JP6906529B2 - 編込みまたは編組管状金属構成体 - Google Patents
編込みまたは編組管状金属構成体 Download PDFInfo
- Publication number
- JP6906529B2 JP6906529B2 JP2018537852A JP2018537852A JP6906529B2 JP 6906529 B2 JP6906529 B2 JP 6906529B2 JP 2018537852 A JP2018537852 A JP 2018537852A JP 2018537852 A JP2018537852 A JP 2018537852A JP 6906529 B2 JP6906529 B2 JP 6906529B2
- Authority
- JP
- Japan
- Prior art keywords
- fiber
- component
- tubular structure
- braided
- components
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 229910052751 metal Inorganic materials 0.000 title claims description 30
- 239000002184 metal Substances 0.000 title claims description 30
- 239000000835 fiber Substances 0.000 claims description 52
- 229910045601 alloy Inorganic materials 0.000 claims description 10
- 239000000956 alloy Substances 0.000 claims description 10
- 229910001000 nickel titanium Inorganic materials 0.000 claims description 10
- HLXZNVUGXRDIFK-UHFFFAOYSA-N nickel titanium Chemical compound [Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni] HLXZNVUGXRDIFK-UHFFFAOYSA-N 0.000 claims description 10
- XEEYBQQBJWHFJM-UHFFFAOYSA-N Iron Chemical compound [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 claims description 6
- 230000004323 axial length Effects 0.000 claims description 5
- 229910001220 stainless steel Inorganic materials 0.000 claims description 5
- 239000010935 stainless steel Substances 0.000 claims description 4
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 claims description 3
- HCHKCACWOHOZIP-UHFFFAOYSA-N Zinc Chemical compound [Zn] HCHKCACWOHOZIP-UHFFFAOYSA-N 0.000 claims description 3
- 230000002745 absorbent Effects 0.000 claims description 3
- 239000002250 absorbent Substances 0.000 claims description 3
- 229910052742 iron Inorganic materials 0.000 claims description 3
- 229910052749 magnesium Inorganic materials 0.000 claims description 3
- 239000011777 magnesium Substances 0.000 claims description 3
- 229910052725 zinc Inorganic materials 0.000 claims description 3
- 239000011701 zinc Substances 0.000 claims description 3
- 229910000531 Co alloy Inorganic materials 0.000 claims description 2
- WAIPAZQMEIHHTJ-UHFFFAOYSA-N [Cr].[Co] Chemical class [Cr].[Co] WAIPAZQMEIHHTJ-UHFFFAOYSA-N 0.000 claims description 2
- 239000000463 material Substances 0.000 description 20
- 210000000988 bone and bone Anatomy 0.000 description 10
- 238000002788 crimping Methods 0.000 description 10
- 238000013461 design Methods 0.000 description 6
- 230000000399 orthopedic effect Effects 0.000 description 6
- 210000001519 tissue Anatomy 0.000 description 6
- 238000009954 braiding Methods 0.000 description 5
- 239000000470 constituent Substances 0.000 description 5
- 239000002131 composite material Substances 0.000 description 4
- 150000002739 metals Chemical class 0.000 description 4
- 238000000034 method Methods 0.000 description 4
- 238000012545 processing Methods 0.000 description 4
- 238000009864 tensile test Methods 0.000 description 4
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical compound [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 description 3
- 230000006378 damage Effects 0.000 description 3
- 238000009826 distribution Methods 0.000 description 3
- 230000006870 function Effects 0.000 description 3
- 239000012535 impurity Substances 0.000 description 3
- 238000004519 manufacturing process Methods 0.000 description 3
- 229920000642 polymer Polymers 0.000 description 3
- 230000009467 reduction Effects 0.000 description 3
- 229910001285 shape-memory alloy Inorganic materials 0.000 description 3
- 210000001562 sternum Anatomy 0.000 description 3
- XKRFYHLGVUSROY-UHFFFAOYSA-N Argon Chemical compound [Ar] XKRFYHLGVUSROY-UHFFFAOYSA-N 0.000 description 2
- PXHVJJICTQNCMI-UHFFFAOYSA-N Nickel Chemical compound [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 description 2
- 208000027418 Wounds and injury Diseases 0.000 description 2
- 150000001875 compounds Chemical class 0.000 description 2
- 238000010276 construction Methods 0.000 description 2
- 238000007796 conventional method Methods 0.000 description 2
- 238000010586 diagram Methods 0.000 description 2
- 238000010438 heat treatment Methods 0.000 description 2
- 239000010936 titanium Substances 0.000 description 2
- 229910052719 titanium Inorganic materials 0.000 description 2
- 229910001040 Beta-titanium Inorganic materials 0.000 description 1
- 229910001069 Ti alloy Inorganic materials 0.000 description 1
- 230000006978 adaptation Effects 0.000 description 1
- 239000003570 air Substances 0.000 description 1
- 229910052786 argon Inorganic materials 0.000 description 1
- 239000011230 binding agent Substances 0.000 description 1
- 230000015572 biosynthetic process Effects 0.000 description 1
- 230000017531 blood circulation Effects 0.000 description 1
- 210000004204 blood vessel Anatomy 0.000 description 1
- 230000008859 change Effects 0.000 description 1
- 239000000788 chromium alloy Substances 0.000 description 1
- 229910017052 cobalt Inorganic materials 0.000 description 1
- 239000010941 cobalt Substances 0.000 description 1
- GUTLYIVDDKVIGB-UHFFFAOYSA-N cobalt atom Chemical compound [Co] GUTLYIVDDKVIGB-UHFFFAOYSA-N 0.000 description 1
- 230000002301 combined effect Effects 0.000 description 1
- 230000000295 complement effect Effects 0.000 description 1
- 230000008602 contraction Effects 0.000 description 1
- 238000005520 cutting process Methods 0.000 description 1
- 230000007423 decrease Effects 0.000 description 1
- 230000001419 dependent effect Effects 0.000 description 1
- 230000010339 dilation Effects 0.000 description 1
- 238000006073 displacement reaction Methods 0.000 description 1
- 230000009977 dual effect Effects 0.000 description 1
- 238000009661 fatigue test Methods 0.000 description 1
- 239000012530 fluid Substances 0.000 description 1
- 230000002496 gastric effect Effects 0.000 description 1
- 239000001307 helium Substances 0.000 description 1
- 229910052734 helium Inorganic materials 0.000 description 1
- SWQJXJOGLNCZEY-UHFFFAOYSA-N helium atom Chemical compound [He] SWQJXJOGLNCZEY-UHFFFAOYSA-N 0.000 description 1
- 239000001257 hydrogen Substances 0.000 description 1
- 229910052739 hydrogen Inorganic materials 0.000 description 1
- 125000004435 hydrogen atom Chemical class [H]* 0.000 description 1
- 208000014674 injury Diseases 0.000 description 1
- 210000003734 kidney Anatomy 0.000 description 1
- 210000003041 ligament Anatomy 0.000 description 1
- 238000005259 measurement Methods 0.000 description 1
- 239000002905 metal composite material Substances 0.000 description 1
- 239000007769 metal material Substances 0.000 description 1
- 239000000203 mixture Substances 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 210000003205 muscle Anatomy 0.000 description 1
- 210000001577 neostriatum Anatomy 0.000 description 1
- 229910052759 nickel Inorganic materials 0.000 description 1
- 230000001737 promoting effect Effects 0.000 description 1
- 229910002059 quaternary alloy Inorganic materials 0.000 description 1
- 210000002254 renal artery Anatomy 0.000 description 1
- 230000008660 renal denervation Effects 0.000 description 1
- 230000008439 repair process Effects 0.000 description 1
- 238000011160 research Methods 0.000 description 1
- 230000000241 respiratory effect Effects 0.000 description 1
- 230000004044 response Effects 0.000 description 1
- 238000007790 scraping Methods 0.000 description 1
- 230000035807 sensation Effects 0.000 description 1
- 239000007787 solid Substances 0.000 description 1
- 230000000638 stimulation Effects 0.000 description 1
- 230000035882 stress Effects 0.000 description 1
- 238000001356 surgical procedure Methods 0.000 description 1
- 230000003655 tactile properties Effects 0.000 description 1
- 229910002058 ternary alloy Inorganic materials 0.000 description 1
- 238000012360 testing method Methods 0.000 description 1
- 230000008646 thermal stress Effects 0.000 description 1
- 238000009941 weaving Methods 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L31/00—Materials for other surgical articles, e.g. stents, stent-grafts, shunts, surgical drapes, guide wires, materials for adhesion prevention, occluding devices, surgical gloves, tissue fixation devices
- A61L31/02—Inorganic materials
- A61L31/022—Metals or alloys
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods, e.g. tourniquets
- A61B17/56—Surgical instruments or methods for treatment of bones or joints; Devices specially adapted therefor
- A61B17/58—Surgical instruments or methods for treatment of bones or joints; Devices specially adapted therefor for osteosynthesis, e.g. bone plates, screws, setting implements or the like
- A61B17/68—Internal fixation devices, including fasteners and spinal fixators, even if a part thereof projects from the skin
- A61B17/80—Cortical plates, i.e. bone plates; Instruments for holding or positioning cortical plates, or for compressing bones attached to cortical plates
- A61B17/8061—Cortical plates, i.e. bone plates; Instruments for holding or positioning cortical plates, or for compressing bones attached to cortical plates specially adapted for particular bones
- A61B17/8076—Cortical plates, i.e. bone plates; Instruments for holding or positioning cortical plates, or for compressing bones attached to cortical plates specially adapted for particular bones for the ribs or the sternum
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods, e.g. tourniquets
- A61B17/56—Surgical instruments or methods for treatment of bones or joints; Devices specially adapted therefor
- A61B17/58—Surgical instruments or methods for treatment of bones or joints; Devices specially adapted therefor for osteosynthesis, e.g. bone plates, screws, setting implements or the like
- A61B17/68—Internal fixation devices, including fasteners and spinal fixators, even if a part thereof projects from the skin
- A61B17/82—Internal fixation devices, including fasteners and spinal fixators, even if a part thereof projects from the skin for bone cerclage
- A61B17/823—Internal fixation devices, including fasteners and spinal fixators, even if a part thereof projects from the skin for bone cerclage for the sternum
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods, e.g. tourniquets
- A61B17/56—Surgical instruments or methods for treatment of bones or joints; Devices specially adapted therefor
- A61B17/58—Surgical instruments or methods for treatment of bones or joints; Devices specially adapted therefor for osteosynthesis, e.g. bone plates, screws, setting implements or the like
- A61B17/68—Internal fixation devices, including fasteners and spinal fixators, even if a part thereof projects from the skin
- A61B17/82—Internal fixation devices, including fasteners and spinal fixators, even if a part thereof projects from the skin for bone cerclage
- A61B17/826—Nets or webs
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods, e.g. tourniquets
- A61B17/56—Surgical instruments or methods for treatment of bones or joints; Devices specially adapted therefor
- A61B17/58—Surgical instruments or methods for treatment of bones or joints; Devices specially adapted therefor for osteosynthesis, e.g. bone plates, screws, setting implements or the like
- A61B17/68—Internal fixation devices, including fasteners and spinal fixators, even if a part thereof projects from the skin
- A61B17/84—Fasteners therefor or fasteners being internal fixation devices
- A61B17/842—Flexible wires, bands or straps
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/02—Prostheses implantable into the body
- A61F2/08—Muscles; Tendons; Ligaments
- A61F2/0811—Fixation devices for tendons or ligaments
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/82—Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/86—Stents in a form characterised by the wire-like elements; Stents in the form characterised by a net-like or mesh-like structure
- A61F2/90—Stents in a form characterised by the wire-like elements; Stents in the form characterised by a net-like or mesh-like structure characterised by a net-like or mesh-like structure
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L31/00—Materials for other surgical articles, e.g. stents, stent-grafts, shunts, surgical drapes, guide wires, materials for adhesion prevention, occluding devices, surgical gloves, tissue fixation devices
- A61L31/14—Materials characterised by their function or physical properties, e.g. injectable or lubricating compositions, shape-memory materials, surface modified materials
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L31/00—Materials for other surgical articles, e.g. stents, stent-grafts, shunts, surgical drapes, guide wires, materials for adhesion prevention, occluding devices, surgical gloves, tissue fixation devices
- A61L31/14—Materials characterised by their function or physical properties, e.g. injectable or lubricating compositions, shape-memory materials, surface modified materials
- A61L31/148—Materials at least partially resorbable by the body
-
- D—TEXTILES; PAPER
- D02—YARNS; MECHANICAL FINISHING OF YARNS OR ROPES; WARPING OR BEAMING
- D02G—CRIMPING OR CURLING FIBRES, FILAMENTS, THREADS, OR YARNS; YARNS OR THREADS
- D02G3/00—Yarns or threads, e.g. fancy yarns; Processes or apparatus for the production thereof, not otherwise provided for
- D02G3/02—Yarns or threads characterised by the material or by the materials from which they are made
- D02G3/12—Threads containing metallic filaments or strips
-
- D—TEXTILES; PAPER
- D03—WEAVING
- D03D—WOVEN FABRICS; METHODS OF WEAVING; LOOMS
- D03D1/00—Woven fabrics designed to make specified articles
-
- D—TEXTILES; PAPER
- D03—WEAVING
- D03D—WOVEN FABRICS; METHODS OF WEAVING; LOOMS
- D03D15/00—Woven fabrics characterised by the material, structure or properties of the fibres, filaments, yarns, threads or other warp or weft elements used
- D03D15/20—Woven fabrics characterised by the material, structure or properties of the fibres, filaments, yarns, threads or other warp or weft elements used characterised by the material of the fibres or filaments constituting the yarns or threads
- D03D15/242—Woven fabrics characterised by the material, structure or properties of the fibres, filaments, yarns, threads or other warp or weft elements used characterised by the material of the fibres or filaments constituting the yarns or threads inorganic, e.g. basalt
- D03D15/25—Metal
-
- D—TEXTILES; PAPER
- D03—WEAVING
- D03D—WOVEN FABRICS; METHODS OF WEAVING; LOOMS
- D03D3/00—Woven fabrics characterised by their shape
- D03D3/02—Tubular fabrics
-
- D—TEXTILES; PAPER
- D04—BRAIDING; LACE-MAKING; KNITTING; TRIMMINGS; NON-WOVEN FABRICS
- D04C—BRAIDING OR MANUFACTURE OF LACE, INCLUDING BOBBIN-NET OR CARBONISED LACE; BRAIDING MACHINES; BRAID; LACE
- D04C1/00—Braid or lace, e.g. pillow-lace; Processes for the manufacture thereof
- D04C1/02—Braid or lace, e.g. pillow-lace; Processes for the manufacture thereof made from particular materials
-
- D—TEXTILES; PAPER
- D04—BRAIDING; LACE-MAKING; KNITTING; TRIMMINGS; NON-WOVEN FABRICS
- D04C—BRAIDING OR MANUFACTURE OF LACE, INCLUDING BOBBIN-NET OR CARBONISED LACE; BRAIDING MACHINES; BRAID; LACE
- D04C1/00—Braid or lace, e.g. pillow-lace; Processes for the manufacture thereof
- D04C1/06—Braid or lace serving particular purposes
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods, e.g. tourniquets
- A61B2017/00831—Material properties
- A61B2017/00862—Material properties elastic or resilient
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/02—Prostheses implantable into the body
- A61F2/08—Muscles; Tendons; Ligaments
- A61F2/0811—Fixation devices for tendons or ligaments
- A61F2002/0847—Mode of fixation of anchor to tendon or ligament
- A61F2002/0852—Fixation of a loop or U-turn, e.g. eyelets, anchor having multiple holes
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2210/00—Particular material properties of prostheses classified in groups A61F2/00 - A61F2/26 or A61F2/82 or A61F9/00 or A61F11/00 or subgroups thereof
- A61F2210/0004—Particular material properties of prostheses classified in groups A61F2/00 - A61F2/26 or A61F2/82 or A61F9/00 or A61F11/00 or subgroups thereof bioabsorbable
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2210/00—Particular material properties of prostheses classified in groups A61F2/00 - A61F2/26 or A61F2/82 or A61F9/00 or A61F11/00 or subgroups thereof
- A61F2210/0014—Particular material properties of prostheses classified in groups A61F2/00 - A61F2/26 or A61F2/82 or A61F9/00 or A61F11/00 or subgroups thereof using shape memory or superelastic materials, e.g. nitinol
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2220/00—Fixations or connections for prostheses classified in groups A61F2/00 - A61F2/26 or A61F2/82 or A61F9/00 or A61F11/00 or subgroups thereof
- A61F2220/0025—Connections or couplings between prosthetic parts, e.g. between modular parts; Connecting elements
- A61F2220/0033—Connections or couplings between prosthetic parts, e.g. between modular parts; Connecting elements made by longitudinally pushing a protrusion into a complementary-shaped recess, e.g. held by friction fit
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L2400/00—Materials characterised by their function or physical properties
- A61L2400/16—Materials with shape-memory or superelastic properties
-
- D—TEXTILES; PAPER
- D10—INDEXING SCHEME ASSOCIATED WITH SUBLASSES OF SECTION D, RELATING TO TEXTILES
- D10B—INDEXING SCHEME ASSOCIATED WITH SUBLASSES OF SECTION D, RELATING TO TEXTILES
- D10B2101/00—Inorganic fibres
- D10B2101/20—Metallic fibres
-
- D—TEXTILES; PAPER
- D10—INDEXING SCHEME ASSOCIATED WITH SUBLASSES OF SECTION D, RELATING TO TEXTILES
- D10B—INDEXING SCHEME ASSOCIATED WITH SUBLASSES OF SECTION D, RELATING TO TEXTILES
- D10B2401/00—Physical properties
- D10B2401/12—Physical properties biodegradable
-
- D—TEXTILES; PAPER
- D10—INDEXING SCHEME ASSOCIATED WITH SUBLASSES OF SECTION D, RELATING TO TEXTILES
- D10B—INDEXING SCHEME ASSOCIATED WITH SUBLASSES OF SECTION D, RELATING TO TEXTILES
- D10B2509/00—Medical; Hygiene
- D10B2509/04—Sutures
-
- D—TEXTILES; PAPER
- D10—INDEXING SCHEME ASSOCIATED WITH SUBLASSES OF SECTION D, RELATING TO TEXTILES
- D10B—INDEXING SCHEME ASSOCIATED WITH SUBLASSES OF SECTION D, RELATING TO TEXTILES
- D10B2509/00—Medical; Hygiene
- D10B2509/06—Vascular grafts; stents
Description
本願は、2016年1月22日付で出願された、WOVEN OR BRAIDED TUBULAR METAL CONSTRUCTと題する米国特許仮出願第62/286062号の利益を主張するものであり、その全開示は参照により本明細書に援用される。
本明細書中の使用では、「ワイヤ」または「ワイヤ製品」は、例えば円形断面を有するワイヤ及び平坦なワイヤまたはリボンを含む非円形断面を有するワイヤ等の、連続的に生産されてその後の流通及び使用のためにスプールに巻回され得る、連続状ワイヤ及びワイヤ製品であり得る。「ワイヤ」または「ワイヤ製品」はまた、特定の用途に応じて特定の長さに生産され得る素線、ケーブル、コイル及び管類等の、上記以外のワイヤ系製品も包含する。一部の例示的実施形態では、本開示に従ったワイヤまたはワイヤ製品は、最大2.5mmの直径を有し得る。
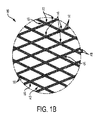
図1Aに示す編組管状構成体10は、外径DO及び内径DIならびに長手方向軸Aを画成する概略管状の構成体を協働して形成する多数の構成要素12を含む。特に、各要素12は生産時にマンドレル(図示せず)周囲に巻回されて螺旋を形成する。それぞれの構成要素12は、十字交差螺旋の規則的重ね合せパターンが形成されて完成管状構成体10を生成するように、相互に編込みまたは編組される。多数の用途で、構成体10は、軸方向(即ち、構成体の軸方向長は長手方向軸Aに沿って印加された力により「引伸ばされ」または「押縮められ」得る)及び半径方向(即ち、長手方向軸Aが曲線または非線形形態に「湾曲され」得る)の両方向に本質的に可撓性である構造体である。本開示目的のために、完成形態の構成体10の直径DO、直径DI及び軸方向長は、構成体10が何らの外力も加えられない「自由状態」にあるときの計測値であるため、構成体10の全体的形状及びサイズは、その編組または編込み形態、その構成要素12の材料、及び熱処理等の任意の加工処理により決定づけられることになる(以下に更に説明する)。
上述のように、構成要素12は金属素線14から作製される。医療用具の目的に対しては、生体適合性金属または移植可能金属は素線14に好適な候補であり、316LVM及び2205等のステンレス鋼;MP35N、35NLT及びL605を含むコバルト系及びコバルトクロム合金;Nitinol(NiTi)、NiTi3元及び4元合金、及びチタンベータ合金を含む超可塑性合金;チタン合金;及びマグネシウム、亜鉛、鉄及びそれらの合金を含む生体吸収性金属が含まれる。これらの金属のうちのいずれも、DFT(上述)材料及び被覆ワイヤ材料等の、一体構造ワイヤまたは複合ワイヤ材料として形成可能である。
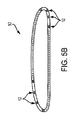
上述して図4に示したように、構成体10の例示的用途は、胸骨切開閉鎖プレート20に関連している。図示の実施形態では、閉鎖プレート20は、骨接合用ネジ24により切開部両側で患者の胸骨に固定され得、更にプレート20及び切開部両側の胸骨の隣接部分の周囲に構成体10を巻付けて締結部を形成することにより固定され得る。圧着具22は図示のように、構成体10の端部を固定してループを閉鎖するように使用し得る。好都合なことに、このような状況での構成体10の使用によって、平坦化及び自己調整された構成要素12(図5B)とこれに隣接する骨または組織の表面との間の接触表面積が最小化されることにより、隣接する骨での応力が最小化される。
Claims (15)
- 多繊条管状構成体であって、
外力の不存在下で内径、外径及び軸方向長さを画成する管状構成体に編込まれるかまたは編組された複数の構成要素を備え、
前記構成要素は各々、実質上相互に平行に延在するそれぞれの長手方向軸を有する複数の金属繊条体を備え、
前記管状構成体の端部に形成された少なくとも1つの摩擦ばめ嵌合部を更に備える、
多繊条管状構成体。 - 多繊条管状構成体であって、
外力の不存在下で内径、外径及び軸方向長さを画成する管状構成体に編込まれるかまたは編組された複数の構成要素を備え、前記構成要素は各々、実質上相互に平行に延在するそれぞれの長手方向軸を有する複数の金属繊条体を備え、
前記構成要素の前記複数の金属繊条体のうちの少なくとも1本は、吸収性金属で形成される、
多繊条管状構成体。 - 前記構成要素は、前記複数の金属繊条体の前記長手方向軸が各々実質上相互に平行に延在する螺旋を画成する撚合せケーブルである、請求項1又は2に記載の多繊条管状構成体。
- 前記構成要素の前記複数の金属繊条体は、2本〜343本の繊条体を備える、請求項1又は2に記載の多繊条管状構成体。
- 前記管状構成体の前記内径は、0.010インチ〜0.200インチである、請求項1又は2に記載の多繊条管状構成体。
- 前記管状構成体の前記外径は、0.014インチ〜0.208インチである、請求項5に記載の多繊条管状構成体。
- 前記複数の構成要素は、8本〜128本の構成要素を備える、請求項1又は2に記載の多繊条管状構成体。
- 前記複数の構成要素は、隣接する構成要素間のそれぞれの交差箇所で複数の打込みを画成し、前記打込みの数は前記管状構成体の外面に沿って軸方向インチ距離当り1〜50である、請求項1又は2に記載の多繊条管状構成体。
- 前記管状構成体の端部に形成された少なくとも1つの摩擦ばめ嵌合部を更に備える、請求項2に記載の多繊条管状構成体。
- 前記摩擦ばめ嵌合部は142lbf未満の分離力によっては前記管状構成体から脱離され得ない、請求項1又は9に記載の多繊条管状構成体。
- 前記吸収性金属は、マグネシウム、亜鉛、鉄及びそれらの合金のうちの少なくとも1種から成る、請求項2又は9に記載の多繊条管状構成体。
- 前記構成要素の前記複数の金属繊条体のうちの少なくとも1本は、ステンレス鋼で形成される、請求項1又は2に記載の多繊条管状構成体。
- 前記構成要素の前記複数の金属繊条体のうちの少なくとも1本は、超可塑性合金で形成される、請求項1又は2に記載の多繊条管状構成体。
- 前記超可塑性合金は、Nitinolから成る、請求項13に記載の多繊条管状構成体。
- 前記構成要素の前記複数の金属繊条体のうちの少なくとも1本は、コバルト系合金また
はコバルトクロム合金で形成される、請求項1又は2に記載の多繊条管状構成体。
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201662286062P | 2016-01-22 | 2016-01-22 | |
| US62/286,062 | 2016-01-22 | ||
| PCT/US2017/014355 WO2017127692A1 (en) | 2016-01-22 | 2017-01-20 | Woven or braided tubular metal construct |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2019503790A JP2019503790A (ja) | 2019-02-14 |
| JP6906529B2 true JP6906529B2 (ja) | 2021-07-21 |
Family
ID=59362082
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2018537852A Active JP6906529B2 (ja) | 2016-01-22 | 2017-01-20 | 編込みまたは編組管状金属構成体 |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US11497538B2 (ja) |
| EP (1) | EP3405145B1 (ja) |
| JP (1) | JP6906529B2 (ja) |
| CN (1) | CN108738305A (ja) |
| ES (1) | ES2921698T3 (ja) |
| PL (1) | PL3405145T3 (ja) |
| WO (1) | WO2017127692A1 (ja) |
Families Citing this family (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9561064B2 (en) | 2012-11-21 | 2017-02-07 | Pioneer Surgical Technology, Inc. | Bone plate system and method |
| US10765465B2 (en) | 2012-11-21 | 2020-09-08 | A&E Advanced Closure Systems, Llc | Tensioning instrument |
| US9999454B2 (en) | 2013-12-05 | 2018-06-19 | A&E Advanced Closure Systems, Llc | Bone plate system and method |
| US10314635B2 (en) | 2014-05-28 | 2019-06-11 | A&E Advanced Closure Systems, Llc | Tensioning instruments |
| AU2017210022B2 (en) | 2016-01-22 | 2021-05-13 | A&E Advanced Closure Systems, Llc | Bone plate having a connector and a connector for a surgical loop |
| CN108738305A (zh) | 2016-01-22 | 2018-11-02 | 韦恩堡金属研究产品公司 | 机织或编织管状金属结构 |
| WO2018022283A1 (en) | 2016-07-29 | 2018-02-01 | Pioneer Surgical Technology, Inc. | Surgical cable tensioner |
| CN107574526B (zh) * | 2017-10-10 | 2019-12-10 | 东华大学 | 结构相转变与交叉转动耦合的径向缩胀管织物及其制备方法与应用 |
| JPWO2019073901A1 (ja) * | 2017-10-11 | 2020-09-17 | 株式会社 京都医療設計 | 動脈瘤塞栓用の部材 |
| US20200146852A1 (en) * | 2018-11-13 | 2020-05-14 | Icad Endovascular Llc | Systems and methods for delivery retrievable stents |
| US11251406B2 (en) | 2019-03-07 | 2022-02-15 | Vitro Flat Glass Llc | Borosilicate light extraction region |
| US20220354489A1 (en) | 2021-05-10 | 2022-11-10 | Cilag Gmbh International | Absorbable staple comprising strain limiting features |
| CN114897902B (zh) * | 2022-07-13 | 2022-11-11 | 深圳金正方科技股份有限公司 | 基于多摄像头的bwfrp管道在线监测方法以及系统 |
Family Cites Families (26)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3125095A (en) * | 1964-03-17 | Flexible stainless steel sutures | ||
| US4184784A (en) * | 1978-07-03 | 1980-01-22 | The Bendix Corporation | Termination and method of terminating ropes or cables of aramid fiber or the like |
| US5358498A (en) * | 1990-02-01 | 1994-10-25 | Deknatel Technology Corporation, Inc. | Needled suture |
| US5318575A (en) * | 1992-02-03 | 1994-06-07 | United States Surgical Corporation | Method of using a surgical repair suture product |
| US5541380A (en) | 1994-09-16 | 1996-07-30 | Methode Electronics, Inc. | Braided cable solidification |
| US5718159A (en) * | 1996-04-30 | 1998-02-17 | Schneider (Usa) Inc. | Process for manufacturing three-dimensional braided covered stent |
| US5755704A (en) * | 1996-10-29 | 1998-05-26 | Medtronic, Inc. | Thinwall guide catheter |
| US6068648A (en) | 1998-01-26 | 2000-05-30 | Orthodyne, Inc. | Tissue anchoring system and method |
| US6494907B1 (en) * | 1998-04-28 | 2002-12-17 | Intratherapeutics, Inc. | Braided stent |
| EP1750619B1 (en) | 2004-05-25 | 2013-07-24 | Covidien LP | Flexible vascular occluding device |
| WO2006053270A2 (en) * | 2004-11-10 | 2006-05-18 | Boston Scientific Limited | Atraumatic stent with reduced deployment force, method for making the same and method and apparatus for deploying and positioning the stent |
| US8308761B2 (en) * | 2004-12-06 | 2012-11-13 | Ecole De Technologie Superieure | Binding component |
| KR101387070B1 (ko) * | 2005-05-04 | 2014-04-18 | 진테스 게엠베하 | 결합 부재 |
| US8152833B2 (en) * | 2006-02-22 | 2012-04-10 | Tyco Healthcare Group Lp | Embolic protection systems having radiopaque filter mesh |
| US20120109129A1 (en) * | 2010-11-02 | 2012-05-03 | Bernstein Oren S | Replacement system for a surgical wire |
| US8151682B2 (en) * | 2009-01-26 | 2012-04-10 | Boston Scientific Scimed, Inc. | Atraumatic stent and method and apparatus for making the same |
| KR20110088975A (ko) | 2010-01-29 | 2011-08-04 | 주식회사 뉴로벤션 | 스텐트 |
| EP2585125B1 (en) * | 2010-06-25 | 2014-11-19 | Fort Wayne Metals Research Products Corporation | Biodegradable composite wire for medical devices |
| WO2012027500A2 (en) * | 2010-08-24 | 2012-03-01 | Edwards Lifesciences Corporation | Flexible annuloplasty ring with select control points |
| JP6546718B2 (ja) * | 2011-01-17 | 2019-07-17 | メタクティブ・メディカル・インコーポレイテッドMetactive Medical, Inc. | ブロックステントデバイスおよび使用方法 |
| CN102650084A (zh) * | 2011-02-28 | 2012-08-29 | Tyco医疗健康集团 | 包含热塑弹性体共聚物和聚烯烃单丝的纱 |
| JP6485642B2 (ja) * | 2012-07-10 | 2019-03-20 | フォート ウェイン メタルス リサーチ プロダクツ コーポレーション | 医療デバイス用生分解性合金ワイヤ |
| JP2014161650A (ja) | 2013-02-27 | 2014-09-08 | Osaka Coat Rope Kk | 骨結束用索体、及び骨結束用索体の製造方法 |
| CN105658175B (zh) | 2013-08-07 | 2017-09-19 | 波士顿科学国际有限公司 | 用于聚合物支架的硅酮回流阀 |
| JP2015119893A (ja) * | 2013-12-25 | 2015-07-02 | 堤総研株式会社 | 生体器具 |
| CN108738305A (zh) | 2016-01-22 | 2018-11-02 | 韦恩堡金属研究产品公司 | 机织或编织管状金属结构 |
-
2017
- 2017-01-20 CN CN201780007543.8A patent/CN108738305A/zh not_active Withdrawn
- 2017-01-20 JP JP2018537852A patent/JP6906529B2/ja active Active
- 2017-01-20 US US16/069,643 patent/US11497538B2/en active Active
- 2017-01-20 WO PCT/US2017/014355 patent/WO2017127692A1/en active Application Filing
- 2017-01-20 EP EP17742019.7A patent/EP3405145B1/en active Active
- 2017-01-20 PL PL17742019.7T patent/PL3405145T3/pl unknown
- 2017-01-20 ES ES17742019T patent/ES2921698T3/es active Active
Also Published As
| Publication number | Publication date |
|---|---|
| EP3405145A4 (en) | 2019-12-11 |
| KR20180111844A (ko) | 2018-10-11 |
| ES2921698T3 (es) | 2022-08-30 |
| US11497538B2 (en) | 2022-11-15 |
| PL3405145T3 (pl) | 2022-10-24 |
| JP2019503790A (ja) | 2019-02-14 |
| EP3405145B1 (en) | 2022-06-22 |
| EP3405145A1 (en) | 2018-11-28 |
| US20190015142A1 (en) | 2019-01-17 |
| WO2017127692A1 (en) | 2017-07-27 |
| CN108738305A (zh) | 2018-11-02 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6906529B2 (ja) | 編込みまたは編組管状金属構成体 | |
| US5417690A (en) | Surgical cable | |
| CA2587737C (en) | Binding component | |
| US5997542A (en) | Surgical wire assembly and method of use | |
| JP4074432B2 (ja) | 血管閉塞コイル | |
| US6589246B1 (en) | Method of applying an active compressive force continuously across a fracture | |
| CN104053409B (zh) | 可植入形状记忆闭塞器械及其制造和使用方法 | |
| US20090216326A1 (en) | Tendon repair apparatus and method | |
| JP5561273B2 (ja) | 塞栓コイル | |
| US20090204118A1 (en) | Surgical cable with malleable leader segment | |
| JP5479242B2 (ja) | 医療用線状部材 | |
| EP2706953B1 (en) | External support for elongated bodily vessels | |
| JP5631722B2 (ja) | 医療用線状部材 | |
| JP6190994B2 (ja) | 骨癒合のための医療用線状部材 | |
| US9936991B2 (en) | Bone fracture treatment apparatus and method | |
| KR102655780B1 (ko) | 직조 또는 편조된 튜브형 금속 구성체 | |
| WO2018183862A1 (en) | Small diameter cable | |
| JP5916138B2 (ja) | 外科用ケーブル及びその製造方法 | |
| JP6127165B2 (ja) | 外科用ケーブル | |
| WO2016013123A1 (ja) | 骨結束用索体、及び骨結束用索体の製造方法 | |
| JP2014161650A (ja) | 骨結束用索体、及び骨結束用索体の製造方法 | |
| II | BINDING COMPONENT | |
| WO2019197684A1 (en) | A lead anchor device |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20200120 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20200413 |
|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20201224 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20210112 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20210412 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20210520 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20210608 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20210629 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 6906529 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| S533 | Written request for registration of change of name |
Free format text: JAPANESE INTERMEDIATE CODE: R313533 |
|
| R350 | Written notification of registration of transfer |
Free format text: JAPANESE INTERMEDIATE CODE: R350 |