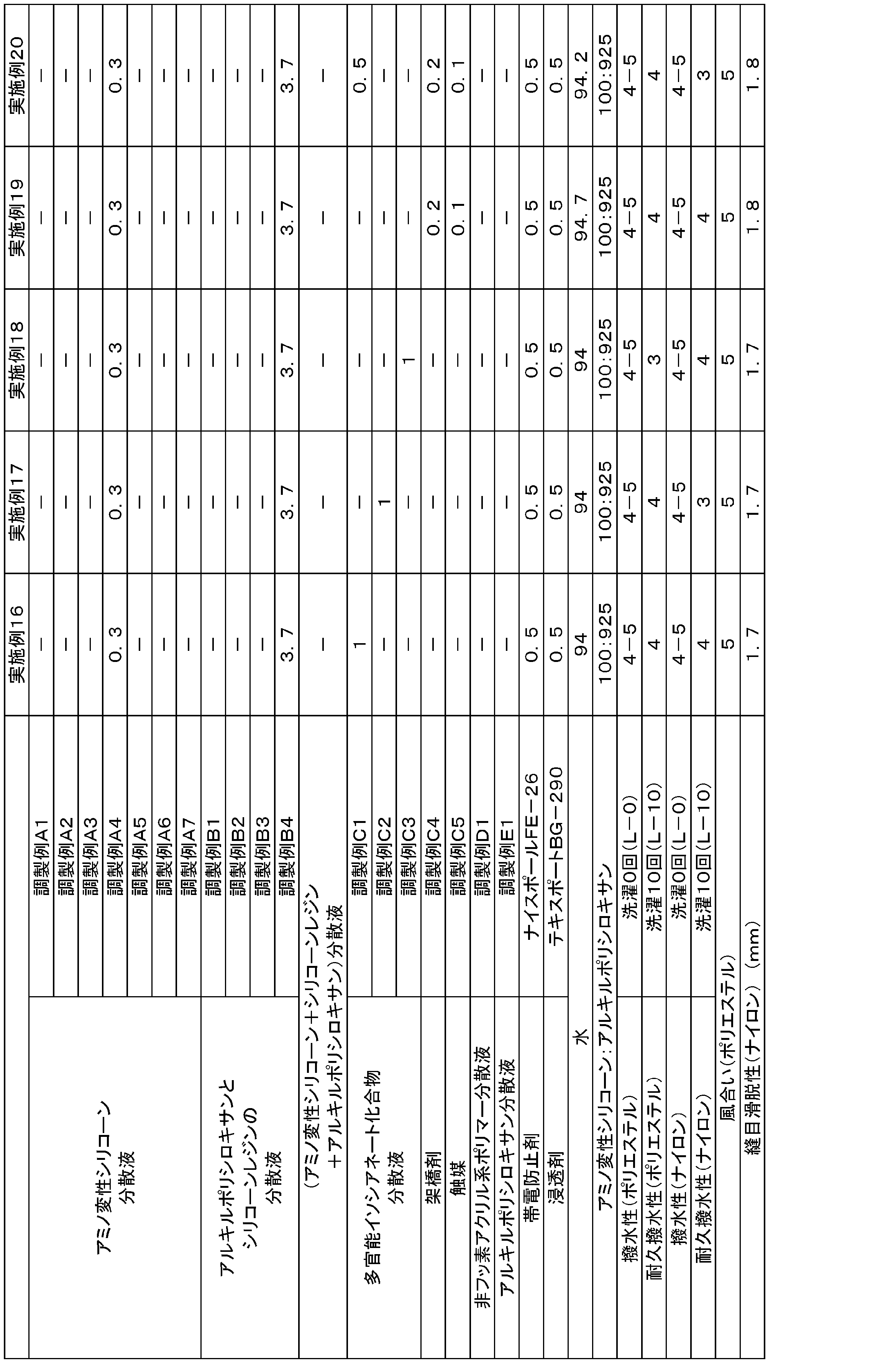
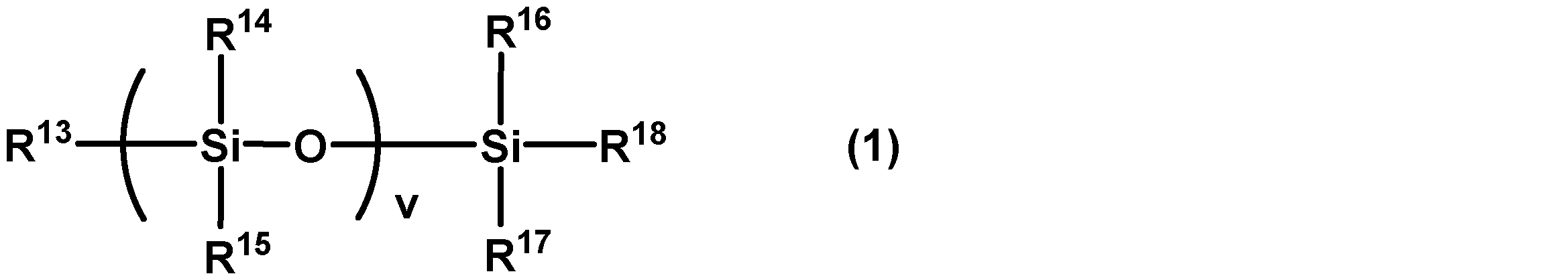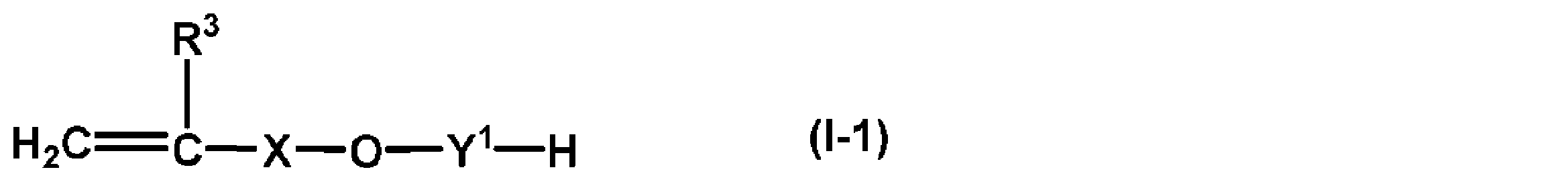WO2019131456A1 - 撥水剤組成物、及び撥水性繊維製品の製造方法 - Google Patents
撥水剤組成物、及び撥水性繊維製品の製造方法 Download PDFInfo
- Publication number
- WO2019131456A1 WO2019131456A1 PCT/JP2018/047085 JP2018047085W WO2019131456A1 WO 2019131456 A1 WO2019131456 A1 WO 2019131456A1 JP 2018047085 W JP2018047085 W JP 2018047085W WO 2019131456 A1 WO2019131456 A1 WO 2019131456A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- group
- mass
- water repellent
- amino
- dispersion
- Prior art date
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K3/00—Materials not provided for elsewhere
- C09K3/18—Materials not provided for elsewhere for application to surfaces to minimize adherence of ice, mist or water thereto; Thawing or antifreeze materials for application to surfaces
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D183/00—Coating compositions based on macromolecular compounds obtained by reactions forming in the main chain of the macromolecule a linkage containing silicon, with or without sulfur, nitrogen, oxygen, or carbon only; Coating compositions based on derivatives of such polymers
- C09D183/04—Polysiloxanes
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D5/00—Coating compositions, e.g. paints, varnishes or lacquers, characterised by their physical nature or the effects produced; Filling pastes
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06M—TREATMENT, NOT PROVIDED FOR ELSEWHERE IN CLASS D06, OF FIBRES, THREADS, YARNS, FABRICS, FEATHERS OR FIBROUS GOODS MADE FROM SUCH MATERIALS
- D06M13/00—Treating fibres, threads, yarns, fabrics or fibrous goods made from such materials, with non-macromolecular organic compounds; Such treatment combined with mechanical treatment
- D06M13/50—Treating fibres, threads, yarns, fabrics or fibrous goods made from such materials, with non-macromolecular organic compounds; Such treatment combined with mechanical treatment with organometallic compounds; with organic compounds containing boron, silicon, selenium or tellurium atoms
- D06M13/51—Compounds with at least one carbon-metal or carbon-boron, carbon-silicon, carbon-selenium, or carbon-tellurium bond
- D06M13/513—Compounds with at least one carbon-metal or carbon-boron, carbon-silicon, carbon-selenium, or carbon-tellurium bond with at least one carbon-silicon bond
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06M—TREATMENT, NOT PROVIDED FOR ELSEWHERE IN CLASS D06, OF FIBRES, THREADS, YARNS, FABRICS, FEATHERS OR FIBROUS GOODS MADE FROM SUCH MATERIALS
- D06M15/00—Treating fibres, threads, yarns, fabrics, or fibrous goods made from such materials, with macromolecular compounds; Such treatment combined with mechanical treatment
- D06M15/19—Treating fibres, threads, yarns, fabrics, or fibrous goods made from such materials, with macromolecular compounds; Such treatment combined with mechanical treatment with synthetic macromolecular compounds
- D06M15/37—Macromolecular compounds obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds
- D06M15/643—Macromolecular compounds obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds containing silicon in the main chain
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G77/00—Macromolecular compounds obtained by reactions forming a linkage containing silicon with or without sulfur, nitrogen, oxygen or carbon in the main chain of the macromolecule
- C08G77/04—Polysiloxanes
- C08G77/14—Polysiloxanes containing silicon bound to oxygen-containing groups
- C08G77/16—Polysiloxanes containing silicon bound to oxygen-containing groups to hydroxyl groups
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G77/00—Macromolecular compounds obtained by reactions forming a linkage containing silicon with or without sulfur, nitrogen, oxygen or carbon in the main chain of the macromolecule
- C08G77/04—Polysiloxanes
- C08G77/14—Polysiloxanes containing silicon bound to oxygen-containing groups
- C08G77/18—Polysiloxanes containing silicon bound to oxygen-containing groups to alkoxy or aryloxy groups
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G77/00—Macromolecular compounds obtained by reactions forming a linkage containing silicon with or without sulfur, nitrogen, oxygen or carbon in the main chain of the macromolecule
- C08G77/04—Polysiloxanes
- C08G77/22—Polysiloxanes containing silicon bound to organic groups containing atoms other than carbon, hydrogen and oxygen
- C08G77/26—Polysiloxanes containing silicon bound to organic groups containing atoms other than carbon, hydrogen and oxygen nitrogen-containing groups
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06M—TREATMENT, NOT PROVIDED FOR ELSEWHERE IN CLASS D06, OF FIBRES, THREADS, YARNS, FABRICS, FEATHERS OR FIBROUS GOODS MADE FROM SUCH MATERIALS
- D06M15/00—Treating fibres, threads, yarns, fabrics, or fibrous goods made from such materials, with macromolecular compounds; Such treatment combined with mechanical treatment
- D06M15/19—Treating fibres, threads, yarns, fabrics, or fibrous goods made from such materials, with macromolecular compounds; Such treatment combined with mechanical treatment with synthetic macromolecular compounds
- D06M15/37—Macromolecular compounds obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds
- D06M15/643—Macromolecular compounds obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds containing silicon in the main chain
- D06M15/6436—Macromolecular compounds obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds containing silicon in the main chain containing amino groups
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06M—TREATMENT, NOT PROVIDED FOR ELSEWHERE IN CLASS D06, OF FIBRES, THREADS, YARNS, FABRICS, FEATHERS OR FIBROUS GOODS MADE FROM SUCH MATERIALS
- D06M2101/00—Chemical constitution of the fibres, threads, yarns, fabrics or fibrous goods made from such materials, to be treated
- D06M2101/16—Synthetic fibres, other than mineral fibres
- D06M2101/30—Synthetic polymers consisting of macromolecular compounds obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds
- D06M2101/32—Polyesters
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06M—TREATMENT, NOT PROVIDED FOR ELSEWHERE IN CLASS D06, OF FIBRES, THREADS, YARNS, FABRICS, FEATHERS OR FIBROUS GOODS MADE FROM SUCH MATERIALS
- D06M2101/00—Chemical constitution of the fibres, threads, yarns, fabrics or fibrous goods made from such materials, to be treated
- D06M2101/16—Synthetic fibres, other than mineral fibres
- D06M2101/30—Synthetic polymers consisting of macromolecular compounds obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds
- D06M2101/34—Polyamides
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06M—TREATMENT, NOT PROVIDED FOR ELSEWHERE IN CLASS D06, OF FIBRES, THREADS, YARNS, FABRICS, FEATHERS OR FIBROUS GOODS MADE FROM SUCH MATERIALS
- D06M2200/00—Functionality of the treatment composition and/or properties imparted to the textile material
- D06M2200/10—Repellency against liquids
- D06M2200/12—Hydrophobic properties
Definitions
- the present invention relates to a water repellent composition and a method for producing a water repellent fiber product.
- a fluorine-based water repellent having a fluorine group is known, and a textile product having water repellency imparted to the surface thereof is known by treating such a fluorine-based water repellent onto a fiber product or the like.
- a fluorine-based water repellent is generally produced by polymerizing or copolymerizing a monomer having a fluoroalkyl group (monomer).
- fiber products treated with a fluorine-based water repellent exhibit excellent water repellency, since the fluoroalkyl group-containing monomer is hard to be decomposed, there are environmental problems.
- Patent Document 1 proposes a water repellent composed of a specific non-fluorinated polymer containing, as a monomer unit, a (meth) acrylic ester having 12 or more carbon atoms in the ester portion.
- Patent Document 2 proposes a soft water repellent agent containing an amino-modified silicone and a polyfunctional isocyanate compound.
- Patent Document 2 Although the silicone-based soft water repellent agent described in Patent Document 2 can obtain good texture, it is difficult to obtain sufficient water repellent performance, and along with the initial water repellency, it is particularly water repellant even after washing. There is a problem that it is difficult to obtain durable water repellency that can be sufficiently maintained.
- the present invention has been made in view of the above circumstances, and is a water repellent composition capable of obtaining a water repellent fiber product excellent in water repellency, durable water repellency and texture, and a water repellent fiber product using the same.
- the purpose is to provide a manufacturing method of
- the present inventors have intensively studied to solve the above problems, and as a result, a specific silicone compound, a silicone resin and an alkylpolysiloxane are combined, and the compounding amount of the specific silicone compound and the compounding amount of the alkylpolysiloxane It has been found that by adjusting the mass ratio, a fiber product having a soft texture can be obtained while exhibiting a high level of water repellency and durable water repellency, and based on this finding, the present invention has been completed.
- the present invention contains an amino-modified silicone, a silicone resin, and an alkylpolysiloxane, and the blending amount of the alkylpolysiloxane is 500 to 15,000 parts by mass with respect to 100 parts by mass of the amino-modified silicone.
- a first water repellent composition is provided.
- a water repellent fiber product excellent in water repellency, durable water repellency and texture can be obtained.
- the reason why such an effect can be obtained is that by combining the above components in a predetermined blending amount, it is possible to improve the water repellency and the durable water repellency without impairing the texture of the fiber, and the fiber is too stretched or hard It is thought that it can be suppressed that it becomes too much.
- the functional group equivalent of the amino-modified silicone may be 100 to 20,000 g / mol from the viewpoint of water repellency, durability water repellency, feel and seam slipperiness.
- the functional group equivalent of amino-modified silicone means the molecular weight of amino-modified silicone per 1 nitrogen atom.
- the present invention also includes a dimethylpolysiloxane having a hydroxyl group and / or an alkoxy group having 1 to 3 carbon atoms, an amino group-containing silane coupling agent, a silicone resin, and an alkylpolysiloxane, and contains an alkylpolysiloxane.
- a second water repellent composition is provided, wherein the amount is 500 to 15,000 parts by mass with respect to 100 parts by mass of the total content of the dimethylpolysiloxane and the amino group-containing silane coupling agent.
- the second water repellent composition of the present invention a water repellent fiber product excellent in water repellency, durable water repellency and texture can be realized.
- the second water repellent composition according to the present invention can also provide a water repellent fiber product which is also excellent in stitch slippage.
- the present invention also provides a method of producing a water repellent fiber product, comprising the step of treating a fiber with a treatment liquid containing the first or second water repellent composition according to the present invention.
- a water-repellent fiber product excellent in water repellency, durable water repellency and texture can be stably produced.
- the water repellent fiber product produced by the method of the present invention can be excellent in stitch slippage.
- a water repellent composition capable of obtaining a water repellent fiber product excellent in water repellency, durable water repellency and texture. Further, according to the water repellent composition of the present invention, excellent seam slipperiness can be imparted to textiles and the like.
- the water repellent composition of the present embodiment comprises a water repellent component comprising an amino-modified silicone (hereinafter sometimes referred to as component (I)) and a silicone resin (hereinafter sometimes referred to as component (II)); It contains siloxane (hereinafter sometimes referred to as component (III)).
- component (I) an amino-modified silicone
- component (II) a silicone resin
- component (III) siloxane
- the amino-modified silicone includes a compound having an organic group containing an amino group and / or an imino group at the side chain or end of the organopolysiloxane.
- an organic group for example, an organic group represented by -R-NH 2 and an organic group represented by -R-NH-R'-NH 2 can be mentioned.
- R and R ' bivalent groups, such as ethylene group and a propylene group, are mentioned.
- Part or all of the amino group and / or imino group may be a blocked amino group and / or imino group.
- the blocked amino group and / or imino group can be obtained, for example, by treating the amino group and / or imino group with a blocking agent.
- blocking agents include fatty acids having 2 to 22 carbon atoms, acid anhydrides of fatty acids having 2 to 22 carbon atoms, acid halides of fatty acids having 2 to 22 carbon atoms, aliphatic monoisocyanates having 1 to 22 carbon atoms, etc. It can be mentioned.
- the functional group equivalent of the amino-modified silicone is preferably 100 to 20,000 g / mol, more preferably 150 to 12,000 g / mol, from the viewpoints of water repellency, durability water repellency, texture and slip resistance. 4,000 g / mol is more preferred.
- the amino-modified silicone is preferably liquid at 25 ° C.
- Kinematic viscosity at 25 ° C. of the amino-modified silicone is preferably 10 ⁇ 100,000mm 2 / s, more preferably 10 ⁇ 30,000mm 2 / s, is 10 ⁇ 5,000mm 2 / s Is more preferred.
- the kinematic viscosity at 25 ° C. means a value measured by the method described in JIS K 2283: 2000 (Ubbelohde viscometer).
- a commercial item can be used as amino modified silicone.
- Examples of commercially available products include KF8005, KF-868, KF-864, and KF-393 (all of which are trade names of Shin-Etsu Chemical Co., Ltd., trade names), XF42-B1989 (Momentive Performance Materials Japan GK Co., Ltd.) Products, SF-8417, BY16-853U (all of which are manufactured by Toray Dow Corning Co., Ltd., trade names), and the like.
- the amino-modified silicone may be used alone or in combination of two or more.
- part or all of the amino group and / or imino group may be neutralized or unneutralized.
- organic acids such as lactic acid, acetic acid, propionic acid, maleic acid, oxalic acid, formic acid, methanesulfonic acid and toluenesulfonic acid; and inorganic acids such as hydrogen chloride, sulfuric acid and nitric acid can be used.
- the compounding amount of the amino-modified silicone in the water repellent composition of the present embodiment is 0 based on the total amount of the water repellent composition. .01 to 1% by mass.
- the compounding amount of the amino-modified silicone can be 0.1 to 50% by mass, and may be 0.2 to 20% by mass, based on the total amount of the water repellent composition. .
- a dimethylpolysiloxane (hereinafter referred to as (IV) component) having a hydroxyl group and / or an alkoxy group having 1 to 3 carbon atoms at its terminal or side chain in place of the amino-modified silicone.
- an amino group-containing silane coupling agent (hereinafter sometimes referred to as component (V)) can be blended.
- the water repellent composition (hereinafter sometimes referred to as a second water repellent composition) contains at least components (IV), (V), (II) and (III).
- hydroxyl group and / or alkoxy group having 1 to 3 carbon atoms in the component (IV) is preferably bonded to the end of the dimethylpolysiloxane skeleton, and more preferably bonded to both ends.
- Kinematic viscosity at 25 ° C. of the reaction product of (IV) component and (V) component is preferably 10 ⁇ 100,000mm 2 / s, more preferably from 10 ⁇ 30,000mm 2 / s, More preferably, it is 10 to 5,000 mm 2 / s.
- the kinematic viscosity at 25 ° C. means a value measured by the method described in JIS K 2283: 2000 (Ubbelohde viscometer).
- amino group-containing silane coupling agent for example, KBM-602 (N-2- (aminoethyl) -3-aminopropylmethyldimethoxysilane) manufactured by Shin-Etsu Chemical Co., Ltd., KBM-603 (N-2- (amino) And silane compounds having an amino group and an alkoxy group having 1 to 3 carbon atoms, such as ethyl) -3-aminopropyltrimethoxysilane) and KBM-903 (3-aminopropyltrimethoxysilane).
- the compounding amounts of the (IV) component and the (V) component in the second water repellent composition is the total amount of the water repellent composition.
- the total amount can be 0.01 to 1% by mass on the basis of
- the compounding amounts of the (IV) component and the (V) component can be 0.1 to 50 mass% in total, based on the total amount of the water repellent composition, and 0.2 to 50 mass%. 20 mass% may be sufficient.
- the compounding ratio of the (IV) component and the (V) component is the functional group of the (IV) component (the total of the hydroxyl group and the alkoxy group having 1 to 3 carbon atoms) and the functional group of the (V) component (1 to 3 carbon atoms)
- the molar ratio [(IV) :( V)] to the sum of alkoxy groups) can be 20: 1 to 1: 1.
- the water repellent composition of the present embodiment contains a silicone resin as a silicone compound other than the amino-modified silicone, the component (IV), and the reaction product of the component (IV) and the component (V).
- a silicone resin it is preferable that it is an organopolysiloxane which contains MQ, MDQ, MT, MTQ, MDT or MDTQ as a structural component, is solid at 25 ° C., and has a three-dimensional structure.
- silicone resins are disclosed in JIS K 6249: 2003 13.
- the hardness measured by a type A durometer according to the hardness test is preferably 20 or more, more preferably 60 or more.
- M, D, T and Q respectively represent (R ′ ′) 3 SiO 0.5 units, (R ′ ′) 2 SiO units, R ′ ′ SiO 1.5 units and SiO 2 units.
- R ′ ′ represents a C 1-10 monovalent aliphatic hydrocarbon group or a C 6-15 monovalent aromatic hydrocarbon group.
- the silicone resin is generally known as MQ resin, MT resin or MDT resin, and may have a portion designated as MDQ, MTQ or MDTQ.
- the silicone resin can also be obtained as a solution in which it is dissolved in a suitable solvent other than alkylpolysiloxane or alkylpolysiloxane.
- suitable solvents other than alkylpolysiloxanes include n-hexane, isopropyl alcohol, methylene chloride, 1,1,1-trichloroethane, and mixtures of these solvents.
- KF7312F trimethylsilyl
- Octamethyl cyclotetrasiloxane 50: 50 mixture
- silicone resin for example, MQ-1600 solid Resin (trimethylsilyl group-containing polysiloxane), MQ-1640 Flake Resin (trimethylsilyl group-containing polysiloxane, polypropylsilsesquioxane) commercially available from Toray Dow Corning Co., Ltd. Etc.
- MQ-1600 solid Resin trimethylsilyl group-containing polysiloxane
- MQ-1640 Flake Resin trimethylsilyl group-containing polysiloxane, polypropylsilsesquioxane
- the compounding amount of the silicone resin in the water repellent composition of the present embodiment is 50 to 15,000 parts by mass with respect to 100 parts by mass of the amino-modified silicone from the viewpoint of water repellency, feeling and seam slippage. And may be 100 to 10,000 parts by mass, may be 150 to 6,000 parts by mass, may be 500 to 15,000 parts by mass, and 900 to 6,000. It may be parts by mass.
- the compounding quantity of the silicone resin in the 2nd water repellent composition of this embodiment is the total compounding quantity 100 mass of (IV) component and (V) component from a viewpoint of water repellency, feel, and a stitch slipperiness.
- the amount is preferably 500 to 15,000 parts by mass, and more preferably 900 to 6,000 parts by mass.
- the alkylpolysiloxane is a compound in which the side chain and the end of the chain organopolysiloxane are saturated hydrocarbon groups, or a compound in which the side chains of cyclic organopolysiloxanes are saturated hydrocarbon groups.
- the compound represented by following General formula (1), the compound represented by following General formula (2), etc. are mentioned, for example.
- R 13 , R 14 , R 15 , R 16 , R 17 , and R 18 each independently represent a monovalent saturated hydrocarbon group having 1 to 18 carbon atoms, and v is 1 or more Represents an integer of ]
- R 19 and R 20 each independently represent a monovalent saturated hydrocarbon group having 1 to 18 carbon atoms, and w represents an integer of 2 to 20].
- R 13 , R 14 , R 15 , R 16 , R 17 and R 18 each independently have 1 to 18 carbon atoms. It is a monovalent saturated hydrocarbon group.
- the carbon number of the saturated hydrocarbon group is preferably 1 to 10 from the viewpoint of easily dissolving the silicone resin in the compound represented by the general formula (1) and easily obtaining the compound.
- the saturated hydrocarbon group may be linear or branched.
- the saturated hydrocarbon group is preferably linear, and more preferably linear alkyl.
- the saturated hydrocarbon group is preferably a methyl group or an ethyl group, and more preferably a methyl group.
- v is an integer of 1 or more. v can be suitably selected so that the kinematic viscosity of the compound represented by the general formula (1) falls within the range of the kinematic viscosity of the following alkylpolysiloxane.
- Examples of the compound represented by the above general formula (1) include dimethylpolysiloxane and diethylpolysiloxane.
- R 19 and R 20 are each independently a monovalent saturated hydrocarbon group having 1 to 18 carbon atoms.
- the carbon number of this saturated hydrocarbon group is preferably 1 to 10.
- the saturated hydrocarbon group may be linear or branched.
- the saturated hydrocarbon group is preferably linear, and more preferably linear alkyl.
- the saturated hydrocarbon group is preferably a methyl group or an ethyl group, and more preferably a methyl group.
- w is an integer of 2 to 20.
- w is preferably 3 to 10, and more preferably 4 or 5.
- the silicone resin tends to be easily dissolved in the compound represented by the general formula (2), and the compound tends to be easily obtained.
- the alkylpolysiloxanes may be used alone or in combination of two or more.
- the alkyl polysiloxane is preferably liquid at 25 ° C.
- Kinematic viscosity at 25 ° C. of alkyl polysiloxane preferably from 0.1 ⁇ 100,000mm 2 / s, more preferably 0.1 ⁇ 10,000mm 2 / s, 0.1 ⁇ 1, more preferably from 000 mm 2 / s, still more preferably 0.1 ⁇ 500mm 2 / s, particularly preferably from 0.1 ⁇ 100mm 2 / s.
- the kinematic viscosity at 25 ° C. means a value measured by the method described in JIS K 2283: 2000 (Ubbelohde viscometer).
- the compounding amount of the alkylpolysiloxane in the water repellent composition of the present embodiment is 500 to 15,000 parts by mass with respect to 100 parts by mass of the amino-modified silicone.
- the compounding amount of the alkylpolysiloxane is preferably 900 to 6,000 parts by mass with respect to 100 parts by mass of the amino-modified silicone from the viewpoint of water repellency, feeling and seam slippage.
- the compounding quantity of the alkylpolysiloxane in the 2nd water repellent composition of this embodiment is 500-15,000 mass parts with respect to 100 mass parts of total compounding quantities of (IV) component and (V) component. It is.
- the compounding amount of the alkylpolysiloxane is 900 to 6,000 parts by mass with respect to 100 parts by mass in total of the (IV) component and the (V) component from the viewpoint of water repellency, texture and seam slipperiness. Is more preferred.
- the mass ratio [(II) :( III)] of the compounding amount of the silicone resin to the compounding amount of the alkylpolysiloxane in the water repellent composition of the present embodiment is 10:90 to 60 from the viewpoint of texture and water repellency. : 40 is preferable, 20: 80 to 55: 45 is more preferable, and 20: 80 to 35: 65 is more preferable.
- water repellent component other than the component (I), the component (II), the component (IV) and the component (V) include, for example, known fluorine-based polymers, aliphatic hydrocarbons, aliphatic carboxylic acids and their esters, and polyolefins And hydrocarbon group-containing compounds such as poly (meth) acrylic acid esters.
- Examples of conventional fluorine-based polymers include NK Guard S-33 (manufactured by Nikka Chemical Co., Ltd.).
- aliphatic hydrocarbons examples include paraffinic hydrocarbons and olefinic hydrocarbons.
- the carbon number of the aliphatic hydrocarbon is preferably 12 or more.
- the aliphatic carboxylic acid may be either saturated or unsaturated, and preferably has 12 or more carbon atoms. Esterified compounds of such aliphatic carboxylic acids may be used.
- polystyrene resin examples include polyethylene, polypropylene, ethylene-propylene copolymer and the like.
- the poly (meth) acrylic acid ester preferably has 12 or more carbon atoms of a hydrocarbon group present via an ester bond. Moreover, it is preferable that carbon number of a hydrocarbon group is 24 or less.
- This hydrocarbon group may be linear or branched, may be saturated hydrocarbon or unsaturated hydrocarbon, and further has an alicyclic or aromatic ring. It may be done. Among these, linear ones are preferable, and linear alkyl groups are more preferable.
- the constituent ratio of the acrylic acid ester or methacrylic acid ester monomer in the polymer is preferably 80 to 100% by mass with respect to the total amount of the monomer units constituting the polymer. Moreover, it is preferable that the weight average molecular weight of this polymer is 500,000 or more. Also, a copolymer of acrylic acid ester and methacrylic acid ester may be used.
- poly (meth) acrylic acid ester (non-fluorinated polymer) for example, a (meth) acrylic acid ester monomer (A) represented by the following general formula (A-1)
- A-1 Non-fluorinated acrylic polymers containing a structural unit derived from component A) are also mentioned.
- R 1 represents hydrogen or a methyl group
- R 2 represents a monovalent hydrocarbon group having 12 or more carbon atoms that may have a substituent.
- the (meth) acrylic acid ester monomer (A) represented by the above general formula (A-1) used in this embodiment has 1 or more carbon atoms of 12 or more which may have a substituent.
- the hydrocarbon group may be linear or branched, may be a saturated hydrocarbon group or an unsaturated hydrocarbon group, and further may be an alicyclic or aromatic cyclic group. May be included. Among these, linear ones are preferable, and linear alkyl groups are more preferable. In this case, the water repellency is more excellent.
- R 2 is preferably a non-substituted hydrocarbon group.
- the carbon number of the hydrocarbon group is preferably 12 to 24.
- the carbon number is 12 or more
- the water repellent composition containing the non-fluorinated acrylic polymer is attached to a fiber product or the like, the water repellency is more easily improved.
- a water repellent composition containing a non-fluorinated acrylic polymer is attached to a fiber product or the like at a carbon number of 24 or less, the texture of the fiber product tends to be further improved.
- the carbon number of the hydrocarbon group is more preferably 12 to 21. When the carbon number is in this range, the water repellency and the texture become particularly excellent.
- Particularly preferable as the hydrocarbon group is a linear alkyl group having 12 to 18 carbon atoms.
- Examples of the component (A) include stearyl (meth) acrylate, cetyl (meth) acrylate, lauryl (meth) acrylate, dodecyl (meth) acrylate, myristyl (meth) acrylate, and (meth) acrylic acid. Pentadecyl, heptadecyl (meth) acrylate, nonadecyl (meth) acrylate, eicosyl (meth) acrylate, heheikosyl (meth) acrylate, behenyl (meth) acrylate, seryl (meth) acrylate and mesylate (meth) acrylate Can be mentioned.
- the component (A) can have at least one functional group selected from the group consisting of a hydroxy group, an amino group, a carboxy group, an epoxy group and an isocyanate group capable of reacting with the crosslinking agent.
- the durable water repellency of the resulting textile can be further improved.
- the isocyanate group may form a blocked isocyanate group protected by a blocking agent.
- the feel of the fiber product obtained can be further improved.
- the component (A) is preferably a monofunctional (meth) acrylic acid ester monomer having one polymerizable unsaturated group in one molecule.
- one type may be used alone, or two or more types may be used in combination.
- the component (A) is preferably used in combination with the acrylic acid ester monomer (a1) and the methacrylic acid ester monomer (a2) from the viewpoint of the durable water repellency of the fiber product to be obtained.
- the ratio (a1) / (a2) of the mass of the component (a1) to the mass of the component (a2) is preferably 30/70 to 90/10 and 40/60 to 85/15. Is more preferable, and 50/50 to 80/20 is more preferable.
- (a1) / (a2) is in the above range, the durable water repellency of the resulting fiber product is better.
- the total component ratio of the monomers of the component (A) in the non-fluorinated acrylic polymer is the total amount of the monomer components constituting the non-fluorinated polymer in terms of water repellency and durable water repellency of the resulting fiber product.
- the content is preferably 50 to 100% by mass, more preferably 55 to 100% by mass, and still more preferably 60 to 100% by mass.
- the non-fluorinated acrylic polymer can improve the water repellency of the resulting fiber product and the emulsion stability of the non-fluorinated acrylic polymer during emulsion polymerization or dispersion polymerization and in the composition after polymerization, (A) In addition to the components, (B1) a compound represented by the following formula (I-1) having an HLB of 7 to 18, (B2) a compound represented by the following formula (II-1) having an HLB of 7 to 18, And / or (B3) at least one type of reactivity selected from compounds obtained by adding an alkylene oxide of 2 to 4 carbon atoms to an oil and fat having a hydroxyl group and a polymerizable unsaturated group having a HLB of 7 to 18 It is preferable to contain the emulsifier (B) (Hereinafter, it is also called "(B) component.”) As a monomer component.
- R 3 represents hydrogen or a methyl group
- X represents a linear or branched alkylene group having 1 to 6 carbon atoms
- Y 1 represents an alkyleneoxy group having 2 to 4 carbon atoms
- R 4 represents a C 13-17 monovalent unsaturated hydrocarbon group having a polymerizable unsaturated group
- Y 2 includes an C 2-4 alkyleneoxy group. Represents a divalent group.
- Reactive emulsifier is an emulsifying dispersant having radical reactivity, that is, a surfactant having one or more polymerizable unsaturated groups in the molecule, such as (meth) acrylic acid ester It can be copolymerized with a monomer.
- HLB is an HLB value calculated by the Griffin method, assuming that the ethyleneoxy group is a hydrophilic group and all other groups are lipophilic groups.
- the HLB of the compounds (B1) to (B3) used in the present embodiment is 7 to 18, and the emulsion polymerization or dispersion polymerization of the non-fluorinated acrylic polymer and the emulsion in the composition after polymerization are performed. From the viewpoint of stability (hereinafter simply referred to as emulsion stability), 9 to 15 is preferable. Furthermore, it is more preferable to use together 2 or more types of reactive emulsifiers (B) which have different HLB within the said range in the storage stability of a water-repellent composition.
- R 3 is hydrogen or a methyl group, and in terms of copolymerizability with the component (A) More preferably, it is a methyl group.
- X is a linear or branched alkylene group having 1 to 6 carbon atoms, and in view of the emulsion stability of the non-fluorinated acrylic polymer of the present embodiment, a linear alkylene group having 2 to 3 carbon atoms is more preferable.
- Y 1 is a divalent group containing an alkyleneoxy group having 2 to 4 carbon atoms.
- the type, combination and addition number of the alkyleneoxy group in Y 1 can be appropriately selected so as to be within the above-mentioned HLB range. Moreover, when the alkyleneoxy group is 2 or more types, they can have a block addition structure or a random addition structure.
- R 3 represents hydrogen or a methyl group
- X represents a linear or branched alkylene group having 1 to 6 carbon atoms
- a 1 O is an alkyleneoxy group having 2 to 4 carbon atoms
- m can be appropriately selected so as to fall within the above HLB range, and specifically, an integer of 1 to 80 is preferable, and when m is 2 or more, m A 1 O are the same. Or they may be different.
- R 3 is hydrogen or a methyl group, and more preferably a methyl group in terms of copolymerizability with the component (A).
- X is a linear or branched alkylene group having 1 to 6 carbon atoms, and in view of the emulsion stability of the non-fluorinated acrylic polymer, a linear alkylene group having 2 to 3 carbon atoms is more preferable.
- a 1 O is an alkyleneoxy group having 2 to 4 carbon atoms. The type and combination of A 1 O and the number of m can be appropriately selected so as to fall within the above-mentioned HLB range.
- m is preferably an integer of 1 to 80, more preferably an integer of 1 to 60.
- m A 1 O may be the same or different. Further, when A 1 O is two or more, they may have a block addition structure or random addition structure.
- the reactive emulsifier (B1) represented by the above general formula (I-2) can be obtained by a conventionally known method, and is not particularly limited. Further, they can be easily obtained from commercial products, and examples thereof include “Latemul PD-420", “Latemul PD-430", “Latemur PD-450” and the like manufactured by Kao Corporation.
- R 4 is a monovalent unsaturated carbon having 13 to 17 carbon atoms having a polymerizable unsaturated group.
- a hydrogen group such as tridecenyl group, tridecadienyl group, tetradecenyl group, tetradienyl group, pentadecenyl group, pentadecadienyl group, pentadecatrienyl group, heptadecenyl group, heptadecadienyl group, heptadecatrienyl group, etc.
- R 4 is more preferably a C 14-16 monovalent unsaturated hydrocarbon group.
- Y 2 is a divalent group containing an alkyleneoxy group having 2 to 4 carbon atoms.
- the type, combination and addition number of the alkyleneoxy group in Y 2 can be appropriately selected so as to fall within the above-mentioned HLB range.
- the alkyleneoxy group is 2 or more types, they can have a block addition structure or a random addition structure.
- the alkyleneoxy group is more preferably an ethyleneoxy group.
- R 4 represents a C 13-17 monovalent unsaturated hydrocarbon group having a polymerizable unsaturated group, and A 2 O represents a C 2-4 alkyleneoxy group; N can be appropriately selected so as to fall within the above HLB range, and specifically, an integer of 1 to 50 is preferable, and when n is 2 or more, n A 2 O are the same. May also be different.
- N can be appropriately selected so as to fall within the above HLB range, and specifically, an integer of 1 to 50 is preferable, and when n is 2 or more, n A 2 O are the same. May also be different.
- R 4 in the compound represented by the above Formula (II-2) are the same as those for R 4 in the above-mentioned general formula (II-1).
- a 2 O is an alkyleneoxy group having 2 to 4 carbon atoms. From the viewpoint of the emulsion stability of the non-fluorinated acrylic polymer, the type and combination of A 2 O and the number of n can be appropriately selected so as to fall within the above-mentioned HLB range.
- a 2 O is more preferably an ethyleneoxy group
- n is preferably an integer of 1 to 50, more preferably an integer of 5 to 20, further preferably an integer of 8 to 14 preferable.
- n A 2 O's may be the same or different. Further, when A 2 O is more than two, they may have a block addition structure or random addition structure.
- the reactive emulsifier (B2) represented by the above general formula (II-2) used in the present embodiment adds an alkylene oxide to phenol having a corresponding unsaturated hydrocarbon group by a conventionally known method.
- the phenol having the corresponding unsaturated hydrocarbon group includes, in addition to pure products or mixtures produced industrially, those present as pure products or mixtures extracted and purified from plants and the like.
- 3- [8 (Z), 11 (Z), 14-pentadecatrienyl] phenol 3- [8 (Z), 11 (Z), extracted from cashew nut shells etc. and collectively referred to as cardanol.
- the reactive emulsifier (B3) used in this embodiment is a compound obtained by adding an alkylene oxide having a carbon number of 2 to 4 to an oil and fat having a hydroxyl group and a polymerizable unsaturated group having an HLB of 7 to 18 .
- unsaturated fatty acids (palmitoleic acid, oleic acid, linoleic acid, ⁇ -linolenic acid, arachidonic acid, eicosapentaenoic acid, docosapentaenoic acid, etc.) may be contained.
- triglycerides of fatty acids including mono- or diglycerides of good fatty acids, at least one hydroxy unsaturated fatty acid (ricinoleic acid, lisinoelliidic acid, 2-hydroxytetracosenoic acid etc).
- an alkylene oxide adduct of triglyceride of fatty acid containing at least one hydroxy unsaturated fatty acid is preferred, and castor oil (triglyceride of fatty acid containing ricinoleic acid) has 2 to 4 carbon atoms
- the alkylene oxide adduct of is more preferred, and the ethylene oxide adduct of castor oil is even more preferred.
- the addition mole number of the alkylene oxide can be appropriately selected so as to fall within the above-mentioned HLB range, and from the viewpoint of the emulsion stability of the non-fluorinated acrylic polymer, 20 to 50 moles are more preferable, and 25 to 45 Molar is more preferred.
- alkylene oxide is 2 or more types, they can have a block addition structure or a random addition structure.
- the reactive emulsifier (B3) used in this embodiment can be synthesized by adding an alkylene oxide to a fat and oil having a hydroxy group and a polymerizable unsaturated group by a conventionally known method, and is particularly limited. It is not a thing. For example, it can be synthesized by adding a predetermined amount of alkylene oxide at 120 to 170 ° C. under pressure using an alkaline catalyst such as caustic soda or potassium hydroxide to triglyceride of fatty acid containing ricinoleic acid, ie castor oil .
- an alkaline catalyst such as caustic soda or potassium hydroxide
- the component ratio of the monomer of the component (B) in the non-fluorinated acrylic polymer is the non-fluorinated acrylic polymer from the viewpoint of being able to further improve the water repellency of the obtained fiber product and the emulsion stability of the non-fluorinated acrylic polymer.
- the amount is preferably 0.5 to 20% by mass, more preferably 1 to 15% by mass, and still more preferably 3 to 10% by mass with respect to the total amount of the monomer components constituting the
- the non-fluorinated acrylic polymer is capable of further improving the durable water repellency of the fiber product to be obtained, and the following (C1), (C2), (C3), (C4) and (C5) Containing at least one second (meth) acrylic acid ester monomer (C) (hereinafter also referred to as “component (C)”) selected from the group consisting of preferable.
- (C1) is a (meth) acrylic acid ester monomer represented by the following general formula (C-1) other than (C5).
- R 5 represents hydrogen or a methyl group
- R 6 represents at least one selected from the group consisting of a hydroxy group, an amino group, a carboxy group, an epoxy group, an isocyanate group and a (meth) acryloyloxy group It represents a C 1-11 monovalent chain hydrocarbon group having one type of functional group. However, the number of (meth) acryloyloxy groups in the molecule is 2 or less.
- (C2) is a (meth) acrylic acid ester monomer represented by the following general formula (C-2).
- R 7 represents hydrogen or a methyl group
- R 8 represents a monovalent cyclic hydrocarbon group having 1 to 11 carbon atoms which may have a substituent.
- (C3) is a methacrylic acid ester monomer represented by the following general formula (C-3).
- R 9 represents an unsubstituted monovalent hydrocarbon chain hydrocarbon group having 1 to 4 carbon atoms.
- (C4) is a (meth) acrylic acid ester monomer represented by the following general formula (C-4).
- R 10 represents hydrogen or a methyl group
- p represents an integer of 2 or more
- S represents an (p + 1) -valent organic group
- T has a polymerizable unsaturated group 1 Represents a valent organic group.
- (C5) is a (meth) acrylic acid ester monomer represented by the following general formula (C-5).
- R 11 represents hydrogen or a methyl group
- R 12 has 3 to 6 carbon atoms having at least one functional group selected from the group consisting of chloro group and bromo group and a hydroxy group And a monovalent chain saturated hydrocarbon group of ]
- the monomer (C1) has a carbon number having at least one functional group selected from the group consisting of a hydroxy group, an amino group, a carboxy group, an epoxy group, an isocyanate group and a (meth) acryloyloxy group in the ester moiety (Meth) acrylic acid ester monomers having 1 to 11 monovalent chain hydrocarbon groups, and (meth) acrylic acid ester monomers other than the above (C5).
- the monovalent chain hydrocarbon group having 1 to 11 carbon atoms is at least one selected from the group consisting of a hydroxy group, an amino group, a carboxy group, an epoxy group, and an isocyanate group from the viewpoint of being capable of reacting with a crosslinking agent. It is preferable to have a functional group.
- the isocyanate group may be a blocked isocyanate group protected by a blocking agent.
- the linear hydrocarbon group may be linear or branched, and may be a saturated hydrocarbon group or an unsaturated hydrocarbon group.
- the chain hydrocarbon group may further have a substituent in addition to the above functional group.
- it is preferably linear and / or a saturated hydrocarbon group.
- the monomer (C1) examples include 2-hydroxyethyl (meth) acrylate, dimethylaminoethyl (meth) acrylate, glycidyl (meth) acrylate, 1,1-bis (acryloyloxymethyl) ethyl An isocyanate etc. are mentioned.
- One of these monomers may be used alone, or two or more of these monomers may be used in combination.
- 2-hydroxyethyl (meth) acrylate, glycidyl (meth) acrylate and 1,1-bis (acryloyloxymethyl) ethyl isocyanate are preferable in that the durable water repellency of the obtained fiber product can be further improved.
- dimethylaminoethyl (meth) acrylate is preferable in that the texture of the obtained fiber product is further improved.
- the constituent ratio of the above-mentioned (C1) monomer in the non-fluorinated acrylic polymer is the total amount of the monomer components constituting the non-fluorinated acrylic polymer from the viewpoint of water repellency and texture of the obtained fiber product,
- the content is preferably 1 to 30% by mass, more preferably 3 to 25% by mass, and still more preferably 5 to 20% by mass.
- the monomer (C2) is a (meth) acrylic acid ester monomer having a monovalent cyclic hydrocarbon group having 1 to 11 carbon atoms in the ester portion, and the cyclic hydrocarbon group is an isobornyl group, A cyclohexyl group, a dicyclopentanyl group etc. are mentioned.
- These cyclic hydrocarbon groups may have a substituent such as an alkyl group.
- a hydrocarbon group is selected such that the total carbon number of the substituent and the cyclic hydrocarbon group is 11 or less.
- it is preferable that these cyclic hydrocarbon groups are directly bonded to an ester bond from the viewpoint of further improving the durable water repellency.
- the cyclic hydrocarbon group may be alicyclic or aromatic, and in the case of alicyclic, it may be a saturated hydrocarbon group or an unsaturated hydrocarbon group.
- Specific examples of the monomer include isobornyl (meth) acrylate, cyclohexyl (meth) acrylate, and dicyclopentanyl (meth) acrylate.
- One of these monomers may be used alone, or two or more of these monomers may be used in combination. Among them, isobornyl (meth) acrylate and cyclohexyl methacrylate are preferable, and isobornyl methacrylate is more preferable, from the viewpoint that the durable water repellency of the obtained fiber product can be further improved.
- the constituent ratio of the above-mentioned (C2) monomer in the non-fluorinated acrylic polymer is, with respect to the water repellency and texture of the obtained fiber product, relative to the total amount of the monomer components constituting the non-fluorinated acrylic polymer,
- the content is preferably 1 to 30% by mass, more preferably 3 to 25% by mass, and still more preferably 5 to 20% by mass.
- the monomer (C3) is a methacrylic acid ester monomer in which an unsubstituted monovalent linear hydrocarbon group having 1 to 4 carbon atoms is directly bonded to the ester bond of the ester portion.
- the chain hydrocarbon group having 1 to 4 carbon atoms a linear hydrocarbon group having 1 to 2 carbon atoms and a branched hydrocarbon group having 3 to 4 carbon atoms are preferable.
- Examples of the chain hydrocarbon group having 1 to 4 carbon atoms include methyl group, ethyl group, n-propyl group, isopropyl group, n-butyl group and t-butyl group.
- the compound examples include methyl methacrylate, ethyl methacrylate, n-propyl methacrylate, isopropyl methacrylate, n-butyl methacrylate, isobutyl methacrylate and t-butyl methacrylate.
- One of these monomers may be used alone, or two or more of these monomers may be used in combination.
- methyl methacrylate, isopropyl methacrylate and t-butyl methacrylate are preferable, and methyl methacrylate is more preferable, since the durable water repellency of the obtained fiber product can be further improved.
- the constituent ratio of the above-mentioned (C3) monomer in the non-fluorinated acrylic polymer is 1 with respect to the total amount of the monomer components constituting the non-fluorinated polymer from the viewpoint of water repellency and texture of the obtained fiber product.
- the content is preferably 30% by mass, more preferably 3 to 25% by mass, and still more preferably 5 to 20% by mass.
- the monomer (C4) is a (meth) acrylic acid ester monomer having three or more polymerizable unsaturated groups in one molecule.
- a multifunctional (meth) acrylic acid ester having three or more (meth) acryloyloxy groups in one molecule, wherein T in the general formula (C-4) is a (meth) acryloyloxy group Preferred are mers.
- p T's may be the same or different.
- Specific compounds include, for example, ethoxylated isocyanuric acid triacrylate, tetramethylolmethane tetraacrylate, tetramethylolmethane tetramethacrylate, trimethylolpropane triacrylate, trimethylolpropane trimethacrylate, pentaerythritol triacrylate, pentaerythritol trimethacrylate, Dipentaerythritol hexaacrylate, dipentaerythritol hexamethacrylate and the like can be mentioned.
- One of these monomers may be used alone, or two or more of these monomers may be used in combination.
- tetramethylolmethane tetraacrylate and ethoxylated isocyanuric acid triacrylate are more preferable in that the durable water repellency of the obtained fiber product can be further improved.
- the constituent ratio of the above-mentioned (C4) monomer in the non-fluorinated acrylic polymer is the total amount of the monomer components constituting the non-fluorinated acrylic polymer from the viewpoint of water repellency and texture of the obtained fiber product,
- the content is preferably 1 to 30% by mass, more preferably 3 to 25% by mass, and still more preferably 5 to 20% by mass.
- the monomer (C5) has a monovalent chain saturated hydrocarbon group having 3 to 6 carbon atoms having at least one functional group selected from the group consisting of a chloro group and a bromo group and a hydroxy group. .
- R 11 is hydrogen or a methyl group. From the viewpoint of the durable water repellency of the resulting textile, R 11 is preferably a methyl group.
- R 12 is a C 3 to C 6 monovalent chain saturated hydrocarbon group having a hydroxy group and at least one functional group selected from the group consisting of a chloro group and a bromo group.
- the chain saturated hydrocarbon group may be linear or branched. When the chain saturated hydrocarbon group is linear, the durable water repellency of the obtained fiber product is more excellent.
- the carbon number of the chain saturated hydrocarbon group is preferably 3 to 4 and more preferably 3 from the viewpoint of the durable water repellency of the resulting fiber product.
- the chain saturated hydrocarbon group preferably has one or two chloro groups and one hydroxy group in view of the durable water repellency of the resulting fiber product, and one chloro group, It is more preferable to have one hydroxy group.
- the linear saturated hydrocarbon group is a hydroxy group at the ⁇ -position (the carbon atom next to the carbon atom bonded to CH 2 CRCR 11 (CO) O—) It is further preferable to have Specific examples of the above linear saturated hydrocarbon group include, for example, 3-chloro-2-hydroxypropyl group, 3-chloro-2-hydroxybutyl group, 5-chloro-2-hydroxypentyl group, 3-chloro-2 And -hydroxy-2-methylpropyl and 3-bromo-2-hydroxypropyl.
- the monomer (C5) include 3-chloro-2-hydroxypropyl (meth) acrylate, 3-chloro-2-hydroxybutyl (meth) acrylate, and 5- (meth) acrylate.
- Examples include chloro-2-hydroxypentyl and 3-bromo-2-hydroxypropyl (meth) acrylate.
- 3-chloro-2-hydroxypropyl (meth) acrylate is preferable, and 3-chloro-2-hydroxypropyl methacrylate is more preferable, in that the durable water repellency of the obtained fiber product can be further improved.
- the component ratio of the above-mentioned (C5) monomer in the non-fluorinated acrylic polymer is 1 with respect to the total amount of the monomer components constituting the non-fluorinated acrylic polymer in view of the durable water repellency of the obtained fiber product.
- the content is preferably 30% by mass, more preferably 3 to 25% by mass, and still more preferably 5 to 20% by mass.
- the total component ratio of the above-mentioned monomer of the component (C) in the non-fluorinated acrylic polymer is the total amount of monomer components constituting the non-fluorinated acrylic polymer from the viewpoint of water repellency and texture of the obtained fiber product. On the other hand, it is preferably 1 to 30% by mass, more preferably 3 to 25% by mass, and still more preferably 5 to 20% by mass.
- the non-fluorinated acrylic polymer is a monofunctional monomer (D) copolymerizable with these (hereinafter also referred to as “(D) component”) Can be contained in the range which does not impair the effect of the present invention.
- Examples of the monomer (D) include (meth) acryloyl morpholine, (meth) acrylic acid ester having a hydrocarbon group other than the (A) component and the (C) component, (meth) acrylic acid, and fumaric acid.
- the (meth) acrylic acid ester having a hydrocarbon group other than the component (A) and the component (C) has a vinyl group, a hydroxy group, an amino group, an epoxy group and an isocyanate group, a blocked isocyanate group as a hydrocarbon group.
- the like and may have a substituent other than a group capable of reacting with the crosslinking agent such as quaternary ammonium group, and may be an ether bond, an ester bond, an amide bond, or a urethane bond And the like.
- Examples of the (meth) acrylic acid ester other than the component (A) and the component (C) include methyl acrylate, 2-ethylhexyl (meth) acrylate, benzyl (meth) acrylate, ethylene glycol di (meth) acrylate, etc. Can be mentioned.
- the component ratio of the monomer of the component (D) in the non-fluorinated acrylic polymer is the total amount of the monomer components constituting the non-fluorinated acrylic polymer from the viewpoint of water repellency and texture of the obtained fiber product. And 10% by mass or less.
- the non-fluorinated acrylic polymer has at least one functional group selected from the group consisting of a hydroxy group, an amino group, a carboxy group, an epoxy group and an isocyanate group capable of reacting with a crosslinking agent, resulting in the durability of the obtained fiber product. It is preferable because the water repellency is further improved.
- the isocyanate group may form a blocked isocyanate group protected by a blocking agent.
- a non-fluorine acrylic polymer has an amino group from the viewpoint of further improving the texture of the obtained fiber product.
- the non-fluorinated acrylic polymer is at least one monomer of vinyl chloride and vinylidene chloride in that it can further improve the water repellency of the resulting fiber product and the peel strength to the coating. It is preferable to contain (E) (hereinafter, also referred to as “component (E)”) as a monomer component.
- the monomer (E) of at least one of vinyl chloride and vinylidene chloride used in the present embodiment is preferably vinyl chloride in view of the water repellency of the obtained fiber product and the peel strength to the coating.
- the composition ratio of the monomer of the component (E) in the non-fluorinated acrylic polymer is the total amount of the monomer components constituting the non-fluorinated acrylic polymer from the viewpoint of being able to further improve the peel strength to the coating of the obtained fiber product.
- the amount is preferably 1 to 45% by mass, more preferably 3 to 40% by mass, and still more preferably 5 to 35% by mass.
- the non-fluorinated acrylic polymer can be produced by a radical polymerization method. Further, among the radical polymerization methods, it is preferable to polymerize by the emulsion polymerization method or the dispersion polymerization method from the viewpoint of the performance and the environment of the obtained water repellent.
- a non-fluorinated acrylic polymer can be obtained by emulsion polymerization or dispersion polymerization of the (meth) acrylic acid ester monomer (A) represented by the above general formula (A-1) in a medium. More specifically, for example, (A) component and optionally the above (B) component, the above (C) component, the above (D) component and the above (E) component, and an emulsifying aid or dispersion in a medium An adjuvant is added and the mixture is emulsified or dispersed to obtain an emulsion or dispersion.
- the polymerization reaction can be initiated to polymerize the monomer and the reactive emulsifier.
- a homomixer, a high pressure emulsifier, an ultrasonic wave etc. are mentioned.
- emulsification adjuvant or dispersion adjuvant As the above-mentioned emulsification adjuvant or dispersion adjuvant (hereinafter, also referred to as "emulsion adjuvant etc.”), nonionic surfactants other than the above-mentioned reactive emulsifier (B), cationic surfactants, anionic surfactants, and One or more selected from amphoteric surfactants can be used.
- the content of the emulsification aid and the like is preferably 0.5 to 30 parts by mass, more preferably 1 to 20 parts by mass, and more preferably 1 to 10 parts by mass with respect to 100 parts by mass of all monomers.
- the dispersion stability of the mixed liquid tends to be further improved when the content of the above-mentioned emulsification adjuvant and the like is 0.5 parts by mass or more, and the content of the emulsification adjuvant and the like is 30 parts by mass or less.
- the water repellency of the water repellent composition tends to be further improved.
- water is preferable, and water and an organic solvent may be mixed as needed.
- organic solvent at this time include alcohols such as methanol and ethanol, esters such as ethyl acetate, ketones such as acetone and methyl ethyl ketone, ethers such as diethyl ether, etc., propylene glycol, dipropylene glycol, tripropylene, etc. Glycols such as glycols can be mentioned.
- the ratio of water to the organic solvent is not particularly limited.
- polymerization initiator As said polymerization initiator, well-known polymerization initiators, such as an azo type, a peroxide type, or a redox type, can be used suitably.
- the content of the polymerization initiator is preferably 0.01 to 2 parts by mass with respect to 100 parts by mass of all the monomers. When the content of the polymerization initiator is in the above range, a non-fluorinated acrylic polymer having a weight average molecular weight of 100,000 or more can be efficiently produced.
- a chain transfer agent such as dodecyl mercaptan or t-butyl alcohol may be used for the purpose of molecular weight adjustment.
- a polymerization inhibitor for molecular weight adjustment.
- a polymerization inhibitor a non-fluorinated acrylic polymer having a desired weight average molecular weight can be easily obtained.
- the temperature of the polymerization reaction is preferably 20 ° C to 150 ° C. When the temperature is 20 ° C. or more, the polymerization tends to be sufficient, and when the temperature is 150 ° C. or less, the control of the heat of reaction tends to be easy.
- the weight average molecular weight of the non-fluorinated acrylic polymer obtained can be adjusted by increasing or decreasing the content of the polymerization initiator, the chain transfer agent, and the polymerization inhibitor described above, and the melt viscosity at 105 ° C. It can adjust by the increase and decrease of content of a functional monomer, and content of a polymerization initiator. In order to reduce the melt viscosity at 105 ° C., the content of the monomer having two or more polymerizable functional groups may be reduced, or the content of the polymerization initiator may be increased.
- the content of the non-fluorinated acrylic polymer in the polymer emulsion or dispersion obtained by emulsion polymerization or dispersion polymerization is 10 to 10 parts of the total amount of the emulsion or dispersion from the viewpoint of storage stability and handling of the composition.
- the content is preferably 50% by mass, and more preferably 20 to 40% by mass.
- Examples of the above-mentioned hydrocarbon group-containing compounds include Neoseed NR-90 (manufactured by Nikka Chemical Co., Ltd.), NR-158 (manufactured by Nikka Chemical Co., Ltd.), TH-44 (manufactured by Nikka Chemical Co., Ltd.) ), PW-182 (Daiwa Chemical Co., Ltd.) Phoball, RSH (Huntsman Japan Co., Ltd.), Palladium ECO-500 (Ohara Paradium Chemical Co., Ltd.), NX018 (Nanotex Co., Ltd.), etc. Can be mentioned.
- the water repellent composition of the present embodiment includes, in addition to the components described above, a polyfunctional isocyanate compound, a surfactant, an antifoamer, an organic acid, an inorganic acid, an alcohol, an antibacterial agent, an antifungal agent, and pH adjustment.
- the polyfunctional isocyanate compound is not particularly limited as long as it is a compound having two or more isocyanate groups in the molecule, and a known polyisocyanate compound can be used.
- polyfunctional isocyanate compounds include diisocyanate compounds such as alkylene diisocyanate, aryl diisocyanates and cycloalkyl diisocyanates, and modified polyisocyanate compounds such as dimers, trimers or tetramers of these diisocyanate compounds.
- the carbon number of the alkylene diisocyanate is preferably 1 to 12.
- diisocyanate compound examples include 2,4 or 2,6-tolylene diisocyanate, ethylene diisocyanate, propylene diisocyanate, 4,4-diphenylmethane diisocyanate, p-phenylene diisocyanate, tetramethylene diisocyanate, hexamethylene diisocyanate, decamethylene diisocyanate, dodeca Methylene diisocyanate, 2,4,4-trimethylhexamethylene-1,6-diisocyanate, phenylene diisocyanate, tolylene or naphthalene diisocyanate, 4,4'-methylene-bis (phenylisocyanate), 2,4'-methylene-bis ( Phenylisocyanate), 3,4'-methylene-bis (phenylisocyanate), 4,4'-ethylene-bis (phenyliso) Anate), ⁇ , ⁇ ′-diisocyanate-1,3-dimethylbenzene, ⁇
- triisocyanate compounds examples include triphenylmethane triisocyanate, tris (isocyanatophenyl) -thiophosphate and the like.
- a tetraisocyanate compound dimethyl triphenyl methane tetraisocyanate etc. are mentioned, for example.
- the modified polyisocyanate compound derived from the diisocyanate compound is not particularly limited as long as it has two or more isocyanate groups, and, for example, biuret structure, isocyanurate structure, urethane structure, uretdione structure, allophanate structure, trimeric Examples thereof include polyisocyanates having a body structure and the like, and adducts of aliphatic isocyanates of trimethylolpropane and the like.
- the polyisocyanate compound can be used singly or in combination of two or more.
- the isocyanate group contained in the polyfunctional isocyanate compound may be as it is or may be a blocked isocyanate group blocked by a blocking agent.
- blocking agents pyrazoles such as 3,5-dimethylpyrazole, 3-methylpyrazole, 3,5-dimethyl-4-nitropyrazole, 3,5-dimethyl-4-bromopyrazole, and pyrazole; phenol, methylphenol, Phenols such as chlorophenol, iso-butylphenol, tert-butylphenol, iso-amylphenol, octylphenol, nonylphenol; lactams such as ⁇ -caprolactam, ⁇ -valerolactam, ⁇ -butyrolactam; malonic acid dimethyl ester, malonic acid diethyl ester , Active methylene compounds such as acetylacetone, methyl acetoacetate and ethyl acetoacetate; formaldoxime, acetoaldoxime, acetone oxime,
- the water dispersible isocyanate which provided water dispersibility to polyisocyanate can also be used by introduce
- known catalysts such as organotin and organozinc can be used in combination to promote the reaction between the amino group and the isocyanate group.
- the compounding amount of the polyfunctional isocyanate compound in the water repellent composition of the present embodiment is 100 parts by mass in total of the compounding amount of the amino-modified silicone and the compounding amount of the silicone resin from the viewpoint of water repellency, durable water repellency and texture.
- the amount is preferably 1 to 200 parts by mass, and more preferably 5 to 100 parts by mass.
- the compounding amount of the polyfunctional isocyanate compound in the second water repellent composition of the present embodiment is the (IV) component, the (V) component, and the (II) component from the viewpoint of water repellency, durable water repellency and texture.
- the amount is preferably 1 to 200 parts by mass, and more preferably 5 to 100 parts by mass with respect to 100 parts by mass of the total blending amount.
- the compounding amount of the polyfunctional isocyanate compound is From the viewpoint of water repellency, durable water repellency and texture, the amount is preferably 1 to 200 parts by mass, and more preferably 5 to 100 parts by mass, with respect to 100 parts by mass of the total amount of water repellent components.
- the total content of the components (I) and (II) in the water repellent component is preferably 1% by mass or more based on the total amount of the water repellent component, and more preferably 10 to 90% by mass. .
- the total blending amount of the components (IV) and (V) and the component (II) in the water repellent component is 1% by mass or more based on the total amount of the water repellent component Is preferable, and 10 to 90% by mass is more preferable.
- the surfactant essentially includes a polyalkylene oxide adduct and may further contain other surfactant.
- the other surfactant is, for example, used to expand the temperature range in which the state of the emulsion is stably maintained, and to adjust the amount of foaming generated when it is mixed with water to prepare a diluted solution.
- the other surfactant may be any one of a nonionic surfactant, an anionic surfactant, a cationic surfactant, and an amphoteric surfactant.
- the other surfactants can be used singly or in combination of two or more.
- the antifoaming agent is not particularly limited, but, for example, a fat-based antifoaming agent such as castor oil, sesame oil, linseed oil, animal or vegetable oil; a fatty acid-based antifoaming agent such as stearic acid, oleic acid or palmitic acid; Fatty acid ester antifoaming agents such as isoamyl, distearyl succinate, ethylene glycol distearate, butyl stearate; polyoxyalkylene monohydric alcohol, di-t-amyl phenoxyethanol, 3-heptanol, 2-ethylhexanol, etc.
- a fat-based antifoaming agent such as castor oil, sesame oil, linseed oil, animal or vegetable oil
- a fatty acid-based antifoaming agent such as stearic acid, oleic acid or palmitic acid
- Fatty acid ester antifoaming agents such as isoa
- Alcohol antifoaming agents such as 3-heptyl cellosolve, nonyl cellosolve, 3-heptyl carbitol; Phosphoric acid ester antifoaming agents such as tributyl phosphate and tris (butoxyethyl) phosphate; amines such as diamylamine Antifoaming agent; Polyalkylene Sulfate-based defoaming agents such as sodium lauryl sulfate; amides, amide-based defoaming agent such as acylate polyamine mineral oil, and the like.
- An antifoamer can be used individually by 1 type or in combination of 2 or more types.
- the organic acid is not particularly limited, and examples thereof include lactic acid, acetic acid, propionic acid, maleic acid, oxalic acid, formic acid, methanesulfonic acid, toluenesulfonic acid and the like.
- An organic acid can be used individually by 1 type or in combination of 2 or more types.
- the inorganic acid is not particularly limited, and examples thereof include hydrogen chloride, sulfuric acid and nitric acid.
- An inorganic acid can be used individually by 1 type or in combination of 2 or more types.
- the alcohol is not particularly limited, and examples thereof include ethanol, isopropanol, glycerin, trimethylolpropane, diethylene glycol, triethylene glycol, dipropylene glycol, propylene glycol and the like.
- the alcohols may be used alone or in combination of two or more.
- the antistatic agent it is preferable to use one that does not easily inhibit the water repellency performance.
- the antistatic agent for example, cationic surfactants such as higher alcohol sulfuric acid ester salt, sulfated oil, sulfonate, quaternary ammonium salt, imidazoline type quaternary salt, polyethylene glycol type, polyhydric alcohol ester type And nonionic surfactants, imidazoline type quaternary salts, amphoteric surfactants such as alanine type and betaine type, and the above-mentioned antistatic polymer and polyalkylamine as polymer compound types.
- the antistatic agent can be used singly or in combination of two or more.
- crosslinking agents other than the said polyfunctional isocyanate compound a melamine resin, glyoxal resin etc. can be mentioned, for example.
- a compound having a melamine skeleton can be used.
- polymethylolmelamine such as trimethylolmelamine and hexamethylolmelamine; some or all of methylol groups of polymethylolmelamine have 1 to 6 carbon atoms Alkoxymethylmelamine which is an alkoxymethyl group having an alkyl group; acyloxymethylmelamine in which a part or all of the methylol group of a polymethylolmelamine is an acyloxymethyl group having an acyl group of 2 to 6 carbon atoms, etc. It can be mentioned.
- These melamine resins may be monomers or multimers of dimers or higher, or mixtures of these may be used.
- melamine resins for example, Beckamine APM, Beckamine M-3, Beckamine M-3 (60), Beckamine MA-S, Beckamine J-101, and Beckamine J-101 LF manufactured by DIC Corporation, Union Chemical Industries, Ltd. Unicardin 380K manufactured by Co., Ltd., Riken Resin MM series manufactured by Miki Riken Kogyo Co., Ltd., etc. may be mentioned.
- glioxar resin conventionally known ones can be used.
- examples of the glioxal resin include 1,3-dimethylglyoxal urea resin, dimethylol dihydroxy ethylene urea resin, and dimethylol dihydroxy propylene urea resin.
- the functional groups of these resins may be substituted with other functional groups.
- glyoxal resin for example, Beckamine N-80, Beckamine NS-11, Beckamine LF-K, Beckamine NS-19, Beckamine LF-55P conc, Beckamine NS-210L, Beckamine NS-200, manufactured by DIC Corporation And Beckamine NF-3, Uniresin GS-20E manufactured by Union Chemical Industry Co., Ltd., Riken Resin RG series manufactured by Miki Riken Kogyo Co., Ltd., and Riken Resin MS series.
- a catalyst for the melamine resin and the glyoxal resin from the viewpoint of promoting the reaction.
- Such catalysts are not particularly limited as long as they are commonly used catalysts.
- borofluorinated compounds such as ammonium borofluoride and borofluorinated sub salts
- Neutral metal salt catalysts such as magnesium chloride and magnesium sulfate
- inorganic acids such as phosphoric acid, hydrochloric acid and boric acid.
- organic acids such as citric acid, tartaric acid, malic acid, maleic acid, lactic acid and the like can be used in combination as a co-catalyst.
- catalyst ACX manufactured by DIC Corporation
- catalyst 376 catalyst O
- catalyst M catalyst G
- catalyst X-110 catalyst GT-3
- Catalyst NFC-1 Uni-Catarist 3-P made by Union Chemical Industry Co., Ltd.
- Uni-Catarist MC-109 Miki Riken Kogyo Co., Ltd.
- Riken Fixer RC Series Riken Fixer MX Series, and Riken Fixer RZ- 5 and the like.
- the water repellent composition according to the present embodiment described above can be suitably used for applications such as textile products processing agents, paper products processing agents, leather products processing agents and the like.
- the water repellent composition of this embodiment can be obtained by mixing the amino-modified silicone, the silicone resin, and the alkylpolysiloxane described above.
- the content of each component described above in the water repellent composition of the present embodiment can be the above-described preferable compounding amount.
- the water repellent composition according to the present embodiment may be a one-component type in which an amino-modified silicone, a silicone resin, and an alkylpolysiloxane are previously mixed, or a one-component type in which two of the above three components are mixed. It may be a two-component type divided into one component and another one-component component, or may be a three-component type in which the above three components are respectively separated.
- the three components are preferably dispersed (including emulsification and dissolution) in an aqueous medium from the viewpoint of ease of handling.
- the water repellent composition of the present embodiment comprises component (I), component (II) and component (II) III) Simultaneously dispersing (emulsifying, dissolving) the components into an aqueous medium, or dispersing the dispersion obtained by dispersing at least one of the three components in the aqueous medium and the other components in the aqueous medium It can be obtained by mixing with the above dispersions or by mixing the respective dispersions of the components (I), (II) and (III).
- a method of dispersing each of the above components in an aqueous medium for example, mixing and stirring each component, an aqueous medium, and, if necessary, a dispersant may be mentioned.
- a conventionally known emulsification disperser such as a milder, a high speed stirrer, a homogenizer, an ultrasonic homogenizer, a homomixer, a bead mill, a bead mill, a pearl mill, an ape mill, a basket mill, a ball mill, a nanomizer, an altimizer, a starburst, etc. It may be used.
- emulsifying and dispersing machines can be used alone or in combination of two or more.
- the aqueous medium is preferably water or a mixed solvent of water and a hydrophilic solvent miscible with water.
- hydrophilic solvents include methanol, ethanol, isopropyl alcohol, ethylene glycol, diethylene glycol, hexylene glycol, glycerin, butyl glycol, butyl diglycol, sorbite, N-methylpyrrolidone, dimethylformamide, dimethyl sulfooxide and the like.
- the dispersion preferably further contains a surfactant from the viewpoint of dispersion stability.
- a surfactant is not particularly limited as long as it can improve the emulsion dispersion stability, and, for example, known nonionic surfactants, anionic surfactants, cationic surfactants, etc. Activators, amphoteric surfactants and the like can be mentioned. These can be used singly or in combination of two or more.
- the dispersion may be used as it is as a treatment liquid, or may be used as a treatment liquid by diluting with an aqueous medium or a hydrophobic organic solvent.
- the dispersion may be used as it is unneutralized (without neutralization), or pH may be adjusted by a method such as neutralization of amino-modified silicone. In the case of pH adjustment, the pH of the treatment solution can be adjusted to 5.5 to 6.5.
- the second water repellent composition of the present embodiment can be produced by using the (IV) component and the (V) component in place of the (I) component.
- the method of the present embodiment comprises the step of treating the fiber with a treatment liquid containing the water repellent composition of the present embodiment described above. Through this process, a water repellent fiber product is obtained.
- the material of the fiber is not particularly limited, and natural fibers such as cotton, hemp, silk and wool, semi-synthetic fibers such as rayon and acetate, synthetic fibers such as nylon, polyester, polyurethane and polypropylene and composite fibers thereof, blended fibers Etc.
- the form of the fiber may be any form such as yarn, cloth, non-woven fabric, paper and the like.
- the fibers may be textiles.
- the treatment liquid containing the components (I), (II) and (III) is used in one step.
- a method of treating, a method of treating in two steps using a treatment liquid containing two of the three components and a treatment liquid containing one other component, and three kinds of dispersions containing the three components separately The method of processing in 3 steps can be mentioned.
- the order of processing the respective components may be any order.
- components (IV) and (V) may be blended in the treatment liquid instead of the component (I).
- the above-mentioned treatment solutions and dispersions may be used unneutralized (without neutralization), and may be adjusted to pH 5.5 to 6.5.
- the pH is adjusted, for example, organic acids such as lactic acid, acetic acid, propionic acid, maleic acid, oxalic acid, formic acid, methanesulfonic acid, toluenesulfonic acid; inorganic acids such as hydrogen chloride, sulfuric acid, nitric acid; sodium hydroxide, water Hydroxides such as potassium oxide; Carbonates such as sodium carbonate, sodium hydrogen carbonate, potassium carbonate, sodium sesquicarbonate; Organic amines such as monoethanolamine, diethanolamine, triethanolamine, triethylamine etc .; Ammonia etc. can be used .
- the water repellent composition contains water, it is preferably dried to remove the water after being attached to the fiber.
- the adhesion amount of the water repellent composition of the present embodiment to the fiber can be appropriately adjusted according to the required degree of water repellency, but the adhesion amount of the water repellent composition relative to 100 g of fiber is 0. It is preferable to adjust to 1 to 5 g, and more preferable to adjust to 0.1 to 3 g.
- the adhesion amount of the water repellent composition is 0.1 g or more, the fiber tends to more easily exhibit sufficient water repellency, and when it is 5 g or less, the feeling of the fiber is further improved and economically Also tend to be advantageous.
- the temperature conditions are not particularly limited, but it is preferable to carry out at 110 to 180 ° C. for 1 to 5 minutes, from the viewpoint of water repellency, durable water repellency and texture.
- the water repellent fiber product of this embodiment exhibits excellent water repellency and a soft texture, so it can be used as a downside, coat, blouson, windbreaker, blouse, dress shirt, skirt, slacks, gloves, hat, and futon side. It is suitably used for textile applications such as clothing applications and non-clothing applications such as grounds, duvet covers, curtains or tents.
- Preparation of Amino-Modified Silicone Dispersion 30 parts by mass of KF 8005 (Shin-Etsu Chemical Co., Ltd., trade name) as amino-modified silicone, 0.3 parts by mass of formic acid, and 1 part by mass of ethylene oxide 5 molar adduct of branched alcohol having 12 to 14 carbon atoms Mixed. Next, 68.7 parts by mass of water was added little by little while mixing to the obtained mixture to obtain a dispersion containing 30% by mass of amino-modified silicone.
- KF 8005 Shin-Etsu Chemical Co., Ltd., trade name
- Preparation Examples A2 to A7 A dispersion containing 30% by mass of amino-modified silicone was obtained in the same manner as in Preparation Example A1 except that the amino-modified silicone was changed from KF 8005 to the amino-modified silicone described in Table 1.
- KF-868, KF-864, and KF-393 are trade names of Shin-Etsu Chemical Co., Ltd.
- SF-8417 and BY16-853U are trade names of Toray Dow Corning, Inc.
- XF42-B1989 is a trade name of Momentive Performance Materials Japan LLC.
- Preparation Example A8 As amino-modified silicone, 30 parts by mass of SF-8417 (trade name, manufactured by Toray Dow Corning Co., Ltd.) and 1 part by mass of ethylene oxide 5 molar adduct of a branched alcohol having 12 to 14 carbon atoms were mixed. Next, 69.0 parts by mass of water was added little by little to the obtained mixture while mixing to obtain a dispersion containing 30% by mass of amino-modified silicone.
- SF-8417 trade name, manufactured by Toray Dow Corning Co., Ltd.
- Preparation Example B3 KF7312L (manufactured by Shin-Etsu Chemical Co., Ltd., trade name, solvent: low viscosity methylpolysiloxane (KF-96L-2cs, dimethylsilicone), trimethylsilyl group-containing polysiloxane: solvent instead of a mixture of MQ-1600 and dimethylsilicone
- KF-96L-2cs low viscosity methylpolysiloxane
- MQ-1600 dimethylsilicone
- Preparation Example B4 25 parts by mass of MQ-1600 (trimethylsilyl group-containing polysiloxane, manufactured by Toray Dow Corning Co., Ltd.) as a silicone resin, and 75 parts by mass of dimethyl silicone (6 cs, manufactured by Toray Dow Corning Co., Ltd.) as an alkylpolysiloxane Parts were added and mixed until the silicone resin was dissolved to obtain a mixture. 30 parts by mass of the obtained mixture and 1 part by mass of a 5-mol ethylene oxide adduct of a branched alcohol having 12 to 14 carbon atoms were mixed. Subsequently, 69 parts by mass of water was added while mixing little by little, to obtain a dispersion containing 22.5% by mass of an alkylpolysiloxane and 7.5% by mass of a silicone resin.
- MQ-1600 trimethylsilyl group-containing polysiloxane, manufactured by Toray Dow Corning Co., Ltd.
- dimethyl silicone 6 cs, manufactured by
- Preparation Example C Dispersion of methyl ethyl ketoxime blocked product of reaction product of trimethylolpropane and toluene diisocyanate
- a reaction product of trimethylolpropane and toluene diisocyanate a reaction product of Polurene AD (trimethylolpropane and toluene diisocyanate (the mass ratio of the 2,4 isomer to the 2,6 isomer is 80:20) Of 75% by mass, solvent: ethyl acetate, manufactured by SAPICI, trade name) was prepared.
- Preparation Example C 2 Dispersion of isocyanurate type methyl ethyl ketoxime blocked compound of hexamethylene diisocyanate To a reaction vessel, add 1 mol of duranate THA-100 (isocyanurate type of hexamethylene diisocyanate, NCO functional group number: 3, content: 100% by mass, manufactured by Asahi Kasei Chemicals Corporation, trade name), and methyl isobutyl ketone, 60 Heated to ⁇ 70 ° C. Then, 3 moles of methyl ethyl ketoxime is slowly charged, and reacted at 60 to 70 ° C.
- duranate THA-100 isocyanurate type of hexamethylene diisocyanate, NCO functional group number: 3, content: 100% by mass, manufactured by Asahi Kasei Chemicals Corporation, trade name
- Preparation Example C3 Dimethylpyrazole block of hexamethylene diisocyanate biuret
- duranate 24A-100 biuret type of hexamethylene diisocyanate, NCO functional number: 3, content 100% by mass, Asahi Kasei Chemicals Co., Ltd., trade name
- methyl isobutyl ketone methyl isobutyl ketone
- 3 moles of 3,5-dimethylpyrazole are slowly charged, and reacted at 60 to 70 ° C. until the isocyanate content confirmed by the infrared spectrophotometer is zero, to give a dimethylpyrazole block polyisocyanate compound 98
- a clear, colorless, viscous liquid composition containing 7% by mass was obtained.
- the mixed solution was irradiated with ultrasonic waves to emulsify and disperse all monomers.
- 0.3 g of azobis (isobutylamidine) dihydrochloride is added to the mixed solution, and radical polymerization is performed at 60 ° C. for 6 hours under a nitrogen atmosphere to obtain a non-fluorinated acrylic polymer containing 20% by mass of a non-fluorinated acrylic polymer. A dispersion was obtained.
- Example 1 0.3% by mass of the amino-modified silicone dispersion obtained in Preparation Example A1, 3.7% by mass of the dispersion of the alkylpolysiloxane and silicone resin obtained in Preparation Example B4, nice-pol FE-26 (Antistatic Agent, manufactured by Nikka Chemical Co., Ltd. (trade name) 0.5% by mass, and Textport BG-290 (penetrant, manufactured by Nicha Chemical Co., Ltd., trade name) 0.5% by mass
- the polyester 100% cloth or the nylon 100% cloth which had been dyed was dipped in the treatment liquid diluted in the above (pickup rate 60% by mass) and then dried at 130 ° C. for 1 minute.
- Examples 2 to 11 Using the amino-modified silicone dispersion shown in Table 2 or 3 instead of the amino-modified silicone dispersion obtained in Preparation Example A1, the blending amount (% by mass) of the amino-modified silicone dispersion and the dispersion of the alkylpolysiloxane and the silicone resin Water-repellent fiber products were obtained in the same manner as in Example 1 except that the above was changed as shown in Table 2 or 3.
- Example 12 1.5 parts by mass of SF-8417 (trade name) manufactured by Toray Dow Corning Co., Ltd., 0.1 parts by mass of formic acid as amino-modified silicone, and MQ-1600 silicone resin (Toray Dow Corning Co., Ltd.) And 28.5 parts by mass of a 1: 1 mixture of dimethyl silicone and dimethyl silicone were mixed with 1 part by mass of a 5-mol ethylene oxide adduct of a branched alcohol having 12 to 14 carbon atoms.
- SF-8417 trade name
- formic acid amino-modified silicone
- MQ-1600 silicone resin Toray Dow Corning Co., Ltd.
- the dispersion obtained above was used in place of the dispersion of the amino-modified silicone dispersion obtained in Preparation Example A1 and the dispersion of the alkylpolysiloxane and silicone resin obtained in Preparation Example B4 to be 4% by mass.
- a water repellent fiber product was obtained in the same manner as Example 1 except for the above.
- Example 13 Using the amino-modified silicone dispersion shown in Table 4 instead of the amino-modified silicone dispersion obtained in Preparation Example A1, the blending amounts (% by mass) of the amino-modified silicone dispersion and the dispersion of the alkylpolysiloxane and the silicone resin in the same manner as in Example 1 except that the non-fluorinated acrylic polymer dispersion obtained in Preparation Example D1 is changed as shown in Table 4 and further added so as to be the blending amount (% by mass) shown in Table 4. Obtained a water repellent fiber product.
- Example 14 and 15 Dispersion of the amino-modified silicone dispersion shown in Table 4 in place of the dispersion of the amino-modified silicone dispersion obtained in Preparation Example A1 and the dispersion of the alkylpolysiloxane and silicone resin obtained in Preparation Example B4 and the dispersion of the alkyl polysiloxane and silicone resin Water-repellent fiber product in the same manner as in Example 1 except that the blending amounts (% by mass) of the amino-modified silicone dispersion and the dispersion of the alkylpolysiloxane and the silicone resin are changed as shown in Table 4 using I got
- Example 16 Using the amino-modified silicone dispersion shown in Table 5 instead of the amino-modified silicone dispersion obtained in Preparation Example A1, the compounding amounts (mass%) shown in Table 5 for the polyfunctional isocyanate compound dispersion obtained in Preparation Example C1 A water repellent fiber product was obtained in the same manner as in Example 1 except that it was further added so as to become.
- Example 17 A water-repellent fiber product was obtained in the same manner as in Example 16 except that the type of the polyfunctional isocyanate compound dispersion was changed as shown in Table 5.
- Example 19 The procedure of Example 16 is repeated except that the crosslinker of Preparation Example C4 and the catalyst of Preparation Example C5 are used in the compounding amounts (% by mass) shown in Table 5 instead of the polyfunctional isocyanate compound dispersion. Obtained a water repellent fiber product.
- Example 20 The blending amount (% by mass) of the polyfunctional isocyanate compound dispersion liquid is changed as shown in Table 5, and the crosslinking agent of Preparation Example C4 and the catalyst of Preparation Example C5 become the blending amounts (% by mass) shown in Table 5.
- a water repellent fiber product was obtained in the same manner as in Example 16 except that it was further added.
- Example 21 Water-repellent fiber products were obtained in the same manner as in Example 1, except that the amino-modified silicone dispersion obtained in Preparation Example A8 was used instead of the amino-modified silicone dispersion obtained in Preparation Example A1. .
- Example 22 0.3% by mass of the amino-modified silicone dispersion obtained in Preparation Example A8, 3.7% by mass of the dispersion of the alkylpolysiloxane and silicone resin obtained in Preparation Example B4, nice-pol FE-26 (Antistatic Agent, manufactured by Nikka Chemical Co., Ltd. (trade name) 0.5% by mass, and Textport BG-290 (penetrant, manufactured by Nicha Chemical Co., Ltd., trade name) 0.5% by mass
- the pH of the treatment solution was adjusted to 6 by adding acetic acid to the treatment solution diluted with.
- the treated solution was dipped in a dyed polyester 100% cloth or nylon 100% cloth (pickup rate: 60% by mass) and then dried at 130 ° C. for 1 minute. Further, a 100% polyester cloth was heat-treated at 180 ° C. for 30 seconds, and a 100% nylon cloth was heat-treated at 170 ° C. for 30 seconds to obtain a water repellent fiber product.
- Comparative example 4 A water-repellent fiber product was obtained in the same manner as in Comparative Example 1 except that the polyfunctional isocyanate compound dispersion was not used.
- Comparative example 5 A water-repellent fiber product was obtained in the same manner as Comparative Example 2 except that the polyfunctional isocyanate compound dispersion was not used.
- the water repellent fiber product was washed 10 times (L-10) according to JIS L 0217 (1995) method 103, and the water repellency after air drying was evaluated in the same manner as the water repellency evaluation method.
- seam slip evaluation of textiles The seam slip resistance force of the water repellent fiber product was measured according to JIS L 1096: 2010 8.23 slip resistance force 8.23.1 seam slip method b) B method. The smaller the value, the better the stitch slipperiness, and the case of 4 mm or less was judged to be good.
- water repellent fiber products treated with the water repellent compositions of Examples 1 to 22 are excellent in water repellency, durable water repellency and feeling, and also excellent in stitch slipperiness.
- a water repellent composition capable of obtaining a water repellent fiber product excellent in water repellency, durable water repellency and texture. Further, according to the water repellent composition of the present invention, excellent seam slipperiness can be imparted to textiles and the like.
Landscapes
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Organic Chemistry (AREA)
- Materials Engineering (AREA)
- Textile Engineering (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Life Sciences & Earth Sciences (AREA)
- Wood Science & Technology (AREA)
- Inorganic Chemistry (AREA)
- Combustion & Propulsion (AREA)
- Treatments For Attaching Organic Compounds To Fibrous Goods (AREA)
- Materials Applied To Surfaces To Minimize Adherence Of Mist Or Water (AREA)
Abstract
Description
アミノ変性シリコーンとしては、オルガノポリシロキサンの側鎖又は末端にアミノ基及び/又はイミノ基を含む有機基を有する化合物が挙げられる。このような有機基としては、例えば、-R-NH2で表される有機基、及び、-R-NH-R’-NH2で表される有機基が挙げられる。R及びR’としては、エチレン基、プロピレン基等の2価の基が挙げられる。アミノ基及び/又はイミノ基の一部又は全部が、封鎖されたアミノ基及び/又はイミノ基であってもよい。封鎖されたアミノ基及び/又はイミノ基は、例えば、アミノ基及び/又はイミノ基を封鎖剤で処理することにより得られる。封鎖剤としては、例えば、炭素数2~22の脂肪酸、炭素数2~22の脂肪酸の酸無水物、炭素数2~22の脂肪酸の酸ハライド、炭素数1~22の脂肪族モノイソシアネートなどが挙げられる。
本実施形態の撥水剤組成物は、上記アミノ変性シリコーン、上記(IV)成分、及び、上記(IV)成分と上記(V)成分との反応物以外のシリコーン化合物として、シリコーンレジンを含む。シリコーンレジンとしては、構成成分としてMQ、MDQ、MT、MTQ、MDT又はMDTQを含み、25℃にて固形状であり、三次元構造を有するオルガノポリシロキサンであることが好ましい。また、シリコーンレジンは、JIS K 6249:2003 13.硬さ試験に従ってタイプAデュロメータにより測定した硬さが20以上であることが好ましく、60以上であることがより好ましい。ここで、M、D、T及びQは、それぞれ(R’’)3SiO0.5単位、(R’’)2SiO単位、R’’SiO1.5単位及びSiO2単位を表す。R’’は、炭素数1~10の1価の脂肪族炭化水素基、又は、炭素数6~15の1価の芳香族炭化水素基を表す。
アルキルポリシロキサンは、鎖状オルガノポリシロキサンの側鎖及び末端が飽和炭化水素基である化合物、又は、環状オルガノポリシロキサンの側鎖が飽和炭化水素基である化合物である。アルキルポリシロキサンとしては、例えば、下記一般式(1)で表される化合物、下記一般式(2)で表される化合物などが挙げられる。
本実施形態の撥水剤組成物は、上記で説明した各成分以外に、多官能イソシアネート化合物、界面活性剤、消泡剤、有機酸、無機酸、アルコール、抗菌剤、防黴剤、pH調整剤、着色剤、シリカ、酸化防止剤、消臭剤、各種触媒、乳化安定剤、アルキルポリシロキサン以外の各種有機溶剤、キレート剤、帯電防止剤、アミノ変性シリコーン以外のオルガノ変性シリコーン、多官能イソシアネート化合物以外の架橋剤などをさらに含有していてもよい。
(調製例A1)
アミノ変性シリコーンとしてKF8005(信越化学工業(株)製、商品名)30質量部と、蟻酸0.3質量部と、炭素数12~14の分岐アルコールのエチレンオキサイド5モル付加物1質量部とを混合した。次いで、得られた混合物に、水68.7質量部を少量ずつ混合しながら添加し、アミノ変性シリコーンを30質量%含む分散液を得た。
アミノ変性シリコーンをKF8005から表1に記載のアミノ変性シリコーンに変えたこと以外は調製例A1と同様にして、アミノ変性シリコーンを30質量%含む分散液を得た。なお、KF-868、KF-864、KF-393は、信越化学工業(株)製の商品名であり、SF-8417、BY16-853Uは、東レ・ダウコーニング(株)製の商品名であり、XF42-B1989は、モメンティブ・パフォーマンス・マテリアルズ・ジャパン合同会社製の商品名である。
上記で用いたアミノ変性シリコーンの官能基当量(単位:g/mоl)、及び25℃における動粘度(単位:mm2/s)を表1に示す。
アミノ変性シリコーンとしてSF-8417(東レ・ダウコーニング(株)製、商品名)30質量部と、炭素数12~14の分岐アルコールのエチレンオキサイド5モル付加物1質量部とを混合した。次いで、得られた混合物に、水69.0質量部を少量ずつ混合しながら添加し、アミノ変性シリコーンを30質量%含む分散液を得た。
(調製例B1)
シリコーンレジンとしてMQ-1600(トリメチルシリル基含有ポリシロキサン、東レ・ダウコーニング(株)製、商品名)50質量部に、アルキルポリシロキサンとしてジメチルシリコーン(6cs、東レ・ダウコーニング(株)製)50質量部を添加し、シリコーンレジンが溶解するまで混合して、混合物を得た。得られた混合物30質量部と、炭素数12~14の分岐アルコールのエチレンオキサイド5モル付加物1質量部とを混合した。次いで、水69質量部を少量ずつ混合しながら添加し、アルキルポリシロキサンを15質量%とシリコーンレジンを15質量%含む分散液を得た。
MQ-1600とジメチルシリコーンとの混合物に代えてKF9021L(信越化学工業(株)製、商品名、溶媒:低粘度メチルポリシロキサン(KF-96L-2cs、ジメチルシリコーン)、トリメチルシリル基含有ポリシロキサン:溶媒(質量比)=50:50)を使用したこと以外は、調製例B1と同様にしてアルキルポリシロキサンを15質量%とシリコーンレジンを15質量%含む分散液を得た。
MQ-1600とジメチルシリコーンとの混合物に代えてKF7312L(信越化学工業(株)製、商品名、溶媒:低粘度メチルポリシロキサン(KF-96L-2cs、ジメチルシリコーン)、トリメチルシリル基含有ポリシロキサン:溶媒(質量比)=50:50)を使用したこと以外は、調製例B1と同様にしてアルキルポリシロキサンを15質量%とシリコーンレジンを15質量%含む分散液を得た。
シリコーンレジンとしてMQ-1600(トリメチルシリル基含有ポリシロキサン、東レ・ダウコーニング(株)製、商品名)25質量部に、アルキルポリシロキサンとしてジメチルシリコーン(6cs、東レ・ダウコーニング(株)製)75質量部を添加し、シリコーンレジンが溶解するまで混合して、混合物を得た。得られた混合物30質量部と、炭素数12~14の分岐アルコールのエチレンオキサイド5モル付加物1質量部とを混合した。次いで、水69質量部を少量ずつ混合しながら添加し、アルキルポリシロキサンを22.5質量%とシリコーンレジンを7.5質量%含む分散液を得た。
まず、トリメチロールプロパンとトルエンジイソシアネートとの反応生成物として、Polurene AD(トリメチロールプロパンとトルエンジイソシアネート(2,4異性体と2,6異性体との質量比は80:20)との反応生成物の含有量75質量%、溶剤:酢酸エチル、SAPICI社製、商品名)を用意した。
反応容器に、デュラネートTHA-100(ヘキサメチレンジイソシアネートのイソシアヌレートタイプ、NCO官能基数:3、含有量100質量%、旭化成ケミカルズ社製、商品名)を1モル、及びメチルイソブチルケトンを添加し、60~70℃まで加熱した。次いで、メチルエチルケトオキシム3モルをゆっくりと仕込み、60~70℃で赤外分光光度計にて確認されるイソシアネート含量がゼロになるまで反応させることにより、メチルエチルケトオキシムブロックポリイソシアネート化合物を98.7質量%含む無色透明の粘稠液状組成物を得た。
反応容器に、デュラネート24A-100(ヘキサメチレンジイソシアネートのビウレットタイプ、NCO官能基数:3、含有量100質量%、旭化成ケミカルズ社製、商品名)を1モル、及びメチルイソブチルケトンを添加し、60~70℃まで加熱した。次いで、3,5-ジメチルピラゾール3モルをゆっくりと仕込み、60~70℃で赤外分光光度計にて確認されるイソシアネート含量がゼロになるまで反応させることにより、ジメチルピラゾールブロックポリイソシアネート化合物を98.7質量%含む無色透明の粘稠液状組成物を得た。
(調製例C4)
ベッカミンM-3(トリメチロールアミン、DIC株式会社製、商品名)を準備した。
(調製例C5)
ベッカミンACX(有機アミン系触媒、DIC株式会社製、商品名)を準備した。
(調製例D1)
500mL耐圧フラスコに、アクリル酸ステアリル30g、アクリル酸ラウリル10g、クロロエチレン20g、3スチレン化フェノール30モルエチレンオキサイド付加物5g、アーカードT-28(ライオン株式会社製、商品名、塩化ステアリルトリメチルアンモニウム)3g、トリプロピレングリコール25g及び水206.7gを入れ、45℃にて混合攪拌し混合液とした。この混合液に超音波を照射して全単量体を乳化分散させた。次いで、アゾビス(イソブチルアミジン)二塩酸塩0.3gを混合液に添加し、窒素雰囲気下で60℃にて6時間ラジカル重合させて、非フッ素アクリル系ポリマーを20質量%含む非フッ素アクリル系ポリマー分散液を得た。
(調製例E1)
ジメチルシリコーン(25℃での動粘度が6cs、東レ・ダウコーニング(株)製)30質量部と、炭素数12~14の分岐アルコールのエチレンオキサイド5モル付加物2質量部とを混合した。次いで、水68質量部を少量ずつ混合しながら添加し、アルキルポリシロキサンを30質量%含む分散液を得た。
(実施例1)
調製例A1で得られたアミノ変性シリコーン分散液を0.3質量%、調製例B4で得られたアルキルポリシロキサンとシリコーンレジンの分散液を3.7質量%、ナイスポールFE-26(帯電防止剤、日華化学(株)製、商品名)を0.5質量%及びテキスポートBG-290(浸透剤、日華化学(株)製、商品名)を0.5質量%となるように水で希釈した処理液に、染色を行ったポリエステル100%布又はナイロン100%布を浸漬処理(ピックアップ率60質量%)した後、130℃で1分間乾燥した。更にポリエステル100%布においては180℃で30秒間熱処理をし、ナイロン100%布においては170℃で30秒間熱処理を行い、撥水性繊維製品を得た。なお、表中に、アミノ変性シリコーンの配合量とアルキルポリシロキサンの配合量との質量比を示す。
調製例A1で得られたアミノ変性シリコーン分散液に代えて表2又は3に示すアミノ変性シリコーン分散液を用い、アミノ変性シリコーン分散液及びアルキルポリシロキサンとシリコーンレジンの分散液の配合量(質量%)を表2又は3に示すように変えたこと以外は実施例1と同様にして、撥水性繊維製品をそれぞれ得た。
アミノ変性シリコーンとしてSF-8417(東レ・ダウコーニング(株)製、商品名)1.5質量部と、蟻酸0.1質量部と、シリコーンレジンであるMQ-1600(東レ・ダウコーニング(株)製、商品名)及びジメチルシリコーンの1:1混合物28.5質量部と、炭素数12~14の分岐アルコールのエチレンオキサイド5モル付加物1質量部とを混合した。次いで、得られた混合物に、水68.9質量部を少量ずつ混合しながら添加し、アミノ変性シリコーンとシリコーンレジンとアルキルポリシロキサンとを合計で30質量%含む分散液(アミノ変性シリコーン:アルキルポリシロキサン(質量比)=100:950)を得た。
調製例A1で得られたアミノ変性シリコーン分散液に代えて表4に示すアミノ変性シリコーン分散液を用い、アミノ変性シリコーン分散液及びアルキルポリシロキサンとシリコーンレジンの分散液の配合量(質量%)を表4に示すように変え、調製例D1で得られた非フッ素アクリル系ポリマー分散液を表4に示す配合量(質量%)となるように更に加えたこと以外は実施例1と同様にして、撥水性繊維製品を得た。
調製例A1で得られたアミノ変性シリコーン分散液及び調製例B4で得られたアルキルポリシロキサンとシリコーンレジンの分散液に代えて表4に示すアミノ変性シリコーン分散液及びアルキルポリシロキサンとシリコーンレジンの分散液を用い、アミノ変性シリコーン分散液及びアルキルポリシロキサンとシリコーンレジンの分散液の配合量(質量%)を表4に示すように変えたこと以外は実施例1と同様にして、撥水性繊維製品を得た。
調製例A1で得られたアミノ変性シリコーン分散液に代えて表5に示すアミノ変性シリコーン分散液を用い、調製例C1で得られた多官能イソシアネート化合物分散液を表5に示す配合量(質量%)となるように更に加えたこと以外は実施例1と同様にして、撥水性繊維製品を得た。
多官能イソシアネート化合物分散液の種類を表5に示すように変えたこと以外は実施例16と同様にして、撥水性繊維製品を得た。
多官能イソシアネート化合物分散液に代えて、調製例C4の架橋剤と調製例C5の触媒とを表5に示す配合量(質量%)となるように用いたこと以外は実施例16と同様にして、撥水性繊維製品を得た。
多官能イソシアネート化合物分散液の配合量(質量%)を表5に示すように変え、調製例C4の架橋剤と調製例C5の触媒とを表5に示す配合量(質量%)となるように更に加えたこと以外は実施例16と同様にして、撥水性繊維製品を得た。
調製例A1で得られたアミノ変性シリコーン分散液に代えて、調製例A8で得られたアミノ変性シリコーン分散液を用たこと以外は実施例1と同様にして、撥水性繊維製品をそれぞれ得た。
調製例A8で得られたアミノ変性シリコーン分散液を0.3質量%、調製例B4で得られたアルキルポリシロキサンとシリコーンレジンの分散液を3.7質量%、ナイスポールFE-26(帯電防止剤、日華化学(株)製、商品名)を0.5質量%及びテキスポートBG-290(浸透剤、日華化学(株)製、商品名)を0.5質量%となるように水で希釈した処理液に、酢酸を添加して処理液のpHを6に調整した。この処理液に染色を行ったポリエステル100%布又はナイロン100%布を浸漬処理(ピックアップ率60質量%)した後、130℃で1分間乾燥した。更にポリエステル100%布においては180℃で30秒間熱処理をし、ナイロン100%布においては170℃で30秒間熱処理を行い、撥水性繊維製品を得た。
アルキルポリシロキサンとシリコーンレジンの分散液を用いず、アミノ変性シリコーン分散液の種類及び配合量(質量%)を表7に示すように変え、調製例C1で得られた多官能イソシアネート化合物分散液を表7に示す配合量(質量%)となるように更に加えたこと以外は実施例1と同様にして、撥水性繊維製品を得た。
アミノ変性シリコーン分散液及びアルキルポリシロキサンとシリコーンレジンの分散液を用いず、調製例C1で得られた多官能イソシアネート化合物分散液、及び調製例D1で得られた非フッ素アクリル系ポリマー分散液を、表7に示す配合量(質量%)となるように更に加えたこと以外は実施例1と同様にして、撥水性繊維製品を得た。
アミノ変性シリコーン分散液を用いず、アルキルポリシロキサンとシリコーンレジンの分散液の種類及び配合量(質量%)を表7に示すように変え、調製例C1で得られた多官能イソシアネート化合物分散液を表7に示す配合量(質量%)となるように更に加えたこと以外は実施例1と同様にして、撥水性繊維製品を得た。
多官能イソシアネート化合物分散液を用いなかったこと以外は比較例1と同様にして、撥水性繊維製品を得た。
多官能イソシアネート化合物分散液を用いなかったこと以外は比較例2と同様にして、撥水性繊維製品を得た。
多官能イソシアネート化合物分散液を用いなかったこと以外は比較例3と同様にして、撥水性繊維製品を得た。
アミノ変性シリコーン分散液の種類及び配合量(質量%)を表8に示すように変え、アルキルポリシロキサンとシリコーンレジンの分散液に代えて調製例E1で得られたアルキルポリシロキサン分散液を表7に示す配合量(質量%)となるように用いたこと以外は実施例1と同様にして、撥水性繊維製品を得た。
アミノ変性シリコーン分散液及びアルキルポリシロキサン分散液の配合量(質量%)を表8に示すように変えたこと以外は比較例7と同様にして、撥水性繊維製品を得た。
調製例A1で得られたアミノ変性シリコーン分散液及び調製例B4で得られたアルキルポリシロキサンとシリコーンレジンの分散液に代えて表8に示すアミノ変性シリコーン分散液及びアルキルポリシロキサンとシリコーンレジンの分散液を用い、アミノ変性シリコーン分散液及びアルキルポリシロキサンとシリコーンレジンの分散液の配合量(質量%)を表8に示すように変えたこと以外は実施例1と同様にして、撥水性繊維製品を得た。
アミノ変性シリコーン分散液及びアルキルポリシロキサンとシリコーンレジンの分散液の配合量(質量%)を表8に示すように変えたこと以外は比較例9と同様にして、撥水性繊維製品を得た。
JIS L 1092(2009)のスプレー法に準じてシャワー水温を20℃として試験をした。結果は目視にて下記の等級で評価した。なお、特性がわずかに良好な場合は等級に「+」をつけ、特性が等級4と等級5との間である場合は、等級を「4-5」とした。
撥水性:状態
5:表面に付着湿潤のないもの
4:表面にわずかに付着湿潤を示すもの
3:表面に部分的湿潤を示すもの
2:表面に湿潤を示すもの
1:表面全体に湿潤を示すもの
0:表裏両面が完全に湿潤を示すもの
上記撥水性繊維製品に対して、JIS L 0217(1995)の103法による洗濯を10回(L-10)行い、風乾後の撥水性を上記撥水性評価方法と同様に評価した。
上記撥水性繊維製品をハンドリングにて下記に示す5段階で評価した。
1:硬い ~ 5:柔らかい
上記撥水性繊維製品の縫目滑脱抵抗力を、JIS L 1096:2010の8.23滑脱抵抗力 8.23.1縫目滑脱法 b)B法により測定した。数値が小さいほど縫目滑脱性に優れていることを示し、中でも4mm以下の場合を良好と判断した。
Claims (4)
- アミノ変性シリコーンと、シリコーンレジンと、アルキルポリシロキサンとを含有し、
前記アルキルポリシロキサンの配合量が、前記アミノ変性シリコーンの配合量100質量部に対して、500~15,000質量部である、撥水剤組成物。 - 前記アミノ変性シリコーンの官能基当量が、100~20,000g/molである、請求項1に記載の撥水剤組成物。
- 水酸基及び/又は炭素数1~3のアルコキシ基を有するジメチルポリシロキサンと、アミノ基含有シランカップリング剤と、シリコーンレジンと、アルキルポリシロキサンとを含有し、
前記アルキルポリシロキサンの配合量が、前記ジメチルポリシロキサン及び前記アミノ基含有シランカップリング剤の合計配合量100質量部に対して、500~15,000質量部である、撥水剤組成物。 - 繊維を、請求項1~3のいずれか一項に記載の撥水剤組成物が含まれる処理液で処理する工程を備える、撥水性繊維製品の製造方法。
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2019561615A JP7300395B2 (ja) | 2017-12-25 | 2018-12-20 | 撥水剤組成物、及び撥水性繊維製品の製造方法 |
| US16/956,696 US20200332148A1 (en) | 2017-12-25 | 2018-12-20 | Water repellent agent composition, and production method of water repellent fiber product |
| EP18895048.9A EP3733809A4 (en) | 2017-12-25 | 2018-12-20 | WATER-REPELLENT COMPOSITION AND METHOD FOR MANUFACTURING A WATER-REPELLENT FIBER PRODUCT |
| KR1020207020754A KR20200100135A (ko) | 2017-12-25 | 2018-12-20 | 발수제 조성물, 및 발수성 섬유 제품의 제조방법 |
| CN201880076237.4A CN111386327B (zh) | 2017-12-25 | 2018-12-20 | 防水剂组合物和防水性纤维制品的制造方法 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2017247815 | 2017-12-25 | ||
| JP2017-247815 | 2017-12-25 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2019131456A1 true WO2019131456A1 (ja) | 2019-07-04 |
Family
ID=67067381
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2018/047085 WO2019131456A1 (ja) | 2017-12-25 | 2018-12-20 | 撥水剤組成物、及び撥水性繊維製品の製造方法 |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US20200332148A1 (ja) |
| EP (1) | EP3733809A4 (ja) |
| JP (1) | JP7300395B2 (ja) |
| KR (1) | KR20200100135A (ja) |
| CN (1) | CN111386327B (ja) |
| TW (1) | TWI812664B (ja) |
| WO (1) | WO2019131456A1 (ja) |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2019173185A (ja) * | 2018-03-27 | 2019-10-10 | 松本油脂製薬株式会社 | 繊維用撥水剤組成物及びその利用 |
| JP7153407B1 (ja) | 2022-01-17 | 2022-10-14 | 竹本油脂株式会社 | ポリエステル合成繊維用処理剤、ポリエステル合成繊維用処理剤含有組成物、及びポリエステル合成繊維 |
| JP7223467B1 (ja) | 2022-01-17 | 2023-02-16 | 竹本油脂株式会社 | ポリエステル合成繊維用処理剤セット、ポリエステル合成繊維用処理剤含有組成物の製造方法、ポリエステル合成繊維用第1処理剤、ポリエステル合成繊維用第2処理剤、ポリエステル合成繊維用第3処理剤、ポリエステル合成繊維用第1処理剤含有組成物、ポリエステル合成繊維用第2処理剤含有組成物、ポリエステル合成繊維の処理方法、及びポリエステル合成繊維の製造方法 |
| WO2024034530A1 (ja) * | 2022-08-09 | 2024-02-15 | 信越化学工業株式会社 | 撥水剤組成物及び繊維処理剤 |
Families Citing this family (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN112062498B (zh) * | 2020-09-11 | 2022-07-05 | 杭州埃比森新材料有限公司 | 一种自乳化型有机硅粉体憎水剂及其制备方法 |
| CN112746365A (zh) * | 2020-12-25 | 2021-05-04 | 江苏锐晟纺织科技有限公司 | 一种高疏水性纯棉超柔华夫格面料的生产工艺及华夫格面料 |
| EP4291611A1 (en) | 2021-02-15 | 2023-12-20 | Dow Silicones Corporation | Aqueous emulsion including interpenetrating network of silicon resin and organic polymers |
| EP4050058A1 (de) | 2021-02-26 | 2022-08-31 | Rudolf GmbH | Effektpermanentes hydrophobierungsmittel |
| CN117480297A (zh) | 2021-06-15 | 2024-01-30 | 美国陶氏有机硅公司 | 用于纺织品的基于聚硅氧烷的防水剂 |
| CN114960199B (zh) * | 2021-07-07 | 2023-11-03 | 上海允继化工新材料有限公司 | 一种纺织品用无氟防水剂及其制备方法 |
| WO2023086692A1 (en) | 2021-11-10 | 2023-05-19 | Dow Silicones Corporation | Textile treatment emulsion formulation including a silicone - (meth)acrylate copolymer and a silicone additive |
| CN114855462B (zh) * | 2022-06-10 | 2023-02-07 | 杭州正立生物科技有限公司 | 水基型有机硅树脂组合物以及制备方法 |
| CN115029042A (zh) * | 2022-07-02 | 2022-09-09 | 成都春天来了建材有限公司 | 一种多彩真石漆及其制备方法 |
| KR102526375B1 (ko) * | 2022-08-23 | 2023-04-26 | 이동철 | 우수한 내오염성을 갖는 1액형 유성 발수제 조성물 및 이의 제조 방법 |
| KR102534952B1 (ko) * | 2022-08-23 | 2023-05-26 | 이동철 | 우수한 내오염성을 갖는 1액형 수성 발수제 조성물 및 이의 제조 방법 |
| CN116478620A (zh) * | 2023-05-04 | 2023-07-25 | 浙江新安化工集团股份有限公司 | 一种有机硅防水剂及其制备方法 |
| CN116641229B (zh) * | 2023-06-06 | 2023-11-03 | 江苏瑞洋安泰新材料科技有限公司 | 一种导电超疏水织物及其制备方法 |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS57149563A (en) * | 1981-02-10 | 1982-09-16 | Wacker Chemie Gmbh | Impregnation of fiber web |
| JP2000336580A (ja) * | 1999-05-28 | 2000-12-05 | Soft 99 Corporation:Kk | 撥水性付与クロス用処理剤及び撥水性付与クロス |
| JP2004059609A (ja) | 2002-07-25 | 2004-02-26 | Miki Riken Kogyo Kk | 柔軟撥水剤 |
| JP2006328624A (ja) | 2005-04-28 | 2006-12-07 | Nicca Chemical Co Ltd | 撥水剤、撥水加工方法及び撥水性繊維製品 |
| JP2010526891A (ja) * | 2007-04-26 | 2010-08-05 | ダウ・コーニング・コーポレイション | 撥水性を与える水性シリコーンエマルジョン |
Family Cites Families (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP3748054B2 (ja) * | 2001-06-28 | 2006-02-22 | 信越化学工業株式会社 | 室温硬化性オルガノポリシロキサン組成物 |
| FR2840912B1 (fr) * | 2002-06-18 | 2005-10-21 | Rhodia Chimie Sa | Emulsion silicone aqueuse pour le revetement de supports fibreux tisses ou non |
| EP2980647B1 (en) * | 2013-03-28 | 2018-05-09 | Toyobo Co., Ltd. | Method for producing flexographic printing plate |
-
2018
- 2018-12-20 CN CN201880076237.4A patent/CN111386327B/zh active Active
- 2018-12-20 US US16/956,696 patent/US20200332148A1/en active Pending
- 2018-12-20 WO PCT/JP2018/047085 patent/WO2019131456A1/ja unknown
- 2018-12-20 JP JP2019561615A patent/JP7300395B2/ja active Active
- 2018-12-20 EP EP18895048.9A patent/EP3733809A4/en active Pending
- 2018-12-20 KR KR1020207020754A patent/KR20200100135A/ko not_active Application Discontinuation
- 2018-12-25 TW TW107146878A patent/TWI812664B/zh active
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS57149563A (en) * | 1981-02-10 | 1982-09-16 | Wacker Chemie Gmbh | Impregnation of fiber web |
| JP2000336580A (ja) * | 1999-05-28 | 2000-12-05 | Soft 99 Corporation:Kk | 撥水性付与クロス用処理剤及び撥水性付与クロス |
| JP2004059609A (ja) | 2002-07-25 | 2004-02-26 | Miki Riken Kogyo Kk | 柔軟撥水剤 |
| JP2006328624A (ja) | 2005-04-28 | 2006-12-07 | Nicca Chemical Co Ltd | 撥水剤、撥水加工方法及び撥水性繊維製品 |
| JP2010526891A (ja) * | 2007-04-26 | 2010-08-05 | ダウ・コーニング・コーポレイション | 撥水性を与える水性シリコーンエマルジョン |
Cited By (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2019173185A (ja) * | 2018-03-27 | 2019-10-10 | 松本油脂製薬株式会社 | 繊維用撥水剤組成物及びその利用 |
| JP7146426B2 (ja) | 2018-03-27 | 2022-10-04 | 松本油脂製薬株式会社 | 繊維用撥水剤組成物及びその利用 |
| JP7153407B1 (ja) | 2022-01-17 | 2022-10-14 | 竹本油脂株式会社 | ポリエステル合成繊維用処理剤、ポリエステル合成繊維用処理剤含有組成物、及びポリエステル合成繊維 |
| JP7223467B1 (ja) | 2022-01-17 | 2023-02-16 | 竹本油脂株式会社 | ポリエステル合成繊維用処理剤セット、ポリエステル合成繊維用処理剤含有組成物の製造方法、ポリエステル合成繊維用第1処理剤、ポリエステル合成繊維用第2処理剤、ポリエステル合成繊維用第3処理剤、ポリエステル合成繊維用第1処理剤含有組成物、ポリエステル合成繊維用第2処理剤含有組成物、ポリエステル合成繊維の処理方法、及びポリエステル合成繊維の製造方法 |
| WO2023136317A1 (ja) * | 2022-01-17 | 2023-07-20 | 竹本油脂株式会社 | ポリエステル合成繊維用処理剤、ポリエステル合成繊維用処理剤含有組成物、及びポリエステル合成繊維 |
| JP2023104858A (ja) * | 2022-01-17 | 2023-07-28 | 竹本油脂株式会社 | ポリエステル合成繊維用処理剤セット、ポリエステル合成繊維用処理剤含有組成物の製造方法、ポリエステル合成繊維用第1処理剤、ポリエステル合成繊維用第2処理剤、ポリエステル合成繊維用第3処理剤、ポリエステル合成繊維用第1処理剤含有組成物、ポリエステル合成繊維用第2処理剤含有組成物、ポリエステル合成繊維の処理方法、及びポリエステル合成繊維の製造方法 |
| JP2023104289A (ja) * | 2022-01-17 | 2023-07-28 | 竹本油脂株式会社 | ポリエステル合成繊維用処理剤、ポリエステル合成繊維用処理剤含有組成物、及びポリエステル合成繊維 |
| WO2024034530A1 (ja) * | 2022-08-09 | 2024-02-15 | 信越化学工業株式会社 | 撥水剤組成物及び繊維処理剤 |
Also Published As
| Publication number | Publication date |
|---|---|
| CN111386327A (zh) | 2020-07-07 |
| JP7300395B2 (ja) | 2023-06-29 |
| TWI812664B (zh) | 2023-08-21 |
| JPWO2019131456A1 (ja) | 2020-12-24 |
| EP3733809A4 (en) | 2021-09-22 |
| TW201930544A (zh) | 2019-08-01 |
| CN111386327B (zh) | 2023-04-18 |
| EP3733809A1 (en) | 2020-11-04 |
| KR20200100135A (ko) | 2020-08-25 |
| US20200332148A1 (en) | 2020-10-22 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2019131456A1 (ja) | 撥水剤組成物、及び撥水性繊維製品の製造方法 | |
| KR102351981B1 (ko) | 발수제 조성물 및 발수성 섬유 제품의 제조 방법 | |
| JP6435320B2 (ja) | 基材の表面特性を改質する組成物のためのウレタン性ポリマーエクステンダー | |
| KR102252767B1 (ko) | 불소화되지 않은 우레탄계 코팅 | |
| JP2017222827A (ja) | 撥水剤組成物、撥水性繊維製品及び撥水性繊維製品の製造方法 | |
| JP2018119250A (ja) | 撥水剤組成物、撥水性繊維製品及び撥水性繊維製品の製造方法 | |
| JP2023085382A (ja) | 撥水剤組成物、撥水性繊維製品及び撥水性繊維製品の製造方法 | |
| JP7469845B2 (ja) | 繊維用撥水剤組成物、撥水性繊維製品及び撥水性繊維製品の製造方法 | |
| CN113227486B (zh) | 拒水剂组合物、及拒水性纤维制品的制造方法 | |
| KR102667486B1 (ko) | 발수제 조성물 및 발수성 섬유 제품의 제조 방법 | |
| TWI835952B (zh) | 撥水劑組合物、及撥水性纖維製品之製造方法 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 18895048 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2019561615 Country of ref document: JP Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 20207020754 Country of ref document: KR Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 2018895048 Country of ref document: EP Effective date: 20200727 |