EP1300514A1 - Verwendung wässriger Aufhellpräparationen zum Aufhellen von natürlichen und synthetischen Materialien - Google Patents
Verwendung wässriger Aufhellpräparationen zum Aufhellen von natürlichen und synthetischen Materialien Download PDFInfo
- Publication number
- EP1300514A1 EP1300514A1 EP02021336A EP02021336A EP1300514A1 EP 1300514 A1 EP1300514 A1 EP 1300514A1 EP 02021336 A EP02021336 A EP 02021336A EP 02021336 A EP02021336 A EP 02021336A EP 1300514 A1 EP1300514 A1 EP 1300514A1
- Authority
- EP
- European Patent Office
- Prior art keywords
- aqueous
- brightener
- water
- preparations according
- paper
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
Classifications
-
- D—TEXTILES; PAPER
- D21—PAPER-MAKING; PRODUCTION OF CELLULOSE
- D21H—PULP COMPOSITIONS; PREPARATION THEREOF NOT COVERED BY SUBCLASSES D21C OR D21D; IMPREGNATING OR COATING OF PAPER; TREATMENT OF FINISHED PAPER NOT COVERED BY CLASS B31 OR SUBCLASS D21G; PAPER NOT OTHERWISE PROVIDED FOR
- D21H21/00—Non-fibrous material added to the pulp, characterised by its function, form or properties; Paper-impregnating or coating material, characterised by its function, form or properties
- D21H21/14—Non-fibrous material added to the pulp, characterised by its function, form or properties; Paper-impregnating or coating material, characterised by its function, form or properties characterised by function or properties in or on the paper
- D21H21/30—Luminescent or fluorescent substances, e.g. for optical bleaching
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06L—DRY-CLEANING, WASHING OR BLEACHING FIBRES, FILAMENTS, THREADS, YARNS, FABRICS, FEATHERS OR MADE-UP FIBROUS GOODS; BLEACHING LEATHER OR FURS
- D06L4/00—Bleaching fibres, filaments, threads, yarns, fabrics, feathers or made-up fibrous goods; Bleaching leather or furs
- D06L4/60—Optical bleaching or brightening
- D06L4/614—Optical bleaching or brightening in aqueous solvents
- D06L4/621—Optical bleaching or brightening in aqueous solvents with anionic brighteners
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06L—DRY-CLEANING, WASHING OR BLEACHING FIBRES, FILAMENTS, THREADS, YARNS, FABRICS, FEATHERS OR MADE-UP FIBROUS GOODS; BLEACHING LEATHER OR FURS
- D06L4/00—Bleaching fibres, filaments, threads, yarns, fabrics, feathers or made-up fibrous goods; Bleaching leather or furs
- D06L4/60—Optical bleaching or brightening
- D06L4/664—Preparations of optical brighteners; Optical brighteners in aerosol form; Physical treatment of optical brighteners
Definitions
- the invention relates to the use of aqueous preparations of optical brighteners for lightening natural and synthetic materials, processes for their manufacture and processes for the optical brightening of paper.
- optical brightening of paper comes in the paper mass, in the glue or Film press and in the paper line for use.
- lightening in particular of paper in the pulp must now increasingly ecological aspects, e.g. in the Meaning of avoidance of organic waste water loads and economic aspects e.g. in the sense of cost-effective procedures.
- State of Technology is the use of liquid brightener formulations, be it in the form of Solutions or suspensions, also called slurries or slurries.
- Stilbene brighteners containing sulfo or carboxylic acid groups are preferred, in particular Triazinylstilbene brighteners are used, which are often characterized by good substantivity on the one hand, but have a limited solubility in cold water on the other hand are marked. They already tend in purely aqueous solutions at low concentrations and ambient temperature ( ⁇ 40 ° C) too crystalline Precipitations. Storage-stable aqueous solutions, e.g. in DE-A-2 928 053 are therefore described, also contain considerable amounts of adjusting agents, Solution brokers such as Urea and / or organic solvents such as e.g. Polyalkylene.
- DE-A-2 715 864 also describes the advantages with regard to the dust behavior of Slurries over dry powders by drying and grinding of the water-moist press cake are obtained.
- DE-A-3 523 207 describes low-dust solid brightener preparations for use described in textile dye baths, which are mixed by spray-dried optical brightener formulation with an aromatic carboxylic acid ester or fatty acid polyol esters and are characterized by good hot water solubility Mark (80 ° C). However, such solutions are in higher Concentrations not long-term stable against crystalline precipitation.
- optical brighteners Common to all methods of applying optical brighteners in paper is that preparations of the optical brighteners are used which are based on a adequate stability and use at normal temperature are coordinated and which, in addition to the "active ingredient" of the optical brightener itself, also contains other ingredients for the Paper process need unwanted auxiliary materials. So require suspensions and Slurry i.A. organic dispersing and stabilizing agents, solid Preparations often require blending agents and / or dedusting agents. Known, at normal temperature, long-term stable solutions of optical brighteners are limited in their concentration at approx. 20% and require in large range of adjusting agents and / or co-solvents.
- aqueous brightener preparations in particular solutions of optical brighteners, with a temperature of 40 ° C to 98 ° C compared to solid preparations much easier and faster in water let bring in.
- the aqueous preparations to be used according to the invention preferably have a high content of optical brightener, are preferred largely free of undesirable organic components, and preferably free of crystalline brightener particles, especially their hydrate forms. they are therefore for the continuous and discontinuous brightening of synthetic or natural materials, preferably fiber materials, especially paper very suitable in terms of mass and / or surface.
- the brightener preparation consists of more than 90% by weight, preferably more than 95% by weight, particularly preferably more than 98% by weight from components a) and b).
- the aqueous brightener preparations are generally liquid.
- R 1 , R 2 and R 3 independently of one another denote -NH 2 , -NH-CH 3 , -NH-C 2 H 5 , -N (CH 3 ) 2 , -N (C 2 H 5 ) 2 , -NH -CH 2 CH 2 OH, -NH-C 2 -C 4 -hydroxyalkyl, -N (C 2 -C 4 -hydroxyalkyl) 2 -NH-CH 2 CH 2 SO 3 H; -NH-CH 2 -CH 2 -O-CH 2 -CH 2 -OH, -OCH 3 , -OCH (CH 3 ) 2 , -O-CH 2 -CH 2 -O-CH 3 , -N (CH 2 -CH 2 -OH) 2 , -N (CH 2 -CHOH-CH 3 ) 2 , morpholino, -SCH 3 , -N (CH 2 -CH 2 -OH) CH 2 -CH 2 -CON
- Preferred amino acid residues derive e.g. from glycine, sarcosine, ⁇ -alanine, Aspartic acid or iminodiacetic acid.
- M is preferably H, Na, Li, K, Ca, Mg, ammonium or ammonium, which is mono-, di-, tri- or terasubstituted by C 1 -C 4 alkyl, C 2 -C 4 hydroxyalkyl or a mixture it.
- optical brighteners are preferably in the form of their salts, in particular as Na or K salts.
- optical brighteners from the distilbene series are, for example, in EP-A-0 385 374.
- the cationic brighteners can, for example, by cationizing anionic, especially brighteners containing sulfo and / or carboxyl groups, for example by means of polymeric quaternary ammonium compounds become.
- Such brighteners are e.g. in WO-A 9967317.
- the aqueous brightener preparations in small amounts contain further, preferably ⁇ 10% by weight of auxiliaries.
- auxiliaries e.g. to name formulation aids such as fillers, surface-active agents and / or defoamers, as well as organic thickeners (protective colloids), preservatives and / or electrolytes.
- the aqueous brightener preparations preferably contain however, for ecological reasons only very small proportions of organic Aggregates, auxiliaries and / or contaminants, in particular in total less than 3% by weight, based on brightener of component a), very particularly less than 1% by weight.
- the aqueous preparations particularly preferably contain no organic co-solvents.
- the electrolyte is sodium chloride, sodium sulfate, sodium carbonate, or one of the corresponding potassium salts or mixtures of the substances mentioned.
- the amount of electrolyte can be less than or equal to 2% by weight, preferably 0.001 to 2 % By weight, based on the total weight of the aqueous preparation, in particular 0.001 to 0.5 wt .-%.
- Formulation aids of component c) contain, preferably
- sulfonated aromatics of Comp. A are also in the context of this application understood sulfomethylated aromatics.
- Preferred sulfonated aromatics are: Naphthalenesulfonic acids, phenolsulfonic acids, dihydroxybenzenesulfonic acids, sulfonated Ditolyl ether, sulfomethylated 4,4'-dihydroxydiphenyl sulfone, sulfonated diphenylmethane, sulfonated biphenyl, sulfonated hydroxybiphenyl, especially 2-hydroxybiphenyl, sulfonated terphenyl or benzenesulfonic acids.
- Aldehydes and / or ketones of component B) include, in particular, aliphatic, cycloaliphatic as well as aromatic in question.
- Aliphatic are preferred Aldehydes, with particular preference formaldehyde and other aliphatic aldehydes with 3 to 5 carbon atoms.
- Examples of unsulfonated aromatics of component C) are phenol, Cresol, 4,4'-dihydroxydiphenylsulfone or dihydroxydiphenylmethane in question.
- urea derivatives for example, dimethylol urea, melamine or Guanidine can be called.
- the condensation product preferably obtained in the condensation preferably has an average degree of condensation of 1 to 150, particularly preferred from 1 to 20, in particular from 1 to 5.
- condensation products of component c) can be in the form of an aqueous solution or Suspension or as a solid, for example as a powder or granules, preferably as spray-dried powder or granules.
- Preferred condensation products of component c) have an inorganic one Salinity below 10% by weight, preferably below 5% by weight, in particular below 1 wt .-%, based on the aqueous solution or suspension used Component or based on the solid of component c) used.
- Low monomer means a residual monomer content of less than 30% by weight, preferably less than 20 wt .-%, based on the condensation product, in particular ⁇ 10% by weight, preferably ⁇ 5% by weight.
- residual monomers in this context are used to produce the condensation product understood reactants used.
- Such low salt and low residual monomer condensation products are for example known from EP-A 816 406.
- the condensation products of component c) can be produced in this way, for example that the sulfonated aromatics of component A) optionally in a mixture with unsulfonated aromatics of component C) by reacting the underlying aromatics with a sulfonating agent preferably sulfuric acid, especially concentrated sulfuric acid, chlorosulfonic acid, Amidosulfonic acid or oleum.
- a sulfonating agent preferably sulfuric acid, especially concentrated sulfuric acid, chlorosulfonic acid, Amidosulfonic acid or oleum.
- the condensation then takes place with aldehydes and / or ketones of the components B), preferably formaldehyde, optionally together with others Compounds of component C).
- the condensation is preferably carried out in aqueous Solution at a pH of 0 to 9.
- aqueous Solution at a pH of 0 to 9.
- preferably per mol of sulfonated aromatics A) or per mole of a mixture of sulfonated aromatics of component A) and unsulfonated aromatics of component C) 0.4 to 1.5 mol, in particular 0.4 to 1.0 mol of component B) are used.
- the separation of the inorganic acid or its salts and the reduction the residual monomer content can, for example, by means of membrane separation processes be performed.
- the preferred membrane separation processes are Ultrafiltration, diffusion dialysis or electrodialysis in question.
- membranes preferably in ultrafiltration
- membranes have a molecular weightcut-off (MWCO) from 1,000 to 10,000 daltons.
- the separation of the inorganic acid using a membrane separation process is preferably carried out by diafiltration with acid-stable ultra or Cross-flow filtration nanofiltration membranes.
- suitable membranes include polyhydantoin membranes such as those found in EP-A 65 20 44 are known.
- Preferred membranes for this purpose have a MWCO level of 2,000 to 10,000 daltons. If necessary, concentration is carried out simultaneously in this process step.
- the aqueous brightener preparations can also contain organic thickeners contain.
- Suitable thickeners are those from the group of anionic or to name non-ionic organic water-soluble polymers. Particularly preferred are organic thickeners that have a solubility in water of> 100 g / l have.
- a compound is preferably used as the organic thickener, whose 4% aqueous solution has a viscosity of ⁇ 2 mPa.s at 20 ° C.
- Polysaccharides in particular, come as preferred anionic polyhydroxy compounds Xanthan and carboxymethyl cellulose in question.
- Copolymers of vinyl acetate are used in particular as thickeners fully or partially hydrolyzed vinyl alcohol copolymers understood, in particular fully hydrolyzed Copolymers of alkyl vinyl ester and vinyl acetate with a share of Alkyl vinyl esters of preferably 5 to 20 mol%, very particularly copolymers Alkyl vinyl acetate and vinyl acetate.
- Homopolymers and copolymers of N-vinylpyrrolidone also come as thickeners in question that completely disperse in water.
- the brighteners of component a), in particular of the general formula (I), are either known or can be prepared by methods known per se and are usually used as the free acid or as its salts, preferably alkali metal salts.
- aqueous brightener preparations e.g. by means of clarification filtration of undissolved Ingredients and unwanted by-products from brightener synthesis (e.g. Triazines) are exempt.
- the aqueous preparations can be washed with water, if necessary, together with the auxiliary substances mentioned, to a desired brightener concentration can be set.
- the use according to the invention is preferably characterized in that the aqueous brightener preparation, if necessary after dilution with water with the natural ones or synthetic materials, especially fiber materials is, preferably in an aqueous pulp mixture, paper coating slip or glue or film press fleet is introduced.
- the aqueous preparations can also be carried out by means of continuous Processes such as Filtered micro or ultrafiltration processes and if necessary be concentrated.
- continuous Processes such as Filtered micro or ultrafiltration processes and if necessary be concentrated.
- salts and low molecular weight still contained Components are eliminated particularly efficiently.
- aqueous brightener preparations for optical Lighten natural and synthetic materials especially fiber materials in aqueous liquors or media there is no restriction according to the invention.
- the use therefore affects e.g. B lightening textiles using known Dyeing process and application for lightening detergents.
- the use according to the invention of the aqueous brightener preparations relates lightening paper pulp in papermaking ("Mass staining"), e.g. Pulp, chemical and mechanical pulp, the Brightening up the coating slips and coatings commonly used in the paper industry lightening of unfilled, but especially filled, paper pulps and pigmented coating slips, as well as lightening in the glue or film press.
- Mass staining e.g. Pulp, chemical and mechanical pulp
- the use according to the invention is preferably characterized in that the aqueous brightener preparation if necessary after dilution with water the natural or synthetic materials, especially fiber materials is, preferably in an aqueous pulp mixture, paper coating slip or glue or film press fleet is introduced.
- the use according to the invention for the optical brightening of paper in the Mass and / or surface is preferably characterized in that the aqueous Brightener preparation continuously or discontinuously, preferably continuously to a concentration of 0.01-40% by weight. to Comp. a), in particular 1 - 25% by weight at a temperature of 15 - 75 ° C, in particular 20 - 55 ° C, and a pH of 6.0 - 12, in particular 6.5 - 11, very particularly 6.5 - 9.5, in the form their solution diluted with water and then the aqueous pulp mixture, Paper coating slip or glue or film press fleet is supplied or preferably undiluted in the form of the aqueous brightener preparations themselves introduced at a temperature of 40 to 98 ° C in the application media mentioned becomes.
- the aqueous brightener preparation itself or a aqueous dilution of the pulp at every stage of the papermaking process be added before leaf formation.
- Continuous is understood to mean that both the aqueous brightener preparation as well as an aqueous medium, generally Water, for example in a desired ratio. continuously brought together in an agitator tank or in a pipeline, and the mixture obtained to the stream or the dilution water the paper machine is fed in the desired ratio.
- aqueous medium generally Water
- the known coating slips generally contain as a binder and others Plastic dispersions based on copolymers of butadiene-styrene, acrylonitrile-butadiene-styrene, Acrylic acid esters, ethylene vinyl chloride or ethylene vinyl acetate or on Based on homopolymers, such as polyvinyl chloride, polyvinylidene chloride, polyethylene, Polyvinyl acetate or polyurethanes.
- a preferred binder is from styrene-butyl acrylate or styrene-butadiene-acrylic acid copolymers. Further polymer latices are described, for example, in US Pat. No. 3,265,654.
- Aluminum silicates such as, are usually used to pigment the coating slips China clay and kaolin, also barium sulfate, satin white, titanium dioxide or calcium carbonate in natural or felled form.
- the coating slip can be produced, for example, by the Components in any order at temperatures from 10 to 100 ° C, preferably 20 to 80 ° C, mixes.
- the components here also include the usual ones Tools used to regulate rheological properties, such as viscosity or water retention that coating slips can be used. Such tools are e.g.
- aqueous brightener preparations to be used according to the invention continue to be characterized by a number of advantages.
- liquid preparations they can be dosed well, at a temperature of 40 - 98 ° C, especially above 60 ° C very well storable, and can easily be stored in insulated, preferably heated, containers such as. Road tank containers are also transported in large units and possibly also be stored. Because of the very good stability of these preparations there is no need for stirred tanks in storage.
- the mixture was filtered at 80-90 ° C. through a depth filter and 890 g of a clear solution were obtained with an E1 / 1 value of 165 (measured at 350 nm wavelength).
- the aqueous brightener preparation thus obtained was over a period of more than 30 days at a temperature of 75 ° C storage stable and showed no crystalline Precipitation or segregation.
- Baykanol®SL (Bayer AG, formaldehyde condensation product of Comp. C) based on sulfonated ditolyl ether) based on the brightening agent added with stirring and completely dissolved.
- This aqueous brightener preparation was over a period of more than 30 days at a temperature of 65 ° C stable in storage and showed no crystalline precipitations or segregation.
- This aqueous brightener preparation was over a period of more than 30 days at a temperature of 65 ° C stable in storage and showed no crystalline precipitations or segregation.
- a solution prepared according to Example 2 was using a single-component nozzle dryer with an air inlet temperature of 220 ° C and an outlet temperature spray dried from 90 ° C with fine material recycling.
- a solid was obtained in Form of microgranules with 5.6% by weight residual moisture (determination by means of IR drying) and an E1 / 1 value of 550, which is deionized in 90 ° C hot Complete water at pH 8.7 with a concentration of up to 75% by weight dissolved.
- This aqueous brightener preparation with an E1 / 1 value of 445 was over a period of more than 14 days storage stable at a temperature of 85 ° C. and showed no crystalline precipitations or segregation. It had a liquid, especially pumpable Consistency.
- Example 5 As described in Example 5, but with the addition of 0.16% by weight of the hot aqueous brightener preparation according to Example 4 were also paper sheets comparable good white aspect and excellent levelness.
- the pH of the coating color was adjusted to 8-8.5 with dilute sodium hydroxide solution and the solids content of the coating color was adjusted to 60-65% by adding water.
- the coating color was divided into 3 parts and in each part of the coating color were the hot aqueous brightener preparations described in Examples 1 to 3 in a quantity of 10 g based on 1 kg of coating color homogeneous introduced and over a period of 10 min. mixed.
- the sheets coated in this way were dried in a drying cylinder at 95 ° C. for 1 min and then stored for 3 hours at 23 ° C and a humidity of 50%, before they were measured. In all 3 cases papers with a very high result high whiteness and good levelness.
- the pH of the liquors was about 7.
- the wet pickup was about 50-60%.
- An aqueous brightener preparation prepared according to Example 1 at one temperature of 75 ° C was continuous over a period of 1 hour (stationary operation) together by means of a heated gear pump in an amount of 350 kg / h with 650 l / h of 45 ° C, partially demineralized water in a 50 l stirred tank initiated with propeller stirrer and at the same time 1000 kg / h of the thus obtained concentrated solution taken from the bottom by means of a rotor-stator pump.
- the aqueous preparation was fed near the stirrer at 70% filled container. The stirrer speed was set so that sufficient Mixing effect with low foaming was achieved.
- the aqueous Auheller preparations could also be made with the same procedure according to the compositions from Examples 2 and 3 with comparably good Result to be resolved continuously.
- the watery Brightener preparation from Example 3 showed even better dissolving stability itself Concentrations above 22% by weight of the brightening agent.
- the whitening agent was added with stirring by adding the appropriate one Amount of hot aqueous brightener preparation from the examples 1-4 and then adding sodium sulfate.
- the liquor pick-up of the fabric was done by squeezing between the paddle rollers set to approx. 80%.
- the goods were then dried immediately through a stenter passage at 100 ° C for 30 seconds. Through this treatment a very good brightening effect was achieved on the goods.
- the whitening agent was added with stirring by adding the corresponding amount of hot aqueous brightener preparation from the examples 1-4.
- the goods were between rolls on a fleet absorption of about 80% of the Squeezed dry weight. Then the drying on the Stenter frame at 100 ° C for 30 seconds. Condensation was also carried out on the Stenter at 150 ° C for 4 minutes.
Landscapes
- Engineering & Computer Science (AREA)
- Textile Engineering (AREA)
- Paper (AREA)
- Cosmetics (AREA)
- Compositions Of Macromolecular Compounds (AREA)
Abstract
Description
- b) 85 - 15 Gew.-% Wasser,
- R1,R2 und R3
- unabhängig voneinander Phenoxy; mono- oder disulfoniertes
Phenoxy; Phenylamino; mono- oder disulfoniertes Phenylamino; Phenylamino
substituiert durch C1-C3-Alkyl, Cyano, Halogen, COOR, CONH-R,
NH-COR, SO2NH-R, O-R; Morpholino; Piperidino; Pyrrolidino; -OC1-C4-Alkyl;
-NH(C1-C4-Alkyl); -N(C1-C4-Alkyl)2; -NHC2-C4-Alkylen-OR; -N(C2-C4-Hydroxyalkyl)2; -NHC2-C4-Alkylen-O-C2-C4-Alkylen-OR; eine Aminosäure oder ein Aminosäureamid, von deren Aminogruppe ein Wasserstoffatom entfernt wurde; -NHCH2CH2OH; -N(CH2CH2OH)2; -N(CH3)(CH2CH2OH); -NH2; -OCH3; -S-C1-C4-Alkyl; -S-Aryl; -Cl; -NH-CH2CH2SO3H; -N(CH2CH2SO3H)2; oder -N(CH2CH2OH)CH2CH2CONH2 bedeuten, und - R
- H oder C1-C3-Alkyl und M den Rest eines Alkali-, Erdalkali-, Ammoniumoder Aminsalzes bedeutet. Unter Aminsalzionen sind solche der Formel H⊕NR4R5R6 bevorzugt, in denen R4, R5 und R6 unabhängig von einander Wasserstoff, Alkyl, Alkenyl, Hydroxyalkyl, Cyanoalkyl, Halogenalkyl oder Phenylalkyl bedeuten oder worin R4 und R5 zusammen die Ergänzung zu einem 5 bis 7-gliedrigen gesättigten Stickstoffheterocycluses darstellen, der noch zusätzlich ein Stickstoff- oder Sauerstoffatom als Ringglied enthalten kann, beispielsweise einen Piperidin, Piperazin, Pyrrolidin-, Imidazolin- oder Morpholinring, während R6 für Wasserstoff steht.
- R1 und R2
- für -N(CH2-CH2-OH)2 und
- R3
- für stehen und
- M
- die obige Bedeutung hat, insbesondere für Na+ oder K+ steht.
- Dextrine oder Cyclodextrine,
- Stärke und Stärkederivate, insbesondere abgebaute oder teilabgebaute Stärke,
- anionische Polyhydroxyverbindungen, insbesondere Xanthan oder Carboxymethylcellulose,
- Cellulosederivate wie z.B. Methylcellulose, insbesondere Hydroxymethyl-, Hydroxyethyl- oder Hydroxypropylcellulose,
- partiell hydrolysierte Polymerisate von Vinylacetat, vorzugsweise Polyvinylalkohol, die zu mehr als 70 % hydrolysiert sind und/oder Vinylalkohol-Copolymerisate, vorzugsweise Copolymere aus Vinylacetat und Alkylvinylester, die partiell oder vollständig verseift sind, sowie Polyinylalkohol selbst,
- Polymerisate von N-Vinylpyrrolidon, oder Copolymerisate mit Vinylestern,
- Polyacrylamide, vorzugsweise nichtionische oder anionische Polyacrylamide.
Claims (13)
- Verwendung wässriger Aufhellerpräparationen zur optischen Aufhellung von natürlichen oder synthetischen Materialien enthaltenddadurch gekennzeichnet, dass die Temperatur der Präparation 40 - 98°C, vorzugsweise 60 - 95°C beträgt.a) 15 bis 85 Gew.-%, insbesondere 20 bis 65 Gew.-% wenigstens eines wasserlöslichen optischen Aufhellers,
- Verwendung wässriger Aufhellerpräparationen gemäß Anspruch 1, dadurch gekennzeichnet, dass die Aufhellerpräparationen frei sind von kristallinen Aufhellerteilchen, insbesondere deren Hydratformen.
- Verwendung wässriger Aufhellerpräparationen gemäß Anspruch 1, dadurch gekennzeichnet, dass der wasserlösliche optische Aufheller der Komponente a) ausgewählt wird aus der Gruppe der sulfo- und/oder carboxylgruppenhaltigen Aufheller, insbesondere der Stilbenverbindungen.
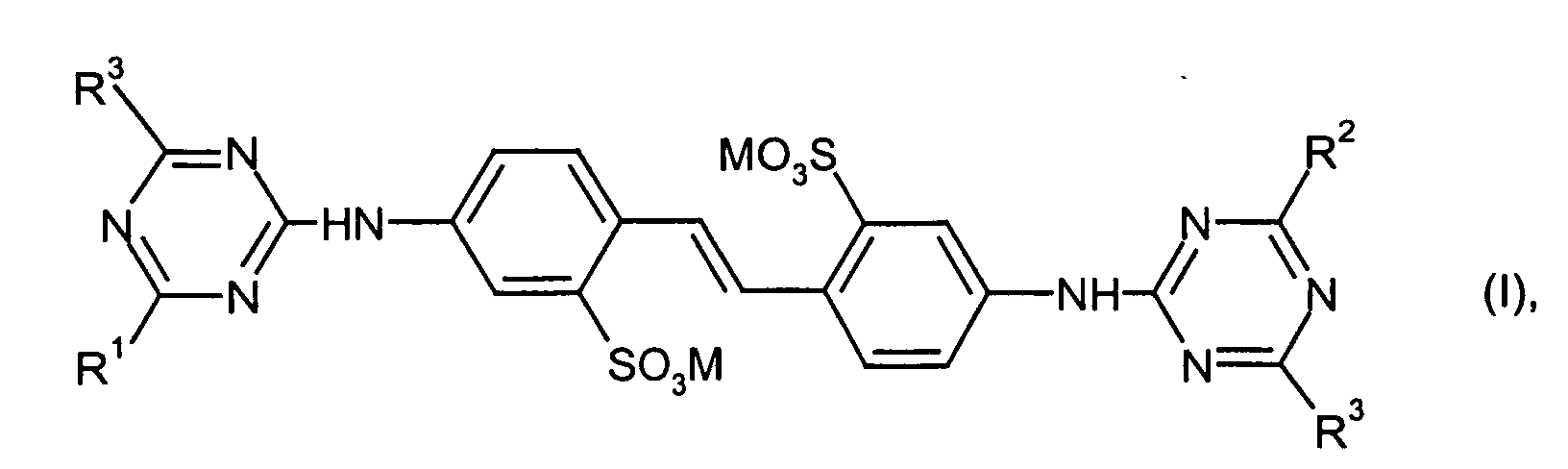
- Verwendung wässriger Aufhellerpräparationen gemäß Anspruch 1, dadurch gekennzeichnet, dass der wasserlösliche optische Aufheller der Komponente a) ausgewählt wird aus der Gruppe Distilbene oder der Triazinylflavonate der allgemeinen Formel (I) worin
- R1,R2 und R3
- unabhängig voneinander Phenoxy; mono- oder disulfoniertes
Phenoxy; Phenylamino; mono- oder disulfoniertes Phenylamino;
Phenylamino substituiert durch C1-C3-Alkyl, Cyano, Halogen, COOR,
CONH-R, NH-COR, SO2NH-R, O-R; Morpholino; Piperidino;
Pyrrolidino; -OC1-C4-Alkyl;
-NH(C1-C4-Alkyl); -N(C1-C4-Alkyl)2; -NHC2-C4-Alkylen-OR; -N(C2-C4-Hydroxyalkyl)2; -NHC2-C4-Alkylen-O-C2-C4-Alkylen-OR; eine Aminosäure oder ein Aminosäureamid, von deren Aminogruppe ein Wasserstoffatom entfernt wurde;
-NHCH2CH2OH; -N(CH2CH2OH)2; -N(CH3)(CH2CH2OH); -NH2; -OCH3;
-S-C1-C4-Alkyl; -S-Aryl; -Cl; -NH-CH2CH2SO3H;
-N(CH2CH2SO3H)2; oder
-N(CH2CH2OH)CH2CH2CONH2 bedeuten, und - R
- H oder C1-C3-Alkyl und M den Rest eines Alkali-, Erdalkali-, Ammonium- oder Aminsalzes bedeutet.
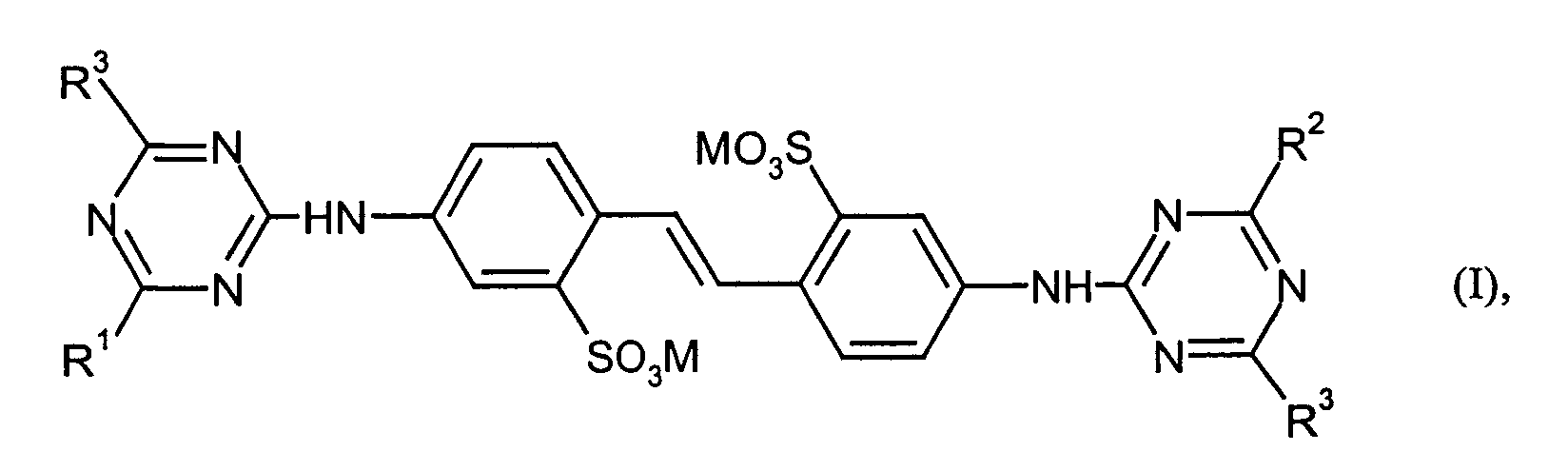
- Verwendung wässriger Aufhellerpräparationen gemäß Anspruch 1, dadurch gekennzeichnet, dass der wasserlösliche optische Aufheller der Komponente a) ausgewählt wird aus der Gruppe der Triazinylflavonate der Formel (I) worin
R1 , R2 und R3 unabhängig voneinander
-NH2, -NH-CH3, -NH-C2H5, -N(CH3)2, -N(C2H5)2, -NH-CH2CH2OH, -NH-C2-C4-Hydroxyalkyl, -N(C2-C4-Hydroxyalkyl)2, -NH-CH2CH2SO3H, -NH-CH2-CH2-O-CH2-CH2-OH, -OCH3, -OCH(CH3)2, -O-CH2-CH2-O-CH3, -N(CH2-CH2-OH)2, -N(CH2-CHOH-CH3)2, Morpholino, -SCH3, -N(CH2-CH2-OH)CH2-CH2-CONH2 sowie einen der nachstehenden Reste bedeuten, und M der Rest eines Alkali-, Erdalkali-, Ammonium- oder Aminsalzes bedeutet. - Verwendung wässriger Aufhellerpräparationen gemäß wenigstens einem der Ansprüche 1-6 enthaltend zusätzlichc) 0,1 - 5 Gew.-% bezogen auf Aufheller der Komp. a) wenigstens eines Kondensationsproduktes auf Basis vonA) sulfonierten AromatenB) Aldehyden und/oder Ketonen und gegebenenfallsC) einer oder mehrerer Verbindungen aus der Gruppe der nicht sulfonierten Aromaten, Harnstoff und Harnstoffderivaten.
- Verwendung wässriger Aufhellerpräparationen gemäß wenigstens einem der Ansprüche 1-7 enthaltend als Komp. c) ein Kondensationsprodukt auf Basis vonA) Ditolylethersulfonsäure undB) Formaldehyd oderA) Naphthalinsulfonsäure,B) Formaldehyd undC) 4,4'-Diphenylsulfon.
- Verwendung wässriger Aufhellerpräparationen gemäß Anspruch 1 zum optischen Aufhellen von Papier in der Masse.
- Verwendung wässriger Aufhellerpräparationen gemäß Anspruch 1 zum optischen Aufhellen von Papier in der Oberfläche, insbesondere in Papierstreichfarben und Leim- bzw. Filmpressenflotten.
- Verwendung wässriger Aufhellerpräparationen gemäß Anspruch 1, dadurch gekennzeichnet, dass die wässrige Aufhellerpräparation gegebenenfalls nach Verdünnung mit Wasser mit den synthetischen oder natürlichen Materialien, insbesondere Fasermaterialien zusammengebracht wird, vorzugsweise in eine wässrige Zellstoffmischung, Papierstreichmasse und/oder Leim- bzw. Filmpressenflotte eingebracht wird.
- Verwendung gemäß Anspruch 1 zum optischen Aufhellen von Papier in der Masse, dadurch gekennzeichnet, dass die wässrige Aufhellerpräparation in Wasser mit einer Konzentration von 0,01 bis 40 Gew.-%, bez. auf die Komponente a), insbesondere 1 bis 25 Gew.-% bei einer Temperatur von 10 bis 75°C, insbesondere 15 bis 55°C, und einem pH von 6,0 - 12, insbesondere 6,5 - 11, ganz besonders 6,5 - 9,5, eingebracht und der wässrigen Papiermasse zugegeben wird.
- Verwendung der wässrigen Aufhellerpräparationen gemäß Anspruch 1 zum Aufhellen von Polyamid, Cellulose, Papier und Waschmittel.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DE10149313A DE10149313A1 (de) | 2001-10-05 | 2001-10-05 | Verwendung wässriger Aufhellerpräparationen zum Aufhellen von natürlichen und synthetischen Materialien |
| DE10149313 | 2001-10-05 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| EP1300514A1 true EP1300514A1 (de) | 2003-04-09 |
| EP1300514B1 EP1300514B1 (de) | 2008-08-13 |
Family
ID=7701614
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP02021336A Expired - Lifetime EP1300514B1 (de) | 2001-10-05 | 2002-09-23 | Verwendung wässriger Aufhellpräparationen zum Aufhellen von natürlichen und synthetischen Materialien |
Country Status (6)
| Country | Link |
|---|---|
| US (1) | US20030089888A1 (de) |
| EP (1) | EP1300514B1 (de) |
| AT (1) | ATE404731T1 (de) |
| CA (1) | CA2406295A1 (de) |
| DE (2) | DE10149313A1 (de) |
| PT (1) | PT1300514E (de) |
Cited By (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2004111330A1 (en) * | 2003-06-11 | 2004-12-23 | Ciba Specialty Chemicals Holding Inc. | Storage-stable fluorescent whitener formulations |
| WO2006119201A1 (en) * | 2005-04-29 | 2006-11-09 | Hewlett-Packard Development Company, L.P. | Amphoteric stilbene composition |
| WO2011054655A3 (de) * | 2009-11-05 | 2011-10-06 | Weipatech Gmbh | Streichfarbe zur verbesserung der qualitäts- und laufeigenschaften von druckträgern und herstellungsverfahren der streichfarbe |
| CN103194086A (zh) * | 2013-03-14 | 2013-07-10 | 浙江传化华洋化工有限公司 | 一种含氨基丙酸衍生物荧光增白剂的合成方法 |
| US8603273B2 (en) | 2007-10-10 | 2013-12-10 | Miba Frictec Gmbh | Method of producing a friction lining |
| US8894815B2 (en) | 2007-12-12 | 2014-11-25 | Clariant Finance (Bvi) Limited | Storage stable solutions of optical brighteners |
Families Citing this family (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2009118248A2 (en) * | 2008-03-26 | 2009-10-01 | Clariant International Ltd | Improved optical brightening compositions |
| WO2006014563A2 (en) * | 2004-07-07 | 2006-02-09 | Novozymes North America, Inc | Use of cyclodextrin for reducing yellowing of pulp and paper |
| US20060124266A1 (en) * | 2004-11-23 | 2006-06-15 | Novozymes North America, Inc. | Use of cyclodextrins for reducing deposits during paper production |
| EP1712677A1 (de) * | 2005-04-08 | 2006-10-18 | Clariant International Ltd. | Wässerige Lösungen von optischen Aufhellern |
| US7682438B2 (en) | 2005-11-01 | 2010-03-23 | International Paper Company | Paper substrate having enhanced print density |
| JP2010500432A (ja) * | 2006-08-08 | 2010-01-07 | クラリアント ファイナンス (ビーブイアイ) リミティド | 光学的増白剤の水溶液 |
| EP2052021A1 (de) * | 2006-08-08 | 2009-04-29 | Clariant International Ltd. | Wässrige lösungen von optischen aufhellern |
| WO2009085308A2 (en) | 2007-12-26 | 2009-07-09 | International Paper Company | A paper substrate containing a wetting agent and having improved print mottle |
| US8613834B2 (en) | 2008-04-03 | 2013-12-24 | Basf Se | Paper coating or binding formulations and methods of making and using same |
| EP2135997B1 (de) * | 2008-06-11 | 2011-01-05 | Kemira Germany GmbH | Zusammensetzung und Verfahren zur Papierbleichung |
| WO2010039996A1 (en) * | 2008-10-01 | 2010-04-08 | International Paper Company | A paper substrate containing a wetting agent and having improved printability |
| EP2412870B1 (de) * | 2010-07-30 | 2013-04-17 | Blankophor GmbH & Co. KG | Zusammensetzung und Verfahren zur Papierbleichung |
| PL3246321T3 (pl) * | 2016-05-17 | 2019-02-28 | Blankophor Gmbh & Co. Kg | Fluorescencyjne środki wybielające i ich mieszaniny |
Citations (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB814579A (en) * | 1956-06-14 | 1959-06-10 | Cassella Farbwerke Mainkur Ag | Optical bleaching agents |
| DE1098485B (de) * | 1957-06-05 | 1961-02-02 | Bayer Ag | Aufhellungsmittel |
| DE1100583B (de) * | 1958-01-08 | 1961-03-02 | Bayer Ag | Aufhellungsmittel |
| US3272805A (en) * | 1960-07-28 | 1966-09-13 | Geigy Ag J R | Bis-triazinylaminostilbene compounds |
| US3954740A (en) * | 1973-02-02 | 1976-05-04 | Ciba-Geigy Corporation | Bis-s-triazinylamino-stilbene-2,2'-disulphonic acids, their manufacture and their use as optical brighteners |
| US3962115A (en) * | 1970-07-09 | 1976-06-08 | Ciba-Geigy Ag | Treatment of optical brightening agents |
| US5076968A (en) * | 1989-02-28 | 1991-12-31 | Ciba-Geigy Corporation | Aqueous storage-stable whitener formulation with an anionic polysaccharide stabilizer |
| EP0808837A1 (de) * | 1996-05-23 | 1997-11-26 | Ciba SC Holding AG | UV Stilbenverbindungen als optische Aufheller, als UV Absorber und deren Verwendung zur Erhöhung des Sonnenschutzfaktors von Geweben |
| EP0825188A1 (de) * | 1996-08-17 | 1998-02-25 | Ciba SC Holding AG | Triazinderivate als optische Aufheller, als UV Absorber und deren Verwendung zur Erhöhung des Sonnenschutzfaktors von Geweben |
| US5888400A (en) * | 1996-06-26 | 1999-03-30 | Bayer Aktiengesellschaft | Process for preparing low-salt preparations of condensation products |
| WO2001019804A1 (en) * | 1999-09-10 | 2001-03-22 | Ciba Specialty Chemicals Holding Inc. | Triazinylaminostilbene derivative as fluorescent whitening agents |
| DE19954959A1 (de) * | 1999-11-16 | 2001-05-17 | Henkel Kgaa | Umhüllte teilchenförmige Peroxoverbindungen |
Family Cites Families (21)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE1495645C3 (de) * | 1961-05-26 | 1974-11-07 | Bayer Ag, 5090 Leverkusen | Verfahren zur Herstellung stabiler, wäßriger Emulsionen bzw. Dispersionen von Äthylen-Mischpolymerisaten |
| BE755820A (fr) * | 1969-09-12 | 1971-03-08 | Sandoz Sa | Composition de blanchiment optique |
| BE786308A (fr) * | 1971-07-16 | 1973-01-15 | Ciba Geigy | Acides bis-s-triazinylamino-stilbene-2,2' -disulfoniques, leur preparation et leur utilisation comme azurants optiques |
| JPS5116060B2 (de) * | 1972-08-04 | 1976-05-21 | ||
| CH587831A5 (de) * | 1973-05-17 | 1977-05-13 | Sandoz Ag | |
| CH583211A5 (de) * | 1973-05-22 | 1976-12-31 | Sandoz Ag | |
| US4167626A (en) * | 1974-08-14 | 1979-09-11 | Sandoz Ltd. | Triazolyl stilbene triazoles |
| CH596290A5 (de) * | 1975-01-22 | 1978-03-15 | Ciba Geigy Ag | |
| US4212763A (en) * | 1977-07-04 | 1980-07-15 | Ciba-Geigy Corporation | Bis-triazinylaminostilbene compounds and their use as fluorescent brightening agents |
| US4364845A (en) * | 1978-07-17 | 1982-12-21 | Ciba-Geigy Corporation | Concentrated aqueous solutions of sulfo group-containing fluorescent brighteners which are stable on storage |
| US4339238A (en) * | 1980-01-14 | 1982-07-13 | Ciba-Geigy Corporation | Stable aqueous formulations of stilbene fluorescent whitening agents |
| CH647021A5 (de) * | 1981-09-22 | 1984-12-28 | Ciba Geigy Ag | Verfahren zur herstellung lagerstabiler aufhellerformulierungen. |
| US4762868A (en) * | 1985-06-13 | 1988-08-09 | North Broken Hill Limited | Coated front copy paper |
| DE4338196A1 (de) * | 1993-11-09 | 1995-05-11 | Bayer Ag | Verfahren zum Reinigen organischer Syntheseprodukte |
| DE4343857A1 (de) * | 1993-12-22 | 1995-06-29 | Hoechst Ag | Öl-in-Wasser-Emulsionen |
| GB9412590D0 (en) * | 1994-06-23 | 1994-08-10 | Sandoz Ltd | Organic compounds |
| US5591489A (en) * | 1995-05-04 | 1997-01-07 | Sequa Chemicals, Inc. | Process for surface sizing paper or paperboard |
| GB9813248D0 (en) * | 1998-06-22 | 1998-08-19 | Clariant Int Ltd | Improvements in or relating to organic compounds |
| AU1143401A (en) * | 1999-10-29 | 2001-05-14 | Ciba Specialty Chemicals Holding Inc. | Process for the preparation of solutions of anionic organic compounds |
| EP1283856B1 (de) * | 2000-05-26 | 2007-01-24 | Ciba SC Holding AG | Verfahren zur herstellung von lösungen anionischer organischer verbindungen |
| GB0100610D0 (en) * | 2001-01-10 | 2001-02-21 | Clariant Int Ltd | Improvements in or relating to organic compounds |
-
2001
- 2001-10-05 DE DE10149313A patent/DE10149313A1/de not_active Withdrawn
-
2002
- 2002-09-20 US US10/251,561 patent/US20030089888A1/en not_active Abandoned
- 2002-09-23 PT PT02021336T patent/PT1300514E/pt unknown
- 2002-09-23 AT AT02021336T patent/ATE404731T1/de active
- 2002-09-23 EP EP02021336A patent/EP1300514B1/de not_active Expired - Lifetime
- 2002-09-23 DE DE50212628T patent/DE50212628D1/de not_active Expired - Lifetime
- 2002-10-02 CA CA002406295A patent/CA2406295A1/en not_active Abandoned
Patent Citations (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB814579A (en) * | 1956-06-14 | 1959-06-10 | Cassella Farbwerke Mainkur Ag | Optical bleaching agents |
| DE1098485B (de) * | 1957-06-05 | 1961-02-02 | Bayer Ag | Aufhellungsmittel |
| DE1100583B (de) * | 1958-01-08 | 1961-03-02 | Bayer Ag | Aufhellungsmittel |
| US3272805A (en) * | 1960-07-28 | 1966-09-13 | Geigy Ag J R | Bis-triazinylaminostilbene compounds |
| US3962115A (en) * | 1970-07-09 | 1976-06-08 | Ciba-Geigy Ag | Treatment of optical brightening agents |
| US3954740A (en) * | 1973-02-02 | 1976-05-04 | Ciba-Geigy Corporation | Bis-s-triazinylamino-stilbene-2,2'-disulphonic acids, their manufacture and their use as optical brighteners |
| US5076968A (en) * | 1989-02-28 | 1991-12-31 | Ciba-Geigy Corporation | Aqueous storage-stable whitener formulation with an anionic polysaccharide stabilizer |
| EP0808837A1 (de) * | 1996-05-23 | 1997-11-26 | Ciba SC Holding AG | UV Stilbenverbindungen als optische Aufheller, als UV Absorber und deren Verwendung zur Erhöhung des Sonnenschutzfaktors von Geweben |
| US5888400A (en) * | 1996-06-26 | 1999-03-30 | Bayer Aktiengesellschaft | Process for preparing low-salt preparations of condensation products |
| EP0825188A1 (de) * | 1996-08-17 | 1998-02-25 | Ciba SC Holding AG | Triazinderivate als optische Aufheller, als UV Absorber und deren Verwendung zur Erhöhung des Sonnenschutzfaktors von Geweben |
| WO2001019804A1 (en) * | 1999-09-10 | 2001-03-22 | Ciba Specialty Chemicals Holding Inc. | Triazinylaminostilbene derivative as fluorescent whitening agents |
| DE19954959A1 (de) * | 1999-11-16 | 2001-05-17 | Henkel Kgaa | Umhüllte teilchenförmige Peroxoverbindungen |
Cited By (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2004111330A1 (en) * | 2003-06-11 | 2004-12-23 | Ciba Specialty Chemicals Holding Inc. | Storage-stable fluorescent whitener formulations |
| WO2006119201A1 (en) * | 2005-04-29 | 2006-11-09 | Hewlett-Packard Development Company, L.P. | Amphoteric stilbene composition |
| US7789917B2 (en) | 2005-04-29 | 2010-09-07 | Hewlett-Packard Development Company, L.P. | Amphoteric stilbene composition |
| CN101263256B (zh) * | 2005-04-29 | 2011-07-06 | 惠普开发有限公司 | 两性二苯乙烯组合物 |
| US8603273B2 (en) | 2007-10-10 | 2013-12-10 | Miba Frictec Gmbh | Method of producing a friction lining |
| US8894815B2 (en) | 2007-12-12 | 2014-11-25 | Clariant Finance (Bvi) Limited | Storage stable solutions of optical brighteners |
| WO2011054655A3 (de) * | 2009-11-05 | 2011-10-06 | Weipatech Gmbh | Streichfarbe zur verbesserung der qualitäts- und laufeigenschaften von druckträgern und herstellungsverfahren der streichfarbe |
| CN103194086A (zh) * | 2013-03-14 | 2013-07-10 | 浙江传化华洋化工有限公司 | 一种含氨基丙酸衍生物荧光增白剂的合成方法 |
| CN103194086B (zh) * | 2013-03-14 | 2014-07-02 | 浙江传化华洋化工有限公司 | 一种含氨基丙酸衍生物荧光增白剂的合成方法 |
Also Published As
| Publication number | Publication date |
|---|---|
| DE50212628D1 (de) | 2008-09-25 |
| CA2406295A1 (en) | 2003-04-05 |
| ATE404731T1 (de) | 2008-08-15 |
| EP1300514B1 (de) | 2008-08-13 |
| PT1300514E (pt) | 2008-11-20 |
| US20030089888A1 (en) | 2003-05-15 |
| DE10149313A1 (de) | 2003-04-17 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP1300514B1 (de) | Verwendung wässriger Aufhellpräparationen zum Aufhellen von natürlichen und synthetischen Materialien | |
| EP1355004B1 (de) | Verwendung von Aufhellern zur Herstellung von Streichmassen | |
| EP1049745B1 (de) | Wässrige pigmentpräparationen | |
| DE3234784C2 (de) | ||
| DE69807397T3 (de) | Triazinylaminostilben Verbindungen | |
| EP0992547B1 (de) | Verfahren zur Herstellung von Farbstoffen und/oder Aufhellerzubereitungen | |
| DE3707221A1 (de) | Kationisch eingestellte pigmentdispersion und streichfarbe | |
| EP0274666B1 (de) | Weisstönerhaltige Papierstreichmassen | |
| DE4237545A1 (de) | Kupferphthalocyanin-Flüssigformierung | |
| DE3535566A1 (de) | Natriumlignosulfonate mit niedrigem elektrolytgehalt | |
| DD144934A5 (de) | Lagerstabile,konzentrierte waessrige loesungen von sulfogruppenhaltigen optischen aufhellern | |
| DE3235747A1 (de) | Verfahren zur herstellung von lagerstabilen farbstoffpraeparaten | |
| EP1624105B1 (de) | Alkanolammoniumhaltige Triazinylflavonataufheller | |
| EP0059782A1 (de) | Verfahren zur Herstellung konzentrierter wässriger Farbstoffpräparate von anionischen Papier- oder Wollfarbstoffen | |
| DE2715864A1 (de) | Verfahren zum optischen aufhellen von papier | |
| EP1177180B1 (de) | Stilbenaufheller | |
| CH689833A5 (de) | Cu-Phthalocyaninfarbstoffmischungen, ihre Herstellung und Verwendung zum Faerben oder Bedrucken von cellulosehaltigem Material oder als Tintenstrahldruckfluessigkeit. | |
| EP1300513A2 (de) | Verwendung fester Aufhellerpräparationen zum Aufhellen von Papier | |
| DE10150894A1 (de) | Feste Aufhellerpräparationen | |
| EP1335001A1 (de) | Feste Aufhellerpräparationen | |
| EP0382054A2 (de) | Chemisch modifizierte Proteine | |
| CN1910241B (zh) | 染料的浓缩含水组合物 | |
| CH648584A5 (en) | Process for preparing concentrated aqueous formulations of anionic dyes | |
| JP5587767B2 (ja) | 安定な液体配合物 | |
| CH694887A5 (de) | Verfahren zur Herstellung von Farbstoffen und/oder optischer Aufheller. |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): AT BE BG CH CY CZ DE DK EE ES FI FR GB GR IE IT LI LU MC NL PT SE SK TR |
|
| AX | Request for extension of the european patent |
Extension state: AL LT LV MK RO SI |
|
| 17P | Request for examination filed |
Effective date: 20031009 |
|
| AKX | Designation fees paid |
Designated state(s): AT BE BG CH CY CZ DE DK EE ES FI FR GB GR IE IT LI LU MC NL PT SE SK TR |
|
| RAP1 | Party data changed (applicant data changed or rights of an application transferred) |
Owner name: BAYER CHEMICALS AG |
|
| 17Q | First examination report despatched |
Effective date: 20040813 |
|
| RAP1 | Party data changed (applicant data changed or rights of an application transferred) |
Owner name: LANXESS DEUTSCHLAND GMBH |
|
| 17Q | First examination report despatched |
Effective date: 20040813 |
|
| RAP1 | Party data changed (applicant data changed or rights of an application transferred) |
Owner name: KEMIRA OYJ |
|
| RAP1 | Party data changed (applicant data changed or rights of an application transferred) |
Owner name: KEMIRA OYJ |
|
| GRAP | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOSNIGR1 |
|
| GRAS | Grant fee paid |
Free format text: ORIGINAL CODE: EPIDOSNIGR3 |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): AT BE BG CH CY CZ DE DK EE ES FI FR GB GR IE IT LI LU MC NL PT SE SK TR |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: FG4D Free format text: NOT ENGLISH |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: EP |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: FG4D Free format text: LANGUAGE OF EP DOCUMENT: GERMAN |
|
| REF | Corresponds to: |
Ref document number: 50212628 Country of ref document: DE Date of ref document: 20080925 Kind code of ref document: P |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: NV Representative=s name: ING. MARCO ZARDI C/O M. ZARDI & CO. S.A. |
|
| REG | Reference to a national code |
Ref country code: PT Ref legal event code: SC4A Free format text: AVAILABILITY OF NATIONAL TRANSLATION Effective date: 20081110 |
|
| REG | Reference to a national code |
Ref country code: SE Ref legal event code: TRGR |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: NL Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20080813 Ref country code: ES Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20081124 |
|
| BERE | Be: lapsed |
Owner name: KEMIRA OYJ Effective date: 20080930 |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: FD4D |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20080813 Ref country code: DK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20080813 Ref country code: BG Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20081113 Ref country code: MC Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20080930 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: CZ Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20080813 |
|
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| 26N | No opposition filed |
Effective date: 20090514 |
|
| GBPC | Gb: european patent ceased through non-payment of renewal fee |
Effective date: 20081113 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: EE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20080813 Ref country code: BE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20080930 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GB Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20081113 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LU Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20080923 Ref country code: CY Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20080813 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: TR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20080813 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20081114 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: IT Payment date: 20100928 Year of fee payment: 9 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R081 Ref document number: 50212628 Country of ref document: DE Owner name: BLANKOPHOR GMBH & CO. KG, DE Free format text: FORMER OWNER: KEMIRA OYJ, HELSINKI, FI Effective date: 20110519 |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PUE Owner name: BLANKOPHOR GMBH & CO. KG Free format text: KEMIRA OYJ#PORKKALANKATU 3#00180 HELSINKI (FI) -TRANSFER TO- BLANKOPHOR GMBH & CO. KG#MARIE-CURIE-STRASSE 10#51377 LEVERKUSEN (DE) |
|
| REG | Reference to a national code |
Ref country code: PT Ref legal event code: PC4A Owner name: BLANKOPHOR GMBH & CO. KG, DE Effective date: 20110908 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: CH Payment date: 20110926 Year of fee payment: 10 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: SK Payment date: 20110916 Year of fee payment: 10 Ref country code: SE Payment date: 20110923 Year of fee payment: 10 Ref country code: FI Payment date: 20110922 Year of fee payment: 10 Ref country code: DE Payment date: 20110916 Year of fee payment: 10 Ref country code: AT Payment date: 20110922 Year of fee payment: 10 Ref country code: FR Payment date: 20111005 Year of fee payment: 10 Ref country code: PT Payment date: 20110909 Year of fee payment: 10 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: TP Owner name: BLANKOPHOR GMBH & CO. KG, DE Effective date: 20111021 |
|
| REG | Reference to a national code |
Ref country code: SK Ref legal event code: PC4A Ref document number: E 4537 Country of ref document: SK Owner name: BLANKOPHOR GMBH & CO. KG, LEVERKUSEN, DE Free format text: FORMER OWNER: KEMIRA OYJ, HELSINKI, FI Effective date: 20110822 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R082 Ref document number: 50212628 Country of ref document: DE Representative=s name: PATENT- UND RECHTSANWAELTE BARDEHLE PAGENBERG, DE |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R082 Ref document number: 50212628 Country of ref document: DE Representative=s name: BARDEHLE PAGENBERG PARTNERSCHAFT PATENTANWAELT, DE Effective date: 20120227 Ref country code: DE Ref legal event code: R081 Ref document number: 50212628 Country of ref document: DE Owner name: BLANKOPHOR GMBH & CO. KG, DE Free format text: FORMER OWNER: BLANKOPHOR GMBH & CO. KG, 51377 LEVERKUSEN, DE Effective date: 20120227 |
|
| REG | Reference to a national code |
Ref country code: PT Ref legal event code: MM4A Free format text: LAPSE DUE TO NON-PAYMENT OF FEES Effective date: 20130325 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20120924 Ref country code: FI Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20120923 |
|
| REG | Reference to a national code |
Ref country code: SE Ref legal event code: EUG Ref country code: CH Ref legal event code: PL |
|
| REG | Reference to a national code |
Ref country code: AT Ref legal event code: MM01 Ref document number: 404731 Country of ref document: AT Kind code of ref document: T Effective date: 20120923 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: PT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20130325 |
|
| REG | Reference to a national code |
Ref country code: SK Ref legal event code: MM4A Ref document number: E 4537 Country of ref document: SK Effective date: 20120923 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: ST Effective date: 20130531 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LI Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20120930 Ref country code: AT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20120923 Ref country code: DE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20130403 Ref country code: SK Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20120923 Ref country code: CH Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20120930 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20120923 Ref country code: FR Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20121001 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R119 Ref document number: 50212628 Country of ref document: DE Effective date: 20130403 |