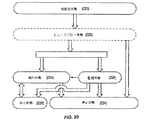
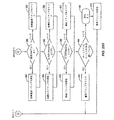
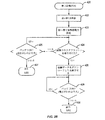
JP5406728B2 - マイボーム腺機能不全を治療するための熱治療のための方法、システム、および装置 - Google Patents
マイボーム腺機能不全を治療するための熱治療のための方法、システム、および装置 Download PDFInfo
- Publication number
- JP5406728B2 JP5406728B2 JP2009546506A JP2009546506A JP5406728B2 JP 5406728 B2 JP5406728 B2 JP 5406728B2 JP 2009546506 A JP2009546506 A JP 2009546506A JP 2009546506 A JP2009546506 A JP 2009546506A JP 5406728 B2 JP5406728 B2 JP 5406728B2
- Authority
- JP
- Japan
- Prior art keywords
- eyelid
- heat
- force
- temperature
- meibomian gland
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 206010065062 Meibomian gland dysfunction Diseases 0.000 title description 74
- 238000000034 method Methods 0.000 title description 32
- 238000002560 therapeutic procedure Methods 0.000 title description 18
- 210000000744 eyelid Anatomy 0.000 claims description 529
- 210000004175 meibomian gland Anatomy 0.000 claims description 264
- 238000011282 treatment Methods 0.000 claims description 157
- 238000010438 heat treatment Methods 0.000 claims description 132
- 208000003556 Dry Eye Syndromes Diseases 0.000 claims description 31
- 230000017531 blood circulation Effects 0.000 claims description 31
- 206010013774 Dry eye Diseases 0.000 claims description 29
- 239000012212 insulator Substances 0.000 claims description 29
- 210000001519 tissue Anatomy 0.000 claims description 29
- 238000009413 insulation Methods 0.000 claims description 15
- 239000004071 soot Substances 0.000 claims description 11
- 210000003786 sclera Anatomy 0.000 claims description 5
- 230000001143 conditioned effect Effects 0.000 claims 2
- 210000001508 eye Anatomy 0.000 description 102
- 210000004907 gland Anatomy 0.000 description 54
- 239000010410 layer Substances 0.000 description 52
- 238000012546 transfer Methods 0.000 description 45
- 150000002632 lipids Chemical class 0.000 description 44
- 230000007246 mechanism Effects 0.000 description 40
- 239000007788 liquid Substances 0.000 description 31
- 230000028327 secretion Effects 0.000 description 26
- 210000002374 sebum Anatomy 0.000 description 25
- 239000012530 fluid Substances 0.000 description 24
- 210000005252 bulbus oculi Anatomy 0.000 description 22
- 239000000463 material Substances 0.000 description 21
- 239000000725 suspension Substances 0.000 description 21
- 238000010586 diagram Methods 0.000 description 20
- 230000001105 regulatory effect Effects 0.000 description 19
- 239000003814 drug Substances 0.000 description 18
- 229940079593 drug Drugs 0.000 description 16
- 230000002829 reductive effect Effects 0.000 description 16
- 230000006835 compression Effects 0.000 description 15
- 238000007906 compression Methods 0.000 description 15
- 150000001875 compounds Chemical class 0.000 description 14
- 239000010408 film Substances 0.000 description 14
- 208000002193 Pain Diseases 0.000 description 13
- 239000003795 chemical substances by application Substances 0.000 description 12
- 238000004090 dissolution Methods 0.000 description 12
- 238000001704 evaporation Methods 0.000 description 12
- 230000008020 evaporation Effects 0.000 description 12
- 230000036407 pain Effects 0.000 description 12
- 239000003098 androgen Substances 0.000 description 11
- 230000006870 function Effects 0.000 description 11
- 238000002844 melting Methods 0.000 description 11
- 230000008018 melting Effects 0.000 description 11
- 238000012544 monitoring process Methods 0.000 description 11
- 210000000795 conjunctiva Anatomy 0.000 description 10
- 230000006378 damage Effects 0.000 description 10
- 230000004044 response Effects 0.000 description 10
- 239000007787 solid Substances 0.000 description 10
- 208000010217 blepharitis Diseases 0.000 description 9
- 208000015181 infectious disease Diseases 0.000 description 9
- 210000004379 membrane Anatomy 0.000 description 9
- 239000012528 membrane Substances 0.000 description 9
- 208000024891 symptom Diseases 0.000 description 9
- 230000009471 action Effects 0.000 description 8
- 238000012790 confirmation Methods 0.000 description 8
- 201000010099 disease Diseases 0.000 description 8
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 8
- 238000004519 manufacturing process Methods 0.000 description 8
- 238000012360 testing method Methods 0.000 description 8
- 206010015943 Eye inflammation Diseases 0.000 description 7
- 206010061218 Inflammation Diseases 0.000 description 7
- 239000006196 drop Substances 0.000 description 7
- 230000004054 inflammatory process Effects 0.000 description 7
- 230000000144 pharmacologic effect Effects 0.000 description 7
- 230000008569 process Effects 0.000 description 7
- 230000001225 therapeutic effect Effects 0.000 description 7
- 239000004098 Tetracycline Substances 0.000 description 6
- 210000004204 blood vessel Anatomy 0.000 description 6
- 230000008859 change Effects 0.000 description 6
- 235000019364 tetracycline Nutrition 0.000 description 6
- 150000003522 tetracyclines Chemical class 0.000 description 6
- 230000000699 topical effect Effects 0.000 description 6
- 229940030486 androgens Drugs 0.000 description 5
- 230000008901 benefit Effects 0.000 description 5
- 230000004397 blinking Effects 0.000 description 5
- 230000036760 body temperature Effects 0.000 description 5
- 238000001514 detection method Methods 0.000 description 5
- 230000004064 dysfunction Effects 0.000 description 5
- 230000000694 effects Effects 0.000 description 5
- 210000003811 finger Anatomy 0.000 description 5
- 206010002091 Anaesthesia Diseases 0.000 description 4
- PMATZTZNYRCHOR-CGLBZJNRSA-N Cyclosporin A Chemical compound CC[C@@H]1NC(=O)[C@H]([C@H](O)[C@H](C)C\C=C\C)N(C)C(=O)[C@H](C(C)C)N(C)C(=O)[C@H](CC(C)C)N(C)C(=O)[C@H](CC(C)C)N(C)C(=O)[C@@H](C)NC(=O)[C@H](C)NC(=O)[C@H](CC(C)C)N(C)C(=O)[C@H](C(C)C)NC(=O)[C@H](CC(C)C)N(C)C(=O)CN(C)C1=O PMATZTZNYRCHOR-CGLBZJNRSA-N 0.000 description 4
- 102000004190 Enzymes Human genes 0.000 description 4
- 108090000790 Enzymes Proteins 0.000 description 4
- MUMGGOZAMZWBJJ-DYKIIFRCSA-N Testostosterone Chemical class O=C1CC[C@]2(C)[C@H]3CC[C@](C)([C@H](CC4)O)[C@@H]4[C@@H]3CCC2=C1 MUMGGOZAMZWBJJ-DYKIIFRCSA-N 0.000 description 4
- 230000037005 anaesthesia Effects 0.000 description 4
- 239000000560 biocompatible material Substances 0.000 description 4
- 210000004027 cell Anatomy 0.000 description 4
- 230000001276 controlling effect Effects 0.000 description 4
- 238000013461 design Methods 0.000 description 4
- 238000002474 experimental method Methods 0.000 description 4
- 239000003889 eye drop Substances 0.000 description 4
- 229940012356 eye drops Drugs 0.000 description 4
- 238000003780 insertion Methods 0.000 description 4
- 230000037431 insertion Effects 0.000 description 4
- 239000000203 mixture Substances 0.000 description 4
- 229920003023 plastic Polymers 0.000 description 4
- 229920003229 poly(methyl methacrylate) Polymers 0.000 description 4
- 239000004926 polymethyl methacrylate Substances 0.000 description 4
- 230000000580 secretagogue effect Effects 0.000 description 4
- 229960002180 tetracycline Drugs 0.000 description 4
- 229930101283 tetracycline Natural products 0.000 description 4
- 206010006803 Burns third degree Diseases 0.000 description 3
- 241000282412 Homo Species 0.000 description 3
- 206010036790 Productive cough Diseases 0.000 description 3
- 210000004087 cornea Anatomy 0.000 description 3
- 230000007423 decrease Effects 0.000 description 3
- 230000007794 irritation Effects 0.000 description 3
- 238000005461 lubrication Methods 0.000 description 3
- 201000003265 lymphadenitis Diseases 0.000 description 3
- 239000002184 metal Substances 0.000 description 3
- 210000003097 mucus Anatomy 0.000 description 3
- 230000037325 pain tolerance Effects 0.000 description 3
- 239000004033 plastic Substances 0.000 description 3
- 210000003802 sputum Anatomy 0.000 description 3
- 208000024794 sputum Diseases 0.000 description 3
- 239000000758 substrate Substances 0.000 description 3
- 238000012549 training Methods 0.000 description 3
- 229930105110 Cyclosporin A Natural products 0.000 description 2
- 108010036949 Cyclosporine Proteins 0.000 description 2
- 206010013786 Dry skin Diseases 0.000 description 2
- 239000004593 Epoxy Substances 0.000 description 2
- 208000003251 Pruritus Diseases 0.000 description 2
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 2
- 102000004887 Transforming Growth Factor beta Human genes 0.000 description 2
- 108090001012 Transforming Growth Factor beta Proteins 0.000 description 2
- 239000003242 anti bacterial agent Chemical class 0.000 description 2
- 229940088710 antibiotic agent Drugs 0.000 description 2
- 230000033228 biological regulation Effects 0.000 description 2
- 230000005540 biological transmission Effects 0.000 description 2
- 229960001265 ciclosporin Drugs 0.000 description 2
- CYQFCXCEBYINGO-IAGOWNOFSA-N delta1-THC Chemical compound C1=C(C)CC[C@H]2C(C)(C)OC3=CC(CCCCC)=CC(O)=C3[C@@H]21 CYQFCXCEBYINGO-IAGOWNOFSA-N 0.000 description 2
- 210000000981 epithelium Anatomy 0.000 description 2
- 239000000262 estrogen Substances 0.000 description 2
- 229940011871 estrogen Drugs 0.000 description 2
- 235000021323 fish oil Nutrition 0.000 description 2
- 230000000762 glandular Effects 0.000 description 2
- 230000006872 improvement Effects 0.000 description 2
- 230000000977 initiatory effect Effects 0.000 description 2
- 230000007803 itching Effects 0.000 description 2
- 210000005128 keratinized epithelium Anatomy 0.000 description 2
- 238000012423 maintenance Methods 0.000 description 2
- 230000014759 maintenance of location Effects 0.000 description 2
- 239000000155 melt Substances 0.000 description 2
- 238000012986 modification Methods 0.000 description 2
- 230000004048 modification Effects 0.000 description 2
- 235000019198 oils Nutrition 0.000 description 2
- 229940012843 omega-3 fatty acid Drugs 0.000 description 2
- 235000020660 omega-3 fatty acid Nutrition 0.000 description 2
- 229940124624 oral corticosteroid Drugs 0.000 description 2
- 239000011253 protective coating Substances 0.000 description 2
- 230000001681 protective effect Effects 0.000 description 2
- 230000005855 radiation Effects 0.000 description 2
- 229940053174 restasis Drugs 0.000 description 2
- 239000011780 sodium chloride Substances 0.000 description 2
- 239000000126 substance Substances 0.000 description 2
- 230000002459 sustained effect Effects 0.000 description 2
- 229960003604 testosterone Drugs 0.000 description 2
- 229940040944 tetracyclines Drugs 0.000 description 2
- ZRKFYGHZFMAOKI-QMGMOQQFSA-N tgfbeta Chemical compound C([C@H](NC(=O)[C@H](C(C)C)NC(=O)CNC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H]([C@@H](C)O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H]([C@@H](C)O)NC(=O)[C@H](CC(C)C)NC(=O)CNC(=O)[C@H](C)NC(=O)[C@H](CO)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@@H](NC(=O)[C@H](C)NC(=O)[C@H](C)NC(=O)[C@@H](NC(=O)[C@H](CC(C)C)NC(=O)[C@@H](N)CCSC)C(C)C)[C@@H](C)CC)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CC=1C=CC=CC=1)C(=O)N[C@@H](C)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](C)C(=O)N[C@@H](CC=1C=CC=CC=1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](C)C(=O)N[C@@H](CC(C)C)C(=O)N1[C@@H](CCC1)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CO)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC(C)C)C(O)=O)C1=CC=C(O)C=C1 ZRKFYGHZFMAOKI-QMGMOQQFSA-N 0.000 description 2
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 2
- LQIAZOCLNBBZQK-UHFFFAOYSA-N 1-(1,2-Diphosphanylethyl)pyrrolidin-2-one Chemical compound PCC(P)N1CCCC1=O LQIAZOCLNBBZQK-UHFFFAOYSA-N 0.000 description 1
- 241000894006 Bacteria Species 0.000 description 1
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 1
- 206010010741 Conjunctivitis Diseases 0.000 description 1
- 201000004624 Dermatitis Diseases 0.000 description 1
- JOYRKODLDBILNP-UHFFFAOYSA-N Ethyl urethane Chemical compound CCOC(N)=O JOYRKODLDBILNP-UHFFFAOYSA-N 0.000 description 1
- 229920004142 LEXAN™ Polymers 0.000 description 1
- 229930189127 Lacrimin Natural products 0.000 description 1
- 239000004418 Lexan Substances 0.000 description 1
- 229920000106 Liquid crystal polymer Polymers 0.000 description 1
- 239000004977 Liquid-crystal polymers (LCPs) Substances 0.000 description 1
- 241000124008 Mammalia Species 0.000 description 1
- 102000015728 Mucins Human genes 0.000 description 1
- 108010063954 Mucins Proteins 0.000 description 1
- 208000012868 Overgrowth Diseases 0.000 description 1
- 239000004734 Polyphenylene sulfide Substances 0.000 description 1
- 241000295644 Staphylococcaceae Species 0.000 description 1
- 208000032159 Vaginal inflammation Diseases 0.000 description 1
- 201000008100 Vaginitis Diseases 0.000 description 1
- 230000002159 abnormal effect Effects 0.000 description 1
- 238000001467 acupuncture Methods 0.000 description 1
- 230000001154 acute effect Effects 0.000 description 1
- 210000001742 aqueous humor Anatomy 0.000 description 1
- 230000001580 bacterial effect Effects 0.000 description 1
- 230000000903 blocking effect Effects 0.000 description 1
- 239000008280 blood Substances 0.000 description 1
- 210000004369 blood Anatomy 0.000 description 1
- 238000009530 blood pressure measurement Methods 0.000 description 1
- 238000009529 body temperature measurement Methods 0.000 description 1
- WYEMLYFITZORAB-UHFFFAOYSA-N boscalid Chemical compound C1=CC(Cl)=CC=C1C1=CC=CC=C1NC(=O)C1=CC=CN=C1Cl WYEMLYFITZORAB-UHFFFAOYSA-N 0.000 description 1
- 229910052799 carbon Inorganic materials 0.000 description 1
- 238000006243 chemical reaction Methods 0.000 description 1
- 239000000701 coagulant Substances 0.000 description 1
- 239000011248 coating agent Substances 0.000 description 1
- 238000000576 coating method Methods 0.000 description 1
- 238000004891 communication Methods 0.000 description 1
- 238000011109 contamination Methods 0.000 description 1
- 238000007796 conventional method Methods 0.000 description 1
- 238000001816 cooling Methods 0.000 description 1
- 239000003246 corticosteroid Substances 0.000 description 1
- 229960001334 corticosteroids Drugs 0.000 description 1
- 239000002537 cosmetic Substances 0.000 description 1
- 230000008878 coupling Effects 0.000 description 1
- 238000010168 coupling process Methods 0.000 description 1
- 238000005859 coupling reaction Methods 0.000 description 1
- 230000001186 cumulative effect Effects 0.000 description 1
- 230000003247 decreasing effect Effects 0.000 description 1
- 230000002950 deficient Effects 0.000 description 1
- 230000000994 depressogenic effect Effects 0.000 description 1
- 206010012601 diabetes mellitus Diseases 0.000 description 1
- 238000002405 diagnostic procedure Methods 0.000 description 1
- 238000009826 distribution Methods 0.000 description 1
- 210000002919 epithelial cell Anatomy 0.000 description 1
- 210000005081 epithelial layer Anatomy 0.000 description 1
- 230000005284 excitation Effects 0.000 description 1
- 238000000605 extraction Methods 0.000 description 1
- 208000030533 eye disease Diseases 0.000 description 1
- 230000008713 feedback mechanism Effects 0.000 description 1
- 239000006260 foam Substances 0.000 description 1
- 239000003292 glue Substances 0.000 description 1
- 230000003054 hormonal effect Effects 0.000 description 1
- 230000002706 hydrostatic effect Effects 0.000 description 1
- 238000003384 imaging method Methods 0.000 description 1
- 230000001771 impaired effect Effects 0.000 description 1
- 230000001050 lubricating effect Effects 0.000 description 1
- 238000005259 measurement Methods 0.000 description 1
- 230000009245 menopause Effects 0.000 description 1
- 238000013508 migration Methods 0.000 description 1
- 230000005012 migration Effects 0.000 description 1
- 229940051875 mucins Drugs 0.000 description 1
- 210000003205 muscle Anatomy 0.000 description 1
- 230000003287 optical effect Effects 0.000 description 1
- PRGUDWLMFLCODA-UHFFFAOYSA-N oxybuprocaine hydrochloride Chemical compound [Cl-].CCCCOC1=CC(C(=O)OCC[NH+](CC)CC)=CC=C1N PRGUDWLMFLCODA-UHFFFAOYSA-N 0.000 description 1
- 230000036961 partial effect Effects 0.000 description 1
- 239000013618 particulate matter Substances 0.000 description 1
- 238000005192 partition Methods 0.000 description 1
- 239000002304 perfume Substances 0.000 description 1
- 230000002093 peripheral effect Effects 0.000 description 1
- 229920000642 polymer Polymers 0.000 description 1
- 229920000069 polyphenylene sulfide Polymers 0.000 description 1
- 238000003825 pressing Methods 0.000 description 1
- 239000011241 protective layer Substances 0.000 description 1
- 230000003362 replicative effect Effects 0.000 description 1
- 230000000284 resting effect Effects 0.000 description 1
- 230000000717 retained effect Effects 0.000 description 1
- 230000002441 reversible effect Effects 0.000 description 1
- 210000001732 sebaceous gland Anatomy 0.000 description 1
- 230000003248 secreting effect Effects 0.000 description 1
- 238000004904 shortening Methods 0.000 description 1
- 230000002269 spontaneous effect Effects 0.000 description 1
- 230000001954 sterilising effect Effects 0.000 description 1
- 238000004659 sterilization and disinfection Methods 0.000 description 1
- 230000004936 stimulating effect Effects 0.000 description 1
- 239000013589 supplement Substances 0.000 description 1
- 230000009897 systematic effect Effects 0.000 description 1
- 230000008719 thickening Effects 0.000 description 1
- 239000010409 thin film Substances 0.000 description 1
- 210000003813 thumb Anatomy 0.000 description 1
- 230000000451 tissue damage Effects 0.000 description 1
- 231100000827 tissue damage Toxicity 0.000 description 1
- 229940125379 topical corticosteroid Drugs 0.000 description 1
- 238000011277 treatment modality Methods 0.000 description 1
- 230000007306 turnover Effects 0.000 description 1
- 210000001215 vagina Anatomy 0.000 description 1
- 238000012795 verification Methods 0.000 description 1
- 230000000007 visual effect Effects 0.000 description 1
- 238000010792 warming Methods 0.000 description 1
- 230000037303 wrinkles Effects 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F7/00—Heating or cooling appliances for medical or therapeutic treatment of the human body
- A61F7/02—Compresses or poultices for effecting heating or cooling
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P27/00—Drugs for disorders of the senses
- A61P27/02—Ophthalmic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F7/00—Heating or cooling appliances for medical or therapeutic treatment of the human body
- A61F2007/0001—Body part
- A61F2007/0002—Head or parts thereof
- A61F2007/0004—Eyes or part of the face surrounding the eyes
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F7/00—Heating or cooling appliances for medical or therapeutic treatment of the human body
- A61F2007/0086—Heating or cooling appliances for medical or therapeutic treatment of the human body with a thermostat
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F7/00—Heating or cooling appliances for medical or therapeutic treatment of the human body
- A61F7/007—Heating or cooling appliances for medical or therapeutic treatment of the human body characterised by electric heating
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F9/00—Methods or devices for treatment of the eyes; Devices for putting-in contact lenses; Devices to correct squinting; Apparatus to guide the blind; Protective devices for the eyes, carried on the body or in the hand
- A61F9/007—Methods or devices for eye surgery
- A61F9/00718—Restoration of lid function
Description
本出願は、2007年1月17日出願の米国暫定特許出願第60/880,850号の表題「Method and Apparatus for Treating Meibomian Gland Obstructive Disease」に対する優先権を主張し、参照することによってその全体が本明細書に組み込まれる。
熱の印加を調節し得るとは、加熱手段または要素を、眼瞼に対して安全であり、マイボーム腺における閉塞または妨害物を溶解、弛緩、または軟化させるために十分な温度で安全である、該温度内および該手段内に制御することを意味する。
Claims (17)
- ドライアイの治療のための装置であって、
熱を印加することができるデバイスを備えており、
前記デバイスが、マイボーム腺における妨害物を溶解、弛緩、または軟化させて除去する温度レベルまで眼瞼の内側へと熱を印加するように構成されている、装置。 - 前記デバイスが、一定時間にわたって前記眼瞼の内側への熱を維持するようにさらに構成されている、請求項1に記載の装置。
- 前記デバイスが、前記眼瞼の内側へと熱を印加するときに熱を調節するようにさらに構成されている、請求項1に記載の装置。
- 前記デバイスが、前記眼瞼の内側へと熱を印加するときに、摂氏39〜47度の間の温度レベルに熱を調節するようにさらに構成されている、請求項1に記載の装置。
- 前記デバイスが、マイボーム腺に近接する組織に熱を印加するようにさらに構成されている、請求項1に記載の装置。
- 前記マイボーム腺が、マイボーム腺チャネルと、該マイボーム腺チャネルの頂部のマイボーム腺開口部とを有し、
前記デバイスが、複数のマイボーム腺の1つ以上のマイボーム腺のマイボーム腺チャネル内の妨害物が溶解状態になるように、複数のマイボーム腺を含む眼瞼に調整された熱を印加し、
マイボーム腺のマイボーム腺開口部を通してマイボーム腺チャネル内から妨害物を圧搾するようにさらに構成されている、請求項1に記載の装置。 - 前記デバイスが、一定時間にわたって前記眼瞼の内側への熱を維持しながらマイボーム腺のマイボーム腺開口部を通してマイボーム腺チャネル内から妨害物を圧搾するようにさらに構成されている、請求項6に記載の装置。
- 前記デバイスが、マイボーム腺のマイボーム腺開口部を通してマイボーム腺チャネル内から妨害物を圧搾するように哺乳類の眼瞼の表面へと力を案内するようにさらに構成されている、請求項6に記載の装置。
- 前記デバイスが、妨害物を圧搾するように一定時間にわたって一定または間欠的な力を維持するようにさらに構成されている、請求項8に記載の装置。
- 前記デバイスが、前記眼瞼の血流を低下させ、前記眼瞼からの対流熱損失を軽減するよう、前記力を維持するようにさらに構成されている、請求項8に記載の装置。
- 前記デバイスが、
断熱体と、
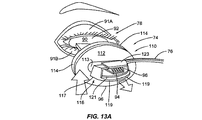
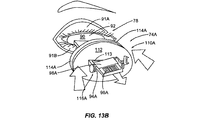
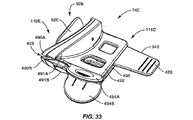
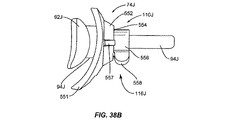
前記断熱体に埋め込まれた加熱要素とを備える瞼加温器をさらに含んでおり、
前記瞼加温器が、前記眼瞼の内側へと前記加熱要素によって熱を印加するように構成されている、請求項1に記載の装置。 - 前記断熱体が、眼の強膜の外部表面上に配置されるようにさらに構成されている、請求項11に記載の装置。
- 前記断熱体が、前記加熱要素に電力が印加されて熱が生じるよう、前記加熱要素に連結した電気的インターフェースをさらに備えている、請求項11に記載の装置。
- 前記電気的インターフェースを介して前記加熱要素へと接続され、前記加熱要素によって生成される熱を調節するコントローラをさらに備えている、請求項13に記載の装置。
- 前記デバイスが、前記眼瞼の外部表面に力を印加するように拡張可能であるブラダーをさらに備えている、請求項11に記載の装置。
- 前記マイボーム腺が、マイボーム腺チャネルと、該マイボーム腺チャネルの頂部のマイボーム腺開口部とを有し、前記デバイスが、マイボーム腺から妨害物を圧搾するように、マイボーム腺チャネルの底部からマイボーム腺チャネルの頂部へ向かう方向にマイボーム腺チャネルに調整され方向付けられた力を印加するようにさらに構成された、請求項1に記載の装置。
- デバイスによって印加される調整され方向付けられた力が、ミルキングタイプの力である、請求項16に記載の装置。
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US88085007P | 2007-01-17 | 2007-01-17 | |
| US60/880,850 | 2007-01-17 | ||
| PCT/US2008/051309 WO2008089327A1 (en) | 2007-01-17 | 2008-01-17 | Methods, systems, and apparatuses for heat treatment for treating meibomian gland dysfunction |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2013226709A Division JP2014087649A (ja) | 2007-01-17 | 2013-10-31 | マイボーム腺機能不全を治療するための熱治療のための方法、システム、および装置 |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| JP2010516343A JP2010516343A (ja) | 2010-05-20 |
| JP2010516343A5 JP2010516343A5 (ja) | 2011-03-10 |
| JP5406728B2 true JP5406728B2 (ja) | 2014-02-05 |
Family
ID=39636379
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2009546506A Active JP5406728B2 (ja) | 2007-01-17 | 2008-01-17 | マイボーム腺機能不全を治療するための熱治療のための方法、システム、および装置 |
| JP2013226709A Pending JP2014087649A (ja) | 2007-01-17 | 2013-10-31 | マイボーム腺機能不全を治療するための熱治療のための方法、システム、および装置 |
Family Applications After (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2013226709A Pending JP2014087649A (ja) | 2007-01-17 | 2013-10-31 | マイボーム腺機能不全を治療するための熱治療のための方法、システム、および装置 |
Country Status (8)
| Country | Link |
|---|---|
| EP (2) | EP3434313A1 (ja) |
| JP (2) | JP5406728B2 (ja) |
| CN (3) | CN103181837B (ja) |
| BR (1) | BRPI0806635A2 (ja) |
| ES (1) | ES2692434T3 (ja) |
| IL (1) | IL199920A0 (ja) |
| SG (2) | SG177990A1 (ja) |
| WO (1) | WO2008089327A1 (ja) |
Families Citing this family (32)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20080114423A1 (en) | 2006-05-15 | 2008-05-15 | Grenon Stephen M | Apparatus for inner eyelid treatment of meibomian gland dysfunction |
| US20070060988A1 (en) | 2005-07-18 | 2007-03-15 | Grenon Stephen M | Melting meibomian gland obstructions |
| US7981145B2 (en) | 2005-07-18 | 2011-07-19 | Tearscience Inc. | Treatment of meibomian glands |
| US20090043365A1 (en) | 2005-07-18 | 2009-02-12 | Kolis Scientific, Inc. | Methods, apparatuses, and systems for reducing intraocular pressure as a means of preventing or treating open-angle glaucoma |
| US8915253B2 (en) | 2005-07-18 | 2014-12-23 | Tearscience, Inc. | Method and apparatus for treating gland dysfunction employing heated medium |
| WO2013003594A2 (en) | 2011-06-28 | 2013-01-03 | Tearscience, Inc. | Methods and systems for treating meibomian gland dysfunction using radio-frequency energy |
| US7981095B2 (en) | 2005-07-18 | 2011-07-19 | Tearscience, Inc. | Methods for treating meibomian gland dysfunction employing fluid jet |
| US8950405B2 (en) | 2006-05-15 | 2015-02-10 | Tearscience, Inc. | Treatment of obstructive disorders of the eye or eyelid |
| US8128673B2 (en) | 2006-05-15 | 2012-03-06 | Tearscience, Inc. | System for inner eyelid heat and pressure treatment for treating meibomian gland dysfunction |
| US9314369B2 (en) | 2006-05-15 | 2016-04-19 | Tearscience, Inc. | System for inner eyelid treatment of meibomian gland dysfunction |
| US8128674B2 (en) | 2006-05-15 | 2012-03-06 | Tearscience, Inc. | System for outer eyelid heat and pressure treatment for treating meibomian gland dysfunction |
| US8137390B2 (en) | 2006-05-15 | 2012-03-20 | Tearscience, Inc. | System for providing heat treatment and heat loss reduction for treating meibomian gland dysfunction |
| US9642520B2 (en) | 2009-04-01 | 2017-05-09 | Tearscience, Inc. | Background reduction apparatuses and methods of ocular surface interferometry (OSI) employing polarization for imaging, processing, and/or displaying an ocular tear film |
| US9888839B2 (en) | 2009-04-01 | 2018-02-13 | Tearscience, Inc. | Methods and apparatuses for determining contact lens intolerance in contact lens wearer patients based on dry eye tear film characteristic analysis and dry eye symptoms |
| US8746883B2 (en) | 2009-04-01 | 2014-06-10 | Tearscience, Inc. | Ocular surface interferometery (OSI) devices and systems for imaging, processing, and/or displaying an ocular tear film |
| AU2013213968A1 (en) * | 2012-02-03 | 2014-09-18 | Eye Comfort Limited | Eye massage device |
| WO2014031857A2 (en) | 2012-08-22 | 2014-02-27 | Tearscience, Inc. | Apparatuses and methods for diagnosing and/or treating lipid transport deficiency in ocular tear films, and related components and devices |
| US9339177B2 (en) | 2012-12-21 | 2016-05-17 | Tearscience, Inc. | Full-eye illumination ocular surface imaging of an ocular tear film for determining tear film thickness and/or providing ocular topography |
| US9763827B2 (en) | 2013-04-30 | 2017-09-19 | Tear Film Innovations, Inc. | Systems and methods for the treatment of eye conditions |
| ES2901406T3 (es) * | 2013-05-03 | 2022-03-22 | Tearscience Inc | Sistemas y métodos de iluminación de párpados para imagenología de las glándulas de Meibomio para análisis de las glándulas de Meibomio |
| US9795290B2 (en) | 2013-11-15 | 2017-10-24 | Tearscience, Inc. | Ocular tear film peak detection and stabilization detection systems and methods for determining tear film layer characteristics |
| JP6927881B2 (ja) * | 2014-10-30 | 2021-09-01 | アルコン インコーポレイティド | 眼の疾患を手当てするシステムおよび方法 |
| CN105520805B (zh) * | 2016-02-04 | 2017-11-07 | 张晨明 | 赶压式睑板腺功能障碍治疗装置 |
| US10974063B2 (en) | 2016-06-30 | 2021-04-13 | Alcon Inc. | Light therapy for eyelash growth |
| TWI772314B (zh) * | 2016-07-12 | 2022-08-01 | 德商得瑪法公司 | 用於瘙癢症的高熱治療之裝置 |
| ES2800914T3 (es) * | 2016-10-11 | 2021-01-05 | Dermapharm Ag | Dispositivo para un tratamiento hipertérmico de picores |
| AU2018263955B2 (en) | 2017-05-05 | 2024-01-04 | Andrew Jaccoma | Devices and methods for treating eyelids |
| WO2019033339A1 (en) * | 2017-08-17 | 2019-02-21 | Xinova, LLC | DISPOSABLE CONTACT LENS FOR OPTICAL PROCESSING SYSTEMS |
| WO2020130176A1 (ko) * | 2018-12-19 | 2020-06-25 | 부산대학교 산학협력단 | 안구 건조증 치료용 장치 |
| CN109771127A (zh) * | 2019-02-27 | 2019-05-21 | 浙江花园润嘉医疗器械有限公司 | 干眼症治疗仪及其控制方法 |
| CN114081716B (zh) * | 2022-01-22 | 2022-04-12 | 南方医科大学深圳医院 | 用于向睑板腺递送药物的装置 |
| CN117562733A (zh) * | 2022-08-08 | 2024-02-20 | 上海臻复医疗科技有限公司 | 眼杯以及眼部治疗装置 |
Family Cites Families (16)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3140390A (en) * | 1963-06-12 | 1964-07-07 | Smith Percy | Therapeutic appliance |
| US6107289A (en) | 1992-04-21 | 2000-08-22 | The Schepens Eye Research Institute, Inc. | Ocular therapy in keratoconjunctivitis sicca using topically applied androgens or TGF-β |
| US5958912A (en) | 1992-04-21 | 1999-09-28 | The Schepens Eye Research Institute, Inc. | Ocular therapy in keratoconjunctivitis sicca using topically applied androgens of TGF-β |
| JPH06269473A (ja) * | 1993-03-19 | 1994-09-27 | Santen Pharmaceut Co Ltd | 携帯式眼部加温器 |
| WO1997020578A1 (en) | 1995-12-04 | 1997-06-12 | University Of Miami | Non-preserved topical corticosteroid for treatment of dry eye, filamentary keratitis, and delayed tear clearance |
| JPH11221247A (ja) * | 1997-08-25 | 1999-08-17 | Seputo:Kk | 温熱付眼鏡 |
| ATE319456T1 (de) | 1998-05-08 | 2006-03-15 | Univ Miami | Verwendung von tetracyclinen zur behandlung von störungen der meibomschen drüsen |
| TW539564B (en) * | 1999-05-25 | 2003-07-01 | Iomed Inc | Methods and apparatus for ocular iontophoresis |
| JP3613634B2 (ja) * | 2001-11-06 | 2005-01-26 | 得一郎 長谷川 | 視力矯正装置 |
| GB2384430B (en) * | 2002-01-24 | 2003-12-03 | John Robert Fuller | Therapeutic eye and eyelid cover |
| GB0225795D0 (en) * | 2002-11-05 | 2002-12-11 | Guillon Michael | Wipes |
| US7513893B2 (en) * | 2004-03-12 | 2009-04-07 | Abraham Ebbie Soroudi | Device and method for treatment of eyelid diseases |
| CN100553588C (zh) * | 2004-11-23 | 2009-10-28 | 小爱德华·K·王 | 控制眼和眼周围组织温度以及治疗眼疾病的医疗装置 |
| JP2006198249A (ja) * | 2005-01-21 | 2006-08-03 | Rohto Pharmaceut Co Ltd | ドライアイ治療用具 |
| US8915253B2 (en) | 2005-07-18 | 2014-12-23 | Tearscience, Inc. | Method and apparatus for treating gland dysfunction employing heated medium |
| CN101547666B (zh) * | 2006-08-21 | 2012-06-20 | 眼泪科学公司 | 用于治疗腺体功能障碍的器械 |
-
2008
- 2008-01-17 EP EP18192086.9A patent/EP3434313A1/en not_active Withdrawn
- 2008-01-17 SG SG2012003638A patent/SG177990A1/en unknown
- 2008-01-17 BR BRPI0806635-3A patent/BRPI0806635A2/pt not_active Application Discontinuation
- 2008-01-17 EP EP08727830.5A patent/EP2121109B1/en active Active
- 2008-01-17 JP JP2009546506A patent/JP5406728B2/ja active Active
- 2008-01-17 CN CN201310017761.3A patent/CN103181837B/zh active Active
- 2008-01-17 CN CN201310017764.7A patent/CN103083131B/zh active Active
- 2008-01-17 WO PCT/US2008/051309 patent/WO2008089327A1/en active Application Filing
- 2008-01-17 CN CN2008800087417A patent/CN101663064B/zh active Active
- 2008-01-17 ES ES08727830.5T patent/ES2692434T3/es active Active
- 2008-01-17 SG SG2012003646A patent/SG177991A1/en unknown
-
2009
- 2009-07-16 IL IL199920A patent/IL199920A0/en unknown
-
2013
- 2013-10-31 JP JP2013226709A patent/JP2014087649A/ja active Pending
Also Published As
| Publication number | Publication date |
|---|---|
| CN103083131B (zh) | 2015-07-22 |
| ES2692434T3 (es) | 2018-12-03 |
| CN103181837B (zh) | 2015-04-22 |
| BRPI0806635A2 (pt) | 2011-09-06 |
| SG177991A1 (en) | 2012-02-28 |
| EP2121109A1 (en) | 2009-11-25 |
| CN101663064A (zh) | 2010-03-03 |
| WO2008089327A1 (en) | 2008-07-24 |
| EP2121109A4 (en) | 2013-01-23 |
| JP2014087649A (ja) | 2014-05-15 |
| SG177990A1 (en) | 2012-02-28 |
| CN103181837A (zh) | 2013-07-03 |
| IL199920A0 (en) | 2010-04-15 |
| CN103083131A (zh) | 2013-05-08 |
| JP2010516343A (ja) | 2010-05-20 |
| EP2121109B1 (en) | 2018-09-05 |
| EP3434313A1 (en) | 2019-01-30 |
| CN101663064B (zh) | 2013-03-06 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5406728B2 (ja) | マイボーム腺機能不全を治療するための熱治療のための方法、システム、および装置 | |
| US20210205131A1 (en) | Methods and apparatuses for treatment of meibomian gland dysfunction | |
| US8617229B2 (en) | System for outer eyelid heat and pressure treatment for treating meibomian gland dysfunction | |
| US7976573B2 (en) | Inner eyelid heat and pressure treatment for treating meibomian gland dysfunction | |
| US9314369B2 (en) | System for inner eyelid treatment of meibomian gland dysfunction | |
| US8523928B2 (en) | System for inner eyelid heat and pressure treatment for treating meibomian gland dysfunction | |
| US7981147B2 (en) | Outer eyelid heat and pressure treatment for treating meibomian gland dysfunction | |
| US8137390B2 (en) | System for providing heat treatment and heat loss reduction for treating meibomian gland dysfunction | |
| US7981146B2 (en) | Inner eyelid treatment for treating meibomian gland dysfunction | |
| US8007524B2 (en) | Heat treatment and heat loss reduction for treating meibomian gland dysfunction | |
| US10940074B2 (en) | Melting meibomian gland obstructions | |
| US20090306588A1 (en) | Balloon catheter systems and methods for treating uterine disorders | |
| EP2053997A1 (en) | Melting meibomian gland obstructions | |
| JP2010504769A (ja) | マイボーム腺閉塞の溶解 | |
| WO2008039221A1 (en) | Providing gas and vapor to a patient |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20110117 |
|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20110117 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20120904 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20121130 |
|
| A602 | Written permission of extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A602 Effective date: 20121207 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20130301 |
|
| RD04 | Notification of resignation of power of attorney |
Free format text: JAPANESE INTERMEDIATE CODE: A7424 Effective date: 20130314 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20130903 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20131002 |
|
| A602 | Written permission of extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A602 Effective date: 20131008 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20131101 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 5406728 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |