JP7581347B2 - 炭素被覆ニッケル-アルミニウムナノ複合材料、その調製方法およびその適用 - Google Patents
炭素被覆ニッケル-アルミニウムナノ複合材料、その調製方法およびその適用 Download PDFInfo
- Publication number
- JP7581347B2 JP7581347B2 JP2022523622A JP2022523622A JP7581347B2 JP 7581347 B2 JP7581347 B2 JP 7581347B2 JP 2022523622 A JP2022523622 A JP 2022523622A JP 2022523622 A JP2022523622 A JP 2022523622A JP 7581347 B2 JP7581347 B2 JP 7581347B2
- Authority
- JP
- Japan
- Prior art keywords
- nickel
- aluminum
- carbon
- nanocomposite
- salt
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
Images
Classifications
-
- F—MECHANICAL ENGINEERING; LIGHTING; HEATING; WEAPONS; BLASTING
- F23—COMBUSTION APPARATUS; COMBUSTION PROCESSES
- F23C—METHODS OR APPARATUS FOR COMBUSTION USING FLUID FUEL OR SOLID FUEL SUSPENDED IN A CARRIER GAS OR AIR
- F23C13/00—Apparatus in which combustion takes place in the presence of catalytic material
- F23C13/08—Apparatus in which combustion takes place in the presence of catalytic material characterised by the catalytic material
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D53/00—Separation of gases or vapours; Recovering vapours of volatile solvents from gases; Chemical or biological purification of waste gases, e.g. engine exhaust gases, smoke, fumes, flue gases, aerosols
- B01D53/34—Chemical or biological purification of waste gases
- B01D53/74—General processes for purification of waste gases; Apparatus or devices specially adapted therefor
- B01D53/86—Catalytic processes
- B01D53/8621—Removing nitrogen compounds
- B01D53/8625—Nitrogen oxides
- B01D53/8628—Processes characterised by a specific catalyst
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D53/00—Separation of gases or vapours; Recovering vapours of volatile solvents from gases; Chemical or biological purification of waste gases, e.g. engine exhaust gases, smoke, fumes, flue gases, aerosols
- B01D53/34—Chemical or biological purification of waste gases
- B01D53/74—General processes for purification of waste gases; Apparatus or devices specially adapted therefor
- B01D53/86—Catalytic processes
- B01D53/864—Removing carbon monoxide or hydrocarbons
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D53/00—Separation of gases or vapours; Recovering vapours of volatile solvents from gases; Chemical or biological purification of waste gases, e.g. engine exhaust gases, smoke, fumes, flue gases, aerosols
- B01D53/34—Chemical or biological purification of waste gases
- B01D53/74—General processes for purification of waste gases; Apparatus or devices specially adapted therefor
- B01D53/86—Catalytic processes
- B01D53/8668—Removing organic compounds not provided for in B01D53/8603 - B01D53/8665
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D53/00—Separation of gases or vapours; Recovering vapours of volatile solvents from gases; Chemical or biological purification of waste gases, e.g. engine exhaust gases, smoke, fumes, flue gases, aerosols
- B01D53/34—Chemical or biological purification of waste gases
- B01D53/74—General processes for purification of waste gases; Apparatus or devices specially adapted therefor
- B01D53/86—Catalytic processes
- B01D53/8678—Removing components of undefined structure
- B01D53/8687—Organic components
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J21/00—Catalysts comprising the elements, oxides, or hydroxides of magnesium, boron, aluminium, carbon, silicon, titanium, zirconium, or hafnium
- B01J21/18—Carbon
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J23/00—Catalysts comprising metals or metal oxides or hydroxides, not provided for in group B01J21/00
- B01J23/70—Catalysts comprising metals or metal oxides or hydroxides, not provided for in group B01J21/00 of the iron group metals or copper
- B01J23/74—Iron group metals
- B01J23/755—Nickel
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J23/00—Catalysts comprising metals or metal oxides or hydroxides, not provided for in group B01J21/00
- B01J23/70—Catalysts comprising metals or metal oxides or hydroxides, not provided for in group B01J21/00 of the iron group metals or copper
- B01J23/76—Catalysts comprising metals or metal oxides or hydroxides, not provided for in group B01J21/00 of the iron group metals or copper combined with metals, oxides or hydroxides provided for in groups B01J23/02 - B01J23/36
- B01J23/78—Catalysts comprising metals or metal oxides or hydroxides, not provided for in group B01J21/00 of the iron group metals or copper combined with metals, oxides or hydroxides provided for in groups B01J23/02 - B01J23/36 with alkali- or alkaline earth metals
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J35/00—Catalysts, in general, characterised by their form or physical properties
- B01J35/40—Catalysts, in general, characterised by their form or physical properties characterised by dimensions, e.g. grain size
- B01J35/45—Nanoparticles
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J35/00—Catalysts, in general, characterised by their form or physical properties
- B01J35/70—Catalysts, in general, characterised by their form or physical properties characterised by their crystalline properties, e.g. semi-crystalline
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J37/00—Processes, in general, for preparing catalysts; Processes, in general, for activation of catalysts
- B01J37/02—Impregnation, coating or precipitation
- B01J37/0215—Coating
- B01J37/0219—Coating the coating containing organic compounds
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J37/00—Processes, in general, for preparing catalysts; Processes, in general, for activation of catalysts
- B01J37/02—Impregnation, coating or precipitation
- B01J37/0215—Coating
- B01J37/0221—Coating of particles
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J37/00—Processes, in general, for preparing catalysts; Processes, in general, for activation of catalysts
- B01J37/02—Impregnation, coating or precipitation
- B01J37/03—Precipitation; Co-precipitation
- B01J37/031—Precipitation
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J37/00—Processes, in general, for preparing catalysts; Processes, in general, for activation of catalysts
- B01J37/02—Impregnation, coating or precipitation
- B01J37/03—Precipitation; Co-precipitation
- B01J37/031—Precipitation
- B01J37/033—Using Hydrolysis
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J37/00—Processes, in general, for preparing catalysts; Processes, in general, for activation of catalysts
- B01J37/08—Heat treatment
- B01J37/082—Decomposition and pyrolysis
- B01J37/088—Decomposition of a metal salt
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J37/00—Processes, in general, for preparing catalysts; Processes, in general, for activation of catalysts
- B01J37/08—Heat treatment
- B01J37/10—Heat treatment in the presence of water, e.g. steam
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J37/00—Processes, in general, for preparing catalysts; Processes, in general, for activation of catalysts
- B01J37/12—Oxidising
- B01J37/14—Oxidising with gases containing free oxygen
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J37/00—Processes, in general, for preparing catalysts; Processes, in general, for activation of catalysts
- B01J37/16—Reducing
- B01J37/18—Reducing with gases containing free hydrogen
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B22—CASTING; POWDER METALLURGY
- B22F—WORKING METALLIC POWDER; MANUFACTURE OF ARTICLES FROM METALLIC POWDER; MAKING METALLIC POWDER; APPARATUS OR DEVICES SPECIALLY ADAPTED FOR METALLIC POWDER
- B22F1/00—Metallic powder; Treatment of metallic powder, e.g. to facilitate working or to improve properties
- B22F1/16—Metallic particles coated with a non-metal
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B35/00—Shaped ceramic products characterised by their composition; Ceramics compositions; Processing powders of inorganic compounds preparatory to the manufacturing of ceramic products
- C04B35/01—Shaped ceramic products characterised by their composition; Ceramics compositions; Processing powders of inorganic compounds preparatory to the manufacturing of ceramic products based on oxide ceramics
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B35/00—Shaped ceramic products characterised by their composition; Ceramics compositions; Processing powders of inorganic compounds preparatory to the manufacturing of ceramic products
- C04B35/622—Forming processes; Processing powders of inorganic compounds preparatory to the manufacturing of ceramic products
- C04B35/626—Preparing or treating the powders individually or as batches ; preparing or treating macroscopic reinforcing agents for ceramic products, e.g. fibres; mechanical aspects section B
- C04B35/628—Coating the powders or the macroscopic reinforcing agents
- C04B35/62802—Powder coating materials
- C04B35/62828—Non-oxide ceramics
- C04B35/62839—Carbon
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B35/00—Shaped ceramic products characterised by their composition; Ceramics compositions; Processing powders of inorganic compounds preparatory to the manufacturing of ceramic products
- C04B35/622—Forming processes; Processing powders of inorganic compounds preparatory to the manufacturing of ceramic products
- C04B35/626—Preparing or treating the powders individually or as batches ; preparing or treating macroscopic reinforcing agents for ceramic products, e.g. fibres; mechanical aspects section B
- C04B35/628—Coating the powders or the macroscopic reinforcing agents
- C04B35/62884—Coating the powders or the macroscopic reinforcing agents by gas phase techniques
-
- F—MECHANICAL ENGINEERING; LIGHTING; HEATING; WEAPONS; BLASTING
- F23—COMBUSTION APPARATUS; COMBUSTION PROCESSES
- F23G—CREMATION FURNACES; CONSUMING WASTE PRODUCTS BY COMBUSTION
- F23G7/00—Incinerators or other apparatus for consuming industrial waste, e.g. chemicals
- F23G7/06—Incinerators or other apparatus for consuming industrial waste, e.g. chemicals of waste gases or noxious gases, e.g. exhaust gases
- F23G7/07—Incinerators or other apparatus for consuming industrial waste, e.g. chemicals of waste gases or noxious gases, e.g. exhaust gases in which combustion takes place in the presence of catalytic material
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M10/00—Secondary cells; Manufacture thereof
- H01M10/05—Accumulators with non-aqueous electrolyte
- H01M10/052—Li-accumulators
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/04—Processes of manufacture in general
- H01M4/0471—Processes of manufacture in general involving thermal treatment, e.g. firing, sintering, backing particulate active material, thermal decomposition, pyrolysis
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/362—Composites
- H01M4/366—Composites as layered products
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/48—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/48—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides
- H01M4/52—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides of nickel, cobalt or iron
- H01M4/523—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides of nickel, cobalt or iron for non-aqueous cells
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/58—Selection of substances as active materials, active masses, active liquids of inorganic compounds other than oxides or hydroxides, e.g. sulfides, selenides, tellurides, halogenides or LiCoFy; of polyanionic structures, e.g. phosphates, silicates or borates
- H01M4/583—Carbonaceous material, e.g. graphite-intercalation compounds or CFx
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D2255/00—Catalysts
- B01D2255/20—Metals or compounds thereof
- B01D2255/202—Alkali metals
- B01D2255/2022—Potassium
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D2255/00—Catalysts
- B01D2255/20—Metals or compounds thereof
- B01D2255/207—Transition metals
- B01D2255/20753—Nickel
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D2255/00—Catalysts
- B01D2255/20—Metals or compounds thereof
- B01D2255/209—Other metals
- B01D2255/2092—Aluminium
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D2255/00—Catalysts
- B01D2255/70—Non-metallic catalysts, additives or dopants
- B01D2255/702—Carbon
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D2255/00—Catalysts
- B01D2255/90—Physical characteristics of catalysts
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D2255/00—Catalysts
- B01D2255/90—Physical characteristics of catalysts
- B01D2255/92—Dimensions
- B01D2255/9202—Linear dimensions
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D2257/00—Components to be removed
- B01D2257/40—Nitrogen compounds
- B01D2257/402—Dinitrogen oxide
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D2257/00—Components to be removed
- B01D2257/40—Nitrogen compounds
- B01D2257/404—Nitrogen oxides other than dinitrogen oxide
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D2257/00—Components to be removed
- B01D2257/70—Organic compounds not provided for in groups B01D2257/00 - B01D2257/602
- B01D2257/702—Hydrocarbons
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D2257/00—Components to be removed
- B01D2257/70—Organic compounds not provided for in groups B01D2257/00 - B01D2257/602
- B01D2257/702—Hydrocarbons
- B01D2257/7022—Aliphatic hydrocarbons
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D2257/00—Components to be removed
- B01D2257/70—Organic compounds not provided for in groups B01D2257/00 - B01D2257/602
- B01D2257/708—Volatile organic compounds V.O.C.'s
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J2235/00—Indexing scheme associated with group B01J35/00, related to the analysis techniques used to determine the catalysts form or properties
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J2235/00—Indexing scheme associated with group B01J35/00, related to the analysis techniques used to determine the catalysts form or properties
- B01J2235/15—X-ray diffraction
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J2235/00—Indexing scheme associated with group B01J35/00, related to the analysis techniques used to determine the catalysts form or properties
- B01J2235/30—Scanning electron microscopy; Transmission electron microscopy
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B2235/00—Aspects relating to ceramic starting mixtures or sintered ceramic products
- C04B2235/02—Composition of constituents of the starting material or of secondary phases of the final product
- C04B2235/30—Constituents and secondary phases not being of a fibrous nature
- C04B2235/32—Metal oxides, mixed metal oxides, or oxide-forming salts thereof, e.g. carbonates, nitrates, (oxy)hydroxides, chlorides
- C04B2235/3201—Alkali metal oxides or oxide-forming salts thereof
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B2235/00—Aspects relating to ceramic starting mixtures or sintered ceramic products
- C04B2235/02—Composition of constituents of the starting material or of secondary phases of the final product
- C04B2235/30—Constituents and secondary phases not being of a fibrous nature
- C04B2235/32—Metal oxides, mixed metal oxides, or oxide-forming salts thereof, e.g. carbonates, nitrates, (oxy)hydroxides, chlorides
- C04B2235/3217—Aluminum oxide or oxide forming salts thereof, e.g. bauxite, alpha-alumina
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B2235/00—Aspects relating to ceramic starting mixtures or sintered ceramic products
- C04B2235/02—Composition of constituents of the starting material or of secondary phases of the final product
- C04B2235/30—Constituents and secondary phases not being of a fibrous nature
- C04B2235/32—Metal oxides, mixed metal oxides, or oxide-forming salts thereof, e.g. carbonates, nitrates, (oxy)hydroxides, chlorides
- C04B2235/327—Iron group oxides, their mixed metal oxides, or oxide-forming salts thereof
- C04B2235/3279—Nickel oxides, nickalates, or oxide-forming salts thereof
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B2235/00—Aspects relating to ceramic starting mixtures or sintered ceramic products
- C04B2235/02—Composition of constituents of the starting material or of secondary phases of the final product
- C04B2235/50—Constituents or additives of the starting mixture chosen for their shape or used because of their shape or their physical appearance
- C04B2235/54—Particle size related information
- C04B2235/5418—Particle size related information expressed by the size of the particles or aggregates thereof
- C04B2235/5454—Particle size related information expressed by the size of the particles or aggregates thereof nanometer sized, i.e. below 100 nm
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B2235/00—Aspects relating to ceramic starting mixtures or sintered ceramic products
- C04B2235/65—Aspects relating to heat treatments of ceramic bodies such as green ceramics or pre-sintered ceramics, e.g. burning, sintering or melting processes
- C04B2235/656—Aspects relating to heat treatments of ceramic bodies such as green ceramics or pre-sintered ceramics, e.g. burning, sintering or melting processes characterised by specific heating conditions during heat treatment
- C04B2235/6562—Heating rate
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B2235/00—Aspects relating to ceramic starting mixtures or sintered ceramic products
- C04B2235/65—Aspects relating to heat treatments of ceramic bodies such as green ceramics or pre-sintered ceramics, e.g. burning, sintering or melting processes
- C04B2235/656—Aspects relating to heat treatments of ceramic bodies such as green ceramics or pre-sintered ceramics, e.g. burning, sintering or melting processes characterised by specific heating conditions during heat treatment
- C04B2235/6567—Treatment time
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B2235/00—Aspects relating to ceramic starting mixtures or sintered ceramic products
- C04B2235/70—Aspects relating to sintered or melt-casted ceramic products
- C04B2235/74—Physical characteristics
- C04B2235/76—Crystal structural characteristics, e.g. symmetry
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B2235/00—Aspects relating to ceramic starting mixtures or sintered ceramic products
- C04B2235/70—Aspects relating to sintered or melt-casted ceramic products
- C04B2235/80—Phases present in the sintered or melt-cast ceramic products other than the main phase
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B2235/00—Aspects relating to ceramic starting mixtures or sintered ceramic products
- C04B2235/70—Aspects relating to sintered or melt-casted ceramic products
- C04B2235/95—Products characterised by their size, e.g. microceramics
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M2004/026—Electrodes composed of, or comprising, active material characterised by the polarity
- H01M2004/027—Negative electrodes
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02C—CAPTURE, STORAGE, SEQUESTRATION OR DISPOSAL OF GREENHOUSE GASES [GHG]
- Y02C20/00—Capture or disposal of greenhouse gases
- Y02C20/10—Capture or disposal of greenhouse gases of nitrous oxide (N2O)
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02C—CAPTURE, STORAGE, SEQUESTRATION OR DISPOSAL OF GREENHOUSE GASES [GHG]
- Y02C20/00—Capture or disposal of greenhouse gases
- Y02C20/20—Capture or disposal of greenhouse gases of methane
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02P—CLIMATE CHANGE MITIGATION TECHNOLOGIES IN THE PRODUCTION OR PROCESSING OF GOODS
- Y02P20/00—Technologies relating to chemical industry
- Y02P20/30—Improvements relating to adipic acid or caprolactam production
Landscapes
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Materials Engineering (AREA)
- Organic Chemistry (AREA)
- Environmental & Geological Engineering (AREA)
- General Chemical & Material Sciences (AREA)
- Manufacturing & Machinery (AREA)
- Health & Medical Sciences (AREA)
- Biomedical Technology (AREA)
- Analytical Chemistry (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Ceramic Engineering (AREA)
- Electrochemistry (AREA)
- Inorganic Chemistry (AREA)
- Mechanical Engineering (AREA)
- General Engineering & Computer Science (AREA)
- Structural Engineering (AREA)
- Thermal Sciences (AREA)
- Physics & Mathematics (AREA)
- Combustion & Propulsion (AREA)
- Composite Materials (AREA)
- Catalysts (AREA)
- Exhaust Gas Treatment By Means Of Catalyst (AREA)
- Inorganic Compounds Of Heavy Metals (AREA)
- Dispersion Chemistry (AREA)
Description
本発明は触媒の技術分野に関し、特に、炭素被覆ニッケル-アルミニウムナノ複合材料、その調製方法および使用、例えば揮発性有機化合物を触媒燃焼させる方法に関する。
遷移金属酸化物は優れた触媒性能および電磁性能を有し、無機材料の分野における研究のホットスポットであり、エネルギー貯蔵材料、触媒材料、磁気記録材料および生物医学において広く使用されている。近年、炭素被覆ナノ材料は、電気触媒、スーパーキャパシタ材料、リチウムイオン電池アノード材料、バイオエンジニアリングなどの分野で広く使用されている。
本発明の主な目的は、先行技術の上述の欠点の少なくとも1つを克服することおよび炭素被覆ニッケル-アルミニウムナノ複合材料を提供することである。当該ナノ複合材料は、黒鉛化炭素シェルと酸化ニッケルおよびアルミナの内側コアとを含むコア-シェル構造を有し、このナノ複合材料は触媒としての活性に優れ、亜酸化窒素の分解を有効に触媒することができ、アジピン酸プラント、硝酸プラント等の製造プロセスで発生する高濃度のN2O排気ガスの除去の課題を解決するために役立つ。ナノ複合材料はまた、揮発性有機化合物の触媒燃焼の方法のための触媒として使用することができ、低温でのVOCの酸化および燃焼を高効率で触媒することができ、VOCの浄化の課題を解決するために有益である。
本発明の第1の態様は、炭素被覆ニッケル-アルミニウムナノ複合材料であって、シェルとしての外側膜および内側コアを有するコア-シェル構造を含み、前記外側膜が黒鉛化炭素膜であり、前記内側コアが酸化ニッケルおよびアルミナを含み、前記ナノ複合材料の総重量に基づいて、酸化ニッケル含有量が59%~80%、アルミナ含有量が19%~40%、炭素含有量が1%以下である、炭素被覆ニッケル-アルミニウムナノ複合材料を提供する。
本発明により提供される炭素被覆ニッケル-アルミニウムナノ複合材料は、黒鉛化炭素膜のシェルと酸化ニッケルおよびアルミナのコアとを有するコア-シェル構造を含み、独特の構造および組成によりN2Oの分解反応を触媒するための触媒として使用される場合に優れた活性を有し、より低温での工業的製造において生成される高濃度の亜酸化窒素排ガスを触媒および分解するために使用することができ、ここで分解速度は99%を超えることができ、前記炭素被覆ニッケル-アルミニウムナノ複合材料は、工業的排ガス中のN2Oを希釈し、次いで処理する必要がある従来の触媒と比較して、環境を保護し、大気汚染を低減するために重要な意義を有し、良好な工業的用途の見通しを有する。
本発明により提供される揮発性有機化合物の触媒燃焼方法は優れた触媒活性を有し、低温でのVOCの酸化燃焼を高効率で触媒することができ、VOCの浄化の問題を解決するのに有益であり、大気汚染を低減し、良好な工業的用途の見通しを有する。
図面は本発明をさらに理解するために提供され、以下の実施形態と共に、本発明を限定せずに本発明の原理を説明するのに役立ち、本明細書の一部を構成する。
図2aおよび図2bは、種々の倍率における実施例1のナノ複合材料のTEM像である;
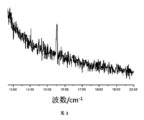
図3は、実施例1のナノ複合材料のラマンスペクトルである;
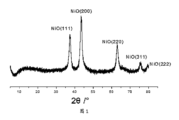
図4は、実施例2のナノ複合材料のX線回折パターンである;
図5aおよび図5bは、種々の倍率での実施例2のナノ複合材料のTEM像である;
図6は、実施例2のナノ複合材料のラマンスペクトルである。
以下の説明は、当業者が本明細書の説明を参照して本発明を実施することを可能にするために、様々な実施形態または実施例を提供する。もちろん、これらは単なる例であり、限定を意図するものではない。本願に開示される範囲の終点および任意の値は、正確な範囲または値に限定されず、これらの範囲または値に近い値を包含すると理解されるべきである。数値範囲についてはそれぞれの範囲の終点間、それぞれの範囲の終点と個々の値との間、および個々の値の間の値の組み合わせは1つ以上の新しい数値範囲をもたらすことができ、そのような新しい数値範囲は本明細書で具体的に開示されているとみなされるべきである。
以下の実施例によって本発明をさらに説明するが、それに限定されるものではない。特に明記しない限り、本発明で使用される全ての試薬は分析的に純粋である。
この実施例は、本発明に係る炭素被覆ニッケル-アルミニウムナノ複合材料の調製を例示した。
図1は実施例1のナノ複合材料のX線回折(XRD)スペクトルであり、図1から、ナノ複合材料中のニッケルは、穏やかな酸化処理後に酸化物として存在することが分かった。図2aおよび図2bはそれぞれ異なった倍率での実施例1のナノ複合材料の透過型電子顕微鏡写真(TEM)であり、そこから、炭素層膜が材料の表面上に形成されており、粒径が約5~20nmであることが観察された。
この実施例は、本発明による炭素被覆ニッケル-アルミニウムナノ複合材料の調製を例示した。
図4は実施例2のナノ複合材料のX線回析(XRD)スペクトルであり、図4から、ナノ複合材料中のニッケルは、穏やかな酸化処理後に酸化物として存在することが分かった。図5aおよび図5bはそれぞれ異なった倍率での実施例2のナノ複合材料の透過型電子顕微鏡写真であり、そこから、炭素層膜が物質の表面上に形成されており、粒径が約5~20nmであることが観察された。
実施例1の工程(1)で得られたニッケル-アルミニウム前駆体を秤量し、セラミックボートに入れ、次いで当該セラミックボートを窒素保護雰囲気下、管状炉に入れ、100mL/分の窒素流量、5℃/分の温度プログラムで、500℃に加熱した後、30mL/分の水素を180分間導入し、水素を閉鎖し、水素雰囲気下で自然に冷却し、中間生成物を得た。
実施例1の工程(1)で得られたニッケル-アルミニウム前駆体を秤量し、セラミックボートに入れ、次いで当該セラミックボートを窒素保護雰囲気下、管状炉に入れ、100mL/分の窒素流量、5℃/分の温度プログラムで、500℃に加熱した後、30mL/分の水素を240分間導入し、水素を閉鎖し、水素雰囲気下で自然に冷却し、中間生成物を得た。
この応用例は、触媒として実施例1のナノ複合材料を使用して亜酸化窒素の分解を触媒する反応を例示した。
実施例2のナノ複合材料を触媒として用いたこと以外は応用例1に従った方法を用いて分解を行い、その結果を表1に示した。
比較例1の材料を触媒として用いたこと以外は、応用例1の方法を用いて分解反応を行い、その結果を表1に示した。
比較例2の材料を触媒として用いたこと以外は、応用例1の方法を用いて分解反応を行い、その結果を表1に示した。
この応用例は、触媒として実施例1のナノ複合材料を使用するVOCの触媒燃焼を例示した。
実施例2のナノ複合材料を触媒として用いたこと以外は、実施例3に係る方法を用いてVOCの触媒燃焼反応を行い、その結果を表2に示した。
比較例1の材料を触媒として用いたこと以外は、応用例3の方法により分解反応を行い、その結果を表2に示した。
比較例2の材料を触媒として用いたこと以外は、応用例3の方法により分解反応を行い、その結果を表2に示した。
Claims (24)
- 炭素被覆ニッケル-アルミニウムナノ複合材料であって、外側シェルおよび内側コアを有するコア-シェル構造を含み、前記外側シェルは黒鉛化炭素膜であり、前記内側コアは酸化ニッケルおよびアルミナを含み、前記ナノ複合材料の総重量に基づいて、酸化ニッケル含有量が69%~79%、アルミナ含有量が20%~30%、炭素含有量が1%以下であることを特徴とする、炭素被覆ニッケル-アルミニウムナノ複合材料。
- 前記酸化ニッケル含有量が69%~79%であり、前記アルミナ含有量が20%~30%であり、前記炭素含有量が0.3%~1%であることを特徴とする、請求項1に記載のナノ複合材料。
- 前記ナノ複合材料において、元素分析により決定された炭素元素含有量に対するX線光電子分光法により決定された炭素元素含有量の重量比が10以上であることを特徴とする、請求項1に記載のナノ複合材料。
- 1320cm-1付近に位置するDピークの強度に対する1580cm-1付近に位置するGピークの強度の比率が2より大きいラマンスペクトルを有することを特徴とする、請求項1に記載のナノ複合材料。
- 前記コアが、ナノ複合材料の重量に対して5重量%以下の量のアルカリ金属酸化物をさらに含むことを特徴とする、請求項1に記載のナノ複合材料。
- 前記ナノ複合材料が1%以下、0.5%以下、0.1%以下、または0.01%以下の量のニッケル元素を含むことを特徴とする、請求項1に記載のナノ複合材料。
- ニッケル-アルミニウム前駆体を調製する工程と、
前記ニッケル-アルミニウム前駆体に対して昇温熱処理を行い、炭素源ガスとして低級アルカンを用いることにより蒸着を行う工程と、
蒸着後に得られた生成物に対して酸素処理を行い、ナノ複合材料を得る工程と、を含むことを特徴とする、請求項1~6のいずれか1項に記載の炭素被覆ニッケル-アルミニウムナノ複合材料を調製する方法。 - 前記酸素処理は、前記蒸着後に得られた生成物に標準ガスを導入して加熱することを含み、前記標準ガスは、酸素とバランスガスとを含み、10~40体積%の酸素濃度を有することを特徴とする、請求項7に記載の方法。
- 前記酸素処理は、200℃~500℃の温度で0.5時間~10時間行われることを特徴とする、請求項7に記載の方法。
- 前記ニッケル-アルミニウム前駆体が、共沈および/または水熱結晶化の様式によって調製されることを特徴とする、請求項7に記載の方法。
- 前記ニッケル-アルミニウム前駆体は、アルカリ溶液と、3価のアルミニウム塩および2価のニッケル塩を含む水溶液とを同時に水中に滴下して沈殿処理し、その結果、3価のアルミニウム塩と2価のニッケル塩とが共沈物を生成することと、前記共沈物を熟成させることと、を含む工程によって調製されることを特徴とする、請求項7に記載の方法。
- 前記3価のアルミニウム塩は硝酸アルミニウムおよび/または塩化アルミニウムを含み、前記2価のニッケル塩は硝酸ニッケルおよび/または塩化ニッケルを含み、前記3価のアルミニウム塩中のアルミニウムと前記2価のニッケル塩中のニッケルとのモル比は1:(2~4)であり;
前記アルカリ溶液は水酸化ナトリウムおよび炭酸ナトリウムを含む水溶液であり、水酸化ナトリウムアルカリ溶液の濃度は0.2~4mol/Lであり、アルカリ溶液中の炭酸ナトリウムの濃度は0.1~2mol/Lであり;
水酸化ナトリウムと前記3価のアルミニウム塩および前記2価のニッケル塩中のアルミニウムおよびニッケルの総モルとのモル比は(2~4):1であり、炭酸ナトリウムと前記3価のアルミニウム塩および前記2価のニッケル塩中のアルミニウムおよびニッケルの総モルとのモル比は(0.5~2):1であることを特徴とする、請求項11に記載の方法。 - 沈殿処理は40℃から100℃未満の温度で行われ、熟成処理は40℃から100℃未満の温度で2~48時間行われることを特徴とする、請求項11に記載の方法。
- 昇温熱処理を受けた前記ニッケル-アルミニウム前駆体を水素と接触させて、500~900℃の処理で120~480分間、30~50ml/(分 gニッケル-アルミニウム前駆体)の水素流量で還元処理を実施することをさらに含む、請求項7に記載の方法。
- 前記ニッケル-アルミニウム前駆体とアルカリ金属の塩溶液とを混合して共沈反応させ、次いで得られた沈殿物に対して昇温熱処理を行うことをさらに含み、ニッケルに対する前記アルカリ金属のモル比が0.2以下である、請求項7に記載の方法。
- 前記昇温熱処理は保護ガスの存在下で前記ニッケル-アルミニウム前駆体の温度を500~900℃に上昇させることを含み、前記保護ガスは窒素および/またはアルゴンであり、前記保護ガスの流量は10~500ml/(分・gニッケル-アルミニウム前駆体)であり、昇温速度は1~5℃/分であり;
前記蒸着は750~900℃で5~240分間行われ;
前記炭素源ガスはメタンまたはエタンであり、これは10~500ml/(分・gニッケル-アルミニウム前駆体)の流量で使用されることを特徴とする、請求項7に記載の方法。 - 触媒材料、エネルギー貯蔵材料または電磁材料としての、請求項1~6のいずれか1項に記載のナノ複合材料の使用。
- 触媒を亜酸化窒素と接触させて触媒分解反応させ、窒素および酸素を生成することを含む、亜酸化窒素の分解のための触媒としての、請求項1~6のいずれか1項に記載のナノ複合材料の使用。
- 揮発性有機化合物の酸化反応を触媒するための触媒として、請求項1~6のいずれか1項に記載のナノ複合材料を使用することを含む、揮発性有機化合物を触媒燃焼させる方法。
- 前記酸化反応は、揮発性有機化合物と、酸素を含む標準ガスとを含む混合ガスを触媒と接触させることによる、前記混合ガスの触媒燃焼を含み、前記標準ガスは、酸素とバランスガスとを含み、10~40体積%の酸素濃度を有することを特徴とする、請求項19に記載の方法。
- 前記混合ガスが、0.01~2体積%の揮発性有機化合物と、5~20体積%の酸素とを含むことを特徴とする、請求項20に記載の方法。
- 揮発性有機化合物が、1~4個の炭素原子を有する炭化水素からなる群から選択される1種以上であることを特徴とする、請求項19に記載の方法。
- 前記酸化反応は、1000~5000mlの反応ガス/(時間・g触媒)の空間速度で行われることを特徴とする、請求項19に記載の方法。
- 前記酸化反応は、300℃~450℃の温度で行われることを特徴とする、請求項19に記載の方法。
Applications Claiming Priority (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201911001564.6A CN112755994A (zh) | 2019-10-21 | 2019-10-21 | 碳包覆镍铝的纳米复合材料及制备方法和应用 |
| CN201911001557.6 | 2019-10-21 | ||
| CN201911001564.6 | 2019-10-21 | ||
| CN201911001557.6A CN112762468B (zh) | 2019-10-21 | 2019-10-21 | 催化燃烧挥发性有机化合物的方法 |
| PCT/CN2020/122099 WO2021078113A1 (zh) | 2019-10-21 | 2020-10-20 | 碳包覆镍铝的纳米复合材料及制备方法和应用 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2022553343A JP2022553343A (ja) | 2022-12-22 |
| JP7581347B2 true JP7581347B2 (ja) | 2024-11-12 |
Family
ID=75619637
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2022523622A Active JP7581347B2 (ja) | 2019-10-21 | 2020-10-20 | 炭素被覆ニッケル-アルミニウムナノ複合材料、その調製方法およびその適用 |
Country Status (5)
| Country | Link |
|---|---|
| US (1) | US12467622B2 (ja) |
| EP (1) | EP4049752A4 (ja) |
| JP (1) | JP7581347B2 (ja) |
| TW (1) | TWI870486B (ja) |
| WO (1) | WO2021078113A1 (ja) |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN116791069B (zh) * | 2023-06-26 | 2025-12-30 | 四川轻化工大学 | 一种镍包石墨复合颗粒及其制备方法 |
Citations (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20040065619A1 (en) | 2002-10-04 | 2004-04-08 | Klabunde Kenneth J. | Carbon-coated metal oxide nanoparticles |
| JP2004230317A (ja) | 2003-01-31 | 2004-08-19 | Nippon Oil Corp | 炭化水素の脱硫触媒、脱硫方法及び燃料電池システム |
| JP2007152263A (ja) | 2005-12-07 | 2007-06-21 | Nippon Shokubai Co Ltd | 亜酸化窒素分解用触媒および亜酸化窒素含有ガスの浄化方法 |
| CN101811052A (zh) | 2010-04-16 | 2010-08-25 | 济南大学 | 一种以类水滑石为前驱物制备的柴油车碳烟燃烧催化剂及其制备方法 |
| JP2011513167A (ja) | 2008-02-28 | 2011-04-28 | ビーエーエスエフ ソシエタス・ヨーロピア | 黒鉛のナノプレートレットおよび組成物 |
| CN104815983A (zh) | 2015-04-20 | 2015-08-05 | 齐鲁工业大学 | 一种碳包覆氧化镍/金属镍及其简单合成方法 |
| JP2015536235A (ja) | 2012-11-16 | 2015-12-21 | ユニヴァーシティ・オブ・バス | 鉄及びカーボンナノチューブを含む改良された触媒 |
| CN106872545A (zh) | 2017-02-06 | 2017-06-20 | 内蒙古科技大学 | 一种蛛网状复合材料、及其制备方法和在生物传感器方面的应用 |
| CN107469824A (zh) | 2017-09-26 | 2017-12-15 | 天津工业大学 | 一种碳包覆的中空晶体氧化镍的乙醇氧化催化剂的制备方法 |
| CN108212035A (zh) | 2017-12-15 | 2018-06-29 | 成都市银隆新能源有限公司 | 一种多孔核壳纳米复合材料及其制备方法 |
| CN108856706A (zh) | 2017-05-10 | 2018-11-23 | 中国石油化工股份有限公司 | 一种碳包覆镍铝复合材料及其制备方法和应用 |
| WO2019020086A1 (zh) | 2017-07-28 | 2019-01-31 | 中国石油化工股份有限公司 | 碳包覆过渡金属纳米复合材料、其制备及应用 |
Family Cites Families (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4191664A (en) * | 1975-06-16 | 1980-03-04 | Union Oil Company Of California | Thermally stable nickel-alumina catalysts useful for methanation and other reactions |
| US10427982B2 (en) * | 2015-09-17 | 2019-10-01 | Korea Institute Of Energy Research | Method of carbon coating on nanoparticle and carbon coated nanoparticle produced by the same |
-
2020
- 2020-10-20 WO PCT/CN2020/122099 patent/WO2021078113A1/zh not_active Ceased
- 2020-10-20 US US17/755,112 patent/US12467622B2/en active Active
- 2020-10-20 EP EP20880137.3A patent/EP4049752A4/en active Pending
- 2020-10-20 JP JP2022523622A patent/JP7581347B2/ja active Active
- 2020-10-21 TW TW109136552A patent/TWI870486B/zh active
Patent Citations (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20040065619A1 (en) | 2002-10-04 | 2004-04-08 | Klabunde Kenneth J. | Carbon-coated metal oxide nanoparticles |
| JP2004230317A (ja) | 2003-01-31 | 2004-08-19 | Nippon Oil Corp | 炭化水素の脱硫触媒、脱硫方法及び燃料電池システム |
| JP2007152263A (ja) | 2005-12-07 | 2007-06-21 | Nippon Shokubai Co Ltd | 亜酸化窒素分解用触媒および亜酸化窒素含有ガスの浄化方法 |
| JP2011513167A (ja) | 2008-02-28 | 2011-04-28 | ビーエーエスエフ ソシエタス・ヨーロピア | 黒鉛のナノプレートレットおよび組成物 |
| CN101811052A (zh) | 2010-04-16 | 2010-08-25 | 济南大学 | 一种以类水滑石为前驱物制备的柴油车碳烟燃烧催化剂及其制备方法 |
| JP2015536235A (ja) | 2012-11-16 | 2015-12-21 | ユニヴァーシティ・オブ・バス | 鉄及びカーボンナノチューブを含む改良された触媒 |
| CN104815983A (zh) | 2015-04-20 | 2015-08-05 | 齐鲁工业大学 | 一种碳包覆氧化镍/金属镍及其简单合成方法 |
| CN106872545A (zh) | 2017-02-06 | 2017-06-20 | 内蒙古科技大学 | 一种蛛网状复合材料、及其制备方法和在生物传感器方面的应用 |
| CN108856706A (zh) | 2017-05-10 | 2018-11-23 | 中国石油化工股份有限公司 | 一种碳包覆镍铝复合材料及其制备方法和应用 |
| WO2019020086A1 (zh) | 2017-07-28 | 2019-01-31 | 中国石油化工股份有限公司 | 碳包覆过渡金属纳米复合材料、其制备及应用 |
| CN107469824A (zh) | 2017-09-26 | 2017-12-15 | 天津工业大学 | 一种碳包覆的中空晶体氧化镍的乙醇氧化催化剂的制备方法 |
| CN108212035A (zh) | 2017-12-15 | 2018-06-29 | 成都市银隆新能源有限公司 | 一种多孔核壳纳米复合材料及其制备方法 |
Also Published As
| Publication number | Publication date |
|---|---|
| US12467622B2 (en) | 2025-11-11 |
| TW202116414A (zh) | 2021-05-01 |
| US20220370989A1 (en) | 2022-11-24 |
| WO2021078113A1 (zh) | 2021-04-29 |
| TWI870486B (zh) | 2025-01-21 |
| EP4049752A1 (en) | 2022-08-31 |
| EP4049752A4 (en) | 2023-11-29 |
| JP2022553343A (ja) | 2022-12-22 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP7752108B2 (ja) | 炭素被覆酸化ニッケルナノ複合材料、その製造方法および応用 | |
| CN108856706B (zh) | 一种碳包覆镍铝复合材料及其制备方法和应用 | |
| Zhao et al. | Unveiling a remarkable enhancement role by designing a confined structure Ho-TNTs@ Mn catalyst for low-temperature NH 3-SCR reaction | |
| Chen et al. | Advances in photochemical deposition for controllable synthesis of heterogeneous catalysts | |
| Cui et al. | Oxidation of toluene over the Pt-embedded mesoporous CeO2 hollow nanospheres with advanced catalytic performances | |
| CN109926060A (zh) | 一种核-壳结构纳米铜铈复合氧化物催化剂、制备方法与应用 | |
| Cui et al. | Oxidation of toluene over mesoporous MnO2/CeO2 nanosphere catalysts: effects of the MnO2 precursor and the type of support | |
| CN113750991B (zh) | 碳包覆氧化镍的催化剂及其制备方法和应用 | |
| JP7581347B2 (ja) | 炭素被覆ニッケル-アルミニウムナノ複合材料、その調製方法およびその適用 | |
| CN113751007B (zh) | 碳包覆氧化镍的催化剂及其制备方法和应用 | |
| CN113751006B (zh) | 碳包覆氧化镍的纳米复合材料及其制备方法和应用 | |
| CN113751042B (zh) | 碳包覆氧化镍的纳米复合材料及其制备方法和应用 | |
| CN113751004B (zh) | 碳包覆过渡金属氧化物的催化剂及其制备方法和应用 | |
| CN112755993B (zh) | 碳包覆氧化镍的纳米复合材料及其制备方法和应用 | |
| CN113751041B (zh) | 碳包覆氧化镍的纳米复合材料及其制备方法和应用 | |
| CN112755994A (zh) | 碳包覆镍铝的纳米复合材料及制备方法和应用 | |
| CN113750781B (zh) | 催化一氧化二氮分解的方法 | |
| CN113751005B (zh) | 碳包覆过渡金属氧化物的催化剂及其制备方法和应用 | |
| CN113757696A (zh) | 催化燃烧挥发性有机化合物的方法 | |
| CN112762468B (zh) | 催化燃烧挥发性有机化合物的方法 | |
| CN113751008B (zh) | 碳包覆氧化镍的纳米复合材料及其制备方法和应用 | |
| CN113751003B (zh) | 碳包覆氧化镍的纳米复合材料及其制备方法和应用 | |
| CN112762469A (zh) | 催化燃烧挥发性有机化合物的方法 | |
| CN112755995A (zh) | 碳包覆镍铝的纳米复合材料及其制备方法和应用 | |
| Wang et al. | Preparation and characterization of mesoporous TiO2-sphere-supported Au-nanoparticle catalysts with high activity for CO oxidation at ambient temperature |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20230724 |
|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20240131 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20240206 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20240411 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20240611 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20240828 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20241008 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20241030 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 7581347 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 |