JP2012144451A - 殺藻・殺菌剤及び殺藻・殺菌方法 - Google Patents
殺藻・殺菌剤及び殺藻・殺菌方法 Download PDFInfo
- Publication number
- JP2012144451A JP2012144451A JP2011002059A JP2011002059A JP2012144451A JP 2012144451 A JP2012144451 A JP 2012144451A JP 2011002059 A JP2011002059 A JP 2011002059A JP 2011002059 A JP2011002059 A JP 2011002059A JP 2012144451 A JP2012144451 A JP 2012144451A
- Authority
- JP
- Japan
- Prior art keywords
- chloro
- water
- algicidal
- dichloro
- formula
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
- 0 C*C*C(N(C)C)=O Chemical compound C*C*C(N(C)C)=O 0.000 description 2
Classifications
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/34—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one nitrogen atom as the only ring hetero atom
- A01N43/36—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one nitrogen atom as the only ring hetero atom five-membered rings
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N25/00—Biocides, pest repellants or attractants, or plant growth regulators, characterised by their forms, or by their non-active ingredients or by their methods of application, e.g. seed treatment or sequential application; Substances for reducing the noxious effect of the active ingredients to organisms other than pests
- A01N25/02—Biocides, pest repellants or attractants, or plant growth regulators, characterised by their forms, or by their non-active ingredients or by their methods of application, e.g. seed treatment or sequential application; Substances for reducing the noxious effect of the active ingredients to organisms other than pests containing liquids as carriers, diluents or solvents
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/34—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one nitrogen atom as the only ring hetero atom
- A01N43/40—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one nitrogen atom as the only ring hetero atom six-membered rings
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/34—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one nitrogen atom as the only ring hetero atom
- A01N43/46—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one nitrogen atom as the only ring hetero atom rings with more than six members
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/48—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with two nitrogen atoms as the only ring hetero atoms
- A01N43/50—1,3-Diazoles; Hydrogenated 1,3-diazoles
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/48—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with two nitrogen atoms as the only ring hetero atoms
- A01N43/54—1,3-Diazines; Hydrogenated 1,3-diazines
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N59/00—Biocides, pest repellants or attractants, or plant growth regulators containing elements or inorganic compounds
-
- C—CHEMISTRY; METALLURGY
- C02—TREATMENT OF WATER, WASTE WATER, SEWAGE, OR SLUDGE
- C02F—TREATMENT OF WATER, WASTE WATER, SEWAGE, OR SLUDGE
- C02F1/00—Treatment of water, waste water, or sewage
- C02F1/50—Treatment of water, waste water, or sewage by addition or application of a germicide or by oligodynamic treatment
-
- C—CHEMISTRY; METALLURGY
- C02—TREATMENT OF WATER, WASTE WATER, SEWAGE, OR SLUDGE
- C02F—TREATMENT OF WATER, WASTE WATER, SEWAGE, OR SLUDGE
- C02F1/00—Treatment of water, waste water, or sewage
- C02F1/72—Treatment of water, waste water, or sewage by oxidation
- C02F1/76—Treatment of water, waste water, or sewage by oxidation with halogens or compounds of halogens
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02W—CLIMATE CHANGE MITIGATION TECHNOLOGIES RELATED TO WASTEWATER TREATMENT OR WASTE MANAGEMENT
- Y02W10/00—Technologies for wastewater treatment
- Y02W10/30—Wastewater or sewage treatment systems using renewable energies
- Y02W10/37—Wastewater or sewage treatment systems using renewable energies using solar energy
Landscapes
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Health & Medical Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Wood Science & Technology (AREA)
- Plant Pathology (AREA)
- Dentistry (AREA)
- Pest Control & Pesticides (AREA)
- Agronomy & Crop Science (AREA)
- Zoology (AREA)
- Environmental Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Water Supply & Treatment (AREA)
- Environmental & Geological Engineering (AREA)
- Hydrology & Water Resources (AREA)
- Organic Chemistry (AREA)
- Toxicology (AREA)
- Inorganic Chemistry (AREA)
- Agricultural Chemicals And Associated Chemicals (AREA)
Abstract
Description
(1)N−クロロ−2−ピロリジノンを含有することを特徴とする殺藻剤、
(2)水又は水と水溶性有機溶剤の混合溶液を更に含む、水溶性の液体製剤であることを特徴とする(1)記載の殺藻剤、
(3)式(I):
で表される環状のN−クロロ化合物又はN,N’−ジクロロ化合物(但し、N−クロロ−2−ピロリジノンを除く)から選ばれる1種以上を含有することを特徴とする殺藻・殺菌剤、
(4)式(I)で表される環状のN−クロロ化合物又はN,N’−ジクロロ化合物が、N−クロロ−2−ピペリドン、N−クロロ−ε−カプロラクタム、N−クロロ−ω−ヘプタラクタム、N−クロロ−2−イミダゾリジノン、N,N’−ジクロロ−2−イミダゾリジノン、N−クロロ−テトラヒドロ−2−ピリミジノン、及びN,N’−ジクロロ−テトラヒドロ−2−ピリミジノンから選ばれる1種以上であることを特徴とする(3)記載の殺藻・殺菌剤、
(5)水又は水と水溶性有機溶剤の混合溶液を更に含む、水溶性の液体製剤であることを特徴とする(3)又は(4)記載の殺藻・殺菌剤、
(6)腐食が望まれない金属部材と接触している水系中に、式(I):
で表される環状のN−クロロ化合物又はN,N’−ジクロロ化合物から選ばれる1種以上を含有する殺藻・殺菌剤を、残留塩素濃度(Cl2換算)として0.2〜10mg/Lとなるように添加する殺藻・殺菌方法、
(7)式(I)で表される環状のN−クロロ化合物又はN,N’−ジクロロ化合物が、N−クロロ−2−ピロリジノン、N−クロロ−2−ピペリドン、N−クロロ−ε−カプロラクタム、N−クロロ−ω−ヘプタラクタム、N−クロロ−2−イミダゾリジノン、N,N’−ジクロロ−2−イミダゾリジノン、N−クロロ−テトラヒドロ−2−ピリミジノン、及びN,N’−ジクロロ−テトラヒドロ−2−ピリミジノンから選ばれる1種以上であることを特徴とする(6)記載の殺藻・殺菌方法
に関する。
2.28(m、4H)、4.21(t、2H)
IR(KBr、cm−1):
2968、1708、1385、1254、1135、820
1.78(br.s、6H)、2.45(d、2H)、3.21(t、2H)
IR(NaCl、cm−1):
2933、2858、1672、1452、1191、980
1.98(m、4H)、2.98(t、2H)
IR(KBr、cm−1):
2966、2887、1697、1475、1277、1172
3367、2887、1676、1490、1278、1172、724
3230、1736、1715、1440、1380、1214、1054
緑藻類としてChlorella vulgaris C−135(以下、クロレラという)を、藍藻類としてMicrocystis aeruginosa Lemmermann NIES−44(以下、ミクロキスティスという)を用いて、実施例及び比較例の殺藻・殺菌剤の殺藻効果を評価した。クロレラは、MDM培地による前培養液を吸光度O.D.420での値が0.5になるように蒸留水で希釈し、これにHEPES(N−2−ヒドロキシエチルピペラジン−N’−2−エタンスルホン酸)を50mMになるように投入して溶解させ、苛性ソーダにてpHを8.5に調整した。ミクロキスティスは、CB培地による前培養液を吸光度O.D.440での値が0.5になるように蒸留水で希釈し、Bicine(N,N−ビス(2−ヒドロキシエチル)グリシン)を100mMになるように投入して溶解させ、苛性ソーダにてpHを9.0に調整した。これらを試験液とし、各10mlをL字型試験管に分注し、光照射型振盪−恒温水槽に設置した。実施例及び比較例の殺藻・殺菌剤を有効塩素濃度として所定濃度になるようにそれぞれ添加し、30℃、10KLx(明:12h、暗:12h)の条件で振盪培養し、1日おきに目視観察し、3日間観察を行った。殺藻・殺菌剤の殺藻効果は表1に記載の判定基準にて評価し、試験結果を表2に示す。
試験水として、某病院空調用冷却塔の冷却水を用いた。冷却水の水質を表3に示す。
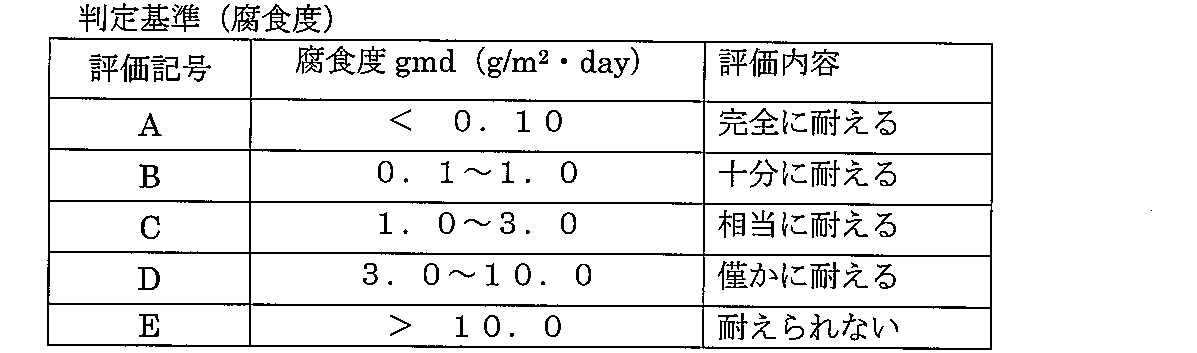
蓋付きガラス製容器中にステンレス鋼SUS304(片面#400研磨仕上、2.5×15×50mm)、鉄(SS−400、2.3×15×30mm)及び銅(C122OP、2.0×20×50mm)の試験片を立て掛け、気相部と溶液浸積部とが形成できる位置にまで上水(静岡県磐田市市水)を所定量計り込み、実施例1及び3、比較例1及び3の殺藻・殺菌剤を有効塩素濃度として2.5mg/L及び5mg/Lとなるように添加し、30℃下で試験片の腐食の程度を観察した。試験液は1週間に一度交換し、8週間後に試験片を取り出し、腐食度(gmd;g/m2・day)を求め下表5のA〜Eの判定基準による評価結果と、外観観察にて薬剤未添加区(上水のみ)での腐食程度に対して各殺藻・殺菌剤の腐食の程度を下表6の判定基準により評価した。結果を組み合わせた記号にて表7に示した。
Claims (7)
- N−クロロ−2−ピロリジノンを含有することを特徴とする殺藻剤。
- 水又は水と水溶性有機溶剤の混合溶液を更に含む、水溶性の液体製剤であることを特徴とする請求項1記載の殺藻剤。
- 式(I)で表される環状のN−クロロ化合物又はN,N’−ジクロロ化合物が、N−クロロ−2−ピペリドン、N−クロロ−ε−カプロラクタム、N−クロロ−ω−ヘプタラクタム、N−クロロ−2−イミダゾリジノン、N,N’−ジクロロ−2−イミダゾリジノン、N−クロロ−テトラヒドロ−2−ピリミジノン、及びN,N’−ジクロロ−テトラヒドロ−2−ピリミジノンから選ばれる1種以上であることを特徴とする請求項3記載の殺藻・殺菌剤。
- 水又は水と水溶性有機溶剤の混合溶液を更に含む、水溶性の液体製剤であることを特徴とする請求項3又は4記載の殺藻・殺菌剤。
- 式(I)で表される環状のN−クロロ化合物又はN,N’−ジクロロ化合物が、N−クロロ−2−ピロリジノン、N−クロロ−2−ピペリドン、N−クロロ−ε−カプロラクタム、N−クロロ−ω−ヘプタラクタム、N−クロロ−2−イミダゾリジノン、N,N’−ジクロロ−2−イミダゾリジノン、N−クロロ−テトラヒドロ−2−ピリミジノン、及びN,N’−ジクロロ−テトラヒドロ−2−ピリミジノンから選ばれる1種以上であることを特徴とする請求項6記載の殺藻・殺菌方法。
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2011002059A JP5643117B2 (ja) | 2011-01-07 | 2011-01-07 | 殺藻・殺菌剤及び殺藻・殺菌方法 |
| KR20137016670A KR20130141624A (ko) | 2011-01-07 | 2011-12-26 | 살조·살균제 및 살조·살균방법 |
| CN201180064265.2A CN103298340B (zh) | 2011-01-07 | 2011-12-26 | 杀藻-杀菌剂及杀藻-杀菌方法 |
| PCT/JP2011/080039 WO2012093608A1 (ja) | 2011-01-07 | 2011-12-26 | 殺藻・殺菌剤及び殺藻・殺菌方法 |
| TW101100718A TW201309198A (zh) | 2011-01-07 | 2012-01-06 | 殺藻殺菌劑及殺藻殺菌方法 |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2011002059A JP5643117B2 (ja) | 2011-01-07 | 2011-01-07 | 殺藻・殺菌剤及び殺藻・殺菌方法 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2012144451A true JP2012144451A (ja) | 2012-08-02 |
| JP5643117B2 JP5643117B2 (ja) | 2014-12-17 |
Family
ID=46457467
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2011002059A Expired - Fee Related JP5643117B2 (ja) | 2011-01-07 | 2011-01-07 | 殺藻・殺菌剤及び殺藻・殺菌方法 |
Country Status (5)
| Country | Link |
|---|---|
| JP (1) | JP5643117B2 (ja) |
| KR (1) | KR20130141624A (ja) |
| CN (1) | CN103298340B (ja) |
| TW (1) | TW201309198A (ja) |
| WO (1) | WO2012093608A1 (ja) |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN104336070A (zh) * | 2014-10-20 | 2015-02-11 | 山东华亚环保科技有限公司 | 一种用于油田输水的杀菌灭藻剂 |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS62230770A (ja) * | 1986-03-31 | 1987-10-09 | ピ−ピ−ジ− インダストリ−ズ インコ−ポレ−テツド | N,n′−ジハロ−2−イミダゾリジノン類 |
Family Cites Families (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB868876A (en) * | 1958-05-05 | 1961-05-25 | Montedison Spa | A method of preparing compounds containing the groupingí¬nclí¬coí¬nclí¬ in a ring |
| US3850920A (en) * | 1967-10-30 | 1974-11-26 | Dow Chemical Co | N-halo-3-morpholinone |
| US3591601A (en) * | 1967-10-30 | 1971-07-06 | Dow Chemical Co | N-chloro or bromo-2-oxazolidinones |
| US3749672A (en) * | 1971-04-19 | 1973-07-31 | Du Pont | Stabilized solutions of n-halo compounds |
| US4492619A (en) * | 1984-02-01 | 1985-01-08 | Phan Xuan T | Photochemical process for preparing alkoxy substituted compounds |
| JPS63101498A (ja) * | 1986-10-17 | 1988-05-06 | ライオン株式会社 | 漂白洗浄剤組成物 |
| US5565109B1 (en) * | 1994-10-14 | 1999-11-23 | Lonza Ag | Hydantoin-enhanced halogen efficacy in pulp and paper applications |
| JP4726329B2 (ja) * | 2001-05-31 | 2011-07-20 | 伯東株式会社 | 微生物コントロール剤 |
| JP3916215B2 (ja) * | 2002-03-11 | 2007-05-16 | ケイ・アイ化成株式会社 | 新規ジブロモニトロ化合物及びその用途 |
-
2011
- 2011-01-07 JP JP2011002059A patent/JP5643117B2/ja not_active Expired - Fee Related
- 2011-12-26 WO PCT/JP2011/080039 patent/WO2012093608A1/ja not_active Ceased
- 2011-12-26 CN CN201180064265.2A patent/CN103298340B/zh not_active Expired - Fee Related
- 2011-12-26 KR KR20137016670A patent/KR20130141624A/ko not_active Withdrawn
-
2012
- 2012-01-06 TW TW101100718A patent/TW201309198A/zh unknown
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS62230770A (ja) * | 1986-03-31 | 1987-10-09 | ピ−ピ−ジ− インダストリ−ズ インコ−ポレ−テツド | N,n′−ジハロ−2−イミダゾリジノン類 |
Also Published As
| Publication number | Publication date |
|---|---|
| CN103298340A (zh) | 2013-09-11 |
| TW201309198A (zh) | 2013-03-01 |
| KR20130141624A (ko) | 2013-12-26 |
| JP5643117B2 (ja) | 2014-12-17 |
| CN103298340B (zh) | 2016-03-30 |
| WO2012093608A1 (ja) | 2012-07-12 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP3072774B2 (ja) | N,n’―ジハロイミダゾリジン―4―オウン類およびn−ハロイミダゾリン−4−オウン類の化合物 | |
| US5126057A (en) | Disinfecting with N,N'-dihaloimidazolidin-4-ones | |
| EP0239896B1 (en) | N,n'-dihalo-2-imidazolidinones | |
| EP2292096B1 (en) | Active bromine containing biocidal compositions and their preparation | |
| KR960013330B1 (ko) | 재순환 물 시스템의 미생물 퇴적 제어 방법 | |
| JP2003146817A (ja) | 殺菌殺藻剤組成物、水系の殺菌殺藻方法及び殺菌殺藻剤組成物の製造方法 | |
| JP5643117B2 (ja) | 殺藻・殺菌剤及び殺藻・殺菌方法 | |
| JP4733851B2 (ja) | アメーバ殺滅剤、アメーバの抑制方法及びレジオネラ属菌の除菌方法 | |
| JP3814085B2 (ja) | 藻類防除剤及び藻類防除方法 | |
| US6881418B2 (en) | Liquid deodorant killing microorganism and method of microorganism-killing deodorization | |
| JP7057091B2 (ja) | 開放循環冷却水系の処理方法 | |
| JP4475920B2 (ja) | 微生物防除剤及び微生物防除方法 | |
| JPH0790639A (ja) | 一液型水処理剤及び水処理方法 | |
| JPS60204704A (ja) | ヒドロキシプロピルメタンチオスルフオネートを含有する相乗的組成物およびその使用法 | |
| US10793586B2 (en) | Quaternary ammonium etidronates | |
| JP4748773B2 (ja) | 変色防止効果能を有する殺菌殺藻剤及び変色防止方法 | |
| JP4297473B2 (ja) | 防菌防藻剤及び防菌防藻方法 | |
| JP4296565B2 (ja) | ヒダントイン含有組成物及びその用途 | |
| US20250059074A1 (en) | Antimicrobial system and method | |
| JP2006232687A (ja) | 循環水系におけるバイオフィルムの形成抑制剤及びバイオフィルムの形成抑制方法 | |
| JP5211274B2 (ja) | トリアジン製剤およびその水性製剤 | |
| JP2003261518A (ja) | 新規ジブロモニトロアルコール誘導体及びその用途 | |
| JP2002161007A (ja) | 殺藻剤 | |
| JP2006206608A (ja) | 殺菌殺藻剤組成物 | |
| JP2014101281A (ja) | 殺藻剤 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20130718 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20140722 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20140919 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20141021 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20141030 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 5643117 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| LAPS | Cancellation because of no payment of annual fees |