JP4884772B2 - 経口徐放性錠剤 - Google Patents
経口徐放性錠剤 Download PDFInfo
- Publication number
- JP4884772B2 JP4884772B2 JP2005512546A JP2005512546A JP4884772B2 JP 4884772 B2 JP4884772 B2 JP 4884772B2 JP 2005512546 A JP2005512546 A JP 2005512546A JP 2005512546 A JP2005512546 A JP 2005512546A JP 4884772 B2 JP4884772 B2 JP 4884772B2
- Authority
- JP
- Japan
- Prior art keywords
- krp
- tablet
- oral sustained
- mass
- granule
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related
Links
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/2004—Excipients; Inactive ingredients
- A61K9/2022—Organic macromolecular compounds
- A61K9/205—Polysaccharides, e.g. alginate, gums; Cyclodextrin
- A61K9/2054—Cellulose; Cellulose derivatives, e.g. hydroxypropyl methylcellulose
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/41—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with two or more ring hetero atoms, at least one of which being nitrogen, e.g. tetrazole
- A61K31/4164—1,3-Diazoles
- A61K31/4174—Arylalkylimidazoles, e.g. oxymetazolin, naphazoline, miconazole
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/30—Macromolecular organic or inorganic compounds, e.g. inorganic polyphosphates
- A61K47/36—Polysaccharides; Derivatives thereof, e.g. gums, starch, alginate, dextrin, hyaluronic acid, chitosan, inulin, agar or pectin
- A61K47/38—Cellulose; Derivatives thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P13/00—Drugs for disorders of the urinary system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P13/00—Drugs for disorders of the urinary system
- A61P13/02—Drugs for disorders of the urinary system of urine or of the urinary tract, e.g. urine acidifiers
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
Description
1) 4−(2−メチル−1−イミダゾリル)−2,2−ジフェニルブチルアミド溶液を流動状態にある賦形剤に噴霧することにより4−(2−メチル−1−イミダゾリル)−2,2−ジフェニルブチルアミドを含有する顆粒状組成物を構成し、当該顆粒状組成物と、ヒドロキシプロピルメチルセルロースを含有する組成物とを混合して打錠用顆粒組成物を構成し、当該打錠用顆粒組成物を圧縮成形することを含む経口徐放性錠剤の製造方法;
2)前記ヒドロキシプロピルメチルセルロースの含有割合が錠剤中において18〜73質量%である1)に記載の経口徐放性錠剤の製造方法;
に関するものである。
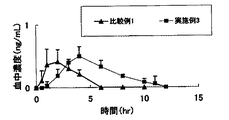
比較例1
実験例1
実験例2
Claims (2)
- 4−(2−メチル−1−イミダゾリル)−2,2−ジフェニルブチルアミド溶液を流動状態にある賦形剤に噴霧することにより4−(2−メチル−1−イミダゾリル)−2,2−ジフェニルブチルアミドを含有する顆粒状組成物を構成し、
前記顆粒状組成物と、ヒドロキシプロピルメチルセルロースを含有する組成物とを混合して打錠用顆粒組成物を構成し、
当該打錠用顆粒組成物を圧縮成形することを含む経口徐放性錠剤の製造方法。 - 前記ヒドロキシプロピルメチルセルロースの含有割合が錠剤中において18〜73質量%である請求項1記載の経口徐放性錠剤の製造方法。
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2005512546A JP4884772B2 (ja) | 2003-08-04 | 2004-08-03 | 経口徐放性錠剤 |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2003286096 | 2003-08-04 | ||
| JP2003286096 | 2003-08-04 | ||
| JP2005512546A JP4884772B2 (ja) | 2003-08-04 | 2004-08-03 | 経口徐放性錠剤 |
| PCT/JP2004/011067 WO2005011682A1 (ja) | 2003-08-04 | 2004-08-03 | 経口徐放性錠剤 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JPWO2005011682A1 JPWO2005011682A1 (ja) | 2007-10-04 |
| JP4884772B2 true JP4884772B2 (ja) | 2012-02-29 |
Family
ID=34113929
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2005512546A Expired - Fee Related JP4884772B2 (ja) | 2003-08-04 | 2004-08-03 | 経口徐放性錠剤 |
Country Status (7)
| Country | Link |
|---|---|
| US (2) | US20070092566A1 (ja) |
| EP (1) | EP1652522A4 (ja) |
| JP (1) | JP4884772B2 (ja) |
| KR (1) | KR101175816B1 (ja) |
| CN (1) | CN1842333A (ja) |
| CA (1) | CA2532714C (ja) |
| WO (1) | WO2005011682A1 (ja) |
Families Citing this family (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20060188554A1 (en) * | 2003-08-04 | 2006-08-24 | Katashi Nakashima | Transdermal absorption preparation |
| JP5112619B2 (ja) * | 2004-05-20 | 2013-01-09 | 大塚製薬株式会社 | 固形医薬製剤 |
| US20080107727A1 (en) * | 2005-01-31 | 2008-05-08 | Katashi Nakashima | Multiple Unit Oral Sustained Release Preparation and Production Method Thereof |
| CA2713679A1 (en) | 2008-01-31 | 2009-08-06 | Kyorin Pharmaceutical Co., Ltd. | Orally rapidly disintegrating tablet comprising imidafenacin |
| CN101983055B (zh) * | 2008-01-31 | 2013-05-08 | 杏林制药株式会社 | 以咪达那新为有效成分的口腔内速崩片的制造方法 |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS6248618A (ja) * | 1985-08-27 | 1987-03-03 | Zeria Shinyaku Kogyo Kk | 徐放性製剤およびその製造法 |
| WO2001034147A1 (fr) * | 1999-11-11 | 2001-05-17 | Kyorin Pharmaceutical Co., Ltd. | Preparation solide orale |
| JP2001172181A (ja) * | 1999-12-17 | 2001-06-26 | Adir | 経口投与後のトリメタジジンの持続性放出を可能とするマトリックス錠 |
| WO2002000219A1 (de) * | 2000-06-26 | 2002-01-03 | Bayer Aktiengesellschaft | Retard-zubereitungen von chinolon-antibiotika und verfahren zu ihrer herstellung |
Family Cites Families (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4571333A (en) * | 1983-06-14 | 1986-02-18 | Syntex (U.S.A.) Inc. | Controlled release naproxen and naproxen sodium tablets |
| US4734285A (en) * | 1985-10-28 | 1988-03-29 | The Dow Chemical Company | Sustained release compositions |
| FR2677886B1 (fr) * | 1991-06-18 | 1995-03-31 | Adir | Comprime matriciel permettant la liberation prolongee d'indapamide apres administration par voie orale. |
| JP3294961B2 (ja) | 1993-12-10 | 2002-06-24 | 杏林製薬株式会社 | 新規イミダゾール誘導体及びその製造法 |
| US5399359A (en) * | 1994-03-04 | 1995-03-21 | Edward Mendell Co., Inc. | Controlled release oxybutynin formulations |
| JPH1067657A (ja) * | 1996-06-18 | 1998-03-10 | Otsuka Pharmaceut Co Ltd | マルチプルユニット型持続性製剤 |
| WO2001076557A1 (fr) * | 2000-04-10 | 2001-10-18 | Sumitomo Pharmaceuticals Co., Ltd. | Preparations a liberation prolongee |
-
2004
- 2004-08-03 WO PCT/JP2004/011067 patent/WO2005011682A1/ja active Application Filing
- 2004-08-03 US US10/566,503 patent/US20070092566A1/en not_active Abandoned
- 2004-08-03 EP EP04748200A patent/EP1652522A4/en not_active Withdrawn
- 2004-08-03 JP JP2005512546A patent/JP4884772B2/ja not_active Expired - Fee Related
- 2004-08-03 KR KR1020067002426A patent/KR101175816B1/ko not_active IP Right Cessation
- 2004-08-03 CN CNA2004800221865A patent/CN1842333A/zh active Pending
- 2004-08-03 CA CA2532714A patent/CA2532714C/en not_active Expired - Fee Related
-
2010
- 2010-08-12 US US12/855,226 patent/US8343544B2/en not_active Expired - Fee Related
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS6248618A (ja) * | 1985-08-27 | 1987-03-03 | Zeria Shinyaku Kogyo Kk | 徐放性製剤およびその製造法 |
| WO2001034147A1 (fr) * | 1999-11-11 | 2001-05-17 | Kyorin Pharmaceutical Co., Ltd. | Preparation solide orale |
| JP2001172181A (ja) * | 1999-12-17 | 2001-06-26 | Adir | 経口投与後のトリメタジジンの持続性放出を可能とするマトリックス錠 |
| WO2002000219A1 (de) * | 2000-06-26 | 2002-01-03 | Bayer Aktiengesellschaft | Retard-zubereitungen von chinolon-antibiotika und verfahren zu ihrer herstellung |
Also Published As
| Publication number | Publication date |
|---|---|
| KR101175816B1 (ko) | 2012-08-24 |
| US20100305179A1 (en) | 2010-12-02 |
| KR20060130006A (ko) | 2006-12-18 |
| WO2005011682A1 (ja) | 2005-02-10 |
| US8343544B2 (en) | 2013-01-01 |
| US20070092566A1 (en) | 2007-04-26 |
| CN1842333A (zh) | 2006-10-04 |
| EP1652522A4 (en) | 2008-11-26 |
| CA2532714C (en) | 2010-11-16 |
| EP1652522A1 (en) | 2006-05-03 |
| CA2532714A1 (en) | 2005-02-10 |
| JPWO2005011682A1 (ja) | 2007-10-04 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP0280571B1 (en) | Sustained release matrix formulations | |
| JP5199084B2 (ja) | 胃保持型製剤およびそれらの製造方法 | |
| CN106943355B (zh) | 药物组合物 | |
| JP2005508331A (ja) | 糖尿病の処置のための投与製剤 | |
| KR100780553B1 (ko) | 메트포르민 서방정 및 그의 제조방법 | |
| WO2011111818A1 (ja) | モサプリドまたはその塩を含む徐放型医薬組成物 | |
| JPWO2006080481A1 (ja) | マルチプルユニット型経口徐放性製剤及びその製造方法 | |
| HU215955B (hu) | Tramadolsót tartalmazó, nyújtott hatóanyag-leadású gyógyászati készítmény | |
| JP2011521927A (ja) | ブリバラセタムを含む医薬組成物 | |
| CN113018273A (zh) | 一种固体制剂及其制备方法和用途 | |
| US8343544B2 (en) | Oral sustained-release tablet | |
| KR101485421B1 (ko) | 이토프라이드 염산염을 함유하는 제어 방출성 경구 제제의 조성물 및 그의 제조방법 | |
| JP2016117738A (ja) | シロドシン−シクロデキストリン包接化合物 | |
| TW202038917A (zh) | 包含托法替尼或其藥學上可接受的鹽類的延長釋放配方及其製備方法 | |
| CN115400128B (zh) | 含盐酸去氧肾上腺素片剂、制备方法及应用 | |
| KR20210081290A (ko) | 메트포르민 서방성 제제 및 이의 제조 방법 | |
| JP2010001242A (ja) | レバミピド固形製剤及びその製造方法 | |
| JPWO2004091600A1 (ja) | 経口固形製剤 | |
| JP2016539109A (ja) | イソニアジドの顆粒およびリファペンチンの顆粒を含むコーティング錠の形態の抗結核性の安定な医薬組成物ならびにその製造方法 | |
| JP4696210B2 (ja) | イソソルビド‐5‐モノニトレートを有効成分とする徐放性錠剤及びその製造方法 | |
| KR20160141045A (ko) | 보센탄을 함유한 약학적 조성물 | |
| JP2022140430A (ja) | リバーロキサバン含有錠剤 | |
| CN114983957A (zh) | 一种艾拉戈克钠缓控释渗透装置及其制备方法 | |
| KR101446066B1 (ko) | 구안파신 함유 경구용 서방성 캡슐제 조성물 | |
| KR101431232B1 (ko) | 당뇨병 치료를 위한 약제학적 복합 제제 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20070723 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20101026 |
|
| A521 | Written amendment |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20101227 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20110201 |
|
| A521 | Written amendment |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20110325 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20111108 |
|
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20111207 |
|
| FPAY | Renewal fee payment (event date is renewal date of database) |
Free format text: PAYMENT UNTIL: 20141216 Year of fee payment: 3 |
|
| R150 | Certificate of patent or registration of utility model |
Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| S531 | Written request for registration of change of domicile |
Free format text: JAPANESE INTERMEDIATE CODE: R313531 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| LAPS | Cancellation because of no payment of annual fees |