EP0814840B1 - Oral pharmaceutical dosage forms comprising a proton pump inhibitor and a prokinetic agent - Google Patents
Oral pharmaceutical dosage forms comprising a proton pump inhibitor and a prokinetic agent Download PDFInfo
- Publication number
- EP0814840B1 EP0814840B1 EP96944725A EP96944725A EP0814840B1 EP 0814840 B1 EP0814840 B1 EP 0814840B1 EP 96944725 A EP96944725 A EP 96944725A EP 96944725 A EP96944725 A EP 96944725A EP 0814840 B1 EP0814840 B1 EP 0814840B1
- Authority
- EP
- European Patent Office
- Prior art keywords
- dosage form
- enteric coating
- proton pump
- pump inhibitor
- pellets
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Lifetime
Links
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K45/00—Medicinal preparations containing active ingredients not provided for in groups A61K31/00 - A61K41/00
- A61K45/06—Mixtures of active ingredients without chemical characterisation, e.g. antiphlogistics and cardiaca
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/44—Non condensed pyridines; Hydrogenated derivatives thereof
- A61K31/445—Non condensed piperidines, e.g. piperocaine
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/04—Drugs for disorders of the alimentary tract or the digestive system for ulcers, gastritis or reflux esophagitis, e.g. antacids, inhibitors of acid secretion, mucosal protectants
Definitions
- the present invention is related to new oral pharmaceutical preparations especially for use in the prevention and treatment of disorders associated with gastro oesophageal reflux.
- the present preparations comprise a gastric acid suppressing agent, such as a proton pump inhibitor, in combination with one or more prokinetic agents in a new fixed unit dosage form, especially a tablet.
- the present invention refers to a method for the manufacture of such preparations and the use of such preparations in medicine, especially in the treatment of gastro oesophageal reflux diseases and other gastrointestinal disorders.
- Gastro oesophageal reflux disease is among the most common disorders seen by gastroenterologists and general practicians. The wide diversity of symptoms and disease severity produced by acid reflux has led to the need for more individualized treatment strategies.
- Therapeutic agents effective in the treatment of GORD include gastric acid suppressing agents, such as H 2 receptor antagonists, proton pump inhibitors, other agents of interest are antacids/alginates and prokinetic agents. These agents can be distinguished by their mechanisms of action, safety profile, pharmacokinetics and indications.
- Antacids and alginates are still widely used. They have a short duration of action but are seen as inexpensive and safe. They do not provide a layterm symptom resolution of GORD.
- H 2 receptor antagonists are widely prescribed for GORD. Their higher cost has been compensated by the clinical results obtained both in terms of symptom relief and healing. These advantages have been related to their mode of action, which offered more potent and longer duration of effect on gastric acidity.
- Proton pump inhibitors such as omeprazole
- H 2 receptor antagonists particularly in reflux oesophagitis.
- Omeprazole is known to offer significant gain over H 2 receptor antagonists in terms of symptom resolution, healing and prevention of relapse for reflux oesophagitis.
- Prokinetic agents of the first generation e.g. bethanecol
- second generation e.g. domperidone and metoclopramide. blocks effects of endogenous dopamine in the gut.
- the results of double-blind placebo controlled trials in GORD patients have been conflicting.
- the action of the third generation of prokinetic agents such as substituted benzamides, e.g. cisapride and mosapride derives primarily, but not exclusively, from facilitating acetylcholine release from neurones of the myenteric plexus via stimulation of 5-HT4 receptors.
- a combination therapy of a prokinetic agent and a gastric acid lowering compound is rational and was shown more effective than mono therapy apart from full dose of proton pump inhibitors.
- Administration of cisapride and ranitidine was shown to further lower the exposure of the oesophagus to acid(s) ( Inauen W et al. Gut 1993; 34: 1025 - 1031).
- Such a therapy was also shown to improve healing rates (de Boer WA et al. Aliment Pharmacol Ther 1994; 8: 147 - 157).
- WO 95/01803 describes a pharmaceutical composition of famitidine, cisapride and optionally simethicone in the treatment of gastrointestinal distress.
- Such combination therapies might be considered for patients whose predominant symptom is regurgitation; those whose symptoms occur mainly at night; those with respiratory problems such as posterior laryngitis, asthma, chronic bronchitis, or recurrent aspiration; those with cough and hoarseness related to reflux disease.
- a combination therapy comprising an acid suppressing agent and a prokinetic agent is attractive, rational and effective.
- An acid suppressing agent plus a prokinetic agent could be an alternative to each of them separately in case of failure.
- the compliance of such a treatment may be a problem. It is well known that patient compliance is a main factor in receiving good results in medical treatments. Administration of two, three or even more different tablets to the patient is not convenient or satisfactory to achieve the most optimal results.
- the present invention now provides new oral dosage forms comprising two or more different active substances combined in one fixed unit dosage form, preferably a tablet.
- the present invention provides oral, fixed unit dosage forms, i.e. a multiple unit tableted dosage forms, multilayered tablets or a capsule filled with more than one pharmaceutically active compound.
- the active compounds present in the dosage form are preferably an acid susceptible proton pump inhibitor which is protected by an enteric coating layer, and one or more prokinetic agents. These new dosage forms will simplify the regimen and improve the patient compliance.
- One object of the invention is to provide an oral, multiple unit tableted dosage form comprising an acid susceptible proton pump inhibitor in the form of individually enteric coating layered units together with one or more prokinetic agents in the form of a powder or granules compressed into a tablet
- the enteric coating layer(s) covering the individual units of the proton pump inhibitor has properties such that the compression of the units into a tablet does not significantly affect the acid resistance of the individually enteric coating layered units.
- the multiple unit tableted dosage form provides a good stability of the active substances during long-term storage.
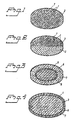
- the new fixed dosage form is preferably in the form of a multiple unit tableted dosage form comprising enteric coating layered units of the one of the active substance which is acid susceptible and granules of the other active substance, i.e. prepared prokinetic granules as shown in Fig. 1.
- the proton pump inhibitor in the form of enteric coating layered units, may also be mixed with pharmaceutically acceptable excipients and compressed into a tablet which is then filmcoated with an aqueous suspension containing the prokinetic substance, see Fig. 4.
- Another object of the invention is to provide a tablet preparation comprising a proton pump inhibitor in admixture with tablet excipients in a tablet core and a separate layer surrounding the tablet core, which layer comprises one or more prokinetic agent(s) presscoated onto the tablet core.
- the tablet core is enteric coating layered before the surrounding layer of prokinetic agents is applied.
- a separating layer also is applied on the tablet before the enteric coating layer, see Fig. 3.
- the prepared tablet is sectioned in separate layers, each one comprising different active substances.
- one layer comprises the proton pump inhibitor in the form of enteric coating layered pellets in admixture with pharmaceutically acceptable excipients and another layer(s) comprises(-e) the prolanetic agent(s) in admixture with pharmaceutically acceptable excipients, respectively, see Fig. 2.
- a further object of the invention is to provide a multiple unit tableted dosage form, which is divisible and easy to handle.
- Such a multiple unit tableted dosage form may be dispersed in an aqueous liquid and can be given to patients with swallowing disorders and in pediatrics.
- Such a suspension of dispersed units/pellets of appropriate size can be used for oral administration and also for feeding through a naso-gastric tube.
- the present invention provides a capsule preparation comprising the proton pump inhibitor in the form of enteric coating layered pellets mixed with one or more prokinetic agents in the form of prepared granules or pellets.
- the new fixed unit dosage forms comprise as active substances one gastric acid suppressing agent, such as an acid susceptible proton pump inhibitor and one or more prokinetic agents.
- the different therapeutically active components used in the dosage forms are defined below.
- the prokinetic part of the formulation may be formulated in the form of instant release, sustained release or extended release formulations. Alternatively, all the components of the formulation may be formulated in an effervescent formulation.
- the gastric acid suppressing agent is preferably an acid susceptible proton pump inhibitor.
- R 1 , R 2 and R 3 are the same or different and selected from hydrogen, alkyl, alkoxy optionally substituted by fluorine, alkylthio, alkoxyalkoxy, dialkylamino, piperidino, morpholino, halogen, phenyl and phenylalkoxy;
- R 4 and R 5 are the same or different and selected from hydrogen, alkyl and aralkyl;
- R 6 ' is hydrogen, halogen, trifluoromethyl, alkyl and alkoxy,
- R 6 -R 9 are the same or different and selected from hydrogen, alkyl
- the proton pump inhibitors used in the dosage forms of the invention may be used in neutral form or in the form of an alkaline salt, such as for instance the Mg 2+, Ca 2+, Na + , K + or Li + salts, preferably the Mg 2+ salts.
- an alkaline salt such as for instance the Mg 2+, Ca 2+, Na + , K + or Li + salts, preferably the Mg 2+ salts.
- the compounds listed above may be used in racemic form or in the form of a substantially pure enantiomer thereof, or alkaline salts of the single enantiomers.
- Suitable proton pump inhibitors are for example disclosed in EP-A1-0005129, EP-A1-174 726, EP-A1-166 287, GB 2 163 747 and WO90/06925, WO91/19711, WO91/1971 and further especially suitable compounds are described in WO95/01977 and WO94/27988.
- the gastric acid suppressing agent is preferably an acid susceptible proton pump inhibitor but other gastric acid suppressing agents such as the H 2 receptor antagonists: ranitidine, cimetidine or famotidine, may be used together with a prolanetic agent in the pharmaceutical compositions according to the present invention.
- gastric acid suppressing agents such as the H 2 receptor antagonists: ranitidine, cimetidine or famotidine, may be used together with a prolanetic agent in the pharmaceutical compositions according to the present invention.
- prolanetic compounds may be used in combination with a suitable proton pump inhibitor in the fixed unit dosage form according to the present invention.
- suitable prokinetic agents include for example cisapride, mosapride, metoclopramide, and domperidone.
- the active prokinetic agents could be in standard forms or used as salts, hydrates, esters etc.
- a combination of two or more of the above described drugs may be used.
- a preferable prokinetic agent for the new fixed dosage form is mosapride or cisapride.
- suitable prokinetic agents are described in EP 0 243 959 and EP 0 076 530.
- the preferred multiple unit tableted dosage form comprising a proton pump inhibitor in the form of a racemat, an alkaline salt or one of its single enantiomers in combination with a prokinetic compound, is characterized in the following way.
- Individually enteric coating layered units small beads, granules or pellets
- the prokinetic compound and tablet excipients may be dry mixed or wet-mixed into granules.
- the mixture of enteric coating layered units, prokinetic agent(s) and optionally excipients are compressed into the multiple unit tableted dosage forms.
- individual units is meant small beads, granules or pellets, in the following referred to as pellets of the proton pump inhibitor.
- the compaction process (compression) for formulating the multiple unit tableted dosage form must not significantly affect the acid resistance of the enteric coating layered pellets.
- the mechanical properties, such as the flexibility and hardness as well as the thickness of the enteric coating layer(s) must secure that the requirements on enteric coated articles in the United States Pharmacopeia are accomplished in that the acid resistance does not decrease more than 10% during the compression of the pellets into tablets.
- the acid resistance is defined as the amount of proton pump inhibitor in the tablets or pellets after being exposed to simulated gastric fluid USP, or to 0.1 M HCl (aq) relative to that of unexposed tablets and pellets, respectively.
- the test is accomplished in the following way. Individual tablets or pellets are exposed to stimulated gastric fluid of a temperature of 37°C. The tablets disintegrate rapidly and release the enteric coating layered pellets to the medium. After two hours the enteric coating layered pellets are removed and analyzed for content of the proton pump inhibitor using High Performance Liquid Chromatography (HPLC).
- HPLC High Performance Liquid Chromatography
- Core material - for enteric coating layered pellets comprising a proton pump inhibitor
- the core material for the individually enteric coating layered pellets can be constituted according to different principles. Seeds layered with the proton pump inhibitor, optionally mixed with alkaline substances, can be used as the core material for the further processing.
- the seeds which are to be layered with the proton pump inhibitor can be water insoluble seeds comprising different oxides, celluloses, organic polymers and other materials, alone or in mixtures or water-soluble seeds comprising different inorganic salts, sugars, non-pareils and other materials, alone or in mixtures. Further, the seeds may comprise the proton pump inhibitor in the form of crystals, agglomerates, compacts etc. The size of the seeds is not essential for the present invention but may vary between approximately 0.1 and 2 mm.
- the seeds layered with the proton pump inhibitor are produced either by powder or solution/suspension layering using for instance granulation or spray coating layering equipment
- the proton pump inhibitor may be mixed with further components.
- Such components can be binders, surfactants fillers, disintegrating agents, alkaline additives or other and/or pharmaceutically acceptable ingredients alone or in mixtures.
- the binders are for example polymers such as hydroxypropyl methylcellulose (HPMC), hydroxypropylcellulose (HPC), carboxymethylcellulose sodium, polyvinyl pyrrolidone (PVP), or sugars, starches or other pharmaceutically acceptable substances with cohesive properties.
- Suitable surfactants are found in the groups of pharmaceutically acceptable non-ionic or ionic surfactants such as for instance sodium lauryl sulfate.
- the proton pump inhibitor optionally mixed with alkaline substances and further mixed with suitable constituents can be formulated into a core material.
- Said core material may be produced by extrusion/spheronization, balling or compression utilizing conventional process equipment.
- the size of the formulated core material is approximately between 0.1 and 4 mm and preferably between 0.1 and 2 mm.
- the manufactured core material can further be layered with additional ingredients comprising the proton pump inhibitor and/or be used for further processing.
- the proton pump inhibitor is mixed with pharmaceutical constituents to obtain preferred handling and processing properties and a suitable concentration of the substance in the final mixture.
- Pharmaceutical constituents such as fillers, binders, lubricants, disintegrating agents, surfactants and other pharmaceutically acceptable additives.
- the proton pump inhibitor may also be mixed with an alkaline, pharmaceutically acceptable substance (or substances).
- substances can be chosen among, but are not restricted to substances such as the sodium, potassium, calcium, magnesium and aluminium salts of phosphoric acid, carbonic acid, citric acid or other suitable weak inorganic or organic acids; aluminium hydroxide/sodium bicarbonate coprecipitate; substances normally used in antacid preparations such as aluminium, calcium and magnesium hydroxides; magnesium oxide or composite substances, such as Al 2 O 3 .6MgO.CO 2 .12H 2 O, (Mg 6 Al 2 (OH) 16 CO 3 .4H 2 O), MgO.Al 2 O 3 . 2SiO 2 .nH 2 O or similar compounds; organic pH-buffering substances such as trihydroxymethyl-aminomethane, basic amino acids and their salts or other similar, pharmaceutically acceptable pH-buffering substances.
- the aforementioned core material can be prepared by using spray drying or spray congealing technique.
- the pellets or tablets Before applying the enteric coating layer(s) onto the core material in the form of individual pellets or tablets, the pellets or tablets may optionally be covered with one or more separating layer(s) comprising pharmaceutical excipients optionally including alkaline compounds such as pH-buffering compounds.
- This/these separating layer(s) separate(s) the core material from the outer layers being enteric coating layer(s).
- the separating layer(s) protecting the core material of a proton pump inhibitor should be water soluble or rapidly disintegrating in water.
- the separating layer(s) can be applied to the core material by coating or layering procedures in suitable equipments such as coating pan, coating granulator or in a fluidized bed apparatus using water and/or organic solvents for the coating process.
- the separating layer(s) can be applied to the core material by using powder coating technique.
- the materials for the separating layers are pharmaceutically acceptable compounds such as, for instance, sugar, polyethylene glycol, polyvinylpyrrolidone, polyvinyl alcohol, polyvinyl acetate, hydroxypropyl cellulose, methylcellulose, ethyl-cellulose, hydroxypropyl methyl cellulose, carboxymethylcellulose sodium and others, used alone or in mixtures.
- Additives such as plasticizers, colorants, pigments, fillers anti-tacking and anti-static agents, such as for instance magnesium stearate, titanium dioxide, talc and other additives may also be included into the separating layer(s).
- the optional separating layer When the optional separating layer is applied to the core material it may constitute a variable thickness.
- the maximum thickness of the separating layer(s) is normally only limited by processing conditions.
- the separating layer may serve as a diffusion barrier and may act as a pH-buffering zone.
- the pH-buffering properties of the separating layer(s) can be further strengthened by introducing into the layer(s) substances chosen from a group of compounds usually used in antacid formulations such as, for instance, magnesium oxide, hydroxide or carbonate, aluminium or calcium hydroxide, carbonate or silicate; composite aluminium/magnesium compounds such as, for instance Al 2 O 3 .6MgO.CO 2 .12H 2 O, (Mg 6 Al 2 (OH) 16 CO 3 .4H 2 O), MgO.Al 2 O 3 .2SiO 2 .nH 2 O, aluminium hydroxide/sodium bicarbonate coprecipitate or similar compounds; or other pharmaceutically acceptable pH-buffering
- Talc or other compounds may be added to increase the thickness of the layer(s) and thereby strenghten the diffusion barrier.
- the optionally applied separating layer(s) is not essential for the invention. However, the separating layer(s) may improve the chemical stability of the active substance and/or the physical properties of the novel multiple unit tableted dosage form.
- the separating layer may be formed in situ by a reaction between an enteric coating polymer layer applied on the core material an alkaline reacting compound in the core material.
- the separating layer formed comprises a salt formed between the enteric coating layer polymer(s) and an alkaline reacting compound which is in the position to form a salt.
- enteric coating layers are applied onto the core material or onto the core material covered with separating layer(s) by using a suitable coating technique.
- the enteric coating layer material may be dispersed or dissolved in either water or in suitable organic solvents.
- enteric coating layer polymers one or more, separately or in combination, of the following can be used, e.g. solutions or dispersions of methacrylic acid copolymers, cellulose acetate phthalate, hydroxypropyl methylcellulose phthalate, hydroxypropyl methylcellulose acetate succinate, polyvinyl acetate phthalate, cellulose acetate trimellitate, carboxymethylethylcellulose, shellac or other suitable enteric coating polymer(s).
- the enteric coating layers may contain pharmaceutically acceptable plasticizers to obtain the desired mechanical properties, such as flexibility and hardness of the enteric coating layers.
- plasticizers are for instance, but not restricted to triacetin, citric acid esters, phthalic acid esters, dibutyl sebacate, cetyl alcohol, polyethylene glycols, polysorbates or other plasticizers.
- the amount of plasticizer is optimized for each enteric coating layer formula, in relation to selected enteric coating layer polymer(s), selected plasticizer(s) and the applied amount of said polymer(s), in such a way that the mechanical properties, i.e. flexibility and hardness of the enteric coating layer(s), for instance exemplified as Vickers hardness, are adjusted so that the acid resistance of the pellets covered with enteric coating layer(s) does not decrease significantly during compression of pellets into tablets.
- the amount of plasticizer is usually above 10 % by weight of the enteric coating layer polymer(s), preferably 15 - 50 % and more preferably 20 - 50 %.
- Additives such as dispersants, colorants, pigments polymers e.g.
- poly (ethylacrylat, methylmethacrylat), anti-tacking and anti-foaming agents may also be included into the enteric coating layer(s).
- Other compounds may be added to increase film thickness and to decrease diffusion of acidic gastric juices into the acid susceptible material.
- the enteric coating layer(s) constitutes a thickness of approximately at least 10 ⁇ m, preferably more than 20 ⁇ m.
- the maximum thickness of the applied enteric coating is normally limited by processing conditions and the desired dissolution profile.
- enteric coating layer described above may be used for enteric coating of conventional tablets comprising an acid susceptible proton pump inhibitor. Said enteric coating layered tablet is thereafter presscoated with a granulation comprising the prolanetic compound.
- Pellets covered with enteric coating layer(s) may further be covered with one or more over-coating layer(s).
- the over-coating layer(s) should be water soluble or rapidly disintegrating in water.
- the over-coating layer(s) can be applied to the enteric coating layered pellets by coating or layering procedures in suitable equipments such as coating pan, coating granulator or in a fluidized bed apparatus using water and/or organic solvents for the coating or layering process.
- the materials for over-coating layers are chosen among pharmaceutically acceptable compounds such as, for instance sugar, polyethylene glycol, polyvinylpyrrolidone, polyvinyl alcohol, polyvinyl acetate, hydroxypropyl cellulose, methylcellulose, ethylcellulose, hydroxypropyl methyl cellulose, carboxymethylcellulose sodium and others, used alone or in mixtures.
- pharmaceutically acceptable compounds such as, for instance sugar, polyethylene glycol, polyvinylpyrrolidone, polyvinyl alcohol, polyvinyl acetate, hydroxypropyl cellulose, methylcellulose, ethylcellulose, hydroxypropyl methyl cellulose, carboxymethylcellulose sodium and others, used alone or in mixtures.
- Additives such as plasticizers, colorants, pigments, fillers, anti-tacking and anti-static agents, such for instance magnesium stearate, titaniumdioxide, talc and other additives may also be included into the over-coating layer(s).
- Said over-coating layer may further prevent potential agglomeration of enteric coating layered pellets, further protect the enteric coating layer towards cracking during the compaction process and enhance the tableting process.
- the maximum thickness of the applied over-coating layer(s) is normally limited by processing conditions and the desired dissolution profile.
- over-coating layer may also be used as a tablet filmcoat to obtain tablets of good appearance.
- the active substance(s) in form of one or more prokinetic compound(s) is dry mixed with inactive excipients and the mixture is wet massed with a granulation liquid.
- the wet mass is dried preferably to a loss on drying of less than 3% by weight. Thereafter the dry mass is milled to a suitable size for the granules, such as smaller than 4 mm, and preferably smaller than 1 mm.
- suitable inactive excipients for the prokinetic mixture are for instance lactose, corn starch low substituted hydroxypropyl cellulose, microciystalline cellulose, sodium starch glycolate and crosslinked polyvinyl pyrrolidone.
- the dry mixture comprising prokinetic compound is wet-mixed with a suitable granulation liquid comprising for instance hydroxy propyl cellulose or polyvinyl pyrrolidone dissolved in purified water or an alcohol or a mixture thereof.
- a suitable granulation liquid comprising for instance hydroxy propyl cellulose or polyvinyl pyrrolidone dissolved in purified water or an alcohol or a mixture thereof.
- the prokinetic agent(s) are dry mixed with pharmaceutically acceptable excipients according to above.
- the prokinetic agent(s) can be applied in a separate layer onto a multiple unit tableted dosage form or surrounding the tablet comprising the proton pump inhibitor.
- the prokinetic agent(s) is dispersed or dissolved in an aqueous solution optionally comprising binders for suspension layering onto the tablet.
- the enteric coating layered pellets comprising a proton pump inhibitor are mixed with the granules comprising prokinetic compound and tablet excipients such as fillers, binders, disintegrants, lubricants and other pharmaceutically acceptable additives.
- the mixture is compressed into a multiple unit tableted dosage form.
- the compressed tablet is optionally covered with a filmforming agent(s) to obtain a smooth surface of the tablet and further enhance the stability of the tablet during packaging and transport
- Such a coating layer may further comprise additives such as anti-tacking agents, colorants and pigments or other additives to obtain a tablet of good appearance.
- enteric coated pellets may be dry mixed with the prokinetic compound and pharmaceutically acceptable tablet excipients according to above, and compressed into tablets (direct compression).
- Suitable lubricants for the tableting process are for instance sodium stearyl fumarate, magnesium stearate and talc.
- the different active substances may be formulated into different layers, wherein the layer comprising the proton pump inhibitor is in the form of a multiple unit tableted dosage form layered with prepared prokinetic granules.
- the two layers may be separated by an anti-tacking layer.
- the proton pump inhibitor is dry mixed with inactive excipients and compressed into a conventional tablet which is coating layered with an enteric coating and optionally a separating layer is applied before the enteric coating. Thereafter the enteric coated tablet is presscoated with a prokinetic preparation.
- the tablet core may also be formulated as a multiple unit tableted dosage form comprising the proton pump inhibitor, the tablet is spray coating layered by a suspension comprising the prokinetic agent(s).
- the fraction of enteric coating layered pellets constitutes less than 75 % by weight of the total tablet weight and preferably less than 60 %.
- the fraction of enteric coating layered pellets of the proton pump inhibitor may be reduced in the multiple unit tableted dosage form.
- the preferred multiple unit tablet formulation consists of enteric coating layered pellets containing one active substance in the form of a proton pump inhibitor, optionally admixed with alkaline reacting compound(s), compressed into tablets together with the prepared prokinetic mixture and optionally tablet excipients.
- a proton pump inhibitor optionally admixed with alkaline reacting compound(s)
- alkaline reacting compound(s) optionally admixed with alkaline reacting compound(s)
- the enteric coating layer(s) is making the pellets of the dosage form insoluble in acidic media, but disintegrating/dissolving in near neutral to alkaline media such as, for instance the liquids present in the proximal part of the small intestine, where dissolution of the proton pump inhibitor is desired.
- the prokinetic agent(s) may be released in the stomach.
- the enteric coating layered pellets may further be covered with an overcoating layer before being formulated into the tablet and they may also contain one or more separating layer(s) optionally containing alkaline substance(s).
- the process for the manufacture of the dosage form represents a further aspect of the invention.
- the pellets After formulation of the pellets by spray coating or layering of the proton pump inhibitor onto seeds, or by extrusion/spheronization or granulation, e.g. rotor granulation of homogeneous pellets, the pellets are first optionally covered with the separating layer(s) and then with the enteric coating layer(s) or a separating layer is spontaneously developed in situ between the alkaline core material and the enteric coating layer material.
- the coating is carried out as described above and in the accompanying examples.
- the preparation of the prokinetic mixture is also described above and in the examples.
- the pharmaceutical processes can preferably be completely water-based.
- the enteric coating layered pellets are mixed with the prepared prokinetic mixture, optionally tablet excipients and other pharmaceutically acceptable additives and compressed into tablets.
- the enteric coating layered pellets may be intimately mixed with tablet excipients and precompressed and further layered with the prepared prokinetic mixture and finally compressed into a tablet.
- the proton pump inhibitor in form of the active substance may be mixed with tablet excipients and compressed into a tablet which is optionally layered with a separating layer and thereafter enteric coating layered. Said tablet is then presscoated with the prepared prokinetic mixture.
- a multiple unit tableted dosage form of the proton pump inhibitor is manufactured as describes above.
- the multiple unit dosage form is spray coating layered by an aqueous suspension comprising the prokinetic agent(s).
- the suspension may optionally comprise binders; such as hydroxypropyl methylcellulose, and an alcohol to solve the binder.
- the proton pump inhibitor in the form of enteric coating layered pellets may also be filled into a capsule together with the prokinetic substance in the form of a granulation optionally mixed with pharmaceutical excipients.
- the dosage forms according to the invention are especially advantageous in the treatment of gastro oesophageal reflux disease and other gastrointestinal disorder. They are administered one to several times a day, preferably once or twice daily.
- the typical daily dose of the active substances varies and will depend on various factors such as the individual requirements of the patients, the mode of administration and disease.
- each dosage form will comprise 0.1-200 mg of the proton pump inhibitor and 0.1-100 mg of the prokinetic compound.
- each dosage form will comprise 10-80 mg of the proton pump inhibitor and 3-80 mg of the prokinetic compound, and more preferably 10-40 mg of proton pump inhibitor and 15 - 40 mg of the prokinetic compound, respectively.
- the multiple unit tablet preparation is also suitable for dispersion in an aqueous liquid with slightly acidic pH-value before being orally administered or fed through a naso-gastric tube.
- Multiple unit dosage form comprising magnesium omeprazole and mosapride (batch size 500 tablets).
- Core material Magnesium omeprazole 5 kg Sugar sphere seeds 10 kg Hydroxypropyl methylcellulose 0.75 kg Water purified 20.7 kg Separating layer Core material (acc. to above) 10.2 kg Hydroxypropyl cellulose 1.02 kg Talc 1.75 kg Magnesium stearate 0.146kg Water purified 21.4 kg Enteric coating layer Pellets covered with separating layer (acc.
- Suspension layering was performed in a fluid bed apparatus. Magnesium omeprazole was sprayed onto sugar sphere seeds from a water suspension containing the dissolved binder. The size of sugar sphere seeds were in the range of 0.25 to 0.35 mm.
- the prepared core material was covered with a separating layer in a fluid bed apparatus with a hydroxypropyl cellulose solution containing talc and magnesium stearate.
- the enteric coating layer consisting of methacrylic acid copolymer, mono- and diglycerides, triethyl citrate and polysorbate was sprayed onto the pellets covered with a separating layer in a fluid bed apparatus.
- enteric coating layered pellets were coated with a hydroxypropyl methylcellulose solution containing magnesium stearate.
- the over-coating layered pellets were classified by sieving.
- the enteric coating layered pellets with an over-coating layer, mosapride citrate dihydrate, microcrystalline cellulose, polyvinyl pyrrolidone crosslinked and sodium stearyl fumarate were dry mixed and compressed into tablets using an excenter tableting machine equipped with 12 mm punches.
- the amount of omeprazole in each tablet was approx. 10 mg and the amount of mosapride was approx. 30 mg.
- the tablet hardness was measured to 70-80 N.
- the obtained tablets are covered with a conventional tablet filmcoating layer.
- Multiple unit dosage form comprising magnesium omeprazole and mosapride (batch size 500 tablets).
- Mosapride granulation Mosapride citrate dihydrate 46.8 g Lactose monohydrate 350 g Corn starch 184 g Hydroxy propyl cellulose LF 25 g Water purified 225 g Hydroxypropyl cellulose (L-HPC) 152 g Magnesium stearate 7.4 g Tablets Enteric coating layered pellets with an over-coating layer (manufacturing and composition as in example 1) 41.2 g Mosapride granulation 190 g Tablet coating solution (for 10 kg tablets) Hydroxypropyl methyl cellulose 250 g Polyethylene glycol 6000 62.5 g Titanium dioxid 62.5 g Water purified 2125 g Hydrogen peroxide 0.75g
- Hydroxypropyl cellulose was dissolved in purified water to form the granulation liquid.
- Mosapride citrate dehydrate, lactose monohydrate and corn starch were dry mixed.
- the granulation liquid was added to the powder mixture and the mass was wet-mixed.
- the wet mass was dried in a steam-oven and milled through sive 1 mm in an oscillating mill equipment.
- the prepared granulation was mixed with low substituted hydroxypropyl cellulose and magnesium stearate.
- the enteric coating layered pellets with an over-coat and prepared granules were mixed and compressed into tablets using an excenter tableting machine equipped with 11 mm punches.
- the amount of omeprazole in each tablet was approx. 10 mg and the amount of mosapride was approx. 15 mg. Tablet hardness was measured to 30 - 40 N.
- the obtained tablets are covered with a conventional tablet filmcoating layer.
- Multiple unit dosage form comprising magnesium omeprazole and mosapride (batch size 500 tablets).
- Core material Magnesium omeprazole 10 kg Sugar sphere seeds 10 kg Hydroxypropyl methylcellulose 1.5 kg Water purified 29.9 kg Separating layer Core material (acc. to above) 20 kg Hydroxypropyl cellulose 2 kg Talc 3.43 kg Magnesium stearate 0.287kg Water purified 41 kg Enteric coating layer Pellets covered with separating layer (acc.
- the enteric coating layered pellets with an over-coating layer prepared as described in Example 1, mosapride citrate dihydrate, microcrystalline cellulose, polyvinyl pyrrolidone crosslinked and sodium stearyl fumarate were dry mixed and compressed into tablets using an excenter tableting machine equipped with 12 mm punches.
- the amount of omeprazole in each tablet was approx. 20 mg and the amount of mosapride was approx. 30 mg.
- the tablet hardness was measured to 70 N.
- the tablets are covered with a conventional tablet filmcoating layer.
- S-omeprazole magnesium salt and mosapride (batch size 300 tablets).
- Core material S-omeprazole magnesium salt 120 g Sugar sphere seeds 150 g Hydroxypropyl methylcellulose 18 g Polysorbate 80 2.4 g Water purified 562 g Separating layer Core material (acc. to above) 200 g Hydroxypropyl cellulose 30 g Talc 51.4 g Magnesium stearate 4.3 g Water purified 600 g Enteric coating layer Pellets covered with separating layer (acc.
- Suspension layering was performed in a fluid bed apparatus.
- S-Omeprazole magnesium salt was sprayed onto sugar sphere seeds from a water suspension containing the dissolved binder and polysorbate 80.
- the size of sugar sphere seedes were in the range of 0.25 to 0.35 mm.
- the prepared core material was covered with a separating layer in a fluid bed apparatus with hydroxypropyl cellulose solution containing talc and magnesium stearate.
- the enteric coating layer consisting of methacrylic acid copolymer, mono-and diglycerides, triethyl citrate and polysorbate was sprayed onto the pellets covered with a separating layer in a fluid bed apparatus.
- the enteric coating layered pellets were classified by sieving.
- the enteric coating layered pellets, mosapride citrate dihydrate, microcrystalline cellulose, polyvinyl pyrrolidone crosslinked and sodium stearyl fumarate were mixed and compressed into tablets using an excenter tableting machine equipped with 12mm punches.
- the amount of S-omeprazole in each tablet was approx. 20 mg and the amount of mosapride was approx. 30 mg.
- the tablet hardness was measured to 65 N.
- Multiple unit dosage form comprising lanzoprazole and mosapride (batch size 500 tablets).
- Core material Lanzoprazole 400 g Sugar sphere seeds 400 g Hydroxypropyl methylcellulose 80 g Sodium laurylsulfate 3 g Water purified 1500 g Separating layer Core material (acc. to above) 400 g Hydroxypropyl cellulose 40 g Talc 69 g Magnesium stearate 6 g Water purified 800 g Enteric coating layer Pellets covered with a separating layer (acc.
- Suspension layering was performed in a fluid bed apparatus. Lansoprazole was sprayed onto the sugar sphere seeds from a suspension containing the dissolved binder in a water solution. Pellets covered with separating layer and enteric coating layer were produced as in example 1.
- enteric coating layered pellets, mosapride citrate dihydrate, microcrystalline cellulose, polyvinyl pyriolidone crosslinked and sodium stearyl fumarate were dry mixed and compressed into tablets using an excenter tableting machine equipped with 10 mm punches.
- the amount of lanzoprazole in each tablet was approx. 10 mg and the amount of mosapride was approx. 30 mg.
- the tablet hardness was measured to 70 N.
- the tablets are covered with a conventional tablet filmcoating layer.
- Magnesium omeprazole and mosapride presscoated tablets (batch size 10.000 tablets).
- Omeprazole tablets Mg-omeprazole 112.5 g Mannitol 287 g Microcrystalline cellulose 94 g Sodium starch glycolate 30 g Hydroxypropyl methylcellulose 30 g Talc 25 g Microcrystalline cellulose 31 g Sodium stearyl fumarate 12.5 g Water purified 200 g Solution for separating layer (for 10 kg tablets) Hydroxypropyl methylcellulose 300 g Hydrogen peroxide (30%) 0.003 g Water purified 2700 g Solution for enteric coating layer (for 10 kg tablets) Methacrylic acid copolymer dispersion (30%) 2450 g Polyethylene glycol 400 80 g Titanium dioxide Colour 100 g Water purified 1960 g Presscoated tablet Mg-Omeprazole tablets 10.000 tabl Mosapride granulation (manufacturing and composition as in example 2) 3800 g Tablet coating solution (for 10 kg tablets) Hydr
- Magnesium omeprazole, mannitol, microcrystalline cellulose, sodium starch glycolate and hydroxypropyl methyl cellulose are dry mixed.
- the powder mixture is moistened with water purified.
- the granulation is dried and milled through sive 1 mm in a suitable mill.
- the prepared granules comprising proton pump inhibitor is mixed with talc, microcrystalline cellulose and sodium stearyl fumarate and compressed into tablets using a rotary tableting machine equipped with 5 mm punches.
- the obtained tablets are coated layered with a separating layer and an enteric coating layer. Said tablets are then presscoated with mosapride granulation using a presscoating machine equipped with 11 mm punched.
- the tablets are covered with a conventional tablet filmcoating layer.
- a capsule formulation comprising magnesium omeprazole and mosapride (batch size 100 capsules).
- Capsules Enteric coating layered pellets with an over-coating layer (manufacturing and composition as in example 3)
- Mosapride granulation manufactured and composition as in example 2
- Enteric coating layered pellets and mosapride granulation are filled into capsules, size 00.
- the amount of omeprazole in each capsule is approx. 20 mg and the amount of mosapride is approx. 15 mg.
- Multiple unit dosage form comprising magnesium omeprazole with a tablet coating layer comprising mosapride (batch size 1 000 tablets).
- Tablets Enteric coating layered pellets with an overcoat (manufacturing and composition as in example 1) 82.4 g Microcrystalline cellulose 179.2 g Polyvinyl pyrrolidone crosslinked 3.7 g Sodium stearyl fumarate 0.4 g Mosapride coating layer suspension Mosapride citrate dihydrate 23.4 g Hydroxypropyl methyl cellulose 13.4 g Ethanol 99 % 132.5 g Water purified 132.5 g Tablet coating solution (for 10 kg tablets) Hydroxypropyl methylcellulose 250 g Polyethylene glycol 6000 62.5 g Titanium dioxid 62.5 g Water purified 2125 g Hydrogen peroxide 0.75g
- the enteric coating layered pellets are mixed with microcrystalline cellulose, polyvidone and sodium stearyl fumarate and compressed into tablets using an excenter tableting machine equipped with 9 mm punches.
- the tablets are then coated layered in a fluid bed apparatus with the suspension comprising mosapride.
- the amount of omeprazole in each tablet is approx. 10 mg and the amount of mosapride is approx. 15 mg.
- the enteric coating layered pellets comprising a proton pump inhibitor may also be prepared as described in the following examples.
- Sodium lauryl sulphate is dissolved in purified water to form the granulation liquid.
- Magnesium omeprazole, mannitol, microcrystalline cellulose and hydroxypropyl cellulose are dry-mixed.
- the granulation liquid is added to the powder mixture and the mass is wet-mixed.
- the wet mass is forced through an extruder equipped with screens of size 0.5 mm.
- the extrudate is spheronized on a friction plate in a spheronizing apparatus.
- the core material is dried in a fluid bed dryer and classified.
- the prepared core material is covered by a separating layer in a fluid bed apparatus with a hydroxypropyl methylcellulose/water solution.
- the enteric coating layer is applied to the pellets covered with separating layer from an aqueous dispersion of methacrylic acid copolymer plasticized with triethyl citrate to which a mono- and diglycerides/polysorbate dispersion has been added.
- the pellets are dried in a fluid bed apparatus.
- Magnesium omeprazole, part of the hydroxypropyl methylcellulose and Aerosil® are dry-mixed forming a powder.
- Sugar sphere seeds (0.25-0.40 mm) are layered with the powder in a centrifugal fluidized coating granulator while spraying a hydroxypropyl methylcellulose solution (6 %, w/w).
- the prepared core material is dried and covered by a separating layer in a centrifugal fluidized coating-granulator.
- a fluid bed apparatus is used for enteric coating layereing.
- the prepared core material is covered with a separating layer in a fluid bed apparatus with a hydroxypropyl methylcellulose solution.
- the enteric coating layer consisting of methacrylic acid copolymer, mono- and diglycerides, polyethylene glycol 400 and polysorbate is sprayed onto the pellets covered with separating layer in a fluid bed apparatus.
- enteric coating layer Pellets covered with separating layer (manufacturing and composition as in example 10) 500 g Methacrylic acid copolymer 250 g Polyethylene glycol 6000 75 g Mono- and diglycerides (NF) 12.5 g Polysorbate 80 1.2 g Water purified 490 g
- enteric coating layered pellets Preparation of enteric coating layered pellets.
- Enteric coating Pellets covered with separating layer 500 g Hydroxypropyl methylcellulose phthalate 250 g Cetanol 50 g Ethanol (95%) 1000 g Acetone 2500 g
- the dry ingredients for producing the core material are well mixed in a mixer. Addition of granulation liquid is made and the mixture is kneeded and granulated to a proper consistency. The wet mass is pressed through an extruder screen and the granules are converted into a spherical form in a spheronizer.
- the core material is dried in a fluid bed apparatus and classified into a suitable particle size range, e.g. 0.5 - 1.0 mm.
- the prepared core material is covered with a separating layer and enteric coating layered as described in previous examples.
- omeprazole used in some of the examples is produced according to the process described in WO95/01977, the single enantiomers of omeprazole salts are prepared as described in WO94/27988 and omeprazole is produced according to the process disclosed in EP-A1 0005129.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Medicinal Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Pharmacology & Pharmacy (AREA)
- Epidemiology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Organic Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- General Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Medicinal Preparation (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| SE9600072A SE9600072D0 (sv) | 1996-01-08 | 1996-01-08 | New oral formulation of two active ingredients II |
| SE9600072 | 1996-01-08 | ||
| PCT/SE1996/001736 WO1997025065A1 (en) | 1996-01-08 | 1996-12-20 | Oral pharmaceutical dosage forms comprising a proton pump inhibitor and a prokinetic agent |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| EP0814840A1 EP0814840A1 (en) | 1998-01-07 |
| EP0814840B1 true EP0814840B1 (en) | 2002-11-20 |
Family
ID=20400970
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP96944725A Expired - Lifetime EP0814840B1 (en) | 1996-01-08 | 1996-12-20 | Oral pharmaceutical dosage forms comprising a proton pump inhibitor and a prokinetic agent |
Country Status (29)
| Country | Link |
|---|---|
| US (1) | US6132771A (xx) |
| EP (1) | EP0814840B1 (xx) |
| JP (1) | JPH11501949A (xx) |
| KR (1) | KR100486057B1 (xx) |
| CN (1) | CN1142792C (xx) |
| AR (1) | AR008750A1 (xx) |
| AT (1) | ATE228010T1 (xx) |
| AU (1) | AU712572B2 (xx) |
| BR (1) | BR9607344A (xx) |
| CA (1) | CA2214033C (xx) |
| CZ (1) | CZ280897A3 (xx) |
| DE (1) | DE69624910T2 (xx) |
| DK (1) | DK0814840T3 (xx) |
| EE (1) | EE9700195A (xx) |
| ES (1) | ES2185816T3 (xx) |
| HU (1) | HUP9904094A3 (xx) |
| ID (1) | ID18383A (xx) |
| IL (1) | IL121652A (xx) |
| IS (1) | IS4550A (xx) |
| MX (1) | MX9706769A (xx) |
| NO (1) | NO316425B1 (xx) |
| NZ (1) | NZ325976A (xx) |
| PL (1) | PL322176A1 (xx) |
| PT (1) | PT814840E (xx) |
| SE (1) | SE9600072D0 (xx) |
| SK (1) | SK117097A3 (xx) |
| TR (1) | TR199700917T1 (xx) |
| WO (1) | WO1997025065A1 (xx) |
| ZA (1) | ZA9610938B (xx) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN1993132B (zh) * | 2004-07-28 | 2010-05-05 | 大日本住友制药株式会社 | 具有多包衣层的膜-包衣片剂 |
Families Citing this family (101)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5840737A (en) | 1996-01-04 | 1998-11-24 | The Curators Of The University Of Missouri | Omeprazole solution and method for using same |
| US6489346B1 (en) | 1996-01-04 | 2002-12-03 | The Curators Of The University Of Missouri | Substituted benzimidazole dosage forms and method of using same |
| US6699885B2 (en) | 1996-01-04 | 2004-03-02 | The Curators Of The University Of Missouri | Substituted benzimidazole dosage forms and methods of using same |
| US6645988B2 (en) | 1996-01-04 | 2003-11-11 | Curators Of The University Of Missouri | Substituted benzimidazole dosage forms and method of using same |
| SE9600072D0 (sv) * | 1996-01-08 | 1996-01-08 | Astra Ab | New oral formulation of two active ingredients II |
| SE510650C2 (sv) * | 1997-05-30 | 1999-06-14 | Astra Ab | Ny förening |
| ES2137862B1 (es) * | 1997-07-31 | 2000-09-16 | Intexim S A | Preparacion farmaceutica oral que comprende un compuesto de actividad antiulcerosa y procedimiento para su obtencion. |
| US6096340A (en) | 1997-11-14 | 2000-08-01 | Andrx Pharmaceuticals, Inc. | Omeprazole formulation |
| JP4546643B2 (ja) * | 1997-12-08 | 2010-09-15 | ニコメッド ゲゼルシャフト ミット ベシュレンクテル ハフツング | 酸不安定な活性化合物を含有する新規の坐剤形 |
| DK173431B1 (da) * | 1998-03-20 | 2000-10-23 | Gea Farmaceutisk Fabrik As | Farmaceutisk formulering omfattende en 2-[[(2-pyridinyl)methyl]sulfinyl]benzimidazol med anti-ulcusaktivitet samt fremgangs |
| US6328994B1 (en) | 1998-05-18 | 2001-12-11 | Takeda Chemical Industries, Ltd. | Orally disintegrable tablets |
| TWI257311B (en) | 1998-07-28 | 2006-07-01 | Takeda Chemical Industries Ltd | Rapidly disintegrable solid preparation |
| SE9803772D0 (sv) * | 1998-11-05 | 1998-11-05 | Astra Ab | Pharmaceutical formulation |
| WO2000059314A1 (en) * | 1999-04-01 | 2000-10-12 | Wm. Wrigley Jr. Company | Long flavor duration releasing structures for chewing gum |
| US6428809B1 (en) * | 1999-08-18 | 2002-08-06 | Microdose Technologies, Inc. | Metering and packaging of controlled release medication |
| US6521255B2 (en) * | 2000-01-13 | 2003-02-18 | Osmotica Corp. | Osmotic device containing ranitidine and a prokinetic agent |
| US6667042B2 (en) | 2000-02-24 | 2003-12-23 | Advancis Pharmaceutical Corp. | Fluroquinilone antibiotic product, use and formulation thereof |
| US6663891B2 (en) | 2000-02-24 | 2003-12-16 | Advancis Pharmaceutical Corp. | Erythromyacin antibiotic product, use and formulation thereof |
| US6638532B2 (en) | 2000-02-24 | 2003-10-28 | Advancis Pharmaceutical Corp. | Tetracycline—doxycycline antibiotic composition |
| US6669948B2 (en) | 2000-02-24 | 2003-12-30 | Advancis Pharmaceutical Corp. | Antibiotic product, use and formulation thereof |
| US6663890B2 (en) | 2000-02-24 | 2003-12-16 | Advancis Pharmaceutical Corp. | Metronidazole antibiotic product, use and formulation thereof |
| US7025989B2 (en) * | 2000-02-24 | 2006-04-11 | Advancis Pharmaceutical Corp. | Multiple-delayed released antibiotic product, use and formulation thereof |
| US6632453B2 (en) | 2000-02-24 | 2003-10-14 | Advancis Pharmaceutical Corp. | Ciprofoxacin-metronidazole antibiotic composition |
| US6627222B2 (en) | 2000-02-24 | 2003-09-30 | Advancis Pharmaceutical Corp. | Amoxicillin-dicloxacillin antibiotic composition |
| US6667057B2 (en) | 2000-02-24 | 2003-12-23 | Advancis Pharmaceutical Corp. | Levofloxacin antibiotic product, use and formulation thereof |
| US6610328B2 (en) | 2000-02-24 | 2003-08-26 | Advancis Pharmaceutical Corp. | Amoxicillin-clarithromycin antibiotic composition |
| US6623758B2 (en) | 2000-02-24 | 2003-09-23 | Advancis Pharmaceutical Corp. | Cephalosporin-metronidazole antibiotic composition |
| WO2001062195A1 (en) | 2000-02-24 | 2001-08-30 | Advancis Pharmaceutical Corporation | Antibiotic and antifungal compositions |
| US6991807B2 (en) * | 2000-02-24 | 2006-01-31 | Advancis Pharmaceutical, Corp. | Antibiotic composition |
| US6544555B2 (en) | 2000-02-24 | 2003-04-08 | Advancis Pharmaceutical Corp. | Antibiotic product, use and formulation thereof |
| US6565882B2 (en) | 2000-02-24 | 2003-05-20 | Advancis Pharmaceutical Corp | Antibiotic composition with inhibitor |
| US6730320B2 (en) | 2000-02-24 | 2004-05-04 | Advancis Pharmaceutical Corp. | Tetracycline antibiotic product, use and formulation thereof |
| GEP20043285B (en) | 2000-03-30 | 2004-07-26 | Bristol Myers Squibb Co | Sustained Release Beadlets Containing Stavudine, Method for Their Production, Pharmaceutical Composition Containing the Same and Use Thereof for Treatment Retroviral Infections |
| US20040146554A1 (en) * | 2000-07-15 | 2004-07-29 | Sharma Virender K. | Method for the administration of acid-labile drugs |
| US20020064555A1 (en) * | 2000-09-29 | 2002-05-30 | Dan Cullen | Proton pump inhibitor formulation |
| US7105174B2 (en) * | 2000-10-13 | 2006-09-12 | Advancis Pharmaceutical Corporation | Multiple-delayed release anti-neoplastic product, use and formulation thereof |
| US7074417B2 (en) * | 2000-10-13 | 2006-07-11 | Advancis Pharmaceutical Corporation | Multiple-delayed release anti-viral product, use and formulation thereof |
| US7157095B2 (en) * | 2000-10-13 | 2007-01-02 | Advancis Pharmaceutical Corporation | Multiple-delayed release antifungal product, use and formulation thereof |
| US6541014B2 (en) | 2000-10-13 | 2003-04-01 | Advancis Pharmaceutical Corp. | Antiviral product, use and formulation thereof |
| US20020197314A1 (en) | 2001-02-23 | 2002-12-26 | Rudnic Edward M. | Anti-fungal composition |
| SE0101379D0 (sv) * | 2001-04-18 | 2001-04-18 | Diabact Ab | Komposition som hämmar utsöndring av magsyra |
| US8206741B2 (en) | 2001-06-01 | 2012-06-26 | Pozen Inc. | Pharmaceutical compositions for the coordinated delivery of NSAIDs |
| JP2006282677A (ja) * | 2001-10-17 | 2006-10-19 | Takeda Chem Ind Ltd | 酸に不安定な薬物の高含量顆粒 |
| JP4331930B2 (ja) * | 2001-10-17 | 2009-09-16 | 武田薬品工業株式会社 | 酸に不安定な薬物の高含量顆粒 |
| US8105626B2 (en) | 2001-10-17 | 2012-01-31 | Takeda Pharmaceutical Company Limited | Granules containing acid-unstable chemical in large amount |
| MXPA04007169A (es) * | 2002-01-25 | 2004-10-29 | Santarus Inc | Suministro transmucosal de inhibidores de bomba de protones. |
| US7125563B2 (en) * | 2002-04-12 | 2006-10-24 | Dava Pharmaceuticals, Inc. | Sustained release pharmaceutical preparations and methods for producing the same |
| US20030228363A1 (en) * | 2002-06-07 | 2003-12-11 | Patel Mahendra R. | Stabilized pharmaceutical compositons containing benzimidazole compounds |
| SE0203065D0 (sv) * | 2002-10-16 | 2002-10-16 | Diabact Ab | Gastric acid secretion inhibiting composition |
| MY148805A (en) * | 2002-10-16 | 2013-05-31 | Takeda Pharmaceutical | Controlled release preparation |
| US9107804B2 (en) * | 2002-12-10 | 2015-08-18 | Nortec Development Associates, Inc. | Method of preparing biologically active formulations |
| US20040114368A1 (en) * | 2002-12-13 | 2004-06-17 | Shyu Shing Jy | Light device having rotatable or movable view |
| EP1596838A2 (en) * | 2003-02-11 | 2005-11-23 | Torrent Pharmaceuticals Ltd | Once a day orally administered pharmaceutical compositions comprising a proton pump inhibitor and a prokinetic agent |
| US20040185119A1 (en) * | 2003-02-26 | 2004-09-23 | Theuer Richard C. | Method and compositions for treating gastric hyperacidity while diminishing the likelihood of producing vitamin deficiency |
| BR8300543U (pt) * | 2003-04-09 | 2004-11-23 | Medley S A Ind Farmaceutica | Embalagem, ou kit de medicamentos, ou apresentação facilitadora da administração de medicamentos por parte dos pacientes |
| US8993599B2 (en) | 2003-07-18 | 2015-03-31 | Santarus, Inc. | Pharmaceutical formulations useful for inhibiting acid secretion and methods for making and using them |
| AU2004258949B2 (en) | 2003-07-21 | 2011-02-10 | Shionogi, Inc. | Antibiotic product, use and formulation thereof |
| CA2533292C (en) | 2003-07-21 | 2013-12-31 | Advancis Pharmaceutical Corporation | Antibiotic product, use and formulation thereof |
| AU2004258944B2 (en) | 2003-07-21 | 2011-02-10 | Shionogi, Inc. | Antibiotic product, use and formulation thereof |
| WO2005011637A1 (ja) * | 2003-08-04 | 2005-02-10 | Eisai Co., Ltd. | 用時分散型製剤 |
| JP2007502296A (ja) | 2003-08-11 | 2007-02-08 | アドバンシス ファーマスーティカル コーポレイション | ロバストペレット |
| US8062672B2 (en) | 2003-08-12 | 2011-11-22 | Shionogi Inc. | Antibiotic product, use and formulation thereof |
| AU2004268641A1 (en) * | 2003-08-29 | 2005-03-10 | Dynogen Pharmaceuticals, Inc. | Compositions useful for treating gastrointestinal motility disorders |
| WO2005023184A2 (en) * | 2003-08-29 | 2005-03-17 | Advancis Pharmaceuticals Corporation | Antibiotic product, use and formulation thereof |
| WO2005027877A1 (en) | 2003-09-15 | 2005-03-31 | Advancis Pharmaceutical Corporation | Antibiotic product, use and formulation thereof |
| TWI372066B (en) * | 2003-10-01 | 2012-09-11 | Wyeth Corp | Pantoprazole multiparticulate formulations |
| US7608674B2 (en) * | 2003-11-03 | 2009-10-27 | Ilypsa, Inc. | Pharmaceutical compositions comprising cross-linked small molecule amine polymers |
| US7767768B2 (en) * | 2003-11-03 | 2010-08-03 | Ilypsa, Inc. | Crosslinked amine polymers |
| US7335795B2 (en) | 2004-03-22 | 2008-02-26 | Ilypsa, Inc. | Crosslinked amine polymers |
| JP4740740B2 (ja) * | 2003-12-09 | 2011-08-03 | 大日本住友製薬株式会社 | 薬物含有粒子および該粒子を含む固形製剤 |
| RS20050796A (xx) * | 2004-01-06 | 2007-08-03 | Panacea Biotec Ltd., | Farmaceutske smeše koje sadrže inhibitor protonske pumpe i prokinetičko sredstvo |
| US8906940B2 (en) | 2004-05-25 | 2014-12-09 | Santarus, Inc. | Pharmaceutical formulations useful for inhibiting acid secretion and methods for making and using them |
| US8815916B2 (en) | 2004-05-25 | 2014-08-26 | Santarus, Inc. | Pharmaceutical formulations useful for inhibiting acid secretion and methods for making and using them |
| WO2005123042A1 (en) * | 2004-06-10 | 2005-12-29 | Glatt Air Techniques, Inc. | Controlled release pharmaceutical formulation |
| WO2006014427A1 (en) | 2004-07-02 | 2006-02-09 | Advancis Pharmaceutical Corporation | Tablet for pulsed delivery |
| AR051654A1 (es) * | 2004-11-04 | 2007-01-31 | Astrazeneca Ab | Nuevas formulaciones de pellets de liberacion modificada para inhibidores de la bomba de protones |
| AR052225A1 (es) * | 2004-11-04 | 2007-03-07 | Astrazeneca Ab | Formulaciones de tabletas de liberacion modificada par inhibidores de la bomba de protones |
| WO2006066932A1 (en) * | 2004-12-24 | 2006-06-29 | Lek Pharmaceuticals D.D. | Stable pharmaceutical composition comprising an active substance in the form of solid solution |
| US8357394B2 (en) | 2005-12-08 | 2013-01-22 | Shionogi Inc. | Compositions and methods for improved efficacy of penicillin-type antibiotics |
| US8778924B2 (en) | 2006-12-04 | 2014-07-15 | Shionogi Inc. | Modified release amoxicillin products |
| CN101405004A (zh) * | 2006-01-20 | 2009-04-08 | 大日本住友制药株式会社 | 新型薄膜衣片剂 |
| EP2012756A4 (en) * | 2006-04-20 | 2013-01-23 | Inventia Healthcare Private Ltd | MULTI-UNIT COMPOSITIONS |
| US8299052B2 (en) | 2006-05-05 | 2012-10-30 | Shionogi Inc. | Pharmaceutical compositions and methods for improved bacterial eradication |
| AU2007266574A1 (en) * | 2006-06-01 | 2007-12-06 | Dexcel Pharma Technologies Ltd. | Multiple unit pharmaceutical formulation |
| US8439033B2 (en) | 2007-10-09 | 2013-05-14 | Microdose Therapeutx, Inc. | Inhalation device |
| CA2736547C (en) * | 2008-09-09 | 2016-11-01 | Pozen Inc. | Method for delivering a pharmaceutical composition to patient in need thereof |
| JP2012518655A (ja) * | 2009-02-23 | 2012-08-16 | アプタリス ファーマテック インコーポレイテッド | プロトンポンプ阻害剤を含む制御放出組成物 |
| US8758779B2 (en) * | 2009-06-25 | 2014-06-24 | Wockhardt Ltd. | Pharmaceutical composition of duloxetine |
| AU2010263304A1 (en) | 2009-06-25 | 2012-02-02 | Astrazeneca Ab | Method for treating a patient at risk for developing an NSAID-associated ulcer |
| WO2011063150A2 (en) * | 2009-11-20 | 2011-05-26 | Handa Pharmaceuticals, Llc | Oral formulation for dexlansoprazole |
| CA2784041C (en) | 2010-01-05 | 2017-11-07 | Microdose Therapeutx, Inc. | Inhalation device and method |
| KR101143997B1 (ko) * | 2010-03-04 | 2012-05-09 | 한미사이언스 주식회사 | 유산균 제제 및 5-ht4 수용체 효능제를 함유하는 경구 투여용 복합 제제 및 이의 제조방법 |
| WO2011126327A2 (en) * | 2010-04-09 | 2011-10-13 | Hyundai Pharm Co., Ltd. | Pharmaceutical composition with controlled-release properties comprising mosapride or levodropropizine, and preparing method thereof |
| EP2563341A1 (en) * | 2010-04-26 | 2013-03-06 | Mahmut Bilgic | Pharmaceutical compositions inducing synergistic effect |
| BRPI1103093A2 (pt) * | 2011-06-03 | 2013-07-02 | Eurofarma Laboratarios Ltda | composiÇço farmacÊutica oral e uso da composiÇço farmacÊutica oral |
| MX2014007935A (es) | 2011-12-28 | 2014-11-14 | Pozen Inc | Composiciones y metodos mejorados para el suministro de omeprazol y de acido acetilsalicilico. |
| JP6273302B2 (ja) * | 2013-03-15 | 2018-01-31 | コリア ユナイテッド ファーマ. インコーポレーテッド | 1日1回の投与で薬理学的臨床効果を提供するモサプリド徐放性製剤 |
| EP3288556A4 (en) | 2015-04-29 | 2018-09-19 | Dexcel Pharma Technologies Ltd. | Orally disintegrating compositions |
| CN108289849A (zh) * | 2015-06-26 | 2018-07-17 | 韩国联合制药株式会社 | 莫沙必利与雷贝拉唑的复合制剂 |
| US10076494B2 (en) | 2016-06-16 | 2018-09-18 | Dexcel Pharma Technologies Ltd. | Stable orally disintegrating pharmaceutical compositions |
| KR102034694B1 (ko) * | 2017-12-14 | 2019-10-22 | 한국유나이티드제약 주식회사 | 모사프리드와 라베프라졸을 함유하는 유핵정 복합제제 |
Family Cites Families (30)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS5535031A (en) * | 1978-09-04 | 1980-03-11 | Shin Etsu Chem Co Ltd | Enteric coating composition |
| JPS5598120A (en) * | 1979-01-16 | 1980-07-25 | Shin Etsu Chem Co Ltd | Preparation of drug having enteric coating |
| FR2471186A1 (fr) * | 1979-12-10 | 1981-06-19 | Roussel Uclaf | Nouveaux comprimes a delitescence colique, ainsi que leur procede de preparation |
| US4340582A (en) * | 1981-01-15 | 1982-07-20 | Abbott Laboratories | Erythromycin base tablets |
| US4377568A (en) * | 1981-08-12 | 1983-03-22 | Merck Sharp & Dohme (I.A.) Corp. | Preparation of aqueous alcoholic dispersions of pH sensitive polymers and plasticizing agents and a method of enteric coating dosage forms using same |
| DK150008C (da) * | 1981-11-20 | 1987-05-25 | Benzon As Alfred | Fremgangsmaade til fremstilling af et farmaceutisk oralt polydepotpraeparat |
| JPS5973529A (ja) * | 1982-10-12 | 1984-04-25 | Sankyo Co Ltd | 被覆固形製剤の製造法 |
| US4525339A (en) * | 1982-10-15 | 1985-06-25 | Hoffmann-La Roche Inc. | Enteric coated oral dosage form |
| US4518433A (en) * | 1982-11-08 | 1985-05-21 | Fmc Corporation | Enteric coating for pharmaceutical dosage forms |
| GB2132887A (en) * | 1982-11-15 | 1984-07-18 | Procter & Gamble | Enteric-coated anti-inflammatory compositions |
| US4556552A (en) * | 1983-09-19 | 1985-12-03 | Colorcon, Inc. | Enteric film-coating compositions |
| IE59287B1 (en) * | 1984-02-10 | 1994-02-09 | Benzon Pharma As | Diffusion coated multiple-units dosage form |
| US4568560A (en) * | 1984-03-16 | 1986-02-04 | Warner-Lambert Company | Encapsulated fragrances and flavors and process therefor |
| SE450087B (sv) * | 1985-10-11 | 1987-06-09 | Haessle Ab | Korn med kontrollerad frisettning av farmaceutiskt aktiva emnen som anbringats teckande pa ett kompakt olosligt kernmaterial |
| GB2189698A (en) * | 1986-04-30 | 1987-11-04 | Haessle Ab | Coated omeprazole tablets |
| US5330982A (en) * | 1986-12-17 | 1994-07-19 | Glaxo Group Limited | Pharmaceutical composition containing a 5-HT receptor antagonist and an H+ K+ Atpase inhibitor and a method of treating gastrointestingal disorders therewith |
| SE8803822D0 (sv) * | 1988-10-26 | 1988-10-26 | Novel dosage form | |
| US5026559A (en) * | 1989-04-03 | 1991-06-25 | Kinaform Technology, Inc. | Sustained-release pharmaceutical preparation |
| NZ235877A (en) * | 1989-11-02 | 1992-09-25 | Mcneil Ppc Inc | Composition comprising acetaminophen or nsaid and an h 1 or h 2 receptor blocker and/or proton pump inhibitor for treating overindulgence |
| JP3142919B2 (ja) * | 1991-11-06 | 2001-03-07 | 旭化成株式会社 | セルロース誘導体ラテックス及びその製法 |
| US5160742A (en) * | 1991-12-31 | 1992-11-03 | Abbott Laboratories | System for delivering an active substance for sustained release |
| SG42869A1 (en) * | 1992-08-05 | 1997-10-17 | Faulding F H & Co Ltd | Pelletised pharmaceutical composition |
| US5330759A (en) * | 1992-08-26 | 1994-07-19 | Sterling Winthrop Inc. | Enteric coated soft capsules and method of preparation thereof |
| CA2166730A1 (en) * | 1993-07-06 | 1995-01-19 | Robert T. Sims | H2 antagonist-gastrointestinal motility agent combinations |
| JP3017906B2 (ja) * | 1993-10-08 | 2000-03-13 | 信越化学工業株式会社 | 腸溶性コーティング剤分散液 |
| JP3710473B2 (ja) * | 1993-10-12 | 2005-10-26 | 三菱ウェルファーマ株式会社 | 腸溶性顆粒含有錠剤 |
| GB2285989A (en) * | 1994-01-28 | 1995-08-02 | Merck Frosst Canada Inc | Aqueous formulations for enteric coatings |
| EP0723436B1 (en) * | 1994-07-08 | 2001-09-26 | AstraZeneca AB | Multiple unit tableted dosage form i |
| SE9402431D0 (sv) * | 1994-07-08 | 1994-07-08 | Astra Ab | New tablet formulation |
| SE9600072D0 (sv) * | 1996-01-08 | 1996-01-08 | Astra Ab | New oral formulation of two active ingredients II |
-
1996
- 1996-01-08 SE SE9600072A patent/SE9600072D0/xx unknown
- 1996-12-20 US US08/750,936 patent/US6132771A/en not_active Expired - Lifetime
- 1996-12-20 WO PCT/SE1996/001736 patent/WO1997025065A1/en active IP Right Grant
- 1996-12-20 PT PT96944725T patent/PT814840E/pt unknown
- 1996-12-20 TR TR97/00917T patent/TR199700917T1/xx unknown
- 1996-12-20 KR KR1019970706235A patent/KR100486057B1/ko not_active IP Right Cessation
- 1996-12-20 PL PL96322176A patent/PL322176A1/xx unknown
- 1996-12-20 EE EE9700195A patent/EE9700195A/xx unknown
- 1996-12-20 HU HU9904094A patent/HUP9904094A3/hu unknown
- 1996-12-20 CN CNB96193607XA patent/CN1142792C/zh not_active Expired - Fee Related
- 1996-12-20 AU AU13240/97A patent/AU712572B2/en not_active Ceased
- 1996-12-20 SK SK1170-97A patent/SK117097A3/sk unknown
- 1996-12-20 DE DE69624910T patent/DE69624910T2/de not_active Expired - Fee Related
- 1996-12-20 JP JP9525130A patent/JPH11501949A/ja not_active Ceased
- 1996-12-20 IL IL12165296A patent/IL121652A/en not_active IP Right Cessation
- 1996-12-20 ES ES96944725T patent/ES2185816T3/es not_active Expired - Lifetime
- 1996-12-20 EP EP96944725A patent/EP0814840B1/en not_active Expired - Lifetime
- 1996-12-20 DK DK96944725T patent/DK0814840T3/da active
- 1996-12-20 AT AT96944725T patent/ATE228010T1/de not_active IP Right Cessation
- 1996-12-20 CZ CZ972808A patent/CZ280897A3/cs unknown
- 1996-12-20 BR BR9607344A patent/BR9607344A/pt not_active IP Right Cessation
- 1996-12-20 MX MX9706769A patent/MX9706769A/es not_active IP Right Cessation
- 1996-12-20 NZ NZ325976A patent/NZ325976A/xx unknown
- 1996-12-20 CA CA002214033A patent/CA2214033C/en not_active Expired - Fee Related
- 1996-12-26 AR ARP960105898A patent/AR008750A1/es not_active Application Discontinuation
- 1996-12-30 ZA ZA9610938A patent/ZA9610938B/xx unknown
-
1997
- 1997-01-08 ID IDP970026A patent/ID18383A/id unknown
- 1997-08-21 IS IS4550A patent/IS4550A/is unknown
- 1997-09-04 NO NO19974070A patent/NO316425B1/no not_active IP Right Cessation
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN1993132B (zh) * | 2004-07-28 | 2010-05-05 | 大日本住友制药株式会社 | 具有多包衣层的膜-包衣片剂 |
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP0814840B1 (en) | Oral pharmaceutical dosage forms comprising a proton pump inhibitor and a prokinetic agent | |
| EP0813424B1 (en) | Oral pharmaceutical dosage forms comprising a proton pump inhibitor and an antacid agent or alginate | |
| EP0814783B1 (en) | Multiple unit effervescent dosage forms comprising protonpump inhibitor | |
| JP3350054B2 (ja) | 複数単位の錠剤化された剤形▲i▼ | |
| EP0814839B1 (en) | Oral pharmaceutical dosage forms comprising a proton pump inhibitor and a nsaid | |
| KR100384961B1 (ko) | 프로톤펌프억제제를함유하는다단위제약제제 | |
| AU759311B2 (en) | Oral pharmaceutical pulsed release dosage form | |
| US6605303B1 (en) | Oral pharmaceutical extended release dosage form |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): AT BE CH DE DK ES FI FR GB GR IE IT LI LU MC NL PT SE |
|
| AX | Request for extension of the european patent |
Free format text: LT PAYMENT 971008;LV PAYMENT 971008;RO PAYMENT 971008;SI PAYMENT 971008 |
|
| 17P | Request for examination filed |
Effective date: 19980119 |
|
| RAP1 | Party data changed (applicant data changed or rights of an application transferred) |
Owner name: ASTRAZENECA AB |
|
| 17Q | First examination report despatched |
Effective date: 20010219 |
|
| RIC1 | Information provided on ipc code assigned before grant |
Free format text: 7A 61K 45/06 A, 7A 61K 31/44 B, 7A 61K 31/445 B, 7A 61K 9/20 B, 7A 61K 9/26 B, 7A 61K 9/48 B, 7A 61P 1/04 B |
|
| GRAG | Despatch of communication of intention to grant |
Free format text: ORIGINAL CODE: EPIDOS AGRA |
|
| GRAG | Despatch of communication of intention to grant |
Free format text: ORIGINAL CODE: EPIDOS AGRA |
|
| GRAG | Despatch of communication of intention to grant |
Free format text: ORIGINAL CODE: EPIDOS AGRA |
|
| GRAH | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOS IGRA |
|
| GRAH | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOS IGRA |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| RIN1 | Information on inventor provided before grant (corrected) |
Inventor name: HALLGREN, AGNETA Inventor name: DEPUI, HELENE |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): AT BE CH DE DK ES FI FR GB GR IE IT LI LU MC NL PT SE |
|
| AX | Request for extension of the european patent |
Free format text: LT PAYMENT 19971008;LV PAYMENT 19971008;RO PAYMENT 19971008;SI PAYMENT 19971008 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20021120 |
|
| REF | Corresponds to: |
Ref document number: 228010 Country of ref document: AT Date of ref document: 20021215 Kind code of ref document: T |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: FG4D |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: EP |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: FG4D |
|
| REF | Corresponds to: |
Ref document number: 69624910 Country of ref document: DE Date of ref document: 20030102 |
|
| REG | Reference to a national code |
Ref country code: DK Ref legal event code: T3 |
|
| REG | Reference to a national code |
Ref country code: GR Ref legal event code: EP Ref document number: 20030400607 Country of ref document: GR |
|
| LTIE | Lt: invalidation of european patent or patent extension |
Effective date: 20021120 |
|
| REG | Reference to a national code |
Ref country code: PT Ref legal event code: SC4A Free format text: AVAILABILITY OF NATIONAL TRANSLATION Effective date: 20030219 |
|
| REG | Reference to a national code |
Ref country code: ES Ref legal event code: FG2A Ref document number: 2185816 Country of ref document: ES Kind code of ref document: T3 |
|
| ET | Fr: translation filed | ||
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| 26N | No opposition filed |
Effective date: 20030821 |
|
| REG | Reference to a national code |
Ref country code: HK Ref legal event code: WD Ref document number: 1001762 Country of ref document: HK |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: NL Payment date: 20081111 Year of fee payment: 13 Ref country code: MC Payment date: 20081104 Year of fee payment: 13 Ref country code: LU Payment date: 20081021 Year of fee payment: 13 Ref country code: IE Payment date: 20081016 Year of fee payment: 13 Ref country code: DK Payment date: 20081112 Year of fee payment: 13 Ref country code: CH Payment date: 20081028 Year of fee payment: 13 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: PT Payment date: 20081006 Year of fee payment: 13 Ref country code: FI Payment date: 20081112 Year of fee payment: 13 Ref country code: ES Payment date: 20081217 Year of fee payment: 13 Ref country code: AT Payment date: 20081110 Year of fee payment: 13 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: SE Payment date: 20081208 Year of fee payment: 13 Ref country code: IT Payment date: 20081213 Year of fee payment: 13 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: FR Payment date: 20081205 Year of fee payment: 13 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: DE Payment date: 20081230 Year of fee payment: 13 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GR Payment date: 20081014 Year of fee payment: 13 Ref country code: GB Payment date: 20081110 Year of fee payment: 13 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: BE Payment date: 20090112 Year of fee payment: 13 |
|
| REG | Reference to a national code |
Ref country code: PT Ref legal event code: MM4A Free format text: LAPSE DUE TO NON-PAYMENT OF FEES Effective date: 20100621 |
|
| BERE | Be: lapsed |
Owner name: *ASTRAZENECA A.B. Effective date: 20091231 |
|
| REG | Reference to a national code |
Ref country code: NL Ref legal event code: V1 Effective date: 20100701 |
|
| EUG | Se: european patent has lapsed | ||
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: PT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20100621 Ref country code: MC Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20100701 |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PL |
|
| REG | Reference to a national code |
Ref country code: DK Ref legal event code: EBP |
|
| GBPC | Gb: european patent ceased through non-payment of renewal fee |
Effective date: 20091220 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: FI Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20091220 Ref country code: AT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20091220 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: ST Effective date: 20100831 |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: MM4A |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: NL Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20100701 Ref country code: LI Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20091231 Ref country code: IE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20091221 Ref country code: FR Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20091231 Ref country code: CH Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20091231 Ref country code: BE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20091231 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20100701 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GB Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20091220 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DK Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20100104 |
|
| REG | Reference to a national code |
Ref country code: ES Ref legal event code: FD2A Effective date: 20110307 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20091220 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LU Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20091220 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20091221 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: ES Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20110304 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: ES Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20091221 |