JP2020194792A - 特性が変化するナノ複合電池電極粒子 - Google Patents
特性が変化するナノ複合電池電極粒子 Download PDFInfo
- Publication number
- JP2020194792A JP2020194792A JP2020139694A JP2020139694A JP2020194792A JP 2020194792 A JP2020194792 A JP 2020194792A JP 2020139694 A JP2020139694 A JP 2020139694A JP 2020139694 A JP2020139694 A JP 2020139694A JP 2020194792 A JP2020194792 A JP 2020194792A
- Authority
- JP
- Japan
- Prior art keywords
- active material
- particles
- matrix material
- skeletal
- matrix
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
- 239000002245 particle Substances 0.000 title claims description 460
- 239000002114 nanocomposite Substances 0.000 title description 3
- 239000011159 matrix material Substances 0.000 claims abstract description 461
- 239000011149 active material Substances 0.000 claims abstract description 449
- 239000011246 composite particle Substances 0.000 claims abstract description 228
- 239000000203 mixture Substances 0.000 claims abstract description 192
- 239000011148 porous material Substances 0.000 claims abstract description 190
- 239000000463 material Substances 0.000 claims abstract description 131
- 238000000034 method Methods 0.000 claims abstract description 95
- 150000002500 ions Chemical class 0.000 claims abstract description 38
- 239000003792 electrolyte Substances 0.000 claims description 83
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 claims description 79
- 229910052799 carbon Inorganic materials 0.000 claims description 63
- 239000002904 solvent Substances 0.000 claims description 52
- 238000000151 deposition Methods 0.000 claims description 24
- 229910052710 silicon Inorganic materials 0.000 claims description 22
- 230000008021 deposition Effects 0.000 claims description 20
- 239000007773 negative electrode material Substances 0.000 claims description 17
- 239000007774 positive electrode material Substances 0.000 claims description 16
- 230000007547 defect Effects 0.000 claims description 15
- 229910021645 metal ion Inorganic materials 0.000 claims description 14
- 238000004519 manufacturing process Methods 0.000 claims description 13
- 239000010703 silicon Substances 0.000 claims description 13
- 238000009826 distribution Methods 0.000 claims description 12
- 230000007423 decrease Effects 0.000 claims description 11
- 125000000524 functional group Chemical group 0.000 claims description 11
- 230000002093 peripheral effect Effects 0.000 claims description 11
- 230000006911 nucleation Effects 0.000 claims description 10
- 238000010899 nucleation Methods 0.000 claims description 10
- 229920001940 conductive polymer Polymers 0.000 claims description 8
- 230000002829 reductive effect Effects 0.000 claims description 8
- 230000001747 exhibiting effect Effects 0.000 claims description 3
- 229910010293 ceramic material Inorganic materials 0.000 claims description 2
- 229910052731 fluorine Inorganic materials 0.000 claims description 2
- 239000011737 fluorine Substances 0.000 claims description 2
- PXGOKWXKJXAPGV-UHFFFAOYSA-N Fluorine Chemical compound FF PXGOKWXKJXAPGV-UHFFFAOYSA-N 0.000 claims 1
- 230000004888 barrier function Effects 0.000 claims 1
- 239000012141 concentrate Substances 0.000 claims 1
- 230000003746 surface roughness Effects 0.000 claims 1
- 238000013461 design Methods 0.000 abstract description 56
- 238000004146 energy storage Methods 0.000 abstract description 4
- 239000006183 anode active material Substances 0.000 abstract 1
- 239000006182 cathode active material Substances 0.000 abstract 1
- 239000002243 precursor Substances 0.000 description 173
- 239000010410 layer Substances 0.000 description 108
- 239000011257 shell material Substances 0.000 description 103
- 239000002105 nanoparticle Substances 0.000 description 83
- 229910001416 lithium ion Inorganic materials 0.000 description 66
- 230000008859 change Effects 0.000 description 65
- 239000000725 suspension Substances 0.000 description 53
- 239000002131 composite material Substances 0.000 description 48
- 229920000642 polymer Polymers 0.000 description 48
- 229910052751 metal Inorganic materials 0.000 description 43
- 239000002184 metal Substances 0.000 description 43
- 230000015572 biosynthetic process Effects 0.000 description 39
- 229910052744 lithium Inorganic materials 0.000 description 38
- 238000006243 chemical reaction Methods 0.000 description 34
- 238000010586 diagram Methods 0.000 description 33
- 239000007784 solid electrolyte Substances 0.000 description 32
- WHXSMMKQMYFTQS-UHFFFAOYSA-N Lithium Chemical compound [Li] WHXSMMKQMYFTQS-UHFFFAOYSA-N 0.000 description 29
- 238000000576 coating method Methods 0.000 description 28
- 150000003839 salts Chemical class 0.000 description 28
- 239000000243 solution Substances 0.000 description 28
- 230000001681 protective effect Effects 0.000 description 27
- 238000010438 heat treatment Methods 0.000 description 26
- 239000011248 coating agent Substances 0.000 description 25
- AMXOYNBUYSYVKV-UHFFFAOYSA-M lithium bromide Chemical compound [Li+].[Br-] AMXOYNBUYSYVKV-UHFFFAOYSA-M 0.000 description 25
- 239000011244 liquid electrolyte Substances 0.000 description 21
- 238000005229 chemical vapour deposition Methods 0.000 description 20
- 230000008018 melting Effects 0.000 description 19
- 238000002844 melting Methods 0.000 description 19
- 150000002739 metals Chemical class 0.000 description 19
- 238000001994 activation Methods 0.000 description 18
- 230000008569 process Effects 0.000 description 18
- HSZCZNFXUDYRKD-UHFFFAOYSA-M lithium iodide Chemical compound [Li+].[I-] HSZCZNFXUDYRKD-UHFFFAOYSA-M 0.000 description 17
- 239000002344 surface layer Substances 0.000 description 17
- 238000003786 synthesis reaction Methods 0.000 description 17
- -1 CuF 2 and FeF 3 ) Chemical class 0.000 description 16
- 230000004913 activation Effects 0.000 description 15
- 239000011133 lead Substances 0.000 description 15
- 239000000843 powder Substances 0.000 description 15
- 239000000126 substance Substances 0.000 description 15
- PQXKHYXIUOZZFA-UHFFFAOYSA-M lithium fluoride Chemical compound [Li+].[F-] PQXKHYXIUOZZFA-UHFFFAOYSA-M 0.000 description 14
- 239000011241 protective layer Substances 0.000 description 14
- 150000004820 halides Chemical class 0.000 description 13
- 230000003993 interaction Effects 0.000 description 13
- 239000007788 liquid Substances 0.000 description 13
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical compound [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 description 12
- 230000001939 inductive effect Effects 0.000 description 12
- 229910052760 oxygen Inorganic materials 0.000 description 12
- 239000011135 tin Substances 0.000 description 12
- 230000002776 aggregation Effects 0.000 description 11
- 239000000956 alloy Substances 0.000 description 11
- 229910045601 alloy Inorganic materials 0.000 description 11
- 239000011230 binding agent Substances 0.000 description 11
- 230000001965 increasing effect Effects 0.000 description 11
- 229910001415 sodium ion Inorganic materials 0.000 description 11
- 239000007787 solid Substances 0.000 description 11
- 230000001976 improved effect Effects 0.000 description 10
- 229910001512 metal fluoride Inorganic materials 0.000 description 10
- 229910052718 tin Inorganic materials 0.000 description 10
- 238000004220 aggregation Methods 0.000 description 9
- 238000013459 approach Methods 0.000 description 9
- 230000009286 beneficial effect Effects 0.000 description 9
- 238000007740 vapor deposition Methods 0.000 description 9
- 150000004645 aluminates Chemical class 0.000 description 8
- 229910052782 aluminium Inorganic materials 0.000 description 8
- 230000008901 benefit Effects 0.000 description 8
- 238000007599 discharging Methods 0.000 description 8
- 239000007772 electrode material Substances 0.000 description 8
- 238000000605 extraction Methods 0.000 description 8
- KWGKDLIKAYFUFQ-UHFFFAOYSA-M lithium chloride Chemical compound [Li+].[Cl-] KWGKDLIKAYFUFQ-UHFFFAOYSA-M 0.000 description 8
- 239000011777 magnesium Substances 0.000 description 8
- 230000003647 oxidation Effects 0.000 description 8
- 238000007254 oxidation reaction Methods 0.000 description 8
- 230000036961 partial effect Effects 0.000 description 8
- 230000009467 reduction Effects 0.000 description 8
- 230000002194 synthesizing effect Effects 0.000 description 8
- KRHYYFGTRYWZRS-UHFFFAOYSA-M Fluoride anion Chemical compound [F-] KRHYYFGTRYWZRS-UHFFFAOYSA-M 0.000 description 7
- 238000003763 carbonization Methods 0.000 description 7
- 230000006870 function Effects 0.000 description 7
- 230000008595 infiltration Effects 0.000 description 7
- 238000001764 infiltration Methods 0.000 description 7
- 229910052749 magnesium Inorganic materials 0.000 description 7
- 229910044991 metal oxide Inorganic materials 0.000 description 7
- 150000004706 metal oxides Chemical class 0.000 description 7
- 150000004767 nitrides Chemical class 0.000 description 7
- 230000000087 stabilizing effect Effects 0.000 description 7
- 238000003860 storage Methods 0.000 description 7
- 229910013870 LiPF 6 Inorganic materials 0.000 description 6
- 229910019142 PO4 Inorganic materials 0.000 description 6
- 238000000137 annealing Methods 0.000 description 6
- 239000010949 copper Substances 0.000 description 6
- 238000000354 decomposition reaction Methods 0.000 description 6
- 238000009792 diffusion process Methods 0.000 description 6
- 238000003487 electrochemical reaction Methods 0.000 description 6
- 238000011049 filling Methods 0.000 description 6
- 230000006872 improvement Effects 0.000 description 6
- 238000003780 insertion Methods 0.000 description 6
- 230000037431 insertion Effects 0.000 description 6
- 239000000178 monomer Substances 0.000 description 6
- 230000000149 penetrating effect Effects 0.000 description 6
- 239000012071 phase Substances 0.000 description 6
- 235000021317 phosphate Nutrition 0.000 description 6
- BTBUEUYNUDRHOZ-UHFFFAOYSA-N Borate Chemical compound [O-]B([O-])[O-] BTBUEUYNUDRHOZ-UHFFFAOYSA-N 0.000 description 5
- 229910016509 CuF 2 Inorganic materials 0.000 description 5
- 229910052787 antimony Inorganic materials 0.000 description 5
- WATWJIUSRGPENY-UHFFFAOYSA-N antimony atom Chemical compound [Sb] WATWJIUSRGPENY-UHFFFAOYSA-N 0.000 description 5
- 238000000231 atomic layer deposition Methods 0.000 description 5
- 239000003054 catalyst Substances 0.000 description 5
- 239000000084 colloidal system Substances 0.000 description 5
- 230000006866 deterioration Effects 0.000 description 5
- 238000004090 dissolution Methods 0.000 description 5
- 238000001035 drying Methods 0.000 description 5
- 230000000694 effects Effects 0.000 description 5
- 229910021480 group 4 element Inorganic materials 0.000 description 5
- 238000009830 intercalation Methods 0.000 description 5
- 230000007246 mechanism Effects 0.000 description 5
- 239000002070 nanowire Substances 0.000 description 5
- 230000035515 penetration Effects 0.000 description 5
- NBIIXXVUZAFLBC-UHFFFAOYSA-K phosphate Chemical compound [O-]P([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-K 0.000 description 5
- 239000010452 phosphate Substances 0.000 description 5
- 239000002002 slurry Substances 0.000 description 5
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 5
- 239000011701 zinc Substances 0.000 description 5
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 4
- 229910013063 LiBF 4 Inorganic materials 0.000 description 4
- UCKMPCXJQFINFW-UHFFFAOYSA-N Sulphide Chemical compound [S-2] UCKMPCXJQFINFW-UHFFFAOYSA-N 0.000 description 4
- 239000007983 Tris buffer Substances 0.000 description 4
- 230000003213 activating effect Effects 0.000 description 4
- 239000007833 carbon precursor Substances 0.000 description 4
- 239000003660 carbonate based solvent Substances 0.000 description 4
- 230000015556 catabolic process Effects 0.000 description 4
- 239000011258 core-shell material Substances 0.000 description 4
- 238000006731 degradation reaction Methods 0.000 description 4
- 238000005516 engineering process Methods 0.000 description 4
- 150000002148 esters Chemical class 0.000 description 4
- 229910002804 graphite Inorganic materials 0.000 description 4
- 239000010439 graphite Substances 0.000 description 4
- 230000002687 intercalation Effects 0.000 description 4
- XEEYBQQBJWHFJM-UHFFFAOYSA-N iron Substances [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 description 4
- 229910001507 metal halide Inorganic materials 0.000 description 4
- 150000005309 metal halides Chemical class 0.000 description 4
- 238000000465 moulding Methods 0.000 description 4
- 150000002825 nitriles Chemical class 0.000 description 4
- 238000012856 packing Methods 0.000 description 4
- 229910052698 phosphorus Inorganic materials 0.000 description 4
- 238000006116 polymerization reaction Methods 0.000 description 4
- 238000001556 precipitation Methods 0.000 description 4
- 238000007789 sealing Methods 0.000 description 4
- VEXZGXHMUGYJMC-UHFFFAOYSA-M Chloride anion Chemical compound [Cl-] VEXZGXHMUGYJMC-UHFFFAOYSA-M 0.000 description 3
- 229910015475 FeF 2 Inorganic materials 0.000 description 3
- 229910013684 LiClO 4 Inorganic materials 0.000 description 3
- 229910002651 NO3 Inorganic materials 0.000 description 3
- NHNBFGGVMKEFGY-UHFFFAOYSA-N Nitrate Chemical compound [O-][N+]([O-])=O NHNBFGGVMKEFGY-UHFFFAOYSA-N 0.000 description 3
- OAICVXFJPJFONN-UHFFFAOYSA-N Phosphorus Chemical compound [P] OAICVXFJPJFONN-UHFFFAOYSA-N 0.000 description 3
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 3
- 238000005275 alloying Methods 0.000 description 3
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 3
- 239000012080 ambient air Substances 0.000 description 3
- 125000004429 atom Chemical group 0.000 description 3
- 229910052793 cadmium Inorganic materials 0.000 description 3
- 125000004432 carbon atom Chemical group C* 0.000 description 3
- 239000002482 conductive additive Substances 0.000 description 3
- 239000004020 conductor Substances 0.000 description 3
- 229910052802 copper Inorganic materials 0.000 description 3
- 230000006378 damage Effects 0.000 description 3
- 230000003247 decreasing effect Effects 0.000 description 3
- 239000004210 ether based solvent Substances 0.000 description 3
- 238000001704 evaporation Methods 0.000 description 3
- 239000000284 extract Substances 0.000 description 3
- 230000002349 favourable effect Effects 0.000 description 3
- 239000007789 gas Substances 0.000 description 3
- 229910052732 germanium Inorganic materials 0.000 description 3
- 229910021478 group 5 element Inorganic materials 0.000 description 3
- 150000004678 hydrides Chemical class 0.000 description 3
- 229910052739 hydrogen Inorganic materials 0.000 description 3
- 229910052738 indium Inorganic materials 0.000 description 3
- 229910052742 iron Inorganic materials 0.000 description 3
- 230000007774 longterm Effects 0.000 description 3
- 229910001510 metal chloride Inorganic materials 0.000 description 3
- 239000002060 nanoflake Substances 0.000 description 3
- 239000002071 nanotube Substances 0.000 description 3
- 150000002823 nitrates Chemical class 0.000 description 3
- 150000002826 nitrites Chemical class 0.000 description 3
- 230000037361 pathway Effects 0.000 description 3
- 239000012466 permeate Substances 0.000 description 3
- 239000011574 phosphorus Substances 0.000 description 3
- 239000004033 plastic Substances 0.000 description 3
- 229920002959 polymer blend Polymers 0.000 description 3
- 239000005518 polymer electrolyte Substances 0.000 description 3
- 239000011253 protective coating Substances 0.000 description 3
- 238000000926 separation method Methods 0.000 description 3
- 238000007086 side reaction Methods 0.000 description 3
- 238000005245 sintering Methods 0.000 description 3
- 238000001179 sorption measurement Methods 0.000 description 3
- 230000032258 transport Effects 0.000 description 3
- 238000011282 treatment Methods 0.000 description 3
- 229910052725 zinc Inorganic materials 0.000 description 3
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 2
- 229910021594 Copper(II) fluoride Inorganic materials 0.000 description 2
- 229910000733 Li alloy Inorganic materials 0.000 description 2
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 2
- 229910020923 Sn-O Inorganic materials 0.000 description 2
- ATJFFYVFTNAWJD-UHFFFAOYSA-N Tin Chemical compound [Sn] ATJFFYVFTNAWJD-UHFFFAOYSA-N 0.000 description 2
- 238000005054 agglomeration Methods 0.000 description 2
- 239000003570 air Substances 0.000 description 2
- 229910052783 alkali metal Inorganic materials 0.000 description 2
- 150000001340 alkali metals Chemical class 0.000 description 2
- 229910052785 arsenic Inorganic materials 0.000 description 2
- RQNWIZPPADIBDY-UHFFFAOYSA-N arsenic atom Chemical compound [As] RQNWIZPPADIBDY-UHFFFAOYSA-N 0.000 description 2
- 229910021383 artificial graphite Inorganic materials 0.000 description 2
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 2
- 229910052788 barium Inorganic materials 0.000 description 2
- 229910002056 binary alloy Inorganic materials 0.000 description 2
- 229910052797 bismuth Inorganic materials 0.000 description 2
- JCXGWMGPZLAOME-UHFFFAOYSA-N bismuth atom Chemical compound [Bi] JCXGWMGPZLAOME-UHFFFAOYSA-N 0.000 description 2
- 229920001400 block copolymer Polymers 0.000 description 2
- 229910052792 caesium Inorganic materials 0.000 description 2
- 229910052791 calcium Inorganic materials 0.000 description 2
- 239000003575 carbonaceous material Substances 0.000 description 2
- 230000022131 cell cycle Effects 0.000 description 2
- 239000000919 ceramic Substances 0.000 description 2
- 238000005234 chemical deposition Methods 0.000 description 2
- GWFAVIIMQDUCRA-UHFFFAOYSA-L copper(ii) fluoride Chemical compound [F-].[F-].[Cu+2] GWFAVIIMQDUCRA-UHFFFAOYSA-L 0.000 description 2
- 238000005530 etching Methods 0.000 description 2
- 230000008020 evaporation Effects 0.000 description 2
- 229910052733 gallium Inorganic materials 0.000 description 2
- GNPVGFCGXDBREM-UHFFFAOYSA-N germanium atom Chemical compound [Ge] GNPVGFCGXDBREM-UHFFFAOYSA-N 0.000 description 2
- 229910052735 hafnium Inorganic materials 0.000 description 2
- 239000011261 inert gas Substances 0.000 description 2
- 230000037427 ion transport Effects 0.000 description 2
- SHXXPRJOPFJRHA-UHFFFAOYSA-K iron(iii) fluoride Chemical compound F[Fe](F)F SHXXPRJOPFJRHA-UHFFFAOYSA-K 0.000 description 2
- 230000001788 irregular Effects 0.000 description 2
- 230000002427 irreversible effect Effects 0.000 description 2
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 2
- 229910052746 lanthanum Inorganic materials 0.000 description 2
- 239000001989 lithium alloy Substances 0.000 description 2
- YQNQTEBHHUSESQ-UHFFFAOYSA-N lithium aluminate Chemical compound [Li+].[O-][Al]=O YQNQTEBHHUSESQ-UHFFFAOYSA-N 0.000 description 2
- 239000002609 medium Substances 0.000 description 2
- 229910001509 metal bromide Inorganic materials 0.000 description 2
- 229910001092 metal group alloy Inorganic materials 0.000 description 2
- 229910001511 metal iodide Inorganic materials 0.000 description 2
- 239000002082 metal nanoparticle Substances 0.000 description 2
- 238000003801 milling Methods 0.000 description 2
- 229910052759 nickel Inorganic materials 0.000 description 2
- PXHVJJICTQNCMI-UHFFFAOYSA-N nickel Substances [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 description 2
- 239000005486 organic electrolyte Substances 0.000 description 2
- JPJBEORAVWZJKS-UHFFFAOYSA-N oxalic acid;propanedioic acid Chemical compound OC(=O)C(O)=O.OC(=O)CC(O)=O JPJBEORAVWZJKS-UHFFFAOYSA-N 0.000 description 2
- 230000001590 oxidative effect Effects 0.000 description 2
- 239000001301 oxygen Substances 0.000 description 2
- 125000005003 perfluorobutyl group Chemical group FC(F)(F)C(F)(F)C(F)(F)C(F)(F)* 0.000 description 2
- 125000005004 perfluoroethyl group Chemical group FC(F)(F)C(F)(F)* 0.000 description 2
- 238000000623 plasma-assisted chemical vapour deposition Methods 0.000 description 2
- 229920000447 polyanionic polymer Polymers 0.000 description 2
- 239000002244 precipitate Substances 0.000 description 2
- 230000002265 prevention Effects 0.000 description 2
- 229910052761 rare earth metal Inorganic materials 0.000 description 2
- 230000004044 response Effects 0.000 description 2
- JHJLBTNAGRQEKS-UHFFFAOYSA-M sodium bromide Chemical compound [Na+].[Br-] JHJLBTNAGRQEKS-UHFFFAOYSA-M 0.000 description 2
- 239000006104 solid solution Substances 0.000 description 2
- 229910052712 strontium Inorganic materials 0.000 description 2
- 150000003457 sulfones Chemical class 0.000 description 2
- 150000003462 sulfoxides Chemical class 0.000 description 2
- 229910052723 transition metal Inorganic materials 0.000 description 2
- 150000003624 transition metals Chemical class 0.000 description 2
- PBIMIGNDTBRRPI-UHFFFAOYSA-N trifluoro borate Chemical compound FOB(OF)OF PBIMIGNDTBRRPI-UHFFFAOYSA-N 0.000 description 2
- TWQULNDIKKJZPH-UHFFFAOYSA-K trilithium;phosphate Chemical class [Li+].[Li+].[Li+].[O-]P([O-])([O-])=O TWQULNDIKKJZPH-UHFFFAOYSA-K 0.000 description 2
- LENZDBCJOHFCAS-UHFFFAOYSA-N tris Chemical compound OCC(N)(CO)CO LENZDBCJOHFCAS-UHFFFAOYSA-N 0.000 description 2
- 230000005641 tunneling Effects 0.000 description 2
- 239000012808 vapor phase Substances 0.000 description 2
- 229910052727 yttrium Inorganic materials 0.000 description 2
- JIAARYAFYJHUJI-UHFFFAOYSA-L zinc dichloride Chemical compound [Cl-].[Cl-].[Zn+2] JIAARYAFYJHUJI-UHFFFAOYSA-L 0.000 description 2
- 229910052726 zirconium Inorganic materials 0.000 description 2
- 229910017982 Ag—Si Inorganic materials 0.000 description 1
- 229910018125 Al-Si Inorganic materials 0.000 description 1
- 229910018520 Al—Si Inorganic materials 0.000 description 1
- 229910015249 Ba—Si Inorganic materials 0.000 description 1
- 229910016292 BiF 5 Inorganic materials 0.000 description 1
- ZOXJGFHDIHLPTG-UHFFFAOYSA-N Boron Chemical compound [B] ZOXJGFHDIHLPTG-UHFFFAOYSA-N 0.000 description 1
- CPELXLSAUQHCOX-UHFFFAOYSA-M Bromide Chemical compound [Br-] CPELXLSAUQHCOX-UHFFFAOYSA-M 0.000 description 1
- JQIZJWJHAZEXCX-UHFFFAOYSA-H C(C(=O)[O-])(=O)[O-].C(C(=O)[O-])(=O)[O-].C(C(=O)[O-])(=O)[O-].[Li+].[Li+].[Li+].[Li+].[Li+].[Li+] Chemical compound C(C(=O)[O-])(=O)[O-].C(C(=O)[O-])(=O)[O-].C(C(=O)[O-])(=O)[O-].[Li+].[Li+].[Li+].[Li+].[Li+].[Li+] JQIZJWJHAZEXCX-UHFFFAOYSA-H 0.000 description 1
- VPIDXLJVGVBFOW-UHFFFAOYSA-N C=1C=[C-]PC=1 Chemical class C=1C=[C-]PC=1 VPIDXLJVGVBFOW-UHFFFAOYSA-N 0.000 description 1
- 229910014458 Ca-Si Inorganic materials 0.000 description 1
- 229920000049 Carbon (fiber) Polymers 0.000 description 1
- 229910004547 Cd—Si Inorganic materials 0.000 description 1
- 229910021583 Cobalt(III) fluoride Inorganic materials 0.000 description 1
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 1
- 229910020711 Co—Si Inorganic materials 0.000 description 1
- 229910019819 Cr—Si Inorganic materials 0.000 description 1
- 229910017758 Cu-Si Inorganic materials 0.000 description 1
- 229910017931 Cu—Si Inorganic materials 0.000 description 1
- 229910017082 Fe-Si Inorganic materials 0.000 description 1
- 229910017133 Fe—Si Inorganic materials 0.000 description 1
- YCKRFDGAMUMZLT-UHFFFAOYSA-N Fluorine atom Chemical compound [F] YCKRFDGAMUMZLT-UHFFFAOYSA-N 0.000 description 1
- GYHNNYVSQQEPJS-UHFFFAOYSA-N Gallium Chemical compound [Ga] GYHNNYVSQQEPJS-UHFFFAOYSA-N 0.000 description 1
- 229910003839 Hf—Si Inorganic materials 0.000 description 1
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 1
- 229910018068 Li 2 O Inorganic materials 0.000 description 1
- 229910018091 Li 2 S Inorganic materials 0.000 description 1
- HBBGRARXTFLTSG-UHFFFAOYSA-N Lithium ion Chemical compound [Li+] HBBGRARXTFLTSG-UHFFFAOYSA-N 0.000 description 1
- 229910021380 Manganese Chloride Inorganic materials 0.000 description 1
- GLFNIEUTAYBVOC-UHFFFAOYSA-L Manganese chloride Chemical compound Cl[Mn]Cl GLFNIEUTAYBVOC-UHFFFAOYSA-L 0.000 description 1
- 229910019064 Mg-Si Inorganic materials 0.000 description 1
- 229910019406 Mg—Si Inorganic materials 0.000 description 1
- 229910018643 Mn—Si Inorganic materials 0.000 description 1
- 229910017305 Mo—Si Inorganic materials 0.000 description 1
- 241000549556 Nanos Species 0.000 description 1
- 229910020010 Nb—Si Inorganic materials 0.000 description 1
- 229910018098 Ni-Si Inorganic materials 0.000 description 1
- 229910018529 Ni—Si Inorganic materials 0.000 description 1
- AUBNQVSSTJZVMY-UHFFFAOYSA-M P(=O)([O-])(O)O.C(C(=O)O)(=O)F.C(C(=O)O)(=O)F.C(C(=O)O)(=O)F.C(C(=O)O)(=O)F.[Li+] Chemical compound P(=O)([O-])(O)O.C(C(=O)O)(=O)F.C(C(=O)O)(=O)F.C(C(=O)O)(=O)F.C(C(=O)O)(=O)F.[Li+] AUBNQVSSTJZVMY-UHFFFAOYSA-M 0.000 description 1
- 229910021074 Pd—Si Inorganic materials 0.000 description 1
- 229910019596 Rh—Si Inorganic materials 0.000 description 1
- 229910018287 SbF 5 Inorganic materials 0.000 description 1
- BUGBHKTXTAQXES-UHFFFAOYSA-N Selenium Chemical compound [Se] BUGBHKTXTAQXES-UHFFFAOYSA-N 0.000 description 1
- 229910007981 Si-Mg Inorganic materials 0.000 description 1
- 229910008316 Si—Mg Inorganic materials 0.000 description 1
- 229910001128 Sn alloy Inorganic materials 0.000 description 1
- 229910008449 SnF 2 Inorganic materials 0.000 description 1
- 229910003526 Sr—Si Inorganic materials 0.000 description 1
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 1
- 229930006000 Sucrose Natural products 0.000 description 1
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 description 1
- 229910004305 Tc—Si Inorganic materials 0.000 description 1
- 229910004339 Ti-Si Inorganic materials 0.000 description 1
- 229910010978 Ti—Si Inorganic materials 0.000 description 1
- 229910008938 W—Si Inorganic materials 0.000 description 1
- 229910009257 Y—Si Inorganic materials 0.000 description 1
- 229910007735 Zr—Si Inorganic materials 0.000 description 1
- IYOHHZVNHNCZNL-UHFFFAOYSA-N [Fe].FOF Chemical compound [Fe].FOF IYOHHZVNHNCZNL-UHFFFAOYSA-N 0.000 description 1
- HIVGXUNKSAJJDN-UHFFFAOYSA-N [Si].[P] Chemical compound [Si].[P] HIVGXUNKSAJJDN-UHFFFAOYSA-N 0.000 description 1
- BTFOWJRRWDOUKQ-UHFFFAOYSA-N [Si]=O.[Sn] Chemical compound [Si]=O.[Sn] BTFOWJRRWDOUKQ-UHFFFAOYSA-N 0.000 description 1
- 238000009825 accumulation Methods 0.000 description 1
- 230000009471 action Effects 0.000 description 1
- 239000012190 activator Substances 0.000 description 1
- 239000013543 active substance Substances 0.000 description 1
- 230000006978 adaptation Effects 0.000 description 1
- 239000011157 advanced composite material Substances 0.000 description 1
- 239000003125 aqueous solvent Substances 0.000 description 1
- 239000002585 base Substances 0.000 description 1
- 238000005452 bending Methods 0.000 description 1
- IPNGSXQUQIUWKO-UHFFFAOYSA-N bismuth;fluoro hypofluorite Chemical compound [Bi].FOF IPNGSXQUQIUWKO-UHFFFAOYSA-N 0.000 description 1
- 229910052796 boron Inorganic materials 0.000 description 1
- YKYOUMDCQGMQQO-UHFFFAOYSA-L cadmium dichloride Chemical compound Cl[Cd]Cl YKYOUMDCQGMQQO-UHFFFAOYSA-L 0.000 description 1
- LVEULQCPJDDSLD-UHFFFAOYSA-L cadmium fluoride Chemical compound F[Cd]F LVEULQCPJDDSLD-UHFFFAOYSA-L 0.000 description 1
- 229910001424 calcium ion Inorganic materials 0.000 description 1
- 239000006229 carbon black Substances 0.000 description 1
- 239000004917 carbon fiber Substances 0.000 description 1
- 238000006555 catalytic reaction Methods 0.000 description 1
- 150000004770 chalcogenides Chemical class 0.000 description 1
- 239000007795 chemical reaction product Substances 0.000 description 1
- 238000005660 chlorination reaction Methods 0.000 description 1
- 239000003245 coal Substances 0.000 description 1
- 239000011247 coating layer Substances 0.000 description 1
- GVPFVAHMJGGAJG-UHFFFAOYSA-L cobalt dichloride Chemical compound [Cl-].[Cl-].[Co+2] GVPFVAHMJGGAJG-UHFFFAOYSA-L 0.000 description 1
- YCYBZKSMUPTWEE-UHFFFAOYSA-L cobalt(ii) fluoride Chemical compound F[Co]F YCYBZKSMUPTWEE-UHFFFAOYSA-L 0.000 description 1
- 230000000295 complement effect Effects 0.000 description 1
- 150000001875 compounds Chemical class 0.000 description 1
- 238000009833 condensation Methods 0.000 description 1
- 230000005494 condensation Effects 0.000 description 1
- 239000000470 constituent Substances 0.000 description 1
- ORTQZVOHEJQUHG-UHFFFAOYSA-L copper(II) chloride Chemical compound Cl[Cu]Cl ORTQZVOHEJQUHG-UHFFFAOYSA-L 0.000 description 1
- 230000007797 corrosion Effects 0.000 description 1
- 238000005260 corrosion Methods 0.000 description 1
- 238000000280 densification Methods 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- ZACMZEXSXVCAIO-UHFFFAOYSA-N difluoro oxalate;lithium Chemical compound [Li].FOC(=O)C(=O)OF ZACMZEXSXVCAIO-UHFFFAOYSA-N 0.000 description 1
- FPHIOHCCQGUGKU-UHFFFAOYSA-L difluorolead Chemical compound F[Pb]F FPHIOHCCQGUGKU-UHFFFAOYSA-L 0.000 description 1
- SMBQBQBNOXIFSF-UHFFFAOYSA-N dilithium Chemical compound [Li][Li] SMBQBQBNOXIFSF-UHFFFAOYSA-N 0.000 description 1
- 239000002019 doping agent Substances 0.000 description 1
- 230000005489 elastic deformation Effects 0.000 description 1
- 230000005684 electric field Effects 0.000 description 1
- 230000005611 electricity Effects 0.000 description 1
- 238000012983 electrochemical energy storage Methods 0.000 description 1
- 238000004070 electrodeposition Methods 0.000 description 1
- 239000000839 emulsion Substances 0.000 description 1
- 150000002170 ethers Chemical class 0.000 description 1
- 230000005496 eutectics Effects 0.000 description 1
- 238000001125 extrusion Methods 0.000 description 1
- 239000000835 fiber Substances 0.000 description 1
- 239000000945 filler Substances 0.000 description 1
- 239000010419 fine particle Substances 0.000 description 1
- 239000011888 foil Substances 0.000 description 1
- 238000009472 formulation Methods 0.000 description 1
- 230000004927 fusion Effects 0.000 description 1
- 150000004676 glycans Chemical class 0.000 description 1
- 229910021389 graphene Inorganic materials 0.000 description 1
- 101150008103 hal gene Proteins 0.000 description 1
- 239000012456 homogeneous solution Substances 0.000 description 1
- 239000010903 husk Substances 0.000 description 1
- 239000001257 hydrogen Substances 0.000 description 1
- 125000004435 hydrogen atom Chemical group [H]* 0.000 description 1
- 238000010348 incorporation Methods 0.000 description 1
- APFVFJFRJDLVQX-UHFFFAOYSA-N indium atom Chemical compound [In] APFVFJFRJDLVQX-UHFFFAOYSA-N 0.000 description 1
- 229910010272 inorganic material Inorganic materials 0.000 description 1
- 239000011147 inorganic material Substances 0.000 description 1
- 230000016507 interphase Effects 0.000 description 1
- 239000002608 ionic liquid Substances 0.000 description 1
- FBAFATDZDUQKNH-UHFFFAOYSA-M iron chloride Chemical compound [Cl-].[Fe] FBAFATDZDUQKNH-UHFFFAOYSA-M 0.000 description 1
- 229910052745 lead Inorganic materials 0.000 description 1
- HWSZZLVAJGOAAY-UHFFFAOYSA-L lead(II) chloride Chemical compound Cl[Pb]Cl HWSZZLVAJGOAAY-UHFFFAOYSA-L 0.000 description 1
- 239000003077 lignite Substances 0.000 description 1
- 239000007791 liquid phase Substances 0.000 description 1
- AHKHZLVXUVZTGF-UHFFFAOYSA-M lithium dihydrogen phosphate oxalic acid Chemical compound P(=O)([O-])(O)O.C(C(=O)O)(=O)O.C(C(=O)O)(=O)O.C(C(=O)O)(=O)O.[Li+] AHKHZLVXUVZTGF-UHFFFAOYSA-M 0.000 description 1
- GELKBWJHTRAYNV-UHFFFAOYSA-K lithium iron phosphate Chemical compound [Li+].[Fe+2].[O-]P([O-])([O-])=O GELKBWJHTRAYNV-UHFFFAOYSA-K 0.000 description 1
- PEXNRZDEKZDXPZ-UHFFFAOYSA-N lithium selenidolithium Chemical compound [Li][Se][Li] PEXNRZDEKZDXPZ-UHFFFAOYSA-N 0.000 description 1
- GLNWILHOFOBOFD-UHFFFAOYSA-N lithium sulfide Chemical compound [Li+].[Li+].[S-2] GLNWILHOFOBOFD-UHFFFAOYSA-N 0.000 description 1
- GKWAQTFPHUTRMG-UHFFFAOYSA-N lithium telluride Chemical compound [Li][Te][Li] GKWAQTFPHUTRMG-UHFFFAOYSA-N 0.000 description 1
- 229910001425 magnesium ion Inorganic materials 0.000 description 1
- 235000002867 manganese chloride Nutrition 0.000 description 1
- 239000011565 manganese chloride Substances 0.000 description 1
- 229940099607 manganese chloride Drugs 0.000 description 1
- SRVINXWCFNHIQZ-UHFFFAOYSA-K manganese(iii) fluoride Chemical compound [F-].[F-].[F-].[Mn+3] SRVINXWCFNHIQZ-UHFFFAOYSA-K 0.000 description 1
- 239000000320 mechanical mixture Substances 0.000 description 1
- 239000012528 membrane Substances 0.000 description 1
- 239000002931 mesocarbon microbead Substances 0.000 description 1
- 150000001247 metal acetylides Chemical class 0.000 description 1
- 229910052987 metal hydride Inorganic materials 0.000 description 1
- 150000004681 metal hydrides Chemical class 0.000 description 1
- 229910000000 metal hydroxide Inorganic materials 0.000 description 1
- 150000004692 metal hydroxides Chemical class 0.000 description 1
- 229910021518 metal oxyhydroxide Inorganic materials 0.000 description 1
- 239000011834 metal-based active material Substances 0.000 description 1
- VNWKTOKETHGBQD-UHFFFAOYSA-N methane Chemical compound C VNWKTOKETHGBQD-UHFFFAOYSA-N 0.000 description 1
- 238000002156 mixing Methods 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 239000002121 nanofiber Substances 0.000 description 1
- 239000002135 nanosheet Substances 0.000 description 1
- QMMRZOWCJAIUJA-UHFFFAOYSA-L nickel dichloride Chemical compound Cl[Ni]Cl QMMRZOWCJAIUJA-UHFFFAOYSA-L 0.000 description 1
- DBJLJFTWODWSOF-UHFFFAOYSA-L nickel(ii) fluoride Chemical compound F[Ni]F DBJLJFTWODWSOF-UHFFFAOYSA-L 0.000 description 1
- 229910052757 nitrogen Inorganic materials 0.000 description 1
- 239000012457 nonaqueous media Substances 0.000 description 1
- 239000003960 organic solvent Substances 0.000 description 1
- 239000007800 oxidant agent Substances 0.000 description 1
- 239000003415 peat Substances 0.000 description 1
- 239000002006 petroleum coke Substances 0.000 description 1
- 150000003013 phosphoric acid derivatives Chemical class 0.000 description 1
- 239000011295 pitch Substances 0.000 description 1
- 101150100677 polo gene Proteins 0.000 description 1
- 229920005596 polymer binder Polymers 0.000 description 1
- 229920005597 polymer membrane Polymers 0.000 description 1
- 239000003505 polymerization initiator Substances 0.000 description 1
- 229920001282 polysaccharide Polymers 0.000 description 1
- 239000005017 polysaccharide Substances 0.000 description 1
- 230000001376 precipitating effect Effects 0.000 description 1
- 239000000047 product Substances 0.000 description 1
- 238000000197 pyrolysis Methods 0.000 description 1
- 230000009257 reactivity Effects 0.000 description 1
- 238000011946 reduction process Methods 0.000 description 1
- 230000002787 reinforcement Effects 0.000 description 1
- 230000000717 retained effect Effects 0.000 description 1
- 238000005096 rolling process Methods 0.000 description 1
- 229910052711 selenium Inorganic materials 0.000 description 1
- 239000011669 selenium Substances 0.000 description 1
- 229940065287 selenium compound Drugs 0.000 description 1
- 150000003343 selenium compounds Chemical class 0.000 description 1
- 150000004760 silicates Chemical class 0.000 description 1
- 239000000377 silicon dioxide Substances 0.000 description 1
- 229910052814 silicon oxide Inorganic materials 0.000 description 1
- 239000011827 silicon-based solvent Substances 0.000 description 1
- 229910052709 silver Inorganic materials 0.000 description 1
- 241000894007 species Species 0.000 description 1
- 239000012798 spherical particle Substances 0.000 description 1
- 238000001694 spray drying Methods 0.000 description 1
- 230000006641 stabilisation Effects 0.000 description 1
- 238000011105 stabilization Methods 0.000 description 1
- 239000010902 straw Substances 0.000 description 1
- 239000000758 substrate Substances 0.000 description 1
- 239000005720 sucrose Substances 0.000 description 1
- 239000011593 sulfur Substances 0.000 description 1
- 229910052717 sulfur Inorganic materials 0.000 description 1
- 238000010189 synthetic method Methods 0.000 description 1
- 229910052714 tellurium Inorganic materials 0.000 description 1
- 229910002058 ternary alloy Inorganic materials 0.000 description 1
- 239000010409 thin film Substances 0.000 description 1
- 150000003568 thioethers Chemical class 0.000 description 1
- XOLBLPGZBRYERU-UHFFFAOYSA-N tin dioxide Chemical compound O=[Sn]=O XOLBLPGZBRYERU-UHFFFAOYSA-N 0.000 description 1
- 229910001887 tin oxide Inorganic materials 0.000 description 1
- HPGGPRDJHPYFRM-UHFFFAOYSA-J tin(iv) chloride Chemical compound Cl[Sn](Cl)(Cl)Cl HPGGPRDJHPYFRM-UHFFFAOYSA-J 0.000 description 1
- YUOWTJMRMWQJDA-UHFFFAOYSA-J tin(iv) fluoride Chemical compound [F-].[F-].[F-].[F-].[Sn+4] YUOWTJMRMWQJDA-UHFFFAOYSA-J 0.000 description 1
- 230000001988 toxicity Effects 0.000 description 1
- 231100000419 toxicity Toxicity 0.000 description 1
- 238000012546 transfer Methods 0.000 description 1
- RIUWBIIVUYSTCN-UHFFFAOYSA-N trilithium borate Chemical class [Li+].[Li+].[Li+].[O-]B([O-])[O-] RIUWBIIVUYSTCN-UHFFFAOYSA-N 0.000 description 1
- NDZWKTKXYOWZML-UHFFFAOYSA-N trilithium;difluoro oxalate;borate Chemical compound [Li+].[Li+].[Li+].[O-]B([O-])[O-].FOC(=O)C(=O)OF NDZWKTKXYOWZML-UHFFFAOYSA-N 0.000 description 1
- 238000009736 wetting Methods 0.000 description 1
- 239000002023 wood Substances 0.000 description 1
- 235000005074 zinc chloride Nutrition 0.000 description 1
- 239000011592 zinc chloride Substances 0.000 description 1
- BHHYHSUAOQUXJK-UHFFFAOYSA-L zinc fluoride Chemical compound F[Zn]F BHHYHSUAOQUXJK-UHFFFAOYSA-L 0.000 description 1
Images
Classifications
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/38—Selection of substances as active materials, active masses, active liquids of elements or alloys
- H01M4/388—Halogens
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/362—Composites
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M10/00—Secondary cells; Manufacture thereof
- H01M10/05—Accumulators with non-aqueous electrolyte
- H01M10/052—Li-accumulators
- H01M10/0525—Rocking-chair batteries, i.e. batteries with lithium insertion or intercalation in both electrodes; Lithium-ion batteries
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/04—Processes of manufacture in general
- H01M4/0402—Methods of deposition of the material
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/13—Electrodes for accumulators with non-aqueous electrolyte, e.g. for lithium-accumulators; Processes of manufacture thereof
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/362—Composites
- H01M4/364—Composites as mixtures
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/362—Composites
- H01M4/366—Composites as layered products
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/38—Selection of substances as active materials, active masses, active liquids of elements or alloys
- H01M4/386—Silicon or alloys based on silicon
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/58—Selection of substances as active materials, active masses, active liquids of inorganic compounds other than oxides or hydroxides, e.g. sulfides, selenides, tellurides, halogenides or LiCoFy; of polyanionic structures, e.g. phosphates, silicates or borates
- H01M4/582—Halogenides
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/62—Selection of inactive substances as ingredients for active masses, e.g. binders, fillers
- H01M4/624—Electric conductive fillers
- H01M4/625—Carbon or graphite
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E60/00—Enabling technologies; Technologies with a potential or indirect contribution to GHG emissions mitigation
- Y02E60/10—Energy storage using batteries
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02P—CLIMATE CHANGE MITIGATION TECHNOLOGIES IN THE PRODUCTION OR PROCESSING OF GOODS
- Y02P70/00—Climate change mitigation technologies in the production process for final industrial or consumer products
- Y02P70/50—Manufacturing or production processes characterised by the final manufactured product
Landscapes
- Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Electrochemistry (AREA)
- General Chemical & Material Sciences (AREA)
- Composite Materials (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Manufacturing & Machinery (AREA)
- Inorganic Chemistry (AREA)
- Battery Electrode And Active Subsutance (AREA)
- Secondary Cells (AREA)
Abstract
Description
本特許出願は、2015年10月13日出願の「特性が変化するナノ複合電池電極粒子(Nanocomposite Battery Electrode Particles with Changing Properties)」と題する米国特許出願第14/882,166号、および2014年10月14日出願の「半径に沿って特性が変化するナノ複合電池電極粒子(Nanocomposite Battery Electrode Particles with Properties Changing Along their Radii)」と題する米国仮特許出願第62/063,493号に基づく優先権を主張するものであり、これらの出願はそれぞれ、参照によって全体が本出願に明示的に組み込まれる。
本開示は概してエネルギー貯蔵装置、より詳細には、粉末ベースの電極などを活用した電池技術に関する。
電気化学エネルギー貯蔵技術は、広範囲に及ぶ重要な用途、いくつかの例を挙げると、エネルギー効率の良い産業用機器、電動型およびハイブリッド電動型の輸送手段(陸上用車両、航空機および船舶を含む)、送電網ならびに家庭用電化製品などの用途に役立つ。リチウムイオン(Liイオン)電池など先進的な金属イオン電池は、一部ではエネルギー密度が比較的高く軽量で長期間にわたり使用できる潜在性があることから、現在、家庭用電化製品および電動型輸送手段の用途で支配的である。しかしながら、様々な種類の電池のさらなる開発および改善が必要とされている。
Claims (18)
- 複合粒子を含む電池電極組成物であって、
各前記複合粒子が、
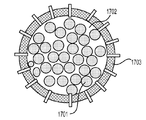
電池作動中にイオンを貯蔵および放出するように設けられ、(i)正極活物質として少なくとも220mAh/gの比容量、または(ii)負極活物質として少なくとも400mAh/gの比容量を示す、高容量活物質と、
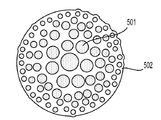
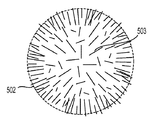
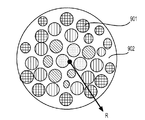
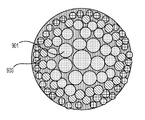
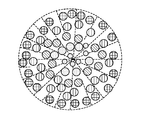
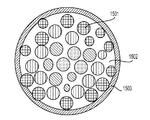
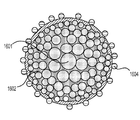
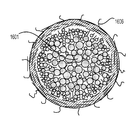
前記活物質が配置される細孔を有する、多孔質の導電性骨格マトリクス材料と、
前記活物質および前記骨格マトリクス材料を少なくとも部分的に包囲するシェルと、
を含み、
前記骨格マトリクス材料が、(i)炭素、(ii)伝導性ポリマー、または(iii)セラミック材料のうちの少なくとも1つを含み、
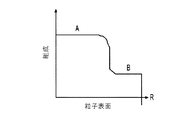
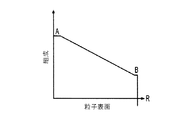
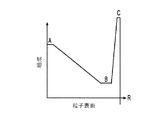
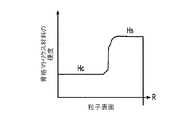
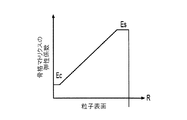
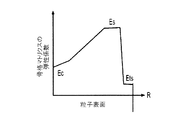
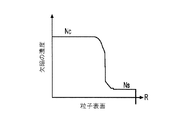
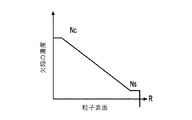
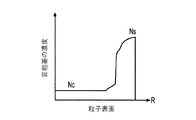
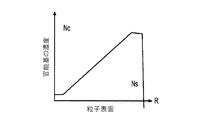
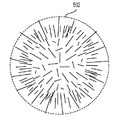
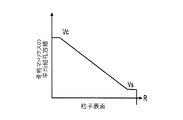
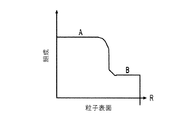
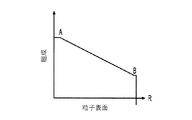
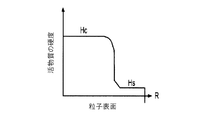
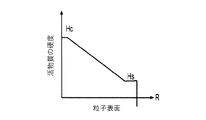
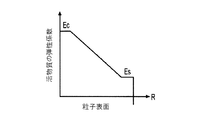
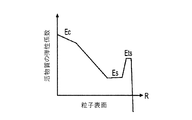
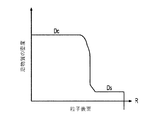
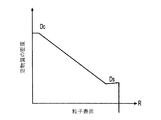
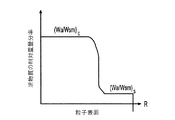
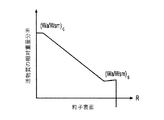
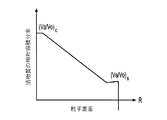
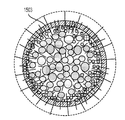
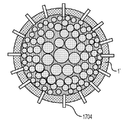
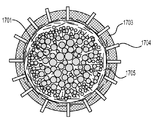
各前記複合粒子が、前記骨格マトリクス材料中において、前記骨格マトリクス材料の中心から周縁にかけて変化している少なくとも1つの平均材料特性を示し、
前記平均材料特性が以下のものを含み、
(i)前記骨格マトリクス材料の材料組成;
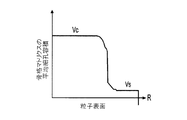
(ii)前記骨格マトリクス材料の、機械的強度、機械的安定性、靱性、硬度を含む機械特性;
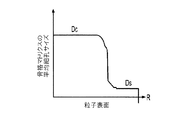
(iii)前記中心から前記周縁に向かって低下する、前記骨格マトリクス材料内の欠陥の濃度または不規則度;
(iv)前記骨格マトリクス材料の平均細孔サイズ;
(v)前記骨格マトリクス材料の細孔配向;
(vi)前記骨格マトリクス材料に対する前記活物質の容積分率;
(vii)前記活物質の材料組成;
(viii)前記複合粒子の密度;
前記(iii)においては、前記欠陥の濃度が高い部分で前記活物質の堆積による核形成が促進される、電池電極組成物。 - 前記活物質が、負極活物質であり、かつシリコンを含む、請求項1に記載の電池電極組成物。
- 前記活物質が、正極活物質であり、かつフッ素を含む、請求項1に記載の電池電極組成物。
- 前記骨格マトリクス材料が炭素を含む、請求項1に記載の電池電極組成物。
- 前記シェルが、前記活物質により貯蔵および放出される前記イオンに対して透過性であり、かつ、前記活物質と電解質溶媒との間にバリアを形成する、請求項1に記載の電池電極組成物。
- 前記機械特性が、前記骨格マトリクス材料の前記周縁と比較して、前記骨格マトリクス材料の前記周縁よりも前記骨格マトリクス材料の前記中心の近くにおいて、より低い弾性係数またはより低い硬度を有する、請求項1に記載の電池電極組成物。
- 前記平均細孔サイズが、前記骨格マトリクス材料の前記周縁と比較して、前記骨格マトリクス材料の前記周縁よりも前記骨格マトリクス材料の前記中心の近くで大きくなる、請求項1に記載の電池電極組成物。
- 前記平均細孔サイズが、前記骨格マトリクス材料の前記中心と前記周縁との間に位置する前記骨格マトリクスの中間領域と比較して、前記骨格マトリクス材料の前記周縁よりも前記中心に近い第1領域および前記骨格マトリクス材料の前記中心よりも前記周縁に近い第2領域で大きくなる、請求項1に記載の電池電極組成物。
- 前記骨格マトリクス材料粒子の前記中心での前記平均細孔サイズが2nmから100nmの範囲である、請求項1に記載の電池電極組成物。
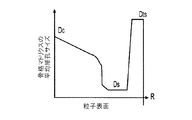
- 前記材料組成が、前記骨格マトリクス材料の前記周縁よりも前記骨格マトリクス材料の前記中心の近くにより多く集中する第1の活物質と、前記骨格マトリクス材料の前記中心よりも前記骨格マトリクス材料の前記周縁の近くにより多く集中する第2の活物質とを含み、
前記第1の活物質が前記第2の活物質より高いエネルギー密度を有する、請求項1に記載の電池電極組成物。 - 前記密度が、前記骨格マトリクス材料の前記周縁と比較して、前記骨格マトリクス材料の前記周縁よりも前記骨格マトリクス材料の前記中心の近くで高い、請求項1に記載の電池電極組成物。
- 前記シェルが、80℃超の温度で導電性またはイオン伝導性が減少する感熱性材料で形成される、請求項1に記載の電池電極組成物。
- 各前記複合粒子が、1nmから500nmの範囲の高低差を有する表面粗度を示す、請求項1に記載の電池電極組成物。
- 前記シェルが、伝導性炭素シェルである、請求項1に記載の電池電極組成物。
- イオン透過性の負極電極および正極電極であり、該電極のうち少なくとも1つが請求項1に記載の電池電極組成物を含む、電極と、
前記負極電極および正極電極をイオン的につなぐ電解質と、
前記負極電極および正極電極を電気的に分離するセパレーターと
を含む金属イオン電池。 - 複合粒子を含む電池電極組成物の製造方法であって、
電池作動中にイオンを貯蔵および放出するための高容量活物質であり、該活物質が(i)正極活物質として少なくとも220mAh/gの比容量、または、(ii)負極活物質として少なくとも400mAh/gの比容量を示す高容量活物質を、用意するステップと、
前記活物質が配置される細孔を有する、多孔質の導電性骨格マトリクス材料を形成するステップと、
前記活物質および前記骨格マトリクス材料を少なくとも部分的に包囲するシェルを形成するステップと、を含み、
各前記複合粒子が、前記骨格マトリクス材料中において、前記骨格マトリクス材料の中心から周縁にかけて変化する少なくとも1つの平均材料特性を伴って形成され、
前記骨格マトリクス材料の中心から周縁にかけて変化する前記平均材料特性が、以下のものを含み、
(i)前記骨格マトリクス材料の材料組成;
(ii)前記骨格マトリクス材料の機械特性;
(iii)前記中心から前記周縁に向かって低下する、前記骨格マトリクス材料内の欠陥の濃度または不規則度;
(iv)前記骨格マトリクス材料の平均細孔サイズ;
(v)前記骨格マトリクス材料の細孔配向;
(vi)前記骨格マトリクス材料に対する前記活物質の容積分率;
(vii)前記活物質の材料組成;
(viii)前記複合粒子の密度、
前記(iii)においては、前記欠陥の濃度が高い部分で前記活物質の堆積による核形成が促進される、方法。 - 前記骨格マトリクス材料を形成するステップが、前記骨格マトリクス材料内に1つまたは複数の官能基を、前記骨格マトリクス材料の前記中心から前記周縁にかけて変化する分布を伴って導入するステップを含む、請求項16に記載の方法。
- 前記骨格マトリクス材料を形成するステップが、前記活物質を前記骨格マトリクス材料に浸透させるステップを含む請求項16に記載の方法。
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201462063493P | 2014-10-14 | 2014-10-14 | |
| US62/063,493 | 2014-10-14 | ||
| US14/882,166 US10340520B2 (en) | 2014-10-14 | 2015-10-13 | Nanocomposite battery electrode particles with changing properties |
| US14/882,166 | 2015-10-13 |
Related Parent Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2017520972A Division JP2017531907A (ja) | 2014-10-14 | 2015-10-14 | 特性が変化するナノ複合電池電極粒子 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2020194792A true JP2020194792A (ja) | 2020-12-03 |
| JP7185668B2 JP7185668B2 (ja) | 2022-12-07 |
Family
ID=55662378
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2017520972A Pending JP2017531907A (ja) | 2014-10-14 | 2015-10-14 | 特性が変化するナノ複合電池電極粒子 |
| JP2020139694A Active JP7185668B2 (ja) | 2014-10-14 | 2020-08-20 | 特性が変化するナノ複合電池電極粒子 |
Family Applications Before (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2017520972A Pending JP2017531907A (ja) | 2014-10-14 | 2015-10-14 | 特性が変化するナノ複合電池電極粒子 |
Country Status (6)
| Country | Link |
|---|---|
| US (4) | US10340520B2 (ja) |
| EP (2) | EP3207580B1 (ja) |
| JP (2) | JP2017531907A (ja) |
| KR (2) | KR102508429B1 (ja) |
| CN (2) | CN107078287A (ja) |
| WO (1) | WO2016061216A1 (ja) |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2023004060A3 (en) * | 2021-07-23 | 2023-04-13 | Lyten, Inc. | Lithium-sulfur battery cathode formed from multiple carbonaceous regions |
| US11735745B2 (en) | 2021-06-16 | 2023-08-22 | Lyten, Inc. | Lithium-air battery |
| US11735740B2 (en) | 2019-10-25 | 2023-08-22 | Lyten, Inc. | Protective carbon layer for lithium (Li) metal anodes |
| US11901580B2 (en) | 2020-01-10 | 2024-02-13 | Lyten, Inc. | Selectively activated metal-air battery |
| US12003003B2 (en) | 2021-03-31 | 2024-06-04 | Lyten, Inc. | Metal-air battery including electrolyte beads |
Families Citing this family (81)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| AU2007319213B2 (en) | 2006-11-15 | 2014-06-12 | Basf Se | Electric double layer capacitance device |
| CN105226284B (zh) | 2009-07-01 | 2017-11-28 | 巴斯夫欧洲公司 | 超纯合成碳材料 |
| US11710854B2 (en) * | 2020-10-30 | 2023-07-25 | Enevate Corporation | Functional epoxides in catalyst-based electrolyte compositions for Li-ion batteries |
| US20120262127A1 (en) | 2011-04-15 | 2012-10-18 | Energ2 Technologies, Inc. | Flow ultracapacitor |
| US10522836B2 (en) | 2011-06-03 | 2019-12-31 | Basf Se | Carbon-lead blends for use in hybrid energy storage devices |
| US9409777B2 (en) | 2012-02-09 | 2016-08-09 | Basf Se | Preparation of polymeric resins and carbon materials |
| US20140272592A1 (en) | 2013-03-14 | 2014-09-18 | Energ2 Technologies, Inc. | Composite carbon materials comprising lithium alloying electrochemical modifiers |
| US10195583B2 (en) | 2013-11-05 | 2019-02-05 | Group 14 Technologies, Inc. | Carbon-based compositions with highly efficient volumetric gas sorption |
| CN107074994B (zh) | 2014-03-14 | 2021-12-14 | 14集团技术公司 | 无溶剂进行溶胶-凝胶聚合并由其产生可调节的碳结构的新方法 |
| US10549650B2 (en) * | 2014-04-08 | 2020-02-04 | StoreDot Ltd. | Internally adjustable modular single battery systems for power systems |
| US10110036B2 (en) * | 2016-12-15 | 2018-10-23 | StoreDot Ltd. | Supercapacitor-emulating fast-charging batteries and devices |
| US11128152B2 (en) | 2014-04-08 | 2021-09-21 | StoreDot Ltd. | Systems and methods for adaptive fast-charging for mobile devices and devices having sporadic power-source connection |
| US10293704B2 (en) | 2014-04-08 | 2019-05-21 | StoreDot Ltd. | Electric vehicles with adaptive fast-charging, utilizing supercapacitor-emulating batteries |
| US10199646B2 (en) | 2014-07-30 | 2019-02-05 | StoreDot Ltd. | Anodes for lithium-ion devices |
| US10326137B2 (en) * | 2014-09-30 | 2019-06-18 | Nec Corporation | Negative electrode active material for a lithium ion secondary battery and production method for same, and negative electrode and lithium ion secondary battery |
| JP6868335B2 (ja) * | 2015-01-12 | 2021-05-12 | アイメック・ヴェーゼットウェーImec Vzw | 固体バッテリおよび製造方法 |
| WO2017011594A1 (en) | 2015-07-13 | 2017-01-19 | Sila Nanotechnologies Inc. | Stable lithium fluoride-based cathodes for metal and metal-ion batteries |
| WO2017030995A1 (en) | 2015-08-14 | 2017-02-23 | Energ2 Technologies, Inc. | Nano-featured porous silicon materials |
| KR102637617B1 (ko) | 2015-08-28 | 2024-02-19 | 그룹14 테크놀로지스, 인코포레이티드 | 극도로 내구성이 우수한 리튬 인터칼레이션을 나타내는 신규 물질 및 그의 제조 방법 |
| DE102015218189A1 (de) * | 2015-09-22 | 2017-03-23 | Bayerische Motoren Werke Aktiengesellschaft | Lithium-Ionen-Zelle |
| WO2017062521A1 (en) | 2015-10-05 | 2017-04-13 | Sila Nanotechnologies Inc. | Protection of battery electrodes against side reactions |
| EP3420607A1 (en) | 2016-02-23 | 2019-01-02 | Maxwell Technologies, Inc. | Elemental metal and carbon mixtures for energy storage devices |
| US10367191B2 (en) | 2016-04-07 | 2019-07-30 | StoreDot Ltd. | Tin silicon anode active material |
| US10367192B2 (en) | 2016-04-07 | 2019-07-30 | StoreDot Ltd. | Aluminum anode active material |
| EP3440726A4 (en) | 2016-04-07 | 2019-09-11 | StoreDot Ltd. | LITHIUM-ION CELLS AND ANODES THEREFOR |
| US10916811B2 (en) | 2016-04-07 | 2021-02-09 | StoreDot Ltd. | Semi-solid electrolytes with flexible particle coatings |
| US10818919B2 (en) | 2016-04-07 | 2020-10-27 | StoreDot Ltd. | Polymer coatings and anode material pre-lithiation |
| US11205796B2 (en) | 2016-04-07 | 2021-12-21 | StoreDot Ltd. | Electrolyte additives in lithium-ion batteries |
| US10096859B2 (en) | 2016-04-07 | 2018-10-09 | StoreDot Ltd. | Electrolytes with ionic liquid additives for lithium ion batteries |
| US10355271B2 (en) | 2016-04-07 | 2019-07-16 | StoreDot Ltd. | Lithium borates and phosphates coatings |
| US10454101B2 (en) * | 2017-01-25 | 2019-10-22 | StoreDot Ltd. | Composite anode material made of core-shell particles |
| US10680289B2 (en) | 2016-04-07 | 2020-06-09 | StoreDot Ltd. | Buffering zone for preventing lithium metallization on the anode of lithium ion batteries |
| WO2018027187A1 (en) * | 2016-08-05 | 2018-02-08 | Amtek Research International Llc | Solidified, conformable porous composites and related devices, methods, and uses |
| DE102016217017A1 (de) | 2016-09-07 | 2018-03-08 | Bayerische Motoren Werke Aktiengesellschaft | Konversionsmaterial |
| US10707531B1 (en) | 2016-09-27 | 2020-07-07 | New Dominion Enterprises Inc. | All-inorganic solvents for electrolytes |
| DE102016221782A1 (de) | 2016-11-07 | 2018-05-09 | Wacker Chemie Ag | Kohlenstoff-beschichtete Siliciumpartikel für Lithiumionen-Batterien |
| EP4243122A3 (en) | 2016-12-02 | 2023-11-15 | Fastcap Systems Corporation | Composite electrode |
| US11177500B2 (en) | 2017-01-17 | 2021-11-16 | Sila Nanotechnologies, Inc. | Electrolytes for improved performance of cells with high-capacity anodes based on micron-scale moderate volume-changing particles |
| EP4303964A3 (en) | 2017-02-21 | 2024-03-20 | Tesla, Inc. | Prelithiated energy storage device |
| CN110582823A (zh) | 2017-03-09 | 2019-12-17 | 14集团技术公司 | 含硅前体在多孔支架材料上的分解 |
| JP6946902B2 (ja) * | 2017-09-27 | 2021-10-13 | トヨタ自動車株式会社 | 金属負極二次電池の製造方法 |
| US10756334B2 (en) * | 2017-12-22 | 2020-08-25 | Lyten, Inc. | Structured composite materials |
| US11374227B2 (en) | 2018-02-02 | 2022-06-28 | Amtek Research International Llc | Dry process electrically conductive composite formation |
| US11515540B2 (en) * | 2018-04-09 | 2022-11-29 | Global Graphene Group, Inc. | Alkali metal-selenium secondary battery containing a graphene foam-protected selenium cathode |
| JP6947119B2 (ja) * | 2018-05-14 | 2021-10-13 | トヨタ自動車株式会社 | 正極活物質およびフッ化物イオン電池 |
| US11380896B2 (en) * | 2018-09-14 | 2022-07-05 | Sila Nanotechnologies, Inc. | Battery electrode composition comprising biomass-derived carbon |
| US11557784B2 (en) | 2018-11-06 | 2023-01-17 | Utility Global, Inc. | Method of making a fuel cell and treating a component thereof |
| US11611097B2 (en) | 2018-11-06 | 2023-03-21 | Utility Global, Inc. | Method of making an electrochemical reactor via sintering inorganic dry particles |
| US11761100B2 (en) | 2018-11-06 | 2023-09-19 | Utility Global, Inc. | Electrochemical device and method of making |
| US11539053B2 (en) | 2018-11-12 | 2022-12-27 | Utility Global, Inc. | Method of making copper electrode |
| US11603324B2 (en) | 2018-11-06 | 2023-03-14 | Utility Global, Inc. | Channeled electrodes and method of making |
| GB201818232D0 (en) | 2018-11-08 | 2018-12-26 | Nexeon Ltd | Electroactive materials for metal-ion batteries |
| GB201818235D0 (en) | 2018-11-08 | 2018-12-26 | Nexeon Ltd | Electroactive materials for metal-ion batteries |
| GB2580033B (en) | 2018-12-19 | 2021-03-10 | Nexeon Ltd | Electroactive materials for metal-Ion batteries |
| WO2020102140A1 (en) * | 2018-11-12 | 2020-05-22 | Utility Global, Inc. | Manufacturing method with particle size control |
| US11905593B2 (en) | 2018-12-21 | 2024-02-20 | Nexeon Limited | Process for preparing electroactive materials for metal-ion batteries |
| US10508335B1 (en) | 2019-02-13 | 2019-12-17 | Nexeon Limited | Process for preparing electroactive materials for metal-ion batteries |
| US11552328B2 (en) * | 2019-01-18 | 2023-01-10 | Sila Nanotechnologies, Inc. | Lithium battery cell including cathode having metal fluoride core-shell particle |
| US10608463B1 (en) | 2019-01-23 | 2020-03-31 | StoreDot Ltd. | Direct charging of battery cell stacks |
| CA3131698A1 (en) | 2019-02-27 | 2020-09-03 | Aspen Aerogels, Inc. | Carbon aerogel-based electrode materials and methods of manufacture thereof |
| CA3134169A1 (en) | 2019-03-22 | 2020-10-01 | Aspen Aerogels, Inc. | Carbon aerogel-based cathodes for lithium-air batteries |
| US11374213B2 (en) | 2019-03-22 | 2022-06-28 | Aspen Aerogels, Inc. | Carbon aerogel-based cathodes for lithium-sulfur batteries |
| CN111799437B (zh) * | 2019-04-08 | 2021-07-09 | 宁德时代新能源科技股份有限公司 | 正极极片及钠离子电池 |
| US11831012B2 (en) | 2019-04-25 | 2023-11-28 | StoreDot Ltd. | Passivated silicon-based anode material particles |
| US11961998B2 (en) * | 2019-05-06 | 2024-04-16 | Honeycomb Battery Company | Method of producing protected anode active material particles for rechargeable lithium batteries |
| CN110348054B (zh) * | 2019-06-11 | 2020-11-06 | 河海大学 | 一种含硬核-软壳结构体的颗粒增强材料导电率计算方法 |
| US11557765B2 (en) | 2019-07-05 | 2023-01-17 | Fastcap Systems Corporation | Electrodes for energy storage devices |
| CN112331906B (zh) * | 2019-08-05 | 2022-08-05 | 辉能科技股份有限公司 | 活性材料球极层结构 |
| TWI756556B (zh) * | 2019-08-05 | 2022-03-01 | 輝能科技股份有限公司 | 活性材料球極層結構 |
| CN112331820B (zh) * | 2019-08-05 | 2022-03-04 | 辉能科技股份有限公司 | 活性材料球复合层 |
| TWI735017B (zh) * | 2019-08-05 | 2021-08-01 | 輝能科技股份有限公司 | 活性材料球複合層 |
| US11367863B2 (en) | 2019-11-15 | 2022-06-21 | International Business Machines Corporation | Porous silicon anode for rechargeable metal halide battery |
| US11677282B2 (en) * | 2020-02-27 | 2023-06-13 | Accelerated Systems Inc. | Composite material for thermal management in an electric motor |
| US10964940B1 (en) | 2020-09-17 | 2021-03-30 | Nexeon Limited | Electroactive materials for metal-ion batteries |
| CN115136347A (zh) | 2020-03-18 | 2022-09-30 | 瓦克化学股份公司 | 碳涂覆的硅颗粒的制造方法 |
| WO2022002404A1 (de) | 2020-07-02 | 2022-01-06 | Wacker Chemie Ag | Verfahren zur herstellung von kohlenstoff-beschichteten siliciumpartikel für lithiumionen-batterien |
| US11639292B2 (en) | 2020-08-18 | 2023-05-02 | Group14 Technologies, Inc. | Particulate composite materials |
| US11174167B1 (en) | 2020-08-18 | 2021-11-16 | Group14 Technologies, Inc. | Silicon carbon composites comprising ultra low Z |
| US11335903B2 (en) | 2020-08-18 | 2022-05-17 | Group14 Technologies, Inc. | Highly efficient manufacturing of silicon-carbon composites materials comprising ultra low z |
| WO2022074031A1 (en) | 2020-10-08 | 2022-04-14 | Umicore | A powder of carbonaceous matrix particles and a composite powder, for use in the negative electrode of a battery, comprising such a powder |
| WO2023184125A1 (zh) * | 2022-03-29 | 2023-10-05 | 宁德新能源科技有限公司 | 电化学装置和电子设备 |
Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2010129545A (ja) * | 2008-12-01 | 2010-06-10 | Samsung Sdi Co Ltd | 負極活物質、負極、及びリチウム二次電池 |
| JP2012124115A (ja) * | 2010-12-10 | 2012-06-28 | Hitachi Chem Co Ltd | リチウムイオン二次電池用負極材及びその製造方法、リチウムイオン二次電池用負極、並びにリチウムイオン二次電池 |
| JP2012124121A (ja) * | 2010-12-10 | 2012-06-28 | Hitachi Chem Co Ltd | リチウム二次電池用負極材の製造方法 |
| JP2012124116A (ja) * | 2010-12-10 | 2012-06-28 | Hitachi Chem Co Ltd | リチウムイオン二次電池用負極材及びその製造方法、リチウムイオン二次電池用負極、並びにリチウムイオン二次電池 |
| WO2013117192A1 (de) * | 2012-02-08 | 2013-08-15 | Studiengesellschaft Kohle Mbh | Verwendung von mesoporösen graphitischen teilchen für elektrochemische anwendungen |
| WO2013192205A1 (en) * | 2012-06-18 | 2013-12-27 | Sila Nanotechnologies Inc. | Multi-shell structures for battery active materials with expansion properties |
| WO2014031929A1 (en) * | 2012-08-24 | 2014-02-27 | Sila Nanotechnologies Inc. | Scaffolding matrix with internal nanoparticles |
Family Cites Families (29)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5780164A (en) | 1994-12-12 | 1998-07-14 | The Dow Chemical Company | Computer disk substrate, the process for making same, and the material made therefrom |
| JP3352316B2 (ja) | 1995-03-17 | 2002-12-03 | キヤノン株式会社 | リチウム二次電池、リチウム二次電池用電極およびそれ等の作製方法 |
| US5919587A (en) | 1996-05-22 | 1999-07-06 | Moltech Corporation | Composite cathodes, electrochemical cells comprising novel composite cathodes, and processes for fabricating same |
| KR101356250B1 (ko) | 2000-10-20 | 2014-02-06 | 매사츄세츠 인스티튜트 오브 테크놀러지 | 2극 장치 |
| TWI246212B (en) * | 2003-06-25 | 2005-12-21 | Lg Chemical Ltd | Anode material for lithium secondary cell with high capacity |
| US20100055459A1 (en) | 2006-08-30 | 2010-03-04 | Liquidia Technologies, Inc. | Nanoparticles Having Functional Additives for Self and Directed Assembly and Methods of Fabricating Same |
| JP5376894B2 (ja) * | 2008-10-20 | 2013-12-25 | 古河電池株式会社 | オリビン構造を有する多元系リン酸型リチウム化合物粒子、その製造方法及びこれを正極材料に用いたリチウム二次電池 |
| US8450012B2 (en) | 2009-05-27 | 2013-05-28 | Amprius, Inc. | Interconnected hollow nanostructures containing high capacity active materials for use in rechargeable batteries |
| WO2011041468A1 (en) | 2009-09-29 | 2011-04-07 | Georgia Tech Research Corporation | Electrodes, lithium-ion batteries, and methods of making and using same |
| US8236452B2 (en) | 2009-11-02 | 2012-08-07 | Nanotek Instruments, Inc. | Nano-structured anode compositions for lithium metal and lithium metal-air secondary batteries |
| WO2011060024A2 (en) | 2009-11-11 | 2011-05-19 | Amprius, Inc. | Open structures in substrates for electrodes |
| CN102122708A (zh) * | 2010-01-08 | 2011-07-13 | 中国科学院物理研究所 | 用于锂离子二次电池的负极材料、含该负极材料的负极及其制备方法以及含该负极的电池 |
| WO2011085327A2 (en) | 2010-01-11 | 2011-07-14 | Amprius Inc. | Variable capacity cell assembly |
| WO2012054766A2 (en) | 2010-10-22 | 2012-04-26 | Amprius Inc. | Composite structures containing high capacity porous active materials constrained in shells |
| US9786947B2 (en) | 2011-02-07 | 2017-10-10 | Sila Nanotechnologies Inc. | Stabilization of Li-ion battery anodes |
| US10256458B2 (en) | 2011-04-01 | 2019-04-09 | Georgia Tech Research Corporation | Curved two-dimensional nanocomposites for battery electrodes |
| US9394165B2 (en) | 2011-06-15 | 2016-07-19 | Georgia Tech Research Corporation | Carbon nanotube array bonding |
| US8932764B2 (en) | 2012-02-28 | 2015-01-13 | Sila Nanotechnologies, Inc. | Core-shell composites for sulfur-based cathodes in metal-ion batteries |
| WO2013187969A2 (en) | 2012-06-07 | 2013-12-19 | Robert Bosch Gmbh | Metal/oxygen battery with internal oxygen reservoir |
| JP5941437B2 (ja) * | 2012-06-29 | 2016-06-29 | Jfeケミカル株式会社 | リチウムイオン二次電池負極用複合粒子及びその製造方法、リチウムイオン二次電池用負極並びにリチウムイオン二次電池 |
| JP5993337B2 (ja) * | 2012-07-03 | 2016-09-14 | Jfeケミカル株式会社 | リチウムイオン二次電池用負極材料およびその製造方法ならびにこれを用いたリチウムイオン二次電池用負極ならびにリチウムイオン二次電池 |
| US10263279B2 (en) * | 2012-12-14 | 2019-04-16 | Sila Nanotechnologies Inc. | Electrodes for energy storage devices with solid electrolytes and methods of fabricating the same |
| WO2014153536A1 (en) | 2013-03-21 | 2014-09-25 | Sila Nanotechnologies Inc. | Electrochemical energy storage devices and components |
| US11721831B2 (en) | 2013-08-30 | 2023-08-08 | Sila Nanotechnologies, Inc. | Electrolyte or electrode additives for increasing metal content in metal-ion batteries |
| US10224537B2 (en) | 2013-11-29 | 2019-03-05 | Sila Nanotechnologies, Inc. | Fluorides in nanoporous, electrically-conductive scaffolding matrix for metal and metal-ion batteries |
| CN103715402B (zh) * | 2013-12-18 | 2015-08-05 | 湘潭大学 | 一种基于火山岩的锂硫电池正极材料及其制备和应用方法 |
| JP6156190B2 (ja) * | 2014-02-27 | 2017-07-05 | ソニー株式会社 | 負極活物質、電池、電池パック、電子機器、電動車両、蓄電装置および電力システム |
| US20150318530A1 (en) | 2014-05-01 | 2015-11-05 | Sila Nanotechnologies, Inc. | Aqueous electrochemical energy storage devices and components |
| KR102331721B1 (ko) * | 2014-12-31 | 2021-11-26 | 삼성전자 주식회사 | 복합 음극 활물질, 그 제조방법, 이를 포함하는 음극 및 리튬이차전지 |
-
2015
- 2015-10-13 US US14/882,166 patent/US10340520B2/en active Active
- 2015-10-14 KR KR1020177012942A patent/KR102508429B1/ko active IP Right Grant
- 2015-10-14 EP EP15850603.0A patent/EP3207580B1/en active Active
- 2015-10-14 KR KR1020237007704A patent/KR102591003B1/ko active IP Right Grant
- 2015-10-14 CN CN201580056096.6A patent/CN107078287A/zh active Pending
- 2015-10-14 JP JP2017520972A patent/JP2017531907A/ja active Pending
- 2015-10-14 EP EP21154047.1A patent/EP3866229B1/en active Active
- 2015-10-14 CN CN202110391187.2A patent/CN113381002A/zh active Pending
- 2015-10-14 WO PCT/US2015/055518 patent/WO2016061216A1/en active Application Filing
-
2019
- 2019-06-28 US US16/457,701 patent/US11233235B2/en active Active
-
2020
- 2020-08-20 JP JP2020139694A patent/JP7185668B2/ja active Active
-
2021
- 2021-11-15 US US17/454,962 patent/US11575129B2/en active Active
-
2023
- 2023-01-13 US US18/154,368 patent/US11855280B2/en active Active
Patent Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2010129545A (ja) * | 2008-12-01 | 2010-06-10 | Samsung Sdi Co Ltd | 負極活物質、負極、及びリチウム二次電池 |
| JP2012124115A (ja) * | 2010-12-10 | 2012-06-28 | Hitachi Chem Co Ltd | リチウムイオン二次電池用負極材及びその製造方法、リチウムイオン二次電池用負極、並びにリチウムイオン二次電池 |
| JP2012124121A (ja) * | 2010-12-10 | 2012-06-28 | Hitachi Chem Co Ltd | リチウム二次電池用負極材の製造方法 |
| JP2012124116A (ja) * | 2010-12-10 | 2012-06-28 | Hitachi Chem Co Ltd | リチウムイオン二次電池用負極材及びその製造方法、リチウムイオン二次電池用負極、並びにリチウムイオン二次電池 |
| WO2013117192A1 (de) * | 2012-02-08 | 2013-08-15 | Studiengesellschaft Kohle Mbh | Verwendung von mesoporösen graphitischen teilchen für elektrochemische anwendungen |
| WO2013192205A1 (en) * | 2012-06-18 | 2013-12-27 | Sila Nanotechnologies Inc. | Multi-shell structures for battery active materials with expansion properties |
| WO2014031929A1 (en) * | 2012-08-24 | 2014-02-27 | Sila Nanotechnologies Inc. | Scaffolding matrix with internal nanoparticles |
Non-Patent Citations (1)
| Title |
|---|
| MIYUKI, TAKUHIRO ET AL., ELECTROCHEMISTRY, vol. 80(6), JPN6022026250, 2012, pages 401 - 404, ISSN: 0004812484 * |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US11735740B2 (en) | 2019-10-25 | 2023-08-22 | Lyten, Inc. | Protective carbon layer for lithium (Li) metal anodes |
| US11901580B2 (en) | 2020-01-10 | 2024-02-13 | Lyten, Inc. | Selectively activated metal-air battery |
| US12003003B2 (en) | 2021-03-31 | 2024-06-04 | Lyten, Inc. | Metal-air battery including electrolyte beads |
| US11735745B2 (en) | 2021-06-16 | 2023-08-22 | Lyten, Inc. | Lithium-air battery |
| WO2023004060A3 (en) * | 2021-07-23 | 2023-04-13 | Lyten, Inc. | Lithium-sulfur battery cathode formed from multiple carbonaceous regions |
Also Published As
| Publication number | Publication date |
|---|---|
| EP3866229B1 (en) | 2023-11-29 |
| KR102591003B1 (ko) | 2023-10-17 |
| EP3207580B1 (en) | 2021-03-10 |
| JP7185668B2 (ja) | 2022-12-07 |
| US11855280B2 (en) | 2023-12-26 |
| EP3866229A1 (en) | 2021-08-18 |
| KR20170067878A (ko) | 2017-06-16 |
| US20230163286A1 (en) | 2023-05-25 |
| CN113381002A (zh) | 2021-09-10 |
| US11575129B2 (en) | 2023-02-07 |
| CN107078287A (zh) | 2017-08-18 |
| JP2017531907A (ja) | 2017-10-26 |
| US20160104882A1 (en) | 2016-04-14 |
| KR102508429B1 (ko) | 2023-03-08 |
| EP3207580A1 (en) | 2017-08-23 |
| US11233235B2 (en) | 2022-01-25 |
| US20190341607A1 (en) | 2019-11-07 |
| KR20230039754A (ko) | 2023-03-21 |
| US20220077462A1 (en) | 2022-03-10 |
| WO2016061216A1 (en) | 2016-04-21 |
| US10340520B2 (en) | 2019-07-02 |
| EP3207580A4 (en) | 2018-04-18 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP7185668B2 (ja) | 特性が変化するナノ複合電池電極粒子 | |
| JP6770565B2 (ja) | 内部ナノ粒子を有する骨格マトリックス | |
| US20220181597A1 (en) | Curved two-dimensional nanocomposites for battery electrodes | |
| US11522176B2 (en) | Electrodes, lithium-ion batteries, and methods of making and using same | |
| JP6715877B2 (ja) | 膨張特性を有する電池活物質用のマルチシェル構造体 | |
| JP2021082599A (ja) | 金属および金属イオン電池用の安定なフッ化リチウム系カソード | |
| CN108370060A (zh) | 用于安全金属和金属离子电池的固态电解质 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20200918 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20211019 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20220119 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20220316 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20220628 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20220823 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20221108 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20221125 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 7185668 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 |