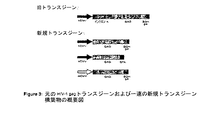
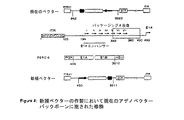
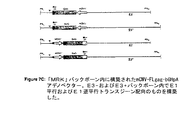
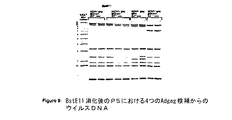
JP2004508064A - コドン最適化hiv1−gag、pol、nefおよび修飾体を発現する増強された第1世代アデノウイルスワクチン - Google Patents
コドン最適化hiv1−gag、pol、nefおよび修飾体を発現する増強された第1世代アデノウイルスワクチン Download PDFInfo
- Publication number
- JP2004508064A JP2004508064A JP2002526335A JP2002526335A JP2004508064A JP 2004508064 A JP2004508064 A JP 2004508064A JP 2002526335 A JP2002526335 A JP 2002526335A JP 2002526335 A JP2002526335 A JP 2002526335A JP 2004508064 A JP2004508064 A JP 2004508064A
- Authority
- JP
- Japan
- Prior art keywords
- adenovirus
- vector
- hiv
- nef
- seq
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Withdrawn
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N7/00—Viruses; Bacteriophages; Compositions thereof; Preparation or purification thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/12—Antivirals
- A61P31/14—Antivirals for RNA viruses
- A61P31/18—Antivirals for RNA viruses for HIV
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/02—Immunomodulators
- A61P37/04—Immunostimulants
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/005—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from viruses
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N15/00—Mutation or genetic engineering; DNA or RNA concerning genetic engineering, vectors, e.g. plasmids, or their isolation, preparation or purification; Use of hosts therefor
- C12N15/09—Recombinant DNA-technology
- C12N15/63—Introduction of foreign genetic material using vectors; Vectors; Use of hosts therefor; Regulation of expression
- C12N15/79—Vectors or expression systems specially adapted for eukaryotic hosts
- C12N15/85—Vectors or expression systems specially adapted for eukaryotic hosts for animal cells
- C12N15/86—Viral vectors
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/51—Medicinal preparations containing antigens or antibodies comprising whole cells, viruses or DNA/RNA
- A61K2039/525—Virus
- A61K2039/5256—Virus expressing foreign proteins
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/51—Medicinal preparations containing antigens or antibodies comprising whole cells, viruses or DNA/RNA
- A61K2039/53—DNA (RNA) vaccination
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/545—Medicinal preparations containing antigens or antibodies characterised by the dose, timing or administration schedule
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/57—Medicinal preparations containing antigens or antibodies characterised by the type of response, e.g. Th1, Th2
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2710/00—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA dsDNA viruses
- C12N2710/00011—Details
- C12N2710/10011—Adenoviridae
- C12N2710/10311—Mastadenovirus, e.g. human or simian adenoviruses
- C12N2710/10341—Use of virus, viral particle or viral elements as a vector
- C12N2710/10343—Use of virus, viral particle or viral elements as a vector viral genome or elements thereof as genetic vector
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2710/00—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA dsDNA viruses
- C12N2710/00011—Details
- C12N2710/10011—Adenoviridae
- C12N2710/10311—Mastadenovirus, e.g. human or simian adenoviruses
- C12N2710/10351—Methods of production or purification of viral material
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2740/00—Reverse transcribing RNA viruses
- C12N2740/00011—Details
- C12N2740/10011—Retroviridae
- C12N2740/16011—Human Immunodeficiency Virus, HIV
- C12N2740/16111—Human Immunodeficiency Virus, HIV concerning HIV env
- C12N2740/16134—Use of virus or viral component as vaccine, e.g. live-attenuated or inactivated virus, VLP, viral protein
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2740/00—Reverse transcribing RNA viruses
- C12N2740/00011—Details
- C12N2740/10011—Retroviridae
- C12N2740/16011—Human Immunodeficiency Virus, HIV
- C12N2740/16311—Human Immunodeficiency Virus, HIV concerning HIV regulatory proteins
- C12N2740/16334—Use of virus or viral component as vaccine, e.g. live-attenuated or inactivated virus, VLP, viral protein
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2830/00—Vector systems having a special element relevant for transcription
- C12N2830/42—Vector systems having a special element relevant for transcription being an intron or intervening sequence for splicing and/or stability of RNA
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Genetics & Genomics (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Virology (AREA)
- General Health & Medical Sciences (AREA)
- Wood Science & Technology (AREA)
- Medicinal Chemistry (AREA)
- Zoology (AREA)
- Immunology (AREA)
- Biotechnology (AREA)
- Biomedical Technology (AREA)
- Biochemistry (AREA)
- Molecular Biology (AREA)
- General Engineering & Computer Science (AREA)
- Pharmacology & Pharmacy (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Biophysics (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Microbiology (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- General Chemical & Material Sciences (AREA)
- Plant Pathology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- AIDS & HIV (AREA)
- Oncology (AREA)
- Physics & Mathematics (AREA)
- Tropical Medicine & Parasitology (AREA)
- Communicable Diseases (AREA)
- Gastroenterology & Hepatology (AREA)
- Medicines Containing Antibodies Or Antigens For Use As Internal Diagnostic Agents (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Medicines Containing Material From Animals Or Micro-Organisms (AREA)
- Micro-Organisms Or Cultivation Processes Thereof (AREA)
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US23318000P | 2000-09-15 | 2000-09-15 | |
| PCT/US2001/028861 WO2002022080A2 (en) | 2000-09-15 | 2001-09-14 | Enhanced first generation adenovirus vaccines expressing codon optimized hiv1-gag, pol, nef and modifications |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2004508064A true JP2004508064A (ja) | 2004-03-18 |
| JP2004508064A5 JP2004508064A5 (OSRAM) | 2005-08-04 |
Family
ID=22876220
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2002526335A Withdrawn JP2004508064A (ja) | 2000-09-15 | 2001-09-14 | コドン最適化hiv1−gag、pol、nefおよび修飾体を発現する増強された第1世代アデノウイルスワクチン |
Country Status (5)
| Country | Link |
|---|---|
| EP (1) | EP1320621A4 (OSRAM) |
| JP (1) | JP2004508064A (OSRAM) |
| AU (2) | AU9456201A (OSRAM) |
| CA (1) | CA2422882A1 (OSRAM) |
| WO (1) | WO2002022080A2 (OSRAM) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2007532656A (ja) * | 2004-04-12 | 2007-11-15 | アメリカ合衆国 | アデノウイルスベクターを用いて免疫応答を誘導するための方法 |
| JP2008539746A (ja) * | 2005-05-12 | 2008-11-20 | グラクソ グループ リミテッド | ワクチン組成物 |
| JP2018035177A (ja) * | 2008-11-18 | 2018-03-08 | ベス イスラエル デアコネス メディカル センター インコーポレイテッド | 細胞性免疫原性が向上した抗ウイルスワクチン |
Families Citing this family (42)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2003516741A (ja) * | 1999-12-17 | 2003-05-20 | メルク エンド カムパニー インコーポレーテッド | コドン最適化HIV−1Nef及び修飾HIV−1Nefを発現するポリヌクレオチドワクチン |
| CA2461380C (en) | 2001-10-11 | 2011-03-22 | Merck & Co., Inc. | Hepatitis c virus vaccine |
| US7479547B2 (en) | 2001-10-31 | 2009-01-20 | The South African Medical Research Council | HIV-1 subtype isolate regulatory/accessory genes, and modifications and derivatives thereof |
| CA2478651A1 (en) * | 2002-03-13 | 2003-09-25 | Merck & Co., Inc. | Method of inducing an enhanced immune response against hiv |
| ATE386824T1 (de) | 2002-03-29 | 2008-03-15 | Merck & Co Inc | Verfahren zur virusproduktion |
| ES2285120T3 (es) | 2002-05-14 | 2007-11-16 | MERCK & CO., INC. | Procedimientos de purificacion de adenovirus. |
| CN1490056A (zh) * | 2002-10-18 | 2004-04-21 | ��¡���ɵ°��̲��о����� | 针对hiv-1的免疫方法和组合物 |
| US20080153083A1 (en) | 2003-10-23 | 2008-06-26 | Crucell Holland B.V. | Settings for recombinant adenoviral-based vaccines |
| WO2004037294A2 (en) | 2002-10-23 | 2004-05-06 | Crucell Holland B.V. | New settings for recombinant adenoviral-based vaccines |
| AU2003298361B2 (en) | 2002-12-17 | 2009-05-14 | Crucell Holland B.V. | Recombinant viral-based malaria vaccines |
| AU2003294023B2 (en) | 2003-01-03 | 2008-01-31 | Istituto Di Ricerche Di Biologia Molecolare P. Angeletti Spa | Rhesus HER2/neu, nucleotides encoding same, and uses thereof |
| CN101173282B (zh) | 2003-07-21 | 2011-04-06 | P.安杰莱蒂分子生物学研究所 | 编码人表皮生长因子2/neu抗原的合成基因及其用途 |
| GB2406336A (en) * | 2003-09-24 | 2005-03-30 | Oxxon Pharmaccines Ltd | HIV Pharmaccines |
| BRPI0507579A (pt) | 2004-02-11 | 2007-07-03 | Angeletti P Ist Richerche Bio | molécula de ácido nucleico, vetor, célula hospedeira, processo para expressar uma proteìna de fusão cea em uma célula hospedeira recombinante, proteìna de fusão cea purificada, método de prevenção ou tratamento de cáncer, plasmìdeo de vacina, e, método de tratamento de um mamìfero sofrendo de ou predisposto a um cáncer associados com cea |
| CN1922308A (zh) | 2004-02-23 | 2007-02-28 | 克鲁塞尔荷兰公司 | 病毒纯化方法 |
| GB0417494D0 (en) | 2004-08-05 | 2004-09-08 | Glaxosmithkline Biolog Sa | Vaccine |
| NZ553889A (en) | 2004-10-13 | 2009-11-27 | Crucell Holland Bv | Replication-defective adenovirus comprising a chimeric hexon protein and replaced hypervariable ragions HVR1-7 |
| ES2317517T5 (es) | 2005-04-11 | 2016-01-21 | Crucell Holland B.V. | Purificación de virus usando ultrafiltración |
| AU2012201827B2 (en) * | 2005-05-12 | 2014-09-04 | Glaxo Group Limited | Vaccine composition |
| US8106176B2 (en) | 2005-10-07 | 2012-01-31 | Instituto di Richerche di Biologia Molecolare P. Angeletti SpA | Matrix metalloproteinase 11 vaccine |
| US9717788B2 (en) | 2007-03-02 | 2017-08-01 | Glaxosmithkline Biologicals Sa | Method of inducing an immune response against HIV employing HIV immunogens, adenoviral vectors encoding said immunogens, and adjuvant |
| CL2008000611A1 (es) * | 2007-03-02 | 2008-09-05 | Glaxosmithkline Biolog Sa | Procedimiento para fomentar respuesta inmunitaria frente a un patogeno que comprende administrar i) uno o mas polipeptidos inmunogenicos derivados del patogeno, ii) uno o mas vectores adenoviricos que comprenden uno o mas polinucleotidos que codifica |
| EP2185192B1 (en) | 2007-08-03 | 2018-10-31 | Institut Pasteur | Lentiviral gene transfer vectors and their medicinal applications |
| EP2047861B1 (en) * | 2007-10-12 | 2019-07-31 | Institut Pasteur | Lentiviral gene transfer vectors suitable for iterative administration and their medicinal applications |
| WO2009022236A2 (en) | 2007-08-16 | 2009-02-19 | Tripep Ab | Immunogen platform |
| US9839684B2 (en) | 2011-04-06 | 2017-12-12 | Biovaxim Limited | Pharmaceutical compositions comprising inactivated HIV viral particles and non-pathogenic lactobacilli for the induction of antigen-specific immunotolerance |
| US8932607B2 (en) | 2012-03-12 | 2015-01-13 | Crucell Holland B.V. | Batches of recombinant adenovirus with altered terminal ends |
| PL2825640T3 (pl) | 2012-03-12 | 2016-10-31 | Partie rekombinowanych adenowirusów o zmienionych końcach | |
| NZ708144A (en) | 2012-11-16 | 2020-03-27 | Beth Israel Deaconess Medical Ct Inc | Recombinant adenoviruses and use thereof |
| US20180052173A1 (en) | 2015-03-18 | 2018-02-22 | Janssen Vaccines & Prevention B.V. | Assays for recombinant expression systems |
| PL3283634T3 (pl) | 2015-04-14 | 2019-10-31 | Janssen Vaccines & Prevention Bv | Rekombinowany adenowirus eksprymujący dwa transgeny z dwukierunkowym promotorem |
| ES2821876T3 (es) | 2016-06-20 | 2021-04-28 | Janssen Vaccines & Prevention Bv | Promotor bidireccional potente y equilibrado |
| AU2018360253B2 (en) | 2017-10-31 | 2023-11-09 | Janssen Vaccines & Prevention B.V. | Adenovirus and uses thereof |
| KR102755022B1 (ko) | 2017-10-31 | 2025-01-20 | 얀센 백신스 앤드 프리벤션 비.브이. | 아데노바이러스 벡터 및 이의 용도 |
| EA202091004A1 (ru) | 2017-10-31 | 2020-08-10 | Янссен Вэксинс Энд Превеншн Б.В. | Аденовирус и пути его применения |
| EP3703722B1 (en) | 2017-10-31 | 2024-06-12 | Janssen Vaccines & Prevention B.V. | Adenovirus and uses thereof |
| US11773142B2 (en) | 2017-12-11 | 2023-10-03 | Beth Israel Deaconess Medical Center, Inc. | Recombinant adenoviruses and uses thereof |
| CN111057716B (zh) * | 2019-07-27 | 2022-04-15 | 中国人民解放军军事科学院军事医学研究院 | 用于包装重组人4型腺病毒的单质粒载体系统及其应用 |
| IL291852A (en) | 2019-10-03 | 2022-06-01 | Janssen Vaccines & Prevention Bv | Adenovirus vectors and their use |
| CN112522276B (zh) * | 2020-12-15 | 2022-07-15 | 武汉纽福斯生物科技有限公司 | 一种emc1核苷酸序列及其应用 |
| WO2025242657A1 (en) | 2024-05-21 | 2025-11-27 | Shape Biopharmaceuticals Ag | Lipopeptide building blocks and aggregates |
| CN118931872B (zh) * | 2024-10-14 | 2025-05-20 | 南京诺唯赞生物科技股份有限公司 | Rna聚合酶变体及其应用 |
Family Cites Families (16)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5643579A (en) * | 1984-11-01 | 1997-07-01 | American Home Products Corporation | Oral vaccines |
| FR2705686B1 (fr) * | 1993-05-28 | 1995-08-18 | Transgene Sa | Nouveaux adénovirus défectifs et lignées de complémentation correspondantes. |
| WO1996009399A2 (en) * | 1994-09-23 | 1996-03-28 | Somatix Therapy Corporation | Chimeric adenovirus for gene delivery |
| US5872005A (en) * | 1994-11-03 | 1999-02-16 | Cell Genesys Inc. | Packaging cell lines for adeno-associated viral vectors |
| WO1996039178A1 (en) * | 1995-06-05 | 1996-12-12 | The Wistar Institute Of Anatomy And Biology | A replication-defective adenovirus human type 5 recombinant as a vaccine carrier |
| US6019978A (en) * | 1995-06-05 | 2000-02-01 | The Wistar Institute Of Anatomy And Biology | Replication-defective adenovirus human type 5 recombinant as a vaccine carrier |
| WO1999002647A2 (de) * | 1997-07-10 | 1999-01-21 | Hepavec Ag Für Gentherapie | Klonierungsvektoren für die herstellung von adenoviralen minimalviren |
| GB9803351D0 (en) * | 1998-02-17 | 1998-04-15 | Oxford Biomedica Ltd | Anti-viral vectors |
| US6670188B1 (en) * | 1998-04-24 | 2003-12-30 | Crucell Holland B.V. | Packaging systems for human recombinant adenovirus to be used in gene therapy |
| WO2000015819A1 (en) * | 1998-09-11 | 2000-03-23 | The Children's Medical Center Corporation | Packaging cell lines for hiv-derived retroviral vector particles |
| EP1980617A1 (en) * | 1998-12-31 | 2008-10-15 | Novartis Vaccines and Diagnostics, Inc. | Improved expression of HIV polypeptides and production of virus-like particles |
| GB9906177D0 (en) * | 1999-03-17 | 1999-05-12 | Oxford Biomedica Ltd | Anti-viral vectors |
| GB9912965D0 (en) * | 1999-06-03 | 1999-08-04 | Oxford Biomedica Ltd | In vivo selection method |
| JP2003520786A (ja) * | 1999-12-22 | 2003-07-08 | メルク エンド カムパニー インコーポレーテッド | コドン最適化hiv−1polおよび修飾hiv−1polを発現するポリヌクレオチドワクチン |
| DE60026199T2 (de) * | 2000-05-18 | 2006-11-23 | Geneart Ag | Synthetische Gene für gagpol und deren Verwendungen |
| JP5052732B2 (ja) * | 2000-08-14 | 2012-10-17 | アメリカ合衆国 | 遺伝的免疫化のための免疫原性を強化するHIVEnv、Gag、およびPolの改変 |
-
2001
- 2001-09-14 CA CA002422882A patent/CA2422882A1/en not_active Abandoned
- 2001-09-14 AU AU9456201A patent/AU9456201A/xx active Pending
- 2001-09-14 WO PCT/US2001/028861 patent/WO2002022080A2/en not_active Ceased
- 2001-09-14 AU AU2001294562A patent/AU2001294562B2/en not_active Ceased
- 2001-09-14 EP EP01975214A patent/EP1320621A4/en not_active Withdrawn
- 2001-09-14 JP JP2002526335A patent/JP2004508064A/ja not_active Withdrawn
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2007532656A (ja) * | 2004-04-12 | 2007-11-15 | アメリカ合衆国 | アデノウイルスベクターを用いて免疫応答を誘導するための方法 |
| JP2008539746A (ja) * | 2005-05-12 | 2008-11-20 | グラクソ グループ リミテッド | ワクチン組成物 |
| JP2013046613A (ja) * | 2005-05-12 | 2013-03-07 | Glaxo Group Ltd | ワクチン組成物 |
| JP2018035177A (ja) * | 2008-11-18 | 2018-03-08 | ベス イスラエル デアコネス メディカル センター インコーポレイテッド | 細胞性免疫原性が向上した抗ウイルスワクチン |
Also Published As
| Publication number | Publication date |
|---|---|
| AU2001294562B2 (en) | 2007-05-24 |
| WO2002022080A9 (en) | 2003-03-06 |
| AU2001294562B8 (en) | 2002-03-26 |
| WO2002022080A3 (en) | 2002-05-02 |
| EP1320621A4 (en) | 2005-11-23 |
| AU9456201A (en) | 2002-03-26 |
| CA2422882A1 (en) | 2002-03-21 |
| WO2002022080A8 (en) | 2003-01-16 |
| WO2002022080A2 (en) | 2002-03-21 |
| EP1320621A2 (en) | 2003-06-25 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP2004508064A (ja) | コドン最適化hiv1−gag、pol、nefおよび修飾体を発現する増強された第1世代アデノウイルスワクチン | |
| US20050070017A1 (en) | Enhanced first generation adenovirus vaccines expressing codon optimized HIV1-Gag, Pol, Nef and modifications | |
| JP2003530307A (ja) | gag遺伝子保有アデノウイルスHIVワクチン | |
| AU2001294562A1 (en) | Enhanced First Generation Adenovirus Vaccines Expressing Codon Optimized HIV1-Gag, Pol, Nef and Modifications | |
| CA2597404A1 (en) | Adenovirus serotype 26 vectors, nucleic acid and viruses produced thereby | |
| AU2003262790A1 (en) | Adenovirus serotype 24 vectors, nucleic acids and virus produced thereby | |
| US20080063656A1 (en) | Adenoviral Vector Compositions | |
| AU2003298554A1 (en) | Adenovirus serotype 34 vectors, nucleic acids and virus produced thereby | |
| US20070054395A1 (en) | Enhanced first generation adenovirus vaccines expressing codon optimized HIV1-Gag, Pol, Nef and modifications | |
| CN1972958B (zh) | 应用腺病毒载体诱导免疫应答的方法 | |
| US20060165664A1 (en) | Method of inducing an enhanced immune response against hiv | |
| US20050106123A1 (en) | Method of inducing an enhanced immune response against hiv | |
| US20070077257A1 (en) | Enhanced first generation adenovirus vaccines expressing condon optimized HIV1-Gag, Pol, Nef and modifications |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A300 | Application deemed to be withdrawn because no request for examination was validly filed |
Free format text: JAPANESE INTERMEDIATE CODE: A300 Effective date: 20081202 |