JP5764792B2 - マクロライド免疫抑制剤を含む新規な医薬組成物 - Google Patents
マクロライド免疫抑制剤を含む新規な医薬組成物 Download PDFInfo
- Publication number
- JP5764792B2 JP5764792B2 JP2012532587A JP2012532587A JP5764792B2 JP 5764792 B2 JP5764792 B2 JP 5764792B2 JP 2012532587 A JP2012532587 A JP 2012532587A JP 2012532587 A JP2012532587 A JP 2012532587A JP 5764792 B2 JP5764792 B2 JP 5764792B2
- Authority
- JP
- Japan
- Prior art keywords
- pharmaceutical composition
- composition according
- tacrolimus
- component
- microemulsion
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 239000008194 pharmaceutical composition Substances 0.000 title claims description 17
- 239000003120 macrolide antibiotic agent Substances 0.000 title description 23
- 239000003018 immunosuppressive agent Substances 0.000 title description 20
- 229960003444 immunosuppressant agent Drugs 0.000 title description 13
- 230000001861 immunosuppressant effect Effects 0.000 title description 6
- 239000000203 mixture Substances 0.000 claims description 90
- QJJXYPPXXYFBGM-LFZNUXCKSA-N Tacrolimus Chemical compound C1C[C@@H](O)[C@H](OC)C[C@@H]1\C=C(/C)[C@@H]1[C@H](C)[C@@H](O)CC(=O)[C@H](CC=C)/C=C(C)/C[C@H](C)C[C@H](OC)[C@H]([C@H](C[C@H]2C)OC)O[C@@]2(O)C(=O)C(=O)N2CCCC[C@H]2C(=O)O1 QJJXYPPXXYFBGM-LFZNUXCKSA-N 0.000 claims description 45
- 239000004530 micro-emulsion Substances 0.000 claims description 43
- 229960001967 tacrolimus Drugs 0.000 claims description 43
- QJJXYPPXXYFBGM-SHYZHZOCSA-N tacrolimus Natural products CO[C@H]1C[C@H](CC[C@@H]1O)C=C(C)[C@H]2OC(=O)[C@H]3CCCCN3C(=O)C(=O)[C@@]4(O)O[C@@H]([C@H](C[C@H]4C)OC)[C@@H](C[C@H](C)CC(=C[C@@H](CC=C)C(=O)C[C@H](O)[C@H]2C)C)OC QJJXYPPXXYFBGM-SHYZHZOCSA-N 0.000 claims description 42
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 22
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 claims description 21
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 claims description 18
- 239000000546 pharmaceutical excipient Substances 0.000 claims description 17
- 239000004094 surface-active agent Substances 0.000 claims description 17
- 239000007788 liquid Substances 0.000 claims description 15
- 201000004681 Psoriasis Diseases 0.000 claims description 11
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 claims description 10
- 238000000034 method Methods 0.000 claims description 10
- 239000003814 drug Substances 0.000 claims description 8
- 210000004400 mucous membrane Anatomy 0.000 claims description 8
- 206010012438 Dermatitis atopic Diseases 0.000 claims description 7
- 201000008937 atopic dermatitis Diseases 0.000 claims description 7
- 229940079593 drug Drugs 0.000 claims description 7
- WCVRQHFDJLLWFE-UHFFFAOYSA-N pentane-1,2-diol Chemical compound CCCC(O)CO WCVRQHFDJLLWFE-UHFFFAOYSA-N 0.000 claims description 7
- 235000014113 dietary fatty acids Nutrition 0.000 claims description 6
- 229930195729 fatty acid Natural products 0.000 claims description 6
- 239000000194 fatty acid Substances 0.000 claims description 6
- 150000004665 fatty acids Chemical class 0.000 claims description 6
- 125000000217 alkyl group Chemical group 0.000 claims description 5
- WGCNASOHLSPBMP-UHFFFAOYSA-N hydroxyacetaldehyde Natural products OCC=O WGCNASOHLSPBMP-UHFFFAOYSA-N 0.000 claims description 5
- 208000027866 inflammatory disease Diseases 0.000 claims description 5
- 238000011200 topical administration Methods 0.000 claims description 5
- XTJFFFGAUHQWII-UHFFFAOYSA-N Dibutyl adipate Chemical compound CCCCOC(=O)CCCCC(=O)OCCCC XTJFFFGAUHQWII-UHFFFAOYSA-N 0.000 claims description 4
- 239000003795 chemical substances by application Substances 0.000 claims description 4
- 229940100539 dibutyl adipate Drugs 0.000 claims description 4
- 150000003904 phospholipids Chemical class 0.000 claims description 4
- JNYAEWCLZODPBN-JGWLITMVSA-N (2r,3r,4s)-2-[(1r)-1,2-dihydroxyethyl]oxolane-3,4-diol Chemical class OC[C@@H](O)[C@H]1OC[C@H](O)[C@H]1O JNYAEWCLZODPBN-JGWLITMVSA-N 0.000 claims description 3
- HBTAOSGHCXUEKI-UHFFFAOYSA-N 4-chloro-n,n-dimethyl-3-nitrobenzenesulfonamide Chemical compound CN(C)S(=O)(=O)C1=CC=C(Cl)C([N+]([O-])=O)=C1 HBTAOSGHCXUEKI-UHFFFAOYSA-N 0.000 claims description 3
- 208000027932 Collagen disease Diseases 0.000 claims description 3
- 239000002202 Polyethylene glycol Substances 0.000 claims description 3
- 150000005690 diesters Chemical class 0.000 claims description 3
- 229940031578 diisopropyl adipate Drugs 0.000 claims description 3
- 150000002191 fatty alcohols Chemical class 0.000 claims description 3
- 238000000265 homogenisation Methods 0.000 claims description 3
- 229940074928 isopropyl myristate Drugs 0.000 claims description 3
- XUGNVMKQXJXZCD-UHFFFAOYSA-N isopropyl palmitate Chemical compound CCCCCCCCCCCCCCCC(=O)OC(C)C XUGNVMKQXJXZCD-UHFFFAOYSA-N 0.000 claims description 3
- 229940075495 isopropyl palmitate Drugs 0.000 claims description 3
- 229920001223 polyethylene glycol Polymers 0.000 claims description 3
- HIQIXEFWDLTDED-UHFFFAOYSA-N 4-hydroxy-1-piperidin-4-ylpyrrolidin-2-one Chemical compound O=C1CC(O)CN1C1CCNCC1 HIQIXEFWDLTDED-UHFFFAOYSA-N 0.000 claims description 2
- 229920001521 polyalkylene glycol ether Polymers 0.000 claims description 2
- UFTFJSFQGQCHQW-UHFFFAOYSA-N triformin Chemical compound O=COCC(OC=O)COC=O UFTFJSFQGQCHQW-UHFFFAOYSA-N 0.000 claims description 2
- 210000003491 skin Anatomy 0.000 description 35
- 238000009472 formulation Methods 0.000 description 25
- 239000004480 active ingredient Substances 0.000 description 15
- 238000011282 treatment Methods 0.000 description 14
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 12
- 150000001875 compounds Chemical class 0.000 description 12
- 239000000839 emulsion Substances 0.000 description 10
- -1 liquid paraffin Substances 0.000 description 10
- 239000012071 phase Substances 0.000 description 10
- 230000000699 topical effect Effects 0.000 description 10
- 239000004615 ingredient Substances 0.000 description 9
- 235000013601 eggs Nutrition 0.000 description 7
- 229940125721 immunosuppressive agent Drugs 0.000 description 7
- 230000001225 therapeutic effect Effects 0.000 description 7
- PMATZTZNYRCHOR-CGLBZJNRSA-N Cyclosporin A Chemical compound CC[C@@H]1NC(=O)[C@H]([C@H](O)[C@H](C)C\C=C\C)N(C)C(=O)[C@H](C(C)C)N(C)C(=O)[C@H](CC(C)C)N(C)C(=O)[C@H](CC(C)C)N(C)C(=O)[C@@H](C)NC(=O)[C@H](C)NC(=O)[C@H](CC(C)C)N(C)C(=O)[C@H](C(C)C)NC(=O)[C@H](CC(C)C)N(C)C(=O)CN(C)C1=O PMATZTZNYRCHOR-CGLBZJNRSA-N 0.000 description 6
- 108010036949 Cyclosporine Proteins 0.000 description 6
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 6
- 229960001265 ciclosporin Drugs 0.000 description 6
- 229930182912 cyclosporin Natural products 0.000 description 6
- 229940043375 1,5-pentanediol Drugs 0.000 description 5
- HKVAMNSJSFKALM-GKUWKFKPSA-N Everolimus Chemical compound C1C[C@@H](OCCO)[C@H](OC)C[C@@H]1C[C@@H](C)[C@H]1OC(=O)[C@@H]2CCCCN2C(=O)C(=O)[C@](O)(O2)[C@H](C)CC[C@H]2C[C@H](OC)/C(C)=C/C=C/C=C/[C@@H](C)C[C@@H](C)C(=O)[C@H](OC)[C@H](O)/C(C)=C/[C@@H](C)C(=O)C1 HKVAMNSJSFKALM-GKUWKFKPSA-N 0.000 description 5
- 230000008901 benefit Effects 0.000 description 5
- 150000002009 diols Chemical class 0.000 description 5
- 229960005167 everolimus Drugs 0.000 description 5
- 238000002156 mixing Methods 0.000 description 5
- 230000035515 penetration Effects 0.000 description 5
- IIZPXYDJLKNOIY-JXPKJXOSSA-N 1-palmitoyl-2-arachidonoyl-sn-glycero-3-phosphocholine Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCC\C=C/C\C=C/C\C=C/C\C=C/CCCCC IIZPXYDJLKNOIY-JXPKJXOSSA-N 0.000 description 4
- 229920003171 Poly (ethylene oxide) Polymers 0.000 description 4
- 239000008186 active pharmaceutical agent Substances 0.000 description 4
- 230000015572 biosynthetic process Effects 0.000 description 4
- 210000004027 cell Anatomy 0.000 description 4
- 210000004207 dermis Anatomy 0.000 description 4
- 238000009792 diffusion process Methods 0.000 description 4
- 230000000694 effects Effects 0.000 description 4
- 238000002474 experimental method Methods 0.000 description 4
- 150000002334 glycols Chemical class 0.000 description 4
- 210000000987 immune system Anatomy 0.000 description 4
- 230000001506 immunosuppresive effect Effects 0.000 description 4
- 239000000787 lecithin Substances 0.000 description 4
- 229940067606 lecithin Drugs 0.000 description 4
- 235000010445 lecithin Nutrition 0.000 description 4
- 210000004877 mucosa Anatomy 0.000 description 4
- 210000000056 organ Anatomy 0.000 description 4
- 230000010412 perfusion Effects 0.000 description 4
- ZAHRKKWIAAJSAO-UHFFFAOYSA-N rapamycin Natural products COCC(O)C(=C/C(C)C(=O)CC(OC(=O)C1CCCCN1C(=O)C(=O)C2(O)OC(CC(OC)C(=CC=CC=CC(C)CC(C)C(=O)C)C)CCC2C)C(C)CC3CCC(O)C(C3)OC)C ZAHRKKWIAAJSAO-UHFFFAOYSA-N 0.000 description 4
- QFJCIRLUMZQUOT-HPLJOQBZSA-N sirolimus Chemical compound C1C[C@@H](O)[C@H](OC)C[C@@H]1C[C@@H](C)[C@H]1OC(=O)[C@@H]2CCCCN2C(=O)C(=O)[C@](O)(O2)[C@H](C)CC[C@H]2C[C@H](OC)/C(C)=C/C=C/C=C/[C@@H](C)C[C@@H](C)C(=O)[C@H](OC)[C@H](O)/C(C)=C/[C@@H](C)C(=O)C1 QFJCIRLUMZQUOT-HPLJOQBZSA-N 0.000 description 4
- 229960002930 sirolimus Drugs 0.000 description 4
- 208000023275 Autoimmune disease Diseases 0.000 description 3
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 3
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 3
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 3
- 206010052779 Transplant rejections Diseases 0.000 description 3
- 230000001413 cellular effect Effects 0.000 description 3
- 201000010099 disease Diseases 0.000 description 3
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 3
- 229940088679 drug related substance Drugs 0.000 description 3
- BXWNKGSJHAJOGX-UHFFFAOYSA-N n-hexadecyl alcohol Natural products CCCCCCCCCCCCCCCCO BXWNKGSJHAJOGX-UHFFFAOYSA-N 0.000 description 3
- 239000002245 particle Substances 0.000 description 3
- KASDHRXLYQOAKZ-ZPSXYTITSA-N pimecrolimus Chemical compound C/C([C@H]1OC(=O)[C@@H]2CCCCN2C(=O)C(=O)[C@]2(O)O[C@@H]([C@H](C[C@H]2C)OC)[C@@H](OC)C[C@@H](C)C/C(C)=C/[C@H](C(C[C@H](O)[C@H]1C)=O)CC)=C\[C@@H]1CC[C@@H](Cl)[C@H](OC)C1 KASDHRXLYQOAKZ-ZPSXYTITSA-N 0.000 description 3
- 229960005330 pimecrolimus Drugs 0.000 description 3
- 239000002798 polar solvent Substances 0.000 description 3
- 238000002360 preparation method Methods 0.000 description 3
- 239000003755 preservative agent Substances 0.000 description 3
- 230000008569 process Effects 0.000 description 3
- 239000000047 product Substances 0.000 description 3
- 210000000434 stratum corneum Anatomy 0.000 description 3
- 239000000126 substance Substances 0.000 description 3
- 208000024891 symptom Diseases 0.000 description 3
- 230000009885 systemic effect Effects 0.000 description 3
- 210000001519 tissue Anatomy 0.000 description 3
- GVJHHUAWPYXKBD-UHFFFAOYSA-N (±)-α-Tocopherol Chemical compound OC1=C(C)C(C)=C2OC(CCCC(C)CCCC(C)CCCC(C)C)(C)CCC2=C1C GVJHHUAWPYXKBD-UHFFFAOYSA-N 0.000 description 2
- ZORQXIQZAOLNGE-UHFFFAOYSA-N 1,1-difluorocyclohexane Chemical compound FC1(F)CCCCC1 ZORQXIQZAOLNGE-UHFFFAOYSA-N 0.000 description 2
- KBPLFHHGFOOTCA-UHFFFAOYSA-N 1-Octanol Chemical compound CCCCCCCCO KBPLFHHGFOOTCA-UHFFFAOYSA-N 0.000 description 2
- RFVNOJDQRGSOEL-UHFFFAOYSA-N 2-hydroxyethyl octadecanoate Chemical compound CCCCCCCCCCCCCCCCCC(=O)OCCO RFVNOJDQRGSOEL-UHFFFAOYSA-N 0.000 description 2
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 2
- CIWBSHSKHKDKBQ-JLAZNSOCSA-N Ascorbic acid Chemical compound OC[C@H](O)[C@H]1OC(=O)C(O)=C1O CIWBSHSKHKDKBQ-JLAZNSOCSA-N 0.000 description 2
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 description 2
- FBOZXECLQNJBKD-ZDUSSCGKSA-N L-methotrexate Chemical compound C=1N=C2N=C(N)N=C(N)C2=NC=1CN(C)C1=CC=C(C(=O)N[C@@H](CCC(O)=O)C(O)=O)C=C1 FBOZXECLQNJBKD-ZDUSSCGKSA-N 0.000 description 2
- CBPNZQVSJQDFBE-FUXHJELOSA-N Temsirolimus Chemical compound C1C[C@@H](OC(=O)C(C)(CO)CO)[C@H](OC)C[C@@H]1C[C@@H](C)[C@H]1OC(=O)[C@@H]2CCCCN2C(=O)C(=O)[C@](O)(O2)[C@H](C)CC[C@H]2C[C@H](OC)/C(C)=C/C=C/C=C/[C@@H](C)C[C@@H](C)C(=O)[C@H](OC)[C@H](O)/C(C)=C/[C@@H](C)C(=O)C1 CBPNZQVSJQDFBE-FUXHJELOSA-N 0.000 description 2
- 239000002253 acid Substances 0.000 description 2
- 239000008346 aqueous phase Substances 0.000 description 2
- 230000004888 barrier function Effects 0.000 description 2
- 230000009286 beneficial effect Effects 0.000 description 2
- 238000001574 biopsy Methods 0.000 description 2
- 239000000872 buffer Substances 0.000 description 2
- 239000003093 cationic surfactant Substances 0.000 description 2
- 229960000541 cetyl alcohol Drugs 0.000 description 2
- 239000012141 concentrate Substances 0.000 description 2
- 239000002537 cosmetic Substances 0.000 description 2
- 230000000968 intestinal effect Effects 0.000 description 2
- 238000001990 intravenous administration Methods 0.000 description 2
- 230000007794 irritation Effects 0.000 description 2
- 150000002596 lactones Chemical group 0.000 description 2
- 239000002502 liposome Substances 0.000 description 2
- 229940057995 liquid paraffin Drugs 0.000 description 2
- 230000010534 mechanism of action Effects 0.000 description 2
- 210000004379 membrane Anatomy 0.000 description 2
- 239000012528 membrane Substances 0.000 description 2
- 239000002674 ointment Substances 0.000 description 2
- 239000008363 phosphate buffer Substances 0.000 description 2
- 229940068196 placebo Drugs 0.000 description 2
- 239000000902 placebo Substances 0.000 description 2
- 230000002265 prevention Effects 0.000 description 2
- 229940112971 protopic Drugs 0.000 description 2
- 239000007787 solid Substances 0.000 description 2
- 239000000243 solution Substances 0.000 description 2
- 229940035049 sorbitan monooleate Drugs 0.000 description 2
- 239000001593 sorbitan monooleate Substances 0.000 description 2
- 235000011069 sorbitan monooleate Nutrition 0.000 description 2
- 230000000638 stimulation Effects 0.000 description 2
- 229960000235 temsirolimus Drugs 0.000 description 2
- QFJCIRLUMZQUOT-UHFFFAOYSA-N temsirolimus Natural products C1CC(O)C(OC)CC1CC(C)C1OC(=O)C2CCCCN2C(=O)C(=O)C(O)(O2)C(C)CCC2CC(OC)C(C)=CC=CC=CC(C)CC(C)C(=O)C(OC)C(O)C(C)=CC(C)C(=O)C1 QFJCIRLUMZQUOT-UHFFFAOYSA-N 0.000 description 2
- 238000012360 testing method Methods 0.000 description 2
- 239000012049 topical pharmaceutical composition Substances 0.000 description 2
- 230000002792 vascular Effects 0.000 description 2
- 210000005166 vasculature Anatomy 0.000 description 2
- 239000001993 wax Substances 0.000 description 2
- CGTADGCBEXYWNE-JUKNQOCSSA-N zotarolimus Chemical compound N1([C@H]2CC[C@@H](C[C@@H](C)[C@H]3OC(=O)[C@@H]4CCCCN4C(=O)C(=O)[C@@]4(O)[C@H](C)CC[C@H](O4)C[C@@H](/C(C)=C/C=C/C=C/[C@@H](C)C[C@@H](C)C(=O)[C@H](OC)[C@H](O)/C(C)=C/[C@@H](C)C(=O)C3)OC)C[C@H]2OC)C=NN=N1 CGTADGCBEXYWNE-JUKNQOCSSA-N 0.000 description 2
- 229950009819 zotarolimus Drugs 0.000 description 2
- NWJQLQGQZSIBAF-MSLXHMNKSA-N (1R,9S,12S,13R,14S,17R,18Z,21S,23S,24R,25S,27R)-1,14-dihydroxy-12-[(E)-1-[(1R,3R,4R)-4-hydroxy-3-methoxycyclohexyl]prop-1-en-2-yl]-23,25-dimethoxy-13,19,21,27-tetramethyl-17-prop-2-enyl-11,28-dioxa-4-azatricyclo[22.3.1.04,9]octacos-18-ene-2,3,10,16-tetrone hydrate Chemical group O.C1C[C@@H](O)[C@H](OC)C[C@@H]1\C=C(/C)[C@@H]1[C@H](C)[C@@H](O)CC(=O)[C@H](CC=C)\C=C(C)/C[C@H](C)C[C@H](OC)[C@H]([C@H](C[C@H]2C)OC)O[C@@]2(O)C(=O)C(=O)N2CCCC[C@H]2C(=O)O1 NWJQLQGQZSIBAF-MSLXHMNKSA-N 0.000 description 1
- CMCBDXRRFKYBDG-UHFFFAOYSA-N 1-dodecoxydodecane Chemical compound CCCCCCCCCCCCOCCCCCCCCCCCC CMCBDXRRFKYBDG-UHFFFAOYSA-N 0.000 description 1
- LXAHHHIGZXPRKQ-UHFFFAOYSA-N 5-fluoro-2-methylpyridine Chemical compound CC1=CC=C(F)C=N1 LXAHHHIGZXPRKQ-UHFFFAOYSA-N 0.000 description 1
- BUROJSBIWGDYCN-GAUTUEMISA-N AP 23573 Chemical compound C1C[C@@H](OP(C)(C)=O)[C@H](OC)C[C@@H]1C[C@@H](C)[C@H]1OC(=O)[C@@H]2CCCCN2C(=O)C(=O)[C@](O)(O2)[C@H](C)CC[C@H]2C[C@H](OC)/C(C)=C/C=C/C=C/[C@@H](C)C[C@@H](C)C(=O)[C@H](OC)[C@H](O)/C(C)=C/[C@@H](C)C(=O)C1 BUROJSBIWGDYCN-GAUTUEMISA-N 0.000 description 1
- 208000026872 Addison Disease Diseases 0.000 description 1
- 206010009900 Colitis ulcerative Diseases 0.000 description 1
- 208000011231 Crohn disease Diseases 0.000 description 1
- 102000004127 Cytokines Human genes 0.000 description 1
- 108090000695 Cytokines Proteins 0.000 description 1
- JDRSMPFHFNXQRB-CMTNHCDUSA-N Decyl beta-D-threo-hexopyranoside Chemical compound CCCCCCCCCCO[C@@H]1O[C@H](CO)C(O)[C@H](O)C1O JDRSMPFHFNXQRB-CMTNHCDUSA-N 0.000 description 1
- 206010012442 Dermatitis contact Diseases 0.000 description 1
- 208000003556 Dry Eye Syndromes Diseases 0.000 description 1
- 206010013774 Dry eye Diseases 0.000 description 1
- 102000002322 Egg Proteins Human genes 0.000 description 1
- 108010000912 Egg Proteins Proteins 0.000 description 1
- 108010008165 Etanercept Proteins 0.000 description 1
- 206010020751 Hypersensitivity Diseases 0.000 description 1
- 206010061218 Inflammation Diseases 0.000 description 1
- 102000000589 Interleukin-1 Human genes 0.000 description 1
- 108010002352 Interleukin-1 Proteins 0.000 description 1
- 102000000588 Interleukin-2 Human genes 0.000 description 1
- 108010002350 Interleukin-2 Proteins 0.000 description 1
- 241001465754 Metazoa Species 0.000 description 1
- 239000004264 Petrolatum Substances 0.000 description 1
- DBMJMQXJHONAFJ-UHFFFAOYSA-M Sodium laurylsulphate Chemical compound [Na+].CCCCCCCCCCCCOS([O-])(=O)=O DBMJMQXJHONAFJ-UHFFFAOYSA-M 0.000 description 1
- 241001647839 Streptomyces tsukubensis Species 0.000 description 1
- 201000006704 Ulcerative Colitis Diseases 0.000 description 1
- 229930003427 Vitamin E Natural products 0.000 description 1
- 206010052428 Wound Diseases 0.000 description 1
- 208000027418 Wounds and injury Diseases 0.000 description 1
- ATBOMIWRCZXYSZ-XZBBILGWSA-N [1-[2,3-dihydroxypropoxy(hydroxy)phosphoryl]oxy-3-hexadecanoyloxypropan-2-yl] (9e,12e)-octadeca-9,12-dienoate Chemical compound CCCCCCCCCCCCCCCC(=O)OCC(COP(O)(=O)OCC(O)CO)OC(=O)CCCCCCC\C=C\C\C=C\CCCCC ATBOMIWRCZXYSZ-XZBBILGWSA-N 0.000 description 1
- 229960003697 abatacept Drugs 0.000 description 1
- 150000007513 acids Chemical class 0.000 description 1
- 230000009471 action Effects 0.000 description 1
- 150000001298 alcohols Chemical class 0.000 description 1
- 208000026935 allergic disease Diseases 0.000 description 1
- AWUCVROLDVIAJX-UHFFFAOYSA-N alpha-glycerophosphate Natural products OCC(O)COP(O)(O)=O AWUCVROLDVIAJX-UHFFFAOYSA-N 0.000 description 1
- 238000010171 animal model Methods 0.000 description 1
- 239000003242 anti bacterial agent Substances 0.000 description 1
- 230000000844 anti-bacterial effect Effects 0.000 description 1
- 230000000340 anti-metabolite Effects 0.000 description 1
- 229940088710 antibiotic agent Drugs 0.000 description 1
- 229940121375 antifungal agent Drugs 0.000 description 1
- 239000000427 antigen Substances 0.000 description 1
- 108091007433 antigens Proteins 0.000 description 1
- 102000036639 antigens Human genes 0.000 description 1
- 229940100197 antimetabolite Drugs 0.000 description 1
- 239000002256 antimetabolite Substances 0.000 description 1
- 239000003963 antioxidant agent Substances 0.000 description 1
- 235000006708 antioxidants Nutrition 0.000 description 1
- 239000003443 antiviral agent Substances 0.000 description 1
- 239000007864 aqueous solution Substances 0.000 description 1
- 235000010323 ascorbic acid Nutrition 0.000 description 1
- 229960005070 ascorbic acid Drugs 0.000 description 1
- 239000011668 ascorbic acid Substances 0.000 description 1
- 230000001363 autoimmune Effects 0.000 description 1
- 229960002170 azathioprine Drugs 0.000 description 1
- LMEKQMALGUDUQG-UHFFFAOYSA-N azathioprine Chemical compound CN1C=NC([N+]([O-])=O)=C1SC1=NC=NC2=C1NC=N2 LMEKQMALGUDUQG-UHFFFAOYSA-N 0.000 description 1
- 239000002585 base Substances 0.000 description 1
- 229960000686 benzalkonium chloride Drugs 0.000 description 1
- CADWTSSKOVRVJC-UHFFFAOYSA-N benzyl(dimethyl)azanium;chloride Chemical compound [Cl-].C[NH+](C)CC1=CC=CC=C1 CADWTSSKOVRVJC-UHFFFAOYSA-N 0.000 description 1
- 210000000481 breast Anatomy 0.000 description 1
- 239000000337 buffer salt Substances 0.000 description 1
- 239000002775 capsule Substances 0.000 description 1
- 238000006243 chemical reaction Methods 0.000 description 1
- 230000001684 chronic effect Effects 0.000 description 1
- 208000025302 chronic primary adrenal insufficiency Diseases 0.000 description 1
- 239000000470 constituent Substances 0.000 description 1
- 208000010247 contact dermatitis Diseases 0.000 description 1
- 239000003246 corticosteroid Substances 0.000 description 1
- 229960001334 corticosteroids Drugs 0.000 description 1
- 238000009109 curative therapy Methods 0.000 description 1
- 229940073499 decyl glucoside Drugs 0.000 description 1
- 150000008266 deoxy sugars Chemical group 0.000 description 1
- 238000001514 detection method Methods 0.000 description 1
- XXJWXESWEXIICW-UHFFFAOYSA-N diethylene glycol monoethyl ether Chemical compound CCOCCOCCO XXJWXESWEXIICW-UHFFFAOYSA-N 0.000 description 1
- 238000010790 dilution Methods 0.000 description 1
- 239000012895 dilution Substances 0.000 description 1
- 125000003438 dodecyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 238000012377 drug delivery Methods 0.000 description 1
- 230000002500 effect on skin Effects 0.000 description 1
- 210000003278 egg shell Anatomy 0.000 description 1
- 238000004945 emulsification Methods 0.000 description 1
- 239000003995 emulsifying agent Substances 0.000 description 1
- 239000003623 enhancer Substances 0.000 description 1
- 230000002708 enhancing effect Effects 0.000 description 1
- 210000002615 epidermis Anatomy 0.000 description 1
- 229960000403 etanercept Drugs 0.000 description 1
- BEFDCLMNVWHSGT-UHFFFAOYSA-N ethenylcyclopentane Chemical compound C=CC1CCCC1 BEFDCLMNVWHSGT-UHFFFAOYSA-N 0.000 description 1
- 230000008020 evaporation Effects 0.000 description 1
- 238000001704 evaporation Methods 0.000 description 1
- 210000001508 eye Anatomy 0.000 description 1
- 230000001815 facial effect Effects 0.000 description 1
- 230000004720 fertilization Effects 0.000 description 1
- 239000012530 fluid Substances 0.000 description 1
- 239000004052 folic acid antagonist Substances 0.000 description 1
- 230000037406 food intake Effects 0.000 description 1
- 108020001507 fusion proteins Proteins 0.000 description 1
- 102000037865 fusion proteins Human genes 0.000 description 1
- WIGCFUFOHFEKBI-UHFFFAOYSA-N gamma-tocopherol Natural products CC(C)CCCC(C)CCCC(C)CCCC1CCC2C(C)C(O)C(C)C(C)C2O1 WIGCFUFOHFEKBI-UHFFFAOYSA-N 0.000 description 1
- 102000054766 genetic haplotypes Human genes 0.000 description 1
- 239000011521 glass Substances 0.000 description 1
- 229930182478 glucoside Natural products 0.000 description 1
- 150000008131 glucosides Chemical class 0.000 description 1
- 239000007902 hard capsule Substances 0.000 description 1
- 210000002216 heart Anatomy 0.000 description 1
- CPBQJMYROZQQJC-UHFFFAOYSA-N helium neon Chemical compound [He].[Ne] CPBQJMYROZQQJC-UHFFFAOYSA-N 0.000 description 1
- 210000002443 helper t lymphocyte Anatomy 0.000 description 1
- 238000004128 high performance liquid chromatography Methods 0.000 description 1
- 230000002209 hydrophobic effect Effects 0.000 description 1
- 230000009610 hypersensitivity Effects 0.000 description 1
- 230000028993 immune response Effects 0.000 description 1
- 238000000338 in vitro Methods 0.000 description 1
- 238000001727 in vivo Methods 0.000 description 1
- 230000004054 inflammatory process Effects 0.000 description 1
- 229960000598 infliximab Drugs 0.000 description 1
- 238000001802 infusion Methods 0.000 description 1
- 239000003112 inhibitor Substances 0.000 description 1
- 230000002401 inhibitory effect Effects 0.000 description 1
- 210000003734 kidney Anatomy 0.000 description 1
- 210000002429 large intestine Anatomy 0.000 description 1
- 150000002605 large molecules Chemical class 0.000 description 1
- 210000000265 leukocyte Anatomy 0.000 description 1
- 230000000670 limiting effect Effects 0.000 description 1
- 210000004185 liver Anatomy 0.000 description 1
- 210000004698 lymphocyte Anatomy 0.000 description 1
- 229960003511 macrogol Drugs 0.000 description 1
- 229940041033 macrolides Drugs 0.000 description 1
- 229920002521 macromolecule Polymers 0.000 description 1
- 210000002540 macrophage Anatomy 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- 238000002844 melting Methods 0.000 description 1
- 230000008018 melting Effects 0.000 description 1
- 229910052751 metal Inorganic materials 0.000 description 1
- 239000002184 metal Substances 0.000 description 1
- 229960000485 methotrexate Drugs 0.000 description 1
- 238000012283 microsurgical operation Methods 0.000 description 1
- 210000000214 mouth Anatomy 0.000 description 1
- 201000006417 multiple sclerosis Diseases 0.000 description 1
- RTGDFNSFWBGLEC-SYZQJQIISA-N mycophenolate mofetil Chemical compound COC1=C(C)C=2COC(=O)C=2C(O)=C1C\C=C(/C)CCC(=O)OCCN1CCOCC1 RTGDFNSFWBGLEC-SYZQJQIISA-N 0.000 description 1
- 229960004866 mycophenolate mofetil Drugs 0.000 description 1
- GOQYKNQRPGWPLP-UHFFFAOYSA-N n-heptadecyl alcohol Natural products CCCCCCCCCCCCCCCCCO GOQYKNQRPGWPLP-UHFFFAOYSA-N 0.000 description 1
- GLDOVTGHNKAZLK-UHFFFAOYSA-N n-octadecyl alcohol Natural products CCCCCCCCCCCCCCCCCCO GLDOVTGHNKAZLK-UHFFFAOYSA-N 0.000 description 1
- 210000003928 nasal cavity Anatomy 0.000 description 1
- 239000013642 negative control Substances 0.000 description 1
- 230000001537 neural effect Effects 0.000 description 1
- 239000003921 oil Substances 0.000 description 1
- 239000003960 organic solvent Substances 0.000 description 1
- 125000000913 palmityl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 239000012188 paraffin wax Substances 0.000 description 1
- 238000005192 partition Methods 0.000 description 1
- 229940066842 petrolatum Drugs 0.000 description 1
- 235000019271 petrolatum Nutrition 0.000 description 1
- WVDDGKGOMKODPV-ZQBYOMGUSA-N phenyl(114C)methanol Chemical compound O[14CH2]C1=CC=CC=C1 WVDDGKGOMKODPV-ZQBYOMGUSA-N 0.000 description 1
- 230000036470 plasma concentration Effects 0.000 description 1
- 239000013641 positive control Substances 0.000 description 1
- 230000002335 preservative effect Effects 0.000 description 1
- 229940072288 prograf Drugs 0.000 description 1
- 238000011321 prophylaxis Methods 0.000 description 1
- RUOJZAUFBMNUDX-UHFFFAOYSA-N propylene carbonate Chemical compound CC1COC(=O)O1 RUOJZAUFBMNUDX-UHFFFAOYSA-N 0.000 description 1
- 108090000623 proteins and genes Proteins 0.000 description 1
- 239000008349 purified phosphatidyl choline Substances 0.000 description 1
- 230000006825 purine synthesis Effects 0.000 description 1
- 108020003175 receptors Proteins 0.000 description 1
- 230000002829 reductive effect Effects 0.000 description 1
- 206010039073 rheumatoid arthritis Diseases 0.000 description 1
- 229960001302 ridaforolimus Drugs 0.000 description 1
- 229960004641 rituximab Drugs 0.000 description 1
- 150000003839 salts Chemical class 0.000 description 1
- 210000000813 small intestine Anatomy 0.000 description 1
- 230000007928 solubilization Effects 0.000 description 1
- 238000005063 solubilization Methods 0.000 description 1
- 230000003381 solubilizing effect Effects 0.000 description 1
- 239000012453 solvate Substances 0.000 description 1
- 238000000527 sonication Methods 0.000 description 1
- 239000004334 sorbic acid Substances 0.000 description 1
- 235000010199 sorbic acid Nutrition 0.000 description 1
- 229940075582 sorbic acid Drugs 0.000 description 1
- 125000004079 stearyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 238000003860 storage Methods 0.000 description 1
- 239000008348 synthetic phosphatidyl choline Substances 0.000 description 1
- 238000009121 systemic therapy Methods 0.000 description 1
- 229960001569 tacrolimus monohydrate Drugs 0.000 description 1
- 239000012085 test solution Substances 0.000 description 1
- 229960003989 tocilizumab Drugs 0.000 description 1
- 231100000331 toxic Toxicity 0.000 description 1
- 230000002588 toxic effect Effects 0.000 description 1
- 231100000820 toxicity test Toxicity 0.000 description 1
- 238000013518 transcription Methods 0.000 description 1
- 230000035897 transcription Effects 0.000 description 1
- 238000002054 transplantation Methods 0.000 description 1
- 150000003626 triacylglycerols Chemical class 0.000 description 1
- PHYFQTYBJUILEZ-IUPFWZBJSA-N triolein Chemical compound CCCCCCCC\C=C/CCCCCCCC(=O)OCC(OC(=O)CCCCCCC\C=C/CCCCCCCC)COC(=O)CCCCCCC\C=C/CCCCCCCC PHYFQTYBJUILEZ-IUPFWZBJSA-N 0.000 description 1
- 235000019165 vitamin E Nutrition 0.000 description 1
- 229940046009 vitamin E Drugs 0.000 description 1
- 239000011709 vitamin E Substances 0.000 description 1
- 230000037303 wrinkles Effects 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/4353—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom ortho- or peri-condensed with heterocyclic ring systems
- A61K31/436—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom ortho- or peri-condensed with heterocyclic ring systems the heterocyclic ring system containing a six-membered ring having oxygen as a ring hetero atom, e.g. rapamycin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/0014—Skin, i.e. galenical aspects of topical compositions
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/0048—Eye, e.g. artificial tears
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/10—Dispersions; Emulsions
- A61K9/107—Emulsions ; Emulsion preconcentrates; Micelles
- A61K9/1075—Microemulsions or submicron emulsions; Preconcentrates or solids thereof; Micelles, e.g. made of phospholipids or block copolymers
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
- A61P17/06—Antipsoriatics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/04—Drugs for skeletal disorders for non-specific disorders of the connective tissue
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P27/00—Drugs for disorders of the senses
- A61P27/02—Ophthalmic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/02—Immunomodulators
- A61P37/06—Immunosuppressants, e.g. drugs for graft rejection
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/08—Antiallergic agents
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N1/00—Sampling; Preparing specimens for investigation
- G01N1/28—Preparing specimens for investigation including physical details of (bio-)chemical methods covered elsewhere, e.g. G01N33/50, C12Q
- G01N1/40—Concentrating samples
- G01N1/4055—Concentrating samples by solubility techniques
- G01N2001/4061—Solvent extraction
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Pharmacology & Pharmacy (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- General Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Animal Behavior & Ethology (AREA)
- Epidemiology (AREA)
- Engineering & Computer Science (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Organic Chemistry (AREA)
- Dermatology (AREA)
- Immunology (AREA)
- Ophthalmology & Optometry (AREA)
- Molecular Biology (AREA)
- Biophysics (AREA)
- Dispersion Chemistry (AREA)
- Rheumatology (AREA)
- Pain & Pain Management (AREA)
- Pulmonology (AREA)
- Transplantation (AREA)
- Biomedical Technology (AREA)
- Physical Education & Sports Medicine (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Medicinal Preparation (AREA)
Description
それぞれの成分を組み合わせ、混合することによって、本発明による4種の異なるマイクロエマルション組成物を製剤化し、調製した。組成を表1から7に示す。混合すると、透明またはわずかに乳白色のマイクロエマルションが自発的に形成された。室温で3カ月保存した後、3種のマイクロエマルションのサンプルを目視によって検査した。物理的不安定性の兆候は認められなかった。
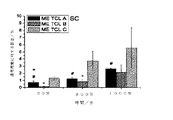
実施例1に従って、ただし活性成分を用いずに調製した3種の製剤A、B、およびCを、皮膚および粘膜に毒性組織作用をもたらす可能性に関して、HET CAM(「鶏卵試験−漿尿膜」)法を用いて試験した。この膜は、受精後8〜10日で、すでに機能的血管系を有するが、神経組織を有さないときに用いる。活性成分ではなく、担体の刺激可能性を判定するために、プラセボ製剤を用いた。
実施例1に従って調製した3種のタクロリムスマイクロエマルションA、B、およびCを、切除したヒト乳房皮膚およびフランツ型拡散セル(Crowne Glass Company、Somerville、NY、USA)を用いて、皮膚透過実験でさらに試験した。リン酸緩衝液pH7.4を受容液として用いた。金属クリップを用いて、皮膚サンプルをフィルタの上に固定した。実験中、蒸発による損失を回避するために、フランツ型セルをガラスの蓋で覆った。一連の試験で3人のドナーの皮膚を用いた。
Claims (11)
- −タクロリムス、
−親水性成分、
−親油性成分、および
−両親媒性成分を含み、
前記親水性成分が、水と、グリセロール、プロピレングリコール、1,2−ペンチレングリコール、およびポリエチレングリコールから選択される液体グリコールとの組み合わせからなり、前記水と液体グリコールの比率が、1:10から10:1の範囲である、
マイクロエマルションとして製剤化されている局所投与用医薬組成物。 - 必須的にすべてのタクロリムスが可溶化形態である、請求項1に記載の医薬組成物。
- マイクロエマルションが液体形態である、請求項1または2のいずれかに記載の医薬組成物。
- 親水性成分が、組成物の重量に対して、少なくとも20重量%の量で存在する、請求項1から3のいずれかに記載の医薬組成物。
- 親水性成分と両親媒性成分の重量比が、1:1以上である、請求項1から4のいずれかに記載の医薬組成物。
- 親油性成分が、イソプロピルミリスタート、イソプロピルパルミタート、ジブチルアジパート、ジイソプロピルアジパート、およびトリグリセリドから選択された少なくとも1種の賦形剤を含む、請求項1から5のいずれかに記載の医薬組成物。
- 両親媒性成分が、リン脂質、アルキルポリグルコシド、脂肪酸とのソルビタンエステル、脂肪アルコールのポリアルキレングリコールエーテル、および脂肪酸とのグリセロールペグ化モノおよびジエステルから選択された少なくとも1種の界面活性剤を含む、請求項1から6のいずれかに記載の医薬組成物。
- 両親媒性成分が、少なくとも2種の界面活性剤を含む、請求項1から7のいずれかに記載の医薬組成物。
- 皮膚、粘膜、または眼に投与する薬剤として使用するための、請求項1から8のいずれかに記載の医薬組成物。
- アトピー性皮膚炎、乾癬、膠原病、または眼の炎症性疾患を予防および/または治療する薬剤として使用するための、請求項1から8のいずれかに記載の医薬組成物。
- 請求項1から8のいずれかに記載の医薬組成物を調製する方法であって、高剪断条件または加圧均質化を適用することなく、組成物の成分を組み合わせ、混合する方法。
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP09012724.2 | 2009-10-08 | ||
| EP09012724A EP2308468A1 (en) | 2009-10-08 | 2009-10-08 | Novel pharmaceutical composition comprising a macrolide immunosuppressant drug |
| PCT/EP2010/064965 WO2011042485A1 (en) | 2009-10-08 | 2010-10-07 | Novel pharmaceutical composition comprising a macrolide immunosuppressant drug |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| JP2013507337A JP2013507337A (ja) | 2013-03-04 |
| JP2013507337A5 JP2013507337A5 (ja) | 2013-11-07 |
| JP5764792B2 true JP5764792B2 (ja) | 2015-08-19 |
Family
ID=41435414
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2012532587A Active JP5764792B2 (ja) | 2009-10-08 | 2010-10-07 | マクロライド免疫抑制剤を含む新規な医薬組成物 |
Country Status (12)
| Country | Link |
|---|---|
| US (1) | US8574562B2 (ja) |
| EP (2) | EP2308468A1 (ja) |
| JP (1) | JP5764792B2 (ja) |
| KR (1) | KR101790257B1 (ja) |
| CN (1) | CN102510752B (ja) |
| AU (1) | AU2010305404B2 (ja) |
| BR (1) | BR112012007332B1 (ja) |
| CA (1) | CA2774720C (ja) |
| ES (1) | ES2552803T3 (ja) |
| IN (1) | IN2012DN01559A (ja) |
| MX (1) | MX2012003800A (ja) |
| WO (1) | WO2011042485A1 (ja) |
Families Citing this family (18)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US10265265B2 (en) | 2007-03-15 | 2019-04-23 | Drug Delivery Solutions Limited | Topical composition |
| MY176112A (en) * | 2011-06-23 | 2020-07-24 | Santen Pharmaceutical Co Ltd | Ophthalmic solution containing hyaluronic acid or salt thereof and propylene glycol |
| ES2797376T3 (es) | 2013-01-24 | 2020-12-02 | Palvella Therapeutics Inc | Composiciones para la administración transdérmica de inhibidores de mTOR |
| CN103830178B (zh) * | 2014-03-18 | 2017-01-11 | 深圳劲创生物技术有限公司 | 一种蒜氨酸微乳及其制备方法和应用 |
| US10525012B2 (en) | 2015-08-11 | 2020-01-07 | Eyesiu Medicines B.V. | Pegylated lipid nanoparticle with bioactive lipophilic compound |
| CN109069418A (zh) | 2016-04-04 | 2018-12-21 | 药品配送方案有限公司 | 包含他克莫司的局部组合物 |
| WO2017207819A1 (en) * | 2016-06-03 | 2017-12-07 | Avexxin As | Combination therapy comprising a polyunsaturated ketone and a calcineurin inhibitor |
| CN106074386A (zh) * | 2016-08-31 | 2016-11-09 | 佛山市弘泰药物研发有限公司 | 一种依维莫司自微乳制剂及其制备方法 |
| KR20190055153A (ko) | 2016-09-21 | 2019-05-22 | 아벡신 에이에스 | 약학 조성물 |
| US10722499B2 (en) | 2017-01-06 | 2020-07-28 | Palvella Therapeutics, Inc. | Anyhydrous compositions of mTOR inhibitors and methods of use |
| US11766421B2 (en) | 2017-09-25 | 2023-09-26 | Surface Ophthalmics, Inc. | Ophthalmic pharmaceutical compositions and methods for treating ocular surface disease |
| EP3542788A1 (en) | 2018-03-19 | 2019-09-25 | MC2 Therapeutics Limited | Topical composition comprising calcipotriol and betamethasone dipropionate |
| WO2019233722A1 (en) | 2018-06-08 | 2019-12-12 | Almirall, S.A. | Pharmaceutical composition comprising tacrolimus |
| EP3817743A4 (en) | 2018-07-02 | 2022-07-06 | Palvella Therapeutics, Inc. | ANHYDROUS COMPOSITIONS OF MTOR INHIBITORS AND METHODS OF USE |
| AU2019417161B2 (en) | 2018-12-27 | 2023-06-15 | Surface Ophthalmics, Inc. | Ophthalmic pharmaceutical compositions and methods for treating ocular surface disease |
| CA3164846A1 (en) * | 2020-01-22 | 2021-07-29 | Nayan Desai | Topical compositions comprising a macrolide immunosuppressant |
| TWI769745B (zh) * | 2021-03-19 | 2022-07-01 | 國泰醫療財團法人國泰綜合醫院 | 醫藥組成物與製藥用途 |
| BE1030538B1 (nl) | 2022-05-18 | 2023-12-19 | Bogaert Gina Van | Liposomaal preparaat met ingekapselde hormonen, werkwijze voor de productie en gebruik ervan |
Family Cites Families (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CH677448A5 (ja) * | 1987-11-09 | 1991-05-31 | Sandoz Ag | |
| GB2222770B (en) * | 1988-09-16 | 1992-07-29 | Sandoz Ltd | Pharmaceutical compositions containing cyclosporins |
| CH686761A5 (de) * | 1993-05-27 | 1996-06-28 | Sandoz Ag | Galenische Formulierungen. |
| GB2315216B (en) * | 1993-10-05 | 1998-10-14 | Ciba Geigy Ag | Microemulsion preconcentrates comprising FK506 or 33-epi-chloro-33-desoxy-ascomycin |
| NZ295170A (en) * | 1994-10-26 | 1999-02-25 | Novartis Ag | Topical pharmaceutical compositions comprising a fk506 class compound, an unsaturated fatty alcohol, water and diether alcohol, alkanediol or ether diol as a solvent |
| GB9723669D0 (en) * | 1997-11-07 | 1998-01-07 | Univ Aberdeen | Skin penetration enhancing components |
| DE10036871A1 (de) * | 2000-07-28 | 2002-02-14 | Pharmasol Gmbh | Dispersionen zur Formulierung wenig oder schwer löslicher Wirkstoffe |
| CN100335036C (zh) * | 2001-11-01 | 2007-09-05 | 耶路撒冷希伯来语大学依苏姆研究开发公司 | 用于干眼症治疗的方法和组合物 |
| CA2470230C (en) * | 2001-12-14 | 2012-01-24 | Jagotec Ag | Pharmaceutical formulation comprising cyclosporin and use thereof |
| CA2570825A1 (en) * | 2004-07-16 | 2006-01-26 | Novartis Ag | Use of a steroid for enhancement of skin permeability |
| WO2006050838A2 (en) * | 2004-11-09 | 2006-05-18 | Novagali Pharma Sa | Ophthalmic oil-in-water type emulsion with stable positive zeta potential |
| KR100678829B1 (ko) * | 2004-12-06 | 2007-02-05 | 한미약품 주식회사 | 타크로리무스의 경구용 마이크로에멀젼 조성물 |
| WO2006123354A2 (en) * | 2005-02-02 | 2006-11-23 | Mega Lifesciences Pvt. Ltd. | Oral pharmaceutical composition |
| US8663639B2 (en) | 2005-02-09 | 2014-03-04 | Santen Pharmaceutical Co., Ltd. | Formulations for treating ocular diseases and conditions |
| CA2597590A1 (en) * | 2005-02-09 | 2006-08-17 | Macusight, Inc. | Formulations for ocular treatment |
-
2009
- 2009-10-08 EP EP09012724A patent/EP2308468A1/en not_active Withdrawn
-
2010
- 2010-10-07 US US13/498,182 patent/US8574562B2/en active Active
- 2010-10-07 ES ES10762917.2T patent/ES2552803T3/es active Active
- 2010-10-07 CA CA2774720A patent/CA2774720C/en active Active
- 2010-10-07 WO PCT/EP2010/064965 patent/WO2011042485A1/en active Application Filing
- 2010-10-07 AU AU2010305404A patent/AU2010305404B2/en active Active
- 2010-10-07 BR BR112012007332-0A patent/BR112012007332B1/pt active IP Right Grant
- 2010-10-07 CN CN201080042067.1A patent/CN102510752B/zh active Active
- 2010-10-07 EP EP10762917.2A patent/EP2485714B1/en active Active
- 2010-10-07 MX MX2012003800A patent/MX2012003800A/es active IP Right Grant
- 2010-10-07 JP JP2012532587A patent/JP5764792B2/ja active Active
- 2010-10-07 KR KR1020127009028A patent/KR101790257B1/ko active IP Right Grant
-
2012
- 2012-02-21 IN IN1559DEN2012 patent/IN2012DN01559A/en unknown
Also Published As
| Publication number | Publication date |
|---|---|
| KR20120093865A (ko) | 2012-08-23 |
| EP2485714B1 (en) | 2015-09-09 |
| BR112012007332B1 (pt) | 2021-07-13 |
| CA2774720C (en) | 2017-05-23 |
| WO2011042485A1 (en) | 2011-04-14 |
| US20120184511A1 (en) | 2012-07-19 |
| EP2485714A1 (en) | 2012-08-15 |
| CN102510752B (zh) | 2015-08-05 |
| JP2013507337A (ja) | 2013-03-04 |
| BR112012007332A2 (pt) | 2016-10-04 |
| EP2308468A1 (en) | 2011-04-13 |
| AU2010305404B2 (en) | 2015-04-02 |
| CN102510752A (zh) | 2012-06-20 |
| CA2774720A1 (en) | 2011-04-14 |
| AU2010305404A1 (en) | 2012-03-08 |
| ES2552803T3 (es) | 2015-12-02 |
| MX2012003800A (es) | 2012-06-28 |
| US8574562B2 (en) | 2013-11-05 |
| KR101790257B1 (ko) | 2017-10-26 |
| IN2012DN01559A (ja) | 2015-06-05 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5764792B2 (ja) | マクロライド免疫抑制剤を含む新規な医薬組成物 | |
| JP2020183427A (ja) | 局所製剤 | |
| JP2018123138A (ja) | 半フッ化アルカンを基礎にした局所薬学的組成物 | |
| KR101930244B1 (ko) | 국소용 코르티코스테로이드 조성물 | |
| JP6030565B2 (ja) | ミニエマルション、同ミニエマルションを形成するための方法および医薬品の製造における同ミニエマルションの使用 | |
| CN114641297B (zh) | 使用高克拉夫特温度阴离子表面活性剂治疗皮肤病症 | |
| MX2007011283A (es) | Mejora de penetracion de macrolidos a traves de la piel humana. | |
| KR20230054682A (ko) | Jak 저해제 및 라우레스-4를 함유하는 국소 제형 | |
| CZ302649B6 (cs) | Farmaceutická formulace zahrnující cyklosporin a její použití | |
| JP2018537524A (ja) | イソトレチノイン製剤ならびにその使用および方法 | |
| US20080145434A1 (en) | Compositions for the reversal and detoxification of anesthetics and other compounds and methods of their use | |
| CA3240662A1 (en) | Stable formulations of shr0302 | |
| CN118660693A (zh) | Shr0302的稳定制剂 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20130919 |
|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20130919 |
|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20140620 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20140701 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20140919 |
|
| A602 | Written permission of extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A602 Effective date: 20140929 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20141015 |
|
| A602 | Written permission of extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A602 Effective date: 20141022 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20141219 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20150415 |
|
| A711 | Notification of change in applicant |
Free format text: JAPANESE INTERMEDIATE CODE: A711 Effective date: 20150512 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20150512 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20150529 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 5764792 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| S111 | Request for change of ownership or part of ownership |
Free format text: JAPANESE INTERMEDIATE CODE: R313113 |
|
| R350 | Written notification of registration of transfer |
Free format text: JAPANESE INTERMEDIATE CODE: R350 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |