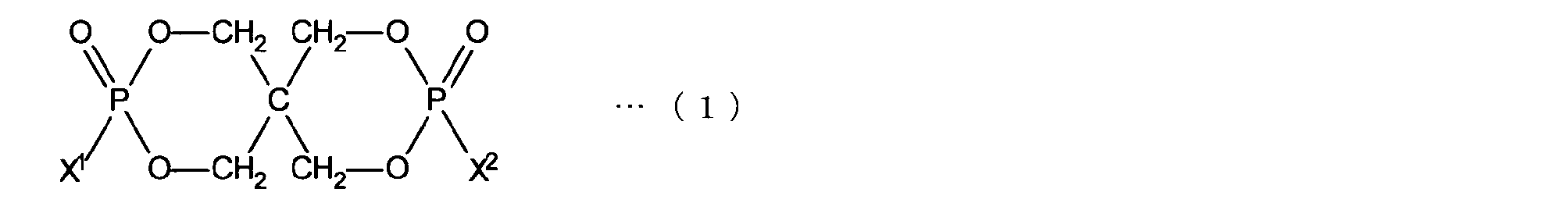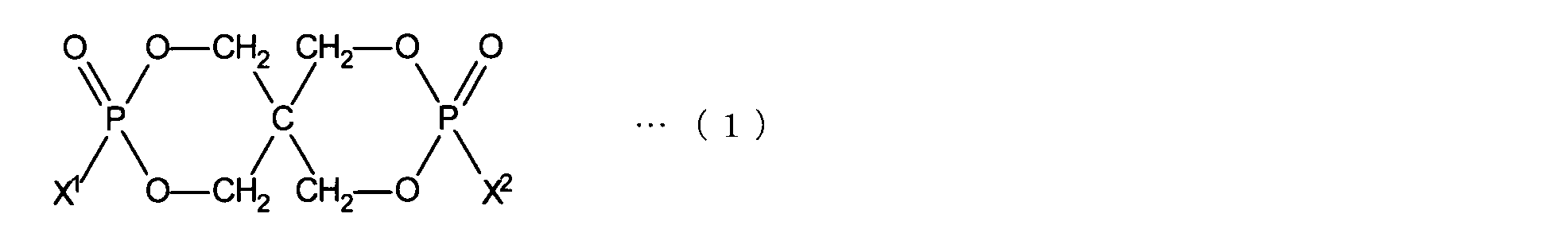WO2016017571A1 - 難燃性樹脂組成物およびそれからの成形品 - Google Patents
難燃性樹脂組成物およびそれからの成形品 Download PDFInfo
- Publication number
- WO2016017571A1 WO2016017571A1 PCT/JP2015/071216 JP2015071216W WO2016017571A1 WO 2016017571 A1 WO2016017571 A1 WO 2016017571A1 JP 2015071216 W JP2015071216 W JP 2015071216W WO 2016017571 A1 WO2016017571 A1 WO 2016017571A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- component
- resin
- resin composition
- flame retardant
- composition according
- Prior art date
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K5/00—Use of organic ingredients
- C08K5/49—Phosphorus-containing compounds
- C08K5/51—Phosphorus bound to oxygen
- C08K5/53—Phosphorus bound to oxygen bound to oxygen and to carbon only
- C08K5/5317—Phosphonic compounds, e.g. R—P(:O)(OR')2
- C08K5/5333—Esters of phosphonic acids
- C08K5/5357—Esters of phosphonic acids cyclic
-
- D—TEXTILES; PAPER
- D01—NATURAL OR MAN-MADE THREADS OR FIBRES; SPINNING
- D01F—CHEMICAL FEATURES IN THE MANUFACTURE OF ARTIFICIAL FILAMENTS, THREADS, FIBRES, BRISTLES OR RIBBONS; APPARATUS SPECIALLY ADAPTED FOR THE MANUFACTURE OF CARBON FILAMENTS
- D01F1/00—General methods for the manufacture of artificial filaments or the like
- D01F1/02—Addition of substances to the spinning solution or to the melt
- D01F1/07—Addition of substances to the spinning solution or to the melt for making fire- or flame-proof filaments
-
- D—TEXTILES; PAPER
- D01—NATURAL OR MAN-MADE THREADS OR FIBRES; SPINNING
- D01F—CHEMICAL FEATURES IN THE MANUFACTURE OF ARTIFICIAL FILAMENTS, THREADS, FIBRES, BRISTLES OR RIBBONS; APPARATUS SPECIALLY ADAPTED FOR THE MANUFACTURE OF CARBON FILAMENTS
- D01F6/00—Monocomponent artificial filaments or the like of synthetic polymers; Manufacture thereof
- D01F6/02—Monocomponent artificial filaments or the like of synthetic polymers; Manufacture thereof from homopolymers obtained by reactions only involving carbon-to-carbon unsaturated bonds
- D01F6/04—Monocomponent artificial filaments or the like of synthetic polymers; Manufacture thereof from homopolymers obtained by reactions only involving carbon-to-carbon unsaturated bonds from polyolefins
Definitions
- the present invention relates to a flame retardant resin composition having flame retardancy and good physical properties, and a molded product formed therefrom. More particularly, the present invention relates to a flame-retardant polyolefin-based resin composition containing a pentaerythritol diphosphonate compound having specific characteristics and substantially halogen-free, and a molded article therefrom.
- Polyolefin resins are used in a wide range of applications due to their characteristics such as good processability, good physical properties, and low specific gravity.
- polyolefin resins have various resin types including polyethylene and polypropylene, and it is possible to select physical properties suitable for various applications.
- polyolefin-based resins are easily combustible materials, various flame retardant studies have been conducted so far.
- non-halogen-based flame retardants As an example of such a flame retardant, conventionally, studies have been made to add a halogen-based flame retardant, but in recent years, non-halogen-based flame retardants have been actively studied due to environmental problems such as generation of toxic gas during combustion. Yes.
- the most frequently practiced non-halogen flame retardant technology for polyolefin resins is a method of adding a metal hydroxide such as magnesium hydroxide or aluminum hydroxide. This flame-retarding technique has been reported in particular as a technique for wire coating materials, but these flame-retardant resin compositions containing magnesium hydroxide, aluminum hydroxide and the like are used to obtain flame-retardant performance.
- Patent Document 1 An in-messentic flame retardant that uses a specific phosphate flame retardant to form a foam layer on the surface of the molded product during combustion, and suppresses the diffusion and heat transfer of decomposition products and exhibits flame retardancy.
- Patent Document 1 A flame retardant has been proposed (Patent Document 1).
- the intumescent flame retardant has excellent flame retardancy, there are problems such as poor dispersion in the resin due to secondary aggregation and deterioration of hygroscopicity due to hydrolysis. For this reason, improvements have been made by co-adding compounds such as phosphate esters, silicone oils and polycarbodiimides having a specific molecular structure, but satisfactory properties have not yet been achieved (Patent Documents 2 to 4).
- the flame retarding technology described above is not a technology that can be applied to various polyolefin resins in general, but a technology that can be applied only to specific resin types among polyolefin resins.
- the high heat-resistant polyolefin resin represented by polymethylpentene resin has a high processing temperature and cannot be handled by a phosphate ester flame retardant.
- An object of the present invention is to provide a flame retardant resin composition having a high degree of flame retardancy and good physical properties, in particular, no deterioration in heat resistance, and a product comprising the same.
- the organophosphorus compound (B) represented by the following formula (1) with respect to 100 parts by weight of the polyolefin-based resin (component A).
- the organic phosphorus compound has an organic purity of 97.0% or more, a chlorine content of 1000 ppm or less, a ⁇ pH of 1.0 or less, and a residual solvent amount of 1000 ppm. It has been found that a polyolefin resin composition capable of achieving both high flame retardancy and good physical properties can be provided by the following.
- AL is a branched or straight-chain aliphatic hydrocarbon group having 1 to 5 carbon atoms
- Ar is a phenyl group, naphthyl group or anthryl group which may have a substituent on the aromatic ring.
- N represents an integer of 1 to 3, and Ar can be bonded to any carbon atom in AL.
- a flame retardant polyolefin-based resin composition which does not substantially contain halogen and has high flame retardancy and good physical properties, particularly no deterioration in heat resistance.
- the polyolefin-based resin used as the component A in the present invention is not particularly limited.
- homopolymers such as ethylene, propylene, 1-butene, and the like, or each of them and 3-methyl-1-butene
- Other ⁇ -olefins having about 2 to 20 carbon atoms such as 1-pentene, 4-methyl-1-pentene, 1-hexene, 1-octene, 1-decene, vinyl acetate, vinyl alcohol, acrylic acid, methacrylic acid , Copolymers with acrylic acid esters, methacrylic acid esters and the like.
- polyolefin resin examples include, for example, ethylene homopolymers such as low, medium and high density polyethylene (branched or linear), ethylene-propylene copolymers, ethylene-1-butene copolymers, ethylene -4-methyl-1-pentene copolymer, ethylene-1-hexene copolymer, ethylene-1-octene copolymer, ethylene-vinyl alcohol copolymer, ethylene-acrylic acid copolymer, ethylene-methacrylic acid Polyethylene resins such as copolymers, polypropylene resins such as propylene homopolymers, propylene-ethylene copolymers, propylene-ethylene-1-butene copolymers, 1-butene homopolymers, 1-butene- Examples thereof include poly 1-butene resins such as ethylene copolymers and 1-butene-propylene copolymers.
- ethylene homopolymers such as low, medium and high density polyethylene (branched or linear)
- polyolefin resins may be modified with an unsaturated carboxylic acid such as maleic anhydride, maleic acid or acrylic acid, or a derivative thereof, an unsaturated silane compound, or the like. Furthermore, you may have a crosslinked structure partially.
- unsaturated carboxylic acid such as maleic anhydride, maleic acid or acrylic acid, or a derivative thereof, an unsaturated silane compound, or the like.
- the polyethylene-based resin means a polymer containing ethylene as a main component as a raw material monomer, and preferably containing 50% by weight or more of ethylene.
- the polypropylene resin means a polymer containing propylene as a main component as a raw material monomer, and preferably containing 50% by weight or more of propylene.
- the poly 1-butene resin means a polymer containing 1-butene as a main component as a main component, and preferably containing 50% by weight or more of 1-butene.
- the chain form in the case of using a copolymer as the polyolefin-based resin is not limited, and any of a block copolymer, a random copolymer, a graft copolymer, and the like may be used. Moreover, a well-known thing can also be used suitably for the polymerization method and the catalyst used for superposition
- polyolefin resins may be used alone or in a mixture of two or more.
- Preferred examples of the polyolefin resin include 60% by weight or more of at least one resin selected from a polyethylene resin, a polypropylene resin, a poly-1-butene resin, and a poly-4-methyl-1-pentene resin.
- a resin component is preferred.
- a melt flow rate measured at 190 ° C. and a load of 2.16 kg in accordance with JIS K7210 standard is preferably 50 g / 10 min or less, more preferably a melt flow rate. Those having a viscosity of 30 g / 10 min or less, more preferably those having a melt flow rate of 25 g / 10 min are suitable. When a melt flow rate higher than 50 g / 10 min is used, a large amount of dripping material is produced when the molded product is burned, and the desired flame retardancy may not be obtained.
- those having a melt flow rate measured at 230 ° C. and a load of 2.16 kg according to JIS K7210 standard are preferably 0.1 to 50 g / 10 min, more preferably melt Suitable are those having a flow rate of 0.2 to 45 g / 10 min, more preferably having a melt flow rate of 0.3 to 40 g / 10 min, and particularly preferably having a melt flow rate of 10 to 35 g / 10 min. Yes.
- a melt flow rate higher than 50 g / 10 min is used, a large amount of dripping material is produced when the molded product is burned, and the desired flame retardancy may not be obtained.
- Preferred examples of the poly-1-butene resin in the present invention are those having a melt flow rate of 20 g / 10 min or less measured at 190 ° C. and a load of 2.16 kg in accordance with JIS K7210 standard, more preferably A flow rate of 15 g / 10 min or less, more preferably a melt flow rate of 10 g / 10 min or less is suitable.
- Preferred examples of the poly-4-methyl-1-pentene resin in the present invention are those having a melt flow rate of 1 to 200 g / 10 min measured at 260 ° C. and a 5.0 kg load in accordance with JIS K7210 standard. More preferably, the melt flow rate is 5 to 180 g / 10 minutes, more preferably the melt flow rate is 5 to 150 g / 10 minutes, and particularly preferably the melt flow rate is 7 to 50 g / 10 minutes. ing.
- a component polyolefin-based resin may contain other thermoplastic resins (polycarbonate resin, polyarylate resin, polyamide resin, polyimide resin, polyetherimide resin, polyurethane resin, silicone resin, polyphenylene sulfide resin, polysulfone resin, polystyrene as necessary.
- AS resin acrylonitrile / styrene copolymer
- ABS resin acrylonitrile / butadiene / styrene copolymer
- polystyrene resin high impact polystyrene resin, syndiot
- AL is a branched or straight-chain aliphatic hydrocarbon group having 1 to 5 carbon atoms
- Ar is a phenyl group, naphthyl group or anthryl group which may have a substituent on the aromatic ring.
- N represents an integer of 1 to 3, and Ar can be bonded to any carbon atom in AL.
- it is an organic phosphorus compound represented by the following formula (3).
- the organophosphorus compound (component B) represented by the formula (1) exhibits an extremely excellent flame retardant effect on the resin.
- component B represented by the formula (1)
- the organophosphorus compound (component B) can be easily made flame retardant by itself in a small amount, and does not impair the original properties of the resin.
- a fluorine-containing resin or other additive is used to reduce the use ratio of the B component, improve the flame retardancy of the molded product, Naturally, it can be added for the purpose of improving the chemical properties of molded articles, improving the chemical properties of molded articles, or other purposes.
- the component B may be produced by a method other than the method described below.
- the B component can be obtained, for example, by reacting pentaerythritol with phosphorus trichloride, subsequently treating the oxidized reaction product with an alkali metal compound such as sodium methoxide, and then reacting with aralkyl halide.
- the organic phosphorus compound (component B) described above has an organic purity measured by HPLC of preferably 97.0% or more, more preferably 98.0% or more, and even more preferably 99.0% or more. Is done.
- the organic purity affects the flame retardancy of the obtained resin composition, and when the organic purity is low, high flame retardancy cannot be obtained.
- B component with low organic purity will exhibit the hue deterioration of the resin composition obtained by the influence of the impurity, the fall of a physical property, especially a heat resistant fall.
- the organic purity of the B component by HPLC can be measured effectively by using the following method.
- the column was Develosil ODS-7 300 mm ⁇ 4 mm ⁇ manufactured by Nomura Chemical Co., Ltd., and the column temperature was 40 ° C.
- a solvent a mixed solution of acetonitrile and water 6: 4 (volume ratio) was used, and 5 ⁇ l was injected.
- the detector used was UV-264 nm.
- the method for removing impurities in component B is not particularly limited, but a method of performing repulp washing with a solvent such as water or methanol (washing with a solvent, repeating filtration several times) is the most effective. It is also advantageous in terms of cost. More effective cleaning is possible by stirring while heating the mixture of the component B and the solvent during cleaning.
- a solvent such as water or methanol
- the component B preferably has a chlorine content of 1000 ppm or less, more preferably 500 ppm or less, and still more preferably 100 ppm or less.
- One of the objects of the present invention is to provide a non-halogen flame retardant resin composition. Therefore, it is preferable to use a B component having a chlorine content within this range. Furthermore, by using the B component having a chlorine content in this range, a molded product having good thermal stability can be obtained, and a molded product having excellent hue can be obtained. When the chlorine content exceeds this range, the thermal stability of the resin composition is lowered, and the hue is lowered due to the occurrence of burns during high temperature molding.
- the chlorine content of the B component can be effectively measured by analyzing by a combustion method and detecting by a titration method in accordance with ASTM D5808.
- Component B preferably has a ⁇ pH of 1.0 or less, more preferably 0.8 or less, even more preferably 0.5 or less, and particularly preferably 0.3 or less. .
- a molded product having good thermal stability can be obtained, and a molded product having excellent hue can be obtained.
- ⁇ pH exceeds this range, the thermal stability of the resin composition is lowered, and the hue is lowered due to the occurrence of burns during high temperature molding.
- the ⁇ pH of the B component can be effectively measured by using the following method.
- ⁇ pH of the present invention can be calculated by the following formula (4).
- the component B preferably has a residual solvent amount of 1000 ppm or less, more preferably 800 ppm or less, still more preferably 500 ppm or less, particularly preferably 100 ppm or less.
- a resin composition having a high degree of flame retardancy can be obtained.
- Polyolefin resins generally have low flame retardancy, and when a B component whose residual solvent amount exceeds this range is used, it becomes difficult to obtain desired flame retardancy.
- the residual solvent amount of component B can be effectively measured by the same method as the organic purity measurement method using HPLC.
- the component B is 1 to 100 parts by weight, preferably 5 to 90 parts by weight, more preferably 10 to 70 parts by weight, and still more preferably 10 to 50 parts by weight with respect to 100 parts by weight of the polyolefin resin (component A).
- the preferred range of the blending ratio of the B component is determined by the desired flame retardancy level, the type of polyolefin resin (A component), and the like. Even if it is other than component A and component B constituting these compositions, other components can be used as necessary as long as they do not impair the purpose of the present invention. Other flame retardants, flame retardant aids, fluorine-containing compounds
- the blending amount of the B component can be changed also by using the resin, and in many cases, the blending ratio of the B component can be reduced by using these resins.
- the flame retardant resin composition of the present invention includes an antioxidant, a heat stabilizer, a stabilizer such as a light stabilizer, a nucleating agent, an inorganic filler, and an organic filler as long as the object of the present invention is not impaired.
- the flame-retardant resin composition of the present invention is prepared by mixing a polyolefin resin (component A), an organophosphorus compound (component B) and other components as required, such as a V-type blender, super mixer, super floater, Henschel mixer, etc.
- a method of premixing using such a mixer, supplying the premixed mixture to a kneader, and melt mixing is preferably employed.
- various melt mixers such as a kneader, a single screw or a twin screw extruder can be used, and in particular, a resin composition is melted at a recommended temperature of various polyolefin resins using a twin screw extruder.
- a method in which a liquid component is injected by a side feeder, extruded, and pelletized by a pelletizer is preferably used.
- the molding method of the molded product is not particularly limited, such as injection molding, blow molding, press molding, etc., but preferably it is molded by injection molding a pellet-shaped resin composition using an injection molding machine. .
- the fiber spinning method is not particularly limited, such as a melt spinning method, a dry spinning method, or a wet spinning method, but in the case of polyolefin resins, the melt spinning method is generally preferably used.
- the method for producing the nonwoven fabric is not particularly limited.
- a fleece formed by a fleece forming method such as a dry method, a wet method, a spun bond method, a melt blow method, a thermal bond method, a chemical bond method, a needle punch method.
- a fleece bonding method such as a spunlace method, a stitch bond method, and a steam jet method.
- a spunbond method or a thermal bond method is preferably used.
- ⁇ pH
- Develosil ODS-7 300 mm ⁇ 4 mm ⁇ manufactured by Nomura Chemical Co., Ltd. was used, and the column temperature was 40 ° C.
- a solvent a mixed solution of acetonitrile and water 6: 4 (volume ratio) was used, and 5 ⁇ l was injected.
- the detector used was UV-264 nm.
- the amount of residual solvent was calculated using a separately prepared calibration curve.
- (6) Hue A molded product having a thickness of 2 mm was prepared by injection molding and visually judged according to the following criteria.
- Heat resistance retention rate (deflection temperature retention rate under load; HDT retention rate)
- the deflection temperature under load (HDT) was measured at a load of 0.45 MPa using a 6.35 mm (1/4 inch) test piece by a method according to ASTM-D648.
- the load deflection temperature retention ratio (M) is determined from the load deflection temperature x (° C.) of the molded product from the used base resin (component A) and the flame retardant resin composition (mixture of base resin and component B).
- the yield was 21.33 g, the yield was 95%, and the 31 PNMR purity was 95%.
- the organic purity measured by the method described in the text was 94%.
- the chlorine content was 2500 ppm. ⁇ pH was 1.5.
- the amount of residual solvent was 1100 ppm.
- TPI DX845 Commercially available poly-4-methyl-1-pentene resin (TPI DX845 manufactured by Mitsui Chemicals; melt flow rate (260 ° C., 5.0 kg load) is 9.6 g / 10 min)) (hereinafter referred to as TPX-1) Called).
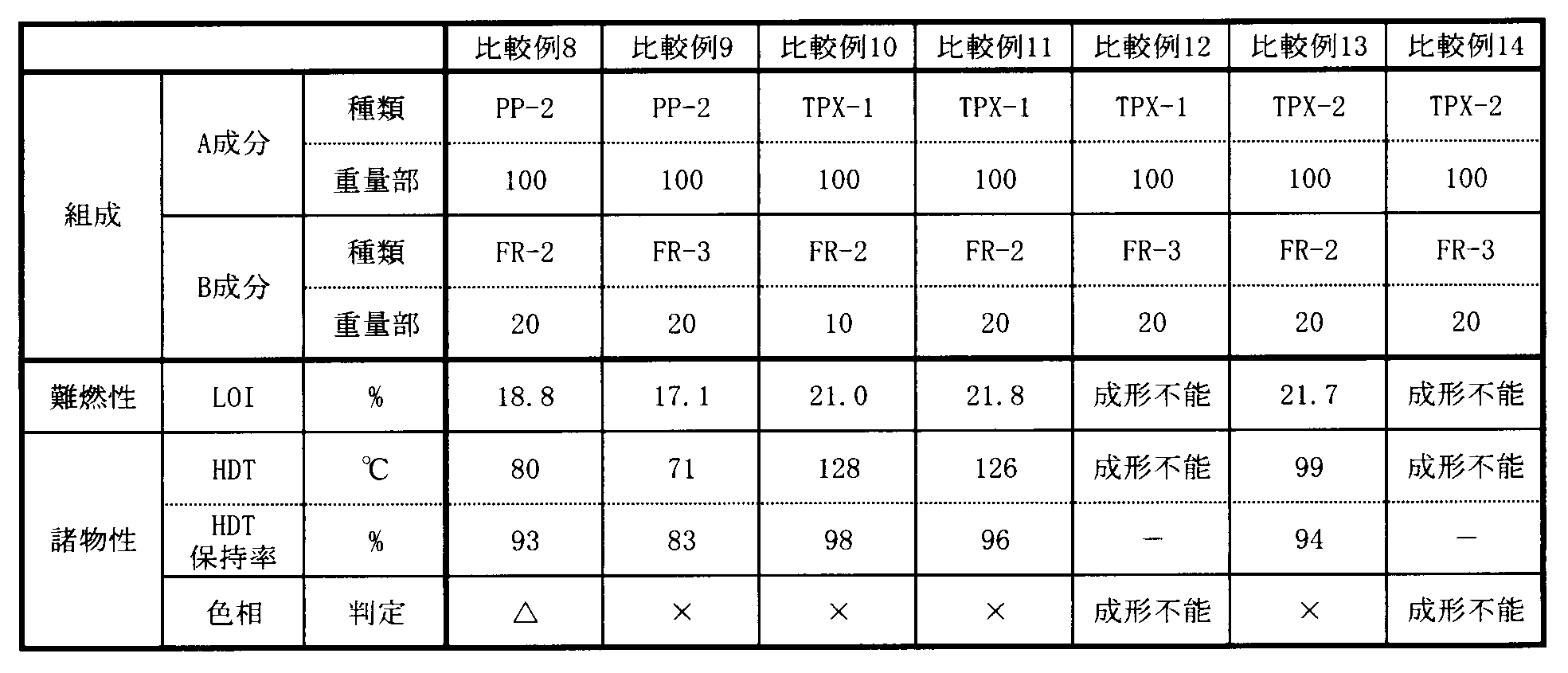
- Examples 1 to 8 and Comparative Examples 1 to 14 Each component described in Tables 1 and 2 is blended in the amount (parts by weight) described in Tables 1 and 2 with a tumbler, pelletized with a 15 mm ⁇ twin screw extruder (Technobel, KZW15), and the resulting pellets are injection molded. Each test piece was formed with a machine (manufactured by Nippon Steel Works, J75Si). The results of evaluation using this test piece are shown in Tables 1 and 2.
- the present invention provides a flame retardant polyolefin-based resin composition that does not substantially contain halogen and has high flame retardancy and good physical properties, in particular, no deterioration in heat resistance. Is useful as a material for various molded articles, fibers and nonwoven fabrics, and is extremely useful industrially.
Landscapes
- Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Engineering & Computer Science (AREA)
- General Chemical & Material Sciences (AREA)
- Textile Engineering (AREA)
- Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
- Organic Chemistry (AREA)
- Manufacturing & Machinery (AREA)
- Compositions Of Macromolecular Compounds (AREA)
- Nonwoven Fabrics (AREA)
Abstract
Description
(ポリオレフィン系樹脂(A成分))
本発明においてA成分として使用するポリオレフィン系樹脂は、特に限定されるものではないが、例えば、エチレン、プロピレン、1-ブテン等の単独重合体、それら同士あるいはそれらと3-メチル-1-ブテン、1-ペンテン、4-メチル-1-ペンテン、1-ヘキセン、1-オクテン、1-デセン等の炭素数2~20程度の他のα-オレフィンや、酢酸ビニル、ビニルアルコール、アクリル酸、メタクリル酸、アクリル酸エステル、メタクリル酸エステル等との共重合体等が挙げられる。
(有機リン化合物(B成分))
本発明において、B成分として使用する有機リン化合物は、下記式(1)で表される有機リン化合物である。
好ましくは下記式(3)で表される有機リン化合物である。
(B成分中の前記式(3)の有機リン化合物の合成法)
ペンタエリスリトールに三塩化リンを反応させ、次いでターシャリーブタノールにより酸化させた反応物を、ナトリウムメトキシドにより処理し、ベンジルブロマイドを反応させることにより得ることができる。
さらにB成分は、その残存溶媒量が1000ppm以下のものが好ましく、より好ましくは800ppm以下のもの、さらに好ましくは500ppm以下のもの、特に好ましくは100ppm以下のものが好適に使用できる。残存溶媒量がこの範囲のB成分を用いることにより、高度な難燃性を有する樹脂組成物を得ることができる。ポリオレフィン系樹脂は一般に難燃性が低く、残存溶媒量がこの範囲を超えるB成分を用いた場合、所望の難燃性を得ることが困難となる。
(1)有機純度
カラムは野村化学(株)製Develosil ODS-7 300mm×4mmφを用い、カラム温度は40℃とした。溶媒としてはアセトニトリルと水の6:4(容量比)の混合溶液を用い、5μlを注入した。検出器はUV-264nmを用いた。測定結果より、面積比をもって有機純度とした。
(2)塩素含有量
ASTM D5808に準拠し、燃焼法にて分析を行い、滴定法にて検出した。
(3)ΔpH
蒸留水99gと分散剤(エタノール)1gを混合し、1分間撹拌後、pH計にてpHを測定する(得られたpH値をpH1とする)。前記蒸留水と分散剤の混合溶液にB成分の有機リン化合物1gを添加し、1分間撹拌する。撹拌後の混合物を濾過し、濾液のpHをpH計にて測定する(得られたpH値をpH2とする)。ΔpHは下記式(4)により算出した。
(4)残存溶媒量
カラムは野村化学(株)製Develosil ODS-7 300mm×4mmφを用い、カラム温度は40℃とした。溶媒としてはアセトニトリルと水の6:4(容量比)の混合溶液を用い、5μlを注入した。検出器はUV-264nmを用いた。別途作成した検量線を用い、残存溶媒量を算出した。
(5)酸素指数
JIS-K-7201に準拠して行った。数値が高いほど難燃性に優れる。
(6)色相
射出成形により2mm厚の成形品を作成し、下記の基準にて目視判定した。
△:若干のヤケが認められるもの
×:明らかなヤケが認められるもの
(7)耐熱性保持率(荷重たわみ温度保持率;HDT保持率)
ASTM-D648に準拠した方法により6.35mm(1/4インチ)試験片を用いて0.45MPa荷重で荷重たわみ温度(HDT)を測定した。また、荷重たわみ温度保持率(M)は、使用したベース樹脂(A成分)からの成形品の荷重たわみ温度x(℃)と難燃性樹脂組成物(ベース樹脂とB成分の混合物)からの成形品の荷重たわみ温度y(℃)を測定し、M=(y/x)×100(%)の計算式により算出した。
[調製例1]
2,4,8,10-テトラオキサ-3,9-ジホスファスピロ[5.5]ウンデカン,3,9-ジベンジル-3,9-ジオキサイド(FR-1)の調製
攪拌機、温度計、コンデンサーを有する反応容器に、3,9-ジベンジロキシ-2,4,8,10-テトラオキサ-3,9-ジホスファスピロ[5.5]ウンデカン22.55g(0.055モル)、ベンジルブロマイド19.01g(0.11モル)およびキシレン33.54g(0.32モル)を充填し、室温下攪拌しながら、乾燥窒素をフローさせた。次いでオイルバスで加熱を開始し、還流温度(約130℃)で4時間加熱、攪拌した。加熱終了後、室温まで放冷し、キシレン20mLを加え、さらに30分攪拌した。析出した結晶をろ過により分離し、キシレン40mLで2回洗浄した。得られた粗精製物とメタノール50mLをコンデンサー、攪拌機を備えた反応容器に入れ、約3時間還流した。室温まで冷却後、結晶をろ過により分離し、メタノール20mLで2回洗浄した後、得られたろ取物を120℃、1.33×102Paで20時間乾燥し、白色の鱗片状結晶を得た。生成物は質量スペクトル分析、1H、31P核磁気共鳴スペクトル分析および元素分析でビスベンジルペンタエリスリトールジホスホネートであることを確認した。収量は19.76g、収率は88%、31PNMR純度は99%であった。また、本文記載の方法で測定した有機純度は99.5%であった。塩素含有量は51ppmであった。ΔpHは0.1であった。残留溶媒量は47ppmであった。
[調製例2]
2,4,8,10-テトラオキサ-3,9-ジホスファスピロ[5.5]ウンデカン,3,9-ジベンジル-3,9-ジオキサイド(FR-2)の調製
キシレン40mlで2回洗浄とメタノール還流洗浄の操作を省略した他は、調製例1と同様の調製方法にて調製した。
(イ)ポリオレフィン系樹脂(A成分)
(i)市販のポリプロピレン樹脂(プライムポリマー製プライムポリプロJ106G;メルトフローレート(230℃、2.16kg荷重)は15.4g/10分)を用いた(以下PP-1と称する)。
(ロ)有機リン化合物(B成分)
(i)調製例1で合成した2,4,8,10-テトラオキサ-3,9-ジホスファスピロ[5,5]ウンデカン,3,9-ジベンジル-3,9-ジオキサイド
前記一般式(3)で示される有機リン系化合物(以下FR-1と称する)。
前記一般式(3)で示される有機リン系化合物(以下FR-2と称する)。
(ハ)その他の有機リン化合物
市販の芳香族リン酸エステル系難燃剤(大八化学工業製PX-200)を用いた(以下FR-3と称する)。
[実施例1~8および比較例1~14]
表1および2記載の各成分を表1および2記載の量(重量部)でタンブラーにて配合し、15mmφ二軸押出機(テクノベル製、KZW15)にてペレット化し、得られたペレットを射出成形機(日本製鋼所製、J75Si)にて各試験片を成形した。この試験片を用いて評価した結果を表1および表2に示した。
Claims (11)
- ポリオレフィン系樹脂(A成分)100重量部に対して、下記式(1)で表される有機リン化合物(B成分)1~100重量部を含み、該有機リン化合物は、有機純度が97.0%以上であり、塩素含有量が1000ppm以下であり、ΔpHが1.0以下であり、残存溶媒量が1000ppm以下である難燃性樹脂組成物。
- A成分のポリオレフィン系樹脂が、ポリエチレン系樹脂、ポリプロピレン系樹脂、ポリ1-ブテン系樹脂、ポリ4-メチル-1-ペンテン系樹脂から選択される少なくとも1種の樹脂を60重量%以上含有する樹脂成分である請求項1記載の難燃性樹脂組成物。
- B成分の有機リン化合物は、有機純度が98.0%以上であり、塩素含有量が500ppm以下であり、ΔpHが0.8以下であり、残存溶媒量が800ppm以下である請求項1記載の難燃性樹脂組成物。
- A成分のポリオレフィン系樹脂が、JIS K7210規格に準じ、190℃、2.16kg荷重にて測定したメルトフローレートが50g/10分以下のポリエチレン系樹脂を少なくとも60重量%以上含有する樹脂成分である請求項1記載の難燃性樹脂組成物。
- A成分のポリオレフィン系樹脂が、JIS K7210規格に準じ、230℃、2.16kg荷重にて測定したメルトフローレートが0.1~50g/10分のポリプロピレン系樹脂を少なくとも60重量%以上含有する樹脂成分である請求項1記載の難燃性樹脂組成物。
- A成分のポリオレフィン系樹脂が、JIS K7210規格に準じ、190℃、2.16kg荷重にて測定したメルトフローレートが20g/10分以下のポリ1-ブテン系樹脂を少なくとも60重量%以上含有する樹脂成分である請求項1記載の難燃性樹脂組成物。
- A成分のポリオレフィン系樹脂が、JIS K7210規格に準じ、260℃、5.0kg荷重にて測定したメルトフローレートが1~200g/10分のポリ4-メチル-1-ペンテン系樹脂を少なくとも60重量%以上含有する樹脂成分である請求項1記載の難燃性樹脂組成物。
- 請求項1記載の難燃性樹脂組成物より形成された成形品。
- 請求項1記載の難燃性樹脂組成物を紡糸することにより得られた繊維および繊維製品。
- 請求項1記載の難燃性樹脂組成物より得られた不織布。
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP15827086.8A EP3176212B1 (en) | 2014-07-31 | 2015-07-27 | Flame-retardant resin composition and article molded from same |
| US15/304,721 US10494506B2 (en) | 2014-07-31 | 2015-07-27 | Flame-retardant resin composition and article molded from same |
| JP2016538334A JP6619340B2 (ja) | 2014-07-31 | 2015-07-27 | 難燃性樹脂組成物およびそれからの成形品 |
| CN201580024515.8A CN106459522B (zh) | 2014-07-31 | 2015-07-27 | 阻燃性树脂组合物以及来自它的成型品 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2014156464 | 2014-07-31 | ||
| JP2014-156464 | 2014-07-31 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2016017571A1 true WO2016017571A1 (ja) | 2016-02-04 |
Family
ID=55217472
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2015/071216 WO2016017571A1 (ja) | 2014-07-31 | 2015-07-27 | 難燃性樹脂組成物およびそれからの成形品 |
Country Status (5)
| Country | Link |
|---|---|
| US (1) | US10494506B2 (ja) |
| EP (1) | EP3176212B1 (ja) |
| JP (1) | JP6619340B2 (ja) |
| CN (2) | CN106459522B (ja) |
| WO (1) | WO2016017571A1 (ja) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2019085553A (ja) * | 2017-11-06 | 2019-06-06 | 株式会社フジクラ | 難燃性樹脂組成物、これを用いた成形体、絶縁電線、ケーブル及び光ファイバケーブル |
| JP2019085552A (ja) * | 2017-11-06 | 2019-06-06 | 株式会社フジクラ | 難燃性樹脂組成物、これを用いた成形体、絶縁電線、ケーブル及び光ファイバケーブル |
| JP2020195732A (ja) * | 2019-06-05 | 2020-12-10 | 積水成型工業株式会社 | すだれ |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP6845348B2 (ja) * | 2018-09-28 | 2021-03-17 | 積水化学工業株式会社 | ポリオレフィン系樹脂発泡体シート |
Citations (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS5060481A (ja) * | 1973-09-25 | 1975-05-24 | ||
| JPS5068978A (ja) * | 1973-04-16 | 1975-06-09 | ||
| JPS50105560A (ja) * | 1973-12-03 | 1975-08-20 | ||
| JP2002003727A (ja) * | 2000-06-22 | 2002-01-09 | Teijin Chem Ltd | 難燃性樹脂組成物 |
| JP2003267984A (ja) * | 2002-03-12 | 2003-09-25 | Teijin Chem Ltd | ペンタエリスリトールジホスホネート化合物およびその製造方法 |
| JP2004010586A (ja) * | 2002-06-11 | 2004-01-15 | Teijin Chem Ltd | 環状ホスホネート化合物 |
| JP2004010587A (ja) * | 2002-06-11 | 2004-01-15 | Teijin Chem Ltd | 環状ホスホネート化合物 |
| JP2004018382A (ja) * | 2002-06-12 | 2004-01-22 | Teijin Chem Ltd | 環状ホスホネート化合物 |
| JP2004018383A (ja) * | 2002-06-12 | 2004-01-22 | Teijin Chem Ltd | 環状ホスホネート化合物 |
| JP2004018380A (ja) * | 2002-06-12 | 2004-01-22 | Teijin Chem Ltd | 環状ホスホネート化合物 |
| JP2004018381A (ja) * | 2002-06-12 | 2004-01-22 | Teijin Chem Ltd | 環状ホスホネート化合物 |
| WO2013147294A1 (ja) * | 2012-03-29 | 2013-10-03 | 帝人株式会社 | 繊維用防炎加工剤 |
Family Cites Families (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3962377A (en) | 1973-09-25 | 1976-06-08 | Ciba-Geigy Corporation | 2,3,5-Trialkyl-4-hydroxybenzylphosphonates and phosphinates |
| US4174343A (en) * | 1978-05-05 | 1979-11-13 | American Cyanamid Company | Pentaerythrityl diphosphonate-ammonium polyphosphate combinations as flame retardants for olefin polymers |
| KR100407724B1 (ko) * | 1999-12-30 | 2003-12-31 | 삼성아토피나주식회사 | 난연성 폴리프로필렌 수지조성물 |
| WO2002092690A1 (en) * | 2001-05-15 | 2002-11-21 | Teijin Chemicals, Ltd. | Flame-retardant resin composition and molded article therefrom |
| JP4753498B2 (ja) | 2001-07-17 | 2011-08-24 | 株式会社Adeka | 難燃性合成樹脂組成物 |
| JP4084143B2 (ja) * | 2002-07-03 | 2008-04-30 | 帝人化成株式会社 | ペンタエリスリトールジホスホネートの製造方法 |
| US20040083537A1 (en) * | 2002-11-06 | 2004-05-06 | Eliaichaloi Mosha | Adjustable undergarment and method of manufacture |
| JP4204342B2 (ja) | 2003-02-07 | 2009-01-07 | 株式会社Adeka | 難燃性合成樹脂組成物 |
| JP2007126538A (ja) * | 2005-11-02 | 2007-05-24 | Kaneka Corp | 難燃性ポリオレフィン系樹脂組成物 |
| JP5414168B2 (ja) | 2007-11-14 | 2014-02-12 | 株式会社Adeka | 加工性の改善された難燃剤組成物、難燃性合成樹脂組成物及びその成形品 |
| JP5424444B2 (ja) | 2008-06-06 | 2014-02-26 | 株式会社Adeka | 難燃性熱可塑性樹脂組成物 |
| WO2012057297A1 (ja) | 2010-10-27 | 2012-05-03 | 帝人化成株式会社 | 難燃性樹脂組成物およびそれからの成形品 |
| EP2860309B1 (en) * | 2012-06-11 | 2017-05-10 | Teijin Limited | Flame-retardant synthetic leather |
-
2015
- 2015-07-27 EP EP15827086.8A patent/EP3176212B1/en active Active
- 2015-07-27 CN CN201580024515.8A patent/CN106459522B/zh active Active
- 2015-07-27 WO PCT/JP2015/071216 patent/WO2016017571A1/ja active Application Filing
- 2015-07-27 JP JP2016538334A patent/JP6619340B2/ja active Active
- 2015-07-27 CN CN202010493709.5A patent/CN111732786A/zh active Pending
- 2015-07-27 US US15/304,721 patent/US10494506B2/en active Active
Patent Citations (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS5068978A (ja) * | 1973-04-16 | 1975-06-09 | ||
| JPS5060481A (ja) * | 1973-09-25 | 1975-05-24 | ||
| JPS50105560A (ja) * | 1973-12-03 | 1975-08-20 | ||
| JP2002003727A (ja) * | 2000-06-22 | 2002-01-09 | Teijin Chem Ltd | 難燃性樹脂組成物 |
| JP2003267984A (ja) * | 2002-03-12 | 2003-09-25 | Teijin Chem Ltd | ペンタエリスリトールジホスホネート化合物およびその製造方法 |
| JP2004010586A (ja) * | 2002-06-11 | 2004-01-15 | Teijin Chem Ltd | 環状ホスホネート化合物 |
| JP2004010587A (ja) * | 2002-06-11 | 2004-01-15 | Teijin Chem Ltd | 環状ホスホネート化合物 |
| JP2004018382A (ja) * | 2002-06-12 | 2004-01-22 | Teijin Chem Ltd | 環状ホスホネート化合物 |
| JP2004018383A (ja) * | 2002-06-12 | 2004-01-22 | Teijin Chem Ltd | 環状ホスホネート化合物 |
| JP2004018380A (ja) * | 2002-06-12 | 2004-01-22 | Teijin Chem Ltd | 環状ホスホネート化合物 |
| JP2004018381A (ja) * | 2002-06-12 | 2004-01-22 | Teijin Chem Ltd | 環状ホスホネート化合物 |
| WO2013147294A1 (ja) * | 2012-03-29 | 2013-10-03 | 帝人株式会社 | 繊維用防炎加工剤 |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2019085553A (ja) * | 2017-11-06 | 2019-06-06 | 株式会社フジクラ | 難燃性樹脂組成物、これを用いた成形体、絶縁電線、ケーブル及び光ファイバケーブル |
| JP2019085552A (ja) * | 2017-11-06 | 2019-06-06 | 株式会社フジクラ | 難燃性樹脂組成物、これを用いた成形体、絶縁電線、ケーブル及び光ファイバケーブル |
| JP7090485B2 (ja) | 2017-11-06 | 2022-06-24 | 株式会社フジクラ | 難燃性樹脂組成物、これを用いた成形体、絶縁電線、ケーブル及び光ファイバケーブル |
| JP7090484B2 (ja) | 2017-11-06 | 2022-06-24 | 株式会社フジクラ | 難燃性樹脂組成物、これを用いた成形体、絶縁電線、ケーブル及び光ファイバケーブル |
| JP2020195732A (ja) * | 2019-06-05 | 2020-12-10 | 積水成型工業株式会社 | すだれ |
Also Published As
| Publication number | Publication date |
|---|---|
| CN106459522A (zh) | 2017-02-22 |
| CN106459522B (zh) | 2020-11-27 |
| JP6619340B2 (ja) | 2019-12-11 |
| EP3176212A1 (en) | 2017-06-07 |
| US20170051132A1 (en) | 2017-02-23 |
| JPWO2016017571A1 (ja) | 2017-05-25 |
| EP3176212B1 (en) | 2018-08-29 |
| CN111732786A (zh) | 2020-10-02 |
| EP3176212A4 (en) | 2017-07-19 |
| US10494506B2 (en) | 2019-12-03 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CA2502289C (en) | Flame retardant compositions | |
| CA2654157C (en) | Liquid phosphite blends as stabilizers | |
| CA2501384C (en) | Flame retardant compositions | |
| CA2476294C (en) | Flame retardant compositions | |
| CA2467711C (en) | Flame retardant compositions | |
| JP6771538B2 (ja) | 難燃性ポリオレフィン物品 | |
| CA2467119C (en) | Novel flame retarding compounds | |
| JP6619340B2 (ja) | 難燃性樹脂組成物およびそれからの成形品 | |
| BR112015014031B1 (pt) | Composição de retardo de chamas e composição de resina sintética de retardo de chamas | |
| TWI761486B (zh) | 難燃劑組合物及含有其之難燃性合成樹脂組合物 | |
| EP1423465A1 (en) | Flame retardant compositions | |
| TWI782135B (zh) | 難燃劑組合物及包含該難燃劑組合物之難燃性熱塑性樹脂組合物 | |
| TW201908471A (zh) | 組合物及難燃性樹脂組合物 | |
| KR20200079255A (ko) | 조성물 및 난연성 수지 조성물 | |
| Ba et al. | Synthesis and characterization of a novel charring agent and its application in intumescent flame retardant polyethylene system | |
| JPWO2018042869A1 (ja) | 難燃剤組成物及び難燃性合成樹脂組成物 | |
| TW202043444A (zh) | 難燃劑組成物及難燃性合成樹脂組成物 | |
| JPWO2018020840A1 (ja) | 難燃剤組成物及び難燃性合成樹脂組成物 | |
| JP2021138664A (ja) | 有機リン化合物、熱可塑性樹脂組成物及び電子機器 | |
| WO2012011519A1 (ja) | 環状アミン塩を含む難燃剤及び難燃性樹脂組成物 | |
| US20230295376A1 (en) | A halogen-free synergist | |
| JPWO2019117049A1 (ja) | 組成物及び難燃性樹脂組成物 | |
| WO2024094455A1 (en) | Flame-retardant composition, polymer composition comprising same and use thereof | |
| TW202309254A (zh) | 難燃劑組成物,難燃性樹脂組成物及其成形品 | |
| JP3899569B2 (ja) | 難燃性ポリオレフィン系樹脂組成物 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 15827086 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 15304721 Country of ref document: US |
|
| REEP | Request for entry into the european phase |
Ref document number: 2015827086 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2015827086 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 2016538334 Country of ref document: JP Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |