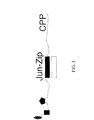
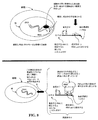
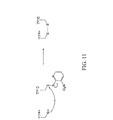
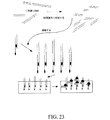
JP2008545394A - 生存細胞中でのリアルタイム核酸分析のための組成物、方法およびキット - Google Patents
生存細胞中でのリアルタイム核酸分析のための組成物、方法およびキット Download PDFInfo
- Publication number
- JP2008545394A JP2008545394A JP2008512475A JP2008512475A JP2008545394A JP 2008545394 A JP2008545394 A JP 2008545394A JP 2008512475 A JP2008512475 A JP 2008512475A JP 2008512475 A JP2008512475 A JP 2008512475A JP 2008545394 A JP2008545394 A JP 2008545394A
- Authority
- JP
- Japan
- Prior art keywords
- molecule
- chimeric
- nucleic acid
- detector
- peptide
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
- 150000007523 nucleic acids Chemical class 0.000 title claims abstract description 327
- 102000039446 nucleic acids Human genes 0.000 title claims abstract description 294
- 108020004707 nucleic acids Proteins 0.000 title claims abstract description 294
- 238000000034 method Methods 0.000 title claims abstract description 239
- 239000000203 mixture Substances 0.000 title abstract description 23
- 238000004458 analytical method Methods 0.000 title description 12
- 238000013518 transcription Methods 0.000 claims abstract description 72
- 230000035897 transcription Effects 0.000 claims abstract description 72
- 238000013519 translation Methods 0.000 claims abstract description 31
- 238000012163 sequencing technique Methods 0.000 claims abstract description 19
- 108090000765 processed proteins & peptides Proteins 0.000 claims description 344
- 102000004163 DNA-directed RNA polymerases Human genes 0.000 claims description 328
- 108091092236 Chimeric RNA Proteins 0.000 claims description 226
- 230000027455 binding Effects 0.000 claims description 218
- 210000004027 cell Anatomy 0.000 claims description 212
- 102000004196 processed proteins & peptides Human genes 0.000 claims description 199
- 108091093037 Peptide nucleic acid Proteins 0.000 claims description 177
- 108091032973 (ribonucleotides)n+m Proteins 0.000 claims description 176
- 229920001184 polypeptide Polymers 0.000 claims description 163
- 210000004708 ribosome subunit Anatomy 0.000 claims description 137
- 108090000626 DNA-directed RNA polymerases Proteins 0.000 claims description 114
- 108020004414 DNA Proteins 0.000 claims description 108
- 230000000295 complement effect Effects 0.000 claims description 106
- 125000003729 nucleotide group Chemical group 0.000 claims description 89
- 239000002773 nucleotide Substances 0.000 claims description 83
- 230000011664 signaling Effects 0.000 claims description 78
- 239000012634 fragment Substances 0.000 claims description 68
- 150000001413 amino acids Chemical group 0.000 claims description 67
- 108010051109 Cell-Penetrating Peptides Proteins 0.000 claims description 63
- 102000020313 Cell-Penetrating Peptides Human genes 0.000 claims description 63
- 239000013598 vector Substances 0.000 claims description 58
- 102000004190 Enzymes Human genes 0.000 claims description 48
- 108090000790 Enzymes Proteins 0.000 claims description 48
- 102000053602 DNA Human genes 0.000 claims description 46
- 108091028043 Nucleic acid sequence Proteins 0.000 claims description 46
- 101710137500 T7 RNA polymerase Proteins 0.000 claims description 45
- 238000002866 fluorescence resonance energy transfer Methods 0.000 claims description 44
- GNBHRKFJIUUOQI-UHFFFAOYSA-N fluorescein Chemical compound O1C(=O)C2=CC=CC=C2C21C1=CC=C(O)C=C1OC1=CC(O)=CC=C21 GNBHRKFJIUUOQI-UHFFFAOYSA-N 0.000 claims description 38
- 108020004999 messenger RNA Proteins 0.000 claims description 38
- 239000002299 complementary DNA Substances 0.000 claims description 37
- ROHFNLRQFUQHCH-YFKPBYRVSA-N L-leucine Chemical compound CC(C)C[C@H](N)C(O)=O ROHFNLRQFUQHCH-YFKPBYRVSA-N 0.000 claims description 36
- ROHFNLRQFUQHCH-UHFFFAOYSA-N Leucine Natural products CC(C)CC(N)C(O)=O ROHFNLRQFUQHCH-UHFFFAOYSA-N 0.000 claims description 36
- WGTODYJZXSJIAG-UHFFFAOYSA-N tetramethylrhodamine chloride Chemical compound [Cl-].C=12C=CC(N(C)C)=CC2=[O+]C2=CC(N(C)C)=CC=C2C=1C1=CC=CC=C1C(O)=O WGTODYJZXSJIAG-UHFFFAOYSA-N 0.000 claims description 35
- FWMNVWWHGCHHJJ-SKKKGAJSSA-N 4-amino-1-[(2r)-6-amino-2-[[(2r)-2-[[(2r)-2-[[(2r)-2-amino-3-phenylpropanoyl]amino]-3-phenylpropanoyl]amino]-4-methylpentanoyl]amino]hexanoyl]piperidine-4-carboxylic acid Chemical compound C([C@H](C(=O)N[C@H](CC(C)C)C(=O)N[C@H](CCCCN)C(=O)N1CCC(N)(CC1)C(O)=O)NC(=O)[C@H](N)CC=1C=CC=CC=1)C1=CC=CC=C1 FWMNVWWHGCHHJJ-SKKKGAJSSA-N 0.000 claims description 32
- 108050008861 SH3 domains Proteins 0.000 claims description 32
- 102000000395 SH3 domains Human genes 0.000 claims description 32
- 235000013930 proline Nutrition 0.000 claims description 28
- 241001465754 Metazoa Species 0.000 claims description 25
- 108010058266 p21-Activated Kinases Proteins 0.000 claims description 23
- 108010076504 Protein Sorting Signals Proteins 0.000 claims description 21
- 238000004422 calculation algorithm Methods 0.000 claims description 21
- 102000009572 RNA Polymerase II Human genes 0.000 claims description 20
- 108010043655 penetratin Proteins 0.000 claims description 20
- 150000003148 prolines Chemical class 0.000 claims description 20
- 108010052853 pVEC peptide Proteins 0.000 claims description 19
- 239000011148 porous material Substances 0.000 claims description 19
- 210000003527 eukaryotic cell Anatomy 0.000 claims description 18
- PYWVYCXTNDRMGF-UHFFFAOYSA-N rhodamine B Chemical compound [Cl-].C=12C=CC(=[N+](CC)CC)C=C2OC2=CC(N(CC)CC)=CC=C2C=1C1=CC=CC=C1C(O)=O PYWVYCXTNDRMGF-UHFFFAOYSA-N 0.000 claims description 18
- 108010062760 transportan Proteins 0.000 claims description 17
- RAHZWNYVWXNFOC-UHFFFAOYSA-N Sulphur dioxide Chemical compound O=S=O RAHZWNYVWXNFOC-UHFFFAOYSA-N 0.000 claims description 16
- 230000008859 change Effects 0.000 claims description 16
- 239000012472 biological sample Substances 0.000 claims description 13
- 241000124008 Mammalia Species 0.000 claims description 12
- 230000001105 regulatory effect Effects 0.000 claims description 11
- ONIBWKKTOPOVIA-UHFFFAOYSA-N Proline Natural products OC(=O)C1CCCN1 ONIBWKKTOPOVIA-UHFFFAOYSA-N 0.000 claims description 8
- 101710175625 Maltose/maltodextrin-binding periplasmic protein Proteins 0.000 claims description 7
- 210000001236 prokaryotic cell Anatomy 0.000 claims description 7
- 108010043121 Green Fluorescent Proteins Proteins 0.000 claims description 6
- 102000004144 Green Fluorescent Proteins Human genes 0.000 claims description 6
- 239000005090 green fluorescent protein Substances 0.000 claims description 6
- 101710154606 Hemagglutinin Proteins 0.000 claims description 5
- 101710093908 Outer capsid protein VP4 Proteins 0.000 claims description 5
- 101710135467 Outer capsid protein sigma-1 Proteins 0.000 claims description 5
- 108091000080 Phosphotransferase Proteins 0.000 claims description 5
- 101710176177 Protein A56 Proteins 0.000 claims description 5
- 239000000185 hemagglutinin Substances 0.000 claims description 5
- 102000020233 phosphotransferase Human genes 0.000 claims description 5
- 241000712461 unidentified influenza virus Species 0.000 claims description 5
- 108010070675 Glutathione transferase Proteins 0.000 claims description 4
- 102000005720 Glutathione transferase Human genes 0.000 claims description 4
- 238000002372 labelling Methods 0.000 claims description 4
- 102000006271 p21-Activated Kinases Human genes 0.000 claims 1
- 238000006243 chemical reaction Methods 0.000 abstract description 27
- 238000011897 real-time detection Methods 0.000 abstract description 3
- 108090000623 proteins and genes Proteins 0.000 description 125
- 102000004169 proteins and genes Human genes 0.000 description 77
- 235000018102 proteins Nutrition 0.000 description 76
- 235000001014 amino acid Nutrition 0.000 description 64
- 229940024606 amino acid Drugs 0.000 description 63
- 230000014509 gene expression Effects 0.000 description 55
- 102000040430 polynucleotide Human genes 0.000 description 37
- 108091033319 polynucleotide Proteins 0.000 description 37
- 239000002157 polynucleotide Substances 0.000 description 37
- 210000001519 tissue Anatomy 0.000 description 36
- 101800001707 Spacer peptide Proteins 0.000 description 35
- 125000003275 alpha amino acid group Chemical group 0.000 description 32
- 238000001514 detection method Methods 0.000 description 27
- 230000014616 translation Effects 0.000 description 27
- 108091034117 Oligonucleotide Proteins 0.000 description 26
- 238000002493 microarray Methods 0.000 description 25
- 239000000523 sample Substances 0.000 description 25
- 230000015572 biosynthetic process Effects 0.000 description 24
- 102100027910 Serine/threonine-protein kinase PAK 1 Human genes 0.000 description 22
- 238000000338 in vitro Methods 0.000 description 20
- 238000001727 in vivo Methods 0.000 description 20
- 238000003786 synthesis reaction Methods 0.000 description 20
- 230000006870 function Effects 0.000 description 19
- 108010077850 Nuclear Localization Signals Proteins 0.000 description 17
- YBJHBAHKTGYVGT-ZKWXMUAHSA-N (+)-Biotin Chemical compound N1C(=O)N[C@@H]2[C@H](CCCCC(=O)O)SC[C@@H]21 YBJHBAHKTGYVGT-ZKWXMUAHSA-N 0.000 description 16
- 229920005989 resin Polymers 0.000 description 16
- 239000011347 resin Substances 0.000 description 16
- 241000282414 Homo sapiens Species 0.000 description 15
- 125000005647 linker group Chemical group 0.000 description 15
- 238000004128 high performance liquid chromatography Methods 0.000 description 14
- 108091033380 Coding strand Proteins 0.000 description 13
- 108060003951 Immunoglobulin Proteins 0.000 description 13
- 102000018358 immunoglobulin Human genes 0.000 description 13
- 108091026890 Coding region Proteins 0.000 description 12
- 108010009460 RNA Polymerase II Proteins 0.000 description 12
- 239000011521 glass Substances 0.000 description 12
- 230000003993 interaction Effects 0.000 description 12
- 239000000047 product Substances 0.000 description 12
- 102000007469 Actins Human genes 0.000 description 11
- 108010085238 Actins Proteins 0.000 description 11
- 238000004519 manufacturing process Methods 0.000 description 11
- 125000006239 protecting group Chemical group 0.000 description 11
- 238000000126 in silico method Methods 0.000 description 10
- CUJMXIQZWPZMNQ-XYYGWQPLSA-N 13,14-dihydro-15-oxo-prostaglandin E2 Chemical compound CCCCCC(=O)CC[C@H]1[C@H](O)CC(=O)[C@@H]1C\C=C/CCCC(O)=O CUJMXIQZWPZMNQ-XYYGWQPLSA-N 0.000 description 9
- 230000000692 anti-sense effect Effects 0.000 description 9
- 210000004899 c-terminal region Anatomy 0.000 description 9
- 238000010195 expression analysis Methods 0.000 description 9
- -1 nucleoside triphosphate Chemical class 0.000 description 9
- 239000013612 plasmid Substances 0.000 description 9
- 125000006850 spacer group Chemical group 0.000 description 9
- 150000007970 thio esters Chemical group 0.000 description 9
- 108700019146 Transgenes Proteins 0.000 description 8
- 241000700605 Viruses Species 0.000 description 8
- JLCPHMBAVCMARE-UHFFFAOYSA-N [3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[5-(2-amino-6-oxo-1H-purin-9-yl)-3-[[3-[[3-[[3-[[3-[[3-[[5-(2-amino-6-oxo-1H-purin-9-yl)-3-[[5-(2-amino-6-oxo-1H-purin-9-yl)-3-hydroxyoxolan-2-yl]methoxy-hydroxyphosphoryl]oxyoxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxyoxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methyl [5-(6-aminopurin-9-yl)-2-(hydroxymethyl)oxolan-3-yl] hydrogen phosphate Polymers Cc1cn(C2CC(OP(O)(=O)OCC3OC(CC3OP(O)(=O)OCC3OC(CC3O)n3cnc4c3nc(N)[nH]c4=O)n3cnc4c3nc(N)[nH]c4=O)C(COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3CO)n3cnc4c(N)ncnc34)n3ccc(N)nc3=O)n3cnc4c(N)ncnc34)n3ccc(N)nc3=O)n3ccc(N)nc3=O)n3ccc(N)nc3=O)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)n3cc(C)c(=O)[nH]c3=O)n3cc(C)c(=O)[nH]c3=O)n3ccc(N)nc3=O)n3cc(C)c(=O)[nH]c3=O)n3cnc4c3nc(N)[nH]c4=O)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)O2)c(=O)[nH]c1=O JLCPHMBAVCMARE-UHFFFAOYSA-N 0.000 description 8
- 229960002685 biotin Drugs 0.000 description 8
- 235000020958 biotin Nutrition 0.000 description 8
- 239000011616 biotin Substances 0.000 description 8
- 230000000694 effects Effects 0.000 description 8
- 239000013604 expression vector Substances 0.000 description 8
- 230000001965 increasing effect Effects 0.000 description 8
- 230000004044 response Effects 0.000 description 8
- 238000012546 transfer Methods 0.000 description 8
- 241001515965 unidentified phage Species 0.000 description 8
- 125000000539 amino acid group Chemical group 0.000 description 7
- 239000000427 antigen Substances 0.000 description 7
- 108091007433 antigens Proteins 0.000 description 7
- 102000036639 antigens Human genes 0.000 description 7
- 150000001875 compounds Chemical class 0.000 description 7
- 235000018417 cysteine Nutrition 0.000 description 7
- 238000009826 distribution Methods 0.000 description 7
- 230000001939 inductive effect Effects 0.000 description 7
- 238000005259 measurement Methods 0.000 description 7
- 238000010369 molecular cloning Methods 0.000 description 7
- 239000012071 phase Substances 0.000 description 7
- 108091008146 restriction endonucleases Proteins 0.000 description 7
- 210000003705 ribosome Anatomy 0.000 description 7
- 238000005070 sampling Methods 0.000 description 7
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 6
- 241000894006 Bacteria Species 0.000 description 6
- ONIBWKKTOPOVIA-BYPYZUCNSA-N L-Proline Chemical compound OC(=O)[C@@H]1CCCN1 ONIBWKKTOPOVIA-BYPYZUCNSA-N 0.000 description 6
- 108020004511 Recombinant DNA Proteins 0.000 description 6
- 108010090804 Streptavidin Proteins 0.000 description 6
- 238000013459 approach Methods 0.000 description 6
- 230000004397 blinking Effects 0.000 description 6
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 6
- 210000000170 cell membrane Anatomy 0.000 description 6
- XUJNEKJLAYXESH-UHFFFAOYSA-N cysteine Natural products SCC(N)C(O)=O XUJNEKJLAYXESH-UHFFFAOYSA-N 0.000 description 6
- 125000000151 cysteine group Chemical class N[C@@H](CS)C(=O)* 0.000 description 6
- 238000009396 hybridization Methods 0.000 description 6
- 239000000463 material Substances 0.000 description 6
- 239000000178 monomer Substances 0.000 description 6
- 210000002569 neuron Anatomy 0.000 description 6
- 238000010647 peptide synthesis reaction Methods 0.000 description 6
- 229920000642 polymer Polymers 0.000 description 6
- 238000000746 purification Methods 0.000 description 6
- 241000894007 species Species 0.000 description 6
- 238000001228 spectrum Methods 0.000 description 6
- 239000000126 substance Substances 0.000 description 6
- 230000008685 targeting Effects 0.000 description 6
- RMVRSNDYEFQCLF-UHFFFAOYSA-N thiophenol Chemical compound SC1=CC=CC=C1 RMVRSNDYEFQCLF-UHFFFAOYSA-N 0.000 description 6
- 230000005945 translocation Effects 0.000 description 6
- 108010047041 Complementarity Determining Regions Proteins 0.000 description 5
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 5
- 241000699666 Mus <mouse, genus> Species 0.000 description 5
- 241000700159 Rattus Species 0.000 description 5
- 101000927794 Rattus norvegicus Rho guanine nucleotide exchange factor 7 Proteins 0.000 description 5
- 238000002474 experimental method Methods 0.000 description 5
- 230000002163 immunogen Effects 0.000 description 5
- 230000000670 limiting effect Effects 0.000 description 5
- 238000001819 mass spectrum Methods 0.000 description 5
- 239000007790 solid phase Substances 0.000 description 5
- 108090001008 Avidin Proteins 0.000 description 4
- 108020004705 Codon Proteins 0.000 description 4
- KRHYYFGTRYWZRS-UHFFFAOYSA-N Fluorane Chemical compound F KRHYYFGTRYWZRS-UHFFFAOYSA-N 0.000 description 4
- 102000016941 Rho Guanine Nucleotide Exchange Factors Human genes 0.000 description 4
- 108010053823 Rho Guanine Nucleotide Exchange Factors Proteins 0.000 description 4
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical class O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 4
- 238000000137 annealing Methods 0.000 description 4
- 230000000890 antigenic effect Effects 0.000 description 4
- 238000003556 assay Methods 0.000 description 4
- 210000003855 cell nucleus Anatomy 0.000 description 4
- 238000004624 confocal microscopy Methods 0.000 description 4
- 239000003814 drug Substances 0.000 description 4
- 238000004520 electroporation Methods 0.000 description 4
- 238000005516 engineering process Methods 0.000 description 4
- 238000011156 evaluation Methods 0.000 description 4
- 230000005284 excitation Effects 0.000 description 4
- RWSXRVCMGQZWBV-WDSKDSINSA-N glutathione Chemical compound OC(=O)[C@@H](N)CCC(=O)N[C@@H](CS)C(=O)NCC(O)=O RWSXRVCMGQZWBV-WDSKDSINSA-N 0.000 description 4
- 230000013595 glycosylation Effects 0.000 description 4
- 238000006206 glycosylation reaction Methods 0.000 description 4
- 238000003384 imaging method Methods 0.000 description 4
- 230000003053 immunization Effects 0.000 description 4
- 238000010348 incorporation Methods 0.000 description 4
- 230000003834 intracellular effect Effects 0.000 description 4
- 238000002955 isolation Methods 0.000 description 4
- 230000033001 locomotion Effects 0.000 description 4
- 238000001254 matrix assisted laser desorption--ionisation time-of-flight mass spectrum Methods 0.000 description 4
- 238000000386 microscopy Methods 0.000 description 4
- 230000004048 modification Effects 0.000 description 4
- 238000012986 modification Methods 0.000 description 4
- 210000004940 nucleus Anatomy 0.000 description 4
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 description 4
- 230000008569 process Effects 0.000 description 4
- 238000012545 processing Methods 0.000 description 4
- 239000000243 solution Substances 0.000 description 4
- 230000009870 specific binding Effects 0.000 description 4
- 125000005931 tert-butyloxycarbonyl group Chemical group [H]C([H])([H])C(OC(*)=O)(C([H])([H])[H])C([H])([H])[H] 0.000 description 4
- 230000001052 transient effect Effects 0.000 description 4
- 230000017105 transposition Effects 0.000 description 4
- DTQVDTLACAAQTR-UHFFFAOYSA-N trifluoroacetic acid Substances OC(=O)C(F)(F)F DTQVDTLACAAQTR-UHFFFAOYSA-N 0.000 description 4
- 239000013638 trimer Substances 0.000 description 4
- 210000004881 tumor cell Anatomy 0.000 description 4
- 125000003088 (fluoren-9-ylmethoxy)carbonyl group Chemical group 0.000 description 3
- WFDIJRYMOXRFFG-UHFFFAOYSA-N Acetic anhydride Chemical compound CC(=O)OC(C)=O WFDIJRYMOXRFFG-UHFFFAOYSA-N 0.000 description 3
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 3
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 3
- 102000017727 Immunoglobulin Variable Region Human genes 0.000 description 3
- KDXKERNSBIXSRK-UHFFFAOYSA-N Lysine Natural products NCCCCC(N)C(O)=O KDXKERNSBIXSRK-UHFFFAOYSA-N 0.000 description 3
- 241000699670 Mus sp. Species 0.000 description 3
- 230000006819 RNA synthesis Effects 0.000 description 3
- 240000004808 Saccharomyces cerevisiae Species 0.000 description 3
- 108020004566 Transfer RNA Proteins 0.000 description 3
- 238000003776 cleavage reaction Methods 0.000 description 3
- 238000010367 cloning Methods 0.000 description 3
- 230000008878 coupling Effects 0.000 description 3
- 238000010168 coupling process Methods 0.000 description 3
- 238000005859 coupling reaction Methods 0.000 description 3
- 238000004132 cross linking Methods 0.000 description 3
- 239000000975 dye Substances 0.000 description 3
- 108020001507 fusion proteins Proteins 0.000 description 3
- 102000037865 fusion proteins Human genes 0.000 description 3
- 239000000499 gel Substances 0.000 description 3
- 230000016784 immunoglobulin production Effects 0.000 description 3
- 238000003780 insertion Methods 0.000 description 3
- 230000037431 insertion Effects 0.000 description 3
- 239000002502 liposome Substances 0.000 description 3
- 229920002521 macromolecule Polymers 0.000 description 3
- 210000004962 mammalian cell Anatomy 0.000 description 3
- SHDMMLFAFLZUEV-UHFFFAOYSA-N n-methyl-1,1-diphenylmethanamine Chemical compound C=1C=CC=CC=1C(NC)C1=CC=CC=C1 SHDMMLFAFLZUEV-UHFFFAOYSA-N 0.000 description 3
- 230000030648 nucleus localization Effects 0.000 description 3
- 239000013600 plasmid vector Substances 0.000 description 3
- 230000007017 scission Effects 0.000 description 3
- 238000010561 standard procedure Methods 0.000 description 3
- 238000012360 testing method Methods 0.000 description 3
- 230000002103 transcriptional effect Effects 0.000 description 3
- 238000001262 western blot Methods 0.000 description 3
- WTKQMHWYSBWUBE-UHFFFAOYSA-N (3-nitropyridin-2-yl) thiohypochlorite Chemical group [O-][N+](=O)C1=CC=CN=C1SCl WTKQMHWYSBWUBE-UHFFFAOYSA-N 0.000 description 2
- 102000040650 (ribonucleotides)n+m Human genes 0.000 description 2
- 229930024421 Adenine Natural products 0.000 description 2
- GFFGJBXGBJISGV-UHFFFAOYSA-N Adenine Chemical compound NC1=NC=NC2=C1N=CN2 GFFGJBXGBJISGV-UHFFFAOYSA-N 0.000 description 2
- 108020004635 Complementary DNA Proteins 0.000 description 2
- 108010014303 DNA-directed DNA polymerase Proteins 0.000 description 2
- 102000016928 DNA-directed DNA polymerase Human genes 0.000 description 2
- 238000002965 ELISA Methods 0.000 description 2
- 241000196324 Embryophyta Species 0.000 description 2
- 108091029865 Exogenous DNA Proteins 0.000 description 2
- 108700028146 Genetic Enhancer Elements Proteins 0.000 description 2
- SXRSQZLOMIGNAQ-UHFFFAOYSA-N Glutaraldehyde Chemical compound O=CCCCC=O SXRSQZLOMIGNAQ-UHFFFAOYSA-N 0.000 description 2
- DHMQDGOQFOQNFH-UHFFFAOYSA-N Glycine Chemical compound NCC(O)=O DHMQDGOQFOQNFH-UHFFFAOYSA-N 0.000 description 2
- NYHBQMYGNKIUIF-UUOKFMHZSA-N Guanosine Chemical compound C1=NC=2C(=O)NC(N)=NC=2N1[C@@H]1O[C@H](CO)[C@@H](O)[C@H]1O NYHBQMYGNKIUIF-UUOKFMHZSA-N 0.000 description 2
- 241000282412 Homo Species 0.000 description 2
- 108010021625 Immunoglobulin Fragments Proteins 0.000 description 2
- 102000008394 Immunoglobulin Fragments Human genes 0.000 description 2
- 108700005091 Immunoglobulin Genes Proteins 0.000 description 2
- 102100034343 Integrase Human genes 0.000 description 2
- 208000009869 Neu-Laxova syndrome Diseases 0.000 description 2
- 108700026244 Open Reading Frames Proteins 0.000 description 2
- 241000283973 Oryctolagus cuniculus Species 0.000 description 2
- NQRYJNQNLNOLGT-UHFFFAOYSA-N Piperidine Chemical compound C1CCNCC1 NQRYJNQNLNOLGT-UHFFFAOYSA-N 0.000 description 2
- 238000002123 RNA extraction Methods 0.000 description 2
- 108010092799 RNA-directed DNA polymerase Proteins 0.000 description 2
- 108700026226 TATA Box Proteins 0.000 description 2
- 208000007536 Thrombosis Diseases 0.000 description 2
- IQFYYKKMVGJFEH-XLPZGREQSA-N Thymidine Chemical compound O=C1NC(=O)C(C)=CN1[C@@H]1O[C@H](CO)[C@@H](O)C1 IQFYYKKMVGJFEH-XLPZGREQSA-N 0.000 description 2
- DRTQHJPVMGBUCF-XVFCMESISA-N Uridine Chemical compound O[C@@H]1[C@H](O)[C@@H](CO)O[C@H]1N1C(=O)NC(=O)C=C1 DRTQHJPVMGBUCF-XVFCMESISA-N 0.000 description 2
- 229960000643 adenine Drugs 0.000 description 2
- OIRDTQYFTABQOQ-KQYNXXCUSA-N adenosine Chemical compound C1=NC=2C(N)=NC=NC=2N1[C@@H]1O[C@H](CO)[C@@H](O)[C@H]1O OIRDTQYFTABQOQ-KQYNXXCUSA-N 0.000 description 2
- 150000001412 amines Chemical class 0.000 description 2
- 230000003321 amplification Effects 0.000 description 2
- 150000008064 anhydrides Chemical class 0.000 description 2
- 238000003491 array Methods 0.000 description 2
- 230000001580 bacterial effect Effects 0.000 description 2
- 230000008901 benefit Effects 0.000 description 2
- 230000004071 biological effect Effects 0.000 description 2
- 230000031018 biological processes and functions Effects 0.000 description 2
- 239000000872 buffer Substances 0.000 description 2
- 229910052799 carbon Inorganic materials 0.000 description 2
- 238000007385 chemical modification Methods 0.000 description 2
- 239000003153 chemical reaction reagent Substances 0.000 description 2
- 239000003795 chemical substances by application Substances 0.000 description 2
- 238000005094 computer simulation Methods 0.000 description 2
- 238000012790 confirmation Methods 0.000 description 2
- 239000000356 contaminant Substances 0.000 description 2
- 239000006059 cover glass Substances 0.000 description 2
- OPTASPLRGRRNAP-UHFFFAOYSA-N cytosine Chemical compound NC=1C=CNC(=O)N=1 OPTASPLRGRRNAP-UHFFFAOYSA-N 0.000 description 2
- 238000012217 deletion Methods 0.000 description 2
- 230000037430 deletion Effects 0.000 description 2
- 238000013461 design Methods 0.000 description 2
- 238000010586 diagram Methods 0.000 description 2
- 125000002228 disulfide group Chemical group 0.000 description 2
- 229940079593 drug Drugs 0.000 description 2
- 230000032050 esterification Effects 0.000 description 2
- 238000005886 esterification reaction Methods 0.000 description 2
- 150000002148 esters Chemical group 0.000 description 2
- OVBPIULPVIDEAO-LBPRGKRZSA-N folic acid Chemical compound C=1N=C2NC(N)=NC(=O)C2=NC=1CNC1=CC=C(C(=O)N[C@@H](CCC(O)=O)C(O)=O)C=C1 OVBPIULPVIDEAO-LBPRGKRZSA-N 0.000 description 2
- 229960003180 glutathione Drugs 0.000 description 2
- UYTPUPDQBNUYGX-UHFFFAOYSA-N guanine Chemical compound O=C1NC(N)=NC2=C1N=CN2 UYTPUPDQBNUYGX-UHFFFAOYSA-N 0.000 description 2
- 239000005556 hormone Substances 0.000 description 2
- 229940088597 hormone Drugs 0.000 description 2
- NPZTUJOABDZTLV-UHFFFAOYSA-N hydroxybenzotriazole Substances O=C1C=CC=C2NNN=C12 NPZTUJOABDZTLV-UHFFFAOYSA-N 0.000 description 2
- 108010023260 immunoglobulin Fv Proteins 0.000 description 2
- 229940072221 immunoglobulins Drugs 0.000 description 2
- 239000012133 immunoprecipitate Substances 0.000 description 2
- 150000002605 large molecules Chemical class 0.000 description 2
- 150000002632 lipids Chemical class 0.000 description 2
- 239000011159 matrix material Substances 0.000 description 2
- 238000001840 matrix-assisted laser desorption--ionisation time-of-flight mass spectrometry Methods 0.000 description 2
- 230000001404 mediated effect Effects 0.000 description 2
- 239000012528 membrane Substances 0.000 description 2
- 239000002184 metal Substances 0.000 description 2
- 229910052751 metal Inorganic materials 0.000 description 2
- 238000003199 nucleic acid amplification method Methods 0.000 description 2
- 210000000056 organ Anatomy 0.000 description 2
- 238000004091 panning Methods 0.000 description 2
- 230000036961 partial effect Effects 0.000 description 2
- 230000001717 pathogenic effect Effects 0.000 description 2
- 238000002823 phage display Methods 0.000 description 2
- 230000010287 polarization Effects 0.000 description 2
- 150000003141 primary amines Chemical class 0.000 description 2
- 239000002096 quantum dot Substances 0.000 description 2
- 230000002285 radioactive effect Effects 0.000 description 2
- 230000003362 replicative effect Effects 0.000 description 2
- 238000011160 research Methods 0.000 description 2
- 238000000926 separation method Methods 0.000 description 2
- 238000002864 sequence alignment Methods 0.000 description 2
- 238000002741 site-directed mutagenesis Methods 0.000 description 2
- 210000000130 stem cell Anatomy 0.000 description 2
- 238000006467 substitution reaction Methods 0.000 description 2
- 150000003457 sulfones Chemical class 0.000 description 2
- 230000002194 synthesizing effect Effects 0.000 description 2
- 229940124597 therapeutic agent Drugs 0.000 description 2
- RWQNBRDOKXIBIV-UHFFFAOYSA-N thymine Chemical compound CC1=CNC(=O)NC1=O RWQNBRDOKXIBIV-UHFFFAOYSA-N 0.000 description 2
- 230000007704 transition Effects 0.000 description 2
- 238000011144 upstream manufacturing Methods 0.000 description 2
- MTCFGRXMJLQNBG-REOHCLBHSA-N (2S)-2-Amino-3-hydroxypropansäure Chemical compound OC[C@H](N)C(O)=O MTCFGRXMJLQNBG-REOHCLBHSA-N 0.000 description 1
- SGKRLCUYIXIAHR-AKNGSSGZSA-N (4s,4ar,5s,5ar,6r,12ar)-4-(dimethylamino)-1,5,10,11,12a-pentahydroxy-6-methyl-3,12-dioxo-4a,5,5a,6-tetrahydro-4h-tetracene-2-carboxamide Chemical compound C1=CC=C2[C@H](C)[C@@H]([C@H](O)[C@@H]3[C@](C(O)=C(C(N)=O)C(=O)[C@H]3N(C)C)(O)C3=O)C3=C(O)C2=C1O SGKRLCUYIXIAHR-AKNGSSGZSA-N 0.000 description 1
- UHDGCWIWMRVCDJ-UHFFFAOYSA-N 1-beta-D-Xylofuranosyl-NH-Cytosine Natural products O=C1N=C(N)C=CN1C1C(O)C(O)C(CO)O1 UHDGCWIWMRVCDJ-UHFFFAOYSA-N 0.000 description 1
- OWEGMIWEEQEYGQ-UHFFFAOYSA-N 100676-05-9 Natural products OC1C(O)C(O)C(CO)OC1OCC1C(O)C(O)C(O)C(OC2C(OC(O)C(O)C2O)CO)O1 OWEGMIWEEQEYGQ-UHFFFAOYSA-N 0.000 description 1
- PIINGYXNCHTJTF-UHFFFAOYSA-N 2-(2-azaniumylethylamino)acetate Chemical group NCCNCC(O)=O PIINGYXNCHTJTF-UHFFFAOYSA-N 0.000 description 1
- WZENSHMKBOPZTM-UHFFFAOYSA-N 2-tritylsulfanylpropanoic acid Chemical compound C=1C=CC=CC=1C(C=1C=CC=CC=1)(SC(C)C(O)=O)C1=CC=CC=C1 WZENSHMKBOPZTM-UHFFFAOYSA-N 0.000 description 1
- JMTMSDXUXJISAY-UHFFFAOYSA-N 2H-benzotriazol-4-ol Chemical compound OC1=CC=CC2=C1N=NN2 JMTMSDXUXJISAY-UHFFFAOYSA-N 0.000 description 1
- 102100038954 60S ribosomal export protein NMD3 Human genes 0.000 description 1
- 229920000936 Agarose Polymers 0.000 description 1
- 108020005098 Anticodon Proteins 0.000 description 1
- 239000004475 Arginine Substances 0.000 description 1
- DCXYFEDJOCDNAF-UHFFFAOYSA-N Asparagine Natural products OC(=O)C(N)CC(N)=O DCXYFEDJOCDNAF-UHFFFAOYSA-N 0.000 description 1
- DWRXFEITVBNRMK-UHFFFAOYSA-N Beta-D-1-Arabinofuranosylthymine Natural products O=C1NC(=O)C(C)=CN1C1C(O)C(O)C(CO)O1 DWRXFEITVBNRMK-UHFFFAOYSA-N 0.000 description 1
- 108091003079 Bovine Serum Albumin Proteins 0.000 description 1
- 239000002126 C01EB10 - Adenosine Substances 0.000 description 1
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 1
- 108091062157 Cis-regulatory element Proteins 0.000 description 1
- 108020004394 Complementary RNA Proteins 0.000 description 1
- MIKUYHXYGGJMLM-GIMIYPNGSA-N Crotonoside Natural products C1=NC2=C(N)NC(=O)N=C2N1[C@H]1O[C@@H](CO)[C@H](O)[C@@H]1O MIKUYHXYGGJMLM-GIMIYPNGSA-N 0.000 description 1
- UHDGCWIWMRVCDJ-PSQAKQOGSA-N Cytidine Natural products O=C1N=C(N)C=CN1[C@@H]1[C@@H](O)[C@@H](O)[C@H](CO)O1 UHDGCWIWMRVCDJ-PSQAKQOGSA-N 0.000 description 1
- 241000701022 Cytomegalovirus Species 0.000 description 1
- 150000008574 D-amino acids Chemical class 0.000 description 1
- NYHBQMYGNKIUIF-UHFFFAOYSA-N D-guanosine Natural products C1=2NC(N)=NC(=O)C=2N=CN1C1OC(CO)C(O)C1O NYHBQMYGNKIUIF-UHFFFAOYSA-N 0.000 description 1
- 241000252212 Danio rerio Species 0.000 description 1
- 241000702421 Dependoparvovirus Species 0.000 description 1
- BWGNESOTFCXPMA-UHFFFAOYSA-N Dihydrogen disulfide Chemical compound SS BWGNESOTFCXPMA-UHFFFAOYSA-N 0.000 description 1
- 102100021238 Dynamin-2 Human genes 0.000 description 1
- 241000588724 Escherichia coli Species 0.000 description 1
- 241000206602 Eukaryota Species 0.000 description 1
- 108700039691 Genetic Promoter Regions Proteins 0.000 description 1
- WHUUTDBJXJRKMK-UHFFFAOYSA-N Glutamic acid Natural products OC(=O)C(N)CCC(O)=O WHUUTDBJXJRKMK-UHFFFAOYSA-N 0.000 description 1
- 108010024636 Glutathione Proteins 0.000 description 1
- 239000004471 Glycine Substances 0.000 description 1
- 101000603190 Homo sapiens 60S ribosomal export protein NMD3 Proteins 0.000 description 1
- 101000817607 Homo sapiens Dynamin-2 Proteins 0.000 description 1
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 1
- 108010054477 Immunoglobulin Fab Fragments Proteins 0.000 description 1
- 102000001706 Immunoglobulin Fab Fragments Human genes 0.000 description 1
- 108010067060 Immunoglobulin Variable Region Proteins 0.000 description 1
- 108091092195 Intron Proteins 0.000 description 1
- QNAYBMKLOCPYGJ-REOHCLBHSA-N L-alanine Chemical compound C[C@H](N)C(O)=O QNAYBMKLOCPYGJ-REOHCLBHSA-N 0.000 description 1
- 150000008575 L-amino acids Chemical class 0.000 description 1
- DCXYFEDJOCDNAF-REOHCLBHSA-N L-asparagine Chemical compound OC(=O)[C@@H](N)CC(N)=O DCXYFEDJOCDNAF-REOHCLBHSA-N 0.000 description 1
- CKLJMWTZIZZHCS-REOHCLBHSA-N L-aspartic acid Chemical compound OC(=O)[C@@H](N)CC(O)=O CKLJMWTZIZZHCS-REOHCLBHSA-N 0.000 description 1
- WHUUTDBJXJRKMK-VKHMYHEASA-N L-glutamic acid Chemical compound OC(=O)[C@@H](N)CCC(O)=O WHUUTDBJXJRKMK-VKHMYHEASA-N 0.000 description 1
- ZDXPYRJPNDTMRX-VKHMYHEASA-N L-glutamine Chemical compound OC(=O)[C@@H](N)CCC(N)=O ZDXPYRJPNDTMRX-VKHMYHEASA-N 0.000 description 1
- AGPKZVBTJJNPAG-WHFBIAKZSA-N L-isoleucine Chemical compound CC[C@H](C)[C@H](N)C(O)=O AGPKZVBTJJNPAG-WHFBIAKZSA-N 0.000 description 1
- COLNVLDHVKWLRT-QMMMGPOBSA-N L-phenylalanine Chemical compound OC(=O)[C@@H](N)CC1=CC=CC=C1 COLNVLDHVKWLRT-QMMMGPOBSA-N 0.000 description 1
- AYFVYJQAPQTCCC-GBXIJSLDSA-N L-threonine Chemical compound C[C@@H](O)[C@H](N)C(O)=O AYFVYJQAPQTCCC-GBXIJSLDSA-N 0.000 description 1
- OUYCCCASQSFEME-QMMMGPOBSA-N L-tyrosine Chemical compound OC(=O)[C@@H](N)CC1=CC=C(O)C=C1 OUYCCCASQSFEME-QMMMGPOBSA-N 0.000 description 1
- KZSNJWFQEVHDMF-BYPYZUCNSA-N L-valine Chemical compound CC(C)[C@H](N)C(O)=O KZSNJWFQEVHDMF-BYPYZUCNSA-N 0.000 description 1
- 101710128836 Large T antigen Proteins 0.000 description 1
- 239000004472 Lysine Substances 0.000 description 1
- GUBGYTABKSRVRQ-PICCSMPSSA-N Maltose Natural products O[C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@@H]1O[C@@H]1[C@@H](CO)OC(O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-PICCSMPSSA-N 0.000 description 1
- BAVYZALUXZFZLV-UHFFFAOYSA-N Methylamine Chemical group NC BAVYZALUXZFZLV-UHFFFAOYSA-N 0.000 description 1
- 241000713333 Mouse mammary tumor virus Species 0.000 description 1
- JGFZNNIVVJXRND-UHFFFAOYSA-N N,N-Diisopropylethylamine (DIPEA) Chemical compound CCN(C(C)C)C(C)C JGFZNNIVVJXRND-UHFFFAOYSA-N 0.000 description 1
- OVBPIULPVIDEAO-UHFFFAOYSA-N N-Pteroyl-L-glutaminsaeure Natural products C=1N=C2NC(N)=NC(=O)C2=NC=1CNC1=CC=C(C(=O)NC(CCC(O)=O)C(O)=O)C=C1 OVBPIULPVIDEAO-UHFFFAOYSA-N 0.000 description 1
- 238000000636 Northern blotting Methods 0.000 description 1
- 102000007999 Nuclear Proteins Human genes 0.000 description 1
- 108010089610 Nuclear Proteins Proteins 0.000 description 1
- 101710163270 Nuclease Proteins 0.000 description 1
- 108091007494 Nucleic acid- binding domains Proteins 0.000 description 1
- 239000004677 Nylon Substances 0.000 description 1
- BZQFBWGGLXLEPQ-UHFFFAOYSA-N O-phosphoryl-L-serine Natural products OC(=O)C(N)COP(O)(O)=O BZQFBWGGLXLEPQ-UHFFFAOYSA-N 0.000 description 1
- 108010038807 Oligopeptides Proteins 0.000 description 1
- 102000015636 Oligopeptides Human genes 0.000 description 1
- 238000012408 PCR amplification Methods 0.000 description 1
- 229910019142 PO4 Inorganic materials 0.000 description 1
- 108010033276 Peptide Fragments Proteins 0.000 description 1
- 102000007079 Peptide Fragments Human genes 0.000 description 1
- 241000255969 Pieris brassicae Species 0.000 description 1
- 108010039918 Polylysine Proteins 0.000 description 1
- 229920000037 Polyproline Polymers 0.000 description 1
- 239000004793 Polystyrene Substances 0.000 description 1
- 238000012952 Resampling Methods 0.000 description 1
- 241000714474 Rous sarcoma virus Species 0.000 description 1
- MTCFGRXMJLQNBG-UHFFFAOYSA-N Serine Natural products OCC(N)C(O)=O MTCFGRXMJLQNBG-UHFFFAOYSA-N 0.000 description 1
- 108020004682 Single-Stranded DNA Proteins 0.000 description 1
- 239000004098 Tetracycline Substances 0.000 description 1
- AYFVYJQAPQTCCC-UHFFFAOYSA-N Threonine Natural products CC(O)C(N)C(O)=O AYFVYJQAPQTCCC-UHFFFAOYSA-N 0.000 description 1
- 239000004473 Threonine Substances 0.000 description 1
- 102000006289 Transcription Factor TFIIA Human genes 0.000 description 1
- 108010083262 Transcription Factor TFIIA Proteins 0.000 description 1
- 102000006290 Transcription Factor TFIID Human genes 0.000 description 1
- 108010083268 Transcription Factor TFIID Proteins 0.000 description 1
- 108091023040 Transcription factor Proteins 0.000 description 1
- 102000040945 Transcription factor Human genes 0.000 description 1
- KZSNJWFQEVHDMF-UHFFFAOYSA-N Valine Natural products CC(C)C(N)C(O)=O KZSNJWFQEVHDMF-UHFFFAOYSA-N 0.000 description 1
- 241000251539 Vertebrata <Metazoa> Species 0.000 description 1
- 241000269370 Xenopus <genus> Species 0.000 description 1
- 125000002777 acetyl group Chemical group [H]C([H])([H])C(*)=O 0.000 description 1
- 230000021736 acetylation Effects 0.000 description 1
- 238000006640 acetylation reaction Methods 0.000 description 1
- 230000009471 action Effects 0.000 description 1
- 230000006978 adaptation Effects 0.000 description 1
- 239000000654 additive Substances 0.000 description 1
- 230000000996 additive effect Effects 0.000 description 1
- 229960005305 adenosine Drugs 0.000 description 1
- 230000004103 aerobic respiration Effects 0.000 description 1
- 239000011543 agarose gel Substances 0.000 description 1
- 230000004931 aggregating effect Effects 0.000 description 1
- 230000002776 aggregation Effects 0.000 description 1
- 238000004220 aggregation Methods 0.000 description 1
- 235000004279 alanine Nutrition 0.000 description 1
- 210000000628 antibody-producing cell Anatomy 0.000 description 1
- 239000011260 aqueous acid Substances 0.000 description 1
- 239000012062 aqueous buffer Substances 0.000 description 1
- 239000007864 aqueous solution Substances 0.000 description 1
- ODKSFYDXXFIFQN-UHFFFAOYSA-N arginine Natural products OC(=O)C(N)CCCNC(N)=N ODKSFYDXXFIFQN-UHFFFAOYSA-N 0.000 description 1
- 235000009582 asparagine Nutrition 0.000 description 1
- 229960001230 asparagine Drugs 0.000 description 1
- 235000003704 aspartic acid Nutrition 0.000 description 1
- 210000001130 astrocyte Anatomy 0.000 description 1
- 210000003719 b-lymphocyte Anatomy 0.000 description 1
- 239000013602 bacteriophage vector Substances 0.000 description 1
- 239000011324 bead Substances 0.000 description 1
- 238000005452 bending Methods 0.000 description 1
- 230000009286 beneficial effect Effects 0.000 description 1
- IQFYYKKMVGJFEH-UHFFFAOYSA-N beta-L-thymidine Natural products O=C1NC(=O)C(C)=CN1C1OC(CO)C(O)C1 IQFYYKKMVGJFEH-UHFFFAOYSA-N 0.000 description 1
- DRTQHJPVMGBUCF-PSQAKQOGSA-N beta-L-uridine Natural products O[C@H]1[C@@H](O)[C@H](CO)O[C@@H]1N1C(=O)NC(=O)C=C1 DRTQHJPVMGBUCF-PSQAKQOGSA-N 0.000 description 1
- OQFSQFPPLPISGP-UHFFFAOYSA-N beta-carboxyaspartic acid Natural products OC(=O)C(N)C(C(O)=O)C(O)=O OQFSQFPPLPISGP-UHFFFAOYSA-N 0.000 description 1
- 230000003851 biochemical process Effects 0.000 description 1
- 238000007622 bioinformatic analysis Methods 0.000 description 1
- 238000003766 bioinformatics method Methods 0.000 description 1
- 230000033228 biological regulation Effects 0.000 description 1
- 229960000074 biopharmaceutical Drugs 0.000 description 1
- 230000000903 blocking effect Effects 0.000 description 1
- 210000004369 blood Anatomy 0.000 description 1
- 239000008280 blood Substances 0.000 description 1
- 210000000601 blood cell Anatomy 0.000 description 1
- 229940098773 bovine serum albumin Drugs 0.000 description 1
- 210000004556 brain Anatomy 0.000 description 1
- 238000010804 cDNA synthesis Methods 0.000 description 1
- 238000004364 calculation method Methods 0.000 description 1
- 239000003560 cancer drug Substances 0.000 description 1
- 150000001718 carbodiimides Chemical class 0.000 description 1
- 230000021523 carboxylation Effects 0.000 description 1
- 238000006473 carboxylation reaction Methods 0.000 description 1
- 210000005056 cell body Anatomy 0.000 description 1
- 238000004113 cell culture Methods 0.000 description 1
- 108091092356 cellular DNA Proteins 0.000 description 1
- 230000001413 cellular effect Effects 0.000 description 1
- 239000001913 cellulose Substances 0.000 description 1
- 229920002678 cellulose Polymers 0.000 description 1
- 210000001175 cerebrospinal fluid Anatomy 0.000 description 1
- 239000002738 chelating agent Substances 0.000 description 1
- 238000010382 chemical cross-linking Methods 0.000 description 1
- 231100000481 chemical toxicant Toxicity 0.000 description 1
- 239000013599 cloning vector Substances 0.000 description 1
- 239000011248 coating agent Substances 0.000 description 1
- 238000000576 coating method Methods 0.000 description 1
- 230000000052 comparative effect Effects 0.000 description 1
- 239000003184 complementary RNA Substances 0.000 description 1
- 239000012141 concentrate Substances 0.000 description 1
- 230000021615 conjugation Effects 0.000 description 1
- 238000010276 construction Methods 0.000 description 1
- 239000002872 contrast media Substances 0.000 description 1
- 150000001944 cysteine derivatives Chemical class 0.000 description 1
- UHDGCWIWMRVCDJ-ZAKLUEHWSA-N cytidine Chemical compound O=C1N=C(N)C=CN1[C@H]1[C@H](O)[C@@H](O)[C@H](CO)O1 UHDGCWIWMRVCDJ-ZAKLUEHWSA-N 0.000 description 1
- 230000009089 cytolysis Effects 0.000 description 1
- 210000000805 cytoplasm Anatomy 0.000 description 1
- 229940104302 cytosine Drugs 0.000 description 1
- 230000006378 damage Effects 0.000 description 1
- 238000007405 data analysis Methods 0.000 description 1
- 230000003247 decreasing effect Effects 0.000 description 1
- 230000022811 deglycosylation Effects 0.000 description 1
- 210000001787 dendrite Anatomy 0.000 description 1
- 230000001419 dependent effect Effects 0.000 description 1
- 238000010511 deprotection reaction Methods 0.000 description 1
- 238000001212 derivatisation Methods 0.000 description 1
- 238000003795 desorption Methods 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- 229950006137 dexfosfoserine Drugs 0.000 description 1
- 239000000539 dimer Substances 0.000 description 1
- 201000010099 disease Diseases 0.000 description 1
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 1
- 229960003722 doxycycline Drugs 0.000 description 1
- 238000005553 drilling Methods 0.000 description 1
- 238000009510 drug design Methods 0.000 description 1
- 239000000428 dust Substances 0.000 description 1
- 239000003623 enhancer Substances 0.000 description 1
- 230000002255 enzymatic effect Effects 0.000 description 1
- 238000001976 enzyme digestion Methods 0.000 description 1
- 210000002919 epithelial cell Anatomy 0.000 description 1
- 230000007717 exclusion Effects 0.000 description 1
- 230000002349 favourable effect Effects 0.000 description 1
- 210000002950 fibroblast Anatomy 0.000 description 1
- 238000001917 fluorescence detection Methods 0.000 description 1
- 238000002875 fluorescence polarization Methods 0.000 description 1
- 239000007850 fluorescent dye Substances 0.000 description 1
- 108020005243 folate receptor Proteins 0.000 description 1
- 102000006815 folate receptor Human genes 0.000 description 1
- 235000019152 folic acid Nutrition 0.000 description 1
- 239000011724 folic acid Substances 0.000 description 1
- 229960000304 folic acid Drugs 0.000 description 1
- 231100000221 frame shift mutation induction Toxicity 0.000 description 1
- 230000037433 frameshift Effects 0.000 description 1
- 125000000524 functional group Chemical group 0.000 description 1
- 230000004927 fusion Effects 0.000 description 1
- 239000010437 gem Substances 0.000 description 1
- 238000012239 gene modification Methods 0.000 description 1
- 230000007274 generation of a signal involved in cell-cell signaling Effects 0.000 description 1
- 230000002068 genetic effect Effects 0.000 description 1
- 230000005017 genetic modification Effects 0.000 description 1
- 235000013617 genetically modified food Nutrition 0.000 description 1
- 239000003862 glucocorticoid Substances 0.000 description 1
- 235000013922 glutamic acid Nutrition 0.000 description 1
- 239000004220 glutamic acid Substances 0.000 description 1
- ZDXPYRJPNDTMRX-UHFFFAOYSA-N glutamine Natural products OC(=O)C(N)CCC(N)=O ZDXPYRJPNDTMRX-UHFFFAOYSA-N 0.000 description 1
- 229960000789 guanidine hydrochloride Drugs 0.000 description 1
- PJJJBBJSCAKJQF-UHFFFAOYSA-N guanidinium chloride Chemical compound [Cl-].NC(N)=[NH2+] PJJJBBJSCAKJQF-UHFFFAOYSA-N 0.000 description 1
- 229940029575 guanosine Drugs 0.000 description 1
- 108060003552 hemocyanin Proteins 0.000 description 1
- 238000004896 high resolution mass spectrometry Methods 0.000 description 1
- 210000004295 hippocampal neuron Anatomy 0.000 description 1
- 238000002744 homologous recombination Methods 0.000 description 1
- 230000006801 homologous recombination Effects 0.000 description 1
- 210000005260 human cell Anatomy 0.000 description 1
- 210000004408 hybridoma Anatomy 0.000 description 1
- 229910052739 hydrogen Inorganic materials 0.000 description 1
- 239000001257 hydrogen Substances 0.000 description 1
- 230000003301 hydrolyzing effect Effects 0.000 description 1
- 229920001600 hydrophobic polymer Polymers 0.000 description 1
- 230000003100 immobilizing effect Effects 0.000 description 1
- 238000010820 immunofluorescence microscopy Methods 0.000 description 1
- 238000011532 immunohistochemical staining Methods 0.000 description 1
- 230000006872 improvement Effects 0.000 description 1
- 238000000099 in vitro assay Methods 0.000 description 1
- 238000005462 in vivo assay Methods 0.000 description 1
- 239000000411 inducer Substances 0.000 description 1
- 238000013383 initial experiment Methods 0.000 description 1
- 230000000977 initiatory effect Effects 0.000 description 1
- 238000002743 insertional mutagenesis Methods 0.000 description 1
- 230000008611 intercellular interaction Effects 0.000 description 1
- 238000005342 ion exchange Methods 0.000 description 1
- 238000004255 ion exchange chromatography Methods 0.000 description 1
- 150000002500 ions Chemical class 0.000 description 1
- 229960000310 isoleucine Drugs 0.000 description 1
- AGPKZVBTJJNPAG-UHFFFAOYSA-N isoleucine Natural products CCC(C)C(N)C(O)=O AGPKZVBTJJNPAG-UHFFFAOYSA-N 0.000 description 1
- 239000003446 ligand Substances 0.000 description 1
- 238000007834 ligase chain reaction Methods 0.000 description 1
- 238000011068 loading method Methods 0.000 description 1
- 210000004698 lymphocyte Anatomy 0.000 description 1
- 210000000415 mammalian chromosome Anatomy 0.000 description 1
- 239000003550 marker Substances 0.000 description 1
- 230000010534 mechanism of action Effects 0.000 description 1
- 230000002503 metabolic effect Effects 0.000 description 1
- 229910021645 metal ion Inorganic materials 0.000 description 1
- 150000002739 metals Chemical class 0.000 description 1
- 238000010208 microarray analysis Methods 0.000 description 1
- 244000000010 microbial pathogen Species 0.000 description 1
- 238000001823 molecular biology technique Methods 0.000 description 1
- 238000012544 monitoring process Methods 0.000 description 1
- 230000000877 morphologic effect Effects 0.000 description 1
- 210000004897 n-terminal region Anatomy 0.000 description 1
- 230000001537 neural effect Effects 0.000 description 1
- 150000002825 nitriles Chemical class 0.000 description 1
- 238000010606 normalization Methods 0.000 description 1
- 238000007899 nucleic acid hybridization Methods 0.000 description 1
- 238000003203 nucleic acid sequencing method Methods 0.000 description 1
- 239000002777 nucleoside Substances 0.000 description 1
- 229920001778 nylon Polymers 0.000 description 1
- 238000002966 oligonucleotide array Methods 0.000 description 1
- 238000002515 oligonucleotide synthesis Methods 0.000 description 1
- 230000003287 optical effect Effects 0.000 description 1
- 230000007170 pathology Effects 0.000 description 1
- 239000013610 patient sample Substances 0.000 description 1
- MCYTYTUNNNZWOK-LCLOTLQISA-N penetratin Chemical group C([C@H](NC(=O)[C@H](CC=1C2=CC=CC=C2NC=1)NC(=O)[C@H]([C@@H](C)CC)NC(=O)[C@H](CCCCN)NC(=O)[C@@H](NC(=O)[C@H](CCC(N)=O)NC(=O)[C@@H](N)CCCNC(N)=N)[C@@H](C)CC)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCCCN)C(N)=O)C1=CC=CC=C1 MCYTYTUNNNZWOK-LCLOTLQISA-N 0.000 description 1
- 230000010363 phase shift Effects 0.000 description 1
- COLNVLDHVKWLRT-UHFFFAOYSA-N phenylalanine Natural products OC(=O)C(N)CC1=CC=CC=C1 COLNVLDHVKWLRT-UHFFFAOYSA-N 0.000 description 1
- NMHMNPHRMNGLLB-UHFFFAOYSA-N phloretic acid Chemical group OC(=O)CCC1=CC=C(O)C=C1 NMHMNPHRMNGLLB-UHFFFAOYSA-N 0.000 description 1
- NBIIXXVUZAFLBC-UHFFFAOYSA-K phosphate Chemical compound [O-]P([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-K 0.000 description 1
- 239000010452 phosphate Substances 0.000 description 1
- 239000008363 phosphate buffer Substances 0.000 description 1
- BZQFBWGGLXLEPQ-REOHCLBHSA-N phosphoserine Chemical compound OC(=O)[C@@H](N)COP(O)(O)=O BZQFBWGGLXLEPQ-REOHCLBHSA-N 0.000 description 1
- USRGIUJOYOXOQJ-GBXIJSLDSA-N phosphothreonine Chemical compound OP(=O)(O)O[C@H](C)[C@H](N)C(O)=O USRGIUJOYOXOQJ-GBXIJSLDSA-N 0.000 description 1
- DCWXELXMIBXGTH-UHFFFAOYSA-N phosphotyrosine Chemical compound OC(=O)C(N)CC1=CC=C(OP(O)(O)=O)C=C1 DCWXELXMIBXGTH-UHFFFAOYSA-N 0.000 description 1
- 230000001766 physiological effect Effects 0.000 description 1
- 239000002504 physiological saline solution Substances 0.000 description 1
- 239000004033 plastic Substances 0.000 description 1
- 229920006122 polyamide resin Polymers 0.000 description 1
- 229920000656 polylysine Polymers 0.000 description 1
- 238000003752 polymerase chain reaction Methods 0.000 description 1
- 238000006116 polymerization reaction Methods 0.000 description 1
- 108010026466 polyproline Proteins 0.000 description 1
- 229920001296 polysiloxane Polymers 0.000 description 1
- 229920005990 polystyrene resin Polymers 0.000 description 1
- 239000002243 precursor Substances 0.000 description 1
- 125000001500 prolyl group Chemical group [H]N1C([H])(C(=O)[*])C([H])([H])C([H])([H])C1([H])[H] 0.000 description 1
- 230000006916 protein interaction Effects 0.000 description 1
- 230000004850 protein–protein interaction Effects 0.000 description 1
- 230000017854 proteolysis Effects 0.000 description 1
- 238000011002 quantification Methods 0.000 description 1
- 238000010791 quenching Methods 0.000 description 1
- 230000000171 quenching effect Effects 0.000 description 1
- 238000010223 real-time analysis Methods 0.000 description 1
- 108020003175 receptors Proteins 0.000 description 1
- 102000005962 receptors Human genes 0.000 description 1
- 230000006798 recombination Effects 0.000 description 1
- 238000005215 recombination Methods 0.000 description 1
- 230000002829 reductive effect Effects 0.000 description 1
- 230000022532 regulation of transcription, DNA-dependent Effects 0.000 description 1
- 230000003252 repetitive effect Effects 0.000 description 1
- 230000010076 replication Effects 0.000 description 1
- 238000010839 reverse transcription Methods 0.000 description 1
- 238000004007 reversed phase HPLC Methods 0.000 description 1
- 238000012552 review Methods 0.000 description 1
- 235000002020 sage Nutrition 0.000 description 1
- 230000019491 signal transduction Effects 0.000 description 1
- 239000000377 silicon dioxide Substances 0.000 description 1
- 238000004557 single molecule detection Methods 0.000 description 1
- 229910052708 sodium Inorganic materials 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- 239000002904 solvent Substances 0.000 description 1
- 238000011895 specific detection Methods 0.000 description 1
- 230000002269 spontaneous effect Effects 0.000 description 1
- 230000003068 static effect Effects 0.000 description 1
- 238000010972 statistical evaluation Methods 0.000 description 1
- 238000012109 statistical procedure Methods 0.000 description 1
- 230000000638 stimulation Effects 0.000 description 1
- 238000007920 subcutaneous administration Methods 0.000 description 1
- 229940037128 systemic glucocorticoids Drugs 0.000 description 1
- 238000012731 temporal analysis Methods 0.000 description 1
- 229960002180 tetracycline Drugs 0.000 description 1
- 229930101283 tetracycline Natural products 0.000 description 1
- 235000019364 tetracycline Nutrition 0.000 description 1
- 150000003522 tetracyclines Chemical class 0.000 description 1
- 230000001225 therapeutic effect Effects 0.000 description 1
- 229940104230 thymidine Drugs 0.000 description 1
- 229940113082 thymine Drugs 0.000 description 1
- 238000000700 time series analysis Methods 0.000 description 1
- 239000003440 toxic substance Substances 0.000 description 1
- 231100000419 toxicity Toxicity 0.000 description 1
- 230000001988 toxicity Effects 0.000 description 1
- 239000003053 toxin Substances 0.000 description 1
- 231100000765 toxin Toxicity 0.000 description 1
- 108700012359 toxins Proteins 0.000 description 1
- 238000012549 training Methods 0.000 description 1
- 230000001131 transforming effect Effects 0.000 description 1
- 239000011043 treated quartz Substances 0.000 description 1
- 239000001226 triphosphate Substances 0.000 description 1
- 235000011178 triphosphate Nutrition 0.000 description 1
- 125000002221 trityl group Chemical group [H]C1=C([H])C([H])=C([H])C([H])=C1C([*])(C1=C(C(=C(C(=C1[H])[H])[H])[H])[H])C1=C([H])C([H])=C([H])C([H])=C1[H] 0.000 description 1
- OUYCCCASQSFEME-UHFFFAOYSA-N tyrosine Natural products OC(=O)C(N)CC1=CC=C(O)C=C1 OUYCCCASQSFEME-UHFFFAOYSA-N 0.000 description 1
- 241000701161 unidentified adenovirus Species 0.000 description 1
- 241001430294 unidentified retrovirus Species 0.000 description 1
- DRTQHJPVMGBUCF-UHFFFAOYSA-N uracil arabinoside Natural products OC1C(O)C(CO)OC1N1C(=O)NC(=O)C=C1 DRTQHJPVMGBUCF-UHFFFAOYSA-N 0.000 description 1
- 229940045145 uridine Drugs 0.000 description 1
- 239000004474 valine Substances 0.000 description 1
- 230000003612 virological effect Effects 0.000 description 1
- 238000012800 visualization Methods 0.000 description 1
- 238000005406 washing Methods 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N9/00—Enzymes; Proenzymes; Compositions thereof; Processes for preparing, activating, inhibiting, separating or purifying enzymes
- C12N9/96—Stabilising an enzyme by forming an adduct or a composition; Forming enzyme conjugates
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N9/00—Enzymes; Proenzymes; Compositions thereof; Processes for preparing, activating, inhibiting, separating or purifying enzymes
- C12N9/10—Transferases (2.)
- C12N9/12—Transferases (2.) transferring phosphorus containing groups, e.g. kinases (2.7)
- C12N9/1241—Nucleotidyltransferases (2.7.7)
- C12N9/1247—DNA-directed RNA polymerase (2.7.7.6)
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N9/00—Enzymes; Proenzymes; Compositions thereof; Processes for preparing, activating, inhibiting, separating or purifying enzymes
- C12N9/10—Transferases (2.)
- C12N9/12—Transferases (2.) transferring phosphorus containing groups, e.g. kinases (2.7)
- C12N9/1241—Nucleotidyltransferases (2.7.7)
- C12N9/127—RNA-directed RNA polymerase (2.7.7.48), i.e. RNA replicase
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Q—MEASURING OR TESTING PROCESSES INVOLVING ENZYMES, NUCLEIC ACIDS OR MICROORGANISMS; COMPOSITIONS OR TEST PAPERS THEREFOR; PROCESSES OF PREPARING SUCH COMPOSITIONS; CONDITION-RESPONSIVE CONTROL IN MICROBIOLOGICAL OR ENZYMOLOGICAL PROCESSES
- C12Q1/00—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions
- C12Q1/68—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions involving nucleic acids
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2319/00—Fusion polypeptide
- C07K2319/70—Fusion polypeptide containing domain for protein-protein interaction
- C07K2319/72—Fusion polypeptide containing domain for protein-protein interaction containing SH2 domain
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2319/00—Fusion polypeptide
- C07K2319/70—Fusion polypeptide containing domain for protein-protein interaction
- C07K2319/73—Fusion polypeptide containing domain for protein-protein interaction containing coiled-coiled motif (leucine zippers)
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2319/00—Fusion polypeptide
- C07K2319/85—Fusion polypeptide containing an RNA binding domain
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Y—ENZYMES
- C12Y207/00—Transferases transferring phosphorus-containing groups (2.7)
- C12Y207/07—Nucleotidyltransferases (2.7.7)
- C12Y207/07006—DNA-directed RNA polymerase (2.7.7.6)
Landscapes
- Life Sciences & Earth Sciences (AREA)
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Wood Science & Technology (AREA)
- Zoology (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Genetics & Genomics (AREA)
- Molecular Biology (AREA)
- General Health & Medical Sciences (AREA)
- Microbiology (AREA)
- Biotechnology (AREA)
- Biochemistry (AREA)
- General Engineering & Computer Science (AREA)
- Medicinal Chemistry (AREA)
- Biomedical Technology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Physics & Mathematics (AREA)
- Analytical Chemistry (AREA)
- Biophysics (AREA)
- Immunology (AREA)
- Measuring Or Testing Involving Enzymes Or Micro-Organisms (AREA)
- Investigating Or Analysing Materials By The Use Of Chemical Reactions (AREA)
- Micro-Organisms Or Cultivation Processes Thereof (AREA)
- Peptides Or Proteins (AREA)
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US68233405P | 2005-05-18 | 2005-05-18 | |
| PCT/US2006/019107 WO2006125012A2 (en) | 2005-05-18 | 2006-05-17 | Compositions, methods and kits for real-time nucleic acid analysis in live cells |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2008545394A true JP2008545394A (ja) | 2008-12-18 |
| JP2008545394A5 JP2008545394A5 (enExample) | 2009-07-02 |
Family
ID=37432106
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2008512475A Pending JP2008545394A (ja) | 2005-05-18 | 2006-05-17 | 生存細胞中でのリアルタイム核酸分析のための組成物、方法およびキット |
Country Status (6)
| Country | Link |
|---|---|
| US (2) | US9260703B2 (enExample) |
| EP (1) | EP1882047A4 (enExample) |
| JP (1) | JP2008545394A (enExample) |
| AU (1) | AU2006247215B2 (enExample) |
| CA (1) | CA2609150A1 (enExample) |
| WO (1) | WO2006125012A2 (enExample) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2023510916A (ja) * | 2020-01-17 | 2023-03-15 | サナ バイオテクノロジー,インコーポレイテッド | 遺伝子発現の制御のための安全スイッチ |
Families Citing this family (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB0804495D0 (en) * | 2008-03-11 | 2008-04-16 | Iti Scotland Ltd | Surface attachment |
| AU2010245304B2 (en) | 2009-04-27 | 2015-06-04 | Pacific Biosciences Of California, Inc. | Real-time sequencing methods and systems |
| US9063156B2 (en) | 2009-06-12 | 2015-06-23 | Pacific Biosciences Of California, Inc. | Real-time analytical methods and systems |
| JP2020530267A (ja) | 2017-06-30 | 2020-10-22 | コデクシス, インコーポレイテッド | T7 rnaポリメラーゼバリアント |
| US10793841B2 (en) | 2017-06-30 | 2020-10-06 | Codexis, Inc. | T7 RNA polymerase variants |
| CN116802267A (zh) * | 2020-10-23 | 2023-09-22 | 斯宾德尔生物科技公司 | 用于rna合成的组合物和方法 |
| WO2025038950A1 (en) * | 2023-08-16 | 2025-02-20 | Spotlight Therapeutics | Cell-penetrating peptide compositions and uses thereof |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2003061684A (ja) * | 2001-06-11 | 2003-03-04 | Nippon Genetech Co Ltd | Dnaの塩基配列決定法 |
| WO2004007692A2 (en) * | 2002-07-17 | 2004-01-22 | U.S.Genomics, Inc. | Methods and compositions for analyzing polymers using chimeric tags |
| JP2004532849A (ja) * | 2001-04-20 | 2004-10-28 | カイロン コーポレイション | 中枢神経系へのポリヌクレオチド薬剤の送達 |
Family Cites Families (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0714980A1 (en) * | 1994-12-02 | 1996-06-05 | Institut Pasteur | Hypermutagenesis |
| US6322901B1 (en) | 1997-11-13 | 2001-11-27 | Massachusetts Institute Of Technology | Highly luminescent color-selective nano-crystalline materials |
| US5990479A (en) | 1997-11-25 | 1999-11-23 | Regents Of The University Of California | Organo Luminescent semiconductor nanocrystal probes for biological applications and process for making and using such probes |
| US6426513B1 (en) | 1998-09-18 | 2002-07-30 | Massachusetts Institute Of Technology | Water-soluble thiol-capped nanocrystals |
| US6251303B1 (en) | 1998-09-18 | 2001-06-26 | Massachusetts Institute Of Technology | Water-soluble fluorescent nanocrystals |
| JP5047437B2 (ja) * | 2000-02-08 | 2012-10-10 | サンガモ バイオサイエンシーズ, インコーポレイテッド | 薬物の発見のための細胞 |
| US20020025317A1 (en) * | 2000-07-20 | 2002-02-28 | Schering Ag | Bispecific monoclonal antibodies to IL-12 and IL-18 |
| US6576291B2 (en) | 2000-12-08 | 2003-06-10 | Massachusetts Institute Of Technology | Preparation of nanocrystallites |
| JP2005504275A (ja) * | 2001-09-18 | 2005-02-10 | ユー.エス. ジェノミクス, インコーポレイテッド | 高分解能線形解析用のポリマーの差示的タグ付け |
| US7343249B2 (en) * | 2005-03-23 | 2008-03-11 | International Business Machines Corporational | Vectorization of sequence alignment computation using distance matrix reshaping |
-
2006
- 2006-05-17 WO PCT/US2006/019107 patent/WO2006125012A2/en not_active Ceased
- 2006-05-17 AU AU2006247215A patent/AU2006247215B2/en active Active
- 2006-05-17 CA CA002609150A patent/CA2609150A1/en not_active Abandoned
- 2006-05-17 US US11/920,618 patent/US9260703B2/en active Active
- 2006-05-17 EP EP06770507A patent/EP1882047A4/en not_active Withdrawn
- 2006-05-17 JP JP2008512475A patent/JP2008545394A/ja active Pending
-
2015
- 2015-12-31 US US14/985,826 patent/US10093917B2/en active Active
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2004532849A (ja) * | 2001-04-20 | 2004-10-28 | カイロン コーポレイション | 中枢神経系へのポリヌクレオチド薬剤の送達 |
| JP2003061684A (ja) * | 2001-06-11 | 2003-03-04 | Nippon Genetech Co Ltd | Dnaの塩基配列決定法 |
| WO2004007692A2 (en) * | 2002-07-17 | 2004-01-22 | U.S.Genomics, Inc. | Methods and compositions for analyzing polymers using chimeric tags |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2023510916A (ja) * | 2020-01-17 | 2023-03-15 | サナ バイオテクノロジー,インコーポレイテッド | 遺伝子発現の制御のための安全スイッチ |
Also Published As
| Publication number | Publication date |
|---|---|
| US9260703B2 (en) | 2016-02-16 |
| CA2609150A1 (en) | 2006-11-23 |
| EP1882047A2 (en) | 2008-01-30 |
| US20160168558A1 (en) | 2016-06-16 |
| EP1882047A4 (en) | 2009-10-28 |
| WO2006125012A3 (en) | 2007-05-03 |
| AU2006247215A1 (en) | 2006-11-23 |
| US10093917B2 (en) | 2018-10-09 |
| US20100041025A1 (en) | 2010-02-18 |
| AU2006247215B2 (en) | 2012-09-13 |
| WO2006125012A2 (en) | 2006-11-23 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US10093917B2 (en) | Compositions, methods and kits for real-time nucleic acid analysis in live cells | |
| RU2360923C2 (ru) | Новый лиганд рецептора цитокина zcytor17 | |
| JP2011516026A (ja) | 免疫関連疾患の治療のための組成物と方法 | |
| JP2002513588A (ja) | 所望の生物学的特性を与えるクローンを発現ライブラリーから同定する新規方法 | |
| JP2005538696A (ja) | Ptlvエンベロープタンパク質の表面成分をコードする配列を起源とするオリゴヌクレオチド及びそれらの使用 | |
| CN1643148B (zh) | 小鼠精子形成基因、人男性不育关连基因、以及利用该基因的诊断系统 | |
| CN109563516B (zh) | Gpr156变体及其用途 | |
| JP4414138B2 (ja) | 蛍光染料を通した分子のエピトープの免疫検出及び分子の相互作用の検出のための方法 | |
| US20030032018A1 (en) | Human smooth muscle myosin heavy chain | |
| US7094890B1 (en) | Arthritis-associated protein | |
| CN111315765A (zh) | Cornulin(CRNN)变体及其用途 | |
| JP5587528B2 (ja) | c−Jun蛋白質と複合体を形成する蛋白質、及び、それをコードする核酸、ならびに、それらの利用方法 | |
| US7670787B2 (en) | Protein forming complex with c-Fos protein, nucleic acid encoding the same and method of using the same | |
| JP7237064B2 (ja) | 単一免疫グロブリンインターロイキン-1受容体関連(sigirr)変異型及びその使用 | |
| RU2815068C2 (ru) | Варианты белка, родственного рецептору интерлейкина-1 и содержащего одиночный домен иммуноглобулина (sigirr), и их применение | |
| US20020155473A1 (en) | Methods for identifying G-protein coupled receptors associated with diseases | |
| US20040234537A1 (en) | Carcinoma-related peptides | |
| WO1997001634A2 (en) | Polypeptide for repairing genetic information, nucleotidic sequence which codes for it and process for the preparation thereof (guanine thymine binding protein - gtbp) | |
| JP2003525636A (ja) | ヒトの細胞外シグナル制御キナーゼ | |
| US20050048502A1 (en) | Human SACH-related gene variants associated with cancers | |
| JP2001503994A (ja) | チロシンリン酸化切断溝関連タンパク質(pstpip) | |
| JP2003523758A (ja) | 新規転写因子carp−2 | |
| Ravi | Kinetic characterization of GreB binding to RNA polymerase and transcription elongation complexes | |
| WO2003004532A2 (en) | Q9ul33/q9nzz4 and q9h5p3 splice variants | |
| US20030207276A1 (en) | Human G protein beta subunit 1-related gene variant associated with lung cancers |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20090518 |
|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20090518 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20111122 |
|
| A02 | Decision of refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A02 Effective date: 20120417 |