WO2020091036A1 - 経口投与用エダラボン懸濁剤 - Google Patents
経口投与用エダラボン懸濁剤 Download PDFInfo
- Publication number
- WO2020091036A1 WO2020091036A1 PCT/JP2019/043013 JP2019043013W WO2020091036A1 WO 2020091036 A1 WO2020091036 A1 WO 2020091036A1 JP 2019043013 W JP2019043013 W JP 2019043013W WO 2020091036 A1 WO2020091036 A1 WO 2020091036A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- edaravone
- suspension
- dispersant
- particles
- polyvinyl alcohol
- Prior art date
Links
- QELUYTUMUWHWMC-UHFFFAOYSA-N edaravone Chemical compound O=C1CC(C)=NN1C1=CC=CC=C1 QELUYTUMUWHWMC-UHFFFAOYSA-N 0.000 title claims abstract description 300
- 229950009041 edaravone Drugs 0.000 title claims abstract description 296
- 239000000725 suspension Substances 0.000 title claims abstract description 145
- 239000002245 particle Substances 0.000 claims description 145
- 239000002270 dispersing agent Substances 0.000 claims description 59
- 229920002451 polyvinyl alcohol Polymers 0.000 claims description 57
- 239000004372 Polyvinyl alcohol Substances 0.000 claims description 56
- 239000000843 powder Substances 0.000 claims description 56
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 44
- 239000000375 suspending agent Substances 0.000 claims description 40
- 239000007864 aqueous solution Substances 0.000 claims description 39
- 239000002562 thickening agent Substances 0.000 claims description 34
- 238000012360 testing method Methods 0.000 claims description 33
- 239000000230 xanthan gum Substances 0.000 claims description 33
- 235000010493 xanthan gum Nutrition 0.000 claims description 33
- 229920001285 xanthan gum Polymers 0.000 claims description 33
- 229940082509 xanthan gum Drugs 0.000 claims description 33
- 239000003814 drug Substances 0.000 claims description 32
- 239000000203 mixture Substances 0.000 claims description 31
- 239000000243 solution Substances 0.000 claims description 30
- 238000009472 formulation Methods 0.000 claims description 27
- 238000002347 injection Methods 0.000 claims description 27
- 239000007924 injection Substances 0.000 claims description 27
- 229920000609 methyl cellulose Polymers 0.000 claims description 27
- 239000001923 methylcellulose Substances 0.000 claims description 27
- 235000010981 methylcellulose Nutrition 0.000 claims description 27
- 235000003599 food sweetener Nutrition 0.000 claims description 19
- 239000003765 sweetening agent Substances 0.000 claims description 19
- 239000003381 stabilizer Substances 0.000 claims description 17
- 238000002156 mixing Methods 0.000 claims description 16
- 241000416162 Astragalus gummifer Species 0.000 claims description 13
- 229920001615 Tragacanth Polymers 0.000 claims description 13
- 239000000654 additive Substances 0.000 claims description 13
- 238000004090 dissolution Methods 0.000 claims description 13
- 239000000196 tragacanth Substances 0.000 claims description 12
- 235000010487 tragacanth Nutrition 0.000 claims description 12
- 229940116362 tragacanth Drugs 0.000 claims description 12
- 229930006000 Sucrose Natural products 0.000 claims description 11
- 239000005720 sucrose Substances 0.000 claims description 11
- 238000007922 dissolution test Methods 0.000 claims description 10
- 239000008247 solid mixture Substances 0.000 claims description 10
- 241000282412 Homo Species 0.000 claims description 9
- 235000014113 dietary fatty acids Nutrition 0.000 claims description 8
- 239000000194 fatty acid Substances 0.000 claims description 8
- 229930195729 fatty acid Natural products 0.000 claims description 8
- -1 sucrose fatty acid ester Chemical class 0.000 claims description 8
- 229940124597 therapeutic agent Drugs 0.000 claims description 8
- 239000001866 hydroxypropyl methyl cellulose Substances 0.000 claims description 6
- 235000010979 hydroxypropyl methyl cellulose Nutrition 0.000 claims description 6
- 229920003088 hydroxypropyl methyl cellulose Polymers 0.000 claims description 6
- 229960003943 hypromellose Drugs 0.000 claims description 6
- 229920000136 polysorbate Polymers 0.000 claims description 6
- 229950008882 polysorbate Drugs 0.000 claims description 5
- 238000013329 compounding Methods 0.000 claims description 4
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical group OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 claims description 3
- 238000007127 saponification reaction Methods 0.000 claims description 3
- 239000012085 test solution Substances 0.000 claims description 3
- 235000019422 polyvinyl alcohol Nutrition 0.000 claims description 2
- 206010002026 amyotrophic lateral sclerosis Diseases 0.000 abstract description 23
- 238000002360 preparation method Methods 0.000 description 40
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 33
- 229940079593 drug Drugs 0.000 description 22
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 20
- 239000007788 liquid Substances 0.000 description 19
- AMTWCFIAVKBGOD-UHFFFAOYSA-N dioxosilane;methoxy-dimethyl-trimethylsilyloxysilane Chemical compound O=[Si]=O.CO[Si](C)(C)O[Si](C)(C)C AMTWCFIAVKBGOD-UHFFFAOYSA-N 0.000 description 17
- 239000000839 emulsion Substances 0.000 description 17
- 229940083037 simethicone Drugs 0.000 description 17
- FBPFZTCFMRRESA-FSIIMWSLSA-N D-Glucitol Natural products OC[C@H](O)[C@H](O)[C@@H](O)[C@H](O)CO FBPFZTCFMRRESA-FSIIMWSLSA-N 0.000 description 16
- FBPFZTCFMRRESA-JGWLITMVSA-N D-glucitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-JGWLITMVSA-N 0.000 description 16
- 238000000034 method Methods 0.000 description 16
- 229960002920 sorbitol Drugs 0.000 description 16
- 239000000126 substance Substances 0.000 description 15
- 239000003002 pH adjusting agent Substances 0.000 description 12
- 239000003755 preservative agent Substances 0.000 description 11
- 230000002335 preservative effect Effects 0.000 description 11
- 229910000147 aluminium phosphate Inorganic materials 0.000 description 10
- 239000000600 sorbitol Substances 0.000 description 10
- 235000010356 sorbitol Nutrition 0.000 description 10
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 9
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 9
- DWAQJAXMDSEUJJ-UHFFFAOYSA-M Sodium bisulfite Chemical compound [Na+].OS([O-])=O DWAQJAXMDSEUJJ-UHFFFAOYSA-M 0.000 description 9
- DPXJVFZANSGRMM-UHFFFAOYSA-N acetic acid;2,3,4,5,6-pentahydroxyhexanal;sodium Chemical compound [Na].CC(O)=O.OCC(O)C(O)C(O)C(O)C=O DPXJVFZANSGRMM-UHFFFAOYSA-N 0.000 description 9
- 230000000996 additive effect Effects 0.000 description 9
- 239000002518 antifoaming agent Substances 0.000 description 9
- 239000008280 blood Substances 0.000 description 9
- 210000004369 blood Anatomy 0.000 description 9
- 230000006870 function Effects 0.000 description 9
- 235000010267 sodium hydrogen sulphite Nutrition 0.000 description 9
- 239000006185 dispersion Substances 0.000 description 8
- 238000005259 measurement Methods 0.000 description 8
- 239000004375 Dextrin Substances 0.000 description 7
- 229920001353 Dextrin Polymers 0.000 description 7
- 235000019425 dextrin Nutrition 0.000 description 7
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 7
- VLSOAXRVHARBEQ-UHFFFAOYSA-N [4-fluoro-2-(hydroxymethyl)phenyl]methanol Chemical compound OCC1=CC=C(F)C=C1CO VLSOAXRVHARBEQ-UHFFFAOYSA-N 0.000 description 6
- 230000001186 cumulative effect Effects 0.000 description 6
- 235000018417 cysteine Nutrition 0.000 description 6
- 238000003860 storage Methods 0.000 description 6
- 208000019505 Deglutition disease Diseases 0.000 description 5
- 150000001945 cysteines Chemical class 0.000 description 5
- 238000010828 elution Methods 0.000 description 5
- 239000007787 solid Substances 0.000 description 5
- 235000000346 sugar Nutrition 0.000 description 5
- 206010003504 Aspiration Diseases 0.000 description 4
- QFOHBWFCKVYLES-UHFFFAOYSA-N Butylparaben Chemical compound CCCCOC(=O)C1=CC=C(O)C=C1 QFOHBWFCKVYLES-UHFFFAOYSA-N 0.000 description 4
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 4
- 239000003963 antioxidant agent Substances 0.000 description 4
- WPYMKLBDIGXBTP-UHFFFAOYSA-N benzoic acid Chemical compound OC(=O)C1=CC=CC=C1 WPYMKLBDIGXBTP-UHFFFAOYSA-N 0.000 description 4
- 239000003795 chemical substances by application Substances 0.000 description 4
- 201000010099 disease Diseases 0.000 description 4
- 239000002612 dispersion medium Substances 0.000 description 4
- 230000000857 drug effect Effects 0.000 description 4
- 239000000796 flavoring agent Substances 0.000 description 4
- 235000019634 flavors Nutrition 0.000 description 4
- 239000011521 glass Substances 0.000 description 4
- QELSKZZBTMNZEB-UHFFFAOYSA-N propylparaben Chemical compound CCCOC(=O)C1=CC=C(O)C=C1 QELSKZZBTMNZEB-UHFFFAOYSA-N 0.000 description 4
- 238000004062 sedimentation Methods 0.000 description 4
- GEHJYWRUCIMESM-UHFFFAOYSA-L sodium sulfite Chemical compound [Na+].[Na+].[O-]S([O-])=O GEHJYWRUCIMESM-UHFFFAOYSA-L 0.000 description 4
- XUJNEKJLAYXESH-UHFFFAOYSA-N Cysteine Chemical compound SCC(N)C(O)=O XUJNEKJLAYXESH-UHFFFAOYSA-N 0.000 description 3
- IFQSXNOEEPCSLW-DKWTVANSSA-N L-cysteine hydrochloride Chemical compound Cl.SC[C@H](N)C(O)=O IFQSXNOEEPCSLW-DKWTVANSSA-N 0.000 description 3
- KWYUFKZDYYNOTN-UHFFFAOYSA-M Potassium hydroxide Chemical compound [OH-].[K+] KWYUFKZDYYNOTN-UHFFFAOYSA-M 0.000 description 3
- 244000131415 Zanthoxylum piperitum Species 0.000 description 3
- 235000008853 Zanthoxylum piperitum Nutrition 0.000 description 3
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 description 3
- 208000035475 disorder Diseases 0.000 description 3
- 238000009826 distribution Methods 0.000 description 3
- 230000000694 effects Effects 0.000 description 3
- 230000007774 longterm Effects 0.000 description 3
- 239000011734 sodium Substances 0.000 description 3
- 238000003756 stirring Methods 0.000 description 3
- 150000008163 sugars Chemical class 0.000 description 3
- 230000001225 therapeutic effect Effects 0.000 description 3
- WRMNZCZEMHIOCP-UHFFFAOYSA-N 2-phenylethanol Chemical compound OCCC1=CC=CC=C1 WRMNZCZEMHIOCP-UHFFFAOYSA-N 0.000 description 2
- 239000005711 Benzoic acid Substances 0.000 description 2
- VTYYLEPIZMXCLO-UHFFFAOYSA-L Calcium carbonate Chemical compound [Ca+2].[O-]C([O-])=O VTYYLEPIZMXCLO-UHFFFAOYSA-L 0.000 description 2
- 229920002134 Carboxymethyl cellulose Polymers 0.000 description 2
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 description 2
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 2
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 2
- WHUUTDBJXJRKMK-VKHMYHEASA-N L-glutamic acid Chemical compound OC(=O)[C@@H](N)CCC(O)=O WHUUTDBJXJRKMK-VKHMYHEASA-N 0.000 description 2
- 241001465754 Metazoa Species 0.000 description 2
- 208000005392 Spasm Diseases 0.000 description 2
- LSNNMFCWUKXFEE-UHFFFAOYSA-N Sulfurous acid Chemical compound OS(O)=O LSNNMFCWUKXFEE-UHFFFAOYSA-N 0.000 description 2
- 102000019197 Superoxide Dismutase Human genes 0.000 description 2
- 108010012715 Superoxide dismutase Proteins 0.000 description 2
- TVXBFESIOXBWNM-UHFFFAOYSA-N Xylitol Natural products OCCC(O)C(O)C(O)CCO TVXBFESIOXBWNM-UHFFFAOYSA-N 0.000 description 2
- 238000002835 absorbance Methods 0.000 description 2
- 238000010521 absorption reaction Methods 0.000 description 2
- 230000003078 antioxidant effect Effects 0.000 description 2
- 239000008122 artificial sweetener Substances 0.000 description 2
- 235000021311 artificial sweeteners Nutrition 0.000 description 2
- 235000010233 benzoic acid Nutrition 0.000 description 2
- 229940067596 butylparaben Drugs 0.000 description 2
- 235000010948 carboxy methyl cellulose Nutrition 0.000 description 2
- 238000007906 compression Methods 0.000 description 2
- 230000006835 compression Effects 0.000 description 2
- 238000001647 drug administration Methods 0.000 description 2
- 239000003480 eluent Substances 0.000 description 2
- 239000003205 fragrance Substances 0.000 description 2
- 239000003168 generic drug Substances 0.000 description 2
- 229930195712 glutamate Natural products 0.000 description 2
- 230000005484 gravity Effects 0.000 description 2
- 208000027866 inflammatory disease Diseases 0.000 description 2
- HEBKCHPVOIAQTA-UHFFFAOYSA-N meso ribitol Natural products OCC(O)C(O)C(O)CO HEBKCHPVOIAQTA-UHFFFAOYSA-N 0.000 description 2
- 229960002900 methylcellulose Drugs 0.000 description 2
- LXCFILQKKLGQFO-UHFFFAOYSA-N methylparaben Chemical compound COC(=O)C1=CC=C(O)C=C1 LXCFILQKKLGQFO-UHFFFAOYSA-N 0.000 description 2
- 230000004770 neurodegeneration Effects 0.000 description 2
- 208000015122 neurodegenerative disease Diseases 0.000 description 2
- 239000003960 organic solvent Substances 0.000 description 2
- 230000036542 oxidative stress Effects 0.000 description 2
- 230000036470 plasma concentration Effects 0.000 description 2
- 235000010232 propyl p-hydroxybenzoate Nutrition 0.000 description 2
- 239000004405 propyl p-hydroxybenzoate Substances 0.000 description 2
- 229960003415 propylparaben Drugs 0.000 description 2
- 239000008213 purified water Substances 0.000 description 2
- 229920006395 saturated elastomer Polymers 0.000 description 2
- 239000007974 sodium acetate buffer Substances 0.000 description 2
- 235000010265 sodium sulphite Nutrition 0.000 description 2
- LSNNMFCWUKXFEE-UHFFFAOYSA-L sulfite Chemical class [O-]S([O-])=O LSNNMFCWUKXFEE-UHFFFAOYSA-L 0.000 description 2
- 208000024891 symptom Diseases 0.000 description 2
- 230000007704 transition Effects 0.000 description 2
- 210000001364 upper extremity Anatomy 0.000 description 2
- 239000000811 xylitol Substances 0.000 description 2
- 235000010447 xylitol Nutrition 0.000 description 2
- HEBKCHPVOIAQTA-SCDXWVJYSA-N xylitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)CO HEBKCHPVOIAQTA-SCDXWVJYSA-N 0.000 description 2
- 229960002675 xylitol Drugs 0.000 description 2
- HDTRYLNUVZCQOY-UHFFFAOYSA-N α-D-glucopyranosyl-α-D-glucopyranoside Natural products OC1C(O)C(O)C(CO)OC1OC1C(O)C(O)C(O)C(CO)O1 HDTRYLNUVZCQOY-UHFFFAOYSA-N 0.000 description 1
- OWEGMIWEEQEYGQ-UHFFFAOYSA-N 100676-05-9 Natural products OC1C(O)C(O)C(CO)OC1OCC1C(O)C(O)C(O)C(OC2C(OC(O)C(O)C2O)CO)O1 OWEGMIWEEQEYGQ-UHFFFAOYSA-N 0.000 description 1
- PWKSKIMOESPYIA-UHFFFAOYSA-N 2-acetamido-3-sulfanylpropanoic acid Chemical compound CC(=O)NC(CS)C(O)=O PWKSKIMOESPYIA-UHFFFAOYSA-N 0.000 description 1
- WBZFUFAFFUEMEI-UHFFFAOYSA-M Acesulfame k Chemical compound [K+].CC1=CC(=O)[N-]S(=O)(=O)O1 WBZFUFAFFUEMEI-UHFFFAOYSA-M 0.000 description 1
- GUBGYTABKSRVRQ-XLOQQCSPSA-N Alpha-Lactose Chemical compound O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@H]1O[C@@H]1[C@@H](CO)O[C@H](O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-XLOQQCSPSA-N 0.000 description 1
- 208000024827 Alzheimer disease Diseases 0.000 description 1
- 244000144730 Amygdalus persica Species 0.000 description 1
- 108010011485 Aspartame Proteins 0.000 description 1
- 241000894006 Bacteria Species 0.000 description 1
- LSNNMFCWUKXFEE-UHFFFAOYSA-M Bisulfite Chemical compound OS([O-])=O LSNNMFCWUKXFEE-UHFFFAOYSA-M 0.000 description 1
- 206010006542 Bulbar palsy Diseases 0.000 description 1
- 206010008025 Cerebellar ataxia Diseases 0.000 description 1
- 241000207199 Citrus Species 0.000 description 1
- 235000005979 Citrus limon Nutrition 0.000 description 1
- 244000131522 Citrus pyriformis Species 0.000 description 1
- 240000000560 Citrus x paradisi Species 0.000 description 1
- 208000028698 Cognitive impairment Diseases 0.000 description 1
- FBPFZTCFMRRESA-KVTDHHQDSA-N D-Mannitol Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-KVTDHHQDSA-N 0.000 description 1
- 206010012335 Dependence Diseases 0.000 description 1
- FEWJPZIEWOKRBE-JCYAYHJZSA-N Dextrotartaric acid Chemical compound OC(=O)[C@H](O)[C@@H](O)C(O)=O FEWJPZIEWOKRBE-JCYAYHJZSA-N 0.000 description 1
- 208000012661 Dyskinesia Diseases 0.000 description 1
- KCXVZYZYPLLWCC-UHFFFAOYSA-N EDTA Chemical compound OC(=O)CN(CC(O)=O)CCN(CC(O)=O)CC(O)=O KCXVZYZYPLLWCC-UHFFFAOYSA-N 0.000 description 1
- 239000004386 Erythritol Substances 0.000 description 1
- UNXHWFMMPAWVPI-UHFFFAOYSA-N Erythritol Natural products OCC(O)C(O)CO UNXHWFMMPAWVPI-UHFFFAOYSA-N 0.000 description 1
- IMROMDMJAWUWLK-UHFFFAOYSA-N Ethenol Chemical compound OC=C IMROMDMJAWUWLK-UHFFFAOYSA-N 0.000 description 1
- 235000016623 Fragaria vesca Nutrition 0.000 description 1
- 240000009088 Fragaria x ananassa Species 0.000 description 1
- 235000011363 Fragaria x ananassa Nutrition 0.000 description 1
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 1
- PWKSKIMOESPYIA-BYPYZUCNSA-N L-N-acetyl-Cysteine Chemical compound CC(=O)N[C@@H](CS)C(O)=O PWKSKIMOESPYIA-BYPYZUCNSA-N 0.000 description 1
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 description 1
- GUBGYTABKSRVRQ-PICCSMPSSA-N Maltose Natural products O[C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@@H]1O[C@@H]1[C@@H](CO)OC(O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-PICCSMPSSA-N 0.000 description 1
- 229930195725 Mannitol Natural products 0.000 description 1
- 206010061296 Motor dysfunction Diseases 0.000 description 1
- 208000010428 Muscle Weakness Diseases 0.000 description 1
- 206010028289 Muscle atrophy Diseases 0.000 description 1
- 208000029578 Muscle disease Diseases 0.000 description 1
- 206010028372 Muscular weakness Diseases 0.000 description 1
- 108010025020 Nerve Growth Factor Proteins 0.000 description 1
- 102000007072 Nerve Growth Factors Human genes 0.000 description 1
- 102000008763 Neurofilament Proteins Human genes 0.000 description 1
- 108010088373 Neurofilament Proteins Proteins 0.000 description 1
- 208000018737 Parkinson disease Diseases 0.000 description 1
- 239000002202 Polyethylene glycol Substances 0.000 description 1
- 235000006040 Prunus persica var persica Nutrition 0.000 description 1
- 241000700159 Rattus Species 0.000 description 1
- CDBYLPFSWZWCQE-UHFFFAOYSA-L Sodium Carbonate Chemical compound [Na+].[Na+].[O-]C([O-])=O CDBYLPFSWZWCQE-UHFFFAOYSA-L 0.000 description 1
- 208000010112 Spinocerebellar Degenerations Diseases 0.000 description 1
- 244000228451 Stevia rebaudiana Species 0.000 description 1
- 208000013200 Stress disease Diseases 0.000 description 1
- 239000004376 Sucralose Substances 0.000 description 1
- 201000009594 Systemic Scleroderma Diseases 0.000 description 1
- 206010042953 Systemic sclerosis Diseases 0.000 description 1
- FEWJPZIEWOKRBE-UHFFFAOYSA-N Tartaric acid Natural products [H+].[H+].[O-]C(=O)C(O)C(O)C([O-])=O FEWJPZIEWOKRBE-UHFFFAOYSA-N 0.000 description 1
- HDTRYLNUVZCQOY-WSWWMNSNSA-N Trehalose Natural products O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@@H]1O[C@@H]1[C@H](O)[C@@H](O)[C@@H](O)[C@@H](CO)O1 HDTRYLNUVZCQOY-WSWWMNSNSA-N 0.000 description 1
- 240000000851 Vaccinium corymbosum Species 0.000 description 1
- 235000003095 Vaccinium corymbosum Nutrition 0.000 description 1
- 240000001717 Vaccinium macrocarpon Species 0.000 description 1
- 235000012545 Vaccinium macrocarpon Nutrition 0.000 description 1
- 235000017537 Vaccinium myrtillus Nutrition 0.000 description 1
- 235000002118 Vaccinium oxycoccus Nutrition 0.000 description 1
- 235000009499 Vanilla fragrans Nutrition 0.000 description 1
- 244000263375 Vanilla tahitensis Species 0.000 description 1
- 235000012036 Vanilla tahitensis Nutrition 0.000 description 1
- 235000009754 Vitis X bourquina Nutrition 0.000 description 1
- 235000012333 Vitis X labruscana Nutrition 0.000 description 1
- 240000006365 Vitis vinifera Species 0.000 description 1
- 235000014787 Vitis vinifera Nutrition 0.000 description 1
- 238000009825 accumulation Methods 0.000 description 1
- 235000010358 acesulfame potassium Nutrition 0.000 description 1
- 229960004998 acesulfame potassium Drugs 0.000 description 1
- 239000000619 acesulfame-K Substances 0.000 description 1
- 229960004308 acetylcysteine Drugs 0.000 description 1
- 239000002253 acid Substances 0.000 description 1
- 230000002378 acidificating effect Effects 0.000 description 1
- 239000008186 active pharmaceutical agent Substances 0.000 description 1
- 230000002411 adverse Effects 0.000 description 1
- 238000004220 aggregation Methods 0.000 description 1
- 230000002776 aggregation Effects 0.000 description 1
- HDTRYLNUVZCQOY-LIZSDCNHSA-N alpha,alpha-trehalose Chemical compound O[C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@@H]1O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 HDTRYLNUVZCQOY-LIZSDCNHSA-N 0.000 description 1
- 239000000605 aspartame Substances 0.000 description 1
- 235000010357 aspartame Nutrition 0.000 description 1
- IAOZJIPTCAWIRG-QWRGUYRKSA-N aspartame Chemical compound OC(=O)C[C@H](N)C(=O)N[C@H](C(=O)OC)CC1=CC=CC=C1 IAOZJIPTCAWIRG-QWRGUYRKSA-N 0.000 description 1
- 229960003438 aspartame Drugs 0.000 description 1
- 239000000305 astragalus gummifer gum Substances 0.000 description 1
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 1
- 230000001363 autoimmune Effects 0.000 description 1
- ZETCGWYACBNPIH-UHFFFAOYSA-N azane;sulfurous acid Chemical compound N.OS(O)=O ZETCGWYACBNPIH-UHFFFAOYSA-N 0.000 description 1
- 230000001580 bacterial effect Effects 0.000 description 1
- 229960004365 benzoic acid Drugs 0.000 description 1
- 235000021028 berry Nutrition 0.000 description 1
- WQZGKKKJIJFFOK-VFUOTHLCSA-N beta-D-glucose Chemical compound OC[C@H]1O[C@@H](O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-VFUOTHLCSA-N 0.000 description 1
- GUBGYTABKSRVRQ-QUYVBRFLSA-N beta-maltose Chemical compound OC[C@H]1O[C@H](O[C@H]2[C@H](O)[C@@H](O)[C@H](O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@@H]1O GUBGYTABKSRVRQ-QUYVBRFLSA-N 0.000 description 1
- 230000005540 biological transmission Effects 0.000 description 1
- 235000021014 blueberries Nutrition 0.000 description 1
- 230000003925 brain function Effects 0.000 description 1
- 239000011575 calcium Substances 0.000 description 1
- 229910000019 calcium carbonate Inorganic materials 0.000 description 1
- GBAOBIBJACZTNA-UHFFFAOYSA-L calcium sulfite Chemical compound [Ca+2].[O-]S([O-])=O GBAOBIBJACZTNA-UHFFFAOYSA-L 0.000 description 1
- 235000010261 calcium sulphite Nutrition 0.000 description 1
- 239000002775 capsule Substances 0.000 description 1
- 150000001720 carbohydrates Chemical class 0.000 description 1
- 238000005119 centrifugation Methods 0.000 description 1
- 206010008118 cerebral infarction Diseases 0.000 description 1
- 208000026106 cerebrovascular disease Diseases 0.000 description 1
- 230000008859 change Effects 0.000 description 1
- 235000020971 citrus fruits Nutrition 0.000 description 1
- 208000010877 cognitive disease Diseases 0.000 description 1
- 238000005056 compaction Methods 0.000 description 1
- 150000001875 compounds Chemical class 0.000 description 1
- 235000004634 cranberry Nutrition 0.000 description 1
- 239000013078 crystal Substances 0.000 description 1
- 229960002433 cysteine Drugs 0.000 description 1
- 230000003436 cytoskeletal effect Effects 0.000 description 1
- 230000003247 decreasing effect Effects 0.000 description 1
- 230000007812 deficiency Effects 0.000 description 1
- 229940078469 dl- cysteine Drugs 0.000 description 1
- 238000012377 drug delivery Methods 0.000 description 1
- 229940126534 drug product Drugs 0.000 description 1
- 229940088679 drug related substance Drugs 0.000 description 1
- 235000019414 erythritol Nutrition 0.000 description 1
- UNXHWFMMPAWVPI-ZXZARUISSA-N erythritol Chemical compound OC[C@H](O)[C@H](O)CO UNXHWFMMPAWVPI-ZXZARUISSA-N 0.000 description 1
- 229940009714 erythritol Drugs 0.000 description 1
- 229960004756 ethanol Drugs 0.000 description 1
- BEFDCLMNVWHSGT-UHFFFAOYSA-N ethenylcyclopentane Chemical compound C=CC1CCCC1 BEFDCLMNVWHSGT-UHFFFAOYSA-N 0.000 description 1
- 229960001617 ethyl hydroxybenzoate Drugs 0.000 description 1
- 235000010228 ethyl p-hydroxybenzoate Nutrition 0.000 description 1
- 239000004403 ethyl p-hydroxybenzoate Substances 0.000 description 1
- NUVBSKCKDOMJSU-UHFFFAOYSA-N ethylparaben Chemical compound CCOC(=O)C1=CC=C(O)C=C1 NUVBSKCKDOMJSU-UHFFFAOYSA-N 0.000 description 1
- 238000011156 evaluation Methods 0.000 description 1
- 230000002461 excitatory amino acid Effects 0.000 description 1
- 239000003257 excitatory amino acid Substances 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 239000000835 fiber Substances 0.000 description 1
- 238000010579 first pass effect Methods 0.000 description 1
- 210000001035 gastrointestinal tract Anatomy 0.000 description 1
- 230000005861 gene abnormality Effects 0.000 description 1
- 239000008103 glucose Substances 0.000 description 1
- 235000011187 glycerol Nutrition 0.000 description 1
- 150000003840 hydrochlorides Chemical class 0.000 description 1
- WGCNASOHLSPBMP-UHFFFAOYSA-N hydroxyacetaldehyde Natural products OCC=O WGCNASOHLSPBMP-UHFFFAOYSA-N 0.000 description 1
- 230000001771 impaired effect Effects 0.000 description 1
- 210000003000 inclusion body Anatomy 0.000 description 1
- 239000004615 ingredient Substances 0.000 description 1
- 238000001990 intravenous administration Methods 0.000 description 1
- 239000008101 lactose Substances 0.000 description 1
- 210000003141 lower extremity Anatomy 0.000 description 1
- VTHJTEIRLNZDEV-UHFFFAOYSA-L magnesium dihydroxide Chemical compound [OH-].[OH-].[Mg+2] VTHJTEIRLNZDEV-UHFFFAOYSA-L 0.000 description 1
- 239000000347 magnesium hydroxide Substances 0.000 description 1
- 229910001862 magnesium hydroxide Inorganic materials 0.000 description 1
- 239000000395 magnesium oxide Substances 0.000 description 1
- CPLXHLVBOLITMK-UHFFFAOYSA-N magnesium oxide Inorganic materials [Mg]=O CPLXHLVBOLITMK-UHFFFAOYSA-N 0.000 description 1
- AXZKOIWUVFPNLO-UHFFFAOYSA-N magnesium;oxygen(2-) Chemical compound [O-2].[Mg+2] AXZKOIWUVFPNLO-UHFFFAOYSA-N 0.000 description 1
- 239000000845 maltitol Substances 0.000 description 1
- 235000010449 maltitol Nutrition 0.000 description 1
- VQHSOMBJVWLPSR-WUJBLJFYSA-N maltitol Chemical compound OC[C@H](O)[C@@H](O)[C@@H]([C@H](O)CO)O[C@H]1O[C@H](CO)[C@@H](O)[C@H](O)[C@H]1O VQHSOMBJVWLPSR-WUJBLJFYSA-N 0.000 description 1
- 229940035436 maltitol Drugs 0.000 description 1
- 239000000594 mannitol Substances 0.000 description 1
- 235000010355 mannitol Nutrition 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- 230000007721 medicinal effect Effects 0.000 description 1
- 239000002609 medium Substances 0.000 description 1
- 235000006109 methionine Nutrition 0.000 description 1
- 150000002742 methionines Chemical class 0.000 description 1
- 235000010270 methyl p-hydroxybenzoate Nutrition 0.000 description 1
- 239000004292 methyl p-hydroxybenzoate Substances 0.000 description 1
- 229960002216 methylparaben Drugs 0.000 description 1
- 210000002161 motor neuron Anatomy 0.000 description 1
- 208000005264 motor neuron disease Diseases 0.000 description 1
- 201000006417 multiple sclerosis Diseases 0.000 description 1
- 210000003205 muscle Anatomy 0.000 description 1
- 230000020763 muscle atrophy Effects 0.000 description 1
- 201000000585 muscular atrophy Diseases 0.000 description 1
- 201000006938 muscular dystrophy Diseases 0.000 description 1
- 210000001087 myotubule Anatomy 0.000 description 1
- 230000001537 neural effect Effects 0.000 description 1
- 210000005044 neurofilament Anatomy 0.000 description 1
- 239000003900 neurotrophic factor Substances 0.000 description 1
- 239000006186 oral dosage form Substances 0.000 description 1
- 230000003647 oxidation Effects 0.000 description 1
- 238000007254 oxidation reaction Methods 0.000 description 1
- 239000001301 oxygen Substances 0.000 description 1
- 229910052760 oxygen Inorganic materials 0.000 description 1
- 239000000546 pharmaceutical excipient Substances 0.000 description 1
- 239000000825 pharmaceutical preparation Substances 0.000 description 1
- WVDDGKGOMKODPV-ZQBYOMGUSA-N phenyl(114C)methanol Chemical compound O[14CH2]C1=CC=CC=C1 WVDDGKGOMKODPV-ZQBYOMGUSA-N 0.000 description 1
- 229920001223 polyethylene glycol Polymers 0.000 description 1
- 229940068886 polyethylene glycol 300 Drugs 0.000 description 1
- 229940068918 polyethylene glycol 400 Drugs 0.000 description 1
- 229920002503 polyoxyethylene-polyoxypropylene Polymers 0.000 description 1
- 229940068965 polysorbates Drugs 0.000 description 1
- DJEHXEMURTVAOE-UHFFFAOYSA-M potassium bisulfite Chemical compound [K+].OS([O-])=O DJEHXEMURTVAOE-UHFFFAOYSA-M 0.000 description 1
- 229940099427 potassium bisulfite Drugs 0.000 description 1
- 235000010259 potassium hydrogen sulphite Nutrition 0.000 description 1
- RWPGFSMJFRPDDP-UHFFFAOYSA-L potassium metabisulfite Chemical compound [K+].[K+].[O-]S(=O)S([O-])(=O)=O RWPGFSMJFRPDDP-UHFFFAOYSA-L 0.000 description 1
- 235000010263 potassium metabisulphite Nutrition 0.000 description 1
- BHZRJJOHZFYXTO-UHFFFAOYSA-L potassium sulfite Chemical compound [K+].[K+].[O-]S([O-])=O BHZRJJOHZFYXTO-UHFFFAOYSA-L 0.000 description 1
- 235000019252 potassium sulphite Nutrition 0.000 description 1
- 238000004321 preservation Methods 0.000 description 1
- 201000002241 progressive bulbar palsy Diseases 0.000 description 1
- OJTDGPLHRSZIAV-UHFFFAOYSA-N propane-1,2-diol Chemical compound CC(O)CO.CC(O)CO OJTDGPLHRSZIAV-UHFFFAOYSA-N 0.000 description 1
- 238000013094 purity test Methods 0.000 description 1
- 150000003254 radicals Chemical class 0.000 description 1
- HELXLJCILKEWJH-NCGAPWICSA-N rebaudioside A Chemical compound O([C@H]1[C@H](O)[C@@H](CO)O[C@H]([C@@H]1O[C@H]1[C@@H]([C@@H](O)[C@H](O)[C@@H](CO)O1)O)O[C@]12C(=C)C[C@@]3(C1)CC[C@@H]1[C@@](C)(CCC[C@]1([C@@H]3CC2)C)C(=O)O[C@H]1[C@@H]([C@@H](O)[C@H](O)[C@@H](CO)O1)O)[C@@H]1O[C@H](CO)[C@@H](O)[C@H](O)[C@H]1O HELXLJCILKEWJH-NCGAPWICSA-N 0.000 description 1
- 235000019204 saccharin Nutrition 0.000 description 1
- CVHZOJJKTDOEJC-UHFFFAOYSA-N saccharin Chemical compound C1=CC=C2C(=O)NS(=O)(=O)C2=C1 CVHZOJJKTDOEJC-UHFFFAOYSA-N 0.000 description 1
- 229940081974 saccharin Drugs 0.000 description 1
- 239000000901 saccharin and its Na,K and Ca salt Substances 0.000 description 1
- 150000003839 salts Chemical class 0.000 description 1
- BHZOKUMUHVTPBX-UHFFFAOYSA-M sodium acetic acid acetate Chemical compound [Na+].CC(O)=O.CC([O-])=O BHZOKUMUHVTPBX-UHFFFAOYSA-M 0.000 description 1
- WXMKPNITSTVMEF-UHFFFAOYSA-M sodium benzoate Chemical compound [Na+].[O-]C(=O)C1=CC=CC=C1 WXMKPNITSTVMEF-UHFFFAOYSA-M 0.000 description 1
- 235000010234 sodium benzoate Nutrition 0.000 description 1
- 239000004299 sodium benzoate Substances 0.000 description 1
- 229960003885 sodium benzoate Drugs 0.000 description 1
- HRZFUMHJMZEROT-UHFFFAOYSA-L sodium disulfite Chemical compound [Na+].[Na+].[O-]S(=O)S([O-])(=O)=O HRZFUMHJMZEROT-UHFFFAOYSA-L 0.000 description 1
- 229940079827 sodium hydrogen sulfite Drugs 0.000 description 1
- VYKVQJFOZDGJLN-UHFFFAOYSA-M sodium hydrogen sulfite sulfurous acid Chemical compound [Na+].OS(O)=O.OS([O-])=O VYKVQJFOZDGJLN-UHFFFAOYSA-M 0.000 description 1
- 235000010199 sorbic acid Nutrition 0.000 description 1
- 239000004334 sorbic acid Substances 0.000 description 1
- 229940075582 sorbic acid Drugs 0.000 description 1
- 238000012306 spectroscopic technique Methods 0.000 description 1
- 208000003265 stomatitis Diseases 0.000 description 1
- 235000019408 sucralose Nutrition 0.000 description 1
- BAQAVOSOZGMPRM-QBMZZYIRSA-N sucralose Chemical compound O[C@@H]1[C@@H](O)[C@@H](Cl)[C@@H](CO)O[C@@H]1O[C@@]1(CCl)[C@@H](O)[C@H](O)[C@@H](CCl)O1 BAQAVOSOZGMPRM-QBMZZYIRSA-N 0.000 description 1
- 229960004793 sucrose Drugs 0.000 description 1
- 230000001629 suppression Effects 0.000 description 1
- 230000009747 swallowing Effects 0.000 description 1
- 230000009885 systemic effect Effects 0.000 description 1
- 239000011975 tartaric acid Substances 0.000 description 1
- 235000002906 tartaric acid Nutrition 0.000 description 1
- 208000019553 vascular disease Diseases 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/10—Dispersions; Emulsions
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/41—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with two or more ring hetero atoms, at least one of which being nitrogen, e.g. tetrazole
- A61K31/415—1,2-Diazoles
- A61K31/4152—1,2-Diazoles having oxo groups directly attached to the heterocyclic ring, e.g. antipyrine, phenylbutazone, sulfinpyrazone
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/26—Carbohydrates, e.g. sugar alcohols, amino sugars, nucleic acids, mono-, di- or oligo-saccharides; Derivatives thereof, e.g. polysorbates, sorbitan fatty acid esters or glycyrrhizin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/30—Macromolecular organic or inorganic compounds, e.g. inorganic polyphosphates
- A61K47/32—Macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds, e.g. carbomers, poly(meth)acrylates, or polyvinyl pyrrolidone
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/30—Macromolecular organic or inorganic compounds, e.g. inorganic polyphosphates
- A61K47/36—Polysaccharides; Derivatives thereof, e.g. gums, starch, alginate, dextrin, hyaluronic acid, chitosan, inulin, agar or pectin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/30—Macromolecular organic or inorganic compounds, e.g. inorganic polyphosphates
- A61K47/36—Polysaccharides; Derivatives thereof, e.g. gums, starch, alginate, dextrin, hyaluronic acid, chitosan, inulin, agar or pectin
- A61K47/38—Cellulose; Derivatives thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/0053—Mouth and digestive tract, i.e. intraoral and peroral administration
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0087—Galenical forms not covered by A61K9/02 - A61K9/7023
- A61K9/0095—Drinks; Beverages; Syrups; Compositions for reconstitution thereof, e.g. powders or tablets to be dispersed in a glass of water; Veterinary drenches
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/14—Particulate form, e.g. powders, Processes for size reducing of pure drugs or the resulting products, Pure drug nanoparticles
- A61K9/16—Agglomerates; Granulates; Microbeadlets ; Microspheres; Pellets; Solid products obtained by spray drying, spray freeze drying, spray congealing,(multiple) emulsion solvent evaporation or extraction
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P21/00—Drugs for disorders of the muscular or neuromuscular system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P21/00—Drugs for disorders of the muscular or neuromuscular system
- A61P21/02—Muscle relaxants, e.g. for tetanus or cramps
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/28—Drugs for disorders of the nervous system for treating neurodegenerative disorders of the central nervous system, e.g. nootropic agents, cognition enhancers, drugs for treating Alzheimer's disease or other forms of dementia
Definitions
- the present invention relates to an edaravone suspension for oral administration and a kit for preparing an edaravone suspension for oral administration.
- Edaravone is 3-methyl-1-phenyl-2-pyrazolin-5-one (see formula below):
- ALS amyotrophic lateral sclerosis
- ALS which is one of the motor neuron diseases, relies on the initial symptoms such as weakness of the hand, dyskinesia of the fingers and fiber spasm of the upper limbs, through muscle atrophy / muscular weakness, bulbar paralysis and muscle fiber spasm to breathe. It is an incurable disease leading to failure.
- ALS is divided into upper limb type, globular type, lower limb type, and mixed type depending on the onset site, and in any type, the muscle groups of the whole body are affected as the symptoms progress.
- the etiology of ALS has not been fully elucidated yet, the main etiology is (1) autoimmune theory (emergence of autoantibodies against Ca channels), (2) excitatory amino acid excess / addiction theory (increased extracellular glutamate).
- Oxidative stress disorder theory Cu / Zn superoxide dismutase (SOD) gene abnormality and free radical-induced neuronal disorder
- Cytoskeletal disorder theory neuroofilament to motor neurons Accumulation and appearance of inclusion bodies
- deficiency of neurotrophic factor have been proposed as hypotheses.
- edaravone is used as a therapeutic agent for ALS, but edaravone as a therapeutic agent for ALS is provided only as an injectable agent, which is less burdensome to patients and caregivers, and a desirable oral administration preparation is desirable in terms of QOL. ing.
- oral preparations that are bioequivalent to injections in oral preparations where various factors such as gastrointestinal tract absorption and first-pass effects affect bioavailability Obtaining formulations is a daunting task.
- Non-Patent Documents 1 to 3 describe CMC-Na suspensions of edaravone, but when these were administered to animals, it was described that all had low bioavailability.
- Patent Document 3 describes a liquid for oral administration of edaravone using an aqueous tragacanth gum solution, and describes that a sufficient blood concentration was obtained in rats.
- Patent Document 4 describes an edaravone solution having excellent absorbability, but this solution has a dose of 100 mL due to the low solubility of edaravone in water. It is also unfavorable for patient adherence.
- An object of the present invention is to provide an oral edaravone suspension for oral administration that can reduce the burden on ALS patients and caregivers and can achieve the same ALS therapeutic effect as an injection.
- the present inventors as a result of diligent studies, by blending a dispersant, to uniformly disperse the edaravone particles in water, and maintain the dispersion state, even when the edaravone particles settled after storage. It was found that an edaravone suspension for oral administration, which is excellent in terms of formulation, can be prepared because it can be rapidly redispersed by light shaking. Furthermore, based on the findings of Non-Patent Documents 1 to 3, the prepared oral edaravone suspension for oral administration is a suspension considered to be disadvantageous in terms of bioavailability based on the results of animal experiments so far. Nevertheless, they found that they exhibited unexpectedly excellent bioavailability when administered to humans, and completed the present invention. Means for solving the problems relate to the following ⁇ 1> to ⁇ 23>, but are not limited to these.
- ⁇ 1> A human edaravone suspension for oral administration containing edaravone particles, a dispersant, and water.
- the suspending agent according to ⁇ 1> which is a dispersant having a transmitted scattered light intensity of 1% or more.
- the suspending agent according to ⁇ 1> which is a dispersant having a contact angle of 80 ° or less.
- ⁇ 5> The suspending agent according to ⁇ 4>, wherein the dispersant is one or two selected from polyvinyl alcohol and methyl cellulose.
- ⁇ 6> The suspending agent according to ⁇ 4> or ⁇ 5>, wherein the dispersant is polyvinyl alcohol.
- ⁇ 7> polyvinyl alcohol, saponification degree is 86.5 to 89.0, and kinematic viscosity at 20 ° C. of a 4% aqueous solution is 3mm 2 /s ⁇ 55.7mm 2 / s, ⁇ 4> ⁇ Any one suspension agent of ⁇ 6>.
- ⁇ 8> The suspending agent according to any one of ⁇ 1> to ⁇ 7>, in which the compounding amount of the dispersant is 0.001% (w / v) to 1.0% (w / v).
- ⁇ 9> The suspending agent according to any one of ⁇ 1> to ⁇ 8>, further including a thickener.
- ⁇ 10> The suspending agent according to ⁇ 9>, wherein the thickening agent is one or two selected from xanthan gum and tragacanth powder.
- ⁇ 11> The suspending agent according to ⁇ 9> or ⁇ 10>, wherein the thickener is xanthan gum.
- ⁇ 12> The suspending agent according to any one of ⁇ 9> to ⁇ 11>, wherein the content of the thickener is 0.1% (w / v) to 1.2% (w / v).
- ⁇ 13> The suspension according to any one of ⁇ 1> to ⁇ 12>, in which the edaravone particles in the suspension have a D50 particle size of 10 ⁇ m to 100 ⁇ m and a D90 particle size of 50 ⁇ m to 300 ⁇ m.
- ⁇ 15> The suspending agent according to any one of ⁇ 1> to ⁇ 14>, further including one or more additives selected from sweeteners, stabilizers and pH adjusters.
- ⁇ 16> The suspending agent according to any one of ⁇ 1> to ⁇ 15>, wherein the viscosity of the suspending agent is 50 mPa ⁇ s to 1750 mPa ⁇ s. ⁇ 17>
- a dissolution test test solution: first solution, paddle rotation speed: 50 rpm
- the dissolution rate of edaravone is 80% or more 30 minutes after the start of the test ⁇ 1> to ⁇ 17.
- a human edaravone suspension preparation kit for oral administration which comprises (A) a solid composition containing edaravone particles and (B) a dispersant solution.
- a human oral edaravone suspension showing an average Cmax of plasma edaravone of 500 to 2500 ng / mL and an average AUC 0- ⁇ of 1000 to 2500 h * ng / mL when 90 to 120 mg of edaravone is orally administered to a human. Turbidity agent.
- ⁇ 22> When a crossover test was conducted in which a suspension containing 90 to 120 mg of edaravone was orally administered to humans using the injection of edaravone as a control formulation, the ratio of the geometric mean Cmax to the control formulation, the AUC 0- ⁇ Edaravone suspension for human oral administration, in which any ratio of geometric mean values falls between 0.8 and 1.25.
- a therapeutic agent for ALS characterized in that a single dose of the formulation by oral administration is 1 to 20 mL, and the dose contains 50 to 210 mg of edaravone.
- an oral edaravone suspension for oral administration that can reduce the burden on ALS patients and caregivers and can achieve the same ALS therapeutic effect as an injection.
- the suspension for human oral administration of the present invention contains edaravone particles, a dispersant, and water.
- the suspension of the present invention reduces the burden on ALS patients and caregivers, and has an ALS therapeutic effect equivalent to that of an injection.
- the suspending agent of the present invention may further contain a thickener, if necessary.
- a thickener when administered to humans, the variation in drug blood concentration among patients becomes very small, and more stable drug effect can be expected.
- the edaravone in the present invention is a known compound, and can be easily synthesized by those skilled in the art by the method described in, for example, Japanese Patent Publication No. 5-31523.
- the edaravone particles contained in the suspending agent of the present invention are solid particles containing edaravone, may be composed only of edaravone, or may contain other components, and edaravone is a crystal. It may be in a state or an amorphous state.
- the particle size of the edaravone particles in the suspension is not particularly limited, but from the viewpoint of maintaining a stable dispersed state in the suspension, rapid absorption in the body, and no feeling of roughness during administration. , D50 particle size (volume-based cumulative 50% particle size) is in the range of 10 ⁇ m to 100 ⁇ m, and D90 particle size (volume-based cumulative 90% particle size) is preferably in the range of 50 ⁇ m to 300 ⁇ m.
- the D50 particle size is in the range of 20 ⁇ m to 80 ⁇ m
- the D90 particle size is in the range of 100 ⁇ m to 250 ⁇ m.
- the cumulative 50% particle size or the cumulative 90% particle size is the particle size on a volume basis.
- the particle size distribution of the edaravone particles in the suspension is measured by dispersing a part of the suspension in a dispersion medium for measurement (edaravone saturated aqueous solution) and using a laser diffraction particle size distribution device (Sympatec / HELOS & CUVETTE).
- the concentration (blending amount) of edaravone particles blended in the suspension of the present invention is the optimal dose of edaravone and the amount of suspension provided to ALS patients (single dose, daily dose, 1 week).
- the dose can be appropriately adjusted depending on the dose, the dose for 10 days, etc., but an amount that can maintain the form as a suspension is suitable.
- the concentration of edaravone particles may be 2 mg / mL or more, which is equal to or higher than the saturated solubility of edaravone in a dispersion medium (for example, water), when the edaravone particles are composed only of edaravone, and preferably 0.06%.
- the suspending agent of the present invention contains a dispersant.
- a dispersant for example, a senor, a senor, a senor, or a senor.
- the suspension of the present invention the edaravone particles maintain a good dispersion state, even if the edaravone particles settled after long-term storage, promptly by shaking (for example, manual or mechanical shaking) Can be redistributed into The behavior of redispersion can be confirmed visually or by spectroscopic techniques (eg, using a laser diffraction particle size analyzer).
- any dispersant may be used as long as it can disperse edaravone particles in water without forming secondary aggregation.
- a dispersant having a transmitted scattered light intensity of 1% or more can be mentioned.
- 1% or more means a range of 1% to 100%.
- a dispersant having a transmitted scattered light intensity of X% or more means a scattered scattered light intensity ( ⁇ T) immediately below the liquid surface of a liquid obtained by mixing 40 mL of a 0.1% (w / v) aqueous dispersant solution and 840 mg of edaravone. %) Means a dispersant of X% or more when measured.
- a dispersant having a transmitted / scattered light intensity of 1% or more is obtained by mixing 40 mL of a 0.1% (w / v) dispersant aqueous solution and 840 mg of edaravone (D50: 37 ⁇ m, D90: 143 ⁇ m) and stirring the mixture for 30 minutes or more. It means a dispersant that is 1% or more when the intensity of transmitted scattered light ( ⁇ T%) immediately below the liquid surface of the liquid obtained by stirring is measured.
- the transmitted scattered light intensity means that 20 mL of the liquid is filled in a cylindrical sample bottle (inner diameter 25 mm ⁇ outer diameter 27.5 mm ⁇ height 72 mm), and is used in TURBISCAN Tower (manufactured by Formulation) (25 ° C.), It means the value 10 minutes after the start of measurement of the intensity of transmitted scattered light at a sample bottle height of 39 to 40 mm.
- the dispersant having a transmitted / scattered light intensity of 1% or more include polyvinyl alcohol, sucrose fatty acid ester, polysorbate, methyl cellulose, and hypromellose.
- a dispersant having a contact angle of 80 ° or less can also be preferably used.
- 80 ° or less means 0 ° to 80 °.
- a dispersant having a contact angle of 80 ° or less means that when a droplet of a saturated edaravone aqueous solution in which 0.1% (w / v) of the dispersant is dissolved is dropped onto an edaravone tablet, the tangent to the droplet and the edaravone tablet surface Means a dispersant having an angle (contact angle) of 80 ° or less.
- the edaravone tablet refers to edaravone (D50: 37 ⁇ m, D90: 143 ⁇ m) 120 mg compression-molded with a tableting pressure of 800 kg with a flat punch having a diameter of 8 mm, and the contact angle is measured by a contact angle measuring device (Kyowa CAX-150 manufactured by Interfacial Science Co., Ltd.) means the measurement under the following conditions.
- Syringe used glass, 1mL Needle: 23 gauge Liquid volume: 1 ⁇ L Measurement time: 3.1 seconds later
- Examples of the dispersant having a contact angle of 80 ° or less include polyvinyl alcohol, sucrose fatty acid ester, polysorbate, hypromellose and the like.
- preferred dispersants include one or more selected from the group consisting of polyvinyl alcohol, sucrose fatty acid ester, polysorbate, methyl cellulose and hypromellose, and particularly preferably from the group consisting of polyvinyl alcohol and methyl cellulose.
- the polyvinyl alcohol, saponification degree is 86.5 to 89.0
- kinematic viscosity range at 20 ° C. of a 4% aqueous solution is 3mm 2 /s ⁇ 55.7mm 2 / s, recommended as pharmaceutical additives
- Gohsenol EG-03P, EG-05P, EG-05PW, EG-18P, EG-22P, EG-30P, EG-30PW, EG-40P, EG-40PW, EG- sold by Nippon Synthetic Chemical Industry. 48P is mentioned.
- the dispersant any one kind may be used alone, or two or more kinds may be used in combination.
- the blending amount of the dispersant may be selected within a range in which the edaravone particles are dispersed and does not adversely affect the manufacturability, and is usually 0.001% (w / v) to 1.0% (w / v), preferably Is 0.005% (w / v) to 0.5% (w / v), most preferably 0.01% (w / v) to 0.1% (w / v).
- the suspending agent of the present invention may contain a thickener in order to maintain the dispersed state of the edaravone particles for a long time.
- a thickener By containing a thickener, even patients with dysphagia can easily drink without the risk of aspiration, and when administered to humans, the variation in blood drug concentration between patients is extremely small, making it more stable. It has the effect of being able to expect the desired medicinal effects.
- the thickener it is possible to use a thickener known in the formulation, specifically, for example, carmellose sodium, dextrin, tragacanth powder, xanthan gum and the like, from the aspect of storage stability of edaravone tragacanth. Finally, xanthan gum is preferred, and xanthan gum is most preferred.
- any one kind may be used alone, or two or more kinds may be used in combination.
- the blending amount of the thickener may be 0.1% (w / v) to 1.2% (w / v), preferably 0.2% (w / v) to 1. 0.0% (w / v), most preferably 0.3% (w / v) to 0.5% (w / v).
- a thickener is advantageous because it can give viscosity to the suspension and makes it easy for a patient with dysphagia to swallow and prevent aspiration.
- the viscosity of such a suspending agent may be within a range not exceeding 1750 mPa ⁇ s, preferably within a range of 50 mPa ⁇ s to 1750 mPa ⁇ s, and most preferably within a range of 150 mPa ⁇ s to 900 mPa ⁇ s. is there.
- the blending amount of the thickener is not limited to the above description, and can be appropriately adjusted to the above viscosity.
- the combination of a thickening agent has an unexpected effect that it can minimize the variation in the blood concentration transition of edaravone among patients when administered to patients, and thus has a stable efficacy for all patients. Can be expected.
- the suspension for oral administration of the present invention may contain a sweetener for the purpose of adjusting the taste as a preparation for oral administration, and increasing the density of the solution of the suspension to delay the sedimentation of edaravone particles.
- a sweetener for the purpose of adjusting the taste as a preparation for oral administration, and increasing the density of the solution of the suspension to delay the sedimentation of edaravone particles.
- sugars can increase the blending ratio as compared with other additives and can contribute to increasing the density of the solution. Further, by dissolving the saccharides, the density of the solution can be made closer to that of the particles, which can also contribute to suppressing the sedimentation rate of the particles.
- the density of the suspending agent is preferably close to that of edaravone particles, and is more than the range exceeding the density of water, preferably in the range of 1 g / mL to 1.5 g / mL, and most preferably 1.05 g / mL to 1.2 g. Within the range of / mL.
- sweetener examples include sugars, artificial sweeteners, non-sugar sweeteners, and the like
- specific examples of sugars include, for example, mannitol, sorbitol, xylitol, maltitol, erythritol, sucrose, trehalose, lactose, Maltose, glucose, glycerin, and the like.
- specific examples of artificial sweeteners include sucralose, aspartame, acesulfame potassium, saccharin, and the like
- non-sugar sweeteners include somatine, stevia extract, and the like. Is mentioned. Of these, sorbitol, xylitol, or sucrose is preferable, sorbitol or sucrose is more preferable, and sorbitol is most preferable.
- the sweetener any one kind may be used alone, or two or more kinds may be used in combination.
- the blending amount of the sweetener can be appropriately adjusted in view of the taste of the user and suppression of sedimentation of particles.
- the blending amount of sorbitol is 5% (w / v) to 70% (w / v). ), Preferably 10% (w / v) to 60% (w / v), more preferably 20% (w / v) to 50% (w / v).
- the pharmaceutical ingredient edaravone in the suspension of the present invention has a property of being easily oxidized by dissolved oxygen in the liquid, it is preferable to add a known stabilizer.
- stabilizers include antioxidants such as sulfite, bisulfite, and pyrosulfite, cysteines, methionines, polyethylene glycol, polyoxyethylene polyoxypropylene glycol, and EDTA.
- antioxidants such as sulfite, bisulfite, and pyrosulfite
- cysteines such as sulfite, bisulfite, and pyrosulfite
- methionines polyethylene glycol, polyoxyethylene polyoxypropylene glycol, and EDTA.
- Ammonium hydrogen sulfite (NH 4 HSO 3 ), and pyrosulfite salts include sodium pyrosulfite (Na 2 S 2 O 5 ), potassium pyrosulfite (K 2 S 2 O 5 ), and the like.
- cysteines include L-cysteine, DL-cysteine, N-acetylcysteine, and their hydrochlorides. Most preferably, sodium bisulfite is used as the antioxidant and L-cysteine hydrochloride is used as the cysteines.
- the amount of the antioxidant and cysteine added is 0.001% (w / v) to 0.5% (w / v), particularly 0.01% (w / v) to 0. 2% (w / v) is preferable, and for cysteines 0.005% (w / v) to 0.5% (w / v), especially 0.01% (w / v) to 0.2% (W / v) is preferred.
- the suspending agent of the present invention contains a pH adjusting agent.
- the pH adjuster can be used in an amount to adjust the pH of the suspension to the range of 2.5 to 6.0, preferably 3.0 to 4.5.
- either a base or an acid can be used to adjust to the desired pH.
- an acidic pH adjuster for example, hydrochloric acid, phosphoric acid, acetic acid, citric acid, tartaric acid and the like can be mentioned, preferably phosphoric acid.
- a basic pH adjuster eg, sodium hydroxide, potassium hydroxide, calcium carbonate, magnesium oxide, magnesium hydroxide, etc. can be mentioned, preferably sodium hydroxide). ..
- the suspension of the present invention may further contain pharmaceutically acceptable formulation additives such as a fragrance, a preservative and an antifoaming agent, if necessary.
- pharmaceutically acceptable formulation additives such as a fragrance, a preservative and an antifoaming agent, if necessary.
- flavors include various flavors such as citrus flavors (orange, lemon, grapefruit, etc.), peach, grape, vanilla, soda, and berry flavors (strawberry, cranberry, blueberry, etc.). For example, it may be 0.05% (w / v) to 0.2% (w / v).
- antifoaming agent examples include simethicone emulsion, fatty acid ester, polysorbates, ethanol and the like, and as the compounding amount of the antifoaming agent, for example, 0.01% (w / v) to 0.05% (w / v) Is mentioned.
- the preservative examples include methylparaben, ethylparaben, propylparaben, butylparaben, benzoic acid, sorbic acid, sodium benzoate, benzyl alcohol, phenylethanol, etc., and the compounding amount of the preservative is, for example, 0.01% ( w / v) to 0.5% (w / v).
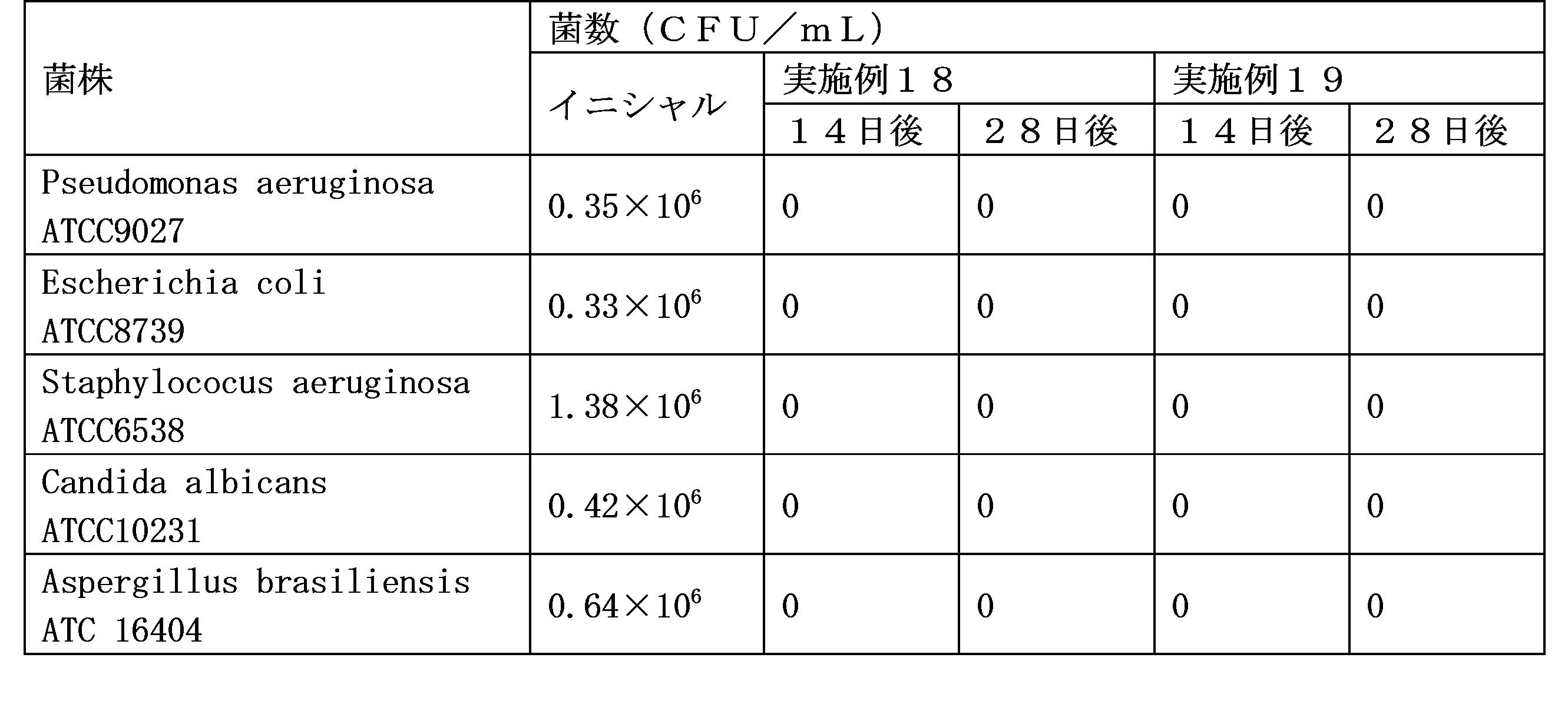
- the suspension of the present invention does not require the addition of a preservative because the growth of bacteria is not observed without the preservative.
- the liquid medium (dispersion medium) used in the suspension for oral administration of the present invention is preferably water, and may contain a pharmaceutically acceptable organic solvent.
- organic solvent examples include propylene glycol (1,2-propanediol), polyethylene glycol 300, polyethylene glycol 400, ethanol and the like.
- composition combinations in the suspension of the present invention are exemplified below, but are not limited thereto.
- Edaravone particles polyvinyl alcohol (dispersant), xanthan gum (thickener), sorbitol (sweetener), sodium bisulfite (stabilizer), L-cysteine hydrochloride (stabilizer), phosphoric acid (pH Regulator), sodium hydroxide (pH regulator), simethicone emulsion (antifoam) and water.
- Edaravone particles polyvinyl alcohol (dispersant), tragacanth powder (thickener), sorbitol (sweetener), sodium bisulfite (stabilizer), L-cysteine hydrochloride (stabilizer), phosphoric acid ( pH adjuster), sodium hydroxide (pH adjuster), simethicone emulsion (antifoam) and water.
- Edaravone particles sucrose fatty acid ester (dispersant), xanthan gum (thickener), sucrose (sweetener), sodium bisulfite (stabilizer), L-cysteine hydrochloride (stabilizer), acetic acid ( pH adjuster), sodium hydroxide (pH adjuster), simethicone emulsion (antifoam) and water.
- Edaravone particles polyvinyl alcohol (dispersant), xanthan gum (thickener), sorbitol (sweetener), sodium bisulfite (stabilizer), phosphoric acid (pH adjuster), sodium hydroxide (pH adjuster) ), Simethicone emulsion (antifoam) and water.
- Edaravone particles polyvinyl alcohol (dispersant), xanthan gum (thickener), sorbitol (sweetener), sodium bisulfite (stabilizer), L-cysteine hydrochloride (stabilizer), phosphoric acid (pH Regulator), sodium hydroxide (pH regulator), simethicone emulsion (antifoam), fragrance and water.
- the suspension of the present invention maintains a good dispersion state of edaravone particles for a long time, so that even if a fixed amount of all liquid is administered, the drug content can always be kept uniform.
- edaravone particles settled after long-term storage they can be rapidly redispersed by shaking, and the chemical stability of edaravone is not impaired during such long-term storage, and preservatives are preserved. It has features such as no bacterial growth without the addition of agents.
- the formulation of the present invention by further blending a thickener, since it has an appropriate viscosity, it is easy to take even for patients with dysphagia and the risk of aspiration is reduced, and when administered to humans. There is also an effect that the variation in the blood concentration of the drug among patients can be reduced, and a stable drug effect can be expected.
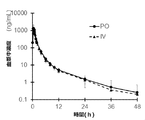
- the suspension of the present invention exhibits excellent bioavailability, it is administered with an edaravone injection currently used as an ALS therapeutic agent in clinical settings, for example, the Japanese trade name "Radicut” (registered trademark) (60 mg as edaravone.
- the drug concentration in the blood is similar to that in the case of intravenous administration over 1 hour), but the oral dose is very low as edaravone dose, specifically 90-120 mg, more specifically 100-110 mg, most Specifically, it can be achieved with 105 mg.
- the oral dose of the suspension of the present invention as edaravone is 90 to 120 mg, preferably 100 to 110 mg, and most preferably 105 mg.
- the control formulation of edaravone injection is 60 mg of edaravone over 1 hour. Be injected intravenously.
- the suspension of the present invention showed that the lower limit of the 90% confidence interval of the ratio of the Cmax geometric mean value with respect to the control preparation exceeded 0.8 and the AUC 0- ⁇ geometric mean value with respect to the control preparation.
- the 90% confidence interval for the ratio can fall within the range of 0.8-1.25.
- the suspension of the present invention had a 90% confidence interval of the ratio of the Cmax geometric mean value with respect to the control preparation within the range of 0.8 to 2.0, and the AUC 0- ⁇ geometric mean value with respect to the control preparation.
- the 90% confidence interval for the ratio of can fall within the range of 0.8 to 1.25.
- the 90% confidence interval of the ratio of the Cmax geometric mean value to the control preparation falls within the range of 0.8 to 1.5, and the AUC 0- ⁇ geometric mean value to the control preparation The 90% confidence interval for the ratio can fall within the range of 0.8-1.25.
- the suspension of the present invention has a ratio of the Cmax geometric mean value to the control preparation and a ratio of the AUC 0- ⁇ geometric mean value to the control preparation both falling within the range of 0.8 to 1.25. obtain.
- the average Cmax is in the range of 500 to 2500 ng / mL
- the average AUC 0- ⁇ falls within the range of 1000 to 2500 h * ng / mL. More specifically, the average Cmax is in the range of 1000 to 2000 ng / mL, and the average AUC 0- ⁇ is in the range of 1500 to 2000 h * ng / mL.
- the average Cmax and the average AUC 0- ⁇ may be either an arithmetic average value or a geometric average value.
- Examples of the suspension equivalent to the above-mentioned edaravone injection include, but are not limited to, the suspensions having the following compositions.
- Edaravone particles 2.1% (w / v) Polyvinyl alcohol (dispersant): 0.1% (w / v) Xanthan gum (thickener): 0.3% (w / v) Sorbitol (sweetener): 40% (w / v) Sodium sulfite (stabilizer): 0.1% (w / v) L-Cysteine hydrochloride (stabilizer): 0.05% (w / v) Sodium hydroxide (pH adjusting agent): appropriate amount Phosphoric acid (pH adjusting agent): appropriate amount Simethicone emulsion (antifoaming agent): 0.05% (w / v) Dispersion medium: water
- the plasma concentration transition becomes the same as when
- the suspension for oral administration of the present invention can be prepared by mixing the above-mentioned edaravone particles, a dispersant and, if necessary, other components (thickener, sweetener, etc.) and water.
- the edaravone particles used preferably have a D50 particle size (volume-based cumulative 50% particle size) of 2 ⁇ m to 50 ⁇ m and a D90 particle size (volume-based cumulative 90% particle size) of 100 ⁇ m to 250 ⁇ m.
- the edaravone bulk powder obtained by the method described in JP-B-5-31523 or the like is known by a pulverizer, for example, By using a jet mill, a hammer mill, a pin mill, a ball mill or the like, edaravone particles having a desired particle diameter can be prepared.
- the particle diameter of the edaravone particles can be measured by a dry method using a laser diffraction particle size distribution device (Sympatec / HELOS & CUVETTE).
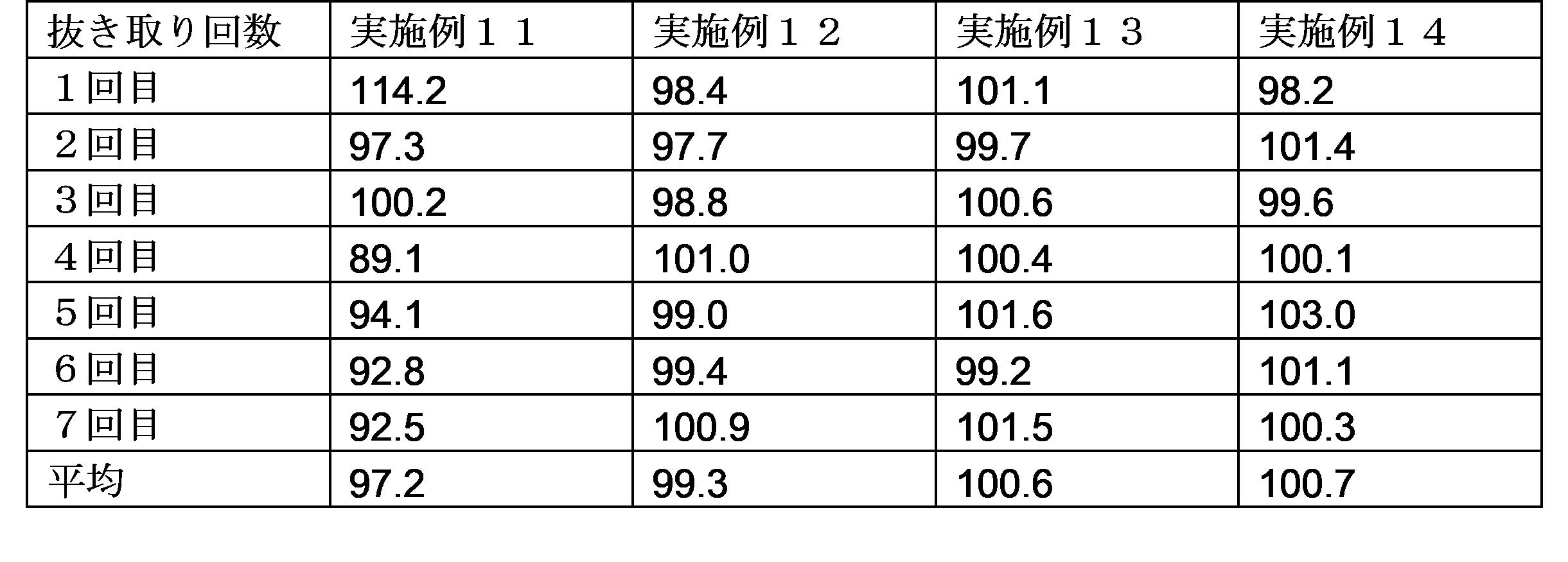
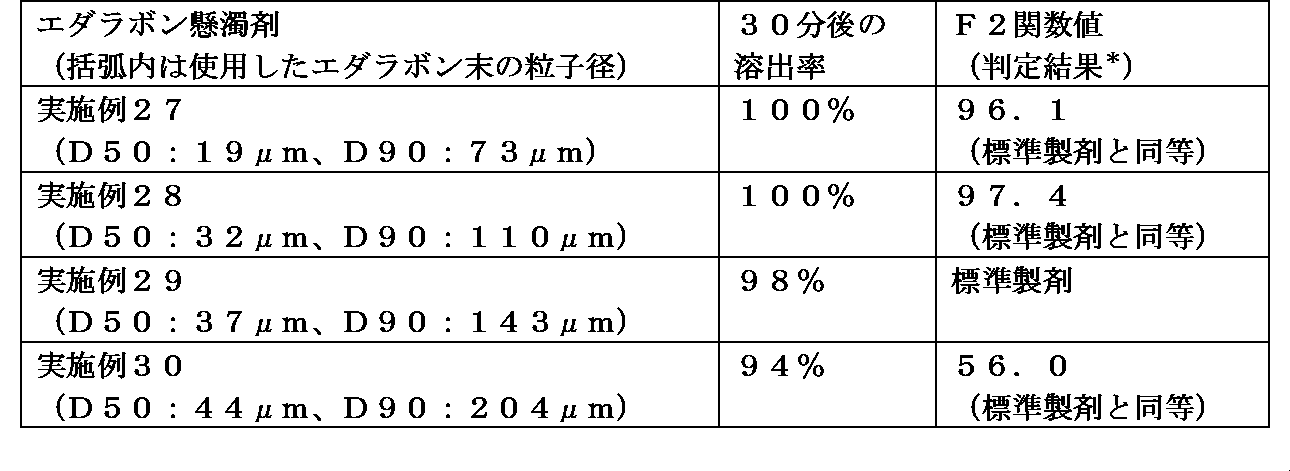
- the dissolution rate of edaravone from the obtained preparation by changing the particle size of the edaravone particles used. Specifically, the smaller the particle size, the faster the elution.
- the D50 particle size is in the range of 10 ⁇ m to 50 ⁇ m
- the D90 particle size is in the range of 50 ⁇ m to 200 ⁇ m
- the D50 particle size is Dissolution test according to the Japanese Pharmacopoeia by setting the D90 particle size within the range of 20 ⁇ m to 40 ⁇ m and 70 ⁇ m to 150 ⁇ m or less (test solution: first solution, second solution, 0.05 mol / L acetic acid / sodium acetate buffer solution)
- a paddle rotation speed 50 rpm to 75 rpm
- the suspension of the present invention can be prepared into a homogeneous suspension by mixing each component and then using a known mixing method such as stirring, shaking, or ultrasonic irradiation.
- a suitable preparation method for example, a method of uniformly dispersing (suspending) the edaravone particles in water in which a dispersant is dissolved (dispersant solution) can be mentioned. In this case, both of adding edaravone particles to the dispersant solution and adding the dispersant solution to the edaravone particles are included.
- a thickener and a sweetener may be added to the dispersant solution in advance before dispersing the edaravone particles, or may be added at the same time when the eradavone particles are dispersed. It may be added after the particles are dispersed. Further, the timing of adding these components may be different for each component.
- the suspension for oral administration of the present invention is preferably provided in a state where the edaravone particles are suspended, which requires less operation during administration, but may be provided as a ready-to-use preparation kit in which edaravone particles and water are separated. Good. Examples of such a form include a solid composition containing (A) edaravone particles, and a edaravone suspension preparation kit containing (B) a dispersant solution.
- the ready-to-use preparation kit can be expected to have storage stability for a longer period of time as compared with a prepared suspension.
- the solid composition containing (A) edaravone particles may be only edaravone particles or may be a mixture with other components.
- the edaravone particles those used for preparing the suspension agent can be used.
- the solid composition containing the edaravone particles may be granulated by a known method and finely granulated, or may be compression molded and tableted.
- the dispersant used in the present invention but also other components such as a thickener and a sweetener may be added to the (B) dispersant solution.
- a medical staff mixes (A) and (B) and shakes to obtain the suspension of the present invention.
- the suspension for oral administration of the present invention may be provided in the form of a solid composition which can be prepared just by adding water, that is, a solid composition containing edaravone particles and a dispersant.
- the present solid composition may contain other components used in the suspending agent of the present invention such as a thickener and a sweetener, and further, the present solid composition can be prepared by a known method. It may be granulated, finely divided, or granulated, or may be compression molded and tableted.
- the suspension agent of the present invention can be obtained by a medical staff mixing a solid composition containing edaravone particles and a dispersant with water and shaking.
- the suspension of the present invention is the intermittent administration method described in WO2005 / 75434 and currently used for ALS treatment using edaravone injection in clinical practice, that is, the administration period and the drug holiday period are defined as one unit. Is administered twice or more times. When the drug administration period and the drug holiday period are repeated twice or more, the final drug holiday period is always brought to the end, but it is not essential to provide the last drug holiday period. That is, for example, when the medication period and the drug holiday period are repeated twice, the “medication period, the drug holiday period, the drug period, and the drug holiday period” is obtained, but the last drug holiday period is not provided. , Dosing period ".
- the drug holiday is a period in which the drug is not administered continuously for 7 days or more, preferably 14 days.
- the dosing period can be 14 days, or 10 days out of 14 days.
- 10 days out of 14 days means any 10 days out of 14 consecutive days, and the 10 days to be administered may be 10 consecutive days or 1 to 4 days without administration 1 It may be 10 consecutive days, which are divided by more than one time period.
- the preferred period of administration can be selected while observing the patient's condition. More specifically, there is a method of providing a 14-day initial drug holiday after the 14-day initial drug period, and then repeating the 10-day drug administration period and the 14-day drug holiday period within 14 days. The number of repetitions of the 10-day administration period and the 14-day withdrawal period is not particularly limited as long as it is once or more.
- the daily dose in the intermittent dosing period can be appropriately selected according to the conditions such as the age and condition of the patient (for example, the severity of the disease), and generally, for adults, edaravone
- the dose is 60 mg to 400 mg, preferably 60 mg to 300 mg, more preferably 90 mg to 210 mg, particularly preferably 90 mg, 100 mg, 105 mg, 180 mg, 200 mg or 210 mg, further preferably 105 mg or 210 mg, most preferably Is 105 mg.
- the suspensions of the present invention may be administered to a patient on a daily or near daily basis repeatedly during the dosing period.
- the daily dose can be appropriately selected according to conditions such as the age and condition of the patient (for example, the severity of the disease), and generally, the dose of edaravone is 60 mg to 400 mg for an adult. , Preferably 60 mg to 300 mg, more preferably 90 mg to 210 mg, particularly preferably 90 mg, 100 mg, 105 mg, 180 mg, 200 mg or 210 mg, further preferably 105 mg or 210 mg, most preferably 105 mg.
- daily administration or intermittent administration there is no limitation on the number of administrations per day, and the preferable number can be selected while observing the patient's condition.
- the suspension of the present invention can contain a high content of edaravone, the dosage of edaravone can be reduced to a small dose, which is convenient for ALS patients with dysphagia.
- the suspension of the present invention can be orally administered at a dose of 1 to 20 mL per formulation, and an ALS therapeutic agent containing 50 to 210 mg of edaravone in the dose can be prepared.
- the suspension of the present invention in addition to ALS, diseases for which the involvement of oxidative stress has been reported, for example, Parkinson's disease, neurodegenerative diseases with motor dysfunction such as spinocerebellar degeneration, muscle diseases such as muscular dystrophy, It is also used for neurodegenerative diseases such as Alzheimer's disease that present cognitive impairment, vascular disorders such as cerebral infarction, systemic inflammatory diseases such as multiple sclerosis and systemic scleroderma, and local inflammatory diseases such as stomatitis. sell.
- Parkinson's disease neurodegenerative diseases with motor dysfunction such as spinocerebellar degeneration
- muscle diseases such as muscular dystrophy
- neurodegenerative diseases such as Alzheimer's disease that present cognitive impairment, vascular disorders such as cerebral infarction, systemic inflammatory diseases such as multiple sclerosis and systemic scleroderma, and local inflammatory diseases such as stomatitis. sell.
- Example 1 200 mg of polyvinyl alcohol (Nippon Gosei Kagaku, EG-05P) was dissolved in 200 mL of water to prepare a 0.1% (w / v) polyvinyl alcohol aqueous solution. 100 mg of edaravone particles (edaravone powder, D50: 37 ⁇ m, D90: 143 ⁇ m) were dispersed in 10 mL of the polyvinyl alcohol aqueous solution to obtain an edaravone suspension for oral administration.
- edaravone particles edaravone powder, D50: 37 ⁇ m, D90: 143 ⁇ m
- Example 2 200 mg of polyvinyl alcohol (Nippon Gosei Kagaku, EG-05P) was dissolved in 200 mL of water, and 1000 mg of xanthan gum (Sansei, KELTROL-CG) was further dissolved in the obtained aqueous solution of polyvinyl alcohol to give 0.1% (w / v). An aqueous polyvinyl alcohol / 0.5% (w / v) xanthan gum solution was prepared.
- edaravone particles 100 mg were dispersed in 10 mL of the polyvinyl alcohol / xanthan gum aqueous solution to obtain an edaravone suspension.
- Example 3 Polyvinyl alcohol (Nippon Gosei Kagaku, EG-05P) 200 mg was dissolved in water 200 ml, and Tragant powder (Rin powder chemical) 1000 mg was further dissolved in the obtained polyvinyl alcohol aqueous solution to obtain 0.1% (w / v) polyvinyl alcohol. /0.5% (w / v) tragacanth powder aqueous solution was prepared. 100 mg of edaravone particles (edaravone powder, D50: 37 ⁇ m, D90: 143 ⁇ m) were dispersed in 10 mL of the polyvinyl alcohol / tragacanth powder aqueous solution to obtain an edaravone suspension.
- edaravone particles edaravone powder, D50: 37 ⁇ m, D90: 143 ⁇ m
- Example 4 200 mg of methyl cellulose (Shin-Etsu Chemical, SM-25) was dissolved in 200 mL of water to prepare a 0.1% (w / v) methyl cellulose aqueous solution. 100 mg of edaravone particles (edaravone powder, D50: 37 ⁇ m, D90: 143 ⁇ m) were dispersed in 10 mL of the methylcellulose aqueous solution to obtain an edaravone suspension.
- edaravone particles edaravone powder, D50: 37 ⁇ m, D90: 143 ⁇ m
- Example 5 200 mg of methylcellulose (Shin-Etsu Chemical, SM-25) was dissolved in 200 mL of water, and 1000 mg of xanthan gum (Sansei, KELTROL-CG) was further dissolved in the obtained aqueous solution of methylcellulose to give 0.1% (w / v) methylcellulose / 0. A 0.5% (w / v) xanthan gum aqueous solution was prepared. 100 mg of edaravone particles (edaravone powder, D50: 37 ⁇ m, D90: 143 ⁇ m) were dispersed in 10 mL of the methylcellulose / xanthan gum aqueous solution to obtain an edaravone suspension.
- edaravone particles edaravone powder, D50: 37 ⁇ m, D90: 143 ⁇ m
- Example 6 200 mg of methylcellulose (Shin-Etsu Chemical, SM-25) was dissolved in 200 mL of water, and 1000 mg of tragacanth powder (Rin powder chemical) was further dissolved in the obtained aqueous solution of methylcellulose to give 0.1% (w / v) methylcellulose / 0.5. % (W / v) tragacanth powder aqueous solution was prepared. 100 mg of edaravone particles (edaravone powder, D50: 37 ⁇ m, D90: 143 ⁇ m) were dispersed in 10 mL of the methylcellulose / tragacanth powder aqueous solution to obtain an edaravone suspension.
- edaravone particles edaravone powder, D50: 37 ⁇ m, D90: 143 ⁇ m
- Example 7 200 mg of polyvinyl alcohol (Nippon Gosei Kagaku, EG-05P) was dissolved in 200 mL of water, and 1600 mg of carmellose sodium (Daicel Finechem, CMC Daicel 1150) was further dissolved in the obtained aqueous solution of polyvinyl alcohol to give 0.1% (w / v) A polyvinyl alcohol / 0.5% (w / v) aqueous solution of carmellose sodium was prepared.
- edaravone particles 100 mg were dispersed in 10 mL of the polyvinyl alcohol / carmellose sodium aqueous solution to obtain an edaravone suspension.
- Example 8 200 mg of polyvinyl alcohol (Nippon Gosei Kagaku, EG-05P) was dissolved in 200 mL of water, and 160 g of dextrin (Nippon Kagaku, Akatama No. 3) was dissolved in the obtained aqueous solution of polyvinyl alcohol to give 0.1% (w / v). ) A polyvinyl alcohol / 0.5% (w / v) dextrin aqueous solution was prepared.
- edaravone particles 100 mg were dispersed in 10 mL of the polyvinyl alcohol / dextrin aqueous solution to obtain an edaravone suspension.
- Example 9 200 mg of methyl cellulose (Shin-Etsu Chemical, SM-25) was dissolved in 200 mL of water, and 1600 mg of carmellose sodium (Daicel Finechem, CMC Daicel 1150) was further dissolved in the obtained aqueous solution of methyl cellulose to give 0.1% (w / v) methyl cellulose. /0.5% (w / v) carmellose sodium aqueous solution was prepared. 100 mg of edaravone particles (edaravone powder, D50: 37 ⁇ m, D90: 143 ⁇ m) were dispersed in 10 mL of the methylcellulose / carmellose sodium aqueous solution to obtain an edaravone suspension.
- edaravone particles edaravone powder, D50: 37 ⁇ m, D90: 143 ⁇ m
- Example 10 200 mg of methyl cellulose (SM-25, Shin-Etsu Chemical Co., Ltd.) was dissolved in 200 mL of water, and 160 g of dextrin (Nippon Kagaku, Akatama No. 3) was further dissolved in the resulting aqueous solution of methyl cellulose to give 0.1% (w / v) methyl cellulose / A 0.5% (w / v) dextrin aqueous solution was prepared. 100 mg of edaravone particles (edaravone powder, D50: 37 ⁇ m, D90: 143 ⁇ m) were dispersed in 10 mL of the methylcellulose / dextrin aqueous solution to obtain an edaravone suspension.
- edaravone particles edaravone powder, D50: 37 ⁇ m, D90: 143 ⁇ m
- Example 11 Polyvinyl alcohol (Nippon Gosei Kagaku, EG-05P) 40 mg, simethicone emulsion (Basildon, PD30S) 20 mg and edaravone particles (edaravone powder, D50: 37 ⁇ m, D90: 143 ⁇ m) 800 mg were dissolved in water to a volume of 40 mL. Dispersion was performed to obtain an edaravone suspension for oral administration.
- edaravone powder edaravone powder, D50: 37 ⁇ m, D90: 143 ⁇ m
- Example 12 40 mg of polyvinyl alcohol (Nippon Gosei Kagaku, EG-05P), 20 mg of simethicone emulsion (Basildon, PD30S), 80 mg of xanthan gum (Sansei, KELTROL-CG) and 800 mg of edaravone particles (edaravone powder, D50: 37 ⁇ m, D90: 143 ⁇ m). It was dissolved and dispersed in water to a volume of 40 mL to obtain an edaravone suspension for oral administration.
- Example 13 40 mg of polyvinyl alcohol (Nippon Gosei Kagaku, EG-05P), 20 mg of simethicone emulsion (Basildon, PD30S), 120 mg of xanthan gum (Sansei, KELTROL-CG) and 800 mg of edaravone particles (edaravone powder, D50: 37 ⁇ m, D90: 143 ⁇ m). It was dissolved and dispersed in water to a volume of 40 mL to obtain an edaravone suspension for oral administration.
- Example 14 40 mg of polyvinyl alcohol (Nippon Gosei Kagaku, EG-05P), 20 mg of simethicone emulsion (Basildon, PD30S), 200 mg of xanthan gum (Sansei, KELTROL-CG) and 800 mg of edaravone particles (edaravone powder, D50: 37 ⁇ m, D90: 143 ⁇ m). It was dissolved and dispersed in water to a volume of 40 mL to obtain an edaravone suspension for oral administration.
- Example 15 40 mg of polyvinyl alcohol (Nippon Gosei Kagaku, EG-05P), 20 mg of simethicone emulsion (Basildon, PD30S), 4 g of D-sorbitol, 40 mg of xanthan gum (Sansei, KELTROL-CG) and edaravone particles (edaravone powder, D50: 37 ⁇ m, D90). (143 ⁇ m) 800 mg was dissolved / dispersed in water to a volume of 40 mL to obtain an edaravone suspension for oral administration.
- Example 16 40 mg of polyvinyl alcohol (Nippon Gosei Kagaku, EG-05P), 20 mg of simethicone emulsion (Basildon, PD30S), 4 g of D-sorbitol, 120 mg of xanthan gum (Sansei, KELTROL-CG) and edaravone particles (edaravone powder, D50: 37 ⁇ m, D90). (143 ⁇ m) 800 mg was dissolved / dispersed in water to a volume of 40 mL to obtain an edaravone suspension for oral administration.
- Example 17 Polyvinyl alcohol (Nippon Gosei Kagaku, EG-05P) 40 mg, simethicone emulsion (Basildon, PD30S) 20 mg, D-sorbitol 4 g, xanthan gum (Sansei, KELTROL-CG) 200 mg, and edaravone particles (edaravone powder, D50: 37 ⁇ m, D90). (143 ⁇ m) 800 mg was dissolved / dispersed in water to a volume of 40 mL to obtain an edaravone suspension for oral administration.
- Example 18 Polyvinyl alcohol (Nippon Gosei Kagaku, EG-05P) 40 mg, sodium bisulfite 40 mg, L-cysteine hydrochloride hydrate 20 mg, simethicone emulsion (Basildon, PD30S) 20 mg, D-sorbitol 4 g, xanthan gum (Sansho, KELTROL-CG) ) 120 mg and 800 mg of edaravone particles (edaravone powder, D50: 37 ⁇ m, D90: 143 ⁇ m) are dissolved / dispersed in water to a volume of 40 mL, and appropriate amounts of sodium hydroxide and phosphoric acid are added to the resulting suspension. The pH was adjusted to 4.20 to obtain an edaravone suspension.
- edaravone particles edaravone powder, D50: 37 ⁇ m, D90: 143 ⁇ m
- Example 19 A preservative solution was prepared by dissolving 40 mg of benzoic acid, 2 mg of propylparaben, and 2 mg of butylparaben in 40 mg of ethanol.
- Polyvinyl alcohol (Nippon Gosei Kagaku, EG-05P) 40 mg, sodium bisulfite 40 mg, L-cysteine hydrochloride hydrate 20 mg, simethicone emulsion (Basildon, PD30S) 20 mg, D-sorbitol 4 g, xanthan gum (Sansho, KELTROL-CG) ) 120 mg and edaravone particles (edaravone powder, D50: 37 ⁇ m, D90: 143 ⁇ m) 800 mg were dissolved / dispersed in water to a volume of 40 mL, and then the preservative solution was added.
- Sodium hydroxide and phosphoric acid were added in appropriate amounts to the obtained suspension to adjust the pH to 4.20 to obtain an edaravone
- Example 20 200 mg of polyvinyl alcohol (Nippon Gosei Kagaku, EG-05P) was dissolved in 200 mL of purified water to obtain a 0.1% (w / v) polyvinyl alcohol solution. 120 mg of edaravone particles (edaravone powder, D50: 37 ⁇ m, D90: 143 ⁇ m) were dispersed in 10 mL of the polyvinyl alcohol solution to obtain an edaravone suspension.
- edaravone particles edaravone powder, D50: 37 ⁇ m, D90: 143 ⁇ m
- Example 21 300 mg of edaravone particles (edaravone powder, D50: 37 ⁇ m, D90: 143 ⁇ m) were dispersed in 10 mL of the 0.1% (w / v) polyvinyl alcohol solution obtained in Example 20 to obtain an edaravone suspension.
- Example 22 200 mg of polyvinyl alcohol (Nippon Gosei Kagaku, EG-05P) and 1000 mg of xanthan gum (Sansei, KELTROL-CG) were dissolved in 200 mL of purified water, and 0.1% (w / v) polyvinyl alcohol 0.5% (w / v) A xanthan gum solution was obtained. 300 mg of edaravone particles (edaravone powder, D50: 37 ⁇ m, D90: 143 ⁇ m) were dispersed in 10 mL of the polyvinyl alcohol / xanthan gum solution to obtain an edaravone suspension.
- edaravone particles edaravone powder, D50: 37 ⁇ m, D90: 143 ⁇ m
- Example 23 In Example 19, the edaravone suspension was obtained by using edaravone particles (edaravone powder, D50: 19 ⁇ m, D90: 73 ⁇ m) instead of the edaravone particles (edaravone powder, D50: 37 ⁇ m, D90: 143 ⁇ m).
- edaravone particles edaravone powder, D50: 19 ⁇ m, D90: 73 ⁇ m
- Example 24 In Example 19, edaravone particles (edaravone powder, D50: 37 ⁇ m, D90: 143 ⁇ m) were replaced with edaravone particles (edaravone powder, D50: 32 ⁇ m, D90: 110 ⁇ m) to obtain an edaravone suspension.
- edaravone particles edaravone powder, D50: 37 ⁇ m, D90: 143 ⁇ m
- edaravone particles edaravone powder, D50: 32 ⁇ m, D90: 110 ⁇ m
- Example 25 In Example 19, an edaravone suspension was obtained by using edaravone particles (edaravone powder, D50: 44 ⁇ m, D90: 204 ⁇ m) instead of edaravone particles (edaravone powder, D50: 37 ⁇ m, D90: 143 ⁇ m).
- edaravone particles edaravone powder, D50: 44 ⁇ m, D90: 204 ⁇ m
- edaravone particles edaravone powder, D50: 37 ⁇ m, D90: 143 ⁇ m
- Example 26 Polyvinyl alcohol (Nippon Gosei Kagaku, EG-05P) 40 mg, sodium hydrogen sulfite 40 mg, L-cysteine hydrochloride hydrate 20 mg, simethicone emulsion (Basildon, PD30S) 20 mg, D-sorbitol 16 g, xanthan gum (Sansho, KELTROL-CG) ) 120 mg and 840 mg of edaravone particles (edaravone powder, D50: 37 ⁇ m, D90: 143 ⁇ m) are dissolved / dispersed in water to a volume of 40 mL, and appropriate amounts of sodium hydroxide and phosphoric acid are added to the resulting suspension. The pH was adjusted to 4.20 to obtain an edaravone suspension.
- edaravone particles edaravone powder, D50: 37 ⁇ m, D90: 143 ⁇ m
- Example 27 an edaravone suspension was obtained by using edaravone particles (edaravone powder, D50: 17 ⁇ m, D90: 64 ⁇ m) instead of edaravone particles (edaravone powder, D50: 37 ⁇ m, D90: 143 ⁇ m).
- Example 28 In Example 26, an edaravone suspension was obtained by using edaravone particles (edaravone powder, D50: 21 ⁇ m, D90: 79 ⁇ m) instead of edaravone particles (edaravone powder, D50: 37 ⁇ m, D90: 143 ⁇ m).
- Example 29 In Example 26, an edaravone suspension was obtained by using edaravone particles (edaravone powder, D50: 31 ⁇ m, D90: 124 ⁇ m) instead of edaravone particles (edaravone powder, D50: 37 ⁇ m, D90: 143 ⁇ m).
- Example 30 In Example 26, an edaravone suspension was obtained by using edaravone particles (edaravone powder, D50: 46 ⁇ m, D90: 185 ⁇ m) instead of edaravone particles (edaravone powder, D50: 37 ⁇ m, D90: 143 ⁇ m).
- Example 31 In Example 26, xanthan gum (Sansei, KELTROL-CG) was replaced with 120 mg, and tragacanth powder (Rin powder chemical) 200 mg was used to obtain an edaravone suspension.
- Test Example 1 (Selection of dispersant) A 0.1% (w / v) aqueous solution of a formulation additive shown in the following table, which is generally known as a substance having a function of dispersing solid particles in a liquid, was prepared at room temperature. To 3600 mg of edaravone particles (edaravone powder, D50: 37 ⁇ m, D90: 143 ⁇ m), 50 mL of each of the above additive aqueous solutions was added and stirred with a stirrer. The results are shown in the table below. From the results in the above table, it was found that only polyvinyl alcohol, methyl cellulose, sucrose fatty acid ester, and hypromellose can suitably disperse the edaravone particles in water.
- Test example 2 (transmitted scattered light intensity test) A 0.1% (w / v) aqueous solution of a formulation additive shown in the following table, which is generally known as a substance having a function of dispersing solid particles in a liquid, was prepared at room temperature. To 840 mg of edaravone particles (edaravone powder, D50: 37 ⁇ m, D90: 143 ⁇ m), 40 mL of each of the above additive aqueous solutions was added and stirred with a stirrer for 30 minutes or more to confirm the dispersion state of edaravone particles.
- a formulation additive shown in the following table which is generally known as a substance having a function of dispersing solid particles in a liquid
- Test Example 3 120 mg of edaravone drug substance (D50: 37 ⁇ m, D90: 143 ⁇ m) was compression-molded with a single-shot tableting machine (compaction analyzer) (punch: diameter 8 mm flat punch, tableting pressure: 800 kgf) to obtain edaravone tablets for contact angle measurement. ..
- a saturated edaravone aqueous solution in which 0.1% (w / v) of a formulation additive shown in the following table, which is generally known as a substance having a function of dispersing solid particles in a liquid, was dissolved was prepared at room temperature, The contact angle of the edaravone tablet produced above was measured with a contact angle measuring device (Kyowa Interface Science, CAX-150). The results are shown in the table below.
- Test Example 4 (stability) 5 mL of each of the edaravone suspensions prepared in Examples 1 to 10 was placed in a glass bottle, tightly stoppered, and stored at 60 ° C. for 4 weeks. After 4 weeks, the amount of edaravone related substance in each suspension was measured according to the purity test related substance (i) described in the Japanese Pharmacopoeia edaravone injection. The results are shown in the table below. From the above results, the production of edaravone related substances was small in any of Examples 1 to 10, but in the suspensions of Examples 1 to 6 in which xanthan gum or tragacanth powder was used as the thickener, It can be seen that the generation of was small.
- Test Example 5 (content uniformity test) From each of the edaravone suspensions (40 mL) obtained in Examples 11 to 14, 5 mL of the suspension was extracted 7 times in total using a syringe, and the content of edaravone in the extracted suspension of 5 mL was measured. The results are shown below (the values in the table are relative values (%) relative to 100 mg of edaravone originally contained in 5 mL). Although the content uniformity was ensured in all of the suspensions, the suspensions of Examples 12 to 14 containing the thickener showed more preferable content uniformity.
- Test Example 6 (Redispersibility test) The suspension obtained in each of Examples 15 to 17 was put into a centrifuge, and 4000 g of gravity was applied for 6.6 hours to forcibly settle the edaravone particles. After the centrifugation, each suspension was shaken lightly by hand, and the edaravone particles were redispersed within 10 seconds for the suspensions of Examples 15 and 16 and within 40 seconds for the preparation of Example 17. The gravity condition is equivalent to the case of being stored for 3 years.
- Example 7 (preservation efficacy test) Using the suspensions obtained in Examples 18 to 19, a preservative efficacy test was conducted according to the Japanese Pharmacopoeia. Although the suspension of Example 18 contained no preservative, it exhibited a preservative effect equivalent to that of the suspension of Example 19 containing a preservative.
- Test Example 8 The edaravone suspension (10 mL) obtained in each of Examples 20, 21, and 22 was orally administered to 6 healthy adult males in a fasted state. Blood was collected before administration and 0.25, 0.5, 1, 1.5, 2, 4, 6, 8, 12, 24, 36, 48 hours after administration, and the concentration of unchanged edaravone in plasma was measured. .. The PK profile obtained is shown in the table below. Cmax of Example 20 ( edaravone 120 mg) and AUC 0-24h are Cmax (1049 ng / mL) of edaravone injection (60 mg) described in Amyotroph Lateral Scler Frontotemporal Degener. 2017; 18 (suppl 1): 80-87.
- Test Example 9 (1) (dissolution test) The suspensions prepared in Examples 19, 23, 24 and 25 were subjected to a dissolution test according to the Japanese Pharmacopoeia Dissolution Test Method 2 (paddle method).
- ⁇ Eluent Japanese Pharmacopoeia Dissolution test 1st liquid (pH 1.2) or 2nd liquid (pH 6.8), 900 ml ⁇
- the dissolution rate of edaravone 30 minutes after the start of the test and the similarity of the dissolution behavior with other examples when the preparation of Example 19 was used as the standard preparation were determined according to the guidelines for bioequivalence of generic drugs, 15, 30, The following table shows the calculation results of the F2 function when the dissolution rate comparison time was 45 minutes.
- the preparations of Examples 23 to 25 exhibited the biologically equivalent dissolution behavior as the preparation of Example 19, and the second liquid (pH 6). .8), the preparations of Examples 23 and 24 showed the biologically equivalent dissolution behavior as the preparation of Example 19.
- the dissolution rate of edaravone 30 minutes after the start of the test and the similarity of the dissolution behavior with other examples when the preparation of Example 29 was used as a standard preparation were evaluated according to the guidelines for bioequivalence of generic drugs, 15, 30, The following table shows the calculation results of the F2 function when the dissolution rate comparison time was 45 minutes.
- Test Example 10 Bioequivalence test
- the suspension obtained in Example 26 was subjected to an open-label, single-dose, randomized crossover test with 42 Japanese healthy subjects using edaravone injection as a target drug.
- the administration of the edaravone suspension in Example 26 was carried out by gently shaking the glass bottle containing the suspension, extracting 5 mL (105 mg of edaravone) with a syringe for oral administration, and orally administering this to a subject in a fasted state. I went by.
- edaravone injection was carried out by intravenously injecting 200 mL of edaravone injection (Radicut injection) (60 mg as edaravone) to a fasted subject for 1 hour.
- edaravone injection RVI injection
- PK profiles of unchanged edaravone in plasma after administration of the suspension described in Example 26 and administration of edaravone injection are shown in the following table and FIG. Further, the results of bioequivalence evaluation of the preparation of Example 26 with respect to the injection of edaravone are shown in the following table.
- the ratio of the geometric mean value of Cmax or AUC 0- ⁇ at the time of administration of the preparation of Example 26 to that at the time of administration of edaravone injection was in the range of 0.8 to 1.25 for both Cmax and AUC 0- ⁇ .
- the 90% confidence interval of the ratio of the geometric mean values is in the range of 0.8 to 1.25 for AUC 0- ⁇ and the lower limit value is in the range of 0.8 to 1.25 for Cmax.
- the upper limit value exceeded the above range.
- the present invention Since the edaravone suspension of the present invention is particularly useful as a therapeutic agent for ALS, the present invention has industrial applicability.
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Pharmacology & Pharmacy (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Medicinal Chemistry (AREA)
- Epidemiology (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Inorganic Chemistry (AREA)
- Neurology (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Organic Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Physical Education & Sports Medicine (AREA)
- Orthopedic Medicine & Surgery (AREA)
- Neurosurgery (AREA)
- Biomedical Technology (AREA)
- Dispersion Chemistry (AREA)
- Nutrition Science (AREA)
- Physiology (AREA)
- Hospice & Palliative Care (AREA)
- Pain & Pain Management (AREA)
- Psychiatry (AREA)
- Biochemistry (AREA)
- Molecular Biology (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Medicinal Preparation (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
Abstract
Description
しかしながら、直接血中に投与される注射剤と異なり、胃腸管からの吸収性、初回通過効果など種々の要因がバイオアベイラビリティに影響を与える経口投与用製剤において、注射剤と生物学的に同等な製剤を得るのは至難の業である。
経口投与用製剤としては、錠剤、カプセル剤などの固形製剤が一般的であるが、これらの製剤形態は直接服用する場合、嚥下能力の低下が予想されるALS患者には服薬が困難であり、これらの患者群に対しては、液剤、懸濁剤などの経口投与形態が望ましい。
非特許文献1~3には、エダラボンのCMC-Na懸濁液が記載されているが、これらを動物に投与したところ、いずれもバイオアベイラビリティが低かったことが記載されている。
特許文献3には、トラガントゴム水溶液を用いたエダラボン経口投与用液が記載されており、ラットにおいて十分な血中濃度が得られたことが記載されている。しかしながら、前記血中濃度は上記非特許文献1~3に記載のあるCMC-Na懸濁液の結果よりも低く、本文献記載製剤も注射剤に匹敵しうるバイオアベイラビリティを有するものではない。
懸濁液における低いバイオアベイラビリティを克服すべく、特許文献4には吸収性に優れたエダラボン溶液剤が記載されているが、本溶液剤は、エダラボンの水に対する溶解度の低さゆえに投与量が100mLにもなり、患者の服薬アドヒアランス上好ましくない。
課題を解決するための手段は以下の<1>~<23>に関するものであるが、決してこれらに限定はされない。
<2> 分散剤が、透過散乱光強度1%以上を示す分散剤である<1>の懸濁剤。
<3> 分散剤が、接触角80°以下を示す分散剤である<1>の懸濁剤。
<4> 分散剤が、ポリビニルアルコール、メチルセルロース、ヒプロメロース、ショ糖脂肪酸エステルおよびポリソルベートから選択される一種または二種である、<1>の懸濁剤。
<5> 分散剤が、ポリビニルアルコールおよびメチルセルロースから選択される一種または二種である、<4>の懸濁剤。
<6> 分散剤が、ポリビニルアルコールである、<4>または<5>の懸濁剤。
<7> ポリビニルアルコールが、ケン化度が86.5~89.0であり、かつ4%水溶液の20℃における動粘度が3mm2/s~55.7mm2/sである、<4>~<6>のいずれか1つの懸濁剤。
<8> 分散剤の配合量が0.001%(w/v)~1.0%(w/v)である、<1>~<7>のいずれか1つの懸濁剤。
<10> 増粘剤がキサンタンガムおよびトラガント末から選択される一種または二種である、<9>の懸濁剤。
<11> 増粘剤がキサンタンガムである、<9>または<10>の懸濁剤。
<12> 増粘剤の配合量が0.1%(w/v)~1.2%(w/v)である、<9>~<11>のいずれか1つの懸濁剤。
<13> 懸濁剤中におけるエダラボン粒子のD50粒子径が、10μm~100μmであり、D90粒子径が50μm~300μmである<1>~<12>のいずれかの1つの懸濁剤。
<14> エダラボン粒子の配合量が0.06%(w/v)~36%(w/v)である、<1>~<13>のいずれか1つの懸濁剤。
<15> さらに甘味剤、安定化剤およびpH調節剤から選択される一種以上の添加剤を含む、<1>~<14>のいずれか1つの懸濁剤。
<16> 懸濁剤の粘度が、50mPa・s~1750mPa・sである<1>~<15>のいずれか1つの懸濁剤。
<17> 懸濁剤の密度が、1g/mL~1.5g/mLである<1>~<16>のいずれか1つの懸濁剤。
<18> 日本薬局方に従い溶出試験(試験液:第一液、パドル回転数:50rpm)を実施した際、試験開始30分後におけるエダラボンの溶出率が80%以上である<1>~<17>のいずれかに記載の懸濁剤。
<19> (A)エダラボン粒子を含む固形組成物、および、(B)分散剤溶液
を含むヒト経口投与用エダラボン懸濁剤調製用キット。
<20> エダラボンとして90~120mgをヒトに経口投与した際、血漿中エダラボンの平均Cmaxが500~2500ng/mL、平均AUC0-∞が1000~2500h*ng/mLを示すヒト経口投与用エダラボン懸濁剤。
<21> エダラボン注射剤を対照製剤として、エダラボン90~120mgを含む懸濁剤をヒトに経口投与するクロスオーバー試験を実施した際、対照製剤に対するCmax幾何平均値の比の90%信頼区間下限値、および対照製剤に対するAUC0-∞幾何平均値の比の90%信頼区間下限値の何れもが0.8を超えるヒト経口投与用エダラボン懸濁剤。
<22> エダラボン注射剤を対照製剤として、エダラボン90~120mgを含む懸濁剤をヒトに経口投与するクロスオーバー試験を実施した際、対照製剤に対するCmax幾何平均値の比、対照製剤に対するAUC0-∞幾何平均値の比の何れもが0.8~1.25の間に入るヒト経口投与用エダラボン懸濁剤。
<23> 経口投与による一回あたりの製剤服用量が1~20mLであり、前記服用量中にエダラボンを50~210mg含有することを特徴とするALS治療剤。
さらに、本明細書において、「%(w/v)」は特に記載のない限り懸濁剤の体積に対する質量%を意味し、「~」を用いて示された数値範囲は、「~」の前後に記載される数値をそれぞれ最小値および最大値として含む範囲を示す。
これにより、嚥下障害を有する患者であっても誤嚥リスクなく飲み易くなると共にエダラボン粒子の分散状態をさらに長時間維持することが可能となる。さらに増粘剤を含有することにより、ヒトに投与した際に患者ごとの薬物血中濃度のばらつきが非常に小さくなり、より安定した薬効が期待できる。
懸濁剤中のエダラボン粒子の粒子径は、特に限定されるものではないが、懸濁剤中における安定的な分散状態の維持、速やかな体内吸収、服用時のざらつき感のなさなどの面から、D50粒子径(体積基準の累積50%粒子径)が10μm~100μmの範囲内、かつD90粒子径(体積基準の累積90%粒子径)が50μm~300μmの範囲内であることが好ましく、さらに好ましくはD50粒子径が20μm~80μmの範囲内、かつD90粒子径が100μm~250μmの範囲内である。なお、本発明において累積50%粒子径もしくは累積90%粒子径とは、それぞれ体積基準での粒子径である。
懸濁剤中におけるエダラボン粒子の粒度分布は、懸濁剤の一部を測定用の分散媒(エダラボン飽和水溶液)に分散させ、レーザー回折粒度分布装置(Sympatec/HELOS&CUVETTE)を用い、測定する。
なお、透過散乱光強度X%以上の分散剤とは、0.1%(w/v)分散剤水溶液40mLとエダラボン840mgを混合して得られた液体の液面直下の透過散乱光強度(ΔT%)を測定した際、X%以上となる分散剤を意味する。例えば、透過散乱光強度1%以上の分散剤とは、0.1%(w/v)分散剤水溶液40mLとエダラボン(D50:37μm,D90:143μm)840mgを混合し、これを30分以上スターラー撹拌して得られた液体の液面直下の透過散乱光強度(ΔT%)を測定した際、1%以上となる分散剤を意味する。さらにここで透過散乱光強度とは、上記液体20mLを円筒形のサンプルボトル(内径25mm×外径27.5mm×高さ72mm)に充填し、TURBISCAN Tower(Formulaction製)にて(25℃)、サンプルボトルの高さ39~40mmにおける透過散乱光強度の測定を開始し、開始後10分後の値を意味する。
透過散乱光強度1%以上の分散剤としては、例えば、ポリビニルアルコール、ショ糖脂肪酸エステル、ポリソルベート、メチルセルロース、ヒプロメロースが挙げられる。
使用シリンジ:ガラス、1mL
針:23ゲージ
液量:1μL
測定時間:3.1秒後
接触角80°以下の分散剤としては、例えば、ポリビニルアルコール、ショ糖脂肪酸エステル、ポリソルベート、ヒプロメロースなどがあげられる。
分散剤は、いずれかの種類を1種単独で使用してもよく、2種以上を併用してもよい。
増粘剤としては、製剤上既知の増粘剤が使用可能であり、具体的には、例えば、カルメロースナトリウム、デキストリン、トラガント末、キサンタンガムなどが挙げられ、エダラボンの保存安定性の面からトラガント末、キサンタンガムが好ましく、最も好ましくは、キサンタンガムである。
増粘剤は、いずれかの種類を1種単独で使用してもよく、2種以上を併用してもよい。
増粘剤の配合量は上記の記載に限らず、上記粘度になるよう適宜調整することができる。
また、増粘剤の配合により、患者に投与した際の患者毎のエダラボン血中濃度推移のばらつきを最小にすることができるという予想外の効果を奏するため、どの患者に対しても安定した薬効が期待できる。
甘味剤としては、例えば、糖類、人工甘味剤、非糖質系甘味剤などが挙げられ、糖類の具体例としては、例えば、マンニトール、ソルビトール、キシリトール、マルチトール、エリスリトール、スクロース、トレハロース、ラクトース、マルトース、グルコース、グリセリンなどが挙げられ、人工甘味剤の具体例としては、スクラロース、アスパルテーム、アセスルファムカリウム、サッカリンなどが挙げられ、非糖質系甘味剤の具体例としては、ソーマチン、ステビア抽出物などが挙げられる。これらのうち、ソルビトール、キシリトール、またはスクロースが好ましく、ソルビトール、またはスクロースがより好ましく、最も好ましくはソルビトールである。
甘味剤の配合量は、服用者の嗜好と粒子の沈降抑制を鑑みて適宜調整することができるが、例えば、ソルビトールの配合量としては、5%(w/v)~70%(w/v)、好ましくは10%(w/v)~60%(w/v)、より好ましくは20%(w/v)~50%(w/v)である。
香料としては、柑橘系フレーバー(オレンジ、レモン、グレープフルーツなど)、ピーチ、グレープ、バニラ、ソーダ、ベリー系フレーバー(ストロベリー、クランベリー、ブルーベリーなど)などの各種フレーバーが挙げられ、香料の好ましい配合量として、例えば、0.05%(w/v)~0.2%(w/v)が挙げられる。
消泡剤としては、シメチコンエマルジョン、脂肪酸エステル、ポリソルベート類、エタノールなどがあげられ、消泡剤の配合量として、例えば0.01%(w/v)~0.05%(w/v)が挙げられる。
(1)エダラボン粒子、ポリビニルアルコール(分散剤)、キサンタンガム(増粘剤)、ソルビトール(甘味剤)、亜硫酸水素ナトリウム(安定化剤)、L-システイン塩酸塩(安定化剤)、リン酸(pH調節剤)、水酸化ナトリウム(pH調節剤)、シメチコンエマルジョン(消泡剤)および水。
(2)エダラボン粒子、ポリビニルアルコール(分散剤)、トラガント末(増粘剤)、ソルビトール(甘味剤)、亜硫酸水素ナトリウム(安定化剤)、L-システイン塩酸塩(安定化剤)、リン酸(pH調節剤)、水酸化ナトリウム(pH調節剤)、シメチコンエマルジョン(消泡剤)および水。
(3)エダラボン粒子、ショ糖脂肪酸エステル(分散剤)、キサンタンガム(増粘剤)、スクロース(甘味剤)、亜硫酸水素ナトリウム(安定化剤)、L-システイン塩酸塩(安定化剤)、酢酸(pH調節剤)、水酸化ナトリウム(pH調節剤)、シメチコンエマルジョン(消泡剤)および水。
(4)エダラボン粒子、ポリビニルアルコール(分散剤)、キサンタンガム(増粘剤)、ソルビトール(甘味剤)、亜硫酸水素ナトリウム(安定化剤)、リン酸(pH調節剤)、水酸化ナトリウム(pH調節剤)、シメチコンエマルジョン(消泡剤)および水。
(5)エダラボン粒子、ポリビニルアルコール(分散剤)、キサンタンガム(増粘剤)、ソルビトール(甘味剤)、亜硫酸水素ナトリウム(安定化剤)、L-システイン塩酸塩(安定化剤)、リン酸(pH調節剤)、水酸化ナトリウム(pH調節剤)、シメチコンエマルジョン(消泡剤)、香料および水。
また上記試験において本発明の懸濁剤は、対照製剤に対するCmax幾何平均値の比の90%信頼区間が0.8~2.0の範囲内に入り、対照製剤に対するAUC0-∞幾何平均値の比の90%信頼区間が0.8~1.25の範囲内に入り得る。
また上記において本発明の懸濁剤は、対照製剤に対するCmax幾何平均値の比の90%信頼区間が0.8~1.5の範囲内に入り、対照製剤に対するAUC0-∞幾何平均値の比の90%信頼区間が0.8~1.25の範囲内に入り得る。
また上記において本発明の懸濁剤は、対照製剤に対するCmax幾何平均値の比、および対照製剤に対するAUC0-∞幾何平均値の比が、何れも0.8~1.25の範囲内に入り得る。
エダラボン粒子:2.1%(w/v)
ポリビニルアルコール(分散剤):0.1%(w/v)
キサンタンガム(増粘剤):0.3%(w/v)
ソルビトール(甘味剤):40%(w/v)
亜硫酸ナトリウム(安定化剤):0.1%(w/v)
L-システイン塩酸塩(安定化剤):0.05%(w/v)
水酸化ナトリウム(pH調節剤):適量
リン酸(pH調節剤):適量
シメチコンエマルジョン(消泡剤):0.05%(w/v)
分散媒:水
上記懸濁剤5mL(エダラボンとして105mg)を経口投与することにより、上記エダラボン注射剤(エダラボンとして60mg)を60分かけて静脈注射した場合と同等の血漿中濃度推移となる。
使用するエダラボン粒子は、D50粒子径(体積基準の累積50%粒子径)が2μm~50μm、かつD90粒子径(体積基準の累積90%粒子径)が100μm~250μmが好ましい。前記粒子径のエダラボン粒子を調製するためには、例えばエダラボン粒子がエダラボンのみから構成される場合、特公平5-31523号などに記載の方法により得られたエダラボン原末を公知の粉砕機、例えば、ジェットミル、ハンマーミル、ピンミル、ボールミルなどを用いることにより、所望の粒子径のエダラボン粒子を調製可能である。エダラボン粒子の粒子径の測定は、レーザー回折粒度分布装置(Sympatec/HELOS&CUVETTE)を用い、乾式方法で測定できる。
使用するエダラボン粒子の粒子径を変化させることにより、得られた製剤からのエダラボンの溶出速度を変化させることも可能である。具体的には、粒子径を小さくするほど、溶出が早くなる。例えば、エダラボン粒子が上記方法により得られたエダラボン原末の粉砕物である場合、D50粒子径を10μm~50μmの範囲内、かつD90粒子径を50μm~200μmの範囲内、好ましくはD50粒子径を20μm~40μmの範囲内、かつD90粒子径を70μm~150μm以下とすることにより、日本薬局方に従い溶出試験(試験液:第一液、第二液、0.05mol/L酢酸・酢酸ナトリウム緩衝液(pH4.0)など、パドル回転数:50rpm~75rpm)を実施した際、試験開始30分後のエダラボン溶出率が80%以上の製剤が得られるため、速やかな薬効を期待できる。
好適な調製方法としては、例えば、分散剤を溶解させた水(分散剤溶液)にエダラボン粒子を均一に分散(懸濁)させる方法が挙げられる。この場合、分散剤溶液にエダラボン粒子を投入、エダラボン粒子に分散剤溶液を添加の何れも含まれる。
増粘剤、甘味剤などのその他の成分は、エダラボン粒子を分散させる前に予め分散剤溶液に添加しておいてもよく、エラダボン粒子を分散させる際に同時に添加してもよく、さらにはエダラボン粒子を分散した後に添加してもよい。また、これら成分の添加タイミングは成分ごとに異なっていてもよい。
(B)分散剤溶液にも、本発明で使用される分散剤のみならず、増粘剤、甘味剤など他の成分が添加されていてもよい。医療従事者が(A)および(B)を混合し、振盪することで本発明の懸濁剤が得られる。
本形態についても、医療従事者がエダラボン粒子および分散剤を含む固形組成物と水を混合し、振盪することで本発明の懸濁剤が得られる。
投薬期間は、14日間、または14日間中の10日とすることができる。14日間中の10日とは、連続する14日間のうちの任意の10日という意味であり、この投薬される10日は、連続する10日間でもよいし、1日~4日間の投薬しない1回以上の期間で分断されている連続ではない10日間でもよい。投薬期間は、患者の状態を観察しながら好ましい期間を選択することができる。
より具体的には、14日間の初回投薬期間後に14日間の初回休薬期間を設け、その後、14日間中10日間の投薬期間および14日間の休薬期間を繰り返す方法があげられ、14日間中10日間の投薬期間および14日間の休薬期間を繰り返す回数は1回以上であれば特に限定されない。
毎日投与、間欠投与の何れにおいても、1日あたりの投与回数に制限はなく、患者の状態を観察しながら好ましい回数を選択することができる。しかし、患者の負担などを考慮して、3回、2回および1回が好ましく、1回がより好ましい。
また、本発明の懸濁剤であれば、高含量のエダラボンを含有しうるため、上記投与量のエダラボンを少ない製剤服用量とすることができ、嚥下障害を有するALS患者にとって好都合である。例えば本発明の懸濁剤であれば、経口投与による一回あたりの製剤服用量が1~20mLであり、前記服用量中にエダラボンを50~210mg含有するALS治療剤の調製が可能である。
ポリビニルアルコール(日本合成化学、EG-05P)200mgを水200mLに溶解し、0.1%(w/v)ポリビニルアルコール水溶液を調製した。エダラボン粒子(エダラボン末、D50:37μm,D90:143μm)100mgを前記ポリビニルアルコール水溶液10mLに分散させ、経口投与用エダラボン懸濁剤を得た。
ポリビニルアルコール(日本合成化学、EG-05P)200mgを水200mLに溶解し、得られたポリビニルアルコール水溶液にさらにキサンタンガム(三晶、KELTROL-CG)1000mgを溶解させ、0.1%(w/v)ポリビニルアルコール/0.5%(w/v)キサンタンガム水溶液を調製した。エダラボン粒子(エダラボン末、D50:37μm,D90:143μm)100mgを前記ポリビニルアルコール/キサンタンガム水溶液10mLに分散させ、エダラボン懸濁剤を得た。
ポリビニルアルコール(日本合成化学、EG-05P)200mgを水200mlに溶解し、得られたポリビニルアルコール水溶液にさらにトラガント末(鈴粉末薬品)1000mgを溶解させ、0.1%(w/v)ポリビニルアルコール/0.5%(w/v)トラガント末水溶液を調製した。エダラボン粒子(エダラボン末、D50:37μm,D90:143μm)100mgを前記ポリビニルアルコール/トラガント末水溶液10mLに分散させ、エダラボン懸濁剤を得た。
メチルセルロース(信越化学、SM-25)200mgを水200mLに溶解し、0.1%(w/v)メチルセルロース水溶液を調製した。エダラボン粒子(エダラボン末、D50:37μm,D90:143μm)100mgを前記メチルセルロース水溶液10mLに分散させ、エダラボン懸濁剤を得た。
メチルセルロース(信越化学、SM-25)200mgを水200mLに溶解し、得られたメチルセルロース水溶液にさらにキサンタンガム(三晶、KELTROL-CG)1000mgを溶解させ、0.1%(w/v)メチルセルロース/0.5%(w/v)キサンタンガム水溶液を調製した。エダラボン粒子(エダラボン末、D50:37μm,D90:143μm)100mgを前記メチルセルロース/キサンタンガム水溶液10mLに分散させ、エダラボン懸濁剤を得た。
メチルセルロース(信越化学、SM-25)200mgを水200mLに溶解し、得られたメチルセルロース水溶液にさらにトラガント末(鈴粉末薬品)1000mgを溶解させ、0.1%(w/v)メチルセルロース/0.5%(w/v)トラガント末水溶液を調製した。エダラボン粒子(エダラボン末、D50:37μm,D90:143μm)100mgを前記メチルセルロース/トラガント末水溶液10mLに分散させ、エダラボン懸濁剤を得た。
ポリビニルアルコール(日本合成化学、EG-05P)200mgを水200mLに溶解し、得られたポリビニルアルコール水溶液にさらにカルメロースナトリウム(ダイセルファインケム、CMCダイセル1150)1600mgを溶解させ、0.1%(w/v)ポリビニルアルコール/0.5%(w/v)カルメロースナトリウム水溶液を調製した。エダラボン粒子(エダラボン末、D50:37μm,D90:143μm)100mgを前記ポリビニルアルコール/カルメロースナトリウム水溶液10mLに分散させ、エダラボン懸濁剤を得た。
ポリビニルアルコール(日本合成化学、EG-05P)200mgを水200mLに溶解し、得られたポリビニルアルコール水溶液にさらにデキストリン(日澱化学、赤玉3号)160gを溶解させ、0.1%(w/v)ポリビニルアルコール/0.5%(w/v)デキストリン水溶液を調製した。エダラボン粒子(エダラボン末、D50:37μm,D90:143μm)100mgを前記ポリビニルアルコール/デキストリン水溶液10mLに分散させ、エダラボン懸濁剤を得た。
メチルセルロース(信越化学、SM-25)200mgを水200mLに溶解し、得られたメチルセルロース水溶液にさらにカルメロースナトリウム(ダイセルファインケム、CMCダイセル1150)1600mgを溶解させ、0.1%(w/v)メチルセルロース/0.5%(w/v)カルメロースナトリウム水溶液を調製した。エダラボン粒子(エダラボン末、D50:37μm,D90:143μm)100mgを前記メチルセルロース/カルメロースナトリウム水溶液10mLに分散させ、エダラボン懸濁剤を得た。
メチルセルロース(信越化学、SM-25)200mgを水200mLに溶解し、得られたメチルセルロース水溶液にさらにデキストリン(日澱化学、赤玉3号)160gを溶解させ、0.1%(w/v)メチルセルロース/0.5%(w/v)デキストリン水溶液を調製した。エダラボン粒子(エダラボン末、D50:37μm,D90:143μm)100mgを前記メチルセルロース/デキストリン水溶液10mLに分散させ、エダラボン懸濁剤を得た。
ポリビニルアルコール(日本合成化学、EG-05P)40mg、シメチコンエマルジョン(Basildon、PD30S)20mgならびにエダラボン粒子(エダラボン末、D50:37μm,D90:143μm)800mgを体積が40mLになるように水に溶解・分散させ、経口投与用エダラボン懸濁剤を得た。
ポリビニルアルコール(日本合成化学、EG-05P)40mg、シメチコンエマルジョン(Basildon、PD30S)20mg、キサンタンガム(三晶、KELTROL-CG)80mgならびにエダラボン粒子(エダラボン末、D50:37μm,D90:143μm)800mgを体積が40mLになるように水に溶解・分散させ、経口投与用エダラボン懸濁剤を得た。
ポリビニルアルコール(日本合成化学、EG-05P)40mg、シメチコンエマルジョン(Basildon、PD30S)20mg、キサンタンガム(三晶、KELTROL-CG)120mgならびにエダラボン粒子(エダラボン末、D50:37μm,D90:143μm)800mgを体積が40mLになるように水に溶解・分散させ、経口投与用エダラボン懸濁剤を得た。
ポリビニルアルコール(日本合成化学、EG-05P)40mg、シメチコンエマルジョン(Basildon、PD30S)20mg、キサンタンガム(三晶、KELTROL-CG)200mgならびにエダラボン粒子(エダラボン末、D50:37μm,D90:143μm)800mgを体積が40mLになるように水に溶解・分散させ、経口投与用エダラボン懸濁剤を得た。
ポリビニルアルコール(日本合成化学、EG-05P)40mg、シメチコンエマルジョン(Basildon、PD30S)20mg、D-ソルビトール4g、キサンタンガム(三晶、KELTROL-CG)40mgならびにエダラボン粒子(エダラボン末、D50:37μm,D90:143μm)800mgを体積が40mLになるように水に溶解・分散させ、経口投与用エダラボン懸濁剤を得た。
ポリビニルアルコール(日本合成化学、EG-05P)40mg、シメチコンエマルジョン(Basildon、PD30S)20mg、D-ソルビトール4g、キサンタンガム(三晶、KELTROL-CG)120mgならびにエダラボン粒子(エダラボン末、D50:37μm,D90:143μm)800mgを体積が40mLになるように水に溶解・分散させ、経口投与用エダラボン懸濁剤を得た。
ポリビニルアルコール(日本合成化学、EG-05P)40mg、シメチコンエマルジョン(Basildon、PD30S)20mg、D-ソルビトール4g、キサンタンガム(三晶、KELTROL-CG)200mgならびにエダラボン粒子(エダラボン末、D50:37μm,D90:143μm)800mgを体積が40mLになるように水に溶解・分散させ、経口投与用エダラボン懸濁剤を得た。
ポリビニルアルコール(日本合成化学、EG-05P)40mg、亜硫酸水素ナトリウム40mg、L-システイン塩酸塩水和物20mg、シメチコンエマルジョン(Basildon、PD30S)20mg、D-ソルビトール4g、キサンタンガム(三晶、KELTROL-CG)120mgならびにエダラボン粒子(エダラボン末、D50:37μm,D90:143μm)800mgを体積が40mLになるように水に溶解・分散させ、得られた懸濁液に水酸化ナトリウム、リン酸を適量添加してpHを4.20に調整し、エダラボン懸濁剤を得た。
安息香酸40mg、プロピルパラベン2mg、ブチルパラベン2mgをエタノール40mgに溶解し防腐剤溶液を調製した。ポリビニルアルコール(日本合成化学、EG-05P)40mg、亜硫酸水素ナトリウム40mg、L-システイン塩酸塩水和物20mg、シメチコンエマルジョン(Basildon、PD30S)20mg、D-ソルビトール4g、キサンタンガム(三晶、KELTROL-CG)120mgならびにエダラボン粒子(エダラボン末、D50:37μm,D90:143μm)800mgを体積が40mLになるように水に溶解・分散させ、その後、前期防腐剤溶液を添加した。得られた懸濁液に水酸化ナトリウム、リン酸を適量添加してpHを4.20に調整し、エダラボン懸濁剤を得た。
ポリビニルアルコール(日本合成化学、EG-05P)200mgを精製水200mLに溶解させ、0.1%(w/v)ポリビニルアルコール溶液を得た。前記ポリビニルアルコール溶液10mLにエダラボン粒子(エダラボン末、D50:37μm,D90:143μm)120mgを分散させ、エダラボン懸濁剤を得た。
実施例20で得た0.1%(w/v)ポリビニルアルコール溶液10mLにエダラボン粒子(エダラボン末、D50:37μm,D90:143μm)300mgを分散させ、エダラボン懸濁剤を得た。
ポリビニルアルコール(日本合成化学、EG-05P)200mg、キサンタンガム(三晶、KELTROL-CG)1000mgを精製水200mLに溶解させ、0.1%(w/v)ポリビニルアルコール・0.5%(w/v)キサンタンガム溶液を得た。前記ポリビニルアルコール・キサンタンガム溶液10mLにエダラボン粒子(エダラボン末、D50:37μm,D90:143μm)300mgを分散させ、エダラボン懸濁剤を得た。
実施例19において、エダラボン粒子(エダラボン末、D50:37μm,D90:143μm)に替えて、エダラボン粒子(エダラボン末、D50:19μm,D90:73μm)を用い、エダラボン懸濁剤を得た。
実施例19において、エダラボン粒子(エダラボン末、D50:37μm,D90:143μm)に替えて、エダラボン粒子(エダラボン末、D50:32μm,D90:110μm)を用い、エダラボン懸濁剤を得た。
実施例19において、エダラボン粒子(エダラボン末、D50:37μm,D90:143μm)に替えて、エダラボン粒子(エダラボン末、D50:44μm、D90:204μm)を用い、エダラボン懸濁剤を得た。
ポリビニルアルコール(日本合成化学、EG-05P)40mg、亜硫酸水素ナトリウム40mg、L-システイン塩酸塩水和物20mg、シメチコンエマルジョン(Basildon、PD30S)20mg、D-ソルビトール16g、キサンタンガム(三晶、KELTROL-CG)120mgならびにエダラボン粒子(エダラボン末、D50:37μm,D90:143μm)840mgを体積が40mLになるように水に溶解・分散させ、得られた懸濁液に水酸化ナトリウム、リン酸を適量添加してpHを4.20に調整し、エダラボン懸濁剤を得た。
実施例26において、エダラボン粒子(エダラボン末、D50:37μm,D90:143μm)に替えて、エダラボン粒子(エダラボン末、D50:17μm,D90:64μm)用い、エダラボン懸濁剤を得た。
実施例26において、エダラボン粒子(エダラボン末、D50:37μm,D90:143μm)に替えて、エダラボン粒子(エダラボン末、D50:21μm,D90:79μm)用い、エダラボン懸濁剤を得た。
実施例26において、エダラボン粒子(エダラボン末、D50:37μm,D90:143μm)に替えて、エダラボン粒子(エダラボン末、D50:31μm,D90:124μm)用い、エダラボン懸濁剤を得た。
実施例26において、エダラボン粒子(エダラボン末、D50:37μm,D90:143μm)に替えて、エダラボン粒子(エダラボン末、D50:46μm,D90:185μm)用い、エダラボン懸濁剤を得た。
実施例26において、キサンタンガム(三晶、KELTROL-CG)120mgに替えて、トラガント末(鈴粉末薬品)200mgを用い、エダラボン懸濁剤を得た。
固体粒子を液中に分散し得る機能をもった物質として一般的に知られる下記表記載の製剤添加剤の0.1%(w/v)水溶液を室温にて調製した。
エダラボン粒子(エダラボン末、D50:37μm,D90:143μm)3600mgに、上記各添加剤水溶液50mLを添加し、スターラーにて撹拌した。結果を以下の表に示す。
固体粒子を液中に分散し得る機能をもった物質として一般的に知られる下記表記載の製剤添加剤の0.1%(w/v)水溶液を室温にて調製した。
エダラボン粒子(エダラボン末、D50:37μm,D90:143μm)840mgに、上記各添加剤水溶液40mLを添加してスターラーにて30分以上撹拌し、エダラボン粒子の分散状態を確認した。
さらに、上記で得られた分散液20mLをサンプルボトル(内径25mm×外径27.5mm×高さ72mm)に充填し、TURBISCAN Tower(Formulaction製)にて、サンプルボトル高さ39~40mmの透過散乱光強度ΔT%の測定を開始し(25℃設定)、測定開始後10分後の値を各添加剤の透過散乱光強度ΔT%とした。各添加剤の透過散乱光強度ΔT%と各添加剤溶液中のエダラボン粒子の分散状態を以下の表に示す。
上記表の結果より、エダラボン粒子を好適に分散しうる分散剤は、1%以上の透過散乱光強度ΔT%を有していることがわかる。
エダラボン原薬(D50:37μm,D90:143μm)120mgを単発打錠機(コンパクションアナライザー)で圧縮成型し(杵:直径8mm平杵、打錠圧:800kgf)、接触角測定用エダラボン錠を得た。
固体粒子を液中に分散し得る機能をもった物質として一般的に知られる下記表記載の製剤添加剤を0.1%(w/v)溶解させた飽和エダラボン水溶液を室温にて調製し、接触角測定装置(協和界面科学、CAX-150)にて上記で製造したエダラボン錠に対する接触角を測定した。結果を以下の表に示した。
使用シリンジ:ガラス、1mL
針:23ゲージ
液量:1μL
測定時間:3.1秒後
上記結果および試験例1もしくは2の結果から、添加剤0.1%(w/v)を含む飽和エダラボン水溶液のエダラボン錠に対する接触角が80°以下を示す物質であれば、好適にエダラボンを水中に分散しうることがわかる。
実施例1~10にて調製したエダラボン懸濁剤各5mLをガラス瓶に入れ密栓し、60℃下にて4週間保存した。4週間後、日本薬局方エダラボン注射液に記載されている純度試験 類縁物質(i)に従い、各懸濁剤中のエダラボン類縁物質量を測定した。結果を以下の表に示す。
上記結果より、実施例1~10の何れにおいても、エダラボン類縁物質の生成は少量であったが、とりわけ増粘剤としてキサンタンガムもしくはトラガント末を使用した実施例1~6の懸濁剤において類縁物質の生成が少なかったことがわかる。
実施例11~14で得たエダラボン懸濁剤(40mL)それぞれから、シリンジを用いて懸濁剤5mLずつ計7回抜き取り、抜き取った懸濁剤5mL中のエダラボン含有量を測定した。その結果を以下に示す(表中の値は、本来5mL中に含まれるエダラボン量100mgに対する相対値(%)である)。
何れの懸濁剤においても含量均一性が確保されているが、とりわけ増粘剤を含む実施例12~14の懸濁剤の方がより好ましい含量均一性を示した。
実施例15~17で得られた懸濁剤を遠心分離機にかけ、4000gの重力を6.6時間加え、強制的にエダラボン粒子を沈降させた。遠心終了後、各懸濁剤を手で軽く振盪したところ、実施例15、16の懸濁剤は10秒以内、実施例17の製剤は40秒以内でエダラボン粒子が再分散された。なお、前記重力条件は、3年間保存した場合と等価である。
実施例18~19で得られた懸濁剤を用いて、日本薬局方に従い保存効力試験を実施した。実施例18の懸濁剤は保存剤を配合していないのにも関わらず、保存剤を配合した実施例19の懸濁剤と同等の保存効力作用を示した。
実施例20、21、22で得たエダラボン懸濁剤(10mL)を、それぞれ絶食状態の健康成人男性6人ずつに経口投与した。投与前、投与0.25、0.5、1、1.5、2、4、6、8、12,24、36、48時間後に採血を行い、血漿中におけるエダラボン未変化体濃度を測定した。得られたPKプロファイルを以下の表に示す。
実施例20(エダラボン120mg)のCmax、AUC0-24hは、Amyotroph Lateral Scler Frontotemporal Degener.2017;18(suppl 1):80-87.に記載されたエダラボン注射剤(60mg)のCmax(1049ng/mL)、AUC0-24h(1374h*ng/mL)を大きく上回り、本発明の懸濁剤が経口投与用製剤として優れたバイオアベイラビリティを有していることがわかる。
さらに、実施例21と22のPKプロファイルの比較から、増粘剤を添加することにより、被験者間のエダラボン血漿中濃度のばらつきが小さくなる、すなわち安定した薬効が期待できることがわかる。
実施例19、23、24、25にて調製した懸濁剤を、日本薬局方溶出試験法第二法(パドル法)に従い溶出試験を行った。
・溶出液:日本薬局方溶出試験第1液(pH1.2)もしくは第2液(pH6.8)、900ml
・測定方法:波長240nmにおける吸光度
・パドル回転数:50rpm
・サンプル数:n=3
試験開始30分後のエダラボンの溶出率および実施例19の製剤を標準製剤とした際の他の実施例との溶出挙動の類似性を後発医薬品の生物学的同等性ガイドラインに従い,15,30,45分を溶出率比較時点としたF2関数の計算結果を以下の表に示す。エダラボン懸濁剤を調製する際に使用したエダラボンの粒子径が小さいほど溶出が早く、エダラボン粒子の粒子径を変化させることにより、溶出速度を制御できることがわかる。一方で、F2関数の値から、第一液(pH1.2)においては、実施例23~25の製剤は実施例19の製剤と生物学的に同等の溶出挙動を示し、第二液(pH6.8)においては、実施例23、24の製剤が実施例19の製剤と生物学的に同等の溶出挙動を示した。
実施例27~30にて調製した懸濁剤を、日本薬局方溶出試験法第二法(パドル法)に従い溶出試験を行った。
・溶出液:0.05mol/L酢酸・酢酸ナトリウム緩衝液(pH4.0)、900ml
・測定方法:波長240nmにおける吸光度
・パドル回転数:50rpm
・サンプル数:n=3
試験開始30分後のエダラボンの溶出率および実施例29の製剤を標準製剤とした際の他の実施例との溶出挙動の類似性を後発医薬品の生物学的同等性ガイドラインに従い,15,30,45分を溶出率比較時点としたF2関数の計算結果を以下の表に示す。エダラボン懸濁剤を調製する際に使用したエダラボンの粒子径が小さいほど溶出が早く、エダラボン粒子の粒子径を変化させることにより、溶出速度を制御できることがわかる。一方で、F2関数の値から、実施例27、28、30の製剤は実施例29の製剤と生物学的に同等の溶出挙動を示すことわかる。
実施例26で得られた懸濁剤について、エダラボン注射剤を対象薬として、日本人健康被験者42人にて、非盲検、単回投与、無作為クロスオーバー試験を実施した。
実施例26のエダラボン懸濁剤の投与は、懸濁剤の入ったガラス瓶を軽く振った後、経口投与用シリンジにて5mL(エダラボンとして105mg)を抜き取り、これを絶食状態の被験者に経口投与することにより行った。
エダラボン注射剤の投与は、エダラボン注射剤(ラジカット注)200mL(エダラボンとして60mg)を1時間かけて絶食状態の被験者に静脈注射することにより行った。
実施例26記載懸濁剤投与時、エダラボン注射剤投与時の血漿中エダラボン未変化体のPKプロファイルを以下の表および図1に示す。
さらに、実施例26製剤のエダラボン注射剤に対する生物学的同等性評価結果を以下の表に示す。
エダラボン注射剤投与時に対する実施例26製剤投与時のCmaxもしくはAUC0-∞の幾何平均値の比は、Cmax、AUC0-∞何れも0.8~1.25の範囲内であった。その幾何平均値の比の90%信頼区間は、AUC0-∞については0.8~1.25の範囲内であり、Cmaxについては、下限値が0.8~1.25の範囲内であったが、上限値が前記範囲を超える結果となった。
Claims (23)
- エダラボン粒子、分散剤および水を含むヒト経口投与用エダラボン懸濁剤。
- 分散剤が、透過散乱光強度1%以上を示す分散剤である請求項1記載の懸濁剤。
- 分散剤が、接触角80°以下を示す分散剤である請求項1記載の懸濁剤。
- 分散剤が、ポリビニルアルコール、メチルセルロース、ヒプロメロース、ショ糖脂肪酸エステルおよびポリソルベートから選択される一種または二種である、請求項1に記載の懸濁剤。
- 分散剤が、ポリビニルアルコールおよびメチルセルロースから選択される一種または二種である、請求項4に記載の懸濁剤。
- 分散剤が、ポリビニルアルコールである、請求項4または5に記載の懸濁剤。
- ポリビニルアルコールが、ケン化度が86.5~89.0であり、かつ4%水溶液の20℃における動粘度が3mm2/s~55.7mm2/sである、請求項4~6のいずれかに記載の懸濁剤。
- 分散剤の配合量が0.001%(w/v)~1.0%(w/v)である、請求項1~7のいずれかに記載の懸濁剤。
- さらに増粘剤を含む、請求項1~8のいずれかに記載の懸濁剤。
- 増粘剤がキサンタンガムおよびトラガント末から選択される一種または二種である、請求項9に記載の懸濁剤。
- 増粘剤がキサンタンガムである、請求項9または10に記載の懸濁剤。
- 増粘剤の配合量が0.1%(w/v)~1.2%(w/v)である、請求項9~11のいずれかに記載の懸濁剤。
- 懸濁剤中におけるエダラボン粒子のD50粒子径が10μm~100μmであり、D90粒子径が50μm~300μmである、請求項1~12のいずれかに記載の懸濁剤。
- エダラボン粒子の配合量が0.06%(w/v)~36%(w/v)である、請求項1~13のいずれかに記載の懸濁剤。
- さらに甘味剤、安定化剤およびpH調節剤から選択される1種以上の添加剤を含む、請求項1~14のいずれかに記載の懸濁剤。
- 懸濁剤の粘度が、50mPa・s~1750mPa・sである請求項1~15のいずれかに記載の懸濁剤。
- 懸濁剤の密度が、1g/mL~1.5g/mLである請求項1~16のいずれかに記載の懸濁剤。
- 日本薬局方に従い溶出試験(試験液:第一液、パドル回転数:50rpm)を実施した際、試験開始30分後におけるエダラボンの溶出率が80%以上である請求項1~17のいずれかに記載の懸濁剤。
- 下記(A)および(B):
(A)エダラボン粒子を含む固形組成物
(B)分散剤溶液
を含むヒト経口投与用エダラボン懸濁剤調製用キット。 - エダラボンとして90~120mgをヒトに経口投与した際、血漿中エダラボンの平均Cmaxが500~2500ng/mL、平均AUC0-∞が1000~2500h*ng/mLを示すヒト経口投与用エダラボン懸濁剤。
- エダラボン注射剤を対照製剤として、エダラボン90~120mgを含む懸濁剤をヒトに経口投与するクロスオーバー試験を実施した際、対照製剤に対するCmax幾何平均値の比の90%信頼区間下限値、および対照製剤に対するAUC0-∞幾何平均値の比の90%信頼区間下限値の何れもが0.8を超えるヒト経口投与用エダラボン懸濁剤。
- エダラボン注射剤を対照製剤として、エダラボン90~120mgを含む懸濁剤をヒトに経口投与するクロスオーバー試験を実施した際、対照製剤に対するCmax幾何平均値の比、対照製剤に対するAUC0-∞幾何平均値の比の何れもが0.8~1.25の間に入るヒト経口投与用エダラボン懸濁剤。
- 経口投与による一回あたりの製剤服用量が1~20mLであり、前記服用量中にエダラボンを50~210mg含有することを特徴とするALS治療剤。
Priority Applications (24)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| SG11202104365QA SG11202104365QA (en) | 2018-11-02 | 2019-11-01 | Edaravone suspension for oral administration |
| CN202311552337.9A CN117398346A (zh) | 2018-11-02 | 2019-11-01 | 用于口服给药的依达拉奉混悬剂 |
| EP24181935.8A EP4420724A2 (en) | 2018-11-02 | 2019-11-01 | Edaravone suspension for oral administration |
| AU2019369843A AU2019369843A1 (en) | 2018-11-02 | 2019-11-01 | Edaravone suspension for oral administration |
| BR112021008197-6A BR112021008197A2 (pt) | 2018-11-02 | 2019-11-01 | suspensão de edaravone para administração oral |
| DK19880770.3T DK3875085T3 (da) | 2018-11-02 | 2019-11-01 | Edaravon-suspension til oral indgivelse |
| CN202311552338.3A CN117379369A (zh) | 2018-11-02 | 2019-11-01 | 依达拉奉在制备用于治疗肌萎缩侧索硬化的口服混悬剂中的用途 |
| KR1020217016462A KR20210087490A (ko) | 2018-11-02 | 2019-11-01 | 경구 투여용 에다라본 현탁액 |
| CN202311552339.8A CN117379370A (zh) | 2018-11-02 | 2019-11-01 | 用于口服给药的依达拉奉混悬剂 |
| FIEP19880770.3T FI3875085T3 (fi) | 2018-11-02 | 2019-11-01 | Edaravonisuspensio annettavaksi oraalisesti |
| JP2020554977A JP7274502B2 (ja) | 2018-11-02 | 2019-11-01 | 経口投与用エダラボン懸濁剤 |
| CN201980072370.7A CN112969459B (zh) | 2018-11-02 | 2019-11-01 | 用于口服给药的依达拉奉混悬剂 |
| EP19880770.3A EP3875085B1 (en) | 2018-11-02 | 2019-11-01 | Edaravone suspension for oral administration |
| MX2021005144A MX2021005144A (es) | 2018-11-02 | 2019-11-01 | Suspension de edaravona para administracion oral. |
| US16/872,741 US10987341B2 (en) | 2018-11-02 | 2020-05-12 | Edaravone suspension for oral administration |
| US17/213,501 US11241416B2 (en) | 2018-11-02 | 2021-03-26 | Edaravone suspension for oral administration |
| IL282773A IL282773A (en) | 2018-11-02 | 2021-04-29 | EDARAVONE SUSPENSION FOR ORAL ADMINISTRATION |
| PH12021550988A PH12021550988A1 (en) | 2018-11-02 | 2021-04-30 | Edaravone suspension for oral administration |
| US17/308,399 US11826352B2 (en) | 2018-11-02 | 2021-05-05 | Edaravone suspension for oral administration |
| US17/560,493 US11478450B2 (en) | 2018-11-02 | 2021-12-23 | Edaravone suspension for oral administration |
| US17/934,234 US11957660B2 (en) | 2018-11-02 | 2022-09-22 | Edaravone suspension for oral administration |
| JP2023075654A JP2023087011A (ja) | 2018-11-02 | 2023-05-01 | 経口投与用エダラボン懸濁剤 |
| US18/477,752 US20240033248A1 (en) | 2018-11-02 | 2023-09-29 | Edaravone suspension for oral administration |
| US18/616,227 US20240226064A1 (en) | 2018-11-02 | 2024-03-26 | Edaravone suspension for oral administration |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2018207646 | 2018-11-02 | ||
| JP2018-207646 | 2018-11-02 |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US16/872,741 Continuation US10987341B2 (en) | 2018-11-02 | 2020-05-12 | Edaravone suspension for oral administration |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2020091036A1 true WO2020091036A1 (ja) | 2020-05-07 |
Family
ID=70463399
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2019/043013 WO2020091036A1 (ja) | 2018-11-02 | 2019-11-01 | 経口投与用エダラボン懸濁剤 |
Country Status (17)
| Country | Link |
|---|---|
| US (5) | US10987341B2 (ja) |
| EP (2) | EP3875085B1 (ja) |
| JP (2) | JP7274502B2 (ja) |
| KR (1) | KR20210087490A (ja) |
| CN (4) | CN117379369A (ja) |
| AR (1) | AR116967A1 (ja) |
| AU (1) | AU2019369843A1 (ja) |
| BR (1) | BR112021008197A2 (ja) |
| DK (1) | DK3875085T3 (ja) |
| FI (1) | FI3875085T3 (ja) |
| IL (1) | IL282773A (ja) |
| MX (3) | MX2021005144A (ja) |
| PH (1) | PH12021550988A1 (ja) |
| PT (1) | PT3875085T (ja) |
| SG (1) | SG11202104365QA (ja) |
| TW (1) | TW202031256A (ja) |
| WO (1) | WO2020091036A1 (ja) |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2022102737A1 (ja) | 2020-11-12 | 2022-05-19 | 田辺三菱製薬株式会社 | エダラボン経口投与用医薬組成物およびその投与方法 |
| KR20230098267A (ko) | 2020-11-12 | 2023-07-03 | 미쓰비시 타나베 파마 코퍼레이션 | 에다라본 경구 투여용 의약 조성물 및 그의 투여 방법 |
| EP3989968A4 (en) * | 2019-07-18 | 2023-07-12 | BDR Pharmaceuticals International Private Limited | ORAL FORMULATIONS OF EDARAVON AND PROCESSES FOR THEIR PREPARATION |
| WO2023149539A1 (ja) | 2022-02-03 | 2023-08-10 | 田辺三菱製薬株式会社 | 筋萎縮性側索硬化症の治療又は進展抑制のための薬剤 |
Families Citing this family (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2019167178A1 (ja) * | 2018-02-28 | 2019-09-06 | 田辺三菱製薬株式会社 | 3-メチル-1-フェニル-2-ピラゾリン-5-オン原薬の分析方法、筋萎縮性側索硬化症の治療および筋萎縮性側索硬化症の進行抑制、ならびに、3-メチル-1-フェニル-2-ピラゾリン-5-オン原薬を含む薬剤の製造方法 |
| US11826352B2 (en) * | 2018-11-02 | 2023-11-28 | Mitsubishi Tanabe Pharma Corporation | Edaravone suspension for oral administration |
| KR20210087490A (ko) | 2018-11-02 | 2021-07-12 | 미쓰비시 타나베 파마 코퍼레이션 | 경구 투여용 에다라본 현탁액 |
| GB202103807D0 (en) | 2021-03-18 | 2021-05-05 | Johnson Matthey Plc | A crystalline salt of edaravone, processes for the preparation and use thereof |
Citations (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH0531523B2 (ja) | 1985-05-20 | 1993-05-12 | Mitsubishi Chem Ind | |
| JPH07121861B2 (ja) | 1986-11-25 | 1995-12-25 | 三菱化学株式会社 | 3−メチル−1−フエニル−2−ピラゾロン−5−オンを含有する安定な注射剤 |
| JPH1129463A (ja) * | 1997-05-14 | 1999-02-02 | Senju Pharmaceut Co Ltd | 再分散性の良い水性懸濁液剤 |
| JP2004091441A (ja) | 2002-09-04 | 2004-03-25 | Mitsubishi Pharma Corp | ピラゾロン誘導体を含有する経口投与製剤 |
| WO2005075434A1 (ja) | 2004-02-09 | 2005-08-18 | Mitsubishi Pharma Corporation | 筋萎縮性側索硬化症(als)又はalsに起因する疾患の新規治療剤 |
| JP3758164B2 (ja) | 2000-10-24 | 2006-03-22 | 三菱ウェルファーマ株式会社 | 筋萎縮性側索硬化症(als)治療剤 |
| JP2012056950A (ja) * | 2010-08-12 | 2012-03-22 | Nisshin Kasei Kk | 可溶化剤 |
| JP2014177415A (ja) * | 2013-03-13 | 2014-09-25 | Daido Kasei Kogyo Kk | ナノ粒子化製剤及びその製造方法 |
| WO2018134243A1 (en) | 2017-01-17 | 2018-07-26 | Treeway Tw001 B.V. | Treatment comprising oral or gastric administration of edaravone |
| WO2018133957A1 (en) * | 2017-01-17 | 2018-07-26 | Treeway Tw001 B.V. | Medical treatment comprising enteral administration of edaravone |
Family Cites Families (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH0531523A (ja) | 1991-07-26 | 1993-02-09 | Aichi Steel Works Ltd | ロ−ラ回転数・間隙測定可能な圧延機用ロ−ラガイド装置 |
| JPH1120463A (ja) | 1997-06-30 | 1999-01-26 | Zexel Corp | 車両用デュアルエアコン |
| WO2014134243A1 (en) | 2013-02-27 | 2014-09-04 | Regents Of The University Of Michigan | Pharmaceutical compounds and use of same in cancer and tauopathies |
| US11147811B2 (en) * | 2016-03-10 | 2021-10-19 | Sumitomo Dainippon Pharma Co., Ltd. | Composition comprising fine particle and process thereof |
| CN109125261B (zh) * | 2016-03-16 | 2021-11-30 | 苏州澳宗生物科技有限公司 | 依达拉奉剂型 |
| EP3648760A1 (en) * | 2017-07-06 | 2020-05-13 | Treeway TW001 B.V. | Use of edaravone in oral treatment of oxidative-stress mediated neurodegenerative disorders |
| KR20210087490A (ko) * | 2018-11-02 | 2021-07-12 | 미쓰비시 타나베 파마 코퍼레이션 | 경구 투여용 에다라본 현탁액 |
| MX2022000762A (es) * | 2019-07-18 | 2022-04-01 | Bdr Pharmaceuticals International Private Ltd | Formulaciones orales de edaravone y metodo de fabricacion de las mismas. |
-
2019
- 2019-11-01 KR KR1020217016462A patent/KR20210087490A/ko unknown
- 2019-11-01 WO PCT/JP2019/043013 patent/WO2020091036A1/ja active Application Filing
- 2019-11-01 CN CN202311552338.3A patent/CN117379369A/zh active Pending
- 2019-11-01 CN CN201980072370.7A patent/CN112969459B/zh active Active
- 2019-11-01 PT PT198807703T patent/PT3875085T/pt unknown
- 2019-11-01 AR ARP190103206A patent/AR116967A1/es unknown
- 2019-11-01 MX MX2021005144A patent/MX2021005144A/es unknown
- 2019-11-01 DK DK19880770.3T patent/DK3875085T3/da active
- 2019-11-01 CN CN202311552337.9A patent/CN117398346A/zh active Pending
- 2019-11-01 EP EP19880770.3A patent/EP3875085B1/en active Active
- 2019-11-01 EP EP24181935.8A patent/EP4420724A2/en active Pending
- 2019-11-01 SG SG11202104365QA patent/SG11202104365QA/en unknown
- 2019-11-01 JP JP2020554977A patent/JP7274502B2/ja active Active
- 2019-11-01 TW TW108139797A patent/TW202031256A/zh unknown
- 2019-11-01 AU AU2019369843A patent/AU2019369843A1/en active Pending
- 2019-11-01 FI FIEP19880770.3T patent/FI3875085T3/fi active
- 2019-11-01 BR BR112021008197-6A patent/BR112021008197A2/pt unknown
- 2019-11-01 CN CN202311552339.8A patent/CN117379370A/zh active Pending
-
2020
- 2020-05-12 US US16/872,741 patent/US10987341B2/en active Active
-
2021
- 2021-03-26 US US17/213,501 patent/US11241416B2/en active Active
- 2021-04-29 IL IL282773A patent/IL282773A/en unknown
- 2021-04-30 PH PH12021550988A patent/PH12021550988A1/en unknown
- 2021-04-30 MX MX2023009973A patent/MX2023009973A/es unknown
- 2021-04-30 MX MX2024000663A patent/MX2024000663A/es unknown
- 2021-12-23 US US17/560,493 patent/US11478450B2/en active Active
-
2022
- 2022-09-22 US US17/934,234 patent/US11957660B2/en active Active
-
2023
- 2023-05-01 JP JP2023075654A patent/JP2023087011A/ja active Pending
-
2024
- 2024-03-26 US US18/616,227 patent/US20240226064A1/en active Pending
Patent Citations (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH0531523B2 (ja) | 1985-05-20 | 1993-05-12 | Mitsubishi Chem Ind | |
| JPH07121861B2 (ja) | 1986-11-25 | 1995-12-25 | 三菱化学株式会社 | 3−メチル−1−フエニル−2−ピラゾロン−5−オンを含有する安定な注射剤 |
| JPH1129463A (ja) * | 1997-05-14 | 1999-02-02 | Senju Pharmaceut Co Ltd | 再分散性の良い水性懸濁液剤 |
| JP3758164B2 (ja) | 2000-10-24 | 2006-03-22 | 三菱ウェルファーマ株式会社 | 筋萎縮性側索硬化症(als)治療剤 |
| JP2004091441A (ja) | 2002-09-04 | 2004-03-25 | Mitsubishi Pharma Corp | ピラゾロン誘導体を含有する経口投与製剤 |
| WO2005075434A1 (ja) | 2004-02-09 | 2005-08-18 | Mitsubishi Pharma Corporation | 筋萎縮性側索硬化症(als)又はalsに起因する疾患の新規治療剤 |
| JP2012056950A (ja) * | 2010-08-12 | 2012-03-22 | Nisshin Kasei Kk | 可溶化剤 |
| JP2014177415A (ja) * | 2013-03-13 | 2014-09-25 | Daido Kasei Kogyo Kk | ナノ粒子化製剤及びその製造方法 |
| WO2018134243A1 (en) | 2017-01-17 | 2018-07-26 | Treeway Tw001 B.V. | Treatment comprising oral or gastric administration of edaravone |
| WO2018133957A1 (en) * | 2017-01-17 | 2018-07-26 | Treeway Tw001 B.V. | Medical treatment comprising enteral administration of edaravone |
Non-Patent Citations (6)
| Title |
|---|
| DRUG DELIVERY, vol. 24, no. 1, pages 962 - 978 |
| INTERNATIONAL JOURNAL OF PHARMACEUTICS, vol. 515, 2016, pages 490 - 500 |
| JOURNAL OF PHARMACEUTICAL SCIENCES, vol. 103, 2014, pages 730 - 742 |
| LI, HAO ET AL.: "Phase I clinical study of edaravone in healthy Chinese volunteer s", DRUGS R D, vol. 12, no. 2, 2012, pages 65 - 70, XP55703976 * |
| PARIKH, ANKIT ET AL.: "Development of a novel oral delivery system of edaravone for enhancing bioavailability", INTERNATIONAL JOURNAL OF PHARMACEUTICS, vol. 515, no. 1, 2016, pages 490 - 500, XP002770642, DOI: 10.1016/j.ijpharm.2016.10.052 * |
| PARIKH, ANKIT ET AL.: "Lipid-based nanosystem of edaravone: development, optimization, characterization and in vitro/in vivo evaluatio n", D RUG DELIV, vol. 24, no. 1, 2017, pages 962 - 978, XP55703974 * |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP3989968A4 (en) * | 2019-07-18 | 2023-07-12 | BDR Pharmaceuticals International Private Limited | ORAL FORMULATIONS OF EDARAVON AND PROCESSES FOR THEIR PREPARATION |
| WO2022102737A1 (ja) | 2020-11-12 | 2022-05-19 | 田辺三菱製薬株式会社 | エダラボン経口投与用医薬組成物およびその投与方法 |
| KR20230098267A (ko) | 2020-11-12 | 2023-07-03 | 미쓰비시 타나베 파마 코퍼레이션 | 에다라본 경구 투여용 의약 조성물 및 그의 투여 방법 |
| WO2023149539A1 (ja) | 2022-02-03 | 2023-08-10 | 田辺三菱製薬株式会社 | 筋萎縮性側索硬化症の治療又は進展抑制のための薬剤 |
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2020091036A1 (ja) | 経口投与用エダラボン懸濁剤 | |
| US8263652B2 (en) | Stabilized pediatric suspension of carisbamate | |
| JP6092629B2 (ja) | リファキシミン使用準備済懸濁液 | |
| EP2377523A1 (de) | Arzneiformen mit verbesserten pharmakokinetischen Eigenschaften | |
| US20240033248A1 (en) | Edaravone suspension for oral administration | |
| TW201244718A (en) | A pharmaceutical composition for treating a disease in the oral cavity comprising rebamipide | |
| IL184319A (en) | Pharmaceutical preparation of glycine-dexibropane syrup and processes for its preparation | |
| JP2021523202A (ja) | 経口溶液製剤 | |
| RU2810789C2 (ru) | Суспензия эдаравона для перорального введения | |
| WO2020212898A1 (en) | Pharmaceutical oral liquid solution of ivacaftor | |
| EP2926810A1 (en) | Oral liquid pharmaceutical formulations of loxoprofen | |
| US20230270722A1 (en) | Pharmaceutical composition for oral administration of edaravone and method of administering same |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 19880770 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2020554977 Country of ref document: JP Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 282773 Country of ref document: IL |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01A Ref document number: 112021008197 Country of ref document: BR |
|
| ENP | Entry into the national phase |
Ref document number: 2019369843 Country of ref document: AU Date of ref document: 20191101 Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 20217016462 Country of ref document: KR Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 2019880770 Country of ref document: EP Effective date: 20210602 |
|
| ENP | Entry into the national phase |
Ref document number: 112021008197 Country of ref document: BR Kind code of ref document: A2 Effective date: 20210428 |