JP5489627B2 - リチウム二次電池 - Google Patents
リチウム二次電池 Download PDFInfo
- Publication number
- JP5489627B2 JP5489627B2 JP2009230909A JP2009230909A JP5489627B2 JP 5489627 B2 JP5489627 B2 JP 5489627B2 JP 2009230909 A JP2009230909 A JP 2009230909A JP 2009230909 A JP2009230909 A JP 2009230909A JP 5489627 B2 JP5489627 B2 JP 5489627B2
- Authority
- JP
- Japan
- Prior art keywords
- positive electrode
- active material
- electrode active
- lithium secondary
- potential
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 229910052744 lithium Inorganic materials 0.000 title claims description 67
- WHXSMMKQMYFTQS-UHFFFAOYSA-N Lithium Chemical compound [Li] WHXSMMKQMYFTQS-UHFFFAOYSA-N 0.000 title claims description 64
- 239000007774 positive electrode material Substances 0.000 claims description 87
- 239000000203 mixture Substances 0.000 claims description 55
- 239000013078 crystal Substances 0.000 claims description 23
- 239000002245 particle Substances 0.000 claims description 12
- 239000011255 nonaqueous electrolyte Substances 0.000 claims description 11
- 238000002441 X-ray diffraction Methods 0.000 claims description 5
- 229910052759 nickel Inorganic materials 0.000 claims description 5
- PXHVJJICTQNCMI-UHFFFAOYSA-N nickel Substances [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 description 48
- 239000008151 electrolyte solution Substances 0.000 description 34
- 238000000354 decomposition reaction Methods 0.000 description 30
- 238000012360 testing method Methods 0.000 description 28
- 239000000843 powder Substances 0.000 description 24
- 208000028659 discharge Diseases 0.000 description 20
- 239000000463 material Substances 0.000 description 18
- 230000000052 comparative effect Effects 0.000 description 16
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 13
- 229910052796 boron Inorganic materials 0.000 description 13
- 239000011521 glass Substances 0.000 description 13
- -1 lithium transition metal Chemical class 0.000 description 12
- 239000004020 conductor Substances 0.000 description 11
- 239000003792 electrolyte Substances 0.000 description 11
- 229910052710 silicon Inorganic materials 0.000 description 11
- 229910052718 tin Inorganic materials 0.000 description 11
- 239000006230 acetylene black Substances 0.000 description 9
- 238000000034 method Methods 0.000 description 9
- ZOXJGFHDIHLPTG-UHFFFAOYSA-N Boron Chemical compound [B] ZOXJGFHDIHLPTG-UHFFFAOYSA-N 0.000 description 7
- 229910052782 aluminium Inorganic materials 0.000 description 7
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 7
- 230000007423 decrease Effects 0.000 description 7
- 229910052751 metal Inorganic materials 0.000 description 7
- 239000002184 metal Substances 0.000 description 7
- 239000002904 solvent Substances 0.000 description 7
- WMFOQBRAJBCJND-UHFFFAOYSA-M Lithium hydroxide Chemical compound [Li+].[OH-] WMFOQBRAJBCJND-UHFFFAOYSA-M 0.000 description 6
- 239000011651 chromium Substances 0.000 description 6
- 238000004519 manufacturing process Methods 0.000 description 6
- 239000010955 niobium Substances 0.000 description 6
- 239000010936 titanium Substances 0.000 description 6
- HBBGRARXTFLTSG-UHFFFAOYSA-N Lithium ion Chemical compound [Li+] HBBGRARXTFLTSG-UHFFFAOYSA-N 0.000 description 5
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical compound [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 description 5
- ATJFFYVFTNAWJD-UHFFFAOYSA-N Tin Chemical compound [Sn] ATJFFYVFTNAWJD-UHFFFAOYSA-N 0.000 description 5
- 238000007599 discharging Methods 0.000 description 5
- 239000011888 foil Substances 0.000 description 5
- 229910001416 lithium ion Inorganic materials 0.000 description 5
- 239000010703 silicon Substances 0.000 description 5
- XTHFKEDIFFGKHM-UHFFFAOYSA-N Dimethoxyethane Chemical compound COCCOC XTHFKEDIFFGKHM-UHFFFAOYSA-N 0.000 description 4
- KMTRUDSVKNLOMY-UHFFFAOYSA-N Ethylene carbonate Chemical compound O=C1OCCO1 KMTRUDSVKNLOMY-UHFFFAOYSA-N 0.000 description 4
- 229910019142 PO4 Inorganic materials 0.000 description 4
- OAICVXFJPJFONN-UHFFFAOYSA-N Phosphorus Chemical compound [P] OAICVXFJPJFONN-UHFFFAOYSA-N 0.000 description 4
- 239000004698 Polyethylene Substances 0.000 description 4
- 239000003125 aqueous solvent Substances 0.000 description 4
- 239000000470 constituent Substances 0.000 description 4
- 238000010586 diagram Methods 0.000 description 4
- 239000010452 phosphate Substances 0.000 description 4
- 229910052698 phosphorus Inorganic materials 0.000 description 4
- 239000011574 phosphorus Substances 0.000 description 4
- 229920000573 polyethylene Polymers 0.000 description 4
- RUOJZAUFBMNUDX-UHFFFAOYSA-N propylene carbonate Chemical compound CC1COC(=O)O1 RUOJZAUFBMNUDX-UHFFFAOYSA-N 0.000 description 4
- 238000010791 quenching Methods 0.000 description 4
- 239000002994 raw material Substances 0.000 description 4
- 150000003839 salts Chemical class 0.000 description 4
- 238000006467 substitution reaction Methods 0.000 description 4
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 3
- VYZAMTAEIAYCRO-UHFFFAOYSA-N Chromium Chemical compound [Cr] VYZAMTAEIAYCRO-UHFFFAOYSA-N 0.000 description 3
- ZOKXTWBITQBERF-UHFFFAOYSA-N Molybdenum Chemical compound [Mo] ZOKXTWBITQBERF-UHFFFAOYSA-N 0.000 description 3
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 3
- 239000002033 PVDF binder Substances 0.000 description 3
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical compound [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 description 3
- 229910052785 arsenic Inorganic materials 0.000 description 3
- RQNWIZPPADIBDY-UHFFFAOYSA-N arsenic atom Chemical compound [As] RQNWIZPPADIBDY-UHFFFAOYSA-N 0.000 description 3
- 239000011230 binding agent Substances 0.000 description 3
- 230000015572 biosynthetic process Effects 0.000 description 3
- 239000003575 carbonaceous material Substances 0.000 description 3
- 229910052804 chromium Inorganic materials 0.000 description 3
- 229910052732 germanium Inorganic materials 0.000 description 3
- GNPVGFCGXDBREM-UHFFFAOYSA-N germanium atom Chemical compound [Ge] GNPVGFCGXDBREM-UHFFFAOYSA-N 0.000 description 3
- 229910052748 manganese Inorganic materials 0.000 description 3
- 238000002156 mixing Methods 0.000 description 3
- 229910052750 molybdenum Inorganic materials 0.000 description 3
- 239000011733 molybdenum Substances 0.000 description 3
- 229910052758 niobium Inorganic materials 0.000 description 3
- GUCVJGMIXFAOAE-UHFFFAOYSA-N niobium atom Chemical compound [Nb] GUCVJGMIXFAOAE-UHFFFAOYSA-N 0.000 description 3
- 229920002981 polyvinylidene fluoride Polymers 0.000 description 3
- 230000000171 quenching effect Effects 0.000 description 3
- 239000007858 starting material Substances 0.000 description 3
- 238000005211 surface analysis Methods 0.000 description 3
- 238000003786 synthesis reaction Methods 0.000 description 3
- 229910052715 tantalum Inorganic materials 0.000 description 3
- GUVRBAGPIYLISA-UHFFFAOYSA-N tantalum atom Chemical compound [Ta] GUVRBAGPIYLISA-UHFFFAOYSA-N 0.000 description 3
- 229910052719 titanium Inorganic materials 0.000 description 3
- 229910052723 transition metal Inorganic materials 0.000 description 3
- 229910052720 vanadium Inorganic materials 0.000 description 3
- GPPXJZIENCGNKB-UHFFFAOYSA-N vanadium Chemical compound [V]#[V] GPPXJZIENCGNKB-UHFFFAOYSA-N 0.000 description 3
- 238000004804 winding Methods 0.000 description 3
- YEJRWHAVMIAJKC-UHFFFAOYSA-N 4-Butyrolactone Chemical compound O=C1CCCO1 YEJRWHAVMIAJKC-UHFFFAOYSA-N 0.000 description 2
- 229920000049 Carbon (fiber) Polymers 0.000 description 2
- OIFBSDVPJOWBCH-UHFFFAOYSA-N Diethyl carbonate Chemical compound CCOC(=O)OCC OIFBSDVPJOWBCH-UHFFFAOYSA-N 0.000 description 2
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 2
- 229910013870 LiPF 6 Inorganic materials 0.000 description 2
- SECXISVLQFMRJM-UHFFFAOYSA-N N-Methylpyrrolidone Chemical compound CN1CCCC1=O SECXISVLQFMRJM-UHFFFAOYSA-N 0.000 description 2
- 239000004743 Polypropylene Substances 0.000 description 2
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 2
- IDSMHEZTLOUMLM-UHFFFAOYSA-N [Li].[O].[Co] Chemical class [Li].[O].[Co] IDSMHEZTLOUMLM-UHFFFAOYSA-N 0.000 description 2
- 239000011149 active material Substances 0.000 description 2
- 230000005540 biological transmission Effects 0.000 description 2
- 239000004917 carbon fiber Substances 0.000 description 2
- 238000010276 construction Methods 0.000 description 2
- 230000003247 decreasing effect Effects 0.000 description 2
- IEJIGPNLZYLLBP-UHFFFAOYSA-N dimethyl carbonate Chemical compound COC(=O)OC IEJIGPNLZYLLBP-UHFFFAOYSA-N 0.000 description 2
- 230000000694 effects Effects 0.000 description 2
- 239000007772 electrode material Substances 0.000 description 2
- 238000002149 energy-dispersive X-ray emission spectroscopy Methods 0.000 description 2
- JBTWLSYIZRCDFO-UHFFFAOYSA-N ethyl methyl carbonate Chemical compound CCOC(=O)OC JBTWLSYIZRCDFO-UHFFFAOYSA-N 0.000 description 2
- XLYOFNOQVPJJNP-UHFFFAOYSA-M hydroxide Chemical compound [OH-] XLYOFNOQVPJJNP-UHFFFAOYSA-M 0.000 description 2
- 239000007788 liquid Substances 0.000 description 2
- 229910000625 lithium cobalt oxide Inorganic materials 0.000 description 2
- URIIGZKXFBNRAU-UHFFFAOYSA-N lithium;oxonickel Chemical class [Li].[Ni]=O URIIGZKXFBNRAU-UHFFFAOYSA-N 0.000 description 2
- 238000007578 melt-quenching technique Methods 0.000 description 2
- 238000002844 melting Methods 0.000 description 2
- 230000008018 melting Effects 0.000 description 2
- 239000002905 metal composite material Substances 0.000 description 2
- VNWKTOKETHGBQD-UHFFFAOYSA-N methane Chemical compound C VNWKTOKETHGBQD-UHFFFAOYSA-N 0.000 description 2
- 239000007773 negative electrode material Substances 0.000 description 2
- 230000003647 oxidation Effects 0.000 description 2
- 238000007254 oxidation reaction Methods 0.000 description 2
- 239000002861 polymer material Substances 0.000 description 2
- 229920001155 polypropylene Polymers 0.000 description 2
- 239000003115 supporting electrolyte Substances 0.000 description 2
- LZDKZFUFMNSQCJ-UHFFFAOYSA-N 1,2-diethoxyethane Chemical compound CCOCCOCC LZDKZFUFMNSQCJ-UHFFFAOYSA-N 0.000 description 1
- WNXJIVFYUVYPPR-UHFFFAOYSA-N 1,3-dioxolane Chemical compound C1COCO1 WNXJIVFYUVYPPR-UHFFFAOYSA-N 0.000 description 1
- RYHBNJHYFVUHQT-UHFFFAOYSA-N 1,4-Dioxane Chemical compound C1COCCO1 RYHBNJHYFVUHQT-UHFFFAOYSA-N 0.000 description 1
- JWUJQDFVADABEY-UHFFFAOYSA-N 2-methyltetrahydrofuran Chemical compound CC1CCCO1 JWUJQDFVADABEY-UHFFFAOYSA-N 0.000 description 1
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 1
- 229910018068 Li 2 O Inorganic materials 0.000 description 1
- 229910013063 LiBF 4 Inorganic materials 0.000 description 1
- 229910013372 LiC 4 Inorganic materials 0.000 description 1
- 229910013684 LiClO 4 Inorganic materials 0.000 description 1
- 229910013528 LiN(SO2 CF3)2 Inorganic materials 0.000 description 1
- 229910013385 LiN(SO2C2F5)2 Inorganic materials 0.000 description 1
- 229910013086 LiNiPO Inorganic materials 0.000 description 1
- 229910004298 SiO 2 Inorganic materials 0.000 description 1
- 229910006404 SnO 2 Inorganic materials 0.000 description 1
- 229910000831 Steel Inorganic materials 0.000 description 1
- 229910000147 aluminium phosphate Inorganic materials 0.000 description 1
- 238000005280 amorphization Methods 0.000 description 1
- 229910003481 amorphous carbon Inorganic materials 0.000 description 1
- 239000000010 aprotic solvent Substances 0.000 description 1
- 238000000498 ball milling Methods 0.000 description 1
- 229910052799 carbon Inorganic materials 0.000 description 1
- 239000003660 carbonate based solvent Substances 0.000 description 1
- 150000004649 carbonic acid derivatives Chemical class 0.000 description 1
- 239000006182 cathode active material Substances 0.000 description 1
- 238000006243 chemical reaction Methods 0.000 description 1
- 239000002131 composite material Substances 0.000 description 1
- 230000001143 conditioned effect Effects 0.000 description 1
- 230000003750 conditioning effect Effects 0.000 description 1
- 238000007796 conventional method Methods 0.000 description 1
- 239000010949 copper Substances 0.000 description 1
- 239000011889 copper foil Substances 0.000 description 1
- 238000007872 degassing Methods 0.000 description 1
- 238000013461 design Methods 0.000 description 1
- SBZXBUIDTXKZTM-UHFFFAOYSA-N diglyme Chemical compound COCCOCCOC SBZXBUIDTXKZTM-UHFFFAOYSA-N 0.000 description 1
- 238000001035 drying Methods 0.000 description 1
- 238000010894 electron beam technology Methods 0.000 description 1
- 238000002003 electron diffraction Methods 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 150000002148 esters Chemical class 0.000 description 1
- 150000002170 ethers Chemical class 0.000 description 1
- 238000011156 evaluation Methods 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 239000000284 extract Substances 0.000 description 1
- 238000000605 extraction Methods 0.000 description 1
- 239000000446 fuel Substances 0.000 description 1
- 239000010439 graphite Substances 0.000 description 1
- 229910002804 graphite Inorganic materials 0.000 description 1
- 238000002347 injection Methods 0.000 description 1
- 239000007924 injection Substances 0.000 description 1
- 238000003780 insertion Methods 0.000 description 1
- 230000037431 insertion Effects 0.000 description 1
- 238000007689 inspection Methods 0.000 description 1
- 150000002596 lactones Chemical class 0.000 description 1
- 229910003002 lithium salt Inorganic materials 0.000 description 1
- 159000000002 lithium salts Chemical class 0.000 description 1
- 239000007769 metal material Substances 0.000 description 1
- 238000003801 milling Methods 0.000 description 1
- 239000012046 mixed solvent Substances 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 238000000465 moulding Methods 0.000 description 1
- 230000007935 neutral effect Effects 0.000 description 1
- 150000002825 nitriles Chemical class 0.000 description 1
- LYGJENNIWJXYER-UHFFFAOYSA-N nitromethane Chemical compound C[N+]([O-])=O LYGJENNIWJXYER-UHFFFAOYSA-N 0.000 description 1
- 239000010450 olivine Substances 0.000 description 1
- 229910052609 olivine Inorganic materials 0.000 description 1
- NBIIXXVUZAFLBC-UHFFFAOYSA-N phosphoric acid Substances OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 1
- 150000003013 phosphoric acid derivatives Chemical class 0.000 description 1
- 229920001721 polyimide Polymers 0.000 description 1
- 239000009719 polyimide resin Substances 0.000 description 1
- 229920005672 polyolefin resin Polymers 0.000 description 1
- FVSKHRXBFJPNKK-UHFFFAOYSA-N propionitrile Chemical compound CCC#N FVSKHRXBFJPNKK-UHFFFAOYSA-N 0.000 description 1
- 229920005989 resin Polymers 0.000 description 1
- 239000011347 resin Substances 0.000 description 1
- 238000007789 sealing Methods 0.000 description 1
- 229910001220 stainless steel Inorganic materials 0.000 description 1
- 239000010935 stainless steel Substances 0.000 description 1
- 239000010959 steel Substances 0.000 description 1
- HXJUTPCZVOIRIF-UHFFFAOYSA-N sulfolane Chemical compound O=S1(=O)CCCC1 HXJUTPCZVOIRIF-UHFFFAOYSA-N 0.000 description 1
- 150000003457 sulfones Chemical class 0.000 description 1
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 1
- 229910000314 transition metal oxide Inorganic materials 0.000 description 1
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 1
- 238000003466 welding Methods 0.000 description 1
Images
Classifications
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/48—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides
- H01M4/50—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides of manganese
- H01M4/505—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides of manganese of mixed oxides or hydroxides containing manganese for inserting or intercalating light metals, e.g. LiMn2O4 or LiMn2OxFy
-
- C—CHEMISTRY; METALLURGY
- C03—GLASS; MINERAL OR SLAG WOOL
- C03C—CHEMICAL COMPOSITION OF GLASSES, GLAZES OR VITREOUS ENAMELS; SURFACE TREATMENT OF GLASS; SURFACE TREATMENT OF FIBRES OR FILAMENTS MADE FROM GLASS, MINERALS OR SLAGS; JOINING GLASS TO GLASS OR OTHER MATERIALS
- C03C3/00—Glass compositions
- C03C3/12—Silica-free oxide glass compositions
- C03C3/16—Silica-free oxide glass compositions containing phosphorus
-
- C—CHEMISTRY; METALLURGY
- C03—GLASS; MINERAL OR SLAG WOOL
- C03C—CHEMICAL COMPOSITION OF GLASSES, GLAZES OR VITREOUS ENAMELS; SURFACE TREATMENT OF GLASS; SURFACE TREATMENT OF FIBRES OR FILAMENTS MADE FROM GLASS, MINERALS OR SLAGS; JOINING GLASS TO GLASS OR OTHER MATERIALS
- C03C3/00—Glass compositions
- C03C3/12—Silica-free oxide glass compositions
- C03C3/16—Silica-free oxide glass compositions containing phosphorus
- C03C3/19—Silica-free oxide glass compositions containing phosphorus containing boron
-
- C—CHEMISTRY; METALLURGY
- C03—GLASS; MINERAL OR SLAG WOOL
- C03C—CHEMICAL COMPOSITION OF GLASSES, GLAZES OR VITREOUS ENAMELS; SURFACE TREATMENT OF GLASS; SURFACE TREATMENT OF FIBRES OR FILAMENTS MADE FROM GLASS, MINERALS OR SLAGS; JOINING GLASS TO GLASS OR OTHER MATERIALS
- C03C4/00—Compositions for glass with special properties
- C03C4/14—Compositions for glass with special properties for electro-conductive glass
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M10/00—Secondary cells; Manufacture thereof
- H01M10/05—Accumulators with non-aqueous electrolyte
- H01M10/052—Li-accumulators
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/48—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides
- H01M4/52—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides of nickel, cobalt or iron
- H01M4/525—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides of nickel, cobalt or iron of mixed oxides or hydroxides containing iron, cobalt or nickel for inserting or intercalating light metals, e.g. LiNiO2, LiCoO2 or LiCoOxFy
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M10/00—Secondary cells; Manufacture thereof
- H01M10/05—Accumulators with non-aqueous electrolyte
- H01M10/056—Accumulators with non-aqueous electrolyte characterised by the materials used as electrolytes, e.g. mixed inorganic/organic electrolytes
- H01M10/0564—Accumulators with non-aqueous electrolyte characterised by the materials used as electrolytes, e.g. mixed inorganic/organic electrolytes the electrolyte being constituted of organic materials only
- H01M10/0566—Liquid materials
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/13—Electrodes for accumulators with non-aqueous electrolyte, e.g. for lithium-accumulators; Processes of manufacture thereof
- H01M4/131—Electrodes based on mixed oxides or hydroxides, or on mixtures of oxides or hydroxides, e.g. LiCoOx
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/58—Selection of substances as active materials, active masses, active liquids of inorganic compounds other than oxides or hydroxides, e.g. sulfides, selenides, tellurides, halogenides or LiCoFy; of polyanionic structures, e.g. phosphates, silicates or borates
- H01M4/5825—Oxygenated metallic salts or polyanionic structures, e.g. borates, phosphates, silicates, olivines
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E60/00—Enabling technologies; Technologies with a potential or indirect contribution to GHG emissions mitigation
- Y02E60/10—Energy storage using batteries
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02T—CLIMATE CHANGE MITIGATION TECHNOLOGIES RELATED TO TRANSPORTATION
- Y02T10/00—Road transport of goods or passengers
- Y02T10/60—Other road transportation technologies with climate change mitigation effect
- Y02T10/70—Energy storage systems for electromobility, e.g. batteries
Description
本発明はかかる点に鑑みてなされたものであり、その主な目的は、高容量で充放電特性に優れたリチウム二次電池を提供することである。
上記正極活物質は、非晶質であって、以下の組成式:
LixA[PaM1−a]yOz (I)
(式中、AはMnまたはNiであり、MはPよりも電気陰性度が低いガラスフォーマー元素であり、x、y、z、aは、それぞれ、1<x≦2.5、0<y≦3、0≦a<1、z=(x+(Aの価数)+(Pの価数)×a×y+(Mの価数)×(1−a)×y)/2)である。);
で表わされる。
LixA[PaM1−a]yOz (I)
(1)非晶質であって、以下の組成式:
LixA[PaM1−a]yOz (I)
(上記式(I)中、AはMnまたはNiであり、MはPよりも電気陰性度が低いガラスフォーマー元素であり、x、y、z、aは、それぞれ、1<x≦2.5、0<y≦3、0≦a<1、z=(x+(Aの価数)+(Pの価数)×a×y+(Mの価数)×(1−a)×y)/2)である。);
で表わされる、リチウム二次電池用正極活物質。
(2)上記組成式(I)中のMは、B(ホウ素),Si(ケイ素),Sn(スズ),Ta(タンタル),Ti(チタン),Nb(ニオブ),Al(アルミニウム),V(バナジウム),Cr(クロム),Ge(ゲルマニウム),Mo(モリブデン)及びAs(ヒ素)の少なくとも一種の元素を含む、上記(1)に記載の正極活物質。
実施例1〜3および比較例1では、Li:Mn:P:Bが下記表1に示す構成比となるように、非晶質のLixMn[PaB1−a]yOzからなる正極活物質を合成した。具体的には、Li源としてのLiOHと、Mn源としてのMnOと、P源としてのP2O5と、B源としてのB2O3とを、これらの材料の構成成分が所定のモル比となるように秤量し、これを混合して原料混合物を得た。次に、原料混合物をAr雰囲気中にて1000℃〜1200℃で溶融し、単ロール溶融急冷装置で急冷することにより、非晶質のLixMn[PaB1−a]yOzからなる正極活物質を合成した。得られた非晶質のLixMn[PaB1−a]yOzを所定の粉砕装置で粉砕して粉末状の非晶質のMn系正極活物質を作製した。
上記得られた非晶質のLixMn[PaB1−a]yOz粉末に対して一般的なボールミル装置を用いて300rpm,3時間の条件でボールミル処理を行った。さらに、導電材としてのアセチレンブラックを、正極活物質とアセチレンブラックとの質量比が70:25となるように秤量して混合し、該混合物に対して300rpm,3時間の条件で更なるボールミル処理を行った。このようにして、平均粒径3μmの非晶質のLixMn[PaB1−a]yOzからなる正極活物質粉末を、上記導電材との混合物として得た。
実施例4,5および比較例2では、Li:Ni:P:Bが下記表2に示す構成比となるように、非晶質のLixNi[PaB1−a]yOzからなる正極活物質を合成した。具体的には、Li源としてのLiOHと、Ni源としてのNiOと、P源としてのP2O5と、B源としてのB2O3とを、これらの材料の構成成分が所定のモル比となるように秤量し、これを混合して原料混合物を得た。次に、原料混合物をAr雰囲気中にて1000℃〜1200℃で溶融し、単ロール溶融急冷装置で急冷することにより、非晶質のLixNi[PaB1−a]yOzからなる正極活物質を合成した。得られた非晶質のLixNi[PaB1−a]yOzを所定の粉砕装置で粉砕して粉末状の非晶質のNi系正極活物質を作製した。
上記得られた非晶質のLixNi[PaB1−a]yOz粉末に対して一般的なボールミル装置を用いて300rpm,3時間の条件でボールミル処理を行った。さらに、導電材としてのアセチレンブラックを、正極活物質とアセチレンブラックとの質量比が70:25となるように秤量して混合し、該混合物に対して300rpm,3時間の条件で更なるボールミル処理を行い、平均粒径3μmの非晶質のLixNi[PaB1−a]yOzからなる正極活物質粉末を、上記導電材との混合物として得た。
続いて、実施例1〜3および比較例1で得られたLixMn[PaB1−a]yOzからなる正極活物質粉末、及び実施例4,5および比較例2で得られたLixNi[PaB1−a]yOzからなる正極活物質粉末をそれぞれ用いてリチウム二次電池の作製し、充放電特性を評価した。具体的には、まず、上記得られた正極活物質粉末とアセチレンブラック(導電材)との混合物(正極活物質とアセチレンブラックとを70:25の質量比で含む。)に、結着剤としてのポリフッ化ビニリデン(PVdF)を、正極活物質とアセチレンブラックとPVdFとの質量比が70:25:5となるように秤量してN−メチルピロリドン(NMP)中で混ぜ合わせ、ペースト状の正極合材層用組成物を調製した。このペースト状正極合材層用組成物をアルミニウム箔(正極集電体)の片面に層状に塗布して乾燥することにより、該正極集電体の片面に正極合材層が設けられた正極シートを得た。
以上のように得られた実施例1〜5および比較例1、2の正極活物質粉末を用いて構築された試験用セルのそれぞれに対して、充放電特性試験を行った。具体的には、実施例1〜3及び比較例1のMn系正極活物質粉末を用いて構築された試験用セルに対しては、約25℃の環境下において、下記の(1)及び(2)の条件で充放電操作を行った。また、実施例4,5及び比較例2のNi系正極活物質粉末を用いて構築された試験用セルに対しては、約25℃の環境下において、下記の(1)及び(2)と同様の条件にて、ただし、充電時のカット電位を4.7V(vs.Li/Li+)、放電時のカット電位を2.0V(vs.Li/Li+)に変更して充放電操作を行った。
(2)0.1mA/cm2の定電流で正極の電位が1.5V(vs.Li/Li+)になるまで放電を行い、その後10分間休止した。
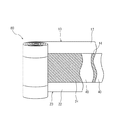
10 正極シート
12 正極集電体
14 正極合材層
20 負極シート
22 正極集電体
24 正極合材層
40 セパレータシート
50 容器
52 容器本体
54 蓋体
60 コインセル
70 正極端子
72 負極端子
80 捲回電極体
100 リチウム二次電池
Claims (2)
- 正極活物質を含む正極と、負極と、非水電解液とを備えたリチウム二次電池であって、
前記正極活物質は、非晶質であって、以下の組成式:
LixA[PaB1−a]yOz (I)
(前記式(I)中、AはNiであり、x、y、z、aは、それぞれ、1<x≦2.5、0<y≦3、0≦a<1、z=(x+(Aの価数)+(Pの価数)×a×y+(Bの価数)×(1−a)×y)/2)である。);
で表わされ、
ここで、前記組成式(I)中のAがNiである非晶質相を主体とし、Ni、NiO及びNi2Pのうち少なくとも一種の結晶相を含み、
前記正極活物質のX線回折パターンにおける2θ=43°付近にNi結晶相を示すピークを有しており、かつ、該Ni結晶相が正極活物質粒子の表面に偏在している、リチウム二次電池。 - 請求項1に記載のリチウム二次電池を搭載する車両。
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2009230909A JP5489627B2 (ja) | 2009-10-02 | 2009-10-02 | リチウム二次電池 |
| CN201080044460.4A CN102598387B (zh) | 2009-10-02 | 2010-09-30 | 锂二次电池 |
| US13/499,336 US8974963B2 (en) | 2009-10-02 | 2010-09-30 | Lithium secondary battery |
| PCT/IB2010/002481 WO2011039619A1 (en) | 2009-10-02 | 2010-09-30 | Lithium secondary battery |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2009230909A JP5489627B2 (ja) | 2009-10-02 | 2009-10-02 | リチウム二次電池 |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| JP2011081923A JP2011081923A (ja) | 2011-04-21 |
| JP2011081923A5 JP2011081923A5 (ja) | 2012-10-25 |
| JP5489627B2 true JP5489627B2 (ja) | 2014-05-14 |
Family
ID=43430907
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2009230909A Active JP5489627B2 (ja) | 2009-10-02 | 2009-10-02 | リチウム二次電池 |
Country Status (4)
| Country | Link |
|---|---|
| US (1) | US8974963B2 (ja) |
| JP (1) | JP5489627B2 (ja) |
| CN (1) | CN102598387B (ja) |
| WO (1) | WO2011039619A1 (ja) |
Families Citing this family (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP6051514B2 (ja) * | 2010-12-02 | 2016-12-27 | ソニー株式会社 | 固体電解質電池および正極活物質 |
| JP5821270B2 (ja) * | 2011-05-16 | 2015-11-24 | ソニー株式会社 | 固体電解質電池および正極活物質 |
| WO2014021395A1 (ja) * | 2012-07-31 | 2014-02-06 | シャープ株式会社 | 非水電解質二次電池用正極活物質 |
| CN103263939A (zh) * | 2013-06-08 | 2013-08-28 | 安徽工业大学 | 一种制备Ni2P催化剂的方法 |
| FR3016245B1 (fr) * | 2014-01-03 | 2021-07-16 | Commissariat Energie Atomique | Cellule electrochimique, systeme de stockage et de restitution d'energie electrique comprenant une telle cellule electrochimique et vehicule comprenant un tel systeme |
| JP6389688B2 (ja) * | 2014-08-06 | 2018-09-12 | Fdk株式会社 | リチウム二次電池用正極活物質およびリチウム二次電池 |
| JP6837278B2 (ja) * | 2015-02-25 | 2021-03-03 | 国立大学法人長岡技術科学大学 | アルカリイオン二次電池用正極活物質 |
Family Cites Families (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0849817A3 (en) * | 1996-12-20 | 1999-03-24 | Japan Storage Battery Company Limited | Positive active material for lithium battery having the same, and method for producing the same |
| JPH1125983A (ja) | 1997-07-04 | 1999-01-29 | Japan Storage Battery Co Ltd | リチウム電池用活物質 |
| US6749648B1 (en) * | 2000-06-19 | 2004-06-15 | Nanagram Corporation | Lithium metal oxides |
| EP1052712B1 (en) * | 1998-12-02 | 2010-02-24 | Panasonic Corporation | Non-aqueous electrolyte secondary cell |
| CA2270771A1 (fr) * | 1999-04-30 | 2000-10-30 | Hydro-Quebec | Nouveaux materiaux d'electrode presentant une conductivite de surface elevee |
| JP3639468B2 (ja) | 1999-08-04 | 2005-04-20 | 三洋電機株式会社 | リチウム二次電池 |
| US6815122B2 (en) * | 2002-03-06 | 2004-11-09 | Valence Technology, Inc. | Alkali transition metal phosphates and related electrode active materials |
| JP4153288B2 (ja) * | 2002-11-25 | 2008-09-24 | 日本電信電話株式会社 | 非水電解質二次電池 |
| US7008566B2 (en) * | 2003-04-08 | 2006-03-07 | Valence Technology, Inc. | Oligo phosphate-based electrode active materials and methods of making same |
| JP2005158673A (ja) * | 2003-10-31 | 2005-06-16 | Toyota Motor Corp | 電極活物質およびその製造方法ならびに非水電解質二次電池 |
| FR2864348B1 (fr) * | 2003-12-18 | 2006-03-10 | Commissariat Energie Atomique | Accumulateur au lithium presentant a la fois un potentiel electrique et une capacite d'insertion du lthium eleves. |
| JP4333752B2 (ja) | 2007-02-19 | 2009-09-16 | トヨタ自動車株式会社 | 電極活物質およびその製造方法 |
| JP4289406B2 (ja) * | 2007-02-19 | 2009-07-01 | トヨタ自動車株式会社 | 電極活物質およびその製造方法 |
| JP5164246B2 (ja) * | 2007-03-30 | 2013-03-21 | 国立大学法人九州大学 | 電極活物質およびリチウム二次電池 |
-
2009
- 2009-10-02 JP JP2009230909A patent/JP5489627B2/ja active Active
-
2010
- 2010-09-30 CN CN201080044460.4A patent/CN102598387B/zh active Active
- 2010-09-30 US US13/499,336 patent/US8974963B2/en active Active
- 2010-09-30 WO PCT/IB2010/002481 patent/WO2011039619A1/en active Application Filing
Also Published As
| Publication number | Publication date |
|---|---|
| CN102598387B (zh) | 2014-12-24 |
| CN102598387A (zh) | 2012-07-18 |
| US20120183857A1 (en) | 2012-07-19 |
| WO2011039619A1 (en) | 2011-04-07 |
| US8974963B2 (en) | 2015-03-10 |
| JP2011081923A (ja) | 2011-04-21 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| KR101702818B1 (ko) | 활성 재료, 비수성 전해질 전지 및 전지 팩 | |
| EP3540825B1 (en) | Lithium secondary battery | |
| JP6320809B2 (ja) | 正極活物質、非水電解質電池および電池パック | |
| JP5389143B2 (ja) | 非水電解質電池 | |
| KR101730957B1 (ko) | 음극 활물질, 그 제조방법, 이를 포함하는 리튬 이차 전지용 음극 및 이를 채용한 리튬 이차 전지 | |
| KR102572648B1 (ko) | 리튬전지용 복합양극활물질, 이를 포함하는 리튬전지용 양극 및 리튬전지 | |
| JP5489627B2 (ja) | リチウム二次電池 | |
| JP5605641B2 (ja) | リチウム二次電池 | |
| US20110217574A1 (en) | Cathode material for lithium secondary battery, lithium secondary battery, and secondary battery module using the battery | |
| JP2005317512A (ja) | 非水電解質電池 | |
| KR20100072047A (ko) | 정극 활물질, 리튬 2차 전지, 및 그 제조 방법 | |
| JP6615660B2 (ja) | 非水電解質電池、電池パック及び車両 | |
| JP2011071103A (ja) | 正極、非水電解質電池及び電池パック | |
| US20190165372A1 (en) | Positive electrode material and lithium secondary battery using the same | |
| JP2011165461A (ja) | リチウムイオン二次電池正極材料 | |
| JP2014029872A (ja) | 非水電解質電池 | |
| US20170170474A1 (en) | Lithium secondary battery | |
| JP2009218112A (ja) | 非水電解質二次電池及びその製造方法 | |
| JP2016051504A (ja) | 非水系二次電池用正極活物質、非水系二次電池用正極、非水系二次電池及び車載用非水系二次電池モジュール | |
| JP2015130272A (ja) | 非水系二次電池用正極、非水系二次電池用正極活物質、非水系二次電池及び車載用非水系二次電池モジュール | |
| JP7040832B1 (ja) | リチウムイオン二次電池用負極活物質、その製造方法、及びリチウムイオン二次電池用負極電極 | |
| JP2013080726A (ja) | 正極、非水電解質電池及び電池パック | |
| JP2018160416A (ja) | 電極、非水電解質電池、電池パック及び車両 | |
| JP2021157936A (ja) | 負極活物質、負極及び二次電池 | |
| CN112599728A (zh) | 电极、二次电池、电池包及车辆 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20120907 |
|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20120907 |
|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20131030 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20131031 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20140106 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20140206 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20140225 |
|
| R151 | Written notification of patent or utility model registration |
Ref document number: 5489627 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R151 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| S531 | Written request for registration of change of domicile |
Free format text: JAPANESE INTERMEDIATE CODE: R313531 |
|
| R350 | Written notification of registration of transfer |
Free format text: JAPANESE INTERMEDIATE CODE: R350 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |