WO2004063283A1 - 光機能材料 - Google Patents
光機能材料 Download PDFInfo
- Publication number
- WO2004063283A1 WO2004063283A1 PCT/JP2004/000267 JP2004000267W WO2004063283A1 WO 2004063283 A1 WO2004063283 A1 WO 2004063283A1 JP 2004000267 W JP2004000267 W JP 2004000267W WO 2004063283 A1 WO2004063283 A1 WO 2004063283A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- group
- photoelectric conversion
- substituted
- dyes
- sensitizing dye
- Prior art date
Links
Classifications
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01G—CAPACITORS; CAPACITORS, RECTIFIERS, DETECTORS, SWITCHING DEVICES OR LIGHT-SENSITIVE DEVICES, OF THE ELECTROLYTIC TYPE
- H01G9/00—Electrolytic capacitors, rectifiers, detectors, switching devices, light-sensitive or temperature-sensitive devices; Processes of their manufacture
- H01G9/20—Light-sensitive devices
- H01G9/2059—Light-sensitive devices comprising an organic dye as the active light absorbing material, e.g. adsorbed on an electrode or dissolved in solution
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F9/00—Compounds containing elements of Groups 5 or 15 of the Periodic System
- C07F9/02—Phosphorus compounds
- C07F9/28—Phosphorus compounds with one or more P—C bonds
- C07F9/38—Phosphonic acids RP(=O)(OH)2; Thiophosphonic acids, i.e. RP(=X)(XH)2 (X = S, Se)
- C07F9/3804—Phosphonic acids RP(=O)(OH)2; Thiophosphonic acids, i.e. RP(=X)(XH)2 (X = S, Se) not used, see subgroups
- C07F9/3826—Acyclic unsaturated acids
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F9/00—Compounds containing elements of Groups 5 or 15 of the Periodic System
- C07F9/02—Phosphorus compounds
- C07F9/28—Phosphorus compounds with one or more P—C bonds
- C07F9/38—Phosphonic acids RP(=O)(OH)2; Thiophosphonic acids, i.e. RP(=X)(XH)2 (X = S, Se)
- C07F9/3804—Phosphonic acids RP(=O)(OH)2; Thiophosphonic acids, i.e. RP(=X)(XH)2 (X = S, Se) not used, see subgroups
- C07F9/3839—Polyphosphonic acids
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F9/00—Compounds containing elements of Groups 5 or 15 of the Periodic System
- C07F9/02—Phosphorus compounds
- C07F9/28—Phosphorus compounds with one or more P—C bonds
- C07F9/38—Phosphonic acids RP(=O)(OH)2; Thiophosphonic acids, i.e. RP(=X)(XH)2 (X = S, Se)
- C07F9/40—Esters thereof
- C07F9/4003—Esters thereof the acid moiety containing a substituent or a structure which is considered as characteristic
- C07F9/4015—Esters of acyclic unsaturated acids
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F9/00—Compounds containing elements of Groups 5 or 15 of the Periodic System
- C07F9/02—Phosphorus compounds
- C07F9/28—Phosphorus compounds with one or more P—C bonds
- C07F9/38—Phosphonic acids RP(=O)(OH)2; Thiophosphonic acids, i.e. RP(=X)(XH)2 (X = S, Se)
- C07F9/40—Esters thereof
- C07F9/4071—Esters thereof the ester moiety containing a substituent or a structure which is considered as characteristic
- C07F9/409—Compounds containing the structure P(=X)-X-acyl, P(=X) -X-heteroatom, P(=X)-X-CN (X = O, S, Se)
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F9/00—Compounds containing elements of Groups 5 or 15 of the Periodic System
- C07F9/02—Phosphorus compounds
- C07F9/547—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom
- C07F9/553—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom having one nitrogen atom as the only ring hetero atom
- C07F9/5537—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom having one nitrogen atom as the only ring hetero atom the heteroring containing the structure -C(=O)-N-C(=O)- (both carbon atoms belong to the heteroring)
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F9/00—Compounds containing elements of Groups 5 or 15 of the Periodic System
- C07F9/02—Phosphorus compounds
- C07F9/547—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom
- C07F9/553—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom having one nitrogen atom as the only ring hetero atom
- C07F9/572—Five-membered rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F9/00—Compounds containing elements of Groups 5 or 15 of the Periodic System
- C07F9/02—Phosphorus compounds
- C07F9/547—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom
- C07F9/553—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom having one nitrogen atom as the only ring hetero atom
- C07F9/572—Five-membered rings
- C07F9/5728—Five-membered rings condensed with carbocyclic rings or carbocyclic ring systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F9/00—Compounds containing elements of Groups 5 or 15 of the Periodic System
- C07F9/02—Phosphorus compounds
- C07F9/547—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom
- C07F9/553—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom having one nitrogen atom as the only ring hetero atom
- C07F9/576—Six-membered rings
- C07F9/60—Quinoline or hydrogenated quinoline ring systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F9/00—Compounds containing elements of Groups 5 or 15 of the Periodic System
- C07F9/02—Phosphorus compounds
- C07F9/547—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom
- C07F9/553—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom having one nitrogen atom as the only ring hetero atom
- C07F9/576—Six-membered rings
- C07F9/64—Acridine or hydrogenated acridine ring systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F9/00—Compounds containing elements of Groups 5 or 15 of the Periodic System
- C07F9/02—Phosphorus compounds
- C07F9/547—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom
- C07F9/645—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom having two nitrogen atoms as the only ring hetero atoms
- C07F9/6503—Five-membered rings
- C07F9/65031—Five-membered rings having the nitrogen atoms in the positions 1 and 2
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F9/00—Compounds containing elements of Groups 5 or 15 of the Periodic System
- C07F9/02—Phosphorus compounds
- C07F9/547—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom
- C07F9/645—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom having two nitrogen atoms as the only ring hetero atoms
- C07F9/6509—Six-membered rings
- C07F9/650952—Six-membered rings having the nitrogen atoms in the positions 1 and 4
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F9/00—Compounds containing elements of Groups 5 or 15 of the Periodic System
- C07F9/02—Phosphorus compounds
- C07F9/547—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom
- C07F9/645—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom having two nitrogen atoms as the only ring hetero atoms
- C07F9/6509—Six-membered rings
- C07F9/6512—Six-membered rings having the nitrogen atoms in positions 1 and 3
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F9/00—Compounds containing elements of Groups 5 or 15 of the Periodic System
- C07F9/02—Phosphorus compounds
- C07F9/547—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom
- C07F9/6527—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom having nitrogen and oxygen atoms as the only ring hetero atoms
- C07F9/653—Five-membered rings
- C07F9/65324—Five-membered rings condensed with carbocyclic rings or carbocyclic ring systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F9/00—Compounds containing elements of Groups 5 or 15 of the Periodic System
- C07F9/02—Phosphorus compounds
- C07F9/547—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom
- C07F9/6527—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom having nitrogen and oxygen atoms as the only ring hetero atoms
- C07F9/6533—Six-membered rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F9/00—Compounds containing elements of Groups 5 or 15 of the Periodic System
- C07F9/02—Phosphorus compounds
- C07F9/547—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom
- C07F9/6536—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom having nitrogen and sulfur atoms with or without oxygen atoms, as the only ring hetero atoms
- C07F9/6539—Five-membered rings
- C07F9/6541—Five-membered rings condensed with carbocyclic rings or carbocyclic ring systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F9/00—Compounds containing elements of Groups 5 or 15 of the Periodic System
- C07F9/02—Phosphorus compounds
- C07F9/547—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom
- C07F9/6536—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom having nitrogen and sulfur atoms with or without oxygen atoms, as the only ring hetero atoms
- C07F9/6544—Six-membered rings
- C07F9/6547—Six-membered rings condensed with carbocyclic rings or carbocyclic ring systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F9/00—Compounds containing elements of Groups 5 or 15 of the Periodic System
- C07F9/02—Phosphorus compounds
- C07F9/547—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom
- C07F9/655—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom having oxygen atoms, with or without sulfur, selenium, or tellurium atoms, as the only ring hetero atoms
- C07F9/6552—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom having oxygen atoms, with or without sulfur, selenium, or tellurium atoms, as the only ring hetero atoms the oxygen atom being part of a six-membered ring
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F9/00—Compounds containing elements of Groups 5 or 15 of the Periodic System
- C07F9/02—Phosphorus compounds
- C07F9/547—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom
- C07F9/655—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom having oxygen atoms, with or without sulfur, selenium, or tellurium atoms, as the only ring hetero atoms
- C07F9/6552—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom having oxygen atoms, with or without sulfur, selenium, or tellurium atoms, as the only ring hetero atoms the oxygen atom being part of a six-membered ring
- C07F9/65522—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom having oxygen atoms, with or without sulfur, selenium, or tellurium atoms, as the only ring hetero atoms the oxygen atom being part of a six-membered ring condensed with carbocyclic rings or carbocyclic ring systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F9/00—Compounds containing elements of Groups 5 or 15 of the Periodic System
- C07F9/02—Phosphorus compounds
- C07F9/547—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom
- C07F9/6553—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom having sulfur atoms, with or without selenium or tellurium atoms, as the only ring hetero atoms
- C07F9/655345—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom having sulfur atoms, with or without selenium or tellurium atoms, as the only ring hetero atoms the sulfur atom being part of a five-membered ring
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F9/00—Compounds containing elements of Groups 5 or 15 of the Periodic System
- C07F9/02—Phosphorus compounds
- C07F9/547—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom
- C07F9/6553—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom having sulfur atoms, with or without selenium or tellurium atoms, as the only ring hetero atoms
- C07F9/655345—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom having sulfur atoms, with or without selenium or tellurium atoms, as the only ring hetero atoms the sulfur atom being part of a five-membered ring
- C07F9/655354—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom having sulfur atoms, with or without selenium or tellurium atoms, as the only ring hetero atoms the sulfur atom being part of a five-membered ring condensed with carbocyclic rings or carbocyclic ring systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F9/00—Compounds containing elements of Groups 5 or 15 of the Periodic System
- C07F9/02—Phosphorus compounds
- C07F9/547—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom
- C07F9/6558—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom containing at least two different or differently substituted hetero rings neither condensed among themselves nor condensed with a common carbocyclic ring or ring system
- C07F9/65583—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom containing at least two different or differently substituted hetero rings neither condensed among themselves nor condensed with a common carbocyclic ring or ring system each of the hetero rings containing nitrogen as ring hetero atom
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F9/00—Compounds containing elements of Groups 5 or 15 of the Periodic System
- C07F9/02—Phosphorus compounds
- C07F9/547—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom
- C07F9/6558—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom containing at least two different or differently substituted hetero rings neither condensed among themselves nor condensed with a common carbocyclic ring or ring system
- C07F9/65586—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom containing at least two different or differently substituted hetero rings neither condensed among themselves nor condensed with a common carbocyclic ring or ring system at least one of the hetero rings does not contain nitrogen as ring hetero atom
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F9/00—Compounds containing elements of Groups 5 or 15 of the Periodic System
- C07F9/02—Phosphorus compounds
- C07F9/547—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom
- C07F9/6561—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom containing systems of two or more relevant hetero rings condensed among themselves or condensed with a common carbocyclic ring or ring system, with or without other non-condensed hetero rings
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09B—ORGANIC DYES OR CLOSELY-RELATED COMPOUNDS FOR PRODUCING DYES, e.g. PIGMENTS; MORDANTS; LAKES
- C09B23/00—Methine or polymethine dyes, e.g. cyanine dyes
- C09B23/10—The polymethine chain containing an even number of >CH- groups
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M14/00—Electrochemical current or voltage generators not provided for in groups H01M6/00 - H01M12/00; Manufacture thereof
- H01M14/005—Photoelectrochemical storage cells
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01G—CAPACITORS; CAPACITORS, RECTIFIERS, DETECTORS, SWITCHING DEVICES OR LIGHT-SENSITIVE DEVICES, OF THE ELECTROLYTIC TYPE
- H01G9/00—Electrolytic capacitors, rectifiers, detectors, switching devices, light-sensitive or temperature-sensitive devices; Processes of their manufacture
- H01G9/20—Light-sensitive devices
- H01G9/2027—Light-sensitive devices comprising an oxide semiconductor electrode
- H01G9/2031—Light-sensitive devices comprising an oxide semiconductor electrode comprising titanium oxide, e.g. TiO2
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E10/00—Energy generation through renewable energy sources
- Y02E10/50—Photovoltaic [PV] energy
- Y02E10/542—Dye sensitized solar cells
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02P—CLIMATE CHANGE MITIGATION TECHNOLOGIES IN THE PRODUCTION OR PROCESSING OF GOODS
- Y02P70/00—Climate change mitigation technologies in the production process for final industrial or consumer products
- Y02P70/50—Manufacturing or production processes characterised by the final manufactured product
Definitions
- the present invention relates to an optical functional material.
- This optical functional material can be used as a photoelectric conversion material, a light emitting material or a light absorbing material. Furthermore, the present invention relates to a sensitizing dye for photoelectric conversion, a photoelectric conversion material, a photoelectric conversion electrode, and a photoelectric conversion cell using the same, using the optical functional material.
- single crystal silicon solar cells polycrystalline silicon solar cells, amorphous silicon solar cells, compound solar cells such as telluride power domium and indium copper selenide are in practical use or are subject to research and development.
- amorphous silicon solar cells compound solar cells such as telluride power domium and indium copper selenide are in practical use or are subject to research and development.
- problems such as high manufacturing cost, difficulty in securing raw materials, and long energy payback time.
- many solar cells using organic materials for increasing the area and cost have been proposed so far, but they have problems such as low conversion efficiency and poor durability.
- This is a so-called dye-sensitized solar cell in which a dye is fixed to the surface of a porous titanium oxide thin film, and more specifically, a titanium oxide porous thin layer spectrally sensitized by a ruthenium complex dye is used as a working electrode
- a dye-sensitized photoelectric conversion cell comprising an electrolyte layer mainly composed of iodine and a counter electrode.
- the first advantage of this method is that it can provide an inexpensive photoelectric conversion device because it uses an inexpensive oxide semiconductor such as titanium oxide, and the second advantage is the ruthenium complex dye used. Since they have wide absorption in the visible light range, they have relatively high conversion efficiency.
- non-ruthenium complex dyes include phenylxanthene dyes, phthalocyanine dyes, coumarin dyes, cyanine dyes, porphyrin dyes, azo dyes and the like. These organic dyes are expected to have high photoelectric conversion efficiency because they have a large absorption coefficient and a large degree of freedom in molecular design compared to ruthenium complexes.
- organic sensitizing dyes because the light absorbing region of the dye is narrow and the charge injection to titanium oxide is inefficient.
- a sensitizing dye having a substituted acrylic acid site has been developed as a sensitizing dye characterized by its adsorption end with titanium oxide, and it has been shown that it has a relatively high conversion efficiency.
- the advantage of this sensitizing dye is that the charge injection efficiency from the sensitizing dye to an inorganic semiconductor such as titanium oxide is improved by combining the dye skeleton with a substituted acrylic acid site.
- the adsorptive power of the sulfoxyl group is weak.
- the present invention relates to an optical functional material having a vinyl phosphonic acid group.
- an optical functional material having a chemical structure represented by the following general formula (1) is preferably used.
- R 1 and R 2 each independently represent a hydrogen atom or a monovalent organic residue
- M 1 and M 2 each independently represent a hydrogen atom
- R 1 and R 2 , R 1 and X, and R 2 and X respectively represent They may be combined with each other to form a ring
- X and R 2 may be interchanged.
- Another present invention relates to a sensitizing dye for photoelectric conversion, which comprises the optical functional material according to the present invention.
- Still another invention of the present invention relates to a photoelectric conversion material comprising: an inorganic semiconductor; and the sensitizing dye for photoelectric conversion according to the invention connected to the inorganic semiconductor.
- Yet another aspect of the present invention relates to a photoelectric conversion electrode including a transparent electrode, and the photoelectric conversion material according to the present invention laminated on the transparent electrode.
- Still another present invention relates to a photoelectric conversion cell including the photoelectric conversion electrode according to the present invention, an electrolyte layer, and a conductive counter electrode.
- FIG. 1 is a cross-sectional view schematically showing a test sample in an example as an example of a photoelectric conversion cell.
- FIG. 2 represents a proton NMR spectrum.
- the upper part shows the ethyl ester form of compound (3 0), and the lower part shows compound (3 0) obtained by hydrolyzing it.
- FIG. 3 shows the IV characteristics of the photoelectric conversion cell using the compound (73).
- FIG. 4 shows an IPCE spectrum of a photoelectric conversion cell using a compound (7 3).
- the optical function material of the present invention is characterized by having a vinyl phosphonic acid group. That is, the optical functional material of the present invention is not particularly limited as long as it has a vinylphosphonic acid group, but in particular, a compound having a chemical structure represented by the following general formula (1) is preferable .
- X represents a monovalent organic residue
- R 1 and R 2 each independently represent a hydrogen atom or a monovalent organic residue
- M 1 and M 2 each independently represent a hydrogen atom
- R 1 and R 2 , R 1 and X, and R 2 and X are And each may combine with each other to form a ring
- X and R 2 may be interchanged.
- the photofunctional material is a new sensitizing effect, heat generation effect, coloring effect, color fading effect, luminous effect, phase change effect, photoelectric conversion effect, photo-optical effect, photocatalytic effect, by absorbing light. It means a material that exhibits functions such as light modulation effect, optical recording effect, and radical generation effect, or conversely, materials having a light emitting function by receiving these effects.
- the optical functional material is, for example, a photoelectric conversion material, a light emitting material, an optical recording material, an image forming material, a photochromic material, an elector luminescence material, a photoconductive material, a dichroic material, a radical generating material, an acid generating material, a base generating Materials, phosphorescent materials, nonlinear optical materials, second harmonic generation materials, third harmonic generation materials, photosensitive materials, light absorbing materials, near infrared absorbing materials, photochemical hole burning materials, light sensing materials, optical materials Widely used as king materials, sensitizing materials for photochemical treatment, optical phase change recording materials, photosintered recording materials, photomagnetic recording materials, dyes for photodynamic therapy, sensitizing dyes for photoelectric conversion, etc. Can be.
- an optical functional material having a vinyl phosphonic acid group particularly having a chemical structure of the general formula (1)
- this optical functional material As a typical application form, it may be referred to as a sensitizing dye for photoelectric conversion or simply as a sensitizing dye, but the broad application described above is not denied.
- the functions required for the sensitizing dye for photoelectric conversion include that the dye has a wide absorption region and that charges can be efficiently injected into an inorganic semiconductor such as titanium oxide.
- an organic residue which widens the absorption region to X in the above general formula (1) In order to widen the absorption region, X is preferably an electron donating organic residue, and an organic residue having an amino group or the like can exhibit a high effect.
- the sensitizing dye In order to inject charges efficiently, the sensitizing dye needs an phanka group to be adsorbed on the surface of the inorganic semiconductor, but since the photofunctional material of the present invention has a phosphonic acid group, it can be used as a sensitizing dye If you meet this condition.
- the phosphonic acid group has a stronger adsorptive ability to an inorganic semiconductor such as titanium oxide than a carboxylic acid group, so that the detachment of the dye is less likely to occur, and therefore, it is expected to prolong the life of the device. it can.
- one phosphorus atom has two acidic groups (-OM 1 and - ⁇ _M 2), by each of the addition effect and chelating effect, it is possible to exert a strong adsorption effect.
- the adsorption rate at the time of adsorbing the dye to the inorganic semiconductor electrode at the time of element production becomes faster, and there is also an advantage that the manufacturing time can be shortened.
- a strong chemical bond is generated between the anchor and the inorganic semiconductor surface, and the electron clouds overlap with each other effectively, so that rapid electron transfer from the sensitizing dye to the inorganic semiconductor surface can be expected.
- the solubility in water, ethanol, etc. is also improved, and it becomes possible to use these solvents with a small environmental load as adsorption solvents, and these become pots as dye solutions. It is expected that it will lead to a long life, which in turn will lead to the effect of lowering the manufacturing cost.
- a substituent can be introduced at the position of R 1 .
- the biphenyl phosphonic acid group becomes stronger and the charge transfer in the molecule becomes more efficient. This is very effective in that charge injection from the sensitizing dye to the inorganic semiconductor such as titanium oxide can be performed more efficiently.
- the vinyl phosphonic acid group has a chemical structure that makes it possible, for the first time, to place an electron withdrawing group in the vicinity of the phosphonic acid while connecting the 7T electron conjugated structure of the chromophoric moiety to the bonding position of the phosphonic acid.
- an electron withdrawing group can not be introduced in the vicinity, and charge injection is more efficient than vinyl phosphonic acid group. Poor efficiency and low photoelectric conversion efficiency.
- a vinylphosphonic acid structure for example, a chemical structure represented by the general formula (1)
- a sensitizing dye having one anchored group can be realized.
- X in the general formula (1) represents a monovalent organic residue.
- the organic residue as referred to herein is not particularly limited. For example, it may be substituted (that is, substituted or unsubstituted) monovalent aromatic hydrocarbon residue, substituted or unsubstituted Monovalent heterocyclic residue, substituted or unsubstituted monovalent aliphatic unsaturated hydrocarbon residue, substituted or unsubstituted monovalent amine And substituted or unsubstituted monovalent organometallic complex residues.
- the aromatic ring of the aromatic hydrocarbon residue is not particularly limited, and examples thereof include benzene, naphthalene, anthracene, naphthacene, pyrene, phenanthrene, indene, azulene, perylene, fluorene, biphenyl and terphenyl.
- the heterocycle of the heterocycle residue is not particularly limited.
- heterocycles may be quaternized and may have a counter ion.
- the counter ion in this case is not particularly limited, and may be a general anion. Examples include halogen ions, perchlorate ions, boron tetrafluoride ions, phosphorus hexafluoride ions, ⁇ oxide ions, methanesulfonic acid ions, toluenesulfonic acid ions. When it does not have a counter ion, it may be neutralized with an acidic group such as an intramolecular or intermolecular carpoxyl group.
- the heterocyclic ring includes dye skeletons used for dyes and pigments.
- dye skeletons used for dyes and pigments.
- the dye skeleton to be used for example, azo dyes, quinacridone dyes, diketopyrrole dyes, squarylium dyes, cyanine dyes, cyanine dyes, merocyanine dyes, trifenylmethane dyes, xanthene dyes, porphyrin dyes Dyes, chlorophyll dyes, ruthenium complex dyes, indigo dyes, perylene dyes, dioxazine dyes, anthraquinone dyes, phthalocyanine dyes, and naphthalene dyes are listed.
- the aliphatic unsaturated hydrocarbon residue is not particularly limited, and examples thereof include a vinyl group, a 1,3-butadienyl group and a 1,3,5-hexatrienyl group, and the like.
- the sum of saturated bonds is preferably in the range of 1 to 20.
- the amino group is not particularly limited, and examples thereof include an amino group, a mono- or dialkylamino group, a mono- or diarylamino group and the like, and specific examples thereof include N-methylamino group, N-acetylamino group, N, N- Cetylamino group, N, N-diisopropylamino group, N, N- dibutylamino group, N- benzylamino group, N, N- dibenzylamino group, N- phenylamino group, N, N- diphenylamino group, N, N- bis (m —Tolyl) amino group, N, N-bis (p-tolyl) amino group, N, N-bis (p-biphenylyl) amino group.
- the organometallic complex of the organometallic complex residue is not particularly limited, and examples thereof include ferrocene, ruthenocene, titanocene, zirconocene, phthalocyanine, naphthenic cyanine, porphyrin and ruthenium bipyridyl complex.
- the substituent is not particularly limited, and, for example, an alkyl group, a aryl group, a heterocyclic group, an alkoxyl group, an acyl group, an aryloxy group, an alkylthio group, an arylthio group, a substituted or unsubstituted amino group, a substituted group Or an unsubstituted amido group, an alkoxyalkyl group, an alkoxyalkyl group, an aryloxy carbonyl group, an aromaticoxy group, a sulfoxy group, a sulfo group, a phosphonic acid group, a cyano group, an isosyano group, a thiosyanate group, an isothiocyanate group, A nitro group, a nitrosyl group, a halogen atom, and a hydroxyl group are mentioned.
- alkyl group a C1-C30 substituted or unsubstituted linear, branched, and cyclic hydrocarbon group is mentioned.
- aryl group examples include the above-mentioned aromatic ring of the aromatic hydrocarbon residue, and these aryl groups may further have a substituent.
- the heterocyclic group includes the heterocyclic ring of the aforementioned heterocyclic residue. These heterocyclic groups may further have a substituent.
- alkoxyl group include alkoxyl groups having 1 to 20 carbon atoms, such as methoxy, ethoxy, propoxy, butoxy, tert-butoxy, alkoxy and tert-alkoxy.
- Examples of the asyl group include an alkylcarponyl group and an arylcarponyl group, and specific examples thereof include an acyl group having a carbon number of 1 to 20, such as an acetyl group, a propionyl group, a benzoyl group and a toluoyl group.
- aryloxy group examples include aryloxy groups having 6 to 20 carbon atoms, such as phenyloxy, 4-tert-butylphenoxy, 1-naphthyloxy, 2-naphthyloxy, and 9-anthryloxy groups.
- alkylthio group examples include alkylthio groups having 1 to 20 carbon atoms, such as a methylthio group, a phenylthio group, a tert-butylthio group, a hexylthio group, and an octylthio group.
- arylthio group examples include arylthio groups having 6 to 20 carbon atoms, such as phenylthio group, 2-methylphenylthio group, 41-tert-peptylphenylthio group and the like.
- Examples of the substituted or unsubstituted amino group include the aforementioned monovalent amino group which may have a substituent.
- amido group an amido group, an alkyl amido group, an aromatic amido group is mentioned, for example.
- alkoxyalkyl group examples include alkoxyalkyl groups having 1 to 20 carbon atoms such as methoxymethyl group, ethoxymethyl group and isopropoxymethyl group.
- alkyloxycarponyl group examples include alkoxycarbonyl group having 1 to 20 carbon atoms, such as methoxycarbonyl group, ethoxycarpyl group, ter t-butoxycarponyl group and the like.
- aryloxy carbonyl group there can be mentioned an alkoxycarboxyl group having 5 to 30 carbon atoms such as a phenyloxy carponyl group and a naphthyloxy carponyl group.
- the acidic group such as a dynamic propoxy group, a phosphonic acid group or a sulfo group may form a metal salt or an ammonium salt.
- substituents bonded to X described above may be the same or different from each other, and the substituents may be bonded to each other to form a ring. Furthermore, X and X, and a substituent linked to X may be linked to R 1 and R 2 described later to form a ring.
- X is a monovalent organic residue having a substituted or unsubstituted amino group, and examples thereof include a dialkylaminophenyl group, a dialkylamino phenyl group and a dialkylaminostyryl group. Can be mentioned. Furthermore, in order to have high photoelectric conversion efficiency, X is a monovalent organic residue having a substituted or unsubstituted amino group, and has a long conjugated chain, and the conjugated chain has a rigid skeleton. Some are more preferable.
- the light absorbing region of the dye is broadened by having a long conjugated chain, and an electron donating substitution such as an amino group is made to the vinyl phosphonic acid group which is a charge injection site to the inorganic semiconductor. This is because intramolecular charge transfer from the donor site to the partial position of the acceptor can efficiently occur by binding to a rigid unit having a group.
- R 1 and R 2 independently represents a hydrogen atom or a monovalent organic residue.
- the organic residue as referred to herein is not particularly limited.
- the same organic residue as the above X substituted or unsubstituted alicyclic hydrocarbon residue, substituted or unsubstituted chain hydrocarbon residue Groups, hydroxyl groups, electron withdrawing groups.
- the cyclic hydrocarbon of the substituted or unsubstituted cyclic hydrocarbon residue is, for example, a saturated cyclic hydrocarbon having 3 to 20 carbon atoms, such as cyclohexane, cyclopentane, etc., a cyclic hexene, cyclopentene Examples thereof include unsaturated cyclic hydrocarbons having 3 to 30 carbon atoms such as cyclohexene and cyclopentene.
- Examples of the chain hydrocarbon group of the substituted or unsubstituted chain hydrocarbon residue include linear or branched alkyl groups having 1 to 30 carbon atoms, and these chain carbon hydrogen groups are not It may have a saturated bond.
- the electron withdrawing group means a group having a Hammett's substituent constant of which value is larger than zero.
- substituents are not particularly limited.
- a cyano group, an amino group, a nitro group, an acyl group, an alkyloxy carponyl group, an aryloxy carbonyl group, an alkylsulfonyl group, an arylsulfonyl group, a substituted or substituted group examples thereof include non-substituted amido group, perfluoroalkyl group, perfluoroalkylthio group, perfluoroalkylcarponyl group, substituted or unsubstituted sulfonamide group, 4-cyanophenyl group, and halogen atom.
- the ⁇ value described in Chem. Rev. 91, 165-195, 1991 is 0. There are bigger ones.
- an acyl group an alkyloxy carponyl group, an aryloxy carponyl group, and a substituted or unsubstituted amido group, exemplified as a substituent in the organic residue represented by X
- substituent in the organic residue represented by X examples thereof include the same as the substituted silyl group, alkyloxy carponyl group, aryloxy carponyl group, substituted or unsubstituted amido group.
- alkylsulfonyl group examples include a mesyl group, a methylsulfonyl group, a propylsulfonyl group and the like.
- arylsulfonyl group examples include benzenesulfonyl group, toluenesulfonyl group and the like.
- perfluoroalkyl group examples include a trifluoromethyl group and a pentafluoroethyl group.
- the perfluoroalkylthio group includes a trifluoromethylthio group, a pentafluorothiolthio group and the like.
- perfluoroalkylcarbonyl group examples include a trifluoroacetyl group and a penfluorofluorethyl carponyl group.
- Examples of the substituted or unsubstituted sulfonamide group include sulfonamide group, dimethylaminosulfonyl group, getylaminosulfonyl group, and diphenylaminosulfone. And the like.
- RR 2 described above may be bonded to each other to form a ring, or RR 2 may be bonded to X to form a ring.
- R 1 and Z or R 2 have a substituent, those substituents may form a bond with each other, and the substituent may be bonded to a substituent of or X or It may form a ring.
- R 1 is preferably an electron-suction bow I-like group. This is because the electron withdrawing group is bonded to the vinyl phosphonic acid group which is an inorganic semiconductor adsorption site and the charge injection site, and thus the biphenyl phosphonic acid group becomes a stronger electron axepeption group. . This enables efficient charge injection to the inorganic semiconductor.
- R 1 is more preferably a cyano group. This is because the cyano group is a strong electron withdrawing group and has high stability.
- M 1 and M 2 each independently represent a hydrogen atom, a substituted or unsubstituted alkyl group, a substituted or unsubstituted aryl group, a substituted or unsubstituted silyl group, or a cation.
- alkyl group refers to a linear, branched or cyclic hydrocarbon group having 1 to 20 carbon atoms, and these hydrocarbon groups may have an unsaturated bond.
- alkyl groups preferable ones include a methyl group, a methyl group, an isopropyl group, a t-butyl group, a benzyl group and the like.
- aryl group examples include aromatic hydrocarbon residues among the organic residues represented by the above X, furan, thiophen, pyrrolyl, oxazolyl, isooxazole, thiazole, isothiazolyl, imidazole and pyrazole And heteroaromatic ring groups such as furazan, pyridine, pyridazine, pyrimidine, pyrazine, indole, benzofuran, benzothiophene, quinoline, carbazole, acridine, xanthene, phenothiazine, phenoxazine and the like. Of these aryl groups, preferred are phenyl and tolyl.
- silyl groups include alkyl silyl groups and aryl silyl groups. Examples thereof include: .trimethylsilyl group, triethylsilyl group and trifenylsilyl group.
- alkyl group, aryl group and silyl group may be optionally substituted by one or more substituents, and examples of the substituent include the same as the substituents to X described above.
- the cation is not particularly limited as long as it forms a salt with phosphonic acid, and examples thereof include metal ions such as lithium, sodium, potassium, magnesium and calcium, tetraptyalmonium, There are quaternary ammonium ions such as pyridinium and imidazolium.
- M 1 and M 2 are preferably a hydrogen atom or a quaternary ammonium salt, but even if MM 2 is other than these, It can be used without problems.
- M 1 and M 2 may be in the form of a phosphonic acid ester such as an alkyl group or a silyl group, and when a phosphonic acid ester is adsorbed to an inorganic semiconductor, an appropriate catalyst or the like may be used. It can also be adsorbed while being hydrolyzed in the system.
- the compound represented by the general formula (1) has a double bond, and thus can take structural isomers such as cis form and trans form, but the steric structure is not particularly limited, and any of them is for example, for photoelectric conversion It can be used favorably as a sensitizing dye. That is, in the formula (1), X and R 2 may be interchanged. For example, in the relationship between R 1 and R 2 , arbitrary geometric isomers can be selected in either cis or trans.
- the compounds represented by the general formula (1) can be synthesized, for example, by a method as shown in the following scheme (1).
- ethanol tetrahydrofuran and the like can be used, but it is not particularly limited thereto.
- Organic Reactions Volume 15, Chapter 2, 196 7 (19 7 8
- the solvents described in (Annual Reprint) may be used. If the reaction does not proceed well, it may be effective to use the reaction without using a solvent.
- the reaction temperature is usually room temperature, but may be heated for reaction as required.
- the reaction may be difficult to progress.
- a methyl group may be substituted for the hydrogen atom in MM 2.
- the desired compound can be obtained by introducing and reacting an ethyl group or the like, and hydrolyzing the obtained compound.
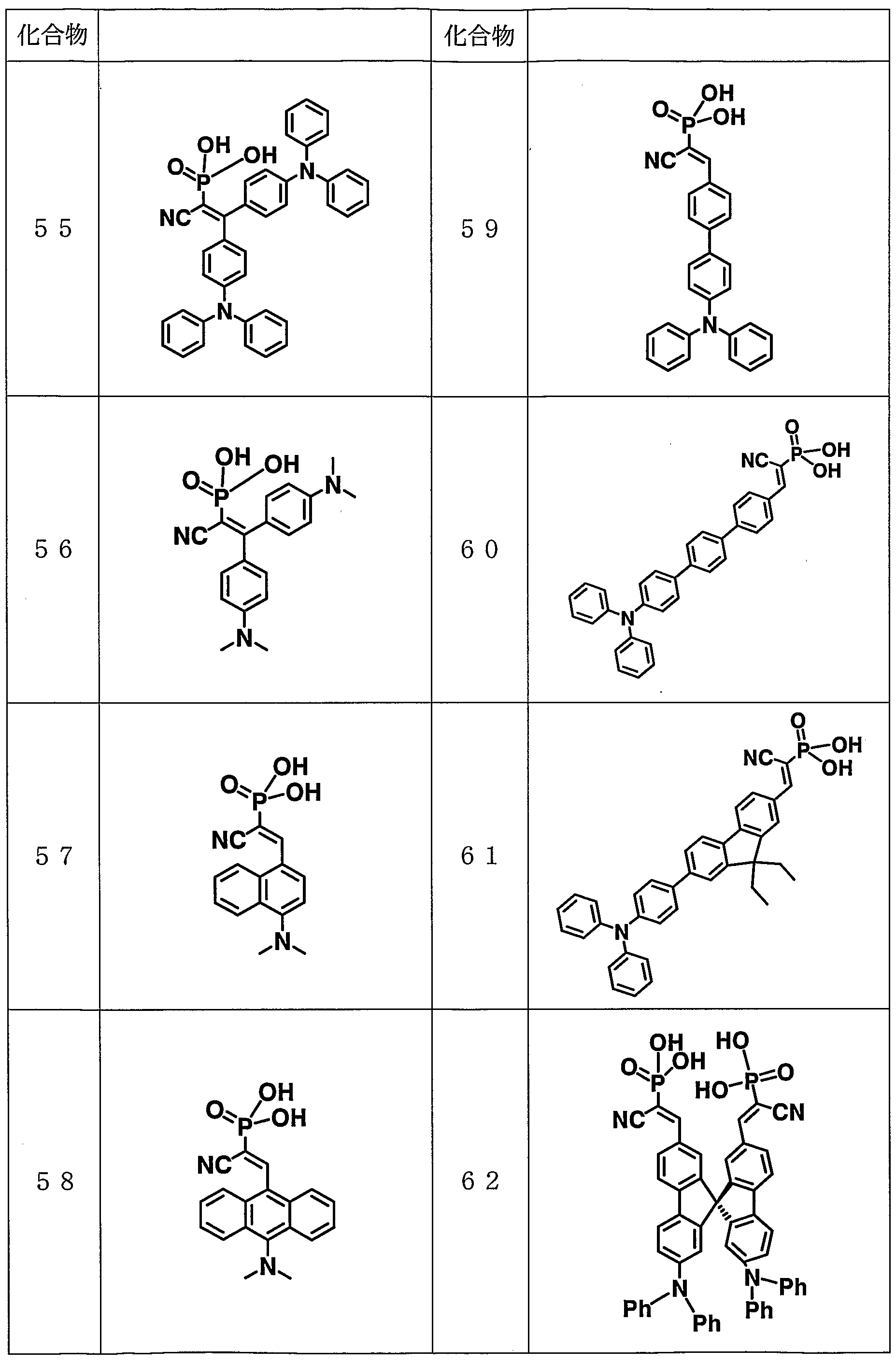
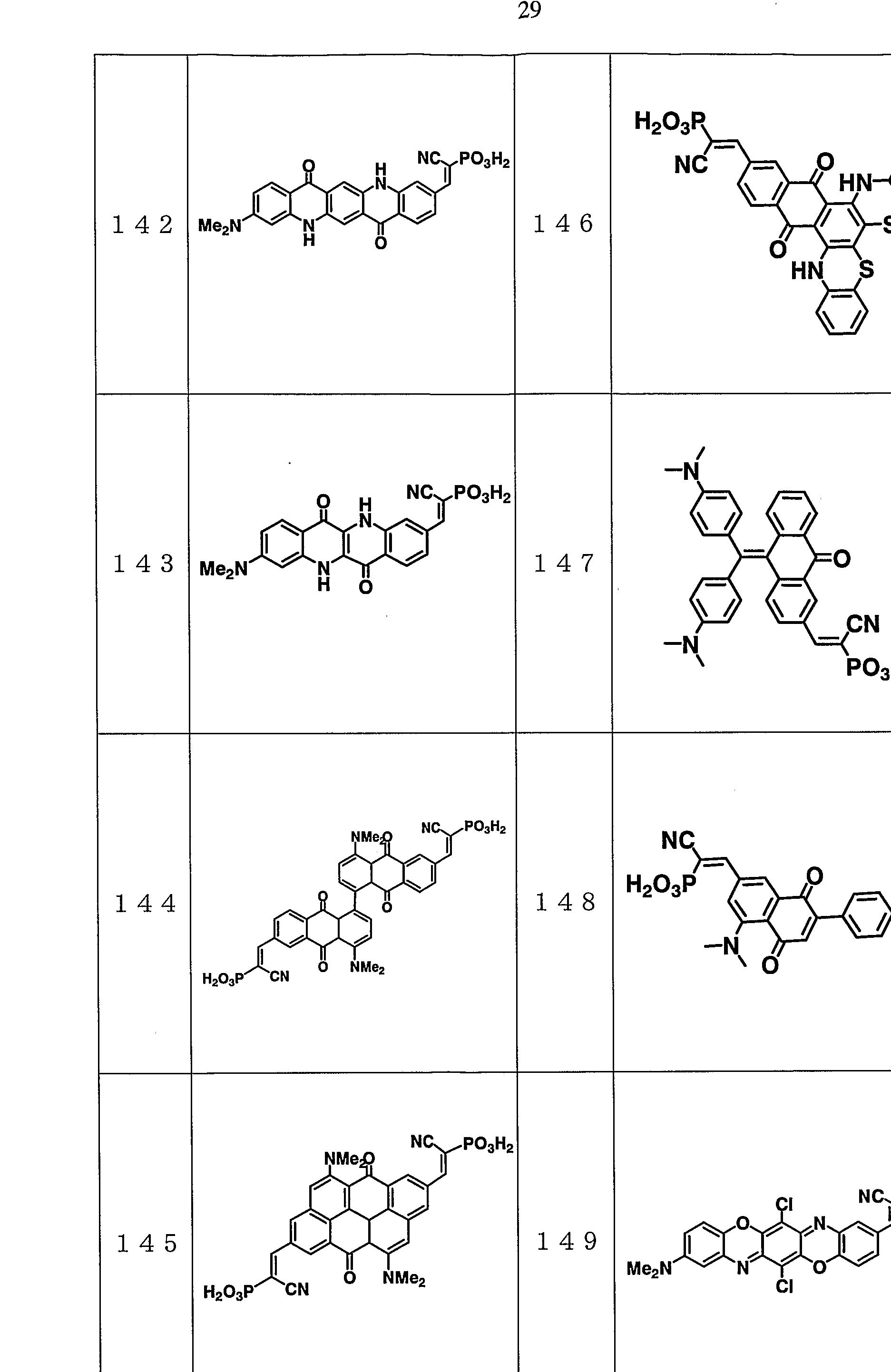
- representative examples of compounds that can be used as the optical functional material of the present invention will be shown, but the present invention is not limited to these.
- the sensitizing dye for photoelectric conversion according to the present invention includes one or more of the above-described optical functional materials according to the present invention, and the photofunctional material having a vinyl phosphonic acid group such as the general formula (1) has a cover
- One or more other light functional materials that is, having no vinyl phosphonic acid group
- the sensitizing dyes represented by the general formula (1) or the like may be used alone or in combination of two or more, and may be used in combination with one or more other sensitizing dyes.
- the blending ratio of the sensitizing dye for photoelectric conversion according to the present invention in combination with other sensitizing dyes there is no particular limitation. It is preferable to use 0.1 to 100 mol of a dye, and it is more preferable to use 0.1 to 10 mol.
- sensitizing dyes include, for example, azo dyes, quinacridone dyes, diketopyrrolopyrrole dyes, squarylium dyes, cyanine dyes, merocyanine dyes, trifenylmethane dyes, xanthene dyes, porphyrin dyes Chlorophyll dyes, ruthenium complex dyes, indigo dyes, perylene dyes, dioxazine dyes, anthraquinone dyes, phthalocyanine dyes, naphtha port cyanine dyes, and derivatives thereof.
- these sensitizing dyes have a functional group capable of being linked to the surface of the inorganic semiconductor in their structure. The reason is that the excited electrons of the photoexcited color can be rapidly transmitted to the conduction band of the inorganic semiconductor.
- the functional group as mentioned herein include a sulfoxyl group, a hydroxy group, a hydroxamic acid group, a sulfonic acid group, a phosphonic acid group, and a phosphinic acid group, and the like.
- the substituent is not limited to these, as long as it is a substituent having a role of rapidly transmitting excited electrons of the metal to the conduction band of the inorganic semiconductor.
- a photoelectric conversion material, a photoelectric conversion electrode, and a photoelectric conversion cell according to the present invention obtained using the above-described sensitizing dye for photoelectric conversion according to the present invention will be described including materials other than the sensitizing dye.
- the photoelectric conversion material in which the inorganic semiconductor is sensitized that is, the inorganic semiconductor and the increased number of linked inorganic semiconductors.
- a photoelectric conversion material containing a dye is obtained.
- linking means that the inorganic semiconductor and the sensitizing dye are chemically or physically bonded, and includes, for example, that both are bonded by adsorption.
- each of a linking group, an anchor group, P and an attachment group is used as a term indicating a group having an equivalent function.
- Inorganic semiconductors generally have a photoelectric conversion function for light in a part of the region, but by connecting a sensitizing dye to this surface, the photoelectric conversion to visible light and Z or near infrared light regions is possible. Conversion is possible.
- a material of the inorganic semiconductor although an inorganic oxide is mainly used, it is not limited to this as long as it is an inorganic semiconductor having a photoelectric conversion function by linking sensitizing dyes.
- silicon, germanium, a group III-V group semiconductor, a metal chalcogenide and the like can be mentioned.
- the conduction band of the inorganic oxide be present at a position where it is easy to receive electrons from the photoexcitation order of the sensitizing dye.
- titanium oxide, tin oxide, zinc oxide, niobium oxide and the like are particularly preferably used.
- titanium oxide is particularly preferably used in terms of price and environmental hygiene.
- These inorganic semiconductors may be used alone or in combination of two or more of them.
- the above inorganic semiconductor is preferably made porous and used as an inorganic semiconductor porous body.
- the inorganic semiconductor porous body has a large surface area by making it porous so that a large amount of sensitizing dye can be linked to its surface, and it can have highly efficient photoelectric conversion ability.
- a method for making porous there is widely known a method in which inorganic oxide particles such as titanium oxide having a particle diameter of several nanometers to several tens of nanometers are sintered and then sintered. The method is not limited to this as long as it is a method of obtaining a large surface area by quality.
- a photoelectric conversion electrode that is, a photoelectric conversion electrode including a transparent electrode and a photoelectric conversion material laminated on the transparent electrode is formed.
- a transparent electrode is usually a conductive layer formed on the surface of a transparent substrate, that is, a conductive surface of a transparent substrate having a conductive surface.
- the conductive surface (transparent electrode) to be used is not particularly limited as long as it is a conductive material which absorbs less light in the visible to near-infrared region of sunlight, but IT ⁇ (indium monotin oxide), tin oxide (fluorine Metal oxides with good conductivity such as zinc oxide, zinc oxide and the like are preferable. Since the sheet resistance (surface resistance) of the substrate (transparent substrate having a conductive surface) is preferably as low as possible, specifically, 20 ⁇ ⁇ ⁇ ( ⁇ / sq.) Or less, the conductive layer is preferably It is preferable to have a thickness corresponding to that.
- the transparent substrate to be used is not particularly limited as long as it is a material that absorbs less light in the visible to near infrared region of sunlight.
- Glass base materials such as quartz, plain glass, glass 7, lead glass, etc .; polyethylene terephthalate, polyethylene naphthalate, polyimid, polyester, polyethylene, polycarbonate, polyvinyl peptate, polypropylene, tetraacetyl cellulose, syndioxide
- resin base materials such as tic polystyrene, polyphenyl rusulfide, polyarylate, polysulfone, polyester sulfone, polyether imide, cyclic polyolefin, brominated fenoxy, chlorinated pinyl and the like can be used.
- a method of laminating a photoelectric conversion material on the conductive surface of a transparent base material having a conductive surface for example, after applying inorganic oxide particles pasted on the conductive surface, it is dried or sintered to obtain an inorganic oxide semiconductor porous material.
- an inorganic oxide semiconductor porous material By forming a body and immersing it in a solution in which the sensitizing dye is dissolved, per each transparent substrate, to utilize the affinity between the porous surface of the inorganic oxide semiconductor and the anchor group of the sensitizing dye to increase
- a method of binding a dye to its porous surface can be mentioned as a general method, it is not limited to this method.
- the inorganic oxide particles may be dispersed in water or an appropriate organic solvent in order to pasteurize the inorganic oxide particles. It is important to prepare a paste with good dispersibility to be laminated as a homogeneous, large surface area inorganic porous material, so if necessary, an acid such as nitrate, acetylacetone, polyethylene glycol, Triton X-1 0 0 And the like are preferably mixed with the paste component and made into a paste using a paint shaker or the like.
- an acid such as nitrate, acetylacetone, polyethylene glycol, Triton X-1 0 0 And the like are preferably mixed with the paste component and made into a paste using a paint shaker or the like.
- a coating method by spin coating As a method of applying the paste to the conductive surface of the transparent substrate, a coating method by spin coating, a screen printing method, a coating method using a squeegee, a dipping method, a spraying method, a coating method, etc. are used. .
- the applied inorganic oxide paste is dried or fired to remove volatile components in the paste, thereby forming an inorganic oxide semiconductor porous body on the conductive surface of the transparent substrate.
- a condition of drying or baking for example, a method of giving thermal energy of about 30 minutes to 1 hour at a temperature of 40.degree. To 500.degree. C., for example, is generally used.
- an alcohol solvent such as ethanol and benzyl alcohol as a solvent
- a nitrile solvent such as acetoditril and propiolodiuril
- Halogen solvents such as benzene in the mouth
- ether solvents such as jetyl ether and tetrahydrofuran
- ester solvents such as ethyl acetate and butyl acetate
- ketone solvents such as acetone, methyl ethyl ketone and cyclohexanone
- Carbonate solvents such as jetyl carbonate and propylene carbonate
- Hydrocarbon solvents such as hexane, octane, benzene and toluene; dimethylformamide, dimethylacetoamide, dimethylsulfoxide, 1,
- the conditions for immersing the inorganic semiconductor porous body in the solution in which the sensitizing dye is dissolved are not particularly limited, and may be suitably set so as to obtain desired photoelectric conversion efficiency. It is preferable that the temperature be about room temperature to about 80 ° C.
- the film thickness of the inorganic semiconductor porous body formed on the conductive surface of the transparent substrate is desirably about 0.5 to 20 m. If the film thickness is less than this range, effective conversion efficiency may not be obtained. On the other hand, if the film thickness is thicker than this range, As a result, it becomes difficult to form a film, for example, peeling occurs, and the distance between the surface layer of the inorganic semiconductor porous body and the conductive surface becomes long, so that the generated charges can not be effectively transmitted to the conductive surface. Conversion efficiency may be difficult to obtain. 3. Photoelectric conversion cell
- a photoelectric conversion cell that is, a photoelectric conversion cell including a photoelectric conversion electrode, an electrolyte layer, and a conductive counter electrode by combining the photoelectric counter electrode obtained as described above with the conductive counter electrode through the electrolyte layer.
- the electrolyte layer preferably comprises an electrolyte, a medium, and an additive.
- electrolyte 1 2 and L i I as iodide
- iodide example, N a I, KI, C s I, M g I 2, C a I 2, C u I, tetraalkyl ammonium Niu Muyo one iodide
- organic molten salt compound refers to an ion pair compound consisting of an organic cation and an inorganic or organic anion and having a melting point of room temperature or less.
- examples of aromatic cations include N-methyl-N'-ethylimidazolium cation, N-methyl-N, -n-propylimidazolium cation, N —Methyl—N′—n—Hexylimidazolium cation and other N-alkyl N′-alkyldiimidazolium cations; N—Hexyl pyridinium cation, N—Butyl pyridinium cation and other N-alkyl pyridinium And mu cations.
- Aliphatic cations include aliphatic cations such as N, N, N-trimethyl-N-propyl ammonium cations and cyclic aliphatic cations such as N, N-methyl pyrrolidinium.
- salt Halide ions such as fluoride ion, bromide ion and iodide ion, phosphorus hexafluoride ion, boron tetrafluoride ion, methane trifluoride fluoride, perchlorate ion, hypochlorite ion, chlorate ion, sulfuric acid Inorganic anions such as ion and phosphate ion; Amide such as bis (trifluoromethylsulfonyl) imide and imido type anions.
- iodides, bromides and the like can be used alone or in combination of two or more.
- an electrolyte obtained by mixing a combination of I 2 and an iodide, for example, I 2 and L i I, pyridinium iodide, or imidazolium iodide is preferably used, but is not limited thereto.
- Preferred electrolyte concentration is 1 2 0 in the medium. 0 1-0.
- a 5 M, iodides and Z or bromide and the like (in the case of more mixtures thereof) is 0. 1 to 1 5 M hereinafter It is.
- the medium used for the electrolyte layer is preferably a compound that can exhibit good ion conductivity.
- liquid media include ether compounds such as dioxan and jetyl ether; ethylene daryl dialkyl ethers, propylene daryl dialkyl ethers, polyethylene glycol dialkyl ethers, linear ethers such as polypropylene glycol dialkyl ethers; methanol, Alcohols such as ethanol, ethylene glycol monoalkyl ether, propylene glycol monoalkyl ether, polyethylene glycol monoalkyl ether, polypropylene glycol monoalkyl ether, etc .; ethylene glycol, propylene glycol, polyethylene glycol, polypropylene glycol, glycerin etc.
- Polyhydric alcohols Acetonitrile, Darutur rogetril, Metexia setonitri , Propionic nitrile, nitrile compounds such as benzonitrile, ethylene carbonate, forces one Poneto compounds such as propylene force one Poneto Heterocyclic compounds such as 3-methyl-2-oxazolidinone; non-proton polar substances such as dimethyl sulfoxide, sulfolane, water, etc. can be used. These may be used alone or in combination of two or more.
- the liquid medium can also contain a polymer.
- a polymer such as polyacrylonitrile or polyvinylidene fluoride is added to the liquid medium, or a polyfunctional monomer having an ethylenically unsaturated group is polymerized in the liquid medium to form a solid medium.
- Cul Cul, Cu SCN (these compounds are P-type semiconductors that do not require a liquid medium and act as electrolytes), etc., Nature, Volume 95, 583-5 8 5 pages (19.98.1 Oct. 08) (the disclosure of which is incorporated herein by reference).
- Hole transport materials such as methoxyphenylamine) -9,9'-spiropifluorene can be used.
- additives may be added to the electrolyte layer for the purpose of improving the durability and the electrical output of the photoelectric conversion cell.
- inorganic salts such as magnesium iodide may be added for the purpose of improving durability
- amines such as t_butylpyridine, 2-picoline, 2,6-lutidine, etc. for the purpose of improving output; deoxycholic acid, etc.
- Monosaccharides such as glucose, darcosamine, glucuronic acid and their sugar alcohols; disaccharides such as maltose; linear oligosaccharides such as raffinose; cyclic oligosaccharides such as cyclodextrin; Hydrolyzed oligosaccharides can also be added.
- the thickness of the electrolyte layer to be formed is not particularly limited, but it is preferable that the thickness be such that the conductive counter electrode is not in direct contact with the inorganic semiconductor layer to which the dye is adsorbed. Specifically, it is preferably about 0.1 to about L 00 m. (Conductive counter electrode)
- the conductive counter electrode functions as the positive electrode of the photoelectric conversion cell.
- conductive materials used for the counter electrode include metals (platinum, gold, silver, copper, aluminum, rhodium, indium, etc.), metal oxides (ITO (indium-tin oxide), tin oxide (fluorine, etc.) Etc.), zinc oxide etc.), carbon etc. may be mentioned.
- the film thickness of the counter electrode is not particularly limited, but is preferably 5 nm or more and 10 m or less.
- a photoelectric conversion cell is formed by combining the photoelectric conversion electrode and the conductive counter electrode through the electrolyte layer. If necessary, seal around the photoelectric conversion cell to prevent leakage and volatilization of the electrolyte layer.
- a thermoplastic resin, a photocurable resin, a glass frit or the like can be used as a sealing material.
- the photoelectric conversion cell can be formed by connecting small-area photoelectric conversion cells as needed. For example, the electromotive voltage can be increased by combining photoelectric conversion cells in series.
- P-Dimethylaminobenzenesaldehyde 10. 0. 0 g (67 mmo 1), 3.0 g (74 mmo 1) of ketomethyl phosphonate, piperidine 0. 1 g into ethanol of 200 m 1 After stirring for 5 hours at room temperature, the solvent was evaporated under reduced pressure to give an orange solid. The crude compound thus obtained was purified by silica gel column chromatography to obtain 1 5. 8 g of [1 -cyano-2-(4 -dimethylamino-phenyl) -vinyl]-jettyl Yield 7 6%).
- FIG. 2 shows a proton NMR spectrum of the obtained compound (30).
- the upper part shows the spectrum of the ethyl ester of compound (30), and the lower part shows the spectrum of compound (30) obtained by hydrolyzing it.
- Synthesis Example 4 Synthesis Method of Compound (73)
- reaction solution was slowly returned to room temperature and stirred for additional 2 hours, then a saturated aqueous solution of ammonium chloride was added to the reaction solution and stirred, and the organic layer in the reaction solution was extracted twice with 5 O ml of toluene.
- Synthesis Example 5 Synthesis Method of Compound (74) A mixture of 10.0 g (72 mmol) of isophorone, 2.8 g (72 mmo 1) of jetyl cyanomethylphosphonate, and 0.77 g (1 Ommo 1) of ammonium acetate at 100 ° C. in a nitrogen stream. Stir for 5 hours. After completion of the reaction, the reaction solution is heated under reduced pressure to remove unreacted starting materials, and then subjected to silica gel chromatography, and [Sigano- (3,5,5-trimethyl-cyclohexyl--2-enylidene] is obtained. 13.4 g of a) -methyl] -gettyl phosphonate was obtained (yield 70%).
- FIG. 1 showing a test sample of the photoelectric conversion cell. The description will be made with reference to.
- a fluorine-doped tin oxide layer (transparent electrode layer) 31 and a glass substrate 51 were used.
- Fluorine-doped tin oxide layer 32 attached glass substrate 52 (Asahi Glass Co., Ltd., type U— A conductive counter electrode was used in which a platinum layer (platinum electrode layer) 4 (150 nm thick) was laminated on the conductive layer 32 of TCTC) by sputtering. Preparation of titanium oxide paste
- Titanium oxide (P25 manufactured by Nippon Aerosil Co., Ltd. P21 particle diameter 21 nm) 6 parts Water (adjusted to pH 2 by adding nitric acid) 14 parts acetylacetalone 0.6 part surfactant (ITN Corporation Tr iton X- 100) 0. 04
- a 60 m thick mending tape is placed on the conductive surface of the transparent electrode (transparent electrode layer 31), and a 1 cm square tape is removed to make a mask, and a few drops of the above-mentioned acid titanium paste are made on the vacant part. After cooking, the excess paste was removed with a squeegee. After air drying, all the masks were removed, and firing was performed in an oven at 450 ° C. for 1 hour to obtain a titanium oxide electrode having a titanium oxide porous layer with an effective area of 1 cm 2 . Adsorption of sensitizing dyes
- the sensitizing dye for photoelectric conversion is dissolved in ethanol or water (concentration: 0.6 mmo 1 / L), the insoluble matter is removed with a membrane filter if necessary, and the above-mentioned titanium oxide electrode is immersed in this dye solution, Or, if necessary, heat and leave it for several hours to several days. Immersion time was set up so that the conversion efficiency was maximized by actually creating cells to obtain the conversion efficiency.
- the electrolyte solution of the following formulation was prepared.
- a solvent methoxyacetonitrile was used.
- test samples of the photoelectric conversion cell were assembled. That is, the above-mentioned transparent electrode (glass substrate 51 with fluorine-doped tin oxide layer 31) having the titanium oxide porous layer 1 adsorbed with the sensitizing dye for photoelectric conversion as described above, and fluorine-doped And a conductive counter electrode in which a platinum layer 4 is laminated on a conductive layer of a glass substrate 52 with a tin oxide layer 32 and a spacer 61, 62 made of resin film (Mitsui 'Dupont Polychemical' HIMIRAN 'film ( The electrolyte solution was injected into the gap to form an electrolyte solution layer 2.
- Conductors 71 and 72 for measuring conversion efficiency were fixed to the glass substrates 51 and 52, respectively. Measurement method of conversion efficiency
- a combination of an ORI EL Solar Shimley Yuichi (# 8116) and an air mass filter is adjusted to a light quantity of 10 OmW / cm 2 with a actinometer to form a measurement light source, and the test sample of the photoelectric conversion cell is irradiated with light.
- I-V curve characteristics were measured using an EKO Seiki-made I-V power butler (MP 160).
- the conversion efficiency 7? was calculated according to the following equation using Vo c (open circuit voltage value), I sc (short circuit current value), and ff (fill factor value) obtained from the I 1 V curve characteristic measurement.
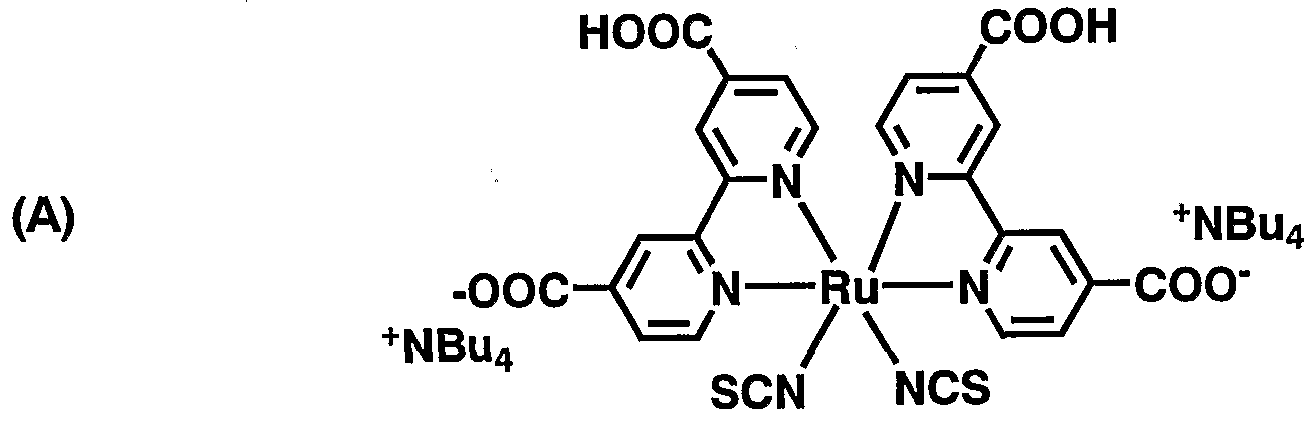
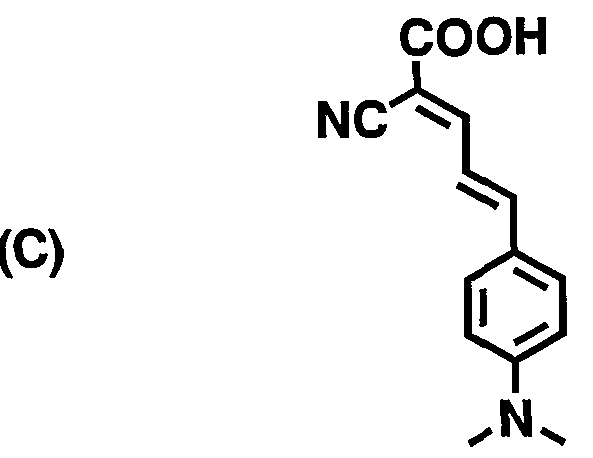
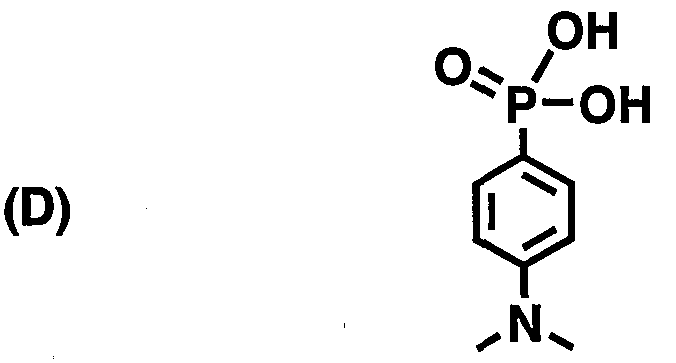
- Compounds (A) to (D) in Table 4 are compounds having the following structures, respectively:
- (A) is a ruthenium complex dye
- compounds (B) and (C) are the compounds described in WO 02Z1 1213 pamphlet (in the structural formula, the substituent of N described by N ⁇ represents a methyl group).
- (D) is p-dimethylamino benzene phosphonic acid.
- Example 4 to 6 and Comparative Examples 2 to 4 and 8 to 10 the substrate after dye adsorption was further immersed in a predetermined aqueous solution to determine the presence or absence of dye detachment and the photoelectric conversion efficiency.
- Example 1 1 1. 7 E t OH Example 2 4 2. 3 ", E t OH Example 3 30 3.2, ⁇ E t OH Example 4 30 3.2 2 ⁇ 5 AE t OH Example 5 30 3. 1 ⁇ 7 AE t OH
- the diffuse reflectance spectrum of the substrate was measured before cell assembly. Those with no decrease in absorbance by the given aqueous solution treatment were evaluated as A, those with a slight decrease were evaluated as B, and those with a decrease were evaluated as C.
- the current-voltage characteristics of the photoelectric conversion cell of Example 8 using the compound (7 3) are shown in FIG. 3, and the IPCE spectrum of the photoelectric conversion cell of the same Example 8 is shown in FIG. The wavelength dependence of the rate of conversion to the child is shown respectively.
- the present invention uses a specific chemical structure, that is, a vinyl phosphonic acid group as an adsorption terminal to an inorganic semiconductor, and according to the present invention, a sensitizing dye having this vinyl phosphonic acid group
- a good photoelectric conversion cell can be formed by connecting the organic semiconductor to the surface of the inorganic semiconductor laminated on the transparent conductive substrate.
- This sensitizing dye can exhibit a stronger adsorptive power than a dye having a carboxylic acid end by having a vinyl phosphonic acid group, so it is stable with high photoelectric conversion efficiency and strong adsorptive power with the interface of the inorganic semiconductor. It can be expected to function as a sensitizing dye for photoelectric conversion.
Description
Claims
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2005508016A JP4341621B2 (ja) | 2003-01-16 | 2004-01-16 | 光機能材料 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2003-008519 | 2003-01-16 | ||
| JP2003008519 | 2003-01-16 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2004063283A1 true WO2004063283A1 (ja) | 2004-07-29 |
Family
ID=32709164
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2004/000267 WO2004063283A1 (ja) | 2003-01-16 | 2004-01-16 | 光機能材料 |
Country Status (3)
| Country | Link |
|---|---|
| JP (1) | JP4341621B2 (ja) |
| TW (1) | TWI318638B (ja) |
| WO (1) | WO2004063283A1 (ja) |
Cited By (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2006120939A1 (ja) * | 2005-05-13 | 2006-11-16 | Agc Seimi Chemical Co., Ltd. | 新規アミノ基含有複素環誘導体および該複素環誘導体を含有する光電変換用増感色素 |
| JP2008044852A (ja) * | 2006-08-10 | 2008-02-28 | Tokyo Institute Of Technology | シクロペンテン骨格を有する有機リン化合物及びその製造方法 |
| WO2008056567A1 (fr) * | 2006-11-07 | 2008-05-15 | Agc Seimi Chemical Co., Ltd. | Nouveau composé aromatique contenant un groupe amino, et colorant de sensibilisation pour conversion photoélectrique, contenant le composé aromatique |
| WO2013008889A1 (ja) * | 2011-07-14 | 2013-01-17 | 積水化学工業株式会社 | 光電変換素子用材料、光電変換素子の製造方法及び光電変換素子 |
| WO2013008951A1 (en) * | 2011-07-13 | 2013-01-17 | National Institute For Materials Science | Organic dye, dye-sensitized metal oxide semiconductor electrode and dye-sensitized solar cell |
| JP2013030466A (ja) * | 2011-06-23 | 2013-02-07 | Konica Minolta Business Technologies Inc | 光電変換素子、光電変換素子の製造方法および太陽電池 |
| JP2013239384A (ja) * | 2012-05-16 | 2013-11-28 | Konica Minolta Inc | 光電変換素子 |
| JP2014053263A (ja) * | 2012-09-10 | 2014-03-20 | Toyota Central R&D Labs Inc | 色素増感型太陽電池、色素増感型太陽電池モジュール及び有機色素の製造方法 |
| JPWO2013129605A1 (ja) * | 2012-03-01 | 2015-07-30 | 関東天然瓦斯開発株式会社 | 色素増感型光電変換素子用の非平面型構造を有する完全有機色素化合物、およびそれを用いた光電変換素子 |
| WO2021010425A1 (en) | 2019-07-16 | 2021-01-21 | Ricoh Company, Ltd. | Solar cell module, electronic device, and power supply module |
| EP3839994A1 (en) | 2019-11-28 | 2021-06-23 | Ricoh Company, Ltd. | Photoelectric conversion element, photoelectric conversion module, electronic device, and power supply module |
| US11286233B2 (en) * | 2017-05-09 | 2022-03-29 | Ambient Photonics, Inc. | Stilbene derivatives for the treatment of CNS and other disorders |
| EP4064355A1 (en) | 2021-03-23 | 2022-09-28 | Ricoh Company, Ltd. | Solar cell module |
| WO2023008085A1 (en) | 2021-07-29 | 2023-02-02 | Ricoh Company, Ltd. | Photoelectric conversion element and solar cell module |
| WO2023175466A1 (en) | 2022-03-18 | 2023-09-21 | Ricoh Company, Ltd. | Photoelectric conversion element, photoelectric conversion module, electronic device, and solar cell module |
Families Citing this family (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8455753B2 (en) * | 2005-01-14 | 2013-06-04 | Semiconductor Energy Laboratory Co., Ltd. | Solar cell and semiconductor device, and manufacturing method thereof |
| CA2655192A1 (en) * | 2006-07-05 | 2008-01-10 | Nippon Kayaku Kabushiki Kaisha | Dye-sensitized solar cell |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0911841A2 (en) * | 1997-10-23 | 1999-04-28 | Fuji Photo Film Co., Ltd. | Photoelectric conversion device and photoelectrochemical cell |
| EP1176618A1 (en) * | 2000-07-25 | 2002-01-30 | Fuji Photo Film Co., Ltd. | Metal complex dye, photoelectric conversion device and photoelectric cell |
-
2004
- 2004-01-16 TW TW093101139A patent/TWI318638B/zh not_active IP Right Cessation
- 2004-01-16 JP JP2005508016A patent/JP4341621B2/ja not_active Expired - Fee Related
- 2004-01-16 WO PCT/JP2004/000267 patent/WO2004063283A1/ja active Application Filing
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0911841A2 (en) * | 1997-10-23 | 1999-04-28 | Fuji Photo Film Co., Ltd. | Photoelectric conversion device and photoelectrochemical cell |
| EP1176618A1 (en) * | 2000-07-25 | 2002-01-30 | Fuji Photo Film Co., Ltd. | Metal complex dye, photoelectric conversion device and photoelectric cell |
Cited By (19)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP5086807B2 (ja) * | 2005-05-13 | 2012-11-28 | Agcセイミケミカル株式会社 | 新規アミノ基含有複素環誘導体および該複素環誘導体を含有する光電変換用増感色素 |
| WO2006120939A1 (ja) * | 2005-05-13 | 2006-11-16 | Agc Seimi Chemical Co., Ltd. | 新規アミノ基含有複素環誘導体および該複素環誘導体を含有する光電変換用増感色素 |
| US8044219B2 (en) | 2005-05-13 | 2011-10-25 | Agc Seimi Chemical., Ltd. | Amino group-containing heterocyclic derivatives and sensitizing dyes for photoelectric conversion containing the heterocyclic derivatives |
| JP2008044852A (ja) * | 2006-08-10 | 2008-02-28 | Tokyo Institute Of Technology | シクロペンテン骨格を有する有機リン化合物及びその製造方法 |
| JP5280853B2 (ja) * | 2006-11-07 | 2013-09-04 | Agcセイミケミカル株式会社 | 新規アミノ基含有芳香族化合物および該芳香族化合物を含有する光電変換用増感色素 |
| WO2008056567A1 (fr) * | 2006-11-07 | 2008-05-15 | Agc Seimi Chemical Co., Ltd. | Nouveau composé aromatique contenant un groupe amino, et colorant de sensibilisation pour conversion photoélectrique, contenant le composé aromatique |
| JP2013030466A (ja) * | 2011-06-23 | 2013-02-07 | Konica Minolta Business Technologies Inc | 光電変換素子、光電変換素子の製造方法および太陽電池 |
| WO2013008951A1 (en) * | 2011-07-13 | 2013-01-17 | National Institute For Materials Science | Organic dye, dye-sensitized metal oxide semiconductor electrode and dye-sensitized solar cell |
| JP5369238B2 (ja) * | 2011-07-14 | 2013-12-18 | 積水化学工業株式会社 | 光電変換素子用材料、光電変換素子の製造方法及び光電変換素子 |
| WO2013008889A1 (ja) * | 2011-07-14 | 2013-01-17 | 積水化学工業株式会社 | 光電変換素子用材料、光電変換素子の製造方法及び光電変換素子 |
| JPWO2013129605A1 (ja) * | 2012-03-01 | 2015-07-30 | 関東天然瓦斯開発株式会社 | 色素増感型光電変換素子用の非平面型構造を有する完全有機色素化合物、およびそれを用いた光電変換素子 |
| JP2013239384A (ja) * | 2012-05-16 | 2013-11-28 | Konica Minolta Inc | 光電変換素子 |
| JP2014053263A (ja) * | 2012-09-10 | 2014-03-20 | Toyota Central R&D Labs Inc | 色素増感型太陽電池、色素増感型太陽電池モジュール及び有機色素の製造方法 |
| US11286233B2 (en) * | 2017-05-09 | 2022-03-29 | Ambient Photonics, Inc. | Stilbene derivatives for the treatment of CNS and other disorders |
| WO2021010425A1 (en) | 2019-07-16 | 2021-01-21 | Ricoh Company, Ltd. | Solar cell module, electronic device, and power supply module |
| EP3839994A1 (en) | 2019-11-28 | 2021-06-23 | Ricoh Company, Ltd. | Photoelectric conversion element, photoelectric conversion module, electronic device, and power supply module |
| EP4064355A1 (en) | 2021-03-23 | 2022-09-28 | Ricoh Company, Ltd. | Solar cell module |
| WO2023008085A1 (en) | 2021-07-29 | 2023-02-02 | Ricoh Company, Ltd. | Photoelectric conversion element and solar cell module |
| WO2023175466A1 (en) | 2022-03-18 | 2023-09-21 | Ricoh Company, Ltd. | Photoelectric conversion element, photoelectric conversion module, electronic device, and solar cell module |
Also Published As
| Publication number | Publication date |
|---|---|
| JPWO2004063283A1 (ja) | 2006-05-18 |
| TW200422379A (en) | 2004-11-01 |
| TWI318638B (en) | 2009-12-21 |
| JP4341621B2 (ja) | 2009-10-07 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN102138249B (zh) | 光电器件 | |
| JP5925541B2 (ja) | 光電変換素子用金属錯体色素、光電変換素子、色素増感太陽電池、色素増感太陽電池用色素吸着組成液、色素増感太陽電池用半導体電極および色素増感太陽電池の製造方法 | |
| JP4341621B2 (ja) | 光機能材料 | |
| WO2007119525A1 (ja) | 有機化合物及びそれを用いた半導体薄膜電極、光電変換素子、光電気化学太陽電池 | |
| JP2010529226A (ja) | 新規な有機染料及びその製造方法 | |
| JP4423857B2 (ja) | 光機能材料 | |
| WO2012060346A1 (ja) | 光電気素子、光電気素子の製造方法、及び光増感剤 | |
| JPWO2010016612A1 (ja) | 色素増感型太陽電池用色素、半導体電極及び色素増感型太陽電池 | |
| JP4442105B2 (ja) | 光機能材料 | |
| JP2007246885A (ja) | 光機能材料 | |
| JP5972849B2 (ja) | 金属錯体、金属錯体色素、光電変換素子、色素増感太陽電池、色素溶液、色素吸着電極および色素増感太陽電池の製造方法 | |
| JP4230228B2 (ja) | 色素増感光電変換素子 | |
| JP4591667B2 (ja) | 光機能材料 | |
| JP2014172835A (ja) | 光機能材料及び光電変換用増感色素 | |
| TW201604245A (zh) | 光電轉換元件、色素增感太陽電池、金屬錯合物色素、色素溶液以及三聯吡啶化合物或它的酯化物 | |
| JP2008226505A (ja) | フェナントロチオフェン系化合物、および、その用途、ならびに製造方法 | |
| JP2011026389A (ja) | 光機能材料 | |
| JP2014209606A (ja) | 光電変換素子、色素増感太陽電池、金属錯体色素を含有する色素吸着液および光電変換素子の製造方法 | |
| JP5913223B2 (ja) | 金属錯体色素、光電変換素子、色素増感太陽電池、色素溶液および色素吸着電極 | |
| JP2007131767A (ja) | 光機能材料 | |
| JP2011057937A (ja) | 捻れたπ共役部分を有するドナー・アクセプター型色素、及びこれを用いた色素増感型太陽電池 | |
| JP2004292743A (ja) | 光機能材料 | |
| JP5401193B2 (ja) | 光電変換素子用色素および光電変換素子 | |
| JP5740984B2 (ja) | 光機能材料 | |
| JP2012131906A (ja) | 色素および色素増感太陽電池 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BW BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EC EE EG ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NA NI NO NZ OM PG PH PL PT RO RU SC SD SE SG SK SL SY TJ TM TN TR TT TZ UA UG US UZ VC VN YU ZA ZM ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): BW GH GM KE LS MW MZ SD SL SZ TZ UG ZM ZW AM AZ BY KG KZ MD RU TJ TM AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IT LU MC NL PT RO SE SI SK TR BF BJ CF CG CI CM GA GN GQ GW ML MR NE SN TD TG |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 2005508016 Country of ref document: JP |
|
| 122 | Ep: pct application non-entry in european phase |