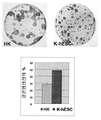
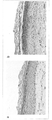
KR20110022713A - 인간 만능줄기세포로부터 인간 피부 대체물을 제조하는 방법 - Google Patents
인간 만능줄기세포로부터 인간 피부 대체물을 제조하는 방법 Download PDFInfo
- Publication number
- KR20110022713A KR20110022713A KR1020117001947A KR20117001947A KR20110022713A KR 20110022713 A KR20110022713 A KR 20110022713A KR 1020117001947 A KR1020117001947 A KR 1020117001947A KR 20117001947 A KR20117001947 A KR 20117001947A KR 20110022713 A KR20110022713 A KR 20110022713A
- Authority
- KR
- South Korea
- Prior art keywords
- human
- stem cells
- cells
- pluripotent stem
- keratinocytes
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Withdrawn
Links
- 238000000034 method Methods 0.000 title claims abstract description 63
- 210000001778 pluripotent stem cell Anatomy 0.000 title claims abstract description 51
- 210000004027 cell Anatomy 0.000 claims abstract description 158
- 210000002510 keratinocyte Anatomy 0.000 claims abstract description 128
- 210000003491 skin Anatomy 0.000 claims abstract description 79
- 230000004069 differentiation Effects 0.000 claims abstract description 37
- 238000000338 in vitro Methods 0.000 claims abstract description 28
- 230000006698 induction Effects 0.000 claims abstract description 18
- 238000012258 culturing Methods 0.000 claims abstract description 14
- 210000003981 ectoderm Anatomy 0.000 claims abstract description 13
- 230000011712 cell development Effects 0.000 claims abstract description 8
- 230000008093 supporting effect Effects 0.000 claims abstract description 7
- 238000004519 manufacturing process Methods 0.000 claims abstract description 4
- CIWBSHSKHKDKBQ-JLAZNSOCSA-N Ascorbic acid Chemical compound OC[C@H](O)[C@H]1OC(=O)C(O)=C1O CIWBSHSKHKDKBQ-JLAZNSOCSA-N 0.000 claims description 41
- 210000002950 fibroblast Anatomy 0.000 claims description 36
- 210000004263 induced pluripotent stem cell Anatomy 0.000 claims description 31
- 102100024505 Bone morphogenetic protein 4 Human genes 0.000 claims description 22
- 150000001875 compounds Chemical class 0.000 claims description 22
- 238000011282 treatment Methods 0.000 claims description 21
- 235000010323 ascorbic acid Nutrition 0.000 claims description 20
- 239000011668 ascorbic acid Substances 0.000 claims description 20
- 229960005070 ascorbic acid Drugs 0.000 claims description 20
- 239000003795 chemical substances by application Substances 0.000 claims description 20
- 230000002500 effect on skin Effects 0.000 claims description 20
- 238000004113 cell culture Methods 0.000 claims description 18
- 230000007170 pathology Effects 0.000 claims description 18
- 239000001963 growth medium Substances 0.000 claims description 17
- 108010049955 Bone Morphogenetic Protein 4 Proteins 0.000 claims description 16
- 230000037380 skin damage Effects 0.000 claims description 13
- 239000008194 pharmaceutical composition Substances 0.000 claims description 10
- 239000000758 substrate Substances 0.000 claims description 10
- 210000001671 embryonic stem cell Anatomy 0.000 claims description 9
- 239000003937 drug carrier Substances 0.000 claims description 5
- 230000001575 pathological effect Effects 0.000 claims description 5
- 239000000546 pharmaceutical excipient Substances 0.000 claims description 5
- 238000012216 screening Methods 0.000 claims description 5
- 206010028980 Neoplasm Diseases 0.000 claims description 4
- 230000002950 deficient Effects 0.000 claims description 2
- 239000010410 layer Substances 0.000 description 36
- 102100040445 Keratin, type I cytoskeletal 14 Human genes 0.000 description 27
- 230000014509 gene expression Effects 0.000 description 26
- 238000010171 animal model Methods 0.000 description 24
- 210000002615 epidermis Anatomy 0.000 description 24
- 239000002609 medium Substances 0.000 description 23
- 102100025756 Keratin, type II cytoskeletal 5 Human genes 0.000 description 21
- 108090000623 proteins and genes Proteins 0.000 description 20
- 102100033421 Keratin, type I cytoskeletal 18 Human genes 0.000 description 19
- NWIBSHFKIJFRCO-WUDYKRTCSA-N Mytomycin Chemical compound C1N2C(C(C(C)=C(N)C3=O)=O)=C3[C@@H](COC(N)=O)[C@@]2(OC)[C@@H]2[C@H]1N2 NWIBSHFKIJFRCO-WUDYKRTCSA-N 0.000 description 19
- 208000027418 Wounds and injury Diseases 0.000 description 15
- 238000001727 in vivo Methods 0.000 description 15
- 108010070553 Keratin-5 Proteins 0.000 description 14
- 238000004458 analytical method Methods 0.000 description 14
- 238000012360 testing method Methods 0.000 description 14
- 108010066321 Keratin-14 Proteins 0.000 description 13
- 238000003753 real-time PCR Methods 0.000 description 12
- 239000003550 marker Substances 0.000 description 11
- 229960004857 mitomycin Drugs 0.000 description 11
- 241001465754 Metazoa Species 0.000 description 10
- 230000000877 morphologic effect Effects 0.000 description 10
- 102100023972 Keratin, type II cytoskeletal 8 Human genes 0.000 description 9
- 241000699666 Mus <mouse, genus> Species 0.000 description 9
- 206010052428 Wound Diseases 0.000 description 9
- 239000007788 liquid Substances 0.000 description 9
- 210000001519 tissue Anatomy 0.000 description 9
- 238000012413 Fluorescence activated cell sorting analysis Methods 0.000 description 8
- 101710183391 Keratin, type I cytoskeletal 14 Proteins 0.000 description 8
- JYGXADMDTFJGBT-VWUMJDOOSA-N hydrocortisone Chemical compound O=C1CC[C@]2(C)[C@H]3[C@@H](O)C[C@](C)([C@@](CC4)(O)C(=O)CO)[C@@H]4[C@@H]3CCC2=C1 JYGXADMDTFJGBT-VWUMJDOOSA-N 0.000 description 8
- NOESYZHRGYRDHS-UHFFFAOYSA-N insulin Chemical compound N1C(=O)C(NC(=O)C(CCC(N)=O)NC(=O)C(CCC(O)=O)NC(=O)C(C(C)C)NC(=O)C(NC(=O)CN)C(C)CC)CSSCC(C(NC(CO)C(=O)NC(CC(C)C)C(=O)NC(CC=2C=CC(O)=CC=2)C(=O)NC(CCC(N)=O)C(=O)NC(CC(C)C)C(=O)NC(CCC(O)=O)C(=O)NC(CC(N)=O)C(=O)NC(CC=2C=CC(O)=CC=2)C(=O)NC(CSSCC(NC(=O)C(C(C)C)NC(=O)C(CC(C)C)NC(=O)C(CC=2C=CC(O)=CC=2)NC(=O)C(CC(C)C)NC(=O)C(C)NC(=O)C(CCC(O)=O)NC(=O)C(C(C)C)NC(=O)C(CC(C)C)NC(=O)C(CC=2NC=NC=2)NC(=O)C(CO)NC(=O)CNC2=O)C(=O)NCC(=O)NC(CCC(O)=O)C(=O)NC(CCCNC(N)=N)C(=O)NCC(=O)NC(CC=3C=CC=CC=3)C(=O)NC(CC=3C=CC=CC=3)C(=O)NC(CC=3C=CC(O)=CC=3)C(=O)NC(C(C)O)C(=O)N3C(CCC3)C(=O)NC(CCCCN)C(=O)NC(C)C(O)=O)C(=O)NC(CC(N)=O)C(O)=O)=O)NC(=O)C(C(C)CC)NC(=O)C(CO)NC(=O)C(C(C)O)NC(=O)C1CSSCC2NC(=O)C(CC(C)C)NC(=O)C(NC(=O)C(CCC(N)=O)NC(=O)C(CC(N)=O)NC(=O)C(NC(=O)C(N)CC=1C=CC=CC=1)C(C)C)CC1=CN=CN1 NOESYZHRGYRDHS-UHFFFAOYSA-N 0.000 description 8
- 238000010186 staining Methods 0.000 description 8
- 210000000130 stem cell Anatomy 0.000 description 8
- 101001056473 Homo sapiens Keratin, type II cytoskeletal 5 Proteins 0.000 description 7
- 102000000426 Integrin alpha6 Human genes 0.000 description 7
- 108010041100 Integrin alpha6 Proteins 0.000 description 7
- 230000006378 damage Effects 0.000 description 7
- 108010028309 kalinin Proteins 0.000 description 7
- 102000004169 proteins and genes Human genes 0.000 description 7
- 102000008186 Collagen Human genes 0.000 description 6
- 108010035532 Collagen Proteins 0.000 description 6
- 101000762379 Homo sapiens Bone morphogenetic protein 4 Proteins 0.000 description 6
- 101000614436 Homo sapiens Keratin, type I cytoskeletal 14 Proteins 0.000 description 6
- 102000012334 Integrin beta4 Human genes 0.000 description 6
- 108010022238 Integrin beta4 Proteins 0.000 description 6
- 102100023970 Keratin, type I cytoskeletal 10 Human genes 0.000 description 6
- 108010066327 Keratin-18 Proteins 0.000 description 6
- 241000699670 Mus sp. Species 0.000 description 6
- 238000012512 characterization method Methods 0.000 description 6
- 229920001436 collagen Polymers 0.000 description 6
- 210000000981 epithelium Anatomy 0.000 description 6
- 230000012010 growth Effects 0.000 description 6
- 230000001965 increasing effect Effects 0.000 description 6
- 208000014674 injury Diseases 0.000 description 6
- 101000998020 Homo sapiens Keratin, type I cytoskeletal 18 Proteins 0.000 description 5
- 101000975496 Homo sapiens Keratin, type II cytoskeletal 8 Proteins 0.000 description 5
- 239000000427 antigen Substances 0.000 description 5
- 108091007433 antigens Proteins 0.000 description 5
- 102000036639 antigens Human genes 0.000 description 5
- 239000003814 drug Substances 0.000 description 5
- 239000007943 implant Substances 0.000 description 5
- 230000004807 localization Effects 0.000 description 5
- 210000000056 organ Anatomy 0.000 description 5
- 210000002220 organoid Anatomy 0.000 description 5
- 239000012679 serum free medium Substances 0.000 description 5
- 210000001082 somatic cell Anatomy 0.000 description 5
- 210000000434 stratum corneum Anatomy 0.000 description 5
- GFFGJBXGBJISGV-UHFFFAOYSA-N Adenine Chemical compound NC1=NC=NC2=C1N=CN2 GFFGJBXGBJISGV-UHFFFAOYSA-N 0.000 description 4
- 229930024421 Adenine Natural products 0.000 description 4
- 108091003079 Bovine Serum Albumin Proteins 0.000 description 4
- 102000009016 Cholera Toxin Human genes 0.000 description 4
- 108010049048 Cholera Toxin Proteins 0.000 description 4
- 108010037362 Extracellular Matrix Proteins Proteins 0.000 description 4
- 102000010834 Extracellular Matrix Proteins Human genes 0.000 description 4
- 102000009123 Fibrin Human genes 0.000 description 4
- 108010073385 Fibrin Proteins 0.000 description 4
- BWGVNKXGVNDBDI-UHFFFAOYSA-N Fibrin monomer Chemical compound CNC(=O)CNC(=O)CN BWGVNKXGVNDBDI-UHFFFAOYSA-N 0.000 description 4
- 208000026350 Inborn Genetic disease Diseases 0.000 description 4
- 102000004877 Insulin Human genes 0.000 description 4
- 108090001061 Insulin Proteins 0.000 description 4
- 108010070511 Keratin-8 Proteins 0.000 description 4
- 229960000643 adenine Drugs 0.000 description 4
- 230000000975 bioactive effect Effects 0.000 description 4
- 230000003247 decreasing effect Effects 0.000 description 4
- 210000004207 dermis Anatomy 0.000 description 4
- 201000010099 disease Diseases 0.000 description 4
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 4
- 229940079593 drug Drugs 0.000 description 4
- 230000000694 effects Effects 0.000 description 4
- 239000012091 fetal bovine serum Substances 0.000 description 4
- 229950003499 fibrin Drugs 0.000 description 4
- 238000001943 fluorescence-activated cell sorting Methods 0.000 description 4
- 208000016361 genetic disease Diseases 0.000 description 4
- 239000008187 granular material Substances 0.000 description 4
- 229960000890 hydrocortisone Drugs 0.000 description 4
- 229940125396 insulin Drugs 0.000 description 4
- 230000007774 longterm Effects 0.000 description 4
- 230000002062 proliferating effect Effects 0.000 description 4
- 238000011160 research Methods 0.000 description 4
- 230000001225 therapeutic effect Effects 0.000 description 4
- 238000002054 transplantation Methods 0.000 description 4
- 108091032973 (ribonucleotides)n+m Proteins 0.000 description 3
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 3
- 101710088660 Filaggrin Proteins 0.000 description 3
- 102100028314 Filaggrin Human genes 0.000 description 3
- 102000006354 HLA-DR Antigens Human genes 0.000 description 3
- 108010058597 HLA-DR Antigens Proteins 0.000 description 3
- 102100033420 Keratin, type I cytoskeletal 19 Human genes 0.000 description 3
- 102100022905 Keratin, type II cytoskeletal 1 Human genes 0.000 description 3
- 108010065038 Keratin-10 Proteins 0.000 description 3
- 229930192392 Mitomycin Natural products 0.000 description 3
- 102100027881 Tumor protein 63 Human genes 0.000 description 3
- 230000000903 blocking effect Effects 0.000 description 3
- 239000002537 cosmetic Substances 0.000 description 3
- 238000011161 development Methods 0.000 description 3
- 230000018109 developmental process Effects 0.000 description 3
- 210000002744 extracellular matrix Anatomy 0.000 description 3
- 210000003780 hair follicle Anatomy 0.000 description 3
- 230000001506 immunosuppresive effect Effects 0.000 description 3
- 238000007918 intramuscular administration Methods 0.000 description 3
- 238000001990 intravenous administration Methods 0.000 description 3
- 239000000463 material Substances 0.000 description 3
- 239000000203 mixture Substances 0.000 description 3
- 244000052769 pathogen Species 0.000 description 3
- 230000008569 process Effects 0.000 description 3
- 230000035755 proliferation Effects 0.000 description 3
- 230000008929 regeneration Effects 0.000 description 3
- 238000011069 regeneration method Methods 0.000 description 3
- 210000002966 serum Anatomy 0.000 description 3
- 239000002356 single layer Substances 0.000 description 3
- 239000000243 solution Substances 0.000 description 3
- 238000007920 subcutaneous administration Methods 0.000 description 3
- 108010049931 Bone Morphogenetic Protein 2 Proteins 0.000 description 2
- 108010049870 Bone Morphogenetic Protein 7 Proteins 0.000 description 2
- 102100024506 Bone morphogenetic protein 2 Human genes 0.000 description 2
- 102100022544 Bone morphogenetic protein 7 Human genes 0.000 description 2
- 101710087047 Cytoskeleton-associated protein 4 Proteins 0.000 description 2
- 206010013786 Dry skin Diseases 0.000 description 2
- 206010058314 Dysplasia Diseases 0.000 description 2
- WSFSSNUMVMOOMR-UHFFFAOYSA-N Formaldehyde Chemical compound O=C WSFSSNUMVMOOMR-UHFFFAOYSA-N 0.000 description 2
- 108010010803 Gelatin Proteins 0.000 description 2
- WZUVPPKBWHMQCE-UHFFFAOYSA-N Haematoxylin Chemical compound C12=CC(O)=C(O)C=C2CC2(O)C1C1=CC=C(O)C(O)=C1OC2 WZUVPPKBWHMQCE-UHFFFAOYSA-N 0.000 description 2
- 101000994365 Homo sapiens Integrin alpha-6 Proteins 0.000 description 2
- 101001015006 Homo sapiens Integrin beta-4 Proteins 0.000 description 2
- 101000851176 Homo sapiens Pro-epidermal growth factor Proteins 0.000 description 2
- 102100032816 Integrin alpha-6 Human genes 0.000 description 2
- 102100033000 Integrin beta-4 Human genes 0.000 description 2
- 102100023967 Keratin, type I cytoskeletal 12 Human genes 0.000 description 2
- 102100025759 Keratin, type II cytoskeletal 3 Human genes 0.000 description 2
- 108010070514 Keratin-1 Proteins 0.000 description 2
- 108010065086 Keratin-12 Proteins 0.000 description 2
- 108010066302 Keratin-19 Proteins 0.000 description 2
- 108010070918 Keratin-3 Proteins 0.000 description 2
- 102000011782 Keratins Human genes 0.000 description 2
- 108010076876 Keratins Proteins 0.000 description 2
- 208000034607 Kindler epidermolysis bullosa Diseases 0.000 description 2
- 201000004290 Kindler syndrome Diseases 0.000 description 2
- 108700021430 Kruppel-Like Factor 4 Proteins 0.000 description 2
- 102000043129 MHC class I family Human genes 0.000 description 2
- 108091054437 MHC class I family Proteins 0.000 description 2
- 241000124008 Mammalia Species 0.000 description 2
- 101710135898 Myc proto-oncogene protein Proteins 0.000 description 2
- 229930040373 Paraformaldehyde Natural products 0.000 description 2
- 238000001190 Q-PCR Methods 0.000 description 2
- 101100247004 Rattus norvegicus Qsox1 gene Proteins 0.000 description 2
- 101150086694 SLC22A3 gene Proteins 0.000 description 2
- 102000040945 Transcription factor Human genes 0.000 description 2
- 108091023040 Transcription factor Proteins 0.000 description 2
- 101710150448 Transcriptional regulator Myc Proteins 0.000 description 2
- 101710140697 Tumor protein 63 Proteins 0.000 description 2
- 230000005856 abnormality Effects 0.000 description 2
- SHGAZHPCJJPHSC-YCNIQYBTSA-N all-trans-retinoic acid Chemical compound OC(=O)\C=C(/C)\C=C\C=C(/C)\C=C\C1=C(C)CCCC1(C)C SHGAZHPCJJPHSC-YCNIQYBTSA-N 0.000 description 2
- 238000003491 array Methods 0.000 description 2
- 238000003556 assay Methods 0.000 description 2
- 229940088623 biologically active substance Drugs 0.000 description 2
- 239000003153 chemical reaction reagent Substances 0.000 description 2
- 230000005757 colony formation Effects 0.000 description 2
- 238000010293 colony formation assay Methods 0.000 description 2
- 230000000052 comparative effect Effects 0.000 description 2
- 230000007423 decrease Effects 0.000 description 2
- 230000037336 dry skin Effects 0.000 description 2
- 210000002257 embryonic structure Anatomy 0.000 description 2
- 210000001339 epidermal cell Anatomy 0.000 description 2
- 210000002514 epidermal stem cell Anatomy 0.000 description 2
- 210000002919 epithelial cell Anatomy 0.000 description 2
- 230000005713 exacerbation Effects 0.000 description 2
- 238000009472 formulation Methods 0.000 description 2
- 239000008273 gelatin Substances 0.000 description 2
- 229920000159 gelatin Polymers 0.000 description 2
- 235000019322 gelatine Nutrition 0.000 description 2
- 235000011852 gelatine desserts Nutrition 0.000 description 2
- 230000002068 genetic effect Effects 0.000 description 2
- ZDXPYRJPNDTMRX-UHFFFAOYSA-N glutamine Natural products OC(=O)C(N)CCC(N)=O ZDXPYRJPNDTMRX-UHFFFAOYSA-N 0.000 description 2
- 238000010166 immunofluorescence Methods 0.000 description 2
- 238000010185 immunofluorescence analysis Methods 0.000 description 2
- 230000005847 immunogenicity Effects 0.000 description 2
- 238000011534 incubation Methods 0.000 description 2
- 210000004379 membrane Anatomy 0.000 description 2
- 239000012528 membrane Substances 0.000 description 2
- 210000003716 mesoderm Anatomy 0.000 description 2
- 238000000386 microscopy Methods 0.000 description 2
- 230000035772 mutation Effects 0.000 description 2
- GVUGOAYIVIDWIO-UFWWTJHBSA-N nepidermin Chemical compound C([C@@H](C(=O)N[C@@H]([C@@H](C)CC)C(=O)NCC(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CS)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC=1C=CC(O)=CC=1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCNC(N)=N)C(O)=O)NC(=O)CNC(=O)[C@@H](NC(=O)[C@@H](NC(=O)[C@H](CS)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CS)NC(=O)[C@H](C)NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@H](CCCCN)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](C)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@@H](NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@H](CCSC)NC(=O)[C@H](CS)NC(=O)[C@@H](NC(=O)CNC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CC=1NC=NC=1)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CS)NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)CNC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CC=1NC=NC=1)NC(=O)[C@H](CO)NC(=O)[C@H](CC(C)C)NC(=O)[C@H]1N(CCC1)C(=O)[C@H](CS)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CO)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CO)NC(=O)[C@@H](N)CC(N)=O)C(C)C)[C@@H](C)CC)C(C)C)C(C)C)C1=CC=C(O)C=C1 GVUGOAYIVIDWIO-UFWWTJHBSA-N 0.000 description 2
- 208000015122 neurodegenerative disease Diseases 0.000 description 2
- 229920002866 paraformaldehyde Polymers 0.000 description 2
- 239000004033 plastic Substances 0.000 description 2
- 229920003023 plastic Polymers 0.000 description 2
- 239000004417 polycarbonate Substances 0.000 description 2
- 229920000515 polycarbonate Polymers 0.000 description 2
- 230000002265 prevention Effects 0.000 description 2
- 230000002829 reductive effect Effects 0.000 description 2
- 229930002330 retinoic acid Natural products 0.000 description 2
- 238000010839 reverse transcription Methods 0.000 description 2
- 208000017520 skin disease Diseases 0.000 description 2
- DAEPDZWVDSPTHF-UHFFFAOYSA-M sodium pyruvate Chemical compound [Na+].CC(=O)C([O-])=O DAEPDZWVDSPTHF-UHFFFAOYSA-M 0.000 description 2
- 241000894007 species Species 0.000 description 2
- 230000004083 survival effect Effects 0.000 description 2
- 230000001988 toxicity Effects 0.000 description 2
- 231100000419 toxicity Toxicity 0.000 description 2
- 230000010474 transient expression Effects 0.000 description 2
- 229960001727 tretinoin Drugs 0.000 description 2
- VBEQCZHXXJYVRD-GACYYNSASA-N uroanthelone Chemical compound C([C@@H](C(=O)N[C@H](C(=O)N[C@@H](CS)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CS)C(=O)N[C@H](C(=O)N[C@@H]([C@@H](C)CC)C(=O)NCC(=O)N[C@@H](CC=1C=CC(O)=CC=1)C(=O)N[C@@H](CO)C(=O)NCC(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CS)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCNC(N)=N)C(O)=O)C(C)C)[C@@H](C)O)NC(=O)[C@H](CO)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CO)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@@H](NC(=O)[C@H](CC=1NC=NC=1)NC(=O)[C@H](CCSC)NC(=O)[C@H](CS)NC(=O)[C@@H](NC(=O)CNC(=O)CNC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CS)NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)CNC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@H](CO)NC(=O)[C@H](CO)NC(=O)[C@H]1N(CCC1)C(=O)[C@H](CS)NC(=O)CNC(=O)[C@H]1N(CCC1)C(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@H](CO)NC(=O)[C@@H](N)CC(N)=O)C(C)C)[C@@H](C)CC)C1=CC=C(O)C=C1 VBEQCZHXXJYVRD-GACYYNSASA-N 0.000 description 2
- DGVVWUTYPXICAM-UHFFFAOYSA-N β‐Mercaptoethanol Chemical compound OCCS DGVVWUTYPXICAM-UHFFFAOYSA-N 0.000 description 2
- YTPMCWYIRHLEGM-BQYQJAHWSA-N 1-[(e)-2-propylsulfonylethenyl]sulfonylpropane Chemical compound CCCS(=O)(=O)\C=C\S(=O)(=O)CCC YTPMCWYIRHLEGM-BQYQJAHWSA-N 0.000 description 1
- RSEBUVRVKCANEP-UHFFFAOYSA-N 2-pyrroline Chemical compound C1CC=CN1 RSEBUVRVKCANEP-UHFFFAOYSA-N 0.000 description 1
- FWBHETKCLVMNFS-UHFFFAOYSA-N 4',6-Diamino-2-phenylindol Chemical compound C1=CC(C(=N)N)=CC=C1C1=CC2=CC=C(C(N)=N)C=C2N1 FWBHETKCLVMNFS-UHFFFAOYSA-N 0.000 description 1
- ZCYVEMRRCGMTRW-UHFFFAOYSA-N 7553-56-2 Chemical compound [I] ZCYVEMRRCGMTRW-UHFFFAOYSA-N 0.000 description 1
- 239000004382 Amylase Substances 0.000 description 1
- 102000013142 Amylases Human genes 0.000 description 1
- 108010065511 Amylases Proteins 0.000 description 1
- 235000009024 Ceanothus sanguineus Nutrition 0.000 description 1
- 102000012422 Collagen Type I Human genes 0.000 description 1
- 108010022452 Collagen Type I Proteins 0.000 description 1
- 230000004544 DNA amplification Effects 0.000 description 1
- 102000007260 Deoxyribonuclease I Human genes 0.000 description 1
- 108010008532 Deoxyribonuclease I Proteins 0.000 description 1
- 208000017452 Epidermal disease Diseases 0.000 description 1
- 101800003838 Epidermal growth factor Proteins 0.000 description 1
- 102000003886 Glycoproteins Human genes 0.000 description 1
- 108090000288 Glycoproteins Proteins 0.000 description 1
- 101001052035 Homo sapiens Fibroblast growth factor 2 Proteins 0.000 description 1
- 101001046960 Homo sapiens Keratin, type II cytoskeletal 1 Proteins 0.000 description 1
- 241000701806 Human papillomavirus Species 0.000 description 1
- 206010061598 Immunodeficiency Diseases 0.000 description 1
- 208000029462 Immunodeficiency disease Diseases 0.000 description 1
- 206010062016 Immunosuppression Diseases 0.000 description 1
- 206010061218 Inflammation Diseases 0.000 description 1
- 101710183399 Keratin, type I cytoskeletal 19 Proteins 0.000 description 1
- ZDXPYRJPNDTMRX-VKHMYHEASA-N L-glutamine Chemical compound OC(=O)[C@@H](N)CCC(N)=O ZDXPYRJPNDTMRX-VKHMYHEASA-N 0.000 description 1
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 description 1
- 102100024629 Laminin subunit beta-3 Human genes 0.000 description 1
- 240000003553 Leptospermum scoparium Species 0.000 description 1
- 235000015459 Lycium barbarum Nutrition 0.000 description 1
- 108700018351 Major Histocompatibility Complex Proteins 0.000 description 1
- 239000004909 Moisturizer Substances 0.000 description 1
- 102100025744 Mothers against decapentaplegic homolog 1 Human genes 0.000 description 1
- 102100030610 Mothers against decapentaplegic homolog 5 Human genes 0.000 description 1
- 101710143113 Mothers against decapentaplegic homolog 5 Proteins 0.000 description 1
- 102100030608 Mothers against decapentaplegic homolog 7 Human genes 0.000 description 1
- 241000283973 Oryctolagus cuniculus Species 0.000 description 1
- 238000010222 PCR analysis Methods 0.000 description 1
- 206010034972 Photosensitivity reaction Diseases 0.000 description 1
- 102100033237 Pro-epidermal growth factor Human genes 0.000 description 1
- 238000011579 SCID mouse model Methods 0.000 description 1
- 101700032040 SMAD1 Proteins 0.000 description 1
- 101700026522 SMAD7 Proteins 0.000 description 1
- 238000010818 SYBR green PCR Master Mix Methods 0.000 description 1
- 206010040840 Skin erosion Diseases 0.000 description 1
- 102000007374 Smad Proteins Human genes 0.000 description 1
- 108010007945 Smad Proteins Proteins 0.000 description 1
- 229920002472 Starch Polymers 0.000 description 1
- 206010042496 Sunburn Diseases 0.000 description 1
- 102000043168 TGF-beta family Human genes 0.000 description 1
- 108091085018 TGF-beta family Proteins 0.000 description 1
- AUYYCJSJGJYCDS-LBPRGKRZSA-N Thyrolar Chemical compound IC1=CC(C[C@H](N)C(O)=O)=CC(I)=C1OC1=CC=C(O)C(I)=C1 AUYYCJSJGJYCDS-LBPRGKRZSA-N 0.000 description 1
- 102000004887 Transforming Growth Factor beta Human genes 0.000 description 1
- 108090001012 Transforming Growth Factor beta Proteins 0.000 description 1
- 229920004890 Triton X-100 Polymers 0.000 description 1
- 239000013504 Triton X-100 Substances 0.000 description 1
- 230000009471 action Effects 0.000 description 1
- 230000003213 activating effect Effects 0.000 description 1
- 239000013543 active substance Substances 0.000 description 1
- 239000000853 adhesive Substances 0.000 description 1
- 230000001070 adhesive effect Effects 0.000 description 1
- 150000001413 amino acids Chemical group 0.000 description 1
- 230000003321 amplification Effects 0.000 description 1
- 235000019418 amylase Nutrition 0.000 description 1
- 230000002424 anti-apoptotic effect Effects 0.000 description 1
- 229940121363 anti-inflammatory agent Drugs 0.000 description 1
- 239000002260 anti-inflammatory agent Substances 0.000 description 1
- 239000003963 antioxidant agent Substances 0.000 description 1
- 235000006708 antioxidants Nutrition 0.000 description 1
- 210000000270 basal cell Anatomy 0.000 description 1
- 210000002469 basement membrane Anatomy 0.000 description 1
- 230000009286 beneficial effect Effects 0.000 description 1
- 239000012867 bioactive agent Substances 0.000 description 1
- 230000004071 biological effect Effects 0.000 description 1
- 230000033228 biological regulation Effects 0.000 description 1
- 230000015572 biosynthetic process Effects 0.000 description 1
- 210000000988 bone and bone Anatomy 0.000 description 1
- 235000014633 carbohydrates Nutrition 0.000 description 1
- 150000001720 carbohydrates Chemical class 0.000 description 1
- 230000022131 cell cycle Effects 0.000 description 1
- 230000005779 cell damage Effects 0.000 description 1
- 208000037887 cell injury Diseases 0.000 description 1
- 230000036755 cellular response Effects 0.000 description 1
- 238000006243 chemical reaction Methods 0.000 description 1
- 239000011248 coating agent Substances 0.000 description 1
- 238000000576 coating method Methods 0.000 description 1
- 239000002299 complementary DNA Substances 0.000 description 1
- 238000007796 conventional method Methods 0.000 description 1
- 239000006071 cream Substances 0.000 description 1
- 238000012136 culture method Methods 0.000 description 1
- 210000004748 cultured cell Anatomy 0.000 description 1
- 230000007547 defect Effects 0.000 description 1
- 230000007123 defense Effects 0.000 description 1
- 230000006735 deficit Effects 0.000 description 1
- 230000003412 degenerative effect Effects 0.000 description 1
- 238000001514 detection method Methods 0.000 description 1
- 235000014113 dietary fatty acids Nutrition 0.000 description 1
- 230000029087 digestion Effects 0.000 description 1
- 230000036267 drug metabolism Effects 0.000 description 1
- 238000001035 drying Methods 0.000 description 1
- 239000000975 dye Substances 0.000 description 1
- 230000004064 dysfunction Effects 0.000 description 1
- 210000001705 ectoderm cell Anatomy 0.000 description 1
- 230000013020 embryo development Effects 0.000 description 1
- 230000007613 environmental effect Effects 0.000 description 1
- 238000001952 enzyme assay Methods 0.000 description 1
- YQGOJNYOYNNSMM-UHFFFAOYSA-N eosin Chemical compound [Na+].OC(=O)C1=CC=CC=C1C1=C2C=C(Br)C(=O)C(Br)=C2OC2=C(Br)C(O)=C(Br)C=C21 YQGOJNYOYNNSMM-UHFFFAOYSA-N 0.000 description 1
- 229940116977 epidermal growth factor Drugs 0.000 description 1
- 239000003797 essential amino acid Substances 0.000 description 1
- 235000020776 essential amino acid Nutrition 0.000 description 1
- 238000011156 evaluation Methods 0.000 description 1
- 238000004299 exfoliation Methods 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 238000000605 extraction Methods 0.000 description 1
- 229930195729 fatty acid Natural products 0.000 description 1
- 239000000194 fatty acid Substances 0.000 description 1
- -1 fatty acid esters Chemical class 0.000 description 1
- 210000003953 foreskin Anatomy 0.000 description 1
- 230000004927 fusion Effects 0.000 description 1
- 239000000499 gel Substances 0.000 description 1
- 239000003102 growth factor Substances 0.000 description 1
- 230000036541 health Effects 0.000 description 1
- 238000009396 hybridization Methods 0.000 description 1
- 229940031574 hydroxymethyl cellulose Drugs 0.000 description 1
- 229920003063 hydroxymethyl cellulose Polymers 0.000 description 1
- 230000002519 immonomodulatory effect Effects 0.000 description 1
- 238000003365 immunocytochemistry Methods 0.000 description 1
- 230000007813 immunodeficiency Effects 0.000 description 1
- 238000003125 immunofluorescent labeling Methods 0.000 description 1
- 238000003364 immunohistochemistry Methods 0.000 description 1
- 230000001771 impaired effect Effects 0.000 description 1
- 238000002513 implantation Methods 0.000 description 1
- 238000010874 in vitro model Methods 0.000 description 1
- 230000001939 inductive effect Effects 0.000 description 1
- 230000004054 inflammatory process Effects 0.000 description 1
- 230000005764 inhibitory process Effects 0.000 description 1
- 230000000977 initiatory effect Effects 0.000 description 1
- 238000002347 injection Methods 0.000 description 1
- 239000007924 injection Substances 0.000 description 1
- 230000002452 interceptive effect Effects 0.000 description 1
- 230000006799 invasive growth in response to glucose limitation Effects 0.000 description 1
- 229910052740 iodine Inorganic materials 0.000 description 1
- 239000011630 iodine Substances 0.000 description 1
- 230000007794 irritation Effects 0.000 description 1
- 238000002955 isolation Methods 0.000 description 1
- 230000002147 killing effect Effects 0.000 description 1
- 239000008101 lactose Substances 0.000 description 1
- 230000003902 lesion Effects 0.000 description 1
- 239000003446 ligand Substances 0.000 description 1
- 230000000670 limiting effect Effects 0.000 description 1
- 239000006210 lotion Substances 0.000 description 1
- 238000012423 maintenance Methods 0.000 description 1
- 230000003211 malignant effect Effects 0.000 description 1
- 238000013411 master cell bank Methods 0.000 description 1
- 239000011159 matrix material Substances 0.000 description 1
- 238000002844 melting Methods 0.000 description 1
- 230000008018 melting Effects 0.000 description 1
- 108020004999 messenger RNA Proteins 0.000 description 1
- 230000000813 microbial effect Effects 0.000 description 1
- 238000007431 microscopic evaluation Methods 0.000 description 1
- 239000007758 minimum essential medium Substances 0.000 description 1
- 230000001333 moisturizer Effects 0.000 description 1
- 230000009456 molecular mechanism Effects 0.000 description 1
- 230000000921 morphogenic effect Effects 0.000 description 1
- 238000003199 nucleic acid amplification method Methods 0.000 description 1
- 235000016709 nutrition Nutrition 0.000 description 1
- 230000035764 nutrition Effects 0.000 description 1
- 239000003921 oil Substances 0.000 description 1
- 235000019198 oils Nutrition 0.000 description 1
- 230000002188 osteogenic effect Effects 0.000 description 1
- 239000012188 paraffin wax Substances 0.000 description 1
- 230000001717 pathogenic effect Effects 0.000 description 1
- 239000002304 perfume Substances 0.000 description 1
- 208000007578 phototoxic dermatitis Diseases 0.000 description 1
- 231100000018 phototoxicity Toxicity 0.000 description 1
- 229920001184 polypeptide Polymers 0.000 description 1
- 230000003389 potentiating effect Effects 0.000 description 1
- 239000002243 precursor Substances 0.000 description 1
- 102000004196 processed proteins & peptides Human genes 0.000 description 1
- 108090000765 processed proteins & peptides Proteins 0.000 description 1
- ZVJHJDDKYZXRJI-UHFFFAOYSA-N pyrroline Natural products C1CC=NC1 ZVJHJDDKYZXRJI-UHFFFAOYSA-N 0.000 description 1
- 238000011002 quantification Methods 0.000 description 1
- 239000001397 quillaja saponaria molina bark Substances 0.000 description 1
- 230000005855 radiation Effects 0.000 description 1
- 238000001959 radiotherapy Methods 0.000 description 1
- 238000012755 real-time RT-PCR analysis Methods 0.000 description 1
- 238000011084 recovery Methods 0.000 description 1
- 230000004043 responsiveness Effects 0.000 description 1
- 239000012266 salt solution Substances 0.000 description 1
- 229930182490 saponin Natural products 0.000 description 1
- 150000007949 saponins Chemical class 0.000 description 1
- 238000007423 screening assay Methods 0.000 description 1
- 230000028327 secretion Effects 0.000 description 1
- 238000000926 separation method Methods 0.000 description 1
- 230000036556 skin irritation Effects 0.000 description 1
- 229940054269 sodium pyruvate Drugs 0.000 description 1
- 239000007787 solid Substances 0.000 description 1
- 239000007921 spray Substances 0.000 description 1
- 230000010473 stable expression Effects 0.000 description 1
- 235000019698 starch Nutrition 0.000 description 1
- 239000008107 starch Substances 0.000 description 1
- 230000004936 stimulating effect Effects 0.000 description 1
- 238000013517 stratification Methods 0.000 description 1
- 239000000126 substance Substances 0.000 description 1
- GECHUMIMRBOMGK-UHFFFAOYSA-N sulfapyridine Chemical compound C1=CC(N)=CC=C1S(=O)(=O)NC1=CC=CC=N1 GECHUMIMRBOMGK-UHFFFAOYSA-N 0.000 description 1
- 230000001629 suppression Effects 0.000 description 1
- 230000020382 suppression by virus of host antigen processing and presentation of peptide antigen via MHC class I Effects 0.000 description 1
- 238000003786 synthesis reaction Methods 0.000 description 1
- 230000009885 systemic effect Effects 0.000 description 1
- 239000008399 tap water Substances 0.000 description 1
- 235000020679 tap water Nutrition 0.000 description 1
- ZRKFYGHZFMAOKI-QMGMOQQFSA-N tgfbeta Chemical compound C([C@H](NC(=O)[C@H](C(C)C)NC(=O)CNC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H]([C@@H](C)O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H]([C@@H](C)O)NC(=O)[C@H](CC(C)C)NC(=O)CNC(=O)[C@H](C)NC(=O)[C@H](CO)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@@H](NC(=O)[C@H](C)NC(=O)[C@H](C)NC(=O)[C@@H](NC(=O)[C@H](CC(C)C)NC(=O)[C@@H](N)CCSC)C(C)C)[C@@H](C)CC)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CC=1C=CC=CC=1)C(=O)N[C@@H](C)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](C)C(=O)N[C@@H](CC=1C=CC=CC=1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](C)C(=O)N[C@@H](CC(C)C)C(=O)N1[C@@H](CCC1)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CO)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC(C)C)C(O)=O)C1=CC=C(O)C=C1 ZRKFYGHZFMAOKI-QMGMOQQFSA-N 0.000 description 1
- 230000002110 toxicologic effect Effects 0.000 description 1
- 231100000759 toxicological effect Toxicity 0.000 description 1
- 238000012546 transfer Methods 0.000 description 1
- 229940035722 triiodothyronine Drugs 0.000 description 1
- 241001430294 unidentified retrovirus Species 0.000 description 1
- 229920002554 vinyl polymer Polymers 0.000 description 1
- 239000013603 viral vector Substances 0.000 description 1
- 238000005406 washing Methods 0.000 description 1
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 1
- 230000029663 wound healing Effects 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/06—Animal cells or tissues; Human cells or tissues
- C12N5/0602—Vertebrate cells
- C12N5/0625—Epidermal cells, skin cells; Cells of the oral mucosa
- C12N5/0629—Keratinocytes; Whole skin
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/06—Animal cells or tissues; Human cells or tissues
- C12N5/0602—Vertebrate cells
- C12N5/0603—Embryonic cells ; Embryoid bodies
- C12N5/0606—Pluripotent embryonic cells, e.g. embryonic stem cells [ES]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/02—Prostheses implantable into the body
- A61F2/10—Hair or skin implants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K35/00—Medicinal preparations containing materials or reaction products thereof with undetermined constitution
- A61K35/12—Materials from mammals; Compositions comprising non-specified tissues or cells; Compositions comprising non-embryonic stem cells; Genetically modified cells
- A61K35/36—Skin; Hair; Nails; Sebaceous glands; Cerumen; Epidermis; Epithelial cells; Keratinocytes; Langerhans cells; Ectodermal cells
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L27/00—Materials for grafts or prostheses or for coating grafts or prostheses
- A61L27/36—Materials for grafts or prostheses or for coating grafts or prostheses containing ingredients of undetermined constitution or reaction products thereof, e.g. transplant tissue, natural bone, extracellular matrix
- A61L27/38—Materials for grafts or prostheses or for coating grafts or prostheses containing ingredients of undetermined constitution or reaction products thereof, e.g. transplant tissue, natural bone, extracellular matrix containing added animal cells
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
- A61P17/02—Drugs for dermatological disorders for treating wounds, ulcers, burns, scars, keloids, or the like
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2500/00—Specific components of cell culture medium
- C12N2500/30—Organic components
- C12N2500/40—Nucleotides, nucleosides or bases
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/01—Modulators of cAMP or cGMP, e.g. non-hydrolysable analogs, phosphodiesterase inhibitors, cholera toxin
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/10—Growth factors
- C12N2501/11—Epidermal growth factor [EGF]
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/10—Growth factors
- C12N2501/155—Bone morphogenic proteins [BMP]; Osteogenins; Osteogenic factor; Bone inducing factor
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/30—Hormones
- C12N2501/33—Insulin
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/30—Hormones
- C12N2501/38—Hormones with nuclear receptors
- C12N2501/395—Thyroid hormones
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2502/00—Coculture with; Conditioned medium produced by
- C12N2502/13—Coculture with; Conditioned medium produced by connective tissue cells; generic mesenchyme cells, e.g. so-called "embryonic fibroblasts"
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2506/00—Differentiation of animal cells from one lineage to another; Differentiation of pluripotent cells
- C12N2506/02—Differentiation of animal cells from one lineage to another; Differentiation of pluripotent cells from embryonic cells
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2506/00—Differentiation of animal cells from one lineage to another; Differentiation of pluripotent cells
- C12N2506/45—Differentiation of animal cells from one lineage to another; Differentiation of pluripotent cells from artificially induced pluripotent stem cells
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Biomedical Technology (AREA)
- Chemical & Material Sciences (AREA)
- Zoology (AREA)
- Bioinformatics & Cheminformatics (AREA)
- General Health & Medical Sciences (AREA)
- Biotechnology (AREA)
- Organic Chemistry (AREA)
- Dermatology (AREA)
- Cell Biology (AREA)
- Genetics & Genomics (AREA)
- Wood Science & Technology (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Animal Behavior & Ethology (AREA)
- Biochemistry (AREA)
- General Engineering & Computer Science (AREA)
- Medicinal Chemistry (AREA)
- Microbiology (AREA)
- Developmental Biology & Embryology (AREA)
- Transplantation (AREA)
- Pharmacology & Pharmacy (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Reproductive Health (AREA)
- Gynecology & Obstetrics (AREA)
- Oral & Maxillofacial Surgery (AREA)
- Epidemiology (AREA)
- Heart & Thoracic Surgery (AREA)
- Cardiology (AREA)
- Virology (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Vascular Medicine (AREA)
- Immunology (AREA)
- General Chemical & Material Sciences (AREA)
- Botany (AREA)
- Micro-Organisms Or Cultivation Processes Thereof (AREA)
- Measuring Or Testing Involving Enzymes Or Micro-Organisms (AREA)
- Materials For Medical Uses (AREA)
Applications Claiming Priority (8)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP08305320 | 2008-06-25 | ||
| EP08305320.7 | 2008-06-25 | ||
| EP08305447 | 2008-08-04 | ||
| EP08305447.8 | 2008-08-04 | ||
| US9095708P | 2008-08-22 | 2008-08-22 | |
| US61/090,957 | 2008-08-22 | ||
| EP09305077.1 | 2009-01-28 | ||
| EP09305077 | 2009-01-28 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| KR20110022713A true KR20110022713A (ko) | 2011-03-07 |
Family
ID=40863382
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020117001947A Withdrawn KR20110022713A (ko) | 2008-06-25 | 2009-06-23 | 인간 만능줄기세포로부터 인간 피부 대체물을 제조하는 방법 |
Country Status (8)
| Country | Link |
|---|---|
| US (1) | US20110165130A1 (enExample) |
| EP (1) | EP2297299A1 (enExample) |
| JP (1) | JP2011525370A (enExample) |
| KR (1) | KR20110022713A (enExample) |
| CN (1) | CN102186969A (enExample) |
| AU (1) | AU2009264336A1 (enExample) |
| BR (1) | BRPI0914422A2 (enExample) |
| WO (1) | WO2009156398A1 (enExample) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2019004533A1 (ko) * | 2017-06-30 | 2019-01-03 | 주식회사 스템랩 | 소변세포로부터 케라티노사이트 줄기세포로의 직접 역분화 방법 및 역분화된 케라티노사이트 줄기세포를 이용한 피부재생 촉진용 조성물의 제조방법 |
Families Citing this family (27)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2010134619A1 (ja) * | 2009-05-18 | 2010-11-25 | 国立大学法人東北大学 | 人工多能性幹細胞からの上皮系前駆細胞・幹細胞群及び角膜上皮細胞群の分化誘導方法 |
| US8529883B2 (en) * | 2010-05-07 | 2013-09-10 | Fibrocell Technologies, Inc. | Dosage unit formulations of autologous dermal fibroblasts |
| RU2458983C1 (ru) * | 2011-07-18 | 2012-08-20 | Российская Федерация, От Имени Которой Выступает Министерство Образования И Науки Российской Федерации | Способ получения индуцированных плюрипотентных стволовых клеток из фибробластов пациентов с болезнью хангингтона |
| AU2014239954B2 (en) * | 2013-03-15 | 2020-07-16 | The Jackson Laboratory | Isolation of non-embryonic stem cells and uses thereof |
| US20160237400A1 (en) * | 2013-03-15 | 2016-08-18 | The Jackson Laboratory | Isolation of non-embryonic stem cells and uses thereof |
| US9545302B2 (en) | 2013-11-20 | 2017-01-17 | Dermagenesis Llc | Skin printing and auto-grafting |
| CN104789521B (zh) * | 2014-01-22 | 2017-11-07 | 广州康睿生物医药科技股份有限公司 | 一种晶状体上皮干细胞的分离培养方法 |
| SG10202103834VA (en) * | 2014-02-21 | 2021-05-28 | Sumitomo Dainippon Pharma Co Ltd | Eye disease treatment agent, screening method therefor, and method for predicting rejection response associated with retinal pigment epithelial cell transplant |
| WO2016057833A1 (en) * | 2014-10-10 | 2016-04-14 | Vygantas Garrett | Methods of manufacturing keratin products and uses thereof |
| JP6736253B2 (ja) * | 2014-12-22 | 2020-08-05 | ザ チャイニーズ ユニバーシティ オブ ホンコン | 体細胞から多能性幹細胞を作製するための機械的操作とプログラミンの併用 |
| US10167451B2 (en) | 2014-12-22 | 2019-01-01 | The Chinese University Of Hong Kong | Combinational use of mechanical manipulation and programin derivatives to increase Oct4, Sox2, or Nanog expression in fibroblasts |
| GB201510913D0 (en) * | 2015-06-22 | 2015-08-05 | Nat University Of Singapore And Agency For Science Technology And Res | Vascularized tissue, skin or mucosa quivalent |
| CA3002164A1 (en) * | 2015-10-21 | 2017-04-27 | Indiana University Research And Technology Corporation | Derivation of human skin organoids from pluripotent stem cells |
| AU2016341982B2 (en) | 2015-10-21 | 2022-07-07 | Indiana University Research And Technology Corporation | Methods of generating human inner ear sensory epithelia and sensory neurons |
| US10273549B2 (en) | 2016-04-21 | 2019-04-30 | Vitrolabs Inc. | Engineered skin equivalent, method of manufacture thereof and products derived therefrom |
| EP3551746A1 (en) * | 2016-12-09 | 2019-10-16 | The University of Chicago | Tissue organoids |
| CN106754652B (zh) * | 2017-03-06 | 2019-04-02 | 广州润虹医药科技股份有限公司 | iPS细胞分化成外胚层祖细胞的无血清诱导培养基及诱导方法 |
| CN106834214B (zh) * | 2017-03-27 | 2020-02-14 | 广州润虹医药科技股份有限公司 | 一种诱导多能干细胞形成角质形成细胞的诱导培养基及诱导方法 |
| CN107988147B (zh) * | 2017-12-19 | 2021-11-09 | 江汉大学 | 基于器官芯片与诱导多能干细胞的定向分化用于3d拟表皮构建的方法 |
| CN110093306B (zh) * | 2018-01-31 | 2022-12-02 | 中山大学 | 人工毛囊及其制备方法和应用 |
| EP3875581A1 (en) | 2020-03-02 | 2021-09-08 | Centre d'Etude des Cellules Souches (CECS) | Automated method for preparing keratinocytes |
| EP3875124A1 (fr) | 2020-03-02 | 2021-09-08 | Urgo Recherche Innovation Et Developpement | Procede d'obtention d'un tissu dermo-epidermique pre-vascularise |
| EP3875580A1 (en) | 2020-03-02 | 2021-09-08 | Centre d'Etude des Cellules Souches (CECS) | Methods for preparing keratinocytes |
| CN112941015B (zh) * | 2021-03-19 | 2022-03-18 | 上海爱萨尔生物科技有限公司 | 一种基于多能干细胞分化制备角质细胞的添加剂及方法 |
| CN112980775B (zh) * | 2021-03-19 | 2024-01-23 | 上海爱萨尔生物科技有限公司 | 一种基于多能干细胞分化制备角质细胞的培养液 |
| CN113046300B (zh) * | 2021-03-19 | 2024-08-06 | 上海爱萨尔生物科技有限公司 | 一种基于多能干细胞分化制备角质细胞的培养方法 |
| JP7315184B2 (ja) * | 2022-02-16 | 2023-07-26 | 株式会社コーセー | 多能性幹細胞から表皮角化細胞への分化誘導方法 |
Family Cites Families (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR2825370A1 (fr) * | 2001-05-31 | 2002-12-06 | Inst Nat Sante Rech Med | Keratinocytes obtenus a partir de cellules souches embryonnaires de mammiferes |
-
2009
- 2009-06-23 CN CN2009801256559A patent/CN102186969A/zh active Pending
- 2009-06-23 JP JP2011515354A patent/JP2011525370A/ja active Pending
- 2009-06-23 AU AU2009264336A patent/AU2009264336A1/en not_active Abandoned
- 2009-06-23 KR KR1020117001947A patent/KR20110022713A/ko not_active Withdrawn
- 2009-06-23 WO PCT/EP2009/057817 patent/WO2009156398A1/en not_active Ceased
- 2009-06-23 US US12/999,129 patent/US20110165130A1/en not_active Abandoned
- 2009-06-23 EP EP09769247A patent/EP2297299A1/en not_active Withdrawn
- 2009-06-23 BR BRPI0914422A patent/BRPI0914422A2/pt not_active IP Right Cessation
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2019004533A1 (ko) * | 2017-06-30 | 2019-01-03 | 주식회사 스템랩 | 소변세포로부터 케라티노사이트 줄기세포로의 직접 역분화 방법 및 역분화된 케라티노사이트 줄기세포를 이용한 피부재생 촉진용 조성물의 제조방법 |
Also Published As
| Publication number | Publication date |
|---|---|
| BRPI0914422A2 (pt) | 2017-03-21 |
| JP2011525370A (ja) | 2011-09-22 |
| US20110165130A1 (en) | 2011-07-07 |
| EP2297299A1 (en) | 2011-03-23 |
| WO2009156398A1 (en) | 2009-12-30 |
| AU2009264336A1 (en) | 2009-12-30 |
| CN102186969A (zh) | 2011-09-14 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| KR20110022713A (ko) | 인간 만능줄기세포로부터 인간 피부 대체물을 제조하는 방법 | |
| JP2011525370A5 (enExample) | ||
| US10344266B2 (en) | Cell mass capable of serving as a primitive organ-like structure comprised of a plurality of cell types of somatic origin | |
| AU2009228756B2 (en) | Methods for producing hair microfollicles and de novo papillae and their use for in vitro tests and in vivo implantations | |
| KR20120120971A (ko) | 인간 다능성 줄기 세포로부터 인간 멜라닌세포를 제조하는 방법 | |
| CN106661547B (zh) | 调控毛发生长的方法和组合物 | |
| US20110321180A1 (en) | Compositions and methods to generate pilosebaceous units | |
| JP6626245B2 (ja) | 再構成皮膚の作製用の組成物および方法 | |
| JP2016516394A (ja) | ヒト線維芽細胞において特徴的な幹細胞であるMuse細胞から容易に分化した機能性メラノサイトの使用 | |
| Sakai | Development and regeneration of salivary gland toward for clinical application | |
| US20120148541A1 (en) | Compositions and methods to generate pilosebaceous units | |
| Yonetani et al. | In vitro expansion of immature melanoblasts and their ability to repopulate melanocyte stem cells in the hair follicle | |
| US20220002678A1 (en) | Composition for reconstituting human skin tissue having hair follicles, human skin tissue model animal, and production method thereof | |
| Dong et al. | Enrichment of epidermal stem cells by rapid adherence and analysis of the reciprocal interaction of epidermal stem cells with neighboring cells using an organotypic system | |
| TW202340454A (zh) | 將誘導性多能幹細胞分化為視網膜色素上皮細胞之方法、視網膜色素上皮細胞以及使用該視網膜色素上皮細胞之方法 | |
| A Rahman | The development of in vitro models of human salivary glands | |
| Shamis | hESC-and iPSC-Derived Fibroblasts for Skin Engineering and Repair |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PA0105 | International application |
Patent event date: 20110125 Patent event code: PA01051R01D Comment text: International Patent Application |
|
| PG1501 | Laying open of application | ||
| PC1203 | Withdrawal of no request for examination | ||
| WITN | Application deemed withdrawn, e.g. because no request for examination was filed or no examination fee was paid |