WO2020071551A1 - 良性腫瘍の予防または治療薬 - Google Patents
良性腫瘍の予防または治療薬Info
- Publication number
- WO2020071551A1 WO2020071551A1 PCT/JP2019/039383 JP2019039383W WO2020071551A1 WO 2020071551 A1 WO2020071551 A1 WO 2020071551A1 JP 2019039383 W JP2019039383 W JP 2019039383W WO 2020071551 A1 WO2020071551 A1 WO 2020071551A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- peptide
- seq
- analog
- cells
- amino acid
- Prior art date
Links
- 206010028980 Neoplasm Diseases 0.000 title claims abstract description 175
- 230000000069 prophylactic effect Effects 0.000 title claims abstract description 61
- 229940043274 prophylactic drug Drugs 0.000 title abstract description 6
- 229940126585 therapeutic drug Drugs 0.000 title abstract description 6
- 108090000765 processed proteins & peptides Proteins 0.000 claims abstract description 629
- 102000040856 WT1 Human genes 0.000 claims abstract description 109
- 108700020467 WT1 Proteins 0.000 claims abstract description 109
- 238000000034 method Methods 0.000 claims abstract description 108
- 150000007523 nucleic acids Chemical class 0.000 claims abstract description 102
- 102000039446 nucleic acids Human genes 0.000 claims abstract description 100
- 108020004707 nucleic acids Proteins 0.000 claims abstract description 100
- 210000004443 dendritic cell Anatomy 0.000 claims abstract description 70
- 201000006107 Familial adenomatous polyposis Diseases 0.000 claims abstract description 69
- 208000029664 classic familial adenomatous polyposis Diseases 0.000 claims abstract description 69
- 210000002443 helper t lymphocyte Anatomy 0.000 claims abstract description 63
- 230000001939 inductive effect Effects 0.000 claims abstract description 48
- 101800000324 Immunoglobulin A1 protease translocator Proteins 0.000 claims abstract description 38
- 125000003275 alpha amino acid group Chemical group 0.000 claims description 166
- 239000003814 drug Substances 0.000 claims description 97
- 239000000203 mixture Substances 0.000 claims description 87
- 208000003200 Adenoma Diseases 0.000 claims description 77
- 229940124597 therapeutic agent Drugs 0.000 claims description 70
- 150000001413 amino acids Chemical class 0.000 claims description 68
- 238000011282 treatment Methods 0.000 claims description 60
- 210000001151 cytotoxic T lymphocyte Anatomy 0.000 claims description 55
- 150000003839 salts Chemical class 0.000 claims description 51
- 239000002671 adjuvant Substances 0.000 claims description 48
- 150000001875 compounds Chemical class 0.000 claims description 41
- 230000002265 prevention Effects 0.000 claims description 39
- 206010001233 Adenoma benign Diseases 0.000 claims description 36
- 210000003819 peripheral blood mononuclear cell Anatomy 0.000 claims description 36
- 230000003449 preventive effect Effects 0.000 claims description 20
- 239000003795 chemical substances by application Substances 0.000 claims description 19
- 238000004519 manufacturing process Methods 0.000 claims description 17
- 238000012217 deletion Methods 0.000 claims description 15
- 230000037430 deletion Effects 0.000 claims description 15
- 238000006467 substitution reaction Methods 0.000 claims description 15
- 201000002758 colorectal adenoma Diseases 0.000 claims description 13
- 108010081208 RMFPNAPYL Proteins 0.000 claims description 10
- FWMNVWWHGCHHJJ-SKKKGAJSSA-N 4-amino-1-[(2r)-6-amino-2-[[(2r)-2-[[(2r)-2-[[(2r)-2-amino-3-phenylpropanoyl]amino]-3-phenylpropanoyl]amino]-4-methylpentanoyl]amino]hexanoyl]piperidine-4-carboxylic acid Chemical compound C([C@H](C(=O)N[C@H](CC(C)C)C(=O)N[C@H](CCCCN)C(=O)N1CCC(N)(CC1)C(O)=O)NC(=O)[C@H](N)CC=1C=CC=CC=1)C1=CC=CC=C1 FWMNVWWHGCHHJJ-SKKKGAJSSA-N 0.000 claims description 9
- 206010027191 meningioma Diseases 0.000 claims description 9
- 210000000056 organ Anatomy 0.000 claims description 9
- 201000005262 Chondroma Diseases 0.000 claims description 8
- 206010024612 Lipoma Diseases 0.000 claims description 8
- 210000004556 brain Anatomy 0.000 claims description 8
- 201000011066 hemangioma Diseases 0.000 claims description 8
- 208000022669 mucinous neoplasm Diseases 0.000 claims description 8
- 208000007538 neurilemmoma Diseases 0.000 claims description 8
- 208000003154 papilloma Diseases 0.000 claims description 8
- 206010046798 Uterine leiomyoma Diseases 0.000 claims description 7
- 201000010260 leiomyoma Diseases 0.000 claims description 7
- 102000004196 processed proteins & peptides Human genes 0.000 abstract description 52
- 235000001014 amino acid Nutrition 0.000 description 69
- 229940024606 amino acid Drugs 0.000 description 68
- 235000002639 sodium chloride Nutrition 0.000 description 51
- 229940029042 WT1 peptide vaccine Drugs 0.000 description 50
- 241000699670 Mus sp. Species 0.000 description 46
- 108090000623 proteins and genes Proteins 0.000 description 46
- 229960005486 vaccine Drugs 0.000 description 40
- 102000017420 CD3 protein, epsilon/gamma/delta subunit Human genes 0.000 description 38
- 210000001744 T-lymphocyte Anatomy 0.000 description 33
- 210000004027 cell Anatomy 0.000 description 33
- 101000621309 Homo sapiens Wilms tumor protein Proteins 0.000 description 28
- 102000046004 human WT1 Human genes 0.000 description 28
- 108091033319 polynucleotide Proteins 0.000 description 27
- 102000040430 polynucleotide Human genes 0.000 description 27
- 239000002157 polynucleotide Substances 0.000 description 27
- 102000004169 proteins and genes Human genes 0.000 description 27
- 235000018102 proteins Nutrition 0.000 description 25
- 108020004414 DNA Proteins 0.000 description 24
- 108091034117 Oligonucleotide Proteins 0.000 description 23
- 230000000694 effects Effects 0.000 description 22
- 239000000427 antigen Substances 0.000 description 21
- 108091007433 antigens Proteins 0.000 description 21
- 102000036639 antigens Human genes 0.000 description 21
- 238000002474 experimental method Methods 0.000 description 21
- 108091005601 modified peptides Proteins 0.000 description 20
- 229920001184 polypeptide Polymers 0.000 description 20
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 19
- 108091032973 (ribonucleotides)n+m Proteins 0.000 description 16
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 16
- 201000011510 cancer Diseases 0.000 description 16
- 125000000539 amino acid group Chemical group 0.000 description 15
- 239000000126 substance Substances 0.000 description 15
- ZHNUHDYFZUAESO-UHFFFAOYSA-N Formamide Chemical compound NC=O ZHNUHDYFZUAESO-UHFFFAOYSA-N 0.000 description 14
- 201000010099 disease Diseases 0.000 description 14
- 230000004048 modification Effects 0.000 description 14
- 238000012986 modification Methods 0.000 description 14
- 239000000243 solution Substances 0.000 description 14
- 210000001239 CD8-positive, alpha-beta cytotoxic T lymphocyte Anatomy 0.000 description 13
- 238000002347 injection Methods 0.000 description 13
- 239000007924 injection Substances 0.000 description 13
- 210000000813 small intestine Anatomy 0.000 description 13
- 230000001225 therapeutic effect Effects 0.000 description 13
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 12
- 230000006870 function Effects 0.000 description 12
- 238000002360 preparation method Methods 0.000 description 12
- 239000003124 biologic agent Substances 0.000 description 11
- 239000002953 phosphate buffered saline Substances 0.000 description 11
- 239000004480 active ingredient Substances 0.000 description 10
- 238000009396 hybridization Methods 0.000 description 10
- 230000003053 immunization Effects 0.000 description 10
- 238000002649 immunization Methods 0.000 description 10
- 239000007788 liquid Substances 0.000 description 10
- 102220019469 rs80358836 Human genes 0.000 description 10
- 241000699666 Mus <mouse, genus> Species 0.000 description 9
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 9
- 238000004458 analytical method Methods 0.000 description 9
- 230000004071 biological effect Effects 0.000 description 9
- 239000002775 capsule Substances 0.000 description 9
- 125000003729 nucleotide group Chemical group 0.000 description 9
- 239000008194 pharmaceutical composition Substances 0.000 description 9
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 9
- 208000037062 Polyps Diseases 0.000 description 8
- 230000004913 activation Effects 0.000 description 8
- 210000000612 antigen-presenting cell Anatomy 0.000 description 8
- 238000001727 in vivo Methods 0.000 description 8
- 210000002429 large intestine Anatomy 0.000 description 8
- 239000002773 nucleotide Substances 0.000 description 8
- 239000011780 sodium chloride Substances 0.000 description 8
- 239000001509 sodium citrate Substances 0.000 description 8
- 108091054437 MHC class I family Proteins 0.000 description 7
- 238000007792 addition Methods 0.000 description 7
- 230000027455 binding Effects 0.000 description 7
- 208000015768 polyposis Diseases 0.000 description 7
- 239000000047 product Substances 0.000 description 7
- NLJMYIDDQXHKNR-UHFFFAOYSA-K sodium citrate Chemical compound O.O.[Na+].[Na+].[Na+].[O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O NLJMYIDDQXHKNR-UHFFFAOYSA-K 0.000 description 7
- 238000010186 staining Methods 0.000 description 7
- 208000024891 symptom Diseases 0.000 description 7
- 238000012360 testing method Methods 0.000 description 7
- 108091054438 MHC class II family Proteins 0.000 description 6
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 6
- 230000008827 biological function Effects 0.000 description 6
- 238000006243 chemical reaction Methods 0.000 description 6
- 238000011161 development Methods 0.000 description 6
- 230000018109 developmental process Effects 0.000 description 6
- 239000002552 dosage form Substances 0.000 description 6
- 229940079593 drug Drugs 0.000 description 6
- 239000012634 fragment Substances 0.000 description 6
- 210000004907 gland Anatomy 0.000 description 6
- 239000012188 paraffin wax Substances 0.000 description 6
- 239000007787 solid Substances 0.000 description 6
- 206010015548 Euthanasia Diseases 0.000 description 5
- CKLJMWTZIZZHCS-REOHCLBHSA-N L-aspartic acid Chemical compound OC(=O)[C@@H](N)CC(O)=O CKLJMWTZIZZHCS-REOHCLBHSA-N 0.000 description 5
- WHUUTDBJXJRKMK-VKHMYHEASA-N L-glutamic acid Chemical compound OC(=O)[C@@H](N)CCC(O)=O WHUUTDBJXJRKMK-VKHMYHEASA-N 0.000 description 5
- 102000043129 MHC class I family Human genes 0.000 description 5
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 5
- 229930006000 Sucrose Natural products 0.000 description 5
- 239000000654 additive Substances 0.000 description 5
- 235000003704 aspartic acid Nutrition 0.000 description 5
- OQFSQFPPLPISGP-UHFFFAOYSA-N beta-carboxyaspartic acid Natural products OC(=O)C(N)C(C(O)=O)C(O)=O OQFSQFPPLPISGP-UHFFFAOYSA-N 0.000 description 5
- 239000000872 buffer Substances 0.000 description 5
- 238000012258 culturing Methods 0.000 description 5
- 125000000151 cysteine group Chemical group N[C@@H](CS)C(=O)* 0.000 description 5
- OPTASPLRGRRNAP-UHFFFAOYSA-N cytosine Chemical compound NC=1C=CNC(=O)N=1 OPTASPLRGRRNAP-UHFFFAOYSA-N 0.000 description 5
- 208000035475 disorder Diseases 0.000 description 5
- 238000003780 insertion Methods 0.000 description 5
- 230000037431 insertion Effects 0.000 description 5
- 108020004999 messenger RNA Proteins 0.000 description 5
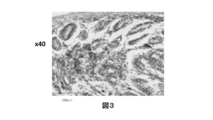
- 238000001000 micrograph Methods 0.000 description 5
- 238000007911 parenteral administration Methods 0.000 description 5
- 108010091748 peptide A Proteins 0.000 description 5
- 238000010647 peptide synthesis reaction Methods 0.000 description 5
- 239000005720 sucrose Substances 0.000 description 5
- 235000000346 sugar Nutrition 0.000 description 5
- 239000013598 vector Substances 0.000 description 5
- 102000004190 Enzymes Human genes 0.000 description 4
- 108090000790 Enzymes Proteins 0.000 description 4
- WSFSSNUMVMOOMR-UHFFFAOYSA-N Formaldehyde Chemical compound O=C WSFSSNUMVMOOMR-UHFFFAOYSA-N 0.000 description 4
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 4
- WHUUTDBJXJRKMK-UHFFFAOYSA-N Glutamic acid Natural products OC(=O)C(N)CCC(O)=O WHUUTDBJXJRKMK-UHFFFAOYSA-N 0.000 description 4
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 4
- 102100028972 HLA class I histocompatibility antigen, A alpha chain Human genes 0.000 description 4
- 108010075704 HLA-A Antigens Proteins 0.000 description 4
- WZUVPPKBWHMQCE-UHFFFAOYSA-N Haematoxylin Chemical compound C12=CC(O)=C(O)C=C2CC2(O)C1C1=CC=C(O)C(O)=C1OC2 WZUVPPKBWHMQCE-UHFFFAOYSA-N 0.000 description 4
- 229920002153 Hydroxypropyl cellulose Polymers 0.000 description 4
- 102000043131 MHC class II family Human genes 0.000 description 4
- WCUXLLCKKVVCTQ-UHFFFAOYSA-M Potassium chloride Chemical compound [Cl-].[K+] WCUXLLCKKVVCTQ-UHFFFAOYSA-M 0.000 description 4
- PYMYPHUHKUWMLA-LMVFSUKVSA-N Ribose Natural products OC[C@@H](O)[C@@H](O)[C@@H](O)C=O PYMYPHUHKUWMLA-LMVFSUKVSA-N 0.000 description 4
- 238000000692 Student's t-test Methods 0.000 description 4
- ISAKRJDGNUQOIC-UHFFFAOYSA-N Uracil Chemical compound O=C1C=CNC(=O)N1 ISAKRJDGNUQOIC-UHFFFAOYSA-N 0.000 description 4
- HMFHBZSHGGEWLO-UHFFFAOYSA-N alpha-D-Furanose-Ribose Natural products OCC1OC(O)C(O)C1O HMFHBZSHGGEWLO-UHFFFAOYSA-N 0.000 description 4
- 238000003556 assay Methods 0.000 description 4
- WQZGKKKJIJFFOK-VFUOTHLCSA-N beta-D-glucose Chemical compound OC[C@H]1O[C@@H](O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-VFUOTHLCSA-N 0.000 description 4
- 229940022399 cancer vaccine Drugs 0.000 description 4
- 238000009566 cancer vaccine Methods 0.000 description 4
- 239000011248 coating agent Substances 0.000 description 4
- 238000010586 diagram Methods 0.000 description 4
- 239000000539 dimer Substances 0.000 description 4
- 239000008103 glucose Substances 0.000 description 4
- 235000013922 glutamic acid Nutrition 0.000 description 4
- 239000004220 glutamic acid Substances 0.000 description 4
- 239000001863 hydroxypropyl cellulose Substances 0.000 description 4
- 235000010977 hydroxypropyl cellulose Nutrition 0.000 description 4
- 238000000338 in vitro Methods 0.000 description 4
- 230000003211 malignant effect Effects 0.000 description 4
- -1 patches Substances 0.000 description 4
- 229920000642 polymer Polymers 0.000 description 4
- 238000002271 resection Methods 0.000 description 4
- 239000006188 syrup Substances 0.000 description 4
- 235000020357 syrup Nutrition 0.000 description 4
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 3
- GUBGYTABKSRVRQ-XLOQQCSPSA-N Alpha-Lactose Chemical compound O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@H]1O[C@@H]1[C@@H](CO)O[C@H](O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-XLOQQCSPSA-N 0.000 description 3
- WVDDGKGOMKODPV-UHFFFAOYSA-N Benzyl alcohol Chemical compound OCC1=CC=CC=C1 WVDDGKGOMKODPV-UHFFFAOYSA-N 0.000 description 3
- 108020004705 Codon Proteins 0.000 description 3
- FBPFZTCFMRRESA-FSIIMWSLSA-N D-Glucitol Natural products OC[C@H](O)[C@H](O)[C@@H](O)[C@H](O)CO FBPFZTCFMRRESA-FSIIMWSLSA-N 0.000 description 3
- FBPFZTCFMRRESA-KVTDHHQDSA-N D-Mannitol Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-KVTDHHQDSA-N 0.000 description 3
- FBPFZTCFMRRESA-JGWLITMVSA-N D-glucitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-JGWLITMVSA-N 0.000 description 3
- HMFHBZSHGGEWLO-SOOFDHNKSA-N D-ribofuranose Chemical compound OC[C@H]1OC(O)[C@H](O)[C@@H]1O HMFHBZSHGGEWLO-SOOFDHNKSA-N 0.000 description 3
- DCXYFEDJOCDNAF-REOHCLBHSA-N L-asparagine Chemical compound OC(=O)[C@@H](N)CC(N)=O DCXYFEDJOCDNAF-REOHCLBHSA-N 0.000 description 3
- ZDXPYRJPNDTMRX-VKHMYHEASA-N L-glutamine Chemical compound OC(=O)[C@@H](N)CCC(N)=O ZDXPYRJPNDTMRX-VKHMYHEASA-N 0.000 description 3
- AGPKZVBTJJNPAG-WHFBIAKZSA-N L-isoleucine Chemical compound CC[C@H](C)[C@H](N)C(O)=O AGPKZVBTJJNPAG-WHFBIAKZSA-N 0.000 description 3
- KDXKERNSBIXSRK-YFKPBYRVSA-N L-lysine Chemical compound NCCCC[C@H](N)C(O)=O KDXKERNSBIXSRK-YFKPBYRVSA-N 0.000 description 3
- FFEARJCKVFRZRR-BYPYZUCNSA-N L-methionine Chemical compound CSCC[C@H](N)C(O)=O FFEARJCKVFRZRR-BYPYZUCNSA-N 0.000 description 3
- COLNVLDHVKWLRT-QMMMGPOBSA-N L-phenylalanine Chemical compound OC(=O)[C@@H](N)CC1=CC=CC=C1 COLNVLDHVKWLRT-QMMMGPOBSA-N 0.000 description 3
- KZSNJWFQEVHDMF-BYPYZUCNSA-N L-valine Chemical compound CC(C)[C@H](N)C(O)=O KZSNJWFQEVHDMF-BYPYZUCNSA-N 0.000 description 3
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 description 3
- KDXKERNSBIXSRK-UHFFFAOYSA-N Lysine Natural products NCCCCC(N)C(O)=O KDXKERNSBIXSRK-UHFFFAOYSA-N 0.000 description 3
- 229930195725 Mannitol Natural products 0.000 description 3
- 108091028043 Nucleic acid sequence Proteins 0.000 description 3
- 101800005149 Peptide B Proteins 0.000 description 3
- DBMJMQXJHONAFJ-UHFFFAOYSA-M Sodium laurylsulphate Chemical compound [Na+].CCCCCCCCCCCCOS([O-])(=O)=O DBMJMQXJHONAFJ-UHFFFAOYSA-M 0.000 description 3
- KZSNJWFQEVHDMF-UHFFFAOYSA-N Valine Natural products CC(C)C(N)C(O)=O KZSNJWFQEVHDMF-UHFFFAOYSA-N 0.000 description 3
- 230000003213 activating effect Effects 0.000 description 3
- WNROFYMDJYEPJX-UHFFFAOYSA-K aluminium hydroxide Chemical compound [OH-].[OH-].[OH-].[Al+3] WNROFYMDJYEPJX-UHFFFAOYSA-K 0.000 description 3
- 210000004899 c-terminal region Anatomy 0.000 description 3
- 230000001472 cytotoxic effect Effects 0.000 description 3
- 238000005516 engineering process Methods 0.000 description 3
- 210000001035 gastrointestinal tract Anatomy 0.000 description 3
- ZDXPYRJPNDTMRX-UHFFFAOYSA-N glutamine Natural products OC(=O)C(N)CCC(N)=O ZDXPYRJPNDTMRX-UHFFFAOYSA-N 0.000 description 3
- 235000004554 glutamine Nutrition 0.000 description 3
- 238000005469 granulation Methods 0.000 description 3
- 230000003179 granulation Effects 0.000 description 3
- 230000028993 immune response Effects 0.000 description 3
- 230000036039 immunity Effects 0.000 description 3
- 230000000968 intestinal effect Effects 0.000 description 3
- 210000000936 intestine Anatomy 0.000 description 3
- AGPKZVBTJJNPAG-UHFFFAOYSA-N isoleucine Natural products CCC(C)C(N)C(O)=O AGPKZVBTJJNPAG-UHFFFAOYSA-N 0.000 description 3
- 229960000310 isoleucine Drugs 0.000 description 3
- 235000014705 isoleucine Nutrition 0.000 description 3
- 239000008101 lactose Substances 0.000 description 3
- 239000000594 mannitol Substances 0.000 description 3
- 235000010355 mannitol Nutrition 0.000 description 3
- 229930182817 methionine Natural products 0.000 description 3
- 235000006109 methionine Nutrition 0.000 description 3
- 238000002156 mixing Methods 0.000 description 3
- 238000010369 molecular cloning Methods 0.000 description 3
- 210000001616 monocyte Anatomy 0.000 description 3
- 210000005087 mononuclear cell Anatomy 0.000 description 3
- 239000003921 oil Substances 0.000 description 3
- 235000019198 oils Nutrition 0.000 description 3
- 102000013415 peroxidase activity proteins Human genes 0.000 description 3
- 108040007629 peroxidase activity proteins Proteins 0.000 description 3
- 239000000546 pharmaceutical excipient Substances 0.000 description 3
- 239000008363 phosphate buffer Substances 0.000 description 3
- 102000054765 polymorphisms of proteins Human genes 0.000 description 3
- 239000001267 polyvinylpyrrolidone Substances 0.000 description 3
- 229920000036 polyvinylpyrrolidone Polymers 0.000 description 3
- 235000013855 polyvinylpyrrolidone Nutrition 0.000 description 3
- 239000002243 precursor Substances 0.000 description 3
- 239000003755 preservative agent Substances 0.000 description 3
- 239000000523 sample Substances 0.000 description 3
- 210000002966 serum Anatomy 0.000 description 3
- 238000002741 site-directed mutagenesis Methods 0.000 description 3
- 239000002904 solvent Substances 0.000 description 3
- 239000000600 sorbitol Substances 0.000 description 3
- 235000010356 sorbitol Nutrition 0.000 description 3
- 230000004936 stimulating effect Effects 0.000 description 3
- 238000007920 subcutaneous administration Methods 0.000 description 3
- 239000003826 tablet Substances 0.000 description 3
- 239000004474 valine Substances 0.000 description 3
- 235000014393 valine Nutrition 0.000 description 3
- MTCFGRXMJLQNBG-REOHCLBHSA-N (2S)-2-Amino-3-hydroxypropansäure Chemical compound OC[C@H](N)C(O)=O MTCFGRXMJLQNBG-REOHCLBHSA-N 0.000 description 2
- PJVXUVWGSCCGHT-ZPYZYFCMSA-N (2r,3s,4r,5r)-2,3,4,5,6-pentahydroxyhexanal;(3s,4r,5r)-1,3,4,5,6-pentahydroxyhexan-2-one Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@@H](O)C=O.OC[C@@H](O)[C@@H](O)[C@H](O)C(=O)CO PJVXUVWGSCCGHT-ZPYZYFCMSA-N 0.000 description 2
- OWEGMIWEEQEYGQ-UHFFFAOYSA-N 100676-05-9 Natural products OC1C(O)C(O)C(CO)OC1OCC1C(O)C(O)C(O)C(OC2C(OC(O)C(O)C2O)CO)O1 OWEGMIWEEQEYGQ-UHFFFAOYSA-N 0.000 description 2
- YXHLJMWYDTXDHS-IRFLANFNSA-N 7-aminoactinomycin D Chemical compound C[C@H]1OC(=O)[C@H](C(C)C)N(C)C(=O)CN(C)C(=O)[C@@H]2CCCN2C(=O)[C@@H](C(C)C)NC(=O)[C@H]1NC(=O)C1=C(N)C(=O)C(C)=C2OC(C(C)=C(N)C=C3C(=O)N[C@@H]4C(=O)N[C@@H](C(N5CCC[C@H]5C(=O)N(C)CC(=O)N(C)[C@@H](C(C)C)C(=O)O[C@@H]4C)=O)C(C)C)=C3N=C21 YXHLJMWYDTXDHS-IRFLANFNSA-N 0.000 description 2
- 108700012813 7-aminoactinomycin D Proteins 0.000 description 2
- 108700001666 APC Genes Proteins 0.000 description 2
- CIWBSHSKHKDKBQ-JLAZNSOCSA-N Ascorbic acid Chemical compound OC[C@H](O)[C@H]1OC(=O)C(O)=C1O CIWBSHSKHKDKBQ-JLAZNSOCSA-N 0.000 description 2
- DCXYFEDJOCDNAF-UHFFFAOYSA-N Asparagine Natural products OC(=O)C(N)CC(N)=O DCXYFEDJOCDNAF-UHFFFAOYSA-N 0.000 description 2
- BSYNRYMUTXBXSQ-UHFFFAOYSA-N Aspirin Chemical compound CC(=O)OC1=CC=CC=C1C(O)=O BSYNRYMUTXBXSQ-UHFFFAOYSA-N 0.000 description 2
- 108091003079 Bovine Serum Albumin Proteins 0.000 description 2
- 125000001433 C-terminal amino-acid group Chemical group 0.000 description 2
- 241000283707 Capra Species 0.000 description 2
- 101800001312 Capsid protein VP1 Proteins 0.000 description 2
- 101800001318 Capsid protein VP4 Proteins 0.000 description 2
- 206010009944 Colon cancer Diseases 0.000 description 2
- 208000001333 Colorectal Neoplasms Diseases 0.000 description 2
- 229920002261 Corn starch Polymers 0.000 description 2
- SNHRIJBANHPWMO-XGEHTFHBSA-N Cys-Met-Thr Chemical compound C[C@H]([C@@H](C(=O)O)NC(=O)[C@H](CCSC)NC(=O)[C@H](CS)N)O SNHRIJBANHPWMO-XGEHTFHBSA-N 0.000 description 2
- 230000006820 DNA synthesis Effects 0.000 description 2
- KCXVZYZYPLLWCC-UHFFFAOYSA-N EDTA Chemical compound OC(=O)CN(CC(O)=O)CCN(CC(O)=O)CC(O)=O KCXVZYZYPLLWCC-UHFFFAOYSA-N 0.000 description 2
- ZGTMUACCHSMWAC-UHFFFAOYSA-L EDTA disodium salt (anhydrous) Chemical compound [Na+].[Na+].OC(=O)CN(CC([O-])=O)CCN(CC(O)=O)CC([O-])=O ZGTMUACCHSMWAC-UHFFFAOYSA-L 0.000 description 2
- 229930091371 Fructose Natural products 0.000 description 2
- RFSUNEUAIZKAJO-ARQDHWQXSA-N Fructose Chemical compound OC[C@H]1O[C@](O)(CO)[C@@H](O)[C@@H]1O RFSUNEUAIZKAJO-ARQDHWQXSA-N 0.000 description 2
- 239000005715 Fructose Substances 0.000 description 2
- 108010010803 Gelatin Proteins 0.000 description 2
- DHMQDGOQFOQNFH-UHFFFAOYSA-N Glycine Chemical compound NCC(O)=O DHMQDGOQFOQNFH-UHFFFAOYSA-N 0.000 description 2
- 102100028976 HLA class I histocompatibility antigen, B alpha chain Human genes 0.000 description 2
- 102100028971 HLA class I histocompatibility antigen, C alpha chain Human genes 0.000 description 2
- 102100036242 HLA class II histocompatibility antigen, DQ alpha 2 chain Human genes 0.000 description 2
- 108010088729 HLA-A*02:01 antigen Proteins 0.000 description 2
- 108010013476 HLA-A24 Antigen Proteins 0.000 description 2
- 108010058607 HLA-B Antigens Proteins 0.000 description 2
- 108010058597 HLA-DR Antigens Proteins 0.000 description 2
- 102000006354 HLA-DR Antigens Human genes 0.000 description 2
- 102000008949 Histocompatibility Antigens Class I Human genes 0.000 description 2
- 102000018713 Histocompatibility Antigens Class II Human genes 0.000 description 2
- 241000282412 Homo Species 0.000 description 2
- 101000986084 Homo sapiens HLA class I histocompatibility antigen, C alpha chain Proteins 0.000 description 2
- 101000930801 Homo sapiens HLA class II histocompatibility antigen, DQ alpha 2 chain Proteins 0.000 description 2
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 2
- 208000026350 Inborn Genetic disease Diseases 0.000 description 2
- XUJNEKJLAYXESH-REOHCLBHSA-N L-Cysteine Chemical compound SC[C@H](N)C(O)=O XUJNEKJLAYXESH-REOHCLBHSA-N 0.000 description 2
- 102000003855 L-lactate dehydrogenase Human genes 0.000 description 2
- 108700023483 L-lactate dehydrogenases Proteins 0.000 description 2
- ROHFNLRQFUQHCH-YFKPBYRVSA-N L-leucine Chemical compound CC(C)C[C@H](N)C(O)=O ROHFNLRQFUQHCH-YFKPBYRVSA-N 0.000 description 2
- AYFVYJQAPQTCCC-GBXIJSLDSA-N L-threonine Chemical compound C[C@@H](O)[C@H](N)C(O)=O AYFVYJQAPQTCCC-GBXIJSLDSA-N 0.000 description 2
- QIVBCDIJIAJPQS-VIFPVBQESA-N L-tryptophane Chemical compound C1=CC=C2C(C[C@H](N)C(O)=O)=CNC2=C1 QIVBCDIJIAJPQS-VIFPVBQESA-N 0.000 description 2
- 239000004166 Lanolin Substances 0.000 description 2
- ROHFNLRQFUQHCH-UHFFFAOYSA-N Leucine Natural products CC(C)CC(N)C(O)=O ROHFNLRQFUQHCH-UHFFFAOYSA-N 0.000 description 2
- 239000004472 Lysine Substances 0.000 description 2
- GUBGYTABKSRVRQ-PICCSMPSSA-N Maltose Natural products O[C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@@H]1O[C@@H]1[C@@H](CO)OC(O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-PICCSMPSSA-N 0.000 description 2
- YNOVBMBQSQTLFM-DCAQKATOSA-N Met-Asn-Leu Chemical compound [H]N[C@@H](CCSC)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CC(C)C)C(O)=O YNOVBMBQSQTLFM-DCAQKATOSA-N 0.000 description 2
- 241001465754 Metazoa Species 0.000 description 2
- 108091005461 Nucleic proteins Proteins 0.000 description 2
- CTQNGGLPUBDAKN-UHFFFAOYSA-N O-Xylene Chemical compound CC1=CC=CC=C1C CTQNGGLPUBDAKN-UHFFFAOYSA-N 0.000 description 2
- 241000283973 Oryctolagus cuniculus Species 0.000 description 2
- 229930040373 Paraformaldehyde Natural products 0.000 description 2
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 2
- 239000002202 Polyethylene glycol Substances 0.000 description 2
- 229920001213 Polysorbate 20 Polymers 0.000 description 2
- 239000004372 Polyvinyl alcohol Substances 0.000 description 2
- 101800001494 Protease 2A Proteins 0.000 description 2
- 101800001066 Protein 2A Proteins 0.000 description 2
- 101800001090 Protein 3A Proteins 0.000 description 2
- 230000006819 RNA synthesis Effects 0.000 description 2
- 102220532739 SPARC-like protein 1_P9A_mutation Human genes 0.000 description 2
- MTCFGRXMJLQNBG-UHFFFAOYSA-N Serine Natural products OCC(N)C(O)=O MTCFGRXMJLQNBG-UHFFFAOYSA-N 0.000 description 2
- 229920002125 Sokalan® Polymers 0.000 description 2
- RAHZWNYVWXNFOC-UHFFFAOYSA-N Sulphur dioxide Chemical compound O=S=O RAHZWNYVWXNFOC-UHFFFAOYSA-N 0.000 description 2
- BJJRNAVDQGREGC-HOUAVDHOSA-N Thr-Trp-Asn Chemical compound C[C@H]([C@@H](C(=O)N[C@@H](CC1=CNC2=CC=CC=C21)C(=O)N[C@@H](CC(=O)N)C(=O)O)N)O BJJRNAVDQGREGC-HOUAVDHOSA-N 0.000 description 2
- AYFVYJQAPQTCCC-UHFFFAOYSA-N Threonine Natural products CC(O)C(N)C(O)=O AYFVYJQAPQTCCC-UHFFFAOYSA-N 0.000 description 2
- 239000004473 Threonine Substances 0.000 description 2
- 239000007983 Tris buffer Substances 0.000 description 2
- NAQBQJOGGYGCOT-QEJZJMRPSA-N Trp-Asn-Gln Chemical compound [H]N[C@@H](CC1=CNC2=C1C=CC=C2)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CCC(N)=O)C(O)=O NAQBQJOGGYGCOT-QEJZJMRPSA-N 0.000 description 2
- QIVBCDIJIAJPQS-UHFFFAOYSA-N Tryptophan Natural products C1=CC=C2C(CC(N)C(O)=O)=CNC2=C1 QIVBCDIJIAJPQS-UHFFFAOYSA-N 0.000 description 2
- 108700025716 Tumor Suppressor Genes Proteins 0.000 description 2
- 102000044209 Tumor Suppressor Genes Human genes 0.000 description 2
- 208000008383 Wilms tumor Diseases 0.000 description 2
- 230000021736 acetylation Effects 0.000 description 2
- 238000006640 acetylation reaction Methods 0.000 description 2
- 229960001138 acetylsalicylic acid Drugs 0.000 description 2
- 230000009471 action Effects 0.000 description 2
- 230000000996 additive effect Effects 0.000 description 2
- 208000025768 adenoma of small intestine Diseases 0.000 description 2
- 229940037003 alum Drugs 0.000 description 2
- ILRRQNADMUWWFW-UHFFFAOYSA-K aluminium phosphate Chemical compound O1[Al]2OP1(=O)O2 ILRRQNADMUWWFW-UHFFFAOYSA-K 0.000 description 2
- 125000003277 amino group Chemical group 0.000 description 2
- 235000009582 asparagine Nutrition 0.000 description 2
- 229960001230 asparagine Drugs 0.000 description 2
- 210000003719 b-lymphocyte Anatomy 0.000 description 2
- SESFRYSPDFLNCH-UHFFFAOYSA-N benzyl benzoate Chemical compound C=1C=CC=CC=1C(=O)OCC1=CC=CC=C1 SESFRYSPDFLNCH-UHFFFAOYSA-N 0.000 description 2
- 230000031018 biological processes and functions Effects 0.000 description 2
- 210000000601 blood cell Anatomy 0.000 description 2
- 229910021538 borax Inorganic materials 0.000 description 2
- KGBXLFKZBHKPEV-UHFFFAOYSA-N boric acid Chemical compound OB(O)O KGBXLFKZBHKPEV-UHFFFAOYSA-N 0.000 description 2
- 239000004327 boric acid Substances 0.000 description 2
- 235000010338 boric acid Nutrition 0.000 description 2
- 229940098773 bovine serum albumin Drugs 0.000 description 2
- 210000000133 brain stem Anatomy 0.000 description 2
- 239000001506 calcium phosphate Substances 0.000 description 2
- 229910000389 calcium phosphate Inorganic materials 0.000 description 2
- 235000011010 calcium phosphates Nutrition 0.000 description 2
- 125000002915 carbonyl group Chemical group [*:2]C([*:1])=O 0.000 description 2
- 239000000969 carrier Substances 0.000 description 2
- 230000008859 change Effects 0.000 description 2
- 239000003153 chemical reaction reagent Substances 0.000 description 2
- OSASVXMJTNOKOY-UHFFFAOYSA-N chlorobutanol Chemical compound CC(C)(O)C(Cl)(Cl)Cl OSASVXMJTNOKOY-UHFFFAOYSA-N 0.000 description 2
- 239000007979 citrate buffer Substances 0.000 description 2
- 210000001072 colon Anatomy 0.000 description 2
- 238000007906 compression Methods 0.000 description 2
- 230000006835 compression Effects 0.000 description 2
- 238000007796 conventional method Methods 0.000 description 2
- 239000008120 corn starch Substances 0.000 description 2
- 239000006071 cream Substances 0.000 description 2
- 230000009089 cytolysis Effects 0.000 description 2
- 229940104302 cytosine Drugs 0.000 description 2
- 230000001934 delay Effects 0.000 description 2
- 239000003085 diluting agent Substances 0.000 description 2
- 238000012143 endoscopic resection Methods 0.000 description 2
- 210000003743 erythrocyte Anatomy 0.000 description 2
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 2
- 238000011156 evaluation Methods 0.000 description 2
- 239000013604 expression vector Substances 0.000 description 2
- 238000011049 filling Methods 0.000 description 2
- 238000000684 flow cytometry Methods 0.000 description 2
- 235000003599 food sweetener Nutrition 0.000 description 2
- 238000009472 formulation Methods 0.000 description 2
- 239000008273 gelatin Substances 0.000 description 2
- 229920000159 gelatin Polymers 0.000 description 2
- 235000019322 gelatine Nutrition 0.000 description 2
- 235000011852 gelatine desserts Nutrition 0.000 description 2
- 235000001727 glucose Nutrition 0.000 description 2
- 150000004676 glycans Chemical class 0.000 description 2
- 235000011187 glycerol Nutrition 0.000 description 2
- 239000008187 granular material Substances 0.000 description 2
- 229910052739 hydrogen Inorganic materials 0.000 description 2
- 125000001165 hydrophobic group Chemical group 0.000 description 2
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 2
- 230000001900 immune effect Effects 0.000 description 2
- 230000006698 induction Effects 0.000 description 2
- 239000000543 intermediate Substances 0.000 description 2
- 229940039717 lanolin Drugs 0.000 description 2
- 235000019388 lanolin Nutrition 0.000 description 2
- 239000002502 liposome Substances 0.000 description 2
- 229940057995 liquid paraffin Drugs 0.000 description 2
- 210000004698 lymphocyte Anatomy 0.000 description 2
- HQKMJHAJHXVSDF-UHFFFAOYSA-L magnesium stearate Chemical compound [Mg+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O HQKMJHAJHXVSDF-UHFFFAOYSA-L 0.000 description 2
- 239000000463 material Substances 0.000 description 2
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 2
- CXKWCBBOMKCUKX-UHFFFAOYSA-M methylene blue Chemical compound [Cl-].C1=CC(N(C)C)=CC2=[S+]C3=CC(N(C)C)=CC=C3N=C21 CXKWCBBOMKCUKX-UHFFFAOYSA-M 0.000 description 2
- 229960000907 methylthioninium chloride Drugs 0.000 description 2
- 238000002493 microarray Methods 0.000 description 2
- 230000002906 microbiologic effect Effects 0.000 description 2
- 229940126619 mouse monoclonal antibody Drugs 0.000 description 2
- 230000035772 mutation Effects 0.000 description 2
- 201000008026 nephroblastoma Diseases 0.000 description 2
- 230000008816 organ damage Effects 0.000 description 2
- 229920002866 paraformaldehyde Polymers 0.000 description 2
- 230000036961 partial effect Effects 0.000 description 2
- 229940031734 peptide cancer vaccine Drugs 0.000 description 2
- 210000005259 peripheral blood Anatomy 0.000 description 2
- 239000011886 peripheral blood Substances 0.000 description 2
- 230000002093 peripheral effect Effects 0.000 description 2
- COLNVLDHVKWLRT-UHFFFAOYSA-N phenylalanine Natural products OC(=O)C(N)CC1=CC=CC=C1 COLNVLDHVKWLRT-UHFFFAOYSA-N 0.000 description 2
- 230000026731 phosphorylation Effects 0.000 description 2
- 238000006366 phosphorylation reaction Methods 0.000 description 2
- 229920001223 polyethylene glycol Polymers 0.000 description 2
- 239000000256 polyoxyethylene sorbitan monolaurate Substances 0.000 description 2
- 235000010486 polyoxyethylene sorbitan monolaurate Nutrition 0.000 description 2
- 229920001282 polysaccharide Polymers 0.000 description 2
- 239000005017 polysaccharide Substances 0.000 description 2
- 229920002451 polyvinyl alcohol Polymers 0.000 description 2
- 239000001103 potassium chloride Substances 0.000 description 2
- 235000011164 potassium chloride Nutrition 0.000 description 2
- 229960004583 pranlukast Drugs 0.000 description 2
- UAJUXJSXCLUTNU-UHFFFAOYSA-N pranlukast Chemical compound C=1C=C(OCCCCC=2C=CC=CC=2)C=CC=1C(=O)NC(C=1)=CC=C(C(C=2)=O)C=1OC=2C=1N=NNN=1 UAJUXJSXCLUTNU-UHFFFAOYSA-N 0.000 description 2
- 230000001376 precipitating effect Effects 0.000 description 2
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 description 2
- 238000012545 processing Methods 0.000 description 2
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 description 2
- 239000008213 purified water Substances 0.000 description 2
- 150000003254 radicals Chemical class 0.000 description 2
- 230000002829 reductive effect Effects 0.000 description 2
- 238000000611 regression analysis Methods 0.000 description 2
- 230000001105 regulatory effect Effects 0.000 description 2
- 230000004044 response Effects 0.000 description 2
- 102220093888 rs745424307 Human genes 0.000 description 2
- 229940037001 sodium edetate Drugs 0.000 description 2
- 235000010339 sodium tetraborate Nutrition 0.000 description 2
- 239000004328 sodium tetraborate Substances 0.000 description 2
- 210000004988 splenocyte Anatomy 0.000 description 2
- 239000007921 spray Substances 0.000 description 2
- 239000003381 stabilizer Substances 0.000 description 2
- 239000007858 starting material Substances 0.000 description 2
- 238000007619 statistical method Methods 0.000 description 2
- 239000003765 sweetening agent Substances 0.000 description 2
- 238000001308 synthesis method Methods 0.000 description 2
- 238000011191 terminal modification Methods 0.000 description 2
- 125000000101 thioether group Chemical group 0.000 description 2
- QORWJWZARLRLPR-UHFFFAOYSA-H tricalcium bis(phosphate) Chemical compound [Ca+2].[Ca+2].[Ca+2].[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O QORWJWZARLRLPR-UHFFFAOYSA-H 0.000 description 2
- LENZDBCJOHFCAS-UHFFFAOYSA-N tris Chemical compound OCC(N)(CO)CO LENZDBCJOHFCAS-UHFFFAOYSA-N 0.000 description 2
- 229940035893 uracil Drugs 0.000 description 2
- 108700026220 vif Genes Proteins 0.000 description 2
- 238000005406 washing Methods 0.000 description 2
- 239000008096 xylene Substances 0.000 description 2
- FTLYMKDSHNWQKD-UHFFFAOYSA-N (2,4,5-trichlorophenyl)boronic acid Chemical compound OB(O)C1=CC(Cl)=C(Cl)C=C1Cl FTLYMKDSHNWQKD-UHFFFAOYSA-N 0.000 description 1
- QMUKFZMAYHSADK-MZDDNWLQSA-N (2S)-2-[[(2S)-2-[[2-[[(2S)-2-[[(2S)-2-[[(2S)-2-[[(2S)-2-[[2-[[(2S)-1-[(2S)-1-[(2S)-2-[[(2S)-2-[[(2S)-2-[[(2S)-2-[[(2S)-2-[[(2S)-1-[(2S)-2-[[(2S)-2-amino-3-(1H-indol-3-yl)propanoyl]amino]propanoyl]pyrrolidine-2-carbonyl]amino]-3-methylbutanoyl]amino]-4-methylpentanoyl]amino]-3-carboxypropanoyl]amino]-3-phenylpropanoyl]amino]propanoyl]pyrrolidine-2-carbonyl]pyrrolidine-2-carbonyl]amino]acetyl]amino]propanoyl]amino]-3-hydroxypropanoyl]amino]propanoyl]amino]-3-(4-hydroxyphenyl)propanoyl]amino]acetyl]amino]-3-hydroxypropanoyl]amino]-4-methylpentanoic acid Chemical compound CC(C)C[C@H](NC(=O)[C@H](CO)NC(=O)CNC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](C)NC(=O)[C@H](CO)NC(=O)[C@H](C)NC(=O)CNC(=O)[C@@H]1CCCN1C(=O)[C@@H]1CCCN1C(=O)[C@H](C)NC(=O)[C@H](Cc1ccccc1)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@@H](NC(=O)[C@@H]1CCCN1C(=O)[C@H](C)NC(=O)[C@@H](N)Cc1c[nH]c2ccccc12)C(C)C)C(O)=O QMUKFZMAYHSADK-MZDDNWLQSA-N 0.000 description 1
- PFBPIHUEFUNOIS-DVQPEGLTSA-N (2S)-2-[[(2S)-6-amino-2-[[(2S)-2-[[(2S)-2-[[(2S)-2-[[(2S)-2-[[(2S)-5-amino-2-[[(2S)-2-[[(2S)-2-[[(2S)-2-[[(2S)-2-[[(2S)-6-amino-2-[[(2S)-2-[[(2S)-2-[[(2S)-5-carbamimidamido-2-[[(2S)-2,6-diaminohexanoyl]amino]pentanoyl]amino]-3-(4-hydroxyphenyl)propanoyl]amino]-3-phenylpropanoyl]amino]hexanoyl]amino]-4-methylpentanoyl]amino]-3-hydroxypropanoyl]amino]-3-(1H-imidazol-4-yl)propanoyl]amino]-4-methylpentanoyl]amino]-5-oxopentanoyl]amino]-4-methylsulfanylbutanoyl]amino]-3-(1H-imidazol-4-yl)propanoyl]amino]-3-hydroxypropanoyl]amino]-5-carbamimidamidopentanoyl]amino]hexanoyl]amino]-3-(1H-imidazol-4-yl)propanoic acid Chemical compound CSCC[C@H](NC(=O)[C@H](CCC(N)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](Cc1c[nH]cn1)NC(=O)[C@H](CO)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CCCCN)NC(=O)[C@H](Cc1ccccc1)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@@H](N)CCCCN)C(=O)N[C@@H](Cc1c[nH]cn1)C(=O)N[C@@H](CO)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](Cc1c[nH]cn1)C(O)=O PFBPIHUEFUNOIS-DVQPEGLTSA-N 0.000 description 1
- ASWBNKHCZGQVJV-UHFFFAOYSA-N (3-hexadecanoyloxy-2-hydroxypropyl) 2-(trimethylazaniumyl)ethyl phosphate Chemical compound CCCCCCCCCCCCCCCC(=O)OCC(O)COP([O-])(=O)OCC[N+](C)(C)C ASWBNKHCZGQVJV-UHFFFAOYSA-N 0.000 description 1
- VGONTNSXDCQUGY-RRKCRQDMSA-N 2'-deoxyinosine Chemical group C1[C@H](O)[C@@H](CO)O[C@H]1N1C(N=CNC2=O)=C2N=C1 VGONTNSXDCQUGY-RRKCRQDMSA-N 0.000 description 1
- SPSPIUSUWPLVKD-UHFFFAOYSA-N 2,3-dibutyl-6-methylphenol Chemical compound CCCCC1=CC=C(C)C(O)=C1CCCC SPSPIUSUWPLVKD-UHFFFAOYSA-N 0.000 description 1
- FJKROLUGYXJWQN-UHFFFAOYSA-M 4-hydroxybenzoate Chemical compound OC1=CC=C(C([O-])=O)C=C1 FJKROLUGYXJWQN-UHFFFAOYSA-M 0.000 description 1
- UJBCLAXPPIDQEE-UHFFFAOYSA-N 5-prop-1-ynyl-1h-pyrimidine-2,4-dione Chemical class CC#CC1=CNC(=O)NC1=O UJBCLAXPPIDQEE-UHFFFAOYSA-N 0.000 description 1
- SLXKOJJOQWFEFD-UHFFFAOYSA-N 6-aminohexanoic acid Chemical compound NCCCCCC(O)=O SLXKOJJOQWFEFD-UHFFFAOYSA-N 0.000 description 1
- 108700028369 Alleles Proteins 0.000 description 1
- 239000004475 Arginine Substances 0.000 description 1
- UEONJSPBTSWKOI-CIUDSAMLSA-N Asn-Gln-Met Chemical compound [H]N[C@@H](CC(N)=O)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCSC)C(O)=O UEONJSPBTSWKOI-CIUDSAMLSA-N 0.000 description 1
- 108010011485 Aspartame Proteins 0.000 description 1
- 241000972773 Aulopiformes Species 0.000 description 1
- BTBUEUYNUDRHOZ-UHFFFAOYSA-N Borate Chemical compound [O-]B([O-])[O-] BTBUEUYNUDRHOZ-UHFFFAOYSA-N 0.000 description 1
- BVKZGUZCCUSVTD-UHFFFAOYSA-L Carbonate Chemical compound [O-]C([O-])=O BVKZGUZCCUSVTD-UHFFFAOYSA-L 0.000 description 1
- WYZLWZNAWQNLGQ-FXQIFTODSA-N Cys-Cys-Met Chemical compound CSCC[C@@H](C(=O)O)NC(=O)[C@H](CS)NC(=O)[C@H](CS)N WYZLWZNAWQNLGQ-FXQIFTODSA-N 0.000 description 1
- GHUVBPIYQYXXEF-SRVKXCTJSA-N Cys-Cys-Tyr Chemical compound SC[C@H](N)C(=O)N[C@@H](CS)C(=O)N[C@H](C(O)=O)CC1=CC=C(O)C=C1 GHUVBPIYQYXXEF-SRVKXCTJSA-N 0.000 description 1
- 108090000695 Cytokines Proteins 0.000 description 1
- 102000004127 Cytokines Human genes 0.000 description 1
- 238000002965 ELISA Methods 0.000 description 1
- 239000004386 Erythritol Substances 0.000 description 1
- UNXHWFMMPAWVPI-UHFFFAOYSA-N Erythritol Natural products OCC(O)C(O)CO UNXHWFMMPAWVPI-UHFFFAOYSA-N 0.000 description 1
- 241000588724 Escherichia coli Species 0.000 description 1
- 229920001917 Ficoll Polymers 0.000 description 1
- 238000012413 Fluorescence activated cell sorting analysis Methods 0.000 description 1
- 208000000321 Gardner Syndrome Diseases 0.000 description 1
- 208000012671 Gastrointestinal haemorrhages Diseases 0.000 description 1
- MSHXWFKYXJTLEZ-CIUDSAMLSA-N Gln-Met-Asn Chemical compound CSCC[C@@H](C(=O)N[C@@H](CC(=O)N)C(=O)O)NC(=O)[C@H](CCC(=O)N)N MSHXWFKYXJTLEZ-CIUDSAMLSA-N 0.000 description 1
- 239000004471 Glycine Substances 0.000 description 1
- 108010035452 HLA-A1 Antigen Proteins 0.000 description 1
- 108010087480 HLA-B40 Antigen Proteins 0.000 description 1
- 108010091938 HLA-B7 Antigen Proteins 0.000 description 1
- 108010010378 HLA-DP Antigens Proteins 0.000 description 1
- 102000015789 HLA-DP Antigens Human genes 0.000 description 1
- 108010062347 HLA-DQ Antigens Proteins 0.000 description 1
- 208000028782 Hereditary disease Diseases 0.000 description 1
- 239000004354 Hydroxyethyl cellulose Substances 0.000 description 1
- 229920000663 Hydroxyethyl cellulose Polymers 0.000 description 1
- 102100037850 Interferon gamma Human genes 0.000 description 1
- 102000006992 Interferon-alpha Human genes 0.000 description 1
- 108010047761 Interferon-alpha Proteins 0.000 description 1
- 108010074328 Interferon-gamma Proteins 0.000 description 1
- ONIBWKKTOPOVIA-BYPYZUCNSA-N L-Proline Chemical compound OC(=O)[C@@H]1CCCN1 ONIBWKKTOPOVIA-BYPYZUCNSA-N 0.000 description 1
- QNAYBMKLOCPYGJ-REOHCLBHSA-N L-alanine Chemical compound C[C@H](N)C(O)=O QNAYBMKLOCPYGJ-REOHCLBHSA-N 0.000 description 1
- ODKSFYDXXFIFQN-BYPYZUCNSA-P L-argininium(2+) Chemical compound NC(=[NH2+])NCCC[C@H]([NH3+])C(O)=O ODKSFYDXXFIFQN-BYPYZUCNSA-P 0.000 description 1
- HNDVDQJCIGZPNO-YFKPBYRVSA-N L-histidine Chemical compound OC(=O)[C@@H](N)CC1=CN=CN1 HNDVDQJCIGZPNO-YFKPBYRVSA-N 0.000 description 1
- OUYCCCASQSFEME-QMMMGPOBSA-N L-tyrosine Chemical compound OC(=O)[C@@H](N)CC1=CC=C(O)C=C1 OUYCCCASQSFEME-QMMMGPOBSA-N 0.000 description 1
- 108700018351 Major Histocompatibility Complex Proteins 0.000 description 1
- 241000124008 Mammalia Species 0.000 description 1
- 208000024556 Mendelian disease Diseases 0.000 description 1
- 206010027476 Metastases Diseases 0.000 description 1
- 229920000168 Microcrystalline cellulose Polymers 0.000 description 1
- 125000001429 N-terminal alpha-amino-acid group Chemical group 0.000 description 1
- 239000000020 Nitrocellulose Substances 0.000 description 1
- 239000004677 Nylon Substances 0.000 description 1
- 108010038807 Oligopeptides Proteins 0.000 description 1
- 102000015636 Oligopeptides Human genes 0.000 description 1
- 108010066305 Parkinson Disease Associated Proteins Proteins 0.000 description 1
- 102000018315 Parkinson Disease Associated Proteins Human genes 0.000 description 1
- 108010033276 Peptide Fragments Proteins 0.000 description 1
- 102000007079 Peptide Fragments Human genes 0.000 description 1
- 108010043958 Peptoids Proteins 0.000 description 1
- 229920003171 Poly (ethylene oxide) Polymers 0.000 description 1
- 239000004698 Polyethylene Substances 0.000 description 1
- ONIBWKKTOPOVIA-UHFFFAOYSA-N Proline Natural products OC(=O)C1CCCN1 ONIBWKKTOPOVIA-UHFFFAOYSA-N 0.000 description 1
- 102220472129 Protein Wnt-2_C23A_mutation Human genes 0.000 description 1
- 238000012300 Sequence Analysis Methods 0.000 description 1
- DWAQJAXMDSEUJJ-UHFFFAOYSA-M Sodium bisulfite Chemical compound [Na+].OS([O-])=O DWAQJAXMDSEUJJ-UHFFFAOYSA-M 0.000 description 1
- 239000004288 Sodium dehydroacetate Substances 0.000 description 1
- 238000002105 Southern blotting Methods 0.000 description 1
- 239000007984 Tris EDTA buffer Substances 0.000 description 1
- AKKYBQGHUAWPJR-MNSWYVGCSA-N Tyr-Thr-Trp Chemical compound C[C@H]([C@@H](C(=O)N[C@@H](CC1=CNC2=CC=CC=C21)C(=O)O)NC(=O)[C@H](CC3=CC=C(C=C3)O)N)O AKKYBQGHUAWPJR-MNSWYVGCSA-N 0.000 description 1
- 208000000260 Warts Diseases 0.000 description 1
- 108700025700 Wilms Tumor Genes Proteins 0.000 description 1
- 101100082060 Xenopus laevis pou5f1.1 gene Proteins 0.000 description 1
- TVXBFESIOXBWNM-UHFFFAOYSA-N Xylitol Natural products OCCC(O)C(O)C(O)CCO TVXBFESIOXBWNM-UHFFFAOYSA-N 0.000 description 1
- 102000038627 Zinc finger transcription factors Human genes 0.000 description 1
- 108091007916 Zinc finger transcription factors Proteins 0.000 description 1
- JLCPHMBAVCMARE-UHFFFAOYSA-N [3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[5-(2-amino-6-oxo-1H-purin-9-yl)-3-[[3-[[3-[[3-[[3-[[3-[[5-(2-amino-6-oxo-1H-purin-9-yl)-3-[[5-(2-amino-6-oxo-1H-purin-9-yl)-3-hydroxyoxolan-2-yl]methoxy-hydroxyphosphoryl]oxyoxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxyoxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methyl [5-(6-aminopurin-9-yl)-2-(hydroxymethyl)oxolan-3-yl] hydrogen phosphate Polymers Cc1cn(C2CC(OP(O)(=O)OCC3OC(CC3OP(O)(=O)OCC3OC(CC3O)n3cnc4c3nc(N)[nH]c4=O)n3cnc4c3nc(N)[nH]c4=O)C(COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3CO)n3cnc4c(N)ncnc34)n3ccc(N)nc3=O)n3cnc4c(N)ncnc34)n3ccc(N)nc3=O)n3ccc(N)nc3=O)n3ccc(N)nc3=O)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)n3cc(C)c(=O)[nH]c3=O)n3cc(C)c(=O)[nH]c3=O)n3ccc(N)nc3=O)n3cc(C)c(=O)[nH]c3=O)n3cnc4c3nc(N)[nH]c4=O)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)O2)c(=O)[nH]c1=O JLCPHMBAVCMARE-UHFFFAOYSA-N 0.000 description 1
- 239000008351 acetate buffer Substances 0.000 description 1
- 230000002378 acidificating effect Effects 0.000 description 1
- 239000013543 active substance Substances 0.000 description 1
- 208000009956 adenocarcinoma Diseases 0.000 description 1
- 235000004279 alanine Nutrition 0.000 description 1
- 150000001298 alcohols Chemical class 0.000 description 1
- 229910000147 aluminium phosphate Inorganic materials 0.000 description 1
- WMGSQTMJHBYJMQ-UHFFFAOYSA-N aluminum;magnesium;silicate Chemical compound [Mg+2].[Al+3].[O-][Si]([O-])([O-])[O-] WMGSQTMJHBYJMQ-UHFFFAOYSA-N 0.000 description 1
- 230000009435 amidation Effects 0.000 description 1
- 238000007112 amidation reaction Methods 0.000 description 1
- 229960002684 aminocaproic acid Drugs 0.000 description 1
- 239000003708 ampul Substances 0.000 description 1
- 230000033115 angiogenesis Effects 0.000 description 1
- 238000010171 animal model Methods 0.000 description 1
- 230000006907 apoptotic process Effects 0.000 description 1
- 238000013459 approach Methods 0.000 description 1
- 239000007864 aqueous solution Substances 0.000 description 1
- 239000003125 aqueous solvent Substances 0.000 description 1
- ODKSFYDXXFIFQN-UHFFFAOYSA-N arginine Natural products OC(=O)C(N)CCCNC(N)=N ODKSFYDXXFIFQN-UHFFFAOYSA-N 0.000 description 1
- 210000001815 ascending colon Anatomy 0.000 description 1
- 235000010323 ascorbic acid Nutrition 0.000 description 1
- 239000011668 ascorbic acid Substances 0.000 description 1
- 229960005070 ascorbic acid Drugs 0.000 description 1
- 239000000605 aspartame Substances 0.000 description 1
- 235000010357 aspartame Nutrition 0.000 description 1
- 229960003438 aspartame Drugs 0.000 description 1
- IAOZJIPTCAWIRG-QWRGUYRKSA-N aspartame Chemical compound OC(=O)C[C@H](N)C(=O)N[C@H](C(=O)OC)CC1=CC=CC=C1 IAOZJIPTCAWIRG-QWRGUYRKSA-N 0.000 description 1
- 239000012752 auxiliary agent Substances 0.000 description 1
- 230000008901 benefit Effects 0.000 description 1
- 229960000686 benzalkonium chloride Drugs 0.000 description 1
- 235000019445 benzyl alcohol Nutrition 0.000 description 1
- 229960002903 benzyl benzoate Drugs 0.000 description 1
- CADWTSSKOVRVJC-UHFFFAOYSA-N benzyl(dimethyl)azanium;chloride Chemical compound [Cl-].C[NH+](C)CC1=CC=CC=C1 CADWTSSKOVRVJC-UHFFFAOYSA-N 0.000 description 1
- 239000011230 binding agent Substances 0.000 description 1
- 238000002306 biochemical method Methods 0.000 description 1
- 239000003181 biological factor Substances 0.000 description 1
- 230000033228 biological regulation Effects 0.000 description 1
- 230000015572 biosynthetic process Effects 0.000 description 1
- 230000037396 body weight Effects 0.000 description 1
- 238000006664 bond formation reaction Methods 0.000 description 1
- 229960002645 boric acid Drugs 0.000 description 1
- 239000007853 buffer solution Substances 0.000 description 1
- 239000006172 buffering agent Substances 0.000 description 1
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 235000010354 butylated hydroxytoluene Nutrition 0.000 description 1
- CHRHZFQUDFAQEQ-UHFFFAOYSA-L calcium;2-hydroxyacetate Chemical compound [Ca+2].OCC([O-])=O.OCC([O-])=O CHRHZFQUDFAQEQ-UHFFFAOYSA-L 0.000 description 1
- 238000004422 calculation algorithm Methods 0.000 description 1
- 238000004364 calculation method Methods 0.000 description 1
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 1
- 239000004359 castor oil Substances 0.000 description 1
- 235000019438 castor oil Nutrition 0.000 description 1
- 210000004534 cecum Anatomy 0.000 description 1
- 230000024245 cell differentiation Effects 0.000 description 1
- 230000004663 cell proliferation Effects 0.000 description 1
- 229920002678 cellulose Polymers 0.000 description 1
- 235000010980 cellulose Nutrition 0.000 description 1
- 238000012512 characterization method Methods 0.000 description 1
- 230000006328 chemical modification of amino acids Effects 0.000 description 1
- 208000013549 childhood kidney neoplasm Diseases 0.000 description 1
- 229960004926 chlorobutanol Drugs 0.000 description 1
- 238000010367 cloning Methods 0.000 description 1
- 238000000576 coating method Methods 0.000 description 1
- 239000003086 colorant Substances 0.000 description 1
- 230000009137 competitive binding Effects 0.000 description 1
- 230000000295 complement effect Effects 0.000 description 1
- 239000002299 complementary DNA Substances 0.000 description 1
- 238000004590 computer program Methods 0.000 description 1
- 230000021615 conjugation Effects 0.000 description 1
- 239000000470 constituent Substances 0.000 description 1
- 239000012050 conventional carrier Substances 0.000 description 1
- 238000005138 cryopreservation Methods 0.000 description 1
- 125000004122 cyclic group Chemical group 0.000 description 1
- XUJNEKJLAYXESH-UHFFFAOYSA-N cysteine Natural products SCC(N)C(O)=O XUJNEKJLAYXESH-UHFFFAOYSA-N 0.000 description 1
- 235000018417 cysteine Nutrition 0.000 description 1
- LEVWYRKDKASIDU-IMJSIDKUSA-N cystine group Chemical group C([C@@H](C(=O)O)N)SSC[C@@H](C(=O)O)N LEVWYRKDKASIDU-IMJSIDKUSA-N 0.000 description 1
- 230000003013 cytotoxicity Effects 0.000 description 1
- 231100000135 cytotoxicity Toxicity 0.000 description 1
- 230000007812 deficiency Effects 0.000 description 1
- 238000001514 detection method Methods 0.000 description 1
- 229960000633 dextran sulfate Drugs 0.000 description 1
- 238000003745 diagnosis Methods 0.000 description 1
- 235000005911 diet Nutrition 0.000 description 1
- 230000037213 diet Effects 0.000 description 1
- LOKCTEFSRHRXRJ-UHFFFAOYSA-I dipotassium trisodium dihydrogen phosphate hydrogen phosphate dichloride Chemical compound P(=O)(O)(O)[O-].[K+].P(=O)(O)([O-])[O-].[Na+].[Na+].[Cl-].[K+].[Cl-].[Na+] LOKCTEFSRHRXRJ-UHFFFAOYSA-I 0.000 description 1
- 239000007884 disintegrant Substances 0.000 description 1
- 239000002270 dispersing agent Substances 0.000 description 1
- 238000004090 dissolution Methods 0.000 description 1
- 239000012153 distilled water Substances 0.000 description 1
- 238000009826 distribution Methods 0.000 description 1
- 230000035622 drinking Effects 0.000 description 1
- 239000003937 drug carrier Substances 0.000 description 1
- 238000007908 dry granulation Methods 0.000 description 1
- 239000003995 emulsifying agent Substances 0.000 description 1
- 239000000839 emulsion Substances 0.000 description 1
- 238000002674 endoscopic surgery Methods 0.000 description 1
- 238000001839 endoscopy Methods 0.000 description 1
- 235000019414 erythritol Nutrition 0.000 description 1
- UNXHWFMMPAWVPI-ZXZARUISSA-N erythritol Chemical compound OC[C@H](O)[C@H](O)CO UNXHWFMMPAWVPI-ZXZARUISSA-N 0.000 description 1
- 229940009714 erythritol Drugs 0.000 description 1
- 238000010195 expression analysis Methods 0.000 description 1
- 210000002950 fibroblast Anatomy 0.000 description 1
- 206010016629 fibroma Diseases 0.000 description 1
- 238000001914 filtration Methods 0.000 description 1
- 239000000796 flavoring agent Substances 0.000 description 1
- 235000013355 food flavoring agent Nutrition 0.000 description 1
- 239000003205 fragrance Substances 0.000 description 1
- 238000004108 freeze drying Methods 0.000 description 1
- 208000030304 gastrointestinal bleeding Diseases 0.000 description 1
- 239000000499 gel Substances 0.000 description 1
- 208000016361 genetic disease Diseases 0.000 description 1
- 230000002068 genetic effect Effects 0.000 description 1
- 239000011521 glass Substances 0.000 description 1
- 229930195712 glutamate Natural products 0.000 description 1
- ZEMPKEQAKRGZGQ-XOQCFJPHSA-N glycerol triricinoleate Natural products CCCCCC[C@@H](O)CC=CCCCCCCCC(=O)OC[C@@H](COC(=O)CCCCCCCC=CC[C@@H](O)CCCCCC)OC(=O)CCCCCCCC=CC[C@H](O)CCCCCC ZEMPKEQAKRGZGQ-XOQCFJPHSA-N 0.000 description 1
- 230000013595 glycosylation Effects 0.000 description 1
- 238000006206 glycosylation reaction Methods 0.000 description 1
- 229930004094 glycosylphosphatidylinositol Natural products 0.000 description 1
- 239000007902 hard capsule Substances 0.000 description 1
- 230000036541 health Effects 0.000 description 1
- 235000019534 high fructose corn syrup Nutrition 0.000 description 1
- HNDVDQJCIGZPNO-UHFFFAOYSA-N histidine Natural products OC(=O)C(N)CC1=CN=CN1 HNDVDQJCIGZPNO-UHFFFAOYSA-N 0.000 description 1
- 235000012907 honey Nutrition 0.000 description 1
- 125000004435 hydrogen atom Chemical group [H]* 0.000 description 1
- 230000002209 hydrophobic effect Effects 0.000 description 1
- 235000019447 hydroxyethyl cellulose Nutrition 0.000 description 1
- 239000001866 hydroxypropyl methyl cellulose Substances 0.000 description 1
- 235000010979 hydroxypropyl methyl cellulose Nutrition 0.000 description 1
- 229920003088 hydroxypropyl methyl cellulose Polymers 0.000 description 1
- UFVKGYZPFZQRLF-UHFFFAOYSA-N hydroxypropyl methyl cellulose Chemical compound OC1C(O)C(OC)OC(CO)C1OC1C(O)C(O)C(OC2C(C(O)C(OC3C(C(O)C(O)C(CO)O3)O)C(CO)O2)O)C(CO)O1 UFVKGYZPFZQRLF-UHFFFAOYSA-N 0.000 description 1
- 229920003132 hydroxypropyl methylcellulose phthalate Polymers 0.000 description 1
- 229940031704 hydroxypropyl methylcellulose phthalate Drugs 0.000 description 1
- 210000002865 immune cell Anatomy 0.000 description 1
- 230000002163 immunogen Effects 0.000 description 1
- 230000016784 immunoglobulin production Effects 0.000 description 1
- 238000012744 immunostaining Methods 0.000 description 1
- 238000007901 in situ hybridization Methods 0.000 description 1
- 238000000099 in vitro assay Methods 0.000 description 1
- 230000008595 infiltration Effects 0.000 description 1
- 238000001764 infiltration Methods 0.000 description 1
- 239000004615 ingredient Substances 0.000 description 1
- 230000002401 inhibitory effect Effects 0.000 description 1
- 230000005764 inhibitory process Effects 0.000 description 1
- 230000003834 intracellular effect Effects 0.000 description 1
- 239000007951 isotonicity adjuster Substances 0.000 description 1
- 108010045069 keyhole-limpet hemocyanin Proteins 0.000 description 1
- 238000002372 labelling Methods 0.000 description 1
- 201000011061 large intestine cancer Diseases 0.000 description 1
- 230000003902 lesion Effects 0.000 description 1
- 210000000265 leukocyte Anatomy 0.000 description 1
- 230000000670 limiting effect Effects 0.000 description 1
- 230000029226 lipidation Effects 0.000 description 1
- 239000012669 liquid formulation Substances 0.000 description 1
- 239000006210 lotion Substances 0.000 description 1
- 239000000314 lubricant Substances 0.000 description 1
- 239000006166 lysate Substances 0.000 description 1
- 235000019359 magnesium stearate Nutrition 0.000 description 1
- 238000012423 maintenance Methods 0.000 description 1
- 239000000845 maltitol Substances 0.000 description 1
- 235000010449 maltitol Nutrition 0.000 description 1
- VQHSOMBJVWLPSR-WUJBLJFYSA-N maltitol Chemical compound OC[C@H](O)[C@@H](O)[C@@H]([C@H](O)CO)O[C@H]1O[C@H](CO)[C@@H](O)[C@H](O)[C@H]1O VQHSOMBJVWLPSR-WUJBLJFYSA-N 0.000 description 1
- 229940035436 maltitol Drugs 0.000 description 1
- 229960001855 mannitol Drugs 0.000 description 1
- 238000005259 measurement Methods 0.000 description 1
- 239000012528 membrane Substances 0.000 description 1
- HEBKCHPVOIAQTA-UHFFFAOYSA-N meso ribitol Natural products OCC(O)C(O)C(O)CO HEBKCHPVOIAQTA-UHFFFAOYSA-N 0.000 description 1
- 230000009401 metastasis Effects 0.000 description 1
- 229920000609 methyl cellulose Polymers 0.000 description 1
- 239000001923 methylcellulose Substances 0.000 description 1
- 235000010981 methylcellulose Nutrition 0.000 description 1
- 239000000693 micelle Substances 0.000 description 1
- 244000005700 microbiome Species 0.000 description 1
- 239000008108 microcrystalline cellulose Substances 0.000 description 1
- 235000019813 microcrystalline cellulose Nutrition 0.000 description 1
- 229940016286 microcrystalline cellulose Drugs 0.000 description 1
- 239000011259 mixed solution Substances 0.000 description 1
- 238000010172 mouse model Methods 0.000 description 1
- 238000002703 mutagenesis Methods 0.000 description 1
- 230000007498 myristoylation Effects 0.000 description 1
- OHDXDNUPVVYWOV-UHFFFAOYSA-N n-methyl-1-(2-naphthalen-1-ylsulfanylphenyl)methanamine Chemical compound CNCC1=CC=CC=C1SC1=CC=CC2=CC=CC=C12 OHDXDNUPVVYWOV-UHFFFAOYSA-N 0.000 description 1
- 210000000822 natural killer cell Anatomy 0.000 description 1
- 208000012108 neoplastic polyp Diseases 0.000 description 1
- 230000007935 neutral effect Effects 0.000 description 1
- 229920001220 nitrocellulos Polymers 0.000 description 1
- 239000002736 nonionic surfactant Substances 0.000 description 1
- 229920001778 nylon Polymers 0.000 description 1
- 239000002674 ointment Substances 0.000 description 1
- 239000002997 ophthalmic solution Substances 0.000 description 1
- 150000002894 organic compounds Chemical class 0.000 description 1
- 239000007800 oxidant agent Substances 0.000 description 1
- 210000000277 pancreatic duct Anatomy 0.000 description 1
- 229940023041 peptide vaccine Drugs 0.000 description 1
- 239000000816 peptidomimetic Substances 0.000 description 1
- WVDDGKGOMKODPV-ZQBYOMGUSA-N phenyl(114C)methanol Chemical compound O[14CH2]C1=CC=CC=C1 WVDDGKGOMKODPV-ZQBYOMGUSA-N 0.000 description 1
- 239000006187 pill Substances 0.000 description 1
- 229920001983 poloxamer Polymers 0.000 description 1
- 229920000573 polyethylene Polymers 0.000 description 1
- 229920005862 polyol Polymers 0.000 description 1
- 150000003077 polyols Chemical class 0.000 description 1
- 239000000244 polyoxyethylene sorbitan monooleate Substances 0.000 description 1
- 235000010482 polyoxyethylene sorbitan monooleate Nutrition 0.000 description 1
- 229920000053 polysorbate 80 Polymers 0.000 description 1
- 229940068968 polysorbate 80 Drugs 0.000 description 1
- 235000019422 polyvinyl alcohol Nutrition 0.000 description 1
- 238000010837 poor prognosis Methods 0.000 description 1
- 230000001323 posttranslational effect Effects 0.000 description 1
- 239000000843 powder Substances 0.000 description 1
- 230000002335 preservative effect Effects 0.000 description 1
- 230000008569 process Effects 0.000 description 1
- 238000004393 prognosis Methods 0.000 description 1
- 238000011321 prophylaxis Methods 0.000 description 1
- 235000013772 propylene glycol Nutrition 0.000 description 1
- 235000004252 protein component Nutrition 0.000 description 1
- 238000002708 random mutagenesis Methods 0.000 description 1
- 239000002994 raw material Substances 0.000 description 1
- 210000000664 rectum Anatomy 0.000 description 1
- 238000006894 reductive elimination reaction Methods 0.000 description 1
- 230000008844 regulatory mechanism Effects 0.000 description 1
- 239000002336 ribonucleotide Substances 0.000 description 1
- 102220214961 rs1060503561 Human genes 0.000 description 1
- 102220328147 rs80356994 Human genes 0.000 description 1
- 235000019204 saccharin Nutrition 0.000 description 1
- CVHZOJJKTDOEJC-UHFFFAOYSA-N saccharin Chemical compound C1=CC=C2C(=O)NS(=O)(=O)C2=C1 CVHZOJJKTDOEJC-UHFFFAOYSA-N 0.000 description 1
- 229940081974 saccharin Drugs 0.000 description 1
- 239000000901 saccharin and its Na,K and Ca salt Substances 0.000 description 1
- 229940085605 saccharin sodium Drugs 0.000 description 1
- 235000019515 salmon Nutrition 0.000 description 1
- 238000012216 screening Methods 0.000 description 1
- 230000028327 secretion Effects 0.000 description 1
- 238000004062 sedimentation Methods 0.000 description 1
- 230000001235 sensitizing effect Effects 0.000 description 1
- 239000008159 sesame oil Substances 0.000 description 1
- 235000011803 sesame oil Nutrition 0.000 description 1
- 230000037384 skin absorption Effects 0.000 description 1
- 231100000274 skin absorption Toxicity 0.000 description 1
- 201000010153 skin papilloma Diseases 0.000 description 1
- 230000000391 smoking effect Effects 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- 229940001607 sodium bisulfite Drugs 0.000 description 1
- 229960001790 sodium citrate Drugs 0.000 description 1
- 235000011083 sodium citrates Nutrition 0.000 description 1
- 235000019259 sodium dehydroacetate Nutrition 0.000 description 1
- 229940079839 sodium dehydroacetate Drugs 0.000 description 1
- 235000010267 sodium hydrogen sulphite Nutrition 0.000 description 1
- 239000001488 sodium phosphate Substances 0.000 description 1
- 229910000162 sodium phosphate Inorganic materials 0.000 description 1
- 239000012064 sodium phosphate buffer Substances 0.000 description 1
- AKHNMLFCWUSKQB-UHFFFAOYSA-L sodium thiosulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=S AKHNMLFCWUSKQB-UHFFFAOYSA-L 0.000 description 1
- 229940001474 sodium thiosulfate Drugs 0.000 description 1
- 235000019345 sodium thiosulphate Nutrition 0.000 description 1
- DSOWAKKSGYUMTF-GZOLSCHFSA-M sodium;(1e)-1-(6-methyl-2,4-dioxopyran-3-ylidene)ethanolate Chemical compound [Na+].C\C([O-])=C1/C(=O)OC(C)=CC1=O DSOWAKKSGYUMTF-GZOLSCHFSA-M 0.000 description 1
- 239000007901 soft capsule Substances 0.000 description 1
- 239000012453 solvate Substances 0.000 description 1
- 229960002920 sorbitol Drugs 0.000 description 1
- 239000003549 soybean oil Substances 0.000 description 1
- 235000012424 soybean oil Nutrition 0.000 description 1
- 241000894007 species Species 0.000 description 1
- 239000012192 staining solution Substances 0.000 description 1
- 230000001954 sterilising effect Effects 0.000 description 1
- 230000000638 stimulation Effects 0.000 description 1
- 238000003756 stirring Methods 0.000 description 1
- 230000035882 stress Effects 0.000 description 1
- 239000006190 sub-lingual tablet Substances 0.000 description 1
- 150000005846 sugar alcohols Polymers 0.000 description 1
- 239000013589 supplement Substances 0.000 description 1
- 230000001629 suppression Effects 0.000 description 1
- 230000020382 suppression by virus of host antigen processing and presentation of peptide antigen via MHC class I Effects 0.000 description 1
- 238000001356 surgical procedure Methods 0.000 description 1
- 239000000375 suspending agent Substances 0.000 description 1
- 239000000725 suspension Substances 0.000 description 1
- 230000008961 swelling Effects 0.000 description 1
- 238000003786 synthesis reaction Methods 0.000 description 1
- 229920001059 synthetic polymer Polymers 0.000 description 1
- 239000002562 thickening agent Substances 0.000 description 1
- 125000003396 thiol group Chemical group [H]S* 0.000 description 1
- 230000025366 tissue development Effects 0.000 description 1
- 238000013518 transcription Methods 0.000 description 1
- 230000035897 transcription Effects 0.000 description 1
- 238000012546 transfer Methods 0.000 description 1
- RYFMWSXOAZQYPI-UHFFFAOYSA-K trisodium phosphate Chemical compound [Na+].[Na+].[Na+].[O-]P([O-])([O-])=O RYFMWSXOAZQYPI-UHFFFAOYSA-K 0.000 description 1
- 210000004881 tumor cell Anatomy 0.000 description 1
- 230000005740 tumor formation Effects 0.000 description 1
- OUYCCCASQSFEME-UHFFFAOYSA-N tyrosine Natural products OC(=O)C(N)CC1=CC=C(O)C=C1 OUYCCCASQSFEME-UHFFFAOYSA-N 0.000 description 1
- 238000011144 upstream manufacturing Methods 0.000 description 1
- 230000035899 viability Effects 0.000 description 1
- 239000008215 water for injection Substances 0.000 description 1
- 229920003169 water-soluble polymer Polymers 0.000 description 1
- 239000000080 wetting agent Substances 0.000 description 1
- 239000000811 xylitol Substances 0.000 description 1
- 235000010447 xylitol Nutrition 0.000 description 1
- HEBKCHPVOIAQTA-SCDXWVJYSA-N xylitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)CO HEBKCHPVOIAQTA-SCDXWVJYSA-N 0.000 description 1
- 229960002675 xylitol Drugs 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
- A61K38/04—Peptides having up to 20 amino acids in a fully defined sequence; Derivatives thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/0005—Vertebrate antigens
- A61K39/0011—Cancer antigens
- A61K39/001152—Transcription factors, e.g. SOX or c-MYC
- A61K39/001153—Wilms tumor 1 [WT1]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K35/00—Medicinal preparations containing materials or reaction products thereof with undetermined constitution
- A61K35/12—Materials from mammals; Compositions comprising non-specified tissues or cells; Compositions comprising non-embryonic stem cells; Genetically modified cells
- A61K35/14—Blood; Artificial blood
- A61K35/15—Cells of the myeloid line, e.g. granulocytes, basophils, eosinophils, neutrophils, leucocytes, monocytes, macrophages or mast cells; Myeloid precursor cells; Antigen-presenting cells, e.g. dendritic cells
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K35/00—Medicinal preparations containing materials or reaction products thereof with undetermined constitution
- A61K35/12—Materials from mammals; Compositions comprising non-specified tissues or cells; Compositions comprising non-embryonic stem cells; Genetically modified cells
- A61K35/14—Blood; Artificial blood
- A61K35/17—Lymphocytes; B-cells; T-cells; Natural killer cells; Interferon-activated or cytokine-activated lymphocytes
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
- A61K38/04—Peptides having up to 20 amino acids in a fully defined sequence; Derivatives thereof
- A61K38/08—Peptides having 5 to 11 amino acids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
- A61K38/04—Peptides having up to 20 amino acids in a fully defined sequence; Derivatives thereof
- A61K38/10—Peptides having 12 to 20 amino acids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
- A61K38/16—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- A61K38/17—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- A61K38/1703—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans from vertebrates
- A61K38/1709—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans from vertebrates from mammals
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/39—Medicinal preparations containing antigens or antibodies characterised by the immunostimulating additives, e.g. chemical adjuvants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/435—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- C07K14/46—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans from vertebrates
- C07K14/47—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans from vertebrates from mammals
- C07K14/4701—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans from vertebrates from mammals not used
- C07K14/4702—Regulators; Modulating activity
- C07K14/4703—Inhibitors; Suppressors
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/82—Translation products from oncogenes
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/06—Animal cells or tissues; Human cells or tissues
- C12N5/0602—Vertebrate cells
- C12N5/0634—Cells from the blood or the immune system
- C12N5/0639—Dendritic cells, e.g. Langherhans cells in the epidermis
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/06—Animal cells or tissues; Human cells or tissues
- C12N5/0602—Vertebrate cells
- C12N5/0693—Tumour cells; Cancer cells
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01K—ANIMAL HUSBANDRY; CARE OF BIRDS, FISHES, INSECTS; FISHING; REARING OR BREEDING ANIMALS, NOT OTHERWISE PROVIDED FOR; NEW BREEDS OF ANIMALS
- A01K2217/00—Genetically modified animals
- A01K2217/07—Animals genetically altered by homologous recombination
- A01K2217/075—Animals genetically altered by homologous recombination inducing loss of function, i.e. knock out
- A01K2217/077—Animals genetically altered by homologous recombination inducing loss of function, i.e. knock out heterozygous knock out animals displaying phenotype
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01K—ANIMAL HUSBANDRY; CARE OF BIRDS, FISHES, INSECTS; FISHING; REARING OR BREEDING ANIMALS, NOT OTHERWISE PROVIDED FOR; NEW BREEDS OF ANIMALS
- A01K2227/00—Animals characterised by species
- A01K2227/10—Mammal
- A01K2227/105—Murine
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01K—ANIMAL HUSBANDRY; CARE OF BIRDS, FISHES, INSECTS; FISHING; REARING OR BREEDING ANIMALS, NOT OTHERWISE PROVIDED FOR; NEW BREEDS OF ANIMALS
- A01K2267/00—Animals characterised by purpose
- A01K2267/03—Animal model, e.g. for test or diseases
- A01K2267/0331—Animal model for proliferative diseases
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/51—Medicinal preparations containing antigens or antibodies comprising whole cells, viruses or DNA/RNA
- A61K2039/53—DNA (RNA) vaccination
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/545—Medicinal preparations containing antigens or antibodies characterised by the dose, timing or administration schedule
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/555—Medicinal preparations containing antigens or antibodies characterised by a specific combination antigen/adjuvant
- A61K2039/55511—Organic adjuvants
- A61K2039/55566—Emulsions, e.g. Freund's adjuvant, MF59
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/70—Multivalent vaccine
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/80—Vaccine for a specifically defined cancer
- A61K2039/82—Colon
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/40—Regulators of development
- C12N2501/415—Wnt; Frizzeled
Definitions
- the present disclosure relates to a medicament, a method, and the like for preventing or treating a benign tumor.
- the present disclosure relates to a prophylactic or therapeutic agent for familial adenomatous disease.
- a benign tumor is a tumor that has no pathologically malignant findings. Benign tumors are understood to be different from malignant tumors and do not show a tendency to metastasize or invade. Most benign tumors are asymptomatic, but large tumors may show symptoms or become malignant due to compression of other tissues and may require treatment or prevention. is there. For this reason, therapeutic and prophylactic agents are not well known.
- Familial adenomatous polyposis is a hereditary disease with a tumor suppressor gene APC heterozygous deficiency.
- APC heterozygous deletion of the APC gene occurs in gland cells having an APC hetero-deficiency in the large intestine, adenomas develop throughout the large intestine, and colorectal cancer develops from the adenomas.
- the adenoma is removed by endoscopy in the early stage. After that, since the adenomas become too dense to be resected and cancer develops from the adenoma, the large intestine is often removed in many cases. It has a penetrance of 100% and is a very tragic disease that occurs around the age of 20 in all carriers. There are about 6,000 patients in Japan.
- Aspirin is known to be effective in preventing and / or inhibiting the development of adenomas and adenocarcinomas in familial adenomatous polyposis, but the effect is weak and there is a risk of gastrointestinal bleeding as a side effect of aspirin. Accompany.
- Wilms tumor gene WT1 was isolated as a gene involved in tumor formation of Wilms tumor, a childhood renal tumor (see Non-Patent Document 1). This gene encodes a zinc finger transcription factor that is involved in the regulatory mechanisms of cell proliferation and differentiation, as well as apoptosis and tissue development.
- the present inventors have conducted intensive studies and found that the WT1 peptide vaccine suppresses and / or delays the onset of adenoma in benign tumors such as familial adenomatous polyposis and suppresses and / or delays the onset of symptoms from adenoma. It was found that there was no radical treatment method after radical resection, and that it was useful for the treatment and prevention of benign tumors such as familial adenomatous polyposis which are considered intractable. The present inventors have found that adenomas of patients with benign tumors such as familial adenomatous polyposis express the WT1 cancer antigen, so that the WT1 cancer vaccine is not suitable for benign tumors such as familial adenomatous polyposis. The present invention was conceived to be effective in suppressing and / or delaying the onset of adenoma, and in suppressing and / or delaying the onset of symptoms from adenoma.
- the present disclosure is based on the surprising discovery that cells of benign tumor adenomas express WT1 protein.
- WT1 protein is highly expressed in cancer cells of malignant tumors, and the usefulness of WT1 peptide vaccine in malignant tumors could be easily expected.
- the present disclosure has revealed for the first time that cells of adenomas of familial adenomatous polyposis which are benign tumors express WT1 protein, and have come to provide a therapeutic or preventive agent for benign tumors in general. .
- the efficacy of the WT1 peptide vaccine depends on the expression mode (expression amount, etc.) of the WT1 protein, even if it is found that the WT1 protein is expressed in adenoma, the efficacy of the WT1 peptide vaccine is high. Sex could not be reasonably predicted.
- the present disclosure has demonstrated for the first time that the WT1 peptide vaccine is also effective against benign tumors, and has found effects that cannot be predicted from the prior art.
- (Item X1) An agent for preventing or treating a benign tumor, comprising a WT1 peptide or an analog thereof.
- (Item X2) The prophylactic or therapeutic agent according to item X1, wherein the WT1 peptide or an analog thereof includes a killer type and / or a helper type.
- (Item X3) The prophylactic or therapeutic agent according to item X1 or item X2, wherein the WT1 peptide or an analog thereof comprises a WT1 126 killer peptide and / or a WT1 35 helper peptide.
- the preventive or therapeutic agent for benign tumor according to any one of items X1 to X3, comprising a nucleic acid molecule encoding a WT1 peptide or an analog thereof.
- the preventive or therapeutic agent according to any one of items X1 to X7, wherein the benign tumor expresses WT1.
- the benign tumors include familial adenomatous polyposis, non-hereditary colorectal adenoma, intraductal papillary mucinous tumor, meningioma of the brain, schwannomas, epithelial adenoma of each organ, papilloma, non-epithelial
- (Item X10) The preventive or therapeutic agent according to any one of Items X1 to X9, wherein the benign tumor is familial adenomatous polyposis.
- (Item X11) The preventive or therapeutic agent according to any one of items X1 to X10, which is administered once a week.
- (Item X12) A peripheral blood mononuclear cell derived from a subject in need of benign tumor treatment is cultured in the presence of the WT1 peptide or the analog thereof according to any one of the above item X, or any of the above item X.
- CTLs cytotoxic T cell
- Immature dendritic cells from a subject in need of benign tumor treatment are cultured in the presence of the WT1 peptide or an analog thereof according to any one of the above items, or any one of the above item X
- a method for preventing or treating a benign tumor comprising a step of introducing a nucleic acid molecule encoding the WT1 peptide or an analog thereof according to the above item into the immature dendritic cells to induce WT1-presenting dendritic cells.
- (Item X13A) A method according to item X12 or 13, further comprising one or more features according to any one or more of items X1 to X11.
- (Item X14) A composition for inducing WT1-specific CTLs and / or WT1-specific helper T cells for use in prevention or treatment of benign tumors, comprising a WT1 peptide or an analog thereof, or a nucleic acid molecule encoding the same .
- (Item X15) A composition for inducing WT1-presenting dendritic cells for use in prevention or treatment of benign tumors, comprising a WT1 peptide or an analog thereof, or a nucleic acid molecule encoding the same.
- composition according to item X14 or X15 The composition according to item X14 or X15, further comprising one or more characteristics according to any one or more of items X1 to X13.
- composition for preventing or treating benign tumors comprising WT1-specific CTLs and / or WT1-specific helper T cells.
- composition for preventing or treating a benign tumor comprising a WT1-presenting dendritic cell.
- composition according to item X16 or X17 further comprising one or more characteristics according to any one or more of items X1 to X13.
- the present disclosure also provides the following.
- An agent for preventing or treating a benign tumor comprising a WT1 peptide or an analog thereof.
- (Item 2) 2. The prophylactic or therapeutic agent according to item 1, wherein the WT1 peptide or an analog thereof includes a killer type and / or a helper type.
- the WT1 peptide or an analog thereof may be a WT1 126 killer peptide, a WT1 235 killer peptide and / or a WT1 35 helper peptide, or a deletion, substitution, and / or deletion of one to several amino acids in any amino acid sequence.
- the prophylactic or therapeutic agent according to item 1 or 2 comprising a peptide having an added amino acid sequence and having CTL inducing activity.
- the WT1 peptide or an analog thereof RMFPNAPYL (SEQ ID NO: 2), RYFPNAPYL (SEQ ID NO: 46), YMFPNAPYL (SEQ ID NO: 14), CYTWNQMNL (SEQ ID NO: 45), CMTWNQMNL (SEQ ID NO: 3), C-CYTWNQMNL (SEQ ID NO: 47) (where the bond between C and C represents a disulfide bond), and C-CMMTWNQMNL (SEQ ID NO: 48) (where the bond between C and C is a disulfide bond) Or a pharmaceutically acceptable salt thereof, or a peptide consisting of any amino acid sequence selected from 4.
- the preventive or therapeutic agent according to any one of items 1 to 3.
- the WT1 peptide or an analog thereof further comprises the following amino acid sequence: WAPVLDFAPPGASAYGSL (SEQ ID NO: 4), CWAPVLDFAPPGASAYGSL (SEQ ID NO: 50) and WAPVLDFAPPGASAYGSLC (SEQ ID NO: 51), 7.
- the benign tumors include familial adenomatous polyposis, non-hereditary colorectal adenoma, intraductal papillary mucinous tumor, meningioma of the brain, schwannomas, epithelial adenoma of each organ, papilloma, non-epithelial 15.
- a peripheral blood mononuclear cell from a subject in need of benign tumor treatment is cultured in the presence of the WT1 peptide or an analog thereof according to any one of the above items, or any one of the above items.
- a nucleic acid molecule encoding the WT1 peptide or an analog thereof according to the above item is introduced into said peripheral blood mononuclear cells, and WT1-specific cytotoxic T cells (CTLs) and / or WT1-specific cells are obtained from said peripheral blood mononuclear cells.
- CTLs cytotoxic T cells
- a method for inducing WT1-specific CTLs and / or WT1-specific helper T cells for use in the prevention or treatment of benign tumors comprising the step of inducing specific helper T cells.
- Immature dendritic cells from a subject in need of benign tumor treatment are cultured in the presence of the WT1 peptide or an analog thereof according to any one of the above items, or any one of the above items.
- a WT1-presenting tree used for the prevention or treatment of a benign tumor which comprises a step of introducing a nucleic acid molecule encoding the WT1 peptide or an analog thereof according to 1 above into said immature dendritic cells, and inducing a WT1-presenting dendritic cell.
- Method for inducing dendritic cells Item 19A
- Item 20 The method of item 18 or 19, further comprising one or more features of any one or more of items 1-17.
- (Item 20) A composition for inducing WT1-specific CTLs and / or WT1-specific helper T cells for use in prevention or treatment of benign tumors, comprising a WT1 peptide or an analog thereof, or a nucleic acid molecule encoding the same .
- (Item 21) A composition for inducing WT1-presenting dendritic cells for use in prevention or treatment of benign tumors, comprising a WT1 peptide or an analog thereof, or a nucleic acid molecule encoding the same.
- (Item 22) A composition for preventing or treating benign tumors, comprising WT1-specific CTLs and / or WT1-specific helper T cells.
- (Item 23) A composition for preventing or treating a benign tumor, comprising a WT1-presenting dendritic cell.
- (Item 23A) Item 24. The composition of item 22 or 23, further comprising one or more features of any one or more of items 1-19.
- (Item A1) A method of preventing or treating a benign tumor in a subject, comprising administering to the subject an effective amount of a WT1 peptide or an analog thereof.
- (Item A2) The method according to item A1, wherein the WT1 peptide or an analog thereof includes a killer type and / or a helper type.
- the WT1 peptide or an analog thereof may be a WT1 126 killer peptide, a WT1 235 killer peptide and / or a WT1 35 helper peptide, or a deletion, substitution, and / or deletion of one to several amino acids in any amino acid sequence.
- the method according to item A1 or A2 comprising a peptide comprising an added amino acid sequence and having CTL inducing activity.
- the WT1 peptide or an analog thereof further comprises the following amino acid sequence: WAPVLDFAPPGASAYGSL (SEQ ID NO: 4), CWAPVLDFAPPGASAYGSL (SEQ ID NO: 50) and WAPVLDFAPPGASAYGSLC (SEQ ID NO: 51),
- (Item A12) The method according to any one of items A1 to A11, wherein the WT1 peptide or an analog thereof, or a nucleic acid molecule encoding the same, is used in combination with an adjuvant.
- (Item A13) The method of any of the preceding items A1 to A12, wherein the adjuvant is Montanide® ISA51 adjuvant.
- (Item A14) The method according to any one of items A1 to A13, wherein the benign tumor expresses WT1.
- the benign tumors include familial adenomatous polyposis, non-hereditary colorectal adenoma, intraductal papillary mucinous tumor, meningiomas of the brain, schwannomas, epithelial adenoma of each organ, papilloma, non-epithelial
- (Item A17) The method according to any one of items A1 to A16, wherein the WT1 peptide or an analog thereof, or a nucleic acid molecule encoding the WT1 peptide is administered once a week.
- (Item A18) A method for preventing or treating a benign tumor in a subject, the method comprising the steps of: treating a peripheral blood mononuclear cell from a subject in need of benign tumor treatment with the WT1 peptide or the analog thereof according to any one of the above items.
- a nucleic acid molecule encoding a WT1 peptide or an analog thereof according to any one of the above items is introduced into the peripheral blood mononuclear cells, whereby the peripheral blood mononuclear cells are cultured.
- a method for preventing or treating a benign tumor in a subject comprising the step of: treating a subject with immature dendritic cells in need of benign tumor treatment with the WT1 peptide or the analog thereof according to any one of the above items.
- (Item A20) A method of preventing or treating a benign tumor in a subject, comprising providing the subject with WT1-specific CTLs and / or WT1-specific CTLs induced by a WT1 peptide or an analog thereof, or a nucleic acid molecule encoding the same. Administering an effective amount of a helper T cell.
- (Item A21) A method of preventing or treating a benign tumor in a subject, comprising administering to said subject an effective amount of a WT1-presenting dendritic cell induced by a WT1 peptide or an analog thereof, or a nucleic acid molecule encoding the same.
- a method comprising the steps of: (Item A21A) A composition according to item A20 or A21, further comprising one or more features according to any one or more of items A1 to A19.
- (Item A22) A method of preventing or treating a benign tumor in a subject, comprising administering to said subject an effective amount of WT1-specific CTLs and / or WT1-specific helper T cells.
- (Item A23) A method of preventing or treating a benign tumor in a subject, comprising administering to the subject an effective amount of a WT1-presenting dendritic cell.
- (Item B1) A WT1 peptide or an analog thereof for preventing or treating a benign tumor.
- Amino acid sequence a composition comprising a peptide consisting of WAPVLDFAPPGASAYGSL (SEQ ID NO: 4) or a pharmaceutically acceptable salt thereof, WT1 peptide or an analog thereof according to any one of items B1 to B8.
- the benign tumors include familial adenomatous polyposis, non-hereditary colorectal adenoma, intraductal papillary mucinous tumor, meningioma of the brain, schwannomas, epithelial adenoma of each organ, papilloma, non-epithelial WT1 peptide or its analog or nucleic acid molecule according to any one of items B1 to B14, selected from the group consisting of fibroids, lipomas, chondromas, and hemangiomas.
- (Item B16) The WT1 peptide or an analog thereof, or a nucleic acid molecule according to any one of Items B1 to B15, wherein the benign tumor is familial adenomatous polyposis.
- (Item B17) WT1 peptide or analog thereof or a nucleic acid molecule according to any one of items B1 to B16, which is administered once a week.
- (Item B18) WT1 peptide or WT1 peptide according to any one of the preceding items for inducing WT1-specific cytotoxic T cells (CTLs) and / or WT1-specific helper T cells for use in the prevention or treatment of benign tumors.
- CTLs cytotoxic T cells
- WT1-specific helper T cells for use in the prevention or treatment of benign tumors.
- WT1 peptide or analog thereof is cultured with peripheral blood mononuclear cells from a subject in need of treatment for a benign tumor.
- the nucleic acid molecule is introduced into the peripheral blood mononuclear cells, whereby the WT1-specific CTLs and / or WT1-specific helper T cells are induced, or a WT1 peptide or analog thereof, or a nucleic acid molecule.
- a nucleic acid molecule wherein the WT1 peptide or an analog thereof is cultured with immature dendritic cells from a subject in need of treatment for a benign tumor, or the nucleic acid molecule is introduced into the immature dendritic cells.
- a WT1 peptide or an analog thereof, or a nucleic acid molecule from which the WT1-presenting dendritic cells are induced. (Item B19A) The method of claim B18 or B19, further comprising one or more features of any one or more of items B1-B17.
- (Item B20) A WT1 peptide or an analog thereof, or a nucleic acid molecule encoding the same, for inducing WT1-specific CTLs and / or WT1-specific helper T cells for use in preventing or treating benign tumors.
- (Item B21) A WT1 peptide or an analog thereof, or a nucleic acid molecule encoding the same, for inducing WT1-presenting dendritic cells for use in the prevention or treatment of a benign tumor.
- (Item B21A) The composition according to item B20 or B21, further comprising one or more features according to any one or more of items B1-B19.
- (Item C1) Use of a WT1 peptide or an analog thereof in the manufacture of a medicament for preventing or treating a benign tumor.
- the WT1 peptide or an analog thereof may be a WT1 126 killer peptide, a WT1 235 killer peptide and / or a WT1 35 helper peptide, or a deletion, substitution, and / or deletion of one to several amino acids in any amino acid sequence.
- the use according to item C1 or C2 comprising a peptide comprising an added amino acid sequence and having CTL inducing activity.
- the WT1 peptide or an analog thereof further comprises the following amino acid sequence: WAPVLDFAPPGASAYGSL (SEQ ID NO: 4), CWAPVLDFAPPGASAYGSL (SEQ ID NO: 50) and WAPVLDFAPPGASAYGSLC (SEQ ID NO: 51),
- Amino acid sequence a composition comprising a peptide consisting of WAPVLDFAPPGASAYGSL (SEQ ID NO: 4) or a pharmaceutically acceptable salt thereof, Use according to any one of items C1 to C8.
- Items C10 Use of a nucleic acid molecule encoding a WT1 peptide or an analog thereof in the manufacture of a medicament for preventing or treating a benign tumor.
- Items C11 Use according to any of the preceding items C1 to C10, wherein the nucleic acid molecule comprises RNA and / or DNA.
- (Item C12) Use according to any one of the preceding items C1 to C11, wherein the WT1 peptide or its analog or nucleic acid molecule is used in combination with an adjuvant.
- (Item C13) Use according to any one of the above items C1-C12, wherein the adjuvant is Montanide® ISA51 adjuvant.
- (Item C14) The use according to any of items C1-C13, wherein the benign tumor expresses WT1.
- the benign tumors include familial adenomatous polyposis, non-hereditary colorectal adenoma, intraductal papillary mucinous tumor, meningioma of the brain, schwannomas, epithelial adenoma of each organ, papilloma, non-epithelial
- nucleic acid molecule is cultured or the nucleic acid molecule is introduced into the peripheral blood mononuclear cells, whereby the WT1-specific CTLs and / or WT1-specific helper T cells are induced.
- said WT1 peptide or analog thereof is cultured with immature dendritic cells from a subject in need of treatment for benign tumor, or said nucleic acid molecule is introduced into said immature dendritic cells, A WT1 peptide or an analog thereof, or a nucleic acid molecule, from which the WT1-presenting dendritic cells are induced.
- (Item C20) Use of a WT1 peptide or an analog thereof or a nucleic acid molecule encoding the same in the production of WT1-specific CTLs and / or WT1-specific helper T cells for use in prevention or treatment of benign tumors.
- (Item C21) Use of a WT1 peptide or an analog thereof, or a nucleic acid molecule encoding the same in the production of WT1-presenting dendritic cells for use in the prevention or treatment of a benign tumor.
- (Item C21A) The composition according to items C20 or C21, further comprising one or more features according to any one or more of items C1-C19.
- (Item C22) Use of WT1-specific CTLs and / or WT1-specific helper T cells in the manufacture of a medicament for preventing or treating benign tumors.
- (Item C23) Use of a WT1-presenting dendritic cell in the manufacture of a medicament for preventing or treating a benign tumor.
- (Item C23A) The composition according to items C22 or C23, further comprising one or more features according to any one or more of items C1-C19.
- prevention, delay and treatment of benign tumors are achieved.
- the invention achieves prevention, delay and treatment of familial adenomatous polyposis.
- the present invention achieves the prevention, delay and treatment of symptoms arising from adenomas of benign tumors (eg, familial adenomatous polyposis).
- the WT1 peptide cancer vaccine of the present disclosure has no serious side effects other than redness and swelling of the skin at the administration site, and is extremely safe. Therefore, the WT1 peptide cancer vaccine can be safely and easily administered to most benign tumor patients and patients with familial adenomatous polyposis, and it is necessary to avoid endoscopic resection and surgery from the viewpoint of QOL of patients. It can be said that it is superior to the conventional technology also from the viewpoint of the economic effect of reducing medical costs due to.
- FIG. 1 is a photomicrograph showing that WT1 protein was expressed in an adenoma of a patient with human familial adenomatous polyposis.
- the photograph on the left shows a photograph of adenoma tissue, and the photograph on the right shows a photograph of a normal gland duct. In both pictures, the light staining indicates WT1 protein, and the dark and circularly stained areas indicate nuclei.
- FIG. 2 is a micrograph showing the expression of WT1 protein in APC Min / + mice. Microphotographs at 5 ⁇ , 10 ⁇ and 20 ⁇ magnification are shown in order from the top.
- FIG. 3 is a photomicrograph showing WT1 protein expression in APC Min / + mice. The magnification is 40 times, and a scale bar indicating the length of 100 ⁇ m is shown at the lower left of the photograph. Intense staining indicates WT1 protein, circularly stained areas indicate nuclei.
- FIG. 3 is a photomicrograph showing WT1 protein expression in APC Min / + mice. The magnification is 40 times, and a scale bar indicating the length of 100 ⁇ m is shown at the lower left of the photograph. Intense staining indicates WT1 protein, circularly stained areas indicate nuclei.
- FIG. 4 shows the administration scheme of an experiment in which WT1 peptide vaccine was administered to APC Min / + mice.
- the upper horizontal axis shows the age of APC Min / + mice, the time of vaccine administration, and the time of euthanasia and analysis.
- the lower numbers indicate the age of the mice
- the short arrows at the top indicate the time of administration of the vaccine
- the long arrows at the top indicate the time of euthanasia and analysis.
- the compositions of the WT1 vaccine and the control vaccine are shown below the horizontal axis.
- FIG. 5 is a graph showing suppression of adenoma development by administration of a WT1 peptide vaccine.
- FIG. 6 is a graph showing that WT1 tetramer + CD3 + CD8 + T cells were increased by the administration of the WT1 peptide vaccine.
- the vertical axis of the graph represents the frequency of H-2D b WT1 tetramer + CD3 + CD8 + T cells in the CD3 + CD8 + cells
- scatter plot on the left shows the results of APC Min / + mice treated with WT1 peptide vaccine
- the scatter plot on the right shows the results for APC Min / + mice receiving the control vaccine.
- the horizontal line in each scatter plot indicates the average value, and the result of the significance test is shown at the top of the graph.
- FIG. 7 is a graph showing a regression analysis between the WT1 peptide vaccine administration group and the control vaccine administration group.
- the vertical axis of the graph represents the frequency of H-2D b WT1 tetramer + CD3 + CD8 + T cells in the CD3 + CD8 + cells, and the horizontal axis represents the number of adenomas per small intestine.
- Each point is a plot of the number of adenomas and the frequency of WT1 tetramer + CD3 + CD8 + T cells between the WT1 peptide vaccine administration group and the control vaccine administration group on a graph.
- the straight line in the graph indicates a regression line, and the right side shows the correlation coefficient (R), the function formula of the regression line, and the coefficient of determination (R 2 ) in order from the top.
- FIG. 8 shows the administration scheme of an experiment in which WT1 peptide vaccine is administered to APC Min / + mice.
- the upper horizontal axis shows the age of APC Min / + mice, the time of vaccine administration, and the time of euthanasia and analysis.
- the numbers at the bottom indicate the age of the mice
- the short arrows at the top indicate the time of administration of the vaccine
- the long arrows at the top indicate the time of euthanasia and analysis.
- the compositions of the WT1 vaccine and the control vaccine are shown below the horizontal axis.
- FIG. 9 is a photomicrograph showing that WT1 protein was expressed in adenomas of human non-hereditary adenomatous polyposis.
- the first photograph from the left shows a photograph of a normal gland duct
- the three photographs on the right show photographs of adenoma tissue obtained from three non-hereditary adenomatous polyposis patients.
- light staining indicates WT1 protein
- the darkly circular or oval stained areas indicate nuclei.
- the expression level of the WT1 protein is shown below each photograph, (-) indicates that the expression level is low, and (+) indicates that the expression level is high.
- Figure 10 is a APC Min / + mice, indicating the dosing scheme of experiments was administered a mixture containing compounds and WT1 35 peptide represented by the formula (3) herein.
- the horizontal axis indicates the age of APC Min / + mice, the time of administration of the vaccine, and the time of euthanasia and analysis.
- the numbers at the bottom indicate the age of the mice, and the arrows at the top indicate the time when the vaccine was administered and the time when the mice were euthanized and subjected to analysis.
- Figure 11 is a graph showing the inhibition of development of adenomas by administration of a mixture comprising the compound and WT1 35 peptide represented by the formula (3) herein.
- FIG. 12 is a graph showing that WT1 tetramer + CD3 + CD8 + T cells increased by administering a combination comprising a compound and WT1 35 peptide represented by the formula (3) herein.
- the vertical axis of the graph represents the frequency of H-2D b WT1 tetramer + CD3 + CD8 + T cells in the CD3 + CD8 + cells, scatter plot on the left compounds and WT1 35 peptide represented by the formula (3) in the present specification the mixture shows the results of APC Min / + mice treated including, right scatter plot shows the results of APC Min / + mice administered with the control vaccine.
- the horizontal line in each scatter plot indicates the average value, and the result of the significance test is shown at the top of the graph.
- Figure 13 is a graph showing a regression analysis between the compound and mixture treated group and the control vaccine group including a WT1 35 peptide represented by the formula (3) herein.
- the vertical axis of the graph represents the frequency of H-2D b WT1 tetramer + CD3 + CD8 + T cells in the CD3 + CD8 + cells, and the horizontal axis represents the number of adenomas per small intestine.
- Each point is a compound represented by the formula (3) in the present specification and WT1 35 mixture treated group comprising a peptide and adenomas number between the control vaccine group and the WT1 tetramer + CD3 + CD8 + T cells often a on the graph Is plotted in FIG.
- the straight line in the graph indicates a regression line, and the right side shows the correlation coefficient (R), the function formula of the regression line, and the coefficient of determination (R 2 ) in order from the top.
- WT1 Wilms Tumor Gene 1
- WT1 protein WT1 protein
- WT1 peptide includes at least a part (whole) of a Wilms tumor (WT1) gene product or an analog thereof.
- WT1 protein specifically, typically, a human WT1 protein (SEQ ID NO: 1) consisting of 449 amino acids, or one or several (preferably about 2 to Proteins comprising an amino acid sequence in which (6) amino acids have been deleted, substituted and / or added are preferred.
- the inserted or substituted amino acids may be unnatural amino acids other than the 20 gene-encoded amino acids.
- WT1 peptide refers to a peptide consisting of a part of the amino acid sequence constituting the WT1 protein.
- WT1 or “WT1 peptide” includes mutant WT1 unless otherwise specified.
- WT1 or WT1 peptide it refers to human WT1 unless otherwise specified.
- the length of the WT1 peptide used in the present disclosure is not particularly limited, but is preferably one comprising about 7 to about 30 amino acids.
- a preferred WT1 peptide has a regularity (motif) of the sequence of the antigen peptide presented by binding to the HLA molecule and has an ability to bind to the HLA molecule.
- the ability to bind to an HLA molecule can be determined by a method known in the art. Such methods include, for example, computer-based methods such as Rankpep, BIMAS, and SYFPEITHI, and competitive binding tests with known WT1 peptides that have the ability to bind to HLA molecules.
- the WT peptides that can be used in the present disclosure are described in the section (WT1 peptide) in the present specification, and are also described in WO2016 / 093326, which are incorporated by reference.
- WT1 peptides used in the present disclosure are those that activate killer T cells and / or helper T cells.
- the activation of killer T cells and / or helper T cells may be borne by a single peptide, or may be borne by a plurality of peptides (division of labor).
- the WT1 peptide used in the pharmaceutical composition of the present disclosure may be one type or a plurality of types.
- the WT1 peptide used in the medicament or the pharmaceutical composition of the present disclosure may be a killer WT1 peptide, or may be a helper WT1 peptide, or a mixture thereof.
- the WT1 peptide of the present disclosure may be a single peptide, or a conjugate, mixture or combination of a plurality of peptides. More preferred WT1 peptides may include a combination of a killer WT1 peptide and a helper WT1 peptide. For example, examples of such combinations include the compounds or compositions described in WO 2014/157692.
- a “benign tumor” is a tumor that has no pathologically malignant findings, and is understood to be different from a malignant tumor. No metastasis or infiltration. Diagnosis of a benign tumor does not necessarily mean that the clinical prognosis is good. For example, a low-grade meningioma arising in the brainstem is a benign tumor, but is difficult to treat, and because of a poor prognosis due to compression of the brainstem, it is clinically malignant and requires treatment or prevention. There are many.
- Benign tumors include familial adenomatous polyposis, non-hereditary colorectal adenoma, papillary mucinous tumor in the pancreatic duct, meningioma of the brain, schwannomas, epithelial adenoma of each organ, papilloma, non-epithelial , Fibromas, lipomas, chondromas, hemangiomas and the like, but are not limited thereto.
- familial adenomatous polyposis includes diseases accompanied by tumors in tissues other than the intestinal tract, such as Gardner's syndrome.
- Representative animal models of familial adenomatous polyposis include, but are not limited to, APC Min / + mice (Jackson Institute, Bar Harbor, Maine, USA).
- the terms “killer type”, “killer peptide” and the like mean a peptide capable of activating cytotoxic T cells (CTL, killer T cells). Activation of killer T cells refers to an increase in the cytotoxic activity of killer T cells and / or an increase in the number of killer T cells. Whether it is a "killer (type)" is typically determined by the following test. That is, it can be determined by expressing CD8 on the surface of T cells by a technique such as flow cytometry. Alternatively, the determination can be made by measuring the target cytotoxic activity by a 51 Cr release assay, a lactate dehydrogenase (LDH) assay, or the like. In a preferred embodiment, the WT1 peptides of the present disclosure have this killer form of activity.
- MHC is called human leukocyte antigen (HLA) in humans.
- HLAs corresponding to MHC class I molecules are classified into subtypes such as HLA-A, B, Cw, F and G.
- MHC class I restriction preferably includes HLA-A restriction, HLA-B restriction or HLA-Cw restriction.
- HLA-A polymorphism examples include 27 or more such as HLA-A1, HLA-A0201, and HLA-A24
- HLA-B polymorphism examples include HLA-B7, HLA-B40, and HLA-B4403.
- HLA-Cw polymorphisms include 10 or more of HLA-Cw0301, HLA-Cw0401, HLA-Cw0602 and the like. Among these polymorphisms, HLA-A0201 and HLA-A24 are preferable.
- ⁇ The“ WT1 peptide ”in the present disclosure is a partial peptide consisting of consecutive 7 to 30 amino acids in the amino acid sequence of human WT1 shown in SEQ ID NO: 1.
- MHC class I restricted in the present disclosure means a property of binding to MHC class I molecules that are class I of major histocompatibility antigen (Major Histocompatibility complex, MHC) to induce CTL.
- MHC class I-restricted WT1 peptide is a peptide that binds to an MHC class I antigen in vitro and / or in vivo and is presented as a complex, and the result of recognition of the complex by precursor T cells Since it means a peptide that induces CTL, it is synonymous with WT1 helper peptide.
- the amino acid residues of the “WT1 class I-restricted WT1 peptide” are 7 to 30, preferably 7 to 15, more preferably 8 to 12, even more preferably 8 to 11, and most preferably 8 or 9.
- MHC class I-restricted WT1 peptide for example, in the amino acid sequence of human WT1 shown in SEQ ID NO: 1, 2, 3, 4, 7, 7, 10, 17, 18, 20, 23, 24, 26, 29, 30, 32, 33, 37, 38, 39, 40, 47, 63, 64, 65, 70 , 73, 80, 81, 82, 83, 84, 85, 86, 88, 92, 93, 96, 98, 99, 100, 101, 104 , 107, 110, 118, 119, 120, 123, 125, 126, 128, 130, 136, 137, 138, 139, 141, 143, 144, 146, 152, 161, 163, 165, 168 , 169, 174, 177, 179, 180, 185, 187, 191, 192, 194, 202, 204, 206, 207, 208, 209, 210 , 211, 213, 217, 218, 219, 221, 222
- MHC class I-restricted WT1 peptide preferably, the following amino acid sequence: RMFPNAPYL (SEQ ID NO: 2), CMTWNQMNL (SEQ ID NO: 3), ALLPAVPSL (SEQ ID NO: 52), SLGEQQYSV (SEQ ID NO: 53) and RVPGVAPTL (SEQ ID NO: 54) Or a modified amino acid sequence containing a modification of an amino acid residue in any of the amino acid sequences selected from SEQ ID NOs: 2, 3, 52, 53 and 54. And peptides having CTL inducing activity. More preferably, a peptide consisting of any amino acid sequence selected from SEQ ID NOs: 2, 3, 52, 53 and 54 is exemplified.
- the“ WT1 peptide or an analog thereof ”in the present disclosure is a killer peptide, it means a peptide containing a modified amino acid sequence containing a modification of an amino acid residue in the amino acid sequence and having CTL-inducing activity.
- The“ peptide containing a modified amino acid sequence containing a modification of an amino acid residue in the amino acid sequence and having CTL inducing activity ”in the present disclosure is also referred to as“ modified killer peptide ”.
- the modified killer peptide consists of an amino acid sequence in which 1 to 3 amino acids have been deleted, substituted and / or added in the amino acid sequence, and refers to a peptide that binds to MHC class I and induces CTL.
- the substitution position of the amino acid to be substituted includes the 1-position (N-terminal), the 2-position, the 3-position and the 9-position.
- the number of amino acids to be added is usually one to several, preferably one to three, more preferably one or two, and still more preferably one. Preferred addition positions include the C-terminal.
- the number of amino acids deleted is preferably one.
- the added or substituted amino acids may be unnatural amino acids other than the 20 gene-encoded amino acids.
- modified killer peptide examples include the following peptides.
- RYFPNAPYL SEQ ID NO: 46
- RMFPNAPYL SEQ ID NO: 2
- FMFPNAPYL SEQ ID NO: 13
- RLFPNAPYL SEQ ID NO: 18
- RMMPNAPYL SEQ ID NO: 25
- RMFPNAPYV SEQ ID NO: 28
- YMFPNAPYL SEQ ID NO: 14
- CYTWNQMNL SEQ ID NO: 45
- Xaa-Met-Thr-Trp-Asn-Gln-Met-Asn-Leu SEQ ID NO: 55
- amino acid sequence which is not a partial peptide consisting of consecutive amino acids of 8 to 35 residues in the amino acid sequence of human WT1 shown in SEQ ID NO: 1 include the following amino acid sequences (WO 2007/063903) No.).
- C-CYTWNQMNL (SEQ ID NO: 47) (where the bond between C and C represents a disulfide bond) or C-CMWTNQMNL (SEQ ID NO: 48) (where the bond between C and C is a disulfide bond) Represents.).
- the modified killer peptide of the present disclosure also includes, for example, the following dimers as well as multimeric peptides (see WO 2014/157692). Equation (2):
- Equation (3) (Wherein the bond between C and C represents a disulfide bond), Equation (3):
- helper type means a peptide capable of activating helper T cells.
- Activation of helper T cells refers to an increase in the function of helper T cells that assists in B-cell antibody production, activation of killer T cells, and / or an increase in the number of helper T cells.
- helper (type) is typically determined by the following test. That is, it can be determined by expressing CD4 on the surface of T cells by a technique such as flow cytometry. Alternatively, it can be determined by stimulating target cells with an antigen and analyzing the production of cytokines such as IFN- ⁇ and IFN- ⁇ in an antigen-specific manner by immunostaining.
- MHC class II restricted in the present disclosure means the property of binding to MHC class II molecules to induce helper T cells.
- HHLAs corresponding to MHC class II molecules are classified into subtypes such as HLA-DR, DQ and DP.
- MHC class II restriction preferably includes HLA-DR restriction, HLA-DQ restriction or HLA-DP restriction.
- the “MHC class II-restricted WT1 peptide” tp in the present disclosure means a peptide that binds to an MHC class II antigen and induces helper T cells in vitro and / or in vivo.
- the number of amino acid residues of the “MHC class II-restricted WT1 peptide” is 7 to 30, preferably 14 to 30.
- helper peptide in the present disclosure is a helper peptide, it means a peptide that has a modified amino acid sequence containing a modification of an amino acid residue in the amino acid sequence and has a helper T cell inducing activity.
- the modified helper peptide is a peptide comprising an amino acid sequence in which 1 to 3 amino acids have been deleted, substituted and / or added in the amino acid sequence, binds to MHC class II, and induces a helper T cell.
- the number of amino acids added (including insertions) is preferably 1-3.
- the number of amino acids deleted is preferably 1-5.
- the added or substituted amino acids may be non-natural amino acids other than the 20 gene-encoded amino acids.
- modified helper peptide examples include the following peptides.
- SGQAYMFPNAPYLPSCLES SEQ ID NO: 70
- SEQ ID NO: 69 a modified helper peptide of SGQARMFPNAPYLPSCLES
- SEQ ID NO: 69 a modified helper peptide of SGQARMFPNAPYLPSCLES
- SEQ ID NO: 72 SGQAYMFPNAPYLPSC
- PGCNKRYFKLSHLQMHSRK SEQ ID NO: 49
- PGCNKRYFKLSHHLQMHSRKH SEQ ID NO: 62
- CNKRYFKLSHHLQMHSRK SEQ ID NO: 64
- CNKRYFKLSHLMHSRKH SEQ ID NO: 65
- CNKRYFKLSHHLQMHSRKHTG SEQ ID NO: 66
- WAPVLDFAPPGASAYGSL SEQ ID NO: 4
- CWAPVFDPAPPGASAYGSL SEQ ID NO: 4
- the “WT1 peptide or an analog thereof” of the present disclosure also has both a killer type and a helper type activity by forming a composition of the killer peptide and the helper peptide.
- Examples of both killer-type and helper-type active substances include the following. Equation (2):
- Equation (3) (Wherein the bond between C and C represents a disulfide bond), Equation (3):
- Amino acid sequence A composition comprising a peptide consisting of WAPVLDFAPPGASAYGSL (SEQ ID NO: 4) or a pharmaceutically acceptable salt thereof; and formula (3):
- Amino acid sequence A composition comprising a peptide consisting of WAPVLDFAPPGASAYGSL (SEQ ID NO: 4) or a pharmaceutically acceptable salt thereof.
- an adjuvant means an auxiliary agent for a main agent (for example, the WT1 peptide in the present disclosure).
- an adjuvant refers to a substance that enhances or improves the immune response elicited by a WT1 peptide in a therapeutic or prophylactic agent.
- the adjuvant may be, for example, a precipitating adjuvant such as sodium hydroxide, aluminum hydroxide, calcium phosphate, aluminum phosphate, alum, pepeth or a carboxyvinyl polymer, or liquid paraffin, lanolin, Freund, It may be an oily adjuvant such as Montanide @ ISA763AVG, Montanide @ ISA51, incomplete Freund's adjuvant or complete Freund's adjuvant.
- a precipitating adjuvant such as sodium hydroxide, aluminum hydroxide, calcium phosphate, aluminum phosphate, alum, pepeth or a carboxyvinyl polymer, or liquid paraffin, lanolin, Freund, It may be an oily adjuvant such as Montanide @ ISA763AVG, Montanide @ ISA51, incomplete Freund's adjuvant or complete Freund's adjuvant.
- adenoma refers to a polyp having a high risk of canceration.
- Polyp is a general term for mushroom-like and wart-like prominent lesions formed on the inner wall of intestinal tissues such as the small intestine, large intestine, and rectum. Adenomas are neoplastic polyps and have a high risk of changing to cancer such as colorectal cancer.
- Colorectal adenomas include hereditary colorectal adenomas (eg, familial colorectal adenomatosis) and non-hereditary colorectal adenomatosis.
- Non-hereditary adenomatous polyposis refers to a disease in which a large number of adenomas are formed in the intestinal tract due to lifestyle, diet, drinking, smoking, stress, etc., without being characterized by mutations in the APC gene. Classified as benign tumor.
- “treatment ” refers to stopping the progression of a disease, disorder or symptom which is already onset and targeted for the present disclosure, and preferably curing it.
- RECIST New Guidelines for the Evaluation of Therapeutic Effects of Solid Cancer
- the term “therapeutic agent (agent)” broadly refers to any drug that can treat a target condition (eg, a disease such as familial adenomatous polyposis).
- the "therapeutic agent” may be a pharmaceutical composition comprising the active ingredient and one or more pharmacologically acceptable carriers.
- the pharmaceutical composition can be produced, for example, by mixing the active ingredient and the above-mentioned carrier and by any method known in the technical field of pharmaceuticals.
- the form of use of the therapeutic agent is not limited as long as it is used for the treatment, and the active ingredient may be a single active ingredient or a mixture of the active ingredient and an optional ingredient.
- the shape of the carrier is not particularly limited, and may be, for example, a solid or a liquid (for example, a buffer).
- preventing means not causing, or at least delaying, by any means, before the disease, disorder or condition targeted by this disclosure occurs. Or a state in which even if the cause of a disease, disorder or symptom occurs, the cause of the disorder does not occur.
- prophylactic agent broadly refers to any drug that can prevent a target condition (eg, a disease such as familial adenomatous polyposis).
- ameliorating refers to halting or reducing the progression of a disease, disorder or condition of the present disclosure that has already developed, whether completely or partially. Say.
- the term “subject (person)” refers to a target (eg, a human or other organism or a cell, blood, serum, or the like extracted from an organism) to which the prevention or treatment of the present disclosure is applied.
- peripheral blood mononuclear cells refers to mononuclear cells or mononuclear cells including monocytes and lymphocytes isolated from peripheral blood.
- Peripheral blood mononuclear cells include various blood cells such as T cells, B cells, NK cells, monocytes and dendritic cells. By stimulating peripheral blood mononuclear cells, they can be differentiated into cytotoxic T cells, helper T cells, and the like.
- cytotoxic T cells and helper T cells specific to WT1 are referred to as “WT1-specific cytotoxic T cells” and “WT1-specific helper T cells”, respectively.
- WT1-specific cytotoxic T cells and / or WT1-specific helper T cells can be induced by using the WT1 peptide or an analog thereof or a nucleic acid molecule encoding the same as described in the present disclosure. .
- the term "immature dendritic cells” refers to dendritic cells capable of sensitizing a peptide, a cell, or the like to present an antigen to the peptide or the cell. By stimulating immature dendritic cells, they can be differentiated into dendritic cells that present a specific peptide.
- dendritic cells that present WT1 are each referred to as “WT1-presenting dendritic cells”.
- WT1-presenting dendritic cells can be induced by using the WT1 peptide or an analog thereof described herein, or a nucleic acid molecule encoding the same.
- killer T cells can be induced or activated by administering a WT1 peptide to the subject.
- killer T cells may be obtained by, for example, reacting a sample containing lymphocytes from a subject with a complex of a WT1 peptide and an HLA molecule.
- a peripheral blood mononuclear cell derived from a subject may be cultured in the presence of a WT1 peptide, and WT1-specific CTL may be induced from the peripheral blood mononuclear cell.
- an antigen-presenting cell that presents a WT1 peptide via an HLA molecule may be induced by culturing an immature antigen-presenting cell derived from a subject in the presence of a WT1 peptide.
- the immature antigen-presenting cells refer to cells that can mature into antigen-presenting cells, and include immature dendritic cells.
- a WT1 peptide may be added to antigen-presenting cells to activate helper T cells.
- the antigen-presenting cells or killer T cells or helper T cells used in the medicament or composition of the present disclosure may be derived or activated using any WT1 peptide or derivative thereof, or a nucleic acid molecule. .
- Antigen-presenting cells or killer T cells or helper T cells thus induced or activated are administered to a subject, preferably to the subject from which these cells were obtained, to produce a benign tumor (eg, familial adenomatous polyposis) Can be treated and prevented.
- a benign tumor eg, familial adenomatous polyposis
- an “analog”, “derivative”, “analog” or “variant” (such as a WT1 peptide) is preferably, but not intended to be, limited to a protein of interest (eg, (E.g., a WT1 peptide) that includes a region substantially homologous to the WT1 peptide, such molecules are, in various embodiments, aligned over amino acid sequences of the same size or by computer homology programs known in the art.
- This refers to a protein that is the product of modifying the protein by amino acid substitutions, deletions and additions, respectively, whose derivatives still exhibit, but not necessarily to the same degree, the biological function of the original protein. .
- the biological function of such proteins can be determined by suitable and available in vitro assays described herein or known in the art.
- “functionally active” or “functionally active” as used herein refers to a biological activity, such as a biological activity, according to the aspect to which the polypeptides, ie, fragments or derivatives of the disclosure are related. Has a structural, regulatory, or biochemical function of a protein.
- a WT1 peptide fragment is a polypeptide that includes any region of the WT1 peptide, and as long as it functions as the object of the present disclosure (eg, a peptide vaccine), it is not necessarily the biological function of the native WT1 peptide. You don't have to have everything.
- protein polypeptide
- oligopeptide and “peptide” are used interchangeably in the present specification and refer to a polymer of amino acids of any length.
- This polymer may be linear, branched or cyclic.
- Amino acids may be natural or non-natural, and may be modified amino acids.
- the term may also include those assembled into a complex of multiple polypeptide chains.
- the term also includes naturally or artificially modified amino acid polymers. Such modifications include, for example, disulfide bond formation, glycosylation, lipidation, acetylation, phosphorylation or any other manipulation or modification (eg, conjugation with a labeling component).
- amino acid is a general term for organic compounds having an amino group and a carboxyl group.
- amino acid sequence may be chemically modified.
- any amino acid in the amino acid sequence may form a salt or a solvate.
- any of the amino acids in the amino acid sequence may be L-type or D-type.
- the protein according to the embodiment of the present disclosure includes the above “specific amino acid sequence”.
- Examples of the chemical modification of amino acids contained in proteins in vivo include N-terminal modification (eg, acetylation, myristoylation, etc.), C-terminal modification (eg, amidation, glycosylphosphatidylinositol addition, etc.), or side chain Modifications (eg, phosphorylation, addition of sugar chains, etc.) are known.
- Amino acids may be natural or non-natural as long as they satisfy the purpose of the present disclosure.
- polynucleotide As used herein, the terms “polynucleotide”, “oligonucleotide” and “nucleic acid” have the same meaning and refer to a polymer of nucleotides of any length. The term also includes “oligonucleotide derivatives" or “polynucleotide derivatives". The term “oligonucleotide derivative” or “polynucleotide derivative” includes oligonucleotides or polynucleotides containing nucleotide derivatives or unusual bonds between nucleotides, and is used interchangeably.
- oligonucleotide examples include 2′-O-methyl-ribonucleotide, an oligonucleotide derivative in which a phosphoric diester bond in an oligonucleotide is converted to a phosphorothioate bond, and a phosphodiester bond in an oligonucleotide.
- nucleic acid sequence also includes conservatively modified variants (eg, degenerate codon substitutions) and complementary sequences thereof, as well as explicitly stated sequences. Is contemplated. Specifically, degenerate codon substitutions create a sequence in which the third position of one or more selected (or all) codons is replaced with a mixed base and / or deoxyinosine residue. (Batzer et al., Nucleic Acid Res. 19: 5081 (1991); Ohtsuka et al., J. Biol. Chem. 260: 2605-2608 (1985); Rossolini et al., El. Probes 8: 91-98 (1994)).
- degenerate codon substitutions create a sequence in which the third position of one or more selected (or all) codons is replaced with a mixed base and / or deoxyinosine residue.
- nucleic acid is also used interchangeably with gene, cDNA, mRNA, oligonucleotide, and polynucleotide.
- nucleotide may be natural or non-natural.
- the term “gene” refers to a factor that defines a genetic trait, and the term “gene” may refer to “polynucleotide”, “oligonucleotide”, and “nucleic acid”.
- homology of a gene refers to the degree of identity between two or more gene sequences, and generally having “homology” means that the degree of identity or similarity is high.
- the higher the homology of a given two genes the higher the identity or similarity of their sequences.
- Whether the two genes have homology can be determined by direct sequence comparison or, in the case of nucleic acids, a hybridization method under stringent conditions.
- the DNA sequences between the gene sequences are typically at least 50% identical, preferably at least 70% identical, more preferably at least 80%, 90% , 95%, 96%, 97%, 98% or 99% identical, the genes are homologous.
- a “homolog” or “homologous gene product” refers to another species, preferably a mammal, that performs the same biological function as the protein component of the complex described further herein. Preferably, it means a protein in human. Such homologues may also be referred to as “orthologous gene products.” It is understood that such homologs, homologous gene products, orthologous gene products, and the like can be used as long as they meet the purpose of the present disclosure.
- Amino acids may be referred to herein by either their commonly known three letter symbols or by the one-letter symbols recommended by IUPAC-IUB Biochemical Nomenclature Commission. Nucleotides may also be referred to by the generally recognized one-letter code.
- BLAST a tool for sequence analysis, using default parameters.
- the search for the identity can be performed using, for example, NCBI's BLAST 2.7.1 (issued 2017.10.19).
- the value of “identity” usually refers to a value when the above-mentioned BLAST is used and aligned under default conditions.
- Similarity is a numerical value calculated for similar amino acids in addition to identity.
- “several” may be, for example, 10, 8, 6, 5, 4, 3, or 2, or may be less than any of those values. It is known that polypeptides in which one or several amino acid residues have been deleted, added, inserted, or substituted with other amino acids maintain their biological activity (Mark et al., Proc. Natl Acad Sci USA 1984 Sep; 81 (18): 5566-5666., Zoller et al., Nucleic Acids Res. 1982 Oct 25; 10 (20): 6487-6500., Wang et al., Science. 29; 224 (4656): 1431-1433.).
- Deleted proteins can be prepared by, for example, site-directed mutagenesis, random mutagenesis, or biopanning using a protein phage library.
- site-directed mutagenesis method for example, KOD-Plus-Mutagenesis Kit (TOYOBO CO., LTD.) Can be used. It is possible to select a protein having the same activity as that of the wild type from the mutant type protein into which a deletion or the like has been introduced, by performing various characterizations such as FACS analysis and ELISA.
- “70% or more” which is a numerical value of identity or the like is, for example, 70% or more, 75% or more, 80% or more, 85% or more, 90% or more, 95% or more, 96% or more. , 97% or more, 98% or more, 99% or more, or 100%, and may be in the range of any two of the starting values.
- the “identity” is calculated by calculating the ratio of the number of homologous amino acids in two or more amino acid sequences according to a known method as described above.
- the amino acid sequences of the amino acid sequence group to be compared are aligned, and a gap is introduced in a part of the amino acid sequence if necessary to maximize the ratio of the same amino acid.
- Methods for alignment, percentage calculation, comparison methods, and their associated computer programs are well known in the art (eg, BLAST, etc., described above).
- identity and “similarity” can be represented by values measured by BLAST of NCBI unless otherwise specified.
- Blastp can be used as a default algorithm for comparing amino acid sequences with BLAST. The measurement result is quantified as Positives or Identities.
- polynucleotide that hybridizes under stringent conditions refers to well-known conditions commonly used in the art.
- a polynucleotide can be obtained by using a polynucleotide selected from the polynucleotides of the present disclosure as a probe and using a colony hybridization method, a plaque hybridization method, a Southern blot hybridization method, or the like. Specifically, after performing hybridization at 65 ° C. in the presence of 0.7 to 1.0 M NaCl using a filter on which DNA derived from colonies or plaques is immobilized, A polynucleotide which can be identified by washing the filter at 65 ° C.
- SSC serum-sodium citrate
- the composition of a 1 ⁇ concentration SSC solution is 150 mM sodium chloride and 15 mM sodium citrate.
- stringent conditions for example, the following conditions can be adopted.
- sequences containing only the A sequence or only the T sequence are preferably excluded from the sequences that hybridize under stringent conditions. Moderate stringent conditions can be readily determined by those skilled in the art, for example, based on the length of the DNA, and are described in Sambrook et al., Molecular Cloning: Alabourory Manual, Vol. 3, Vol. 1.
- a polypeptide for use in the present disclosure is encoded by a nucleic acid molecule that hybridizes under highly or moderately stringent conditions to a nucleic acid molecule encoding a polypeptide specifically described in the present disclosure. Polypeptides are also included.
- WWT1 peptides of the present disclosure may preferably be “purified” or “isolated”.
- the term “purified” substance or biological agent refers to a substance or biological agent from which at least a part of a factor naturally associated with the substance or biological agent has been removed. .
- the purity of the biological agent in the purified biological agent is higher (ie, more concentrated) than in the state in which the biological agent is normally present.
- the term “purified” as used herein preferably refers to at least 75%, more preferably at least 85%, even more preferably at least 95%, and most preferably at least 98% by weight of It means that the same type of biological factor is present.
- the substance or biological agent used in the present disclosure is preferably a "purified” substance.
- an “isolated” substance or biological agent eg, a nucleic acid or protein, etc.
- isolated does not necessarily have to be expressed in purity, as it will vary depending on its purpose, but if necessary, preferably at least 75% by weight, more preferably Means that at least 85%, even more preferably at least 95%, and most preferably at least 98% by weight of the same type of biological agent is present.
- the substance used in the present disclosure is preferably an "isolated" substance or biological agent.
- fragment refers to a polypeptide or polynucleotide having a sequence length of 1 to n ⁇ 1 with respect to a full-length polypeptide or polynucleotide (length is n).
- length is n
- the length of the fragment can be appropriately changed depending on the purpose. For example, in the case of a polypeptide, the lower limit of the length is 3, 4, 5, 6, 7, 8, 9, 10, 15, 20, 25, 30, 40, 50 and more amino acids, and lengths represented by integers not specifically recited herein (eg, 11 and the like) are also suitable as lower limits. obtain.
- nucleotides of 5, 6, 7, 8, 9, 10, 15, 20, 25, 30, 40, 50, 75, 100 and more nucleotides can be mentioned.
- a length represented by a non-integer integer eg, 11, etc.
- such a fragment falls within the scope of the present disclosure, for example, when a full-length fragment functions as a cancer vaccine, as long as the fragment itself also has a function as a cancer vaccine. Is done.
- biological function refers to a specific function that a gene, nucleic acid molecule or polypeptide can have in vivo or in vitro when referring to a gene or a nucleic acid molecule or polypeptide related thereto. This includes, but is not limited to, for example, activation of killer T cells or helper T cells.
- a biological function may be performed by a corresponding “biological activity”.
- biological activity refers to an activity that a certain factor (eg, a polynucleotide, a protein, etc.) may have, and various functions (eg, activity of a killer T cell or a helper T cell). ) Are included.
- the “biological activity” may be an activity exerted in a living body or an activity exerted outside a living body by secretion or the like.
- a factor is an enzyme
- its biological activity includes that enzyme activity.
- Such a biological activity can be measured by techniques well known in the art.
- “activity” indicates or reveals binding (either directly or indirectly); affects response (ie, has a measurable effect in response to some exposure or stimulus); Refers to various measurable indices, such as the affinity of a compound for binding directly to a polypeptide or polynucleotide of the disclosure, or the amount of upstream or downstream proteins or some other Similar functional measures may also be included.
- “ expression ”of a gene, a polynucleotide, a polypeptide, or the like means that the gene or the like undergoes a certain action in vivo to take another form.
- it means that a gene, a polynucleotide, or the like is transcribed and translated to form a polypeptide, but transcription is also an embodiment of expression of mRNA.
- the term "expression product” as used herein includes such a polypeptide or protein, or mRNA. More preferably, such forms of the polypeptide may have undergone post-translational processing.
- the expression level of WT1 can be determined by any method.
- the expression level of WT1 can be known by evaluating the amount of WT1 mRNA, the amount of WT1 protein, and the biological activity of WT1 protein.
- the amount of WT1 mRNA or protein can be determined by methods as detailed elsewhere herein or by other methods known in the art.
- a functional equivalent of the “WT1 peptide” of the present disclosure does not have the same sequence as SEQ ID NO: 1, but is a mutant or variant thereof (eg, an amino acid sequence variant or the like), Those that have the biological action of the WT1 peptide, and those that can change to the mutant or variant having the biological action of the WT1 peptide at the time of action (for example, (Encoding nucleic acids, and vectors, cells, etc. containing the nucleic acids) are understood to be encompassed.
- the biological search include stringent hybridization, a macroarray in which genomic DNA is attached to a nylon membrane or the like, a microarray in which a glass plate is attached (microarray assay), PCR, and in situ hybridization. It is not limited to. In the present specification, it is intended that the gene used in the present disclosure should also include a corresponding gene identified by such electronic search and biological search.
- insertion, substitution or deletion of one or more amino acids, or addition of one or more amino acids to one or both terminals in an amino acid sequence can be used.
- “insertion, substitution, or deletion of one or more amino acids in an amino acid sequence, or addition to one or both ends thereof” refers to a well-known technical technique such as site-directed mutagenesis. It means that the modification has been made by a method or by a natural mutation, such as by substitution of a plurality of amino acids to the extent that can occur naturally.
- the modified amino acid sequence is, for example, insertion, substitution, and / or deletion of 1 to 4, preferably 1 to 3, particularly preferably 1 to 2, or 1 amino acid, or addition to one or both terminals.
- the modified amino acid sequence preferably has one or more (preferably one or several or 1, 2, 3, or 4) conservative substitutions in the amino acid sequence of any of SEQ ID NOs: 1 to 5. May be an amino acid sequence having
- “conservative substitution” means that one or more amino acid residues are substituted with another chemically similar amino acid residue so as not to substantially alter the function of the protein. For example, a case where a certain hydrophobic residue is substituted with another hydrophobic residue, a case where a certain polar residue is substituted with another polar residue having the same charge, and the like can be mentioned. Functionally similar amino acids that can make such substitutions are known in the art for each amino acid.
- non-polar (hydrophobic) amino acids include alanine, valine, isoleucine, leucine, proline, tryptophan, phenylalanine, methionine and the like.
- Polar (neutral) amino acids include glycine, serine, threonine, tyrosine, glutamine, asparagine, cysteine, and the like.
- positively charged (basic) amino acids include arginine, histidine, and lysine.
- negatively charged (acidic) amino acids include aspartic acid and glutamic acid.
- WT1 peptides include peptides derived from the WT1 protein that induce the activation of cytotoxic T cells or helper T cells.
- WT1 protein examples include a WT1 killer peptide that induces activation of cytotoxic T cells and a WT1 helper peptide that induces activation of helper T cells.
- WT1 killer peptide examples include a peptide consisting of 8 to 12 amino acids derived from the WT1 protein, and preferably a peptide consisting of 8 to 9 amino acids.
- the WT1 126 peptide Arg Met Phe Pro Asn Ala Pro Tyr Leu (SEQ ID NO: 2), or the WT1 235 peptide: Cys Met Thr Trp Asn Gln Met Asn Leu (SEQ ID NO: 3), or the formula (3)
- WT1 helper peptide examples include a peptide consisting of 14 to 20 amino acids derived from the WT1 protein, and preferably a peptide consisting of 16 to 18 amino acids.
- WT1 35 peptide (Trp Ala Pro Val Leu Asp Phe Ala Pro Pro Gly Ala Ser Ala Tyr Gly Ser Leu; SEQ ID NO: 4), or WT1 332 peptide: Lys Arg Tyr Phe Lys Leu Ser His Leu Gln Met His Ser Arg Lys His) (SEQ ID NO: 5).
- a modified peptide in which one or several amino acids have been deleted, substituted or added in the WT1 peptide can also be used as the WT1 peptide in the present disclosure.
- modified peptides As such modified peptides, WT1 126 peptide modified peptide, WT1 235 peptide modified peptides, modified peptides of WT1 35 peptides, and WT1 332 peptide modified peptides.
- a modified peptide of the WT1 126 peptide a peptide in which the 4th to 8th amino acid residues from the N-terminal are the same as the 4th to 8th amino acid residues from the N-terminal of the WT1 126 peptide (PNAPY) is preferable.
- PNAPY 4th to 8th amino acid residues from the N-terminal of the WT1 126 peptide
- a modified peptide of the WT1 126 peptide a peptide having an amino acid sequence represented by any of the following SEQ ID NOs: 6 to 44 is preferable.
- WT1 126 P1G peptide (GMFPNAPYL; SEQ ID NO: 6) WT1 126 P1A peptide (AMPFPNAPYL; SEQ ID NO: 7) WT1 126 P1V peptide (VMFPNAPYL; SEQ ID NO: 8) WT1 126 P1L peptide (LMFPNAPYL; SEQ ID NO: 9) WT1 126 P1I peptide (IMFPNAPYL; SEQ ID NO: 10) WT1 126 P1M peptide (MMFPNAPYL; SEQ ID NO: 11) WT1 126 P1W peptide (WMFPNAPYL; SEQ ID NO: 12) WT1 126 P1F peptide (FMFPNAPYL; SEQ ID NO: 13) WT1 126 P1Y peptide (YMFPNAPYL; SEQ ID NO: 14) WT1 126 P2V peptide (RVFPNAPYL; S
- WT1 235 peptide of the modified peptide WT1 235m peptide, HLA-A * 24: 02 ( Japanese most of the HLA type) are restricted WT1 peptide, a high therapeutic efficacy than the WT1 235 peptide of wild-type, It is particularly preferable because of its excellent solubility in water.
- modified peptides include the following. Cys-Cys Tyr Thr Trp Asn Gln Met Asn Leu (SEQ ID NO: 47) (wherein the bond between C and C represents a disulfide bond), or Cys-CysMet Thr Trp Asn Gln Met As No. 48) (wherein the bond between C and C represents a disulfide bond), which is particularly excellent in physicochemical properties and stability by modifying the thiol group of the N-terminal cysteine residue (International Publication No. 2007/063903).
- the modified peptide of the WT1 peptide in the present embodiment preferably includes a plurality of peptides corresponding to different HLA subtypes.
- the pharmaceutical composition has the formula (1):
- a tumor antigen peptide A has the following amino acid sequence: RMFPNAPYL (SEQ ID NO: 2), ALLPAVPSL (SEQ ID NO: 52), SLGEQQYSV (SEQ ID NO: 53), RVPGVAPTL ( SEQ ID NO: 7), a peptide consisting of any amino acid sequence selected from YMFPNAPYL (SEQ ID NO: 14) and VLDFAPPGA (SEQ ID NO: 68), wherein the carbonyl group of the C-terminal amino acid of tumor antigen peptide A has the formula (1 ) Binds to the hydroxyl group in R 1 represents a hydrogen atom or a tumor antigen peptide B;
- the tumor antigen peptide B is a peptide having a sequence different from that of the tumor antigen peptide A and consisting of any one of the following amino acid sequences: CMWTNQMNL (SEQ ID NO: 3) and CYTWNQMNL
- the compound represented by the above formula (1) is excellent in stability against an oxidizing agent or the like in a solution due to, for example, a cysteine residue forming a disulfide bond, and has a certain quality as a drug material. .
- the modified peptide of the WT1 peptide in the present embodiment includes the compound represented by the above formula (1) (a conjugate of the WT1 killer peptide), the disulfide bond between the N-terminal cysteine residues by EERA1 in the body.
- Reductive cleavage cleaves the conjugate and produces two epitopes corresponding to different HLA subtypes.
- a conjugate in which a plurality of types of epitopes corresponding to different HLA subtypes are generated in the body, such as the conjugate represented by the formula (1), can widely correspond to different HLA subtypes depending on the subject, and has a large population.
- the “tumor antigen peptide A” in the present embodiment is an MHC class I restricted WT1 peptide consisting of 7 to 30 amino acids.
- the amino group of the N-terminal amino acid binds to Ya in formula (1), and the carbonyl group of the C-terminal amino acid binds to the hydroxyl group in formula (1).
- the compound represented by the formula (1) is represented by the formula (2):
- the modified peptide of WT1 35 peptide in the amino acid sequence shown in SEQ ID NO: 4, one or several amino acids are not particularly limited as long substituted or deletions or added in the amino acid sequence.
- the modified peptide of the WT1 332 peptide is not particularly limited as long as one or several amino acids are substituted, deleted or added in the amino acid sequence shown in SEQ ID NO: 5.
- WT1 126 P1F peptide SEQ ID NO: 13
- WT1 126 P2L peptide SEQ ID NO: 18
- WT1 126 P3M peptide SEQ ID NO: 25
- WT1 126 P9V peptide SEQ ID NO: 28
- WT1 126 P2L peptide, WT1 126 P3M peptide or WT1 126 P9V peptide is more preferred
- WT1 126 P9V peptide is even more preferred.
- WT1 126 peptide in prophylactic or therapeutic agents of the present disclosure, WT1 126 peptide, WT1 126 P1F peptide, WT1 126 P2L peptide, WT1 126 P3M peptide or WT1 126 P9V peptide is preferred. More preferably, it is WT1 126 peptide, WT1 126 P2L peptide, WT1 126 P3M peptide or WT1 126 P9V peptide, further preferably WT1 126 peptide, or WT1 126 P9V peptide, and particularly preferably WT1 126 peptide.
- a derivative of the WT1 peptide can also be used as the WT1 peptide.
- derivatives of WT1 126 peptide, WT1 235 peptide, WT1 35 peptide or WT1 332 peptide include various substances at the N-terminal and / or C-terminal of the amino acid sequence consisting of 9, 16 or 18 consecutive amino acids. And the like.
- amino acids, peptides, analogs thereof, and the like may be bound.
- WT1 126 peptide, WT1 235 peptide, WT1 35 peptide or WT1 332 peptide, or modified peptides thereof these substances may be, for example,
- the WT1-specific CTL reaction is treated by an in vivo enzyme or the like or by a process such as intracellular processing to finally produce a peptide consisting of the above 9, 16, or 18 amino acids and to be displayed on the cell surface. Can be withdrawn.
- WT1 peptide or its analog in this embodiment may further include a WT1 helper peptide.
- the WT1 peptide or an analog thereof is CNKRYFKLSHHLQMHSRK (SEQ ID NO: 63), CNKRYFKLSHHLQMHSRKH (SEQ ID NO: 64), CNKRYFKLSHLQMHSRKHTG (SEQ ID NO: 65), WAPVLDPAPPGASAYGSLAPGAPSYGAP, and SEQ ID NO. No. 51) further includes a peptide containing another amino acid sequence selected from the above group and / or another WT1 helper peptide other than the above. May be.
- WT1 peptides can be produced by methods commonly used in the art. For example, Peptide Synthesis, Interscience, New York, 1966; The Proteins, Vol 2, Academic Press Inc. ⁇ New York, 1976; ⁇ Peptide Synthesis, Maruzen Co., Ltd., 1975; Basics and Experiments of Peptide Synthesis, Maruzen Co., Ltd. 1985; Development of Pharmaceuticals, Continued, Vol. 14, Peptide Synthesis, Hirokawa Shoten, 1991, etc. It can be synthesized by a peptide synthesis method.
- Methods for screening WT1 peptides and modified peptides include, for example, stimulation of only one peptide using PBMCs (peripheral blood mononuclear cells) from some patients with benign tumors (eg, familial adenomatous polyposis).
- PBMCs peripheral blood mononuclear cells
- benign tumors eg, familial adenomatous polyposis.
- the method of performing an IFN ⁇ assay and selecting a peptide having a good reaction is preferred because it is simple.
- a polynucleotide such as DNA or RNA encoding the above WT1 protein or WT1 peptide can also be used as an active ingredient of a prophylactic or therapeutic agent. That is, by inserting a polynucleotide encoding a WT1 protein or a WT1 peptide into an appropriate vector, preferably an expression vector, and then administering it to animals including humans, cancer immunity can be generated in vivo.
- an appropriate vector preferably an expression vector
- cancer immunity can be generated in vivo.
- the polynucleotide include DNA and RNA, and DNA or RNA is preferable.
- the nucleotide sequence of the polynucleotide can be determined based on the amino acid sequence of a WT1 protein or WT1 peptide that is immunogenic for a patient with a benign tumor (eg, familial adenomatous polyposis).
- the polynucleotide can be produced by, for example, a known DNA or RNA synthesis method, a PCR method, or the like.
- a prophylactic or therapeutic agent containing a DNA encoding the WT1 protein or WT1 peptide is also one of the present disclosures.
- the WT1 protein or WT1 peptide is preferably a WT1 peptide, more preferably a WT1 126 peptide, WT1 235 peptide, WT1 35 peptide or WT1 332 peptide, or a modified peptide thereof, or a combination thereof, and more preferably WT1 126 peptide, WT1 235 peptide or WT1.
- the expression vector into which the DNA is inserted is not particularly limited.
- the RNA can be used as an active ingredient of the composition without inserting it into a vector.
- the prophylactic or therapeutic agent of the present disclosure can contain an adjuvant.
- an adjuvant when a WT1 protein or WT1 peptide serving as an antigen is administered, if administered together with or separately from the WT1 protein or WT1 peptide, any substance that nonspecifically enhances an immune response to the antigen can be used. Not limited.
- Adjuvants include, for example, sedimentation adjuvants or oily adjuvants.
- Precipitating adjuvants include, for example, sodium hydroxide, aluminum hydroxide, calcium phosphate, aluminum phosphate, alum, pepeth or carboxyvinyl polymers.
- the oil adjuvant is preferably one that can form micelles by wrapping an aqueous solution of the antigen with oil, and specific examples include liquid paraffin, lanolin, Freund, Montanide ISA763AVG, Montanide ISA51, incomplete Freund's adjuvant, or complete Freund's adjuvant.
- Can be Adjuvants can be used as a mixture of two or more. Preferably, it is an oily adjuvant.
- the amount of adjuvant in the preventive or therapeutic agent of the present disclosure is not particularly limited as long as it is an amount that nonspecifically enhances an immune response to an antigen, and may be appropriately selected depending on the type of adjuvant and the like.
- the prophylactic or therapeutic agent of the present disclosure can be administered orally or parenterally, for example, intraperitoneally, subcutaneously, intradermally, intramuscularly, intravenously or intranasally.
- a prophylactic or therapeutic agent is applied to the skin or a patch containing the prophylactic or therapeutic agent is applied to the skin, whereby the active ingredient WT1 protein or WT1 peptide is applied.
- An administration method for skin absorption is also included.
- the prophylactic or therapeutic agent of the present disclosure can be administered by inhalation or the like.
- it is administered by parenteral administration. More preferably, it is administered by intradermal or subcutaneous administration.
- the body part to be administered intradermally or subcutaneously is preferably, for example, the upper arm.
- the prophylactic or therapeutic agent of the present disclosure can take various formulation forms depending on the administration route, for example, a solid formulation, a liquid formulation and the like.
- it can be a solid preparation or a liquid preparation for oral administration or an injection for parenteral administration.
- Examples of the solid preparation for oral administration for oral administration include tablets, pills, capsules, powders, and granules.
- the WT1 protein or WT1 peptide may be used as it is, or may be mixed with an additive or granulated (eg, stirring granulation, fluidized bed granulation, dry granulation, tumbling stirred fluidized bed granulation). Etc.) and manufactured according to a conventional method.
- an additive or granulated eg, stirring granulation, fluidized bed granulation, dry granulation, tumbling stirred fluidized bed granulation.
- Etc. granulated
- capsules can be manufactured by filling capsules, and tablets can be manufactured by tableting.
- One or more additives may be appropriately blended.
- additives include excipients such as lactose, mannitol, glucose, microcrystalline cellulose, and corn starch; binders such as hydroxypropyl cellulose, polyvinylpyrrolidone, and magnesium aluminate metasilicate; dispersants such as corn starch; Disintegrants such as calcium glycolate; lubricants such as magnesium stearate; dissolution aids such as glutamic acid and aspartic acid; stabilizers; celluloses such as hydroxypropylcellulose, hydroxypropylmethylcellulose and methylcellulose; polyethylene glycol and polyvinylpyrrolidone Water-soluble polymers such as synthetic polymers such as water and polyvinyl alcohol; sucrose, powdered sugar, sucrose, fructose, glucose, lactose, reduced maltose water, powdered reduced maltose water, glucose fructose liquid sugar Fructose-glucose liquid sugar, honey, sorbitol, maltitol, mannito
- the granules or tablets may be coated with a coating agent or the like, if necessary, and the coating may be composed of two or more layers.
- the coating agent include sucrose, gelatin, hydroxypropylcellulose, hydroxypropylmethylcellulose phthalate and the like.
- the above-mentioned excipients are appropriately selected, uniformly mixed or granulated with pranlukast hydrate, or granulated or coated with an appropriate coating agent to form a capsule.
- glycerin or sorbitol may be added to a suitable capsule base (eg, gelatin) to form a capsule with increased plasticity.
- Coloring agents or preservatives can be added to these capsule bases as necessary.
- Capsules include hard capsules or soft capsules.
- liquid preparations for oral administration include liquid preparations, suspensions / emulsions, syrup preparations, dry syrup preparations and the like, and elixir preparations.
- the WT1 protein or WT1 peptide is dissolved, suspended or emulsified in a diluent generally used in the liquid medicine for internal use.
- the diluent include purified water, ethanol, and a mixed solution thereof.
- this liquid preparation may contain a wetting agent, a suspending agent, an emulsifying agent, a sweetening agent, a flavoring agent, a fragrance, a preservative or a buffering agent and the like.
- the dry syrup can be produced by mixing, for example, pranlukast hydrate with, for example, sucrose, powdered sugar, sucrose, fructose, glucose or lactose. Further, the dry syrup may be granulated according to a conventional method.
- dosage forms for parenteral administration include injections, ointments, gels, creams, patches, sprays, sprays and the like, with injections being preferred.
- injections it is preferable to make an injection with WT1 protein or WT1 peptide and a conventional carrier.
- the injection for parenteral administration may be either an aqueous injection or an oil injection.
- an aqueous injection according to a known method, for example, after mixing a WT1 protein or a WT1 peptide with a solution obtained by appropriately adding a pharmaceutically acceptable additive to an aqueous solvent (water for injection, purified water, or the like), It can be prepared by filtering and sterilizing with a filter or the like, and then filling in a sterile container.
- Pharmaceutically acceptable additives include, for example, the above-mentioned adjuvants; isotonic agents such as sodium chloride, potassium chloride, glycerin, mannitol, sorbitol, boric acid, borax, glucose, propylene glycol; phosphate buffer; Buffers such as acetate buffer, borate buffer, carbonate buffer, citrate buffer, Tris buffer, glutamate buffer, epsilon aminocaproic acid buffer; methyl paraoxybenzoate, ethyl paraoxybenzoate, propyl paraoxybenzoate Preservatives such as butyl, parahydroxybenzoate, chlorobutanol, benzyl alcohol, benzalkonium chloride, sodium dehydroacetate, sodium edetate, boric acid, borax; hydroxyethyl cellulose, hydroxypropyl cellulose, polyvinyl alcohol, polyethylene Thickeners such as recohol; stabilizers such as sodium bisulfite, sodium thiosul
- Injectables include suitable solubilizers, for example, alcohols such as ethanol; polyalcohols such as propylene glycol and polyethylene glycol; nonionic surfactants such as polysorbate 80, polyoxyethylene hydrogenated castor oil 50, lysolecithin, and pluronic polyol.
- An agent or the like may be further added.
- proteins such as bovine serum albumin and keyhole limpet hemocyanin; and polysaccharides such as aminodextran may be contained.
- sesame oil or soybean oil is used as the oily solvent, and benzyl benzoate or benzyl alcohol may be added as a solubilizing agent.
- the prepared injection is usually filled in a suitable ampoule or vial.
- Liquid preparations such as injections can be preserved by removing water by cryopreservation or lyophilization.
- the lyophilized preparation is used after reconstitution by adding distilled water for injection or the like at the time of use.
- a WT1 protein or a WT1 peptide is mixed into a liposome, and further, if necessary, other components incorporated in a polysaccharide and / or a cancer vaccine composition. It can also be contained.
- the amount of the WT1 peptide is preferably about 0.1 ⁇ g to 1 mg / kg per body weight per day.
- the dose of the WT1 peptide is usually 0.0001 mg to 1000 mg, preferably 0.01 mg to 1000 mg, more preferably 0.1 mg to 10 mg, and it is preferable to administer this amount once every several days to several months. .
- PBMCs are collected from peripheral blood of a patient with a benign tumor (eg, familial adenomatous polyposis), and dendritic cells are extracted therefrom.
- a method of pulsing a peptide such as a WT1 126 peptide, a WT1 235 peptide, a WT1 35 peptide or a WT1 332 peptide, or a polynucleotide such as DNA or RNA which is contained as an active ingredient in a drug and returning the patient to the patient by subcutaneous administration or the like is also available.
- the conditions for pulsing dendritic cells with the WT1 peptide or the like are not particularly limited as long as the effects of the present disclosure are exerted, and ordinary conditions can be employed.
- a nucleic acid molecule encoding a WT1 protein or WT1 peptide is used as a prophylactic or therapeutic agent
- the nucleic acid molecule is prevented or introduced into a dendritic cell of a patient with a benign tumor (eg, familial adenomatous polyposis).
- a therapeutic agent is administered.
- Methods for introducing a nucleic acid molecule into dendritic cells of a patient with a benign tumor include, for example, as described above, a patient from a benign tumor (eg, familial adenomatous polyposis).
- introducing a nucleic acid molecule into the dendritic cell by an electric pulse introducing a nucleic acid molecule into the dendritic cell by an electric pulse.
- the dendritic cell pulsed with the nucleic acid molecule can be used to convert a benign tumor (eg, familial adenomatous polyposis). By returning the patient to the body of (1), cancer immunity can be quickly generated in the living body.
- a method for treating or preventing cancer in which a nucleic acid molecule encoding a WT1 protein or WT1 peptide is introduced into dendritic cells of a subject is one of the preferred embodiments of the present disclosure.
- the nucleic acid molecule may be any of DNA and RNA, and is preferably RNA.
- Another aspect of the disclosure is culturing peripheral blood mononuclear cells from a subject in the presence of a WT1 protein or WT1 peptide, or introducing a nucleic acid molecule encoding them into the peripheral blood mononuclear cells.
- the present invention relates to a method for inducing WT1-specific CTLs and / or WT1-specific helper T cells by inducing WT1-specific CTLs and / or WT1-specific helper T cells from the peripheral mononuclear cells.
- the subject from which peripheral blood mononuclear cells are derived is not particularly limited.
- WT1 protein or WT1 peptide examples include WT1 126 peptide, WT1 235 peptide, WT1 35 peptide or WT1 332 peptide, and modified peptides thereof, and WT1 126 peptide, WT1 235 peptide, WT1 35 peptide or WT1 332 peptide are preferred. is there.
- WT1-specific CTLs are induced from CTL precursor cells in peripheral blood mononuclear cells by culturing peripheral blood mononuclear cells from a subject in the presence of WT1 126 peptide or WT1 235 peptide.
- WT1-specific helper T cells are induced from helper T cell precursor cells in peripheral blood mononuclear cells.
- the culture conditions for the peripheral blood mononuclear cells derived from the subject are not particularly limited, and they can be cultured under ordinary conditions.
- the CTLs and helper T cells thus obtained recognize WT1 126 peptide, WT1 235 peptide, WT1 35 peptide and WT1 332 peptide, respectively.
- WT1-specific CTLs and / or WT1-specific helper T cells induced according to the present disclosure can specifically injure WT1-high expressing tumor cells and benign tumor tumors (eg, familial tumors) Adenomatous polyposis) can be treated and / or prevented.
- the method of administering WT1-specific CTLs and / or WT1-specific helper T cells to a subject is not particularly limited, and for example, can be administered in the same manner as the above-described prophylactic or therapeutic agent.
- kits for inducing WT1-specific CTLs and / or WT1-specific helper T cells comprising a WT1 protein or WT1 peptide as an essential component.
- the kit is used for the method of inducing WT1-specific CTLs and / or WT1-specific helper T cells from the subject.
- a kit may include, for example, a means for obtaining peripheral blood mononuclear cells, an adjuvant, a reaction container, and the like, in addition to the WT1 protein or the WT1 peptide.
- WT1-specific CTLs and / or WT1-specific helper T cells that recognize cancer antigens such as WT1 126 peptide, WT1 235 peptide, WT1 35 peptide or WT1 332 peptide. Can be.
- immature dendritic cells from a subject are cultured in the presence of a WT1 protein or WT1 peptide, or by introducing a nucleic acid molecule encoding them into the immature dendritic cells. And a method for inducing dendritic cells presenting the WT1 protein or WT1 peptide from the immature dendritic cells, and inducing dendritic cells presenting the WT1 protein or WT1 peptide.
- WT1 protein or WT1 peptide examples include WT1 126 peptide, WT1 235 peptide, WT1 35 peptide or WT1 332 peptide or a modified peptide thereof, and WT1 126 peptide, WT1 235 peptide, WT1 35 peptide or WT1 332 peptide are preferable. .
- nucleic acid molecule encoding the WT1 protein or WT1 peptide examples include those encoding WT1 126 peptide, WT1 235 peptide, WT1 35 peptide or WT1 332 peptide or a modified peptide thereof, and include WT1 126 peptide, WT1 235 peptide, WT1 35 Those encoding the peptide or the WT1 332 peptide are preferred. Any of DNA and RNA may be used as the nucleic acid molecule, and RNA is preferred.
- the subject from which the immature dendritic cells are derived is not particularly limited.
- immature dendritic cells are contained in, for example, peripheral blood mononuclear cells, such cells may be cultured in the presence of WT1 126 peptide, WT1 235 peptide, WT1 35 peptide, or WT1 332 peptide.
- WT1 126 peptide WT1 235 peptide
- WT1 35 peptide WT1 35 peptide
- WT1 332 peptide WT1 332 peptide.
- the present disclosure provides an agent for preventing or treating a benign tumor, comprising a WT1 peptide or an analog thereof.
- WT1 was shown to be highly expressed in cancer cells of malignant tumors, but its expression in benign tumors was unknown.
- the present disclosure is based on the surprising discovery that WT1 protein is expressed in cells of benign tumors and provides a new treatment for benign tumors.
- a benign tumor of the present disclosure expresses WT1.
- the WT1 peptide of the present disclosure acts as a cancer antigen and enhances the cytotoxic activity of CTLs and / or the activity of helper T cells, thereby causing cytotoxicity to cells of benign tumors. Due to their activity, the WT1 peptide vaccines of the present disclosure have therapeutic efficacy against WT1-expressing benign tumors.
- the expression of the WT1 protein is not limited to specific benign tumors, but is observed in various hereditary and non-hereditary benign tumors.
- WT1 protein may be expressed not only in hereditary but also acquired benign tumors, and such types of benign tumors can be treated or prevented using the technology of the present disclosure. It is understood that it is possible.
- the benign tumor is familial adenomatous polyposis, non-hereditary colorectal adenoma, intraductal papillary mucinous tumor, meningioma of the brain, schwannomas, epithelial adenoma of each organ, papilloma , Non-epithelial fibroids, lipomas, chondromas, and hemangiomas.
- the present disclosure provides an agent for preventing or treating familial adenomatous polyposis comprising a WT1 peptide or an analog thereof.
- the WT1 peptide or analog thereof has been shown to be effective in the treatment of angiogenesis, it is intractable and has been used in the treatment or prevention of familial adenomatous polyposis, which had previously been virtually resected only through resection. I could not expect it to work.
- the present disclosure provides a new treatment for familial adenomatous polyposis, for which there was virtually no cure for resection, a treatment or prophylaxis that does not require resection, and which improves Quality of Life (QOL). It is useful as a contributor.
- QOL Quality of Life
- the WT1 peptide of the present disclosure may be of one type or a plurality of types.
- the WT1 peptide used in the medicament or the composition of the present disclosure may be a killer WT1 peptide, or may be a helper WT1 peptide, or a mixture thereof. More preferably, it contains both a killer type WT1 peptide and a helper type WT1 peptide.
- a dimer of the WT1 peptide may be used.
- a dimer of a WT1 peptide may be obtained by forming a disulfide bond between two WT1 peptides having cysteine residues.
- the WT1 peptide used in the pharmaceutical composition of the present disclosure may be a single type or a plurality of types.
- Whether a WT1 peptide exerts a therapeutic or prophylactic effect in a subject depends on whether the WT1 peptide corresponds to the HLA type of the subject. At present, it is known which HLA type is compatible with many WT1 peptides, so that the WT1 peptide used in the present disclosure can be selected according to the HLA type of interest. In addition, a plurality of types of WT1 peptides may be used in the pharmaceutical composition of the present disclosure to cover a wide range of subjects.
- the WT1 peptide used in the present disclosure is a WT1 126 killer peptide, a WT1 235 killer peptide, a WT1 35 helper peptide and / or a WT1 332 helper peptide.
- the WT1 peptide includes both a killer peptide selected from the WT1 126 peptide and the WT1 235 peptide, and a helper peptide selected from the WT1 35 and WT1 332 peptides.
- the WT1 peptide has an effect beyond that used alone when it includes a killer peptide and a helper peptide.
- WT1 peptide of the present disclosure includes both compounds and WT1 35 helper peptide represented by the formula (3).
- Combinations of a compound of the WT1 35 helper peptide represented by the formula (3) is the knowledge of the Applicant, to achieve the prevention and / or therapeutic efficacy against benign tumors was not known.
- the present disclosure provides an agent for preventing or treating a benign tumor, comprising a nucleic acid molecule encoding a WT1 peptide or an analog thereof.
- the disclosure provides a prophylactic or therapeutic agent for familial adenomatous polyposis comprising a nucleic acid molecule encoding a WT1 peptide or analog thereof.
- the active ingredient in the pharmaceutical composition of the present disclosure is a polynucleotide encoding a WT1 peptide, but is not limited thereto.
- the nucleotide sequence of the polynucleotide can be determined based on the amino acid sequence of the WT1 peptide.
- the polynucleotide can be produced by, for example, a known DNA or RNA synthesis method such as a chemical synthesis method, a PCR method, or the like.
- a prophylactic or therapeutic agent of the present disclosure comprises a DNA encoding the WT1 peptide or an analog thereof.
- the prophylactic or therapeutic agent of the present disclosure comprises RNA encoding the WT1 peptide or an analog thereof.
- a prophylactic or therapeutic agent of the present disclosure comprises RNA and DNA encoding the WT1 peptide or an analog thereof.
- a prophylactic or therapeutic agent of the present disclosure comprises an adjuvant in addition to the above.
- the adjuvant used in the present disclosure comprises Montanide® ISA51 adjuvant.
- a prophylactic or therapeutic agent of the present disclosure is administered once a week.
- the medicament or composition of the present disclosure may be used in combination with a medicament used for either or both treatment and prevention of benign tumors (eg, familial adenomatous polyposis).
- benign tumors eg, familial adenomatous polyposis
- the route of administration of the medicament or composition of the present disclosure is not particularly limited, but examples of preferred routes of administration include intradermal administration, subcutaneous administration, transdermal administration, and transmucosal administration (eg, ophthalmic, nasal, sublingual, etc.).
- routes of administration include intradermal administration, subcutaneous administration, transdermal administration, and transmucosal administration (eg, ophthalmic, nasal, sublingual, etc.).
- the dosage form of the medicament or composition of the present disclosure is not particularly limited, and examples thereof include dosage forms such as injection solutions, ophthalmic solutions, nasal solutions, lotions, creams, patches, sublingual tablets, and troches. You may. These dosage forms can be prepared and administered by methods well known to those skilled in the art.
- the dose of the WT1 peptide when using the medicine or composition of the present disclosure is appropriately changed in consideration of the type of the WT1 peptide, the administration route, the dosage form, the type of the disease, the degree of the disease, the health condition of the subject, and the like. be able to. Generally, the dose of the WT1 peptide will be 0.1 ⁇ g / kg to 1 mg / kg per adult per day.
- the type, administration route, and dosage form of the WT1 peptide can also be appropriately changed in the same manner.
- the medicament or composition of the present disclosure may contain, in addition to pharmaceutically acceptable carriers and excipients, a suitable adjuvant such as, for example, aluminum hydroxide.
- a medicament or composition of the present disclosure may include a WT1 peptide encapsulated in liposomes.
- the dose of the WT1 peptide which is a compound represented by the formula (2) or (3), is 3.5 mg per adult day every 2 weeks in the induction period, for a total of 1 to 5 times.
- 3.5 mg per adult per day can be administered intradermally every 3 months, every 1 or 2 months.
- the dose is 1.0 mg or more, 2.5 mg or more, 5.0 mg or more, 10 mg or more, 15 mg or more, 20 mg or more, or 25 mg or more, or 100 mg or less, 50 mg or less, 40 mg or less, 30 mg or less, 25 mg or less per day for adults. It can be administered in a range of 20 mg or less, 15 mg or less, 10 mg or less, 5.0 mg or less, or 2.5 mg or less.
- the specific dose is, for example, 0.5 mg, 1.0 mg, 1.5 mg, 2.0 mg, 2.5 mg, 3.0 mg, 3.5 mg, 4.0 mg, 4.5 mg, 5.0 mg per adult. 5.5 mg, 6.0 mg, 6.5 mg, 7.0 mg, 7.5 mg, 8.0 mg, 8.5 mg, 9.0 mg, 9.5 mg or 10 mg.
- the administration interval can be appropriately selected from one week to one year. For example, one day or more, one week or more, two weeks or more, three weeks or more, one month or more, two months or more, three months or more, 1 month, 5 months, 6 months, 1 year, 9 months, 6 months, 5 months, 4 months, 3 months, 2 months, 1 month, 3 weeks, 2 weeks It can be administered at intervals of up to one week or less. Specifically, as the administration interval, for example, 1 day, 3 days, 5 days, 1 week, 2 weeks, 3 weeks, 1 month, 2 months, 3 months, 4 months, 5 months, 6 months, 9 months or 1 month The year may be used.
- the number of times of administration can be appropriately selected from a total of 1 to 100 times, for example, 1 time or more, 2 times or more, 3 times or more, 4 times or more, 5 times or more, 6 times or more, 7 times or more, 8 times or more. Or more, 9 or more or 10 or more, or 100 or less, 50 or less, 40 or less, 30 or less, 20 or less, 15 or less, 10 or less, 9 or less, 8 or less, 7 times In the following, the administration can be performed 6 times or less, 5 times or less, 4 times or less, 3 times or less, or 2 times or less.
- the specific number of administrations may be, for example, one, two, three, four, five, six, seven, eight, nine, ten, eleven, or twelve. .
- the disclosure encodes a WT1 peptide or analog thereof, or a WT1 peptide or analog thereof, for either or both treatment and prevention of benign tumors (eg, familial adenomatous polyposis).
- benign tumors eg, familial adenomatous polyposis.
- the disclosure is directed to either or both treatment and prevention of benign tumors (eg, familial adenomatous polyposis) or to treatment and prevention of benign tumors (eg, familial adenomatous polyposis).
- benign tumors eg, familial adenomatous polyposis
- benign tumors eg, familial adenomatous polyposis
- a WT1 peptide or an analog thereof, or a nucleic acid molecule encoding a WT1 peptide or an analog thereof for the manufacture of a pharmaceutical composition for either or both.
- the disclosure further includes administering to a subject in need thereof an effective amount of a WT1 peptide or analog thereof, or a nucleic acid molecule encoding the WT1 peptide or analog thereof, to a benign tumor (eg, familial colon) For the treatment and / or prevention of adenomatosis).
- a benign tumor eg, familial colon
- the present disclosure provides a WT1 peptide or analog thereof, or a WT1 peptide or analog thereof, for the manufacture of a medicament for either or both treatment and prevention of benign tumors (eg, familial adenomatous polyposis). It relates to the use of a body-encoding nucleic acid molecule.
- the disclosure provides a WT1 peptide or an analog thereof, or a WT1 peptide or an analog thereof in a subject in need of either or both treatment and prevention of a benign tumor (eg, familial adenomatous polyposis). And / or a method for treating and / or preventing a benign tumor (eg, familial adenomatous polyposis), which comprises administering a nucleic acid molecule encoding the same.
- a benign tumor eg, familial adenomatous polyposis
- the present disclosure provides peripheral blood mononuclear cells from a subject in need of treatment for a benign tumor (eg, familial adenomatous polyposis) with any of the WT1 peptides described herein or a WT1 peptide thereof.
- WT1-specific cytotoxic T cells (CTLs) from the peripheral monocytes, by culturing in the presence of the analogs or by introducing nucleic acid molecules encoding them into the peripheral blood mononuclear cells, and / or Or WT1-specific helper T cells are induced.
- CTLs cytotoxic T cells
- WT1-specific CTLs and / or WT1-specific helpers used for prevention or treatment of benign tumors (eg, familial adenomatous polyposis) Methods for inducing T cells are provided. It will be understood by those skilled in the art that any description given in (Prophylactic or Therapeutic Agents) also applies in this guidance method.
- the disclosure provides for immature dendritic cells from a subject in need of treatment for a benign tumor (eg, familial adenomatous polyposis) by any of the WT1 peptides described herein or analogs thereof.
- a benign tumor eg, familial adenomatous polyposis
- Inducing a WT1-presenting dendritic cell by culturing in the presence of a body or introducing a nucleic acid molecule encoding the same into the immature dendritic cell.
- the present invention provides a method for inducing WT1-presenting dendritic cells for use in the prevention or treatment of adenomatous polyposis. It will be understood by those skilled in the art that any description given in (Prophylactic or Therapeutic Agents) also applies in this method of induction.
- the present disclosure provides a WT1-specific for use in the prevention or treatment of a benign tumor (eg, familial adenomatous polyposis), comprising a WT1 peptide or analog thereof, or a nucleic acid molecule encoding the same.
- a benign tumor eg, familial adenomatous polyposis
- Compositions for inducing cytotoxic T cells and / or WT1-specific helper T cells are provided. It will be appreciated by those skilled in the art that any statements made in (Prophylactic or Therapeutic) also apply in this composition.
- the present disclosure provides a WT1 presentation for use in the prevention or treatment of a benign tumor (eg, familial adenomatous polyposis) comprising a WT1 peptide or analog thereof, or a nucleic acid molecule encoding the same.
- a benign tumor eg, familial adenomatous polyposis
- compositions for inducing dendritic cells are provided. It will be appreciated by those skilled in the art that any statements made in (Prophylactic or Therapeutic) also apply in this composition.
- compositions for preventing or treating benign tumors comprising WT1-specific cytotoxic T cells and / or WT1-specific helper T cells. Offer things. It will be appreciated by those skilled in the art that any statements made in (Prophylactic or Therapeutic) also apply in this composition.
- the present disclosure provides a composition for preventing or treating a benign tumor (eg, familial adenomatous polyposis), comprising a WT1-presenting dendritic cell.
- a benign tumor eg, familial adenomatous polyposis
- WT1-presenting dendritic cell e.g, familial adenomatous polyposis
- the peptide or derivative or nucleic acid molecule of the present disclosure can be produced by a production method commonly used in the art. These production methods can be appropriately modified based on the knowledge of those skilled in immunological techniques, molecular biological techniques, biochemical techniques, and microbiological techniques. Specifically, the peptide or derivative or nucleic acid molecule of the present disclosure may be designed based on the amino acid sequence (eg, SEQ ID NO: 1) or nucleic acid sequence of a natural WT1 protein, and may be produced by a microorganism expression system or the like. May be.
- the starting materials and intermediates in the production method can be purchased as commercial products, or can be obtained according to known methods from known methods or known compounds. As these starting materials and intermediates, analogs thereof may be used as long as they do not hinder the production process.
- mice In the following examples, Apc Min / + mice (C57BL / 6J) mice (Jackson Institute, Bar Harbor, Maine, USA) were used as mice. The mice were bred in a specific microorganism-free (SPF) containment facility at an animal experiment facility attached to the Osaka University School of Medicine, in compliance with the Osaka University Animal Experiment Regulations.
- SPF microorganism-free
- the MHC class I (H-2D b ) -binding peptide, WT1 126-134 (RMFPNAPYL, 9a.a), and the MHC class II (H-2I-A b ) -binding peptide, WT1 35-52 (WAPVLDFAPPGASAYGSL, 18 aa) was purchased from SIGMA Genosys (Ishikari, Japan). Each raw material used for the mixture of the compound of the formula (3) and the WT135-52 helper peptide was provided by Sumitomo Dainippon Pharma (Osaka, Japan). Peptides were stored at -20 0 C until use.
- the peptide was dissolved in phosphate buffered saline (PBS; 137 mM NaCl, 2.7 mM KCl, 8.1 mM Na 2 HPO 4 , 1.47 mM KH 2 PO 4 ) and stored at ⁇ 20 ° C.
- PBS phosphate buffered saline
- IFA Incomplete Adjuvant
- Montanide ISA 51 is available from Seppic S.D. A. (Orsay, France).
- Example 1 Expression of WT1 protein in adenoma of familial adenomatous polyposis
- Example 1 The results of Example 1 are shown in Table 1 and FIG. Table 1 shows the expression levels of WT1 protein in adenomas, normal ducts and fibroblasts in specimens from each patient. Table 1 shows that the expression level of the WT1 protein in the adenoma is significantly higher than that in the normal duct. In FIG. 1, light staining indicates the WT1 protein, and the darkly stained area indicates the nucleus. WT1 protein expression was observed in adenomas, but WT1 protein expression was not observed in normal gland ducts.
- Example 2 Expression of WT1 protein in APC Min / + mice
- This example shows that WT1 protein was expressed in small intestinal adenoma of APC Min / + mouse, which is a model mouse for familial adenomatous polyposis.
- Example 2 The results of Example 2 are shown in FIGS.
- FIG. 2 shows micrographs at 5 ⁇ , 10 ⁇ , and 20 ⁇ magnification in order from the top
- FIG. 3 shows micrographs at 40 ⁇ magnification.
- intense staining indicates WT1 protein and circularly stained areas indicate nuclei.
- WT1 protein expression was observed in small intestinal adenomas of Apc Min / + mice, which are model mice of familial adenomatous polyposis.
- Example 3 Administration of WT1 peptide vaccine to familial adenomatous polyposis model mouse
- This example shows an administration scheme of an experiment in which a WT1 peptide vaccine was administered to APC Min / + mice.
- the administration scheme of the WT1 peptide vaccine is shown in FIG.
- WT1 peptide vaccine or control vaccine was administered intradermally to the flanks of 4-5 week old Apc Min / + mice. Immunization was started 5 weeks after birth, and was performed 8 times, once a week. Mice immunized 10 days after the final immunization were euthanized and further analysis was performed. In addition, administration of the WT1 peptide vaccine did not cause organ damage as shown in Table 3 below.
- Example 4 Prevention and treatment of adenoma by administration of WT1 peptide vaccine
- This example shows that administration of the WT1 peptide vaccine to APC Min / + mice prevented the onset of adenoma and treated the adenoma.
- mice obtained by the administration scheme described in Example 3 were used.
- Whole intestine was collected from mice euthanized 10 days after the last immunization.
- the small intestine was divided into 8-10 fractions, and the large intestine was divided into cecum and ascending colon.
- Each fraction was cut in the longitudinal direction, washed with PBS, sandwiched between filter papers, and fixed in 4% paraformaldehyde / phosphate buffer at 4 ° C. for 24 hours or more.
- the fixed intestinal tissue was stained with 1% methylene blue solution, and then observed under a stereoscopic microscope, and the number of adenomas (polyps) was counted.
- the difference in the number of adenomas (polyps) between the WT1 peptide vaccine administration group and the control vaccine administration group was statistically analyzed by Student's t test.
- Example 4 The result of Example 4 is shown in FIG.
- the horizontal axis of the graph indicates the group to which the WT1 vaccine was administered on the left side, the group to which the control vaccine was administered on the right side, and the vertical axis indicates the number of adenomas per small intestine.
- the average number of adenomas per small intestine was about 29 in the WT1 peptide vaccine administration group, whereas it was about 34 in the control vaccine administration group.
- the administration of the WT1 peptide vaccine significantly suppressed the development of adenomas And showed that the adenoma had been treated.
- Example 5 Increase in WT1 tetramer + CD3 + CD8 + T cells by administration of WT1 peptide vaccine
- This example shows that administration of a WT1 peptide vaccine to APC Min / + mice increased WT1 tetramer + CD3 + CD8 + T cells.
- mice obtained by the administration scheme described in Example 3 were used. Splenocytes were collected from mice euthanized 10 days after the final immunization. After lysis of the erythrocytes, the cells were stained with H-2D b WT1 tetramer (MBL, Aichi, Japan), followed by anti-mouse CD3 antibody (17A2, BioLegend, San Diego, CA) and anti-mouse CD8 antibody (KT15, MBL, Aichi). , Japan). Stained cells were analyzed by FACSCanto, it was measured the frequency of CD3 + CD8 + T H-2D b WT1 tetramer + CD3 + CD8 + T cells in a cell. The difference in the frequency of H-2D b WT1 tetramer + CD3 + CD8 + T cells between the WT1 peptide vaccine administration group and the control vaccine administration group was statistically analyzed by Student's t test.
- Example 5 The results of Example 5 are shown in FIG.
- the horizontal axis of the graph indicates the group to which the WT1 peptide vaccine was administered on the left side, the group to which the control vaccine was administered on the right side, and the vertical axis indicates the frequency of H-2D b WT1 tetramer in CD3 + CD8 + cells.
- the WT1 vaccine administration group, CD3 + CD8 + T mean frequency of H-2D b WT1 tetramer + CD3 + CD8 + T cells in the cell while it was about 0.6% in the control vaccine group Approximately 0.2%, indicating that administration of the WT1 vaccine increased WT1 tetramer + CD3 + CD8 + T cells.
- Example 6 Statistical analysis
- Example 4 adenomas per intestine obtained from Example 4, and a frequency of H-2D b WT1 tetramer + CD3 + CD8 + T cells in CD3 + CD8 + T cells obtained from Example 5, Microsoft Excel ( ® plotted on a scatter plot. Further, for these results, the correlation was statistically analyzed by Excel's Pearson correlation coefficient.
- Example 7 Example with a single agent
- adenomas were administered to APC Min / + mice when both killer-type and helper-type WT1 peptide vaccines were administered and when only a killer-type or helper-type WT1 peptide vaccine was administered alone.
- 2 shows a comparison between the effects of preventing and treating the onset of the disease.
- the administration scheme of the WT1 peptide vaccine is shown in FIG.
- a mixture of 100 ⁇ g of WT1 126-134 killer peptide and 45 ⁇ g of WT1 35-52 helper peptide, 100 ⁇ g of WT1 126-134 killer peptide alone, 45 ⁇ g of WT1 35-52 helper peptide alone or PBS were each treated with IFA Montanide ISA51 adjuvant. Emulsify. Control vaccines containing a mixture of killer peptide and helper peptide, killer peptide alone, helper peptide alone, or PBS alone are intradermally administered to the flanks of 5-week-old Apc Min / + mice. Immunization is started 5 weeks after birth, and is performed 8 times a week. Mice immunized 10 days after the last immunization are euthanized and further analysis is performed.
- Example 8 Expression of WT1 in other benign tumors
- Adenoma tissue was excised from three human patients who developed non-hereditary colorectal adenoma, and WT1 protein and nuclei were stained by the same method as in Example 1.
- Example 8 The result of Example 8 is shown in FIG.
- the first image from the left shows a microscope image of a normal gland duct
- the three images on the right show a microscopic image of a non-hereditary adenoma adenoma tissue.
- the light staining indicates the WT1 protein
- the darkly circular or oval stained area indicates the nucleus. WT1 protein expression was observed in adenomas, but WT1 protein expression was not observed in normal gland ducts.
- Example 9 Administration of a mixture of the compound of the formula (3) and the WT135-52 helper peptide to a familial adenomatous polyposis model mouse
- This example shows a dosing scheme for experiments in which APC Min / + mice were administered a mixture of a compound of formula (3), a WT1 peptide vaccine, and a WT1 35-52 helper peptide.
- the administration scheme of the WT1 peptide vaccine is shown in FIG.
- the WT1 peptide vaccine was dissolved in PBS, and the lysate containing the WT1 peptide vaccine and PBS alone were emulsified with IFA Montanide ISA51 adjuvant to give a WT1 peptide vaccine or a control vaccine, respectively.
- WT1 peptide vaccine or control vaccine was administered intradermally to the flanks of 4-5 week old Apc Min / + mice. Immunization was started 4 to 5 weeks after birth, and was performed 6 times a week. The immunized mice were euthanized 7 days after the final immunization and further analysis was performed.
- Example 10 Prevention and treatment of adenoma by administration of a mixture of a compound of formula (3) and WT1 35-52 helper peptide
- This example shows that the administration of a mixture of the compound of formula (3) and the WT135-52 helper peptide to APC Min / + mice prevented the development of adenoma and treated the adenoma.
- the administration of the mixture of the compound of the formula (3) and the WT135-52 helper peptide did not cause organ damage as shown in Table 4 below.
- mice obtained by the administration scheme described in Example 9 were used. Seven days after the final immunization, each organ was excised from the euthanized mouse and its weight was measured. The small intestine was divided into 8-10 fractions. Each fraction was cut in the longitudinal direction, washed with PBS, sandwiched between filter papers, and fixed in 4% paraformaldehyde / phosphate buffer at 4 ° C. for 24 hours or more. The fixed intestinal tissue was stained with 1% methylene blue solution, and then observed under a stereoscopic microscope, and the number of adenomas (polyps) was counted. The difference in the number of adenomas (polyps) between the WT1 peptide vaccine administration group and the control vaccine administration group was statistically analyzed by Student's t test.
- Example 10 The result of Example 10 is shown in FIG.
- the horizontal axis of the graph indicates the group to which the WT1 peptide vaccine was administered on the left side, the group to which the control vaccine was administered on the right side, and the vertical axis indicates the number of adenomas per small intestine.
- the average value of the number of adenomas per small intestine was about 37.2 in the WT1 peptide vaccine administration group, whereas it was about 43.7 in the control vaccine administration group. Therefore, the administration of the WT1 peptide vaccine suppressed the onset of adenoma and tended to treat adenoma.
- Example 11 Increase of WT1 tetramer + CD3 + CD8 + T cells by administration of a mixture of a compound of formula (3) and WT1 35-52 helper peptide
- This example shows that administration of a mixture of the compound of formula (3) and the WT1 35-52 helper peptide to APC Min / + mice increased WT1 tetramer + CD3 + CD8 + T cells.
- mice obtained by the administration scheme described in Example 9 were used. Splenocytes were collected from mice euthanized 7 days after the final immunization. After lysis of erythrocytes, the cells were stained with H-2D b WT1 tetramer (MBL, Aichi, Japan), and further, anti-mouse CD3 antibody (17A2, BioLegend, San Diego, CA), anti-mouse CD8 antibody (KT15, MBL, Aichi) , Japan) and 7-AAD Viability Staining Solution (Bioscience, San Diego, CA).
- Example 11 The result of Example 11 is shown in FIG.
- the horizontal axis of the graph indicates the group to which the WT1 peptide vaccine was administered on the left side, the group to which the control vaccine was administered on the right side, and the vertical axis indicates the frequency of H-2D b WT1 tetramer in CD3 + CD8 + cells.
- the WT1 peptide vaccine administration group whereas the mean value of the frequency of H-2D b WT1 tetramer + CD3 + CD8 + T cells in CD3 + CD8 + T cells was about 0.13%, the control vaccinated group was about 0.04%, indicating that the administration of the WT1 peptide vaccine increased WT1 tetramer + CD3 + CD8 + T cells.
- Example 12 Statistical analysis
- Example 10 adenomas per intestine obtained from Example 10, and a frequency of H-2D b WT1 tetramer + CD3 + CD8 + T cells in the CD3 + CD8 + T cells obtained from Example 11, Microsoft Excel ( ® plotted on a scatter plot. Further, for these results, the correlation was statistically analyzed by Excel's Pearson correlation coefficient.
- the present disclosure is useful for preventing, delaying and treating benign tumors (eg, familial adenomatous polyposis) and for preventing, delaying and treating symptoms arising from adenomas of benign tumors (eg, familial adenomatous polyposis).
- benign tumors eg, familial adenomatous polyposis
- adenomas of benign tumors eg, familial adenomatous polyposis.
- the WT1 peptide vaccine of the present disclosure has utility in that it avoids surgery such as endoscopic resection, has no serious side effects, and is extremely safe.
Abstract
Description
(項目X1)
WT1ペプチドまたはその類縁体を含む、良性腫瘍の予防または治療薬。
(項目X2)
前記WT1ペプチドまたはその類縁体は、キラー型および/またはヘルパー型を含む、項目X1に記載の予防または治療薬。
(項目X3)
前記WT1ペプチドまたはその類縁体は、WT1126キラーペプチドおよび/またはWT135ヘルパーペプチドを含む、項目X1または項目X2に記載の予防または治療薬。
(項目X4)
WT1ペプチドまたはその類縁体をコードする核酸分子を含む、項目X1~項目X3のいずれか一項に記載の良性腫瘍の予防または治療薬。
(項目X5)
前記核酸分子は、RNAおよび/またはDNAを含む、項目X1~項目X4のいずれか一項に記載の予防または治療薬。
(項目X6)
アジュバントをさらに含む、項目X1~項目X5のいずれか一項に記載の予防または治療薬。
(項目X7)
前記アジュバントがMontanide(登録商標) ISA51アジュバントである、項目X1~項目X6のいずれか一項に記載の予防または治療薬。
(項目X8)
前記良性腫瘍は、WT1を発現する、項目X1~項目X7のいずれか一項に記載の予防または治療薬。
(項目X9)
前記良性腫瘍は、家族性大腸腺腫症、非遺伝性の大腸腺腫、膵管内乳頭粘液性腫瘍、脳の髄膜腫、神経鞘腫、各臓器の上皮性の腺腫、乳頭腫、非上皮性の筋腫、脂肪腫、軟骨腫、血管腫からなる群より選択される、項目X1~項目X8のいずれか一項に記載の予防または治療薬。
(項目X10)
前記良性腫瘍は、家族性大腸腺腫症である、項目X1~項目X9のいずれか一項に記載の予防または治療薬。
(項目X11)
1週間に1回投与されることを特徴とする、項目X1~項目X10のいずれか一項に記載の予防または治療薬。
(項目X12)
良性腫瘍の処置を必要とする被験体由来の末梢血単核球を、上記項目Xのいずれか一項に記載のWT1ペプチドまたはその類縁体の存在下で培養し、あるいは、上記項目Xのいずれか一項に記載のWT1ペプチドまたはその類縁体をコードする核酸分子を該末梢血単核球に導入し、該末梢単核球からWT1特異的細胞傷害性T細胞(CTLs)、および/またはWT1特異的ヘルパーT細胞を誘導する工程を含むことを特徴とする、良性腫瘍の予防または治療のために使用するWT1特異的CTLsおよび/またはWT1特異的ヘルパーT細胞の誘導方法。
(項目X13)
良性腫瘍の処置を必要とする被験体由来の未熟樹状細胞を、上記項目のいずれか一項に記載のWT1ペプチドまたはその類縁体の存在下で培養し、あるいは、上記項目Xのいずれか一項に記載のWT1ペプチドまたはその類縁体をコードする核酸分子を該未熟樹状細胞に導入し、WT1提示樹状細胞を誘導する工程を含むことを特徴とする、良性腫瘍の予防または治療のために使用するWT1提示樹状細胞の誘導方法。
(項目X13A)
項目X1~項目X11のいずれか1つまたは複数に記載の1つまたは複数の特徴をさらに含む、項目X12または13に記載の方法。
(項目X14)
WT1ペプチドもしくはその類縁体、またはそれをコードする核酸分子を含む、良性腫瘍の予防または治療のために使用する、WT1特異的CTLs、および/またはWT1特異的ヘルパーT細胞を誘導するための組成物。
(項目X15)
WT1ペプチドもしくはその類縁体、またはそれをコードする核酸分子を含む、良性腫瘍の予防または治療のために使用する、WT1提示樹状細胞を誘導するための組成物。
(項目X15A)
項目X1~項目X13のいずれか1つまたは複数に記載の1つまたは複数の特徴をさらに含む、項目X14または項目X15に記載の組成物。
(項目X16)
WT1特異的CTLs、および/またはWT1特異的ヘルパーT細胞を含む、良性腫瘍の予防または治療のための組成物。
(項目X17)
WT1提示樹状細胞を含む、良性腫瘍の予防または治療のための組成物。
(項目X17A)
項目X1~項目X13のいずれか1つまたは複数に記載の1つまたは複数の特徴をさらに含む、項目X16または項目X17に記載の組成物。
(項目1)
WT1ペプチドまたはその類縁体を含む、良性腫瘍の予防または治療薬。
(項目2)
前記WT1ペプチドまたはその類縁体は、キラー型および/またはヘルパー型を含む、項目1に記載の予防または治療薬。
(項目3)
前記WT1ペプチドまたはその類縁体は、WT1126キラーペプチド、WT1235キラーペプチドおよび/またはWT135ヘルパーペプチド、またはいずれかのアミノ酸配列において、1個~数個のアミノ酸が、欠失、置換、および/または付加されたアミノ酸配列を含み且つCTL誘導活性を有するペプチドを含む、項目1または2に記載の予防または治療薬。
(項目4)
前記WT1ペプチドまたはその類縁体は、
RMFPNAPYL (配列番号2)、
RYFPNAPYL (配列番号46)、
YMFPNAPYL (配列番号14)、
CYTWNQMNL (配列番号45)、
CMTWNQMNL (配列番号3)、
C-CYTWNQMNL(配列番号47)(式中、CとCの間の結合はジスルフィド結合を表す。)、および
C-CMTWNQMNL(配列番号48)(式中、CとCの間の結合はジスルフィド結合を表す。)から選択されるいずれかのアミノ酸配列からなるペプチド、またはその薬学上許容される塩である、
項目1~3のいずれか一項に記載の予防または治療薬。
(項目5)
前記WT1ペプチドまたはその類縁体が、
式(2):
で表される化合物、またはその薬学上許容される塩である、
項目1~4のいずれか一項に記載の予防または治療薬。
(項目6)
前記WT1ペプチドまたはその類縁体が、
式(3):
で表される化合物、またはその薬学上許容される塩である、
項目1~5のいずれか一項に記載の予防または治療薬。
(項目7)
前記WT1ペプチドまたはその類縁体が、さらに
以下のアミノ酸配列:
WAPVLDFAPPGASAYGSL (配列番号4)、
CWAPVLDFAPPGASAYGSL (配列番号50)および
WAPVLDFAPPGASAYGSLC (配列番号51)、
からなる群から選択されるアミノ酸配列からなるペプチドまたはその薬学上許容される塩を一つ以上含む組成物である、項目1~6のいずれか一項に記載の予防または治療薬。
(項目8)
前記WT1ペプチドまたはその類縁体が、
式(2):
で表される化合物またはその薬学上許容される塩と、
アミノ酸配列: WAPVLDFAPPGASAYGSL (配列番号4)からなるペプチドまたはその薬学上許容される塩を含む組成物である、
項目1~7のいずれか一項に記載の予防または治療薬。
(項目9)
前記WT1ペプチドまたはその類縁体が、
式(3):
で表される化合物またはその薬学上許容される塩と、
アミノ酸配列: WAPVLDFAPPGASAYGSL (配列番号4)からなるペプチドまたはその薬学上許容される塩を含む組成物である、
項目1~8のいずれか一項に記載の予防または治療薬。
(項目10)
WT1ペプチドまたはその類縁体をコードする核酸分子を含む、良性腫瘍の予防または治療薬。
(項目11)
前記核酸分子は、RNAおよび/またはDNAを含む、項目A1~A10のいずれか一項に記載の予防または治療薬。
(項目12)
アジュバントをさらに含む、項目1~11のいずれか一項に記載の予防または治療薬。
(項目13)
前記アジュバントがMontanide(登録商標) ISA51アジュバントである、項目1~12のいずれか一項に記載の予防または治療薬。
(項目14)
前記良性腫瘍は、WT1を発現する、項目1~13のいずれか一項に記載の予防または治療薬。
(項目15)
前記良性腫瘍は、家族性大腸腺腫症、非遺伝性の大腸腺腫、膵管内乳頭粘液性腫瘍、脳の髄膜腫、神経鞘腫、各臓器の上皮性の腺腫、乳頭腫、非上皮性の筋腫、脂肪腫、軟骨腫、血管腫からなる群より選択される、項目1~14のいずれか一項に記載の予防または治療薬。
(項目16)
前記良性腫瘍は、家族性大腸腺腫症である、項目1~15のいずれか一項に記載の予防または治療薬。
(項目17)
1週間に1回投与されることを特徴とする、項目1~16のいずれか一項に記載の予防または治療薬。
(項目18)
良性腫瘍の処置を必要とする被験体由来の末梢血単核球を、上記項目のいずれか一項に記載のWT1ペプチドまたはその類縁体の存在下で培養し、あるいは、上記項目のいずれか一項に記載のWT1ペプチドまたはその類縁体をコードする核酸分子を該末梢血単核球に導入し、該末梢血単核球からWT1特異的細胞傷害性T細胞(CTLs)、および/またはWT1特異的ヘルパーT細胞を誘導する工程を含む、良性腫瘍の予防または治療のために使用するWT1特異的CTLsおよび/またはWT1特異的ヘルパーT細胞の誘導方法。
(項目19)
良性腫瘍の処置を必要とする被験体由来の未熟樹状細胞を、上記項目のいずれか一項に記載のWT1ペプチドまたはその類縁体の存在下で培養し、あるいは、上記項目のいずれか一項に記載のWT1ペプチドまたはその類縁体をコードする核酸分子を該未熟樹状細胞に導入し、WT1提示樹状細胞を誘導する工程を含む、良性腫瘍の予防または治療のために使用するWT1提示樹状細胞の誘導方法。
(項目19A)
項目1~17のいずれか1つまたは複数に記載の1つまたは複数の特徴をさらに含む、項目18または19に記載の方法。
(項目20)
WT1ペプチドもしくはその類縁体、またはそれをコードする核酸分子を含む、良性腫瘍の予防または治療のために使用する、WT1特異的CTLs、および/またはWT1特異的ヘルパーT細胞を誘導するための組成物。
(項目21)
WT1ペプチドもしくはその類縁体、またはそれをコードする核酸分子を含む、良性腫瘍の予防または治療のために使用する、WT1提示樹状細胞を誘導するための組成物。
(項目21A)
項目1~19のいずれか1つまたは複数に記載の1つまたは複数の特徴をさらに含む、項目20または21に記載の組成物。
(項目22)
WT1特異的CTLs、および/またはWT1特異的ヘルパーT細胞を含む、良性腫瘍の予防または治療のための組成物。
(項目23)
WT1提示樹状細胞を含む、良性腫瘍の予防または治療のための組成物。
(項目23A)
項目1~19のいずれか1つまたは複数に記載の1つまたは複数の特徴をさらに含む、項目22または23に記載の組成物。
(項目A1)
被験体における良性腫瘍を予防または治療する方法であって、該被験体に、有効量のWT1ペプチドまたはその類縁体を投与する工程を含む、方法。
(項目A2)
前記WT1ペプチドまたはその類縁体は、キラー型および/またはヘルパー型を含む、項目A1に記載の方法。
(項目A3)
前記WT1ペプチドまたはその類縁体は、WT1126キラーペプチド、WT1235キラーペプチドおよび/またはWT135ヘルパーペプチド、またはいずれかのアミノ酸配列において、1個~数個のアミノ酸が、欠失、置換、および/または付加されたアミノ酸配列を含み且つCTL誘導活性を有するペプチドを含む、項目A1またはA2に記載の方法。
(項目A4)
前記WT1ペプチドまたはその類縁体が、
RMFPNAPYL (配列番号2)、
RYFPNAPYL (配列番号46)、
YMFPNAPYL (配列番号14)、
CYTWNQMNL (配列番号45)、
CMTWNQMNL (配列番号3)、
C-CYTWNQMNL(配列番号47)(式中、CとCの間の結合はジスルフィド結合を表す。)、および
C-CMTWNQMNL(配列番号48)(式中、CとCの間の結合はジスルフィド結合を表す。)から選択されるいずれかのアミノ酸配列からなるペプチド、またはその薬学上許容される塩である、
項目A1~A3のいずれか一項に記載の予防または治療薬。
(項目A5)
前記WT1ペプチドまたはその類縁体が、
式(2):
で表される化合物、またはその薬学上許容される塩である、
項目A1~A4のいずれか一項に記載の方法。
(項目A6)
前記WT1ペプチドまたはその類縁体が、
式(3):
で表される化合物、またはその薬学上許容される塩である、
項目A1~A5のいずれか一項に記載の方法。
(項目A7)
前記WT1ペプチドまたはその類縁体が、さらに
以下のアミノ酸配列:
WAPVLDFAPPGASAYGSL (配列番号4)、
CWAPVLDFAPPGASAYGSL (配列番号50)および
WAPVLDFAPPGASAYGSLC (配列番号51)、
からなる群から選択されるアミノ酸配列からなるペプチドまたはその薬学上許容される塩を一つ以上含む組成物である、項目A1~A6のいずれか一項に記載の方法。
(項目A8)
前記WT1ペプチドまたはその類縁体が、
式(2):
で表される化合物またはその薬学上許容される塩と、
アミノ酸配列: WAPVLDFAPPGASAYGSL (配列番号4)からなるペプチドまたはその薬学上許容される塩を含む組成物である、
項目A1~A7のいずれか一項に記載の方法。
(項目A9)
前記WT1ペプチドまたはその類縁体が、
式(3):
で表される化合物またはその薬学上許容される塩と、
アミノ酸配列: WAPVLDFAPPGASAYGSL (配列番号4)からなるペプチドまたはその薬学上許容される塩を含む組成物である、
項目A1~A8のいずれか一項に記載の方法。
(項目A10)
被験体における良性腫瘍を予防または治療する方法であって、該被験体に、有効量のWT1ペプチドまたはその類縁体をコードする核酸分子を投与する工程を含む、方法。
(項目A11)
前記核酸分子は、RNAおよび/またはDNAを含む、項目A1~A10のいずれか一項に記載の方法。
(項目A12)
前記WT1ペプチドもしくはその類縁体、またはそれをコードする核酸分子が、アジュバントと併用される、項目A1~A11のいずれか一項に記載の方法。
(項目A13)
前記アジュバントがMontanide(登録商標) ISA51アジュバントである、項目A1~A12のいずれか一項に記載の方法。
(項目A14)
前記良性腫瘍は、WT1を発現する、項目A1~A13のいずれか一項に記載の方法。
(項目A15)
前記良性腫瘍は、家族性大腸腺腫症、非遺伝性の大腸腺腫、膵管内乳頭粘液性腫瘍、脳の髄膜腫、神経鞘腫、各臓器の上皮性の腺腫、乳頭腫、非上皮性の筋腫、脂肪腫、軟骨腫、血管腫からなる群より選択される、項目A1~A14のいずれか一項に記載の方法。
(項目A16)
前記良性腫瘍は、家族性大腸腺腫症である、項目A1~A15のいずれか一項に記載の方法。
(項目A17)
前記WT1ペプチドもしくはその類縁体、またはそれをコードする核酸分子が、1週間に1回投与されることを特徴とする、項目A1~A16のいずれか一項に記載の方法。
(項目A18)
被験体における良性腫瘍を予防または治療する方法であって、良性腫瘍の処置を必要とする被験体由来の末梢血単核球を、上記項目のいずれか一項に記載のWT1ペプチドまたはその類縁体の存在下で培養し、あるいは、上記項目のいずれか一項に記載のWT1ペプチドまたはその類縁体をコードする核酸分子を該末梢血単核球に導入し、それにより、該末梢血単核球からWT1特異的細胞傷害性T細胞(CTLs)、および/またはWT1特異的ヘルパーT細胞を誘導する工程、および生成された該WT1特異的CTLsおよび/またはWT1特異的ヘルパーT細胞を、該被験体に投与する工程を含む、方法。
(項目A19)
被験体における良性腫瘍を予防または治療する方法であって、良性腫瘍の処置を必要とする被験体由来の未熟樹状細胞を、上記項目のいずれか一項に記載のWT1ペプチドまたはその類縁体の存在下で培養し、あるいは、上記項目のいずれか一項に記載のWT1ペプチドまたはその類縁体をコードする核酸分子を該未熟樹状細胞に導入し、それにより、WT1提示樹状細胞を誘導する工程、および生成された該樹状細胞を、該被験体に投与する工程を含む、方法。
(項目A19A)
項目A1~A17のいずれか1つまたは複数に記載の1つまたは複数の特徴をさらに含む、項目A18またはA19に記載の方法。
(項目A20)
被験体における良性腫瘍を予防または治療する方法であって、該被験体に、WT1ペプチドもしくはその類縁体、またはそれをコードする核酸分子により誘導された、WT1特異的CTLs、および/またはWT1特異的ヘルパーT細胞の有効量を投与する工程を含む、方法。
(項目A21)
被験体における良性腫瘍を予防または治療する方法であって、該被験体に、WT1ペプチドもしくはその類縁体、またはそれをコードする核酸分子により誘導された、WT1提示樹状細胞の有効量を投与する工程を含む、方法。
(項目A21A)
項目A1~A19のいずれか1つまたは複数に記載の1つまたは複数の特徴をさらに含む、項目A20またはA21に記載の組成物。
(項目A22)
被験体における良性腫瘍を予防または治療する方法であって、該被験体に、有効量のWT1特異的CTLs、および/またはWT1特異的ヘルパーT細胞を投与する工程を含む、方法。
(項目A23)
被験体における良性腫瘍を予防または治療する方法であって、該被験体に、有効量のWT1提示樹状細胞を投与する工程を含む、方法。
(項目A23A)
項目A1~A19のいずれか1つまたは複数に記載の1つまたは複数の特徴をさらに含む、項目A22またはA23に記載の組成物。
(項目B1)
良性腫瘍を予防または治療するための、WT1ペプチドまたはその類縁体。
(項目B2)
キラー型および/またはヘルパー型を含む、項目B1に記載のWT1ペプチドまたはその類縁体。
(項目B3)
WT1126キラーペプチド、WT1235キラーペプチドおよび/またはWT135ヘルパーペプチド、またはいずれかのアミノ酸配列において、1個~数個のアミノ酸が、欠失、置換、および/または付加されたアミノ酸配列を含み且つCTL誘導活性を有するペプチドを含む、項目B1またはB2に記載のWT1ペプチドまたはその類縁体。
(項目B4)
RMFPNAPYL (配列番号2)、
RYFPNAPYL (配列番号46)、
YMFPNAPYL (配列番号14)、
CYTWNQMNL (配列番号45)、
CMTWNQMNL (配列番号3)、
C-CYTWNQMNL(配列番号47)(式中、CとCの間の結合はジスルフィド結合を表す。)、および
C-CMTWNQMNL(配列番号48)(式中、CとCの間の結合はジスルフィド結合を表す。)から選択されるいずれかのアミノ酸配列からなるペプチド、またはその薬学上許容される塩である、
項目B1~B3のいずれか一項に記載のWT1ペプチドまたはその類縁体。
(項目B5)
式(2):
で表される化合物、またはその薬学上許容される塩である、
項目B1~B4のいずれか一項に記載のWT1ペプチドまたはその類縁体。
(項目B6)
式(3):
で表される化合物、またはその薬学上許容される塩である、
項目B1~B5のいずれか一項に記載のWT1ペプチドまたはその類縁体。
(項目B7)
さらに、以下のアミノ酸配列:
WAPVLDFAPPGASAYGSL (配列番号4)、
CWAPVLDFAPPGASAYGSL (配列番号50)および
WAPVLDFAPPGASAYGSLC (配列番号51)、
からなる群から選択されるアミノ酸配列からなるペプチドまたはその薬学上許容される塩を一つ以上含む組成物である、項目B1~B6のいずれか一項に記載のWT1ペプチドまたはその類縁体。
(項目B8)
式(2):
で表される化合物またはその薬学上許容される塩と、
アミノ酸配列: WAPVLDFAPPGASAYGSL (配列番号4)からなるペプチドまたはその薬学上許容される塩を含む組成物である、
項目B1~B7のいずれか一項に記載のWT1ペプチドまたはその類縁体。
(項目B9)
式(3):
で表される化合物またはその薬学上許容される塩と、
アミノ酸配列: WAPVLDFAPPGASAYGSL (配列番号4)からなるペプチドまたはその薬学上許容される塩を含む組成物である、
項目B1~B8のいずれか一項に記載のWT1ペプチドまたはその類縁体。
(項目B10)
被験体における良性腫瘍を予防または治療するための、WT1ペプチドまたはその類縁体をコードする核酸分子。
(項目B11)
RNAおよび/またはDNAを含む、項目B1~B10のいずれか一項に記載の核酸分子。
(項目B12)
アジュバントと併用される、項目B1~B11のいずれか一項に記載のWT1ペプチドもしくはその類縁体、または核酸分子。
(項目B13)
前記アジュバントがMontanide(登録商標) ISA51アジュバントである、項目B1~B12のいずれか一項に記載のWT1ペプチドもしくはその類縁体、または核酸分子。
(項目B14)
前記良性腫瘍は、WT1を発現する、項目B1~B13のいずれか一項に記載のWT1ペプチドもしくはその類縁体、または核酸分子。
(項目B15)
前記良性腫瘍は、家族性大腸腺腫症、非遺伝性の大腸腺腫、膵管内乳頭粘液性腫瘍、脳の髄膜腫、神経鞘腫、各臓器の上皮性の腺腫、乳頭腫、非上皮性の筋腫、脂肪腫、軟骨腫、血管腫からなる群より選択される、項目B1~B14のいずれか一項に記載のWT1ペプチドもしくはその類縁体、または核酸分子。
(項目B16)
前記良性腫瘍は、家族性大腸腺腫症である、項目B1~B15のいずれか一項に記載のWT1ペプチドもしくはその類縁体、または核酸分子。
(項目B17)
1週間に1回投与されることを特徴とする、項目B1~B16のいずれか一項に記載のWT1ペプチドもしくはその類縁体、または核酸分子。
(項目B18)
良性腫瘍の予防または治療のために使用するWT1特異的細胞傷害性T細胞(CTLs)および/またはWT1特異的ヘルパーT細胞を誘導するための、上記項目のいずれか一項に記載のWT1ペプチドもしくはその類縁体、または上記項目のいずれか一項に記載の核酸分子であって、該WT1ペプチドもしくはその類縁体が、良性腫瘍の処置を必要とする被験体由来の末梢血単核球と共に培養され、あるいは、該核酸分子が、該末梢血単核球に導入され、それにより、該WT1特異的CTLsおよび/またはWT1特異的ヘルパーT細胞が誘導される、WT1ペプチドもしくはその類縁体、または核酸分子。
(項目B19)
良性腫瘍の予防または治療のために使用するWT1提示樹状細胞を誘導するための、上記項目のいずれか一項に記載のWT1ペプチドまたはその類縁体、または上記項目のいずれか一項に記載の核酸分子であって、該WT1ペプチドまたはその類縁体が、良性腫瘍の処置を必要とする被験体由来の未熟樹状細胞と共に培養され、あるいは、該核酸分子が、該未熟樹状細胞に導入され、それにより、該WT1提示樹状細胞が誘導される、WT1ペプチドもしくはその類縁体、または核酸分子。
(項目B19A)
項目B1~B17のいずれか1つまたは複数に記載の1つまたは複数の特徴をさらに含む、項目B18またはB19に記載の方法。
(項目B20)
良性腫瘍の予防または治療のために使用する、WT1特異的CTLs、および/またはWT1特異的ヘルパーT細胞を誘導するための、WT1ペプチドもしくはその類縁体、またはそれをコードする核酸分子。
(項目B21)
良性腫瘍の予防または治療のために使用する、WT1提示樹状細胞を誘導するための、WT1ペプチドもしくはその類縁体、またはそれをコードする核酸分子。
(項目B21A)
項目B1~B19のいずれか1つまたは複数に記載の1つまたは複数の特徴をさらに含む、項目B20またはB21に記載の組成物。
(項目B22)
被験体における良性腫瘍を予防または治療するための、WT1特異的CTLs、および/またはWT1特異的ヘルパーT細胞。
(項目B23)
被験体における良性腫瘍を予防または治療するための、WT1提示樹状細胞。
(項目B23A)
項目B1~B19のいずれか1つまたは複数に記載の1つまたは複数の特徴をさらに含む、項目B22またはB23に記載の組成物。
(項目C1)
良性腫瘍の予防または治療薬の製造における、WT1ペプチドまたはその類縁体の使用。
(項目C2)
前記WT1ペプチドまたはその類縁体は、キラー型および/またはヘルパー型を含む、項目C1に記載の使用。
(項目C3)
前記WT1ペプチドまたはその類縁体は、WT1126キラーペプチド、WT1235キラーペプチドおよび/またはWT135ヘルパーペプチド、またはいずれかのアミノ酸配列において、1個~数個のアミノ酸が、欠失、置換、および/または付加されたアミノ酸配列を含み且つCTL誘導活性を有するペプチドを含む、項目C1またはC2に記載の使用。
(項目C4)
前記WT1ペプチドまたはその類縁体が、
RMFPNAPYL (配列番号2)、
RYFPNAPYL (配列番号46)、
YMFPNAPYL (配列番号14)、
CYTWNQMNL (配列番号45)、
CMTWNQMNL (配列番号3)、
C-CYTWNQMNL(配列番号47)(式中、CとCの間の結合はジスルフィド結合を表す。)、および
C-CMTWNQMNL(配列番号48)(式中、CとCの間の結合はジスルフィド結合を表す。)から選択されるいずれかのアミノ酸配列からなるペプチド、またはその薬学上許容される塩である、
項目C1~C3のいずれか一項に記載の使用。
(項目C5)
前記WT1ペプチドまたはその類縁体が、
式(2):
で表される化合物、またはその薬学上許容される塩である、
項目C1~C4のいずれか一項に記載の使用。
(項目C6)
前記WT1ペプチドまたはその類縁体が、
式(3):
で表される化合物、またはその薬学上許容される塩である、
項目C1~C5のいずれか一項に記載の使用。
(項目C7)
前記WT1ペプチドまたはその類縁体が、さらに
以下のアミノ酸配列:
WAPVLDFAPPGASAYGSL (配列番号4)、
CWAPVLDFAPPGASAYGSL (配列番号50)および
WAPVLDFAPPGASAYGSLC (配列番号51)、
からなる群から選択されるアミノ酸配列からなるペプチドまたはその薬学上許容される塩を一つ以上含む組成物である、項目C1~C6のいずれか一項に記載の使用。
(項目C8)
前記WT1ペプチドまたはその類縁体が、
式(2):
で表される化合物またはその薬学上許容される塩と、
アミノ酸配列: WAPVLDFAPPGASAYGSL (配列番号4)からなるペプチドまたはその薬学上許容される塩を含む組成物である、
項目C1~C7のいずれか一項に記載の使用。
(項目C9)
前記WT1ペプチドまたはその類縁体が、
式(3):
で表される化合物またはその薬学上許容される塩と、
アミノ酸配列: WAPVLDFAPPGASAYGSL (配列番号4)からなるペプチドまたはその薬学上許容される塩を含む組成物である、
項目C1~C8のいずれか一項に記載の使用。
(項目C10)
良性腫瘍の予防または治療薬の製造における、WT1ペプチドまたはその類縁体をコードする核酸分子の使用。
(項目C11)
前記核酸分子は、RNAおよび/またはDNAを含む、項目C1~C10のいずれか一項に記載の使用。
(項目C12)
前記WT1ペプチドもしくはその類縁体、または核酸分子が、アジュバントと併用される、項目C1~C11のいずれか一項に記載の使用。
(項目C13)
前記アジュバントがMontanide(登録商標) ISA51アジュバントである、項目C1~C12のいずれか一項に記載の使用。
(項目C14)
前記良性腫瘍は、WT1を発現する、項目C1~C13のいずれか一項に記載の使用。
(項目C15)
前記良性腫瘍は、家族性大腸腺腫症、非遺伝性の大腸腺腫、膵管内乳頭粘液性腫瘍、脳の髄膜腫、神経鞘腫、各臓器の上皮性の腺腫、乳頭腫、非上皮性の筋腫、脂肪腫、軟骨腫、血管腫からなる群より選択される、項目C1~C14のいずれか一項に記載の使用。
(項目C16)
前記良性腫瘍は、家族性大腸腺腫症である、項目C1~C15のいずれか一項に記載の使用。
(項目C17)
前記WT1ペプチドもしくはその類縁体、またはそれをコードする核酸分子が、1週間に1回投与されることを特徴とする、項目C1~C16のいずれか一項に記載の使用。
(項目C18)
良性腫瘍の予防または治療のために使用するWT1特異的細胞傷害性T細胞(CTLs)、および/またはWT1特異的ヘルパーT細胞の製造における、上記項目のいずれか一項に記載のWT1ペプチドもしくはその類縁体、または、上記項目のいずれか一項に記載の核酸分子の使用であって、該WT1ペプチドもしくはその類縁体が、良性腫瘍の処置を必要とする被験体由来の末梢血単核球と共に培養され、あるいは、該核酸分子が、該末梢血単核球に導入され、それにより、該WT1特異的CTLsおよび/またはWT1特異的ヘルパーT細胞が誘導される、使用。
(項目C19)
良性腫瘍の予防または治療のために使用するWT1提示樹状細胞の製造における、上記項目のいずれか一項に記載のWT1ペプチドまたはその類縁体、または上記項目のいずれか一項に記載の核酸分子であって、該WT1ペプチドまたはその類縁体が、良性腫瘍の処置を必要とする被験体由来の未熟樹状細胞と共に培養され、あるいは、該核酸分子が、該未熟樹状細胞に導入され、それにより、該WT1提示樹状細胞が誘導される、WT1ペプチドもしくはその類縁体、または核酸分子。
(項目C19A)
項目C1~C17のいずれか1つまたは複数に記載の1つまたは複数の特徴をさらに含む、項目C18またはC19に記載の方法。
(項目C20)
良性腫瘍の予防または治療のために使用する、WT1特異的CTLs、および/またはWT1特異的ヘルパーT細胞の製造における、WT1ペプチドもしくはその類縁体、またはそれをコードする核酸分子の使用。
(項目C21)
良性腫瘍の予防または治療のために使用する、WT1提示樹状細胞の製造における、WT1ペプチドもしくはその類縁体、またはそれをコードする核酸分子の使用。
(項目C21A)
項目C1~C19のいずれか1つまたは複数に記載の1つまたは複数の特徴をさらに含む、項目C20またはC21に記載の組成物。
(項目C22)
良性腫瘍の予防または治療薬の製造における、WT1特異的CTLs、および/またはWT1特異的ヘルパーT細胞の使用。
(項目C23)
良性腫瘍の予防または治療薬の製造における、WT1提示樹状細胞の使用。
(項目C23A)
項目C1~C19のいずれか1つまたは複数に記載の1つまたは複数の特徴をさらに含む、項目C22またはC23に記載の組成物。
本明細書において「WT1(Wilms Tumor Gene 1)」、「WT1タンパク質」または「WT1ペプチド」とは、ウィルムス腫瘍(WT1)遺伝子産物またはその類縁体の少なくとも一部(全部であってよい)を含むポリペプチドをいう。「WT1タンパク質」としては、具体的には、代表的には、449個のアミノ酸からなるヒトWT1タンパク質(配列番号1)、または、このアミノ酸配列において、1もしくは数個(好ましくは、約2~6個)のアミノ酸が欠失、置換および/もしくは付加されたアミノ酸配列からなるタンパク質が好ましく挙げられる。挿入されるアミノ酸または置換されるアミノ酸は、遺伝子によりコードされる20種類のアミノ酸以外の非天然アミノ酸であってもよい。「WT1ペプチド」とは、WT1タンパク質を構成するアミノ酸配列の一部からなるペプチドをいう。本明細書において、「WT1」または「WT1ペプチド」という場合、特に断らない限り、変異型のWT1を包含するものとする。また、本明細書において、WT1またはWT1ペプチドに言及する場合、特に断らない限り、ヒトのWT1をいう。
RMFPNAPYL (配列番号2)、
CMTWNQMNL (配列番号3)、
ALLPAVPSL (配列番号52)、
SLGEQQYSV (配列番号53)および
RVPGVAPTL (配列番号54)
の中から選ばれるいずれかのアミノ酸配列を含むペプチド、または配列番号2、3、52、53および54の中から選ばれるいずれかのアミノ酸配列中にアミノ酸残基の改変を含有する改変アミノ酸配列を含み且つCTL誘導活性を有するペプチドが挙げられる。さらに好ましくは、配列番号2、3、52、53および54の中から選ばれるいずれかのアミノ酸配列からなるペプチドが挙げられる。
RMFPNAPYL (配列番号2)の改変キラーペプチドである
RYFPNAPYL (配列番号46)(国際公開第2003/106682号参照);
FMFPNAPYL (配列番号13)、
RLFPNAPYL (配列番号18)、
RMMPNAPYL (配列番号25)、
RMFPNAPYV (配列番号28)もしくは
YMFPNAPYL (配列番号14)(国際公開第2009/072610号参照);
CMTWNQMNL (配列番号3)の改変キラーペプチドである
CYTWNQMNL (配列番号45)(国際公開第2002/79253号参照);
Xaa-Met-Thr-Trp-Asn-Gln-Met-Asn-Leu (配列番号55)(本配列中XaaはSerまたはAlaを表す)もしくは
Xaa-Tyr-Thr-Trp-Asn-Gln-Met-Asn-Leu (配列番号56)(本配列中XaaはSer、Ala、Abu、Arg、Lys、Orn、Cit、Lue、PheまたはAsnを表す)(国際公開第2004/026897号参照);
ALLPAVPSL (配列番号52)の改変キラーペプチドである
AYLPAVPSL (配列番号57)(国際公開第2003/106682号参照);
SLGEQQYSV (配列番号53)の改変キラーペプチドである
FLGEQQYSV (配列番号58)、
SMGEQQYSV (配列番号59)もしくは
SLMEQQYSV (配列番号60)(国際公開第2009/072610号参照);または
RVPGVAPTL (配列番号54)の改変キラーペプチドである
RYPGVAPTL (配列番号61)(国際公開2003/106682号参照)。
C-CMTWNQMNL (配列番号48)(式中、CとCの間の結合はジスルフィド結合を表す。)。
式(2):
SGQARMFPNAPYLPSCLES (配列番号69)の改変ヘルパーペプチドである
SGQAYMFPNAPYLPSCLES (配列番号70)(国際公開第2004/063217号参照)
SGQARMFPNAPYLPSC (配列番号71)もしくは
SGQAYMFPNAPYLPSC (配列番号72);または
PGCNKRYFKLSHLQMHSRK (配列番号49)、
PGCNKRYFKLSHLQMHSRKH (配列番号62)、
CNKRYFKLSHLQMHSRK (配列番号64)、
CNKRYFKLSHLQMHSRKH (配列番号65)もしくは
CNKRYFKLSHLQMHSRKHTG (配列番号66);または
WAPVLDFAPPGASAYGSL (配列番号4)、
CWAPVLDFAPPGASAYGSL (配列番号50)もしくは
WAPVLDFAPPGASAYGSLC (配列番号51)。
式(2):
以下のアミノ酸配列:
CNKRYFKLSHLQMHSRK (配列番号63)、
CNKRYFKLSHLQMHSRKH (配列番号64)、
CNKRYFKLSHLQMHSRKHTG (配列番号65)、
WAPVLDFAPPGASAYGSL (配列番号4)、
CWAPVLDFAPPGASAYGSL (配列番号50)および
WAPVLDFAPPGASAYGSLC (配列番号51)
からなる群から選択されるアミノ酸配列からなるペプチド、または
その薬学上許容される塩を一つ以上含む組成物;
式(2):
で表される化合物またはその薬学上許容される塩と、
アミノ酸配列: WAPVLDFAPPGASAYGSL (配列番号4)からなるペプチドまたはその薬学上許容される塩を含む組成物;および
式(3):
で表される化合物またはその薬学上許容される塩と、
アミノ酸配列: WAPVLDFAPPGASAYGSL (配列番号4)からなるペプチドまたはその薬学上許容される塩を含む組成物。
本開示において、WT1ペプチドとしては、細胞傷害性T細胞またはヘルパーT細胞の活性化を誘導する、WT1タンパク質由来のペプチドが挙げられる。WT1タンパク質としては、細胞傷害性T細胞の活性化を誘導するWT1キラーペプチドと、ヘルパーT細胞の活性化を誘導するWT1ヘルパーペプチドとが挙げられる。WT1キラーペプチドとしては、WT1タンパク質由来の8~12個のアミノ酸からなるペプチドが挙げられ、好ましくは8~9個のアミノ酸からなるペプチドが挙げられる。また、特に好ましくは、WT1126ペプチド:Arg Met Phe Pro Asn Ala Pro Tyr Leu(配列番号2)、またはWT1235ペプチド:Cys Met Thr Trp Asn Gln Met Asn Leu(配列番号3)、または式(3)
に示される、WT1126ペプチドとWT1235ペプチドとのコンジュゲートである。WT1ヘルパーペプチドとしては、WT1タンパク質由来の14~20個のアミノ酸からなるペプチドが挙げられ、好ましくは16~18個のアミノ酸からなるペプチドが挙げられる。また、特に好ましくは、WT135ペプチド(Trp Ala Pro Val Leu Asp Phe Ala Pro Pro Gly Ala Ser Ala Tyr Gly Ser Leu;配列番号4)、またはWT1332ペプチド:Lys Arg Tyr Phe Lys Leu Ser His Leu Gln Met His Ser Arg Lys His)(配列番号5)である。WT1ペプチドにおいて1もしくは数個のアミノ酸が欠失、置換もしくは付加された改変ペプチドも、本開示におけるWT1ペプチドとして用いることができる。このような改変ペプチドとしては、WT1126ペプチドの改変ペプチド、WT1235ペプチドの改変ペプチド、WT135ペプチドの改変ペプチド、およびWT1332ペプチドの改変ペプチドが挙げられる。
WT1126P1Gペプチド(GMFPNAPYL;配列番号6)
WT1126P1Aペプチド(AMFPNAPYL;配列番号7)
WT1126P1Vペプチド(VMFPNAPYL;配列番号8)
WT1126P1Lペプチド(LMFPNAPYL;配列番号9)
WT1126P1Iペプチド(IMFPNAPYL;配列番号10)
WT1126P1Mペプチド(MMFPNAPYL;配列番号11)
WT1126P1Wペプチド(WMFPNAPYL;配列番号12)
WT1126P1Fペプチド(FMFPNAPYL;配列番号13)
WT1126P1Yペプチド(YMFPNAPYL;配列番号14)
WT1126P2Vペプチド(RVFPNAPYL;配列番号15)
WT1126P2Qペプチド(RQFPNAPYL;配列番号16)
WT1126P2Aペプチド(RAFPNAPYL;配列番号17)
WT1126P2Lペプチド(RLFPNAPYL;配列番号18)
WT1126P2Iペプチド(RIFPNAPYL;配列番号19)
WT1126P3Iペプチド(RMIPNAPYL;配列番号20)
WT1126P3Lペプチド(RMLPNAPYL;配列番号21)
WT1126P3Gペプチド(RMGPNAPYL;配列番号22)
WT1126P3Aペプチド(RMAPNAPYL;配列番号23)
WT1126P3Vペプチド(RMVPNAPYL;配列番号24)
WT1126P3Mペプチド(RMMPNAPYL;配列番号25)
WT1126P3Pペプチド(RMPPNAPYL;配列番号26)
WT1126P3Wペプチド(RMWPNAPYL;配列番号27)
WT1126P9Vペプチド(RMFPNAPYV;配列番号28)
WT1126P9Aペプチド(RMFPNAPYA;配列番号29)
WT1126P9Iペプチド(RMFPNAPYI;配列番号30)
WT1126P9Mペプチド(RMFPNAPYM;配列番号31)
WT1126P1Dペプチド(DMFPNAPYL;配列番号32)
WT1126P1Eペプチド(EMFPNAPYL;配列番号33)
WT1126P1Hペプチド(HMFPNAPYL;配列番号34)
WT1126P1Kペプチド(KMFPNAPYL;配列番号35)
WT1126P1Nペプチド(NMFPNAPYL;配列番号36)
WT1126P1Pペプチド(PMFPNAPYL;配列番号37)
WT1126P1Qペプチド(QMFPNAPYL;配列番号38)
WT1126P1Sペプチド(SMFPNAPYL;配列番号39)
WT1126P1Tペプチド(TMFPNAPYL;配列番号40)
WT1126P2I&P9Iペプチド(RIFPNAPYI;配列番号41)
WT1126P2I&P9Vペプチド(RIFPNAPYV;配列番号42)
WT1126P2L&P9Iペプチド(RLFPNAPYI;配列番号43)
WT1126P2L&P9Vペプチド(RLFPNAPYV;配列番号44)
WT1235ペプチドの改変ペプチドとしては、WT1235:Cys Met Thr Trp Asn Gln Met Asn Leu(配列番号3)、またはWT1235m:Cys Tyr Thr Trp Asn Gln Met Asn Leu(配列番号45)を含む、あるいはからなるペプチド、配列番号3または45のアミノ酸配列を含む、あるいはからなるペプチドの二量体、配列番号3または45のアミノ酸配列を含む、あるいはからなるペプチドのシスチン体、および上記アミノ酸配列において1個ないし数個のアミノ酸が置換、欠失または付加されているアミノ酸配列を含む、あるいはからなるペプチドなどが例示されるが、これらに限定されない。WT1235ペプチドの改変ペプチドの中でも、WT1235mペプチドは、HLA-A*24:02(日本人最多のHLA型)拘束性WT1ペプチドであり、野生型のWT1235ペプチドよりも治療有効性が高く、水への可溶性に優れることから、特に好ましい。
Cys-Cys Tyr Thr Trp Asn Gln Met Asn Leu(配列番号47)(式中、CとCの間の結合はジスルフィド結合を表す。)、またはCys-Cys Met Thr Trp Asn Gln Met Asn Leu(配列番号48)(式中、CとCの間の結合はジスルフィド結合を表す。)は、N末端システイン残基のチオール基を修飾することによって、特に物理化学的性質および安定性に優れる(国際公開第2007/063903号参照)。
R1は、水素原子または腫瘍抗原ペプチドBを表し、
腫瘍抗原ペプチドBは、腫瘍抗原ペプチドAとは配列が異なり且つ以下のアミノ酸配列:CMTWNQMNL(配列番号3)およびCYTWNQMNL(配列番号45)の中から選ばれるいずれかのアミノ酸配列からなるペプチドを表し、腫瘍抗原ペプチドBのシステイン残基のチオエーテル基が式(1)中のチオエーテル基と結合する。]
で表される化合物またはその薬学上許容される塩を含むものであってもよい。
本実施形態における「腫瘍抗原ペプチドA」とは、7~30残基のアミノ酸からなるMHCクラスI拘束性WT1ペプチドである。式(1)において腫瘍抗原ペプチドAは、N末端アミノ酸のアミノ基が式(1)中のYaと結合し、C末端アミノ酸のカルボニル基が式(1)中の水酸基と結合する。
また、上記式(1)で表される化合物は、式(2):
第3位:フェニルアラニン、トリプトファン、バリン、イソロイシン、ロイシン、メチオニン、
第6位:バリン、イソロイシン、メチオニン、アスパラギン酸、グルタミン酸、
第8位:アスパラギン、セリン、スレオニン、グルタミン、リジン、アスパラギン酸、
第11位:アスパラギン酸、グルタミン酸、グルタミン、
の中から選択されるいずれかのアミノ酸残基に置換されたアミノ酸配列からなるペプチドが好適である。
以下に本開示の好ましい実施形態を説明する。以下に提供される実施形態は、本開示のよりよい理解のために提供されるものであり、本開示の範囲は以下の記載に限定されるべきでないことが理解される。従って、当業者は、本明細書中の記載を参酌して、本開示の範囲内で適宜改変を行うことができることは明らかである。また、本開示の以下の実施形態は単独でも使用されあるいはそれらを組み合わせて使用することができることが理解される。
一つの局面において、本開示は、WT1ペプチドまたはその類縁体を含む、良性腫瘍の予防または治療薬を提供する。WT1は、悪性腫瘍のがん細胞において高発現していることは示されていたが、良性腫瘍における発現は未知であった。本開示は、良性腫瘍の細胞においてWT1タンパク質が発現するとの驚くべき発見に基づくものであり、良性腫瘍の新たな治療法を提供するものである。
別の局面において、本開示は、良性腫瘍(例えば、家族性大腸腺腫症)の処置を必要とする被験体由来の末梢血単核球を、本明細書に記載のいずれかのWT1ペプチドまたはその類縁体の存在下で培養する、または、それらをコードする核酸分子を上記末梢血単核球に導入することによって、上記末梢単核球からWT1特異的細胞傷害性T細胞(CTLs)、および/またはWT1特異的ヘルパーT細胞を誘導する工程を含むことを特徴とする、良性腫瘍(例えば、家族性大腸腺腫症)の予防または治療のために使用するWT1特異的CTLsおよび/またはWT1特異的ヘルパーT細胞の誘導方法を提供する。(予防または治療薬)に記載される任意の説明は、この誘導方法においても当てはまることが当業者に理解される。
本開示のペプチドまたは誘導体、あるいは核酸分子は、当技術分野において通常用いられる製造法によって製造することができる。これらの製造法は、免疫学的手法、分子生物学的手法、生化学的手法、微生物学的手法に習熟している者の知識に基づき、適宜改良することができる。具体的には、本開示のペプチドまたは誘導体、あるいは核酸分子は、天然のWT1タンパク質のアミノ酸配列(例えば、配列番号1)または核酸配列に基づく設計を行ってもよく、微生物発現系などによって、製造してもよい。製造法における出発原料および中間体は、市販品として購入可能であるか、または公知文献に記載された方法もしくは公知化合物から公知の方法に準じて入手可能である。また、これらの出発原料および中間体は、製造工程に支障をきたさない限り、それらの類縁体を用いてもよい。
本明細書において用いられる免疫学的手法、分子生物学的手法、生化学的手法、微生物学的手法は、当該分野において周知であり慣用されるものであり、Sambrook J. et al.(1989). Molecular Cloning: A Laboratory Manual, Cold Spring Harborおよびその3rd Ed.(2001); Ausubel, F.M.(1987).Current Protocols in Molecular Biology, Greene Pub. Associates and Wiley-Interscience; Ausubel, F.M.(1989). Short Protocols in Molecular Biology: A Compendium of Methods from Current Protocols in Molecular Biology, Greene Pub. Associates and Wiley-Interscience; Innis, M.A.(1990).PCR Protocols: A Guide to Methods and Applications, Academic Press; Ausubel, F.M.(1992).Short Protocols in Molecular Biology: A Compendium of Methods from Current Protocols in Molecular Biology, Greene Pub. Associates; Ausubel, F.M. (1995).Short Protocols in Molecular Biology: A Compendium of Methods from Current Protocols in Molecular Biology, Greene Pub. Associates; Innis, M.A. et al.(1995).PCR Strategies, Academic Press; Ausubel, F.M.(1999).Short Protocols in Molecular Biology: A Compendium of Methods from Current Protocols in Molecular Biology, Wiley, and annual updates; Sninsky, J.J. et al.(1999). PCR Applications: Protocols for Functional Genomics, Academic Press、別冊実験医学「遺伝子導入&発現解析実験法」羊土社、1997などに記載されており、これらは本明細書において関連する部分(全部であり得る)が参考として援用される。
本明細書において「または」は、文章中に列挙されている事項の「少なくとも1つ以上」を採用できるときに使用される。「もしくは」も同様である。本明細書において「2つの値の範囲内」と明記した場合、その範囲には2つの値自体も含む。
以下の実施例において、マウスは、ApcMin/+(C57BL/6J)マウス(Jackson研究所、Bar Harbor、Maine、USA)を使用した。マウスは、大阪大学動物実験規程を遵守し、大阪大学医学部付属動物実験施設の特定微生物フリー(SPF)封じ込め施設にて飼育した。
以下の実施例において、MHC class I(H-2Db)-結合ペプチドであるWT1126-134(RMFPNAPYL、9a.a)、およびMHC class II(H-2I-Ab)-結合ペプチドであるWT135-52(WAPVLDFAPPGASAYGSL、18a.a.)は、SIGMA Genosys(石狩、日本)から購入した。式(3)の化合物およびWT135-52 ヘルパーペプチドの混合物に用いる各原料は、大日本住友製薬株式会社(大阪、日本)より供与された。ペプチドは、使用まで-20℃において保存した。ペプチドはリン酸緩衝生理食塩水(PBS;137mM NaCl、2.7mM KCl、8.1mM Na2HPO4、1.47mM KH2PO4)に溶解し、-20℃で保存した。フロインドの不完全アジュバント(IFA)、Montanide ISA 51は、Seppic S.A.(Orsay、France)より購入した。
本実施例では、ヒト家族性大腸腺腫症患者の腺腫において、WT1タンパク質が発現されたことを示す。
家族性大腸腺腫症のヒト患者の小腸および大腸を、ホルマリンにより固定後、パラフィンで包埋し、パラフィンブロックを3μmの厚さに薄切した。内因性ペルオキシダーゼ反応を軽減させるため、パラフィン切片を3% H2O2溶液で処理後、キシレンで脱パラフィンし、エタノールで段階的に再水和した後、10mMのトリスEDTA緩衝液((10mM Tris、1mM EDTA(pH9.0))中で、オイルバスにおいて70℃で25分間、110℃で25分間加熱し、3時間室温で冷まして抗原賦活化した。処理した切片を、PBST(PBS+0.05% Tween20(登録商標))で100倍希釈した抗WT1 6FH2マウス単クローン抗体(Dako Cytomation,Inc.、Carpinteria、CA)により、4℃で一晩反応させた。その後、Dako REAL Envision HRP RABBIT/MOUSE(Dako Cytomation)を37℃で30分間反応させた。Dako REAL Envision Detection System Peroxidase/DAB+、Rabbit/Mouse(Dako Cytomation)を用い、取扱説明書に従い、WT1タンパク質を可視化させた。その後切片をヘマトキシリン染色した。
実施例1の結果を、表1および図1に示す。表1は、各患者由来の標本における、腺腫、正常腺管および線維芽細胞におけるWT1タンパク質の発現量を示す。表1において、腺腫におけるWT1タンパク質の発現量が、正常腺管における発現量よりも顕著に増大していることが示される。図1において、薄い染色はWT1タンパク質を示し、濃く円形に染色された領域は核を示す。腺腫においてはWT1タンパク質の発現が認められたが、正常腺管においては、WT1タンパク質の発現が認められなかった。
本実施例では、家族性大腸腺腫症のモデルマウスであるAPCMin/+マウスの小腸腺腫において、WT1タンパク質が発現したことを示す。
20週齢のApcMin/+(C57BL/6J)マウスの小腸および大腸を、ホルマリンにより固定後、パラフィンで包埋し、パラフィンブロックを3μmの厚さに薄切した。パラフィン切片をキシレンで脱パラフィンし、エタノールで段階的に再水和した後、10mMのクエン酸緩衝液((10mM クエン酸ナトリウム、0.05% Tween20(pH6.0))中で、15分間マイクロ波を照射し、抗原賦活化した。処理した切片を、ヤギ血清含有PBSで100倍希釈した抗WT1 6FH2マウス単クローン抗体(Dako Cytomation,Inc.、Carpinteria、CA)により、4℃で一晩反応させた。そして、ヤギ血清含有PBSで100倍希釈したビオチン化抗マウスIgG抗体(Vector Labs.、Burlingame、CA)を、37℃で30分間反応させた。内因性ペルオキシダーゼ反応を軽減させるため、3% H2O2溶液で処理後、Vectastain ABC kit(Vector Labs.)を用い、取扱説明書に従い、WT1タンパク質を可視化させた。その後切片をヘマトキシリン染色した。
実施例2の結果を図2および3に示す。図2は、上から順に、5倍、10倍、20倍の倍率での顕微鏡写真を示し、図3は、40倍の倍率での顕微鏡写真を示す。図2および3において、濃い染色はWT1タンパク質を示し、円形に染色された領域は核を示す。家族性大腸腺腫症のモデルマウスであるApcMin/+マウスの小腸腺腫において、WT1タンパク質の発現が認められた。
本実施例では、APCMin/+マウスに、WT1ペプチドワクチンを投与した実験の投与スキームを示す。
WT1ペプチドワクチンの投与スキームを図4に示す。100μgのWT1126-134 キラーペプチドと45μgのWT135-52 ヘルパーペプチドとの混合物、またはPBSを、それぞれIFA Montanide ISA51アジュバントでエマルション化し、それぞれWT1ペプチドワクチンまたはコントロールワクチンとした。4~5週齢のApcMin/+のマウスの脇腹に、WT1ペプチドワクチンまたはコントロールワクチンを皮内投与した。免疫は、生後5週目から開始し、1週間に1回の頻度で8回行った。最終免疫の10日後に免疫したマウスを安楽死させ、さらなる解析を行った。なお、WT1ペプチドワクチンの投与は、以下の表3において示されるとおり、臓器傷害をもたらさないものであった。
本実施例では、APCMin/+マウスに、WT1ペプチドワクチンを投与することによって、腺腫の発症が予防され、腺腫を治療したことを示す。
実施例3に記載の投与スキームによって得られたマウスを使用した。最終免疫の10日後に安楽死させたマウスから全腸を採取した。小腸は8~10分画に分け、大腸は盲腸と上行結腸とに分けた。各分画は縦方向に切開し、PBSにて洗浄後、ろ紙に挟んで、4%パラホルムアルデヒド/リン酸緩衝液において、4℃で24時間以上固定した。固定した腸組織は、1% メチレンブルー液で染色後、実体顕微鏡下で観察し、腺腫(ポリープ)数を数えた。WT1ペプチドワクチン投与群とコントロールワクチン投与群との間の腺腫(ポリープ)数の差は、Student’s t testにより統計解析した。
実施例4の結果を図5に示す。グラフの横軸は左側にWT1ワクチンを投与した群を、右側にコントロールワクチンを投与した群を示し、縦軸は小腸あたりの腺腫数を示す。WT1ペプチドワクチン投与群では小腸あたりの腺腫数の平均値が約29個であったのに対し、コントロールワクチン投与群では約34個であり、WT1ペプチドワクチンの投与によって、腺腫の発症が有意に抑制され、さらに腺腫が治療されたことが示された。
本実施例では、APCMin/+マウスに、WT1ペプチドワクチンを投与することによって、WT1テトラマー+CD3+CD8+T細胞が増加したことを示す。
実施例3に記載の投与スキームによって得られたマウスを使用した。最終免疫の10日後に安楽死させたマウスから脾細胞を採取した。赤血球を溶解後、細胞をH-2Db WT1 テトラマー(MBL、愛知、日本)で染色し、さらに抗マウスCD3抗体(17A2、BioLegend、San Diego、CA)および抗マウスCD8抗体(KT15、MBL、愛知、日本)で染色した。染色した細胞はFACSCantoにて解析し、CD3+CD8+T細胞中のH-2Db WT1 テトラマー+CD3+CD8+T細胞の頻度を測定した。WT1ペプチドワクチン投与群とコントロールワクチン投与群との間のH-2Db WT1 テトラマー+CD3+CD8+T細胞の頻度の差は、Student’s t testにより統計解析した。
実施例5の結果を図6に示す。グラフの横軸は左側にWT1ペプチドワクチンを投与した群を、右側にコントロールワクチンを投与した群を示し、縦軸はCD3+CD8+細胞中のH-2Db WT1 テトラマー頻度を示す。WT1ワクチン投与群では、CD3+CD8+T細胞中のH-2Db WT1 テトラマー+CD3+CD8+T細胞の頻度の平均値が約0.6%であったのに対し、コントロールワクチン投与群では約0.2%であり、WT1ワクチンの投与によって、WT1テトラマー+CD3+CD8+T細胞が増加したことが示された。
本実施例では、実施例4における腺腫数と、実施例5におけるWT1テトラマー+CD3+CD8+T細胞に相関が認められたことを示す。
実施例4から得られた小腸あたりの腺腫数と、実施例5から得られたCD3+CD8+T細胞中のH-2Db WT1 テトラマー+CD3+CD8+T細胞の頻度とを、Microsoft Excel(登録商標)によって散布図にプロットした。さらにこれらの結果について、Excelのピアソン相関係数によって相関関係を統計解析した。
実施例6の結果から、WT1ペプチドワクチン投与群とコントロールワクチン投与群との間の、腺腫(ポリープ)数およびWT1テトラマー頻度の差は、弱いながらも相関関係を示すことが認められた。
本実施例では、APCMin/+マウスに、キラー型およびヘルパー型の両方のWT1ペプチドワクチンを投与する場合と、キラー型またはヘルパー型のWT1ペプチドワクチンの単剤のみを投与する場合との、腺腫の発症の予防および治療効果における比較を示す。
(実験手法)
WT1ペプチドワクチンの投与スキームを図8に示す。100μgのWT1126-134 キラーペプチドと45μgのWT135-52 ヘルパーペプチドとの混合物、100μgのWT1126-134 キラーペプチド単独、45μgのWT135-52 ヘルパーペプチド単独またはPBSを、それぞれIFA Montanide ISA51アジュバントでエマルション化する。5週齢のApcMin/+のマウスの脇腹に、キラーペプチドとヘルパーペプチドとの混合物、キラーペプチド単独、ヘルパーペプチド単独、またはPBSのみのコントロールワクチンを皮内投与する。免疫は、生後5週目から開始し、1週間に1回の頻度で8回行う。最終免疫の10日後に免疫するマウスを安楽死させ、さらなる解析を行う。
本実施例では、非遺伝性の大腸腺腫においても、WT1が発現することを示す。
非遺伝性の大腸腺腫を発症したヒト患者(3名)から腺腫組織を摘出し、実施例1と同様の手法によって、WT1タンパク質および核を染色した。
実施例8の結果を図9に示す。図9において、左から1番目は、正常腺管の顕微鏡画像を示し、右側の3枚の画像は、非遺伝性の大腸腺腫の腺腫組織の顕微鏡画像を示す。図9において、薄い染色はWT1タンパク質を示し、濃く円形または楕円形に染色された領域は核を示す。腺腫においてはWT1タンパク質の発現が認められたが、正常腺管においては、WT1タンパク質の発現が認められなかった。
本実施例では、APCMin/+マウスに、WT1ペプチドワクチンである、式(3)の化合物およびWT135-52 ヘルパーペプチドの混合物を投与した実験の投与スキームを示す。
WT1ペプチドワクチンの投与スキームを図10に示す。WT1ペプチドワクチンをPBSで溶解し、WT1ペプチドワクチンを含む溶解液およびPBS単独を、IFA Montanide ISA51アジュバントでエマルション化し、それぞれWT1ペプチドワクチンまたはコントロールワクチンとした。4~5週齢のApcMin/+のマウスの脇腹に、WT1ペプチドワクチンまたはコントロールワクチンを皮内投与した。免疫は、生後4~5週目から開始し、1週間に1回の頻度で6回行った。最終免疫の7日後に免疫したマウスを安楽死させ、さらなる解析を行った。
本実施例では、APCMin/+マウスに、式(3)の化合物およびWT135-52 ヘルパーペプチドの混合物を投与することによって、腺腫の発症が予防され、腺腫を治療したことを示す。なお、式(3)の化合物およびWT135-52 ヘルパーペプチドの混合物の投与は、以下の表4において示されるとおり、臓器傷害をもたらさないものであった。
実施例9に記載の投与スキームによって得られたマウスを使用した。最終免疫の7日後に安楽死させたマウスから各臓器を摘出し、その重量を測定した。小腸は8~10分画に分けた。各分画は縦方向に切開し、PBSにて洗浄後、ろ紙に挟んで、4%パラホルムアルデヒド/リン酸緩衝液において、4℃で24時間以上固定した。固定した腸組織は、1% メチレンブルー液で染色後、実体顕微鏡下で観察し、腺腫(ポリープ)数を数えた。WT1ペプチドワクチン投与群とコントロールワクチン投与群との間の腺腫(ポリープ)数の差は、Student’s t testにより統計解析した。
実施例10の結果を図11に示す。グラフの横軸は左側にWT1ペプチドワクチンを投与した群を、右側にコントロールワクチンを投与した群を示し、縦軸は小腸あたりの腺腫数を示す。WT1ペプチドワクチン投与群では小腸あたりの腺腫数の平均値が約37.2個であったのに対し、コントロールワクチン投与群では約43.7個であった。したがって、WT1ペプチドワクチンの投与によって、腺腫の発症が抑制され、さらに腺腫が治療される傾向が認められた。
本実施例では、APCMin/+マウスに、式(3)の化合物およびWT135-52 ヘルパーペプチドの混合物を投与することによって、WT1テトラマー+CD3+CD8+T細胞が増加したことを示す。
実施例9に記載の投与スキームによって得られたマウスを使用した。最終免疫の7日後に安楽死させたマウスから脾細胞を採取した。赤血球を溶解後、細胞をH-2Db WT1 テトラマー(MBL、愛知、日本)で染色し、さらに抗マウスCD3抗体(17A2、BioLegend、San Diego、CA)、抗マウスCD8抗体(KT15、MBL、愛知、日本)および7-AAD Viability Staining Solution(Bioscience、San Diego、CA)で染色した。染色した細胞はFACSCantoにて解析し、7-AAD+の死細胞分画を除去し、CD3+CD8+T細胞中のH-2Db WT1 テトラマー+CD3+CD8+T細胞の頻度を測定した。WT1ペプチドワクチン投与群とコントロールワクチン投与群との間のH-2Db WT1 テトラマー+CD3+CD8+T細胞の頻度の差は、Student’s t testにより統計解析した。
実施例11の結果を図12に示す。グラフの横軸は左側にWT1ペプチドワクチンを投与した群を、右側にコントロールワクチンを投与した群を示し、縦軸はCD3+CD8+細胞中のH-2Db WT1 テトラマー頻度を示す。WT1ペプチドワクチン投与群では、CD3+CD8+T細胞中のH-2Db WT1 テトラマー+CD3+CD8+T細胞の頻度の平均値が約0.13%であったのに対し、コントロールワクチン投与群では約0.04%であり、WT1ペプチドワクチンの投与によって、WT1テトラマー+CD3+CD8+T細胞が増加したことが示された。
本実施例では、実施例10における腺腫数と、実施例11におけるWT1テトラマー+CD3+CD8+T細胞に相関が認められたことを示す。
実施例10から得られた小腸あたりの腺腫数と、実施例11から得られたCD3+CD8+T細胞中のH-2Db WT1 テトラマー+CD3+CD8+T細胞の頻度とを、Microsoft Excel(登録商標)によって散布図にプロットした。さらにこれらの結果について、Excelのピアソン相関係数によって相関関係を統計解析した。
実施例12の結果から、WT1ペプチドワクチン投与群とコントロールワクチン投与群との間の、腺腫(ポリープ)数およびWT1テトラマー頻度の差は、高い相関関係を示し、WT1特異的免疫が腫瘍の抑制に強く作用していることが実証された。
以上のように、本開示の好ましい実施形態を用いて本開示を例示してきたが、本開示は、特許請求の範囲によってのみその範囲が解釈されるべきであることが理解される。本明細書において引用した特許、特許出願および他の文献は、その内容自体が具体的に本明細書に記載されているのと同様にその内容が本明細書に対する参考として援用されるべきであることが理解される。本願は日本国特許庁に2018年10月5日に提出された特願2018-190461に対して優先権主張を伴うものであり、その内容はすべて本明細書に対する参考として援用されるべきであることが理解される。
配列番号2 ヒトWT1126ペプチドのアミノ酸配列
配列番号3 ヒトWT1235ペプチドのアミノ酸配列
配列番号4 ヒトWT135ペプチドのアミノ酸配列
配列番号5 ヒトWT1332ペプチドのアミノ酸配列
配列番号6 ヒトWT1126P1Gペプチドのアミノ酸配列
配列番号7 ヒトWT1126P1Aペプチドのアミノ酸配列
配列番号8 ヒトWT1126P1Vペプチドのアミノ酸配列
配列番号9 ヒトWT1126P1Lペプチドのアミノ酸配列
配列番号10 ヒトWT1126P1Iペプチドのアミノ酸配列
配列番号11 ヒトWT1126P1Mペプチドのアミノ酸配列
配列番号12 ヒトWT1126P1Wペプチドのアミノ酸配列
配列番号13 ヒトWT1126P1Fペプチドのアミノ酸配列
配列番号14 ヒトWT1126P1Yペプチドのアミノ酸配列
配列番号15 ヒトWT1126P2Vペプチドのアミノ酸配列
配列番号16 ヒトWT1126P2Qペプチドのアミノ酸配列
配列番号17 ヒトWT1126P2Aペプチドのアミノ酸配列
配列番号18 ヒトWT1126P2Lペプチドのアミノ酸配列
配列番号19 ヒトWT1126P2Iペプチドのアミノ酸配列
配列番号20 ヒトWT1126P3Iペプチドのアミノ酸配列
配列番号21 ヒトWT1126P3Lペプチドのアミノ酸配列
配列番号22 ヒトWT1126P3Gペプチドのアミノ酸配列
配列番号23 ヒトWT1126P3Aペプチドのアミノ酸配列
配列番号24 ヒトWT1126P3Vペプチドのアミノ酸配列
配列番号25 ヒトWT1126P3Mペプチドのアミノ酸配列
配列番号26 ヒトWT1126P3Pペプチドのアミノ酸配列
配列番号27 ヒトWT1126P3Wペプチドのアミノ酸配列
配列番号28 ヒトWT1126P9Vペプチドのアミノ酸配列
配列番号29 ヒトWT1126P9Aペプチドのアミノ酸配列
配列番号30 ヒトWT1126P9Iペプチドのアミノ酸配列
配列番号31 ヒトWT1126P9Mペプチドのアミノ酸配列
配列番号32 ヒトWT1126P1Dペプチドのアミノ酸配列
配列番号33 ヒトWT1126P1Eペプチドのアミノ酸配列
配列番号34 ヒトWT1126P1Hペプチドのアミノ酸配列
配列番号35 ヒトWT1126P1Kペプチドのアミノ酸配列
配列番号36 ヒトWT1126P1Nペプチドのアミノ酸配列
配列番号37 ヒトWT1126P1Pペプチドのアミノ酸配列
配列番号38 ヒトWT1126P1Qペプチドのアミノ酸配列
配列番号39 ヒトWT1126P1Sペプチドのアミノ酸配列
配列番号40 ヒトWT1126P1Tペプチドのアミノ酸配列
配列番号41 ヒトWT1126P2I&P9Iペプチドのアミノ酸配列
配列番号42 ヒトWT1126P2I&P9Vペプチドのアミノ酸配列
配列番号43 ヒトWT1126P2L&P9Iペプチドのアミノ酸配列
配列番号44 ヒトWT1126P2L&P9Vペプチドのアミノ酸配列
配列番号45 ヒトWT1235mペプチドのアミノ酸配列
配列番号46 ヒトWT1126ペプチドの改変体のアミノ酸配列
配列番号47 ヒトWT1235ペプチドの改変体のアミノ酸配列
配列番号48 ヒトWT1235ペプチドの改変体のアミノ酸配列
配列番号49 ヒトWT1122ペプチドの改変体のアミノ酸配列
配列番号50 ヒトWT135ペプチドの改変体のアミノ酸配列
配列番号51 ヒトWT135ペプチドの改変体のアミノ酸配列
配列番号52 ヒトWT110ペプチドのアミノ酸配列
配列番号53 ヒトWT1187ペプチドのアミノ酸配列
配列番号54 ヒトWT1126ペプチドの改変体のアミノ酸配列
配列番号55 ヒトWT1235ペプチドの改変体のアミノ酸配列
配列番号56 ヒトWT1235ペプチドの改変体のアミノ酸配列
配列番号57 ヒトWT1235ペプチドの改変体のアミノ酸配列
配列番号58 ヒトWT110ペプチドの改変体のアミノ酸配列
配列番号59 ヒトWT1187ペプチドの改変体のアミノ酸配列
配列番号60 ヒトWT1187ペプチドの改変体のアミノ酸配列
配列番号61 ヒトWT1126ペプチドの改変体のアミノ酸配列
配列番号62 ヒトWT1122ペプチドの改変体のアミノ酸配列
配列番号63 ヒトWT135ペプチドの改変体のアミノ酸配列
配列番号64 ヒトWT135ペプチドの改変体のアミノ酸配列
配列番号65 ヒトWT135ペプチドの改変体のアミノ酸配列
配列番号66 ヒトWT135ペプチドの改変体のアミノ酸配列
配列番号67 ヒトWT135ペプチドの改変体のアミノ酸配列
配列番号68 ヒトWT137ペプチドのアミノ酸配列
配列番号69 ヒトWT1122ペプチドのアミノ酸配列
配列番号70 ヒトWT1122ペプチドの改変体のアミノ酸配列
配列番号71 ヒトWT1122ペプチドの改変体のアミノ酸配列
配列番号72 ヒトWT1122ペプチドの改変体のアミノ酸配列
Claims (26)
- WT1ペプチドまたはその類縁体を含む、良性腫瘍の予防または治療薬。
- 前記WT1ペプチドまたはその類縁体は、キラー型および/またはヘルパー型を含む、請求項1に記載の予防または治療薬。
- 前記WT1ペプチドまたはその類縁体は、WT1126キラーペプチド、WT1235キラーペプチドおよび/またはWT135ヘルパーペプチド、またはいずれかのアミノ酸配列において、1個~数個のアミノ酸が、欠失、置換、および/または付加されたアミノ酸配列を含み且つCTL誘導活性を有するペプチドを含む、請求項1または2に記載の予防または治療薬。
- 前記WT1ペプチドまたはその類縁体は、
RMFPNAPYL (配列番号2)、
RYFPNAPYL (配列番号46)、
YMFPNAPYL (配列番号14)、
CYTWNQMNL (配列番号45)、
CMTWNQMNL (配列番号3)、
C-CYTWNQMNL(配列番号47)(式中、CとCの間の結合はジスルフィド結合を表す。)、および
C-CMTWNQMNL(配列番号48)(式中、CとCの間の結合はジスルフィド結合を表す。)から選択されるいずれかのアミノ酸配列からなるペプチド、またはその薬学上許容される塩である、
請求項3に記載の予防または治療薬。 - 前記WT1ペプチドまたはその類縁体が、さらに
以下のアミノ酸配列:
WAPVLDFAPPGASAYGSL (配列番号4)、
CWAPVLDFAPPGASAYGSL (配列番号50)および
WAPVLDFAPPGASAYGSLC (配列番号51)、
からなる群から選択されるアミノ酸配列からなるペプチドまたはその薬学上許容される塩を一つ以上含む組成物である、請求項1~6のいずれか一項に記載の予防または治療薬。 - WT1ペプチドまたはその類縁体をコードする核酸分子を含む、良性腫瘍の予防または治療薬。
- 前記核酸分子は、RNAおよび/またはDNAを含む、請求項10に記載の予防または治療薬。
- アジュバントをさらに含む、請求項1~11のいずれか一項に記載の予防または治療薬。
- 前記アジュバントがMontanide(登録商標) ISA51アジュバントである、請求項12に記載の予防または治療薬。
- 前記良性腫瘍は、WT1を発現する、請求項1~13のいずれか一項に記載の予防または治療薬。
- 前記良性腫瘍は、家族性大腸腺腫症、非遺伝性の大腸腺腫、膵管内乳頭粘液性腫瘍、脳の髄膜腫、神経鞘腫、各臓器の上皮性の腺腫、乳頭腫、非上皮性の筋腫、脂肪腫、軟骨腫、血管腫からなる群より選択される、請求項1~14のいずれか一項に記載の予防または治療薬。
- 前記良性腫瘍は、家族性大腸腺腫症である、請求項1~15のいずれか一項に記載の予防または治療薬。
- 1週間に1回投与されることを特徴とする、請求項1~16のいずれか一項に記載の予防または治療薬。
- 良性腫瘍の処置を必要とする被験体由来の末梢血単核球を、請求項1~9のいずれか一項に記載のWT1ペプチドまたはその類縁体の存在下で培養し、あるいは、請求項10または11に記載のWT1ペプチドまたはその類縁体をコードする核酸分子を該末梢血単核球に導入し、該末梢血単核球からWT1特異的細胞傷害性T細胞(CTLs)、および/またはWT1特異的ヘルパーT細胞を誘導する工程を含む、良性腫瘍の予防または治療のために使用するWT1特異的CTLsおよび/またはWT1特異的ヘルパーT細胞の誘導方法。
- 良性腫瘍の処置を必要とする被験体由来の未熟樹状細胞を、請求項1~9のいずれか一項に記載のWT1ペプチドまたはその類縁体の存在下で培養し、あるいは、請求項10または11に記載のWT1ペプチドまたはその類縁体をコードする核酸分子を該未熟樹状細胞に導入し、WT1提示樹状細胞を誘導する工程を含む、良性腫瘍の予防または治療のために使用するWT1提示樹状細胞の誘導方法。
- WT1ペプチドもしくはその類縁体、またはそれをコードする核酸分子を含む、良性腫瘍の予防または治療のために使用する、WT1特異的CTLs、および/またはWT1特異的ヘルパーT細胞を誘導するための組成物。
- WT1ペプチドもしくはその類縁体、またはそれをコードする核酸分子を含む、良性腫瘍の予防または治療のために使用する、WT1提示樹状細胞を誘導するための組成物。
- WT1特異的CTLs、および/またはWT1特異的ヘルパーT細胞を含む、良性腫瘍の予防または治療のための組成物。
- WT1提示樹状細胞を含む、良性腫瘍の予防または治療のための組成物。
- 被験体における良性腫瘍を予防または治療する方法であって、該被験体に、有効量のWT1ペプチドまたはその類縁体を投与する工程を含む、方法。
- 良性腫瘍を予防または治療するための、WT1ペプチドまたはその類縁体。
- 良性腫瘍の予防または治療薬の製造における、WT1ペプチドまたはその類縁体の使用。
Priority Applications (9)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2020551127A JP7393752B2 (ja) | 2018-10-05 | 2019-10-04 | 良性腫瘍の予防または治療薬 |
| EP19868580.2A EP3865146A4 (en) | 2018-10-05 | 2019-10-04 | Prophylactic or therapeutic drug for benign tumor |
| MX2021003938A MX2021003938A (es) | 2018-10-05 | 2019-10-04 | Composicion para prevenir o tratar un tumor benigno. |
| CA3115240A CA3115240A1 (en) | 2018-10-05 | 2019-10-04 | Composition for preventing or treating benign tumor |
| CN201980080769.XA CN113395976A (zh) | 2018-10-05 | 2019-10-04 | 良性肿瘤的预防或治疗药 |
| KR1020217012141A KR20210073540A (ko) | 2018-10-05 | 2019-10-04 | 양성 종양의 예방 또는 치료약 |
| AU2019352354A AU2019352354A1 (en) | 2018-10-05 | 2019-10-04 | Prophylactic or therapeutic drug for benign tumor |
| US17/282,172 US20220008528A1 (en) | 2018-10-05 | 2019-10-04 | Composition for preventing or treating benign tumor |
| NZ774631A NZ774631A (en) | 2018-10-05 | 2019-10-04 | Composition for preventing or treating benign tumor |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2018190461 | 2018-10-05 | ||
| JP2018-190461 | 2018-10-05 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2020071551A1 true WO2020071551A1 (ja) | 2020-04-09 |
Family
ID=70054693
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2019/039383 WO2020071551A1 (ja) | 2018-10-05 | 2019-10-04 | 良性腫瘍の予防または治療薬 |
Country Status (11)
| Country | Link |
|---|---|
| US (1) | US20220008528A1 (ja) |
| EP (1) | EP3865146A4 (ja) |
| JP (1) | JP7393752B2 (ja) |
| KR (1) | KR20210073540A (ja) |
| CN (1) | CN113395976A (ja) |
| AU (1) | AU2019352354A1 (ja) |
| CA (1) | CA3115240A1 (ja) |
| MX (1) | MX2021003938A (ja) |
| NZ (1) | NZ774631A (ja) |
| TW (1) | TW202027777A (ja) |
| WO (1) | WO2020071551A1 (ja) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2021230638A1 (en) * | 2020-05-12 | 2021-11-18 | Lg Chem, Ltd. | Multimeric t-cell modulatory polypeptides and methods of use thereof |
Citations (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2002079253A1 (fr) | 2001-03-22 | 2002-10-10 | Haruo Sugiyama | Peptide modifie wt1 |
| WO2003106682A1 (ja) | 2002-06-12 | 2003-12-24 | 中外製薬株式会社 | Hla−a24拘束性癌抗原ペプチド |
| WO2004026897A1 (ja) | 2002-09-20 | 2004-04-01 | Chugai Seiyaku Kabushiki Kaisha | Wt1置換型ペプチド |
| WO2007063903A1 (ja) | 2005-11-30 | 2007-06-07 | International Institute Of Cancer Immunology, Inc. | 新規ペプチド化合物 |
| WO2009072610A1 (ja) | 2007-12-05 | 2009-06-11 | International Institute Of Cancer Immunology, Inc. | 癌ワクチン組成物 |
| WO2014157692A1 (ja) | 2013-03-29 | 2014-10-02 | 大日本住友製薬株式会社 | Wt1抗原ペプチドコンジュゲートワクチン |
| WO2016093326A1 (ja) | 2014-12-11 | 2016-06-16 | 株式会社癌免疫研究所 | 血管新生病の免疫療法 |
| JP2017523784A (ja) * | 2014-08-04 | 2017-08-24 | フレッド ハッチンソン キャンサー リサーチ センター | Wt−1に特異的なt細胞免疫治療 |
| WO2018181648A1 (ja) * | 2017-03-30 | 2018-10-04 | 大日本住友製薬株式会社 | Wt1癌抗原ペプチドおよびこれを含むペプチドコンジュゲート体 |
| JP2018190461A (ja) | 2018-08-07 | 2018-11-29 | カシオ計算機株式会社 | 注文処理装置及びプログラム |
Family Cites Families (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CA2337743C (en) * | 1998-07-31 | 2015-07-07 | Yoshihiro Oka | Tumor antigen based on products of the tumor suppressor gene wt1 |
| AU5161800A (en) * | 1999-05-24 | 2000-12-12 | Board Of Regents, The University Of Texas System | Methods and compositions for non-viral gene therapy for treatment of hyperproliferative diseases |
| AU2003262094A1 (en) * | 2002-09-12 | 2004-04-30 | Haruo Sugiyama | Cancer antigen peptide preparation |
| WO2012037551A2 (en) * | 2010-09-17 | 2012-03-22 | Irx Therapeutics, Inc. | Primary cell-derived biologic and wt1 synthetic long peptide vaccine |
| JP6117515B2 (ja) * | 2011-11-01 | 2017-04-19 | 国立大学法人名古屋大学 | 髄膜腫治療用医薬組成物 |
| WO2016186177A1 (ja) * | 2015-05-20 | 2016-11-24 | 大日本住友製薬株式会社 | Wt1抗原ペプチドおよび免疫調節剤の併用 |
| AU2016356708B2 (en) * | 2015-11-20 | 2022-02-03 | Memorial Sloan Kettering Cancer Center | Methods and compositions for treating cancer |
-
2019
- 2019-10-04 AU AU2019352354A patent/AU2019352354A1/en active Pending
- 2019-10-04 US US17/282,172 patent/US20220008528A1/en active Pending
- 2019-10-04 CN CN201980080769.XA patent/CN113395976A/zh active Pending
- 2019-10-04 MX MX2021003938A patent/MX2021003938A/es unknown
- 2019-10-04 KR KR1020217012141A patent/KR20210073540A/ko active Search and Examination
- 2019-10-04 NZ NZ774631A patent/NZ774631A/en unknown
- 2019-10-04 JP JP2020551127A patent/JP7393752B2/ja active Active
- 2019-10-04 EP EP19868580.2A patent/EP3865146A4/en active Pending
- 2019-10-04 TW TW108136125A patent/TW202027777A/zh unknown
- 2019-10-04 WO PCT/JP2019/039383 patent/WO2020071551A1/ja unknown
- 2019-10-04 CA CA3115240A patent/CA3115240A1/en active Pending
Patent Citations (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2002079253A1 (fr) | 2001-03-22 | 2002-10-10 | Haruo Sugiyama | Peptide modifie wt1 |
| WO2003106682A1 (ja) | 2002-06-12 | 2003-12-24 | 中外製薬株式会社 | Hla−a24拘束性癌抗原ペプチド |
| WO2004026897A1 (ja) | 2002-09-20 | 2004-04-01 | Chugai Seiyaku Kabushiki Kaisha | Wt1置換型ペプチド |
| WO2007063903A1 (ja) | 2005-11-30 | 2007-06-07 | International Institute Of Cancer Immunology, Inc. | 新規ペプチド化合物 |
| WO2009072610A1 (ja) | 2007-12-05 | 2009-06-11 | International Institute Of Cancer Immunology, Inc. | 癌ワクチン組成物 |
| WO2014157692A1 (ja) | 2013-03-29 | 2014-10-02 | 大日本住友製薬株式会社 | Wt1抗原ペプチドコンジュゲートワクチン |
| JP2017523784A (ja) * | 2014-08-04 | 2017-08-24 | フレッド ハッチンソン キャンサー リサーチ センター | Wt−1に特異的なt細胞免疫治療 |
| WO2016093326A1 (ja) | 2014-12-11 | 2016-06-16 | 株式会社癌免疫研究所 | 血管新生病の免疫療法 |
| WO2018181648A1 (ja) * | 2017-03-30 | 2018-10-04 | 大日本住友製薬株式会社 | Wt1癌抗原ペプチドおよびこれを含むペプチドコンジュゲート体 |
| JP2018190461A (ja) | 2018-08-07 | 2018-11-29 | カシオ計算機株式会社 | 注文処理装置及びプログラム |
Non-Patent Citations (25)
| Title |
|---|
| ALTSCHUL ET AL., J. MOL. BIOL., vol. 215, 1990, pages 403 - 410 |
| AMINI NIK S. ET AL.: "Upregulation of Wlms' tumor gene 1 (WT1) in desmoid tumors", INTERNATIONAL JOURNAL OF CANCER, vol. 114, no. 2, 2005, pages 202 - 208, XP055700055 * |
| ASLAN, A. ET AL.: "Investigation of Insulin-Like Growth Factor-1 (IGF-1), P53, and Wilms' Tumor 1 (WT1) Expression Levels in the Colon Polyp Subtypes in Colon Cancer", MEDICAL SCIENCE MONITOR, vol. 25, 25 July 2019 (2019-07-25), pages 5510 - 5517, XP055700063 * |
| AUSUBEL ET AL.: "Current Protocols in Molecular Biology", 1987, GREENE PUB. ASSOCIATES AND WILEY-INTERSCIENCE, article "Current Protocols in Molecular Biology, Supplement 1-38, DNA Cloning 1: Core Techniques, A Practical Approach" |
| BATZER, NUCLEIC ACID RES., vol. 19, 1991, pages 5081 |
| BAUTZ, D. J. ET AL.: "Prophylactic vaccination targeting ERBB3 decreases polyp burden in a mouse model of human colorectal cancer", ONCOIMMUNOLOGY, vol. 6, no. 1, 2016, pages 1 - 6, XP055700061 * |
| GESSLER, M. ET AL., NATURE, vol. 343, 1990, pages 774 - 778 |
| INNIS, M.A.: "PCR Protocols: A Guide to Methods and Applications", 1990, ACADEMIC PRESS |
| KOIDO, S. ET AL.: "Treatment with chemotherapy and dendritic cells pulsed with multiple Wilms' tumor 1 (WT1)-specific MHC class I/II-restricted epitopes for pancreatic cancer", CLINICAL CANCER RESEARCH, vol. 20, no. 16, 2014, pages 4228 - 4239, XP002780582, DOI: 10.1158/1078-0432.CCR-14-0314 * |
| LANGMAN, G. ET AL.: "WT1 expression in salivary gland pleomorphic adenomas: a reliable marker of the neoplastic myoepithelium", MODERN PATHOLOGY, vol. 24, no. 2, 2011, pages 168 - 174, XP055700050 * |
| MARK ET AL., PROC NATL ACAD SCI USA., vol. 81, no. 18, pages 5662 - 5666 |
| NAPEKOSKI, K. M. ET AL.: "Microvenular hemangioma: a clinicopathologic review of 13 cases", JOURNAL OF CUTANEOUS PATHOLOGY, vol. 41, no. 11, 2014, pages 816 - 822, XP055700043 * |
| NEEDLEMANWUNSCH, J. MOL. BIOL., vol. 48, 1970, pages 443 - 453 |
| NOMURA, MASAYA ET AL.: "A Case of Heterochronous Ascending Colon Carcinoid 7 Years after Curative Resection of Descending Colon Carcinoid", THE JAPANESE JOURNAL OF GASTROENTEROLOGICAL SURGERY, vol. 37, no. 2, 2004, pages 217 - 222, XP055700057, Retrieved from the Internet <URL:https://doi.org/10.5833/jjgs.37.217> * |
| OHTSUKA ET AL., J. BIOL. CHEM., vol. 260, 1985, pages 2605 - 2608 |
| OUCHI, TOMOYUKI; KAKU, TOHRU: "Carcinoma ex pleomorphic adenoma mm metastasizing pleomorphic adenoma", THE DENTAL JOURNAL OF HEALTH SCIENCES UNIVERSITY OF HOKKAIDO, vol. 26, no. 2, 30 November 2006 (2006-11-30), JP, pages 85 - 86, XP009527511, ISSN: 0910-9722 * |
| PEARSONLIPMAN, PROC. NATL. ACAD. SCI., USA, vol. 85, 1988, pages 2444 - 2448 |
| ROSSOLINI ET AL., MOL. CELL. PROBES, vol. 8, 1994, pages 91 - 98 |
| See also references of EP3865146A4 |
| SHIRAKATA, T. ET AL.: "WT1 peptide therapy for a patient with chemotherapy-resistant salivary gland cancer", ANTICANCER RESEARCH, vol. 32, no. 3, 2012, pages 1081 - 1085, XP055700049, ISSN: 0250-7005 * |
| SMITHWATERMAN, J. MOL. BIOL., vol. 147, 1981, pages 195 - 197 |
| SNINSKY, J.J ET AL.: "Short Protocols in Molecular Biology: A Compendium of Methods from Current Protocols in Molecular Biology", vol. 1, 1999, GREENE PUB. ASSOCIATES AND WILEY-INTERSCIENCE, pages: 7.42 - 7.45 |
| WANG ET AL., SCIENCE, vol. 224, no. 4656, 29 June 1984 (1984-06-29), pages 1431 - 1433 |
| WEBER, G. ET AL.: "WT1 peptide-specific T cells generated from peripheral blood of healthy donors: possible implications for adoptive immunotherapy after allogeneic stem cell transplantation", LEUKEMIA, vol. 23, no. 9, 2009, pages 1634 - 1642, XP055700053 * |
| ZOLLER ET AL., NUCLEIC ACIDS RES., vol. 10, no. 20, 25 October 1982 (1982-10-25), pages 6487 - 6500 |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2021230638A1 (en) * | 2020-05-12 | 2021-11-18 | Lg Chem, Ltd. | Multimeric t-cell modulatory polypeptides and methods of use thereof |
Also Published As
| Publication number | Publication date |
|---|---|
| EP3865146A4 (en) | 2022-06-29 |
| KR20210073540A (ko) | 2021-06-18 |
| NZ774631A (en) | 2024-03-22 |
| AU2019352354A1 (en) | 2021-05-13 |
| JP7393752B2 (ja) | 2023-12-07 |
| CN113395976A (zh) | 2021-09-14 |
| US20220008528A1 (en) | 2022-01-13 |
| EP3865146A1 (en) | 2021-08-18 |
| TW202027777A (zh) | 2020-08-01 |
| JPWO2020071551A1 (ja) | 2021-09-24 |
| MX2021003938A (es) | 2021-08-11 |
| CA3115240A1 (en) | 2020-04-09 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US11707512B2 (en) | Cancer vaccine composition | |
| CN105377291B (zh) | 免疫原性wt-1肽和其使用方法 | |
| EP2010209B1 (en) | Immunogenic wt-1 peptides and methods of use thereof | |
| PT1731605E (pt) | Péptidos antigénicos de cancro derivados de wt1 | |
| AU2006304573A1 (en) | WT1 HLA class II-binding peptides and compositions and methods comprising same | |
| JP6112171B2 (ja) | 免疫誘導剤 | |
| US20170072038A1 (en) | Combination therapy with wt1 peptide vaccine and temozolomide | |
| KR20200067862A (ko) | 종양-관련된 항원 에피토프의 미생물 서열 변이체 | |
| JPWO2006062094A1 (ja) | 新規癌抗原ペプチド及びその用途 | |
| JP2003516344A (ja) | Hlaクラスia2腫瘍関連抗原ペプチドおよびワクチン組成物 | |
| JP2012102105A (ja) | 血管内皮増殖因子受容体1に由来するエピトープ・ペプチドおよびこれらのペプチドを含むワクチン | |
| WO2020071551A1 (ja) | 良性腫瘍の予防または治療薬 | |
| WO2018032501A1 (zh) | 一种新的肿瘤特异性多肽及其应用 | |
| WO2015005479A1 (ja) | 腫瘍抗原ペプチド | |
| US20140147490A1 (en) | Hla class i a2 tumor associated antigen peptides and vaccine compositions |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 19868580 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 3115240 Country of ref document: CA Ref document number: 2020551127 Country of ref document: JP Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 20217012141 Country of ref document: KR Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 2019352354 Country of ref document: AU Date of ref document: 20191004 Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 2019868580 Country of ref document: EP Effective date: 20210506 |