WO2022118619A1 - 感光性ポリイミド樹脂組成物、樹脂膜及び電子装置 - Google Patents
感光性ポリイミド樹脂組成物、樹脂膜及び電子装置 Download PDFInfo
- Publication number
- WO2022118619A1 WO2022118619A1 PCT/JP2021/041279 JP2021041279W WO2022118619A1 WO 2022118619 A1 WO2022118619 A1 WO 2022118619A1 JP 2021041279 W JP2021041279 W JP 2021041279W WO 2022118619 A1 WO2022118619 A1 WO 2022118619A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- polyimide resin
- group
- resin composition
- photosensitive polyimide
- composition according
- Prior art date
Links
- 229920001721 polyimide Polymers 0.000 title claims abstract description 184
- 239000009719 polyimide resin Substances 0.000 title claims abstract description 168
- 239000000203 mixture Substances 0.000 title claims abstract description 105
- 239000011347 resin Substances 0.000 title claims description 48
- 229920005989 resin Polymers 0.000 title claims description 48
- 150000001875 compounds Chemical class 0.000 claims abstract description 56
- 125000000524 functional group Chemical group 0.000 claims abstract description 35
- 239000002904 solvent Substances 0.000 claims description 23
- 125000004432 carbon atom Chemical group C* 0.000 claims description 20
- 239000004642 Polyimide Substances 0.000 claims description 14
- 125000004122 cyclic group Chemical group 0.000 claims description 13
- 125000004435 hydrogen atom Chemical group [H]* 0.000 claims description 13
- 239000007787 solid Substances 0.000 claims description 12
- 239000003999 initiator Substances 0.000 claims description 11
- 239000011342 resin composition Substances 0.000 claims description 10
- 125000002723 alicyclic group Chemical group 0.000 claims description 8
- 125000002029 aromatic hydrocarbon group Chemical group 0.000 claims description 8
- 238000002834 transmittance Methods 0.000 claims description 8
- 125000002015 acyclic group Chemical group 0.000 claims description 7
- 125000001931 aliphatic group Chemical group 0.000 claims description 7
- 239000003086 colorant Substances 0.000 claims description 7
- NKYXYJFTTIPZDE-UHFFFAOYSA-N 4-[4-amino-2-(trifluoromethyl)phenoxy]-3-(trifluoromethyl)aniline Chemical compound FC(F)(F)C1=CC(N)=CC=C1OC1=CC=C(N)C=C1C(F)(F)F NKYXYJFTTIPZDE-UHFFFAOYSA-N 0.000 claims description 6
- 125000003647 acryloyl group Chemical group O=C([*])C([H])=C([H])[H] 0.000 claims description 6
- 125000003118 aryl group Chemical group 0.000 claims description 6
- NVKGJHAQGWCWDI-UHFFFAOYSA-N 4-[4-amino-2-(trifluoromethyl)phenyl]-3-(trifluoromethyl)aniline Chemical group FC(F)(F)C1=CC(N)=CC=C1C1=CC=C(N)C=C1C(F)(F)F NVKGJHAQGWCWDI-UHFFFAOYSA-N 0.000 claims description 5
- UKJLNMAFNRKWGR-UHFFFAOYSA-N cyclohexatrienamine Chemical group NC1=CC=C=C[CH]1 UKJLNMAFNRKWGR-UHFFFAOYSA-N 0.000 claims description 5
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 claims description 5
- 125000005375 organosiloxane group Chemical group 0.000 claims description 5
- 229910052727 yttrium Inorganic materials 0.000 claims description 5
- KWOIWTRRPFHBSI-UHFFFAOYSA-N 4-[2-[3-[2-(4-aminophenyl)propan-2-yl]phenyl]propan-2-yl]aniline Chemical compound C=1C=CC(C(C)(C)C=2C=CC(N)=CC=2)=CC=1C(C)(C)C1=CC=C(N)C=C1 KWOIWTRRPFHBSI-UHFFFAOYSA-N 0.000 claims description 4
- 238000010526 radical polymerization reaction Methods 0.000 claims description 2
- 239000010408 film Substances 0.000 description 107
- NIXOWILDQLNWCW-UHFFFAOYSA-M Acrylate Chemical compound [O-]C(=O)C=C NIXOWILDQLNWCW-UHFFFAOYSA-M 0.000 description 33
- 150000004985 diamines Chemical class 0.000 description 22
- YEJRWHAVMIAJKC-UHFFFAOYSA-N 4-Butyrolactone Chemical compound O=C1CCCO1 YEJRWHAVMIAJKC-UHFFFAOYSA-N 0.000 description 21
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 18
- -1 acrylic compound Chemical class 0.000 description 17
- 238000000059 patterning Methods 0.000 description 17
- GTDPSWPPOUPBNX-UHFFFAOYSA-N ac1mqpva Chemical compound CC12C(=O)OC(=O)C1(C)C1(C)C2(C)C(=O)OC1=O GTDPSWPPOUPBNX-UHFFFAOYSA-N 0.000 description 16
- 150000000000 tetracarboxylic acids Chemical class 0.000 description 16
- 238000006243 chemical reaction Methods 0.000 description 12
- 239000000243 solution Substances 0.000 description 12
- 125000003277 amino group Chemical group 0.000 description 11
- 230000000052 comparative effect Effects 0.000 description 11
- SECXISVLQFMRJM-UHFFFAOYSA-N N-Methylpyrrolidone Chemical compound CN1CCCC1=O SECXISVLQFMRJM-UHFFFAOYSA-N 0.000 description 10
- 238000000034 method Methods 0.000 description 10
- 239000000758 substrate Substances 0.000 description 10
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 9
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 9
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 9
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 8
- CURBACXRQKTCKZ-UHFFFAOYSA-N cyclobutane-1,2,3,4-tetracarboxylic acid Chemical compound OC(=O)C1C(C(O)=O)C(C(O)=O)C1C(O)=O CURBACXRQKTCKZ-UHFFFAOYSA-N 0.000 description 8
- 239000004065 semiconductor Substances 0.000 description 8
- 239000000126 substance Substances 0.000 description 8
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical compound [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 description 7
- 239000002253 acid Substances 0.000 description 7
- NIXOWILDQLNWCW-UHFFFAOYSA-N acrylic acid group Chemical group C(C=C)(=O)O NIXOWILDQLNWCW-UHFFFAOYSA-N 0.000 description 7
- 239000000412 dendrimer Substances 0.000 description 7
- 229920000736 dendritic polymer Polymers 0.000 description 7
- 239000000047 product Substances 0.000 description 7
- 239000010703 silicon Substances 0.000 description 7
- 229910052710 silicon Inorganic materials 0.000 description 7
- 239000002966 varnish Substances 0.000 description 7
- RZIPTXDCNDIINL-UHFFFAOYSA-N cyclohexane-1,1,2,2-tetracarboxylic acid Chemical compound OC(=O)C1(C(O)=O)CCCCC1(C(O)=O)C(O)=O RZIPTXDCNDIINL-UHFFFAOYSA-N 0.000 description 6
- 125000003700 epoxy group Chemical group 0.000 description 6
- 239000011229 interlayer Substances 0.000 description 6
- 235000012431 wafers Nutrition 0.000 description 6
- FXHOOIRPVKKKFG-UHFFFAOYSA-N N,N-Dimethylacetamide Chemical compound CN(C)C(C)=O FXHOOIRPVKKKFG-UHFFFAOYSA-N 0.000 description 5
- 239000006087 Silane Coupling Agent Substances 0.000 description 5
- ZJCCRDAZUWHFQH-UHFFFAOYSA-N Trimethylolpropane Chemical compound CCC(CO)(CO)CO ZJCCRDAZUWHFQH-UHFFFAOYSA-N 0.000 description 5
- 238000011161 development Methods 0.000 description 5
- IQPQWNKOIGAROB-UHFFFAOYSA-N isocyanate group Chemical group [N-]=C=O IQPQWNKOIGAROB-UHFFFAOYSA-N 0.000 description 5
- 239000002243 precursor Substances 0.000 description 5
- 229940042596 viscoat Drugs 0.000 description 5
- TXBCBTDQIULDIA-UHFFFAOYSA-N 2-[[3-hydroxy-2,2-bis(hydroxymethyl)propoxy]methyl]-2-(hydroxymethyl)propane-1,3-diol Chemical compound OCC(CO)(CO)COCC(CO)(CO)CO TXBCBTDQIULDIA-UHFFFAOYSA-N 0.000 description 4
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 4
- ZPAKUZKMGJJMAA-UHFFFAOYSA-N Cyclohexane-1,2,4,5-tetracarboxylic acid Chemical compound OC(=O)C1CC(C(O)=O)C(C(O)=O)CC1C(O)=O ZPAKUZKMGJJMAA-UHFFFAOYSA-N 0.000 description 4
- RDOXTESZEPMUJZ-UHFFFAOYSA-N anisole Chemical compound COC1=CC=CC=C1 RDOXTESZEPMUJZ-UHFFFAOYSA-N 0.000 description 4
- 239000003054 catalyst Substances 0.000 description 4
- 239000007822 coupling agent Substances 0.000 description 4
- BGTOWKSIORTVQH-UHFFFAOYSA-N cyclopentanone Chemical compound O=C1CCCC1 BGTOWKSIORTVQH-UHFFFAOYSA-N 0.000 description 4
- 238000004519 manufacturing process Methods 0.000 description 4
- WXZMFSXDPGVJKK-UHFFFAOYSA-N pentaerythritol Chemical compound OCC(CO)(CO)CO WXZMFSXDPGVJKK-UHFFFAOYSA-N 0.000 description 4
- FDPIMTJIUBPUKL-UHFFFAOYSA-N pentan-3-one Chemical compound CCC(=O)CC FDPIMTJIUBPUKL-UHFFFAOYSA-N 0.000 description 4
- 229920000642 polymer Polymers 0.000 description 4
- 230000001681 protective effect Effects 0.000 description 4
- YLHUPYSUKYAIBW-UHFFFAOYSA-N 1-acetylpyrrolidin-2-one Chemical compound CC(=O)N1CCCC1=O YLHUPYSUKYAIBW-UHFFFAOYSA-N 0.000 description 3
- LVUQCTGSDJLWCE-UHFFFAOYSA-N 1-benzylpyrrolidin-2-one Chemical compound O=C1CCCN1CC1=CC=CC=C1 LVUQCTGSDJLWCE-UHFFFAOYSA-N 0.000 description 3
- WMYINDVYGQKYMI-UHFFFAOYSA-N 2-[2,2-bis(hydroxymethyl)butoxymethyl]-2-ethylpropane-1,3-diol Chemical compound CCC(CO)(CO)COCC(CC)(CO)CO WMYINDVYGQKYMI-UHFFFAOYSA-N 0.000 description 3
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 3
- XDTMQSROBMDMFD-UHFFFAOYSA-N Cyclohexane Chemical compound C1CCCCC1 XDTMQSROBMDMFD-UHFFFAOYSA-N 0.000 description 3
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 3
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 3
- CTQNGGLPUBDAKN-UHFFFAOYSA-N O-Xylene Chemical compound CC1=CC=CC=C1C CTQNGGLPUBDAKN-UHFFFAOYSA-N 0.000 description 3
- DAKWPKUUDNSNPN-UHFFFAOYSA-N Trimethylolpropane triacrylate Chemical compound C=CC(=O)OCC(CC)(COC(=O)C=C)COC(=O)C=C DAKWPKUUDNSNPN-UHFFFAOYSA-N 0.000 description 3
- MPIAGWXWVAHQBB-UHFFFAOYSA-N [3-prop-2-enoyloxy-2-[[3-prop-2-enoyloxy-2,2-bis(prop-2-enoyloxymethyl)propoxy]methyl]-2-(prop-2-enoyloxymethyl)propyl] prop-2-enoate Chemical compound C=CC(=O)OCC(COC(=O)C=C)(COC(=O)C=C)COCC(COC(=O)C=C)(COC(=O)C=C)COC(=O)C=C MPIAGWXWVAHQBB-UHFFFAOYSA-N 0.000 description 3
- 150000001408 amides Chemical class 0.000 description 3
- 150000004984 aromatic diamines Chemical class 0.000 description 3
- 230000015572 biosynthetic process Effects 0.000 description 3
- 238000004132 cross linking Methods 0.000 description 3
- STZIXLPVKZUAMV-UHFFFAOYSA-N cyclopentane-1,1,2,2-tetracarboxylic acid Chemical class OC(=O)C1(C(O)=O)CCCC1(C(O)=O)C(O)=O STZIXLPVKZUAMV-UHFFFAOYSA-N 0.000 description 3
- SBZXBUIDTXKZTM-UHFFFAOYSA-N diglyme Chemical compound COCCOCCOC SBZXBUIDTXKZTM-UHFFFAOYSA-N 0.000 description 3
- 238000011156 evaluation Methods 0.000 description 3
- RAXXELZNTBOGNW-UHFFFAOYSA-N imidazole Natural products C1=CNC=N1 RAXXELZNTBOGNW-UHFFFAOYSA-N 0.000 description 3
- 239000010410 layer Substances 0.000 description 3
- 239000000463 material Substances 0.000 description 3
- QSHDDOUJBYECFT-UHFFFAOYSA-N mercury Chemical compound [Hg] QSHDDOUJBYECFT-UHFFFAOYSA-N 0.000 description 3
- 229910052753 mercury Inorganic materials 0.000 description 3
- 239000003960 organic solvent Substances 0.000 description 3
- 239000002994 raw material Substances 0.000 description 3
- 230000035484 reaction time Effects 0.000 description 3
- 239000012487 rinsing solution Substances 0.000 description 3
- 238000003786 synthesis reaction Methods 0.000 description 3
- YFNKIDBQEZZDLK-UHFFFAOYSA-N triglyme Chemical compound COCCOCCOCCOC YFNKIDBQEZZDLK-UHFFFAOYSA-N 0.000 description 3
- 239000008096 xylene Substances 0.000 description 3
- QASBHTCRFDZQAM-UHFFFAOYSA-N (2-isocyanato-2-methyl-3-prop-2-enoyloxypropyl) prop-2-enoate Chemical compound C=CC(=O)OCC(C)(COC(=O)C=C)N=C=O QASBHTCRFDZQAM-UHFFFAOYSA-N 0.000 description 2
- QEGNUYASOUJEHD-UHFFFAOYSA-N 1,1-dimethylcyclohexane Chemical compound CC1(C)CCCCC1 QEGNUYASOUJEHD-UHFFFAOYSA-N 0.000 description 2
- 239000012956 1-hydroxycyclohexylphenyl-ketone Substances 0.000 description 2
- PAMIQIKDUOTOBW-UHFFFAOYSA-N 1-methylpiperidine Chemical compound CN1CCCCC1 PAMIQIKDUOTOBW-UHFFFAOYSA-N 0.000 description 2
- STMDPCBYJCIZOD-UHFFFAOYSA-N 2-(2,4-dinitroanilino)-4-methylpentanoic acid Chemical compound CC(C)CC(C(O)=O)NC1=CC=C([N+]([O-])=O)C=C1[N+]([O-])=O STMDPCBYJCIZOD-UHFFFAOYSA-N 0.000 description 2
- FDSUVTROAWLVJA-UHFFFAOYSA-N 2-[[3-hydroxy-2,2-bis(hydroxymethyl)propoxy]methyl]-2-(hydroxymethyl)propane-1,3-diol;prop-2-enoic acid Chemical compound OC(=O)C=C.OC(=O)C=C.OC(=O)C=C.OC(=O)C=C.OC(=O)C=C.OCC(CO)(CO)COCC(CO)(CO)CO FDSUVTROAWLVJA-UHFFFAOYSA-N 0.000 description 2
- RLHGFJMGWQXPBW-UHFFFAOYSA-N 2-hydroxy-3-(1h-imidazol-5-ylmethyl)benzamide Chemical compound NC(=O)C1=CC=CC(CC=2NC=NC=2)=C1O RLHGFJMGWQXPBW-UHFFFAOYSA-N 0.000 description 2
- DPNXHTDWGGVXID-UHFFFAOYSA-N 2-isocyanatoethyl prop-2-enoate Chemical compound C=CC(=O)OCCN=C=O DPNXHTDWGGVXID-UHFFFAOYSA-N 0.000 description 2
- KRPRVQWGKLEFKN-UHFFFAOYSA-N 3-(3-aminopropoxy)propan-1-amine Chemical compound NCCCOCCCN KRPRVQWGKLEFKN-UHFFFAOYSA-N 0.000 description 2
- UCOJMQGVYMIIEM-UHFFFAOYSA-N 5-methoxycarbonylcyclohexane-1,2,4-tricarboxylic acid Chemical compound COC(=O)C1CC(C(O)=O)C(C(O)=O)CC1C(O)=O UCOJMQGVYMIIEM-UHFFFAOYSA-N 0.000 description 2
- 101001074560 Arabidopsis thaliana Aquaporin PIP1-2 Proteins 0.000 description 2
- RGSFGYAAUTVSQA-UHFFFAOYSA-N Cyclopentane Chemical compound C1CCCC1 RGSFGYAAUTVSQA-UHFFFAOYSA-N 0.000 description 2
- QIGBRXMKCJKVMJ-UHFFFAOYSA-N Hydroquinone Chemical compound OC1=CC=C(O)C=C1 QIGBRXMKCJKVMJ-UHFFFAOYSA-N 0.000 description 2
- QISSLHPKTCLLDL-UHFFFAOYSA-N N-Acetylcaprolactam Chemical compound CC(=O)N1CCCCCC1=O QISSLHPKTCLLDL-UHFFFAOYSA-N 0.000 description 2
- UFWIBTONFRDIAS-UHFFFAOYSA-N Naphthalene Chemical compound C1=CC=CC2=CC=CC=C21 UFWIBTONFRDIAS-UHFFFAOYSA-N 0.000 description 2
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 2
- JUJWROOIHBZHMG-UHFFFAOYSA-N Pyridine Chemical compound C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 description 2
- SMWDFEZZVXVKRB-UHFFFAOYSA-N Quinoline Chemical compound N1=CC=CC2=CC=CC=C21 SMWDFEZZVXVKRB-UHFFFAOYSA-N 0.000 description 2
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 2
- GUCYFKSBFREPBC-UHFFFAOYSA-N [phenyl-(2,4,6-trimethylbenzoyl)phosphoryl]-(2,4,6-trimethylphenyl)methanone Chemical compound CC1=CC(C)=CC(C)=C1C(=O)P(=O)(C=1C=CC=CC=1)C(=O)C1=C(C)C=C(C)C=C1C GUCYFKSBFREPBC-UHFFFAOYSA-N 0.000 description 2
- 230000002411 adverse Effects 0.000 description 2
- 150000001412 amines Chemical group 0.000 description 2
- 238000007611 bar coating method Methods 0.000 description 2
- 239000002585 base Substances 0.000 description 2
- IOJUPLGTWVMSFF-UHFFFAOYSA-N benzothiazole Chemical compound C1=CC=C2SC=NC2=C1 IOJUPLGTWVMSFF-UHFFFAOYSA-N 0.000 description 2
- BKDVBBSUAGJUBA-UHFFFAOYSA-N bicyclo[2.2.2]oct-7-ene-2,3,5,6-tetracarboxylic acid Chemical compound C1=CC2C(C(O)=O)C(C(=O)O)C1C(C(O)=O)C2C(O)=O BKDVBBSUAGJUBA-UHFFFAOYSA-N 0.000 description 2
- MQDJYUACMFCOFT-UHFFFAOYSA-N bis[2-(1-hydroxycyclohexyl)phenyl]methanone Chemical compound C=1C=CC=C(C(=O)C=2C(=CC=CC=2)C2(O)CCCCC2)C=1C1(O)CCCCC1 MQDJYUACMFCOFT-UHFFFAOYSA-N 0.000 description 2
- 238000000576 coating method Methods 0.000 description 2
- 239000012792 core layer Substances 0.000 description 2
- JHIVVAPYMSGYDF-UHFFFAOYSA-N cyclohexanone Chemical compound O=C1CCCCC1 JHIVVAPYMSGYDF-UHFFFAOYSA-N 0.000 description 2
- WOSVXXBNNCUXMT-UHFFFAOYSA-N cyclopentane-1,2,3,4-tetracarboxylic acid Chemical compound OC(=O)C1CC(C(O)=O)C(C(O)=O)C1C(O)=O WOSVXXBNNCUXMT-UHFFFAOYSA-N 0.000 description 2
- ZUOUZKKEUPVFJK-UHFFFAOYSA-N diphenyl Chemical compound C1=CC=CC=C1C1=CC=CC=C1 ZUOUZKKEUPVFJK-UHFFFAOYSA-N 0.000 description 2
- USIUVYZYUHIAEV-UHFFFAOYSA-N diphenyl ether Chemical compound C=1C=CC=CC=1OC1=CC=CC=C1 USIUVYZYUHIAEV-UHFFFAOYSA-N 0.000 description 2
- KZTYYGOKRVBIMI-UHFFFAOYSA-N diphenyl sulfone Chemical compound C=1C=CC=CC=1S(=O)(=O)C1=CC=CC=C1 KZTYYGOKRVBIMI-UHFFFAOYSA-N 0.000 description 2
- VOZRXNHHFUQHIL-UHFFFAOYSA-N glycidyl methacrylate Chemical compound CC(=C)C(=O)OCC1CO1 VOZRXNHHFUQHIL-UHFFFAOYSA-N 0.000 description 2
- 238000010438 heat treatment Methods 0.000 description 2
- 125000000623 heterocyclic group Chemical group 0.000 description 2
- NAQMVNRVTILPCV-UHFFFAOYSA-N hexane-1,6-diamine Chemical compound NCCCCCCN NAQMVNRVTILPCV-UHFFFAOYSA-N 0.000 description 2
- 239000004615 ingredient Substances 0.000 description 2
- 239000003112 inhibitor Substances 0.000 description 2
- HJOVHMDZYOCNQW-UHFFFAOYSA-N isophorone Chemical compound CC1=CC(=O)CC(C)(C)C1 HJOVHMDZYOCNQW-UHFFFAOYSA-N 0.000 description 2
- 239000007788 liquid Substances 0.000 description 2
- 239000004973 liquid crystal related substance Substances 0.000 description 2
- AMXOYNBUYSYVKV-UHFFFAOYSA-M lithium bromide Chemical compound [Li+].[Br-] AMXOYNBUYSYVKV-UHFFFAOYSA-M 0.000 description 2
- RBQRWNWVPQDTJJ-UHFFFAOYSA-N methacryloyloxyethyl isocyanate Chemical compound CC(=C)C(=O)OCCN=C=O RBQRWNWVPQDTJJ-UHFFFAOYSA-N 0.000 description 2
- UZKWTJUDCOPSNM-UHFFFAOYSA-N methoxybenzene Substances CCCCOC=C UZKWTJUDCOPSNM-UHFFFAOYSA-N 0.000 description 2
- 238000002156 mixing Methods 0.000 description 2
- 229910052757 nitrogen Inorganic materials 0.000 description 2
- RPQRDASANLAFCM-UHFFFAOYSA-N oxiran-2-ylmethyl prop-2-enoate Chemical compound C=CC(=O)OCC1CO1 RPQRDASANLAFCM-UHFFFAOYSA-N 0.000 description 2
- NWVVVBRKAWDGAB-UHFFFAOYSA-N p-methoxyphenol Chemical compound COC1=CC=C(O)C=C1 NWVVVBRKAWDGAB-UHFFFAOYSA-N 0.000 description 2
- 239000002798 polar solvent Substances 0.000 description 2
- 229920005575 poly(amic acid) Polymers 0.000 description 2
- 238000006116 polymerization reaction Methods 0.000 description 2
- 229920001451 polypropylene glycol Polymers 0.000 description 2
- CYIDZMCFTVVTJO-UHFFFAOYSA-N pyromellitic acid Chemical compound OC(=O)C1=CC(C(O)=O)=C(C(O)=O)C=C1C(O)=O CYIDZMCFTVVTJO-UHFFFAOYSA-N 0.000 description 2
- 150000003254 radicals Chemical class 0.000 description 2
- 238000005507 spraying Methods 0.000 description 2
- 125000001424 substituent group Chemical group 0.000 description 2
- CNHDIAIOKMXOLK-UHFFFAOYSA-N toluquinol Chemical compound CC1=CC(O)=CC=C1O CNHDIAIOKMXOLK-UHFFFAOYSA-N 0.000 description 2
- IMNIMPAHZVJRPE-UHFFFAOYSA-N triethylenediamine Chemical compound C1CN2CCN1CC2 IMNIMPAHZVJRPE-UHFFFAOYSA-N 0.000 description 2
- GETQZCLCWQTVFV-UHFFFAOYSA-N trimethylamine Chemical compound CN(C)C GETQZCLCWQTVFV-UHFFFAOYSA-N 0.000 description 2
- RIOQSEWOXXDEQQ-UHFFFAOYSA-N triphenylphosphine Chemical compound C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1 RIOQSEWOXXDEQQ-UHFFFAOYSA-N 0.000 description 2
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 2
- FHBXQJDYHHJCIF-UHFFFAOYSA-N (2,3-diaminophenyl)-phenylmethanone Chemical compound NC1=CC=CC(C(=O)C=2C=CC=CC=2)=C1N FHBXQJDYHHJCIF-UHFFFAOYSA-N 0.000 description 1
- MAOBFOXLCJIFLV-UHFFFAOYSA-N (2-aminophenyl)-phenylmethanone Chemical compound NC1=CC=CC=C1C(=O)C1=CC=CC=C1 MAOBFOXLCJIFLV-UHFFFAOYSA-N 0.000 description 1
- RXCOGDYOZQGGMK-UHFFFAOYSA-N (3,4-diaminophenyl)-phenylmethanone Chemical compound C1=C(N)C(N)=CC=C1C(=O)C1=CC=CC=C1 RXCOGDYOZQGGMK-UHFFFAOYSA-N 0.000 description 1
- RBKHNGHPZZZJCI-UHFFFAOYSA-N (4-aminophenyl)-phenylmethanone Chemical compound C1=CC(N)=CC=C1C(=O)C1=CC=CC=C1 RBKHNGHPZZZJCI-UHFFFAOYSA-N 0.000 description 1
- AVQQQNCBBIEMEU-UHFFFAOYSA-N 1,1,3,3-tetramethylurea Chemical compound CN(C)C(=O)N(C)C AVQQQNCBBIEMEU-UHFFFAOYSA-N 0.000 description 1
- WNXJIVFYUVYPPR-UHFFFAOYSA-N 1,3-dioxolane Chemical compound C1COCO1 WNXJIVFYUVYPPR-UHFFFAOYSA-N 0.000 description 1
- WZCQRUWWHSTZEM-UHFFFAOYSA-N 1,3-phenylenediamine Chemical compound NC1=CC=CC(N)=C1 WZCQRUWWHSTZEM-UHFFFAOYSA-N 0.000 description 1
- RYHBNJHYFVUHQT-UHFFFAOYSA-N 1,4-Dioxane Chemical compound C1COCCO1 RYHBNJHYFVUHQT-UHFFFAOYSA-N 0.000 description 1
- CBCKQZAAMUWICA-UHFFFAOYSA-N 1,4-phenylenediamine Chemical compound NC1=CC=C(N)C=C1 CBCKQZAAMUWICA-UHFFFAOYSA-N 0.000 description 1
- CGSKOGYKWHUSLC-UHFFFAOYSA-N 1-(4-aminophenyl)-1,3,3-trimethyl-2h-inden-5-amine Chemical compound C12=CC=C(N)C=C2C(C)(C)CC1(C)C1=CC=C(N)C=C1 CGSKOGYKWHUSLC-UHFFFAOYSA-N 0.000 description 1
- ONQBOTKLCMXPOF-UHFFFAOYSA-N 1-ethylpyrrolidine Chemical compound CCN1CCCC1 ONQBOTKLCMXPOF-UHFFFAOYSA-N 0.000 description 1
- AVFZOVWCLRSYKC-UHFFFAOYSA-N 1-methylpyrrolidine Chemical compound CN1CCCC1 AVFZOVWCLRSYKC-UHFFFAOYSA-N 0.000 description 1
- HXYTVQUCDXBNCR-UHFFFAOYSA-N 1h-inden-5-amine Chemical compound NC1=CC=C2CC=CC2=C1 HXYTVQUCDXBNCR-UHFFFAOYSA-N 0.000 description 1
- KWVGIHKZDCUPEU-UHFFFAOYSA-N 2,2-dimethoxy-2-phenylacetophenone Chemical compound C=1C=CC=CC=1C(OC)(OC)C(=O)C1=CC=CC=C1 KWVGIHKZDCUPEU-UHFFFAOYSA-N 0.000 description 1
- PTJWCLYPVFJWMP-UHFFFAOYSA-N 2-[[3-hydroxy-2-[[3-hydroxy-2,2-bis(hydroxymethyl)propoxy]methyl]-2-(hydroxymethyl)propoxy]methyl]-2-(hydroxymethyl)propane-1,3-diol Chemical compound OCC(CO)(CO)COCC(CO)(CO)COCC(CO)(CO)CO PTJWCLYPVFJWMP-UHFFFAOYSA-N 0.000 description 1
- BFSVOASYOCHEOV-UHFFFAOYSA-N 2-diethylaminoethanol Chemical compound CCN(CC)CCO BFSVOASYOCHEOV-UHFFFAOYSA-N 0.000 description 1
- PCKZAVNWRLEHIP-UHFFFAOYSA-N 2-hydroxy-1-[4-[[4-(2-hydroxy-2-methylpropanoyl)phenyl]methyl]phenyl]-2-methylpropan-1-one Chemical compound C1=CC(C(=O)C(C)(O)C)=CC=C1CC1=CC=C(C(=O)C(C)(C)O)C=C1 PCKZAVNWRLEHIP-UHFFFAOYSA-N 0.000 description 1
- XMLYCEVDHLAQEL-UHFFFAOYSA-N 2-hydroxy-2-methyl-1-phenylpropan-1-one Chemical compound CC(C)(O)C(=O)C1=CC=CC=C1 XMLYCEVDHLAQEL-UHFFFAOYSA-N 0.000 description 1
- LWRBVKNFOYUCNP-UHFFFAOYSA-N 2-methyl-1-(4-methylsulfanylphenyl)-2-morpholin-4-ylpropan-1-one Chemical compound C1=CC(SC)=CC=C1C(=O)C(C)(C)N1CCOCC1 LWRBVKNFOYUCNP-UHFFFAOYSA-N 0.000 description 1
- QTWJRLJHJPIABL-UHFFFAOYSA-N 2-methylphenol;3-methylphenol;4-methylphenol Chemical compound CC1=CC=C(O)C=C1.CC1=CC=CC(O)=C1.CC1=CC=CC=C1O QTWJRLJHJPIABL-UHFFFAOYSA-N 0.000 description 1
- SMDGQEQWSSYZKX-UHFFFAOYSA-N 3-(2,3-dicarboxyphenoxy)phthalic acid Chemical compound OC(=O)C1=CC=CC(OC=2C(=C(C(O)=O)C=CC=2)C(O)=O)=C1C(O)=O SMDGQEQWSSYZKX-UHFFFAOYSA-N 0.000 description 1
- NBAUUNCGSMAPFM-UHFFFAOYSA-N 3-(3,4-dicarboxyphenyl)phthalic acid Chemical compound C1=C(C(O)=O)C(C(=O)O)=CC=C1C1=CC=CC(C(O)=O)=C1C(O)=O NBAUUNCGSMAPFM-UHFFFAOYSA-N 0.000 description 1
- RNLHGQLZWXBQNY-UHFFFAOYSA-N 3-(aminomethyl)-3,5,5-trimethylcyclohexan-1-amine Chemical compound CC1(C)CC(N)CC(C)(CN)C1 RNLHGQLZWXBQNY-UHFFFAOYSA-N 0.000 description 1
- RHRNYXVSZLSRRP-UHFFFAOYSA-N 3-(carboxymethyl)cyclopentane-1,2,4-tricarboxylic acid Chemical compound OC(=O)CC1C(C(O)=O)CC(C(O)=O)C1C(O)=O RHRNYXVSZLSRRP-UHFFFAOYSA-N 0.000 description 1
- TYKLCAKICHXQNE-UHFFFAOYSA-N 3-[(2,3-dicarboxyphenyl)methyl]phthalic acid Chemical compound OC(=O)C1=CC=CC(CC=2C(=C(C(O)=O)C=CC=2)C(O)=O)=C1C(O)=O TYKLCAKICHXQNE-UHFFFAOYSA-N 0.000 description 1
- DFSUKONUQMHUKQ-UHFFFAOYSA-N 3-[2-(2,3-dicarboxyphenyl)-1,1,1,3,3,3-hexafluoropropan-2-yl]phthalic acid Chemical compound OC(=O)C1=CC=CC(C(C=2C(=C(C(O)=O)C=CC=2)C(O)=O)(C(F)(F)F)C(F)(F)F)=C1C(O)=O DFSUKONUQMHUKQ-UHFFFAOYSA-N 0.000 description 1
- PAHZZOIHRHCHTH-UHFFFAOYSA-N 3-[2-(2,3-dicarboxyphenyl)propan-2-yl]phthalic acid Chemical compound C=1C=CC(C(O)=O)=C(C(O)=O)C=1C(C)(C)C1=CC=CC(C(O)=O)=C1C(O)=O PAHZZOIHRHCHTH-UHFFFAOYSA-N 0.000 description 1
- IKYAJDOSWUATPI-UHFFFAOYSA-N 3-[dimethoxy(methyl)silyl]propane-1-thiol Chemical compound CO[Si](C)(OC)CCCS IKYAJDOSWUATPI-UHFFFAOYSA-N 0.000 description 1
- OXYZDRAJMHGSMW-UHFFFAOYSA-N 3-chloropropyl(trimethoxy)silane Chemical compound CO[Si](OC)(OC)CCCCl OXYZDRAJMHGSMW-UHFFFAOYSA-N 0.000 description 1
- PBXQRBDNLCEEAR-UHFFFAOYSA-N 3-methoxycarbonylbicyclo[2.2.2]oct-7-ene-2,5,6-tricarboxylic acid Chemical compound C1=CC2C(C(O)=O)C(C(O)=O)C1C(C(=O)OC)C2C(O)=O PBXQRBDNLCEEAR-UHFFFAOYSA-N 0.000 description 1
- LUTRJLBPCOJZIR-UHFFFAOYSA-N 3-methoxycarbonylcyclopentane-1,2,4-tricarboxylic acid Chemical compound COC(=O)C1C(C(O)=O)CC(C(O)=O)C1C(O)=O LUTRJLBPCOJZIR-UHFFFAOYSA-N 0.000 description 1
- SJECZPVISLOESU-UHFFFAOYSA-N 3-trimethoxysilylpropan-1-amine Chemical compound CO[Si](OC)(OC)CCCN SJECZPVISLOESU-UHFFFAOYSA-N 0.000 description 1
- UUEWCQRISZBELL-UHFFFAOYSA-N 3-trimethoxysilylpropane-1-thiol Chemical compound CO[Si](OC)(OC)CCCS UUEWCQRISZBELL-UHFFFAOYSA-N 0.000 description 1
- YBRVSVVVWCFQMG-UHFFFAOYSA-N 4,4'-diaminodiphenylmethane Chemical compound C1=CC(N)=CC=C1CC1=CC=C(N)C=C1 YBRVSVVVWCFQMG-UHFFFAOYSA-N 0.000 description 1
- ADRCREMWPUFGDU-UHFFFAOYSA-N 4-(1,3-benzothiazol-2-yl)-n,n-diethylaniline Chemical compound C1=CC(N(CC)CC)=CC=C1C1=NC2=CC=CC=C2S1 ADRCREMWPUFGDU-UHFFFAOYSA-N 0.000 description 1
- MYHNMZPERVYEKS-UHFFFAOYSA-N 4-(1,3-benzoxazol-2-yl)-n,n-diethylaniline Chemical compound C1=CC(N(CC)CC)=CC=C1C1=NC2=CC=CC=C2O1 MYHNMZPERVYEKS-UHFFFAOYSA-N 0.000 description 1
- NLWWHMRHFRTAII-UHFFFAOYSA-N 4-(1,3-benzoxazol-2-yl)-n,n-dimethylaniline Chemical compound C1=CC(N(C)C)=CC=C1C1=NC2=CC=CC=C2O1 NLWWHMRHFRTAII-UHFFFAOYSA-N 0.000 description 1
- KOECQEGGMWDHEV-UHFFFAOYSA-N 4-(1h-benzimidazol-2-yl)-n,n-diethylaniline Chemical compound C1=CC(N(CC)CC)=CC=C1C1=NC2=CC=CC=C2N1 KOECQEGGMWDHEV-UHFFFAOYSA-N 0.000 description 1
- ZKBBGUJBGLTNEK-UHFFFAOYSA-N 4-(1h-benzimidazol-2-yl)-n,n-dimethylaniline Chemical compound C1=CC(N(C)C)=CC=C1C1=NC2=CC=CC=C2N1 ZKBBGUJBGLTNEK-UHFFFAOYSA-N 0.000 description 1
- UITKHKNFVCYWNG-UHFFFAOYSA-N 4-(3,4-dicarboxybenzoyl)phthalic acid Chemical compound C1=C(C(O)=O)C(C(=O)O)=CC=C1C(=O)C1=CC=C(C(O)=O)C(C(O)=O)=C1 UITKHKNFVCYWNG-UHFFFAOYSA-N 0.000 description 1
- AIVVXPSKEVWKMY-UHFFFAOYSA-N 4-(3,4-dicarboxyphenoxy)phthalic acid Chemical compound C1=C(C(O)=O)C(C(=O)O)=CC=C1OC1=CC=C(C(O)=O)C(C(O)=O)=C1 AIVVXPSKEVWKMY-UHFFFAOYSA-N 0.000 description 1
- LFBALUPVVFCEPA-UHFFFAOYSA-N 4-(3,4-dicarboxyphenyl)phthalic acid Chemical compound C1=C(C(O)=O)C(C(=O)O)=CC=C1C1=CC=C(C(O)=O)C(C(O)=O)=C1 LFBALUPVVFCEPA-UHFFFAOYSA-N 0.000 description 1
- HLBLWEWZXPIGSM-UHFFFAOYSA-N 4-Aminophenyl ether Chemical compound C1=CC(N)=CC=C1OC1=CC=C(N)C=C1 HLBLWEWZXPIGSM-UHFFFAOYSA-N 0.000 description 1
- IWXCYYWDGDDPAC-UHFFFAOYSA-N 4-[(3,4-dicarboxyphenyl)methyl]phthalic acid Chemical compound C1=C(C(O)=O)C(C(=O)O)=CC=C1CC1=CC=C(C(O)=O)C(C(O)=O)=C1 IWXCYYWDGDDPAC-UHFFFAOYSA-N 0.000 description 1
- DZIHTWJGPDVSGE-UHFFFAOYSA-N 4-[(4-aminocyclohexyl)methyl]cyclohexan-1-amine Chemical compound C1CC(N)CCC1CC1CCC(N)CC1 DZIHTWJGPDVSGE-UHFFFAOYSA-N 0.000 description 1
- APXJLYIVOFARRM-UHFFFAOYSA-N 4-[2-(3,4-dicarboxyphenyl)-1,1,1,3,3,3-hexafluoropropan-2-yl]phthalic acid Chemical compound C1=C(C(O)=O)C(C(=O)O)=CC=C1C(C(F)(F)F)(C(F)(F)F)C1=CC=C(C(O)=O)C(C(O)=O)=C1 APXJLYIVOFARRM-UHFFFAOYSA-N 0.000 description 1
- GEYAGBVEAJGCFB-UHFFFAOYSA-N 4-[2-(3,4-dicarboxyphenyl)propan-2-yl]phthalic acid Chemical compound C=1C=C(C(O)=O)C(C(O)=O)=CC=1C(C)(C)C1=CC=C(C(O)=O)C(C(O)=O)=C1 GEYAGBVEAJGCFB-UHFFFAOYSA-N 0.000 description 1
- UZGVMZRBRRYLIP-UHFFFAOYSA-N 4-[5-[4-(diethylamino)phenyl]-1,3,4-oxadiazol-2-yl]-n,n-diethylaniline Chemical compound C1=CC(N(CC)CC)=CC=C1C1=NN=C(C=2C=CC(=CC=2)N(CC)CC)O1 UZGVMZRBRRYLIP-UHFFFAOYSA-N 0.000 description 1
- LGKCTTQKRNEUAL-UHFFFAOYSA-N 4-[5-[4-(diethylamino)phenyl]-1,3,4-thiadiazol-2-yl]-n,n-diethylaniline Chemical compound C1=CC(N(CC)CC)=CC=C1C1=NN=C(C=2C=CC(=CC=2)N(CC)CC)S1 LGKCTTQKRNEUAL-UHFFFAOYSA-N 0.000 description 1
- VIERZIUSJOSEGK-UHFFFAOYSA-N 4-cyclopenta[c][1,2]benzoxazin-2-yl-n,n-dimethylaniline Chemical compound C1=CC(N(C)C)=CC=C1C1=CC2=NOC3=CC=CC=C3C2=C1 VIERZIUSJOSEGK-UHFFFAOYSA-N 0.000 description 1
- RODDOGQBSBJOCQ-UHFFFAOYSA-N 4-cyclopenta[c][2,1]benzoxazin-2-yl-n,n-dimethylaniline Chemical compound C1=CC(N(C)C)=CC=C1C1=CC2=C3C=CC=CC3=NOC2=C1 RODDOGQBSBJOCQ-UHFFFAOYSA-N 0.000 description 1
- WLZKDDQDRZSHGX-UHFFFAOYSA-N 4-methoxycarbonylcyclobutane-1,2,3-tricarboxylic acid Chemical compound COC(=O)C1C(C(O)=O)C(C(O)=O)C1C(O)=O WLZKDDQDRZSHGX-UHFFFAOYSA-N 0.000 description 1
- VBBDHKXGNHTBBB-UHFFFAOYSA-N 5-methoxy-5-oxopentane-1,2,3-tricarboxylic acid Chemical compound COC(=O)CC(C(O)=O)C(C(O)=O)CC(O)=O VBBDHKXGNHTBBB-UHFFFAOYSA-N 0.000 description 1
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 1
- PMPVIKIVABFJJI-UHFFFAOYSA-N Cyclobutane Chemical compound C1CCC1 PMPVIKIVABFJJI-UHFFFAOYSA-N 0.000 description 1
- MQJKPEGWNLWLTK-UHFFFAOYSA-N Dapsone Chemical compound C1=CC(N)=CC=C1S(=O)(=O)C1=CC=C(N)C=C1 MQJKPEGWNLWLTK-UHFFFAOYSA-N 0.000 description 1
- OTMSDBZUPAUEDD-UHFFFAOYSA-N Ethane Chemical compound CC OTMSDBZUPAUEDD-UHFFFAOYSA-N 0.000 description 1
- KGWDUNBJIMUFAP-KVVVOXFISA-N Ethanolamine Oleate Chemical compound NCCO.CCCCCCCC\C=C/CCCCCCCC(O)=O KGWDUNBJIMUFAP-KVVVOXFISA-N 0.000 description 1
- PIICEJLVQHRZGT-UHFFFAOYSA-N Ethylenediamine Chemical compound NCCN PIICEJLVQHRZGT-UHFFFAOYSA-N 0.000 description 1
- YCKRFDGAMUMZLT-UHFFFAOYSA-N Fluorine atom Chemical compound [F] YCKRFDGAMUMZLT-UHFFFAOYSA-N 0.000 description 1
- CERQOIWHTDAKMF-UHFFFAOYSA-M Methacrylate Chemical compound CC(=C)C([O-])=O CERQOIWHTDAKMF-UHFFFAOYSA-M 0.000 description 1
- CERQOIWHTDAKMF-UHFFFAOYSA-N Methacrylic acid Chemical compound CC(=C)C(O)=O CERQOIWHTDAKMF-UHFFFAOYSA-N 0.000 description 1
- UEEJHVSXFDXPFK-UHFFFAOYSA-N N-dimethylaminoethanol Chemical compound CN(C)CCO UEEJHVSXFDXPFK-UHFFFAOYSA-N 0.000 description 1
- HTLZVHNRZJPSMI-UHFFFAOYSA-N N-ethylpiperidine Chemical compound CCN1CCCCC1 HTLZVHNRZJPSMI-UHFFFAOYSA-N 0.000 description 1
- AHVYPIQETPWLSZ-UHFFFAOYSA-N N-methyl-pyrrolidine Natural products CN1CC=CC1 AHVYPIQETPWLSZ-UHFFFAOYSA-N 0.000 description 1
- 239000002202 Polyethylene glycol Substances 0.000 description 1
- 239000004793 Polystyrene Substances 0.000 description 1
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical compound [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 description 1
- GSEJCLTVZPLZKY-UHFFFAOYSA-N Triethanolamine Chemical compound OCCN(CCO)CCO GSEJCLTVZPLZKY-UHFFFAOYSA-N 0.000 description 1
- XZAHJRZBUWYCBM-UHFFFAOYSA-N [1-(aminomethyl)cyclohexyl]methanamine Chemical compound NCC1(CN)CCCCC1 XZAHJRZBUWYCBM-UHFFFAOYSA-N 0.000 description 1
- FDLQZKYLHJJBHD-UHFFFAOYSA-N [3-(aminomethyl)phenyl]methanamine Chemical compound NCC1=CC=CC(CN)=C1 FDLQZKYLHJJBHD-UHFFFAOYSA-N 0.000 description 1
- SSOONFBDIYMPEU-UHFFFAOYSA-N [3-hydroxy-2-[[3-hydroxy-2,2-bis(hydroxymethyl)propoxy]methyl]-2-(hydroxymethyl)propyl] prop-2-enoate Chemical compound OCC(CO)(CO)COCC(CO)(CO)COC(=O)C=C SSOONFBDIYMPEU-UHFFFAOYSA-N 0.000 description 1
- OXIKYYJDTWKERT-UHFFFAOYSA-N [4-(aminomethyl)cyclohexyl]methanamine Chemical compound NCC1CCC(CN)CC1 OXIKYYJDTWKERT-UHFFFAOYSA-N 0.000 description 1
- ISKQADXMHQSTHK-UHFFFAOYSA-N [4-(aminomethyl)phenyl]methanamine Chemical compound NCC1=CC=C(CN)C=C1 ISKQADXMHQSTHK-UHFFFAOYSA-N 0.000 description 1
- 239000000654 additive Substances 0.000 description 1
- 230000000996 additive effect Effects 0.000 description 1
- 150000004645 aluminates Chemical class 0.000 description 1
- 229910000147 aluminium phosphate Inorganic materials 0.000 description 1
- 125000000129 anionic group Chemical group 0.000 description 1
- 238000013459 approach Methods 0.000 description 1
- RWCCWEUUXYIKHB-UHFFFAOYSA-N benzophenone Chemical compound C=1C=CC=CC=1C(=O)C1=CC=CC=C1 RWCCWEUUXYIKHB-UHFFFAOYSA-N 0.000 description 1
- 239000012965 benzophenone Substances 0.000 description 1
- 150000008366 benzophenones Chemical class 0.000 description 1
- 230000001588 bifunctional effect Effects 0.000 description 1
- 235000010290 biphenyl Nutrition 0.000 description 1
- 239000004305 biphenyl Substances 0.000 description 1
- TUQQUUXMCKXGDI-UHFFFAOYSA-N bis(3-aminophenyl)methanone Chemical compound NC1=CC=CC(C(=O)C=2C=C(N)C=CC=2)=C1 TUQQUUXMCKXGDI-UHFFFAOYSA-N 0.000 description 1
- ZLSMCQSGRWNEGX-UHFFFAOYSA-N bis(4-aminophenyl)methanone Chemical compound C1=CC(N)=CC=C1C(=O)C1=CC=C(N)C=C1 ZLSMCQSGRWNEGX-UHFFFAOYSA-N 0.000 description 1
- QDVNNDYBCWZVTI-UHFFFAOYSA-N bis[4-(ethylamino)phenyl]methanone Chemical compound C1=CC(NCC)=CC=C1C(=O)C1=CC=C(NCC)C=C1 QDVNNDYBCWZVTI-UHFFFAOYSA-N 0.000 description 1
- HXTBYXIZCDULQI-UHFFFAOYSA-N bis[4-(methylamino)phenyl]methanone Chemical compound C1=CC(NC)=CC=C1C(=O)C1=CC=C(NC)C=C1 HXTBYXIZCDULQI-UHFFFAOYSA-N 0.000 description 1
- 235000013877 carbamide Nutrition 0.000 description 1
- 150000001732 carboxylic acid derivatives Chemical class 0.000 description 1
- 238000005266 casting Methods 0.000 description 1
- 230000015556 catabolic process Effects 0.000 description 1
- 125000002091 cationic group Chemical group 0.000 description 1
- 239000003795 chemical substances by application Substances 0.000 description 1
- 239000011248 coating agent Substances 0.000 description 1
- 229920001577 copolymer Polymers 0.000 description 1
- 239000011889 copper foil Substances 0.000 description 1
- 229930003836 cresol Natural products 0.000 description 1
- PQRHREUWMOAJOW-UHFFFAOYSA-N ctk1a6096 Chemical class N[SiH](N)[SiH3] PQRHREUWMOAJOW-UHFFFAOYSA-N 0.000 description 1
- 230000001186 cumulative effect Effects 0.000 description 1
- 150000004292 cyclic ethers Chemical class 0.000 description 1
- 150000003997 cyclic ketones Chemical class 0.000 description 1
- XXKOQQBKBHUATC-UHFFFAOYSA-N cyclohexylmethylcyclohexane Chemical compound C1CCCCC1CC1CCCCC1 XXKOQQBKBHUATC-UHFFFAOYSA-N 0.000 description 1
- MAWOHFOSAIXURX-UHFFFAOYSA-N cyclopentylcyclopentane Chemical compound C1CCCC1C1CCCC1 MAWOHFOSAIXURX-UHFFFAOYSA-N 0.000 description 1
- 239000012024 dehydrating agents Substances 0.000 description 1
- 230000018044 dehydration Effects 0.000 description 1
- 238000006297 dehydration reaction Methods 0.000 description 1
- 238000007607 die coating method Methods 0.000 description 1
- 125000005442 diisocyanate group Chemical group 0.000 description 1
- 238000003618 dip coating Methods 0.000 description 1
- CZZYITDELCSZES-UHFFFAOYSA-N diphenylmethane Chemical compound C=1C=CC=CC=1CC1=CC=CC=C1 CZZYITDELCSZES-UHFFFAOYSA-N 0.000 description 1
- MZRQZJOUYWKDNH-UHFFFAOYSA-N diphenylphosphoryl-(2,3,4-trimethylphenyl)methanone Chemical compound CC1=C(C)C(C)=CC=C1C(=O)P(=O)(C=1C=CC=CC=1)C1=CC=CC=C1 MZRQZJOUYWKDNH-UHFFFAOYSA-N 0.000 description 1
- KPUWHANPEXNPJT-UHFFFAOYSA-N disiloxane Chemical class [SiH3]O[SiH3] KPUWHANPEXNPJT-UHFFFAOYSA-N 0.000 description 1
- 238000001035 drying Methods 0.000 description 1
- 239000003480 eluent Substances 0.000 description 1
- FWDBOZPQNFPOLF-UHFFFAOYSA-N ethenyl(triethoxy)silane Chemical compound CCO[Si](OCC)(OCC)C=C FWDBOZPQNFPOLF-UHFFFAOYSA-N 0.000 description 1
- NKSJNEHGWDZZQF-UHFFFAOYSA-N ethenyl(trimethoxy)silane Chemical compound CO[Si](OC)(OC)C=C NKSJNEHGWDZZQF-UHFFFAOYSA-N 0.000 description 1
- AIJZIRPGCQPZSL-UHFFFAOYSA-N ethylenetetracarboxylic acid Chemical compound OC(=O)C(C(O)=O)=C(C(O)=O)C(O)=O AIJZIRPGCQPZSL-UHFFFAOYSA-N 0.000 description 1
- 229910052731 fluorine Inorganic materials 0.000 description 1
- 239000011737 fluorine Substances 0.000 description 1
- 125000001153 fluoro group Chemical group F* 0.000 description 1
- 239000011888 foil Substances 0.000 description 1
- 238000009472 formulation Methods 0.000 description 1
- 239000007863 gel particle Substances 0.000 description 1
- 239000011521 glass Substances 0.000 description 1
- 230000009477 glass transition Effects 0.000 description 1
- 238000007756 gravure coating Methods 0.000 description 1
- DMEGYFMYUHOHGS-UHFFFAOYSA-N heptamethylene Natural products C1CCCCCC1 DMEGYFMYUHOHGS-UHFFFAOYSA-N 0.000 description 1
- GNOIPBMMFNIUFM-UHFFFAOYSA-N hexamethylphosphoric triamide Chemical compound CN(C)P(=O)(N(C)C)N(C)C GNOIPBMMFNIUFM-UHFFFAOYSA-N 0.000 description 1
- 229920001519 homopolymer Polymers 0.000 description 1
- 230000001771 impaired effect Effects 0.000 description 1
- 239000001023 inorganic pigment Substances 0.000 description 1
- 239000013067 intermediate product Substances 0.000 description 1
- 230000001678 irradiating effect Effects 0.000 description 1
- ZFSLODLOARCGLH-UHFFFAOYSA-N isocyanuric acid Chemical compound OC1=NC(O)=NC(O)=N1 ZFSLODLOARCGLH-UHFFFAOYSA-N 0.000 description 1
- AWJUIBRHMBBTKR-UHFFFAOYSA-N isoquinoline Chemical compound C1=NC=CC2=CC=CC=C21 AWJUIBRHMBBTKR-UHFFFAOYSA-N 0.000 description 1
- 229940018564 m-phenylenediamine Drugs 0.000 description 1
- 238000005259 measurement Methods 0.000 description 1
- 229910052751 metal Inorganic materials 0.000 description 1
- 239000002184 metal Substances 0.000 description 1
- HGUBFVKLMFGDJZ-UHFFFAOYSA-N methanol;silicon Chemical compound [Si].OC HGUBFVKLMFGDJZ-UHFFFAOYSA-N 0.000 description 1
- 150000004702 methyl esters Chemical class 0.000 description 1
- PHQOGHDTIVQXHL-UHFFFAOYSA-N n'-(3-trimethoxysilylpropyl)ethane-1,2-diamine Chemical compound CO[Si](OC)(OC)CCCNCCN PHQOGHDTIVQXHL-UHFFFAOYSA-N 0.000 description 1
- HOSQZKZVJBFALN-UHFFFAOYSA-N n'-benzyl-n-(3-trimethoxysilylpropyl)ethane-1,2-diamine;hydron;chloride Chemical compound Cl.CO[Si](OC)(OC)CCCNCCNCC1=CC=CC=C1 HOSQZKZVJBFALN-UHFFFAOYSA-N 0.000 description 1
- SAQPQTKYCWAAMJ-UHFFFAOYSA-N n,n-diethyl-4-pyridin-2-ylaniline Chemical compound C1=CC(N(CC)CC)=CC=C1C1=CC=CC=N1 SAQPQTKYCWAAMJ-UHFFFAOYSA-N 0.000 description 1
- MLNHFFKCCKXSAW-UHFFFAOYSA-N n,n-diethyl-4-pyrimidin-2-ylaniline Chemical compound C1=CC(N(CC)CC)=CC=C1C1=NC=CC=N1 MLNHFFKCCKXSAW-UHFFFAOYSA-N 0.000 description 1
- KVEFSERWIAYNPO-UHFFFAOYSA-N n,n-diethyl-4-quinolin-2-ylaniline Chemical compound C1=CC(N(CC)CC)=CC=C1C1=CC=C(C=CC=C2)C2=N1 KVEFSERWIAYNPO-UHFFFAOYSA-N 0.000 description 1
- QTBGSBRUWVUWCS-UHFFFAOYSA-N n,n-dimethyl-4-pyridin-2-ylaniline Chemical compound C1=CC(N(C)C)=CC=C1C1=CC=CC=N1 QTBGSBRUWVUWCS-UHFFFAOYSA-N 0.000 description 1
- RJJJWZAJSLSBKC-UHFFFAOYSA-N n,n-dimethyl-4-pyrimidin-2-ylaniline Chemical compound C1=CC(N(C)C)=CC=C1C1=NC=CC=N1 RJJJWZAJSLSBKC-UHFFFAOYSA-N 0.000 description 1
- KLNWLOCDPWWNDS-UHFFFAOYSA-N n,n-dimethyl-4-quinolin-2-ylaniline Chemical compound C1=CC(N(C)C)=CC=C1C1=CC=C(C=CC=C2)C2=N1 KLNWLOCDPWWNDS-UHFFFAOYSA-N 0.000 description 1
- KBJFYLLAMSZSOG-UHFFFAOYSA-N n-(3-trimethoxysilylpropyl)aniline Chemical compound CO[Si](OC)(OC)CCCNC1=CC=CC=C1 KBJFYLLAMSZSOG-UHFFFAOYSA-N 0.000 description 1
- KQSABULTKYLFEV-UHFFFAOYSA-N naphthalene-1,5-diamine Chemical compound C1=CC=C2C(N)=CC=CC2=C1N KQSABULTKYLFEV-UHFFFAOYSA-N 0.000 description 1
- OTLDLKLSNZMTTA-UHFFFAOYSA-N octahydro-1h-4,7-methanoindene-1,5-diyldimethanol Chemical compound C1C2C3C(CO)CCC3C1C(CO)C2 OTLDLKLSNZMTTA-UHFFFAOYSA-N 0.000 description 1
- 230000003287 optical effect Effects 0.000 description 1
- 125000000962 organic group Chemical group 0.000 description 1
- 239000012860 organic pigment Substances 0.000 description 1
- 150000002903 organophosphorus compounds Chemical class 0.000 description 1
- 239000003973 paint Substances 0.000 description 1
- 238000005192 partition Methods 0.000 description 1
- 238000002161 passivation Methods 0.000 description 1
- XNLICIUVMPYHGG-UHFFFAOYSA-N pentan-2-one Chemical compound CCCC(C)=O XNLICIUVMPYHGG-UHFFFAOYSA-N 0.000 description 1
- MIVZUXGHPJSKRI-UHFFFAOYSA-N pentane-1,1,1,2-tetracarboxylic acid Chemical compound CCCC(C(O)=O)C(C(O)=O)(C(O)=O)C(O)=O MIVZUXGHPJSKRI-UHFFFAOYSA-N 0.000 description 1
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 1
- 230000000704 physical effect Effects 0.000 description 1
- 239000002985 plastic film Substances 0.000 description 1
- 229920006255 plastic film Polymers 0.000 description 1
- 239000003495 polar organic solvent Substances 0.000 description 1
- 229920000058 polyacrylate Polymers 0.000 description 1
- 229920002577 polybenzoxazole Polymers 0.000 description 1
- 229920000728 polyester Polymers 0.000 description 1
- 229920001223 polyethylene glycol Polymers 0.000 description 1
- 229920002223 polystyrene Polymers 0.000 description 1
- 238000007639 printing Methods 0.000 description 1
- ODLMAHJVESYWTB-UHFFFAOYSA-N propylbenzene Chemical compound CCCC1=CC=CC=C1 ODLMAHJVESYWTB-UHFFFAOYSA-N 0.000 description 1
- UMJSCPRVCHMLSP-UHFFFAOYSA-N pyridine Natural products COC1=CC=CN=C1 UMJSCPRVCHMLSP-UHFFFAOYSA-N 0.000 description 1
- 238000006798 ring closing metathesis reaction Methods 0.000 description 1
- 238000007650 screen-printing Methods 0.000 description 1
- 238000007086 side reaction Methods 0.000 description 1
- 238000004528 spin coating Methods 0.000 description 1
- 238000003756 stirring Methods 0.000 description 1
- 150000003457 sulfones Chemical class 0.000 description 1
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 1
- 239000010409 thin film Substances 0.000 description 1
- 125000003396 thiol group Chemical group [H]S* 0.000 description 1
- IMFACGCPASFAPR-UHFFFAOYSA-N tributylamine Chemical compound CCCCN(CCCC)CCCC IMFACGCPASFAPR-UHFFFAOYSA-N 0.000 description 1
- DQZNLOXENNXVAD-UHFFFAOYSA-N trimethoxy-[2-(7-oxabicyclo[4.1.0]heptan-4-yl)ethyl]silane Chemical compound C1C(CC[Si](OC)(OC)OC)CCC2OC21 DQZNLOXENNXVAD-UHFFFAOYSA-N 0.000 description 1
- BPSIOYPQMFLKFR-UHFFFAOYSA-N trimethoxy-[3-(oxiran-2-ylmethoxy)propyl]silane Chemical compound CO[Si](OC)(OC)CCCOCC1CO1 BPSIOYPQMFLKFR-UHFFFAOYSA-N 0.000 description 1
- YFTHZRPMJXBUME-UHFFFAOYSA-N tripropylamine Chemical compound CCCN(CCC)CCC YFTHZRPMJXBUME-UHFFFAOYSA-N 0.000 description 1
- 150000003672 ureas Chemical class 0.000 description 1
- 125000000391 vinyl group Chemical group [H]C([*])=C([H])[H] 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F2/00—Processes of polymerisation
- C08F2/46—Polymerisation initiated by wave energy or particle radiation
- C08F2/48—Polymerisation initiated by wave energy or particle radiation by ultraviolet or visible light
- C08F2/50—Polymerisation initiated by wave energy or particle radiation by ultraviolet or visible light with sensitising agents
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F290/00—Macromolecular compounds obtained by polymerising monomers on to polymers modified by introduction of aliphatic unsaturated end or side groups
- C08F290/02—Macromolecular compounds obtained by polymerising monomers on to polymers modified by introduction of aliphatic unsaturated end or side groups on to polymers modified by introduction of unsaturated end groups
- C08F290/06—Polymers provided for in subclass C08G
- C08F290/065—Polyamides; Polyesteramides; Polyimides
-
- G—PHYSICS
- G03—PHOTOGRAPHY; CINEMATOGRAPHY; ANALOGOUS TECHNIQUES USING WAVES OTHER THAN OPTICAL WAVES; ELECTROGRAPHY; HOLOGRAPHY
- G03F—PHOTOMECHANICAL PRODUCTION OF TEXTURED OR PATTERNED SURFACES, e.g. FOR PRINTING, FOR PROCESSING OF SEMICONDUCTOR DEVICES; MATERIALS THEREFOR; ORIGINALS THEREFOR; APPARATUS SPECIALLY ADAPTED THEREFOR
- G03F7/00—Photomechanical, e.g. photolithographic, production of textured or patterned surfaces, e.g. printing surfaces; Materials therefor, e.g. comprising photoresists; Apparatus specially adapted therefor
- G03F7/004—Photosensitive materials
- G03F7/038—Macromolecular compounds which are rendered insoluble or differentially wettable
- G03F7/0387—Polyamides or polyimides
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F2/00—Processes of polymerisation
- C08F2/44—Polymerisation in the presence of compounding ingredients, e.g. plasticisers, dyestuffs, fillers
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G18/00—Polymeric products of isocyanates or isothiocyanates
- C08G18/06—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen
- C08G18/70—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen characterised by the isocyanates or isothiocyanates used
- C08G18/81—Unsaturated isocyanates or isothiocyanates
- C08G18/8108—Unsaturated isocyanates or isothiocyanates having only one isocyanate or isothiocyanate group
- C08G18/8116—Unsaturated isocyanates or isothiocyanates having only one isocyanate or isothiocyanate group esters of acrylic or alkylacrylic acid having only one isocyanate or isothiocyanate group
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G73/00—Macromolecular compounds obtained by reactions forming a linkage containing nitrogen with or without oxygen or carbon in the main chain of the macromolecule, not provided for in groups C08G12/00 - C08G71/00
- C08G73/06—Polycondensates having nitrogen-containing heterocyclic rings in the main chain of the macromolecule
- C08G73/10—Polyimides; Polyester-imides; Polyamide-imides; Polyamide acids or similar polyimide precursors
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G73/00—Macromolecular compounds obtained by reactions forming a linkage containing nitrogen with or without oxygen or carbon in the main chain of the macromolecule, not provided for in groups C08G12/00 - C08G71/00
- C08G73/06—Polycondensates having nitrogen-containing heterocyclic rings in the main chain of the macromolecule
- C08G73/10—Polyimides; Polyester-imides; Polyamide-imides; Polyamide acids or similar polyimide precursors
- C08G73/1039—Polyimides; Polyester-imides; Polyamide-imides; Polyamide acids or similar polyimide precursors comprising halogen-containing substituents
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G73/00—Macromolecular compounds obtained by reactions forming a linkage containing nitrogen with or without oxygen or carbon in the main chain of the macromolecule, not provided for in groups C08G12/00 - C08G71/00
- C08G73/06—Polycondensates having nitrogen-containing heterocyclic rings in the main chain of the macromolecule
- C08G73/10—Polyimides; Polyester-imides; Polyamide-imides; Polyamide acids or similar polyimide precursors
- C08G73/1046—Polyimides containing oxygen in the form of ether bonds in the main chain
- C08G73/105—Polyimides containing oxygen in the form of ether bonds in the main chain with oxygen only in the diamino moiety
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D151/00—Coating compositions based on graft polymers in which the grafted component is obtained by reactions only involving carbon-to-carbon unsaturated bonds; Coating compositions based on derivatives of such polymers
- C09D151/08—Coating compositions based on graft polymers in which the grafted component is obtained by reactions only involving carbon-to-carbon unsaturated bonds; Coating compositions based on derivatives of such polymers grafted on to macromolecular compounds obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds
-
- G—PHYSICS
- G03—PHOTOGRAPHY; CINEMATOGRAPHY; ANALOGOUS TECHNIQUES USING WAVES OTHER THAN OPTICAL WAVES; ELECTROGRAPHY; HOLOGRAPHY
- G03F—PHOTOMECHANICAL PRODUCTION OF TEXTURED OR PATTERNED SURFACES, e.g. FOR PRINTING, FOR PROCESSING OF SEMICONDUCTOR DEVICES; MATERIALS THEREFOR; ORIGINALS THEREFOR; APPARATUS SPECIALLY ADAPTED THEREFOR
- G03F7/00—Photomechanical, e.g. photolithographic, production of textured or patterned surfaces, e.g. printing surfaces; Materials therefor, e.g. comprising photoresists; Apparatus specially adapted therefor
- G03F7/004—Photosensitive materials
-
- G—PHYSICS
- G03—PHOTOGRAPHY; CINEMATOGRAPHY; ANALOGOUS TECHNIQUES USING WAVES OTHER THAN OPTICAL WAVES; ELECTROGRAPHY; HOLOGRAPHY
- G03F—PHOTOMECHANICAL PRODUCTION OF TEXTURED OR PATTERNED SURFACES, e.g. FOR PRINTING, FOR PROCESSING OF SEMICONDUCTOR DEVICES; MATERIALS THEREFOR; ORIGINALS THEREFOR; APPARATUS SPECIALLY ADAPTED THEREFOR
- G03F7/00—Photomechanical, e.g. photolithographic, production of textured or patterned surfaces, e.g. printing surfaces; Materials therefor, e.g. comprising photoresists; Apparatus specially adapted therefor
- G03F7/004—Photosensitive materials
- G03F7/027—Non-macromolecular photopolymerisable compounds having carbon-to-carbon double bonds, e.g. ethylenic compounds
-
- G—PHYSICS
- G03—PHOTOGRAPHY; CINEMATOGRAPHY; ANALOGOUS TECHNIQUES USING WAVES OTHER THAN OPTICAL WAVES; ELECTROGRAPHY; HOLOGRAPHY
- G03F—PHOTOMECHANICAL PRODUCTION OF TEXTURED OR PATTERNED SURFACES, e.g. FOR PRINTING, FOR PROCESSING OF SEMICONDUCTOR DEVICES; MATERIALS THEREFOR; ORIGINALS THEREFOR; APPARATUS SPECIALLY ADAPTED THEREFOR
- G03F7/00—Photomechanical, e.g. photolithographic, production of textured or patterned surfaces, e.g. printing surfaces; Materials therefor, e.g. comprising photoresists; Apparatus specially adapted therefor
- G03F7/004—Photosensitive materials
- G03F7/027—Non-macromolecular photopolymerisable compounds having carbon-to-carbon double bonds, e.g. ethylenic compounds
- G03F7/032—Non-macromolecular photopolymerisable compounds having carbon-to-carbon double bonds, e.g. ethylenic compounds with binders
- G03F7/037—Non-macromolecular photopolymerisable compounds having carbon-to-carbon double bonds, e.g. ethylenic compounds with binders the binders being polyamides or polyimides
-
- G—PHYSICS
- G03—PHOTOGRAPHY; CINEMATOGRAPHY; ANALOGOUS TECHNIQUES USING WAVES OTHER THAN OPTICAL WAVES; ELECTROGRAPHY; HOLOGRAPHY
- G03F—PHOTOMECHANICAL PRODUCTION OF TEXTURED OR PATTERNED SURFACES, e.g. FOR PRINTING, FOR PROCESSING OF SEMICONDUCTOR DEVICES; MATERIALS THEREFOR; ORIGINALS THEREFOR; APPARATUS SPECIALLY ADAPTED THEREFOR
- G03F7/00—Photomechanical, e.g. photolithographic, production of textured or patterned surfaces, e.g. printing surfaces; Materials therefor, e.g. comprising photoresists; Apparatus specially adapted therefor
- G03F7/004—Photosensitive materials
- G03F7/039—Macromolecular compounds which are photodegradable, e.g. positive electron resists
Definitions
- the present invention relates to a photosensitive polyimide resin composition, a resin film, and an electronic device.
- a resin film obtained by exposing a photosensitive polyimide resin composition having excellent heat resistance and insulating properties is widely used.
- Examples of the technique relating to such a photosensitive polyimide resin composition include those described in Patent Documents 1 and 2.
- Patent Document 1 describes a photosensitive resin composition containing an alkali-soluble resin, a radically polymerizable compound, a photopolymerization initiator and a colorant, wherein the alkali-soluble resin is a polyimide, a polyimide precursor, or a polybenzoxazole precursor. And / or a bifunctional or higher functional (meth) acrylic compound containing a copolymer thereof and having a glass transition temperature of 150 ° C. or higher when the radically polymerizable compound is used as a homopolymer, and other tetrafunctional or higher functional compounds.
- a photosensitive resin composition containing the (meth) acrylic compound of the above is described.
- Patent Document 2 includes a precursor of a heterocyclic-containing polymer, a thermobase generator, and a radically polymerizable compound, and the precursor of the heterocyclic ring-containing polymer has a radically polymerizable group, and the above-mentioned radically polymerizable compound is described.
- a photosensitive resin composition comprising at least one radically polymerizable compound selected from the group consisting of a compound having four or more polymerizable functional groups and a compound having three polymerizable functional groups and having a molecular weight of 400 or less. Have been described.
- the photosensitive resin composition described in Patent Document 1 is inferior in thick film developability because the light transmittance is low and the light at the time of exposure does not easily reach the lower part of the film when the film is thick.
- the resin film tends to shrink when the polyimide precursor is dehydrated and closed to form polyimide, so that the pattern shape during thick film development deteriorates or the pattern cracks. Is likely to occur.
- the photosensitive resin composition when the photosensitive resin composition is a thick film, the amount of exposure required for curing is large, so that the pattern shape tends to be more tapered. That is, the composition containing the conventional photosensitive polyimide resin and the polyfunctional radically polymerizable compound has room for improvement in terms of thick film patterning property.
- the present invention has been made in view of the above circumstances, and provides a photosensitive polyimide resin composition having excellent thick film patterning property.
- the present inventors have made extensive studies to solve the above problems. As a result, by combining the closed ring polyimide resin (A) having a specific structure and molecular weight with the polyfunctional radically polymerizable compound (B) having 4 or more and 100 or less radically polymerizable functional groups, the photosensitive is photosensitive. We have found that the thick film patterning property of the polyimide resin composition can be improved, and completed the present invention.
- the photosensitive polyimide resin composition the resin film, and the electronic device shown below are provided.
- R is a tetravalent group having a cyclic structure, an acyclic structure, or a cyclic structure and a non-cyclic structure and having 4 or more and 10 or less carbon atoms.
- A has at least one group selected from the group consisting of an aliphatic hydrocarbon group, an alicyclic hydrocarbon group, an aromatic hydrocarbon group, and an organosiloxane group, and has 2 or more and 39 or less carbon atoms. It is the basis of the price.
- the main chain of A is selected from the group consisting of -O-, -SO 2- , -CO-, -CH 2- , -C (CH 3 ) 2- , -C 2 H 4 O-, and -S-. At least one group may be intervened.
- n indicates the number of repeating units.
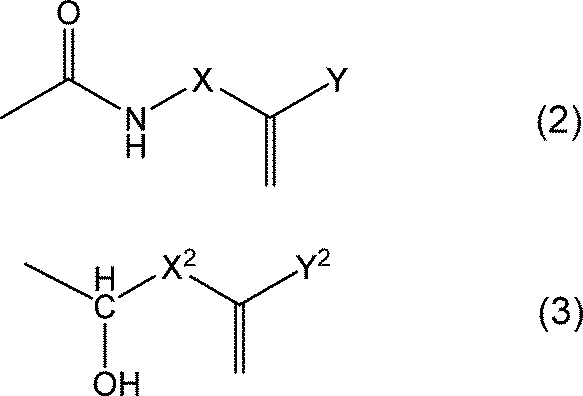
- the end of the above formula (1) is either the group represented by the following formula (2) or the formula (3), or a hydrogen atom, and at least one of the ends is the following formula (2) or the formula (3). It is the group shown.
- X and X 2 are independently groups having 2 or more and 15 or less carbon atoms, and at least one group selected from the group consisting of an ester bond and a double bond is used. You may have.
- Y and Y 2 are independently hydrogen atoms or methyl groups, respectively.
- the content of the closed ring polyimide resin (A) is 30% by mass or more when the total solid content of the photosensitive polyimide resin composition is 100% by mass, according to the above [1] or [2].
- Photosensitive polyimide resin composition [4]
- the content of the polyfunctional radically polymerizable compound (B) is 20 parts by mass or more and 100 parts by mass when the amount of the closed ring polyimide resin (A) contained in the photosensitive polyimide resin composition is 100 parts by mass.
- the photosensitive polyimide resin composition according to any one of the above [1] to [3] below.
- the content of the colorant is less than 10 parts by mass when the amount of the closed ring polyimide resin (A) contained in the photosensitive polyimide resin composition is 100 parts by mass.
- [1] to [4] The photosensitive polyimide resin composition according to any one of.
- [6] The photosensitive polyimide resin composition according to any one of [1] to [5] above, wherein the radically polymerizable functional group contains a (meth) acryloyl group.
- the closed ring polyimide resin (A) is 4,4'-diamino-2,2'-bis (trifluoromethyl) biphenyl, 1- (4-aminophenyl) -2,3-dihydro-1,3,3. -Trimethyl-1H-inden-5-amine, 4,4'-oxybis [3- (trifluoromethyl) benzeneamine], or 1,3-bis [2- (4-aminophenyl) -2-propyl] benzene
- the photosensitive polyimide resin composition according to any one of the above [1] to [9], which comprises at least one unit composed of.
- Polyimide resin composition [12] The photosensitive polyimide resin composition according to any one of the above [1] to [11], which is used for forming an insulating film. [13] A resin film comprising the photosensitive polyimide resin composition according to any one of the above [1] to [12] or a cured product of the photosensitive polyimide resin composition. [14] The resin film [15] according to the above [13], which has a film thickness of 20 ⁇ m or more. An electronic device provided with the resin film according to the above [14].
- the present embodiment The embodiment for carrying out the present invention (hereinafter, simply referred to as "the present embodiment") will be described in detail.
- the following embodiments are examples for explaining the present invention, and do not limit the contents of the present invention.
- the present invention can be appropriately modified and carried out within the scope of the gist thereof.
- the preferable provisions can be arbitrarily adopted, and it can be said that the combination of preferable ones is more preferable.
- the description of "XX to YY" means "XX or more and YY or less”.
- (meth) acrylate in the present embodiment means both “acrylate” and “methacrylate”. The same applies to other similar terms (“(meth) acrylic acid”, “(meth) acryloyl group”, etc.).
- Photosensitive polyimide resin composition The photosensitive polyimide resin composition according to the present embodiment has a structure represented by the following formula (1) and has a weight average molecular weight of 70,000 or less with the closed ring polyimide resin (A). 4. The polyfunctional radically polymerizable compound (B) having 4 or more and 100 or less radically polymerizable functional groups is contained.
- R is a tetravalent group having a cyclic structure, an acyclic structure, or a cyclic structure and an acyclic structure and having 4 or more and 10 or less carbon atoms.
- A has at least one group selected from the group consisting of an aliphatic hydrocarbon group, an alicyclic hydrocarbon group, an aromatic hydrocarbon group, and an organosiloxane group, and has 2 or more and 39 or less carbon atoms. It is the basis of the price.
- the main chain of A is selected from the group consisting of -O-, -SO 2- , -CO-, -CH 2- , -C (CH 3 ) 2- , -C 2 H 4 O-, and -S-.
- n indicates the number of repeating units.
- the end of the above formula (1) is either the group represented by the following formula (2) or the formula (3), or a hydrogen atom, and at least one of the ends is the following formula (2) or the formula (3). It is the group shown.
- X and X 2 are independently groups having 2 or more and 15 or less carbon atoms, and have at least one group selected from the group consisting of an ester bond and a double bond. You may.
- Y and Y 2 are independently hydrogen atoms or methyl groups, respectively.
- the photosensitive polyimide resin composition according to the present embodiment has a structure represented by the above formula (1), and has a weight average molecular weight of 70,000 or less as a closed ring polyimide resin (A) and 4 or more and 100 or less. It is a composition containing a polyfunctional radically polymerizable compound (B) having a radically polymerizable functional group of the above and having excellent thick film patterning property.
- excellent in thick film patterning property means that when the film thickness of the resin film is, for example, a thick film of 20 ⁇ m or more, the pattern shape is good and the developability is excellent.
- a good pattern shape means a shape in which the lower width / upper width of the pattern is, for example, 0.80 or more and 1.20 or less, preferably 0.90 or more and 1.10 or less, and is excellent in developability. It means that there is little residue in the unexposed area after development.
- the photosensitive polyimide resin composition according to the present embodiment has a structure represented by the above formula (1), and has a weight average molecular weight of 70,000 or less as a closed ring polyimide resin (A) and 4 or more and 100 or less.
- the polyfunctional radically polymerizable compound (B) having the radically polymerizable functional group of the above the thick film patterning property is excellent.
- the reason is not clear, but it can be considered as follows. First, the developability is better as the difference in solubility in the developing solution between the exposed part and the unexposed part (hereinafter, also simply referred to as “developer solubility”) is larger. It is important to widen the difference in developer solubility in unexposed areas.
- the closed ring polyimide resin (A) having the structure represented by the above formula (1) and having a weight average molecular weight of 70,000 or less has high developer solubility, and therefore is not yet thickened even if the resin film is thickened. It is considered that the developer solubility of the exposed part is high. Further, since the closed ring polyimide resin (A) having the structure represented by the above formula (1) is excellent in transparency, even if the resin film is thickened, the light at the time of exposure easily reaches the lower part of the film, and the resin film is further formed.
- the cross-linking can be uniformly performed, and further, by combining with the polyfunctional radically polymerizable compound (B), the cross-linking density of the resin film can be improved and the solubility of the developing solution in the exposed portion can be lowered. Further, since the closed ring polyimide resin (A) is a resin for which imidization has already been completed, a dehydration ring closure step is not required. Therefore, it is considered possible to reduce the developer solubility of the exposed portion while suppressing deformation such as curing shrinkage of the resin film during exposure by combining with the polyfunctional radically polymerizable compound (B).
- the photosensitive polyimide resin composition according to the present embodiment by combining the closed ring polyimide resin (A) and the polyfunctional radically polymerizable compound (B), the developing liquid in the exposed portion and the unexposed portion is used. Since the difference in solubility can be increased, the thick film developability can be improved, and further, deformation such as curing shrinkage of the resin film during exposure can be suppressed, so that the thick film patterning property can be improved. It is thought that it was.
- the closed ring polyimide resin (A) has a structure represented by the following formula (1) and has a weight average molecular weight of 70,000 or less.
- R is a tetravalent group having a cyclic structure, an acyclic structure, or a cyclic structure and an acyclic structure and having 4 or more and 10 or less carbon atoms.
- A has at least one group selected from the group consisting of an aliphatic hydrocarbon group, an alicyclic hydrocarbon group, an aromatic hydrocarbon group, and an organosiloxane group, and has 2 or more and 39 or less carbon atoms. It is the basis of the price.
- the main chain of A is selected from the group consisting of -O-, -SO 2- , -CO-, -CH 2- , -C (CH 3 ) 2- , -C 2 H 4 O-, and -S-.
- n indicates the number of repeating units.
- the end of the above formula (1) is either the group represented by the following formula (2) or the formula (3), or a hydrogen atom, and at least one of the ends is the following formula (2) or the formula (3). It is the group shown.
- X and X 2 are independently groups having 2 or more and 15 or less carbon atoms, and have at least one group selected from the group consisting of an ester bond and a double bond. You may.
- Y and Y 2 are independently hydrogen atoms or methyl groups, respectively.
- R in the above formula (1) has at least a cyclic structure
- the cyclic structure includes, for example, cyclohexane, cyclopentane, cyclobutane, bicyclopentane and steric isomers thereof except for four hydrogen atoms.
- examples thereof include tetravalent groups to be formed. More specifically, the tetravalent group includes a group represented by the following structural formula.
- a tetravalent group formed by removing four hydrogen atoms from cyclohexane is preferable.
- a in the above formula (1) has at least one group selected from the group consisting of an aliphatic hydrocarbon group, an alicyclic hydrocarbon group, an aromatic hydrocarbon group, and an organosiloxane group, and has 2 carbon atoms. It is a divalent group of 39 or more and 39 or less.
- the main chain of A is selected from the group consisting of -O-, -SO 2- , -CO-, -CH 2- , -C (CH 3 ) 2- , -C 2 H 4 O-, and -S-. At least one group may be intervened.
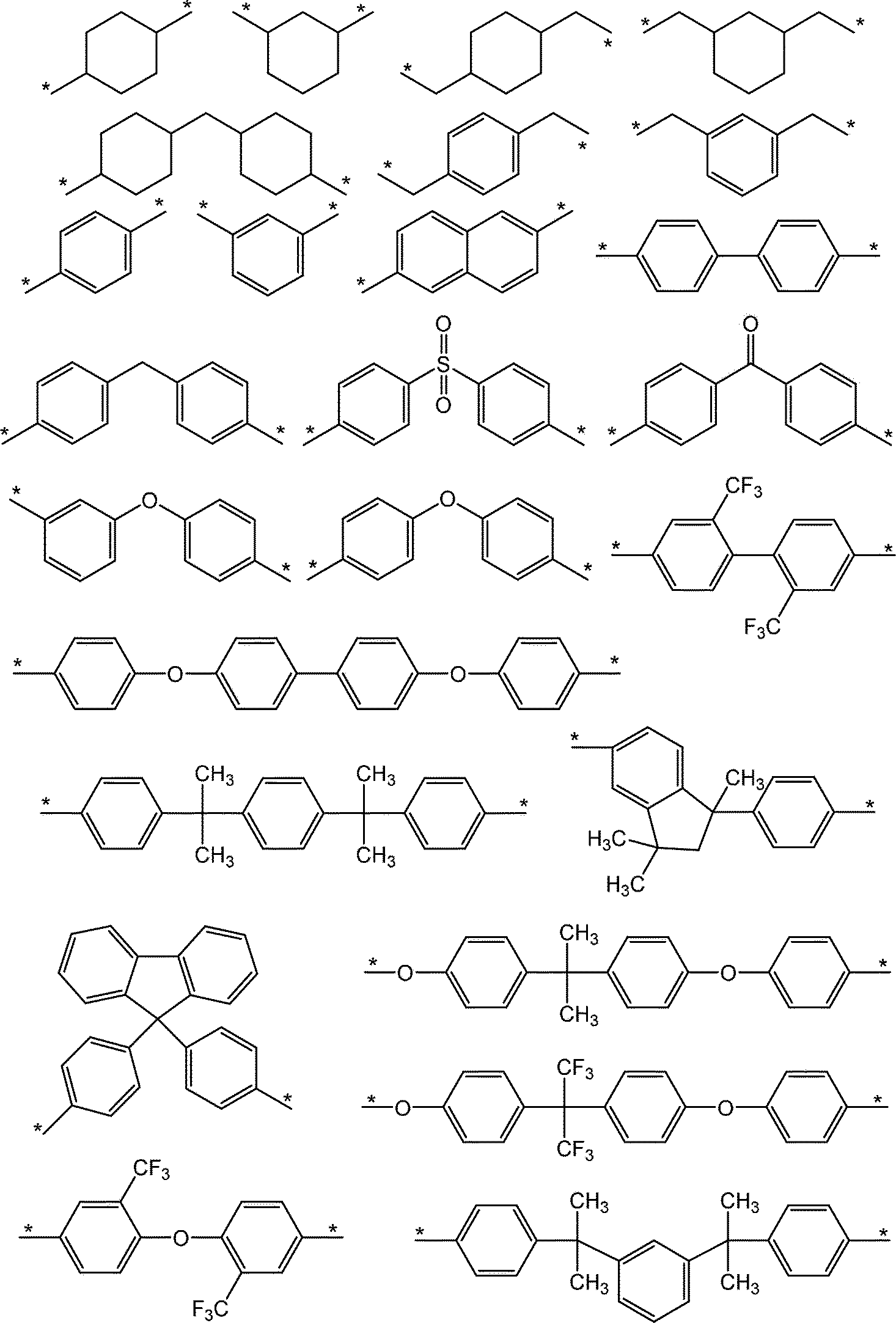
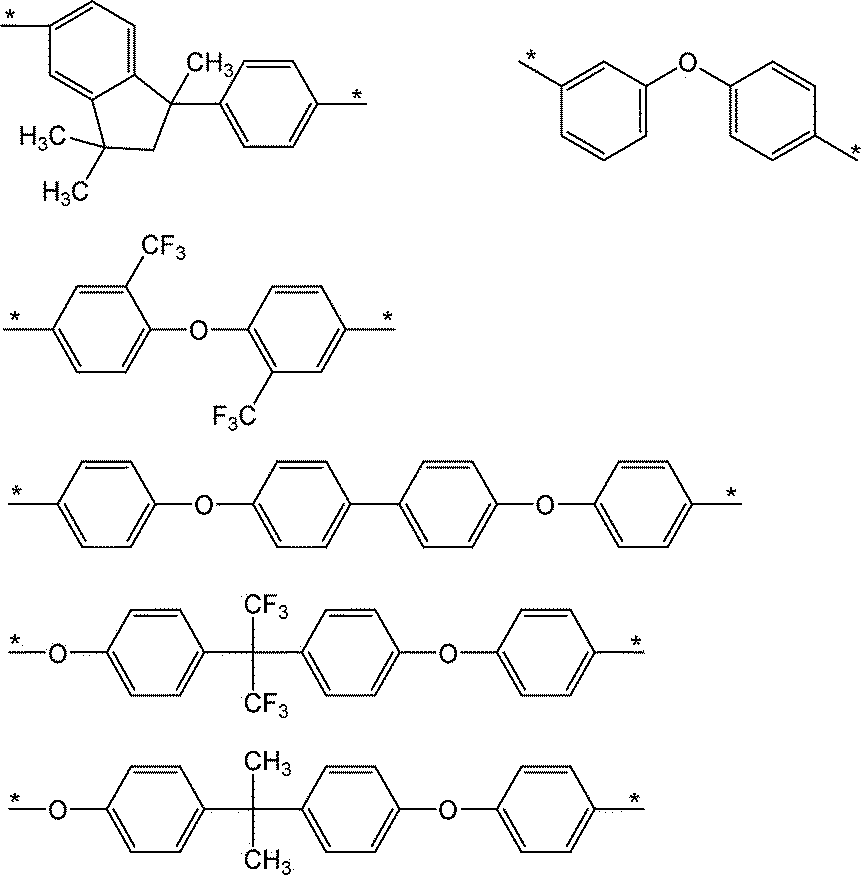
- A is cyclohexane, dicyclohexylmethane, dimethylcyclohexane, isophorone, norbornan and alkyl-substituted products thereof, and halogen-substituted products thereof; benzene, naphthalene, biphenyl, diphenylmethane, diphenyl ether, diphenyl sulfone, benzophenone and these. Alkyl-substituted products of the above, as well as these halogen-substituted products; divalent groups formed by removing two hydrogen atoms from compounds such as organo (poly) siloxane.
- A preferably has a cyclic structure, and more preferably has at least one selected from an alicyclic hydrocarbon group and an aromatic ring. It is more preferable that A has an aromatic ring as an aromatic hydrocarbon group. More specifically, a divalent group having 6 or more and 27 or less carbon atoms represented by the following structural formula is preferably mentioned.
- the divalent group having 2 or more and 39 or less carbon atoms indicated by A contains at least one selected from the group consisting of the structures shown below.
- the divalent group having 2 or more and 39 or less carbon atoms indicated by A includes at least one group (IA) selected from the group consisting of the structures shown below.
- the divalent group having 2 or more and 39 or less carbon atoms indicated by A includes at least one group (Ib) selected from the group consisting of the structures shown below.
- the closed ring polyimide resin (A) according to the present embodiment has at least one configuration selected from the above (Ia), (Ib) and (Ic) as A in the above formula (1).
- the unit ratio is preferably 60 mol% or more from the viewpoint of developer solubility.
- the ratio of at least one structural unit selected from the above (Ia), (Ib) and (Ic) in A in the above formula (1) is more preferably 70 mol% or more, still more preferably 70 mol% or more. It is 80 mol% or more, more preferably 95 mol% or more, still more preferably 100 mol%. Above all, it is preferable to include the structural unit derived from the diamine represented by the formula (Ic) in the above ratio.
- N indicating the number of repeating units of the structural unit represented by the above formula (1) is preferably 5 or more, more preferably 10 or more, still more preferably 15 or more, from the viewpoint of further improving the mechanical properties of the obtained resin film. From the viewpoint of further improving the developer solubility of the obtained photosensitive polyimide resin composition, it is preferably 250 or less, more preferably 200 or less, still more preferably 150 or less.
- the closed ring polyimide resin (A) has either a group represented by the above formula (2) or the above formula (3) or a hydrogen atom at the end, and at least one of the ends is the above formula (2). ) Or the group represented by the formula (3).
- the closed ring polyimide resin (A) may have one end having a structure represented by the above formula (2) or the above formula (3), or both ends may be represented by the above formula (2) or the above formula (3). It may have a structure to be used.
- the group represented by X or X2 in the above formula ( 2 ) or formula (3) is a group having 2 or more carbon atoms and 15 or less carbon atoms, and is at least one selected from the group consisting of an ester bond and a double bond. It may have a group.
- the group represented by Y or Y 2 is a hydrogen atom or a methyl group.
- the structure represented by the above formula (2) or formula (3) more specifically corresponds to a structure obtained by reacting a terminal amine of a polyimide resin with a functional group-containing compound.
- the functional group-containing compound include compounds having an isocyanate group or an epoxy group and a (meth) acrylic group.
- the compound include 2-isocyanatoethyl methacrylate, 2-isocyanatoethyl acrylate, 1,1-bis (acryloyloxymethyl) ethyl isocyanate, glycidyl methacrylate, glycidyl acrylate, and allyl glycidyl ether.
- the structure represented by the above formula (2) or the formula (3) may have a structure in which the compound and the amine terminal of the polyimide resin are reacted.
- the weight average molecular weight of the closed ring polyimide resin (A) is 70,000 or less, preferably 60,000 or less, more preferably 50,000 or less, from the viewpoint of improving the developer solubility of the photosensitive polyimide resin composition. It is more preferably 45,000 or less, further preferably 40,000 or less, still more preferably 37,000 or less, and preferably 5,000 or more, more preferably 5,000 or more, from the viewpoint of further improving the mechanical properties of the obtained resin film. It is 10,000 or more, more preferably 13,000 or more, still more preferably 15,000 or more.
- the weight average molecular weight of the closed ring polyimide resin (A) is in the above range, the residual film ratio of the unexposed portion can be further lowered, and the developability of the photosensitive polyimide resin composition can be further improved.
- the weight average molecular weight is a polystyrene-equivalent weight average molecular weight.
- the closed ring polyimide resin (A) can be obtained by reacting the diamine component described in detail below with the tetracarboxylic acid component.
- diamine component examples include diamines, diisocyanates and diaminodisilanes, and diamines are preferable.
- the diamine content in the diamine component used as a raw material is preferably 50 mol% or more, more preferably 70 mol% or more, further preferably 90 mol% or more, still more preferably 95 mol% or more, and preferably 100 mol%. It is as follows.
- the diamine may be either an aliphatic diamine or an aromatic diamine, or a mixture thereof.
- the "aromatic diamine” represents a diamine in which an amino group is directly bonded to an aromatic ring, and a part of its structure contains an aliphatic group, an alicyclic group, and other substituents. May be good.
- the "aliphatic diamine” represents a diamine in which an amino group is directly bonded to an aliphatic group or an alicyclic group, and an aromatic group or other substituent may be contained as a part of the structure thereof.
- Examples of the aliphatic diamine include 4,4'-diaminodicyclohexylmethane, ethylenediamine, hexamethylenediamine, polyethylene glycol bis (3-aminopropyl) ether, polypropylene glycol bis (3-aminopropyl) ether, and 1,3-.
- Examples thereof include bis (aminomethyl) cyclohexane, 1,4-bis (aminomethyl) cyclohexane, m-xylylene diamine, p-xylylene diamine, isophorone diamine, norbornan diamine and siloxane diamines.
- aromatic diamine examples include 4,4'-diaminodiphenyl ether, 4,4'-diaminodiphenylmethane, 4,4'-diaminodiphenylsulfone, m-phenylenediamine, p-phenylenediamine, diaminobenzophenone, 2,6.
- the diamine component is 4,4'-diamino-2,2'-bis (trifluoromethyl) biphenyl, 1- (4-aminophenyl) -2,3-dihydro-1,3,3-trimethyl-1H-. At least selected from inden-5-amine, 4,4'-oxybis [3- (trifluoromethyl) benzeneamine], and 1,3-bis [2- (4-aminophenyl) -2-propyl] benzene. It is preferable to include one kind. By containing at least these diamines as the diamine component, the light transmittance and the developer solubility of the obtained photosensitive polyimide resin composition at a specific wavelength can be further improved.
- the closed ring polyimide resin (A) is 4,4'-diamino-2,2'-bis (trifluoromethyl) biphenyl, 1- (4-aminophenyl) -2,3-dihydro-1. , 3,3-trimethyl-1H-inden-5-amine, 4,4'-oxybis [3- (trifluoromethyl) benzeneamine] or 1,3-bis [2- (4-aminophenyl) -2- It preferably contains at least one unit composed of [propyl] benzene.
- tetracarboxylic acid component examples include cyclohexanetetracarboxylic acid, cyclohexanetetracarboxylic acid esters, cyclohexanetetracarboxylic acid dianhydride, cyclobutanetetracarboxylic acid, cyclobutanetetracarboxylic acid esters, cyclobutanetetracarboxylic acid dianhydride, and cyclo.
- the tetracarboxylic acid component preferably contains at least one selected from cyclohexanetetracarboxylic acid dianhydride, cyclobutanetetracarboxylic acid dianhydride, and cyclopentanetetracarboxylic acid dianhydride, and cyclohexanetetracarboxylic acid. More preferably, it contains an acid dianhydride.
- the various tetracarboxylic acid components described above contain positional isomers.
- tetracarboxylic acid component is 1,2,4,5-cyclohexanetetracarboxylic acid, 1,2,4,5-cyclohexanetetracarboxylic acid dianhydride, 1,2,4,5- Cyclohexanetetracarboxylic acid methyl ester, 1,2,3,4-butanetetracarboxylic acid, 1,2,3,4-butanetetracarboxylic acid dianhydride, 1,2,3,4-butanetetracarboxylic acid methyl ester , 1,2,3,4-cyclobutanetetracarboxylic acid, 1,2,3,4-cyclobutanetetracarboxylic acid dianhydride, 1,2,3,4-cyclobutanetetracarboxylic acid methyl ester, 1,2,4 , 5-Cyclopentanetetracarboxylic acid, 1,2,4,5-cyclopentanetetracarboxylic acid dianhydride, 1,2,4,5-cyclopentanetetracarboxylic acid dianhydride
- the tetracarboxylic acid component is 1,2,4,5-cyclohexanetetra because it is easy to increase the molecular weight when producing a polyimide resin and it is advantageous in that a flexible resin film can be easily obtained.
- the tetracarboxylic acid component may contain another tetracarboxylic acid or a derivative thereof, for example, as long as the flexibility and thermocompression bonding property of the resin film are not impaired.
- these other tetracarboxylic acids or derivatives thereof include pyromellitic acid, 3,3', 4,4'-biphenyltetracarboxylic acid, 2,3,3', 4'-biphenyltetracarboxylic acid, 2, 2-Bis (3,4-dicarboxyphenyl) propane, 2,2-bis (2,3-dicarboxyphenyl) propane, 2,2-bis (3,4-dicarboxyphenyl) -1,1,1 , 3,3,3-hexafluoropropane, 2,2-bis (2,3-dicarboxyphenyl) -1,1,1,3,3,3-hexafluoropropane, bis (3,4-dicarboxyphenyl) Phenyl) sulfone, bis (3,
- the light transmittance at a wavelength of 365 nm when the closed ring polyimide resin (A) according to the present embodiment has a solid content concentration of 3% by mass is a viewpoint of more uniformly cross-linking the resin film and further improving the thick film patterning property. Therefore, it is preferably 80% or more, more preferably 85% or more, still more preferably 88% or more, still more preferably 90% or more.
- the closed ring polyimide resin (A) according to the present embodiment can be obtained, for example, by a production method including the following steps (1) and (2).
- Step (1) The tetracarboxylic acid component and the diamine component are reacted to obtain a polyimide resin having an amino group at the terminal.
- Step (2) The polyimide resin having an amino group at the terminal obtained in the above step (1) is reacted with the above functional group-containing compound (a compound having an isocyanate group or an epoxy group and a (meth) acrylic group). Let me.
- Step (1) the tetracarboxylic acid and the diamine component are reacted to obtain a polyimide resin having an amino group at the terminal.
- the organic solvent used for reacting the tetracarboxylic acid component and the diamine component is not particularly limited, but is an organic solvent containing at least one selected from the group consisting of, for example, cyclic ethers, cyclic ketones, cyclic esters, amides and ureas. Is preferable.
- the suitable solvent are not particularly limited, but ⁇ -butyrolactone, N, N-dimethylacetamide, N, N-dimethylformamide, N-methyl-2-pyrrolidone, dimethyl sulfoxide, hexamethylphosphoramide, cyclo.
- aprotonic polar organic solvents such as pentanone, cyclohexanone, 1,3-dioxolane, 1,4-dioxane, tetramethylurea and tetrahydrofuran can be mentioned.
- ⁇ -butyrolactone, N, N-dimethylacetamide, N, N-dimethylformamide and N-methyl-2-pyrrolidone is more preferable.
- An imidization catalyst can be used when the tetracarboxylic acid component and the diamine component are reacted in the step (1).
- a tertiary amine compound is preferable, and specifically, trimethylamine, triethylamine, tripropylamine, tributylamine, triethanolamine, N, N-dimethylethanolamine, N, N-diethylethanolamine and triethylenediamine.
- the reaction temperature in the step (1) is, for example, 160 ° C. or higher and 230 ° C. or lower, preferably 170 ° C. or higher and 210 ° C. or lower, and more preferably 180 ° C. or higher and 200 ° C. or lower.
- the reaction temperature in the step (1) is equal to or higher than the above lower limit value, imidization and molecular weight increase can be promoted more effectively.
- the reaction temperature in the step (1) is not more than the above upper limit value, the solution viscosity can be maintained more appropriately, and problems such as the resin being burnt on the wall surface of the reaction vessel can be further avoided.
- an azeotropic dehydrating agent such as toluene or xylene may be used.
- the reaction pressure is usually normal pressure, but if necessary, the reaction can be carried out under pressure.
- the holding time of the reaction temperature is preferably 1 hour or longer, more preferably 3 hours or longer. When the holding time of the reaction temperature is equal to or longer than the above lower limit value, imidization and molecular weight increase can be promoted more effectively. There is no particular upper limit to the reaction time, but the reaction time can be, for example, 10 hours or less.
- the step (1) it is preferable to react the tetracarboxylic acid component “A mol” and the diamine component “B mol” in the range of 0.80 ⁇ A / B ⁇ 0.99, preferably 0.85 ⁇ A. It is more preferable to react in the range of / B ⁇ 0.97.
- a / B ⁇ 0.99
- it is possible to make the end of the polyimide an excess of diamine a polyimide resin having an amino group at the end can be obtained, and the molecular weight has sufficient developer solubility.
- Polyimide resin can be obtained.
- 0.80 ⁇ A / B a polyimide resin having a molecular weight that exhibits sufficient flexibility can be obtained.
- a / B approaches 1.0, a polyimide resin having a high molecular weight can be obtained. Therefore, by appropriately adjusting the A / B, a polyimide resin having a desired molecular weight can be obtained.
- the step (2) is a step of modifying the end of the polyimide resin obtained in the above step (1). Specifically, as described above, the polyimide is reacted with the functional group-containing compound (a compound having an isocyanate group or an epoxy group and a (meth) acrylic group) to have a (meth) acrylic group at the terminal. Obtain a polyimide resin.
- the functional group-containing compound a compound having an isocyanate group or an epoxy group and a (meth) acrylic group
- the functional group-containing compound that modifies the terminal of the polyimide resin is a compound having an isocyanate group or an epoxy group and a (meth) acrylic group, and specifically, 2-isocyanatoethyl methacrylate and 2-isocyanatoethyl. Examples thereof include acrylate, 1,1-bis (acryloyloxymethyl) ethyl isocyanate, glycidyl methacrylate, glycidyl acrylate, and allyl glycidyl ether. These functional group-containing compounds may be used alone or in combination of two or more.
- the functional group-containing compound is preferably used at a ratio of 0.1 to 30 mol times that of the polyimide resin having an amino group at the terminal.
- the reaction temperature in the step (2) is preferably in the range of 30 ° C. or higher and 100 ° C. or lower, and the reaction time is preferably 1 hour or longer and 10 hours or lower.
- the reaction may be carried out as it is, or may be reacted in the presence of a catalyst if necessary.
- the catalyst include amine compounds such as triethylamine and organophosphorus compounds such as triphenylphosphine, which may be used alone or in combination of two or more.
- a polymerization inhibitor may be used to suppress side reactions during the reaction. Examples of the polymerization inhibitor include hydroquinone, hydroquinone monomethyl ether, and methylhydroquinone, and these may be used alone or in combination of two or more.
- the content of the closed ring polyimide resin (A) in the photosensitive polyimide resin composition according to the present embodiment further improves the thick film patterning property when the total solid content of the photosensitive polyimide resin composition is 100% by mass. From the viewpoint of the above, it is preferably 30% by mass or more, more preferably 40% by mass or more, further preferably 50% by mass or more, still more preferably 60% by mass or more, and preferably 90% by mass or less, more preferably 80% by mass. % Or less, more preferably 70% by mass or less.
- the total solid content of the photosensitive polyimide resin composition is a component that remains as a solid content when the photosensitive polyimide resin composition is cured, and is volatilized by heating, for example, a solvent or the like. Ingredients are excluded.
- the components incorporated into the resin film when heat-cured are included in the total solid content.
- the polyfunctional radically polymerizable compound (B) has 4 or more and 100 or less radically polymerizable functional groups.
- the polyfunctional radically polymerizable compound (B) contains two or more kinds of polyfunctional radically polymerizable compounds having different numbers of radically polymerizable functional groups, the polyfunctional radically polymerizable compound (B) is used.
- the number of radically polymerizable functional groups a weighted average value of the number of radically polymerizable functional groups of each polyfunctional radically polymerizable compound can be adopted.
- the number of radically polymerizable functional groups of the polyfunctional radically polymerizable compound (B) according to the present embodiment is 4 or more and 100 or less, but is preferably 5 or more from the viewpoint of further improving the thick film patterning property.
- the number is preferably 50 or less, more preferably 30 or less, still more preferably 20 or less, still more preferably 15 or less.
- Examples of the radically polymerizable functional group include a (meth) acryloyl group, a vinyl group and the like, and a (meth) acryloyl group is preferable.
- Examples of the polyfunctional radically polymerizable compound constituting the polyfunctional radically polymerizable compound (B) include tricyclodecanedimethanol di (meth) acrylate, polypropylene glycol di (meth) acrylate, and trimethylolpropane tri (meth) acrylate.
- polyglycerin-based (meth) acrylate refers to a compound having a polyglycerin skeleton and a (meth) acryloyl group.
- examples of the polyglycerin-based (meth) acrylate include SA-TE6, SA-TE60, and SA-ZE12 manufactured by Sakamoto Yakuhin Kogyo Co., Ltd.
- ethoxylated trimethylolpropane tri (meth) acrylate examples include ethoxylated (3) trimethylolpropane triacrylate and ethoxylated (6) trimethylolpropane triacrylate and ethoxylated (15) trimethylolpropane manufactured by Arkema Co., Ltd. Triacrylate or the like can be used.
- the dendrimer (meth) acrylate refers to a polyfunctional (meth) acrylate having a dendrimer structure (including a hyperbranched structure).
- Examples of the dendrimer (meth) acrylate include Viscoat 1000, Viscoat 1020, and STAR-501 (SIRIUS-501, SUBARU-501) manufactured by Osaka Organic Chemical Industry Co., Ltd.
- the biscoat 1000 and the biscoat 1020 are mainly composed of a dendrimer type polyester acrylate having an acrylate group at the terminal.
- the molecular weight of the biscoat 1000 is about 1000 to 2000, and the molecular weight of the viscoat 1020 is about 1000 to 3000.
- STAR-501 contains a core derived from dipentaerythritol and contains a dipentaerythritol hexaacrylate-linked multi-branched polyacrylate having an acrylate group at the terminal as a main component.
- the molecular weight of STAR501 is about 16000 to 24000.
- the content of the polyfunctional radically polymerizable compound (B) in the photosensitive polyimide resin composition according to the present embodiment is 100 parts by mass based on the amount of the closed ring polyimide resin (A) contained in the photosensitive polyimide resin composition.
- the shape of the obtained thick film pattern is preferably 20 parts by mass or more, more preferably 30 parts by mass or more, still more preferably 40 parts by mass or more.
- the viewpoint of further suppressing stickiness of the thick film pattern preferably 100 parts by mass or less, more preferably 90 parts by mass or less, still more preferably 80 parts by mass or less, still more preferably 70 parts by mass or less, further. It is preferably 60 parts by mass or less.
- the photosensitive polyimide resin composition according to the present embodiment contains, for example, a photopolymerization initiator, a solvent, and an adhesion improver as other components other than the closed ring polyimide resin (A) and the polyfunctional radically polymerizable compound (B). It is preferable to further contain at least one selected from the group consisting of a surface conditioner and a sensitizer, and more preferably to further contain at least one selected from the group consisting of a photopolymerization initiator and a solvent.
- the photopolymerization initiator is not particularly limited, and known photopolymerization initiators can be used.
- photopolymerization initiators may be used alone or in combination of two or more.
- the content of the photopolymerization initiator in the photosensitive polyimide resin composition according to the present embodiment is, for example, 0 when the amount of the closed ring polyimide resin (A) contained in the photosensitive polyimide resin composition is 100 parts by mass. .1 part by mass or more and 10 parts by mass or less.
- solvent an aprotic polar solvent is desirable from the viewpoint of solubility.
- the solvent include N-methyl-2-pyrrolidone, N-acetyl-2-pyrrolidone, N-benzyl-2-pyrrolidone, N, N-dimethylformamide, N, N-dimethylacetamide, dimethyl sulfoxide, and hexamethylphospho.
- examples thereof include rutriamide, N-acetyl- ⁇ -caprolactum, dimethylimidazolidonene, diethylene glycol dimethyl ether, triethylene glycol dimethyl ether and ⁇ -butyrolactone. These solvents may be used alone or in combination of two or more.
- solvents such as toluene, xylene, diethyl ketone, methoxybenzene and cyclopentanone may be mixed as long as they do not adversely affect the solubility of the polymer.
- solvents such as toluene, xylene, diethyl ketone, methoxybenzene and cyclopentanone may be mixed as long as they do not adversely affect the solubility of the polymer.
- the photosensitive polyimide resin composition according to the present embodiment can be used in a solution (varnish) state, and the film forming property can be improved.
- the adhesion improver is not particularly limited, and known ones can be used, and an amino group-containing silane coupling agent, an epoxy group-containing silane coupling agent, a mercapto group-containing silane coupling agent, and a (meth) acrylic group-containing silane coupling agent can be used.
- Known coupling agents such as silane coupling agents such as agents, titanate coupling agents, and aluminate coupling agents can be used.
- the coupling agent include KP-390, KA-1003, KBM-1003, KBE-1003, KBM-303, KBM-403, KBE-402, KBE-403, KBM-1403, KBM-502, KBM-.
- the content of the adhesion improver in the photosensitive polyimide resin composition according to the present embodiment is, for example, 0 when the amount of the closed ring polyimide resin (A) contained in the photosensitive polyimide resin composition is 100 parts by mass. It is 0005 parts by mass or more and 20 parts by mass or less.
- the surface conditioner is not particularly limited, and known ones can be used.
- a silicon-based surface conditioner an acrylic surface conditioner, a fluorine-based surface conditioner, a nonionic surface conditioner, a cationic surface conditioner, and an anionic surface conditioner can be used.
- Various surface conditioners such as surface conditioners can be used. These may be used alone or in combination of two or more.
- the content of the surface conditioner in the photosensitive polyimide resin composition according to the present embodiment is, for example, 0. It is 001 parts by mass or more and 20 parts by mass or less.
- the sensitizer is not particularly limited, and known ones can be used.
- an amino group-containing sensitizer can be mentioned, and a compound having an amino group and a phenyl group in the same molecule can be preferably exemplified.
- the content of the colorant in the photosensitive polyimide resin composition according to the present embodiment is the resin obtained when the amount of the closed ring polyimide resin (A) contained in the photosensitive polyimide resin composition is 100 parts by mass. From the viewpoint of further improving the transparency of the film and further improving the developability of the thick film, it is preferably less than 10 parts by mass, more preferably less than 5 parts by mass, still more preferably less than 1 part by mass, still more preferably 0.5 parts by mass. It is more preferably less than a portion, more preferably less than 0.1 part by mass, and the photosensitive polyimide resin composition according to the present embodiment does not contain a coloring agent.
- the colorant refers to an organic pigment, an inorganic pigment or a dye generally used in the field of photosensitive materials.
- the photosensitive polyimide resin composition according to the present embodiment is not particularly limited, and for example, the closed ring polyimide resin (A) and the polyfunctional radically polymerizable compound (B) are, if necessary, a photopolymerization initiator, a solvent, and the like. It can be obtained by mixing at least one selected from the group consisting of an adhesion improver, a surface conditioner, and a sensitizer.
- the photosensitive polyimide resin composition according to the present embodiment can be used, for example, to form a resin film such as a resist used in manufacturing an electronic device or a permanent film (cured film) constituting the electronic device.
- the resin film according to the present embodiment is made of a photosensitive polyimide resin composition or a cured product of the photosensitive polyimide resin composition.
- the resist is composed of, for example, a resin film obtained by applying the photosensitive polyimide resin composition according to the present embodiment on a substrate and removing a solvent as needed.
- the permanent film is composed of a cured film obtained by exposing and developing the resin film, patterning it into a desired shape, and then curing it by heat treatment or the like.
- the permanent film can be suitably used as an insulating film such as a surface protective film and an interlayer insulating film.
- the surface protective film is formed on the surface of an electronic component or electronic device or the surface of wiring of a wiring board, and refers to an insulating film for protecting the surface, and the type thereof is not particularly limited.
- Examples of such a surface protective film include a passivation film or buffer coat layer provided on a semiconductor element, or a cover coat provided on a flexible substrate.
- the interlayer insulating film refers to an insulating film provided in a multilayer structure, and the type thereof is not particularly limited.
- the interlayer film include those used in semiconductor device applications such as an interlayer insulating film constituting a multilayer wiring structure of a semiconductor element, a build-up layer or a core layer constituting a wiring substrate, and the like.
- the interlayer film for example, a flattening film covering a thin film transistor in a display device, a liquid crystal alignment film, a protrusion provided on a color filter substrate of the liquid crystal display device, a partition wall for forming a cathode of an organic EL element, or the like. Some are used in display device applications.
- the method for applying the photosensitive polyimide resin composition according to the present embodiment to the substrate is not particularly limited, and for example, an inkjet method, a spin coating method, a casting method, a micro gravure method, a gravure coating method, a bar coating method, and the like. Examples thereof include a roll coating method, a wire bar coating method, a dip coating method, a spray coating method, a screen printing method, a flexographic printing method, and a die coating method.
- the solid content concentration of the photosensitive polyimide resin composition according to the present embodiment When applied onto a substrate, it is preferable to adjust the solid content concentration of the photosensitive polyimide resin composition according to the present embodiment so as to be in the range of 5 to 50% by mass.
- an aprotic polar solvent is preferable from the viewpoint of solubility. Specifically, N-methyl-2-pyrrolidone, N-acetyl-2-pyrrolidone, N-benzyl-2-pyrrolidone, N, N-dimethylformamide, N, N-dimethylacetamide, dimethyl sulfoxide, hexamethylphosphortri.
- Suitable examples include amide, N-acetyl- ⁇ -caprolactam, dimethylimidazolidonene, diethylene glycol dimethyl ether, triethylene glycol dimethyl ether, ⁇ -butyrolactone and the like. These solvents may be used alone or in combination of two or more. In order to further improve the coatability, solvents such as toluene, xylene, diethyl ketone, methoxybenzene and cyclopentanone may be mixed as long as they do not adversely affect the solubility of the polymer.
- Examples of the base material include glass, silicon wafers, metal foils, plastic films and the like. Among the above base materials, silicon wafers and copper foils are preferable.
- the exposure of the film may be performed, for example, by irradiating a film made of a photosensitive polyimide resin composition formed on a substrate with light (usually using ultraviolet rays) through a photomask having a predetermined pattern. can.
- the exposed film has a portion where light is blocked by a photomask and a portion irradiated with light, that is, an exposed portion and an unexposed portion.
- the polyimide resin in the photosensitive polyimide resin composition is crosslinked to form a crosslinked polyimide film, and a pattern is formed by the next developing step.
- the polyimide resin since the polyimide resin is not crosslinked in the unexposed portion, it becomes an uncrosslinked polyimide film that is dissolved and removed by development.
- the ultraviolet irradiation amount is preferably 100 to 8,000 mJ / cm 2 and more preferably 200 to 6,000 mJ / cm 2 as the integrated irradiation amount.
- an organic solvent as the developing solution.
- the developer is not particularly limited as long as it can dissolve the photosensitive polyimide resin composition according to the present embodiment. Specifically, N-methyl-2-pyrrolidone, N-acetyl-2-pyrrolidone, N-benzyl-2-pyrrolidone, N, N-dimethylformamide, N, N-dimethylacetamide, dimethyl sulfoxide, hexamethylphosphortri.
- Preferable examples include amide, N-acetyl- ⁇ -caprolactam, dimethylimidazolidonene, diethylene glycol dimethyl ether, triethylene glycol dimethyl ether, ⁇ -butyrolactone and the like. These developers may be used alone or in combination of two or more.
- the relief pattern formed by development is washed with a rinsing solution to remove the developer.
- a rinsing solution include methanol, ethanol, isopropyl alcohol, and water, which are miscible with the developing solution.
- the relief pattern obtained by the above-mentioned treatment is heat-treated at a temperature selected from the range of 80 to 250 ° C., the solvent is dried, and the photosensitive polyimide resin composition according to the present embodiment is cured. Pattern) can be obtained.
- a photosensitive polyimide resin composition having excellent developability that is, a photosensitive polyimide resin composition in which the exposed portion is sufficiently cured and the unexposed portion is sufficiently removed, the obtained relief pattern can be obtained with high resolution. Can be done.
- the line represents the pattern width and the space represents the spacing between adjacent patterns.
- the fact that the line / space 30 ⁇ m / 30 ⁇ m patterning is possible means that “a pattern having no residue in the space portion (unexposed portion) can be obtained”.
- the film thickness of the resin film according to the present embodiment is preferably 20 ⁇ m or more, more preferably 25 ⁇ m or more, further preferably 30 ⁇ m or more, and preferably 85 ⁇ m or less, more preferably 60 ⁇ m or less.
- the film thickness is in the above range, it can be used as an excellent insulating film.
- the thicker the film that is, the more photosensitive polyimide resin composition to be applied to the substrate), the more often problems occur in the developer solubility of the polyimide resin.
- the closed ring polyimide resin (A) having a specific structure and a specific terminal structure and having a specific molecular weight range, and a plurality of radically polymerizable functional groups having 4 or more and 100 or less.
- the resin film according to the present embodiment can be suitably used, for example, in an insulating film application where a high voltage is expected to be applied.
- the resin film obtained from the photosensitive polyimide resin composition according to the present embodiment containing the closed ring polyimide resin (A) and the polyfunctional radically polymerizable compound (B) can effectively suppress the generation of cracks and the like. It can be made and has excellent physical properties.
- the electronic device according to the present embodiment includes the resin film according to the present embodiment.
- the electronic device according to the present embodiment is not particularly limited as long as it includes a resin film formed by the photosensitive polyimide resin composition according to the present embodiment.
- the resin film according to the present embodiment is flattened.
- Semiconductor device Examples thereof include a semiconductor device using the resin film according to the present embodiment as a build-up layer or a core layer constituting a wiring substrate.
- the semiconductor device is preferable as the electronic device according to the present embodiment.
- the electronic device according to the present embodiment can be manufactured based on known information except that the resin film according to the present embodiment is used. Since the electronic device according to the present embodiment includes a resin film having a good patterning shape, dielectric breakdown and the like are unlikely to occur, and the electronic device is excellent in reliability.
- Weight average molecular weight (Mw) Mw was determined by GPC analysis.
- the equipment and analysis conditions used for the analysis are as follows.
- a photosensitive polyimide resin composition varnish was prepared in Examples and Comparative Examples described below.
- the varnishes obtained in each Example and Comparative Example were applied on a silicon wafer with a spin coater so that the film thickness after removing the solvent was 30 ⁇ m, and then heated at 100 ° C. for 5 minutes to remove the solvent.
- the unexposed part is removed by spraying ⁇ -butyrolactone as a developer for 30 seconds, washed with methanol as a rinsing solution, and placed under air flow. The solvent was removed.
- Example 1 10.0 g of the closed ring polyimide resin (A1) obtained in Synthesis Example 1 was dissolved in 12.2 g of GBL, and a mixture of dipentaerythritol pentaacrylate and dipentaerythritol hexaacrylate (DIPE-A, Shin-Nakamura Chemical Industry Co., Ltd.) , Number of radically polymerizable functional groups: 5.5) 5.0 g, 1-hydroxycyclohexylphenylketone (manufactured by BASF, Omnirad 184) 0.15 g, bis (2,4,6-trimethylbenzoyl) phenylphosphine oxide (manufactured by BASF) , Omnirad819) 0.35g, LE-304 (Kyoeisha Chemical Co., Ltd., silicon-based surface conditioner) 0.01g as a surface conditioner, KP-390 (Shinetsu Chemical Industry Co., Ltd., paint additive) as an adhe
- DIPE-A dipentaery
- This photosensitive polyimide resin composition varnish was applied onto a silicon wafer so that the film thickness after drying was 30 ⁇ m, and dried at 100 ° C. for 5 minutes.
- a mask aligner (MA-10B) manufactured by Mikasa Co., Ltd. was used to irradiate and expose the light of a high-pressure mercury lamp whose wavelength was less than 365 nm with an integrated irradiation amount of 4000 mJ / cm 2 (calculated from the illuminance of 365 nm).
- a pattern with a space of 30 ⁇ m / 30 ⁇ m was prepared.
- Examples 2 to 4 and Comparative Examples 1 to 3 A photosensitive resin composition was prepared in the same manner as in Example 1 above, and its characteristics were evaluated, except that the formulations and integrated irradiation doses shown in Table 1 below were used. The results are shown in Table 1. However, in Examples 1 to 4 and Comparative Examples 1 and 2, the integrated irradiation amount (exposure amount), which was the most excellent in developability when exposed with a plurality of different integrated irradiation amounts, was adopted. Comparative Example 3 is an example in which the integrated irradiation amount is changed in Comparative Example 2. The unit of the blending amount shown in Table 1 is "g".
- the polyfunctional radically polymerizable compounds used in Examples and Comparative Examples are as follows.
- TMP-A Trimethylolpropane triacrylate, manufactured by Osaka Organic Chemical Industry Co., Ltd., number of radically polymerizable functional groups: 3
- DIPE-A Mixture of dipentaerythritol pentaacrylate and dipentaerythritol hexaacrylate, manufactured by Shin Nakamura Chemical Industry Co., Ltd., number of radically polymerizable functional groups: 5.5
- Viscoat 802 Mixture of tripentaerythritol acrylate and mono and dipentaerythritol acrylate and polypentaerythritol acrylate, manufactured by Osaka Organic Chemical Industry Co., Ltd., number of radically polymerizable functional groups: 7.8 Viscoat 1000LT: (Dendrimer (meth) acrylate, manufactured by Osaka Organic Chemical Industry Co., Ltd., radically polymerizable functional group number: 14)
- the photosensitive polyimide resin composition of the example when used, a pattern without a residue was obtained in the space portion, and the photosensitive polyimide resin composition of the example was excellent in thick film developability. Further, when the photosensitive polyimide resin composition of the example was used, a good pattern shape could be obtained with a BTM width / TOP width close to 1.0. That is, the photosensitive polyimide resin composition of the example was excellent in thick film patterning property. On the other hand, the photosensitive polyimide resin compositions of Comparative Examples 1 to 3 were inferior in at least one of the thick film developability and the pattern shape (BTM width / TOP width), and were inferior in the thick film patterning property.
Abstract
Description
このような感光性ポリイミド樹脂組成物に関する技術としては、例えば、特許文献1及び2に記載のものが挙げられる。
特許文献2には、複素環含有ポリマーの前駆体と熱塩基発生剤とラジカル重合性化合物とを含み、上記複素環含有ポリマーの前駆体がラジカル重合性基を有し、上記ラジカル重合性化合物が重合性官能基を4つ以上有する化合物及び重合性官能基を3つ有しかつ分子量が400以下である化合物からなる群から選ばれる少なくとも1つのラジカル重合性化合物を含む、感光性樹脂組成物が記載されている。
ここで、特許文献1に記載の感光性樹脂組成物は、光透過性が低く、厚膜とした場合に膜の下部まで露光時の光が届きにくいため、厚膜現像性に劣っている。特許文献2に記載の感光性樹脂組成物は、ポリイミド前駆体が脱水閉環してポリイミドが生成する際に樹脂膜が収縮しやすいため、厚膜現像時のパターン形状が悪化したり、パターンにクラックが発生したりしやすい。また、一般的に、感光性樹脂組成物を厚膜とした場合、硬化に必要な露光量が多くなるため、パターン形状がよりテーパーになりやすい。
すなわち、従来の感光性ポリイミド樹脂と多官能ラジカル重合性化合物とを含む組成物は、厚膜パターニング性の点で改善の余地があった。
下記式(1)で示される構造を有し、重量平均分子量が70,000以下である既閉環ポリイミド樹脂(A)と、4個以上100個以下のラジカル重合性官能基を有する多官能ラジカル重合性化合物(B)と、を含む、感光性ポリイミド樹脂組成物。
[上記式(1)中、Rは、環状構造、非環状構造、又は環状構造と非環状構造とを有する炭素数4以上10以下の4価の基である。Aは、脂肪族炭化水素基、脂環式炭化水素基、芳香族炭化水素基、及びオルガノシロキサン基からなる群から選択される少なくとも1種の基を有し、炭素数2以上39以下の2価の基である。Aの主鎖には-O-、-SO2-、-CO-、-CH2-、-C(CH3)2-、-C2H4O-、及び-S-からなる群から選択される少なくとも1種の基が介在していてもよい。nは繰り返し単位数を示す。上記式(1)の末端は、下記式(2)若しくは式(3)で示される基、又は水素原子のいずれかであって、末端の少なくとも一方は下記式(2)若しくは式(3)で示される基である。]
[上記式(2)及び(3)中、X及びX2はそれぞれ独立に、炭素数2以上15以下の基であり、エステル結合及び二重結合からなる群から選択される少なくとも1つの基を有してもよい。Y及びY2はそれぞれ独立に、水素原子又はメチル基である。]
[2]
上記既閉環ポリイミド樹脂(A)の重量平均分子量が5,000以上である、上記[1]に記載の感光性ポリイミド樹脂組成物。
[3]
上記既閉環ポリイミド樹脂(A)の含有量が、上記感光性ポリイミド樹脂組成物の全固形分を100質量%としたとき、30質量%以上である、上記[1]又は[2]に記載の感光性ポリイミド樹脂組成物。
[4]
上記多官能ラジカル重合性化合物(B)の含有量が、上記感光性ポリイミド樹脂組成物に含まれる上記既閉環ポリイミド樹脂(A)の量を100質量部としたとき、20質量部以上100質量部以下である、上記[1]~[3]のいずれかに記載の感光性ポリイミド樹脂組成物。
[5]
着色剤の含有量が、上記感光性ポリイミド樹脂組成物に含まれる上記既閉環ポリイミド樹脂(A)の量を100質量部としたとき、10質量部未満である、上記[1]~[4]のいずれかに記載の感光性ポリイミド樹脂組成物。
[6]
上記ラジカル重合性官能基が(メタ)アクリロイル基を含む、上記[1]~[5]のいずれかに記載の感光性ポリイミド樹脂組成物。
[7]
上記既閉環ポリイミド樹脂(A)の固形分濃度3質量%溶液とした際の波長365nmにおける光線透過率が80%以上である、上記[1]~[6]のいずれかに記載の感光性ポリイミド樹脂組成物。
[8]
上記式(1)中のAが芳香族炭化水素基として芳香環を含む、上記[1]~[7]のいずれかに記載の感光性ポリイミド樹脂組成物。
[9]
上記式(1)のAが、以下に示される構造からなる群から選択される少なくとも1種以上を含む、上記[1]~[8]のいずれかに記載の感光性ポリイミド樹脂組成物。
[式中、*は結合手を示す。]
[10]
上記既閉環ポリイミド樹脂(A)が、4,4’-ジアミノ-2,2’-ビス(トリフルオロメチル)ビフェニル、1-(4-アミノフェニル)-2,3-ジヒドロ-1,3,3-トリメチル-1H-インデン-5-アミン、4,4’-オキシビス[3-(トリフルオロメチル)ベンゼンアミン]、又は1,3-ビス[2-(4-アミノフェニル)-2-プロピル]ベンゼンから構成される単位の少なくとも1種を含む、上記[1]~[9]のいずれかに記載の感光性ポリイミド樹脂組成物。
[11]
光重合開始剤、溶剤、密着性向上剤、表面調整剤、及び増感剤からなる群から選択される少なくとも1種を更に含む、上記[1]~[10]のいずれかに記載の感光性ポリイミド樹脂組成物。
[12]
絶縁膜形成用である、上記[1]~[11]のいずれかに記載の感光性ポリイミド樹脂組成物。
[13]
上記[1]~[12]のいずれかに記載の感光性ポリイミド樹脂組成物又は前記感光性ポリイミド樹脂組成物の硬化物からなる、樹脂膜。
[14]
膜厚が20μm以上である、上記[13]に記載の樹脂膜
[15]
上記[14]に記載の樹脂膜を備える電子装置。
本実施形態に係る感光性ポリイミド樹脂組成物は、下記式(1)で示される構造を有し、重量平均分子量が70,000以下である既閉環ポリイミド樹脂(A)と、4個以上100個以下のラジカル重合性官能基を有する多官能ラジカル重合性化合物(B)と、を含む。
上記式(1)中、Rは、環状構造、非環状構造、又は環状構造と非環状構造とを有する炭素数4以上10以下の4価の基である。Aは、脂肪族炭化水素基、脂環式炭化水素基、芳香族炭化水素基、及びオルガノシロキサン基からなる群から選択される少なくとも1種の基を有し、炭素数2以上39以下の2価の基である。Aの主鎖には-O-、-SO2-、-CO-、-CH2-、-C(CH3)2-、-C2H4O-、及び-S-からなる群から選択される少なくとも1種の基が介在していてもよい。nは繰り返し単位数を示す。上記式(1)の末端は、下記式(2)若しくは式(3)で示される基、又は水素原子のいずれかであって、末端の少なくとも一方は下記式(2)若しくは式(3)で示される基である。
上記式(2)及び(3)中、X及びX2はそれぞれ独立に、炭素数2以上15以下の基であり、エステル結合及び二重結合からなる群から選択される少なくとも1つの基を有してもよい。Y及びY2はそれぞれ独立に、水素原子又はメチル基である。
まず、現像性は露光部と未露光部の現像液に対する溶解性(以下、単に「現像液溶解性」ともいう。)の差が大きいほど良好であり、現像性を向上させるには露光部と未露光部の現像液溶解性の差を広げることが重要である。また、パターン形状を良好にするためには、現像性を向上させながら、露光時の樹脂膜の収縮等の変形を抑制することが重要である。
ここで、上記式(1)で示される構造を有し、重量平均分子量が70,000以下である既閉環ポリイミド樹脂(A)は現像液溶解性が高いため、樹脂膜を厚くしても未露光部の現像液溶解性が高いと考えられる。
また、上記式(1)で示される構造を有する既閉環ポリイミド樹脂(A)は透明性に優れるため、樹脂膜を厚くしても露光時の光が膜の下部まで届きやすく、樹脂膜をより均一に架橋させることができ、更に多官能ラジカル重合性化合物(B)と組み合わせることにより、樹脂膜の架橋密度が向上し、露光部の現像液溶解性を低下させることができると考えられる。
更に、既閉環ポリイミド樹脂(A)は既にイミド化が完了している樹脂であるため、脱水閉環工程が不要である。そのため、多官能ラジカル重合性化合物(B)と組み合わせることにより、露光時の樹脂膜の硬化収縮等の変形を抑制させつつ、露光部の現像液溶解性を低下させることが可能と考えられる。
以上から、本実施形態に係る感光性ポリイミド樹脂組成物によれば、既閉環ポリイミド樹脂(A)と多官能ラジカル重合性化合物(B)とを組み合わせることによって、露光部と未露光部の現像液溶解性の差を大きくすることができるため、厚膜現像性を向上させることができ、更に露光時の樹脂膜の硬化収縮等の変形を抑制することができるため、厚膜パターニング性を向上できたと考えられる。
本実施形態に係る既閉環ポリイミド樹脂(A)は、下記式(1)で示される構造を有し、重量平均分子量が70,000以下である。
上記式(1)中、Rは、環状構造、非環状構造、又は環状構造と非環状構造とを有する炭素数4以上10以下の4価の基である。Aは、脂肪族炭化水素基、脂環式炭化水素基、芳香族炭化水素基、及びオルガノシロキサン基からなる群から選択される少なくとも1種の基を有し、炭素数2以上39以下の2価の基である。Aの主鎖には-O-、-SO2-、-CO-、-CH2-、-C(CH3)2-、-C2H4O-、及び-S-からなる群から選択される少なくとも1種の基が介在していてもよい。nは繰り返し単位数を示す。上記式(1)の末端は、下記式(2)若しくは式(3)で示される基、又は水素原子のいずれかであって、末端の少なくとも一方は下記式(2)若しくは式(3)で示される基である。
上記式(2)及び(3)中、X及びX2はそれぞれ独立に、炭素数2以上15以下の基であり、エステル結合及び二重結合からなる群から選択される少なくとも1つの基を有してもよい。Y及びY2はそれぞれ独立に、水素原子又はメチル基である。
上記式(2)又は式(3)中のX又はX2で表される基は、炭素数2以上15以下の基であり、エステル結合及び二重結合からなる群から選択される少なくとも1つの基を有してもよい。Y又はY2で示される基は、水素原子又はメチル基である。
ジアミン成分としては、例えば、ジアミン、ジイソシアネート及びジアミノジシラン類等を挙げることができ、ジアミンであることが好ましい。原料として用いるジアミン成分中のジアミン含量は、好ましくは50モル%以上、より好ましくは70モル%以上、更に好ましくは90モル%以上、更に好ましくは95モル%以上であり、そして好ましくは100モル%以下である。
ジアミン成分として、これらのジアミンを少なくとも含むことにより、得られる感光性ポリイミド樹脂組成物の、特定波長における光線透過率及び現像液溶解性をより向上させることができる。そのため、露光部の硬化性を向上でき、かつ未露光部の残膜率を低くすることができるため、得られる感光性ポリイミド樹脂組成物の現像性をより向上させることができる。
本実施形態に係る既閉環ポリイミド樹脂(A)は、4,4’-ジアミノ-2,2’-ビス(トリフルオロメチル)ビフェニル、1-(4-アミノフェニル)-2,3-ジヒドロ-1,3,3-トリメチル-1H-インデン-5-アミン、4,4’-オキシビス[3-(トリフルオロメチル)ベンゼンアミン]又は1,3-ビス[2-(4-アミノフェニル)-2-プロピル]ベンゼンから構成される単位の少なくとも1種を含むことが好ましい。
テトラカルボン酸成分としては、例えば、シクロヘキサンテトラカルボン酸、シクロヘキサンテトラカルボン酸エステル類、シクロヘキサンテトラカルボン酸二無水物、シクロブタンテトラカルボン酸、シクロブタンテトラカルボン酸エステル類、シクロブタンテトラカルボン酸二無水物、シクロペンタンテトラカルボン酸、シクロペンタンテトラカルボン酸エステル類、シクロペンタンテトラカルボン酸二無水物、ビシクロペンタンテトラカルボン酸二無水物等が挙げられる。これらの中でも、テトラカルボン酸成分は、シクロヘキサンテトラカルボン酸二無水物、シクロブタンテトラカルボン酸二無水物、及びシクロペンタンテトラカルボン酸二無水物から選択される少なくとも一種を含むことが好ましく、シクロヘキサンテトラカルボン酸二無水物を含むことがより好ましい。上記した各種テトラカルボン酸成分は位置異性体を含む。
これらの中でも、ポリイミド樹脂を製造する際に高分子量化が容易で、フレキシブルな樹脂膜が得られ易い面で有利である点から、テトラカルボン酸成分は、1,2,4,5-シクロヘキサンテトラカルボン酸、1,2,4,5-シクロヘキサンテトラカルボン酸二無水物、1,2,3,4-シクロブタンテトラカルボン酸二無水物、及び1,2,4,5-シクロヘキサンテトラカルボン酸メチルエステルから選択される少なくとも一種を含むことが好ましい。
本実施形態に係る既閉環ポリイミド樹脂(A)は、例えば、以下の工程(1)及び(2)を含む製造方法により得ることができる。
工程(1):テトラカルボン酸成分とジアミン成分を反応させ、末端にアミノ基を有するポリイミド樹脂を得る。
工程(2):上記工程(1)で得られた末端にアミノ基を有するポリイミド樹脂と、上記官能基含有化合物(イソシアネート基又はエポキシ基と、(メタ)アクリル基とを有する化合物)とを反応させる。
工程(1)では、テトラカルボン酸とジアミン成分とを反応させて、末端にアミノ基を有するポリイミド樹脂を得る。
テトラカルボン酸成分とジアミン成分とを反応させる際に使用する有機溶媒は特に限定されないが、例えば環状エーテル、環状ケトン、環状エステル、アミド及びウレアからなる群から選択される少なくとも1種を含む有機溶媒が好ましい。好適な溶媒の具体例としては、特に限定されないが、γ-ブチロラクトン、N,N-ジメチルアセトアミド、N,N-ジメチルホルムアミド、N-メチル-2-ピロリドン、ジメチルスルホキシド、ヘキサメチルホスホルアミド、シクロペンタノン、シクロヘキサノン、1,3-ジオキソラン、1,4-ジオキサン、テトラメチルウレア及びテトラヒドロフラン等の非プロトン性の極性有機溶媒からなる群から選択される少なくとも1種を挙げることができる。これらの中でも、γ-ブチロラクトン、N,N-ジメチルアセトアミド、N,N-ジメチルホルムアミド及びN-メチル-2-ピロリドンからなる群から選択される1種以上であることがより好ましい。
A/Bが1.0に近づくほど高分子量のポリイミド樹脂が得られるため、A/Bを適宜調整することで、目的の分子量のポリイミド樹脂を得ることができる。
工程(2)は、上記工程(1)で得られたポリイミド樹脂の末端を変性する工程である。具体的には、上述した通り、ポリイミドと、上記官能基含有化合物(イソシアネート基又はエポキシ基と、(メタ)アクリル基とを有する化合物)とを反応させて、末端に(メタ)アクリル基を有するポリイミド樹脂を得る。
ポリイミド樹脂のアミノ基末端と、官能基含有化合物のイソシアネート基又はエポキシ基とを反応させる際には、そのまま反応させてもよいし、必要に応じて触媒の存在下で反応させてもよい。該触媒としては、トリエチルアミン等のアミン化合物、トリフェニルホスフィン等の有機リン系化合物等が挙げられ、これらを単独で用いてもよいし、2種類以上を組み合わせて用いてもよい。反応時の副反応を抑制させるために重合禁止剤を使用してもよい。重合禁止剤としては、ハイドロキノン、ハイドロキノンモノメチルエーテル、及びメチルハイドロキノン等が挙げられ、これらを単独で用いてもよいし、2種類以上を組み合わせて用いてもよい。
ここで、本実施形態において、感光性ポリイミド樹脂組成物の全固形分とは、当該感光性ポリイミド樹脂組成物を硬化した際に固形分として残る成分であり、例えば、溶剤等の加熱により揮発する成分は除かれる。一方で、液状成分でも、加熱硬化した際に樹脂膜に取り込まれる成分は全固形分に含まれる。
本実施形態に係る多官能ラジカル重合性化合物(B)は、4個以上100個以下のラジカル重合性官能基を有する。ここで、本実施形態において、多官能ラジカル重合性化合物(B)が、ラジカル重合性官能基数が異なる2種類以上の多官能ラジカル重合性化合物を含む場合、多官能ラジカル重合性化合物(B)のラジカル重合性官能基数は、各多官能ラジカル重合性化合物のラジカル重合性官能基数の加重平均値を採用することができる。
本実施形態に係る多官能ラジカル重合性化合物(B)のラジカル重合性官能基数は、4個以上100個以下であるが、厚膜パターニング性をより向上させる観点から、好ましくは5個以上であり、そして、好ましくは50個以下、更に好ましくは30個以下、更に好ましくは20個以下、更に好ましくは15個以下である。
多官能ラジカル重合性化合物(B)を構成する多官能ラジカル重合性化合物としては、例えば、トリシクロデカンジメタノールジ(メタ)アクリレート、ポリプロピレングリコールジ(メタ)アクリレート、トリメチロールプロパントリ(メタ)アクリレート、エトキシ化トリメチロールプロパントリ(メタ)アクリレート、プロポキシ化トリメチロールプロパントリ(メタ)アクリレート、ペンタエリスリトールテトラ(メタ)アクリレート、ペンタエリスリトールトリ(メタ)アクリレート、トリス-(2-(メタ)アクリロキシエチル)イソシアヌレート、ジトリメチロールプロパンテトラ(メタ)アクリレート、ジトリメチロールプロパンペンタ(メタ)アクリレート、ジトリメチロールプロパンヘキサ(メタ)アクリレート、ジペンタエリスリトールペンタ(メタ)アクリレート、ジペンタエリスリトールヘキサ(メタ)アクリレート、トリペンタエリスリトール(メタ)アクリレート、モノ及びジペンタエリスリトール(メタ)アクリレート、ポリペンタエリスリトール(メタ)アクリレート、ポリグリセリン系(メタ)アクリレート、デンドリマー(メタ)アクリレート等が挙げられる。これらの多官能ラジカル重合性化合物は単独で用いてもよいし、2種以上を組み合わせて用いてもよい。また、これらの多官能ラジカル重合性化合物を単独で用いてラジカル重合性官能基数を上記範囲内に調整してもよいし、2種以上を組み合わせてラジカル重合性官能基数を上記範囲内に調整してもよい。
ここで、ポリグリセリン系(メタ)アクリレートとは、ポリグリセリン骨格及び(メタ)アクリロイル基を有する化合物のことをいう。ポリグリセリン系(メタ)アクリレートとしては、例えば、阪本薬品工業株式会社製のSA-TE6、SA-TE60、SA-ZE12等が挙げられる。
エトキシ化トリメチロールプロパントリ(メタ)アクリレートとしては、例えば、アルケマ株式会社製のエトキシ化(3)トリメチロールプロパントリアクリレート、エトキシ化(6)トリメチロールプロパントリアクリレート、エトキシ化(15)トリメチロールプロパントリアクリレート等を使用することができる。
また、デンドリマー(メタ)アクリレートとは、デンドリマー構造(ハイパーブランチ構造を含む)を有する多官能(メタ)アクリレートをいう。デンドリマー(メタ)アクリレートとしては、例えば、大阪有機化学工業株式会社製のビスコート1000、ビスコート1020、STAR-501(SIRIUS-501、SUBARU-501)等が挙げられる。ビスコート1000及びビスコート1020は、末端にアクリレート基を有するデンドリマー型ポリエステルアクリレートを主成分とする。また、ビスコート1000の分子量は、1000~2000程度であり、ビスコート1020の分子量は1000~3000程度である。STAR-501は、ジペンタエリスリトールに由来するコアを含み、末端にアクリレート基を有する、ジペンタエリスリトールヘキサアクリレート連結型の多分岐ポリアクリレートを主成分とする。STAR501の分子量は、16000~24000程度である。
本実施形態に係る感光性ポリイミド樹脂組成物は、既閉環ポリイミド樹脂(A)及び多官能ラジカル重合性化合物(B)以外のその他の成分として、例えば、光重合開始剤、溶剤、密着性向上剤、表面調整剤、及び増感剤からなる群から選択される少なくとも1種を更に含むことが好ましく、光重合開始剤及び溶剤からなる群から選択される少なくとも1種を更に含むことがより好ましい。
光重合開始剤は特に限定されず公知のものを使用することができる。例えば、2,2-ジメトキシ-1,2-ジフェニルエタン-1-オン、1-ヒドロキシシクロヘキシルフェニルケトン、2-ヒドロキシ-2-メチル-1-フェニル-プロパン-1-オン、1-[4-(2-ヒドロキシエトキシ)-フェニル]-2-ヒドロキシ-2-メチル-1-プロパン-1-オン、2-ヒドロキシ-1-{4-[4-(2-ヒドロキシ-2-メチル-プロピオニル)-ベンジル]フェニル}-2-メチル-プロパン-1-オン、2-メチル-1-(4-メチルチオフェニル)-2-モルホリノプロパン-1-オン、2-ベンジル-2-ジメチルアミノ-1-(4-モルホリノフェニル)-ブタノン-1,2-(ジメチルアミノ)-2-[(4-メチルフェニル)メチル]-1-[4-(4-モルホリニル)フェニル]-1-ブタノン、2,4,6-トリメチルベンゾイル-ジフェニルホスフィンオキサイド及びビス(2,4,6-トリメチルベンゾイル)フェニルホスフィンオキサイド等を挙げることができる。これらの光重合開始剤は単独で用いてもよいし、2種以上を組み合わせて用いてもよい。
本実施形態に係る感光性ポリイミド樹脂組成物中の光重合開始剤の含有量は、感光性ポリイミド樹脂組成物に含まれる既閉環ポリイミド樹脂(A)の量を100質量部としたとき、例えば0.1質量部以上10質量部以下である。
溶剤としては、溶解性の観点から非プロトン性極性溶媒が望ましい。溶剤としては、例えば、N-メチル-2-ピロリドン、N-アセチル-2-ピロリドン、N-ベンジル-2-ピロリドン、N,N-ジメチルホルムアミド、N,N-ジメチルアセトアミド、ジメチルスルホキシド、ヘキサメチルホスホルトリアミド、N-アセチル-ε-カプロラクタム、ジメチルイミダゾリジノン、ジエチレングリコールジメチルエーテル、トリエチレングリコールジメチルエーテル及びγ-ブチロラクトン等を挙げることができる。これらの溶剤は単独で用いてもよいし、2種以上を組み合わせて用いてもよい。塗布性をより改善するために、トルエン、キシレン、ジエチルケトン、メトキシベンゼン、シクロペンタノン等の溶媒をポリマーの溶解性に悪影響を及ぼさない範囲で混合してもよい。
適切な溶剤を用いることで、本実施形態に係る感光性ポリイミド樹脂組成物を溶液(ワニス)状態で使用することができ、成膜性を向上させることができる。
密着性向上剤は特に限定されず、公知のものを使用でき、アミノ基含有シランカップリング剤、エポキシ基含有シランカップリング剤、メルカプト基含有シランカップリング剤、(メタ)アクリル基含有シランカップリング剤等のシランカップリング剤、チタネートカップリング剤、アルミネートカップリング剤等の公知のカップリング剤等を用いることができる。カップリング剤としては、例えば、KP-390、KA-1003、KBM-1003、KBE-1003、KBM-303、KBM-403、KBE-402、KBE-403、KBM-1403、KBM-502、KBM-503、KBE-502、KBE-503、KBM-5103、KBM-602、KBM-603、KBE-603、KBM-903、KBE-903、KBE-9103、KBM-9103、KBM-573、KBM-575、KBM-6123、KBE-585、KBM-703、KBM-802、KBM-803、KBE-846、KBE-9007(いずれも商品名;信越化学工業株式会社製)等を挙げることができる。これらは1種を単独で用いてもよく2種以上を併用してもよい。
本実施形態に係る感光性ポリイミド樹脂組成物中の密着性向上剤の含有量は、感光性ポリイミド樹脂組成物に含まれる既閉環ポリイミド樹脂(A)の量を100質量部としたとき、例えば0.0005質量部以上20質量部以下である。
表面調整剤は特に限定されず、公知のものを使用でき、例えば、シリコン系表面調整剤、アクリル系表面調整剤、フッ素系表面調整剤、ノニオン性表面調整剤、カチオン性表面調整剤、アニオン性表面調整剤等の各種表面調整剤等を用いることができる。これらは1種を単独で用いてもよく2種以上を併用してもよい。
本実施形態に係る感光性ポリイミド樹脂組成物中の表面調整剤の含有量は、感光性ポリイミド樹脂組成物に含まれる既閉環ポリイミド樹脂(A)の量を100質量部としたとき、例えば0.001質量部以上20質量部以下である。
増感剤は特に限定されず、公知のものを使用できる。例えば、アミノ基含有増感剤を挙げることができ、アミノ基及びフェニル基を同一分子内に有する化合物を好ましくは例示できる。より具体的には、4,4’-ジメチルアミノベンゾフェノン、4,4’-ジエチルアミノベンゾフェノン、2-アミノベンゾフェノン、4-アミノベンゾフェノン、4,4’-ジアミノベンゾフェノン、3,3’-ジアミノベンゾフェノン、3,4-ジアミノベンゾフェノン等のベンゾフェノン系化合物;2-(p-ジメチルアミノフェニル)ベンゾオキサゾール、2-(p-ジエチルアミノフェニル)ベンゾオキサゾール、2-(p-ジメチルアミノフェニル)ベンゾ[4,5]ベンゾオキサゾール、2-(p-ジメチルアミノフェニル)ベンゾ[6,7]ベンゾオキサゾール、2,5-ビス(p-ジエチルアミノフェニル)-1,3,4-オキサジアゾール、2-(p-ジメチルアミノフェニル)ベンゾチアゾール、2-(p-ジエチルアミノフェニル)ベンゾチアゾール、2-(p-ジメチルアミノフェニル)ベンズイミダゾール、2-(p-ジエチルアミノフェニル)ベンズイミダゾール、2,5-ビス(p-ジエチルアミノフェニル)-1,3,4-チアジアゾール、(p-ジメチルアミノフェニル)ピリジン、(p-ジエチルアミノフェニル)ピリジン、(p-ジメチルアミノフェニル)キノリン、(p-ジエチルアミノフェニル)キノリン、(p-ジメチルアミノフェニル)ピリミジン、(p-ジエチルアミノフェニル)ピリミジン等のp-ジアルキルアミノフェニル基含有化合物等を挙げることができる。これらの増感剤は単独で用いてもよいし、2種以上を組み合わせて用いてもよい。
本実施形態に係る感光性ポリイミド樹脂組成物中の増感剤の含有量は、感光性ポリイミド樹脂組成物に含まれる既閉環ポリイミド樹脂(A)の量を100質量部としたとき、例えば0.001質量部以上10質量部以下である。
本実施形態に係る感光性ポリイミド樹脂組成物中の着色剤の含有量は、感光性ポリイミド樹脂組成物に含まれる上記既閉環ポリイミド樹脂(A)の量を100質量部としたとき、得られる樹脂膜の透明性をより向上させ、厚膜現像性をより向上させる観点から、好ましくは10質量部未満、より好ましくは5質量部未満、更に好ましくは1質量部未満、更に好ましくは0.5質量部未満、更に好ましくは0.1質量部未満であり、本実施形態に係る感光性ポリイミド樹脂組成物は着色剤を含まないことが更に好ましい。
ここで、着色剤とは、感光性材料分野で一般的に用いられる、有機顔料、無機顔料又は染料をいう。
本実施形態に係る感光性ポリイミド樹脂組成物は、例えば、電子装置を製造する際に用いるレジストや電子装置を構成する永久膜(硬化膜)等の樹脂膜を形成するために用いることができる。すなわち、本実施形態に係る樹脂膜は、感光性ポリイミド樹脂組成物又は感光性ポリイミド樹脂組成物の硬化物からなる。
上記レジストは、例えば、本実施形態に係る感光性ポリイミド樹脂組成物を基材上に塗布し、必要に応じて溶剤を除去することにより得られた樹脂膜で構成される。
上記永久膜は、上記樹脂膜に対して露光及び現像を行い、所望の形状にパターニングした後、熱処理等によって硬化させることにより得られた硬化膜で構成される。
永久膜は、例えば、表面保護膜、層間絶縁膜等の絶縁膜として好適に用いることができる。
紫外線照射量は、積算照射量として好ましくは100~8,000mJ/cm2、より好ましくは200~6,000mJ/cm2である。
これらの現像液は単独で用いてもよいし、2種以上を組み合わせて用いてもよい。
上述する処理によって得られたレリーフパターンを、80~250℃の範囲から選ばれる温度で加熱処理し、溶剤を乾燥し、本実施形態に係る感光性ポリイミド樹脂組成物を硬化してなる硬化膜(パターン)を得ることができる。本実施形態によれば、優れた現像性、すなわち露光部は十分に硬化し、未露光部は十分に除去される感光性ポリイミド樹脂組成物を用いるため、得られるレリーフパターンを高解像度で得ることができる。
したがって、本実施形態に係る樹脂膜は、例えば、高い電圧の印加が想定される絶縁性膜用途において好適に用いることができる。既閉環ポリイミド樹脂(A)及び多官能ラジカル重合性化合物(B)を含む、本実施形態に係る感光性ポリイミド樹脂組成物から得られる樹脂膜は、クラック等の発生を効果的に抑制することができ、物性に優れる。
本実施形態に係る電子装置は、本実施形態に係る樹脂膜を備える。
本実施形態に係る電子装置は、本実施形態に係る感光性ポリイミド樹脂組成物により形成される樹脂膜を備えるものであれば特に限定されないが、例えば、本実施形態に係る樹脂膜を平坦化膜やマイクロレンズとして有する表示装置;本実施形態に係る樹脂膜を層間絶縁膜として用いた多層配線構造を備える半導体装置;本実施形態に係る樹脂膜を半導体素子や配線基板の表面保護膜として用いた半導体装置;本実施形態に係る樹脂膜を配線基板を構成するビルドアップ層もしくはコア層として用いた半導体装置等が挙げられる。これらの中でも、本実施形態に係る電子装置としては半導体装置が好ましい。
本実施形態に係る電子装置は、本実施形態に係る樹脂膜を使用する以外は、公知の情報に基づいて製造することができる。
本実施形態に係る電子装置は、良好なパターニング形状を有する樹脂膜を備えるため、絶縁破壊等が起こり難く、信頼性に優れている。
本実施例及び比較例で採用した評価方法は以下の通りである。
GPC分析により、Mwを求めた。分析に用いた装置及び分析条件は下記の通りである。
装置:Ecosec Elite HLC8420GPC(東ソー株式会社製)
カラム:TSKGelsuperAWM-H(カラムサイズ:6.0mm×15cm、充填ゲル粒子径:9μm)×2(東ソー株式会社製)
溶離液:(20mM臭化リチウム及び100mMリン酸)添加ジメチルホルムアミド
標準ポリスチレン:PStQuick Kit-H(東ソー株式会社製)
流速:0.6mL/min
カラム温度:40℃
検出器:RI(屈折率検出器)
以下に記載する実施例及び比較例にて、感光性ポリイミド樹脂組成物ワニスを調製した。スピンコーターでシリコンウェハー上に、各実施例及び比較例でそれぞれ得られたワニスを溶媒除去後の膜厚が30μmとなるよう塗布した後、100℃で5分間加熱し、溶媒を除去した。マスクアライナーにて所定量露光し、15分間室温で静置した後、現像液のγ-ブチロラクトンを30秒間吹きかけて未露光部を除去し、リンス液としてメタノールを用いて洗浄し、空気流通下にて、溶媒を除去した。
マスクアライナー:MA-10B(ミカサ株式会社製)
光源:高圧水銀ランプ
照度:12W/cm2(波長365nm)
積算照射量:1,000~5000mJ/cm2
現像液:γ-ブチロラクトン
リンス液:メタノール
シリコンウェハー:4インチシリコンウェハー(株式会社アドバンテック製)
上記(2)で作製したパターンを割断し、断面をSEM(TM3030plus(株式会社日立製作所製))で観察し、パターンの下部幅(BTM幅)/上部幅(TOP幅)を測定した。更に、スペース部(未露光部)における残渣の有無を確認した。スペース部に残渣がない場合を「〇」とし、スペース部に残渣がある場合を「×」とした。
合成例で得られた既閉環ポリイミド樹脂(A)をγ-ブチロラクトンに溶解し、固形分濃度3質量%のポリイミド樹脂溶液を得た。次いで、このポリイミド樹脂溶液の波長365nmにおける光線透過率を株式会社日立製作所製の分光光度計「製品名:U3900H」及び光路長10mmのセルを用いて測定した。
合成例1
窒素導入管、撹拌装置、温度計、冷却器を備えた1,000mLの5つ口フラスコに、窒素導入下、4,4’-オキシビス[3-(トリフルオロメチル)ベンゼンアミン]を184.927g(0.55mol)、トリエチルアミンを2.644g(0.026mol)、γ-ブチロラクトン(以下、GBL)を369.471g投入し、撹拌しながら70℃まで加熱した。ここに、1,2,4,5-シクロヘキサンテトラカルボン酸二無水物117.129g(0.523mol)を加え、190℃で5時間反応させた。GBL290.788gで希釈し、固形分濃度30質量%のポリイミドワニス943gを得た。GPCでの測定の結果、得られたポリイミドのポリスチレン換算重量平均分子量は34,046であった。
次いで、撹拌装置、温度計、冷却器を備えた500mLの4つ口フラスコに、得られたポリイミドワニス151.88g、2-イソシアナトエチルアクリレート(昭和電工株式会社製、カレンズAOI)3.50gを入れ、50℃で5時間反応させた。その後、反応液を水中に滴下してポリイミドを析出させ、70℃で一晩乾燥させ、既閉環ポリイミド樹脂(A1)を得た。得られた既閉環ポリイミド樹脂(A1)のポリスチレン換算重量平均分子量は34,664であった。また、得られた既閉環ポリイミド樹脂(A1)の波長365nmにおける光線透過率は92.0%であった。
実施例1
合成例1で得られた既閉環ポリイミド樹脂(A1)10.0gをGBL12.2gに溶解させ、ジペンタエリスリトールペンタアクリレートとジペンタエリスリトールヘキサアクリレートとの混合物(DIPE-A、新中村化学工業株式会社、ラジカル重合性官能基数:5.5)5.0g、1-ヒドロキシシクロヘキシルフェニルケトン(BASF社製、Omnirad184)0.15g、ビス(2,4,6-トリメチルベンゾイル)フェニルホスフィンオキサイド(BASF社製、Omnirad819)0.35g、表面調整剤としてLE-304(共栄社化学株式会社製、シリコン系表面調整剤)0.01g、密着性向上剤としてKP-390(信越化学工業株式会社製、塗料添加剤)0.005g、を加え、溶解するまで撹拌し、感光性ポリイミド樹脂組成物ワニスを得た。この感光性ポリイミド樹脂組成物ワニスを、乾燥後の膜厚が30μmとなるようシリコンウェハー上に塗布し、100℃で5分間乾燥させた。次いで、ミカサ株式会社製マスクアライナー(MA-10B)で波長365nm未満の波長をカットした高圧水銀灯の光を積算照射量4000mJ/cm2(365nmの照度から算出)で照射して露光し、ライン/スペース=30μm/30μmのパターンを作製した。室温で15分間静置した後、GBLを30秒間かけて未露光部のスペース部を洗い流し、メタノールでリンスし、空気流通下で溶媒を除去した。パターンの断面形状をSEMで観察し、スペース部の残渣有無(現像性)と、ライン部のパターン形状(BTM幅/TOP幅)を確認した。スペース部の残渣は無く、BTM幅/TOP幅=1.02と、現像性、パターン形状ともに良好であった。結果を表1に示す。
下記表1に示す配合及び積算照射量としたこと以外は、上記実施例1と同様に感光性樹脂組成物を調製し、その特性を評価した。結果を表1に合わせて示す。
ただし、各実施例1~4及び比較例1~2において、積算照射量(露光量)は、複数の異なる積算照射量で露光したとき、現像性が最も優れていた積算照射量を採用した。比較例3は、比較例2において、積算照射量を変化させた例である。また表1に記載した配合量の単位は「g」である。
TMP-A:トリメチロールプロパントリアクリレート、大阪有機化学工業株式会社製、ラジカル重合性官能基数:3
DIPE-A:ジペンタエリスリトールペンタアクリレートとジペンタエリスリトールヘキサアクリレートとの混合物、新中村化学工業株式会社製、ラジカル重合性官能基数:5.5
ビスコート802:トリペンタエリスリトールアクリレートとモノ及びジペンタエリスリトールアクリレートとポリペンタエリスリトールアクリレートとの混合物、大阪有機化学工業株式会社製、ラジカル重合性官能基数:7.8
ビスコート1000LT:(デンドリマー(メタ)アクリレート、大阪有機化学工業株式会社製、ラジカル重合性官能基数:14)
SIRIUS-501:(デンドリマー(メタ)アクリレート、大阪有機化学工業株式会社製、ラジカル重合性官能基数:168)
TMP-AとSIRIUS-501の等モル混合物:ラジカル重合性官能基数:85.5
一方、比較例1~3の感光性ポリイミド樹脂組成物は、厚膜現像性及びパターン形状(BTM幅/TOP幅)のうち少なくとも一方が劣っており、厚膜パターニング性に劣っていた。
Claims (15)
- 下記式(1)で示される構造を有し、重量平均分子量が70,000以下である既閉環ポリイミド樹脂(A)と、4個以上100個以下のラジカル重合性官能基を有する多官能ラジカル重合性化合物(B)と、を含む、感光性ポリイミド樹脂組成物。
[前記式(1)中、Rは、環状構造、非環状構造、又は環状構造と非環状構造とを有する炭素数4以上10以下の4価の基である。Aは、脂肪族炭化水素基、脂環式炭化水素基、芳香族炭化水素基、及びオルガノシロキサン基からなる群から選択される少なくとも1種の基を有し、炭素数2以上39以下の2価の基である。Aの主鎖には-O-、-SO2-、-CO-、-CH2-、-C(CH3)2-、-C2H4O-、及び-S-からなる群から選択される少なくとも1種の基が介在していてもよい。nは繰り返し単位数を示す。前記式(1)の末端は、下記式(2)若しくは式(3)で示される基、又は水素原子のいずれかであって、末端の少なくとも一方は下記式(2)若しくは式(3)で示される基である。]
[前記式(2)及び(3)中、X及びX2はそれぞれ独立に、炭素数2以上15以下の基であり、エステル結合及び二重結合からなる群から選択される少なくとも1つの基を有してもよい。Y及びY2はそれぞれ独立に、水素原子又はメチル基である。] - 前記既閉環ポリイミド樹脂(A)の重量平均分子量が5,000以上である、請求項1に記載の感光性ポリイミド樹脂組成物。
- 前記既閉環ポリイミド樹脂(A)の含有量が、前記感光性ポリイミド樹脂組成物の全固形分を100質量%としたとき、30質量%以上である、請求項1又は2に記載の感光性ポリイミド樹脂組成物。
- 前記多官能ラジカル重合性化合物(B)の含有量が、前記感光性ポリイミド樹脂組成物に含まれる前記既閉環ポリイミド樹脂(A)の量を100質量部としたとき、20質量部以上100質量部以下である、請求項1~3のいずれかに記載の感光性ポリイミド樹脂組成物。
- 着色剤の含有量が、前記感光性ポリイミド樹脂組成物に含まれる前記既閉環ポリイミド樹脂(A)の量を100質量部としたとき、10質量部未満である、請求項1~4のいずれかに記載の感光性ポリイミド樹脂組成物。
- 前記ラジカル重合性官能基が(メタ)アクリロイル基を含む、請求項1~5のいずれかに記載の感光性ポリイミド樹脂組成物。
- 前記既閉環ポリイミド樹脂(A)の固形分濃度3質量%溶液とした際の波長365nmにおける光線透過率が80%以上である、請求項1~6のいずれかに記載の感光性ポリイミド樹脂組成物。
- 前記式(1)中のAが芳香族炭化水素基として芳香環を含む、請求項1~7のいずれかに記載の感光性ポリイミド樹脂組成物。
- 前記既閉環ポリイミド樹脂(A)が、4,4’-ジアミノ-2,2’-ビス(トリフルオロメチル)ビフェニル、1-(4-アミノフェニル)-2,3-ジヒドロ-1,3,3-トリメチル-1H-インデン-5-アミン、4,4’-オキシビス[3-(トリフルオロメチル)ベンゼンアミン]、又は1,3-ビス[2-(4-アミノフェニル)-2-プロピル]ベンゼンから構成される単位の少なくとも1種を含む、請求項1~9のいずれかに記載の感光性ポリイミド樹脂組成物。
- 光重合開始剤、溶剤、密着性向上剤、表面調整剤、及び増感剤からなる群から選択される少なくとも1種を更に含む、請求項1~10のいずれかに記載の感光性ポリイミド樹脂組成物。
- 絶縁膜形成用である、請求項1~11のいずれかに記載の感光性ポリイミド樹脂組成物。
- 請求項1~12のいずれかに記載の感光性ポリイミド樹脂組成物又は前記感光性ポリイミド樹脂組成物の硬化物からなる、樹脂膜。
- 膜厚が20μm以上である、請求項13に記載の樹脂膜。
- 請求項14に記載の樹脂膜を備える電子装置。
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP21900374.6A EP4257622A1 (en) | 2020-12-02 | 2021-11-10 | Photosensitive polyimide resin composition, resin film, and electronic device |
| US18/038,574 US20240085789A1 (en) | 2020-12-02 | 2021-11-10 | Photosensitive polyimide resin composition, resin film, and electronic device |
| JP2022566807A JPWO2022118619A1 (ja) | 2020-12-02 | 2021-11-10 | |
| CN202180080165.2A CN116547327A (zh) | 2020-12-02 | 2021-11-10 | 感光性聚酰亚胺树脂组合物、树脂膜和电子装置 |
| KR1020237018092A KR20230113556A (ko) | 2020-12-02 | 2021-11-10 | 감광성 폴리이미드 수지 조성물, 수지막 및 전자장치 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2020-200269 | 2020-12-02 | ||
| JP2020200269 | 2020-12-02 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2022118619A1 true WO2022118619A1 (ja) | 2022-06-09 |
Family
ID=81853106
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2021/041279 WO2022118619A1 (ja) | 2020-12-02 | 2021-11-10 | 感光性ポリイミド樹脂組成物、樹脂膜及び電子装置 |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US20240085789A1 (ja) |
| EP (1) | EP4257622A1 (ja) |
| JP (1) | JPWO2022118619A1 (ja) |
| KR (1) | KR20230113556A (ja) |
| CN (1) | CN116547327A (ja) |
| TW (1) | TW202231722A (ja) |
| WO (1) | WO2022118619A1 (ja) |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2006064867A1 (ja) * | 2004-12-16 | 2006-06-22 | Nippon Kayaku Kabushiki Kaisha | 不飽和基含有ポリイミド樹脂、それを含有する感光性樹脂組成物及びその硬化物 |
| WO2017159876A1 (ja) * | 2016-03-18 | 2017-09-21 | 東レ株式会社 | ネガ型感光性樹脂組成物、硬化膜、硬化膜を具備する表示装置、及びその製造方法 |
| JP2018070829A (ja) * | 2016-11-02 | 2018-05-10 | 東レ株式会社 | 樹脂組成物 |
| WO2018180592A1 (ja) | 2017-03-28 | 2018-10-04 | 東レ株式会社 | 感光性樹脂組成物、硬化膜、硬化膜を具備する素子、硬化膜を具備する有機el表示装置、硬化膜の製造方法、および有機el表示装置の製造方法 |
| WO2019189110A1 (ja) | 2018-03-29 | 2019-10-03 | 富士フイルム株式会社 | 感光性樹脂組成物、硬化膜、積層体、硬化膜の製造方法、および半導体デバイス |
| WO2021187355A1 (ja) * | 2020-03-18 | 2021-09-23 | 東レ株式会社 | 感光性樹脂組成物、感光性シート、硬化膜、硬化膜の製造方法、電子部品、アンテナ素子、半導体パッケージおよび表示装置 |
-
2021
- 2021-11-10 JP JP2022566807A patent/JPWO2022118619A1/ja active Pending
- 2021-11-10 EP EP21900374.6A patent/EP4257622A1/en active Pending
- 2021-11-10 CN CN202180080165.2A patent/CN116547327A/zh active Pending
- 2021-11-10 KR KR1020237018092A patent/KR20230113556A/ko unknown
- 2021-11-10 US US18/038,574 patent/US20240085789A1/en active Pending
- 2021-11-10 WO PCT/JP2021/041279 patent/WO2022118619A1/ja active Application Filing
- 2021-11-15 TW TW110142325A patent/TW202231722A/zh unknown
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2006064867A1 (ja) * | 2004-12-16 | 2006-06-22 | Nippon Kayaku Kabushiki Kaisha | 不飽和基含有ポリイミド樹脂、それを含有する感光性樹脂組成物及びその硬化物 |
| WO2017159876A1 (ja) * | 2016-03-18 | 2017-09-21 | 東レ株式会社 | ネガ型感光性樹脂組成物、硬化膜、硬化膜を具備する表示装置、及びその製造方法 |
| JP2018070829A (ja) * | 2016-11-02 | 2018-05-10 | 東レ株式会社 | 樹脂組成物 |
| WO2018180592A1 (ja) | 2017-03-28 | 2018-10-04 | 東レ株式会社 | 感光性樹脂組成物、硬化膜、硬化膜を具備する素子、硬化膜を具備する有機el表示装置、硬化膜の製造方法、および有機el表示装置の製造方法 |
| WO2019189110A1 (ja) | 2018-03-29 | 2019-10-03 | 富士フイルム株式会社 | 感光性樹脂組成物、硬化膜、積層体、硬化膜の製造方法、および半導体デバイス |
| WO2021187355A1 (ja) * | 2020-03-18 | 2021-09-23 | 東レ株式会社 | 感光性樹脂組成物、感光性シート、硬化膜、硬化膜の製造方法、電子部品、アンテナ素子、半導体パッケージおよび表示装置 |
Also Published As
| Publication number | Publication date |
|---|---|
| CN116547327A (zh) | 2023-08-04 |
| KR20230113556A (ko) | 2023-07-31 |
| EP4257622A1 (en) | 2023-10-11 |
| US20240085789A1 (en) | 2024-03-14 |
| JPWO2022118619A1 (ja) | 2022-06-09 |
| TW202231722A (zh) | 2022-08-16 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5887867B2 (ja) | 感光性樹脂組成物、これを用いた感光性樹脂フィルム、並びにこれらを用いた電子部品 | |
| JP2010256532A (ja) | 感光性樹脂組成物、感光性エレメント及びこれを用いたレジストパターンの形成方法 | |
| JP5239818B2 (ja) | 感光性樹脂組成物、sawフィルタ及びその製造方法 | |
| JPWO2019146611A1 (ja) | 感光性樹脂組成物、樹脂、硬化膜、積層体、硬化膜の製造方法、半導体デバイス | |
| JP6238593B2 (ja) | 膜形成用組成物 | |
| TWI819121B (zh) | 硬化膜的製造方法、硬化膜、積層體的製造方法及半導體元件的製造方法 | |
| WO2022118619A1 (ja) | 感光性ポリイミド樹脂組成物、樹脂膜及び電子装置 | |
| WO2022118620A1 (ja) | 感光性ポリイミド樹脂組成物、樹脂膜及び電子装置 | |
| WO2022118618A1 (ja) | パターン付き基板の製造方法 | |
| CN113892058B (zh) | 感光性树脂组合物和其固化膜 | |
| WO2021140845A1 (ja) | ポリイミド樹脂、感光性樹脂組成物、樹脂膜及び電子装置 | |
| JP6738353B2 (ja) | 感光性樹脂組成物、ドライフィルム、硬化物およびプリント配線板 | |
| JP2010001352A (ja) | 新規なポリイミド前駆体組成物及びその利用 | |
| WO2020246349A1 (ja) | 感光性樹脂組成物及びその硬化膜 | |
| JP7319381B2 (ja) | ネガ型硬化性組成物、硬化膜、積層体、硬化膜の製造方法、及び、半導体デバイス | |
| JP7339103B2 (ja) | 硬化性樹脂組成物、ドライフィルム、硬化物、および、電子部品 | |
| WO2022107747A1 (ja) | 硬化性樹脂組成物およびドライフィルム | |
| CN110537146A (zh) | 感光性树脂组合物 | |
| JP2021089442A (ja) | 感光性樹脂組成物、感光性樹脂フィルム、硬化物の製造方法、積層体、及び電子部品 | |
| WO2020195804A1 (ja) | 感光性樹脂組成物、ドライフィルム、硬化物、及び、電子部品 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 21900374 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2022566807 Country of ref document: JP Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 18038574 Country of ref document: US |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 202180080165.2 Country of ref document: CN |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2021900374 Country of ref document: EP Effective date: 20230703 |