WO2004091663A1 - 神経再生薬 - Google Patents
神経再生薬 Download PDFInfo
- Publication number
- WO2004091663A1 WO2004091663A1 PCT/JP2004/005503 JP2004005503W WO2004091663A1 WO 2004091663 A1 WO2004091663 A1 WO 2004091663A1 JP 2004005503 W JP2004005503 W JP 2004005503W WO 2004091663 A1 WO2004091663 A1 WO 2004091663A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- dihydro
- benzazepine
- group
- indolo
- substituted
- Prior art date
Links
- 239000003814 drug Substances 0.000 title claims abstract description 54
- 210000005036 nerve Anatomy 0.000 title claims abstract description 38
- 229940079593 drug Drugs 0.000 title claims abstract description 22
- 238000011069 regeneration method Methods 0.000 title claims description 12
- 230000008929 regeneration Effects 0.000 title claims description 10
- 108010014905 Glycogen Synthase Kinase 3 Proteins 0.000 claims abstract description 129
- 239000000126 substance Substances 0.000 claims abstract description 106
- 230000004766 neurogenesis Effects 0.000 claims abstract description 89
- 230000000694 effects Effects 0.000 claims abstract description 80
- 210000002569 neuron Anatomy 0.000 claims abstract description 59
- 230000002401 inhibitory effect Effects 0.000 claims abstract description 22
- 239000004480 active ingredient Substances 0.000 claims abstract description 16
- 208000024827 Alzheimer disease Diseases 0.000 claims abstract description 15
- 238000012258 culturing Methods 0.000 claims abstract description 12
- 201000010099 disease Diseases 0.000 claims abstract description 10
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 claims abstract description 10
- 208000018737 Parkinson disease Diseases 0.000 claims abstract description 9
- 210000000130 stem cell Anatomy 0.000 claims abstract description 9
- 208000028683 bipolar I disease Diseases 0.000 claims abstract description 7
- 206010002026 amyotrophic lateral sclerosis Diseases 0.000 claims abstract description 6
- 208000026106 cerebrovascular disease Diseases 0.000 claims abstract description 6
- 206010015037 epilepsy Diseases 0.000 claims abstract description 6
- 208000019901 Anxiety disease Diseases 0.000 claims abstract description 5
- 208000023105 Huntington disease Diseases 0.000 claims abstract description 5
- 208000006011 Stroke Diseases 0.000 claims abstract description 5
- 201000006417 multiple sclerosis Diseases 0.000 claims abstract description 5
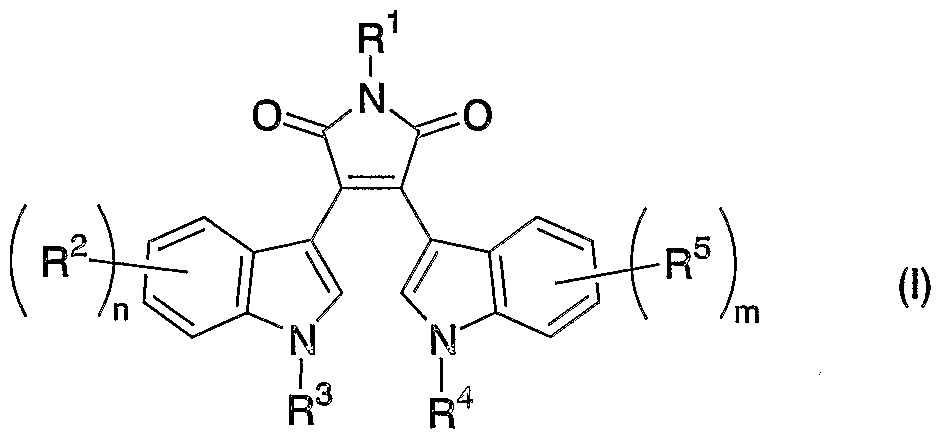
- -1 hydroxy, nitro, amino Chemical group 0.000 claims description 130
- 102000002254 Glycogen Synthase Kinase 3 Human genes 0.000 claims description 128
- 125000000217 alkyl group Chemical group 0.000 claims description 100
- 239000003795 chemical substances by application Substances 0.000 claims description 73
- 150000003839 salts Chemical class 0.000 claims description 73
- 125000004435 hydrogen atom Chemical group [H]* 0.000 claims description 62
- 150000001875 compounds Chemical class 0.000 claims description 60
- 125000005842 heteroatom Chemical group 0.000 claims description 59
- 210000001178 neural stem cell Anatomy 0.000 claims description 59
- 230000001737 promoting effect Effects 0.000 claims description 57
- 125000003118 aryl group Chemical group 0.000 claims description 52
- 125000003545 alkoxy group Chemical group 0.000 claims description 41
- 229910052736 halogen Inorganic materials 0.000 claims description 37
- 150000002367 halogens Chemical class 0.000 claims description 35
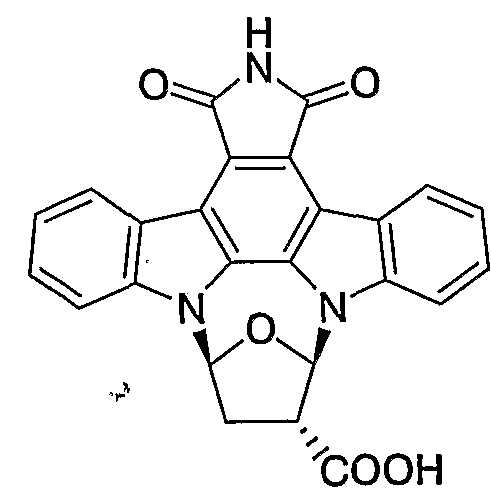
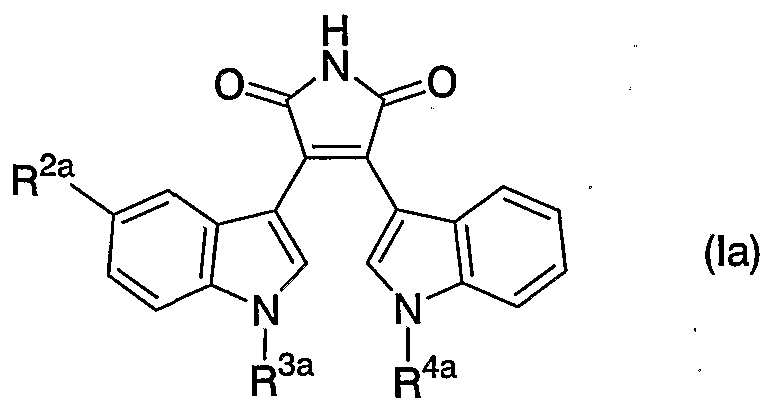
- CRDNMYFJWFXOCH-YPKPFQOOSA-N (3z)-3-(3-oxo-1h-indol-2-ylidene)-1h-indol-2-one Chemical class N/1C2=CC=CC=C2C(=O)C\1=C1/C2=CC=CC=C2NC1=O CRDNMYFJWFXOCH-YPKPFQOOSA-N 0.000 claims description 31
- 125000004432 carbon atom Chemical group C* 0.000 claims description 27
- 125000000753 cycloalkyl group Chemical group 0.000 claims description 27
- WHXSMMKQMYFTQS-UHFFFAOYSA-N Lithium Chemical compound [Li] WHXSMMKQMYFTQS-UHFFFAOYSA-N 0.000 claims description 26
- 125000003342 alkenyl group Chemical group 0.000 claims description 26
- 229910052744 lithium Inorganic materials 0.000 claims description 26
- 230000001172 regenerating effect Effects 0.000 claims description 24
- 125000003277 amino group Chemical group 0.000 claims description 23
- 125000004356 hydroxy functional group Chemical group O* 0.000 claims description 22
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 claims description 20
- QOXOZONBQWIKDA-UHFFFAOYSA-N 3-hydroxypropyl Chemical group [CH2]CCO QOXOZONBQWIKDA-UHFFFAOYSA-N 0.000 claims description 19
- 208000012902 Nervous system disease Diseases 0.000 claims description 17
- 208000025966 Neurological disease Diseases 0.000 claims description 17
- 125000002887 hydroxy group Chemical group [H]O* 0.000 claims description 17
- QQUXFYAWXPMDOE-UHFFFAOYSA-N kenpaullone Chemical compound C1C(=O)NC2=CC=CC=C2C2=C1C1=CC(Br)=CC=C1N2 QQUXFYAWXPMDOE-UHFFFAOYSA-N 0.000 claims description 16
- WGYKZJWCGVVSQN-UHFFFAOYSA-N propylamine Chemical group CCCN WGYKZJWCGVVSQN-UHFFFAOYSA-N 0.000 claims description 16
- 125000001273 sulfonato group Chemical group [O-]S(*)(=O)=O 0.000 claims description 16
- 229910052739 hydrogen Inorganic materials 0.000 claims description 15
- 239000001257 hydrogen Substances 0.000 claims description 15
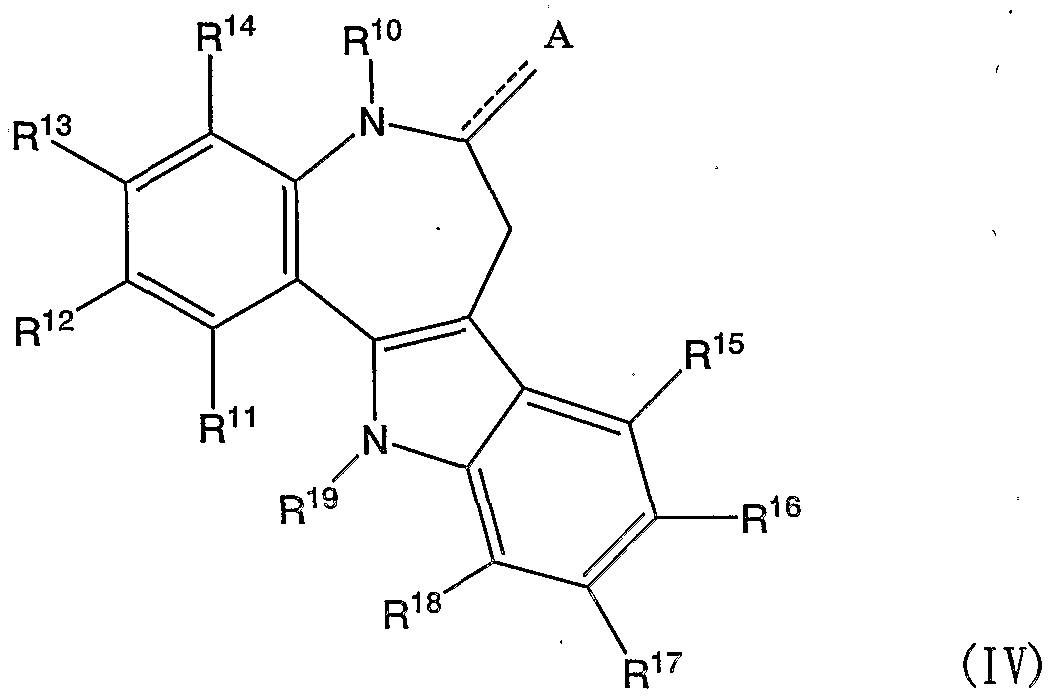
- OLRJMJBCFIJRIT-UHFFFAOYSA-N 4H-azepino[4,5-b]indol-5-one Chemical compound O=C1CN=CC=C2C1=Nc1ccccc21 OLRJMJBCFIJRIT-UHFFFAOYSA-N 0.000 claims description 14
- CRDNMYFJWFXOCH-BUHFOSPRSA-N Couroupitine B Natural products N\1C2=CC=CC=C2C(=O)C/1=C1/C2=CC=CC=C2NC1=O CRDNMYFJWFXOCH-BUHFOSPRSA-N 0.000 claims description 14
- CRDNMYFJWFXOCH-UHFFFAOYSA-N isoindigotin Natural products N1C2=CC=CC=C2C(=O)C1=C1C2=CC=CC=C2NC1=O CRDNMYFJWFXOCH-UHFFFAOYSA-N 0.000 claims description 14
- 125000000449 nitro group Chemical group [O-][N+](*)=O 0.000 claims description 14
- DQFQCHIDRBIESA-UHFFFAOYSA-N 1-benzazepine Chemical compound N1C=CC=CC2=CC=CC=C12 DQFQCHIDRBIESA-UHFFFAOYSA-N 0.000 claims description 13
- 125000003710 aryl alkyl group Chemical group 0.000 claims description 13
- 125000002252 acyl group Chemical group 0.000 claims description 12
- 125000004453 alkoxycarbonyl group Chemical group 0.000 claims description 12
- 238000006243 chemical reaction Methods 0.000 claims description 12
- PEEHTFAAVSWFBL-UHFFFAOYSA-N Maleimide Chemical compound O=C1NC(=O)C=C1 PEEHTFAAVSWFBL-UHFFFAOYSA-N 0.000 claims description 11
- 125000004104 aryloxy group Chemical group 0.000 claims description 11
- 238000004519 manufacturing process Methods 0.000 claims description 11
- AZRRZGIBBLWSSQ-UHFFFAOYSA-N 4-ethyl-7-phenyl-3,5-diazabicyclo[2.2.2]octane-2,6-dione Chemical compound N1C(=O)C2C(=O)NC1(CC)CC2C1=CC=CC=C1 AZRRZGIBBLWSSQ-UHFFFAOYSA-N 0.000 claims description 10
- 125000005233 alkylalcohol group Chemical group 0.000 claims description 10
- 229930182470 glycoside Natural products 0.000 claims description 10
- 150000002338 glycosides Chemical class 0.000 claims description 10
- 230000001537 neural effect Effects 0.000 claims description 10
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 claims description 9
- SIKJAQJRHWYJAI-UHFFFAOYSA-N Indole Chemical compound C1=CC=C2NC=CC2=C1 SIKJAQJRHWYJAI-UHFFFAOYSA-N 0.000 claims description 9
- BAPJBEWLBFYGME-UHFFFAOYSA-N Methyl acrylate Chemical compound COC(=O)C=C BAPJBEWLBFYGME-UHFFFAOYSA-N 0.000 claims description 9
- 125000003282 alkyl amino group Chemical group 0.000 claims description 9
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 claims description 8
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 claims description 8
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 claims description 8
- FQCPPVRJPILDIK-UHFFFAOYSA-N chembl126077 Chemical compound N1C2=CC=CC=C2C(N=O)=C1C1=C(O)NC2=CC=CC=C21 FQCPPVRJPILDIK-UHFFFAOYSA-N 0.000 claims description 8
- 150000002016 disaccharides Chemical class 0.000 claims description 8
- 150000002772 monosaccharides Chemical class 0.000 claims description 8
- 125000004433 nitrogen atom Chemical group N* 0.000 claims description 8
- 229910052760 oxygen Inorganic materials 0.000 claims description 8
- 239000001301 oxygen Substances 0.000 claims description 8
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims description 8
- LFGREXWGYUGZLY-UHFFFAOYSA-N phosphoryl Chemical group [P]=O LFGREXWGYUGZLY-UHFFFAOYSA-N 0.000 claims description 8
- 229910052717 sulfur Inorganic materials 0.000 claims description 8
- 150000003924 bisindolylmaleimides Chemical class 0.000 claims description 7
- 150000001732 carboxylic acid derivatives Chemical class 0.000 claims description 7
- 101150090422 gsk-3 gene Proteins 0.000 claims description 7
- 229910052757 nitrogen Inorganic materials 0.000 claims description 7
- VGMDAWVZNAXVDG-UHFFFAOYSA-N paullone Chemical compound C12=CC=CC=C2NC(=O)CC2=C1NC1=CC=CC=C21 VGMDAWVZNAXVDG-UHFFFAOYSA-N 0.000 claims description 7
- KIZLESRPUNYYNU-UHFFFAOYSA-N 2,9-dibromo-7,12-dihydro-5h-indolo[3,2-d][1]benzazepin-6-one Chemical compound C12=CC(Br)=CC=C2NC(=O)CC2=C1NC1=CC=C(Br)C=C21 KIZLESRPUNYYNU-UHFFFAOYSA-N 0.000 claims description 6
- LBVIZFWUIQSZHP-UHFFFAOYSA-N 6-oxo-7,12-dihydro-5h-indolo[3,2-d][1]benzazepine-9-carbonitrile Chemical compound C12=CC=CC=C2NC(=O)CC2=C1NC1=CC=C(C#N)C=C21 LBVIZFWUIQSZHP-UHFFFAOYSA-N 0.000 claims description 6
- 208000020925 Bipolar disease Diseases 0.000 claims description 6
- 229930182474 N-glycoside Natural products 0.000 claims description 6
- 125000005843 halogen group Chemical group 0.000 claims description 6
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 claims description 6
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 claims description 6
- 238000006467 substitution reaction Methods 0.000 claims description 6
- 125000002023 trifluoromethyl group Chemical group FC(F)(F)* 0.000 claims description 6
- BLRHMMGNCXNXJL-UHFFFAOYSA-N 1-methylindole Chemical compound C1=CC=C2N(C)C=CC2=C1 BLRHMMGNCXNXJL-UHFFFAOYSA-N 0.000 claims description 5
- 125000004201 2,4-dichlorophenyl group Chemical group [H]C1=C([H])C(*)=C(Cl)C([H])=C1Cl 0.000 claims description 5
- XOAKRYSSJJAKLL-UHFFFAOYSA-N 9-methoxy-7,12-dihydro-5h-indolo[3,2-d][1]benzazepin-6-one Chemical compound C1C(=O)NC2=CC=CC=C2C2=C1C1=CC(OC)=CC=C1N2 XOAKRYSSJJAKLL-UHFFFAOYSA-N 0.000 claims description 5
- 229930182473 O-glycoside Chemical group 0.000 claims description 5
- 150000008444 O-glycosides Chemical group 0.000 claims description 5
- 125000004429 atom Chemical group 0.000 claims description 5
- 125000004093 cyano group Chemical group *C#N 0.000 claims description 5
- HBDSHCUSXQATPO-BGBJRWHRSA-N indirubin-3'-monoxime Chemical compound O=C/1NC2=CC=CC=C2C\1=C\1/C(=N/O)/C2=CC=CC=C2N/1 HBDSHCUSXQATPO-BGBJRWHRSA-N 0.000 claims description 5
- WQGWDDDVZFFDIG-UHFFFAOYSA-N pyrogallol Chemical compound OC1=CC=CC(O)=C1O WQGWDDDVZFFDIG-UHFFFAOYSA-N 0.000 claims description 5
- 208000020431 spinal cord injury Diseases 0.000 claims description 5
- DNIAPMSPPWPWGF-GSVOUGTGSA-N (R)-(-)-Propylene glycol Chemical compound C[C@@H](O)CO DNIAPMSPPWPWGF-GSVOUGTGSA-N 0.000 claims description 4
- YAKJZHVHEIOVIR-UHFFFAOYSA-N 2,3-dimethoxy-7,12-dihydro-5h-indolo[3,2-d][1]benzazepin-6-one Chemical compound C1=2C=C(OC)C(OC)=CC=2NC(=O)CC2=C1NC1=CC=CC=C21 YAKJZHVHEIOVIR-UHFFFAOYSA-N 0.000 claims description 4
- KYRQRRGDKAIFMA-UHFFFAOYSA-N 2,3-dimethoxy-9-nitro-7,12-dihydro-5h-indolo[3,2-d][1]benzazepin-6-one Chemical compound C1=2C=C(OC)C(OC)=CC=2NC(=O)CC2=C1NC1=CC=C([N+]([O-])=O)C=C21 KYRQRRGDKAIFMA-UHFFFAOYSA-N 0.000 claims description 4
- 125000004182 2-chlorophenyl group Chemical group [H]C1=C([H])C(Cl)=C(*)C([H])=C1[H] 0.000 claims description 4
- 125000000954 2-hydroxyethyl group Chemical group [H]C([*])([H])C([H])([H])O[H] 0.000 claims description 4
- SYGNSFGBDJEBOK-UHFFFAOYSA-N 8,10-dichloro-7,12-dihydro-5h-indolo[3,2-d][1]benzazepin-6-one Chemical compound C1C(=O)NC2=CC=CC=C2C2=C1C1=C(Cl)C=C(Cl)C=C1N2 SYGNSFGBDJEBOK-UHFFFAOYSA-N 0.000 claims description 4
- NLHHRLWOUZZQLW-UHFFFAOYSA-N Acrylonitrile Chemical compound C=CC#N NLHHRLWOUZZQLW-UHFFFAOYSA-N 0.000 claims description 4
- 201000010374 Down Syndrome Diseases 0.000 claims description 4
- BUGBHKTXTAQXES-UHFFFAOYSA-N Selenium Chemical compound [Se] BUGBHKTXTAQXES-UHFFFAOYSA-N 0.000 claims description 4
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 claims description 4
- 125000005907 alkyl ester group Chemical group 0.000 claims description 4
- 150000001408 amides Chemical class 0.000 claims description 4
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 claims description 4
- AOHZJKSTSDMBIZ-UHFFFAOYSA-N chembl111433 Chemical compound C1=CC=C2C(=O)C(C3=C(O)NC4=CC=C(C=C43)C)=NC2=C1 AOHZJKSTSDMBIZ-UHFFFAOYSA-N 0.000 claims description 4
- 125000001841 imino group Chemical group [H]N=* 0.000 claims description 4
- PZOUSPYUWWUPPK-UHFFFAOYSA-N indole Natural products CC1=CC=CC2=C1C=CN2 PZOUSPYUWWUPPK-UHFFFAOYSA-N 0.000 claims description 4
- RKJUIXBNRJVNHR-UHFFFAOYSA-N indolenine Natural products C1=CC=C2CC=NC2=C1 RKJUIXBNRJVNHR-UHFFFAOYSA-N 0.000 claims description 4
- 150000002576 ketones Chemical class 0.000 claims description 4
- 125000000018 nitroso group Chemical group N(=O)* 0.000 claims description 4
- 125000004430 oxygen atom Chemical group O* 0.000 claims description 4
- 229910052711 selenium Inorganic materials 0.000 claims description 4
- 239000011669 selenium Substances 0.000 claims description 4
- 239000011593 sulfur Substances 0.000 claims description 4
- 125000004434 sulfur atom Chemical group 0.000 claims description 4
- 229910052714 tellurium Inorganic materials 0.000 claims description 4
- PORWMNRCUJJQNO-UHFFFAOYSA-N tellurium atom Chemical group [Te] PORWMNRCUJJQNO-UHFFFAOYSA-N 0.000 claims description 4
- 229940124597 therapeutic agent Drugs 0.000 claims description 4
- 125000004665 trialkylsilyl group Chemical group 0.000 claims description 4
- 125000005106 triarylsilyl group Chemical group 0.000 claims description 4
- SFMHJRSZXNCRAM-UHFFFAOYSA-N 1-(1-methylindol-3-yl)pyrrole-2,5-dione Chemical compound C12=CC=CC=C2N(C)C=C1N1C(=O)C=CC1=O SFMHJRSZXNCRAM-UHFFFAOYSA-N 0.000 claims description 3
- TUMYZJPVQGGVEN-UHFFFAOYSA-N 2,3-dimethoxy-6-oxo-7,12-dihydro-5h-indolo[3,2-d][1]benzazepine-9-carbonitrile Chemical compound C1=2C=C(OC)C(OC)=CC=2NC(=O)CC2=C1NC1=CC=C(C#N)C=C21 TUMYZJPVQGGVEN-UHFFFAOYSA-N 0.000 claims description 3
- HLNLXECKTICCDT-UHFFFAOYSA-N 4-hydroxy-7,12-dihydro-5h-indolo[3,2-d][1]benzazepin-6-one Chemical compound C1=2NC3=CC=CC=C3C=2CC(=O)NC2=C1C=CC=C2O HLNLXECKTICCDT-UHFFFAOYSA-N 0.000 claims description 3
- YBTFSTRMFPNVDQ-UHFFFAOYSA-N 9-bromo-10-hydroxy-7,12-dihydro-5h-indolo[3,2-d][1]benzazepin-6-one Chemical compound C1C(=O)NC2=CC=CC=C2C(N2)=C1C1=C2C=C(O)C(Br)=C1 YBTFSTRMFPNVDQ-UHFFFAOYSA-N 0.000 claims description 3
- FDMHWATUIRUWOF-UHFFFAOYSA-N 9-methyl-7,12-dihydro-5h-indolo[3,2-d][1]benzazepin-6-one Chemical compound C1C(=O)NC2=CC=CC=C2C2=C1C1=CC(C)=CC=C1N2 FDMHWATUIRUWOF-UHFFFAOYSA-N 0.000 claims description 3
- 239000004593 Epoxy Substances 0.000 claims description 3
- 125000001931 aliphatic group Chemical group 0.000 claims description 3
- HSFWRNGVRCDJHI-UHFFFAOYSA-N alpha-acetylene Natural products C#C HSFWRNGVRCDJHI-UHFFFAOYSA-N 0.000 claims description 3
- 125000000539 amino acid group Chemical group 0.000 claims description 3
- 125000003917 carbamoyl group Chemical group [H]N([H])C(*)=O 0.000 claims description 3
- XMPHNZMDLSUDQI-UHFFFAOYSA-N chembl173682 Chemical compound C1=CC=C2C(=O)C(C=3C4=CC(Br)=CC=C4NC=3O)=NC2=C1 XMPHNZMDLSUDQI-UHFFFAOYSA-N 0.000 claims description 3
- 125000002534 ethynyl group Chemical group [H]C#C* 0.000 claims description 3
- 125000001188 haloalkyl group Chemical group 0.000 claims description 3
- 230000009689 neuronal regeneration Effects 0.000 claims description 3
- FVSKHRXBFJPNKK-UHFFFAOYSA-N propionitrile Chemical compound CCC#N FVSKHRXBFJPNKK-UHFFFAOYSA-N 0.000 claims description 3
- QAOWNCQODCNURD-UHFFFAOYSA-L sulfate group Chemical group S(=O)(=O)([O-])[O-] QAOWNCQODCNURD-UHFFFAOYSA-L 0.000 claims description 3
- 125000000565 sulfonamide group Chemical group 0.000 claims description 3
- ITMCEJHCFYSIIV-UHFFFAOYSA-M triflate Chemical compound [O-]S(=O)(=O)C(F)(F)F ITMCEJHCFYSIIV-UHFFFAOYSA-M 0.000 claims description 3
- JGELUIOSDAYQLO-UHFFFAOYSA-N 9-bromo-11-(hydroxymethyl)-7,12-dihydro-5h-indolo[3,2-d][1]benzazepin-6-one Chemical compound C1C(=O)NC2=CC=CC=C2C2=C1C(C=C(Br)C=C1CO)=C1N2 JGELUIOSDAYQLO-UHFFFAOYSA-N 0.000 claims description 2
- CSJPCQXZZVBCHG-UHFFFAOYSA-N 9-bromo-2,3-dihydroxy-7,12-dihydro-5h-indolo[3,2-d][1]benzazepin-6-one Chemical compound C1=2C=C(O)C(O)=CC=2NC(=O)CC2=C1NC1=CC=C(Br)C=C21 CSJPCQXZZVBCHG-UHFFFAOYSA-N 0.000 claims description 2
- IZRNPQQLTKFPRZ-UHFFFAOYSA-N 9-bromo-4-methoxy-7,12-dihydro-5h-indolo[3,2-d][1]benzazepin-6-one Chemical compound C1=2NC3=CC=C(Br)C=C3C=2CC(=O)NC2=C1C=CC=C2OC IZRNPQQLTKFPRZ-UHFFFAOYSA-N 0.000 claims description 2
- MHKWLZUWJABEIX-UHFFFAOYSA-N 9-bromo-7,12-dihydro-5h-indolo[3,2-d][1]benzazepine-6-thione Chemical compound C1C(=S)NC2=CC=CC=C2C2=C1C1=CC(Br)=CC=C1N2 MHKWLZUWJABEIX-UHFFFAOYSA-N 0.000 claims description 2
- OKMGGPIYQLLQFZ-UHFFFAOYSA-N 9-chloro-7,12-dihydro-5h-indolo[3,2-d][1]benzazepin-6-one Chemical compound C1C(=O)NC2=CC=CC=C2C2=C1C1=CC(Cl)=CC=C1N2 OKMGGPIYQLLQFZ-UHFFFAOYSA-N 0.000 claims description 2
- 125000004051 hexyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 claims description 2
- JYXPWUYLZPYOAH-UHFFFAOYSA-N methylhydrazine Chemical compound CN=N JYXPWUYLZPYOAH-UHFFFAOYSA-N 0.000 claims description 2
- 125000002467 phosphate group Chemical group [H]OP(=O)(O[H])O[*] 0.000 claims description 2
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 claims description 2
- ABLZXFCXXLZCGV-UHFFFAOYSA-N Phosphorous acid Chemical group OP(O)=O ABLZXFCXXLZCGV-UHFFFAOYSA-N 0.000 claims 3
- ISVJYSRTVAYNMI-UHFFFAOYSA-N 1,4-dihydroindol-5-one Chemical compound C1=CC(=O)CC2=C1NC=C2 ISVJYSRTVAYNMI-UHFFFAOYSA-N 0.000 claims 2
- UNIYXBRZYVYAAJ-UHFFFAOYSA-N 1-methyl-7,12-dihydro-5h-indolo[3,2-d][1]benzazepin-6-one Chemical compound C1=2C(C)=CC=CC=2NC(=O)CC2=C1NC1=CC=CC=C21 UNIYXBRZYVYAAJ-UHFFFAOYSA-N 0.000 claims 2
- DUZUWJBAONJCTE-UHFFFAOYSA-N 9-bromo-2,3-dimethoxy-7,12-dihydro-5h-indolo[3,2-d][1]benzazepin-6-one Chemical compound C1=2C=C(OC)C(OC)=CC=2NC(=O)CC2=C1NC1=CC=C(Br)C=C21 DUZUWJBAONJCTE-UHFFFAOYSA-N 0.000 claims 2
- LOOYKDRIOSVHMM-UHFFFAOYSA-N 11-ethyl-7,12-dihydro-5h-indolo[3,2-d][1]benzazepin-6-one Chemical compound C1C(=O)NC2=CC=CC=C2C2=C1C(C=CC=C1CC)=C1N2 LOOYKDRIOSVHMM-UHFFFAOYSA-N 0.000 claims 1
- XWKMAJPZELTMLH-UHFFFAOYSA-N 11-methyl-7,12-dihydro-5h-indolo[3,2-d][1]benzazepin-6-one Chemical compound C1C(=O)NC2=CC=CC=C2C2=C1C(C=CC=C1C)=C1N2 XWKMAJPZELTMLH-UHFFFAOYSA-N 0.000 claims 1
- FZSXLUDKOYPBMM-UHFFFAOYSA-N 2,3-dihydroxy-7,12-dihydro-5h-indolo[3,2-d][1]benzazepin-6-one Chemical compound C1=2C=C(O)C(O)=CC=2NC(=O)CC2=C1NC1=CC=CC=C21 FZSXLUDKOYPBMM-UHFFFAOYSA-N 0.000 claims 1
- CZOVMHCLKVCOEM-UHFFFAOYSA-N 2-bromo-9-(trifluoromethyl)-7,12-dihydro-5h-indolo[3,2-d][1]benzazepin-6-one Chemical compound C1C(=O)NC2=CC=C(Br)C=C2C2=C1C1=CC(C(F)(F)F)=CC=C1N2 CZOVMHCLKVCOEM-UHFFFAOYSA-N 0.000 claims 1
- OMZWEMXAKFVJGS-UHFFFAOYSA-N 3-(2-chlorophenyl)-4-(1-methylindol-3-yl)pyrrole-2,5-dione Chemical compound C12=CC=CC=C2N(C)C=C1C(C(NC1=O)=O)=C1C1=CC=CC=C1Cl OMZWEMXAKFVJGS-UHFFFAOYSA-N 0.000 claims 1
- JCAZXVOHALWELW-UHFFFAOYSA-N 4,5-dihydroazepin-3-one Chemical compound O=C1CCC=CN=C1 JCAZXVOHALWELW-UHFFFAOYSA-N 0.000 claims 1
- ATOAEKSFKWKAIO-UHFFFAOYSA-N 4-methoxy-7,12-dihydro-5h-indolo[3,2-d][1]benzazepin-6-one Chemical compound C1=2NC3=CC=CC=C3C=2CC(=O)NC2=C1C=CC=C2OC ATOAEKSFKWKAIO-UHFFFAOYSA-N 0.000 claims 1
- GWFCNMFOAORLDH-UHFFFAOYSA-N 9-bromo-12-(2-hydroxyethyl)-5,7-dihydroindolo[3,2-d][1]benzazepin-6-one Chemical compound C1C(=O)NC2=CC=CC=C2C2=C1C1=CC(Br)=CC=C1N2CCO GWFCNMFOAORLDH-UHFFFAOYSA-N 0.000 claims 1
- VPNVMVMFLHEGBV-UHFFFAOYSA-N 9-bromo-4-hydroxy-7,12-dihydro-5h-indolo[3,2-d][1]benzazepin-6-one Chemical compound C1=2NC3=CC=C(Br)C=C3C=2CC(=O)NC2=C1C=CC=C2O VPNVMVMFLHEGBV-UHFFFAOYSA-N 0.000 claims 1
- NNDLCXNIMGREEF-UHFFFAOYSA-N 9-fluoro-7,12-dihydro-5h-indolo[3,2-d][1]benzazepin-6-one Chemical compound C1C(=O)NC2=CC=CC=C2C2=C1C1=CC(F)=CC=C1N2 NNDLCXNIMGREEF-UHFFFAOYSA-N 0.000 claims 1
- 150000001298 alcohols Chemical class 0.000 claims 1
- 125000000033 alkoxyamino group Chemical group 0.000 claims 1
- OLUKILHGKRVDCT-UHFFFAOYSA-N alsterpaullone Chemical compound C1C(=O)NC2=CC=CC=C2C2=C1C1=CC([N+](=O)[O-])=CC=C1N2 OLUKILHGKRVDCT-UHFFFAOYSA-N 0.000 claims 1
- ZMKPMNLSLLYATH-UHFFFAOYSA-N azepino[4,5-b]indole Chemical compound C1=CN=CC=C2C3=CC=CC=C3N=C21 ZMKPMNLSLLYATH-UHFFFAOYSA-N 0.000 claims 1
- 125000004744 butyloxycarbonyl group Chemical group 0.000 claims 1
- 150000001735 carboxylic acids Chemical class 0.000 claims 1
- 239000003610 charcoal Substances 0.000 claims 1
- KZGUEWREZGMORL-UHFFFAOYSA-N ditert-butyl 9-bromo-6-oxo-7,12-dihydroindolo[3,2-d][1]benzazepine-5,7-dicarboxylate Chemical compound C12=CC=CC=C2N(C(=O)OC(C)(C)C)C(=O)C(C(=O)OC(C)(C)C)C2=C1NC1=CC=C(Br)C=C21 KZGUEWREZGMORL-UHFFFAOYSA-N 0.000 claims 1
- SNWQUNCRDLUDEX-UHFFFAOYSA-N inden-1-one Chemical compound C1=CC=C2C(=O)C=CC2=C1 SNWQUNCRDLUDEX-UHFFFAOYSA-N 0.000 claims 1
- 125000001820 oxy group Chemical group [*:1]O[*:2] 0.000 claims 1
- 125000002572 propoxy group Chemical group [*]OC([H])([H])C(C([H])([H])[H])([H])[H] 0.000 claims 1
- 201000000377 schizophrenia 3 Diseases 0.000 claims 1
- BDHFUVZGWQCTTF-UHFFFAOYSA-M sulfonate Chemical compound [O-]S(=O)=O BDHFUVZGWQCTTF-UHFFFAOYSA-M 0.000 claims 1
- 238000000034 method Methods 0.000 abstract description 33
- 206010056677 Nerve degeneration Diseases 0.000 abstract description 4
- 102000001267 GSK3 Human genes 0.000 abstract 1
- 208000020339 Spinal injury Diseases 0.000 abstract 1
- 230000004064 dysfunction Effects 0.000 abstract 1
- 230000010354 integration Effects 0.000 abstract 1
- 208000011580 syndromic disease Diseases 0.000 abstract 1
- 210000004027 cell Anatomy 0.000 description 83
- KWGKDLIKAYFUFQ-UHFFFAOYSA-M lithium chloride Chemical compound [Li+].[Cl-] KWGKDLIKAYFUFQ-UHFFFAOYSA-M 0.000 description 60
- 230000004069 differentiation Effects 0.000 description 34
- 239000002609 medium Substances 0.000 description 34
- 239000000243 solution Substances 0.000 description 28
- 210000004556 brain Anatomy 0.000 description 26
- 230000006698 induction Effects 0.000 description 23
- NOESYZHRGYRDHS-UHFFFAOYSA-N insulin Chemical compound N1C(=O)C(NC(=O)C(CCC(N)=O)NC(=O)C(CCC(O)=O)NC(=O)C(C(C)C)NC(=O)C(NC(=O)CN)C(C)CC)CSSCC(C(NC(CO)C(=O)NC(CC(C)C)C(=O)NC(CC=2C=CC(O)=CC=2)C(=O)NC(CCC(N)=O)C(=O)NC(CC(C)C)C(=O)NC(CCC(O)=O)C(=O)NC(CC(N)=O)C(=O)NC(CC=2C=CC(O)=CC=2)C(=O)NC(CSSCC(NC(=O)C(C(C)C)NC(=O)C(CC(C)C)NC(=O)C(CC=2C=CC(O)=CC=2)NC(=O)C(CC(C)C)NC(=O)C(C)NC(=O)C(CCC(O)=O)NC(=O)C(C(C)C)NC(=O)C(CC(C)C)NC(=O)C(CC=2NC=NC=2)NC(=O)C(CO)NC(=O)CNC2=O)C(=O)NCC(=O)NC(CCC(O)=O)C(=O)NC(CCCNC(N)=N)C(=O)NCC(=O)NC(CC=3C=CC=CC=3)C(=O)NC(CC=3C=CC=CC=3)C(=O)NC(CC=3C=CC(O)=CC=3)C(=O)NC(C(C)O)C(=O)N3C(CCC3)C(=O)NC(CCCCN)C(=O)NC(C)C(O)=O)C(=O)NC(CC(N)=O)C(O)=O)=O)NC(=O)C(C(C)CC)NC(=O)C(CO)NC(=O)C(C(C)O)NC(=O)C1CSSCC2NC(=O)C(CC(C)C)NC(=O)C(NC(=O)C(CCC(N)=O)NC(=O)C(CC(N)=O)NC(=O)C(NC(=O)C(N)CC=1C=CC=CC=1)C(C)C)CC1=CN=CN1 NOESYZHRGYRDHS-UHFFFAOYSA-N 0.000 description 20
- 108020004414 DNA Proteins 0.000 description 19
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 19
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 18
- OHCQJHSOBUTRHG-KGGHGJDLSA-N FORSKOLIN Chemical compound O=C([C@@]12O)C[C@](C)(C=C)O[C@]1(C)[C@@H](OC(=O)C)[C@@H](O)[C@@H]1[C@]2(C)[C@@H](O)CCC1(C)C OHCQJHSOBUTRHG-KGGHGJDLSA-N 0.000 description 16
- 241000700159 Rattus Species 0.000 description 16
- 108090000623 proteins and genes Proteins 0.000 description 15
- 108091032973 (ribonucleotides)n+m Proteins 0.000 description 14
- 239000013642 negative control Substances 0.000 description 14
- 241001465754 Metazoa Species 0.000 description 13
- 239000002299 complementary DNA Substances 0.000 description 13
- 239000000203 mixture Substances 0.000 description 13
- 108090000765 processed proteins & peptides Proteins 0.000 description 13
- 239000013598 vector Substances 0.000 description 13
- 241000282414 Homo sapiens Species 0.000 description 12
- JCSGFHVFHSKIJH-UHFFFAOYSA-N 3-(2,4-dichlorophenyl)-4-(1-methyl-3-indolyl)pyrrole-2,5-dione Chemical compound C12=CC=CC=C2N(C)C=C1C(C(NC1=O)=O)=C1C1=CC=C(Cl)C=C1Cl JCSGFHVFHSKIJH-UHFFFAOYSA-N 0.000 description 11
- 102000004877 Insulin Human genes 0.000 description 10
- 108090001061 Insulin Proteins 0.000 description 10
- 230000001965 increasing effect Effects 0.000 description 10
- 230000001939 inductive effect Effects 0.000 description 10
- 239000003112 inhibitor Substances 0.000 description 10
- 229940125396 insulin Drugs 0.000 description 10
- 239000006144 Dulbecco’s modified Eagle's medium Substances 0.000 description 9
- 108010051975 Glycogen Synthase Kinase 3 beta Proteins 0.000 description 9
- 102000019058 Glycogen Synthase Kinase 3 beta Human genes 0.000 description 9
- ISWSIDIOOBJBQZ-UHFFFAOYSA-N Phenol Chemical compound OC1=CC=CC=C1 ISWSIDIOOBJBQZ-UHFFFAOYSA-N 0.000 description 9
- 239000013078 crystal Substances 0.000 description 9
- 230000002829 reductive effect Effects 0.000 description 9
- SUZLHDUTVMZSEV-UHFFFAOYSA-N Deoxycoleonol Natural products C12C(=O)CC(C)(C=C)OC2(C)C(OC(=O)C)C(O)C2C1(C)C(O)CCC2(C)C SUZLHDUTVMZSEV-UHFFFAOYSA-N 0.000 description 8
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 8
- 230000015572 biosynthetic process Effects 0.000 description 8
- 239000000872 buffer Substances 0.000 description 8
- OHCQJHSOBUTRHG-UHFFFAOYSA-N colforsin Natural products OC12C(=O)CC(C)(C=C)OC1(C)C(OC(=O)C)C(O)C1C2(C)C(O)CCC1(C)C OHCQJHSOBUTRHG-UHFFFAOYSA-N 0.000 description 8
- HQKMJHAJHXVSDF-UHFFFAOYSA-L magnesium stearate Chemical compound [Mg+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O HQKMJHAJHXVSDF-UHFFFAOYSA-L 0.000 description 8
- 125000001424 substituent group Chemical group 0.000 description 8
- 230000001629 suppression Effects 0.000 description 8
- 241000699666 Mus <mouse, genus> Species 0.000 description 7
- 239000002585 base Substances 0.000 description 7
- 230000004770 neurodegeneration Effects 0.000 description 7
- 238000012360 testing method Methods 0.000 description 7
- 102000004219 Brain-derived neurotrophic factor Human genes 0.000 description 6
- 108090000715 Brain-derived neurotrophic factor Proteins 0.000 description 6
- 241000282693 Cercopithecidae Species 0.000 description 6
- 102000003974 Fibroblast growth factor 2 Human genes 0.000 description 6
- 108090000379 Fibroblast growth factor 2 Proteins 0.000 description 6
- 239000012580 N-2 Supplement Substances 0.000 description 6
- 239000000284 extract Substances 0.000 description 6
- 208000015122 neurodegenerative disease Diseases 0.000 description 6
- 239000000825 pharmaceutical preparation Substances 0.000 description 6
- 241001430294 unidentified retrovirus Species 0.000 description 6
- 108010001483 Glycogen Synthase Proteins 0.000 description 5
- 241000700605 Viruses Species 0.000 description 5
- 150000001413 amino acids Chemical group 0.000 description 5
- 230000006907 apoptotic process Effects 0.000 description 5
- 210000001320 hippocampus Anatomy 0.000 description 5
- 238000000338 in vitro Methods 0.000 description 5
- 230000001272 neurogenic effect Effects 0.000 description 5
- 230000004031 neuronal differentiation Effects 0.000 description 5
- 239000013600 plasmid vector Substances 0.000 description 5
- 102000004169 proteins and genes Human genes 0.000 description 5
- 239000012264 purified product Substances 0.000 description 5
- 239000008223 sterile water Substances 0.000 description 5
- 238000003786 synthesis reaction Methods 0.000 description 5
- WOVKYSAHUYNSMH-RRKCRQDMSA-N 5-bromodeoxyuridine Chemical compound C1[C@H](O)[C@@H](CO)O[C@H]1N1C(=O)NC(=O)C(Br)=C1 WOVKYSAHUYNSMH-RRKCRQDMSA-N 0.000 description 4
- GUBGYTABKSRVRQ-XLOQQCSPSA-N Alpha-Lactose Chemical compound O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@H]1O[C@@H]1[C@@H](CO)O[C@H](O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-XLOQQCSPSA-N 0.000 description 4
- 108091003079 Bovine Serum Albumin Proteins 0.000 description 4
- 102220480531 GSK3B-interacting protein_K85R_mutation Human genes 0.000 description 4
- DHMQDGOQFOQNFH-UHFFFAOYSA-N Glycine Chemical compound NCC(O)=O DHMQDGOQFOQNFH-UHFFFAOYSA-N 0.000 description 4
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 description 4
- 108091000080 Phosphotransferase Proteins 0.000 description 4
- 210000004102 animal cell Anatomy 0.000 description 4
- 238000010171 animal model Methods 0.000 description 4
- 230000030833 cell death Effects 0.000 description 4
- 239000012228 culture supernatant Substances 0.000 description 4
- OPTASPLRGRRNAP-UHFFFAOYSA-N cytosine Chemical compound NC=1C=CNC(=O)N=1 OPTASPLRGRRNAP-UHFFFAOYSA-N 0.000 description 4
- 230000006378 damage Effects 0.000 description 4
- 230000001605 fetal effect Effects 0.000 description 4
- 108010049611 glycogen synthase kinase 3 alpha Proteins 0.000 description 4
- 239000001963 growth medium Substances 0.000 description 4
- 230000005764 inhibitory process Effects 0.000 description 4
- 239000008101 lactose Substances 0.000 description 4
- 235000019359 magnesium stearate Nutrition 0.000 description 4
- CTSLXHKWHWQRSH-UHFFFAOYSA-N oxalyl chloride Chemical compound ClC(=O)C(Cl)=O CTSLXHKWHWQRSH-UHFFFAOYSA-N 0.000 description 4
- 102000020233 phosphotransferase Human genes 0.000 description 4
- 238000002360 preparation method Methods 0.000 description 4
- 239000011780 sodium chloride Substances 0.000 description 4
- 125000003107 substituted aryl group Chemical group 0.000 description 4
- 210000001519 tissue Anatomy 0.000 description 4
- IEEQWYMIVLKPFP-UHFFFAOYSA-N 2-bromo-7,12-dihydro-5h-indolo[3,2-d][1]benzazepin-6-one Chemical compound C12=CC(Br)=CC=C2NC(=O)CC2=C1NC1=CC=CC=C21 IEEQWYMIVLKPFP-UHFFFAOYSA-N 0.000 description 3
- 101150090724 3 gene Proteins 0.000 description 3
- 229930024421 Adenine Natural products 0.000 description 3
- GFFGJBXGBJISGV-UHFFFAOYSA-N Adenine Chemical compound NC1=NC=NC2=C1N=CN2 GFFGJBXGBJISGV-UHFFFAOYSA-N 0.000 description 3
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 3
- 241000282412 Homo Species 0.000 description 3
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 3
- 241000124008 Mammalia Species 0.000 description 3
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 3
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 3
- AWKLQZCLFIUTSR-UHFFFAOYSA-N N1C(=O)C=C2C3=CC=CC=C3N=C2C2=CC=CC=C21 Chemical class N1C(=O)C=C2C3=CC=CC=C3N=C2C2=CC=CC=C21 AWKLQZCLFIUTSR-UHFFFAOYSA-N 0.000 description 3
- 239000004372 Polyvinyl alcohol Substances 0.000 description 3
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 3
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 3
- 229930006000 Sucrose Natural products 0.000 description 3
- 206010044688 Trisomy 21 Diseases 0.000 description 3
- 239000002253 acid Substances 0.000 description 3
- 230000009471 action Effects 0.000 description 3
- 229960000643 adenine Drugs 0.000 description 3
- 230000003321 amplification Effects 0.000 description 3
- DZHSAHHDTRWUTF-SIQRNXPUSA-N amyloid-beta polypeptide 42 Chemical compound C([C@@H](C(=O)N[C@@H](C)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@H](C(=O)NCC(=O)N[C@@H](CO)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CCCCN)C(=O)NCC(=O)N[C@@H](C)C(=O)N[C@H](C(=O)N[C@@H]([C@@H](C)CC)C(=O)NCC(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](C(C)C)C(=O)NCC(=O)NCC(=O)N[C@@H](C(C)C)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](C)C(O)=O)[C@@H](C)CC)C(C)C)NC(=O)[C@H](CC=1C=CC=CC=1)NC(=O)[C@@H](NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CCCCN)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@H](CC=1N=CNC=1)NC(=O)[C@H](CC=1N=CNC=1)NC(=O)[C@@H](NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)CNC(=O)[C@H](CO)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CC=1N=CNC=1)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CC=1C=CC=CC=1)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](C)NC(=O)[C@@H](N)CC(O)=O)C(C)C)C(C)C)C1=CC=CC=C1 DZHSAHHDTRWUTF-SIQRNXPUSA-N 0.000 description 3
- 239000012472 biological sample Substances 0.000 description 3
- 230000008859 change Effects 0.000 description 3
- 238000001816 cooling Methods 0.000 description 3
- 239000000287 crude extract Substances 0.000 description 3
- 238000001514 detection method Methods 0.000 description 3
- 238000001914 filtration Methods 0.000 description 3
- 239000000796 flavoring agent Substances 0.000 description 3
- 235000019634 flavors Nutrition 0.000 description 3
- 239000012634 fragment Substances 0.000 description 3
- 150000002431 hydrogen Chemical class 0.000 description 3
- 238000002347 injection Methods 0.000 description 3
- 239000007924 injection Substances 0.000 description 3
- 238000001990 intravenous administration Methods 0.000 description 3
- 239000007788 liquid Substances 0.000 description 3
- 230000016273 neuron death Effects 0.000 description 3
- 238000003199 nucleic acid amplification method Methods 0.000 description 3
- 239000002773 nucleotide Substances 0.000 description 3
- 125000003729 nucleotide group Chemical group 0.000 description 3
- 238000007911 parenteral administration Methods 0.000 description 3
- 230000036961 partial effect Effects 0.000 description 3
- 238000006366 phosphorylation reaction Methods 0.000 description 3
- 239000013612 plasmid Substances 0.000 description 3
- 229920002451 polyvinyl alcohol Polymers 0.000 description 3
- 229920001592 potato starch Polymers 0.000 description 3
- 201000000980 schizophrenia Diseases 0.000 description 3
- 230000000862 serotonergic effect Effects 0.000 description 3
- 239000005720 sucrose Substances 0.000 description 3
- 239000006228 supernatant Substances 0.000 description 3
- 238000005406 washing Methods 0.000 description 3
- YKGKBQYSHBGZQB-UHFFFAOYSA-N 11-bromo-7,12-dihydro-5h-indolo[3,2-d][1]benzazepin-6-one Chemical compound C1C(=O)NC2=CC=CC=C2C2=C1C(C=CC=C1Br)=C1N2 YKGKBQYSHBGZQB-UHFFFAOYSA-N 0.000 description 2
- NAYAHQPYRSIGCX-UHFFFAOYSA-N 2-(2,4-dichlorophenyl)acetamide Chemical compound NC(=O)CC1=CC=C(Cl)C=C1Cl NAYAHQPYRSIGCX-UHFFFAOYSA-N 0.000 description 2
- WIQRSJOCVVPMPS-UHFFFAOYSA-N 3-(1h-indol-2-yl)pyrrole-2,5-dione Chemical class O=C1NC(=O)C(C=2NC3=CC=CC=C3C=2)=C1 WIQRSJOCVVPMPS-UHFFFAOYSA-N 0.000 description 2
- HEAJGTIEQPBAMJ-UHFFFAOYSA-N 9-bromo-12-ethyl-5,7-dihydroindolo[3,2-d][1]benzazepin-6-one Chemical compound C1C(=O)NC2=CC=CC=C2C2=C1C1=CC(Br)=CC=C1N2CC HEAJGTIEQPBAMJ-UHFFFAOYSA-N 0.000 description 2
- 102000002260 Alkaline Phosphatase Human genes 0.000 description 2
- 108020004774 Alkaline Phosphatase Proteins 0.000 description 2
- 102100021569 Apoptosis regulator Bcl-2 Human genes 0.000 description 2
- 102000016928 DNA-directed DNA polymerase Human genes 0.000 description 2
- 108010014303 DNA-directed DNA polymerase Proteins 0.000 description 2
- 102000016911 Deoxyribonucleases Human genes 0.000 description 2
- 108010053770 Deoxyribonucleases Proteins 0.000 description 2
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 2
- 102000004190 Enzymes Human genes 0.000 description 2
- 108090000790 Enzymes Proteins 0.000 description 2
- WSFSSNUMVMOOMR-UHFFFAOYSA-N Formaldehyde Chemical compound O=C WSFSSNUMVMOOMR-UHFFFAOYSA-N 0.000 description 2
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 2
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 2
- 239000004471 Glycine Substances 0.000 description 2
- 108010043121 Green Fluorescent Proteins Proteins 0.000 description 2
- 102000004144 Green Fluorescent Proteins Human genes 0.000 description 2
- SQUHHTBVTRBESD-UHFFFAOYSA-N Hexa-Ac-myo-Inositol Natural products CC(=O)OC1C(OC(C)=O)C(OC(C)=O)C(OC(C)=O)C(OC(C)=O)C1OC(C)=O SQUHHTBVTRBESD-UHFFFAOYSA-N 0.000 description 2
- 101000971171 Homo sapiens Apoptosis regulator Bcl-2 Proteins 0.000 description 2
- YNAVUWVOSKDBBP-UHFFFAOYSA-N Morpholine Chemical compound C1COCCN1 YNAVUWVOSKDBBP-UHFFFAOYSA-N 0.000 description 2
- 229910019142 PO4 Inorganic materials 0.000 description 2
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 2
- NQRYJNQNLNOLGT-UHFFFAOYSA-N Piperidine Chemical compound C1CCNCC1 NQRYJNQNLNOLGT-UHFFFAOYSA-N 0.000 description 2
- KAESVJOAVNADME-UHFFFAOYSA-N Pyrrole Chemical compound C=1C=CNC=1 KAESVJOAVNADME-UHFFFAOYSA-N 0.000 description 2
- 229940124639 Selective inhibitor Drugs 0.000 description 2
- 108020004459 Small interfering RNA Proteins 0.000 description 2
- UIIMBOGNXHQVGW-UHFFFAOYSA-M Sodium bicarbonate Chemical compound [Na+].OC([O-])=O UIIMBOGNXHQVGW-UHFFFAOYSA-M 0.000 description 2
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 2
- DTQVDTLACAAQTR-UHFFFAOYSA-N Trifluoroacetic acid Chemical compound OC(=O)C(F)(F)F DTQVDTLACAAQTR-UHFFFAOYSA-N 0.000 description 2
- 208000027418 Wounds and injury Diseases 0.000 description 2
- 210000004504 adult stem cell Anatomy 0.000 description 2
- 230000001640 apoptogenic effect Effects 0.000 description 2
- 239000007864 aqueous solution Substances 0.000 description 2
- 239000011230 binding agent Substances 0.000 description 2
- 230000037396 body weight Effects 0.000 description 2
- 210000004958 brain cell Anatomy 0.000 description 2
- 125000004369 butenyl group Chemical group C(=CCC)* 0.000 description 2
- 238000007796 conventional method Methods 0.000 description 2
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 2
- 229940104302 cytosine Drugs 0.000 description 2
- 239000007884 disintegrant Substances 0.000 description 2
- 210000005064 dopaminergic neuron Anatomy 0.000 description 2
- 238000005516 engineering process Methods 0.000 description 2
- 229940088598 enzyme Drugs 0.000 description 2
- 238000011156 evaluation Methods 0.000 description 2
- 239000013604 expression vector Substances 0.000 description 2
- 239000012091 fetal bovine serum Substances 0.000 description 2
- 239000012894 fetal calf serum Substances 0.000 description 2
- GNBHRKFJIUUOQI-UHFFFAOYSA-N fluorescein Chemical compound O1C(=O)C2=CC=CC=C2C21C1=CC=C(O)C=C1OC1=CC(O)=CC=C21 GNBHRKFJIUUOQI-UHFFFAOYSA-N 0.000 description 2
- 239000008103 glucose Substances 0.000 description 2
- 239000008187 granular material Substances 0.000 description 2
- 239000005090 green fluorescent protein Substances 0.000 description 2
- UYTPUPDQBNUYGX-UHFFFAOYSA-N guanine Chemical compound O=C1NC(N)=NC2=C1N=CN2 UYTPUPDQBNUYGX-UHFFFAOYSA-N 0.000 description 2
- 238000010438 heat treatment Methods 0.000 description 2
- 238000001727 in vivo Methods 0.000 description 2
- 238000011065 in-situ storage Methods 0.000 description 2
- 208000014674 injury Diseases 0.000 description 2
- CDAISMWEOUEBRE-GPIVLXJGSA-N inositol Chemical compound O[C@H]1[C@H](O)[C@@H](O)[C@H](O)[C@H](O)[C@@H]1O CDAISMWEOUEBRE-GPIVLXJGSA-N 0.000 description 2
- 229960000367 inositol Drugs 0.000 description 2
- 208000037906 ischaemic injury Diseases 0.000 description 2
- 208000028867 ischemia Diseases 0.000 description 2
- 239000000314 lubricant Substances 0.000 description 2
- VFIJGAWYVXDYLK-UHFFFAOYSA-N methyl 2-(1h-indol-3-yl)-2-oxoacetate Chemical compound C1=CC=C2C(C(=O)C(=O)OC)=CNC2=C1 VFIJGAWYVXDYLK-UHFFFAOYSA-N 0.000 description 2
- 150000004702 methyl esters Chemical class 0.000 description 2
- 125000000325 methylidene group Chemical group [H]C([H])=* 0.000 description 2
- 230000015656 negative regulation of neurogenesis Effects 0.000 description 2
- 210000005170 neoplastic cell Anatomy 0.000 description 2
- 239000002858 neurotransmitter agent Substances 0.000 description 2
- 239000003921 oil Substances 0.000 description 2
- 235000019198 oils Nutrition 0.000 description 2
- DIVDFFZHCJEHGG-UHFFFAOYSA-N oxidopamine Chemical compound NCCC1=CC(O)=C(O)C=C1O DIVDFFZHCJEHGG-UHFFFAOYSA-N 0.000 description 2
- 239000000546 pharmaceutical excipient Substances 0.000 description 2
- 239000010452 phosphate Substances 0.000 description 2
- UEZVMMHDMIWARA-UHFFFAOYSA-M phosphonate Chemical compound [O-]P(=O)=O UEZVMMHDMIWARA-UHFFFAOYSA-M 0.000 description 2
- 230000026731 phosphorylation Effects 0.000 description 2
- 239000004014 plasticizer Substances 0.000 description 2
- 239000000843 powder Substances 0.000 description 2
- 239000003755 preservative agent Substances 0.000 description 2
- 230000002062 proliferating effect Effects 0.000 description 2
- 238000000746 purification Methods 0.000 description 2
- CDAISMWEOUEBRE-UHFFFAOYSA-N scyllo-inosotol Natural products OC1C(O)C(O)C(O)C(O)C1O CDAISMWEOUEBRE-UHFFFAOYSA-N 0.000 description 2
- BTIHMVBBUGXLCJ-OAHLLOKOSA-N seliciclib Chemical compound C=12N=CN(C(C)C)C2=NC(N[C@@H](CO)CC)=NC=1NCC1=CC=CC=C1 BTIHMVBBUGXLCJ-OAHLLOKOSA-N 0.000 description 2
- QZAYGJVTTNCVMB-UHFFFAOYSA-N serotonin Chemical compound C1=C(O)C=C2C(CCN)=CNC2=C1 QZAYGJVTTNCVMB-UHFFFAOYSA-N 0.000 description 2
- 210000002966 serum Anatomy 0.000 description 2
- 238000010561 standard procedure Methods 0.000 description 2
- 230000000638 stimulation Effects 0.000 description 2
- 238000003756 stirring Methods 0.000 description 2
- 239000004094 surface-active agent Substances 0.000 description 2
- 238000001356 surgical procedure Methods 0.000 description 2
- 208000024891 symptom Diseases 0.000 description 2
- 239000006188 syrup Substances 0.000 description 2
- 235000020357 syrup Nutrition 0.000 description 2
- 239000003826 tablet Substances 0.000 description 2
- 230000001225 therapeutic effect Effects 0.000 description 2
- 210000000689 upper leg Anatomy 0.000 description 2
- 102000040650 (ribonucleotides)n+m Human genes 0.000 description 1
- KZKAYEGOIJEWQB-UHFFFAOYSA-N 1,3-dibromopropane;n,n,n',n'-tetramethylhexane-1,6-diamine Chemical compound BrCCCBr.CN(C)CCCCCCN(C)C KZKAYEGOIJEWQB-UHFFFAOYSA-N 0.000 description 1
- KMJPCMBQOGKNAK-UHFFFAOYSA-N 1-ethyl-7,12-dihydro-5h-indolo[3,2-d][1]benzazepin-6-one Chemical compound C1=2C(CC)=CC=CC=2NC(=O)CC2=C1NC1=CC=CC=C21 KMJPCMBQOGKNAK-UHFFFAOYSA-N 0.000 description 1
- IXPNQXFRVYWDDI-UHFFFAOYSA-N 1-methyl-2,4-dioxo-1,3-diazinane-5-carboximidamide Chemical compound CN1CC(C(N)=N)C(=O)NC1=O IXPNQXFRVYWDDI-UHFFFAOYSA-N 0.000 description 1
- LAAHPFCQVWTSIK-UHFFFAOYSA-N 1-propylindole Chemical compound C1=CC=C2N(CCC)C=CC2=C1 LAAHPFCQVWTSIK-UHFFFAOYSA-N 0.000 description 1
- 238000005160 1H NMR spectroscopy Methods 0.000 description 1
- YBYIRNPNPLQARY-UHFFFAOYSA-N 1H-indene Natural products C1=CC=C2CC=CC2=C1 YBYIRNPNPLQARY-UHFFFAOYSA-N 0.000 description 1
- RVTOIQOTZUKDTE-UHFFFAOYSA-N 2,3-dimethoxy-9-(trifluoromethyl)-7,12-dihydro-5h-indolo[3,2-d][1]benzazepin-6-one Chemical compound C1=2C=C(OC)C(OC)=CC=2NC(=O)CC2=C1NC1=CC=C(C(F)(F)F)C=C21 RVTOIQOTZUKDTE-UHFFFAOYSA-N 0.000 description 1
- GXMWLJKTGBZMBH-UHFFFAOYSA-N 2-(2,4-dichlorophenyl)acetic acid Chemical compound OC(=O)CC1=CC=C(Cl)C=C1Cl GXMWLJKTGBZMBH-UHFFFAOYSA-N 0.000 description 1
- JABNPSKWVNCGMX-UHFFFAOYSA-N 2-(4-ethoxyphenyl)-6-[6-(4-methylpiperazin-1-yl)-1h-benzimidazol-2-yl]-1h-benzimidazole;trihydrochloride Chemical compound Cl.Cl.Cl.C1=CC(OCC)=CC=C1C1=NC2=CC=C(C=3NC4=CC(=CC=C4N=3)N3CCN(C)CC3)C=C2N1 JABNPSKWVNCGMX-UHFFFAOYSA-N 0.000 description 1
- IAJGQMXAKJOLBA-UHFFFAOYSA-N 2-bromo-9-nitro-7,12-dihydro-5h-indolo[3,2-d][1]benzazepin-6-one Chemical compound C1C(=O)NC2=CC=C(Br)C=C2C2=C1C1=CC([N+](=O)[O-])=CC=C1N2 IAJGQMXAKJOLBA-UHFFFAOYSA-N 0.000 description 1
- LSBDFXRDZJMBSC-UHFFFAOYSA-N 2-phenylacetamide Chemical compound NC(=O)CC1=CC=CC=C1 LSBDFXRDZJMBSC-UHFFFAOYSA-N 0.000 description 1
- DWLMLPCWSGBXMZ-UHFFFAOYSA-N 3,4-bis(1-methylindol-3-yl)pyrrole-2,5-dione Chemical compound C12=CC=CC=C2N(C)C=C1C1=C(C=2C3=CC=CC=C3N(C)C=2)C(=O)NC1=O DWLMLPCWSGBXMZ-UHFFFAOYSA-N 0.000 description 1
- MVWNPTRDUDXOPY-UHFFFAOYSA-N 3-[6-oxo-9-(trifluoromethyl)-7,12-dihydro-5h-indolo[3,2-d][1]benzazepin-2-yl]prop-2-enenitrile Chemical compound C1C(=O)NC2=CC=C(C=CC#N)C=C2C2=C1C1=CC(C(F)(F)F)=CC=C1N2 MVWNPTRDUDXOPY-UHFFFAOYSA-N 0.000 description 1
- SHBOZSJPVRMXBA-UHFFFAOYSA-N 3-[6-oxo-9-(trifluoromethyl)-7,12-dihydro-5h-indolo[3,2-d][1]benzazepin-2-yl]propanenitrile Chemical compound C1C(=O)NC2=CC=C(CCC#N)C=C2C2=C1C1=CC(C(F)(F)F)=CC=C1N2 SHBOZSJPVRMXBA-UHFFFAOYSA-N 0.000 description 1
- WUQNGTJFNCCRKY-UHFFFAOYSA-N 3-indol-1-ylpropan-1-amine Chemical compound C1=CC=C2N(CCCN)C=CC2=C1 WUQNGTJFNCCRKY-UHFFFAOYSA-N 0.000 description 1
- FWMNVWWHGCHHJJ-SKKKGAJSSA-N 4-amino-1-[(2r)-6-amino-2-[[(2r)-2-[[(2r)-2-[[(2r)-2-amino-3-phenylpropanoyl]amino]-3-phenylpropanoyl]amino]-4-methylpentanoyl]amino]hexanoyl]piperidine-4-carboxylic acid Chemical compound C([C@H](C(=O)N[C@H](CC(C)C)C(=O)N[C@H](CCCCN)C(=O)N1CCC(N)(CC1)C(O)=O)NC(=O)[C@H](N)CC=1C=CC=CC=1)C1=CC=CC=C1 FWMNVWWHGCHHJJ-SKKKGAJSSA-N 0.000 description 1
- 150000005168 4-hydroxybenzoic acids Chemical class 0.000 description 1
- ZCYVEMRRCGMTRW-UHFFFAOYSA-N 7553-56-2 Chemical compound [I] ZCYVEMRRCGMTRW-UHFFFAOYSA-N 0.000 description 1
- GJZCFRVBDYYNIV-UHFFFAOYSA-N 9-bromo-12-prop-2-enyl-5,7-dihydroindolo[3,2-d][1]benzazepin-6-one Chemical compound C1C(=O)NC2=CC=CC=C2C2=C1C1=CC(Br)=CC=C1N2CC=C GJZCFRVBDYYNIV-UHFFFAOYSA-N 0.000 description 1
- ZNJYKYATCKBLHO-UHFFFAOYSA-N 9-bromo-5-methyl-7,12-dihydroindolo[3,2-d][1]benzazepin-6-one Chemical compound C1C(=O)N(C)C2=CC=CC=C2C2=C1C1=CC(Br)=CC=C1N2 ZNJYKYATCKBLHO-UHFFFAOYSA-N 0.000 description 1
- RZVAJINKPMORJF-UHFFFAOYSA-N Acetaminophen Chemical compound CC(=O)NC1=CC=C(O)C=C1 RZVAJINKPMORJF-UHFFFAOYSA-N 0.000 description 1
- QTBSBXVTEAMEQO-UHFFFAOYSA-M Acetate Chemical compound CC([O-])=O QTBSBXVTEAMEQO-UHFFFAOYSA-M 0.000 description 1
- 229920000936 Agarose Polymers 0.000 description 1
- 239000012103 Alexa Fluor 488 Substances 0.000 description 1
- QGZKDVFQNNGYKY-UHFFFAOYSA-O Ammonium Chemical compound [NH4+] QGZKDVFQNNGYKY-UHFFFAOYSA-O 0.000 description 1
- NLXLAEXVIDQMFP-UHFFFAOYSA-N Ammonium chloride Substances [NH4+].[Cl-] NLXLAEXVIDQMFP-UHFFFAOYSA-N 0.000 description 1
- VHUUQVKOLVNVRT-UHFFFAOYSA-N Ammonium hydroxide Chemical compound [NH4+].[OH-] VHUUQVKOLVNVRT-UHFFFAOYSA-N 0.000 description 1
- 208000037259 Amyloid Plaque Diseases 0.000 description 1
- 206010002091 Anaesthesia Diseases 0.000 description 1
- 108090000145 Bacillolysin Proteins 0.000 description 1
- 201000006474 Brain Ischemia Diseases 0.000 description 1
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical compound [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 description 1
- CPAWWGVWNXHNIH-UHFFFAOYSA-N CC(C)(C)OC(C1=CC=CC(NC(C2)=O)=C1C1=C2C2=CC=CC=C2N1)=O Chemical compound CC(C)(C)OC(C1=CC=CC(NC(C2)=O)=C1C1=C2C2=CC=CC=C2N1)=O CPAWWGVWNXHNIH-UHFFFAOYSA-N 0.000 description 1
- 241000283707 Capra Species 0.000 description 1
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 1
- 206010008120 Cerebral ischaemia Diseases 0.000 description 1
- 241000282994 Cervidae Species 0.000 description 1
- VEXZGXHMUGYJMC-UHFFFAOYSA-M Chloride anion Chemical compound [Cl-] VEXZGXHMUGYJMC-UHFFFAOYSA-M 0.000 description 1
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 description 1
- 108050006400 Cyclin Proteins 0.000 description 1
- 102000016736 Cyclin Human genes 0.000 description 1
- 102000004127 Cytokines Human genes 0.000 description 1
- 108090000695 Cytokines Proteins 0.000 description 1
- FBPFZTCFMRRESA-KVTDHHQDSA-N D-Mannitol Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-KVTDHHQDSA-N 0.000 description 1
- SRBFZHDQGSBBOR-IOVATXLUSA-N D-xylopyranose Chemical group O[C@@H]1COC(O)[C@H](O)[C@H]1O SRBFZHDQGSBBOR-IOVATXLUSA-N 0.000 description 1
- KCXVZYZYPLLWCC-UHFFFAOYSA-N EDTA Chemical compound OC(=O)CN(CC(O)=O)CCN(CC(O)=O)CC(O)=O KCXVZYZYPLLWCC-UHFFFAOYSA-N 0.000 description 1
- 101800003838 Epidermal growth factor Proteins 0.000 description 1
- 102400001368 Epidermal growth factor Human genes 0.000 description 1
- 102000003951 Erythropoietin Human genes 0.000 description 1
- 108090000394 Erythropoietin Proteins 0.000 description 1
- 241000588724 Escherichia coli Species 0.000 description 1
- 208000009331 Experimental Sarcoma Diseases 0.000 description 1
- 102000003971 Fibroblast Growth Factor 1 Human genes 0.000 description 1
- 108090000386 Fibroblast Growth Factor 1 Proteins 0.000 description 1
- 102000014250 Fibroblast growth factor 12 Human genes 0.000 description 1
- 108050003239 Fibroblast growth factor 12 Proteins 0.000 description 1
- PXGOKWXKJXAPGV-UHFFFAOYSA-N Fluorine Chemical compound FF PXGOKWXKJXAPGV-UHFFFAOYSA-N 0.000 description 1
- 235000016623 Fragaria vesca Nutrition 0.000 description 1
- 240000009088 Fragaria x ananassa Species 0.000 description 1
- 235000011363 Fragaria x ananassa Nutrition 0.000 description 1
- 229930091371 Fructose Natural products 0.000 description 1
- RFSUNEUAIZKAJO-ARQDHWQXSA-N Fructose Chemical compound OC[C@H]1O[C@](O)(CO)[C@@H](O)[C@@H]1O RFSUNEUAIZKAJO-ARQDHWQXSA-N 0.000 description 1
- 239000005715 Fructose Substances 0.000 description 1
- 108091007911 GSKs Proteins 0.000 description 1
- 102000002464 Galactosidases Human genes 0.000 description 1
- 108010093031 Galactosidases Proteins 0.000 description 1
- 108010010803 Gelatin Proteins 0.000 description 1
- 102000004103 Glycogen Synthase Kinases Human genes 0.000 description 1
- 239000000028 HMX Substances 0.000 description 1
- 239000012981 Hank's balanced salt solution Substances 0.000 description 1
- 229920000209 Hexadimethrine bromide Polymers 0.000 description 1
- 101000599951 Homo sapiens Insulin-like growth factor I Proteins 0.000 description 1
- 229920002153 Hydroxypropyl cellulose Polymers 0.000 description 1
- 102100037852 Insulin-like growth factor I Human genes 0.000 description 1
- VLSMHEGGTFMBBZ-OOZYFLPDSA-M Kainate Chemical compound CC(=C)[C@H]1C[NH2+][C@H](C([O-])=O)[C@H]1CC([O-])=O VLSMHEGGTFMBBZ-OOZYFLPDSA-M 0.000 description 1
- 241000713666 Lentivirus Species 0.000 description 1
- CSNNHWWHGAXBCP-UHFFFAOYSA-L Magnesium sulfate Chemical compound [Mg+2].[O-][S+2]([O-])([O-])[O-] CSNNHWWHGAXBCP-UHFFFAOYSA-L 0.000 description 1
- 229930195725 Mannitol Natural products 0.000 description 1
- 244000246386 Mentha pulegium Species 0.000 description 1
- 235000016257 Mentha pulegium Nutrition 0.000 description 1
- 235000004357 Mentha x piperita Nutrition 0.000 description 1
- 229940123685 Monoamine oxidase inhibitor Drugs 0.000 description 1
- 244000131360 Morinda citrifolia Species 0.000 description 1
- 241001553014 Myrsine salicina Species 0.000 description 1
- AFCARXCZXQIEQB-UHFFFAOYSA-N N-[3-oxo-3-(2,4,6,7-tetrahydrotriazolo[4,5-c]pyridin-5-yl)propyl]-2-[[3-(trifluoromethoxy)phenyl]methylamino]pyrimidine-5-carboxamide Chemical compound O=C(CCNC(=O)C=1C=NC(=NC=1)NCC1=CC(=CC=C1)OC(F)(F)F)N1CC2=C(CC1)NN=N2 AFCARXCZXQIEQB-UHFFFAOYSA-N 0.000 description 1
- 229910002651 NO3 Inorganic materials 0.000 description 1
- 108010025020 Nerve Growth Factor Proteins 0.000 description 1
- 102000007072 Nerve Growth Factors Human genes 0.000 description 1
- 208000028389 Nerve injury Diseases 0.000 description 1
- 102000035092 Neutral proteases Human genes 0.000 description 1
- 108091005507 Neutral proteases Proteins 0.000 description 1
- NHNBFGGVMKEFGY-UHFFFAOYSA-N Nitrate Chemical compound [O-][N+]([O-])=O NHNBFGGVMKEFGY-UHFFFAOYSA-N 0.000 description 1
- 108091028043 Nucleic acid sequence Proteins 0.000 description 1
- 241000233855 Orchidaceae Species 0.000 description 1
- 108090000526 Papain Proteins 0.000 description 1
- 102000005877 Peptide Initiation Factors Human genes 0.000 description 1
- 108010044843 Peptide Initiation Factors Proteins 0.000 description 1
- 229940123932 Phosphodiesterase 4 inhibitor Drugs 0.000 description 1
- 239000002202 Polyethylene glycol Substances 0.000 description 1
- 108010020346 Polyglutamic Acid Proteins 0.000 description 1
- 239000004365 Protease Substances 0.000 description 1
- 108091030071 RNAI Proteins 0.000 description 1
- 238000011530 RNeasy Mini Kit Methods 0.000 description 1
- 241000700157 Rattus norvegicus Species 0.000 description 1
- 101001032571 Rattus norvegicus Glycogen synthase kinase-3 beta Proteins 0.000 description 1
- 108091081021 Sense strand Proteins 0.000 description 1
- KEAYESYHFKHZAL-UHFFFAOYSA-N Sodium Chemical compound [Na] KEAYESYHFKHZAL-UHFFFAOYSA-N 0.000 description 1
- 229920002472 Starch Polymers 0.000 description 1
- 108700025695 Suppressor Genes Proteins 0.000 description 1
- 102000004142 Trypsin Human genes 0.000 description 1
- 108090000631 Trypsin Proteins 0.000 description 1
- 241001211763 Yukara Species 0.000 description 1
- 230000032683 aging Effects 0.000 description 1
- 229910052783 alkali metal Inorganic materials 0.000 description 1
- 229910052784 alkaline earth metal Inorganic materials 0.000 description 1
- 125000005079 alkoxycarbonylmethyl group Chemical group 0.000 description 1
- VREFGVBLTWBCJP-UHFFFAOYSA-N alprazolam Chemical compound C12=CC(Cl)=CC=C2N2C(C)=NN=C2CN=C1C1=CC=CC=C1 VREFGVBLTWBCJP-UHFFFAOYSA-N 0.000 description 1
- AZDRQVAHHNSJOQ-UHFFFAOYSA-N alumane Chemical class [AlH3] AZDRQVAHHNSJOQ-UHFFFAOYSA-N 0.000 description 1
- 229910000147 aluminium phosphate Inorganic materials 0.000 description 1
- 150000001412 amines Chemical class 0.000 description 1
- QGZKDVFQNNGYKY-UHFFFAOYSA-N ammonia Natural products N QGZKDVFQNNGYKY-UHFFFAOYSA-N 0.000 description 1
- 235000011114 ammonium hydroxide Nutrition 0.000 description 1
- 150000003863 ammonium salts Chemical class 0.000 description 1
- AVKUERGKIZMTKX-NJBDSQKTSA-N ampicillin Chemical compound C1([C@@H](N)C(=O)N[C@H]2[C@H]3SC([C@@H](N3C2=O)C(O)=O)(C)C)=CC=CC=C1 AVKUERGKIZMTKX-NJBDSQKTSA-N 0.000 description 1
- 229960000723 ampicillin Drugs 0.000 description 1
- 230000037005 anaesthesia Effects 0.000 description 1
- 230000003042 antagnostic effect Effects 0.000 description 1
- 230000008485 antagonism Effects 0.000 description 1
- 125000005427 anthranyl group Chemical group 0.000 description 1
- 230000000692 anti-sense effect Effects 0.000 description 1
- 239000000935 antidepressant agent Substances 0.000 description 1
- 229940005513 antidepressants Drugs 0.000 description 1
- 125000000637 arginyl group Chemical group N[C@@H](CCCNC(N)=N)C(=O)* 0.000 description 1
- 125000002619 bicyclic group Chemical group 0.000 description 1
- 238000001574 biopsy Methods 0.000 description 1
- 230000000903 blocking effect Effects 0.000 description 1
- 210000004369 blood Anatomy 0.000 description 1
- 239000008280 blood Substances 0.000 description 1
- GDTBXPJZTBHREO-UHFFFAOYSA-N bromine Substances BrBr GDTBXPJZTBHREO-UHFFFAOYSA-N 0.000 description 1
- 229910052794 bromium Inorganic materials 0.000 description 1
- 125000001246 bromo group Chemical group Br* 0.000 description 1
- FUSUHKVFWTUUBE-UHFFFAOYSA-N buten-2-one Chemical compound CC(=O)C=C FUSUHKVFWTUUBE-UHFFFAOYSA-N 0.000 description 1
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 159000000007 calcium salts Chemical class 0.000 description 1
- 125000000609 carbazolyl group Chemical class C1(=CC=CC=2C3=CC=CC=C3NC12)* 0.000 description 1
- 229910052799 carbon Inorganic materials 0.000 description 1
- 229910002091 carbon monoxide Inorganic materials 0.000 description 1
- 125000002915 carbonyl group Chemical group [*:2]C([*:1])=O 0.000 description 1
- 238000004113 cell culture Methods 0.000 description 1
- 210000003169 central nervous system Anatomy 0.000 description 1
- 206010008118 cerebral infarction Diseases 0.000 description 1
- IUCQPIROBHBEID-UHFFFAOYSA-N chembl334031 Chemical compound C12=CC(Cl)=CC=C2NC2=C1CC(=O)NC1=C2SC=C1 IUCQPIROBHBEID-UHFFFAOYSA-N 0.000 description 1
- 239000000460 chlorine Substances 0.000 description 1
- 229910052801 chlorine Inorganic materials 0.000 description 1
- 125000001309 chloro group Chemical group Cl* 0.000 description 1
- 150000001860 citric acid derivatives Chemical class 0.000 description 1
- 230000000295 complement effect Effects 0.000 description 1
- 238000010276 construction Methods 0.000 description 1
- 239000002875 cyclin dependent kinase inhibitor Substances 0.000 description 1
- 229940043378 cyclin-dependent kinase inhibitor Drugs 0.000 description 1
- 125000001995 cyclobutyl group Chemical group [H]C1([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 1
- 125000000596 cyclohexenyl group Chemical group C1(=CCCCC1)* 0.000 description 1
- 125000000640 cyclooctyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C([H])([H])C1([H])[H] 0.000 description 1
- 125000001511 cyclopentyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 1
- 125000001559 cyclopropyl group Chemical group [H]C1([H])C([H])([H])C1([H])* 0.000 description 1
- 230000003247 decreasing effect Effects 0.000 description 1
- 125000002704 decyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 230000007850 degeneration Effects 0.000 description 1
- 230000001419 dependent effect Effects 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- 230000018109 developmental process Effects 0.000 description 1
- 235000014113 dietary fatty acids Nutrition 0.000 description 1
- 239000003085 diluting agent Substances 0.000 description 1
- BFMYDTVEBKDAKJ-UHFFFAOYSA-L disodium;(2',7'-dibromo-3',6'-dioxido-3-oxospiro[2-benzofuran-1,9'-xanthene]-4'-yl)mercury;hydrate Chemical compound O.[Na+].[Na+].O1C(=O)C2=CC=CC=C2C21C1=CC(Br)=C([O-])C([Hg])=C1OC1=C2C=C(Br)C([O-])=C1 BFMYDTVEBKDAKJ-UHFFFAOYSA-L 0.000 description 1
- 108010007093 dispase Proteins 0.000 description 1
- 239000006185 dispersion Substances 0.000 description 1
- MRTWJQDNVKEBIF-UHFFFAOYSA-N ditert-butyl 9-bromo-6-oxo-7h-indolo[3,2-d][1]benzazepine-5,12-dicarboxylate Chemical compound C1C(=O)N(C(=O)OC(C)(C)C)C2=CC=CC=C2C2=C1C1=CC(Br)=CC=C1N2C(=O)OC(C)(C)C MRTWJQDNVKEBIF-UHFFFAOYSA-N 0.000 description 1
- 239000002552 dosage form Substances 0.000 description 1
- 239000003937 drug carrier Substances 0.000 description 1
- 235000013601 eggs Nutrition 0.000 description 1
- 238000000921 elemental analysis Methods 0.000 description 1
- 108010084315 endopolyphosphatase Proteins 0.000 description 1
- 230000007613 environmental effect Effects 0.000 description 1
- 229940116977 epidermal growth factor Drugs 0.000 description 1
- 229940105423 erythropoietin Drugs 0.000 description 1
- 235000019441 ethanol Nutrition 0.000 description 1
- ZMMJGEGLRURXTF-UHFFFAOYSA-N ethidium bromide Chemical compound [Br-].C12=CC(N)=CC=C2C2=CC=C(N)C=C2[N+](CC)=C1C1=CC=CC=C1 ZMMJGEGLRURXTF-UHFFFAOYSA-N 0.000 description 1
- 229960005542 ethidium bromide Drugs 0.000 description 1
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 239000004744 fabric Substances 0.000 description 1
- 239000000194 fatty acid Substances 0.000 description 1
- 229930195729 fatty acid Natural products 0.000 description 1
- 229940029303 fibroblast growth factor-1 Drugs 0.000 description 1
- 238000000799 fluorescence microscopy Methods 0.000 description 1
- 229910052731 fluorine Inorganic materials 0.000 description 1
- 239000011737 fluorine Substances 0.000 description 1
- 238000009472 formulation Methods 0.000 description 1
- 239000000499 gel Substances 0.000 description 1
- 239000008273 gelatin Substances 0.000 description 1
- 229920000159 gelatin Polymers 0.000 description 1
- 235000019322 gelatine Nutrition 0.000 description 1
- 235000011852 gelatine desserts Nutrition 0.000 description 1
- 230000009368 gene silencing by RNA Effects 0.000 description 1
- 230000002068 genetic effect Effects 0.000 description 1
- 239000006481 glucose medium Substances 0.000 description 1
- 235000011187 glycerol Nutrition 0.000 description 1
- 150000002334 glycols Chemical class 0.000 description 1
- 125000003187 heptyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 229950007870 hexadimethrine bromide Drugs 0.000 description 1
- 125000006038 hexenyl group Chemical group 0.000 description 1
- 230000000971 hippocampal effect Effects 0.000 description 1
- 235000001050 hortel pimenta Nutrition 0.000 description 1
- 238000009396 hybridization Methods 0.000 description 1
- 239000001863 hydroxypropyl cellulose Substances 0.000 description 1
- 235000010977 hydroxypropyl cellulose Nutrition 0.000 description 1
- 238000007654 immersion Methods 0.000 description 1
- 230000006872 improvement Effects 0.000 description 1
- 125000003454 indenyl group Chemical group C1(C=CC2=CC=CC=C12)* 0.000 description 1
- 208000015181 infectious disease Diseases 0.000 description 1
- 230000000977 initiatory effect Effects 0.000 description 1
- 238000007918 intramuscular administration Methods 0.000 description 1
- 238000007914 intraventricular administration Methods 0.000 description 1
- 239000011630 iodine Substances 0.000 description 1
- 229910052740 iodine Inorganic materials 0.000 description 1
- INQOMBQAUSQDDS-UHFFFAOYSA-N iodomethane Chemical compound IC INQOMBQAUSQDDS-UHFFFAOYSA-N 0.000 description 1
- 125000000959 isobutyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 description 1
- 238000002955 isolation Methods 0.000 description 1
- 125000001972 isopentyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 238000002372 labelling Methods 0.000 description 1
- 210000003140 lateral ventricle Anatomy 0.000 description 1
- 229910003002 lithium salt Inorganic materials 0.000 description 1
- 159000000002 lithium salts Chemical class 0.000 description 1
- 125000003588 lysine group Chemical group [H]N([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])(N([H])[H])C(*)=O 0.000 description 1
- 159000000003 magnesium salts Chemical class 0.000 description 1
- VZCYOOQTPOCHFL-UPHRSURJSA-N maleic acid Chemical compound OC(=O)\C=C/C(O)=O VZCYOOQTPOCHFL-UPHRSURJSA-N 0.000 description 1
- 239000000594 mannitol Substances 0.000 description 1
- 235000010355 mannitol Nutrition 0.000 description 1
- 239000003550 marker Substances 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- 230000007246 mechanism Effects 0.000 description 1
- 238000002844 melting Methods 0.000 description 1
- 230000008018 melting Effects 0.000 description 1
- 108020004999 messenger RNA Proteins 0.000 description 1
- 229910052751 metal Inorganic materials 0.000 description 1
- 239000002184 metal Substances 0.000 description 1
- KFKXSMSQHIOMSO-UHFFFAOYSA-N methyl 2-oxoacetate Chemical compound COC(=O)C=O KFKXSMSQHIOMSO-UHFFFAOYSA-N 0.000 description 1
- 229940060942 methylin Drugs 0.000 description 1
- 230000000813 microbial effect Effects 0.000 description 1
- 238000009629 microbiological culture Methods 0.000 description 1
- 238000002156 mixing Methods 0.000 description 1
- 239000003068 molecular probe Substances 0.000 description 1
- 239000002899 monoamine oxidase inhibitor Substances 0.000 description 1
- 125000002950 monocyclic group Chemical group 0.000 description 1
- 239000004050 mood stabilizer Substances 0.000 description 1
- 230000035772 mutation Effects 0.000 description 1
- 125000001624 naphthyl group Chemical group 0.000 description 1
- 125000001971 neopentyl group Chemical group [H]C([*])([H])C(C([H])([H])[H])(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 210000001577 neostriatum Anatomy 0.000 description 1
- 210000002682 neurofibrillary tangle Anatomy 0.000 description 1
- 239000003900 neurotrophic factor Substances 0.000 description 1
- 230000007935 neutral effect Effects 0.000 description 1
- 235000017524 noni Nutrition 0.000 description 1
- 231100000252 nontoxic Toxicity 0.000 description 1
- 230000003000 nontoxic effect Effects 0.000 description 1
- 125000001400 nonyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 238000010899 nucleation Methods 0.000 description 1
- 108020004707 nucleic acids Proteins 0.000 description 1
- 102000039446 nucleic acids Human genes 0.000 description 1
- 150000007523 nucleic acids Chemical class 0.000 description 1
- 125000003261 o-tolyl group Chemical group [H]C1=C([H])C(*)=C(C([H])=C1[H])C([H])([H])[H] 0.000 description 1
- 125000004365 octenyl group Chemical group C(=CCCCCCC)* 0.000 description 1
- UZGLIIJVICEWHF-UHFFFAOYSA-N octogen Chemical compound [O-][N+](=O)N1CN([N+]([O-])=O)CN([N+]([O-])=O)CN([N+]([O-])=O)C1 UZGLIIJVICEWHF-UHFFFAOYSA-N 0.000 description 1
- 125000002347 octyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 210000000956 olfactory bulb Anatomy 0.000 description 1
- 239000004006 olive oil Substances 0.000 description 1
- 235000008390 olive oil Nutrition 0.000 description 1
- 239000012044 organic layer Substances 0.000 description 1
- 229940055729 papain Drugs 0.000 description 1
- 235000019834 papain Nutrition 0.000 description 1
- 125000002255 pentenyl group Chemical group C(=CCCC)* 0.000 description 1
- 125000001147 pentyl group Chemical group C(CCCC)* 0.000 description 1
- 230000002093 peripheral effect Effects 0.000 description 1
- 210000002856 peripheral neuron Anatomy 0.000 description 1
- 230000000144 pharmacologic effect Effects 0.000 description 1
- 239000002587 phosphodiesterase IV inhibitor Substances 0.000 description 1
- 229920001223 polyethylene glycol Polymers 0.000 description 1
- 229920002643 polyglutamic acid Polymers 0.000 description 1
- 239000004800 polyvinyl chloride Substances 0.000 description 1
- 229920000915 polyvinyl chloride Polymers 0.000 description 1
- 239000013641 positive control Substances 0.000 description 1
- 229910052700 potassium Inorganic materials 0.000 description 1
- 239000011591 potassium Substances 0.000 description 1
- XAEFZNCEHLXOMS-UHFFFAOYSA-M potassium benzoate Chemical compound [K+].[O-]C(=O)C1=CC=CC=C1 XAEFZNCEHLXOMS-UHFFFAOYSA-M 0.000 description 1
- LPNYRYFBWFDTMA-UHFFFAOYSA-N potassium tert-butoxide Chemical compound [K+].CC(C)(C)[O-] LPNYRYFBWFDTMA-UHFFFAOYSA-N 0.000 description 1
- OXCMYAYHXIHQOA-UHFFFAOYSA-N potassium;[2-butyl-5-chloro-3-[[4-[2-(1,2,4-triaza-3-azanidacyclopenta-1,4-dien-5-yl)phenyl]phenyl]methyl]imidazol-4-yl]methanol Chemical compound [K+].CCCCC1=NC(Cl)=C(CO)N1CC1=CC=C(C=2C(=CC=CC=2)C2=N[N-]N=N2)C=C1 OXCMYAYHXIHQOA-UHFFFAOYSA-N 0.000 description 1
- 102000004196 processed proteins & peptides Human genes 0.000 description 1
- 239000000047 product Substances 0.000 description 1
- 235000013772 propylene glycol Nutrition 0.000 description 1
- 210000002763 pyramidal cell Anatomy 0.000 description 1
- 239000011541 reaction mixture Substances 0.000 description 1
- 238000009256 replacement therapy Methods 0.000 description 1
- 238000011160 research Methods 0.000 description 1
- 230000001177 retroviral effect Effects 0.000 description 1
- 238000003757 reverse transcription PCR Methods 0.000 description 1
- 239000003161 ribonuclease inhibitor Substances 0.000 description 1
- 239000012266 salt solution Substances 0.000 description 1
- 229920006395 saturated elastomer Polymers 0.000 description 1
- 125000002914 sec-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 229940076279 serotonin Drugs 0.000 description 1
- 239000008159 sesame oil Substances 0.000 description 1
- 235000011803 sesame oil Nutrition 0.000 description 1
- 150000004760 silicates Chemical class 0.000 description 1
- 210000003625 skull Anatomy 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- 239000000661 sodium alginate Substances 0.000 description 1
- 235000010413 sodium alginate Nutrition 0.000 description 1
- 229940005550 sodium alginate Drugs 0.000 description 1
- 235000017557 sodium bicarbonate Nutrition 0.000 description 1
- 229910000030 sodium bicarbonate Inorganic materials 0.000 description 1
- 238000002415 sodium dodecyl sulfate polyacrylamide gel electrophoresis Methods 0.000 description 1
- 239000012312 sodium hydride Substances 0.000 description 1
- 229910000104 sodium hydride Inorganic materials 0.000 description 1
- 159000000000 sodium salts Chemical class 0.000 description 1
- 239000003549 soybean oil Substances 0.000 description 1
- 235000012424 soybean oil Nutrition 0.000 description 1
- 239000008107 starch Substances 0.000 description 1
- 235000019698 starch Nutrition 0.000 description 1
- 230000003068 static effect Effects 0.000 description 1
- 238000007920 subcutaneous administration Methods 0.000 description 1
- 125000000547 substituted alkyl group Chemical group 0.000 description 1
- 235000000346 sugar Nutrition 0.000 description 1
- 150000008163 sugars Chemical class 0.000 description 1
- 125000005420 sulfonamido group Chemical group S(=O)(=O)(N*)* 0.000 description 1
- 125000000472 sulfonyl group Chemical group *S(*)(=O)=O 0.000 description 1
- 239000013589 supplement Substances 0.000 description 1
- 239000000725 suspension Substances 0.000 description 1
- 239000000454 talc Substances 0.000 description 1
- 229910052623 talc Inorganic materials 0.000 description 1
- 102000013498 tau Proteins Human genes 0.000 description 1
- 108010026424 tau Proteins Proteins 0.000 description 1
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 1
- QEMXHQIAXOOASZ-UHFFFAOYSA-N tetramethylammonium Chemical compound C[N+](C)(C)C QEMXHQIAXOOASZ-UHFFFAOYSA-N 0.000 description 1
- 238000002560 therapeutic procedure Methods 0.000 description 1
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 description 1
- 238000001890 transfection Methods 0.000 description 1
- 239000012096 transfection reagent Substances 0.000 description 1
- 230000014616 translation Effects 0.000 description 1
- 238000002054 transplantation Methods 0.000 description 1
- 239000012588 trypsin Substances 0.000 description 1
- 230000004614 tumor growth Effects 0.000 description 1
- 241000701161 unidentified adenovirus Species 0.000 description 1
- VBEQCZHXXJYVRD-GACYYNSASA-N uroanthelone Chemical compound C([C@@H](C(=O)N[C@H](C(=O)N[C@@H](CS)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CS)C(=O)N[C@H](C(=O)N[C@@H]([C@@H](C)CC)C(=O)NCC(=O)N[C@@H](CC=1C=CC(O)=CC=1)C(=O)N[C@@H](CO)C(=O)NCC(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CS)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCNC(N)=N)C(O)=O)C(C)C)[C@@H](C)O)NC(=O)[C@H](CO)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CO)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@@H](NC(=O)[C@H](CC=1NC=NC=1)NC(=O)[C@H](CCSC)NC(=O)[C@H](CS)NC(=O)[C@@H](NC(=O)CNC(=O)CNC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CS)NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)CNC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@H](CO)NC(=O)[C@H](CO)NC(=O)[C@H]1N(CCC1)C(=O)[C@H](CS)NC(=O)CNC(=O)[C@H]1N(CCC1)C(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@H](CO)NC(=O)[C@@H](N)CC(N)=O)C(C)C)[C@@H](C)CC)C1=CC=C(O)C=C1 VBEQCZHXXJYVRD-GACYYNSASA-N 0.000 description 1
- 239000013603 viral vector Substances 0.000 description 1
- 238000001262 western blot Methods 0.000 description 1
- 150000003751 zinc Chemical class 0.000 description 1
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/40—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with one nitrogen as the only ring hetero atom, e.g. sulpiride, succinimide, tolmetin, buflomedil
- A61K31/403—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with one nitrogen as the only ring hetero atom, e.g. sulpiride, succinimide, tolmetin, buflomedil condensed with carbocyclic rings, e.g. carbazole
- A61K31/404—Indoles, e.g. pindolol
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/40—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with one nitrogen as the only ring hetero atom, e.g. sulpiride, succinimide, tolmetin, buflomedil
- A61K31/403—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with one nitrogen as the only ring hetero atom, e.g. sulpiride, succinimide, tolmetin, buflomedil condensed with carbocyclic rings, e.g. carbazole
- A61K31/404—Indoles, e.g. pindolol
- A61K31/4045—Indole-alkylamines; Amides thereof, e.g. serotonin, melatonin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/40—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with one nitrogen as the only ring hetero atom, e.g. sulpiride, succinimide, tolmetin, buflomedil
- A61K31/407—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with one nitrogen as the only ring hetero atom, e.g. sulpiride, succinimide, tolmetin, buflomedil condensed with other heterocyclic ring systems, e.g. ketorolac, physostigmine
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/55—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having seven-membered rings, e.g. azelastine, pentylenetetrazole
- A61K31/553—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having seven-membered rings, e.g. azelastine, pentylenetetrazole having at least one nitrogen and one oxygen as ring hetero atoms, e.g. loxapine, staurosporine
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/08—Antiepileptics; Anticonvulsants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/14—Drugs for disorders of the nervous system for treating abnormal movements, e.g. chorea, dyskinesia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/14—Drugs for disorders of the nervous system for treating abnormal movements, e.g. chorea, dyskinesia
- A61P25/16—Anti-Parkinson drugs
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/18—Antipsychotics, i.e. neuroleptics; Drugs for mania or schizophrenia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/22—Anxiolytics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/24—Antidepressants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/28—Drugs for disorders of the nervous system for treating neurodegenerative disorders of the central nervous system, e.g. nootropic agents, cognition enhancers, drugs for treating Alzheimer's disease or other forms of dementia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
Definitions
- the present invention relates to a nerve regenerating agent containing a substance that inhibits the activity of glycogencin zekinase-1 (hereinafter abbreviated as GSK-3) as an active ingredient, a substance that inhibits the activity of GSK-3.
- GSK-3 glycogencin zekinase-1
- the present invention relates to a neurogenesis promoting agent contained as an active ingredient, a neuron obtained by culturing a neural stem cell in the presence of the neurogenesis promoting agent, and a method for producing the neuron.
- Neurological diseases include neurodegenerative diseases in which brain and peripheral neurons are damaged by genetic factors, environmental factors, aging factors, etc., as well as depression and manic depression that do not involve nerve degeneration. Collectively. Specific examples of neurodegenerative diseases include Parkinson's disease, Alheimer's disease, polyglutamic acid disease, amyotrophic lateral sclerosis, multiple neurobati, spinal cord injury, cerebrovascular disease and the like. A common treatment for these neurodegenerative diseases is to replace neurotransmitters lost due to neuronal injury, but the improvement of symptoms is limited to Parkinson's disease, Alzheimer's disease, etc. . Neurotransmitter replacement therapy does not stop neuronal cell death.
- Regenerative medicine that regenerates the central nervous system has been studied as a treatment to positively restore the function of dopaminergic neurons lost in Parkinson's disease. Due to various problems, it is not used for general use.
- neural stem cells obtained from fetal brain ES ES cells obtained from fertilized human eggs are cultured in large quantities in vitro, and then converted to target neurons and used for transplantation.
- There is no established technology for accurately differentiating into neurons and there is a problem arising from the use of fetal neural stem cells ⁇ human ES cells, and clinical application has not progressed.
- neural stem cells are isolated from adult brain and that neurogenesis occurs in human adult brain over a lifetime.
- Methods for treating neurodegenerative diseases by inducing regeneration by stimulation with a drug or the like are being studied.
- Insulin-like growth factor-1 1 [J. Neuroscience, 20; 2896-2903 (2000)], fibroblast growth factor-1 2 [Pro. Nat. Acad. Sci. USA, 98, 5874-5879 (2001)], Stem cell factor [J. Clin. Invest., 110, 311-319 (2002)], erythropoietin [J. Neuroscience, 21, 9733-9743 (2001)], global cerebral ischemia [J. Neuroscience, 18, 7768-7778 (1998)], epilepsy stimulation [J. Neuroscience, 22, 3174-3188 (2002)], etc. It has been reported that intracerebral administration of cytokines and disease models promote neurogenesis in the hippocampus and olfactory bulb. ing.
- Peripherally administrable low molecular compounds that can induce neurogenesis include antidepressants such as monoamine oxidase inhibitors, serotonin-specific transporter inhibitors, and phosphodiesterase IV inhibitors. It has been reported [Neuropsychopharmacology, 25, 836-844 (2001)]. The mechanism by which these drugs induce nerve regeneration in the brain is that they act directly or indirectly on serotonergic receptor signals of serotonergic neurons to produce neurotrophic factors, It is thought to promote peripheral neurogenesis. Therefore, these drugs may not be available as nerve regenerating agents in most neurological disorders that are not associated with degeneration of serotonergic neurons.
- lithium a mood stabilizing drug, induces the expression of the cell death suppressor gene bcl-2, thereby protecting neonatal neurons, which are constantly growing in the hippocampus, from neuronal death and preventing neurogenesis in the hippocampus. Apparent increase has been reported [J. Neurochemis try, 75, 1729-1734 (2000)].
- Lithium is the neurotrophic factor BDNF. It has also been reported to induce actuality '[Neuropharmacology, 43, 1173-1179 (2002)]. However, it has not been reported that lithium acts directly on neural stem cells to promote neuronal differentiation, thereby promoting neurogenesis, nor has it been reported that lithium promotes neurogenesis outside of the hippocampus. . It is also unknown why lithium has a therapeutic effect on depression and manic depression without neurodegeneration.
- GSK-3 darikogen synthase kinase-3 (hereinafter abbreviated as GSK-3) excessively phosphorylates the tau protein, a small ⁇ -associated protein, to form neurofibrillary tangles, resulting in neuronal cell death.
- GSK-3 darikogen synthase kinase-3
- a hypothesis of induction has been proposed [Trends in Molecular Medicine, 8, 126-132 (2002)].
- the substance that inhibits the activity of GSK- 3 is reported to be active to protect the mature neuronal cells in in vitro [J.
- An object of the present invention is to provide a nerve regenerating agent containing a substance that inhibits the activity of GSK-3 as an active ingredient, an agent for promoting neurogenesis of neural stem cells containing the substance as an active ingredient,
- An object of the present invention is to provide a neuron obtained by culturing a neural stem cell in the presence of an agent and a method for producing the neuron.
- the present invention relates to the following (1) to (41).
- a nerve regeneration drug containing, as an active ingredient, a substance that inhibits the activity of glycogen synthase kinase 13 (hereinafter abbreviated as GSK-3).
- Neurological disorders include Parkinson's disease, Alzheimer's disease, Down's syndrome, cerebrovascular disease Is a neurological disease selected from the group consisting of harm, stroke, spinal cord injury, Huntington's chorea, multiple sclerosis, amyotrophic lateral sclerosis, epilepsy, anxiety disorder, schizophrenia, depression and manic depression (2) The medicament.
- the substances that inhibit the activity of GSK-3 are bis-indolylmaleimide derivatives, 3-aryl-4-indolylmaleimide derivatives, indole-active rubazole derivatives, and indolo [3,2-d] [1] benzazepine- 6 Any one of the above (1) to (3) ′, which is a (5H) -one derivative, an indirubin derivative, or a pharmacologically acceptable salt thereof.
- n and m are the same or different and each represents an integer of 1 to 3
- RR 3 and R 4 are the same or different and each represents a hydrogen atom, a substituted or unsubstituted lower alkyl, a substituted or unsubstituted lower alkyl.
- R 6 is a hydrogen atom, a substituted or unsubstituted lower alkyl, a substituted or unsubstituted lower alkenyl, a substituted or unsubstituted aryl or a substituted or unsubstituted cycloalkyl represented), -C00R 7 (wherein, R 7 represents hydrogen atom, a substituted or unsubstituted lower alkyl, substituted or unsubstituted Ariru or substitution or unsubstituted cycloalkyl) or - oR 8 (wherein , R 8 represents a hydrogen atom, substitution or unsubstituted lower alkyl, Ku substituted or unsubstituted Ariru or if substituted an unsubstituted cycloalkyl), R 2 and R 5 are the same or Hydrogen atom, substituted or unsubstituted lower alkyl, substituted or unsubstituted lower alkenyl, a substituted or unsubsti
- na, ma, R 1A , R 2A , R 3A and R 5A have the same meanings as n, m, RR 2 , R 3 and above) or a compound of formula (III) ′
- nb, mb, R 1B , R 2B and have the same meanings as n, m, RR z and R 5 , respectively, and R 3B and R 4B are the same or different and are each a hydrogen atom, substituted or unsubstituted. of lower alkyl, substituted or unsubstituted lower alkenyl, -C0R 6 (wherein, R 6 is as defined above), -C00R 7 (wherein R 7 is as defined above) or - oR 8 ( Wherein R 8 is as defined above, or R 3B and R 4B are taken together,
- k 1 or 2
- X represents C3 ⁇ 4, NH, an oxygen atom or a sulfur atom
- R 9 represents hydroxy, carbonyl, carbamoyl or lower alkoxycarbonyl. Or a pharmacologically acceptable salt thereof.
- R 2 a is a hydrogen atom, lower alkoxy, lower alk snow deer Lupo alkenyl represents Ariru or nitro
- R 3 a and R 4 a are the same or different and each represents a substituted or unsubstituted lower alkyl
- R 9 has the same meaning as described above
- R 9 has the same meaning as described above
- a pharmaceutically acceptable salt thereof or the pharmaceutically acceptable salt of any one of the above (1) to (3).
- the substances that inhibit the activity of GSK-3 are 3,4-bis (1-methylindole-13- ⁇ -per) -1H-pyrroyl-2,5-dione, 3- (1-methylindole 1-4- (1-propylindole-3-yl)-1H-pyrrolyl 1, 2, 5-dione, 3-[1-(3-cyanopropyl) indole-3- -]
- R 1 consists of a hydrogen atom, aryl, lower aliphatic substituent, especially alkyl and lower alkyl ester
- R "to R 14 are alkoxy, amino, acyl, aliphatic substituents, especially alkyl, alkenyl and alkynyl substituents, aliphatic alcohols, especially alkyl alcohols, aliphatic nitriles, especially alkyl nitriles
- R 15 to R 18 are independently selected from the group consisting of cyano, nitro, carboxyl, halogen, hydrogen, hydroxyl, imino and ⁇ -unsaturated ketone, wherein R 15 to R 18 are aliphatic substituents, especially alkenyl and alkynyl substituents, Especially lower aliphatic substituents, aliphatic alcohols, especially alkyl alcohols, alkoxy, acyl, cyano
- R 19 is individually selected from
- benzazepine-6 (5H) -one, 9-bromo-7,12-dihydro-2,3-dimethoxy-indolo [3,2_d] [1] benzazepine_6 (5H)- On, 9_bromo-7,12-dihydro-2,3-dihydroxy-indolo [3,2-d] [1] benzazepine-6 (5H) -one, 7,12-dihydro-2,3-dimethoxy- Indro [3, 2-d] [1] benzazepine-6 (5H) -one, 7,12-dihydro-9-trifluoromethyl-indro [3, 2-d] [1] benzazepine-6 (5H) -One, 7,12-di'hydro-2,3-dimethoxy-9-trifluoromethyl-indro [3,2-d] [1] benzazepin-6 (5H) -one, 2-bromo-7 , 12-D
- Substances that inhibit GSK-3 are 9-cyano-7,12-dihydro-indolo [3,2-d] [1] benzazepin-6 (5H) -one, 9-bromo_7,12- Dihydro-2,3-dimethoxy-indolo [3,2-d] [1] benzazepin-6 (5H) -one, 2_bromo-7,12-dihydro-9-trifluoromethyl-indro [3,2-d ] [1] Benzazepine-6 (5H) -one ..
- benzazepine-6 (5H) -one, 2-bromo-7,12-dihydro-9-trifluoromethyl-indro [3,2-d] [1] benzazepine-6 (5H) -one, 7,12-dihydro-2,3-dimethoxy-9-trifluoromethyl-ind [3,2-d] [1] benzazepine-6 (5H) -one, 2, 9 -Dibromo-7,12-dihydro-indolo [3,2-d] [1] benzazepine-6 (5H) -one, 7,12-dihydro-9-trifluoromethyl-indolo [3,2-d] [l] benzazepine-6 (5H) -one, 9-chloro-7,12-dihydro-indolo [3,2-d] [1] benzazepine-6 (5H) -one, 8-bromo-6, 11-d
- the substance that inhibits GSK-3 is 9_bromo-7,12-dihydro-indolo [3,2-d] [1] benzazepin-6 (5H) -one or its pharmacologically acceptable
- R 20 and R 25 may be the same or different; a hydrogen atom; a halogen; a hydroxy group; a methylenehydroxy group; a linear or branched C ⁇ ds-alkyl or alkoxy or a methylenealkoxy group; A cycloalkyl group having 3 to 7 carbon atoms, including one or more heteroatoms; optionally substituted or unsubstituted aryl, aralkyl or aryloxy having one or more heteroatoms.
- a straight or branched chain C ⁇ ds alkyl group, substituted or unsubstituted, if necessary, represents an aryl group containing one or more hetero
- R 2 o and R 24 and R 25 and R 29 each together form, independently of each other, a ring having from 1 to 4 optionally substituted CH 2 groups; May be the same or different Y and Z are oxygen; O ⁇ ; selenium; tellurium atom; NR 33 group (where R 33 is a hydrogen atom, one if necessary or force Lupo A straight-chain or branched ( ⁇ -di) ⁇ alkyl group substituted with a xyl, phosphoryl or sulfonate group, a substituted or unsubstituted aryl group containing one or more heteroatoms, if necessary, an aralkyl group or represents a sulfonate group); or single NOR 33 (wherein the R 33 groups are acceptable salt compound represented by or their pharmacologically and represented] a have) the meanings of (1) to ( 3) The pharmaceutical of any one of the above.
- Substances that inhibit GSK-3 are: indirubin, 5-iodo-indirubin, 5-bromo-indirubin, 5-chloro-indirubin, 5-fluoro-indirubin, 5-methyl-indirubin, 5-12-troindiene Rubin, 5- S_ ⁇ 3 H- indirubin, 5'-bromo-one ⁇ gamma Njirubin, 5-5 '- dibromo - Injirubi emissions and 5' - selected from the group consisting Promoted indirubine 5-sulfonic acid compound or a Any one of the above (1) to (3), which is a pharmacologically acceptable salt
- An agent for promoting neural stem cell neurogenesis comprising as an active ingredient a substance that inhibits the activity of GSK-3.
- Substances that inhibit the activity of GSK-3 are bis-indolylmaleimide derivatives, 3-aryl-41-indolylmaleimide derivatives, indole-active rubazole derivatives, and indolo [3,2-d] [1]
- the neurogenesis-promoting agent according to the above (20) which is a benzazepine-6 (5H) -one derivative or an indirubin derivative or a pharmacologically acceptable salt thereof.
- n and m are the same or different and each represents an integer of 1 to 3, and RR 3 and R 4 are the same or different and are a hydrogen atom, a substituted or unsubstituted lower alkyl, a substituted or unsubstituted Lower alkenyl, -C0R 6 (wherein R 6 represents a hydrogen atom, substituted or unsubstituted lower alkyl, substituted or unsubstituted lower alkenyl, substituted or unsubstituted aryl, or substituted or unsubstituted cycloalkyl ), -C00R 7 (wherein R 7 represents a hydrogen atom, a substituted or unsubstituted lower alkyl, a substituted or unsubstituted aryl or a substituted or unsubstituted cycloalkyl) or -OR 8 (wherein R 8 represents a hydrogen atom, substituted or unsubstituted lower alkyl, substituted or
- na, ma, R 1A , R 2A , R 3A and R 5A have the same meanings as n, m, RR 2 , R 3 and R 5 respectively
- nb, mb, R 1B , R 2B and R 5B have the same meanings as n, m, RR 2 and R 5 , respectively, and R 3B and R 4B are the same or different and each represents a hydrogen atom, a substituted or # lower alkyl substituted, substituted or unsubstituted lower alkenyl, - C0R 6 (wherein, R 6 is as defined above), -C00R 7 (wherein, R 7 is as defined above) or - OR 8 (wherein R 8 is as defined above), or R 3B and R 4B together
- k 1 or 2
- X represents CH 2 , NH, an oxygen atom or a sulfur atom
- R 9 represents hydroxy, .carboxy., Carbamoyl or lower alkoxycarbonyl.
- la A compound represented by the above formula (la), which is a compound represented by the formula:
- R 2a represents a hydrogen atom, lower alkoxy, lower alkoxy force Ruponiru, a nitro were Ariru or, R 3a and R 4 a are the same or different and each represents a lower alkyl substituted or unsubstituted
- the neurogenesis promoting agent according to the above (20) which is a compound represented by the formula or a pharmacologically acceptable salt thereof.
- R 3 Aa represents a substituted or unsubstituted lower alkyl
- R 5 Aa represents a halogen, or a pharmaceutically acceptable compound thereof.
- Substances that inhibit the activity of GSK-3 are 3,4-bis (1-methylindole-1-yl) -1H-pyrroyl-2,5-dione, 3- (1-methylindone-1) 3- (yl)-4- (1-propyl-indole-l-yl)-1H-pyrrole-2,5-dione, 3-(1- (3-cyanopropyl) -dyl--3--yl ] 1 4 1 (1-methylindole 1-3-yl) 1 1H-pyrrole-2,5-dione, 3-[1-(3-aminopropyl) indole-3-yl] -4- (1-methylin 1-Hyl-pyrroyl 2,5-dione, 3- [1- (3-carboxypropyl) indole-3-yl] -4-1- (1-methylindole 3-yl) 1) 1 H-pyrroyl 2,5—dione, 3— [1— (3—rubamoyl)
- R 1 consists of hydrogen, aryl, lower aliphatic substituents, especially alkyl and lower alkyl esters is selected from the group
- R "to R 14 are alkoxy, Amino, Ashiru, aliphatic substituents, particularly alkyl, alkenyl and alkynyl substituents, aliphatic alcohols - le, in particular alkyl alcohols, aliphatic nitriles, particularly alkyl nitriles , Cyano, nitro, carboxyl, halogen, hydrogen, hydroxyl, imino and ⁇ -unsaturated ketones, wherein R 15 -R 18 are aliphatic substituents, especially alkyl, alkenyl and alkynyl substituents.
- R 19 is independently selected from the group consisting of ano, nitro, epoxy, haloalkyl, halogen, hydrogen and hydroxyl, and R 19 is an aliphatic group, especially a lower alkyl group, an aliphatic alcohol, especially an alkyl alcohol, and a carboxylic acid. Or a pharmacologically acceptable salt thereof, wherein the agent is selected from the group consisting of hydrogen and hydrogen.
- Substances that inhibit GSK-3 are 7,12-dihydro-indolo [3,2-d] [1] benzoazepin-6 (5H) -one, 2-bromo-7,12-dihydro-indolo [3, 2-d] [1] benzazepine-6 (5H) -one, 9_bromo-7,12-dihydro-indolo [3,2-d] [1] benzazepine-6 (5H) -one, 9-black mouth-7,12-dihydro-indro [3,2-d] [1] benzazepine-6 (5H) -one, 1 black mouth-7,12-dihydro-indolo [3,2-d] [l] benzazepine-6 (5H) -one, 10-bromo-7,12-dihydro-indro [3,2-d] [1] benzazepine-6 (5H) -one, 8-bromo-6,11- Dihydro-thieno
- benzazepine-6 (5H) -one, 9-promo-7,12-dihydro-2,3-dimethoxy-indolo [3,2-d] [1] benzazepine-6 ( 5H) -one, 9-promo-7,12-dihydro-2,3-dihydroxy-indolo [3,2-d] [1] benzazepin-6 (5H) -one, 7,12-dihydro-2,3 -Dimethoxy-indolo [3,2-d] [1] benzazepine-6 (5H) -one, 7,12-dihydro-9-trifluoromethyl-indro [3,2-d] [1] benzazepine-6 (5H) -one, 7,12-dihydro-2,3-dimethoxy-9-trifluoromethyl-indro [3,2-d] [1] benzazepine-6 (5H) -one, 7,12-dihydro-2,3-dime
- the substance that inhibits GSK-3 is 9_cyano-7,12-dihydro-indolo.
- benzazepine-6 (5H) -one, 9-bromo-7,12-dihydro-2,3-dimethoxy-indolo [3,2-d] [1] benzazepine-6 ( 5H) -one, 2-bromo_7,12-dihydro-9-trifluoromethyl-ind, mouth [3,2-d] [1] benzazepin-6 (5H) -one, 7,12-dihydro-2, 3-dimethoxy-9-trifluoromethyl-indolo [3,2-d] [1] benzazepine-6 (5H) _one, 2,9-dibromo-7,12-dihydro-indolo [3,2-d ] [1] Benzezepine-6 (5H) -one, 7,12-dihid-9-Trifluoromethyl-indro [3,2-d] [1] Benzazepine-6 (5H) -one, 9- Black mouth-7,
- benzazepine-6 (5H) -one, 9-bromo-7,12-dihydro-2,3-dihydroxy-indolo [3,2-d] [1] benzazepine_6 ( 5H) -one, 2-bromo-7,12-dihydro-indro [3,2-d] [1] benzazepin-6 (5H) -one, 7,12-dihydro-2,3-dimethoxy-indro [3,2-d] [1] benzazepin-6 (5H) -one, 9-bromo_7,12-dihydro-12-methyl Ruindro [3,2-d] [1] Benzazepine-6 (5H) -one, 9-promo-7, 12-dihydro-5-methyloxycarbonylmethyl-indro [3, 2-d] [ 1] A compound selected from the group consisting of benzazepine-6 (5H) -one and 7,12-dihydrido-ind
- benzazepine-6 (5H) -one, 9-bromo-7,12-dihydro-2,3-dimethoxy-indolo [3,2-d] [1] benzazepine-6 ( 5H) -one, 2-bromo-7,12-dihydro_9-trifluoromethyl-indolo [3,2-d] [1] benzazepine-6 (5H): one, .7,12-dihydric mouth-2,3 -Dimethoxy-9-trifluoromethyl-indro [3,2-d] [1] benzapine_6 (5H) -one, 2,9-dibromo-7,12-dihydro-indolo [3,2-d] [1] Benzezepine-6 (5 ⁇ -one, 7,12-dihydro-9-trifluoromethyl-indolo [3,2-d] [1] Benzezepine-6 (5H) -one,
- R 2 o and R 25 may be the same or different are a hydrogen atom; a halogen; a hydroxy group; a methylenehydroxy group; a linear or branched chain; Or a methylenealkoxy group; a cycloalkyl group having 3 to 7 carbon atoms, optionally containing one or more heteroatoms; substituted or unsubstituted, optionally having one or more heteroatoms
- An aryl, aralkyl or aryloxy group a mono-, di- or trialkylsilyl group having from 1 to 6 carbon atoms in a linear or branched alkyl group independently of each other; Mono-, di- or triarylsilyl groups having a substituted or unsubstituted aryl group independently of each other; trifluoromethyl group; —C OM; —CO OM; ] ⁇ Group (where M is a hydrogen atom, a linear or branched C i -C 18 -alkyl
- Substances that inhibit GSK-3 are indirubin, 5-phosphate-indirubin, 5-bromo-indirubin, 5-cloin-indirubin, 5-fluorindirubin, 5-methyl-indirubin, 5-nitro-one indirubin, 5- S0 3
- the substance that inhibits GSK- 3 is selected from the group consisting of indirubin-1,3-monoxime, 5-iodine-indirubin-3, -monoxime and 5-S N3Na-indirubin-1,3-monoxime
- the neuronal stem cell is cultured to generate neurons, and the neurons are collected from the culture. Method of manufacturing neurons.
- a nerve regeneration method comprising administering a substance that inhibits GSK_3.
- the substance that inhibits the activity of GSK-3 may be any compound that inhibits the activity of GSK-3.
- Examples thereof include lithium, bisindolylmaleimide derivatives, 3-aryl-4-indolylmaleimide derivatives, and indolo.
- Examples of the bis (indolylmaleimide) derivative, 3-aryl-4-indolylmaleimide derivative, and indolorubazole derivative include, for example, compounds of formulas (I) to (III) The compound represented by is mentioned. Among them, 3,4-bis (1-methylindole-3-yl) -1H-pyrrole-2,5-dione, 3- (1-methylindole-3-yl) 4- (1-propylindole) 3-yl) 1H-pyrroyl 2,5-dione, 3- [1- (3-cyanopropyl) indole-3-3-yl] -4-1- (1-methylindole-3-yl) -1H —Pyrrole—2,5-dione, 3— [1- (3-Aminopropyl) indole-3-yl] 1-4— (1—methylindole—l—3-yl) 1H—pyrrole—2,5-dion, 3— [1— (3
- indolo [3,2-d] [1] benzazepin-6 (5H) -one derivative include a compound represented by the formula (IV).
- benzazepine-6 (5H) -one, 9-bromo-7,12-dihydro-5-methyloxy-propylonylmethyl-indolo [3,2-d] [1] benzazepine-6 ( 5H) -one, 9-bromo-7,12-dihydro-12-methyloxyl-ponylmethyl-indolo [3,2-d] [1] benzazepine-6 (5H) -one, 9-bromo-7,12- Dihydro-12- (2-hydroxyethyl) -indolo
- Benzazepine-6 (5H) -one, 9-bromo-7, 12-dihydro-5-methyl-ind [3, 2-d] [1] Benzazepine-6 ( 5H) -one, 5-benzyl-9-bromo-7,12-dihydro-5-methyl-indro [3,2-d] [1] benzazepine-6 (5H) -one, 9-bromo-7,12 -Dihydro-12-methyl-indro [3,2-d] [1] benzazepine-6 (5H) -one, 9-bromo-12-ethyl-7,12-dihydro-indolo [3,2-d ] [1] Benzazepine-6 (5H) -one.
- benzazepine-6 (5H) -one, 9-bromo-7,12-dihydro-2- (carboxylic acid) -ind [3,2-d] [1] benzazepine- 6 (5H) -one, 9-bromo-7,12-dihydro-10-hydroxy-indolo [3,2-d] [1] benzazepine-6 (5H) -one, 9-promo-7,12-dihydro -U-hydroxymethyl-indolo [3,2-d] [1] benzazepine-6 (5H) -one, 7,12-dihydro-4-hydroxy-indolo [3, 2_d] [1] benzazepine-6 (5H ) -One, 7,12-dihydroxy-2,3-dihydroxy-indro [3,2-d] [1] benzazepine-6 (5H) -one, 2,3-dimethoxy-9-nitro-7, 12-di
- Zazepine-6 (5H) -one, 2,3-dimethoxy-9-cyano-7,12-dihydro-indolo [3,2-d] [1] ben Zazepine-6 (5H) -one, 9-Nitro-7,12-dihydro-indro [3,2-d] [1] Benzazepine-6 (5H) -one, 3- (6-oxo-9-9 Trifluoromethyl-5,6,7,12-tetrahydrido-indro [3,2-d] [1] benzazepine-2-yl) propionitrile, 2-bromo-9-dito-7 , 12-Dihydro-indro [3,2-d] [1] benzazepine-6 (5H) -one, 3- (6-oxo-9-trifluoromethyl-5,6,7,12-tetrahydro-indolo [3,2-d] [1] benzazepine-2-yl) acrylonitrile, 23-hydroxy-
- indirubin derivative examples include, for example, a compound represented by the formula (V).
- indirubin 5-® one Doinjirubin, 5-bromo-r down Jirubin, 5 black port one indirubin, 5-fluoro over indirubine, 5-methyl-one indirubin, 5-two Toro one indirubin, 5-S0 3 H- indirubin , 5'-bromo-indirubin, 5-5, -moindirubin, 5'-bromo-indirubin 5-sulfonic acid, indirubin-1,3-monoxime, 5-odo-indirubine-1,3-monoxime and 5- S0 3 Na—Indirubin—3′—Monoxy And the like.
- Examples of the lower alkyl moiety of lower alkyl, lower alkoxy and lower alkoxyl-propyl include, for example, linear or branched alkyl having 1 to 10 carbon atoms, and specifically, methyl, ethyl ', propyl, Examples include isopropyl, butyl, isobutyl, sec-butyl, tert-butyl, pentyl, isopentyl, neopentyl, hexyl, heptyl, octyl, 6-methylheptyl, isooctyl, nonyl, and decyl.
- cycloalkyl examples include cycloalkyl having 3 to 8 carbon atoms, and specific examples thereof include cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cyclohexyl and cyclooctyl.
- Examples of lower alkenyl are-.
- straight-chain, branched or cyclic alkenyl having 2 to 8 carbon atoms such as pinyl, aryl, 1-proenyl, butenyl, pentenyl, hexenyl , Heptenyl, octenyl, cyclohexenyl, 2,6-octenyl and the like.
- the lower alkyl portion of the mono- or di-lower alkylamino has the same meaning as the lower alkyl, and the two lower alkyl portions of the di-lower alkylamino may be the same or different.
- (V) octogen represents each atom of fluorine, chlorine, bromine and iodine.
- aryl examples include monocyclic, bicyclic or tricyclic aryl having 6 to 14 carbon atoms, specifically, phenyl, naphthyl, indenyl, anthranyl and the like. .
- Substituents in substituted lower alkyl, substituted lower alkenyl, substituted lower alkoxy and substituted lower alkoxycarbonyl are the same or different, for example, having 1 to 3 substituents, halogen, cyano, nitro, hydroxy, propyloxy, Levalmoyl, amino, mono- or di-lower alkylamino, cycloalkyl, lower alkanoyl, lower Examples include alkoxy, aryl, substituted aryl, aryloxy, substituted aryloxy, lower alkoxycarbonyl, lower alkoxyyloxy, and the like.
- halogen the mono- or di-lower alkylamino, cycloalkyl, the aryl moiety of aryl and aryloxy, and the lower alkyl moiety of lower alkoxy, lower alkoxycarbonyl, lower alkyl and lower alkyloxy, respectively, are as described above. Synonymous with halogen (v), mono- or di-lower alkylamino (iv), cycloalkyl (ii), aryl (vi) and lower alkyl (i).
- the substituents in the substituted aryl and substituted aryloxy shown here may be the same or different and include, for example, lower alkyl, lower alkoxy, lower alkoxyl alkoxyl having 1 to 3 substituents, Halogen, cyano, nitro, hydroxy, propyloxy, amino and the like.
- halogen and the lower alkyl portion of the lower alkyl, lower alkoxy and lower alkoxycarbonyl shown herein have the same meaning as the above-mentioned halogen (V) and lower alkyl (i), respectively.
- Examples of the substituent in the substituted aryl and the substituted alkyl have the same groups as those described above for the substituent (vii) in the substituted lower alkyl, and include, for example, lower alkyl, substituted lower alkyl and the like. '
- substituent in the substituted lower alkyl shown here are the same or different, and include, for example, lower alkoxy, lower alkoxyl propyl, halogen, cyano, nitro, hydroxy, propyloxy, amino and the like having 1 to 3 substituents. .
- halogen and the lower alkyl moiety of the lower alkoxy and the lower alkoxycarbonyl shown herein have the same meaning as the halogen (V) and the lower alkyl ⁇ , respectively.
- the lower alkyl shown here has the same meaning as the lower alkyl (i).
- Bisindolylmaleimide derivatives 3-aryl-4_indolylmaleimide derivatives, indolo rubazole derivatives, indolo [3,2-d] [1] benzazepine-6 (5H) -one derivatives, indirubin derivatives, compounds (I As the pharmacologically acceptable salts of) to (V) and compounds (la) to (Ilia), non-toxic, water-soluble salts are preferable.
- hydrochloride, sulfate, nitrate, phosphate Inorganic acid salts such as acetate, maleate, fuma Organic salts such as-,, silicates, citrates and the like are listed.
- 'Pharmacologically acceptable metal salts include alkali metal salts such as sodium salt and potassium salt, magnesium salt, calcium salt and the like. Salts include alkaline earth metal salts, aluminum salts, zinc salts, etc., and pharmacologically acceptable ammonium salts include salts such as ammonium, tetramethylammonium, and the like. Examples of the organic amine addition salt include addition salts such as morpholine and piperidine.
- siRNA short interference RNA
- the siRNA against GSK-3 can suppress the expression of GSK-3 due to its RNAi activity, thereby inhibiting the activity of GSK-3.
- siRNA can inhibit GSK-3 activity by introducing itself into cells, and also by introducing vectors that express siRNA into cells. .
- the guanine or cytosine content is 3052% in any 19 of the messenger RNA sequences of human GSK-3. That at least 3 out of the 5 bases at the 3 'end are adenine or peridine, the melting temperature is 2 (less than T, the 3rd base is adenine, and the 10th base is The condition that the 13th base is other than glycine, the 19th base is adenine, and the 19th base is not glycine or cytosine.
- a target sequence that is highly effective By selecting a sequence that satisfies the above criteria, a target sequence that is highly effective can be selected.
- Sense strand oligoRNA with 2 nucleotides added to 3 'end of oligo RNA of the target sequence and antisense strand with 2 nucleotides added to 3' end of oligo RNA of sequence complementary to the target sequence By hybridizing both oligo RNAs, an effective siRNA against human GSK-3 can be produced. Synthesis, purification, and hybridization of siRNA can be performed by various methods (for example, it can be performed by using a 7 siRNA Construction Kit (manufactured by Ambion) and following the attached protocol).
- Vectors to be expressed can be expressed by various viral vectors such as various plasmid vectors, retrovirus vectors, lentivirus vectors, and adenovirus vectors.
- the vector can be expressed in the iGENEhU6 Vector (iGENE) by the method described above. It can be prepared by incorporating oligo DNA of the selected target sequence according to the attached protocol.Introduction of siRNA or a vector expressing the siRNA into cells can be carried out by various methods, for example, Nucleofector Device (Manufactured by Amaxa) according to the attached protocol.
- the method of searching for a substance that inhibits the activity of GSK-3 is based on [i] coexistence of GSK-3 3 ⁇ 4 GSK-3 phosphorylated peptide and ATP in the presence of the test substance, and [ii] test substance [Iii] Measured and compared the amount of phosphorylated peptide in the presence of GSK-3, GSK-3 phosphorylated peptide and ATP as described in [i] above in the absence of [Iv]
- the test substance is not particularly limited.
- peptides, proteins, cell extracts, purified products derived from the extracts, cell culture supernatants, purified products derived from the supernatants, biological samples such as serum, and biological samples derived from the biological samples Purified product, microbial cell extract, purified product derived from the extract, microbial culture supernatant, purified product derived from the supernatant, compound, compound synthesized using combinatorial chemistry, etc. it can.
- GSK-3 is not particularly limited as long as it has GSK-3 activity. But preferably mammalian, more preferably rat, mouse, monkey or human-derived GSK-3 ⁇ or / 3, and more specifically, a protein having an amino acid sequence represented by SEQ ID NO: 1. Can be given.
- GSK-3 can be obtained by introducing an expression vector having a gene encoding GSK-3 into animal cells, and culturing the animal cells.
- the gene encoding GSK-3 is not particularly limited as long as it encodes GSK-3, but is preferably GSK-3 ⁇ or / 3 derived from mammals, more preferably rat, mouse, monkey or human.
- a gene to be encoded can be mentioned, and specifically, a gene having the base sequence represented by SEQ ID NO: 2 can be mentioned.
- Examples of the peptide phosphorylated by GSK-3 include glycogen synthase.
- Examples of glycogen synthase include a peptide having an amino acid sequence represented by SEQ ID NO: 3. .
- Daricogen synthase can be obtained by, for example, introducing an expression vector having a gene encoding Daricogen synthase into animal cells, and culturing the animal cells.
- the gene encoding glycogen synthase is not particularly limited as long as it encodes glycogen synthase, but preferably encodes glycogen synthase derived from mammals, more preferably rat, mouse, monkey or human. And, specifically, a gene having the nucleotide sequence represented by SEQ ID NO: 4.
- a peptide having an amino acid sequence containing a site that is phosphorylated by GSK-3 is also phosphorylated by GSK-3. It can be used as a peptide, and specifically, a peptide having an amino acid sequence represented by SEQ ID NO: 5 can be obtained.
- GSK- 3 activity for example as AT P is a donor of phosphoric acid - with [ ⁇ 33 P] ATP, and have your test substance the presence or test substance in the absence, GSK- Phosphorylation reaction of dalycogen synthase or a peptide containing a phosphorylation site of the enzyme by step 3 and measuring the amount of 33 P. incorporated into the enzyme or peptide using a liquid scintillation counter or the like. Can be raised You.
- the nerve regenerating agent refers to an agent having an effect of directly acting on neural stem cells in the brain of a human or animal to promote neuron formation and increase the number of neurons in the brain.
- the nerve regenerating agent can be used as a therapeutic agent for a nerve disease accompanied by nerve degeneration or damage.
- the neurological diseases include Parkinson's disease, Alzheimer's disease, Down's syndrome, cerebrovascular disorder, stroke, spinal cord injury, Huntington's chorea, multiple sclerosis, amyotrophic lateral sclerosis, epilepsy, anxiety disorder, schizophrenia , Depression and manic depression.
- a substance that inhibits the activity of GSK-3 or a pharmacologically acceptable salt thereof can be administered alone as it is as a nerve regenerating agent, but it is usually provided as various pharmaceutical preparations. desirable.
- the pharmaceutical preparations are used for animals and humans.
- the nerve regenerating agent of the present invention contains, as an active ingredient, a substance that inhibits the activity of GSK-3 or a pharmacologically acceptable salt thereof alone or as a mixture with any other active ingredient for treatment. can do.
- these pharmaceutical preparations are prepared by mixing the active ingredient with one or more pharmaceutically acceptable carriers and by any method well known in the technical field of pharmaceuticals.
- the route of administration may be oral, for which it is desirable to use the most effective one for treatment, or parenteral, for example, intravenously.
- Dosage forms include tablets, powders, granules, syrups, and injections.
- Liquid preparations suitable for oral administration include water, sugars such as sucrose, sorbit, fructose, glycols such as polyethylene glycol, propylene glycol, sesame oil, olive oil, soybean oil It can be produced by using oils such as E. coli, preservatives such as P-hydroxybenzoic acid esters, and flavors such as strawberry flavor and peppermint. Tablets, powders, granules, etc.
- Formulations suitable for parenteral administration comprise sterile aqueous solutions containing the active compound, which is preferably isotonic with the blood of the recipient.
- a solution for injection is prepared using a carrier comprising a salt solution, a glucose solution, or a mixture of saline and a glucose solution.
- parenteral preparations one selected from the diluents, preservatives, flavors, excipients, disintegrants, lubricants, binders, surfactants, plasticizers, etc. exemplified for the oral preparations Alternatively, more auxiliary components can be added. .
- the dosage and frequency of administration of a substance that inhibits the activity of GSK-3 or a pharmacologically acceptable salt thereof will vary depending on the mode of administration, the age and weight of the patient, the nature or severity of the condition to be treated, Usually, in the case of oral administration, 0.01 mg to lg, preferably 0.05 to 50 mg per adult is administered once to several times a day. In the case of parenteral administration such as intravenous administration, 0.001 to 100 mg, preferably 0.01 to 10 mg, per adult is administered once to several times a day. 4. Agent for promoting neurogenesis of neural stem cells
- the agent for promoting neuronal generation of neural stem cells refers to an agent that promotes neuronal generation of neural stem cells when brought into contact with neural stem cells in vivo or in vitro.
- Neural stem cells are not particularly limited as long as they are neural stem cells, but adult neural stem cells of the brain are preferred.
- the brain may be the brain of any animal, but is preferably a mammal, more preferably a brain of a rat, mouse, monkey, human or the like.
- a substance that inhibits the activity of GSK-3 or a pharmacologically acceptable salt thereof can be used alone as it is as an agent for promoting neurogenesis of neural stem cells, but is usually provided as various pharmaceutical preparations Is desirable.
- the pharmaceutical preparations are used for animals and humans.
- the agent for promoting neurogenesis of neural stem cells of the present invention may be a substance which inhibits the activity of GSK-3 or a pharmacologically acceptable salt thereof alone or as an active ingredient for any other treatment. Can be contained as a mixture. These pharmaceutical preparations can be produced by the same method as the preparation of the above-mentioned nerve regeneration drug, and can be administered by the same administration method.
- the neurogenesis-promoting agent for neural stem cells according to the present invention is characterized in that, in vitro, the neural stem cells are brought into contact with neural stem cells, and the neurons are cultured to generate new neurons, and the neurons are collected from the culture. It can be used for the manufacturing method of the neuron which performs.
- a substance that inhibits the activity of GSK-3 or a pharmacologically acceptable salt thereof may be dissolved in the substance or the salt. It is preferable to use it by dissolving it in a solution that can be used. Examples of the solution include water and DMS0.
- Neurons obtained by culturing neural stem cells in the presence of the neurogenesis promoter of the present invention are obtained by culturing neural stem cells in the presence of the neurogenesis promoter of the present invention.
- the animal neural stem cells may be neural stem cells of any animal, and preferably include mammalian, more preferably rat, mouse, monkey and human-derived neural stem cells.
- Examples of neural stem cells include: Examples include neural stem cells derived from the brain.
- the neural stem cells may be cells derived from animals of any age or age, but preferably include adult neural stem cells.
- Obtaining adult neural stem cells from an animal The method described in J. Neuroscinece, 19, 8487 (1999) and Genes S Development, 10, 3129 (1996) is performed by a surgical technique. A method of extracting a brain from an adult animal to prepare a crude extract of brain cells, and concentrating adult stem cells from the crude extract can be mentioned.
- tissue is collected from the lateral ventricle wall of a neurological disease patient by biopsy. And a method of preparing a crude extract of brain cells and enriching adult stem cells from the extract.
- the agent for promoting neurogenesis is applied to about 1.8 ⁇ 10 5 cells / cm 2 of adult neural stem cells. It is preferred to work at a concentration of mol / l. However, lithium or a pharmacologically acceptable salt thereof is preferably used at a concentration of 100 mol / l to 10 mmol / l.
- 5% CO at 37 ° C Culture can be promoted by static culture in two atmospheres, with replacement of the whole or partial medium every 4 days for 4 to 14 days.
- the medium used for culturing adult neural stem cells may be any medium as long as it does not prevent promotion of neurogenesis, such as DMEMZF12 medium (Invitrogeri) containing 1% N2 supplement (Invitrogen). It is preferable to use
- the neurons obtained by the above culture can be recovered from the culture medium and transplanted to the affected site of a neurological disease patient by a surgical technique to be used for the treatment of the neurological disease.
- the neurological diseases include Parkinson's disease, Alzheimer's disease, Down's syndrome, cerebrovascular disorder, stroke, spinal cord injury, Huntington's chorea, multiple sclerosis', amyotrophic lateral sclerosis, epilepsy, anxiety disorder, schizophrenia, Depression and manic depression can be cited.
- Evaluation method of nerve regenerating agent of the present invention The following method can be used to confirm that the nerve regenerating agent of the present invention can regenerate neurons in vivo and treat nerve diseases.
- the above-described nerve regenerating agent of the present invention is administered to an experimental animal such as a rat or a monkey.
- the experimental animal may be a healthy animal without injury, but it is possible to effectively observe neurogenesis by giving hippocampal ischemic injury [Cell, no, 429 (2002 )], Animals in which the brain has been injured by a method such as ischemia, 6-hydroxydopamine (6-0HDA) administration or kainate administration.
- Administration routes include oral, buccal, subcutaneous, intramuscular, intravenous and intraventricular administration.
- the dose and the administration method are, for example, lOO ⁇ glOing, preferably 500 g to 500 ng per kg of body weight, administered once or several times a day.
- parenteral administration such as intravenous administration, 10 g to 1 mg, preferably 100 g to 100 ng per 1 kg of body weight is administered once or several times a day.
- Newborn neurons can be detected by the following method.
- a retroviral vector capable of expressing a cell-labelable gene such as bromodoxyperidine (BrdU), which can label proliferating cells, or Green Fluorescent Protein (GFP) or galactosidase, is used as the first substance.
- a cell-labelable gene such as bromodoxyperidine (BrdU), which can label proliferating cells, or Green Fluorescent Protein (GFP) or galactosidase
- the number of BrdU-positive cells per unit area and the ratio of the number of Tuj 1-positive cells, which is a neuronal marker, to the number of BrdU-positive cells are calculated as follows: Compare with negative control.
- the neurogenesis promoting effect of the nerve regenerating agent of the present invention and the therapeutic effect on nerve disease can be evaluated.
- ANSC-7 cells obtained by the method described in Reference Example 2 were collected from a culture medium containing DMEM / F12 medium containing 1 ml of N2 supplement (Invitrogen) and 20 ng / ml of FGF-2 (PeproTech). In a 12-well culture dish coated with two tins and laminin, spread 1.8 ⁇ 10 5 per well and incubate. The whole amount of the culture solution is replaced with a DMEM / F12 medium (manufactured by Invitrogen, hereinafter referred to as a differentiation induction medium) containing 0.5% fetal calf serum and 1% N2supplement without FGF2 to induce differentiation.
- a differentiation induction medium manufactured by Invitrogen
- PBS Invitrogen
- a substance which inhibits the activity of GSK-3 which is serially diluted in a range of 0.01 mmol / l to 5 mol / l with DMS0, is added in a volume of 1 / 1,000. Add the same volume of PBS or DMS0 as a negative control.
- the culture medium is exchanged every two days with a differentiation-inducing medium containing a substance that inhibits the activity of each GSK-3, and after a total of six days of differentiation induction, a 15% neutral buffered formalin solution (Wako Pure Chemical Industries, Ltd.) ) And fix for 20 minutes. Then, washing for 5 minutes using PBS containing 0.3% TritonX-100 (manufactured by Nacalai Tesque) is repeated three times. Next, after blocking the cells for 2 hours using 10% fetal fetal serum (manufactured by DAK0) diluted with PBS, mouse anti-Tujl (
- H33342 Fluorochrome, Trihydrochloride (manufactured by Calbiochem, hereinafter referred to as H33342) is added to a final concentration of lg / ml to stain the nuclei. After immersion in PBS, the cells are observed with an inverted fluorescence microscope (manufactured by Nikon Corporation), and the number of Tujl-positive neurons per 2.44 square millimeters is counted.
- an inverted fluorescence microscope manufactured by Nikon Corporation
- ANSC-7 cells 4.5 x 10 5 per well in a 6-well culture dish coated with polyorditin and laminin in DMEM / F12 medium containing 2 ml of orchid N2 supplement and 20 ng / ml FGF-2 7 holes were soaked and incubated.
- Total RNA was obtained from the cells in one well using an RNeasy mini kit (manufactured by QIAGEN) according to the attached protocol.
- the medium in the remaining 6 wells was completely replaced with a differentiation induction medium to induce differentiation.
- 3 mol / l of lithium chloride was added to 1 / 1,000 volume of the culture medium in 2 wells, and 1 mol / l volume of 1 mol / l of lithium chloride was added to the other 2 wells.
- the same volume of PBS was added to the remaining two wells to make a control.
- 5XDNase buffer of IO I 5XDNase buffer of IO I
- RNase inhibitor of 0.51 40U / 1
- RNase-free DNaseldU / ⁇ 1 2.5 I (promega)
- the total volume was adjusted to 501 with sterile water. After reacting at 37 ° C for 30 minutes, phenol was treated with chloroform and precipitated with ethanol.
- cDNA was prepared from all RMs of rat brain as a positive control. 1 il l of the cDNA was added to a set of 2, "1 ⁇ , mo 1/1 primer, 11 DMS0 (manufactured by Nacalai Tesque), 21 lOXExTaq buffer, 1.6 ⁇ 1 dNTPmix, 0.1 l After adding ExTaq (above, manufactured by Yu-Kara Bio Inc.) and treating at 94 ° C for 1 minute using a thermal cycler, the cycle of 94 for 1 minute, 60 for 1 minute, and 74 for 1 minute is amplified by Be2. The cDNA fragment was amplified by repeating 27 cycles of PCR for PCR and 35 cycles of PCR for amplification of BDNF, respectively.
- BDNF For amplification of BDNF, a synthetic DNA consisting of the nucleotide sequences represented by SEQ ID NOs: 8 and 9 was used as a format.
- the amplified DNA was electrophoresed on a 1.8% agarose (Nacalai Tesque) gel, stained with ethidium bromide (Nacalai Tesque), and detected with a transilluminator (Toyobo).
- the Bcl-2 band intensity did not change due to the difference in the concentration of lithium chloride at 24 hours and 6 days after the start of differentiation.
- BDNF was not expressed regardless of the difference in lithium chloride concentration.
- lithium ANSC- The effect of inhibiting apoptosis on 7 cells was analyzed.
- lithium chloride was added to a medium containing ANSC-7 cells to a final concentration of 3 ol / l, cultured for 6 days, and the ANSC-7 cells cultured with lithium chloride were added.
- ANSC-7 cells cultured with PBS were reacted with the cells using Fluorescein (manufactured by Roche Diagnostics), an in situ cell death detection kit, according to the attached protocol. The cells were observed using an inverted fluorescence microscope (manufactured by Nikon Corporation), and the number of apoptotic cells per 2.44 square millimeters was counted.
- the target molecule of lithium, GSK- 3 and inositol ⁇ Ichiru - 1-phosphate Fosufu ⁇ evening - Zeya inositol Bok - Lou poly phosphatase are known [Na t ur e, 417, 292-295 (2002 )], And insulin and forskolin are known to indirectly inhibit the activity of GSK-3 [Mol. Cell. Biol., 19, 4989-5000 (1999)].
- Lithium promotes neurogenesis of neural stem cells by inhibiting the activity of GSK-3, as lithium, insulin and forskolin are thought to share a common target molecule for the promotion of neurogenesis. It was shown that.
- a substance having an activity of selectively inhibiting GSK-3 can be used as an agent for promoting neuronal formation of neural stem cells and as a therapeutic agent for nerve regeneration.
- Example 6 9-bromo-7,12-dihydro-indolo [3,2_d] [1] benzazepine-6 (5H) -one (Kenpaullone), a GSK-3 inhibitor, promotes neurogenesis GSK-3 and cyclin dependence in a manner similar to that shown Kenpaullone [Biochem.
- CDK an inhibitor of kinase
- the DMS0 solution was dissolved in DMS0, and the DMS0 solution in a volume of 1 / 1,000 of the culture solution was added to the medium containing ANSC-7 cells, and the number of Tujl-positive neurons on day 6 after induction of differentiation was analyzed.
- the number of Tujl-positive neurons was 1.3 times higher than that of the negative control with Kenpaullone at a final concentration of 0.5 ⁇ mol / l, 2.7 times at 2 xmol / l, and 3.7 times at 5 ⁇ mol / l, depending on the concentration of Kenpaullone. Significantly increased.
- Roscovitine at a final concentration of 2 xmol / 1 was 1.0-fold compared to the negative control, 1.2-fold at 5 mol / 1, and 1.1-fold at 10.mol / l. No increase in numbers was observed. Therefore, it was clarified that Kenpaullone has a neurogenesis-promoting action, and that the action is not due to the CDK inhibitory activity of Kenpau 11 one but to the GSK-3 inhibitory activity.
- the cDNA encoding the wild-type rat GSK-3 gene was prepared as follows using the rat brain-derived cDNA described in Experimental Example 2 as type II. A primer set of 1 ⁇ ⁇ ⁇ ⁇ ⁇ ⁇ ⁇ 1 / 1 consisting of 31 SEQ ID NOS: 10 and 11 to 2.5 xl type II cDNA
- a mutant rat GSK-3 ⁇ gene in which the lysine residue at position 85, which is a mutation that loses the kinase activity of GSK-3 ⁇ , is mutated to an arginine residue [Proc. Nat. Acad. Sci. USA, 92, 8498-8502 (1995)] was similarly prepared as follows.
- the amplification reaction solution of each of the wild type and the mutant type was treated with phenol / cloth form, precipitated by adding ethanol, and purified using a QIAquick PCR purification kit (manufactured by QIAGEN) according to the attached protocol.
- the reaction solution was treated with phenolic orifice, and ethanol was added to precipitate the DNA.
- the DNA was dissolved in 431 of sterilized water.
- 5 10 of 10X Alkaline Phosphatase buffer and 21 of Alkaline Phosphatase buffer were added. (Manufactured by La Bio Inc.) and reacted at 65 ° C for 30 minutes.
- the reaction solution was treated with phenol / chloroform and ethanol was added to precipitate the DNA, which was dissolved in sterilized water.
- 3 g of the cut pCLNCX plasmid vector is mixed with 3 g of the wild-type or mutant rat GSK-3] 3 gene cDNA prepared above, and sterile water is added thereto.
- 4 l was added, and ligation high (manufactured by Toyobo Co., Ltd.) of 41 was added to react at 16 ° C for 12 hours. This was transformed into an iDH5o! Competent cell (Dakara Bio Inc.).
- the ampicillin-resistant colonies were cultured in a liquid LB medium by a standard method, and pCLNC-GSK3j3 plasmid DNA and pCLNC-GSK3 / 3 (K85R) plasmid DNA were prepared using Endofree Plasmid Maxi Kit (Qiagen) according to the attached protocol. .
- a virus vector was prepared as follows, and the functions involved in neuronal differentiation of ANSC-7 cells were analyzed.
- 2 ml of PCLNC-GSK3; 3, pCLNC-GSK3 ⁇ (K85R), or the pCLNCX plasmid vector, which is a negative control, and pMD.G plasmid vector DNA (distributed from US The cells were dissolved in D-MEM high glucose medium (Invitrogen) and transfected into 293gp cells prepared on the previous day (distributed from the US Solk Laboratories) using Transfast transfection reagent (Promega) according to the attached protocol. Fiction was performed.
- the culture supernatant was filtered with a 0.4511 m filter (Mi 11 ipore) to recover a solution containing the virus vector.
- the virus vector solution was transferred to a poly-aloma tube (manufactured by Hitachi, Ltd.) and centrifuged at 50,000 ⁇ g, 18 ° C. for 90 minutes using an ultracentrifuge (manufactured by Hitachi, Ltd.). Remove the supernatant and remove the precipitated virus from DMEM / F12 containing 2% 1 supplement, 20 ng / ml FGF-2 and 8, "g / uil hexadimethrine bromide (Sydama Aldrich).
- GSK-3 ⁇ was highly expressed and differentiated by infecting a retrovirus made from CLNC-GSK3; 3 compared to ANSC-7 cells differentiated. In cells, the number of new neurons was significantly reduced by 33%.
- Alzheimer's disease is shown to have the potential to develop by inhibiting the self-renewal ability of the brain by suppressing neurogenesis by promoting the phosphorylation of the target molecule by high expression of GSK-3) 3.
- — 3 inhibitors have been shown to be potential therapeutics for neurological disorders such as Alzheimer's disease.
- Beta-amyloid peptide (hereinafter referred to as A ⁇ ) is a major component of senile plaques and a substance thought to be the cause of Alzheimer's disease [Pro Nat. Acad. Sci., USA, 98, 11039. -11041 (2001)].
- S [1-40] (BI0S0URCE) was dissolved in 0.1% (v / v) trifluoroacetic acid (Nacalai Tesque) to make lOmg / ml .. Incubate at 25 for 1 hour, then PBS To 0.5 mg / ml. The cells were incubated at 25 ° C for 24 hours to form complexes (J. Biol.
- Alzheimer's disease one of the neurological diseases, may have the potential to develop by inhibiting the self-renewal ability of the brain by suppressing the neuronal renewal by associative A / 3.
- ANSC-7 cells of associated A3 Apoptosis-promoting effect on Analyzed.
- ANSC-7 cells induced to differentiate for 6 days with a differentiation-inducing medium containing associative type A] 3 at a final concentration of 0.1 mg / ml in the same manner as in Experimental Example 8, and a differentiation-inducing medium containing PBS as a control MSC-7 cells induced to differentiate by the above method were transformed with the cells using an in situ cell death detection kit, Fluorescein (manufactured by Roche Diagnostics) according to the attached protocol.
- the cells were observed using an inverted fluorescence microscope (manufactured by Nikon Corporation), and the number of apoptotic cells per 2.44 square millimeters was counted.
- the number of apoptosis-positive cells increased by 14% with the addition of associative A jS, but there was no significant difference. It has been revealed that suppression is mainly caused by.
- a compound having an activity of selectively inhibiting GSK-3 is a nerve regenerating agent for neurological diseases such as Alzheimer's disease, and that the compound is a neurogenesis promoting agent for neural stem cells.
- Indirubin-3, -monoxime reported as an inhibitor of GSK-3 [J. Biol. Chem., 276, 251-260 (2001), Sigma-Aldrich, Inc.] Was dissolved in DMS0 to lmmol / 1, and the culture solution was Was added to the medium containing ANSC-7 cells, and the number of Tujl-positive neurons on day 6 after induction of differentiation was analyzed. The same volume of DMS0 was added as a negative control. As a result, when Indirubin-3'-monoxime was added at a final concentration of l ⁇ mol / 1, the number of Tujl-positive neurons significantly increased 1.4-fold compared to the negative control. Therefore, it was revealed that Indirubin-3'-monoxime has a neurogenic effect.
- siRNA Short interference RNA
- siRNA_Bl consisting of SEQ ID NOS: 14 and 15
- double-stranded siRNA consisting of SEQ ID NOs: 16 and 17-B2
- Dharmacon was introduced according to the attached protocol.
- Non-specific Control Duplex IX (473 ⁇ 4GC Content) siRNA was introduced.
- the cells were suspended in 5 ml of a differentiation induction medium and inoculated on a 6-cm culture dish coated with polyorutin and laminin. 48 hours after seeding, the cells were cultivated with 1% Noni de t P_40, 50 mM Tris-HC1 (pH 7.4), 50 mM NaCl (all manufactured by Nacalai Tesque), and one tablet per 10 ml of Complete mini, EDTA free ( The product was dissolved and recovered in an aqueous solution containing Roche Diagnostics, and separated by SDS-PAGE according to a standard method, and then the amount of GSK-3 ⁇ was detected by Western blotting.
- a compound having an activity of inhibiting GSK-3 and a nucleic acid containing siRNA serve as an agent for promoting neurogenesis of neural stem cells and as a medicament for regenerative treatment of neurological diseases.
- Step 2 Synthesis of 2- (1-methylindole-1-yl) -12-oxoacetic acid methyl ester
- Reference Example 2 Isolation and culture of adult neural stem cells from rat brain
- a 7-week-old Sprague Dawley rat was put to sleep under ether anesthesia and then decapitated.
- the skull was incised from the top of the head to remove the brain.
- the tissue including the periventricular region was separated from the excised brain under a microscope using ophthalmic scissors and tweezers.
- the tissue including the periventricular region is cut into fragments of about lmm 3 using ophthalmic scissors and a scalpel, and then 2.5 U / ml papain and 250 U / ml DNase (both manufactured by Worth'ington, Freehold, NJ) 5 ml HBSS buffer containing lu / ml neutral protease (Dispase: Boehringer Maneim)
- the obtained crude cell extract was cultured on a 10-cm culture dish in a 37 ° C incubator using a DMEM / F12 medium (Invitrogen) containing 10% fetal calf serum. The next day, the medium was replaced with DMEM / F12 containing 1% N2 supplement (Invitrogen) and 2 Ong / ml FGF-2 (PeproTech), and culture was started. Once every three days, half of the medium was replaced with DMEM / F12 containing fresh 1% N2 supplement and 20 ng / ml FGF-2, and the culture was continued. When small colonies of small cells were formed, the cells were treated with 1% trypsin for about 30 seconds to 1 minute, and the detached cells were collected.
- the cells were incubated at room temperature with lO zg / ml of polyorditin (manufactured by Shidama) and at 37 ° C. with Sg / ml of mouse EHS tumor-derived laminin (Becton Dickinson). It was spread on a multi-coated culture dish (manufactured by Fisher Scientific), which had been coated, and the culture was continued. By continuing the above culture, thick small cells having small projections were concentrated. The cells were used in the above experiments as the adult neural stem cell line ANSC-7. BEST MODE FOR CARRYING OUT THE INVENTION
- a tablet having the following composition is prepared by a conventional method.
- Lithium chloride was dissolved in PBS to a concentration of 3 ⁇ 1/1 by a conventional method to prepare an agent for promoting the formation of neural stem cells that contains lithium chloride.
- Example 3 Agent for promoting neurogenesis of neural stem cells (2)
- a neurogenesis promoter for neural stem cells containing Indirubin-3'-monoxime was prepared. . Industrial Applicability
- a nerve regenerating agent comprising, as an active ingredient, a substance that inhibits the activity of Daricogen synthase kinase-3, an agent for promoting neuronal stem cell neurogenesis, comprising the substance as an active ingredient, It is possible to provide a neuron obtained by culturing a neural stem cell in the presence of the neurogenesis promoter, and a method for producing the neuron. Sequence listing free text
- SEQ ID NO: 6 Description of artificial sequence: Synthetic DNA
- SEQ ID NO: 7 Description of artificial sequence: Synthetic DNA
- SEQ ID NO: 10 Description of Artificial Sequence: Synthetic DNA ′
- SEQ ID NO: 11 Description of Artificial Sequence: Synthetic DNA
Landscapes
- Health & Medical Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Chemical & Material Sciences (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Medicinal Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Pharmacology & Pharmacy (AREA)
- Life Sciences & Earth Sciences (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Engineering & Computer Science (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Organic Chemistry (AREA)
- Biomedical Technology (AREA)
- Neurology (AREA)
- Neurosurgery (AREA)
- Epidemiology (AREA)
- Psychiatry (AREA)
- Pain & Pain Management (AREA)
- Psychology (AREA)
- Heart & Thoracic Surgery (AREA)
- Cardiology (AREA)
- Hospice & Palliative Care (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Micro-Organisms Or Cultivation Processes Thereof (AREA)
- Nitrogen Condensed Heterocyclic Rings (AREA)
- Plural Heterocyclic Compounds (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
Abstract
Description
Claims
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CA002522712A CA2522712A1 (en) | 2003-04-18 | 2004-04-16 | Nerve regenerating drug |
| US10/552,061 US20060217368A1 (en) | 2003-04-18 | 2004-04-16 | Drug for nerve regeneration |
| JP2005505483A JPWO2004091663A1 (ja) | 2003-04-18 | 2004-04-16 | 神経再生薬 |
| EP04728057A EP1645286A1 (en) | 2003-04-18 | 2004-04-16 | Drug for nerve regeneration |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2003114579 | 2003-04-18 | ||
| JP2003-114579 | 2003-04-18 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2004091663A1 true WO2004091663A1 (ja) | 2004-10-28 |
Family
ID=33296154
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2004/005503 WO2004091663A1 (ja) | 2003-04-18 | 2004-04-16 | 神経再生薬 |
Country Status (6)
| Country | Link |
|---|---|
| US (1) | US20060217368A1 (ja) |
| EP (1) | EP1645286A1 (ja) |
| JP (1) | JPWO2004091663A1 (ja) |
| CN (1) | CN1774265A (ja) |
| CA (1) | CA2522712A1 (ja) |
| WO (1) | WO2004091663A1 (ja) |
Cited By (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2004112794A3 (en) * | 2003-06-18 | 2005-06-16 | Novartis Ag | New pharmaceutical uses of staurosporine derivatives |
| WO2006077824A1 (ja) * | 2005-01-18 | 2006-07-27 | Kyoto University | 神経細胞再生のための医薬 |
| EP1757607A1 (en) | 2005-08-24 | 2007-02-28 | Molisa GmbH | N5-substituted benzo¬2,3|azepino¬4,5-b|indol-6-ones for treating tropical diseases |
| WO2008081989A1 (ja) | 2006-12-28 | 2008-07-10 | Suntory Holdings Limited | 神経再生剤 |
| US8202907B2 (en) | 2004-09-17 | 2012-06-19 | Suntory Holdings Limited | Composition with preventive or improvement effect on symptoms or diseases associated with stress-induced behavior disorders |
| US9168241B2 (en) | 2005-06-30 | 2015-10-27 | Suntory Holdings Limited | Compositions ameliorating a reduced diurnal activity and/or depressive symptoms |
| WO2016114322A1 (ja) * | 2015-01-13 | 2016-07-21 | 国立大学法人京都大学 | 筋萎縮性側索硬化症の予防及び/又は治療剤 |
| US10744146B2 (en) | 2001-08-02 | 2020-08-18 | Suntory Holdings Limited | Composition having effects of preventing or ameliorating conditions or diseases caused by brain hypofunction |
Families Citing this family (24)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| PT1542670E (pt) * | 2002-09-24 | 2013-07-11 | Suntory Holdings Ltd | Composição com efeitos na prevenção do declínio, melhoramento ou aumento de respostas normais de aptidões cognitivas de uma pessoa saudável |
| JP4522075B2 (ja) * | 2003-10-29 | 2010-08-11 | サントリーホールディングス株式会社 | 血管の老化に起因する症状あるいは疾患の予防又は改善作用を有する組成物 |
| JP2006083136A (ja) * | 2004-09-17 | 2006-03-30 | Suntory Ltd | ストレスに起因する脳機能の低下およびそれに伴う症状あるいは疾患の予防又は改善作用を有する組成物 |
| JP5697293B2 (ja) * | 2005-06-30 | 2015-04-08 | サントリーホールディングス株式会社 | 器質的脳障害に起因する高次脳機能の低下に対する改善作用を有する組成物 |
| ES2446269T3 (es) * | 2006-12-19 | 2014-03-06 | The Board Of Trustees Of The University Of Illinois | 3-Benzofuranil-4-indolil-maleimidas como potentes inhibidores de GSK-3 para trastornos neurodegenerativos |
| US20100292209A1 (en) * | 2007-03-30 | 2010-11-18 | Massachusetts Institute Of Technology | Methods for identifying compounds that modulate neurotrophic factor signaling |
| WO2008130921A1 (en) * | 2007-04-16 | 2008-10-30 | Modgene, Llc | Methods and compositions for diagnosis and treatment of depression and anxiety |
| EP2014281A1 (en) * | 2007-06-19 | 2009-01-14 | Institut National De La Sante Et De La Recherche Medicale (Inserm) | Use of inhibitors of sirtuins and/or ampk for the preparation of a medicament for the treatment of polyalanine diseases. |
| US8642334B2 (en) | 2009-02-17 | 2014-02-04 | Memorial Sloan Kettering Cancer Center | Methods of neural conversion of human embryonic stem cells |
| CN101548987B (zh) * | 2009-04-10 | 2012-08-22 | 清华大学 | 一种用于降解β淀粉样蛋白的细胞培养提取物及其制备方法和应用 |
| EP2305274A1 (en) * | 2009-09-21 | 2011-04-06 | Centre National de la Recherche Scientifique (CNRS) | Use of WNT stimulators for treating demyelinating diseases |
| US20120277218A1 (en) * | 2009-09-22 | 2012-11-01 | Neuronascent, Inc. | Methods and pharmaceutical compositions for treating down syndrome |
| EP3578988A1 (en) | 2010-05-25 | 2019-12-11 | Memorial Sloan-Kettering Cancer Center | Method of nociceptor differentiantion of human embryonic stem cells and uses thereof |
| GB201111427D0 (en) * | 2011-07-05 | 2011-08-17 | Amakem Nv | Novel bisindolylmaleimides, pan-pkc inhibitors |
| AU2012332269B2 (en) | 2011-11-04 | 2018-05-31 | Memorial Sloan-Kettering Cancer Center | Midbrain dopamine (DA) neurons for engraftment |
| WO2013142817A2 (en) | 2012-03-23 | 2013-09-26 | Dennis Brown | Compositions and methods to improve the therapeutic benefit of indirubin and analogs thereof, including meisoindigo |
| GB201217621D0 (en) * | 2012-10-02 | 2012-11-14 | Univ Leuven Kath | Anticonvulsant activity of gsk-3b inhibitors |
| WO2014176606A1 (en) | 2013-04-26 | 2014-10-30 | Memorial Sloan-Kettering Center Center | Cortical interneurons and other neuronal cells produced by the directed differentiation of pluripotent and multipotent cells |
| WO2016008966A1 (en) * | 2014-07-17 | 2016-01-21 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods for treating neuromuscular junction-related diseases |
| WO2018178194A1 (en) * | 2017-03-28 | 2018-10-04 | Institut National de la Santé et de la Recherche Médicale | Pharmaceutical compositions for use in the treatment of brain injuries or demyelinating disorders |
| CN107235966B (zh) * | 2017-05-04 | 2019-12-03 | 浙江工业大学 | 一类3,4-双吲哚-1,2,4-三氮唑酮类化合物及其制备方法和应用 |
| JP7417322B2 (ja) * | 2019-11-04 | 2024-01-18 | シーケー・リジョン・インコーポレイテッド | 神経変性疾患及び/又はその臨床的状態を抑制及び/又は治療するための組成物及び方法 |
| WO2023060268A1 (en) | 2021-10-08 | 2023-04-13 | Azora Therapeutics, Inc. | Derivatives of aryl hydrocarbon receptor agonists |
| CN114470164A (zh) * | 2022-02-28 | 2022-05-13 | 浙江卫未生物医药科技有限公司 | 一种毛囊干细胞(orsc)制剂及其在神经损伤中的应用 |
Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH02306974A (ja) * | 1989-05-05 | 1990-12-20 | Goedecke Ag | マレインイミド誘導体および医薬としてのそれらの使用 |
| JPH07508268A (ja) * | 1992-05-30 | 1995-09-14 | ゲデッケ・アクチェンゲゼルシャフト | インドロカルバゾールイミドおよびその使用 |
| WO1999042100A1 (fr) * | 1998-02-23 | 1999-08-26 | Sagami Chemical Research Center | Inhibiteurs de la mort cellulaire |
| WO2000038675A1 (en) * | 1998-12-23 | 2000-07-06 | Smithkline Beecham Plc | Treatment of conditions with a need for the inhibition of gsk-3 |
| WO2001013916A1 (fr) * | 1999-08-20 | 2001-03-01 | Sagami Chemical Research Center | Medicaments inhibant la mort cellulaire |
| WO2001037819A2 (fr) * | 1999-11-23 | 2001-05-31 | Centre National De La Recherche Scientifique (C.N.R.S.) | Utilisation de derives d'indirubine pour la fabrication de medicaments |
| WO2002062387A1 (en) * | 2001-02-07 | 2002-08-15 | Smithkline Beecham P.L.C. | Treatment of conditions with a need of gsk-3 inhibition |
Family Cites Families (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5753506A (en) * | 1996-05-23 | 1998-05-19 | Cns Stem Cell Technology, Inc. | Isolation propagation and directed differentiation of stem cells from embryonic and adult central nervous system of mammals |
| WO2002000261A2 (en) * | 2000-06-26 | 2002-01-03 | Monsanto Technology Llc | Surfactant-containing formulations for extended release of somatotropin |
| CA2452596C (en) * | 2001-07-05 | 2010-12-21 | Takeda Chemical Industries, Ltd. | Benzo-fused 5-membered heterocyclic compounds, their production and use |
| US20040247525A1 (en) * | 2003-01-23 | 2004-12-09 | Rene Hen | Brain progenitor cell-based assay, and related methods and compositions |
-
2004
- 2004-04-16 EP EP04728057A patent/EP1645286A1/en not_active Withdrawn
- 2004-04-16 CN CNA200480010384XA patent/CN1774265A/zh active Pending
- 2004-04-16 US US10/552,061 patent/US20060217368A1/en not_active Abandoned
- 2004-04-16 JP JP2005505483A patent/JPWO2004091663A1/ja not_active Abandoned
- 2004-04-16 CA CA002522712A patent/CA2522712A1/en not_active Abandoned
- 2004-04-16 WO PCT/JP2004/005503 patent/WO2004091663A1/ja not_active Application Discontinuation
Patent Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH02306974A (ja) * | 1989-05-05 | 1990-12-20 | Goedecke Ag | マレインイミド誘導体および医薬としてのそれらの使用 |
| JPH07508268A (ja) * | 1992-05-30 | 1995-09-14 | ゲデッケ・アクチェンゲゼルシャフト | インドロカルバゾールイミドおよびその使用 |
| WO1999042100A1 (fr) * | 1998-02-23 | 1999-08-26 | Sagami Chemical Research Center | Inhibiteurs de la mort cellulaire |
| WO2000038675A1 (en) * | 1998-12-23 | 2000-07-06 | Smithkline Beecham Plc | Treatment of conditions with a need for the inhibition of gsk-3 |
| WO2001013916A1 (fr) * | 1999-08-20 | 2001-03-01 | Sagami Chemical Research Center | Medicaments inhibant la mort cellulaire |
| WO2001037819A2 (fr) * | 1999-11-23 | 2001-05-31 | Centre National De La Recherche Scientifique (C.N.R.S.) | Utilisation de derives d'indirubine pour la fabrication de medicaments |
| WO2002062387A1 (en) * | 2001-02-07 | 2002-08-15 | Smithkline Beecham P.L.C. | Treatment of conditions with a need of gsk-3 inhibition |
Non-Patent Citations (2)
| Title |
|---|
| CHEN G. ET AL: "Enhancement of Hippocampal Neurogenesis by Lithium", JOURNAL OF NEUROCHEMISTRY, vol. 75, 2000, pages 1729 - 1734, XP002951304 * |
| LEOST M. ET AL: "Paullones are potent inhibitors of glycogen synthase kinase-3beta and cyclin-dependent kinase 5/p25", EUR. J. BIOCHEM., vol. 267, 2000, pages 5983 - 5994, XP000981256 * |
Cited By (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US10744146B2 (en) | 2001-08-02 | 2020-08-18 | Suntory Holdings Limited | Composition having effects of preventing or ameliorating conditions or diseases caused by brain hypofunction |
| WO2004112794A3 (en) * | 2003-06-18 | 2005-06-16 | Novartis Ag | New pharmaceutical uses of staurosporine derivatives |
| US8575146B2 (en) | 2003-06-18 | 2013-11-05 | Novartis Ag | Pharmaceutical uses of staurosporine derivatives |
| US8202907B2 (en) | 2004-09-17 | 2012-06-19 | Suntory Holdings Limited | Composition with preventive or improvement effect on symptoms or diseases associated with stress-induced behavior disorders |
| US8367729B2 (en) | 2004-09-17 | 2013-02-05 | Suntory Holdings Limited | Composition with preventive or improvement effect on symptoms or diseases associated with stress-induced behavior disorders |
| WO2006077824A1 (ja) * | 2005-01-18 | 2006-07-27 | Kyoto University | 神経細胞再生のための医薬 |
| US9168241B2 (en) | 2005-06-30 | 2015-10-27 | Suntory Holdings Limited | Compositions ameliorating a reduced diurnal activity and/or depressive symptoms |
| EP1757607A1 (en) | 2005-08-24 | 2007-02-28 | Molisa GmbH | N5-substituted benzo¬2,3|azepino¬4,5-b|indol-6-ones for treating tropical diseases |
| US8383677B2 (en) | 2006-12-28 | 2013-02-26 | Suntory Holdings Limited | Nerve-regenerating agent |
| EP3593797A1 (en) | 2006-12-28 | 2020-01-15 | Suntory Holdings Limited | Nerve-regenerating agent |
| WO2008081989A1 (ja) | 2006-12-28 | 2008-07-10 | Suntory Holdings Limited | 神経再生剤 |
| WO2016114322A1 (ja) * | 2015-01-13 | 2016-07-21 | 国立大学法人京都大学 | 筋萎縮性側索硬化症の予防及び/又は治療剤 |
| JPWO2016114322A1 (ja) * | 2015-01-13 | 2017-10-19 | 国立大学法人京都大学 | 筋萎縮性側索硬化症の予防及び/又は治療剤 |
| CN107530430A (zh) * | 2015-01-13 | 2018-01-02 | 国立大学法人京都大学 | 用于预防和/或治疗肌萎缩性侧索硬化症的药剂 |
Also Published As
| Publication number | Publication date |
|---|---|
| CA2522712A1 (en) | 2004-10-28 |
| US20060217368A1 (en) | 2006-09-28 |
| JPWO2004091663A1 (ja) | 2006-07-06 |
| CN1774265A (zh) | 2006-05-17 |
| EP1645286A1 (en) | 2006-04-12 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2004091663A1 (ja) | 神経再生薬 | |
| JP6352368B2 (ja) | トリプトファン代謝によって媒介される免疫抑制の阻害剤 | |
| Saraswati et al. | Glycogen synthase kinase-3 and its inhibitors: Potential target for various therapeutic conditions | |
| EP3125883B1 (en) | Indole derivatives for use in medicine | |
| US10590086B2 (en) | Pharmaceutical compound | |
| EP3182972B1 (en) | Pharmaceutical compound | |
| KR20190137843A (ko) | 단백질 인산화효소 억제제로서의 1h-이미다조[4,5-h]퀴나졸린 화합물 | |
| CN101014345A (zh) | 三环酰苯胺螺环内酰胺cgrp受体拮抗剂 | |
| IL255726A (en) | Compounds that inhibit the notch signaling pathway | |
| CA2804069C (en) | Pyrazoloquinolines | |
| JP2010528016A (ja) | 細胞を刺激するための方法および組成物 | |
| KR20130106367A (ko) | Dna-pk 저해제로서의 이미다조[4,5-c]퀴놀린 | |
| ES2930106T3 (es) | Compuesto derivado de pirrol-piridina, procedimiento para preparar el mismo y composición farmacéutica que contiene el mismo como ingrediente activo para la prevención o el tratamiento de enfermedades relacionadas con la proteína quinasa | |
| KR102338568B1 (ko) | 퀴나졸린 유도체 | |
| US11944625B2 (en) | Calmodulin inhibitors, Chk2 inhibitors and RSK inhibitors for the treatment of ribosomal disorders and ribosomapathies | |
| CA3189181A1 (en) | Inhibitors of sarm1 | |
| CA2358724C (fr) | Nouveaux derives de cyclobutene-dione, leur procede de preparation et les compositons pharmaceutiques qui les contiennent | |
| CA2923765A1 (en) | Stem cell modulation ii | |
| EP2866803A1 (en) | Psoralen derivatives for preventing or treating heart failure or cardiac hypertrophy | |
| CN101006055B (zh) | 作为酪蛋白激酶Iε抑制剂的3-取代的-5-和6-氨基烷基吲哚-2-羧酸酰胺以及相关类似物 | |
| US20220073880A1 (en) | Natural killer cells | |
| CN105793421A (zh) | 耐性突变型90kDa热休克蛋白质 | |
| KR20070102671A (ko) | 항종양 활성을 갖는 인돌 유도체 | |
| WO2013168807A1 (ja) | 移植細胞懸濁液用の添加剤及び治療用組成物 | |
| WO2023193809A1 (zh) | Sarm1抑制剂化合物、包含其的药物组合物及其制备方法和用途 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BW BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EC EE EG ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NA NI NO NZ OM PG PH PL PT RO RU SC SD SE SG SK SL SY TJ TM TN TR TT TZ UA UG US UZ VC VN YU ZA ZM ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): BW GH GM KE LS MW MZ SD SL SZ TZ UG ZM ZW AM AZ BY KG KZ MD RU TJ TM AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IT LU MC NL PL PT RO SE SI SK TR BF BJ CF CG CI CM GA GN GQ GW ML MR NE SN TD TG |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 2006217368 Country of ref document: US Ref document number: 10552061 Country of ref document: US |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2005505483 Country of ref document: JP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2004810384X Country of ref document: CN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2522712 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2004728057 Country of ref document: EP |
|
| WWP | Wipo information: published in national office |
Ref document number: 2004728057 Country of ref document: EP |
|
| WWP | Wipo information: published in national office |
Ref document number: 10552061 Country of ref document: US |
|
| WWW | Wipo information: withdrawn in national office |
Ref document number: 2004728057 Country of ref document: EP |