WO2018124227A1 - 被膜の製造方法 - Google Patents
被膜の製造方法 Download PDFInfo
- Publication number
- WO2018124227A1 WO2018124227A1 PCT/JP2017/047079 JP2017047079W WO2018124227A1 WO 2018124227 A1 WO2018124227 A1 WO 2018124227A1 JP 2017047079 W JP2017047079 W JP 2017047079W WO 2018124227 A1 WO2018124227 A1 WO 2018124227A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- film
- component
- coating
- composition
- skin
- Prior art date
Links
Images
Classifications
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B05—SPRAYING OR ATOMISING IN GENERAL; APPLYING FLUENT MATERIALS TO SURFACES, IN GENERAL
- B05B—SPRAYING APPARATUS; ATOMISING APPARATUS; NOZZLES
- B05B5/00—Electrostatic spraying apparatus; Spraying apparatus with means for charging the spray electrically; Apparatus for spraying liquids or other fluent materials by other electric means
- B05B5/16—Arrangements for supplying liquids or other fluent material
- B05B5/1691—Apparatus to be carried on or by a person or with a container fixed to the discharge device
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/72—Cosmetics or similar toiletry preparations characterised by the composition containing organic macromolecular compounds
- A61K8/81—Cosmetics or similar toiletry preparations characterised by the composition containing organic macromolecular compounds obtained by reactions involving only carbon-to-carbon unsaturated bonds
- A61K8/8141—Compositions of homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by only one carboxyl radical, or of salts, anhydrides, esters, amides, imides or nitriles thereof; Compositions of derivatives of such polymers
- A61K8/8158—Homopolymers or copolymers of amides or imides, e.g. (meth) acrylamide; Compositions of derivatives of such polymers
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/02—Cosmetics or similar toiletry preparations characterised by special physical form
- A61K8/0216—Solid or semisolid forms
- A61K8/022—Powders; Compacted Powders
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/02—Cosmetics or similar toiletry preparations characterised by special physical form
- A61K8/0216—Solid or semisolid forms
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/02—Cosmetics or similar toiletry preparations characterised by special physical form
- A61K8/0241—Containing particulates characterized by their shape and/or structure
- A61K8/027—Fibers; Fibrils
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/19—Cosmetics or similar toiletry preparations characterised by the composition containing inorganic ingredients
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/31—Hydrocarbons
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/33—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing oxygen
- A61K8/34—Alcohols
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/33—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing oxygen
- A61K8/34—Alcohols
- A61K8/342—Alcohols having more than seven atoms in an unbroken chain
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/33—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing oxygen
- A61K8/34—Alcohols
- A61K8/345—Alcohols containing more than one hydroxy group
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/33—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing oxygen
- A61K8/35—Ketones, e.g. benzophenone
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/33—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing oxygen
- A61K8/37—Esters of carboxylic acids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/72—Cosmetics or similar toiletry preparations characterised by the composition containing organic macromolecular compounds
- A61K8/81—Cosmetics or similar toiletry preparations characterised by the composition containing organic macromolecular compounds obtained by reactions involving only carbon-to-carbon unsaturated bonds
- A61K8/8129—Compositions of homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by an alcohol, ether, aldehydo, ketonic, acetal or ketal radical; Compositions of hydrolysed polymers or esters of unsaturated alcohols with saturated carboxylic acids; Compositions of derivatives of such polymers, e.g. polyvinylmethylether
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/72—Cosmetics or similar toiletry preparations characterised by the composition containing organic macromolecular compounds
- A61K8/84—Cosmetics or similar toiletry preparations characterised by the composition containing organic macromolecular compounds obtained by reactions otherwise than those involving only carbon-carbon unsaturated bonds
- A61K8/89—Polysiloxanes
- A61K8/891—Polysiloxanes saturated, e.g. dimethicone, phenyl trimethicone, C24-C28 methicone or stearyl dimethicone
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q19/00—Preparations for care of the skin
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B05—SPRAYING OR ATOMISING IN GENERAL; APPLYING FLUENT MATERIALS TO SURFACES, IN GENERAL
- B05D—PROCESSES FOR APPLYING FLUENT MATERIALS TO SURFACES, IN GENERAL
- B05D1/00—Processes for applying liquids or other fluent materials
- B05D1/02—Processes for applying liquids or other fluent materials performed by spraying
- B05D1/04—Processes for applying liquids or other fluent materials performed by spraying involving the use of an electrostatic field
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K2800/00—Properties of cosmetic compositions or active ingredients thereof or formulation aids used therein and process related aspects
- A61K2800/20—Chemical, physico-chemical or functional or structural properties of the composition as a whole
- A61K2800/26—Optical properties
- A61K2800/262—Transparent; Translucent
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K2800/00—Properties of cosmetic compositions or active ingredients thereof or formulation aids used therein and process related aspects
- A61K2800/80—Process related aspects concerning the preparation of the cosmetic composition or the storage or application thereof
- A61K2800/83—Electrophoresis; Electrodes; Electrolytic phenomena
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K2800/00—Properties of cosmetic compositions or active ingredients thereof or formulation aids used therein and process related aspects
- A61K2800/80—Process related aspects concerning the preparation of the cosmetic composition or the storage or application thereof
- A61K2800/87—Application Devices; Containers; Packaging
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M35/00—Devices for applying media, e.g. remedies, on the human body
- A61M35/003—Portable hand-held applicators having means for dispensing or spreading integral media
Definitions
- the present invention relates to a method for producing a coating.
- Patent Document 1 describes a cosmetic sheet in which cosmetics or cosmetic ingredients are held.
- Patent Documents 2 and 3 describe a method of forming a film by electrostatic spraying.
- the present invention is a method for producing a film, which forms a film on the surface of a film formation target.
- the method for producing a coating includes an electrostatic spraying step in which a composition is directly sprayed on the coating formation target to form a coating composed of fiber deposits, and the composition comprises the following components (a ), Component (b) and component (c).
- (A) One or more volatile substances selected from the group consisting of water, alcohol and ketone.
- B A polymer having a film-forming ability.
- C The liquid agent containing 1 type, or 2 or more types selected from a liquid oil and a polyol at 20 degreeC.
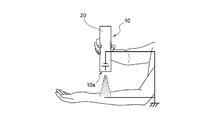
- FIG. 1 is a schematic view showing the configuration of an electrostatic spray apparatus suitably used in the present invention.
- FIG. 2 is a schematic diagram illustrating a state where an electrostatic spray method is performed using an electrostatic spray apparatus.
- FIG. 3 is an SEM image of the coating film of Example 3.
- FIG. 4 is an SEM image of the coating film of Comparative Example 1.
- the cosmetic sheet described in Patent Document 1 holds cosmetics or cosmetic ingredients, the cosmetics and the like can be used effectively on the skin.
- the cosmetic sheet described in Patent Document 1 contains cosmetics or the like in a sheet prepared in advance, it easily peels off from the sheet when frictional force is applied during wearing.
- the fiber of the sheet prepared in advance in addition to cosmetics etc., when stored in a state containing non-volatile oil or polyol such as almond oil, avocado oil, olive oil or the like, the fiber form of the constituent fibers of the sheet is The collapsed sheet form tends to collapse, and the storage stability deteriorates.
- the skin treatment method by electrostatic spray described in Patent Document 2 is a method of treating skin by electrostatic application of particles that are particulate powder substances. Therefore, since the film is not a fiber deposit, it is difficult to maintain the form of a single film, and the durability is inferior, for example, particles are partially dropped during use, and it is difficult to peel off after use.
- Patent Document 3 the method of forming a coating by electrostatic spray described in Patent Document 3 can be handled as a single film because the formed coating is a fiber deposit, and is easy to peel off after use.
- the adhesion between the film formed by electrostatic spraying and the substrate is not sufficient, and the film may be damaged or peeled due to external force such as friction.
- Patent Document 3 does not describe anything about transparentizing a coating made of fiber deposits and covering the skin in a natural state.
- the present invention relates to a method for producing a coating that can eliminate the above-mentioned drawbacks of the prior art.
- the production method of the present invention is a method for producing a film that forms a film on the surface of a film formation target.
- a composition containing a predetermined component is directly applied to the surface of human skin, which is an example of a film formation target, to form a film.
- an electrostatic spray method is employed as a method for forming a coating film.
- the electrostatic spray method is a method in which a positive or negative high voltage is applied to the composition to charge the composition, and the charged composition is sprayed toward the film formation target.
- the sprayed composition spreads into the space while being repeatedly refined by Coulomb repulsion, and in the process or after adhering to the film formation target, the solvent, which is a volatile substance, is dried, so that the film formation target A film is formed on the surface.
- composition used in the present invention is liquid in an environment (for example, 20 ° C.) in which an electrostatic spray method is performed. .
- This composition contains the following component (a), component (b), and component (c).
- component (a) One or more volatile substances selected from water, alcohol and ketone.
- component (B) A polymer having a film-forming ability.
- C) The liquid agent containing 1 type, or 2 or more types selected from a liquid oil and a polyol at 20 degreeC.
- composition for spraying is used in a method for producing a film that forms a transparent or translucent film made of a deposit containing fibers on human skin, which is an example of a film forming object, by electrostatic spraying.
- a method for producing a film that forms a transparent or translucent film made of a deposit containing fibers on human skin which is an example of a film forming object, by electrostatic spraying.
- each composition will be described.
- the volatile substance of component (a) is a substance that is volatile in the liquid state.
- the component (a) is sufficiently charged to the spray composition placed in an electric field, and then discharged from the tip of the nozzle toward the film formation target, for example, the skin, so that the component (a) ) Evaporates, the charge density of the spray composition becomes excessive, and the component (a) further evaporates while being further refined by Coulomb repulsion, and is finally formulated to form a dry film.
- the vapor pressure of the volatile substance is preferably 0.01 kPa or more and 106.66 kPa or less, more preferably 0.13 kPa or more and 66.66 kPa or less at 20 ° C. It is still more preferably 67 kPa or more and 40.00 kPa or less, and further preferably 1.33 kPa or more and 40.00 kPa or less.
- a monovalent chain aliphatic alcohol for example, a monovalent chain aliphatic alcohol, a monovalent cyclic aliphatic alcohol, or a monovalent aromatic alcohol is preferably used.
- a monovalent chain aliphatic alcohol for example, a monovalent chain aliphatic alcohol, a monovalent cyclic aliphatic alcohol, or a monovalent aromatic alcohol is preferably used.
- Specific examples thereof include ethanol, isopropyl alcohol, butyl alcohol, phenylethyl alcohol, propanol, pentanol and the like. These alcohols can use 1 type, or 2 or more types chosen from these.
- ketone examples include acetone, methyl ethyl ketone, and methyl isobutyl ketone. These ketones can be used alone or in combination of two or more.
- the volatile substance of component (a) is more preferably one or more selected from ethanol, isopropyl alcohol, butyl alcohol, and water, more preferably one or two selected from ethanol and butyl alcohol. More than species, and most preferably ethanol.
- the volatile substance of component (a) is selected from (a1) ethanol, isopropyl alcohol, and butyl alcohol from the viewpoint of dispersibility of component (b) when component (b) described later is a water-insoluble polymer. From the viewpoint of charge imparting, (a2) a mixed solution with water is preferable.
- the mass ratio (a2) / (a1) between the component (a2) and the component (a1) is 0.0025 or more and 0.3 or less from the viewpoint of the fiber formability and the coating adhesion by the spray composition. Is preferred.
- the composition for spraying contains a polymer having a film-forming ability as the component (b) together with the component (a).
- the component (b), which has a film-forming ability is generally a substance that can be dissolved in the volatile substance of the component (a).
- dissolving means being in a dispersed state at 20 ° C., and means that the dispersed state is a visually uniform state, preferably a visually transparent or translucent state.
- polymers having a film-forming ability are roughly classified into water-soluble polymers and water-insoluble polymers.
- water-soluble polymer means that 1 g of a polymer is weighed in an environment of 1 atm and 23 ° C. and then immersed in 10 g of ion-exchanged water. The thing which has the property which 5g or more melt
- water-insoluble polymer means that 1 g of a polymer is weighed in an environment of 1 atm and 23 ° C. and then immersed in 10 g of ion-exchanged water. It has the property that more than 5g does not dissolve.
- the polymer having a film forming ability preferably includes a water-insoluble polymer.
- water-soluble polymer having a film-forming ability examples include, for example, pullulan, hyaluronic acid, chondroitin sulfate, poly- ⁇ -glutamic acid, modified corn starch, ⁇ -glucan, gluco-oligosaccharide, heparin, keratosulfuric mucopolysaccharide, cellulose, pectin, etc.
- Xylan lignin, glucomannan, galacturonic acid, psyllium seed gum, tamarind seed gum, gum arabic, tragacanth gum, soybean water-soluble polysaccharides, alginic acid, carrageenan, laminaran, agar (agarose), fucoidan, methylcellulose, hydroxypropylcellulose, hydroxypropyl Natural polymers such as methylcellulose, partially saponified polyvinyl alcohol (when not used in combination with a crosslinking agent), low saponified polyvinyl alcohol, polyvinyl pyrrolidone (PVP), polyethylene Side, and synthetic polymers such as sodium polyacrylate and the like.
- PVP polyvinyl pyrrolidone
- water-soluble polymers can be used alone or in combination of two or more.
- polyethylene oxide is used as the water-soluble polymer, the number average molecular weight is preferably 50,000 or more and 3 million or less, and more preferably 100,000 or more and 2.5 million or less.
- examples of the water-insoluble polymer having film-forming ability include, for example, fully saponified polyvinyl alcohol that can be insolubilized after film formation, partially saponified polyvinyl alcohol that can be crosslinked after film formation by using in combination with a crosslinking agent, and poly (N-propylene).
- Noylethyleneimine graft-dimethylsiloxane / ⁇ -aminopropylmethylsiloxane copolymer and other oxazoline-modified silicones, polyvinyl acetal diethylaminoacetate, zein (a major component of corn protein), polyester, polylactic acid (PLA), polyacrylonitrile resin, Acrylic resin such as polymethacrylic acid resin, polystyrene resin, polyvinyl butyral resin, polyethylene terephthalate resin, polybutylene terephthalate resin, polyurethane resin, polyamide resin , Polyimide resins, polyamide-imide resins.
- water-insoluble polymers can be used alone (one kind) or in combination of two or more kinds.
- water-insoluble polymers fully saponified polyvinyl alcohol that can be insolubilized after film formation, partially saponified polyvinyl alcohol that can be cross-linked after film formation by using in combination with a crosslinking agent, polyvinyl butyral resin, (alkyl acrylate / octylamide)
- an acrylic resin such as a polymer, an oxazoline-modified silicone such as a poly (N-propanoylethyleneimine) graft-dimethylsiloxane / ⁇ -aminopropylmethylsiloxane copolymer, a polyester, or a tane.
- the composition for spraying contains, together with the component (a) and the component (b), a liquid agent containing one or more selected from oil and polyol liquid at 20 ° C. which is the component (c).
- Component (c) is preferably non-volatile in the liquid state.
- the component (c) is generally a substance that can be dissolved in the volatile substance of the component (a), like the polymer of the component (b).
- “dissolved” means that the polymer is in a dispersed state at 20 ° C., and the dispersed state is a visually uniform state, preferably a visually transparent or translucent state.
- oil that is liquid at 20 ° C. As the oil that is liquid at 20 ° C. as the component (c), a suitable oil is used according to the nature of the volatile substance of the component (a).
- the oil include hydrocarbon oils that are liquid at 20 ° C., ester oils, silicone oils, and higher alcohols. Liquid oils selected from these may be used alone or in combination of two or more.
- oil that is liquid at 20 ° C. is also referred to as “liquid oil”.
- the ester oil includes, in addition to oils such as triacylglyceride (triglycerin fatty acid ester) contained in vegetable oil, compounds having an ester structure with an HLB value of 10 or less.
- the HLB value is an index indicating a hydrophilic-lipophilic balance.
- the component (c) preferably contains a liquid oil at 20 ° C. from the viewpoint of further improving the adhesion of the polymer of the component (b) to the film-forming target, and has a polarity, and the component (b) From the viewpoint of improving the adhesion of the polymer to the coating object, it is preferable to contain one or more selected from ester oils and higher alcohols, and one or two selected from ester oils. It is preferable to contain seeds or more.
- Examples of the hydrocarbon oil that is liquid at 20 ° C. described above as the component (c) include liquid paraffin, squalane, squalene, n-octane, n-heptane, cyclohexane, light isoparaffin, liquid isoparaffin, and the like. From the viewpoint, liquid paraffin and squalane are preferable. Further, from the viewpoint of bringing the electrostatically sprayed film into close contact with the skin, the viscosity of the hydrocarbon oil at 30 ° C. is preferably 10 mPa ⁇ s or more, and more preferably 30 mPa ⁇ s or more.
- the content in the spray composition of isododecane, isohexadecane, and hydrogenated polyisobutene having a viscosity of less than 10 mPa ⁇ s at 30 ° C. is preferably 10% by mass or less, more preferably 5% by mass or less. More preferably, it is 1 mass% or less, More preferably, it is 0.5 mass% or less, and does not need to contain.
- the viscosity is measured at 30 ° C. with a BM viscometer (manufactured by Tokimec, measurement conditions: rotor No. 1, 60 rpm, 1 minute).
- a component (c) is a liquid at 20 degreeC
- the upper limit of the viscosity in 30 degreeC should just be a fluid range, Preferably it is 2000 mPa * s or less.
- glycerol fatty acid ester monoglycerol fatty acid ester, diglycerol fatty acid ester, and triglycerol fatty acid ester are contained.
- alkyl Selected from alkyl, isononyl isononanoate, tri (caprylic acid / capric acid) glycerin, and triacyl glyceride having one or more selected from oleic acid, eicosenoic acid, icosenoic acid and docosenoic acid
- tri (caprylic acid / capric acid) glycerin Selected from alkyl, isononyl isononanoate, tri (caprylic acid / capric acid) glycerin, and triacyl glyceride having one or more selected from oleic acid, eicosenoic acid, icosenoic acid and docosenoic acid
- oleic acid eicosenoic acid, icosenoic acid and docosenoic acid
- vegetable oils such as olive oil, jojoba oil, macadamia nut oil, medfoam oil, castor oil, safflower oil, sunflower oil, avocado oil, canola oil, kyonin oil, rice germ oil, rice bran oil, etc. Animal oils containing lanolin and the like can also be used.
- the polyglyceryl fatty acid ester contained in the ester oil that is liquid at 20 ° C. described above as the component (c) has an HLB value of 10 or less, polyglyceryl isostearate, polyglyceryl diisostearate, polyglyceryl triisostearate, polyglyceryl stearate, Examples thereof include polyglyceryl oleate and polyglyceryl sesquicaprate.
- sorbitan fatty acid ester sorbitan monostearate, sorbitan monooleate, sorbitan sesquioleate, sorbitan sesquiisostearate, sorbitan monopalmitate, sorbitan tristearate, sorbitan tristearate having an HLB value of 10 or less.
- coconut oil fatty acid sorbitan sorbitan.
- polyglyceryl isostearate polyglyceryl diisostearate, polyglyceryl triisostearate, polyglyceryl stearate, oleic acid from the viewpoint of adhering the electrostatic sprayed film to the skin and improving the feel when applied to the skin
- polyglyceryl and polyglyceryl sesquicaprate are preferable, and polyglyceryl diisostearate is more preferable.
- the content of the silicone oil in the spray composition is preferably 10% by mass or less, more preferably 7% by mass or less, from the viewpoint of improving adhesion to the skin or the like. Preferably, it is 0.1 mass% or more, More preferably, it is 1 mass% or more.
- the silicone oil may not be contained in the spray composition of the present invention.
- the content is 5% by mass or less, more preferably 1% by mass or less, and still more preferably. It is 0.1 mass% or less.
- the kinematic viscosity of the silicone oil at 25 ° C. is preferably 3 mm 2 / s or more, more preferably 4 mm 2 / s or more, and further preferably 5 mm from the viewpoint of bringing the electrostatically sprayed film into close contact with the skin or the like. 2 / s or more, preferably 30 mm 2 / s or less, more preferably 20 mm 2 / s or less, and further preferably 10 mm 2 / s or less.
- a silicone oil contains a dimethylpolysiloxane from a viewpoint of adhering the electrostatic sprayed film.
- Examples of the higher alcohol liquid at 20 ° C. described above as the component (c) include liquid higher alcohols having 12 to 20 carbon atoms, preferably higher alcohols of branched fatty acids or unsaturated fatty acids, isostearyl alcohol and oleyl alcohol. More preferred.
- the component (c) is a polyol
- the polyol includes alkylene glycol such as ethylene glycol, propylene glycol, 1,3-propanediol, 1,3-butanediol; diethylene glycol, dipropylene glycol, polyethylene glycol having a number average molecular weight of 1000 or less, polypropylene Polyalkylene glycol such as glycol; glycerin such as glycerin, diglycerin, triglycerin or polyglyceryl.
- ethylene glycol, propylene glycol, 1,3-butanediol, dipropylene glycol, polyethylene glycol, glycerin, and diglycerin are preferable from the viewpoint of improving the feeling of use, and propylene glycol, 1,3-butanediol, glycerin.
- Dipropylene glycol is more preferable.
- Polyethylene glycol has a number average molecular weight of preferably 600 or less, and more preferably 400 or less.
- the oil and polyol which are liquid at 20 ° C. described above as an example of the component (c) can be used alone or in combination of two or more.
- the component (c) is preferably a polymer plasticizer having film-forming ability.
- the component (c) is an oil that is liquid at 20 ° C. selected from hydrocarbon oils, ester oils, silicone oils, and higher alcohols, and polyols selected from alkylene glycol, polyalkylene glycol, glycerin and triglycerin. It is preferable that it is 1 type, or 2 or more types of substances selected from these.
- the content of the component (a) in the spray composition is preferably 50% by mass or more, more preferably 55% by mass or more, and further preferably 60% by mass or more. Moreover, it is preferable that it is 98 mass% or less, it is still more preferable that it is 96 mass% or less, and it is still more preferable that it is 94 mass% or less.
- the content of the component (a) in the spray composition is preferably 50% by mass to 98% by mass, more preferably 55% by mass to 96% by mass, and 60% by mass to 94% by mass. More preferably, it is as follows. By blending the component (a) in the spray composition at this ratio, the spray composition can be sufficiently volatilized when the electrostatic spray method is performed.
- the spray composition in the present invention can contain solid or semi-solid oil, that is, oil other than the component (c), as long as the effects of the present invention are not impaired.
- oil other than the component (c) is 10% by mass or less in the spray composition. It is preferably 8% by mass or less, more preferably 6% by mass or less.
- the content of the component (b) in the spray composition is preferably 2% by mass or more from the viewpoint of appropriately adjusting the thickness of the fiber by setting the viscosity of the spray composition to an appropriate range,
- the content is more preferably 4% by mass or more, and further preferably 6% by mass or more. Further, it is preferably 50% by mass or less, more preferably 45% by mass or less, and further preferably 40% by mass or less.
- the content of the component (b) in the spray composition is preferably 2% by mass or more and 50% by mass or less, more preferably 4% by mass or more and 45% by mass or less, and more preferably 6% by mass or more and 40% by mass. More preferably, it is as follows. By blending the component (b) in the spray composition at this ratio, the target film can be successfully formed.
- the content of the component (c) in the spray composition is preferably 0.5% by mass or more, more preferably 1.0% by mass or more, and 1.5% by mass or more. Is more preferable. Further, it is preferably 30% by mass or less, more preferably 25% by mass or less, and further preferably 20% by mass or less.
- the content of the component (c) in the spray composition is preferably 0.5% by mass or more and 30% by mass or less, more preferably 1% by mass or more and 25% by mass or less, and 1.5% by mass. More preferably, it is 20 mass% or less.
- composition for spraying only the component (a), the component (b) and the component (c) described above may be contained, or the component (a) and the component are within the range not impairing the effects of the present invention.
- Other components may be included in addition to (b) and component (c).
- Other components include, for example, colored pigments, extender pigments, dyes, surfactants having an HLB value of more than 10, UV protection agents, fragrances, repellents, antioxidants, stabilizers, preservatives, antiperspirants, various types Vitamins and the like are listed.
- each of these agents is not limited to the use as each agent, and other uses, for example, an antiperspirant can be used as a fragrance according to the purpose.
- the blending ratio of the other components is preferably 0.1% by mass or more and 30% by mass or less, and 0.5% by mass or more and 20% by mass or less. More preferably it is.
- a spray composition having a viscosity of preferably 1 mPa ⁇ s or more, more preferably 10 mPa ⁇ s or more, and further preferably 50 mPa ⁇ s or more at 25 ° C. is used. Further, at 25 ° C., the viscosity is preferably 5000 mPa ⁇ s or less, more preferably 2000 mPa ⁇ s or less, and still more preferably 1500 mPa ⁇ s or less.
- the viscosity of the spray composition is preferably 1 mPa ⁇ s or more and 5000 mPa ⁇ s or less, more preferably 10 mPa ⁇ s or more and 2000 mPa ⁇ s or less, and further preferably 50 mPa ⁇ s or more and 1500 mPa ⁇ s or less at 25 ° C. It is.
- a spray composition having a viscosity in this range a coating film, specifically, a porous coating film composed of a fiber deposit can be successfully formed by an electrostatic spray method.
- the viscosity of the spray composition is more preferably 50 mPa ⁇ s or more and 1000 mPa ⁇ s or less at 25 ° C.
- the viscosity of the spray composition is measured at 25 ° C. using an E-type viscometer.
- E type viscometer for example, an E type viscometer manufactured by Tokyo Keiki Co., Ltd. can be used.
- rotor rotor No. 43 can be used.
- the conditions for measuring the viscosity specifically, the rotor model number, rotation speed, rotation time, etc., are determined by the viscosity in each E-type viscometer.
- the composition for spraying is sprayed directly onto a film forming target, for example, human skin, by an electrostatic spray method.
- the electrostatic spraying method includes a step of forming a film by electrostatically spraying the spray composition on the skin using an electrostatic spraying device in an electrostatic spraying step.
- the electrostatic spray device includes a container for storing a spraying composition, a nozzle for discharging the spraying composition, a supply device for supplying the spraying composition contained in the container to the nozzle, and a voltage applied to the nozzle. And a power source for applying a voltage.
- 1 is a schematic diagram showing the configuration of an electrostatic spray apparatus preferably used in the present invention.
- An electrostatic spray apparatus 10 shown in FIG. 1 includes a low voltage power supply 11.
- the low voltage power supply 11 can generate a voltage of several volts to several tens of volts.
- the low voltage power supply 11 is preferably composed of one or two or more batteries. Further, by using a battery as the low voltage power supply 11, there is an advantage that replacement can be easily performed as necessary. Instead of the battery, an AC adapter or the like can be used as the low voltage power supply 11.
- the electrostatic spray device 10 also includes a high voltage power supply 12.
- the high voltage power supply 12 is connected to the low voltage power supply 11 and includes an electronic circuit (not shown) that boosts the voltage generated by the low voltage power supply 11 to a high voltage.
- the step-up electronic circuit is generally composed of a transformer, a capacitor, a semiconductor element, and the like.
- the electrostatic spray device 10 further includes an auxiliary electric circuit 13.
- the auxiliary electric circuit 13 is interposed between the low voltage power supply 11 and the high voltage power supply 12 described above, and has a function of adjusting the voltage of the low voltage power supply 11 and causing the high voltage power supply 12 to operate stably. Further, the auxiliary electric circuit 13 has a function of controlling the rotational speed of a motor provided in the micro gear pump 14 described later. By controlling the number of rotations of the motor, the amount of the spray composition supplied from the spray composition container 15 to be described later to the micro gear pump 14 is controlled.
- a switch SW is attached between the auxiliary electric circuit 13 and the low voltage power source 11, and the electrostatic spray device 10 can be operated / stopped by turning on / off the switch SW.
- the electrostatic spray device 10 further includes a nozzle 16.
- the nozzle 16 is made of various conductors including metals and non-conductors such as plastic, rubber, and ceramic, and has a shape that allows the spray composition to be discharged from its tip.
- a minute space through which the spraying composition flows is formed along the longitudinal direction of the nozzle 16.
- the size of the cross section of the minute space is preferably 100 ⁇ m or more and 1000 ⁇ m or less in terms of diameter.
- the nozzle 16 communicates with the micro gear pump 14 through a pipe line 17.
- the conduit 17 may be a conductor or a non-conductor.
- the nozzle 16 is electrically connected to the high voltage power supply 12. As a result, a high voltage can be applied to the nozzle 16.
- the nozzle 16 and the high voltage power supply 12 are electrically connected via the current limiting resistor 19 in order to prevent an excessive current from flowing when the human body directly touches the nozzle 16.
- the micro gear pump 14 that communicates with the nozzle 16 via the pipe line 17 functions as a supply device that supplies the spray composition contained in the container 15 to the nozzle 16.
- the micro gear pump 14 operates by receiving power from the low voltage power source 11. Further, the micro gear pump 14 is configured to supply a predetermined amount of the spray composition to the nozzle 16 under the control of the auxiliary electric circuit 13.
- the container 15 is connected to the micro gear pump 14 through a flexible pipe 18.
- the container 15 contains a spraying composition.
- the container 15 is preferably in a cartridge-type replaceable form.
- FIG. 2 shows a handy type electrostatic spray apparatus 10 having a size that can be held with one hand.
- the electrostatic spray device 10 shown in the figure all members in the configuration diagram shown in FIG. 1 are accommodated in a cylindrical housing 20.
- a nozzle (not shown) is disposed at one end 10 a in the longitudinal direction of the housing 20.
- the nozzle is arranged in the casing 20 so that the blowing direction of the composition coincides with the longitudinal direction of the casing 20 and is convex toward the skin side that is the film forming target.
- FIG. 2 shows a state where one end 10a of the electrostatic spray device 10 is directed to the inner side of the user's forearm. Under this state, the apparatus 10 is turned on to perform the electrostatic spray method. When the apparatus 10 is turned on, an electric field is generated between the nozzle and the skin.
- the apparatus 10 is turned on, an electric field is generated between the nozzle and the skin.
- a positive high voltage is applied to the nozzle and the skin becomes the negative electrode.
- the spray composition at the tip of the nozzle is polarized by electrostatic induction so that the tip becomes a cone shape, and droplets of the spray composition charged from the tip of the cone are charged.
- component (a) which is a solvent, evaporates from the spray composition charged and discharged into space, the charge density on the surface of the spray composition becomes excessive and spreads into the space while repeating miniaturization by Coulomb repulsion.
- the sprayed composition can reach the skin in the form of droplets.
- the component (a) of the volatile substance that is a solvent is volatilized from the composition, and a polymer having a film-forming ability that is a solute is solidified and stretched and deformed by a potential difference.
- a polymer having a film-forming ability that is a solute is solidified and stretched and deformed by a potential difference.
- fibers and deposit them are formed on the surface of the skin. For example, increasing the viscosity of the spray composition tends to deposit the composition on the surface of the skin in the form of fibers. As a result, a film composed of fiber deposits is formed on the surface of the skin.
- a film made of fiber deposits can also be formed by adjusting the distance between the nozzle and the skin and the voltage applied to the nozzle.
- the electrostatic spray method a high potential difference is generated between the skin, which is a film formation target, and the nozzle.
- the impedance is very large, the current flowing through the human body is extremely small.
- the present inventor has confirmed that the current flowing through the human body during the electrostatic spray method is several orders of magnitude smaller than the current flowing through the human body due to static electricity generated in normal life.
- the thickness of the fiber is preferably 10 nm or more, and more preferably 50 nm or more, when expressed in terms of a circle equivalent diameter. Moreover, it is preferable that it is 3000 nm or less, and it is still more preferable that it is 1000 nm or less.
- the thickness of the fiber is observed by, for example, scanning electron microscope (SEM) observation by magnifying the fiber 10,000 times, and removing defects (fiber clumps, fiber intersections, droplets) from the two-dimensional image, It can be measured by selecting 10 fibers arbitrarily, drawing a line perpendicular to the longitudinal direction of the fiber, and directly reading the fiber diameter.
- SEM scanning electron microscope
- the coating film which is a fiber deposit formed by electrostatic spraying, has a liquid-carrying coating film containing component (c) on the surface side of the constituent fibers.
- the surface side of the fiber means the surface or a part of the surface and between the fibers.
- the component (c) is more likely to bleed out from the constituent fibers, and the liquid-supported coating is easily formed between the constituent fibers, while the components ( If content of c) is less than 1 mass%, the said liquid agent carrying
- the transparent or translucent film is a state in which the film is formed on the film formation target. For example, when the film target is human skin, the human being the film formation target through the film.
- the skin color preferably a state where a fine pattern or the like can be visually recognized, the skin color or the like can be confirmed with the naked eye, and preferably the fine structure such as the skin skin can be visually recognized with the naked eye It is more preferable that the presence of the coating is difficult to understand.
- the color of the film is preferably white from the viewpoint of making it difficult to understand the presence of the film on the surface of a film-forming object such as skin.
- the L value when electrosprayed is preferably 80 or more, more preferably 90 or more. From the same viewpoint, the a value and the b value are preferably ⁇ 20 to 30, and more preferably ⁇ 10 to 20, more preferably 0-10.
- the L value is a value defined in CIE 1976 (L *, a *, b *) color space (CIELAB), where 100 is white and 0 is black. Furthermore, since the persistence of the adhesion is increased, it is effective for the moisture retention of the skin, which is a film formation target, or for improving the skin condition.
- the component (c) is combined in the fiber, so that the fiber is swollen and easily plasticized.
- the change in the fiber diameter over time when observed, the fiber electrostatically sprayed on the skin surface becomes larger in diameter due to swelling than the fiber electrostatically sprayed on the metal surface.
- the coating film containing fibers formed by electrostatic spraying is plasticized with oil and moisture in the skin and becomes softer, so that the followability of the fibers themselves to the texture of the skin is improved.
- the liquid agent that is, the component (c) bleeds out and exists between the fiber surface and the fiber, so that the coating film containing the fiber becomes translucent or transparent, and natural appearance is imparted.
- the fiber diameter due to swelling satisfies the following formula (1). (Fiber diameter after 30 seconds of spinning on the skin)> (fiber diameter after 30 seconds of spinning on the metal plate) (1)
- Mass ratio of component (c) to total amount of component (c) and component (b) ((c) / ((b) in a coating or spraying composition, which is a fiber deposit formed by electrostatic spraying. ) + (C))) is preferably 0.05 or more, more preferably 0.08 or more, and still more preferably 0.1 from the viewpoint of improving the adhesion of the coating and the appearance of the coating. From the viewpoint of film formability by electrostatic spraying, it is preferably 0.75 or less, more preferably 0.7 or less, and even more preferably 0.55 or less.
- the mass ratio ((c) / ((b) + (c))) of component (c) to the total amount of component (c) and component (b) in the coating or spray composition is: It is preferably from 0.05 to 0.75, more preferably from 0.08 to 0.7, and even more preferably from 0.1 to 0.55. If the value of ((c) / ((b) + (c))) is within the above range, fibers are easily formed, stickiness of the coating surface formed by electrostatic spraying is suppressed, The touch is improved.
- the contents of the component (a), the component (b) and the component (c) which are the spray composition are measured as follows. Since the component (a) which is a volatile substance does not exist in the formed film or volatilizes even if it exists, the formed film contains only the component (b) and the component (c). The content is measured as follows.
- ⁇ Method of measuring the content of component (b) and component (c) in the formed film> A solvent capable of dissolving the film is searched, and after the film is dissolved in the solvent, separation and identification by liquid chromatography (HPLC) or infrared spectrophotometer (IR) is performed.
- HPLC liquid chromatography
- IR infrared spectrophotometer
- the fiber forming the coating is a continuous fiber having an infinite length on the principle of production, but preferably has a length at least 100 times the thickness of the fiber.
- the formed film contains a fiber containing the component (b), preferably having a length of 10 ⁇ m or more, more preferably 50 ⁇ m or more, and even more preferably 100 ⁇ m or more.
- a fiber having a length of 100 times or more the thickness of the fiber is defined as “continuous fiber”.
- the cross-sectional shape of the fiber is preferably a circle or an ellipse, and the thickness of the fiber is a diameter in the case of a circle and a length of a major axis in the case of an ellipse.
- the film manufactured by the electrostatic spray method is a porous discontinuous film composed of a deposit of one or two or more continuous fibers.
- the coating in such a form is not only easy to handle as a single sheet as an aggregate because the fiber itself is flexible and soft, but also has a very soft feature, and it is difficult to break apart even if shearing force is applied to it, There is an advantage that it is excellent in following the movement of the body. There is also an advantage that the complete removal of the coating is easy.
- the continuous film having no pores is not easily peeled off and has low sweat dissipation, so that there is a risk of stuffiness on the skin.
- a porous discontinuous film composed of aggregates of particles is required to be completely removed without damaging the skin, for example, an operation such as rubbing the entire film is required to completely remove the film. Have difficulty.
- the coating film formed by the composition for spraying contains the component (c) includes a bonding portion that bonds at a portion where the fibers intersect.
- the coating film formed by the composition for spraying contains the component (c) includes a bonding portion that bonds at a portion where the fibers intersect.
- the spraying composition that has been electrostatically sprayed into a fibrous form is charged with the component (b) and the component (c) while the component (a) evaporates. Reach the skin directly in the state.
- the fibers adhere to the skin in the form of a single film by electrostatic force. Since fine irregularities such as texture are formed on the surface of the skin, the fibers are more closely attached to the surface of the skin in the form of a single film coupled with the anchor effect due to the irregularities.
- electrostatic sprayer 10 is turned off.
- the fiber which comprises a film contains the component (c), even if it does not apply
- the fibers constituting the film are difficult to reflect light, and the appearance of the film becomes transparent. It is easy to cover the skin with a natural appearance.
- the distance between the nozzle and the skin depends on the voltage applied to the nozzle, but is preferably 50 mm or more and 150 mm or less in order to successfully form the coating.
- the distance between the nozzle and the skin can be measured by a generally used non-contact sensor or the like.
- the basis weight of the coating is preferably 0.1 g / m 2 or more, and preferably 1 g / m 2 or more. Is more preferable. Moreover, it is preferable that it is 50 g / m ⁇ 2 > or less, and it is still more preferable that it is 40 g / m ⁇ 2 > or less.
- the basis weight of the coating is preferably 0.1 g / m 2 or more 50 g / m 2 or less, further preferably 1 g / m 2 or more 40 g / m 2 or less.
- the film is directly formed on the skin, which is the film formation target.
- the film formation target is a metal part other than the skin, such as stainless steel or tableware constituting the body of a car or a system kitchen. Or ceramics.
- the film formation target is not a base for forming a film on a base and then depositing the film on the target as in the prior art, but an object that is directly sprayed to form a film. Therefore, even if the surface of the film forming object is a surface with large and small irregularities, a moving object such as bending and stretching, or a surface with waviness, the film of the present invention obtains high adhesion and excellent appearance. be able to.
- the film formation target include curved portions and stepped tableware, cars, walls with fine unevenness and pores, skin, nails, and the like, more preferably skin and nails, The skin is preferred.
- the film manufacturing method of the present invention is useful as various beauty methods not intended for human surgery, treatment or diagnosis.
- the method for producing a coating of the present invention is used for cosmetic purposes, whitening the skin, hiding skin spots, hiding skin dullness / bearing, hiding skin wrinkles, blurring skin, protecting skin from UV rays, skin Can be applied for moisturizing.
- various kinds of actions for protecting the skin personally at home for example, protection of various wounds such as abrasions, cuts, lacerations and stab wounds, prevention of pressure sores, etc.
- the method can also be applied. Even if the formed film is formed on a site where the degree of expansion and contraction of the skin such as a joint is large or a site where the curvature is large such as a shoulder, the film is hardly peeled off or broken.
- the electrostatic spray device 10 is a device that can be gripped by a human hand, and is an electrostatic spray device having an operation unit that includes a spray nozzle that can be gripped by a human hand. Also good.
- the present invention further discloses the following method for producing a coating film.
- the composition comprises the following component (a), component (b) and component (c).
- B A polymer having a film-forming ability.
- C The liquid agent containing 1 type, or 2 or more types selected from a liquid oil and a polyol at 20 degreeC.
- the said coating film is a manufacturing method of the film as described in said ⁇ 1> which has the liquid agent carrying
- the value of the mass ratio ((c) / ((b) + (c))) of the component (c) to the total amount of the component (b) and the component (c) in the coating or spray composition is Preferably, it is 0.05 or more and 0.75 or less, More preferably, it is 0.08 or more and 0.7 or less, The manufacturing method of the film as described in said ⁇ 1> or ⁇ 2>.
- ⁇ 4> The value of the mass ratio ((c) / ((b) + (c))) of component (c) to the total amount of component (b) and component (c) in the coating or spray composition is preferably The manufacturing method of the film as described in said ⁇ 1> or ⁇ 2> which is 0.1 or more and 0.55 or less.
- ⁇ 5> Any one of ⁇ 1> to ⁇ 4>, wherein the component (c) is one or more substances selected from hydrocarbon oils, ester oils, silicone oils, higher alcohols, and polyols. The manufacturing method of the coating film of description.
- the composition is electrostatically sprayed onto the film forming object using an electrostatic spray apparatus to form a film made of a fiber deposit
- the electrostatic spray apparatus includes the composition A container for containing an object, a nozzle for discharging the composition, a supply device for supplying the composition contained in the container to the nozzle, and a power source for applying a voltage to the nozzle. 1> thru
- the volatile substance of component (a) has a vapor pressure of 0.01 kPa to 106.66 kPa at 20 ° C., preferably 0.13 kPa to 66.66 kPa, and more preferably 0.67 kPa to 40.
- the volatile substance of component (a) contains an alcohol, preferably one or two selected from monovalent chain aliphatic alcohols, monovalent cycloaliphatic alcohols, and monovalent aromatic alcohols.
- any one of the above ⁇ 1> to ⁇ 7> which contains one or more, more preferably one or more selected from ethanol, isopropyl alcohol, butyl alcohol, phenylethyl alcohol, propanol, and pentanol.
- the volatile substance of component (a) is a mixture of (a1) one or more selected from ethanol, isopropyl alcohol, and butyl alcohol, and (a2) water, preferably component ((a2) and The method for producing a coating film according to any one of ⁇ 1> to ⁇ 8>, wherein the mass ratio (a2) / (a1) of the component (a1) is 0.0025 or more and 0.3 or less.
- ⁇ 10> Any one of ⁇ 1> to ⁇ 9>, wherein the volatile substance of component (a) contains a ketone, preferably contains one or more selected from acetone, methyl ethyl ketone, and methyl isobutyl ketone.
- the volatile substance of component (a) is one or more selected from (a1) ethanol, isopropyl alcohol, butyl alcohol, and (a2) water, preferably 1 selected from ethanol and butyl alcohol.
- the film-forming polymer contains a water-soluble polymer, and preferably the water-soluble polymer having a film-forming ability is pullulan, hyaluronic acid, chondroitin sulfate, poly- ⁇ -glutamic acid, modified corn starch, ⁇ -glucan , Mucopolysaccharides such as gluco-oligosaccharide, heparin, keratosulfuric acid, cellulose, pectin, xylan, lignin, glucomannan, galacturonic acid, psyllium seed gum, tamarind seed gum, gum arabic, tragacanth gum, soybean water-soluble polysaccharide, alginic acid, carrageenan, laminaran , Natural polymers such as agar (agarose), fucoidan, methylcellulose, hydroxypropylcellulose, hydroxypropylmethylcellulose, partially saponified polyvinyl alcohol (not used in combination with a crosslinking agent), low
- the polymer having a film-forming ability contains a water-insoluble polymer, and the water-insoluble polymer having a film-forming ability is used together with a completely saponified polyvinyl alcohol that can be insolubilized after the film is formed, and a crosslinking agent.
- PVA polylactic acid
- polyacrylonitrile resin acrylic resin, polystyrene resin, polyvinyl butyral resin, polyethylene terephthalate resin, polybutylene terephthalate that can be crosslinked after formation
- the component (c) oil which is liquid at 20 ° C. is one or more selected from hydrocarbon oil, ester oil, silicone oil and higher alcohol which are liquid at 20 ° C., preferably ester oil, And one or more selected from higher alcohols, more preferably one or more selected from ester oils, the film according to any one of the above ⁇ 1> to ⁇ 13> Production method.
- Method. ⁇ 16> The oil that is liquid at 20 ° C.
- the component (c) is preferably one or more selected from hydrocarbon oil, ester oil, silicone oil, and higher alcohol that are liquid at 20 ° C., more preferably.
- the component (c) liquid hydrocarbon oil at 20 ° C. is one or more selected from liquid paraffin, squalane, squalene, n-octane, n-heptane, cyclohexane, light isoparaffin, and liquid isoparaffin.
- the viscosity of the hydrocarbon oil at 30 ° C. is preferably 10 mPa ⁇ s or more, more preferably 30 mPa ⁇ s or more, and the method for producing a coating film according to any one of ⁇ 14> to ⁇ 17>.
- the content of isododecane, isohexadecane, and hydrogenated polyisobutene in the composition having a viscosity of less than 10 mPa ⁇ s at 30 ° C. is preferably 10% by mass or less, more preferably 5% by mass or less, More preferably 1% by weight or less, even more preferably 0.5% by weight or less, or preferably the composition has a viscosity of less than 10 mPa ⁇ s at 30 ° C., and isododecane, isohexadecane, and The method for producing a coating film according to any one of ⁇ 1> to ⁇ 18>, wherein the coating film does not contain hydrogenated polyisobutene.
- the component oil (c), which is liquid at 20 ° C., is an ester compound having an HLB value of 10 or less, and includes fatty acid esters, fatty acid alcohol esters, polyhydric alcohol esters, glycerin fatty acid esters, polyglycerin fatty acid esters, sorbitan fatty acids.
- the manufacturing method of the film as described in said ⁇ 14> or ⁇ 16> which is 1 type, or 2 or more types chosen from ester.
- component (c) is isopropyl myristate, cetyl octanoate, octyldodecyl myristate, isopropyl stearate, butyl stearate, hexyl laurate, myristyl myristate, decyl oleate, dimethyl Hexyldecyl octoate, cetyl lactate, myristyl lactate, lanolin acetate, isocetyl stearate, isocetyl isostearate, cholesteryl 12-hydroxystearylate, ethylene glycol di-2-ethylhexanoate, dipentaerythritol fatty acid ester, N-alkyl monoisostearate Glycol, neopentyl glycol dicaprate, diisostearyl malate, glycerin di-2-heptylundecanoate, trimethylolpro tri-2-ethyl
- the fatty acid ester, fatty acid alcohol ester, polyhydric alcohol ester, and glycerin fatty acid ester of ester oil that is liquid at 20 ° C. as the component (c) are preferably octyldodecyl myristate, myristyl myristate, isocetyl stearate, isostearic acid Isocetyl, cetearyl isononanoate, isobutyl adipate, diethyl sebacate, di-2-ethylhexyl sebacate, isopropyl myristate, isopropyl palmitate, diisostearyl malate, neopentyl glycol dicaprate, benzoic acid (12 to 12 carbon atoms) 15) One or more selected from alkyl, isononyl isononanoate, tri (caprylic acid / capric acid) glycerin, triacyl glycer
- sorbitan fatty acid ester of an ester oil which is liquid at 20 ° C. has an HLB value of 10 or less, sorbitan monostearate, sorbitan monooleate, sorbitan sesquioleate, sorbitan sesquiisostearate, monopalmitin
- the component (c), which is a liquid silicone oil at 20 ° C., is one or two selected from dimethylpolysiloxane, dimethylcyclopolysiloxane, methylphenylpolysiloxane, methylhydrogenpolysiloxane, and higher alcohol-modified organopolysiloxane.
- the content of the silicone oil in the composition is 10% by mass or less, preferably 5% by mass or less, more preferably 1% by mass or less, still more preferably 0.1% by mass or less, The kinematic viscosity of the silicone oil at 25 ° C.
- the said silicone oil is a manufacturing method of the film as described in said ⁇ 25> containing dimethylpolysiloxane.
- the component (c) liquid higher alcohol at 20 ° C. is a liquid higher alcohol having 12 to 20 carbon atoms, and the higher alcohol is one or two selected from branched fatty acids or unsaturated fatty acid higher alcohols.
- the polyol as component (c) is ethylene glycol, propylene glycol, 1,3-propanediol, 1,3-butanediol, diethylene glycol, dipropylene glycol, polyethylene glycol having a number average molecular weight of 1000 or less, polypropylene glycol, glycerin.
- the component (c) liquid oil and polyol at 20 ° C. are oils selected at 20 ° C. selected from hydrocarbon oils, ester oils, silicone oils, and higher alcohols, and alkylene glycols, polyalkylene glycols, glycerin and triglycerides.
- the component (a) contains (a1) ethanol and (a2) water, and the component (b) is fully saponified polyvinyl alcohol, partially saponified polyvinyl alcohol, polyvinyl butyral resin, (alkyl acrylate / octylamide) copolymer, oxazoline ⁇ 1 or 2 or more types selected from modified silicone, polyester and twein, and component (c) is 1 or 2 or more types selected from hydrocarbon oils, ester oils, silicone oils and polyols ⁇ 1 >
- component (c) is 1 or 2 or more types selected from hydrocarbon oils, ester oils, silicone oils and polyols ⁇ 1 >
- To ⁇ 29> The method for producing a coating film according to any one of ⁇ 29>.
- the component (a) contains (a1) ethanol and (a2) water, and the component (b) is one or more selected from polyvinyl butyral resin and (alkyl acrylate / octylamide) copolymer, ⁇ 1> thru
- Content of the said component (a) in the said composition becomes like this. Preferably they are 50 to 98 mass%, More preferably, they are 55 to 96 mass%, More preferably, they are 60 to 94 mass%. %.
- the oil other than the component (c) has a content in the composition of preferably 10% by mass or less, more preferably 8% by mass or less, and further preferably 6% by mass or less, ⁇ 1 >
- the content of the component (b) in the composition is preferably 2% by mass or more and 50% by mass or less, more preferably 4% by mass or more and 45% by mass or less, and further preferably 6% by mass or more and 40% by mass. %.
- the content of the component (c) in the composition is preferably 0.5% by mass or more and 30% by mass or less, more preferably 1% by mass or more and 25% by mass or less, and further preferably 1.5% by mass. % Or more and 20 mass% or less, The manufacturing method of the film as described in any one of said ⁇ 1> thru
- the content of the component (a) in the composition is 55% by mass to 96% by mass, the content of the component (b) is 4% by mass to 45% by mass, and the content of the component (c) is The method for producing a film according to any one of ⁇ 1> to ⁇ 35>, which is 1% by mass or more and 25% by mass or less, and more preferably 1.5% by mass or more and 20% by mass or less.
- the composition contains only the component (a), the component (b) and the component (c), or the component (a), the component (b) and the component (c).
- the other components include coloring pigments, extender pigments, dyes, surfactants with HLB values exceeding 10, UV protection agents, fragrances, repellents, antioxidants, stabilizers, preservatives, antiperspirants, and various vitamins.
- ⁇ 38> The coating ratio according to ⁇ 37>, wherein the blending ratio of the other components in the composition is 0.1% by mass or more and 30% by mass or less, preferably 0.5% by mass or more and 20% by mass or less.
- the composition has a viscosity of 1 mPa ⁇ s or more at 25 ° C., preferably 10 mPa ⁇ s or more, more preferably 50 mPa ⁇ s or more, and at 25 ° C., 5000 mPa ⁇ s or less, preferably 2000 mPa ⁇ s or less.
- ⁇ 40> The method for producing a film according to any one of ⁇ 1> to ⁇ 39>, wherein the object for forming a film is a surface of human skin.
- the said coating film is a manufacturing method of the coating film as described in said ⁇ 40> which is transparent or semi-transparent which can visually recognize the color of skin through a film after formation of a film.
- ⁇ 42> The method for producing a coating film according to any one of ⁇ 1> to ⁇ 41>, wherein the composition is liquid at 20 ° C., and the fibers are formed by electrostatic spraying of the liquid composition.
- ⁇ 43> The method for producing a coating according to any one of ⁇ 1> to ⁇ 42>, wherein the fibers forming the coating are continuous fibers.
- the said coating film is a manufacturing method of the coating film as described in any one of said ⁇ 1> thru
- the film formation target is human skin, and any one of ⁇ 1> to ⁇ 44> for forming a transparent or translucent film made of a deposit containing fibers on the skin by electrostatic spraying.
- ⁇ 46> The use for producing a film by electrostatic spraying according to any one of ⁇ 1> to ⁇ 44> of the composition, wherein the film forming object is human skin.
- the device used for the electrostatic spraying is an electrostatic spraying device that can be gripped by a human hand or an electrostatic spraying device having an operation unit including a spray nozzle that can be gripped by a human hand.
- the manufacturing method of the film as described in any one of>.
- Example 1 (1) Preparation of spraying composition Ethanol (made by Wako Pure Chemical Industries, Ltd .: trade name ethanol (99.5)) was used as component (a) of the spraying composition.
- Polyvinyl butyral manufactured by Sekisui Chemical Co., Ltd .: trade name S-LEC B BM-1 was used as component (b) of the spray composition.
- component (c) of the spray composition glycerin (manufactured by Kao Corporation: trade name: concentrated glycerin for cosmetics) was used.
- the blending ratio in the spray composition is as shown in Tables 1 and 2.
- the amounts of ethanol and glycerin shown in Tables 1 and 2 are effective amounts and do not include water.
- Example 2 to 5 An electrostatic spray process was performed in the same manner as in Example 1 except that the components (a), (b) and (c) in the spray composition were changed to the conditions shown in Table 1 below, A coating was obtained.
- Example 6 to 10 The component (c) in the spray composition was changed to polyglyceryl diisostearate-2 (manufactured by Nisshin Oillio Group Co., Ltd .: trade name Cosmol 42V), and the conditions were as shown in Table 1 below. Then, an electrostatic spray process was performed to obtain a film made of fiber deposits.
- Example 11 The component (a) in the spray composition was changed to the ethanol used in Example 1 with 1-butanol (manufactured by Wako Pure Chemical Industries, Ltd .: trade name: 1 Butanol) added to the component (b). (Alkyl acrylate / octyl acrylamide) copolymer (manufactured by Akzobel: trade name DERMACRYL 79) and component (c) is changed to polyglyceryl-10 diisostearate (trade name: Matsumoto MI-102, made by Matsumoto Fine Chemical Co., Ltd.) The electrostatic spraying process was performed in the same manner as in Example 1 except that the conditions shown in Table 1 below were used, to obtain a film composed of a fiber deposit.
- Example 1 An electrostatic spraying process was performed in the same manner as in Example 1 except that the spray composition did not contain the component (c) to obtain a coating film composed of fiber deposits.
- Example 2 The spray composition does not contain the component (c), and the component (a) is further added with 1-butanol (trade name: 1 Butanol) manufactured by Wako Pure Chemical Industries, Ltd. to the ethanol used in Example 1.
- 1-butanol trade name: 1 Butanol
- the component (b) is changed to (alkyl acrylate / octyl acrylamide) copolymer (manufactured by Akzobel: trade name DERMACRYL 79)
- an electrostatic spraying process is performed, and a fiber is obtained. A film consisting of a deposit was obtained.
- ⁇ Adhesion with skin> 1 Almost all of the coating is peeled off immediately after spinning in the electrostatic spray process.
- 2 The film adheres immediately after spinning in the electrostatic spraying process, but 50% or more of the film area peels off after 30 minutes in a stationary state.
- 3 The film adheres immediately after spinning in the electrostatic spraying process, but less than 50% of the film area peels off after 30 minutes in a stationary state.
- 4 After spinning in the electrostatic spraying process, the film adheres for 30 minutes in a static state, but peels off when a shearing force is applied in the parallel direction with a finger.
- 5 After spinning in the electrostatic spraying process, the film adheres for 30 minutes in a stationary state, and does not peel even when a shearing force is applied in the parallel direction with a finger.
- ⁇ Appearance of coating> 1 The appearance of the film appears white. 2: Appearance of the coating looks translucent close to white. 3: The appearance of the film appears translucent. 4: The appearance of the film appears thin and translucent. 5: The appearance of the film appears transparent.
- Example 12 Using the spray composition of Example 3, in the electrostatic spraying process of Example 1, the objects (d) and (e) shown below were subjected to electrostatic spraying for 5 seconds. A film composed of a fiber deposit 30 seconds after the attachment was obtained.
- Example 13 Using the spray composition of Example 7, an electrostatic spray process was carried out in the same manner as in Example 12 to obtain a film composed of fiber deposits.
- Example 14 An electrostatic spray process was performed in the same manner as in Example 12 except that the components (a), (b), and (c) in the spray composition were changed to the conditions shown in Table 1 below, and the fiber deposits were used. A coating was obtained.
- Comparative Example 3 Using the composition for spraying of Comparative Example 1, the electrostatic spraying process of Example 1 was carried out in the same manner as in Example 12 to obtain a film composed of fiber deposits.
- the film formed by the method of each example has higher adhesion to the skin and higher transparency than the film formed by the method of the comparative example. It was recognized that
- Examples 15 to 28 About the composition for spraying shown in Table 3, Table 4, it carried out similarly to Example 1, and performed the electrostatic spray process, and obtained the film which consists of fiber deposits. In the same manner as in Example 1, the adhesion to the skin and the appearance of the coating were evaluated, and the touch of the coating described below was evaluated. The evaluation results are shown in Tables 3 and 4.
- the viscosity of the spray composition used for forming the coating films of Examples 23 and 25 to 28 was measured under the following conditions. Table 5 shows the measurement results of the viscosity and the evaluation of the adhesion and feel of the coatings of Examples 23 and 25 to 28.
- the coatings of Examples 26 and 28 shown in Table 5 have a moist feel with high adhesion to the skin, but have a slight tack force, and the tack force is expressed without the component (b) being completely dried. It is thought that it is the touch by having done.
- the coating film of Example 27, in which the content of component (c) is large relative to component (b) has a slightly sticky feel with high adhesion to the skin, and is considered to be sticky derived from component (c). It is done.
- Viscosity was measured under the condition that the spray composition was stored at 25 ° C. for one day, and then at 25 ° C., the rotor No. 43, the number of revolutions was 1 rpm when 1280 mPa ⁇ s or more, 10 rpm when 128 mPa ⁇ s or more and less than 1280 mPa ⁇ s, and 100 rpm when less than 128 mPa ⁇ s.
- the SEM image is an image obtained by observing and photographing with an electron microscope (JEOL, JSM-6510) under the conditions of an acceleration voltage of 10 kV and 5000 times after 30 minutes from the formation of the film.
- JEOL, JSM-6510 an electron microscope
- FIG. 3 in the coating of Example 3, the presence of bonding points bonded to the intersections of the fibers is recognized.
- Example 29 About the spray composition of Table 6, it carried out similarly to Example 1, and performed the electrostatic spray process, and obtained the film which consists of fiber deposits. In the same manner as in Example 1, the appearance of the coating was evaluated. The evaluation results are shown in Table 6.
- Example 29 was translucent in appearance.
- the adhesion between the film forming object and the film formed by electrostatic spraying is enhanced, and the appearance of the film is transparent and the film forming object can be coated in a natural state.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Animal Behavior & Ethology (AREA)
- Epidemiology (AREA)
- Birds (AREA)
- Emergency Medicine (AREA)
- Dermatology (AREA)
- Chemical & Material Sciences (AREA)
- Inorganic Chemistry (AREA)
- Hematology (AREA)
- Heart & Thoracic Surgery (AREA)
- Biomedical Technology (AREA)
- Anesthesiology (AREA)
- Engineering & Computer Science (AREA)
- Cosmetics (AREA)
- Application Of Or Painting With Fluid Materials (AREA)
Abstract
Description
(a)水、アルコール及びケトンから選ばれる1種又は2種以上の揮発性物質。
(b)被膜形成能を有するポリマー。
(c)20℃で液体の油及びポリオールから選択される1種又は2種以上を含有する液剤。
噴霧用組成物は、静電スプレー法によって、被膜形成対象物の一例であるヒトの皮膚に、繊維を含む堆積物からなる透明又は半透明の被膜を形成する被膜の製造方法に使用される。
以下、各組成物について説明する。
HLB=(Σ無機性値/Σ有機性値)×10
成分(c)は、成分(b)のポリマーの被膜形成対象物への密着性をより向上する観点から、20℃において液体の油を含有することが好ましく、極性を有し、成分(b)のポリマーの被膜対象物へ密着性をより良好にする観点から、好ましくはエステル油、及び高級アルコールから選ばれる1種又は2種以上を含有することが好ましく、エステル油から選ばれる1種又は2種以上を含有することが好ましい。
これらの中では、静電噴霧された被膜を皮膚に密着させる観点及び皮膚に塗布した際の感触を向上させる観点から、イソステアリン酸ポリグリセリル、ジイソステアリン酸ポリグリセリル、トリイソステアリン酸ポリグリセリル、ステアリン酸ポリグリセリル、オレイン酸ポリグリセリル、セスキカプリン酸ポリグリセリルが好ましく、ジイソステアリン酸ポリグリセリルがより好ましい。
25℃におけるシリコーン油の動粘度は、静電噴霧された被膜を皮膚等に密着させる観点から、好ましくは3mm2/s以上であり、より好ましくは4mm2/s以上であり、更に好ましくは5mm2/s以上であり、好ましくは30mm2/s以下であり、より好ましくは20mm2/s以下であり、更に好ましくは10mm2/s以下である。これらの中では静電噴霧された被膜を密着させる観点から、シリコーン油はジメチルポリシロキサンを含むことが好ましい。
本発明における噴霧用組成物には、本発明の効果を損なわない範囲で、固体状や半固体状の油、すなわち、成分(c)以外の油を含有することができる。被膜の被膜形成対象物への密着性を向上させる観点から、噴霧用組成物の安定性の観点から、成分(c)以外の油は、噴霧用組成物中には、10質量%以下であることが好ましく、8質量%以下であることがより好ましく、6質量%以下であることが更に好ましい。
(皮膚に対して紡糸し、30秒後の繊維径)>(金属板に対して紡糸し、30秒後の繊維径)・・・(1)
溶液状態にて液体クロマトグラフ(HPLC)による分離同定や、赤外分光光度計(IR)にて同定する方法がある。液体クロマトグラフでは、分子量の大きい成分から溶出するため、分子量の予測や、成分の溶出位置によって組成を同定することもできる。IR分析では個々の吸収体より官能基を帰属し同定することも可能であり、一般的には市販添加剤の標準チャートと成分のIRチャートを比較することで同定することが可能である。
被膜を溶解可能な溶媒の探索を行い、溶媒に被膜を溶解後、液体クロマトグラフ(HPLC)による分離同定や、赤外分光光度計(IR)にて同定する。
<1>
被膜形成対象物の表面に被膜を形成する被膜の製造方法であって、前記被膜形成対象物に組成物を直接に静電スプレーして繊維を含む堆積物からなる被膜を形成する静電スプレー工程を具備し、前記組成物が、以下の成分(a)、成分(b)及び成分(c)を含む、被膜の製造方法。(a)水、アルコール及びケトンからなる群より選択される1種又は2種以上の揮発性物質。(b)被膜形成能を有するポリマー。(c)20℃で液体の油及びポリオールから選択される1種又は2種以上を含有する液剤。
前記被膜は、構成する前記繊維の表面側に、前記成分(c)が存在する液剤担持被膜を有している、前記<1>に記載の被膜の製造方法。
<3>
前記被膜あるいは噴霧用組成物における、前記成分(b)と前記成分(c)の合計量に対する前記成分(c)の質量比((c)/((b)+(c)))の値が、好ましくは0.05以上0.75以下であり、より好ましくは0.08以上0.7以下である、前記<1>又は<2>に記載の被膜の製造方法。
<4>
前記被膜あるいは噴霧用組成物における、成分(b)と成分(c)の合計量に対する成分(c)の質量比((c)/((b)+(c)))の値が、好ましくは0.1以上0.55以下である、前記<1>又は<2>に記載の被膜の製造方法。
<5>
前記成分(c)が、炭化水素油、エステル油、シリコーン油、高級アルコール、及びポリオールから選択される1種又は2種以上の物質である、前記<1>ないし<4>のいずれか一に記載の被膜の製造方法。
<6>
前記静電スプレー工程において、静電スプレー装置を用いて前記被膜形成対象物に前記組成物を静電スプレーして、繊維の堆積物からなる被膜を形成し、前記静電スプレー装置が、前記組成物を収容する容器と、前記組成物を吐出するノズルと、前記容器中に収容されている前記組成物を前記ノズルに供給する供給装置と、前記ノズルに電圧を印加する電源とを備える前記<1>ないし<5>のいずれか一に記載の被膜の製造方法。
前記成分(a)の揮発性物質は、その蒸気圧が20℃において0.01kPa以上106.66kPa以下であり、好ましくは0.13kPa以上66.66kPa以下であり、更に好ましくは0.67kPa以上40.00kPa以下であり、より一層好ましくは1.33kPa以上40.00kPa以下である、前記<1>ないし<6>のいずれか一に記載の被膜の製造方法。
<8>
前記成分(a)の揮発性物質は、アルコールを含有し、好ましくは一価の鎖式脂肪族アルコール、一価の環式脂肪族アルコール、及び一価の芳香族アルコールから選ばれる1種又は2種以上を含有し、より好ましくは、エタノール、イソプロピルアルコール、ブチルアルコール、フェニルエチルアルコール、プロパノール、及びペンタノールから選ばれる1種又は2種以上を含有する、前記<1>ないし<7>のいずれか一に記載の被膜の製造方法。
<9>
前記成分(a)の揮発性物質が、(a1)エタノール、イソプロピルアルコール、ブチルアルコールから選ばれる1種又は2種以上と、(a2)水の混合液であり、好ましくは成分((a2)と成分(a1)の質量比(a2)/(a1)が0.0025以上0.3以下である、前記<1>ないし<8>のいずれか一に記載の被膜の製造方法。
<10>
前記成分(a)の揮発性物質は、ケトンを含有し、好ましくはアセトン、メチルエチルケトン、及びメチルイソブチルケトンから選ばれる1種又は2種以上を含有する、前記<1>ないし<9>のいずれか一に記載の被膜の製造方法。
<11>
前記成分(a)の揮発性物質は、(a1)エタノール、イソプロピルアルコール、ブチルアルコール、及び(a2)水から選ばれる1種又は2種以上であり、好ましくはエタノール、及びブチルアルコールから選ばれる1種又は2種及び(a2)水であり、より好ましくは(a1)エタノール及び(a2)水である、前記<1>ないし<10>のいずれか一に記載の被膜の製造方法。
被膜形成能を有するポリマーは水溶性ポリマーを含有し、好ましくは、前記水溶性である被膜形成能を有するポリマーは、プルラン、ヒアルロン酸、コンドロイチン硫酸、ポリ-γ-グルタミン酸、変性コーンスターチ、β-グルカン、グルコオリゴ糖、ヘパリン、ケラト硫酸等のムコ多糖、セルロース、ペクチン、キシラン、リグニン、グルコマンナン、ガラクツロン酸、サイリウムシードガム、タマリンド種子ガム、アラビアガム、トラガントガム、大豆水溶性多糖、アルギン酸、カラギーナン、ラミナラン、寒天(アガロース)、フコイダン、メチルセルロース、ヒドロキシプロピルセルロース、ヒドロキシプロピルメチルセルロース等の天然高分子、部分鹸化ポリビニルアルコール(架橋剤と併用しない)、低鹸化ポリビニルアルコール、ポリビニルピロリドン(PVP)、ポリエチレンオキサイド、及びポリアクリル酸ナトリウムから選ばれる1種又は2種以上である、前記<1>ないし<11>のいずれか一に記載の被膜の製造方法。
<13>
成分(b)被膜形成能を有するポリマーは水不溶性ポリマーを含有し、前記水不溶性である被膜形成能を有するポリマーは、被膜形成後に不溶化処理できる完全鹸化ポリビニルアルコール、架橋剤と併用することで被膜形成後に架橋処理できる部分鹸化ポリビニルアルコール、オキサゾリン変性シリコーン、ポリビニルアセタールジエチルアミノアセテート、ツエイン、ポリエステル、ポリ乳酸(PLA)、ポリアクリロニトリル樹脂、アクリル樹脂、ポリスチレン樹脂、ポリビニルブチラール樹脂、ポリエチレンテレフタレート樹脂、ポリブチレンテレフタレート樹脂、ポリウレタン樹脂、ポリアミド樹脂、ポリイミド樹脂、及びポリアミドイミド樹脂から選ばれる1種又は2種以上であり、より好ましくは成分(b)は水不溶性ポリマーである、前記<1>ないし<12>のいずれか一に記載の被膜の製造方法。
<14>
前記成分(c)である20℃で液体の油は、20℃において液状の炭化水素油、エステル油、シリコーン油、及び高級アルコールから選ばれる1種又は2種以上であり、好ましくはエステル油、及び高級アルコールから選ばれる1種又は2種以上であり、より好ましくはエステル油から選ばれる1種又は2種以上を含有する、前記<1>ないし<13>のいずれか一に記載の被膜の製造方法。
<15>
前記成分(c)が、炭化水素油、エステル油、シリコーン油、及びポリオールから選択される1種又は2種以上である、前記<1>ないし<13>のいずれか一に記載の被膜の製造方法。
<16>
前記成分(c)である20℃で液体の油は、好ましくは20℃において液状の炭化水素油、エステル油、シリコーン油、及び高級アルコールから選ばれる1種又は2種以上であり、より好ましくは20℃において液状の炭化水素油、エステル油、及びシリコーン油から選ばれる1種又は2種以上を含む、前記<1>ないし<14>のいずれか一に記載の被膜の製造方法。
<17>
前記成分(c)である20℃で液体の炭化水素油は、流動パラフィン、スクワラン、スクワレン、n-オクタン、n-ヘプタン、シクロヘキサン、軽質イソパラフィン、及び流動イソパラフィンから選ばれる1種又は2種以上であり、好ましくは流動パラフィン、及びスクワランから選ばれる1種又は2種である、前記<14>ないし<16>のいずれか一に記載の被膜の製造方法。
<18>
前記炭化水素油の30℃における粘度は、好ましくは10mPa・s以上であり、より好ましくは30mPa・s以上である、前記<14>ないし<17>のいずれか一に記載の被膜の製造方法。
30℃において粘度が10mPa・s未満である、イソドデカン、イソヘキサデカン、及び水添ポリイソブテンの前記組成物中の含有量は、好ましくは10質量%以下であり、より好ましくは5質量%以下であり、更に好ましくは1質量%以下であり、より更に好ましくは0.5質量%以下であるか、または、好ましくは前記組成物は30℃において粘度が10mPa・s未満である、イソドデカン、イソヘキサデカン、及び水添ポリイソブテンを含有しない、前記<1>ないし<18>のいずれか一に記載の被膜の製造方法。
<20>
前記成分(c)である20℃で液体のエステル油は、HLB値が10以下のエステル化合物であり、脂肪酸エステル、脂肪酸アルコールエステル、多価アルコールエステル、グリセリン脂肪酸エステル、ポリグリセリン脂肪酸エステル、ソルビタン脂肪酸エステルから選ばれる1種又は2種以上である、前記<14>又は<16>に記載の被膜の製造方法。
<21>
前記成分(c)である20℃で液体のエステル油は、ミリスチン酸イソプロピル、オクタン酸セチル、ミリスチン酸オクチルドデシル、パルミチン酸イソプロピル、ステアリン酸ブチル、ラウリン酸ヘキシル、ミリスチン酸ミリスチル、オレイン酸デシル、ジメチルオクタン酸ヘキシルデシル、乳酸セチル、乳酸ミリスチル、酢酸ラノリン、ステアリン酸イソセチル、イソステアリン酸イソセチル、12-ヒドロキシステアリル酸コレステリル、ジ2-エチルヘキサン酸エチレングリコール、ジペンタエリスリトール脂肪酸エステル、モノイソステアリン酸N-アルキルグリコール、ジカプリン酸ネオペンチルグリコール、リンゴ酸ジイソステアリル、ジ2-ヘプチルウンデカン酸グリセリン、トリ2-エチルヘキサン酸トリメチロールプロパン、トリイソステアリン酸トリメチロールプロパン、テトラ2-エチルヘキサン酸ペンタエリスリット、トリ2-エチルヘキサン酸グリセリル、トリイソステアリン酸トリメチロールプロパン、セチル2-エチルヘキサノエート、2-エチルヘキシルパルミテート、ナフタレンジカルボン酸ジエチルヘキシル、安息香酸(炭素数12~15)アルキル、イソノニルイソノナノエート、セテアリルイソノナノエート、トリ(カプリル酸・カプリン酸)グリセリン、(ジカプリル酸/カプリン酸)ブチレングリコール、トリラウリン酸グリセリル、トリミリスチン酸グリセリル、トリパルミチン酸グリセリル、トリイソステアリン酸グリセリル、トリ2-ヘプチルウンデカン酸グリセリル、トリベヘン酸グリセリル、トリヤシ油脂肪酸グリセリル、トリオレイン酸グリセリル、トリリノール酸グリセリル、ヒマシ油脂肪酸メチルエステル、オレイン酸オレイル、パルミチン酸2-ヘプチルウンデシル、アジピン酸ジイソブチル、N-ラウロイル-L-グルタミン酸-2-オクチルドデシルエステル、アジピン酸ジ2-ヘプチルウンデシル、エチルラウレート、アジピン酸イソブチル、セバシン酸ジエチル、セバシン酸ジ2-エチルヘキシル、ミリスチン酸2-ヘキシルデシル、パルミチン酸2-ヘキシルデシル、アジピン酸2-ヘキシルデシル、セバシン酸ジイソプロピル、コハク酸ジ2-エチルヘキシル、クエン酸トリエチル、パラメトキシケイ皮酸2-エチルヘキシル、ジピバリン酸トリプロピレングリコールから選ばれる1種又は2種以上である、前記<20>に記載の被膜の製造方法。
<22>
前記成分(c)である20℃で液体のエステル油の脂肪酸エステル、脂肪酸アルコールエステル、多価アルコールエステル、及びグリセリン脂肪酸エステルは、好ましくはミリスチン酸オクチルドデシル、ミリスチン酸ミリスチル、ステアリン酸イソセチル、イソステアリン酸イソセチル、セテアリルイソノナノエート、アジピン酸イソブチル、セバシン酸ジエチル、セバシン酸ジ2-エチルヘキシル、ミリスチン酸イソプロピル、パルミチン酸イソプロピル、リンゴ酸ジイソステアリル、ジカプリン酸ネオペンチルグリコール、安息香酸(炭素数12~15)アルキル、イソノニルイソノナノエート、トリ(カプリル酸・カプリン酸)グリセリン、トリアシルグリセライドから選ばれる1種又は2種以上であり、より好ましくはミリスチン酸イソプロピル、パルミチン酸イソプロピル、リンゴ酸ジイソステアリル、セバシン酸ジエチル、ジカプリン酸ネオペンチルグリコール、安息香酸(炭素数12~15)アルキル、イソノニルイソノナノエート、トリ(カプリル酸・カプリン酸)グリセリン、及びトリアシルグリセライドから選ばれる1種又は2種以上である、前記<21>に記載の被膜の製造方法。
<23>
前記成分(c)である20℃で液体のエステル油のポリグリセリン脂肪酸エステルは、HLB値が10以下である、イソステアリン酸ポリグリセリル、ジイソステアリン酸ポリグリセリル、トリイソステアリン酸ポリグリセリル、ステアリン酸ポリグリセリル、オレイン酸ポリグリセリル、及びセスキカプリン酸ポリグリセリルから選ばれる1種又は2種以であり、より好ましくはジイソステアリン酸ポリグリセリルである前記<20>に記載の被膜の製造方法。
前記成分(c)である20℃で液体のエステル油のソルビタン脂肪酸エステルは、HLB値が10以下である、モノステアリン酸ソルビタン、モノオレイン酸ソルビタン、セスキオレイン酸ソルビタン、セスキイソステアリン酸ソルビタン、モノパルミチン酸ソルビタン、トリステアリン酸ソルビタン、トリオレイン酸ソルビタン、ヤシ油脂肪酸ソルビタンから選ばれる1種又は2種以上である、前記<20>に記載の被膜の製造方法。
<25>
前記成分(c)である20℃で液体のシリコーン油は、ジメチルポリシロキサン、ジメチルシクロポリシロキサン、メチルフェニルポリシロキサン、メチルハイドロジェンポリシロキサン、高級アルコール変性オルガノポリシロキサンから選ばれる1種又は2種以上である、前記<14>又は<16>に記載の被膜の製造方法。
<26>
前記シリコーン油の組成物中の含有量は、10質量%以下、好ましくは5質量%以下、更に好ましくは1質量%以下、より更に好ましくは0.1質量%以下であり、
25℃における前記シリコーン油の動粘度は、3mm2/s以上、好ましくは4mm2/s以上、更に好ましくは5mm2/s以上であり、30mm2/s以下、好ましくは20mm2/s以下、更に好ましくは10mm2/s以下であり、
前記シリコーン油はジメチルポリシロキサンを含む、前記<25>に記載の被膜の製造方法。
<27>
前記成分(c)である20℃で液体の高級アルコールは、炭素数12~20の液状の高級アルコールであり、前記高級アルコールは、分岐脂肪酸あるいは不飽和脂肪酸の高級アルコールから選ばれる1種又は2種以上であり、好ましくはイソステアリルアルコール、及びオレイルアルコールから選ばれる1種又は2種である、前記<14>又は<16>に記載の被膜の製造方法。
前記成分(c)であるポリオールは、エチレングリコール、プロピレングリコール、1,3-プロパンジオール、1,3-ブタンジオール、ジエチレングリコール、ジプロピレングリコール、数平均分子量が1000以下のポリエチレングリコール、ポリプロピレングリコール、グリセリン、ジグリセリン、及びトリグリセリンから選ばれる1種又は2種以上であり、好ましくは数平均分子量が600以下のエチレングリコール、プロピレングリコール、1,3-ブタンジオール、ジプロピレングリコール、ポリエチレングリコール、グリセリン、及びジグリセリンから選ばれる1種又は2種以上であり、より好ましくはプロピレングリコール、1,3-ブタンジオール、グリセリン、及びジプロピレングリコールから選ばれる1種又は2種以上である、前記<1>ないし<27>のいずれか一に記載の被膜の製造方法。
<29>
前記成分(c)の20℃で液体の油及びポリオールは、炭化水素油、エステル油、シリコーン油、及び高級アルコールから選ばれる20℃で液体の油、並びにアルキレングリコール、ポリアルキレングリコール、グリセリン及びトリグリセリンから選ばれるポリオールから選択される1種又は2種以上の物質であり、好ましくは、流動パラフィン、スクワラン、ミリスチン酸イソプロピル、パルミチン酸イソプロピル、リンゴ酸ジイソステアリル、セバシン酸ジエチル、ジカプリン酸ネオペンチルグリコール、安息香酸(炭素数12~15)アルキル、イソノニルイソノナノエート、トリ(カプリル酸・カプリン酸)グリセリン、トリアシルグリセライド、HLB値が10以下である、イソステアリン酸ポリグリセリル、ジイソステアリン酸ポリグリセリル、トリイソステアリン酸ポリグリセリル、ステアリン酸ポリグリセリル、オレイン酸ポリグリセリル、セスキカプリン酸ポリグリセリル、イソステアリン酸ポリグリセリル、ジイソステアリン酸ポリグリセリル、トリイソステアリン酸ポリグリセリル、ステアリン酸ポリグリセリル、オレイン酸ポリグリセリル、セスキカプリン酸ポリグリセリル、ジメチルポリシロキサン、数平均分子量が600以下のエチレングリコール、プロピレングリコール、1,3-ブタンジオール、ジプロピレングリコール、ポリエチレングリコール、グリセリン、及びジグリセリンから選ばれる1種又は2種以上である、前記<1>ないし<28>のいずれか一に記載の被膜の製造方法。
<30>
前記成分(a)が(a1)エタノールと(a2)水を含み、成分(b)が完全鹸化ポリビニルアルコール、部分鹸化ポリビニルアルコール、ポリビニルブチラール樹脂、(アクリル酸アルキル・オクチルアミド)共重合体、オキサゾリン変性シリコーン、ポリエステル及びツエインから選ばれる1種又は2種以上であり、成分(c)が炭化水素油、エステル油、シリコーン油、及びポリオールから選ばれる1種又は2種以上である、前記<1>ないし<29>のいずれか一に記載の被膜の製造方法。
<31>
前記成分(a)が(a1)エタノールと(a2)水を含み、成分(b)がポリビニルブチラール樹脂、(アクリル酸アルキル・オクチルアミド)共重合体から選ばれる1種又は2種以上であり、成分(c)が炭化水素油、エステル油、シリコーン油、及びポリオールから選ばれる1種又は2種以上である、前記<1>ないし<29>のいずれか一に記載の被膜の製造方法。
<32>
前記組成物における前記成分(a)の含有量は、好ましくは50質量%以上98質量%以下であり、より好ましくは55質量%以上96質量%以下であり、更に好ましくは60質量%以上94質量%以下である、前記<1>ないし<31>のいずれか一に記載の被膜の製造方法。
<33>
成分(c)以外の油は、前記組成物中の含有量が、好ましくは10質量%以下であり、より好ましくは8質量%以下であり、さらに好ましくは6質量%以下である、前記<1>ないし<32>のいずれか一に記載の被膜の製造方法。
<34>
前記組成物における前記成分(b)の含有量は、好ましくは2質量%以上50質量%以下であり、より好ましくは4質量%以上45質量%以下であり、更に好ましくは6質量%以上40質量%以下である、前記<1>ないし<33>のいずれか一に記載の被膜の製造方法。
前記組成物における前記成分(c)の含有量は、好ましくは0.5質量%以上30質量%以下であり、より好ましくは1質量%以上25質量%以下であり、更に好ましくは1.5質量%以上20質量%以下である、前記<1>ないし<34>のいずれか一に記載の被膜の製造方法。
<36>
前記組成物における成分(a)の含有量が55質量%以上96質量%以下であり、成分
(b)の含有量が4質量%以上45質量%以下であり、成分(c)の含有量が1質量%以上25質量%以下であり、より好ましくは1.5質量%以上20質量%以下である、前記<1>ないし<35>のいずれか一に記載の被膜の製造方法。
<37>
前記組成物中には、前記成分(a)、前記成分(b)及び前記成分(c)のみが含まれているか、あるいは、該成分(a)、該成分(b)及び該成分(c)に加えて他の成分が含まれており、
前記他の成分は、着色顔料、体質顔料、染料、HLB値が10超の界面活性剤、UV防御剤、香料、忌避剤、酸化防止剤、安定剤、防腐剤、制汗剤、各種ビタミンである、前記<1>ないし<36>のいずれか一に記載の被膜の製造方法。
<38>
前記組成物中の前記他の成分の配合割合は、0.1質量%以上30質量%以下、好ましくは0.5質量%以上20質量%以下である、前記<37>に記載の被膜の製造方法。
<39>
前記組成物は、その粘度が、25℃において、1mPa・s以上、好ましくは10mPa・s以上、更に好ましくは50mPa・s以上であり、また、25℃において、5000mPa・s以下、好ましくは2000mPa・s以下、更に好ましくは1500mPa・s以下であり、好ましくは1mPa・s以上5000mPa・s以下であり、より好ましくは10mPa・s以上2000mPa・s以下である、更に好ましくは50mPa・s以上1500mPa・s以下である、前記<1>ないし<38>のいずれか一に記載の被膜の製造方法。
前記被膜形成対象物がヒトの皮膚の表面である、前記<1>ないし<39>のいずれか一に記載の被膜の製造方法。
<41>
前記被膜は、被膜の形成後に被膜を通して皮膚の色を視認できる透明又は半透明である、前記<40>に記載の被膜の製造方法。
<42>
前記組成物は20℃で液状であって、前記繊維は前記液状の組成物が静電スプレーされて形成される、前記<1>ないし<41>のいずれか一に記載の被膜の製造方法。
<43>
前記被膜を形成する繊維が連続繊維である、前記<1>ないし<42>のいずれか一に記載の被膜の製造方法。
<44>
前記被膜は、形成する繊維の交差する部分における結合部を含む、前記<1>ないし<43>のいずれか一に記載の被膜の製造方法。
<45>
前記被膜形成対象物がヒトの皮膚であり、静電スプレーにより皮膚に繊維を含む堆積物からなる透明又は半透明の被膜を形成するための前記<1>ないし<44>のいずれか一に記載の被膜の製造方法のための前記組成物の使用。
<46>
前記被膜形成対象物がヒトの皮膚であり、前記組成物の前記<1>ないし<44>に記載のいずれか一に記載の静電スプレーによる被膜の製造のための使用。
<47>
前記静電スプレーに用いる装置が、ヒトの手で把持可能な静電スプレー装置又はヒトの手で把持可能なスプレーノズルを備える操作部を有する静電スプレー装置である、前記<1>ないし<44>のいずれか一に記載の被膜の製造方法。
(1)噴霧用組成物の調製 噴霧用組成物の成分(a)としてエタノール(和光純薬工業(株)社製:商品名エタノール(99.5))を用いた。噴霧用組成物の成分(b)としてポリビニルブチラール(積水化学工業(株)社製:商品名S-LEC B BM-1)を用いた。噴霧用組成物の成分(c)としてグリセリン(花王(株)社製:商品名化粧品用濃グリセリン)を用いた。噴霧用組成物における配合割合は、表1及び2に示すとおりである。なお、表1及び2に示すエタノール及びグリセリンの量は、有効量であり水を含まない。
(2)静電スプレー工程
図1に示す構成を有し、図2に示す外観を有する静電スプレー装置10を用い、直接皮膚に向けて静電スプレー法を20秒間行った。静電スプレー法の条件は以下に示すとおりとした。
・印加電圧:10kV
・ノズルと皮膚との距離:100mm
・噴霧用組成物の吐出量:5ml/h
・環境:25℃、30%RH
この静電スプレーによって、皮膚の表面に繊維の堆積物からなる一枚の膜の形態である被膜が形成された。被膜は直径約4cmの円であり、質量は約3.8mgであった。上述した方法で測定された繊維の太さは660nmであった。
噴霧用組成物における成分(a)、(b)及び(c)を、以下の表1に示す条件とした以外は実施例1と同様にして、静電スプレー工程を行い、繊維の堆積物からなる被膜を得た。
噴霧用組成物における成分(c)をジイソステアリン酸ポリグリセリル―2(日清オイリオグループ株式会社製:商品名コスモール42V)に変更し、以下の表1に示す条件とした以外は実施例1と同様にして、静電スプレー工程を行い、繊維の堆積物からなる被膜を得た。
噴霧用組成物における成分(a)を実施例1に用いたエタノールに1-ブタノール(和光純薬工業(株)社製:商品名 1Butanol)を更に加えたものに変更し、成分(b)を(アクリル酸アルキル・オクチルアクリルアミド)コポリマー(Akzonobel社製:商品名DERMACRYL 79)に変更し、成分(c)をジイソステアリン酸ポリグリセリル―10(マツモトファインケミカル株式会社製:商品名マツネートMI-102)に変更し、以下の表1に示す条件とした以外は実施例1と同様にして、静電スプレー工程を行い、繊維の堆積物からなる被膜を得た。
噴霧用組成物に成分(c)を含有しない以外は、実施例1と同様にして、静電スプレー工程を行い、繊維の堆積物からなる被膜を得た。
噴霧用組成物に成分(c)を含有せず、成分(a)を実施例1に用いたエタノールに1-ブタノール( 和光純薬工業(株)社製:商品名 1Butanol)を更に加えたものに変更し、成分(b)を(アクリル酸アルキル・オクチルアクリルアミド)コポリマー(Akzonobel社製:商品名DERMACRYL 79)に変更する以外は、実施例1と同様にして、静電スプレー工程を行い、繊維の堆積物からなる被膜を得た。
実施例及び比較例で形成された被膜について、皮膚との密着性、被膜の外観を、以下の基準で評価した。その結果を表1に示す。
1:静電スプレー工程での紡糸直後から被膜がほぼすべて剥離する。
2:静電スプレー工程での紡糸直後では被膜が貼り着くが、静止状態30分後に被膜面積の50%以上が剥離する。
3:静電スプレー工程での紡糸直後では被膜が貼り着くが、静止状態30分後に被膜面積の50%未満が剥離する。
4:静電スプレー工程での紡糸後、静止状態で30分間は被膜が貼り着くが、指で平行方向に剪断力を与えると剥離する。
5:静電スプレー工程での紡糸後、静止状態で30分間は被膜が貼り着き、指で平行方向に剪断力を与えても剥離しない。
1:被膜の外観が白く見える。
2:被膜の外観が白に近い半透明に見える。
3:被膜の外観が半透明に見える。
4:被膜の外観が薄い半透明に見える。
5:被膜の外観が透明に見える。
実施例3の噴霧用組成物を用いて、実施例1の静電スプレー工程にて、以下に示す対象物(d)及び(e)に静電スプレー法を5秒間行った。付着してから30秒経過後の繊維の堆積物からなる被膜を得た。
(d)手のひら
(e)100mm×100mm×10mmのアルミ金属板表面
実施例7の噴霧用組成物を用いて、実施例12と同様にして静電スプレー工程を行い繊維の堆積物からなる被膜を得た。
噴霧用組成物における成分(a)、(b)、(c)を、以下の表1に示す条件とした以外は実施例12と同様にして、静電スプレー工程を行い、繊維の堆積物からなる被膜を得た。
比較例1の噴霧用組成物を用いて、実施例1の静電スプレー工程にて、実施例12と同様にして、静電スプレー工程を行い、繊維の堆積物からなる被膜を得た。
対象物(d)と(e)に静電スプレーして30秒間付着させた後に、被膜を剥がし、走査型電子顕微鏡(SEM)観察によって、繊維径を直接読み取ることで平均繊維径を得た。繊維の太さは、走査型電子顕微鏡(SEM)観察によって、繊維を10000倍に拡大して観察し、その二次元画像から欠陥(繊維の塊、繊維の交差部分、液滴)を除き、繊維を任意に10本選び出し、繊維の長手方向に直交する線を引き、繊維径を直接読み取ることで測定した。(1)式を満たす場合は皮膚との密着性や被膜の外観が良好となり、(2)式を満たす場合は皮膚と密着しにくく、外観も白く不自然な状態であった。結果を表2に示す。
(d)の平均繊維径>(e)の平均繊維径・・・(1)
(d)の平均繊維径≦(e)の平均繊維径・・・(2)
表3、表4に示す噴霧用組成物について、実施例1と同様にして、静電スプレー工程を行い、繊維堆積物からなる被膜を得た。実施例1と同様にして、皮膚との密着性、被膜の外観の評価と、下記の被膜の感触の評価を行った。評価結果を表3及び表4に示す。
実施例15~実施例28の被膜について、被膜形成後30分後の被膜を指で押したときの感触を以下の評価基準により評価した。評価は3名で行い、協議の結果を表3に示す。
1:べとべとする
2:ややべとつく
3:しっとりした感触
4:なめらか
5:すべるようになめらか
―:密着性の評価が5未満
表5に示す実施例26、28の被膜は、皮膚との密着性が高くしっとりした感触の被膜であるが、ややタック力があり、成分(b)が完全に乾燥せずにタック力が発現したことによる感触であると考えられる。一方、成分(b)に対して成分(c)の含有量が多い実施例27の被膜は、皮膚との密着性が高くややべとつく感触であり、成分(c)に由来するべとつきであると考えられる。
粘度の測定条件は、噴霧組成物を調製後1日25℃にて保存後、25℃において、E型粘度計(VisconicEMD、東京計器株式会社製)により、ローターNo.43を用い、回転数は1280mPa・s以上の場合は1rpm、128mPa・s以上1280mPa・s未満の場合は10rpm、128mPa・s未満の場合は100rpmとした。
表6に記載の噴霧組成物について、実施例1と同様にして、静電スプレー工程を行い、繊維堆積物からなる被膜を得た。実施例1と同様にして、被膜の外観の評価を行った。評価結果を表6に示す。
Claims (15)
- 被膜形成対象物の表面に被膜を形成する被膜の製造方法であって、
前記被膜形成対象物に組成物を直接に静電スプレーして繊維を含む堆積物からなる被膜を形成する静電スプレー工程を具備し、
前記組成物が、以下の成分(a)、成分(b)及び成分(c)を含む、被膜の製造方法。
(a)水、アルコール及びケトンから選択される1種又は2種以上の揮発性物質。
(b)被膜形成能を有するポリマー。
(c)20℃で液体の油及びポリオールから選択される1種又は2種以上を含有する液剤。 - 前記被膜は、構成する前記繊維の表面側に、前記成分(c)が存在する液剤担持被膜を有している、請求項1に記載の被膜の製造方法。
- 前記被膜あるいは噴霧用組成物における、前記成分(b)と前記成分(c)の合計量に対する前記成分(c)の質量比((c)/((b)+(c)))の値が、0.05以上0.75以下である、請求項1又は2に記載の被膜の製造方法。
- 前記成分(c)が、炭化水素油、エステル油、シリコーン油、高級アルコール、及びポリオールから選択される1種又は2種以上の物質である、請求項1ないし3のいずれか一項に記載の被膜の製造方法。
- 前記静電スプレー工程において、静電スプレー装置を用いて前記被膜形成対象物に前記組成物を静電スプレーして、繊維の堆積物からなる被膜を形成し、
前記静電スプレー装置が、前記組成物を収容する容器と、前記組成物を吐出するノズルと、前記容器中に収容されている前記組成物を前記ノズルに供給する供給装置と、前記ノズルに電圧を印加する電源とを備える請求項1ないし4のいずれか一項に記載の被膜の製造方法。 - 前記被膜形成対象物がヒトの皮膚の表面である請求項1ないし5のいずれか一項に記載の被膜の製造方法。
- 成分(a)が成分(a1)エタノール、イソプロピルアルコール、及びブチルアルコールから選ばれる1種又は2種以上と成分(a2)水とを含有する請求項1ないし6のいずれか一項に記載の被膜の製造方法。
- 成分(c)が、炭化水素油、エステル油、シリコーン油、及びポリオールから選択される1種又は2種以上である請求項1ないし7のいずれか一項に記載の被膜の製造方法。
- 前記組成物は20℃で液状であって、前記繊維は前記液状の組成物が静電スプレーされて形成される請求項1ないし8のいずれか一項に記載の被膜の製造方法。
- 前記被膜形成対象物がヒトの皮膚であって、被膜は被膜の形成後に被膜を通して皮膚の色を視認できる透明又は半透明である、請求項1ないし9のいずれか一項に記載の被膜の製造方法。
- 成分(b)が水不溶性ポリマーである請求項1ないし10のいずれか一項に記載の被膜の製造方法。
- 前記被膜を形成する繊維が連続繊維である請求項1ないし11のいずれか一項に記載の被膜の製造方法。
- 前記被膜は、形成する繊維の交差する部分における結合部を含む請求項1ないし12のいずれか一項に記載の被膜の製造方法。
- 成分(a)が(a1)エタノールと(a2)水を含み、成分(b)が完全鹸化ポリビニルアルコール、部分鹸化ポリビニルアルコール、ポリビニルブチラール樹脂、(アクリル酸アルキル・オクチルアミド)共重合体、オキサゾリン変性シリコーン、ポリエステル、及びツエインから選ばれる1種又は2種以上であり、成分(c)が炭化水素油、エステル油、シリコーン油、及びポリオールから選ばれる1種又は2種以上である請求項1ないし13のいずれか一項に記載の被膜の製造方法。
- 前記静電スプレーの装置が、ヒトの手で把持可能な静電スプレー装置又はヒトの手で把持可能なスプレーノズルを備える操作部を有する静電スプレー装置である請求項1ないし14のいずれか一項に記載の被膜の製造方法。
Priority Applications (8)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| BR112019013464-6A BR112019013464A2 (pt) | 2016-12-28 | 2017-12-27 | Método para fabricar um filme de revestimento |
| CN202011403956.8A CN112402296B (zh) | 2016-12-28 | 2017-12-27 | 覆膜的制造方法 |
| CN202210545584.5A CN114917147B (zh) | 2016-12-28 | 2017-12-27 | 覆膜的制造方法 |
| AU2017388849A AU2017388849B2 (en) | 2016-12-28 | 2017-12-27 | Method for manufacturing coating film |
| KR1020197017671A KR102020751B1 (ko) | 2016-12-28 | 2017-12-27 | 피막의 제조 방법 |
| US16/474,435 US11571371B2 (en) | 2016-12-28 | 2017-12-27 | Method for manufacturing coating film by electrostatic spraying |
| EP17886241.3A EP3563935B1 (en) | 2016-12-28 | 2017-12-27 | Method for manufacturing coating film |
| CN201780080954.XA CN110121391B (zh) | 2016-12-28 | 2017-12-27 | 覆膜的制造方法 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2016-255100 | 2016-12-28 | ||
| JP2016255100 | 2016-12-28 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2018124227A1 true WO2018124227A1 (ja) | 2018-07-05 |
Family
ID=62709348
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2017/047079 WO2018124227A1 (ja) | 2016-12-28 | 2017-12-27 | 被膜の製造方法 |
Country Status (9)
| Country | Link |
|---|---|
| US (1) | US11571371B2 (ja) |
| EP (1) | EP3563935B1 (ja) |
| JP (2) | JP6453434B2 (ja) |
| KR (1) | KR102020751B1 (ja) |
| CN (2) | CN112402296B (ja) |
| AU (1) | AU2017388849B2 (ja) |
| BR (1) | BR112019013464A2 (ja) |
| TW (1) | TWI682783B (ja) |
| WO (1) | WO2018124227A1 (ja) |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2020033266A1 (en) * | 2018-08-09 | 2020-02-13 | Kao Corporation | Method for producing coating |
| WO2021085394A1 (ja) | 2019-10-28 | 2021-05-06 | 花王株式会社 | 繊維堆積体の製造方法、膜の製造方法及び膜の付着方法 |
| WO2021085393A1 (ja) | 2019-10-28 | 2021-05-06 | 花王株式会社 | 繊維堆積体の製造方法、膜の製造方法及び膜の付着方法 |
| CN113165008A (zh) * | 2018-12-07 | 2021-07-23 | 花王株式会社 | 涂膜的形成方法 |
| CN113165011A (zh) * | 2018-12-07 | 2021-07-23 | 花王株式会社 | 涂膜的形成方法 |
Families Citing this family (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP7169813B2 (ja) * | 2018-08-09 | 2022-11-11 | 花王株式会社 | 被膜の製造方法 |
| KR102655471B1 (ko) * | 2018-11-26 | 2024-04-05 | 카오카부시키가이샤 | 피부용 피막 |
| KR102407900B1 (ko) * | 2019-05-31 | 2022-06-10 | 카오카부시키가이샤 | 피막 형성 조성물 및 극세 단섬유 |
| JP6894552B2 (ja) * | 2019-05-31 | 2021-06-30 | 花王株式会社 | 極細短繊維 |
| JP6903794B2 (ja) * | 2019-05-31 | 2021-07-14 | 花王株式会社 | 皮膜形成組成物 |
| JP6903793B2 (ja) * | 2019-05-31 | 2021-07-14 | 花王株式会社 | 皮膜形成組成物 |
| WO2021025118A1 (ja) * | 2019-08-07 | 2021-02-11 | 花王株式会社 | 被膜形成用組成物 |
| EP4101436A1 (en) * | 2021-06-11 | 2022-12-14 | IONIQ Skincare GmbH & Co. KG | Electrostatically sprayable skin caring formulation |
| EP4313320A1 (en) * | 2021-07-19 | 2024-02-07 | Coty Inc. | Antiperspirant composition |
| TW202327546A (zh) * | 2021-08-26 | 2023-07-16 | 日商花王股份有限公司 | 覆膜之製造方法 |
| TW202345771A (zh) * | 2022-04-04 | 2023-12-01 | 日商花王股份有限公司 | 覆膜形成用組成物 |
| CN115317655A (zh) * | 2022-08-11 | 2022-11-11 | 百达联康生物科技(深圳)有限公司 | 一种纤维膜及其制备方法 |
| CN115418796A (zh) * | 2022-09-14 | 2022-12-02 | 百达联康生物科技(深圳)有限公司 | 一种抗菌纤维膜及其制备方法 |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH07173031A (ja) * | 1991-07-15 | 1995-07-11 | Unilever Nv | 化粧品送出システム |
| WO1998003267A1 (en) | 1996-07-23 | 1998-01-29 | Electrosols Ltd. | A dispensing device and method for forming material |
| US6531142B1 (en) | 1999-08-18 | 2003-03-11 | The Procter & Gamble Company | Stable, electrostatically sprayable topical compositions |
| JP2005523981A (ja) * | 2002-04-30 | 2005-08-11 | ハイドロマー インコーポレイテッド | 親水性多用途のためのコーティング組成物 |
| JP2007526224A (ja) * | 2003-06-19 | 2007-09-13 | ザ プロクター アンド ギャンブル カンパニー | シリコーン中ポリオール型エマルション |
| JP2008179629A (ja) | 2006-12-27 | 2008-08-07 | Snt Co | 化粧用シート |
Family Cites Families (16)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB9224191D0 (en) | 1992-11-18 | 1993-01-06 | Unilever Plc | Cosmetic delivery system |
| US6252129B1 (en) * | 1996-07-23 | 2001-06-26 | Electrosols, Ltd. | Dispensing device and method for forming material |
| US20080119772A1 (en) * | 2001-01-11 | 2008-05-22 | Ronald Alan Coffee | Dispensing device and method for forming material |
| US6461626B1 (en) * | 1999-08-18 | 2002-10-08 | The Procter & Gamble Company | Wear resistant topical compositions having improved feel |
| US6514504B1 (en) | 1999-08-18 | 2003-02-04 | The Procter & Gamble Company | Discontinuous films from skin care compositions |
| US6558682B2 (en) | 1999-08-18 | 2003-05-06 | The Procter & Gamble Company | Discontinuous films from skin care compositions |
| FR2816831B1 (fr) * | 2000-11-23 | 2002-12-27 | Oreal | Composition cosmetique a phase continue lipophile contenant des fibres |
| US20020187181A1 (en) | 2001-05-14 | 2002-12-12 | 3M Innovative Properties Company | System for delivering cosmetics and pharmaceuticals |
| WO2005075090A1 (en) * | 2004-02-09 | 2005-08-18 | Matsushita Electric Works, Ltd. | Electrostatic spraying device |
| EP1924328A1 (en) * | 2005-09-09 | 2008-05-28 | The Procter and Gamble Company | Solid skin care composition comprising multiple layers based on water-in-oil emulsions |
| JP4777760B2 (ja) * | 2005-12-01 | 2011-09-21 | 株式会社Snt | 網目状構造体を含む複合構造体 |
| FR2910295B3 (fr) * | 2006-12-20 | 2009-09-18 | Oreal | Procede de maquillage des matieres keratiniques et kit pour la mise en oeuvre d'un tel procede |
| WO2011143131A1 (en) * | 2010-05-11 | 2011-11-17 | Shaosheng Dong | Film forming personal care compositions and methods |
| EP3366270B1 (en) | 2015-10-19 | 2021-02-17 | Kao Corporation | Method for producing coating film |
| JP2018177725A (ja) | 2017-04-18 | 2018-11-15 | 花王株式会社 | 外用薬 |
| JP2018177724A (ja) | 2017-04-18 | 2018-11-15 | 花王株式会社 | 外用薬 |
-
2017
- 2017-12-27 EP EP17886241.3A patent/EP3563935B1/en active Active
- 2017-12-27 AU AU2017388849A patent/AU2017388849B2/en not_active Ceased
- 2017-12-27 WO PCT/JP2017/047079 patent/WO2018124227A1/ja unknown
- 2017-12-27 CN CN202011403956.8A patent/CN112402296B/zh active Active
- 2017-12-27 TW TW106146363A patent/TWI682783B/zh active
- 2017-12-27 BR BR112019013464-6A patent/BR112019013464A2/pt not_active Application Discontinuation
- 2017-12-27 US US16/474,435 patent/US11571371B2/en active Active
- 2017-12-27 KR KR1020197017671A patent/KR102020751B1/ko active IP Right Grant
- 2017-12-27 CN CN201780080954.XA patent/CN110121391B/zh active Active
- 2017-12-27 JP JP2017252237A patent/JP6453434B2/ja active Active
-
2018
- 2018-12-12 JP JP2018232325A patent/JP2019038856A/ja active Pending
Patent Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH07173031A (ja) * | 1991-07-15 | 1995-07-11 | Unilever Nv | 化粧品送出システム |
| WO1998003267A1 (en) | 1996-07-23 | 1998-01-29 | Electrosols Ltd. | A dispensing device and method for forming material |
| US6531142B1 (en) | 1999-08-18 | 2003-03-11 | The Procter & Gamble Company | Stable, electrostatically sprayable topical compositions |
| JP2006104211A (ja) * | 1999-08-18 | 2006-04-20 | Procter & Gamble Co | 皮膚の処理方法 |
| JP2005523981A (ja) * | 2002-04-30 | 2005-08-11 | ハイドロマー インコーポレイテッド | 親水性多用途のためのコーティング組成物 |
| JP2007526224A (ja) * | 2003-06-19 | 2007-09-13 | ザ プロクター アンド ギャンブル カンパニー | シリコーン中ポリオール型エマルション |
| JP2008179629A (ja) | 2006-12-27 | 2008-08-07 | Snt Co | 化粧用シート |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP3563935A4 |
Cited By (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2020033266A1 (en) * | 2018-08-09 | 2020-02-13 | Kao Corporation | Method for producing coating |
| US11583487B2 (en) | 2018-08-09 | 2023-02-21 | Kao Corporation | Method for producing coating |
| CN113165008A (zh) * | 2018-12-07 | 2021-07-23 | 花王株式会社 | 涂膜的形成方法 |
| CN113165011A (zh) * | 2018-12-07 | 2021-07-23 | 花王株式会社 | 涂膜的形成方法 |
| CN113165008B (zh) * | 2018-12-07 | 2022-12-23 | 花王株式会社 | 涂膜的形成方法 |
| CN113165011B (zh) * | 2018-12-07 | 2023-01-03 | 花王株式会社 | 涂膜的形成方法 |
| US11866600B2 (en) | 2018-12-07 | 2024-01-09 | Kao Corporation | Method for forming coating film |
| WO2021085394A1 (ja) | 2019-10-28 | 2021-05-06 | 花王株式会社 | 繊維堆積体の製造方法、膜の製造方法及び膜の付着方法 |
| WO2021085393A1 (ja) | 2019-10-28 | 2021-05-06 | 花王株式会社 | 繊維堆積体の製造方法、膜の製造方法及び膜の付着方法 |
| US11773512B2 (en) | 2019-10-28 | 2023-10-03 | Kao Corporation | Fiber deposit production method, membrane production method, and membrane adhesion method |
Also Published As
| Publication number | Publication date |
|---|---|
| US11571371B2 (en) | 2023-02-07 |
| TW201841604A (zh) | 2018-12-01 |
| CN110121391B (zh) | 2022-05-03 |
| EP3563935A4 (en) | 2020-08-19 |
| JP6453434B2 (ja) | 2019-01-16 |
| AU2017388849B2 (en) | 2022-02-17 |
| JP2018108991A (ja) | 2018-07-12 |
| US20190343731A1 (en) | 2019-11-14 |
| BR112019013464A2 (pt) | 2020-01-07 |
| KR20190078655A (ko) | 2019-07-04 |
| EP3563935B1 (en) | 2021-09-08 |
| KR102020751B1 (ko) | 2019-09-11 |
| CN112402296A (zh) | 2021-02-26 |
| CN112402296B (zh) | 2022-03-11 |
| AU2017388849A1 (en) | 2019-07-11 |
| JP2019038856A (ja) | 2019-03-14 |
| CN110121391A (zh) | 2019-08-13 |
| EP3563935A1 (en) | 2019-11-06 |
| CN114917147A (zh) | 2022-08-19 |
| TWI682783B (zh) | 2020-01-21 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6453434B2 (ja) | 被膜の製造方法 | |
| JP6203362B2 (ja) | 被膜の製造方法 | |
| JP6203363B2 (ja) | 被膜の製造方法 | |
| WO2018194086A1 (ja) | 紫外線防御被膜の製造方法 | |
| WO2018194130A1 (ja) | 被膜形成用組成物 | |
| WO2018194085A1 (ja) | 化粧被膜の製造方法 | |
| WO2018194087A1 (ja) | 紫外線防御被膜の製造方法 | |
| WO2023027151A1 (ja) | 被膜の製造方法 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 17886241 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 20197017671 Country of ref document: KR Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01A Ref document number: 112019013464 Country of ref document: BR |
|
| ENP | Entry into the national phase |
Ref document number: 2017388849 Country of ref document: AU Date of ref document: 20171227 Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 2017886241 Country of ref document: EP Effective date: 20190729 |
|
| ENP | Entry into the national phase |
Ref document number: 112019013464 Country of ref document: BR Kind code of ref document: A2 Effective date: 20190627 |