WO2012147625A1 - 撥水撥油剤組成物、その製造方法および物品 - Google Patents
撥水撥油剤組成物、その製造方法および物品 Download PDFInfo
- Publication number
- WO2012147625A1 WO2012147625A1 PCT/JP2012/060625 JP2012060625W WO2012147625A1 WO 2012147625 A1 WO2012147625 A1 WO 2012147625A1 JP 2012060625 W JP2012060625 W JP 2012060625W WO 2012147625 A1 WO2012147625 A1 WO 2012147625A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- monomer
- group
- water
- film
- surfactant
- Prior art date
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F214/00—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by a halogen
- C08F214/02—Monomers containing chlorine
- C08F214/04—Monomers containing two carbon atoms
- C08F214/06—Vinyl chloride
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K3/00—Materials not provided for elsewhere
- C09K3/18—Materials not provided for elsewhere for application to surfaces to minimize adherence of ice, mist or water thereto; Thawing or antifreeze materials for application to surfaces
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F214/00—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by a halogen
- C08F214/18—Monomers containing fluorine
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F214/00—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by a halogen
- C08F214/18—Monomers containing fluorine
- C08F214/182—Monomers containing fluorine not covered by the groups C08F214/20 - C08F214/28
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F214/00—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by a halogen
- C08F214/18—Monomers containing fluorine
- C08F214/184—Monomers containing fluorine with fluorinated vinyl ethers
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F220/00—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and only one being terminated by only one carboxyl radical or a salt, anhydride ester, amide, imide or nitrile thereof
- C08F220/02—Monocarboxylic acids having less than ten carbon atoms; Derivatives thereof
- C08F220/10—Esters
- C08F220/22—Esters containing halogen
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K5/00—Use of organic ingredients
- C08K5/04—Oxygen-containing compounds
- C08K5/06—Ethers; Acetals; Ketals; Ortho-esters
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K5/00—Use of organic ingredients
- C08K5/16—Nitrogen-containing compounds
- C08K5/20—Carboxylic acid amides
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L33/00—Compositions of homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and only one being terminated by only one carboxyl radical, or of salts, anhydrides, esters, amides, imides or nitriles thereof; Compositions of derivatives of such polymers
- C08L33/04—Homopolymers or copolymers of esters
- C08L33/14—Homopolymers or copolymers of esters of esters containing halogen, nitrogen, sulfur, or oxygen atoms in addition to the carboxy oxygen
- C08L33/16—Homopolymers or copolymers of esters containing halogen atoms
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D5/00—Coating compositions, e.g. paints, varnishes or lacquers, characterised by their physical nature or the effects produced; Filling pastes
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06M—TREATMENT, NOT PROVIDED FOR ELSEWHERE IN CLASS D06, OF FIBRES, THREADS, YARNS, FABRICS, FEATHERS OR FIBROUS GOODS MADE FROM SUCH MATERIALS
- D06M15/00—Treating fibres, threads, yarns, fabrics, or fibrous goods made from such materials, with macromolecular compounds; Such treatment combined with mechanical treatment
- D06M15/19—Treating fibres, threads, yarns, fabrics, or fibrous goods made from such materials, with macromolecular compounds; Such treatment combined with mechanical treatment with synthetic macromolecular compounds
- D06M15/21—Macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds
- D06M15/244—Macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds of halogenated hydrocarbons
- D06M15/248—Macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds of halogenated hydrocarbons containing chlorine
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06M—TREATMENT, NOT PROVIDED FOR ELSEWHERE IN CLASS D06, OF FIBRES, THREADS, YARNS, FABRICS, FEATHERS OR FIBROUS GOODS MADE FROM SUCH MATERIALS
- D06M15/00—Treating fibres, threads, yarns, fabrics, or fibrous goods made from such materials, with macromolecular compounds; Such treatment combined with mechanical treatment
- D06M15/19—Treating fibres, threads, yarns, fabrics, or fibrous goods made from such materials, with macromolecular compounds; Such treatment combined with mechanical treatment with synthetic macromolecular compounds
- D06M15/21—Macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds
- D06M15/263—Macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds of unsaturated carboxylic acids; Salts or esters thereof
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06M—TREATMENT, NOT PROVIDED FOR ELSEWHERE IN CLASS D06, OF FIBRES, THREADS, YARNS, FABRICS, FEATHERS OR FIBROUS GOODS MADE FROM SUCH MATERIALS
- D06M15/00—Treating fibres, threads, yarns, fabrics, or fibrous goods made from such materials, with macromolecular compounds; Such treatment combined with mechanical treatment
- D06M15/19—Treating fibres, threads, yarns, fabrics, or fibrous goods made from such materials, with macromolecular compounds; Such treatment combined with mechanical treatment with synthetic macromolecular compounds
- D06M15/21—Macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds
- D06M15/263—Macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds of unsaturated carboxylic acids; Salts or esters thereof
- D06M15/273—Macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds of unsaturated carboxylic acids; Salts or esters thereof of unsaturated carboxylic esters having epoxy groups
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06M—TREATMENT, NOT PROVIDED FOR ELSEWHERE IN CLASS D06, OF FIBRES, THREADS, YARNS, FABRICS, FEATHERS OR FIBROUS GOODS MADE FROM SUCH MATERIALS
- D06M15/00—Treating fibres, threads, yarns, fabrics, or fibrous goods made from such materials, with macromolecular compounds; Such treatment combined with mechanical treatment
- D06M15/19—Treating fibres, threads, yarns, fabrics, or fibrous goods made from such materials, with macromolecular compounds; Such treatment combined with mechanical treatment with synthetic macromolecular compounds
- D06M15/21—Macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds
- D06M15/263—Macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds of unsaturated carboxylic acids; Salts or esters thereof
- D06M15/277—Macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds of unsaturated carboxylic acids; Salts or esters thereof containing fluorine
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06M—TREATMENT, NOT PROVIDED FOR ELSEWHERE IN CLASS D06, OF FIBRES, THREADS, YARNS, FABRICS, FEATHERS OR FIBROUS GOODS MADE FROM SUCH MATERIALS
- D06M15/00—Treating fibres, threads, yarns, fabrics, or fibrous goods made from such materials, with macromolecular compounds; Such treatment combined with mechanical treatment
- D06M15/19—Treating fibres, threads, yarns, fabrics, or fibrous goods made from such materials, with macromolecular compounds; Such treatment combined with mechanical treatment with synthetic macromolecular compounds
- D06M15/21—Macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds
- D06M15/356—Macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds of other unsaturated compounds containing nitrogen, sulfur, silicon or phosphorus atoms
- D06M15/3562—Macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds of other unsaturated compounds containing nitrogen, sulfur, silicon or phosphorus atoms containing nitrogen
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06M—TREATMENT, NOT PROVIDED FOR ELSEWHERE IN CLASS D06, OF FIBRES, THREADS, YARNS, FABRICS, FEATHERS OR FIBROUS GOODS MADE FROM SUCH MATERIALS
- D06M15/00—Treating fibres, threads, yarns, fabrics, or fibrous goods made from such materials, with macromolecular compounds; Such treatment combined with mechanical treatment
- D06M15/19—Treating fibres, threads, yarns, fabrics, or fibrous goods made from such materials, with macromolecular compounds; Such treatment combined with mechanical treatment with synthetic macromolecular compounds
- D06M15/21—Macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds
- D06M15/356—Macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds of other unsaturated compounds containing nitrogen, sulfur, silicon or phosphorus atoms
- D06M15/3564—Macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds of other unsaturated compounds containing nitrogen, sulfur, silicon or phosphorus atoms containing phosphorus
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06M—TREATMENT, NOT PROVIDED FOR ELSEWHERE IN CLASS D06, OF FIBRES, THREADS, YARNS, FABRICS, FEATHERS OR FIBROUS GOODS MADE FROM SUCH MATERIALS
- D06M2200/00—Functionality of the treatment composition and/or properties imparted to the textile material
- D06M2200/10—Repellency against liquids
- D06M2200/11—Oleophobic properties
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06M—TREATMENT, NOT PROVIDED FOR ELSEWHERE IN CLASS D06, OF FIBRES, THREADS, YARNS, FABRICS, FEATHERS OR FIBROUS GOODS MADE FROM SUCH MATERIALS
- D06M2200/00—Functionality of the treatment composition and/or properties imparted to the textile material
- D06M2200/10—Repellency against liquids
- D06M2200/12—Hydrophobic properties
Definitions
- the present invention relates to a water / oil repellent composition, a method for producing the same, and an article whose surface has been treated with the water / oil repellent composition.
- a copolymer having a structural unit based on a monomer having a polyfluoroalkyl group (hereinafter referred to as Rf group) is aqueous.
- Rf group a monomer having a polyfluoroalkyl group
- drying at room temperature is referred to as air drying.
- water / oil repellency of the article hereinafter, the water / oil repellency of the article when air-dried is referred to as water / oil repellency after air drying. Is insufficient.
- the following water / oil repellent composition has been proposed as a water / oil repellent composition capable of imparting water / oil repellency after air drying to the surface of the article.
- a structural unit based on a monomer having an R f group (II) a structural unit based on a monomer having a urethane or urea bond; and (III) a single polymer having a glass transition temperature of 50 ° C. or less.
- a copolymer (A) having a structural unit based on a monomer, (IV) a structural unit based on a monomer having a hydrophilic group, (V) a structural unit based on a monomer having a chlorine atom, and a film-forming aid A water / oil repellent composition in which (B) is dispersed in an aqueous medium (Patent Document 1).
- R F group perfluoroalkyl group having carbon atoms
- R F group compounds having decomposes environment in vivo degradation product It has been pointed out that there is a possibility that things accumulate, that is, the environmental load is high. Therefore, it comprises a copolymer having a structural unit based on a monomer having an R F group having 6 or less carbon atoms and having no structural unit based on a monomer having an R F group having 7 or more carbon atoms. There is a need for water and oil repellent compositions.
- the present invention provides a water- and oil-repellent composition that can impart sufficient water- and oil-repellency after air drying to the surface of the article and has a low environmental load, a method for producing the same, and an article whose surface is treated with the water- and oil-repellent composition I will provide a.
- the water / oil repellent composition of the present invention comprises a copolymer (A) and an aqueous medium (B), wherein the copolymer (A) comprises a structural unit based on the following monomer (a), A structural unit based on the monomer (b), at least one structural unit selected from a structural unit based on the following monomer (c) and a structural unit based on the following monomer (d),
- the aqueous medium (B) contains water and at least one film-forming aid selected from the following film-forming aid (x) and the following film-forming aid (y).
- Z is a polyfluoroalkyl group having 1 to 6 carbon atoms or a group represented by the following formula (2)
- Y is a divalent organic group having no fluorine atom or a single bond
- X is one of the groups represented by the following formulas (3-1) to (3-5) when n is 1, and when n is 2,
- i is an integer of 1 to 6
- j is an integer of 0 to 10
- X 1 and X 2 are each a fluorine atom or a trifluoromethyl group.
- R is a hydrogen atom, a methyl group, or a halogen atom, and ⁇ is a phenylene group.
- Monomer (d) a monomer having no polyfluoroalkyl group and having a functional group capable of crosslinking.
- Film-forming auxiliary (x) a compound represented by the following formula (5).
- r is an integer of 0 to 3
- s is 0 or 1
- R 1 is an alkyl group having 1 to 4 carbon atoms
- R 2 and R 3 are each independently 1 to 3 carbon atoms.
- R 4 is a hydrogen atom or a methyl group.
- Film-forming aid (y) a compound represented by the following formula (6).
- t is 3 or 4
- R 5 and R 6 are each independently an alkyl group having 1 to 4 carbon atoms.
- the copolymer (A) comprises a structural unit based on the monomer (a), a structural unit based on the monomer (b), a structural unit based on the monomer (c), and the single unit. It is preferable to have a structural unit based on the monomer (d).
- Z is a perfluoroalkyl group having 4 to 6 carbon atoms
- Y is an alkylene group having 1 to 4 carbon atoms
- n is 1
- X is A monomer that is a group (3-3) [wherein R is a hydrogen atom or a methyl group] is preferable.
- the monomer (c) is preferably an acrylate or methacrylate having a cycloalkyl group or an alkyl group having 16 to 25 carbon atoms. Moreover, it is preferable that the functional group which can be bridge
- the film-forming aid (x) is preferably 3-alkoxy-N, N-dialkylpropionamide, and the film-forming aid (y) is triethylene glycol dimethyl ether or tetraethylene glycol dimethyl ether. preferable.
- the total ratio of the film-forming auxiliary (x) and the film-forming auxiliary (y) is preferably 0.1 to 20% by mass in the aqueous medium (B) (100% by mass).
- the total amount of the film-forming auxiliary (x) and the film-forming auxiliary (y) is preferably 10 to 4000 parts by mass with respect to 100 parts by mass of the copolymer (A).
- the water / oil repellent composition of the present invention is at least one selected from the film-forming aid (x) and the film-forming aid (y) in an emulsion obtained by emulsion polymerization of the monomer in an aqueous medium.
- An emulsion obtained by blending seeds is preferred.
- the method for producing the water / oil repellent composition of the present invention after the monomer is polymerized in an aqueous medium in the presence of a surfactant and a polymerization initiator, an emulsion of the copolymer (A) is produced.
- the obtained emulsion is blended with at least one film-forming auxiliary selected from film-forming auxiliary (x) and film-forming auxiliary (y).
- the method for producing the water / oil repellent composition of the present invention also comprises at least one film-forming aid selected from a film-forming aid (x) and a film-forming aid (y) in the presence of a surfactant and a polymerization initiator.
- a copolymer (A) emulsion is produced by polymerizing the monomer in an aqueous medium containing a film aid.
- the article of the present invention is an article having a coating film of the copolymer (A) whose surface is treated with the water / oil repellent composition of the present invention.
- the water and oil repellent composition of the present invention can impart sufficient water and oil repellency after air drying to the surface of an article, and has a low environmental load.
- the article of the present invention has sufficient water and oil repellency after air drying and has a low environmental load.
- a compound represented by the formula (1) is referred to as a compound (1).
- group represented by Formula (2) is described as group (2).
- Groups represented by other formulas are also described in the same manner.
- (meth) acrylate in this specification means an acrylate or a methacrylate.
- the monomer in this specification means the compound which has a polymerizable unsaturated group.
- the R f group in the present specification is a group in which part or all of the hydrogen atoms of the alkyl group are substituted with fluorine atoms.
- the R F group in the present specification is a group in which all of the hydrogen atoms of the alkyl group are substituted with fluorine atoms.
- the water / oil repellent composition of the present invention contains a specific copolymer (A) and a specific aqueous medium (B) as essential components, and optionally contains a surfactant and an additive.
- the water / oil repellent composition of the present invention is preferably an emulsion in which fine particles of the copolymer (A) are stably dispersed in the aqueous medium (B). Contains an active agent.
- the copolymer (A) includes a structural unit based on the monomer (a), a structural unit based on the monomer (b), a structural unit based on the monomer (c), and the monomer (d). With either or both of the building blocks based.
- the copolymer (A) is composed of a structural unit based on the monomer (a) and a structural unit based on the monomer (b) in terms of water and oil repellency after air drying and dynamic water repellency (heavy rain water repellency). And a structural unit based on the monomer (c) and a structural unit based on the monomer (d).
- the copolymer (A) may have a structural unit based on the monomer (e) other than the monomer (a) to the monomer (d).
- Monomer (a) is compound (1). (ZY) n X (1). By having a structural unit based on the monomer (a), water and oil repellency can be imparted to the article.
- Z is an R f group having 1 to 6 carbon atoms (provided that the R f group may contain an etheric oxygen atom) or a group (2).
- i is an integer of 1 to 6
- j is an integer of 0 to 10
- X 1 and X 2 are each independently a fluorine atom or a trifluoromethyl group.
- Z is preferably an R f group having 4 to 6 carbon atoms, more preferably an R f group having 6 carbon atoms.
- the R f group is preferably an R F group.
- the R f group may be linear or branched, and is preferably linear.
- the number of carbon atoms in the R f group is preferably 4-6.
- Z examples include the following groups. F (CF 2) 4 -, F (CF 2 ) 5- , F (CF 2 ) 6- , (CF 3 ) 2 CF (CF 2 ) 2- , C k F 2k + 1 O [CF (CF 3 ) CF 2 O] h —CF (CF 3 ) — and the like. However, k is an integer of 1 to 6, and h is an integer of 0 to 10.
- Y is a divalent organic group having no fluorine atom or a single bond.
- an alkylene group is preferable.
- the alkylene group may be linear or branched.
- the alkylene group preferably has 2 to 6 carbon atoms.
- the divalent organic group includes —O—, —NH—, —CO—, —S—, —SO 2 —, —CD 1 ⁇ CD 2 — (wherein D 1 and D 2 are each independently a hydrogen atom or A methyl group) and the like.
- Examples of Y include the following groups. —CH 2 —, —CH 2 CH 2 — -(CH 2 ) 3- , -CH 2 CH 2 CH (CH 3 )-, —CH ⁇ CH—CH 2 —, -S-CH 2 CH 2- , —CH 2 CH 2 —S—CH 2 CH 2 —, —CH 2 CH 2 —SO 2 —CH 2 CH 2 —, —W—OC (O) NH—A—NHC (O) O— (C p H 2p ) — and the like.
- A is unbranched contrasting alkylene group, an arylene group or an aralkylene group, -C 6 H 12 -, - ⁇ -CH 2 - ⁇ -, - ⁇ - (although, phi is a phenylene group.) Is preferred.
- W is any of the following groups. —SO 2 N (R 7 ) —C d H 2d —, -CONHC d H 2d -, —CH (R F1 ) —C e H 2e —, -C q H 2q- .
- R 7 is a hydrogen atom or an alkyl group having 1 to 4 carbon atoms
- d is an integer of 2 to 8
- R F1 is a perfluoroalkyl group having 1 to 6 carbon atoms
- e is It is an integer from 0 to 6
- q is an integer from 1 to 20.
- R F1 is preferably a C 4 or C 6 perfluoroalkyl group.
- n 1 or 2.
- X is any one of groups (3-1) to (3-5) when n is 1, and when n is 2, X is any of groups (4-1) to (4-4). Either.
- R is a hydrogen atom, a methyl group, or a halogen atom, and ⁇ is a phenylene group.
- R is a hydrogen atom, methyl group or halogen atom, and m is an integer of 0-4.
- the Z is a R F group
- Y is preferably an alkylene radical
- n is preferably 1.
- X is preferably a group (3-3) to a group (3-5), more preferably a group (3-3).
- R is preferably a hydrogen atom or a methyl group.
- (meth) acrylates having 4 to 6 carbon R F groups are preferred.
- Z is an R F group having 4-6 carbon atoms
- Y is an alkylene group having 1 to 4 carbon atoms
- n is 1
- X is a group (3-3) [where And R is particularly preferably a hydrogen atom or a methyl group.
- Monomer (b) is vinylidene chloride.
- the affinity of the copolymer (A) for the article and the film-forming property are improved. Therefore, the article after being immersed in the water / oil repellent composition is air-dried. Even in such a case, the copolymer (A) penetrates into every part of the article (textile product etc.) where the coating with the copolymer (A) is difficult to be formed, such as between the fibers. A uniform and ideal film according to A) can be formed. As a result, sufficient water and oil repellency can be imparted to the article after air drying.
- the monomer (c) is a monomer having no R f group and a crosslinkable functional group described later, and having a glass transition temperature of the homopolymer (hereinafter referred to as Tg) of 20 ° C. or higher. .
- Tg glass transition temperature of the homopolymer
- Vinyl chloride which is a kind of monomer (c) is a kind of chlorinated olefin, similarly to vinylidene chloride of monomer (b), and the structural unit based on vinyl chloride is the copolymer (A).
- a more preferable copolymer (A) is a copolymer having substantially no structural unit based on vinyl chloride.
- Tg of the homopolymer of monomer (c) is preferably 22 ° C. or higher, more preferably 40 ° C. or higher, further preferably 60 ° C. or higher, and particularly preferably 100 ° C. or higher. Moreover, it is preferable that the upper limit of Tg is 200 degreeC.
- the Tg of the homopolymer of monomer (c) is the midpoint glass transition temperature measured by the differential scanning calorimetry (DSC) method according to JIS K 7121: 1987.
- the monomer (c) is preferably a (meth) acrylate having a cycloalkyl group or an alkyl group having 16 to 25 carbon atoms from the viewpoint of water / oil repellency after air drying and dynamic water repellency (heavy rain water repellency).
- a (meth) acrylate having a cycloalkyl group or an alkyl group having 16 to 25 carbon atoms from the viewpoint of water / oil repellency after air drying and dynamic water repellency (heavy rain water repellency).
- stearyl acrylate, behenyl acrylate, behenyl methacrylate, isobornyl acrylate, isobornyl methacrylate and the like are preferable.
- the monomer (d) is a monomer having no functional group capable of crosslinking without having an R f group. By having the structural unit based on the monomer (d), it is possible to improve water and oil repellency after air drying and to impart dynamic water repellency (heavy rain water repellency) to the article.
- the functional group capable of crosslinking is preferably a functional group having at least one bond among a covalent bond, an ionic bond and a hydrogen bond, or a functional group capable of forming a crosslinked structure by the interaction of the bonds. Further, it may be a compound having an active organic group or an element such as hydrogen or halogen in the molecule.
- the functional group include a hydroxyl group, an isocyanate group, a blocked isocyanate group, an alkoxysilyl group, an amino group, an N-alkoxymethylamide group, a silanol group, an ammonium group, an amide group, an epoxy group, an oxazoline group, a carboxy group, and an alkenyl group.
- a sulfo group and the like are preferable, and a hydroxyl group, a blocked isocyanate group, an amino group, and an epoxy group are particularly preferable.
- (meth) acrylates, acrylamides, vinyl ethers, or vinyl esters are preferable.
- the monomer (d) the following compounds may be mentioned.
- t-butyl (meth) acrylamide sulfonic acid (meth) acrylamide, N-methyl (meth) acrylamide, N-methylol (meth) acrylamide, N-butoxymethyl (meth) acrylamide, glycidyl (meth) acrylate, 2-hydroxyethyl (Meth) acrylate, 2-hydroxypropyl (meth) acrylate, 4-hydroxybutyl (meth) acrylate, 3-chloro-2-hydroxypropyl methacrylate, polyoxyalkylene glycol mono (meth) acrylate, (meth) acrylic acid, 2 -(Meth) acryloyloxyethyl succinic acid, 2- (meth) acryloyloxyhexahydrophthalic acid, 2- (meth) acryloyloxyethyl acid phosphate, allyl (meth) acrylate, 2-vinyl 2-oxazoline, 2-vinyl-4-methyl - (2-vinyl
- a polycaprolactone ester of (meth) acrylate (Placcel FA, FM series, manufactured by Daicel Chemical Industries) is preferable.
- the monomer (e) is a monomer other than the monomer (a), the monomer (b), the monomer (c), and the monomer (d). Examples of the monomer (e) include known compounds.
- the proportion of the structural unit based on the monomer (a) is, among the structural units based on all monomers (100% by mass), from the viewpoint of water / oil repellency after air drying and dynamic water repellency (heavy rain water repellency). 30 to 80% by mass is preferable, 50 to 80% by mass is more preferable, and 65 to 75% by mass is particularly preferable.
- the proportion of the structural unit based on the monomer (b) is, among the structural units based on all monomers (100% by mass), from the viewpoint of water / oil repellency after air drying and dynamic water repellency (heavy rain water repellency). It is preferably 5 to 40% by mass, more preferably 10 to 30% by mass, and particularly preferably 10 to 20% by mass.
- the total proportion of the structural unit based on the monomer (c) and the structural unit based on the monomer (d) is determined from the viewpoint of water and oil repellency after air drying and dynamic water repellency (heavy rain water repellency).
- the proportion of the structural unit based on the monomer (e) is preferably from 0 to 50% by weight, more preferably from 0 to 30% by weight, among the structural units based on all monomers (100% by weight), from 0 to 10% by mass is particularly preferred.
- the proportion of structural units based on monomers in the present invention is determined from NMR analysis and elemental analysis. In addition, when it cannot obtain
- the weight average molecular weight (Mw) of the copolymer (A) is 50000 or more, and 60000 or more is particularly preferable. Within this range, the water and oil repellency and dynamic water repellency (heavy rain water repellency) after air drying can be further improved.
- the weight average molecular weight (Mw) of the copolymer (A) is preferably 500,000 or less, particularly preferably 300,000 or less, from the viewpoint of film forming properties and storage stability.
- the number average molecular weight (Mn) of the copolymer (A) is preferably 12,000 or more, particularly preferably 15000 or more.
- the number average molecular weight (Mn) of the copolymer (A) is preferably 300000 or less, particularly preferably 150,000 or less.
- the mass average molecular weight (Mw) and the number average molecular weight (Mn) of the copolymer (A) are molecular weights in terms of polystyrene measured by gel permeation chromatography (GPC). Specifically, the following method is used. Measure with
- the copolymer (A) is dissolved in tetrahydrofuran (hereinafter referred to as THF) to form a 0.5% by mass solution, which is passed through a 0.2 ⁇ m filter to obtain an analysis sample. About this sample, a number average molecular weight (Mn) and a mass average molecular weight (Mw) are measured on condition of the following. Measurement temperature: 23 ° C. Injection volume: 0.2 mL, Outflow rate: 1 mL / min, Eluent: THF.
- THF tetrahydrofuran
- the copolymer (A) is preferably dispersed as fine particles in an aqueous medium.
- the fine particles of the copolymer (A) are particularly preferably fine particles obtained by emulsion polymerization of a monomer in an aqueous medium.
- the average particle size of the copolymer (A) is preferably 10 to 1000 nm, more preferably 10 to 300 nm, and particularly preferably 10 to 250 nm. If the average particle diameter is within this range, it is not necessary to use a large amount of a surfactant or the like, the water repellency is good, and no discoloration occurs when dyed fabrics are treated. Thus, the dispersed particles can exist stably and do not settle.
- the average particle size of the copolymer (A) is measured by a dynamic light scattering device, an electron microscope or the like.
- the aqueous medium contains water, at least one film-forming aid selected from the film-forming aid (x) and the film-forming aid (y), and other media as necessary. Since the film-forming auxiliary (x) and the film-forming auxiliary (y) have good compatibility with the copolymer (A), the copolymer (A) can be effectively swollen or dissolved. . Furthermore, since the film-forming aid (x) and the film-forming aid (y) have a relatively high boiling point, they are less likely to evaporate than water when air-dried.
- the copolymer (A) is penetrated by the film-forming auxiliary (x) or the film-forming auxiliary (y) even in the portion where the film made of the polymer (A) is difficult to form, and the uniform film is formed by the copolymer (A). Can be formed. As a result, sufficient water and oil repellency can be imparted to the article after air drying.
- the water / oil repellent composition of the present invention contains at least one film-forming aid selected from a film-forming aid (x) and a film-forming aid (y).
- a film-forming aid selected from a film-forming aid (x) and a film-forming aid (y).
- the film-forming auxiliary (x) and the film-forming auxiliary (y) are collectively referred to as a film-forming auxiliary.
- the film-forming auxiliary (x) is the compound (5).
- r is an integer of 0 to 3
- s is 0 or 1
- R 1 is an alkyl group having 1 to 4 carbon atoms
- R 2 and R 3 are each independently 1 to 3 carbon atoms.
- An alkyl group (which may contain an etheric oxygen atom)
- R 4 is a hydrogen atom or a methyl group
- R 1 may be linear or branched, and is preferably linear.
- R 1 include a methyl group, an ethyl group, a propyl group, an isopropyl group, and an n-butyl group, and an n-butyl group is preferable.
- R 2 and R 3 include a methyl group, an ethyl group, a propyl group, an isopropyl group, a methoxyethyl group, and the like, and all are preferably a methyl group.
- R 4 is preferably a hydrogen atom, and r + s is preferably 1 or 2.
- Examples of the compound (5) include the following compounds.
- 3-alkoxy-N, N-dialkylpropionamide is preferable from the viewpoint of excellent compatibility with the copolymer (A), and 3-n-butoxy-N, N-dimethylpropionamide ( Boiling point: 252 ° C.), 3-methoxy-N, N-dimethylpropionamide (boiling point: 204 ° C.) is more preferable, and 3-n-butoxy-N, N-dimethylpropionamide is particularly preferable.
- Film-forming aid (y) is the compound (6).
- t is 3 or 4
- R 5 and R 6 are each independently an alkyl group having 1 to 4 carbon atoms.
- R 5 and R 6 may be linear or branched and are preferably linear.
- R 5 and R 6 include a methyl group, an ethyl group, a propyl group, an isopropyl group, and an n-butyl group, and a methyl group is preferable.
- Examples of the compound (6) include the following compounds.
- Tetraethylene glycol dimethyl ether Tetraethylene glycol dimethyl ether, triethylene glycol dimethyl ether, tetraethylene glycol diethyl ether, triethylene glycol diethyl ether, tetraethylene glycol di n-propyl ether, triethylene glycol di n-propyl ether, tetraethylene glycol isopropyl ether, triethylene glycol diisopropyl Ether, tetraethylene glycol di-n-butyl ether, triethylene glycol di-n-butyl ether and the like.
- tetraethylene glycol dimethyl ether (boiling point: 275 ° C.) and triethylene glycol dimethyl ether (boiling point: 216 ° C.) are preferable from the viewpoint of excellent compatibility with the copolymer (A), and tetraethylene glycol dimethyl ether. Is particularly preferred.
- the boiling points of the film-forming auxiliary (x) and the film-forming auxiliary (y) are each preferably 100 ° C. or higher, more preferably 200 ° C. or higher, and particularly preferably 250 ° C. or higher. If the boiling point is 100 ° C. or higher, the film-forming aid can form a uniform copolymer (A) film on the substrate when air-dried, and can further improve the water / oil repellency after air-drying.
- the ratio of the film-forming auxiliary in the aqueous medium is 0.1 to 0.1% of the aqueous medium (B) (100% by mass). 20% by mass is preferable, 0.5 to 10% by mass is more preferable, and 1 to 5% by mass is particularly preferable. If the ratio of the film-forming aid is 0.1% by mass or more, the water / oil repellency after air drying can be further improved. When the proportion of the film-forming aid is 20% by mass or less, the stability of the emulsified state is improved, precipitation or the like in the water / oil repellent composition is unlikely to occur, and an ideal processing state can be realized.
- the amount of the film-forming auxiliary with respect to the copolymer (A), that is, the total amount of the film-forming auxiliary (x) and the film-forming auxiliary (y) with respect to the copolymer (A) depends on the copolymer (A).
- the amount is preferably 10 to 4000 parts by mass, more preferably 50 to 2000 parts by mass, still more preferably 300 to 1000 parts by mass, and particularly preferably 500 to 1000 parts by mass with respect to 100 parts by mass.
- the amount of the film-forming auxiliary is 10 parts by mass or more, the compatibility with the copolymer (A) is improved, and a uniform film is easily formed on the surface of the article.
- the amount of the film-forming auxiliary is 4000 parts by mass or less, a uniform copolymer (A) film can be formed on the substrate, and water and oil repellency can be imparted to the article after one layer of air drying. .
- Other media include alcohols, glycols, glycol ethers, halogen compounds, ketones, esters, ethers, nitrogen compounds, sulfur compounds, organic acids, and the like.
- Alcohols include methanol, ethanol, 1-propanol, 2-propanol, 1-butanol, 2-butanol, 2-methylpropanol, 1,1-dimethylethanol, 1-pentanol, 2-pentanol, and 3-pentanol.
- glycol and glycol ether ethylene glycol, ethylene glycol monomethyl ether, ethylene glycol monoethyl ether, ethylene glycol monobutyl ether, ethylene glycol monomethyl ether acetate, ethylene glycol monoethyl ether acetate, ethylene glycol monobutyl ether acetate, hexylene Glycol, propylene glycol, dipropylene glycol, dipropylene glycol monomethyl ether, propylene glycol monomethyl ether acetate, propylene glycol monomethyl ether, propylene glycol monoethyl ether, propylene glycol dimethyl ether, dipropylene glycol dimethyl ether, dipropylene glycol monoether Ether, tripropylene glycol, tripropylene glycol monomethyl ether, polypropylene glycol, and the like.
- halogen compound examples include halogenated hydrocarbons and halogenated ethers.
- halogenated hydrocarbon examples include hydrochlorofluorocarbon, hydrofluorocarbon, hydrobromocarbon and the like.
- Examples of the halogenated ether include hydrofluoroether.
- Examples of hydrofluoroethers include separated hydrofluoroethers and non-separable hydrofluoroethers.
- the separated hydrofluoroether is a compound in which an R F group or a perfluoroalkylene group, and an alkyl group or an alkylene group are bonded via an etheric oxygen atom.
- the non-separable hydrofluoroether is a hydrofluoroether containing a partially fluorinated alkyl group or alkylene group.
- Examples of the ketone include acetone, methyl ethyl ketone, 2-pentanone, 3-pentanone, 2-hexanone, and methyl isobutyl ketone.
- Examples of the ester include methyl acetate, ethyl acetate, butyl acetate, methyl propionate, methyl lactate, ethyl lactate, and pentyl lactate.
- Examples of the ether include diisopropyl ether, dioxane, THF and the like.
- nitrogen compounds include formamide, monomethylformamide, dimethylformamide, monoethylformamide, diethylformamide, amides such as N, N-dimethylformamide, acetamide, N, N-dimethylacetamide, N-methylpyrrolidone, pyridine, and the like.
- sulfur compound include dimethyl sulfoxide and sulfolane.
- organic acid include acetic acid, propionic acid, malic acid, and lactic acid.
- the proportion of the other medium is preferably 50% by mass or less, more preferably 40% by mass or less, and particularly preferably 0 to 30% by mass in the aqueous medium (100% by mass).
- the water / oil repellent composition of the present invention usually contains a surfactant.
- This surfactant is required for stably dispersing the fine particles of the copolymer (A) in the aqueous medium (B).
- a surfactant is added to the aqueous medium used for emulsion polymerization.
- At least a part of the surfactant in the water / oil repellent composition of the present invention is the surfactant used in the polymerization.
- a surfactant can be further added to the emulsion obtained by emulsion polymerization.
- the surfactant examples include a hydrocarbon-based surfactant and a fluorine-based surfactant, and an anionic surfactant, a nonionic surfactant, a cationic surfactant, and an amphoteric surfactant, respectively.
- a combination of a nonionic surfactant and an amphoteric surfactant is preferable from the viewpoint of compatibility with the additive, and a nonionic surfactant is used alone from the viewpoint of adhesion to the substrate, and A combination of a nonionic surfactant and a cationic surfactant is preferred.
- the ratio of the nonionic surfactant to the cationic surfactant is preferably 97/3 to 40/60 (mass ratio).
- the nonionic surfactant is preferably at least one selected from the group consisting of surfactants s 1 to s 6 .
- the surfactant s 1 is a polyoxyalkylene monoalkyl ether, a polyoxyalkylene monoalkenyl ether, a polyoxyalkylene monoalkapolyenyl ether, or a polyoxyalkylene monopolyfluoroalkyl ether.
- the surfactant s 1 is preferably polyoxyalkylene monoalkyl ether, polyoxyalkylene monoalkenyl ether, or polyoxyalkylene monopolyfluoroalkyl ether.
- Surfactant s 1 may be used alone or in combination of two or more thereof.
- the alkyl group, alkenyl group, alkapolyenyl group or polyfluoroalkyl group (hereinafter, the alkyl group, alkenyl group, alkapolyenyl group and polyfluoroalkyl group are collectively referred to as R s group) has 4 carbon atoms. Groups of ⁇ 26 are preferred.
- the R s group may be linear or branched.
- the branched R s group is preferably a secondary alkyl group, a secondary alkenyl group, or a secondary alkapolyenyl group.
- part or all of the hydrogen atoms may be substituted with fluorine atoms.
- R s group examples include octyl group, dodecyl group, tetradecyl group, hexadecyl group, stearyl group (octadecyl group), behenyl group (docosyl group), oleyl group (9-octadecenyl group), heptadecylfluorooctyl group, Examples thereof include a tridecylfluorohexyl group, 1H, 1H, 2H, 2H-tridecylfluorooctyl group, 1H, 1H, 2H, 2H-nonafluorohexyl group and the like.
- the polyoxyalkylene (hereinafter referred to as POA) chain includes a chain in which two or more polyoxyethylene (hereinafter referred to as POE) chains and / or polyoxypropylene (hereinafter referred to as POP) chains are connected.
- POA chain may be a chain composed of one kind of POA chain or a chain composed of two or more kinds of POA chains. When composed of two or more POA chains, each POA chain is preferably linked in a block form.
- R 10 O [CH 2 CH (CH 3 ) O] u2 — (CH 2 CH 2 O) u1 H (s 11 ).
- R 10 is an alkyl group having 8 or more carbon atoms or an alkenyl group having 8 or more carbon atoms, u1 is an integer of 5 to 50, and u2 is an integer of 0 to 20.
- a part of hydrogen atoms may be substituted with fluorine atoms.
- u1 is 5 or more, it is soluble in water and uniformly dissolved in an aqueous medium, so that the permeability of the water / oil repellent composition to the article is good.
- u1 is 50 or less, hydrophilicity is suppressed and water repellency is improved.
- u2 is 20 or less, it becomes soluble in water and is uniformly dissolved in an aqueous medium, so that the permeability of the water / oil repellent composition to the article is good.
- R 10 is preferably linear or branched.
- u1 is preferably an integer of 10 to 30.
- u2 is preferably an integer of 0 to 10.
- Examples of the compound (s 11 ) include the following compounds. However, the POE chain and the POP chain are linked in a block shape. C 18 H 37 O [CH 2 CH (CH 3 ) O] 2 — (CH 2 CH 2 O) 30 H, C 17 H 35 O— (CH 2 CH 2 O) 30 H, C 16 H 33 O [CH 2 CH (CH 3 ) O] 5 — (CH 2 CH 2 O) 20 H, C 12 H 25 O [CH 2 CH (CH 3 ) O] 2 — (CH 2 CH 2 O) 15 H, (C 8 H 17 ) (C 6 H 13 ) CHO— (CH 2 CH 2 O) 15 H, C 10 H 21 O [CH 2 CH (CH 3 ) O] 2 — (CH 2 CH 2 O) 15 H, C 6 F 13 CH 2 CH 2 O— (CH 2 CH 2 O) 15 H, C 6 F 13 CH 2 CH 2 O [CH 2 CH (CH 3) O] 2 - (CH 2 CH 2 O) 15 H, C 4 F 9 CH 2 CH 2 O [
- Surfactant s 2 Surfactant s 2, one or more carbons in the molecule - is a nonionic surfactant made of a compound having a carbon triple bond and at least one hydroxy group.
- the surfactant s 2 is preferably a nonionic surfactant made of a compound having one carbon-carbon triple bond and one or two hydroxyl groups in the molecule.
- Surfactant s 2 may have a POA chain in the molecule. Examples of the POA chain include a POE chain, a POP chain, a chain in which a POE chain and a POP chain are connected randomly, or a chain in which a POE chain and a POP chain are connected in a block form.
- compounds (s 21 ) to (s 24 ) are preferable.
- a 1 to A 3 are each an alkylene group.
- v and w are each an integer of 0 or more, and (v + w) is an integer of 1 or more.
- x is an integer of 1 or more.
- the POA chain is preferably a POE chain, a POP chain, or a chain containing a POE chain and a POP chain.
- the number of repeating units of the POA chain is preferably 1-50.
- R 11 to R 16 are each a hydrogen atom or an alkyl group.
- the alkyl group an alkyl group having 1 to 12 carbon atoms is preferable, and an alkyl group having 1 to 4 carbon atoms is more preferable.
- the alkyl group include a methyl group, an ethyl group, a propyl group, a butyl group, and an isobutyl group.
- the compound (s 22 ) is preferable.
- y and z are each an integer of 0 to 100.
- Compound (s 25) may be used alone or in combination of two or more thereof.
- Surfactant s 3 is a nonionic surfactant comprising a compound in which a POE chain is linked to a POA chain in which two or more oxyalkylenes having 3 or more carbon atoms are continuously linked, and both ends are hydroxyl groups. It is.
- the POA chain is preferably polyoxybutene (hereinafter referred to as POB) and / or POP chain.
- Surfactant s 3, compound (s 31) or compound (s 32) is preferably. HO (CH 2 CH 2 O) g 1 (C 3 H 6 O) g 2 (CH 2 CH 2 O) g 3 H (s 31 ), HO (CH 2 CH 2 O) g1 (CH 2 CH 2 CH 2 CH 2 O) g2 (CH 2 CH 2 O) g3 H ⁇ (s 32).
- g1 is an integer of 0 to 200.
- g2 is an integer of 2 to 100.
- g3 is an integer of 0 to 200.
- g1 is 0, g3 is an integer of 2 or more.
- g1 is an integer of 2 or more.
- —C 3 H 6 O— may be —CH (CH 3 ) CH 2 O—, may be —CH 2 CH (CH 3 ) O—, and —CH (CH 3 ) CH 2 O—.
- a mixture of — and —CH 2 CH (CH 3 ) O— may be used.
- the POA chain is block-shaped.
- Examples of the surfactant s 3 include the following compounds. HO— (CH 2 CH 2 O) 15 — (C 3 H 6 O) 35 — (CH 2 CH 2 O) 15 H, HO— (CH 2 CH 2 O) 8 — (C 3 H 6 O) 35 — (CH 2 CH 2 O) 8 H, HO— (CH 2 CH 2 O) 45 — (C 3 H 6 O) 17 — (CH 2 CH 2 O) 45 H, HO— (CH 2 CH 2 O) 34 — (CH 2 CH 2 CH 2 CH 2 O) 28 — (CH 2 CH 2 O) 34 H.
- Surfactant s 4 is a nonionic surfactant having an amine oxide moiety in the molecule.
- Surfactant s 4, compound (s 41) is preferably. (R 17 ) (R 18 ) (R 19 ) N ( ⁇ O) (s 41 ).
- R 17 to R 19 are each a monovalent hydrocarbon group.
- a surfactant having amine oxide (N ⁇ O) is treated as a nonionic surfactant.
- Compound (s 41) may be used alone or in combination of two or more thereof.
- the compound (s 41 ) is preferable from the viewpoint of dispersion stability of the copolymer (A).
- (R 20 ) (CH 3 ) 2 N ( ⁇ O) (s 42 ).
- R 20 is an alkyl group having 6 to 22 carbon atoms, an alkenyl group having 6 to 22 carbon atoms, a phenyl group to which an alkyl group having 6 to 22 carbon atoms is bonded, or an alkenyl group having 6 to 22 carbon atoms. Or a polyfluoroalkyl group having 6 to 13 carbon atoms.
- R 20 is preferably an alkyl group having 8 to 22 carbon atoms, an alkenyl group having 8 to 22 carbon atoms, or a polyfluoroalkyl group having 4 to 9 carbon atoms.
- Examples of the compound (s 42 ) include the following compounds. [H (CH 2 ) 12 ] (CH 3 ) 2 N ( ⁇ O), [H (CH 2 ) 14 ] (CH 3 ) 2 N ( ⁇ O), [H (CH 2 ) 16 ] (CH 3 ) 2 N ( ⁇ O), [H (CH 2 ) 18 ] (CH 3 ) 2 N ( ⁇ O), [F (CF 2 ) 6 (CH 2 ) 2 ] (CH 3 ) 2 N ( ⁇ O), [F (CF 2 ) 4 (CH 2 ) 2 ] (CH 3 ) 2 N ( ⁇ O).
- Surfactant s 5 is a nonionic surfactant made of a polyoxyethylene mono (substituted phenyl) ether condensate or polyoxyethylene mono (substituted phenyl) ether.
- the substituted phenyl group is preferably a phenyl group substituted with a monovalent hydrocarbon group, and more preferably a phenyl group substituted with an alkyl group, an alkenyl group or a styryl group.
- Surfactant s 5 includes polyoxyethylene mono (alkylphenyl) ether condensate, polyoxyethylene mono (alkenylphenyl) ether condensate, polyoxyethylene mono (alkylphenyl) ether, polyoxyethylene mono (alkenyl). Phenyl) ether or polyoxyethylene mono [(alkyl) (styryl) phenyl] ether is preferred.
- Polyoxyethylene mono (substituted phenyl) ether condensate or polyoxyethylene mono (substituted phenyl) ether includes formaldehyde condensate of polyoxyethylene mono (nonylphenyl) ether, polyoxyethylene mono (nonylphenyl) ether, poly Oxyethylene mono (octylphenyl) ether, polyoxyethylenemono (oleylphenyl) ether, polyoxyethylenemono [(nonyl) (styryl) phenyl] ether, polyoxyethylenemono [(oleyl) (styryl) phenyl] ether, etc. Can be mentioned.
- Surfactant s 6 is a nonionic surfactant made of a fatty acid ester of a polyol.
- the polyol represents glycerin, sorbitan, sorbit, polyglycerin, polyethylene glycol, polyoxyethylene glyceryl ether, polyoxyethylene sorbitan ether, polyoxyethylene sorbit ether.
- a 1: 1 (molar ratio) ester of stearic acid and polyethylene glycol As the surfactant s 6 , a 1: 1 (molar ratio) ester of stearic acid and polyethylene glycol, a 1: 4 (molar ratio) ester of ether of sorbite and polyethylene glycol and oleic acid, polyoxyethylene glycol, 1: 1 (molar ratio) ester of ether of sorbitan and stearic acid, 1: 1 (molar ratio) ester of ether of polyethylene glycol and sorbitan and oleic acid, 1: 1 (molar ratio) of dodecanoic acid and sorbitan Ratio) ester, 1: 1 or 2: 1 (molar ratio) ester of oleic acid and decaglycerin, and 1: 1 or 2: 1 (molar ratio) ester of stearic acid and decaglycerin.
- Surfactant s 7 When the surfactant includes a cationic surfactant, the surfactant s 7 is preferable as the cationic surfactant. Surfactant s 7 is a cationic surfactant in the form of a substituted ammonium salt.
- the surfactant s 7 is preferably an ammonium salt in which one or more hydrogen atoms bonded to a nitrogen atom are substituted with an alkyl group, an alkenyl group, or a POA chain having a terminal hydroxyl group, and the compound (s 71 ) is more preferable. preferable. [(R 21) 4 N + ] ⁇ X - ⁇ (s 71).
- R 21 is a hydrogen atom, an alkyl group having 1 to 22 carbon atoms, an alkenyl group having 2 to 22 carbon atoms, a polyfluoroalkyl group having 1 to 9 carbon atoms, or a POA chain having a terminal hydroxyl group.
- the four R 21 may be the same or different, but the four R 21 are not hydrogen atoms at the same time.
- R 21 is preferably a long-chain alkyl group having 6 to 22 carbon atoms, a long-chain alkenyl group having 6 to 22 carbon atoms, or a fluoroalkyl group having 1 to 9 carbon atoms.
- R 21 is an alkyl group other than a long-chain alkyl group
- R 21 is preferably a methyl group or an ethyl group.
- R 21 is a POA chain having a hydroxyl group at the end, the POA chain is preferably a POE chain.
- X ⁇ is a counter ion.
- X ⁇ is preferably a chlorine ion, an ethyl sulfate ion, or an acetate ion.
- Examples of the compound (s 71 ) include monostearyl trimethylammonium chloride, monostearyldimethylmonoethylammonium ethyl sulfate, mono (stearyl) monomethyldi (polyethylene glycol) ammonium chloride, monofluorohexyltrimethylammonium chloride, di (tallow alkyl) dimethylammonium. Examples include chloride and dimethyl monococonut amine acetate.
- Surfactant s 8 When the surfactant contains an amphoteric surfactant, the amphoteric surfactant, surfactant s 8 is preferred.
- Surfactant s 8 is alanine compounds, imidazolinium betaines, amide betaines or betaine acetate.
- the hydrophobic group is preferably a long-chain alkyl group having 6 to 22 carbon atoms, a long-chain alkenyl group having 6 to 22 carbon atoms, or a polyfluoroalkyl group having 1 to 9 carbon atoms.
- Surfactant s 8 dodecyl betaine, stearyl betaine, dodecyl carboxymethyl hydroxyethyl imidazolinium betaine, dodecyl dimethylamino betaine, such as fatty acid amidopropyl dimethylamino acetic acid betaine.
- lauryldimethylaminoacetic acid betaine aqueous solution (NIKKOL AM-301, manufactured by Nikko Chemicals), coconut oil fatty acid amidopropyldimethylaminoacetic acid betaine aqueous solution (manufactured by Nikko Chemicals, NIKKOL AM-3130N), N-coconut oil fatty acid acyl-N. -Carboxymethyl-N-hydroxyethyl ethylenediamine sodium aqueous solution (Nikko Chemicals NIKKOL AM-101) and the like.
- Surfactant s 9 is a block copolymer, random copolymer, or hydrophilic copolymer of a hydrophilic monomer and a hydrocarbon-based hydrophobic monomer and / or a fluorine-based hydrophobic monomer. It is a polymer surfactant comprising a hydrophobic modified product.
- Surfactant s 9 a block or random copolymer derived from polyethylene glycol (meth) acrylate with a long chain alkyl acrylate, a block or random copolymer derived from polyethylene glycol (meth) acrylate and fluoro (meth) acrylate, acetate Block or random copolymer of vinyl and long chain alkyl vinyl ether, block or random copolymer of vinyl acetate and long chain alkyl vinyl ester, polymer of styrene and maleic anhydride, condensation of polyvinyl alcohol and stearic acid Products, condensation products of polyvinyl alcohol and stearyl mercaptan, condensation products of polyallylamine and stearic acid, condensation products of polyethyleneimine and stearyl alcohol, methyl cellulose, hydroxypropyl methylcellulose And hydroxyethyl methylcellulose.
- surfactant s 9 includes Kuraray's MP polymer (product numbers: MP-103, MP-203), Elf Atchem's SMA resin, Shin-Etsu Chemical's Metros, Nippon Shokubai's Epomin RP, and Seimi Chemical.
- the company's Surflon product numbers: S-381, S-393) and the like.
- Surfactant s 9 when the medium is an organic solvent or when the mixing ratio of the organic solvent is large, the surfactant s 91 is preferable.
- Surfactant s 91 a polymer surfactant comprising a block copolymer or a random copolymer (modified polyfluoroalkyl thereof) of a lipophilic monomer and a fluorine monomer.
- Surfactant s 91 includes a copolymer of alkyl acrylate and fluoro (meth) acrylate, a copolymer of alkyl vinyl ether and fluoroalkyl vinyl ether, and the like.
- a commercially available product of Surfactant s 91 is Surflon (product number: S-383, SC-100 series) manufactured by Seimi Chemical Co., Ltd.
- a combination of surfactant s 1 , surfactant s 3 and surfactant s 7 or a combination of surfactant s 1 , surfactant s 2 , surfactant s 3 and surfactant s 7 is preferred,
- the above combination in which the surfactant s 7 is the compound (s 71 ) is more preferable.
- surfactant s 1 and / or a combination of surfactant s 2 and surfactant s 8 , or surfactant s 1 , surfactant s 2 and surfactant s the combination of 3 and a surfactant s 8 is preferred.
- HLB is a value representing the degree of affinity of a surfactant with water and oil, and takes a value of 0 to 20. The smaller the value, the more lipophilic, and the larger the value, the more hydrophilic.
- Several methods have been proposed for calculating this value. For example, in the Griffin method, it is defined as the sum of the formula weight of 20 ⁇ hydrophilic part / molecular weight.
- surfactant having an HLB of 12 or less the following are preferable.
- Surfactant s 1 Of compounds (s 11 ), a compound in which s + r is an integer of 2 to 10 (preferably 3 to 8) and r is an integer of 2 to 10 (preferably 3 to 8) is preferable .
- Surfactant s 2 Of the compounds (s 25 ), compounds wherein x + y is 4 or less are preferred.
- Surfactant s 6 Among sorbitan fatty acid esters, compounds in which the fatty acid has 5 to 30 carbon atoms are preferred.
- the amount of the surfactant is preferably 1 to 10 parts by mass, more preferably 1 to 9 parts by mass, and particularly preferably 1 to 7 parts by mass with respect to 100 parts by mass of the copolymer (A).
- Additives include penetrants, antifoaming agents, water absorbing agents, antistatic agents, antistatic polymers, antifungal agents, texture modifiers, water-soluble polymers (polyacrylamide, polyvinyl alcohol, etc.), thermosetting agents ( Melamine resin, urethane resin, triazine ring-containing compound, isocyanate compound, etc.), epoxy curing agent (isophthalic acid dihydrazide, adipic acid dihydrazide, sebacic acid dihydrazide, dodecanedioic acid dihydrazide, 1,6-hexamethylenebis (N, N-) Dimethyl semicarbazide), 1,1,1 ′, 1′-tetramethyl-4,4 ′-(methylene-di-para-phenylene) disemicarbazide, spiroglycol, etc.), thermosetting catalyst, crosslinking catalyst (organic acids, chloride) Ammonium), synthetic resins, fiber stabilizers, inorganic fine particles, and
- the water / oil repellent composition of the present invention may be a copolymer other than the copolymer (A) that can exhibit water repellency and / or oil repellency, if necessary (for example, a commercially available water repellent, a commercially available water repellent).
- a commercially available water repellent for example, a commercially available water repellent, a commercially available water repellent.
- oil repellent commercially available water / oil repellent, commercially available SR agent, etc.
- a water repellent compound having no fluorine atom examples include paraffinic compounds, aliphatic amide compounds, alkylethylene urea compounds, silicone compounds, and the like.
- the water / oil repellent composition of the present invention is produced, for example, by the following method ( ⁇ ) or ( ⁇ ).
- ⁇ After the monomer component is polymerized in an aqueous medium in the presence of a surfactant and a polymerization initiator to produce a dispersion or emulsion of the copolymer (A), a film-forming aid ( A method of blending at least one film-forming aid selected from x) and a film-forming aid (y).
- a medium such as water, a surfactant, an additive, or the like can be blended with the produced dispersion or emulsion as necessary.
- a method for producing a dispersion or emulsion of a copolymer (A) by polymerizing a body component If necessary, a medium such as water, a surfactant, an additive, or the like can be added to the produced dispersion or emulsion. Moreover, at least 1 type of film-forming aid chosen from film-forming aid (x) and film-forming aid (y) can also be mix
- Examples of the polymerization method include a dispersion polymerization method, an emulsion polymerization method, a suspension polymerization method, and the like, and emulsion polymerization is preferable.
- the polymerization method may be batch polymerization or multistage polymerization.
- As a polymerization method an emulsion of a copolymer (A) is obtained by emulsion polymerization of a monomer component in an aqueous medium containing water and, if necessary, another medium in the presence of a surfactant and a polymerization initiator. Is preferred.
- a mixture comprising a monomer, a surfactant and an aqueous medium is mixed and dispersed with an ultrasonic stirrer, a homomixer or a high-pressure emulsifier.
- polymerization initiator examples include a thermal polymerization initiator, a photopolymerization initiator, a radiation polymerization initiator, a radical polymerization initiator, an ionic polymerization initiator, and the like, and a water-soluble or oil-soluble radical polymerization initiator is preferable.
- radical polymerization initiator a general-purpose initiator such as an azo polymerization initiator, a peroxide polymerization initiator, or a redox initiator is used depending on the polymerization temperature.
- an azo compound is particularly preferable, and when polymerization is performed in an aqueous medium, a salt of the azo compound is more preferable.
- azo compounds and salts of azo compounds include 2,2′-azobis (4-methoxy-2,4-dimethylvaleronitrile), 2,2′-azobis (2,4-dimethylvaleronitrile), dimethyl 2, 2'-azobis (2-methylpropionate), 2,2'-azobis (2-methylbutyronitrile), 2,2'-azobis (2-methylpropionamide) dihydrochloride, 2,2'- Azobis [2- (2-imidazolin-2-yl) propane] disulfate, 2,2′-azobis [2- (2-imidazolin-2-yl) propane] dihydrochloride, 2,2′-azobis [ 2- (2-imidazolin-2-yl) propane] acetate, 2,2′-azobis [N- (2-carboxyethyl) 2-methylpropionamidine] hydrate, 2,2′-azobis (1- Imino 1-pyrrolidino -2-methylpropane) dihydrochloride.
- the polymerization temperature is
- a molecular weight modifier may be used.
- aromatic compounds, mercapto alcohols or mercaptans are preferable, and alkyl mercaptans are particularly preferable.
- molecular weight modifiers include mercaptoethanol, n-octyl mercaptan, n-dodecyl mercaptan, t-dodecyl mercaptan, stearyl mercaptan, thioglycerol, ⁇ -methylstyrene dimer (CH 2 ⁇ C (Ph) CH 2 C (CH 3 ) 2 Ph and Ph are phenyl groups), diethylene glycol bis (3-mercaptobutyrate), pentaerythritol tetrakis (3-mercaptobutyrate), 2,4,6-trimercaptotriazine, 1,3,5-tris And polyfunctional mercapto compounds such as (3-mercaptobutyl
- the proportion of the monomers (a) to (e) is used in the same manner as the proportion of the structural units based on the monomers (a) to (e) because these monomers are copolymerized at almost 100%.
- the preferred embodiments are also the same.
- the aqueous medium used for polymerizing the monomer component does not contain the film-forming aid and is made of water or, in some cases, a mixture of a medium other than water and water.
- the film-forming aid By blending the film-forming aid into the dispersion or emulsion obtained by polymerization, the aqueous medium in the dispersion or emulsion after blending becomes the aqueous medium (B).
- water and other media can be blended and diluted in the dispersion or emulsion obtained by polymerization.
- the said surfactant and additive can also be mix
- the surfactant to be blended may be the same type of surfactant as the surfactant added to the aqueous medium during polymerization or a different surfactant. Some or all of the additives can be added to the aqueous medium used in the polymerization.
- the aqueous medium used for polymerizing the monomer component contains the film-forming aid, and the aqueous medium in the dispersion or emulsion obtained by polymerization is the aqueous medium (B). .
- the film-forming aid, water, and other media can be further blended and diluted as necessary in the dispersion or emulsion obtained by polymerization.
- the said surfactant and additive can also be mix
- the surfactant to be blended may be the same type of surfactant as the surfactant added to the aqueous medium used in the polymerization or a different surfactant. Some or all of the additives can be added to the aqueous medium used in the polymerization.
- the polymerization method is preferably emulsion polymerization, and an emulsion in which the fine particles of the copolymer (A) are stably dispersed in the aqueous medium is obtained by emulsion polymerization. It is done.
- This emulsion has good dispersion stability even when water, a film-forming aid, other media, or the like is added thereto.
- the water / oil repellent composition of the present invention is preferably an emulsion produced by the method ( ⁇ ). In particular, by the method ( ⁇ ), it is possible to easily obtain a water / oil repellent composition having a relatively large amount of a film-forming aid and having good dispersion stability.
- the ratio of the film-forming auxiliary can be adjusted more easily than the method ( ⁇ ) according to the type of article and the processing conditions for the article. Furthermore, the adjustment of the composition of the aqueous medium in the water / oil repellent composition is easy.
- the optimum amount of the film-forming aid can be easily set according to the type of article such as cloth and the processing conditions (bath temperature, air drying temperature, etc.).
- the solid content concentration of the emulsion is preferably 20 to 40% by mass in the emulsion (100% by mass) immediately after emulsion polymerization (that is, before dilution).
- concentration is a density
- the proportion of the copolymer (A) in the emulsion is preferably 18 to 40% by mass immediately after the emulsion polymerization.
- the solid content concentration of the water / oil repellent composition of the present invention is preferably 0.2 to 5% by mass in the water / oil repellent composition (100% by mass) when the article is processed. For this reason, usually emulsions produced by emulsion polymerization are diluted with one or more of water, film-forming aids, or other media, and the diluted emulsion is used to treat the article.
- the solid content concentration of the emulsion or water / oil repellent composition is calculated from the mass of the emulsion or water / oil repellent composition before heating and the mass after drying for 4 hours in a convection dryer at 120 ° C. .
- the water / oil repellent composition of the present invention described above contains at least one selected from water, a film-forming aid (x) and a film-forming aid (y), including the copolymer (A). Since the aqueous medium (B) containing a seed film-forming auxiliary is an essential component, sufficient air and water / oil repellency can be imparted to the surface of the article after air drying.
- copolymer (A) since it does not have structural units based on a monomer having an R F group number is 7 or more carbon atoms, to the environment
- the content of perfluorooctanoic acid (PFOA), perfluorooctanesulfonic acid (PFOS), its precursor, and analogs (contents when the solid content concentration is 20% by mass), which has been pointed out to be affected, is disclosed in International Publication No. 2009
- the analysis value of LC-MS / MS by the method described in Japanese Patent No. 0881822 can be made below the detection limit.
- the article of the present invention is an article having a surface treated with the water / oil repellent composition of the present invention and having a coating of the copolymer (A) on the surface.
- articles to be treated include fibers (natural fibers, synthetic fibers, blended fibers, etc.), various fiber products, nonwoven fabrics, resins, paper, leather, metals, stones, concrete, gypsum, glass and the like.
- a coating solution containing a water / oil repellent composition is applied to an article by a known coating method and then dried, or the article is immersed in a coating liquid containing a water / oil repellent composition And then drying.
- waterproofing include processing for providing a waterproof film.
- the waterproof membrane include a porous membrane obtained from a urethane resin or an acrylic resin, a nonporous membrane obtained from a urethane resin or an acrylic resin, a polytetrafluoroethylene membrane, or a moisture-permeable waterproof membrane combining these.
- a film of the copolymer (A) is formed on the article surface, and water / oil repellency can be imparted to the article.
- the formed coating is excellent in adhesion to the article surface, and water and oil repellency can be imparted even by curing at a low temperature. Moreover, there is little deterioration in performance due to friction or washing, and the initial performance can be stably maintained.
- excellent size and water / oil repellency can be imparted to the paper even under low temperature drying conditions.
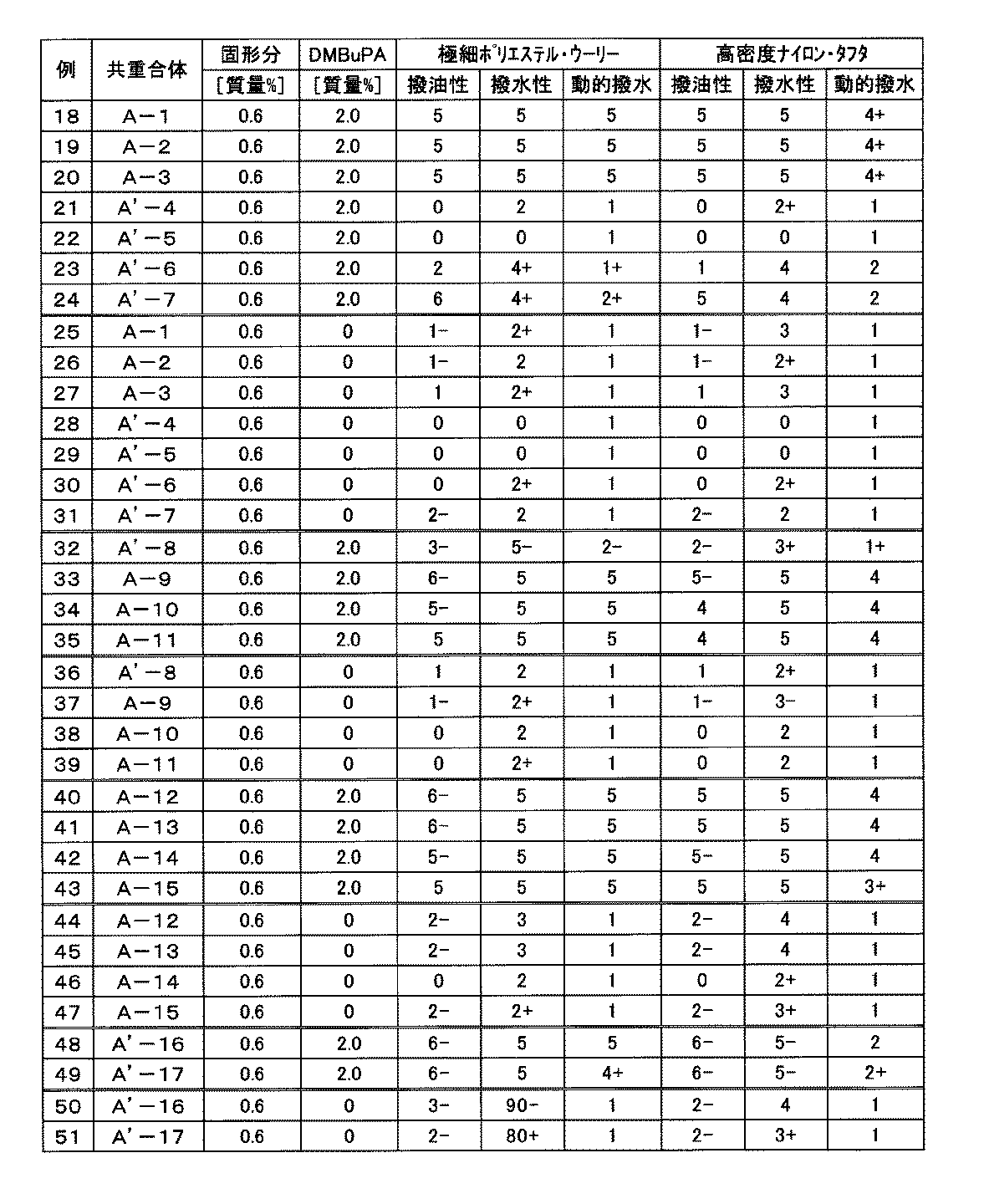
- Examples 1 to 17 are production examples, examples 18 to 20, 33 to 35, 40 to 43, 52, 53, and 57 are examples, and examples 21 to 32, 36 to 39, 44 to 51, and 54 to 56 are examples.
- 58 to 65 are comparative examples.
- VdCl Vinylidene chloride (Tg: -18 ° C).
- VCl Vinyl chloride (Tg: 82 ° C.)
- VA behenyl acrylate (Tg: 66 ° C.)
- StA stearyl acrylate (Tg: 47 ° C.)
- StMA Stearyl methacrylate (Tg: 35 ° C.)
- CetMA Cetyl methacrylate (Tg: 23.5 ° C.)
- CyhMA cyclohexyl methacrylate (Tg: 66 ° C.)
- n-BuMA n-butyl methacrylate (Tg: 20 ° C.)
- IB-X Isobornyl methacrylate (Tg: 180 ° C.).
- (Surfactant) E-430 polyoxyethylene oleyl ether (manufactured by Kao Corporation, Emulgen E430), SFN-465: 2,4,7,9-tetramethyl-5-decyne-4,7-diol ethylene oxide adduct (manufactured by Nissin Chemical Industry Co., Ltd., Surfinol 465), P-204: ethylene oxide propylene oxide polymer (manufactured by NOF Corporation, Pronon 204), Aq-18: Stearyl trimethylammonium chloride (manufactured by Lion Corporation, ARCARD 18-63), Aq-C: Lauryltrimethylammonium chloride (manufactured by Lion Corporation, ARCARD C-50).
- StSH Stearyl mercaptan
- VA-061A 2,2′-azobis [2- (2-imidazolin-2-yl) propane] (Wako Pure Chemical Industries, Ltd., VA-061) acetate.
- DMBuPA 3-n-butoxy-N, N-dimethylpropionamide (manufactured by Idemitsu Kosan Co., Ltd., Ecamide B100, boiling point: 252 ° C.).
- TeEGDME Tetraethylene glycol dimethyl ether (boiling point: 275 ° C.).
- DPG Dipropylene glycol (boiling point: 232 ° C.)
- EGDMA ethylene glycol dimethyl ether (boiling point: 83 ° C.)
- DEGDMA Diethylene glycol dimethyl ether (boiling point: 162 ° C.)
- TrPG tripropylene glycol (boiling point: 232 ° C.)
- DPGMMA Dipropylene glycol monomethyl ether (boiling point: 190 ° C.).
- Example 1 In a glass beaker, 178.0 g of C6FMA, 12.8 g of VA, 12.8 g of IB-X, 1.28 g of HEA, 5.11 g of E-430, 2.55 g of SFN-465, P-204 2.55 g of Aq-18, 2.55 g of Aq-18, 1.28 g of Aq-C, 1.28 g of StSH, 76.6 g of DPG, 404.7 g of water, and heated at 65 ° C. for 40 minutes A preliminary emulsion was obtained using a mixer (manufactured by SMT, HIGH-FLEX DISPERSER HG-92).
- the obtained pre-emulsified liquid was processed at an emulsification pressure of 40 MPa and 1 pass using a high-pressure emulsifier (LAB 60 manufactured by Gorin Co., Ltd.) while maintaining 60 ° C. to obtain an emulsified liquid.
- the obtained emulsion was put into a stainless steel reaction vessel, cooled to 20 ° C. or lower, and 48.5 g of VdCl and 1.28 g of VA-061A were added.
- the gas phase was replaced with nitrogen, and a polymerization reaction was carried out at 60 ° C. for 12 hours to obtain a copolymer (A-1) emulsion.
- Table 2 shows the proportion of structural units based on each monomer.
- Examples 2 to 17 A copolymer emulsion was obtained in the same manner as in Example 1 except that the charged amount of each monomer was changed to the proportion of the structural units based on each monomer shown in Table 2.
- Table 2 shows the proportion of structural units based on each monomer.
- Example 18 The emulsion of copolymer (A-1) was diluted with water, and DMBuPA was added to obtain a water / oil repellent composition having a solid content concentration of 0.6% by mass and a DMBuPA concentration of 2.0%. After immersing ultra-fine polyester wooly and high-density nylon taffeta in the water / oil repellent composition, the wet pick-up was squeezed to 63 mass% and 57 mass%, respectively. These were air-dried at a temperature of 23 ° C. and a humidity of 50% for 24 hours to obtain a test cloth. The test fabric was evaluated. The results are shown in Table 3.
- Example 19 A test cloth was obtained in the same manner as in Example 18 except that the copolymer (A-1) was changed to that shown in Table 3. The evaluation results of the test cloth are shown in Table 3.
- Example 25 The emulsion of copolymer (A-1) was diluted with water to obtain a water / oil repellent composition having a solid content concentration of 0.6% by mass.
- a test cloth was obtained in the same manner as in Example 18 except that the water / oil repellent composition was used. The evaluation results of the test cloth are shown in Table 3.
- Examples 26 to 31, 36 to 39, 44 to 47, 50, 51 A test cloth was obtained in the same manner as in Example 25 except that the copolymer (A-1) was changed to that shown in Table 3. The evaluation results of the test cloth are shown in Table 3.
- Example 52 and 53 A test cloth was obtained in the same manner as in Example 18 except that the DMBuPA concentration was changed to the concentration shown in Table 4. Table 4 shows the evaluation results of the test cloth.
- Example 54 to 56 A test cloth was obtained in the same manner as in Example 23 except that the DMBuPA concentration was changed to the concentration shown in Table 4. Table 4 shows the evaluation results of the test cloth.
- Example 57 to 59 A test cloth was obtained in the same manner as in Example 18 except that DMBuPA was changed to the film-forming aid shown in Table 5. The evaluation results of the test cloth are shown in Table 5.
- Example 60 to 62 A test cloth was obtained in the same manner as in Example 23 except that DMBuPA was changed to the film-forming aid shown in Table 5. The evaluation results of the test cloth are shown in Table 5.
- Example 63 to 65 A test cloth was obtained in the same manner as in Example 18 except that DMBuPA was changed to the film-forming aid shown in Table 6. The evaluation results of the test cloth are shown in Table 6.
- the water / oil repellent composition of the present invention comprises fiber products (clothing articles (sportswear, coats, blousons, work clothes, uniforms, etc.), bags, industrial materials, etc.), non-woven fabrics, leather products, stone materials, concrete building materials. It is useful for applications that impart water and oil repellency to the like. Moreover, it is useful as a repair agent for products with deteriorated wear performance. Moreover, it is useful as a coating agent for filter materials and a surface protective agent. Furthermore, it is also useful for applications that impart water and oil repellency by mixing and molding with fiber such as polypropylene and nylon. It should be noted that all of the description, claims and abstract of Japanese Patent Application No. 2011-099960 filed on April 27, 2011 and Japanese Patent Application No. 2011-269960 filed on December 9, 2011 The contents of which are hereby incorporated herein by reference.
Landscapes
- Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Organic Chemistry (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
- Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Textile Engineering (AREA)
- Materials Engineering (AREA)
- Life Sciences & Earth Sciences (AREA)
- Wood Science & Technology (AREA)
- Combustion & Propulsion (AREA)
- Addition Polymer Or Copolymer, Post-Treatments, Or Chemical Modifications (AREA)
- Materials Applied To Surfaces To Minimize Adherence Of Mist Or Water (AREA)
- Treatments For Attaching Organic Compounds To Fibrous Goods (AREA)
- Compositions Of Macromolecular Compounds (AREA)
- Polymerisation Methods In General (AREA)
Abstract
Description
(I)Rf基を有する単量体に基づく構成単位と(II)ウレタンまたはウレア結合を有する単量体に基づく構成単位と(III)単独重合体のガラス転移温度が50℃以下である単量体に基づく構成単位と(IV)親水基を有する単量体に基づく構成単位と(V)塩素原子を有する単量体に基づく構成単位とする共重合体(A)と、造膜助剤(B)とが水性媒体に分散した撥水撥油剤組成物(特許文献1)。
単量体(a):下式(1)で表される化合物。
(Z-Y)nX ・・・(1)。
ただし、Zは、炭素数が1~6のポリフルオロアルキル基、または下式(2)で表される基であり、Yは、フッ素原子を有しない2価有機基または単結合であり、nは、1または2であり、Xは、nが1の場合は、下式(3-1)~(3-5)で表される基のいずれかであり、nが2の場合は、下式(4-1)~(4-4)で表される基のいずれかである。
CiF2i+1O(CFX1CF2O)jCFX2- ・・・(2)。
ただし、iは、1~6の整数であり、jは、0~10の整数であり、X1およびX2は、それぞれフッ素原子またはトリフルオロメチル基である。
-CR=CH2 ・・・(3-1)、
-C(O)OCR=CH2 ・・・(3-2)、
-OC(O)CR=CH2 ・・・(3-3)、
-OCH2-φ-CR=CH2 ・・・(3-4)、
-OCH=CH2 ・・・(3-5)。
ただし、Rは、水素原子、メチル基またはハロゲン原子であり、φはフェニレン基である。
-CH[-(CH2)mCR=CH2]- ・・・(4-1)、
-CH[-(CH2)mC(O)OCR=CH2]- ・・・(4-2)、
-CH[-(CH2)mOC(O)CR=CH2]- ・・・(4-3)、
-OC(O)CH=CHC(O)O- ・・・(4-4)。
ただし、Rは、水素原子、メチル基またはハロゲン原子であり、mは0~4の整数である。
単量体(b):塩化ビニリデン。
単量体(c):ポリフルオロアルキル基および架橋しうる官能基を有さず、単独重合体のガラス転移温度が20℃以上である単量体。
単量体(d):ポリフルオロアルキル基を有さず、架橋しうる官能基を有する単量体。
造膜助剤(x):下式(5)で表される化合物。
R1O(CH2)r(CHR4)sC(O)NR2R3 ・・・(5)。
ただし、rは0~3の整数であり、sは0または1であり、R1は炭素数が1~4のアルキル基であり、R2およびR3はそれぞれ独立に炭素数が1~3のアルキル基(ただし、エーテル性の酸素原子を含んでいてもよい。)であり、R4は水素原子またはメチル基である。
造膜助剤(y):下式(6)で表される化合物。
R5-O-(CH2CH2O)t-R6 ・・・(6)。
ただし、tは3または4であり、R5およびR6はそれぞれ独立に炭素数が1~4のアルキル基である。
前記単量体(a)としては、式(1)においてZが炭素数4~6のペルフルオロアルキル基であり、Yが炭素数1~4のアルキレン基であり、nが1であり、Xが基(3-3)[ただし、Rは水素原子またはメチル基]である単量体が好ましい。
前記単量体(c)は、シクロアルキル基または炭素数16~25のアルキル基を有する、アクリレートもしくはメタクリレートであることが好ましい。また、前記単量体(d)における架橋しうる官能基は、水酸基、ブロックドイソシアネート基、アミノ基またはエポキシ基であることが好ましい。
前記造膜助剤(x)および前記造膜助剤(y)の合計の割合は、前記水性媒体(B)(100質量%)のうち、0.1~20質量%であることが好ましい。
前記造膜助剤(x)および前記造膜助剤(y)の合計量は、前記共重合体(A)の100質量部に対して、10~4000質量部であることが好ましい。
本発明の撥水撥油剤組成物は、水性媒体中で前記単量体を乳化重合して得られるエマルションに前記造膜助剤(x)および前記造膜助剤(y)から選ばれる少なくとも1種を配合して得られるエマルションであることが好ましい。
本発明の撥水撥油剤組成物の製造方法は、また、界面活性剤および重合開始剤の存在下、造膜助剤(x)および造膜助剤(y)から選ばれる少なくとも1種の造膜助剤を含む水性媒体中で、前記単量体を重合して共重合体(A)のエマルションを製造することを特徴とする。
本発明の物品は、本発明の撥水撥油剤組成物によって表面が処理された、前記共重合体(A)の被膜を表面に有する物品である。
本発明の物品は、充分な風乾後撥水撥油性を有し、かつ環境負荷が低い。
また、本明細書においては、式(2)で表される基を基(2)と記す。他の式で表される基も同様に記す。
また、本明細書における(メタ)アクリレートは、アクリレートまたはメタクリレートを意味する。
また、本明細書における単量体は、重合性不飽和基を有する化合物を意味する。
また、本明細書におけるRf基は、アルキル基の水素原子の一部またはすべてがフッ素原子に置換された基である。
また、本明細書におけるRF基は、アルキル基の水素原子のすべてがフッ素原子に置換された基である。
本発明の撥水撥油剤組成物は、特定の共重合体(A)と、特定の水性媒体(B)とを必須成分として含み、必要に応じて、界面活性剤、添加剤を含む。本発明の撥水撥油剤組成物は、共重合体(A)の微粒子が水性媒体(B)中に安定して分散しているエマルションであることが好ましく、この安定的分散のために通常界面活性剤を含む。
共重合体(A)は、単量体(a)に基づく構成単位と、単量体(b)に基づく構成単位と、単量体(c)に基づく構成単位および単量体(d)に基づく構成単位のいずれかまたは両方とを有する。
共重合体(A)は、風乾後撥水撥油性および動的撥水性(豪雨撥水性)の点から、単量体(a)に基づく構成単位と、単量体(b)に基づく構成単位と、単量体(c)に基づく構成単位と、単量体(d)に基づく構成単位とを有することが好ましい。
共重合体(A)は、単量体(a)~単量体(d)以外の単量体(e)に基づく構成単位を有していてもよい。
単量体(a)は、化合物(1)である。
(Z-Y)nX ・・・(1)。
単量体(a)に基づく構成単位を有することにより、物品に撥水撥油性を付与できる。
CiF2i+1O(CFX1CF2O)jCFX2- ・・・(2)。
ただし、iは、1~6の整数であり、jは、0~10の整数であり、X1およびX2は、それぞれ独立にフッ素原子またはトリフルオロメチル基である。
Zとしては、炭素数4~6のRf基が好ましく、炭素数6のRf基がより好ましい。
Rf基としては、RF基が好ましい。Rf基は、直鎖状であってもよく、分岐状であってもよく、直鎖状が好ましい。Rf基の炭素数は4~6が好ましい。
F(CF2)4-、
F(CF2)5-、
F(CF2)6-、
(CF3)2CF(CF2)2-、
CkF2k+1O[CF(CF3)CF2O]h-CF(CF3)-等。
ただし、kは、1~6の整数であり、hは0~10の整数である。
2価有機基としては、アルキレン基が好ましい。アルキレン基は、直鎖状であってもよく、分岐状であってもよい。アルキレン基の炭素数は2~6が好ましい。2価有機基は、-O-、-NH-、-CO-、-S-、-SO2-、-CD1=CD2-(ただし、D1、D2は、それぞれ独立に水素原子またはメチル基である。)等を有していてもよい。
-CH2-、
-CH2CH2-
-(CH2)3-、
-CH2CH2CH(CH3)-、
-CH=CH-CH2-、
-S-CH2CH2-、
-CH2CH2-S-CH2CH2-、
-CH2CH2-SO2-CH2CH2-、
-W-OC(O)NH-A-NHC(O)O-(CpH2p)-等。
Aは、分岐のない対照的なアルキレン基、アリーレン基またはアラルキレン基であり、-C6H12-、-φ-CH2-φ-、-φ-(ただし、φはフェニレン基である。)が好ましい。
Wは、下記の基のいずれかである。
-SO2N(R7)-CdH2d-、
-CONHCdH2d-、
-CH(RF1)-CeH2e-、
-CqH2q-。
ただし、R7は、水素原子または炭素数1~4のアルキル基であり、dは、2~8の整数であり、RF1は、炭素数1~6のペルフルオロアルキル基であり、eは、0~6の整数であり、qは、1~20の整数である。RF1としては、炭素数4または6のペルフルオロアルキル基が好ましい。
Xは、nが1の場合は、基(3-1)~基(3-5)のいずれかであり、nが2の場合は、基(4-1)~基(4-4)のいずれかである。
-C(O)OCR=CH2 ・・・(3-2)、
-OC(O)CR=CH2 ・・・(3-3)、
-OCH2-φ-CR=CH2 ・・・(3-4)、
-OCH=CH2 ・・・(3-5)。
ただし、Rは、水素原子、メチル基またはハロゲン原子であり、φはフェニレン基である。
-CH[-(CH2)mC(O)OCR=CH2]- ・・・(4-2)、
-CH[-(CH2)mOC(O)CR=CH2]- ・・・(4-3)、
-OC(O)CH=CHC(O)O- ・・・(4-4)。
ただし、Rは、水素原子、メチル基またはハロゲン原子であり、mは0~4の整数である。
化合物(1)としては、Zが炭素数4~6のRF基であり、Yが炭素数1~4のアルキレン基であり、nが1であり、Xが基(3-3)[ただし、Rは水素原子またはメチル基]である化合物が特に好ましい。
単量体(b)は、塩化ビニリデンである。
単量体(b)に基づく構成単位を有することにより、物品に対する共重合体(A)の親和性、造膜性が向上するため、撥水撥油剤組成物に浸漬した後の物品を風乾した場合であっても、物品(繊維製品等)における繊維間のように共重合体(A)による被膜が形成されにくい部分にも共重合体(A)が隅々まで浸透し、共重合体(A)による均一で理想的な被膜を形成できる。その結果、物品に充分な風乾後撥水撥油性を付与できる。
単量体(c)は、Rf基および後述する架橋しうる官能基を有さず、単独重合体のガラス転移温度(以下、Tgと記す。)が20℃以上である単量体である。
単量体(c)に基づく構成単位を有することにより、風乾後撥水撥油性を向上させるとともに物品に動的撥水性(豪雨撥水性)を付与できる。
単量体(c)の1種である塩化ビニルは、単量体(b)の塩化ビニリデンと同様に塩素化オレフィンの1種であり、塩化ビニルに基づく構成単位は共重合体(A)に単量体(b)に基づく構成単位と同様の特性を与える。従って、共重合体(A)が単量体(b)に基づく構成単位を有することより、さらに塩化ビニルに基づく構成単位を有する必要性は少ない。したがって、共重合体(A)が塩化ビニルに基づく構成単位を有する場合であっても、共重合体(A)における塩化ビニルに基づく構成単位の含有量は、塩化ビニルに基づく構成単位と単量体(b)に基づく構成単位との合計量に対して40質量%以下であることが好ましい。より好ましい共重合体(A)は塩化ビニルに基づく構成単位を実質的に有しない共重合体である。
単量体(c)の単独重合体のTgは、JIS K 7121:1987に準じ、示差走査熱量測定(DSC)法で測定した中間点ガラス転移温度とする。
メチルメタクリレート(Tg:105℃)、エチルメタアクリレート(Tg:65℃)、n-プロピルメタアクリレート(Tg:33℃)、イソプロピルメタアクリレート(Tg:81℃)、n-ブチルメタアクリレート(Tg:20℃)、イソブチルメタアクリレート(Tg:48℃)、t-ブチルアクリレート(Tg:41℃)、t-ブチルメタクリレート(Tg:107℃)、セチルメタアクリレート(Tg:23.5℃)、セチルアクリート(Tg:34℃)、ステアリルメタアクリレート(Tg:35℃)、ステアリルアクリレート(Tg:47℃)、ベヘニルメタクリレート(Tg:55℃)、ベヘニルアクリレート(Tg:66℃)、イソボルニルアクリレート(Tg:90℃)、イソボルニルメタクリレート(Tg:180℃)、スチレン(Tg:100℃)、塩化ビニル(Tg:82℃)、ベンジルメタアクレリート(Tg:55℃)、シクロヘキシルメタアクリレート(Tg:66℃)、テトラヒドロフリルメタアクリレート(Tg:60℃)、フェノキシエチルメタアクリレート(Tg:36℃)、シクロペンタニルメタアクリレート(Tg:175℃)、シクロペンタニルアクリレート(Tg:120℃)、シクロペンテニルオキシエチルメタアクリレート(Tg:45℃)。
単量体(d)は、Rf基を有さず、架橋しうる官能基を有する単量体である。
単量体(d)に基づく構成単位を有することにより、風乾後撥水撥油性を向上させるとともに物品に動的撥水性(豪雨撥水性)を付与できる。
該官能基としては、水酸基、イソシアネート基、ブロックドイソシアネート基、アルコキシシリル基、アミノ基、N-アルコキシメチルアミド基、シラノール基、アンモニウム基、アミド基、エポキシ基、オキサゾリン基、カルボキシ基、アルケニル基、スルホ基等が好ましく、水酸基、ブロックドイソシアネート基、アミノ基、エポキシ基が特に好ましい。
単量体(d)としては、下記の化合物が挙げられる。
単量体(e)は、単量体(a)、単量体(b)、単量体(c)および単量体(d)以外の単量体である。
単量体(e)としては、公知の化合物が挙げられる。
単量体(b)に基づく構成単位の割合は、風乾後撥水撥油性および動的撥水性(豪雨撥水性)の点から、全ての単量体に基づく構成単位(100質量%)のうち、5~40質量%が好ましく、10~30質量%がより好ましく、10~20質量%が特に好ましい。
単量体(e)に基づく構成単位の割合は、全ての単量体に基づく構成単位(100質量%)のうち、0~50質量%が好ましく、0~30質量%がより好ましく、0~10質量%が特に好ましい。
共重合体(A)の数平均分子量(Mn)は、12000以上が好ましく、15000以上が特に好ましい。共重合体(A)の数平均分子量(Mn)は、300000以下が好ましく、150000以下が特に好ましい。
共重合体(A)の質量平均分子量(Mw)および数平均分子量(Mn)は、ゲルパーミエイションクロマトグラフィ(GPC)によって測定される、ポリスチレン換算の分子量であり、具体的には、下記の方法で測定する。
測定温度:23℃、
注入量:0.2mL、
流出速度:1mL/分、
溶離液:THF。
共重合体(A)の平均粒子径は、10~1000nmが好ましく、10~300nmがより好ましく、10~250nmが特に好ましい。平均粒子径が該範囲内であれば、界面活性剤等を多量に用いる必要がなく、撥水性が良好であり、染色された布帛類を処理した場合に色落ちが発生せず、水性媒体中で分散粒子が安定に存在できて沈降することがない。
共重合体(A)の平均粒子径は、動的光散乱装置、電子顕微鏡等によって測定される。
水性媒体は、水と、造膜助剤(x)および造膜助剤(y)から選ばれる少なくとも1種の造膜助剤と、必要に応じて他の媒体とを含む。
造膜助剤(x)および造膜助剤(y)は、共重合体(A)との相溶性が良好であることから共重合体(A)を効果的に膨潤または溶解させることができる。さらに、造膜助剤(x)および造膜助剤(y)は、沸点が比較的高いため、風乾時に水よりも蒸発しにくいことから、物品(繊維製品等)における繊維間のように共重合体(A)による被膜が形成されにくい部分にも造膜助剤(x)または造膜助剤(y)によって共重合体(A)が浸透し、共重合体(A)による均一な被膜を形成できる。その結果、物品に充分な風乾後撥水撥油性を付与できる。
造膜助剤(x):
造膜助剤(x)は、化合物(5)である。
R1O(CH2)r(CHR4)sC(O)NR2R3 ・・・(5)。
ただし、rは0~3の整数であり、sは0または1であり、R1は炭素数が1~4のアルキル基であり、R2およびR3はそれぞれ独立に炭素数が1~3のアルキル基(ただし、エーテル性の酸素原子を含んでいてもよい。)であり、R4は水素原子またはメチル基である。
R1としては、たとえば、メチル基、エチル基、プロピル基、イソプロピル基、n-ブチル基等が挙げられ、n-ブチル基が好ましい。
R2およびR3としては、たとえば、メチル基、エチル基、プロピル基、イソプロピル基、メトキシエチル基等が挙げられ、いずれもメチル基であることが好ましい。
R4は水素原子であることが好ましく、r+sは1または2であることが好ましい。
化合物(5)としては、下記の化合物が挙げられる。
造膜助剤(y)は、化合物(6)である。
R5-O-(CH2CH2O)t-R6 ・・・(6)。
ただし、tは3または4であり、R5およびR6はそれぞれ独立に炭素数が1~4のアルキル基である。
R5、R6は、直鎖状であってもよく、分岐状であってもよく、直鎖状が好ましい。
R5、R6としては、たとえば、メチル基、エチル基、プロピル基、イソプロピル基、n-ブチル基等が挙げられ、メチル基が好ましい。
化合物(6)としては、下記の化合物が挙げられる。
ハロゲン化炭化水素としては、ハイドロクロロフルオロカーボン、ハイドロフルオロカーボン、ハイドロブロモカーボン等が挙げられる。
ハイドロフルオロエーテルとしては、分離型ハイドロフルオロエーテル、非分離型ハイドロフルオロエーテル等が挙げられる。分離型ハイドロフルオロエーテルとは、エーテル性酸素原子を介してRF基またはペルフルオロアルキレン基、および、アルキル基またはアルキレン基が結合している化合物である。非分離型ハイドロフルオロエーテルとは、部分的にフッ素化されたアルキル基またはアルキレン基を含むハイドロフルオロエーテルである。
エステルとしては、酢酸メチル、酢酸エチル、酢酸ブチル、プロピオン酸メチル、乳酸メチル、乳酸エチル、乳酸ペンチル等が挙げられる。
エーテルとしては、ジイソプロピルエーテル、ジオキサン、THF等が挙げられる。
硫黄化合物としては、ジメチルスルホキシド、スルホラン等が挙げられる。
有機酸としては、酢酸、プロピオン酸、りんご酸、乳酸等が挙げられる。
本発明の撥水撥油剤組成物は通常界面活性剤を含有する。この界面活性剤は共重合体(A)の微粒子が水性媒体(B)中で安定的に分散させるために必要とされる。前記単量体を水性媒体中で乳化重合して得られる共重合体(A)のエマルションを製造するために、乳化重合に使用する水性媒体は界面活性剤が配合される。本発明の撥水撥油剤組成物中の界面活性剤の少なくとも一部はこの重合の際に使用された界面活性剤である。また、乳化重合で得られたエマルションにさらに界面活性剤を配合することもできる。
ノニオン性界面活性剤とカチオン性界面活性剤との比(ノニオン性界面活性剤/カチオン性界面活性剤)は、97/3~40/60(質量比)が好ましい。
界面活性剤s1は、ポリオキシアルキレンモノアルキルエーテル、ポリオキシアルキレンモノアルケニルエーテル、ポリオキシアルキレンモノアルカポリエニルエーテル、またはポリオキシアルキレンモノポリフルオロアルキルエーテルである。
界面活性剤s1としては、ポリオキシアルキレンモノアルキルエーテル、ポリオキシアルキレンモノアルケニルエーテル、またはポリオキシアルキレンモノポリフルオロアルキルエーテルが好ましい。界面活性剤s1は、1種を単独で用いてもよく、2種以上を併用してもよい。
R10O[CH2CH(CH3)O]u2-(CH2CH2O)u1H ・・・(s11)。
ただし、R10は、炭素数が8以上のアルキル基または炭素数が8以上のアルケニル基であり、u1は、5~50の整数であり、u2は、0~20の整数である。R10は、水素原子の一部がフッ素原子に置換されていてもよい。
u2が20以下であれば、水に可溶となり、水系媒体中に均一に溶解するため、撥水撥油剤組成物の物品への浸透性が良好となる。
R10としては、直鎖状または分岐状のものが好ましい。
u1は、10~30の整数が好ましい。
u2は、0~10の整数が好ましい。
C18H37O[CH2CH(CH3)O]2-(CH2CH2O)30H、
C17H35O-(CH2CH2O)30H、
C16H33O[CH2CH(CH3)O]5-(CH2CH2O)20H、
C12H25O[CH2CH(CH3)O]2-(CH2CH2O)15H、
(C8H17)(C6H13)CHO-(CH2CH2O)15H、
C10H21O[CH2CH(CH3)O]2-(CH2CH2O)15H、
C6F13CH2CH2O-(CH2CH2O)15H、
C6F13CH2CH2O[CH2CH(CH3)O]2-(CH2CH2O)15H、
C4F9CH2CH2O[CH2CH(CH3)O]2-(CH2CH2O)15H。
界面活性剤s2は、分子中に1個以上の炭素-炭素三重結合および1個以上の水酸基を有する化合物からなるノニオン性界面活性剤である。
界面活性剤s2としては、分子中に1個の炭素-炭素三重結合、および1個または2個の水酸基を有する化合物からなるノニオン性界面活性剤が好ましい。
界面活性剤s2は、分子中にPOA鎖を有してもよい。POA鎖としては、POE鎖、POP鎖、POE鎖とPOP鎖とがランダム状に連結された鎖、またはPOE鎖とPOP鎖とがブロック状に連結された鎖が挙げられる。
HO-C(R11)(R12)-C≡C-(CR13)(R14)-OH ・・・(s21)、
HO-(A1O)V-(CR11)(R12)-C≡C-C(R13)(R14)-(OA2)W-OH ・・・(s22)、
HO-C(R15)(R16)-C≡C-H ・・・(s23)、
HO-(A3O)X-C(R15)(R16)-C≡C-H ・・・(s24)。
vおよびwは、それぞれ0以上の整数であり、(v+w)は、1以上の整数である。
xは、1以上の整数である。
v、w、xが、それぞれ2以上である場合、A1~A3は、それぞれ同一であってもよく、異なっていてもよい。
POA鎖としては、POE鎖、POP鎖、またはPOE鎖とPOP鎖とを含む鎖が好ましい。POA鎖の繰り返し単位の数は、1~50が好ましい。
アルキル基としては、炭素数が1~12のアルキル基が好ましく、炭素数が1~4のアルキル基がより好ましい。アルキル基としては、メチル基、エチル基、プロピル基、ブチル基、イソブチル基等が挙げられる。
化合物(s25)は、1種を単独で用いてもよく、2種以上を併用してもよい。
化合物(s25)としては、xおよびyが0である化合物、yとzとの和が平均1~4である化合物、またはyとzとの和が平均10~30である化合物が好ましい。
界面活性剤s3は、POE鎖と、炭素数が3以上のオキシアルキレンが2個以上連続して連なったPOA鎖とが連結し、かつ両末端が水酸基である化合物からなるノニオン性界面活性剤である。
該POA鎖としては、ポリオキシブテン(以下、POBと記す。)および/またはPOP鎖が好ましい。
HO(CH2CH2O)g1(C3H6O)g2(CH2CH2O)g3H ・・・(s31)、
HO(CH2CH2O)g1(CH2CH2CH2CH2O)g2(CH2CH2O)g3H ・・・(s32)。
g2は、2~100の整数である。
g3は、0~200の整数である。
g1が0の場合、g3は、2以上の整数である。g3が0の場合、g1は、2以上の整数である。
-C3H6O-は、-CH(CH3)CH2O-であってもよく、-CH2CH(CH3)O-であってもよく、-CH(CH3)CH2O-と-CH2CH(CH3)O-とが混在したものであってもよい。
POA鎖は、ブロック状である。
HO-(CH2CH2O)15-(C3H6O)35-(CH2CH2O)15H、
HO-(CH2CH2O)8-(C3H6O)35-(CH2CH2O)8H、
HO-(CH2CH2O)45-(C3H6O)17-(CH2CH2O)45H、
HO-(CH2CH2O)34-(CH2CH2CH2CH2O)28-(CH2CH2O)34H。
界面活性剤s4は、分子中にアミンオキシド部分を有するノニオン性界面活性剤である。
界面活性剤s4としては、化合物(s41)が好ましい。
(R17)(R18)(R19)N(→O) ・・・(s41)。
本発明においては、アミンオキシド(N→O)を有する界面活性剤をノニオン性界面活性剤として扱う。
化合物(s41)は、1種を単独で用いてもよく、2種以上を併用してもよい。
(R20)(CH3)2N(→O) ・・・(s42)。
[H(CH2)12](CH3)2N(→O)、
[H(CH2)14](CH3)2N(→O)、
[H(CH2)16](CH3)2N(→O)、
[H(CH2)18](CH3)2N(→O)、
[F(CF2)6(CH2)2](CH3)2N(→O)、
[F(CF2)4(CH2)2](CH3)2N(→O)。
界面活性剤s5は、ポリオキシエチレンモノ(置換フェニル)エーテルの縮合物またはポリオキシエチレンモノ(置換フェニル)エーテルからなるノニオン性界面活性剤である。
置換フェニル基としては、1価炭化水素基で置換されたフェニル基が好ましく、アルキル基、アルケニル基またはスチリル基で置換されたフェニル基がより好ましい。
界面活性剤s6は、ポリオールの脂肪酸エステルからなるノニオン性界面活性剤である。
ポリオールとは、グリセリン、ソルビタン、ソルビット、ポリグリセリン、ポリエチレングリコール、ポリオキシエチレングリセリルエーテル、ポリオキシエチレンソルビタンエーテル、ポリオキシエチレンソルビットエーテルを表わす。
界面活性剤がカチオン性界面活性剤を含む場合、該カチオン性界面活性剤としては、界面活性剤s7が好ましい。
界面活性剤s7は、置換アンモニウム塩形のカチオン性界面活性剤である。
[(R21)4N+]・X- ・・・(s71)。
R21としては、炭素数が6~22の長鎖アルキル基、炭素数が6~22の長鎖アルケニル基、または炭素数が1~9のフルオロアルキル基が好ましい。
R21が長鎖アルキル基以外のアルキル基の場合、R21としては、メチル基またはエチル基が好ましい。
R21が、末端が水酸基であるPOA鎖の場合、POA鎖としては、POE鎖が好ましい。
X-としては、塩素イオン、エチル硫酸イオン、または酢酸イオンが好ましい。
界面活性剤が両性界面活性剤を含む場合、該両性界面活性剤としては、界面活性剤s8が好ましい。
界面活性剤s8は、アラニン類、イミダゾリニウムベタイン類、アミドベタイン類または酢酸ベタインである。
界面活性剤s8としては、ドデシルベタイン、ステアリルベタイン、ドデシルカルボキシメチルヒドロキシエチルイミダゾリニウムベタイン、ドデシルジメチルアミノ酢酸ベタイン、脂肪酸アミドプロピルジメチルアミノ酢酸ベタイン等が挙げられる。
具体例としては、ラウリルジメチルアミノ酢酸ベタイン水溶液(日光ケミカルズ製 NIKKOL AM-301)、ヤシ油脂肪酸アミドプロピルジメチルアミノ酢酸ベタイン水溶液(日光ケミカルズ製、NIKKOL AM-3130N),N-ヤシ油脂肪酸アシル-N-カルボキシメチル-N-ヒドロキシエチルエチレンジアミンナトリウム水溶液(日光ケミカルズ NIKKOL AM-101)等が挙げられる。
界面活性剤s9は、親水性単量体と炭化水素系疎水性単量体および/またはフッ素系疎水性単量体との、ブロック共重合体、ランダム共重合体、または親水性共重合体の疎水性変性物からなる高分子界面活性剤である。
界面活性剤s91:親油性単量体とフッ素系単量体とのブロック共重合体またはランダム共重合体(そのポリフルオロアルキル変性体)からなる高分子界面活性剤。
界面活性剤s91の市販品としては、セイミケミカル社のサーフロン(商品番号:S-383、SC-100シリーズ)が挙げられる。
界面活性剤s1:化合物(s11)のうち、s+rが2~10(好ましくは3~8)の整数であり、rが2~10(好ましくは3~8)の整数である化合物が好ましい。
界面活性剤s2:化合物(s25)のうち、x+yが4以下である化合物が好ましい。
界面活性剤s6:ソルビタン脂肪酸エステルのうち、脂肪酸の炭素数が5~30である化合物が好ましい。具体例としては、ポリオキシエチレン2モル付加物オレイルエーテル(日光ケミカルズ社製、NIKKOL BO-2V、HLB=7.5)、ポリオキシエチレン7モル付加物オレイルエーテル(日光ケミカルズ社製、NIKKOL BO-7V、HLB=10.5)、ポリオキシエチレン5モル付加物ベヘニルエーテル(日光ケミカルズ社製、NIKKOL BB-5、HLB=10.5)、ポリオキシエチレン4モル付加物C12-15アルキルエーテル(日光ケミカルズ社製、NIKKOL BD-4、HLB=10.5)、ポリオキシエチレン1モルポリオキシプロピレン4モル付加物セチルエーテル(日光ケミカルズ社製、NIKKOL PBC-31、HLB=9.5)、2,4,7,9-テトラメチル-5-デシン-4,7-ジオールエチレンオキサイド3.5モル付加物(日信化学工業社製、サーフィノール440、HLB=8)、2,4,7,9-テトラメチル-5-デシン-4,7-ジオールエチレンオキサイド1.3モル付加物(日信化学工業社製、サーフィノール420、HLB=4)、2,4,7,9-テトラメチル-5-デシン-4,7-ジオール(日信化学工業社製、サーフィノール104、HLB=4)、ソルビタンパルミテート(日油社製、ノニオンPP-40R,HLB=6.7)等が挙げられる。
添加剤としては、浸透剤、消泡剤、吸水剤、帯電防止剤、制電性重合体、防皺剤、風合い調整剤、水溶性高分子(ポリアクリルアミド、ポリビニルアルコール等)、熱硬化剤(メラミン樹脂、ウレタン樹脂、トリアジン環含有化合物、イソシアネート系化合物等)、エポキシ硬化剤(イソフタル酸ジヒドラジド、アジピン酸ジヒドラジド、セバチン酸ジヒドラジド、ドデカン二酸ジヒドラジド、1,6-ヘキサメチレンビス(N,N-ジメチルセミカルバジド)、1,1,1’,1’-テトラメチル-4,4’-(メチレン-ジ-パラ-フェニレン)ジセミカルバジド、スピログリコール等)、熱硬化触媒、架橋触媒(有機酸類、塩化アンモニウム等)、合成樹脂、繊維安定剤、無機微粒子等が挙げられる。
本発明の撥水撥油剤組成物は、たとえば、下記の方法(α)または(β)で製造される。
(α)界面活性剤および重合開始剤の存在下、水性媒体中で前記単量体成分を重合して共重合体(A)の分散液またはエマルションを製造した後、これに造膜助剤(x)および造膜助剤(y)から選ばれる少なくとも1種の造膜助剤を配合する方法。造膜助剤の配合と同様に、製造された分散液やエマルションに、必要に応じて、水などの媒体、界面活性剤、添加剤等を配合することができる。
(β)界面活性剤および重合開始剤の存在下、造膜助剤(x)および造膜助剤(y)から選ばれる少なくとも1種の造膜助剤を含む水性媒体中で、前記単量体成分を重合して共重合体(A)の分散液またはエマルションを製造する方法。製造された分散液やエマルションに、必要に応じて、水などの媒体、界面活性剤、添加剤等を配合することができる。また、製造された分散液やエマルションに、さらに造膜助剤(x)および造膜助剤(y)から選ばれる少なくとも1種の造膜助剤を配合することもできる。
なお、これらの方法において、重合により得られた分散液やエマルションに造膜助剤や水などの媒体を配合することを以下希釈ともいう。
重合法としては、界面活性剤および重合開始剤の存在下、水および必要に応じて他の媒体を含む水性媒体中にて、単量体成分を乳化重合して共重合体(A)のエマルションを得る方法が好ましい。
重合開始剤の添加量は、単量体成分の100質量部に対して、0.1~5質量部が好ましく、0.1~3質量部がより好ましい。
分子量調整剤の添加量は、単量体成分の100質量部に対して、0~5質量部が好ましく、0~2質量部がより好ましい。
本発明の撥水撥油剤組成物は、方法(α)で製造されたエマルションであることが好ましい。特に、方法(α)により、比較的多量の造膜助剤を有する、分散安定性が良好な撥水撥油剤組成物を容易に得ることができる。また、方法(α)は方法(β)よりも物品の種類、物品への処理条件に応じて簡便に造膜助剤の割合を調整することができる。さらに、撥水撥油剤組成物における水性媒体の組成の調整も容易である。たとえば、方法(α)では、布等の物品の種類や処理条件(浴温度、風乾温度等)などに応じて造膜助剤の最適量を容易に設定できる。
エマルションまたは撥水撥油剤組成物の固形分濃度は、加熱前のエマルションまたは撥水撥油剤組成物の質量と、120℃の対流式乾燥機にて4時間乾燥した後の質量とから計算される。
以上説明した本発明の撥水撥油剤組成物にあっては、共重合体(A)を含み、かつ水と、造膜助剤(x)および造膜助剤(y)から選ばれる少なくとも1種の造膜助剤を含む水性媒体(B)とを必須成分とするため、物品の表面に充分な風乾後撥水撥油性を付与できる。
本発明の物品は、本発明の撥水撥油剤組成物によって表面が処理されたものであり、共重合体(A)の被膜を表面に有する物品である。
処理される物品としては、繊維(天然繊維、合成繊維、混紡繊維等)、各種繊維製品、不織布、樹脂、紙、皮革、金属、石、コンクリート、石膏、ガラス等が挙げられる。
処理方法としては、たとえば、公知の塗工方法によって物品に撥水撥油剤組成物を含む塗布液を塗布した後、乾燥する方法、または物品を、撥水撥油剤組成物を含む塗布液に浸漬した後、乾燥する方法が挙げられる。
防水加工としては、防水膜を付与する加工が挙げられる。防水膜としては、ウレタン樹脂やアクリル樹脂から得られる多孔質膜、ウレタン樹脂やアクリル樹脂から得られる無孔質膜、ポリテトラフルオロエチレン膜、またはこれらを組み合わせた透湿防水膜が挙げられる。
例1~17は製造例であり、例18~20、33~35、40~43、52、53、57は実施例であり、例21~32、36~39、44~51、54~56、58~65は比較例である。
(撥油性)
試験布について、AATCC-TM118-1966の試験方法にしたがって撥油性を評価した。撥油性は、表1に示す等級で表した。等級に+(-)を記したものは、それぞれの性質がわずかに良い(悪い)ことを示す。
試験布について、JIS L1092-1992のスプレー試験にしたがって撥水性を評価した。撥水性は、1~5の5段階の等級で表した。点数が大きいほど撥水性が良好であることを示す。等級に+(-)を記したものは、当該等級の標準的なものと比べてそれぞれの性質がわずかに良い(悪い)ことを示す。
試験布について、JIS L1092(C)法に記載の方法(ブンデスマン試験)にしたがって、降雨量を80cc/分、降雨水温を20℃、降雨時間10分とする条件で降雨させ、撥水性を評価した。撥水性は、1~5の5段階の等級で表した。点数が大きいほど撥水性が良好であることを示す。等級に+(-)を記したものは、それぞれの性質がわずかに良い(悪い)ことを示す。
(単量体(a))
C6FMA:F(CF2)6CH2CH2OC(O)C(CH3)=CH2。
VdCl:塩化ビニリデン(Tg:-18℃)。
VCl:塩化ビニル(Tg:82℃)、
VA:ベヘニルアクリレート(Tg:66℃)、
StA:ステアリルアクリレート(Tg:47℃)、
StMA:ステアリルメタクリレート(Tg:35℃)、
CetMA:セチルメタクリレート(Tg:23.5℃)、
CyhMA:シクロヘキシルメタクリレート(Tg:66℃)、
n-BuMA:n-ブチルメタクリレート(Tg:20℃)、
IB-X:イソボルニルメタクリレート(Tg:180℃)。
HEA:2-ヒドロキシエチルアクリレート。
CmFA:F(CF2)mCH2CH2OC(O)CH=CH2(mが6~16の混合物であり、mが8以上のものが99質量%以上であり、mの平均値は9である。)、
LaA:ラウリルアクリレート(Tg:15℃)、
EtA:エチルアクリレート(Tg:-22℃)。
E-430:ポリオキシエチレンオレイルエーテル(花王社製、エマルゲンE430)、
SFN-465:2,4,7,9-テトラメチル-5-デシン-4,7-ジオールエチレンオキシド付加物(日信化学工業社製、サーフィノール465)、
P-204:エチレンオキシドプロピレンオキシド重合物(日油社製、プロノン204)、
Aq-18:ステアリルトリメチルアンモニウムクロライド(ライオン社製、アーカード18-63)、
Aq-C:ラウリルトリメチルアンモニウムクロライド(ライオン社製、アーカードC-50)。
StSH:ステアリルメルカプタン。
VA-061A:2,2’-アゾビス[2-(2-イミダゾリン-2-イル)プロパン](和光純薬社製、VA-061)の酢酸塩。
水:脱イオン水。
DMBuPA:3-n-ブトキシ-N,N-ジメチルプロピオンアミド(出光興産社製、エクアミドB100、沸点:252℃)。
TeEGDME:テトラエチレングリコールジメチルエーテル(沸点:275℃)。
DPG:ジプロピレングリコール(沸点:232℃)、
EGDMA:エチレングリコールジメチルエーテル(沸点:83℃)、
DEGDMA:ジエチレングリコールジメチルエーテル(沸点:162℃)、
TrPG:トリプロピレングリコール(沸点:232℃)、
DPGMMA:ジプロピレングリコールモノメチルエーテル(沸点:190℃)。
ガラス製ビーカーに、C6FMAの178.0g、VAの12.8g、IB-Xの12.8g、HEAの1.28g、E-430の5.11g、SFN-465の2.55g、P-204の2.55g、Aq-18の2.55g、Aq-Cの1.28g、StSHの1.28g、DPGの76.6g、水の404.7gを入れ、65℃で40分間加温した後、ミキサー(SMT社製、HIGH-FLEX DISPERSER HG-92)を用いて予備乳化液を得た。
各単量体の仕込み量が、表2に示す各単量体に基づく構成単位の割合となるように変更した以外は、例1と同様にして共重合体のエマルションを得た。各単量体に基づく構成単位の割合を表2に示す。
共重合体(A-1)のエマルションを水で希釈し、DMBuPAを加えて、固形分濃度:0.6質量%、DMBuPA濃度:2.0%の撥水撥油剤組成物を得た。
撥水撥油剤組成物に極細ポリエステル・ウーリーおよび高密度ナイロン・タフタを浸漬した後、ウェットピックアップがそれぞれ63質量%、57質量%となるように絞った。これらを温度:23℃、湿度:50%にて24時間風乾したものを試験布とした。該試験布を評価した。結果を表3に示す。
共重合体(A-1)を表3に示すものに変更した以外は、例18と同様にして試験布を得た。試験布の評価結果を表3に示す。
共重合体(A-1)のエマルションを水で希釈し、固形分濃度:0.6質量%の撥水撥油剤組成物を得た。
該撥水撥油剤組成物を用いた以外は、例18と同様にして試験布を得た。試験布の評価結果を表3に示す。
共重合体(A-1)を表3に示すものに変更した以外は、例25と同様にして試験布を得た。試験布の評価結果を表3に示す。
DMBuPA濃度を表4に示す濃度に変更した以外は、例18と同様にして試験布を得た。試験布の評価結果を表4に示す。
DMBuPA濃度を表4に示す濃度に変更した以外は、例23と同様にして試験布を得た。試験布の評価結果を表4に示す。
DMBuPAを表5に示す造膜助剤に変更した以外は、例18と同様にして試験布を得た。試験布の評価結果を表5に示す。
DMBuPAを表5に示す造膜助剤に変更した以外は、例23と同様にして試験布を得た。試験布の評価結果を表5に示す。
DMBuPAを表6に示す造膜助剤に変更した以外は、例18と同様にして試験布を得た。試験布の評価結果を表6に示す。
なお、2011年4月27日に出願された日本特許出願2011-099960号および2011年12月9日に出願された日本特許出願2011-269960号の明細書、特許請求の範囲及び要約書の全内容をここに引用し、本発明の明細書の開示として、取り入れるものである。
Claims (13)
- 共重合体(A)と水性媒体(B)とを含み、
前記共重合体(A)が、下記単量体(a)に基づく構成単位と、下記単量体(b)に基づく構成単位と、下記単量体(c)に基づく構成単位および下記単量体(d)に基づく構成単位から選ばれる少なくとも1種の構成単位と、を有し、
前記水性媒体(B)が、水と、下記造膜助剤(x)および下記造膜助剤(y)から選ばれる少なくとも1種の造膜助剤を含む、撥水撥油剤組成物。
単量体(a):下式(1)で表される化合物。
(Z-Y)nX ・・・(1)。
ただし、Zは、炭素数が1~6のポリフルオロアルキル基、または下式(2)で表される基であり、Yは、フッ素原子を有しない2価有機基または単結合であり、nは、1または2であり、Xは、nが1の場合は、下式(3-1)~(3-5)で表される基のいずれかであり、nが2の場合は、下式(4-1)~(4-4)で表される基のいずれかである。
CiF2i+1O(CFX1CF2O)jCFX2- ・・・(2)。
ただし、iは、1~6の整数であり、jは、0~10の整数であり、X1およびX2は、それぞれフッ素原子またはトリフルオロメチル基である。
-CR=CH2 ・・・(3-1)、
-C(O)OCR=CH2 ・・・(3-2)、
-OC(O)CR=CH2 ・・・(3-3)、
-OCH2-φ-CR=CH2 ・・・(3-4)、
-OCH=CH2 ・・・(3-5)。
ただし、Rは、水素原子、メチル基またはハロゲン原子であり、φはフェニレン基である。
-CH[-(CH2)mCR=CH2]- ・・・(4-1)、
-CH[-(CH2)mC(O)OCR=CH2]- ・・・(4-2)、
-CH[-(CH2)mOC(O)CR=CH2]- ・・・(4-3)、
-OC(O)CH=CHC(O)O- ・・・(4-4)。
ただし、Rは、水素原子、メチル基またはハロゲン原子であり、mは0~4の整数である。
単量体(b):塩化ビニリデン。
単量体(c):ポリフルオロアルキル基および架橋しうる官能基を有さず、単独重合体のガラス転移温度が20℃以上である単量体。
単量体(d):ポリフルオロアルキル基を有さず、架橋しうる官能基を有する単量体。
造膜助剤(x):下式(5)で表される化合物。
R1O(CH2)r(CHR4)sC(O)NR2R3 ・・・(5)。
ただし、rは0~3の整数であり、sは0または1であり、R1は炭素数が1~4のアルキル基であり、R2およびR3はそれぞれ独立に炭素数が1~3のアルキル基(ただし、エーテル性の酸素原子を含んでいてもよい。)であり、R4は水素原子またはメチル基である。
造膜助剤(y):下式(6)で表される化合物。
R5-O-(CH2CH2O)t-R6 ・・・(6)。
ただし、tは3または4であり、R5およびR6はそれぞれ独立に炭素数が1~4のアルキル基である。 - 前記共重合体(A)が、前記単量体(a)に基づく構成単位と、前記単量体(b)に基づく構成単位と、前記単量体(c)に基づく構成単位と、前記単量体(d)に基づく構成単位とを有する、請求項1に記載の撥水撥油剤組成物。
- Zが炭素数4~6のペルフルオロアルキル基であり、Yが炭素数1~4のアルキレン基であり、nが1であり、Xが基(3-3)[ただし、Rは水素原子またはメチル基]である、請求項1または2に記載の撥水撥油剤組成物。
- 単量体(c)が、シクロアルキル基または炭素数16~25のアルキル基を有する、アクリレートもしくはメタクリレートである、請求項1~3のいずれか一項に記載の撥水撥油剤組成物。
- 単量体(d)における架橋しうる官能基が水酸基、ブロックドイソシアネート基、アミノ基またはエポキシ基である、請求項1~4のいずれか一項に記載の撥水撥油剤組成物。
- 前記造膜助剤(x)が、3-アルコキシ-N,N-ジアルキルプロピオンアミドである、請求項1~5のいずれか一項に記載の撥水撥油剤組成物。
- 前記造膜助剤(y)が、トリエチレングリコールジメチルエーテルまたはテトラエチレングリコールジメチルエーテルである、請求項1~6のいずれか一項に記載の撥水撥油剤組成物。
- 前記造膜助剤(x)および前記造膜助剤(y)の合計の割合が、前記水性媒体(B)(100質量%)のうち、0.1~20質量%である、請求項1~7のいずれか一項に記載の撥水撥油剤組成物。
- 前記造膜助剤(x)および前記造膜助剤(y)の合計量が、前記共重合体(A)の100質量部に対して、10~4000質量部である、請求項1~8のいずれか一項に記載の撥水撥油剤組成物。
- 撥水撥油剤組成物が、水性媒体中で前記単量体を乳化重合して得られるエマルションに前記造膜助剤(x)および前記造膜助剤(y)から選ばれる少なくとも1種を配合して得られるエマルションである、請求項1~9のいずれか一項に記載の撥水撥油剤組成物。
- 界面活性剤および重合開始剤の存在下、水性媒体中で前記単量体を重合して共重合体(A)のエマルションを製造した後、得られたエマルションに造膜助剤(x)および造膜助剤(y)から選ばれる少なくとも1種の造膜助剤を配合することを特徴とする請求項1~10のいずれか一項に記載の撥水撥油剤組成物の製造方法。
- 界面活性剤および重合開始剤の存在下、造膜助剤(x)および造膜助剤(y)から選ばれる少なくとも1種の造膜助剤を含む水性媒体中で、前記単量体を重合して共重合体(A)のエマルションを製造することを特徴とする請求項1~10のいずれか一項に記載の撥水撥油剤組成物の製造方法。
- 請求項1~10のいずれか一項に記載の撥水撥油剤組成物によって表面が処理された、前記共重合体(A)の被膜を表面に有する物品。
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2013512313A JP6070545B2 (ja) | 2011-04-27 | 2012-04-19 | 撥水撥油剤組成物、その製造方法および物品 |
| CN201280020087.8A CN103492521B (zh) | 2011-04-27 | 2012-04-19 | 拒水拒油剂组合物、其制造方法及物品 |
| EP12777818.1A EP2703466B1 (en) | 2011-04-27 | 2012-04-19 | Water-repellent and oil-repellent agent compound, manufacturing method therefor, and article |
| KR1020137028064A KR101892775B1 (ko) | 2011-04-27 | 2012-04-19 | 발수발유제 조성물, 그 제조 방법 및 물품 |
| US14/062,579 US9040625B2 (en) | 2011-04-27 | 2013-10-24 | Water/oil repellent composition, method for its production and article |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2011099960 | 2011-04-27 | ||
| JP2011-099960 | 2011-04-27 | ||
| JP2011-269960 | 2011-12-09 | ||
| JP2011269960 | 2011-12-09 |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US14/062,579 Continuation US9040625B2 (en) | 2011-04-27 | 2013-10-24 | Water/oil repellent composition, method for its production and article |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2012147625A1 true WO2012147625A1 (ja) | 2012-11-01 |
Family
ID=47072145
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2012/060625 WO2012147625A1 (ja) | 2011-04-27 | 2012-04-19 | 撥水撥油剤組成物、その製造方法および物品 |
Country Status (6)
| Country | Link |
|---|---|
| US (1) | US9040625B2 (ja) |
| EP (1) | EP2703466B1 (ja) |
| JP (1) | JP6070545B2 (ja) |
| KR (1) | KR101892775B1 (ja) |
| CN (1) | CN103492521B (ja) |
| WO (1) | WO2012147625A1 (ja) |
Cited By (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2014021106A1 (ja) * | 2012-07-31 | 2014-02-06 | ダイキン工業株式会社 | マスク用フィルター |
| WO2014200029A1 (ja) * | 2013-06-14 | 2014-12-18 | 旭硝子株式会社 | 撥水撥油剤組成物、撥水撥油剤組成物の製造方法および物品 |
| US20150166863A1 (en) * | 2012-08-21 | 2015-06-18 | Asahi Glass Company, Limited | Fluorinated copolymer and process for its production, water repellent composition, and article |
| JP2015166465A (ja) * | 2013-11-22 | 2015-09-24 | ダイキン工業株式会社 | 水系の表面処理剤 |
| JP2015172198A (ja) * | 2013-11-22 | 2015-10-01 | ダイキン工業株式会社 | 水系エマルションの表面処理剤 |
| WO2016117592A1 (ja) * | 2015-01-20 | 2016-07-28 | 三菱マテリアル株式会社 | 低屈折率膜形成用液組成物 |
| JP2016135838A (ja) * | 2015-01-20 | 2016-07-28 | 三菱マテリアル株式会社 | 低屈折率膜形成用液組成物 |
| EP3075813A4 (en) * | 2013-11-26 | 2016-10-26 | Asahi Glass Co Ltd | WATER- AND OIL-REPELLING COMPOSITION, ARTICLES AND METHOD FOR PRODUCING THE ARTICLE |
| WO2016208512A1 (ja) * | 2015-06-25 | 2016-12-29 | 旭硝子株式会社 | 撥水剤組成物、撥水剤組成物の製造方法および物品 |
| JP2020172715A (ja) * | 2019-04-09 | 2020-10-22 | Agc株式会社 | 透湿防水布帛の製造方法 |
| JP2020535275A (ja) * | 2017-09-27 | 2020-12-03 | ハンワ・ソリューションズ・コーポレイション | 塩化ビニル系樹脂組成物およびその製造方法 |
Families Citing this family (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR102159595B1 (ko) * | 2016-09-08 | 2020-09-25 | 다이킨 고교 가부시키가이샤 | 퍼플루오로(폴리)에테르 변성 아미드실란 화합물을 포함하는 조성물 |
| EP3604467A4 (en) * | 2017-03-23 | 2021-07-14 | Tokyo Ohka Kogyo Co., Ltd. | SURFACE TREATMENT LIQUID AND SURFACE TREATMENT METHOD |
| KR20210132113A (ko) * | 2019-04-18 | 2021-11-03 | 디아이씨 가부시끼가이샤 | 구조체, 구조체의 제조 방법, 열교환기용 부재 및 열교환기 |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH10273879A (ja) | 1997-01-30 | 1998-10-13 | Daikin Ind Ltd | 新規組成物および処理剤 |
| JP2004250353A (ja) * | 2003-02-19 | 2004-09-09 | Idemitsu Petrochem Co Ltd | β−アルコキシプロピオンアミド類の製造方法 |
| WO2006075373A1 (ja) * | 2005-01-13 | 2006-07-20 | Idemitsu Kosan Co., Ltd. | β−アルコキシプロピオンアミド類、溶剤、洗浄剤および液状薬剤組成物、並びにβ−アルコキシプロピオンアミド類の製造方法 |
| WO2009081822A1 (ja) | 2007-12-26 | 2009-07-02 | Asahi Glass Company, Limited | 撥水撥油剤組成物中の炭素数が20以下である低分子の有機化合物の分析方法 |
| WO2010123042A1 (ja) * | 2009-04-23 | 2010-10-28 | 旭硝子株式会社 | 撥水撥油剤組成物、その製造方法および物品の処理方法 |
| WO2010140668A1 (ja) * | 2009-06-05 | 2010-12-09 | 旭硝子株式会社 | 撥水撥油剤組成物、その製造方法および物品の処理方法 |
Family Cites Families (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR100889714B1 (ko) * | 2001-04-13 | 2009-03-23 | 아사히 가라스 가부시키가이샤 | 발수발유제 조성물 |
| CN101506257B (zh) * | 2006-08-25 | 2012-08-08 | 克莱里安特财务(Bvi)有限公司 | 防油、防水和防污的甲基丙烯酸全氟烷基乙酯共聚物 |
| KR101544627B1 (ko) | 2007-09-28 | 2015-08-17 | 아사히 가라스 가부시키가이샤 | 발수 발유제 조성물 및 물품 |
| JP2010100766A (ja) * | 2008-10-24 | 2010-05-06 | Asahi Glass Co Ltd | 撥水撥油剤組成物およびその製造方法 |
| JP2009185079A (ja) * | 2009-05-27 | 2009-08-20 | Idemitsu Kosan Co Ltd | β−アルコキシプロピオンアミド類の製造方法 |
-
2012
- 2012-04-19 JP JP2013512313A patent/JP6070545B2/ja active Active
- 2012-04-19 KR KR1020137028064A patent/KR101892775B1/ko active IP Right Grant
- 2012-04-19 CN CN201280020087.8A patent/CN103492521B/zh active Active
- 2012-04-19 WO PCT/JP2012/060625 patent/WO2012147625A1/ja active Application Filing
- 2012-04-19 EP EP12777818.1A patent/EP2703466B1/en active Active
-
2013
- 2013-10-24 US US14/062,579 patent/US9040625B2/en active Active
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH10273879A (ja) | 1997-01-30 | 1998-10-13 | Daikin Ind Ltd | 新規組成物および処理剤 |
| JP2004250353A (ja) * | 2003-02-19 | 2004-09-09 | Idemitsu Petrochem Co Ltd | β−アルコキシプロピオンアミド類の製造方法 |
| WO2006075373A1 (ja) * | 2005-01-13 | 2006-07-20 | Idemitsu Kosan Co., Ltd. | β−アルコキシプロピオンアミド類、溶剤、洗浄剤および液状薬剤組成物、並びにβ−アルコキシプロピオンアミド類の製造方法 |
| WO2009081822A1 (ja) | 2007-12-26 | 2009-07-02 | Asahi Glass Company, Limited | 撥水撥油剤組成物中の炭素数が20以下である低分子の有機化合物の分析方法 |
| WO2010123042A1 (ja) * | 2009-04-23 | 2010-10-28 | 旭硝子株式会社 | 撥水撥油剤組成物、その製造方法および物品の処理方法 |
| WO2010140668A1 (ja) * | 2009-06-05 | 2010-12-09 | 旭硝子株式会社 | 撥水撥油剤組成物、その製造方法および物品の処理方法 |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP2703466A4 |
Cited By (25)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2014223623A (ja) * | 2012-07-31 | 2014-12-04 | ダイキン工業株式会社 | マスク用フィルター |
| WO2014021106A1 (ja) * | 2012-07-31 | 2014-02-06 | ダイキン工業株式会社 | マスク用フィルター |
| US9487689B2 (en) * | 2012-08-21 | 2016-11-08 | Asahi Glass Company, Limited | Fluorinated copolymer and process for its production, water repellent composition, and article |
| US20150166863A1 (en) * | 2012-08-21 | 2015-06-18 | Asahi Glass Company, Limited | Fluorinated copolymer and process for its production, water repellent composition, and article |
| EP2889316A4 (en) * | 2012-08-21 | 2016-01-13 | Asahi Glass Co Ltd | FLUORINATED COPOLYMER, PROCESS FOR PRODUCTION THEREOF, WATER-REPELLENT COMPOSITION AND ARTICLE |
| WO2014200029A1 (ja) * | 2013-06-14 | 2014-12-18 | 旭硝子株式会社 | 撥水撥油剤組成物、撥水撥油剤組成物の製造方法および物品 |
| US10081908B2 (en) | 2013-06-14 | 2018-09-25 | AGC Inc. | Water/oil repellent composition, method for producing water/oil repellent composition and article |
| CN108467453A (zh) * | 2013-06-14 | 2018-08-31 | 旭硝子株式会社 | 拒水拒油剂组合物、拒水拒油剂组合物的制造方法及物品 |
| CN105247005A (zh) * | 2013-06-14 | 2016-01-13 | 旭硝子株式会社 | 拒水拒油剂组合物、拒水拒油剂组合物的制造方法及物品 |
| CN108467453B (zh) * | 2013-06-14 | 2020-03-27 | Agc株式会社 | 拒水拒油剂组合物、拒水拒油剂组合物的制造方法及物品 |
| JPWO2014200029A1 (ja) * | 2013-06-14 | 2017-02-23 | 旭硝子株式会社 | 撥水撥油剤組成物、撥水撥油剤組成物の製造方法および物品 |
| JP2015166465A (ja) * | 2013-11-22 | 2015-09-24 | ダイキン工業株式会社 | 水系の表面処理剤 |
| JP2015172198A (ja) * | 2013-11-22 | 2015-10-01 | ダイキン工業株式会社 | 水系エマルションの表面処理剤 |
| EP3075813A4 (en) * | 2013-11-26 | 2016-10-26 | Asahi Glass Co Ltd | WATER- AND OIL-REPELLING COMPOSITION, ARTICLES AND METHOD FOR PRODUCING THE ARTICLE |
| JPWO2015080060A1 (ja) * | 2013-11-26 | 2017-03-16 | 旭硝子株式会社 | 撥水撥油剤組成物、物品、および物品の製造方法 |
| JP2016135838A (ja) * | 2015-01-20 | 2016-07-28 | 三菱マテリアル株式会社 | 低屈折率膜形成用液組成物 |
| WO2016117592A1 (ja) * | 2015-01-20 | 2016-07-28 | 三菱マテリアル株式会社 | 低屈折率膜形成用液組成物 |
| TWI662081B (zh) * | 2015-01-20 | 2019-06-11 | 日商三菱綜合材料股份有限公司 | 低折射率膜形成用液體組成物 |
| WO2016208512A1 (ja) * | 2015-06-25 | 2016-12-29 | 旭硝子株式会社 | 撥水剤組成物、撥水剤組成物の製造方法および物品 |
| EP3315578A4 (en) * | 2015-06-25 | 2019-01-23 | AGC Inc. | WATER-REPELLING COMPOSITION, METHOD FOR PRODUCING A WATER-REPELLING COMPOSITION AND ARTICLES |
| JPWO2016208512A1 (ja) * | 2015-06-25 | 2018-05-24 | 旭硝子株式会社 | 撥水剤組成物、撥水剤組成物の製造方法および物品 |
| JP2020535275A (ja) * | 2017-09-27 | 2020-12-03 | ハンワ・ソリューションズ・コーポレイション | 塩化ビニル系樹脂組成物およびその製造方法 |
| JP7177149B2 (ja) | 2017-09-27 | 2022-11-22 | ハンワ ソリューションズ コーポレイション | 塩化ビニル系樹脂組成物およびその製造方法 |
| JP2020172715A (ja) * | 2019-04-09 | 2020-10-22 | Agc株式会社 | 透湿防水布帛の製造方法 |
| JP7279480B2 (ja) | 2019-04-09 | 2023-05-23 | Agc株式会社 | 透湿防水布帛の製造方法 |
Also Published As
| Publication number | Publication date |
|---|---|
| EP2703466A4 (en) | 2014-11-19 |
| JPWO2012147625A1 (ja) | 2014-07-28 |
| CN103492521A (zh) | 2014-01-01 |
| EP2703466A1 (en) | 2014-03-05 |
| EP2703466B1 (en) | 2018-07-11 |
| KR20140022392A (ko) | 2014-02-24 |
| CN103492521B (zh) | 2015-08-19 |
| KR101892775B1 (ko) | 2018-08-28 |
| JP6070545B2 (ja) | 2017-02-01 |
| US9040625B2 (en) | 2015-05-26 |
| US20140051797A1 (en) | 2014-02-20 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6070545B2 (ja) | 撥水撥油剤組成物、その製造方法および物品 | |
| JP5353701B2 (ja) | 撥水撥油剤組成物および物品 | |
| JP5585078B2 (ja) | 撥水撥油剤組成物、その製造方法および物品 | |
| EP2423292B1 (en) | Water and oil repellent composition, method for producing same, and method for treating article | |
| JP5572385B2 (ja) | 撥水撥油剤組成物、その製造方法および物品 | |
| EP2166026B1 (en) | Method for production of water-repellant/oil-repellant composition, and article | |
| JP6070546B2 (ja) | 撥水撥油剤組成物、その製造方法および物品 | |
| EP2439249B1 (en) | Water-repellent oil-repellent agent composition, process for production thereof, and method for treatment of article | |
| WO2012147573A1 (ja) | 撥水撥油剤組成物、その製造方法および物品 | |
| WO2010047258A1 (ja) | 撥水撥油剤組成物およびその製造方法 | |
| WO2009148098A1 (ja) | 共重合体、その製造方法および撥水撥油剤組成物 | |
| EP2172530B1 (en) | Method for production of water-repellent and oil-repellant composition, and article | |
| WO2011078135A1 (ja) | 撥水撥油剤組成物、その製造方法および物品の処理方法 | |
| JPWO2009041650A1 (ja) | 撥水撥油剤組成物および物品 | |
| JP2010100766A (ja) | 撥水撥油剤組成物およびその製造方法 | |
| JP2011021081A (ja) | 撥水撥油剤組成物およびその製造方法 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 12777818 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2013512313 Country of ref document: JP Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2012777818 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 20137028064 Country of ref document: KR Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |