WO2010058762A1 - 鋼板および表面処理鋼板ならびにそれらの製造方法 - Google Patents
鋼板および表面処理鋼板ならびにそれらの製造方法 Download PDFInfo
- Publication number
- WO2010058762A1 WO2010058762A1 PCT/JP2009/069464 JP2009069464W WO2010058762A1 WO 2010058762 A1 WO2010058762 A1 WO 2010058762A1 JP 2009069464 W JP2009069464 W JP 2009069464W WO 2010058762 A1 WO2010058762 A1 WO 2010058762A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- steel sheet
- rolling
- steel
- cold
- less
- Prior art date
Links
- 229910000831 Steel Inorganic materials 0.000 title claims abstract description 163
- 239000010959 steel Substances 0.000 title claims abstract description 163
- 238000004519 manufacturing process Methods 0.000 title claims description 18
- 238000005204 segregation Methods 0.000 claims abstract description 28
- 239000000203 mixture Substances 0.000 claims abstract description 17
- 239000000126 substance Substances 0.000 claims abstract description 17
- 229910052758 niobium Inorganic materials 0.000 claims abstract description 14
- 229910052720 vanadium Inorganic materials 0.000 claims abstract description 14
- 229910052804 chromium Inorganic materials 0.000 claims abstract description 13
- 229910052802 copper Inorganic materials 0.000 claims abstract description 13
- 229910052750 molybdenum Inorganic materials 0.000 claims abstract description 13
- 229910052759 nickel Inorganic materials 0.000 claims abstract description 12
- 229910052726 zirconium Inorganic materials 0.000 claims abstract description 8
- 229910052748 manganese Inorganic materials 0.000 claims abstract description 6
- 229910052797 bismuth Inorganic materials 0.000 claims abstract description 5
- 229910052796 boron Inorganic materials 0.000 claims abstract description 5
- 229910052757 nitrogen Inorganic materials 0.000 claims abstract description 5
- 229910052698 phosphorus Inorganic materials 0.000 claims abstract description 5
- 229910052710 silicon Inorganic materials 0.000 claims abstract description 5
- 229910052717 sulfur Inorganic materials 0.000 claims abstract description 5
- 238000005096 rolling process Methods 0.000 claims description 35
- 238000007747 plating Methods 0.000 claims description 27
- 238000000137 annealing Methods 0.000 claims description 25
- 239000010960 cold rolled steel Substances 0.000 claims description 25
- 238000000034 method Methods 0.000 claims description 22
- 238000005098 hot rolling Methods 0.000 claims description 19
- 238000005097 cold rolling Methods 0.000 claims description 14
- 229910052719 titanium Inorganic materials 0.000 claims description 13
- 238000009749 continuous casting Methods 0.000 claims description 11
- 238000007711 solidification Methods 0.000 claims description 10
- 230000008023 solidification Effects 0.000 claims description 10
- 238000001953 recrystallisation Methods 0.000 claims description 4
- 229910052782 aluminium Inorganic materials 0.000 claims description 3
- 229910052799 carbon Inorganic materials 0.000 claims description 3
- 238000005452 bending Methods 0.000 abstract description 47
- 238000012360 testing method Methods 0.000 description 25
- 239000010410 layer Substances 0.000 description 19
- 238000001816 cooling Methods 0.000 description 9
- 238000009826 distribution Methods 0.000 description 9
- 238000000265 homogenisation Methods 0.000 description 9
- 239000000463 material Substances 0.000 description 9
- 229910000734 martensite Inorganic materials 0.000 description 7
- 229910001335 Galvanized steel Inorganic materials 0.000 description 6
- 238000005275 alloying Methods 0.000 description 6
- 230000000694 effects Effects 0.000 description 6
- 239000008397 galvanized steel Substances 0.000 description 6
- 229910001563 bainite Inorganic materials 0.000 description 5
- 238000010438 heat treatment Methods 0.000 description 5
- 238000005554 pickling Methods 0.000 description 5
- 229910045601 alloy Inorganic materials 0.000 description 4
- 239000000956 alloy Substances 0.000 description 4
- 238000005246 galvanizing Methods 0.000 description 4
- 239000012535 impurity Substances 0.000 description 4
- 229910000859 α-Fe Inorganic materials 0.000 description 4
- 238000007796 conventional method Methods 0.000 description 3
- 238000005336 cracking Methods 0.000 description 3
- 239000013078 crystal Substances 0.000 description 3
- 238000007598 dipping method Methods 0.000 description 3
- 238000009713 electroplating Methods 0.000 description 3
- 239000002344 surface layer Substances 0.000 description 3
- 238000009864 tensile test Methods 0.000 description 3
- 238000004458 analytical method Methods 0.000 description 2
- 238000005266 casting Methods 0.000 description 2
- 238000006243 chemical reaction Methods 0.000 description 2
- 210000001787 dendrite Anatomy 0.000 description 2
- 230000006866 deterioration Effects 0.000 description 2
- 238000005516 engineering process Methods 0.000 description 2
- 238000011156 evaluation Methods 0.000 description 2
- 238000000227 grinding Methods 0.000 description 2
- 238000002844 melting Methods 0.000 description 2
- 230000008018 melting Effects 0.000 description 2
- 239000000523 sample Substances 0.000 description 2
- 238000005728 strengthening Methods 0.000 description 2
- 230000003746 surface roughness Effects 0.000 description 2
- 238000004804 winding Methods 0.000 description 2
- 229910018134 Al-Mg Inorganic materials 0.000 description 1
- 229910021365 Al-Mg-Si alloy Inorganic materials 0.000 description 1
- 229910018467 Al—Mg Inorganic materials 0.000 description 1
- 229910007570 Zn-Al Inorganic materials 0.000 description 1
- 229910007567 Zn-Ni Inorganic materials 0.000 description 1
- 229910007614 Zn—Ni Inorganic materials 0.000 description 1
- 230000002411 adverse Effects 0.000 description 1
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 1
- 239000000538 analytical sample Substances 0.000 description 1
- 230000015572 biosynthetic process Effects 0.000 description 1
- 230000015271 coagulation Effects 0.000 description 1
- 238000005345 coagulation Methods 0.000 description 1
- 230000003750 conditioning effect Effects 0.000 description 1
- 230000007797 corrosion Effects 0.000 description 1
- 238000005260 corrosion Methods 0.000 description 1
- 230000000593 degrading effect Effects 0.000 description 1
- 238000009792 diffusion process Methods 0.000 description 1
- 239000006185 dispersion Substances 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 239000000446 fuel Substances 0.000 description 1
- 238000007654 immersion Methods 0.000 description 1
- 230000001771 impaired effect Effects 0.000 description 1
- 230000006698 induction Effects 0.000 description 1
- 238000005461 lubrication Methods 0.000 description 1
- 239000011159 matrix material Substances 0.000 description 1
- 230000003647 oxidation Effects 0.000 description 1
- 238000007254 oxidation reaction Methods 0.000 description 1
- 230000000704 physical effect Effects 0.000 description 1
- OXNIZHLAWKMVMX-UHFFFAOYSA-N picric acid Chemical compound OC1=C([N+]([O-])=O)C=C([N+]([O-])=O)C=C1[N+]([O-])=O OXNIZHLAWKMVMX-UHFFFAOYSA-N 0.000 description 1
- 239000002244 precipitate Substances 0.000 description 1
- 238000003825 pressing Methods 0.000 description 1
- 238000012545 processing Methods 0.000 description 1
- 239000000047 product Substances 0.000 description 1
- 238000007670 refining Methods 0.000 description 1
- 230000003014 reinforcing effect Effects 0.000 description 1
- 229920006395 saturated elastomer Polymers 0.000 description 1
- 239000006104 solid solution Substances 0.000 description 1
- 238000010301 surface-oxidation reaction Methods 0.000 description 1
- 230000009466 transformation Effects 0.000 description 1
- 230000000007 visual effect Effects 0.000 description 1
- 230000037303 wrinkles Effects 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/06—Ferrous alloys, e.g. steel alloys containing aluminium
-
- C—CHEMISTRY; METALLURGY
- C21—METALLURGY OF IRON
- C21D—MODIFYING THE PHYSICAL STRUCTURE OF FERROUS METALS; GENERAL DEVICES FOR HEAT TREATMENT OF FERROUS OR NON-FERROUS METALS OR ALLOYS; MAKING METAL MALLEABLE, e.g. BY DECARBURISATION OR TEMPERING
- C21D8/00—Modifying the physical properties by deformation combined with, or followed by, heat treatment
- C21D8/02—Modifying the physical properties by deformation combined with, or followed by, heat treatment during manufacturing of plates or strips
- C21D8/021—Modifying the physical properties by deformation combined with, or followed by, heat treatment during manufacturing of plates or strips involving a particular fabrication or treatment of ingot or slab
- C21D8/0215—Rapid solidification; Thin strip casting
-
- C—CHEMISTRY; METALLURGY
- C21—METALLURGY OF IRON
- C21D—MODIFYING THE PHYSICAL STRUCTURE OF FERROUS METALS; GENERAL DEVICES FOR HEAT TREATMENT OF FERROUS OR NON-FERROUS METALS OR ALLOYS; MAKING METAL MALLEABLE, e.g. BY DECARBURISATION OR TEMPERING
- C21D8/00—Modifying the physical properties by deformation combined with, or followed by, heat treatment
- C21D8/02—Modifying the physical properties by deformation combined with, or followed by, heat treatment during manufacturing of plates or strips
- C21D8/0221—Modifying the physical properties by deformation combined with, or followed by, heat treatment during manufacturing of plates or strips characterised by the working steps
- C21D8/0226—Hot rolling
-
- C—CHEMISTRY; METALLURGY
- C21—METALLURGY OF IRON
- C21D—MODIFYING THE PHYSICAL STRUCTURE OF FERROUS METALS; GENERAL DEVICES FOR HEAT TREATMENT OF FERROUS OR NON-FERROUS METALS OR ALLOYS; MAKING METAL MALLEABLE, e.g. BY DECARBURISATION OR TEMPERING
- C21D8/00—Modifying the physical properties by deformation combined with, or followed by, heat treatment
- C21D8/02—Modifying the physical properties by deformation combined with, or followed by, heat treatment during manufacturing of plates or strips
- C21D8/0221—Modifying the physical properties by deformation combined with, or followed by, heat treatment during manufacturing of plates or strips characterised by the working steps
- C21D8/0236—Cold rolling
-
- C—CHEMISTRY; METALLURGY
- C21—METALLURGY OF IRON
- C21D—MODIFYING THE PHYSICAL STRUCTURE OF FERROUS METALS; GENERAL DEVICES FOR HEAT TREATMENT OF FERROUS OR NON-FERROUS METALS OR ALLOYS; MAKING METAL MALLEABLE, e.g. BY DECARBURISATION OR TEMPERING
- C21D8/00—Modifying the physical properties by deformation combined with, or followed by, heat treatment
- C21D8/02—Modifying the physical properties by deformation combined with, or followed by, heat treatment during manufacturing of plates or strips
- C21D8/0247—Modifying the physical properties by deformation combined with, or followed by, heat treatment during manufacturing of plates or strips characterised by the heat treatment
- C21D8/0273—Final recrystallisation annealing
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/002—Ferrous alloys, e.g. steel alloys containing In, Mg, or other elements not provided for in one single group C22C38/001 - C22C38/60
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/005—Ferrous alloys, e.g. steel alloys containing rare earths, i.e. Sc, Y, Lanthanides
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/02—Ferrous alloys, e.g. steel alloys containing silicon
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/04—Ferrous alloys, e.g. steel alloys containing manganese
Definitions
- the present invention relates to a steel plate, a surface-treated steel plate, and methods for producing them. More specifically, the present invention relates to a high-strength steel sheet and a surface-treated steel sheet suitable as materials for automobile reinforcing members and automobile seat members, and methods for producing them.
- high-strength steel sheets with excellent workability are attracting more and more attention because they are effective in improving the safety of automobiles and reducing the fuel consumption.
- the number of members made from high-strength steel sheets per car is increasing. For this reason, a very high level of bendability is required even for a high-strength steel sheet having a tensile strength of 590 MPa or more.
- a member including a processed portion having a small bending radius such as a seat rail or a side sill of an automobile is required to have unprecedented bendability and higher strength.
- Patent Document 1 discloses a high-strength thin steel sheet in which the hardness difference between the bainite and the martensite phase and the low-hardness ferrite phase around the bainite and martensite phase is reduced by reducing the hardness of the bainite and martensite phase.
- Patent Document 2 and Patent Document 3 disclose that a ferrite crystal grain is made ultrafine to achieve a cold stretch that has both high stretch strength and stretch strength that require local deformability as well as bendability.
- a rolled steel sheet and a hot dip galvanized steel sheet are disclosed.
- High strength steel sheets generally contain a large amount of Mn in order to increase the strength. Mn is easily segregated in steel. For this reason, the chemical composition of the high-strength steel plate locally varies due to segregation of Mn. Due to the local variation of the chemical composition, a non-uniform structure is formed in the high-strength steel plate. For this reason, as disclosed in Patent Document 1, it is actually extremely difficult to precisely control the hardness of the ferrite phase, the bainite, and the martensite phase throughout the high-strength steel sheet.
- FIG. 1 is an explanatory view showing surface properties after bending deformation of a high-strength steel plate.
- a non-uniform structure is formed on a high-strength steel sheet, as shown in FIG. Since this unevenness promotes non-uniform deformation during bending, cracks are induced in the processed portion, and bendability deteriorates. Even when cracks do not occur, the unevenness present in the processed part remains in the member made from this high-strength steel plate, so that the collision characteristics of this member deteriorate.
- Patent Document 4 discloses a high-strength cold-rolled steel sheet having improved bendability by using a martensite single-phase structure that is the ultimate uniform structure.
- the steel structure is a martensite single-phase structure, the flatness of the high-strength steel sheet is impaired, so that it is difficult to use the steel structure as a material for automobile members that require high dimensional accuracy.
- Patent Document 5 discloses a thin steel plate having a hole expansion ratio and strength increased by making the matrix a ferrite single-phase structure.
- Patent Document 6 discloses a steel material heat treatment method that diffuses segregation by homogenizing a steel material at a high temperature of 1250 ° C. or higher for 10 hours or longer. However, this method cannot completely eliminate segregation. For this reason, a heterogeneous structure is formed by segregation, the unevenness of the processed part is not removed, and the bendability cannot be improved sufficiently.
- the average cooling rate at the position of (1/4) ts of the slab thickness ts is set to 100 ° C./min or more, and the condition is continuously cooled from the liquidus temperature to the solidus temperature.
- a hot dip galvanized steel sheet having excellent seizure reduction and excellent hole expansibility by casting is disclosed.
- this technique cannot be applied to continuous casting of a normal slab having a thickness of 200 to 300 mm.
- Japanese Patent Laid-Open No. 62-13533 Japanese Patent Laid-Open No. 2004-211126 JP 2004-250774 A JP 2002-161336 A JP 2002-322539 A JP-A-4-191322 JP 2007-70649 A JP 2007-70659 A
- An object of this invention is to provide the steel plate and surface-treated steel plate which are excellent in bendability with a tensile strength of 590 Mpa or more, and a manufacturing method thereof.
- excellent bendability means that the minimum bend radius that does not cause cracking in a 180 ° bend test in which the bend axis is the rolling direction is 1.0 t or less, and bending in the same bending direction is performed. It means that irregularities do not appear on the surface of the processed part visually after 90 ° V bending with a radius of 1.0 t. Therefore, unless otherwise specified, the bendability in this specification is evaluated by such physical properties of a steel plate and visual observation of a member produced from the steel plate by bending.
- the minimum bending radius in a 180 ° bending test is 0.5 t or less, and the 90 ° at a bending radius of 0.5 t. It is preferable that irregularities do not appear on the surface of the processed part visually after V bending.
- the present invention can achieve a desired Mn concentration distribution by optimizing the chemical composition and production conditions in a high-strength steel sheet, thereby suppressing the generation of a heterogeneous structure due to segregation of Mn. Therefore, it is based on the knowledge that a high-strength steel sheet excellent in bendability with a tensile strength of 590 MPa or more can be produced.
- the present invention relates to C, Si, Mn, P, S, sol. Content of Al, N, Bi, Ti, Nb, V, Cr, Mo, Cu, Ni, Ca, Mg, REM, Zr and B (In this specification, “%” regarding chemical composition is unless otherwise specified) C: 0.03 to 0.20%, Si: 0.005 to 2.0%, Mn: 1.2 to 3.5%, P ⁇ 0.1%, S ⁇ 0.01%, sol.
- Mn segregation ratio (Mn max / Mn av ) calculated from the average Mn concentration (Mn av ) and the maximum Mn concentration (Mn max ) is less than 1.30.
- the chemical composition contains at least one selected from the following (a) to (d): (a) one or more selected from the group consisting of Ti: 0.003-0.3%, Nb: 0.003-0.3% and V: 0.003-0.3%; (b) One or two selected from the group consisting of Cr: 0.01 to 1%, Mo: 0.01 to 1%, Cu: 0.01 to 1%, and Ni: 0.01 to 1% Or more; (c) From the group consisting of Ca: 0.0001-0.01%, Mg: 0.0001-0.01%, REM: 0.0001-0.01% and Zr: 0.0001-0.01% One or more selected; and (d) B: 0.0003-0.01%.
- the present invention is a surface-treated steel sheet comprising the steel sheet and a plating layer formed on at least one surface of the steel sheet. From another point of view, the present invention is a method for producing a steel sheet, comprising the following steps (A) to (C).
- a continuous casting process in which molten steel having the above-described chemical composition is cast into a slab having a thickness of 200 to 300 mm at a solidification rate at a depth of 10 mm from the surface at 100 to 1000 ° C./min;
- C a continuous annealing process in which the cold-rolled steel sheet obtained in the rolling process is subjected to recrystallization annealing in a temperature range of 750 to 950 ° C.
- the present invention is a method for producing a surface-treated steel sheet, wherein a steel sheet obtained by the above-described production method is subjected to a plating treatment for forming a plating layer on at least one surface thereof.
- a high-strength steel sheet having a strength of 590 MPa or more and excellent bendability can be obtained.
- the steel sheet according to the present invention can be widely used in various industrial fields, particularly in the automobile field.
- the chemical composition of the steel sheet according to the present invention is as follows. (C: 0.03 to 0.20%) C contributes to improving the strength of the steel sheet.
- the C content is set to 0.03% or more in order to increase the tensile strength of the steel plate to 590 MPa or more. If the C content is more than 0.20%, the weldability deteriorates. For this reason, C content is made into 0.20% or less.
- the C content is preferably 0.05% or more in order to easily obtain a tensile strength of 980 MPa or more.
- Si 0.005 to 2.0% If Si is contained in an amount of 0.005% or more, the strength of the steel sheet can be improved without significantly degrading the bendability. If the Si content exceeds 2.0%, the chemical conversion processability deteriorates in the case of a non-plated steel sheet, and the wettability, alloying processability and plating adhesion of a hot dip galvanized steel sheet deteriorate. To do. For this reason, Si content shall be 0.005% or more and 2.0% or less.
- the Si content exceeds 1.5%, an oxide containing Si is formed on the surface of the steel sheet, and the surface properties may deteriorate. For this reason, the Si content is preferably 1.5% or less.
- the Si content is 0.4% or more, 980 MPa or more The tensile strength of can be easily obtained. For this reason, the Si content is preferably 0.4% or more.
- Mn contributes to improving the strength of the steel sheet.
- the Mn content needs to be 1.2% or more in order to make the tensile strength of the steel plate 590 MPa or more. If the Mn content exceeds 3.5%, not only the melting and refining of the steel in the converter becomes difficult, but also the weldability deteriorates. For this reason, Mn content shall be 1.2% or more and 3.5% or less.
- Mn promotes the heterogeneous structure of steel, as described later, since the adverse effect of Mn is mitigated by containing Bi and the structure becomes uniform, deterioration of bendability is suppressed and the strength is improved. .
- the Mn content is preferably 1.8% or more in order to make the tensile strength 980 MPa or more.
- P is an impurity inevitably contained.
- P is also a solid solution strengthening element, and is effective for strengthening the steel sheet, so it may be positively incorporated.
- the P content exceeds 0.1%, the weldability deteriorates. For this reason, the P content is 0.1% or less.
- the P content is preferably 0.003% or more in order to strengthen the steel plate more reliably.
- S is an impurity inevitably contained in the steel.
- the S content is preferably as low as possible from the viewpoints of bendability and weldability. For this reason, S content shall be 0.01% or less.
- the S content is preferably 0.005% or less, and more preferably 0.003% or less.
- Al is contained in the steel for deoxidation of the steel. Al effectively works to improve the yield of carbonitride-forming elements such as Ti.
- Sol. Al content is 0.001% or more. sol. If the Al content is more than 1.0%, the weldability deteriorates and the oxide inclusions increase, so that the surface properties deteriorate. For this reason, sol. Al content shall be 0.001% or more and 1.0% or less. Note that sol. The Al content is preferably 0.01% or more and 0.2% or less.
- N (N ⁇ 0.01%) N is an impurity inevitably contained in the steel.
- the N content is preferably as low as possible from the viewpoint of bendability, so is set to 0.01% or less.
- the N content is preferably 0.006% or less.
- Bi plays an important role in the present invention.
- the solidified structure of the slab becomes finer, and even if the steel contains a large amount of Mn, the structure of the steel sheet becomes uniform, and deterioration of the bendability is suppressed. Therefore, a Bi content of 0.0001% or more is necessary to ensure the desired bendability.
- the Bi content is more than 0.05%, hot workability deteriorates and hot rolling becomes difficult. For this reason, Bi content shall be 0.0001% or more and 0.05% or less.
- the Bi content is preferably 0.0010% or more in order to further improve the bendability.
- Ti, Nb, and V are all optional elements that contribute to improving the strength of the steel sheet and can be contained in the steel as necessary. It is effective for steel to contain one or more of Ti, Nb and V in order to ensure a tensile strength of 980 MPa or more. In order to obtain this effect more reliably, it is preferable to contain 0.003% or more of any one or more elements of Ti, Nb and V. If the content of each of Ti, Nb and V is more than 0.3%, inclusions containing Ti, Nb and V increase, so that the surface properties of the steel sheet deteriorate. For this reason, when it contains at least 1 sort (s) of Ti, Nb, and V, each content shall be 0.3% or less.
- Cr, Mo, Cu, and Ni are all optional elements that can be contained in steel as necessary because they all contribute to improving the strength of the steel sheet.
- the fact that the steel contains one or more of Cr, Mo, Cu and Ni means that when the cold-rolled steel sheet is produced by setting the cooling stop temperature of continuous annealing to 300 ° C. or more and 420 ° C. or less, or hot dip galvanization.
- it is effective to ensure a tensile strength of 980 MPa or more.
- each content of Cr, Mo, Cu and Ni is more than 1%, not only is the above effect saturated and economically wasted, but the hot-rolled steel sheet after hot rolling is hard Therefore, it becomes difficult to perform cold rolling on the hot-rolled steel sheet. For this reason, when containing at least 1 sort (s) of Cr, Mo, Cu, and Ni, each content shall be 1% or less.
- Ca, Mg, REM and Zr all contribute to the inclusion control of the steel, particularly the fine dispersion of the inclusion, and further improve the bendability of the steel sheet, so that it can be contained in the steel as necessary. It is an optional element that can be made. However, when Ca, Mg, REM, and Zr are contained excessively, the surface properties of the steel sheet deteriorate. Therefore, when one or more of Ca, Mg, REM and Zr are contained, the respective contents are set to 0.01% or less. In order to obtain the above effects more reliably, the contents of Ca, Mg, REM, and Zr are preferably set to 0.0001% or more.
- B (B ⁇ 0.01%) B is effective not only for improving the bendability of the steel sheet, but also for securing a tensile strength of 980 MPa or more in the case of producing a hot-dip galvanized steel sheet. It is an optional element that can be made. However, if the B content exceeds 0.01%, the hot-rolled steel sheet becomes hard, and it is difficult to perform cold rolling on the hot-rolled steel sheet. Therefore, the B content is preferably 0.01% or less. In the case of containing B, the B content is preferably set to 0.0003% or more in order to obtain the above effect more reliably.
- the balance other than the components described above consists essentially of Fe and impurities.
- Mn segregation ratio ⁇ 1.30 The steel sheet according to the present invention has a specific Mn distribution. That is, the Mn distribution of the steel sheet satisfies the condition that the Mn segregation ratio (Mn max / Mn av ) is less than 1.30.
- the Mn segregation ratio (Mn max / Mn av ) is an average obtained by analyzing a region not containing MnS with EPMA (Electron Probe Micro Analyzer) at a (1/20) depth position of the plate thickness from the steel plate surface. It is calculated as the ratio of the maximum Mn concentration (Mn max ) to the Mn concentration (Mn av ).
- a Mn segregation ratio of less than 1.30 is an indicator that the structure is uniform. As a result, the bendability of the steel sheet is improved, and unevenness in the bent portion is less likely to occur.
- the chemical composition of steel contains Bi, and the Mn segregation ratio of the steel sheet can be made less than 1.30 by setting the casting speed to a predetermined condition as will be described later.
- the Mn segregation ratio is preferably less than 1.20 in order to further improve the bendability.
- the bendability of the steel sheet is affected by the Mn distribution at the steel sheet surface layer. This is because the deformation in the bending process is larger in the steel plate surface layer than in the thickness center of the steel plate, and the bendability is governed by the deformability of the steel plate surface layer.
- concentration is measured in said depth position near the surface of a steel plate, and Mn segregation ratio is calculated
- the EPMA analysis can measure an area that can sufficiently evaluate the local variation of the Mn distribution, such as a rectangular region of 500 ⁇ m in the rolling direction and a total of 4 mm in the direction perpendicular to the rolling direction. preferable.
- the steel sheet according to the present invention can be used as a surface-treated steel sheet by forming a plating layer on one or both surfaces for the purpose of improving corrosion resistance or the like.
- the plating layer to be formed may be an electroplating layer or a hot dipping layer.
- the electroplating layer include an electrogalvanizing layer and an electro Zn—Ni alloy plating layer.
- the hot dip plating layer hot dip galvanization layer, alloyed hot dip galvanization layer, hot dip aluminum plating layer, hot dip Zn-Al alloy plating layer, hot dip Zn-Al-Mg alloy plating layer and hot dip Zn-Al-Mg-Si alloy A plating layer etc. are illustrated.
- the adhesion amount (or thickness) of these plating layers may be generally employed for this type of plated steel sheet. Two or more plating layers may be provided as desired.
- the thickness of a slab is 200-300mm.
- the thickness of the slab is less than 200 mm, it is difficult to ensure a total rolling reduction of 99.0% or more in hot rolling and cold rolling described later.
- the thickness of the slab exceeds 300 mm, it becomes difficult to ensure a Mn segregation ratio of less than 1.30 at a (1/20) depth position from the steel plate surface. Therefore, the thickness of a slab shall be 200 mm or more and 300 mm or less.
- the slab obtained by this continuous casting process is subjected to a homogenization treatment for 20 minutes or more in a temperature range of 1200 to 1350 ° C., and then a finishing temperature: 800 to 950 ° C. and a winding temperature: 400 to 750
- a hot rolled steel sheet is formed by hot rolling at 0 ° C., and the hot rolled steel sheet is cold rolled to form a cold rolled steel sheet, and the total rolling reduction in hot rolling and cold rolling is set to 99.0% or more.
- the homogenization treatment time is more preferably 1.0 hour or more and 3 hours or less.
- the Mn segregation ratio can be less than 1.20, and the bendability of the steel sheet can be further improved.
- the homogenization time is 3 hours or less, scale loss can be suppressed, productivity can be improved, and manufacturing costs can be reduced.
- Winding temperature 400 ° C to 750 ° C
- the coiling temperature in hot rolling is 400 ° C. or higher, the formation of hard bainite and martensite is suppressed, and subsequent cold rolling can be easily performed.
- the coiling temperature is 750 ° C. or lower, oxidation of the steel sheet surface is suppressed, and good surface properties can be ensured.
- the rough bar after the rough rolling and before the finish rolling is heated by induction heating or the like, and the temperature of the rough bar is made uniform over the entire length. Thereby, the characteristic fluctuation
- Total reduction ratio in hot rolling and cold rolling 99.0% or more
- the hot-rolled steel sheet obtained by the hot rolling step is usually descaled by a conventional method such as pickling, and then cold-rolled to obtain a cold-rolled steel sheet. At this time, it is preferable that the total rolling reduction in hot rolling and cold rolling is 99.0% or more.
- the total rolling reduction is calculated by the following equation.
- Total rolling reduction (%) ⁇ 1 ⁇ (thickness of cold-rolled steel sheet) / (thickness of slab to be subjected to hot rolling) ⁇ ⁇ 100
- the surface roughness of the processed part that occurs after the bending of the steel sheet is not only the Mn segregation ratio, but also the Mn enrichment in which the Mn segregated part caused by solidification segregation becomes a band-like region that extends in the rolling direction in the subsequent rolling process. It is also affected by the thickness of the strip in the thickness direction. By reducing the thickness of the Mn-concentrated band, surface irregularities after processing can be more reliably suppressed, and thereby the bendability of the steel sheet can be improved. For this purpose, it is effective to set the total rolling reduction to 99.0% or more.
- the rolling reduction of cold rolling is preferably 30% or more in order to make the structure of the steel sheet after continuous annealing uniform. Moreover, it is preferable from a viewpoint of ensuring the flatness of a steel plate to perform mild rolling with a rolling reduction of 5% or less before or after pickling to correct the shape. In addition, by performing this mild rolling before pickling, pickling performance is improved and removal of surface concentrating elements is promoted. In the case of a cold-rolled steel sheet, the surface properties are improved.
- Continuous annealing is performed on the cold-rolled steel sheet obtained by the rolling process including the hot rolling and the cold rolling.
- the annealing temperature is preferably 750 ° C. or higher and 950 ° C. or lower. It is preferable from the viewpoint of productivity that the rate of temperature rise to the recrystallization annealing temperature is 1 ° C./sec or more.
- Annealing temperature 750-950 ° C
- the annealing temperature is 750 ° C. or higher, the remaining of the non-recrystallized structure is suppressed and a uniform structure can be obtained with certainty, so that the bendability is further improved.
- an annealing temperature is 950 degrees C or less, the damage of an annealing furnace is suppressed and productivity improves.
- the annealing time is preferably set to 10 seconds or longer in order to completely remove the non-recrystallized structure and stably ensure good bendability.
- the annealing time is preferably 300 seconds or less from the viewpoint of productivity.
- the average cooling rate from 650 ° C. to 550 ° C. may be set to 5 ° C./sec or more in order to secure a high tensile strength of 590 MPa or more while suppressing the addition of alloy elements leading to high costs. preferable.
- temper rolling it is preferable to perform temper rolling on the steel sheet after annealing (after plating when hot dip galvanization is applied). By performing temper rolling, it is possible to suppress the occurrence of yield point elongation and to prevent seizure and galling during pressing.
- the elongation of temper rolling is preferably 0.05% or more and 1% or less.
- Hot dip galvanization may be performed by a conventional method.
- the steel sheet is immersed in a hot dip galvanizing bath at 410 ° C. or higher and 490 ° C. or lower, and the plating adhesion amount is controlled by a gas wiping nozzle immediately after leaving the plating bath.
- Alloying treatment may be performed after immersion in a hot dip galvanizing bath.
- the alloying treatment temperature is preferably 460 ° C. or more and 600 ° C. or less.
- the alloying treatment temperature is less than 460 ° C., an unalloyed portion is generated, and the surface properties of the steel sheet are likely to deteriorate.
- the alloying treatment temperature is higher than 600 ° C., powdering of the plating film tends to occur.
- the steel sheet cooled after annealing may be electroplated after an appropriate surface conditioning treatment according to a conventional method.
- the obtained plated steel sheet may be subjected to well-known post-treatment (for example, chemical conversion treatment, lubrication treatment).
- the Mn segregation ratio at the (1/20) depth position of the plate thickness from the steel plate surface is less than 1.30, and the steel plate and the surface excellent in bendability while being high strength A treated steel sheet can be easily manufactured.
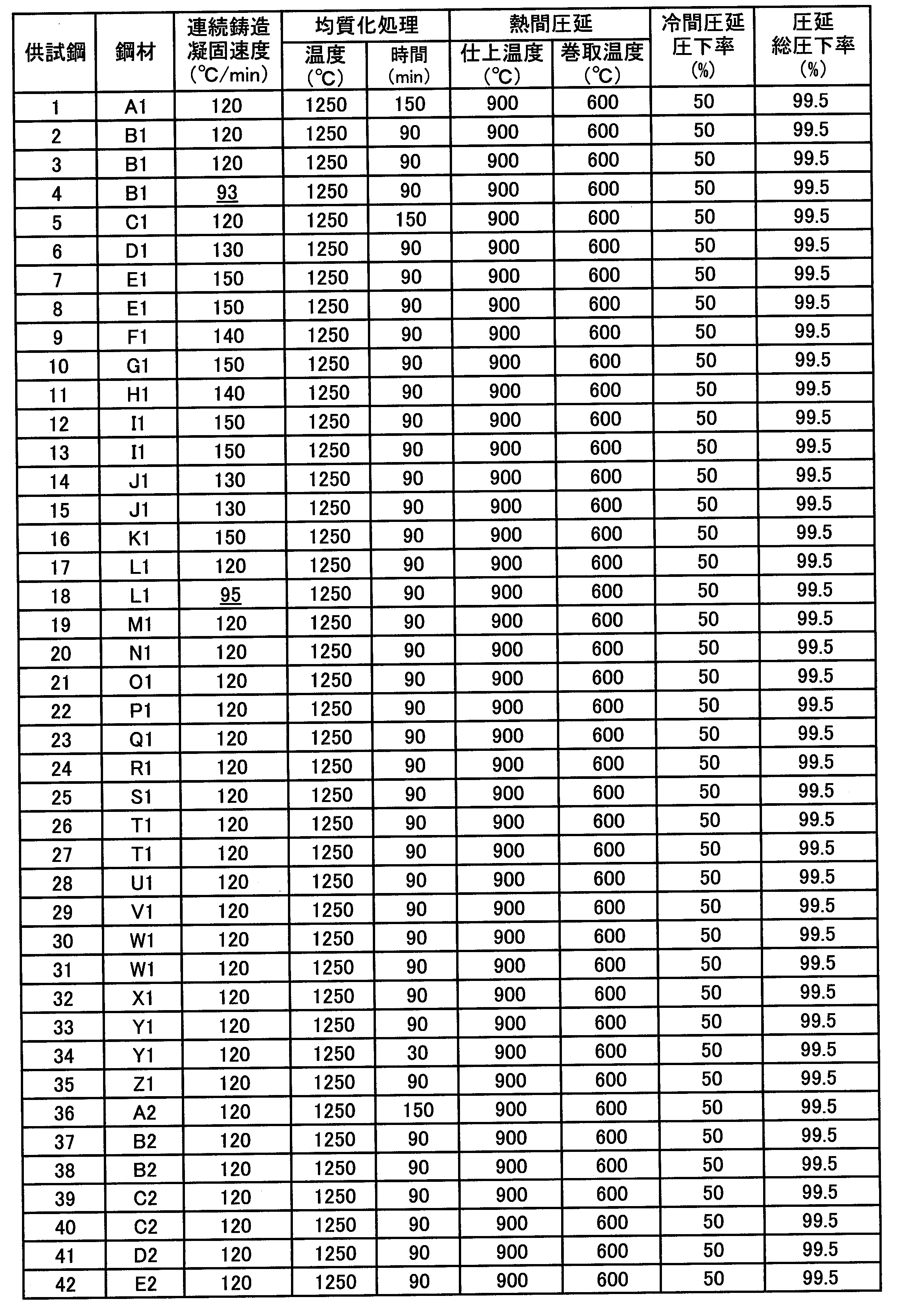
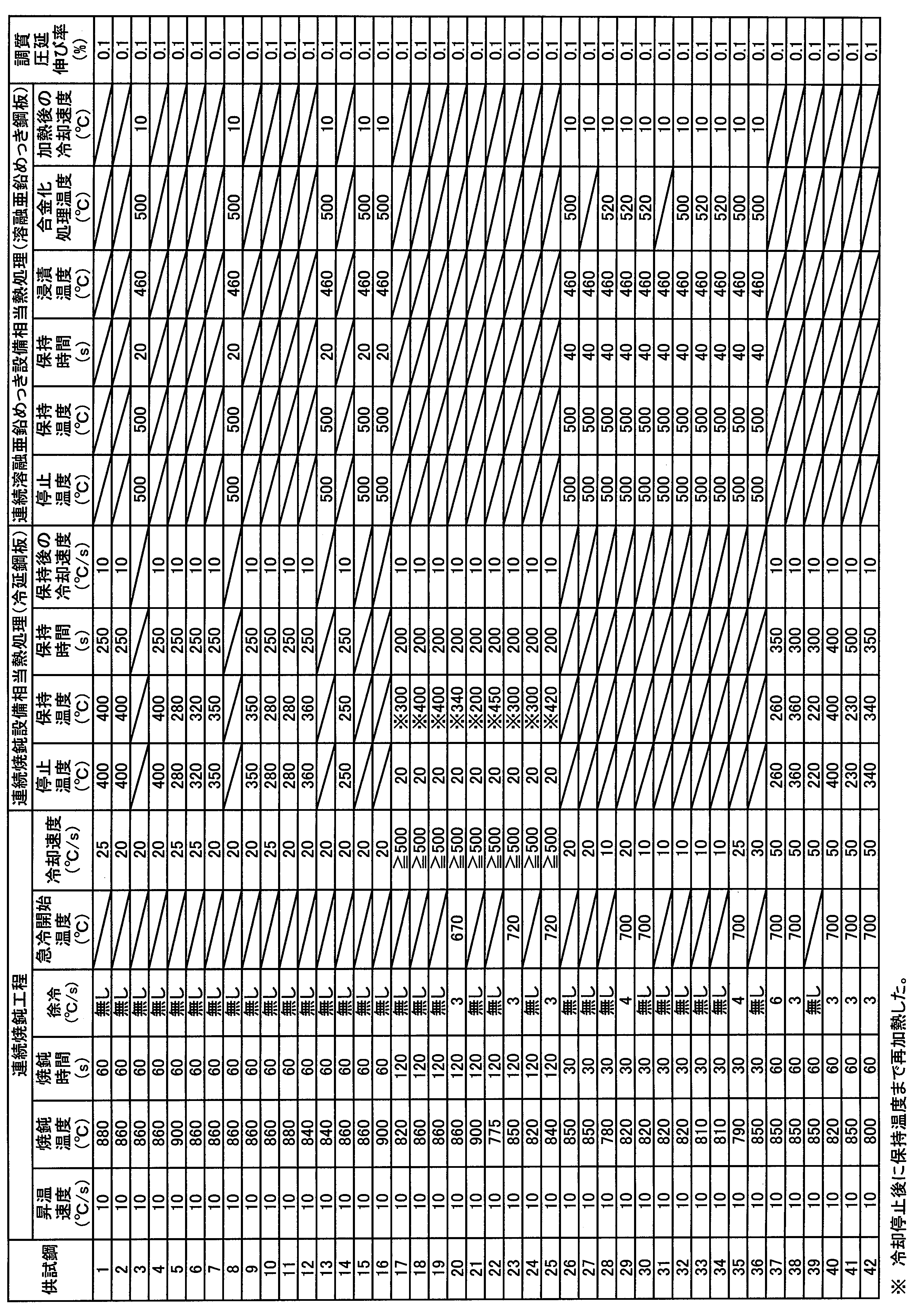
- a test material for heat treatment was collected from the obtained cold-rolled steel sheet, and heat treatment corresponding to a heat pattern in a continuous annealing facility or a continuous hot dip galvanizing facility was performed as shown in Table 3.
- the specimen cold-rolled steel sheets obtained under various production conditions were analyzed by EPMA to investigate the Mn distribution.
- a tensile test and a bending test in which the bending ridge line is in the rolling direction were performed on the test cold-rolled steel sheet to evaluate mechanical properties.
- Tensile test A JIS No. 5 tensile test piece was taken from each test cold-rolled steel sheet from the direction perpendicular to the rolling direction, and the tensile strength (TS) was measured.
- the bending radius standardized by the thickness (t) is obtained, and the minimum bending when no crack is observed after the test.
- the radius shown as Rmin in Table 4
- the minimum bending radius was set to> 1.0 t.
- a 90 ° V bending test (bending radius 1.0t) was performed on this test piece by pushing it with a 90 ° punch with a radius of 1.2mm at the tip, and the presence or absence of surface irregularities was confirmed visually. did. In the evaluation of the surface properties, those having irregularities were judged as bad and those having no irregularities as good. For cold-rolled steel sheets having good surface properties and a minimum bending radius of 0.5 t or less in the above bending test, 90 ° having a radius of 0.6 mm at the tip with respect to the test piece collected in the same manner as above. Further, a 90 ° V bending test (bending radius of 0.5 t) was performed by pushing with a punch, and the presence or absence of surface irregularities was confirmed visually. The evaluation of the surface property is the same as described above.
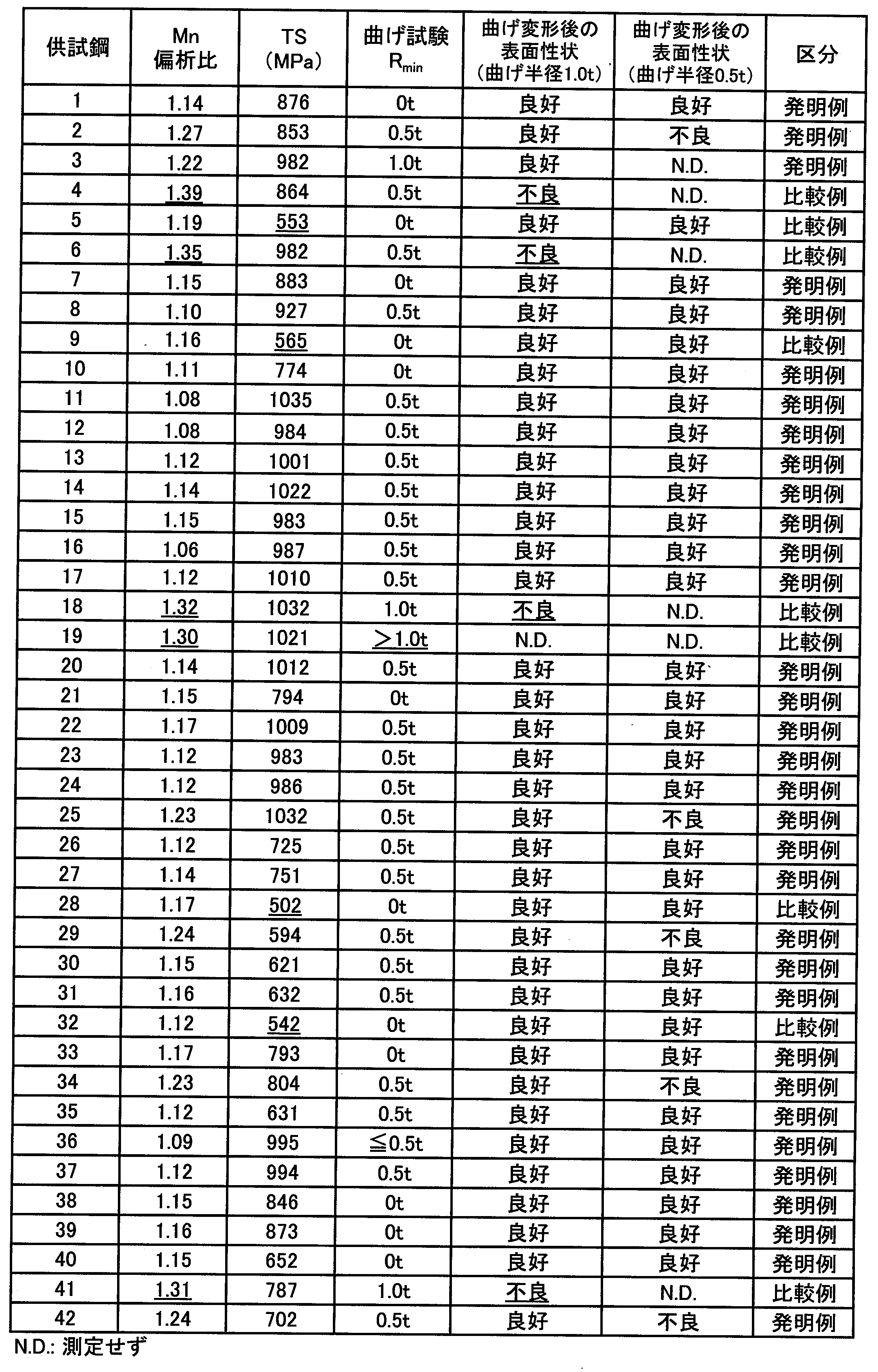
- Specimen Nos. 1 to 3, 7, 8, 10 to 17, 20 to 27, 29 to 31, 33 to 40 and 42 in Table 4 are steel plates of the present invention examples that satisfy all the conditions of the present invention. .
- sample materials Nos. 4 and 18 had a Mn segregation ratio exceeding 1.30 because the solidification rate at a position 10 mm deep from the surface in the continuous casting process was below the lower limit defined in the present invention. The bendability was poor, or the surface properties after bending deformation were poor.
- test materials No. 6, 19 and 41 did not contain Bi, the Mn segregation ratio exceeded 1.30 and the bendability was poor, or the surface properties after bending deformation were poor.
- the C content or the Mn content was lower than the lower limit defined in the present invention, and thus the desired tensile strength could not be obtained.
- the steel sheets of the examples of the present invention all had a tensile strength of 590 MPa or more, and had good bendability and surface properties after bending deformation.
- the steel plates 1, 7, 8, 10 to 17, 20 to 24, 26, 27, 30, 31, 33, and 35 to 40 have a Bi content of 0.0010% or more, 0 or more, which is the above-described preferable range.
- the homogenization temperature and the homogenization time are 1200 ° C. or higher, 1350 ° C. or lower, 1.0 hour or longer and 3 hours or shorter, and the Mn segregation ratio is 1. Since it was less than 20, the tensile strength was 590 MPa or more, and the bendability was further excellent.
Landscapes
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Mechanical Engineering (AREA)
- Materials Engineering (AREA)
- Metallurgy (AREA)
- Organic Chemistry (AREA)
- Physics & Mathematics (AREA)
- Thermal Sciences (AREA)
- Crystallography & Structural Chemistry (AREA)
- Heat Treatment Of Sheet Steel (AREA)
Abstract
Description
不均一組織の起源である偏析そのものを拡散によって解消する技術も提案されている。特許文献6には、鋼材を1250℃以上の高温に10時間以上の長時間保持する均質化(homogenization)を行うことによって偏析を拡散させる鋼材の熱処理方法が開示される。しかし、この方法は偏析を完全には消滅させることができない。このため、偏析によって不均一組織が形成され、加工部の凹凸が除去されず、曲げ性を十分に改善できない。
本発明に関して「曲げ性に優れる」とは、曲げ稜線(bend axis)が圧延方向となる180゜曲げ試験における割れを生じない最小曲げ半径が1.0t以下であって、同じ曲げ方向での曲げ半径1.0tの90°V曲げ後に目視で加工部の表面に凹凸が出現しないことを意味する。したがって、本明細書における曲げ性は、特に断りがない限り、鋼板のそのような物性とその鋼板から曲げ加工で作製された部材の目視観察とによって評価される。本発明に係る鋼板を、厳しい曲げ性が要求されるシートレールの素材として用いる場合には、180゜曲げ試験における最小曲げ半径が0.5t以下であって、曲げ半径0.5tでの90°V曲げ後に目視で加工部の表面に凹凸が出現しないことが好ましい。
(a)Ti:0.003~0.3%、Nb:0.003~0.3%およびV:0.003~0.3%からなる群から選ばれた1種または2種以上;
(b)Cr:0.01~1%、Mo:0.01~1%、Cu:0.01~1%およびNi:0.01~1%からなる群から選ばれた1種または2種以;
(c)Ca:0.0001~0.01%、Mg:0.0001~0.01%、REM:0.0001~0.01%およびZr:0.0001~0.01%からなる群から選ばれた1種または2種以上;ならびに
(d)B:0.0003~0.01%。
別の観点からは、本発明は、下記工程(A)~(C)を備えることを特徴とする鋼板の製造方法である。
(B)前記連続鋳造工程で得られたスラブに熱間圧延を施して熱延鋼板とし、この熱延鋼板に冷間圧延を施して冷延鋼板とする、熱間圧延および冷間圧延を含む圧延工程;ならびに
(C)前記圧延工程で得られた冷延鋼板に、750~950℃の温度域で再結晶焼鈍を施す連続焼鈍工程。
(C:0.03~0.20%)
Cは鋼板の強度向上に寄与する。C含有量は、鋼板の引張強度を590MPa以上にするために、0.03%以上とする。C含有量が0.20%超であると溶接性が劣化する。このため、C含有量は0.20%以下とする。C含有量は、980MPa以上の引張強度を容易に得るために、好ましくは0.05%以上である。
Siは、0.005%以上含有させると、曲げ性をあまり劣化することなく、鋼板の強度を向上することができる。Si含有量が2.0%超であると、非めっき鋼板の場合には化成処理性が劣化し、溶融亜鉛めっき鋼板の場合にはめっきの濡れ性、合金化処理性及びめっき密着性が劣化する。このため、Si含有量は0.005%以上、2.0%以下とする。
Mnは、鋼板の強度向上に寄与する。Mn含有量は、鋼板の引張強度を590MPa以上にするために1.2%以上が必要である。Mn含有量が3.5%超であると、転炉における鋼の溶解や精錬が困難になるだけでなく、溶接性が劣化する。このため、Mn含有量は1.2%以上、3.5%以下とする。Mnは鋼の不均一組織を助長するが、後述するように、Biを含有することによってMnのこの悪影響が緩和されて組織が均一となるので、曲げ性の劣化が抑制されて強度が向上する。なお、Mn含有量は、引張強度を980MPa以上にするために、好ましくは1.8%以上である。
Pは、一般には不可避的に含有される不純物である。しかし、Pは固溶強化元素でもあり、鋼板の強化に有効であるので、積極的に含有させてもよい。しかし、P含有量が0.1%超であると溶接性が劣化する。このため、P含有量は0.1%以下とする。P含有量は、鋼板をより確実に強化するために、好ましくは0.003%以上である。
Sは、鋼に不可避的に含有される不純物である。S含有量は、曲げ性及び溶接性の観点からは低いほど好ましい。このため、S含有量は0.01%以下とする。S含有量は、好ましくは0.005%以下であり、さらに好ましくは0.003%以下である。
Alは、鋼の脱酸のために鋼中に含有される。Alは、Ti等の炭窒化物形成元素の歩留まりを向上させるのに有効に作用する。それに必要なsol.Al含有量は0.001%以上である。sol.Al含有量が1.0%超であると、溶接性が劣化するとともに、酸化物系介在物が増加するために表面性状が劣化する。このため、sol.Al含有量は0.001%以上、1.0%以下とする。なお、sol.Al含有量は、好ましくは0.01%以上、0.2%以下である。
Nは、鋼に不可避的に含有される不純物である。N含有量は、曲げ性の観点からは低いほど好ましいので、0.01%以下とする。N含有量は好ましくは0.006%以下である。
Biは、本発明において重要な役割を果たす。鋼がBiを含有することによって、スラブの凝固組織が微細化し、鋼がMnを多量に含有しても鋼板の組織が均一となり、その曲げ性の劣化が抑制される。したがって、所望の曲げ性を確保するために0.0001%以上のBi含有量が必要である。しかし、Bi含有量が0.05%超であると、熱間加工性が劣化して熱間圧延が困難になる。このため、Bi含有量は0.0001%以上、0.05%以下とする。Bi含有量は、曲げ性をさらに向上させるために、好ましくは0.0010%以上である。
Ti、NbおよびVは、いずれも鋼板の強度向上に寄与し、必要に応じて鋼に含有させることができる任意元素である。鋼がTi、NbおよびVの1種または2種以上を含有することは、980MPa以上の引張強度を確保するために有効である。この効果をより確実に得るには、Ti、NbおよびVの何れか1種以上の元素を0.003%以上含有することが好ましい。Ti、NbおよびVそれぞれの含有量が0.3%超であると、Ti、NbやVを含む介在物が増加するため、鋼板の表面性状が劣化する。このため、Ti、NbおよびVの少なくとも1種を含有させる場合、それぞれの含有量は0.3%以下とする。
Cr、Mo、CuおよびNiは、いずれも鋼板の強度向上に寄与するので、必要に応じて鋼に含有させることができる任意元素である。鋼がCr、Mo、CuおよびNiの1種または2種以上を含有することは、連続焼鈍の冷却停止温度を300℃以上、420℃以下にして冷延鋼板を製造する場合や、溶融亜鉛めっき鋼板を製造する場合において、980MPa以上の引張強度を確保するのに有効である。この効果をより確実に得るには、Cr、Mo、CuおよびNiの少なくとも1種を0.01%以上含有することが好ましい。ただし、Cr、Mo、CuおよびNiのそれぞれの含有量が1%超であると、上記効果が飽和して経済的に無駄となるだけでなく、熱間圧延を終了した熱延鋼板が硬質となるため、この熱延鋼板に対して冷間圧延を行うことが困難になる。このため、Cr、Mo、CuおよびNiの少なくとも1種を含有させる場合、それぞれの含有量は1%以下とする。
Ca、Mg、REMおよびZrは、いずれも、鋼の介在物制御、特に介在物の微細分散化に寄与して、鋼板の曲げ性をさらに向上するので、必要に応じて鋼に含有させることができる任意元素である。しかし、Ca、Mg、REM、Zrを過剰に含有すると、鋼板の表面性状が劣化する。そこで、Ca、Mg、REMおよびZrの1種以上を含有させる場合、それぞれの含有量は0.01%以下とする。上記効果をより確実に得るためには、Ca、Mg、REMおよびZrのそれぞれの含有量を0.0001%以上とすることが好ましい。
Bは、鋼板の曲げ性向上に寄与するだけでなく、溶融亜鉛めっき鋼板を製造する場合において、980MPa以上の引張強度を確保するのに有効であるので、必要に応じて鋼に含有させることができる任意元素である。ただし、B含有量が0.01%超であると、熱延鋼板が硬質となって、この熱延鋼板に対して冷間圧延を行うことが困難になる。このため、B含有量は0.01%以下とすることが好ましい。Bを含有させる場合、上記効果をより確実に得るには、B含有量を0.0003%以上とすることが好ましい。
(Mn偏析比<1.30)
本発明に係る鋼板は特定のMn分布を有する。即ち、鋼板のMn分布は、Mn偏析比(Mnmax/Mnav)が1.30未満という条件を満たす。Mn偏析比(Mnmax/Mnav)は、鋼板表面から板厚の(1/20)深さ位置において、MnSを含まない領域をEPMA(Electron Probe Micro Analyzer)で分析することにより求められる、平均のMn濃度(Mnav)に対する最大Mn濃度(Mnmax)の比として算出される。Mn偏析比が1.30未満であることは、組織が均一であることの指標である。その結果、鋼板の曲げ性が向上して、曲げ加工部における凹凸が発生し難くなる。鋼の化学組成がBiを含有するとともに、後述するように、鋳造速度を所定の条件とすることによって、鋼板のMn偏析比を1.30未満とすることができる。Mn偏析比は、曲げ性をさらに向上するために、好ましくは1.20未満である。
本発明に係る鋼板は、その片面または両面の表面に耐食性の向上等を目的としてめっき層を形成することによって、表面処理鋼板として用いることもできる。
[連続鋳造工程]
上述した化学組成を有する溶鋼を、転炉や電気炉等の公知の溶製方法によって溶製する。この溶鋼を、スラブ表面から10mm深さ位置における凝固速度を100~1000℃/minとして、200~300mmの厚さのスラブに連続鋳造する。
連続鋳造工程におけるスラブ表面から10mm深さ位置における凝固速度が100℃/min未満であると、スラブ表面からスラブ厚の(1/20)深さ位置におけるデンドライト一次アーム間隔を微細化することができず、Mnの偏析が十分に防止されず、鋼板の曲げ性を改善できない場合がある。凝固速度が1000℃/minを超えると、スラブの表面割れが誘発される場合がある。そのため、この凝固速度は100℃/min以上、1000℃/min以下とする。
スラブの厚さが200mm未満であると、後述する熱間圧延および冷間圧延において99.0%以上の総圧下率を確保することが困難となる。スラブの厚さが300mmを超えると、鋼板表面から板厚の(1/20)深さ位置において、1.30未満のMn偏析比を確保することが困難となる。そのため、スラブの厚さは200mm以上,300mm以下とする。
上記連続鋳造工程により得られたスラブに、熱間圧延を施して熱延鋼板とし、さらにこの熱延鋼板に冷間圧延を施して冷延鋼板とする。
熱間圧延に供するスラブを1200℃以上の温度域に20分間以上保持することにより、Mnの偏析に起因する不均一組織がさらに解消し、鋼板の曲げ性がさらに向上する。均質化処理温度が1350℃以下であることは、スケールロスの抑制、加熱炉の損傷の防止および生産性の向上といった観点から好ましい。
熱間圧延の仕上温度が800℃以上であると、熱間圧延時の変形抵抗を小さくすることができ、操業をより容易に行うことができる。仕上温度が950℃以下であると、スケールによる疵をさらに確実に抑制することができ、良好な表面性状を確保できる。
熱間圧延における巻取温度が400℃以上であると、硬質なベイナイトやマルテンサイトの生成が抑制され、その後の冷間圧延を容易に行うことができる。また、巻取温度が750℃以下であることにより、鋼板表面の酸化が抑制され、良好な表面性状を確保できる。
上記熱間圧延工程により得られた熱延鋼板は、通常は酸洗等の常法により脱スケール処理を受けた後、冷間圧延が行われて冷延鋼板とされる。このときの熱間圧延および冷間圧延における総圧下率を99.0%以上とすることが好ましい。ここで、総圧下率は次式で算出される。
鋼板の曲げ加工後に発生する加工部の表面凹凸は、Mn偏析比だけでなく、凝固偏析により生じたMn偏析部がその後の圧延工程において圧延方向に展伸した帯状の領域となったMn濃化帯の板厚方向の厚みにも影響される。このMn濃化帯の厚みを低減することによって、加工後の表面凹凸をより確実に抑制することができ、これにより、鋼板の曲げ性を改善できる。このために、総圧下率を99.0%以上とすることが有効である。
上記熱間圧延および冷間圧延を含む圧延工程により得られた冷延鋼板に連続焼鈍を行う。焼鈍温度は750℃以上、950℃以下とすることが好ましい。再結晶焼鈍温度までの昇温速度が1℃/sec以上であることが、生産性の観点から好ましい。
焼鈍温度が750℃以上であると、未再結晶組織の残存が抑制されて均一な組織を確実に得ることができるので、曲げ性がさらに向上する。また、焼鈍温度が950℃以下であることにより、焼鈍炉の損傷が抑制されて生産性が向上する。
鋼板の表面に溶融亜鉛めっきを施す場合には、焼鈍工程において焼鈍後の冷却を460℃以上、550℃以下で停止し、焼鈍した鋼板を直ちに溶融めっき浴に浸漬して連続的にめっきを行うことが好ましい。この時の冷却停止温度が460℃未満であると、めっき浴浸入時の抜熱が大きくなり、めっき操業が困難になることがある。冷却停止温度が550℃超である場合にも、めっき操業が困難になることがある。
溶融めっきまたは電気めっきの後、用途によっては、得られためっき鋼板に、周知の後処理(例えば、化成処理、潤滑処理など)を施してもよい。
(平均凝固速度)
得られたスラブの断面をピクリン酸によりエッチングし、スラブ表皮より内部に10mmの位置において、5箇所のデンドライト2次アーム間隔λ(μm)を測定し、下記式に基づいて、その値からスラブの液相線温度~固相線温度内における冷却速度A(℃/min)を算出した。
(EPMA分析)
各供試用冷延鋼板の圧延面を研削およびバフ研磨し、表面から板厚の(1/20)深さ位置の分析面を現出させた分析用サンプルを作製して、EPMAでMn分布を調査した。MnSを含まない領域を選択し、ビーム径を10μmとし、圧延方向に500μm、圧延方向に対して直角方向に総計4mmの領域を測定し、500μm幅で平均された圧延方向に対して直角方向のMn濃度分布を解析した。得られたMn濃度分布より、平均のMn濃度(Mnav)と最大のMn濃度(Mnmax)からMn偏析比(Mnmax/Mnav)を算出した。
各供試用冷延鋼板から、圧延方向に対して直角方向からJIS 5号引張試験片を採取し、引張強度(TS)を測定した。
各供試用冷延鋼板から、曲げ稜線が圧延方向となるように、圧延方向に対して直角方向が長手方向と一致する曲げ試験片(幅40mm×長さ100mm×板厚1.2mm)を採取した。厚み2.4mmの鋼板を挟んだ180゜曲げ試験(曲げ半径1.0t)を実施し、割れの有無を目視にて確認した。割れが無かった冷延鋼板については、上と同様に採取した曲げ試験片に対して厚み1.2mmの鋼板を挟んだ180゜曲げ試験(曲げ半径0.5t)をさらに実施し、同様に割れの有無を確認した。この試験でも割れが無い冷延鋼板に対しては、鋼板を挟まない180゜曲げ試験(密着曲げ試験、曲げ半径0t)をさらに実施し、同様に割れの有無を確認した。
(曲げ変形後の表面性状)
上記曲げ試験において最小曲げ半径が1.0t以下であった各冷延鋼板から、曲げ稜線が圧延方向となるように、圧延方向に対して直角方向が長手方向と一致する曲げ試験片(幅40mm×長さ60mm×板厚1.2mm)を採取した。この試験片に対して、先端に1.2mmの半径を持つ90゜のポンチで押し込むことにより90°V曲げ試験(曲げ半径1.0t)を実施し、表面の凹凸の有無を目視にて確認した。表面性状の評価は、凹凸が有るものを不良、無いものを良好とした。表面性状が良好で、かつ上記曲げ試験における最小曲げ半径が0.5t以下であった冷延鋼板については、上と同様に採取した試験片に対して先端に0.6mmの半径を持つ90゜のポンチで押し込むことによる90°V曲げ試験(曲げ半径0.5t)をさらに実施し、表面の凹凸の有無を目視にて確認した。表面性状の評価は上記と同じである。
これらの結果を表4に示す。
これに対し、供試材No.4および18は、連続鋳造工程における、表面から10mm深さ位置における凝固速度が本発明で規定する下限値を下回っていたため、Mn偏析比が1.30超となり、曲げ性が悪いか、または曲げ変形後の表面性状が不良となった。
供試材No.5、9、28および32は、C含有量もしくはMn含有量が本発明で規定する下限値を下回っていたため、所望の引張強度を得られなかった。
Claims (8)
- C、Si、Mn、P、S、sol.Al、N、Bi、Ti、Nb、V、Cr、Mo、Cu、Ni、Ca、Mg、REM、ZrおよびBの含有量が、質量%で、C:0.03~0.20%、Si:0.005~2.0%、Mn:1.2~3.5%、P≦0.1%、S≦0.01%、sol.Al:0.001~1.0%、N≦0.01%、Bi:0.0001~0.05%、Ti:0~0.3%、Nb:0~0.3%、V:0~0.3%、Cr:0~1%、Mo:0~1%、Cu:0~1%、Ni:0~1%、Ca:0~0.01%、Mg:0~0.01%、REM:0~0.01%、Zr:0~0.01%、およびB:0~0.01%である化学組成を有し、かつ鋼板表面から板厚の(1/20)深さ位置における平均Mn濃度(Mnav)と最大Mn濃度(Mnmax)とから算出されるMn偏析比(Mnmax/Mnav)が1.30未満であることを特徴とする鋼板。
- 前記化学組成が、質量%で、Ti:0.003~0.3%、Nb:0.003~0.3%およびV:0.003~0.3%からなる群から選ばれた1種または2種以上を含有する、請求項1に記載の鋼板。
- 前記化学組成が、質量%で、Cr:0.01~1%、Mo:0.01~1%、Cu:0.01~1%およびNi:0.01~1%からなる群から選ばれた1種または2種以上を含有する、請求項1または請求項2に記載の鋼板。
- 前記化学組成が、質量%で、Ca:0.0001~0.01%、Mg:0.0001~0.01%、REM:0.0001~0.01%およびZr:0.0001~0.01%からなる群から選ばれた1種または2種以上を含有する、請求項1から請求項3までのいずれか1項に記載の鋼板。
- 前記化学組成が、質量%で、B:0.0003~0.01%を含有する、請求項1から請求項4までのいずれか1項に記載の鋼板。
- 請求項1から請求項5までのいずれか1項に記載の鋼板と、該鋼板の少なくとも片面の表面に形成されためっき層とを備えることを特徴とする表面処理鋼板。
- 下記工程(A)~(C)を含むことを特徴とする鋼板の製造方法:
(A)請求項1から請求項5までのいずれか1項に記載の化学組成を有する溶鋼を、表面から10mm深さ位置における凝固速度を100~1000℃/minとして200~300mmの厚さのスラブに鋳造する連続鋳造工程;
(B)前記連続鋳造工程により得られたスラブに熱間圧延を施して熱延鋼板とし、前記熱延鋼板に冷間圧延を施して冷延鋼板とする、熱間圧延および冷間圧延を含む圧延工程;ならびに
(C)前記圧延工程で得られた冷延鋼板に750~950℃の温度域で再結晶焼鈍を施す連続焼鈍工程。 - 請求項7に記載の製造方法により得られた鋼板に、その少なくとも片面の表面にめっき層を形成するめっき処理を施すことを特徴とする表面処理鋼板の製造方法。
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP09827543.1A EP2371978B1 (en) | 2008-11-19 | 2009-11-17 | Steel sheet and surface-treated steel sheet |
| ES09827543.1T ES2672070T3 (es) | 2008-11-19 | 2009-11-17 | Chapa de acero y chapa de acero tratada superficialmente |
| KR1020117013957A KR101304009B1 (ko) | 2008-11-19 | 2009-11-17 | 강판 및 표면 처리 강판 및 그들의 제조 방법 |
| PL09827543T PL2371978T3 (pl) | 2008-11-19 | 2009-11-17 | Blacha stalowa cienka i blacha stalowa cienka poddana obróbce powierzchni |
| CN200980154581.1A CN102282280B (zh) | 2008-11-19 | 2009-11-17 | 钢板和表面处理钢板以及钢板和表面处理钢板的制造方法 |
Applications Claiming Priority (8)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2008-295900 | 2008-11-19 | ||
| JP2008-295898 | 2008-11-19 | ||
| JP2008295900A JP5206352B2 (ja) | 2008-11-19 | 2008-11-19 | 鋼板およびその製造方法 |
| JP2008295897A JP5206349B2 (ja) | 2008-11-19 | 2008-11-19 | 鋼板および表面処理鋼板ならびにそれらの製造方法 |
| JP2008-295899 | 2008-11-19 | ||
| JP2008-295897 | 2008-11-19 | ||
| JP2008295899A JP5206351B2 (ja) | 2008-11-19 | 2008-11-19 | 鋼板およびその製造方法 |
| JP2008295898A JP5206350B2 (ja) | 2008-11-19 | 2008-11-19 | 溶融亜鉛めっき鋼板およびその製造方法 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2010058762A1 true WO2010058762A1 (ja) | 2010-05-27 |
Family
ID=42198202
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2009/069464 WO2010058762A1 (ja) | 2008-11-19 | 2009-11-17 | 鋼板および表面処理鋼板ならびにそれらの製造方法 |
Country Status (6)
| Country | Link |
|---|---|
| EP (1) | EP2371978B1 (ja) |
| KR (1) | KR101304009B1 (ja) |
| CN (1) | CN102282280B (ja) |
| ES (1) | ES2672070T3 (ja) |
| PL (1) | PL2371978T3 (ja) |
| WO (1) | WO2010058762A1 (ja) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2011231367A (ja) * | 2010-04-27 | 2011-11-17 | Sumitomo Metal Ind Ltd | 溶融亜鉛めっき鋼板およびその製造方法 |
| JP2012125824A (ja) * | 2010-12-17 | 2012-07-05 | Sumitomo Metal Ind Ltd | 高強度鋼板用の連続鋳造鋳片およびその連続鋳造方法、ならびに高強度鋼板 |
Families Citing this family (17)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| ES2437715B1 (es) * | 2012-07-10 | 2014-10-24 | Gerdau Investigación Y Desarrollo Europa, S.A. | Procedimiento para la fabricación de acero |
| KR101750643B1 (ko) * | 2013-10-02 | 2017-06-23 | 신닛테츠스미킨 카부시키카이샤 | 시효 경화성 강 |
| WO2015092929A1 (ja) | 2013-12-20 | 2015-06-25 | 新日鐵住金株式会社 | 熱間プレス鋼板部材、その製造方法及び熱間プレス用鋼板 |
| US10253387B2 (en) | 2013-12-27 | 2019-04-09 | Nippon Steel & Sumitomo Metal Corporation | Hot-pressed steel sheet member, method of manufacturing the same, and steel sheet for hot pressing |
| MX2016008169A (es) | 2013-12-27 | 2016-09-29 | Nippon Steel & Sumitomo Metal Corp | Miembro de lamina de acero prensada en caliente, metodo de fabricacion del mismo, y lamina de acero para prensado en caliente. |
| ES2745428T3 (es) | 2014-01-06 | 2020-03-02 | Nippon Steel Corp | Acero y método para fabricar el mismo |
| US10508317B2 (en) | 2014-07-18 | 2019-12-17 | Nippon Steel Corporation | Steel product and manufacturing method of the same |
| KR101630976B1 (ko) * | 2014-12-08 | 2016-06-16 | 주식회사 포스코 | 표면품질 및 도금 밀착성이 우수한 초고강도 용융아연도금강판 및 그 제조방법 |
| RU2686715C1 (ru) | 2015-04-08 | 2019-04-30 | Ниппон Стил Энд Сумитомо Метал Корпорейшн | Элемент из термообработанного стального листа и способ его производства |
| US10822680B2 (en) * | 2015-04-08 | 2020-11-03 | Nippon Steel Corporation | Steel sheet for heat treatment |
| BR112017019994A2 (ja) | 2015-04-08 | 2018-06-19 | Nippon Steel & Sumitomo Metal Corporation | A heat treatment steel plate member and a manufacturing method for the same |
| WO2017017961A1 (ja) * | 2015-07-29 | 2017-02-02 | Jfeスチール株式会社 | 冷延鋼板、めっき鋼板及びこれらの製造方法 |
| JP6737338B2 (ja) | 2016-08-08 | 2020-08-05 | 日本製鉄株式会社 | 鋼板 |
| SG11201903691UA (en) * | 2016-11-04 | 2019-05-30 | Nippon Steel Corp | Surface-treated steel sheet |
| CN107365944A (zh) * | 2017-07-29 | 2017-11-21 | 华北理工大学 | 热轧u型钢板桩及其成形方法 |
| CN108057862A (zh) * | 2017-12-28 | 2018-05-22 | 安徽东升精密铸钢件有限公司 | 一种双辊带坯的铸造方法 |
| WO2020044445A1 (ja) * | 2018-08-28 | 2020-03-05 | Jfeスチール株式会社 | 熱延鋼板及びその製造方法、冷延鋼板及びその製造方法、冷延焼鈍鋼板の製造方法、並びに溶融亜鉛めっき鋼板の製造方法 |
Citations (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS6213533A (ja) | 1985-07-09 | 1987-01-22 | Nippon Steel Corp | 曲げ特性の優れた高強度薄鋼板の製造方法 |
| JPH04191322A (ja) | 1990-11-26 | 1992-07-09 | Nkk Corp | 鋼材の均質化処理方法 |
| JP2002161336A (ja) | 2000-09-12 | 2002-06-04 | Nkk Corp | 超高張力冷延鋼板およびその製造方法 |
| JP2002322539A (ja) | 2001-01-31 | 2002-11-08 | Nkk Corp | プレス成形性に優れた薄鋼板およびその加工方法 |
| JP2004211126A (ja) | 2002-12-27 | 2004-07-29 | Jfe Steel Kk | 超微細粒組織を有し伸びフランジ性に優れる溶融亜鉛めっき冷延鋼板およびその製造方法 |
| JP2004250774A (ja) | 2002-03-29 | 2004-09-09 | Jfe Steel Kk | 超微細粒組織を有する冷延鋼板およびその製造方法 |
| JP2007070649A (ja) | 2005-09-02 | 2007-03-22 | Nippon Steel Corp | 耐食性と穴拡げ性に優れた溶融亜鉛めっき高強度鋼板、合金化溶融亜鉛めっき高強度鋼板、およびそれらの製造方法 |
| JP2007070659A (ja) | 2005-09-05 | 2007-03-22 | Nippon Steel Corp | 耐食性と伸びと穴拡げ性に優れた溶融亜鉛めっき鋼板および合金化溶融亜鉛めっき鋼板、およびそれらの製造方法 |
| JP2007197763A (ja) * | 2006-01-25 | 2007-08-09 | Kobe Steel Ltd | 耐食性および靭性に優れた船舶用鋼材 |
| JP2008274379A (ja) * | 2007-05-02 | 2008-11-13 | Kobe Steel Ltd | 耐ピット性に優れた鋼板およびその製造方法 |
Family Cites Families (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP3292671B2 (ja) * | 1997-02-10 | 2002-06-17 | 川崎製鉄株式会社 | 深絞り性と耐時効性の良好な冷延鋼板用の熱延鋼帯 |
| CA2231760A1 (en) * | 1998-03-11 | 1999-09-11 | Nisshin Steel Co., Ltd. | Cold-rolled steel strip and hot-dip coated cold-rolled steel strip for use as building material and manufacturing method thereof |
| JP4267367B2 (ja) * | 2002-06-19 | 2009-05-27 | 新日本製鐵株式会社 | 原油油槽用鋼およびその製造方法、原油油槽およびその防食方法 |
-
2009
- 2009-11-17 CN CN200980154581.1A patent/CN102282280B/zh not_active Expired - Fee Related
- 2009-11-17 KR KR1020117013957A patent/KR101304009B1/ko active IP Right Grant
- 2009-11-17 EP EP09827543.1A patent/EP2371978B1/en not_active Not-in-force
- 2009-11-17 PL PL09827543T patent/PL2371978T3/pl unknown
- 2009-11-17 ES ES09827543.1T patent/ES2672070T3/es active Active
- 2009-11-17 WO PCT/JP2009/069464 patent/WO2010058762A1/ja active Application Filing
Patent Citations (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS6213533A (ja) | 1985-07-09 | 1987-01-22 | Nippon Steel Corp | 曲げ特性の優れた高強度薄鋼板の製造方法 |
| JPH04191322A (ja) | 1990-11-26 | 1992-07-09 | Nkk Corp | 鋼材の均質化処理方法 |
| JP2002161336A (ja) | 2000-09-12 | 2002-06-04 | Nkk Corp | 超高張力冷延鋼板およびその製造方法 |
| JP2002322539A (ja) | 2001-01-31 | 2002-11-08 | Nkk Corp | プレス成形性に優れた薄鋼板およびその加工方法 |
| JP2004250774A (ja) | 2002-03-29 | 2004-09-09 | Jfe Steel Kk | 超微細粒組織を有する冷延鋼板およびその製造方法 |
| JP2004211126A (ja) | 2002-12-27 | 2004-07-29 | Jfe Steel Kk | 超微細粒組織を有し伸びフランジ性に優れる溶融亜鉛めっき冷延鋼板およびその製造方法 |
| JP2007070649A (ja) | 2005-09-02 | 2007-03-22 | Nippon Steel Corp | 耐食性と穴拡げ性に優れた溶融亜鉛めっき高強度鋼板、合金化溶融亜鉛めっき高強度鋼板、およびそれらの製造方法 |
| JP2007070659A (ja) | 2005-09-05 | 2007-03-22 | Nippon Steel Corp | 耐食性と伸びと穴拡げ性に優れた溶融亜鉛めっき鋼板および合金化溶融亜鉛めっき鋼板、およびそれらの製造方法 |
| JP2007197763A (ja) * | 2006-01-25 | 2007-08-09 | Kobe Steel Ltd | 耐食性および靭性に優れた船舶用鋼材 |
| JP2008274379A (ja) * | 2007-05-02 | 2008-11-13 | Kobe Steel Ltd | 耐ピット性に優れた鋼板およびその製造方法 |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2011231367A (ja) * | 2010-04-27 | 2011-11-17 | Sumitomo Metal Ind Ltd | 溶融亜鉛めっき鋼板およびその製造方法 |
| JP2012125824A (ja) * | 2010-12-17 | 2012-07-05 | Sumitomo Metal Ind Ltd | 高強度鋼板用の連続鋳造鋳片およびその連続鋳造方法、ならびに高強度鋼板 |
Also Published As
| Publication number | Publication date |
|---|---|
| EP2371978B1 (en) | 2018-05-02 |
| CN102282280A (zh) | 2011-12-14 |
| ES2672070T3 (es) | 2018-06-12 |
| KR101304009B1 (ko) | 2013-09-04 |
| EP2371978A1 (en) | 2011-10-05 |
| CN102282280B (zh) | 2015-01-07 |
| PL2371978T3 (pl) | 2018-09-28 |
| KR20110084545A (ko) | 2011-07-25 |
| EP2371978A4 (en) | 2016-05-04 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2010058762A1 (ja) | 鋼板および表面処理鋼板ならびにそれらの製造方法 | |
| JP6409917B2 (ja) | 熱延鋼板の製造方法および冷延フルハード鋼板の製造方法 | |
| JP4725415B2 (ja) | 熱間プレス用鋼板および熱間プレス鋼板部材ならびにそれらの製造方法 | |
| JP5636727B2 (ja) | 溶融亜鉛めっき鋼板およびその製造方法 | |
| CN112930413A (zh) | 高强度钢板及其制造方法 | |
| WO2013047810A1 (ja) | 合金化溶融亜鉛めっき鋼板 | |
| JP2004211157A (ja) | 高強度高延性溶融亜鉛めっき鋼板とその製造方法 | |
| WO2017131054A1 (ja) | 高強度亜鉛めっき鋼板、高強度部材及び高強度亜鉛めっき鋼板の製造方法 | |
| CN112955575B (zh) | 高强度构件、高强度构件的制造方法和高强度构件用钢板的制造方法 | |
| JP6384623B2 (ja) | 高強度鋼板およびその製造方法 | |
| JP7216933B2 (ja) | 鋼板およびその製造方法 | |
| EP3875616A1 (en) | Steel sheet, member, and manufacturing method of these | |
| JP5206350B2 (ja) | 溶融亜鉛めっき鋼板およびその製造方法 | |
| JP6409916B2 (ja) | 熱延鋼板の製造方法および冷延フルハード鋼板の製造方法 | |
| US11408047B2 (en) | Alloyed hot-dip galvanized steel sheet and alloyed hot-dip galvanized steel sheet production method | |
| JP5245475B2 (ja) | 鋼板およびその製造方法 | |
| JP5206349B2 (ja) | 鋼板および表面処理鋼板ならびにそれらの製造方法 | |
| JP4360319B2 (ja) | 高張力溶融亜鉛めっき鋼板とその製造方法 | |
| CN115768915A (zh) | 镀锌钢板、构件和它们的制造方法 | |
| JP5206352B2 (ja) | 鋼板およびその製造方法 | |
| JP3613129B2 (ja) | 成形性にすぐれた合金化溶融亜鉛めっき高張力鋼板及びその製造方法 | |
| JP5206351B2 (ja) | 鋼板およびその製造方法 | |
| JP5387501B2 (ja) | 鋼板および表面処理鋼板ならびにそれらの製造方法 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 200980154581.1 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 09827543 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 3958/DELNP/2011 Country of ref document: IN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2009827543 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 20117013957 Country of ref document: KR Kind code of ref document: A |