RU2671089C2 - Би- или полиспецифические полипептиды, связывающие поверхностные антигены иммунных эффекторных клеток и антигены hbv для лечения инфекций bv и ассоциированных с ними состояний - Google Patents
Би- или полиспецифические полипептиды, связывающие поверхностные антигены иммунных эффекторных клеток и антигены hbv для лечения инфекций bv и ассоциированных с ними состояний Download PDFInfo
- Publication number
- RU2671089C2 RU2671089C2 RU2016114504A RU2016114504A RU2671089C2 RU 2671089 C2 RU2671089 C2 RU 2671089C2 RU 2016114504 A RU2016114504 A RU 2016114504A RU 2016114504 A RU2016114504 A RU 2016114504A RU 2671089 C2 RU2671089 C2 RU 2671089C2
- Authority
- RU
- Russia
- Prior art keywords
- hbv
- cells
- antigen
- polypeptide
- polypeptides
- Prior art date
Links
- 239000000427 antigen Substances 0.000 title claims abstract description 162
- 108091007433 antigens Proteins 0.000 title claims abstract description 162
- 102000036639 antigens Human genes 0.000 title claims abstract description 162
- 108090000765 processed proteins & peptides Proteins 0.000 title claims abstract description 123
- 102000004196 processed proteins & peptides Human genes 0.000 title claims abstract description 118
- 229920001184 polypeptide Polymers 0.000 title claims abstract description 116
- 208000015181 infectious disease Diseases 0.000 title claims abstract description 49
- 229940121354 immunomodulator Drugs 0.000 title claims abstract description 46
- 239000012642 immune effector Substances 0.000 title claims abstract description 45
- 230000027455 binding Effects 0.000 title claims abstract description 38
- 210000004027 cell Anatomy 0.000 title claims description 273
- 241000700721 Hepatitis B virus Species 0.000 claims abstract description 174
- 239000000203 mixture Substances 0.000 claims abstract description 29
- 208000014018 liver neoplasm Diseases 0.000 claims abstract description 14
- 201000007270 liver cancer Diseases 0.000 claims abstract description 13
- 208000019425 cirrhosis of liver Diseases 0.000 claims abstract description 12
- 108020004707 nucleic acids Proteins 0.000 claims abstract description 6
- 102000039446 nucleic acids Human genes 0.000 claims abstract description 6
- 150000007523 nucleic acids Chemical class 0.000 claims abstract description 6
- 108010047041 Complementarity Determining Regions Proteins 0.000 claims description 57
- 210000000822 natural killer cell Anatomy 0.000 claims description 39
- 239000003446 ligand Substances 0.000 claims description 31
- 210000001151 cytotoxic T lymphocyte Anatomy 0.000 claims description 29
- 238000006471 dimerization reaction Methods 0.000 claims description 22
- 101000914514 Homo sapiens T-cell-specific surface glycoprotein CD28 Proteins 0.000 claims description 18
- 102100032852 Natural cytotoxicity triggering receptor 3 Human genes 0.000 claims description 18
- 102100027213 T-cell-specific surface glycoprotein CD28 Human genes 0.000 claims description 18
- 239000012634 fragment Substances 0.000 claims description 18
- 101000581981 Homo sapiens Neural cell adhesion molecule 1 Proteins 0.000 claims description 15
- 210000003494 hepatocyte Anatomy 0.000 claims description 15
- 101000917858 Homo sapiens Low affinity immunoglobulin gamma Fc region receptor III-A Proteins 0.000 claims description 14
- 101000917839 Homo sapiens Low affinity immunoglobulin gamma Fc region receptor III-B Proteins 0.000 claims description 14
- 102100029185 Low affinity immunoglobulin gamma Fc region receptor III-B Human genes 0.000 claims description 14
- 102100027347 Neural cell adhesion molecule 1 Human genes 0.000 claims description 14
- 125000006850 spacer group Chemical group 0.000 claims description 14
- 102100022153 Tumor necrosis factor receptor superfamily member 4 Human genes 0.000 claims description 13
- 201000009030 Carcinoma Diseases 0.000 claims description 11
- 206010016654 Fibrosis Diseases 0.000 claims description 11
- 101001109501 Homo sapiens NKG2-D type II integral membrane protein Proteins 0.000 claims description 11
- 102100022680 NKG2-D type II integral membrane protein Human genes 0.000 claims description 11
- 108010004222 Natural Cytotoxicity Triggering Receptor 3 Proteins 0.000 claims description 11
- 101710165473 Tumor necrosis factor receptor superfamily member 4 Proteins 0.000 claims description 11
- 230000007882 cirrhosis Effects 0.000 claims description 11
- 108060003951 Immunoglobulin Proteins 0.000 claims description 8
- 102000018358 immunoglobulin Human genes 0.000 claims description 8
- 101000589307 Homo sapiens Natural cytotoxicity triggering receptor 3 Proteins 0.000 claims description 7
- 238000000338 in vitro Methods 0.000 claims description 6
- 239000008194 pharmaceutical composition Substances 0.000 claims description 6
- 108010087819 Fc receptors Proteins 0.000 claims description 5
- 102000009109 Fc receptors Human genes 0.000 claims description 5
- 230000035772 mutation Effects 0.000 claims description 3
- 230000001747 exhibiting effect Effects 0.000 claims description 2
- 125000003275 alpha amino acid group Chemical group 0.000 claims 4
- 230000009089 cytolysis Effects 0.000 abstract description 21
- 206010073071 hepatocellular carcinoma Diseases 0.000 abstract description 16
- 230000000694 effects Effects 0.000 abstract description 13
- 239000003814 drug Substances 0.000 abstract description 3
- 201000010099 disease Diseases 0.000 abstract 1
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 abstract 1
- 231100000844 hepatocellular carcinoma Toxicity 0.000 abstract 1
- 239000000126 substance Substances 0.000 abstract 1
- 210000001744 T-lymphocyte Anatomy 0.000 description 72
- 210000003819 peripheral blood mononuclear cell Anatomy 0.000 description 71
- 239000002609 medium Substances 0.000 description 34
- 108010002350 Interleukin-2 Proteins 0.000 description 32
- 102000000588 Interleukin-2 Human genes 0.000 description 32
- 150000001413 amino acids Chemical class 0.000 description 29
- 230000008030 elimination Effects 0.000 description 26
- 238000003379 elimination reaction Methods 0.000 description 26
- 230000035899 viability Effects 0.000 description 26
- 238000000034 method Methods 0.000 description 23
- 235000001014 amino acid Nutrition 0.000 description 22
- 229940024606 amino acid Drugs 0.000 description 22
- 239000012636 effector Substances 0.000 description 22
- 108060008682 Tumor Necrosis Factor Proteins 0.000 description 21
- 230000000638 stimulation Effects 0.000 description 21
- 108010074328 Interferon-gamma Proteins 0.000 description 20
- 239000006228 supernatant Substances 0.000 description 20
- 102000017420 CD3 protein, epsilon/gamma/delta subunit Human genes 0.000 description 19
- 108050005493 CD3 protein, epsilon/gamma/delta subunit Proteins 0.000 description 19
- 102100040247 Tumor necrosis factor Human genes 0.000 description 19
- 102100037850 Interferon gamma Human genes 0.000 description 18
- 230000028327 secretion Effects 0.000 description 18
- MZOFCQQQCNRIBI-VMXHOPILSA-N (3s)-4-[[(2s)-1-[[(2s)-1-[[(1s)-1-carboxy-2-hydroxyethyl]amino]-4-methyl-1-oxopentan-2-yl]amino]-5-(diaminomethylideneamino)-1-oxopentan-2-yl]amino]-3-[[2-[[(2s)-2,6-diaminohexanoyl]amino]acetyl]amino]-4-oxobutanoic acid Chemical compound OC[C@@H](C(O)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CCCN=C(N)N)NC(=O)[C@H](CC(O)=O)NC(=O)CNC(=O)[C@@H](N)CCCCN MZOFCQQQCNRIBI-VMXHOPILSA-N 0.000 description 17
- 102100032937 CD40 ligand Human genes 0.000 description 16
- 230000004913 activation Effects 0.000 description 16
- 238000002474 experimental method Methods 0.000 description 16
- 206010028980 Neoplasm Diseases 0.000 description 14
- 102000004127 Cytokines Human genes 0.000 description 12
- 108090000695 Cytokines Proteins 0.000 description 12
- 102000005962 receptors Human genes 0.000 description 12
- 108020003175 receptors Proteins 0.000 description 12
- 108090000623 proteins and genes Proteins 0.000 description 11
- 101000851370 Homo sapiens Tumor necrosis factor receptor superfamily member 9 Proteins 0.000 description 10
- 102100036856 Tumor necrosis factor receptor superfamily member 9 Human genes 0.000 description 10
- 230000004069 differentiation Effects 0.000 description 10
- 230000001404 mediated effect Effects 0.000 description 10
- 102000004169 proteins and genes Human genes 0.000 description 10
- 241000699670 Mus sp. Species 0.000 description 9
- 238000004458 analytical method Methods 0.000 description 9
- 235000018102 proteins Nutrition 0.000 description 9
- 238000002965 ELISA Methods 0.000 description 8
- 241001465754 Metazoa Species 0.000 description 8
- 210000004369 blood Anatomy 0.000 description 8
- 239000008280 blood Substances 0.000 description 8
- 231100000433 cytotoxic Toxicity 0.000 description 8
- 230000001472 cytotoxic effect Effects 0.000 description 8
- 230000003013 cytotoxicity Effects 0.000 description 8
- 231100000135 cytotoxicity Toxicity 0.000 description 8
- 230000003247 decreasing effect Effects 0.000 description 8
- JYGXADMDTFJGBT-VWUMJDOOSA-N hydrocortisone Chemical compound O=C1CC[C@]2(C)[C@H]3[C@@H](O)C[C@](C)([C@@](CC4)(O)C(=O)CO)[C@@H]4[C@@H]3CCC2=C1 JYGXADMDTFJGBT-VWUMJDOOSA-N 0.000 description 8
- 230000002147 killing effect Effects 0.000 description 8
- 239000003550 marker Substances 0.000 description 8
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 8
- MTCFGRXMJLQNBG-REOHCLBHSA-N (2S)-2-Amino-3-hydroxypropansäure Chemical compound OC[C@H](N)C(O)=O MTCFGRXMJLQNBG-REOHCLBHSA-N 0.000 description 7
- DHMQDGOQFOQNFH-UHFFFAOYSA-N Glycine Chemical compound NCC(O)=O DHMQDGOQFOQNFH-UHFFFAOYSA-N 0.000 description 7
- 102100035133 Lysosome-associated membrane glycoprotein 1 Human genes 0.000 description 7
- 101710116782 Lysosome-associated membrane glycoprotein 1 Proteins 0.000 description 7
- 108700012920 TNF Proteins 0.000 description 7
- 238000003501 co-culture Methods 0.000 description 7
- 230000006378 damage Effects 0.000 description 7
- 230000006870 function Effects 0.000 description 7
- 239000013612 plasmid Substances 0.000 description 7
- 102100035360 Cerebellar degeneration-related antigen 1 Human genes 0.000 description 6
- 108020004414 DNA Proteins 0.000 description 6
- 238000001943 fluorescence-activated cell sorting Methods 0.000 description 6
- 239000013642 negative control Substances 0.000 description 6
- 238000010606 normalization Methods 0.000 description 6
- UCSJYZPVAKXKNQ-HZYVHMACSA-N streptomycin Chemical compound CN[C@H]1[C@H](O)[C@@H](O)[C@H](CO)O[C@H]1O[C@@H]1[C@](C=O)(O)[C@H](C)O[C@H]1O[C@@H]1[C@@H](NC(N)=N)[C@H](O)[C@@H](NC(N)=N)[C@H](O)[C@H]1O UCSJYZPVAKXKNQ-HZYVHMACSA-N 0.000 description 6
- 230000002195 synergetic effect Effects 0.000 description 6
- 108020004638 Circular DNA Proteins 0.000 description 5
- 239000006144 Dulbecco’s modified Eagle's medium Substances 0.000 description 5
- 238000012413 Fluorescence activated cell sorting analysis Methods 0.000 description 5
- 241000700605 Viruses Species 0.000 description 5
- 230000015572 biosynthetic process Effects 0.000 description 5
- 230000003833 cell viability Effects 0.000 description 5
- 239000002245 particle Substances 0.000 description 5
- 101000638251 Homo sapiens Tumor necrosis factor ligand superfamily member 9 Proteins 0.000 description 4
- 102000018697 Membrane Proteins Human genes 0.000 description 4
- 108010052285 Membrane Proteins Proteins 0.000 description 4
- 102100032101 Tumor necrosis factor ligand superfamily member 9 Human genes 0.000 description 4
- 239000013543 active substance Substances 0.000 description 4
- 239000003153 chemical reaction reagent Substances 0.000 description 4
- 229960000890 hydrocortisone Drugs 0.000 description 4
- JTEGQNOMFQHVDC-NKWVEPMBSA-N lamivudine Chemical compound O=C1N=C(N)C=CN1[C@H]1O[C@@H](CO)SC1 JTEGQNOMFQHVDC-NKWVEPMBSA-N 0.000 description 4
- 238000004519 manufacturing process Methods 0.000 description 4
- 230000004048 modification Effects 0.000 description 4
- 238000012986 modification Methods 0.000 description 4
- 238000010186 staining Methods 0.000 description 4
- 230000009261 transgenic effect Effects 0.000 description 4
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 3
- 101000991061 Homo sapiens MHC class I polypeptide-related sequence B Proteins 0.000 description 3
- ZDXPYRJPNDTMRX-VKHMYHEASA-N L-glutamine Chemical compound OC(=O)[C@@H](N)CCC(N)=O ZDXPYRJPNDTMRX-VKHMYHEASA-N 0.000 description 3
- 229930182816 L-glutamine Natural products 0.000 description 3
- 102100030300 MHC class I polypeptide-related sequence B Human genes 0.000 description 3
- 229930182555 Penicillin Natural products 0.000 description 3
- JGSARLDLIJGVTE-MBNYWOFBSA-N Penicillin G Chemical compound N([C@H]1[C@H]2SC([C@@H](N2C1=O)C(O)=O)(C)C)C(=O)CC1=CC=CC=C1 JGSARLDLIJGVTE-MBNYWOFBSA-N 0.000 description 3
- MTCFGRXMJLQNBG-UHFFFAOYSA-N Serine Natural products OCC(N)C(O)=O MTCFGRXMJLQNBG-UHFFFAOYSA-N 0.000 description 3
- 102000000852 Tumor Necrosis Factor-alpha Human genes 0.000 description 3
- WOZSCQDILHKSGG-UHFFFAOYSA-N adefovir depivoxil Chemical compound N1=CN=C2N(CCOCP(=O)(OCOC(=O)C(C)(C)C)OCOC(=O)C(C)(C)C)C=NC2=C1N WOZSCQDILHKSGG-UHFFFAOYSA-N 0.000 description 3
- 230000010056 antibody-dependent cellular cytotoxicity Effects 0.000 description 3
- 239000003443 antiviral agent Substances 0.000 description 3
- 210000000170 cell membrane Anatomy 0.000 description 3
- 238000003570 cell viability assay Methods 0.000 description 3
- 239000002299 complementary DNA Substances 0.000 description 3
- 125000000151 cysteine group Chemical group N[C@@H](CS)C(=O)* 0.000 description 3
- 230000001419 dependent effect Effects 0.000 description 3
- 239000010432 diamond Substances 0.000 description 3
- 210000002472 endoplasmic reticulum Anatomy 0.000 description 3
- 230000004927 fusion Effects 0.000 description 3
- 230000013595 glycosylation Effects 0.000 description 3
- 238000006206 glycosylation reaction Methods 0.000 description 3
- 210000004408 hybridoma Anatomy 0.000 description 3
- 210000002865 immune cell Anatomy 0.000 description 3
- 238000001727 in vivo Methods 0.000 description 3
- 238000002955 isolation Methods 0.000 description 3
- 210000005229 liver cell Anatomy 0.000 description 3
- 229910052618 mica group Inorganic materials 0.000 description 3
- 125000003729 nucleotide group Chemical group 0.000 description 3
- 229940049954 penicillin Drugs 0.000 description 3
- 230000002829 reductive effect Effects 0.000 description 3
- 229960005322 streptomycin Drugs 0.000 description 3
- 238000007920 subcutaneous administration Methods 0.000 description 3
- 230000005945 translocation Effects 0.000 description 3
- 210000004881 tumor cell Anatomy 0.000 description 3
- 238000005406 washing Methods 0.000 description 3
- 108091003079 Bovine Serum Albumin Proteins 0.000 description 2
- 108010029697 CD40 Ligand Proteins 0.000 description 2
- 108010019670 Chimeric Antigen Receptors Proteins 0.000 description 2
- 108020004635 Complementary DNA Proteins 0.000 description 2
- 108010073807 IgG Receptors Proteins 0.000 description 2
- 102000009490 IgG Receptors Human genes 0.000 description 2
- 102000018071 Immunoglobulin Fc Fragments Human genes 0.000 description 2
- 108010091135 Immunoglobulin Fc Fragments Proteins 0.000 description 2
- 108010078049 Interferon alpha-2 Proteins 0.000 description 2
- UQSXHKLRYXJYBZ-UHFFFAOYSA-N Iron oxide Chemical compound [Fe]=O UQSXHKLRYXJYBZ-UHFFFAOYSA-N 0.000 description 2
- 241000699666 Mus <mouse, genus> Species 0.000 description 2
- 108010069196 Neural Cell Adhesion Molecules Proteins 0.000 description 2
- 239000012124 Opti-MEM Substances 0.000 description 2
- 239000012980 RPMI-1640 medium Substances 0.000 description 2
- 230000006044 T cell activation Effects 0.000 description 2
- 108091008874 T cell receptors Proteins 0.000 description 2
- 102000016266 T-Cell Antigen Receptors Human genes 0.000 description 2
- 230000003213 activating effect Effects 0.000 description 2
- 229960003205 adefovir dipivoxil Drugs 0.000 description 2
- 230000000840 anti-viral effect Effects 0.000 description 2
- 238000002869 basic local alignment search tool Methods 0.000 description 2
- 230000037396 body weight Effects 0.000 description 2
- KQNZDYYTLMIZCT-KQPMLPITSA-N brefeldin A Chemical compound O[C@@H]1\C=C\C(=O)O[C@@H](C)CCC\C=C\[C@@H]2C[C@H](O)C[C@H]21 KQNZDYYTLMIZCT-KQPMLPITSA-N 0.000 description 2
- JUMGSHROWPPKFX-UHFFFAOYSA-N brefeldin-A Natural products CC1CCCC=CC2(C)CC(O)CC2(C)C(O)C=CC(=O)O1 JUMGSHROWPPKFX-UHFFFAOYSA-N 0.000 description 2
- 239000000872 buffer Substances 0.000 description 2
- 210000004899 c-terminal region Anatomy 0.000 description 2
- 230000001684 chronic effect Effects 0.000 description 2
- 238000011260 co-administration Methods 0.000 description 2
- 239000000470 constituent Substances 0.000 description 2
- 238000007796 conventional method Methods 0.000 description 2
- 230000004940 costimulation Effects 0.000 description 2
- 230000000139 costimulatory effect Effects 0.000 description 2
- 230000016396 cytokine production Effects 0.000 description 2
- 230000007402 cytotoxic response Effects 0.000 description 2
- 238000013480 data collection Methods 0.000 description 2
- 229940079593 drug Drugs 0.000 description 2
- 239000003937 drug carrier Substances 0.000 description 2
- 239000000839 emulsion Substances 0.000 description 2
- 238000005516 engineering process Methods 0.000 description 2
- QDGZDCVAUDNJFG-FXQIFTODSA-N entecavir (anhydrous) Chemical compound C1=2NC(N)=NC(=O)C=2N=CN1[C@H]1C[C@H](O)[C@@H](CO)C1=C QDGZDCVAUDNJFG-FXQIFTODSA-N 0.000 description 2
- 239000012894 fetal calf serum Substances 0.000 description 2
- 208000002672 hepatitis B Diseases 0.000 description 2
- 229940072221 immunoglobulins Drugs 0.000 description 2
- 238000011534 incubation Methods 0.000 description 2
- 230000006698 induction Effects 0.000 description 2
- 230000005764 inhibitory process Effects 0.000 description 2
- 239000007924 injection Substances 0.000 description 2
- 238000002347 injection Methods 0.000 description 2
- NOESYZHRGYRDHS-UHFFFAOYSA-N insulin Chemical compound N1C(=O)C(NC(=O)C(CCC(N)=O)NC(=O)C(CCC(O)=O)NC(=O)C(C(C)C)NC(=O)C(NC(=O)CN)C(C)CC)CSSCC(C(NC(CO)C(=O)NC(CC(C)C)C(=O)NC(CC=2C=CC(O)=CC=2)C(=O)NC(CCC(N)=O)C(=O)NC(CC(C)C)C(=O)NC(CCC(O)=O)C(=O)NC(CC(N)=O)C(=O)NC(CC=2C=CC(O)=CC=2)C(=O)NC(CSSCC(NC(=O)C(C(C)C)NC(=O)C(CC(C)C)NC(=O)C(CC=2C=CC(O)=CC=2)NC(=O)C(CC(C)C)NC(=O)C(C)NC(=O)C(CCC(O)=O)NC(=O)C(C(C)C)NC(=O)C(CC(C)C)NC(=O)C(CC=2NC=NC=2)NC(=O)C(CO)NC(=O)CNC2=O)C(=O)NCC(=O)NC(CCC(O)=O)C(=O)NC(CCCNC(N)=N)C(=O)NCC(=O)NC(CC=3C=CC=CC=3)C(=O)NC(CC=3C=CC=CC=3)C(=O)NC(CC=3C=CC(O)=CC=3)C(=O)NC(C(C)O)C(=O)N3C(CCC3)C(=O)NC(CCCCN)C(=O)NC(C)C(O)=O)C(=O)NC(CC(N)=O)C(O)=O)=O)NC(=O)C(C(C)CC)NC(=O)C(CO)NC(=O)C(C(C)O)NC(=O)C1CSSCC2NC(=O)C(CC(C)C)NC(=O)C(NC(=O)C(CCC(N)=O)NC(=O)C(CC(N)=O)NC(=O)C(NC(=O)C(N)CC=1C=CC=CC=1)C(C)C)CC1=CN=CN1 NOESYZHRGYRDHS-UHFFFAOYSA-N 0.000 description 2
- 210000004020 intracellular membrane Anatomy 0.000 description 2
- 238000001990 intravenous administration Methods 0.000 description 2
- 229960001627 lamivudine Drugs 0.000 description 2
- 108020004999 messenger RNA Proteins 0.000 description 2
- 230000002503 metabolic effect Effects 0.000 description 2
- 238000012544 monitoring process Methods 0.000 description 2
- 230000009871 nonspecific binding Effects 0.000 description 2
- 239000002773 nucleotide Substances 0.000 description 2
- 239000000546 pharmaceutical excipient Substances 0.000 description 2
- 230000004481 post-translational protein modification Effects 0.000 description 2
- 230000008569 process Effects 0.000 description 2
- 230000000770 proinflammatory effect Effects 0.000 description 2
- 230000009467 reduction Effects 0.000 description 2
- 230000010076 replication Effects 0.000 description 2
- 230000003248 secreting effect Effects 0.000 description 2
- 239000002356 single layer Substances 0.000 description 2
- IQFYYKKMVGJFEH-CSMHCCOUSA-N telbivudine Chemical compound O=C1NC(=O)C(C)=CN1[C@H]1O[C@@H](CO)[C@H](O)C1 IQFYYKKMVGJFEH-CSMHCCOUSA-N 0.000 description 2
- 210000001519 tissue Anatomy 0.000 description 2
- 238000001890 transfection Methods 0.000 description 2
- 239000012096 transfection reagent Substances 0.000 description 2
- 230000029812 viral genome replication Effects 0.000 description 2
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 2
- KQNZDYYTLMIZCT-KFKPYADVSA-N (2e,7s,10e,12r,13r,15s)-12,15-dihydroxy-7-methyl-8-oxabicyclo[11.3.0]hexadeca-2,10-dien-9-one Chemical compound O[C@@H]1\C=C\C(=O)O[C@@H](C)CCC\C=C\C2C[C@H](O)C[C@H]21 KQNZDYYTLMIZCT-KFKPYADVSA-N 0.000 description 1
- FDKWRPBBCBCIGA-REOHCLBHSA-N (2r)-2-azaniumyl-3-$l^{1}-selanylpropanoate Chemical compound [Se]C[C@H](N)C(O)=O FDKWRPBBCBCIGA-REOHCLBHSA-N 0.000 description 1
- QNRATNLHPGXHMA-XZHTYLCXSA-N (r)-(6-ethoxyquinolin-4-yl)-[(2s,4s,5r)-5-ethyl-1-azabicyclo[2.2.2]octan-2-yl]methanol;hydrochloride Chemical compound Cl.C([C@H]([C@H](C1)CC)C2)CN1[C@@H]2[C@H](O)C1=CC=NC2=CC=C(OCC)C=C21 QNRATNLHPGXHMA-XZHTYLCXSA-N 0.000 description 1
- 108091032973 (ribonucleotides)n+m Proteins 0.000 description 1
- NFGXHKASABOEEW-UHFFFAOYSA-N 1-methylethyl 11-methoxy-3,7,11-trimethyl-2,4-dodecadienoate Chemical compound COC(C)(C)CCCC(C)CC=CC(C)=CC(=O)OC(C)C NFGXHKASABOEEW-UHFFFAOYSA-N 0.000 description 1
- JKMHFZQWWAIEOD-UHFFFAOYSA-N 2-[4-(2-hydroxyethyl)piperazin-1-yl]ethanesulfonic acid Chemical compound OCC[NH+]1CCN(CCS([O-])(=O)=O)CC1 JKMHFZQWWAIEOD-UHFFFAOYSA-N 0.000 description 1
- 108020005065 3' Flanking Region Proteins 0.000 description 1
- 125000004042 4-aminobutyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])N([H])[H] 0.000 description 1
- 108020005029 5' Flanking Region Proteins 0.000 description 1
- IPWKGIFRRBGCJO-IMJSIDKUSA-N Ala-Ser Chemical compound C[C@H]([NH3+])C(=O)N[C@@H](CO)C([O-])=O IPWKGIFRRBGCJO-IMJSIDKUSA-N 0.000 description 1
- 108010011170 Ala-Trp-Arg-His-Pro-Gln-Phe-Gly-Gly Proteins 0.000 description 1
- 241000415078 Anemone hepatica Species 0.000 description 1
- DCXYFEDJOCDNAF-UHFFFAOYSA-N Asparagine Natural products OC(=O)C(N)CC(N)=O DCXYFEDJOCDNAF-UHFFFAOYSA-N 0.000 description 1
- 108700031361 Brachyury Proteins 0.000 description 1
- 241000283707 Capra Species 0.000 description 1
- 108091026890 Coding region Proteins 0.000 description 1
- FDKWRPBBCBCIGA-UWTATZPHSA-N D-Selenocysteine Natural products [Se]C[C@@H](N)C(O)=O FDKWRPBBCBCIGA-UWTATZPHSA-N 0.000 description 1
- 150000008574 D-amino acids Chemical class 0.000 description 1
- 101100193633 Danio rerio rag2 gene Proteins 0.000 description 1
- 101710142246 External core antigen Proteins 0.000 description 1
- 229930182566 Gentamicin Natural products 0.000 description 1
- CEAZRRDELHUEMR-URQXQFDESA-N Gentamicin Chemical compound O1[C@H](C(C)NC)CC[C@@H](N)[C@H]1O[C@H]1[C@H](O)[C@@H](O[C@@H]2[C@@H]([C@@H](NC)[C@@](C)(O)CO2)O)[C@H](N)C[C@@H]1N CEAZRRDELHUEMR-URQXQFDESA-N 0.000 description 1
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 1
- XCLCVBYNGXEVDU-WHFBIAKZSA-N Gly-Asn-Ser Chemical compound NCC(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CO)C(O)=O XCLCVBYNGXEVDU-WHFBIAKZSA-N 0.000 description 1
- BCCRXDTUTZHDEU-VKHMYHEASA-N Gly-Ser Chemical group NCC(=O)N[C@@H](CO)C(O)=O BCCRXDTUTZHDEU-VKHMYHEASA-N 0.000 description 1
- 239000004471 Glycine Substances 0.000 description 1
- 239000007995 HEPES buffer Substances 0.000 description 1
- 206010019695 Hepatic neoplasm Diseases 0.000 description 1
- 208000017604 Hodgkin disease Diseases 0.000 description 1
- 208000021519 Hodgkin lymphoma Diseases 0.000 description 1
- 208000010747 Hodgkins lymphoma Diseases 0.000 description 1
- 101100166600 Homo sapiens CD28 gene Proteins 0.000 description 1
- 101000599940 Homo sapiens Interferon gamma Proteins 0.000 description 1
- 101001109508 Homo sapiens NKG2-A/NKG2-B type II integral membrane protein Proteins 0.000 description 1
- 101000851376 Homo sapiens Tumor necrosis factor receptor superfamily member 8 Proteins 0.000 description 1
- PMMYEEVYMWASQN-DMTCNVIQSA-N Hydroxyproline Chemical compound O[C@H]1CN[C@H](C(O)=O)C1 PMMYEEVYMWASQN-DMTCNVIQSA-N 0.000 description 1
- 206010061598 Immunodeficiency Diseases 0.000 description 1
- 206010062016 Immunosuppression Diseases 0.000 description 1
- 229930010555 Inosine Natural products 0.000 description 1
- UGQMRVRMYYASKQ-KQYNXXCUSA-N Inosine Chemical compound O[C@@H]1[C@H](O)[C@@H](CO)O[C@H]1N1C2=NC=NC(O)=C2N=C1 UGQMRVRMYYASKQ-KQYNXXCUSA-N 0.000 description 1
- 102000004877 Insulin Human genes 0.000 description 1
- 108090001061 Insulin Proteins 0.000 description 1
- 102000008070 Interferon-gamma Human genes 0.000 description 1
- 108010050904 Interferons Proteins 0.000 description 1
- 102000014150 Interferons Human genes 0.000 description 1
- AHLPHDHHMVZTML-BYPYZUCNSA-N L-Ornithine Chemical compound NCCC[C@H](N)C(O)=O AHLPHDHHMVZTML-BYPYZUCNSA-N 0.000 description 1
- DCXYFEDJOCDNAF-REOHCLBHSA-N L-asparagine Chemical compound OC(=O)[C@@H](N)CC(N)=O DCXYFEDJOCDNAF-REOHCLBHSA-N 0.000 description 1
- WHUUTDBJXJRKMK-VKHMYHEASA-N L-glutamic acid Chemical compound OC(=O)[C@@H](N)CCC(O)=O WHUUTDBJXJRKMK-VKHMYHEASA-N 0.000 description 1
- ROHFNLRQFUQHCH-YFKPBYRVSA-N L-leucine Chemical compound CC(C)C[C@H](N)C(O)=O ROHFNLRQFUQHCH-YFKPBYRVSA-N 0.000 description 1
- ZFOMKMMPBOQKMC-KXUCPTDWSA-N L-pyrrolysine Chemical compound C[C@@H]1CC=N[C@H]1C(=O)NCCCC[C@H]([NH3+])C([O-])=O ZFOMKMMPBOQKMC-KXUCPTDWSA-N 0.000 description 1
- AYFVYJQAPQTCCC-GBXIJSLDSA-N L-threonine Chemical compound C[C@@H](O)[C@H](N)C(O)=O AYFVYJQAPQTCCC-GBXIJSLDSA-N 0.000 description 1
- OUYCCCASQSFEME-QMMMGPOBSA-N L-tyrosine Chemical compound OC(=O)[C@@H](N)CC1=CC=C(O)C=C1 OUYCCCASQSFEME-QMMMGPOBSA-N 0.000 description 1
- 101710084021 Large envelope protein Proteins 0.000 description 1
- ROHFNLRQFUQHCH-UHFFFAOYSA-N Leucine Natural products CC(C)CC(N)C(O)=O ROHFNLRQFUQHCH-UHFFFAOYSA-N 0.000 description 1
- 241000124008 Mammalia Species 0.000 description 1
- 241000699660 Mus musculus Species 0.000 description 1
- 101100193635 Mus musculus Rag2 gene Proteins 0.000 description 1
- 230000004988 N-glycosylation Effects 0.000 description 1
- 108010057466 NF-kappa B Proteins 0.000 description 1
- 102000003945 NF-kappa B Human genes 0.000 description 1
- 102000000835 NK Cell Lectin-Like Receptor Subfamily B Human genes 0.000 description 1
- 108010001882 NK Cell Lectin-Like Receptor Subfamily B Proteins 0.000 description 1
- 102100022682 NKG2-A/NKG2-B type II integral membrane protein Human genes 0.000 description 1
- 102000001068 Neural Cell Adhesion Molecules Human genes 0.000 description 1
- 102100023616 Neural cell adhesion molecule L1-like protein Human genes 0.000 description 1
- 108091028043 Nucleic acid sequence Proteins 0.000 description 1
- 230000004989 O-glycosylation Effects 0.000 description 1
- AHLPHDHHMVZTML-UHFFFAOYSA-N Orn-delta-NH2 Natural products NCCCC(N)C(O)=O AHLPHDHHMVZTML-UHFFFAOYSA-N 0.000 description 1
- UTJLXEIPEHZYQJ-UHFFFAOYSA-N Ornithine Natural products OC(=O)C(C)CCCN UTJLXEIPEHZYQJ-UHFFFAOYSA-N 0.000 description 1
- 238000012408 PCR amplification Methods 0.000 description 1
- 108090000526 Papain Proteins 0.000 description 1
- 108091093037 Peptide nucleic acid Proteins 0.000 description 1
- 102000003992 Peroxidases Human genes 0.000 description 1
- 206010035226 Plasma cell myeloma Diseases 0.000 description 1
- 241000276498 Pollachius virens Species 0.000 description 1
- 239000004365 Protease Substances 0.000 description 1
- 239000012979 RPMI medium Substances 0.000 description 1
- PLXBWHJQWKZRKG-UHFFFAOYSA-N Resazurin Chemical compound C1=CC(=O)C=C2OC3=CC(O)=CC=C3[N+]([O-])=C21 PLXBWHJQWKZRKG-UHFFFAOYSA-N 0.000 description 1
- 239000006146 Roswell Park Memorial Institute medium Substances 0.000 description 1
- RJFAYQIBOAGBLC-BYPYZUCNSA-N Selenium-L-methionine Chemical compound C[Se]CC[C@H](N)C(O)=O RJFAYQIBOAGBLC-BYPYZUCNSA-N 0.000 description 1
- RJFAYQIBOAGBLC-UHFFFAOYSA-N Selenomethionine Natural products C[Se]CCC(N)C(O)=O RJFAYQIBOAGBLC-UHFFFAOYSA-N 0.000 description 1
- 238000012300 Sequence Analysis Methods 0.000 description 1
- 101710137302 Surface antigen S Proteins 0.000 description 1
- 230000024932 T cell mediated immunity Effects 0.000 description 1
- AYFVYJQAPQTCCC-UHFFFAOYSA-N Threonine Natural products CC(O)C(N)C(O)=O AYFVYJQAPQTCCC-UHFFFAOYSA-N 0.000 description 1
- 239000004473 Threonine Substances 0.000 description 1
- 108010028230 Trp-Ser- His-Pro-Gln-Phe-Glu-Lys Proteins 0.000 description 1
- 108060008683 Tumor Necrosis Factor Receptor Proteins 0.000 description 1
- 102100036857 Tumor necrosis factor receptor superfamily member 8 Human genes 0.000 description 1
- 206010058874 Viraemia Diseases 0.000 description 1
- 108010003533 Viral Envelope Proteins Proteins 0.000 description 1
- 108010067390 Viral Proteins Proteins 0.000 description 1
- 230000001594 aberrant effect Effects 0.000 description 1
- 238000010521 absorption reaction Methods 0.000 description 1
- 125000002777 acetyl group Chemical group [H]C([H])([H])C(*)=O 0.000 description 1
- 230000001464 adherent effect Effects 0.000 description 1
- 150000001371 alpha-amino acids Chemical class 0.000 description 1
- 235000008206 alpha-amino acids Nutrition 0.000 description 1
- 150000001408 amides Chemical class 0.000 description 1
- 150000001412 amines Chemical class 0.000 description 1
- 125000003277 amino group Chemical group 0.000 description 1
- 235000009582 asparagine Nutrition 0.000 description 1
- 229960001230 asparagine Drugs 0.000 description 1
- -1 aspartate side chain carboxylate Chemical class 0.000 description 1
- 229940002637 baraclude Drugs 0.000 description 1
- 230000008901 benefit Effects 0.000 description 1
- 150000001576 beta-amino acids Chemical class 0.000 description 1
- 201000011510 cancer Diseases 0.000 description 1
- 239000000969 carrier Substances 0.000 description 1
- 238000004113 cell culture Methods 0.000 description 1
- 239000006143 cell culture medium Substances 0.000 description 1
- 230000006037 cell lysis Effects 0.000 description 1
- 230000001413 cellular effect Effects 0.000 description 1
- 238000005119 centrifugation Methods 0.000 description 1
- 230000008859 change Effects 0.000 description 1
- 238000012512 characterization method Methods 0.000 description 1
- 239000007795 chemical reaction product Substances 0.000 description 1
- 239000003795 chemical substances by application Substances 0.000 description 1
- 238000003776 cleavage reaction Methods 0.000 description 1
- 238000010367 cloning Methods 0.000 description 1
- 239000012881 co-culture medium Substances 0.000 description 1
- 239000003086 colorant Substances 0.000 description 1
- 238000002648 combination therapy Methods 0.000 description 1
- 150000001875 compounds Chemical class 0.000 description 1
- 108091008034 costimulatory receptors Proteins 0.000 description 1
- 230000009260 cross reactivity Effects 0.000 description 1
- 239000012228 culture supernatant Substances 0.000 description 1
- 238000012258 culturing Methods 0.000 description 1
- 230000034994 death Effects 0.000 description 1
- 231100000517 death Toxicity 0.000 description 1
- 238000012217 deletion Methods 0.000 description 1
- 230000037430 deletion Effects 0.000 description 1
- 238000000432 density-gradient centrifugation Methods 0.000 description 1
- 239000003085 diluting agent Substances 0.000 description 1
- 239000000539 dimer Substances 0.000 description 1
- PMMYEEVYMWASQN-UHFFFAOYSA-N dl-hydroxyproline Natural products OC1C[NH2+]C(C([O-])=O)C1 PMMYEEVYMWASQN-UHFFFAOYSA-N 0.000 description 1
- 231100000673 dose–response relationship Toxicity 0.000 description 1
- 229960000980 entecavir Drugs 0.000 description 1
- 229940074057 epivir hbv Drugs 0.000 description 1
- 230000028023 exocytosis Effects 0.000 description 1
- 239000013604 expression vector Substances 0.000 description 1
- 210000004700 fetal blood Anatomy 0.000 description 1
- 238000011010 flushing procedure Methods 0.000 description 1
- 238000009472 formulation Methods 0.000 description 1
- 229960002518 gentamicin Drugs 0.000 description 1
- 239000008103 glucose Substances 0.000 description 1
- 229930195712 glutamate Natural products 0.000 description 1
- 125000003630 glycyl group Chemical group [H]N([H])C([H])([H])C(*)=O 0.000 description 1
- XKUKSGPZAADMRA-UHFFFAOYSA-N glycyl-glycyl-glycine Chemical compound NCC(=O)NCC(=O)NCC(O)=O XKUKSGPZAADMRA-UHFFFAOYSA-N 0.000 description 1
- PCHJSUWPFVWCPO-UHFFFAOYSA-N gold Chemical compound [Au] PCHJSUWPFVWCPO-UHFFFAOYSA-N 0.000 description 1
- 239000010931 gold Substances 0.000 description 1
- 229910052737 gold Inorganic materials 0.000 description 1
- 210000002288 golgi apparatus Anatomy 0.000 description 1
- 239000008187 granular material Substances 0.000 description 1
- 230000036541 health Effects 0.000 description 1
- 229940097709 hepsera Drugs 0.000 description 1
- 239000000833 heterodimer Substances 0.000 description 1
- 239000000710 homodimer Substances 0.000 description 1
- 102000049018 human NCAM1 Human genes 0.000 description 1
- 230000028996 humoral immune response Effects 0.000 description 1
- 229960002591 hydroxyproline Drugs 0.000 description 1
- 230000006058 immune tolerance Effects 0.000 description 1
- 239000002955 immunomodulating agent Substances 0.000 description 1
- 230000001506 immunosuppresive effect Effects 0.000 description 1
- 238000009169 immunotherapy Methods 0.000 description 1
- 230000001976 improved effect Effects 0.000 description 1
- 238000010348 incorporation Methods 0.000 description 1
- 230000001939 inductive effect Effects 0.000 description 1
- 230000002458 infectious effect Effects 0.000 description 1
- 238000001802 infusion Methods 0.000 description 1
- 239000003112 inhibitor Substances 0.000 description 1
- 230000002401 inhibitory effect Effects 0.000 description 1
- 230000000977 initiatory effect Effects 0.000 description 1
- 239000002054 inoculum Substances 0.000 description 1
- 229960003786 inosine Drugs 0.000 description 1
- 238000003780 insertion Methods 0.000 description 1
- 230000037431 insertion Effects 0.000 description 1
- 229940125396 insulin Drugs 0.000 description 1
- 229960003521 interferon alfa-2a Drugs 0.000 description 1
- 229960003507 interferon alfa-2b Drugs 0.000 description 1
- 229940047124 interferons Drugs 0.000 description 1
- 230000003834 intracellular effect Effects 0.000 description 1
- 238000010212 intracellular staining Methods 0.000 description 1
- 238000007918 intramuscular administration Methods 0.000 description 1
- 238000007912 intraperitoneal administration Methods 0.000 description 1
- 239000010410 layer Substances 0.000 description 1
- 210000000265 leukocyte Anatomy 0.000 description 1
- 210000004185 liver Anatomy 0.000 description 1
- 230000007774 longterm Effects 0.000 description 1
- 210000004698 lymphocyte Anatomy 0.000 description 1
- 239000006166 lysate Substances 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- 238000005259 measurement Methods 0.000 description 1
- MYWUZJCMWCOHBA-VIFPVBQESA-N methamphetamine Chemical compound CN[C@@H](C)CC1=CC=CC=C1 MYWUZJCMWCOHBA-VIFPVBQESA-N 0.000 description 1
- 239000010445 mica Substances 0.000 description 1
- 239000004005 microsphere Substances 0.000 description 1
- 238000010369 molecular cloning Methods 0.000 description 1
- 210000001616 monocyte Anatomy 0.000 description 1
- 201000000050 myeloid neoplasm Diseases 0.000 description 1
- 108010045147 myrcludex-B Proteins 0.000 description 1
- 238000011580 nude mouse model Methods 0.000 description 1
- 229960003104 ornithine Drugs 0.000 description 1
- 229940055729 papain Drugs 0.000 description 1
- 235000019834 papain Nutrition 0.000 description 1
- 230000035515 penetration Effects 0.000 description 1
- 230000035699 permeability Effects 0.000 description 1
- 108040007629 peroxidase activity proteins Proteins 0.000 description 1
- 239000008024 pharmaceutical diluent Substances 0.000 description 1
- 239000008363 phosphate buffer Substances 0.000 description 1
- 230000004962 physiological condition Effects 0.000 description 1
- 238000002360 preparation method Methods 0.000 description 1
- 239000000047 product Substances 0.000 description 1
- 230000000069 prophylactic effect Effects 0.000 description 1
- 125000006239 protecting group Chemical group 0.000 description 1
- 230000001681 protective effect Effects 0.000 description 1
- 238000000746 purification Methods 0.000 description 1
- 238000011002 quantification Methods 0.000 description 1
- 230000007420 reactivation Effects 0.000 description 1
- 230000006798 recombination Effects 0.000 description 1
- 238000005215 recombination Methods 0.000 description 1
- 230000003362 replicative effect Effects 0.000 description 1
- 238000011160 research Methods 0.000 description 1
- HSSLDCABUXLXKM-UHFFFAOYSA-N resorufin Chemical compound C1=CC(=O)C=C2OC3=CC(O)=CC=C3N=C21 HSSLDCABUXLXKM-UHFFFAOYSA-N 0.000 description 1
- 230000000284 resting effect Effects 0.000 description 1
- 230000001177 retroviral effect Effects 0.000 description 1
- 230000002441 reversible effect Effects 0.000 description 1
- 230000007017 scission Effects 0.000 description 1
- 235000016491 selenocysteine Nutrition 0.000 description 1
- ZKZBPNGNEQAJSX-UHFFFAOYSA-N selenocysteine Natural products [SeH]CC(N)C(O)=O ZKZBPNGNEQAJSX-UHFFFAOYSA-N 0.000 description 1
- 229940055619 selenocysteine Drugs 0.000 description 1
- 229960002718 selenomethionine Drugs 0.000 description 1
- 238000000926 separation method Methods 0.000 description 1
- 125000003607 serino group Chemical group [H]N([H])[C@]([H])(C(=O)[*])C(O[H])([H])[H] 0.000 description 1
- 210000002966 serum Anatomy 0.000 description 1
- 238000005549 size reduction Methods 0.000 description 1
- 239000000243 solution Substances 0.000 description 1
- 230000009870 specific binding Effects 0.000 description 1
- 210000004989 spleen cell Anatomy 0.000 description 1
- 239000008174 sterile solution Substances 0.000 description 1
- 230000004936 stimulating effect Effects 0.000 description 1
- 238000006467 substitution reaction Methods 0.000 description 1
- 235000020357 syrup Nutrition 0.000 description 1
- 239000006188 syrup Substances 0.000 description 1
- 230000009897 systematic effect Effects 0.000 description 1
- 230000008685 targeting Effects 0.000 description 1
- 229960005311 telbivudine Drugs 0.000 description 1
- SGOIRFVFHAKUTI-ZCFIWIBFSA-N tenofovir (anhydrous) Chemical compound N1=CN=C2N(C[C@@H](C)OCP(O)(O)=O)C=NC2=C1N SGOIRFVFHAKUTI-ZCFIWIBFSA-N 0.000 description 1
- 238000012360 testing method Methods 0.000 description 1
- 230000001225 therapeutic effect Effects 0.000 description 1
- 238000002560 therapeutic procedure Methods 0.000 description 1
- 125000000341 threoninyl group Chemical group [H]OC([H])(C([H])([H])[H])C([H])(N([H])[H])C(*)=O 0.000 description 1
- 238000004448 titration Methods 0.000 description 1
- FGMPLJWBKKVCDB-UHFFFAOYSA-N trans-L-hydroxy-proline Natural products ON1CCCC1C(O)=O FGMPLJWBKKVCDB-UHFFFAOYSA-N 0.000 description 1
- 230000032258 transport Effects 0.000 description 1
- 230000006433 tumor necrosis factor production Effects 0.000 description 1
- 102000003298 tumor necrosis factor receptor Human genes 0.000 description 1
- OUYCCCASQSFEME-UHFFFAOYSA-N tyrosine Natural products OC(=O)C(N)CC1=CC=C(O)C=C1 OUYCCCASQSFEME-UHFFFAOYSA-N 0.000 description 1
- 229940063032 tyzeka Drugs 0.000 description 1
- 210000003462 vein Anatomy 0.000 description 1
- 230000028973 vesicle-mediated transport Effects 0.000 description 1
- 210000002845 virion Anatomy 0.000 description 1
- 230000003612 virological effect Effects 0.000 description 1
- 238000001262 western blot Methods 0.000 description 1
- 239000000080 wetting agent Substances 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/28—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants
- C07K16/2803—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants against the immunoglobulin superfamily
- C07K16/2809—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants against the immunoglobulin superfamily against the T-cell receptor (TcR)-CD3 complex
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K40/00—Cellular immunotherapy
- A61K40/10—Cellular immunotherapy characterised by the cell type used
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K40/00—Cellular immunotherapy
- A61K40/40—Cellular immunotherapy characterised by antigens that are targeted or presented by cells of the immune system
- A61K40/46—Viral antigens
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/16—Drugs for disorders of the alimentary tract or the digestive system for liver or gallbladder disorders, e.g. hepatoprotective agents, cholagogues, litholytics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/12—Antivirals
- A61P31/20—Antivirals for DNA viruses
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/02—Immunomodulators
- A61P37/04—Immunostimulants
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/08—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from viruses
- C07K16/081—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from viruses from DNA viruses
- C07K16/082—Hepadnaviridae, e.g. hepatitis B virus
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/28—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants
- C07K16/2803—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants against the immunoglobulin superfamily
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/28—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants
- C07K16/2803—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants against the immunoglobulin superfamily
- C07K16/2818—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants against the immunoglobulin superfamily against CD28 or CD152
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/28—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants
- C07K16/2803—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants against the immunoglobulin superfamily
- C07K16/283—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants against the immunoglobulin superfamily against Fc-receptors, e.g. CD16, CD32, CD64
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/06—Animal cells or tissues; Human cells or tissues
- C12N5/0602—Vertebrate cells
- C12N5/0634—Cells from the blood or the immune system
- C12N5/0636—T lymphocytes
- C12N5/0638—Cytotoxic T lymphocytes [CTL] or lymphokine activated killer cells [LAK]
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/06—Animal cells or tissues; Human cells or tissues
- C12N5/0602—Vertebrate cells
- C12N5/0634—Cells from the blood or the immune system
- C12N5/0646—Natural killers cells [NK], NKT cells
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/505—Medicinal preparations containing antigens or antibodies comprising antibodies
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/505—Medicinal preparations containing antigens or antibodies comprising antibodies
- A61K2039/507—Comprising a combination of two or more separate antibodies
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K2239/00—Indexing codes associated with cellular immunotherapy of group A61K40/00
- A61K2239/31—Indexing codes associated with cellular immunotherapy of group A61K40/00 characterized by the route of administration
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K2239/00—Indexing codes associated with cellular immunotherapy of group A61K40/00
- A61K2239/38—Indexing codes associated with cellular immunotherapy of group A61K40/00 characterised by the dose, timing or administration schedule
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K2239/00—Indexing codes associated with cellular immunotherapy of group A61K40/00
- A61K2239/46—Indexing codes associated with cellular immunotherapy of group A61K40/00 characterised by the cancer treated
- A61K2239/53—Liver
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/10—Immunoglobulins specific features characterized by their source of isolation or production
- C07K2317/14—Specific host cells or culture conditions, e.g. components, pH or temperature
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/30—Immunoglobulins specific features characterized by aspects of specificity or valency
- C07K2317/31—Immunoglobulins specific features characterized by aspects of specificity or valency multispecific
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/30—Immunoglobulins specific features characterized by aspects of specificity or valency
- C07K2317/35—Valency
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/50—Immunoglobulins specific features characterized by immunoglobulin fragments
- C07K2317/52—Constant or Fc region; Isotype
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/50—Immunoglobulins specific features characterized by immunoglobulin fragments
- C07K2317/52—Constant or Fc region; Isotype
- C07K2317/524—CH2 domain
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/50—Immunoglobulins specific features characterized by immunoglobulin fragments
- C07K2317/52—Constant or Fc region; Isotype
- C07K2317/526—CH3 domain
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/50—Immunoglobulins specific features characterized by immunoglobulin fragments
- C07K2317/56—Immunoglobulins specific features characterized by immunoglobulin fragments variable (Fv) region, i.e. VH and/or VL
- C07K2317/565—Complementarity determining region [CDR]
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/60—Immunoglobulins specific features characterized by non-natural combinations of immunoglobulin fragments
- C07K2317/62—Immunoglobulins specific features characterized by non-natural combinations of immunoglobulin fragments comprising only variable region components
- C07K2317/622—Single chain antibody (scFv)
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/70—Immunoglobulins specific features characterized by effect upon binding to a cell or to an antigen
- C07K2317/73—Inducing cell death, e.g. apoptosis, necrosis or inhibition of cell proliferation
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/70—Immunoglobulins specific features characterized by effect upon binding to a cell or to an antigen
- C07K2317/73—Inducing cell death, e.g. apoptosis, necrosis or inhibition of cell proliferation
- C07K2317/732—Antibody-dependent cellular cytotoxicity [ADCC]
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2319/00—Fusion polypeptide
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Immunology (AREA)
- Life Sciences & Earth Sciences (AREA)
- Organic Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Genetics & Genomics (AREA)
- Biochemistry (AREA)
- Biomedical Technology (AREA)
- Medicinal Chemistry (AREA)
- Molecular Biology (AREA)
- Biophysics (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Biotechnology (AREA)
- Zoology (AREA)
- Wood Science & Technology (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Animal Behavior & Ethology (AREA)
- Hematology (AREA)
- Cell Biology (AREA)
- General Engineering & Computer Science (AREA)
- Microbiology (AREA)
- Epidemiology (AREA)
- Virology (AREA)
- Communicable Diseases (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Pharmacology & Pharmacy (AREA)
- General Chemical & Material Sciences (AREA)
- Gastroenterology & Hepatology (AREA)
- Oncology (AREA)
- Peptides Or Proteins (AREA)
- Micro-Organisms Or Cultivation Processes Thereof (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Medicines Containing Antibodies Or Antigens For Use As Internal Diagnostic Agents (AREA)
- Preparation Of Compounds By Using Micro-Organisms (AREA)
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP13184635.4 | 2013-09-16 | ||
| EP13184635 | 2013-09-16 | ||
| PCT/EP2014/069675 WO2015036606A1 (en) | 2013-09-16 | 2014-09-16 | Bi- or multispecific polypeptides binding immune effector cell surface antigens and hbv antigens for treating hbv infections and associated conditions |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| RU2016114504A RU2016114504A (ru) | 2017-10-23 |
| RU2016114504A3 RU2016114504A3 (cg-RX-API-DMAC7.html) | 2018-04-25 |
| RU2671089C2 true RU2671089C2 (ru) | 2018-10-29 |
Family
ID=49165642
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| RU2016114504A RU2671089C2 (ru) | 2013-09-16 | 2014-09-16 | Би- или полиспецифические полипептиды, связывающие поверхностные антигены иммунных эффекторных клеток и антигены hbv для лечения инфекций bv и ассоциированных с ними состояний |
Country Status (8)
| Country | Link |
|---|---|
| US (2) | US10059767B2 (cg-RX-API-DMAC7.html) |
| EP (1) | EP3046938B1 (cg-RX-API-DMAC7.html) |
| JP (1) | JP6599338B2 (cg-RX-API-DMAC7.html) |
| CN (2) | CN105636982B (cg-RX-API-DMAC7.html) |
| CA (1) | CA2924252C (cg-RX-API-DMAC7.html) |
| ES (1) | ES2773306T3 (cg-RX-API-DMAC7.html) |
| RU (1) | RU2671089C2 (cg-RX-API-DMAC7.html) |
| WO (1) | WO2015036606A1 (cg-RX-API-DMAC7.html) |
Families Citing this family (28)
| Publication number | Priority date | Publication date | Assignee | Title |
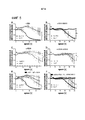
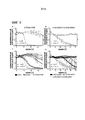
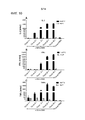
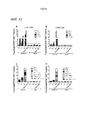
|---|---|---|---|---|
| CA2747011C (en) | 2008-12-18 | 2018-06-19 | Dana-Farber Cancer Institute, Inc. | Nkg2d-fc for immunotherapy |
| US10059767B2 (en) | 2013-09-16 | 2018-08-28 | Helmholtz Zentrum München—Deutsches Forschungszentrum für Gesundheit und Umwelt (GmbH) | Means and methods for treating HBV infection and associated conditions |
| EP3271387B1 (en) * | 2015-03-16 | 2023-12-06 | Helmholtz Zentrum München - Deutsches Forschungszentrum für Gesundheit und Umwelt (GmbH) | Trispecific binding molecules for treating hbv infection and associated conditions |
| CN106349391A (zh) * | 2015-07-17 | 2017-01-25 | 中国科学院深圳先进技术研究院 | Hbv特异性双靶向抗体及其制备方法和应用、含该双靶向抗体表达盒的微环dna及应用 |
| JP7037480B2 (ja) * | 2015-11-13 | 2022-03-16 | デイナ ファーバー キャンサー インスティチュート,インコーポレイテッド | がんの免疫療法のためのnkg2d-ig融合タンパク質 |
| CN105505872B (zh) * | 2016-02-16 | 2019-01-15 | 广州赛莱拉干细胞科技股份有限公司 | 一种致敏nk细胞的方法及组合物 |
| CN107129537A (zh) * | 2016-02-29 | 2017-09-05 | 北京赛诺泰生物科技有限公司 | 抗乙型肝炎病毒的免疫反应性细胞 |
| WO2018064611A1 (en) * | 2016-09-30 | 2018-04-05 | Baylor College Of Medicine | Antibody based gene therapy with tissue-directed expression |
| US11219646B2 (en) * | 2016-09-30 | 2022-01-11 | Baylor College Of Medicine | Chimeric antigen receptor therapy with reduced cytotoxicity for viral disease |
| KR20250089564A (ko) | 2017-02-08 | 2025-06-18 | 드래곤플라이 쎄라퓨틱스, 인크. | 천연 킬러 세포의 활성화를 위한 다중-특이적 결합 단백질 및 암 치료에서의 그의 치료적 용도 |
| KR20190120782A (ko) | 2017-02-20 | 2019-10-24 | 드래곤플라이 쎄라퓨틱스, 인크. | Her2, nkg2d 및 cd16에 결합하는 단백질 |
| PE20210375A1 (es) | 2018-02-08 | 2021-03-02 | Dragonfly Therapeutics Inc | Terapia de combinacion del cancer que incluye proteinas de union multiespecificas que activan a las celulas asesinas naturales |
| CA3090244A1 (en) | 2018-02-08 | 2019-08-15 | Dragonfly Therapeutics, Inc. | Antibody variable domains targeting the nkg2d receptor |
| CN112119093A (zh) | 2018-02-20 | 2020-12-22 | 蜻蜓治疗公司 | 结合cd33、nkg2d和cd16的多特异性结合蛋白和使用方法 |
| EA202091888A1 (ru) | 2018-08-08 | 2020-10-23 | Драгонфлай Терапьютикс, Инк. | Вариабельные домены антител, нацеленные на рецептор nkg2d |
| CA3108427A1 (en) | 2018-08-08 | 2020-02-13 | Dragonfly Therapeutics, Inc. | Multi-specific binding proteins that bind bcma, nkg2d and cd16, and methods of use |
| BR112021002276A2 (pt) | 2018-08-08 | 2021-05-04 | Dragonfly Therapeutics, Inc. | proteínas de ligação a nkg2d, cd16 e um antígeno associado ao tumor |
| CN109265559B (zh) * | 2018-09-25 | 2021-12-07 | 山东兴瑞生物科技有限公司 | 嵌合抗原受体、其制备方法、利用其修饰的nk细胞及治疗hbv感染的应用 |
| SG11202105948TA (en) * | 2018-12-19 | 2021-07-29 | Regeneron Pharma | Bispecific anti-muc16 x anti-cd28 antibodies and uses thereof |
| GB201904328D0 (en) * | 2019-03-28 | 2019-05-15 | Immunocore Ltd | Specific binding molecules |
| KR102503349B1 (ko) | 2019-05-14 | 2023-02-23 | 프로벤션 바이오, 인코포레이티드 | 제1형 당뇨병을 예방하기 위한 방법 및 조성물 |
| US12157771B2 (en) | 2020-05-06 | 2024-12-03 | Dragonfly Therapeutics, Inc. | Proteins binding NKG2D, CD16 and CLEC12A |
| WO2021252917A2 (en) | 2020-06-11 | 2021-12-16 | Provention Bio, Inc. | Methods and compositions for preventing type 1 diabetes |
| WO2022081529A1 (en) * | 2020-10-12 | 2022-04-21 | Greffex, Inc. | Antibody constructs to target t cell responses to sars-cov protein expressing cells, their design and uses |
| CN112592902B (zh) * | 2020-12-16 | 2022-03-29 | 熊猫乳品集团股份有限公司 | 一种生物活性肽aseppvldvkrpflc及其制备方法和应用 |
| WO2022187539A1 (en) | 2021-03-03 | 2022-09-09 | Dragonfly Therapeutics, Inc. | Methods of treating cancer using multi-specific binding proteins that bind nkg2d, cd16 and a tumor-associated antigen |
| CN114573705B (zh) * | 2022-03-17 | 2024-05-14 | 杭州师范大学 | 特异性启动抗乙型肝炎病毒t细胞免疫的双特异性抗体及其应用 |
| KR20250042205A (ko) * | 2022-06-27 | 2025-03-26 | 에스씨지 셀 테라피 피티이. 리미티드 | HBV 표면 항원 (HBsAg) 및 CD3에 대한 이중 특이적 항체 및 이의 용도 |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20050158829A1 (en) * | 2004-01-16 | 2005-07-21 | Fandl James P. | Fusion polypeptides capable of activating receptors |
| US20060127392A1 (en) * | 2002-09-13 | 2006-06-15 | Laboratoire Francais Du Fractionnement Et Des Bioetchnologies | Antibody for adcc and inducing cytokine production |
| WO2008119567A2 (en) * | 2007-04-03 | 2008-10-09 | Micromet Ag | Cross-species-specific cd3-epsilon binding domain |
| EA013564B1 (ru) * | 2000-08-03 | 2010-06-30 | Терапеутик Хьюман Поликлоналз Инк. | Гуманизированный иммуноглобулин и содержащая его фармацевтическая композиция |
| WO2011057124A1 (en) * | 2009-11-06 | 2011-05-12 | Transtarget, Inc. | Polyclonal bispecific antibody compositions and method of use |
| EP2524699A1 (en) * | 2011-05-17 | 2012-11-21 | Trion Research GmbH | Vaccine preparation containing trifunctional antibodies with antigen immunogenicity enhancer properties |
Family Cites Families (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB8928874D0 (en) * | 1989-12-21 | 1990-02-28 | Celltech Ltd | Humanised antibodies |
| CN1294148C (zh) | 2001-04-11 | 2007-01-10 | 中国科学院遗传与发育生物学研究所 | 环状单链三特异抗体 |
| CN100376599C (zh) | 2004-04-01 | 2008-03-26 | 北京安波特基因工程技术有限公司 | 基因工程重组抗cea抗cd3抗cd28线性单链三特异抗体 |
| PT2520590T (pt) * | 2007-04-03 | 2018-11-14 | Amgen Res Munich Gmbh | Domínio de ligação específico de espécies cruzadas |
| WO2012156430A1 (en) * | 2011-05-17 | 2012-11-22 | Trion Research Gmbh | Vaccine preparation containing trifunctional antibodies with antigen immunogenicity enhancer properties |
| EP2797629A4 (en) * | 2011-12-28 | 2015-09-30 | Novelmed Therapeutics Inc | AGLYCOSYLATED HUMAN ANTIBODY AND FUSION PROTEIN AND USES THEREOF |
| US10059767B2 (en) | 2013-09-16 | 2018-08-28 | Helmholtz Zentrum München—Deutsches Forschungszentrum für Gesundheit und Umwelt (GmbH) | Means and methods for treating HBV infection and associated conditions |
-
2014
- 2014-09-16 US US15/021,916 patent/US10059767B2/en active Active
- 2014-09-16 JP JP2016542337A patent/JP6599338B2/ja active Active
- 2014-09-16 EP EP14771250.9A patent/EP3046938B1/en active Active
- 2014-09-16 ES ES14771250T patent/ES2773306T3/es active Active
- 2014-09-16 CN CN201480050976.8A patent/CN105636982B/zh active Active
- 2014-09-16 CA CA2924252A patent/CA2924252C/en active Active
- 2014-09-16 CN CN202010449532.9A patent/CN111606998B/zh active Active
- 2014-09-16 WO PCT/EP2014/069675 patent/WO2015036606A1/en not_active Ceased
- 2014-09-16 RU RU2016114504A patent/RU2671089C2/ru active
-
2018
- 2018-08-24 US US16/112,431 patent/US10730943B2/en active Active
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EA013564B1 (ru) * | 2000-08-03 | 2010-06-30 | Терапеутик Хьюман Поликлоналз Инк. | Гуманизированный иммуноглобулин и содержащая его фармацевтическая композиция |
| US20060127392A1 (en) * | 2002-09-13 | 2006-06-15 | Laboratoire Francais Du Fractionnement Et Des Bioetchnologies | Antibody for adcc and inducing cytokine production |
| US20050158829A1 (en) * | 2004-01-16 | 2005-07-21 | Fandl James P. | Fusion polypeptides capable of activating receptors |
| WO2008119567A2 (en) * | 2007-04-03 | 2008-10-09 | Micromet Ag | Cross-species-specific cd3-epsilon binding domain |
| WO2011057124A1 (en) * | 2009-11-06 | 2011-05-12 | Transtarget, Inc. | Polyclonal bispecific antibody compositions and method of use |
| EP2524699A1 (en) * | 2011-05-17 | 2012-11-21 | Trion Research GmbH | Vaccine preparation containing trifunctional antibodies with antigen immunogenicity enhancer properties |
Non-Patent Citations (2)
| Title |
|---|
| BOHNE F. et al., "T Cells Redirected Against Hepatitis B Virus Surface Proteins Eliminate Infected Hepatocytes", Gastroenterology, 2008, 134: 239-247. * |
| PIZARRO J.C. et al., "Structural and functional characterization of a monoclonal antibody specific for the preS1 region of hepatitis B virus", FEBS Lett., 2001, 509(3):463-8. * |
Also Published As
| Publication number | Publication date |
|---|---|
| ES2773306T3 (es) | 2020-07-10 |
| US10730943B2 (en) | 2020-08-04 |
| RU2016114504A (ru) | 2017-10-23 |
| JP2016530889A (ja) | 2016-10-06 |
| CA2924252A1 (en) | 2015-03-19 |
| RU2016114504A3 (cg-RX-API-DMAC7.html) | 2018-04-25 |
| US20180362649A1 (en) | 2018-12-20 |
| US20160200798A1 (en) | 2016-07-14 |
| CN105636982B (zh) | 2020-06-23 |
| US10059767B2 (en) | 2018-08-28 |
| CA2924252C (en) | 2022-01-04 |
| CN105636982A (zh) | 2016-06-01 |
| CN111606998B (zh) | 2024-05-24 |
| EP3046938A1 (en) | 2016-07-27 |
| JP6599338B2 (ja) | 2019-10-30 |
| EP3046938B1 (en) | 2019-12-04 |
| CN111606998A (zh) | 2020-09-01 |
| WO2015036606A1 (en) | 2015-03-19 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| RU2671089C2 (ru) | Би- или полиспецифические полипептиды, связывающие поверхностные антигены иммунных эффекторных клеток и антигены hbv для лечения инфекций bv и ассоциированных с ними состояний | |
| RU2733496C2 (ru) | Триспецифические связывающие молекулы для лечения вирусной инфекции гепатита в и связанных состояний | |
| US12269881B2 (en) | Antigen binding proteins specifically binding MAGE-A | |
| US20220348689A1 (en) | ADOPTIVE T-CELL THERAPY USING EMPD-SPECIFIC CHIMERIC ANTIGEN RECEPTORS FOR TREATING lgE-MEDIATED ALLERGIC DISEASES | |
| CN107922489A (zh) | κ骨髓瘤抗原嵌合性抗原受体和其用途 | |
| KR20230113578A (ko) | Cldn18.2 항체 및 그의 용도 | |
| JP2023532807A (ja) | Psma及びガンマ-デルタt細胞受容体に結合する抗体 | |
| KR20200039783A (ko) | Cxcr5에 결합하는 키메라 항원 수용체 및 car-t 세포 | |
| JP7278623B2 (ja) | 抗cd27抗体およびその使用 | |
| US20220267420A1 (en) | Foxp3 targeting agent compositions and methods of use for adoptive cell therapy | |
| KR20230069961A (ko) | Cd33에 대한 단일 도메인 항체 | |
| CN119487069A (zh) | 结合剂及其使用方法 | |
| CA2975660C (en) | Trispecific binding molecules for treating hbv infection and associated conditions | |
| JP2025525941A (ja) | EGFRvIIIを標的とする抗体および細胞免疫治療におけるその使用 |