WO2014157156A1 - タンパク質組成物およびその製造方法 - Google Patents
タンパク質組成物およびその製造方法 Download PDFInfo
- Publication number
- WO2014157156A1 WO2014157156A1 PCT/JP2014/058209 JP2014058209W WO2014157156A1 WO 2014157156 A1 WO2014157156 A1 WO 2014157156A1 JP 2014058209 W JP2014058209 W JP 2014058209W WO 2014157156 A1 WO2014157156 A1 WO 2014157156A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- milk basic
- protein fraction
- basic protein
- milk
- protein
- Prior art date
Links
- 102000004169 proteins and genes Human genes 0.000 title claims abstract description 132
- 108090000623 proteins and genes Proteins 0.000 title claims abstract description 132
- 239000000203 mixture Substances 0.000 title claims abstract description 131
- 238000004519 manufacturing process Methods 0.000 title description 5
- 235000013336 milk Nutrition 0.000 claims abstract description 167
- 239000008267 milk Substances 0.000 claims abstract description 167
- 210000004080 milk Anatomy 0.000 claims abstract description 167
- 101710093543 Probable non-specific lipid-transfer protein Proteins 0.000 claims abstract description 165
- 239000003381 stabilizer Substances 0.000 claims abstract description 56
- 150000004676 glycans Chemical class 0.000 claims abstract description 19
- 229920001285 xanthan gum Polymers 0.000 claims abstract description 17
- 235000010493 xanthan gum Nutrition 0.000 claims abstract description 17
- 239000000230 xanthan gum Substances 0.000 claims abstract description 17
- 229940082509 xanthan gum Drugs 0.000 claims abstract description 17
- 235000010469 Glycine max Nutrition 0.000 claims abstract description 16
- 229920001282 polysaccharide Polymers 0.000 claims abstract description 16
- 239000005017 polysaccharide Substances 0.000 claims abstract description 16
- 244000068988 Glycine max Species 0.000 claims abstract description 14
- 235000013305 food Nutrition 0.000 claims abstract description 14
- 239000003814 drug Substances 0.000 claims abstract description 13
- 229920001277 pectin Polymers 0.000 claims abstract description 13
- 239000001814 pectin Substances 0.000 claims abstract description 13
- 235000010987 pectin Nutrition 0.000 claims abstract description 13
- 244000215068 Acacia senegal Species 0.000 claims abstract description 10
- 102000011632 Caseins Human genes 0.000 claims abstract description 10
- 108010076119 Caseins Proteins 0.000 claims abstract description 10
- 229920000084 Gum arabic Polymers 0.000 claims abstract description 10
- 235000010489 acacia gum Nutrition 0.000 claims abstract description 10
- 239000000205 acacia gum Substances 0.000 claims abstract description 10
- IIZPXYDJLKNOIY-JXPKJXOSSA-N 1-palmitoyl-2-arachidonoyl-sn-glycero-3-phosphocholine Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCC\C=C/C\C=C/C\C=C/C\C=C/CCCCC IIZPXYDJLKNOIY-JXPKJXOSSA-N 0.000 claims abstract description 9
- 229920002134 Carboxymethyl cellulose Polymers 0.000 claims abstract description 9
- 229920000161 Locust bean gum Polymers 0.000 claims abstract description 9
- 239000001768 carboxy methyl cellulose Substances 0.000 claims abstract description 9
- 235000010948 carboxy methyl cellulose Nutrition 0.000 claims abstract description 9
- 239000008112 carboxymethyl-cellulose Substances 0.000 claims abstract description 9
- 235000010418 carrageenan Nutrition 0.000 claims abstract description 9
- 239000000679 carrageenan Substances 0.000 claims abstract description 9
- 229920001525 carrageenan Polymers 0.000 claims abstract description 9
- 229940113118 carrageenan Drugs 0.000 claims abstract description 9
- 239000000787 lecithin Substances 0.000 claims abstract description 9
- 235000010445 lecithin Nutrition 0.000 claims abstract description 9
- 229940067606 lecithin Drugs 0.000 claims abstract description 9
- 235000010420 locust bean gum Nutrition 0.000 claims abstract description 9
- 239000000711 locust bean gum Substances 0.000 claims abstract description 9
- UHVMMEOXYDMDKI-JKYCWFKZSA-L zinc;1-(5-cyanopyridin-2-yl)-3-[(1s,2s)-2-(6-fluoro-2-hydroxy-3-propanoylphenyl)cyclopropyl]urea;diacetate Chemical compound [Zn+2].CC([O-])=O.CC([O-])=O.CCC(=O)C1=CC=C(F)C([C@H]2[C@H](C2)NC(=O)NC=2N=CC(=CC=2)C#N)=C1O UHVMMEOXYDMDKI-JKYCWFKZSA-L 0.000 claims abstract description 9
- 229940080237 sodium caseinate Drugs 0.000 claims abstract description 7
- 229940105329 carboxymethylcellulose Drugs 0.000 claims abstract description 6
- 235000018102 proteins Nutrition 0.000 claims description 127
- 238000010438 heat treatment Methods 0.000 claims description 58
- 238000002415 sodium dodecyl sulfate polyacrylamide gel electrophoresis Methods 0.000 claims description 47
- 150000001413 amino acids Chemical class 0.000 claims description 23
- 238000000034 method Methods 0.000 claims description 19
- 229920000591 gum Polymers 0.000 claims description 11
- 102000010445 Lactoferrin Human genes 0.000 claims description 6
- 108010063045 Lactoferrin Proteins 0.000 claims description 6
- 108010023244 Lactoperoxidase Proteins 0.000 claims description 6
- 102000045576 Lactoperoxidases Human genes 0.000 claims description 6
- 229940079593 drug Drugs 0.000 claims description 6
- CSSYQJWUGATIHM-IKGCZBKSSA-N l-phenylalanyl-l-lysyl-l-cysteinyl-l-arginyl-l-arginyl-l-tryptophyl-l-glutaminyl-l-tryptophyl-l-arginyl-l-methionyl-l-lysyl-l-lysyl-l-leucylglycyl-l-alanyl-l-prolyl-l-seryl-l-isoleucyl-l-threonyl-l-cysteinyl-l-valyl-l-arginyl-l-arginyl-l-alanyl-l-phenylal Chemical compound C([C@H](N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CS)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(C)C)C(=O)NCC(=O)N[C@@H](C)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CO)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CS)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](C)C(=O)N[C@@H](CC=1C=CC=CC=1)C(O)=O)C1=CC=CC=C1 CSSYQJWUGATIHM-IKGCZBKSSA-N 0.000 claims description 6
- 229940078795 lactoferrin Drugs 0.000 claims description 6
- 235000021242 lactoferrin Nutrition 0.000 claims description 6
- 229940057428 lactoperoxidase Drugs 0.000 claims description 6
- 235000013361 beverage Nutrition 0.000 claims description 4
- 230000006872 improvement Effects 0.000 abstract description 2
- 244000106483 Anogeissus latifolia Species 0.000 abstract 1
- 235000011514 Anogeissus latifolia Nutrition 0.000 abstract 1
- 239000001922 Gum ghatti Substances 0.000 abstract 1
- 230000009849 deactivation Effects 0.000 abstract 1
- 235000019314 gum ghatti Nutrition 0.000 abstract 1
- 239000000243 solution Substances 0.000 description 91
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Chemical compound O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 39
- 239000008367 deionised water Substances 0.000 description 37
- 229910021641 deionized water Inorganic materials 0.000 description 37
- 239000007788 liquid Substances 0.000 description 37
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 36
- 238000001556 precipitation Methods 0.000 description 36
- 238000004220 aggregation Methods 0.000 description 34
- 230000002776 aggregation Effects 0.000 description 34
- 239000000523 sample Substances 0.000 description 34
- JVTAAEKCZFNVCJ-UHFFFAOYSA-N lactic acid Chemical compound CC(O)C(O)=O JVTAAEKCZFNVCJ-UHFFFAOYSA-N 0.000 description 22
- 230000001954 sterilising effect Effects 0.000 description 22
- 238000004659 sterilization and disinfection Methods 0.000 description 21
- 239000000047 product Substances 0.000 description 20
- 230000000694 effects Effects 0.000 description 16
- 230000002378 acidificating effect Effects 0.000 description 13
- 230000007935 neutral effect Effects 0.000 description 13
- 238000002264 polyacrylamide gel electrophoresis Methods 0.000 description 12
- 235000011121 sodium hydroxide Nutrition 0.000 description 12
- 230000015556 catabolic process Effects 0.000 description 11
- 238000006731 degradation reaction Methods 0.000 description 11
- 239000004310 lactic acid Substances 0.000 description 11
- 235000014655 lactic acid Nutrition 0.000 description 11
- 239000003708 ampul Substances 0.000 description 10
- 238000004458 analytical method Methods 0.000 description 10
- 230000000007 visual effect Effects 0.000 description 10
- 235000020183 skimmed milk Nutrition 0.000 description 9
- 230000009471 action Effects 0.000 description 7
- 239000000843 powder Substances 0.000 description 7
- 239000002244 precipitate Substances 0.000 description 6
- 239000000796 flavoring agent Substances 0.000 description 5
- 235000019634 flavors Nutrition 0.000 description 5
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 4
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 4
- 229930006000 Sucrose Natural products 0.000 description 4
- 102000007544 Whey Proteins Human genes 0.000 description 4
- 108010046377 Whey Proteins Proteins 0.000 description 4
- 235000014113 dietary fatty acids Nutrition 0.000 description 4
- -1 ester compound Chemical class 0.000 description 4
- 239000000194 fatty acid Substances 0.000 description 4
- 229930195729 fatty acid Natural products 0.000 description 4
- 239000012528 membrane Substances 0.000 description 4
- 239000005720 sucrose Substances 0.000 description 4
- NWUYHJFMYQTDRP-UHFFFAOYSA-N 1,2-bis(ethenyl)benzene;1-ethenyl-2-ethylbenzene;styrene Chemical compound C=CC1=CC=CC=C1.CCC1=CC=CC=C1C=C.C=CC1=CC=CC=C1C=C NWUYHJFMYQTDRP-UHFFFAOYSA-N 0.000 description 3
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 3
- IAJILQKETJEXLJ-UHFFFAOYSA-N Galacturonsaeure Natural products O=CC(O)C(O)C(O)C(O)C(O)=O IAJILQKETJEXLJ-UHFFFAOYSA-N 0.000 description 3
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 3
- 240000004584 Tamarindus indica Species 0.000 description 3
- 235000004298 Tamarindus indica Nutrition 0.000 description 3
- 239000005862 Whey Substances 0.000 description 3
- PYMYPHUHKUWMLA-UHFFFAOYSA-N arabinose Natural products OCC(O)C(O)C(O)C=O PYMYPHUHKUWMLA-UHFFFAOYSA-N 0.000 description 3
- SRBFZHDQGSBBOR-UHFFFAOYSA-N beta-D-Pyranose-Lyxose Natural products OC1COC(O)C(O)C1O SRBFZHDQGSBBOR-UHFFFAOYSA-N 0.000 description 3
- 210000000988 bone and bone Anatomy 0.000 description 3
- 239000005018 casein Substances 0.000 description 3
- BECPQYXYKAMYBN-UHFFFAOYSA-N casein, tech. Chemical compound NCCCCC(C(O)=O)N=C(O)C(CC(O)=O)N=C(O)C(CCC(O)=N)N=C(O)C(CC(C)C)N=C(O)C(CCC(O)=O)N=C(O)C(CC(O)=O)N=C(O)C(CCC(O)=O)N=C(O)C(C(C)O)N=C(O)C(CCC(O)=N)N=C(O)C(CCC(O)=N)N=C(O)C(CCC(O)=N)N=C(O)C(CCC(O)=O)N=C(O)C(CCC(O)=O)N=C(O)C(COP(O)(O)=O)N=C(O)C(CCC(O)=N)N=C(O)C(N)CC1=CC=CC=C1 BECPQYXYKAMYBN-UHFFFAOYSA-N 0.000 description 3
- 235000021240 caseins Nutrition 0.000 description 3
- 239000003729 cation exchange resin Substances 0.000 description 3
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 description 3
- 238000010828 elution Methods 0.000 description 3
- 230000000415 inactivating effect Effects 0.000 description 3
- 210000000963 osteoblast Anatomy 0.000 description 3
- 230000035755 proliferation Effects 0.000 description 3
- 230000009257 reactivity Effects 0.000 description 3
- 239000011347 resin Substances 0.000 description 3
- 229920005989 resin Polymers 0.000 description 3
- 239000011734 sodium Substances 0.000 description 3
- 229910052708 sodium Inorganic materials 0.000 description 3
- 238000003756 stirring Methods 0.000 description 3
- 238000005728 strengthening Methods 0.000 description 3
- SRBFZHDQGSBBOR-IOVATXLUSA-N D-xylopyranose Chemical compound O[C@@H]1COC(O)[C@H](O)[C@H]1O SRBFZHDQGSBBOR-IOVATXLUSA-N 0.000 description 2
- 238000002965 ELISA Methods 0.000 description 2
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 2
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 2
- UIIMBOGNXHQVGW-UHFFFAOYSA-M Sodium bicarbonate Chemical compound [Na+].OC([O-])=O UIIMBOGNXHQVGW-UHFFFAOYSA-M 0.000 description 2
- 230000002159 abnormal effect Effects 0.000 description 2
- IAJILQKETJEXLJ-QTBDOELSSA-N aldehydo-D-glucuronic acid Chemical compound O=C[C@H](O)[C@@H](O)[C@H](O)[C@H](O)C(O)=O IAJILQKETJEXLJ-QTBDOELSSA-N 0.000 description 2
- PYMYPHUHKUWMLA-WDCZJNDASA-N arabinose Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)C=O PYMYPHUHKUWMLA-WDCZJNDASA-N 0.000 description 2
- 150000001768 cations Chemical class 0.000 description 2
- 239000012141 concentrate Substances 0.000 description 2
- 235000008504 concentrate Nutrition 0.000 description 2
- 238000000909 electrodialysis Methods 0.000 description 2
- 239000003995 emulsifying agent Substances 0.000 description 2
- 239000003205 fragrance Substances 0.000 description 2
- 229930182830 galactose Natural products 0.000 description 2
- 229940097043 glucuronic acid Drugs 0.000 description 2
- 230000002779 inactivation Effects 0.000 description 2
- 229910052500 inorganic mineral Inorganic materials 0.000 description 2
- 235000010755 mineral Nutrition 0.000 description 2
- 239000011707 mineral Substances 0.000 description 2
- 238000002156 mixing Methods 0.000 description 2
- 230000035790 physiological processes and functions Effects 0.000 description 2
- 238000002360 preparation method Methods 0.000 description 2
- 239000002994 raw material Substances 0.000 description 2
- 238000001223 reverse osmosis Methods 0.000 description 2
- 239000011780 sodium chloride Substances 0.000 description 2
- 230000006641 stabilisation Effects 0.000 description 2
- 238000011105 stabilization Methods 0.000 description 2
- 238000003860 storage Methods 0.000 description 2
- 239000011782 vitamin Substances 0.000 description 2
- 229940088594 vitamin Drugs 0.000 description 2
- 235000013343 vitamin Nutrition 0.000 description 2
- 229930003231 vitamin Natural products 0.000 description 2
- 150000003722 vitamin derivatives Chemical class 0.000 description 2
- DGVVWUTYPXICAM-UHFFFAOYSA-N β‐Mercaptoethanol Chemical compound OCCS DGVVWUTYPXICAM-UHFFFAOYSA-N 0.000 description 2
- QKNYBSVHEMOAJP-UHFFFAOYSA-N 2-amino-2-(hydroxymethyl)propane-1,3-diol;hydron;chloride Chemical compound Cl.OCC(N)(CO)CO QKNYBSVHEMOAJP-UHFFFAOYSA-N 0.000 description 1
- FHVDTGUDJYJELY-UHFFFAOYSA-N 6-{[2-carboxy-4,5-dihydroxy-6-(phosphanyloxy)oxan-3-yl]oxy}-4,5-dihydroxy-3-phosphanyloxane-2-carboxylic acid Chemical compound O1C(C(O)=O)C(P)C(O)C(O)C1OC1C(C(O)=O)OC(OP)C(O)C1O FHVDTGUDJYJELY-UHFFFAOYSA-N 0.000 description 1
- 239000004475 Arginine Substances 0.000 description 1
- 241000283690 Bos taurus Species 0.000 description 1
- 241000283707 Capra Species 0.000 description 1
- BVKZGUZCCUSVTD-UHFFFAOYSA-L Carbonate Chemical compound [O-]C([O-])=O BVKZGUZCCUSVTD-UHFFFAOYSA-L 0.000 description 1
- 229920001661 Chitosan Polymers 0.000 description 1
- 229920002261 Corn starch Polymers 0.000 description 1
- FBPFZTCFMRRESA-FSIIMWSLSA-N D-Glucitol Natural products OC[C@H](O)[C@H](O)[C@@H](O)[C@H](O)CO FBPFZTCFMRRESA-FSIIMWSLSA-N 0.000 description 1
- WQZGKKKJIJFFOK-QTVWNMPRSA-N D-mannopyranose Chemical compound OC[C@H]1OC(O)[C@@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-QTVWNMPRSA-N 0.000 description 1
- 229920001353 Dextrin Polymers 0.000 description 1
- 239000004375 Dextrin Substances 0.000 description 1
- 241000283086 Equidae Species 0.000 description 1
- 229920002148 Gellan gum Polymers 0.000 description 1
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 1
- 229920002907 Guar gum Polymers 0.000 description 1
- 241000282412 Homo Species 0.000 description 1
- ODKSFYDXXFIFQN-BYPYZUCNSA-P L-argininium(2+) Chemical compound NC(=[NH2+])NCCC[C@H]([NH3+])C(O)=O ODKSFYDXXFIFQN-BYPYZUCNSA-P 0.000 description 1
- HNDVDQJCIGZPNO-YFKPBYRVSA-N L-histidine Chemical compound OC(=O)[C@@H](N)CC1=CN=CN1 HNDVDQJCIGZPNO-YFKPBYRVSA-N 0.000 description 1
- KDXKERNSBIXSRK-YFKPBYRVSA-N L-lysine Chemical compound NCCCC[C@H](N)C(O)=O KDXKERNSBIXSRK-YFKPBYRVSA-N 0.000 description 1
- KDXKERNSBIXSRK-UHFFFAOYSA-N Lysine Natural products NCCCCC(N)C(O)=O KDXKERNSBIXSRK-UHFFFAOYSA-N 0.000 description 1
- 239000004472 Lysine Substances 0.000 description 1
- 235000019482 Palm oil Nutrition 0.000 description 1
- 241001494479 Pecora Species 0.000 description 1
- 235000004347 Perilla Nutrition 0.000 description 1
- 244000124853 Perilla frutescens Species 0.000 description 1
- 235000019764 Soybean Meal Nutrition 0.000 description 1
- 108010073771 Soybean Proteins Proteins 0.000 description 1
- 229920002472 Starch Polymers 0.000 description 1
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 1
- 241000282887 Suidae Species 0.000 description 1
- 235000021307 Triticum Nutrition 0.000 description 1
- 244000098338 Triticum aestivum Species 0.000 description 1
- IAJILQKETJEXLJ-RSJOWCBRSA-N aldehydo-D-galacturonic acid Chemical compound O=C[C@H](O)[C@@H](O)[C@@H](O)[C@H](O)C(O)=O IAJILQKETJEXLJ-RSJOWCBRSA-N 0.000 description 1
- 229940072056 alginate Drugs 0.000 description 1
- 235000010443 alginic acid Nutrition 0.000 description 1
- 229920000615 alginic acid Polymers 0.000 description 1
- WQZGKKKJIJFFOK-PHYPRBDBSA-N alpha-D-galactose Chemical compound OC[C@H]1O[C@H](O)[C@H](O)[C@@H](O)[C@H]1O WQZGKKKJIJFFOK-PHYPRBDBSA-N 0.000 description 1
- 229910000147 aluminium phosphate Inorganic materials 0.000 description 1
- ODKSFYDXXFIFQN-UHFFFAOYSA-N arginine Natural products OC(=O)C(N)CCCNC(N)=N ODKSFYDXXFIFQN-UHFFFAOYSA-N 0.000 description 1
- 230000036772 blood pressure Effects 0.000 description 1
- 238000009395 breeding Methods 0.000 description 1
- 230000001488 breeding effect Effects 0.000 description 1
- 239000000872 buffer Substances 0.000 description 1
- MKJXYGKVIBWPFZ-UHFFFAOYSA-L calcium lactate Chemical compound [Ca+2].CC(O)C([O-])=O.CC(O)C([O-])=O MKJXYGKVIBWPFZ-UHFFFAOYSA-L 0.000 description 1
- 239000001527 calcium lactate Substances 0.000 description 1
- 235000011086 calcium lactate Nutrition 0.000 description 1
- 229960002401 calcium lactate Drugs 0.000 description 1
- 229920002678 cellulose Polymers 0.000 description 1
- 239000001913 cellulose Substances 0.000 description 1
- 235000013351 cheese Nutrition 0.000 description 1
- 239000003795 chemical substances by application Substances 0.000 description 1
- 239000007979 citrate buffer Substances 0.000 description 1
- 239000011248 coating agent Substances 0.000 description 1
- 238000000576 coating method Methods 0.000 description 1
- 230000037319 collagen production Effects 0.000 description 1
- 239000002285 corn oil Substances 0.000 description 1
- 235000005687 corn oil Nutrition 0.000 description 1
- 239000008120 corn starch Substances 0.000 description 1
- 238000000354 decomposition reaction Methods 0.000 description 1
- 238000004925 denaturation Methods 0.000 description 1
- 230000036425 denaturation Effects 0.000 description 1
- 235000019425 dextrin Nutrition 0.000 description 1
- 230000006806 disease prevention Effects 0.000 description 1
- 150000004665 fatty acids Chemical class 0.000 description 1
- 238000011049 filling Methods 0.000 description 1
- 235000021323 fish oil Nutrition 0.000 description 1
- 235000013312 flour Nutrition 0.000 description 1
- 239000000499 gel Substances 0.000 description 1
- 235000010492 gellan gum Nutrition 0.000 description 1
- 239000000216 gellan gum Substances 0.000 description 1
- 239000011521 glass Substances 0.000 description 1
- 239000008103 glucose Substances 0.000 description 1
- 125000002791 glucosyl group Chemical group C1([C@H](O)[C@@H](O)[C@H](O)[C@H](O1)CO)* 0.000 description 1
- 239000008187 granular material Substances 0.000 description 1
- 235000010417 guar gum Nutrition 0.000 description 1
- 239000000665 guar gum Substances 0.000 description 1
- 229960002154 guar gum Drugs 0.000 description 1
- HNDVDQJCIGZPNO-UHFFFAOYSA-N histidine Natural products OC(=O)C(N)CC1=CN=CN1 HNDVDQJCIGZPNO-UHFFFAOYSA-N 0.000 description 1
- 230000007062 hydrolysis Effects 0.000 description 1
- 238000006460 hydrolysis reaction Methods 0.000 description 1
- 125000001165 hydrophobic group Chemical group 0.000 description 1
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 1
- 210000000987 immune system Anatomy 0.000 description 1
- 230000037356 lipid metabolism Effects 0.000 description 1
- 150000002632 lipids Chemical class 0.000 description 1
- 239000000845 maltitol Substances 0.000 description 1
- 235000010449 maltitol Nutrition 0.000 description 1
- VQHSOMBJVWLPSR-WUJBLJFYSA-N maltitol Chemical compound OC[C@H](O)[C@@H](O)[C@@H]([C@H](O)CO)O[C@H]1O[C@H](CO)[C@@H](O)[C@H](O)[C@H]1O VQHSOMBJVWLPSR-WUJBLJFYSA-N 0.000 description 1
- 229940035436 maltitol Drugs 0.000 description 1
- 239000003550 marker Substances 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- 150000007522 mineralic acids Chemical class 0.000 description 1
- 235000015097 nutrients Nutrition 0.000 description 1
- 239000003921 oil Substances 0.000 description 1
- 235000019198 oils Nutrition 0.000 description 1
- 150000007524 organic acids Chemical class 0.000 description 1
- 239000002540 palm oil Substances 0.000 description 1
- 239000002245 particle Substances 0.000 description 1
- 208000028169 periodontal disease Diseases 0.000 description 1
- 229920002401 polyacrylamide Polymers 0.000 description 1
- 150000003839 salts Chemical class 0.000 description 1
- 239000012723 sample buffer Substances 0.000 description 1
- 238000007789 sealing Methods 0.000 description 1
- 235000015424 sodium Nutrition 0.000 description 1
- 235000017557 sodium bicarbonate Nutrition 0.000 description 1
- 229910000030 sodium bicarbonate Inorganic materials 0.000 description 1
- 239000000600 sorbitol Substances 0.000 description 1
- 229940001941 soy protein Drugs 0.000 description 1
- 239000004455 soybean meal Substances 0.000 description 1
- 239000003549 soybean oil Substances 0.000 description 1
- 235000012424 soybean oil Nutrition 0.000 description 1
- 239000008107 starch Substances 0.000 description 1
- 235000019698 starch Nutrition 0.000 description 1
- 235000000346 sugar Nutrition 0.000 description 1
- 150000008163 sugars Chemical class 0.000 description 1
- 239000006188 syrup Substances 0.000 description 1
- 235000020357 syrup Nutrition 0.000 description 1
- 238000005406 washing Methods 0.000 description 1
- 235000021119 whey protein Nutrition 0.000 description 1
Classifications
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS, OR NON-ALCOHOLIC BEVERAGES, NOT COVERED BY SUBCLASSES A21D OR A23B-A23J; THEIR PREPARATION OR TREATMENT, e.g. COOKING, MODIFICATION OF NUTRITIVE QUALITIES, PHYSICAL TREATMENT; PRESERVATION OF FOODS OR FOODSTUFFS, IN GENERAL
- A23L33/00—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof
- A23L33/10—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof using additives
- A23L33/17—Amino acids, peptides or proteins
- A23L33/19—Dairy proteins
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23J—PROTEIN COMPOSITIONS FOR FOODSTUFFS; WORKING-UP PROTEINS FOR FOODSTUFFS; PHOSPHATIDE COMPOSITIONS FOR FOODSTUFFS
- A23J1/00—Obtaining protein compositions for foodstuffs; Bulk opening of eggs and separation of yolks from whites
- A23J1/20—Obtaining protein compositions for foodstuffs; Bulk opening of eggs and separation of yolks from whites from milk, e.g. casein; from whey
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23K—FODDER
- A23K20/00—Accessory food factors for animal feeding-stuffs
- A23K20/10—Organic substances
- A23K20/142—Amino acids; Derivatives thereof
- A23K20/147—Polymeric derivatives, e.g. peptides or proteins
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS, OR NON-ALCOHOLIC BEVERAGES, NOT COVERED BY SUBCLASSES A21D OR A23B-A23J; THEIR PREPARATION OR TREATMENT, e.g. COOKING, MODIFICATION OF NUTRITIVE QUALITIES, PHYSICAL TREATMENT; PRESERVATION OF FOODS OR FOODSTUFFS, IN GENERAL
- A23L2/00—Non-alcoholic beverages; Dry compositions or concentrates therefor; Their preparation
- A23L2/52—Adding ingredients
- A23L2/66—Proteins
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS, OR NON-ALCOHOLIC BEVERAGES, NOT COVERED BY SUBCLASSES A21D OR A23B-A23J; THEIR PREPARATION OR TREATMENT, e.g. COOKING, MODIFICATION OF NUTRITIVE QUALITIES, PHYSICAL TREATMENT; PRESERVATION OF FOODS OR FOODSTUFFS, IN GENERAL
- A23L29/00—Foods or foodstuffs containing additives; Preparation or treatment thereof
- A23L29/10—Foods or foodstuffs containing additives; Preparation or treatment thereof containing emulsifiers
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS, OR NON-ALCOHOLIC BEVERAGES, NOT COVERED BY SUBCLASSES A21D OR A23B-A23J; THEIR PREPARATION OR TREATMENT, e.g. COOKING, MODIFICATION OF NUTRITIVE QUALITIES, PHYSICAL TREATMENT; PRESERVATION OF FOODS OR FOODSTUFFS, IN GENERAL
- A23L29/00—Foods or foodstuffs containing additives; Preparation or treatment thereof
- A23L29/20—Foods or foodstuffs containing additives; Preparation or treatment thereof containing gelling or thickening agents
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS, OR NON-ALCOHOLIC BEVERAGES, NOT COVERED BY SUBCLASSES A21D OR A23B-A23J; THEIR PREPARATION OR TREATMENT, e.g. COOKING, MODIFICATION OF NUTRITIVE QUALITIES, PHYSICAL TREATMENT; PRESERVATION OF FOODS OR FOODSTUFFS, IN GENERAL
- A23L29/00—Foods or foodstuffs containing additives; Preparation or treatment thereof
- A23L29/20—Foods or foodstuffs containing additives; Preparation or treatment thereof containing gelling or thickening agents
- A23L29/206—Foods or foodstuffs containing additives; Preparation or treatment thereof containing gelling or thickening agents of vegetable origin
- A23L29/231—Pectin; Derivatives thereof
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS, OR NON-ALCOHOLIC BEVERAGES, NOT COVERED BY SUBCLASSES A21D OR A23B-A23J; THEIR PREPARATION OR TREATMENT, e.g. COOKING, MODIFICATION OF NUTRITIVE QUALITIES, PHYSICAL TREATMENT; PRESERVATION OF FOODS OR FOODSTUFFS, IN GENERAL
- A23L29/00—Foods or foodstuffs containing additives; Preparation or treatment thereof
- A23L29/20—Foods or foodstuffs containing additives; Preparation or treatment thereof containing gelling or thickening agents
- A23L29/206—Foods or foodstuffs containing additives; Preparation or treatment thereof containing gelling or thickening agents of vegetable origin
- A23L29/262—Cellulose; Derivatives thereof, e.g. ethers
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS, OR NON-ALCOHOLIC BEVERAGES, NOT COVERED BY SUBCLASSES A21D OR A23B-A23J; THEIR PREPARATION OR TREATMENT, e.g. COOKING, MODIFICATION OF NUTRITIVE QUALITIES, PHYSICAL TREATMENT; PRESERVATION OF FOODS OR FOODSTUFFS, IN GENERAL
- A23L29/00—Foods or foodstuffs containing additives; Preparation or treatment thereof
- A23L29/20—Foods or foodstuffs containing additives; Preparation or treatment thereof containing gelling or thickening agents
- A23L29/269—Foods or foodstuffs containing additives; Preparation or treatment thereof containing gelling or thickening agents of microbial origin, e.g. xanthan or dextran
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS, OR NON-ALCOHOLIC BEVERAGES, NOT COVERED BY SUBCLASSES A21D OR A23B-A23J; THEIR PREPARATION OR TREATMENT, e.g. COOKING, MODIFICATION OF NUTRITIVE QUALITIES, PHYSICAL TREATMENT; PRESERVATION OF FOODS OR FOODSTUFFS, IN GENERAL
- A23L29/00—Foods or foodstuffs containing additives; Preparation or treatment thereof
- A23L29/20—Foods or foodstuffs containing additives; Preparation or treatment thereof containing gelling or thickening agents
- A23L29/269—Foods or foodstuffs containing additives; Preparation or treatment thereof containing gelling or thickening agents of microbial origin, e.g. xanthan or dextran
- A23L29/27—Xanthan not combined with other microbial gums
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS, OR NON-ALCOHOLIC BEVERAGES, NOT COVERED BY SUBCLASSES A21D OR A23B-A23J; THEIR PREPARATION OR TREATMENT, e.g. COOKING, MODIFICATION OF NUTRITIVE QUALITIES, PHYSICAL TREATMENT; PRESERVATION OF FOODS OR FOODSTUFFS, IN GENERAL
- A23L29/00—Foods or foodstuffs containing additives; Preparation or treatment thereof
- A23L29/20—Foods or foodstuffs containing additives; Preparation or treatment thereof containing gelling or thickening agents
- A23L29/275—Foods or foodstuffs containing additives; Preparation or treatment thereof containing gelling or thickening agents of animal origin, e.g. chitin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K35/00—Medicinal preparations containing materials or reaction products thereof with undetermined constitution
- A61K35/12—Materials from mammals; Compositions comprising non-specified tissues or cells; Compositions comprising non-embryonic stem cells; Genetically modified cells
- A61K35/20—Milk; Whey; Colostrum
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
- A61K38/01—Hydrolysed proteins; Derivatives thereof
- A61K38/012—Hydrolysed proteins; Derivatives thereof from animals
- A61K38/018—Hydrolysed proteins; Derivatives thereof from animals from milk
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
- A61K38/16—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- A61K38/17—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- A61K38/22—Hormones
- A61K38/30—Insulin-like growth factors, i.e. somatomedins, e.g. IGF-1, IGF-2
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
- A61K38/16—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- A61K38/17—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- A61K38/40—Transferrins, e.g. lactoferrins, ovotransferrins
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
- A61K38/16—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- A61K38/43—Enzymes; Proenzymes; Derivatives thereof
- A61K38/44—Oxidoreductases (1)
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/24—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite containing atoms other than carbon, hydrogen, oxygen, halogen, nitrogen or sulfur, e.g. cyclomethicone or phospholipids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/30—Macromolecular organic or inorganic compounds, e.g. inorganic polyphosphates
- A61K47/36—Polysaccharides; Derivatives thereof, e.g. gums, starch, alginate, dextrin, hyaluronic acid, chitosan, inulin, agar or pectin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/30—Macromolecular organic or inorganic compounds, e.g. inorganic polyphosphates
- A61K47/36—Polysaccharides; Derivatives thereof, e.g. gums, starch, alginate, dextrin, hyaluronic acid, chitosan, inulin, agar or pectin
- A61K47/38—Cellulose; Derivatives thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/30—Macromolecular organic or inorganic compounds, e.g. inorganic polyphosphates
- A61K47/42—Proteins; Polypeptides; Degradation products thereof; Derivatives thereof, e.g. albumin, gelatin or zein
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/02—Stomatological preparations, e.g. drugs for caries, aphtae, periodontitis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/02—Immunomodulators
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/12—Antihypertensives
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23V—INDEXING SCHEME RELATING TO FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES AND LACTIC OR PROPIONIC ACID BACTERIA USED IN FOODSTUFFS OR FOOD PREPARATION
- A23V2002/00—Food compositions, function of food ingredients or processes for food or foodstuffs
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23V—INDEXING SCHEME RELATING TO FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES AND LACTIC OR PROPIONIC ACID BACTERIA USED IN FOODSTUFFS OR FOOD PREPARATION
- A23V2250/00—Food ingredients
- A23V2250/02—Acid
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23V—INDEXING SCHEME RELATING TO FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES AND LACTIC OR PROPIONIC ACID BACTERIA USED IN FOODSTUFFS OR FOOD PREPARATION
- A23V2250/00—Food ingredients
- A23V2250/15—Inorganic Compounds
- A23V2250/156—Mineral combination
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23V—INDEXING SCHEME RELATING TO FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES AND LACTIC OR PROPIONIC ACID BACTERIA USED IN FOODSTUFFS OR FOOD PREPARATION
- A23V2250/00—Food ingredients
- A23V2250/15—Inorganic Compounds
- A23V2250/156—Mineral combination
- A23V2250/1578—Calcium
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23V—INDEXING SCHEME RELATING TO FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES AND LACTIC OR PROPIONIC ACID BACTERIA USED IN FOODSTUFFS OR FOOD PREPARATION
- A23V2250/00—Food ingredients
- A23V2250/18—Lipids
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23V—INDEXING SCHEME RELATING TO FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES AND LACTIC OR PROPIONIC ACID BACTERIA USED IN FOODSTUFFS OR FOOD PREPARATION
- A23V2250/00—Food ingredients
- A23V2250/18—Lipids
- A23V2250/184—Emulsifier
- A23V2250/1842—Lecithin
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23V—INDEXING SCHEME RELATING TO FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES AND LACTIC OR PROPIONIC ACID BACTERIA USED IN FOODSTUFFS OR FOOD PREPARATION
- A23V2250/00—Food ingredients
- A23V2250/50—Polysaccharides, gums
- A23V2250/502—Gums
- A23V2250/5036—Carrageenan
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23V—INDEXING SCHEME RELATING TO FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES AND LACTIC OR PROPIONIC ACID BACTERIA USED IN FOODSTUFFS OR FOOD PREPARATION
- A23V2250/00—Food ingredients
- A23V2250/50—Polysaccharides, gums
- A23V2250/502—Gums
- A23V2250/5056—Ghatti, indian gum
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23V—INDEXING SCHEME RELATING TO FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES AND LACTIC OR PROPIONIC ACID BACTERIA USED IN FOODSTUFFS OR FOOD PREPARATION
- A23V2250/00—Food ingredients
- A23V2250/50—Polysaccharides, gums
- A23V2250/502—Gums
- A23V2250/507—Locust bean, carob
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23V—INDEXING SCHEME RELATING TO FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES AND LACTIC OR PROPIONIC ACID BACTERIA USED IN FOODSTUFFS OR FOOD PREPARATION
- A23V2250/00—Food ingredients
- A23V2250/50—Polysaccharides, gums
- A23V2250/502—Gums
- A23V2250/5072—Pectine, pectinate
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23V—INDEXING SCHEME RELATING TO FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES AND LACTIC OR PROPIONIC ACID BACTERIA USED IN FOODSTUFFS OR FOOD PREPARATION
- A23V2250/00—Food ingredients
- A23V2250/50—Polysaccharides, gums
- A23V2250/502—Gums
- A23V2250/5086—Xanthan
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23V—INDEXING SCHEME RELATING TO FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES AND LACTIC OR PROPIONIC ACID BACTERIA USED IN FOODSTUFFS OR FOOD PREPARATION
- A23V2250/00—Food ingredients
- A23V2250/50—Polysaccharides, gums
- A23V2250/51—Polysaccharide
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23V—INDEXING SCHEME RELATING TO FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES AND LACTIC OR PROPIONIC ACID BACTERIA USED IN FOODSTUFFS OR FOOD PREPARATION
- A23V2250/00—Food ingredients
- A23V2250/50—Polysaccharides, gums
- A23V2250/51—Polysaccharide
- A23V2250/5108—Cellulose
- A23V2250/51082—Carboxymethyl cellulose
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23V—INDEXING SCHEME RELATING TO FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES AND LACTIC OR PROPIONIC ACID BACTERIA USED IN FOODSTUFFS OR FOOD PREPARATION
- A23V2250/00—Food ingredients
- A23V2250/50—Polysaccharides, gums
- A23V2250/51—Polysaccharide
- A23V2250/5114—Dextrins, maltodextrins
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23V—INDEXING SCHEME RELATING TO FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES AND LACTIC OR PROPIONIC ACID BACTERIA USED IN FOODSTUFFS OR FOOD PREPARATION
- A23V2250/00—Food ingredients
- A23V2250/50—Polysaccharides, gums
- A23V2250/51—Polysaccharide
- A23V2250/5118—Starch
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23V—INDEXING SCHEME RELATING TO FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES AND LACTIC OR PROPIONIC ACID BACTERIA USED IN FOODSTUFFS OR FOOD PREPARATION
- A23V2250/00—Food ingredients
- A23V2250/54—Proteins
- A23V2250/542—Animal Protein
- A23V2250/5424—Dairy protein
- A23V2250/54246—Casein
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23V—INDEXING SCHEME RELATING TO FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES AND LACTIC OR PROPIONIC ACID BACTERIA USED IN FOODSTUFFS OR FOOD PREPARATION
- A23V2250/00—Food ingredients
- A23V2250/54—Proteins
- A23V2250/542—Animal Protein
- A23V2250/5424—Dairy protein
- A23V2250/54248—Lactoferrin
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23V—INDEXING SCHEME RELATING TO FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES AND LACTIC OR PROPIONIC ACID BACTERIA USED IN FOODSTUFFS OR FOOD PREPARATION
- A23V2250/00—Food ingredients
- A23V2250/54—Proteins
- A23V2250/542—Animal Protein
- A23V2250/5424—Dairy protein
- A23V2250/5425—Lactoperoxidase
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23V—INDEXING SCHEME RELATING TO FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES AND LACTIC OR PROPIONIC ACID BACTERIA USED IN FOODSTUFFS OR FOOD PREPARATION
- A23V2250/00—Food ingredients
- A23V2250/54—Proteins
- A23V2250/542—Animal Protein
- A23V2250/5424—Dairy protein
- A23V2250/54252—Whey protein
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23V—INDEXING SCHEME RELATING TO FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES AND LACTIC OR PROPIONIC ACID BACTERIA USED IN FOODSTUFFS OR FOOD PREPARATION
- A23V2250/00—Food ingredients
- A23V2250/60—Sugars, e.g. mono-, di-, tri-, tetra-saccharides
- A23V2250/64—Sugar alcohols
- A23V2250/6416—Maltitol
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23V—INDEXING SCHEME RELATING TO FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES AND LACTIC OR PROPIONIC ACID BACTERIA USED IN FOODSTUFFS OR FOOD PREPARATION
- A23V2250/00—Food ingredients
- A23V2250/60—Sugars, e.g. mono-, di-, tri-, tetra-saccharides
- A23V2250/64—Sugar alcohols
- A23V2250/642—Sorbitol
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23V—INDEXING SCHEME RELATING TO FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES AND LACTIC OR PROPIONIC ACID BACTERIA USED IN FOODSTUFFS OR FOOD PREPARATION
- A23V2250/00—Food ingredients
- A23V2250/70—Vitamins
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Y—ENZYMES
- C12Y111/00—Oxidoreductases acting on a peroxide as acceptor (1.11)
- C12Y111/01—Peroxidases (1.11.1)
- C12Y111/01007—Peroxidase (1.11.1.7), i.e. horseradish-peroxidase
Definitions
- the present invention relates to a protein composition having high heat resistance and a method for producing the same. More specifically, the present invention relates to a highly heat-resistant protein composition containing a milk basic protein fraction and a stabilizer, and a method for producing the same.
- the basic protein fraction derived from milk has various physiological functions such as bone strengthening action, periodontal disease prevention action, lipid metabolism improvement action, blood pressure lowering action, skin collagen production promotion action, immune system control action, etc. It has been reported. In order to effectively use these physiological functions, various foods, drinks, feeds, and medicines containing a milk basic protein fraction have been developed. However, it is known that the milk basic protein fraction is unstable to heating in the neutral region and precipitates when heated at 80 ° C. for 10 minutes (see, for example, Patent Document 1). For this reason, a method of heat-treating a milk basic protein fraction in an acidic region is generally performed. If this method is used, it has been reported that the milk basic protein fraction hardly changes its bone strengthening effect even when heat-treated (see Non-Patent Document 1, for example).
- the heat stability of the milk basic protein fraction is low with respect to sterilization exceeding 90 ° C., particularly retort sterilization treatment, and (1) heat sterilization at 90 ° C. or higher because heat resistance is not sufficient.
- the milk basic protein fraction tends to aggregate and precipitate, and (2) the milk basic protein fraction tends to be inactivated when heated and sterilized in an environment of neutral range (about pH 7) or higher. It was. Therefore, when heat-treating the liquid containing the milk basic protein fraction (particularly at 90 ° C or higher), the milk basic protein fraction may be inactivated, and an ultra-high temperature heat treatment such as retort sterilization is adopted. What is not possible is the actual situation, and there are limitations when blended into foods and drinks, feeds and medicines without inactivating the milk basic protein fraction.
- An object of the present invention is to provide a protein composition capable of heating a protein composition at 90 ° C. or higher without inactivating the milk basic protein fraction and a method for producing the same.
- the present invention has the following configuration.
- the protein composition according to (1), wherein the milk basic protein fraction has the following properties.
- SDS-PAGE sodium dodecyl sulfate-polyacrylamide gel electrophoresis
- proteins having a molecular weight ranging from 3,000 to 80,000.
- 95% by weight or more is protein and contains a small amount of other fat and ash.
- Protein mainly consists of lactoferrin and lactoperoxidase.
- the amino acid composition of the protein contains 15% by weight or more of basic amino acids.
- the milk basic protein fraction contains at least one stabilizer of soybean polysaccharide, xanthan gum, pectin, gum arabic, gati gum, carrageenan, locust bean gum, sodium caseinate, lecithin, carboxymethyl cellulose, 90
- a method for heat-treating a milk basic protein fraction that is heated at a temperature of at least C (6) The method for heat-treating a milk basic protein fraction according to (5), wherein the milk basic protein fraction contains 15% by weight or more of a basic amino acid in the amino acid composition.
- the method for heat-treating a milk basic protein fraction according to (5), wherein the milk basic protein fraction has the following properties.
- SDS-PAGE sodium dodecyl sulfate-polyacrylamide gel electrophoresis
- proteins having a molecular weight ranging from 3,000 to 80,000.
- 95% by weight or more is protein and contains a small amount of other fat and ash.
- Protein mainly consists of lactoferrin and lactoperoxidase.
- the amino acid composition of the protein contains 15% by weight or more of basic amino acids.
- the protein composition can be heated at 90 ° C. or higher without inactivating the milk basic protein fraction.
- the present inventors have made extensive studies on the thermal stabilization of the milk basic protein fraction, and as a result, contain the milk basic protein fraction and a stabilizer such as soybean polysaccharide or xanthan gum. It was found that the heat resistance of the milk basic protein fraction was remarkably improved by using the protein composition, and the present invention was completed. That is, the protein composition according to the embodiment of the present invention comprises a milk basic protein fraction and soy polysaccharide, xanthan gum, pectin, gum arabic, gati gum, carrageenan, locust bean gum, sodium caseinate, lecithin, and carboxymethyl cellulose. And at least one stabilizer selected from the group.
- This protein composition has high heat resistance even in a high temperature heat treatment of 90 ° C. or higher and a retort sterilization treatment of 120 ° C. or higher in a wide pH range from acidic to neutral and alkaline.
- the milk basic protein fraction preferably contains 15% by weight or more of basic amino acids in the amino acid composition.
- the milk basic protein fraction can be used from any origin, but for example, the milk basic protein having the following properties known to have bone strengthening action, It is possible to heat at 90 ° C. or higher without losing activity. 1) According to sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE), it consists of several kinds of proteins having a molecular weight ranging from 3,000 to 80,000.
- SDS-PAGE sodium dodecyl sulfate-polyacrylamide gel electrophoresis
- the amino acid composition of the protein contains 15% by weight or more of milk basic amino acids.
- milk basic amino acids include lysine, histidine, and arginine.
- the basic protein fraction is obtained by bringing a milk material such as skim milk or whey into contact with a cation exchange resin to adsorb the basic protein, and the basic protein fraction adsorbed on the resin is 0.1 M. Elution with a 1M salt concentration eluate, collect this elution fraction, desalinate and concentrate by reverse osmosis (RO) membrane, electrodialysis (ED) method, etc., and dry as necessary Can do.
- RO reverse osmosis
- ED electrodialysis
- the source include milk such as cows, buffalos, humans, pigs, sheep, goats and horses.
- milk or a milk-derived raw material is brought into contact with a cation exchanger to adsorb milk basic protein, and then adsorbed to the cation exchanger.
- a method for obtaining a milk basic protein fraction by elution with an eluate having a pH of more than 5 and an ionic strength of more than 0.5 JP-A-5-202098
- Method using an alginate gel JP-A 61-61) No. 246198
- Method of obtaining from whey using inorganic porous particles Japanese Patent Laid-Open No. 1-86839
- Method of obtaining from milk using sulfated ester compound Japanese Patent No. 63-255300, etc.
- the milk basic protein fraction obtained by such a method can be used.
- the stabilizer which has as a main component the component which has the following property is more desirable.
- a stabilizer having a large molecular weight and a coating property such as soybean polysaccharide.
- the stabilizer having these properties prevents inactivation due to aggregation / precipitation of the milk basic protein fraction during heating, and increases the heat resistance of the milk basic protein fraction.
- stabilizers that are not preferred for milk basic protein compositions include those having the following properties.
- a stabilizer having a hydrophobic group and a hydrophilic group such as a sucrose fatty acid ester formed by reacting a fatty acid with a hydroxyl group of sucrose, and having a multimolecular layer adsorptivity.
- a stabilizer having a linear structure such as gellan gum or a stabilizer having a small side chain ratio to the straight chain such as guar gum and tamarind gum.
- the heat resistance of the milk basic protein fraction during heating is not so improved.
- the thing containing a part of these stabilizers is not limited.
- soybean polysaccharide, xanthan gum, pectin, gum arabic, gati gum, carrageenan, locust bean gum, casein sodium, lecithin, Carboxymethylcellulose is preferred, and at least one of these stabilizers is mixed with the milk basic protein fraction to form a milk basic protein composition.
- Some of these stabilizers may have various other functions such as a function as an emulsifier, but can be used without any problem.
- the content ratio of the milk basic protein fraction and the stabilizer in the protein composition is not particularly limited, but the stabilizer is used in an amount of 0.5 to 100 (weight / weight) with respect to the milk basic protein fraction. ), Preferably 1 to 40 (weight / weight).
- the method for preparing the protein composition is not particularly limited.
- the milk basic protein fraction and the stabilizer are suspended or dissolved in deionized water, and stirred and mixed. It is prepared and used in the form of food and drink, feed and medicine.
- the milk basic protein fraction and the stabilizer are sufficiently mixed, and stirring and mixing using an ultradisperser or the like while heating to about 40 to 80 ° C. as necessary.
- the protein composition solution may be concentrated with a UF membrane or lyophilized, if necessary, so that it can be easily used in foods, drinks, feeds and medicines.
- the protein composition has high heat stability in a wide range from the acidic range to the neutral and alkaline range, and can be subjected to high-temperature heat treatment and retort sterilization treatment that are usually used in the production of foods and drinks, feeds, and pharmaceuticals. Dry heat sterilization in the form is also possible. Therefore, various forms of foods and drinks, feeds, and pharmaceuticals such as liquid, gel, powder, and granule can be prepared using the protein composition.
- the protein composition it is possible to adjust the pH by using an inorganic acid such as hydrochloric acid or phosphoric acid, an organic acid such as citric acid or acetic acid, or an alkaline agent such as caustic soda or sodium bicarbonate.
- an inorganic acid such as hydrochloric acid or phosphoric acid
- an organic acid such as citric acid or acetic acid
- an alkaline agent such as caustic soda or sodium bicarbonate.
- high-temperature heat treatment and retort sterilization can be performed without particularly adjusting the pH, but the protein composition is included.
- the heat sterilization conditions and pH can be appropriately selected according to the quality required for foods and drinks, feeds, and medicines.
- the protein composition may be used as a food, drink, feed, or medicine using only the protein composition, and appropriately mixed with other foods, feeds, and raw materials that are usually contained in a medicine, such as sugars, lipids, and flavors. It may be a food or drink, feed or medicine.
- the eluate was desalted with a reverse osmosis (RO) membrane, concentrated, and then freeze-dried to obtain 21 g of a powdery basic protein fraction (Reference Example Product A).
- the obtained milk basic protein fraction was measured by sodium lauryl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE).
- SDS-PAGE sodium lauryl sulfate-polyacrylamide gel electrophoresis
- the molecular weight was distributed in the range of 3,000 to 80,000, and the component composition was It was as shown in Table 1.
- Table 2 shows the results of analyzing the amino acid composition with an amino acid analyzer (L-8500, manufactured by Hitachi, Ltd.) after hydrolysis with 6N hydrochloric acid at 110 ° C. for 24 hours.
- the protein composition was analyzed by the ELISA method, as shown in Table 3, 40% or more of lactoferrin and lactoperoxidase were contained.
- SDS-PAGE was performed according to the following method. SDS-PAGE: 15 ⁇ l of each sample was added to 15 ⁇ l of sample buffer (1.25 ml of 0.5 M Tris-HCl (pH 6.8), 1.0 ml of glycerol, 2.0 ml of 10% SDS, 0.5 ml of 2-mercaptoethanol, 0.1 % BPB (0.25 ml) and heated at 100 ° C. for 5 minutes. Thereafter, each sample was electrophoresed on a 14% polyacrylamide gel (TEFCO SDS-PAGE mini) at 15 ⁇ l. Kaleidoscope Prestained Standards (BioRad) was used as a molecular weight marker. The results are shown in Table 4.
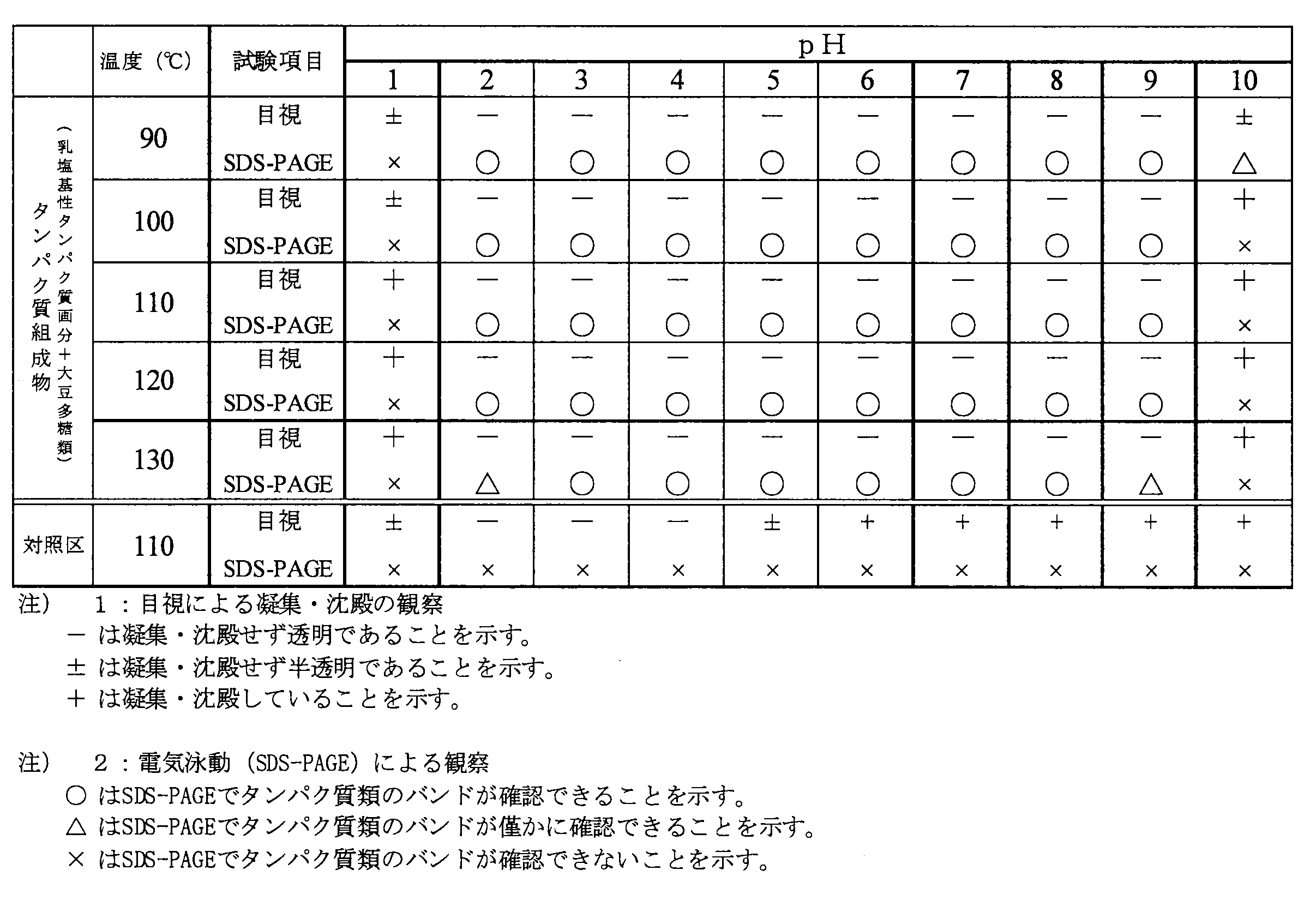
- the solution containing the protein composition containing the milk basic protein fraction and xanthan gum does not cause visual aggregation / precipitation at pH 2 to 9, and the milk basic protein fraction is analyzed by SDS-PAGE. I was able to confirm the band. Therefore, it was found that this protein composition is not only stable on the acidic side, but also extremely heat stable on the neutral and alkaline sides. Further, when the heating time was extended to test whether the milk basic protein fraction could be confirmed, at pH 2-9, no aggregation or precipitation occurred even when heated at 130 ° C. for 10 minutes. A band of fractions was confirmed. Therefore, it was revealed that this protein composition can sufficiently retain the activity of the milk basic protein fraction even after retort sterilization.
- the solution of the protein composition containing milk basic protein and locust bean gum does not cause visual aggregation / precipitation at pH 4 to 7, and the milk basic protein fraction is analyzed by SDS-PAGE. I was able to confirm the band. Therefore, it was found that the protein composition is not only stable on the acidic side, but also extremely heat stable on the neutral side. Further, when the heating time was extended to test whether the milk basic protein fraction could be confirmed, at pH 4-7, it did not aggregate or precipitate even when heated at 120 ° C. for 5 minutes. A band of fractions was confirmed. From this test result, it was revealed that this protein composition can sufficiently retain the activity of the milk basic protein fraction even after the retort sterilization treatment.
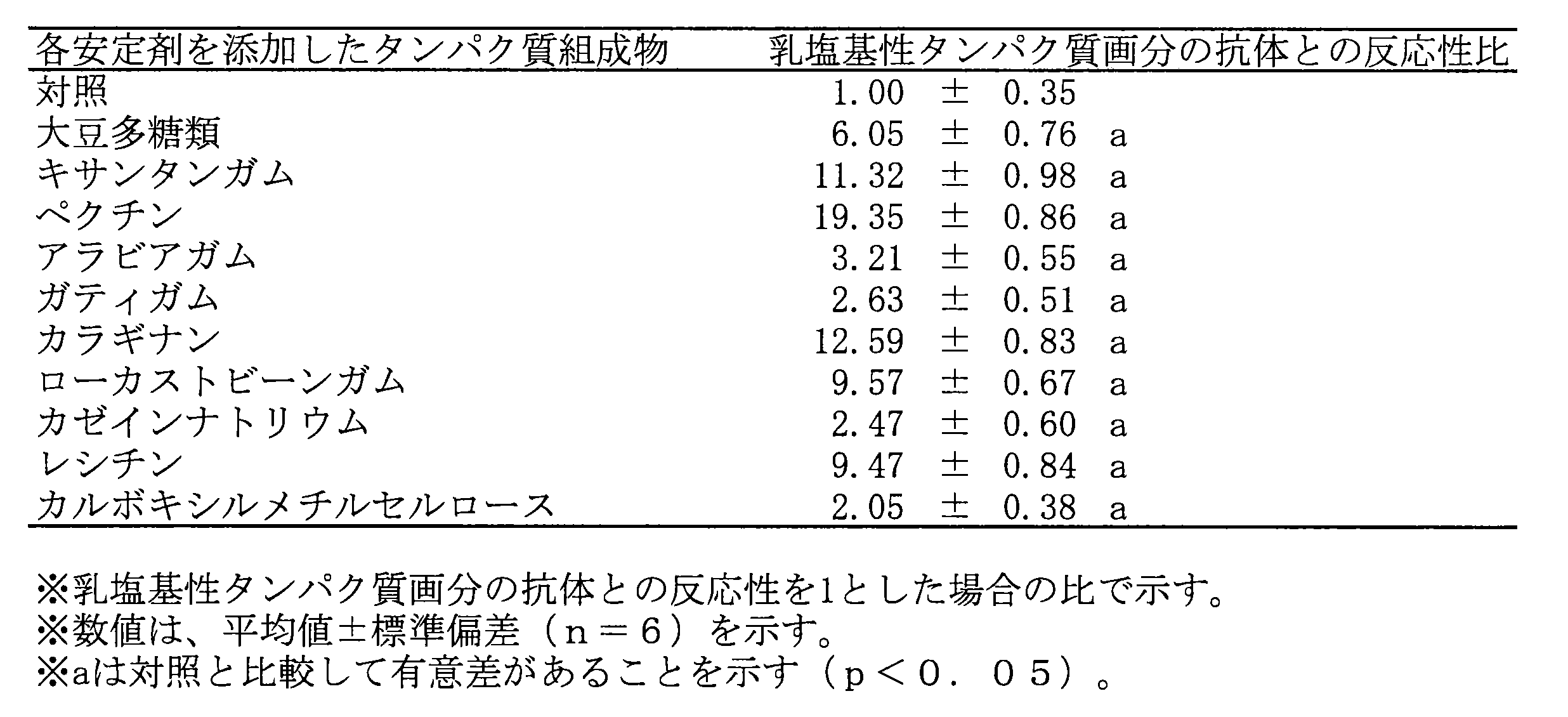
- Test Example 11 For the samples prepared in Test Example 1 to Test Example 10, the pH was adjusted to 7, and the antigenicity when heated at 140 ° C. for 5 minutes was measured by ELISA using an antibody against the milk basic protein fraction. For comparison, the milk basic protein fraction not mixed with the stabilizer was heated at 140 ° C. for 5 minutes to react with the antibody, and the reactivity of each protein composition with the antibody was defined as 1. The ratio is calculated as shown in Table 7.
- Test Example 12 For the samples prepared in Test Example 1 to Test Example 10, the pH was adjusted to 7, and the osteoblast proliferation activity when heated at 140 ° C. for 5 minutes was measured. For comparison, the milk basic protein fraction not mixed with the stabilizer was heated at 140 ° C. for 5 minutes and the osteoblast proliferation activity was taken as 1, and the ratio of the osteoblast proliferation activity of the protein composition to this was shown in Table 8. Show.
- Test Example 13 A solution containing a protein composition (milk basic protein fraction 50 mg%, xanthan gum 0.04% by weight) prepared in the same manner as in Test Example 2 was adjusted to pH 2-9, and 150 ml each was filled in a retort pouch. Sealed. As a control, a solution containing only LF adjusted to pH 2-9 was filled in 150 ml portions of the retort pouch and sealed. These solutions were heated at 120 ° C. for 4 minutes by a retort sterilizer (type 1 pressure vessel, TYPE: RCS-4CRTGN, manufactured by Nisaka Seisakusho). Each sample after heating is stored at 25 ° C., and the presence or absence of aggregation / precipitation is visually observed over time.
- a protein composition milk basic protein fraction 50 mg%, xanthan gum 0.04% by weight
- the band pattern of the milk basic protein fraction is analyzed by polyacrylamide gel electrophoresis (SDS-PAGE).
- SDS-PAGE polyacrylamide gel electrophoresis
- Test Example 14 200 g of a protein composition prepared in the same manner as in Test Example 2 (milk basic protein fraction 50 mg%, xanthan gum 0.04 wt%) is mixed with 800 g of a reduced skim milk solution (skim milk powder 3 wt%) to obtain a protein composition.
- a solution (1) containing a product was prepared.
- 200 g of milk basic protein fraction solution (milk basic protein fraction 50 mg% solution) 200 g of reduced skim milk powder solution (3% by weight of skim milk powder) (2) and reduced skim milk powder solution (skim milk powder)
- a solution (3) of only 1000 g (3 wt%) was prepared.
- 150 ml of each solution was filled into a retort pouch and sealed. These solutions were heated at 120 ° C.
- the milk basic protein fraction of Reference Example Product B was dissolved in deionized water at a concentration of 50 mg% (A solution, 300 g). As a stabilizer, 0.4% by weight of soybean polysaccharide was dissolved in deionized water (solution B, 300 g). Liquid A and liquid B were mixed and stirred and mixed at 50 ° C. and 9500 rpm for 3 minutes in an ultradisperser (ULTRA-TURRAX T-25; manufactured by IKA Japan) to prepare 600 g of the protein composition of the present invention.
- the milk basic protein fraction of Reference Example Product A was dissolved in deionized water at a concentration of 40 mg% (A solution, 10 kg).
- xanthan gum was dissolved in deionized water at 0.08% by weight (solution B, 10 kg).
- Liquid A and liquid B are mixed, mixed with TK homomixer (MARK II II 160 type, manufactured by Tokki Kika Kogyo Co., Ltd.) at 3600 rpm for 30 minutes, and further concentrated on a UF membrane with a molecular weight cut off of 10 kDa. 10 kg of the protein composition was prepared.
- the milk basic protein fraction of Reference Example Product B was dissolved in deionized water at a concentration of 100 mg% (A solution, 1000 kg). As a stabilizer, 0.4% by weight of pectin was dissolved in deionized water (solution B, 1000 kg). 2. Solution A and Solution B are mixed, stirred and mixed at 40 ° C. and 3600 rpm for 40 minutes with a TK homomixer (MARK II II-2500, manufactured by Tokushu Kika Kogyo Co., Ltd.), and freeze-dried to obtain the protein composition of the present invention. 9 kg was prepared.
- TK homomixer MARK II II-2500, manufactured by Tokushu Kika Kogyo Co., Ltd.
- the milk basic protein fraction of Reference Example Product A was dissolved in deionized water at a concentration of 100 mg% (A solution, 500 g).
- a solution, 500 g As a stabilizer, 0.4% by weight of soybean polysaccharide was dissolved in deionized water (solution B, 500 g).
- Liquid A and liquid B were mixed and stirred and mixed for 3 minutes at 40 ° C. and 9500 rpm with an ultradisperser (ULTRA-TURRAX T-25; manufactured by IKA Japan).
- the milk basic protein fraction of Reference Example Product B was dissolved in deionized water at a concentration of 500 mg% (A solution, 200 g).
- As a stabilizer 4% by weight of soybean polysaccharide was dissolved in deionized water (solution B, 200 g).
- This protein composition was filled into a 50 ml glass bottle, sterilized at 90 ° C. for 15 minutes, sealed, and 20 beverages containing the protein composition of the present invention were prepared. In all the prepared beverages, no precipitation was observed, and no abnormal flavor was felt.
- Example 2 0.2 kg of protein composition prepared in Example 2 (milk basic protein fraction 20 mg%, xanthan gum 0.04 wt%), soybean meal 12 kg, skim milk powder 14 kg, soybean oil 4 kg, corn oil 2 kg, palm oil 28 kg, Corn starch (15 kg), wheat flour (9 kg), bran (2 kg), vitamin mixture (9 kg), cellulose (2.8 kg), and mineral mixture (2 kg) were mixed and sterilized at 120 ° C. for 4 minutes to prepare dog breeding feed (100 kg).
- Example 3 kg of the protein composition prepared in Example 3 milk basic protein fraction 50 mg%, pectin 0.2 wt%), 5 kg of casein, 5 kg of soy protein, 1 kg of fish oil, 3 kg of perilla oil, 19 kg of dextrin, 6 kg of mineral mixture, Vitamin mixture 1.95kg, emulsifier 2kg, stabilizer 4kg, flavor 0.05kg, 200ml retort pouch filled, retort sterilizer (type 1 pressure vessel, TYPE: RCS-4CRTGN, manufactured by Nisaka Seisakusho) Was sterilized at 121 ° C. for 20 minutes to prepare 50 kg of enteral nutrient.
- milk basic protein fraction 50 mg%, pectin 0.2 wt%) 5 kg of casein, 5 kg of soy protein, 1 kg of fish oil, 3 kg of perilla oil, 19 kg of dextrin, 6 kg of mineral mixture, Vitamin mixture 1.95kg, emulsifier 2kg, stabilizer 4kg, flavor 0.05kg, 200ml ret
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Polymers & Plastics (AREA)
- Food Science & Technology (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Medicinal Chemistry (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Pharmacology & Pharmacy (AREA)
- Nutrition Science (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Epidemiology (AREA)
- Dispersion Chemistry (AREA)
- Immunology (AREA)
- Zoology (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Gastroenterology & Hepatology (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Organic Chemistry (AREA)
- Inorganic Chemistry (AREA)
- Molecular Biology (AREA)
- Diabetes (AREA)
- Biochemistry (AREA)
- Endocrinology (AREA)
- Mycology (AREA)
- Animal Husbandry (AREA)
- Biomedical Technology (AREA)
- Virology (AREA)
- Developmental Biology & Embryology (AREA)
- Cell Biology (AREA)
- Biotechnology (AREA)
- Biophysics (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Hematology (AREA)
- Cardiology (AREA)
Abstract
Description
(1)乳塩基性タンパク質画分と、大豆多糖類、キサンタンガム、ペクチン、アラビアガム、ガティガム、カラギナン、ローカストビーンガム、カゼインナトリウム、レシチン、カルボキシメチルセルロースからなる群から選択される少なくとも1種の安定化剤と、からなる加熱安定性のあるタンパク質組成物。
(2)乳塩基性タンパク質画分が、アミノ酸組成中に塩基性アミノ酸を15重量%以上含有するものである(1)記載のタンパク質組成物。
(3)乳塩基性タンパク質画分が、以下の性質を有する(1)記載のタンパク質組成物。
1)ソジウムドデシルサルフェート-ポリアクリルアミドゲル電気泳動(SDS-PAGE)によると分子量3,000~80,000の範囲の数種のタンパク質よりなる。
2)95重量%以上がタンパク質であって、その他少量の脂肪、灰分を含む。
3)タンパク質は主としてラクトフェリン及びラクトパーオキシダーゼよりなる。
4)タンパク質のアミノ酸組成は、塩基性アミノ酸を15重量%以上含有する。
(4)(1)~(3)のいずれかに記載のタンパク質組成物を配合した飲食品、飼料及び医薬。
(5)乳塩基性タンパク質画分に、大豆多糖類、キサンタンガム、ペクチン、アラビアガム、ガティガム、カラギナン、ローカストビーンガム、カゼインナトリウム、レシチン、カルボキシメチルセルロースの少なくとも1種の安定化剤を含有させ、90℃以上の温度で加熱する乳塩基性タンパク質画分の加熱処理方法。
(6)乳塩基性タンパク質画分が、アミノ酸組成中に塩基性アミノ酸を15重量%以上含有するものである(5)記載の乳塩基性タンパク質画分の加熱処理方法。
(7)乳塩基性タンパク質画分が、以下の性質を有する(5)記載の乳塩基性タンパク質画分の加熱処理方法。
1)ソジウムドデシルサルフェート-ポリアクリルアミドゲル電気泳動(SDS-PAGE)によると分子量3,000~80,000の範囲の数種のタンパク質よりなる。
2)95重量%以上がタンパク質であって、その他少量の脂肪、灰分を含む。
3)タンパク質は主としてラクトフェリン及びラクトパーオキシダーゼよりなる。
4)タンパク質のアミノ酸組成は、塩基性アミノ酸を15重量%以上含有する。
即ち、本発明の実施形態に係るタンパク質組成物は、乳塩基性タンパク質画分と、大豆多糖類、キサンタンガム、ペクチン、アラビアガム、ガティガム、カラギナン、ローカストビーンガム、カゼインナトリウム、レシチン、カルボキシメチルセルロースからなる群から選択される少なくとも1種の安定化剤と、からなる。このタンパク質組成物は、酸性から中性、アルカリ性の広いpH範囲で、90℃以上の高温加熱処理及び120℃以上のレトルト殺菌処理においても高い耐熱性を有する。
乳塩基性タンパク質画分が、アミノ酸組成中に塩基性アミノ酸を15重量%以上含有することが好ましい。乳塩基性タンパク質画分としては、どのような由来のものであっても使用可能であるが、例えば、骨強化作用を有することが知られている以下の性質を有する乳塩基性タンパク質については、活性を失うことなく90℃以上で加熱することが可能である。
1)ソジウムドデシルサルフェート-ポリアクリルアミドゲル電気泳動(SDS-PAGE)によると分子量3,000~80,000の範囲の数種のタンパク質よりなる。
2)95重量%以上がタンパク質であって、その他少量の脂肪、灰分を含む。
3)タンパク質は主としてラクトフェリン及びラクトパーオキシダーゼよりなる。
4)タンパク質のアミノ酸組成は、乳塩基性アミノ酸を15重量%以上含有する。
なお、乳塩基性アミノ酸としては、例えばリジン、ヒスチジン、アルギニンが挙げられる。
(1)大豆多糖類等のように分子量が大きく、被膜性のある安定剤。
(2)キサンタンガム等のようにグルコース主鎖にマンノースとグルクロン酸が結合しているような、直鎖に対して側鎖の割合が大きい安定剤。
(3)ペクチンのガラクツロン酸主鎖にガラクトース、アラビノース、キシロース等からなる多種類の側鎖を有する安定剤。
(4)アラビアガム等のようにガラクトース主鎖にアラビノースとグルクロン酸が結合しているような、直鎖に対して側鎖の割合が大きい安定剤。
これらの性質を有する安定剤は、加熱時の乳塩基性タンパク質画分の凝集・沈澱による失活を防ぎ、乳塩基性タンパク質画分の耐熱性を高める。
(1)ショ糖のヒドロキシル基に脂肪酸が反応してできるショ糖脂肪酸エステル等のように疎水基と親水基を持ち、多分子層吸着性を有する安定剤。
(2)ジェランガムのように直鎖状の構造をなす安定剤又はグアガム、タマリンドガムのように直鎖に対して側鎖の割合が大きくない安定剤。
これらの安定剤を使用すると加熱時の乳塩基性タンパク質画分の耐熱性はそれほど向上しない。しかし、これらの安定剤を一部含む物はその限りでは無い。
陽イオン交換樹脂のスルホン化キトパール(富士紡績株式会社製)400gを充填したカラム(直径5cm×高さ30cm)を脱イオン水で十分洗浄した後、このカラムに未殺菌脱脂乳40リットル(pH6.7)を流速25ml/minで通液した。通液後、このカラムを脱イオン水で十分洗浄し、0.98M塩化ナトリウムを含む0.02M炭酸緩衝液(pH7.0)で樹脂に吸着した塩基性タンパク質画分を溶出した。そして、この溶出液を逆浸透(RO)膜により脱塩して、濃縮した後、凍結乾燥して粉末状の塩基性タンパク質画分21gを得た(参考例品A)。得られた乳塩基性タンパク質画分について、ラウリル硫酸ナトリウム-ポリアクリルアミドゲル電気泳動(SDS-PAGE)により測定したところ、分子量は3,000~80,000の範囲に分布しており、成分組成は表1に示すとおりであった。また、6N塩酸で110℃、24時間加水分解した後、アミノ酸分析装置(L-8500型、日立製作所製)でそのアミノ酸組成を分析した結果を表2に示した。さらに、ELISA法によりにより、そのタンパク質組成を分析したところ、表3に示すように、40%以上のラクトフェリン及びラクトパーオキシダーゼが含まれていた。
陽イオン交換樹脂のSPトーヨーパール(東ソー株式会社製)30kgを充填したカラム(直径100cm×高さ10cm)を脱イオン水で十分洗浄した後、このカラムに121℃で30秒間加熱殺菌したチーズホエー3t(pH6.2)を、流速10リットル/minで通液した。通液後、このカラムを脱イオン水で十分洗浄し、0.9M塩化ナトリウムを含む0.1Mクエン酸緩衝液(pH5.7)で樹脂に吸着した塩基性タンパク質画分を溶出した。そして、この溶出液を電気透析(ED)法により脱塩し、濃縮した後、凍結乾燥して粉末状の乳塩基性タンパク質画分183gを得た(参考例品B)。
参考例品Aの乳塩基性タンパク質画分を100mg%濃度で脱イオン水に溶解した(A液)。安定剤として大豆多糖類0.2重量%を脱イオン水に溶解した(B液)。A液とB液を混合し、ウルトラディスパーサー(ULTRA-TURRAX T-25;IKAジャパン社製)にて、50℃、8000rpmで3分間撹拌混合してタンパク質組成物を調製した。次いで、このタンパク質組成物を乳酸または水酸化ナトリウム溶液を用いてpH1~10の10試料に調整した。これをアンプル管に2mlずつ分注し、90℃、100℃、110℃、120℃、130℃にて4分間加熱した。対照区として安定剤を含まない乳塩基性タンパク質画分のみの溶液(A液)を上記と同様の方法で、pHを調整して110℃にて4分間加熱した。加熱処理後の各試料及び対照区の凝集・沈殿状態を目視により判定した。
一方、加熱処理による乳塩基性タンパク質画分の分解の程度を確認するため、加熱処理後の各試料及び対照区のサンプルについて、ポリアクリルアミドゲル電気泳動(SDS-PAGE)により、乳塩基性タンパク質画分のバンドパターンの解析を行った。SDS-PAGEは以下の方法にしたがって実施した。
SDS-PAGE:各試料15μlをサンプルバッファー15μl(0.5M Tris-HCl(pH6.8)1.25ml、グリセロール1.0ml、10%SDS 2.0ml、2-メルカプトエタノール0.5ml、0.1%BPB 0.25ml)にて希釈し、100℃で5分間加熱した。その後各試料を15μlずつ14%ポリアクリルアミドゲル(TEFCO SDS-PAGE mini)にて電気泳動した。分子量マーカーとしてはKaleidoscope Prestained Standards(BioRad)を用いた。この結果を表4に示す。
参考例品Bの乳塩基性タンパク質画分を100mg%濃度で脱イオン水に溶解した(A液)。安定剤としてキサンタンガムを0.04%(重量%)で脱イオン水に溶解した(B液)。A液とB液を混合し、ウルトラディスパーサー(ULTRA-TURRAXT-25;IKAジャパン社製)にて、50℃、9500rpmで3分間撹拌混合してタンパク質組成物を調製した。次いで、このタンパク質組成物を乳酸または水酸化ナトリウム溶液を用いてpH1~10の10試料に調整した。これをアンプル管に2mlずつ分注し、90℃、100℃、110℃、120℃、130℃にて4分間加熱した。対照区として安定剤を含まない乳塩基性タンパク質画分のみの溶液(A液)を上記と同様の方法で、pHを調整して110℃にて4分間加熱した。加熱処理後の各試料及び対照区の凝集・沈殿状態を目視により判定した。また、加熱処理による乳塩基性タンパク質画分の分解の程度を確認するため、加熱処理後の各試料及び対照区のサンプルについて、ポリアクリルアミドゲル電気泳動(SDS-PAGE)により、乳塩基性タンパク質画分のバンドパターンの解析を行った。SDS-PAGEは試験例1と同じ方法で実施した。結果を表5に示す。
参考例品Aの乳塩基性タンパク質画分を100mg%濃度で脱イオン水に溶解した(A液)。安定剤としてペクチン0.2重量%を脱イオン水に溶解した(B液)。A液とB液を混合し、ウルトラディスパーサー(ULTRA-TURRAX T-25;IKAジャパン社製)にて、40℃、8000rpmで3分間撹拌混合してタンパク質組成物を調製した。次いで、このタンパク質組成物を、乳酸または水酸化ナトリウム溶液を用いてpH1~10の10試料に調整した。これをアンプル管に2mlずつ分注し、90℃、100℃、110℃、120℃、130℃にて4分間加熱した。対照区として安定剤を含まない乳塩基性タンパク質画分のみの溶液(A液)を上記と同様の方法で、pHを調整して110℃にて4分間加熱した。加熱処理後の各試料及び対照区の凝集・沈殿状態を目視により判定した。また、加熱処理による乳塩基性タンパク質画分の分解の程度を確認するため、加熱処理後の各試料及び対照区のサンプルについて、ポリアクリルアミドゲル電気泳動(SDS-PAGE)により、乳塩基性タンパク質画分のバンドパターンの解析を行った。SDS-PAGEは試験例1と同じ方法で実施した。結果を表6に示す。
参考例品Bの乳塩基性タンパク質画分を100mg%濃度で脱イオン水に溶解した(A液)。安定剤としてアラビアガム0.1重量%を脱イオン水に溶解した(B液)。A液とB液を混合し、ウルトラディスパーサー(ULTRA-TURRAX T-25;IKAジャパン社製)にて、40℃、8000rpmで3分間撹拌混合してタンパク質組成物を調製した。次いで、このタンパク質組成物を乳酸または水酸化ナトリウム溶液を用いてpH1~10の10試料に調整した。これをアンプル管に2mlずつ分注し、90℃、100℃、110℃、120℃、130℃にて4分間加熱した。対照区として安定剤を含まない乳塩基性タンパク質画分のみの溶液(A液)を上記と同様の方法で、pHを調整して110℃にて4分間加熱した。加熱処理後の各試料及び対照区の凝集・沈殿状態を目視により判定した。また、加熱処理による乳塩基性タンパク質画分の分解の程度を確認するため、加熱処理後の各試料及び対照区のサンプルについて、ポリアクリルアミドゲル電気泳動(SDS-PAGE)により、乳塩基性タンパク質画分のバンドパターンの解析を行った。SDS-PAGEは試験例1と同じ方法で実施した。
参考例品Aの乳塩基性タンパク質画分を100mg%濃度で脱イオン水に溶解した(A液)。安定剤としてガディガム0.1重量%を脱イオン水に溶解した(B液)。A液とB液を混合し、ウルトラディスパーサー(ULTRA-TURRAX T-25;IKAジャパン社製)にて、40℃、8000rpmで4分間撹拌混合してタンパク質組成物を調製した。次いで、このタンパク質組成物を乳酸または水酸化ナトリウム溶液を用いてpH1~10の10試料に調整した。これをアンプル管に2mlずつ分注し、90℃、100℃、110℃、120℃、130℃にて4分間加熱した。対照区として安定剤を含まない乳塩基性タンパク質画分のみの溶液(A液)を上記と同様の方法で、pHを調整して110℃にて4分間加熱した。加熱処理後の各試料及び対照区の凝集・沈殿状態を目視により判定した。また、加熱処理による乳塩基性タンパク質画分の分解の程度を確認するため、加熱処理後の各試料及び対照区のサンプルについて、ポリアクリルアミドゲル電気泳動(SDS-PAGE)により、乳塩基性タンパク質画分のバンドパターンの解析を行った。SDS-PAGEは試験例1と同じ方法で実施した。
参考例品Bの乳塩基性タンパク質画分を100mg%濃度で脱イオン水に溶解した(A液)。安定剤としてカラギナン0.2重量%を脱イオン水に溶解した(B液)。A液とB液を混合し、ウルトラディスパーサー(ULTRA-TURRAX T-25;IKAジャパン社製)にて、40℃、8000rpmで3分間撹拌混合してタンパク質組成物を調製した。次いで、このタンパク質組成物を乳酸または水酸化ナトリウム溶液を用いてpH1~10の10試料に調整した。これをアンプル管に2mlずつ分注し、90℃、100℃、110℃、120℃、130℃にて4分間加熱した。対照区として安定剤を含まない乳塩基性タンパク質画分のみの溶液(A液)を上記と同様の方法で、pHを調整して110℃にて4分間加熱した。加熱処理後の各試料及び対照区の凝集・沈殿状態を目視により判定した。また、加熱処理による乳塩基性タンパク質画分の分解の程度を確認するため、加熱処理後の各試料及び対照区のサンプルについて、ポリアクリルアミドゲル電気泳動(SDS-PAGE)により、乳塩基性タンパク質画分のバンドパターンの解析を行った。SDS-PAGEは試験例1と同じ方法で実施した。
参考例品Aの乳塩基性タンパク質画分を100mg%濃度で脱イオン水に溶解した(A液)。安定剤としてローカストビーンガム0.15重量%を脱イオン水に溶解した(B液)。A液とB液を混合し、ウルトラディスパーサー(ULTRA-TURRAX T-25;IKAジャパン社製)にて、40℃、8000rpmで3分間撹拌混合してタンパク質組成物を調製した。次いで、このタンパク質組成物を乳酸または水酸化ナトリウム溶液を用いてpH1~10の10試料に調整した。これをアンプル管に2mlずつ分注し、90℃、100℃、110℃、120℃、130℃にて4分間加熱した。対照区として安定剤を含まない乳塩基性タンパク質画分のみの溶液(A液)を上記と同様の方法で、pHを調整して110℃にて4分間加熱した。加熱処理後の各試料及び対照区の凝集・沈殿状態を目視により判定した。また、加熱処理による乳塩基性タンパク質画分の分解の程度を確認するため、加熱処理後の各試料及び対照区のサンプルについて、ポリアクリルアミドゲル電気泳動(SDS-PAGE)により、乳塩基性タンパク質画分のバンドパターンの解析を行った。SDS-PAGEは試験例1と同じ方法で実施した。
参考例品Bの乳塩基性タンパク質画分を100mg%濃度で脱イオン水に溶解した(A液)。安定剤としてカゼインナトリウム 0.15重量%を脱イオン水に溶解した(B液)。A液とB液を混合し、ウルトラディスパーサー(ULTRA-TURRAX T-25;IKAジャパン社製)にて、40℃、9500rpmで3分間撹拌混合してタンパク質組成物を調製した。次いで、このタンパク質組成物を乳酸または水酸化ナトリウム溶液を用いてpH1~10の10試料に調整した。これをアンプル管に2mlずつ分注し、90℃、100℃、110℃、120℃、130℃にて4分間加熱した。対照区として安定剤を含まない乳塩基性タンパク質画分のみの溶液(A液)を上記と同様の方法で、pHを調整して110℃にて4分間加熱した。加熱処理後の各試料及び対照区の凝集・沈殿状態を目視により判定した。また、加熱処理による乳塩基性タンパク質画分の分解の程度を確認するため、加熱処理後の各試料及び対照区のサンプルについて、ポリアクリルアミドゲル電気泳動(SDS-PAGE)により、乳塩基性タンパク質画分のバンドパターンの解析を行った。SDS-PAGEは試験例1と同じ方法で実施した。
参考例品Aの乳塩基性タンパク質画分を100mg%濃度で脱イオン水に溶解した(A液)。安定剤としてレシチン0.25重量%を脱イオン水に溶解した(B液)。A液とB液を混合し、ウルトラディスパーサー(ULTRA-TURRAX T-25;IKAジャパン社製)にて、40℃、8000rpmで3分間撹拌混合してタンパク質組成物を調製した。次いで、このタンパク質組成物を乳酸または水酸化ナトリウム溶液を用いて~10の10試料に調整した。これをアンプル管に2mlずつ分注し、90℃、100℃、110℃、120℃、130℃にて4分間加熱した。対照区として安定剤を含まない乳塩基性タンパク質画分のみの溶液(A液)を上記と同様の方法で、pHを調整して110℃にて4分間加熱した。加熱処理後の各試料及び対照区の凝集・沈殿状態を目視により判定した。また、加熱処理による乳塩基性タンパク質画分の分解の程度を確認するため、加熱処理後の各試料及び対照区のサンプルについて、ポリアクリルアミドゲル電気泳動(SDS-PAGE)により、乳塩基性タンパク質画分のバンドパターンの解析を行った。SDS-PAGEは試験例1と同じ方法で実施した。
参考例品Bの乳塩基性タンパク質画分を10mg%濃度で脱イオン水に溶解した(A液)。安定剤としてカルボキシメチルセルロース0.15重量%を脱イオン水に溶解した(B液)。A液とB液を混合し、ウルトラディスパーサー(ULTRA-TURRAX T-25;IKAジャパン社製)にて、40℃、8000rpmで3分間撹拌混合してタンパク質組成物を調製した。次いで、このタンパク質組成物を、乳酸または水酸化ナトリウム溶液を用いてpH1~10の10試料に調整した。これをアンプル管に2mlずつ分注し、90℃、100℃、110℃、120℃、130℃にて4分間加熱した。対照区として安定剤を含まない乳塩基性タンパク質画分のみの溶液(A液)を上記と同様の方法で、pHを調整して110℃にて4分間加熱した。加熱処理後の各試料及び対照区の凝集・沈殿状態を目視により判定した。また、加熱処理による乳塩基性タンパク質画分の分解の程度を確認するため、加熱処理後の各試料及び対照区のサンプルについて、ポリアクリルアミドゲル電気泳動(SDS-PAGE)により、乳塩基性タンパク質画分のバンドパターンの解析を行った。SDS-PAGEは試験例1と同じ方法で実施した。
試験例1~試験例10で調製した試料について、pHを7に調整し、140℃で5分間加熱した際の抗原性を、乳塩基性タンパク質画分に対する抗体を用いてELISA法により測定した。比較対象として、安定剤と混合しない乳塩基性タンパク質画分を140℃で5分間加熱し、抗体と反応させた時の反応性を1とし、各タンパク質組成物の抗体との反応性を、これに対する比として算出し、表7に示す。
試験例1~試験例10で調製した試料について、pHを7に調整し、140℃で5分間加熱した際の骨芽細胞増殖活性を測定した。比較対象として、安定剤と混合しない乳塩基性タンパク質画分を140℃で5分間加熱したものの骨芽細胞増殖活性を1とし、これに対するタンパク質組成物の骨芽細胞増殖活性の比を表8に示す。
試験例2と同様の方法で調製したタンパク質組成物(乳塩基性タンパク質画分50mg%、キサンタンガム0.04重量%)を含有する溶液をpH2~9に調整し、150mlずつレトルトパウチに充填し、密封した。対照としてLFのみを含む溶液をpH2~9に調整したものを150mlずつレトルトパウチに充填し、密封した。これらの溶液を レトルト殺菌機(第1種圧力容器、TYPE: RCS-4CRTGN、日阪製作所製)により120℃、4分間加熱した。加熱後のそれぞれの試料を25℃で保存して、経時的に凝集・沈殿の有無を目視により観察し、また、ポリアクリルアミドゲル電気泳動(SDS-PAGE)により乳塩基性タンパク質画分のバンドパターンの解析を行った。
その結果、対照としたLFのみを含む溶液をpH2~9に調整したものは、1日目で凝集・沈澱が確認できたが、タンパク質組成物を含有する溶液をpH2~9に調整したものは、保存後1ヶ月経っても凝集・沈澱は認められなかった。また、SDS-PAGEにて乳塩基性タンパク質画分のバンドパターンを解析したところ、タンパク質組成物を含む溶液をpH2~9に調整したものは保存開始時から保存後1ヶ月においても乳塩基性タンパク質画分のバンドが確認でき、他の変化は認められなかった。この試験結果により、本発明によるタンパク質組成物はレトルト殺菌処理の場合にも有効であることが判明した。
試験例2と同様の方法で調製したタンパク質組成物200g(乳塩基性タンパク質画分50mg%、キサンタンガム0.04重量%)に還元脱脂粉乳溶液(脱脂粉乳3重量%)800gを混合し、タンパク質組成物を含有する溶液(1)を調製した。対照として乳塩基性タンパク質画分溶液(乳塩基性タンパク質画分50mg%溶液)200gに還元脱脂粉乳溶液(脱脂粉乳3重量%)800gを混合した溶液(2)、及び還元脱脂粉乳溶液(脱脂粉乳3重量%)1000gのみの溶液(3)を調製した。各溶液を150mlずつレトルトパウチに充填し、密封した。これらの溶液をレトルト殺菌機 (第1種圧力容器、TYPE:RCS-4CRTGN、日阪製作所製)により120℃、20分加熱した。その結果、溶液(1)、(3)においては凝集・沈殿は認められず、風味も良好であったが、(2)の溶液では凝集・沈殿が認められた。この試験結果により、本発明によるタンパク質組成物はレトルト殺菌処理の場合に大変有効であることが判明した。
参考例品Aの乳塩基性タンパク質画分を100mg%濃度で脱イオン水に溶解した(A液)。安定剤として大豆多糖類0.4重量%、キサンタンガム0.04重量%、タマリンドガム0.05重量%及びショ糖脂肪酸エステル0.1重量%をそれぞれ脱イオン水に溶解した(B液)。A液とB液を混合し、ウルトラディスパーサー(ULTRA-TURRAX T-25;IKAジャパン社製)にて、50℃、8000rpmで3分間撹拌混合してタンパク質組成物を調製した。このタンパク質組成物を乳酸または水酸化ナトリウム溶液を用いてpH6.5に調製した。得られた試料について、試験例1の方法に従い、120℃で4分間の加熱の前後において、各試料の凝集・沈澱状態を目視により確認し、タンパク質組成物の分解の程度をSDS-PAGEにより確認した。結果を表9に示す。
Claims (7)
- 乳塩基性タンパク質画分と、
大豆多糖類、キサンタンガム、ペクチン、アラビアガム、ガティガム、カラギナン、ローカストビーンガム、カゼインナトリウム、レシチン、カルボキシメチルセルロースからなる群から選択される少なくとも1種の安定化剤と、
からなる加熱安定性のあるタンパク質組成物。 - 前記乳塩基性タンパク質画分が、アミノ酸組成中に塩基性アミノ酸を15重量%以上含有するものである請求項1記載のタンパク質組成物。
- 前記乳塩基性タンパク質画分が、以下の性質を有する請求項1記載のタンパク質組成物。
1)ソジウムドデシルサルフェート-ポリアクリルアミドゲル電気泳動(SDS-PAGE)によると分子量3,000~80,000の範囲の数種のタンパク質よりなる。
2)95重量%以上がタンパク質であって、その他少量の脂肪、灰分を含む。
3)タンパク質は主としてラクトフェリン及びラクトパーオキシダーゼよりなる。
4)タンパク質のアミノ酸組成は、塩基性アミノ酸を15重量%以上含有する。 - 請求項1~3のいずれか1項に記載のタンパク質組成物を配合した飲食品、飼料及び医薬。
- 乳塩基性タンパク質画分に、大豆多糖類、キサンタンガム、ペクチン、アラビアガム、ガティガム、カラギナン、ローカストビーンガム、カゼインナトリウム、レシチン、カルボキシメチルセルロースからなる群から選択される少なくとも1種の安定化剤を含有させ、90℃以上の温度で加熱する乳塩基性タンパク質画分の加熱処理方法。
- 前記乳塩基性タンパク質画分が、アミノ酸組成中に塩基性アミノ酸を15重量%以上含有するものである請求項5記載の乳塩基性タンパク質画分の加熱処理方法。
- 前記乳塩基性タンパク質画分が、以下の性質を有する請求項5記載の乳塩基性タンパク質画分の加熱処理方法。
1)ソジウムドデシルサルフェート-ポリアクリルアミドゲル電気泳動(SDS-PAGE)によると分子量3,000~80,000の範囲の数種のタンパク質よりなる。
2)95重量%以上がタンパク質であって、その他少量の脂肪、灰分を含む。
3)タンパク質は主としてラクトフェリン及びラクトパーオキシダーゼよりなる。
4)タンパク質のアミノ酸組成は、塩基性アミノ酸を15重量%以上含有する。
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP14773646.6A EP2984935A4 (en) | 2013-03-28 | 2014-03-25 | PROTEIN COMPOSITION AND METHOD FOR MANUFACTURING THE SAME |
| CN201480018942.0A CN105072920A (zh) | 2013-03-28 | 2014-03-25 | 蛋白质组合物及其生产方法 |
| US14/779,647 US9861121B2 (en) | 2013-03-28 | 2014-03-25 | Milk basic protein composition and production process therefor |
| PH12015501937A PH12015501937A1 (en) | 2013-03-28 | 2015-09-02 | Protein composition and production process therefor |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2013-070417 | 2013-03-28 | ||
| JP2013070417A JP6309202B2 (ja) | 2013-03-28 | 2013-03-28 | タンパク質組成物およびその製造方法 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2014157156A1 true WO2014157156A1 (ja) | 2014-10-02 |
Family
ID=51624127
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2014/058209 WO2014157156A1 (ja) | 2013-03-28 | 2014-03-25 | タンパク質組成物およびその製造方法 |
Country Status (8)
| Country | Link |
|---|---|
| US (1) | US9861121B2 (ja) |
| EP (1) | EP2984935A4 (ja) |
| JP (1) | JP6309202B2 (ja) |
| CN (1) | CN105072920A (ja) |
| MY (1) | MY169153A (ja) |
| PH (1) | PH12015501937A1 (ja) |
| TW (1) | TWI675621B (ja) |
| WO (1) | WO2014157156A1 (ja) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN108366589A (zh) * | 2015-12-11 | 2018-08-03 | 雀巢产品技术援助有限公司 | 无菌rtd花生乳及其制备方法 |
Families Citing this family (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN108463124B (zh) * | 2015-12-25 | 2023-04-18 | 大塚制药株式会社 | 凝胶样食品组合物 |
| US10736342B2 (en) * | 2016-03-03 | 2020-08-11 | General Mills, Inc. | Aseptic protein beverage and method of preparation |
| WO2019176246A1 (ja) | 2018-03-12 | 2019-09-19 | 森永乳業株式会社 | ラクトフェリン含有水溶液の製造方法 |
| WO2020136536A1 (en) * | 2018-12-24 | 2020-07-02 | Societe Des Produits Nestle Sa | Pet foods comprising gravy topping comprising methylcellulose and methods of making such pet foods |
| CN113475655A (zh) * | 2021-05-31 | 2021-10-08 | 仙乐健康科技股份有限公司 | 一种含乳饮品及其制备方法 |
Citations (19)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS61246198A (ja) | 1985-02-07 | 1986-11-01 | オレオフイナ・ソシエテ・アノニム | 蛋白の精製法 |
| JPS63255300A (ja) | 1987-04-10 | 1988-10-21 | Snow Brand Milk Prod Co Ltd | 硫酸エステル化物を用いて乳からラクトフエリンを分離、精製する方法 |
| JPS6486839A (en) | 1987-06-19 | 1989-03-31 | Entamonto Sa | Method for selectively extracting metal protein from whey |
| JPH05202098A (ja) | 1992-01-29 | 1993-08-10 | Snow Brand Milk Prod Co Ltd | 乳質原料から生理活性物質の製造法 |
| JPH07147906A (ja) * | 1993-06-11 | 1995-06-13 | Soc Prod Nestle Sa | タン白およびタン白含有エマルジョン用の安定性組成物 |
| JPH09163A (ja) * | 1995-05-08 | 1997-01-07 | Campina Melkunie Bv | 加水分解物で安定化された熱安定性水中油型エマルション |
| JPH09191856A (ja) * | 1996-01-17 | 1997-07-29 | Snow Brand Milk Prod Co Ltd | 栄養補給用組成物 |
| JPH09191858A (ja) | 1996-01-23 | 1997-07-29 | Snow Brand Milk Prod Co Ltd | 塩基性タンパク質組成物、塩基性ペプチド組成物及びその利用 |
| JPH09238614A (ja) * | 1996-03-06 | 1997-09-16 | Snow Brand Milk Prod Co Ltd | ホエーの調製方法 |
| JPH09294537A (ja) * | 1996-04-26 | 1997-11-18 | Snow Brand Milk Prod Co Ltd | 乳児用調製乳 |
| JPH11103788A (ja) * | 1997-09-30 | 1999-04-20 | Snow Brand Milk Prod Co Ltd | 家畜飼料 |
| JP2002234844A (ja) * | 2000-12-05 | 2002-08-23 | Toyo Seito Kk | 骨密度向上剤およびその利用 |
| JP2002281895A (ja) * | 2001-03-26 | 2002-10-02 | Snow Brand Milk Prod Co Ltd | 弱酸性乳飲料及びその製造方法 |
| JP2007166913A (ja) * | 2005-12-19 | 2007-07-05 | Sanei Gen Ffi Inc | 酸性蛋白含有飲料及びその製造方法 |
| JP2007246413A (ja) * | 2006-03-14 | 2007-09-27 | Snow Brand Milk Prod Co Ltd | 乳由来塩基性タンパク質含有組成物 |
| JP2007259733A (ja) * | 2006-03-28 | 2007-10-11 | Sanei Gen Ffi Inc | 発酵乳食品用安定剤および該安定剤を含有する発酵乳食品 |
| JP2007306865A (ja) * | 2006-05-19 | 2007-11-29 | Sanei Gen Ffi Inc | 密封容器入り乳飲料用安定剤 |
| JP2007330256A (ja) * | 2006-05-19 | 2007-12-27 | Sanei Gen Ffi Inc | 乳成分入り飲料用安定剤 |
| WO2012102100A1 (ja) * | 2011-01-26 | 2012-08-02 | 雪印メグミルク株式会社 | 感覚改善剤 |
Family Cites Families (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP4082782B2 (ja) * | 1998-04-30 | 2008-04-30 | 雪印乳業株式会社 | 歯周病予防及び改善剤 |
| US20030040475A1 (en) | 2001-01-16 | 2003-02-27 | Yasuhiro Toba | Agents for improving lipid metabolism and reducing high blood pressure |
| JP4592260B2 (ja) * | 2003-05-29 | 2010-12-01 | 雪印乳業株式会社 | ラクトフェリン組成物 |
| CA2704144C (en) | 2007-11-01 | 2015-12-29 | Snow Brand Milk Products Co., Ltd. | Food material for promoting the differentiation of osteoblast and inhibiting the differentiation of osteoclast |
| JP2010180219A (ja) | 2010-03-04 | 2010-08-19 | Snow Brand Milk Prod Co Ltd | ラクトフェリン組成物 |
-
2013
- 2013-03-28 JP JP2013070417A patent/JP6309202B2/ja active Active
-
2014
- 2014-03-25 EP EP14773646.6A patent/EP2984935A4/en not_active Withdrawn
- 2014-03-25 WO PCT/JP2014/058209 patent/WO2014157156A1/ja active Application Filing
- 2014-03-25 CN CN201480018942.0A patent/CN105072920A/zh active Pending
- 2014-03-25 US US14/779,647 patent/US9861121B2/en not_active Expired - Fee Related
- 2014-03-25 MY MYPI2015703020A patent/MY169153A/en unknown
- 2014-03-27 TW TW103111562A patent/TWI675621B/zh active
-
2015
- 2015-09-02 PH PH12015501937A patent/PH12015501937A1/en unknown
Patent Citations (19)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS61246198A (ja) | 1985-02-07 | 1986-11-01 | オレオフイナ・ソシエテ・アノニム | 蛋白の精製法 |
| JPS63255300A (ja) | 1987-04-10 | 1988-10-21 | Snow Brand Milk Prod Co Ltd | 硫酸エステル化物を用いて乳からラクトフエリンを分離、精製する方法 |
| JPS6486839A (en) | 1987-06-19 | 1989-03-31 | Entamonto Sa | Method for selectively extracting metal protein from whey |
| JPH05202098A (ja) | 1992-01-29 | 1993-08-10 | Snow Brand Milk Prod Co Ltd | 乳質原料から生理活性物質の製造法 |
| JPH07147906A (ja) * | 1993-06-11 | 1995-06-13 | Soc Prod Nestle Sa | タン白およびタン白含有エマルジョン用の安定性組成物 |
| JPH09163A (ja) * | 1995-05-08 | 1997-01-07 | Campina Melkunie Bv | 加水分解物で安定化された熱安定性水中油型エマルション |
| JPH09191856A (ja) * | 1996-01-17 | 1997-07-29 | Snow Brand Milk Prod Co Ltd | 栄養補給用組成物 |
| JPH09191858A (ja) | 1996-01-23 | 1997-07-29 | Snow Brand Milk Prod Co Ltd | 塩基性タンパク質組成物、塩基性ペプチド組成物及びその利用 |
| JPH09238614A (ja) * | 1996-03-06 | 1997-09-16 | Snow Brand Milk Prod Co Ltd | ホエーの調製方法 |
| JPH09294537A (ja) * | 1996-04-26 | 1997-11-18 | Snow Brand Milk Prod Co Ltd | 乳児用調製乳 |
| JPH11103788A (ja) * | 1997-09-30 | 1999-04-20 | Snow Brand Milk Prod Co Ltd | 家畜飼料 |
| JP2002234844A (ja) * | 2000-12-05 | 2002-08-23 | Toyo Seito Kk | 骨密度向上剤およびその利用 |
| JP2002281895A (ja) * | 2001-03-26 | 2002-10-02 | Snow Brand Milk Prod Co Ltd | 弱酸性乳飲料及びその製造方法 |
| JP2007166913A (ja) * | 2005-12-19 | 2007-07-05 | Sanei Gen Ffi Inc | 酸性蛋白含有飲料及びその製造方法 |
| JP2007246413A (ja) * | 2006-03-14 | 2007-09-27 | Snow Brand Milk Prod Co Ltd | 乳由来塩基性タンパク質含有組成物 |
| JP2007259733A (ja) * | 2006-03-28 | 2007-10-11 | Sanei Gen Ffi Inc | 発酵乳食品用安定剤および該安定剤を含有する発酵乳食品 |
| JP2007306865A (ja) * | 2006-05-19 | 2007-11-29 | Sanei Gen Ffi Inc | 密封容器入り乳飲料用安定剤 |
| JP2007330256A (ja) * | 2006-05-19 | 2007-12-27 | Sanei Gen Ffi Inc | 乳成分入り飲料用安定剤 |
| WO2012102100A1 (ja) * | 2011-01-26 | 2012-08-02 | 雪印メグミルク株式会社 | 感覚改善剤 |
Non-Patent Citations (2)
| Title |
|---|
| See also references of EP2984935A4 |
| SEIICHIRO AOE ET AL., BIOSCIENCE BIOTECHNOLOGY AND BIOCHEMISTRY, vol. 65, no. 4, 2001, pages 913 - 918 |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN108366589A (zh) * | 2015-12-11 | 2018-08-03 | 雀巢产品技术援助有限公司 | 无菌rtd花生乳及其制备方法 |
Also Published As
| Publication number | Publication date |
|---|---|
| JP6309202B2 (ja) | 2018-04-11 |
| US9861121B2 (en) | 2018-01-09 |
| PH12015501937B1 (en) | 2016-01-11 |
| MY169153A (en) | 2019-02-18 |
| EP2984935A1 (en) | 2016-02-17 |
| TWI675621B (zh) | 2019-11-01 |
| CN105072920A (zh) | 2015-11-18 |
| US20160044949A1 (en) | 2016-02-18 |
| JP2014193125A (ja) | 2014-10-09 |
| TW201517807A (zh) | 2015-05-16 |
| EP2984935A4 (en) | 2016-11-30 |
| PH12015501937A1 (en) | 2016-01-11 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6309202B2 (ja) | タンパク質組成物およびその製造方法 | |
| JP6918188B2 (ja) | ホエイプロテインを含む高プロテイン飲料 | |
| KR19990076831A (ko) | 칼슘복합체에 의한 식품의 영양가 보강 | |
| JP4804951B2 (ja) | 酸性飲食品の製造方法 | |
| AU2005331240B2 (en) | Iron composition containing milk protein | |
| JP5991882B2 (ja) | 酸性ゼリー飲食品及びその製造方法 | |
| JP4592260B2 (ja) | ラクトフェリン組成物 | |
| JP2010180219A (ja) | ラクトフェリン組成物 | |
| JP4410074B2 (ja) | 酸性ゲル状食品 | |
| JP2020043772A (ja) | 加熱凝固卵白の製造方法 | |
| JP2004298047A (ja) | 酸性ゲル状食品 | |
| JP5955615B2 (ja) | 弱酸性のタンパク質含有ゲル状飲食品 | |
| JP4005103B2 (ja) | 未変性ラクトフェリン入り殺菌飲料及びその製造法 | |
| JP5969225B2 (ja) | 骨強化剤 | |
| JP7450397B2 (ja) | タンパク質凝固剤、タンパク質含有飲食品、タンパク質含有飲食品の物性改善剤、及びタンパク質含有飲食品の物性を改善する方法 | |
| JP2024054490A (ja) | タンパク質のゲル化又は増粘を抑制するために用いる組成物 | |
| JP6279851B2 (ja) | 筋萎縮防止及び/又は筋合成促進剤 | |
| JP4059169B2 (ja) | カルシウム添加豆乳およびその製造法 | |
| Mehra et al. | β-Lactoglobulin: A Whey Protein Fraction with Enhanced Functionality | |
| JP2007175065A (ja) | 未変性ラクトフェリン入り殺菌飲料及びその製造法 | |
| NZ562043A (en) | Iron composition containing milk protein |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 201480018942.0 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 14773646 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 12015501937 Country of ref document: PH |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 14779647 Country of ref document: US |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2014773646 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: IDP00201506902 Country of ref document: ID |