EP0132612A1 - Procédé pour la production d'hydrocarbures liquides - Google Patents
Procédé pour la production d'hydrocarbures liquides Download PDFInfo
- Publication number
- EP0132612A1 EP0132612A1 EP84107378A EP84107378A EP0132612A1 EP 0132612 A1 EP0132612 A1 EP 0132612A1 EP 84107378 A EP84107378 A EP 84107378A EP 84107378 A EP84107378 A EP 84107378A EP 0132612 A1 EP0132612 A1 EP 0132612A1
- Authority
- EP
- European Patent Office
- Prior art keywords
- solvent
- liquid hydrocarbons
- pressure
- waste
- bar
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
- 229930195733 hydrocarbon Natural products 0.000 title claims abstract description 27
- 150000002430 hydrocarbons Chemical class 0.000 title claims abstract description 27
- 239000007788 liquid Substances 0.000 title claims abstract description 27
- 238000000034 method Methods 0.000 title claims abstract description 24
- 238000004519 manufacturing process Methods 0.000 title claims abstract description 7
- 239000002904 solvent Substances 0.000 claims abstract description 29
- -1 polyethylene Polymers 0.000 claims abstract description 28
- 239000002699 waste material Substances 0.000 claims abstract description 24
- 239000004698 Polyethylene Substances 0.000 claims abstract description 14
- 239000004743 Polypropylene Substances 0.000 claims abstract description 14
- 229920000573 polyethylene Polymers 0.000 claims abstract description 14
- 229920001155 polypropylene Polymers 0.000 claims abstract description 14
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 claims description 54
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 claims description 6
- 239000000470 constituent Substances 0.000 claims description 5
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 3
- CTQNGGLPUBDAKN-UHFFFAOYSA-N O-Xylene Chemical compound CC1=CC=CC=C1C CTQNGGLPUBDAKN-UHFFFAOYSA-N 0.000 claims description 2
- 239000003502 gasoline Substances 0.000 claims description 2
- 239000008096 xylene Substances 0.000 claims description 2
- YNQLUTRBYVCPMQ-UHFFFAOYSA-N Ethylbenzene Chemical compound CCC1=CC=CC=C1 YNQLUTRBYVCPMQ-UHFFFAOYSA-N 0.000 claims 2
- 238000004821 distillation Methods 0.000 abstract description 6
- 239000012071 phase Substances 0.000 description 9
- XLOMVQKBTHCTTD-UHFFFAOYSA-N Zinc monoxide Chemical compound [Zn]=O XLOMVQKBTHCTTD-UHFFFAOYSA-N 0.000 description 8
- 229920001971 elastomer Polymers 0.000 description 8
- 238000000197 pyrolysis Methods 0.000 description 8
- 239000005060 rubber Substances 0.000 description 8
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 description 6
- 238000009835 boiling Methods 0.000 description 6
- 239000003921 oil Substances 0.000 description 6
- 239000000047 product Substances 0.000 description 6
- 239000011593 sulfur Substances 0.000 description 6
- 229910052717 sulfur Inorganic materials 0.000 description 6
- 239000000945 filler Substances 0.000 description 5
- 239000007789 gas Substances 0.000 description 5
- RRHGJUQNOFWUDK-UHFFFAOYSA-N Isoprene Chemical compound CC(=C)C=C RRHGJUQNOFWUDK-UHFFFAOYSA-N 0.000 description 4
- 229910052751 metal Inorganic materials 0.000 description 4
- 239000002184 metal Substances 0.000 description 4
- 239000002245 particle Substances 0.000 description 4
- 229920003023 plastic Polymers 0.000 description 4
- 239000004033 plastic Substances 0.000 description 4
- 239000011787 zinc oxide Substances 0.000 description 4
- 229910000831 Steel Inorganic materials 0.000 description 3
- 239000006229 carbon black Substances 0.000 description 3
- 238000004939 coking Methods 0.000 description 3
- 239000004744 fabric Substances 0.000 description 3
- 238000010438 heat treatment Methods 0.000 description 3
- 150000002739 metals Chemical class 0.000 description 3
- 238000000926 separation method Methods 0.000 description 3
- 239000007787 solid Substances 0.000 description 3
- 239000010959 steel Substances 0.000 description 3
- KAKZBPTYRLMSJV-UHFFFAOYSA-N Butadiene Chemical compound C=CC=C KAKZBPTYRLMSJV-UHFFFAOYSA-N 0.000 description 2
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 description 2
- 241000196324 Embryophyta Species 0.000 description 2
- VQTUBCCKSQIDNK-UHFFFAOYSA-N Isobutene Chemical compound CC(C)=C VQTUBCCKSQIDNK-UHFFFAOYSA-N 0.000 description 2
- QQONPFPTGQHPMA-UHFFFAOYSA-N Propene Chemical compound CC=C QQONPFPTGQHPMA-UHFFFAOYSA-N 0.000 description 2
- 230000003712 anti-aging effect Effects 0.000 description 2
- 239000011324 bead Substances 0.000 description 2
- 125000004432 carbon atom Chemical group C* 0.000 description 2
- 239000007795 chemical reaction product Substances 0.000 description 2
- 239000003795 chemical substances by application Substances 0.000 description 2
- 239000000460 chlorine Substances 0.000 description 2
- 229910052801 chlorine Inorganic materials 0.000 description 2
- 238000002485 combustion reaction Methods 0.000 description 2
- 150000001875 compounds Chemical class 0.000 description 2
- 239000010791 domestic waste Substances 0.000 description 2
- 239000007791 liquid phase Substances 0.000 description 2
- 239000000203 mixture Substances 0.000 description 2
- 150000002894 organic compounds Chemical class 0.000 description 2
- 239000004014 plasticizer Substances 0.000 description 2
- 229920000642 polymer Polymers 0.000 description 2
- 239000011347 resin Substances 0.000 description 2
- 229920005989 resin Polymers 0.000 description 2
- 239000007858 starting material Substances 0.000 description 2
- 229920003051 synthetic elastomer Polymers 0.000 description 2
- 239000005061 synthetic rubber Substances 0.000 description 2
- 238000004073 vulcanization Methods 0.000 description 2
- VGGSQFUCUMXWEO-UHFFFAOYSA-N Ethene Chemical compound C=C VGGSQFUCUMXWEO-UHFFFAOYSA-N 0.000 description 1
- 244000043261 Hevea brasiliensis Species 0.000 description 1
- 238000004566 IR spectroscopy Methods 0.000 description 1
- 239000012190 activator Substances 0.000 description 1
- 150000001335 aliphatic alkanes Chemical class 0.000 description 1
- 150000001338 aliphatic hydrocarbons Chemical class 0.000 description 1
- 150000004945 aromatic hydrocarbons Chemical class 0.000 description 1
- 230000005540 biological transmission Effects 0.000 description 1
- 230000015572 biosynthetic process Effects 0.000 description 1
- MTAZNLWOLGHBHU-UHFFFAOYSA-N butadiene-styrene rubber Chemical compound C=CC=C.C=CC1=CC=CC=C1 MTAZNLWOLGHBHU-UHFFFAOYSA-N 0.000 description 1
- 239000013064 chemical raw material Substances 0.000 description 1
- 150000008280 chlorinated hydrocarbons Chemical class 0.000 description 1
- 150000001805 chlorine compounds Chemical class 0.000 description 1
- YACLQRRMGMJLJV-UHFFFAOYSA-N chloroprene Chemical compound ClC(=C)C=C YACLQRRMGMJLJV-UHFFFAOYSA-N 0.000 description 1
- 238000004587 chromatography analysis Methods 0.000 description 1
- 239000000571 coke Substances 0.000 description 1
- 239000004020 conductor Substances 0.000 description 1
- 238000001816 cooling Methods 0.000 description 1
- 238000007334 copolymerization reaction Methods 0.000 description 1
- 150000001924 cycloalkanes Chemical class 0.000 description 1
- 238000000354 decomposition reaction Methods 0.000 description 1
- 150000002148 esters Chemical class 0.000 description 1
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 1
- 238000009413 insulation Methods 0.000 description 1
- 229920002521 macromolecule Polymers 0.000 description 1
- 238000002156 mixing Methods 0.000 description 1
- 229920003052 natural elastomer Polymers 0.000 description 1
- 229920001194 natural rubber Polymers 0.000 description 1
- 150000007524 organic acids Chemical class 0.000 description 1
- 235000005985 organic acids Nutrition 0.000 description 1
- 239000003960 organic solvent Substances 0.000 description 1
- 150000002898 organic sulfur compounds Chemical class 0.000 description 1
- 239000013502 plastic waste Substances 0.000 description 1
- 238000006116 polymerization reaction Methods 0.000 description 1
- 230000000379 polymerizing effect Effects 0.000 description 1
- 239000004800 polyvinyl chloride Substances 0.000 description 1
- 229920000915 polyvinyl chloride Polymers 0.000 description 1
- 230000002040 relaxant effect Effects 0.000 description 1
- 239000004071 soot Substances 0.000 description 1
- 125000004434 sulfur atom Chemical group 0.000 description 1
- 229920002994 synthetic fiber Polymers 0.000 description 1
- 239000012209 synthetic fiber Substances 0.000 description 1
- 238000004056 waste incineration Methods 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10G—CRACKING HYDROCARBON OILS; PRODUCTION OF LIQUID HYDROCARBON MIXTURES, e.g. BY DESTRUCTIVE HYDROGENATION, OLIGOMERISATION, POLYMERISATION; RECOVERY OF HYDROCARBON OILS FROM OIL-SHALE, OIL-SAND, OR GASES; REFINING MIXTURES MAINLY CONSISTING OF HYDROCARBONS; REFORMING OF NAPHTHA; MINERAL WAXES
- C10G1/00—Production of liquid hydrocarbon mixtures from oil-shale, oil-sand, or non-melting solid carbonaceous or similar materials, e.g. wood, coal
- C10G1/10—Production of liquid hydrocarbon mixtures from oil-shale, oil-sand, or non-melting solid carbonaceous or similar materials, e.g. wood, coal from rubber or rubber waste
Definitions
- the invention relates to a process for the production of liquid hydrocarbons from used tires, cable, polyethylene and / or polypropylene waste.
- the liquid hydrocarbons that are produced by this process include alkanes, cycloalkanes and aromatics, which have a boiling point of 20 to about 350 ° C and whose molecules contain 5 to about 30 carbon atoms.
- the liquid hydrocarbons produced by the process can contain organic sulfur and chlorine compounds.
- the liquid hydrocarbons can be used as heating oil or for blending heating oil or as a chemical raw material.
- Used tires belong to the waste products of the automotive industry and consist of one or more rubber compounds, a bead ring and a fabric.
- the rubber mixture contains rubber, soot, oils and resins, zinc oxide, vulcanization accelerators, anti-aging agents and sulfur.
- the rubber is composed of chain-like macromolecules which interengage 29lun g en and are additionally linked by sulfur atoms.
- the tires are manufactured using natural rubber and synthetic rubber, with the synthetic rubbers being polymerized by the polymerization of isoprene, butadiene, chlorobutadiene and the copolymerization of butadiene Styrene or iso-butene can be produced with isoprene.
- the carbon black serves as a filler and to improve the rubber properties, because it forms a very close bond with the rubber during tire manufacture.
- the oils and resins act as plasticizers and improve the processability of the rubber mixture.
- the zinc oxide serves as a filler and as an activator for the vulcanization accelerators.
- the bead ring stabilizes the tire against radial forces and is made of steel wire or steel cable.
- the fabric in the tire consists of synthetic fibers or steel wires.
- the weight proportion of the rubber compound in the old tires is usually between 75 and 80%.
- Cable waste arises from the processing and renewal of cables that consist of a metallic electrical conductor, a plastic insulation and a plastic jacket and are used for the transmission of messages or electrical energy.
- Polyethylene and polypropylene waste is produced in the manufacture and processing of plastics and accumulates there in a relatively pure form. These pure wastes can be processed into liquid hydrocarbons by the process according to the invention, while the polyethylene and polypropylene wastes contained in the household waste cannot be fed to the process according to the invention, since they are difficult to separate from the other plastics contained in the household waste, in particular polyvinyl chloride.
- Polyethylene and polypropylene are produced by polymerizing ethene or propene and, in addition to the polymers, also contain fillers.
- used tires are usually removed by incineration, which takes place either in specially designed ovens or in waste incineration plants.
- the exhaust gases from the incineration plants, in which used tires are burned, must be dedusted and desulphurized, in particular due to the high ZnO and S0 2 content.
- used tires can be removed by pyrolysis processes and processed into reusable products.
- the pyrolysis of old tires produces a pyrolysis coke, a flammable pyrolysis gas that mostly serves to cover the energy requirements of the pyrolysis process, and a pyrolysis oil that is used as heating oil.
- the polyethylene and polypropylene waste is mostly incinerated.
- the cable waste is usually deposited in a landfill.
- the combustion processes have the disadvantage that they are difficult to carry out due to the nature of the old tires and plastic waste and that the combustion exhaust gases have to be cleaned using complex processes.
- the disadvantage of the pyrolysis process is that a very large part of the organic compounds, which are valuable per se, is coked and thus converted into a less valuable state.
- the invention has for its object to provide a method with which old tires, cable, polyethylene and / or polypropylene waste can be removed without damage and processed into valuable reusable liquid hydrocarbons, the formation of gaseous and coke-like decomposition products should be largely avoided.
- the object underlying the invention is achieved in that the shredded old tires, cable, polyethylene and / or polypropylene waste at a temperature of 150 to 500 ° C and a pressure of 20 to 300 bar with a liquid at 1 bar and 20 ° C.
- Solvents are treated, that the solvent phase laden with the liquid hydrocarbons is separated off and broken down into its constituents by lowering the pressure or by lowering the pressure and temperature, and that the solvent is circulated.
- the polymer molecules are cleaved under the influence of the high pressure and the high temperature, liquid hydrocarbons in particular having about 5 to 30 carbon atoms being formed without any noticeable coking occurring.
- the liquid hydrocarbons are absorbed and removed comparatively quickly by the solvent.
- the plasticizers and anti-aging agents are released from the solvent and only slightly decomposed under the influence of pressure and temperature.
- the carbon black, the fillers, the metals and the fabric remain as a solid, insoluble residue, which also contains the small amounts of coking products that may arise during the process.
- the process has the advantage that the majority of the organic compounds present in the waste materials are converted into liquid reusable hydrocarbons.
- the process according to the invention can be carried out particularly successfully if gasoline, benzene, toluene, xylene, ethyl bezole and / or water is used as the solvent, the weight ratio of the waste materials to the solvent being between 1: 3 and 1:30.
- the organic solvents to be used according to the invention have an optimal dissolving power for the liquid hydrocarbons formed, while the water to be used according to the invention provides products with a lower molecular weight.
- the pressure and temperature are reduced in several stages and that the constituents of the loaded solvent phase are obtained in several fractions. This procedure allows the liquid hydrocarbons to be broken down into fractions suitable for various purposes.
- a suitable fraction can be returned to the process according to the invention as a solvent.
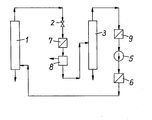
- the reactor 1 there is a bed of shredded old tires, cable, polyethylene and / or polypropylene waste.
- the sulfur and chlorine content of the old tires is advantageously diluted by the polyethylene and polypropylene waste.
- the solvent is heated in the heat exchanger 6 to the process temperature of 150 to 500 ° C and brought to a process pressure of 20 to 300 bar by the compressor 5 and conveyed into the reactor 1.
- the solvent flows through the reactor 1 and removes the liquid hydrocarbons formed in the reactor 1 and the gaseous reaction products formed in a small amount.
- the solvent phase laden with the liquid hydrocarbons is removed, expanded in the expansion valve 2, cooled in the heat exchanger 7, freed from the gaseous reaction products in the separation vessel 8 and then fed to the distillation column 3.
- the higher-boiling hydrocarbons are taken off at the bottom of the distillation column 3, while the low-boiling hydrocarbons are taken off together with the solvent at the top of the distillation column 3.
- the low-boiling fraction is then cooled in the heat exchanger 9 and, after liquefaction, is returned from the compressor 5 to the reactor 1. It is possible to carry out the expansion and cooling of the solvent phase laden with the liquid hydrocarbons in several stages, which is not shown in the drawing.
- the solid residue which contains the insoluble constituents of the waste materials used and the coking products is removed from the reactor 1.
- Used tires and cable waste were shredded so that particles with an edge length of approx. 2 cm were obtained. 602 g of these particles were treated with toluene at 350 ° C. and 80 bar for 4 hours.
- the solvent phase laden with the liquid hydrocarbons was then separated off and let down to 10 bar and cooled to 310 ° C.
- a separation was carried out into a toluene-containing liquid phase and into a gas phase, which mainly consisted of toluene.
- the liquid phase which contained about 85% toluene, was distilled to remove the toluene and the low-boiling liquid hydrocarbons after relaxing to atmospheric pressure.
- the solid residue obtained after the toluene treatment was powdery and mainly consisted of carbon black, ZnO, other tire fillers and pieces of metal. Its sulfur content was 2.37%, while the sulfur content of the particles used was 1.70%.
- 12 kg of toluene were used for 1 kg of starting material, and these can largely be recovered and recycled.
- the metals present in the starting material were found in the residue in unchanged form. This is an advantage over pyrolysis carried out at higher temperatures. There, the metals present in the old tires and cable waste melt together in an undesirable manner, while in the process according to the invention they can be reduced at a low rate with mechanical separation processes! Energy expenditure can be sorted out and recycled.
Landscapes
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Life Sciences & Earth Sciences (AREA)
- Wood Science & Technology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Production Of Liquid Hydrocarbon Mixture For Refining Petroleum (AREA)
- Separation, Recovery Or Treatment Of Waste Materials Containing Plastics (AREA)
- Processes Of Treating Macromolecular Substances (AREA)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| AT84107378T ATE30045T1 (de) | 1983-07-21 | 1984-06-27 | Verfahren zur herstellung fluessiger kohlenwasserstoffe. |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DE3326284 | 1983-07-21 | ||
| DE3326284A DE3326284C2 (de) | 1983-07-21 | 1983-07-21 | Verfahren zur Herstellung flüssiger Kohlenwasserstoffe |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| EP0132612A1 true EP0132612A1 (fr) | 1985-02-13 |
| EP0132612B1 EP0132612B1 (fr) | 1987-09-30 |
Family
ID=6204535
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP84107378A Expired EP0132612B1 (fr) | 1983-07-21 | 1984-06-27 | Procédé pour la production d'hydrocarbures liquides |
Country Status (6)
| Country | Link |
|---|---|
| US (1) | US4642401A (fr) |
| EP (1) | EP0132612B1 (fr) |
| JP (1) | JPS6040193A (fr) |
| AT (1) | ATE30045T1 (fr) |
| DE (1) | DE3326284C2 (fr) |
| ES (1) | ES8504897A1 (fr) |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1994020590A2 (fr) * | 1993-03-10 | 1994-09-15 | The University Of Wyoming Research Corporation Doing Business As Western Research Institute | Procede de recyclage de dechets plastiques |
| FR2706168A1 (fr) * | 1993-04-01 | 1994-12-16 | Yamasaki Nakamichi | Procédé pour la conversion en huiles de caoutchoucs vulcanisés. |
| US6861568B1 (en) | 1998-05-15 | 2005-03-01 | Wyoming Research Corporation | Process for waste plastic recycling |
| EP1770148A1 (fr) * | 2005-10-03 | 2007-04-04 | CHUNG BONG, ChOON | Procédé et appareil de production d'huile à partir de déchets plastiques |
Families Citing this family (57)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE3710021A1 (de) * | 1987-03-30 | 1988-10-20 | Veba Oel Entwicklungs Gmbh | Verfahren zur hydrierenden konversion von schwer- und rueckstandsoelen |
| US5099086A (en) * | 1989-06-28 | 1992-03-24 | Universite Laval | Extraction of commercially valuable chemicals from tire-derived pyrolytic oils |
| US5158982A (en) * | 1991-10-04 | 1992-10-27 | Iit Research Institute | Conversion of municipal waste to useful oils |
| US5158983A (en) * | 1991-10-04 | 1992-10-27 | Iit Research Institute | Conversion of automotive tire scrap to useful oils |
| US5230777A (en) * | 1991-12-13 | 1993-07-27 | James Jarrell | Apparatus for producing fuel and carbon black from rubber tires |
| US5296515A (en) * | 1992-06-03 | 1994-03-22 | Phillips Petroleum Company | Hydrovisbreaking of hydrocarbon polymers |
| US5364996A (en) * | 1992-06-09 | 1994-11-15 | Texaco Inc. | Partial oxidation of scrap rubber tires and used motor oil |
| US5569801A (en) * | 1992-08-27 | 1996-10-29 | Fina Research, S.A. | Polymer conversion process |
| US5288934A (en) * | 1992-08-27 | 1994-02-22 | Petrofina, S.A. | Process for the conversion of polymers |
| US5301834A (en) * | 1993-01-21 | 1994-04-12 | The Lift Ticket | Card vending machine |
| US5799626A (en) * | 1993-01-28 | 1998-09-01 | Ponsford; Thomas E. | Methods for using styrene oil (as heat transfer fluid, hydraulic fluid, lubricant) |
| US5406010A (en) * | 1993-01-28 | 1995-04-11 | Ponsford; Thomas E. | Method of reclaiming styrene and other products from polystyrene based products |
| US5639934A (en) * | 1993-04-01 | 1997-06-17 | Nakamichi Yamasaki | Process for the desulfurization of sulfur-containing compositions by hydrothermal reaction |
| DE4411483C2 (de) * | 1993-04-01 | 2002-10-24 | Nakamichi Yamasaki | Verfahren zur Entschwefelung von schwefelhaltigen Zusammensetzungen durch hydrothermale Reaktion |
| DE4447937C2 (de) * | 1993-04-01 | 2002-12-05 | Nakamichi Yamasaki | Verfahren zur Umwandlung vulkanisierter Kautschuke in Öle unter Verwendung überkritischer wässriger Flüssigkeiten |
| US5395404A (en) * | 1993-08-27 | 1995-03-07 | The Jerrold Corporation | Apparatus for pyrolyzing tires |
| US5445659A (en) * | 1993-10-04 | 1995-08-29 | Texaco Inc. | Partial oxidation of products of liquefaction of plastic materials |
| EP0722480B1 (fr) * | 1993-10-04 | 2000-04-19 | Texaco Development Corporation | Liquefaction et oxydation partielle de materiaux plastiques |
| GB2301112B (en) * | 1995-05-24 | 1998-12-16 | California Engineering Consult | Liquefaction of solid wastes and separation of solid inorganic matter therefrom |
| ES2119657B1 (es) * | 1995-09-25 | 1999-07-16 | Marin Victorino Luengo | Procedimiento e instalacion para la separacion y aprovechamiento de los componentes organicos e inorganicos de plasticos, cauchos y neumaticos, por disolucion calorifica con aceite y posteriormente en eter. |
| US5824193A (en) * | 1997-03-27 | 1998-10-20 | Edwards; Raymond S. | Method of thermally treating plastics material |
| US5977294A (en) * | 1997-05-13 | 1999-11-02 | Prs, Llc | Polymer deformulation by solvent solution filtration |
| US6078888A (en) * | 1997-07-16 | 2000-06-20 | Gilbarco Inc. | Cryptography security for remote dispenser transactions |
| US6143940A (en) * | 1998-12-30 | 2000-11-07 | Chevron U.S.A. Inc. | Method for making a heavy wax composition |
| JP2003041045A (ja) * | 2001-07-26 | 2003-02-13 | Yokohama Rubber Co Ltd:The | ゴム成形品からの材料回収方法および回収材料 |
| US7048832B2 (en) * | 2002-01-07 | 2006-05-23 | Material Conversion Corp. | Method and system for extracting hydrocarbon fuel products from plastic material |
| US6822126B2 (en) * | 2002-04-18 | 2004-11-23 | Chevron U.S.A. Inc. | Process for converting waste plastic into lubricating oils |
| US6774272B2 (en) | 2002-04-18 | 2004-08-10 | Chevron U.S.A. Inc. | Process for converting heavy Fischer Tropsch waxy feeds blended with a waste plastic feedstream into high VI lube oils |
| US6703535B2 (en) * | 2002-04-18 | 2004-03-09 | Chevron U.S.A. Inc. | Process for upgrading fischer-tropsch syncrude using thermal cracking and oligomerization |
| GB0211052D0 (en) * | 2002-05-15 | 2002-06-26 | Tucker Alan C N | Incineration of plastic waste suspended in paraffin gel |
| CA2426253A1 (fr) * | 2003-04-22 | 2004-10-22 | Hurdon A. Hooper | Reduction du caoutchouc |
| EA010464B1 (ru) * | 2004-02-26 | 2008-08-29 | Игорь Антонович Рожновский | Устройство для переработки углеродсодержащих отходов |
| US7893307B2 (en) * | 2007-02-23 | 2011-02-22 | Smith David G | Apparatus and process for converting feed material into reusable hydrocarbons |
| US20090007484A1 (en) * | 2007-02-23 | 2009-01-08 | Smith David G | Apparatus and process for converting biomass feed materials into reusable carbonaceous and hydrocarbon products |
| US7834226B2 (en) * | 2007-12-12 | 2010-11-16 | Chevron U.S.A. Inc. | System and method for producing transportation fuels from waste plastic and biomass |
| US8664458B2 (en) * | 2010-07-15 | 2014-03-04 | Greenmantra Recycling Technologies Ltd. | Method for producing waxes and grease base stocks through catalytic depolymerisation of waste plastics |
| RU2603965C2 (ru) | 2011-01-05 | 2016-12-10 | Игнайт Ресорсиз Пти Лтд | Переработка органического материала |
| US8480880B2 (en) | 2011-01-18 | 2013-07-09 | Chevron U.S.A. Inc. | Process for making high viscosity index lubricating base oils |
| AT511772B1 (de) | 2011-05-05 | 2018-03-15 | Omv Refining & Marketing Gmbh | Verfahren und vorrichtung zur energieeffizienten aufbereitung sekundärer lagerstätten |
| CA3229291A1 (fr) | 2013-01-17 | 2014-07-24 | Greenmantra Recycling Technologies Ltd. | Depolymerisation catalytique de matieres polymeres |
| US10472487B2 (en) | 2015-12-30 | 2019-11-12 | Greenmantra Recycling Technologies Ltd. | Reactor for continuously treating polymeric material |
| BR112018016499B1 (pt) | 2016-02-13 | 2022-03-29 | Greenmantra Recycling Technologies Ltd | Método para formar uma cera e empregar a referida cera para modificar asfalto |
| BR112018068992B1 (pt) | 2016-03-24 | 2023-01-31 | Greenmantra Recycling Technologies Ltd | Método para produzir um polímero modificado com um índice de fluidez melhorado a partir de uma cera despolimerizada |
| EP3519487A4 (fr) | 2016-09-29 | 2020-07-29 | GreenMantra Recycling Technologies Ltd | Réacteur pour le traitement de matériau de polystyrène |
| US10723858B2 (en) | 2018-09-18 | 2020-07-28 | Greenmantra Recycling Technologies Ltd. | Method for purification of depolymerized polymers using supercritical fluid extraction |
| CA3164216A1 (fr) | 2019-12-23 | 2021-07-01 | Chevron U.S.A. Inc. | Economie circulaire de dechets plastiques en polypropylene par l'intermediaire d'une unite fcc de raffinerie |
| MX2022007132A (es) | 2019-12-23 | 2022-09-19 | Chevron Usa Inc | Economia circular para residuos plasticos en polipropileno a traves de craqueo catalitico de fluidos (fcc) de refineria y unidades de alquilacion. |
| MX2022007303A (es) | 2019-12-23 | 2022-10-21 | Chevron Usa Inc | Economia circular para residuos plasticos en polietileno a traves de una unidad de crudo de refineria. |
| BR112022011773A2 (pt) | 2019-12-23 | 2022-08-30 | Chevron Usa Inc | Economia circular para resíduos de plástico para polietileno e produtos químicos via unidade de cru de refinaría |
| US11518945B2 (en) | 2019-12-23 | 2022-12-06 | Chevron U.S.A. Inc. | Circular economy for plastic waste to polypropylene and lubricating oil via refinery FCC and isomerization dewaxing units |
| JP2023508353A (ja) | 2019-12-23 | 2023-03-02 | シェブロン ユー.エス.エー. インコーポレイテッド | 精製fcc及びアルキレーションユニットを介したポリエチレンへの廃プラスチックのサーキュラーエコノミー |
| CA3164223A1 (fr) | 2019-12-23 | 2021-07-01 | Chevron U.S.A. Inc. | Economie circulaire de dechets plastiques en polyethylene et huile lubrifiante par l'intermediaire d'unites de brut et de deparaffinage par isomerisation |
| US11566182B2 (en) | 2020-03-30 | 2023-01-31 | Chevron U.S.A. Inc. | Circular economy for plastic waste to polyethylene via refinery FCC feed pretreater and FCC units |
| US11306253B2 (en) | 2020-03-30 | 2022-04-19 | Chevron U.S.A. Inc. | Circular economy for plastic waste to polyethylene via refinery FCC or FCC/alkylation units |
| WO2021216873A1 (fr) | 2020-04-22 | 2021-10-28 | Chevron U.S.A. Inc. | Économie circulaire de déchets plastiques en polypropylène par raffinage d'huile avec filtration et traitement d'oxyde métallique d'huile de pyrolyse |
| KR20230004713A (ko) | 2020-04-22 | 2023-01-06 | 셰브런 유.에스.에이.인크. | 열분해 오일의 여과 및 금속 산화물 처리가 있는 오일 정제를 통한 플라스틱 폐기물의 폴리에틸렌으로의 순환 경제 |
| WO2024133944A1 (fr) | 2022-12-23 | 2024-06-27 | Borealis Ag | Procédé de recyclage de plastique |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB1372400A (en) * | 1970-12-24 | 1974-10-30 | Exxon Research Engineering Co | Conversion of waste polymers to useful products |
| DD144171A5 (de) * | 1978-06-05 | 1980-10-01 | Bipronaft | Verfahren zur umwandlung von altgummi |
| GB2084182A (en) * | 1980-09-29 | 1982-04-07 | Inst Francais Du Petrole | Conversion of unwanted tyres to liquid and gaseous materials |
Family Cites Families (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3704108A (en) * | 1970-09-25 | 1972-11-28 | Hydrocarbon Research Inc | Hydroconversion of waste natural and synthetic rubbers |
| JPS4897972A (fr) * | 1972-03-27 | 1973-12-13 | ||
| US3974206A (en) * | 1974-07-09 | 1976-08-10 | Mitsubishi Gas Chemical Company, Inc. | Process for the thermal decomposition of thermoplastic resins with a heat transfer medium |
| US4175211A (en) * | 1977-03-14 | 1979-11-20 | Mobil Oil Corporation | Method for treatment of rubber and plastic wastes |
| US4118281A (en) * | 1977-04-15 | 1978-10-03 | Mobil Oil Corporation | Conversion of solid wastes to fuel coke and gasoline/light oil |
| US4235676A (en) * | 1977-09-16 | 1980-11-25 | Deco Industries, Inc. | Apparatus for obtaining hydrocarbons from rubber tires and from industrial and residential waste |
| IT1123572B (it) * | 1979-09-10 | 1986-04-30 | Anic Spa | Procedimento per la preparazione di additivi miglioratori dell'indice di viscosita'di oli lubrificanti per cracking in fase liquida di gomme sintetiche |
| JPS56118431A (en) * | 1980-02-25 | 1981-09-17 | Bridgestone Corp | Decomposition method of vulcanized rubber |
| DE3037829C2 (de) * | 1980-10-07 | 1983-08-25 | Rütgerswerke AG, 6000 Frankfurt | Verfahren zur Herstellung modifizierter Peche und niedrigsiedender Aromaten oder Olefine und Verwendung dieser Peche |
| US4384150A (en) * | 1981-08-20 | 1983-05-17 | Lyakhevich Genrikh D | Method of making either a softener for rubber mixtures or a furnace fuel oil |
| US4463203A (en) * | 1981-11-13 | 1984-07-31 | Gi Kim D | Process for the preparation of fuel oil, fuel gas and pyrolysis coke by pyrolysis |
| SE455703B (sv) * | 1983-06-17 | 1988-08-01 | Bruss Ti Kirova | Apparatur for termisk nedbrytning av polymera material |
-
1983
- 1983-07-21 DE DE3326284A patent/DE3326284C2/de not_active Expired
-
1984
- 1984-06-27 AT AT84107378T patent/ATE30045T1/de not_active IP Right Cessation
- 1984-06-27 EP EP84107378A patent/EP0132612B1/fr not_active Expired
- 1984-07-16 US US06/631,420 patent/US4642401A/en not_active Expired - Fee Related
- 1984-07-20 JP JP59149782A patent/JPS6040193A/ja active Pending
- 1984-07-20 ES ES534485A patent/ES8504897A1/es not_active Expired
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB1372400A (en) * | 1970-12-24 | 1974-10-30 | Exxon Research Engineering Co | Conversion of waste polymers to useful products |
| DD144171A5 (de) * | 1978-06-05 | 1980-10-01 | Bipronaft | Verfahren zur umwandlung von altgummi |
| GB2084182A (en) * | 1980-09-29 | 1982-04-07 | Inst Francais Du Petrole | Conversion of unwanted tyres to liquid and gaseous materials |
Cited By (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1994020590A2 (fr) * | 1993-03-10 | 1994-09-15 | The University Of Wyoming Research Corporation Doing Business As Western Research Institute | Procede de recyclage de dechets plastiques |
| WO1994020590A3 (fr) * | 1993-03-10 | 1994-11-10 | Univ Wyoming | Procede de recyclage de dechets plastiques |
| US5753086A (en) * | 1993-03-10 | 1998-05-19 | The University Of Wyoming Research Corp. | Process for waste plastic recycling |
| FR2706168A1 (fr) * | 1993-04-01 | 1994-12-16 | Yamasaki Nakamichi | Procédé pour la conversion en huiles de caoutchoucs vulcanisés. |
| US6861568B1 (en) | 1998-05-15 | 2005-03-01 | Wyoming Research Corporation | Process for waste plastic recycling |
| EP1770148A1 (fr) * | 2005-10-03 | 2007-04-04 | CHUNG BONG, ChOON | Procédé et appareil de production d'huile à partir de déchets plastiques |
Also Published As
| Publication number | Publication date |
|---|---|
| ES534485A0 (es) | 1985-04-16 |
| JPS6040193A (ja) | 1985-03-02 |
| EP0132612B1 (fr) | 1987-09-30 |
| DE3326284C2 (de) | 1985-08-14 |
| ES8504897A1 (es) | 1985-04-16 |
| ATE30045T1 (de) | 1987-10-15 |
| DE3326284A1 (de) | 1985-02-21 |
| US4642401A (en) | 1987-02-10 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP0132612B1 (fr) | Procédé pour la production d'hydrocarbures liquides | |
| EP0692009B1 (fr) | Procede de traitement de matieres plastiques usees ou de dechets de matieres plastiques | |
| EP0182309B1 (fr) | Procédé pour la réutilisation par hydration de déchets contenant du carbone d'origine synthétique | |
| EP0710270B1 (fr) | Procede de recyclage de matieres plastiques dans un vapocraqueur | |
| DE69413576T2 (de) | Verfahren zum Cracken von Altreifen | |
| EP0717094B1 (fr) | Procédé pour la récupération de paraffines solides à partir de déchets polyoléfiniques pollués | |
| EP2209753B1 (fr) | Procédé de fabrication industrielle de carbure de calcium dans des fours électriques à cuve basse | |
| WO1995032262A1 (fr) | Dispositif de depolymerisation de matieres plastiques usagees et de recuperation | |
| DE3037829C2 (de) | Verfahren zur Herstellung modifizierter Peche und niedrigsiedender Aromaten oder Olefine und Verwendung dieser Peche | |
| EP0659867A2 (fr) | Procédé et installation pour la dépolymérisation des matériaux plastiques | |
| EP0468073B1 (fr) | Procédé pour la valorisation complète de déchets de polymères à haut poids moléculaire | |
| EP0568791B1 (fr) | Procédé pour la régénération de matériaux d'emballage | |
| EP0713906A1 (fr) | Procédé de recyclage de matériaux plastiques dans une unité de vapocraquage | |
| DE4344846C1 (de) | Verfahren zur Gewinnung von festen und flüssigen Kohlenwasserstoffen aus Polyolefinen | |
| DE2415412C3 (de) | Verfahren und Vorrichtung zur Behandlung von hochmolekularen Hydrocarbonaten oder Altolen | |
| DE2530229A1 (de) | Verfahren zur umwandlung von altreifen, gummi und/oder anderen kunststoffen | |
| DE19512029A1 (de) | Verfahren zur Herstellung von Paraffinen, Wachsen und Basisölen | |
| DE10037229B4 (de) | Verfahren zur Herstellung von hochmolekularen Wachsen aus Polyolefinen | |
| EP0669388A2 (fr) | Récuperation de produits de valeur commerciale à partir de pneumatiques usés en caoutchouc | |
| DE19702539A1 (de) | Verfahren zur Gewinnung von Paraffinen und/oder hochschmelzenden Wachsen aus Altkunststoffen | |
| DE4344845C1 (de) | Verfahren zur Herstellung von Kohlenwasserstoffschmelzen aus Polyolefinen | |
| EP3959294B1 (fr) | Procédé de dépolymérisation | |
| DE19707305B4 (de) | Verfahren zur Gewinnung von Mikrowachsen, Paraffinen und Ölen aus Altkunststoffen oder Altkunststoffgemischen | |
| DE2623331A1 (de) | Tieftemperaturpyrolyse von polyolefinen zu fluessigen und gasfoermigen kohlenwasserstoffen | |
| DD255353A1 (de) | Verfahren zur rueckgewinnung von russ |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| AK | Designated contracting states |
Designated state(s): AT BE CH FR GB IT LI NL |
|
| 17P | Request for examination filed |
Effective date: 19850619 |
|
| 17Q | First examination report despatched |
Effective date: 19860620 |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): AT BE CH FR GB IT LI NL |
|
| REF | Corresponds to: |
Ref document number: 30045 Country of ref document: AT Date of ref document: 19871015 Kind code of ref document: T |
|
| GBT | Gb: translation of ep patent filed (gb section 77(6)(a)/1977) | ||
| ITF | It: translation for a ep patent filed | ||
| ET | Fr: translation filed | ||
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: AT Effective date: 19880627 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LI Effective date: 19880630 Ref country code: CH Effective date: 19880630 |
|
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| 26N | No opposition filed | ||
| BERE | Be: lapsed |
Owner name: FRIED. KRUPP G.M.B.H. Effective date: 19880630 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: NL Effective date: 19890101 |
|
| NLV4 | Nl: lapsed or anulled due to non-payment of the annual fee | ||
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: FR Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 19890228 |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PL |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: ST |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GB Effective date: 19890627 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: BE Effective date: 19890630 |
|
| GBPC | Gb: european patent ceased through non-payment of renewal fee |