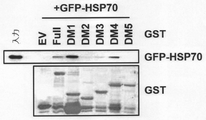
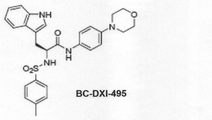
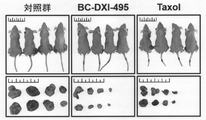
JP6914269B2 - Aimp2−dx2とhsp70との結合を阻害する抗がん剤スクリーニング方法 - Google Patents
Aimp2−dx2とhsp70との結合を阻害する抗がん剤スクリーニング方法 Download PDFInfo
- Publication number
- JP6914269B2 JP6914269B2 JP2018548001A JP2018548001A JP6914269B2 JP 6914269 B2 JP6914269 B2 JP 6914269B2 JP 2018548001 A JP2018548001 A JP 2018548001A JP 2018548001 A JP2018548001 A JP 2018548001A JP 6914269 B2 JP6914269 B2 JP 6914269B2
- Authority
- JP
- Japan
- Prior art keywords
- aimp2
- cancer
- hsp70
- binding
- protein
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related
Links
Images
Classifications
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/50—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing
- G01N33/5005—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving human or animal cells
- G01N33/5008—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving human or animal cells for testing or evaluating the effect of chemical or biological compounds, e.g. drugs, cosmetics
- G01N33/5011—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving human or animal cells for testing or evaluating the effect of chemical or biological compounds, e.g. drugs, cosmetics for testing antineoplastic activity
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/40—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with one nitrogen as the only ring hetero atom, e.g. sulpiride, succinimide, tolmetin, buflomedil
- A61K31/403—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with one nitrogen as the only ring hetero atom, e.g. sulpiride, succinimide, tolmetin, buflomedil condensed with carbocyclic rings, e.g. carbazole
- A61K31/404—Indoles, e.g. pindolol
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/535—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with at least one nitrogen and one oxygen as the ring hetero atoms, e.g. 1,2-oxazines
- A61K31/5375—1,4-Oxazines, e.g. morpholine
- A61K31/5377—1,4-Oxazines, e.g. morpholine not condensed and containing further heterocyclic rings, e.g. timolol
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/70—Carbohydrates; Sugars; Derivatives thereof
- A61K31/7088—Compounds having three or more nucleosides or nucleotides
- A61K31/7105—Natural ribonucleic acids, i.e. containing only riboses attached to adenine, guanine, cytosine or uracil and having 3'-5' phosphodiester links
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K48/00—Medicinal preparations containing genetic material which is inserted into cells of the living body to treat genetic diseases; Gene therapy
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/50—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/50—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing
- G01N33/53—Immunoassay; Biospecific binding assay; Materials therefor
- G01N33/575—Immunoassay; Biospecific binding assay; Materials therefor for cancer
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N2500/00—Screening for compounds of potential therapeutic value
- G01N2500/02—Screening involving studying the effect of compounds C on the interaction between interacting molecules A and B (e.g. A = enzyme and B = substrate for A, or A = receptor and B = ligand for the receptor)
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Chemical & Material Sciences (AREA)
- Immunology (AREA)
- Molecular Biology (AREA)
- Biomedical Technology (AREA)
- Medicinal Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Hematology (AREA)
- Urology & Nephrology (AREA)
- Biotechnology (AREA)
- Public Health (AREA)
- Animal Behavior & Ethology (AREA)
- Epidemiology (AREA)
- Pharmacology & Pharmacy (AREA)
- Veterinary Medicine (AREA)
- Biochemistry (AREA)
- General Physics & Mathematics (AREA)
- Pathology (AREA)
- Analytical Chemistry (AREA)
- Physics & Mathematics (AREA)
- Food Science & Technology (AREA)
- Microbiology (AREA)
- Cell Biology (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Toxicology (AREA)
- Tropical Medicine & Parasitology (AREA)
- Genetics & Genomics (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Hospice & Palliative Care (AREA)
- Oncology (AREA)
- Investigating Or Analysing Biological Materials (AREA)
- Measuring Or Testing Involving Enzymes Or Micro-Organisms (AREA)
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR10-2016-0027077 | 2016-03-07 | ||
| KR1020160027077A KR102297505B1 (ko) | 2016-03-07 | 2016-03-07 | Aimp2-dx2와 hsp70의 결합을 저해하는 항암제 스크리닝 방법 |
| PCT/KR2017/002442 WO2017155277A1 (ko) | 2016-03-07 | 2017-03-07 | Aimp2-dx2와 hsp70의 결합을 저해하는 항암제 스크리닝 방법 |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| JP2019512693A JP2019512693A (ja) | 2019-05-16 |
| JP2019512693A5 JP2019512693A5 (enExample) | 2020-06-18 |
| JP6914269B2 true JP6914269B2 (ja) | 2021-08-04 |
Family
ID=59790548
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2018548001A Expired - Fee Related JP6914269B2 (ja) | 2016-03-07 | 2017-03-07 | Aimp2−dx2とhsp70との結合を阻害する抗がん剤スクリーニング方法 |
Country Status (6)
| Country | Link |
|---|---|
| US (1) | US11442057B2 (enExample) |
| EP (1) | EP3418739B1 (enExample) |
| JP (1) | JP6914269B2 (enExample) |
| KR (1) | KR102297505B1 (enExample) |
| CN (1) | CN109073638B (enExample) |
| WO (1) | WO2017155277A1 (enExample) |
Families Citing this family (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR101831435B1 (ko) * | 2016-03-10 | 2018-02-22 | 재단법인 의약바이오컨버젼스연구단 | Aimp2-dx2 단백질에 특이적으로 결합하는 항체 |
| RU2764430C1 (ru) | 2018-01-29 | 2022-01-17 | Филип Моррис Продактс С.А. | Блок подсветки для систем, генерирующих аэрозоль |
| JP7491510B2 (ja) | 2018-04-26 | 2024-05-28 | メディシナル バイオコンバージェンス リサーチ センター | mTOR阻害剤としての新規化合物及びその用途 |
| JP2024516783A (ja) * | 2021-03-30 | 2024-04-17 | ボード オブ リージェンツ,ザ ユニバーシティ オブ テキサス システム | がんのバイオマーカーおよび治療標的としてのシトルリン化タンパク質 |
| CN115786504B (zh) * | 2022-08-31 | 2023-09-19 | 中国人民解放军总医院京南医疗区 | Hspa4抑制剂在治疗黑素瘤中的应用 |
Family Cites Families (21)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8039218B2 (en) * | 2002-11-14 | 2011-10-18 | John Wayne Cancer Institute | Detection of cancer cells in body fluids |
| US7459529B2 (en) * | 2004-11-24 | 2008-12-02 | Seoul National University Industry Foundation | AIMP2-DX2 and its uses |
| US8003780B2 (en) | 2004-11-24 | 2011-08-23 | Neomics Co., Ltd. | AIMP2-DX2 gene and SiRNA targeting AIMP2-DX2 |
| KR20060057992A (ko) * | 2004-11-24 | 2006-05-29 | 재단법인서울대학교산학협력재단 | p38-DX2 및 이의 용도 |
| EP1872124A4 (en) * | 2005-04-19 | 2008-06-11 | Prediction Sciences Llc | DIAGNOSIS MARKET FOR BREAST CANCER TREATMENT AND PROGRESSION AND METHOD OF USE THEREOF |
| WO2007011856A2 (en) * | 2005-07-15 | 2007-01-25 | The Trustees Of Columbia University In The City Of New York | Compositions and methods for differential diagnosis of chronic lymphocytic leukemia |
| KR20070113926A (ko) * | 2006-05-26 | 2007-11-29 | 재단법인서울대학교산학협력재단 | 암의 진단과 치료를 위한 aimp2dx2의 용도 |
| WO2009023846A2 (en) * | 2007-08-15 | 2009-02-19 | The Research Foundation Of State University Of New York | Methods for heat shock protein dependent cancer treatment |
| KR101067816B1 (ko) * | 2007-11-09 | 2011-09-27 | (주)네오믹스 | Aimp2-dx2의 억제제를 유효성분으로 포함하는 염증성질환 예방 및 치료용 조성물 |
| US20120178111A1 (en) * | 2009-09-23 | 2012-07-12 | Diamandis Eleftherios P | Methods and compositions for the detection of lung cancers |
| JP6008844B2 (ja) * | 2010-05-04 | 2016-10-19 | エータイアー ファーマ, インコーポレイテッド | p38MULTI−tRNA合成酵素複合体のタンパク質フラグメントに関連した治療用、診断用および抗体組成物の革新的発見 |
| WO2013006076A1 (en) * | 2011-07-04 | 2013-01-10 | New York University | The use of intranasally administered hsp70 protein to treat neurodegenerative diseases |
| KR20130016041A (ko) * | 2011-08-04 | 2013-02-14 | 재단법인 의약바이오컨버젼스연구단 | 신규한 아닐린 유도체 및 이의 용도 |
| KR101500804B1 (ko) * | 2012-04-20 | 2015-03-11 | 단국대학교 산학협력단 | Aimp2 유전자의 스플라이싱 변이체 특이적인 트랜스-스플라이싱 활성을 갖는 핵산 분자 및 이를 이용한 폐암 치료용 약학 조성물 및 폐암 진단용 조성물 |
| KR20130118612A (ko) * | 2012-04-20 | 2013-10-30 | (주)네오믹스 | 신규한 아미노피리딘 유도체 및 이의 용도 |
| KR101464360B1 (ko) * | 2012-11-09 | 2014-11-21 | 주식회사 대웅 | 리보자임과 shRNA를 함유하는 아데노바이러스 및 이를 포함하는 암 치료용 조성물 |
| KR101577315B1 (ko) | 2013-03-14 | 2015-12-15 | 부산대학교 산학협력단 | AIMP2-DX2와 p14/ARF의 상호작용을 이용한 암 치료제의 스크리닝 방법 |
| KR101762433B1 (ko) * | 2013-06-05 | 2017-07-28 | 재단법인 의약바이오컨버젼스연구단 | 신규한 말레인산 유도체 및 이의 제조방법 및 이를 포함하는 항암용 조성물 |
| KR101514320B1 (ko) * | 2013-06-14 | 2015-04-22 | 재단법인 의약바이오컨버젼스연구단 | 신규한 암 예방 또는 치료용 약학적 조성물 |
| KR101621963B1 (ko) * | 2014-03-06 | 2016-05-17 | 한국과학기술원 | Aimp2의 엑손2 결손체 돌연변이 검출용 분자신호기 및 이를 이용한 aimp2 돌연변이의 검출방법 |
| EP3162803B1 (en) * | 2014-06-30 | 2019-09-11 | Pusan National University Industry-University Cooperation Foundation | New compound for inhibiting binding between dx2 protein and p14/arf protein, and pharmaceutical composition for treating or preventing cancer disease containing same as effective ingredient |
-
2016
- 2016-03-07 KR KR1020160027077A patent/KR102297505B1/ko active Active
-
2017
- 2017-03-07 WO PCT/KR2017/002442 patent/WO2017155277A1/ko not_active Ceased
- 2017-03-07 JP JP2018548001A patent/JP6914269B2/ja not_active Expired - Fee Related
- 2017-03-07 EP EP17763538.0A patent/EP3418739B1/en active Active
- 2017-03-07 CN CN201780027761.8A patent/CN109073638B/zh active Active
-
2018
- 2018-09-07 US US16/125,113 patent/US11442057B2/en active Active
Also Published As
| Publication number | Publication date |
|---|---|
| EP3418739B1 (en) | 2021-05-12 |
| KR20170104263A (ko) | 2017-09-15 |
| EP3418739A4 (en) | 2019-03-06 |
| WO2017155277A8 (ko) | 2018-11-08 |
| US20190033295A1 (en) | 2019-01-31 |
| WO2017155277A1 (ko) | 2017-09-14 |
| CN109073638A (zh) | 2018-12-21 |
| KR102297505B1 (ko) | 2021-09-01 |
| EP3418739A1 (en) | 2018-12-26 |
| CN109073638B (zh) | 2022-04-26 |
| JP2019512693A (ja) | 2019-05-16 |
| US11442057B2 (en) | 2022-09-13 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| Meng et al. | USP5 promotes epithelial-mesenchymal transition by stabilizing SLUG in hepatocellular carcinoma | |
| JP6914269B2 (ja) | Aimp2−dx2とhsp70との結合を阻害する抗がん剤スクリーニング方法 | |
| Lagadec et al. | Radiation-induced Notch signaling in breast cancer stem cells | |
| Lu et al. | Rottlerin induces Wnt co-receptor LRP6 degradation and suppresses both Wnt/β-catenin and mTORC1 signaling in prostate and breast cancer cells | |
| Chen et al. | Targeting USP9x/SOX2 axis contributes to the anti-osteosarcoma effect of neogambogic acid | |
| Tang et al. | Targeting Trop2 by Bruceine D suppresses breast cancer metastasis by blocking Trop2/β-catenin positive feedback loop | |
| US9597369B2 (en) | Inhibitors of atypical protein kinase C and their use in treating hedgehog pathway-dependent cancers | |
| Cai et al. | Overexpression of caveolin‐1 induces alteration of multidrug resistance in Hs578T breast adenocarcinoma cells | |
| Tan et al. | Gypensapogenin H suppresses tumor growth and cell migration in triple-negative breast cancer by regulating PI3K/AKT/NF-κB/MMP-9 signaling pathway | |
| Gong et al. | Kindlin-2 controls sensitivity of prostate cancer cells to cisplatin-induced cell death | |
| Zhao et al. | Noxa and Puma genes regulated by hTERT promoter can mitigate growth and induce apoptosis in hepatocellular carcinoma mouse model | |
| EP2857392A1 (en) | Small compound targeting at tacc3 | |
| Zhu et al. | Human inhibitor of growth 1 inhibits hepatoma cell growth and influences p53 stability in a variant‐dependent manner | |
| KR101367832B1 (ko) | 종양 억제 타겟으로서 Hades의 용도 | |
| EP3801473B1 (en) | Setbp1 inhibitors for the treatment of myeloid neoplasms and solid tumors | |
| Wang et al. | RGS-GAIP–interacting protein controls breast cancer progression | |
| Mrozek et al. | Anti-cancer small-molecule Inauhzin-C confines its cytotoxicity to cancer cells by targeting GRP78 | |
| Ludwig et al. | The tumor suppressor, p190RhoGAP, differentially initiates apoptosis and confers docetaxel sensitivity to breast cancer cells | |
| Jia et al. | Raddeanin A (RA) Inhibited EMT and Stemness in Glioblastoma via downregulating Skp2 | |
| Fan et al. | High-fat diet promotes colorectal carcinogenesis through SERCA2 mediated serine phosphorylation of Annexin A2 | |
| Miao et al. | A Small Molecule Selectively Targets N-Myc to Suppress Neuroblastoma Cancer Progression | |
| CN119925343B (zh) | squamocin的新应用 | |
| CN115590945B (zh) | 一种用于治疗肿瘤的多肽及其应用 | |
| JP7638527B2 (ja) | 性ホルモン非感受性greb1陽性腫瘍の治療剤 | |
| KR100880850B1 (ko) | 고형암 세포에서 사이클로필린 a의 발현을 억제하는물질을 포함하는 시스플라틴에 대한 내성 억제용 약제학적조성물 및 그를 이용하여 고형암의 시스플라틴에 대한내성을 억제하는 방법 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A524 | Written submission of copy of amendment under article 19 pct |
Free format text: JAPANESE INTERMEDIATE CODE: A524 Effective date: 20200305 |
|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20200305 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20200511 |
|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20210226 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20210309 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20210531 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20210615 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20210713 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 6914269 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| LAPS | Cancellation because of no payment of annual fees |