JP7181880B2 - 免疫療法のためのコア/シェル構造プラットホーム - Google Patents
免疫療法のためのコア/シェル構造プラットホーム Download PDFInfo
- Publication number
- JP7181880B2 JP7181880B2 JP2019540384A JP2019540384A JP7181880B2 JP 7181880 B2 JP7181880 B2 JP 7181880B2 JP 2019540384 A JP2019540384 A JP 2019540384A JP 2019540384 A JP2019540384 A JP 2019540384A JP 7181880 B2 JP7181880 B2 JP 7181880B2
- Authority
- JP
- Japan
- Prior art keywords
- cells
- mrna
- composition
- cancer
- antigen
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 238000009169 immunotherapy Methods 0.000 title description 8
- 239000000203 mixture Substances 0.000 claims description 196
- 108020004999 messenger RNA Proteins 0.000 claims description 191
- 210000004027 cell Anatomy 0.000 claims description 143
- 206010028980 Neoplasm Diseases 0.000 claims description 136
- 108090000765 processed proteins & peptides Proteins 0.000 claims description 82
- 108090000623 proteins and genes Proteins 0.000 claims description 78
- 108091007433 antigens Proteins 0.000 claims description 67
- 102000036639 antigens Human genes 0.000 claims description 67
- 239000000427 antigen Substances 0.000 claims description 66
- 210000004443 dendritic cell Anatomy 0.000 claims description 63
- 102000004196 processed proteins & peptides Human genes 0.000 claims description 62
- 201000011510 cancer Diseases 0.000 claims description 61
- 102000004169 proteins and genes Human genes 0.000 claims description 60
- 238000011282 treatment Methods 0.000 claims description 54
- 241000282414 Homo sapiens Species 0.000 claims description 46
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 claims description 45
- 229920001184 polypeptide Polymers 0.000 claims description 44
- 241001465754 Metazoa Species 0.000 claims description 36
- 208000024891 symptom Diseases 0.000 claims description 34
- 241000124008 Mammalia Species 0.000 claims description 33
- 239000003814 drug Substances 0.000 claims description 31
- 210000000612 antigen-presenting cell Anatomy 0.000 claims description 29
- 230000000890 antigenic effect Effects 0.000 claims description 27
- 201000010099 disease Diseases 0.000 claims description 26
- 210000004881 tumor cell Anatomy 0.000 claims description 26
- 108020004414 DNA Proteins 0.000 claims description 24
- 150000001875 compounds Chemical class 0.000 claims description 24
- 239000002245 particle Substances 0.000 claims description 24
- 102000007327 Protamines Human genes 0.000 claims description 23
- 108010007568 Protamines Proteins 0.000 claims description 23
- 239000000232 Lipid Bilayer Substances 0.000 claims description 22
- 229940048914 protamine Drugs 0.000 claims description 22
- GSHCNPAEDNETGJ-HKOLQMFGSA-N 2-[2,3-bis[[(z)-octadec-9-enoyl]oxy]propoxy-ethoxyphosphoryl]oxyethyl-trimethylazanium Chemical compound CCCCCCCC\C=C/CCCCCCCC(=O)OCC(COP(=O)(OCC)OCC[N+](C)(C)C)OC(=O)CCCCCCC\C=C/CCCCCCCC GSHCNPAEDNETGJ-HKOLQMFGSA-N 0.000 claims description 20
- 229920000642 polymer Polymers 0.000 claims description 20
- 239000003795 chemical substances by application Substances 0.000 claims description 19
- 208000035475 disorder Diseases 0.000 claims description 19
- 108091032973 (ribonucleotides)n+m Proteins 0.000 claims description 18
- -1 antibodies Proteins 0.000 claims description 17
- 239000002246 antineoplastic agent Substances 0.000 claims description 17
- 229940127089 cytotoxic agent Drugs 0.000 claims description 17
- 229940124597 therapeutic agent Drugs 0.000 claims description 17
- 230000001225 therapeutic effect Effects 0.000 claims description 16
- 210000004962 mammalian cell Anatomy 0.000 claims description 15
- 150000003904 phospholipids Chemical class 0.000 claims description 15
- 229940022511 therapeutic cancer vaccine Drugs 0.000 claims description 15
- 241000282412 Homo Species 0.000 claims description 14
- 210000002540 macrophage Anatomy 0.000 claims description 14
- 210000003719 b-lymphocyte Anatomy 0.000 claims description 13
- 239000003112 inhibitor Substances 0.000 claims description 13
- 206010061289 metastatic neoplasm Diseases 0.000 claims description 13
- 108090000467 Interferon-beta Proteins 0.000 claims description 12
- 239000002671 adjuvant Substances 0.000 claims description 12
- 239000007771 core particle Substances 0.000 claims description 11
- 230000004064 dysfunction Effects 0.000 claims description 11
- 239000012634 fragment Substances 0.000 claims description 11
- 230000002159 abnormal effect Effects 0.000 claims description 10
- 108020004201 indoleamine 2,3-dioxygenase Proteins 0.000 claims description 10
- 102000006639 indoleamine 2,3-dioxygenase Human genes 0.000 claims description 10
- 229920001223 polyethylene glycol Polymers 0.000 claims description 10
- 102000004190 Enzymes Human genes 0.000 claims description 9
- 108090000790 Enzymes Proteins 0.000 claims description 9
- 102100026720 Interferon beta Human genes 0.000 claims description 9
- 230000002519 immonomodulatory effect Effects 0.000 claims description 9
- 239000002502 liposome Substances 0.000 claims description 9
- 241000894007 species Species 0.000 claims description 9
- LKKMLIBUAXYLOY-UHFFFAOYSA-N 3-Amino-1-methyl-5H-pyrido[4,3-b]indole Chemical compound N1C2=CC=CC=C2C2=C1C=C(N)N=C2C LKKMLIBUAXYLOY-UHFFFAOYSA-N 0.000 claims description 8
- AOJJSUZBOXZQNB-TZSSRYMLSA-N Doxorubicin Chemical compound O([C@H]1C[C@@](O)(CC=2C(O)=C3C(=O)C=4C=CC=C(C=4C(=O)C3=C(O)C=21)OC)C(=O)CO)[C@H]1C[C@H](N)[C@H](O)[C@H](C)O1 AOJJSUZBOXZQNB-TZSSRYMLSA-N 0.000 claims description 8
- 102100031413 L-dopachrome tautomerase Human genes 0.000 claims description 8
- 101710093778 L-dopachrome tautomerase Proteins 0.000 claims description 8
- 239000002202 Polyethylene glycol Substances 0.000 claims description 8
- 239000000872 buffer Substances 0.000 claims description 8
- MWRBNPKJOOWZPW-CLFAGFIQSA-N dioleoyl phosphatidylethanolamine Chemical compound CCCCCCCC\C=C/CCCCCCCC(=O)OCC(COP(O)(=O)OCCN)OC(=O)CCCCCCC\C=C/CCCCCCCC MWRBNPKJOOWZPW-CLFAGFIQSA-N 0.000 claims description 8
- 238000001990 intravenous administration Methods 0.000 claims description 8
- 230000034701 macropinocytosis Effects 0.000 claims description 8
- 239000000546 pharmaceutical excipient Substances 0.000 claims description 8
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 claims description 8
- 239000003085 diluting agent Substances 0.000 claims description 7
- 239000003937 drug carrier Substances 0.000 claims description 7
- 230000001394 metastastic effect Effects 0.000 claims description 7
- 239000002105 nanoparticle Substances 0.000 claims description 7
- 229940088597 hormone Drugs 0.000 claims description 6
- 239000005556 hormone Substances 0.000 claims description 6
- 239000003981 vehicle Substances 0.000 claims description 6
- 102000053642 Catalytic RNA Human genes 0.000 claims description 5
- 108090000994 Catalytic RNA Proteins 0.000 claims description 5
- 102000002227 Interferon Type I Human genes 0.000 claims description 5
- 108010014726 Interferon Type I Proteins 0.000 claims description 5
- 206010027480 Metastatic malignant melanoma Diseases 0.000 claims description 5
- 108020004459 Small interfering RNA Proteins 0.000 claims description 5
- 239000002254 cytotoxic agent Substances 0.000 claims description 5
- 231100000599 cytotoxic agent Toxicity 0.000 claims description 5
- 108700014844 flt3 ligand Proteins 0.000 claims description 5
- 238000004519 manufacturing process Methods 0.000 claims description 5
- 208000037819 metastatic cancer Diseases 0.000 claims description 5
- 208000011575 metastatic malignant neoplasm Diseases 0.000 claims description 5
- 208000021039 metastatic melanoma Diseases 0.000 claims description 5
- 230000002265 prevention Effects 0.000 claims description 5
- 108091092562 ribozyme Proteins 0.000 claims description 5
- 206010055113 Breast cancer metastatic Diseases 0.000 claims description 4
- 229930012538 Paclitaxel Natural products 0.000 claims description 4
- JXLYSJRDGCGARV-WWYNWVTFSA-N Vinblastine Natural products O=C(O[C@H]1[C@](O)(C(=O)OC)[C@@H]2N(C)c3c(cc(c(OC)c3)[C@]3(C(=O)OC)c4[nH]c5c(c4CCN4C[C@](O)(CC)C[C@H](C3)C4)cccc5)[C@@]32[C@H]2[C@@]1(CC)C=CCN2CC3)C JXLYSJRDGCGARV-WWYNWVTFSA-N 0.000 claims description 4
- 230000000692 anti-sense effect Effects 0.000 claims description 4
- 229960004679 doxorubicin Drugs 0.000 claims description 4
- 229960001592 paclitaxel Drugs 0.000 claims description 4
- 238000007910 systemic administration Methods 0.000 claims description 4
- RCINICONZNJXQF-MZXODVADSA-N taxol Chemical compound O([C@@H]1[C@@]2(C[C@@H](C(C)=C(C2(C)C)[C@H](C([C@]2(C)[C@@H](O)C[C@H]3OC[C@]3([C@H]21)OC(C)=O)=O)OC(=O)C)OC(=O)[C@H](O)[C@@H](NC(=O)C=1C=CC=CC=1)C=1C=CC=CC=1)O)C(=O)C1=CC=CC=C1 RCINICONZNJXQF-MZXODVADSA-N 0.000 claims description 4
- 229960003048 vinblastine Drugs 0.000 claims description 4
- JXLYSJRDGCGARV-XQKSVPLYSA-N vincaleukoblastine Chemical compound C([C@@H](C[C@]1(C(=O)OC)C=2C(=CC3=C([C@]45[C@H]([C@@]([C@H](OC(C)=O)[C@]6(CC)C=CCN([C@H]56)CC4)(O)C(=O)OC)N3C)C=2)OC)C[C@@](C2)(O)CC)N2CCC2=C1NC1=CC=CC=C21 JXLYSJRDGCGARV-XQKSVPLYSA-N 0.000 claims description 4
- YPBKTZBXSBLTDK-PKNBQFBNSA-N (3e)-3-[(3-bromo-4-fluoroanilino)-nitrosomethylidene]-4-[2-(sulfamoylamino)ethylamino]-1,2,5-oxadiazole Chemical compound NS(=O)(=O)NCCNC1=NON\C1=C(N=O)/NC1=CC=C(F)C(Br)=C1 YPBKTZBXSBLTDK-PKNBQFBNSA-N 0.000 claims description 3
- AOJJSUZBOXZQNB-VTZDEGQISA-N 4'-epidoxorubicin Chemical compound O([C@H]1C[C@@](O)(CC=2C(O)=C3C(=O)C=4C=CC=C(C=4C(=O)C3=C(O)C=21)OC)C(=O)CO)[C@H]1C[C@H](N)[C@@H](O)[C@H](C)O1 AOJJSUZBOXZQNB-VTZDEGQISA-N 0.000 claims description 3
- KLWPJMFMVPTNCC-UHFFFAOYSA-N Camptothecin Natural products CCC1(O)C(=O)OCC2=C1C=C3C4Nc5ccccc5C=C4CN3C2=O KLWPJMFMVPTNCC-UHFFFAOYSA-N 0.000 claims description 3
- GAGWJHPBXLXJQN-UORFTKCHSA-N Capecitabine Chemical compound C1=C(F)C(NC(=O)OCCCCC)=NC(=O)N1[C@H]1[C@H](O)[C@H](O)[C@@H](C)O1 GAGWJHPBXLXJQN-UORFTKCHSA-N 0.000 claims description 3
- GAGWJHPBXLXJQN-UHFFFAOYSA-N Capecitabine Natural products C1=C(F)C(NC(=O)OCCCCC)=NC(=O)N1C1C(O)C(O)C(C)O1 GAGWJHPBXLXJQN-UHFFFAOYSA-N 0.000 claims description 3
- DLGOEMSEDOSKAD-UHFFFAOYSA-N Carmustine Chemical compound ClCCNC(=O)N(N=O)CCCl DLGOEMSEDOSKAD-UHFFFAOYSA-N 0.000 claims description 3
- 229940046168 CpG oligodeoxynucleotide Drugs 0.000 claims description 3
- CMSMOCZEIVJLDB-UHFFFAOYSA-N Cyclophosphamide Chemical group ClCCN(CCCl)P1(=O)NCCCO1 CMSMOCZEIVJLDB-UHFFFAOYSA-N 0.000 claims description 3
- HTIJFSOGRVMCQR-UHFFFAOYSA-N Epirubicin Natural products COc1cccc2C(=O)c3c(O)c4CC(O)(CC(OC5CC(N)C(=O)C(C)O5)c4c(O)c3C(=O)c12)C(=O)CO HTIJFSOGRVMCQR-UHFFFAOYSA-N 0.000 claims description 3
- GHASVSINZRGABV-UHFFFAOYSA-N Fluorouracil Chemical compound FC1=CNC(=O)NC1=O GHASVSINZRGABV-UHFFFAOYSA-N 0.000 claims description 3
- FBOZXECLQNJBKD-ZDUSSCGKSA-N L-methotrexate Chemical compound C=1N=C2N=C(N)N=C(N)C2=NC=1CN(C)C1=CC=C(C(=O)N[C@@H](CCC(O)=O)C(O)=O)C=C1 FBOZXECLQNJBKD-ZDUSSCGKSA-N 0.000 claims description 3
- 239000002136 L01XE07 - Lapatinib Substances 0.000 claims description 3
- 206010050017 Lung cancer metastatic Diseases 0.000 claims description 3
- ZDZOTLJHXYCWBA-VCVYQWHSSA-N N-debenzoyl-N-(tert-butoxycarbonyl)-10-deacetyltaxol Chemical compound O([C@H]1[C@H]2[C@@](C([C@H](O)C3=C(C)[C@@H](OC(=O)[C@H](O)[C@@H](NC(=O)OC(C)(C)C)C=4C=CC=CC=4)C[C@]1(O)C3(C)C)=O)(C)[C@@H](O)C[C@H]1OC[C@]12OC(=O)C)C(=O)C1=CC=CC=C1 ZDZOTLJHXYCWBA-VCVYQWHSSA-N 0.000 claims description 3
- YGACXVRLDHEXKY-WXRXAMBDSA-N O[C@H](C[C@H]1c2c(cccc2F)-c2cncn12)[C@H]1CC[C@H](O)CC1 Chemical group O[C@H](C[C@H]1c2c(cccc2F)-c2cncn12)[C@H]1CC[C@H](O)CC1 YGACXVRLDHEXKY-WXRXAMBDSA-N 0.000 claims description 3
- 229920002873 Polyethylenimine Polymers 0.000 claims description 3
- 229940037003 alum Drugs 0.000 claims description 3
- 239000002260 anti-inflammatory agent Substances 0.000 claims description 3
- 229940121363 anti-inflammatory agent Drugs 0.000 claims description 3
- 239000003524 antilipemic agent Substances 0.000 claims description 3
- 229940034982 antineoplastic agent Drugs 0.000 claims description 3
- QKSKPIVNLNLAAV-UHFFFAOYSA-N bis(2-chloroethyl) sulfide Chemical compound ClCCSCCCl QKSKPIVNLNLAAV-UHFFFAOYSA-N 0.000 claims description 3
- VSJKWCGYPAHWDS-FQEVSTJZSA-N camptothecin Chemical compound C1=CC=C2C=C(CN3C4=CC5=C(C3=O)COC(=O)[C@]5(O)CC)C4=NC2=C1 VSJKWCGYPAHWDS-FQEVSTJZSA-N 0.000 claims description 3
- 229940127093 camptothecin Drugs 0.000 claims description 3
- 229960004117 capecitabine Drugs 0.000 claims description 3
- 229960004562 carboplatin Drugs 0.000 claims description 3
- 229960005243 carmustine Drugs 0.000 claims description 3
- DQLATGHUWYMOKM-UHFFFAOYSA-L cisplatin Chemical compound N[Pt](N)(Cl)Cl DQLATGHUWYMOKM-UHFFFAOYSA-L 0.000 claims description 3
- 229960004316 cisplatin Drugs 0.000 claims description 3
- 229960004397 cyclophosphamide Drugs 0.000 claims description 3
- 239000000824 cytostatic agent Substances 0.000 claims description 3
- 239000003599 detergent Substances 0.000 claims description 3
- VSJKWCGYPAHWDS-UHFFFAOYSA-N dl-camptothecin Natural products C1=CC=C2C=C(CN3C4=CC5=C(C3=O)COC(=O)C5(O)CC)C4=NC2=C1 VSJKWCGYPAHWDS-UHFFFAOYSA-N 0.000 claims description 3
- 229960003668 docetaxel Drugs 0.000 claims description 3
- 229950006370 epacadostat Drugs 0.000 claims description 3
- 229960001904 epirubicin Drugs 0.000 claims description 3
- UFNVPOGXISZXJD-XJPMSQCNSA-N eribulin Chemical compound C([C@H]1CC[C@@H]2O[C@@H]3[C@H]4O[C@H]5C[C@](O[C@H]4[C@H]2O1)(O[C@@H]53)CC[C@@H]1O[C@H](C(C1)=C)CC1)C(=O)C[C@@H]2[C@@H](OC)[C@@H](C[C@H](O)CN)O[C@H]2C[C@@H]2C(=C)[C@H](C)C[C@H]1O2 UFNVPOGXISZXJD-XJPMSQCNSA-N 0.000 claims description 3
- 229960003649 eribulin Drugs 0.000 claims description 3
- VJJPUSNTGOMMGY-MRVIYFEKSA-N etoposide Chemical compound COC1=C(O)C(OC)=CC([C@@H]2C3=CC=4OCOC=4C=C3[C@@H](O[C@H]3[C@@H]([C@@H](O)[C@@H]4O[C@H](C)OC[C@H]4O3)O)[C@@H]3[C@@H]2C(OC3)=O)=C1 VJJPUSNTGOMMGY-MRVIYFEKSA-N 0.000 claims description 3
- 229960005420 etoposide Drugs 0.000 claims description 3
- 229960002949 fluorouracil Drugs 0.000 claims description 3
- 230000004927 fusion Effects 0.000 claims description 3
- SDUQYLNIPVEERB-QPPQHZFASA-N gemcitabine Chemical compound O=C1N=C(N)C=CN1[C@H]1C(F)(F)[C@H](O)[C@@H](CO)O1 SDUQYLNIPVEERB-QPPQHZFASA-N 0.000 claims description 3
- 229960005277 gemcitabine Drugs 0.000 claims description 3
- BCFGMOOMADDAQU-UHFFFAOYSA-N lapatinib Chemical compound O1C(CNCCS(=O)(=O)C)=CC=C1C1=CC=C(N=CN=C2NC=3C=C(Cl)C(OCC=4C=C(F)C=CC=4)=CC=3)C2=C1 BCFGMOOMADDAQU-UHFFFAOYSA-N 0.000 claims description 3
- 229960004891 lapatinib Drugs 0.000 claims description 3
- HAWPXGHAZFHHAD-UHFFFAOYSA-N mechlorethamine Chemical class ClCCN(C)CCCl HAWPXGHAZFHHAD-UHFFFAOYSA-N 0.000 claims description 3
- 229960004961 mechlorethamine Drugs 0.000 claims description 3
- 229960000485 methotrexate Drugs 0.000 claims description 3
- 239000011859 microparticle Substances 0.000 claims description 3
- 229960001156 mitoxantrone Drugs 0.000 claims description 3
- KKZJGLLVHKMTCM-UHFFFAOYSA-N mitoxantrone Chemical compound O=C1C2=C(O)C=CC(O)=C2C(=O)C2=C1C(NCCNCCO)=CC=C2NCCNCCO KKZJGLLVHKMTCM-UHFFFAOYSA-N 0.000 claims description 3
- 239000002353 niosome Substances 0.000 claims description 3
- 229940115272 polyinosinic:polycytidylic acid Drugs 0.000 claims description 3
- 230000005855 radiation Effects 0.000 claims description 3
- 239000000018 receptor agonist Substances 0.000 claims description 3
- 229940044601 receptor agonist Drugs 0.000 claims description 3
- 229940044551 receptor antagonist Drugs 0.000 claims description 3
- 239000002464 receptor antagonist Substances 0.000 claims description 3
- 230000000306 recurrent effect Effects 0.000 claims description 3
- 208000016691 refractory malignant neoplasm Diseases 0.000 claims description 3
- 150000003431 steroids Chemical class 0.000 claims description 3
- 229960000575 trastuzumab Drugs 0.000 claims description 3
- GBABOYUKABKIAF-GHYRFKGUSA-N vinorelbine Chemical compound C1N(CC=2C3=CC=CC=C3NC=22)CC(CC)=C[C@H]1C[C@]2(C(=O)OC)C1=CC([C@]23[C@H]([C@]([C@H](OC(C)=O)[C@]4(CC)C=CCN([C@H]34)CC2)(O)C(=O)OC)N2C)=C2C=C1OC GBABOYUKABKIAF-GHYRFKGUSA-N 0.000 claims description 3
- 229960002066 vinorelbine Drugs 0.000 claims description 3
- 229960005475 antiinfective agent Drugs 0.000 claims description 2
- 244000144972 livestock Species 0.000 claims description 2
- 239000011856 silicon-based particle Substances 0.000 claims description 2
- 239000004599 antimicrobial Substances 0.000 claims 1
- 190000008236 carboplatin Chemical compound 0.000 claims 1
- GDFYVGREOHNWMM-UHFFFAOYSA-N platinum(4+);tetranitrate Chemical compound [Pt+4].[O-][N+]([O-])=O.[O-][N+]([O-])=O.[O-][N+]([O-])=O.[O-][N+]([O-])=O GDFYVGREOHNWMM-UHFFFAOYSA-N 0.000 claims 1
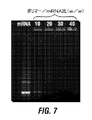
- 229940126582 mRNA vaccine Drugs 0.000 description 115
- 108700021021 mRNA Vaccine Proteins 0.000 description 111
- 108010058846 Ovalbumin Proteins 0.000 description 84
- 229940092253 ovalbumin Drugs 0.000 description 77
- 229960005486 vaccine Drugs 0.000 description 60
- 150000007523 nucleic acids Chemical class 0.000 description 59
- 238000000034 method Methods 0.000 description 53
- 210000001744 T-lymphocyte Anatomy 0.000 description 48
- 235000018102 proteins Nutrition 0.000 description 46
- 241000699670 Mus sp. Species 0.000 description 45
- 102000039446 nucleic acids Human genes 0.000 description 44
- 108020004707 nucleic acids Proteins 0.000 description 44
- 235000001014 amino acid Nutrition 0.000 description 29
- 102000040430 polynucleotide Human genes 0.000 description 28
- 108091033319 polynucleotide Proteins 0.000 description 28
- 239000002157 polynucleotide Substances 0.000 description 28
- 229940024606 amino acid Drugs 0.000 description 26
- 150000001413 amino acids Chemical class 0.000 description 26
- 150000002632 lipids Chemical class 0.000 description 26
- 229920001212 Poly(beta amino esters) Polymers 0.000 description 23
- 230000000694 effects Effects 0.000 description 23
- 230000035772 mutation Effects 0.000 description 21
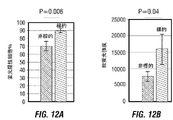
- 230000005809 anti-tumor immunity Effects 0.000 description 19
- 238000009472 formulation Methods 0.000 description 19
- 206010006187 Breast cancer Diseases 0.000 description 18
- 230000000295 complement effect Effects 0.000 description 18
- 239000000243 solution Substances 0.000 description 18
- 208000026310 Breast neoplasm Diseases 0.000 description 17
- 102100025064 Cellular tumor antigen p53 Human genes 0.000 description 17
- 102000004127 Cytokines Human genes 0.000 description 17
- 108090000695 Cytokines Proteins 0.000 description 17
- 125000003275 alpha amino acid group Chemical group 0.000 description 16
- 229940038694 mRNA-based vaccine Drugs 0.000 description 16
- 238000012384 transportation and delivery Methods 0.000 description 16
- 239000002585 base Substances 0.000 description 15
- 210000001519 tissue Anatomy 0.000 description 15
- 108091028043 Nucleic acid sequence Proteins 0.000 description 14
- 108010078814 Tumor Suppressor Protein p53 Proteins 0.000 description 14
- 230000008901 benefit Effects 0.000 description 14
- 238000011534 incubation Methods 0.000 description 14
- 108010002350 Interleukin-2 Proteins 0.000 description 13
- 102000000588 Interleukin-2 Human genes 0.000 description 13
- 238000000338 in vitro Methods 0.000 description 13
- 230000028327 secretion Effects 0.000 description 13
- 210000001151 cytotoxic T lymphocyte Anatomy 0.000 description 12
- 238000011161 development Methods 0.000 description 12
- 230000018109 developmental process Effects 0.000 description 12
- 210000004072 lung Anatomy 0.000 description 12
- 201000001441 melanoma Diseases 0.000 description 12
- 239000002773 nucleotide Substances 0.000 description 12
- 125000003729 nucleotide group Chemical group 0.000 description 12
- 150000003839 salts Chemical class 0.000 description 12
- 239000000523 sample Substances 0.000 description 12
- 101000605639 Homo sapiens Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform Proteins 0.000 description 11
- 102000013462 Interleukin-12 Human genes 0.000 description 11
- 108010065805 Interleukin-12 Proteins 0.000 description 11
- 230000030741 antigen processing and presentation Effects 0.000 description 11
- 229940117681 interleukin-12 Drugs 0.000 description 11
- 230000035800 maturation Effects 0.000 description 11
- 239000013612 plasmid Substances 0.000 description 11
- 230000003389 potentiating effect Effects 0.000 description 11
- 238000002360 preparation method Methods 0.000 description 11
- 230000000638 stimulation Effects 0.000 description 11
- KWVJHCQQUFDPLU-YEUCEMRASA-N 2,3-bis[[(z)-octadec-9-enoyl]oxy]propyl-trimethylazanium Chemical compound CCCCCCCC\C=C/CCCCCCCC(=O)OCC(C[N+](C)(C)C)OC(=O)CCCCCCC\C=C/CCCCCCCC KWVJHCQQUFDPLU-YEUCEMRASA-N 0.000 description 10
- 238000002965 ELISA Methods 0.000 description 10
- 206010027476 Metastases Diseases 0.000 description 10
- 239000003153 chemical reaction reagent Substances 0.000 description 10
- LOKCTEFSRHRXRJ-UHFFFAOYSA-I dipotassium trisodium dihydrogen phosphate hydrogen phosphate dichloride Chemical compound P(=O)(O)(O)[O-].[K+].P(=O)(O)([O-])[O-].[Na+].[Na+].[Cl-].[K+].[Cl-].[Na+] LOKCTEFSRHRXRJ-UHFFFAOYSA-I 0.000 description 10
- 238000000684 flow cytometry Methods 0.000 description 10
- 230000028993 immune response Effects 0.000 description 10
- 239000000463 material Substances 0.000 description 10
- 239000002953 phosphate buffered saline Substances 0.000 description 10
- 230000004936 stimulating effect Effects 0.000 description 10
- 238000002255 vaccination Methods 0.000 description 10
- 238000011740 C57BL/6 mouse Methods 0.000 description 9
- 241000699666 Mus <mouse, genus> Species 0.000 description 9
- 239000013543 active substance Substances 0.000 description 9
- 238000003556 assay Methods 0.000 description 9
- WLNARFZDISHUGS-MIXBDBMTSA-N cholesteryl hemisuccinate Chemical compound C1C=C2C[C@@H](OC(=O)CCC(O)=O)CC[C@]2(C)[C@@H]2[C@@H]1[C@@H]1CC[C@H]([C@H](C)CCCC(C)C)[C@@]1(C)CC2 WLNARFZDISHUGS-MIXBDBMTSA-N 0.000 description 9
- 230000009401 metastasis Effects 0.000 description 9
- 238000012360 testing method Methods 0.000 description 9
- 239000013598 vector Substances 0.000 description 9
- 101001012157 Homo sapiens Receptor tyrosine-protein kinase erbB-2 Proteins 0.000 description 8
- 108700018351 Major Histocompatibility Complex Proteins 0.000 description 8
- 108010011536 PTEN Phosphohydrolase Proteins 0.000 description 8
- 102100030086 Receptor tyrosine-protein kinase erbB-2 Human genes 0.000 description 8
- 230000006044 T cell activation Effects 0.000 description 8
- 239000004480 active ingredient Substances 0.000 description 8
- 210000001185 bone marrow Anatomy 0.000 description 8
- 229940022399 cancer vaccine Drugs 0.000 description 8
- 238000009566 cancer vaccine Methods 0.000 description 8
- 239000005090 green fluorescent protein Substances 0.000 description 8
- 230000002163 immunogen Effects 0.000 description 8
- 238000002347 injection Methods 0.000 description 8
- 239000007924 injection Substances 0.000 description 8
- 210000000952 spleen Anatomy 0.000 description 8
- 238000006467 substitution reaction Methods 0.000 description 8
- 230000020382 suppression by virus of host antigen processing and presentation of peptide antigen via MHC class I Effects 0.000 description 8
- 229940021747 therapeutic vaccine Drugs 0.000 description 8
- 238000013518 transcription Methods 0.000 description 8
- 230000035897 transcription Effects 0.000 description 8
- 102000016607 Diphtheria Toxin Human genes 0.000 description 7
- 108010053187 Diphtheria Toxin Proteins 0.000 description 7
- DHMQDGOQFOQNFH-UHFFFAOYSA-N Glycine Chemical compound NCC(O)=O DHMQDGOQFOQNFH-UHFFFAOYSA-N 0.000 description 7
- 102100038332 Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform Human genes 0.000 description 7
- 238000004458 analytical method Methods 0.000 description 7
- 238000001727 in vivo Methods 0.000 description 7
- 230000005764 inhibitory process Effects 0.000 description 7
- 210000001165 lymph node Anatomy 0.000 description 7
- 210000000056 organ Anatomy 0.000 description 7
- 239000008194 pharmaceutical composition Substances 0.000 description 7
- 230000004614 tumor growth Effects 0.000 description 7
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 6
- MUBZPKHOEPUJKR-UHFFFAOYSA-N Oxalic acid Chemical compound OC(=O)C(O)=O MUBZPKHOEPUJKR-UHFFFAOYSA-N 0.000 description 6
- 125000000539 amino acid group Chemical group 0.000 description 6
- 238000002619 cancer immunotherapy Methods 0.000 description 6
- 230000015556 catabolic process Effects 0.000 description 6
- HVYWMOMLDIMFJA-DPAQBDIFSA-N cholesterol Chemical compound C1C=C2C[C@@H](O)CC[C@]2(C)[C@@H]2[C@@H]1[C@@H]1CC[C@H]([C@H](C)CCCC(C)C)[C@@]1(C)CC2 HVYWMOMLDIMFJA-DPAQBDIFSA-N 0.000 description 6
- 230000000875 corresponding effect Effects 0.000 description 6
- 238000006731 degradation reaction Methods 0.000 description 6
- 238000001514 detection method Methods 0.000 description 6
- 230000001965 increasing effect Effects 0.000 description 6
- 230000000869 mutational effect Effects 0.000 description 6
- UCSJYZPVAKXKNQ-HZYVHMACSA-N streptomycin Chemical compound CN[C@H]1[C@H](O)[C@@H](O)[C@H](CO)O[C@H]1O[C@@H]1[C@](C=O)(O)[C@H](C)O[C@H]1O[C@@H]1[C@@H](NC(N)=N)[C@H](O)[C@@H](NC(N)=N)[C@H](O)[C@H]1O UCSJYZPVAKXKNQ-HZYVHMACSA-N 0.000 description 6
- 238000011269 treatment regimen Methods 0.000 description 6
- MTCFGRXMJLQNBG-REOHCLBHSA-N (2S)-2-Amino-3-hydroxypropansäure Chemical compound OC[C@H](N)C(O)=O MTCFGRXMJLQNBG-REOHCLBHSA-N 0.000 description 5
- 210000001266 CD8-positive T-lymphocyte Anatomy 0.000 description 5
- 208000035473 Communicable disease Diseases 0.000 description 5
- WQZGKKKJIJFFOK-QTVWNMPRSA-N D-mannopyranose Chemical compound OC[C@H]1OC(O)[C@@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-QTVWNMPRSA-N 0.000 description 5
- 229940043367 IDO1 inhibitor Drugs 0.000 description 5
- 108010074328 Interferon-gamma Proteins 0.000 description 5
- DCXYFEDJOCDNAF-REOHCLBHSA-N L-asparagine Chemical compound OC(=O)[C@@H](N)CC(N)=O DCXYFEDJOCDNAF-REOHCLBHSA-N 0.000 description 5
- CKLJMWTZIZZHCS-REOHCLBHSA-N L-aspartic acid Chemical compound OC(=O)[C@@H](N)CC(O)=O CKLJMWTZIZZHCS-REOHCLBHSA-N 0.000 description 5
- WHUUTDBJXJRKMK-VKHMYHEASA-N L-glutamic acid Chemical compound OC(=O)[C@@H](N)CCC(O)=O WHUUTDBJXJRKMK-VKHMYHEASA-N 0.000 description 5
- ZDXPYRJPNDTMRX-VKHMYHEASA-N L-glutamine Chemical compound OC(=O)[C@@H](N)CCC(N)=O ZDXPYRJPNDTMRX-VKHMYHEASA-N 0.000 description 5
- AGPKZVBTJJNPAG-WHFBIAKZSA-N L-isoleucine Chemical compound CC[C@H](C)[C@H](N)C(O)=O AGPKZVBTJJNPAG-WHFBIAKZSA-N 0.000 description 5
- ROHFNLRQFUQHCH-YFKPBYRVSA-N L-leucine Chemical compound CC(C)C[C@H](N)C(O)=O ROHFNLRQFUQHCH-YFKPBYRVSA-N 0.000 description 5
- KDXKERNSBIXSRK-YFKPBYRVSA-N L-lysine Chemical compound NCCCC[C@H](N)C(O)=O KDXKERNSBIXSRK-YFKPBYRVSA-N 0.000 description 5
- AYFVYJQAPQTCCC-GBXIJSLDSA-N L-threonine Chemical compound C[C@@H](O)[C@H](N)C(O)=O AYFVYJQAPQTCCC-GBXIJSLDSA-N 0.000 description 5
- KDXKERNSBIXSRK-UHFFFAOYSA-N Lysine Natural products NCCCCC(N)C(O)=O KDXKERNSBIXSRK-UHFFFAOYSA-N 0.000 description 5
- 206010027458 Metastases to lung Diseases 0.000 description 5
- 102000014160 PTEN Phosphohydrolase Human genes 0.000 description 5
- 102100039390 Toll-like receptor 7 Human genes 0.000 description 5
- KZSNJWFQEVHDMF-UHFFFAOYSA-N Valine Natural products CC(C)C(N)C(O)=O KZSNJWFQEVHDMF-UHFFFAOYSA-N 0.000 description 5
- 230000001093 anti-cancer Effects 0.000 description 5
- 230000000259 anti-tumor effect Effects 0.000 description 5
- 238000013459 approach Methods 0.000 description 5
- 230000004071 biological effect Effects 0.000 description 5
- 230000022534 cell killing Effects 0.000 description 5
- 230000004700 cellular uptake Effects 0.000 description 5
- 239000011258 core-shell material Substances 0.000 description 5
- 229940079593 drug Drugs 0.000 description 5
- 238000005516 engineering process Methods 0.000 description 5
- 230000006870 function Effects 0.000 description 5
- 239000000499 gel Substances 0.000 description 5
- 239000001963 growth medium Substances 0.000 description 5
- 238000009396 hybridization Methods 0.000 description 5
- 230000002209 hydrophobic effect Effects 0.000 description 5
- 230000001404 mediated effect Effects 0.000 description 5
- 230000004048 modification Effects 0.000 description 5
- 238000012986 modification Methods 0.000 description 5
- 238000010172 mouse model Methods 0.000 description 5
- 238000004806 packaging method and process Methods 0.000 description 5
- 230000001737 promoting effect Effects 0.000 description 5
- 230000011664 signaling Effects 0.000 description 5
- 239000000126 substance Substances 0.000 description 5
- 230000036962 time dependent Effects 0.000 description 5
- 210000003171 tumor-infiltrating lymphocyte Anatomy 0.000 description 5
- MZOFCQQQCNRIBI-VMXHOPILSA-N (3s)-4-[[(2s)-1-[[(2s)-1-[[(1s)-1-carboxy-2-hydroxyethyl]amino]-4-methyl-1-oxopentan-2-yl]amino]-5-(diaminomethylideneamino)-1-oxopentan-2-yl]amino]-3-[[2-[[(2s)-2,6-diaminohexanoyl]amino]acetyl]amino]-4-oxobutanoic acid Chemical compound OC[C@@H](C(O)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CCCN=C(N)N)NC(=O)[C@H](CC(O)=O)NC(=O)CNC(=O)[C@@H](N)CCCCN MZOFCQQQCNRIBI-VMXHOPILSA-N 0.000 description 4
- WHTVZRBIWZFKQO-AWEZNQCLSA-N (S)-chloroquine Chemical compound ClC1=CC=C2C(N[C@@H](C)CCCN(CC)CC)=CC=NC2=C1 WHTVZRBIWZFKQO-AWEZNQCLSA-N 0.000 description 4
- 239000004475 Arginine Substances 0.000 description 4
- DCXYFEDJOCDNAF-UHFFFAOYSA-N Asparagine Natural products OC(=O)C(N)CC(N)=O DCXYFEDJOCDNAF-UHFFFAOYSA-N 0.000 description 4
- 238000011725 BALB/c mouse Methods 0.000 description 4
- 101150014003 Batf3 gene Proteins 0.000 description 4
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 4
- ZHNUHDYFZUAESO-UHFFFAOYSA-N Formamide Chemical compound NC=O ZHNUHDYFZUAESO-UHFFFAOYSA-N 0.000 description 4
- 108010043121 Green Fluorescent Proteins Proteins 0.000 description 4
- 102000004144 Green Fluorescent Proteins Human genes 0.000 description 4
- 108010074032 HLA-A2 Antigen Proteins 0.000 description 4
- 102000025850 HLA-A2 Antigen Human genes 0.000 description 4
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 4
- 102100022297 Integrin alpha-X Human genes 0.000 description 4
- COLNVLDHVKWLRT-QMMMGPOBSA-N L-phenylalanine Chemical compound OC(=O)[C@@H](N)CC1=CC=CC=C1 COLNVLDHVKWLRT-QMMMGPOBSA-N 0.000 description 4
- OUYCCCASQSFEME-QMMMGPOBSA-N L-tyrosine Chemical compound OC(=O)[C@@H](N)CC1=CC=C(O)C=C1 OUYCCCASQSFEME-QMMMGPOBSA-N 0.000 description 4
- KZSNJWFQEVHDMF-BYPYZUCNSA-N L-valine Chemical compound CC(C)[C@H](N)C(O)=O KZSNJWFQEVHDMF-BYPYZUCNSA-N 0.000 description 4
- ROHFNLRQFUQHCH-UHFFFAOYSA-N Leucine Natural products CC(C)CC(N)C(O)=O ROHFNLRQFUQHCH-UHFFFAOYSA-N 0.000 description 4
- 208000011738 Lichen planopilaris Diseases 0.000 description 4
- 239000004472 Lysine Substances 0.000 description 4
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 4
- MTCFGRXMJLQNBG-UHFFFAOYSA-N Serine Natural products OCC(N)C(O)=O MTCFGRXMJLQNBG-UHFFFAOYSA-N 0.000 description 4
- 229940125971 TLR7/8 inhibitor Drugs 0.000 description 4
- AYFVYJQAPQTCCC-UHFFFAOYSA-N Threonine Natural products CC(O)C(N)C(O)=O AYFVYJQAPQTCCC-UHFFFAOYSA-N 0.000 description 4
- 239000004473 Threonine Substances 0.000 description 4
- 102000040945 Transcription factor Human genes 0.000 description 4
- 108091023040 Transcription factor Proteins 0.000 description 4
- 108060008682 Tumor Necrosis Factor Proteins 0.000 description 4
- 102000000852 Tumor Necrosis Factor-alpha Human genes 0.000 description 4
- 241000251539 Vertebrata <Metazoa> Species 0.000 description 4
- 241000700605 Viruses Species 0.000 description 4
- 239000002253 acid Substances 0.000 description 4
- ODKSFYDXXFIFQN-UHFFFAOYSA-N arginine Natural products OC(=O)C(N)CCCNC(N)=N ODKSFYDXXFIFQN-UHFFFAOYSA-N 0.000 description 4
- 235000009582 asparagine Nutrition 0.000 description 4
- 229960001230 asparagine Drugs 0.000 description 4
- 230000015572 biosynthetic process Effects 0.000 description 4
- 230000010261 cell growth Effects 0.000 description 4
- 230000003833 cell viability Effects 0.000 description 4
- 229960003677 chloroquine Drugs 0.000 description 4
- WHTVZRBIWZFKQO-UHFFFAOYSA-N chloroquine Natural products ClC1=CC=C2C(NC(C)CCCN(CC)CC)=CC=NC2=C1 WHTVZRBIWZFKQO-UHFFFAOYSA-N 0.000 description 4
- 230000004041 dendritic cell maturation Effects 0.000 description 4
- 210000002889 endothelial cell Anatomy 0.000 description 4
- 239000003623 enhancer Substances 0.000 description 4
- 238000002474 experimental method Methods 0.000 description 4
- 239000012091 fetal bovine serum Substances 0.000 description 4
- ZDXPYRJPNDTMRX-UHFFFAOYSA-N glutamine Natural products OC(=O)C(N)CCC(N)=O ZDXPYRJPNDTMRX-UHFFFAOYSA-N 0.000 description 4
- HNDVDQJCIGZPNO-UHFFFAOYSA-N histidine Natural products OC(=O)C(N)CC1=CN=CN1 HNDVDQJCIGZPNO-UHFFFAOYSA-N 0.000 description 4
- 230000003463 hyperproliferative effect Effects 0.000 description 4
- 210000002865 immune cell Anatomy 0.000 description 4
- 239000004615 ingredient Substances 0.000 description 4
- 230000003993 interaction Effects 0.000 description 4
- 229960000310 isoleucine Drugs 0.000 description 4
- AGPKZVBTJJNPAG-UHFFFAOYSA-N isoleucine Natural products CCC(C)C(N)C(O)=O AGPKZVBTJJNPAG-UHFFFAOYSA-N 0.000 description 4
- 230000000670 limiting effect Effects 0.000 description 4
- 208000020816 lung neoplasm Diseases 0.000 description 4
- 239000003550 marker Substances 0.000 description 4
- 239000002609 medium Substances 0.000 description 4
- 239000013642 negative control Substances 0.000 description 4
- 208000011797 pustulosis palmaris et plantaris Diseases 0.000 description 4
- 230000004044 response Effects 0.000 description 4
- 239000004055 small Interfering RNA Substances 0.000 description 4
- 150000003384 small molecules Chemical class 0.000 description 4
- 238000001228 spectrum Methods 0.000 description 4
- 238000007920 subcutaneous administration Methods 0.000 description 4
- 239000004474 valine Substances 0.000 description 4
- 241000271566 Aves Species 0.000 description 3
- 101150013553 CD40 gene Proteins 0.000 description 3
- 102000053602 DNA Human genes 0.000 description 3
- 208000001382 Experimental Melanoma Diseases 0.000 description 3
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 3
- 239000004471 Glycine Substances 0.000 description 3
- 101000669402 Homo sapiens Toll-like receptor 7 Proteins 0.000 description 3
- 102100037850 Interferon gamma Human genes 0.000 description 3
- 102000006992 Interferon-alpha Human genes 0.000 description 3
- 108010047761 Interferon-alpha Proteins 0.000 description 3
- 102000003996 Interferon-beta Human genes 0.000 description 3
- QNAYBMKLOCPYGJ-REOHCLBHSA-N L-alanine Chemical compound C[C@H](N)C(O)=O QNAYBMKLOCPYGJ-REOHCLBHSA-N 0.000 description 3
- FFEARJCKVFRZRR-BYPYZUCNSA-N L-methionine Chemical compound CSCC[C@H](N)C(O)=O FFEARJCKVFRZRR-BYPYZUCNSA-N 0.000 description 3
- QIVBCDIJIAJPQS-VIFPVBQESA-N L-tryptophane Chemical compound C1=CC=C2C(C[C@H](N)C(O)=O)=CNC2=C1 QIVBCDIJIAJPQS-VIFPVBQESA-N 0.000 description 3
- 108060001084 Luciferase Proteins 0.000 description 3
- FBKMWOJEPMPVTQ-UHFFFAOYSA-N N'-(3-bromo-4-fluorophenyl)-N-hydroxy-4-[2-(sulfamoylamino)ethylamino]-1,2,5-oxadiazole-3-carboximidamide Chemical compound NS(=O)(=O)NCCNC1=NON=C1C(=NO)NC1=CC=C(F)C(Br)=C1 FBKMWOJEPMPVTQ-UHFFFAOYSA-N 0.000 description 3
- 108020005187 Oligonucleotide Probes Proteins 0.000 description 3
- 229930182555 Penicillin Natural products 0.000 description 3
- JGSARLDLIJGVTE-MBNYWOFBSA-N Penicillin G Chemical compound N([C@H]1[C@H]2SC([C@@H](N2C1=O)C(O)=O)(C)C)C(=O)CC1=CC=CC=C1 JGSARLDLIJGVTE-MBNYWOFBSA-N 0.000 description 3
- KWYUFKZDYYNOTN-UHFFFAOYSA-M Potassium hydroxide Chemical compound [OH-].[K+] KWYUFKZDYYNOTN-UHFFFAOYSA-M 0.000 description 3
- 241000288906 Primates Species 0.000 description 3
- ONIBWKKTOPOVIA-UHFFFAOYSA-N Proline Natural products OC(=O)C1CCCN1 ONIBWKKTOPOVIA-UHFFFAOYSA-N 0.000 description 3
- 239000012980 RPMI-1640 medium Substances 0.000 description 3
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 3
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 3
- QIVBCDIJIAJPQS-UHFFFAOYSA-N Tryptophan Natural products C1=CC=C2C(CC(N)C(O)=O)=CNC2=C1 QIVBCDIJIAJPQS-UHFFFAOYSA-N 0.000 description 3
- 102100040245 Tumor necrosis factor receptor superfamily member 5 Human genes 0.000 description 3
- 230000003213 activating effect Effects 0.000 description 3
- 230000004913 activation Effects 0.000 description 3
- 235000004279 alanine Nutrition 0.000 description 3
- 230000004075 alteration Effects 0.000 description 3
- XSDQTOBWRPYKKA-UHFFFAOYSA-N amiloride Chemical compound NC(=N)NC(=O)C1=NC(Cl)=C(N)N=C1N XSDQTOBWRPYKKA-UHFFFAOYSA-N 0.000 description 3
- 229960002576 amiloride Drugs 0.000 description 3
- 239000007864 aqueous solution Substances 0.000 description 3
- 229940009098 aspartate Drugs 0.000 description 3
- 229920005601 base polymer Polymers 0.000 description 3
- 238000005415 bioluminescence Methods 0.000 description 3
- 230000029918 bioluminescence Effects 0.000 description 3
- 239000006172 buffering agent Substances 0.000 description 3
- 230000001413 cellular effect Effects 0.000 description 3
- ZPEIMTDSQAKGNT-UHFFFAOYSA-N chlorpromazine Chemical compound C1=C(Cl)C=C2N(CCCN(C)C)C3=CC=CC=C3SC2=C1 ZPEIMTDSQAKGNT-UHFFFAOYSA-N 0.000 description 3
- 229960001076 chlorpromazine Drugs 0.000 description 3
- 235000012000 cholesterol Nutrition 0.000 description 3
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 description 3
- 235000018417 cysteine Nutrition 0.000 description 3
- XUJNEKJLAYXESH-UHFFFAOYSA-N cysteine Natural products SCC(N)C(O)=O XUJNEKJLAYXESH-UHFFFAOYSA-N 0.000 description 3
- 229940029030 dendritic cell vaccine Drugs 0.000 description 3
- 230000001419 dependent effect Effects 0.000 description 3
- 238000003745 diagnosis Methods 0.000 description 3
- 238000009826 distribution Methods 0.000 description 3
- 230000002255 enzymatic effect Effects 0.000 description 3
- 102000015694 estrogen receptors Human genes 0.000 description 3
- 108010038795 estrogen receptors Proteins 0.000 description 3
- 238000000799 fluorescence microscopy Methods 0.000 description 3
- 230000002068 genetic effect Effects 0.000 description 3
- 229940045109 genistein Drugs 0.000 description 3
- TZBJGXHYKVUXJN-UHFFFAOYSA-N genistein Natural products C1=CC(O)=CC=C1C1=COC2=CC(O)=CC(O)=C2C1=O TZBJGXHYKVUXJN-UHFFFAOYSA-N 0.000 description 3
- 235000006539 genistein Nutrition 0.000 description 3
- ZCOLJUOHXJRHDI-CMWLGVBASA-N genistein 7-O-beta-D-glucoside Chemical compound O[C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@H]1OC1=CC(O)=C2C(=O)C(C=3C=CC(O)=CC=3)=COC2=C1 ZCOLJUOHXJRHDI-CMWLGVBASA-N 0.000 description 3
- 229930195712 glutamate Natural products 0.000 description 3
- 238000003384 imaging method Methods 0.000 description 3
- 210000000987 immune system Anatomy 0.000 description 3
- 230000001976 improved effect Effects 0.000 description 3
- 230000008595 infiltration Effects 0.000 description 3
- 238000001764 infiltration Methods 0.000 description 3
- 230000002757 inflammatory effect Effects 0.000 description 3
- 230000002401 inhibitory effect Effects 0.000 description 3
- 238000011081 inoculation Methods 0.000 description 3
- 230000010354 integration Effects 0.000 description 3
- 229960001388 interferon-beta Drugs 0.000 description 3
- 238000007918 intramuscular administration Methods 0.000 description 3
- 238000002955 isolation Methods 0.000 description 3
- 239000007788 liquid Substances 0.000 description 3
- 238000005259 measurement Methods 0.000 description 3
- 230000007246 mechanism Effects 0.000 description 3
- 229930182817 methionine Natural products 0.000 description 3
- 238000012544 monitoring process Methods 0.000 description 3
- 238000010899 nucleation Methods 0.000 description 3
- 239000002751 oligonucleotide probe Substances 0.000 description 3
- 229940049954 penicillin Drugs 0.000 description 3
- 229940023041 peptide vaccine Drugs 0.000 description 3
- 210000003819 peripheral blood mononuclear cell Anatomy 0.000 description 3
- COLNVLDHVKWLRT-UHFFFAOYSA-N phenylalanine Natural products OC(=O)C(N)CC1=CC=CC=C1 COLNVLDHVKWLRT-UHFFFAOYSA-N 0.000 description 3
- YVUQSNJEYSNKRX-UHFFFAOYSA-N pimozide Chemical compound C1=CC(F)=CC=C1C(C=1C=CC(F)=CC=1)CCCN1CCC(N2C(NC3=CC=CC=C32)=O)CC1 YVUQSNJEYSNKRX-UHFFFAOYSA-N 0.000 description 3
- 229960003634 pimozide Drugs 0.000 description 3
- 230000008569 process Effects 0.000 description 3
- 230000002685 pulmonary effect Effects 0.000 description 3
- 230000001105 regulatory effect Effects 0.000 description 3
- 238000011160 research Methods 0.000 description 3
- 239000002904 solvent Substances 0.000 description 3
- 229960005322 streptomycin Drugs 0.000 description 3
- 239000000758 substrate Substances 0.000 description 3
- 238000003786 synthesis reaction Methods 0.000 description 3
- 230000008685 targeting Effects 0.000 description 3
- 238000011830 transgenic mouse model Methods 0.000 description 3
- 230000014616 translation Effects 0.000 description 3
- 238000004627 transmission electron microscopy Methods 0.000 description 3
- OUYCCCASQSFEME-UHFFFAOYSA-N tyrosine Natural products OC(=O)C(N)CC1=CC=C(O)C=C1 OUYCCCASQSFEME-UHFFFAOYSA-N 0.000 description 3
- YBJHBAHKTGYVGT-ZKWXMUAHSA-N (+)-Biotin Chemical compound N1C(=O)N[C@@H]2[C@H](CCCCC(=O)O)SC[C@@H]21 YBJHBAHKTGYVGT-ZKWXMUAHSA-N 0.000 description 2
- JVJGCCBAOOWGEO-RUTPOYCXSA-N (2s)-2-[[(2s)-2-[[(2s)-2-[[(2s)-2-[[(2s)-4-amino-2-[[(2s,3s)-2-[[(2s,3s)-2-[[(2s)-2-azaniumyl-3-hydroxypropanoyl]amino]-3-methylpentanoyl]amino]-3-methylpentanoyl]amino]-4-oxobutanoyl]amino]-3-phenylpropanoyl]amino]-4-carboxylatobutanoyl]amino]-6-azaniumy Chemical compound OC[C@H](N)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@H](C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(C)C)C(O)=O)CC1=CC=CC=C1 JVJGCCBAOOWGEO-RUTPOYCXSA-N 0.000 description 2
- KDCGOANMDULRCW-UHFFFAOYSA-N 7H-purine Chemical compound N1=CNC2=NC=NC2=C1 KDCGOANMDULRCW-UHFFFAOYSA-N 0.000 description 2
- 206010067484 Adverse reaction Diseases 0.000 description 2
- CIWBSHSKHKDKBQ-JLAZNSOCSA-N Ascorbic acid Chemical compound OC[C@H](O)[C@H]1OC(=O)C(O)=C1O CIWBSHSKHKDKBQ-JLAZNSOCSA-N 0.000 description 2
- 241000972773 Aulopiformes Species 0.000 description 2
- 206010005003 Bladder cancer Diseases 0.000 description 2
- 241000283690 Bos taurus Species 0.000 description 2
- 108700031361 Brachyury Proteins 0.000 description 2
- 108091033409 CRISPR Proteins 0.000 description 2
- 241000282421 Canidae Species 0.000 description 2
- 241000282472 Canis lupus familiaris Species 0.000 description 2
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 description 2
- 108091026890 Coding region Proteins 0.000 description 2
- 241000938605 Crocodylia Species 0.000 description 2
- RGHNJXZEOKUKBD-SQOUGZDYSA-N D-gluconic acid Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@@H](O)C(O)=O RGHNJXZEOKUKBD-SQOUGZDYSA-N 0.000 description 2
- 108010041986 DNA Vaccines Proteins 0.000 description 2
- 230000004544 DNA amplification Effects 0.000 description 2
- 229940021995 DNA vaccine Drugs 0.000 description 2
- FEWJPZIEWOKRBE-JCYAYHJZSA-N Dextrotartaric acid Chemical compound OC(=O)[C@H](O)[C@@H](O)C(O)=O FEWJPZIEWOKRBE-JCYAYHJZSA-N 0.000 description 2
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 2
- 241000283086 Equidae Species 0.000 description 2
- 241000588724 Escherichia coli Species 0.000 description 2
- 241000282326 Felis catus Species 0.000 description 2
- VZCYOOQTPOCHFL-OWOJBTEDSA-N Fumaric acid Chemical compound OC(=O)\C=C\C(O)=O VZCYOOQTPOCHFL-OWOJBTEDSA-N 0.000 description 2
- 241000287828 Gallus gallus Species 0.000 description 2
- 108700028146 Genetic Enhancer Elements Proteins 0.000 description 2
- WHUUTDBJXJRKMK-UHFFFAOYSA-N Glutamic acid Natural products OC(=O)C(N)CCC(O)=O WHUUTDBJXJRKMK-UHFFFAOYSA-N 0.000 description 2
- 108010017213 Granulocyte-Macrophage Colony-Stimulating Factor Proteins 0.000 description 2
- 102100039620 Granulocyte-macrophage colony-stimulating factor Human genes 0.000 description 2
- 208000017891 HER2 positive breast carcinoma Diseases 0.000 description 2
- 108090000144 Human Proteins Proteins 0.000 description 2
- 229940076838 Immune checkpoint inhibitor Drugs 0.000 description 2
- 102000008070 Interferon-gamma Human genes 0.000 description 2
- 102000004388 Interleukin-4 Human genes 0.000 description 2
- 108090000978 Interleukin-4 Proteins 0.000 description 2
- 208000008839 Kidney Neoplasms Diseases 0.000 description 2
- 239000005089 Luciferase Substances 0.000 description 2
- 206010058467 Lung neoplasm malignant Diseases 0.000 description 2
- 108700011259 MicroRNAs Proteins 0.000 description 2
- 241000699660 Mus musculus Species 0.000 description 2
- PXHVJJICTQNCMI-UHFFFAOYSA-N Nickel Chemical compound [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 description 2
- 108091034117 Oligonucleotide Proteins 0.000 description 2
- 108700020796 Oncogene Proteins 0.000 description 2
- 206010061902 Pancreatic neoplasm Diseases 0.000 description 2
- 229930040373 Paraformaldehyde Natural products 0.000 description 2
- 241001494479 Pecora Species 0.000 description 2
- 108091093037 Peptide nucleic acid Proteins 0.000 description 2
- 206010057249 Phagocytosis Diseases 0.000 description 2
- CZPWVGJYEJSRLH-UHFFFAOYSA-N Pyrimidine Chemical compound C1=CN=CN=C1 CZPWVGJYEJSRLH-UHFFFAOYSA-N 0.000 description 2
- 241000700159 Rattus Species 0.000 description 2
- 206010038389 Renal cancer Diseases 0.000 description 2
- 102000039471 Small Nuclear RNA Human genes 0.000 description 2
- 102000042773 Small Nucleolar RNA Human genes 0.000 description 2
- 108020003224 Small Nucleolar RNA Proteins 0.000 description 2
- 208000005718 Stomach Neoplasms Diseases 0.000 description 2
- 241000282887 Suidae Species 0.000 description 2
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 2
- 108091008874 T cell receptors Proteins 0.000 description 2
- 102000016266 T-Cell Antigen Receptors Human genes 0.000 description 2
- 101150080074 TP53 gene Proteins 0.000 description 2
- FEWJPZIEWOKRBE-UHFFFAOYSA-N Tartaric acid Natural products [H+].[H+].[O-]C(=O)C(O)C(O)C([O-])=O FEWJPZIEWOKRBE-UHFFFAOYSA-N 0.000 description 2
- 108010060825 Toll-Like Receptor 7 Proteins 0.000 description 2
- 108010060752 Toll-Like Receptor 8 Proteins 0.000 description 2
- 102000002689 Toll-like receptor Human genes 0.000 description 2
- 108020000411 Toll-like receptor Proteins 0.000 description 2
- 102100033110 Toll-like receptor 8 Human genes 0.000 description 2
- 108700029229 Transcriptional Regulatory Elements Proteins 0.000 description 2
- 208000007097 Urinary Bladder Neoplasms Diseases 0.000 description 2
- 241000021375 Xenogenes Species 0.000 description 2
- 235000011054 acetic acid Nutrition 0.000 description 2
- RJURFGZVJUQBHK-UHFFFAOYSA-N actinomycin D Natural products CC1OC(=O)C(C(C)C)N(C)C(=O)CN(C)C(=O)C2CCCN2C(=O)C(C(C)C)NC(=O)C1NC(=O)C1=C(N)C(=O)C(C)=C2OC(C(C)=CC=C3C(=O)NC4C(=O)NC(C(N5CCCC5C(=O)N(C)CC(=O)N(C)C(C(C)C)C(=O)OC4C)=O)C(C)C)=C3N=C21 RJURFGZVJUQBHK-UHFFFAOYSA-N 0.000 description 2
- 210000000577 adipose tissue Anatomy 0.000 description 2
- 230000000240 adjuvant effect Effects 0.000 description 2
- 230000006838 adverse reaction Effects 0.000 description 2
- 239000011543 agarose gel Substances 0.000 description 2
- PYMYPHUHKUWMLA-LMVFSUKVSA-N aldehydo-D-ribose Chemical compound OC[C@@H](O)[C@@H](O)[C@@H](O)C=O PYMYPHUHKUWMLA-LMVFSUKVSA-N 0.000 description 2
- 229910000147 aluminium phosphate Inorganic materials 0.000 description 2
- 230000002924 anti-infective effect Effects 0.000 description 2
- 230000005904 anticancer immunity Effects 0.000 description 2
- 230000005975 antitumor immune response Effects 0.000 description 2
- VSRXQHXAPYXROS-UHFFFAOYSA-N azanide;cyclobutane-1,1-dicarboxylic acid;platinum(2+) Chemical compound [NH2-].[NH2-].[Pt+2].OC(=O)C1(C(O)=O)CCC1 VSRXQHXAPYXROS-UHFFFAOYSA-N 0.000 description 2
- 230000001580 bacterial effect Effects 0.000 description 2
- 208000010572 basal-like breast carcinoma Diseases 0.000 description 2
- WPYMKLBDIGXBTP-UHFFFAOYSA-N benzoic acid Chemical compound OC(=O)C1=CC=CC=C1 WPYMKLBDIGXBTP-UHFFFAOYSA-N 0.000 description 2
- 230000008827 biological function Effects 0.000 description 2
- 210000004369 blood Anatomy 0.000 description 2
- 239000008280 blood Substances 0.000 description 2
- 210000001124 body fluid Anatomy 0.000 description 2
- 239000010839 body fluid Substances 0.000 description 2
- 230000003139 buffering effect Effects 0.000 description 2
- RFCBNSCSPXMEBK-INFSMZHSSA-N c-GMP-AMP Chemical compound C([C@H]1O2)OP(O)(=O)O[C@H]3[C@@H](O)[C@H](N4C5=NC=NC(N)=C5N=C4)O[C@@H]3COP(O)(=O)O[C@H]1[C@@H](O)[C@@H]2N1C(N=C(NC2=O)N)=C2N=C1 RFCBNSCSPXMEBK-INFSMZHSSA-N 0.000 description 2
- 230000030833 cell death Effects 0.000 description 2
- 230000008859 change Effects 0.000 description 2
- 238000012512 characterization method Methods 0.000 description 2
- 235000013330 chicken meat Nutrition 0.000 description 2
- 238000010367 cloning Methods 0.000 description 2
- 238000003501 co-culture Methods 0.000 description 2
- 239000000084 colloidal system Substances 0.000 description 2
- 238000004590 computer program Methods 0.000 description 2
- 210000000172 cytosol Anatomy 0.000 description 2
- 231100000433 cytotoxic Toxicity 0.000 description 2
- 230000001472 cytotoxic effect Effects 0.000 description 2
- 230000003013 cytotoxicity Effects 0.000 description 2
- 231100000135 cytotoxicity Toxicity 0.000 description 2
- BFMYDTVEBKDAKJ-UHFFFAOYSA-L disodium;(2',7'-dibromo-3',6'-dioxido-3-oxospiro[2-benzofuran-1,9'-xanthene]-4'-yl)mercury;hydrate Chemical compound O.[Na+].[Na+].O1C(=O)C2=CC=CC=C2C21C1=CC(Br)=C([O-])C([Hg])=C1OC1=C2C=C(Br)C([O-])=C1 BFMYDTVEBKDAKJ-UHFFFAOYSA-L 0.000 description 2
- 239000006185 dispersion Substances 0.000 description 2
- 239000002612 dispersion medium Substances 0.000 description 2
- 238000001962 electrophoresis Methods 0.000 description 2
- 238000004520 electroporation Methods 0.000 description 2
- 238000005538 encapsulation Methods 0.000 description 2
- 239000002158 endotoxin Substances 0.000 description 2
- 206010017758 gastric cancer Diseases 0.000 description 2
- 208000005017 glioblastoma Diseases 0.000 description 2
- 230000012010 growth Effects 0.000 description 2
- 229940093915 gynecological organic acid Drugs 0.000 description 2
- 201000010536 head and neck cancer Diseases 0.000 description 2
- 208000014829 head and neck neoplasm Diseases 0.000 description 2
- 239000012274 immune-checkpoint protein inhibitor Substances 0.000 description 2
- 230000036039 immunity Effects 0.000 description 2
- 230000006872 improvement Effects 0.000 description 2
- 238000001802 infusion Methods 0.000 description 2
- 229960003130 interferon gamma Drugs 0.000 description 2
- 229940028885 interleukin-4 Drugs 0.000 description 2
- 238000007912 intraperitoneal administration Methods 0.000 description 2
- 201000010982 kidney cancer Diseases 0.000 description 2
- 230000002147 killing effect Effects 0.000 description 2
- 238000011813 knockout mouse model Methods 0.000 description 2
- 238000002372 labelling Methods 0.000 description 2
- 229920006008 lipopolysaccharide Polymers 0.000 description 2
- 201000007270 liver cancer Diseases 0.000 description 2
- 208000014018 liver neoplasm Diseases 0.000 description 2
- 201000005202 lung cancer Diseases 0.000 description 2
- 208000037841 lung tumor Diseases 0.000 description 2
- 208000015486 malignant pancreatic neoplasm Diseases 0.000 description 2
- 125000000311 mannosyl group Chemical group C1([C@@H](O)[C@@H](O)[C@H](O)[C@H](O1)CO)* 0.000 description 2
- 230000010534 mechanism of action Effects 0.000 description 2
- 239000002679 microRNA Substances 0.000 description 2
- 150000007522 mineralic acids Chemical class 0.000 description 2
- 238000002156 mixing Methods 0.000 description 2
- 229940035032 monophosphoryl lipid a Drugs 0.000 description 2
- 230000007935 neutral effect Effects 0.000 description 2
- 208000002154 non-small cell lung carcinoma Diseases 0.000 description 2
- 239000002777 nucleoside Substances 0.000 description 2
- 125000003835 nucleoside group Chemical group 0.000 description 2
- 150000007524 organic acids Chemical class 0.000 description 2
- 235000005985 organic acids Nutrition 0.000 description 2
- 235000006408 oxalic acid Nutrition 0.000 description 2
- 108700025694 p53 Genes Proteins 0.000 description 2
- 230000000242 pagocytic effect Effects 0.000 description 2
- WLJNZVDCPSBLRP-UHFFFAOYSA-N pamoic acid Chemical compound C1=CC=C2C(CC=3C4=CC=CC=C4C=C(C=3O)C(=O)O)=C(O)C(C(O)=O)=CC2=C1 WLJNZVDCPSBLRP-UHFFFAOYSA-N 0.000 description 2
- 201000002528 pancreatic cancer Diseases 0.000 description 2
- 208000008443 pancreatic carcinoma Diseases 0.000 description 2
- 229920002866 paraformaldehyde Polymers 0.000 description 2
- 230000036961 partial effect Effects 0.000 description 2
- 210000005259 peripheral blood Anatomy 0.000 description 2
- 239000011886 peripheral blood Substances 0.000 description 2
- 230000008782 phagocytosis Effects 0.000 description 2
- 230000000144 pharmacologic effect Effects 0.000 description 2
- BASFCYQUMIYNBI-UHFFFAOYSA-N platinum Chemical compound [Pt] BASFCYQUMIYNBI-UHFFFAOYSA-N 0.000 description 2
- 238000003752 polymerase chain reaction Methods 0.000 description 2
- 238000012545 processing Methods 0.000 description 2
- 239000000047 product Substances 0.000 description 2
- 230000000069 prophylactic effect Effects 0.000 description 2
- 230000002285 radioactive effect Effects 0.000 description 2
- 102000005962 receptors Human genes 0.000 description 2
- 108020003175 receptors Proteins 0.000 description 2
- 230000009467 reduction Effects 0.000 description 2
- 230000002829 reductive effect Effects 0.000 description 2
- 230000010076 replication Effects 0.000 description 2
- 235000019515 salmon Nutrition 0.000 description 2
- 108091029842 small nuclear ribonucleic acid Proteins 0.000 description 2
- 239000011780 sodium chloride Substances 0.000 description 2
- 239000001488 sodium phosphate Substances 0.000 description 2
- 229910000162 sodium phosphate Inorganic materials 0.000 description 2
- 201000011549 stomach cancer Diseases 0.000 description 2
- 235000000346 sugar Nutrition 0.000 description 2
- 239000006228 supernatant Substances 0.000 description 2
- 230000001629 suppression Effects 0.000 description 2
- 239000000725 suspension Substances 0.000 description 2
- 239000011975 tartaric acid Substances 0.000 description 2
- 235000002906 tartaric acid Nutrition 0.000 description 2
- 238000002560 therapeutic procedure Methods 0.000 description 2
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 description 2
- 238000012546 transfer Methods 0.000 description 2
- 238000013519 translation Methods 0.000 description 2
- GETQZCLCWQTVFV-UHFFFAOYSA-N trimethylamine Chemical compound CN(C)C GETQZCLCWQTVFV-UHFFFAOYSA-N 0.000 description 2
- RYFMWSXOAZQYPI-UHFFFAOYSA-K trisodium phosphate Chemical compound [Na+].[Na+].[Na+].[O-]P([O-])([O-])=O RYFMWSXOAZQYPI-UHFFFAOYSA-K 0.000 description 2
- 208000029729 tumor suppressor gene on chromosome 11 Diseases 0.000 description 2
- 201000005112 urinary bladder cancer Diseases 0.000 description 2
- 210000003462 vein Anatomy 0.000 description 2
- 230000009385 viral infection Effects 0.000 description 2
- 230000003442 weekly effect Effects 0.000 description 2
- DGVVWUTYPXICAM-UHFFFAOYSA-N β‐Mercaptoethanol Chemical compound OCCS DGVVWUTYPXICAM-UHFFFAOYSA-N 0.000 description 2
- QBYIENPQHBMVBV-HFEGYEGKSA-N (2R)-2-hydroxy-2-phenylacetic acid Chemical compound O[C@@H](C(O)=O)c1ccccc1.O[C@@H](C(O)=O)c1ccccc1 QBYIENPQHBMVBV-HFEGYEGKSA-N 0.000 description 1
- BJEPYKJPYRNKOW-REOHCLBHSA-N (S)-malic acid Chemical compound OC(=O)[C@@H](O)CC(O)=O BJEPYKJPYRNKOW-REOHCLBHSA-N 0.000 description 1
- 102000040650 (ribonucleotides)n+m Human genes 0.000 description 1
- QFMZQPDHXULLKC-UHFFFAOYSA-N 1,2-bis(diphenylphosphino)ethane Chemical compound C=1C=CC=CC=1P(C=1C=CC=CC=1)CCP(C=1C=CC=CC=1)C1=CC=CC=C1 QFMZQPDHXULLKC-UHFFFAOYSA-N 0.000 description 1
- KILNVBDSWZSGLL-KXQOOQHDSA-N 1,2-dihexadecanoyl-sn-glycero-3-phosphocholine Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCCCCCCCCCCCCCC KILNVBDSWZSGLL-KXQOOQHDSA-N 0.000 description 1
- SLKDGVPOSSLUAI-PGUFJCEWSA-N 1,2-dihexadecanoyl-sn-glycero-3-phosphoethanolamine zwitterion Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](COP(O)(=O)OCCN)OC(=O)CCCCCCCCCCCCCCC SLKDGVPOSSLUAI-PGUFJCEWSA-N 0.000 description 1
- TUSDEZXZIZRFGC-UHFFFAOYSA-N 1-O-galloyl-3,6-(R)-HHDP-beta-D-glucose Natural products OC1C(O2)COC(=O)C3=CC(O)=C(O)C(O)=C3C3=C(O)C(O)=C(O)C=C3C(=O)OC1C(O)C2OC(=O)C1=CC(O)=C(O)C(O)=C1 TUSDEZXZIZRFGC-UHFFFAOYSA-N 0.000 description 1
- LNETULKMXZVUST-UHFFFAOYSA-N 1-naphthoic acid Chemical compound C1=CC=C2C(C(=O)O)=CC=CC2=C1 LNETULKMXZVUST-UHFFFAOYSA-N 0.000 description 1
- NHBKXEKEPDILRR-UHFFFAOYSA-N 2,3-bis(butanoylsulfanyl)propyl butanoate Chemical compound CCCC(=O)OCC(SC(=O)CCC)CSC(=O)CCC NHBKXEKEPDILRR-UHFFFAOYSA-N 0.000 description 1
- NIQFAJBKEHPUAM-UHFFFAOYSA-N 2-[2-[2-(2-aminoethoxy)ethoxy]ethoxy]ethanamine Chemical compound NCCOCCOCCOCCN NIQFAJBKEHPUAM-UHFFFAOYSA-N 0.000 description 1
- OSBLTNPMIGYQGY-UHFFFAOYSA-N 2-amino-2-(hydroxymethyl)propane-1,3-diol;2-[2-[bis(carboxymethyl)amino]ethyl-(carboxymethyl)amino]acetic acid;boric acid Chemical compound OB(O)O.OCC(N)(CO)CO.OC(=O)CN(CC(O)=O)CCN(CC(O)=O)CC(O)=O OSBLTNPMIGYQGY-UHFFFAOYSA-N 0.000 description 1
- QKNYBSVHEMOAJP-UHFFFAOYSA-N 2-amino-2-(hydroxymethyl)propane-1,3-diol;hydron;chloride Chemical compound Cl.OCC(N)(CO)CO QKNYBSVHEMOAJP-UHFFFAOYSA-N 0.000 description 1
- ASJSAQIRZKANQN-CRCLSJGQSA-N 2-deoxy-D-ribose Chemical compound OC[C@@H](O)[C@@H](O)CC=O ASJSAQIRZKANQN-CRCLSJGQSA-N 0.000 description 1
- BMYNFMYTOJXKLE-UHFFFAOYSA-N 3-azaniumyl-2-hydroxypropanoate Chemical compound NCC(O)C(O)=O BMYNFMYTOJXKLE-UHFFFAOYSA-N 0.000 description 1
- JHWGFJBTMHEZME-UHFFFAOYSA-N 4-prop-2-enoyloxybutyl prop-2-enoate Chemical compound C=CC(=O)OCCCCOC(=O)C=C JHWGFJBTMHEZME-UHFFFAOYSA-N 0.000 description 1
- LQGKDMHENBFVRC-UHFFFAOYSA-N 5-aminopentan-1-ol Chemical compound NCCCCCO LQGKDMHENBFVRC-UHFFFAOYSA-N 0.000 description 1
- 102100038222 60 kDa heat shock protein, mitochondrial Human genes 0.000 description 1
- STQGQHZAVUOBTE-UHFFFAOYSA-N 7-Cyan-hept-2t-en-4,6-diinsaeure Natural products C1=2C(O)=C3C(=O)C=4C(OC)=CC=CC=4C(=O)C3=C(O)C=2CC(O)(C(C)=O)CC1OC1CC(N)C(O)C(C)O1 STQGQHZAVUOBTE-UHFFFAOYSA-N 0.000 description 1
- 208000035657 Abasia Diseases 0.000 description 1
- 206010069754 Acquired gene mutation Diseases 0.000 description 1
- 102000002260 Alkaline Phosphatase Human genes 0.000 description 1
- 108020004774 Alkaline Phosphatase Proteins 0.000 description 1
- VHUUQVKOLVNVRT-UHFFFAOYSA-N Ammonium hydroxide Chemical compound [NH4+].[OH-] VHUUQVKOLVNVRT-UHFFFAOYSA-N 0.000 description 1
- 108090001008 Avidin Proteins 0.000 description 1
- 239000005711 Benzoic acid Substances 0.000 description 1
- 108091003079 Bovine Serum Albumin Proteins 0.000 description 1
- 241001598984 Bromius obscurus Species 0.000 description 1
- 238000010354 CRISPR gene editing Methods 0.000 description 1
- 229940045513 CTLA4 antagonist Drugs 0.000 description 1
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 1
- 241000283707 Capra Species 0.000 description 1
- 208000024172 Cardiovascular disease Diseases 0.000 description 1
- 102000014914 Carrier Proteins Human genes 0.000 description 1
- 241000700198 Cavia Species 0.000 description 1
- 102000000844 Cell Surface Receptors Human genes 0.000 description 1
- 108010001857 Cell Surface Receptors Proteins 0.000 description 1
- 241000282693 Cercopithecidae Species 0.000 description 1
- 108010058432 Chaperonin 60 Proteins 0.000 description 1
- 108020004705 Codon Proteins 0.000 description 1
- 206010009944 Colon cancer Diseases 0.000 description 1
- 208000001333 Colorectal Neoplasms Diseases 0.000 description 1
- 206010052358 Colorectal cancer metastatic Diseases 0.000 description 1
- 108091035707 Consensus sequence Proteins 0.000 description 1
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 1
- 241001137251 Corvidae Species 0.000 description 1
- 241000699800 Cricetinae Species 0.000 description 1
- IGXWBGJHJZYPQS-SSDOTTSWSA-N D-Luciferin Chemical compound OC(=O)[C@H]1CSC(C=2SC3=CC=C(O)C=C3N=2)=N1 IGXWBGJHJZYPQS-SSDOTTSWSA-N 0.000 description 1
- RGHNJXZEOKUKBD-UHFFFAOYSA-N D-gluconic acid Natural products OCC(O)C(O)C(O)C(O)C(O)=O RGHNJXZEOKUKBD-UHFFFAOYSA-N 0.000 description 1
- 108010092160 Dactinomycin Proteins 0.000 description 1
- 108010016626 Dipeptides Proteins 0.000 description 1
- 238000008157 ELISA kit Methods 0.000 description 1
- 101150029707 ERBB2 gene Proteins 0.000 description 1
- 102100025137 Early activation antigen CD69 Human genes 0.000 description 1
- 238000011510 Elispot assay Methods 0.000 description 1
- 101100001675 Emericella variicolor andJ gene Proteins 0.000 description 1
- PIICEJLVQHRZGT-UHFFFAOYSA-N Ethylenediamine Chemical compound NCCN PIICEJLVQHRZGT-UHFFFAOYSA-N 0.000 description 1
- 239000001263 FEMA 3042 Substances 0.000 description 1
- 108091092584 GDNA Proteins 0.000 description 1
- 206010064571 Gene mutation Diseases 0.000 description 1
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 1
- 206010019280 Heart failures Diseases 0.000 description 1
- 102400001369 Heparin-binding EGF-like growth factor Human genes 0.000 description 1
- 101800001649 Heparin-binding EGF-like growth factor Proteins 0.000 description 1
- 241001272567 Hominoidea Species 0.000 description 1
- 101000934374 Homo sapiens Early activation antigen CD69 Proteins 0.000 description 1
- 101000738771 Homo sapiens Receptor-type tyrosine-protein phosphatase C Proteins 0.000 description 1
- 102000003839 Human Proteins Human genes 0.000 description 1
- 229920002153 Hydroxypropyl cellulose Polymers 0.000 description 1
- 102000014150 Interferons Human genes 0.000 description 1
- 108010050904 Interferons Proteins 0.000 description 1
- 108090001005 Interleukin-6 Proteins 0.000 description 1
- 102000004889 Interleukin-6 Human genes 0.000 description 1
- ODKSFYDXXFIFQN-BYPYZUCNSA-P L-argininium(2+) Chemical compound NC(=[NH2+])NCCC[C@H]([NH3+])C(O)=O ODKSFYDXXFIFQN-BYPYZUCNSA-P 0.000 description 1
- LEVWYRKDKASIDU-IMJSIDKUSA-N L-cystine Chemical compound [O-]C(=O)[C@@H]([NH3+])CSSC[C@H]([NH3+])C([O-])=O LEVWYRKDKASIDU-IMJSIDKUSA-N 0.000 description 1
- HNDVDQJCIGZPNO-YFKPBYRVSA-N L-histidine Chemical compound OC(=O)[C@@H](N)CC1=CN=CN1 HNDVDQJCIGZPNO-YFKPBYRVSA-N 0.000 description 1
- 102100026358 Lipoma-preferred partner Human genes 0.000 description 1
- 102000008072 Lymphokines Human genes 0.000 description 1
- 108010074338 Lymphokines Proteins 0.000 description 1
- 206010025323 Lymphomas Diseases 0.000 description 1
- 210000004322 M2 macrophage Anatomy 0.000 description 1
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 1
- 108010031099 Mannose Receptor Proteins 0.000 description 1
- 239000012901 Milli-Q water Substances 0.000 description 1
- 102000013967 Monokines Human genes 0.000 description 1
- 108010050619 Monokines Proteins 0.000 description 1
- 108090000143 Mouse Proteins Proteins 0.000 description 1
- 208000034578 Multiple myelomas Diseases 0.000 description 1
- 102000010168 Myeloid Differentiation Factor 88 Human genes 0.000 description 1
- 108010077432 Myeloid Differentiation Factor 88 Proteins 0.000 description 1
- 229930182474 N-glycoside Natural products 0.000 description 1
- GRYLNZFGIOXLOG-UHFFFAOYSA-N Nitric acid Chemical compound O[N+]([O-])=O GRYLNZFGIOXLOG-UHFFFAOYSA-N 0.000 description 1
- 101710163270 Nuclease Proteins 0.000 description 1
- 108010038807 Oligopeptides Proteins 0.000 description 1
- 102000015636 Oligopeptides Human genes 0.000 description 1
- 102000043276 Oncogene Human genes 0.000 description 1
- 108700026244 Open Reading Frames Proteins 0.000 description 1
- 238000010222 PCR analysis Methods 0.000 description 1
- 241000282579 Pan Species 0.000 description 1
- 229920002230 Pectic acid Polymers 0.000 description 1
- LRBQNJMCXXYXIU-PPKXGCFTSA-N Penta-digallate-beta-D-glucose Natural products OC1=C(O)C(O)=CC(C(=O)OC=2C(=C(O)C=C(C=2)C(=O)OC[C@@H]2[C@H]([C@H](OC(=O)C=3C=C(OC(=O)C=4C=C(O)C(O)=C(O)C=4)C(O)=C(O)C=3)[C@@H](OC(=O)C=3C=C(OC(=O)C=4C=C(O)C(O)=C(O)C=4)C(O)=C(O)C=3)[C@H](OC(=O)C=3C=C(OC(=O)C=4C=C(O)C(O)=C(O)C=4)C(O)=C(O)C=3)O2)OC(=O)C=2C=C(OC(=O)C=3C=C(O)C(O)=C(O)C=3)C(O)=C(O)C=2)O)=C1 LRBQNJMCXXYXIU-PPKXGCFTSA-N 0.000 description 1
- 108010033276 Peptide Fragments Proteins 0.000 description 1
- 102000007079 Peptide Fragments Human genes 0.000 description 1
- 108010038988 Peptide Hormones Proteins 0.000 description 1
- 102000015731 Peptide Hormones Human genes 0.000 description 1
- 102000003992 Peroxidases Human genes 0.000 description 1
- 206010035226 Plasma cell myeloma Diseases 0.000 description 1
- 108010020346 Polyglutamic Acid Proteins 0.000 description 1
- 108010039918 Polylysine Proteins 0.000 description 1
- 241000282405 Pongo abelii Species 0.000 description 1
- OFOBLEOULBTSOW-UHFFFAOYSA-N Propanedioic acid Natural products OC(=O)CC(O)=O OFOBLEOULBTSOW-UHFFFAOYSA-N 0.000 description 1
- 102000004245 Proteasome Endopeptidase Complex Human genes 0.000 description 1
- 108090000708 Proteasome Endopeptidase Complex Proteins 0.000 description 1
- IWYDHOAUDWTVEP-UHFFFAOYSA-N R-2-phenyl-2-hydroxyacetic acid Natural products OC(=O)C(O)C1=CC=CC=C1 IWYDHOAUDWTVEP-UHFFFAOYSA-N 0.000 description 1
- 102100037422 Receptor-type tyrosine-protein phosphatase C Human genes 0.000 description 1
- 108020004511 Recombinant DNA Proteins 0.000 description 1
- 108010008281 Recombinant Fusion Proteins Proteins 0.000 description 1
- 102000007056 Recombinant Fusion Proteins Human genes 0.000 description 1
- 206010038111 Recurrent cancer Diseases 0.000 description 1
- 206010070308 Refractory cancer Diseases 0.000 description 1
- 108091027981 Response element Proteins 0.000 description 1
- 241000283984 Rodentia Species 0.000 description 1
- 239000006146 Roswell Park Memorial Institute medium Substances 0.000 description 1
- 108020004682 Single-Stranded DNA Proteins 0.000 description 1
- VMHLLURERBWHNL-UHFFFAOYSA-M Sodium acetate Chemical compound [Na+].CC([O-])=O VMHLLURERBWHNL-UHFFFAOYSA-M 0.000 description 1
- 238000000692 Student's t-test Methods 0.000 description 1
- KDYFGRWQOYBRFD-UHFFFAOYSA-N Succinic acid Natural products OC(=O)CCC(O)=O KDYFGRWQOYBRFD-UHFFFAOYSA-N 0.000 description 1
- 230000005867 T cell response Effects 0.000 description 1
- 239000008051 TBE buffer Substances 0.000 description 1
- 238000003917 TEM image Methods 0.000 description 1
- 239000004809 Teflon Substances 0.000 description 1
- 229920006362 Teflon® Polymers 0.000 description 1
- RYYWUUFWQRZTIU-UHFFFAOYSA-N Thiophosphoric acid Chemical class OP(O)(S)=O RYYWUUFWQRZTIU-UHFFFAOYSA-N 0.000 description 1
- 108091036066 Three prime untranslated region Proteins 0.000 description 1
- 229940125970 Toll-Like Receptor inhibitor Drugs 0.000 description 1
- 206010066901 Treatment failure Diseases 0.000 description 1
- 208000003721 Triple Negative Breast Neoplasms Diseases 0.000 description 1
- 206010050283 Tumour ulceration Diseases 0.000 description 1
- 108010046334 Urease Proteins 0.000 description 1
- 229940122803 Vinca alkaloid Drugs 0.000 description 1
- 208000036142 Viral infection Diseases 0.000 description 1
- 241000710886 West Nile virus Species 0.000 description 1
- HCHKCACWOHOZIP-UHFFFAOYSA-N Zinc Chemical compound [Zn] HCHKCACWOHOZIP-UHFFFAOYSA-N 0.000 description 1
- 238000009825 accumulation Methods 0.000 description 1
- 230000021736 acetylation Effects 0.000 description 1
- 238000006640 acetylation reaction Methods 0.000 description 1
- RJURFGZVJUQBHK-IIXSONLDSA-N actinomycin D Chemical compound C[C@H]1OC(=O)[C@H](C(C)C)N(C)C(=O)CN(C)C(=O)[C@@H]2CCCN2C(=O)[C@@H](C(C)C)NC(=O)[C@H]1NC(=O)C1=C(N)C(=O)C(C)=C2OC(C(C)=CC=C3C(=O)N[C@@H]4C(=O)N[C@@H](C(N5CCC[C@H]5C(=O)N(C)CC(=O)N(C)[C@@H](C(C)C)C(=O)O[C@@H]4C)=O)C(C)C)=C3N=C21 RJURFGZVJUQBHK-IIXSONLDSA-N 0.000 description 1
- 238000000516 activation analysis Methods 0.000 description 1
- 239000012190 activator Substances 0.000 description 1
- 230000033289 adaptive immune response Effects 0.000 description 1
- 238000000246 agarose gel electrophoresis Methods 0.000 description 1
- 239000000783 alginic acid Substances 0.000 description 1
- 229920000615 alginic acid Polymers 0.000 description 1
- 235000010443 alginic acid Nutrition 0.000 description 1
- 229960001126 alginic acid Drugs 0.000 description 1
- 150000004781 alginic acids Chemical class 0.000 description 1
- 239000003513 alkali Substances 0.000 description 1
- 230000000172 allergic effect Effects 0.000 description 1
- BJEPYKJPYRNKOW-UHFFFAOYSA-N alpha-hydroxysuccinic acid Natural products OC(=O)C(O)CC(O)=O BJEPYKJPYRNKOW-UHFFFAOYSA-N 0.000 description 1
- 229910052782 aluminium Inorganic materials 0.000 description 1
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 1
- 230000009435 amidation Effects 0.000 description 1
- 238000007112 amidation reaction Methods 0.000 description 1
- 125000003277 amino group Chemical group 0.000 description 1
- 239000000908 ammonium hydroxide Substances 0.000 description 1
- 230000003321 amplification Effects 0.000 description 1
- 239000002269 analeptic agent Substances 0.000 description 1
- 229940045799 anthracyclines and related substance Drugs 0.000 description 1
- 230000006023 anti-tumor response Effects 0.000 description 1
- 230000005875 antibody response Effects 0.000 description 1
- 230000014102 antigen processing and presentation of exogenous peptide antigen via MHC class I Effects 0.000 description 1
- 239000012736 aqueous medium Substances 0.000 description 1
- PYMYPHUHKUWMLA-UHFFFAOYSA-N arabinose Natural products OCC(O)C(O)C(O)C=O PYMYPHUHKUWMLA-UHFFFAOYSA-N 0.000 description 1
- 229910052785 arsenic Inorganic materials 0.000 description 1
- 239000011668 ascorbic acid Substances 0.000 description 1
- 229960005070 ascorbic acid Drugs 0.000 description 1
- 235000010323 ascorbic acid Nutrition 0.000 description 1
- 235000003704 aspartic acid Nutrition 0.000 description 1
- 208000010668 atopic eczema Diseases 0.000 description 1
- 229910052788 barium Inorganic materials 0.000 description 1
- DSAJWYNOEDNPEQ-UHFFFAOYSA-N barium atom Chemical compound [Ba] DSAJWYNOEDNPEQ-UHFFFAOYSA-N 0.000 description 1
- 235000010233 benzoic acid Nutrition 0.000 description 1
- SRBFZHDQGSBBOR-UHFFFAOYSA-N beta-D-Pyranose-Lyxose Natural products OC1COC(O)C(O)C1O SRBFZHDQGSBBOR-UHFFFAOYSA-N 0.000 description 1
- WQZGKKKJIJFFOK-VFUOTHLCSA-N beta-D-glucose Chemical compound OC[C@H]1O[C@@H](O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-VFUOTHLCSA-N 0.000 description 1
- OQFSQFPPLPISGP-UHFFFAOYSA-N beta-carboxyaspartic acid Natural products OC(=O)C(N)C(C(O)=O)C(O)=O OQFSQFPPLPISGP-UHFFFAOYSA-N 0.000 description 1
- 108091008324 binding proteins Chemical group 0.000 description 1
- 239000012472 biological sample Substances 0.000 description 1
- 229960002685 biotin Drugs 0.000 description 1
- 235000020958 biotin Nutrition 0.000 description 1
- 239000011616 biotin Substances 0.000 description 1
- 229910052797 bismuth Inorganic materials 0.000 description 1
- JCXGWMGPZLAOME-UHFFFAOYSA-N bismuth atom Chemical compound [Bi] JCXGWMGPZLAOME-UHFFFAOYSA-N 0.000 description 1
- 230000000903 blocking effect Effects 0.000 description 1
- 230000037396 body weight Effects 0.000 description 1
- 210000002798 bone marrow cell Anatomy 0.000 description 1
- 210000000481 breast Anatomy 0.000 description 1
- 239000007853 buffer solution Substances 0.000 description 1
- KDYFGRWQOYBRFD-NUQCWPJISA-N butanedioic acid Chemical compound O[14C](=O)CC[14C](O)=O KDYFGRWQOYBRFD-NUQCWPJISA-N 0.000 description 1
- 210000004899 c-terminal region Anatomy 0.000 description 1
- 229910052793 cadmium Inorganic materials 0.000 description 1
- BDOSMKKIYDKNTQ-UHFFFAOYSA-N cadmium atom Chemical compound [Cd] BDOSMKKIYDKNTQ-UHFFFAOYSA-N 0.000 description 1
- 229910052791 calcium Inorganic materials 0.000 description 1
- 239000011575 calcium Substances 0.000 description 1
- AXCZMVOFGPJBDE-UHFFFAOYSA-L calcium dihydroxide Chemical compound [OH-].[OH-].[Ca+2] AXCZMVOFGPJBDE-UHFFFAOYSA-L 0.000 description 1
- 239000000920 calcium hydroxide Substances 0.000 description 1
- 229910001861 calcium hydroxide Inorganic materials 0.000 description 1
- 239000002775 capsule Substances 0.000 description 1
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 1
- 125000002091 cationic group Chemical group 0.000 description 1
- 150000001768 cations Chemical class 0.000 description 1
- 230000027448 caveolin-mediated endocytosis Effects 0.000 description 1
- 238000004113 cell culture Methods 0.000 description 1
- 230000003915 cell function Effects 0.000 description 1
- 230000011748 cell maturation Effects 0.000 description 1
- 210000000170 cell membrane Anatomy 0.000 description 1
- 230000004663 cell proliferation Effects 0.000 description 1
- 108091092328 cellular RNA Proteins 0.000 description 1
- 238000006243 chemical reaction Methods 0.000 description 1
- 238000002512 chemotherapy Methods 0.000 description 1
- 210000000349 chromosome Anatomy 0.000 description 1
- 235000015165 citric acid Nutrition 0.000 description 1
- 210000003690 classically activated macrophage Anatomy 0.000 description 1
- 230000006395 clathrin-mediated endocytosis Effects 0.000 description 1
- 238000003776 cleavage reaction Methods 0.000 description 1
- 239000011248 coating agent Substances 0.000 description 1
- 238000000576 coating method Methods 0.000 description 1
- 229910017052 cobalt Inorganic materials 0.000 description 1
- 239000010941 cobalt Substances 0.000 description 1
- GUTLYIVDDKVIGB-UHFFFAOYSA-N cobalt atom Chemical compound [Co] GUTLYIVDDKVIGB-UHFFFAOYSA-N 0.000 description 1
- 230000001268 conjugating effect Effects 0.000 description 1
- 230000021615 conjugation Effects 0.000 description 1
- 238000007796 conventional method Methods 0.000 description 1
- 229910052802 copper Inorganic materials 0.000 description 1
- 239000010949 copper Substances 0.000 description 1
- 230000002596 correlated effect Effects 0.000 description 1
- 238000012258 culturing Methods 0.000 description 1
- 229960003067 cystine Drugs 0.000 description 1
- 230000001086 cytosolic effect Effects 0.000 description 1
- 229960000640 dactinomycin Drugs 0.000 description 1
- 230000006378 damage Effects 0.000 description 1
- 229960000975 daunorubicin Drugs 0.000 description 1
- STQGQHZAVUOBTE-VGBVRHCVSA-N daunorubicin Chemical compound O([C@H]1C[C@@](O)(CC=2C(O)=C3C(=O)C=4C=CC=C(C=4C(=O)C3=C(O)C=21)OC)C(C)=O)[C@H]1C[C@H](N)[C@H](O)[C@H](C)O1 STQGQHZAVUOBTE-VGBVRHCVSA-N 0.000 description 1
- 230000034994 death Effects 0.000 description 1
- 231100000517 death Toxicity 0.000 description 1
- 230000003111 delayed effect Effects 0.000 description 1
- 238000012217 deletion Methods 0.000 description 1
- 230000037430 deletion Effects 0.000 description 1
- 239000005549 deoxyribonucleoside Substances 0.000 description 1
- 238000001212 derivatisation Methods 0.000 description 1
- 238000013461 design Methods 0.000 description 1
- 239000000032 diagnostic agent Substances 0.000 description 1
- 229940039227 diagnostic agent Drugs 0.000 description 1
- 238000010586 diagram Methods 0.000 description 1
- 238000000502 dialysis Methods 0.000 description 1
- 230000006334 disulfide bridging Effects 0.000 description 1
- 239000002552 dosage form Substances 0.000 description 1
- 230000037437 driver mutation Effects 0.000 description 1
- 230000009977 dual effect Effects 0.000 description 1
- 238000002296 dynamic light scattering Methods 0.000 description 1
- 230000002500 effect on skin Effects 0.000 description 1
- 210000003162 effector t lymphocyte Anatomy 0.000 description 1
- 235000013601 eggs Nutrition 0.000 description 1
- 230000009881 electrostatic interaction Effects 0.000 description 1
- 230000012202 endocytosis Effects 0.000 description 1
- 210000001163 endosome Anatomy 0.000 description 1
- 230000007515 enzymatic degradation Effects 0.000 description 1
- 210000003743 erythrocyte Anatomy 0.000 description 1
- 238000011156 evaluation Methods 0.000 description 1
- 230000005284 excitation Effects 0.000 description 1
- 210000001808 exosome Anatomy 0.000 description 1
- 229960004887 ferric hydroxide Drugs 0.000 description 1
- 239000012458 free base Substances 0.000 description 1
- 239000001530 fumaric acid Substances 0.000 description 1
- 235000011087 fumaric acid Nutrition 0.000 description 1
- 230000005714 functional activity Effects 0.000 description 1
- LRBQNJMCXXYXIU-QWKBTXIPSA-N gallotannic acid Chemical compound OC1=C(O)C(O)=CC(C(=O)OC=2C(=C(O)C=C(C=2)C(=O)OC[C@H]2[C@@H]([C@@H](OC(=O)C=3C=C(OC(=O)C=4C=C(O)C(O)=C(O)C=4)C(O)=C(O)C=3)[C@H](OC(=O)C=3C=C(OC(=O)C=4C=C(O)C(O)=C(O)C=4)C(O)=C(O)C=3)[C@@H](OC(=O)C=3C=C(OC(=O)C=4C=C(O)C(O)=C(O)C=4)C(O)=C(O)C=3)O2)OC(=O)C=2C=C(OC(=O)C=3C=C(O)C(O)=C(O)C=3)C(O)=C(O)C=2)O)=C1 LRBQNJMCXXYXIU-QWKBTXIPSA-N 0.000 description 1
- 238000007429 general method Methods 0.000 description 1
- 238000012268 genome sequencing Methods 0.000 description 1
- 239000011521 glass Substances 0.000 description 1
- 239000000174 gluconic acid Substances 0.000 description 1
- 235000012208 gluconic acid Nutrition 0.000 description 1
- 239000008103 glucose Substances 0.000 description 1
- 235000013922 glutamic acid Nutrition 0.000 description 1
- 239000004220 glutamic acid Substances 0.000 description 1
- 230000013595 glycosylation Effects 0.000 description 1
- 238000006206 glycosylation reaction Methods 0.000 description 1
- 208000019622 heart disease Diseases 0.000 description 1
- 244000144980 herd Species 0.000 description 1
- 210000005260 human cell Anatomy 0.000 description 1
- 239000000017 hydrogel Substances 0.000 description 1
- 239000001257 hydrogen Substances 0.000 description 1
- 229910052739 hydrogen Inorganic materials 0.000 description 1
- 239000001863 hydroxypropyl cellulose Substances 0.000 description 1
- 235000010977 hydroxypropyl cellulose Nutrition 0.000 description 1
- 239000012216 imaging agent Substances 0.000 description 1
- 230000003053 immunization Effects 0.000 description 1
- 238000002649 immunization Methods 0.000 description 1
- 230000005847 immunogenicity Effects 0.000 description 1
- 239000002955 immunomodulating agent Substances 0.000 description 1
- 239000003018 immunosuppressive agent Substances 0.000 description 1
- 229940125721 immunosuppressive agent Drugs 0.000 description 1
- 238000011065 in-situ storage Methods 0.000 description 1
- 230000006698 induction Effects 0.000 description 1
- 239000012678 infectious agent Substances 0.000 description 1
- 208000015181 infectious disease Diseases 0.000 description 1
- 108091006086 inhibitor proteins Proteins 0.000 description 1
- 239000007972 injectable composition Substances 0.000 description 1
- 208000014674 injury Diseases 0.000 description 1
- 150000007529 inorganic bases Chemical class 0.000 description 1
- 238000003780 insertion Methods 0.000 description 1
- 230000037431 insertion Effects 0.000 description 1
- 230000002452 interceptive effect Effects 0.000 description 1
- 229940079322 interferon Drugs 0.000 description 1
- 230000003834 intracellular effect Effects 0.000 description 1
- 230000002601 intratumoral effect Effects 0.000 description 1
- IEECXTSVVFWGSE-UHFFFAOYSA-M iron(3+);oxygen(2-);hydroxide Chemical compound [OH-].[O-2].[Fe+3] IEECXTSVVFWGSE-UHFFFAOYSA-M 0.000 description 1
- JJWLVOIRVHMVIS-UHFFFAOYSA-N isopropylamine Chemical compound CC(C)N JJWLVOIRVHMVIS-UHFFFAOYSA-N 0.000 description 1
- 239000000644 isotonic solution Substances 0.000 description 1
- 230000003902 lesion Effects 0.000 description 1
- 231100001231 less toxic Toxicity 0.000 description 1
- 208000032839 leukemia Diseases 0.000 description 1
- 210000000265 leukocyte Anatomy 0.000 description 1
- 239000003446 ligand Substances 0.000 description 1
- 239000006193 liquid solution Substances 0.000 description 1
- 210000004185 liver Anatomy 0.000 description 1
- 238000011068 loading method Methods 0.000 description 1
- 238000007422 luminescence assay Methods 0.000 description 1
- 239000012139 lysis buffer Substances 0.000 description 1
- 239000011777 magnesium Substances 0.000 description 1
- 229910052749 magnesium Inorganic materials 0.000 description 1
- VZCYOOQTPOCHFL-UPHRSURJSA-N maleic acid Chemical compound OC(=O)\C=C/C(O)=O VZCYOOQTPOCHFL-UPHRSURJSA-N 0.000 description 1
- 239000011976 maleic acid Substances 0.000 description 1
- 239000001630 malic acid Substances 0.000 description 1
- 235000011090 malic acid Nutrition 0.000 description 1
- 230000036210 malignancy Effects 0.000 description 1
- 238000007726 management method Methods 0.000 description 1
- 229960002510 mandelic acid Drugs 0.000 description 1
- 239000011159 matrix material Substances 0.000 description 1
- 229910052751 metal Inorganic materials 0.000 description 1
- 239000002184 metal Substances 0.000 description 1
- YACKEPLHDIMKIO-UHFFFAOYSA-N methylphosphonic acid Chemical class CP(O)(O)=O YACKEPLHDIMKIO-UHFFFAOYSA-N 0.000 description 1
- 230000000813 microbial effect Effects 0.000 description 1
- 239000004005 microsphere Substances 0.000 description 1
- 230000001617 migratory effect Effects 0.000 description 1
- 239000000178 monomer Substances 0.000 description 1
- 230000036457 multidrug resistance Effects 0.000 description 1
- 238000002703 mutagenesis Methods 0.000 description 1
- 231100000350 mutagenesis Toxicity 0.000 description 1
- VMGAPWLDMVPYIA-HIDZBRGKSA-N n'-amino-n-iminomethanimidamide Chemical compound N\N=C\N=N VMGAPWLDMVPYIA-HIDZBRGKSA-N 0.000 description 1
- ACYBVNYNIZTUIL-UHFFFAOYSA-N n'-benzylethane-1,2-diamine Chemical compound NCCNCC1=CC=CC=C1 ACYBVNYNIZTUIL-UHFFFAOYSA-N 0.000 description 1
- OHDXDNUPVVYWOV-UHFFFAOYSA-N n-methyl-1-(2-naphthalen-1-ylsulfanylphenyl)methanamine Chemical compound CNCC1=CC=CC=C1SC1=CC=CC2=CC=CC=C12 OHDXDNUPVVYWOV-UHFFFAOYSA-N 0.000 description 1
- 239000002077 nanosphere Substances 0.000 description 1
- YZMHQCWXYHARLS-UHFFFAOYSA-N naphthalene-1,2-disulfonic acid Chemical compound C1=CC=CC2=C(S(O)(=O)=O)C(S(=O)(=O)O)=CC=C21 YZMHQCWXYHARLS-UHFFFAOYSA-N 0.000 description 1
- PSZYNBSKGUBXEH-UHFFFAOYSA-N naphthalene-1-sulfonic acid Chemical compound C1=CC=C2C(S(=O)(=O)O)=CC=CC2=C1 PSZYNBSKGUBXEH-UHFFFAOYSA-N 0.000 description 1
- 230000012106 negative regulation of microtubule depolymerization Effects 0.000 description 1
- 229910052759 nickel Inorganic materials 0.000 description 1
- 229910017604 nitric acid Inorganic materials 0.000 description 1
- 229910052757 nitrogen Inorganic materials 0.000 description 1
- 230000005937 nuclear translocation Effects 0.000 description 1
- 238000003199 nucleic acid amplification method Methods 0.000 description 1
- 230000001293 nucleolytic effect Effects 0.000 description 1
- 108010038765 octaarginine Proteins 0.000 description 1
- 239000003921 oil Substances 0.000 description 1
- 229940046166 oligodeoxynucleotide Drugs 0.000 description 1
- 238000011275 oncology therapy Methods 0.000 description 1
- 150000007530 organic bases Chemical class 0.000 description 1
- 150000002892 organic cations Chemical class 0.000 description 1
- 230000020477 pH reduction Effects 0.000 description 1
- 244000052769 pathogen Species 0.000 description 1
- 230000037361 pathway Effects 0.000 description 1
- 239000000813 peptide hormone Substances 0.000 description 1
- 108040007629 peroxidase activity proteins Proteins 0.000 description 1
- 150000004713 phosphodiesters Chemical class 0.000 description 1
- 230000026731 phosphorylation Effects 0.000 description 1
- 238000006366 phosphorylation reaction Methods 0.000 description 1
- 238000005375 photometry Methods 0.000 description 1
- 230000036470 plasma concentration Effects 0.000 description 1
- 230000008488 polyadenylation Effects 0.000 description 1
- 108010011110 polyarginine Proteins 0.000 description 1
- 239000010318 polygalacturonic acid Substances 0.000 description 1
- 229920002643 polyglutamic acid Polymers 0.000 description 1
- 229920000656 polylysine Polymers 0.000 description 1
- 229920002959 polymer blend Polymers 0.000 description 1
- 229920001282 polysaccharide Polymers 0.000 description 1
- 239000005017 polysaccharide Substances 0.000 description 1
- 150000004804 polysaccharides Chemical class 0.000 description 1
- 238000010837 poor prognosis Methods 0.000 description 1
- 239000013641 positive control Substances 0.000 description 1
- 230000025613 positive regulation of adaptive immune response Effects 0.000 description 1
- 230000004481 post-translational protein modification Effects 0.000 description 1
- 230000001124 posttranscriptional effect Effects 0.000 description 1
- 230000001323 posttranslational effect Effects 0.000 description 1
- 230000004537 potential cytotoxicity Effects 0.000 description 1
- 239000003755 preservative agent Substances 0.000 description 1
- 230000002335 preservative effect Effects 0.000 description 1
- MFDFERRIHVXMIY-UHFFFAOYSA-N procaine Chemical compound CCN(CC)CCOC(=O)C1=CC=C(N)C=C1 MFDFERRIHVXMIY-UHFFFAOYSA-N 0.000 description 1
- 229960004919 procaine Drugs 0.000 description 1
- 229940002612 prodrug Drugs 0.000 description 1
- 239000000651 prodrug Substances 0.000 description 1
- 102000003998 progesterone receptors Human genes 0.000 description 1
- 108090000468 progesterone receptors Proteins 0.000 description 1
- 238000004393 prognosis Methods 0.000 description 1
- 230000000770 proinflammatory effect Effects 0.000 description 1
- 230000002062 proliferating effect Effects 0.000 description 1
- 229940021993 prophylactic vaccine Drugs 0.000 description 1
- 238000011321 prophylaxis Methods 0.000 description 1
- 229950008679 protamine sulfate Drugs 0.000 description 1
- 230000001681 protective effect Effects 0.000 description 1
- 238000001243 protein synthesis Methods 0.000 description 1
- 229940023143 protein vaccine Drugs 0.000 description 1
- 230000006337 proteolytic cleavage Effects 0.000 description 1
- 210000001938 protoplast Anatomy 0.000 description 1
- 230000005180 public health Effects 0.000 description 1
- 150000003212 purines Chemical class 0.000 description 1
- 238000011002 quantification Methods 0.000 description 1
- 230000006798 recombination Effects 0.000 description 1
- 238000005215 recombination Methods 0.000 description 1
- 230000022532 regulation of transcription, DNA-dependent Effects 0.000 description 1
- 238000005067 remediation Methods 0.000 description 1
- BOLDJAUMGUJJKM-LSDHHAIUSA-N renifolin D Natural products CC(=C)[C@@H]1Cc2c(O)c(O)ccc2[C@H]1CC(=O)c3ccc(O)cc3O BOLDJAUMGUJJKM-LSDHHAIUSA-N 0.000 description 1
- 230000003362 replicative effect Effects 0.000 description 1
- 230000000717 retained effect Effects 0.000 description 1
- 239000002342 ribonucleoside Substances 0.000 description 1
- 238000002390 rotary evaporation Methods 0.000 description 1
- 230000007017 scission Effects 0.000 description 1
- 238000012163 sequencing technique Methods 0.000 description 1
- 238000003307 slaughter Methods 0.000 description 1
- 239000001632 sodium acetate Substances 0.000 description 1
- 235000017281 sodium acetate Nutrition 0.000 description 1
- 239000007787 solid Substances 0.000 description 1
- 230000037439 somatic mutation Effects 0.000 description 1
- 230000003595 spectral effect Effects 0.000 description 1
- 238000002798 spectrophotometry method Methods 0.000 description 1
- 238000007619 statistical method Methods 0.000 description 1
- 210000000130 stem cell Anatomy 0.000 description 1
- 239000008223 sterile water Substances 0.000 description 1
- 230000001954 sterilising effect Effects 0.000 description 1
- 238000004659 sterilization and disinfection Methods 0.000 description 1
- 238000003756 stirring Methods 0.000 description 1
- 238000003860 storage Methods 0.000 description 1
- 239000007929 subcutaneous injection Substances 0.000 description 1
- 238000010254 subcutaneous injection Methods 0.000 description 1
- 150000008163 sugars Chemical class 0.000 description 1
- 239000004094 surface-active agent Substances 0.000 description 1
- 230000004083 survival effect Effects 0.000 description 1
- 239000000375 suspending agent Substances 0.000 description 1
- 238000013268 sustained release Methods 0.000 description 1
- 239000012730 sustained-release form Substances 0.000 description 1
- 230000002195 synergetic effect Effects 0.000 description 1
- 229920002258 tannic acid Polymers 0.000 description 1
- 235000015523 tannic acid Nutrition 0.000 description 1
- 229940033123 tannic acid Drugs 0.000 description 1
- 125000003831 tetrazolyl group Chemical group 0.000 description 1
- 210000001541 thymus gland Anatomy 0.000 description 1
- 210000002303 tibia Anatomy 0.000 description 1
- 239000012929 tonicity agent Substances 0.000 description 1
- 230000000699 topical effect Effects 0.000 description 1
- 230000002110 toxicologic effect Effects 0.000 description 1
- 231100000759 toxicological effect Toxicity 0.000 description 1
- 238000001890 transfection Methods 0.000 description 1
- 230000009466 transformation Effects 0.000 description 1
- 230000001052 transient effect Effects 0.000 description 1
- 230000008733 trauma Effects 0.000 description 1
- 230000010304 tumor cell viability Effects 0.000 description 1
- 238000007039 two-step reaction Methods 0.000 description 1
- 241001430294 unidentified retrovirus Species 0.000 description 1
- 210000000689 upper leg Anatomy 0.000 description 1
- 238000001291 vacuum drying Methods 0.000 description 1
- 230000002792 vascular Effects 0.000 description 1
- 230000035899 viability Effects 0.000 description 1
- 231100000747 viability assay Toxicity 0.000 description 1
- 238000003026 viability measurement method Methods 0.000 description 1
- 229960004528 vincristine Drugs 0.000 description 1
- OGWKCGZFUXNPDA-XQKSVPLYSA-N vincristine Chemical compound C([N@]1C[C@@H](C[C@]2(C(=O)OC)C=3C(=CC4=C([C@]56[C@H]([C@@]([C@H](OC(C)=O)[C@]7(CC)C=CCN([C@H]67)CC5)(O)C(=O)OC)N4C=O)C=3)OC)C[C@@](C1)(O)CC)CC1=C2NC2=CC=CC=C12 OGWKCGZFUXNPDA-XQKSVPLYSA-N 0.000 description 1
- OGWKCGZFUXNPDA-UHFFFAOYSA-N vincristine Natural products C1C(CC)(O)CC(CC2(C(=O)OC)C=3C(=CC4=C(C56C(C(C(OC(C)=O)C7(CC)C=CCN(C67)CC5)(O)C(=O)OC)N4C=O)C=3)OC)CN1CCC1=C2NC2=CC=CC=C12 OGWKCGZFUXNPDA-UHFFFAOYSA-N 0.000 description 1
- 239000013603 viral vector Substances 0.000 description 1
- 230000000007 visual effect Effects 0.000 description 1
- 238000003260 vortexing Methods 0.000 description 1
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Chemical compound O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 1
- 229910052725 zinc Inorganic materials 0.000 description 1
- 239000011701 zinc Substances 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/0005—Vertebrate antigens
- A61K39/0011—Cancer antigens
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/0005—Vertebrate antigens
- A61K39/0011—Cancer antigens
- A61K39/001102—Receptors, cell surface antigens or cell surface determinants
- A61K39/001103—Receptors for growth factors
- A61K39/001104—Epidermal growth factor receptors [EGFR]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/0005—Vertebrate antigens
- A61K39/0011—Cancer antigens
- A61K39/001102—Receptors, cell surface antigens or cell surface determinants
- A61K39/001103—Receptors for growth factors
- A61K39/001106—Her-2/neu/ErbB2, Her-3/ErbB3 or Her 4/ErbB4
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/0005—Vertebrate antigens
- A61K39/0011—Cancer antigens
- A61K39/001154—Enzymes
- A61K39/001156—Tyrosinase and tyrosinase related proteinases [TRP-1 or TRP-2]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/39—Medicinal preparations containing antigens or antibodies characterised by the immunostimulating additives, e.g. chemical adjuvants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/46—Cellular immunotherapy
- A61K39/461—Cellular immunotherapy characterised by the cell type used
- A61K39/4612—B-cells
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/46—Cellular immunotherapy
- A61K39/461—Cellular immunotherapy characterised by the cell type used
- A61K39/4614—Monocytes; Macrophages
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/46—Cellular immunotherapy
- A61K39/461—Cellular immunotherapy characterised by the cell type used
- A61K39/4615—Dendritic cells
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/46—Cellular immunotherapy
- A61K39/462—Cellular immunotherapy characterized by the effect or the function of the cells
- A61K39/4622—Antigen presenting cells
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/46—Cellular immunotherapy
- A61K39/464—Cellular immunotherapy characterised by the antigen targeted or presented
- A61K39/4643—Vertebrate antigens
- A61K39/4644—Cancer antigens
- A61K39/464402—Receptors, cell surface antigens or cell surface determinants
- A61K39/464403—Receptors for growth factors
- A61K39/464406—Her-2/neu/ErbB2, Her-3/ErbB3 or Her 4/ ErbB4
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/46—Cellular immunotherapy
- A61K39/464—Cellular immunotherapy characterised by the antigen targeted or presented
- A61K39/4643—Vertebrate antigens
- A61K39/4644—Cancer antigens
- A61K39/46449—Melanoma antigens
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K45/00—Medicinal preparations containing active ingredients not provided for in groups A61K31/00 - A61K41/00
- A61K45/06—Mixtures of active ingredients without chemical characterisation, e.g. antiphlogistics and cardiaca
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/62—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being a protein, peptide or polyamino acid
- A61K47/64—Drug-peptide, drug-protein or drug-polyamino acid conjugates, i.e. the modifying agent being a peptide, protein or polyamino acid which is covalently bonded or complexed to a therapeutically active agent
- A61K47/645—Polycationic or polyanionic oligopeptides, polypeptides or polyamino acids, e.g. polylysine, polyarginine, polyglutamic acid or peptide TAT
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/69—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the conjugate being characterised by physical or galenical forms, e.g. emulsion, particle, inclusion complex, stent or kit
- A61K47/6921—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the conjugate being characterised by physical or galenical forms, e.g. emulsion, particle, inclusion complex, stent or kit the form being a particulate, a powder, an adsorbate, a bead or a sphere
- A61K47/6925—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the conjugate being characterised by physical or galenical forms, e.g. emulsion, particle, inclusion complex, stent or kit the form being a particulate, a powder, an adsorbate, a bead or a sphere the form being a microcapsule, nanocapsule, microbubble or nanobubble
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/10—Dispersions; Emulsions
- A61K9/127—Liposomes
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/10—Dispersions; Emulsions
- A61K9/127—Liposomes
- A61K9/1271—Non-conventional liposomes, e.g. PEGylated liposomes, liposomes coated with polymers
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/48—Preparations in capsules, e.g. of gelatin, of chocolate
- A61K9/50—Microcapsules having a gas, liquid or semi-solid filling; Solid microparticles or pellets surrounded by a distinct coating layer, e.g. coated microspheres, coated drug crystals
- A61K9/5005—Wall or coating material
- A61K9/5015—Organic compounds, e.g. fats, sugars
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/51—Medicinal preparations containing antigens or antibodies comprising whole cells, viruses or DNA/RNA
- A61K2039/53—DNA (RNA) vaccination
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/555—Medicinal preparations containing antigens or antibodies characterised by a specific combination antigen/adjuvant
- A61K2039/55511—Organic adjuvants
- A61K2039/55522—Cytokines; Lymphokines; Interferons
- A61K2039/55527—Interleukins
- A61K2039/55533—IL-2
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/555—Medicinal preparations containing antigens or antibodies characterised by a specific combination antigen/adjuvant
- A61K2039/55511—Organic adjuvants
- A61K2039/55555—Liposomes; Vesicles, e.g. nanoparticles; Spheres, e.g. nanospheres; Polymers
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/60—Medicinal preparations containing antigens or antibodies characteristics by the carrier linked to the antigen
- A61K2039/6018—Lipids, e.g. in lipopeptides
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/0019—Injectable compositions; Intramuscular, intravenous, arterial, subcutaneous administration; Compositions to be administered through the skin in an invasive manner
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/0019—Injectable compositions; Intramuscular, intravenous, arterial, subcutaneous administration; Compositions to be administered through the skin in an invasive manner
- A61K9/0021—Intradermal administration, e.g. through microneedle arrays, needleless injectors
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- Epidemiology (AREA)
- Mycology (AREA)
- Immunology (AREA)
- Microbiology (AREA)
- Cell Biology (AREA)
- Oncology (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Dispersion Chemistry (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Organic Chemistry (AREA)
- Molecular Biology (AREA)
- Nanotechnology (AREA)
- Dermatology (AREA)
- Medicines Containing Antibodies Or Antigens For Use As Internal Diagnostic Agents (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Medicinal Preparation (AREA)
- Peptides Or Proteins (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Micro-Organisms Or Cultivation Processes Thereof (AREA)
- Medicines Containing Material From Animals Or Micro-Organisms (AREA)
Description
本出願は、PCT国際特許出願PCT/US2018/015601(係属中;同時に出願;Atty.Dkt 37182.215WO01)および、2017年1月27に出願された米国仮特許出願62/451,575(係属中;Atty.Dkt 37182.215PV01)に対する優先権を主張し、その内容は、その全体が本明細書に、明白な参照により具体的に援用される。
本発明は、アメリカ国防総省により与えられたW81XWH-12-1-0414の助成金ならびに、アメリカ国立衛生研究所により与えられた1R01-CA193880-01A1およびU43-CA210181の助成金の下、政府支援によりなされた。米国政府は、本発明における一定の権利を有する。
ワクチンは通常、身体の免疫系によって認識され得る抗原および身体の免疫反応を増強し得る1または複数のアジュバントから構成されている。しかし治療用がんワクチンは感染性疾患を防止するために使用される予防用ワクチンとは全く異なっている。それは、ワクチンは病原体と戦う抗体を産生する代わりに、がん細胞を殺滅する免疫細胞を生成するのに十分に強力でなければならないからである。治療用ワクチンは、がんおよび感染性疾患を含む複数の種類の生命を脅かす疾患の処置において大きな潜在性を有している。がんワクチン接種の成功を決定する重要な課題は、選択した抗原に対する抗腫瘍応答の強力な誘導である。タンパク質ペプチドおよびDNAプラスミドの両方は、従来よりワクチン開発のための抗原として役立ってきた(Meleroら、2014年;Schwartzentruberら、2011年;Kantoffら、2010年)。タンパク質およびペプチドは調製が比較的容易で、大スケールで産生することができるが、抗原ペプチドの選択は患者の独特の主要組織適合性複合体(MHC)タンパク質の型に依存し、したがって個別の患者に適合するようにカスタム化する必要がある。一方、DNAワクチンは効力が低いという欠点があり、制御できないゲノム組込みのリスクがある(McNamaraら、2015年)。
(a)約30%~約70%の1,2-ジオレオイル-sn-グリセロ-3-エチルホスホコリン(「EDOPC」)、
(b)約70%~約30%の1,2-ジオレオイル-sn-グリセロ-3-ホスファチジル-エタノールアミン(「DOPE」)、または
(c)約0.5~約5%の1,2-ジステアロイル-sn-グリセロ-3-ホスホエタノールアミン-N-[アミノ(ポリエチレングリコール)-2000](「DSPE-PEG」)
を含む。
(a)約45%~約55%のEDOPC、
(b)約55%~約45%のDOPE、および
(c)約1%~約2%のDSPE-PEG
を、含んでよい。
配列番号1は、野生型TP53ペプチド由来の例示的な細胞傷害性T細胞エピトープであり、本開示の1または複数の態様に従って有用である。
説明的実施形態の記述
がん
mRNAに基づく治療用がんワクチン
mRNAをパッケージするためのリポポリプレックス組成物
医薬製剤
医薬の調製のための組成物
例示的定義
生物学的機能等価
(実施例1)
リポポリプレックスはmRNAに基づくワクチンの抗腫瘍免疫を増強する
材料および方法
結果
(実施例2)
リポポリプレックスはmRNAに基づくワクチンの抗腫瘍免疫を増強する
(実施例3)
mRNAワクチンプラットフォームの一般化した説明
(実施例4)
2つのmRNAを、デュアル免疫療法のために単一シェル中に共パッケージ化することができる
(実施例5)
個別化がん免疫療法のためのmRNAに基づくワクチン
a)より効果的で毒性が低く、生存期間に影響を与える処置レジメンに置き換えることにより、処置レジメンに革命をもたらすこと;
b)転移性乳がんに関連する死亡率を排除すること;および
c)乳がん細胞が何年も休眠状態であり、その後再出現する理由と方法を解明し、この情報を使用してそのような再発を防止すること。
参考文献
本発明は、例えば、以下の項目を提供する。
(項目1)
少なくとも第1の腫瘍抗原をコードするmRNA分子の集団を含む治療用がんワクチンを含む組成物であって、該集団が少なくとも第1の正電荷ポリマーまたはタンパク質を含む複数のポリプレックスまたはタンパク質コア粒子の中に含まれ、さらに該複数のポリプレックスまたはタンパク質コア粒子がそれ自体、第1の生体適合性脂質二重層シェルにカプセル化されている、組成物。
(項目2)
前記第1の生体適合性脂質二重層シェルが、1または複数の哺乳動物の抗原提示細胞による前記複数のポリプレックスまたはタンパク質コア粒子のマクロピノサイトーシスを促進する、項目1に記載の組成物。
(項目3)
前記生体適合性脂質二重層の中にカプセル化された、CpG、ポリ(I:C)、アラム、およびそれらの任意の組合せからなる群から選択されるアジュバントをさらに含む、項目1または項目2に記載の組成物。
(項目4)
前記生体適合性脂質二重層の間の空間の中にカプセル化された、IL-12p70タンパク質、FLT3リガンド、またはインドールアミン2,3-ジオキシゲナーゼ(IDO-1)阻害剤等の免疫調節化合物をさらに含む、先行する項目のいずれかに記載の組成物。
(項目5)
前記インドールアミン2,3-ジオキシゲナーゼ(IDO-1)阻害剤が、GDC-0919、INCB24360、またはそれらの組合せである、項目4に記載の組成物。
(項目6)
前記正電荷ポリマーまたはタンパク質が、プロタミン、ポリエチレンイミン、ポリ-(B-アミノエステル)、またはそれらの任意の組合せを含む、先行する項目のいずれかに記載の組成物。
(項目7)
前記生体適合性脂質二重層が、1,2-ジオレオイル-sn-グリセロ-3-エチルホスホコリン(EDOPC)、1,2-ジオレオイル-sn-グリセロ-3-ホスファチジル-エタノールアミン(DOPE)、1,2-ジステアロイル-sn-グリセロ-3-ホスホエタノールアミン-N-[アミノ(ポリエチレングリコール)-2000](DSPE-PEG)、およびそれらの組合せの1または複数を含む、先行する項目のいずれかに記載の組成物。
(項目8)
前記生体適合性脂質二重層が、
(a)約30%~約70%の1,2-ジオレオイル-sn-グリセロ-3-エチルホスホコリン(EDOPC)、
(b)約70%~約30%の1,2-ジオレオイル-sn-グリセロ-3-ホスファチジル-エタノールアミン(DOPE)、または
(c)約0.5~約5%の1,2-ジステアロイル-sn-グリセロ-3-ホスホエタノールアミン-N-[アミノ(ポリエチレングリコール)-2000](DSPE-PEG)
を含む、先行する項目のいずれかに記載の組成物。
(項目9)
前記生体適合性脂質二重層が、
(a)約45%~約55%の1,2-ジオレオイル-sn-グリセロ-3-エチルホスホコリン(EDOPC)、
(b)約55%~約45%の1,2-ジオレオイル-sn-グリセロ-3-ホスファチジルエタノールアミン(DOPE)、および(c)約1~約2%の1,2-ジステアロイル-sn-グリセロ-3-ホスホエタノールアミン-N-[アミノ(ポリエチレングリコール)-2000](DSPE-PEG)
を含む、先行する項目のいずれかに記載の組成物。
(項目10)
mRNA分子の前記集団が、哺乳動物のがん細胞に特異的な少なくとも1つの抗原をコードする、先行する項目のいずれかに記載の組成物。
(項目11)
mRNA分子の前記集団が、少なくとも1つのがんもしくは腫瘍特異的タンパク質、ポリペプチドもしくはペプチド、またはその抗原性断片をコードする、先行する項目のいずれかに記載の組成物。
(項目12)
mRNA分子の前記集団が、HER2p66、HER2E75、HER2 YVMA 、TRP2、p66、およびそれらの任意の組合せからなる群から選択される少なくとも1つのヒト腫瘍特異的ポリペプチドまたはペプチドをコードする、先行する項目のいずれかに記載の組成物。
(項目13)
好適な哺乳動物の細胞に導入されたときにI型インターフェロン(IFN-I)の発現を増加させるように、好ましくはIFN-α4、IFN-β、またはそれらの組合せの発現を増加させるように、適合され構成された、先行する項目のいずれかに記載の組成物。
(項目14)
哺乳動物の抗原提示細胞、例えば、ヒト樹状細胞、マクロファージ細胞、B細胞、がん細胞、またはそれらの組合せの集団に導入されたときに、IFN-Iの発現を増加させる、先行する項目のいずれかに記載の組成物。
(項目15)
治療剤、例えば、免疫調節剤、抗新生物剤、細胞傷害性薬剤、細胞増殖抑制剤、神経活性薬剤、抗炎症剤、抗脂血症剤、ホルモン、受容体アゴニスト、受容体アンタゴニスト、抗感染症剤、タンパク質、ペプチド、抗体、抗原結合性断片、酵素、RNA、DNA、siRNA、mRNA、リボザイム、ホルモン、コファクター、ステロイド、アンチセンス分子、またはそれらの任意の組合せからなる群から選択される薬剤をさらに含む、先行する項目のいずれかに記載の組成物。
(項目16)
化学療法剤が、シクロホスファミド、ドキソルビシン、5-フルオロウラシル、ドセタキセル、パクリタキセル、トラスツズマブ、メトトレキセート、エピルビシン、シスプラチン、カルボプラチン、ビノレルビン、カペシタビン、ゲムシタビン、ミトキサントロン、イサベピロン、エリブリン、ラパチニブ、カルムスチン、窒素マスタード、硫黄マスタード、四硝酸プラチン、ビンブラスチン、エトポシド、カンプトテシン、およびそれらの任意の組合せからなる群から選択される化合物を含む、項目13に記載の組成物。
(項目17)
抗原ポリペプチド、抗原性融合ポリペプチド、抗原性ペプチド、またはその断片をさらに含む、先行する項目のいずれかに記載の組成物。
(項目18)
メソ多孔質ケイ素粒子、ナノ粒子、ミクロ粒子、またはそれらの任意の組合せの集団の中に含まれた、先行する項目のいずれかに記載の組成物。
(項目19)
1または複数の界面活性剤、リポソーム、ニオソーム、エトソーム、トランスフェロソーム、リン脂質、スフィンゴソーム、またはそれらの任意の組合せと混合された、先行する項目のいずれかに記載の組成物。
(項目20)
1または複数の薬学的に許容される担体、緩衝剤、希釈剤、ビヒクル、または賦形剤をさらに含む、先行する項目のいずれかに記載の組成物。
(項目21)
哺乳動物への全身投与のため、好ましくはヒトへの皮内または静脈内投与のために製剤化された、先行する項目のいずれかに記載の組成物。
(項目22)
哺乳動物の抗原提示細胞、例えば、がん細胞、腫瘍細胞、マクロファージ細胞、B細胞、樹状細胞、またはそれらの任意の組合せの単離された集団の中に含まれた、先行する項目のいずれかに記載の組成物。
(項目23)
前記組成物および該組成物の投与を必要とするヒトへの該組成物の投与のための少なくとも第1のセットの指示を含む治療用キットの一部として適合され構成された、先行する項目のいずれかに記載の組成物。
(項目24)
哺乳動物のがんの1または複数の症状の予防、治療、または改善における使用のための、先行する項目のいずれかに記載の組成物。
(項目25)
ヒトのがんの1または複数の症状の治療または改善における使用のための、先行する項目のいずれかに記載の組成物。
(項目26)
ヒトの転移がんの1または複数の症状の治療または改善における使用のための、先行する項目のいずれかに記載の組成物。
(項目27)
先行する項目のいずれかに記載の組成物を含む、哺乳動物細胞の単離された集団。
(項目28)
抗原提示細胞、好ましくは、ヒト樹状細胞、マクロファージ、B細胞、またはそれらの組合せとして特徴付けられる、項目27に記載の哺乳動物細胞の単離された集団。
(項目29)
哺乳動物対象における疾患、障害、または機能不全、例えばがんの少なくとも1つの症状を処置しまたは改善するための医薬の製造における、項目1から26のいずれか一項に記載の組成物の使用。
(項目30)
前記哺乳動物対象がヒト、非ヒト霊長類、伴侶動物、外来種、家畜、または原料動物である、項目29に記載の使用。
(項目31)
1)項目1から26のいずれか一項に記載の組成物、および2)哺乳動物における疾患、障害、異常な状態、または機能不全、例えば、がんまたは過剰増殖性障害の1または複数の症状の防止、処置、または改善のためのレジメンの一部としての、該組成物の投与を必要とする該哺乳動物への該組成物の投与のための指示、を含むキット。
(項目32)
がんの1または複数の症状の処置または改善を必要とする哺乳動物におけるがんの1または複数の症状を処置しまたは改善する方法であって、該哺乳動物における疾患、障害、異常な状態、または機能不全の1または複数の症状を処置しまたは改善するのに十分な時間、有効量の項目1から26のいずれか一項に記載の組成物を該動物に投与するステップを含む、方法。
(項目33)
前記疾患が、難治性、転移、再発、または治療抵抗性がんとして診断されまたは同定されている、項目32に記載の方法。
(項目34)
前記がんが、転移乳がん、転移肺がん、または転移黒色腫である、項目33に記載の方法。
(項目35)
前記哺乳動物がヒトである、項目32から34のいずれか一項に記載の方法。
(項目36)
前記哺乳動物に治療有効量の放射線または追加の化学療法剤を投与するステップをさらに含む、項目32から35のいずれか一項に記載の方法。
(項目37)
前記組成物が、単回投与で、あるいは1日もしくはそれより長い日数からの期間にわたる、1週もしくはそれより長い週数の期間にわたる、または1か月もしくはそれより長い月数の期間にわたる、またはそれより長い期間にわたる一連の複数回投与で、前記哺乳動物に全身投与される、項目32から36のいずれか一項に記載の方法。
(項目38)
前記組成物が、化学療法剤、または第2の異なる治療用がんワクチンをさらに含む、項目32から37のいずれか一項に記載の方法。
(項目39)
治療用抗がん抗原をコードするmRNAの投与を必要とする哺乳動物対象の体内のがん細胞の集団に、治療用抗がん抗原をコードするmRNAを投与する方法であって、有効量の項目1から26のいずれか一項に記載の組成物を該対象に投与するステップを含む、方法。
Claims (38)
- 抗原をコードするmRNA分子の集団を含む組成物であって、mRNA分子の該集団が少なくともポリマーまたはタンパク質を含む複数のポリプレックスまたはタンパク質コア粒子の中に含まれ、該複数のポリプレックスまたはタンパク質コア粒子がそれ自体、生体適合性脂質二重層シェルにカプセル化されており、
該生体適合性脂質二重層シェルが、
(a)約45%~約55%の1,2-ジオレオイル-sn-グリセロ-3-エチルホスホコリン(EDOPC)、
(b)約55%~約45%の1,2-ジオレオイル-sn-グリセロ-3-ホスファチジルエタノールアミン(DOPE)、および
(c)約1%~約2%の1,2-ジステアロイル-sn-グリセロ-3-ホスホエタノールアミン-N-[アミノ(ポリエチレングリコール)-2000](DSPE-PEG)を含む、組成物。 - 前記生体適合性脂質二重層シェルが、1または複数の哺乳動物の抗原提示細胞による前記複数のポリプレックスまたはタンパク質コア粒子のマクロピノサイトーシスを促進する、請求項1に記載の組成物。
- 前記生体適合性脂質二重層シェルの中にカプセル化された、CpG、ポリ(I:C)、アラム、およびそれらの任意の組合せからなる群から選択されるアジュバントをさらに含む、請求項1または請求項2に記載の組成物。
- IL-12p70タンパク質、FLT3リガンド、およびインドールアミン2,3-ジオキシゲナーゼ(IDO-1)阻害剤からなる群より選択される免疫調節化合物をさらに含み、該免疫調節化合物は、前記生体適合性脂質二重層シェルの間の空間の中にカプセル化されており、該インドールアミン2,3-ジオキシゲナーゼ(IDO-1)阻害剤が、GDC-0919、INCB24360、またはそれらの組合せである、請求項1~3のいずれかに記載の組成物。
- 前記ポリマーまたはタンパク質が、プロタミン、ポリエチレンイミン、ポリ-(B-アミノエステル)、またはそれらの任意の組合せを含む、請求項1~4のいずれかに記載の組成物。
- 前記抗原が哺乳動物の細胞に特異的である、請求項1~5のいずれかに記載の組成物。
- mRNA分子の前記集団が、少なくとも1つのがんもしくは腫瘍特異的タンパク質、ポリペプチドもしくはペプチド、またはその抗原性断片をコードする、請求項1~6のいずれかに記載の組成物。
- mRNA分子の前記集団が、HER2p66、HER2E75、HER2YVMA、TRP2、p66、およびそれらの任意の組合せからなる群から選択される少なくとも1つのヒト腫瘍特異的ポリペプチドまたはペプチドをコードする、請求項1~7のいずれかに記載の組成物。
- 好適な哺乳動物の細胞に導入されたときにI型インターフェロン(IFN-I)の発現を増加させるように、好ましくはIFN-α4、IFN-β、またはそれらの組合せの発現を増加させるように、適合され構成された、請求項1~8のいずれかに記載の組成物。
- ヒト樹状細胞、マクロファージ細胞、B細胞、がん細胞、またはそれらの組合せを含む哺乳動物の抗原提示細胞の集団に導入されたときに、IFN-Iの発現を増加させる、請求項1~9のいずれかに記載の組成物。
- 免疫調節剤、抗新生物剤、細胞傷害性薬剤、細胞増殖抑制剤、神経活性薬剤、抗炎症剤、抗脂血症剤、ホルモン、受容体アゴニスト、受容体アンタゴニスト、抗感染症剤、タンパク質、ペプチド、抗体、抗原結合性断片、酵素、RNA、DNA、siRNA、mRNA、リボザイム、ホルモン、コファクター、ステロイド、アンチセンス分子、およびそれらの任意の組合せからなる群から選択される治療剤をさらに含む、請求項1~10のいずれかに記載の組成物。
- 前記治療剤が、シクロホスファミド、ドキソルビシン、5-フルオロウラシル、ドセタキセル、パクリタキセル、トラスツズマブ、メトトレキセート、エピルビシン、シスプラチン、カルボプラチン、ビノレルビン、カペシタビン、ゲムシタビン、ミトキサントロン、イサベピロン、エリブリン、ラパチニブ、カルムスチン、窒素マスタード、硫黄マスタード、四硝酸プラチン、ビンブラスチン、エトポシド、カンプトテシン、およびそれらの任意の組合せからなる群から選択される、請求項11に記載の組成物。
- 抗原ポリペプチド、抗原性融合ポリペプチド、抗原性ペプチド、またはその断片をさらに含む、請求項1~12のいずれかに記載の組成物。
- メソ多孔質ケイ素粒子、ナノ粒子、ミクロ粒子、またはそれらの任意の組合せの集団の中に含まれた、請求項1~13のいずれかに記載の組成物。
- 1または複数の界面活性剤、リポソーム、ニオソーム、エトソーム、トランスフェロソーム、リン脂質、スフィンゴソーム、またはそれらの任意の組合せをさらに含む、請求項1~14のいずれかに記載の組成物。
- 1または複数の薬学的に許容される担体、緩衝剤、希釈剤、ビヒクル、または賦形剤をさらに含む、請求項1~15のいずれかに記載の組成物。
- 哺乳動物への全身投与のため、好ましくはヒトへの皮内または静脈内投与のために製剤化された、請求項1~16のいずれかに記載の組成物。
- がん細胞、腫瘍細胞、マクロファージ細胞、B細胞、樹状細胞、またはそれらの任意の組合せを含む哺乳動物の抗原提示細胞の単離された集団の中に含まれた、請求項1~17のいずれかに記載の組成物。
- 前記組成物および該組成物の投与を必要とするヒトへの該組成物の投与のための少なくともセットの指示を含む治療用キットの一部として適合され構成された、請求項1~18のいずれかに記載の組成物。
- 哺乳動物のがんの1または複数の症状の予防、治療、または改善における使用のための、請求項1~19のいずれかに記載の組成物。
- ヒトのがんの1または複数の症状の治療または改善における使用のための、請求項1~20のいずれかに記載の組成物。
- ヒトの転移がんの1または複数の症状の治療または改善における使用のための、請求項1~21のいずれかに記載の組成物。
- 前記ポリマーまたはタンパク質は正に荷電している、請求項1~22のいずれか一項に記載の組成物。
- 請求項1~23のいずれかに記載の組成物を含む、哺乳動物細胞の単離された集団。
- 前記哺乳動物細胞は、抗原提示細胞、好ましくは、ヒト樹状細胞、マクロファージ、B細胞、またはそれらの組合せからなる群から選択される、請求項24に記載の哺乳動物細胞の単離された集団。
- 哺乳動物対象における疾患、障害、または機能不全の少なくとも1つの症状を処置しまたは改善するための医薬の製造における、請求項1から23のいずれか一項に記載の組成物の使用。
- 前記哺乳動物対象がヒト、非ヒト霊長類、伴侶動物、外来種、家畜、または原料動物である、請求項26に記載の使用。
- 1)請求項1から23のいずれか一項に記載の組成物、および2)哺乳動物における疾患、障害、異常な状態、または機能不全の1または複数の症状の防止、処置、または改善のためのレジメンの一部としての、該組成物の投与を必要とする該哺乳動物への該組成物の投与のための指示、を含むキット。
- 疾患、障害、異常な状態、または機能不全の1または複数の症状の処置または改善を必要とする哺乳動物における疾患、障害、異常な状態、または機能不全の1または複数の症状を処置しまたは改善するための、請求項1から23のいずれか一項に記載の組成物であって、前記組成物が、該哺乳動物における疾患、障害、異常な状態、または機能不全の1または複数の症状を処置しまたは改善するのに十分な時間投与されることを特徴とする、組成物。
- 前記疾患が、難治性、転移、再発、または治療抵抗性がんとして診断されまたは同定されている、請求項29に記載の組成物。
- 前記がんが、転移乳がん、転移肺がん、または転移黒色腫である、請求項30に記載の組成物。
- 前記哺乳動物がヒトである、請求項29から31のいずれか一項に記載の組成物。
- 前記組成物が、前記哺乳動物に放射線または追加の化学療法剤と組み合わせて投与されることを特徴とする、請求項29から32のいずれか一項に記載の組成物。
- 前記組成物が、単回投与で、あるいは1日もしくはそれより長い日数からの期間にわたる、1週もしくはそれより長い週数の期間にわたる、または1か月もしくはそれより長い月数の期間にわたる、またはそれより長い期間にわたる一連の複数回投与で、前記哺乳動物に全身投与される、請求項29から33のいずれか一項に記載の組成物。
- 前記組成物が、化学療法剤、または治療用がんワクチンをさらに含む、請求項29から34のいずれか一項に記載の組成物。
- 抗原をコードするmRNAの投与を必要とする哺乳動物対象の体内の細胞の集団に、抗原をコードするmRNAを投与するための、請求項1から23のいずれか一項に記載の組成物。
- 哺乳動物対象における疾患、障害、または機能不全の少なくとも1つの症状を処置しまたは改善するための、請求項1から23のいずれか一項に記載の組成物。
- 前記哺乳動物対象がヒト、非ヒト霊長類、伴侶動物、外来種、家畜、または原料動物である、請求項37に記載の組成物。
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2022184788A JP2023015346A (ja) | 2017-01-27 | 2022-11-18 | 免疫療法のためのコア/シェル構造プラットホーム |
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201762451575P | 2017-01-27 | 2017-01-27 | |
| US62/451,575 | 2017-01-27 | ||
| PCT/US2018/015601 WO2018140826A1 (en) | 2017-01-27 | 2018-01-26 | Core/shell structure platform for immunotherapy |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2022184788A Division JP2023015346A (ja) | 2017-01-27 | 2022-11-18 | 免疫療法のためのコア/シェル構造プラットホーム |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| JP2020505403A JP2020505403A (ja) | 2020-02-20 |
| JP2020505403A5 JP2020505403A5 (ja) | 2021-03-11 |
| JP7181880B2 true JP7181880B2 (ja) | 2022-12-01 |
Family
ID=61899339
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2019540384A Active JP7181880B2 (ja) | 2017-01-27 | 2018-01-26 | 免疫療法のためのコア/シェル構造プラットホーム |
| JP2022184788A Pending JP2023015346A (ja) | 2017-01-27 | 2022-11-18 | 免疫療法のためのコア/シェル構造プラットホーム |
Family Applications After (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2022184788A Pending JP2023015346A (ja) | 2017-01-27 | 2022-11-18 | 免疫療法のためのコア/シェル構造プラットホーム |
Country Status (11)
| Country | Link |
|---|---|
| US (2) | US11382964B2 (ja) |
| EP (3) | EP4008344B1 (ja) |
| JP (2) | JP7181880B2 (ja) |
| KR (2) | KR20240014586A (ja) |
| CN (2) | CN115845040A (ja) |
| AU (1) | AU2018212887A1 (ja) |
| CA (1) | CA3051136A1 (ja) |
| DK (2) | DK3573656T3 (ja) |
| ES (1) | ES2905591T3 (ja) |
| SG (1) | SG11201906744VA (ja) |
| WO (1) | WO2018140826A1 (ja) |
Families Citing this family (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US11433143B2 (en) | 2017-05-18 | 2022-09-06 | The Regents Of The University Of California | Nano-enabled immunotherapy in cancer |
| WO2018213631A1 (en) * | 2017-05-18 | 2018-11-22 | The Regents Of The University Of California | Nano-enabled immunotherapy in cancer |
| JP2022506839A (ja) * | 2018-11-07 | 2022-01-17 | モデルナティエックス インコーポレイテッド | Rnaがんワクチン |
| CN109260157A (zh) * | 2018-11-19 | 2019-01-25 | 上海交通大学 | 一种正电荷多肽脂质体纳米制剂及其制备方法与应用 |
| CA3133175A1 (en) * | 2019-03-14 | 2020-09-17 | Engene, Inc. | Reversible coating of chitosan-nucleic acid nanoparticles and methods of their use |
| CN114502204A (zh) * | 2019-08-14 | 2022-05-13 | 库尔维科公司 | 具有降低的免疫刺激性质的rna组合和组合物 |
| DE102019122014A1 (de) * | 2019-08-15 | 2021-02-18 | Technische Universität Darmstadt | Reduktion der Knochenresorption, insbesondere bei chronischen Gelenkerkrankungen |
| WO2021081043A1 (en) * | 2019-10-22 | 2021-04-29 | The Johns Hopkins University | Mucus penetrating particle compositions and methods of use thereof enhancing immune response |
| CN112245575A (zh) * | 2020-10-26 | 2021-01-22 | 上海杏禾医疗科技有限公司 | 含支化聚合物和mRNA的核壳结构的抗肿瘤疫苗及其用途 |
| WO2023034957A1 (en) * | 2021-09-03 | 2023-03-09 | University Of Connecticut | Stabilization of antigens for long term administration in transdermal microneedle patches |
| CN114699420A (zh) * | 2022-03-08 | 2022-07-05 | 南方科技大学 | 一种用于治疗前列腺癌组合物及其制备方法与应用 |
| CN114699538A (zh) * | 2022-04-02 | 2022-07-05 | 中国药科大学 | 一种核-壳式高效基因药物递送系统及其制备方法 |
| CN117442552B (zh) * | 2023-11-20 | 2024-03-26 | 山东大学 | 一种淋巴结内t细胞区靶向纳米粒及其水凝胶 |
Family Cites Families (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4554101A (en) | 1981-01-09 | 1985-11-19 | New York Blood Center, Inc. | Identification and preparation of epitopes on antigens and allergens on the basis of hydrophilicity |
| US5399363A (en) | 1991-01-25 | 1995-03-21 | Eastman Kodak Company | Surface modified anticancer nanoparticles |
| US5543158A (en) | 1993-07-23 | 1996-08-06 | Massachusetts Institute Of Technology | Biodegradable injectable nanoparticles |
| IE80468B1 (en) | 1995-04-04 | 1998-07-29 | Elan Corp Plc | Controlled release biodegradable nanoparticles containing insulin |
| WO2010037408A1 (en) * | 2008-09-30 | 2010-04-08 | Curevac Gmbh | Composition comprising a complexed (m)rna and a naked mrna for providing or enhancing an immunostimulatory response in a mammal and uses thereof |
| MX2014004415A (es) * | 2011-10-14 | 2015-06-05 | Stc Unm | Bicapas lipidicas soportadas por nanoparticulas porosas (protecelulas) para suministro dirigido, incluido el suministro transdermico de carga, y los metodos relacionados. |
| CN103181896B (zh) * | 2011-12-30 | 2015-10-14 | 沈阳药科大学 | 一种含有小檗胺类药物的脂质体制剂及其制备方法 |
| BR112015022868B1 (pt) * | 2013-03-14 | 2023-05-16 | Ethris Gmbh | Composições de mrna de cftr e usos e métodos relacionados |
| CN103550783A (zh) * | 2013-04-27 | 2014-02-05 | 中国人民解放军军事医学科学院放射与辐射医学研究所 | 一种核酸类药物靶向递送系统及其制备方法 |
| US20160151284A1 (en) * | 2013-07-23 | 2016-06-02 | Protiva Biotherapeutics, Inc. | Compositions and methods for delivering messenger rna |
| US20150110857A1 (en) * | 2013-10-22 | 2015-04-23 | Shire Human Genetic Therapies, Inc. | Cns delivery of mrna and uses thereof |
| CN107683142A (zh) * | 2015-04-02 | 2018-02-09 | 卫理公会医院 | 基于多孔硅微粒的癌症疫苗和增强抗肿瘤免疫性的方法 |
-
2018
- 2018-01-26 EP EP21210308.9A patent/EP4008344B1/en active Active
- 2018-01-26 DK DK18715827.4T patent/DK3573656T3/da active
- 2018-01-26 CN CN202310076857.0A patent/CN115845040A/zh active Pending
- 2018-01-26 AU AU2018212887A patent/AU2018212887A1/en active Pending
- 2018-01-26 US US15/881,637 patent/US11382964B2/en active Active
- 2018-01-26 DK DK21210308.9T patent/DK4008344T3/da active
- 2018-01-26 SG SG11201906744VA patent/SG11201906744VA/en unknown
- 2018-01-26 JP JP2019540384A patent/JP7181880B2/ja active Active
- 2018-01-26 KR KR1020247001662A patent/KR20240014586A/ko active Search and Examination
- 2018-01-26 EP EP18715827.4A patent/EP3573656B1/en active Active
- 2018-01-26 CA CA3051136A patent/CA3051136A1/en active Pending
- 2018-01-26 WO PCT/US2018/015601 patent/WO2018140826A1/en unknown
- 2018-01-26 CN CN201880001680.5A patent/CN109152830B/zh active Active
- 2018-01-26 EP EP23214393.3A patent/EP4309739A3/en active Pending
- 2018-01-26 KR KR1020197024702A patent/KR102627347B1/ko active IP Right Grant
- 2018-01-26 ES ES18715827T patent/ES2905591T3/es active Active
-
2022
- 2022-05-24 US US17/751,812 patent/US20220305103A1/en active Pending
- 2022-11-18 JP JP2022184788A patent/JP2023015346A/ja active Pending
Non-Patent Citations (1)
| Title |
|---|
| Biomaterials,2022年02月,Vol. 125,pp. 81-89 |
Also Published As
| Publication number | Publication date |
|---|---|
| CN109152830A (zh) | 2019-01-04 |
| EP3573656A1 (en) | 2019-12-04 |
| WO2018140826A9 (en) | 2018-10-11 |
| EP4008344B1 (en) | 2023-12-06 |
| EP3573656B1 (en) | 2021-12-22 |
| ES2905591T3 (es) | 2022-04-11 |
| EP4309739A3 (en) | 2024-04-17 |
| JP2020505403A (ja) | 2020-02-20 |
| CA3051136A1 (en) | 2018-08-02 |
| EP4309739A2 (en) | 2024-01-24 |
| JP2023015346A (ja) | 2023-01-31 |
| KR20190108150A (ko) | 2019-09-23 |
| WO2018140826A1 (en) | 2018-08-02 |
| EP4008344A1 (en) | 2022-06-08 |
| US11382964B2 (en) | 2022-07-12 |
| KR20240014586A (ko) | 2024-02-01 |
| CN115845040A (zh) | 2023-03-28 |
| US20220305103A1 (en) | 2022-09-29 |
| KR102627347B1 (ko) | 2024-01-22 |
| DK3573656T3 (da) | 2022-03-14 |
| DK4008344T3 (da) | 2024-01-29 |
| CN109152830B (zh) | 2023-11-03 |
| AU2018212887A1 (en) | 2019-08-08 |
| US20180360756A1 (en) | 2018-12-20 |
| SG11201906744VA (en) | 2019-08-27 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP7181880B2 (ja) | 免疫療法のためのコア/シェル構造プラットホーム | |
| Zhang et al. | DP7-C-modified liposomes enhance immune responses and the antitumor effect of a neoantigen-based mRNA vaccine | |
| JP6862369B2 (ja) | 細胞透過性ペプチド、カーゴ、及びtlrペプチドアゴニストを含む新規複合体 | |
| US9770467B2 (en) | Compositions and methods for cancer immunotherapy | |
| Nguyen et al. | Enhanced cancer DNA vaccine via direct transfection to host dendritic cells recruited in injectable scaffolds | |
| WO2019001339A1 (zh) | 一种新型的肿瘤疫苗及其用途 | |
| Yang et al. | Synthetic multiepitope neoantigen DNA vaccine for personalized cancer immunotherapy | |
| US20200384104A1 (en) | Structure-Function Relationships in the Development of Immunotherapeutic Agents | |
| WO2020146906A2 (en) | Self-assembled peptide nanoparticle and use thereof | |
| CN117797250A (zh) | 个体化mRNA组合物、载体、mRNA疫苗及其应用 | |
| US20240165263A1 (en) | Targeting multiple t cell types using spherical nucleic acid vaccine architecture | |
| US20180117176A1 (en) | Porous silicon microparticle-based cancer vaccines and methods for potentiating anti-tumor immunity | |
| Mohseninia et al. | Harnessing self-assembling peptide nanofibers to prime robust tumor-specific CD8 T cell responses in mice | |
| Cui et al. | Uplifting Anti‐tumor Immunotherapy with Lymph Node‐targeted and Ratio‐controlled Co‐delivery of Tumor Cell Lysate and Adjuvant |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20191001 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20210126 |
|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20210126 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20220309 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20220607 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20221021 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20221118 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 7181880 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| RD02 | Notification of acceptance of power of attorney |
Free format text: JAPANESE INTERMEDIATE CODE: R3D02 |