JP5954259B2 - 排気ガス浄化用触媒の製造方法 - Google Patents
排気ガス浄化用触媒の製造方法 Download PDFInfo
- Publication number
- JP5954259B2 JP5954259B2 JP2013110769A JP2013110769A JP5954259B2 JP 5954259 B2 JP5954259 B2 JP 5954259B2 JP 2013110769 A JP2013110769 A JP 2013110769A JP 2013110769 A JP2013110769 A JP 2013110769A JP 5954259 B2 JP5954259 B2 JP 5954259B2
- Authority
- JP
- Japan
- Prior art keywords
- catalyst
- layer
- nox
- exhaust gas
- gas
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related
Links
- 239000003054 catalyst Substances 0.000 title claims description 104
- 238000004519 manufacturing process Methods 0.000 title claims description 19
- 239000010410 layer Substances 0.000 claims description 64
- 229910021536 Zeolite Inorganic materials 0.000 claims description 28
- 238000000746 purification Methods 0.000 claims description 28
- 239000010457 zeolite Substances 0.000 claims description 28
- HNPSIPDUKPIQMN-UHFFFAOYSA-N dioxosilane;oxo(oxoalumanyloxy)alumane Chemical compound O=[Si]=O.O=[Al]O[Al]=O HNPSIPDUKPIQMN-UHFFFAOYSA-N 0.000 claims description 26
- 230000003647 oxidation Effects 0.000 claims description 16
- 238000007254 oxidation reaction Methods 0.000 claims description 16
- 239000011232 storage material Substances 0.000 claims description 16
- 229910052751 metal Inorganic materials 0.000 claims description 13
- 239000002184 metal Substances 0.000 claims description 13
- 239000000203 mixture Substances 0.000 claims description 12
- PNEYBMLMFCGWSK-UHFFFAOYSA-N aluminium oxide Inorganic materials [O-2].[O-2].[O-2].[Al+3].[Al+3] PNEYBMLMFCGWSK-UHFFFAOYSA-N 0.000 claims description 10
- 230000003197 catalytic effect Effects 0.000 claims description 8
- 239000002344 surface layer Substances 0.000 claims description 7
- 239000011358 absorbing material Substances 0.000 claims description 5
- 239000011248 coating agent Substances 0.000 claims description 5
- 238000000576 coating method Methods 0.000 claims description 5
- 238000000034 method Methods 0.000 claims description 5
- 229910052788 barium Inorganic materials 0.000 claims description 2
- 229910052712 strontium Inorganic materials 0.000 claims description 2
- 230000001180 sulfating effect Effects 0.000 claims 1
- MWUXSHHQAYIFBG-UHFFFAOYSA-N Nitric oxide Chemical compound O=[N] MWUXSHHQAYIFBG-UHFFFAOYSA-N 0.000 description 244
- 239000007789 gas Substances 0.000 description 93
- 239000000463 material Substances 0.000 description 38
- 239000000843 powder Substances 0.000 description 20
- 239000002002 slurry Substances 0.000 description 17
- 210000004027 cell Anatomy 0.000 description 14
- 230000000052 comparative effect Effects 0.000 description 10
- 239000000446 fuel Substances 0.000 description 10
- 210000002421 cell wall Anatomy 0.000 description 8
- 238000001179 sorption measurement Methods 0.000 description 8
- 230000007423 decrease Effects 0.000 description 6
- 238000005259 measurement Methods 0.000 description 6
- 238000012360 testing method Methods 0.000 description 6
- 239000003463 adsorbent Substances 0.000 description 5
- 239000002612 dispersion medium Substances 0.000 description 5
- 229910052739 hydrogen Inorganic materials 0.000 description 5
- 238000002360 preparation method Methods 0.000 description 5
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 5
- 229910052784 alkaline earth metal Inorganic materials 0.000 description 4
- 238000001354 calcination Methods 0.000 description 4
- 229910002091 carbon monoxide Inorganic materials 0.000 description 4
- 238000006243 chemical reaction Methods 0.000 description 4
- 238000010828 elution Methods 0.000 description 4
- 229910052697 platinum Inorganic materials 0.000 description 4
- 238000003860 storage Methods 0.000 description 4
- GRYLNZFGIOXLOG-UHFFFAOYSA-N Nitric acid Chemical compound O[N+]([O-])=O GRYLNZFGIOXLOG-UHFFFAOYSA-N 0.000 description 3
- 150000001342 alkaline earth metals Chemical class 0.000 description 3
- 239000007864 aqueous solution Substances 0.000 description 3
- 230000000694 effects Effects 0.000 description 3
- -1 first Substances 0.000 description 3
- 238000010438 heat treatment Methods 0.000 description 3
- 229910017604 nitric acid Inorganic materials 0.000 description 3
- 229910052760 oxygen Inorganic materials 0.000 description 3
- 239000012466 permeate Substances 0.000 description 3
- 230000009467 reduction Effects 0.000 description 3
- 229910002651 NO3 Inorganic materials 0.000 description 2
- NHNBFGGVMKEFGY-UHFFFAOYSA-N Nitrate Chemical compound [O-][N+]([O-])=O NHNBFGGVMKEFGY-UHFFFAOYSA-N 0.000 description 2
- 230000032683 aging Effects 0.000 description 2
- 229910052783 alkali metal Inorganic materials 0.000 description 2
- 150000001340 alkali metals Chemical class 0.000 description 2
- 239000011230 binding agent Substances 0.000 description 2
- 239000003638 chemical reducing agent Substances 0.000 description 2
- 238000011278 co-treatment Methods 0.000 description 2
- 229910052878 cordierite Inorganic materials 0.000 description 2
- 230000003247 decreasing effect Effects 0.000 description 2
- JSKIRARMQDRGJZ-UHFFFAOYSA-N dimagnesium dioxido-bis[(1-oxido-3-oxo-2,4,6,8,9-pentaoxa-1,3-disila-5,7-dialuminabicyclo[3.3.1]nonan-7-yl)oxy]silane Chemical compound [Mg++].[Mg++].[O-][Si]([O-])(O[Al]1O[Al]2O[Si](=O)O[Si]([O-])(O1)O2)O[Al]1O[Al]2O[Si](=O)O[Si]([O-])(O1)O2 JSKIRARMQDRGJZ-UHFFFAOYSA-N 0.000 description 2
- 230000003993 interaction Effects 0.000 description 2
- 239000002609 medium Substances 0.000 description 2
- 150000002739 metals Chemical class 0.000 description 2
- 229910052763 palladium Inorganic materials 0.000 description 2
- 239000013618 particulate matter Substances 0.000 description 2
- 229910052703 rhodium Inorganic materials 0.000 description 2
- 239000000243 solution Substances 0.000 description 2
- 238000003756 stirring Methods 0.000 description 2
- 230000019635 sulfation Effects 0.000 description 2
- 238000005670 sulfation reaction Methods 0.000 description 2
- BVKZGUZCCUSVTD-UHFFFAOYSA-L Carbonate Chemical compound [O-]C([O-])=O BVKZGUZCCUSVTD-UHFFFAOYSA-L 0.000 description 1
- VGGSQFUCUMXWEO-UHFFFAOYSA-N Ethene Chemical compound C=C VGGSQFUCUMXWEO-UHFFFAOYSA-N 0.000 description 1
- 239000005977 Ethylene Substances 0.000 description 1
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 1
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 description 1
- 239000002253 acid Substances 0.000 description 1
- 230000002378 acidificating effect Effects 0.000 description 1
- 230000002411 adverse Effects 0.000 description 1
- 229910000323 aluminium silicate Inorganic materials 0.000 description 1
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 1
- 238000006555 catalytic reaction Methods 0.000 description 1
- 230000008859 change Effects 0.000 description 1
- 238000002485 combustion reaction Methods 0.000 description 1
- 239000002131 composite material Substances 0.000 description 1
- 238000003795 desorption Methods 0.000 description 1
- 239000006185 dispersion Substances 0.000 description 1
- 238000011156 evaluation Methods 0.000 description 1
- 238000001704 evaporation Methods 0.000 description 1
- 230000008020 evaporation Effects 0.000 description 1
- 238000010304 firing Methods 0.000 description 1
- 239000011521 glass Substances 0.000 description 1
- 238000000227 grinding Methods 0.000 description 1
- 229930195733 hydrocarbon Natural products 0.000 description 1
- 150000002430 hydrocarbons Chemical class 0.000 description 1
- 239000001257 hydrogen Substances 0.000 description 1
- 238000002156 mixing Methods 0.000 description 1
- IJGRMHOSHXDMSA-UHFFFAOYSA-N nitrogen Substances N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 1
- 229910052757 nitrogen Inorganic materials 0.000 description 1
- QJGQUHMNIGDVPM-UHFFFAOYSA-N nitrogen group Chemical group [N] QJGQUHMNIGDVPM-UHFFFAOYSA-N 0.000 description 1
- TVMXDCGIABBOFY-UHFFFAOYSA-N octane Chemical compound CCCCCCCC TVMXDCGIABBOFY-UHFFFAOYSA-N 0.000 description 1
- 230000001590 oxidative effect Effects 0.000 description 1
- 239000001301 oxygen Substances 0.000 description 1
- 230000035515 penetration Effects 0.000 description 1
- 231100000572 poisoning Toxicity 0.000 description 1
- 230000000607 poisoning effect Effects 0.000 description 1
- 238000012545 processing Methods 0.000 description 1
- 239000000047 product Substances 0.000 description 1
- 230000001737 promoting effect Effects 0.000 description 1
- QQONPFPTGQHPMA-UHFFFAOYSA-N propylene Natural products CC=C QQONPFPTGQHPMA-UHFFFAOYSA-N 0.000 description 1
- 125000004805 propylene group Chemical group [H]C([H])([H])C([H])([*:1])C([H])([H])[*:2] 0.000 description 1
- 238000010298 pulverizing process Methods 0.000 description 1
- 238000010926 purge Methods 0.000 description 1
- 239000007787 solid Substances 0.000 description 1
- 229910052717 sulfur Inorganic materials 0.000 description 1
- 239000011593 sulfur Substances 0.000 description 1
- 230000001629 suppression Effects 0.000 description 1
- 230000008719 thickening Effects 0.000 description 1
Images
Landscapes
- Exhaust Gas Treatment By Means Of Catalyst (AREA)
- Catalysts (AREA)
- Exhaust Gas After Treatment (AREA)
Description
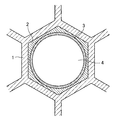
2 DOC(酸化触媒)層
3 LNT(リーンNOxトラップ)層
4 排気ガス通路
Claims (2)
- Ba又はSrよりなるNOx吸蔵材とゼオライトとを含む排気ガス浄化用触媒の製造方法であって、
担体上に、ゼオライト、アルミナ、Ce含有酸化物及び触媒金属を含み且つ前記NOx吸蔵材を添加していない酸化触媒層を形成する工程と、
アルミナとCe含有酸化物との混合物に、前記NOx吸蔵材と触媒金属とを担持する工程と、
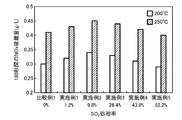
前記担持された混合物にSO 2 ガス処理をすることにより、前記NOx吸蔵材の表層部を硫酸塩化する工程と、
前記SO2ガス処理された混合物をスラリー化し、前記酸化触媒層にコーティングすることによりLNT層を形成する工程と、
前記NOx吸蔵材をCOガスにより炭酸塩化する工程とを備えていることを特徴とする排気ガス浄化用触媒の製造方法。 - 前記担体として、セル断面形状が六角形である六角セルハニカム構造の担体を用いることを特徴とする請求項1に記載の排気ガス浄化用触媒の製造方法。
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2013110769A JP5954259B2 (ja) | 2013-05-27 | 2013-05-27 | 排気ガス浄化用触媒の製造方法 |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2013110769A JP5954259B2 (ja) | 2013-05-27 | 2013-05-27 | 排気ガス浄化用触媒の製造方法 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2014226651A JP2014226651A (ja) | 2014-12-08 |
| JP5954259B2 true JP5954259B2 (ja) | 2016-07-20 |
Family
ID=52126981
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2013110769A Expired - Fee Related JP5954259B2 (ja) | 2013-05-27 | 2013-05-27 | 排気ガス浄化用触媒の製造方法 |
Country Status (1)
| Country | Link |
|---|---|
| JP (1) | JP5954259B2 (ja) |
Families Citing this family (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP6311944B2 (ja) * | 2016-03-28 | 2018-04-18 | 株式会社豊田中央研究所 | ハニカム状触媒 |
| EP3495634A4 (en) * | 2016-08-04 | 2020-03-11 | N.E. Chemcat Corporation | COLD START COMPATIBLE UREA SCR SYSTEM |
| GB2560939A (en) * | 2017-03-29 | 2018-10-03 | Johnson Matthey Plc | NOx Adsorber catalyst |
Family Cites Families (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4335023A (en) * | 1980-01-24 | 1982-06-15 | Engelhard Corporation | Monolithic catalyst member and support therefor |
| JP3799651B2 (ja) * | 1995-04-28 | 2006-07-19 | マツダ株式会社 | 排気ガス浄化用触媒 |
| JP3859940B2 (ja) * | 1999-08-06 | 2006-12-20 | 日産自動車株式会社 | 排気ガス浄化用触媒及びその製造方法 |
| JP2004130269A (ja) * | 2002-10-15 | 2004-04-30 | Nissan Motor Co Ltd | 排気ガス浄化触媒及びその製造方法 |
| JP2004321894A (ja) * | 2003-04-23 | 2004-11-18 | Nissan Motor Co Ltd | 排気ガス浄化触媒及びその製造方法 |
| JP4706784B2 (ja) * | 2008-09-11 | 2011-06-22 | 株式会社デンソー | 六角セルハニカム構造体 |
| JP5785406B2 (ja) * | 2011-03-18 | 2015-09-30 | 日本碍子株式会社 | ハニカム構造体 |
-
2013
- 2013-05-27 JP JP2013110769A patent/JP5954259B2/ja not_active Expired - Fee Related
Also Published As
| Publication number | Publication date |
|---|---|
| JP2014226651A (ja) | 2014-12-08 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6206327B2 (ja) | 排気ガス浄化用触媒及びその製造方法 | |
| KR101868176B1 (ko) | 개선된 no 산화 활성을 갖는 가솔린 린번 엔진용 촉매 | |
| US9242242B2 (en) | Catalyst for gasoline lean burn engines with improved NO oxidation activity | |
| US8950174B2 (en) | Catalysts for gasoline lean burn engines with improved NH3-formation activity | |
| CN102083530B (zh) | 用于净化汽车所排放废气的蜂窝结构型催化剂,其制备方法及使用该催化剂净化废气的方法 | |
| EP3277406B1 (en) | Lean nox trap with enhanced high and low temperature performance | |
| JP6236995B2 (ja) | 排気ガス浄化用触媒及びその製造方法並びにそれを用いた排気ガス浄化方法 | |
| JP2012518531A (ja) | パラジウム担持触媒複合体 | |
| JP7489761B2 (ja) | アンモニア酸化触媒装置 | |
| JP2020515395A (ja) | 三層NOx吸着体触媒 | |
| KR101855537B1 (ko) | Rh 로딩량이 감소된 NOx 저장 촉매 | |
| JP2024164024A (ja) | 超低NOxとコールドスタートのための排気ガス処理システム | |
| JP5391664B2 (ja) | 排気ガス浄化用触媒 | |
| JP5954259B2 (ja) | 排気ガス浄化用触媒の製造方法 | |
| JP5994730B2 (ja) | 排気ガス浄化用触媒の製造方法 | |
| JP5954260B2 (ja) | 排気ガス浄化用触媒及びその製造方法 | |
| JP7682799B2 (ja) | 調整可能なNOx吸着剤 | |
| JP5949662B2 (ja) | 排気ガス浄化用触媒及びその製造方法 | |
| JP6569637B2 (ja) | 内燃機関の排気浄化装置 | |
| JP6102699B2 (ja) | 排気ガス浄化用触媒の製造方法 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20150312 |
|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20160210 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20160216 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20160415 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20160517 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20160530 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 5954259 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| LAPS | Cancellation because of no payment of annual fees |