EP1533392B1 - Stahlprodukt zum schweissen mit hohem wärmeeintrag und verfahren zu seiner herstellung - Google Patents
Stahlprodukt zum schweissen mit hohem wärmeeintrag und verfahren zu seiner herstellung Download PDFInfo
- Publication number
- EP1533392B1 EP1533392B1 EP02763002.9A EP02763002A EP1533392B1 EP 1533392 B1 EP1533392 B1 EP 1533392B1 EP 02763002 A EP02763002 A EP 02763002A EP 1533392 B1 EP1533392 B1 EP 1533392B1
- Authority
- EP
- European Patent Office
- Prior art keywords
- mass
- steel
- toughness
- weld
- content
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Lifetime
Links
- 229910000831 Steel Inorganic materials 0.000 title claims description 60
- 239000010959 steel Substances 0.000 title claims description 60
- 238000003466 welding Methods 0.000 title claims description 35
- 238000004519 manufacturing process Methods 0.000 title description 8
- 239000011575 calcium Substances 0.000 claims description 33
- 239000010936 titanium Substances 0.000 claims description 16
- 239000011572 manganese Substances 0.000 claims description 13
- PXHVJJICTQNCMI-UHFFFAOYSA-N nickel Substances [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 claims description 11
- 229910052791 calcium Inorganic materials 0.000 claims description 10
- 239000011651 chromium Substances 0.000 claims description 9
- 239000010949 copper Substances 0.000 claims description 9
- 239000000203 mixture Substances 0.000 claims description 9
- 229910052717 sulfur Inorganic materials 0.000 claims description 9
- 239000010955 niobium Substances 0.000 claims description 8
- 229910052760 oxygen Inorganic materials 0.000 claims description 8
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 claims description 6
- 229910052748 manganese Inorganic materials 0.000 claims description 5
- 229910052757 nitrogen Inorganic materials 0.000 claims description 5
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 claims description 4
- 229910052796 boron Inorganic materials 0.000 claims description 4
- 229910052799 carbon Inorganic materials 0.000 claims description 4
- 229910052804 chromium Inorganic materials 0.000 claims description 4
- 229910052802 copper Inorganic materials 0.000 claims description 4
- XEEYBQQBJWHFJM-UHFFFAOYSA-N iron Substances [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 claims description 4
- 229910052750 molybdenum Inorganic materials 0.000 claims description 4
- 239000001301 oxygen Substances 0.000 claims description 4
- 229910052710 silicon Inorganic materials 0.000 claims description 4
- 229910052719 titanium Inorganic materials 0.000 claims description 4
- 229910052720 vanadium Inorganic materials 0.000 claims description 4
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical compound [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 claims description 3
- 229910052782 aluminium Inorganic materials 0.000 claims description 3
- 229910052759 nickel Inorganic materials 0.000 claims description 3
- 229910052758 niobium Inorganic materials 0.000 claims description 3
- 229910052698 phosphorus Inorganic materials 0.000 claims description 3
- ZOXJGFHDIHLPTG-UHFFFAOYSA-N Boron Chemical compound [B] ZOXJGFHDIHLPTG-UHFFFAOYSA-N 0.000 claims description 2
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 claims description 2
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 claims description 2
- VYZAMTAEIAYCRO-UHFFFAOYSA-N Chromium Chemical compound [Cr] VYZAMTAEIAYCRO-UHFFFAOYSA-N 0.000 claims description 2
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 claims description 2
- PWHULOQIROXLJO-UHFFFAOYSA-N Manganese Chemical compound [Mn] PWHULOQIROXLJO-UHFFFAOYSA-N 0.000 claims description 2
- ZOKXTWBITQBERF-UHFFFAOYSA-N Molybdenum Chemical compound [Mo] ZOKXTWBITQBERF-UHFFFAOYSA-N 0.000 claims description 2
- OAICVXFJPJFONN-UHFFFAOYSA-N Phosphorus Chemical compound [P] OAICVXFJPJFONN-UHFFFAOYSA-N 0.000 claims description 2
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 claims description 2
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 claims description 2
- 239000012535 impurity Substances 0.000 claims description 2
- 239000011733 molybdenum Substances 0.000 claims description 2
- GUCVJGMIXFAOAE-UHFFFAOYSA-N niobium atom Chemical compound [Nb] GUCVJGMIXFAOAE-UHFFFAOYSA-N 0.000 claims description 2
- 239000011574 phosphorus Substances 0.000 claims description 2
- 239000010703 silicon Substances 0.000 claims description 2
- 239000011593 sulfur Substances 0.000 claims description 2
- 239000004411 aluminium Substances 0.000 claims 1
- 229910052742 iron Inorganic materials 0.000 claims 1
- LEONUFNNVUYDNQ-UHFFFAOYSA-N vanadium atom Chemical compound [V] LEONUFNNVUYDNQ-UHFFFAOYSA-N 0.000 claims 1
- 238000000034 method Methods 0.000 description 27
- 230000000694 effects Effects 0.000 description 22
- 229910052751 metal Inorganic materials 0.000 description 21
- 239000002184 metal Substances 0.000 description 21
- ATJFFYVFTNAWJD-UHFFFAOYSA-N Tin Chemical compound [Sn] ATJFFYVFTNAWJD-UHFFFAOYSA-N 0.000 description 16
- 238000001816 cooling Methods 0.000 description 11
- 229910000859 α-Fe Inorganic materials 0.000 description 10
- 229910001566 austenite Inorganic materials 0.000 description 7
- 230000000052 comparative effect Effects 0.000 description 7
- 230000009466 transformation Effects 0.000 description 7
- 239000006185 dispersion Substances 0.000 description 6
- -1 REM sulfides Chemical class 0.000 description 4
- 230000009471 action Effects 0.000 description 4
- 230000001276 controlling effect Effects 0.000 description 4
- 238000010438 heat treatment Methods 0.000 description 4
- 230000002411 adverse Effects 0.000 description 3
- 230000006866 deterioration Effects 0.000 description 3
- 238000005098 hot rolling Methods 0.000 description 3
- 230000006911 nucleation Effects 0.000 description 3
- 238000010899 nucleation Methods 0.000 description 3
- 230000008569 process Effects 0.000 description 3
- 238000009628 steelmaking Methods 0.000 description 3
- 125000000101 thioether group Chemical group 0.000 description 3
- UCKMPCXJQFINFW-UHFFFAOYSA-N Sulphide Chemical compound [S-2] UCKMPCXJQFINFW-UHFFFAOYSA-N 0.000 description 2
- 238000010276 construction Methods 0.000 description 2
- 238000005336 cracking Methods 0.000 description 2
- 230000003247 decreasing effect Effects 0.000 description 2
- 230000001747 exhibiting effect Effects 0.000 description 2
- 230000006872 improvement Effects 0.000 description 2
- 229910001562 pearlite Inorganic materials 0.000 description 2
- 230000009467 reduction Effects 0.000 description 2
- 238000011160 research Methods 0.000 description 2
- 229920006395 saturated elastomer Polymers 0.000 description 2
- 239000006104 solid solution Substances 0.000 description 2
- 238000007711 solidification Methods 0.000 description 2
- 230000008023 solidification Effects 0.000 description 2
- 230000007704 transition Effects 0.000 description 2
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 1
- 229910000746 Structural steel Inorganic materials 0.000 description 1
- 229910001563 bainite Inorganic materials 0.000 description 1
- 230000015572 biosynthetic process Effects 0.000 description 1
- 230000003749 cleanliness Effects 0.000 description 1
- 150000001875 compounds Chemical class 0.000 description 1
- 239000000470 constituent Substances 0.000 description 1
- 238000009749 continuous casting Methods 0.000 description 1
- 238000007796 conventional method Methods 0.000 description 1
- 230000007797 corrosion Effects 0.000 description 1
- 238000005260 corrosion Methods 0.000 description 1
- 238000002425 crystallisation Methods 0.000 description 1
- 230000008025 crystallization Effects 0.000 description 1
- 238000007872 degassing Methods 0.000 description 1
- 230000002542 deteriorative effect Effects 0.000 description 1
- 238000004090 dissolution Methods 0.000 description 1
- 229910052739 hydrogen Inorganic materials 0.000 description 1
- 239000001257 hydrogen Substances 0.000 description 1
- 238000009863 impact test Methods 0.000 description 1
- 230000008018 melting Effects 0.000 description 1
- 238000002844 melting Methods 0.000 description 1
- 150000002739 metals Chemical class 0.000 description 1
- 238000001000 micrograph Methods 0.000 description 1
- 238000001556 precipitation Methods 0.000 description 1
- 238000010791 quenching Methods 0.000 description 1
- 230000000171 quenching effect Effects 0.000 description 1
- 230000001105 regulatory effect Effects 0.000 description 1
- 238000003303 reheating Methods 0.000 description 1
- 238000005096 rolling process Methods 0.000 description 1
- 230000001629 suppression Effects 0.000 description 1
- 238000005496 tempering Methods 0.000 description 1
- GPPXJZIENCGNKB-UHFFFAOYSA-N vanadium Chemical compound [V]#[V] GPPXJZIENCGNKB-UHFFFAOYSA-N 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/002—Ferrous alloys, e.g. steel alloys containing In, Mg, or other elements not provided for in one single group C22C38/001 - C22C38/60
-
- C—CHEMISTRY; METALLURGY
- C21—METALLURGY OF IRON
- C21D—MODIFYING THE PHYSICAL STRUCTURE OF FERROUS METALS; GENERAL DEVICES FOR HEAT TREATMENT OF FERROUS OR NON-FERROUS METALS OR ALLOYS; MAKING METAL MALLEABLE, e.g. BY DECARBURISATION OR TEMPERING
- C21D8/00—Modifying the physical properties by deformation combined with, or followed by, heat treatment
- C21D8/02—Modifying the physical properties by deformation combined with, or followed by, heat treatment during manufacturing of plates or strips
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/02—Ferrous alloys, e.g. steel alloys containing silicon
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/04—Ferrous alloys, e.g. steel alloys containing manganese
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/06—Ferrous alloys, e.g. steel alloys containing aluminium
Definitions
- the present invention relates to steels used for various structures such as those in the fields of shipbuilding, construction, and civil engineering. More specifically, the present invention relates to steels suitably usable for high heat input welding with a heat input exceeding 400 kJ/cm.
- steels used in the fields of shipbuilding, construction, and civil engineering are processed by welding to fabricate structures having desired shapes.
- the individual steels are required not only to exhibit a high parent-metal toughness as a matter of course but also to exhibit high weld-zone toughness.
- high-efficiency high heat input welding techniques such as a submerged arc welding, an electrogas welding, and an electroslag welding, are employed.
- steels exhibiting high weld-zone toughness need to be used for the weld-fabrication using the high heat input welding.
- the JP-A-2 267241 discloses a steel for a line pipe having excellent hydrogen induced cracking resistance and sulfide stress corrosion cracking resistance.
- a technique that has already been put into practical use employs a method of suppressing the coarsening of austenite grains according to fine dispersion of TiN and actions of TiN serving as a ferrite transformation nucleus.
- techniques have been developed for dispersing oxides of Ti ( Japanese Unexamined Patent Application Publication No. 57-51243 ) and for combining ferrite nucleation abilities BN ( Japanese Unexamined Patent Application Publication No. 62-170459 ).
- known techniques include a technique in which a high toughness is obtained by adding Ca and controlling the sulfide form ( Japanese Unexamined Patent Application Publication No. 60-204863 ) and a technique in which a high toughness is obtained by adding REM and controlling the sulfide form ( Japanese Unexamined Patent Application Publication No. 62-260041 ).
- JP 2002235114 discloses a method for producing a steel where ACR value is between 0 and 1 by which sufficient toughness can be obtained in a high heat input weld zone of >350 kJ/cm.
- an object of the present invention is to solve the above-described problems experienced with the conventional arts and to thereby provide a steel that enables a high weld HAZ toughness at the same level as that of a parent metal of the steel even after welding is performed with a high heat input exceeding 400 kJ/cm.
- the inventors carried out exhaustive researches. As a result, the inventors discovered that appropriate inclusion of Ca that is necessary for controlling the sulfide form is essential to improve the toughness of a weld heat affected zone (HAZ) even after welding performed with a high heat input exceeding 400 kJ/cm. More specifically, in order to improve the toughness of a high heat input weld HAZ, the inventors discovered that it is essential to suppress the coarsening of austenite grains in the high temperature region and to cause fine dispersion of the ferrite transformation nucleus that is necessary to accelerate ferrite transformation in the subsequent cooling stage. The conventional arts were insufficient in capability of achieving these essential factors.
- CaS is crystallized in the stage of solidification during formation of a steel plate from molten steel.
- CaS since CaS is crystallized at a lower temperature, CaS can be finely dispersed.
- MnS is precipitated over the surface of CaS when a sufficient amount of solute S after crystallization of CaS is secured by controlling the contents of Ca and S and the amount of oxygen dissolved in the steel.
- MnS itself has the ferrite nucleation ability and is effective to accelerate the ferrite transformation by forming Mn depleted zones.
- the inventors discovered that the ferrite transformation is further accelerated by causing ferrite nucleation nuclei of TiN, AlN, and the like that are precipitated over MnS. These countermeasures described above enable fine dispersion of the ferrite-transformation generation nucleus that does not dissolve even at the high temperature during the high heat input welding. Consequently, the weld HAZ structure can be transformed into a ferrite and pearlite microstructure having a high toughness.
- the present invention provides a steel for high heat input welding as defined in claim 1.
- the lower limit of the C content is set to 0.03 mass% to secure a strength necessary for using the steel as a structural steel.
- the upper limit of the C content is set to 0.15 mass% in consideration of deterioration in weld-crack resistance. More suitably, the C content is preferably limited to a range from 0.05 to 0.10 mass%.
- the Si content is required to be at least 0.05 mass% for steelmaking. However, with an Si content exceeding 0.25 mass%, the parent-metal toughness is deteriorated, and M-A (Martensite-Austenite) constituent is formed in a high heat input weld HAZ, whereby the toughness of the HAZ is deteriorated.
- M-A Martensite-Austenite
- the Si content is preferably limited to a range of from 0.13 to 0.22 mass%.
- Mn content of 0.5 mass% Mn or higher is required to secure a sufficient parent-metal strength.
- Mn content is preferably limited to a range of from 0.8 to 1.6 mass%.
- the P content is limited to 0.01 mass% or lower.
- An S content exceeding 0.0030 mass% acts to deteriorate the parent-metal toughness.
- the S content is limited to a range of from 0.0015 to 0.0025 mass% to generate CaS and MnS.
- an Al content of 0.015 mass% or higher is required to generate CaS and MnS.
- an Al content exceeding 0.1 mass% acts to deteriorate the parent-metal toughness and the weld metal toughness.
- the S content is preferably limited to a range of from 0.02 to 0.06 mass%.
- Ti is precipitated in the form of TiN upon solidification, thereby contributing to suppression of coarsening of austenite grains in the weld HAZ, and contributes, as a ferrite transformation nucleus, to improvement in the toughness of the weld HAZ.
- Ti content lower than 0.004 mass%, the above-described effect is low; and with a Ti content exceeding 0.03 mass%, TiN grains are coarsened, and a desired effect cannot be obtained. More suitably, the Ti content is preferably limited to a range of from 0.008 to 0.02 mass%.
- N is an element necessary to secure a required amount of TiN.
- a sufficient amount of TiN cannot be secured.
- an N content exceeding 0.0070 mass% the toughness is significantly deteriorated due to an increase in the amount of solute N in a region where TiN is dissolved by a weld-heating cycle. More suitably, the N content is preferably limited to a range of from 0.0030 to 0.0055 mass%.
- Ca has a toughness-improving effect with S being fixed.
- the Ca content is at least 0.0005 mass% to cause this effect to exhibit.
- the effect is saturated eveb with a Ca content exceeding 0.0030 mass%.
- the content is limited to the range of from 0.0005 to 0.0030 mass%. More suitably, the Ca content is preferably limited to a range of from 0.0010 to 0.0020 mass%.
- Fig. 1 shows the results of synthetic HAZ tests performed under two simulated hea-input conditions in which Ca was diversely added into a fundamental composition of the steel of the present invention.
- the toughness is significantly increased according to the relation 0.3 ⁇ ACR ⁇ 0.8 in either case where the time of 800-500°C cooling is 153 seconds or 270 seconds (improved by about 30°C in terms of vTrs).
- the composition appears in the form of a compound sulfide with either MnS or MnS and TiN precipitated over CaS.
- Fig. 3 is a schematic view showing the relationship between ACR and the sulfide to be precipitated.
- products formed by simultaneous precipitation of the compound sulfides of CaS and MnS and TiN exist.
- the quantity of the products is in a range of from 5 ⁇ 10 2 to 1 ⁇ 10 4 pieces/mm 2 , and the average grain size thereof is in a range of from 0.1 to 5 ⁇ m.
- the present invention allows a steel of an embodiment to contain at least one or two selected elements from the elements V, Nb, Cu, Ni, Cr, and Mo that have a strength-improving function, as described hereunder.
- B is effective in increasing hardenability during steel-plate manufacture. To secure this effect, the B content needs to be 0.0004 mass% or higher. However, addition of B exceeding 0.0010 mass% increases the hardenability, thereby decreasing the toughness of the weld HAZ.
- V has the effect of improving the parent-metal strength and toughness. This effect can be secured with a V content of 0.01 mass% or higher. However, addition of V exceeding 0.2 mass% causes deterioration in the toughness.
- Nb has the effect of enabling the parent-metal strength and toughness and the weld-joint strength to be secured. This effect can be secured with an Ni content of 0.007 mass% or higher. Addition Nb exceeding 0.05 mass% causes deterioration in the weld-HAZ toughness.
- Ni has the effect of maintaining high parent-metal toughness and concurrently increasing the strength thereof. This effect can be secured with an Nb content of 0.10 mass% or higher. With a Ni content exceeding 1.5 mass%, since the effect is saturated, the content is specified to be the upper limit.
- Cu exhibits an effect similar to that of Ni. This effect can be secured by including a Cu content of 0.10 mass% or higher. However, a Cu content exceeding 1.0 mass% causes hot embrittlement, thereby deteriorating the steel surface condition.
- Cr has the effect of increasing the parent-metal strength. This effect is secured with a Cr content of 0.05 mass% or higher. However, since addition of an excessive amount causes adverse effects on the toughness, the upper limit is set to 0.7 mass%.
- Mo Mo (Molybdenum): 0.7 Mass% Maximum
- Mo has the effect of increasing the parent-metal strength. This effect is secured with a Cr content of 0.05 mass% or higher. However, since addition of an excessive amount causes adverse effects on the toughness, the upper limit is set to 0.7 mass%.
- the compositions are each regulated in the content to the limited range. Therefore, the steel exhibiting a high toughness in the weld HAZ in the high heat input welding can be provided.
- the steel of the present invention is manufactured in, for example, a procedure as described hereunder.
- molten steel is refined using a convertor into steel.
- an RH (Ruhrstahl-Heraeus) degassing process is performed, and the steel is formed into slabs through continuous casting or ingot-casting-blooming steps.
- each of the slabs is reheated to a temperature of 1, 250°C or lower, and is then hot-rolled to a predetermined thickness in a temperature range of from a heating temperature to 650°C.
- the hot-rolled steel is subjected to either an air-cooling process or an accelerated cooling process at a cooling rate of from 1 to 40°C/sec.
- the cooling process is terminated at a temperature range of from 200 to 600°C, and air cooling is performed.
- the hot-rolled steel is directly hardened from a temperature range of 650°C or higher, and is then tempered to a temperature of 500°C ⁇ 150°C.
- the steel can be also manufactured according to a method selected from the steps wherein the hot-rolled steel is subjected to quenching after reheating in a temperature range of 850°C to 950°C and then, tempering to a temperature of 500°C ⁇ 150°C; the hot-rolled steel is reheated to a temperature of 1,000°C or lower and normalized; or the hot-rolled steel is reheated to a temperature of 1,000°C or lower and normalized and subsequently, tempered to a temperature of 650°C or lower.
- the manufacture can be achieved even under manufacturing conditions ordinarily used in hot rolling with a tandem roller being used.
- the steel plate according to the present invention is either a thick steel plate having a thickness of 6 mm or larger or a hot-rolled steel plate.
- a welding method to be used for the steel plate of the present invention is not limited to a specific one.
- the welding method may be an arc welding method, a submerged arc welding method, an electroslag welding method, an electrogas welding method, or any one of other heating-source welding techniques.
- test specimens having a size of 80 mm (width) x 80 mm (length) x 15 mm (thickness) were prepared to measure properties after being subjected to welding thermal cycles. These test specimens were subjected to a welding thermal cycle set such that the rate of cooling from 800°C to 500°C after heating to 1,400°C was set to 1°C/sec (equivalent to a weld HAZ in electrogas welding with a heat input of 450 kJ/cm). Then, the test specimens were each evaluated for the weld-HAZ toughness according to the result of a 2-mm V-notch Charpy impact test.
- Table 3 shows thus-obtained weld-HAZ toughnesses together with parent-metal strengths and toughnesses.
- the parent-metal strengths were each obtained such that two JIS-Z2201 based test specimens were prepared from 1/2t-thick portions in the rolled direction of each of the rolled plates. The two test specimens were each tested in conformity with
- the toughnesses were each measured such that three JIS-Z2201 based V-notch test specimens were prepared from 1/2t thick portions in the direction perpendicular to the rolled direction of the rolled plate. The three test specimens were each tested in conformity to JIS-Z2242 to measure a brittle-ductile fracture transition temperature (vTrs). The toughness (represented by the fracture transition temperature) of each of the parent metals and the weld HAZs was determined to be excellent in accordance with a criterion set to a vTrs of -40°C or lower.
- any one of inventive examples a high weld-HAZ toughness satisfying vTrs ⁇ -40°C was obtained.
- comparative examples were found to include those individually having low weld-HAZ toughnesses and even those individually having low parent-metal toughnesses.
- at least one of the value of (Ca - (0.18 + 130 x Ca) x O)/1.25/S and the contents of the compositions such as Ca, Ti, C, Mn, Si, S, N, Cu, Cr, Mo, V, and B was found to be out of the range specified in the present invention.
- a steel plate having a thickness of 60 mm was produced by hot rolling.
- a weld joint was produced by electrogas welding with a heat input of 450 kJ/cm, and a microstructure of a representative weld HAZ of a 1/4t thick portion was observed.
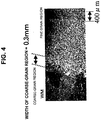
- Fig. 4 shows a microstructure taken of the inventive example steel 16
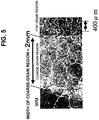
- Fig. 5 shows a microstructure taken of the comparative example steel 23. From these microstructures, grain-coarsening in the weld HAZ was found to appear conspicuously in the comparative example steel 23 shown in Fig. 5 . In comparison, however, the microstructure of the weld HAZ in the inventive example steel 16 shown in Fig. 4 was found to have been refined to the same level as that of the microstructure of the parent metal.
- a steel having a weld-HAZ toughness even after welding is performed with a high heat input of 400 kJ/cm or higher can be obtained.
- the present invention greatly contributes to improvement in the quality of a large structure that is fabricated by high heat input welding, such as submerged arc welding, electrogas welding, and/or electroslag welding.
- the steel has a high weld-HAZ toughness in a heat-input range of 400 kJ/cm or lower.
Landscapes
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Mechanical Engineering (AREA)
- Metallurgy (AREA)
- Organic Chemistry (AREA)
- Physics & Mathematics (AREA)
- Thermal Sciences (AREA)
- Crystallography & Structural Chemistry (AREA)
- Heat Treatment Of Steel (AREA)
Claims (1)
- Stahl zum Schweißen mit hohem Wärmeeintrag, dadurch gekennzeichnet, dass eine Zusammensetzung des Stalles umfasst:
C (Kohlenstoff) 0,03 bis 0,15 Masseprozent (Masse-%); Si (Silizium) 0,05 bis 0,25 Masse-%; Mn (Mangan) 0,5 bis 2,0 Masse-%; P (Phosphor) s 0,01 Masse-%; S (Schwefel) 0,0015 bis 0,0030 Masse-%; Al (Aluminium) 0,015 bis 0,1 Masse-%; Ti (Titan) 0,004 bis 0,03 Masse-%; N (Stickstoff) 0,0020 bis 0,0070 Masse-%; Ca (Calcium) 0,0005 bis 0,0030 Masse-%, B (Bor) 0,0004 bis 0,0010 Masse-%; und O (Sauerstoff) ≤ 0,0045 Masse-%; die Einzelgehalte von Ca, O (Sauerstoff) und S den folgenden Ausdruck (1) erfüllen und der Rest der Zusammensetzung Fe (Eisen) und unvermeidbare Verunreinigungen umfasst:V (Vanadium) maximal 0,2 Masse-%; Cu (Kupfer) maximal 1,0 Masse-%; Ni (Nickel) maximal 1,5 Masse-%; Cr (Chrom) maximal 0,7 Masse-%; Nb (Niob) maximal 0,05 Masse-% und Mo (Molybdän) maximal 0,7 Masse-%.
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| PCT/JP2002/008977 WO2004022807A1 (ja) | 2002-09-04 | 2002-09-04 | 大入熱溶接用鋼材およびその製造方法 |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| EP1533392A1 EP1533392A1 (de) | 2005-05-25 |
| EP1533392A4 EP1533392A4 (de) | 2005-12-07 |
| EP1533392B1 true EP1533392B1 (de) | 2017-08-02 |
Family
ID=31972301
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP02763002.9A Expired - Lifetime EP1533392B1 (de) | 2002-09-04 | 2002-09-04 | Stahlprodukt zum schweissen mit hohem wärmeeintrag und verfahren zu seiner herstellung |
Country Status (4)
| Country | Link |
|---|---|
| EP (1) | EP1533392B1 (de) |
| KR (1) | KR100622888B1 (de) |
| CN (1) | CN100402688C (de) |
| WO (1) | WO2004022807A1 (de) |
Families Citing this family (19)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP5124988B2 (ja) * | 2005-05-30 | 2013-01-23 | Jfeスチール株式会社 | 耐遅れ破壊特性に優れた引張強度900MPa以上の高張力鋼板およびその製造方法 |
| KR100722393B1 (ko) * | 2005-12-26 | 2007-05-28 | 주식회사 포스코 | 미세 복합석출물을 이용한 대입열 용접부 인성이 우수한고강도 강재 및 그 제조방법 |
| KR100722388B1 (ko) * | 2005-12-26 | 2007-05-28 | 주식회사 포스코 | 대입열 용접에 적용되는 용접용 강재 및 이의 제조 방법,그리고 대입열 용접에 의해 제조된 용접 구조물 |
| KR100957940B1 (ko) * | 2007-12-13 | 2010-05-13 | 주식회사 포스코 | 대입열 충격인성이 우수한 용접이음부를 포함하는용접구조용강 |
| JP5439887B2 (ja) * | 2008-03-31 | 2014-03-12 | Jfeスチール株式会社 | 高張力鋼およびその製造方法 |
| CN101736194B (zh) * | 2008-11-19 | 2012-11-28 | 攀钢集团研究院有限公司 | 一种车轮钢及其制备方法和车轮 |
| KR101333307B1 (ko) * | 2009-03-25 | 2013-11-27 | 신닛테츠스미킨 카부시키카이샤 | 가공성 및 담금질 후의 피로 특성이 우수한 전봉 강관 |
| CN101899614B (zh) * | 2010-08-27 | 2012-07-04 | 攀钢集团钢铁钒钛股份有限公司 | 一种含V、Nb复合微合金化的热轧钢板及其制备方法 |
| JP5480215B2 (ja) * | 2011-09-08 | 2014-04-23 | 株式会社神戸製鋼所 | 引張強さ780MPa以上の低降伏比厚肉円形鋼管用鋼板およびその製造方法、並びに引張強さ780MPa以上の低降伏比厚肉円形鋼管 |
| JP5857693B2 (ja) * | 2011-12-05 | 2016-02-10 | Jfeスチール株式会社 | 大入熱用鋼板およびその製造方法 |
| KR20160121712A (ko) | 2015-04-10 | 2016-10-20 | 동국제강주식회사 | 초대입열 용접용 강판의 제조 방법과 이에 의해 제조된 초대입열 용접용 강판 |
| WO2017135179A1 (ja) * | 2016-02-03 | 2017-08-10 | Jfeスチール株式会社 | 大入熱溶接用鋼材 |
| CN105839003B (zh) * | 2016-05-31 | 2017-09-26 | 江阴兴澄特种钢铁有限公司 | 一种正火态交货的180~200mm厚EH36钢板及其制备方法 |
| KR101889189B1 (ko) * | 2016-12-22 | 2018-08-16 | 주식회사 포스코 | 수소유기균열 저항성이 우수한 인장강도 450MPa급 후육 강재 및 그 제조방법 |
| CN107385353B (zh) * | 2017-06-19 | 2019-06-25 | 江阴兴澄特种钢铁有限公司 | 一种海洋平台用250mm 特厚EH36钢板及其制备方法 |
| CN108063108A (zh) * | 2018-01-22 | 2018-05-22 | 广安市嘉乐电子科技有限公司 | 一种mb桥堆焊接机 |
| KR102209581B1 (ko) | 2018-11-29 | 2021-01-28 | 주식회사 포스코 | 용접열영향부 인성이 우수한 강재 및 이의 제조방법 |
| KR20210105418A (ko) * | 2019-03-19 | 2021-08-26 | 제이에프이 스틸 가부시키가이샤 | 고망간강 주편의 제조 방법, 고망간강 강편 및 고망간강 강판의 제조 방법 |
| CN111519098B (zh) * | 2020-05-12 | 2021-06-15 | 首钢集团有限公司 | 一种低碳钢及控制低碳钢中夹杂物的脱氧方法 |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2002235114A (ja) * | 2001-02-05 | 2002-08-23 | Kawasaki Steel Corp | 大入熱溶接部靱性に優れた厚肉高張力鋼の製造方法 |
Family Cites Families (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS61157628A (ja) * | 1984-12-28 | 1986-07-17 | Nippon Steel Corp | 高靭性耐サワ−鋼管用ホツトコイルの製造方法 |
| JPS61270333A (ja) * | 1985-05-23 | 1986-11-29 | Sumitomo Metal Ind Ltd | 溶接部cod特性の優れた高張力鋼の製造方法 |
| JPS62112722A (ja) * | 1985-11-13 | 1987-05-23 | Nippon Steel Corp | 耐水素誘起割れ性及び耐硫化物応力腐食割れ性の優れた鋼板の製造方法 |
| JP2655911B2 (ja) * | 1989-04-07 | 1997-09-24 | 川崎製鉄株式会社 | 耐水素誘起割れ性および耐硫化物応力腐食割れ性に優れるラインパイプ用鋼 |
| JPH07173536A (ja) * | 1993-12-16 | 1995-07-11 | Nippon Steel Corp | 耐サワー性の優れた高強度ラインパイプ用鋼板の製造法 |
| JPH0995731A (ja) * | 1995-10-02 | 1997-04-08 | Nkk Corp | 低温用建築向け鋼材の製造方法 |
| WO2001027342A1 (fr) * | 1999-10-12 | 2001-04-19 | Nippon Steel Corporation | Acier pour structure soudee dont la tenacite de zone thermiquement affectee ne depend pas d'un apport de chaleur, et procede de production associe |
-
2002
- 2002-09-04 EP EP02763002.9A patent/EP1533392B1/de not_active Expired - Lifetime
- 2002-09-04 CN CNB028193466A patent/CN100402688C/zh not_active Expired - Lifetime
- 2002-09-04 WO PCT/JP2002/008977 patent/WO2004022807A1/ja not_active Ceased
- 2002-09-04 KR KR1020047004838A patent/KR100622888B1/ko not_active Expired - Lifetime
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2002235114A (ja) * | 2001-02-05 | 2002-08-23 | Kawasaki Steel Corp | 大入熱溶接部靱性に優れた厚肉高張力鋼の製造方法 |
Also Published As
| Publication number | Publication date |
|---|---|
| WO2004022807A1 (ja) | 2004-03-18 |
| KR100622888B1 (ko) | 2006-09-14 |
| CN100402688C (zh) | 2008-07-16 |
| EP1533392A4 (de) | 2005-12-07 |
| EP1533392A1 (de) | 2005-05-25 |
| CN1561403A (zh) | 2005-01-05 |
| KR20040040485A (ko) | 2004-05-12 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP2272994B1 (de) | Stahl mit hoher bruchfestigkeit und herstellungsverfahren dafür | |
| EP1533392B1 (de) | Stahlprodukt zum schweissen mit hohem wärmeeintrag und verfahren zu seiner herstellung | |
| EP2434027B1 (de) | Stahlmaterial zum schweissen mit hoher wärmebelastung | |
| CN101652495B (zh) | 高温特性和韧性优良的钢材及其制造方法 | |
| KR101846759B1 (ko) | 강판 및 그 제조 방법 | |
| JP4207334B2 (ja) | 溶接性と耐応力腐食割れ性に優れた高強度鋼板およびその製造方法 | |
| EP2400041B1 (de) | Stahlmaterial zum schweissen und herstellungsverfahren dafür | |
| EP3128033B1 (de) | Stahlplatte mit hoher zugfestigkeit und verfahren zur herstellung davon | |
| JP2002256379A (ja) | 大入熱溶接用鋼材 | |
| JPH0860292A (ja) | 溶接熱影響部靱性の優れた高張力鋼 | |
| JPH06128631A (ja) | 低温靱性の優れた高マンガン超高張力鋼の製造方法 | |
| JP2003166017A (ja) | 大入熱溶接部靱性に優れた厚肉高張力鋼材の製造方法 | |
| JP5233364B2 (ja) | 大入熱溶接用鋼材 | |
| JP5233365B2 (ja) | 大入熱溶接用鋼材 | |
| JP3440710B2 (ja) | フィレット部靱性に優れたh形鋼およびその製造方法 | |
| JP2688312B2 (ja) | 高強度高靭性鋼板 | |
| JP4981262B2 (ja) | 溶接部靭性に優れた低温用低降伏比鋼材の製造方法 | |
| JPH09194990A (ja) | 溶接熱影響部靱性の優れた高張力鋼 | |
| CN118679276A (zh) | 大线能量焊接用钢板及其制造方法 | |
| JP2002371338A (ja) | レーザー溶接部の靭性に優れた鋼 | |
| JPH093591A (ja) | 極厚高張力鋼板およびその製造方法 | |
| JP2000104116A (ja) | 強度と靱性に優れた鋼材の製造法 | |
| JP3736209B2 (ja) | 溶接部靭性に優れた高張力鋼及びその製造方法 | |
| JPH05154672A (ja) | 高強度および高靱性クラッド鋼板の製造法 | |
| CN117337341B (zh) | 高强度钢板及其制造方法 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| 17P | Request for examination filed |
Effective date: 20040330 |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): AT BE BG CH CY CZ DE DK EE ES FI FR GB GR IE IT LI LU MC NL PT SE SK TR |
|
| A4 | Supplementary search report drawn up and despatched |
Effective date: 20051020 |
|
| RIC1 | Information provided on ipc code assigned before grant |
Ipc: 7C 22C 38/04 B Ipc: 7C 22C 38/18 B Ipc: 7C 22C 38/02 A Ipc: 7C 22C 38/06 B Ipc: 7C 22C 38/14 B |
|
| RBV | Designated contracting states (corrected) |
Designated state(s): AT BE BG DE FR IT |
|
| 17Q | First examination report despatched |
Effective date: 20090310 |
|
| GRAP | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOSNIGR1 |
|
| INTG | Intention to grant announced |
Effective date: 20170216 |
|
| RBV | Designated contracting states (corrected) |
Designated state(s): DE FR IT |
|
| GRAS | Grant fee paid |
Free format text: ORIGINAL CODE: EPIDOSNIGR3 |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): DE FR IT |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R096 Ref document number: 60249001 Country of ref document: DE |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: PLFP Year of fee payment: 16 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: FR Payment date: 20170930 Year of fee payment: 16 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: IT Payment date: 20171031 Year of fee payment: 16 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R097 Ref document number: 60249001 Country of ref document: DE |
|
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| 26N | No opposition filed |
Effective date: 20180503 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20180904 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: FR Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20180930 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: DE Payment date: 20210727 Year of fee payment: 20 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R071 Ref document number: 60249001 Country of ref document: DE |