EP1001942B1 - Polyhydroxybutylpyrazines, leur preparation et medicaments les contenant - Google Patents
Polyhydroxybutylpyrazines, leur preparation et medicaments les contenant Download PDFInfo
- Publication number
- EP1001942B1 EP1001942B1 EP98939677A EP98939677A EP1001942B1 EP 1001942 B1 EP1001942 B1 EP 1001942B1 EP 98939677 A EP98939677 A EP 98939677A EP 98939677 A EP98939677 A EP 98939677A EP 1001942 B1 EP1001942 B1 EP 1001942B1
- Authority
- EP
- European Patent Office
- Prior art keywords
- pyrazin
- butane
- radical
- dihydroxybutyl
- amino
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Lifetime
Links
- 0 *[Si]1O[Si](*)(*)OCC(C(C(c2ncc(/C=C(\C(CO3)O[Si](*)(*)OI3S(*)*)/O)nc2)O)O)O1 Chemical compound *[Si]1O[Si](*)(*)OCC(C(C(c2ncc(/C=C(\C(CO3)O[Si](*)(*)OI3S(*)*)/O)nc2)O)O)O1 0.000 description 15
- BYRBMGCYGVYXRM-UHFFFAOYSA-N CC(C)(OC1)OC1C(Cc1ncc(C2OC(C)(C)OC2C2OC(C)(C)OC2)nc1)O Chemical compound CC(C)(OC1)OC1C(Cc1ncc(C2OC(C)(C)OC2C2OC(C)(C)OC2)nc1)O BYRBMGCYGVYXRM-UHFFFAOYSA-N 0.000 description 3
- FWQUGRFQILYGDL-UHFFFAOYSA-N CC(C)(OC1)OC1C1OC(C)(C)OC1C1NC=C(CC2OC(C)(C)OCC2O)N=C1 Chemical compound CC(C)(OC1)OC1C1OC(C)(C)OC1C1NC=C(CC2OC(C)(C)OCC2O)N=C1 FWQUGRFQILYGDL-UHFFFAOYSA-N 0.000 description 2
- VCHQERIEGVPPRN-UHFFFAOYSA-N CC(C)(OC1)OC1C1OC(C)(C)OC1C1N(C)C=C(CC2OC(C)(C)OCC2O)N=C1 Chemical compound CC(C)(OC1)OC1C1OC(C)(C)OC1C1N(C)C=C(CC2OC(C)(C)OCC2O)N=C1 VCHQERIEGVPPRN-UHFFFAOYSA-N 0.000 description 1
- BTWMGQSTXIBPIO-UHFFFAOYSA-N CC(C)(OC1)OC1C1OC(C)(C)OC1c1ncc(CC2OC(C)(C)OCC2O)nc1 Chemical compound CC(C)(OC1)OC1C1OC(C)(C)OC1c1ncc(CC2OC(C)(C)OCC2O)nc1 BTWMGQSTXIBPIO-UHFFFAOYSA-N 0.000 description 1
- RNZITFYEFXOHMY-UHFFFAOYSA-N CC(Cc1ncc(C2OC(C)(C)OC2C2OC(C)(C)OC2)nc1)C1OC(C)(C)OC1 Chemical compound CC(Cc1ncc(C2OC(C)(C)OC2C2OC(C)(C)OC2)nc1)C1OC(C)(C)OC1 RNZITFYEFXOHMY-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D241/00—Heterocyclic compounds containing 1,4-diazine or hydrogenated 1,4-diazine rings
- C07D241/02—Heterocyclic compounds containing 1,4-diazine or hydrogenated 1,4-diazine rings not condensed with other rings
- C07D241/10—Heterocyclic compounds containing 1,4-diazine or hydrogenated 1,4-diazine rings not condensed with other rings having three double bonds between ring members or between ring members and non-ring members
- C07D241/12—Heterocyclic compounds containing 1,4-diazine or hydrogenated 1,4-diazine rings not condensed with other rings having three double bonds between ring members or between ring members and non-ring members with only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, directly attached to ring carbon atoms
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/08—Drugs for disorders of the metabolism for glucose homeostasis
- A61P3/10—Drugs for disorders of the metabolism for glucose homeostasis for hyperglycaemia, e.g. antidiabetics
Definitions
- the present invention relates to medicaments containing as active principle at least one compound of formula: or one of its stereoisomers or one of its salts, the new compounds of formula (I), their stereoisomers and their salts and their preparation process.
- radicals and portions alkyl and alkoxy contain 1 to 6 carbon atoms in a straight chain or branched and the halogen atoms are the chlorine, fluorine, iodine and bromine.
- the compounds of formula (I) comprising several asymmetric carbons have stereoisomeric forms. These different stereoisomers are part of the invention.
- Het represents a heterocycle chosen from cycles 2-, 3-, or 4-pyridyl, imidazolyl, thiazolyl and oxazolyl.
- the compound of formula: is known (Carbohydr. Res., 77, 205 (1979)) but no pharmacological property is described for it.
- the preferred compounds of formula (I) are the compounds mentioned in lists of preferred drugs except the compound of formula (A).
- R 9 and R 10 each represent a radical -CH 2 OH and either R 3 represents a methylene radical
- R 4 represents a radical -CHOH- and one of the radicals R 1 , R 2 , R 5 and R 6 represent a carbonyl radical and the others each represent a radical -CHOH-
- ie R 1 and R 6 each represent a radical -CHOH-
- R 3 represents a methylene radical and R 2 , R 4 and R 5 are identical and each represents a carbonyl radical
- R 2 and R 5 each represent a radical -CHOH-
- R 3 represents a methylene radical and R 1 , R 4 and R 6 are identical and each represent a carbonyl radical
- Ra represents a trialkylsilyl, alkyldiphenylsilyl or dialkylphenylsilyl radical
- Rb represents a phenyl radical optionally substituted by at least one
- Preferred trialkylsilyl, alkyldiphenylsilyl or dialkylphenylsilyl radicals are the trimethylsilyl, tert-butyldiphenylsilyl radicals and dimethylphenylsilyl.
- the oxidation reaction is carried out by any known method of oxidation of alcohol functions and in particular those described by D. SWERN et al., Synthesis, 165 (1981) and T.T. TIDWELL, Synthesis, 857 (1990). Of preferably, this oxidation is carried out in dimethyl sulfoxide, in presence of oxalyl chloride and triethylamine, at a temperature between -78 ° C and 0 ° C.
- the deprotection of the hydroxyls is carried out by any known method of deprotection and in particular those described by T.W. GREENE, Protective Groups in Organic Synthesis, J. Wiley-Interscience Publication (1991) or by S.V. LEY et al., Tetrahedron, 46, 4995 (1990).
- T.W. GREENE Protective Groups in Organic Synthesis, J. Wiley-Interscience Publication (1991) or by S.V. LEY et al., Tetrahedron, 46, 4995 (1990).
- trifluoroacetic acid at a temperature between 0 ° C and 100 ° C or tetra (n-butyl) ammonium fluoride, within tetrahydrofuran, at a temperature close to 25 ° C.
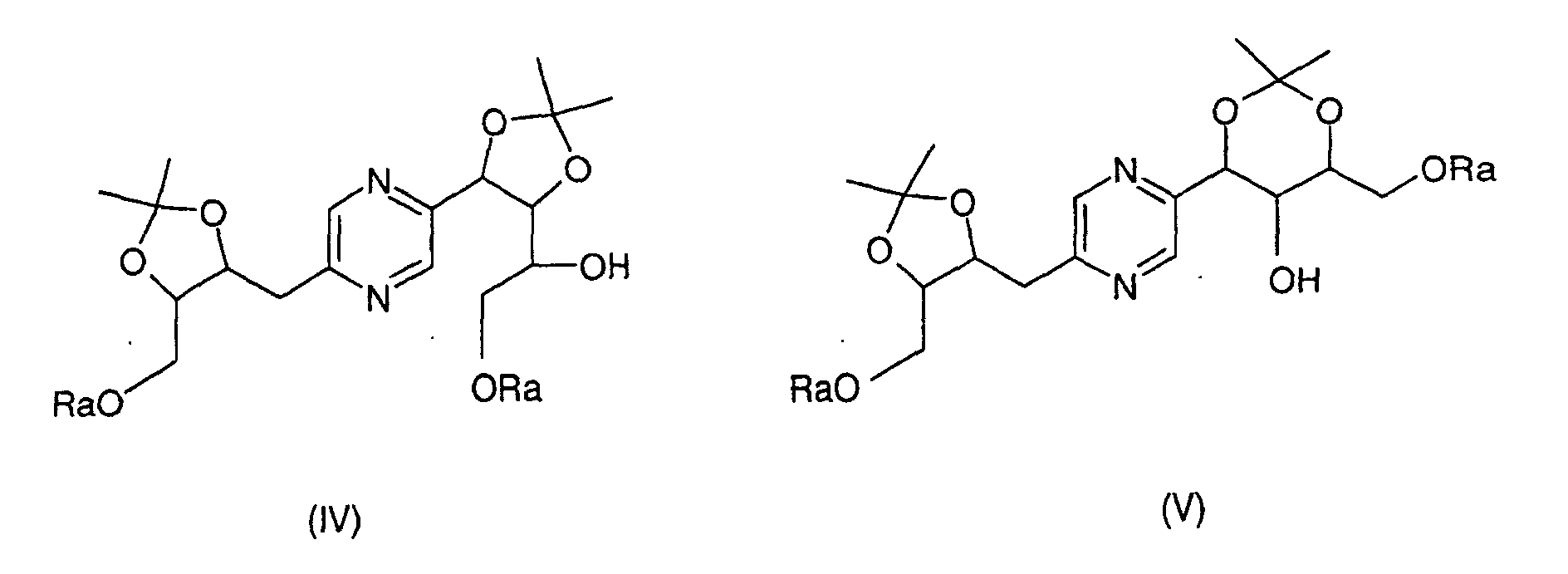
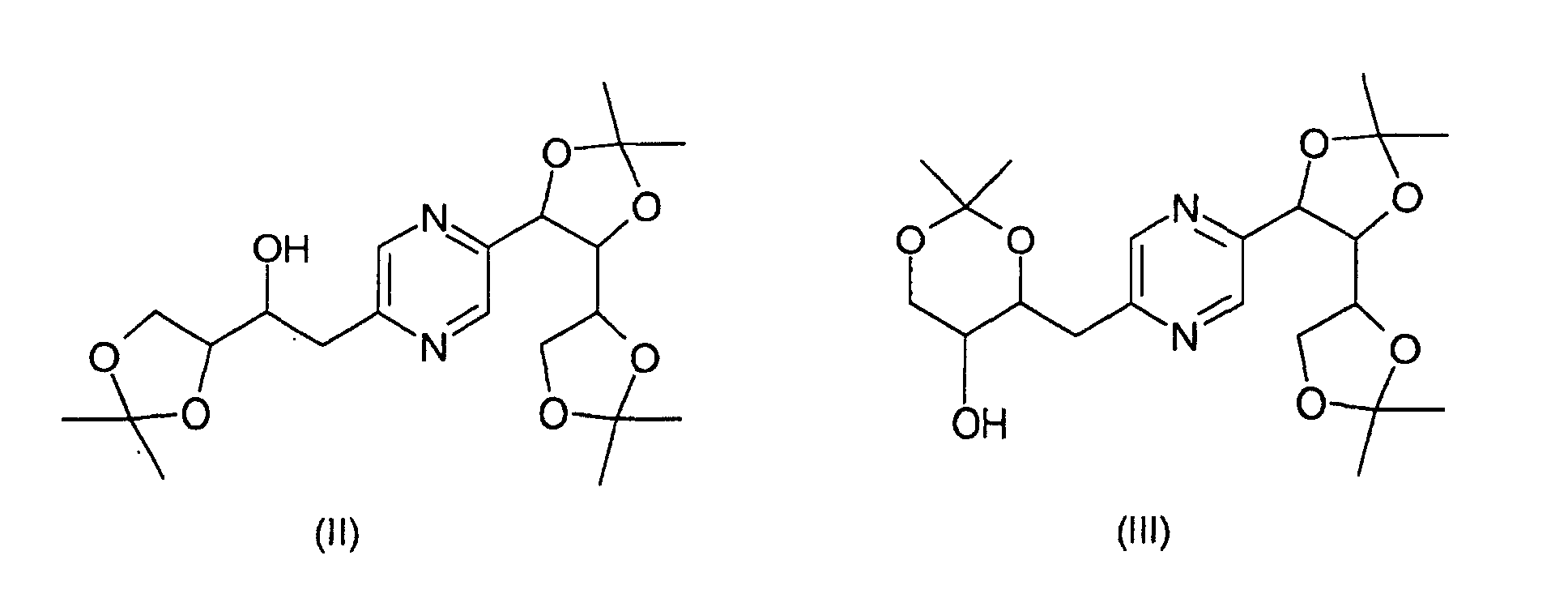
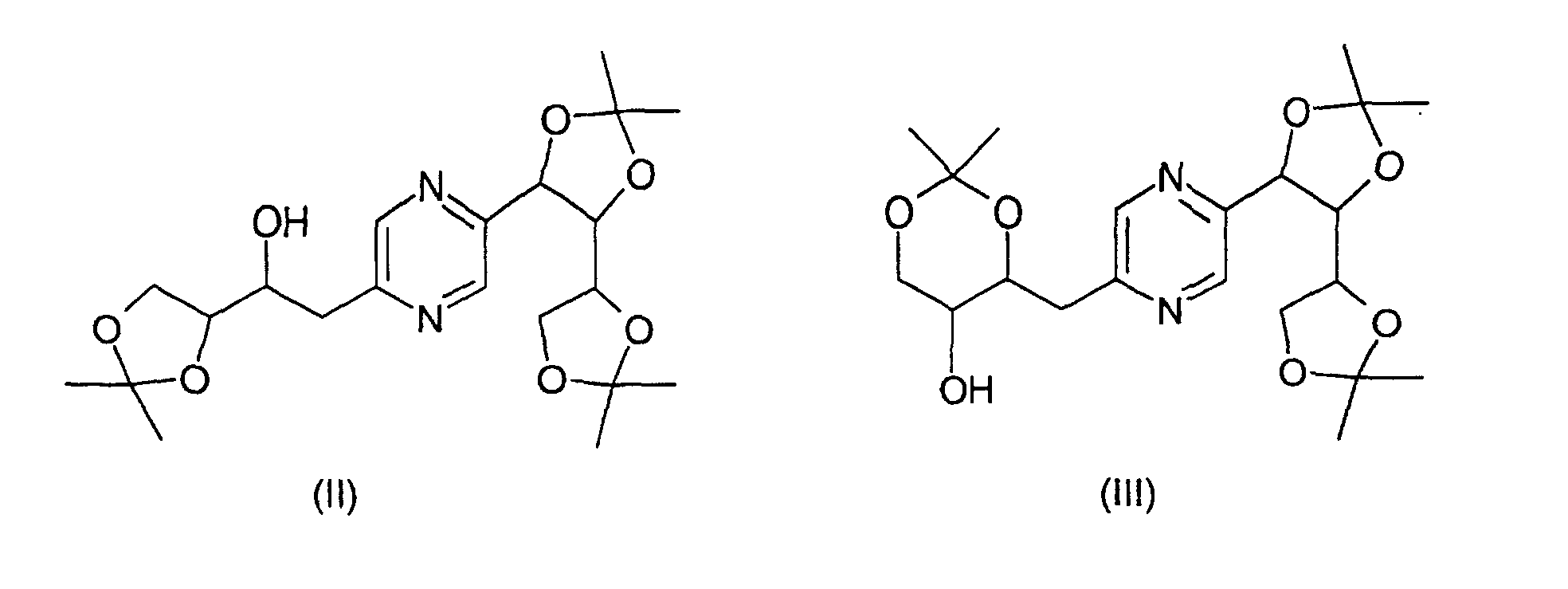
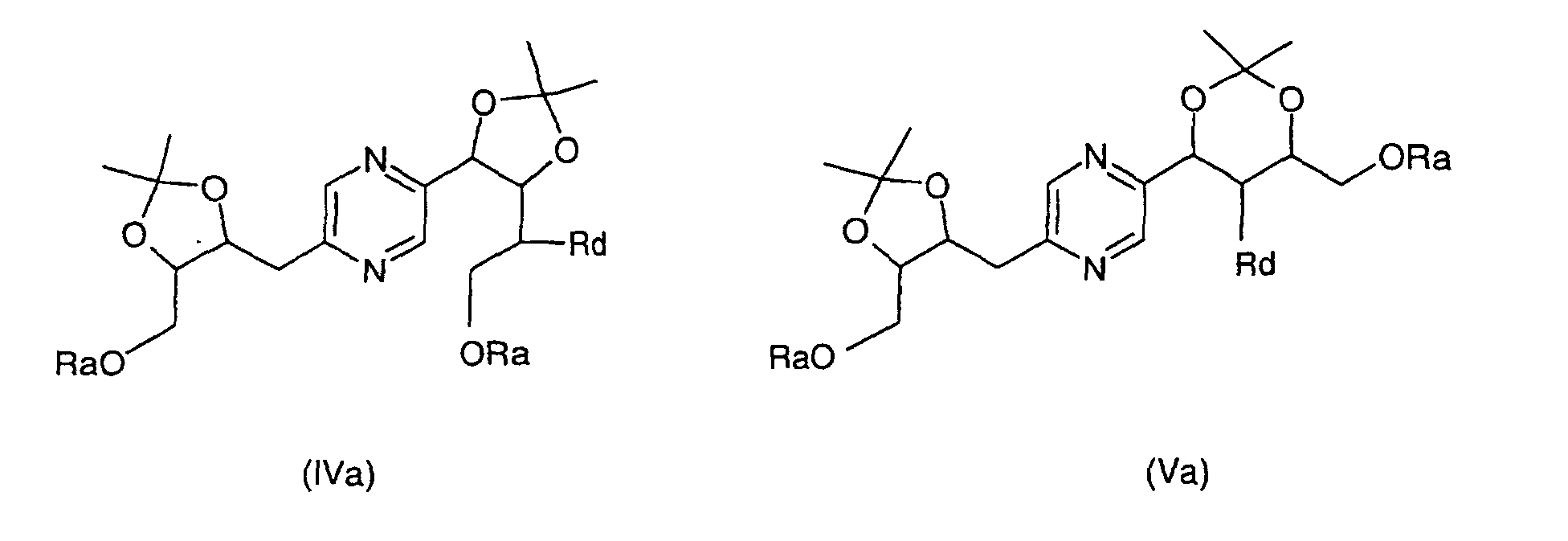
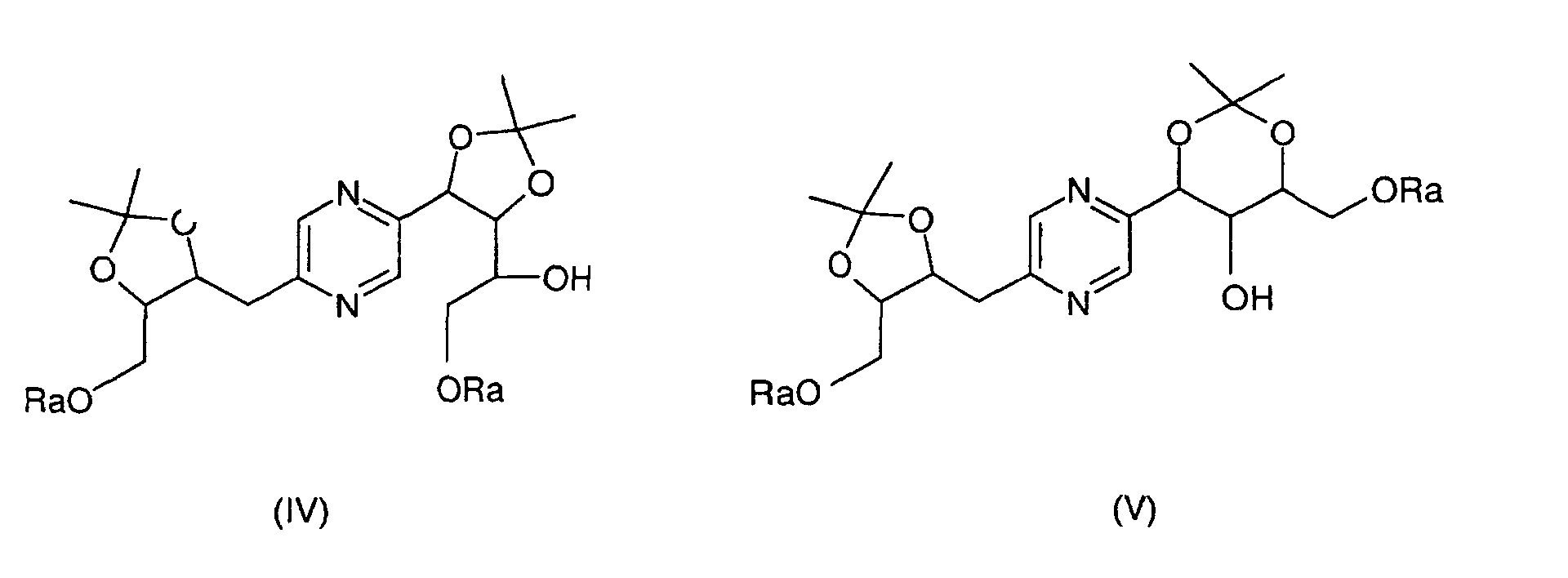
- the derivatives of formulas (II) and (III) can be obtained by the action of 2,2-dimethoxypropane on 2- (1,2,3,4-tetrahydroxybutyl) -5- (2,3,4-trihydroxybutyl) -pyrazine or one of its stereoisomers and separation of derivatives (II) and (III).
- This reaction is generally carried out according to the reaction conditions described by T.W. GREENE, Protective Groups in Organic Synthesis, J. Wiley-Interscience Publication (1991).
- an acid such as paratoluenesulfonic acid
- an inert solvent such as dimethylformamide
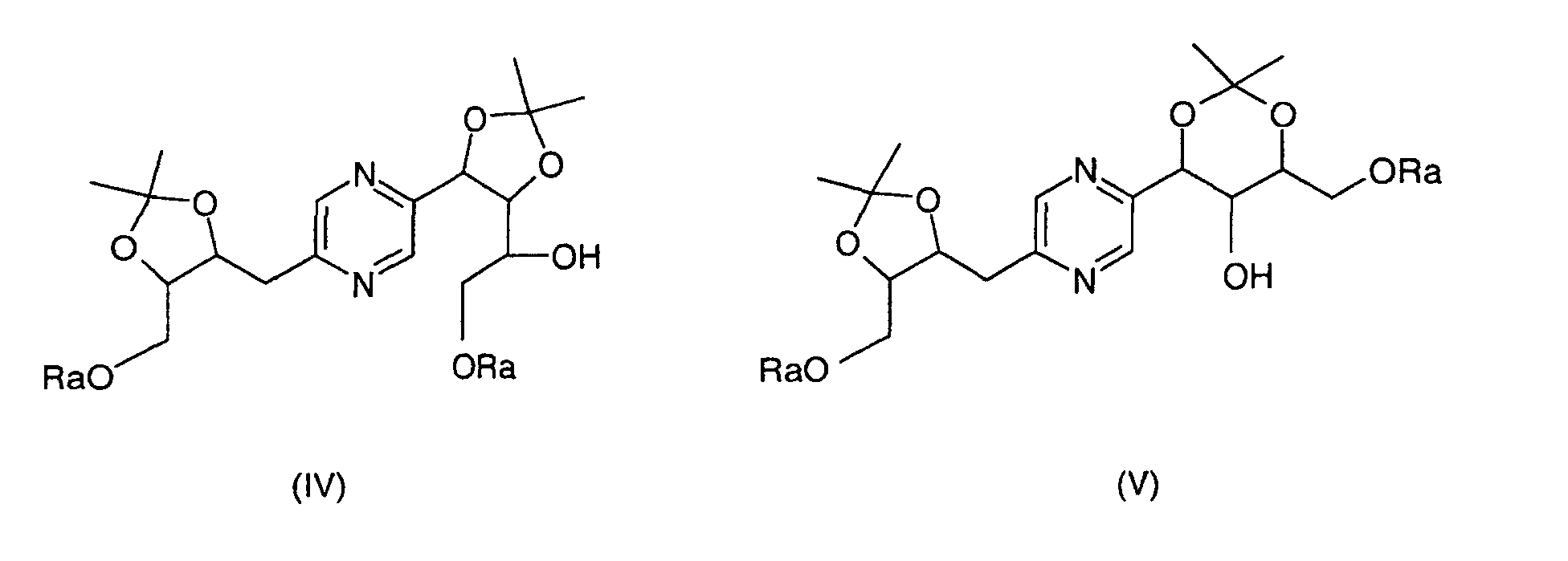
- the derivatives of formula (IV) and (V) can be obtained by the action of 2,2-dimethoxypropane on 2- (1,2,3,4-tetrahydroxybutyl) -5- (2,3,4-trihydroxybutyl) pyrazine whose hydroxyls at the end of the chain have been previously blocked with trialkylsilyl chloride or chloride alkyldiphenylsilyl or dialkyphenylsilyl chloride or a stereoisomer of such a derivative, then separation of the derivatives (IV) and (V).
- This reaction is generally carried out according to the reaction conditions described by T.W. GREENE, Protective Groups in Organic Synthesis, J. Wiley-Interscience Publication (1991).
- an acid such as paratoluenesulfonic acid
- an inert solvent such as dimethylformamide
- the protection of hydroxyls at the end of the chain is generally carried out according to the reaction conditions described by T.W. GREENE, Protective Groups in Organic Synthesis, J. Wiley-Interscience Publication (1991). Of preferably, one operates within the pyridine, at a temperature comprised between 0 ° C and 30 ° C.
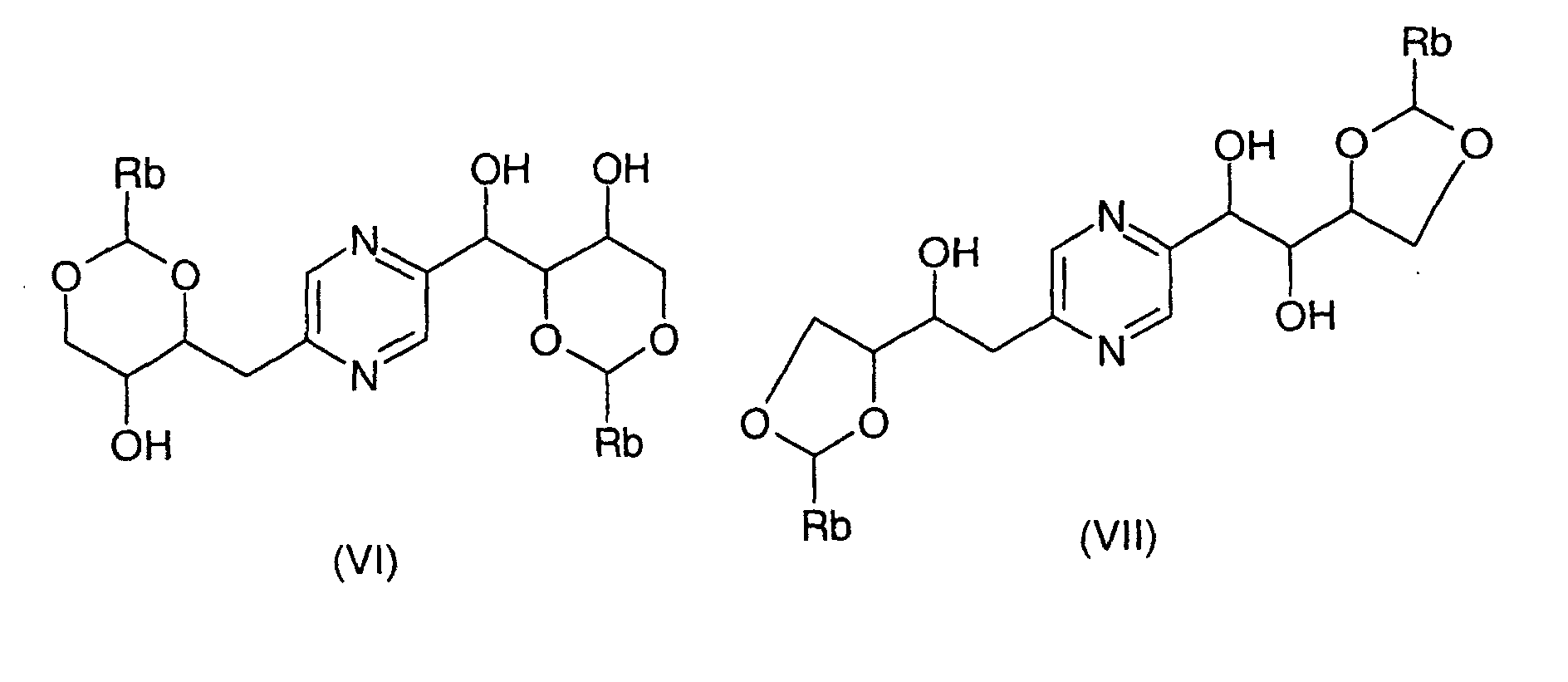
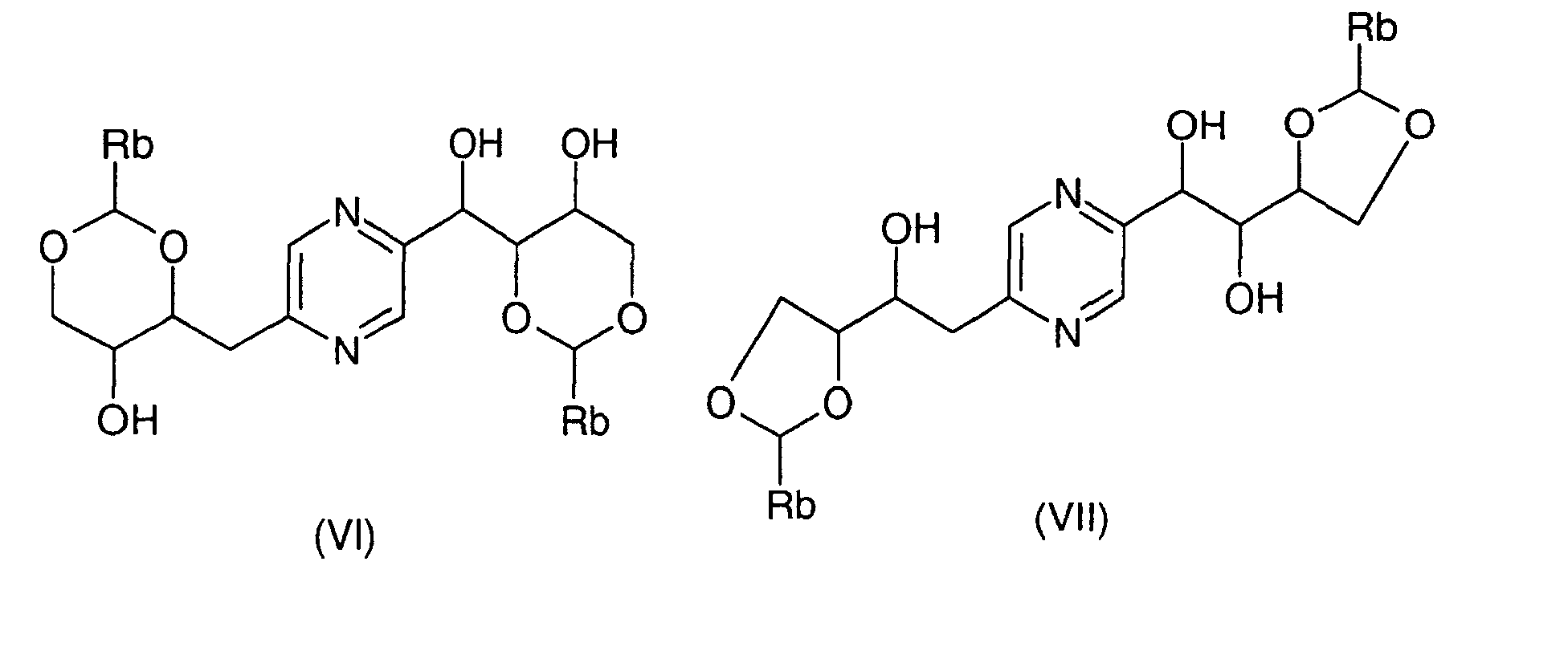
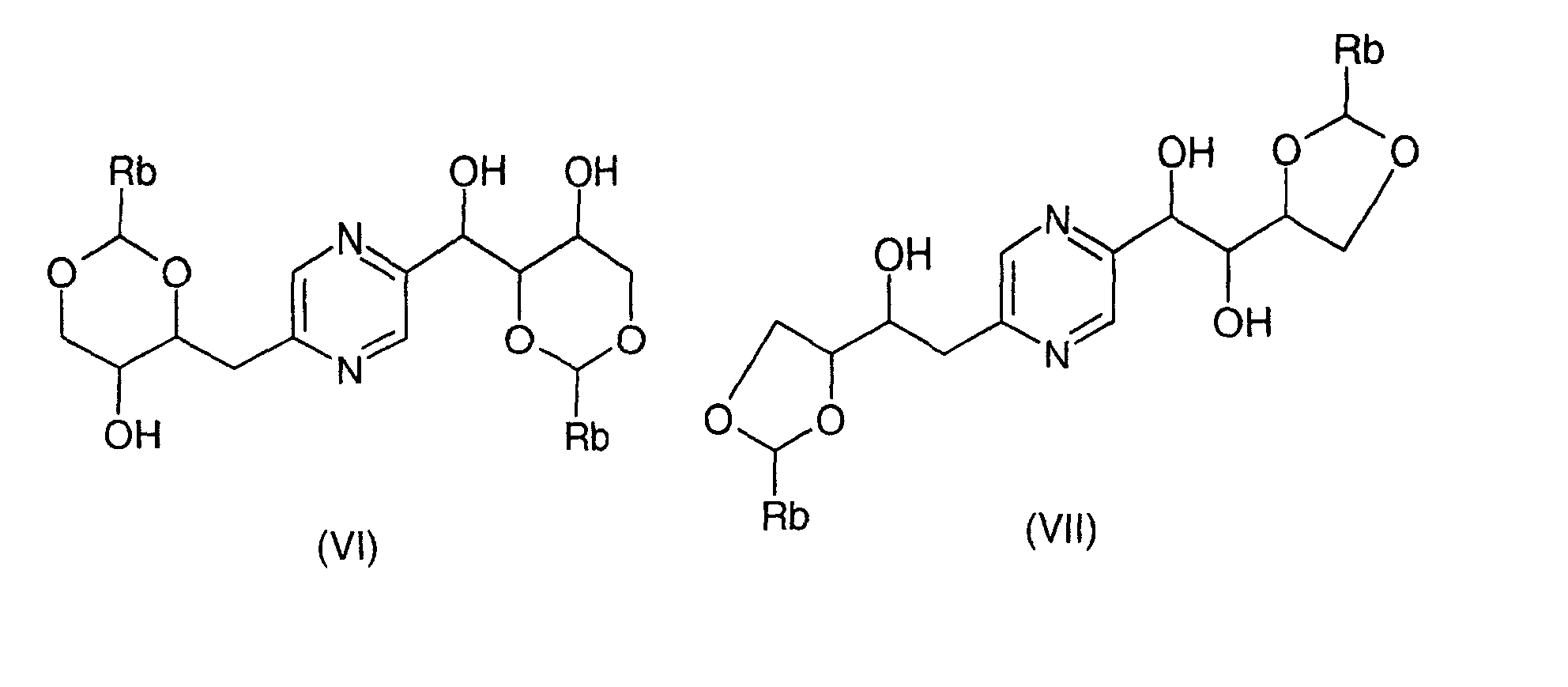
- the derivatives of formula (VI) and (VII) can be obtained by the action of a benzaldehyde whose phenyl is optionally substituted by at least one alkoxy radical or a dialkylacetal derivative of benzaldehyde in which the phenyl is optionally substituted by at least one alkoxy radical (benzaldehyde dimethyl acetal for example) on 2- (1,2,3,4-tetrahydroxybutyl) -5- (2,3,4-trihydroxybutyl) pyrazine or one of his stereoisomers.
- This reaction is generally carried out according to the reaction conditions described by R.S. COLEMAN et al., J. Org. Chem., 57, 3732 (1992). Of preferably, one operates in the presence of an acid such as D-camphosulfonic acid, in an inert solvent such as dimethylformamide, at a temperature between 0 ° C and 100 ° C.
- an acid such as D-camphosulfonic acid
- an inert solvent such as dimethylformamide
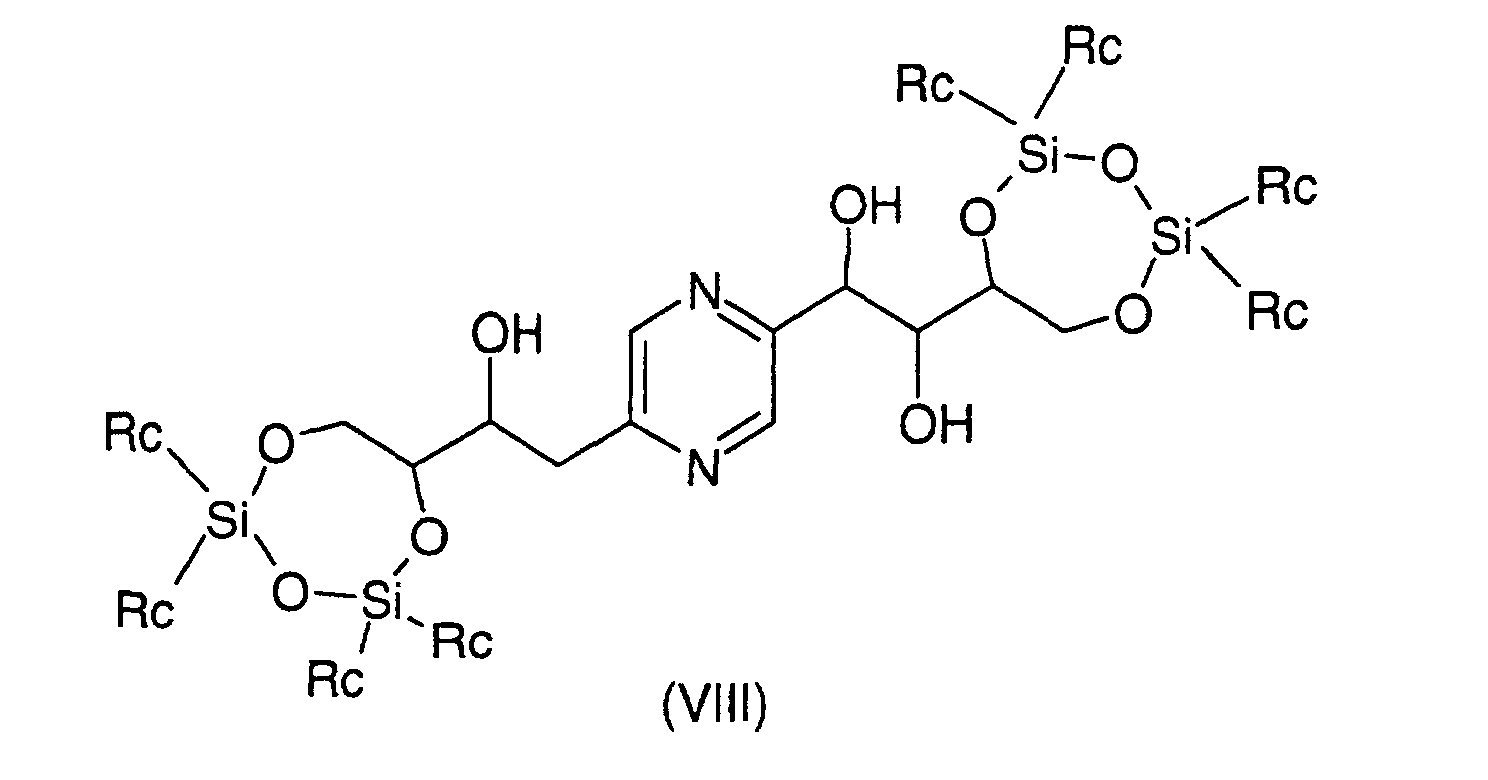
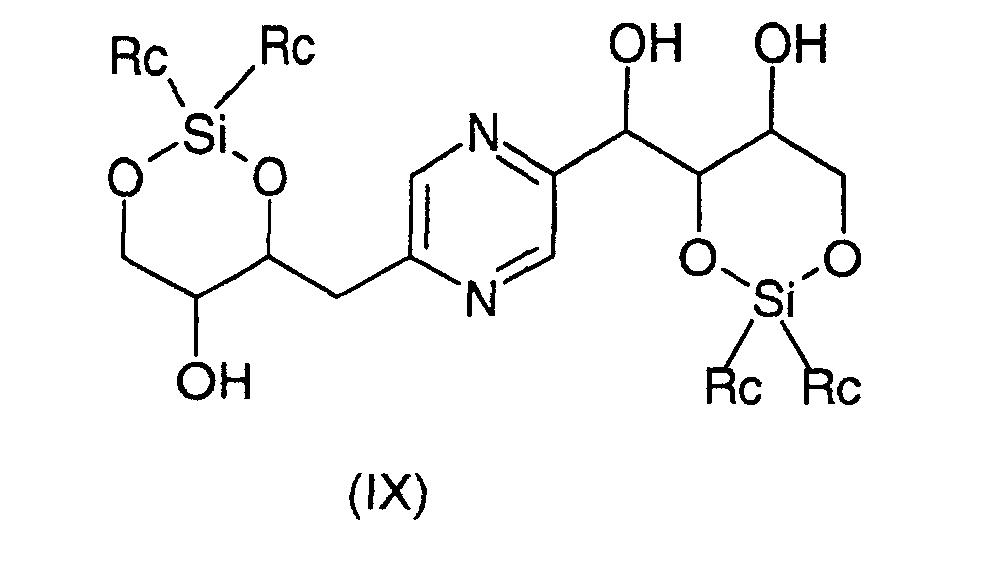
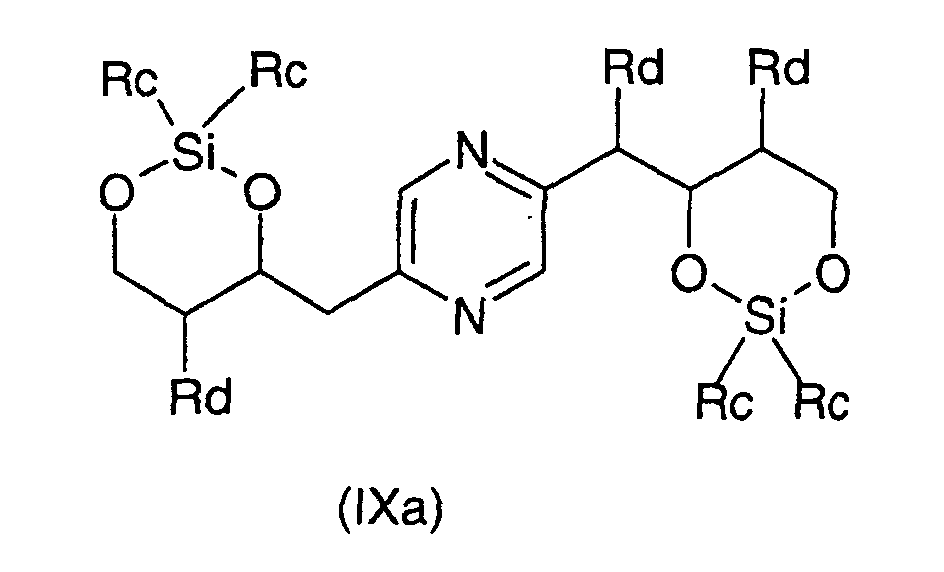
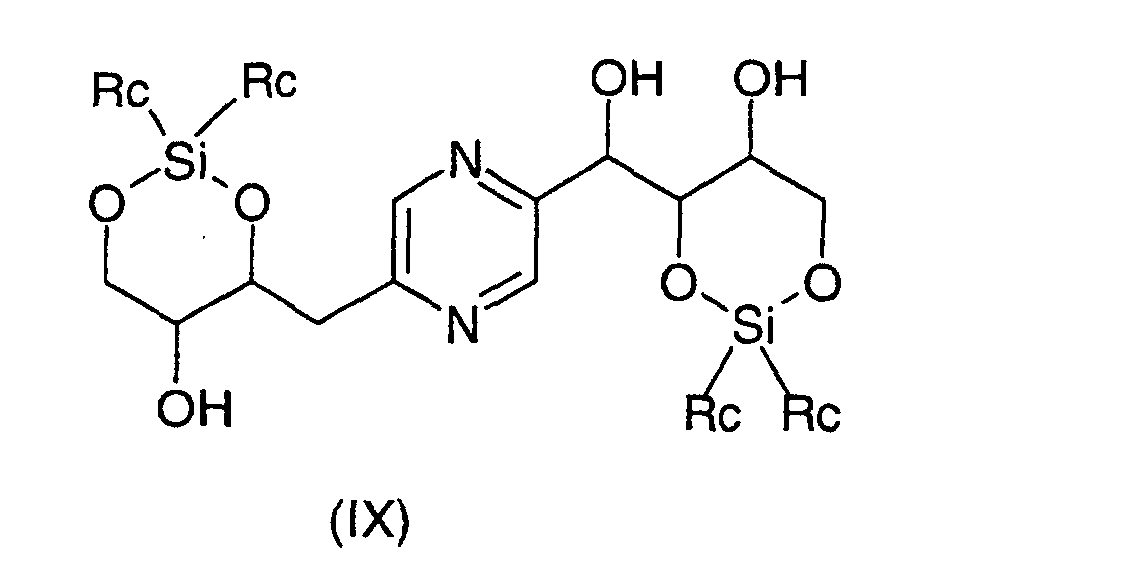
- the derivatives of formula (VIII) and (IX) can be obtained respectively by action of 1,3-dichloro-1,1,3,3-tetraalkyldisiloxane or 1,3-dichloro-1,1,3,3-tetraphenyldisiloxane on the one hand, or dialkylsilyl bis (chloride) or bis (trifluoromethanesulfonate) dialkylsilyl or bis (chloride) diphenylsilyl or diphenylsilyl bis (trifluoromethanesulfonate) else hand, on 2- (1,2,3,4-tetrahydroxybutyl) -5- (2,3,4-trihydroxybutyl) pyrazine or one of its stereoisomers generally according to the reaction conditions described by T.W.
- 2- (1,2,3,4-tetrahydroxybutyl) -5- (2,3,4-trihydroxybutyl) pyrazine and its stereoisomers can be obtained either from one or two amino aldoses OHC-CH (NH 2 ) - (CHOH) 3 -CH 2 OH or one of its stereoisomers, in an acid medium and more particularly in acetic acid medium and preferably operating at a temperature between 15 ° C and 100 ° C; either from one or two ketoses HOCH 2 -CO- (CHOH) 3 -CH 2 OH or one of its stereoisomers, by the action of ammonium formate and preferably operating at a temperature between 15 ° C and 100 ° C, and preferably in an aqueous medium.
- R 9 and R 10 each represent a radical -CH 2 OH and either R 3 represents a methylene radical
- R 4 represents a radical -CHOH- and one of the radicals R 1 , R 2 , R 5 and R 6 represent a methylene radical and the others each represent a radical -CHOH-, that is R 1 and R 6 each represent a radical -CHOH-
- R 3 represents a methylene radical and R 2 , R 4 and R 5 are identical and each represents a methylene radical
- R 2 and R 5 each represent a radical -CHOH- and R 1 , R 3 , R 4 and R 6 each represent a methylene radical
- Ra represents a trialkylsilyl, alkyldiphenylsily
- an inert solvent such than a chlorinated solvent (dichloromethane for example), an ether (diethyl ether, tetrahydrofuran, dioxane for example), in the presence of an acceptor acid such as an organic base such as pyridine or 4-dimethylaminopyridine, at a temperature in the region of 20 ° C and, for the reduction, operation is carried out using tributyltin hydride and azobis (2-methylpropionitrile), within an inert solvent such as an aromatic solvent (benzene, toluene for example), at a temperature between 80 ° C and 110 ° C.
- an inert solvent such as a chlorinated solvent (dichloromethane for example), an ether (diethyl ether, tetrahydrofuran, dioxane for example), in the presence of an acceptor acid such as an organic base such as pyridine or 4-dimethylaminopyridine, at a temperature in the region of
- R 1 , R 2 , R 4 , R 5 and R 6 each represent a radical -CHOH-, R 3 represents a methylene radical, R 9 represents a radical -CH 2 F or -CH 2 OH, R 10 represents a radical -CH 2 F or -CH 2 OH but R 9 and R 10 not both being a radical -CH 2 OH are obtained as a mixture with the compounds prepared from the intermediate (IV).
- This fluorination is generally carried out according to the operating conditions described by W.J. MIDDLETON, J. Org. Chem., 40, 574 (1975).
- the fluorination is carried out by means of a trifluoride dialkylaminosulfide (eg diethylaminosulfide trifluoride), within an inert solvent such as a chlorinated solvent (dichloromethane for example), or an ether (tetrahydrofuran for example), at a temperature comprised between -78 ° C and 20 ° C. Deprotection takes place as mentioned previously.
- R 9 and R 10 each represent a radical -CH 2 OH and either R 3 represents a methylene radical
- R 4 represents a radical -CHOH- and one of the radicals R 1 , R 2 , R 5 and R 6 represent a radical -CH (NHR 7 ) - and the others each represent a radical -CHOH-, that is R 1 and R 6 each represent a radical -CHOH-
- R 3 represents a methylene radical and R 2
- R 4 and R 5 are identical and each represents a radical -CH (NHR 7 ) -
- R 2 and R 5 each represent a radical -CHOH-
- R 3 represents a methylene radical and R 1 , R 4 and R 6 are identical and each represents a radical -CH (NHR 7 ) -
- Ra represents a trialkylsilyl, alkyldiphenylsilyl or dialkylphenylsily
- the reduction is generally carried out according to the reaction conditions described by R.C. LAROCK, Comprehensive Organic Transformations, VCH Publication (1989).
- a catalyst such as palladium
- an inert solvent such than an aliphatic alcohol 1-4C (methanol for example)
- an alkali metal hydride alkali metal borohydride such as sodium borohydride
- lithium aluminum hydride in an inert solvent such as an ether (tetrahydrofuran, diethyl ether for example), at a temperature included between -78 ° C and 100 ° C.
- the reaction with the derivative HalR 7 is generally carried out according to the reaction conditions described by RC LAROCK, Comprehensive Organic Transformations, VCH Publication (1989).
- the operation is carried out in the presence of an organic base such as an amine (trialkylamine such as triethylamine, pyridine) or an organometallic base such as an alkali metal dialkylamide (sodium, lithium for example) or an alkali metal hydride (sodium hydride for example) or a mineral base such as an alkali metal hydroxide (sodium hydroxide, potassium hydroxide for example), in an inert solvent such as an ether (diethyl ether, tetrahydrofuran, dioxane for example), an aliphatic alcohol 1-4C (methanol, ethanol for example), a chlorinated solvent (dichloromethane for example), dimethylformamide or dimethylsulfoxide, at a temperature between 0 ° C and the boiling temperature of the reaction medium.
- the deprotection is carried out as mentioned previously.
- This reaction is generally carried out according to the operating conditions described by A.C. RICHARDSON, Methods Carbohydr. Chem., 6, 218 (1972).
- the operation is carried out in an inert solvent such as dimethylformamide, at a temperature between 0 ° C and 100 ° C.
- This reaction is generally carried out according to the operating conditions described by A.C. RICHARDSON, Methods Carbohydr. Chem., 6, 218, (1972) and H. PAULSEN, Liebigs Ann. Chem., 735 (1992).
- an inert solvent such as a chlorinated solvent (dichloromethane by example)
- an organic base such as pyridine
- R 9 and R 10 each represent a radical -CH 2 OH and either R 3 represents a methylene radical
- R 4 represents a radical -CHOH- and one of the radicals R 1 , R 2 , R 5 and R 6 represent a radical -CH (OR 8 ) and the others each represent a radical -CHOH-
- ie R 1 and R 6 each represent a radical -CHOH-
- R 3 represents a methylene radical and R 2 , R 4 and R 5 are identical and each represents a radical -CH (OR 8 ), or
- R 2 and R 5 each represent a radical -CHOH-
- R 3 represents a methylene radical and R 1 , R 4 and R 6 are identical and each represent a radical -CH (OR 8 )
- R 3 represents a methylene radical and R 1 , R 4 and R 6 are identical and each represent a radical -CH (OR 8 )
- This reaction is generally carried out according to the reaction conditions described R.C. LAROCK, Comprehensive Organic Transformations, VCH Publications (1989).
- a base organic such as an amine (trialkylamine such as triethylamine, pyridine), an organometallic base such as an alkali metal dialkylamide (lithium diisopropylamide, for example) or an alkali metal hydride (sodium hydride for example) or a mineral base (metal hydroxide alkaline (e.g.
- an inert solvent such as ether (tetrahydrofuran, diethyl ether for example), an aliphatic alcohol (methanol, ethanol for example), a chlorinated solvent (dichloromethane by example), dimethylformamide or dimethylsulfoxide, at a temperature between 0 ° C and the boiling temperature of the reaction medium.
- the deprotection is carried out as mentioned previously.
- This reaction is generally carried out according to the reaction conditions described R.C. LAROCK, Comprehensive Organic Transformations, VCH Publications (1989).
- a base organic such as an amine (trialkylamine such as triethylamine or 1,8-diazabicyclo [5.4.0] undéc-7-ene), an organometallic base (alcoholate of alkali metal such as sodium ethanolate) or a dialkylamide of a metal alkaline or alkaline earth (for example lithium diisopropylamide) or a mineral base such as an alkali metal hydroxide (sodium hydroxide or potassium for example), in an inert solvent such as an aliphatic alcohol 1-4C (methanol, ethanol for example), an ether (diethyl ether, tetrahydrofuran for example), a chlorinated solvent (dichloromethane by example) or dimethylformamide, at a temperature between 0 ° C and the boiling point of the reaction medium
- a base organic
- This reaction can also be carried out according to the reaction conditions described by O. MITSUNOBU, Synthesis, p1 (1981).
- an inert solvent such as an ether (diethyl ether, tetrahydrofuran for example), in the presence of trialkylphosphine (triphenylphosphine for example) and dialkyl azodicarboxylate (diethylazocarboxylate for example), at a temperature between 0 ° C and the boiling point of the reaction medium.
- stereoisomers of the compounds of formula (I) are obtained from corresponding stereoisomers of the various intermediates (II), (III), (IV), (V), (VI), (VII), (VIII) or (IX).
- protective groups for the functions amino, hydroxy and carboxy are those that allow to be eliminated without touching the rest of the molecule.
- protecting groups for the amino function we Mention may be made of tert-butyl or methyl carbamates which may be regenerated using iodotrimethylsilane.
- groups protectors of the hydroxy function mention may be made of trialkylsilyl (triethylsilyl for example), benzyl.
- esters methoxymethylester, tetrahydropyranylester, benzylester for example
- oxazoles 2-alkyl-1,3-oxazolines.
- other protecting groups which can be used in these processes are also described by W. GREENE et al., Protective Groups in Organic Synthesis, second edition, 1991, Jonh Wiley & Sons and P.J. KOCIENSKI, Protecting groups, editor Thieme Verlag (1994).
- reaction mixtures obtained by the various processes described above are treated according to conventional physical methods (evaporation, extraction, distillation, chromatography, crystallization by example) or chemical (eg salt formation).
- the compounds of formula (I) can optionally be transformed into addition salts with a mineral or organic acid by the action of such an acid in an organic solvent such as an alcohol, a ketone, an ether or a chlorinated solvent.
- organic solvent such as an alcohol, a ketone, an ether or a chlorinated solvent.
- the compounds of formula (I) comprising an acid residue can optionally be transformed into metal salts or addition salts with bases nitrogenous according to methods known per se.
- These salts can be obtained by the action of a metal base (alkaline or alkaline earth by example), ammonia, an amine or a salt of an amine on a compound of formula (I), in a solvent.
- the salt formed is separated by methods usual.
- salts may be cited addition salts with mineral or organic acids (such as acetate, propionate, succinate, benzoate, fumarate, maleate, oxalate, methanesulfonate, isethionate, theophyllinacetate, salicylate, methylene-bis- ⁇ -oxynaphtoate, hydrochloride, sulfate, nitrate and phosphate), the salts with alkali metals (sodium, potassium, lithium) or with alkaline earth metals (calcium, magnesium), ammonium salt, nitrogen base salts (ethanolamine, trimethylamine, methylamine, benzylamine, N-benzyl- ⁇ -phenethylamine, choline, arginine, leucine, lysine, N-methyl glucamine).
- mineral or organic acids such as acetate, propionate, succinate, benzoate, fumarate, maleate, oxalate, methanesulfonate, ise
- the oil obtained is taken up in 2 cm 3 of absolute ethanol and placed in an ice bath for 2 hours.
- the precipitate obtained is filtered through a sintered glass, drained and then dried in a desiccator under reduced pressure (2.7 kPa) at a temperature of 25 ° C.
- a beige solid is obtained which is recrystallized from a water / absolute ethanol mixture (1: 8 by volume).
- the crystals are filtered on sintered glass, washed with 0.2 cm 3 of absolute ethanol, drained and then dried under reduced pressure (2.7 kPa) at a temperature of 40 ° C.
- the reaction mixture is heated to a temperature of 80 ° C for 45 minutes and then to reflux at a temperature of about 110 ° C for 70 hours. After filtration through a paper filter, the mixture is concentrated under reduced pressure (2.7 kPa) at a temperature of 40 ° C.
- the residual oil is chromatographed on a silica column (0.020-0.045 mm) eluted with an ethyl acetate / cyclohexane mixture (1: 4 by volume) at a pressure of about 1.5 ⁇ 10 5 Pa.
- the fractions containing the expected product are combined and concentrated under reduced pressure (2.7 kPa) at a temperature of 40 ° C.
- the reaction mixture is stirred at a temperature of approximately 25 ° C for 1 day, then it is diluted with a mixture of 20 cm 3 of water and 20 cm 3 of dichloromethane. After decantation, the organic phase is washed 3 times with 20 cm 3 of water, dried over magnesium sulfate, then concentrated under reduced pressure (2.7 kPa) at a temperature in the region of 45 ° C.
- the residual oil is chromatographed on a silica column (0.020-0.045 mm) eluted with an ethyl acetate / cyclohexane mixture (1: 4 by volume) at a pressure of about 1.5 ⁇ 10 5 Pa.
- the reaction mixture is stirred at a temperature of approximately 25 ° C for 20 hours, then 10 cm 3 of 2,2-dimethoxypropane are added and stirring is continued for 3 hours.
- the mixture is then heated at a temperature of 50 ° C for 21 hours.
- concentration under reduced pressure (2.7 kPa) at a temperature of 60 ° C the residual oil is dissolved in 300 cm 3 of dichloromethane and washed twice with 100 cm 3 of an aqueous solution of sodium bicarbonate at 5 % then 2 times with 200 cm 3 of water.
- the organic phase is dried over magnesium sulfate and then concentrated under reduced pressure (2.7 kPa) at a temperature of 40 ° C.
- the oil obtained is chromatographed on a silica column (0.020-0.045 mm) eluted with an ethyl acetate / cyclohexane mixture (1: 1 by volume) at a pressure of approximately 1.5 ⁇ 10 5 Pa.
- the fractions containing the expected products are combined and concentrated under reduced pressure (2.7 kPa) at a temperature of 40 ° C.
- Deoxyfructosazine can be prepared according to the method described by K. Sumoto et al. in Chem. Pharm. Bull., 39 , 792 (1991).
- La 2- (2,2,2 ', 2'-tetramethyl- [4R, 4'R] bi [[1,3] dioxolanyl] -5S-yl) -5- (2- [2,2-dimethyl- [1,3] dioxolan-4S-yl] -E-ethenyl) -pyrazine is prepared according to the following method: To a solution of 0.5 g of 1S- (2,2-Dimethyl- [1,3] dioxolan- 4R-yl) -2- [5- (2,2,2 ', 2'-tetramethyl- [4R, 4'R] bi [[1,3] dioxolanyl] -5S-yl) pyrazin-2-yl ] -ethanol in 9.6 cm 3 of tetrahydrofuran are successively added 132 mg of succinimide and 340 mg of triphenylphosphine.
- a 40% solution of diethyl azodicarboxylate in toluene (0.62 cm 3 ) is then added dropwise and the reaction mixture is stirred at a temperature in the region of 25 ° C for 4 hours at the end of which 396 mg of succinimide 1.02 g of triphenylphosphine and 1.87 cm 3 of 40% diethyl azodicarboxylate solution in toluene are added.
- the reaction mixture is concentrated under reduced pressure (2.7 kPa) at a temperature in the region of 45 ° C.
- the residual brown lacquer is taken up 3 times in ether and then reconcentrated under reduced pressure (2.7 kPa) at a temperature of 45 ° C.
- the residual paste is taken up in 4 cm 3 of ethanol and recrystallized.
- the precipitate obtained is filtered through a sintered glass, washed with ethanol, drained and then dried under reduced pressure (2.7 kPa) at a temperature of 40 ° C.
- La 2- (2,2,2 ', 2'-tetramethyl- [4R, 4'R] bi [[1,3] dioxolanyl] -5S-yl) -5- (2S- [2,2-dimethyl- [1,3] dioxolan-4R-yl] -2-methoxyethyl) -pyrazine is prepared according to the following method: To a solution of 1.0 g of 1S- (2,2-Dimethyl- [1,3] dioxolan- 4R-yl) -2- [5- (2,2,2 ', 2'-tetramethyl- [4R, 4'R] bi [[1,3] dioxolanyl] -5S-yl) pyrazin-2-yl ] -ethanol in 5 cm 3 of dimethylformamide is added a suspension of 104 mg of sodium hydride (60% in oil) in 5 cm 3 of dimethylformamide.
- reaction mixture is stirred at a temperature in the region of 25 ° C for 30 minutes, then 0.15 cm 3 of methyl iodide is added. After 48 hours of stirring, the reaction mixture is treated with 25 cm 3 of water and 25 cm 3 of ethyl acetate and then decanted. The aqueous phase is extracted twice with 25 cm 3 of ethyl acetate and the organic extracts are combined, dried over magnesium sulfate, then concentrated under reduced pressure (2.7 kPa) at a temperature in the region of 45 ° C.
- the residual oil is chromatographed on a silica column (0.020-0.045 mm) eluted with an ethanol / n-butanol / aqueous solution of ammonia (8/1/1 by volume). The fractions containing the expected products are combined and concentrated under reduced pressure (2.7 kPa) at a temperature of 50 ° C.
- La 2- (2,2,2 ', 2'-tetramethyl- [4R, 4'R] bi [[1,3] dioxolanyl] -5S-yl) -5- (2R- [2,2-dimethyl- [1,3] dioxolan-4R-yl] -2-fluoroethyl) -pyrazine is prepared according to the following method: To a solution of 2.0 g of 1S- (2,2-Dimethyl- [1,3] dioxolan- 4R-yl) -2- [5- (2,2,2 ', 2'-tetramethyl- [4R, 4'R] bi [[1,3] dioxolanyl] -5S-yl) pyrazin-2-yl ] -ethanol in 40 cm 3 of tetrahydrofuran cooled to a temperature of -78 ° C and under nitrogen, are added dropwise 1.25 cm 3 of diethylaminosulfide trifluoride.
- the reaction mixture is allowed to warm to a temperature of about -10 ° C. After 4 hours at this temperature of -10 ° C, the reaction mixture is treated with 60 cm 3 of methanol then stirred for 30 minutes and concentrated under reduced pressure (2.7 kPa) at a temperature in the region of 40 ° C.
- the residual brown oil is chromatographed on a silica column (0.020-0.045 mm) eluted with an ethyl acetate / cyclohexane mixture (1: 3 by volume) The fractions containing the expected products are combined and concentrated under reduced pressure (2 , 7 kPa) at a temperature in the region of 40 ° C.
- the precipitate obtained is filtered through a sintered glass, washed with ethanol, drained and then dried under reduced pressure (2.7 kPa) at a temperature in the region of 40 ° C. 88 mg of 1- [5- (2S, 4-dihydroxy-3R-methoxy-butyl) -pyrazin-2-yl] -butane-1R, 2S, 3R, 4-tetraol are thus isolated in the form of a crystalline powder.
- La 2- (2,2,2 ', 2'-tetramethyl- [4R, 4'R] bi [[1,3] dioxolanyl] -5S-yl) -5 - ([5R-methoxy-2,2- dimethyl- [1,3] dioxan-4S-yl] methyl) -pyrazine is prepared according to the following method: To a suspension of 0.14 g of sodium hydride (60% in oil) in 10 cm 3 of dimethylformamide cooled to a temperature close to 0 ° C.
- the reaction mixture is treated with 25 cm 3 of water and 25 cm 3 of ethyl acetate and then decanted.
- the aqueous phase is extracted twice with 25 cm 3 of ethyl acetate and the organic extracts are combined, dried over magnesium sulphate, then concentrated under reduced pressure (2.7 kPa) at a temperature of 50 ° C.
- the residue obtained is chromatographed on a silica column (0.020-0.045 mm) eluted with an ethyl acetate / cyclohexane mixture (1: 2.3 by volume). The fractions containing the expected product are combined and concentrated under reduced pressure (2.7 kPa) at a temperature of 50 ° C.
- the yellow solution is kept at 50 ° C with stirring for 20 hours, then allowed to cool to room temperature and 60 cm 3 of water are added.
- the mixture is extracted with ethyl acetate (2 times) and washed with 20 cm 3 of a saturated solution of sodium hydrogencarbonate, then dried over magnesium sulfate and filtered.
- the organic phases are concentrated under reduced pressure (2.7 kPa) at a temperature of 40 ° C to give an orange oily residue which is chromatographed on a silica column (0.040-0.063 mm), eluting with a dichloromethane / methanol mixture ( 95/5 by volume).
- the reaction mixture is stirred at a temperature of about 25 ° C for 71 hours.
- concentration under reduced pressure (2.7 kPa) at a temperature of 50 ° C the residual oil is taken up 3 times in 30 cm 3 of pentane and then reconcentrated under the same conditions.
- the residual oil obtained is dissolved in 50 cm 3 of dichloromethane and washed successively 2 times with 30 cm 3 of a 1N aqueous hydrochloric acid solution, 2 times with 30 cm 3 of water, then 2 times with 30 cm 3 of a saturated aqueous solution of sodium chloride.
- the organic phase is dried over magnesium sulfate and then concentrated under reduced pressure (2.7 kPa) at a temperature of 40 ° C.
- the meringue obtained is chromatographed on a silica column (0.020-0.045 mm) eluted with an ethyl acetate / cyclohexane mixture (1: 1 by volume) at a pressure of approximately 1.5 ⁇ 10 5 Pa.
- the fractions containing the product expected are combined and concentrated under reduced pressure (2.7 kPa) at a temperature of 40 ° C.
- the meringue obtained is chromatographed on a silica column (0.020-0.045 mm) eluted with an ethyl acetate / cyclohexane mixture (1: 1 by volume) at a pressure of approximately 1.5 ⁇ 10 5 Pa.
- 2-fluoro-1S- [5R- (5 - ⁇ [5R- (hydroxymethyl) -2,2-dimethyl- [1,3] dioxolan-4S-yl] methyl ⁇ -pyrazin-2-yl) -2, 2-dimethyl- [1,3] dioxolan-4R-yl] -ethanol is prepared according to the following method: To a solution of 210 mg of 2-fluoro-1S- [5R- (5 - ⁇ [5R- (tert- butyl-diphenyl-silanyloxymethyl) -2,2-dimethyl- [1,3] dioxolan-4S-yl] methyl ⁇ pyrazin-2-yl) -2,2-dimethyl- [1,3] dioxolan-4R-yl ] -ethanol in 20 cm 3 of tetrahydrofuran under argon, 0.84 cm 3 of a 1.0 M solution of tetra (n-butyl) ammonium fluoride in
- the reaction mixture is stirred at a temperature in the region of 25 ° C for 1.5 hours.
- concentration under reduced pressure (2.7 kPa) at a temperature in the region of 40 ° C the residual amber oil is chromatographed on a silica column (0.040-0.063 mm) eluted with an ethyl acetate / cyclohexane 9 mixture: 1 by volume at a pressure of approximately 1.5 ⁇ 10 5 Pa.
- the fractions containing the expected product are combined and concentrated under reduced pressure (2.7 kPa) at a temperature of 40 ° C.
- the reaction mixture is stirred at a temperature of -45 ° C for 2 hours, then it is treated dropwise with 0.2 cm 3 of methanol. It is then allowed to warm to a temperature of 25 ° C and then concentrated under reduced pressure (2.7 kPa) at a temperature of 40 ° C.
- the residual brown oil is dissolved in 50 cm 3 of dichloromethane and extracted 3 times with 5 cm 3 of water. The organic phase is dried over magnesium sulfate and then concentrated under reduced pressure (2.7 kPa) at a temperature of 40 ° C.
- the residual brown oil is chromatographed on a silica column (0.020-0.045 mm) eluted with a ethyl acetate / cyclohexane mixture 1: 9 by volume at a pressure of about 1.5 ⁇ 10 5 Pa.
- the fractions containing the product expected are combined and concentrated under reduced pressure (2.7 kPa) at a temperature of 40 ° C. 0.17 g of 2-fluoro-1S- [5R- (5 - ⁇ [5R- (tert-butyl-diphenyl-silanyloxymethyl) -2,2-dimethyl- [1,3] dioxolan-4S-yl is thus obtained.
- the reaction mixture is stirred at a temperature of 60 ° C for 42 hours. After concentration under reduced pressure (2.7 kPa) at a temperature of 60 ° C, the residual oil is dissolved in 60 cm 3 of dichloromethane and washed twice with 20 cm 3 of an aqueous solution of sodium bicarbonate at 5 % then 2 times with 20 cm 3 of water. The organic phase is dried over magnesium sulfate and then concentrated under reduced pressure (2.7 kPa) at a temperature of 40 ° C.
- the oil obtained is chromatographed on a silica column (0.020-0.045 mm) eluted with an ethyl acetate / cyclohexane mixture (1: 9 by volume) at a pressure of about 1.5 ⁇ 10 5 Pa.
- the fractions containing the expected products are combined and concentrated under reduced pressure (2.7 kPa) at a temperature of 40 ° C.
- the reaction mixture is stirred at a temperature of approximately 25 ° C for 68 hours, then 0.5 cm 3 of terbutyldiphenylchlorosilane is added and stirring is continued for 8 days.
- concentration under reduced pressure (2.7 kPa) at a temperature of 60 ° C the residual oil is dissolved in 100 cm 3 of dichloromethane and washed 3 times with 30 cm 3 of water.
- the organic phase is dried over magnesium sulfate and then concentrated under reduced pressure (2.7 kPa) at a temperature of 40 ° C.
- the oil obtained is chromatographed on a silica column (0.020-0.045 mm) eluted with a methanol / dichloromethane mixture (1:49 by volume) at a pressure of approximately 1.5 ⁇ 10 5 Pa.
- the fractions containing the expected product are combined and concentrated under reduced pressure (2.7 kPa) at a temperature of 40 ° C.
- the compounds of formula (I) have pharmacological properties interesting. They are hypoglycemic agents.
- the hypoglycaemic activity of the compounds of formula (I) was determined on hyperglycemic response to oral glucose administration in normoglycemic mice, according to the following protocol: Mice Swiss albinos weighing between 22 and 26 g are left on an empty stomach for 2 hours. At the end of this period, blood sugar is measured and, immediately after, a dose of glucose (2 g / kg) is administered orally. Thirty minutes later, blood sugar is measured again. The responding mice with hyperglycemia higher than 170 mg / dl are selected and used for detecting the hypoglycaemic activity of the compounds according to the invention.
- mice thus chosen are divided into groups of at least 10 animals. Separate groups receive a solution of 3 to 50 mg / kg of the test product in a vehicle such as water or a mixture of methylcellulose / tween and water or from the vehicle once a day by gastric tubing.
- the treatment lasts 4 days.
- the 4 th day after the last treatment the animals receive a dose of glucose (2 g / kg) and the glycaemia is measured 20 to 40 minutes later.
- the percentage inhibition of the hyperglycemic response to glucose administration is calculated relative to the response measured in the group treated by the vehicle.
- the compounds according to the invention have a percentage blood glucose inhibition greater than or equal to 10%.
- the compounds of general formula (I) according to the invention have a low toxicity.
- Their LD50 is greater than 2000 mg / kg orally in mice.
- these products are useful in the prevention and treatment of diabetes and in particular type II diabetes (diabetes IDN), obese diabetes, fiftieth diabetes, metaplethoric diabetes, diabetes in the elderly and mild diabetes. They can be used in complement of insulin therapy in insulin dependent diabetes where they allow to gradually decrease the dose of insulin, unstable diabetes, insulin-resistant diabetes, in addition to sulphonylureas when these do not determine a sufficient drop in blood sugar. These products can also be used in complications diabetes such as hyperlipemia, disorders of lipid metabolism, dyslipemias, obesity.
- Atherosclerotic lesions and their complications coronopathies, myocardial infarction, cardiomyopathies, evolution of these three complications towards left ventricular failure, arteriopathies various, arteritis of the lower limbs with lameness and progression to ulcers and gangrene, cerebrovascular insufficiency and its complications, sexual impotence of vascular origin), diabetic retinopathy and all of its manifestations (increased capillary permeability, dilation and capillary thrombosis, microaneurysms, arteriovenous shunt, venous dilation, punctual and macular hemorrhages, exudates, macular edema, manifestations of proliferative retinopathy: neovessels, proliferative retinitis scars, vitreous hemorrhage, detachment retina), diabetic cataract, diabetic neuropathy in its various forms (peripheral polyneuropathies and its
- the medicaments according to the invention consist of a compound according to the invention or a combination of these products, either pure or in form of a composition in which it is associated with any other pharmaceutical product compatible, may be inert or physiologically active.
- the drugs according to the invention can be used orally, parenterally, rectal or topical.
- compositions for oral administration can be used tablets, pills, powders (gelatin capsules, cachets) or granules.
- the active principle according to the invention is mixed with one or more inert diluents, such as starch, cellulose, sucrose, lactose or silica, under a stream of argon.
- inert diluents such as starch, cellulose, sucrose, lactose or silica
- These compositions can also include substances other than diluents, for example one or more lubricants such as magnesium stearate or talc, a coloring, coating (dragees) or varnish.
- compositions for oral administration pharmaceutical solutions, suspensions, emulsions, syrups and elixirs acceptable containing inert diluents such as water, ethanol, glycerol, vegetable oils or paraffin oil.
- inert diluents such as water, ethanol, glycerol, vegetable oils or paraffin oil.
- These compositions may include substances other than diluents, for example example of wetting, sweetening, thickening, flavoring or stabilizers.
- the sterile compositions for parenteral administration can be of preferably aqueous or non-aqueous solutions, suspensions or emulsions.
- solvent or vehicle water, propylene glycol, polyethylene glycol, vegetable oils, in particular oil olive, injectable organic esters, for example ethyl oleate or other suitable organic solvents.
- These compositions can also contain adjuvants, in particular wetting agents, isotonizing agents, emulsifiers, dispersants and stabilizers. Sterilization can be done in several ways, for example by aseptic filtration, by incorporating the composition of the sterilizing agents, by irradiation or by heating. They can also be prepared in the form of sterile solid compositions which can be dissolved at the time of use in sterile water or any other sterile injectable medium.
- compositions for rectal administration are suppositories or rectal capsules which contain, in addition to the active product, excipients such than cocoa butter, semi-synthetic glycerides or polyethylene glycols.
- compositions for topical administration may, for example, be creams, lotions, eye drops, mouthwashes, nose drops or aerosols.
- the doses depend on the desired effect, the duration of the treatment and the route of administration used; they are generally between 150 mg and 600 mg daily orally for an adult with doses units ranging from 50 mg to 200 mg of active substance.
- the doctor will determine the appropriate dosage dependent on age, weight and all other factors specific to the subject treat.
- the invention also relates to the use of the compounds of general formula (I) for the preparation of pharmaceutical compositions useful for the treatment or prevention of diabetes and complications of diabetes.
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- General Health & Medical Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Life Sciences & Earth Sciences (AREA)
- Pharmacology & Pharmacy (AREA)
- Medicinal Chemistry (AREA)
- Diabetes (AREA)
- Epidemiology (AREA)
- Obesity (AREA)
- Endocrinology (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Hematology (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Engineering & Computer Science (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Emergency Medicine (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Medicinal Preparation (AREA)
- Cosmetics (AREA)
- Agricultural Chemicals And Associated Chemicals (AREA)
- Plural Heterocyclic Compounds (AREA)
- Nitrogen Condensed Heterocyclic Rings (AREA)
- Preparation Of Compounds By Using Micro-Organisms (AREA)
Description
R9 et R10 représentent chacun un radical -CH2OH, et soit
R8 représente un radical alkyle, -alk-COOH ou -alk-OH,
alk représente un radical alkyle,
Ar représente un radical phényle ou phényle substitué par un ou plusieurs substituants choisis parmi un atome d'halogène, un radical alkyle, alcoxy, alcoxycarbonyle, amino, monoalkylamino ou dialkylamino,
Het représente un hétérocycle mono, di ou tricyclique saturé ou insaturé contenant 1 à 9 atomes de carbone et un ou plusieurs hétéroatomes choisis parmi oxygène, soufre et azote.
- par des réactions d'épimérisation par application ou adaptation des méthodes décrites dans Adv. Carbohydr. Chem., 13, 63, (1958) notamment en milieu basique au moyen d'une solution aqueuse diluée de soude (0,03 à 0.05%), à une température comprise entre 20 et 40°C,
- par des réactions d'allongement de chaíne par application ou adaptation des méthodes décrites dans «The Carbohydrates», éditeurs: W. Pigman et D. Horton, Academic Press, New-York, Volume IA, 133 (1972) et notamment en formant la cyanhydrine de l'aldose de départ (par exemple par action du cyanure de sodium en solution aqueuse, à une température comprise entre 10 et 30°C et en présence de soude, à un pH voisin de 9) puis hydrolyse de la fonction nitrile ainsi formée en acide correspondant par application ou adaptation des méthodes décrites dans Organic Synthesis volume I page 436 et volume III page 85 (par exemple à l'aide d'acide chlorhydrique ou d'acide sulfurique concentré, en solution aqueuse, à une température comprise entre 20°C et la température d'ébullition du milieu réactionnel), puis réduction de la fonction acide carboxylique en aldéhyde correspondant par application ou adaptation des méthodes décrites dans J. Am. Chem. Soc. 71, 122 (1949) notamment à l'aide d'un borohydrure d'un métal alcalin (le borohydrure de sodium par exemple), en solution aqueuse à une température comprise entre 20°C et la température d'ébullition du milieu réactionnel,
- par des réactions de racourcissement de chaínes par application ou adaptation des méthodes décrites dans «The Carbohydrates», éditeurs: W. Pigman et D. Horton, Academic Press, New-York, Volume IB, 1980, page 929 ou Chem. Ber., 83, 559 (1950) et notamment en transformant la fonction aldéhyde de l'aldose en hydroxylamine correspondant par application ou adaptation des méthodes décrites dans Organic Synthesis volume II page 314 (par exemple à l'aide de chlorhydrate d'hydroxylamine, en solution aqueuse et en présence d'une base telle que le carbonate de sodium à une température comprise entre 20 et 50°C), puis action du 3,4-dinitro-fluorobenzéne en présence de dioxyde de carbone et d'une base telle le d'hydrogénocarbonate de sodium en solution aqueuse et d'un alcool aliphatique (alcool isopropylique par exemple), à une température comprise entre 50 et 80°C,
- Produit actif 50 mg
- Cellulose 18 mg
- Lactose 55 mg
- Silice colloïdale 1 mg
- Carboxyméthylamidon sodique 10 mg
- Talc 10 mg
- Stéarate de magnésium 1 mg
- Produit actif 50 mg
- Lactose 104 mg
- Cellulose 40 mg
- Polyvidone 10 mg
- Carboxyméthylamidon sodique 22 mg
- Talc 10 mg
- Stéarate de magnésium 2 mg
- Silice colloïdale 2 mg
- Mélange d'hydroxyméthylcellulose, glycérine, oxyde de titane (72-3,5-24,5) q.s.p. 1 comprimé pelliculé terminé à 245 mg
- Produit actif 50 mg
- Acide benzoïque 80 mg
- Alcool benzylique 0,06 ml
- Benzoate de sodium 80 mg
- Ethanol à 95 % 0,4 ml
- Hydroxyde de sodium 24 mg
- Propylène glycol 1,6 ml
- Eau q.s.p. 4 ml
Claims (13)
- Médicaments contenant en tant que principe actif au moins un composé de formule : dans laquelle
R9 et R10 sont identiques et représentent un radical -CH2OH, et soitR7 représente un atome d'hydrogène ou un radical alkyle, -CO-alk, -CO-Ar ou -CO-Het,a) R3 représenté un radical méthylène, R4 représente un radical -CHOH- et l'un des radicaux R1, R2, R5 et R6 représente un radical carbonyle, méthylène, -CHF-, -CH(NHR7)- ou -CH(OR8) et les autres représentent chacun un radical -CHOH-,b) R2 et R5 représentent chacun un radical -CHOH-, R3 représente un radical méthylène et R1, R4 et R6 sont identiques et représentent chacun un radical carbonyle, méthylène, -CHF-, -CH(NHR7)- ou -CH(OR8),c) R1 et R6 représentent chacun un radical -CHOH-, R3 représente un radical méthylène et R2, R4 et R5 sont identiques et représentent chacun un radical carbonyle, méthylène, -CHF-, -CH(NHR7)- ou -CH(OR8),d) R1, R4, R5 et R6 représentent chacun un radical -CHOH- et -R2-R3-représente un radical -CH=CH-,
R8 représente un radical alkyle, -alk-COOH ou -alk-OH,
alk représente un radical alkyle,
Ar représente un radical phényle ou phényle substitué par un ou plusieurs substituants choisis parmi un atome d'halogène, un radical alkyle, alcoxy, alcoxycarbonyle, amino, monoalkylamino ou dialkylamino,
Het représente un hétérocycle mono, di ou tricyclique saturé ou insaturé contenant 1 à 9 atomes de carbone et un ou plusieurs hétéroatomes choisis parmi oxygène, soufre et azote,
les radicaux et portions alkyle et alcoxy contenant 1 à 6 atomes de carbone en chaíne droite ou ramifiée,
ou un de ses stéréoisomères ou pour le composé pour lequel -R2-R3-représente un radical -CH=CH- ses formes cis ou trans ou un de ses sels avec un acide minéral ou organiques pharmaceutiquement acceptable. - Médicaments selon la revendication 1 pour lesquels dans les composés de formule (I) Het représente un hétérocycle choisi parmi les cycles 2-, 3-, ou 4-pyridyle, imidazolyle, thiazolyle et oxazolyle.
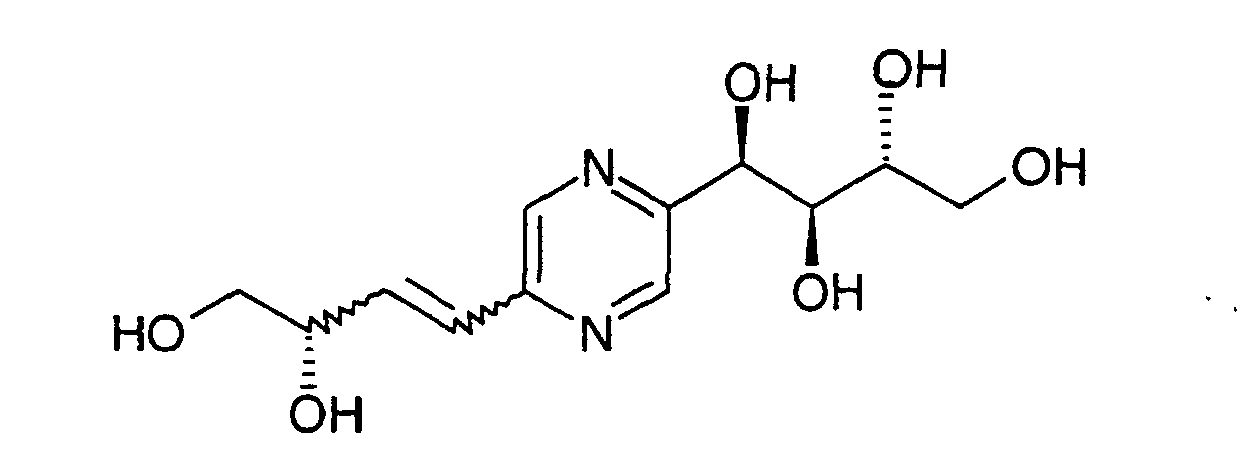
- Médicaments selon la revendication 1 pour lesquels le composé de formule (I) est choisi parmi les composés suivants :et leurs sels avec un acide minéral ou organique pharmaceutiquement acceptable.1-[5-(3R,4-Dihydroxy-2-oxo-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(3S,4-Dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(2S-Fluoro-3R,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(2R-Fluoro-3R,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(2S-Amino-3S,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(2R-Amino-3S,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(2S-(N-Méthyl)amino-3S,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(2R-(N-Méthyl)amino-3S,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(2S-(N-Ethyl)amino-3S,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(2R-(N-Ethyl)amino-3S,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(2S-(N-n-Butyl)amino-3S,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(2R-(N-n-Butyl)amino-3S,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(2S-(N-Benzyl)amino-3S,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(2R-(N-Benzyl)amino-3S,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(2S-(N-Acétyl)amino-3S,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(2R-(N-Acétyl)amino-3S,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(2S-(N-Butanoyl)amino-3S,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(2R-(N-Butanoyl)amino-3S,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(2S-(N-Benzoyl)amino-3S,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(2R-(N-Benzoyl)amino-3S,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(2S-Méthoxy-3R,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(2R-Méthoxy-3R,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(2S-Ethoxy-3R,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(2R-Ethoxy-3R,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(2S-n-Butoxy-3S,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(2R-n-Butoxy-3R,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(2S-(2-Hydroxyéthyl)oxy-3R,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(2R-(2-Hydroxyéthyl)oxy-3R,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(2S-(3-Hydroxy-n-propyl)oxy-3R,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(2R-(3-Hydroxy-n-propyl)oxy-3R,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(2S-(Carboxyméthyl)oxy-3R,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(2R-(Carboxyméthyl)oxy-3R,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(2S-(3-Carboxy-n-propyl)oxy-3R,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(2R-(3-Carboxy-n-propyl)oxy-3R,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(2S,4-Dihydroxy-3-oxo-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(2S,4-Dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(3R-Fluoro-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(3S-Fluoro-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(3R-Amino-2R,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(3S-Amino-2R,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(3R-(N-Méthyl)amino-2R,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(3S-(N-Méthyl)amino-2R,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(3R-(N-Ethyl)amino-2R,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(3S-(N-Ethyl)amino-2R,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(3R-(N-n-Butyl)amino-2R,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(3S-(N-n-Butyl)amino-2R,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(3R-(N-Benzyl)amino-2R,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(3S-(N-Benzyl)amino-2R,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(3R-(N-Acétyl)amino-2R,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(3S-(N-Acétyl)amino-2R,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(3R-(N-Butanoyl)amino-2R,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(3S-(N-Butanoyl)amino-2R,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(3R-(N-Benzoyl)amino-2R,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(3S-(N-Benzoyl)amino-2R,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(3R-Méthoxy-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(3S-Méthoxy-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(3R-Ethoxy-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(3S-Ethoxy-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(3R-n-Butoxy-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(3S-n-Butoxy-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(3R-(2-Hydroxyéthyl)oxy-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(3S-(2-Hydroxyéthyl)oxy-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(3R-(3-Hydroxy-n-propyl)oxy-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(3S-(3-Hydroxy-n-propyl)oxy-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(3R-(Carboxyméthyl)oxy-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(3S-(Carboxyméthyl)oxy-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(3R-(3-Carboxy-n-propyl)oxy-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,1-[5-(3S-(3-Carboxy-n-propyl)oxy-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,4-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-1,3S,4R-trihydroxy-butane-2-one,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2R,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-3R-fluoro-butane-1R,2S,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-3S-fluoro-butane-1R,2S,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-3R-amine-butane-1R,2R,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-3S-amino-butane-1R,2R,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-3R-(N-méthyl)amino-butane-1R,2R,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-3S-(N-méthyl)amino-butane-1R,2R,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-3R-(N-éthyl)amino-butane-1R,2R,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-3S-(N-éthyl)amino-butane-1R,2R,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-3R-(N-n-butyl)amino-butane-1R,2R,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-3S-(N-n-butyl)amino-butane-1R,2R,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-3R-(N-benzyl)amino-butane-1R,2R,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-3S-(N-benzyl)amino-butane-1R,2R,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-3R-(N-acétyl)amino-butane-1R,2R,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-3S-(N-acétyl)amino-butane-1R,2R,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-3R-(N-Butanoyl)amino-butane-1R,2R,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-3S-(N-Butanoyl)amino-butane-1R,2R,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-3R-(N-Benzoyl)amino-butane-1R,2R,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-3S-(N-Benzoyl)amino-butane-1R,2R,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-3R-méthoxy-butane-1R,2S,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-3S-méthoxy-butane-1R,2S,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-3R-éthoxy-butane-1R,2S,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-3S-éthoxy-butane-1R,2S,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-3R-n-butoxy-butane-1R,2S,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-3S-n-butoxy-butane-1R,2S,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-3R-(2-hydroxyéthyl)oxy-butane-1R,2S,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-3S-(2-hydroxyéthyl)oxy-butane-1R,2S,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-3R-(3-hydroxy-n-propyl)oxy-butane-1R,2S,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-3S-(3-hydroxy-n-propyl)oxy-butane-1R,2S,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-3R-(carboxyméthyl)oxy-butane-1R,2S,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-3S-(carboxyméthyl)oxy-butane-1R,2S,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-3R-(3-carboxy-n-propyl)oxy-butane-1R,2S,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-3S-(3-carboxy-n-propyl)oxy-butane-1R,2S,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-1R,3R,4-trihydroxy-butane-2-one,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-butane-1R,3R,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-2R-fluoro-butane-1R,3R,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-2S-fluoro-butane-1R,3R,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-2R-amino-butane-1S,3S,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-2S-amino-butane-1S,3S,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-2R-(N-méthyl)amino-butane-1S,3S,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl)-2S-(N-méthyl)amino-butane-1S,3S,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-2R-(N-éthyl)amino-butane-1S,3S,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-2S-(N-éthyl)amino-butane-1S,3S,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-2R-(N-n-butyl)amino-butane-1S,3S,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-2S-(N-n-butyl)amino-butane-1S,3S,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-2R-(N-benzyl)amino-butane-1S,3S,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-2S-(N-benzyl)amine-butane-1S,3S,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-2R-(N-acétyl)amino-butane-1S,3S,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-2S-(N-acétyl)amino-butane-1S,3S,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-2R-(N-Butanoyl)amino-butane-1S,3S,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-2S-(N-Butanoyl)amino-butane-1S,3S,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-2R-(N-Benzoyl)amino-butane-1S,3S,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-2S-(N-Benzoyl)amino-butane-1S,3S,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-2R-méthoxy-butane-1R,3R,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-2S-méthoxy-butane-1R,3R,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-2R-éthoxy-butane-1R,3R,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-2S-éthoxy-butane-1R,3R,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-2R-n-butoxy-butane-1R,3R,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-2S-n-butoxy-butane-1R,3R,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-2R-(2-hydroxyéthyl)oxy-butane-1R,3R,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-2S-(2-hydroxyéthyl)oxy-butane-1R,3R,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-2R-(3-hydroxy-n-propyl)oxy-butane-1R,3R,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-2S-(3-hydroxy-n-propyl)oxy-butane-1R,3R,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-2R-(carboxyméthyl)oxy-butane-1R,3R,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-2S-(carboxyméthyl)oxy-butane-1R,3R,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-2R-(3-carboxy-n-propyl)oxy-butane-1R,3R,4-triol,1-[5-(2S,3R,4-Trihydroxy-butyl)-pyrazin-2-yl]-2S-(3-carboxy-n-propyl)oxy-butane-1R,3R,4-triol,1-[5-(2S,4-Dihydroxy-3-oxo-butyl)-pyrazin-2-yl]-2S,4-dihydroxy-butane-1,3-dione,4-[5-(2S,4-Dihydroxy-butyl)-pyrazin-2-yl]-butane-1,3S-diol,4-[5-(3R-Fluoro-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2R,4R-difluoro-butane-1,3S-diol,4-[5-(3S-Fluoro-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2S,4S-difluoro-butane-1,3S-diol,1-[5-(3R-Amino-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2R,4R-diamino-butane-1,3S-diol,1-[5-(3S-Amino-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2S,4S-diamino-butane-1,3S-diol,1-[5-(3R-(N-Méthyl)amino-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2R,4R-di-(N-Méthyl)amino-butane-1,3S-diol,1-[5-(3S-(N-Méthyl)amino-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2S,4S-di-(N-Méthyl)amino-butane-1,3S-diol,1-[5-(3R-(N-Ethyl)amino-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2R,4R-di-(N-éthyl)amino-butane-1,3S-diol,1-[5-(3S-(N-Ethyl)amino-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2S,4S-di-(N-éthyl)amino-butane-1,3S-diol,1-[5-(3R-(N-n-Butyl)amino-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2R,4R-di-(N-n-Butyl)amino-butane-1,3S-diol,1-[5-(3S-(N-n-Butyl)amino-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2S,4S-di-(N-n-Butyl)amino-butane-1,3S-diol,1-[5-(3R-(N-Benzyl)amino-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2R,4R-di-(N-Benzyl)amino-butane-1,3S-diol,1-[5-(3S-(N-Benzyl)amino-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2S,4S-di-(N-Benzyl)amino-butane-1,3S-diol,1-[5-(3R-(N-Acétyl)amino-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2R,4R-di-(N-Acétyl)amino-butane-1,3S-diol,1-[5-(3S-(N-Acétyl)amino-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2S,4S-di-(N-Acétyl)amino-butane-1,3S-diol,1-[5-(3R-(N-Butanoyl)amino-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2R,4R-di-(N-Butanoyl)amino-butane-1,3S-diol,1-[5-(3S-(N-Butanoyl)amino-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2S,4S-di-(N-Butanoyl)amino-butane-1,3S-diol,1-[5-(3R-(N-Benzoyl)amino-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2R,4R-di-(N-Benzoyl)amino-butane-1,3S-diol,1-[5-(3S-(N-Benzoyl)amino-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2S,4S-di-(N-Benzoyl)amino-butane-1,3S-diol,1-[5-(3R-Méthoxy-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2R,4R-di-méthoxy-butane-1,3S-diol,1-[5-(3S-Méthoxy-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2S,4S-di-méthoxy-butane-1,3S-diol,1-[5-(3R-Ethoxy-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2R,4R-di-éthoxy-butane-1,3S-diol,1-[5-(3S-Ethoxy-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2S,4S-di-éthoxy-butane-1,3S-diol,1-[5-(3R-n-Butoxy-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2R,4R-di-n-butoxy-butane-1,3S-diol,1-[5-(3S-n-Butoxy-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2S,4S-di-n-butoxy-butane-1,3S-diol,1-[5-(3R-(2-Hydroxyéthyl)oxy-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2R,4R-di-(2-hydroxyéthyl)oxy-butane-1,3S-diol,1-[5-(3S-(2-Hydroxyéthyl)oxy-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2S,4S-di-(2-hydroxyéthyl)oxy-butane-1,3S-diol,1-[5-(3R-(3-Hydroxy-n-propyl)oxy-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2R,4R-di-(3-hydroxy-n-propyl)oxy-butane-1,3S-diol,1-[5-(3S-(3-Hydroxy-n-propyl)oxy-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2S,4S-di-(3-hydroxy-n-propyl)oxy-butane-1,3S-diol,1-[5-(3R-(Carboxyméthyl)oxy-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2R,4R-di-(carboxyméthyl)oxy-butane-1,3S-diol,1-[5-(3S-(Carboxyméthyl)oxy-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2S,4S-di-(carboxyméthyl)oxy-butane-1,3S-diol,1-[5-(3R-(3-Carboxy-n-propyl)oxy-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2R,4R-di-(3-carboxy-n-propyl)oxy-butane-1,3S-diol,1-[5-(3S-(3-Carboxy-n-propyl)oxy-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2S,4S-di-(3-carboxy-n-propyl)oxy-butane-1,3S-diol,1-[5-(3R,4-Dihydroxy-2-oxo-butyl)-pyrazin-2-yl]-3R,4-dihydroxy-butane-1,2-dione,4-[5-(2S,4-Dihydroxy-butyl)-pyrazin-2-yl]-butane-1,3S-diol,4-[5-(3R-Fluoro-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2R,4R-difluoro-butane-1,3S-diol,4-[5-(3S-Fluoro-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2S,4S-difluoro-butane-1,3S-diol,4-[5-(3R-Amino-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2R,4R-diamino-butane-1,3S-diol,4-[5-(3S-Amino-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2S,4S-diamino-butane-1,3S-diol,4-[5-(3R-(N-Méthyl)amino-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2R,4R-di-(N-méthyl)amino-butane-1,3S-diol,4-[5-(3S-(N-Méthyl)amino-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2S,4S-di-(N-méthyl)amino-butane-1,3S-diol,4-[5-(3R-(N-Ethyl)amino-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2R,4R-di-(N-éthyl)amino-butane-1,3S-diol,4-[5-(3S-(N-Ethyl)amino-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2S,4S-di-(N-éthyl)amino-butane-1,3S-diol,4-[5-(3R-(N-n-Butyl)amino-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2R,4R-di-(N-n-butyl)amino-butane-1,3S-diol,4-[5-(3S-(N-n-Butyl)amino-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2S,4S-di-(N-n-butyl)amino-butane-1,3S-diol,4-[5-(3R-(N-Benzyl)amino-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2R,4R-di-(N-benzyl)amino-butane-1,3S-diol,4-[5-(3S-(N-Benzyl)amino-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2S,4S-di-(N-benzyl)amino-butane-1,3S-diol,4-[5-(3R-(N-Acétyl)amino-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2R,4R-di-(N-acétyl)amino-butane-1,3S-diol,4-[5-(3S-(N-Acétyl)amino-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2S,4S-di-(N-acétyl)amino-butane-1,3S-diol,4-[5-(3R-(N-Butanoyl)amino-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2R,4R-di-(N-butanoyl)amino-butane-1,3S-diol,4-[5-(3S-(N-Butanoyl)amino-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2S,4S-di-(N-butanoyl)amino-butane-1,3S-diol,4-[5-(3R-(N-Benzoyl)amino-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2R,4R-di-(N-benzoyl)amino-butane-1,3S-diol,4-[5-(3S-(N-Benzoyl)amino-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2S,4S-di-(N-benzoyl)amino-butane-1,3S-diol,4-[5-(3R-Méthoxy-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2R,4R-diméthoxy-butane-1,3S-diol,4-[5-(3S-Méthoxy-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2S,4S-diméthoxy-butane-1,3S-diol,4-[5-(3R-Ethoxy-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2R,4R-diéthoxy-butane-1,3S-diol,4-[5-(3S-Ethoxy-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2S,4S-diéthoxy-butane-1,3S-diol,4-[5-(3R-n-Butoxy-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2R,4R-di-n-Butoxy-butane-1,3S-diol,4-[5-(3S-n-Butoxy-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2S,4S-di-n-Butoxy-butane-1,3S-diol,4-[5-(3R-(2-Hydroxyéthyl)oxy-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2R,4R-di-(2-hydroxyéthyl)oxy-butane-1,3S-diol,4-[5-(3S-(2-Hydroxyéthyl)oxy-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2S,4S-di-(2-hydroxyéthyl)oxy-butane-1,3S-diol,4-[5-(3R-(3-Hydroxy-n-propyl)oxy-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2R,4R-di-(3-hydroxy-n-propyl)oxy-butane-1,3S-diol,4-[5-(3S-(3-Hydroxy-n-propyl)oxy-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2S,4S-di-(3-hydroxy-n-propyl)oxy-butane-1,3S-diol,4-[5-(3R-(Carboxyméthyl)oxy-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2R,4R-di-(carboxyméthyl)oxy-butane-1,3S-diol,4-[5-(3S-(Carboxyméthyl)oxy-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2S,4S-di-(carboxyméthyl)oxy-butane-1,3S-diol,4-[5-(3R-(3-Carboxy-n-propyl)oxy-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2R,4R-di-(3-carboxy-n-propyl)oxy-butane-1,3S-diol,4-[5-(3S-(3-Carboxy-n-propyl)oxy-2S,4-dihydroxy-butyl)-pyrazin-2-yl]-2S,4S-di-(3-carboxy-n-propyl)oxy-butane-1,3S-diol,1-[5-(3S,4-Dihydroxy-1E-butényl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol,
- Médicaments contenant en tant que principe actif au moins un composé de formule (I) selon la revendication 1 pour lesquels
R9 et R10 représentent chacun un radical -CH2OH, et soitR8 représente un radical alkyle,a) R3 représente un radical méthylène, R4 représente un radical -CHOH- et l'un des radicaux R1, R2, R5 et R6 représente un radical -CHFou -CH(OR8) et les autres représentent chacun un radical -CHOH-,b) R2 et R5 représentent chacun un radical -CHOH-, R3 représente un radical méthylène et R1, R4 et R6 sont identiques et représentent chacun un radical -CH(OR8),c) R1 et R6 représentent chacun un radical -CHOH-, R3 représente un radical méthylène et R2, R4 et R5 sont identiques et représentent chacun un radical -CH(OR8),d) R1, R4, R5 et R6 représentent chacun un radical -CHOH- et -R2-R3-représente un radical -CH=CH-,
leurs stéréoisomères, les formes cis et trans du composé pour lequel -R2-R3-représente une chaíne -CH=CH- et leurs sels avec un acide minéral ou organique pharmaceutiquement acceptable. - Médicaments contenant en tant que principe actif au moins un composé de formule (I) selon la revendication 1 choisi parmi les composés suivants :et leurs sels avec un acide minéral ou organique pharmaceutiquement acceptable.1-[5-(3S,4-Dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol1-[5-(3S,4-Dihydroxy-1E-butényl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol1-[5-(2S-Méthoxy-3R,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol1-[5-(2R-fluoro-3R,4-dihydroxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol1-[5-(2S,4-dihydroxy-3R-méthoxy-butyl)-pyrazin-2-yl]-butane-1R,2S,3R,4-tétraol4-[5-(3R,4-dihydroxy-2S-méthoxy-butyl)]-pyrazin-2-yl]-3R,4R-diméthoxy-butane-1,2-diol
- Composés de formule : dans laquelle
R9 et R10 représentent chacun un radical -CH2OH, et soitR7 représente un atome d'hydrogène ou un radical alkyle, -CO-alk, -CO-Ar ou -CO-Het,a) R3 représente un radical méthylène, R4 représente un radical -CHOH- et l'un des radicaux R1, R2, R5 et R6 représente un radical carbonyle, méthylène, -CHF-, -CH(NHR7)- ou -CH(OR8) et les autres représentent chacun un radical -CHOH-,b) R2 et R5 représentent chacun un radical -CHOH-, R3 représente un radical méthylène et R1, R4 et R6 sont identiques et représentent chacun un radical carbonyle, méthylène, -CHF-, -CH(NHR7)- ou -CH(OR8),c) R1 et R6 représentent chacun un radical -CHOH-, R3 représente un radical méthylène et R2, R4 et R5 sont identiques et représentent chacun un radical carbonyle, méthylène, -CHF-, -CH(NHR7)- ou -CH(OR8),d) R1, R4, R5 et R6 représentent chacun un radical -CHOH- et -R2-R3-représente un radical -CH=CH-,
R8 représente un radical alkyle, -alk-COOH ou -alk-OH,
alk représente un radical alkyle,
Ar représente un radical phényle ou phényle substitué par un ou plusieurs substituants choisis parmi un atome d'halogène, un radical alkyle, alcoxy, alcoxycarbonyle, amino, monoalkylamino ou dialkylamino,
Het représente un hétérocycle mono, di ou tricyclique saturé ou insaturé contenant 1 à 9 atomes de carbone et un ou plusieurs hétéroatomes choisis parmi oxygène, soufre et azote,
les radicaux et portions alkyle et alcoxy contenant 1 à 6 atomes de carbone en chaíne droite ou ramifiée,
ou un de ses stéréoisomères ou pour les composés pour lesquels -R2-R3-représente un radical -CH=CH- leurs formes cis ou trans ou un de ses sels, à l'exception du composé de formule : - Procédé de préparation des composés de formule (I) selon la revendication 6 pour lesquels R9 et R10 représentent chacun un radical -CH2OH et soit R3 représente un radical méthylène, R4 représente un radical -CHOH- et l'un des radicaux R1, R2, R5 et R6 représente un radical carbonyle et les autres représentent chacun un radical -CHOH-, soit R1 et R6 représentent chacun un radical -CHOH-, R3 représente un radical méthylène et R2, R4 et R5 sont identiques et représentent chacun un radical carbonyle, soit R2 et R5 représentent chacun un radical -CHOH-, R3 représente un radical méthylène et R1, R4 et R6 sont identiques et représentent chacun un radical carbonyle caractérisé en ce que l'on oxyde un dérivé choisi parmi les formules : dans lesquelles Ra représente un radical trialkylsilyle, alkyldiphénylsilyle ou dialkylphénylsilyle, Rb représente un radical phényle éventuellement substitué par au moins un radical alcoxy et Rc représente un radical alkyle ou phényle, les radicaux et portions alkyle et alcoxy contenant 1 à 6 atomes de carbone en chaíne droite ou ramifiée, ou un stéréoisomère d'un tel dérivé, puis déprotège les hydroxyles, isole le produit et le transforme éventuellement en sel.
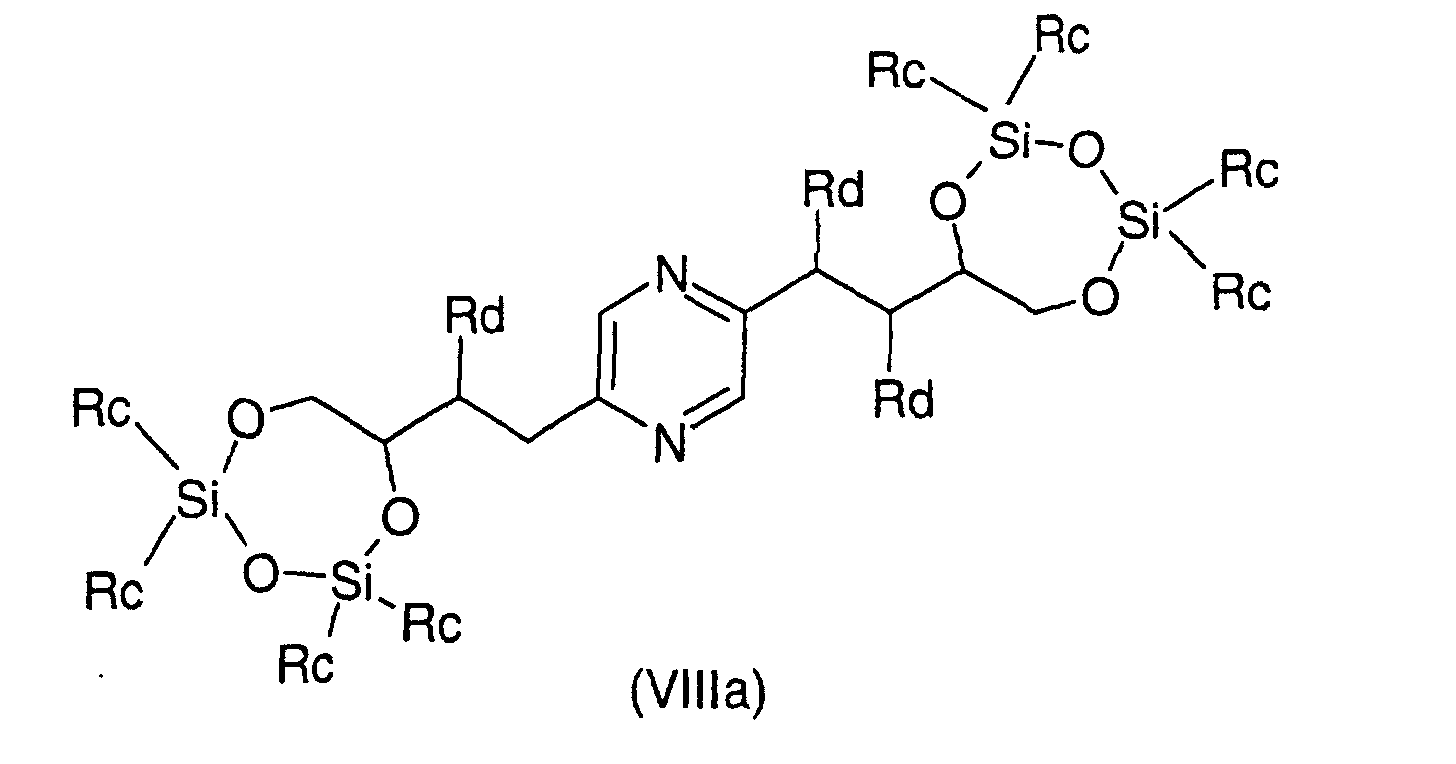
- Procédé de préparation des composés de formule (I) selon la revendication 6 pour lesquels R9 et R10 représentent chacun un radical -CH2OH et soit R3 représente un radical méthylène, R4 représente un radical -CHOH- et l'un des radicaux R1, R2, R5 et R6 représente un radical méthylène et les autres représentent chacun un radical -CHOH-, soit R1 et R6 représentent chacun un radical -CHOH-, R3 représente un radical méthylène et R2, R4 et R5 sont identiques et représentent chacun un radical méthylène, soit R2 et R5 représentent chacun un radical -CHOH- et R1, R3, R4 et R6 représentent chacun un radical méthylène caractérisé en ce que l'on condense un chlorothionocarbonate d'alkyle ou de phényle sur un dérivé choisi parmi les formules suivantes : dans lesquelles Ra représente un radical trialkylsilyle, alkyldiphénylsilyle ou dialkylphénylsilyle, Rb représente un radical phényle éventuellement substitué par au moins un radical alcoxy et Rc représente un radical alkyle ou phényle, les radicaux et portions alkyle et alcoxy contenant 1 à 6 atomes de carbone en chaíne droite ou ramifiée, ou un stéréoisomère d'un tel dérivé, puis déprotège les hydroxyles, isole le produit et le transforme éventuellement en sel.
- Procédé de préparation des composés de formule (I) selon la revendication 6 pour lesquels R9 et R10 représentent chacun un radical -CH2OH et soit R3 représente un radical méthylène, R4 représente un radical -CHOH- et l'un des radicaux R1, R2, R5 et R6 représente un radical -CHF- et les autres représentent chacun un radical -CHOH-, soit R1 et R6 représentent chacun un radical -CHOH-, R3 représente un radical méthylène et R2, R4 et R5 sont identiques et représentent chacun un radical -CHF-, soit R2 et R5 représentent chacun un radical -CHOH-, R3 représente un radical méthylène et R1, R4 et R6 sont identiques et représentent chacun un radical -CHF-caractérisé en ce que l'on fluorure un dérivé choisi parmi les formules: dans lesquelles Ra représente un radical trialkylsilyle, alkyldiphénylsilyle ou dialkylphénylsilyle, Rb représente un radical phényle éventuellement substitué par au moins un radical alcoxy et Rc représente un radical alkyle ou phényle, les radicaux et portions alkyle et alcoxy contenant 1 à 6 atomes de carbone en chaíne droite ou ramifiée, ou un stéréoisomère d'un tel dérivé, puis déprotège les hydroxyles, isole le produit et le transforme éventuellement en sel.
- Procédé de préparation des composés de formule (I) selon la revendication 6 pour lesquels R9 et R10 représentent chacun un radical -CH2OH et soit R3 représente un radical méthylène, R4 représente un radical -CHOH- et l'un des radicaux R1, R2, R5 et R6 représente un radical -CH(NHR7)- et les autres représentent chacun un radical -CHOH-, soit R1 et R6 représentent chacun un radical -CHOH-, R3 représente un radical méthylène et R2, R4 et R5 sont identiques et représentent chacun un radical -CH(NHR7)-, soit R2 et R5 représentent chacun un radical -CHOH-, R3 représente un radical méthylène et R1, R4 et R6 sont identiques et représentent chacun un radical -CH(NHR7)- caractérisé en ce que l'on réduit un dérivé choisi parmi les formules : dans lesquelles Ra représente un radical trialkylsilyle, alkyldiphénylsilyle ou dialkylphénylsilyle, Rb représente un radical phényle éventuellement substitué par au moins un radical alcoxy, Rc représente un radical alkyle ou phényle et Rd représente un radical azido, les radicaux et portions alkyle et alcoxy contenant 1 à 6 atomes de carbone en chaíne droite ou ramifiée, ou un stéréoisomère d'un tel dérivé, suivie éventuellement de la réaction d'un dérivé de formule HalR7 pour lequel R7 a les mêmes significations que dans la revendication 4 sauf hydrogène, Hal représente un atome d'halogène, déprotège les hydroxyles, isole le produit et le transforme éventuellement en sel.
- Procédé de préparation des composés de formule (I) selon la revendication 6 pour lesquels R9 et R10 représentent chacun un radical -CH2OH et soit R3 représente un radical méthylène, R4 représente un radical -CHOH- et l'un des radicaux R1, R2, R5 et R6 représente un radical -CH(OR8) et les autres représentent chacun un radical -CHOH-, soit R1 et R6 représentent chacun un radical -CHOH-, R3 représente un radical méthylène et R2, R4 et R5 sont identiques et représentent chacun un radical -CH(OR8), soit R2 et R5 représentent chacun un radical -CHOH-, R3 représente un radical méthylène et R1, R4 et R6 sont identiques et représentent chacun un radical -CH(OR8) caractérisé en ce que l'on fait réagir un dérivé de formule dans lesquelles Ra représente un radical trialkylsilyle, alkyldiphénylsilyle ou dialkylphénylsilyle, Rb représente un radical phényle éventuellement substitué par au moins un radical alcoxy et Rc représente un radical alkyle ou phényle, les radicaux et portions alkyle et alcoxy contenant 1 à 6 atomes de carbone en chaíne droite ou ramifiée, ou un stéréoisomère d'un tel dérivé, sur un dérivé HaIR8 dans lequel R8 a les mêmes significations que dans la revendication 4, puis déprotège les hydroxyles, isole le produit et le transforme éventuellement en sel.
- Procédé de préparation des composés de formule (I) selon la revendication 6 pour lesquels R9 et R10 représentent chacun un radical -CH2OH et R1, R4, R5 et R6 représentent chacun un radical -CHOH- et -R2-R3- représente un radical -CH=CH- caractérisé en ce que l'on déshydrate un dérivé de formule : dans laquelle Rd représente un radical -OSO2-Re et Re représente un radical méthyle, trifluorométhyle ou 4-méthylphényle ou un stéréoisomère d'un tel dérivé, puis déprotège les hydroxyles, isole le produit et le transforme éventuellement en sel.
- Utilisation des composés de formule générale : dans laquelle
R9 et R10 sont identiques et représentent un radical -CH2OH, et soitR7 représente un atome d'hydrogène ou un radical alkyle, -CO-alk, -CO-Ar ou -CO-Het,a) R3 représente un radical méthylène, R4 représente un radical -CHOH- et l'un des radicaux R1, R2, R5 et R6 représente un radical carbonyle, méthylène, -CHF-, -CH(NHR7)- ou -CH(OR8) et les autres représentent chacun un radical -CHOH-,b) R2 et R5 représentent chacun un radical -CHOH-, R3 représente un radical méthylène et R1, R4 et R6 sont identiques et représentent chacun un radical carbonyle, méthylène, -CHF-, -CH(NHR7)- ou -CH(OR8),c) R1 et R6 représentent chacun un radical -CHOH-, R3 représente un radical méthylène et R2, R4 et R5 sont identiques et représentent chacun un radical carbonyle, méthylène, -CHF-, -CH(NHR7)- ou -CH(OR8),d) R1, R4, R5 et R6 représentent chacun un radical -CHOH- et -R2-R3-représente un radical -CH=CH-,
R8 représente un radical alkyle, -alk-COOH ou -alk-OH,
alk représente un radical alkyle,
Ar représente un radical phényle ou phényle substitué par un ou plusieurs substituants choisis parmi un atome d'halogène, un radical alkyle, alcoxy, alcoxycarbonyle, amino, monoalkylamino ou dialkylamino,
Het représente un hétérocycle mono, di ou tricyclique saturé ou insaturé contenant 1 à 9 atomes de carbone et un ou plusieurs hétéroatomes choisis parmi oxygène, soufre et azote,
les radicaux et portions alkyle et alcoxy contenant 1 à 6 atomes de carbone en chaíne droite ou ramifiée,
ou un de ses stéréoisomères ou pour le composé pour lequel -R2-R3-représente un radical -CH=CH- ses formes cis ou trans ou un de ses sels avec un acide minéral ou organique pharmaceutiquement acceptable pour la préparation d'un médicament utile pour le traitement ou la prévention du diabète et des complications du diabète.
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| FR9709059 | 1997-07-17 | ||
| FR9709059A FR2766183B1 (fr) | 1997-07-17 | 1997-07-17 | Medicaments contenant des polyhydroxybutylpyrazines, les polyhydroxybutylpyrazines nouvelles et leur preparation |
| PCT/FR1998/001543 WO1999003841A1 (fr) | 1997-07-17 | 1998-07-15 | Polyhydroxybutylpyrazines, leur preparation et medicaments les contenant |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| EP1001942A1 EP1001942A1 (fr) | 2000-05-24 |
| EP1001942B1 true EP1001942B1 (fr) | 2003-06-25 |
Family
ID=9509301
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP98939677A Expired - Lifetime EP1001942B1 (fr) | 1997-07-17 | 1998-07-15 | Polyhydroxybutylpyrazines, leur preparation et medicaments les contenant |
Country Status (25)
| Country | Link |
|---|---|
| US (1) | US6288066B1 (fr) |
| EP (1) | EP1001942B1 (fr) |
| JP (1) | JP4443033B2 (fr) |
| KR (1) | KR20010021871A (fr) |
| CN (1) | CN1264368A (fr) |
| AT (1) | ATE243683T1 (fr) |
| AU (1) | AU745905B2 (fr) |
| BR (1) | BR9810595A (fr) |
| CA (1) | CA2296725C (fr) |
| CZ (1) | CZ290896B6 (fr) |
| DE (1) | DE69815854T2 (fr) |
| DK (1) | DK1001942T3 (fr) |
| ES (1) | ES2196595T3 (fr) |
| FR (1) | FR2766183B1 (fr) |
| HU (1) | HUP0004661A3 (fr) |
| IL (1) | IL134039A0 (fr) |
| MX (1) | MXPA00000437A (fr) |
| NO (1) | NO20000199L (fr) |
| NZ (1) | NZ502279A (fr) |
| PL (1) | PL338147A1 (fr) |
| PT (1) | PT1001942E (fr) |
| RU (1) | RU2186773C2 (fr) |
| SK (1) | SK462000A3 (fr) |
| WO (1) | WO1999003841A1 (fr) |
| ZA (1) | ZA986336B (fr) |
Families Citing this family (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR2802815A1 (fr) * | 1999-12-23 | 2001-06-29 | Aventis Pharma Sa | Association de deoxyfructosazine et d'un antidiabetique agoniste du recepteur gamma-active par le proliferateur de peroxisome |
| FR2802813A1 (fr) * | 1999-12-23 | 2001-06-29 | Aventis Pharma Sa | Association de deoxyfructosazine et d'un antidiabetique de la famille des biguandes |
| FR2802816A1 (fr) * | 1999-12-23 | 2001-06-29 | Aventis Pharma Sa | Association de deoxyfructosazine et d'un antidiabetique inhibiteur d'alpha-glucosidase |
| US6794506B2 (en) * | 2000-07-21 | 2004-09-21 | Elan Pharmaceuticals, Inc. | 3-(heteroaryl) alanine derivatives-inhibitors of leukocyte adhesion mediated by VLA-4 |
| DE102005047647A1 (de) * | 2005-10-05 | 2007-04-12 | Merck Patent Gmbh | a,a'-Dihydroxyketonderivate und deren Verwendung als UV-Filter |
| CN101970409A (zh) | 2007-03-19 | 2011-02-09 | 帝斯曼知识产权资产管理有限公司 | 紫外光遮蔽剂化合物 |
Family Cites Families (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR2744365B1 (fr) * | 1996-02-06 | 1998-02-27 | Inst Malgache Rech Appl | Melanges isolables a partir de graines d'eugenia jambolana lamarck, leur preparation et l'utilisation de ces melanges et certains de leurs constituants comme medicaments |
-
1997
- 1997-07-17 FR FR9709059A patent/FR2766183B1/fr not_active Expired - Fee Related
-
1998
- 1998-07-15 EP EP98939677A patent/EP1001942B1/fr not_active Expired - Lifetime
- 1998-07-15 IL IL13403998A patent/IL134039A0/xx unknown
- 1998-07-15 NZ NZ502279A patent/NZ502279A/en unknown
- 1998-07-15 AU AU88103/98A patent/AU745905B2/en not_active Ceased
- 1998-07-15 HU HU0004661A patent/HUP0004661A3/hu unknown
- 1998-07-15 RU RU2000103447/04A patent/RU2186773C2/ru not_active IP Right Cessation
- 1998-07-15 JP JP2000503066A patent/JP4443033B2/ja not_active Expired - Fee Related
- 1998-07-15 DK DK98939677T patent/DK1001942T3/da active
- 1998-07-15 WO PCT/FR1998/001543 patent/WO1999003841A1/fr not_active Application Discontinuation
- 1998-07-15 CA CA002296725A patent/CA2296725C/fr not_active Expired - Fee Related
- 1998-07-15 SK SK46-2000A patent/SK462000A3/sk unknown
- 1998-07-15 BR BR9810595-7A patent/BR9810595A/pt not_active IP Right Cessation
- 1998-07-15 AT AT98939677T patent/ATE243683T1/de not_active IP Right Cessation
- 1998-07-15 DE DE69815854T patent/DE69815854T2/de not_active Expired - Lifetime
- 1998-07-15 MX MXPA00000437A patent/MXPA00000437A/es not_active IP Right Cessation
- 1998-07-15 CN CN98807197A patent/CN1264368A/zh active Pending
- 1998-07-15 KR KR1020007000435A patent/KR20010021871A/ko not_active Application Discontinuation
- 1998-07-15 PL PL98338147A patent/PL338147A1/xx not_active Application Discontinuation
- 1998-07-15 PT PT98939677T patent/PT1001942E/pt unknown
- 1998-07-15 CZ CZ2000121A patent/CZ290896B6/cs not_active IP Right Cessation
- 1998-07-15 ES ES98939677T patent/ES2196595T3/es not_active Expired - Lifetime
- 1998-07-16 ZA ZA986336A patent/ZA986336B/xx unknown
-
2000
- 2000-01-14 NO NO20000199A patent/NO20000199L/no not_active Application Discontinuation
- 2000-01-18 US US09/484,342 patent/US6288066B1/en not_active Expired - Fee Related
Also Published As
| Publication number | Publication date |
|---|---|
| RU2186773C2 (ru) | 2002-08-10 |
| IL134039A0 (en) | 2001-04-30 |
| DE69815854T2 (de) | 2004-05-13 |
| PL338147A1 (en) | 2000-09-25 |
| SK462000A3 (en) | 2000-06-12 |
| ES2196595T3 (es) | 2003-12-16 |
| US6288066B1 (en) | 2001-09-11 |
| FR2766183A1 (fr) | 1999-01-22 |
| KR20010021871A (ko) | 2001-03-15 |
| ZA986336B (en) | 1999-01-27 |
| CN1264368A (zh) | 2000-08-23 |
| DE69815854D1 (de) | 2003-07-31 |
| HUP0004661A2 (hu) | 2001-11-28 |
| EP1001942A1 (fr) | 2000-05-24 |
| AU745905B2 (en) | 2002-04-11 |
| ATE243683T1 (de) | 2003-07-15 |
| FR2766183B1 (fr) | 1999-08-27 |
| JP4443033B2 (ja) | 2010-03-31 |
| BR9810595A (pt) | 2000-09-12 |
| WO1999003841A1 (fr) | 1999-01-28 |
| NO20000199D0 (no) | 2000-01-14 |
| CZ2000121A3 (cs) | 2000-05-17 |
| MXPA00000437A (es) | 2002-04-17 |
| DK1001942T3 (da) | 2003-10-20 |
| HUP0004661A3 (en) | 2001-12-28 |
| NZ502279A (en) | 2002-04-26 |
| CA2296725C (fr) | 2008-11-18 |
| CZ290896B6 (cs) | 2002-11-13 |
| NO20000199L (no) | 2000-01-14 |
| PT1001942E (pt) | 2003-11-28 |
| AU8810398A (en) | 1999-02-10 |
| JP2001510185A (ja) | 2001-07-31 |
| CA2296725A1 (fr) | 1999-01-28 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP1001942B1 (fr) | Polyhydroxybutylpyrazines, leur preparation et medicaments les contenant | |
| EP1001944B1 (fr) | Derives de pyrazine, leur preparation et les medicaments les contenant | |
| EP0998465B1 (fr) | Derives de polyhydroxylalkylpyrazine, leur preparation, medicaments les contenant | |
| EP0998464B1 (fr) | Derives de polyhydroxyalkylpyrazines, leur preparation et les medicaments les contenant | |
| EP1001943B1 (fr) | Derives de polyhydroxyalkylpyrazines, leur preparation et medicaments les contenant | |
| EP1140861B1 (fr) | Nouveaux derives de polyhydroxypyrazines, leur preparation et les compositions pharmaceutiques qui les contiennent | |
| CA2337350C (fr) | Derives de polyhydroxyalkylpyrazine, leur preparation et compositions pharmaceutiques les contenant | |
| FR2766180A1 (fr) | Medicaments contenant en tant que principe actif un derive de 2-(1,2,3,4-tetrahydroxybutyl)pyrazine,nouveaux derives de 2-(1,2,3,4-tetrahydroxybutyl) pyrazine et leur preparation | |
| WO1999003840A1 (fr) | Medicaments contenant des derives de polyhydroxyalkylpyrazine, les nouveaux derives de polyhydroxyalkylpyrazine et leur preparation | |
| FR2766181A1 (fr) | Derives de polyhydroxybutylpyrazines, leur preparation et les medicaments les contenant | |
| FR2766186A1 (fr) | Medicaments contenant des derives de polyhydroxyalkylpyrazine, les nouveaux derives de polyhydroxyalkylpyrazine et leur preparation | |
| CZ2000119A3 (cs) | Deriváty polyhydroxyalkylpyrazinu, způsob jejich přípravy a léčiva, která je obsahují |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| 17P | Request for examination filed |
Effective date: 20000121 |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): AT BE CH CY DE DK ES FI FR GB GR IE IT LI LU NL PT SE |
|
| 17Q | First examination report despatched |
Effective date: 20020426 |
|
| GRAH | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOS IGRA |
|
| GRAH | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOS IGRA |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Designated state(s): AT BE CH CY DE DK ES FI FR GB GR IE IT LI LU NL PT SE |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: FG4D Free format text: NOT ENGLISH |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: EP |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: CY Payment date: 20030704 Year of fee payment: 6 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: AT Payment date: 20030709 Year of fee payment: 6 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: LU Payment date: 20030711 Year of fee payment: 6 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: FI Payment date: 20030714 Year of fee payment: 6 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: CH Payment date: 20030723 Year of fee payment: 6 |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: FG4D Free format text: FRENCH |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: SE Payment date: 20030728 Year of fee payment: 6 |
|
| REG | Reference to a national code |
Ref country code: SE Ref legal event code: TRGR |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: IE Payment date: 20030731 Year of fee payment: 6 Ref country code: NL Payment date: 20030731 Year of fee payment: 6 |
|
| REF | Corresponds to: |
Ref document number: 69815854 Country of ref document: DE Date of ref document: 20030731 Kind code of ref document: P |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: BE Payment date: 20030819 Year of fee payment: 6 |
|
| REG | Reference to a national code |
Ref country code: GR Ref legal event code: EP Ref document number: 20030402459 Country of ref document: GR |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: NV Representative=s name: PATENTANWAELTE FELDMANN & PARTNER AG |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: PT Payment date: 20030929 Year of fee payment: 6 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: DK Payment date: 20030930 Year of fee payment: 6 |
|
| GBT | Gb: translation of ep patent filed (gb section 77(6)(a)/1977) | ||
| REG | Reference to a national code |
Ref country code: ES Ref legal event code: FG2A Ref document number: 2196595 Country of ref document: ES Kind code of ref document: T3 |
|
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| 26N | No opposition filed |
Effective date: 20040326 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LU Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20040715 Ref country code: IE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20040715 Ref country code: FI Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20040715 Ref country code: CY Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20040715 Ref country code: AT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20040715 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20040716 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LI Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20040731 Ref country code: CH Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20040731 Ref country code: BE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20040731 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DK Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20040802 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: PT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20050117 |
|
| BERE | Be: lapsed |
Owner name: S.A. *AVENTIS PHARMA Effective date: 20040731 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: NL Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20050201 |
|
| REG | Reference to a national code |
Ref country code: DK Ref legal event code: EBP |
|
| EUG | Se: european patent has lapsed | ||
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PL |
|
| REG | Reference to a national code |
Ref country code: PT Ref legal event code: MM4A Free format text: LAPSE DUE TO NON-PAYMENT OF FEES Effective date: 20050117 |
|
| NLV4 | Nl: lapsed or anulled due to non-payment of the annual fee |
Effective date: 20050201 |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: MM4A |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GR Payment date: 20050928 Year of fee payment: 8 |
|
| BERE | Be: lapsed |
Owner name: S.A. *AVENTIS PHARMA Effective date: 20040731 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GR Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20070202 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GB Payment date: 20120711 Year of fee payment: 15 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: FR Payment date: 20120719 Year of fee payment: 15 Ref country code: ES Payment date: 20120824 Year of fee payment: 15 Ref country code: DE Payment date: 20120711 Year of fee payment: 15 Ref country code: IT Payment date: 20120714 Year of fee payment: 15 |
|
| GBPC | Gb: european patent ceased through non-payment of renewal fee |
Effective date: 20130715 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R119 Ref document number: 69815854 Country of ref document: DE Effective date: 20140201 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: ST Effective date: 20140331 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GB Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20130715 Ref country code: DE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20140201 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20130715 Ref country code: FR Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20130731 |
|
| REG | Reference to a national code |
Ref country code: ES Ref legal event code: FD2A Effective date: 20140908 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: ES Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20130716 |