EP1194121B1 - Steroid solution aerosol products with enhanced chemical stability - Google Patents
Steroid solution aerosol products with enhanced chemical stability Download PDFInfo
- Publication number
- EP1194121B1 EP1194121B1 EP00941435A EP00941435A EP1194121B1 EP 1194121 B1 EP1194121 B1 EP 1194121B1 EP 00941435 A EP00941435 A EP 00941435A EP 00941435 A EP00941435 A EP 00941435A EP 1194121 B1 EP1194121 B1 EP 1194121B1
- Authority
- EP
- European Patent Office
- Prior art keywords
- ketosteroid
- container
- coating
- group
- budesonide
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Lifetime
Links
- 239000000443 aerosol Substances 0.000 title claims abstract description 44
- 150000003431 steroids Chemical class 0.000 title claims abstract description 31
- 239000000126 substance Substances 0.000 title abstract description 23
- 239000000203 mixture Substances 0.000 claims abstract description 87
- 238000009472 formulation Methods 0.000 claims abstract description 77
- 125000002887 hydroxy group Chemical group [H]O* 0.000 claims abstract description 25
- 238000002144 chemical decomposition reaction Methods 0.000 claims abstract description 19
- 229910052755 nonmetal Inorganic materials 0.000 claims abstract description 8
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 claims description 100
- 238000000034 method Methods 0.000 claims description 54
- 238000000576 coating method Methods 0.000 claims description 49
- 239000011248 coating agent Substances 0.000 claims description 45
- VOVIALXJUBGFJZ-KWVAZRHASA-N Budesonide Chemical compound C1CC2=CC(=O)C=C[C@]2(C)[C@@H]2[C@@H]1[C@@H]1C[C@H]3OC(CCC)O[C@@]3(C(=O)CO)[C@@]1(C)C[C@@H]2O VOVIALXJUBGFJZ-KWVAZRHASA-N 0.000 claims description 40
- 229960004436 budesonide Drugs 0.000 claims description 39
- 229910052751 metal Inorganic materials 0.000 claims description 36
- 239000002184 metal Substances 0.000 claims description 36
- 239000003380 propellant Substances 0.000 claims description 28
- 230000008569 process Effects 0.000 claims description 26
- 229910052782 aluminium Inorganic materials 0.000 claims description 23
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 claims description 23
- 229960002117 triamcinolone acetonide Drugs 0.000 claims description 17
- YNDXUCZADRHECN-JNQJZLCISA-N triamcinolone acetonide Chemical compound C1CC2=CC(=O)C=C[C@]2(C)[C@]2(F)[C@@H]1[C@@H]1C[C@H]3OC(C)(C)O[C@@]3(C(=O)CO)[C@@]1(C)C[C@@H]2O YNDXUCZADRHECN-JNQJZLCISA-N 0.000 claims description 17
- 229960003957 dexamethasone Drugs 0.000 claims description 16
- UREBDLICKHMUKA-CXSFZGCWSA-N dexamethasone Chemical compound C1CC2=CC(=O)C=C[C@]2(C)[C@]2(F)[C@@H]1[C@@H]1C[C@@H](C)[C@@](C(=O)CO)(O)[C@@]1(C)C[C@@H]2O UREBDLICKHMUKA-CXSFZGCWSA-N 0.000 claims description 16
- 229910052739 hydrogen Inorganic materials 0.000 claims description 14
- 239000011521 glass Substances 0.000 claims description 13
- 229960000676 flunisolide Drugs 0.000 claims description 12
- MIXMJCQRHVAJIO-TZHJZOAOSA-N qk4dys664x Chemical compound O.C1([C@@H](F)C2)=CC(=O)C=C[C@]1(C)[C@@H]1[C@@H]2[C@@H]2C[C@H]3OC(C)(C)O[C@@]3(C(=O)CO)[C@@]2(C)C[C@@H]1O.C1([C@@H](F)C2)=CC(=O)C=C[C@]1(C)[C@@H]1[C@@H]2[C@@H]2C[C@H]3OC(C)(C)O[C@@]3(C(=O)CO)[C@@]2(C)C[C@@H]1O MIXMJCQRHVAJIO-TZHJZOAOSA-N 0.000 claims description 12
- 239000000463 material Substances 0.000 claims description 10
- 229910044991 metal oxide Inorganic materials 0.000 claims description 10
- 150000004706 metal oxides Chemical class 0.000 claims description 10
- 239000004480 active ingredient Substances 0.000 claims description 9
- 229910052801 chlorine Inorganic materials 0.000 claims description 9
- 239000000460 chlorine Substances 0.000 claims description 9
- 239000006184 cosolvent Substances 0.000 claims description 9
- 229940071648 metered dose inhaler Drugs 0.000 claims description 9
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 claims description 8
- 239000004411 aluminium Substances 0.000 claims description 8
- 239000001257 hydrogen Substances 0.000 claims description 8
- SNHRLVCMMWUAJD-SUYDQAKGSA-N betamethasone valerate Chemical compound C1CC2=CC(=O)C=C[C@]2(C)[C@]2(F)[C@@H]1[C@@H]1C[C@H](C)[C@@](C(=O)CO)(OC(=O)CCCC)[C@@]1(C)C[C@@H]2O SNHRLVCMMWUAJD-SUYDQAKGSA-N 0.000 claims description 6
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 claims description 5
- NDAUXUAQIAJITI-UHFFFAOYSA-N albuterol Chemical compound CC(C)(C)NCC(O)C1=CC=C(O)C(CO)=C1 NDAUXUAQIAJITI-UHFFFAOYSA-N 0.000 claims description 5
- BPZSYCZIITTYBL-UHFFFAOYSA-N formoterol Chemical compound C1=CC(OC)=CC=C1CC(C)NCC(O)C1=CC=C(O)C(NC=O)=C1 BPZSYCZIITTYBL-UHFFFAOYSA-N 0.000 claims description 5
- 229960002848 formoterol Drugs 0.000 claims description 5
- 150000005828 hydrofluoroalkanes Chemical class 0.000 claims description 5
- 229960001361 ipratropium bromide Drugs 0.000 claims description 5
- KEWHKYJURDBRMN-ZEODDXGYSA-M ipratropium bromide hydrate Chemical compound O.[Br-].O([C@H]1C[C@H]2CC[C@@H](C1)[N@@+]2(C)C(C)C)C(=O)C(CO)C1=CC=CC=C1 KEWHKYJURDBRMN-ZEODDXGYSA-M 0.000 claims description 5
- 239000004033 plastic Substances 0.000 claims description 5
- 229920003023 plastic Polymers 0.000 claims description 5
- 229960002052 salbutamol Drugs 0.000 claims description 5
- 239000010935 stainless steel Substances 0.000 claims description 5
- 229910001220 stainless steel Inorganic materials 0.000 claims description 5
- 239000005028 tinplate Substances 0.000 claims description 5
- OHYGPBKGZGRQKT-XGQKBEPLSA-N [(8s,9r,10s,11s,13s,14s,16s,17r)-9-chloro-11-hydroxy-17-(2-hydroxyacetyl)-10,13,16-trimethyl-3-oxo-6,7,8,11,12,14,15,16-octahydrocyclopenta[a]phenanthren-17-yl] propanoate Chemical compound C1CC2=CC(=O)C=C[C@]2(C)[C@]2(Cl)[C@@H]1[C@@H]1C[C@H](C)[C@@](C(=O)CO)(OC(=O)CC)[C@@]1(C)C[C@@H]2O OHYGPBKGZGRQKT-XGQKBEPLSA-N 0.000 claims description 4
- 238000006243 chemical reaction Methods 0.000 claims description 4
- 230000003993 interaction Effects 0.000 claims description 4
- 239000004922 lacquer Substances 0.000 claims description 4
- 238000007740 vapor deposition Methods 0.000 claims description 4
- 229940006003 beclomethasone 17-monopropionate Drugs 0.000 claims description 3
- 229960003662 desonide Drugs 0.000 claims description 3
- 238000011049 filling Methods 0.000 claims description 3
- LVGUZGTVOIAKKC-UHFFFAOYSA-N 1,1,1,2-tetrafluoroethane Chemical compound FCC(F)(F)F LVGUZGTVOIAKKC-UHFFFAOYSA-N 0.000 claims description 2
- VHRSUDSXCMQTMA-PJHHCJLFSA-N 6alpha-methylprednisolone Chemical compound C([C@@]12C)=CC(=O)C=C1[C@@H](C)C[C@@H]1[C@@H]2[C@@H](O)C[C@]2(C)[C@@](O)(C(=O)CO)CC[C@H]21 VHRSUDSXCMQTMA-PJHHCJLFSA-N 0.000 claims description 2
- MKPDWECBUAZOHP-AFYJWTTESA-N Paramethasone Chemical compound C1([C@@H](F)C2)=CC(=O)C=C[C@]1(C)[C@@H]1[C@@H]2[C@@H]2C[C@@H](C)[C@@](C(=O)CO)(O)[C@@]2(C)C[C@@H]1O MKPDWECBUAZOHP-AFYJWTTESA-N 0.000 claims description 2
- 229960000552 alclometasone Drugs 0.000 claims description 2
- FJXOGVLKCZQRDN-PHCHRAKRSA-N alclometasone Chemical compound C([C@H]1Cl)C2=CC(=O)C=C[C@]2(C)[C@@H]2[C@@H]1[C@@H]1C[C@@H](C)[C@@](C(=O)CO)(O)[C@@]1(C)C[C@@H]2O FJXOGVLKCZQRDN-PHCHRAKRSA-N 0.000 claims description 2
- 229940092705 beclomethasone Drugs 0.000 claims description 2
- 229960002537 betamethasone Drugs 0.000 claims description 2
- UREBDLICKHMUKA-DVTGEIKXSA-N betamethasone Chemical compound C1CC2=CC(=O)C=C[C@]2(C)[C@]2(F)[C@@H]1[C@@H]1C[C@H](C)[C@@](C(=O)CO)(O)[C@@]1(C)C[C@@H]2O UREBDLICKHMUKA-DVTGEIKXSA-N 0.000 claims description 2
- 229960002842 clobetasol Drugs 0.000 claims description 2
- CBGUOGMQLZIXBE-XGQKBEPLSA-N clobetasol propionate Chemical compound C1CC2=CC(=O)C=C[C@]2(C)[C@]2(F)[C@@H]1[C@@H]1C[C@H](C)[C@@](C(=O)CCl)(OC(=O)CC)[C@@]1(C)C[C@@H]2O CBGUOGMQLZIXBE-XGQKBEPLSA-N 0.000 claims description 2
- 229960004299 clocortolone Drugs 0.000 claims description 2
- YMTMADLUXIRMGX-RFPWEZLHSA-N clocortolone Chemical compound C1([C@@H](F)C2)=CC(=O)C=C[C@]1(C)[C@]1(Cl)[C@@H]2[C@@H]2C[C@@H](C)[C@H](C(=O)CO)[C@@]2(C)C[C@@H]1O YMTMADLUXIRMGX-RFPWEZLHSA-N 0.000 claims description 2
- WBGKWQHBNHJJPZ-LECWWXJVSA-N desonide Chemical compound C1CC2=CC(=O)C=C[C@]2(C)[C@@H]2[C@@H]1[C@@H]1C[C@H]3OC(C)(C)O[C@@]3(C(=O)CO)[C@@]1(C)C[C@@H]2O WBGKWQHBNHJJPZ-LECWWXJVSA-N 0.000 claims description 2
- 229960002593 desoximetasone Drugs 0.000 claims description 2
- VWVSBHGCDBMOOT-IIEHVVJPSA-N desoximetasone Chemical compound C1CC2=CC(=O)C=C[C@]2(C)[C@]2(F)[C@@H]1[C@@H]1C[C@@H](C)[C@H](C(=O)CO)[C@@]1(C)C[C@@H]2O VWVSBHGCDBMOOT-IIEHVVJPSA-N 0.000 claims description 2
- 229960002344 dexamethasone sodium phosphate Drugs 0.000 claims description 2
- PLCQGRYPOISRTQ-FCJDYXGNSA-L dexamethasone sodium phosphate Chemical compound [Na+].[Na+].C1CC2=CC(=O)C=C[C@]2(C)[C@]2(F)[C@@H]1[C@@H]1C[C@@H](C)[C@@](C(=O)COP([O-])([O-])=O)(O)[C@@]1(C)C[C@@H]2O PLCQGRYPOISRTQ-FCJDYXGNSA-L 0.000 claims description 2
- 229960004154 diflorasone Drugs 0.000 claims description 2
- WXURHACBFYSXBI-XHIJKXOTSA-N diflorasone Chemical compound C1([C@@H](F)C2)=CC(=O)C=C[C@]1(C)[C@]1(F)[C@@H]2[C@@H]2C[C@H](C)[C@@](C(=O)CO)(O)[C@@]2(C)C[C@@H]1O WXURHACBFYSXBI-XHIJKXOTSA-N 0.000 claims description 2
- 229960003469 flumetasone Drugs 0.000 claims description 2
- WXURHACBFYSXBI-GQKYHHCASA-N flumethasone Chemical compound C1([C@@H](F)C2)=CC(=O)C=C[C@]1(C)[C@]1(F)[C@@H]2[C@@H]2C[C@@H](C)[C@@](C(=O)CO)(O)[C@@]2(C)C[C@@H]1O WXURHACBFYSXBI-GQKYHHCASA-N 0.000 claims description 2
- 229960001347 fluocinolone acetonide Drugs 0.000 claims description 2
- FEBLZLNTKCEFIT-VSXGLTOVSA-N fluocinolone acetonide Chemical compound C1([C@@H](F)C2)=CC(=O)C=C[C@]1(C)[C@]1(F)[C@@H]2[C@@H]2C[C@H]3OC(C)(C)O[C@@]3(C(=O)CO)[C@@]2(C)C[C@@H]1O FEBLZLNTKCEFIT-VSXGLTOVSA-N 0.000 claims description 2
- 229960001048 fluorometholone Drugs 0.000 claims description 2
- FAOZLTXFLGPHNG-KNAQIMQKSA-N fluorometholone Chemical compound C([C@@]12C)=CC(=O)C=C1[C@@H](C)C[C@@H]1[C@]2(F)[C@@H](O)C[C@]2(C)[C@@](O)(C(C)=O)CC[C@H]21 FAOZLTXFLGPHNG-KNAQIMQKSA-N 0.000 claims description 2
- 229960004584 methylprednisolone Drugs 0.000 claims description 2
- 229960002858 paramethasone Drugs 0.000 claims description 2
- 229960005205 prednisolone Drugs 0.000 claims description 2
- OIGNJSKKLXVSLS-VWUMJDOOSA-N prednisolone Chemical compound O=C1C=C[C@]2(C)[C@H]3[C@@H](O)C[C@](C)([C@@](CC4)(O)C(=O)CO)[C@@H]4[C@@H]3CCC2=C1 OIGNJSKKLXVSLS-VWUMJDOOSA-N 0.000 claims description 2
- YFMFNYKEUDLDTL-UHFFFAOYSA-N 1,1,1,2,3,3,3-heptafluoropropane Chemical compound FC(F)(F)C(F)C(F)(F)F YFMFNYKEUDLDTL-UHFFFAOYSA-N 0.000 claims 1
- NBMKJKDGKREAPL-DVTGEIKXSA-N beclomethasone Chemical compound C1CC2=CC(=O)C=C[C@]2(C)[C@]2(Cl)[C@@H]1[C@@H]1C[C@H](C)[C@@](C(=O)CO)(O)[C@@]1(C)C[C@@H]2O NBMKJKDGKREAPL-DVTGEIKXSA-N 0.000 claims 1
- 239000003246 corticosteroid Substances 0.000 claims 1
- 230000000063 preceeding effect Effects 0.000 claims 1
- 229960005294 triamcinolone Drugs 0.000 claims 1
- GFNANZIMVAIWHM-OBYCQNJPSA-N triamcinolone Chemical compound O=C1C=C[C@]2(C)[C@@]3(F)[C@@H](O)C[C@](C)([C@@]([C@H](O)C4)(O)C(=O)CO)[C@@H]4[C@@H]3CCC2=C1 GFNANZIMVAIWHM-OBYCQNJPSA-N 0.000 claims 1
- 239000000243 solution Substances 0.000 description 34
- TWNQGVIAIRXVLR-UHFFFAOYSA-N oxo(oxoalumanyloxy)alumane Chemical compound O=[Al]O[Al]=O TWNQGVIAIRXVLR-UHFFFAOYSA-N 0.000 description 27
- 239000000047 product Substances 0.000 description 22
- 230000015556 catabolic process Effects 0.000 description 19
- 238000006731 degradation reaction Methods 0.000 description 19
- 238000011084 recovery Methods 0.000 description 13
- 239000000725 suspension Substances 0.000 description 13
- 239000004593 Epoxy Substances 0.000 description 10
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 9
- 229910052799 carbon Inorganic materials 0.000 description 8
- 238000012360 testing method Methods 0.000 description 8
- -1 MDI steroid Chemical class 0.000 description 7
- 239000010410 layer Substances 0.000 description 6
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 6
- 239000003814 drug Substances 0.000 description 5
- 229920001343 polytetrafluoroethylene Polymers 0.000 description 5
- LCGLNKUTAGEVQW-UHFFFAOYSA-N Dimethyl ether Chemical compound COC LCGLNKUTAGEVQW-UHFFFAOYSA-N 0.000 description 4
- 230000008901 benefit Effects 0.000 description 4
- 238000000354 decomposition reaction Methods 0.000 description 4
- 229940079593 drug Drugs 0.000 description 4
- 230000000694 effects Effects 0.000 description 4
- 239000012535 impurity Substances 0.000 description 4
- 230000007935 neutral effect Effects 0.000 description 4
- WRIDQFICGBMAFQ-UHFFFAOYSA-N (E)-8-Octadecenoic acid Natural products CCCCCCCCCC=CCCCCCCC(O)=O WRIDQFICGBMAFQ-UHFFFAOYSA-N 0.000 description 3
- LQJBNNIYVWPHFW-UHFFFAOYSA-N 20:1omega9c fatty acid Natural products CCCCCCCCCCC=CCCCCCCCC(O)=O LQJBNNIYVWPHFW-UHFFFAOYSA-N 0.000 description 3
- QSBYPNXLFMSGKH-UHFFFAOYSA-N 9-Heptadecensaeure Natural products CCCCCCCC=CCCCCCCCC(O)=O QSBYPNXLFMSGKH-UHFFFAOYSA-N 0.000 description 3
- KUVIULQEHSCUHY-XYWKZLDCSA-N Beclometasone Chemical compound C1CC2=CC(=O)C=C[C@]2(C)[C@]2(Cl)[C@@H]1[C@@H]1C[C@H](C)[C@@](C(=O)COC(=O)CC)(OC(=O)CC)[C@@]1(C)C[C@@H]2O KUVIULQEHSCUHY-XYWKZLDCSA-N 0.000 description 3
- 239000005642 Oleic acid Substances 0.000 description 3
- ZQPPMHVWECSIRJ-UHFFFAOYSA-N Oleic acid Natural products CCCCCCCCC=CCCCCCCCC(O)=O ZQPPMHVWECSIRJ-UHFFFAOYSA-N 0.000 description 3
- 125000000217 alkyl group Chemical group 0.000 description 3
- 239000011247 coating layer Substances 0.000 description 3
- 239000002274 desiccant Substances 0.000 description 3
- 239000005350 fused silica glass Substances 0.000 description 3
- 239000007789 gas Substances 0.000 description 3
- 238000004128 high performance liquid chromatography Methods 0.000 description 3
- QXJSBBXBKPUZAA-UHFFFAOYSA-N isooleic acid Natural products CCCCCCCC=CCCCCCCCCC(O)=O QXJSBBXBKPUZAA-UHFFFAOYSA-N 0.000 description 3
- ZQPPMHVWECSIRJ-KTKRTIGZSA-N oleic acid Chemical compound CCCCCCCC\C=C/CCCCCCCC(O)=O ZQPPMHVWECSIRJ-KTKRTIGZSA-N 0.000 description 3
- ISWSIDIOOBJBQZ-UHFFFAOYSA-N phenol group Chemical group C1(=CC=CC=C1)O ISWSIDIOOBJBQZ-UHFFFAOYSA-N 0.000 description 3
- 229920000642 polymer Polymers 0.000 description 3
- 229920005989 resin Polymers 0.000 description 3
- 239000011347 resin Substances 0.000 description 3
- GVJHHUAWPYXKBD-UHFFFAOYSA-N (±)-α-Tocopherol Chemical compound OC1=C(C)C(C)=C2OC(CCCC(C)CCCC(C)CCCC(C)C)(C)CCC2=C1C GVJHHUAWPYXKBD-UHFFFAOYSA-N 0.000 description 2
- 229910000838 Al alloy Inorganic materials 0.000 description 2
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical group [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 2
- 239000011786 L-ascorbyl-6-palmitate Substances 0.000 description 2
- 239000004813 Perfluoroalkoxy alkane Substances 0.000 description 2
- 229920001807 Urea-formaldehyde Polymers 0.000 description 2
- 230000003110 anti-inflammatory effect Effects 0.000 description 2
- 239000003963 antioxidant agent Substances 0.000 description 2
- 235000006708 antioxidants Nutrition 0.000 description 2
- 230000004888 barrier function Effects 0.000 description 2
- 229950000210 beclometasone dipropionate Drugs 0.000 description 2
- 238000005229 chemical vapour deposition Methods 0.000 description 2
- 238000000151 deposition Methods 0.000 description 2
- 230000008021 deposition Effects 0.000 description 2
- 238000013461 design Methods 0.000 description 2
- 239000005007 epoxy-phenolic resin Substances 0.000 description 2
- QHSJIZLJUFMIFP-UHFFFAOYSA-N ethene;1,1,2,2-tetrafluoroethene Chemical group C=C.FC(F)=C(F)F QHSJIZLJUFMIFP-UHFFFAOYSA-N 0.000 description 2
- 229920000840 ethylene tetrafluoroethylene copolymer Polymers 0.000 description 2
- 229930195733 hydrocarbon Natural products 0.000 description 2
- 150000002430 hydrocarbons Chemical class 0.000 description 2
- 238000013101 initial test Methods 0.000 description 2
- 229920011301 perfluoro alkoxyl alkane Polymers 0.000 description 2
- 238000005240 physical vapour deposition Methods 0.000 description 2
- 239000007921 spray Substances 0.000 description 2
- 125000001424 substituent group Chemical group 0.000 description 2
- CIWBSHSKHKDKBQ-JLAZNSOCSA-N Ascorbic acid Natural products OC[C@H](O)[C@H]1OC(=O)C(O)=C1O CIWBSHSKHKDKBQ-JLAZNSOCSA-N 0.000 description 1
- 0 CC(CC1O)(C(CC2*3)C(C(*)C4*)C1(*)C(C)(C=C1)C4=CC1O)C23C(*)=O Chemical compound CC(CC1O)(C(CC2*3)C(C(*)C4*)C1(*)C(C)(C=C1)C4=CC1O)C23C(*)=O 0.000 description 1
- LUKZNWIVRBCLON-GXOBDPJESA-N Ciclesonide Chemical compound C1([C@H]2O[C@@]3([C@H](O2)C[C@@H]2[C@@]3(C[C@H](O)[C@@H]3[C@@]4(C)C=CC(=O)C=C4CC[C@H]32)C)C(=O)COC(=O)C(C)C)CCCCC1 LUKZNWIVRBCLON-GXOBDPJESA-N 0.000 description 1
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 1
- BQTXJHAJMDGOFI-NJLPOHDGSA-N Dexamethasone 21-(4-Pyridinecarboxylate) Chemical compound O=C([C@]1(O)[C@@]2(C)C[C@H](O)[C@]3(F)[C@@]4(C)C=CC(=O)C=C4CC[C@H]3[C@@H]2C[C@H]1C)COC(=O)C1=CC=NC=C1 BQTXJHAJMDGOFI-NJLPOHDGSA-N 0.000 description 1
- KCXVZYZYPLLWCC-UHFFFAOYSA-N EDTA Chemical compound OC(=O)CN(CC(O)=O)CCN(CC(O)=O)CC(O)=O KCXVZYZYPLLWCC-UHFFFAOYSA-N 0.000 description 1
- 229920002943 EPDM rubber Polymers 0.000 description 1
- KRHYYFGTRYWZRS-UHFFFAOYSA-M Fluoride anion Chemical compound [F-] KRHYYFGTRYWZRS-UHFFFAOYSA-M 0.000 description 1
- 235000000072 L-ascorbyl-6-palmitate Nutrition 0.000 description 1
- QAQJMLQRFWZOBN-LAUBAEHRSA-N L-ascorbyl-6-palmitate Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](O)[C@H]1OC(=O)C(O)=C1O QAQJMLQRFWZOBN-LAUBAEHRSA-N 0.000 description 1
- 239000004677 Nylon Substances 0.000 description 1
- CBENFWSGALASAD-UHFFFAOYSA-N Ozone Chemical compound [O-][O+]=O CBENFWSGALASAD-UHFFFAOYSA-N 0.000 description 1
- 239000004952 Polyamide Substances 0.000 description 1
- 239000004962 Polyamide-imide Substances 0.000 description 1
- 239000004642 Polyimide Substances 0.000 description 1
- 239000004721 Polyphenylene oxide Substances 0.000 description 1
- 239000004734 Polyphenylene sulfide Substances 0.000 description 1
- XBDQKXXYIPTUBI-UHFFFAOYSA-M Propionate Chemical compound CCC([O-])=O XBDQKXXYIPTUBI-UHFFFAOYSA-M 0.000 description 1
- 206010039085 Rhinitis allergic Diseases 0.000 description 1
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 1
- PRXRUNOAOLTIEF-ADSICKODSA-N Sorbitan trioleate Chemical compound CCCCCCCC\C=C/CCCCCCCC(=O)OC[C@@H](OC(=O)CCCCCCC\C=C/CCCCCCCC)[C@H]1OC[C@H](O)[C@H]1OC(=O)CCCCCCC\C=C/CCCCCCCC PRXRUNOAOLTIEF-ADSICKODSA-N 0.000 description 1
- 239000004147 Sorbitan trioleate Substances 0.000 description 1
- 229930003427 Vitamin E Natural products 0.000 description 1
- 238000010521 absorption reaction Methods 0.000 description 1
- 239000013543 active substance Substances 0.000 description 1
- 239000000654 additive Substances 0.000 description 1
- 238000005054 agglomeration Methods 0.000 description 1
- 230000002776 aggregation Effects 0.000 description 1
- 201000010105 allergic rhinitis Diseases 0.000 description 1
- 238000004458 analytical method Methods 0.000 description 1
- 238000002048 anodisation reaction Methods 0.000 description 1
- 239000013011 aqueous formulation Substances 0.000 description 1
- 229960005070 ascorbic acid Drugs 0.000 description 1
- 235000010385 ascorbyl palmitate Nutrition 0.000 description 1
- 208000006673 asthma Diseases 0.000 description 1
- IWLBIFVMPLUHLK-UHFFFAOYSA-N azane;formaldehyde Chemical compound N.O=C IWLBIFVMPLUHLK-UHFFFAOYSA-N 0.000 description 1
- 239000003637 basic solution Substances 0.000 description 1
- 230000004071 biological effect Effects 0.000 description 1
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 229960001927 cetylpyridinium chloride Drugs 0.000 description 1
- YMKDRGPMQRFJGP-UHFFFAOYSA-M cetylpyridinium chloride Chemical compound [Cl-].CCCCCCCCCCCCCCCC[N+]1=CC=CC=C1 YMKDRGPMQRFJGP-UHFFFAOYSA-M 0.000 description 1
- 230000008859 change Effects 0.000 description 1
- 229960003728 ciclesonide Drugs 0.000 description 1
- 230000000052 comparative effect Effects 0.000 description 1
- 229910052802 copper Inorganic materials 0.000 description 1
- 239000010949 copper Substances 0.000 description 1
- 239000013078 crystal Substances 0.000 description 1
- 125000004122 cyclic group Chemical group 0.000 description 1
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 1
- 125000001511 cyclopentyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 1
- 239000007857 degradation product Substances 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- 238000012377 drug delivery Methods 0.000 description 1
- 229920001971 elastomer Polymers 0.000 description 1
- 239000000806 elastomer Substances 0.000 description 1
- 230000002708 enhancing effect Effects 0.000 description 1
- 229920006334 epoxy coating Polymers 0.000 description 1
- 239000003822 epoxy resin Substances 0.000 description 1
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 239000012632 extractable Substances 0.000 description 1
- 239000000945 filler Substances 0.000 description 1
- 239000000796 flavoring agent Substances 0.000 description 1
- 229920002313 fluoropolymer Polymers 0.000 description 1
- 235000013355 food flavoring agent Nutrition 0.000 description 1
- WIGCFUFOHFEKBI-UHFFFAOYSA-N gamma-tocopherol Natural products CC(C)CCCC(C)CCCC(C)CCCC1CCC2C(C)C(O)C(C)C(C)C2O1 WIGCFUFOHFEKBI-UHFFFAOYSA-N 0.000 description 1
- 125000004051 hexyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 150000002576 ketones Chemical class 0.000 description 1
- TYQCGQRIZGCHNB-JLAZNSOCSA-N l-ascorbic acid Chemical compound OC[C@H](O)[C@H]1OC(O)=C(O)C1=O TYQCGQRIZGCHNB-JLAZNSOCSA-N 0.000 description 1
- 210000004072 lung Anatomy 0.000 description 1
- 230000007246 mechanism Effects 0.000 description 1
- 229910021645 metal ion Inorganic materials 0.000 description 1
- 239000007769 metal material Substances 0.000 description 1
- 150000002739 metals Chemical class 0.000 description 1
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 229920006120 non-fluorinated polymer Polymers 0.000 description 1
- 229920001778 nylon Polymers 0.000 description 1
- 239000003921 oil Substances 0.000 description 1
- 230000003647 oxidation Effects 0.000 description 1
- 238000007254 oxidation reaction Methods 0.000 description 1
- 238000010525 oxidative degradation reaction Methods 0.000 description 1
- 239000011236 particulate material Substances 0.000 description 1
- 238000002161 passivation Methods 0.000 description 1
- 125000001147 pentyl group Chemical group C(CCCC)* 0.000 description 1
- 239000000546 pharmaceutical excipient Substances 0.000 description 1
- 238000011020 pilot scale process Methods 0.000 description 1
- 229920002647 polyamide Polymers 0.000 description 1
- 229920002312 polyamide-imide Polymers 0.000 description 1
- 229920000647 polyepoxide Polymers 0.000 description 1
- 229920000570 polyether Polymers 0.000 description 1
- 229920006393 polyether sulfone Polymers 0.000 description 1
- 229920000139 polyethylene terephthalate Polymers 0.000 description 1
- 239000005020 polyethylene terephthalate Substances 0.000 description 1
- 229920001721 polyimide Polymers 0.000 description 1
- 229920000069 polyphenylene sulfide Polymers 0.000 description 1
- 239000000843 powder Substances 0.000 description 1
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 238000011160 research Methods 0.000 description 1
- 239000003352 sequestering agent Substances 0.000 description 1
- 238000002791 soaking Methods 0.000 description 1
- HUAUNKAZQWMVFY-UHFFFAOYSA-M sodium;oxocalcium;hydroxide Chemical compound [OH-].[Na+].[Ca]=O HUAUNKAZQWMVFY-UHFFFAOYSA-M 0.000 description 1
- 230000007928 solubilization Effects 0.000 description 1
- 238000005063 solubilization Methods 0.000 description 1
- 230000003381 solubilizing effect Effects 0.000 description 1
- 229960000391 sorbitan trioleate Drugs 0.000 description 1
- 235000019337 sorbitan trioleate Nutrition 0.000 description 1
- 238000001179 sorption measurement Methods 0.000 description 1
- 238000005507 spraying Methods 0.000 description 1
- 239000003381 stabilizer Substances 0.000 description 1
- 238000005556 structure-activity relationship Methods 0.000 description 1
- 125000001174 sulfone group Chemical group 0.000 description 1
- 239000004094 surface-active agent Substances 0.000 description 1
- 230000001225 therapeutic effect Effects 0.000 description 1
- 229920001187 thermosetting polymer Polymers 0.000 description 1
- 235000019165 vitamin E Nutrition 0.000 description 1
- 229940046009 vitamin E Drugs 0.000 description 1
- 239000011709 vitamin E Substances 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/007—Pulmonary tract; Aromatherapy
- A61K9/0073—Sprays or powders for inhalation; Aerolised or nebulised preparations generated by other means than thermal energy
- A61K9/008—Sprays or powders for inhalation; Aerolised or nebulised preparations generated by other means than thermal energy comprising drug dissolved or suspended in liquid propellant for inhalation via a pressurized metered dose inhaler [MDI]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/56—Compounds containing cyclopenta[a]hydrophenanthrene ring systems; Derivatives thereof, e.g. steroids
- A61K31/57—Compounds containing cyclopenta[a]hydrophenanthrene ring systems; Derivatives thereof, e.g. steroids substituted in position 17 beta by a chain of two carbon atoms, e.g. pregnane or progesterone
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/56—Compounds containing cyclopenta[a]hydrophenanthrene ring systems; Derivatives thereof, e.g. steroids
- A61K31/57—Compounds containing cyclopenta[a]hydrophenanthrene ring systems; Derivatives thereof, e.g. steroids substituted in position 17 beta by a chain of two carbon atoms, e.g. pregnane or progesterone
- A61K31/573—Compounds containing cyclopenta[a]hydrophenanthrene ring systems; Derivatives thereof, e.g. steroids substituted in position 17 beta by a chain of two carbon atoms, e.g. pregnane or progesterone substituted in position 21, e.g. cortisone, dexamethasone, prednisone or aldosterone
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/56—Compounds containing cyclopenta[a]hydrophenanthrene ring systems; Derivatives thereof, e.g. steroids
- A61K31/58—Compounds containing cyclopenta[a]hydrophenanthrene ring systems; Derivatives thereof, e.g. steroids containing heterocyclic rings, e.g. danazol, stanozolol, pancuronium or digitogenin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/10—Dispersions; Emulsions
- A61K9/12—Aerosols; Foams
- A61K9/124—Aerosols; Foams characterised by the propellant
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/02—Stomatological preparations, e.g. drugs for caries, aphtae, periodontitis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P11/00—Drugs for disorders of the respiratory system
- A61P11/06—Antiasthmatics
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B65—CONVEYING; PACKING; STORING; HANDLING THIN OR FILAMENTARY MATERIAL
- B65D—CONTAINERS FOR STORAGE OR TRANSPORT OF ARTICLES OR MATERIALS, e.g. BAGS, BARRELS, BOTTLES, BOXES, CANS, CARTONS, CRATES, DRUMS, JARS, TANKS, HOPPERS, FORWARDING CONTAINERS; ACCESSORIES, CLOSURES, OR FITTINGS THEREFOR; PACKAGING ELEMENTS; PACKAGES
- B65D83/00—Containers or packages with special means for dispensing contents
- B65D83/14—Containers for dispensing liquid or semi-liquid contents by internal gaseous pressure, i.e. aerosol containers comprising propellant
- B65D83/38—Details of the container body
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B65—CONVEYING; PACKING; STORING; HANDLING THIN OR FILAMENTARY MATERIAL
- B65D—CONTAINERS FOR STORAGE OR TRANSPORT OF ARTICLES OR MATERIALS, e.g. BAGS, BARRELS, BOTTLES, BOXES, CANS, CARTONS, CRATES, DRUMS, JARS, TANKS, HOPPERS, FORWARDING CONTAINERS; ACCESSORIES, CLOSURES, OR FITTINGS THEREFOR; PACKAGING ELEMENTS; PACKAGES
- B65D83/00—Containers or packages with special means for dispensing contents
- B65D83/14—Containers for dispensing liquid or semi-liquid contents by internal gaseous pressure, i.e. aerosol containers comprising propellant
- B65D83/44—Valves specially adapted for the discharge of contents; Regulating devices
- B65D83/52—Metering valves; Metering devices
Definitions
- the present invention relates to medicinal aerosol products and, in particular, to aerosol products such as metered dose inhalers for delivery of steroids.
- the invention is particularly related to certain 20-ketosteroids that have been found to be highly susceptible to chemical degradation when formulated as solution aerosol products, and provides a way of enhancing chemical stability of such steroids.
- Structure I shown below is a typical core structure for a large number of natural and synthetic 20-ketosteroids, with the standard IUPAC numbering system of the carbon positions 1 to 21 indicated.
- These types of steroids have many well-known therapeutic uses, especially based upon their anti-inflammatory activity. It is often desirable to deliver such steroids topically using aerosol spray devices, such as metered dose inhalers (MDIs).
- MDIs are commonly used to deliver steroids, e.g., beclomethasone dipropionate, to the airways of patients via oral or nasal inhalation for the treatment of asthma and allergic rhinitis.
- HFC non-CFC hydrofluorocarbon
- solution formulations are disclosed in U.S. 5,766,573 which are surprisingly chemically stable in propellant HFC 134a and/or 227 and ethanol in a conventional aluminum canister.
- ciclesonide formulations are surprisingly chemically stable in certain solution MDI formulations disclosed in WO 98/52542.
- Solution formulations of flunisolide are disclosed in U.S.
- WO 98/13031 discloses recent work on reformulating budesonide as a non-CFC suspension formulation.
- suspension formulations of a medicament are more likely to encounter problems with physical instability (e.g., agglomeration , crystal growth and deposition on the container wall, all resulting in inconsistent dosage delivery).
- a drug delivery device providing medicinal steroid solution formulations with enhanced chemical stability would offer some significant advantages over suspension formulations. Besides homogeneity, solution formulations have been found in some cases ⁇ e.g., using HFC propellants and low ethanol content--to give dramatically higher respirable fractions (i.e., the percentage of active ingredient able to reach the airways of the lung) compared to a particulate suspension of the steroid drug. See U.S. 5,776,432. Furthermore, an aerosol product providing a chemically and physically stable aerosol steroid formulation using non-CFC propellant would offer the advantage of being more ozone friendly than currently available aerosol products with CFCs.
- Patent publications WO9947195 and WO0030608 form part of the state of the art pursuant to Art. 54(3) EPC relevant to the assessment of novelty as far as they are entitled to the priority dates claimed.
- WO9947195 does not mention that a medicinal aerosol steroid formulation can be chemically stabilized by use of an inert coating onto the surface of a pressurized canister.
- MI98A002559 the first priority document of WO0030608, describes aerosol formulations of budesonide or combination of budesonide with ipratropium bromide, salbutamol or formoterol, comprising a hydrofluoroalkane propellant containing no chlorine and an alcohol as co-solvent.
- the formulation is stored in a pressurized metered dose inhaler made of aluminium or tin plate with interior surfaces of stainless steel, anodised aluminium or inert organic coating in order to improve chemical stability of the active agent.
- Patent publication US5776433 is directed to a flunisolide formulation which may be delivered by metered dose inhaler with a canister that is inert to flunisolide.
- 20-ketosteroid having an OH group at the C-17 or C-21 position, or both, and being other than flunisolide may be chemically stabilized during storage in a metered dose inhaler formulation by use of an inert coating onto the inert surface of a pressurized canister.
- C-17/21 OH 20-ketosteroids those steroids in particular having a C-20 ketone and an OH group at the C-17 position or, especially, the C-21 position or both (hereafter collectively referred to as "C-17/21 OH 20-ketosteroids") are subject to enhanced chemical degradation when stored in contact with a metal container (particularly the metal oxide e.g., Al 2 O 3 layer that forms on the interior surface of the container).
- a metal container particularly the metal oxide e.g., Al 2 O 3 layer that forms on the interior surface of the container.
- the vast majority of MDI's on the market, including all MDI steroid products use aerosol containers made of metal, usually aluminum. By utilizing an aerosol container having a non-metal interior surface it is possible to produce solution aerosol formulations of C-17/21 OH 20-ketosteroids having enhanced chemical stability.
- a process for making a chemically stable steroid solution aerosol product by filling into a container a medicinal aerosol formulation comprising a dissolved 20-ketosteroid other than flunisolide and having an OH group
- the present invention is especially preferred with respect to C-21 OH 20-ketosteroids (with or without a C-17 OH group).
- the C-21 OH group can substantially increase biological activity of a steroid, but such steroids are also much more susceptible to chemical degradation in the presence of metal.
- Particularly preferred 20-ketosteroids are budesonide, triamcinolone acetonide, dexamethasone, and betamethasone 17-valerate, all of which have an OH group at the C-21 position.
- These steroids are all currently on the market as CFC particulate suspension formulations in metered dose inhalers and, at least in the case of budesonide, work has been conducted to reformulate this important steroid as a non-CFC suspension product in HFA propellants. See PCT published application WO98/13031.
- the most preferred type of container for use in the present invention is a conventional aluminum (or aluminum alloy) aerosol canister, but with an interior coating of an inert material, such as a spray-coated, baked epoxy-phenolic lacquer (available from Cebal Printal U.K. Ltd.).
- an inert material such as a spray-coated, baked epoxy-phenolic lacquer (available from Cebal Printal U.K. Ltd.).
- Other metals, such as stainless steel may likewise be used with an inert interior coating.
- the internal surfaces of metal valve components in contact with the formulation are similarly coated with an inert material.
- Another preferred coating for the inside of the container is perfluoroethylenepropylene (FEP).
- a preferred coating for the metal valve components is a very thin layer of glass, or other material, deposited by gas vapor deposition. Such coating is preferably used on all of the metal valve components in contact with the formulation, including the inside and outside of the metering chamber, inside and outside of the bottle emptier (if any), and the inside and outside of the valve stem (if metal), and may also be used to coat the inside of the canister.
- the preferred such coating technique is the SilcosteelTM process available from Restek Corporation, Bellefonte, PA.
- the SilcosteelTM aspect of the invention is useful even outside the context of the chemical degradation problem, for both solution and suspension formulations.
- Preferred formulations use a liquified propellant such as hydrogen-containing (non-CFC) propellants, more preferably hydrofluorocarbons, such as 134a and/or 227.
- a liquified propellant such as hydrogen-containing (non-CFC) propellants, more preferably hydrofluorocarbons, such as 134a and/or 227.
- Particularly preferred formulations include about 0.1 to 0.5% C-17/21 OH 20-ketosteroid about 75 to 99% 134a and/or 227, and about 1 to 25 % w/w ethanol, more preferably about 80 to 95% 134a and/or 227, and about 5 to 20% ethanol.
- the most preferred medicinal aerosol products according to the invention are MDI's comprising about 0.1 to 0.5% budesonide or triamcinolone acetonide dissolved in about 80-95% 134a and/or 227 and about 5-20% ethanol, contained in a coated aluminum aerosol canister equipped with a metering valve.
- the present invention provides a medicinal aerosol steroid solution formulation product with enhanced chemical stability.
- Such product includes a container equipped with a dispensing valve and containing a medicinal aerosol formulation having a 20-ketosteroid drug dissolved therein.
- the 20-ketosteroid is other than flunisolide and has an OH group at the C-17 or C-21 position or both, and the container is provided with a non-metal interior surface so as to reduce chemical degradation.
- the invention further provides a process for making a chemically stable 20-ketosteroid solution aerosol product, by filling into a container an aerosol formulation comprising a dissolved 20-ketosteroid other than flunisolide, said 20-ketosteroid having an OH group at the C-17 position or C-21 position or both, and said container having an inert non-metal interior surface so as to avoid chemical degradation of the 20-ketosteroid due to interaction with the container.
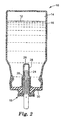
- a medicinal aerosol device 10 comprising a pressurizable metal aerosol container 16 equipped with a metering valve 18.
- the metal container 16 is preferably made of aluminum (i.e. aluminum or aluminum alloy) and has an inert interior coating layer 14.
- the metering valve 18 includes a metal metering chamber 20 with a coating layer 22.
- a plastic valve stem 24 instead of metal.
- Fig. 2 shows an alternative preferred embodiment that is essentially the same as Fig. 1, but utilizes a fixed bottle emptier 26, with coating layer 28. Also, a solution gasket 30 is used to further prevent contact of the formulation with metal components.
- the medicinal aerosol formulation 12 preferably includes a non-CFC propellant, a cosolvent (if necessary), and a therapeutically effective amount of dissolved C-17/21 OH 20-ketosteroid.
- a therapeutically effective amount will nomally be a concentration so as to provide in the range of about 100 to 1500 mg per day using one to eight puffs.
- Preferred propellants include hydrogen containing propellants, such as HFCs 134a and/or 227.
- Ethanol is a preferred cosolvent, although other cosolvents and solubilization aids (e.g., surfactants) may be used.
- the amount of cosolvent used is preferably an amount sufficient to completely dissolve the 20-ketosteroid.
- the formulation 12 may also include other excipients, such as stabilizers, antioxidants, flavoring agents, and the like.
- R 1 is H or an alkyl group
- R 2 is an alkyl group
- X is not H.
- R 1 and/or R 2 alkyl groups are methyl, ethyl, propyl, butyl, pentyl, and hexyl groups, including their branched and cyclic isomers.
- R 1 may form a ring with R 2 , preferably a cyclopentyl or cyclohexyl ring.
- Examples of known 20-ketosteroids according to structure V include budesonide, triamcinolone acetonide, desonide, and fluocinolone acetonide.
- Q is OH, H, Cl, or PO(ONa) 2 ;
- X is H, Cl, or F;
- Y is H, F, or Me;
- Z is H or Cl;
- R 1 is H, OH or propionate provided that when Q is not OH, then R 1 must be OH; and
- R 2 is H, OH, or Me.
- Examples of known 20-ketosteroids according to structure VI include alclometasone, beclomethasone, beclomethasone 17-monopropionate, betamethasone, betamethasone 17-valerate, clocortolone, desoximetasone, dexamethasone, dexamethasone sodium phosphate, dexamethasone 21-isonicotinate, diflorasone, flumethasone, methylprednisolone, paramethasone, prednisolone, tramcinolone, clobetasol, and fluorometholone.
- C-17/21 OH 20-ketosteroids for particular structural examples, four preferred C-17/21 OH 20-ketosteroids according to the present invention, budesonide, triamcinolone acetonide, betamethasone 17-valerate, and dexamethasone, have the following structures:
- the C-17/21 OH 20-ketosteroid is dissolved in the aerosol formulation and preferably contained in an inertly coated metal container.
- coated simply refer to a non-metal interior coating that does not promote degradation at the OH substituents on C-17/21 OH 20-ketosteroids.
- Inert coating materials include any suitable polymer, lacquer, resin, or other coating treatment that creates a barrier to chemical interaction of the dissolved C-17/21 OH 20-ketosteroid and metal on the container (especially metal oxides).
- suitable interior coatings include epoxy-phenolic resins, epoxy-urea-formaldehyde resins, polytetrafluroethylene (PTFE), perfluoroethylenepropylene (FEP), perfluoroalkoxyalkane (PFA), ethylene tetrafluoroethylene (ETFE), poly(vinyldiene fluoride) (PVDF), and chlorinated ethylene tetrafluoroethylene.
- PTFE polytetrafluroethylene
- FEP perfluoroethylenepropylene
- PFA perfluoroalkoxyalkane
- ETFE ethylene tetrafluoroethylene
- PVDF poly(vinyldiene fluoride)
- Chinated ethylene tetrafluoroethylene tetrafluoroethylene
- Blends may be used of fluorinated polymers with non-fluorinated polymers such as polyamides, polyimides, polyethersulfones, polyphenylene sulfides, and amine
- Specific blends include PTFE/FEP/polyamideimide, PTFE/polyether sulphone (PES) and FEP-benzoguanamine.
- Preferred interior coating materials are epoxy-phenolic resins, epoxy-urea-formaldehyde resins, PTFE, and FEP. Additional information regarding interior can coatings is taught in, e.g., EP 642992, WO 96/32099, WO 96/32150, WO 96/32151, and WO 96/32345, which disclose interior can coatings for drug suspension formulation products.
- a preferred coating for the metal valve components is a very thin layer of fused silica glass, or other material, deposited by gas vapor deposition. Such coating is preferably used on all of the metal valve components in contact with the formulation, including the inside and outside of the metering chamber, inside and outside of the bottle emptier (if any), and the inside and outside of the valve stem (if metal).
- the preferred such coating technique is the SilcosteelTM process available from Restek Corporation, Bellefonte, PA. This process deposits a submicron layer of fused silica glass on the metal components and can be used both on the valve components and on the interior of the canister.
- the SilcosteelTM aspect of the invention is thus useful even outside the context of the chemical degradation problem, for both solution and suspension formulations.
- the Silcosteel process is performed at a temperature of 400°C, which has the added benefit of thermally removing residual oils on the metal surface.
- containers made from non-metal materials such as glass or plastic (e.g., polyethylene terephthalate, from Precise Plastic Ltd., U.K.).
- glass containers it is preferred to use glass having a low metal oxide content.
- ASTM American Society for Testing and Materials
- Type I/Class A glass contains 2% aluminum oxide
- Type 11/Class B glass contains 7% aluminum oxide. The former is therefore preferred.
- Type III (soda-lime) glass vials may also be used (available from Wheaton Coated Products).
- High metal oxide content glass should also be avoided during development and testing of formulations containing C-17 and/or C-21 OH 20-ketosteroids because such use could cause unsuspected chemical degradation due to the glass metal oxide content.
- Coated containers can be made by pre-coating the metal roll stock before forming the container or by coating the container after it is made, by various techniques known in the art of coating.
- suitable coating procedures include plasma coating, electrostatic dry powder coating, impregnating/spraying, hard anodization with polymer deposition, chemical vapor deposition (CVD), physical vapor deposition (PVD), as well as other procedures known in the art for this purpose.
- the containers are coated with an epoxy resin and then baked.
- Suitable coated containers can be obtained from, for example, Cebal Printal U.K. Ltd.
- Preferred products/devices according to the present invention are pressurized aerosols such as MDIs that use liquefied gas propellants, including chlorofluorocarbons (CFCs), hydrofluorocarbons (HFCs), fluorocarbons (FCs), hydrocarbons (HCs), hydrochlorofluorocarbons (HCFCs), and dimethyl ether (DME).
- CFCs chlorofluorocarbons
- HFCs hydrofluorocarbons
- FCs fluorocarbons
- HCs hydrocarbons
- HCFCs hydrochlorofluorocarbons
- DME dimethyl ether
- Propellants containing hydrogen are preferred.
- Ethanol may also be included to assist in solubilizing the 20-ketosteroid, preferably in an amount of about 1-25%.
- Formulations comprising dissolved C-17/21 OH 20-ketosteroid, HFC propellant, such as 134a and/or 227, with 5-20% ethanol are particularly preferred
- the formulations are essentially non-aqueous, meaning that they do not include added water, although very small amounts of water may be present due to water ingress and/or as a residual in formulation components (such as ethanol).
- Formulations will preferably have less than about 3%, and more preferably less than about 0.5 % water content.
- the containers can be equipped with any suitable conventional or unconventional dispensing valve, preferably a metered dose valve.
- Example 1 illustrates the effect of aluminum oxide in the degradation of another C-17/21 OH 20-ketosteroid, in this case triamcinolone acetonide, which has an OH attached to the C-21 carbon position.
- Example illustrates the effect of aluminum oxide in the degradation of C-17/21 OH 20-ketosteroid, in the case dexamethasone, which has one OH attached to C-21 and another OH attached to C-17.
- dexamethasone At ambient temperature, a 3 mL ethanol solution of dexamethasone (0.007 M) was mixed with 0.1 g of aluminum oxide (Aldrich, neutral, activated Brockmann I, 150 mesh), which mimics aluminum oxide existing on the inner surface of aluminum containers. The mixture was held at 75°C for several hours, and an aliquot was taken at each of the time intervals given in Table 3. Each aliquote was filtered with a syringe filter and diluted with acetonitrile by 20 times for HPLC analysis to determine the recoveries of dexamethasone with respect to the inital concentration. The recoveries of dexamethasone were compared with a reference, which contained the same concentration of dexamethasone but in ethanol solution without aluminum oxide.
- aluminum oxide Aldrich, neutral, activated Brockmann I, 150 mesh
- Example 5 and 6 were manufactured on a pilot-scale cold filler and filled into metered dose inhaler vials having various different components: different valve types, coated versus uncoated aluminum cans, and with and without a nylon desiccant insert. The units were then inverted and stored at either 40°C/85% RH for 14 days or 30°C/30% RH for 17 days prior to analysis of budesonide degradation.
- valve used also had an impact on chemical stability.
- the greatest chemical stability enhancement was found in units using a Bespak 357 valve with EPDM seals, a Cebal epoxy lacquered can, and the formulation of Example 6. Nearly as stable were units equipped with a 3M Neotechnic SpraymiserTM HFA valve, a Cebal epoxy lacquered can, and the formulations of Examples 5 or 6. Also, none of the valves tested were coated and it is expected that this would further enhance chemical stability.
- Formulations using propellant 227 appeared to be slightly more stable. Containers having a desiccant generally exhibited reduced degradation compared to those without desiccant inserts. This suggests that water may increase degradation and that using dried ethanol may thus help further enhance chemical stability. Also, the use of certain antioxidants such as ascorbic acid and ascorbyl palmitate (but not vitamin E) appeared to enhance chemical stability, while the use of oleic acid appeared to reduce chemical stability.
- Graph A shows comparative tests results on a budesonide solution formulation comprising 0.22% w/w budesonide, 11 % w/w ethanol, and the remainder 134a.
- Three lots were stored at 40°C/75% RH.
- One lot (identified as "Coated A") was contained in aluminum cans coated with an epoxy-phenolic coating (Cebal Printal Ltd. U.K.) and capped with a blind ferrule and a continuous gasket (made of DFDB 1085 elastomer, Union Carbide) so as to completely isolate the formulation from contact with metal surfaces.
- Coated B Another lot (“Coated B”) was contained in the same type of cans as Coated A, but equipped with a functional valve ferrule (3M Neotechnic SpraymiserTM solution valve) having a solution gasket to partially prevent contact of the formulation with the underside of the valve ferrule.
- a third lot (“Uncoated”) was contained in uncoated aluminum cans equipped with the same valves as for Coated B.
- Example 8 the formulation contained 0.22% w/w budesonide dissolved in a mixture of 134a and 13% w/w ethanol.
- the Example 9 formulation had 0.17% w/w budesonide dissolved in 134a and 15% w/w ethanol. In both cases, shown in Graphs B and C, respectively, the degradation rate was much greater in the Uncoated can, and degradation was least for Coated A.
- Table 6 Container Closure System Initial Testing* 1 month @40°C/75%RH Total Impurities %w/w Total Impurities %w/w UV/Epoxy lot A 1.09 0.98 CV/FEP lot B 1.17 1.36 UV/FEP lot C 1.36 2.31 CV/Epoxy lot D 1.17 0.95 Table 7.
- the data indicates the epoxy can is superior to the FEP in preventing degradation. Without wishing to be held to any particular mechanism to explain the difference in results between the two coating types, it is hypothisized that it is not that the phenolic epoxy provides a better barrier, but rather that some adsorption of degradation products is taking place. Essentially, the phenolic epoxy may be "soaking up" the degradates. A small amount of budesonide may also be adsorbed too, but the amount is apparently too small to affect the target content amount.
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Engineering & Computer Science (AREA)
- Epidemiology (AREA)
- Dispersion Chemistry (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Mechanical Engineering (AREA)
- Pulmonology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Organic Chemistry (AREA)
- Otolaryngology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Medicinal Preparation (AREA)
- Cosmetics (AREA)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP06005429A EP1693053B1 (en) | 1999-06-18 | 2000-06-15 | Steroid solution aerosol products with enhanced chemical stability |
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US13996199P | 1999-06-18 | 1999-06-18 | |
| US139961P | 1999-06-18 | ||
| PCT/US2000/016462 WO2000078286A1 (en) | 1999-06-18 | 2000-06-15 | Steroid solution aerosol products with enhanced chemical stability |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP06005429A Division EP1693053B1 (en) | 1999-06-18 | 2000-06-15 | Steroid solution aerosol products with enhanced chemical stability |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| EP1194121A1 EP1194121A1 (en) | 2002-04-10 |
| EP1194121B1 true EP1194121B1 (en) | 2006-03-22 |
Family
ID=22489103
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP00941435A Expired - Lifetime EP1194121B1 (en) | 1999-06-18 | 2000-06-15 | Steroid solution aerosol products with enhanced chemical stability |
| EP06005429A Expired - Lifetime EP1693053B1 (en) | 1999-06-18 | 2000-06-15 | Steroid solution aerosol products with enhanced chemical stability |
Family Applications After (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP06005429A Expired - Lifetime EP1693053B1 (en) | 1999-06-18 | 2000-06-15 | Steroid solution aerosol products with enhanced chemical stability |
Country Status (8)
| Country | Link |
|---|---|
| US (4) | US6315985B1 (enExample) |
| EP (2) | EP1194121B1 (enExample) |
| JP (1) | JP4777560B2 (enExample) |
| AT (2) | ATE523190T1 (enExample) |
| AU (1) | AU777505B2 (enExample) |
| CA (1) | CA2371931A1 (enExample) |
| DE (1) | DE60026840T2 (enExample) |
| WO (1) | WO2000078286A1 (enExample) |
Families Citing this family (76)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20010031244A1 (en) * | 1997-06-13 | 2001-10-18 | Chiesi Farmaceutici S.P.A. | Pharmaceutical aerosol composition |
| DZ2947A1 (fr) * | 1998-11-25 | 2004-03-15 | Chiesi Farma Spa | Inhalateur à compteur de dose sous pression. |
| US6315985B1 (en) | 1999-06-18 | 2001-11-13 | 3M Innovative Properties Company | C-17/21 OH 20-ketosteroid solution aerosol products with enhanced chemical stability |
| IT1313553B1 (it) | 1999-07-23 | 2002-09-09 | Chiesi Farma Spa | Formulazioni ottimizzate costituite da soluzioni di steroidi dasomministrare per inalazione. |
| WO2001019342A2 (en) * | 1999-09-11 | 2001-03-22 | Glaxo Group Limited | Pharmaceutical formulation of fluticasone propionate |
| JP2003518484A (ja) * | 1999-12-24 | 2003-06-10 | グラクソ グループ リミテッド | サルメテロールおよびプロピオン酸フルチカゾンの医薬用エアゾル製剤 |
| IT1317846B1 (it) | 2000-02-22 | 2003-07-15 | Chiesi Farma Spa | Formulazioni contenenti un farmaco anticolinergico per il trattamentodella broncopneumopatia cronica ostruttiva. |
| IT1318514B1 (it) * | 2000-05-12 | 2003-08-27 | Chiesi Farma Spa | Formulazioni contenenti un farmaco glucocorticosteroide per iltrattamento di patologie broncopolmonari. |
| BR0015884A (pt) * | 2000-05-22 | 2003-07-08 | Chiesi Farma Spa | Formulações de soluções farmacêuticas estáveis para inaladores de dose medida pressurizados |
| TW523409B (en) * | 2000-09-15 | 2003-03-11 | Baxter Int | Container for inhalation anesthetic |
| US20040089293A1 (en) * | 2000-10-13 | 2004-05-13 | Paul Johnson | Medicament dispenser |
| GB0025092D0 (en) * | 2000-10-13 | 2000-11-29 | Glaxo Group Ltd | Medicament dispenser |
| US20020085978A1 (en) * | 2000-11-10 | 2002-07-04 | Mina Buenafe | Degradation-resistant glucocorticosteroid formulations |
| DE10062564A1 (de) * | 2000-12-15 | 2002-06-20 | Linde Ag | Schutzgas und Verfahren zum Lichtbogenschweißen |
| EP1241113A1 (en) | 2001-03-12 | 2002-09-18 | CHIESI FARMACEUTICI S.p.A. | Inhaler with means for improving chemical stability of medicinal aerosol solution contained therein |
| GB0106046D0 (en) * | 2001-03-12 | 2001-05-02 | Glaxo Group Ltd | Canister |
| ATE267591T1 (de) * | 2001-07-02 | 2004-06-15 | Chiesi Farma Spa | Optimierte tobramycin-formulierung zur aerosolbildung |
| GB0122725D0 (en) * | 2001-09-21 | 2001-11-14 | Glaxo Group Ltd | Drug dispensing components |
| EP1458354A1 (en) * | 2001-12-21 | 2004-09-22 | 3M Innovative Properties Company | Medicinal aerosol formulations comprising ion pair complexes |
| KR100947409B1 (ko) * | 2002-03-01 | 2010-03-12 | 키에시 파르마슈티시 엣스. 피. 에이. | 포모테롤 초미세 조성물 |
| US7344707B2 (en) * | 2002-05-15 | 2008-03-18 | The Procter & Gamble Company | Low combustion aerosol products in plastic packages having a reduced fire hazard classification that subsequently reduces storage costs |
| US20030215400A1 (en) * | 2002-05-15 | 2003-11-20 | The Procter & Gamble Company | Pressurized package made of a polyamide resin and containing dimethyl ether |
| JP3691459B2 (ja) * | 2002-06-14 | 2005-09-07 | 久光メディカル株式会社 | 粉末状吸入剤組成物 |
| ATE470468T1 (de) * | 2003-08-04 | 2010-06-15 | Alexza Pharmaceuticals Inc | Substrate für eine medikamentenverabreichungsvorrichtung und verfahren zur bereitung |
| AU2004266641A1 (en) | 2003-08-12 | 2005-03-03 | 3M Innovative Properties Company | Oxime substituted imidazo-containing compounds |
| EP1658076B1 (en) | 2003-08-27 | 2013-03-06 | 3M Innovative Properties Company | Aryloxy and arylalkyleneoxy substituted imidazoquinolines |
| US20050054665A1 (en) | 2003-09-05 | 2005-03-10 | 3M Innovative Properties Company | Treatment for CD5+ B cell lymphoma |
| US7544697B2 (en) | 2003-10-03 | 2009-06-09 | Coley Pharmaceutical Group, Inc. | Pyrazolopyridines and analogs thereof |
| ES2544477T3 (es) | 2003-10-03 | 2015-08-31 | 3M Innovative Properties Company | Imidazoquinolinas sustituidas con alcoxi |
| AR041873A1 (es) | 2003-10-30 | 2005-06-01 | Pablo Cassara Srl Lab | Una formulacion farmaceutica en aerosol adecuada para la inhalacion oral o nasal que contienen glucocorticoides en solucion estable al almacenamiento; un metodo para estabilzar formulaciones y uso de un agente estabilizante |
| US7897767B2 (en) | 2003-11-14 | 2011-03-01 | 3M Innovative Properties Company | Oxime substituted imidazoquinolines |
| JP2007511535A (ja) | 2003-11-14 | 2007-05-10 | スリーエム イノベイティブ プロパティズ カンパニー | ヒドロキシルアミン置換イミダゾ環化合物 |
| RU2409576C2 (ru) | 2003-11-25 | 2011-01-20 | 3М Инновейтив Пропертиз Компани | Системы, содержащие имидазольное кольцо с заместителями, и способы их получения |
| US20050121025A1 (en) | 2003-12-04 | 2005-06-09 | Gamard Stephan C.F. | Portable gas operating inhaler |
| WO2005066170A1 (en) | 2003-12-29 | 2005-07-21 | 3M Innovative Properties Company | Arylalkenyl and arylalkynyl substituted imidazoquinolines |
| CA2551399A1 (en) | 2003-12-30 | 2005-07-21 | 3M Innovative Properties Company | Imidazoquinolinyl, imidazopyridinyl, and imidazonaphthyridinyl sulfonamides |
| EP1595531A1 (en) | 2004-05-13 | 2005-11-16 | CHIESI FARMACEUTICI S.p.A. | Stable pharmaceutical solution formulations for pressurized metered dose inhalers |
| EP1730143A2 (en) | 2004-03-24 | 2006-12-13 | 3M Innovative Properties Company | Amide substituted imidazopyridines, imidazoquinolines, and imidazonaphthyridines |
| WO2005123080A2 (en) | 2004-06-15 | 2005-12-29 | 3M Innovative Properties Company | Nitrogen-containing heterocyclyl substituted imidazoquinolines and imidazonaphthyridines |
| WO2006038923A2 (en) | 2004-06-18 | 2006-04-13 | 3M Innovative Properties Company | Aryl substituted imidazonaphthyridines |
| WO2006009826A1 (en) | 2004-06-18 | 2006-01-26 | 3M Innovative Properties Company | Aryloxy and arylalkyleneoxy substituted thiazoloquinolines and thiazolonaphthyridines |
| WO2006065280A2 (en) | 2004-06-18 | 2006-06-22 | 3M Innovative Properties Company | Isoxazole, dihydroisoxazole, and oxadiazole substituted imidazo ring compounds and methods |
| CA2592904C (en) | 2004-12-30 | 2015-04-07 | 3M Innovative Properties Company | Chiral fused [1,2]imidazo[4,5-c] ring compounds |
| AU2005326708C1 (en) | 2004-12-30 | 2012-08-30 | 3M Innovative Properties Company | Substituted chiral fused [1,2]imidazo[4,5-c] ring compounds |
| US9248127B2 (en) | 2005-02-04 | 2016-02-02 | 3M Innovative Properties Company | Aqueous gel formulations containing immune response modifiers |
| WO2006086634A2 (en) | 2005-02-11 | 2006-08-17 | Coley Pharmaceutical Group, Inc. | Oxime and hydroxylamine substituted imidazo[4,5-c] ring compounds and methods |
| EP1850831B1 (en) * | 2005-02-25 | 2015-12-02 | CHIESI FARMACEUTICI S.p.A. | Pharmaceutical aerosol formulations for pressurized metered dose inhalers comprising a sequestering agent |
| US7943610B2 (en) | 2005-04-01 | 2011-05-17 | 3M Innovative Properties Company | Pyrazolopyridine-1,4-diamines and analogs thereof |
| AU2006232375A1 (en) | 2005-04-01 | 2006-10-12 | Coley Pharmaceutical Group, Inc. | 1-substituted pyrazolo (3,4-c) ring compounds as modulators of cytokine biosynthesis for the treatment of viral infections and neoplastic diseases |
| US8277780B2 (en) * | 2005-05-27 | 2012-10-02 | Taro Pharmaceutical North America, Inc. | Stable liquid desoximethasone compositions with reduced oxidized impurity |
| US20090050143A1 (en) * | 2006-03-24 | 2009-02-26 | Boardman Larry D | Medicinal formulation container with a treated metal surface |
| US20070231396A1 (en) * | 2006-03-29 | 2007-10-04 | Ray Charles D | Medication spray formulation |
| US20070286814A1 (en) * | 2006-06-12 | 2007-12-13 | Medispray Laboratories Pvt. Ltd. | Stable aerosol pharmaceutical formulations |
| GB0613761D0 (en) * | 2006-07-11 | 2006-08-23 | Norton Healthcare Ltd | A pharmaceutical formulation and method for the preparation thereof |
| US7906506B2 (en) | 2006-07-12 | 2011-03-15 | 3M Innovative Properties Company | Substituted chiral fused [1,2] imidazo [4,5-c] ring compounds and methods |
| MX2009005359A (es) * | 2006-11-20 | 2009-06-08 | Cipla Ltd | Dispositivo en aerosol. |
| WO2008112661A2 (en) | 2007-03-09 | 2008-09-18 | Alexza Pharmaceuticals, Inc. | Heating unit for use in a drug delivery device |
| MX2010005036A (es) * | 2007-11-07 | 2010-05-27 | Astrazeneca Ab | Formulaciones de polvo seco que comprenden derivados del acido ascorbico. |
| US8227027B2 (en) * | 2007-12-07 | 2012-07-24 | Presspart Gmbh & Co. Kg | Method for applying a polymer coating to an internal surface of a container |
| US20090208424A1 (en) * | 2008-02-19 | 2009-08-20 | Todd Maibach | Compositions and methods for delivery of solution to the skin |
| US9278048B2 (en) * | 2009-05-06 | 2016-03-08 | Baxter International, Inc. | Pharmaceutical product and method of use |
| US20120272951A1 (en) | 2009-12-16 | 2012-11-01 | 3M Innovative Properties Company | Formulations and methods for controlling mdi particle size delivery |
| RS54037B1 (sr) * | 2010-10-12 | 2015-10-30 | Ivax Pharmaceuticals Ireland | Uređaj za nazalni sprej |
| US20120204871A1 (en) * | 2011-02-10 | 2012-08-16 | Julio Cesar Vega | Stable, non-corrosive formulations for pressurized metered dose inhalers |
| DE102014112595A1 (de) * | 2014-09-02 | 2016-03-03 | Reinhard Caliebe | Dose für ein medizinisches, pharmazeutisches oder kosmetisches Fluid |
| EP3746138B1 (en) | 2018-02-02 | 2025-12-10 | Alexza Pharmaceuticals, Inc. | Electrical condensation aerosol device |
| CN111939143A (zh) * | 2019-05-16 | 2020-11-17 | 鲁南制药集团股份有限公司 | 一种布地奈德溶液型气雾剂及其制备方法 |
| US12178979B2 (en) | 2021-10-13 | 2024-12-31 | Satio, Inc. | Dermal patch for delivering a pharmaceutical |
| US12214346B2 (en) | 2021-10-13 | 2025-02-04 | Satio, Inc. | Dermal patch with a diagnostic test strip |
| US12053284B2 (en) | 2021-11-08 | 2024-08-06 | Satio, Inc. | Dermal patch for collecting a physiological sample |
| US12023156B2 (en) | 2021-10-13 | 2024-07-02 | Satio, Inc. | Dermal patch for collecting a physiological sample |
| US12048543B2 (en) | 2021-11-08 | 2024-07-30 | Satio, Inc. | Dermal patch for collecting a physiological sample with removable vial |
| US11877848B2 (en) | 2021-11-08 | 2024-01-23 | Satio, Inc. | Dermal patch for collecting a physiological sample |
| US11452474B1 (en) | 2021-04-14 | 2022-09-27 | Satio, Inc. | Dual lever dermal patch system |
| US11964121B2 (en) | 2021-10-13 | 2024-04-23 | Satio, Inc. | Mono dose dermal patch for pharmaceutical delivery |
| US20250197609A1 (en) | 2023-12-13 | 2025-06-19 | Blue Evolution, Inc. | Ecofriendly, biodegradable biological polysaccharide composition for packaging materials |
Family Cites Families (62)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US577433A (en) * | 1897-02-23 | Door-alarm | ||
| BE555319A (enExample) * | 1956-03-21 | 1900-01-01 | ||
| US3361306A (en) * | 1966-03-31 | 1968-01-02 | Merck & Co Inc | Aerosol unit dispensing uniform amounts of a medically active ingredient |
| IT1034011B (it) * | 1969-06-26 | 1979-09-10 | Vister Vismara Terapeutici S P | Processo per la prapratzione di 17 monoesteri di 17 a 21 diossisteroidi per idrolisi die corpispondenti 17 21 ortoesteri ciclici a ph control lato |
| US3622053A (en) * | 1969-12-10 | 1971-11-23 | Schering Corp | Aerosol inhaler with flip-up nozzle |
| US3923484A (en) * | 1974-01-11 | 1975-12-02 | Corning Glass Works | Flame method of producing glass |
| US4185100A (en) * | 1976-05-13 | 1980-01-22 | Johnson & Johnson | Topical anti-inflammatory drug therapy |
| US4499108A (en) * | 1983-06-08 | 1985-02-12 | Schering Corporation | Stable pleasant-tasting albuterol sulfate pharmaceutical formulations |
| GB8334494D0 (en) * | 1983-12-24 | 1984-02-01 | Tanabe Seiyaku Co | Carbostyril derivatives |
| IT1196142B (it) * | 1984-06-11 | 1988-11-10 | Sicor Spa | Procedimento per la preparazione di 16,17-acetali di derivati pregnanici e nuovi composti ottenuti |
| US5192528A (en) * | 1985-05-22 | 1993-03-09 | Liposome Technology, Inc. | Corticosteroid inhalation treatment method |
| EP0338670B1 (en) | 1988-03-22 | 1994-05-11 | FISONS plc | Pharmaceutical Compositions |
| US5225183A (en) * | 1988-12-06 | 1993-07-06 | Riker Laboratories, Inc. | Medicinal aerosol formulations |
| US5766573A (en) | 1988-12-06 | 1998-06-16 | Riker Laboratories, Inc. | Medicinal aerosol formulations |
| MX9203481A (es) | 1990-10-18 | 1992-07-01 | Minnesota Mining & Mfg | Formulaciones. |
| US6006745A (en) * | 1990-12-21 | 1999-12-28 | Minnesota Mining And Manufacturing Company | Device for delivering an aerosol |
| WO1993004718A1 (de) * | 1991-08-29 | 1993-03-18 | Sroka, Peter-Christian | Medizinisches gerät zur inhalation von dosier-aerosolen |
| DE4230876A1 (de) * | 1992-03-17 | 1993-09-23 | Asta Medica Ag | Druckgaspackungen unter verwendung von polyoxyethylen-glyceryl-oleaten |
| WO1994013263A1 (en) * | 1992-12-09 | 1994-06-23 | Jager Paul D | Stabilized medicinal aerosol solution formulations |
| SE9203743D0 (sv) * | 1992-12-11 | 1992-12-11 | Astra Ab | Efficient use |
| US6596260B1 (en) | 1993-08-27 | 2003-07-22 | Novartis Corporation | Aerosol container and a method for storage and administration of a predetermined amount of a pharmaceutically active aerosol |
| CA2178473C (en) | 1993-12-20 | 2004-08-24 | Tsi-Zong Tzou | Flunisolide aerosol formulations |
| US5653961A (en) * | 1995-03-31 | 1997-08-05 | Minnesota Mining And Manufacturing Company | Butixocort aerosol formulations in hydrofluorocarbon propellant |
| BR9604976A (pt) * | 1995-04-14 | 1998-06-09 | Glaxo Wellcome Inc | Inalador de dose medida sistema inalador de dose medida e uso do mesmo |
| EP1908488A3 (en) * | 1995-04-14 | 2008-04-16 | SmithKline Beecham Corporation | Metered dose inhaler for salmeterol |
| DE69631476T2 (de) * | 1995-04-14 | 2005-01-13 | Smithkline Beecham Corp. | Vorrichtung zur dosierten inhalierung von beclomethason-diproprionat |
| WO1996032151A1 (en) * | 1995-04-14 | 1996-10-17 | Glaxo Wellcome Inc. | Metered dose inhaler for fluticasone propionate |
| JPH11507359A (ja) | 1995-06-07 | 1999-06-29 | ローヌ−プーラン ローラー ファーマシューティカルズ インコーポレイテッド | 安定化ステロイド組成物 |
| US5776573A (en) | 1996-04-16 | 1998-07-07 | Cd Magic, Inc. | Compact disc revitalizer formulations and revitalizer |
| US5743251A (en) | 1996-05-15 | 1998-04-28 | Philip Morris Incorporated | Aerosol and a method and apparatus for generating an aerosol |
| US6039932A (en) * | 1996-09-27 | 2000-03-21 | 3M Innovative Properties Company | Medicinal inhalation aerosol formulations containing budesonide |
| GB9620187D0 (en) | 1996-09-27 | 1996-11-13 | Minnesota Mining & Mfg | Medicinal aerosol formulations |
| AU726510B2 (en) | 1996-12-04 | 2000-11-09 | Consort Medical Plc | Pharmaceutical compositions and devices for their administration |
| US6413496B1 (en) * | 1996-12-04 | 2002-07-02 | Biogland Ireland (R&D) Limited | Pharmaceutical compositions and devices for their administration |
| US6126919A (en) * | 1997-02-07 | 2000-10-03 | 3M Innovative Properties Company | Biocompatible compounds for pharmaceutical drug delivery systems |
| US5891420A (en) * | 1997-04-21 | 1999-04-06 | Aeropharm Technology Limited | Environmentally safe triancinolone acetonide aerosol formulations for oral inhalation |
| US5891419A (en) * | 1997-04-21 | 1999-04-06 | Aeropharm Technology Limited | Environmentally safe flunisolide aerosol formulations for oral inhalation |
| US6120752A (en) | 1997-05-21 | 2000-09-19 | 3M Innovative Properties Company | Medicinal aerosol products containing formulations of ciclesonide and related steroids |
| US20010031244A1 (en) * | 1997-06-13 | 2001-10-18 | Chiesi Farmaceutici S.P.A. | Pharmaceutical aerosol composition |
| NZ504021A (en) * | 1997-10-17 | 2003-04-29 | Systemic Pulmonary Delivery Lt | Method and apparatus for delivering aerosolized medication having air discharged through air tube directly into plume of aerosolized medication |
| GB9805938D0 (en) | 1998-03-19 | 1998-05-13 | Glaxo Group Ltd | Valve for aerosol container |
| US6045784A (en) * | 1998-05-07 | 2000-04-04 | The Procter & Gamble Company | Aerosol package compositions containing fluorinated hydrocarbon propellants |
| US6451285B2 (en) * | 1998-06-19 | 2002-09-17 | Baker Norton Pharmaceuticals, Inc. | Suspension aerosol formulations containing formoterol fumarate and a fluoroalkane propellant |
| US6241969B1 (en) * | 1998-06-26 | 2001-06-05 | Elan Corporation Plc | Aqueous compositions containing corticosteroids for nasal and pulmonary delivery |
| DE19847969A1 (de) * | 1998-10-17 | 2000-04-20 | Boehringer Ingelheim Pharma | Lagerfähig flüssige Formulierung mit Formoterol |
| DK1131059T3 (da) * | 1998-11-13 | 2003-06-30 | Jago Res Ag | Tørpulver til inhalation |
| DZ2947A1 (fr) * | 1998-11-25 | 2004-03-15 | Chiesi Farma Spa | Inhalateur à compteur de dose sous pression. |
| IT1303788B1 (it) * | 1998-11-25 | 2001-02-23 | Chiesi Farma Spa | Formulazioni di aerosol medicinali. |
| US6261539B1 (en) * | 1998-12-10 | 2001-07-17 | Akwete Adjei | Medicinal aerosol formulation |
| US6290930B1 (en) * | 1998-12-18 | 2001-09-18 | Baker Norton Pharmaceuticals, Inc. | Pharmaceutical solution aerosol formulations containing fluoroalkanes and budesonide |
| US6004537A (en) * | 1998-12-18 | 1999-12-21 | Baker Norton Pharmaceuticals, Inc. | Pharmaceutical solution aerosol formulations containing fluoroalkanes, budesonide and formoterol |
| US6315985B1 (en) | 1999-06-18 | 2001-11-13 | 3M Innovative Properties Company | C-17/21 OH 20-ketosteroid solution aerosol products with enhanced chemical stability |
| US6322053B1 (en) * | 2000-02-08 | 2001-11-27 | Valiant Machine & Tool Inc. | Balancing system with a force multiplying device |
| IT1317846B1 (it) * | 2000-02-22 | 2003-07-15 | Chiesi Farma Spa | Formulazioni contenenti un farmaco anticolinergico per il trattamentodella broncopneumopatia cronica ostruttiva. |
| IT1318514B1 (it) * | 2000-05-12 | 2003-08-27 | Chiesi Farma Spa | Formulazioni contenenti un farmaco glucocorticosteroide per iltrattamento di patologie broncopolmonari. |
| BR0015884A (pt) * | 2000-05-22 | 2003-07-08 | Chiesi Farma Spa | Formulações de soluções farmacêuticas estáveis para inaladores de dose medida pressurizados |
| US20060257324A1 (en) * | 2000-05-22 | 2006-11-16 | Chiesi Farmaceutici S.P.A. | Pharmaceutical solution formulations for pressurised metered dose inhalers |
| EP1415647A1 (en) * | 2002-10-23 | 2004-05-06 | CHIESI FARMACEUTICI S.p.A. | "Long-acting beta-2 agonists ultrafine formulations" |
| EP1340492A1 (en) * | 2002-03-01 | 2003-09-03 | CHIESI FARMACEUTICI S.p.A. | Aerosol formulations for pulmonary administration of medicaments having systemic effects |
| KR100947409B1 (ko) * | 2002-03-01 | 2010-03-12 | 키에시 파르마슈티시 엣스. 피. 에이. | 포모테롤 초미세 조성물 |
| CA2388352A1 (en) * | 2002-05-31 | 2003-11-30 | Voiceage Corporation | A method and device for frequency-selective pitch enhancement of synthesized speed |
| EP1595531A1 (en) * | 2004-05-13 | 2005-11-16 | CHIESI FARMACEUTICI S.p.A. | Stable pharmaceutical solution formulations for pressurized metered dose inhalers |
-
2000
- 2000-06-13 US US09/592,885 patent/US6315985B1/en not_active Expired - Lifetime
- 2000-06-15 AU AU56142/00A patent/AU777505B2/en not_active Ceased
- 2000-06-15 AT AT06005429T patent/ATE523190T1/de not_active IP Right Cessation
- 2000-06-15 WO PCT/US2000/016462 patent/WO2000078286A1/en not_active Ceased
- 2000-06-15 EP EP00941435A patent/EP1194121B1/en not_active Expired - Lifetime
- 2000-06-15 AT AT00941435T patent/ATE320794T1/de not_active IP Right Cessation
- 2000-06-15 DE DE60026840T patent/DE60026840T2/de not_active Expired - Lifetime
- 2000-06-15 CA CA002371931A patent/CA2371931A1/en not_active Abandoned
- 2000-06-15 EP EP06005429A patent/EP1693053B1/en not_active Expired - Lifetime
- 2000-06-15 JP JP2001504351A patent/JP4777560B2/ja not_active Expired - Fee Related
-
2001
- 2001-10-04 US US09/970,746 patent/US6610273B2/en not_active Expired - Lifetime
-
2003
- 2003-07-30 US US10/630,414 patent/US20040033201A1/en not_active Abandoned
-
2005
- 2005-02-22 US US11/061,529 patent/US20050220717A1/en not_active Abandoned
Also Published As
| Publication number | Publication date |
|---|---|
| EP1693053A3 (en) | 2007-06-27 |
| CA2371931A1 (en) | 2000-12-28 |
| EP1693053A2 (en) | 2006-08-23 |
| AU5614200A (en) | 2001-01-09 |
| US20050220717A1 (en) | 2005-10-06 |
| ATE523190T1 (de) | 2011-09-15 |
| US20040033201A1 (en) | 2004-02-19 |
| DE60026840D1 (de) | 2006-05-11 |
| EP1693053B1 (en) | 2011-09-07 |
| US20020071810A1 (en) | 2002-06-13 |
| US6315985B1 (en) | 2001-11-13 |
| JP4777560B2 (ja) | 2011-09-21 |
| WO2000078286A1 (en) | 2000-12-28 |
| ATE320794T1 (de) | 2006-04-15 |
| JP2003502357A (ja) | 2003-01-21 |
| DE60026840T2 (de) | 2006-10-12 |
| US6610273B2 (en) | 2003-08-26 |
| AU777505B2 (en) | 2004-10-21 |
| EP1194121A1 (en) | 2002-04-10 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP1194121B1 (en) | Steroid solution aerosol products with enhanced chemical stability | |
| EP1787639B1 (en) | Stable pharmaceutical solution formulations for pressurised metered dose inhalers | |
| EP3515425B1 (en) | Pharmaceutical composition comprising salmeterol | |
| EP3787598B1 (en) | Pharmaceutical composition comprising salbutamol | |
| US7381402B2 (en) | Stable pharmaceutical solution formulations for pressurized metered dose inhalers | |
| US20040126325A1 (en) | Medicinal aerosol solution formulation products with improved chemical stability | |
| EP1368253B1 (en) | Inhaler with means for improving chemical stability of medicinal aerosol solution contained therein | |
| US8877164B2 (en) | Pharmaceutical aerosol formulations for pressurized metered dose inhalers comprising a sequestering agent | |
| US20070025920A1 (en) | Stable Pharmaceutical Solution Formulations for Pressurized Metered Dose Inhalers | |
| MXPA06009584A (en) | Stable pharmaceutical solution formulations for pressurized metered dose inhalers | |
| HK1103280B (en) | Stable pharmaceutical solution formulations for pressurized metered dose inhalers |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| 17P | Request for examination filed |
Effective date: 20020118 |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): AT BE CH CY DE DK ES FI FR GB GR IE IT LI LU MC NL PT SE |
|
| AX | Request for extension of the european patent |
Free format text: AL;LT;LV;MK;RO;SI |
|
| 17Q | First examination report despatched |
Effective date: 20040909 |
|
| GRAP | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOSNIGR1 |
|
| GRAS | Grant fee paid |
Free format text: ORIGINAL CODE: EPIDOSNIGR3 |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): AT BE CH CY DE DK ES FI FR GB GR IE IT LI LU MC NL PT SE |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: AT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20060322 Ref country code: CH Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20060322 Ref country code: NL Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20060322 Ref country code: LI Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20060322 Ref country code: BE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20060322 |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: FG4D |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: EP |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: FG4D |
|
| REF | Corresponds to: |
Ref document number: 60026840 Country of ref document: DE Date of ref document: 20060511 Kind code of ref document: P |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20060615 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20060622 Ref country code: DK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20060622 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: MC Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20060630 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: ES Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20060703 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: PT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20060822 |
|
| NLV1 | Nl: lapsed or annulled due to failure to fulfill the requirements of art. 29p and 29m of the patents act | ||
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PL |
|
| ET | Fr: translation filed | ||
| PLBI | Opposition filed |
Free format text: ORIGINAL CODE: 0009260 |
|
| PLAX | Notice of opposition and request to file observation + time limit sent |
Free format text: ORIGINAL CODE: EPIDOSNOBS2 |
|
| 26 | Opposition filed |
Opponent name: CHIESI FARMACEUTICI S.P.A. Effective date: 20061222 |
|
| PLAF | Information modified related to communication of a notice of opposition and request to file observations + time limit |
Free format text: ORIGINAL CODE: EPIDOSCOBS2 |
|
| PLAF | Information modified related to communication of a notice of opposition and request to file observations + time limit |
Free format text: ORIGINAL CODE: EPIDOSCOBS2 |
|
| PLBB | Reply of patent proprietor to notice(s) of opposition received |
Free format text: ORIGINAL CODE: EPIDOSNOBS3 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20060623 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: FI Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20060322 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LU Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20060615 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: CY Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20060322 |
|
| PLBP | Opposition withdrawn |
Free format text: ORIGINAL CODE: 0009264 |
|
| PLBD | Termination of opposition procedure: decision despatched |
Free format text: ORIGINAL CODE: EPIDOSNOPC1 |
|
| PLBM | Termination of opposition procedure: date of legal effect published |
Free format text: ORIGINAL CODE: 0009276 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: OPPOSITION PROCEDURE CLOSED |
|
| 27C | Opposition proceedings terminated |
Effective date: 20090613 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: PLFP Year of fee payment: 17 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: PLFP Year of fee payment: 18 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: CH Payment date: 20170921 Year of fee payment: 10 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: IT Payment date: 20170619 Year of fee payment: 18 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: PLFP Year of fee payment: 19 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: FR Payment date: 20180511 Year of fee payment: 19 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GB Payment date: 20180403 Year of fee payment: 19 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R119 Ref document number: 60026840 Country of ref document: DE |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20180615 Ref country code: DE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20190101 |
|
| GBPC | Gb: european patent ceased through non-payment of renewal fee |
Effective date: 20190615 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GB Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20190615 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: FR Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20190630 |