EP0343971A2 - Procédé de fabrication de feutre non réticulé - Google Patents
Procédé de fabrication de feutre non réticulé Download PDFInfo
- Publication number
- EP0343971A2 EP0343971A2 EP89305276A EP89305276A EP0343971A2 EP 0343971 A2 EP0343971 A2 EP 0343971A2 EP 89305276 A EP89305276 A EP 89305276A EP 89305276 A EP89305276 A EP 89305276A EP 0343971 A2 EP0343971 A2 EP 0343971A2
- Authority
- EP
- European Patent Office
- Prior art keywords
- fibers
- felt
- slurry
- weight
- solids
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Withdrawn
Links
Images
Classifications
-
- D—TEXTILES; PAPER
- D21—PAPER-MAKING; PRODUCTION OF CELLULOSE
- D21H—PULP COMPOSITIONS; PREPARATION THEREOF NOT COVERED BY SUBCLASSES D21C OR D21D; IMPREGNATING OR COATING OF PAPER; TREATMENT OF FINISHED PAPER NOT COVERED BY CLASS B31 OR SUBCLASS D21G; PAPER NOT OTHERWISE PROVIDED FOR
- D21H17/00—Non-fibrous material added to the pulp, characterised by its constitution; Paper-impregnating material characterised by its constitution
- D21H17/20—Macromolecular organic compounds
- D21H17/33—Synthetic macromolecular compounds
- D21H17/46—Synthetic macromolecular compounds obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds
- D21H17/47—Condensation polymers of aldehydes or ketones
- D21H17/48—Condensation polymers of aldehydes or ketones with phenols
-
- D—TEXTILES; PAPER
- D21—PAPER-MAKING; PRODUCTION OF CELLULOSE
- D21H—PULP COMPOSITIONS; PREPARATION THEREOF NOT COVERED BY SUBCLASSES D21C OR D21D; IMPREGNATING OR COATING OF PAPER; TREATMENT OF FINISHED PAPER NOT COVERED BY CLASS B31 OR SUBCLASS D21G; PAPER NOT OTHERWISE PROVIDED FOR
- D21H13/00—Pulp or paper, comprising synthetic cellulose or non-cellulose fibres or web-forming material
- D21H13/36—Inorganic fibres or flakes
-
- D—TEXTILES; PAPER
- D21—PAPER-MAKING; PRODUCTION OF CELLULOSE
- D21H—PULP COMPOSITIONS; PREPARATION THEREOF NOT COVERED BY SUBCLASSES D21C OR D21D; IMPREGNATING OR COATING OF PAPER; TREATMENT OF FINISHED PAPER NOT COVERED BY CLASS B31 OR SUBCLASS D21G; PAPER NOT OTHERWISE PROVIDED FOR
- D21H17/00—Non-fibrous material added to the pulp, characterised by its constitution; Paper-impregnating material characterised by its constitution
- D21H17/20—Macromolecular organic compounds
- D21H17/33—Synthetic macromolecular compounds
- D21H17/46—Synthetic macromolecular compounds obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds
-
- D—TEXTILES; PAPER
- D21—PAPER-MAKING; PRODUCTION OF CELLULOSE
- D21H—PULP COMPOSITIONS; PREPARATION THEREOF NOT COVERED BY SUBCLASSES D21C OR D21D; IMPREGNATING OR COATING OF PAPER; TREATMENT OF FINISHED PAPER NOT COVERED BY CLASS B31 OR SUBCLASS D21G; PAPER NOT OTHERWISE PROVIDED FOR
- D21H17/00—Non-fibrous material added to the pulp, characterised by its constitution; Paper-impregnating material characterised by its constitution
- D21H17/20—Macromolecular organic compounds
- D21H17/33—Synthetic macromolecular compounds
- D21H17/46—Synthetic macromolecular compounds obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds
- D21H17/47—Condensation polymers of aldehydes or ketones
-
- D—TEXTILES; PAPER
- D21—PAPER-MAKING; PRODUCTION OF CELLULOSE
- D21H—PULP COMPOSITIONS; PREPARATION THEREOF NOT COVERED BY SUBCLASSES D21C OR D21D; IMPREGNATING OR COATING OF PAPER; TREATMENT OF FINISHED PAPER NOT COVERED BY CLASS B31 OR SUBCLASS D21G; PAPER NOT OTHERWISE PROVIDED FOR
- D21H17/00—Non-fibrous material added to the pulp, characterised by its constitution; Paper-impregnating material characterised by its constitution
- D21H17/20—Macromolecular organic compounds
- D21H17/33—Synthetic macromolecular compounds
- D21H17/46—Synthetic macromolecular compounds obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds
- D21H17/47—Condensation polymers of aldehydes or ketones
- D21H17/49—Condensation polymers of aldehydes or ketones with compounds containing hydrogen bound to nitrogen
- D21H17/51—Triazines, e.g. melamine
-
- D—TEXTILES; PAPER
- D21—PAPER-MAKING; PRODUCTION OF CELLULOSE
- D21H—PULP COMPOSITIONS; PREPARATION THEREOF NOT COVERED BY SUBCLASSES D21C OR D21D; IMPREGNATING OR COATING OF PAPER; TREATMENT OF FINISHED PAPER NOT COVERED BY CLASS B31 OR SUBCLASS D21G; PAPER NOT OTHERWISE PROVIDED FOR
- D21H17/00—Non-fibrous material added to the pulp, characterised by its constitution; Paper-impregnating material characterised by its constitution
- D21H17/20—Macromolecular organic compounds
- D21H17/33—Synthetic macromolecular compounds
- D21H17/46—Synthetic macromolecular compounds obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds
- D21H17/52—Epoxy resins
-
- D—TEXTILES; PAPER
- D21—PAPER-MAKING; PRODUCTION OF CELLULOSE
- D21H—PULP COMPOSITIONS; PREPARATION THEREOF NOT COVERED BY SUBCLASSES D21C OR D21D; IMPREGNATING OR COATING OF PAPER; TREATMENT OF FINISHED PAPER NOT COVERED BY CLASS B31 OR SUBCLASS D21G; PAPER NOT OTHERWISE PROVIDED FOR
- D21H17/00—Non-fibrous material added to the pulp, characterised by its constitution; Paper-impregnating material characterised by its constitution
- D21H17/20—Macromolecular organic compounds
- D21H17/33—Synthetic macromolecular compounds
- D21H17/46—Synthetic macromolecular compounds obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds
- D21H17/53—Polyethers; Polyesters
-
- D—TEXTILES; PAPER
- D21—PAPER-MAKING; PRODUCTION OF CELLULOSE
- D21H—PULP COMPOSITIONS; PREPARATION THEREOF NOT COVERED BY SUBCLASSES D21C OR D21D; IMPREGNATING OR COATING OF PAPER; TREATMENT OF FINISHED PAPER NOT COVERED BY CLASS B31 OR SUBCLASS D21G; PAPER NOT OTHERWISE PROVIDED FOR
- D21H17/00—Non-fibrous material added to the pulp, characterised by its constitution; Paper-impregnating material characterised by its constitution
- D21H17/20—Macromolecular organic compounds
- D21H17/33—Synthetic macromolecular compounds
- D21H17/46—Synthetic macromolecular compounds obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds
- D21H17/58—Synthetic macromolecular compounds obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds obtained by reactions forming in the main chain of the macromolecule a linkage containing sulfur
-
- D—TEXTILES; PAPER
- D21—PAPER-MAKING; PRODUCTION OF CELLULOSE
- D21H—PULP COMPOSITIONS; PREPARATION THEREOF NOT COVERED BY SUBCLASSES D21C OR D21D; IMPREGNATING OR COATING OF PAPER; TREATMENT OF FINISHED PAPER NOT COVERED BY CLASS B31 OR SUBCLASS D21G; PAPER NOT OTHERWISE PROVIDED FOR
- D21H21/00—Non-fibrous material added to the pulp, characterised by its function, form or properties; Paper-impregnating or coating material, characterised by its function, form or properties
- D21H21/06—Paper forming aids
- D21H21/10—Retention agents or drainage improvers
Definitions
- This invention relates to a process for making uncured felt and, more particularly, to a wet process for making uncured felt.
- felt (or “felting”) is used herein to refer to a fibrous material made up of interlocked fibers held together by mechanical or chemical action, heat or moisture.
- Typical felts are made of mineral fibers such as ceramic fibers, glass fibers or mineral wool fibers, which are bonded together by a binder material.
- a problem in making such felts relates to providing a uniform distribution of the binder material throughout the felt product.
- a number of techniques have been suggested for dispersing binder materials in felt products.
- One of these suggestions involves dry spraying the binder material into fine streams of fiber filaments before the felt product is formed. See, for example, U.S. Patents 3,220,812; 3,337,669; 3,669,638; 3,745,060; 3,850,601; and 4,300,931.
- U.S. Patent 3,338,996 discloses the addition of a pulverized dry binder to a mass of glass fibers in dry layer form.
- U.S. Patents 2,566,960 and 2,782,458 disclose dipping fibrous felts in binder solutions.
- U.S. Patent 3,118,807 discloses a process for making bonded self-supported fibrous felts or blankets by the steps of (1) preparing a binder component comprising dried bentonite clay and particulate thermosetting resin, e.g., phenol formaldehyde resin (B-stage), (2) blending the binder component with a high refractory fiber, e.g., mineral or glass fiber, and (3) blowing the blended binder components with the fiber into a conventional felting unit.
- a binder component comprising dried bentonite clay and particulate thermosetting resin, e.g., phenol formaldehyde resin (B-stage)
- B-stage phenol formaldehyde resin
- a high refractory fiber e.g., mineral or glass fiber
- This invention provides for a process for making an uncured felt product which can be subsequently cured or thermoset to form hardened products such as heat shields, gaskets, pipe insulation jackets, etc.
- the process comprises the steps of (A) mixing refractory fibers and curable particulate polymeric solids of at least one thermosetting polymeric material other than a two-stage phenolic in water to form a slurry; (B) dewatering said slurry and forming felt comprising said solid fibers and said polymeric solids; and (C) drying said felt at a temperature and for a time sufficient to provide desired water removal but not high enough or long enough to cure said polymeric solids, the water content of said felt subsequent to said drying being up to about 25% by weight.
- the refractory fibers used in accordance with the inventive process can be selected from a wide range of amorphous or polycrystalline fibers that can be used at temperatures generally above about 1093°C (2000°F). These fibers can be oxide-containing or non-oxide fibers.
- the former includes alumina-silica fibers and chemical modifications of the alumina chemical system, high silica fibers (>99% SiO2), polycrystalline zirconia, polycrystalline mullite and alumina fibers.
- These fibers preferably have average diameters in the range from about 0.5 to about 10 microns, more preferably from about 2 to about 6 microns.
- the average lengths of these fibers typically range from about 0.025 to about 25 cm, more preferably from about 0.025 to about 10 cm.
- These fibers may contain up to about 60% by weight unfiberized particles. These unfiberized particles, commonly known as shot, are usually the result of melt fiberization and are often associated with alumina-silica fibers.
- the non-oxide forms include silicon carbide, silicon nitride and boron nitride. These forms typically have average diameters in the range of about 0.1 to about 50 microns, more preferably from about 0.1 to about 10 microns. These fibers preferably have average lengths in the range of about 0.025 to about 10 cm, more preferably from about 0.1 to about 5 cm.
- Preferred refractory fibers include ceramics, fiberglass and mineral wool, with ceramics being particularly preferred. These fibers preferably have average lengths in the range of about 0.025 to about 25 cm, more preferably from about 0.025 to about 10 cm; and average diameters in the range of about 0.5 to about 6 microns, more preferably about 2 to about 6 microns.
- the ceramic fibers can be kaolin based or high-purity based materials, made by either blowing or spinning fiberization techniques.
- Typical compositions of blown fibers are: Kaolin Based(Wt%) High-Purity Based(Wt%) SiO2 46-52% 48-53% Al2O3 42-51% 47-53% Na2O 0.1-0.2% 0.1-0.2% Fe2O3 0.8-1.1% Trace TiO2 1.0-1.8% Trace Typical characteristics of these ceramic fibers are: Blown Fibers Spun Fibers Shot Content(Wt%) 45-60% 45-55% Fiber Diameter (Avg., Microns) 2-4 3-6 Fiber Length (Avg., cm.) 0.025-5 0.05-25
- Fiberfrax Spun Bulk (a product of the Carborundum Company identified as lubricated alumina silica fibers). These fibers are formed using a spinning process, have average diameters of about 3 to about 4 microns, and average lengths of up to about 25 cm. Typical chemical analysis for this product is as follows (all parts and percentages are by weight): Al2O3 40% SiO2 53% Fe2O3 1% Na2O 0.1% TiO2 1% ZrO2 5% Leachable Chlorides ⁇ 10 ppm.
- the curable particulate solids that are intermixed with the refractory fibers in the inventive process can be any thermoset polymeric material, other than a two-stage phenolic, that is stable at the temperatures of intended use for the cured or thermoset products produced from the uncured felt products of the invention.
- the term "two-stage phenolic" (sometimes referred as "novolac") is used herein to refer to a low-molecular weight polymer made by reacting a phenol with an aldehyde, in the proportion of less than one mole of the phenol per mole of aldehyde, and requiring the addition of a curing agent to cure the resin.
- thermosetting polymeric materials that can be used in making curable particulate solids used in the inventive process include single-stage phenolics, alkyds, allyls, aminos, epoxies, furanes, polyesters, melamine-formaldehydes and silicones.
- the single-stage phenolics are particularly preferred.
- These curable particulate solids preferably have a Tyler standard screen size of about -200 (the designation "-200" meaning particles smaller than 200 mesh).
- single-stage phenolics (sometimes referred to as "resols") is used herein to refer to resins made by the reaction between a phenolic and an excess of aldehyde with an alkaline or acid catalyst. The ingredients are reacted to produce a low molecular weight prepolymer. The reaction is stopped before the resin becomes infusible so that it can be subsequently cured. In a subsequent high-temperature process, such as would occur by the user of the uncured felt products of the invention, the reaction between the phenol and the aldehyde is then completed, producing an infusable cross-linked thermoset material.
- the phenols that can be used include phenol, cresol, xylenols, p-t-butyl-phenol, p-phenylphenol, bisphenols and resorcinol.
- a commercially available phenolic that is useful is Varcum Synthetic Resin 29-217 (a product of BTL Speciality Resins Corp. identified as a highly condensed, one step, pulverized phenolic resin).
- the alkyd resins are made by reacting an unsaturated polyester with unsaturated monomers such as styrene, diallyl phthalate, diacetone acrylamide or vinyl toluene to form alkyd resins.
- unsaturated monomers such as styrene, diallyl phthalate, diacetone acrylamide or vinyl toluene to form alkyd resins.
- a peroxide catalyst is used to initiate cross-linking between the polyester resin and the monomers, which results in a cured thermoset system.
- the allyl resins are formed by the addition polymerization of compounds containing the group CH2:CH-CH2, such as esters of allyl alcohol and dibasic acids. They are commercially available as partially polymerized prepolymers and as molding compounds.
- the partially polymerized prepolymers can be cured with a peroxide catalyst. Examples include diallyl phthalate, diallyl isophthalate, diallyl maleate and diallyl chlorendate.
- the amino resins are polyalkylene amides which are nitrogen-rich polymers containing nitrogen in the amino form, NH2.
- the starting amino-bearing material is usually reacted with formaldehyde to form a reactive monomer, which is polymerized to a thermosetting resin.
- examples include urea, melamine, copolymers of both with formaldehyde, thiourea, aniniline, dicyanodiamide, toluene sulfonamide, benzoguanamine, ethylene urea and acrylamide.
- the epoxy resins are thermosetting resins containing the group These resins normally comprise diglicidyl ethers of bisphenol A and modifications thereof. These resins can also be formed by other means such as the oxidation of olefins with peracetic acid. These resins can be modified with other resins and unsaturated fatty acids, resulting in the formation of vinyl esters.
- furan resins sometimes referred to as furfuryl resins
- the furan resins can be obtained by the condensation polymerization of furfuryl alcohol in the presence of strong acids, sometimes in combination with formaldehyde and furfuryl aldehyde.
- resins made by condensing phenol with furfuryl alcohol or furfuryl, and furfuryl-ketone polymers are also included.
- the polyesters are characterized by vinyl unsaturation in the polyester backbone which enables subsequent hardening or curing.
- These unsaturated polyesters are derived from glycols, unsaturated dibasic acids or anhydrides and, sometimes in order to control the reaction and modified properties, saturated dibasic acids or anhydrides.
- Peroxide catalysts are typically used for curing.
- Aromatic polyesters such as homopolymers of repeating p-oxybenzoyl units, and copolymers containing the p-oxybenzoyl unit in combination moieties derived from aromatic dicarboxylic acids and bisphenols, can also be used.
- the melamine-formaldehyde resins are thermosetting resins made by reacting melamine with formaldehyde. These resins are typically in the form of spray-dried solids. The application of heat, in the presence of acid catalysts, converts these resins into hard, infusible materials.
- the silicone resins are cross-linked siloxane systems.
- the cross-linking components are usually introduced as trifunctional or tetrafunctional silanes. Examples of commonly used monomers include CH3SiCl3, C6H5SiCl3, (CH3)2SiCl2, (C6H5)2SiCl2 and CH3(C6H5)SiCl2. Silicon resins are typically cured through the formation of siloxane linkages by condensation of silanols.
- the flocculating agents that are useful include inorganic and synthetic organic agents as well as agents derived from natural products (naturally occurring organic polymers which may have been modified chemically).
- inorganic flocculating agents include aluminium derivatives; aluminium sulfate hydrate (alum) which can be represented by the formula Al2(SO4)3 ⁇ xH2O wherein x is about 14; iron derivatives such as ferric chloride; and lime.
- Examples of synthetic organic agents include poly(ethyleneamine); poly(2-hydroxypropyl-1-N-methylammonium chloride); poly-(2-hydroxypropyl-1, 1-N-dimethylammonium chloride); poly-[N-(dimethylaminomethyl)acrylamide]; poly(2-vinylimid-azolinum bisulfate); poly(diallyldimethylammonium chloride); poly(N,N-dimethylaminoethylmethacrylate), neutralized or quaternized; poly[N-(dimethylaminopropyl)-methacrylamide]; poly(sodium or ammonium acrylate); poly(sodium styrenesulfonate); polyacrylamide; poly(ethylene oxide); and poly(vinylpyrrolidinone).
- natur ally occurring organic materials that are useful as flocculating agents include guar gum and protein colloid.
- Commercially available flocculating agents that are useful include Betz 1260 (a product of Betz PaperChem, Inc., identified as a quaternary ammonium polyacrylamide) and Betz CDP-713 (a product of betz PaperChem, Inc., identified as polyacrylamide).
- the high temperture binders that are useful include colloidal silica, colloidal alumina and sodium silicate.
- the inorganic fillers that are useful include kaolin clay and alumina.
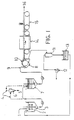
- the inventive process involves introducing the refractory fibers, the curable particulate polymeric solids and water as required, at point 1 to form a slurry 2 in a slurry mix tank 3.
- the fibers are preferably present at a concentration of about 0.3% to about 1% by weight, more preferably about 0.5% by weight, based on the total weight of the slurry.
- the curable particulate polymeric solids are preferably present at a concentration in the range of about 3% to about 15% by weight, more preferably about 11% by weight, based on the weight of the fibers. If any high temperature binders are used, such binders are added to the slurry in the mix tank.
- high temperature binders are preferably present at a concentration of up to about 100% by weight, more preferably about 25% to about 100% by weight, more preferably about 80% by weight, based on the weight of the fibers.
- Any inorganic fillers that are to be used, are also added to the slurry in the mix tank. These inorganic fillers are preferably present at a concentration of up to about 100% by weight, more preferably in the range of about 30% to about 80% by weight, based on the weight of the fibers.
- the slurry 2 is blended in the mix tank 3 until uniform.
- An optional step in the inventive process is the shot reduction system, which is indicated in Figure 1 by the dashed lines at 4.
- shot is removed at point 5 from the fiber using a wet-size separator 6 such as elutriator, jigger or hydrocyclone.
- a wet-size separator 6 such as elutriator, jigger or hydrocyclone.
- elutriator elutriator
- jigger jigger or hydrocyclone.
- Another optional step, not shown in Figure 1 is to remove or reduce shot from the fibers prior slurrying the fibers with the resin and other ingredients in the slurry mix tank 3.
- the slurry from the mix tank 3 or, alternatively, the slurry from the shot reduction system, is advanced to a surge tank 7. From the surge tank, the slurry is advanced to a web-forming vacuum filter 8 which is preferably a rotary drum, screen-type filter assisted by a water syphon or mechanical vacuum system. If flocculant is to be used, it is preferably to add the flocculant to the slurry at point 9 prior to entry in the web-forming apparatus.
- the flocculant is preferably added at a concentration sufficient to enhance retention of the curable polymeric solids on the refractory fibers.
- the flocculant is preferably added at a concentration in the range of about 0.01% to about 0.04% by weight, based on the weight of the fibers entering the web-forming vacuum filter 8.
- the slurry is de-watered and a continuous wet mat or web 10 with the refractory fibers intermeshed with each other and the curable particulate polymeric solids adhered to the fibers is formed.
- the refractory fibers are also intermixed with any high temperature binder, inorganic filler or flocculating agent that may have been added to the slurry.
- the solids content in the wet mat 10 is from about 30% to about 70% by weight, more preferably from about 50% to about 60% by weight, based on the weight of the wet mat.
- the water that is removed is advanced to a separator 11 connected to a vacuum pump 12. In the separator 11, solids are removed, the water is then advanced to a recycle water tank 13, and then advanced to the slurry mix tank 3 as needed. Make-up water is also added to the slurry mix tank 3 along with the recycled water as needed.
- the wet web is advanced from the web-forming vacuum filter 8 to the drying oven 14 wherein most or all of the free moisture in the web is removed.
- the drying oven 14 is preferably an electrically fired tunnel dryer wherein hot dry air is circulated over the web sufficiently to remove moisture.
- the web is dried sufficiently to provide a felt product with a water content of up to about 25% by weight, more preferably from about 3% to about 15% by weight.
- the temperature of the drying oven 14 and the residence time of the web in the oven is sufficient to provide the desired moisture removal, but not high enough or long enough to cure or thermoset the uncured polymeric material in the web.
- the drying temperature is in the range of about 105°C to about 190°C, more preferably in the range of about 115°C to about 175°C
- the residence time in the web in the drying oven 14 is preferably in the range of about 0.15 to about 0.5 hours, more preferably in the range of about 0.25 to about 0.4 hours.
- the uncured felt products produced therefrom preferably have a thickness in the range of about 1/8 to about 1 inch, more preferably about 1/4 to about 1 inch, more preferively about 3/8 to about 3/4 inch. These uncured felt products preferably have a density in the range of about 4 to about 10 pounds per cubic foot, more preferably about 6 to about 8 pounds per cubic foot. These products preferably contain from about 2% to about 20% by weight, more preferably from about 3% to about 12% by weight, more preferably about 3% to about 6% by weight uncured resin.
- the moisture content of these uncured felt products can be up to about 25% by weight, more preferably about 3% to about 20% by weight, more preferably from about 3% to about 15% by weight.
- Useful products for aerospace applications have moisture contents in the range of about 3% to about 5% by weight, and useful products for automotive applications have moisture contents of about 5% to about 20% by weight.
- the uncured felt products produced in accordance with the inventive process can be used in hot-compression molding operations to form hardened parts such as heat shields, gaskets, pipe insulation jackets, casting tips, and the like. These products have commercial use in the automotive, aerospace and metal foundry industries.
- Formulation 1 Feed Product Ceramic Fiber (kaolin based-spun) 80 82 Single-Stage Phenolic Resin Powder 9 6 Water 20,000 12 Betz CDP-713 0.012 0.012 Formulation 2 Feed Product Ceramic Fiber (kaolin based-blown) 100 91 Single-Stage Phenolic Resin Powder 4 4 Water 20,000 5
- An advantage of this inventive process is that a variety of fibers or fiber blends can be easily incorporated into the formulation by addition to the initial slurry in the slurry mix tank. Uncured felt products with higher resin contents and a wider variety of fillers and other additives can be provided with the inventive process than with prior art techniques.
Landscapes
- Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Inorganic Chemistry (AREA)
- Treatments For Attaching Organic Compounds To Fibrous Goods (AREA)
- Nonwoven Fabrics (AREA)
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US19961888A | 1988-05-27 | 1988-05-27 | |
| US199618 | 1988-05-27 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| EP0343971A2 true EP0343971A2 (fr) | 1989-11-29 |
| EP0343971A3 EP0343971A3 (fr) | 1991-01-30 |
Family
ID=22738316
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP19890305276 Withdrawn EP0343971A3 (fr) | 1988-05-27 | 1989-05-24 | Procédé de fabrication de feutre non réticulé |
Country Status (1)
| Country | Link |
|---|---|
| EP (1) | EP0343971A3 (fr) |
Family Cites Families (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS55130764A (en) * | 1979-03-30 | 1980-10-09 | Sumitomo Bakelite Co | Preparation of laminate suitable for electricccommunication apparatus |
| US4654100A (en) * | 1985-03-04 | 1987-03-31 | The Dow Chemical Company | Method for preparing random-fiber thermoset composites |
-
1989
- 1989-05-24 EP EP19890305276 patent/EP0343971A3/fr not_active Withdrawn
Also Published As
| Publication number | Publication date |
|---|---|
| EP0343971A3 (fr) | 1991-01-30 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US4810576A (en) | Treated glass fibers and aqueous dispersion and nonwoven mat of the glass fibers | |
| EP0133307B1 (fr) | Procédé de préparation d'un corps moulé en céramique | |
| JP3090462B2 (ja) | 水性分散液からのシートの製造法 | |
| KR0161985B1 (ko) | 플록 및 피브리드로부터 제조된 고강도 종이 | |
| GB2040273A (en) | Silica fibres production and use as friction linings | |
| JPS63230312A (ja) | 強化熱可塑性材料及びその製造方法 | |
| EP0321688A2 (fr) | Produits de vermiculite non dispersables | |
| EP0343971A2 (fr) | Procédé de fabrication de feutre non réticulé | |
| JPH06316892A (ja) | 温度安定性の電気絶縁紙及びその製法 | |
| JP3482610B2 (ja) | 多孔性炭素質成形板用プリプレグシート | |
| JP3275187B2 (ja) | 耐熱紙及びそれよりなる触媒担体 | |
| JPS58104059A (ja) | 繊維質成形体用組成物 | |
| EP0078617B1 (fr) | Matériau composé de laine de roche | |
| JPH0656551A (ja) | コージライト多孔質耐熱材の製造方法 | |
| JP3483036B2 (ja) | 熱硬化性複合材料及びその製造方法 | |
| JP3351599B2 (ja) | 発泡ボ−ド | |
| JPH0253385B2 (fr) | ||
| JPS59169989A (ja) | 耐熱性繊維質成形体の製造法 | |
| JPH0648860A (ja) | 多孔質耐熱材の製造方法 | |
| KR950000990B1 (ko) | 무기섬유 슬러리 성형용 수분산성 수지 조성물 | |
| JP2603974B2 (ja) | 多孔質セラミック焼結体およびその製造方法 | |
| JPS6287439A (ja) | ガラス繊維およびその水性分散液の製造方法 | |
| JPH0911430A (ja) | 耐熱性ボード | |
| JP2000191808A (ja) | プリント配線板用プリプレグシ―トの製造法 | |
| JP2819352B2 (ja) | 低密度断熱構造体の製造法 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| AK | Designated contracting states |
Kind code of ref document: A2 Designated state(s): AT BE CH DE ES FR GB GR IT LI LU NL SE |
|
| PUAL | Search report despatched |
Free format text: ORIGINAL CODE: 0009013 |
|
| RAP1 | Party data changed (applicant data changed or rights of an application transferred) |
Owner name: THE CARBORUNDUM COMPANY |
|
| RHK1 | Main classification (correction) |
Ipc: D21H 23/04 |
|
| AK | Designated contracting states |
Kind code of ref document: A3 Designated state(s): AT BE CH DE ES FR GB GR IT LI LU NL SE |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: THE APPLICATION IS DEEMED TO BE WITHDRAWN |
|
| 18D | Application deemed to be withdrawn |
Effective date: 19910731 |