JP4985763B2 - 育毛・発毛用経口組成物 - Google Patents
育毛・発毛用経口組成物 Download PDFInfo
- Publication number
- JP4985763B2 JP4985763B2 JP2009503893A JP2009503893A JP4985763B2 JP 4985763 B2 JP4985763 B2 JP 4985763B2 JP 2009503893 A JP2009503893 A JP 2009503893A JP 2009503893 A JP2009503893 A JP 2009503893A JP 4985763 B2 JP4985763 B2 JP 4985763B2
- Authority
- JP
- Japan
- Prior art keywords
- hair growth
- hair
- oral composition
- kombu
- temporary root
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 230000003779 hair growth Effects 0.000 title claims description 56
- 239000000203 mixture Substances 0.000 title claims description 40
- 239000000843 powder Substances 0.000 claims description 25
- 239000000284 extract Substances 0.000 claims description 23
- 239000000463 material Substances 0.000 claims description 12
- 230000001737 promoting effect Effects 0.000 claims description 11
- 230000017531 blood circulation Effects 0.000 claims description 8
- 239000002775 capsule Substances 0.000 claims description 8
- 239000002994 raw material Substances 0.000 claims description 8
- 239000000126 substance Substances 0.000 claims description 8
- 240000004808 Saccharomyces cerevisiae Species 0.000 claims description 7
- 150000001413 amino acids Chemical class 0.000 claims description 7
- YKPUWZUDDOIDPM-SOFGYWHQSA-N capsaicin Chemical compound COC1=CC(CNC(=O)CCCC\C=C\C(C)C)=CC=C1O YKPUWZUDDOIDPM-SOFGYWHQSA-N 0.000 claims description 6
- 108010076876 Keratins Proteins 0.000 claims description 5
- 102000011782 Keratins Human genes 0.000 claims description 5
- 239000003795 chemical substances by application Substances 0.000 claims description 5
- 235000013343 vitamin Nutrition 0.000 claims description 5
- 239000011782 vitamin Substances 0.000 claims description 5
- 229940088594 vitamin Drugs 0.000 claims description 5
- 229930003231 vitamin Natural products 0.000 claims description 5
- 235000020741 pine bark extract Nutrition 0.000 claims description 4
- 229940106587 pine bark extract Drugs 0.000 claims description 4
- 235000017663 capsaicin Nutrition 0.000 claims description 3
- 229960002504 capsaicin Drugs 0.000 claims description 3
- 239000008187 granular material Substances 0.000 claims description 3
- 235000015110 jellies Nutrition 0.000 claims description 3
- 239000008274 jelly Substances 0.000 claims description 3
- 238000004519 manufacturing process Methods 0.000 claims description 3
- 239000003826 tablet Substances 0.000 claims description 3
- 239000008280 blood Substances 0.000 claims 1
- 210000004369 blood Anatomy 0.000 claims 1
- 230000000694 effects Effects 0.000 description 26
- 235000013305 food Nutrition 0.000 description 24
- 210000004209 hair Anatomy 0.000 description 19
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 12
- 235000015872 dietary supplement Nutrition 0.000 description 9
- 238000000034 method Methods 0.000 description 9
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 7
- 235000001014 amino acid Nutrition 0.000 description 7
- 239000011591 potassium Substances 0.000 description 7
- 229910052700 potassium Inorganic materials 0.000 description 7
- 201000004384 Alopecia Diseases 0.000 description 6
- 238000004458 analytical method Methods 0.000 description 6
- 235000013325 dietary fiber Nutrition 0.000 description 6
- 239000000047 product Substances 0.000 description 6
- 241000512259 Ascophyllum nodosum Species 0.000 description 5
- 235000014680 Saccharomyces cerevisiae Nutrition 0.000 description 5
- 230000003676 hair loss Effects 0.000 description 5
- 208000024963 hair loss Diseases 0.000 description 5
- 239000011734 sodium Substances 0.000 description 5
- 241000264877 Hippospongia communis Species 0.000 description 4
- 239000004615 ingredient Substances 0.000 description 4
- 229910052500 inorganic mineral Inorganic materials 0.000 description 4
- 239000011707 mineral Substances 0.000 description 4
- 235000010755 mineral Nutrition 0.000 description 4
- 210000004761 scalp Anatomy 0.000 description 4
- 238000012360 testing method Methods 0.000 description 4
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Chemical compound O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 4
- 230000009471 action Effects 0.000 description 3
- 238000009472 formulation Methods 0.000 description 3
- 235000013402 health food Nutrition 0.000 description 3
- 230000035764 nutrition Effects 0.000 description 3
- 239000000546 pharmaceutical excipient Substances 0.000 description 3
- 238000002360 preparation method Methods 0.000 description 3
- 238000010298 pulverizing process Methods 0.000 description 3
- 239000000243 solution Substances 0.000 description 3
- -1 sulfur amino acids Chemical class 0.000 description 3
- 230000003813 thin hair Effects 0.000 description 3
- 239000011573 trace mineral Substances 0.000 description 3
- 235000013619 trace mineral Nutrition 0.000 description 3
- YBJHBAHKTGYVGT-ZKWXMUAHSA-N (+)-Biotin Chemical compound N1C(=O)N[C@@H]2[C@H](CCCCC(=O)O)SC[C@@H]21 YBJHBAHKTGYVGT-ZKWXMUAHSA-N 0.000 description 2
- GHOKWGTUZJEAQD-ZETCQYMHSA-N (D)-(+)-Pantothenic acid Chemical compound OCC(C)(C)[C@@H](O)C(=O)NCCC(O)=O GHOKWGTUZJEAQD-ZETCQYMHSA-N 0.000 description 2
- IXPNQXFRVYWDDI-UHFFFAOYSA-N 1-methyl-2,4-dioxo-1,3-diazinane-5-carboximidamide Chemical compound CN1CC(C(N)=N)C(=O)NC1=O IXPNQXFRVYWDDI-UHFFFAOYSA-N 0.000 description 2
- 229920000855 Fucoidan Polymers 0.000 description 2
- 108010010803 Gelatin Proteins 0.000 description 2
- WHUUTDBJXJRKMK-UHFFFAOYSA-N Glutamic acid Natural products OC(=O)C(N)CCC(O)=O WHUUTDBJXJRKMK-UHFFFAOYSA-N 0.000 description 2
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 2
- DFPAKSUCGFBDDF-UHFFFAOYSA-N Nicotinamide Chemical compound NC(=O)C1=CC=CN=C1 DFPAKSUCGFBDDF-UHFFFAOYSA-N 0.000 description 2
- 108010009736 Protein Hydrolysates Proteins 0.000 description 2
- 239000000654 additive Substances 0.000 description 2
- 230000000259 anti-tumor effect Effects 0.000 description 2
- 235000013361 beverage Nutrition 0.000 description 2
- HVYWMOMLDIMFJA-DPAQBDIFSA-N cholesterol Chemical compound C1C=C2C[C@@H](O)CC[C@]2(C)[C@@H]2[C@@H]1[C@@H]1CC[C@H]([C@H](C)CCCC(C)C)[C@@]1(C)CC2 HVYWMOMLDIMFJA-DPAQBDIFSA-N 0.000 description 2
- 230000003247 decreasing effect Effects 0.000 description 2
- 239000002552 dosage form Substances 0.000 description 2
- 230000029142 excretion Effects 0.000 description 2
- 238000000605 extraction Methods 0.000 description 2
- 239000008273 gelatin Substances 0.000 description 2
- 229920000159 gelatin Polymers 0.000 description 2
- 235000019322 gelatine Nutrition 0.000 description 2
- 235000011852 gelatine desserts Nutrition 0.000 description 2
- 235000013922 glutamic acid Nutrition 0.000 description 2
- 239000004220 glutamic acid Substances 0.000 description 2
- 239000007902 hard capsule Substances 0.000 description 2
- 210000003128 head Anatomy 0.000 description 2
- 239000005556 hormone Substances 0.000 description 2
- 229940088597 hormone Drugs 0.000 description 2
- 230000006872 improvement Effects 0.000 description 2
- 238000002156 mixing Methods 0.000 description 2
- 235000016709 nutrition Nutrition 0.000 description 2
- 239000002245 particle Substances 0.000 description 2
- QKFJKGMPGYROCL-UHFFFAOYSA-N phenyl isothiocyanate Chemical compound S=C=NC1=CC=CC=C1 QKFJKGMPGYROCL-UHFFFAOYSA-N 0.000 description 2
- 230000003658 preventing hair loss Effects 0.000 description 2
- 239000003531 protein hydrolysate Substances 0.000 description 2
- 210000003491 skin Anatomy 0.000 description 2
- 229910052708 sodium Inorganic materials 0.000 description 2
- 235000010413 sodium alginate Nutrition 0.000 description 2
- 239000000661 sodium alginate Substances 0.000 description 2
- 229940005550 sodium alginate Drugs 0.000 description 2
- 230000009469 supplementation Effects 0.000 description 2
- 235000019583 umami taste Nutrition 0.000 description 2
- YBJHBAHKTGYVGT-UHFFFAOYSA-N 5-(2-oxo-1,3,3a,4,6,6a-hexahydrothieno[3,4-d]imidazol-4-yl)pentanoic acid Chemical compound N1C(=O)NC2C(CCCCC(=O)O)SCC21 YBJHBAHKTGYVGT-UHFFFAOYSA-N 0.000 description 1
- FHVDTGUDJYJELY-UHFFFAOYSA-N 6-{[2-carboxy-4,5-dihydroxy-6-(phosphanyloxy)oxan-3-yl]oxy}-4,5-dihydroxy-3-phosphanyloxane-2-carboxylic acid Chemical compound O1C(C(O)=O)C(P)C(O)C(O)C1OC1C(C(O)=O)OC(OP)C(O)C1O FHVDTGUDJYJELY-UHFFFAOYSA-N 0.000 description 1
- 241001474374 Blennius Species 0.000 description 1
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 1
- GHOKWGTUZJEAQD-UHFFFAOYSA-N Chick antidermatitis factor Natural products OCC(C)(C)C(O)C(=O)NCCC(O)=O GHOKWGTUZJEAQD-UHFFFAOYSA-N 0.000 description 1
- 102000008186 Collagen Human genes 0.000 description 1
- 108010035532 Collagen Proteins 0.000 description 1
- 239000004129 EU approved improving agent Substances 0.000 description 1
- 239000004606 Fillers/Extenders Substances 0.000 description 1
- 239000009429 Ginkgo biloba extract Substances 0.000 description 1
- 244000068988 Glycine max Species 0.000 description 1
- 235000010469 Glycine max Nutrition 0.000 description 1
- 241000546188 Hypericum Species 0.000 description 1
- 235000017309 Hypericum perforatum Nutrition 0.000 description 1
- LEVWYRKDKASIDU-IMJSIDKUSA-N L-cystine Chemical compound [O-]C(=O)[C@@H]([NH3+])CSSC[C@H]([NH3+])C([O-])=O LEVWYRKDKASIDU-IMJSIDKUSA-N 0.000 description 1
- FFEARJCKVFRZRR-BYPYZUCNSA-N L-methionine Chemical compound CSCC[C@H](N)C(O)=O FFEARJCKVFRZRR-BYPYZUCNSA-N 0.000 description 1
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 1
- PVNIIMVLHYAWGP-UHFFFAOYSA-N Niacin Chemical compound OC(=O)C1=CC=CN=C1 PVNIIMVLHYAWGP-UHFFFAOYSA-N 0.000 description 1
- 240000007594 Oryza sativa Species 0.000 description 1
- 235000007164 Oryza sativa Nutrition 0.000 description 1
- 244000131316 Panax pseudoginseng Species 0.000 description 1
- 235000005035 Panax pseudoginseng ssp. pseudoginseng Nutrition 0.000 description 1
- 235000003140 Panax quinquefolius Nutrition 0.000 description 1
- 241000199919 Phaeophyceae Species 0.000 description 1
- AUNGANRZJHBGPY-SCRDCRAPSA-N Riboflavin Chemical compound OC[C@@H](O)[C@@H](O)[C@@H](O)CN1C=2C=C(C)C(C)=CC=2N=C2C1=NC(=O)NC2=O AUNGANRZJHBGPY-SCRDCRAPSA-N 0.000 description 1
- 235000017276 Salvia Nutrition 0.000 description 1
- 240000007164 Salvia officinalis Species 0.000 description 1
- 244000062793 Sorghum vulgare Species 0.000 description 1
- 229930006000 Sucrose Natural products 0.000 description 1
- 241001261506 Undaria pinnatifida Species 0.000 description 1
- 239000003929 acidic solution Substances 0.000 description 1
- 230000002378 acidificating effect Effects 0.000 description 1
- 230000003213 activating effect Effects 0.000 description 1
- 239000012190 activator Substances 0.000 description 1
- 229940072056 alginate Drugs 0.000 description 1
- 235000010443 alginic acid Nutrition 0.000 description 1
- 229920000615 alginic acid Polymers 0.000 description 1
- 229940024606 amino acid Drugs 0.000 description 1
- 230000002429 anti-coagulating effect Effects 0.000 description 1
- 229940121363 anti-inflammatory agent Drugs 0.000 description 1
- 239000002260 anti-inflammatory agent Substances 0.000 description 1
- 238000009360 aquaculture Methods 0.000 description 1
- 244000144974 aquaculture Species 0.000 description 1
- 239000007864 aqueous solution Substances 0.000 description 1
- 235000019606 astringent taste Nutrition 0.000 description 1
- 229960002685 biotin Drugs 0.000 description 1
- 235000020958 biotin Nutrition 0.000 description 1
- 239000011616 biotin Substances 0.000 description 1
- 235000019658 bitter taste Nutrition 0.000 description 1
- 230000036772 blood pressure Effects 0.000 description 1
- 235000008429 bread Nutrition 0.000 description 1
- 239000011575 calcium Substances 0.000 description 1
- 229910052791 calcium Inorganic materials 0.000 description 1
- FAPWYRCQGJNNSJ-UBKPKTQASA-L calcium D-pantothenic acid Chemical compound [Ca+2].OCC(C)(C)[C@@H](O)C(=O)NCCC([O-])=O.OCC(C)(C)[C@@H](O)C(=O)NCCC([O-])=O FAPWYRCQGJNNSJ-UBKPKTQASA-L 0.000 description 1
- 229960002079 calcium pantothenate Drugs 0.000 description 1
- CJZGTCYPCWQAJB-UHFFFAOYSA-L calcium stearate Chemical compound [Ca+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O CJZGTCYPCWQAJB-UHFFFAOYSA-L 0.000 description 1
- 235000013539 calcium stearate Nutrition 0.000 description 1
- 239000008116 calcium stearate Substances 0.000 description 1
- 230000008859 change Effects 0.000 description 1
- 235000012000 cholesterol Nutrition 0.000 description 1
- 229920001436 collagen Polymers 0.000 description 1
- 239000003086 colorant Substances 0.000 description 1
- 235000009508 confectionery Nutrition 0.000 description 1
- 238000011109 contamination Methods 0.000 description 1
- 238000007796 conventional method Methods 0.000 description 1
- 235000014510 cooky Nutrition 0.000 description 1
- 229910052802 copper Inorganic materials 0.000 description 1
- 229960003067 cystine Drugs 0.000 description 1
- 239000007857 degradation product Substances 0.000 description 1
- 210000003298 dental enamel Anatomy 0.000 description 1
- 238000010586 diagram Methods 0.000 description 1
- 235000005911 diet Nutrition 0.000 description 1
- 230000000378 dietary effect Effects 0.000 description 1
- 235000014113 dietary fatty acids Nutrition 0.000 description 1
- 239000012153 distilled water Substances 0.000 description 1
- 230000035622 drinking Effects 0.000 description 1
- 230000001747 exhibiting effect Effects 0.000 description 1
- 230000001815 facial effect Effects 0.000 description 1
- 229930195729 fatty acid Natural products 0.000 description 1
- 239000000194 fatty acid Substances 0.000 description 1
- 239000000796 flavoring agent Substances 0.000 description 1
- 235000019634 flavors Nutrition 0.000 description 1
- 238000004186 food analysis Methods 0.000 description 1
- 230000037406 food intake Effects 0.000 description 1
- 235000003599 food sweetener Nutrition 0.000 description 1
- 229940068052 ginkgo biloba extract Drugs 0.000 description 1
- 235000020686 ginkgo biloba extract Nutrition 0.000 description 1
- 235000008434 ginseng Nutrition 0.000 description 1
- 230000003648 hair appearance Effects 0.000 description 1
- 210000003780 hair follicle Anatomy 0.000 description 1
- 238000010438 heat treatment Methods 0.000 description 1
- 238000004128 high performance liquid chromatography Methods 0.000 description 1
- 238000007654 immersion Methods 0.000 description 1
- 230000003752 improving hair Effects 0.000 description 1
- 230000000968 intestinal effect Effects 0.000 description 1
- GOMNOOKGLZYEJT-UHFFFAOYSA-N isoflavone Chemical compound C=1OC2=CC=CC=C2C(=O)C=1C1=CC=CC=C1 GOMNOOKGLZYEJT-UHFFFAOYSA-N 0.000 description 1
- CJWQYWQDLBZGPD-UHFFFAOYSA-N isoflavone Natural products C1=C(OC)C(OC)=CC(OC)=C1C1=COC2=C(C=CC(C)(C)O3)C3=C(OC)C=C2C1=O CJWQYWQDLBZGPD-UHFFFAOYSA-N 0.000 description 1
- 235000008696 isoflavones Nutrition 0.000 description 1
- 239000011777 magnesium Substances 0.000 description 1
- 229910052749 magnesium Inorganic materials 0.000 description 1
- 229910052748 manganese Inorganic materials 0.000 description 1
- 230000004060 metabolic process Effects 0.000 description 1
- 229930182817 methionine Natural products 0.000 description 1
- 235000019713 millet Nutrition 0.000 description 1
- 229960003966 nicotinamide Drugs 0.000 description 1
- 235000005152 nicotinamide Nutrition 0.000 description 1
- 239000011570 nicotinamide Substances 0.000 description 1
- 229960003512 nicotinic acid Drugs 0.000 description 1
- 235000001968 nicotinic acid Nutrition 0.000 description 1
- 239000011664 nicotinic acid Substances 0.000 description 1
- 235000015097 nutrients Nutrition 0.000 description 1
- 150000007524 organic acids Chemical class 0.000 description 1
- 235000020007 pale lager Nutrition 0.000 description 1
- 229940055726 pantothenic acid Drugs 0.000 description 1
- 235000019161 pantothenic acid Nutrition 0.000 description 1
- 239000011713 pantothenic acid Substances 0.000 description 1
- 230000000144 pharmacologic effect Effects 0.000 description 1
- 229940117953 phenylisothiocyanate Drugs 0.000 description 1
- 239000000419 plant extract Substances 0.000 description 1
- 235000007686 potassium Nutrition 0.000 description 1
- 230000008569 process Effects 0.000 description 1
- ZUFQODAHGAHPFQ-UHFFFAOYSA-N pyridoxine hydrochloride Chemical compound Cl.CC1=NC=C(CO)C(CO)=C1O ZUFQODAHGAHPFQ-UHFFFAOYSA-N 0.000 description 1
- 238000011084 recovery Methods 0.000 description 1
- 230000001105 regulatory effect Effects 0.000 description 1
- 235000009566 rice Nutrition 0.000 description 1
- 239000011435 rock Substances 0.000 description 1
- 229940109850 royal jelly Drugs 0.000 description 1
- 150000003839 salts Chemical class 0.000 description 1
- 239000004576 sand Substances 0.000 description 1
- 229930182490 saponin Natural products 0.000 description 1
- 150000007949 saponins Chemical class 0.000 description 1
- 235000017709 saponins Nutrition 0.000 description 1
- 210000004927 skin cell Anatomy 0.000 description 1
- 235000014347 soups Nutrition 0.000 description 1
- 239000005720 sucrose Substances 0.000 description 1
- 239000011593 sulfur Substances 0.000 description 1
- 229910052717 sulfur Inorganic materials 0.000 description 1
- 239000006228 supernatant Substances 0.000 description 1
- 230000001629 suppression Effects 0.000 description 1
- 239000003765 sweetening agent Substances 0.000 description 1
- DPJRMOMPQZCRJU-UHFFFAOYSA-M thiamine hydrochloride Chemical compound Cl.[Cl-].CC1=C(CCO)SC=[N+]1CC1=CN=C(C)N=C1N DPJRMOMPQZCRJU-UHFFFAOYSA-M 0.000 description 1
- 229940124549 vasodilator Drugs 0.000 description 1
- 239000003071 vasodilator agent Substances 0.000 description 1
- 235000019156 vitamin B Nutrition 0.000 description 1
- 239000011720 vitamin B Substances 0.000 description 1
- 230000037303 wrinkles Effects 0.000 description 1
- 239000012138 yeast extract Substances 0.000 description 1
- 229910052725 zinc Inorganic materials 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K36/00—Medicinal preparations of undetermined constitution containing material from algae, lichens, fungi or plants, or derivatives thereof, e.g. traditional herbal medicines
- A61K36/02—Algae
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS, OR NON-ALCOHOLIC BEVERAGES, NOT COVERED BY SUBCLASSES A21D OR A23B-A23J; THEIR PREPARATION OR TREATMENT, e.g. COOKING, MODIFICATION OF NUTRITIVE QUALITIES, PHYSICAL TREATMENT; PRESERVATION OF FOODS OR FOODSTUFFS, IN GENERAL
- A23L17/00—Food-from-the-sea products; Fish products; Fish meal; Fish-egg substitutes; Preparation or treatment thereof
- A23L17/60—Edible seaweed
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS, OR NON-ALCOHOLIC BEVERAGES, NOT COVERED BY SUBCLASSES A21D OR A23B-A23J; THEIR PREPARATION OR TREATMENT, e.g. COOKING, MODIFICATION OF NUTRITIVE QUALITIES, PHYSICAL TREATMENT; PRESERVATION OF FOODS OR FOODSTUFFS, IN GENERAL
- A23L33/00—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof
- A23L33/10—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof using additives
- A23L33/105—Plant extracts, their artificial duplicates or their derivatives
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/16—Amides, e.g. hydroxamic acids
- A61K31/165—Amides, e.g. hydroxamic acids having aromatic rings, e.g. colchicine, atenolol, progabide
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K36/00—Medicinal preparations of undetermined constitution containing material from algae, lichens, fungi or plants, or derivatives thereof, e.g. traditional herbal medicines
- A61K36/06—Fungi, e.g. yeasts
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
- A61K38/01—Hydrolysed proteins; Derivatives thereof
- A61K38/012—Hydrolysed proteins; Derivatives thereof from animals
- A61K38/014—Hydrolysed proteins; Derivatives thereof from animals from connective tissue peptides, e.g. gelatin, collagen
- A61K38/015—Hydrolysed proteins; Derivatives thereof from animals from connective tissue peptides, e.g. gelatin, collagen from keratin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/40—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing nitrogen
- A61K8/44—Aminocarboxylic acids or derivatives thereof, e.g. aminocarboxylic acids containing sulfur; Salts; Esters or N-acylated derivatives thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/96—Cosmetics or similar toiletry preparations characterised by the composition containing materials, or derivatives thereof of undetermined constitution
- A61K8/97—Cosmetics or similar toiletry preparations characterised by the composition containing materials, or derivatives thereof of undetermined constitution from algae, fungi, lichens or plants; from derivatives thereof
- A61K8/9706—Algae
- A61K8/9711—Phaeophycota or Phaeophyta [brown algae], e.g. Fucus
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/96—Cosmetics or similar toiletry preparations characterised by the composition containing materials, or derivatives thereof of undetermined constitution
- A61K8/97—Cosmetics or similar toiletry preparations characterised by the composition containing materials, or derivatives thereof of undetermined constitution from algae, fungi, lichens or plants; from derivatives thereof
- A61K8/9728—Fungi, e.g. yeasts
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/96—Cosmetics or similar toiletry preparations characterised by the composition containing materials, or derivatives thereof of undetermined constitution
- A61K8/97—Cosmetics or similar toiletry preparations characterised by the composition containing materials, or derivatives thereof of undetermined constitution from algae, fungi, lichens or plants; from derivatives thereof
- A61K8/9755—Gymnosperms [Coniferophyta]
- A61K8/9767—Pinaceae [Pine family], e.g. pine or cedar
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/96—Cosmetics or similar toiletry preparations characterised by the composition containing materials, or derivatives thereof of undetermined constitution
- A61K8/97—Cosmetics or similar toiletry preparations characterised by the composition containing materials, or derivatives thereof of undetermined constitution from algae, fungi, lichens or plants; from derivatives thereof
- A61K8/9783—Angiosperms [Magnoliophyta]
- A61K8/9789—Magnoliopsida [dicotyledons]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
- A61P17/14—Drugs for dermatological disorders for baldness or alopecia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q7/00—Preparations for affecting hair growth
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23V—INDEXING SCHEME RELATING TO FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES AND LACTIC OR PROPIONIC ACID BACTERIA USED IN FOODSTUFFS OR FOOD PREPARATION
- A23V2002/00—Food compositions, function of food ingredients or processes for food or foodstuffs
Description
また、本発明経口組成物は、粉末剤、錠剤、顆粒剤、カプセル剤、ドリンク剤またはゼリーの形態であるのが好ましい。
上記表に示す通り、養殖コンブ仮根は食物繊維に富んでいることが明らかである。なお、養殖コンブ仮根はアルギン酸ナトリウム約7g/100 g、フコイダン分画約4g/100 gを含む。これらは水溶性食物繊維として有用で、アルギン酸ナトリウムにはコレステロール排泄促進、血圧降下作用、整腸作用、有害物排泄促進作用などがあり、フコイダンには抗血液凝固作用、抗腫瘍作用が知られている。
この結果から、養殖コンブ仮根には旨み成分であるグルタミン酸が通常のコンブの1/10程度しか含まれていないことが分かる。なお、メチオニンとシスチンはいずれの試料からも検出できなかった。
(参考例1)
養殖コンブ仮根粉末の調製
乾燥した養殖コンブ仮根100 kgを粗砕機(カッターミル)で粗く粉砕した後、微砕機(サンプルミル)で微粉末にした。微砕機のスクリーンを1mmφにした場合は200 メッシュ以下が30%、100 〜200 メッシュが20%、48〜100 メッシュが30%、48メッシュ以上が20%であった。この粉砕物をさらに0.5 mmφのスクリーンを用いて粉砕すると、200 メッシュ以下が50%、100 〜200 メッシュが30%、48〜100 メッシュが18%、48メッシュ以上が2%の粉体が得られた(収量は92kg)。
(参考例2)
養殖コンブ仮根エキスの調製
粗く裁断した養殖コンブ仮根を、5%酢酸溶液(1kg当たり10L)で下記の条件(室温で14日間および110 ℃で1時間)で抽出して、それぞれ下記表1に示す量および無機質組成のエキスを得た。(単位は試料1kg当たりのg量)
(1カプセル中)
養殖コンブ仮根粉末 250mg
養殖コンブ仮根エキス粉末 50mg
ビール酵母 60mg
ショ糖脂肪酸エステル 10mg
(1カプセル中)
養殖コンブ仮根粉末 250.00mg
松樹皮エキス 2.50mg
ケラチンSHパウダー 30.00mg
ビタミンB1 塩酸塩 2.00mg
ビタミンB2 1.00mg
ビタミンB6 塩酸塩 0.84mg
ニコチン酸アミド 1.25mg
パントテン酸カルシウム 2.50mg
食添ビオチン1% 0.05mg
ヒハツエキスパウダーMF 47.50mg
乾燥ビール酵母A 8.87mg
ステアリン酸カルシウム 3.50mg
(試験例1)
参考例1のようにして製造した養殖コンブ仮根粉末を用いて育毛・発毛効果を試験した。薄毛、抜け毛、白髪などが気になる年齢30〜70歳の男性、女性合わせて40人のボランティアに、朝と夕方の2回、養殖コンブ仮根粉末を3ヶ月間摂取させた (そのうち2人は養殖コンブ仮根粉末無摂取の対照) 。1000mg摂取群には1日合計1000mgを、1500mg摂取群には1日合計1500mgを摂取させた。1ヶ月おきに以下の判定方法により効果を評価し、さらに頭皮の写真撮影およびマイクロスコープによる毛根撮影を行った。
育毛効果
a:服用前に比べ抜け毛が減った
b:髪の毛が増えた、または生えてきた
c:髪の太さが太くなった
d:髪にハリやコシが出てきた
e:白髪が減ってきた
効果判定スコア基準(各項目に対する)
3:著効
2:有効
1:やや有効
0:無効
上記育毛効果に関するa〜eの5項目の点数を合計し、下記の基準により総合的な効果を判定した。
0 :無効
1〜2:やや有効
3〜5:有効
6以上:著効
結果は以下の表7に示す通りである。
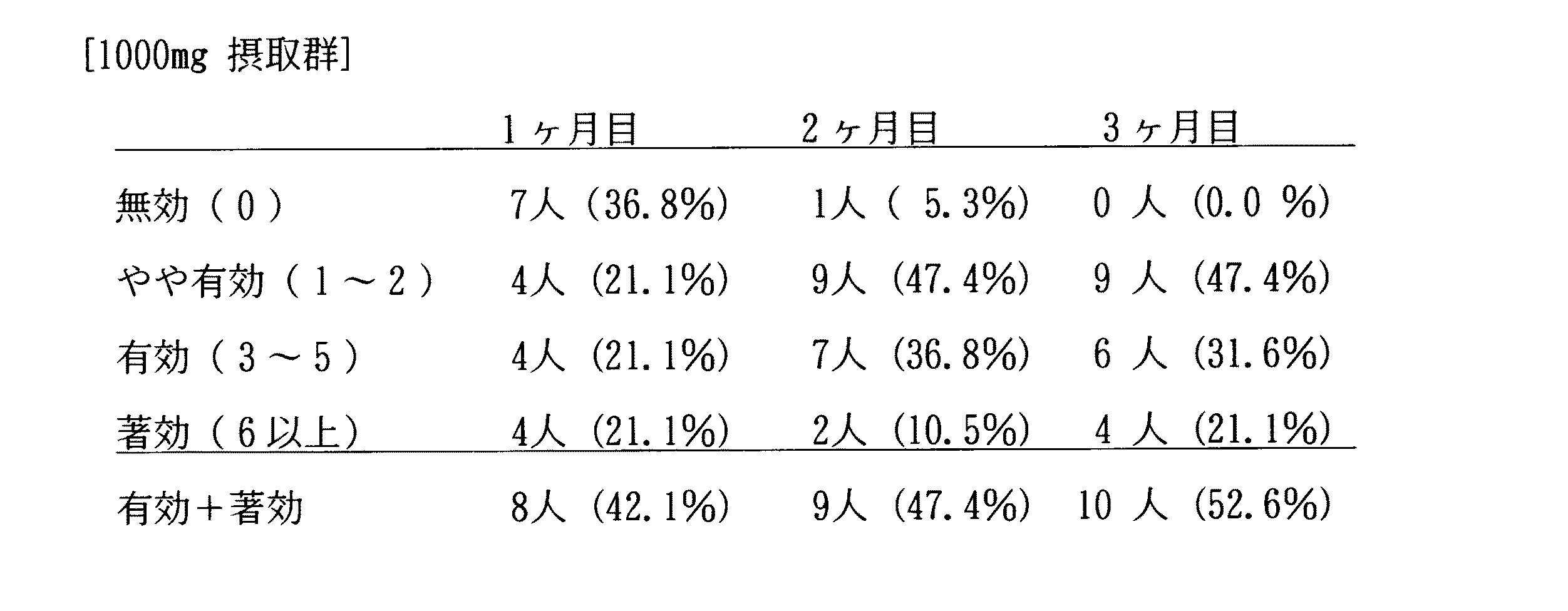
実施例2で製造したカプセル剤を用いて試験例1と同様にして育毛・発毛効果を試験した。ただし、ボランティアは107 人であり、摂取量は養殖コンブ仮根粉末として1日1000mgであった。結果を表9に示す。
Claims (7)
- 養殖コンブ仮根部を原料とする素材を含む育毛・発毛のための経口組成物。
- さらに、ケラチン蛋白質もしくはその加水分解物、ビタミン類、血行促進物質、酵母およびアミノ酸類から選ばれる1種または2種以上を含有する、請求項1記載の経口組成物。
- 血行促進物質がヒハツエキス、松樹皮エキスおよびカプサイシンから選ばれる1種または2種以上である、請求項3記載の経口組成物。
- 養殖コンブ仮根部を原料とする素材が粉末形態である請求項1、3、4のいずれかの項記載の経口組成物。
- 養殖コンブ仮根部を原料とする素材が養殖コンブ仮根部の抽出物の形態である請求項1、3、4のいずれかの項記載の経口組成物。
- 粉末剤、錠剤、顆粒剤、カプセル剤、ドリンク剤またはゼリーの形態である請求項1、3、4、5、6のいずれかの項記載の経口組成物。
- 経口用育毛・発毛剤の製造のための、養殖コンブ仮根部を原料とする素材の使用。
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2009503893A JP4985763B2 (ja) | 2007-03-15 | 2007-12-07 | 育毛・発毛用経口組成物 |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2007067380 | 2007-03-15 | ||
| JP2007067380 | 2007-03-15 | ||
| PCT/JP2007/073661 WO2008111271A1 (ja) | 2007-03-15 | 2007-12-07 | 育毛・発毛用経口組成物 |
| JP2009503893A JP4985763B2 (ja) | 2007-03-15 | 2007-12-07 | 育毛・発毛用経口組成物 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JPWO2008111271A1 JPWO2008111271A1 (ja) | 2010-06-24 |
| JP4985763B2 true JP4985763B2 (ja) | 2012-07-25 |
Family
ID=39759215
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2009503893A Active JP4985763B2 (ja) | 2007-03-15 | 2007-12-07 | 育毛・発毛用経口組成物 |
Country Status (2)
| Country | Link |
|---|---|
| JP (1) | JP4985763B2 (ja) |
| WO (1) | WO2008111271A1 (ja) |
Families Citing this family (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2010148437A (ja) * | 2008-12-25 | 2010-07-08 | Kushiro Konbu Kenkyusho:Kk | 天然コンブ仮根を原料とした食用粉末の製造方法 |
| JP2011168533A (ja) * | 2010-02-18 | 2011-09-01 | Kyosei Seiyaku Kk | 抗インフルエンザウイルス剤 |
| JP2011184305A (ja) * | 2010-03-04 | 2011-09-22 | Kyosei Seiyaku Kk | アディポネクチン産生促進剤 |
| JP2012167060A (ja) * | 2011-02-15 | 2012-09-06 | Asahi Group Holdings Ltd | 経口用組成物 |
| JP5772678B2 (ja) * | 2012-03-26 | 2015-09-02 | 国立大学法人北見工業大学 | 保水性、吸水性およびヒアルロニダーゼ阻害活性を有する化粧品素材および機能性食品素材 |
| JP6200267B2 (ja) * | 2013-10-07 | 2017-09-20 | カイゲンファーマ株式会社 | 5αレダクターゼ阻害剤 |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2001321121A (ja) * | 2000-05-11 | 2001-11-20 | Ichimaru Pharcos Co Ltd | 育毛養毛用飲食品 |
| JP2003081862A (ja) * | 2001-09-14 | 2003-03-19 | Kyosei Seiyaku Kk | コンブ仮根を用いる抗腫瘍剤および食品 |
| JP2004141140A (ja) * | 2001-12-27 | 2004-05-20 | Jigyo Sozo Kenkyusho:Kk | 海藻類の防腐処理方法、防腐処理した海藻類の加工食品、梅塩の製造方法、並びに海藻類及び梅を利用した飲み物 |
Family Cites Families (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS63277610A (ja) * | 1987-05-09 | 1988-11-15 | Suntory Ltd | 養毛料 |
| JPH0366606A (ja) * | 1989-08-03 | 1991-03-22 | Shimizu Takao | 複合生薬育毛剤 |
| JP3407064B2 (ja) * | 1993-11-13 | 2003-05-19 | サンスター株式会社 | 育毛養毛用食品 |
| JPH09175947A (ja) * | 1995-12-28 | 1997-07-08 | Takasago Internatl Corp | 育毛剤組成物 |
| JPH10114649A (ja) * | 1996-10-15 | 1998-05-06 | Dokutaazu Kosumeteikusu:Kk | 津液改善剤及びそれを含有する経口投与用組成物 |
| JP2000038340A (ja) * | 1998-05-20 | 2000-02-08 | Kumahiro Miyama | 育毛剤および食品 |
| JP4448266B2 (ja) * | 2001-07-30 | 2010-04-07 | 丸善製薬株式会社 | 冷え性改善剤及び冷え性改善用組成物 |
| JP2004217604A (ja) * | 2003-01-17 | 2004-08-05 | Kaigen:Kk | 花粉症の症状を改善する食品 |
| JP2006298857A (ja) * | 2005-04-22 | 2006-11-02 | Toyo Shinyaku:Kk | 育毛剤および育毛用キット |
-
2007
- 2007-12-07 WO PCT/JP2007/073661 patent/WO2008111271A1/ja active Search and Examination
- 2007-12-07 JP JP2009503893A patent/JP4985763B2/ja active Active
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2001321121A (ja) * | 2000-05-11 | 2001-11-20 | Ichimaru Pharcos Co Ltd | 育毛養毛用飲食品 |
| JP2003081862A (ja) * | 2001-09-14 | 2003-03-19 | Kyosei Seiyaku Kk | コンブ仮根を用いる抗腫瘍剤および食品 |
| JP2004141140A (ja) * | 2001-12-27 | 2004-05-20 | Jigyo Sozo Kenkyusho:Kk | 海藻類の防腐処理方法、防腐処理した海藻類の加工食品、梅塩の製造方法、並びに海藻類及び梅を利用した飲み物 |
Also Published As
| Publication number | Publication date |
|---|---|
| JPWO2008111271A1 (ja) | 2010-06-24 |
| WO2008111271A1 (ja) | 2008-09-18 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| Berry et al. | The Middle Eastern and biblical origins of the Mediterranean diet | |
| JP4738464B2 (ja) | アディポネクチン分泌促進用飲食品 | |
| JP4985763B2 (ja) | 育毛・発毛用経口組成物 | |
| JP2012171901A (ja) | 男性用の発毛および育毛用経口摂取食品 | |
| JP2009023984A (ja) | 脂肪分解促進剤及び飲食品 | |
| JP5264107B2 (ja) | 抗酸化作用を有する組成物 | |
| JP4397663B2 (ja) | 筋肉量増加剤 | |
| Munglue | Effects of dietary Nelumbo nucifera (lotus) peduncle extract on growth performance of Nile tilapia (Oreochromis niloticus) | |
| KR100762448B1 (ko) | 잔대를 포함하는 생약재 추출물 및 이를 유효성분으로함유하는 간질환 예방 및 치료용 건강식품 조성물 | |
| JP5171080B2 (ja) | 抗酸化作用を有する組成物 | |
| JP2010265252A (ja) | 育毛剤 | |
| JP6108352B2 (ja) | 疲労改善組成物 | |
| JP2008161077A (ja) | 血液流動性改善作用をもたらす機能性食品 | |
| WO2007007994A1 (en) | Food composition for improving liver function comprising a lonicera caerulea l. var. edulis extract | |
| JP6182808B2 (ja) | ボタンボウフウ又はその抽出物を含む精子改善剤及び精液改善剤 | |
| JP5341422B2 (ja) | 蜂の子およびローヤルゼリーを含有する抗酸化組成物 | |
| JP6541047B1 (ja) | 経口組成物 | |
| JP2005304369A (ja) | 高麗人参及び霊芝又はこれらの抽出物を含有する健康補助食品 | |
| JP7296611B2 (ja) | 一酸化窒素産生促進剤 | |
| KR20180010432A (ko) | 곡물 꿀 칼슘 먹물 해초 생선을 살균배합으로 응고 없는 조성물 및 제조방법 | |
| Thummek et al. | Growth performance and intestinal morphology of Common lowland frog (Rana rugulosa) fed diets supplemented with lotus (Nelumbo nucifera Gaertn.) stamen extract | |
| JP5264106B2 (ja) | 抗酸化作用を有する組成物 | |
| JP7131785B1 (ja) | 皮膚保湿剤および皮膚バリア機能改善剤 | |
| JP6734603B1 (ja) | 経口組成物 | |
| KR102030436B1 (ko) | 견사단백질을 갖는 익힌 누에가공물을 함유하는 발모 촉진용 조성물 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20120403 |
|
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20120416 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 4985763 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| FPAY | Renewal fee payment (event date is renewal date of database) |
Free format text: PAYMENT UNTIL: 20150511 Year of fee payment: 3 |
|
| S111 | Request for change of ownership or part of ownership |
Free format text: JAPANESE INTERMEDIATE CODE: R313115 |
|
| S533 | Written request for registration of change of name |
Free format text: JAPANESE INTERMEDIATE CODE: R313533 |
|
| R350 | Written notification of registration of transfer |
Free format text: JAPANESE INTERMEDIATE CODE: R350 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| RD02 | Notification of acceptance of power of attorney |
Free format text: JAPANESE INTERMEDIATE CODE: R3D02 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |