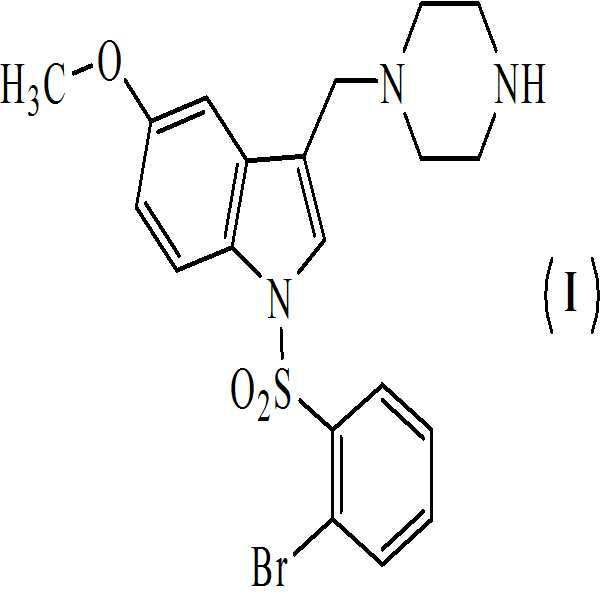
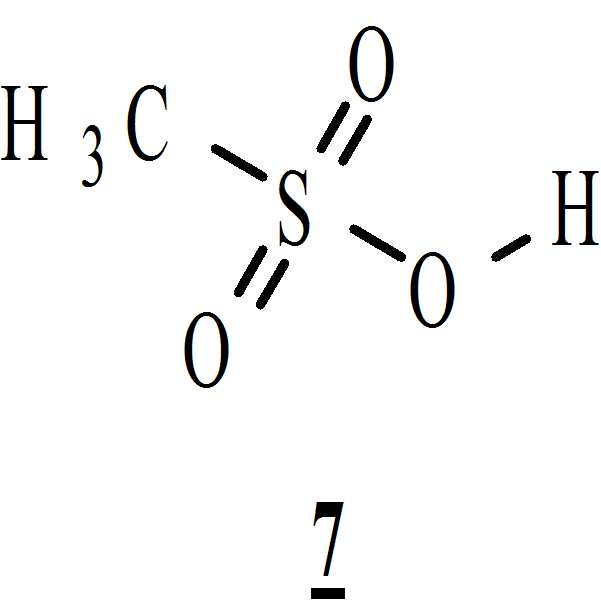KR20170031237A - 1-[(2-브로모페닐) 술포닐]-5-메톡시-3-[(4-메틸-1-피페라지닐) 메틸]-1h-인돌 디메실레이트 모노히드레이트의 활성 대사산물 및 상기 활성 대사산물의 디메실레이트 디히드레이트 염 - Google Patents
1-[(2-브로모페닐) 술포닐]-5-메톡시-3-[(4-메틸-1-피페라지닐) 메틸]-1h-인돌 디메실레이트 모노히드레이트의 활성 대사산물 및 상기 활성 대사산물의 디메실레이트 디히드레이트 염 Download PDFInfo
- Publication number
- KR20170031237A KR20170031237A KR1020177004616A KR20177004616A KR20170031237A KR 20170031237 A KR20170031237 A KR 20170031237A KR 1020177004616 A KR1020177004616 A KR 1020177004616A KR 20177004616 A KR20177004616 A KR 20177004616A KR 20170031237 A KR20170031237 A KR 20170031237A
- Authority
- KR
- South Korea
- Prior art keywords
- methyl
- methoxy
- formula
- sulfonyl
- bromophenyl
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
- -1 dihydrate salt Chemical class 0.000 title claims abstract description 27
- 239000002207 metabolite Substances 0.000 title claims description 6
- XKDCUJBWVGDNSH-UHFFFAOYSA-N 1-(2-bromophenyl)sulfonyl-5-methoxy-3-[(4-methylpiperazin-1-yl)methyl]indole;methanesulfonic acid;hydrate Chemical class O.CS(O)(=O)=O.CS(O)(=O)=O.C12=CC(OC)=CC=C2N(S(=O)(=O)C=2C(=CC=CC=2)Br)C=C1CN1CCN(C)CC1 XKDCUJBWVGDNSH-UHFFFAOYSA-N 0.000 title abstract description 8
- 150000001875 compounds Chemical class 0.000 claims abstract description 39
- QZAYGJVTTNCVMB-UHFFFAOYSA-N serotonin Chemical compound C1=C(O)C=C2C(CCN)=CNC2=C1 QZAYGJVTTNCVMB-UHFFFAOYSA-N 0.000 claims abstract description 22
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 claims abstract description 13
- 201000010099 disease Diseases 0.000 claims abstract description 12
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 claims description 50
- 125000006276 2-bromophenyl group Chemical group [H]C1=C([H])C(Br)=C(*)C([H])=C1[H] 0.000 claims description 37
- 125000000472 sulfonyl group Chemical group *S(*)(=O)=O 0.000 claims description 37
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 claims description 28
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 claims description 24
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 claims description 23
- KWYUFKZDYYNOTN-UHFFFAOYSA-M Potassium hydroxide Chemical compound [OH-].[K+] KWYUFKZDYYNOTN-UHFFFAOYSA-M 0.000 claims description 21
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 claims description 18
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical compound CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 claims description 15
- WSFSSNUMVMOOMR-UHFFFAOYSA-N Formaldehyde Chemical compound O=C WSFSSNUMVMOOMR-UHFFFAOYSA-N 0.000 claims description 14
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 claims description 14
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 13
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 claims description 12
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 claims description 12
- 208000024827 Alzheimer disease Diseases 0.000 claims description 10
- KLWSFLZYJPOGSH-UHFFFAOYSA-N COC1=CC=C2NC=C(CN3CCN(CC3)C(=O)OC(C)(C)C)C2=C1 Chemical compound COC1=CC=C2NC=C(CN3CCN(CC3)C(=O)OC(C)(C)C)C2=C1 KLWSFLZYJPOGSH-UHFFFAOYSA-N 0.000 claims description 9
- 239000008194 pharmaceutical composition Substances 0.000 claims description 9
- 201000000980 schizophrenia Diseases 0.000 claims description 9
- 208000018737 Parkinson disease Diseases 0.000 claims description 8
- 208000006096 Attention Deficit Disorder with Hyperactivity Diseases 0.000 claims description 7
- 208000036864 Attention deficit/hyperactivity disease Diseases 0.000 claims description 7
- AFVFQIVMOAPDHO-UHFFFAOYSA-N Methanesulfonic acid Chemical compound CS(O)(=O)=O AFVFQIVMOAPDHO-UHFFFAOYSA-N 0.000 claims description 7
- 208000015802 attention deficit-hyperactivity disease Diseases 0.000 claims description 7
- CWXPZXBSDSIRCS-UHFFFAOYSA-N tert-butyl piperazine-1-carboxylate Chemical compound CC(C)(C)OC(=O)N1CCNCC1 CWXPZXBSDSIRCS-UHFFFAOYSA-N 0.000 claims description 7
- 239000000126 substance Substances 0.000 claims description 6
- 238000000034 method Methods 0.000 claims description 5
- 239000000546 pharmaceutical excipient Substances 0.000 claims description 5
- 235000011121 sodium hydroxide Nutrition 0.000 claims description 4
- OFZBXXDIKHYMDA-UHFFFAOYSA-N COC1=CC=C2N(C=C(CN3CCNCC3)C2=C1)S(=O)(=O)C1=C(Br)C=CC=C1 Chemical compound COC1=CC=C2N(C=C(CN3CCNCC3)C2=C1)S(=O)(=O)C1=C(Br)C=CC=C1 OFZBXXDIKHYMDA-UHFFFAOYSA-N 0.000 claims description 3
- HEFIWZORGMNALZ-UHFFFAOYSA-N Cl.Cl.COc1ccc2n(cc(CN3CCNCC3)c2c1)S(=O)(=O)c1ccccc1Br Chemical compound Cl.Cl.COc1ccc2n(cc(CN3CCNCC3)c2c1)S(=O)(=O)c1ccccc1Br HEFIWZORGMNALZ-UHFFFAOYSA-N 0.000 claims description 3
- 239000003814 drug Substances 0.000 claims description 3
- 229940098779 methanesulfonic acid Drugs 0.000 claims description 3
- 238000002360 preparation method Methods 0.000 claims description 3
- 239000003751 serotonin 6 antagonist Substances 0.000 claims description 3
- IKCZUPRWPVLSLF-UHFFFAOYSA-N 2-methoxy-1h-indole Chemical compound C1=CC=C2NC(OC)=CC2=C1 IKCZUPRWPVLSLF-UHFFFAOYSA-N 0.000 claims 2
- PJGOLCXVWIYXRQ-UHFFFAOYSA-N 3-bromobenzenesulfonyl chloride Chemical compound ClS(=O)(=O)C1=CC=CC(Br)=C1 PJGOLCXVWIYXRQ-UHFFFAOYSA-N 0.000 claims 2
- SIKJAQJRHWYJAI-UHFFFAOYSA-N Indole Chemical compound C1=CC=C2NC=CC2=C1 SIKJAQJRHWYJAI-UHFFFAOYSA-N 0.000 claims 2
- GLUUGHFHXGJENI-UHFFFAOYSA-N Piperazine Chemical compound C1CNCCN1 GLUUGHFHXGJENI-UHFFFAOYSA-N 0.000 claims 2
- 208000015114 central nervous system disease Diseases 0.000 claims 2
- UAAKJEVCDBPTQS-UHFFFAOYSA-N methanesulfonic acid;dihydrate Chemical compound O.O.CS(O)(=O)=O UAAKJEVCDBPTQS-UHFFFAOYSA-N 0.000 claims 1
- 150000004682 monohydrates Chemical class 0.000 claims 1
- 239000002464 receptor antagonist Substances 0.000 abstract description 4
- 229940044551 receptor antagonist Drugs 0.000 abstract description 4
- 239000000463 material Substances 0.000 description 61
- 238000006243 chemical reaction Methods 0.000 description 53
- 239000000243 solution Substances 0.000 description 37
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 36
- 239000000047 product Substances 0.000 description 24
- 239000012044 organic layer Substances 0.000 description 16
- 238000010992 reflux Methods 0.000 description 13
- 238000005481 NMR spectroscopy Methods 0.000 description 12
- 238000003756 stirring Methods 0.000 description 12
- 230000015572 biosynthetic process Effects 0.000 description 9
- 239000007787 solid Substances 0.000 description 8
- 238000003786 synthesis reaction Methods 0.000 description 7
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 6
- 241001465754 Metazoa Species 0.000 description 6
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 description 6
- 239000000376 reactant Substances 0.000 description 6
- 238000001819 mass spectrum Methods 0.000 description 5
- 239000002002 slurry Substances 0.000 description 5
- HPALAKNZSZLMCH-UHFFFAOYSA-M sodium;chloride;hydrate Chemical class O.[Na+].[Cl-] HPALAKNZSZLMCH-UHFFFAOYSA-M 0.000 description 5
- VFPWGZNNRSQPBT-UHFFFAOYSA-N 2-bromobenzenesulfonyl chloride Chemical compound ClS(=O)(=O)C1=CC=CC=C1Br VFPWGZNNRSQPBT-UHFFFAOYSA-N 0.000 description 4
- DWAQDRSOVMLGRQ-UHFFFAOYSA-N 5-methoxyindole Chemical compound COC1=CC=C2NC=CC2=C1 DWAQDRSOVMLGRQ-UHFFFAOYSA-N 0.000 description 4
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 4
- WCUXLLCKKVVCTQ-UHFFFAOYSA-M Potassium chloride Chemical compound [Cl-].[K+] WCUXLLCKKVVCTQ-UHFFFAOYSA-M 0.000 description 4
- 238000004519 manufacturing process Methods 0.000 description 4
- 239000000203 mixture Substances 0.000 description 4
- 108020003175 receptors Proteins 0.000 description 4
- 239000002904 solvent Substances 0.000 description 4
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 3
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 3
- 241000700159 Rattus Species 0.000 description 3
- 239000005557 antagonist Substances 0.000 description 3
- 238000011161 development Methods 0.000 description 3
- 238000010438 heat treatment Methods 0.000 description 3
- 239000010410 layer Substances 0.000 description 3
- 230000004060 metabolic process Effects 0.000 description 3
- 229910052757 nitrogen Inorganic materials 0.000 description 3
- 239000008188 pellet Substances 0.000 description 3
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 2
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 2
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 2
- HEDRZPFGACZZDS-MICDWDOJSA-N Trichloro(2H)methane Chemical compound [2H]C(Cl)(Cl)Cl HEDRZPFGACZZDS-MICDWDOJSA-N 0.000 description 2
- 210000004556 brain Anatomy 0.000 description 2
- 230000019771 cognition Effects 0.000 description 2
- 230000037213 diet Effects 0.000 description 2
- 235000005911 diet Nutrition 0.000 description 2
- 150000004683 dihydrates Chemical class 0.000 description 2
- 230000000694 effects Effects 0.000 description 2
- 239000003480 eluent Substances 0.000 description 2
- 239000008098 formaldehyde solution Substances 0.000 description 2
- 238000010230 functional analysis Methods 0.000 description 2
- 239000011521 glass Substances 0.000 description 2
- 239000005457 ice water Substances 0.000 description 2
- 238000002955 isolation Methods 0.000 description 2
- 238000004895 liquid chromatography mass spectrometry Methods 0.000 description 2
- 239000002609 medium Substances 0.000 description 2
- BDAGIHXWWSANSR-UHFFFAOYSA-N methanoic acid Natural products OC=O BDAGIHXWWSANSR-UHFFFAOYSA-N 0.000 description 2
- 238000012544 monitoring process Methods 0.000 description 2
- 238000002414 normal-phase solid-phase extraction Methods 0.000 description 2
- 230000000144 pharmacologic effect Effects 0.000 description 2
- 239000012071 phase Substances 0.000 description 2
- 239000001103 potassium chloride Substances 0.000 description 2
- 235000011164 potassium chloride Nutrition 0.000 description 2
- 239000000843 powder Substances 0.000 description 2
- 238000011160 research Methods 0.000 description 2
- 238000012552 review Methods 0.000 description 2
- 229940076279 serotonin Drugs 0.000 description 2
- 239000012265 solid product Substances 0.000 description 2
- 239000007858 starting material Substances 0.000 description 2
- 239000012257 stirred material Substances 0.000 description 2
- 208000024891 symptom Diseases 0.000 description 2
- 238000012360 testing method Methods 0.000 description 2
- 238000004809 thin layer chromatography Methods 0.000 description 2
- IMSOOSHLIQJKBL-UHFFFAOYSA-N 1-(2-bromophenyl)sulfonyl-5-(piperazin-1-ylmethyl)indole;dihydrochloride Chemical compound Cl.Cl.BrC1=CC=CC=C1S(=O)(=O)N1C2=CC=C(CN3CCNCC3)C=C2C=C1 IMSOOSHLIQJKBL-UHFFFAOYSA-N 0.000 description 1
- JIFDEFRYXOCNOI-UHFFFAOYSA-N 1-[(4-methylpiperazin-1-yl)methyl]indole Chemical compound C1CN(C)CCN1CN1C2=CC=CC=C2C=C1 JIFDEFRYXOCNOI-UHFFFAOYSA-N 0.000 description 1
- OSWFIVFLDKOXQC-UHFFFAOYSA-N 4-(3-methoxyphenyl)aniline Chemical compound COC1=CC=CC(C=2C=CC(N)=CC=2)=C1 OSWFIVFLDKOXQC-UHFFFAOYSA-N 0.000 description 1
- 108091005435 5-HT6 receptors Proteins 0.000 description 1
- USFZMSVCRYTOJT-UHFFFAOYSA-N Ammonium acetate Chemical compound N.CC(O)=O USFZMSVCRYTOJT-UHFFFAOYSA-N 0.000 description 1
- 239000005695 Ammonium acetate Substances 0.000 description 1
- ATRHOXITHDTKAU-UHFFFAOYSA-N CS(O)(=O)=O.CS(O)(=O)=O.COc1ccc2n(cc(CN3CCNCC3)c2c1)S(=O)(=O)c1ccccc1Br Chemical compound CS(O)(=O)=O.CS(O)(=O)=O.COc1ccc2n(cc(CN3CCNCC3)c2c1)S(=O)(=O)c1ccccc1Br ATRHOXITHDTKAU-UHFFFAOYSA-N 0.000 description 1
- 108060001084 Luciferase Proteins 0.000 description 1
- 239000005089 Luciferase Substances 0.000 description 1
- 208000026139 Memory disease Diseases 0.000 description 1
- 239000012124 Opti-MEM Substances 0.000 description 1
- 108700008625 Reporter Genes Proteins 0.000 description 1
- XDJLMELDRAPCKG-UHFFFAOYSA-N S(C)(=O)(=O)O.S(C)(=O)(=O)O.BrC1=C(C=CC=C1)S(=O)(=O)N1C=C(C2=CC(=CC=C12)OC)C Chemical compound S(C)(=O)(=O)O.S(C)(=O)(=O)O.BrC1=C(C=CC=C1)S(=O)(=O)N1C=C(C2=CC(=CC=C12)OC)C XDJLMELDRAPCKG-UHFFFAOYSA-N 0.000 description 1
- 239000000556 agonist Substances 0.000 description 1
- 235000019257 ammonium acetate Nutrition 0.000 description 1
- 229940043376 ammonium acetate Drugs 0.000 description 1
- 238000004458 analytical method Methods 0.000 description 1
- 238000010171 animal model Methods 0.000 description 1
- 238000003556 assay Methods 0.000 description 1
- 230000009286 beneficial effect Effects 0.000 description 1
- 238000004166 bioassay Methods 0.000 description 1
- 150000001720 carbohydrates Chemical class 0.000 description 1
- 235000014633 carbohydrates Nutrition 0.000 description 1
- 210000003169 central nervous system Anatomy 0.000 description 1
- 239000003153 chemical reaction reagent Substances 0.000 description 1
- 238000004587 chromatography analysis Methods 0.000 description 1
- 208000010877 cognitive disease Diseases 0.000 description 1
- 230000037410 cognitive enhancement Effects 0.000 description 1
- 238000004440 column chromatography Methods 0.000 description 1
- 230000002950 deficient Effects 0.000 description 1
- 239000008367 deionised water Substances 0.000 description 1
- 229910021641 deionized water Inorganic materials 0.000 description 1
- 230000001934 delay Effects 0.000 description 1
- OKKJLVBELUTLKV-MZCSYVLQSA-N deuterated methanol Substances [2H]OC([2H])([2H])[2H] OKKJLVBELUTLKV-MZCSYVLQSA-N 0.000 description 1
- 239000003085 diluting agent Substances 0.000 description 1
- 238000004090 dissolution Methods 0.000 description 1
- 238000001035 drying Methods 0.000 description 1
- 230000008030 elimination Effects 0.000 description 1
- 238000003379 elimination reaction Methods 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 239000000706 filtrate Substances 0.000 description 1
- 238000003818 flash chromatography Methods 0.000 description 1
- 235000013305 food Nutrition 0.000 description 1
- 235000019253 formic acid Nutrition 0.000 description 1
- 208000013403 hyperactivity Diseases 0.000 description 1
- 239000012535 impurity Substances 0.000 description 1
- 238000011534 incubation Methods 0.000 description 1
- 239000003446 ligand Substances 0.000 description 1
- 238000004020 luminiscence type Methods 0.000 description 1
- 239000012139 lysis buffer Substances 0.000 description 1
- 238000002844 melting Methods 0.000 description 1
- 230000008018 melting Effects 0.000 description 1
- ZXGSCMYBKMTEQA-UHFFFAOYSA-M potassium 2-bromobenzenesulfonyl chloride hydroxide Chemical compound BrC1=C(C=CC=C1)S(=O)(=O)Cl.[OH-].[K+] ZXGSCMYBKMTEQA-UHFFFAOYSA-M 0.000 description 1
- 238000000425 proton nuclear magnetic resonance spectrum Methods 0.000 description 1
- 238000000746 purification Methods 0.000 description 1
- 238000000611 regression analysis Methods 0.000 description 1
- 150000003839 salts Chemical class 0.000 description 1
- 210000002966 serum Anatomy 0.000 description 1
- 239000012679 serum free medium Substances 0.000 description 1
- 239000000741 silica gel Substances 0.000 description 1
- 229910002027 silica gel Inorganic materials 0.000 description 1
- 229910052938 sodium sulfate Inorganic materials 0.000 description 1
- 235000011152 sodium sulphate Nutrition 0.000 description 1
- 239000011343 solid material Substances 0.000 description 1
- 230000001225 therapeutic effect Effects 0.000 description 1
- 231100000027 toxicology Toxicity 0.000 description 1
- 239000003643 water by type Substances 0.000 description 1
- 238000010626 work up procedure Methods 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/40—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with one nitrogen as the only ring hetero atom, e.g. sulpiride, succinimide, tolmetin, buflomedil
- A61K31/403—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with one nitrogen as the only ring hetero atom, e.g. sulpiride, succinimide, tolmetin, buflomedil condensed with carbocyclic rings, e.g. carbazole
- A61K31/4035—Isoindoles, e.g. phthalimide
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/40—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with one nitrogen as the only ring hetero atom, e.g. sulpiride, succinimide, tolmetin, buflomedil
- A61K31/403—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with one nitrogen as the only ring hetero atom, e.g. sulpiride, succinimide, tolmetin, buflomedil condensed with carbocyclic rings, e.g. carbazole
- A61K31/404—Indoles, e.g. pindolol
- A61K31/4045—Indole-alkylamines; Amides thereof, e.g. serotonin, melatonin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/496—Non-condensed piperazines containing further heterocyclic rings, e.g. rifampin, thiothixene or sparfloxacin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/14—Drugs for disorders of the nervous system for treating abnormal movements, e.g. chorea, dyskinesia
- A61P25/16—Anti-Parkinson drugs
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/18—Antipsychotics, i.e. neuroleptics; Drugs for mania or schizophrenia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/28—Drugs for disorders of the nervous system for treating neurodegenerative disorders of the central nervous system, e.g. nootropic agents, cognition enhancers, drugs for treating Alzheimer's disease or other forms of dementia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C309/00—Sulfonic acids; Halides, esters, or anhydrides thereof
- C07C309/01—Sulfonic acids
- C07C309/02—Sulfonic acids having sulfo groups bound to acyclic carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C317/00—Sulfones; Sulfoxides
- C07C317/14—Sulfones; Sulfoxides having sulfone or sulfoxide groups bound to carbon atoms of six-membered aromatic rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D209/00—Heterocyclic compounds containing five-membered rings, condensed with other rings, with one nitrogen atom as the only ring hetero atom
- C07D209/02—Heterocyclic compounds containing five-membered rings, condensed with other rings, with one nitrogen atom as the only ring hetero atom condensed with one carbocyclic ring
- C07D209/04—Indoles; Hydrogenated indoles
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D209/00—Heterocyclic compounds containing five-membered rings, condensed with other rings, with one nitrogen atom as the only ring hetero atom
- C07D209/02—Heterocyclic compounds containing five-membered rings, condensed with other rings, with one nitrogen atom as the only ring hetero atom condensed with one carbocyclic ring
- C07D209/04—Indoles; Hydrogenated indoles
- C07D209/10—Indoles; Hydrogenated indoles with substituted hydrocarbon radicals attached to carbon atoms of the hetero ring
- C07D209/14—Radicals substituted by nitrogen atoms, not forming part of a nitro radical
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D403/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00
- C07D403/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing two hetero rings
- C07D403/06—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing two hetero rings linked by a carbon chain containing only aliphatic carbon atoms
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Veterinary Medicine (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Engineering & Computer Science (AREA)
- Neurosurgery (AREA)
- Biomedical Technology (AREA)
- Neurology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Epidemiology (AREA)
- Psychiatry (AREA)
- Hospice & Palliative Care (AREA)
- Psychology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Indole Compounds (AREA)
- Plural Heterocyclic Compounds (AREA)
Abstract
Description
도 2는 1-[(2-브로모 페닐)술포닐]-5-메톡시-3-[(1-피페라지닐) 메틸]-1H-인돌 (실시예 1)의 질량 스펙트럼 및 MS-단편 화학 구조를 나타낸다.
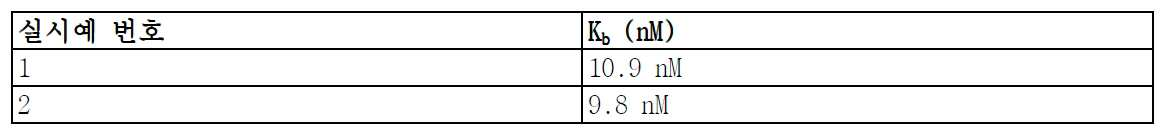
도 3은 실시예 1의 백분율 선택 정확도를 나타낸다.
도 4는 실시예 2의 백분율 선택 정확도를 나타낸다.
Claims (16)
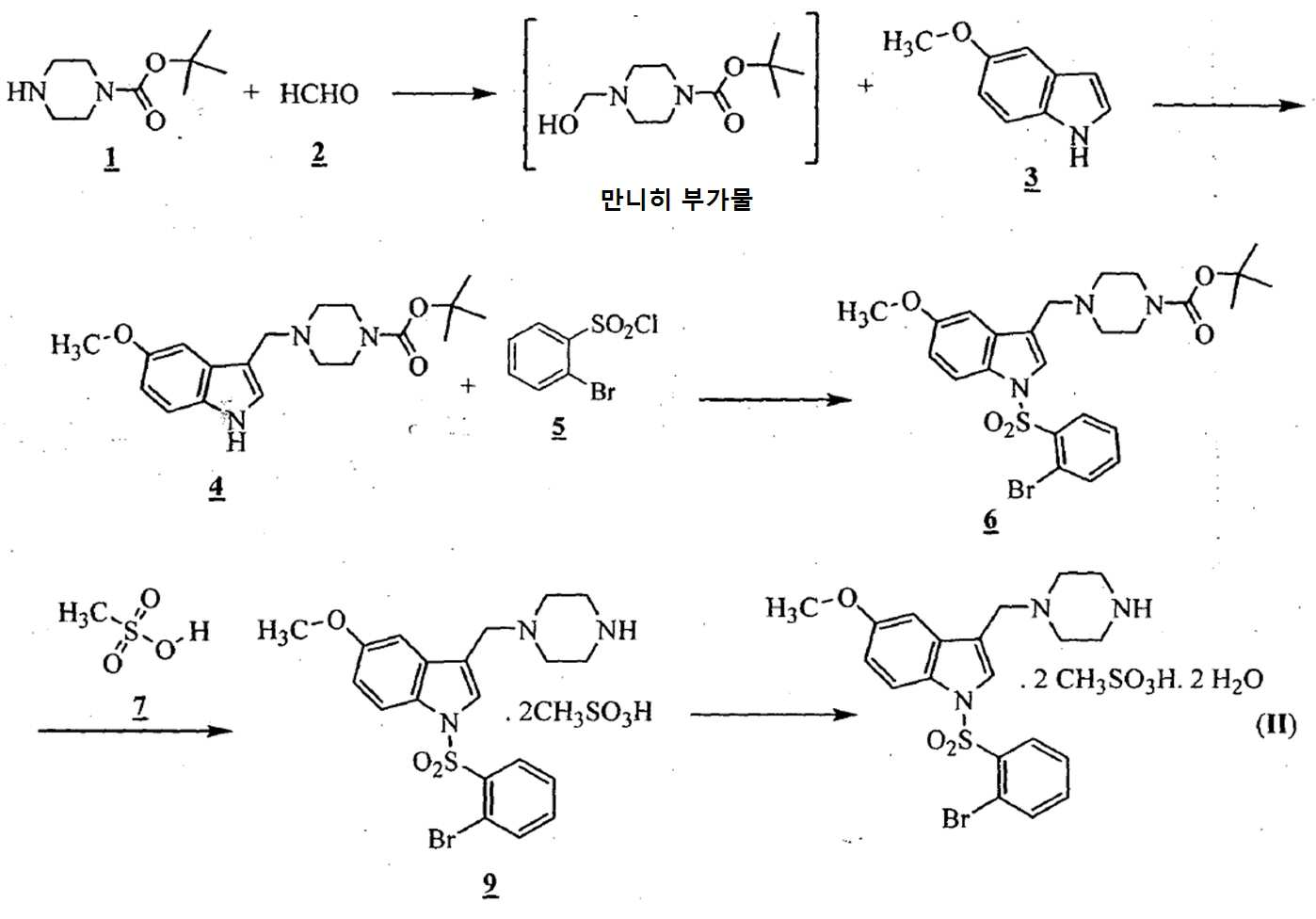
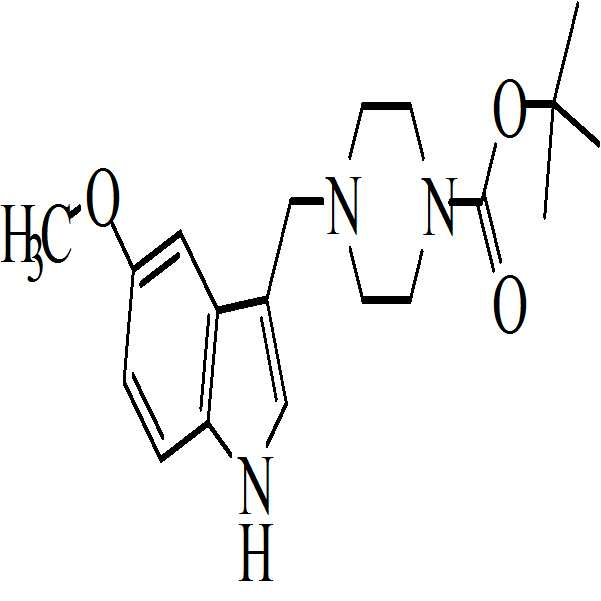
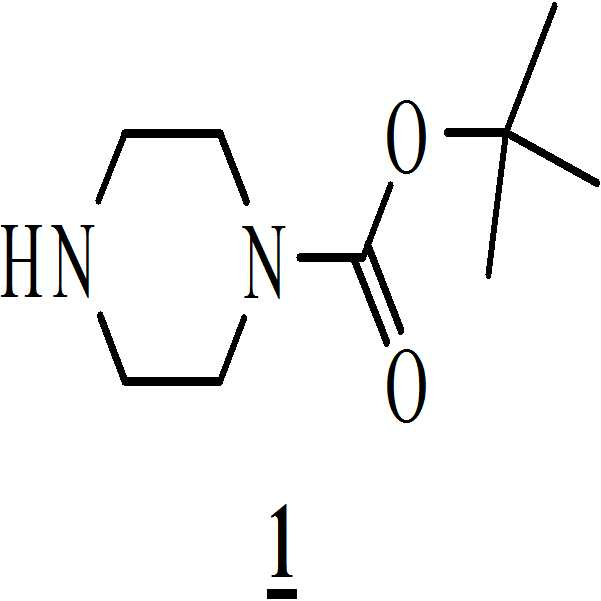
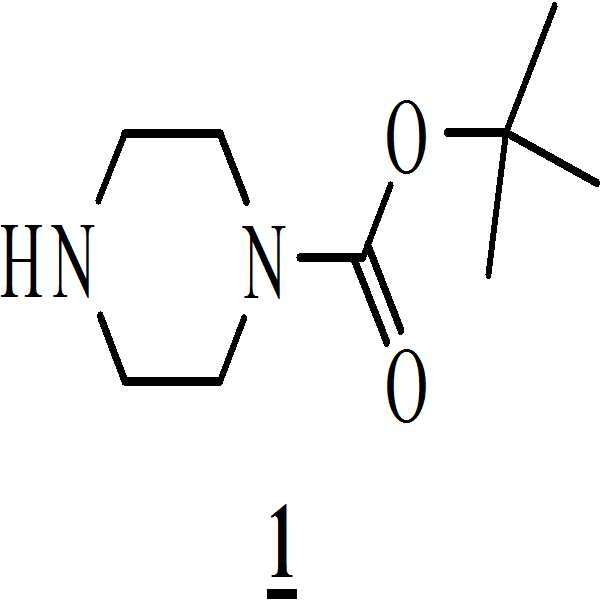
- 아세트산 및 하기의 화학식 2의 수성 포름알데히드 존재하에 하기의 화학식 1의 N-Boc 피페라진을 반응시켜 만니히 부가물(Mannich adduct)을 수득하는 단계 (i)
;
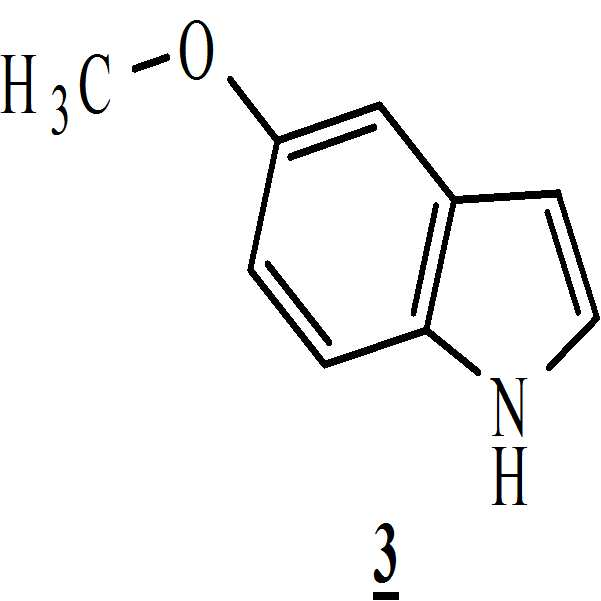
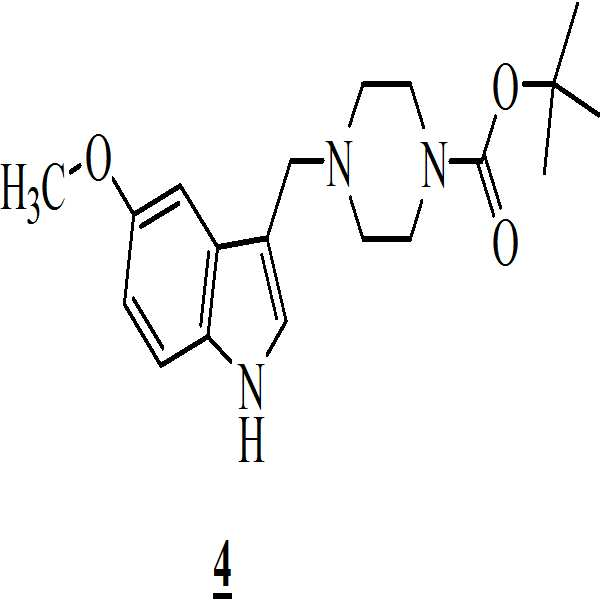
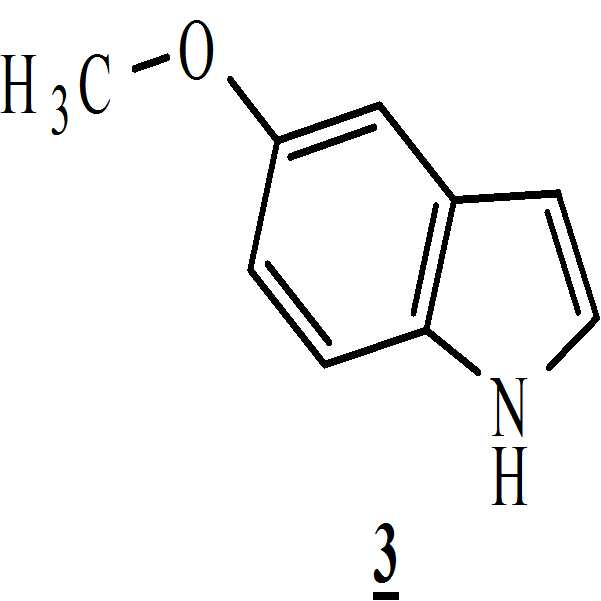
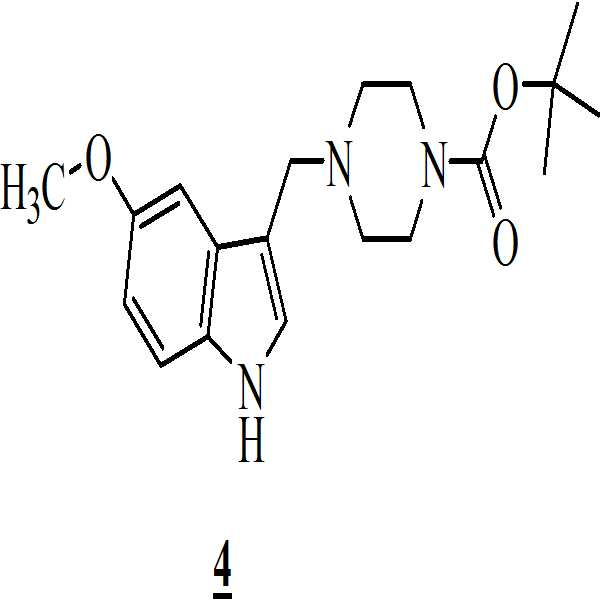
메탄올 존재하에 상기 만니히 부가물을 하기의 화학식 3의 5-메톡시인돌과 반응시켜 화학식 4의 3-[(1-t-부틸옥시카보닐 피페라진-4-일)메틸]-5-메톡시-1H-인돌을 수득하는 단계 (ii)
;
n-헥산을 이용함으로써 상기 화학식 4의 3-[(1-t-부틸옥시카보닐 피페라진-4-일)메틸]-5-메톡시-1H-인돌을 정제하는 단계 (iii);
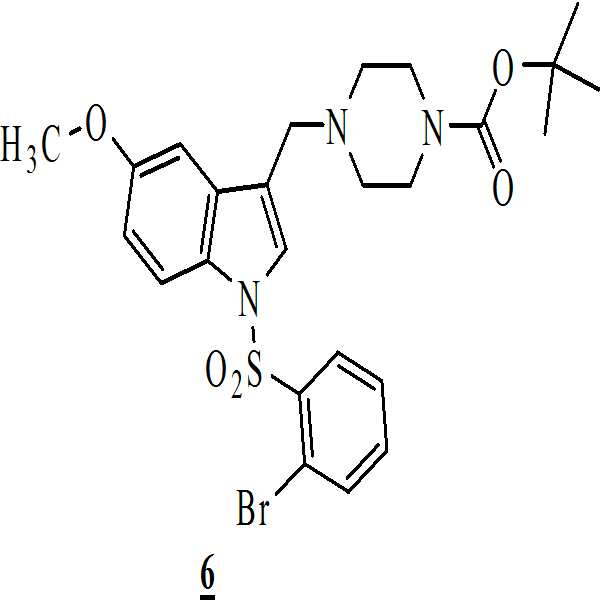
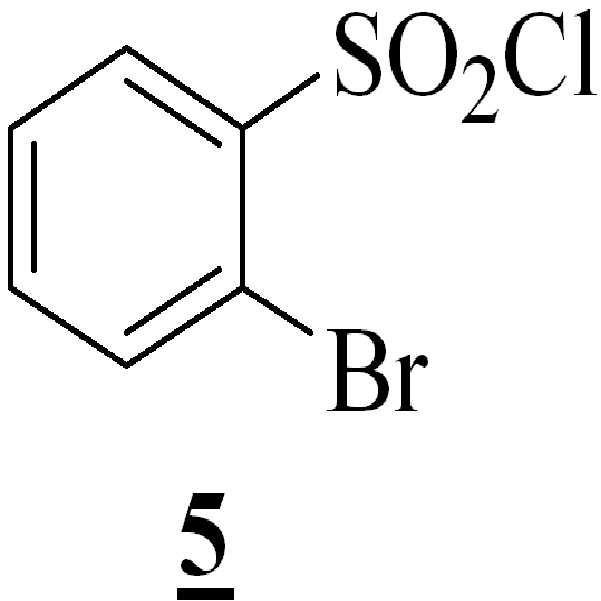
수산화칼륨의 존재하에서 테트라히드로퓨란 중에 상기 수득된 화학식 4의 3-[(1-t-부틸옥시카보닐 피페라진-4-일)메틸]-5-메톡시-1H-인돌을 하기의 화학식 5의 2-브로모페닐술포닐 클로리드와 반응시켜 하기의 화학식 6의 1-[(2-브로모페닐)술포닐]-5-메톡시-3-[(1-t-부틸옥시카보닐 피페라진-4-일)메틸]-1H-인돌을 수득하는 단계 (iv)
;
이소프로판올 및 메탄올을 이용함으로써 상기 화학식 6의 1-[(2-브로모페닐)술포닐]-5-메톡시-3-[(1-t-부틸옥시카보닐 피페라진-4-일)메틸]-1H-인돌을 정제하는 단계 (v);
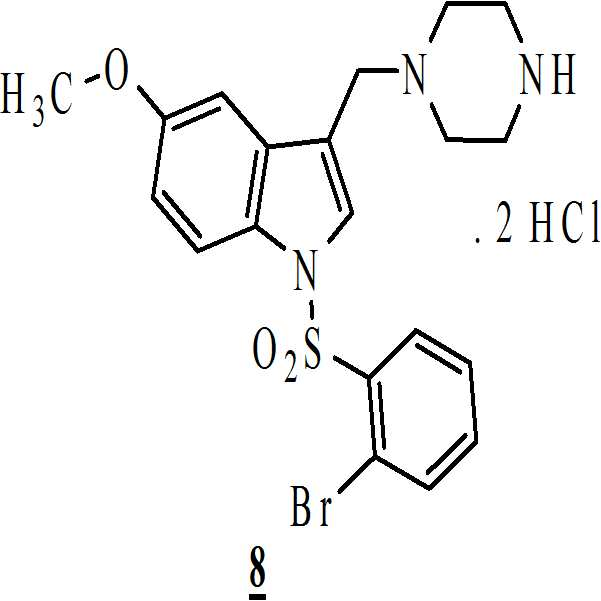
무수 에탄올(absolute ethanol) 및 수성 염산의 존재하에 상기 수득된 화학식 6의 1-[(2-브로모페닐)술포닐]-5-메톡시-3-[(1-t-부틸옥시카보닐 피페라진-4-일)메틸]-1H-인돌을 전환시켜 하기의 화학식 8의 1-[(2-브로모페닐)술포닐]-5-메톡시-3-[(1-피페라지닐)메틸]-1H-인돌 디히드로클로리드를 수득하는 단계 (vi)
; 및
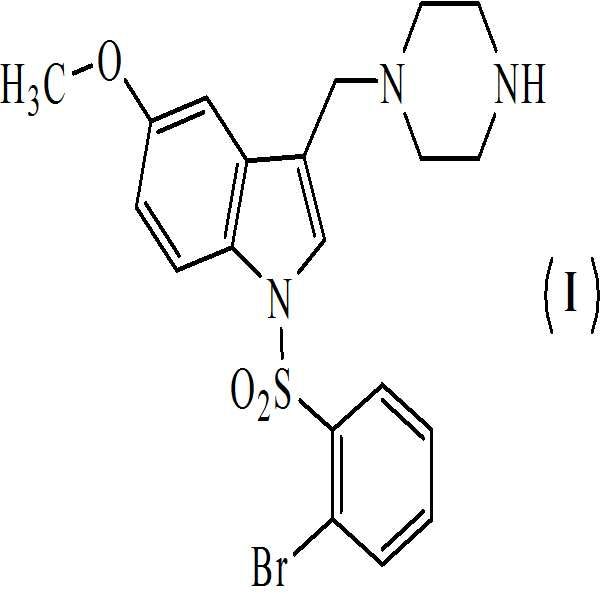
상기 수득된 화학식 8의 1-[(2-브로모페닐)술포닐]-5-메톡시-3-[(피페라진-1-일)메틸]-1H-인돌 디히드로클로리드를 물에 용해시키고, 40 % (w/w) 가성소다(lye) 용액을 첨가함으로써 pH를 10.5 내지 11로 염기성화시켜 화학식 (Ⅰ)의 1-[(2-브로모페닐)술포닐]-5-메톡시-3-[(1-피페라지닐)메틸]-1H-인돌을 수득하는 단계 (vii)를 포함하는 청구항 1의 화학식 (Ⅰ)의 화합물을 제조하는 방법. - 아세트산 및 하기의 화학식 2의 수성 포름알데히드의 존재하에 하기의 화학식 1의 N-Boc 피페라진을 반응시켜 만니히 부가물을 수득하는 단계 (i)
;
메탄올 존재하에 상기 만니히 부가물을 하기의 화학식 3의 5-메톡시인돌과 반응시켜 하기의 화학식 4의 3-[(1-t-부틸옥시카보닐 피페라진-4-일)메틸]-5-메톡시-1H-인돌을 수득하는 단계 (ii)
;
n-헥산을 이용함으로써 상기 화학식 4의 3-[(1-t-부틸옥시카보닐 피페라진-4-일)메틸]-5-메톡시-1H-인돌을 정제하는 단계 (iii);
수산화칼륨의 존재하에서 테트라히드로퓨란 중에 상기 수득된 화학식 4의 3-[(1-t-부틸옥시카보닐 피페라진-4-일)메틸]-5-메톡시-1H-인돌을 하기의 화학식 5의 2-브로모페닐술포닐 클로리드와 반응시켜 하기의 화학식 6의 1-[(2-브로모페닐)술포닐]-5-메톡시-3-[(1-t-부틸옥시카보닐 피페라진-4-일)메틸]-1H-인돌을 수득하는 단계 (iv)
;
이소프로판올 및 메탄올을 이용함으로써 상기 화학식 6의 1-[(2-브로모페닐)술포닐]-5-메톡시-3-[(1-t-부틸옥시카보닐 피페라진-4-일)메틸]-1H-인돌을 정제하는 단계 (v);
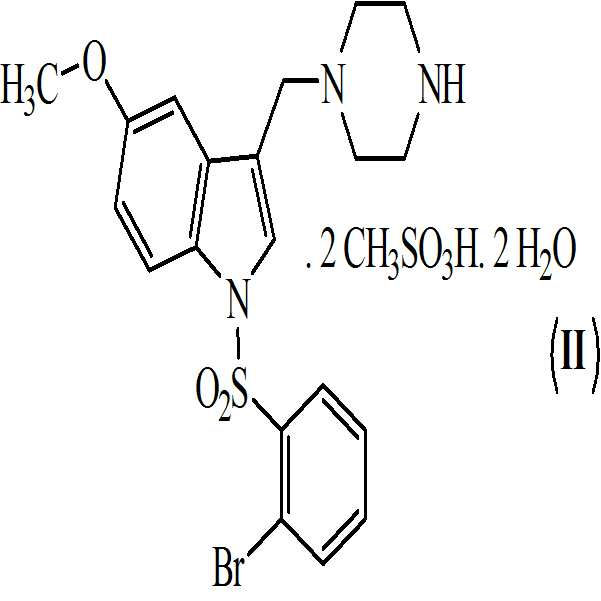
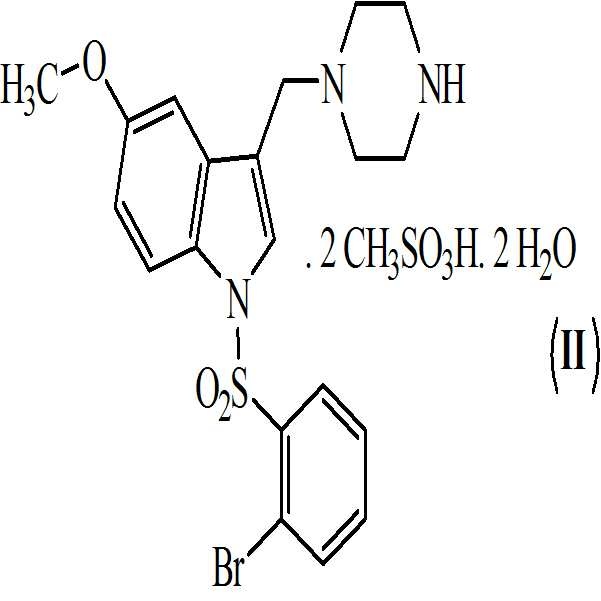
아세톤 및 하기의 화학식 7의 메탄술폰산의 존재하에 상기 수득된 화학식 6의 1-[(2-브로모페닐)술포닐]-5-메톡시-3-[(1-t-부틸옥시카보닐 피페라진-4-일)메틸]-1H-인돌을 전환시켜 하기의 화학식 9의 1-[(2-브로모페닐)술포닐]-5-메톡시-3-[(1-피페라지닐)메틸]-1H-인돌 디메실레이트를 수득하는 단계 (vi)
; 및
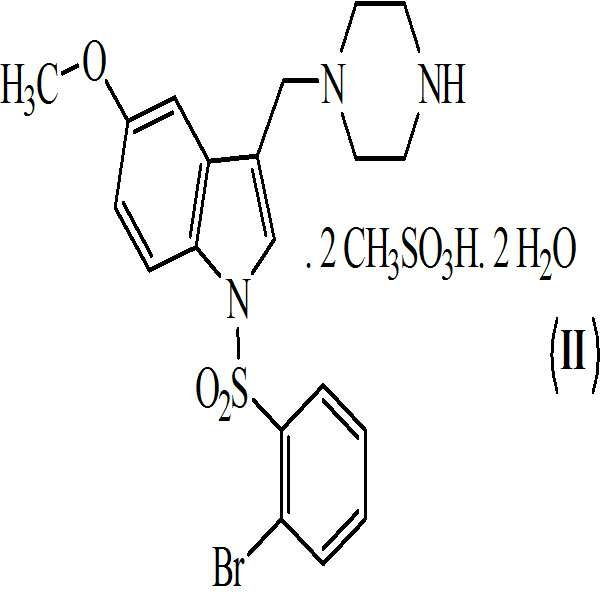
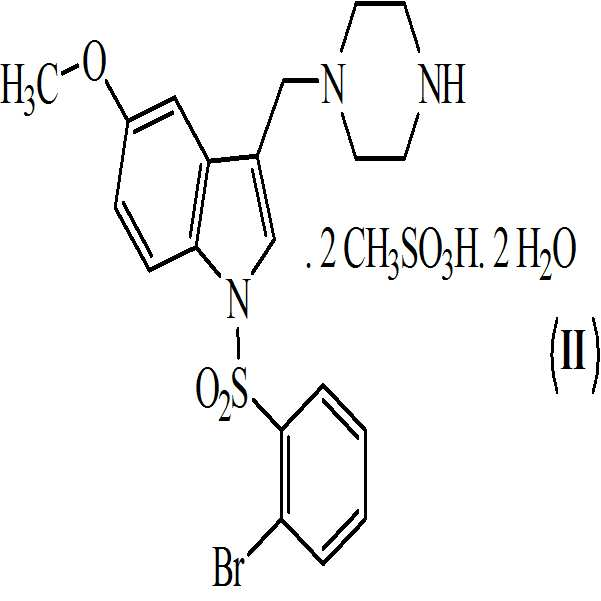
상기 수득된 화학식 9의 1-[(2-브로모페닐)술포닐]-5-메톡시-3-[(1-피페라지닐)메틸]-1H-인돌 디메실레이트를 55 내지 60℃로 가열함으로써 물 및 아세톤에 용해시켜, 화학식 (Ⅱ)의 1-[(2-브로모페닐)술포닐]-5-메톡시-3-[(1-피페라지닐)메틸]-1H-인돌 디메실레이트 디히드레이트를 수득하는 단계 (vii)를 포함하는 청구항 3의 화학식 (Ⅱ)의 화합물을 제조하는 방법. - 청구항 1의 화합물 및 약학적으로 허용가능한 부형제를 포함하는 약학적 조성물.
- 청구항 5에 있어서, 알츠하이머병, 주의력 결핍 과다활동 장애, 파킨슨병 및 정신분열증의 치료를 위한 것인 약학적 조성물.
- 청구항 1의 화합물 유효량을 이를 필요로 하는 환자에게 투여하는 단계를 포함하는 알츠하이머병, 주의력 결핍 과다활동 장애, 파킨슨병 및 정신분열증을 치료하는 방법.
- 5-HT6 수용체 길항제와 관련된 질환의 치료를 위한 청구항 1의 화합물의 용도.
- 청구항 6에 있어서, 알츠하이머병, 주의력 결핍 과다활동 장애, 파킨슨병 및 정신분열증 치료를 위한 것인 용도.
- 청구항 3의 화합물 및 약학적으로 허용가능한 부형제를 포함하는 약학적 조성물.
- 청구항 10에 있어서, 알츠하이머병, 주의력 결핍 과다활동 장애, 파킨슨병 및 정신분열증의 치료를 위한 것인 약학적 조성물.
- 청구항 3의 화합물 유효량을 이를 필요로하는 환자에게 투여하는 단계를 포함하는 알츠하이머병, 주의력 결핍 과다활동 장애, 파킨슨병 및 정신분열증을 치료하는 방법.
- 5-HT6 수용체 길항제와 관련된 질환의 치료를 위한 청구항 3의 화합물의 용도.
- 청구항 13에 있어서, 알츠하이머병, 주의력 결핍 과다활동 장애, 파킨슨병 및 정신분열증의 치료를 위한 것인 용도.
- 청구항 1에 있어서, 5-HT6 수용체와 관련되거나 이에 영향받는 중추 신경계 질환의 치료용 약제의 제조를 위한 것인, 화학식 (Ⅰ)의 화합물.
- 청구항 3에 있어서, 5-HT6 수용체와 관련되거나 이에 영향받는 중추 신경계 질환의 치료용 약제의 제조를 위한 것인, 화학식 (Ⅱ)의 화합물.
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| IN4011/CHE/2014 | 2014-08-16 | ||
| IN4011CH2014 | 2014-08-16 | ||
| PCT/IN2014/000667 WO2016027276A1 (en) | 2014-08-16 | 2014-10-20 | Active metabolite of 1-[(2-bromophenyl) sulfonyl]-5-methoxy-3- [(4-methyl-1-piperazinyl) methyl]-1h-indole dimesylate monohydrate and dimesylate dihydrate salt of active metabolite |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| KR20170031237A true KR20170031237A (ko) | 2017-03-20 |
| KR101938408B1 KR101938408B1 (ko) | 2019-01-14 |
Family
ID=52345478
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020177004616A Active KR101938408B1 (ko) | 2014-08-16 | 2014-10-20 | 1-[(2-브로모페닐) 술포닐]-5-메톡시-3-[(4-메틸-1-피페라지닐) 메틸]-1h-인돌 디메실레이트 모노히드레이트의 활성 대사산물 및 상기 활성 대사산물의 디메실레이트 디히드레이트 염 |
Country Status (28)
| Country | Link |
|---|---|
| US (1) | US10010527B2 (ko) |
| EP (1) | EP3180001B1 (ko) |
| JP (1) | JP6629322B2 (ko) |
| KR (1) | KR101938408B1 (ko) |
| CN (1) | CN106794170A (ko) |
| AP (1) | AP2017009752A0 (ko) |
| AU (1) | AU2014404000B2 (ko) |
| BR (1) | BR112017002921B1 (ko) |
| CA (1) | CA2957497C (ko) |
| CY (1) | CY1121393T1 (ko) |
| DK (1) | DK3180001T3 (ko) |
| EA (1) | EA031319B1 (ko) |
| ES (1) | ES2715113T3 (ko) |
| HR (1) | HRP20190446T1 (ko) |
| HU (1) | HUE044011T2 (ko) |
| IL (1) | IL250411B (ko) |
| LT (1) | LT3180001T (ko) |
| MX (1) | MX375538B (ko) |
| NZ (1) | NZ728907A (ko) |
| PL (1) | PL3180001T3 (ko) |
| PT (1) | PT3180001T (ko) |
| RS (1) | RS58575B1 (ko) |
| SG (1) | SG11201701106RA (ko) |
| SI (1) | SI3180001T1 (ko) |
| SM (1) | SMT201900147T1 (ko) |
| TR (1) | TR201903344T4 (ko) |
| WO (1) | WO2016027276A1 (ko) |
| ZA (1) | ZA201700915B (ko) |
Families Citing this family (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| HUE050819T2 (hu) * | 2016-05-18 | 2021-01-28 | Suven Life Sciences Ltd | Tiszta 5-HT6 receptor antagonisták kombinációja acetilkolinészteráz inhibitorokkal |
| BR112018073396A2 (pt) * | 2016-05-18 | 2019-03-19 | Suven Life Sciences Limited | combinação de antagonistas, uso de uma combinação, método para o tratamento de desordens cognitivas, composto, método para o tratamento de doença de alzheimer e composição farmacêutica |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR20050084185A (ko) * | 2002-12-18 | 2005-08-26 | 수벤 라이프 사이언시스 리미티드 | 세로토닌 수용체 친화력을 가지는 테트라사이클릭 3-치환인돌 |
| KR20050086868A (ko) * | 2002-11-28 | 2005-08-30 | 수벤 라이프 사이언시스 리미티드 | 세로토닌 수용체 친화력을 가지는 엔-아릴술포닐-3-치환인돌, 그 제조방법 및 그를 포함하는 약제학적 조성물 |
| KR100823908B1 (ko) * | 2000-10-20 | 2008-04-21 | 바이오비트럼 에이비(피유비엘) | 2-, 3-, 4-, 또는 5-치환-n1-(벤젠술포닐)인돌 및 이의치료 용도 |
| KR20100014413A (ko) * | 2007-02-16 | 2010-02-10 | 메모리 파마슈티칼스 코포레이션 | 5-ht6 수용체 친화력을 갖는 6' 치환된 인돌 및 인다졸 유도체 |
| KR20100069707A (ko) * | 2007-10-26 | 2010-06-24 | 수벤 라이프 사이언시스 리미티드 | 아미노 아릴설폰아마이드 화합물 및 5―ht6 리간드로서의 이들의 용도 |
Family Cites Families (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE60322186D1 (de) * | 2002-02-12 | 2008-08-28 | Organon Nv | 1-arylsulfonyl-3-substituierte indol und indolinederivate verwendbar zur behandlung von erkrankungen des zentralnervensystem |
-
2014
- 2014-10-20 PL PL14825443T patent/PL3180001T3/pl unknown
- 2014-10-20 AU AU2014404000A patent/AU2014404000B2/en active Active
- 2014-10-20 TR TR2019/03344T patent/TR201903344T4/tr unknown
- 2014-10-20 CA CA2957497A patent/CA2957497C/en active Active
- 2014-10-20 NZ NZ728907A patent/NZ728907A/en unknown
- 2014-10-20 EA EA201790396A patent/EA031319B1/ru unknown
- 2014-10-20 CN CN201480081234.1A patent/CN106794170A/zh active Pending
- 2014-10-20 AP AP2017009752A patent/AP2017009752A0/en unknown
- 2014-10-20 JP JP2017527993A patent/JP6629322B2/ja active Active
- 2014-10-20 BR BR112017002921-9A patent/BR112017002921B1/pt active IP Right Grant
- 2014-10-20 KR KR1020177004616A patent/KR101938408B1/ko active Active
- 2014-10-20 HR HRP20190446TT patent/HRP20190446T1/hr unknown
- 2014-10-20 SM SM20190147T patent/SMT201900147T1/it unknown
- 2014-10-20 WO PCT/IN2014/000667 patent/WO2016027276A1/en not_active Ceased
- 2014-10-20 US US15/504,014 patent/US10010527B2/en active Active
- 2014-10-20 PT PT14825443T patent/PT3180001T/pt unknown
- 2014-10-20 SI SI201431124T patent/SI3180001T1/sl unknown
- 2014-10-20 ES ES14825443T patent/ES2715113T3/es active Active
- 2014-10-20 SG SG11201701106RA patent/SG11201701106RA/en unknown
- 2014-10-20 DK DK14825443.6T patent/DK3180001T3/en active
- 2014-10-20 MX MX2017002045A patent/MX375538B/es active IP Right Grant
- 2014-10-20 EP EP14825443.6A patent/EP3180001B1/en active Active
- 2014-10-20 RS RS20190286A patent/RS58575B1/sr unknown
- 2014-10-20 HU HUE14825443A patent/HUE044011T2/hu unknown
- 2014-10-20 LT LTEP14825443.6T patent/LT3180001T/lt unknown
-
2017
- 2017-02-02 IL IL250411A patent/IL250411B/en active IP Right Grant
- 2017-02-06 ZA ZA2017/00915A patent/ZA201700915B/en unknown
-
2019
- 2019-03-08 CY CY20191100280T patent/CY1121393T1/el unknown
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR100823908B1 (ko) * | 2000-10-20 | 2008-04-21 | 바이오비트럼 에이비(피유비엘) | 2-, 3-, 4-, 또는 5-치환-n1-(벤젠술포닐)인돌 및 이의치료 용도 |
| KR20050086868A (ko) * | 2002-11-28 | 2005-08-30 | 수벤 라이프 사이언시스 리미티드 | 세로토닌 수용체 친화력을 가지는 엔-아릴술포닐-3-치환인돌, 그 제조방법 및 그를 포함하는 약제학적 조성물 |
| KR20050084185A (ko) * | 2002-12-18 | 2005-08-26 | 수벤 라이프 사이언시스 리미티드 | 세로토닌 수용체 친화력을 가지는 테트라사이클릭 3-치환인돌 |
| KR20100014413A (ko) * | 2007-02-16 | 2010-02-10 | 메모리 파마슈티칼스 코포레이션 | 5-ht6 수용체 친화력을 갖는 6' 치환된 인돌 및 인다졸 유도체 |
| KR20100069707A (ko) * | 2007-10-26 | 2010-06-24 | 수벤 라이프 사이언시스 리미티드 | 아미노 아릴설폰아마이드 화합물 및 5―ht6 리간드로서의 이들의 용도 |
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US7645752B2 (en) | Sulfonyl substituted 1H-indoles as ligands for the 5-hydroxytryptamine receptors | |
| JP2019518776A (ja) | Egfr阻害剤としてのアニリンピリミジン化合物の結晶 | |
| US9540321B2 (en) | Process for large scale production of 1-[(2-bromophenyl)sulfonyl]-5-methoxy-3-[(4-methyl-1-piperazinyl)methyl]-1H-indole dimesylate monohydrate | |
| WO2024112611A1 (en) | Targeted protein modification | |
| JP2009538910A (ja) | 5−ヒドロキシトリプタミン−6リガンドとしての1−スルホニルインダゾリルアミン誘導体および1−スルホニルインダゾリルアミド誘導体 | |
| JP2008542433A (ja) | CDK−1インヒビターとしてのα−カルボリン | |
| JP2001512727A (ja) | 5ht−1受容体のリガンドとしてのニ環式化合物 | |
| BG66457B1 (bg) | Индолови производни и тяхното използване като лекарствени средства | |
| KR20160071384A (ko) | 고혈압 및/또는 섬유증 치료용 조성물 | |
| KR101938408B1 (ko) | 1-[(2-브로모페닐) 술포닐]-5-메톡시-3-[(4-메틸-1-피페라지닐) 메틸]-1h-인돌 디메실레이트 모노히드레이트의 활성 대사산물 및 상기 활성 대사산물의 디메실레이트 디히드레이트 염 | |
| EA017007B1 (ru) | Соединения 5-(гетероциклил)алкил-n-(арилсульфонил)индола и их применение в качестве лигандов 5-ht | |
| KR20120112726A (ko) | 5?ht6 수용체 리간드인 술폰 화합물 | |
| TW201103905A (en) | 5-alkynyl-pyridines | |
| CN109476643B (zh) | 作为jak抑制剂的吡唑基氨基苯并咪唑衍生物 | |
| WO2019096112A1 (zh) | 一种取代的苯并咪唑化合物及包含该化合物的组合物 | |
| WO2007046112A1 (en) | Arylthioether tryptamine derivatives as functional 5-ht6 ligands | |
| US7501449B2 (en) | 2H -or 3H-benzo[e]indazol-1-yl carbamate derivatives, the preparation and therapeutic use thereof | |
| HK1236833A1 (en) | Active metabolite of 1-[(2-bromophenyl) sulfonyl]-5-methoxy-3- [(4-methyl-1-piperazinyl) methyl]-1h-indole dimesylate monohydrate and dimesylate dihydrate salt of active metabolite | |
| US5512566A (en) | Tricyclic compounds having affinity for the 5-HT1A receptor | |
| US20070099912A1 (en) | Pyrrolo[2,3-F] and [3,2-F]Isoquinolinone derivatives as 5-hydroxytryptamine-6 ligands | |
| US10449186B2 (en) | Phenylethynyl-substituted benzenes and heterocycles for the treatment of cancer | |
| CZ2000947A3 (cs) | Substituované chromanové deriváty |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PA0105 | International application |
Patent event date: 20170220 Patent event code: PA01051R01D Comment text: International Patent Application |
|
| A201 | Request for examination | ||
| PA0201 | Request for examination |
Patent event code: PA02012R01D Patent event date: 20170221 Comment text: Request for Examination of Application |
|
| PG1501 | Laying open of application | ||
| E902 | Notification of reason for refusal | ||
| PE0902 | Notice of grounds for rejection |
Comment text: Notification of reason for refusal Patent event date: 20180307 Patent event code: PE09021S01D |
|
| E701 | Decision to grant or registration of patent right | ||
| PE0701 | Decision of registration |
Patent event code: PE07011S01D Comment text: Decision to Grant Registration Patent event date: 20181022 |
|
| GRNT | Written decision to grant | ||
| PR0701 | Registration of establishment |
Comment text: Registration of Establishment Patent event date: 20190108 Patent event code: PR07011E01D |
|
| PR1002 | Payment of registration fee |
Payment date: 20190109 End annual number: 3 Start annual number: 1 |
|
| PG1601 | Publication of registration | ||
| PR1001 | Payment of annual fee |
Payment date: 20211230 Start annual number: 4 End annual number: 4 |
|
| PR1001 | Payment of annual fee |
Payment date: 20241226 Start annual number: 7 End annual number: 7 |