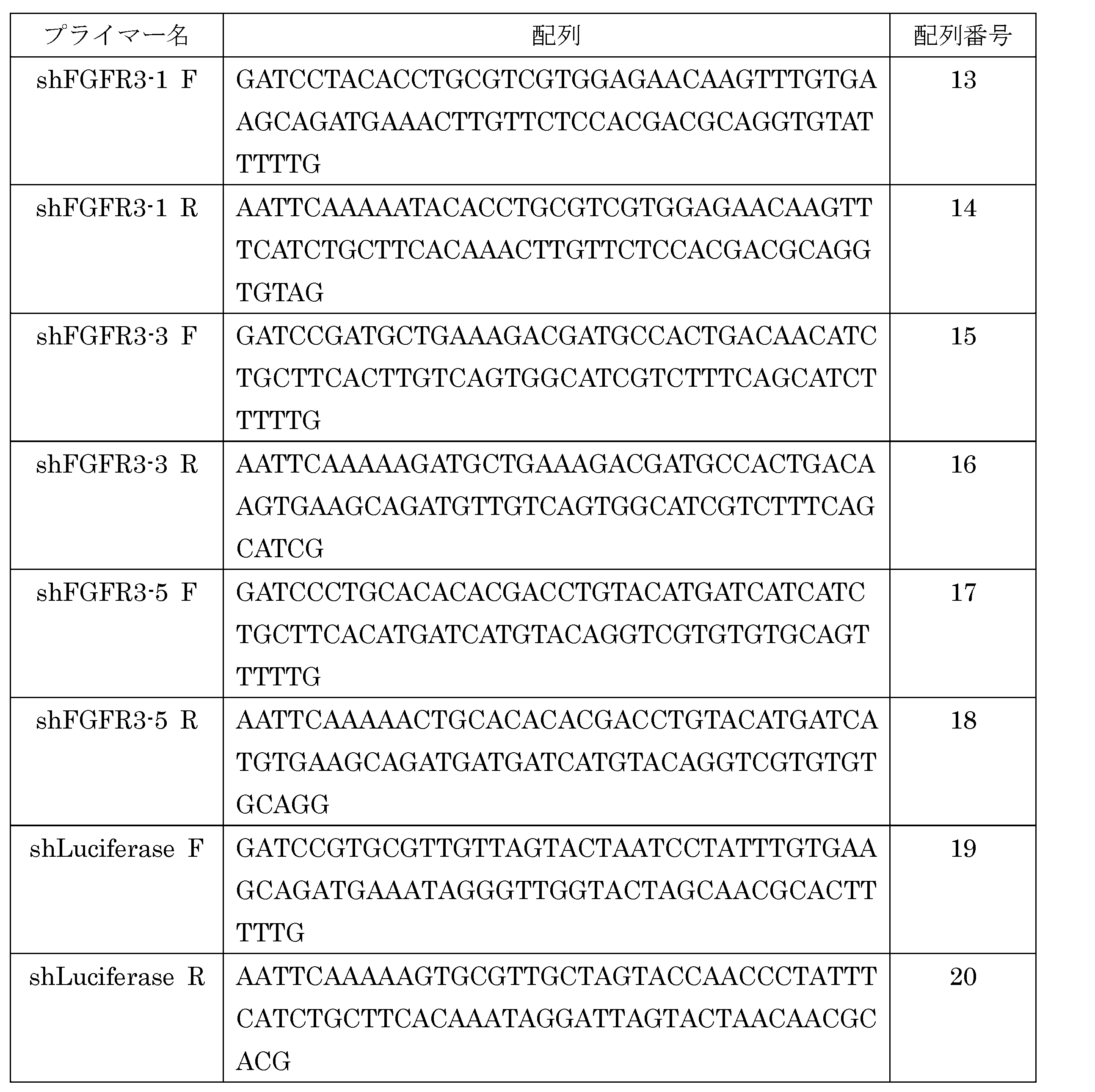
WO2015083582A1 - Fgfr3病の予防および治療剤ならびにそのスクリーニング方法 - Google Patents
Fgfr3病の予防および治療剤ならびにそのスクリーニング方法 Download PDFInfo
- Publication number
- WO2015083582A1 WO2015083582A1 PCT/JP2014/081093 JP2014081093W WO2015083582A1 WO 2015083582 A1 WO2015083582 A1 WO 2015083582A1 JP 2014081093 W JP2014081093 W JP 2014081093W WO 2015083582 A1 WO2015083582 A1 WO 2015083582A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- fgfr3
- cells
- ipsc
- culture
- medium
- Prior art date
Links
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/335—Heterocyclic compounds having oxygen as the only ring hetero atom, e.g. fungichromin
- A61K31/365—Lactones
- A61K31/366—Lactones having six-membered rings, e.g. delta-lactones
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/50—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing
- G01N33/5005—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving human or animal cells
- G01N33/5008—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving human or animal cells for testing or evaluating the effect of chemical or biological compounds, e.g. drugs, cosmetics
- G01N33/5044—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving human or animal cells for testing or evaluating the effect of chemical or biological compounds, e.g. drugs, cosmetics involving specific cell types
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/21—Esters, e.g. nitroglycerine, selenocyanates
- A61K31/215—Esters, e.g. nitroglycerine, selenocyanates of carboxylic acids
- A61K31/22—Esters, e.g. nitroglycerine, selenocyanates of carboxylic acids of acyclic acids, e.g. pravastatin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/40—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with one nitrogen as the only ring hetero atom, e.g. sulpiride, succinimide, tolmetin, buflomedil
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/40—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with one nitrogen as the only ring hetero atom, e.g. sulpiride, succinimide, tolmetin, buflomedil
- A61K31/403—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with one nitrogen as the only ring hetero atom, e.g. sulpiride, succinimide, tolmetin, buflomedil condensed with carbocyclic rings, e.g. carbazole
- A61K31/404—Indoles, e.g. pindolol
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/505—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/08—Drugs for skeletal disorders for bone diseases, e.g. rachitism, Paget's disease
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/06—Animal cells or tissues; Human cells or tissues
- C12N5/0602—Vertebrate cells
- C12N5/0652—Cells of skeletal and connective tissues; Mesenchyme
- C12N5/0655—Chondrocytes; Cartilage
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/06—Animal cells or tissues; Human cells or tissues
- C12N5/0602—Vertebrate cells
- C12N5/0696—Artificially induced pluripotent stem cells, e.g. iPS
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Q—MEASURING OR TESTING PROCESSES INVOLVING ENZYMES, NUCLEIC ACIDS OR MICROORGANISMS; COMPOSITIONS OR TEST PAPERS THEREFOR; PROCESSES OF PREPARING SUCH COMPOSITIONS; CONDITION-RESPONSIVE CONTROL IN MICROBIOLOGICAL OR ENZYMOLOGICAL PROCESSES
- C12Q1/00—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions
- C12Q1/68—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions involving nucleic acids
- C12Q1/6876—Nucleic acid products used in the analysis of nucleic acids, e.g. primers or probes
- C12Q1/6883—Nucleic acid products used in the analysis of nucleic acids, e.g. primers or probes for diseases caused by alterations of genetic material
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/50—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing
- G01N33/5005—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving human or animal cells
- G01N33/5008—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving human or animal cells for testing or evaluating the effect of chemical or biological compounds, e.g. drugs, cosmetics
- G01N33/5044—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving human or animal cells for testing or evaluating the effect of chemical or biological compounds, e.g. drugs, cosmetics involving specific cell types
- G01N33/5073—Stem cells
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/10—Growth factors
- C12N2501/115—Basic fibroblast growth factor (bFGF, FGF-2)
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/10—Growth factors
- C12N2501/119—Other fibroblast growth factors, e.g. FGF-4, FGF-8, FGF-10
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/10—Growth factors
- C12N2501/15—Transforming growth factor beta (TGF-β)
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/10—Growth factors
- C12N2501/155—Bone morphogenic proteins [BMP]; Osteogenins; Osteogenic factor; Bone inducing factor
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/10—Growth factors
- C12N2501/19—Growth and differentiation factors [GDF]
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2506/00—Differentiation of animal cells from one lineage to another; Differentiation of pluripotent cells
- C12N2506/45—Differentiation of animal cells from one lineage to another; Differentiation of pluripotent cells from artificially induced pluripotent stem cells
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2510/00—Genetically modified cells
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Q—MEASURING OR TESTING PROCESSES INVOLVING ENZYMES, NUCLEIC ACIDS OR MICROORGANISMS; COMPOSITIONS OR TEST PAPERS THEREFOR; PROCESSES OF PREPARING SUCH COMPOSITIONS; CONDITION-RESPONSIVE CONTROL IN MICROBIOLOGICAL OR ENZYMOLOGICAL PROCESSES
- C12Q2600/00—Oligonucleotides characterized by their use
- C12Q2600/136—Screening for pharmacological compounds
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Q—MEASURING OR TESTING PROCESSES INVOLVING ENZYMES, NUCLEIC ACIDS OR MICROORGANISMS; COMPOSITIONS OR TEST PAPERS THEREFOR; PROCESSES OF PREPARING SUCH COMPOSITIONS; CONDITION-RESPONSIVE CONTROL IN MICROBIOLOGICAL OR ENZYMOLOGICAL PROCESSES
- C12Q2600/00—Oligonucleotides characterized by their use
- C12Q2600/158—Expression markers
Definitions
- the present invention relates to a method for screening a therapeutic and / or prophylactic agent for fibroblast growth factor receptor 3 (FGFR3) disease.
- the present invention also relates to a therapeutic agent for FGFR3 disease.
- FGFR3 fibroblast growth factor receptor 3
- Non-Patent Document 1 Jonquoy et al. By tyrosine kinase inhibitors (Non-Patent Document 1), Rauchenberger et al. By FGFR3 neutralizing antibody (Non-Patent Document 2), and Yasoda et al. By c-type natriuretic peptide (CNP) (Non-patent Document 1).
- Patent Document 3 reports a method of inhibiting excessive signal transduction from FGFR3. Some of these methods actually restore bone growth in model mice with cartilage dysplasia associated with FGFR3.
- iPS cells mouse and human induced pluripotent stem cells
- Yamanaka et al. Succeeded in establishing iPS cells by introducing 4 genes of Oct3 / 4, Sox2, Klf4 and c-Myc into human skin-derived fibroblasts (Patent Document 1 and Non-Patent Document 4).
- iPS cells obtained in this way can be differentiated into cells of each tissue after being prepared using cells from the patient to be treated, it is possible to reproduce the pathology in vitro. It is thought that there is.
- FGFR3 fibroblast growth factor receptor 3
- iPS cells derived from somatic cells of patients with FGFR3 disease found a tendency to suppress differentiation into chondrocytes as compared with iPS cells derived from normal individuals.
- the drugs used for the treatment of hyperlipidemia are converted to chondrocytes. It was found to promote differentiation induction.
- the present invention has been completed based on such findings.
- a pharmaceutical for treatment and / or prevention of FGFR3 disease comprising an HMG-CoA reductase inhibitor as an active ingredient.
- the HMG-CoA reductase inhibitor is a drug selected from the group consisting of mevastatin, atorvastatin, pravastatin, rosuvastatin, fluvastatin and lovastatin.
- the FGFR3 disease is lethal osteodysplasia (TD) and / or achondroplasia (ACH).
- [4] A method for treating and / or preventing FGFR3 disease, comprising administering an HMG-CoA reductase inhibitor.
- the HMG-CoA reductase inhibitor is a drug selected from the group consisting of mevastatin, atorvastatin, pravastatin, rosuvastatin, fluvastatin and lovastatin.
- the FGFR3 disease is lethal osteodysplasia (TD) and / or achondroplasia (ACH).
- HMG-CoA reductase inhibitor in the manufacture of a medicament for the treatment and / or prevention of FGFR3 disease.
- HMG-CoA reductase inhibitor is a drug selected from the group consisting of mevastatin, atorvastatin, pravastatin, rosuvastatin, fluvastatin and lovastatin.
- FGFR3 disease is lethal osteodysplasia (TD) and / or achondroplasia (ACH).
- a method for screening for a therapeutic and / or prophylactic agent for FGFR3 disease comprising the following steps: (A) differentiating an induced pluripotent stem (iPS) cell having a mutation in FGFR3 into a chondrocyte under the condition of contacting with or not contacting the test substance; (B) measuring one or more indicators selected from the group consisting of the amount of chondrocyte extracellular matrix, chondrocyte marker gene and fibroblast marker gene expression in the culture obtained in step (a) And (c) when the amount of the chondrocyte extracellular matrix or the chondrocyte marker gene is increased or the expression level of the fibroblast marker gene is decreased under the condition of contacting with the test substance under the condition of not contacting, A step of selecting a test substance as a therapeutic or preventive for FGFR3 disease.
- iPS induced pluripotent stem
- the method according to [10], wherein the differentiation into chondrocytes in the step (a) includes the following steps: (I) a step of inducing mesoderm cells by adhesion culture of pluripotent stem cells, (Ii) an adhesion culture of the cells obtained in the step (i) in a culture medium containing bFGF, ascorbic acid, BMP2, TGF ⁇ , GDF5 and a test substance; and (iii) obtained in the step (ii). A suspension culture of the cultured cells in a culture medium containing ascorbic acid, BMP2, TGF ⁇ , GDF5 and a test substance.
- chondrocyte marker gene is at least one gene selected from the group consisting of SOX9, AGGRECAN and COL2.
- the fibroblast marker gene is COL1A1 and / or COL1A2.
- the mutation of FGFR3 is an Arg248Cys or Gly380Arg mutation in FGFR3.
- the FGFR3 disease is lethal osteodysplasia (TD) and / or achondroplasia (ACH).
- HMG-CoA reductase inhibitor used for treating and / or preventing FGFR3 disease.
- HMG-CoA reductase inhibitor used for treating and / or preventing FGFR3 disease.
- TD lethal osteodysplasia
- ACH achondroplasia
- the present invention enables screening for therapeutic and / or prophylactic agents for FGFR3 disease using a novel tool.
- Fig. 1 shows the results of confirming the characteristics of TD-iPSC IV.
- the left diagram shows the phase image of TD-iPSC (TD714-3), and the right diagram shows the fluorescence image of TD-iPSC (TD714-3) for SSEA4 and TRA1-60.
- the scale bar is 50 ⁇ m.
- (b) shows the histological image of teratoma formed after transplantation of TD-iPSC (TD10749-2) into SCID mice.
- the scale bar is 50 ⁇ m.
- FIG. 2 shows TD-iPSC and WT-iPSC differentiated chondrogenically.
- FIG. 3 shows the analysis results of chondrocytes induced to differentiate from TD-iPSC and WT-iPSC.
- (a) The results of real-time RT-PCR expression analysis of chondrocyte marker genes (SOX9, COL2 and AGGRECAN) and fibroblast marker genes (COL1A1 and COL1A2) on the 28th day of induction of chondrocyte differentiation from each iPSC Show.
- (b) shows an immunohistological image on day 42 after induction of differentiation from iPSC into chondrocytes. The scale bar is 50 ⁇ m.
- (c) shows immunostained images of type II collagen in cartilage particles derived from TD-iPSC (TD-714-3) and WT-iPSC (409B2).
- FIG. 4 shows the analysis results of chondrocytes induced to differentiate from TD-iPSC and WT-iPSC.
- (a) shows the results of real-time RT-PCR expression analysis of FGFR3 mRNA in the process of chondrogenic differentiation of TD-iPSC and WT-iPSC.
- the chondrocytes as a positive control show the results of human fetal chondrocytes (402RD-R10f) purchased from Cell Application, Inc., and show the results of fibroblasts as a negative control.
- (b) shows the results of quantitative PCR measuring the expression level of FGFR3 mRNA on day 28 of cartilage differentiation derived from TD-iPSC and WT-iPSC.
- FIG. 5 shows the analysis results of chondrocytes induced to differentiate from TD-iPSC and WT-iPSC.
- FIG. 6 shows the analysis results of chondrocyte differentiation induction from TD-iPSC (TD714-3) introduced with FGFR3 shRNA.
- FIG. 7 shows the result of inducing chondrocytes of TD-iPSC (TD714-3) by adding an FGFR3 neutralizing antibody.
- (a) shows the histology of particles on day 42 when FGFR3 neutralizing antibody was added to the medium during the induction of chondrocyte differentiation of TD-iPSC. IgG was added instead of the FGFR3 neutralizing antibody and used as a negative control. The scale bar is 50 ⁇ m.
- (b) Counting the number of particles substantially stained with safranin O for any three particles derived from TD-iPSC supplemented with FGFR3 neutralizing antibody (Anti- FGFR3) and negative control (IgG) Results are shown.
- FIG. 8 shows the results of inducing chondrocytes of TD-iPSC (TD714-3) in the presence of the test substance.
- ⁇ TD-iPSC chondrocyte differentiation inducing step shows the particle histology on day 42 when the test substance was added to the medium and cultured.
- the scale bar is 50 ⁇ m.
- (b) shows the results of counting the number of particles substantially stained with safranin O for any 10 particles derived from TD-iPSC to which each test substance was added.
- FIG. 9 shows the results of chondrocyte induction of TD-iPSC (TD714-3) in the presence of lovastatin.
- A shows the histology of particles on day 42 when lovastatin was added to the medium in the chondrocyte differentiation induction step of TD-iPSC. The scale bar is 50 ⁇ m.
- FIG. 11 shows the results of chondrocyte induction of ACH-iPSC in the presence of lovastatin or CNP.
- FIG. 12 shows the results of bone growth analysis when rosuvastatin was administered to ACH model mice (FGFR3 Ach ).
- FIG. 13 shows the analysis results of the size of the rods when rosuvastatin was administered to ACH model mice (FGFR3 Ach ). The X-ray images of the maximum weight (Highest) and the minimum weight (Lowest) in each group are shown, and the body weight is described below the figure. The scale bar is 10 mm.
- FIG. 14 shows the analysis results of bone growth when lovastatin is administered to ACH model mice.
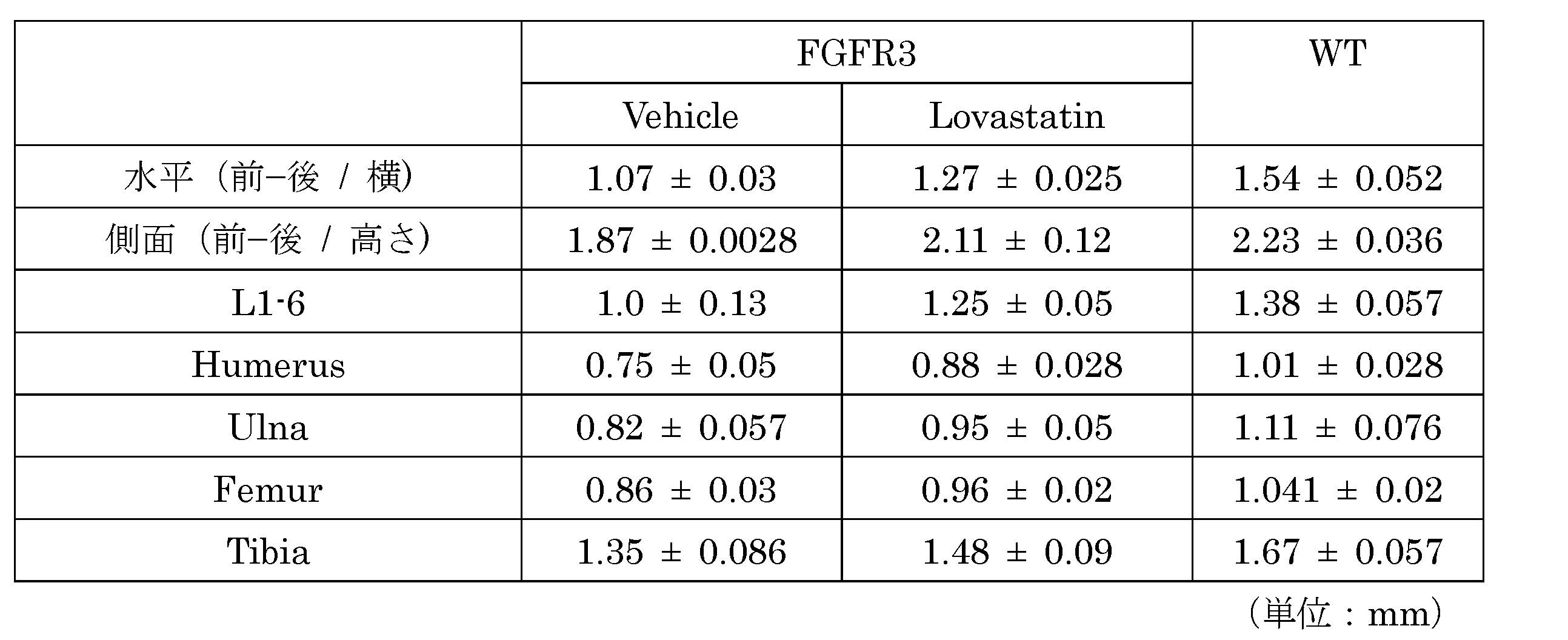
- FIG. 15 shows the measurement results of the length of each bone site in FGFR3 Ach mice treated with robustin.
- FIG. 16 shows the results of culturing chondrocytes with the addition of lovastatin.
- (b) shows the results of measuring the length of primitive cartilage on day 1 and day 7 of organ culture.
- (c) shows stained images with BrdU antibody and safranin O after treatment with BrdU on day 7 of organ culture of metatarsal primitive cartilage.
- (d) shows the results of analyzing the content of BrdU positive cells.
- (e) Safranin O-stained image on day 14 of pellet culture.
- (f) The measurement results of quantitative PCR on pellet culture day 14 (Sox9, Col2a1 and Acan) and day 28 (Runx2 and Col10a1) are shown.
- (g) shows the results of immunoblotting using FGFR3 antibody after culturing primary chondrocytes of wild type and FGFR3 Ach mice with MG132 or Bafilomycun A1 (Baf A1) in the presence of lovastatin.
- FGFR3 disease means any bone morphogenetic disease in which abnormal bone formation is caused by having a mutation in FGFR3.
- FGFR3 disease preferably means a series of diseases belonging to the FGFR3 disease group described in the international classification of bone system diseases (Warman et al., Am J Med Genet 155A (5): 943-68 (2011)).
- TD lethal osteodysplasia
- ACH achondroplasia
- hypochondral dysplasia Camptodactyly, tallature stature, and hearing loss syndrome
- CASHL Crouzon-like craniosynostosis with acanthosis nigricans
- Examples include early skull fusion.
- the mutation occurring in FGFR3 may be either a gain-of-function type mutation or a function-deficient type mutation, but may preferably be a gain-of-function type mutation.
- an iPS cell introduces a specific nuclear reprogramming substance into a somatic cell in the form of DNA or protein, or an endogenous mRNA and protein of the nuclear reprogramming substance by a drug.
- It is an artificial stem cell derived from a somatic cell that has almost the same characteristics as ES cells, such as differentiation pluripotency and proliferation ability by self-replication, which can be produced by increasing the expression of K. Takahashi and S. Yamanaka (2006) Cell, 126: 663-676, K. Takahashi et al. (2007) Cell, 131: 861-872, J. Yu et al. (2007) Science, 318: 1917-1920, M. Nakagawa et al.
- the nuclear reprogramming substance is not particularly limited as long as it is a gene specifically expressed in ES cells, a gene that plays an important role in maintaining undifferentiation of ES cells, or a gene product thereof.
- nucleotide sequences of mouse and human cDNAs of each of the above nuclear reprogramming substances and amino acid sequence information of proteins encoded by the cDNAs refer to NCBI accession numbers described in WO 2007/069666, and L-Myc, Lin28 , Lin28b, Esrrb, Esrrg and Glis1 mouse and human cDNA sequences and amino acid sequence information can be obtained by referring to the following NCBI accession numbers, respectively.
- a person skilled in the art can prepare a desired nuclear reprogramming substance by a conventional method based on the cDNA sequence or amino acid sequence information.
- nuclear reprogramming substances may be introduced into somatic cells in the form of proteins, for example, by lipofection, binding to cell membrane permeable peptides, microinjection, or in the form of DNA, for example, It can be introduced into somatic cells by techniques such as viruses, plasmids, artificial chromosomes, vectors, lipofection, liposomes, and microinjection.
- Virus vectors include retrovirus vectors, lentivirus vectors (cell, 126, pp.663-676, 2006; Cell, 131, pp.861-872, 2007; Science, 318, pp.1917-1920, 2007 ), Adenovirus vectors (Science, 322, 945-949, 2008), adeno-associated virus vectors, Sendai virus vectors (Proc Jpn Acad Ser B Phys Biol Sci. 85, 348-62, 2009) and the like.
- artificial chromosome vectors include human artificial chromosomes (HAC), yeast artificial chromosomes (YAC), and bacterial artificial chromosomes (BAC, PAC).
- a plasmid for mammalian cells can be used (Science, 322: 949-953, 2008).
- the vector can contain regulatory sequences such as a promoter, an enhancer, a ribosome binding sequence, a terminator, and a polyadenylation site so that a nuclear reprogramming substance can be expressed.
- promoter used examples include EF1 ⁇ promoter, CAG promoter, SR ⁇ promoter, SV40 promoter, LTR promoter, CMV (cytomegalovirus) promoter, RSV (rous sarcoma virus) promoter, MoMuLV (Moloney murine leukemia virus) LTR, HSV- A TK (herpes simplex virus thymidine kinase) promoter or the like is used.
- EF1 ⁇ promoter, CAG promoter, MoMuLV LTR, CMV promoter, SR ⁇ promoter and the like can be mentioned.
- drug resistance genes for example, kanamycin resistance gene, ampicillin resistance gene, puromycin resistance gene, etc.
- thymidine kinase gene diphtheria toxin gene and other selectable marker sequences
- green fluorescent protein (GFP) green fluorescent protein
- GUS ⁇ -glucuronidase
- reporter gene sequences such as FLAG, and the like.
- the above vector contains a LoxP sequence before and after the gene or promoter encoding the nuclear reprogramming substance and the gene encoding the nuclear reprogramming substance that binds to it. You may have.
- transposons include, for example, piggyBac, a transposon derived from lepidopterous insects (Kaji, K. et al., (2009), Nature, 458: 771-775, Woltjen et al., (2009), Nature, 458: 766-770, WO 2010/012077).
- the vector replicates without chromosomal integration and is episomal, so that the origin and replication of lymphotropic herpesvirus (lymphotrophic herpes virus), BK virus and bovine papillomavirus
- sequence which concerns on may be included. Examples include EBNA-1 and oriP or LargeLT and SV40ori sequences (WO 2009/115295, WO 2009/157201 and WO 2009/149233).
- an expression vector for polycistronic expression may be used.
- the gene coding sequence may be linked by an IRES or foot-and-mouth disease virus (FMDV) 2A coding region (Science, 322: 949-953, 2008 and WO 2009/092042 2009/152529).
- HDAC histone deacetylase
- VPA valproate PA
- MC 1293 MC 1293, M344
- siRNA and shRNA against HDAC e.g.
- HDAC1 siRNA Smartpool® ⁇ (Millipore), HuSH 29mer shRNA Constructs nucleic acid expression inhibitors such as HDAC1 (OriGene) etc.], DNA methyltransferase inhibitors (eg 5'-azacytidine) (Nat. Biotechnol., 26 (7): 795-797 (2008)), G9a histone Methyltransferase inhibitors [for example, small molecule inhibitors such as BIX-01294 (Cell Stem Cell, 2: 525-528 (2008)), siRNA and shRNA against G9a (eg G9a siRNA (human) (Santa Cruz Biotechnology) etc.), etc.
- BIX-01294 Cell Stem Cell, 2: 525-528 (2008)
- siRNA and shRNA against G9a eg G9a siRNA (human) (Santa Cruz Biotechnology) etc.
- Nucleic acid expression inhibitors, etc.] L-channel calcium agonist ( For example, Bayk8644) (Cell Stem Cell, 3, 568-574 (2008)), p53 inhibitors (eg siRNA and shRNA against p53) (Cell Stem Cell, 3, 475-479 (2008)), Wnt Signaling activator (eg soluble Wnt3a) (Cell Stem Cell, 3, 132-135 (2008)), growth factors such as LIF or bFGF, ALK5 inhibitors (eg SB431542) (Nat.
- a method of increasing the expression of the endogenous protein of the nuclear reprogramming substance by a drug may be used.
- a drug include 6-bromoindirubin-3'-oxime, indirubin-5-nitro-3'-oxime, valproic acid, 2- (3- (6-methylpyridin-2-yl) -lH-pyrazol-4-yl) -1,5-naphthyridine, 1- (4-methylphenyl) -2- (4,5,6, Examples include 7-tetrahydro-2-imino-3 (2H) -benzothiazolyl) ethanoneBrHBr (pifithrin-alpha), prostaglandin J2, prostaglandin2E2, and the like (WO 2010/068955).
- Examples of the culture medium for iPS cell induction include (1) DMEM, DMEM / F12 or DME medium containing 10-15% FBS (these media include LIF, penicillin / streptomycin, puromycin, L-glutamine). , (2) ES cell culture medium containing bFGF or SCF, such as mouse ES cell culture medium (for example, TX-WES medium, thrombos. X) or primate ES cell culture medium (for example, primate (human & monkey) ES cell culture medium (Reprocell, Kyoto, Japan), mTeSR-1).
- mouse ES cell culture medium for example, TX-WES medium, thrombos. X
- primate ES cell culture medium for example, primate (human & monkey) ES cell culture medium (Reprocell, Kyoto, Japan), mTeSR-1).
- a somatic cell and a nuclear reprogramming substance are brought into contact with each other in DMEM or DMEM / F12 medium containing 10% FBS in the presence of 5% CO 2 at 37 ° C.
- Culture for ⁇ 7 days then re-spread the cells onto feeder cells (eg mitomycin C-treated STO cells, SNL cells, etc.), and culture of bFGF-containing primate ES cells approximately 10 days after contact between the somatic cells and the nuclear reprogramming substance
- the cells can be cultured in a working medium to generate ES cell-like colonies about 30 to about 45 days or more after the contact.
- the cells may be cultured under conditions of an oxygen concentration as low as 5-10%.
- an extracellular matrix may be used instead of feeder cells, and as the extracellular matrix, collagen, gelatin, laminin (for example, laminin 111, 411 or 511, or a fragment thereof), heparan sulfate proteoglycan, or entactin, and These fragments, or combinations thereof are exemplified.
- FBS-containing DMEM medium for example, LIF, penicillin / streptomycin, puromycin, L-glutamine, non-glutinin
- feeder cells eg, mitomycin C-treated STO cells, SNL cells.
- Essential amino acids, ⁇ -mercaptoethanol, etc. can be included as appropriate.
- ES-like colonies can be formed after about 25 to about 30 days or more.
- the medium is replaced with a fresh medium once a day from the second day after the start of the culture.
- the number of somatic cells used for nuclear reprogramming is not limited, but ranges from about 5 ⁇ 10 3 to about 5 ⁇ 10 6 cells per 100 cm 2 of culture dish.
- a marker gene-expressing cell When a gene containing a drug resistance gene is used as a marker gene, a marker gene-expressing cell can be selected by culturing in a medium (selective medium) containing the corresponding drug.
- the marker gene is a fluorescent protein gene
- the marker gene-expressing cells can be obtained by observing with a fluorescence microscope, by adding a luminescent substrate in the case of a luminescent enzyme gene, and by adding a chromogenic substrate in the case of a chromogenic enzyme gene. Can be detected.
- a “somatic cell” may be any cell other than a germ cell derived from a mammal (eg, human, mouse, monkey, pig, rat, etc.), for example, keratinized epithelial cell (Eg, keratinized epidermal cells), mucosal epithelial cells (eg, epithelial cells of the tongue surface), exocrine glandular epithelial cells (eg, mammary cells), hormone-secreting cells (eg, adrenal medullary cells), cells for metabolism and storage (Eg, hepatocytes), luminal epithelial cells that make up the interface (eg, type I alveolar cells), luminal epithelial cells (eg, vascular endothelial cells) in the inner chain, and ciliated cells that are capable of transporting (Eg, airway epithelial cells), extracellular matrix secreting cells (eg, fibroblasts), contractile cells (eg, smooth muscle cells), blood and immune system cells (e
- undifferentiated progenitor cells including somatic stem cells
- terminally differentiated mature cells can be used as the source of somatic cells in the invention.
- undifferentiated progenitor cells include tissue stem cells (somatic stem cells) such as neural stem cells, hematopoietic stem cells, mesenchymal stem cells, and dental pulp stem cells.
- tissue stem cells such as neural stem cells, hematopoietic stem cells, mesenchymal stem cells, and dental pulp stem cells.
- blood cells or fibroblasts particularly human skin fibroblasts (HDF)
- HDF human skin fibroblasts
- the mammal individual from which somatic cells are collected is not particularly limited, but is preferably a human.
- iPS cells prepared from somatic cells derived from a subject suffering from FGFR3 disease may have a mutation unique to FGFR3 disease patients in the FGFR3 gene. Mutations peculiar to FGFR3 disease patients include Arg246Cys, Arg248Cys, Ser249Cys, Gly370Cys, Ser371Cys, Thr373Cys, Lys650Glu, X807Arg, X807Cys, etc. However, it is not limited to these.
- a method for inducing differentiation of chondrocytes from differentiated inducing method iPS cells into chondrocytes can be adopted any method used in the art, not only the method known to those skilled in the art at the time of the Applicant It is also possible to adopt the differentiation induction method developed after filing this application.
- As a method for inducing differentiation of chondrocytes for example, Koyama, N. et al. Stem cells and development 22, 102-113 (2013), Hwang, NS, et al. PLoS ONE 3, e2498 (2008), Oldershaw, RA et al. Nat. Biotechnol. 28, 1187-1194 (2010), Bai, HY, et al. Journal of biomedical materials research. Part A 94, 539-546 (2010), Yamashita, A. et al. Scientific Reports 3 (2013) and the like, but are not limited thereto.
- chondrocyte means a cell that produces an extracellular matrix that forms cartilage such as collagen, or a precursor cell that becomes such a cell.
- Such chondrocytes may be cells that express a chondrocyte marker, and examples of chondrocyte markers include type II collagen (COL2A1), SOX9, and AGGRECAN.
- COL2A1 includes, as a NCBI accession number, a gene having a nucleotide sequence described in NM_001844 or NM_033150 for humans, NM_001113515 or NM_031163 for humans, and a protein encoded by the gene, and these Naturally occurring variants having the following functions are included.
- SOX9 has a gene having a nucleotide sequence described as NCBI accession number NM_000346 for humans and NM_011448 for mice, a protein encoded by the gene, and these functions. Naturally occurring variants are included.
- AGGRECAN includes NCBI accession numbers as follows: in the case of humans, NM_001135 or NM_013227, in the case of mice, the gene having the nucleotide sequence described in NM_007424, the protein encoded by the gene, and their functions Naturally occurring variants having are included.
- the cartilage extracellular matrix in the present invention is an extracellular matrix that can be stained with safranin O and its analogs.
- the chondrocyte may be in the state of a culture (particle) composed of an extracellular matrix produced from the chondrocyte (cartilage-like tissue).
- iPS cells are cultured in a basal medium supplemented with Wnt3A, Activin and FGF2.
- the cells may be cultured while adhering to the culture vessel, or may be cultured while floating in the culture solution.
- a culture vessel coated with matrigel (BD) type I collagen, type IV collagen, gelatin, laminin, heparan sulfate proteoglycan, or entactin, and combinations thereof can be used.
- basal medium examples include IMDM medium, Medium 199 medium, Eagle's'MinimummEssential Medium (EMEM) medium, ⁇ MEM medium, Dulbecco's modified Eagle's Medium (DMEM) medium, Ham's F12 medium, RPMI 1640 medium, and Fischer's medium.
- the medium may contain serum (for example, FBS) or may be serum-free.
- albumin, transferrin, KnockOut Serum Replacement (KSR) serum substitute for FBS during ES cell culture
- N2 supplement Invitrogen
- B27 supplement Invitrogen
- fatty acids insulin
- It may contain one or more serum substitutes such as sodium selenate, collagen precursor, trace elements, 2-mercaptoethanol, 3'-thiolglycerol, lipids, amino acids, L-glutamine, GlutaMAX (Invitrogen), non It may also contain one or more substances such as essential amino acids (NEAA), vitamins, growth factors, low molecular weight compounds, antibiotics, antioxidants, pyruvate, buffers, inorganic salts and the like.
- the basal medium is DMEM / F12 containing insulin, transferrin, sodium selenite, and 1% serum.
- the concentration of Wnt in the basal medium is, for example, in the range of 1-200 ng / ml, preferably in the range of 10-50 ng / ml, for example 1 ng / ml, 10 ng / ml, 20 ng / ml, 25 ng / ml ml, 30ng / ml, 40ng / ml, 50ng / ml, 60ng / ml, 70ng / ml, 80ng / ml, 90ng / ml, 100ng / ml, 110ng / ml, 120ng / ml, 130ng / ml, 140ng / ml, Examples include, but are not limited to, 150 ng / ml, 160 ng / ml, 170 ng / ml, 180 ng / ml, 190 ng / ml, and 200 ng / ml.
- it is
- the concentration of Activin in the basal medium is, for example, in the range of 1-200 ng / ml, preferably in the range of 10-50 ng / ml, for example 1 ng / ml, 10 ng / ml, 20 ng / ml, 25 ng / ml 30ng / ml, 40ng / ml, 50ng / ml, 60ng / ml, 70ng / ml, 80ng / ml, 90ng / ml, 100ng / ml, 110ng / ml, 120ng / ml, 130ng / ml, 140ng / ml, 150ng / ml, 160 ng / ml, 170 ng / ml, 180 ng / ml, 190 ng / ml, 200 ng / ml, but not limited thereto.
- the concentration of Activin in the medium can be changed during the culture period. For example, after culturing at 50 ng / ml for 1 day, culturing for 1 day after changing to 25 ng / ml, and further culturing for 1 day after changing to 10 ng / ml To do.
- the concentration of FGF2 in the basal medium is, for example, in the range of 1-100 ng / ml, preferably in the range of 20-40 ng / ml, for example, 1 ng / ml, 10 ng / ml, 15 ng / ml, 20 ng / ml, 25 ng / ml, 30 ng / ml, 40 ng / ml, 50 ng / ml, 60 ng / ml, 70 ng / ml, 80 ng / ml, 90 ng / ml, 100 ng / m, but not limited thereto.
- it is 20 ng / ml.
- the concentration of BMP4 in the basal medium is, for example, in the range of 1-100 ng / ml, preferably in the range of 20-40 ng / ml, for example, 1 ng / ml, 10 ng / ml, 20 ng / ml, 25 ng / ml, 30 ng / ml, 35ng / ml, 40ng / ml, 50ng / ml, 60ng / ml, 70ng / ml, 80ng / ml, 90ng / ml, 100ng / ml, but not limited to these.
- it is 40 ng / ml.
- the concentration of BMP4 in the medium can be changed during the culture period. For example, after culturing at 40 ng / ml for 6 days, culturing for 2 days instead of 25 ng / ml can be mentioned.
- the concentration of Follistatin in the basal medium is, for example, in the range of 1-200 ng / ml, preferably in the range of 50-150 ng / ml, for example 1 ng / ml, 10 ng / ml, 20 ng / ml, 30 ng / ml.
- ng / ml 40ng / ml, 50ng / ml, 60ng / ml, 70ng / ml, 80ng / ml, 90ng / ml, 100ng / ml, 110ng / ml, 120ng / ml, 130ng / ml, 140ng / ml, 150ng / ml, 160 ng / ml, 170 ng / ml, 180 ng / ml, 190 ng / ml, and 200 ng / ml are not limited to these. Preferably, it is 100 ng / ml.
- the concentration of GDF5 in the basal medium is, for example, in the range of 1-100 ng / ml, preferably in the range of 20-40 ng / ml, for example, 1 ng / ml, 10 ng / ml, 20 ng / ml, 25 ng / ml, 30 ng / ml, 35ng / ml, 40ng / ml, 50ng / ml, 60ng / ml, 70ng / ml, 80ng / ml, 90ng / ml, 100ng / m, but not limited thereto.
- it is 40 ng / ml.
- the concentration of GDF5 in the medium can be changed during the culture period. For example, after culturing at 20 ng / ml for 2 days, culturing for 3 days instead of 40 ng / ml can be mentioned.
- the concentration of NT4 in the basal medium is, for example, in the range of 1-10 ng / ml, preferably in the range of 1-5 ng / ml, for example, 1 ng / ml, 1.5 ng / ml, 2 ng / ml, 2.5 ng / ml 3 ng / ml, 4 ng / ml, 5 ng / ml, 6 ng / ml, 7 ng / ml, 8 ng / ml, 9 ng / ml, 10 ng / ml, but not limited thereto.
- it is 2 ng / ml.
- it is a method of inducing chondrocytes from iPS cells including the following modified step; (i) a step of inducing mesoderm cells by adhesion culture of iPS cells, (ii) the above step ( a step of adhesion-culturing the cells obtained in i) in a culture medium containing bFGF, ascorbic acid, BMP2, TGF ⁇ and GDF5; and (iii) the cells obtained in the step (ii) ascorbic acid, BMP2, TGF ⁇ and This is performed by a method including a step of suspension culture in a culture solution containing GDF5.
- the mesoderm cells mean cells that develop between the endoderm and the outer lung lobe in the gastrulation stage of the animal.
- it means a cell positive for BRACHYURY.
- BRACHYURY includes NCBI accession numbers as follows. Naturally occurring variants having are included.
- a method for inducing mesoderm cells from iPS cells is not particularly limited, and a method of culturing in a culture solution containing Activin A and a GSK-3 ⁇ inhibitor is exemplified.
- iPS cells are adherently cultured under conditions that do not contain feeder cells, they become an appropriate size (a cell mass containing 1 ⁇ 10 5 to 2 ⁇ 10 5 cells)
- the medium is replaced with a culture solution containing Activin A and a GSK-3 ⁇ inhibitor and cultured.
- adhesion culture can be performed by culturing using a culture vessel coated with an extracellular matrix.
- the coating treatment can be performed by placing a solution containing an extracellular matrix in a culture container and then removing the solution as appropriate.
- the extracellular matrix is a supramolecular structure that exists outside the cell, and may be naturally derived or artificial (recombinant). Examples thereof include substances such as collagen, proteoglycan, fibronectin, hyaluronic acid, tenascin, entactin, elastin, fibrillin, laminin or fragments thereof. These extracellular substrates may be used in combination, and may be prepared from cells such as BD-Matrigel (TM). Examples of the artifact include laminin fragments. In the present invention, laminin is a protein having a heterotrimeric structure having one ⁇ chain, one ⁇ chain, and one ⁇ chain, and is not particularly limited.
- the ⁇ chain includes ⁇ 1, ⁇ 2, ⁇ 3, It is ⁇ 4 or ⁇ 5, the ⁇ chain is ⁇ 1, ⁇ 2 or ⁇ 3, and the ⁇ chain is exemplified by ⁇ 1, ⁇ 2 or ⁇ 3.
- the laminin fragment is not particularly limited as long as it is a laminin fragment having integrin-binding activity, and examples thereof include an E8 fragment which is a fragment obtained by digestion with elastase.
- the culture solution used in this step (i) can be prepared by adding Activin A and a GSK-3 ⁇ inhibitor to a basal medium used for culturing animal cells.
- basal medium include IMDM medium, Medium 199 medium, Eagle's'MinimummEssential Medium (EMEM) medium, ⁇ MEM medium, Dulbecco's modified Eagle's Medium (DMEM) medium, Ham's F12 medium, RPMI 1640 medium, and Fischer's medium.
- the medium may contain serum (for example, FBS) or may be serum-free.
- albumin, transferrin, KnockOut Serum Replacement (KSR) serum substitute for FBS during ES cell culture
- N2 supplement Invitrogen
- B27 supplement Invitrogen
- fatty acids insulin
- It may contain one or more serum substitutes such as sodium selenate, collagen precursor, trace elements, 2-mercaptoethanol, 3'-thiolglycerol, lipids, amino acids, L-glutamine, GlutaMAX (Invitrogen), non It may also contain one or more substances such as essential amino acids (NEAA), vitamins, growth factors, low molecular compounds, antibiotics, antioxidants, pyruvate, buffers, inorganic salts and the like.
- the basal medium is DMEM / F12 containing insulin, transferrin, sodium selenite, and 1% serum.
- Activin A includes Activin A derived from humans and other animals, and functional variants thereof.
- commercially available products such as R & D systems may be used. it can.
- the concentration of Activin A used in this step is 0.1 ng / ml to 1000 ng / ml, preferably 1 ng / ml to 100 ng / ml, more preferably 5 ng / ml to 50 ng / ml, more preferably 10 ng / ml.
- the GSK-3 ⁇ inhibitor is not particularly limited as long as it can directly or indirectly inhibit GSK-3 ⁇ function, for example, kinase activity.
- Wnt3a BIO which is an indirubin derivative (Also known as GSK-3 ⁇ inhibitor IX; 6-bromoindirubin 3′-oxime), a maleimide derivative SB216763 (3- (2,4-dichlorophenyl) -4- (1-methyl-1H-indole-3- Yl) -1H-pyrrole-2,5-dione), GSK-3 ⁇ inhibitor VII (4-dibromoacetophenone) which is a phenyl ⁇ bromomethyl ketone compound, L803-mts (also known as a cell membrane permeation type phosphorylated peptide) GSK-3 ⁇ peptide inhibitors; Myr-N-GKEAPPAPPQSpP-NH 2 ) and CHIR99021 (Nature (2008) 453: 519-523)
- a preferred GSK-3 ⁇ inhibitor used in this step includes Wnt3a.
- Wnt3a includes Wnt3a derived from humans and other animals, and functional variants thereof.
- commercially available products such as R & D systems can be used.
- the concentration of the GSK-3 ⁇ inhibitor used in this step can be appropriately selected by those skilled in the art depending on the GSK-3 ⁇ inhibitor to be used.
- Wnt3a when Wnt3a is used as the GSK-3 ⁇ inhibitor, 0.1 ng / ml To 1000 ng / ml, preferably 1 ng / ml to 100 ng / ml, more preferably 5 ng / ml to 50 ng / ml, still more preferably 10 ng / ml.
- the culture temperature is not particularly limited, but is about 30 to 40 ° C., preferably about 37 ° C., and the culture is performed in an atmosphere of CO 2 -containing air.
- the CO 2 concentration is about 2-5%, preferably about 5%.
- the culture time in this step is, for example, culture for 5 days or less, and preferably 3 days.
- Step (Ii) Step of adhesion culture of the cells obtained in the step (i) in a culture medium containing bFGF, ascorbic acid, BMP2, TGF ⁇ and GDF5
- the cells obtained in the step (i) were obtained. It can be carried out by removing the culture solution of the cell culture and adding a culture solution containing bFGF, ascorbic acid, BMP2, TGF ⁇ and GDF5. Therefore, since the cell culture is adhered to the culture dish in the step (i), this step (ii) can be performed by adhesion culture.
- the culture solution used in this step (ii) can be prepared by adding bFGF, ascorbic acid, BMP2, TGF ⁇ and GDF5 to the basal medium used for culturing animal cells.
- the basal medium include IMDM medium, Medium 199 medium, Eagle's'MinimummEssential Medium (EMEM) medium, ⁇ MEM medium, Dulbecco's modified Eagle's Medium (DMEM) medium, Ham's F12 medium, RPMI 1640 medium, and Fischer's medium.
- the medium may contain serum (for example, FBS) or may be serum-free.
- albumin transferrin, KnockOut Serum Replacement (KSR) (serum substitute for FBS during ES cell culture) (Invitrogen), N2 supplement (Invitrogen), B27 supplement (Invitrogen), fatty acid, insulin, collagen It may contain one or more serum replacements such as precursors, trace elements, 2-mercaptoethanol, 3'-thiolglycerol, lipids, amino acids, L-glutamine, GlutaMAX (Invitrogen), non-essential amino acids (NEAA) It may also contain one or more substances such as vitamins, growth factors, low molecular compounds, antibiotics, antioxidants, pyruvate, buffers, inorganic salts and the like.
- the basal medium is DMEM containing insulin, transferrin, sodium selenite, and 1% serum.
- bFGF includes human and other animal-derived bFGF and functional variants thereof, and for example, commercially available products such as WAKO can be used.
- concentration of bFGF used in this step is 0.1 ng / ml to 1000 ng / ml, preferably 1 ng / ml to 100 ng / ml, more preferably 5 ng / ml to 50 ng / ml, more preferably 10 ng / ml.
- ascorbic acid may be a commercially available product such as Nakarai.
- concentration of ascorbic acid used in this step is 5 to 500 ⁇ g / ml, preferably 10 to 100 ⁇ g / ml, more preferably 50 to ⁇ g / ml.
- BMP2 includes BMP2 derived from humans and other animals, and functional variants thereof, and for example, commercially available products such as Osteopharma can be used.
- the concentration of BMP2 used in this step is 0.1 ng / ml to 1000 ng / ml, preferably 1 ng / ml to 100 ng / ml, more preferably 5 ng / ml to 50 ng / ml, more preferably 10 ng / ml.
- TGF ⁇ includes TGF ⁇ derived from humans and other animals, and functional variants thereof.
- concentration of TGF ⁇ used in this step is 0.1 ng / ml to 1000 ng / ml, preferably 1 ng / ml to 100 ng / ml, more preferably 5 ng / ml to 50 ng / ml, more preferably 10 ng / ml.
- GDF5 includes GDF5 derived from humans and other animals, and functional modifications thereof.
- GDF5 derived from humans and other animals, and functional modifications thereof.
- commercially available products such as PeproTech can be used.
- the concentration of GDF5 used in this step is 0.1 ng / ml to 1000 ng / ml, preferably 1 ng / ml to 100 ng / ml, more preferably 5 ng / ml to 50 ng / ml, more preferably 10 ng / ml.
- the culture temperature is not particularly limited, but is about 30 to 40 ° C., preferably about 37 ° C., and the culture is performed in an atmosphere of CO 2 -containing air.
- the CO 2 concentration is about 2-5%, preferably about 5%.
- the culture time in this step is, for example, 15 days or less, preferably 11 days.
- the cell culture obtained in the step (ii) This can be done by detaching the product from the culture vessel and subjecting it to suspension culture.
- the method of detaching the cell culture is preferably performed by a mechanical separation method (such as pipetting), and a separation solution having protease activity and / or collagenase activity (for example, containing trypsin and collagenase)
- TM Accutase
- TM Accumax
- the suspension culture used in the method of the present invention is culturing cells in a non-adherent state on a culture dish, and is not particularly limited, but is artificially treated for the purpose of improving adhesion with cells ( For example, using a culture vessel that has not been coated with an extracellular matrix or the like, or a culture vessel that has been artificially inhibited from adhesion (eg, coated with polyhydroxyethylmethacrylate (poly-HEMA)) Preferably it is done.
- the culture solution used in this step (iii) can be prepared by adding ascorbic acid, BMP2, TGF ⁇ and GDF5 to the basal medium used for culturing animal cells.
- the basal medium include IMDM medium, Medium 199 medium, Eagle's'MinimummEssential Medium (EMEM) medium, ⁇ MEM medium, Dulbecco's modified Eagle's Medium (DMEM) medium, Ham's F12 medium, RPMI 1640 medium, and Fischer's medium.
- the medium may contain serum (for example, FBS) or may be serum-free.
- albumin transferrin, KnockOut Serum Replacement (KSR) (serum substitute for FBS during ES cell culture) (Invitrogen), N2 supplement (Invitrogen), B27 supplement (Invitrogen), fatty acid, insulin, collagen It may contain one or more serum replacements such as precursors, trace elements, 2-mercaptoethanol, 3'-thiolglycerol, lipids, amino acids, L-glutamine, GlutaMAX (Invitrogen), non-essential amino acids (NEAA) It may also contain one or more substances such as vitamins, growth factors, low molecular compounds, antibiotics, antioxidants, pyruvate, buffers, inorganic salts and the like.
- the basal medium is DMEM containing insulin, transferrin, sodium selenite, and 1% serum.
- ascorbic acid may be a commercially available product such as Nakarai.
- concentration of ascorbic acid used in this step is 5 to 500 ⁇ g / ml, preferably 10 to 100 ⁇ g / ml, more preferably 50 to ⁇ g / ml.
- BMP2 includes BMP2 derived from humans and other animals, and functional variants thereof, and for example, commercially available products such as Osteopharma can be used.
- the concentration of BMP2 used in this step is 0.1 ng / ml to 1000 ng / ml, preferably 1 ng / ml to 100 ng / ml, more preferably 5 ng / ml to 50 ng / ml, more preferably 10 ng / ml.
- TGF ⁇ includes TGF ⁇ derived from humans and other animals, and functional variants thereof.
- concentration of TGF ⁇ used in this step is 0.1 ng / ml to 1000 ng / ml, preferably 1 ng / ml to 100 ng / ml, more preferably 5 ng / ml to 50 ng / ml, more preferably 10 ng / ml.
- GDF5 includes GDF5 derived from humans and other animals, and functional modifications thereof.
- GDF5 derived from humans and other animals, and functional modifications thereof.
- concentration of GDF5 used in this step is 0.1 ng / ml to 1000 ng / ml, preferably 1 ng / ml to 100 ng / ml, more preferably 5 ng / ml to 50 ng / ml, more preferably 10 ng / ml.
- the culture temperature is not particularly limited, but is about 30 to 40 ° C., preferably about 37 ° C., and the culture is performed in an atmosphere of CO 2 -containing air.
- the CO 2 concentration is about 2-5%, preferably about 5%.
- the culture time in this step is, for example, 10 days or more and 30 days or less, preferably 14 days or more and 28 days or less.
- Step of further culturing cells obtained in the step (iii) in suspension culture The chondrocytes can be produced by the step (iii), but in order to obtain a more mature chondrocyte, the step The cell culture obtained in (iii) may be further subjected to suspension culture.
- the culture solution used in this step (iv) is a basal medium used for animal cell culture.
- the basal medium include IMDM medium, Medium 199 medium, Eagle's'MinimummEssential Medium (EMEM) medium, ⁇ MEM medium, Dulbecco's modified Eagle's Medium (DMEM) medium, Ham's F12 medium, RPMI 1640 medium, and Fischer's medium.
- the medium may contain serum (for example, FBS) or may be serum-free.
- albumin transferrin, KnockOut Serum Replacement (KSR) (serum substitute for FBS during ES cell culture) (Invitrogen), N2 supplement (Invitrogen), B27 supplement (Invitrogen), fatty acid, insulin, collagen It may contain one or more serum replacements such as precursors, trace elements, 2-mercaptoethanol, 3'-thiolglycerol, lipids, amino acids, L-glutamine, GlutaMAX (Invitrogen), non-essential amino acids (NEAA) It may also contain one or more substances such as vitamins, growth factors, low molecular compounds, antibiotics, antioxidants, pyruvate, buffers, inorganic salts and the like. In one embodiment of this step, the basal medium is DMEM with 10% serum.
- the culture temperature is not particularly limited, but is about 30 to 40 ° C., preferably about 37 ° C., and the culture is performed in an atmosphere of CO 2 -containing air.
- the CO 2 concentration is about 2-5%, preferably about 5%. Since the culture time of this step is not particularly long and causes no problems in the production of chondrocytes, for example, a culture period of 20 days or longer is exemplified, and preferably 28 days or longer.
- Chondrocytes obtained by the method of the present invention can be extracted by selectively excluding cells adhering to the culture vessel in view of the fact that many cells are floating in the culture medium. preferable.
- the present invention relates to a method for screening a therapeutic and / or prophylactic agent for FGFR3 disease, wherein the iPS cell origin obtained as described above is brought into contact with a test substance, and each index is used to treat and / or prevent FGFR3 disease.
- Methods for screening drug test substances are provided.
- a therapeutic and / or prophylactic agent for FGFR3 disease can be screened by a method comprising the following steps; (A) a step of differentiating an induced pluripotent stem (iPS) cell produced from a somatic cell derived from a subject suffering from FGFR3 disease into a chondrocyte under a condition in which the test substance is brought into contact with and not in contact with the test substance; (B) measuring one or more indicators selected from the group consisting of the amount of chondrocyte extracellular matrix, chondrocyte marker gene and fibroblast marker gene expression in the culture obtained in step (a) And (c) when the amount of the chondrocyte extracellular matrix or the chondrocyte marker gene is increased or the expression level of the fibroblast marker gene is decreased under the condition of contacting with the test substance under the condition of not contacting, A step of selecting a test substance as a therapeutic or preventive for FGFR3 disease.
- iPS induced pluripotent stem
- Step (a) is the above-described step of inducing differentiation into chondrocytes, and the test substance may be brought into contact with the cells in all steps, preferably, the step (2) and / or the step (3). Alternatively, they may be contacted in the step (ii) and / or the step (iii), and more preferably in contact with the cells in the step (ii) and the step (iii).
- iPS cells prepared from somatic cells derived from a subject suffering from FGFR3 disease may have mutations specific to FGFR3 disease patients in the FGFR3 gene. Mutations peculiar to FGFR3 disease patients include Arg246Cys, Arg248Cys, Ser249Cys, Gly370Cys, Ser371Cys, Thr373Cys, Lys650Glu, X807Arg, X807Cys, etc. However, it is not limited to these.
- any test substance can be used, and any known compound and novel compound may be used, for example, cell extracts, cell culture supernatants, microbial fermentation products, marine organism-derived extracts.
- Product plant extract, purified protein or crude protein, peptide, non-peptide compound, synthetic low molecular weight compound, natural compound and the like.
- the test substance is also (1) biological library method, (2) synthetic library method using deconvolution, (3) “one-beadoneone-compound” live Can be obtained using any of a number of approaches in combinatorial library methods known in the art, including rally methods and (4) synthetic library methods using affinity chromatography sorting.
- Biological library methods using affinity chromatography sorting are limited to peptide libraries, but the other four approaches can be applied to small molecule compound libraries of peptides, non-peptide oligomers, or compounds (Lam (1997) Anticancer Drug Des. 12: 145-67).
- Examples of methods for the synthesis of molecular libraries can be found in the art (DeWitt et al. (1993) Proc. Natl. Acad. Sci. USA 90: 6909-13; Erb et al. (1994) Proc. Natl. Acad. Sci. USA 91: 11422-6; Zuckermann et al. (1994) J. Med. Chem. 37: 2678-85; Cho et al.
- the target disease in the screening method of the present invention may be a disease related to abnormal bone formation, particularly FGFR3 disease.
- FGFR3 disease includes, but is not limited to, fatal osteodysplasia TD (TD), achondroplasia A (ACH), and hypochondral dysplasia in order of severity.
- the target disease in the screening method of the present invention is preferably lethal bone dysplasia (TD) and / or achondroplasia (ACH).
- this can be achieved by measuring the expression level of each marker gene in chondrocytes.
- the marker gene include a chondrocyte marker gene and a fibroblast marker gene.
- the chondrocyte marker gene include, but are not limited to, SOX9, AGGRECAN, COL2, and the like.
- fibroblast marker genes include, but are not limited to, COL1A1, COL1A2, and the like.
- the amount of chondrocytes can also be measured by a method such as antibody staining that targets a gene (protein) specifically expressed in chondrocytes.
- the amount of the cartilage extracellular matrix in the present invention can be measured using a substance that specifically stains cartilage-like tissue.
- a substance that specifically stains cartilage-like tissue include, but are not limited to, safranin O and its analogs.
- the substance obtained by the screening method of the present invention is useful as an active ingredient for various therapeutic and / or prophylactic agents for cartilage diseases.
- the cartilage disease to be treated according to the present invention may be any disease that can be treated and / or prevented by increasing the amount of cartilage that is decreased than usual by some means. Examples of the cartilage disease to be treated according to the present invention include, but are not limited to, osteoarthritis, cartilage damage, and cartilage dysplasia.
- the cartilage disease to be treated according to the present invention is most preferably indicated as a disease caused by cartilage dysplasia in the cartilage growth process, and is not particularly limited as long as it is any disease, for example, Examples include pathological conditions having mutations in FGFR (FGFR1, FGFR2, and FGFR3). Examples of such pathological conditions include the diseases described in Warman et al., Am J Med Genet 155A (5): 943-68 (2011). In using the therapeutic agents found in the present invention, FGFR3 disease is a preferred indication.
- a preferable therapeutic and / or prophylactic agent for a disease having a mutation of FGFR is a medicine containing an HMG-CoA reductase inhibitor.
- HMG-CoA reductase inhibitor examples include mevastatin (compactin) (see USP3983140), pravastatin (see Japanese Patent Application Laid-Open No. 57-2240 (USP4346227)), lovastatin (Japanese Patent Application Laid-Open No.
- the HMG-CoA reductase inhibitor in the present invention is preferably a drug selected from the group consisting of mevastatin, atorvastatin, pravastatin, rosuvastatin, fluvastatin and lovastatin.
- the HMG-CoA reductase inhibitor mevastatin, pravastatin, lovastatin, simvastatin, fluvastatin, atorvastatin, rosuvastatin or pitavastatin of the present invention is a lactone ring ring or a pharmacologically acceptable salt thereof (preferably , Sodium salt or calcium salt).
- compositions for oral administration include solid or liquid dosage forms, specifically tablets (including dragees and film-coated tablets), pills, granules, powders, capsules (including soft capsules). Syrup, emulsion, suspension and the like.
- compositions for parenteral administration for example, injections, suppositories and the like are used, and injections include intravenous injections, subcutaneous injections, intradermal injections, intramuscular injections, infusions, and the like. Dosage forms may be included.
- excipients eg sugar derivatives such as lactose, sucrose, sucrose, mannitol, sorbitol; starch derivatives such as corn starch, potato starch, alpha starch, dextrin; cellulose derivatives such as crystalline cellulose; Gum arabic; dextran; organic excipients such as pullulan; and silicate derivatives such as light anhydrous silicic acid, synthetic aluminum silicate, calcium silicate and magnesium metasilicate aluminate; phosphates such as calcium hydrogen phosphate; Carbonates such as calcium; inorganic excipients such as sulfates such as calcium sulfate), lubricants (eg, stearic acid metal salts such as stearic acid, calcium stearate, magnesium stearate; talc; Colloidal silica; like bead wax, gay wax Boric acid; adipic acid; sulfate such as sodium sulfate; glycol; fumaric excipient
- the dose of the drug of the present invention to a patient varies depending on the type of pathological condition to be treated, symptoms and severity of the disease, patient age, sex or body weight, administration method, etc. By determining in consideration of the above situation, an appropriate dose can be determined as appropriate.
- the lower limit is 0.1 mg (preferably 0.5 mg) and the upper limit is 1000 mg (preferably 500 mg).
- the lower limit is 0.01 mg (preferably 0.05 mg) and an upper limit of 100 mg (preferably 50 mg) can be administered to adults 1 to 6 times per day. The dose may be increased or decreased depending on the symptoms.
- HDFs (TD-714, TD10749, and TD-315H) from three TD patients were obtained from the Coryell Medical Research Institute and Saitama Children's Medical Center. Sequence analysis of genomic cDNA extracted from these HDFs confirmed the heterozygous mutation (Arg248Cys) of the FGFR3 gene in all TD patients.
- iPS cells were prepared from the HDF derived from each patient by the method described below (hereinafter referred to as TD-iPSC), and one iPS cell line (TD-714-3, TD10749-2 and TD315H) was obtained from each patient. -2) was used for the analysis.
- Control HDFs from two different neonates (Strain # 01491 and # 01439) were purchased from KURABO and initialized in a similar manner to generate control iPS cells (KF4009-1 and HDF-11).
- iPS cell line (409B2) derived from healthy individuals obtained from K. Okita and S. Yamanaka (iPS Cell Research Institute, Kyoto University) (Okita, K., et al. Nature methods 8, 409-412 (2011)) were also used as control iPS cells (KF4009-1, HDF-11 and 409B2 are hereinafter referred to as WT-iPSC).
- iPS cells were produced by the following method. Specifically, each obtained human fibroblast (HDF) was cultured in DMEM (Sigma) supplemented with 10% FBS (Invitrogen), 50 U / ml penicillin and 50 ⁇ g / ml streptomycin. Subsequently, episomal plasmid vectors (Mixture Y4: OCT3 / 4, SOX2, KLF4, L-MYC, LIN28 and p53 shRNA) were electroporated into each HDF by Neon transfection system (Invitrogen) (Okita, K ., et al. Nature methods 8, 409-412 (2011)).
- Neon transfection system Invitrogen
- Example 2 Differentiation from each iPS cell was induced to chondrocytes according to the method described below, which was modified from a method reported before cartilage induction (Oldershaw, RA, et al., Nat Biotechnol 28, 1187-1194 (2010)).
- Each iPS cell was seeded on a Matrigel (Invitrogen) coated dish, a medium supplemented with 50 units / ml penicillin and 50 ⁇ g / ml streptomycin was added to Essential 8 medium (Life Technologies), and cultured under feeder-free conditions.
- colonies consisting of 1-2 ⁇ 10 5 cells were transformed into mesodermal differentiation medium (10 ng / ml Wnt3A (R & D), 10 ng / ml activin A (R & D), 1% insulin for DMEM / F12) -Transferrin-sodium selenite (Invitrogen), 1% fetal bovine serum, 50 units / ml penicillin, 50 ⁇ g / ml prepared by mixing streptomycin) (Day 0 of differentiation induction).
- mesodermal differentiation medium (10 ng / ml Wnt3A (R & D), 10 ng / ml activin A (R & D), 1% insulin for DMEM / F12) -Transferrin-sodium selenite (Invitrogen), 1% fetal bovine serum, 50 units / ml penicillin, 50 ⁇ g / ml prepared by mixing streptomycin) (Day 0 of differentiation induction).
- cartilage differentiation medium 50 ⁇ g / ml ascorbic acid, 10 ng / ml BMP2 (Osteopharma), 10 ng / ml TGF ⁇ (Pepro Tech), 10 ng / ml GDF5, 1% insulin-transferrin-sub DMEM / F12) containing sodium selenate, 1% FBS, 50 units / ml penicillin and 50 ⁇ g / ml streptomycin, and after 11 days (14 days of differentiation induction), the cells are detached from the dish and the same medium is used. Particles formed by suspension culture after 14 days (differentiation induction day 28) and 28 days (differentiation induction day 42) were collected and analyzed. During differentiation induction, the medium was changed every 2 to 7 days.
- each chondrocyte marker gene SOX9, COL2 and AGGRECAN
- type I collagen gene on the 28th day of differentiation induction was examined.
- the expression of each chondrocyte marker gene decreased and the expression of type I collagen gene increased (FIG. 3a).
- chondrocytes derived from TD-iPSC and WT-iPSC on the 28th day were compared with chondrocytes derived from WT-iPSC. A significant decrease in growth rate was shown (FIGS. 5a and b).
- TUNEL assay was performed on TD-iPSC-derived cartilage particles on the differentiation induction day 21, an increase in the number of TUNEL-positive cells, that is, an increase in cells that caused apoptosis was confirmed (FIGS. 5c and d). .
- Example 3 Recovery from disturbed cartilage formation of chondrogenically differentiated TD-iPSC TD-iPSC knocks FGFR3 to confirm whether the gain-of-function mutation of FGFR3 is due to chondrocyte differentiation deficiency An experiment to go down was conducted.
- ShRNA PB vector and transposase expression vector were obtained from A. Hotta (Center iPS Cell Research and Application (CiRA), KyotoUniversity, Kyoto, Japan).
- a piggybac vector FGFR3 shRNA PB vector
- shRNA short hairpin RNAs
- FGFR3 shRNA PB vector and PBaseII were introduced into TD-iPSC (TD714-3) using Ncleofection (Amaxa) according to the manufacturer's instructions to prepare TD-iPSC in which FGFR3 was knocked down.
- differentiation induction into chondrocytes was performed according to the method described in Example 2, and evaluation was performed using particles on the 28th and 42nd days of differentiation induction.
- RNA expression analysis was performed according to the method described in Example 1.
- FGFR3FGFshRNA PB vectors were confirmed to have a function of knocking down the FGFR3 gene (Fig. 6b).
- FGFR3 of TD-iPSC was knocked down using these FGFR3 shRNA PB vectors, it was confirmed that particles having a chondrocyte extracellular matrix were formed (FIGS. 6c and d).
- the TD-iPSC-derived particles into which FGFR3 shRNA was introduced showed a chondrocyte marker compared to the case of introducing the negative control shLuciferase (shRNA targeting the luciferase gene sequence). It was found that gene expression increased and fibroblast marker gene (COL1A1 and COL1A2) expression decreased (FIG. 6e).
- FGFR3 neutralizing antibody was added to the medium to suppress the activity of FGFR3, and chondrocyte formation was confirmed.
- the FGFR3 neutralizing antibody (Santa Cruz (sc-13121)) was added by adding 1 ⁇ antibody solution (200 ng / ml) to 1 ml cartilage differentiation medium.
- IgG Cell Signaling, # 27295.
- Induction of chondrocyte differentiation was performed according to the method described in Example 2 and evaluated on the 28th and 42nd days of differentiation induction.
- Example 4 Evaluation of known drugs to normalize chondrocyte differentiation failure of TD-iPSC Screening for substances that normalize chondrocyte differentiation from TD-iPSC in order to find effective drugs for the treatment of FGFR3 disease It was.
- CNP chondrocyte differentiation from TD-iPSC using drugs reported to be involved in FGFR3 signaling and / or chondrocyte differentiation
- CNP drugs reported to be involved in FGFR3 signaling and / or chondrocyte differentiation
- CNP Sigma, N8768
- NF449 abcam, ab120415
- FGFR inhibitor PD 173047, Cayman
- IGF1R inhibitor IGF-1R inhibitor, PPP. Calbiochem, 407247
- Example 2 After adding the stock solution to the cartilage differentiation medium so that the final concentrations of CNP, NF449, FGFRinhibitor and IGF1Rinhibitor are 100 nM, 25 ⁇ M, 1 ⁇ M and 1 nM, respectively, described in Example 2 According to the above method, differentiation into chondrocytes was induced and evaluated on the 28th and 42nd days after induction of differentiation. As a control, an equal volume of water or DMSO was added to the medium. mRNA expression analysis was performed according to the method described in Example 1.
- Example 5 Search for drugs to normalize TD-iPSC chondrocyte insufficiency statins are substances that form a class of drugs that are widely characterized as lipid-lowering agents and inhibit mevalonic acid synthesis, resulting in total cholesterol It is known to reduce the amount and low density lipoprotein (LDL) levels. Since all statins have a beneficial effect on cardiovascular disease, nervous system, immune system, skeletal system and tumor growth, there is an increasing interest in the multifaceted effects of statins. Furthermore, lovastatin has been reported to suppress the loss of type II collagen due to dedifferentiation caused by monolayer culture of cells isolated from nucleus pulposus (Hu et al., Artif Organs. 35, 411- 416 2011).
- lovastatin normalizes chondrocyte differentiation from TD-iPSC
- evaluation was performed in the same manner as in Example 4.
- lovastatin (TCI, L0214) was prepared by dissolving it in DMSO at a concentration of 10 MmM, and used so that the final concentration of the cartilage differentiation medium was 1 ⁇ M.
- lovastatin normalizes chondrocyte differentiation from TD-iPSC (FIGS. 9a and b).
- the addition of lovastatin increased the expression of chondrocyte marker gene and decreased the expression of fibroblast marker gene (FIG. 9c).
- BrdU incorporation of TD-iPSC-derived cartilage particles cultured in the presence and absence of rosuvastatin was confirmed, it was confirmed that the number of BrdU-positive cells was significantly higher in the rosuvastatin addition group (FIGS. 10a and b) .
- statins were prepared as a 10 mM DMSO solution and used by appropriately adding to the culture solution so that the final concentration was 1 ⁇ M.
- Example 6 Effect of Lovastatin on ACH To examine whether lovastatin is effective against ACH, the same evaluation was performed using iPS cells derived from ACH patients.
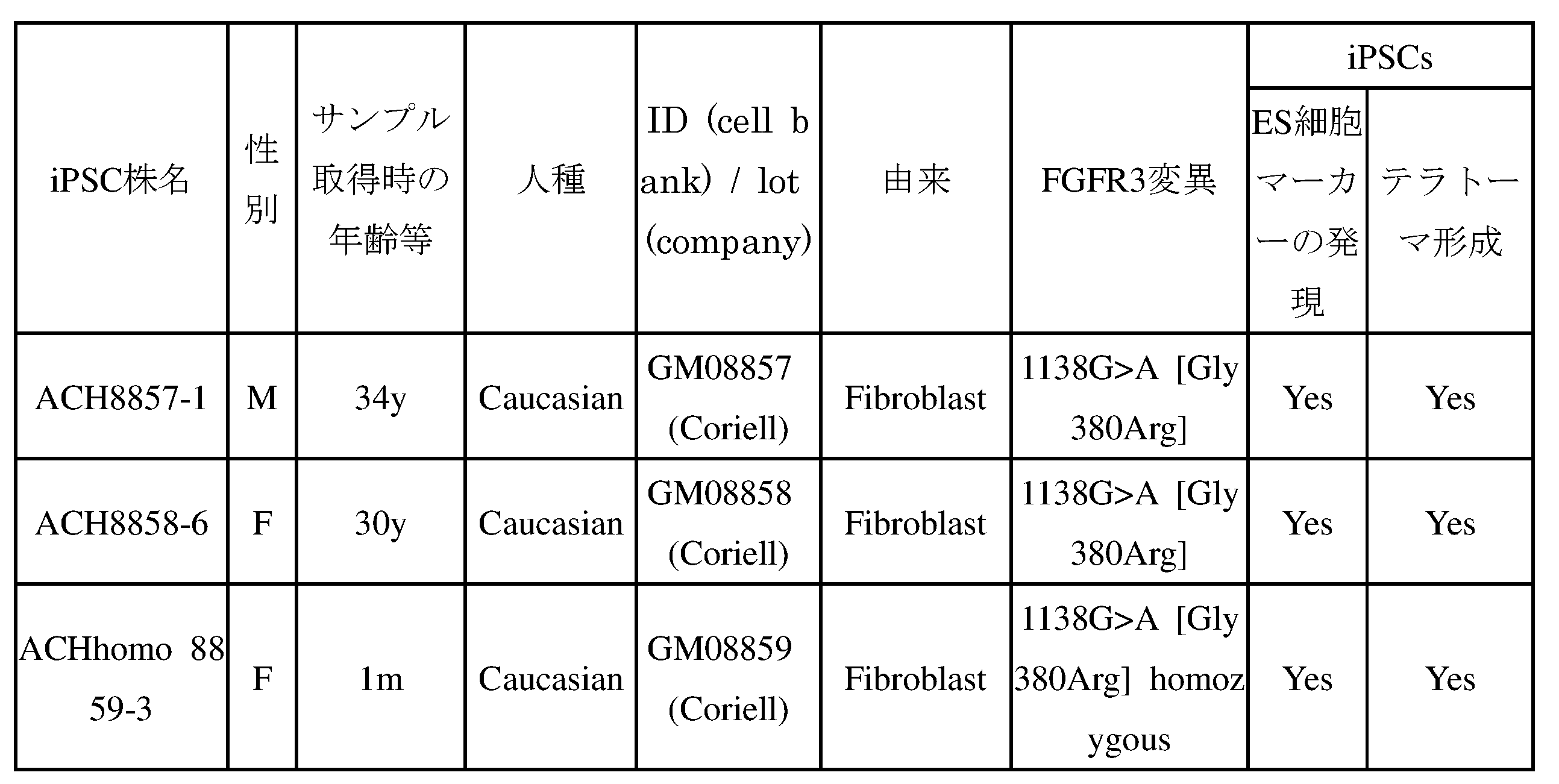
- IPS cells from ACH patients are HDF (ACH8857 and ACH8858) of two ACH patients with a Gly380Arg heterozygous mutation in the FGFR3 gene, and HDF (ACHhomo-8859) showing more severe chondrogenesis than ACH was obtained from the Coryell Institute of Medicine and prepared by the method described in Example 1.
- ACHhomo-8859 had a Gly380Arg homozygous mutation in the FGFR3 gene.
- One iPSC strain (ACH8857-1, ACH8858-6 and ACHhomo8859-3) was prepared from each patient (hereinafter referred to as ACH8857-1 and ACH8858-6 as ACH-iPSC, and ACHhomo8859-3 as ACHhomo-iPSC) ). It was confirmed that ACH-iPSC and ACHhomo-iPSC express SSEA4 and TRA1-60 and form teratomas including three germ layers (Table 4).
- lovastatin can normalize cartilage formation in both TD-iPSC and ACH-iPSC models.
- Example 7 Restoration of reduced bone growth in ACH model mice by statins
- statins can normalize the FGFR3 disease phenotype in vivo.
- FGFR3 Ach mice Naski et al., Development 125, 4977-4988 (1998)) obtained from David Ornitz (Washington University School of Medicine) were crossed with wild type mice (C57BL / 6 background). From 3 to 14 days after birth of the crossbred mice, a 1.0 mg / kg rosuvastatin solution was intraperitoneally administered 6 times a week, and euthanized on the 15th day. X-ray images of rods and bone formation (Faxitron DX-50 ). At this time, genomic DNA was extracted from the toes and examined for genotype.
- lovastatin solution was intraperitoneally administered 6 times a week from the 3rd to 28th day after birth of the crossbred mice, and euthanized on the 29th day to evaluate rods and bone formation.
- FGFR3 Ach mice are dwarfism-like and show short limb bones and short noses.
- intraperitoneal administration of rosuvastatin anteroposterior length of skull (Cranial AP), length of ulna (Ulna), length of femur (Femur) and tibia (Tibia) of 15-day-old FGFR3 Ach mice The length was increased ( Figures 12a and b and Figure 13).
- rosuvastatin administration improved to the same shape as the wild-type.
- lovastatin administration increased body length and long bone length of FGFR3 Ach mice (FIGS. 14a and b, FIG. 15 and Table 5).
- administration of lovastatin showed an effect of normalizing the length of the nose of FGFR3 Ach mice to some extent (FIGS. 14c and d, FIG. 15 and Table 5).
- FGFR3 Ach model mice were also shown to alleviate bone formation inhibition by statin administration.
- Metatarsal bones were collected from 15.5 dpc mouse embryos obtained by mating FVB ⁇ C57Bl / 6 and performed according to Ikegami, D. et al., Osteoarthritis Cartilage 19, 233-241, 2011. Briefly, genotype confirmation was performed by treating metatarsals from FGFR3 Ach mouse embryos with 1 ⁇ M lovastatin or substrate.
- statins promotes the induction of cartilage differentiation by increasing the expression of Sox9 and Runx2, and further matures the cartilage by hypertrophy.
- the present invention is based on the successful reproduction of the pathology of FGFR3 disease by causing iPS cells derived from somatic cells of FGFR3 disease patients to form chondrocytes through induction of differentiation into chondrocytes. Therefore, it is possible to screen for therapeutic and / or prophylactic agents for FGFR3 disease using the cells.
- the present invention provides a substance that promotes differentiation into chondrocytes obtained as a result of the screening, and can be used as a new therapeutic agent and / or preventive agent for FGFR3 disease.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Chemical & Material Sciences (AREA)
- Biomedical Technology (AREA)
- General Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Organic Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Biotechnology (AREA)
- Epidemiology (AREA)
- Zoology (AREA)
- Wood Science & Technology (AREA)
- Genetics & Genomics (AREA)
- Cell Biology (AREA)
- Immunology (AREA)
- Biochemistry (AREA)
- Microbiology (AREA)
- Molecular Biology (AREA)
- Hematology (AREA)
- Urology & Nephrology (AREA)
- General Engineering & Computer Science (AREA)
- Rheumatology (AREA)
- Analytical Chemistry (AREA)
- Developmental Biology & Embryology (AREA)
- Pathology (AREA)
- Physics & Mathematics (AREA)
- Food Science & Technology (AREA)
- Toxicology (AREA)
- General Physics & Mathematics (AREA)
- Tropical Medicine & Parasitology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Emergency Medicine (AREA)
- Transplantation (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Chemical Kinetics & Catalysis (AREA)
Abstract
Description
[1] 有効成分としてHMG-CoA還元酵素阻害薬を含む、FGFR3病の治療および/または予防用医薬。
[2] 前記HMG-CoA還元酵素阻害薬が、メバスタチン、アトルバスタチン、プラバスタチン、ロスバスタチン、フルバスタチンおよびロバスタチンから成る群より選択される薬剤である、[1]に記載の医薬。
[3] 前記FGFR3病が、致死性骨異形成症 (TD) および/または軟骨無形成症 (ACH)である、[1]または[2]に記載の医薬。
[4] FGFR3病を治療および/または予防するための方法であって、HMG-CoA還元酵素阻害薬を投与することを含む、方法。
[5] 前記HMG-CoA還元酵素阻害薬が、メバスタチン、アトルバスタチン、プラバスタチン、ロスバスタチン、フルバスタチンおよびロバスタチンから成る群より選択される薬剤である、[4]に記載の方法。
[6] 前記FGFR3病が、致死性骨異形成症 (TD) および/または軟骨無形成症 (ACH)である、[4]または[5]に記載の方法。
[7] FGFR3病の治療および/または予防用医薬の製造におけるHMG-CoA還元酵素阻害薬の使用。
[8] 前記HMG-CoA還元酵素阻害薬が、メバスタチン、アトルバスタチン、プラバスタチン、ロスバスタチン、フルバスタチンおよびロバスタチンから成る群より選択される薬剤である、[7]に記載の使用。
[9] 前記FGFR3病が、致死性骨異形成症 (TD) および/または軟骨無形成症 (ACH)である、[7]または[8]に記載の使用。
[10] 下記の工程を含む、FGFR3病の治療薬および/または予防薬をスクリーニングする方法;
(a)FGFR3に変異を有する人工多能性幹(iPS)細胞を、被験物質と接触させるおよび接触させない条件下で軟骨細胞へと分化させる工程、
(b)工程(a)で得られた培養物中における軟骨細胞外マトリックスの量、軟骨細胞マーカー遺伝子および線維芽細胞マーカー遺伝子の発現量から成る群より選択される一つ以上の指標を測定する工程、および
(c)被験物質と接触させる条件下において、接触させない条件下より軟骨細胞外マトリックス量または軟骨細胞マーカー遺伝子が増加した場合、もしくは線維芽細胞マーカー遺伝子の発現量が減少した場合、当該被験物質をFGFR3病の治療薬または予防薬として選出する工程。
[11] 前記工程(a)における軟骨細胞への分化が下記の工程を含む、[10]に記載の方法:
(i)多能性幹細胞を接着培養することにより中胚葉細胞を誘導する工程、
(ii)前記工程(i)で得られた細胞をbFGF、アスコルビン酸、BMP2、TGFβ、GDF5および被験物質を含む培養液中で接着培養する工程、および
(iii)前記工程(ii)で得られた細胞をアスコルビン酸、BMP2、TGFβ、GDF5および被験物質を含む培養液中で浮遊培養する工程。
[12] 前記軟骨細胞マーカー遺伝子が、SOX9、AGGRECANおよびCOL2からなる群から選択される少なくとも一つの遺伝子である、[10]または[11]に記載の方法。
[13] 前記線維芽細胞マーカー遺伝子が、COL1A1および/またはCOL1A2である、[10]または[11]に記載の方法。
[14] 前記FGFR3の変異が、FGFR3中のArg248CysまたはGly380Arg変異である、[10]から[13]のいずれか1項に記載の方法。
[15] 前記FGFR3病が、致死性骨異形成症 (TD)および/または軟骨無形成症 (ACH)である、[10]から[14]のいずれか1項に記載の方法。
[16] FGFR3病を治療および/または予防するために使用されるHMG-CoA還元酵素阻害薬。
[17] 前記HMG-CoA還元酵素阻害薬がメバスタチン、アトルバスタチン、プラバスタチン、ロスバスタチン、フルバスタチンロバスタチンから成る群より選択される薬剤である、[16]に記載のHMG-CoA還元酵素阻害薬。
[18] 前記FGFR3病が、致死性骨異形成症 (TD) および/または軟骨無形成症 (ACH)である、[17]または[18]に記載のHMG-CoA還元酵素阻害薬。
本発明において、iPS細胞は、ある特定の核初期化物質を、DNAまたはタンパク質の形態で体細胞に導入すること、または薬剤によって当該核初期化物質の内在性のmRNAおよびタンパク質の発現を上昇させることによって作製することができる、ES細胞とほぼ同等の特性、例えば分化多能性と自己複製による増殖能を有する体細胞由来の人工の幹細胞である(K. Takahashi and S. Yamanaka (2006) Cell, 126: 663-676、K. Takahashi et al. (2007) Cell, 131: 861-872、J. Yu et al. (2007) Science, 318: 1917-1920、M. Nakagawa et al. (2008) Nat. Biotechnol., 26: 101-106、国際公開WO 2007/069666および国際公開WO 2010/068955)。核初期化物質は、ES細胞に特異的に発現している遺伝子またはES細胞の未分化維持に重要な役割を果たす遺伝子もしくはその遺伝子産物であればよく、特に限定されないが、例えば、Oct3/4, Klf4, Klf1, Klf2, Klf5, Sox2, Sox1, Sox3, Sox15, Sox17, Sox18, c-Myc, L-Myc, N-Myc, TERT, SV40 Large T antigen, HPV16 E6, HPV16 E7, Bmil, Lin28, Lin28b, Nanog, Esrrb, Esrrg, Glis1などが例示される。これらの初期化物質は、iPS細胞樹立の際には、組み合わされて使用されてもよい。例えば、上記初期化物質を、少なくとも1つ、2つもしくは3つ含む組み合わせであり、好ましくは4つもしくは5つを含む組み合わせである。
遺伝子名 マウス ヒト
L-Myc NM_008506 NM_001033081
Lin28 NM_145833 NM_024674
Lin28b NM_001031772 NM_001004317
Esrrb NM_011934 NM_004452
Esrrg NM_011935 NM_001438
Glis1 NM_147221 NM_147193
iPS細胞から軟骨細胞を分化誘導する方法としては、当分野において用いられる任意の方法を採用することができ、本願出願時において当業者に知られていた方法のみならず、本願出願後において開発された分化誘導方法を採用することも可能である。軟骨細胞を分化誘導する方法として、例えば、Koyama, N. et al. Stem cells and development 22, 102-113 (2013)、Hwang, N.S., et al. PLoS ONE 3, e2498 (2008)、Oldershaw, R.A. et al. Nat. Biotechnol. 28, 1187-1194 (2010)、Bai, H.Y., et al. Journal of biomedical materials research. Part A 94, 539-546 (2010)、Yamashita, A. et al. Scientific Reports 3 (2013)などに記載の方法が挙げられるが、これらに限定されない。
本発明において、中胚葉細胞とは、動物の発生期の原腸胚期において、内胚葉と外肺葉の間に発生する細胞を意味し、好ましくは、BRACHYURYが陽性である細胞を意味する。本発明において、BRACHYURYには、NCBIのアクセッション番号として、ヒトの場合、NM_001270484またはNM_003181、マウスの場合、NM_009309に記載されたヌクレオチド配列を有する遺伝子並びに当該遺伝子にコードされるタンパク質、ならびにこれらの機能を有する天然に存在する変異体が包含される。
本工程(ii)では、前記工程(i)で得られた細胞培養物の培養液を除去し、bFGF、アスコルビン酸、BMP2、TGFβおよびGDF5を含有する培養液を添加して行い得る。従って、前記工程(i)において細胞培養物は、培養皿へ接着していることから、本工程(ii)は接着培養によって行い得る。
本工程(iii)では、前記工程(ii)で得られた細胞培養物を培養容器より剥離させ、浮遊培養することで行い得る。本工程(iii)において、細胞培養物を剥離させる方法は、力学的分離方法(ピペッティング等)により行うことが好ましく、プロテアーゼ活性および/またはコラゲナーゼ活性を有する分離溶液(例えば、トリプシンとコラゲナーゼの含有溶液であるAccutase(TM)およびAccumax(TM)(Innovative Cell Technologies, Inc)が挙げられる)を用いない方法が好ましい。
前記工程(iii)により、軟骨細胞を製造することが可能であるが、より成熟した軟骨細胞を得るために、前記工程(iii)で得られた細胞培養物をさらに、浮遊培養してもよい。
本発明は、前述のように得られたiPS細胞由と被験物質とを接触させ、各指標を用いて、FGFR3病の治療薬および/または予防薬の被験物質をスクリーニングする方法を提供する。
(a)FGFR3病を患う対象由来の体細胞から作製された人工多能性幹(iPS)細胞を、被験物質と接触させるおよび接触させない条件下で軟骨細胞へと分化させる工程、
(b)工程(a)で得られた培養物中における軟骨細胞外マトリックスの量、軟骨細胞マーカー遺伝子および線維芽細胞マーカー遺伝子の発現量から成る群より選択される一つ以上の指標を測定する工程、および
(c)被験物質と接触させる条件下において、接触させない条件下より軟骨細胞外マトリックス量または軟骨細胞マーカー遺伝子が増加した場合、もしくは線維芽細胞マーカー遺伝子の発現量が減少した場合、当該被験物質をFGFR3病の治療薬または予防薬として選出する工程。
本発明のスクリーニング方法によって得られる物質は、種々の軟骨疾患の治療剤および/または予防剤の有効成分として有用である。本発明の処置対象となる軟骨疾患は、通常よりも減少した軟骨の量を何らかの手段で増加させることにより治療および/または予防が可能である疾患であれば何でもよい。本発明の処置対象となる軟骨疾患として、例えば、変形性関節症、軟骨損傷、軟骨形成異常症などが挙げられるがこれらに限定されない。本発明の処置対象となる軟骨疾患は、軟骨の成長過程における軟骨形成不全による疾患が適応症として最も好ましく、このような病態であれば、いずれの疾患でもあっても特に限定されないが、例えば、FGFR(FGFR1、FGFR2およびFGFR3)における変異を有する病態が挙げられる。このような病態として、Warman et al., Am J Med Genet 155A(5):943-68 (2011)に記載の疾患が例示される。本発明において見出された治療剤を使用するにあたり、FGFR3病が好ましい適応症例である。
iPS細胞の作製
以下のすべての実験は、治験審査委員会、動物実験委員会および施設内生物安全委員会、ならびに京都大学の承認を得て行われた。
3人のTD患者由来のHDF (TD-714, TD10749およびTD-315H) を、コリエル医学研究所および埼玉県立小児医療センターから入手した。これらのHDFから抽出したゲノムcDNAを配列解析したところ、すべてのTD患者においてFGFR3遺伝子のヘテロ接合突然変異(Arg248Cys)が確認された。続いて各患者由来のHDFから以下に記載する方法でiPS細胞を作製し(以下、TD-iPSCと言う)、各患者から1種のiPS細胞株(TD-714-3、TD10749-2およびTD315H-2)を解析に用いた。また、2人の異なる新生児由来の対照HDFs(Strain #01491および#01439)をKURABOから購入し、同様の方法で初期化し、対照iPS細胞を作製した(KF4009-1およびHDF-11)。このほかにも、K. OkitaおよびS. Yamanaka(京都大学iPS細胞研究所)から入手した健常な個体由来のiPS細胞株(409B2)(Okita, K., et al. Nature methods 8, 409-412 (2011))も対照iPS細胞として用いた(以下、KF4009-1、HDF-11および409B2を、WT-iPSCと言う)。
mRNAは、RNeasy Mini Kit (Qiagen)を用いて、各細胞から単離した。得られた全RNAのうち500 ngを鋳型として、ReverTra Ace (TOYOBO)を用いて逆転写によりcDNAを合成した。定量PCR(リアルタイムPCR)のための標準曲線を作成して解析を行った。リアルタイムPCR解析は、KAPA SYBR FAST qPCR kit Master Mix ABI prism (KAPA BIOSYSTEMS)を用いて、Step One system (ABI)により測定した。使用したプライマーの配列およびAssayIDを表2に示す。
細胞溶解液は、SDS-PAGE法を用いて分離し、anti-FGFR3 antibody(Cell SIgnaling社 #4574)、anti-phosphorylated MAPK antibody(Cell Signaling #9109)またはanti β-actin antibody(Cell Signaling社 #49776)を用いて免疫染色した。
軟骨誘導
以前に報告された方法(Oldershaw, R.A., et al., Nat Biotechnol 28, 1187-1194 (2010))を修正した以下に記載した方法に従って、各iPS細胞から軟骨細胞に分化誘導した。
軟骨形成的に分化させた TD-iPSCの妨害された軟骨形成からの回復
TD-iPSCがFGFR3の機能獲得型突然変異が、軟骨細胞分化不全に起因しているかを確認するために、FGFR3をノックダウンする実験を行った。
TD-iPSCの軟骨細胞分化不全を正常化させるための既知薬剤の評価
FGFR3病の治療に有効な薬剤を見出すために、TD-iPSCからの軟骨細胞分化を正常化させるための物質についてスクリーニングを行った。
TD-iPSCの軟骨細胞分化不全を正常化させるための薬剤の探索
スタチンは、広く脂質降下剤として特徴づけられる薬剤クラスを形成する物質であり、メバロン酸合成を阻害し、その結果として、全コレステロール量および低密度リポタンパク質(LDL)レベルを減少させることが知られている。すべてのスタチン類は、心血管系疾患、神経系、免疫系、骨格系および腫瘍成長に対して有利な影響を与えることから、スタチン類の多面的な効果について関心が集まってきている。さらに、ロバスタチンは、髄核から単離した細胞を単層培養することで起きる脱分化によるII型コラーゲンの脱落を抑制することが報告されている(Hu et al., Artif Organs. 35, 411-416 2011)。
ACHにおけるロバスタチンの効果
ロバスタチンがACHに対しても有効であるか否かを調べるため、ACH患者由来のiPS細胞を用いて同様に評価を行った。
スタチンによるACHモデルマウスにおける減少した骨成長の回復
スタチンがin vivoでFGFR3病の表現型を正常化できるか否かを調べた。David Ornitz (Washington University School of Medicine)から入手したFGFR3Achマウス(Naski et al., Development 125, 4977-4988 (1998))を野生型マウスと交雑した(C57BL/6 background)。交雑マウスの出生後3日から14日まで、週に6回1.0 mg/kgロスバスタチン溶液を腹腔内に投与し、15日目に安楽死させ、躯体および骨形成をX線イメージ(Faxitron DX-50)によって評価した。このとき、ゲノムDNAをつま先から抽出し、遺伝子型を調べた。また同様に、交雑マウスの出生後3日から28日まで、週に6回0.4mg/kgロバスタチン溶液を腹腔内に投与し、29日目に安楽死させ、躯体および骨形成を評価した。
Claims (15)
- 有効成分としてHMG-CoA還元酵素阻害薬を含む、FGFR3病の治療および/または予防用医薬。
- 前記HMG-CoA還元酵素阻害薬が、メバスタチン、アトルバスタチン、プラバスタチン、ロスバスタチン、フルバスタチンおよびロバスタチンから成る群より選択される薬剤である、請求項1に記載の医薬。
- 前記FGFR3病が、致死性骨異形成症 (TD) および/または軟骨無形成症 (ACH)である、請求項1または2に記載の医薬。
- FGFR3病を治療および/または予防するための方法であって、HMG-CoA還元酵素阻害薬を投与することを含む、方法。
- 前記HMG-CoA還元酵素阻害薬が、メバスタチン、アトルバスタチン、プラバスタチン、ロスバスタチン、フルバスタチンおよびロバスタチンから成る群より選択される薬剤である、請求項4に記載の方法。
- 前記FGFR3病が、致死性骨異形成症 (TD) および/または軟骨無形成症 (ACH)である、請求項4または5に記載の方法。
- FGFR3病の治療および/または予防用医薬の製造におけるHMG-CoA還元酵素阻害薬の使用。
- 前記HMG-CoA還元酵素阻害薬が、メバスタチン、アトルバスタチン、プラバスタチン、ロスバスタチン、フルバスタチンおよびロバスタチンから成る群より選択される薬剤である、請求項7に記載の使用。
- 前記FGFR3病が、致死性骨異形成症 (TD) および/または軟骨無形成症 (ACH)である、請求項7または8に記載の使用。
- 下記の工程を含む、FGFR3病の治療薬および/または予防薬をスクリーニングする方法;
(a)FGFR3に変異を有する人工多能性幹(iPS)細胞を、被験物質と接触させるおよび接触させない条件下で軟骨細胞へと分化させる工程、
(b)工程(a)で得られた培養物中における軟骨細胞外マトリックスの量、軟骨細胞マーカー遺伝子および線維芽細胞マーカー遺伝子の発現量から成る群より選択される一つ以上の指標を測定する工程、および
(c)被験物質と接触させる条件下において、接触させない条件下より軟骨細胞外マトリックス量または軟骨細胞マーカー遺伝子が増加した場合、もしくは線維芽細胞マーカー遺伝子の発現量が減少した場合、当該被験物質をFGFR3病の治療薬または予防薬として選出する工程。 - 前記工程(a)における軟骨細胞への分化が下記の工程を含む、請求項10に記載の方法:
(i)多能性幹細胞を接着培養することにより中胚葉細胞を誘導する工程、
(ii)前記工程(i)で得られた細胞をbFGF、アスコルビン酸、BMP2、TGFβ、GDF5および被験物質を含む培養液中で接着培養する工程、および
(iii)前記工程(ii)で得られた細胞をアスコルビン酸、BMP2、TGFβ、GDF5および被験物質を含む培養液中で浮遊培養する工程。 - 前記軟骨細胞マーカー遺伝子が、SOX9、AGGRECANおよびCOL2からなる群から選択される少なくとも一つの遺伝子である、請求項10または11に記載の方法。
- 前記線維芽細胞マーカー遺伝子が、COL1A1および/またはCOL1A2である、請求項10または11に記載の方法。
- 前記FGFR3の変異が、FGFR3中のArg248CysまたはGly380Arg変異である、請求項10から13のいずれか1項に記載の方法。
- 前記FGFR3病が、致死性骨異形成症 (TD)および/または軟骨無形成症 (ACH)である、請求項10から14のいずれか1項に記載の方法。
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020167017763A KR102265024B1 (ko) | 2013-12-02 | 2014-11-25 | Fgfr3병의 예방 및 치료제 및 그 스크리닝 방법 |
| EP14867427.8A EP3078387B1 (en) | 2013-12-02 | 2014-11-25 | Prophylactic and therapeutic agents for fgfr3 diseases and screening method for the same |
| EP19158004.2A EP3513789B1 (en) | 2013-12-02 | 2014-11-25 | Prophylactic and therapeutic agents for fgfr3 diseases |
| US15/100,732 US10073083B2 (en) | 2013-12-02 | 2014-11-25 | Prophylactic and therapeutic agents for FGFR3 diseases and screening method for the same |
| US16/059,462 US20180348206A1 (en) | 2013-12-02 | 2018-08-09 | Prophylactic and therapeutic agents for fgfr3 diseases and screening method for the same |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2013-249221 | 2013-12-02 | ||
| JP2013249221 | 2013-12-02 | ||
| JP2014171078A JP6536871B2 (ja) | 2013-12-02 | 2014-08-26 | Fgfr3病の予防および治療剤ならびにそのスクリーニング方法 |
| JP2014-171078 | 2014-08-26 |
Related Child Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US15/100,732 A-371-Of-International US10073083B2 (en) | 2013-12-02 | 2014-11-25 | Prophylactic and therapeutic agents for FGFR3 diseases and screening method for the same |
| US16/059,462 Division US20180348206A1 (en) | 2013-12-02 | 2018-08-09 | Prophylactic and therapeutic agents for fgfr3 diseases and screening method for the same |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2015083582A1 true WO2015083582A1 (ja) | 2015-06-11 |
Family
ID=53273345
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2014/081093 WO2015083582A1 (ja) | 2013-12-02 | 2014-11-25 | Fgfr3病の予防および治療剤ならびにそのスクリーニング方法 |
Country Status (5)
| Country | Link |
|---|---|
| US (2) | US10073083B2 (ja) |
| EP (2) | EP3078387B1 (ja) |
| JP (1) | JP6536871B2 (ja) |
| KR (1) | KR102265024B1 (ja) |
| WO (1) | WO2015083582A1 (ja) |
Cited By (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2016133208A1 (ja) * | 2015-02-19 | 2016-08-25 | 国立大学法人京都大学 | 新規軟骨細胞誘導方法 |
| WO2017170981A1 (ja) * | 2016-04-01 | 2017-10-05 | 国立大学法人京都大学 | Fgfr3病の予防または治療剤 |
| WO2018060314A1 (en) * | 2016-09-29 | 2018-04-05 | Ascendis Pharma Growth Disorders A/S | Combination therapy with controlled-release cnp agonists |
| JP2018517703A (ja) * | 2015-06-18 | 2018-07-05 | 高雄醫學大學 | 軟骨細胞の生成促進薬物を製造するための医薬組成物の用途 |
| JP2020115771A (ja) * | 2019-01-22 | 2020-08-06 | 国立大学法人京都大学 | 多能性幹細胞から軟骨組織を製造する方法 |
| US11311604B2 (en) | 2016-01-08 | 2022-04-26 | Ascendis Pharma Growth Disorders A/S | Controlled-release CNP agonists with low NPR-C binding |
| US11389510B2 (en) | 2016-01-08 | 2022-07-19 | Ascendis Pharma Growth Disorders A/S | Controlled-release CNP agonists with low initial NPR-B activity |
| US11389511B2 (en) | 2016-01-08 | 2022-07-19 | Ascendis Pharma Growth Disorders A/S | Controlled-release CNP agonists with reduced side-effects |
| US11413351B2 (en) | 2016-01-08 | 2022-08-16 | Ascendis Pharma Growth Disorders A/S | CNP prodrugs with carrier attachment at the ring moiety |
| US12083182B2 (en) | 2016-01-08 | 2024-09-10 | Ascendis Pharma Growth Disorders A/S | Controlled-release CNP agonists with increased NEP stability |
Families Citing this family (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2019201584A (ja) * | 2018-05-23 | 2019-11-28 | 国立大学法人神戸大学 | 糖尿病予防又は治療剤スクリーニング方法、及び当該方法に用いる細胞 |
| JP7180458B2 (ja) | 2019-03-07 | 2022-11-30 | マツダ株式会社 | 構造部材およびその製造方法 |
Citations (23)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3983140A (en) | 1974-06-07 | 1976-09-28 | Sankyo Company Limited | Physiologically active substances |
| US4231938A (en) | 1979-06-15 | 1980-11-04 | Merck & Co., Inc. | Hypocholesteremic fermentation products and process of preparation |
| JPS56122375A (en) | 1980-02-04 | 1981-09-25 | Merck & Co Inc | Novel antihypercholesteremic compound* its intermediate and its manufacture |
| JPS572240A (en) | 1980-06-06 | 1982-01-07 | Sankyo Co Ltd | Ml-236b derivative |
| US4346227A (en) | 1980-06-06 | 1982-08-24 | Sankyo Company, Limited | ML-236B Derivatives and their preparation |
| US4444784A (en) | 1980-08-05 | 1984-04-24 | Merck & Co., Inc. | Antihypercholesterolemic compounds |
| JPS60500015A (ja) | 1982-11-22 | 1985-01-10 | サンド・アクチエンゲゼルシヤフト | メバロラクトン同族体とその誘導体、これらの製造法およびこれらを含有する製薬学的組成物 |
| US4739073A (en) | 1983-11-04 | 1988-04-19 | Sandoz Pharmaceuticals Corp. | Intermediates in the synthesis of indole analogs of mevalonolactone and derivatives thereof |
| JPH01279866A (ja) | 1987-08-20 | 1989-11-10 | Nissan Chem Ind Ltd | キノリン系メバロノラクトン類 |
| JPH0358967A (ja) | 1989-07-21 | 1991-03-14 | Warner Lambert Co | 〔R―(R‖,R‖)〕―2―(4―フルオロフエニル)―β,δ―ジヒドロキシ―5―(1―メチルエチル)―3―フエニル―4―〔(フエニルアミノ)カルボニル〕―1H―ピロール―1―ヘプタン酸、そのラクトン体およびその塩 |
| US5223409A (en) | 1988-09-02 | 1993-06-29 | Protein Engineering Corp. | Directed evolution of novel binding proteins |
| JPH05178841A (ja) | 1991-07-01 | 1993-07-20 | Shionogi & Co Ltd | ピリミジン誘導体 |
| US5854259A (en) | 1987-08-20 | 1998-12-29 | Nissan Chemical Industries Ltd. | Quinoline type mevalonolactones |
| US20020103360A1 (en) | 1998-09-01 | 2002-08-01 | Yang Pan | Novel protein related to melanoma-inhibiting protein and uses thereof |
| WO2007069666A1 (ja) | 2005-12-13 | 2007-06-21 | Kyoto University | 核初期化因子 |
| WO2009092042A1 (en) | 2008-01-18 | 2009-07-23 | Nevada Cancer Institute | Reprogramming of differentiated progenitor or somatic cells using homologous recombination |
| JP2009529051A (ja) * | 2006-03-07 | 2009-08-13 | オステオスクリーン アイピー, エルエルシー | HMGCo−Aレダクターゼ阻害剤による骨および軟骨の強化 |
| WO2009115295A1 (en) | 2008-03-17 | 2009-09-24 | Helmholtz Zentrum München - Deutsches Forschungszentrum für Gesundheit und Umwelt (GmbH) | Vectors and methods for generating vector-free induced pluripotent stem (ips) cells using site-specific recombination |
| WO2009149233A1 (en) | 2008-06-04 | 2009-12-10 | Stem Cell Products, Inc. | Methods for the production of ips cells using non-viral approach |
| WO2009152529A2 (en) | 2008-06-13 | 2009-12-17 | Whitehead Institute For Biomedical Research Nine Cambridge Center | Programming and reprogramming of cells |
| WO2009157201A1 (en) | 2008-06-26 | 2009-12-30 | Osaka University | Method and kit for preparing ips cells |
| WO2010012077A1 (en) | 2008-07-28 | 2010-02-04 | Mount Sinai Hospital | Compositions, methods and kits for reprogramming somatic cells |
| WO2010068955A2 (en) | 2008-12-13 | 2010-06-17 | Dna Microarray | MICROENVIRONMENT NICHE ASSAY FOR CiPS SCREENING |
Family Cites Families (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| IL128380A0 (en) | 1999-02-04 | 2000-01-31 | Yeda Res & Dev | A method of screening for agonists and antagonists of FGFR |
| US20040109850A1 (en) | 2000-12-18 | 2004-06-10 | Neelam Jaiswal | Treatment of bone disorders by modulation of fgfr3 |
| EP1964837A4 (en) * | 2005-11-22 | 2010-12-22 | Eisai R&D Man Co Ltd | Antitumor agent against multiple myeloma |
| US8129187B2 (en) | 2005-12-13 | 2012-03-06 | Kyoto University | Somatic cell reprogramming by retroviral vectors encoding Oct3/4. Klf4, c-Myc and Sox2 |
| US20090227032A1 (en) | 2005-12-13 | 2009-09-10 | Kyoto University | Nuclear reprogramming factor and induced pluripotent stem cells |
| US8278104B2 (en) | 2005-12-13 | 2012-10-02 | Kyoto University | Induced pluripotent stem cells produced with Oct3/4, Klf4 and Sox2 |
-
2014
- 2014-08-26 JP JP2014171078A patent/JP6536871B2/ja active Active
- 2014-11-25 EP EP14867427.8A patent/EP3078387B1/en not_active Not-in-force
- 2014-11-25 EP EP19158004.2A patent/EP3513789B1/en active Active
- 2014-11-25 KR KR1020167017763A patent/KR102265024B1/ko active IP Right Grant
- 2014-11-25 US US15/100,732 patent/US10073083B2/en active Active
- 2014-11-25 WO PCT/JP2014/081093 patent/WO2015083582A1/ja active Application Filing
-
2018
- 2018-08-09 US US16/059,462 patent/US20180348206A1/en not_active Abandoned
Patent Citations (29)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3983140A (en) | 1974-06-07 | 1976-09-28 | Sankyo Company Limited | Physiologically active substances |
| US4231938A (en) | 1979-06-15 | 1980-11-04 | Merck & Co., Inc. | Hypocholesteremic fermentation products and process of preparation |
| JPS57163374A (en) | 1979-06-15 | 1982-10-07 | Merck & Co Inc | Hypocholesterolemic fermentation product and manufacture |
| JPS56122375A (en) | 1980-02-04 | 1981-09-25 | Merck & Co Inc | Novel antihypercholesteremic compound* its intermediate and its manufacture |
| JPS572240A (en) | 1980-06-06 | 1982-01-07 | Sankyo Co Ltd | Ml-236b derivative |
| US4346227A (en) | 1980-06-06 | 1982-08-24 | Sankyo Company, Limited | ML-236B Derivatives and their preparation |
| US4444784A (en) | 1980-08-05 | 1984-04-24 | Merck & Co., Inc. | Antihypercholesterolemic compounds |
| JPS60500015A (ja) | 1982-11-22 | 1985-01-10 | サンド・アクチエンゲゼルシヤフト | メバロラクトン同族体とその誘導体、これらの製造法およびこれらを含有する製薬学的組成物 |
| US4739073A (en) | 1983-11-04 | 1988-04-19 | Sandoz Pharmaceuticals Corp. | Intermediates in the synthesis of indole analogs of mevalonolactone and derivatives thereof |
| JPH01279866A (ja) | 1987-08-20 | 1989-11-10 | Nissan Chem Ind Ltd | キノリン系メバロノラクトン類 |
| US5856336A (en) | 1987-08-20 | 1999-01-05 | Nissan Chemical Industries Ltd. | Quinoline type mevalonolactones |
| US5854259A (en) | 1987-08-20 | 1998-12-29 | Nissan Chemical Industries Ltd. | Quinoline type mevalonolactones |
| US5223409A (en) | 1988-09-02 | 1993-06-29 | Protein Engineering Corp. | Directed evolution of novel binding proteins |
| US5571698A (en) | 1988-09-02 | 1996-11-05 | Protein Engineering Corporation | Directed evolution of novel binding proteins |
| US5403484A (en) | 1988-09-02 | 1995-04-04 | Protein Engineering Corporation | Viruses expressing chimeric binding proteins |
| US5273995A (en) | 1989-07-21 | 1993-12-28 | Warner-Lambert Company | [R-(R*R*)]-2-(4-fluorophenyl)-β,δ-dihydroxy-5-(1-methylethyl-3-phenyl-4-[(phenylamino) carbonyl]- 1H-pyrrole-1-heptanoic acid, its lactone form and salts thereof |
| JPH0358967A (ja) | 1989-07-21 | 1991-03-14 | Warner Lambert Co | 〔R―(R‖,R‖)〕―2―(4―フルオロフエニル)―β,δ―ジヒドロキシ―5―(1―メチルエチル)―3―フエニル―4―〔(フエニルアミノ)カルボニル〕―1H―ピロール―1―ヘプタン酸、そのラクトン体およびその塩 |
| US5260440A (en) | 1991-07-01 | 1993-11-09 | Shionogi Seiyaku Kabushiki Kaisha | Pyrimidine derivatives |
| JPH05178841A (ja) | 1991-07-01 | 1993-07-20 | Shionogi & Co Ltd | ピリミジン誘導体 |
| US20020103360A1 (en) | 1998-09-01 | 2002-08-01 | Yang Pan | Novel protein related to melanoma-inhibiting protein and uses thereof |
| WO2007069666A1 (ja) | 2005-12-13 | 2007-06-21 | Kyoto University | 核初期化因子 |
| JP2009529051A (ja) * | 2006-03-07 | 2009-08-13 | オステオスクリーン アイピー, エルエルシー | HMGCo−Aレダクターゼ阻害剤による骨および軟骨の強化 |
| WO2009092042A1 (en) | 2008-01-18 | 2009-07-23 | Nevada Cancer Institute | Reprogramming of differentiated progenitor or somatic cells using homologous recombination |
| WO2009115295A1 (en) | 2008-03-17 | 2009-09-24 | Helmholtz Zentrum München - Deutsches Forschungszentrum für Gesundheit und Umwelt (GmbH) | Vectors and methods for generating vector-free induced pluripotent stem (ips) cells using site-specific recombination |
| WO2009149233A1 (en) | 2008-06-04 | 2009-12-10 | Stem Cell Products, Inc. | Methods for the production of ips cells using non-viral approach |
| WO2009152529A2 (en) | 2008-06-13 | 2009-12-17 | Whitehead Institute For Biomedical Research Nine Cambridge Center | Programming and reprogramming of cells |
| WO2009157201A1 (en) | 2008-06-26 | 2009-12-30 | Osaka University | Method and kit for preparing ips cells |
| WO2010012077A1 (en) | 2008-07-28 | 2010-02-04 | Mount Sinai Hospital | Compositions, methods and kits for reprogramming somatic cells |
| WO2010068955A2 (en) | 2008-12-13 | 2010-06-17 | Dna Microarray | MICROENVIRONMENT NICHE ASSAY FOR CiPS SCREENING |
Non-Patent Citations (66)
| Title |
|---|
| BAI, H. Y. ET AL., JOURNAL OF BIOMEDICAL MATERIALS RESEARCH, vol. 94, 2010, pages 539 - 546 |
| CARELL ET AL., ANGEW. CHEM. INT. ED. ENGL, vol. 33, 1994, pages 2061 |
| CARELL ET AL., ANGEW. CHEM. INT. ED. ENGL., vol. 33, 1994, pages 2059 |
| CELL STEM CELL, vol. 2, 2008, pages 525 - 528 |
| CELL STEM CELL, vol. 3, 2008, pages 132 - 135 |
| CELL STEM CELL, vol. 3, 2008, pages 475 - 479 |
| CELL STEM CELL, vol. 3, 2008, pages 568 - 574 |
| CELL, vol. 126, 2006, pages 663 - 676 |
| CELL, vol. 131, 2007, pages 861 - 872 |
| CHEN, L. ET AL.: "Gly369Cys mutation in mouse FGFR3 causes achondroplasia by affecting both chondrogenesis and osteogenesis", THE JOURNAL OF CLINICAL INVESTIGATION, vol. 104, no. 11, 1999, pages 1517 - 1525, XP002961269 * |
| CHO ET AL., SCIENCE, vol. 261, 1993, pages 1303 - 1305 |
| CULL ET AL., PROC. NATL. ACAD. SCI. USA, vol. 89, 1992, pages 1865 - 1869 |
| CWIRLA ET AL., PROC. NATL. ACAD. SCI. USA, vol. 87, 1990, pages 6378 - 6382 |
| DEVLIN, SCIENCE, vol. 249, 1990, pages 404 - 406 |
| DEWITT ET AL., PROC. NATL. ACAD. SCI. USA, vol. 90, 1993, pages 6909 - 6913 |
| ERB ET AL., PROC. NATL. ACAD. SCI. USA, vol. 91, 1994, pages 11422 - 11426 |
| FELICI, J. MOL. BIOL., vol. 222, 1991, pages 301 - 310 |
| FODOR, NATURE, vol. 364, 1993, pages 555 - 556 |
| GALLOP ET AL., J. MED. CHEM., vol. 37, 1994, pages 1233 - 1251 |
| GOSSET, M ET AL., NATURE PROTOCOLS, vol. 3, 2008, pages 1253 - 1260 |
| HOUGHTEN, BIO/TECHNIQUES, vol. 13, 1992, pages 412 - 421 |
| HU ET AL., ARTIF ORGANS, vol. 35, 2011, pages 411 - 416 |
| HWANG, N. S. ET AL., PLOS ONE, vol. 3, 2008, pages E2498 |
| IKEGAMI, D. ET AL., OSTEOARTHRITIS CARTILAGE, vol. 19, 2011, pages 233 - 241 |
| J. YU ET AL., SCIENCE, vol. 318, 2007, pages 1917 - 1920 |
| JADHAV,S.B. ET AL.: "Statins and osteoporosis: new role for old drugs", J PHARM PHARMACOL, vol. 58, no. 1, 2006, pages 3 - 18, XP055347915 * |
| JONQUOY, A. ET AL., HUM MOL GENET, vol. 21, 2012, pages 841 - 851 |
| K. TAKAHASHI ET AL., CELL, vol. 131, 2007, pages 861 - 872 |
| K. TAKAHASHI; S. YAMANAKA, CELL, vol. 126, 2006, pages 663 - 676 |
| KAJI, K. ET AL., NATURE, vol. 458, 2009, pages 771 - 775 |
| KEIICHI OZONO ET AL.: "Undoki no 10 Nen' Sekai Undo ni Okeru Kin · Kokkakukei Shikkan no Yobo · Chiryo Senryaku Kotsukeito Shikkan Kenkyu no Genjo to Tenbo", THE BONE, vol. 19, no. 4, 2005, pages 51.403 - 54.406, XP008183882 * |
| KOYAMA, N. ET AL., STEM CELLS AND DEVELOPMENT, vol. 22, 2013, pages 102 - 113 |
| KRAMER, J. ET AL.: "Simvastatin modulates mouse embryonic stem cell -derived chondrogenesis in vitro", TOXICOLOGY IN VITRO, vol. 26, no. 7, 2012, pages 1170 - 1176, XP055347917 * |
| LAM, ANTICANCER DRUG DES., vol. 12, 1997, pages 145 - 167 |
| LAM, NATURE, vol. 354, 1991, pages 82 - 84 |
| LEGEAI-MALLET, L. ET AL.: "Overexpression of FGFR3, Stst1, Stat5, and p21Cip1 correlates with phenotypic severity and defective chondrocyte differentiation in FGFR3-related chondrodysplasias", BONE, vol. 34, 2004, pages 26 - 36, XP055347916 * |
| M. NAKAGAWA ET AL., NAT. BIOTECHNOL., vol. 26, 2008, pages 101 - 106 |
| NASKI ET AL., DEVELOPMENT, vol. 125, 1998, pages 4977 - 4988 |
| NAT METHODS, vol. 6, 2009, pages 805 - 808 |
| NAT. BIOTECHNOL., vol. 26, no. 7, 2008, pages 795 - 797 |
| NATURE, vol. 453, 2008, pages 519 - 523 |
| NORIYUKI TSUMAKI: "Chishisei Kotsuikeiseisho no Shindan to Yogo ni Kansuru Kenkyu Chishisei Kotsuikeiseisho Kanja no Hifu Sen'iga Saibo kara no Nankotsu Saibo Yudo no Kokoromi", CHISHISEI KOTSUIKEISEISHO NO SHINDAN TO YOGO NI KANSURU KENKYU HEISEI 23 NENDO SOKATSU · BUNTAN KENKYU HOKOKUSHO, 2012, pages 40 - 42, XP008183793 * |
| OKITA, K. ET AL., NATURE METHODS, vol. 8, 2011, pages 409 - 412 |
| OLDERSHAW, R. A ET AL., NAT. BIOTECHNOL., vol. 28, 2010, pages 1187 - 1194 |
| OLDERSHAW, R. A. ET AL., NAT BIOTECHNOL, vol. 28, 2010, pages 1187 - 1194 |
| OLDERSHAW, R. A. ET AL., NAT. BIOTECHNOL., vol. 28, 2010, pages 1187 - 1194 |
| PLOS BIOLOGY, vol. 6, no. 10, 2008, pages 2237 - 2247 |
| PROC JPN ACAD SER B PHYS BIOL SCI., vol. 85, 2009, pages 348 - 362 |
| R. L. JUDSON ET AL., NAT. BIOTECH., vol. 27, 2009, pages 459 - 461 |
| RAUCHENBERGER, R. ET AL., J BIOL CHEM, vol. 278, 2003, pages 38194 - 38205 |
| SCIENCE, vol. 318, 2007, pages 1917 - 1920 |
| SCIENCE, vol. 322, 2008, pages 945 - 949 |
| SCIENCE, vol. 322, 2008, pages 949 - 953 |
| SCOTT; SMITH, SCIENCE, vol. 249, 1990, pages 386 - 390 |
| See also references of EP3078387A4 |
| SHOHEI KASUGAI: "Kotsutaisha Shikkan no Seiin to Chiryo -Kiso to Rinsho o Tsunagu Mono- Statin no Kyokusho Tekiyo ni yoru Kotsuzosei", CLIN CALCIUM, vol. 15, no. 7, 2005, pages 1165 - 1170, XP008183710 * |
| SIMOPOULOU, T. ET AL.: "Protective Effect of Atorvastatin in Cultured Osteoarthritic Chondrocytes", JOURNAL OF ORTHOPAEDIC RESEARCH, vol. 28, 2010, pages 110 - 115, XP055347918 * |
| TAKAHASHI, K ET AL., CELL, vol. 131, 2007, pages 861 - 872 |
| WARMAN ET AL., AM J MED GENET, vol. 155A, no. 5, 2011, pages 943 - 968 |
| WOLTJEN ET AL., NATURE, vol. 458, 2009, pages 766 - 770 |
| YAMASHITA, A. ET AL., SCIENTIFIC REPORTS, vol. 3, 2013 |
| YAMASHITA, A. ET AL.: "Statin treatment rescues FGFR3 skeletal dysplasia phenotypes", NATURE, vol. 513, no. 7519, 2014, pages 507 - 511, XP055347914 * |
| YASATO KOMATSU ET AL.: "Statin no Nankotsu Bunka eno Sayo", THE BONE, vol. 16, no. 4, 2002, pages 33.359 - 36.362, XP008183881 * |
| YASODA, A. ET AL., ENDOCRINOLOGY, vol. 150, 2009, pages 3138 - 3144 |
| YUKIO AKASAKI ET AL.: "HMG-CoA Kangen Koso Sogaizai (Statin) wa Kotsuzui Kan'yokei Saibo no Nankotsu Tokuiteki Idenshi Hatsugen o Sokushin suru", THE JOURNAL OF THE JAPANESE ORTHOPAEDIC ASSOCIATION, vol. 80, no. 8, 2006, pages S1106, XP008183757 * |
| ZUCKERMANN ET AL., J. MED. CHEM., vol. 37, 1994, pages 2678 - 2685 |
Cited By (17)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2016133208A1 (ja) * | 2015-02-19 | 2016-08-25 | 国立大学法人京都大学 | 新規軟骨細胞誘導方法 |
| US10557121B2 (en) | 2015-02-19 | 2020-02-11 | Kyoto University | Method for chondrogenic induction |
| JP2018517703A (ja) * | 2015-06-18 | 2018-07-05 | 高雄醫學大學 | 軟骨細胞の生成促進薬物を製造するための医薬組成物の用途 |
| US11311604B2 (en) | 2016-01-08 | 2022-04-26 | Ascendis Pharma Growth Disorders A/S | Controlled-release CNP agonists with low NPR-C binding |
| US12083182B2 (en) | 2016-01-08 | 2024-09-10 | Ascendis Pharma Growth Disorders A/S | Controlled-release CNP agonists with increased NEP stability |
| US11413351B2 (en) | 2016-01-08 | 2022-08-16 | Ascendis Pharma Growth Disorders A/S | CNP prodrugs with carrier attachment at the ring moiety |
| US11389511B2 (en) | 2016-01-08 | 2022-07-19 | Ascendis Pharma Growth Disorders A/S | Controlled-release CNP agonists with reduced side-effects |
| US11389510B2 (en) | 2016-01-08 | 2022-07-19 | Ascendis Pharma Growth Disorders A/S | Controlled-release CNP agonists with low initial NPR-B activity |
| JPWO2017170981A1 (ja) * | 2016-04-01 | 2019-02-07 | 国立大学法人京都大学 | Fgfr3病の予防または治療剤 |
| WO2017170981A1 (ja) * | 2016-04-01 | 2017-10-05 | 国立大学法人京都大学 | Fgfr3病の予防または治療剤 |
| IL265592A (en) * | 2016-09-29 | 2019-05-30 | Ascendis Pharma Growth Disorders As | Combined therapy with CNP agonists with controlled release |
| WO2018060314A1 (en) * | 2016-09-29 | 2018-04-05 | Ascendis Pharma Growth Disorders A/S | Combination therapy with controlled-release cnp agonists |
| US11564974B2 (en) | 2016-09-29 | 2023-01-31 | Ascendis Pharma Growth Disorders A/S | Combination therapy with controlled-release CNP agonists |
| AU2017336253B2 (en) * | 2016-09-29 | 2023-02-23 | Ascendis Pharma Growth Disorders A/S | Combination therapy with controlled-release CNP agonists |
| EP4223309A1 (en) * | 2016-09-29 | 2023-08-09 | Ascendis Pharma Growth Disorders A/S | Combination therapy with controlled-release cnp agonists |
| JP2020115771A (ja) * | 2019-01-22 | 2020-08-06 | 国立大学法人京都大学 | 多能性幹細胞から軟骨組織を製造する方法 |
| JP7269620B2 (ja) | 2019-01-22 | 2023-05-09 | 国立大学法人京都大学 | 多能性幹細胞から軟骨組織を製造する方法 |
Also Published As
| Publication number | Publication date |
|---|---|
| US10073083B2 (en) | 2018-09-11 |
| EP3513789A3 (en) | 2019-09-25 |
| US20160327544A1 (en) | 2016-11-10 |
| EP3078387B1 (en) | 2019-02-27 |
| US20180348206A1 (en) | 2018-12-06 |
| EP3078387A1 (en) | 2016-10-12 |
| EP3513789B1 (en) | 2020-11-25 |
| EP3513789A2 (en) | 2019-07-24 |
| JP6536871B2 (ja) | 2019-07-03 |
| EP3078387A4 (en) | 2017-11-15 |
| JP2015129110A (ja) | 2015-07-16 |
| KR102265024B1 (ko) | 2021-06-14 |
| KR20160085910A (ko) | 2016-07-18 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6536871B2 (ja) | Fgfr3病の予防および治療剤ならびにそのスクリーニング方法 | |
| JP5936134B2 (ja) | ヒト人工多能性幹細胞の選択方法 | |
| JP2022188229A (ja) | 中間中胚葉細胞から腎前駆細胞への分化誘導方法、および多能性幹細胞から腎前駆細胞への分化誘導方法 | |
| JP6949336B2 (ja) | 人工多能性幹細胞由来腸管幹細胞の維持培養 | |
| JP6153232B2 (ja) | 筋萎縮性側索硬化症の予防および治療薬とそのスクリーニング方法 | |
| JP2011121949A (ja) | 筋萎縮性側索硬化症の予防および治療用医薬組成物 | |
| JP6673586B2 (ja) | 異所性骨化の予防・治療剤及びそのスクリーニング方法 | |
| JP5995086B2 (ja) | 心筋症特異的多能性幹細胞およびその用途 | |
| JP6856266B2 (ja) | Fgfr3病の予防および治療剤ならびにそのスクリーニング方法 | |
| JP6598185B2 (ja) | 軟骨過形成疾患の予防および治療剤ならびにそのスクリーニング方法 | |
| JP6335117B2 (ja) | アルツハイマー病の治療薬および/または予防薬のスクリーニング方法 | |
| EP3760728B1 (en) | Evaluation of cells using directional movement ability. | |
| JP6083874B2 (ja) | ミトコンドリア病特異的人工多能性幹細胞、その製造方法及び使用 | |
| WO2021193408A1 (ja) | 先天性尿素サイクル異常症モデルおよびその利用 | |
| WO2021015245A1 (ja) | 繊毛関連疾患モデルおよびその利用 | |
| JP2023071631A (ja) | Alport症候群の予防又は治療薬のスクリーニング又は評価方法 | |
| WO2014189144A1 (ja) | 骨髄異形成症候群等の治療/予防薬のスクリーニング方法 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 14867427 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| REEP | Request for entry into the european phase |
Ref document number: 2014867427 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2014867427 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 20167017763 Country of ref document: KR Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 15100732 Country of ref document: US |