WO2016047708A1 - タイヤ - Google Patents
タイヤ Download PDFInfo
- Publication number
- WO2016047708A1 WO2016047708A1 PCT/JP2015/076980 JP2015076980W WO2016047708A1 WO 2016047708 A1 WO2016047708 A1 WO 2016047708A1 JP 2015076980 W JP2015076980 W JP 2015076980W WO 2016047708 A1 WO2016047708 A1 WO 2016047708A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- polyamide
- tire
- thermoplastic elastomer
- based thermoplastic
- acid
- Prior art date
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G69/00—Macromolecular compounds obtained by reactions forming a carboxylic amide link in the main chain of the macromolecule
- C08G69/40—Polyamides containing oxygen in the form of ether groups
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B60—VEHICLES IN GENERAL
- B60C—VEHICLE TYRES; TYRE INFLATION; TYRE CHANGING; CONNECTING VALVES TO INFLATABLE ELASTIC BODIES IN GENERAL; DEVICES OR ARRANGEMENTS RELATED TO TYRES
- B60C1/00—Tyres characterised by the chemical composition or the physical arrangement or mixture of the composition
- B60C1/0041—Compositions of the carcass layers
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G69/00—Macromolecular compounds obtained by reactions forming a carboxylic amide link in the main chain of the macromolecule
- C08G69/02—Polyamides derived from amino-carboxylic acids or from polyamines and polycarboxylic acids
- C08G69/36—Polyamides derived from amino-carboxylic acids or from polyamines and polycarboxylic acids derived from amino acids, polyamines and polycarboxylic acids
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G2380/00—Tyres
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G69/00—Macromolecular compounds obtained by reactions forming a carboxylic amide link in the main chain of the macromolecule
- C08G69/02—Polyamides derived from amino-carboxylic acids or from polyamines and polycarboxylic acids
- C08G69/08—Polyamides derived from amino-carboxylic acids or from polyamines and polycarboxylic acids derived from amino-carboxylic acids
- C08G69/14—Lactams
Definitions
- the present invention relates to a tire mounted on a rim, and particularly relates to a tire in which at least a part of a tire case is formed of a resin material.
- thermoplastic polymer materials such as thermoplastic elastomers and thermoplastic resin materials
- thermoplastic polymer materials have many advantages from the viewpoint of improving productivity, such as being capable of injection molding.
- a tire manufactured using a polyamide-based thermoplastic elastomer as the thermoplastic polymer material has been proposed (see Japanese Patent Application Laid-Open No. 2012-45790).
- thermoplastic polymer materials are easier to manufacture and lower in cost than conventional rubber tires, but they are more efficient than other conventional rubber tires while increasing manufacturing efficiency. Therefore, it is required to achieve performance that is comparable to that of tires (required characteristics of tires).
- the required characteristics of the tire include, for example, low rolling resistance (low loss), heat resistance, and excellent rim assemblage (the rim can be assembled and the internal air is difficult to leak when the rim is assembled. ) And the like.
- the characteristics required to increase the manufacturing efficiency of the tire include, for example, a reaction rate (reactivity) when synthesizing the polymer material, and excellent injection moldability when the polymer material is injection molded. Can be mentioned.
- a tire using a thermoplastic polymer material is required to have a balance of high production efficiency, low loss, heat resistance, and excellent rim assembly.
- an embodiment of the present invention aims to provide a tire that is formed using a resin material and that is excellent in balance between manufacturing efficiency, low loss, heat resistance, and rim assembly. .
- the polyamide-based thermoplastic elastomer includes a hard segment and a soft segment composed of structural units derived from polypropylene glycol, And a bonded portion composed of a structural unit derived from a dicarboxylic acid having 6 to 20 carbon atoms bonded to two or more segments, and the weight average molecular weight of the polyamide-based thermoplastic elastomer is 20,000 to 250,000
- a tire that is:
- a tire that is formed using a resin material and that is excellent in production efficiency, low loss, heat resistance, and rim assembly.
- FIG. 1 is a perspective view showing a partial cross section of a tire according to an embodiment of the present invention. It is sectional drawing of the bead part with which the rim
- the tire according to an embodiment of the present invention is formed of a resin material containing a polyamide-based thermoplastic elastomer and has an annular tire skeleton.
- the polyamide-based thermoplastic elastomer is composed of a hard segment, a soft segment composed of a structural unit derived from polypropylene glycol, and a structural unit derived from a dicarboxylic acid having 6 to 20 carbon atoms by combining two or more segments.
- the polyamide thermoplastic elastomer has a weight average molecular weight of 20,000 or more and 250,000 or less.
- the tire skeleton includes a polyamide-based thermoplastic elastomer that satisfies the above-mentioned conditions for all of the structural unit of the soft segment, the structural unit of the bonding portion, and the weight average molecular weight.
- Excellent balance between heat resistance, heat resistance and rim assembly Specifically, for example, in one embodiment of the present invention, while maintaining the production efficiency, heat resistance, and rim assembly property as compared with the case where the soft segment includes a structural unit derived from polytetramethylene ether glycol (PTMG). Especially, the low-loss property can be improved.
- PTMG polytetramethylene ether glycol
- the production efficiency is improved while maintaining low loss as compared with the case where the number of carbon atoms of the dicarboxylic acid forming the bonding portion is out of the above range, and in addition, heat resistance and rim assembly properties are improved. An excellent and well-balanced tire can be obtained.
- the reason why the production efficiency is improved when the carbon number of the dicarboxylic acid is in the above range is not clear, but by using the dicarboxylic acid having the carbon number, the reaction rate (reactivity) during the synthesis of the polyamide-based thermoplastic elastomer is increased. It is estimated that the manufacturing efficiency is improved as a result.
- a tire having excellent balance of manufacturing efficiency, low loss property, heat resistance, and rim assembly property compared to a case where the weight average molecular weight of the polyamide-based thermoplastic elastomer is out of the above range, a tire having excellent balance of manufacturing efficiency, low loss property, heat resistance, and rim assembly property. Is obtained.
- the production efficiency is particularly excellent in injection moldability and low loss and rim assembly property, and the weight average molecular weight is high.
- the injection moldability, rim assembly property, and heat resistance are excellent.
- the tire to which the polyamide-based thermoplastic elastomer is applied is formed using a resin material, and the manufacturing efficiency, low loss property, heat resistance, and rim assembly property are excellent in a well-balanced manner.
- the tire has a tire skeleton using a resin material containing a polyamide-based thermoplastic elastomer.
- the polyamide-based thermoplastic elastomer comprises at least a hard segment, a soft segment composed of a structural unit derived from polypropylene glycol, and a structural unit derived from a dicarboxylic acid having 6 to 20 carbon atoms by combining two or more segments. And a weight average molecular weight of 20,000 or more and 250,000 or less.
- the “joining part” is a joining part that joins two or more segments.
- the connecting portion include a connecting portion between a hard segment and a soft segment, a connecting portion between hard segments, and a connecting portion between soft segments.
- the binding part include a part bound by a chain extender described later.
- the resin material may contain a thermoplastic elastomer other than the thermoplastic elastomer and an arbitrary component.
- “resin” is a concept including a thermoplastic resin and a thermosetting resin, but does not include natural rubber.
- a numerical range expressed using “to” means a range including numerical values described before and after “to” as a lower limit value and an upper limit value.
- the amount of each component in the composition is the sum of the plurality of substances present in the composition unless there is a specific indication when there are a plurality of substances corresponding to each component in the composition. Means quantity.
- polyamide-based thermoplastic elastomer means a copolymer having a crystalline polymer having a high melting point and a non-crystalline polymer having a low glass transition temperature. Which has an amide bond (—CONH—) in the main chain of the polymer constituting the hard segment.
- polyamide-based thermoplastic elastomer As the polyamide-based thermoplastic elastomer, at least polyamide is a hard segment that is crystalline and has a high melting point, polypropylene glycol is amorphous and a soft segment that has a low glass transition temperature, and is a dicarboxylic acid having 6 to 20 carbon atoms. Examples thereof include materials in which a chain extender that is an acid constitutes a bond.
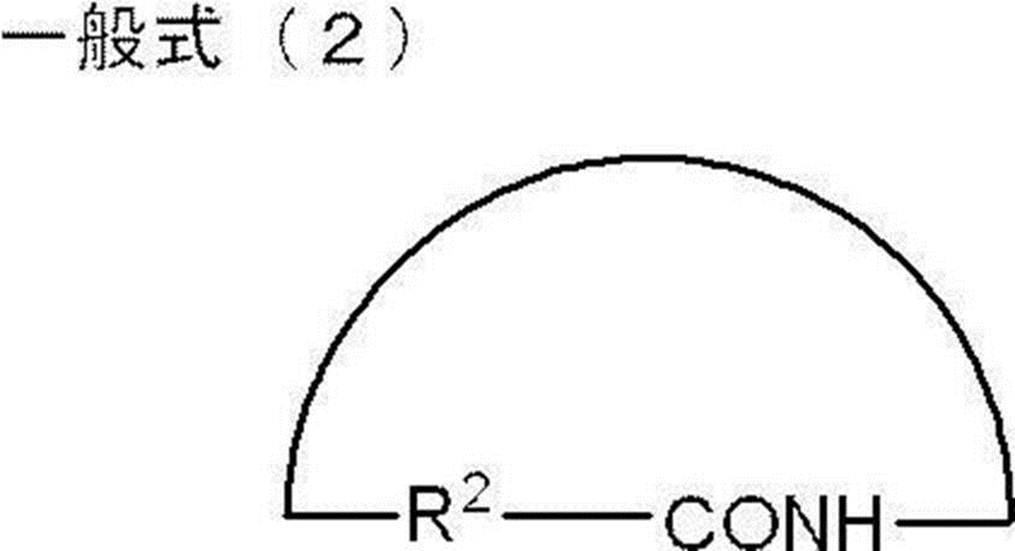
- polyamide forming the hard segment examples include polyamides synthesized using monomers represented by the following general formula (1) or general formula (2).
- R 1 represents a hydrocarbon molecular chain having 2 to 20 carbon atoms or an alkylene group having 2 to 20 carbon atoms.
- R 2 represents a hydrocarbon molecular chain having 3 to 20 carbon atoms or an alkylene group having 3 to 20 carbon atoms.
- R 1 is preferably a hydrocarbon molecular chain having 3 to 18 carbon atoms or an alkylene group having 3 to 18 carbon atoms, and a hydrocarbon molecular chain having 4 to 15 carbon atoms or 4 carbon atoms.
- An alkylene group having 15 to 15 carbon atoms is more preferable, and a molecular chain of a hydrocarbon having 10 to 15 carbon atoms or an alkylene group having 10 to 15 carbon atoms is particularly preferable.
- R 2 is preferably a hydrocarbon molecular chain having 3 to 18 carbon atoms or an alkylene group having 3 to 18 carbon atoms, and a hydrocarbon molecular chain having 4 to 15 carbon atoms or carbon.
- alkylene group having 4 to 15 carbon atoms is more preferable, and a hydrocarbon molecular chain having 10 to 15 carbon atoms or an alkylene group having 10 to 15 carbon atoms is particularly preferable.
- the monomer represented by the general formula (1) or the general formula (2) include ⁇ -aminocarboxylic acid and lactam.
- the polyamide forming the hard segment include polycondensates of these ⁇ -aminocarboxylic acids and lactams, and copolycondensation polymers of diamines and dicarboxylic acids.
- Examples of the ⁇ -aminocarboxylic acid include 6-aminocaproic acid, 7-aminoheptanoic acid, 8-aminooctanoic acid, 10-aminocapric acid, 11-aminoundecanoic acid, and 12-aminododecanoic acid.
- Examples of the lactam include aliphatic lactams having 5 to 20 carbon atoms such as lauryl lactam, ⁇ -caprolactam, undecane lactam, ⁇ -enantolactam, and 2-pyrrolidone.
- diamine examples include ethylenediamine, trimethylenediamine, tetramethylenediamine, hexamethylenediamine, heptamethylenediamine, octamethylenediamine, nonamethylenediamine, decamethylenediamine, undecamethylenediamine, dodecamethylenediamine, 2,2, Examples thereof include diamine compounds such as aliphatic diamines having 2 to 20 carbon atoms such as 4-trimethylhexamethylenediamine, 2,4,4-trimethylhexamethylenediamine, 3-methylpentamethylenediamine, and metaxylenediamine.
- the dicarboxylic acid can be represented by HOOC- (R 3 ) m-COOH (R 3 : a hydrocarbon molecular chain having 3 to 20 carbon atoms, m: 0 or 1).
- R 3 a hydrocarbon molecular chain having 3 to 20 carbon atoms, m: 0 or 1.
- oxalic acid, succinic acid And aliphatic dicarboxylic acids having 2 to 22 carbon atoms such as glutaric acid, adipic acid, pimelic acid, suberic acid, azelaic acid, sebacic acid and dodecanedioic acid.
- polyamide (polyamide 6) obtained by ring-opening polycondensation of ⁇ -caprolactam
- polyamide (polyamide 11) obtained by ring-opening polycondensation of undecane lactam
- polyamide (polyamide) obtained by ring-opening polycondensation of lauryl lactam 12
- polycondensation polyamide (polyamide 66) of diamine and dibasic acid polyamide (polyamide 612) obtained by polymerizing hexamethylenediamine and dodecanedicarboxylic acid
- polyamide (amide MX) having metaxylenediamine as a structural unit can be used.
- the polyamide 6 can be represented by, for example, ⁇ CO— (CH 2 ) 5 —NH ⁇ n (n represents an arbitrary number of repeating units). For example, n is preferably 2 to 100, and 3 to 50 Is more preferable.
- the polyamide 11 can be represented by, for example, ⁇ CO— (CH 2 ) 10 —NH ⁇ n (n represents an arbitrary number of repeating units). For example, n is preferably 2 to 100, and 3 to 50 Is more preferable.
- the polyamide 12 can be represented by, for example, ⁇ CO— (CH 2 ) 11 —NH ⁇ n (n represents an arbitrary number of repeating units). For example, n is preferably 2 to 100, and 3 to 50 Is more preferable.
- the polyamide 66 can be represented by, for example, ⁇ CO (CH 2 ) 4 CONH (CH 2 ) 6 NH ⁇ n (n represents an arbitrary number of repeating units). For example, n is preferably 2 to 100 3 to 50 are more preferable.
- the polyamide 612 can be represented by, for example, ⁇ CO (CH 2 ) 12 CONH (CH 2 ) 6 NH ⁇ n (n represents an arbitrary number of repeating units). For example, n is preferably 2 to 100 3 to 50 are more preferable.
- the amide MX having meta-xylenediamine as a structural unit can be represented, for example, by the following structural unit (A-1) [in (A-1), n represents an arbitrary number of repeating units], for example, n is preferably 2 to 100, and more preferably 3 to 50.
- the polyamide-based thermoplastic elastomer preferably has a polyamide (polyamide 12) having a unit structure represented by — [CO— (CH 2 ) 11 —NH] — as a hard segment.
- the polyamide 12 can be obtained by ring-opening polycondensation of lauryl lactam or polycondensation of 12-aminododecanoic acid.
- the polyamide-based thermoplastic elastomer is particularly represented by — [CO— (CH 2 ) 11 —NH] — as a hard segment from the viewpoint of a balance between low loss, heat resistance, and excellent rim assembly.
- a polyamide having a unit structure (polyamide 12), a polyamide having a unit structure represented by — [CO— (CH 2 ) 5 —NH] — (polyamide 6), and — [CO (CH 2 ) 12 CONH ( It is preferable to have at least one of polyamides (polyamide 612) having a unit structure represented by CH 2 ) 6 NH] —.
- the number average molecular weight of the polymer (polyamide) forming the hard segment is preferably from 300 to 15000 from the viewpoint of melt moldability.
- the soft segment is composed of structural units derived from polypropylene glycol. That is, the soft segment is formed of only polypropylene glycol.
- the polymer (polypropylene glycol) that forms the soft segment include polypropylene glycol derivatives in which both ends are hydroxyl groups, as well as polypropylene glycol derivatives having functional groups introduced at the ends.
- the functional group at the terminal of the polypropylene glycol derivative only needs to react with the carboxy group of the dicarboxylic acid, which is a chain extender, so that the polypropylene glycol and the dicarboxylic acid are bonded, and examples thereof include an amino group.
- examples of the polypropylene glycol derivative include polyoxypropylene diamine obtained by reacting ammonia or the like with the end of polypropylene glycol having both ends being hydroxyl groups.
- the polypropylene glycol whose both ends are hydroxyl groups and the polypropylene glycol derivative may be collectively referred to as “polypropylene glycol”.
- the number average molecular weight of polypropylene glycol forming the soft segment is preferably 200 or more and 6000 or less, more preferably 400 or more and 4000 or less, and particularly preferably 650 or more and 2000 or less, from the viewpoint of toughness and low temperature flexibility.
- Polypropylene glycol can be produced by a known method. Specifically, for example, polypropylene glycol can be obtained by anion ring-opening polymerization reaction of propylene oxide. Moreover, you may form a soft segment using commercially available polypropylene glycol.
- the combination of the hard segment and the soft segment the combination of the hard segment and the soft segment mentioned above can be given.
- a combination of ring-opening polycondensate of lauryl lactam / polypropylene glycol and a combination of polycondensate of aminododecanoic acid / polypropylene glycol are preferable.
- the bonding portion is composed of a structural unit derived from a dicarboxylic acid having 6 to 20 carbon atoms. That is, the bond is formed only with a chain extender that is a dicarboxylic acid having 6 to 20 carbon atoms.
- the chain extender that forms the bonding portion may be a dicarboxylic acid having 6 to 20 carbon atoms. Specifically, for example, an aliphatic dicarboxylic acid, an alicyclic dicarboxylic acid, an aromatic dicarboxylic acid, and the like. These may be used alone or in combination of two or more.
- the chain extender is preferably an aliphatic dicarboxylic acid, more preferably a linear aliphatic dicarboxylic acid, still more preferably a linear aliphatic dicarboxylic acid having 8 to 20 carbon atoms, and more preferably 10 to 18 carbon atoms.
- the straight chain aliphatic dicarboxylic acid is particularly preferable, and a straight chain aliphatic dicarboxylic acid having 12 to 18 carbon atoms is more preferable.
- dicarboxylic acid examples include adipic acid (hexanedioic acid), 1,10-decanedicarboxylic acid (dodecanedioic acid), pimelic acid (heptanedioic acid), suberic acid (octanedioic acid), azelaic acid (nonane).
- the weight average molecular weight of the polyamide-based thermoplastic elastomer contained in the resin material is 20,000 or more and 250,000 or less. If the weight average molecular weight of the polyamide-based thermoplastic elastomer is less than 20,000, the rim assembly property is lowered. Further, if the weight average molecular weight of the polyamide-based thermoplastic elastomer exceeds 250,000, the melt viscosity becomes high, and there is a risk of insufficient filling when forming a tire skeleton, so the molding temperature, mold temperature May need to be high. When the molding temperature and the mold temperature are increased in order to prevent insufficient filling, the cycle time becomes longer and the productivity (manufacturing efficiency) is inferior.
- the weight average molecular weight of the polyamide-based thermoplastic elastomer is preferably from 50,000 to 200,000, more preferably from 60,000 to 200,000, further preferably from 80,000 to 160,000, 000 to 150,000 is particularly preferable.
- the weight average molecular weight of the polyamide-based thermoplastic elastomer may be 100,000 or more and 180,000 or less, particularly 150,000 or more and 180,000 or less, particularly from the viewpoint of heat resistance and rim assembly. It may be 170,000 or more and 180,000 or less.
- the weight average molecular weight of the polyamide-based thermoplastic elastomer can be measured by gel permeation chromatography (GPC).
- GPC gel permeation chromatography
- HLC-8320GPC EcoSEC manufactured by Tosoh Corporation may be used. Can be used.
- the ratio (x / y) of the mass (x) of the hard segment to the mass (y) of the soft segment is, for example, 40/60 to 90/10, and low loss property, From the viewpoint of achieving a balance between heat resistance and rim assembly, 54/46 to 90/10 is preferable, 54/46 to 88/12 is more preferable, 55/45 to 85/15 is more preferable, and 60/40 ⁇ 75 / 25 is particularly preferred.
- x represents the mass of the hard segment contained in the polyamide-based thermoplastic elastomer
- y represents the mass of the soft segment contained in the polyamide-based thermoplastic elastomer.
- X / y” indicates “mass of hard segment (x) / mass of soft segment (y)”.
- the content of the hard segment in the polyamide thermoplastic elastomer is preferably 5 to 95% by mass, more preferably 10 to 90% by mass, and particularly preferably 15 to 90% by mass with respect to the total amount of the polyamide thermoplastic elastomer. .
- the content of the soft segment in the polyamide thermoplastic elastomer is preferably 10 to 95% by mass, more preferably 10 to 90% by mass, based on the total amount of the polyamide thermoplastic elastomer.
- the content of the chain extender is set so that the terminal functional group (for example, hydroxyl group or amino group) of the polypropylene glycol constituting the soft segment and the carboxyl group of the chain extender are approximately equimolar. It is preferable.
- the polyamide-based thermoplastic elastomer can be synthesized by copolymerizing the polymer forming the hard segment and the polymer forming the soft segment by a known method.
- the polyamide-based thermoplastic elastomer includes a monomer constituting a hard segment (for example, ⁇ -aminocarboxylic acid such as 12-aminododecanoic acid or lactam such as lauryl lactam) and a chain extender (for example, adipic acid or Decanedicarboxylic acid) is polymerized in a container, and then a polymer (polypropylene glycol) constituting a soft segment is added and further polymerized.
- a monomer constituting a hard segment for example, ⁇ -aminocarboxylic acid such as 12-aminododecanoic acid or lactam such as lauryl lactam
- a chain extender for example, adipic acid or Decanedicarboxylic acid
- ⁇ -aminocarboxylic acid when used as the monomer constituting the hard segment, it can be synthesized by performing atmospheric pressure melt polymerization or atmospheric pressure melt polymerization, and further under reduced pressure melt polymerization.
- lactam When used as the monomer constituting the hard segment, an appropriate amount of water can coexist, and from melt polymerization under pressure of 0.1 to 5 MPa, followed by normal pressure melt polymerization and / or reduced pressure melt polymerization.
- These synthesis reactions can be carried out either batchwise or continuously.
- a batch-type reaction tank, a single tank type or multi-tank type continuous reaction apparatus, a tubular continuous reaction apparatus, or the like may be used alone or in appropriate combination.
- the polymerization temperature is preferably 150 to 300 ° C, more preferably 160 to 280 ° C.
- the polymerization time can be appropriately determined depending on the relationship between the polymerization average molecular weight of the polyamide-based thermoplastic elastomer to be synthesized and the polymerization temperature. For example, it is preferably 0.5 to 30 hours, and more preferably 0.5 to 20 hours.
- monoamines or diamines such as laurylamine, stearylamine, hexamethylenediamine, and metaxylylenediamine for the purpose of adjusting the molecular weight and stabilizing the melt viscosity at the time of molding as necessary.
- An additive such as acetic acid, benzoic acid, stearic acid, adipic acid, sebacic acid, dodecanedioic acid or the like monocarboxylic acid or dicarboxylic acid; These additives can be appropriately selected in relation to the molecular weight and viscosity of the resulting polyamide-based thermoplastic elastomer within a range that does not adversely affect the effects of the present invention.
- a catalyst can be used as necessary.
- the catalyst includes at least one selected from the group consisting of P, Ti, Ge, Zn, Fe, Sn, Mn, Co, Zr, V, Ir, La, Ce, Li, Ca, and Hf.
- Compounds include inorganic phosphorus compounds, organic titanium compounds, organic zirconium compounds, and organic tin compounds.
- examples of the inorganic phosphorus compound include phosphoric acid, pyrophosphoric acid, polyphosphoric acid, phosphorous acid, hypophosphorous acid and other phosphorus-containing acids, phosphorus-containing acid alkali metal salts, and phosphorus-containing acid alkaline earths. A metal salt etc.
- Examples of the organic titanium compound include titanium alkoxide [titanium tetrabutoxide, titanium tetraisopropoxide, and the like].
- Examples of the organic zirconium compound include zirconium alkoxide (zirconium tetrabutoxide (also referred to as “Zr (OBu) 4 ” or “Zr (OC 4 H 8 ) 4 )”).
- Examples of organotin compounds include distannoxane compounds [1-hydroxy-3-isothiocyanate-1,1,3,3-tetrabutyl distanoxane, etc.], tin acetate, dibutyltin dilaurate, butyltin hydroxide oxide hydrate, and the like. Can be mentioned.
- the catalyst addition amount and the catalyst addition timing are not particularly limited as long as the target product can be obtained quickly.
- polyamide-based thermoplastic elastomer examples include a lauryl lactam ring-opening polycondensate / polypropylene glycol / adipic acid combination, a lauryl lactam ring-opening polycondensate / polypropylene glycol / decane dicarboxylic acid combination, and aminododecanoic acid.
- a combination of a polycondensate / polypropylene glycol / adipic acid and a polycondensate of aminododecanoic acid / polypropylene glycol / decane dicarboxylic acid are preferred.
- polyamide-type thermoplastic elastomer what combined the preferable aspect mentioned above about the combination of a structural unit, the structural ratio, molecular weight, etc. can be used.
- additives such as rubber, various fillers (for example, silica, calcium carbonate, clay), anti-aging agents, oils, plasticizers, colorants, weathering agents, and reinforcing materials are added to the resin material as desired. You may make it contain.
- the content of the additive in the resin material (tire frame) is not particularly limited, and can be appropriately used as long as the effects of the present invention are not impaired.
- the content of the resin component in the resin material is preferably 50% by mass or more, and more preferably 90% by mass or more based on the total amount of the resin material.
- the content of the resin component in the resin material is the balance obtained by subtracting the total content of various additives from the total amount of the resin component.
- the tire frame body uses the above-mentioned resin material.
- the melting point (or softening point) of the resin material (tire frame) itself is usually 100 ° C. to 350 ° C., preferably about 100 ° C. to 250 ° C., but from the viewpoint of tire productivity, 120 ° C. to 250 ° C. The degree is preferable, and 140 ° C to 230 ° C is more preferable.
- a resin material having a melting point of 120 ° C. to 250 ° C. for example, when a tire skeleton is formed by fusing the divided bodies (frame pieces), the periphery of 120 ° C. to 250 ° C. Even if the frame body is fused in the temperature range, the bonding strength between the tire frame pieces is sufficient.
- the heating temperature is preferably 10 ° C to 150 ° C higher than the melting point (or softening point) of the resin material forming the tire frame piece, and more preferably 10 ° C to 100 ° C higher.
- the resin material can be obtained by adding various additives as necessary and mixing them appropriately by a known method (for example, melt mixing).
- the resin material obtained by melt mixing can be used in the form of pellets if necessary.
- the tensile yield strength specified in JIS K7113: 1995 of the resin material (tire frame) itself is preferably 5 MPa or more, more preferably 5 MPa to 20 MPa, and further preferably 5 MPa to 17 MPa.
- the resin material can withstand deformation against a load applied to the tire during traveling.
- the tensile yield elongation defined by JIS K7113: 1995 of the resin material (tire frame) itself is preferably 10% or more, preferably 10% to 70%, and more preferably 15% to 60%.
- the tensile yield elongation of the resin material is 10% or more, the elastic region is large and the air sealability can be improved.
- the tensile elongation at break specified in JIS K7113: 1995 of the resin material (tire frame) itself is preferably 50% or more, preferably 100% or more, more preferably 150% or more, and particularly preferably 200% or more.
- the rim assembly property is good and it is possible to make it difficult to break against a collision.
- the deflection temperature under load (when loaded with 0.45 MPa) as defined in ISO 75-2 or ASTM D648 of the resin material (tire frame) itself is preferably 50 ° C. or more, preferably 50 ° C. to 150 ° C., and preferably 50 ° C. to 50 ° C. 130 ° C. is more preferable.
- the deflection temperature under load of the resin material is 50 ° C. or higher, deformation of the tire skeleton can be suppressed even when vulcanization is performed in the manufacture of the tire.
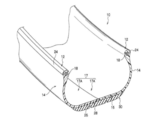
- FIG. 1A is a perspective view showing a partial cross section of a tire according to an embodiment of the present invention.
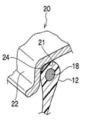
- FIG. 1B is a cross-sectional view of a bead portion attached to a rim.
- the tire 10 of the present embodiment has a cross-sectional shape that is substantially the same as a conventional general rubber pneumatic tire.
- the tire 10 includes a pair of bead portions 12 that contact the bead seat 21 and the rim flange 22 of the rim 20 shown in FIG. 1B, and side portions 14 that extend outward from the bead portion 12 in the tire radial direction.
- a tire case 17 (tire frame) comprising a crown portion 16 (outer peripheral portion) for connecting a tire radial direction outer end of one side portion 14 and a tire radial direction outer end of the other side portion 14.
- the tire case 17 of the present embodiment includes, as a resin material, for example, a hard segment, a soft segment composed of a structural unit derived from polypropylene glycol, and two or more segments and having 6 to 20 carbon atoms.
- a polyamide thermoplastic elastomer having a weight average molecular weight of 20,000 or more and 250,000 or less and including each additive can be used.
- the tire case 17 is formed of a single resin material.
- the present invention is not limited to this configuration, and each part of the tire case 17 is similar to a conventional general rubber pneumatic tire.
- a reinforcing material (polymer material, metal fiber, cord, nonwoven fabric, woven fabric, etc.) is embedded in the tire case 17 (for example, the bead portion 12, the side portion 14, the crown portion 16 and the like), and the reinforcing material is provided.
- the tire case 17 may be reinforced.
- the tire case 17 of the present embodiment is obtained by joining a pair of tire case halves (tire frame pieces) 17A formed of a resin material.
- the tire case half 17A is formed by injection molding or the like so that one bead portion 12, one side portion 14, and a half-width crown portion 16 are integrated with each other so as to face each other. It is formed by joining at the tire equator part.
- the tire case 17 is not limited to the one formed by joining two members, and may be formed by joining three or more members.
- the tire case half 17A formed of the resin material can be formed by, for example, vacuum forming, pressure forming, injection molding, melt casting, or the like. For this reason, it is not necessary to perform vulcanization compared to the case where the tire case is molded with rubber as in the prior art, the manufacturing process can be greatly simplified, and the molding time can be omitted.
- the tire case half body 17A has a symmetrical shape, that is, the one tire case half body 17A and the other tire case half body 17A have the same shape. There is also an advantage that only one type of mold is required.
- an annular bead core 18 made of a steel cord is embedded in the bead portion 12, similar to a conventional general pneumatic tire.
- the present invention is not limited to this configuration, and the bead core 18 can be omitted if the rigidity of the bead portion 12 is ensured and there is no problem in fitting with the rim 20.
- an organic fiber cord, a resin-coated organic fiber cord, or a hard resin may be used.
- An annular seal layer 24 made of is formed.
- the seal layer 24 may also be formed at a portion where the tire case 17 (bead portion 12) and the bead sheet 21 are in contact with each other.
- a material having better sealing properties than the resin material constituting the tire case 17 a softer material than the resin material constituting the tire case 17 can be used.
- thermoplastic resin thermoplastic elastomer
- examples of such other thermoplastic resins include polyurethane resins, polyolefin resins, polystyrene thermoplastic resins, polyester resins, and the like, and blends of these resins with rubbers or elastomers.
- Thermoplastic elastomers can also be used, for example, polyester-based thermoplastic elastomers, polyurethane-based thermoplastic elastomers, polystyrene-based thermoplastic elastomers, polyolefin-based thermoplastic elastomers, combinations of these elastomers, and blends with rubber. Thing etc. are mentioned.
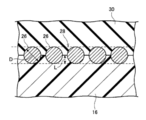
- a reinforcement cord 26 having higher rigidity than the resin material constituting the tire case 17 is wound around the crown portion 16 in the circumferential direction of the tire case 17.
- the reinforcing cord 26 is wound spirally in a state in which at least a part thereof is embedded in the crown portion 16 in a cross-sectional view along the axial direction of the tire case 17, thereby forming a reinforcing cord layer 28.
- FIG. 2 is a cross-sectional view along the tire rotation axis showing a state where a reinforcing cord is embedded in the crown portion of the tire case of the tire of the first embodiment.
- the reinforcing cord 26 is spirally wound in a state in which at least a part is embedded in the crown portion 16 in a sectional view along the axial direction of the tire case 17.
- a reinforcing cord layer 28 indicated by a broken line portion in FIG. 2 is formed together with a part of the outer peripheral portion 17.
- the portion embedded in the crown portion 16 of the reinforcing cord 26 is in close contact with the resin material constituting the crown portion 16 (tire case 17).
- a monofilament such as a metal fiber or an organic fiber, or a multifilament (twisted wire) obtained by twisting these fibers such as a steel cord twisted with a steel fiber
- a steel cord is used as the reinforcing cord 26.
- the burying amount L indicates the burying amount of the reinforcing cord 26 in the tire rotation axis direction with respect to the tire case 17 (crown portion 16).
- the embedding amount L of the reinforcing cord 26 in the crown portion 16 is preferably 1/5 or more of the diameter D of the reinforcing cord 26, and more preferably more than 1/2. Most preferably, the entire reinforcing cord 26 is embedded in the crown portion 16. When the embedment amount L of the reinforcing cord 26 exceeds 1/2 of the diameter D of the reinforcing cord 26, it is difficult to jump out of the embedded portion due to the size of the reinforcing cord 26.
- the reinforcing cord layer 28 corresponds to a belt disposed on the outer peripheral surface of the carcass of a conventional rubber pneumatic tire.
- the tread 30 is disposed on the outer peripheral side of the reinforcing cord layer 28 in the tire radial direction.
- the rubber used for the tread 30 is preferably the same type of rubber as that used in conventional rubber pneumatic tires.
- a tread formed of another type of resin material that is more excellent in wear resistance than the resin material constituting the tire case 17 may be used.
- the tread 30 is formed with a tread pattern including a plurality of grooves on the ground contact surface with the road surface in the same manner as a conventional rubber pneumatic tire.
- the manufacturing method of the tire of this embodiment is explained.
- a tire case half is formed using a resin material containing a resin composition containing the polyamide-based thermoplastic resin elastomer. These tire cases are preferably formed by injection molding.
- the tire case halves supported by the thin metal support ring face each other.
- a joining mold (not shown) is installed so as to be in contact with the outer peripheral surface of the abutting portion of the tire case half.
- die is comprised so that the periphery of the junction part (butting part) of the tire case half body 17A may be pressed with a predetermined pressure.
- the periphery of the joint portion of the tire case half is pressed at a temperature equal to or higher than the melting point (or softening point) of the resin material constituting the tire case.
- the joint portion of the tire case half is heated or pressed by the joining mold, the joint portion is melted and the tire case halves are fused together, and the tire case 17 is formed by integrating these members.
- the joining portion of the tire case half is heated using a joining mold, but the present invention is not limited to this.
- the joining portion is heated by a separately provided high-frequency heater or the like.
- the tire case halves may be joined by softening or melting in advance by irradiation with hot air, infrared rays, or the like, and pressurizing with a joining mold.
- the heated reinforcing cord 26 is wound while being embedded in the outer peripheral surface of the crown portion 16 using a reel, a cord heating device, and a cord supply device provided with various rollers.
- the reinforcing cord layer 28 can be formed on the outer peripheral side of the crown portion 16 of the tire case 17.
- the reinforcing cord layer 28 is formed on the outer peripheral side of the crown portion 16 of the tire case 17 by winding the heated reinforcing cord 26 while being embedded in the outer peripheral surface of the crown portion 16.
- the vulcanized belt-like tread 30 is wound around the outer peripheral surface of the tire case 17 by one turn, and the tread 30 is bonded to the outer peripheral surface of the tire case 17 using an adhesive or the like.
- the precure tread used for the retread tire conventionally known can be used for the tread 30, for example. This step is the same step as the step of bonding the precure tread to the outer peripheral surface of the base tire of the retreaded tire.
- the seal layer 24 made of vulcanized rubber is bonded to the bead portion 12 of the tire case 17 using an adhesive or the like, the tire 10 is completed.
- the tire case 17 is derived from a hard segment, a soft segment composed of a structural unit derived from polypropylene glycol, and a dicarboxylic acid having 6 to 20 carbon atoms by combining two or more segments. And a bond part composed of structural units, and a resin material containing a polyamide-based thermoplastic elastomer having a weight average molecular weight of 20,000 to 250,000. For this reason, the tire 10 of the present embodiment is excellent in balance between manufacturing efficiency, low loss, heat resistance, and rim assembly.
- a reinforcing cord 26 having a rigidity higher than that of the resin material is spirally wound in the circumferential direction on the outer peripheral surface of the crown portion 16 of the tire case 17 formed of a resin material. Therefore, puncture resistance, cut resistance, and circumferential rigidity of the tire 10 are improved. In addition, creep of the tire case 17 formed of a resin material is prevented by improving the circumferential rigidity of the tire 10.
- the reinforcing cord 26 is embedded in the outer peripheral surface of the crown portion 16 of the tire case 17 formed of a resin material in a cross-sectional view along the axial direction of the tire case 17 (the cross section shown in FIG. 1A).
- the reinforcing cord 26 since it is in close contact with the resin material, entry of air at the time of manufacture is suppressed, and movement of the reinforcing cord 26 due to input during travel is suppressed. Thereby, it is suppressed that peeling etc. arise in the reinforcement cord 26, the tire case 17, and the tread 30, and durability of the tire 10 improves.
- the embedding amount L of the reinforcement cord 26 is 1/5 or more of the diameter D as shown in FIG. 2, the air entry at the time of manufacture is suppressed effectively, the input at the time of driving, etc. This further suppresses the movement of the reinforcing cord 26.
- annular bead core 18 made of a metal material is embedded in the bead portion 12, the tire case 17, that is, the tire 10 is strong against the rim 20 like the conventional rubber pneumatic tire. Retained.
- a seal layer 24 made of a rubber material having a sealing property than the resin material constituting the tire case 17 is provided at a portion of the bead portion 12 that contacts the rim 20, the tire 10 and the rim 20 The sealing performance between the two is improved. For this reason, compared with the case where it seals only with the rim
- the reinforcing cord 26 is heated.
- the outer periphery of the reinforcing cord 26 may be covered with the same resin material as the tire case 17. In this case, the covering reinforcing cord is used.
- the resin material covered with the reinforcing cord 26 is also heated, so that the air can be effectively suppressed when being embedded in the crown portion 16.
- the tire 10 of the first embodiment is a so-called tubeless tire in which an air chamber is formed between the tire 10 and the rim 20 by attaching the bead portion 12 to the rim 20, but the present invention is limited to this configuration. It may be a complete tube shape.
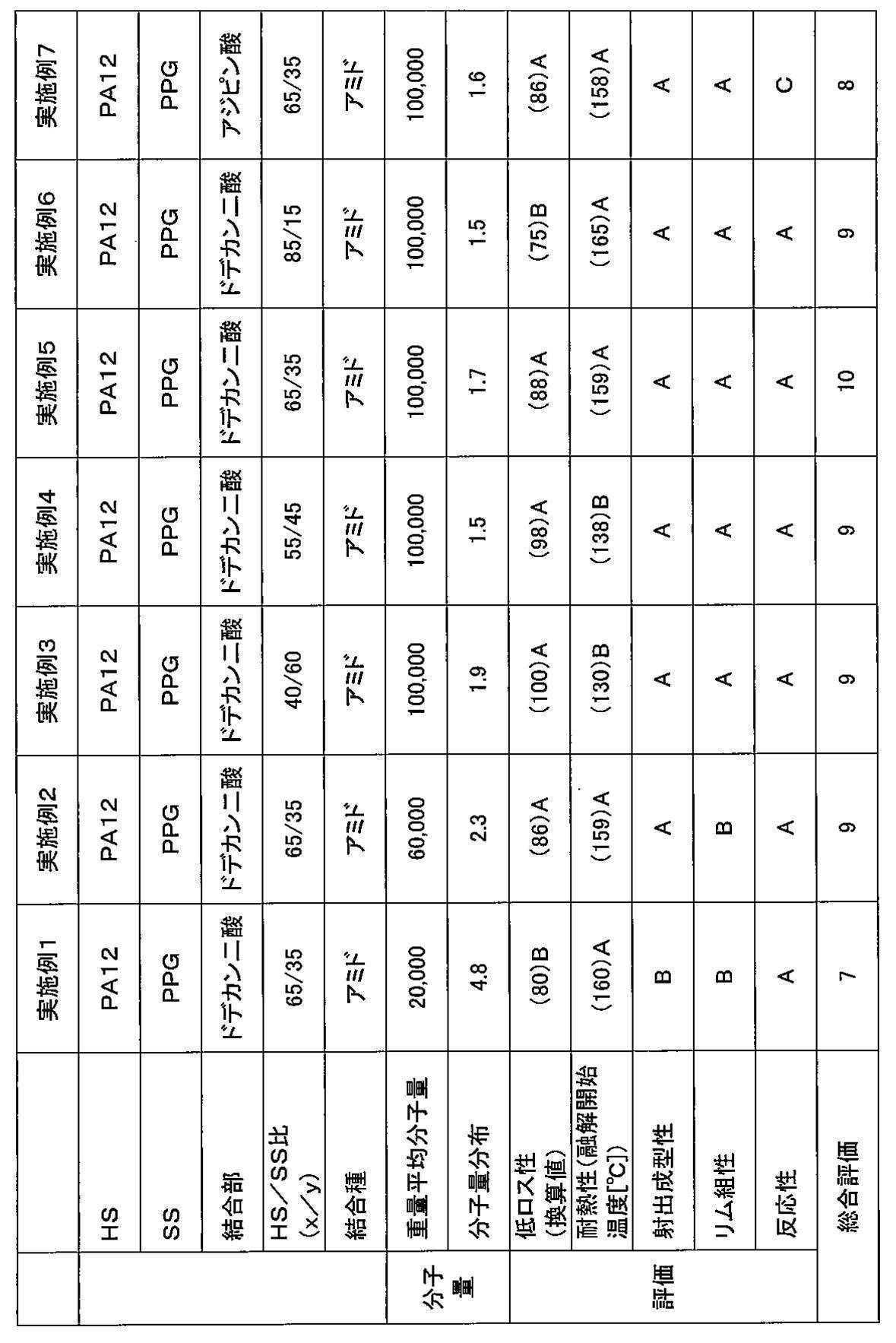
- Example 1 In a reaction vessel with a volume of 2 liters equipped with a stirrer, a nitrogen gas inlet, and a condensed water outlet, 1,6.4 aminododecanoic acid (manufactured by Aldrich), 500 g aminododecanolactam, dodecanedioic acid (bonding part) 17.7 g of the chain length extending agent), and the inside of the container was sufficiently purged with nitrogen, and then the temperature was raised to 280 ° C. and reacted under a pressure of 0.4 MPa for 4 hours.
- 1,6.4 aminododecanoic acid manufactured by Aldrich
- 500 g aminododecanolactam 500 g aminododecanolactam
- nylon 12 polymer having a molecular weight of 3700 (polymerization reaction A).
- the obtained nylon 12 polymer is obtained by bonding dodecanedioic acid as a chain extender to the end of polyamide (nylon 12) having a molecular weight of 3200 as a hard segment.
- Example 2 A polyamide-based thermoplastic elastomer was obtained in the same manner as in Example 1, except that the amount of polyoxypropylenediamine added in the polymerization reaction B was 131.3 g, the reaction temperature and the reaction time were 230 ° C. and 4 hours, respectively.
- Example 3 A polyamide-based thermoplastic elastomer was obtained in the same manner as in Example 1 except that the amount of polyoxypropylenediamine added was 175 g and the reaction temperature and reaction time in the polymerization reaction B were 230 ° C. and 5 hours, respectively.
- Example 4 A polyamide-based thermoplastic elastomer is obtained in the same manner as in Example 1 except that the amount of polyoxypropylenediamine added is 127.3 g and the reaction temperature and reaction time in the polymerization reaction B are 230 ° C. and 5 hours, respectively. It was.
- Example 5 A polyamide-based thermoplastic elastomer is obtained in the same manner as in Example 1 except that the amount of polyoxypropylenediamine added is 107.7 g and the reaction temperature and reaction time in the polymerization reaction B are 230 ° C. and 5 hours, respectively. It was.
- Example 6 A polyamide-based thermoplastic elastomer is obtained in the same manner as in Example 1 except that the amount of polyoxypropylenediamine added is 82.4 g and the reaction temperature and reaction time in the polymerization reaction B are 230 ° C. and 5 hours, respectively. It was.
- Example 7 The type and amount of chain extender used are 14.0 g of adipic acid, the reaction temperature and reaction time in polymerization reaction A are 280 ° C. and 4 hours, respectively, and the reaction temperature and reaction time in polymerization reaction B are 230 ° C. and A polyamide-based thermoplastic elastomer was obtained in the same manner as in Example 1 except that the time was 6.5 hours.
- Example 8 The type and amount of chain extender used were 29.0 g eicosane diacid, the reaction temperature and reaction time in polymerization reaction A were 280 ° C. and 4 hours, respectively, and the reaction temperature and reaction time in polymerization reaction B were 230 ° C., respectively.
- a polyamide-based thermoplastic elastomer was obtained in the same manner as in Example 1 except that the time was 5.5 hours.
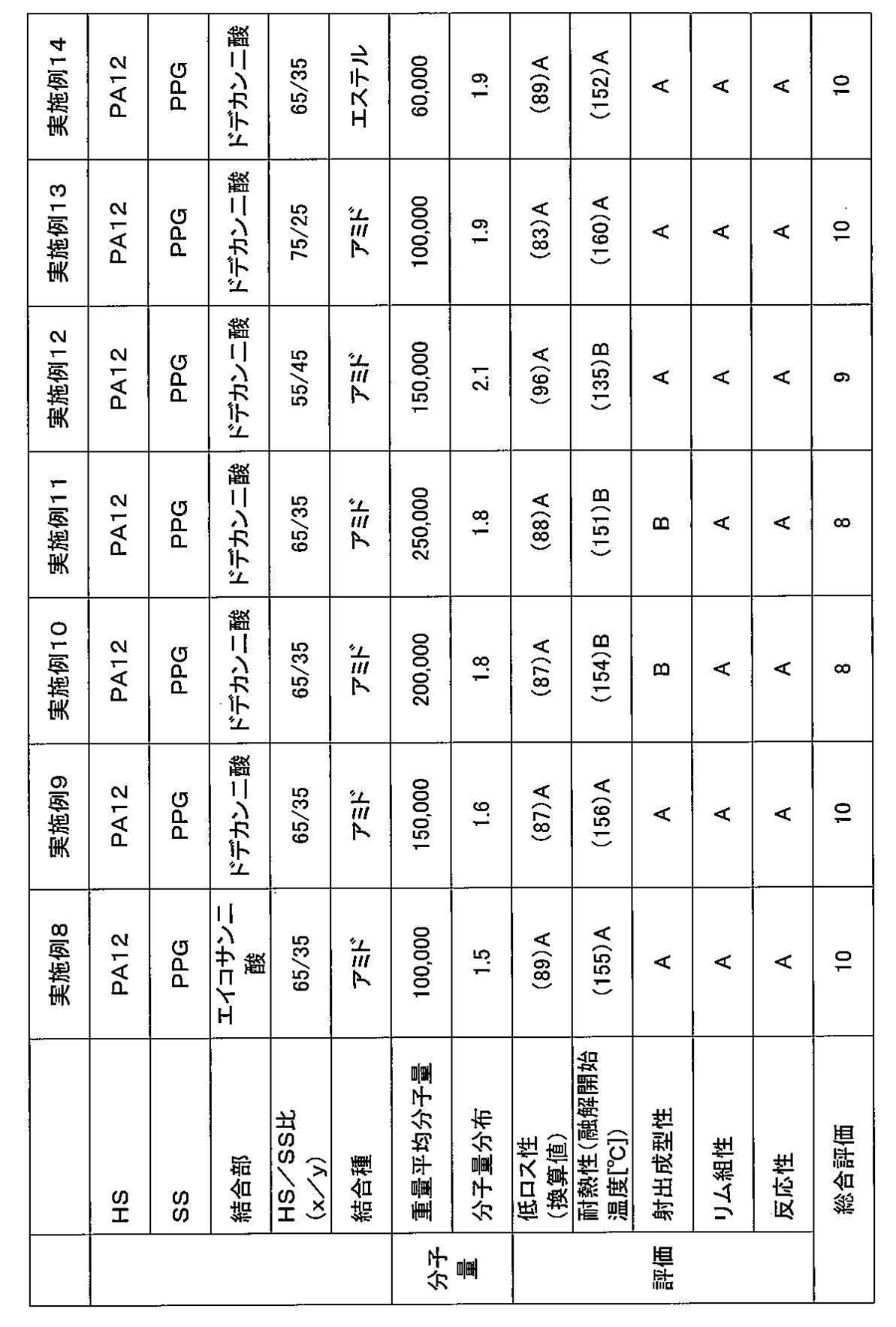
- Example 9 A polyamide-based thermoplastic elastomer is obtained in the same manner as in Example 1 except that the amount of polyoxypropylenediamine added is 107.7 g and the reaction temperature and reaction time in the polymerization reaction B are 230 ° C. and 6 hours, respectively. It was.
- Example 10 A polyamide-based thermoplastic elastomer is obtained in the same manner as in Example 1 except that the amount of polyoxypropylenediamine added is 107.7 g and the reaction temperature and reaction time in the polymerization reaction B are 230 ° C. and 7 hours, respectively. It was.
- Example 11 The polyamide-based thermoplastic elastomer was the same as in Example 1 except that the amount of polyoxypropylenediamine added was 107.7 g and the reaction temperature and reaction time in the polymerization reaction B were 230 ° C. and 8.5 hours, respectively. Got.
- Example 12 A polyamide-based thermoplastic elastomer is obtained in the same manner as in Example 1 except that the amount of polyoxypropylenediamine added is 127.3 g and the reaction temperature and reaction time in the polymerization reaction B are 230 ° C. and 6 hours, respectively. It was.
- Example 13 A polyamide-based thermoplastic elastomer is obtained in the same manner as in Example 1 except that the amount of polyoxypropylenediamine added is 93.3 g and the reaction temperature and reaction time in the polymerization reaction B are 230 ° C. and 6 hours, respectively. It was.
- Example 14 Instead of polyoxypropylenediamine, 131.3 g of polypropylene glycol (manufactured by Wako Pure Chemical Industries, Ltd., product number: 164-05895, number average molecular weight: 2000) was used, and zirconium tetrabutoxide (Tokyo Chemical Industry Co., Ltd.) was used as a polymerization catalyst. ), Product number: Z0016) was used, and a polyamide-based thermoplastic elastomer was obtained in the same manner as in Example 1 except that the reaction temperature and reaction time in the polymerization reaction B were 260 ° C. and 6 hours, respectively.
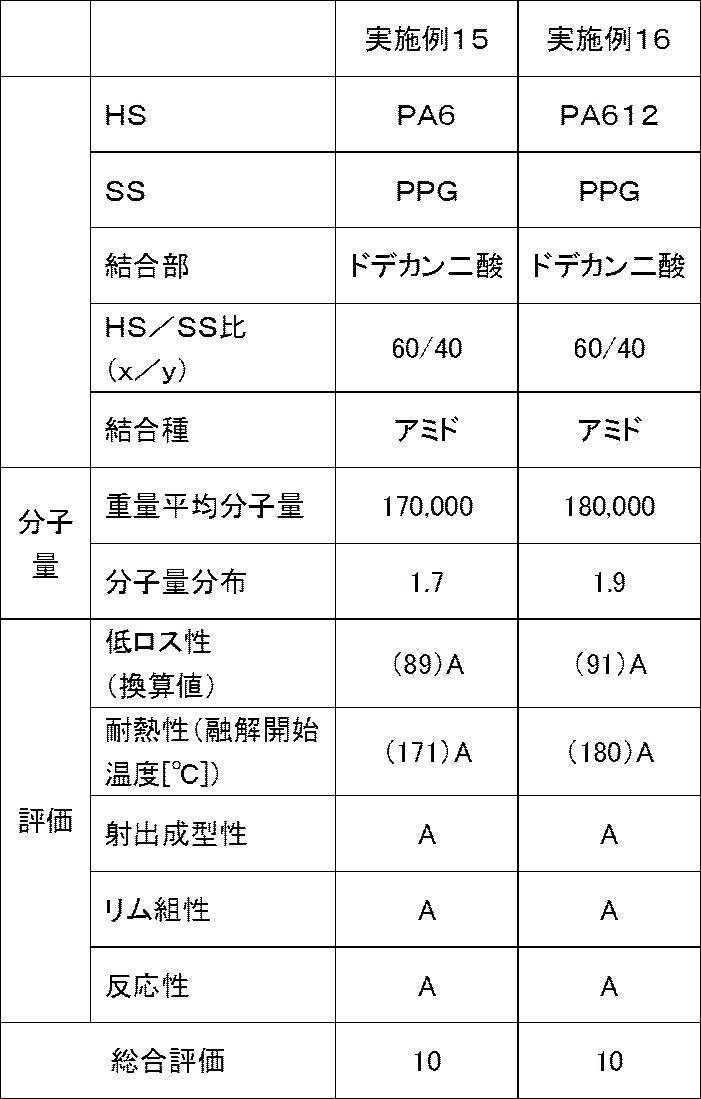
- Example 15 A reaction vessel having a volume of 2 liters equipped with a stirrer, a nitrogen gas inlet, and a condensed water outlet was charged with 500 g of Aldrich caprolactam, 124 g of dodecanedioic acid, and 77 g of aminohexanoic acid. The temperature was raised to 0 ° C. and reacted for 4 hours under a pressure of 0.6 MPa. After releasing the pressure, the mixture was further reacted for 1 hour under a nitrogen stream, and a white solid which was a nylon 6 (polyamide 6) polymer having a number average molecular weight of 3000 was obtained through a water washing step.
- a white solid which was a nylon 6 (polyamide 6) polymer having a number average molecular weight of 3000 was obtained through a water washing step.
- nylon 6 polyamide 6
- polyoxypropylene diamine Elastamine RP-2009, number average molecular weight: 2000, manufactured by HUNTSMAN
- Irganox 1010 was added to obtain a white polyamide-based thermoplastic elastomer.
- Example 16 207 g of dodecanedioic acid, 93 g of hexamethylene diamine, and 100 g of purified water were placed in a reaction vessel having a volume of 2 liters equipped with a stirrer, a nitrogen gas inlet, and a condensed water outlet, and the inside of the vessel was sufficiently purged with nitrogen. The reaction was continued for 4 hours under a pressure of 0.6 MPa. After releasing the pressure, the mixture is further reacted for 1 hour under a nitrogen stream, taken out from the container, and subjected to Soxhlet extraction with ethanol for 24 hours to obtain a white solid which is a nylon 612 (polyamide 612) polymer having a number average molecular weight of 3000. It was.
- nylon 612 polyamide 612
- polyoxypropylene diamine Elastamine RP-2009, number average molecular weight: 2000, manufactured by HUNTSMAN
- Irganox 1010 was added to obtain a white polyamide-based thermoplastic elastomer.
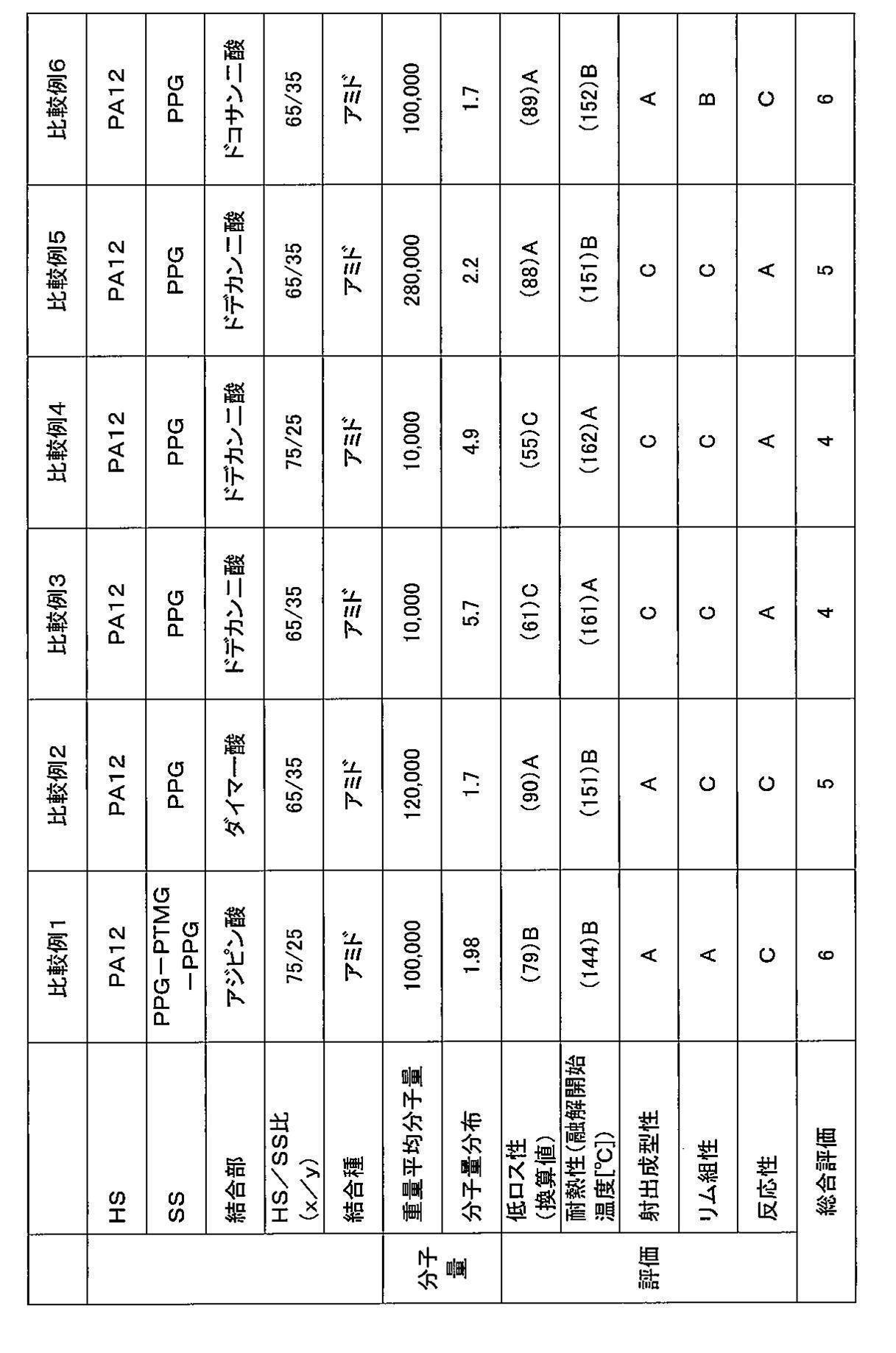
- Example 3 A polyamide-based thermoplastic elastomer is obtained in the same manner as in Example 1 except that the amount of polyoxypropylenediamine added is 269.2 g and the reaction temperature and reaction time in the polymerization reaction B are 250 ° C. and 1 hour, respectively. It was.
- Example 4 A polyamide-based thermoplastic elastomer is obtained in the same manner as in Example 1 except that the amount of polyoxypropylenediamine added is 233.3 g and the reaction temperature and reaction time in the polymerization reaction B are 250 ° C. and 1 hour, respectively. It was.
- Example 5 A polyamide-based thermoplastic elastomer is obtained in the same manner as in Example 1 except that the amount of polyoxypropylenediamine added is 107.7 g and the reaction temperature and reaction time in the polymerization reaction B are 230 ° C. and 9 hours, respectively. It was.
- thermoplastic elastomers obtained from the examples and comparative examples were evaluated using the thermoplastic elastomers obtained from the examples and comparative examples. Specifically, the obtained polyamide-based thermoplastic elastomer was pelletized and injection molded at 200 to 240 ° C. to obtain a sample piece. Various measurements were carried out using a sample obtained by punching a test piece from this sample piece. The results are shown in Tables 1 to 4.
- tan ⁇ a loss tangent (tan ⁇ ) was measured at a temperature of 30 ° C., a strain of 1%, and a frequency of 20 Hz using a sample having a diameter of 8 mm and a thickness of 2 mm, and evaluated according to the following criteria. . In addition, it is excellent in low-loss property, so that tan-delta is small.
- thermoplastic elastomers obtained in each of the examples and comparative examples were measured from 0 ° C to 200 ° C. The temperature was raised at 10 ° C / min.
- the evaluation criteria are as follows. A: Melting start temperature is 155 ° C. or higher B: Melting start temperature is 130 ° C. or higher and lower than 155 ° C. C: Melting start temperature is lower than 130 ° C.
- injection molding is performed under conditions of a molding temperature of 200 ° C to 235 ° C, a mold temperature of 50 ° C to 70 ° C, and a load of 15 tons. The time to completion was measured (weighing time). From the obtained results, the following criteria were evaluated. A: Flat plate can be formed at a load of 15t, and handling properties are good (weighing time is less than 30 seconds) B: Although a flat plate can be formed at a load of 15 t, handling properties are inferior (weighing time is 30 seconds or more). C: Filling failure is observed at a load of 15t.
- thermoplastic elastomers obtained in each of the examples and comparative examples were subjected to the above evaluations (low loss, heat resistance, injection moldability, rim assembly, and reactivity).
- a total evaluation was performed with a total of 10 points, with “point” and “B” being “+1 point” and “C” being “0 point”. If the overall evaluation is 7 points or more, a tire having a well-balanced manufacturing efficiency, low loss, heat resistance, and rim assembly can be obtained.
- thermoplastic elastomer obtained in each example and comparative example was measured by gel permeation chromatography (GPC) using “HLC-8320GPC EcoSEC” manufactured by Tosoh Corporation. The results are shown in Tables 1 to 4.
- HS means a hard segment
- SS means a soft segment
- PA12 means derived from nylon 12 (polyamide 12)
- PA6 means derived from nylon 6 (polyamide 6)
- PA612 represents nylon. It is derived from 612 (polyamide 612).
- PPG means derived from polypropylene glycol. Further, “PPG-PTMG-PPG” means a terpolymer having a structural unit derived from PTMG and a structural unit derived from PPG.
- Comparative Example 1 in which the soft segment is a triblock copolymer containing PTMG, and Comparative Example 2 in which the carbon number of the dicarboxylic acid forming the bonding portion is out of the above range.
- Comparative Example 6 and Comparative Example 3 to Comparative Example 5 in which the weight average molecular weight of the polyamide-based thermoplastic elastomer is out of the above range, the overall evaluation is high, the production efficiency (that is, injection moldability and reactivity), and the low loss property It was found that the heat resistance and rim assemblability were excellent in a well-balanced manner.
Landscapes
- Chemical & Material Sciences (AREA)
- Health & Medical Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
- Organic Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Engineering & Computer Science (AREA)
- Mechanical Engineering (AREA)
- Tires In General (AREA)
- Polyamides (AREA)
Abstract
Description
近年では、軽量化や、成形の容易さ、リサイクルのしやすさから、樹脂材料、特に熱可塑性樹脂や熱可塑性エラストマーなどをタイヤ材料として用いることが検討されている。これら熱可塑性の高分子材料(熱可塑性エラストマー、熱可塑性樹脂材料等)は、射出成形が可能であるなど、生産性の向上の観点から有利な点が多い。例えば、前記熱可塑性の高分子材料としてポリアミド系熱可塑性エラストマーを用いて製造されたタイヤが提案されている(特開2012-45790号参照)。
このように、熱可塑性の高分子材料を用いたタイヤにおいては、高い製造効率、低ロス性、耐熱性、及び優れたリム組み性のバランスが求められている。
具体的には、例えば本発明の一実施形態では、ソフトセグメントがポリテトラメチレンエーテルグリコール(PTMG)に由来する構成単位を含む場合に比べ、製造効率、耐熱性、及びリム組み性を維持しつつ、特に低ロス性を向上させることができる。その理由は定かではないが、ソフトセグメントを形成するポリマーとして用いられるポリプロピレングリコールが非晶性であるため、結晶性を有するPTMGを含む場合に比べて、タイヤの低ロス性が優れると推測される。
以上のように、上記ポリアミド系熱可塑性エラストマーを適用した前記タイヤは、樹脂材料を用いて形成され、製造効率、低ロス性、耐熱性、及びリム組み性がバランスよく優れることとなる。
上述のように、前記タイヤは、ポリアミド系熱可塑性エラストマーを含む樹脂材料を用いたタイヤ骨格体を有する。そして、ポリアミド系熱可塑性エラストマーは、少なくとも、ハードセグメントと、ポリプロピレングリコールに由来する構成単位からなるソフトセグメントと、2つ以上のセグメントを結合し炭素数6以上20以下のジカルボン酸に由来する構成単位からなる結合部と、を有し、重量平均分子量が20,000以上250,000以下である。
なお、前記樹脂材料は、上記熱可塑性エラストマー以外の熱可塑性エラストマーや、任意の成分を含んでいてもよい。また、本明細書において「樹脂」とは、熱可塑性樹脂及び熱硬化性樹脂を含む概念であるが、天然ゴムは含まない。
また本明細書において、「~」を用いて表される数値範囲は、「~」の前後に記載される数値を下限値及び上限値として含む範囲を意味する。
また本明細書において、組成物中の各成分の量は、各成分に該当する物質が組成物中に複数存在する場合には、特に断らない限り、組成物中に存在する複数の物質の合計量を意味する。
本明細書において、「ポリアミド系熱可塑性エラストマー」とは、結晶性で融点の高いハードセグメントを構成するポリマーと非晶性でガラス転移温度の低いソフトセグメントを構成するポリマーとを有する共重合体からなる熱可塑性エラストマーであって、ハードセグメントを構成するポリマーの主鎖にアミド結合(-CONH-)を有するものを意味する。
前記ハードセグメントを形成するポリアミドとしては、例えば、下記一般式(1)又は一般式(2)で表されるモノマーを用いて合成されるポリアミドを挙げることができる。
前記一般式(1)又は一般式(2)で表されるモノマーとしては、ω-アミノカルボン酸やラクタムが挙げられる。また、前記ハードセグメントを形成するポリアミドとしては、これらω-アミノカルボン酸やラクタムの重縮合体や、ジアミンとジカルボン酸との共縮重合体等が挙げられる。
前記ジアミンとしては、例えば、エチレンジアミン、トリメチレンジアミン、テトラメチレンジアミン、ヘキサメチレンジアミン、ヘプタメチレンジアミン、オクタメチレンジアミン、ノナメチレンジアミン、デカメチレンジアミン、ウンデカメチレンジアミン、ドデカメチレンジアミン、2,2,4-トリメチルヘキサメチレンジアミン、2,4,4-トリメチルヘキサメチレンジアミン、3-メチルペンタメチレンジアミン、メタキシレンジアミンなどの炭素数2~20の脂肪族ジアミンなどのジアミン化合物を挙げることができる。また、ジカルボン酸は、HOOC-(R3)m-COOH(R3:炭素数3~20の炭化水素の分子鎖、m:0又は1)で表すことができ、例えば、シュウ酸、コハク酸、グルタル酸、アジピン酸、ピメリン酸、スベリン酸、アゼライン酸、セバシン酸、ドデカン二酸などの炭素数2~22の脂肪族ジカルボン酸を挙げることができる。
前記ポリアミド11は、例えば、{CO-(CH2)10-NH}n(nは任意の繰り返し単位数を表す)で表すことができ、例えば、nとしては2~100が好ましく、3~50が更に好ましい。
前記ポリアミド12は、例えば、{CO-(CH2)11-NH}n(nは任意の繰り返し単位数を表す)で表すことができ、例えば、nとしては2~100が好ましく、3~50が更に好ましい。
前記ポリアミド66は、例えば、{CO(CH2)4CONH(CH2)6NH}n(nは任意の繰り返し単位数を表す)で表すことができ、例えば、nとしては2~100が好ましく、3~50が更に好ましい。
前記ポリアミド612は、例えば、{CO(CH2)12CONH(CH2)6NH}n(nは任意の繰り返し単位数を表す)で表すことができ、例えば、nとしては2~100が好ましく、3~50が更に好ましい。
また、前記ポリアミド系熱可塑性エラストマーは、低ロス性、耐熱性、及び優れたリム組み性のバランスの観点から、特に、ハードセグメントとして、-[CO-(CH2)11-NH]-で表される単位構造を有するポリアミド(ポリアミド12)、-[CO-(CH2)5-NH]-で表される単位構造を有するポリアミド(ポリアミド6)、及び-[CO(CH2)12CONH(CH2)6NH]-で表される単位構造を有するポリアミド(ポリアミド612)の少なくとも1つを有することが好ましい。
前記の通りソフトセグメントは、ポリプロピレングリコールに由来する構成単位からなる。すなわち前記ソフトセグメントは、ポリプロピレングリコールのみで形成される。
前記ソフトセグメントを形成するポリマー(ポリプロピレングリコール)としては、両末端が水酸基であるポリプロピレングリコールのほか、末端に官能基を導入したポリプロピレングリコール誘導体が挙げられる。
すなわちポリプロピレングリコール誘導体としては、例えば、両末端が水酸基であるポリプロピレングリコールの末端にアンモニア等を反応させることによって得られるポリオキシプロピレンジアミン等が挙げられる。
以下、両末端が水酸基であるポリプロピレングリコールと前記ポリプロピレングリコール誘導体とを総称して「ポリプロピレングリコール」と称する場合がある。
前記の通り結合部は、炭素数6以上20以下のジカルボン酸に由来する構成単位からなる。すなわち結合部は、炭素数6以上20以下のジカルボン酸である鎖長延長剤のみで形成される。
前記結合部を形成する鎖長延長剤は、炭素数6以上20以下のジカルボン酸であればよく、具体的には、例えば、脂肪族ジカルボン酸、脂環式ジカルボン酸、芳香族ジカルボン酸等が挙げられ、これらを1種のみ用いてもよく、2種以上併用してもよい。これらの中でも鎖長延長剤としては、脂肪族ジカルボン酸が好ましく、直鎖脂肪族ジカルボン酸がより好ましく、炭素数8以上20以下の直鎖脂肪族ジカルボン酸がさらに好ましく、炭素数10以上18以下の直鎖脂肪族ジカルボン酸が特に好ましく、炭素数12以上18以下の直鎖脂肪族ジカルボン酸がさらに好ましい。
樹脂材料に含まれるポリアミド系熱可塑性エラストマーの重量平均分子量は、20,000以上250,000以下である。前記ポリアミド系熱可塑性エラストマーの重量平均分子量が20,000未満であると、リム組み性が低下してしまう。また、前記ポリアミド系熱可塑性エラストマーの重量平均分子量が250,000を超えると、溶融粘度が高くなり、タイヤ骨格体を形成する際に充填不足が発生するおそれがあるため、成形温度、金型温度を高くする必要がある場合がある。そして、充填不足を防ぐために成形温度及び金型温度を高くした場合、サイクルタイムが長くなり、生産性(製造効率)が劣る。
前記ポリアミド系熱可塑性エラストマーの重量平均分子量は、ゲルパーミエーションクロマトグラフィー(GPC)により測定することができ、例えば、東ソー株式会社製の「HLC-8320GPC EcoSEC」等のGPC(ゲル浸透クロマトグラフィー)を用いることができる。
なお、本明細書を通じて、「x」は、ポリアミド系熱可塑性エラストマーに含まれるハードセグメントの質量を示し、「y」は、ポリアミド系熱可塑性エラストマーに含まれるソフトセグメントの質量を示す。また「x/y」は、「前記ハードセグメントの質量(x)/前記ソフトセグメントの質量(y)」を示す。
前記ポリアミド系熱可塑性エラストマー中のソフトセグメントの含有量は、ポリアミド系熱可塑性エラストマー全量に対して、10~95質量%が好ましく、10~90質量%が更に好ましい。
前記鎖長延長剤の含有量は前記ソフトセグメントを構成するポリプロピレングリコールの末端の官能基(例えば水酸基又はアミノ基)と、鎖長延長剤のカルボキシル基とが、ほぼ等モルになるように設定されることが好ましい。
前記ポリアミド系熱可塑性エラストマーは、前記ハードセグメントを形成するポリマー及びソフトセグメントを形成するポリマーを公知の方法によって共重合することで合成することができる。
例えば、前記ポリアミド系熱可塑性エラストマーは、ハードセグメントを構成するモノマー(例えば、12-アミノドデカン酸などのω-アミノカルボン酸や、ラウリルラクタムなどのラクタム)と鎖長延長剤(例えば、アジピン酸又はデカンジカルボン酸)とを容器内で重合させた後、ソフトセグメントを構成するポリマー(ポリプロピレングリコール)を添加し、さらに重合させることで得ることができる。
例えば、無機系リン化合物、有機チタン化合物、有機ジルコニウム化合物、有機スズ化合物等が挙げられる。
具体的には、無機系リン化合物としては、リン酸、ピロリン酸、ポリリン酸、亜リン酸、次亜リン酸等のリン含有酸、リン含有酸のアルカリ金属塩、リン含有酸のアルカリ土類金属塩等が挙げられる。
有機チタン化合物としては、チタンアルコキシド〔チタンテトラブトキシド、チタンテトライソプロポキシド等〕等が挙げられる。
有機ジルコニウム化合物としては、ジルコニウムアルコキシド〔ジルコニウムテトラブトキシド(「Zr(OBu)4」または「Zr(OC4H8)4」とも称される)等〕等が挙げられる。
有機スズ化合物としては、ジスタノキサン化合物〔1-ヒドロキシ-3-イソチオシアネート-1,1,3,3-テトラブチルジスタノキサン等〕、酢酸スズ、ジラウリン酸ジブチルスズ、ブチルチンヒドロキシドオキシドヒドレート等が挙げられる。
触媒添加量及び触媒添加時期は、目的物を速やかに得られる条件であれば特に制限されない。
次に、タイヤ骨格体を構成する樹脂材料の好ましい物性について説明する。前記タイヤ骨格体は、上述の樹脂材料を用いるものである。
このように、融点が120℃~250℃の樹脂材料を用いることで、例えばタイヤの骨格体を、その分割体(骨格片)を融着して形成する場合に、120℃~250℃の周辺温度範囲で融着された骨格体であってもタイヤ骨格片同士の接着強度が十分である。このため、前記タイヤは耐パンク性や耐摩耗性など走行時における耐久性に優れる。尚、前記加熱温度は、タイヤ骨格片を形成する樹脂材料の融点(又は軟化点)よりも10℃~150℃高い温度が好ましく、10℃~100℃高い温度が更に好ましい。
溶融混合して得られた樹脂材料は、必要に応じてペレット状にして用いることができる。
以下に、図面に従って本発明の第1の実施形態に係るタイヤを説明する。
本実施形態のタイヤ10について説明する。図1Aは、本発明の一実施形態に係るタイヤの一部の断面を示す斜視図である。図1Bは、リムに装着したビード部の断面図である。図1Aに示すように、本実施形態のタイヤ10は、従来一般のゴム製の空気入りタイヤと略同様の断面形状を呈している。
また、本実施形態では、タイヤケース半体17Aは左右対称形状、即ち、一方のタイヤケース半体17Aと他方のタイヤケース半体17Aとが同一形状とされているので、タイヤケース半体17Aを成形する金型が1種類で済むメリットもある。
以下、本実施形態のタイヤの製造方法について説明する。
まず、上述のように前記ポリアミド系熱可塑性樹脂エラストマーを含む樹脂組成物を含む樹脂材料を用いて、タイヤケース半体を形成する。これらタイヤケースの形成は、射出成形で行うことが好ましい。次に、薄い金属の支持リングに支持されたタイヤケース半体同士を互いに向かい合わせる。次いで、タイヤケース半体の突き当て部分の外周面と接するように図を省略する接合金型を設置する。ここで、前記接合金型はタイヤケース半体17Aの接合部(突き当て部分)周辺を所定の圧力で押圧するように構成されている。次いで、タイヤケース半体の接合部周辺を、タイヤケースを構成する樹脂材料の融点(又は軟化点)以上で押圧する。タイヤケース半体の接合部が接合金型によって加熱や加圧されると、前記接合部が溶融しタイヤケース半体同士が融着しこれら部材が一体となってタイヤケース17が形成される。尚、本実施形態においては接合金型を用いてタイヤケース半体の接合部を加熱したが、本発明はこれに限定されず、例えば、別に設けた高周波加熱機等によって前記接合部を加熱してもよく、予め熱風、赤外線の照射等によって軟化又は溶融させ、接合金型によって加圧し、タイヤケース半体を接合させてもよい。
次に、図を省略するが、補強コード26を巻き付けたリール、コード加熱装置、各種ローラ等を備えたコード供給装置を用い、加熱した補強コード26をクラウン部16の外周面に埋設しながら巻き付けることで、タイヤケース17のクラウン部16の外周側に補強コード層28を形成することができる。
本実施形態のタイヤ10は、タイヤケース17が、ハードセグメントと、ポリプロピレングリコールに由来する構成単位からなるソフトセグメントと、2つ以上のセグメントを結合し炭素数6以上20以下のジカルボン酸に由来する構成単位からなる結合部と、を有し、重量平均分子量が20,000以上250,000以下であるポリアミド系熱可塑性エラストマーを含む樹脂材料によって形成される。このため、本実施形態のタイヤ10は、製造効率、低ロス性、耐熱性、及びリム組み性がバランスよく優れる。
攪拌機、窒素ガス導入口、及び縮合水排出口を備えた容積2リットルの反応容器に、1,2-アミノドデカン酸(アルドリッチ製)36.4g、アミノドデカノラクタム500g、ドデカン二酸(結合部となる鎖長延長剤)17.7gを入れ、容器内を十分窒素置換した後、280℃まで昇温し、0.4MPaの加圧下で4時間反応させた。圧力を解放したあと、窒素気流下でさらに1時間反応させ、分子量3700のナイロン12重合物である白色固体を得た(重合反応A)。
なお、得られたナイロン12重合物は、ハードセグメントである分子量3200のポリアミド(ナイロン12)の末端に鎖長延長剤であるドデカン二酸が結合したものである。
重合反応Bにおけるポリオキシプロピレンジアミンの添加量を131.3g、反応温度及び反応時間をそれぞれ230℃及び4時間に以外は、実施例1と同様にして、ポリアミド系熱可塑性エラストマーを得た。
添加するポリオキシプロピレンジアミンの量を175gとし、重合反応Bにおける反応温度及び反応時間をそれぞれ230℃及び5時間にした以外は、実施例1と同様にして、ポリアミド系熱可塑性エラストマーを得た。
添加するポリオキシプロピレンジアミンの量を127.3gとし、重合反応Bにおける反応温度及び反応時間をそれぞれ230℃及び5時間にした以外は、実施例1と同様にして、ポリアミド系熱可塑性エラストマーを得た。
添加するポリオキシプロピレンジアミンの量を107.7gとし、重合反応Bにおける反応温度及び反応時間をそれぞれ230℃及び5時間にした以外は、実施例1と同様にして、ポリアミド系熱可塑性エラストマーを得た。
添加するポリオキシプロピレンジアミンの量を82.4gとし、重合反応Bにおける反応温度及び反応時間をそれぞれ230℃及び5時間にした以外は、実施例1と同様にして、ポリアミド系熱可塑性エラストマーを得た。
用いる鎖長延長剤の種類及び添加量をアジピン酸14.0gとし、重合反応Aにおける反応温度及び反応時間をそれぞれ280℃及び4時間にし、重合反応Bにおける反応温度及び反応時間をそれぞれ230℃及び6.5時間にした以外は、実施例1と同様にして、ポリアミド系熱可塑性エラストマーを得た。
用いる鎖長延長剤の種類及び添加量をエイコサン二酸29.0gとし、重合反応Aにおける反応温度及び反応時間をそれぞれ280℃及び4時間にし、重合反応Bにおける反応温度及び反応時間をそれぞれ230℃及び5.5時間にした以外は、実施例1と同様にして、ポリアミド系熱可塑性エラストマーを得た。
添加するポリオキシプロピレンジアミンの量を107.7gとし、重合反応Bにおける反応温度及び反応時間をそれぞれ230℃及び6時間にした以外は、実施例1と同様にして、ポリアミド系熱可塑性エラストマーを得た。
添加するポリオキシプロピレンジアミンの量を107.7gとし、重合反応Bにおける反応温度及び反応時間をそれぞれ230℃及び7時間にした以外は、実施例1と同様にして、ポリアミド系熱可塑性エラストマーを得た。
添加するポリオキシプロピレンジアミンの量を107.7gとし、重合反応Bにおける反応温度及び反応時間をそれぞれ230℃及び8.5時間にした以外は、実施例1と同様にして、ポリアミド系熱可塑性エラストマーを得た。
添加するポリオキシプロピレンジアミンの量を127.3gとし、重合反応Bにおける反応温度及び反応時間をそれぞれ230℃及び6時間にした以外は、実施例1と同様にして、ポリアミド系熱可塑性エラストマーを得た。
添加するポリオキシプロピレンジアミンの量を93.3gとし、重合反応Bにおける反応温度及び反応時間をそれぞれ230℃及び6時間にした以外は、実施例1と同様にして、ポリアミド系熱可塑性エラストマーを得た。
ポリオキシプロピレンジアミンの代わりにポリプロピレングリコール(和光純薬工業(株)社製、品番:164-05895、数平均分子量:2000)を131.3g用い、重合触媒としてジルコニウムテトラブトキシド(東京化成工業(株)、品番:Z0016)を0.04g用い、重合反応Bにおける反応温度及び反応時間をそれぞれ260℃及び6時間にした以外は、実施例1と同様にして、ポリアミド系熱可塑性エラストマーを得た。
攪拌機、窒素ガス導入口、及び縮合水排出口を備えた容積2リットルの反応容器に、アルドリッチ製カプロラクタム500g、ドデカン二酸124g、アミノヘキサン酸77gを入れ、容器内を十分窒素置換した後、250℃まで昇温し、0.6MPaの加圧下で4時間反応させた。圧力を解放したあと、窒素気流下でさらに1時間反応させ、水洗工程を経て、数平均分子量3000のナイロン6(ポリアミド6)重合物である白色固体を得た。
攪拌機、窒素ガス導入口、及び縮合水排出口を備えた容積2リットルの反応容器に、ドデカン二酸207g、ヘキサメチレンジアミン93g、精製水100gを入れ、容器内を十分窒素置換した後、280℃まで昇温し、0.6MPaの加圧下で4時間反応させた。圧力を解放したあと、窒素気流下でさらに1時間反応させ、容器から取り出し、エタノールにて24時間ソックスレー抽出することで、数平均分子量3000のナイロン612(ポリアミド612)重合物である白色固体を得た。
ポリオキシプロピレンジアミンの代わりにABA型トリブロックポリエーテルジアミン(PPG-PTMG-PPGトリブロック重合体、HUNTSMAN社製、品番:XTJ-542、数平均分子量:1000)を113.8g用い、重合反応Bにおける反応温度及び反応時間をそれぞれ230℃及び6時間にした以外は、実施例1と同様にして、ポリアミド系熱可塑性エラストマーを得た。
なお「PPG」はポリプロピレングリコールに由来する構成単位を意味し、「PTMG」はポリテトラエチレンエーテルグリコールに由来する構成単位を意味する。
用いる鎖長延長剤の種類及び添加量を水添ダイマー酸(ユニケマ社製プリポール1009)45.2gとし、重合反応Aにおける反応温度及び反応時間をそれぞれ280℃及び4時間にし、重合反応Bにおける反応温度及び反応時間をそれぞれ230℃及び8時間にした以外は、実施例1と同様にして、ポリアミド系熱可塑性エラストマーを得た。
添加するポリオキシプロピレンジアミンの量を269.2gとし、重合反応Bにおける反応温度及び反応時間をそれぞれ250℃及び1時間にした以外は、実施例1と同様にして、ポリアミド系熱可塑性エラストマーを得た。
添加するポリオキシプロピレンジアミンの量を233.3gとし、重合反応Bにおける反応温度及び反応時間をそれぞれ250℃及び1時間にした以外は、実施例1と同様にして、ポリアミド系熱可塑性エラストマーを得た。
添加するポリオキシプロピレンジアミンの量を107.7gとし、重合反応Bにおける反応温度及び反応時間をそれぞれ230℃及び9時間にした以外は、実施例1と同様にして、ポリアミド系熱可塑性エラストマーを得た。
用いる鎖長延長剤の種類及び添加量をドコサン二酸29.2gとし、重合反応Aにおける反応温度及び反応時間をそれぞれ280℃及び4時間にし、重合反応Bにおける反応温度及び反応時間をそれぞれ230℃及び6時間にした以外は、実施例1と同様にして、ポリアミド系熱可塑性エラストマーを得た。
実施例及び比較例から得た熱可塑性エラストマーを用いて、以下の項目について評価した。具体的には、得られたポリアミド系熱可塑性エラストマーをペレット化し、200~240℃で射出成形し、サンプル片を得た。各種測定は、このサンプル片から試験片を打ち抜いたサンプルを用いて実施した。結果を表1~表4に示す。
粘弾性測定装置(レオメトリックス社製)を使用し、φ8mm、厚さ2mmのサンプルを用いて温度30℃、歪み1%、周波数20Hzで損失正接(tanδ)を測定し、以下の基準で評価した。なお、tanδが小さい程、低ロス性に優れている。
A:tanδが0.08以下
B:tanδが0.08より大きく0.12以下
C:tanδが0.12より大きい
式:換算値=(tanδ3/tanδ)×100
示差走査型熱量分析(DSC)装置〔ティー・エイ・インスツルメント・ジャパン株式会社製、DSC Q2000〕を用い、各実施例及び比較例で得られた熱可塑性エラストマーを、0℃から200℃まで10℃/分で昇温した。評価基準は以下の通りである。
A:融解開始温度が155℃以上
B:融解開始温度が130℃以上155℃未満
C:融解開始温度が130℃未満
射出成形機として住友重機械工業(株)製「SE30D」を用い、成形温度200℃~235℃、金型温度50℃~70℃、荷重15tの条件での射出成形を行い、射出開始から射出終了までの時間を測定した(計量時間)。得られた結果から、以下の基準で評価した。
A:荷重15tにおいて平板の成形が可能であり、かつ、ハンドリング性が良好である(計量時間が30秒未満)
B:荷重15tにおいて平板の成形が可能であるが、ハンドリング性が劣る(計量時間が30秒以上)
C:荷重15tにおいて充填不良が見られる。
各実施例及び比較例で得られた熱可塑性エラストマーを用いて、それぞれ上述の第1の実施形態を参照し、タイヤを形成した。次いで、タイヤをリムに装着し、以下の基準で評価した。
A:エアシール性が確保でき、かつ、ハンドリング性が良好
B:エアシール性の確保はできたが、リム組みの際に硬くてハンドリング性に劣る
C:エアシール性が確保できなかった
各実施例及び比較例で熱可塑性エラストマーを得る過程において、前記重合反応Bにおける重合時間に対するトルク変化の割合から、反応性を評価した。
具体的には、撹拌モーターのトルクが0.07N・mに達するまで要する時間を計測し、以下の基準で反応性を評価した。
A:1.5時間以内に到達する
B:2.0時間以内に到達する
C:3.0時間以上要する
各実施例及び比較例で得られた熱可塑性エラストマーについて前記評価(低ロス性、耐熱性、射出成形性、リム組み性、及び反応性)を行い、各評価において「A」のものを「+2点」、「B」のものを「+1点」、「C」のものを「0点」として合計10満点で総合評価を行った。総合評価が7点以上であれば、製造効率、低ロス性、耐熱性、及びリム組み性がバランスよく優れたタイヤが得られる。
各実施例及び比較例で得られた熱可塑性エラストマーについて、東ソー株式会社製の「HLC-8320GPC EcoSEC」を用いて、ゲルパーミエーションクロマトグラフィー(GPC)により測定した。結果を表1~表4に示す。
また、表1~表4中、「PA12」はナイロン12(ポリアミド12)に由来することを意味し、「PA6」はナイロン6(ポリアミド6)に由来することを意味し、「PA612」はナイロン612(ポリアミド612)に由来することを意味する。また「PPG」はポリプロピレングリコールに由来することを意味する。さらに、「PPG-PTMG-PPG」は、PTMG由来の構成単位とPPG由来の構成単位を有する三元共重合体を意味する。
本明細書に記載された全ての文献、特許出願、および技術規格は、個々の文献、特許出願、および技術規格が参照により取り込まれることが具体的かつ個々に記された場合と同程度に、本明細書中に参照により取り込まれる。
Claims (3)
- ポリアミド系熱可塑性エラストマーを含む樹脂材料で形成され且つ環状のタイヤ骨格体を有し、
前記ポリアミド系熱可塑性エラストマーは、ハードセグメントと、ポリプロピレングリコールに由来する構成単位からなるソフトセグメントと、2つ以上のセグメントを結合し炭素数6以上20以下のジカルボン酸に由来する構成単位からなる結合部と、を有し、前記ポリアミド系熱可塑性エラストマーの重量平均分子量が20,000以上250,000以下である、タイヤ。 - 前記ポリアミド系熱可塑性エラストマーにおける、前記ハードセグメントの質量(x)と前記ソフトセグメントの質量(y)との比(x/y)は、54/46~90/10である、請求項1に記載のタイヤ。
- 前記ポリアミド系熱可塑性エラストマーにおける、前記ハードセグメントの質量(x)と前記ソフトセグメントの質量(y)との比(x/y)は、54/46~88/12である、請求項1に記載のタイヤ。
Priority Applications (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2016550367A JP6723157B2 (ja) | 2014-09-24 | 2015-09-24 | タイヤ |
| EP15844608.8A EP3199567B1 (en) | 2014-09-24 | 2015-09-24 | Tire |
| US15/513,989 US10654975B2 (en) | 2014-09-24 | 2015-09-24 | Tire |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2014193427 | 2014-09-24 | ||
| JP2014-193427 | 2014-09-24 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2016047708A1 true WO2016047708A1 (ja) | 2016-03-31 |
Family
ID=55581227
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2015/076980 WO2016047708A1 (ja) | 2014-09-24 | 2015-09-24 | タイヤ |
Country Status (4)
| Country | Link |
|---|---|
| US (1) | US10654975B2 (ja) |
| EP (1) | EP3199567B1 (ja) |
| JP (1) | JP6723157B2 (ja) |
| WO (1) | WO2016047708A1 (ja) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20180086140A1 (en) * | 2015-04-10 | 2018-03-29 | Bridgestone Corporation | Polyamide-based thermoplastic elastomer and tire |
Families Citing this family (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2016047707A1 (ja) | 2014-09-24 | 2016-03-31 | 株式会社ブリヂストン | タイヤ |
| CN106715531B (zh) | 2014-09-29 | 2019-08-06 | 株式会社普利司通 | 轮胎 |
| CN107075113B (zh) * | 2014-09-29 | 2019-11-22 | 株式会社普利司通 | 轮胎 |
| CN106715530B (zh) | 2014-09-29 | 2019-07-16 | 株式会社普利司通 | 轮胎 |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS63151502A (ja) * | 1986-12-17 | 1988-06-24 | Okura Ind Co Ltd | 二輪車用軽量チユ−ブ |
| JPS63156855A (ja) * | 1986-12-22 | 1988-06-29 | Okura Ind Co Ltd | 空気保持性に優れた二輪車用軽量チユ−ブ |
| JP2009255489A (ja) * | 2008-03-27 | 2009-11-05 | Ube Ind Ltd | 多層構造体 |
| US20130059142A1 (en) * | 2010-03-31 | 2013-03-07 | Kolon Industries, Inc. | Film for a tyre inner liner and production method therefor |
| US20130192736A1 (en) * | 2011-06-29 | 2013-08-01 | Kolon Industries, Inc. | Film for a tire inner liner and preparation method therefor |
| JP2014062259A (ja) * | 2008-01-23 | 2014-04-10 | Ube Ind Ltd | チェーファー用ゴム組成物及びそれを用いたタイヤ |
Family Cites Families (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4700927A (en) | 1986-11-12 | 1987-10-20 | Rodney Hunt Company | Slide gate |
| US6590065B1 (en) * | 2001-12-10 | 2003-07-08 | E. I. Du Pont De Nemours And Company | Polytrimethylene ether ester amide and use thereof |
| JP5623825B2 (ja) | 2010-08-25 | 2014-11-12 | 株式会社ブリヂストン | タイヤの製造方法 |
| CN103958580B (zh) * | 2011-09-30 | 2017-03-08 | 可隆工业株式会社 | 用于轮胎内衬层的膜及其制备方法 |
| JP6333806B2 (ja) | 2013-03-29 | 2018-05-30 | 株式会社ブリヂストン | タイヤ |
| JP6086782B2 (ja) | 2013-03-29 | 2017-03-01 | 株式会社ブリヂストン | タイヤ |
| CN105073834A (zh) | 2013-03-29 | 2015-11-18 | 株式会社普利司通 | 轮胎 |
| JP6001488B2 (ja) | 2013-03-29 | 2016-10-05 | 株式会社ブリヂストン | タイヤ |
| WO2016047709A1 (ja) | 2014-09-24 | 2016-03-31 | 株式会社ブリヂストン | タイヤ |
| CN107075113B (zh) | 2014-09-29 | 2019-11-22 | 株式会社普利司通 | 轮胎 |
| CN106715531B (zh) | 2014-09-29 | 2019-08-06 | 株式会社普利司通 | 轮胎 |
| CN106715530B (zh) | 2014-09-29 | 2019-07-16 | 株式会社普利司通 | 轮胎 |
-
2015
- 2015-09-24 WO PCT/JP2015/076980 patent/WO2016047708A1/ja active Application Filing
- 2015-09-24 EP EP15844608.8A patent/EP3199567B1/en active Active
- 2015-09-24 US US15/513,989 patent/US10654975B2/en active Active
- 2015-09-24 JP JP2016550367A patent/JP6723157B2/ja active Active
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS63151502A (ja) * | 1986-12-17 | 1988-06-24 | Okura Ind Co Ltd | 二輪車用軽量チユ−ブ |
| JPS63156855A (ja) * | 1986-12-22 | 1988-06-29 | Okura Ind Co Ltd | 空気保持性に優れた二輪車用軽量チユ−ブ |
| JP2014062259A (ja) * | 2008-01-23 | 2014-04-10 | Ube Ind Ltd | チェーファー用ゴム組成物及びそれを用いたタイヤ |
| JP2009255489A (ja) * | 2008-03-27 | 2009-11-05 | Ube Ind Ltd | 多層構造体 |
| US20130059142A1 (en) * | 2010-03-31 | 2013-03-07 | Kolon Industries, Inc. | Film for a tyre inner liner and production method therefor |
| US20130192736A1 (en) * | 2011-06-29 | 2013-08-01 | Kolon Industries, Inc. | Film for a tire inner liner and preparation method therefor |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20180086140A1 (en) * | 2015-04-10 | 2018-03-29 | Bridgestone Corporation | Polyamide-based thermoplastic elastomer and tire |
Also Published As
| Publication number | Publication date |
|---|---|
| EP3199567B1 (en) | 2018-11-14 |
| EP3199567A4 (en) | 2017-08-02 |
| EP3199567A1 (en) | 2017-08-02 |
| US20170291991A1 (en) | 2017-10-12 |
| JP6723157B2 (ja) | 2020-07-15 |
| US10654975B2 (en) | 2020-05-19 |
| JPWO2016047708A1 (ja) | 2017-07-06 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6333806B2 (ja) | タイヤ | |
| JP6086782B2 (ja) | タイヤ | |
| JP6001488B2 (ja) | タイヤ | |
| JP6723157B2 (ja) | タイヤ | |
| JP6560973B2 (ja) | タイヤ | |
| WO2016047710A1 (ja) | タイヤ | |
| WO2016047707A1 (ja) | タイヤ | |
| WO2014157559A1 (ja) | タイヤ | |
| JPWO2016047709A1 (ja) | タイヤ | |
| JP6001813B2 (ja) | タイヤ | |
| JP6001812B2 (ja) | タイヤ | |
| WO2017146069A1 (ja) | タイヤ | |
| JP5989280B1 (ja) | タイヤ骨格体およびタイヤ | |
| JP2016084424A (ja) | タイヤ | |
| WO2016195112A1 (ja) | タイヤ | |
| JP5989279B1 (ja) | タイヤ骨格体およびタイヤ | |
| JP6474273B2 (ja) | タイヤ | |
| JP6694263B2 (ja) | タイヤ | |
| JP6373652B2 (ja) | 積層体及びこれを用いたタイヤ |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 15844608 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2016550367 Country of ref document: JP Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 15513989 Country of ref document: US |
|
| REEP | Request for entry into the european phase |
Ref document number: 2015844608 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2015844608 Country of ref document: EP |