WO2014128111A1 - 4-substituierte pyrrolo- und pyrazolo-diazepine - Google Patents
4-substituierte pyrrolo- und pyrazolo-diazepine Download PDFInfo
- Publication number
- WO2014128111A1 WO2014128111A1 PCT/EP2014/053099 EP2014053099W WO2014128111A1 WO 2014128111 A1 WO2014128111 A1 WO 2014128111A1 EP 2014053099 W EP2014053099 W EP 2014053099W WO 2014128111 A1 WO2014128111 A1 WO 2014128111A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- alkyl
- alkoxy
- ring atoms
- halogen
- amino
- Prior art date
Links
- 0 C*(*)CCC=C(C)C(c1c(*)[n]cc1NC(OC(C)(C)C)=O)=O Chemical compound C*(*)CCC=C(C)C(c1c(*)[n]cc1NC(OC(C)(C)C)=O)=O 0.000 description 3
- CJDYFMIDIQXELO-UHFFFAOYSA-N CC(C)(C)OC(N1CCN(C)CC1)=O Chemical compound CC(C)(C)OC(N1CCN(C)CC1)=O CJDYFMIDIQXELO-UHFFFAOYSA-N 0.000 description 2
- SJRJJKPEHAURKC-UHFFFAOYSA-N CN1CCOCC1 Chemical compound CN1CCOCC1 SJRJJKPEHAURKC-UHFFFAOYSA-N 0.000 description 2
- VKMAYFRJLADGMY-UHFFFAOYSA-N FC(C1)CN1I Chemical compound FC(C1)CN1I VKMAYFRJLADGMY-UHFFFAOYSA-N 0.000 description 2
- LFFIQXPAJSWCFZ-UHFFFAOYSA-N O=C(CC1)CN1I Chemical compound O=C(CC1)CN1I LFFIQXPAJSWCFZ-UHFFFAOYSA-N 0.000 description 2
- FDHKDPNLKXVBPO-UHFFFAOYSA-N O=C(CC1)N(CC2)C1CN2I Chemical compound O=C(CC1)N(CC2)C1CN2I FDHKDPNLKXVBPO-UHFFFAOYSA-N 0.000 description 2
- JFOLPOYZBFMBIY-UHFFFAOYSA-N O=S(CC1)(CCN1I)=O Chemical compound O=S(CC1)(CCN1I)=O JFOLPOYZBFMBIY-UHFFFAOYSA-N 0.000 description 2
- AELBLAHIAVCDLK-UHFFFAOYSA-N CC(C)(C)OC(Nc1c[n](C)c(C)c1C(c1ccccc1)=O)=O Chemical compound CC(C)(C)OC(Nc1c[n](C)c(C)c1C(c1ccccc1)=O)=O AELBLAHIAVCDLK-UHFFFAOYSA-N 0.000 description 1
- OYVXVLSZQHSNDK-UHFFFAOYSA-N CC(N(C)OC)=O Chemical compound CC(N(C)OC)=O OYVXVLSZQHSNDK-UHFFFAOYSA-N 0.000 description 1
- NMPCBSLMMRJXOM-UHFFFAOYSA-N CCNC(CC(c1nnc(C)[n]1-c1c[n](C)nc11)N=C1c(cc1)ccc1Cl)=O Chemical compound CCNC(CC(c1nnc(C)[n]1-c1c[n](C)nc11)N=C1c(cc1)ccc1Cl)=O NMPCBSLMMRJXOM-UHFFFAOYSA-N 0.000 description 1
- CWOXFODHGGGNSS-HNNXBMFYSA-N Cc1c2C(c(cc3)ccc3Cl)=N[C@@H](CC(OC)=O)c3nnc(C)[n]3-c2c[n]1C Chemical compound Cc1c2C(c(cc3)ccc3Cl)=N[C@@H](CC(OC)=O)c3nnc(C)[n]3-c2c[n]1C CWOXFODHGGGNSS-HNNXBMFYSA-N 0.000 description 1
- UHBPUVRZIYOPJO-AWEZNQCLSA-N Cc1cnc2[n]1-c1c[n](C)nc1C(c(cc1)ccc1Cl)=N[C@H]2CC(OC)=O Chemical compound Cc1cnc2[n]1-c1c[n](C)nc1C(c(cc1)ccc1Cl)=N[C@H]2CC(OC)=O UHBPUVRZIYOPJO-AWEZNQCLSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D487/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00
- C07D487/12—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00 in which the condensed system contains three hetero rings
- C07D487/14—Ortho-condensed systems
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/55—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having seven-membered rings, e.g. azelastine, pentylenetetrazole
- A61K31/551—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having seven-membered rings, e.g. azelastine, pentylenetetrazole having two nitrogen atoms, e.g. dilazep
- A61K31/5513—1,4-Benzodiazepines, e.g. diazepam or clozapine
- A61K31/5517—1,4-Benzodiazepines, e.g. diazepam or clozapine condensed with five-membered rings having nitrogen as a ring hetero atom, e.g. imidazobenzodiazepines, triazolam
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P13/00—Drugs for disorders of the urinary system
- A61P13/08—Drugs for disorders of the urinary system of the prostate
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P15/00—Drugs for genital or sexual disorders; Contraceptives
- A61P15/08—Drugs for genital or sexual disorders; Contraceptives for gonadal disorders or for enhancing fertility, e.g. inducers of ovulation or of spermatogenesis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P15/00—Drugs for genital or sexual disorders; Contraceptives
- A61P15/16—Masculine contraceptives
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/28—Drugs for disorders of the nervous system for treating neurodegenerative disorders of the central nervous system, e.g. nootropic agents, cognition enhancers, drugs for treating Alzheimer's disease or other forms of dementia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/04—Antibacterial agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/12—Antivirals
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
- A61P35/02—Antineoplastic agents specific for leukemia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/02—Immunomodulators
- A61P37/06—Immunosuppressants, e.g. drugs for graft rejection
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/10—Drugs for disorders of the cardiovascular system for treating ischaemic or atherosclerotic diseases, e.g. antianginal drugs, coronary vasodilators, drugs for myocardial infarction, retinopathy, cerebrovascula insufficiency, renal arteriosclerosis
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D519/00—Heterocyclic compounds containing more than one system of two or more relevant hetero rings condensed among themselves or condensed with a common carbocyclic ring system not provided for in groups C07D453/00 or C07D455/00
Definitions
- the present invention relates to BET protein-inhibiting, in particular BRD2, BRD3 and BRD4-inhibitory, 4-substituted pyrrolo and pyrazolo-diazepines, pharmaceutical compositions containing the compounds of the invention and their prophylactic and therapeutic use in hyper-proliferative diseases, especially in tumor diseases ,
- this invention relates to the use of BET protein inhibitors in benign hyperplasia, in atherosclerotic diseases, in sepsis, in autoimmune diseases, in vascular diseases, in viral infections, in neurodegenerative diseases, in
- the human BET family (bromodomain and extra C-terminal domain family) has four members (BRD2, BRD3, BRD4 and BRDT) containing two related bromodomains and one extra-terminal domain (Wu and Chiang, J. Biol. Chem., 2007 , 282: 13141-13145).
- the bromodomains are protein regions that recognize acetylated lysine residues. Such acetylated lysines are often found at the N-terminal end of histones (eg, histone H3 or histone H4) and are features of open chromatin structure and active gene transcription (Kuo and Allis, Bioessays, 1998, 20: 615). 626).
- bromodomains can recognize additional acetylated proteins.
- BRD4 binds to RelA, resulting in the stimulation of NF- ⁇ B and transcriptional activity of inflammatory genes (Huang et al., Mol. Cell Biol., 2009, 29: 1375-1387; Zhang et al., J. Biol Chem., 2012, doi / 10.1074 / jbc.Ml 12.359505).
- BRD2, BRD3 and BRD4 interacts with several proteins that have a role in chromatin modulation and regulation of gene expression (Rahman et al., Mol. Cell Biol., 2011, 31: 2641-2652).
- BET proteins play an important role in cell growth and cell cycle. Biol. Cell, 2009, 20: 4899-4909; Yang et al., Mol. Cell. Biol., 2008, 28: 967-976). BRD4 is important for the post-mitotic reactivation of gene transcription (Zhao et al., Nat Cell Biol., 2011, 13: 1295-1304). It has been shown that BRD4 is essential for the
- RNA polymerase II Transcription elongation and for the recruitment of the elongation complex P-TEFb, which consists of CDK9 and cyclin Tl, which leads to the activation of RNA polymerase II (Yang et al., Mol. Cell, 2005, 19: 535-545; Schröder et al. J. Biol. Chem., 2012, 287: 1090-1099).
- RNA polymerase II Yang et al., Mol. Cell, 2005, 19: 535-545; Schröder et al. J. Biol. Chem., 2012, 287: 1090-1099.
- genes involved in cell proliferation is stimulated, such as c-myc and aurora B (You et al., Mol. Cell. Biol., 2009, 29: 5094-5103; Zuber et al. Nature, 2011, 478: 524-528).
- BRD2 and BRD3 bind to transcribed genes in hyperacetylated chromatin regions
- BRD4 binds to promoter regions of several genes activated in the Gl phase, such as cyclin D1 and D2 (Mochizuki et al., J. Biol. Chem.
- BRD2 and BRD4 knockout mice die prematurely during embryogenesis (Gyuris et al., Biochim Biophys Acta, 2009, 1789: 413-421; Houzelstein et al., Mol. Cell. Biol., 2002, 22: 3794-3802 ).
- BET proteins play an important role in various tumor types.
- the fusion protein prevents cell differentiation and promotes proliferation (Yan et al., J. Biol. Chem., 2011, 286: 27663-27675).
- BRD4 inhibitor The growth of derived in vivo models is inhibited by a BRD4 inhibitor (Filippakopoulos et al., Nature, 2010, 468: 1067-1073). Screening for therapeutic targets in an acute myeloid leukemia cell line (AML) showed that BRD4 plays an important role in this tumor (Zuber et al., Nature, 2011, doi: 10.1038). Reduction of BRD4 expression leads to selective cell cycle arrest and apoptosis. Treatment with a BRD4 inhibitor prevents the proliferation of an AML xenograft in vivo. Amplification of the DNA region containing the BRD4 gene has been detected in primary breast tumors (Kadota et al., Cancer Res.
- BET proteins are also involved in viral infections.
- BRD4 binds to the E2 protein of various papillomaviruses and is important for survival of the viruses in latently infected cells (Wu et al., Genes Dev., 2006, 20: 2383-2396; Vosa et al., J. Viral., 2012 , 86: 348-357).
- the herpesvirus responsible for Kaposi's sarcoma interacts with various BET proteins, which is important for disease resistance (Viejo-Borbolla et al., J. Viral., 2005,
- BRD4 By binding to P-TEFb, BRD4 also plays an important role in the replication of HIV (Bisgrove et al., Proc Natl Acad., USA, 2007, 104: 13690-13695).
- BET proteins are also involved in inflammatory processes.
- BRD2-hypomorphic mice show reduced inflammation in adipose tissue (Wang et al., Biochem J., 2009, 425: 71-83).
- the infiltration of macrophages into white adipose tissue is also reduced in BRD2-deficient mice (Wang et al., Biochem J., 2009, 425: 71-83). It has also been shown that BRD4 has a number of
- a BRD4 inhibitor prevents the expression of inflammatory genes, such as IL-1 or IL-6 (Nicodeme et al., Nature, 2010, 468: 1119-1123).
- BET proteins also regulate the expression of the ApoAl gene, which plays an important role in
- Apolipoprotein AI is a major component of high density lipoproteins (HDL) and increased expression of ApoAl results in elevated blood cholesterol levels (Degoma and Rader, Nat. Rev. Cardiol., 2011, 8: 266-277). Elevated HDL levels are associated with a decreased risk of atherosclerosis (Chapman et al., Eur. Heart J., 2011, 32: 1345-1361).
- the first published BRD4 inhibitors are phenyl-thieno-triazolo-l, 4-diazepine (4-phenyl-6-thieno [3,2-] [l, 2,4] triazolo [4,3-a] [ l, 4] diazepines) as described in WO2009 / 084693 (Mitsubishi Tanabe Pharma Corporation) and with the compound JQ1 in WO2011 / 143669 (Dana Farber Cancer Institute). Replacement of the thieno by a benzo moiety also results in active inhibitors (J. Med. Chem., 2011, 54, 3827-3838, E. Nicodeme et al., Nature 2010, 468, 1119). This and another publication indicate that the 1,4-benzodiazepine or thieno-1,4-diazepine
- Ring system fused pyrazole unit is actively involved in the binding of the target protein BRD4 (P. Filippakopoulos et al., Nature 2010, 468, 1067).
- Other 4-phenyl-6 # -thieno [3,2- /] [l, 2,4] triazolo [4,3-a] [l, 4] diazepines and related compounds with alternative rings as the fusion partner instead of the benzo moiety are generically addressed or directly described in WO2012 / 075456 (Constellation Pharmaceuticals).
- WO2012 / 075383 (Constellation Pharmaceuticals) describes 6-substituted-4 / f-isoxazolo [5,4-öf] [2] benzazepines and 4 / f-isoxazolo [3,4-öf] [2] benzazepines, including compounds disclosed in U.S. Pat Position 6 optionally substituted phenyl, as BRD4 inhibitors and also analogs with alternative heterocyclic fusion partners instead of the benzo moiety, eg Thieno or pyridoazepines.
- WO2013 / 184876 and WO2013 / 184878 (Constellation Pharmaceuticals) describe further benzoisoxazoloazepine derivatives as inhibitors of bromodomain-containing proteins.
- BRD4 inhibitors is 7-isoxazoloquinolines and related quinoline derivatives (WO2011 / 054843, Bioorganic & Medicinal Chemistry Letters 22 (2012) 2963-2967, GlaxoSmithKline).
- Pyridinones and pyridazinones (WO 2013/185284, WO 2013/188381; Abbott Laboratories) and isoindolones (WO 2013/155695, WO 2013/158952; Abbott Laboratories) are inhibitors of binding of bromodomains of BET proteins to N-acetylated lysine. Residues containing proteins described.
- Receptors are described in WO2006 / 051312 (James Black Foundation). They also include substituted 3,5-dihydro-4 / i-2,3-benzodiazepin-4-ones derived from the compounds of the invention mainly by the obligatory oxo group in position 4 and by a distinguish obligatory carbonyl-containing alkyl chain in position 5. Finally, substituted 3,5-dihydro-4 / i-2,3-benzodiazepin-4-ones are also described as AMPA antagonists in WO97 / 34878 (Cocensys Inc.). Despite a very broad generic claim regarding the possible substitution patterns on the benzodiazepine skeleton, the embodiments are limited to a narrow range.
- EP 102602 further describes 6-aryl-diazepinones with fused pyrrole ring, which are used as decongestants and in anxiety states. These can carry side chains in position 4, which are linked by an oxygen or nitrogen atom. A linkage via a carbon atom has not been described.
- DE3435973 describes 6-aryl-triazolo-diazepines which carry an annulated pyrrole ring with N in position 2.
- the compounds are used in the treatment of pathological conditions and diseases involving acetylglyceryl-ester phosphorylcholine (PAF).
- PAF acetylglyceryl-ester phosphorylcholine
- these compounds have no side chain in position 4. Only substitution with a methyl group has been described on a diazepinone system with a pyrazole annulation (J. Med. Chem. 24, (1981), p982ff, DeWald et al.).
- hyperproliferative diseases especially in tumor diseases and as BET protein inhibitors in viral infections, benign hyperplasia in neurodegenerative diseases, in inflammatory diseases, atherosclerotic diseases, autoimmune diseases, vascular diseases, sepsis and in the male
- novel compounds are novel 4-substituted pyrrolo and pyrazolo-diazepines, which surprisingly BET protein-inhibitory, in particular BRD2, BRD3 and BRD
- the compounds of the invention inhibit the interaction between BRD4 and an acetylated histone H4 peptide and inhibit the growth of cancer cells. They thus represent new structures for the therapy of human and animal diseases, in particular of cancers.
- X is a carbon or a nitrogen atom
- n and m are independently 0 or 1
- p 1, 2, 3 or 4
- R 1 , R 4 and R 5 are each independently hydrogen, hydroxy, cyano, nitro, amino,
- R 2 represents hydrogen or represents Ci-COE-alkyl, aminocarbonyl, Ci-COE-alkylcarbonyl, CI-C ⁇ -
- C3-Cio-Cycloalkyl- which is optionally mono- or polysubstituted by identical or different substituents with halogen, amino, hydroxy, Ci-Cö-alkyl, CI-C ⁇ - alkoxy, Ci-C6-alkoxy-Ci -C6-alkyl, Ci-Cö-alkylamino, amino-Ci-Cö-alkyl, Ci-C6-alkylamino-Ci-C6-alkyl, halogen-Ci-Cö-alkyl, halogen-Ci-Cö-alkoxy or a monocyclic heterocyclyl radical having 4 to 8 ring atoms,
- Ci-Cö-alkyl is phenyl, which is optionally monosubstituted or polysubstituted, identically or differently, by halogen, amino, hydroxyl, cyano, nitro, carboxy, C 1 -C 6 -alkyl, C 1 -C 6 -alkoxy, C 1 -C 6 -alkoxy Ci-Cö-alkyl, Ci-Cö-alkylamino, amino-Ci-Cö-alkyl, Ci-Cö-alkylaminocarbonyl, Ci-Cö-alkylaminosulfonyl, Ci-Cö-alkylamino-Ci-Ce-alkyl, Hydroxy-C 1 -C 6 -alkyl, halogen-C 1 -C 6 -alkyl, halogen-C 1 -C 6 -alkoxy, C 1 -C 6 -alkylcarbonyl, C 1 -C 6 -alkylsulfony
- Phenylsulfonyl, Ci-COE-alkylamino, Ci-COE-alkylcarbonylamino, CI-C ⁇ - alkylaminocarbonyl or C-Coe-alkylaminosulfonyl group which is optionally substituted singly or multiply, identically or differently substituted with halogen, amino, Hydroxy, carboxy, hydroxy-Ci-Cö-alkyl, Ci-Cö-alkoxy, Ci-Ce-alkoxy-Ci-Cö-alkyl, Ci-Cö-alkylamino or amino-Ci-Cö-alkyl-, if X for one
- Ci-COE-alkyl represents hydrogen or Ci-COE-alkyl, Ci-COE-alkoxy, Ci-COE-alkylcarbonyl, CI-C ⁇ - alkylaminocarbonyl, Ci-COE-alkylsulfonyl, phenylsulfonyl or CI-C ⁇ - alkylaminosulfonyl which are optionally monosubstituted or polysubstituted by identical or different substituents with halogen, amino, hydroxy, carboxy, hydroxy-Ce-Ce-alkyl, Ci-Ce-alkoxy, Ci-Ce-alkoxy-Ci-Ce-alkyl -, Ci-Ce-alkylamino or amino-Ci-Cö-alkyl-, when X is a nitrogen atom,
- G-G-alkyl independently of one another for hydrogen, G-G-alkyl or for a monocyclic or bicyclic heterocyclyl radical having 4 to 12 ring atoms or a
- a monocyclic or bicyclic heterocyclyl radical having 4 to 12 ring atoms or a monocyclic or bicyclic heteroaryl radical having 5 to 10 ring atoms or a partially saturated bicyclic aryl or heteroaryl radical having 7 to 11 ring atoms, where the radicals mentioned are optionally - or polysubstituted by identical or different substituents with halogen, cyano, nitro, hydroxy, amino, oxo, carboxy, GG-alkyl, G-Ce-alkoxy, Ci-Ce-alkoxy-G-Ce-alkyl, hydroxy -G-Ce-alkyl, GG-alkylamino, GG-alkylcarbonylamino, amino-GG-alkyl, GG-alkylamino-G-Ce-alkyl, halogeno-G-Ce-alkyl, halogeno-G-Ce Alkoxy, C 3 -
- R lj is Ci-Ce-alkyl, halo-Ci-C 3 -alkyl, hydroxy-Ci-C 3 -alkyl, Ci-C 3 -alkoxy-C
- X is a carbon or a nitrogen atom
- n and m are independently 0 or 1
- p stands for 1, independently of one another represent hydrogen, hydroxyl, cyano, nitro, amino,
- C 1 -C 6 -alkyl is hydrogen or C 1 -C 6 -alkyl, C 1 -C 6 -alkylcarbonyl, phenylsulfonyl or C 1 -C 6 -alkylsulfonyl, which are optionally mono- or polysubstituted, identically or differently, by halogen, amino, hydroxyl, carboxy, Hydroxy-G-G-alkyl, GG-alkoxy, GG-alkoxy-GG-alkyl, GG-alkylamino, aminoGG-alkyl, monocyclic heterocyclyl having 4 to 8 ring atoms or monocyclic heteroaryl with 5 or 6 ring atoms in which the monocyclic heterocyclyl and heteroaryl are optionally monosubstituted with GG-alkyl,
- C 3 -Cio-cycloalkyl- which is optionally mono- or polysubstituted by identical or different substituents with halogen, amino, hydroxy, GG-alkyl, GG-alkoxy, Ci-C6-alkoxy-Ci-C6-alkyl -, GG-alkylamino, amino-GG-alkyl, Ci-C6-alkylamino-Ci-C6-alkyl, halogen-GG-alkyl, halogen-Ci-Cö-alkoxy or a monocyclic heterocyclyl radical having 4 to 8 ring atoms .
- phenyl which is optionally monosubstituted or polysubstituted, identically or differently, by halogen, amino, hydroxyl, cyano, nitro, carboxy, GG-alkyl, GG-alkoxy, C 1 -C 6 -alkoxy-C 1 -C 6 -alkyl, Alkyl, GG-alkylamino, amino-GG-alkyl, GG-alkylaminocarbonyl, GG-alkylaminosulfonyl, GG-alkylamino-GG-alkyl, hydroxy-GG-alkyl, halogen-GG-alkyl, halogen GG-alkoxy-GG-alkylsulfonyl, C3-Cio-cycloalkyl or a monocyclic
- GG-alkyl is hydrogen or GG-alkyl, GG-alkylcarbonyl, GG-alkylsulfonyl or phenylsulfonyl, which are optionally monosubstituted or polysubstituted, identically or differently, by halogen, amino, hydroxyl, carboxy, hydroxy-C 1 -C 4 -alkyl, Alkyl, GG-alkoxy, C 1 -C 6 -alkoxy-C 1 -C 6 -alkyl, GG-alkylamino or amino-GG-alkyl- when X is a carbon atom,
- GG-alkyl is hydrogen or GG-alkyl, GG-alkylcarbonyl, GG-alkylsulfonyl or phenylsulfonyl, which are optionally monosubstituted or polysubstituted, identically or differently, by halogen, amino, hydroxyl, carboxy, hydroxy-C 1 -C 4 -alkyl, Alkyl, GG-alkoxy, C 1 -C 6 -alkoxy-C 1 -C 6 -alkyl, G-C 1 -C 4 -alkylamino or amino-GG-alkyl- when X is a nitrogen atom,
- heteroaromatic or heterocyclic ring having 5 to 7 ring atoms, which is optionally mono- or polysubstituted by identical or different substituents with halogen, amino, hydroxy, cyano, nitro, carboxy, GG-alkyl , GG-alkoxy, GG-alkoxy-GG-alkyl, GG-alkylamino, amino-GG-alkyl, G-C0-alkylaminocarbonyl, GC- O -alkylaminosulfonyl, GG-alkylcarbonyl, GG-alkylsulfonyl, GG-alkylamino-GG-alkyl, hydroxy-GG-alkyl, halogeno-G-C6-alkyl, halogeno-G-C0-alkoxy, C3-Cio-cycloalkyl or a monocyclic heterocyclyl radical having 4 to 8 ring
- monocyclic or bicyclic heterocyclyl radical having 4 to 12 ring atoms or a monocyclic or bicyclic heteroaryl radical having 5 to 10 ring atoms or a partially saturated bicyclic aryl or heteroaryl radical having 7 to 11 ring atoms, wherein said radicals optionally one or more times, the same or different are substituted by halogen, cyano, nitro, hydroxy, amino, oxo, carboxy, Ci-Ce-alkyl, GG-alkoxy, GG-alkoxy-GG-alkyl, hydroxy-GG-alkyl, GG-alkylamino, GG-alkylcarbonylamino, amino-GG-alkyl, GG-alkylamino-GG-alkyl, halogen-GG-alkyl, halogen-GG-alkoxy, C3-G0-cycloalkyl, phenyl, halophenyl, phenyl C 1 -C 6 -alkyl
- C3-alkyl, C3-C8-cycloalkyl, phenyl, a monocyclic or bicyclic heterocyclyl radical having 4 to 12 ring atoms or a monocyclic or bicyclic heteroaryl radical having 5 to 10 ring atoms or a partially saturated bicyclic aryl or heteroaryl radical having 7 to 11 Ring atoms is, where the radicals mentioned are optionally mono- or polysubstituted by identical or different substituents with halogen, cyano, nitro, hydroxy, amino, oxo, carboxy, CI-C ⁇ -alkyl, Ci-Ce-alkoxy, Ci-C 6 alkoxy-C 6 alkyl, hydroxy-Ci-Ce-alkyl, GC 6 - alkylamino, Ci-COE-alkylcarbonylamino, amino-Ci-COE-alkyl, Ci-COE-alkylamino C -Ce-alkyl, halo-Ci
- R lj is Ci-Ce-alkyl, halo-Ci-C 3 -alkyl, hydroxy-Ci-C 3 -alkyl, Ci-C 3 -alkoxy-C
- X is a carbon or a nitrogen atom
- n and m are independently 0 or 1
- p stands for 1, independently of one another represent hydrogen, hydroxyl, cyano, aminocarbonyl, halogen or optionally mono- or polysubstituted, identically or differently, with halogen, amino, hydroxyl, carboxy, hydroxy-GG-alkyl, GG-alkoxy, GG-alkoxy-G G-alkyl, GG-alkylamino or amino-GG-alkyl-substituted GG-alkyl, GG-alkoxy, GG-alkylamino, GG-alkylcarbonylamino, GG-alkylaminocarbonyl or GG-alkylaminosulphonyl stand,
- G-G-alkyl is hydrogen or G-G-alkyl, G-G-alkylcarbonyl, phenylsulfonyl or G-G-alkylsulfonyl- which are optionally mono- or polysubstituted, identically or differently, by halogen, amino, hydroxyl or carboxy, or
- phenyl which is optionally mono- or polysubstituted, identically or differently, by halogen, hydroxyl, cyano, carboxy, GG-alkyl, GG-alkoxy, halogen-GG-alkyl, halogeno-GG-alkoxy-, C 3 -Cio-cycloalkyl or a monocyclic heterocyclyl radical having 4 to 8 ring atoms, and
- GG-alkyl is hydrogen or GG-alkyl, GG-alkylcarbonyl, phenylsulfonyl or GG-alkylsulfonyl- which are optionally mono- or polysubstituted by identical or different substituents with halogen, amino, hydroxy, carboxy, hydroxy-G-G-alkyl , GG-alkoxy, C 1 -C 6 -alkoxy-C 1 -C 6 -alkyl, GG-alkylamino or amino-GG-alkyl- when X is a carbon atom,
- GG-alkyl is hydrogen or GG-alkyl, C 1 -C 6 -alkylcarbonyl, phenylsulfonyl or C 1 -C 6 -alkylsulfonyl, which are optionally mono- or polysubstituted, identically or differently, by halogen, amino, hydroxyl, carboxy, hydroxyl C 1 -C 6 -alkyl, C 1 -C 6 -alkoxy, C 1 -C 6 -alkoxy-C 1 -C 6 -alkyl, G-C 1 -C 6 -alkylamino or amino-G-C 6 -alkyl- when X is a nitrogen atom stands,
- Ce-alkyl C 1 -C 6 -alkylamino-C 1 -C 6 -alkyl, halogen-C 1 -C 6 -alkyl or halogeno-C 1 -C 6 -alkoxy,
- R is hydrogen, Ci-C 3 alkyl, cyclopropyl, or D1-C 1 -C 3 6 and R 7 independently of one another - alkylamino-Ci-C3-alkyl are,
- R 8 is hydroxy, C 1 -C 6 -alkyl, C 1 -C 6 -alkoxy, halogeno-C 1 -C 3 -alkyl-, hydroxy-C 1 -C 3 -
- R 10 and R 11 are independently hydrogen, Ci-Cö-alkyl or a
- R 10 and R 11 together with the adjacent nitrogen atom stand for a monocyclic or bicyclic heterocyclyl radical having 4 to 12 ring atoms or a monocyclic or bicyclic heteroaryl radical having 5 to 10 ring atoms or a partially saturated bicyclic aryl or heteroaryl radical having 7 to 11 ring atoms, where the radicals mentioned are optionally monosubstituted or polysubstituted, identically or differently, by halogen, cyano, hydroxyl, amino, oxo, C 1 -C 6 -alkyl-,
- R 13 represents C 1 -C 6 -alkyl, halogeno-C 1 -C 3 -alkyl, hydroxy-C 1 -C 3 -alkyl, C 1 -C 3 -alkoxy-G,
- X is a carbon or a nitrogen atom
- n and m are independently 0 or 1
- R 1 , R 4 and R 5 independently of one another represent hydrogen or halogen
- R 2 is hydrogen or GC ⁇ - alkyl-
- R 3 is C 1 -C 6 -alkyl- when X is a carbon atom
- R 3 is hydrogen or C 1 -C 6 -alkyl, when X is a nitrogen atom,
- Heteroaryl having 5 or 6 ring atoms, wherein phenyl, heteroaryl and
- Heterocyclyl optionally mono- or polysubstituted by identical or different substituents with halogen, cyano, nitro, hydroxy, amino, oxo, carboxy, Ci-Cö-alkyl, Ci-Ce-alkoxy, Ci-Ce-alkoxy-ci Ce-alkyl, hydroxy-C 1 -C 6 -alkyl, C 1 -C 6 -alkylamino, amino-C 1 -C 6 -alkyl, C 1 -C 6 -alkylamino-C 1 -C 6 -alkyl, halogeno-G-C
- R 6 and R 7 independently of one another represent hydrogen, C 1 -C 3 -alkyl, cyclopropyl or C 1 -C 3 -alkylamino-C 1 -C 3 -alkyl,
- R 8 is hydroxy, C 1 -C 6 -alkyl, G-C 1 -alkoxy, halogen-C 1 -C 3 -alkyl, hydroxy-C 1 -C 3 -
- R 9 is C 1 -C 6 -alkyl-
- R 10 and R 11 independently of one another are hydrogen, C 1 -C 6 -alkyl or a monocyclic or bicyclic heterocyclyl radical having 4 to 12 ring atoms or a monocyclic or bicyclic heteroaryl radical having 5 to 10 ring atoms or a partially saturated bicyclic aryl or heteroaryl radical 7 to 11 ring atoms, where the radicals mentioned are optionally monosubstituted or polysubstituted, identically or differently, by halogen, cyano, nitro, hydroxyl, amino, oxo, carboxy, C 1 -C 6 -alkyl, G-C 1 -alkoxy, Ci-C 6 alkoxy-C 6 -alkyl, hydroxy-C 6 - alkyl, CI-COE alkylamino, Ci-COE-alkylcarbonylamino, amino-Ci-COE-alkyl, CI C 6 -C 18 -alkylamino-C 1 -
- R 10 and R 11 together with the adjacent nitrogen atom stand for a monocyclic or bicyclic heterocyclyl radical having 4 to 12 ring atoms or a monocyclic or bicyclic heteroaryl radical having 5 to 10 ring atoms or a partially saturated bicyclic aryl or heteroaryl radical having 7 to 11 ring atoms, where the radicals mentioned are optionally monosubstituted or polysubstituted, identically or differently, by halogen, cyano, hydroxyl, amino, oxo, C 1 -C 6 -alkyl-,
- R 12 is Ci-Ce-alkyl, halo-Ci-C 3 alkyl, hydroxy-C 3 alkyl, Ci-C 3 alkoxy-Ci- C 3 - alkyl, C3-C 8 - Cycloalkyl, phenyl, a monocyclic or bicychschen
- X is a carbon or a nitrogen atom
- n and m are independently 0 or 1
- R 1 , R 4 and R 5 independently of one another represent hydrogen or halogen
- R 2 is hydrogen or C 1 -C 6 -alkyl-
- R 3 is C 1 -C 6 -alkyl- when X is a carbon atom, or
- Ring atoms which itself is optionally mono- or polysubstituted by identical or different substituents with halogen, cyano, nitro, hydroxy, amino, oxo, carboxy, C 1 -C 6 -alkyl, C 1 -C 6 -alkoxy, C 1 -C 6 -alkoxy -Ci-C 6 alkyl, hydroxy-Ci-C 6 alkyl, GC 6 - alkylamino, amino-Ci-Cö-alkyl, Ci-Ce-alkylamino-Ci-Cö-alkyl, halogen-G -Cö-alkyl or halo-Ci-Cö-alkoxy,
- Ci-C 3 alkyl are each independently hydrogen, Ci-C 3 alkyl, cyclopropyl, or D1-C 1 -C 3 - alkylamino-Ci-C 3 are alkyl,
- Ci-Ce-alkyl is hydroxy, Ci-Ce-alkyl, Ci-Ce-alkoxy, halo-Ci-C 3 alkyl, hydroxy-C 3 - alkyl, Ci-C 3 alkoxy-Ci-C 3 alkyl , Cs-Cs-cycloalkyl, phenyl, monocyclic heterocyclyl having 4 to 8 ring atoms or monocyclic heteroaryl having 5 or 6 ring atoms, in which phenyl, heteroaryl and heterocyclyl are optionally monosubstituted or disubstituted by halogen, C 1 -C 3 -alkoxy- or C 1 -C 3 -alkyl-, represents C 1 -C 6 -alkyl-,
- R 10 and R 11 together with the adjacent nitrogen atom stand for a monocyclic or bicyclic heterocyclyl radical having 4 to 12 ring atoms or a monocyclic or bicyclic heteroaryl radical having 5 to 10 ring atoms or a partially saturated bicyclic aryl or heteroaryl radical having 7 to 11 ring atoms, where the radicals mentioned are optionally monosubstituted or polysubstituted, identically or differently, by halogen, cyano, hydroxyl, amino, oxo, C 1 -C 6 -alkyl-,
- R 12 is Ci-Cö-alkyl, Cs-Cs-cycloalkyl or a monocyclic or bicyclic
- Heterocyclyl radical having 4 to 12 ring atoms, wherein said radicals are optionally mono- or polysubstituted by identical or different substituents
- X is a carbon or a nitrogen atom
- n and m are independently 0 or 1
- R 1 , R 4 and R 5 independently of one another represent hydrogen or halogen
- R 2 is hydrogen or C 1 -C 6 -alkyl-
- R 3 is C 1 -C 6 -alkyl- when X is a carbon atom
- R 3 is hydrogen or C 1 -C 6 -alkyl, when X is a nitrogen atom,
- Heteroaryl- with 5 ring atoms which is optionally mono- or polysubstituted with Ci-Cö-alkyl or C3-Cio-cycloalkyl-,
- R 6 and R 7 independently of one another represent hydrogen, C 1 -C 3 -alkyl-, cyclopropyl-, or C 1 -C -Cl 3
- R 8 is hydroxy, Ci-Ce-alkyl, Ci-Ce-alkoxy, halo-Ci-C 3 alkyl, hydroxy-C 3 - alkyl, Ci-C 3 alkoxy-Ci-C 3 Alkyl, Cs-Cs-cycloalkyl, phenyl, monocyclic
- Ring atoms in which phenyl, heteroaryl and heterocyclyl optionally have a or are doubly substituted by halogen, C 1 -C 3 -alkoxy- or C 1 -C 3 -alkyl-, R 9 is C 1 -C 6 -alkyl-,
- R 12 is Ci-Cö-alkyl, Cs-Cs-cycloalkyl or a monocyclic or bicyclic
- Heterocyclyl radical having 4 to 12 ring atoms, where the radicals mentioned are optionally mono- or polysubstituted by identical or different substituents with halogen, oxo, C 1 -C 6 -alkyl- or phenoxy-,
- X is a carbon or a nitrogen atom
- n and m are independently 0 or 1
- R 1 is hydrogen or halogen
- R 2 is hydrogen or C 1 -C 6 -alkyl-
- R 3 is C 1 -C 6 -alkyl- when X is a carbon atom
- R 3 is hydrogen or C 1 -C 6 -alkyl, when X is a nitrogen atom, R 4 and R 5 are hydrogen,
- Heteroaryl- with 5 ring atoms which is optionally mono- or polysubstituted with Ci-Cö-alkyl or C3-Cio-cycloalkyl-,
- R 8 is C 1 -C 6 -alkoxy-
- R 10 and R 11 are independently hydrogen, Ci-Cö-alkyl or a
- R 10 and R 11 together with the adjacent nitrogen atom stand for a monocyclic or bicyclic heterocyclyl radical having 4 to 12 ring atoms, which is optionally monosubstituted or polysubstituted by identical or different substituents with halogen, oxo,
- R 12 is Ci-Cö-alkyl, Cs-Cs-cycloalkyl or a monocyclic or bicyclic
- Heterocyclyl radical having 4 to 12 ring atoms, where the radicals mentioned are optionally mono- or polysubstituted by identical or different substituents with halogen, oxo, C 1 -C 6 -alkyl- or phenoxy-,
- X is a carbon or a nitrogen atom
- n and m are independently 0 or 1
- R 1 is hydrogen or chlorine
- R 2 is hydrogen or methyl
- R 3 is methyl when X is a carbon atom
- R 3 is hydrogen or methyl when X is a nitrogen atom
- R 4 and R 5 are hydrogen
- R 10 is hydrogen or methyl
- R 11 is ethyl or one of the following groups
- R 12 is methyl or ieri-butyl
- R 1 is chlorine in the para position and R 4 and R 5 are hydrogen.
- X is preferably a carbon atom or a nitrogen atom.
- X is preferably a carbon atom. In the general formula (I), X is preferably a nitrogen atom. In the general formula (I), n is preferably 0 or 1. In the general formula (I), m is preferably 0 or 1. In the general formula (I), m is preferably 0 or 1.
- p is preferably 1.
- R 1 , R 4 and R 5 may be the same or different and are preferably hydrogen or halogen.
- R 1 is particularly preferably hydrogen or halogen.
- R 4 and R 5 are more preferably hydrogen.
- those compounds are particularly preferred in which R 1 is hydrogen or halogen and R 4 and R 5 are hydrogen.
- R 1 very particularly preferably represents hydrogen or chlorine.
- those compounds are particularly preferred in which R 1 is chlorine injara position and R 4 and R 5 are hydrogen.
- R 2 is preferably hydrogen or C 1 -C 6 -alkyl-. In the general formula (I), R 2 very particularly preferably represents hydrogen or methyl. In the general formula (I), R 2 is very particularly preferably hydrogen. In the general formula (I), R 2 is very particularly preferably methyl.
- R 3 is preferably C 1 -C 6 -alkyl- when X is a carbon atom or hydrogen or C 1 -C 6 -alkyl- when X is a nitrogen atom.
- R 3 is very particularly preferably methyl, when X is a
- ** which may be substituted with methyl, ethyl, iso-propyl, ieri.-butyl or cyclopropyl, and wherein "*" denotes the point of attachment to the remainder of the molecule.
- R 6 and R 7 are each, independently of one another, hydrogen, C 1 -C 3 -alkyl, cyclopropyl, or di-C 1 -C 3 -alkylamino-C 1 -C 3 - Alkyl stand.
- R 8 is preferably hydroxy, C 1 -C 6 -alkyl, C 1 -C 6 -alkoxy, halogen-C 1 -C 8 -alkyl, hydroxy-C 1 -C 5 -alkyl, C 1 -C 8 -synyl Alkoxy-Ci-Cs-alkyl, Cs-Cs-cycloalkyl, phenyl, monocyclic heterocyclyl having 4 to 8 ring atoms or monocyclic heteroaryl having 5 or 6 ring atoms, wherein phenyl, heteroaryl and heterocyclyl optionally one or are doubly substituted with halogen, Ci-C3-alkoxy or Ci-C 3 -alkyl-.
- R 8 is preferably hydroxy, C 1 -C 6 -alkyl, C 1 -C 6 -alkoxy, C 3 -C 8 -cycloalkyl, phenyl, monocyclic heterocyclyl having 4 to 8 ring atoms or
- R 8 is particularly preferably C 1 -C 6 -alkoxy-.
- R 9 is preferably C 1 -C 6 -alkyl-.
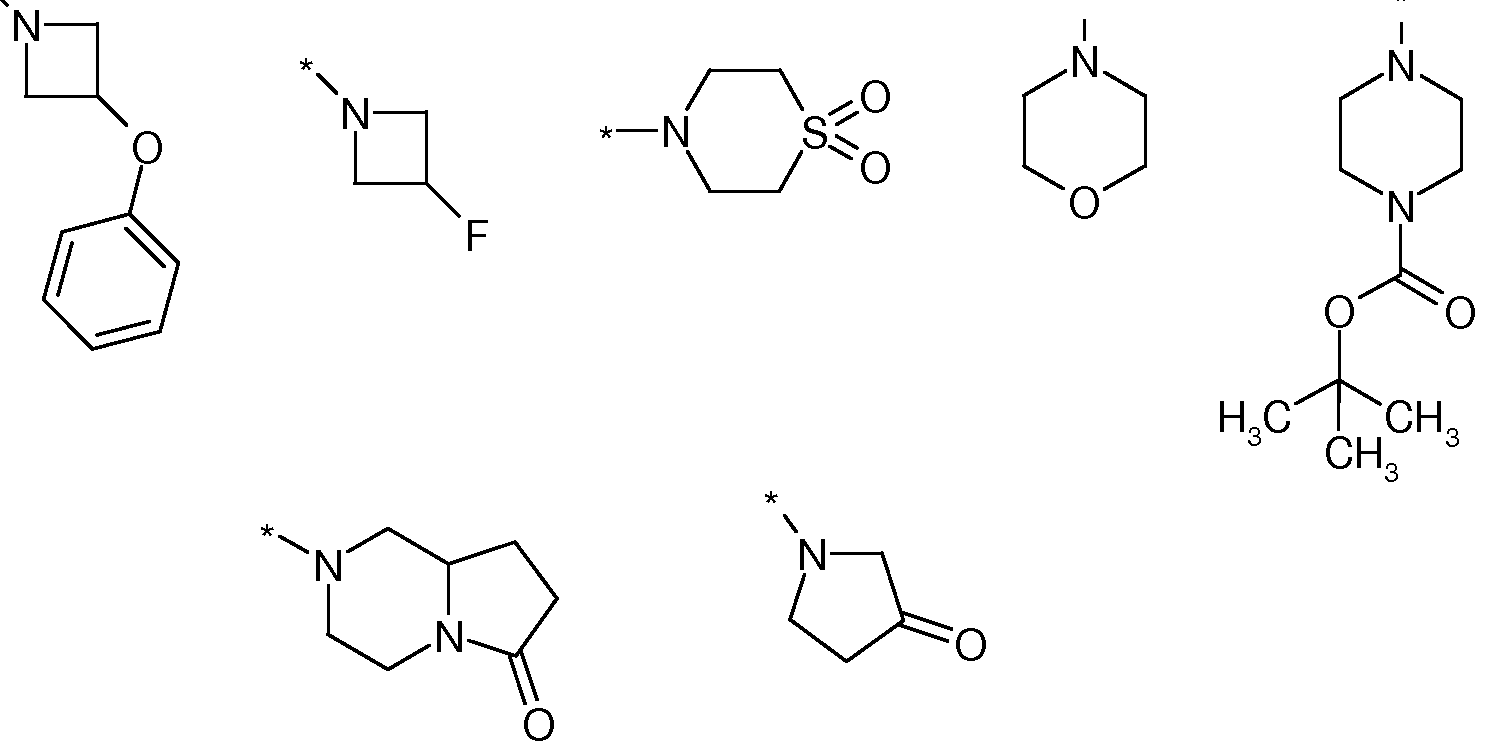
- R 10 and R 11 independently of one another are hydrogen, C 1 -C 6 -alkyl or monocyclic or bicyclic
- Heterocyclyl radical having 4 to 12 ring atoms or a monocyclic or bicyclic
- Heteroaryl radical having 5 to 10 ring atoms or a partially saturated bicyclic aryl or
- Heteroaryl radical having 7 to 11 ring atoms wherein said radicals are optionally mono- or polysubstituted by identical or different substituents with halogen, cyano, nitro, hydroxy, amino, oxo, carboxy, GC 6 alkyl, Ci-C 6 alkoxy C 1 -C 6 -alkoxy-C 1 -C 6 -alkyl, C 1 -C 6 -alkyl, C 1 -C 6 -alkylamino, C 1 -C 6 -alkylcarbonylamino, amino-C 1 -C 6 -alkyl-,
- R 10 and R 11 independently of one another are hydrogen, GG-alkyl- or a monocyclic or bicyclic
- Heterocyclyl radical having 4 to 12 ring atoms or a monocyclic or bicyclic
- Heteroaryl radical having 5 to 10 ring atoms or a partially saturated bicyclic aryl or
- Heteroaryl radical having 7 to 11 ring atoms, wherein the radicals mentioned optionally substituted one or more times, identically or differently with halogen, cyano, nitro, hydroxy, amino, oxo, carboxy, GC 6 alkyl, Ci-C 6 alkoxy C 1 -C 6 -alkoxy-C 1 -C 6 -alkyl, C 1 -C 6 -alkyl, C 1 -C 6 -alkylamino, GG-alkylcarbonylamino, amino-GG-alkyl,
- Heterocyclyl radical having 4 to 8 ring atoms.
- R 10 and R 11 are independently hydrogen, GG-alkyl or a monocyclic or bicyclic heterocyclyl radical having 4 to 12 ring atoms or a monocyclic or bicyclic heteroaryl radical with 5 to 10 ring atoms or a partially saturated aryl or bicyclic
- R 10 and R 11 together with the adjacent nitrogen atom stand for a monocyclic or bicyclic heterocyclyl radical having 4 to 12 ring atoms or a monocyclic or bicyclic heteroaryl radical having 5 to 10 ring atoms or a partially saturated bicyclic aryl or heteroaryl radical having 7 to 11 ring atoms
- the radicals mentioned may optionally contain 1, 2 or 3 heteroatoms from the group of nitrogen, oxygen or sulfur and which are optionally monosubstituted or polysubstituted, identically or differently, by halogen, cyano, amino, oxo, C 1 -C 6 -alkyl , C 1 -C 6 -alkoxy, C 1 -C 6 -alkylamino, C 1 -C 6 -alkylcarbonylamino, halogeno-C 1 -C 6 -alkyl, halogeno-C 1 -C 6 -alkoxy, C 3 -C 10 -cycloalkyl
- R 10 and R 11 are independently hydrogen, Ci-Cö-alkyl or a monocyclic or bicyclic heterocyclyl radical having 4 to 12 ring atoms or a monocyclic or bicyclic heteroaryl radical having 5 to 10 ring atoms or a partially saturated bicyclic aryl or
- radicals having 4 to 12 ring atoms or a monocyclic or bicyclic heteroaryl radical having 5 to 10 ring atoms or a partially saturated bicyclic aryl or heteroaryl radical having 7 to 11 ring atoms, where said radicals are optionally 1, 2, or 3 heteroatoms from the Group may contain nitrogen, oxygen or sulfur and which are optionally mono- or polysubstituted by identical or different substituents with halogen, cyano, amino, oxo,
- R 10 and R 11 are independently hydrogen, Ci-Cö-alkyl or a monocyclic or bicyclic heterocyclyl radical having 4 to 12 ring atoms or a monocyclic or bicyclic Heteroaryl radical having 5 to 10 ring atoms or a partially saturated bicyclic aryl or
- R 10 and R 11 are independently hydrogen, Ci-Cö-alkyl or a monocyclic or bicyclic heterocyclyl radical having 4 to 12 ring atoms or a monocyclic or bicyclic Heteroaryl radical having 5 to 10 ring atoms or a partially saturated bicyclic aryl or
- those compounds are particularly preferred in which R 10 is hydrogen or methyl and R 11 is ethyl or one of the following groups
- R 12 is preferably Ci-Cö-alkyl, Cs-Cs-cycloalkyl or a monocyclic or bicyclic heterocyclyl radical having 4 to 12 ring atoms, wherein said radicals optionally one or more times, the same or substituted differently with halogen, oxo, Ci-Cö-alkyl or phenoxy.
- R 12 is particularly preferably Ci-Cö-alkyl or Cs-Cs-cycloalkyl.
- R 12 is very particularly preferably methyl or tert. Butyl.
- inventive compounds are present.
- the circle should represent both as possible position double bonds, as shown here:
- Alkyl is a linear or branched, saturated, monovalent hydrocarbon radical having generally 1 to 6 (C 1 -C 6 -alkyl), preferably 1 to 4 (C 1 -C 4 -alkyl), and particularly preferably 1 to 3 carbon atoms (C 1 -C 4 -alkyl) C3 alkyl).
- Particularly preferred is a methyl, ethyl, propyl, isopropyl or tert-butyl radical.
- Cycloalkyl is a monocyclic, saturated, monovalent hydrocarbon radical having generally 3 to 10 (C 3 -C 10 -cycloalkyl), preferably 3 to 8 (C 5 -C 8 -cycloalkyl), and particularly preferably 3 to 7 (C 3 -C 7 -cycloalkyl) carbon atoms.
- Alkoxy represents a linear or branched, saturated alkyl ether radical of the formula -O-alkyl having generally 1 to 6 (CI-C ⁇ - alkoxy), preferably 1 to 4 (Ci-C t-alkoxy), and particularly preferably 1 to 3 (C 1 -C 3 -alkoxy) carbon atoms.
- Alkoxyalkyl is an alkoxy-substituted alkyl radical, for example Ci-Ce-alkoxy-Ci-Cö-alkyl or Ci-Cs-alkoxy-Ci-Cs-alkyl.
- Ci-Ce-alkoxy-Ci-Cö-alkyl- means that the alkoxyalkyl group is bonded via the alkyl moiety to the rest of the molecule.
- Oxo may be attached to atoms of suitable valence, for example to a saturated carbon atom or to sulfur.
- the bond to carbon is to form a carbonyl group or bond to sulfur to form a sulfinyl or sulfonyl group.
- Alkylamino is an amino radical having one or two (independently selected)
- Alkyl substituents with generally 1 to 6 (C 1 -C 6 -alkylamino), preferably 1 to 3 carbon atoms (C 1 -C 3 -alkylamino).
- (C 1 -C 3) -Alkylamino is, for example, a monoalkylamino radical having 1 to 3 carbon atoms or a dialkylamino radical having in each case 1 to 3 carbon atoms per alkyl substituent. Examples include:
- Examples include acetyl and propanoyl.
- alkylsulfonyl preferably 1 to 4, and particularly preferably 1 to 3 carbon atoms in the alkyl part.
- Methylsulfonyl ethylsulfonyl, propylsulfonyl.
- Methylaminosulfonyl ethylaminosulfonyl, dimethylaminosulfonyl.
- phenyl-Ci-Cö-alkyl- is meant a group which is composed of a
- alkyl radical here has the meanings given above under alkyl.
- benzyl examples which may be mentioned are benzyl, phenethyl, phenylpropyl, phenylpentyl, benzyl being preferred.
- phenyl-O-C6-alkoxy- is meant a group which is composed of an optionally substituted phenyl radical and a Ci-Cö-alkoxy group, and bound via the Ci-Cö-alkoxy group to the rest of the molecule is.
- the alkoxy radical here has the meanings given above under alkoxy.
- benzoxy examples which may be mentioned are benzoxy, phenethoxy, phenylpropyloxy, phenylpentyloxy, benzoxy being preferred.
- Examples include phenylsulfonyl, o- or p-toluylsulfonyl, m-chlorophenylsulfonyl. heteroatoms
- Heteroatoms are oxygen, nitrogen or sulfur atoms.
- heteroaryl
- Heteroaryl means a monovalent aromatic ring system having 1, 2, 3, 4, 5 or 6 heteroatoms. As heteroatoms nitrogen atoms, oxygen atoms and / or sulfur atoms may occur.
- the binding valency can be attached to any aromatic carbon atom or to a
- a monocyclic heteroaryl group according to the present invention has 5 or 6 ring atoms.
- heteroaryl radicals having 5 ring atoms include the rings:
- Heteroaryl radicals having 6 ring atoms include, for example, the rings:
- a bicyclic heteroaryl group according to the present invention has 9 or 10 ring atoms.
- heteroaryl radicals having 9 ring atoms include the rings:
- Heteroaryl radicals with 10 ring atoms include, for example, the rings:
- a partially saturated bicyclic aryl radical or heteroaryl radical represents a bicyclic group consisting of a phenyl radical or a monocyclic, 5- or 6-membered heteroaryl radical which is fused via two immediately adjacent ring atoms to an aliphatic cyclic radical having 4 to 7 ring atoms, respectively optionally contain one or two heteroatoms, which may be the same or different.
- heteroatoms nitrogen atoms, oxygen atoms and / or sulfur atoms may occur.
- Partially saturated bicyclic aryl radicals include, for example, the groups: Tetrahydronaphthyl, 2,3-dihydro-l, 4-benzodioxinyl, 2,3-dihydro-l-benzofuranyl and 1,3-benzodioxolyl.
- Partially saturated bicyclic heteroaryl radicals include, for example, the groups:
- Monocyclic heterocyclyl means a non-aromatic monocyclic ring system having 1, 2, 3, 4, 5 or 6 heteroatoms. As heteroatoms nitrogen atoms, oxygen atoms and / or sulfur atoms may occur.
- a monocyclic heterocyclyl ring according to the present invention may have 4 to 8, preferably 4 to 7, more preferably 5 or 6 ring atoms.
- Exemplary and preferred for monocyclic heterocyclyl radicals having 4 ring atoms are: azetidinyl, oxetanyl.
- Exemplary and preferred for monocyclic heterocyclyl radicals having 5 ring atoms are: pyrrolidinyl, imidazolidinyl, pyrazolidinyl, pyrrolinyl, dioxolanyl and tetrahydrofuranyl.
- Exemplary and preferred for monocyclic heterocyclyl radicals having 6 ring atoms are: piperidinyl, piperazinyl, morpholinyl, dioxanyl, tetrahydropyranyl and thiomorpholinyl
- Exemplary and preferred for monocyclic heterocyclyl radicals having 7 ring atoms are: azepanyl, oxepanyl, 1,3-diazepanyl, 1,4-diazepanyl.
- monocyclic heterocyclyl radicals having 8 ring atoms are: oxocanyl, azocanyl.
- monocyclic heterocyclyl radicals are 3- to 8-membered, preferably 4 to 7-membered, more preferably 5 to 6-membered saturated heterocyclyl radicals having up to two heteroatoms from the series O, N and S.
- Carbon atoms through heteroatoms as defined above in any combination is one To understand fusion of two saturated ring systems, which share two atoms directly adjacent to each other. Examples are bicyclo [2.2.0] hexyl, bicyclo [3.3.0] octyl, bicyclo [4.4.0] decyl, bicyclo [5.4.0] undecyl, bicyclo [3.2.0] heptyl, bicyclo [4.2 .0] octyl, bicyclo [5.2.0] nonyl,
- halogen includes fluorine, chlorine, bromine and iodine.
- fluorine, chlorine and bromine Preference is given to fluorine, chlorine and bromine, in particular fluorine or chlorine.
- Haloalkyl is an alkyl radical having at least one halogen substituent.
- a halo-C 1 -C 6 -alkyl radical is an alkyl radical having 1-6 carbon atoms and at least one halogen substituent. If several halogen substituents are present, they may also be different.
- Trifluoromethyl 2,2,2-trifluoroethyl, pentafluoroethyl, 4,4,5,5,5-pentafluoropentyl or 3,3,4,4,5,5,5-heptafluoropentyl group.
- perfluorinated alkyl radicals such as trifluoromethyl or pentafluoroethyl.
- Haloalkoxy is an alkoxy radical having at least one halogen substituent.
- a halogeno-C 1 -C 6 -alkoxy radical is an alkoxy radical having 1-6 carbon atoms and at least one halogen substituent. If several halogen substituents are present, they may also be different. Preferred are fluoroalkoxy radicals.
- Hydroxyalkyl is an alkyl radical having at least one hydroxy substituent.
- a hydroxy-C 1 -C 6 -alkyl radical is an alkyl radical having 1-6 carbon atoms and at least one Hydroxy substituent.
- Aminoalkyl is an alkyl radical having at least one amino substituent.
- An amino-Ci-Cö-alkyl radical is an alkyl radical consisting of 1-6 carbon atoms and at least one amino substituent.
- Alkylaminoalkyl- represents an alkyl radical substituted with alkylamino as defined above, for example C 1 -C 6 -alkylamino-C 1 -C 6 -alkyl- or C 1 -C 3 -alkylamino-C 1 -C 3 -alkyl-.
- Ci-Ce-alkylamino-Ci-Cö-alkyl- means that the alkylaminoalkyl group is bonded via the alkyl moiety to the rest of the molecule.
- Di-alkylamino-alkyl for example di-C 1 -C 3 -alkylamino-C 1 -C 3 -alkyl-, means that the abovementioned alkylamino part obligatorily contains two alkyl groups, which may be identical or different.
- alkylaminoalkyl examples include A, N-dimethylaminoethyl, A, N-dimethylaminomethyl, N, N-diethylaminoethyl, A, N-dimethylaminopropyl, N-methylaminoethyl, N-methylaminomethyl.
- radical definitions specified in detail in the respective combinations or preferred combinations of radicals are also replaced, irrespective of the particular combinations of radicals indicated, by any definitions of radicals of other combinations.
- Compounds according to the invention are the compounds of the formula (I) and their salts, solvates and solvates of the salts comprising the compounds of the formulas below and their salts, solvates and solvates of the salts and of the formula (I) encompassed by formula (I), hereinafter referred to as exemplary compounds and their salts, solvates and solvates of the salts, as far as the compounds of formula (I), the compounds mentioned below are not already salts, solvates and solvates of the salts.
- Salts used in the context of the present invention are physiologically acceptable salts of the compounds according to the invention. However, also included are salts which are not suitable for pharmaceutical applications themselves but can be used, for example, for the isolation or purification of the compounds according to the invention.
- Physiologically acceptable salts of the compounds of the invention include acid addition salts of mineral acids, carboxylic acids and sulfonic acids, e.g. Salts of hydrochloric, hydrobromic, sulfuric, phosphoric, methanesulfonic, ethanesulfonic,
- Physiologically acceptable salts of the compounds of the invention further include base addition salts of, for example, alkali metals such as sodium or potassium, alkaline earth metals such as calcium or magnesium, or ammonium salts derived from ammonia or organic amines containing from 1 to 16 carbon atoms, such as Example methylamine, ethylamine, diethylamine, triethylamine, ethyldiisopropylamine, monoethanolamine, diethanolamine, Triethanolamine, dicyclohexylamine, dimethylaminoethanol, procaine, dibenzylamine, N-methylmorpholine, arginine, lysine, ethylenediamine, N-methylpiperidine, N-methylglucamine, dimethylglucamine, ethylglucamine, 1,6-hexadiamine, glucosamine, sarcosine, serinol,
- alkali metals such as sodium or potassium
- alkaline earth metals such
- the compounds according to the invention can form base addition salts with quaternary ammonium ions, which are obtained, for example, by quanitization of corresponding amines with etches such as lower alkyl halides, for example methyl, ethyl, propyl and
- Examples of such quaternary ammonium ions are tetramethylammonium, tetraethylammonium, tetra (w-propyl) ammonium, tetra (w-butyl) ammonium, and
- Another object of the present invention are all possible crystalline and polymorphic forms of the compounds of the invention, wherein the polymorphs can be present either as a single polymorph or as a mixture of several polymorphs in all concentration ranges.
- Another object of the present invention are pharmaceutical compositions containing the compounds of the invention and at least one or more other active ingredients, in particular for the prophylaxis and / or therapy of tumor diseases.
- Solvates in the context of the invention are those forms of the compounds according to the invention which form a complex in the solid or liquid state by coordination with solvent molecules. Hydrates are a special form of solvates that coordinate with water. As solvates, hydrates are preferred in the context of the present invention.
- the compounds according to the invention may exist in different stereoisomeric forms, ie in the form of configurational isomers or optionally also as conformational isomers.
- the compounds according to the invention have an asymmetric center on the carbon atom of the diazepine skeleton (C-4) which is bonded via Y - (CH 2 ) P - to Y. They can therefore be present as pure enantiomers, racemates but also as diastereomers or mixtures thereof, if one or more of the substituents described in the formula (I) another
- Asymmetrieelement contains, for example, a chiral carbon atom.
- the present invention therefore also encompasses enantiomers and diastereomers and their respective mixtures. From such mixtures, the pure enantiomers and diastereomers can be isolated in a known manner; Preferably, for this purpose, chromatographic methods are used, in particular HPLC chromatography on chiral or achiral phase.
- the enantiomers according to the invention inhibit the target to different degrees and are active in different ways in the cancer cell lines investigated.
- the present invention encompasses all tautomeric forms.
- the present invention also includes all suitable isotopic variants of the compounds of the invention.
- An isotopic variant of a compound according to the invention is understood to mean a compound in which at least one atom within the compound according to the invention is exchanged for another atom of the same atomic number but with a different atomic mass than the atomic mass that usually or predominantly occurs in nature.
- isotopes which can be incorporated into a compound of the invention are those of hydrogen, carbon, nitrogen, oxygen, phosphorus, sulfur, fluorine, chlorine, bromine and iodine, such as 2 H (deuterium), 3 H (tritium), 13 C, 14 C, 15 N, 17 0, 18 0, 32 P, 33 P, 33 S, M S, 35 S, 36 S, 18 F, 36 Cl, 82 Br, 123 1, 124 1, 129 I and 131 I.
- Certain isotopic variants of a compound of the invention may be useful, for example, to study the mechanism of action or distribution of drug in the body; Due to the comparatively easy production and detectability, compounds labeled with 3 H or 14 C isotopes in particular are suitable for this purpose.
- the incorporation of isotopes such as deuterium may result in certain therapeutic benefits as a result of greater metabolic stability of the compound, such as prolonging the body's half-life or reducing the required effective dose;
- Such modifications of the compounds of the invention may therefore optionally also constitute a preferred embodiment of the present invention.
- Isotopic variants of the compounds according to the invention can be prepared by the processes known to the person skilled in the art, for example by the methods described below and the rules given in the exemplary embodiments, by using appropriate isotopic modifications of the respective reagents and / or starting compounds.
- the present invention also includes prodrugs of the compounds of the invention.
- prodrugs includes compounds which may themselves be biologically active or inactive, but which are converted during their residence time in the body into compounds of the invention (for example metabolically or hydrolytically).
- the compounds according to the invention can act systemically and / or locally. For this purpose, it may be applied in a suitable manner, such as, for example, orally, parenterally, pulmonarily, nasally, sublingually, lingually, buccally, rectally, dermally, transdermally, conjunctivally, otically or as an implant or stent.
- the compounds of the invention can be used in suitable manner, such as, for example, orally, parenterally, pulmonarily, nasally, sublingually, lingually, buccally, rectally, dermally, transdermally, conjunctivally, otically or as an implant or stent.
- the compounds of the invention can be used in suitable manner, such as, for example, or
- Administration forms are administered.
- Parenteral administration can be done by bypassing a resorption step (e.g.
- intravenously, intraarterially, intracardially, intraspinally or intralumbarally) or with the involvement of resorption e.g., intramuscularly, subcutaneously, intracutaneously, percutaneously or intraperitoneally.
- parenteral administration are suitable as application forms u.a. Injection and
- Infusion preparations in the form of solutions, suspensions, emulsions, lyophilisates or sterile powders.
- Inhalation medicines including powder inhalers, nebulizers
- nasal drops solutions, sprays
- lingual, sublingual or buccal tablets to be applied, films / wafers or capsules, suppositories, ear or eye preparations
- Vaginal capsules aqueous suspensions (lotions, shake mixtures), lipophilic suspensions, ointments, creams, transdermal therapeutic systems (such as patches), milk, pastes, foams, powdered powders, implants or stents.
- the compounds according to the invention can be converted into the stated administration forms. This can be done in a conventional manner by mixing with inert, non-toxic, pharmaceutically suitable excipients.
- adjuvants include, among others.
- Carriers e.g., microcrystalline cellulose, lactose, mannitol
- solvents e.g., liquid
- Polyethylene glycols Polyethylene glycols
- emulsifiers for example, polyethylene glycols
- dispersing or wetting agents for example, polyethylene glycols, emulsifiers and dispersing or wetting agents
- binders for example, polyvinylpyrrolidone
- synthetic and natural polymers for example, albumin
- stabilizers eg, antioxidants such as ascorbic acid
- dyes eg, inorganic pigments such as iron oxides
- flavor and / or odor remedies for example, sodium dodecylsulfate, polyoxysorbitanoleate
- binders for example, polyvinylpyrrolidone
- synthetic and natural polymers for example, albumin
- stabilizers eg, antioxidants such as ascorbic acid
- dyes eg, inorganic pigments such as iron oxides
- compositions containing the compounds of the invention usually together with one or more inert, non-toxic, pharmaceutically suitable excipients, and their use for the purposes mentioned above.
- the formulation of the compounds according to the invention into pharmaceutical preparations is carried out in a manner known per se by converting the active substance (s) into the desired administration form with the auxiliaries customary in galenicals.
- excipients examples include excipients, fillers, disintegrants, binders, humectants, lubricants, ab- and adsorbents, diluents, solvents, cosolvents, emulsifiers, solubilizers, flavoring agents, colorants,
- Preservatives, stabilizers, wetting agents, salts for changing the osmotic pressure or buffers are used. Reference may be made to Remington's Pharmaceutical Science, 15th ed. Mack Publishing Company, East Pennsylvania (1980).
- the pharmaceutical formulations can be used.
- auxiliaries for the purposes of the invention may be, for example, salts, saccharides (mono-, di-, tri-, oligo-, and / or polysaccharides), proteins, amino acids, peptides, fats, waxes, oils, hydrocarbons and derivatives thereof, the auxiliaries may be of natural origin or synthetically or partially synthetically obtained.
- the present invention relates to the compounds of the invention.
- the compounds of the invention can be used for the prophylaxis and treatment of human diseases, in particular tumors.
- the compounds of the invention can be used in particular to the
- the compounds of the invention are particularly suitable for the prophylaxis and / or treatment of hyper-proliferative diseases such as
- BPH benign prostate hyperplasia
- tumors according to the invention for example, tumors of the breast, tumors of the breast, tumors of the breast, tumors of the breast, tumors of the breast, tumors of the breast, tumors of the breast, tumors of the breast, tumors of the breast, tumors of the breast, tumors of the breast, tumors of the breast, tumors of the breast, tumors of the breast, tumors of the breast, tumors of the breast, tumors of the breast, tumors of the breast,
- Genitourinary tract eye, liver, skin, head and neck, thyroid, parathyroid gland, bone, connective tissue and metastases of these tumors.
- hematological tumors are treatable
- treatable as breast tumors are:
- tumors of the respiratory tract are treatable.
- non-small cell lung carcinomas such as squamous cell carcinoma, adenocarcinoma, large cell carcinoma and
- tumors of the brain are treatable.
- the tumors of the male reproductive organs are treatable: prostate carcinomas,
- Penis cancer For example, tumors of the female reproductive organs are treatable:
- tumors of the gastrointestinal tract are treatable:
- tumors of the urogenital tract are treatable:
- tumors of the eye are treatable:
- Intraocular melanomas For example, tumors of the liver are treatable:
- tumors of the skin are treatable:
- tumors of the head and neck are treatable:
- Carcinomas of the midline structures (such as NMC, CA French, Annu Rev. Pathol., 2012, 7: 247-265)
- sarcomas are treatable:
- lymphomas are treatable:
- Treatable as leukemias for example:
- the compounds according to the invention can be used for the prophylaxis and / or therapy of leukemias, in particular acute myeolemic leukemias, prostate carcinomas, in particular androgen receptor-positive prostate carcinomas, cervical carcinomas, breast cancers, in particular of hormone receptor-negative, hormone receptor-positive or BRCA-associated breast carcinomas, pancreatic carcinomas, renal cell carcinomas,
- leukemias in particular acute myeolemic leukemias
- prostate carcinomas in particular androgen receptor-positive prostate carcinomas
- cervical carcinomas cervical carcinomas
- breast cancers in particular of hormone receptor-negative, hormone receptor-positive or BRCA-associated breast carcinomas, pancreatic carcinomas, renal cell carcinomas,
- Prophylaxis and / or therapy of leukemias in particular acute myeloid leukemias, prostate carcinomas, in particular androgen receptor-positive prostate carcinomas,
- Breast cancer in particular estrogen receptor-alpha negative breast carcinoma, melanoma or multiple myeloma.
- the compounds according to the invention are also suitable for the prophylaxis and / or therapy of benign hyperproliferative diseases such as, for example, endometriosis, leiomyoma and benign prostatic hyperplasia.
- the compounds according to the invention are also suitable for the fertility control of the man.
- the compounds according to the invention are also suitable for the prophylaxis and / or therapy of systemic inflammatory diseases, in particular LPS-induced endotoxic shock and / or bacteria-induced sepsis.
- the compounds according to the invention are also suitable for the prophylaxis and / or therapy of inflammatory or autoimmune diseases such as, for example:
- Lung diseases especially allergic alveolitis; all forms of pulmonary edema, especially toxic pulmonary edema; Sarcoidoses and granulomatoses, in particular Boeck's disease - Rheumatic diseases / autoimmune diseases / joint diseases with
- inflammatory, allergic and / or proliferative processes all forms of rheumatic diseases, in particular rheumatoid arthritis, acute rheumatic fever, polymyalgia rheumatica; reactive arthritis; Inflammatory soft tissue diseases of other origin; arthritic symptoms of degenerative joint disease (arthrosis); traumatic arthritis; Collagenosis of any genesis, eg systemic lupus erythematosus, scleroderma, polymyositis, dermatomyositis, Sjögren's syndrome, Still's syndrome, Felty syndrome Allergies associated with inflammatory and / or proliferative processes: all forms of allergic reactions, eg, Quincke's edema, hay fever, insect bites, allergic reactions to drugs, blood derivatives, contrast agents, etc., anaphylactic shock, urticaria, contact dermatitis
- Vascular damage vacular damage (vasculature): Panarteritis nodosa, temporal arteritis, erythema nodosum
- Dermatological disorders associated with inflammatory, allergic and / or proliferative processes atopic dermatitis; Psoriasis; Pityriasis rubra pilaris; erythematous diseases induced by different noxae, e.g.
- Liver diseases associated with inflammatory, allergic and / or proliferative processes acute liver cell decay; acute hepatitis of different causes, e.g. viral, toxic, drug-induced; Chronic aggressive and / or chronic intermittent hepatitis
- Gastrointestinal disorders associated with inflammatory, allergic and / or proliferative processes regional enteritis (Crohn's disease); Ulcerative colitis; Gastritis; reflux esophagitis; Gastroenteritides of other genesis, e.g. native sprue
- Proctological diseases associated with inflammatory, allergic and / or proliferative processes analgesic; fissures; Hemorrhoids; idiopathic proctitis Eye diseases associated with inflammatory, allergic and / or proliferative processes: allergic keratitis, uveitis, ulceris; conjunctivitis; blepharitis; Neuritis nervi optici; Chlorioditis; Opthalmia sympathica
- Neurological diseases associated with inflammatory, allergic and / or proliferative processes brain edema, especially tumor-related cerebral edema; Multiple sclerosis; acute encephalomyelitis; Meningitis; various forms of seizures, e.g. BNS cramps
- Blood disorders associated with inflammatory, allergic and / or proliferative processes acquired hemolytic anemia; idiopathic thrombocytopenia
- Tumor diseases associated with inflammatory, allergic and / or proliferative processes acute lymphoblastic leukemia; malignant lymphomas; Lymphogranulomatosen; lymphosarcoma; extensive metastases, especially in mammary, bronchial and prostate carcinoma
- Endocrine disorders associated with inflammatory, allergic and / or proliferative processes endocrine orbitopathy; thyrotoxic crisis; Thyreoditis de Quervain; Hashimoto's thyroiditis; Graves' disease
- Severe states of shock e.g. anaphylactic shock, systemic inflammatory response syndrome (SIRS)
- SIRS systemic inflammatory response syndrome
- Emesis associated with inflammatory, allergic and / or proliferative processes e.g. in combination with a 5-HT3 antagonist in cytostatic vomiting
- Pain of inflammatory genesis e.g. lumbago
- the compounds according to the invention are also suitable for the treatment of viral
- Diseases such as infections caused by papilloma viruses, herpes viruses, Epstein-Barr viruses, hepatitis B or C viruses, and human immunodeficiency viruses.
- the compounds of the invention are also useful in the treatment of atherosclerosis, dyslipidemia, hypercholesterolemia, hypertriglyceridemia, periferous vascular diseases, cardiovascular diseases, angina, ischemia, stroke, myocardial infarction, angioplasty restenosis, hypertension, thrombosis, obesity, endotoxemia.
- the compounds of the invention are also useful in the treatment of neurodegenerative diseases such as multiple sclerosis, Alzheimer's disease and Parkinson's disease.
- Another object of the present application are the compounds of the invention for use as medicaments, in particular for the prophylaxis and / or therapy of
- a further subject of the present application is the compound according to the invention prophylaxis and / or therapy of leukemias, in particular acute myeolemic leukemias, Prostate carcinomas, in particular androgen receptor-positive prostate carcinomas, cervical carcinomas, breast cancers, in particular of hormone receptor-negative,
- Hormone receptor-positive or BRCA-associated breast cancers pancreatic carcinomas, renal cell carcinomas, hepatocellular carcinomas, melanomas and other skin tumors, non-small cell lung carcinomas, endometrial carcinomas and colorectal carcinomas.
- a further subject of the present application are the compounds according to the invention for the prophylaxis and / or therapy of leukemias, in particular acute myeloid leukemias, prostate carcinomas, in particular androgen receptor-positive prostate carcinomas,
- Breast cancer in particular estrogen receptor-alpha negative breast carcinoma, melanoma or multiple myeloma.
- Another object of the invention is the use of the compounds of the invention for the manufacture of a medicament.
- Another object of the present application is the use of the compounds of the invention for the manufacture of a medicament for the prophylaxis and / or therapy of
- Another object of the present application is the use of the invention
- leukemias in particular acute myeolemic leukemias, prostate carcinomas, in particular androgen receptor-positive prostate carcinomas, cervical carcinomas, breast cancers, in particular of
- Hormone receptor-negative, hormone receptor-positive or BRCA-associated breast carcinomas pancreatic carcinomas, renal cell carcinomas, hepatocellular carcinomas, melanomas and other skin tumors, non-small cell lung cancers, endometrial carcinomas and colorectal carcinomas.
- Another object of the present application is the use of the compounds of the invention for the manufacture of a medicament for the prophylaxis and / or therapy of leukemias, especially acute myeloid leukemias, prostate cancer, especially androgen receptor-positive prostate cancer, breast cancer, in particular estrogen receptor-negative alpha breast carcinomas, melanomas or multiple myelomas.
- leukemias especially acute myeloid leukemias, prostate cancer, especially androgen receptor-positive prostate cancer, breast cancer, in particular estrogen receptor-negative alpha breast carcinomas, melanomas or multiple myelomas.
- Another object of the present application is the use of the compounds of the invention for the prophylaxis and / or therapy of tumor diseases.
- the present application further relates to the use of the compounds according to the invention for the prophylaxis and / or therapy of leukemias, in particular acute myeolemic leukemias, prostate carcinomas, in particular androgen receptor-positive prostate carcinomas, cervical carcinomas, breast cancers, in particular of hormone receptor negative,
- leukemias in particular acute myeolemic leukemias, prostate carcinomas, in particular androgen receptor-positive prostate carcinomas, cervical carcinomas, breast cancers, in particular of hormone receptor negative,
- Hormone receptor-positive or BRCA-associated breast carcinoma pancreatic carcinoma
- Renal cell carcinoma hepatocellular carcinoma, melanoma and other skin tumors, non-small cell lung carcinoma, endometrial carcinoma and colorectal carcinoma.
- a further subject of the present application is the use of the compounds according to the invention for the prophylaxis and / or therapy of leukemias, in particular acute myeloid leukemias, prostate carcinomas, in particular androgen receptor-positive prostate carcinomas, breast cancers, in particular estrogen receptor-alpha negative breast carcinomas, melanomas or multiple myelomas.
- a further subject of the present application are pharmaceutical formulations in the form of tablets containing one of the compounds according to the invention for the prophylaxis and / or therapy of leukemias, in particular acute myeolemic leukemias, prostate carcinomas, in particular androgen receptor-positive prostate carcinomas, cervical carcinomas,
- breast cancer in particular hormone receptor-negative, hormone receptor-positive or BRCA-associated breast carcinoma, pancreatic carcinoma, renal cell carcinoma,
- Another object of the present application are pharmaceutical formulations in the form of tablets containing one of the compounds of the invention for the prophylaxis and / or treatment of leukemias, especially acute myeloid leukemias, prostate cancer, especially androgen receptor-positive prostate cancer, breast cancer, especially estrogen receptor-alpha negative breast carcinoma, melanoma or multiple myeloma.
- leukemias especially acute myeloid leukemias
- prostate cancer especially androgen receptor-positive prostate cancer
- breast cancer especially estrogen receptor-alpha negative breast carcinoma
- melanoma or multiple myeloma are also used for the treatment of diseases associated with proliferative processes.
- Another object of the invention is the use of the compounds of the invention for the treatment of benign hyperplasia, inflammatory diseases, autoimmune
- the compounds of the invention may be used alone or as needed in combination with one or more other pharmacologically active substances, as long as they are
- Another object of the present invention are therefore pharmaceutical compositions containing a compound of the invention and one or more other active ingredients, in particular for the prophylaxis and / or therapy of the aforementioned diseases.
- the compounds according to the invention can be combined with known anti-hyperproliferative, cytostatic or cytotoxic substances for the treatment of cancers.
- the combination of the compounds according to the invention with other substances commonly used for cancer therapy or also with radiotherapy is particularly indicated.
- Affinitak e.g., aldesleukin, alendronic acid, alfaferone, alitretinoin, allopurinol, aloprim, aloxi, alpharadin, altretamine, aminoglutethimide, aminopterin, amifostine, amrubicin, amsacrine,
- Anastrozole Anzmet, Apatinib, Aranesp, Arglabin, Arsenic trioxide, Aromasin, Arzoxifen, Asoprisnil, L-asparaginase, Atamestan, Atrasentan, Avastin, Axitinib, 5-Azacytidine, Azathioprine, BCG or tice- BCG, Bendamustine, Bestatin, Beta methasone acetate, betamethasone sodium phosphate, bexarotene, bicalutamide, bleomycin sulfate, broxuridine, bortezomib, bosutinib, busulfan, cabazitaxel,
- Daunorubicin DaunoXome, Decadron, Decadron Phosphate, Decitabine, Degarelix, Delestrogen, Denileukin Diftitox, Depomedrol, Deslorelin, Dexrazoxane, Diethylstilbestrol, Diflucan, 2 ', 2'-Difluorodeoxycytidine, DN-101, Docetaxel, Doxifluridine, Doxorubicin (adriamycin), Dronabinol, dSLIM, Dutasteride, DW-166HC, Edotecarin, Efl Ornithine, Eligard, Elitek, Ellence, Emend,
- Enzalutamide Epirubicin, Epoetin-alfa, Epogen, Epothilone and its derivatives, Eptaplatin, Ergamisol, Erlotinib, Erythro-hydroxynonyladenine, Estrace, Estradiol, Estramustine sodium phosphate,
- Leuprolide leuprolide acetate, levamisole, levofolic acid calcium salt, levothroid, levoxyl, libra, liposomal MTP-PE, lomustine, lonafarnib, lonidamine, marinol, mechlorethamine, mecobalamin, medroxyprogesterone acetate, megestrol acetate, melphalan, menest, 6-mercaptopurine , Mesna, Methotrexate, Metvix, Miltefosine, Minocycline, Minodronate, Miproxifen, Mitomycin C, Mitotane, Mitoxantrone, Modrenal, MS-209, MX-6, Myocet, Nafarelin, Nedaplatin, Nelarabine, Nemorubicin, Neovastat, Neratinib, Neulasta, Neumega, Neupogen, Nilotimibe, Nilutamide, Nimustin, Nola
- the compounds of this invention may also be treated with biological therapeutics such as antibodies (e.g., aflibercept, alemtuzumab, bevacizumab, brentuximumab, catumaxomab, cetuximab, denosumab, edrecolomab, gemtuzumab, ibritumomab, ipilimumab,
- biological therapeutics such as antibodies (e.g., aflibercept, alemtuzumab, bevacizumab, brentuximumab, catumaxomab, cetuximab, denosumab, edrecolomab, gemtuzumab, ibritumomab, ipilimumab,
- panitumumab panitumumab, pertuzumab, rituximab, tositumumab, trastuzumab
- panitumumab panitumumab, pertuzumab, rituximab, tositumumab, trastuzumab
- recombinant combine th proteins.
- the compounds of the invention may also be used in combination with others, against the
- Angiogenesis-targeted therapies such as bevacizumab, axitinib, regorafenib, cediranib, sorafenib, sunitinib or thalidomide. Combinations with antihormones and steroidal metabolic enzyme inhibitors are because of their favorable
- the compounds of the invention may also be used in conjunction with radiotherapy and / or surgical intervention.
- R 1 , R 2 , R 3 , R 4 and R 5 and n and m here have the meanings given under the general formula I.
- the execution of the reaction sequence a) and b) for ring closure of the pyrrole is a sequence known to the skilled person (II Farmaco, Ediette Scientifica (1984), 39, p 538ff, Tarzia et al.).
- alkyl substituents R 2 according to general formula (I) can be introduced by methods known to the person skilled in the art.
- acyl halides or acyl anhydrides or aryl and alkylsulfonyl chlorides By reaction with acyl or aryl or alkylsulfonyl substituents can be introduced as R 2 according to general formula (I) by methods known to the person skilled in the art.
- Aryl and heteroaryl radicals as R 2 can by reaction with the corresponding aryl or
- LG is to be understood as a leaving group, which, as described here, for example, a halogen or a boronic acid may be.
- 4-aminopyrazolo-acetophenones can be prepared according to a reaction sequence shown in Scheme 2.
- R 1 , R 2 , R 3 , R 4 and R 5 and n and m here have the meanings given under the general formula I.
- R 2 is halogen, K 2 C0 3 , DMF;
- ArR R 4 R 5 , AlCb;
- Fe Fe, NH 4 Cl, water, EtOH
- alkyl halides or alkyl sulfates in step a) alkyl substituents R 2 according to general formula (I) can be introduced by methods known to those skilled in the art. By reaction with acyl halides or acyl anhydrides or aryl and Alkylsulfonylchloriden acyl or aryl or alkylsulfonyl substituents as R 2 according to general formula (I) by the person skilled in the art known methods are introduced. Aryl and heteroaryl radicals as R 2 can be introduced by reaction with the corresponding aryl or heteroaryl halides and a transition metal catalyst of palladium or copper (J. Am. Chem. Soc. (1998), 120, pp.
- LG is to be understood as a leaving group, which, as described here, for example, a halogen or a boronic acid may be.
- R 1 , R 2 , R 3 , R 4 and R 5 and n and m have the meanings given under the general formula I.
- R 1 , R 2 , R 3 , R 4 , R 5 , R 12 and X and n, m and p have the meanings given under the general formula I.
- the circle means a presence of possible double bond isomers in the case of X is nitrogen, as shown here: a) eg HATU, FMOC-ASP (OR 12 ) -OH; b) eg piperidine, RT, then HOAc excess The coupling a) is shown here with HATU, but can also be carried out under other conditions.
- step b a strong acid such as trifluoroacetic acid or hydrochloric acid is used.
- R 1 , R 2 , R 3 , R 4 , R 5 , R 12 and X and n, m and p here have the meanings given under the general formula I.
- Y stands for -C (O) OR 12 as defined for the general formula I.
- the cycle means a presence of possible double bond isomers in the case of X equal to nitrogen, as shown here:
- alkyl groups are methyl, ethyl or longer homologous esters.
- Reactions can be preferably carried out using alkali hydroxides such as lithium hydroxide, sodium hydroxide or potassium hydroxide in aqueous alcoholic solutions.
- alkali hydroxides such as lithium hydroxide, sodium hydroxide or potassium hydroxide in aqueous alcoholic solutions.
- Branched alkyl groups such as tert-butyl esters can preferably be hydrolyzed under acidic conditions.
- a variety of methods are known to those skilled in the art. By way of example only the use of e.g. HCl in organic solvents or pure or diluted
- amides of the general formula I according to the invention are thus prepared by reacting the carboxylic acids, for example with the amines generally available commercially in the working examples, with additional activation by a method which is generally known to the person skilled in the art.
- a method which is generally known to the person skilled in the art.
- HATU, HBTU, PyBOB or T3P with addition of a suitable base.
- Carboxylic acids in their amides are generally described in reference books such as Compendium of Organic Synthetic Methods, Volume I-VI (Wiley Interscience) or The Practice of Peptides
- R 1 , R 2 , R 3 , R 4 , R 5 , R 12 , X and Y and n, m and p have the meanings given under the general formula I.
- Y stands for -C (O) R 13 as defined for the general formula I.
- the cycle means a presence of possible double bond isomers in the case of X equal to nitrogen, as shown here:
- step b) the coupling to a Weinreb amide known to the skilled worker is carried out in step b) by reaction with ⁇ , ⁇ -dimethylhydroxylamine.
- step c) is then, for example, with a known for those skilled alkylmagnesium (Grignard) or Alkyllithium reagent to compounds of general formula (I) implemented.
- alkylmagnesium or alkyllithium reagents are well known to those skilled in the art and can be carried out starting from corresponding alkyl halides such as iodides, bromides or chlorides with, for example, the elemental metal, for example magnesium or lithium, or by reaction with a correspondingly reactive magnesium or lithium -Alkylreagenz such as di-iso-propyl magnesium or butyllithium.
- corresponding alkyl halides such as iodides, bromides or chlorides with, for example, the elemental metal, for example magnesium or lithium
- a correspondingly reactive magnesium or lithium -Alreagenz such as di-iso-propyl magnesium or butyllithium.
- R 1 , R 2 , R 3 , R 4 , R 5 and X and n, m and p have the meanings given under the general formula I.
- Y stands for phenyl or heteroaryl as defined for the general formula I.
- the cycle means a presence of possible double bond isomers in the case of X is nitrogen, as shown here:
- Ring atoms are also prepared as described in Scheme 8, Part A, and Part B:
- R 1 , R 2 , R 3 , R 4 , R 5 , R 12 and X and n, m and p here have the meanings given under the general formula I.
- Y is heteroaryl having 5 ring atoms as defined for the general formula I.
- the circle means a presence of possible double bond isomers in the case of X is nitrogen, as shown here:
- reagents known in the art for the separation of water such as, for example, 1,3,4-oxadiazoles (J. Med. Chem. (2005), 48, p4068ff Garcia et al.) Or, using Lawesson's reagent or phosphorous pentasulfide, to 1,3,4-thiadiazoles (Eur. J. Med. Chem. (2010), 45, p4664ff Kumar et al.), Optionally can carry various other substituents.
- NMR signals are given with their respective recognizable multiplicity or their combinations.
- s singlet
- d doublet
- t triplet
- q quartet
- qi quintet
- m Multiplet
- b wide signal.
- the mother liquor which contained still further product, was purified by chromatography on silica gel, whereby an additional 3.2 g of the desired ieri-butyl- Ar - [4- (4-chlorobenzoyl) -l, 5-dimethyl-1/1-pyrrole-3 -yl] carbamate.
- Example 2 A solution of 550 mg of Example 1, 1.5 ml of sodium hydroxide solution (IN) and 2.5 ml of methanol were stirred for 14 hours at RT. The solvent was removed completely in vacuo to give 587 mg of 2- [(4S) -6- (4-chlorophenyl) -1,7,8-trimethyl-4,8-dihydropyrrolo [3,4-] [1,2 , 4] triazolo [4,3-a] [l, 4] diazepin-4-yl] acetic acid, sodium salt. The substance was used without further purification in the next step.
- the mixture was then stirred at 120 ° C. for 2 h and then at 25 ° C. for 16 h.
- the reaction mixture was diluted with 250 ml of ethyl acetate and extracted with 150 ml of water. After phase separation, the aqueous phase was extracted three times with 150 ml of ethyl acetate.
- the combined organic phases were washed with water and saturated sodium chloride solution, dried over sodium sulfate and concentrated in vacuo.
- the crude product thus obtained was purified by chromatography on silica gel (hexane / ethyl acetate gradient). This gave 14.7 g of (4-chlorophenyl) (1-methyl-4-nitro-1H-pyrazol-3-yl) ketone.
- Example 6 To a solution of 132 mg of Example 6 in 2.6 ml of methanol was added 0.38 ml of an IN
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Veterinary Medicine (AREA)
- Medicinal Chemistry (AREA)
- Public Health (AREA)
- Animal Behavior & Ethology (AREA)
- Pharmacology & Pharmacy (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- General Chemical & Material Sciences (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Engineering & Computer Science (AREA)
- Immunology (AREA)
- Reproductive Health (AREA)
- Oncology (AREA)
- Epidemiology (AREA)
- Neurosurgery (AREA)
- Biomedical Technology (AREA)
- Urology & Nephrology (AREA)
- Communicable Diseases (AREA)
- Heart & Thoracic Surgery (AREA)
- Endocrinology (AREA)
- Cardiology (AREA)
- Neurology (AREA)
- Rheumatology (AREA)
- Virology (AREA)
- Hematology (AREA)
- Transplantation (AREA)
- Psychiatry (AREA)
- Vascular Medicine (AREA)
- Pain & Pain Management (AREA)
- Hospice & Palliative Care (AREA)
- Physical Education & Sports Medicine (AREA)
- Dermatology (AREA)
- Gynecology & Obstetrics (AREA)
- Pregnancy & Childbirth (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
Description
Claims
Priority Applications (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP14705164.3A EP2958923A1 (de) | 2013-02-22 | 2014-02-18 | 4-substituierte pyrrolo- und pyrazolo-diazepine |
| CN201480022721.0A CN105164135A (zh) | 2013-02-22 | 2014-02-18 | 4-取代的吡咯并二氮杂卓和吡唑并二氮杂卓 |
| US14/769,921 US20150376196A1 (en) | 2013-02-22 | 2014-02-18 | 4-substituted pyrrolo- and pyrazolo-diazepines |
| CA2901805A CA2901805A1 (en) | 2013-02-22 | 2014-02-18 | 4-substituted pyrrolo- and pyrazolo-diazepines |
| JP2015558420A JP2016513118A (ja) | 2013-02-22 | 2014-02-18 | 4−置換されたピロロ−およびピラゾロジアゼピン類 |
| HK16100318.7A HK1212345A1 (en) | 2013-02-22 | 2016-01-13 | 4-substituted pyrrolo- and pyrazolo-diazepines 4- |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DE102013202991 | 2013-02-22 | ||
| DE102013202991.8 | 2013-02-22 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2014128111A1 true WO2014128111A1 (de) | 2014-08-28 |
Family
ID=50115891
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2014/053099 WO2014128111A1 (de) | 2013-02-22 | 2014-02-18 | 4-substituierte pyrrolo- und pyrazolo-diazepine |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US20150376196A1 (de) |
| EP (1) | EP2958923A1 (de) |
| JP (1) | JP2016513118A (de) |
| CN (1) | CN105164135A (de) |
| CA (1) | CA2901805A1 (de) |
| HK (1) | HK1212345A1 (de) |
| WO (1) | WO2014128111A1 (de) |
Cited By (27)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9271978B2 (en) | 2012-12-21 | 2016-03-01 | Zenith Epigenetics Corp. | Heterocyclic compounds as bromodomain inhibitors |
| US9278940B2 (en) | 2012-11-21 | 2016-03-08 | Zenith Epigenetics Corp. | Cyclic amines as bromodomain inhibitors |
| WO2016061144A1 (en) | 2014-10-14 | 2016-04-21 | The Regents Of The University Of California | Use of cdk9 and brd4 inhibitors to inhibit inflammation |
| WO2016146755A1 (en) | 2015-03-19 | 2016-09-22 | Glaxosmithkline Intellectual Property Development Limited | Covalent conjugates of bet inhibitors and alpha amino acid esters |
| US9636328B2 (en) | 2013-06-21 | 2017-05-02 | Zenith Epigenetics Ltd. | Substituted bicyclic compounds as bromodomain inhibitors |
| US9663520B2 (en) | 2013-06-21 | 2017-05-30 | Zenith Epigenetics Ltd. | Bicyclic bromodomain inhibitors |
| US9765039B2 (en) | 2012-11-21 | 2017-09-19 | Zenith Epigenetics Ltd. | Biaryl derivatives as bromodomain inhibitors |
| WO2017059319A3 (en) * | 2015-10-02 | 2017-10-12 | Dana-Farber Cancer Institute, Inc. | Combination therapy of bromodomain inhibitors and checkpoint blockade |
| US9855271B2 (en) | 2013-07-31 | 2018-01-02 | Zenith Epigenetics Ltd. | Quinazolinones as bromodomain inhibitors |
| US10124009B2 (en) | 2014-10-27 | 2018-11-13 | Tensha Therapeutics, Inc. | Bromodomain inhibitors |
| US10179125B2 (en) | 2014-12-01 | 2019-01-15 | Zenith Epigenetics Ltd. | Substituted pyridines as bromodomain inhibitors |
| US10231953B2 (en) | 2014-12-17 | 2019-03-19 | Zenith Epigenetics Ltd. | Inhibitors of bromodomains |
| US10292968B2 (en) | 2014-12-11 | 2019-05-21 | Zenith Epigenetics Ltd. | Substituted heterocycles as bromodomain inhibitors |
| US10407441B2 (en) | 2010-05-14 | 2019-09-10 | Dana-Farber Cancer Institute, Inc. | Compositions and methods for treating neoplasia, inflammatory disease and other disorders |
| WO2019199816A1 (en) | 2018-04-13 | 2019-10-17 | Arvinas Operations, Inc. | Cereblon ligands and bifunctional compounds comprising the same |
| US10676484B2 (en) | 2010-05-14 | 2020-06-09 | Dana-Farber Cancer Institute, Inc. | Compositions and methods for treating leukemia |
| US10710992B2 (en) | 2014-12-01 | 2020-07-14 | Zenith Epigenetics Ltd. | Substituted pyridinones as bromodomain inhibitors |
| US10730870B2 (en) | 2015-03-18 | 2020-08-04 | Arvinas Operations, Inc. | Compounds and methods for the enhanced degradation of targeted proteins |
| US10730862B2 (en) | 2012-01-12 | 2020-08-04 | Yale University | Compounds and methods for the enhanced degradation of targeted proteins and other polypeptides by an E3 ubiquitin ligase |
| US10772962B2 (en) | 2015-08-19 | 2020-09-15 | Arvinas Operations, Inc. | Compounds and methods for the targeted degradation of bromodomain-containing proteins |
| WO2020207395A1 (en) * | 2019-04-09 | 2020-10-15 | Ranok Therapeutics (Hangzhou) Co., Ltd. | Methods and compositions for targeted protein degradation |
| US10925881B2 (en) | 2014-02-28 | 2021-02-23 | Tensha Therapeutics, Inc. | Treatment of conditions associated with hyperinsulinaemia |
| EP3683214A4 (de) * | 2017-09-14 | 2021-06-30 | Hinova Pharmaceuticals Inc. | Brd4-inhibitor |
| US11220515B2 (en) | 2018-01-26 | 2022-01-11 | Yale University | Imide-based modulators of proteolysis and associated methods of use |
| US11446309B2 (en) | 2013-11-08 | 2022-09-20 | Dana-Farber Cancer Institute, Inc. | Combination therapy for cancer using bromodomain and extra-terminal (BET) protein inhibitors |
| US11883393B2 (en) | 2019-12-19 | 2024-01-30 | Arvinas Operations, Inc. | Compounds and methods for the targeted degradation of androgen receptor |
| RU2820673C2 (ru) * | 2019-04-09 | 2024-06-07 | Ранок Терапьютикс (Ханчжоу) Ко., Лтд. | Способы и композиции для направленной деградации белка |
Families Citing this family (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CA2901799A1 (en) * | 2013-02-22 | 2014-08-28 | Bayer Pharma Aktiengesellschaft | Pyrrolo- and pyrazolo-triazolodiazepines as bet-protein inhibitors for treating hyperproliferative diseases |
| CN105440041A (zh) * | 2015-12-30 | 2016-03-30 | 上海药明康德新药开发有限公司 | 7-叔丁基-2-乙基-8-甲基-5,6-二氢咪唑[1,2-a]吡嗪-2,7(8H)-二羧酸的合成方法 |
| CN109071562B (zh) * | 2016-02-15 | 2022-03-22 | 密执安大学评议会 | 作为bet溴结构域抑制剂的稠合1,4-氧氮杂䓬和相关类似物 |
| CN107629057B (zh) * | 2016-07-19 | 2020-03-27 | 上海勋和医药科技有限公司 | Bet蛋白抑制剂及其应用 |
| CN107216332B (zh) * | 2017-06-29 | 2019-04-30 | 上海药明康德新药开发有限公司 | 叔丁基-7-羟甲基-7,8-二氢4h吡唑并二氮杂卓5(6h)甲酸基酯的合成方法 |
| CN111372937B (zh) * | 2017-09-22 | 2021-10-01 | 石药集团中奇制药技术(石家庄)有限公司 | 噻吩并二氮杂卓衍生物及其应用 |
| CN108129484B (zh) * | 2017-12-19 | 2021-04-27 | 郭守东 | 杂芳环衍生物 |
| CN108084193A (zh) * | 2017-12-19 | 2018-05-29 | 郭守东 | 一种brd4蛋白抑制剂 |
| WO2020063976A1 (zh) * | 2018-09-29 | 2020-04-02 | 如东凌达生物医药科技有限公司 | 一类稠杂环联芳基苄醇类化合物、制备方法和用途 |
| US20210147441A1 (en) * | 2019-09-26 | 2021-05-20 | Agency For Science, Technology And Research (A*Star) | Therapeutic compounds and methods of use thereof |
| CN112321594B (zh) * | 2020-12-07 | 2022-05-20 | 扬子江药业集团有限公司 | 一种苯并二氮杂䓬类药物的制备方法 |
| WO2024069007A2 (en) * | 2022-09-30 | 2024-04-04 | Universität Basel | Immunosuppressive compounds |
Citations (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3657271A (en) | 1970-06-22 | 1972-04-18 | Abbott Lab | 1 3-dimethyl-5-methylamino-8-phenylpyrazolo(4 3-e)(1 4)diazepine |
| EP0102602A1 (de) | 1982-09-08 | 1984-03-14 | GRUPPO LEPETIT S.p.A. | 1,7-Dihydro-pyrrolo(3,4-e)(1,4)diazepin-2(3H)-onderivate, Verfahren zu ihrer Herstellung und ihre Verwendung als Anticonvulsiva und Mittel gegen Angstzustände |
| DE3435973A1 (de) | 1984-10-01 | 1986-04-10 | Boehringer Ingelheim KG, 6507 Ingelheim | Diazepine enthaltende pharmazeutische zusammensetzungen mit paf-antagonistischer wirkung |
| WO1994026718A1 (en) | 1993-05-13 | 1994-11-24 | Yoshitomi Pharmaceutical Industries, Ltd. | 3-aminoazepine compound and pharmaceutical use thereof |
| WO1997034878A1 (en) | 1996-03-21 | 1997-09-25 | Cocensys, Inc. | Substituted 2,3-benzodiazepin-4-ones and the use thereof |
| WO2006051312A1 (en) | 2004-11-15 | 2006-05-18 | James Black Foundation Limited | Gastrin and cholecystokinin receptor ligands |
| WO2009084693A1 (ja) | 2007-12-28 | 2009-07-09 | Mitsubishi Tanabe Pharma Corporation | 抗癌剤 |
| WO2011054843A1 (en) | 2009-11-05 | 2011-05-12 | Glaxosmithkline Llc | Bromodomain inhibitors for treating autoimmune and inflammatory diseases |
| WO2011143669A2 (en) | 2010-05-14 | 2011-11-17 | Dana-Farber Cancer Institute, Inc | Compositions and methods for treating neoplasia, inflammatory disease and other disorders |
| WO2012075456A1 (en) | 2010-12-02 | 2012-06-07 | Constellation Pharmaceuticals | Bromodomain inhibitors and uses thereof |
| WO2012075383A2 (en) | 2010-12-02 | 2012-06-07 | Constellation Pharmaceuticals, Inc. | Bromodomain inhibitors and uses thereof |
| WO2013155695A1 (en) | 2012-04-20 | 2013-10-24 | Abbott Laboratories | Isoindolone derivatives |
| WO2013184878A1 (en) | 2012-06-06 | 2013-12-12 | Constellation Pharmaceuticals, Inc. | Benzo [b] isoxazoloazepine bromodomain inhibitors and uses thereof |
| WO2013184876A1 (en) | 2012-06-06 | 2013-12-12 | Constellation Pharmaceuticals, Inc. | Benzo [c] isoxazoloazepine bromodomain inhibitors and uses thereof |
| WO2013185284A1 (en) | 2012-06-12 | 2013-12-19 | Abbott Laboratories | Pyridinone and pyridazinone derivatives |
-
2014
- 2014-02-18 EP EP14705164.3A patent/EP2958923A1/de not_active Withdrawn
- 2014-02-18 US US14/769,921 patent/US20150376196A1/en not_active Abandoned
- 2014-02-18 CN CN201480022721.0A patent/CN105164135A/zh active Pending
- 2014-02-18 JP JP2015558420A patent/JP2016513118A/ja active Pending
- 2014-02-18 WO PCT/EP2014/053099 patent/WO2014128111A1/de active Application Filing
- 2014-02-18 CA CA2901805A patent/CA2901805A1/en not_active Abandoned
-
2016
- 2016-01-13 HK HK16100318.7A patent/HK1212345A1/xx unknown
Patent Citations (18)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3657271A (en) | 1970-06-22 | 1972-04-18 | Abbott Lab | 1 3-dimethyl-5-methylamino-8-phenylpyrazolo(4 3-e)(1 4)diazepine |
| EP0102602A1 (de) | 1982-09-08 | 1984-03-14 | GRUPPO LEPETIT S.p.A. | 1,7-Dihydro-pyrrolo(3,4-e)(1,4)diazepin-2(3H)-onderivate, Verfahren zu ihrer Herstellung und ihre Verwendung als Anticonvulsiva und Mittel gegen Angstzustände |
| DE3435973A1 (de) | 1984-10-01 | 1986-04-10 | Boehringer Ingelheim KG, 6507 Ingelheim | Diazepine enthaltende pharmazeutische zusammensetzungen mit paf-antagonistischer wirkung |
| WO1994026718A1 (en) | 1993-05-13 | 1994-11-24 | Yoshitomi Pharmaceutical Industries, Ltd. | 3-aminoazepine compound and pharmaceutical use thereof |
| EP0703222A1 (de) | 1993-05-13 | 1996-03-27 | Yoshitomi Pharmaceutical Industries, Ltd. | 3-aminoazepin-verbindung und ihre pharmazeutische verwendung |
| WO1997034878A1 (en) | 1996-03-21 | 1997-09-25 | Cocensys, Inc. | Substituted 2,3-benzodiazepin-4-ones and the use thereof |
| WO2006051312A1 (en) | 2004-11-15 | 2006-05-18 | James Black Foundation Limited | Gastrin and cholecystokinin receptor ligands |
| WO2009084693A1 (ja) | 2007-12-28 | 2009-07-09 | Mitsubishi Tanabe Pharma Corporation | 抗癌剤 |
| WO2011054843A1 (en) | 2009-11-05 | 2011-05-12 | Glaxosmithkline Llc | Bromodomain inhibitors for treating autoimmune and inflammatory diseases |
| WO2011143669A2 (en) | 2010-05-14 | 2011-11-17 | Dana-Farber Cancer Institute, Inc | Compositions and methods for treating neoplasia, inflammatory disease and other disorders |
| WO2012075456A1 (en) | 2010-12-02 | 2012-06-07 | Constellation Pharmaceuticals | Bromodomain inhibitors and uses thereof |
| WO2012075383A2 (en) | 2010-12-02 | 2012-06-07 | Constellation Pharmaceuticals, Inc. | Bromodomain inhibitors and uses thereof |
| WO2013155695A1 (en) | 2012-04-20 | 2013-10-24 | Abbott Laboratories | Isoindolone derivatives |
| WO2013158952A1 (en) | 2012-04-20 | 2013-10-24 | Abbvie Inc. | Isoindolone derivatives |
| WO2013184878A1 (en) | 2012-06-06 | 2013-12-12 | Constellation Pharmaceuticals, Inc. | Benzo [b] isoxazoloazepine bromodomain inhibitors and uses thereof |
| WO2013184876A1 (en) | 2012-06-06 | 2013-12-12 | Constellation Pharmaceuticals, Inc. | Benzo [c] isoxazoloazepine bromodomain inhibitors and uses thereof |
| WO2013185284A1 (en) | 2012-06-12 | 2013-12-19 | Abbott Laboratories | Pyridinone and pyridazinone derivatives |
| WO2013188381A1 (en) | 2012-06-12 | 2013-12-19 | Abbvie Inc. | Pyridinone and pyridazinone derivatives |
Non-Patent Citations (58)
| Title |
|---|
| "Compendium of Organic Synthetic Methods", vol. I-VI, WILEY INTERSCIENCE |
| "Remington's Pharmaceutical Science", 1980, MACK PUBLISHING COMPANY |
| BIOORGANIC & MEDICINAL CHEMISTRY LETTERS, vol. 22, 2012, pages 2963 - 2967 |
| BISGROVE ET AL., PROC. NATL ACAD. SCI. USA, vol. 104, 2007, pages 13690 - 13695 |
| BODANSKY: "The Practice of Peptide Synthesis", SPRINGER VERLAG |
| CHAPMAN ET AL., EUR. HEART J., vol. 32, 2011, pages 1345 - 1361 |
| CHUNG ET AL., J. MED. CHEM, vol. 54, 2011, pages 3827 - 3838 |
| CHUN-WA CHUNG ET AL., PROGRESS IN MEDICINAL CHEMISTRY, vol. 51, 2012, pages 1 - 55 |
| DAWSON ET AL., NATURE, vol. 478, 2011, pages 529 - 533 |
| DEGOMA; RADER, NAT. REV. CARDIOL., vol. 8, 2011, pages 266 - 277 |
| DELMORE ET AL., CELL, vol. 146, 2011, pages 1 - 14 |
| DEWALD, J MED. CHEM., vol. 16, 1973, pages 1346FF |
| DEWALD, J. MED. CHEM., vol. 24, 1981, pages 982FF |
| DEY ET AL., MOL. BIOL. CELL, vol. 20, 2009, pages 4899 - 4909 |
| E. NICODEME ET AL., NATURE, vol. 468, 2010, pages 1119 |
| FILIPPAKOPOULOS ET AL., CELL, vol. 149, 2012, pages 214 - 231 |
| FILIPPAKOPOULOS ET AL., NATURE, vol. 468, 2010, pages 1067 - 1073 |
| FILIPPAKOPOULOS ET AL., NATURE, vol. 468, 2010, pages 1119 - 1123 |
| FRENCH, CANCER GENET. CYTOGENET., vol. 203, 2010, pages 16 - 20 |
| GARCIA, J. MED. CHEM., vol. 48, 2005, pages 4068FF |
| GREENWALL ET AL., BLOOD, vol. 103, 2005, pages 1475 - 1484 |
| GYURIS ET AL., BIOCHIM. BIOPHYS. ACTA, vol. 1789, 2009, pages 413 - 421 |
| HARTWIG, J. AM. CHEM. SOC., vol. 120, 1998, pages 827 - 8 |
| HESTER, J. MED. CHEM., vol. 23, 1980, pages 392FF |
| HOUZELSTEIN ET AL., MOL. CELL. BIOL., vol. 22, 2002, pages 3794 - 3802 |
| HUANG ET AL., MOL. CELL. BIOL., vol. 29, 2009, pages 1375 - 1387 |
| J. MED. CHEM., vol. 54, 2011, pages 3827 - 3838 |
| J. ORG. CHEM., vol. 41, 1976, pages 2720FF |
| KADOTA ET AL., CANCER RES, vol. 69, 2009, pages 7357 - 7365 |
| KUMAR, EUR. J. MED. CHEM., vol. 45, 2010, pages 4664FF |
| KUO; ALLIS, BIOESSAYS, vol. 20, 1998, pages 615 - 626 |
| LEROY ET AL., MOL. CELL, vol. 30, 2008, pages 51 - 60 |
| MERTZ ET AL., PROC. NATL. ACAD. SCI. USA, vol. 108, 2011, pages 16669 - 16674 |
| MOCHIZUKI ET AL., J. BIOL. CHEM., vol. 283, 2008, pages 9040 - 9048 |
| MOFFETT, J. HETEROCYCLIC CHEM., vol. 16, 1979, pages 793FF |
| NICODEME ET AL., NATURE, vol. 468, 2010, pages 1119 - 1123 |
| NMC, C.A. FRENCH, ANNU. REV. PATHOL., vol. 7, 2012, pages 247 - 265 |
| P. FILIPPAKOPOULOS ET AL., NATURE, vol. 468, 2010, pages 1067 |
| RAHMAN ET AL., MOL. CELL. BIOL., vol. 31, 2011, pages 2641 - 2652 |
| SCHRÖDER ET AL., J. BIOL. CHEM., vol. 287, 2012, pages 1090 - 1099 |
| TARZIA, IL FARMACO, EDIZIONE SCIENTIFICA, vol. 39, 1984, pages 538FF |
| UMEHARA ET AL., J. BIOL. CHEM., vol. 285, 2010, pages 7610 - 7618 |
| VIEJO-BORBOLLA ET AL., J. VIROL., vol. 79, 2005, pages 13618 - 13629 |
| VOSA ET AL., J. VIROL., vol. 86, 2012, pages 348 - 357 |
| W. DU, TETRAHEDRON LETT., vol. 47, 2006, pages 4271 - 4 |
| WANG ET AL., BIOCHEM. J., vol. 425, 2009, pages 71 - 83 |
| WU ET AL., GENES DEV., vol. 20, 2006, pages 2383 - 2396 |
| WU; CHIANG, J. BIOL. CHEM., vol. 282, 2007, pages 13141 - 13145 |
| XIE, BIOORG. MED CHEM. LETT., vol. 21, 2011, pages 4306FF |
| YAN ET AL., J. BIOL. CHEM., vol. 286, 2011, pages 27663 - 27675 |
| YANG ET AL., MOL. CELL, vol. 19, 2005, pages 535 - 545 |
| YANG ET AL., MOL. CELL. BIOL., vol. 28, 2008, pages 967 - 976 |
| YOU ET AL., J. VIROL., vol. 80, 2006, pages 8909 - 8919 |
| YOU ET AL., MOL. CELL. BIOL., vol. 29, 2009, pages 5094 - 5103 |
| ZHANG ET AL., J. BIOL. CHEM., 2012 |
| ZHAO ET AL., NAT. CELL. BIOL., vol. 13, 2011, pages 1295 - 1304 |
| ZUBER ET AL., NATURE, 2011 |
| ZUBER ET AL., NATURE, vol. 478, 2011, pages 524 - 528 |
Cited By (41)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US10407441B2 (en) | 2010-05-14 | 2019-09-10 | Dana-Farber Cancer Institute, Inc. | Compositions and methods for treating neoplasia, inflammatory disease and other disorders |
| US10676484B2 (en) | 2010-05-14 | 2020-06-09 | Dana-Farber Cancer Institute, Inc. | Compositions and methods for treating leukemia |
| US10730862B2 (en) | 2012-01-12 | 2020-08-04 | Yale University | Compounds and methods for the enhanced degradation of targeted proteins and other polypeptides by an E3 ubiquitin ligase |
| US9765039B2 (en) | 2012-11-21 | 2017-09-19 | Zenith Epigenetics Ltd. | Biaryl derivatives as bromodomain inhibitors |
| US9278940B2 (en) | 2012-11-21 | 2016-03-08 | Zenith Epigenetics Corp. | Cyclic amines as bromodomain inhibitors |
| US9598367B2 (en) | 2012-12-21 | 2017-03-21 | Zenith Epigenetics Ltd. | Heterocyclic compounds as bromodomain inhibitors |
| US9271978B2 (en) | 2012-12-21 | 2016-03-01 | Zenith Epigenetics Corp. | Heterocyclic compounds as bromodomain inhibitors |
| US9861637B2 (en) | 2012-12-21 | 2018-01-09 | Zenith Epigenetics Ltd. | Heterocyclic compounds as bromodomain inhibitors |
| US9662311B2 (en) | 2013-06-21 | 2017-05-30 | Zenith Epigenetics Ltd. | Substituted bicyclic compounds as bromodomain inhibitors |
| US9663520B2 (en) | 2013-06-21 | 2017-05-30 | Zenith Epigenetics Ltd. | Bicyclic bromodomain inhibitors |
| US9636328B2 (en) | 2013-06-21 | 2017-05-02 | Zenith Epigenetics Ltd. | Substituted bicyclic compounds as bromodomain inhibitors |
| US10772892B2 (en) | 2013-06-21 | 2020-09-15 | Zenith Epigenetics Ltd. | Bicyclic bromodomain inhibitors |
| US10010556B2 (en) | 2013-06-21 | 2018-07-03 | Zenith Epigenetics Ltd. | Bicyclic bromodomain inhibitors |
| US10363257B2 (en) | 2013-06-21 | 2019-07-30 | Zenith Epigenetics Ltd. | Bicyclic bromodomain inhibitors |
| US10166215B2 (en) | 2013-06-21 | 2019-01-01 | Zenith Epigenetics Ltd. | Substituted bicyclic compounds as bromodomain inhibitors |
| US11026926B2 (en) | 2013-06-21 | 2021-06-08 | Zenith Epigenetics Ltd. | Substituted bicyclic compounds as bromodomain inhibitors |
| US10226451B2 (en) | 2013-06-21 | 2019-03-12 | Zenith Epigenetics Ltd. | Substituted bicyclic compounds as bromodomain inhibitors |
| US11446306B2 (en) | 2013-06-21 | 2022-09-20 | Zenith Epigenetics Ltd. | Bicyclic bromodomain inhibitors |
| US9855271B2 (en) | 2013-07-31 | 2018-01-02 | Zenith Epigenetics Ltd. | Quinazolinones as bromodomain inhibitors |
| US10500209B2 (en) | 2013-07-31 | 2019-12-10 | Zenith Epigenetics Ltd. | Quinazolinones as bromodomain inhibitors |
| US11446309B2 (en) | 2013-11-08 | 2022-09-20 | Dana-Farber Cancer Institute, Inc. | Combination therapy for cancer using bromodomain and extra-terminal (BET) protein inhibitors |
| US10925881B2 (en) | 2014-02-28 | 2021-02-23 | Tensha Therapeutics, Inc. | Treatment of conditions associated with hyperinsulinaemia |
| WO2016061144A1 (en) | 2014-10-14 | 2016-04-21 | The Regents Of The University Of California | Use of cdk9 and brd4 inhibitors to inhibit inflammation |
| US10124009B2 (en) | 2014-10-27 | 2018-11-13 | Tensha Therapeutics, Inc. | Bromodomain inhibitors |
| US10710992B2 (en) | 2014-12-01 | 2020-07-14 | Zenith Epigenetics Ltd. | Substituted pyridinones as bromodomain inhibitors |
| US10179125B2 (en) | 2014-12-01 | 2019-01-15 | Zenith Epigenetics Ltd. | Substituted pyridines as bromodomain inhibitors |
| US10292968B2 (en) | 2014-12-11 | 2019-05-21 | Zenith Epigenetics Ltd. | Substituted heterocycles as bromodomain inhibitors |
| US10231953B2 (en) | 2014-12-17 | 2019-03-19 | Zenith Epigenetics Ltd. | Inhibitors of bromodomains |
| US10730870B2 (en) | 2015-03-18 | 2020-08-04 | Arvinas Operations, Inc. | Compounds and methods for the enhanced degradation of targeted proteins |
| US11512083B2 (en) | 2015-03-18 | 2022-11-29 | Arvinas Operations, Inc. | Compounds and methods for the enhanced degradation of targeted proteins |
| WO2016146755A1 (en) | 2015-03-19 | 2016-09-22 | Glaxosmithkline Intellectual Property Development Limited | Covalent conjugates of bet inhibitors and alpha amino acid esters |
| US10772962B2 (en) | 2015-08-19 | 2020-09-15 | Arvinas Operations, Inc. | Compounds and methods for the targeted degradation of bromodomain-containing proteins |
| US11554171B2 (en) | 2015-08-19 | 2023-01-17 | Arvinas Operations, Inc. | Compounds and methods for the targeted degradation of bromodomain-containing proteins |
| WO2017059319A3 (en) * | 2015-10-02 | 2017-10-12 | Dana-Farber Cancer Institute, Inc. | Combination therapy of bromodomain inhibitors and checkpoint blockade |
| EP3683214A4 (de) * | 2017-09-14 | 2021-06-30 | Hinova Pharmaceuticals Inc. | Brd4-inhibitor |
| US11220515B2 (en) | 2018-01-26 | 2022-01-11 | Yale University | Imide-based modulators of proteolysis and associated methods of use |
| US11834460B2 (en) | 2018-01-26 | 2023-12-05 | Yale University | Imide-based modulators of proteolysis and associated methods of use |
| WO2019199816A1 (en) | 2018-04-13 | 2019-10-17 | Arvinas Operations, Inc. | Cereblon ligands and bifunctional compounds comprising the same |
| WO2020207395A1 (en) * | 2019-04-09 | 2020-10-15 | Ranok Therapeutics (Hangzhou) Co., Ltd. | Methods and compositions for targeted protein degradation |
| RU2820673C2 (ru) * | 2019-04-09 | 2024-06-07 | Ранок Терапьютикс (Ханчжоу) Ко., Лтд. | Способы и композиции для направленной деградации белка |
| US11883393B2 (en) | 2019-12-19 | 2024-01-30 | Arvinas Operations, Inc. | Compounds and methods for the targeted degradation of androgen receptor |
Also Published As
| Publication number | Publication date |
|---|---|
| CA2901805A1 (en) | 2014-08-28 |
| US20150376196A1 (en) | 2015-12-31 |
| JP2016513118A (ja) | 2016-05-12 |
| HK1212345A1 (en) | 2016-06-10 |
| EP2958923A1 (de) | 2015-12-30 |
| CN105164135A (zh) | 2015-12-16 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP2958923A1 (de) | 4-substituierte pyrrolo- und pyrazolo-diazepine | |
| EP3027614B1 (de) | Substituierte dihydropyrido[3,4-b]pyrazinone als duale inhibitoren von bet-proteinen und polo-like kinasen | |
| WO2014128070A1 (de) | Pyrrolo- und pyrazolo-triazolodiazepine als bet-proteininhibitoren zur behandlung von hyper-proliferativen erkrankungen | |
| EP2900672B1 (de) | Bet-proteininhibitorische 5-aryl-triazolo-azepine | |
| EP2885286B1 (de) | 2,3-benzodiazepine | |
| EP3010909A1 (de) | Substituierte phenyl-2,3-benzodiazepine | |
| EP3019493B1 (de) | Modifizierte bet-proteininhibitorische dihydrochinoxalinone und dihydropyridopyrazinone | |
| WO2014095774A1 (de) | Bet-proteininhibitorische dihydropyridopyrazinone | |
| WO2014095775A1 (de) | Bet-proteininhibitorische dihydrochinoxalinone | |
| DE102011082013A1 (de) | 6H-Thieno[3,2-f][1,2,4]triazolo[4,3-a][1,4]diazepine | |
| EP2958909A1 (de) | Bicyclo- und spirocyclisch substituierte 2,3-benzodiazepine | |
| WO2015193219A1 (de) | Bet-proteininhibitorische 3,4-dihydropyrido[2,3-b]pyrazinone mit meta-substituierter aromatischer amino- oder ethergruppe | |
| WO2015121268A1 (de) | 1-phenyl-3h-2,3-benzodiazepine und ihre verwendung als bromodomänen-inhibitoren | |
| WO2015121230A1 (de) | 9-substituierte 2,3-benzodiazepine | |
| WO2015121226A1 (de) | 6-substituierte 2,3-benzodiazepine | |
| WO2015121227A1 (de) | 6,9-disubstituierte 1 -phenyl-3h-2,3-benzodiazepine und ihre verwendung als bromodomänen- inhibitoren | |
| WO2015193228A1 (de) | Bet-proteininhibitorische 1,4-dihydropyrido[3,4-b]pyrazinone mit para-substituierter aromatischer amino- oder ethergruppe | |
| DE102017005091A1 (de) | Substituierte 3,4-Dihydropyrido[2,3-b]pyrazin-2(1H)-one | |
| WO2015193217A1 (de) | Bet-proteininhibitorische dihydropyrido[2,3-b]pyrazinon-derivate mit para-substituierter aromatischer amino- oder ethergruppe | |
| DE102017005089A1 (de) | Substitulerte 3,4-Dihydrochinoxalin-2(1H)-one | |
| WO2015193229A1 (de) | Bet-proteininhibitorische 1,4-dihydropyrido[3,4-b]pyrazinone mit meta-substituierter aromatischer amino- oder ethergruppe |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 201480022721.0 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 14705164 Country of ref document: EP Kind code of ref document: A1 |
|
| REEP | Request for entry into the european phase |
Ref document number: 2014705164 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2014705164 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 2901805 Country of ref document: CA |
|
| ENP | Entry into the national phase |
Ref document number: 2015558420 Country of ref document: JP Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 14769921 Country of ref document: US |