JP2022546292A - フリースタンディングフィルム型リチウム二次電池用正極材、この製造方法及びこれを含むリチウム二次電池 - Google Patents
フリースタンディングフィルム型リチウム二次電池用正極材、この製造方法及びこれを含むリチウム二次電池 Download PDFInfo
- Publication number
- JP2022546292A JP2022546292A JP2022511087A JP2022511087A JP2022546292A JP 2022546292 A JP2022546292 A JP 2022546292A JP 2022511087 A JP2022511087 A JP 2022511087A JP 2022511087 A JP2022511087 A JP 2022511087A JP 2022546292 A JP2022546292 A JP 2022546292A
- Authority
- JP
- Japan
- Prior art keywords
- positive electrode
- sulfur
- carbon
- secondary battery
- lithium secondary
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
Images
Classifications
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/362—Composites
- H01M4/364—Composites as mixtures
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/13—Electrodes for accumulators with non-aqueous electrolyte, e.g. for lithium-accumulators; Processes of manufacture thereof
- H01M4/133—Electrodes based on carbonaceous material, e.g. graphite-intercalation compounds or CFx
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01B—NON-METALLIC ELEMENTS; COMPOUNDS THEREOF; METALLOIDS OR COMPOUNDS THEREOF NOT COVERED BY SUBCLASS C01C
- C01B17/00—Sulfur; Compounds thereof
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M10/00—Secondary cells; Manufacture thereof
- H01M10/05—Accumulators with non-aqueous electrolyte
- H01M10/052—Li-accumulators
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M10/00—Secondary cells; Manufacture thereof
- H01M10/05—Accumulators with non-aqueous electrolyte
- H01M10/052—Li-accumulators
- H01M10/0525—Rocking-chair batteries, i.e. batteries with lithium insertion or intercalation in both electrodes; Lithium-ion batteries
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/04—Processes of manufacture in general
- H01M4/0471—Processes of manufacture in general involving thermal treatment, e.g. firing, sintering, backing particulate active material, thermal decomposition, pyrolysis
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/13—Electrodes for accumulators with non-aqueous electrolyte, e.g. for lithium-accumulators; Processes of manufacture thereof
- H01M4/139—Processes of manufacture
- H01M4/1393—Processes of manufacture of electrodes based on carbonaceous material, e.g. graphite-intercalation compounds or CFx
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/38—Selection of substances as active materials, active masses, active liquids of elements or alloys
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/58—Selection of substances as active materials, active masses, active liquids of inorganic compounds other than oxides or hydroxides, e.g. sulfides, selenides, tellurides, halogenides or LiCoFy; of polyanionic structures, e.g. phosphates, silicates or borates
- H01M4/583—Carbonaceous material, e.g. graphite-intercalation compounds or CFx
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/58—Selection of substances as active materials, active masses, active liquids of inorganic compounds other than oxides or hydroxides, e.g. sulfides, selenides, tellurides, halogenides or LiCoFy; of polyanionic structures, e.g. phosphates, silicates or borates
- H01M4/583—Carbonaceous material, e.g. graphite-intercalation compounds or CFx
- H01M4/587—Carbonaceous material, e.g. graphite-intercalation compounds or CFx for inserting or intercalating light metals
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/62—Selection of inactive substances as ingredients for active masses, e.g. binders, fillers
- H01M4/624—Electric conductive fillers
- H01M4/625—Carbon or graphite
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M2004/021—Physical characteristics, e.g. porosity, surface area
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M2004/026—Electrodes composed of, or comprising, active material characterised by the polarity
- H01M2004/028—Positive electrodes
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/38—Selection of substances as active materials, active masses, active liquids of elements or alloys
- H01M4/381—Alkaline or alkaline earth metals elements
- H01M4/382—Lithium
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E60/00—Enabling technologies; Technologies with a potential or indirect contribution to GHG emissions mitigation
- Y02E60/10—Energy storage using batteries
Landscapes
- Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Electrochemistry (AREA)
- General Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Manufacturing & Machinery (AREA)
- Inorganic Chemistry (AREA)
- Composite Materials (AREA)
- Materials Engineering (AREA)
- Organic Chemistry (AREA)
- Battery Electrode And Active Subsutance (AREA)
- Secondary Cells (AREA)
Abstract
Description
(S2)上記(S1)段階で形成された混合物を熱処理する段階;及び
(S3)上記(S2)段階で形成された硫黄‐炭素複合体を容器に充填した後、加圧して炭素を含む硫黄溶融物を形成する段階;を含むリチウム二次電池用正極材の製造方法を提供する。
本発明はフリースタンディングフィルム形態のリチウム二次電池用正極材であって、上記正極材は炭素を含む硫黄溶融物を含むものであってもよい。上記正極材は硫黄溶融物に多孔性炭素材が分散された形態であってもよい。また、上記正極材は後述するように、硫黄と多孔性炭素材のみを原料にして乾式工程で製造されたもので、もっぱら硫黄と多孔性炭素材のみで構成されたものであってもよい。
図1は、リチウム二次電池用正極材で使用されることができる硫黄、炭素及び硫黄‐炭素複合体を加圧する場合の物性変化及び成形性を示す模式図である。
(S1)硫黄と多孔性炭素材を混合する段階;
(S2)上記(S1)段階で形成された混合物を熱処理する段階;及び
(S3)上記(S2)段階で形成された硫黄‐炭素複合体を容器に充填した後、加圧して炭素を含む硫黄溶融物を形成する段階。
本発明はまた、上記のような炭素を含む硫黄溶融物を含むフリースタンディングフィルム型正極材を正極で含むリチウム二次電池に関する。
硫黄(S)と炭素ナノチューブ(CNT)を固体状態で65:35の質量比で均一に混合した後、100rpmの条件で1時間ボールミーリングして混合物を製造した。
(1)フリースタンディングフィルム型正極材の製造
上記製造例1の硫黄‐炭素複合体(S‐CNT)をモールドに充填させた後、油圧プレスを利用して1MPaの圧力で加圧することで炭素を含む硫黄溶融物を形成して、図2に示すようにフリースタンディングフィルム型正極材を製造した。
上記製造されたフリースタンディングフィルム型正極材を正極で用意し、厚さ150μmのリチウム金属を負極で用意した。
上記硫黄‐炭素複合体の加圧時の圧力を1.5Mpaにしたことを除いて実施例1と同様の方法で実施した。
上記硫黄‐炭素複合体の加圧時の圧力を3Mpaにしたことを除いて実施例1と同様の方法で実施した。
上記製造例1の硫黄‐炭素複合体(S‐CNT)、導電材及びバインダーを90:5:5の質量比で混合してスラリーを製造した後、アルミニウムホイルの上にコーティングした後、乾燥及び圧延して正極を製造したことを除いて実施例1と同様の方法でリチウム‐硫黄二次電池を製造した。この時、上記導電材はVGCF(Vapor grown carbon fiber)を使用し、上記バインダーはSBR(Styrene Butadiene Rubber)を使用した。
圧延された厚さを比較例1に比べて相対的に減少させ、正極の気孔率が65%になるようにしたことを除いて比較例1と同様の方法でリチウム‐硫黄二次電池を製造した。
圧延された厚さを比較例2に比べて相対的に減少させ、正極の気孔率が58%になるようにしたことを除いて比較例1と同様の方法でリチウム‐硫黄二次電池を製造した。
加圧時の圧力を0.5Mpaにしたことを除いて実施例1と同様の方法で実施した。
加圧時の圧力を20Mpaにしたことを除いて実施例1と同様の方法でリチウム‐硫黄二次電池を製造した。
硫黄‐炭素複合体と一緒に導電材であるCNTを10質量%のモールドに充填して加圧したことを除いて、実施例1と同様の方法でリチウム‐硫黄二次電池を製造した。この時、導電材の含量は硫黄‐炭素複合体と導電材の全体質量を基準にしたものである。
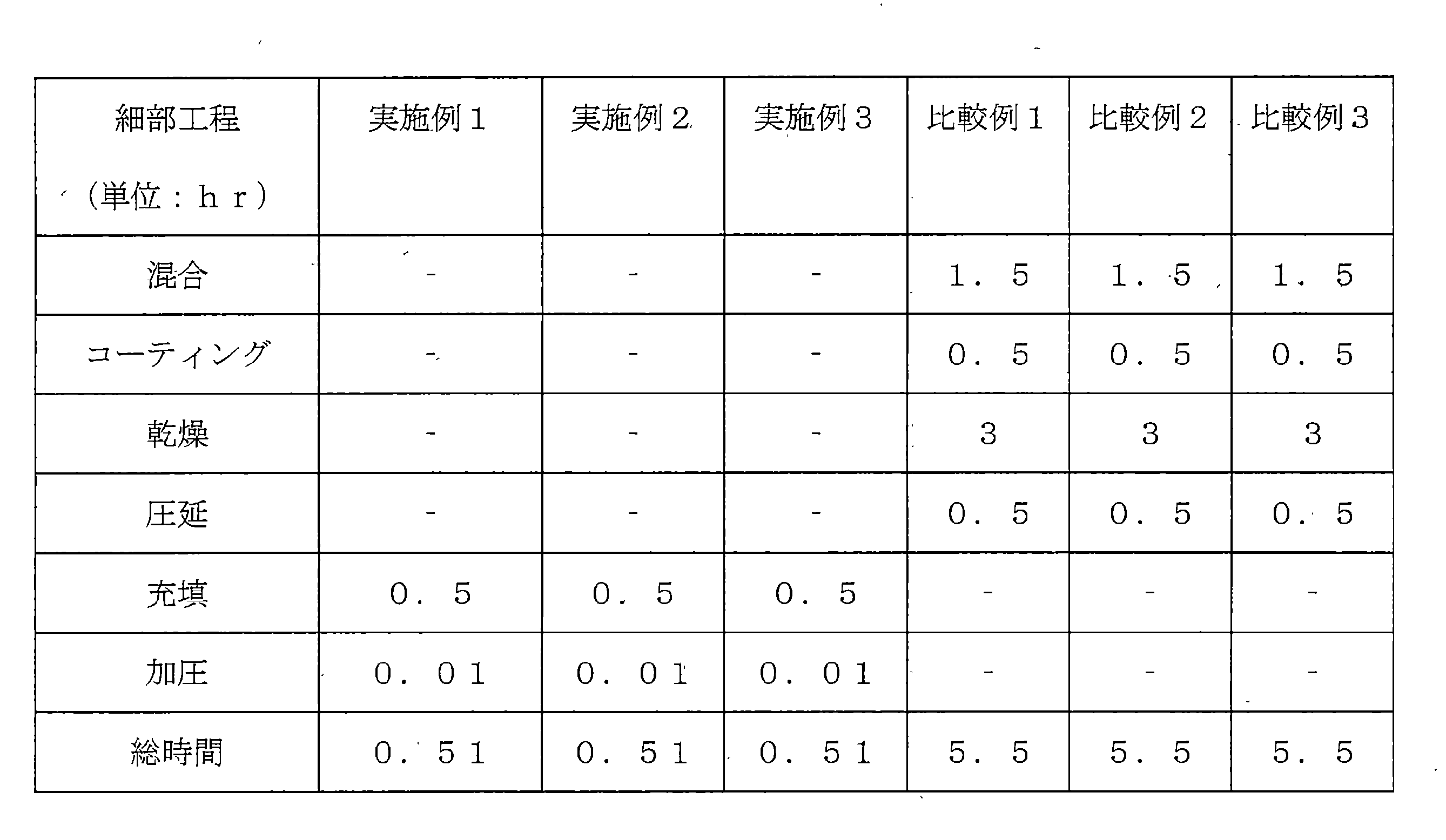
実施例及び比較例で正極材を製造するために工程に要される時間を測定して比べた。工程に必要な時間は下記表1に記載したとおりである。
実施例及び比較例でそれぞれ製造された正極の気孔率を測定して比べ、その結果は表2に記載したとおりである。
実施例及び比較例で製作された電池に対して25℃で0.1C放電、0.1C充電して容量(Capacity、mAh)を測定し、充放電を繰り返して測定することで容量及び充電効率を測定し、その結果を下記表3及び図4aないし図4eに示す。上記表2に示すように、比較例4は加圧圧力が低くて硫黄‐炭素複合体の間の結集力が不足して電極が製造されなかったので、実施例1ないし3と比較例1ないし3及び5と6に対して実験を行った。
製造例1で原料として使われた炭素ナノチューブ(CNT)と硫黄‐炭素複合体(S‐CNT)を加圧工程に適用した後、内部形態を確認した。加圧工程は実施例1の(1)に示すように適用した。
Claims (12)
- フリースタンディングフィルム形態のリチウム二次電池用正極材であって、
上記正極材は炭素を含む硫黄溶融物を含むものである、リチウム二次電池用正極材。 - 上記炭素を含む硫黄溶融物は、上記硫黄溶融物の内部に多孔性炭素材が分散された形態である、請求項1に記載のリチウム二次電池用正極材。
- 上記炭素を含む硫黄溶融物は、硫黄50質量%ないし80質量%及び多孔性炭素材20質量%ないし50質量%で構成されたものである、請求項1に記載のリチウム二次電池用正極材。
- 上記正極材内部の接着力は10gf/cm以上である、請求項1に記載のリチウム二次電池用正極材。
- 上記多孔性炭素材は、グラファイト(graphite);グラフェン(graphene);デンカブラック、アセチレンブラック、ケッチェンブラック、チャンネルブラック、ファーネスブラック、ランプブラック及びサーマルブラックの中で選択されるカーボンブラック;単一壁炭素ナノチューブ(SWCNT)及び多重壁炭素ナノチューブ(MWCNT)が選択される炭素ナノチューブ(CNT);グラファイトナノファイバー(GNF)、カーボンナノファイバー(CNF)及び活性化炭素ファイバー(ACF)の中で選択される炭素繊維;及び活性炭素からなる群から選択される1種以上を含むものである、請求項2に記載のリチウム二次電池用正極材。
- 上記正極材の気孔率は68%以下である、請求項1に記載のリチウム二次電池用正極材。
- (S1)硫黄と多孔性炭素材を混合する段階;
(S2)上記(S1)段階で形成された混合物を熱処理する段階;及び
(S3)上記(S2)段階で形成された硫黄‐炭素複合体を容器に充填した後、加圧して炭素を含む硫黄溶融物を形成する段階;を含むリチウム二次電池用正極材の製造方法。 - 上記熱処理は130℃ないし170℃で行われる、請求項7に記載のリチウム二次電池用正極材の製造方法。
- 上記加圧は0.8MPaないし15MPaの圧力下で行われる、請求項7に記載のリチウム二次電池用正極材の製造方法。
- 請求項1ないし請求項6のいずれか1項に記載の正極材を含む正極;
リチウム金属またはリチウム合金を含む負極;
上記正極と負極の間に位置する分離膜;及び
上記正極、負極及び分離膜が含浸された電解液;
を含むリチウム二次電池。 - 上記リチウム二次電池はリチウム‐硫黄二次電池である、請求項10に記載のリチウム二次電池。
- 上記正極のローディング量は3.0mAh/cm2ないし5.0mAh/cm2である、請求項10に記載のリチウム二次電池。
Applications Claiming Priority (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR10-2020-0063882 | 2020-05-27 | ||
| KR20200063882 | 2020-05-27 | ||
| KR1020210064083A KR102917426B1 (ko) | 2020-05-27 | 2021-05-18 | 프리스탠딩 필름형 리튬 이차전지용 양극재, 이의 제조방법 및 이를 포함하는 리튬 이차전지 |
| KR10-2021-0064083 | 2021-05-18 | ||
| PCT/KR2021/006415 WO2021241959A1 (ko) | 2020-05-27 | 2021-05-24 | 프리스탠딩 필름형 리튬 이차전지용 양극재, 이의 제조방법 및 이를 포함하는 리튬 이차전지 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2022546292A true JP2022546292A (ja) | 2022-11-04 |
| JP7431947B2 JP7431947B2 (ja) | 2024-02-15 |
Family
ID=78744694
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2022511087A Active JP7431947B2 (ja) | 2020-05-27 | 2021-05-24 | フリースタンディングフィルム型リチウム二次電池用正極材、この製造方法及びこれを含むリチウム二次電池 |
Country Status (5)
| Country | Link |
|---|---|
| US (1) | US20220359861A1 (ja) |
| EP (1) | EP4002516A4 (ja) |
| JP (1) | JP7431947B2 (ja) |
| CN (1) | CN114207875B (ja) |
| WO (1) | WO2021241959A1 (ja) |
Families Citing this family (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN114792813B (zh) * | 2022-04-27 | 2023-07-14 | 电子科技大学 | 锂-二氧化碳电池无碳自支撑正极膜材料的制备及电池 |
| US20250286141A1 (en) * | 2024-03-06 | 2025-09-11 | Lyten, Inc. | Electrolyte systems including performance-enhancing additives, and electrochemical cells including the same |
| CN118299671A (zh) * | 2024-06-05 | 2024-07-05 | 中能瑞新(深圳)能源科技有限公司 | 一种锂离子电芯及其制备方法和锂离子电池 |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR20160051610A (ko) * | 2014-10-31 | 2016-05-11 | 주식회사 엘지화학 | 프리스탠딩 필름형 리튬-설퍼 전지용 양극재 및 이를 포함하는 리튬-설퍼 전지 |
| US20180212252A1 (en) * | 2017-01-26 | 2018-07-26 | Caitlin Nicole Dillard | Rapid sulfur melt diffusion into carbon host for making electrodes |
Family Cites Families (16)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR20050052258A (ko) * | 2003-11-29 | 2005-06-02 | 삼성에스디아이 주식회사 | 리튬 설퍼 전지 및 그 제조방법 |
| KR101384198B1 (ko) * | 2011-05-31 | 2014-04-25 | 한양대학교 산학협력단 | 카본 황 복합체의 제조 방법, 이에 의하여 제조된 카본 황 복합체 및 이를 포함하는 리튬황 전지 |
| US20180100037A1 (en) * | 2015-07-13 | 2018-04-12 | Arizona Board Of Regents On Behalf Of The University Of Arizona | Copolymerization of elemental sulfur and epoxy functional styrenics |
| US9306207B2 (en) * | 2012-12-28 | 2016-04-05 | Hyundai Motor Company | Method of fabricating sulfur-infiltrated mesoporous conductive nanocomposites for cathode of lithium-sulfur secondary battery |
| KR101669316B1 (ko) * | 2013-07-30 | 2016-10-25 | 주식회사 엘지화학 | 황-탄소 복합체 및 그의 제조방법 |
| US20170047581A1 (en) * | 2014-02-11 | 2017-02-16 | Batelle Memorial Institute | Additives to enhance electrode wetting and performance and methods of making electrodes comprising the same |
| KR101683963B1 (ko) * | 2014-05-26 | 2016-12-07 | 현대자동차주식회사 | 이중 건식 복합화를 통한 황-탄소 복합체 제조 방법 |
| WO2017053962A1 (en) * | 2015-09-24 | 2017-03-30 | Massachusetts Institute Of Technology | Systems and methods of preparing lithium sulfur electrode using sacrificial template |
| CN109873120A (zh) | 2017-12-05 | 2019-06-11 | 中国科学院大连化学物理研究所 | 无金属集流体、自支撑石墨烯基锂硫电池正极的制备方法 |
| KR102268184B1 (ko) * | 2018-08-08 | 2021-06-22 | 주식회사 엘지화학 | 황-탄소 복합체, 이의 제조방법, 이를 포함하는 양극 및 리튬 이차전지 |
| US20200052279A1 (en) * | 2018-08-13 | 2020-02-13 | Tuqiang Chen | Method of preparing energy storage electrodes |
| CN111224079B (zh) * | 2018-11-27 | 2021-11-05 | 清华大学 | 锂硫电池电极、锂硫电池电极的制备方法及锂硫电池 |
| KR102150848B1 (ko) | 2018-11-28 | 2020-09-02 | 주식회사 보아스에스이 | 신재생에너지 발전시스템 고장 감지방법 |
| BR112021018814A2 (pt) * | 2019-03-22 | 2021-11-30 | Aspen Aerogels Inc | Catodos baseados em aerogel de carbono para baterias de lítio enxofre |
| KR102081775B1 (ko) | 2019-08-22 | 2020-02-26 | 주식회사 엘지화학 | 탄소계 2차 입자를 포함하는 리튬-황 전지용 양극, 이의 제조방법 및 이를 포함하는 리튬-황 전지 |
| US11454837B2 (en) | 2019-11-22 | 2022-09-27 | Eagle Technology, Llc | AOM system with interface board and signal vias and related methods |
-
2021
- 2021-05-24 CN CN202180004710.XA patent/CN114207875B/zh active Active
- 2021-05-24 EP EP21812418.8A patent/EP4002516A4/en active Pending
- 2021-05-24 WO PCT/KR2021/006415 patent/WO2021241959A1/ko not_active Ceased
- 2021-05-24 US US17/634,114 patent/US20220359861A1/en active Pending
- 2021-05-24 JP JP2022511087A patent/JP7431947B2/ja active Active
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR20160051610A (ko) * | 2014-10-31 | 2016-05-11 | 주식회사 엘지화학 | 프리스탠딩 필름형 리튬-설퍼 전지용 양극재 및 이를 포함하는 리튬-설퍼 전지 |
| US20180212252A1 (en) * | 2017-01-26 | 2018-07-26 | Caitlin Nicole Dillard | Rapid sulfur melt diffusion into carbon host for making electrodes |
Also Published As
| Publication number | Publication date |
|---|---|
| JP7431947B2 (ja) | 2024-02-15 |
| EP4002516A1 (en) | 2022-05-25 |
| EP4002516A4 (en) | 2022-11-09 |
| CN114207875A (zh) | 2022-03-18 |
| CN114207875B (zh) | 2024-11-08 |
| WO2021241959A1 (ko) | 2021-12-02 |
| US20220359861A1 (en) | 2022-11-10 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| KR102006727B1 (ko) | 황-탄소 복합체 및 이를 포함하는 리튬-황 전지 | |
| KR101774683B1 (ko) | 전극 활물질 슬러리, 이의 제조 방법 및 이를 포함하는 전고체 이차전지 | |
| WO2020041767A1 (en) | Hybrid and solid-state battery architectures with high loading and methods of manufacture thereof | |
| KR102726743B1 (ko) | 흑연을 포함하지 않는 전고체 전지용 복합 음극 및 이의 제조방법 | |
| KR102917426B1 (ko) | 프리스탠딩 필름형 리튬 이차전지용 양극재, 이의 제조방법 및 이를 포함하는 리튬 이차전지 | |
| US11394054B2 (en) | Polymer microspheres as binders for composite electrolytes | |
| JP5515257B2 (ja) | 双極型二次電池 | |
| KR102771681B1 (ko) | 음극 및 상기 음극을 포함하는 이차 전지 | |
| JP7431947B2 (ja) | フリースタンディングフィルム型リチウム二次電池用正極材、この製造方法及びこれを含むリチウム二次電池 | |
| CN115803904B (zh) | 锂二次电池用正极、其制造方法及包含其的锂二次电池 | |
| CN106848379A (zh) | 含吸湿性物质的锂二次电池用电极及包含其的锂二次电池 | |
| JP5711825B2 (ja) | 一体型電極アセンブリ及び、該一体型電極アセンブリを用いた二次電池 | |
| KR101586536B1 (ko) | 전고상 리튬이차전지용 탄소섬유 시트 집전체의 제조방법 및 탄소섬유 시트 집전체를 포함하는 전고상 리튬이차전지 | |
| JP6212305B2 (ja) | 複合集電体、およびそれを用いた電極と二次電池 | |
| JP2022550821A (ja) | 穴あけされた集電体を含むリチウム二次電池用電極、その製造方法及び前記電極を含むリチウム二次電池 | |
| CN116250099A (zh) | 硫-碳复合材料、其制备方法和包含其的锂硫电池 | |
| CN119895624A (zh) | 全固态电池用正极以及包含该正极的全固态电池 | |
| CN118575323A (zh) | 固体电解质膜及包含其的全固态电池 | |
| JP7039846B2 (ja) | 非水電解質二次電池の製造方法 | |
| CN118843959A (zh) | 包含不同类型的导电材料的锂硫电池用正极和包含其的锂离子二次电池 | |
| CN116235308A (zh) | 制造膜型正极的设备、其制造方法、以及包含其的锂二次电池、电池模块和电池组 | |
| JP2011029136A (ja) | 二次電池用電極、二次電池、及び二次電池用電極の製造方法 | |
| US12126004B2 (en) | Fabrication of Si-MWCNT nanocomposites (SMC) as anodes for lithium-ion batteries | |
| JP7497527B2 (ja) | リチウム二次電池用正極、この製造方法及びこれを含むリチウム二次電池 | |
| JP2024174503A (ja) | 非水電解質二次電池 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20220218 |
|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20230216 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20230403 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20230628 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20230807 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20231101 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20240122 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20240202 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 7431947 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 |