KR20120052943A - 제약 제제 - Google Patents
제약 제제 Download PDFInfo
- Publication number
- KR20120052943A KR20120052943A KR1020127002290A KR20127002290A KR20120052943A KR 20120052943 A KR20120052943 A KR 20120052943A KR 1020127002290 A KR1020127002290 A KR 1020127002290A KR 20127002290 A KR20127002290 A KR 20127002290A KR 20120052943 A KR20120052943 A KR 20120052943A
- Authority
- KR
- South Korea
- Prior art keywords
- formula
- surfactant
- ethanol
- compound
- mixture
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- 239000008194 pharmaceutical composition Substances 0.000 title claims abstract description 24
- 150000001875 compounds Chemical class 0.000 claims abstract description 49
- 239000002253 acid Substances 0.000 claims abstract description 13
- 150000003839 salts Chemical class 0.000 claims abstract description 12
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 claims description 88
- 239000004094 surface-active agent Substances 0.000 claims description 55
- 239000000203 mixture Substances 0.000 claims description 43
- 238000009472 formulation Methods 0.000 claims description 18
- ULQISTXYYBZJSJ-UHFFFAOYSA-N 12-hydroxyoctadecanoic acid Chemical compound CCCCCCC(O)CCCCCCCCCCC(O)=O ULQISTXYYBZJSJ-UHFFFAOYSA-N 0.000 claims description 16
- 150000005690 diesters Chemical class 0.000 claims description 14
- 229920001223 polyethylene glycol Polymers 0.000 claims description 13
- 239000002202 Polyethylene glycol Substances 0.000 claims description 10
- 238000010790 dilution Methods 0.000 claims description 9
- 239000012895 dilution Substances 0.000 claims description 9
- 239000000644 isotonic solution Substances 0.000 claims description 9
- 239000007788 liquid Substances 0.000 claims description 9
- 229940114072 12-hydroxystearic acid Drugs 0.000 claims description 8
- 238000002360 preparation method Methods 0.000 claims description 8
- 230000010412 perfusion Effects 0.000 claims description 7
- 238000007865 diluting Methods 0.000 claims description 6
- 238000000034 method Methods 0.000 claims description 6
- 239000003186 pharmaceutical solution Substances 0.000 claims description 6
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 claims description 5
- 238000001914 filtration Methods 0.000 claims description 5
- 229960003511 macrogol Drugs 0.000 claims description 5
- 230000001954 sterilising effect Effects 0.000 claims description 5
- SHBUUTHKGIVMJT-UHFFFAOYSA-N Hydroxystearate Chemical compound CCCCCCCCCCCCCCCCCC(=O)OO SHBUUTHKGIVMJT-UHFFFAOYSA-N 0.000 claims description 4
- 238000010438 heat treatment Methods 0.000 claims description 4
- 229940072106 hydroxystearate Drugs 0.000 claims description 4
- 238000001816 cooling Methods 0.000 claims description 3
- 150000007513 acids Chemical class 0.000 claims 1
- 238000004519 manufacturing process Methods 0.000 claims 1
- 239000004480 active ingredient Substances 0.000 abstract description 2
- 239000000243 solution Substances 0.000 description 13
- 101000656751 Haloarcula marismortui (strain ATCC 43049 / DSM 3752 / JCM 8966 / VKM B-1809) 30S ribosomal protein S24e Proteins 0.000 description 10
- 206010028980 Neoplasm Diseases 0.000 description 6
- 239000002246 antineoplastic agent Substances 0.000 description 5
- 239000012535 impurity Substances 0.000 description 5
- 241000699670 Mus sp. Species 0.000 description 4
- 239000012141 concentrate Substances 0.000 description 4
- 239000000126 substance Substances 0.000 description 4
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 3
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 3
- 201000011510 cancer Diseases 0.000 description 3
- 239000008103 glucose Substances 0.000 description 3
- 238000004659 sterilization and disinfection Methods 0.000 description 3
- IAYPIBMASNFSPL-UHFFFAOYSA-N Ethylene oxide Chemical compound C1CO1 IAYPIBMASNFSPL-UHFFFAOYSA-N 0.000 description 2
- 239000002033 PVDF binder Substances 0.000 description 2
- 229920002556 Polyethylene Glycol 300 Polymers 0.000 description 2
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 2
- 238000007046 ethoxylation reaction Methods 0.000 description 2
- 239000011521 glass Substances 0.000 description 2
- 208000032839 leukemia Diseases 0.000 description 2
- 239000000693 micelle Substances 0.000 description 2
- 239000003921 oil Substances 0.000 description 2
- 229920002981 polyvinylidene fluoride Polymers 0.000 description 2
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 2
- FBOUIAKEJMZPQG-AWNIVKPZSA-N (1E)-1-(2,4-dichlorophenyl)-4,4-dimethyl-2-(1,2,4-triazol-1-yl)pent-1-en-3-ol Chemical compound C1=NC=NN1/C(C(O)C(C)(C)C)=C/C1=CC=C(Cl)C=C1Cl FBOUIAKEJMZPQG-AWNIVKPZSA-N 0.000 description 1
- ZCYVEMRRCGMTRW-UHFFFAOYSA-N 7553-56-2 Chemical compound [I] ZCYVEMRRCGMTRW-UHFFFAOYSA-N 0.000 description 1
- 241001465754 Metazoa Species 0.000 description 1
- 229920002565 Polyethylene Glycol 400 Polymers 0.000 description 1
- 239000000654 additive Substances 0.000 description 1
- 230000000996 additive effect Effects 0.000 description 1
- 150000001298 alcohols Chemical class 0.000 description 1
- 239000003963 antioxidant agent Substances 0.000 description 1
- 230000003078 antioxidant effect Effects 0.000 description 1
- WQZGKKKJIJFFOK-VFUOTHLCSA-N beta-D-glucose Chemical compound OC[C@H]1O[C@@H](O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-VFUOTHLCSA-N 0.000 description 1
- 239000000872 buffer Substances 0.000 description 1
- 239000004359 castor oil Substances 0.000 description 1
- 235000019438 castor oil Nutrition 0.000 description 1
- 230000015556 catabolic process Effects 0.000 description 1
- 239000003054 catalyst Substances 0.000 description 1
- 239000006184 cosolvent Substances 0.000 description 1
- 238000006731 degradation reaction Methods 0.000 description 1
- 230000001419 dependent effect Effects 0.000 description 1
- 235000014113 dietary fatty acids Nutrition 0.000 description 1
- 238000009792 diffusion process Methods 0.000 description 1
- 239000012897 dilution medium Substances 0.000 description 1
- 239000002552 dosage form Substances 0.000 description 1
- 150000002148 esters Chemical class 0.000 description 1
- 239000003925 fat Substances 0.000 description 1
- 229930195729 fatty acid Natural products 0.000 description 1
- 239000000194 fatty acid Substances 0.000 description 1
- 150000004665 fatty acids Chemical class 0.000 description 1
- 239000012530 fluid Substances 0.000 description 1
- ZEMPKEQAKRGZGQ-XOQCFJPHSA-N glycerol triricinoleate Natural products CCCCCC[C@@H](O)CC=CCCCCCCCC(=O)OC[C@@H](COC(=O)CCCCCCCC=CC[C@@H](O)CCCCCC)OC(=O)CCCCCCCC=CC[C@H](O)CCCCCC ZEMPKEQAKRGZGQ-XOQCFJPHSA-N 0.000 description 1
- 238000004128 high performance liquid chromatography Methods 0.000 description 1
- 239000012456 homogeneous solution Substances 0.000 description 1
- 238000005984 hydrogenation reaction Methods 0.000 description 1
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 1
- 238000007912 intraperitoneal administration Methods 0.000 description 1
- 238000001990 intravenous administration Methods 0.000 description 1
- 229910052740 iodine Inorganic materials 0.000 description 1
- 239000011630 iodine Substances 0.000 description 1
- 239000002609 medium Substances 0.000 description 1
- 238000002844 melting Methods 0.000 description 1
- 230000008018 melting Effects 0.000 description 1
- 239000000825 pharmaceutical preparation Substances 0.000 description 1
- 229920000136 polysorbate Polymers 0.000 description 1
- 238000001556 precipitation Methods 0.000 description 1
- 239000003755 preservative agent Substances 0.000 description 1
- 230000002335 preservative effect Effects 0.000 description 1
- 238000011028 process validation Methods 0.000 description 1
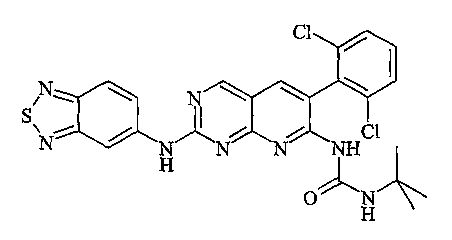
- 159000000018 pyrido[2,3-d]pyrimidines Chemical class 0.000 description 1
- 239000011780 sodium chloride Substances 0.000 description 1
- 239000007787 solid Substances 0.000 description 1
- 239000000271 synthetic detergent Substances 0.000 description 1
- 238000010200 validation analysis Methods 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/505—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim
- A61K31/519—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim ortho- or peri-condensed with heterocyclic rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K45/00—Medicinal preparations containing active ingredients not provided for in groups A61K31/00 - A61K41/00
- A61K45/06—Mixtures of active ingredients without chemical characterisation, e.g. antiphlogistics and cardiaca
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/08—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite containing oxygen, e.g. ethers, acetals, ketones, quinones, aldehydes, peroxides
- A61K47/14—Esters of carboxylic acids, e.g. fatty acid monoglycerides, medium-chain triglycerides, parabens or PEG fatty acid esters
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/0019—Injectable compositions; Intramuscular, intravenous, arterial, subcutaneous administration; Compositions to be administered through the skin in an invasive manner
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
- A61P35/02—Antineoplastic agents specific for leukemia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/08—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite containing oxygen, e.g. ethers, acetals, ketones, quinones, aldehydes, peroxides
- A61K47/10—Alcohols; Phenols; Salts thereof, e.g. glycerol; Polyethylene glycols [PEG]; Poloxamers; PEG/POE alkyl ethers
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Veterinary Medicine (AREA)
- Animal Behavior & Ethology (AREA)
- Life Sciences & Earth Sciences (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Public Health (AREA)
- Epidemiology (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Dermatology (AREA)
- Engineering & Computer Science (AREA)
- Organic Chemistry (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Hematology (AREA)
- Oncology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Medicinal Preparation (AREA)
- Nitrogen Condensed Heterocyclic Rings (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
- Medical Preparation Storing Or Oral Administration Devices (AREA)
- Nitrogen And Oxygen Or Sulfur-Condensed Heterocyclic Ring Systems (AREA)
Abstract
Description
Claims (17)
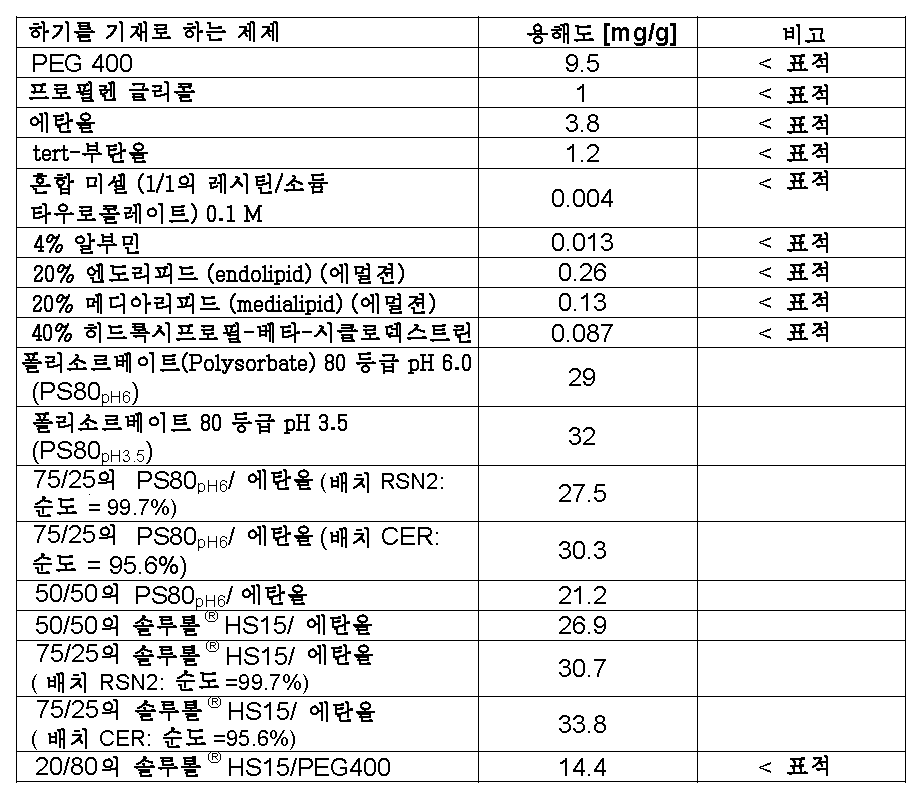
- 제1항 또는 제2항에 있어서, 계면활성제/에탄올의 중량비가 73/27 내지 77/23의 범위인 것을 특징으로 하는 제약 제제.
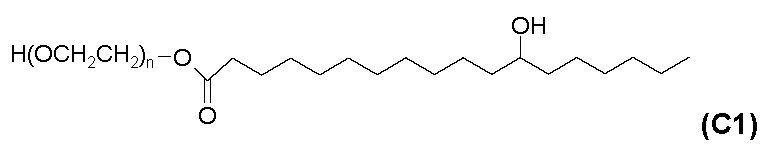
- 제2항에 있어서, 계면활성제가 35 중량% 내지 55 중량%의 모노에스테르 및 디에스테르와 30 중량% 내지 40 중량%의 폴리에틸렌 글리콜 H(OCH2CH2)n-OH를 포함하는 것인 제제.
- 제4항에 있어서, 계면활성제가 주 성분으로서 35 중량% 내지 55 중량%의 모노에스테르 및 디에스테르와 30 중량% 내지 40 중량%의 폴리에틸렌 글리콜 H(OCH2CH2)n-OH, 및 또한 100%가 되도록 나머지를 구성하는 기타 화합물을 포함하는 것인 제제.
- 제1항 또는 제2항에 있어서, 계면활성제가 10 중량% 내지 20 중량%의 모노에스테르, 25 중량% 내지 35 중량%의 디에스테르 및 30 중량% 내지 40 중량%의 폴리에틸렌 글리콜 H(OCH2CH2)n-OH, 및 또한 100%가 되도록 나머지를 구성하는 기타 화합물을 포함하는 것인 제제.
- 제1항 내지 제6항 중 어느 한 항에 있어서, 계면활성제/에탄올의 비가 73/27 내지 77/23의 범위이며 화학식 I의 화합물의 농도가 5 내지 25 mg/ml의 범위인 것인 제약 제제.
- 제1항 내지 제7항 중 어느 한 항에 있어서, 관류액을 형성하도록 희석시키기 위한 것인 제약 제제.
- - 계면활성제를 액체가 될 때까지 가열하는 단계;
- 에탄올을 첨가하는 단계;
- 계면활성제/에탄올 혼합물을 주위 온도로 냉각시키는 단계;
- 화학식 I의 화합물을 냉각된 혼합물에 첨가하는 단계;
- 최종 혼합물을 살균하는 단계
를 포함하는, 제1항 내지 제8항 중 어느 한 항에 따른 제약 제제를 제조하는 방법. - 제9항에 있어서, 혼합물을 여과에 의해 살균하는 방법.
- 제11항 또는 제12항에 있어서, 인간에게 투여하기 위한 관류액.
- 제1항 내지 제8항 중 어느 한 항에 따른 1배 부피의 제약 용액을 20 내지 500배 부피의 등장액 중에 희석시키는 단계로 구성된, 관류액을 제조하는 방법.
- 제1항 내지 제8항 중 어느 한 항에 개시된 제약 용액을 함유하는 병.
- 제11항 내지 제13항 중 어느 한 항의 관류액을 함유하는 점적 백.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| FR09/03742 | 2009-07-30 | ||
| FR0903742A FR2948568B1 (fr) | 2009-07-30 | 2009-07-30 | Formulation pharmaceutique |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| KR20120052943A true KR20120052943A (ko) | 2012-05-24 |
Family
ID=41570922
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020127002290A Ceased KR20120052943A (ko) | 2009-07-30 | 2010-07-29 | 제약 제제 |
Country Status (39)
| Country | Link |
|---|---|
| US (1) | US20120202831A1 (ko) |
| EP (1) | EP2459221B1 (ko) |
| JP (1) | JP5658754B2 (ko) |
| KR (1) | KR20120052943A (ko) |
| CN (1) | CN102470176B (ko) |
| AR (1) | AR077338A1 (ko) |
| AU (1) | AU2010277406B2 (ko) |
| BR (1) | BR112012002105A2 (ko) |
| CA (1) | CA2769477A1 (ko) |
| CL (1) | CL2012000231A1 (ko) |
| CO (1) | CO6491065A2 (ko) |
| CR (1) | CR20120047A (ko) |
| CY (1) | CY1115043T1 (ko) |
| DK (1) | DK2459221T3 (ko) |
| DO (1) | DOP2012000011A (ko) |
| EA (1) | EA021059B1 (ko) |
| ES (1) | ES2458419T3 (ko) |
| FR (1) | FR2948568B1 (ko) |
| HN (1) | HN2012000182A (ko) |
| HR (1) | HRP20140355T1 (ko) |
| IL (1) | IL217762A0 (ko) |
| MA (1) | MA33462B1 (ko) |
| MX (1) | MX2012001386A (ko) |
| MY (1) | MY183312A (ko) |
| NI (1) | NI201200017A (ko) |
| NZ (1) | NZ597963A (ko) |
| PE (1) | PE20120619A1 (ko) |
| PL (1) | PL2459221T3 (ko) |
| PT (1) | PT2459221E (ko) |
| RS (1) | RS53266B (ko) |
| SG (1) | SG177754A1 (ko) |
| SI (1) | SI2459221T1 (ko) |
| SM (1) | SMT201400069B (ko) |
| TN (1) | TN2011000659A1 (ko) |
| TW (1) | TWI478921B (ko) |
| UA (1) | UA105229C2 (ko) |
| UY (1) | UY32816A (ko) |
| WO (1) | WO2011012816A2 (ko) |
| ZA (1) | ZA201200719B (ko) |
Families Citing this family (23)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2012088266A2 (en) | 2010-12-22 | 2012-06-28 | Incyte Corporation | Substituted imidazopyridazines and benzimidazoles as inhibitors of fgfr3 |
| US9611267B2 (en) | 2012-06-13 | 2017-04-04 | Incyte Holdings Corporation | Substituted tricyclic compounds as FGFR inhibitors |
| US9388185B2 (en) | 2012-08-10 | 2016-07-12 | Incyte Holdings Corporation | Substituted pyrrolo[2,3-b]pyrazines as FGFR inhibitors |
| US9266892B2 (en) | 2012-12-19 | 2016-02-23 | Incyte Holdings Corporation | Fused pyrazoles as FGFR inhibitors |
| TWI649318B (zh) | 2013-04-19 | 2019-02-01 | 英塞特控股公司 | 作為fgfr抑制劑之雙環雜環 |
| US10851105B2 (en) | 2014-10-22 | 2020-12-01 | Incyte Corporation | Bicyclic heterocycles as FGFR4 inhibitors |
| MA41551A (fr) | 2015-02-20 | 2017-12-26 | Incyte Corp | Hétérocycles bicycliques utilisés en tant qu'inhibiteurs de fgfr4 |
| WO2016134294A1 (en) | 2015-02-20 | 2016-08-25 | Incyte Corporation | Bicyclic heterocycles as fgfr4 inhibitors |
| AU2016219822B2 (en) | 2015-02-20 | 2020-07-09 | Incyte Holdings Corporation | Bicyclic heterocycles as FGFR inhibitors |
| AR111960A1 (es) | 2017-05-26 | 2019-09-04 | Incyte Corp | Formas cristalinas de un inhibidor de fgfr y procesos para su preparación |
| ES2991427T3 (es) | 2018-05-04 | 2024-12-03 | Incyte Corp | Formas sólidas de un inhibidor de FGFR y procedimientos para preparar las mismas |
| TW201946630A (zh) | 2018-05-04 | 2019-12-16 | 美商英塞特公司 | Fgfr抑制劑之鹽 |
| US11628162B2 (en) | 2019-03-08 | 2023-04-18 | Incyte Corporation | Methods of treating cancer with an FGFR inhibitor |
| WO2021007269A1 (en) | 2019-07-09 | 2021-01-14 | Incyte Corporation | Bicyclic heterocycles as fgfr inhibitors |
| WO2021067374A1 (en) | 2019-10-01 | 2021-04-08 | Incyte Corporation | Bicyclic heterocycles as fgfr inhibitors |
| GEAP202415945A (en) | 2019-10-14 | 2024-04-25 | Incyte Corp | Bicyclic heterocycles as fgfr inhibitors |
| WO2021076728A1 (en) | 2019-10-16 | 2021-04-22 | Incyte Corporation | Bicyclic heterocycles as fgfr inhibitors |
| CA3163875A1 (en) | 2019-12-04 | 2021-06-10 | Incyte Corporation | Tricyclic heterocycles as fgfr inhibitors |
| AU2020395185A1 (en) | 2019-12-04 | 2022-06-02 | Incyte Corporation | Derivatives of an FGFR inhibitor |
| WO2021146424A1 (en) | 2020-01-15 | 2021-07-22 | Incyte Corporation | Bicyclic heterocycles as fgfr inhibitors |
| EP4323405A1 (en) | 2021-04-12 | 2024-02-21 | Incyte Corporation | Combination therapy comprising an fgfr inhibitor and a nectin-4 targeting agent |
| AR126101A1 (es) | 2021-06-09 | 2023-09-13 | Incyte Corp | Heterociclos tricíclicos como inhibidores de fgfr |
| CA3220274A1 (en) | 2021-06-09 | 2022-12-15 | Incyte Corporation | Tricyclic heterocycles as fgfr inhibitors |
Family Cites Families (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE2911241A1 (de) | 1979-03-22 | 1980-10-02 | Basf Ag | Alkoxylierte fettsaeuren, verfahren zu deren herstellung und ihre anwendung als loesungsvermittler |
| HU201567B (en) * | 1988-07-21 | 1990-11-28 | Gyogyszerkutato Intezet | Process for production of intravenous medical compositions containing cyclosphorin |
| WO1996011007A1 (en) * | 1994-10-05 | 1996-04-18 | Glaxo Wellcome Inc. | Parenteral pharmaceutical compositions containing gf120918a |
| US5922754A (en) * | 1998-10-02 | 1999-07-13 | Abbott Laboratories | Pharmaceutical compositions containing paclitaxel |
| US8618085B2 (en) * | 2000-04-28 | 2013-12-31 | Koasn Biosciences Incorporated | Therapeutic formulations of desoxyepothilones |
| KR100866728B1 (ko) * | 2004-11-12 | 2008-11-03 | 주식회사종근당 | 타크로리무스를 함유하는 주사제 |
| FR2879932B1 (fr) * | 2004-12-27 | 2007-03-23 | Aventis Pharma Sa | Formulations injectable ou administrable par voie orale de derives d'azetidine |
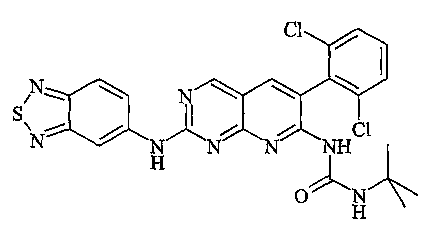
| FR2887882B1 (fr) * | 2005-07-01 | 2007-09-07 | Sanofi Aventis Sa | Derives de pyrido[2,3-d] pyrimidine, leur preparation, leur application en therapeutique |
| FR2910813B1 (fr) * | 2006-12-28 | 2009-02-06 | Sanofi Aventis Sa | Nouvelle utilisation therapeutique pour le traitement des leucemies |
| DE102007021862A1 (de) * | 2007-05-10 | 2008-11-13 | Merck Patent Gmbh | Wässrige pharmazeutische Zubereitung |
-
2009
- 2009-07-30 FR FR0903742A patent/FR2948568B1/fr not_active Expired - Fee Related
-
2010
- 2010-07-29 PE PE2012000117A patent/PE20120619A1/es not_active Application Discontinuation
- 2010-07-29 UA UAA201202339A patent/UA105229C2/uk unknown
- 2010-07-29 WO PCT/FR2010/051611 patent/WO2011012816A2/fr not_active Ceased
- 2010-07-29 MX MX2012001386A patent/MX2012001386A/es active IP Right Grant
- 2010-07-29 SI SI201030587T patent/SI2459221T1/sl unknown
- 2010-07-29 KR KR1020127002290A patent/KR20120052943A/ko not_active Ceased
- 2010-07-29 AR ARP100102748A patent/AR077338A1/es unknown
- 2010-07-29 AU AU2010277406A patent/AU2010277406B2/en not_active Ceased
- 2010-07-29 EA EA201270216A patent/EA021059B1/ru not_active IP Right Cessation
- 2010-07-29 MA MA34577A patent/MA33462B1/fr unknown
- 2010-07-29 NZ NZ597963A patent/NZ597963A/en not_active IP Right Cessation
- 2010-07-29 BR BR112012002105A patent/BR112012002105A2/pt not_active IP Right Cessation
- 2010-07-29 DK DK10762950.3T patent/DK2459221T3/da active
- 2010-07-29 HR HRP20140355AT patent/HRP20140355T1/hr unknown
- 2010-07-29 PT PT107629503T patent/PT2459221E/pt unknown
- 2010-07-29 MY MYPI2012000425A patent/MY183312A/en unknown
- 2010-07-29 JP JP2012522231A patent/JP5658754B2/ja not_active Expired - Fee Related
- 2010-07-29 CA CA2769477A patent/CA2769477A1/fr not_active Abandoned
- 2010-07-29 CN CN201080033686.4A patent/CN102470176B/zh not_active Expired - Fee Related
- 2010-07-29 SG SG2012005435A patent/SG177754A1/en unknown
- 2010-07-29 EP EP10762950.3A patent/EP2459221B1/fr active Active
- 2010-07-29 RS RS20140195A patent/RS53266B/sr unknown
- 2010-07-29 PL PL10762950T patent/PL2459221T3/pl unknown
- 2010-07-29 ES ES10762950.3T patent/ES2458419T3/es active Active
- 2010-07-30 TW TW099125517A patent/TWI478921B/zh not_active IP Right Cessation
- 2010-07-30 UY UY0001032816A patent/UY32816A/es not_active Application Discontinuation
-
2011
- 2011-12-22 TN TNP2011000659A patent/TN2011000659A1/fr unknown
-
2012
- 2012-01-19 DO DO2012000011A patent/DOP2012000011A/es unknown
- 2012-01-20 CO CO12008121A patent/CO6491065A2/es not_active Application Discontinuation
- 2012-01-24 CR CR20120047A patent/CR20120047A/es unknown
- 2012-01-26 IL IL217762A patent/IL217762A0/en unknown
- 2012-01-26 CL CL2012000231A patent/CL2012000231A1/es unknown
- 2012-01-27 HN HN2012000182A patent/HN2012000182A/es unknown
- 2012-01-27 NI NI201200017A patent/NI201200017A/es unknown
- 2012-01-30 US US13/361,309 patent/US20120202831A1/en not_active Abandoned
- 2012-01-30 ZA ZA2012/00719A patent/ZA201200719B/en unknown
-
2014
- 2014-04-22 CY CY20141100298T patent/CY1115043T1/el unknown
- 2014-06-12 SM SM201400069T patent/SMT201400069B/xx unknown
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5658754B2 (ja) | 医薬製剤 | |
| JP4009877B2 (ja) | 親脂性に富んだカンプトテシン誘導体治療薬 | |
| US20070249632A1 (en) | Pharmaceutical compositions and use thereof | |
| JPH07505628A (ja) | 静脈注射用のラパマイシン製剤 | |
| JP6356873B2 (ja) | タキサン系活性成分含有液体組成物及び液体製剤 | |
| JP6736765B2 (ja) | ベンダムスチン溶液製剤 | |
| KR20180030479A (ko) | 레파뮬린의 주사가능한 약제학적 제형 | |
| US20090069350A1 (en) | Pharmaceutical compositions and use thereof | |
| US20110130446A1 (en) | Injectable taxane pharmaceutical composition | |
| CN115666579A (zh) | 卡非佐米的稳定的即稀释型制剂 | |
| JP2822049B2 (ja) | 水性製剤組成物 | |
| HK1169960B (en) | Pharmaceutical formulation | |
| JP2011231109A (ja) | 組成物 | |
| US20180028470A1 (en) | Curcumin-based pharmaceutical compositions and methods for fabricating thereof | |
| KR20220124891A (ko) | 나파모스타트 또는 이의 염을 포함하는 에멀젼을 포함하는 약제학적 제제 및 이의 제조 방법 | |
| JP2025537030A (ja) | 高濃度のオピランセリンを含む医薬組成物 | |
| HK1142281A (en) | Aqueous pharmaceutical preparation |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PA0105 | International application |
Patent event date: 20120127 Patent event code: PA01051R01D Comment text: International Patent Application |
|
| PG1501 | Laying open of application | ||
| A201 | Request for examination | ||
| PA0201 | Request for examination |
Patent event code: PA02012R01D Patent event date: 20150616 Comment text: Request for Examination of Application |
|
| E902 | Notification of reason for refusal | ||
| PE0902 | Notice of grounds for rejection |
Comment text: Notification of reason for refusal Patent event date: 20160609 Patent event code: PE09021S01D |
|
| E601 | Decision to refuse application | ||
| PE0601 | Decision on rejection of patent |
Patent event date: 20160922 Comment text: Decision to Refuse Application Patent event code: PE06012S01D Patent event date: 20160609 Comment text: Notification of reason for refusal Patent event code: PE06011S01I |