JP2017532178A - 創傷被覆用具 - Google Patents
創傷被覆用具 Download PDFInfo
- Publication number
- JP2017532178A JP2017532178A JP2017540337A JP2017540337A JP2017532178A JP 2017532178 A JP2017532178 A JP 2017532178A JP 2017540337 A JP2017540337 A JP 2017540337A JP 2017540337 A JP2017540337 A JP 2017540337A JP 2017532178 A JP2017532178 A JP 2017532178A
- Authority
- JP
- Japan
- Prior art keywords
- fibroin
- wound dressing
- layer
- wound
- dressing device
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
- 108010022355 Fibroins Proteins 0.000 claims abstract description 124
- 208000027418 Wounds and injury Diseases 0.000 claims abstract description 120
- 206010052428 Wound Diseases 0.000 claims abstract description 117
- 239000002250 absorbent Substances 0.000 claims abstract description 56
- 230000002745 absorbent Effects 0.000 claims abstract description 56
- 239000002121 nanofiber Substances 0.000 claims abstract description 38
- 238000000034 method Methods 0.000 claims abstract description 12
- 239000000835 fiber Substances 0.000 claims description 32
- BDAGIHXWWSANSR-UHFFFAOYSA-N methanoic acid Natural products OC=O BDAGIHXWWSANSR-UHFFFAOYSA-N 0.000 claims description 18
- 239000000463 material Substances 0.000 claims description 17
- 241000894006 Bacteria Species 0.000 claims description 16
- 238000011282 treatment Methods 0.000 claims description 13
- 239000003899 bactericide agent Substances 0.000 claims description 11
- 229920000297 Rayon Polymers 0.000 claims description 10
- 229920000728 polyester Polymers 0.000 claims description 10
- OSWFIVFLDKOXQC-UHFFFAOYSA-N 4-(3-methoxyphenyl)aniline Chemical compound COC1=CC=CC(C=2C=CC(N)=CC=2)=C1 OSWFIVFLDKOXQC-UHFFFAOYSA-N 0.000 claims description 9
- 235000019253 formic acid Nutrition 0.000 claims description 9
- 239000000203 mixture Substances 0.000 claims description 7
- 238000004519 manufacturing process Methods 0.000 claims description 6
- 229920000742 Cotton Polymers 0.000 claims description 5
- 238000010521 absorption reaction Methods 0.000 claims description 4
- 239000006260 foam Substances 0.000 claims description 4
- 239000007788 liquid Substances 0.000 claims description 4
- 239000002245 particle Substances 0.000 claims description 4
- 229920003043 Cellulose fiber Polymers 0.000 claims description 3
- 239000004480 active ingredient Substances 0.000 claims description 2
- 238000013019 agitation Methods 0.000 claims description 2
- 210000001124 body fluid Anatomy 0.000 claims description 2
- 238000001523 electrospinning Methods 0.000 claims description 2
- 238000003756 stirring Methods 0.000 claims description 2
- 239000010839 body fluid Substances 0.000 claims 1
- 239000000126 substance Substances 0.000 abstract description 21
- 230000035876 healing Effects 0.000 abstract description 5
- 230000000694 effects Effects 0.000 abstract description 3
- 239000010410 layer Substances 0.000 description 78
- 239000003814 drug Substances 0.000 description 11
- 208000004210 Pressure Ulcer Diseases 0.000 description 8
- 230000000844 anti-bacterial effect Effects 0.000 description 8
- 238000001878 scanning electron micrograph Methods 0.000 description 8
- 208000015181 infectious disease Diseases 0.000 description 7
- 206010011985 Decubitus ulcer Diseases 0.000 description 5
- 230000001684 chronic effect Effects 0.000 description 5
- 229940079593 drug Drugs 0.000 description 5
- 230000003993 interaction Effects 0.000 description 5
- 239000011159 matrix material Substances 0.000 description 5
- 239000002086 nanomaterial Substances 0.000 description 5
- 229920000642 polymer Polymers 0.000 description 5
- 230000001846 repelling effect Effects 0.000 description 5
- 206010017533 Fungal infection Diseases 0.000 description 4
- 208000031888 Mycoses Diseases 0.000 description 4
- 206010042496 Sunburn Diseases 0.000 description 4
- 208000002847 Surgical Wound Diseases 0.000 description 4
- 239000000853 adhesive Substances 0.000 description 4
- 230000001070 adhesive effect Effects 0.000 description 4
- 210000000416 exudates and transudate Anatomy 0.000 description 4
- 239000003102 growth factor Substances 0.000 description 4
- 231100000397 ulcer Toxicity 0.000 description 4
- RDEIXVOBVLKYNT-VQBXQJRRSA-N (2r,3r,4r,5r)-2-[(1s,2s,3r,4s,6r)-4,6-diamino-3-[(2r,3r,6s)-3-amino-6-(1-aminoethyl)oxan-2-yl]oxy-2-hydroxycyclohexyl]oxy-5-methyl-4-(methylamino)oxane-3,5-diol;(2r,3r,4r,5r)-2-[(1s,2s,3r,4s,6r)-4,6-diamino-3-[(2r,3r,6s)-3-amino-6-(aminomethyl)oxan-2-yl]o Chemical compound OS(O)(=O)=O.O1C[C@@](O)(C)[C@H](NC)[C@@H](O)[C@H]1O[C@@H]1[C@@H](O)[C@H](O[C@@H]2[C@@H](CC[C@@H](CN)O2)N)[C@@H](N)C[C@H]1N.O1C[C@@](O)(C)[C@H](NC)[C@@H](O)[C@H]1O[C@@H]1[C@@H](O)[C@H](O[C@@H]2[C@@H](CC[C@H](O2)C(C)N)N)[C@@H](N)C[C@H]1N.O1[C@H](C(C)NC)CC[C@@H](N)[C@H]1O[C@H]1[C@H](O)[C@@H](O[C@@H]2[C@@H]([C@@H](NC)[C@@](C)(O)CO2)O)[C@H](N)C[C@@H]1N RDEIXVOBVLKYNT-VQBXQJRRSA-N 0.000 description 3
- 108010010803 Gelatin Proteins 0.000 description 3
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 3
- 229920003171 Poly (ethylene oxide) Polymers 0.000 description 3
- 239000012790 adhesive layer Substances 0.000 description 3
- 239000002998 adhesive polymer Substances 0.000 description 3
- 230000006378 damage Effects 0.000 description 3
- 239000008273 gelatin Substances 0.000 description 3
- 229920000159 gelatin Polymers 0.000 description 3
- 235000019322 gelatine Nutrition 0.000 description 3
- 235000011852 gelatine desserts Nutrition 0.000 description 3
- 208000014674 injury Diseases 0.000 description 3
- 239000004005 microsphere Substances 0.000 description 3
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 3
- 230000029663 wound healing Effects 0.000 description 3
- 108010037362 Extracellular Matrix Proteins Proteins 0.000 description 2
- 102000010834 Extracellular Matrix Proteins Human genes 0.000 description 2
- 208000025865 Ulcer Diseases 0.000 description 2
- 230000001580 bacterial effect Effects 0.000 description 2
- 239000011230 binding agent Substances 0.000 description 2
- 210000004027 cell Anatomy 0.000 description 2
- 230000010261 cell growth Effects 0.000 description 2
- 229920002678 cellulose Polymers 0.000 description 2
- 239000001913 cellulose Substances 0.000 description 2
- 150000001875 compounds Chemical class 0.000 description 2
- 238000007796 conventional method Methods 0.000 description 2
- 210000002744 extracellular matrix Anatomy 0.000 description 2
- 239000012530 fluid Substances 0.000 description 2
- 230000012010 growth Effects 0.000 description 2
- 230000002209 hydrophobic effect Effects 0.000 description 2
- 238000012606 in vitro cell culture Methods 0.000 description 2
- OKJPEAGHQZHRQV-UHFFFAOYSA-N iodoform Chemical compound IC(I)I OKJPEAGHQZHRQV-UHFFFAOYSA-N 0.000 description 2
- AMXOYNBUYSYVKV-UHFFFAOYSA-M lithium bromide Chemical compound [Li+].[Br-] AMXOYNBUYSYVKV-UHFFFAOYSA-M 0.000 description 2
- 244000005700 microbiome Species 0.000 description 2
- 230000004048 modification Effects 0.000 description 2
- 238000012986 modification Methods 0.000 description 2
- 230000000877 morphologic effect Effects 0.000 description 2
- 238000009581 negative-pressure wound therapy Methods 0.000 description 2
- 230000010399 physical interaction Effects 0.000 description 2
- 108090000623 proteins and genes Proteins 0.000 description 2
- 102000004169 proteins and genes Human genes 0.000 description 2
- 229910052709 silver Inorganic materials 0.000 description 2
- 239000004332 silver Substances 0.000 description 2
- 229940124597 therapeutic agent Drugs 0.000 description 2
- 210000001519 tissue Anatomy 0.000 description 2
- 241000255789 Bombyx mori Species 0.000 description 1
- 208000035473 Communicable disease Diseases 0.000 description 1
- 208000034656 Contusions Diseases 0.000 description 1
- 206010056340 Diabetic ulcer Diseases 0.000 description 1
- 229920001410 Microfiber Polymers 0.000 description 1
- 239000002253 acid Substances 0.000 description 1
- 230000009471 action Effects 0.000 description 1
- 239000002313 adhesive film Substances 0.000 description 1
- 230000002411 adverse Effects 0.000 description 1
- 238000004458 analytical method Methods 0.000 description 1
- 239000003242 anti bacterial agent Substances 0.000 description 1
- 229940088710 antibiotic agent Drugs 0.000 description 1
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 1
- 230000003796 beauty Effects 0.000 description 1
- 239000012620 biological material Substances 0.000 description 1
- 239000010836 blood and blood product Substances 0.000 description 1
- 229940125691 blood product Drugs 0.000 description 1
- 230000015556 catabolic process Effects 0.000 description 1
- 230000021164 cell adhesion Effects 0.000 description 1
- 230000004663 cell proliferation Effects 0.000 description 1
- 239000011248 coating agent Substances 0.000 description 1
- 238000000576 coating method Methods 0.000 description 1
- 239000002537 cosmetic Substances 0.000 description 1
- 238000002425 crystallisation Methods 0.000 description 1
- 230000008025 crystallization Effects 0.000 description 1
- 238000006731 degradation reaction Methods 0.000 description 1
- 239000000645 desinfectant Substances 0.000 description 1
- 239000004205 dimethyl polysiloxane Substances 0.000 description 1
- 235000013870 dimethyl polysiloxane Nutrition 0.000 description 1
- 230000009881 electrostatic interaction Effects 0.000 description 1
- 239000007789 gas Substances 0.000 description 1
- 239000000499 gel Substances 0.000 description 1
- 229910052602 gypsum Inorganic materials 0.000 description 1
- 239000010440 gypsum Substances 0.000 description 1
- 230000036074 healthy skin Effects 0.000 description 1
- 239000000017 hydrogel Substances 0.000 description 1
- 238000011221 initial treatment Methods 0.000 description 1
- 230000007774 longterm Effects 0.000 description 1
- 238000005259 measurement Methods 0.000 description 1
- 239000003658 microfiber Substances 0.000 description 1
- CXQXSVUQTKDNFP-UHFFFAOYSA-N octamethyltrisiloxane Chemical compound C[Si](C)(C)O[Si](C)(C)O[Si](C)(C)C CXQXSVUQTKDNFP-UHFFFAOYSA-N 0.000 description 1
- 230000003287 optical effect Effects 0.000 description 1
- 239000001301 oxygen Substances 0.000 description 1
- 229910052760 oxygen Inorganic materials 0.000 description 1
- 230000002093 peripheral effect Effects 0.000 description 1
- 230000035699 permeability Effects 0.000 description 1
- 239000012466 permeate Substances 0.000 description 1
- 238000004987 plasma desorption mass spectroscopy Methods 0.000 description 1
- 239000011505 plaster Substances 0.000 description 1
- 229920000435 poly(dimethylsiloxane) Polymers 0.000 description 1
- 229920006254 polymer film Polymers 0.000 description 1
- 229920001296 polysiloxane Polymers 0.000 description 1
- 230000002980 postoperative effect Effects 0.000 description 1
- 238000012545 processing Methods 0.000 description 1
- 230000001737 promoting effect Effects 0.000 description 1
- 150000003856 quaternary ammonium compounds Chemical class 0.000 description 1
- 231100000241 scar Toxicity 0.000 description 1
- 210000003802 sputum Anatomy 0.000 description 1
- 238000013269 sustained drug release Methods 0.000 description 1
- 230000000699 topical effect Effects 0.000 description 1
- 238000012546 transfer Methods 0.000 description 1
- 238000005406 washing Methods 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L15/00—Chemical aspects of, or use of materials for, bandages, dressings or absorbent pads
- A61L15/16—Bandages, dressings or absorbent pads for physiological fluids such as urine or blood, e.g. sanitary towels, tampons
- A61L15/22—Bandages, dressings or absorbent pads for physiological fluids such as urine or blood, e.g. sanitary towels, tampons containing macromolecular materials
- A61L15/32—Proteins, polypeptides; Degradation products or derivatives thereof, e.g. albumin, collagen, fibrin, gelatin
-
- A61F13/01012—
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F13/00—Bandages or dressings; Absorbent pads
- A61F13/02—Adhesive plasters or dressings
- A61F13/0203—Adhesive plasters or dressings having a fluid handling member
- A61F13/0206—Adhesive plasters or dressings having a fluid handling member the fluid handling member being absorbent fibrous layer, e.g. woven or nonwoven absorbent pad, island dressings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F13/00—Bandages or dressings; Absorbent pads
- A61F13/02—Adhesive plasters or dressings
- A61F13/0246—Adhesive plasters or dressings characterised by the skin adhering layer
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F13/00—Bandages or dressings; Absorbent pads
- A61F13/15—Absorbent pads, e.g. sanitary towels, swabs or tampons for external or internal application to the body; Supporting or fastening means therefor; Tampon applicators
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L15/00—Chemical aspects of, or use of materials for, bandages, dressings or absorbent pads
- A61L15/16—Bandages, dressings or absorbent pads for physiological fluids such as urine or blood, e.g. sanitary towels, tampons
- A61L15/18—Bandages, dressings or absorbent pads for physiological fluids such as urine or blood, e.g. sanitary towels, tampons containing inorganic materials
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L15/00—Chemical aspects of, or use of materials for, bandages, dressings or absorbent pads
- A61L15/16—Bandages, dressings or absorbent pads for physiological fluids such as urine or blood, e.g. sanitary towels, tampons
- A61L15/22—Bandages, dressings or absorbent pads for physiological fluids such as urine or blood, e.g. sanitary towels, tampons containing macromolecular materials
- A61L15/26—Macromolecular compounds obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds; Derivatives thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L15/00—Chemical aspects of, or use of materials for, bandages, dressings or absorbent pads
- A61L15/16—Bandages, dressings or absorbent pads for physiological fluids such as urine or blood, e.g. sanitary towels, tampons
- A61L15/22—Bandages, dressings or absorbent pads for physiological fluids such as urine or blood, e.g. sanitary towels, tampons containing macromolecular materials
- A61L15/28—Polysaccharides or their derivatives
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L15/00—Chemical aspects of, or use of materials for, bandages, dressings or absorbent pads
- A61L15/16—Bandages, dressings or absorbent pads for physiological fluids such as urine or blood, e.g. sanitary towels, tampons
- A61L15/42—Use of materials characterised by their function or physical properties
- A61L15/44—Medicaments
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L15/00—Chemical aspects of, or use of materials for, bandages, dressings or absorbent pads
- A61L15/16—Bandages, dressings or absorbent pads for physiological fluids such as urine or blood, e.g. sanitary towels, tampons
- A61L15/42—Use of materials characterised by their function or physical properties
- A61L15/46—Deodorants or malodour counteractants, e.g. to inhibit the formation of ammonia or bacteria
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L15/00—Chemical aspects of, or use of materials for, bandages, dressings or absorbent pads
- A61L15/16—Bandages, dressings or absorbent pads for physiological fluids such as urine or blood, e.g. sanitary towels, tampons
- A61L15/42—Use of materials characterised by their function or physical properties
- A61L15/58—Adhesives
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F13/00—Bandages or dressings; Absorbent pads
- A61F2013/00089—Wound bandages
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F13/00—Bandages or dressings; Absorbent pads
- A61F13/15—Absorbent pads, e.g. sanitary towels, swabs or tampons for external or internal application to the body; Supporting or fastening means therefor; Tampon applicators
- A61F2013/15008—Absorbent pads, e.g. sanitary towels, swabs or tampons for external or internal application to the body; Supporting or fastening means therefor; Tampon applicators characterized by the use
- A61F2013/15048—Absorbent pads, e.g. sanitary towels, swabs or tampons for external or internal application to the body; Supporting or fastening means therefor; Tampon applicators characterized by the use for protection against contamination, or protection in using body disinfecting wipes
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F13/00—Bandages or dressings; Absorbent pads
- A61F13/15—Absorbent pads, e.g. sanitary towels, swabs or tampons for external or internal application to the body; Supporting or fastening means therefor; Tampon applicators
- A61F13/53—Absorbent pads, e.g. sanitary towels, swabs or tampons for external or internal application to the body; Supporting or fastening means therefor; Tampon applicators characterised by the absorbing medium
- A61F2013/530131—Absorbent pads, e.g. sanitary towels, swabs or tampons for external or internal application to the body; Supporting or fastening means therefor; Tampon applicators characterised by the absorbing medium being made in fibre but being not pulp
- A61F2013/530343—Absorbent pads, e.g. sanitary towels, swabs or tampons for external or internal application to the body; Supporting or fastening means therefor; Tampon applicators characterised by the absorbing medium being made in fibre but being not pulp being natural fibres
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L2300/00—Biologically active materials used in bandages, wound dressings, absorbent pads or medical devices
- A61L2300/40—Biologically active materials used in bandages, wound dressings, absorbent pads or medical devices characterised by a specific therapeutic activity or mode of action
- A61L2300/404—Biocides, antimicrobial agents, antiseptic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L2400/00—Materials characterised by their function or physical properties
- A61L2400/12—Nanosized materials, e.g. nanofibres, nanoparticles, nanowires, nanotubes; Nanostructured surfaces
Abstract
Description
PEOは、使用者の体内に放出される蚕の繭、医薬品、活性成分、及び/又は殺菌性物質の前処理及び処理の間には、決して使用されない。
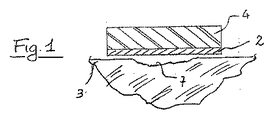
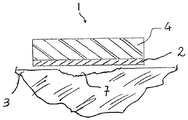
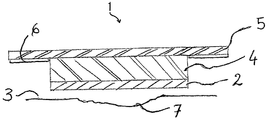
図1〜図2Bを参照して、本発明の一態様を説明する。創傷被覆用具1は、吸収剤層4,及び前述した特徴をもつフィブロインのナノ繊維で製造された創傷接触層を備えている。フィブロイン層は、創傷7と接触している。フィブロインナノ繊維の層2の一部、通常は周辺部分は、使用者の皮膚3とも接触している。
a.純正フィブロインメッシュを、室温、穏やかな攪拌条件下で、CaCl2、及び蟻酸と接触させて、可溶性にする。
b.工程(a)で得た粘稠溶液を、適当な容器に注ぎ、放置して残留蟻酸を気化させる。
c.工程(b)のフィブロイン及びCaCl2の混合物を、CaCl2から精製して、フィブロインフィルムを得る。
d.工程(c)のフィルムを剥離し、放置乾燥させる。
e.フィルムを、穏やかな攪拌条件下、蟻酸で処理し、フィブロインとび蟻酸の溶液を、シリンジに移し、静電紡糸する。
2:フィブロインナノ繊維の層
3:皮膚
4:吸収剤層
5:フィルム裏打ち材
6:ポリマー層
7:創傷
Claims (23)
- 少なくとも1枚の絹フィブロインナノ繊維の層(2)を含む創傷被覆用具であって、
前記フィブロインナノ繊維の層(2)において、フィブロインは、ナノ繊維から製造されていること、前記フィブロインナノ繊維の層(2)は、10〜40g/m2の範囲の質量を有していること、及び前記用具は、更に、吸収剤層(4)を含んでいることを特徴とする創傷被覆用具。 - 前記フィブロインナノ繊維の層(2)は、900nm未満の平均繊維直径を有している、請求項1に記載の創傷被覆用具。
- 前記フィブロインナノ繊維の層(2)は、200〜800nmの範囲、好ましくは400〜700nmの範囲の平均繊維直径を有している、請求項1又は2に記載の創傷被覆用具。
- 前記フィブロインナノ繊維の層(2)は、静電紡糸層である、請求項1又は2に記載の創傷被覆用具。
- 前記フィブロインナノ繊維の層(2)は、医薬品、活性成分又は殺菌性物質を含まない、請求項1〜4のいずれか1項に記載の創傷被覆用具。
- 前記フィブロインナノ繊維の層(2)は、25〜35g/m2の範囲の質量を有している、請求項1〜5のいずれか1項に記載の創傷被覆用具。
- 前記フィブロインナノ繊維の層(2)は、200〜800nmの範囲の平均繊維直径を有している、請求項1〜6のいずれか1項に記載の創傷被覆用具。
- 前記吸収剤層(4)は、フィブロインの2つの層の間に配置されている、請求項1〜7のいずれか1項に記載の創傷被覆用具。
- 1枚のフィブロイン層及び1枚の吸収剤層(4)から製造された用具を折り畳むことによって製造された請求項8に記載の創傷被覆用具。
- 前記吸収剤層(4)は、ビスコース繊維、ポリエステル繊維、セルロース繊維、コットン繊維、発泡体、ゲル形成粒子、ゲル形成繊維、ガーゼ、及び液体吸収性能がある創傷被覆部材から選択される少なくとも1つの材料である、請求項1〜9のいずれか1項に記載の創傷被覆用具。
- 前記吸収剤層(4)は、ビスコース/ポリエステルの質量比70/30の混合物である、請求項9に記載の創傷被覆用具。
- 前記吸収剤層(4)は、更に、殺菌性物質を含んでいる、請求項1〜11のいずれか1項に記載の創傷被覆用具。
- さらに、前記創傷被覆用具の少なくとも一部を、使用者の皮膚(3)に接着する手段(5,6)を含む、請求項1〜12のいずれか1項に記載の創傷被覆用具。
- 900nm未満の平均繊維直径を有するナノ繊維で製造されたフィブロイン層であって、創傷(7)を治療する用具(1)内に細菌を封じ込める要素として使用されるフィブロイン層。
- 前記平均繊維直径は、400〜700nmの範囲にある、請求項13に記載のフィブロイン層。
- 前記フィブロイン層は、静電紡糸フィブロインである、請求項14または15に記載のフィブロイン層。
- 体液吸収剤として使用するための請求項1〜13のいずれか1項に記載の創傷被覆用具(1)。
- 創傷の治療のために使用される請求項1〜13のいずれか1項に記載の創傷被覆用具(1)。
- 負圧創傷治療(NPWT)に使用される、請求項1〜13のいずれか1項に記載の創傷被覆用具(1)。
- 創傷被覆用具を製造する方法であって;
a.純正フィブロインメッシュを、室温、穏やかな攪拌条件下で、CaCl2、及び蟻酸と接触させて、可溶性にすること;
b.工程(a)で得た粘稠溶液を、適当な容器に注ぎ、放置して残留蟻酸を気化させること;
c.工程(b)のフィブロイン及びCaCl2の混合物を、CaCl2から精製して、フィブロインフルムを得ること;
d.工程(c)のフィルムを剥離し、放置乾燥すること;
e.フィルムを、穏やかな攪拌条件下、蟻酸で処理し、フィブロインとび蟻酸の溶液を、シリンジに移し、静電紡糸することを含む創傷被覆用具を製造する方法。 - 請求項20の方法により製造された創傷被覆用具。
- 前記吸収剤層は、ビスコース繊維、ポリエステル繊維、セルロース繊維、コットン繊維、発泡体、ゲル形成粒子、ゲル形成繊維、ガーゼ、及び液体吸収性能がある創傷被覆部材から選択される、請求項21に記載の創傷被覆用具。
- 前記吸収剤は、ビスコース/ポリエステルの質量比70/30の混合物である、請求項21に記載の創傷被覆用具。
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP14189451.9 | 2014-10-17 | ||
| EP14189451 | 2014-10-17 | ||
| PCT/IB2015/057965 WO2016059611A1 (en) | 2014-10-17 | 2015-10-16 | "device for wound dressing" |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| JP2017532178A true JP2017532178A (ja) | 2017-11-02 |
Family
ID=51751968
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2017540337A Pending JP2017532178A (ja) | 2014-10-17 | 2015-10-16 | 創傷被覆用具 |
Country Status (14)
| Country | Link |
|---|---|
| US (1) | US20170216478A1 (ja) |
| EP (2) | EP3517142A1 (ja) |
| JP (1) | JP2017532178A (ja) |
| KR (1) | KR20170085504A (ja) |
| CN (1) | CN107106722A (ja) |
| AU (1) | AU2015331990A1 (ja) |
| BR (1) | BR112017007849A2 (ja) |
| CA (1) | CA2963454A1 (ja) |
| ES (1) | ES2734730T3 (ja) |
| IL (1) | IL251493A0 (ja) |
| MX (1) | MX2017004844A (ja) |
| PL (1) | PL3206725T3 (ja) |
| RU (1) | RU2017116964A (ja) |
| WO (1) | WO2016059611A1 (ja) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2021066078A1 (ja) * | 2019-09-30 | 2021-04-08 | Spiber株式会社 | 筋組織再生剤 |
Families Citing this family (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN109232464B (zh) | 2017-07-10 | 2022-11-15 | 上海新理念生物医药科技有限公司 | 噁二唑型连接子及其应用 |
| IT201700091328A1 (it) * | 2017-08-07 | 2019-02-07 | Leonardino S R L | Matrice elettrofilata a base di proteine della seta, procedimento per la sua preparazione, manufatti che la comprendono e loro uso in terapia ed in cosmetica |
| US20210236644A1 (en) | 2017-11-10 | 2021-08-05 | Cocoon Biotech Inc. | Ocular applications of silk-based products |
| CN108939129A (zh) * | 2018-07-08 | 2018-12-07 | 东莞市联洲知识产权运营管理有限公司 | 一种聚吡咯/蚕丝蛋白晶体的制备方法及其制备的医用敷料 |
| CA3142608A1 (en) | 2019-06-04 | 2020-12-10 | Cocoon Biotech Inc. | Silk-based products, formulations, and methods of use |
| CN110215350B (zh) * | 2019-06-06 | 2021-09-07 | 杭州贝高母婴用品有限公司 | 一种干爽、透气的纸尿裤及其生产工艺 |
| US11311641B1 (en) | 2020-11-20 | 2022-04-26 | Mehrdad Mark Mofid | Scaffold wound dressing |
| CN113813437B (zh) * | 2021-09-28 | 2022-07-01 | 振德医疗用品股份有限公司 | 基于细菌非特异性粘附的抗感染防黏连创面敷料 |
| CN114631934A (zh) * | 2022-03-14 | 2022-06-17 | 中国人民解放军联勤保障部队第九二〇医院 | 一种可营造低氧微环境促急性创面早期愈合的敷料 |
| CN115137557B (zh) * | 2022-07-12 | 2023-01-10 | 浙江理工大学 | 一种具有定向导液功能的纤维基敷料的制备方法 |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2009523591A (ja) * | 2006-01-24 | 2009-06-25 | アイダブリューエムティー インテレクチャル プロパティ ホールディングス(プロプライエタリー)リミテッド | 創傷被覆材 |
| JP2012533354A (ja) * | 2009-07-14 | 2012-12-27 | タフツ ユニバーシティー/トラスティーズ オブ タフツ カレッジ | 創傷治癒のための電界紡糸絹材料システム |
| CN103341209A (zh) * | 2013-07-08 | 2013-10-09 | 苏州大学 | 一种丝素蛋白纳米纤维膜及其制备方法 |
Family Cites Families (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN100482189C (zh) * | 2005-09-07 | 2009-04-29 | 浙江大学 | 一种生物敷料及其制备方法 |
| CN101736430B (zh) * | 2010-02-05 | 2012-07-04 | 东华大学 | 具有护肤功效的丝素蛋白纳米纤维的制备方法 |
| SG10201607262PA (en) * | 2010-08-30 | 2016-10-28 | Harvard College | A High Strength Chitin Composite Material and Method of Making |
| WO2012040310A2 (en) | 2010-09-22 | 2012-03-29 | Virginia Commonwealth University | Preparation rich in growth factor-based fibrous matrices for tissue engineering, growth factor delivery, and wound healing |
| EP2545943A1 (de) | 2011-07-12 | 2013-01-16 | Dr. Suwelack Skin & Health Care AG | Perforiertes, geschichtetes Wundbehandlungsmaterial |
| GB2504872B (en) * | 2011-11-01 | 2015-07-01 | Brightwake Ltd | Wound dressings, and yarn useful therein |
| US9101508B2 (en) * | 2011-12-07 | 2015-08-11 | Esmaeil Mirzaei | Electro spun nanofibrous wound dressing and a method of synthesizing the same |
| CN102560888B (zh) * | 2012-01-17 | 2014-08-13 | 东华大学 | 负载水溶性维生素e的丝素蛋白纳米纤维膜及其制备方法 |
| CN103341214B (zh) * | 2013-07-08 | 2014-12-31 | 苏州大学 | 一种丝素蛋白膜及其制备方法 |
| CN103446617A (zh) | 2013-08-28 | 2013-12-18 | 暨南大学 | 负载硫酸庆大霉素/明胶微球复合物的丝素蛋白支架及制备方法 |
-
2015
- 2015-10-16 JP JP2017540337A patent/JP2017532178A/ja active Pending
- 2015-10-16 EP EP19162289.3A patent/EP3517142A1/en not_active Withdrawn
- 2015-10-16 WO PCT/IB2015/057965 patent/WO2016059611A1/en active Application Filing
- 2015-10-16 PL PL15798194T patent/PL3206725T3/pl unknown
- 2015-10-16 AU AU2015331990A patent/AU2015331990A1/en not_active Abandoned
- 2015-10-16 MX MX2017004844A patent/MX2017004844A/es unknown
- 2015-10-16 BR BR112017007849A patent/BR112017007849A2/pt not_active Application Discontinuation
- 2015-10-16 KR KR1020177012899A patent/KR20170085504A/ko unknown
- 2015-10-16 CA CA2963454A patent/CA2963454A1/en not_active Abandoned
- 2015-10-16 US US15/518,632 patent/US20170216478A1/en not_active Abandoned
- 2015-10-16 EP EP15798194.5A patent/EP3206725B1/en active Active
- 2015-10-16 CN CN201580068180.XA patent/CN107106722A/zh active Pending
- 2015-10-16 RU RU2017116964A patent/RU2017116964A/ru not_active Application Discontinuation
- 2015-10-16 ES ES15798194T patent/ES2734730T3/es active Active
-
2017
- 2017-04-02 IL IL251493A patent/IL251493A0/en unknown
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2009523591A (ja) * | 2006-01-24 | 2009-06-25 | アイダブリューエムティー インテレクチャル プロパティ ホールディングス(プロプライエタリー)リミテッド | 創傷被覆材 |
| JP2012533354A (ja) * | 2009-07-14 | 2012-12-27 | タフツ ユニバーシティー/トラスティーズ オブ タフツ カレッジ | 創傷治癒のための電界紡糸絹材料システム |
| CN103341209A (zh) * | 2013-07-08 | 2013-10-09 | 苏州大学 | 一种丝素蛋白纳米纤维膜及其制备方法 |
Non-Patent Citations (1)
| Title |
|---|
| KIM, SO HYN: "Silk Fibroin Nanofiber. Electrospinning, Properties, and Structure", POLYMER JOURNAL, vol. 35, no. 2, JPN7019002818, 1 January 2003 (2003-01-01), pages 185 - 190, XP055239412, ISSN: 0004248641, DOI: 10.1295/polymj.35.185 * |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2021066078A1 (ja) * | 2019-09-30 | 2021-04-08 | Spiber株式会社 | 筋組織再生剤 |
| CN114555108A (zh) * | 2019-09-30 | 2022-05-27 | 丝芭博株式会社 | 肌肉组织再生剂 |
Also Published As
| Publication number | Publication date |
|---|---|
| RU2017116964A3 (ja) | 2019-04-23 |
| ES2734730T3 (es) | 2019-12-11 |
| RU2017116964A (ru) | 2018-11-19 |
| AU2015331990A1 (en) | 2017-05-04 |
| EP3206725B1 (en) | 2019-04-17 |
| PL3206725T3 (pl) | 2019-09-30 |
| EP3206725A1 (en) | 2017-08-23 |
| US20170216478A1 (en) | 2017-08-03 |
| IL251493A0 (en) | 2017-05-29 |
| WO2016059611A1 (en) | 2016-04-21 |
| KR20170085504A (ko) | 2017-07-24 |
| MX2017004844A (es) | 2018-01-24 |
| BR112017007849A2 (pt) | 2018-01-16 |
| CN107106722A (zh) | 2017-08-29 |
| EP3517142A1 (en) | 2019-07-31 |
| CA2963454A1 (en) | 2016-04-21 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP2017532178A (ja) | 創傷被覆用具 | |
| EP2747792B1 (de) | Wundauflage aufweisend einen anteil an kupfer bzw. kupferionen | |
| EP2809363B1 (de) | Wundpflegeartikel, aufweisend mindestens eine oberfläche mit abrasiven eigenschaften | |
| KR20090112707A (ko) | 박테리아 흡착 구성물 및 수분 유지 시스템을 포함하는 상처 드레싱 | |
| Bielecki et al. | Wound dressings and cosmetic materials from bacterial nanocellulose | |
| JP2022523780A (ja) | 抗菌ドレッシング、ドレッシング構成要素、及び方法 | |
| RU2578458C2 (ru) | Медицинская многослойная повязка с многофункциональными наномембранами и изделия на ее основе | |
| DE102012100842A1 (de) | Wundpflegeartikel, aufweisend mindestens eine Oberfläche mit abrasiven Eigenschaften | |
| JP6290184B2 (ja) | 創傷ドレッシング | |
| US20140236107A1 (en) | Absorbent hygienic or personal care product having a strip-like component with a content of at least one heavy metal present in elemental or ionic form | |
| CA2902382C (en) | Skin substitute / wound dressing with variable pore sizes | |
| JP2005102790A (ja) | 創傷被覆材 | |
| Mao et al. | Nonwoven wound dressings | |
| RU2240140C2 (ru) | Медицинская многослойная повязка и изделия на ее основе | |
| WO2009004282A2 (en) | Medical dressing | |
| Song et al. | Nonwoven materials and technologies for medical applications | |
| KR200359390Y1 (ko) | 의료용 패치 | |
| CA2902387A1 (en) | Chronic wound dressing with variable pore sizes | |
| CN207047481U (zh) | 一种医用耗材制备装置 | |
| RU2197272C1 (ru) | Абсорбирующий медицинский материал и изделия на его основе | |
| Ghanim et al. | Insight into Topical Preparations for Wound Healing: Traditional and Modern Dressings | |
| RU2194534C1 (ru) | Медицинская марля и перевязочные изделия из нее | |
| GORA | ELECTROSPUN ANTIMICROBIAL SCAFFOLDS FOR WOUND MANAGEMENT AND METHODS OF APPLICATIONS | |
| JP2000176000A (ja) | 創傷用包帯 | |
| CN106988013A (zh) | 一种医用耗材制备装置 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20170615 |
|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20180926 |
|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20190827 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20190910 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20191205 |
|
| A02 | Decision of refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A02 Effective date: 20200407 |