WO2020184570A1 - 潤滑油用添加剤、潤滑油用添加剤組成物およびこれらを含有する潤滑油組成物 - Google Patents
潤滑油用添加剤、潤滑油用添加剤組成物およびこれらを含有する潤滑油組成物 Download PDFInfo
- Publication number
- WO2020184570A1 WO2020184570A1 PCT/JP2020/010345 JP2020010345W WO2020184570A1 WO 2020184570 A1 WO2020184570 A1 WO 2020184570A1 JP 2020010345 W JP2020010345 W JP 2020010345W WO 2020184570 A1 WO2020184570 A1 WO 2020184570A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- lubricating oil
- group
- additive
- carbon atoms
- formula
- Prior art date
Links
- 239000010687 lubricating oil Substances 0.000 title claims abstract description 118
- 239000000654 additive Substances 0.000 title claims abstract description 85
- 230000000996 additive effect Effects 0.000 title claims abstract description 75
- 239000000203 mixture Substances 0.000 title claims description 93
- 125000004432 carbon atom Chemical group C* 0.000 claims abstract description 37
- 125000005702 oxyalkylene group Chemical group 0.000 claims abstract description 19
- 150000007942 carboxylates Chemical class 0.000 claims abstract description 16
- QGZKDVFQNNGYKY-UHFFFAOYSA-O Ammonium Chemical compound [NH4+] QGZKDVFQNNGYKY-UHFFFAOYSA-O 0.000 claims abstract description 9
- 125000002915 carbonyl group Chemical group [*:2]C([*:1])=O 0.000 claims abstract description 4
- 239000002199 base oil Substances 0.000 claims description 39
- WMYJOZQKDZZHAC-UHFFFAOYSA-H trizinc;dioxido-sulfanylidene-sulfido-$l^{5}-phosphane Chemical compound [Zn+2].[Zn+2].[Zn+2].[O-]P([O-])([S-])=S.[O-]P([O-])([S-])=S WMYJOZQKDZZHAC-UHFFFAOYSA-H 0.000 claims description 17
- 150000002430 hydrocarbons Chemical group 0.000 claims description 4
- 239000004215 Carbon black (E152) Substances 0.000 claims description 2
- 229930195733 hydrocarbon Natural products 0.000 claims description 2
- 125000001183 hydrocarbyl group Chemical group 0.000 abstract description 24
- 238000005260 corrosion Methods 0.000 description 36
- 230000007797 corrosion Effects 0.000 description 36
- 229910052751 metal Inorganic materials 0.000 description 36
- 239000002184 metal Substances 0.000 description 36
- 150000001875 compounds Chemical class 0.000 description 31
- -1 phosphonate ester Chemical class 0.000 description 28
- 239000003921 oil Substances 0.000 description 18
- 235000019198 oils Nutrition 0.000 description 17
- 239000003795 chemical substances by application Substances 0.000 description 10
- 125000000217 alkyl group Chemical group 0.000 description 9
- 238000011156 evaluation Methods 0.000 description 9
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 8
- 150000001732 carboxylic acid derivatives Chemical class 0.000 description 8
- 238000009472 formulation Methods 0.000 description 8
- 238000002360 preparation method Methods 0.000 description 8
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 7
- 238000005299 abrasion Methods 0.000 description 7
- 230000000052 comparative effect Effects 0.000 description 7
- 229930195734 saturated hydrocarbon Natural products 0.000 description 7
- 229930195735 unsaturated hydrocarbon Natural products 0.000 description 7
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 description 6
- HCHKCACWOHOZIP-UHFFFAOYSA-N Zinc Chemical compound [Zn] HCHKCACWOHOZIP-UHFFFAOYSA-N 0.000 description 6
- 239000002253 acid Substances 0.000 description 6
- 230000015572 biosynthetic process Effects 0.000 description 6
- 238000002156 mixing Methods 0.000 description 6
- 229910052698 phosphorus Inorganic materials 0.000 description 6
- 229910052717 sulfur Inorganic materials 0.000 description 6
- 239000011593 sulfur Substances 0.000 description 6
- 239000011701 zinc Substances 0.000 description 6
- 229910052725 zinc Inorganic materials 0.000 description 6
- OAICVXFJPJFONN-UHFFFAOYSA-N Phosphorus Chemical compound [P] OAICVXFJPJFONN-UHFFFAOYSA-N 0.000 description 5
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 5
- 239000011521 glass Substances 0.000 description 5
- 238000004519 manufacturing process Methods 0.000 description 5
- 239000011574 phosphorus Substances 0.000 description 5
- 239000004721 Polyphenylene oxide Substances 0.000 description 4
- 125000002947 alkylene group Chemical group 0.000 description 4
- 229910052799 carbon Inorganic materials 0.000 description 4
- 230000000694 effects Effects 0.000 description 4
- 239000007789 gas Substances 0.000 description 4
- 238000000034 method Methods 0.000 description 4
- 239000010705 motor oil Substances 0.000 description 4
- 229910052757 nitrogen Inorganic materials 0.000 description 4
- 125000001147 pentyl group Chemical group C(CCCC)* 0.000 description 4
- 229920013639 polyalphaolefin Polymers 0.000 description 4
- 229920000570 polyether Polymers 0.000 description 4
- 150000003839 salts Chemical class 0.000 description 4
- 238000003786 synthesis reaction Methods 0.000 description 4
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 3
- KWYUFKZDYYNOTN-UHFFFAOYSA-M Potassium hydroxide Chemical compound [OH-].[K+] KWYUFKZDYYNOTN-UHFFFAOYSA-M 0.000 description 3
- 150000008065 acid anhydrides Chemical class 0.000 description 3
- 238000007792 addition Methods 0.000 description 3
- 150000001734 carboxylic acid salts Chemical class 0.000 description 3
- 239000003054 catalyst Substances 0.000 description 3
- 125000004051 hexyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 3
- 125000004491 isohexyl group Chemical group C(CCC(C)C)* 0.000 description 3
- 230000001050 lubricating effect Effects 0.000 description 3
- 150000002739 metals Chemical class 0.000 description 3
- YWFWDNVOPHGWMX-UHFFFAOYSA-N n,n-dimethyldodecan-1-amine Chemical compound CCCCCCCCCCCCN(C)C YWFWDNVOPHGWMX-UHFFFAOYSA-N 0.000 description 3
- 230000003472 neutralizing effect Effects 0.000 description 3
- 125000001117 oleyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])/C([H])=C([H])\C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 3
- 229920006395 saturated elastomer Polymers 0.000 description 3
- ALSTYHKOOCGGFT-KTKRTIGZSA-N (9Z)-octadecen-1-ol Chemical compound CCCCCCCC\C=C/CCCCCCCCO ALSTYHKOOCGGFT-KTKRTIGZSA-N 0.000 description 2
- FALRKNHUBBKYCC-UHFFFAOYSA-N 2-(chloromethyl)pyridine-3-carbonitrile Chemical compound ClCC1=NC=CC=C1C#N FALRKNHUBBKYCC-UHFFFAOYSA-N 0.000 description 2
- 0 CCC(C1(C)C)*1(C1)C11C(CC(CC2)C2C2C*C2)CC2C1C2 Chemical compound CCC(C1(C)C)*1(C1)C11C(CC(CC2)C2C2C*C2)CC2C1C2 0.000 description 2
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 2
- IAYPIBMASNFSPL-UHFFFAOYSA-N Ethylene oxide Chemical compound C1CO1 IAYPIBMASNFSPL-UHFFFAOYSA-N 0.000 description 2
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 2
- 230000032683 aging Effects 0.000 description 2
- 150000001412 amines Chemical class 0.000 description 2
- 150000001721 carbon Chemical group 0.000 description 2
- 238000006243 chemical reaction Methods 0.000 description 2
- 229910052802 copper Inorganic materials 0.000 description 2
- 239000010949 copper Substances 0.000 description 2
- 150000002148 esters Chemical class 0.000 description 2
- 125000000816 ethylene group Chemical group [H]C([H])([*:1])C([H])([H])[*:2] 0.000 description 2
- 239000004744 fabric Substances 0.000 description 2
- 125000004435 hydrogen atom Chemical group [H]* 0.000 description 2
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 2
- 125000000959 isobutyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 description 2
- 125000001972 isopentyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])C([H])([H])* 0.000 description 2
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 2
- 239000002480 mineral oil Substances 0.000 description 2
- 125000002347 octyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 2
- 229940055577 oleyl alcohol Drugs 0.000 description 2
- XMLQWXUVTXCDDL-UHFFFAOYSA-N oleyl alcohol Natural products CCCCCCC=CCCCCCCCCCCO XMLQWXUVTXCDDL-UHFFFAOYSA-N 0.000 description 2
- 125000006353 oxyethylene group Chemical group 0.000 description 2
- 125000004437 phosphorous atom Chemical group 0.000 description 2
- 238000005498 polishing Methods 0.000 description 2
- 239000000047 product Substances 0.000 description 2
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 description 2
- 238000003756 stirring Methods 0.000 description 2
- 229940014800 succinic anhydride Drugs 0.000 description 2
- DNIAPMSPPWPWGF-GSVOUGTGSA-N (R)-(-)-Propylene glycol Chemical compound C[C@@H](O)CO DNIAPMSPPWPWGF-GSVOUGTGSA-N 0.000 description 1
- 125000003903 2-propenyl group Chemical group [H]C([*])([H])C([H])=C([H])[H] 0.000 description 1
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerol Natural products OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 1
- 230000002159 abnormal effect Effects 0.000 description 1
- NIXOWILDQLNWCW-UHFFFAOYSA-N acrylic acid group Chemical group C(C=C)(=O)O NIXOWILDQLNWCW-UHFFFAOYSA-N 0.000 description 1
- 125000003342 alkenyl group Chemical group 0.000 description 1
- 239000003963 antioxidant agent Substances 0.000 description 1
- 125000003710 aryl alkyl group Chemical group 0.000 description 1
- 125000003118 aryl group Chemical group 0.000 description 1
- 125000004429 atom Chemical group 0.000 description 1
- 125000002511 behenyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 238000007664 blowing Methods 0.000 description 1
- 239000007795 chemical reaction product Substances 0.000 description 1
- 238000004140 cleaning Methods 0.000 description 1
- 238000002485 combustion reaction Methods 0.000 description 1
- 238000007906 compression Methods 0.000 description 1
- 230000006835 compression Effects 0.000 description 1
- 238000006482 condensation reaction Methods 0.000 description 1
- 238000001816 cooling Methods 0.000 description 1
- 125000004122 cyclic group Chemical group 0.000 description 1
- 125000000753 cycloalkyl group Chemical group 0.000 description 1
- 125000002704 decyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 235000014113 dietary fatty acids Nutrition 0.000 description 1
- ZBCBWPMODOFKDW-UHFFFAOYSA-N diethanolamine Chemical compound OCCNCCO ZBCBWPMODOFKDW-UHFFFAOYSA-N 0.000 description 1
- HPNMFZURTQLUMO-UHFFFAOYSA-O diethylammonium Chemical compound CC[NH2+]CC HPNMFZURTQLUMO-UHFFFAOYSA-O 0.000 description 1
- NAPSCFZYZVSQHF-UHFFFAOYSA-N dimantine Chemical compound CCCCCCCCCCCCCCCCCCN(C)C NAPSCFZYZVSQHF-UHFFFAOYSA-N 0.000 description 1
- 229910001873 dinitrogen Inorganic materials 0.000 description 1
- LAWOZCWGWDVVSG-UHFFFAOYSA-N dioctylamine Chemical compound CCCCCCCCNCCCCCCCC LAWOZCWGWDVVSG-UHFFFAOYSA-N 0.000 description 1
- 239000002270 dispersing agent Substances 0.000 description 1
- 125000003438 dodecyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 238000004945 emulsification Methods 0.000 description 1
- 238000005886 esterification reaction Methods 0.000 description 1
- 125000005678 ethenylene group Chemical group [H]C([*:1])=C([H])[*:2] 0.000 description 1
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 1
- QUSNBJAOOMFDIB-UHFFFAOYSA-O ethylaminium Chemical compound CC[NH3+] QUSNBJAOOMFDIB-UHFFFAOYSA-O 0.000 description 1
- 229940117927 ethylene oxide Drugs 0.000 description 1
- 239000000194 fatty acid Substances 0.000 description 1
- 229930195729 fatty acid Natural products 0.000 description 1
- 238000001914 filtration Methods 0.000 description 1
- 239000012530 fluid Substances 0.000 description 1
- 125000000524 functional group Chemical group 0.000 description 1
- 235000011187 glycerol Nutrition 0.000 description 1
- 238000010438 heat treatment Methods 0.000 description 1
- 125000003187 heptyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 230000001771 impaired effect Effects 0.000 description 1
- 230000000415 inactivating effect Effects 0.000 description 1
- 239000003112 inhibitor Substances 0.000 description 1
- JEIPFZHSYJVQDO-UHFFFAOYSA-N iron(III) oxide Inorganic materials O=[Fe]O[Fe]=O JEIPFZHSYJVQDO-UHFFFAOYSA-N 0.000 description 1
- 125000005645 linoleyl group Chemical group 0.000 description 1
- 239000007788 liquid Substances 0.000 description 1
- 239000000314 lubricant Substances 0.000 description 1
- 238000005555 metalworking Methods 0.000 description 1
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 1
- 125000001421 myristyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000001971 neopentyl group Chemical group [H]C([*])([H])C(C([H])([H])[H])(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 125000004433 nitrogen atom Chemical group N* 0.000 description 1
- 125000001312 palmitoyl group Chemical group O=C([*])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000000913 palmityl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 239000005077 polysulfide Substances 0.000 description 1
- 229920001021 polysulfide Polymers 0.000 description 1
- 150000008117 polysulfides Polymers 0.000 description 1
- 230000003449 preventive effect Effects 0.000 description 1
- 239000011541 reaction mixture Substances 0.000 description 1
- 230000009257 reactivity Effects 0.000 description 1
- 231100000241 scar Toxicity 0.000 description 1
- 229910001220 stainless steel Inorganic materials 0.000 description 1
- 239000010935 stainless steel Substances 0.000 description 1
- 125000004079 stearyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 239000000126 substance Substances 0.000 description 1
- 150000005846 sugar alcohols Polymers 0.000 description 1
- 125000004434 sulfur atom Chemical group 0.000 description 1
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- ZMANZCXQSJIPKH-UHFFFAOYSA-O triethylammonium ion Chemical compound CC[NH+](CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-O 0.000 description 1
- XTAZYLNFDRKIHJ-UHFFFAOYSA-O trioctylazanium Chemical compound CCCCCCCC[NH+](CCCCCCCC)CCCCCCCC XTAZYLNFDRKIHJ-UHFFFAOYSA-O 0.000 description 1
- 235000015112 vegetable and seed oil Nutrition 0.000 description 1
- 235000019871 vegetable fat Nutrition 0.000 description 1
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10M—LUBRICATING COMPOSITIONS; USE OF CHEMICAL SUBSTANCES EITHER ALONE OR AS LUBRICATING INGREDIENTS IN A LUBRICATING COMPOSITION
- C10M129/00—Lubricating compositions characterised by the additive being an organic non-macromolecular compound containing oxygen
- C10M129/02—Lubricating compositions characterised by the additive being an organic non-macromolecular compound containing oxygen having a carbon chain of less than 30 atoms
- C10M129/68—Esters
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10M—LUBRICATING COMPOSITIONS; USE OF CHEMICAL SUBSTANCES EITHER ALONE OR AS LUBRICATING INGREDIENTS IN A LUBRICATING COMPOSITION
- C10M129/00—Lubricating compositions characterised by the additive being an organic non-macromolecular compound containing oxygen
- C10M129/02—Lubricating compositions characterised by the additive being an organic non-macromolecular compound containing oxygen having a carbon chain of less than 30 atoms
- C10M129/68—Esters
- C10M129/76—Esters containing free hydroxy or carboxyl groups
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C211/00—Compounds containing amino groups bound to a carbon skeleton
- C07C211/01—Compounds containing amino groups bound to a carbon skeleton having amino groups bound to acyclic carbon atoms
- C07C211/02—Compounds containing amino groups bound to a carbon skeleton having amino groups bound to acyclic carbon atoms of an acyclic saturated carbon skeleton
- C07C211/03—Monoamines
- C07C211/08—Monoamines containing alkyl groups having a different number of carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C69/00—Esters of carboxylic acids; Esters of carbonic or haloformic acids
- C07C69/34—Esters of acyclic saturated polycarboxylic acids having an esterified carboxyl group bound to an acyclic carbon atom
- C07C69/40—Succinic acid esters
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10M—LUBRICATING COMPOSITIONS; USE OF CHEMICAL SUBSTANCES EITHER ALONE OR AS LUBRICATING INGREDIENTS IN A LUBRICATING COMPOSITION
- C10M133/00—Lubricating compositions characterised by the additive being an organic non-macromolecular compound containing nitrogen
- C10M133/02—Lubricating compositions characterised by the additive being an organic non-macromolecular compound containing nitrogen having a carbon chain of less than 30 atoms
- C10M133/04—Amines, e.g. polyalkylene polyamines; Quaternary amines
- C10M133/06—Amines, e.g. polyalkylene polyamines; Quaternary amines having amino groups bound to acyclic or cycloaliphatic carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10M—LUBRICATING COMPOSITIONS; USE OF CHEMICAL SUBSTANCES EITHER ALONE OR AS LUBRICATING INGREDIENTS IN A LUBRICATING COMPOSITION
- C10M137/00—Lubricating compositions characterised by the additive being an organic non-macromolecular compound containing phosphorus
- C10M137/02—Lubricating compositions characterised by the additive being an organic non-macromolecular compound containing phosphorus having no phosphorus-to-carbon bond
- C10M137/04—Phosphate esters
- C10M137/10—Thio derivatives
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10M—LUBRICATING COMPOSITIONS; USE OF CHEMICAL SUBSTANCES EITHER ALONE OR AS LUBRICATING INGREDIENTS IN A LUBRICATING COMPOSITION
- C10M141/00—Lubricating compositions characterised by the additive being a mixture of two or more compounds covered by more than one of the main groups C10M125/00 - C10M139/00, each of these compounds being essential
- C10M141/10—Lubricating compositions characterised by the additive being a mixture of two or more compounds covered by more than one of the main groups C10M125/00 - C10M139/00, each of these compounds being essential at least one of them being an organic phosphorus-containing compound
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10M—LUBRICATING COMPOSITIONS; USE OF CHEMICAL SUBSTANCES EITHER ALONE OR AS LUBRICATING INGREDIENTS IN A LUBRICATING COMPOSITION
- C10M145/00—Lubricating compositions characterised by the additive being a macromolecular compound containing oxygen
- C10M145/18—Macromolecular compounds obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds
- C10M145/24—Polyethers
- C10M145/26—Polyoxyalkylenes
- C10M145/38—Polyoxyalkylenes esterified
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10M—LUBRICATING COMPOSITIONS; USE OF CHEMICAL SUBSTANCES EITHER ALONE OR AS LUBRICATING INGREDIENTS IN A LUBRICATING COMPOSITION
- C10M169/00—Lubricating compositions characterised by containing as components a mixture of at least two types of ingredient selected from base-materials, thickeners or additives, covered by the preceding groups, each of these compounds being essential
- C10M169/04—Mixtures of base-materials and additives
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10M—LUBRICATING COMPOSITIONS; USE OF CHEMICAL SUBSTANCES EITHER ALONE OR AS LUBRICATING INGREDIENTS IN A LUBRICATING COMPOSITION
- C10M2205/00—Organic macromolecular hydrocarbon compounds or fractions, whether or not modified by oxidation as ingredients in lubricant compositions
- C10M2205/02—Organic macromolecular hydrocarbon compounds or fractions, whether or not modified by oxidation as ingredients in lubricant compositions containing acyclic monomers
- C10M2205/028—Organic macromolecular hydrocarbon compounds or fractions, whether or not modified by oxidation as ingredients in lubricant compositions containing acyclic monomers containing aliphatic monomers having more than four carbon atoms
- C10M2205/0285—Organic macromolecular hydrocarbon compounds or fractions, whether or not modified by oxidation as ingredients in lubricant compositions containing acyclic monomers containing aliphatic monomers having more than four carbon atoms used as base material
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10M—LUBRICATING COMPOSITIONS; USE OF CHEMICAL SUBSTANCES EITHER ALONE OR AS LUBRICATING INGREDIENTS IN A LUBRICATING COMPOSITION
- C10M2207/00—Organic non-macromolecular hydrocarbon compounds containing hydrogen, carbon and oxygen as ingredients in lubricant compositions
- C10M2207/28—Esters
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10M—LUBRICATING COMPOSITIONS; USE OF CHEMICAL SUBSTANCES EITHER ALONE OR AS LUBRICATING INGREDIENTS IN A LUBRICATING COMPOSITION
- C10M2207/00—Organic non-macromolecular hydrocarbon compounds containing hydrogen, carbon and oxygen as ingredients in lubricant compositions
- C10M2207/28—Esters
- C10M2207/287—Partial esters
- C10M2207/288—Partial esters containing free carboxyl groups
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10M—LUBRICATING COMPOSITIONS; USE OF CHEMICAL SUBSTANCES EITHER ALONE OR AS LUBRICATING INGREDIENTS IN A LUBRICATING COMPOSITION
- C10M2209/00—Organic macromolecular compounds containing oxygen as ingredients in lubricant compositions
- C10M2209/10—Macromolecular compoundss obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds
- C10M2209/103—Polyethers, i.e. containing di- or higher polyoxyalkylene groups
- C10M2209/104—Polyethers, i.e. containing di- or higher polyoxyalkylene groups of alkylene oxides containing two carbon atoms only
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10M—LUBRICATING COMPOSITIONS; USE OF CHEMICAL SUBSTANCES EITHER ALONE OR AS LUBRICATING INGREDIENTS IN A LUBRICATING COMPOSITION
- C10M2209/00—Organic macromolecular compounds containing oxygen as ingredients in lubricant compositions
- C10M2209/10—Macromolecular compoundss obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds
- C10M2209/103—Polyethers, i.e. containing di- or higher polyoxyalkylene groups
- C10M2209/105—Polyethers, i.e. containing di- or higher polyoxyalkylene groups of alkylene oxides containing three carbon atoms only
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10M—LUBRICATING COMPOSITIONS; USE OF CHEMICAL SUBSTANCES EITHER ALONE OR AS LUBRICATING INGREDIENTS IN A LUBRICATING COMPOSITION
- C10M2209/00—Organic macromolecular compounds containing oxygen as ingredients in lubricant compositions
- C10M2209/10—Macromolecular compoundss obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds
- C10M2209/103—Polyethers, i.e. containing di- or higher polyoxyalkylene groups
- C10M2209/109—Polyethers, i.e. containing di- or higher polyoxyalkylene groups esterified
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10M—LUBRICATING COMPOSITIONS; USE OF CHEMICAL SUBSTANCES EITHER ALONE OR AS LUBRICATING INGREDIENTS IN A LUBRICATING COMPOSITION
- C10M2215/00—Organic non-macromolecular compounds containing nitrogen as ingredients in lubricant compositions
- C10M2215/02—Amines, e.g. polyalkylene polyamines; Quaternary amines
- C10M2215/04—Amines, e.g. polyalkylene polyamines; Quaternary amines having amino groups bound to acyclic or cycloaliphatic carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10M—LUBRICATING COMPOSITIONS; USE OF CHEMICAL SUBSTANCES EITHER ALONE OR AS LUBRICATING INGREDIENTS IN A LUBRICATING COMPOSITION
- C10M2223/00—Organic non-macromolecular compounds containing phosphorus as ingredients in lubricant compositions
- C10M2223/02—Organic non-macromolecular compounds containing phosphorus as ingredients in lubricant compositions having no phosphorus-to-carbon bonds
- C10M2223/04—Phosphate esters
- C10M2223/045—Metal containing thio derivatives
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10N—INDEXING SCHEME ASSOCIATED WITH SUBCLASS C10M RELATING TO LUBRICATING COMPOSITIONS
- C10N2010/00—Metal present as such or in compounds
- C10N2010/04—Groups 2 or 12
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10N—INDEXING SCHEME ASSOCIATED WITH SUBCLASS C10M RELATING TO LUBRICATING COMPOSITIONS
- C10N2030/00—Specified physical or chemical properties which is improved by the additive characterising the lubricating composition, e.g. multifunctional additives
- C10N2030/06—Oiliness; Film-strength; Anti-wear; Resistance to extreme pressure
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10N—INDEXING SCHEME ASSOCIATED WITH SUBCLASS C10M RELATING TO LUBRICATING COMPOSITIONS
- C10N2030/00—Specified physical or chemical properties which is improved by the additive characterising the lubricating composition, e.g. multifunctional additives
- C10N2030/12—Inhibition of corrosion, e.g. anti-rust agents or anti-corrosives
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10N—INDEXING SCHEME ASSOCIATED WITH SUBCLASS C10M RELATING TO LUBRICATING COMPOSITIONS
- C10N2030/00—Specified physical or chemical properties which is improved by the additive characterising the lubricating composition, e.g. multifunctional additives
- C10N2030/40—Low content or no content compositions
- C10N2030/45—Ash-less or low ash content
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10N—INDEXING SCHEME ASSOCIATED WITH SUBCLASS C10M RELATING TO LUBRICATING COMPOSITIONS
- C10N2040/00—Specified use or application for which the lubricating composition is intended
- C10N2040/08—Hydraulic fluids, e.g. brake-fluids
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10N—INDEXING SCHEME ASSOCIATED WITH SUBCLASS C10M RELATING TO LUBRICATING COMPOSITIONS
- C10N2040/00—Specified use or application for which the lubricating composition is intended
- C10N2040/20—Metal working
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10N—INDEXING SCHEME ASSOCIATED WITH SUBCLASS C10M RELATING TO LUBRICATING COMPOSITIONS
- C10N2040/00—Specified use or application for which the lubricating composition is intended
- C10N2040/25—Internal-combustion engines
Definitions
- the present invention relates to an additive for a lubricating oil, an additive composition for a lubricating oil, and a lubricating oil composition containing these. More specifically, the present invention can stably impart various functions such as abrasion resistance and metal corrosion resistance to a base oil for lubricating oil (hereinafter, also simply referred to as "base oil”) over time. In addition, it is a multifunctional additive for ash-free lubricating oil that does not contain metals such as zinc, phosphorus, and sulfur, and does not generate ash when used; various types such as load resistance and metal corrosion resistance to base oil.
- the present invention relates to an additive composition for a lubricating oil capable of stably imparting the above-mentioned functions over time; and a lubricating oil composition containing each of these additives for a lubricating oil or an additive composition for a lubricating oil.
- Lubricating oil used for engine oil, hydraulic fluid, metalworking oil, etc. consists of base oil (base oil) and additives with various functions. Among the functions of the lubricating oil, wear resistance and load resistance are particularly important, and ZnDTP (zinc dithiophosphate) is generally used as a typical additive for imparting wear resistance and load resistance to the lubricating oil. Is used as a target.
- base oil base oil
- ZnDTP zinc dithiophosphate
- ZnDTP is a compound containing zinc, phosphorus, and sulfur, and metals such as zinc produce ash by combustion.
- ZnDTP is contained in the engine oil of a diesel vehicle, ash is generated by driving the engine, and this ash may promote clogging of the DPF (Diesel Particulate Filter) mounted on the diesel vehicle.
- DPF Diesel Particulate Filter
- the inclusion of phosphorus or sulfur may increase the effect on the three-way catalyst used to purify the exhaust gas of automobiles. Therefore, an ash-free type wear-resistant agent that does not contain metals such as zinc, phosphorus, and sulfur and does not generate ash is desired.
- Patent Document 1 discloses a neutralized salt composed of a monoester carboxylic acid composed of a basic acid and an aliphatic alcohol and an aliphatic amine.
- lubricating oil In addition to wear resistance, lubricating oil is required to have various performances such as emulsification resistance and metal corrosion resistance, and it is common to use a plurality of additives in combination in addition to wear resistance. Is.
- Patent Document 2 discloses a neutralized product of a condensation reaction mixture obtained by reacting a polyhydric alcohol with a polyvalent carboxylic acid for the purpose of improving metal corrosion resistance.
- a polyhydric alcohol obtained by reacting a polyhydric alcohol with a polyvalent carboxylic acid for the purpose of improving metal corrosion resistance.
- Patent Document 3 discloses a lubricating oil agent containing a polysulfide extreme pressure agent and ZnDTP in combination
- Patent Document 4 discloses a lubricating oil composition containing a phosphonate ester and ZnDTP in combination. ..
- the lubricating oils used in these machines are required to exhibit excellent lubricating performance for a long period of time even under high pressure, high load, and high temperature conditions. Further, the lubricating oil is required to have various performances such as metal corrosion resistance in addition to the load resistance, and further improvement of the lubricating oil additive has been desired.
- Patent Document 5 discloses an engine oil composition containing a combination of a glycerin fatty acid partial ester and ZnDTP.
- this engine oil composition does not have sufficient load bearing capacity, and it has been desired to develop an additive for lubricating oil which is further improved in terms of stability over time.
- An object of the present invention is to solve the above problems. Specifically, it is possible to stably impart various functions such as wear resistance and metal corrosion resistance to a base oil over time, and zinc and the like. It is an object of the present invention to provide an ashless type multifunctional additive for lubricating oil which does not contain metal, phosphorus and sulfur and does not generate ash by use, and a lubricating oil composition containing the same. Another object of the present invention is the addition of a lubricating oil capable of stably imparting various functions such as load resistance and metal corrosion resistance to the base oil while reducing the amount of ZnDTP added. It is to provide an agent composition and a lubricating oil composition containing it.
- a neutralized salt of a monoester carboxylic acid composed of monool and dibasic acid and an amine is contained in the base oil as an additive for lubricating oil.
- a lubricating oil having excellent wear resistance and metal corrosion resistance can be obtained.
- R 1 represents a single bond in which the carbons of the carbonyl group are bonded to each other, or a divalent hydrocarbon group having 1 to 4 carbon atoms
- R 2 is a hydrocarbon having 1 to 22 carbon atoms.
- AO represents one single oxyalkylene group selected from oxyalkylene groups having 2 to 4 carbon atoms or a mixed oxyalkylene group of two or more kinds
- n is the average number of moles of oxyalkylene groups represented by AO.
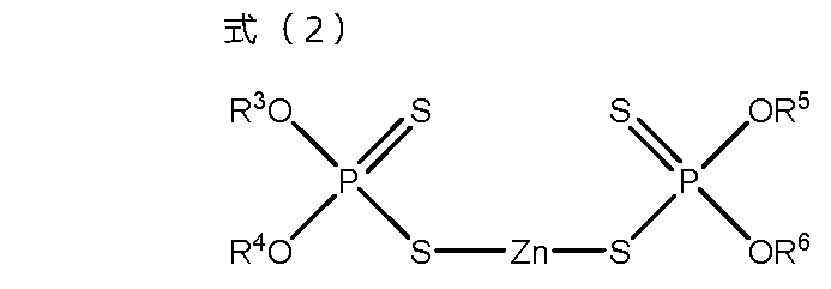
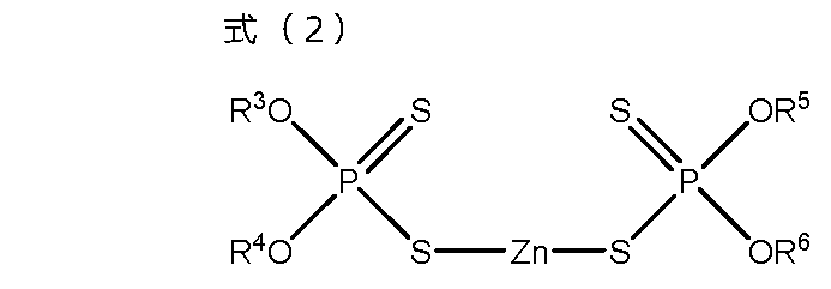
- M represents organic ammonium.
- the additive for lubricating oil of the above [1] and zinc dithiophosphate (C) represented by the formula (2) are contained, and the mass of the monoester carboxylate (A) and zinc dithiophosphate (B).
- An additive composition for a lubricating oil having a ratio (A): (B) 99: 1 to 1:99.
- R 3 to R 6 independently represent hydrocarbon groups having 1 to 24 carbon atoms.
- the additive for lubricating oil of the present invention can stably impart various functions such as wear resistance and metal corrosion resistance to the base oil for lubricating oil over time. Further, since the additive for lubricating oil of the present invention is an ash-free type additive for lubricating oil that does not generate ash with use, it does not cause clogging of a filter such as DPF, and phosphorus atom or sulfur. Since it does not contain atoms, the effect on the three-way catalyst is reduced. Therefore, the lubricating oil composition containing the lubricating oil additive and the lubricating oil base oil of the present invention is excellent in each function of abrasion resistance and metal corrosion resistance even when the amount of ZnDTP added is completely absent.
- the additive composition for lubricating oil of the present invention stably imparts various functions such as load resistance and metal corrosion resistance to the base oil for lubricating oil while reducing the amount of ZnDTP added. be able to. Therefore, the lubricating oil composition containing the lubricating oil additive composition and the lubricating oil base oil of the present invention is excellent in sustainability of each function of load bearing resistance and metal corrosion resistance, and reduces ash formation. can do.
- additive for lubricating oil of the present invention
- additive composition for lubricating oil of the present invention
- additive or An embodiment of a lubricating oil composition containing an additive composition and a base oil for a lubricating oil will be described in detail.
- the numerical range defined by using the symbol “-" includes the numerical values at both ends (upper limit and lower limit) of "-".
- “2 to 10” represents 2 or more and 10 or less.
- any higher concentration or amount can be associated with any lower concentration or amount.
- concentration or amount is specified, any higher concentration or amount can be associated with any lower concentration or amount.
- the additive of the present invention is a compound represented by the following formula (1), and this compound is a monoester carboxylic acid composed of monool and dibasic acid, an organic amine and a neutralizing salt.
- the compound represented by the formula (1) is also simply referred to as "monoester carboxylate (A)" below.
- the monoester carboxylate (A) can be used alone or in combination of two or more.
- R 1 represents a single bond in which carbons of a carbonyl group are bonded to each other, or a divalent hydrocarbon group having 1 to 4 carbon atoms.
- a divalent hydrocarbon group having 1 to 4 carbon atoms is a functional group consisting of a carbon atom and a hydrogen atom, is one selected from an alkylene group and an alkaneylene group, and is in either a linear form or a branched form. There may be. When the number of carbon atoms of the hydrocarbon group is 5 or more, the chain length becomes long, so that sufficient wear resistance and load resistance may not be obtained.
- R 1 is preferably an alkylene group or an alkaneylene group having 2 carbon atoms, and specific examples thereof include an ethylene group and an ethenylene group, but an ethylene group is more preferable.
- R 2 represents a saturated or unsaturated hydrocarbon group having 1 to 22 carbon atoms, and may be in either a linear form or a branched form.
- the R 2 for example, a methyl group, an ethyl group, a propyl group, a butyl group, a hexyl group, heptyl group, octyl group, lauryl group, myristyl group, palmityl group, stearyl group, linear saturated hydrocarbons such as behenyl group Group: Isopropyl group, isobutyl group, t-butyl group, isopentyl group, isooctyl group, 2-ethylhexyl group, isononyl group, 3,5,5-trimethylhexyl group, isodecyl group, isostearyl group, 2-octyldecyl group, Branched saturated hydrocarbon groups such as 2-octyldodecy
- R 2 is preferably a linear or branched saturated hydrocarbon group having 4 to 18 carbon atoms or a linear or branched unsaturated hydrocarbon group, and more preferably carbon. It is a branched saturated hydrocarbon group or a branched unsaturated hydrocarbon group having the number 8 to 18, and more preferably a branched unsaturated hydrocarbon group having 16 to 18 carbon atoms.
- a 2-ethylhexyl group, an isodecyl group, an isostearyl group, and an oleyl group are preferable, and an oleyl group is particularly preferable.
- AO is an oxyalkylene group having 2 to 4 carbon atoms, and may be in either a linear form or a branched form.
- examples of the AO include an oxyethylene group, an oxypropylene group, an oxybutylene group, an oxytetramethylene group and the like. It is preferably an oxyalkylene group having 2 to 3 carbon atoms, and more preferably an oxyethylene group having 2 carbon atoms.
- n represents the average number of moles of oxyalkylene group added, and n is 0 to 5. From the viewpoint of wear resistance, load resistance, and stability over time, n is preferably 1 or more. Further, n is preferably 4 or less, and particularly preferably 3 or less.
- one kind of oxyalkylene group may be bonded to a plurality (single oxyalkylene group), or two or more kinds of oxyalkylene groups may be mixed and bonded to each other (mixed oxy).
- Alkylene group When n is 2 to 5, one kind of oxyalkylene group may be bonded to a plurality (single oxyalkylene group), or two or more kinds of oxyalkylene groups may be mixed and bonded to each other (mixed oxy).
- M represents organic ammonium.
- the organic ammonium include primary, secondary, tertiary or quaternary ammonium cations in which a saturated or unsaturated hydrocarbon group having 1 to 24 carbon atoms is bonded to a nitrogen atom, and these ammonium cations are used. It may be linear, branched or cyclic. Further, the plurality of hydrocarbon groups in the secondary, tertiary and quaternary ammonium cations may be the same, or at least one hydrocarbon group may be different.
- organic ammonium examples include ethyl ammonium, diethyl ammonium, dioctyl ammonium, triethyl ammonium, trioctyl ammonium, dimethyl lauryl ammonium, dimethyl stearyl ammonium and the like. From the viewpoint of metal corrosion resistance and stability over time, tertiary ammonium is preferable.
- AO and M organic ammonium
- the value of the following formula (3) 0.5 It is preferably about 2.0, more preferably 0.6 to 1.8, and particularly preferably 0.7 to 1.5.
- the method for producing the monoester carboxylate (A) represented by the formula (1) is not particularly limited, and for example, the first step of producing the monoester carboxylic acid and the monoester obtained in the first step.
- the monoester carboxylic acid salt (A) represented by the formula (1) can be produced through a second step of neutralizing the carboxylic acid with an amine compound.
- the first step will be described.
- a method of subjecting an alcohol having a hydrocarbon group having 4 to 22 carbon atoms, or a polyether compound obtained by adding an alkylene oxide to the alcohol, and a dibasic acid, for example, at 60 to 180 ° C. Can be mentioned.
- an acid anhydride as the dibasic acid from the viewpoint of reactivity.
- the monoester carboxylic acid salt (A) can be produced by neutralizing the monoester carboxylic acid produced by the above production method with the amine compound, for example, at 20 to 60 ° C.
- the monoester carboxylic acid: amine compound is preferably in the range of 60:40 to 40:60 in molar ratio, more preferably in the range of 55:45 to 45:55. Yes, more preferably in the range of 52:48 to 48:52.
- the additive composition of the present invention contains the above monoester carboxylate (A) and the following zinc dithiophosphate (B).
- Zinc dithiophosphate (B) is a compound represented by the following formula (2), and one type can be used alone or two or more types can be used in combination.
- R 3 to R 6 independently represent hydrocarbon groups having 1 to 24 carbon atoms, and R 3 to R 6 may be the same as or different from each other.
- the hydrocarbon group having 1 to 24 carbon atoms is a saturated or unsaturated hydrocarbon group composed of a carbon atom and a hydrogen atom, and may be in either a linear form or a branched form.
- Examples of the hydrocarbon group having 1 to 24 carbon atoms include an alkyl group, an alkenyl group, a cycloalkyl group, an aryl group and an aralkyl group.
- R 3 to R 6 are preferably linear or branched alkyl groups having 3 to 18 carbon atoms, more preferably linear or branched alkyl groups having 3 to 12 carbon atoms, and branched alkyl groups having 3 to 12 carbon atoms. Is even more preferable.
- Examples of the linear alkyl group having 3 to 12 carbon atoms include a propyl group, a butyl group, a pentyl group, a hexyl group, an octyl group, a decyl group and the like, but a butyl group and a pentyl group are more preferable.
- zinc dithiophosphate (C) preferably has two or more of the above linear alkyl groups as R 3 to R 6 , and has both a linear butyl group and a linear pentyl group. Is particularly preferred.
- the branched alkyl group having 3 to 12 carbon atoms include an isopropyl group, an isobutyl group, an isopentyl group, a neopentyl group, an isohexyl group, a 2-ethylhexyl group, a 3,5,5-trimethylhexyl group and an isodecyl group.
- an isohexyl group, a 2-ethylhexyl group, and a 3,5,5-trimethylhexyl group are more preferable, and an isohexyl group is further preferable.
- Typical examples of such ZnDTP include LUBRIZOL 677A and LUBRIZOL 1371, which are commercially available from Lubrizol.
- the mixing ratio of the monoester carboxylate (A) represented by the formula (1) and the zinc dithiophosphate (B) represented by the formula (2) is 99: 1 to 1:99 in terms of mass ratio, and is preferable. It is 90:10 to 10:90, more preferably 80:20 to 20:80, and even more preferably 60:40 to 40:60. If the content of the monoester carboxylate (A) is too high, the load bearing capacity may decrease, and if the content of the monoester carboxylate (A) is too low, the load bearing capacity may be stable over time. The sex may be reduced.
- the additive composition of the present invention contains at least monoester carboxylate (A) and zinc dithiophosphate (B), and is an extreme pressure agent and wear resistant as long as the effect of the additive composition of the present invention is not impaired. It may further contain other additives such as agents, antioxidants and the like.
- the lubricating oil composition of the present invention contains the additive of the present invention or the additive composition of the present invention, and a base oil for lubricating oil.
- the lubricating oil composition containing the additive of the present invention and the base oil for lubricating oil is referred to as "lubricating oil composition (1)", and the lubricating oil containing the additive composition of the present invention and the base oil for lubricating oil is described.
- the composition is referred to as "lubricating oil composition (2)".
- various base oils for lubricating oils can be used as the base oils for lubricating oils.
- conventionally used base oils for lubricating oils such as mineral oils, highly refined mineral oils, animal and vegetable fats and oils, synthetic esters, poly ⁇ -olefins, and GTL (gas two liquid) oils can be mentioned.
- the content of the base oil for lubricating oil and the additive in the lubricating oil composition (1) of the present invention is 70 to 99.99% by mass for the base oil for lubricating oil and 0.01 to 30% by mass for the additive. ..
- the content of the lubricating base oil is preferably 80 to 99.95% by mass, more preferably 90 to 99.9% by mass.
- the content of the additive is preferably 0.05 to 20% by mass, more preferably 0.1 to 10% by mass. If the content of the additive in the lubricating oil composition (1) of the present invention is too small, sufficient wear resistance may not be obtained. If the content of the additive is too large, the metal corrosion resistance commensurate with the amount of the additive may not be obtained.
- the total content of the base oil for lubricating oil and the additives is 100% by mass.
- the contents of the base oil for lubricating oil and the additive composition in the lubricating oil composition (2) of the present invention are 70 to 99.99% by mass for the base oil for lubricating oil and 0.01 to 30 for the additive composition. It is mass%.
- the content of the lubricating base oil is preferably 80 to 99.95% by mass, more preferably 90 to 99.9% by mass.
- the content of the additive composition is preferably 0.05 to 20% by mass, more preferably 0.1 to 10% by mass. If the content of the additive composition in the lubricating oil composition (2) of the present invention is too small, sufficient load bearing capacity may not be obtained. If the content of the additive composition is too large, it may not be possible to obtain load bearing resistance and metal corrosion resistance commensurate with the added amount.
- the total content of the base oil for lubricating oil and the additive composition is 100% by mass.
- the lubricating oil compositions (1) and (2) of the present invention also require additives such as a cleaning dispersant, a viscosity index improver, a rust preventive, a corrosion inhibitor, a pour point lowering agent, and a metal inactivating agent. It can be contained accordingly.
- the order of blending, mixing, and addition of each additive is not particularly limited, and various methods can be adopted.
- monoester carboxylate (A), zinc dithiophosphate (B), and in some cases various additives are added to the base oil for lubricating oil. Then, a method of heating and mixing, a method of preparing a high-concentration solution of each additive in advance, and mixing this with a base oil for lubricating oil may be used.
- the reaction product was taken out from the pressure-resistant container and neutralized with hydrochloric acid to adjust the pH to 6 to 7, and in order to remove the contained water, it was subjected to a reduced pressure treatment at 100 ° C. for 1 hour, and finally the salt was removed by filtration. 1,200 g of the polyether compound was obtained.
- the hydroxyl value of the obtained polyether compound was 180, and the molecular weight obtained from the hydroxyl group was 312.
- 312 g (1 mol) of the above-mentioned polyether compound and 100 g (1 mol) of succinic anhydride were charged into a 1-liter glass reaction vessel equipped with a stirrer, a thermometer and a nitrogen introduction tube, and the temperature was 100 ° C.
- the compound (A) of the formula (1) shown in Table 1 is obtained by appropriately changing the oleyl alcohol, ethylene oxide, succinic anhydride, and dimethyllaurylamine in Synthesis Example 1 to other compounds and performing the operation according to Synthesis Example 1. -2)-(A-7) were synthesized. Using these compounds (A-2) to (A-7) as additives for lubricating oil, lubricating oil compositions (1-1) to (1-7) were prepared as shown in Formulation Example 1. Table 1 shows the relationships between the compounds (A-1) to (A-7) with the symbols in the formula (1) and the values in the above formula (3).
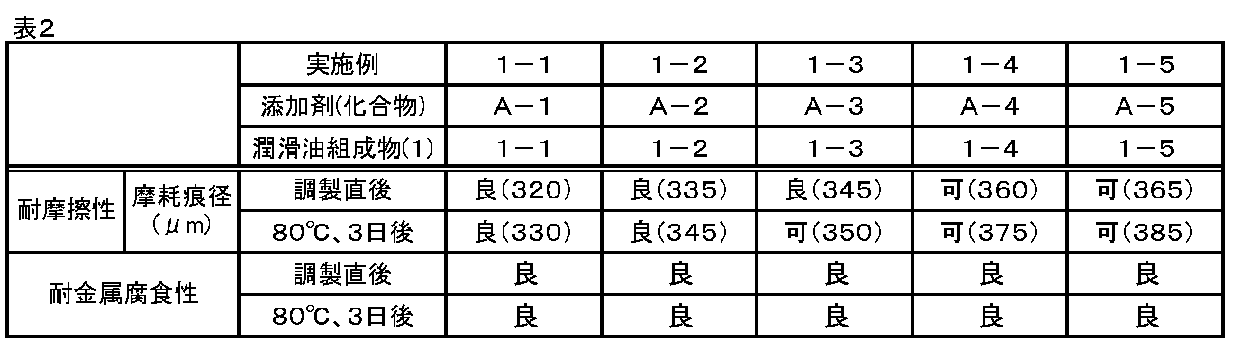
- Abrasion resistance test Abrasion resistance was evaluated with an SRV tester (OPTIMOL, Schwingungs Reihungundund Verschleiss tester type 4). The SRV test was performed with a ball / disc, and each test piece was made of SUJ-2. The test conditions were a test temperature of 150 ° C., a load of 100 N, an amplitude of 1 mm, and a frequency of 50 Hz, and the wear scar diameter was measured after the test time of 25 minutes had elapsed. The evaluation was good: less than 350 ⁇ m, acceptable: 350 ⁇ m or more and less than 400 ⁇ m, and unacceptable: 400 ⁇ m or more.
- test oil 100 ml of the test oil is placed in a 100 ml glass bottle, sealed in an air atmosphere, and allowed to stand in a constant temperature bath at 80 ° C. for 3 days, and then the abrasion resistance of the lubricating oil compositions (1-1) to (1-7). was evaluated under the same conditions as above.
- test oil 100 ml of the test oil is put into a 100 mL glass bottle, a copper wire is immersed therein, the oil is sealed in an air atmosphere, and the lubricating oil composition is allowed to stand in a constant temperature bath at 80 ° C. for 3 days.
- the metal corrosion resistance of 1-7) was evaluated under the same conditions as above.
- the compounds (A-1) to (A-5) according to the additives of the present invention have excellent wear resistance and metal corrosion resistance with respect to the base oil for lubricating oil. Can be stably applied over time. Further, since the compounds (A-1) to (A-5) do not contain a metal component such as zinc, Examples (1-1) to (A-5) in which these compounds (A-1) to (A-5) are blended are blended. The lubricating oil compositions (1-1) to (1-5) of 1-5) do not generate ash with use, and are less likely to cause clogging of filters such as DPF.
- the compounds (A-1) to (A-5) do not contain phosphorus atoms or sulfur atoms, the lubricating oil compositions (1-1) to (1) of Examples (1-1) to (1-5) are contained. The effect on the three-way catalyst due to the use of -5) is reduced.
- the compound (A-6) in which M in the formula (1) is outside the range specified in the present invention had good wear resistance, but wear resistance after aging. And the metal corrosion resistance was inferior. Further, in the compound (A-7) in which n in the formula (1) is outside the range specified in the present invention, the wear resistance after aging and the metal corrosion resistance were good, but the wear resistance immediately after production. was inferior.
- LUBRIZOL 677A alkyl group: branched hexyl group
- LUBRIZOL 1395 alkyl group: linear butyl group and linear pentyl group
- Table 4 shows the relationship between the symbols in the formula (2) and the compounds.
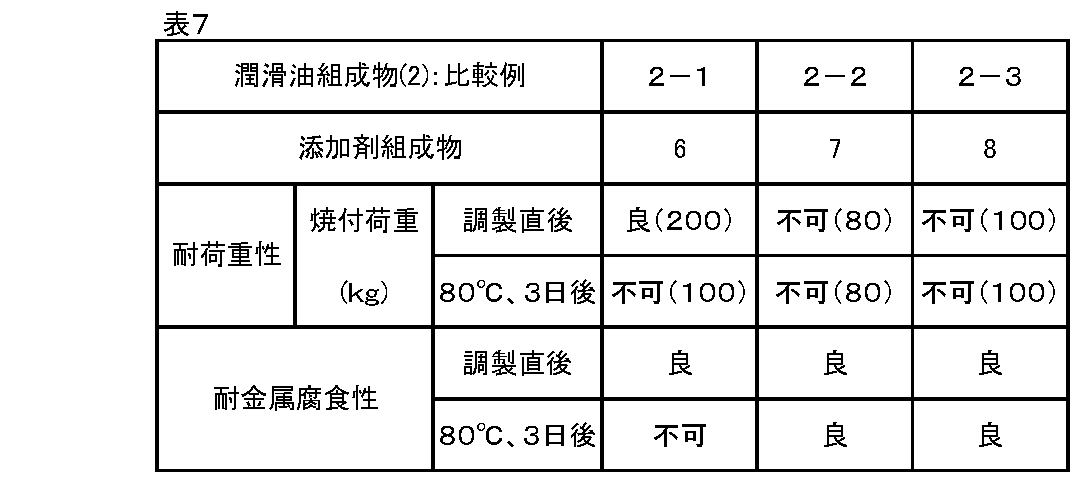
- the seizure load was evaluated with a shell 4-ball tester.
- the test piece used was made by SUJ-2.
- the test conditions were as follows: test temperature 25 ° C., rotation speed 1,800 rpm, test time 10 seconds, load 50 kg, 63 kg, 80 kg, 100 kg, 126 kg, 160 kg, 200 kg in this order.
- phenomena such as a sudden increase in friction torque and generation of abnormal noise occurred, and the load at which seizure marks were generated on the worn surface was taken as the seizure load.
- the evaluation was good: 160 kg or more, acceptable: 126 kg or more and less than 160 kg, and unacceptable: less than 126 kg.
- test oil 100 ml of the test oil is put into a 100 ml glass bottle, sealed in an air atmosphere, and allowed to stand in a constant temperature bath at 80 ° C. for 3 days, and then the load bearing capacity of the lubricating oil composition (2) (test oil) is the same as above. It was evaluated under the condition of.
- test oil 100 ml of the test oil is placed in a 100 mL glass bottle, a copper wire is immersed therein, the oil is sealed in an air atmosphere, and the lubricating oil composition is allowed to stand in a constant temperature bath at 80 ° C. for 3 days.
- Test oil The metal corrosion resistance of the above was evaluated under the same conditions as above.
- the lubricating oil compositions (2) of Examples (2-1) to (2-5) using the additive compositions 1 to 5 according to the present invention were excellent. Load resistance and metal corrosion resistance can be obtained stably over time. That is, the additive compositions 1 to 5 can impart load resistance and metal corrosion resistance to the base oil, and are also excellent in sustainability thereof. Further, since the additive compositions 1 to 5 can reduce the blending amount of zinc dithiophosphate (B) with respect to the base oil for lubricating oil (PAO), the formation of ash can be reduced.
- PEO zinc dithiophosphate
- Comparative Example (2-1) using the additive composition 6 containing the compound (A-6) in which M in the formula (1) is outside the scope specified in the present invention the lubricating oil composition immediately after preparation is used.
- Comparative Example (2-3) using the additive composition 8 composed only of chisel both the metal corrosion resistance and its durability were good, but the load resistance was inferior immediately after the preparation. ..
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Lubricants (AREA)
Abstract
Description
また本発明の他の目的は、ZnDTPの添加量を削減しつつ、基油に対して耐荷重性、耐金属腐食性などの多種の機能を経時安定的に付与することができる潤滑油用添加剤組成物およびこれを含有する潤滑油組成物を提供することである。
また濃度または量を特定した場合、任意のより高い方の濃度または量と、任意のより低い方の濃度または量とを関連づけることができる。例えば「2~10質量%」および「好ましくは4~8質量%」の記載がある場合、「2~4質量%」、「2~8質量%」、「4~10質量%」および「8~10質量%」の記載も包含される。
本発明の添加剤は下記の式(1)で示される化合物であり、この化合物はモノオールおよび二塩基酸からなるモノエステルカルボン酸と有機アミンと中和塩である。なお、式(1)で示される化合物を以下では単に「モノエステルカルボン酸塩(A)」とも言う。モノエステルカルボン酸塩(A)は、1種を単独で、または2種以上を組み合わせて用いることができる。
R1として好ましくは炭素数2のアルキレン基またはアルケニレン基であり、具体的にはエチレン基またはエテニレン基が挙げられるが、より好ましくはエチレン基である。
nはオキシアルキレン基の平均付加モル数を表し、nは0~5である。耐摩耗性、耐荷重性、経時安定性の観点から、nは1以上が好ましい。またnは4以下であることが好ましく、3以下であることが特に好ましい。nが2~5のとき、1種のオキシアルキレン基が複数結合していてもよく(単独オキシアルキレン基)、2種以上のオキシアルキレン基が混合して複数結合していてもよい(混合オキシアルキレン基)。
〔(有機アンモニウムの総炭素数)〕/〔(R2の炭素数)+(AOの炭素数)×n〕 ・・・ 式(3)
式(1)で示されるモノエステルカルボン酸塩(A)の製造法としては特に限定されず、例えば、モノエステルカルボン酸を製造する第一の工程と、第一の工程で得られたモノエステルカルボン酸をアミン化合物により中和させる第二の工程とを経て、式(1)で示されるモノエステルカルボン酸塩(A)を製造することができる。
炭素数が4~22の炭化水素基を有するアルコール、または前記アルコールにアルキレンオキサイドを付加させることによって得られるポリエーテル化合物と、二塩基酸とを、例えば60~180℃でエステル化反応を行う方法が挙げられる。本化合物を製造するためのエステル化反応では、反応性の観点から、二塩基酸として酸無水物を用いることが好ましい。また、酸無水物に対してモル比で等量のアルコールを用いて行うことが好ましい。
上記製造法で製造したモノエステルカルボン酸と、アミン化合物とを、例えば20~60℃で中和反応を行うことで、モノエステルカルボン酸塩(A)を製造することができる。耐摩耗性や耐荷重性の観点から、モノエステルカルボン酸:アミン化合物がモル比で60:40~40:60の範囲であることが好ましく、より好ましくは55:45~45:55の範囲であり、さらに好ましくは52:48~48:52の範囲である。
本発明の添加剤組成物は、上記モノエステルカルボン酸塩(A)と、下記ジチオリン酸亜鉛(B)とを含有する。
ジチオリン酸亜鉛(B)は下記の式(2)で示される化合物であり、1種を単独で、または2種以上を組み合わせて用いることができる。
R3~R6として好ましくは炭素数3~18の直鎖または分岐アルキル基であり、より好ましくは炭素数3~12の直鎖または分岐アルキル基であり、炭素数3~12の分岐アルキル基がさらに好ましい。
炭素数3~12の直鎖アルキル基としては、例えば、プロピル基、ブチル基、ペンチル基、ヘキシル基、オクチル基、デシル基などが挙げられるが、ブチル基、ペンチル基がより好ましい。また、ジチオリン酸亜鉛(C)は、R3~R6として上記直鎖アルキル基のうち2種以上を有していることが好ましく、直鎖ブチル基と直鎖ペンチル基の両方を有していることが特に好ましい。
炭素数3~12の分岐アルキル基としては、例えば、イソプロピル基、イソブチル基、イソペンチル基、ネオペンチル基、イソヘキシル基、2-エチルヘキシル基、3,5,5-トリメチルヘキシル基、イソデシル基などが挙げられるが、イソヘキシル基、2-エチルヘキシル基、3,5,5-トリメチルヘキシル基がより好ましく、イソヘキシル基がさらに好ましい。
このようなZnDTPの代表例として、Lubrizol社から市販されているLUBRIZOL 677A、LUBRIZOL 1371などが挙げられる。
本発明の潤滑油組成物は、本発明の添加剤または本発明の添加剤組成物と、潤滑油用基油とを含有する。本発明の添加剤および潤滑油用基油を含有する潤滑油組成物を「潤滑油組成物(1)」と表記し、本発明の添加剤組成物および潤滑油用基油を含有する潤滑油組成物を「潤滑油組成物(2)」と表記する。
なお、潤滑油用基油および添加剤の各含有量の合計は100質量%である。
なお、潤滑油用基油および添加剤組成物の各含有量の合計は100質量%である。
各添加剤の配合、混合、添加の順序については特に限定されず、種々の方法を採ることができる。例えば、本発明の潤滑油組成物(2)を調製する場合であれば、潤滑油用基油に、モノエステルカルボン酸塩(A)およびジチオリン酸亜鉛(B)、場合により各種添加剤を添加し、加熱混合する方法や、あらかじめ各添加剤の高濃度溶液を調製し、これを潤滑油用基油と混合する方法などを用いても良い。
式(1)で示されるモノエステルカルボン酸塩(A)の製造例を下記合成例1に示す。また、モノエステルカルボン酸塩(A)を含有する潤滑油組成物(1)の調製例を下記配合例1に示す。
攪拌機、圧力計、温度計、安全弁、ガス吹き込み管、排気管、冷却用コイル、および蒸気ジャケットを装備したステンレス製の5リットル容の耐圧容器に、オレイルアルコール1,070g(4mol)および水酸化カリウム1.3gを仕込み、窒素置換後、攪拌しながら120℃に昇温した。攪拌下、120℃、0.05~0.50MPa(ゲージ圧)の条件で、別に用意した耐圧容器からエチレンオキサイド180g(4mol)を、ガス吹き込み管を通して、窒素ガスにより加圧しながら添加した。添加終了後、同条件で内圧が一定となるまで反応させた。その後、耐圧容器から反応物を取り出し、塩酸で中和してpH6~7とし、含有する水分を除去するため、100℃、1時間、減圧処理を行い、最後に濾過により塩を除去して、1,200gのポリエーテル化合物を得た。得られたポリエーテル化合物の水酸基価は180であり、水酸基から求められる分子量は312であった。
次に攪拌装置、温度計および窒素導入管を装備したガラス製の1リットル容の反応容器に、上記で得られたポリエーテル化合物312g(1mol)と無水コハク酸100g(1mol)を仕込み、100℃で2時間反応させた。酸価の測定で99%以上の酸無水物がハーフエステル化していることを確認した後、室温まで冷却した。その後、ジメチルラウリルアミン213g(1mol)を仕込み、60℃以下で0.5時間、攪拌し中和した。これにより化合物(A-1)を得た。
化合物(A-1)~(A-7)について、式(1)中の記号との関係および上記式(3)の値を併せて表1に示す。
潤滑油用基油(ポリαオレフィン、動粘度(40℃):約50mm2/s)に対して上記の化合物(A-1)~(A-7)をそれぞれ0.5質量%配合して、実施例(1-1)~(1-5)および比較例(1-1)~(1-2)の潤滑油組成物(1-1)~(1-7)を得た。得られた潤滑油組成物(試験油)について下記の評価試験を行なった。実施例(1-1)~(1-5)の評価結果を下記表2に、比較例(1-1)~(1-2)の評価結果を下記表3に示す。
SRV試験機(OPTIMOL 社製、Schwingungs Reihungundund Verschleiss 試験機4型)にて耐摩耗性を評価した。SRV試験はボール/ディスクで行い、試験片はそれぞれSUJ-2製を用いた。試験条件は試験温度150℃、荷重100N、振幅1mm、振動数50Hzであり、試験時間25min経過後の摩耗痕径を測定した。
評価は、良:350μm未満、可:350μm以上かつ400μm未満、不可:400μm以上、とした。
耐金属腐食性として耐銅腐食性を評価した。長さ4cmに切断した銅線をP150番研磨布で研磨した。5mlスクリュー管へ試験油を2ml入れ、そこへ銅線を浸し、100℃で3時間加熱した。試験前後での表面状態を比較し、腐食の有無を評価した。
評価は、良:腐食なし、不可:腐食あり、とした。
また式(1)中のnが本発明規定の範囲外である化合物(A-7)では、経時変化後の耐摩耗性および耐金属腐食性は良好であったが、製造直後の耐摩耗性が劣っていた。
ジチオリン酸亜鉛として、Lubrizol社のLUBRIZOL 677A(アルキル基:分岐ヘキシル基)およびLUBRIZOL 1395(アルキル基:直鎖ブチル基および直鎖ペンチル基)を使用した。化合物(B-1)がLUBRIZOL 677Aであり、化合物(B-2)がLUBRIZOL 1395である。
式(2)中の記号と化合物との関係を表4に示す。
300mL~1Lの4つ口フラスコに、温度計および窒素導入管を差し込み、表5に記載の各添加剤を25℃で1時間攪拌配合して、添加剤組成物1~8を得た。
潤滑油用基油(ポリαオレフィン、動粘度(40℃):約50mm2/s)に対して表5に記載の添加剤組成物1~8をそれぞれ0.5質量%配合して、実施例(2-1)~(2-5)および比較例(2-1)~(2-3)の潤滑油組成物を得た。得られた潤滑油組成物(2)(試験油)について下記の評価試験を行なった。評価結果を表6および7に示す。
シェル4球試験機にて焼付荷重を評価した。試験片はSUJ-2製を用いた。試験条件は試験温度25℃、回転数1,800rpm、試験時間10秒、荷重50kg、63kg、80kg、100kg、126kg、160kg、200kgの順に荷重をかけて実施した。試験中に摩擦トルクの急増、異常音の発生などの現象が起き、摩耗面に焼付条痕が生成した荷重をもって焼付荷重とした。
評価は、良:160kg以上、可:126kg以上かつ160kg未満、不可:126kg未満、とした。
耐金属腐食性として耐銅腐食性を評価した。長さ4cmに切断した銅線をP150番研磨布で研磨した。5mlスクリュー管へ試験油を2ml入れ、そこへ銅線を浸し、100℃で3時間加熱した。試験前後での表面状態を比較し、腐食の有無を評価した。
評価は、良:腐食なし、不可:腐食あり、とした。
また、式(1)中のnが本発明規定の範囲外である化合物(A-7)を含有する添加剤組成物7を用いた比較例(2-2)、およびジチオリン酸亜鉛(B)のみからなる添加剤組成物8を用いた比較例(2-3)では、耐金属腐食性とその持続性はいずれも良好であったが、耐荷重性については調製直後の時点で劣っていた。
本願は、2019年3月14日出願の日本国特許出願(特願2019-047823)および2020年2月20日出願の日本国特許出願(特願2020-027132)に基づく優先権の利益を享受し、その全ての内容が参照によりここに組み込まれる。
Claims (4)
- 潤滑油用基油を70~99.99質量%、請求項1に記載の潤滑油用添加剤を0.01~30質量%含有する潤滑油組成物。
- 潤滑油用基油を70~99.99質量%、請求項2に記載の潤滑油用添加剤組成物を0.01~30質量%含有する潤滑油組成物。
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US17/439,087 US11739280B2 (en) | 2019-03-14 | 2020-03-10 | Lubricant additive, lubricant additive composition, and lubricating oil composition containing the same |
| CN202080020952.3A CN113574144B (zh) | 2019-03-14 | 2020-03-10 | 润滑油用添加剂、润滑油用添加剂组合物及含有该添加剂或添加剂组合物的润滑油组合物 |
| JP2021505088A JP7400806B2 (ja) | 2019-03-14 | 2020-03-10 | 潤滑油用添加剤、潤滑油用添加剤組成物およびこれらを含有する潤滑油組成物 |
| EP20769802.8A EP3940045A4 (en) | 2019-03-14 | 2020-03-10 | LUBRICANT OIL SUPPLEMENT, LUBRICANT OIL COMPOSITION AND LUBRICANT OIL COMPOSITION CONTAINING THESE |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2019047823 | 2019-03-14 | ||
| JP2019-047823 | 2019-03-14 | ||
| JP2020-027132 | 2020-02-20 | ||
| JP2020027132 | 2020-02-20 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2020184570A1 true WO2020184570A1 (ja) | 2020-09-17 |
Family
ID=72426406
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2020/010345 WO2020184570A1 (ja) | 2019-03-14 | 2020-03-10 | 潤滑油用添加剤、潤滑油用添加剤組成物およびこれらを含有する潤滑油組成物 |
Country Status (5)
| Country | Link |
|---|---|
| US (1) | US11739280B2 (ja) |
| EP (1) | EP3940045A4 (ja) |
| JP (1) | JP7400806B2 (ja) |
| CN (1) | CN113574144B (ja) |
| WO (1) | WO2020184570A1 (ja) |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP7505341B2 (ja) | 2020-09-08 | 2024-06-25 | 日油株式会社 | 潤滑油用添加剤組成物およびこれを含有する潤滑油組成物 |
Citations (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS5334062A (en) * | 1976-09-08 | 1978-03-30 | Mobil Oil | Lubricating composition |
| GB1548253A (en) * | 1975-05-14 | 1979-07-11 | Rohm & Haas | Malonic esters and their use as fuel and oil additives |
| JPH1067995A (ja) | 1996-08-28 | 1998-03-10 | Yachiyo Res Kk | 耐荷重性潤滑油組成物 |
| JP2004531614A (ja) * | 2001-05-22 | 2004-10-14 | ビーエーエスエフ アクチェンゲゼルシャフト | 殊にポリイソブチレンをベースとする低分子及び高分子乳化剤並びに該乳化剤の混合物 |
| JP2005002215A (ja) | 2003-06-11 | 2005-01-06 | Nippon Oil Corp | 内燃機関用潤滑油組成物 |
| JP2006096849A (ja) * | 2004-09-29 | 2006-04-13 | Japan Energy Corp | 潤滑油、流体軸受用潤滑油及びそれを用いた流体軸受 |
| JP2007131792A (ja) | 2005-11-11 | 2007-05-31 | Cosmo Sekiyu Lubricants Kk | エンジン油組成物 |
| JP2008255239A (ja) * | 2007-04-05 | 2008-10-23 | Japan Energy Corp | ギヤ油組成物 |
| JP2011214004A (ja) * | 2010-03-31 | 2011-10-27 | Infineum Internatl Ltd | 潤滑油組成物 |
| JP4806198B2 (ja) | 2005-01-25 | 2011-11-02 | ユシロ化学工業株式会社 | 環境適応型潤滑油剤 |
| JP2015168813A (ja) | 2014-03-11 | 2015-09-28 | 富士フイルム株式会社 | 潤滑剤組成物および潤滑剤組成物の製造方法 |
| WO2018178687A1 (en) * | 2017-03-30 | 2018-10-04 | Innospec Limited | Compositions and methods and uses relating thereto |
| JP2019047823A (ja) | 2018-12-27 | 2019-03-28 | アサヒビール株式会社 | 非発酵ビール様発泡性飲料 |
| JP2020027132A (ja) | 2018-08-09 | 2020-02-20 | 富士ゼロックス株式会社 | 情報処理装置およびプログラム |
Family Cites Families (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2795911B2 (ja) * | 1989-07-13 | 1998-09-10 | 出光興産株式会社 | 潤滑油組成物 |
| WO2008070307A2 (en) * | 2006-10-23 | 2008-06-12 | The Lubrizol Corporation | Antiwear agent and lubricating composition thereof |
| CA2688091C (en) * | 2007-05-24 | 2015-04-14 | The Lubrizol Corporation | Lubricating composition containing sulphur, phosphorous and ashfree antiwear agent and amine containing friction modifier |
| JP2012131879A (ja) | 2010-12-21 | 2012-07-12 | Showa Shell Sekiyu Kk | 潤滑油組成物 |
| GB201313423D0 (en) * | 2013-07-26 | 2013-09-11 | Innospec Ltd | Compositions and methods |
| JP5807833B1 (ja) * | 2015-05-16 | 2015-11-10 | 有限会社タービュランス・リミテッド | 潤滑油剤用の添加剤、及び潤滑油剤組成物 |
| JP6467377B2 (ja) * | 2016-06-29 | 2019-02-13 | 株式会社Adeka | 潤滑性組成物及び該潤滑性組成物からなるエンジン油組成物 |
-
2020
- 2020-03-10 US US17/439,087 patent/US11739280B2/en active Active
- 2020-03-10 WO PCT/JP2020/010345 patent/WO2020184570A1/ja active Application Filing
- 2020-03-10 EP EP20769802.8A patent/EP3940045A4/en active Pending
- 2020-03-10 CN CN202080020952.3A patent/CN113574144B/zh active Active
- 2020-03-10 JP JP2021505088A patent/JP7400806B2/ja active Active
Patent Citations (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB1548253A (en) * | 1975-05-14 | 1979-07-11 | Rohm & Haas | Malonic esters and their use as fuel and oil additives |
| JPS5334062A (en) * | 1976-09-08 | 1978-03-30 | Mobil Oil | Lubricating composition |
| JPH1067995A (ja) | 1996-08-28 | 1998-03-10 | Yachiyo Res Kk | 耐荷重性潤滑油組成物 |
| JP2004531614A (ja) * | 2001-05-22 | 2004-10-14 | ビーエーエスエフ アクチェンゲゼルシャフト | 殊にポリイソブチレンをベースとする低分子及び高分子乳化剤並びに該乳化剤の混合物 |
| JP2005002215A (ja) | 2003-06-11 | 2005-01-06 | Nippon Oil Corp | 内燃機関用潤滑油組成物 |
| JP2006096849A (ja) * | 2004-09-29 | 2006-04-13 | Japan Energy Corp | 潤滑油、流体軸受用潤滑油及びそれを用いた流体軸受 |
| JP4806198B2 (ja) | 2005-01-25 | 2011-11-02 | ユシロ化学工業株式会社 | 環境適応型潤滑油剤 |
| JP2007131792A (ja) | 2005-11-11 | 2007-05-31 | Cosmo Sekiyu Lubricants Kk | エンジン油組成物 |
| JP2008255239A (ja) * | 2007-04-05 | 2008-10-23 | Japan Energy Corp | ギヤ油組成物 |
| JP2011214004A (ja) * | 2010-03-31 | 2011-10-27 | Infineum Internatl Ltd | 潤滑油組成物 |
| JP2015168813A (ja) | 2014-03-11 | 2015-09-28 | 富士フイルム株式会社 | 潤滑剤組成物および潤滑剤組成物の製造方法 |
| WO2018178687A1 (en) * | 2017-03-30 | 2018-10-04 | Innospec Limited | Compositions and methods and uses relating thereto |
| JP2020027132A (ja) | 2018-08-09 | 2020-02-20 | 富士ゼロックス株式会社 | 情報処理装置およびプログラム |
| JP2019047823A (ja) | 2018-12-27 | 2019-03-28 | アサヒビール株式会社 | 非発酵ビール様発泡性飲料 |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP3940045A4 |
Also Published As
| Publication number | Publication date |
|---|---|
| JPWO2020184570A1 (ja) | 2020-09-17 |
| JP7400806B2 (ja) | 2023-12-19 |
| EP3940045A1 (en) | 2022-01-19 |
| US20220154094A1 (en) | 2022-05-19 |
| CN113574144B (zh) | 2022-12-13 |
| US11739280B2 (en) | 2023-08-29 |
| EP3940045A4 (en) | 2022-12-14 |
| CN113574144A (zh) | 2021-10-29 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP3599231B2 (ja) | トラクションドライブ用流体 | |
| CN106967479A (zh) | 润滑油组合物及其制造方法 | |
| CN104411674A (zh) | 羧酸酯作为润滑剂的用途 | |
| JP6803344B2 (ja) | 多機能性モリブデン含有化合物、製造方法および使用方法、ならびにそれを含む潤滑油組成物 | |
| JP2009523862A (ja) | 潤滑油および潤滑油添加剤濃厚液組成物 | |
| CN113710783A (zh) | 油剂添加剂及油剂组合物 | |
| JP7400806B2 (ja) | 潤滑油用添加剤、潤滑油用添加剤組成物およびこれらを含有する潤滑油組成物 | |
| JP7331919B2 (ja) | 潤滑油用添加剤、潤滑油用添加剤組成物およびこれらを含有する潤滑油組成物 | |
| JP7101779B2 (ja) | 変性油溶性ポリアルキレングリコール | |
| TWI824376B (zh) | 低抗剪強度潤滑流體 | |
| CN113825825B (zh) | 作为润滑剂添加剂的低腐蚀性有机钼化合物 | |
| US3702824A (en) | Friction reducing agent for lubricants | |
| CN114502703B (zh) | 润滑油用添加剂和含有其的润滑油组合物 | |
| JP7317188B2 (ja) | 変性油溶性ポリアルキレングリコール | |
| US12098343B2 (en) | Lubricant additive and lubricant composition comprising same | |
| JPH01301793A (ja) | 潤滑油 | |
| JP7505341B2 (ja) | 潤滑油用添加剤組成物およびこれを含有する潤滑油組成物 | |
| GB2117787A (en) | A gearbox lubricant composition based on a polyoxyalkylene fluid | |
| JP7541836B2 (ja) | モリブデンジチオカルバメートおよびそれを含有する潤滑剤組成物 | |
| EP3013928A1 (en) | Lubricating oil additive and lubricating oil composition |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 20769802 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2021505088 Country of ref document: JP Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2020769802 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 2020769802 Country of ref document: EP Effective date: 20211014 |