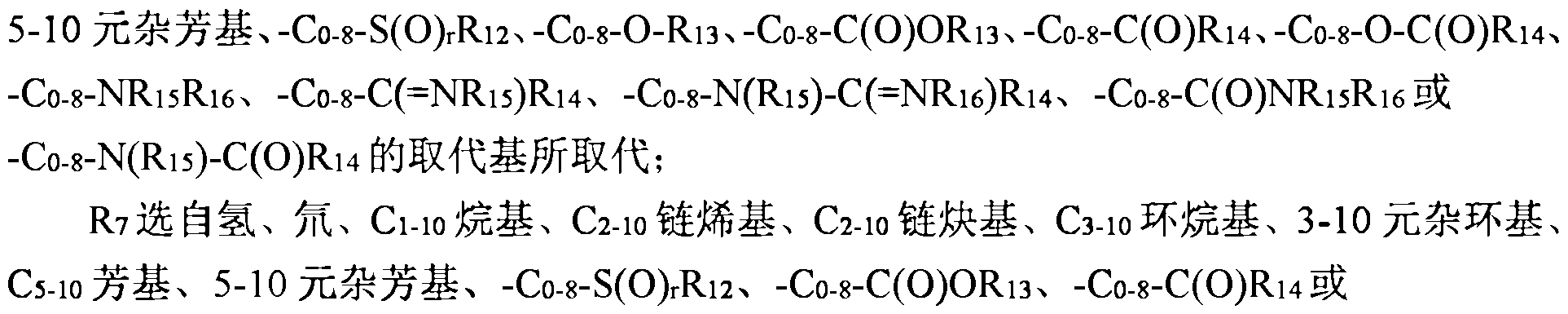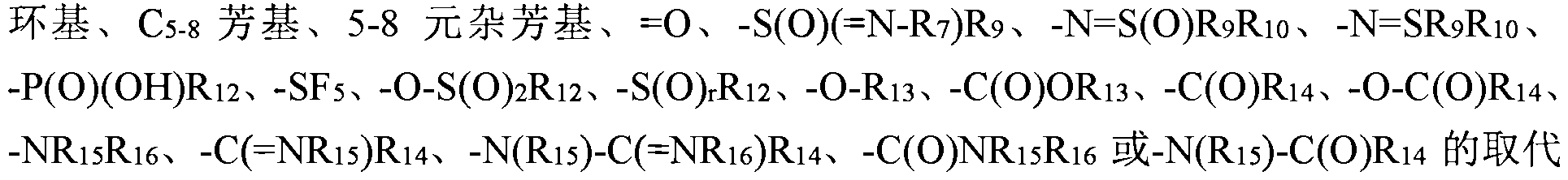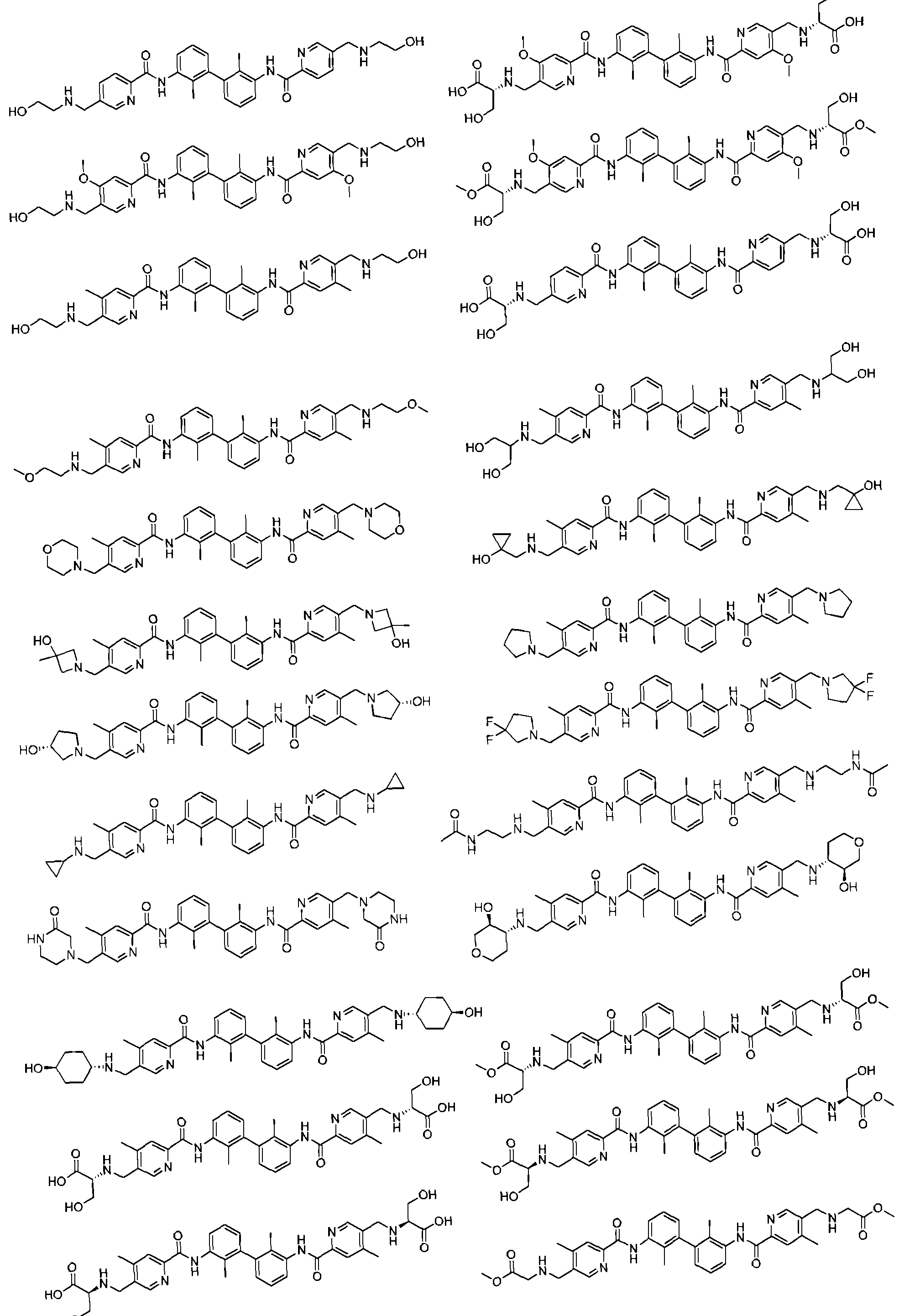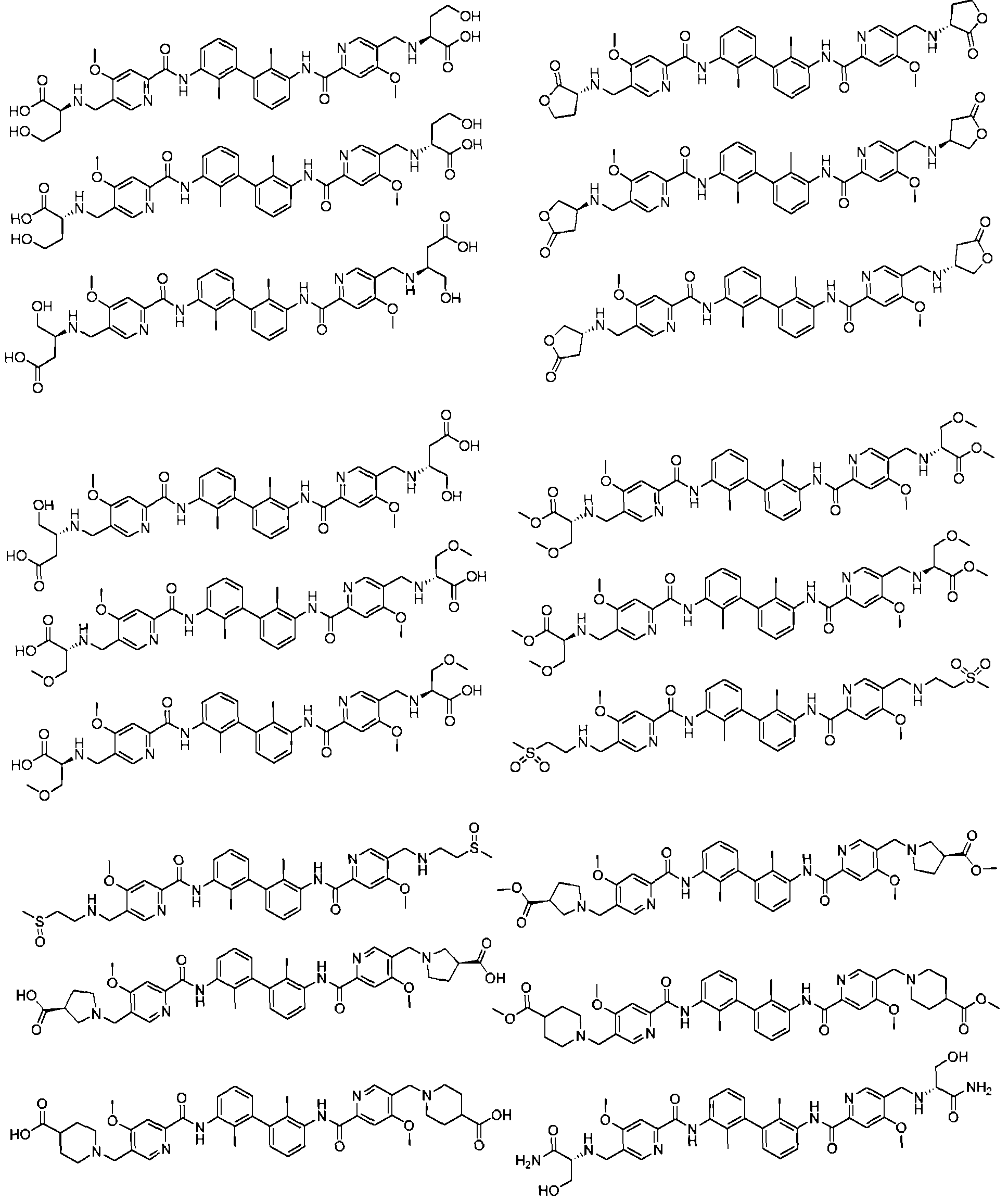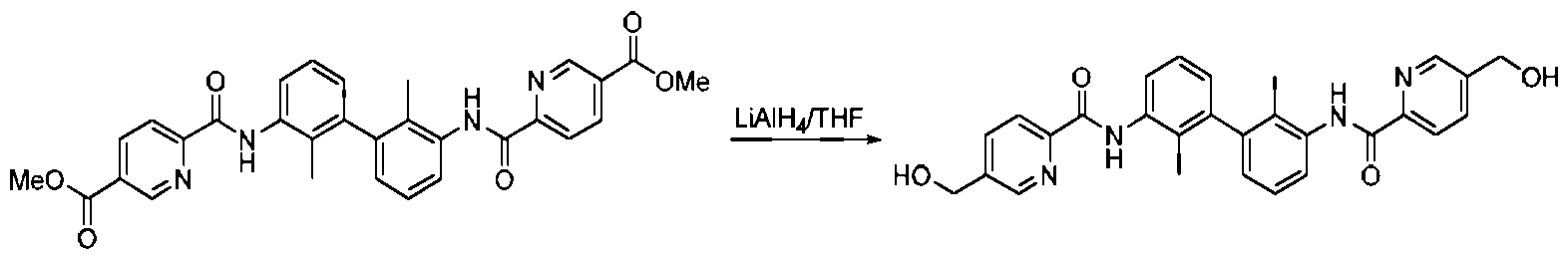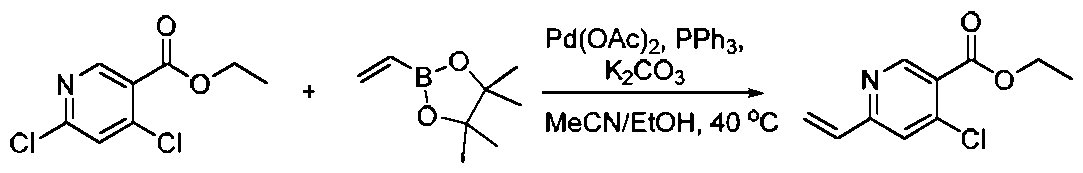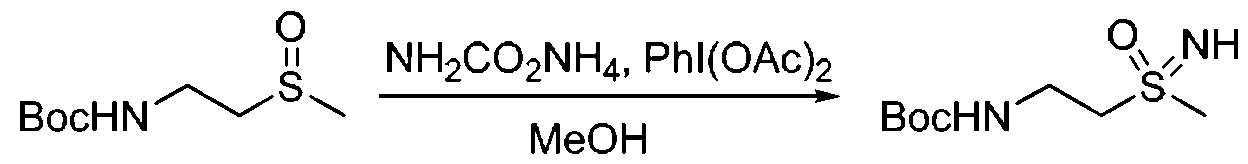WO2019149183A1 - 一种联芳基衍生物、其制备方法和在药学上的应用 - Google Patents
一种联芳基衍生物、其制备方法和在药学上的应用 Download PDFInfo
- Publication number
- WO2019149183A1 WO2019149183A1 PCT/CN2019/073594 CN2019073594W WO2019149183A1 WO 2019149183 A1 WO2019149183 A1 WO 2019149183A1 CN 2019073594 W CN2019073594 W CN 2019073594W WO 2019149183 A1 WO2019149183 A1 WO 2019149183A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- group
- hydrazine
- substituted
- alkyl
- cyano
- Prior art date
Links
- 0 CC(*)(C(CCCC1)CCCC1C(N(*)C(C(*)=C(*=C)C(C(*)=C(*=*)N(*)C(/C(/C1)=C/C=C/C(/C(*)(*)N(*)*)=C/*1=*)=O)=*)=I)=O)N(C)* Chemical compound CC(*)(C(CCCC1)CCCC1C(N(*)C(C(*)=C(*=C)C(C(*)=C(*=*)N(*)C(/C(/C1)=C/C=C/C(/C(*)(*)N(*)*)=C/*1=*)=O)=*)=I)=O)N(C)* 0.000 description 5
- JNLQYXWNWXVVTC-VOTSOKGWSA-N CC/C(/C(OCC)=O)=C\N=C Chemical compound CC/C(/C(OCC)=O)=C\N=C JNLQYXWNWXVVTC-VOTSOKGWSA-N 0.000 description 1
- PMCAALXWRQCXMF-UHFFFAOYSA-N N#CCN=S1(CCCC1)=O Chemical compound N#CCN=S1(CCCC1)=O PMCAALXWRQCXMF-UHFFFAOYSA-N 0.000 description 1
- KSNROUHXOGHUIK-UHFFFAOYSA-N NCCN=S1(CCCC1)=O Chemical compound NCCN=S1(CCCC1)=O KSNROUHXOGHUIK-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D213/00—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members
- C07D213/02—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members
- C07D213/04—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D213/60—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D213/78—Carbon atoms having three bonds to hetero atoms, with at the most one bond to halogen, e.g. ester or nitrile radicals
- C07D213/81—Amides; Imides
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D519/00—Heterocyclic compounds containing more than one system of two or more relevant hetero rings condensed among themselves or condensed with a common carbocyclic ring system not provided for in groups C07D453/00 or C07D455/00
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/44—Non condensed pyridines; Hydrogenated derivatives thereof
- A61K31/4427—Non condensed pyridines; Hydrogenated derivatives thereof containing further heterocyclic ring systems
- A61K31/444—Non condensed pyridines; Hydrogenated derivatives thereof containing further heterocyclic ring systems containing a six-membered ring with nitrogen as a ring heteroatom, e.g. amrinone
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/535—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with at least one nitrogen and one oxygen as the ring hetero atoms, e.g. 1,2-oxazines
- A61K31/5375—1,4-Oxazines, e.g. morpholine
- A61K31/5377—1,4-Oxazines, e.g. morpholine not condensed and containing further heterocyclic rings, e.g. timolol
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/02—Immunomodulators
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings
- C07D401/12—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/14—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D405/00—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom
- C07D405/14—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D409/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms
- C07D409/14—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D413/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms
- C07D413/14—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D491/00—Heterocyclic compounds containing in the condensed ring system both one or more rings having oxygen atoms as the only ring hetero atoms and one or more rings having nitrogen atoms as the only ring hetero atoms, not provided for by groups C07D451/00 - C07D459/00, C07D463/00, C07D477/00 or C07D489/00
- C07D491/02—Heterocyclic compounds containing in the condensed ring system both one or more rings having oxygen atoms as the only ring hetero atoms and one or more rings having nitrogen atoms as the only ring hetero atoms, not provided for by groups C07D451/00 - C07D459/00, C07D463/00, C07D477/00 or C07D489/00 in which the condensed system contains two hetero rings
- C07D491/10—Spiro-condensed systems
- C07D491/107—Spiro-condensed systems with only one oxygen atom as ring hetero atom in the oxygen-containing ring
Definitions
- a biaryl derivative, a preparation method thereof and application in pharmacy TECHNICAL FIELD belongs to the field of pharmaceutical synthesis, and particularly relates to a biaryl derivative. , its preparation method and its application in pharmacy.
- the immune system plays a very important role in controlling and eradicating diseases such as cancer.
- tumor cells are often able to develop strategies to escape or inhibit the monitoring of the immune system to promote their own malignant growth.
- One of the most important mechanisms is to alter the expression of immunological checkpoint molecules on co-stimulation and co-suppression on immune cells.
- Block immune checkpoint molecules like The signaling pathway has proven to be a promising and effective treatment.
- Programmed cell death molecule 1 Also known as 00279, it is a receptor molecule expressed on activated Ding cells, natural killer cells, :6 cells, and macrophage surfaces. Its structure contains an extracellular immunoglobulin variable region similar. a domain, a transmembrane region and an intracellular region, wherein the intracellular region contains two phosphorylation sites located in an immunoreceptor tyrosine kinase-based inhibition domain and an immunoreceptor tyrosine kinase-based translation domain , the signal path.
- Protein in macrophages and dendritic cells in lipopolysaccharide (1 ⁇ 5) and granulocyte-macrophage colony-stimulating factor It is up-regulated after treatment and is also up-regulated after stimulation of butyl and :6 cells via the butyl cell receptor and the 6-cell receptor signaling pathway. It is also highly expressed in almost all tumor cells, and in interferon ( ⁇ ) There will be an up-regulation of expression after stimulation, in fact, tumor The expression status is considered to have a prognostic correlation in a variety of tumor types, and conversely, is more concentrated, mainly expressed on dendritic cells.
- 0-1-expressing I cells When 0-1-expressing I cells are contacted with cells expressed by their ligands, functional activities such as cell proliferation, cytokine release, and cell lytic activity after stimulation of those antigens are inhibited. therefore, The interaction with its ligand is functionally an intrinsic negative feedback regulation mechanism that prevents people from over-activation of butyl cells during infection, immune tolerance or tumorigenesis, thereby reducing autoimmune diseases and promoting autoimmune tolerance.
- Long-term antigenic stimulation such as occurs in tumors or long-term infections, can cause high levels of butyl cell expression? 0-1, and lack of activity in the response to these long-term antigens, no function, that is, the depletion of the function of the cells. : 6 cells and their ligands bring about inhibition and corresponding functional failure.
- Correction page (Article 91) ⁇ 0 2019/149183 ⁇ (:17(: ⁇ 2019/073594 should, including in response to a long-term infection, a therapeutic vaccine).
- the object of the invention is to provide blocking? 0-1/?0 1 interacting compounds, which is expected to develop a new generation Inhibitor.
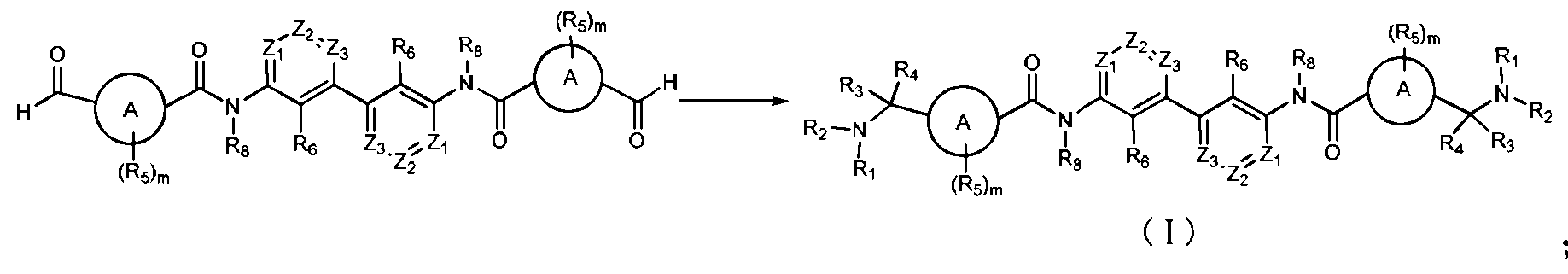
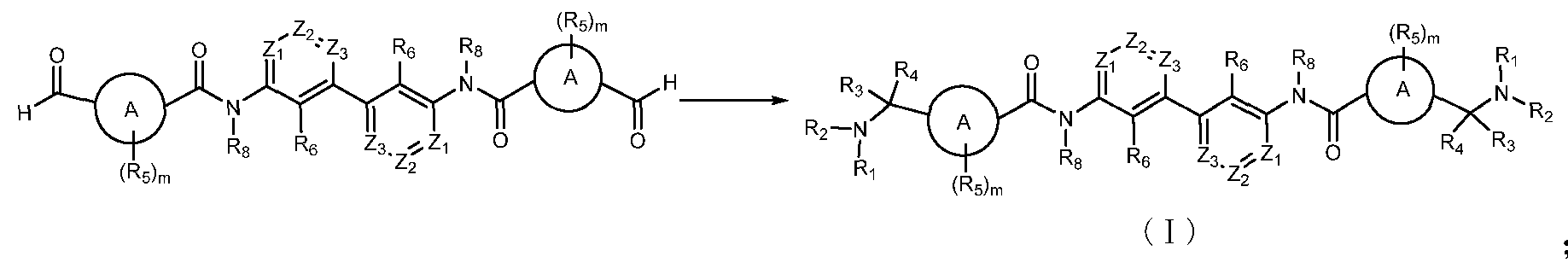
- a first aspect of the invention provides a compound of formula (I), a stereoisomer thereof, a prodrug or a pharmaceutically acceptable salt thereof:
- ring eight is selected from a 5-7 membered aromatic ring or a 5-7 membered heteroaryl ring;
- ⁇ , 1, 3 are each independently selected from (:(1111) or
- III, 1 ⁇ 2 are each independently selected from the group consisting of ammonia, hydrazine, light base, ( ⁇ 1-10 fluorenyl, ⁇ 2-10 chain group, ⁇ 2-10 chain block group, ⁇ 3-10 cyclodecyl group, ⁇ 3-10cyclodecyl 1-8 fluorenyl, 3-10 fluorenyl heterocyclic, 3-10 fluorenyl 1-8 fluorenyl, fluorene 5-10 aryl, fluorene 5-10 aryl (: 1- Or a 5-10 membered heteroaryl group, or 1 ⁇ 1 and 1 ⁇ 2 together with a nitrogen atom directly bonded thereto form a 3-10 membered heterocyclic group, the above group optionally further selected by one or more Since hydrazine, halogen, cyano, nitro, azide, (:1-10
- 3 ⁇ 4, 1 ⁇ 4 are each independently selected from the group consisting of ammonia, hydrazine, aryl, aryl, nitro, azide, 1-10 fluorenyl, ⁇ 2-10 chain
- the group is optionally further selected from one or more selected from the group consisting of hydrazine, halogen, cyano, nitro, azide, ( ⁇ 4()alkyl, Alkene
- 115 is selected from the group consisting of hydrogen, hydrazine, halogen, cyano, nitro, azide, alkyl, ( 32 ⁇ alkenyl, alkynyl,
- the above group is optionally further further selected from one or more selected from the group consisting of an anthraquinone, a cyano group, a nitro group, a nitro group, an azide group, Alkyl, ⁇ 2-10
- 116 is selected from the group consisting of hydrogen, hydrazine, halogen, cyano, nitro, azide, alkyl, Alkenyl, Alkynyl, ⁇ 3-10 cycloalkyl, 3-10 membered heterocyclic, ⁇ 5-10 aryl, 5-10 membered heteroaryl, - ⁇ 0-8-8( ⁇ 12 > - ⁇ 0 -8- ⁇ 13 >
- the group is optionally further selected from one or more selected from the group consisting of hydrazine, halogen, cyano, nitro, azide, Alkyl, (: ⁇ .alkenyl, ( : 2-10 chain block, substituted ⁇ 1-10 alkyl, ⁇ substituted ⁇ 1-10 ⁇ , ⁇ 3-10 ring, 3-1) Heterocyclic group, ⁇ 5-10 aryl group,
- the above group is optionally further selected from one or more selected from the group consisting of ruthenium, halogen, cyano, nitro, azide, ⁇ 1-10 , ⁇ 2-10 chain sieve group, ⁇ 2-10 chain block group, substituted ⁇ ⁇ ⁇ group, ⁇ substituted ⁇ ⁇ ⁇ group, ⁇ 3-10 ring yard group, 3-10 member heterocyclic group, ⁇ 5-1. aryl, 5-10 membered heteroaryl, - ⁇ -8-5(0 ⁇ 12 > -0)-8-0-1113,
- each of 118 is independently selected from the group consisting of ammonia, hydrazine, 1_10, ⁇ 2_10 chain, ⁇ 2_10 chain, ⁇ 3_10 ring, 3-10 membered heterocyclic , ⁇ 5-1.
- the above group is optionally further further selected from one or more selected from the group consisting of an anthracene, a cyano group, a nitro group, a nitro group, an azide group, ⁇ 2-10 chain sieve group, ⁇ 2-10 chain block group, substituted ⁇ 1-10 yard group, ⁇ substitution ( ⁇ 1-10 yard base, ⁇ 3-10 ring yard group, 3-10 member heterocyclic group, ⁇ 5-1 ⁇ aryl, 5-10 ⁇ aryl, _(].-8-8(0) 12, -0 ⁇ -8-0- ⁇ 3 %
- 119, 1110 are each independently selected from hydrogen, deuterium, hydroxy, alkyl, - 1 () alkenyl, (3 ⁇ 0 alkynyl group, a cycloalkyl group ⁇ 3-10, 3-10 membered heterocyclyl, ( Or an aryl group or a 5-10 membered heteroaryl group, or 119 together with a sulfur atom to which the hydrazine and its directly attached form a 3-10 membered heterocyclic group, the above group optionally further selected from one or more selected from the group consisting of hydrazine and halogen. , cyano, nitro, azido, ⁇ 1-10, ⁇ 2-10 chain sieve, ⁇ 2-10 chain block, substituted ⁇ 1-10, ⁇ substituted ⁇ 1-10
- Each is independently selected from the group consisting of hydrogen, hydrazine, halogen, cyano, nitro, azide, alkyl, (: 2 - 10 chain
- the above group is optionally further further selected from one or more selected from the group consisting of an anthraquinone, a cyano group, a nitro group, a nitro group, an azide group, Alkyl, ⁇ 2-10
- 5-10 membered heteroaryl, 5-10 membered heteroaryloxy or -1 ⁇ 151116 the above group optionally further selected from one or more selected from the group consisting of anthraquinone, a fluorene, a light group, a hydrazine, a hydrazine Base, ⁇ 1-10 oxy, ⁇ 3-10 cycloalkyl, ⁇ 3-10 ring oxime, 3-10 membered heterocyclic, 3-10 membered heterocyclooxy, 5-10 aryl, Substituting a 5-10 aryloxy group, a 5-10 membered heteroaryl group, a 5-10 membered heteroaryloxy group or a substituent of -1 ⁇ 11151116;
- 1114 each independently selected from hydrogen, a deuterium, hydroxy, alkyl ⁇ 1-10, ⁇ 1-10 alkoxy, (: 2-10 alkenyl, ⁇ 2-10 chain block group, ⁇ 3-10 Cycloalkyl, ⁇ 3-10 cyclodecyloxy, 3-10 membered heterocyclic, 3-10 membered heterocyclooxy, ⁇ 5-10 aryl, ⁇ 5-10 aryloxy, 5-10 membered Aryl, 5-10 membered heteroaryloxy or - 1 ⁇ 11151116, the above group optionally further selected from one or more selected from the group consisting of anthraquinone, a light, an amino group, a fluorene 1-10, a ⁇ 1-10 Alkoxy, ⁇ 3-10 ring, ⁇ 3-10 ring, oxy, 3-10 membered heterocyclic, aryl, Aryloxy, 5-10 membered heteroaryl, 5-10 membered heteroaryloxy or - ⁇ 151116
- Each of 15 and 1 ⁇ 16 is independently selected from the group consisting of ammonia, hydrazine, light base, ⁇ 1-10 oxy, ⁇ 1-10, ⁇ 2-10 chain, alkyncycloalkyl Aryl, 5-10 membered heteroaryl, sulfinyl, sulfonyl
- 3 ⁇ 45, 1 ⁇ 16 and its directly attached nitrogen atom together form a 4-1 10 membered heteroaryl group
- the above group optionally further selected from one or more selected from the group consisting of hydrazine, halogen, hydroxy, alkenyl, (: 2-10 chain block group, substituted 1-10 fluorenyl group, hydrazine substituted ⁇ ⁇ ⁇ group, 1-10 ⁇ ⁇ 3-10 cyclodecyloxy group, 3-10 membered heterocyclic group, 3-10 yuan Epoxy group, ⁇ 5-10 aryl group, ⁇ 5-1 ⁇ aryl oxygen Base, 5-10 yuan heterofang
- the compound of formula (the I), acceptable salt 1, 2, 2 3 are each independently selected from (a stereoisomer thereof, or a pharmaceutically acceptable prodrug: (1111) or each 1111
- Each is independently selected from the group consisting of hydrogen, hydrazine, fluorine, chlorine, cyano, methyl, isopropyl, vinyl, allyl, ethynyl, cyclopropyl, 3-oxetanyl, 3-azene heterocycle Butyl, phenyl, pyridyl, azole, triazol, methanesulfonyl, aminosulfonyl, methoxy, methoxyacyl, acetyl, acetoxy, amino, dimethylamino, aminoacyl or An acetylamino group, the above group optionally further selected from one or more selected from the group consisting of hydrazine, fluorine, chlorine, cyano, methyl, trifluoromethyl, cycloprop
- the salt 21, 2, 23 are each independently selected from the compound of formula acceptable (the I), a stereoisomer thereof, or a pharmaceutically acceptable prodrug (:(1111) or ⁇
- Each 1111 is independently selected from the group consisting of hydrogen, hydrazine, fluorine, chlorine, cyano, a group, a cyclopropyl group, a phenyl group, a pyridyl group, a diazoxide, a diazonium or a methoxy group, the above group optionally further selected from one or more selected from the group consisting of ruthenium, fluorine, chlorine, cyano, methyl, and tri Substituted by a substituent of a fluoromethyl group, a cyclopropyl group, a phenyl group, a pyridyl group, a methanesulfonyl group, a methoxy group or an amino group.
- Correction page (Article 91) ⁇ 0 2019/149183 ⁇ (:17(: ⁇ 2019/073594 is a preferred embodiment, wherein the compound of the formula (I), its stereoisomer, prodrug or a pharmaceutically acceptable salt thereof is selected from the group consisting of ammonia, ⁇ , ⁇ , ⁇ , ( ⁇ 1-4 ⁇ , ⁇ 2-4 chain coal, ⁇ 2-4 chain block, ⁇ 3-8cyclodecyl, 3-8 ⁇ heterocycle
- the compound of the formula (I), a stereoisomer thereof, a prodrug or a pharmaceutically acceptable salt thereof is selected from the group consisting of hydrogen, hydrazine, fluorine, chlorine, cyano, methyl, isopropyl.
- 116 of the compound of formula (I), a stereoisomer thereof, a prodrug or a pharmaceutically acceptable salt thereof is selected from the group consisting of hydrogen, hydrazine, fluorine, chlorine, cyano, methyl, ethyl , isopropyl, trimethyl, trifluoromethyl, difluoromethyl, cyanomethyl, aminomethyl, cyclopropylmethyl, methoxymethyl, vinyl, ethynyl, cyclopropyl, cyclobutyl base,
- the compound of the formula (I), a stereoisomer thereof, a prodrug or a pharmaceutically acceptable salt thereof is each independently selected from the group consisting of ammonia, hydrazine, amino, amino, and oxime 1- 4 ⁇ base, ⁇ 2-4 chain base, ⁇ 2-4 chain block base, ⁇ 3-8 ring ⁇
- the pharmaceutically acceptable salts are independently selected from the group consisting of hydrogen, hydrazine, fluorine, chlorine, cyano, methyl, cyclopropyl, phenyl, pyridyl, azole, triazole or methoxy, Alternatively, 3 ⁇ 4 together with 114 and its directly attached carbon atom form a cyclopropyl, cyclobutyl, 3-oxetanyl or 3-azetidinyl group, the above group optionally further selected by one or more Substituting a substituent of hydrazine, fluorine, chlorine, cyano, methyl, trifluoromethyl, cyclopropyl, phenyl, pyridyl, methanesulfonyl, methoxy or amino.
- the compound of the formula (I), a stereoisomer thereof, a prodrug or a pharmaceutically acceptable salt thereof is selected from the group consisting of ammonia, hydrazine, , Atmosphere group, (1-4 ⁇ firing group, ⁇ 2-4 refining chain group, ⁇ 2-4 chain block group, ⁇ 3-8 firing ring group, a heterocyclic 3-8 Wu
- the above group is further optionally further selected from one or more selected from the group consisting of hydrazine, halogen, cyano, ( ⁇ -4 alkyl, ( 2-4 -alkenyl, ⁇ 2-4 alkynyl) , halo-substituted (: 1-4 alkyl, (: 3-8 cycloalkyl, 3-8 membered heterocyclyl, (: 5-8 aryl, 5-8
- 115 of the compound of the formula (I), a stereoisomer thereof, a prodrug or a pharmaceutically acceptable salt thereof is selected from the group consisting of hydrogen, hydrazine, fluorine, chlorine, cyano, methyl, ethyl, Isopropyl, vinyl, allyl, ethynyl, cyclopropyl, cyclobutyl, 3-oxetanyl, 3-azetidinyl, phenyl, pyridyl, diazo, trinitrogen Wow, -8?
- the above group optionally further selected from one or more selected from the group consisting of hydrazine, fluorine, chlorine, cyano, methyl, trifluoromethyl, cyclopropyl, phenyl, pyridyl, azole, triazole, A Substituted by a substituent of a sulfonyl group, an aminosulfonyl group, a methoxy group or an amino group, the above group is further optionally further selected from one or
- the compound of the formula (I), a stereoisomer thereof, a prodrug or a pharmaceutically acceptable salt thereof is selected from the group consisting of 5 a 6-membered aromatic ring or a 5-6-membered heteroaryl ring.
- the compound of the formula (I), a stereoisomer thereof, a prodrug or a pharmaceutically acceptable salt thereof is selected from the group consisting of group:
- the compound of the formula (I) wherein the compound of the formula (I), a stereoisomer thereof, a prodrug or a pharmaceutically acceptable salt thereof has the compound structure of the following formula (II):
- 1 ⁇ 5 is selected from the group consisting of ammonia, hydrazine, gas, chlorine, amino, methyl, ethyl, isopropyl, ethene, propyl, ethyl, cyclopropyl, cyclobutyl, 3-oxocyclo Butyl, 3-azetidinyl, phenyl, pyridyl, diazo, trinitrogen, -8?
- 116 is selected from the group consisting of hydrogen, hydrazine, fluorine, chlorine, cyano, methyl, ethyl, isopropyl, vinyl, allyl, ethynyl, cyclopropyl, cyclobutyl, 3-oxetanyl, 3-azetidinyl, phenyl, pyridyl, diaza, triazole, methylsulfonyl, aminosulfonyl, methoxy, methoxy, acetyl, acetoxy, amino, dimethyl
- An amino group, an aminoacyl group or an acetylamino group, the above group optionally further selected from one or more selected from the group consisting of hydrazine, fluorine, chlorine, cyano, methyl, trifluoromethyl, cyclopropyl, phenyl, pyridyl, methylsulfonate Substituted by a substituent of an acyl group
- Each! is independently selected from the group consisting of hydrogen, hydrazine, fluorine, chlorine, cyano, methyl, isopropyl, vinyl, allyl, ethynyl, cyclopropyl, 3-oxetanyl, 3- Azetidinyl, phenyl, pyridyl, azole, triazole, methylsulfonyl, aminosulfonyl, methoxy, methoxy, acetyl, acetoxy, amino, dimethylamino, Amino correction page (Article 91) ⁇ 0 2019/149183 ⁇ (:17(: ⁇ 2019/073594 acyl or acetamido, the above group optionally further one or more selected from the group consisting of hydrazine, fluorine, chlorine, cyano, methyl, trifluoromethyl Substituted with a substituent of a cyclopropyl group, a phenyl group, a
- 116 of the compound of formula (I), a stereoisomer thereof, a prodrug or a pharmaceutically acceptable salt thereof is selected from the group consisting of hydrogen, hydrazine, fluorine, chlorine, cyano, methyl, ethyl , isopropyl, trimethyl, trifluoromethyl, difluoromethyl, cyanomethyl, aminomethyl, cyclopropylmethyl, methoxymethyl, vinyl, ethynyl, cyclopropyl, cyclobutyl , 3-oxetanyl, 3-azetidinyl, methoxy, trimethylmethoxy, difluoromethoxy or trifluoromethoxy;
- Each of ! is independently selected from the group consisting of hydrogen, hydrazine, fluorine, chlorine, cyano, methyl, cyclopropyl, phenyl, pyridyl, azole, triazole or methoxy, and the above groups are optionally further Substituted by one or more substituents selected from the group consisting of hydrazine, fluorine, chlorine, cyano, methyl, trifluoromethyl, cyclopropyl, phenyl, pyridyl, methylsulfonyl, methoxy or amino.
- the compound of the formula (I) wherein the compound of the formula (I), a stereoisomer thereof, a prodrug or a pharmaceutically acceptable salt thereof has the compound structure of the following formula (III):
- the compound of the formula (I) wherein the compound of the formula (I), a stereoisomer thereof, a prodrug or a pharmaceutically acceptable salt thereof has the formula (IV compound, formula (1) , formula (1 ( ⁇ compound or formula (1 ⁇ ) compound structure:
- each of 111, 3 ⁇ 4 is independently as described for the compound of formula (I).
- the compound of the formula (I), a stereoisomer thereof, a prodrug or a pharmaceutically acceptable salt thereof is each independently selected from the group consisting of hydrogen, hydrazine, hydroxyl group, (: 1- 4-alkyl, (: 2-4 alkenyl, ⁇ : 2-4 alkynyl, ⁇ : 3-8 cyclodecyl, 3-8 cycloalkyl 1-4 fluorenyl, 3-8 membered heterocyclic , a 5-8 aryl group or a 5-8 membered heteroaryl group, or 111 and 112 together with a nitrogen atom directly bonded thereto form a 3-8 membered heterocyclic group, wherein the hetero atom in the heterocyclic group is selected from the group consisting of X, 0, 8 and/or 81, the above group optionally further selected from one or more selected from the group consisting of hydrazine
- the compound of the formula (I), a stereoisomer thereof, a prodrug or a pharmaceutically acceptable salt thereof is selected from the group consisting of hydrogen, hydrazine or methyl; 112 is selected from the group consisting of hydrogen, hydrazine, hydroxy, ( ⁇ -4 alkyl, (: 3-8 cycloalkyl, ( : 3 -8 cycloalkyl (31-4 fluorenyl, 3-8 membered heterocyclyl, fluorene 5-8 aryl or 5-8 membered)
- the hetero atom in the heterocyclic group is selected from the group consisting of 1 ⁇ , 0, 8, and/or 8 ⁇ , the above group
- the hetero atom in the heterocyclic group is selected from
- 117 is selected from hydrogen, deuterium, ( ⁇ -4 alkyl, (: 2-4 alkenyl group, (: 3-6 cycloalkyl, 3-6 membered heterocyclyl, phenyl, 5-8 membered heteroaryl Or a methylsulfonyl group or an acetyl group, the above group optionally further selected from one or more selected from the group consisting of hydrazine, fluorine, chlorine, cyano, hydroxy, methyl, ethyl, trifluoromethyl, difluoromethyl, triterpene Methyl, dimethyl, cyclopropyl, phenyl, dinitrogen
- 119, 1 ⁇ 0 are each independently selected from the group consisting of hydrogen, hydrazine, hydroxy, ( ⁇ -4 alkyl, (: 2 -4 alkenyl, (: 3 -6 cycloalkyl, 3-6 membered heterocyclyl, benzene) Or a 5-8 membered heteroaryl group, or 119 and 1110 together with a sulfur atom directly attached thereto form a 3-6 membered heterocyclic group, the above group optionally further selected from one or more selected from the group consisting of ruthenium, fluorine, and chlorine , cyano, hydroxy, methyl, ethyl, trifluoromethyl, difluoromethyl, trimethyl, dimethyl, cyclopropyl, phenyl, diazole, triazol, methoxy Substituted with a substituent of an ethoxy group, a carboxyl group, a methoxycarbonyl group, an ethoxycarbonyl group, an acetyl group
- Each of 11 12 is independently selected from the group consisting of hydrogen, hydrazine, hydroxy, halogen, ( ⁇ -4 alkyl, (: 3-6 cycloalkyl, 3-6 membered heterocyclyl, phenyl, 5-8 membered heteroaryl) Or the group - 151116, the above group optionally further selected from one or more selected from the group consisting of hydrazine, fluorine, chlorine, light, a few, ( ⁇ 1-4 fluorenyl, ( ⁇ 1-4 methoxy, oxime) Substituted with a substituent of 3-6 cyclodecyl, ⁇ 3-6 cyclodecyloxy, 3-6 fluorenyl heterocyclyl, phenyl, 5-8 membered heteroaryl, amino or dimethylamino;
- Each of 11 13 is independently selected from the group consisting of hydrogen, hydrazine, 0-4 fluorenyl, (: 2-4 alkenyl, (3-6 cycloalkyl, 3-6 membered heterocyclyl, phenyl or 5-)
- An 8-membered heteroaryl group the above group optionally further selected from one or more selected from the group consisting of hydrazine, halogen, hydroxy, carbonyl, cyano, ( ⁇ 1-4 fluorenyl, ( ⁇ 1-4 methoxy, ⁇ 3- 6-ring fluorenyl, fluorene 3-6 cyclodecyloxy, 3-6 fluorenyl heterocyclic, 3-6 fluorenyloxy, phenyl, phenoxy, 5-8 membered heteroaryl, 5-8 member Substituted by a heteroaryloxy group or a substituent of -1 ⁇ 151116;
- Each 1114 is independently selected from the group consisting of hydrogen, hydrazine, hydroxy, (: 1-4 alkyl, (: 1-4 methoxy), (: 2-4 alkenyl, (: 2-4 alkynyl, ( : 3_6 cycloalkyl, (: 3_6 cycloalkoxy, 3-6 membered heterocyclic, 3-6 membered heterocyclooxy, phenyl, phenoxy, 5-8 membered heteroaryl, 5-8 member a heteroaryloxy group or -1 ⁇ 151116, the above group optionally further selected from one or more selected from the group consisting of hydrazine, halogen, light base, an aryl group, ( ⁇ 1-4 fluorenyl, ( ⁇ 1-4 methoxy, ⁇ 3-6cyclodecyl, ⁇ 3-6cyclodecyloxy, 3-6 ⁇ heterocyclyl, 3-6 ⁇ heterocyclyloxy, phenyl, phenoxy, 5-8 membered heteroaryl, 5 Substit
- Each of 1115, 1116 is independently selected from the group consisting of hydrogen, hydrazine, hydroxy, (: 1-4 alkoxy, (: 1-4 alkyl, (: 2-4 alkenyl), (: 2 -4 alkynyl) , (: 3_6 cycloalkyl, 3-6 membered heterocyclic, phenyl, 5-8 membered heteroaryl, sulfinyl, sulfonyl, methylsulfonyl, isopropylsulfonyl, cyclopropylsulfonyl, p- a tosyl group, an aminosulfonyl group, a dimethylaminosulfonyl group, an amino group, a monoalkylamino group, a dialkylamino group or a ⁇ -4 alkanoyl group, the above group optionally further selected from one or more selected from the group consisting of hydrazine, halogen, Hydroxy, (: 1-4
- 3 ⁇ 45, 1 ⁇ 16 and its directly attached nitrogen atom together form a 4-6 membered heterocyclic group or a 4-6 membered heteroaryl group, and the above group is optionally further selected from one or more selected from the group consisting of hydrazine, halogen, and hydroxy group.
- I ⁇ is independently 0, 1, or 2.
- the compound of the formula (I), a stereoisomer thereof, a prodrug or a pharmaceutically acceptable salt thereof includes, but is not limited to, the following compounds:
- a process for the preparation of a compound of the formula (I), a stereoisomer thereof, a prodrug or a pharmaceutically acceptable salt thereof comprising the steps of:
- a third aspect of the invention provides a pharmaceutical composition comprising a compound of the above formula (I), a stereoisomer thereof, a prodrug or a pharmaceutically acceptable compound thereof Receiving a salt and a pharmaceutically acceptable carrier.
- a fourth aspect of the invention provides a compound of the above formula (I), a stereoisomer thereof, a prodrug or a pharmaceutically acceptable salt thereof, or a pharmaceutical composition as described above for the preparation of a prophylactic and/or Or for the treatment of a drug associated with the disease mediated by the ?-1/?1 signaling pathway.
- the signaling pathway mediated diseases are selected from cancer or tumors, immune related diseases and disorders, infectious diseases, infectious diseases or metabolic diseases.
- the infectious disease is selected from the group consisting of a bacterial infectious disease, a viral infectious disease, or a fungal infectious disease.
- the aforementioned cancer or tumor is selected from the group consisting of lymphoma (including but not limited to lymphocytic lymphoma, primary central nervous system lymphoma, buty cell lymphoma, diffuse large 8-cell lymphoma, follicular center) Lymphoma, Hodgkin's lymphoma, non-Hodgkin's lymphoma or primary mediastinal large 6-cell lymphoma), sarcoma (including but not limited to Kaposi's sarcoma, fibrosarcoma, liposarcoma, chondrosarcoma, osteosarcoma, Leiomyosarcoma, rhabdomyosarcoma, soft tissue sarcoma, angiosarcoma or lymphangiosarcoma), melanoma, glioblastoma, synovial tumor, meningioma, biliary tract, thymic neoplasm, neuroma
- lymphoma
- the aforementioned immune-related diseases and disorders are selected from the group consisting of rheumatoid arthritis, renal failure, lupus erythematosus, asthma, psoriasis, ulcerative colitis, pancreatitis, allergy, fibrosis, anemia fibromyalgia, Alz Mellow heart disease, congestive heart failure, stroke, aortic stenosis, arteriosclerosis, osteoporosis, Parkinson's disease, infection, Crohn's disease, ulcerative colitis, allergic contact dermatitis and eczema, systemic sclerosis And multiple sclerosis.
- the aforementioned infectious disease or infectious disease is selected from the group consisting of sepsis, liver infection, 111, hepatitis A, hepatitis B, hepatitis C, hepatitis D, herpes virus, papilloma virus or influenza.
- a fifth aspect of the present invention provides a compound of the above formula (I), a stereoisomer thereof, a prodrug or a pharmaceutically acceptable substance thereof.
- the fifth aspect of the invention provides a compound of the above formula (I), a stereoisomer thereof, a prodrug or a pharmaceutically acceptable substance thereof.
- a salt, or a pharmaceutical composition as described above for use as a prophylactic and/or therapeutic agent for cancer or tumors, immune related diseases and disorders, infectious diseases, infectious diseases or metabolism mediated by the ?-1/?1 signaling pathway Drugs for sexual diseases.
- a method for cancer or tumor, immune-related diseases and disorders, infectious diseases, infectious diseases or metabolic diseases mediated by the ⁇ -1/? ⁇ 1 signaling pathway comprising administering to the patient a compound of the above formula (I) A construct, a prodrug or a pharmaceutically acceptable salt thereof, or a pharmaceutical composition as described above.
- a seventh aspect of the invention provides an enhancement, stimulation, modulation and/or increase in a subject in need thereof
- a method of signaling pathway-mediated immune response comprising administering to a patient a compound of the above formula (I), a stereoisomer thereof, a prodrug or a pharmaceutically acceptable salt thereof, or a pharmaceutical composition as described above.
- An eighth aspect of the invention provides a method of inhibiting growth, proliferation or metastasis of a tumor cell comprising administering to a patient a compound of the above formula (I), a stereoisomer thereof, a prodrug or a pharmaceutically acceptable salt thereof, or a combination thereof Things.
- a ninth aspect of the invention provides a treatment by A method of signaling pathway-mediated infectious diseases, infectious diseases, metabolic diseases or disorder diseases, comprising administering to a patient a compound of the above formula (I), a stereoisomer thereof, a prodrug or a pharmaceutically acceptable salt thereof, or The aforementioned pharmaceutical composition.
- BEST MODE FOR CARRYING OUT THE INVENTION The inventors of the present application have extensively and intensively studied to develop, for the first time, a series of compounds of the general formula (I), especially fully symmetric compounds, which are differentiated in the overall structure under symmetry. This series of compounds is inhibited
- biaryl derivatives their preparation and their use in pharmacy.
- Serialization of the invention The interaction has a strong inhibitory effect and can be widely used in the preparation of prevention and / or treatment? Drugs that are mediated by the ⁇ -1/? ⁇ 1 signaling pathway for cancer or tumors, immune-related diseases and disorders, infectious diseases, infectious diseases, or metabolic diseases, are expected to be developed into a new generation? 0-1/?0 1 inhibitor. On the basis of this, the present invention has been completed.
- Alkyl means a straight or branched saturated aliphatic hydrocarbon group, for example, .
- Alkyl means a straight-chain alkyl group having from 1 to 10 carbon atoms and a branched alkyl group, including but not limited to methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, tert Butyl, sec-butyl, n-pentyl, 1,1-dimethylpropyl, 1,2-dimethylpropyl, 2,2-dimethylpropyl, 1-ethylpropyl, 2- Methyl butyl, 3-methylbutyl, n-hexyl, 1-ethyl-2-methylpropyl, 1,1,2-trimethylpropyl, 1,1-dimethylbutyl, 1 , 2 - dimethyl butyl, 2, 2 - dimethyl butyl, 1, 3- dimethyl butyl, 2-ethyl butyl,
- the alkyl group may be optionally substituted or unsubstituted, and when substituted, the substituent is preferably one or more of the following groups, independently selected from the group consisting of hydrazine, halogen, cyano, nitro, azide, alkyl, Alkenyl, Alkenyl group, substituted ⁇ 1-1 fluorenyl group, fluorene substituted ⁇ 1-1 fluorenyl group, ⁇ 3-1 ⁇ cyclodecyl group, 3-10 membered heterocyclic group, ⁇ 5-1 ⁇ aryl group, 5- 10 yuan
- Cycloalkyl or “carbocyclic” refers to a saturated or partially unsaturated monocyclic or polycyclic cyclic hydrocarbon substituent, for example, cycloalkyl” refers to a cycloalkyl group of from 3 to 10 carbon atoms, which is divided into single rings.
- a cycloalkyl, polycyclic cycloalkyl group wherein: a monocyclic cycloalkyl group includes, but is not limited to, a cyclopropyl group, a cyclobutyl group, a cyclopentyl group, a cyclopentenyl group, a cyclohexyl group, a cyclohexenyl group, a cyclohexadiene group.
- Base cycloheptyl, cycloheptatrienyl, cyclooctyl and the like.
- Polycyclic cycloalkyl groups include spiro, fused, and bridged cycloalkyl groups.
- “Spirocycloalkyl” refers to a polycyclic group that shares a carbon atom (called a spiro atom) between the individual rings. These may contain one or more double bonds, but none of the rings have a fully conjugated II electronic system.
- the spirocycloalkyl group is classified into a monospirocycloalkyl group, a bispirocycloalkyl group or a polyspirocycloalkyl group depending on the number of common spiro atoms between the ring and the ring, and the spirocycloalkyl group includes, but is not limited to:
- fused cycloalkyl means an all-carbon polycyclic group in which each ring in the system shares an adjacent pair of carbon atoms with other rings in the system, wherein one or more of the rings may contain one or more double bonds, but None of the rings have a fully conjugated electronic system. Depending on the number of constituent rings, it may be classified into bicyclic, tricyclic, tetracyclic or polycyclic fused ring alkyl groups, and fused cycloalkyl groups include, but are not limited to:
- Bridge cycloalkyl refers to an all-carbon polycyclic group in which two rings share two carbon atoms that are not directly bonded. These may contain one or more double bonds, but none of the rings have a fully conjugated II electronic system. . Depending on the number of constituent rings, it may be classified into bicyclic, tricyclic, tetracyclic or polycyclic bridged cycloalkyl groups, and bridged cycloalkyl groups include, but are not limited to:
- the cycloalkyl ring may be fused to an aryl, heteroaryl or heterocycloalkyl ring, wherein the ring to which the parent structure is attached is a cycloalkyl group, including but not limited to indanyl, tetrahydronaphthyl , benzocycloheptyl and the like.
- the cycloalkyl group may be optionally substituted or unsubstituted, and when substituted, the substituent is preferably one or more of the following groups, independently selected from the group consisting of hydrazine, halogen, cyano, nitro, azide, ( 3 ⁇ 0 alkyl, Alkenyl, Alkenyl group, substituted 1-1 fluorenyl group, fluorene substituted ⁇ 1-1 fluorenyl group, ⁇ 3-1 ⁇ cyclodecyl group, 3-10 membered heterocyclic group, ⁇ 5-1 ⁇ aryl group, 5-10
- Heterocyclyl or “heterocycle” refers to a saturated or partially unsaturated monocyclic or polycyclic cyclic hydrocarbon substituent wherein one or more of the ring atoms are selected from nitrogen, oxygen, silicon or 8 (0 ⁇ (where is an integer) 0, 1, 2) heteroatoms, but not including -0-0 -, - the ring portion, the remaining ring atoms are carbon.
- “4-10 membered heterocyclic group” means a ring group containing 4 to 10 ring atoms
- 3-6 membered heterocyclic group means a ring group containing 3 to 6 ring atoms.
- Monocyclic heterocyclic groups include, but are not limited to, pyrrolidinyl, piperidinyl, piperazinyl, morpholinyl, thiomorpholinyl, homopiperazinyl and the like.
- the polycyclic heterocyclic group includes a spiro ring, a fused ring, and a heterocyclic group of a bridged ring.
- spiroheterocyclyl refers to a polycyclic heterocyclic group in which one atom (referred to as a spiro atom) is shared between a single ring, wherein one or more ring atoms are selected from nitrogen, oxygen, silicon or 3 (0) where is an integer 0 1, 2, 2) heteroatoms, the remaining ring atoms are carbon. These may contain one or more double bonds, but none of the rings have a fully conjugated I! electronic system.
- the spiroheterocyclyl group is classified into a monospiroheterocyclic group, a dispirocyclic heterocyclic group or a polyspiroheterocyclic group depending on the number of common spiro atoms between the ring and the ring.
- Spiroheterocyclyl includes, but is not limited to:
- “Fused heterocyclic group” means that each ring in the system shares an adjacent pair of atomic polycyclic heterocyclic groups with other rings in the system, and one or more rings may contain one or more double bonds, but none
- the ring has a fully conjugated electronic system wherein one or more ring atoms are selected from nitrogen, oxygen, silicon or 3 (0) wherein I. is a heteroatom of the integers 0, 1, 2) and the remaining ring atoms are carbon.
- the bicyclic, tricyclic, tetracyclic or polycyclic fused heterocycloalkyl group may be classified according to the number of constituent rings, and the condensed heterocyclic group includes, but is not limited to:
- Bridge heterocyclyl refers to a polycyclic heterocyclic group in which two rings share two atoms which are not directly bonded, and these may contain one or more double bonds, but none of the rings have a fully conjugated electronic system, wherein The one or more ring atoms are selected from nitrogen, oxygen, silicon or 3 (0) wherein r is a hetero atom of the integers 0, 1, 2) and the remaining ring atoms are carbon.
- the bicyclic, tricyclic, tetracyclic or polycyclic bridged heterocyclic group may be classified according to the number of constituent rings, and the bridged heterocyclic group includes but is not limited to:
- the heterocyclyl ring may be fused to an aryl, heteroaryl or cycloalkyl ring, wherein the ring to which the parent structure is attached is a heterocyclic group, including but not limited to:
- the heterocyclic group may be optionally substituted or unsubstituted, and when substituted, the substituent is preferably one or more of the following groups, independently selected from the group consisting of an anthracene, a amide, an amino group, a nitro group, an azide group, and 1- 10 fluorenyl, ⁇ 2-10 chain dilute, ⁇ 2-10 chain block, substituted ⁇ 1-1 fluorenyl, hydrazine substituted ⁇ 1-1 fluorenyl, ⁇ 3-1 fluorenyl fluorenyl, 3- 10 membered heterocyclic group, ⁇ 5-1 ⁇ aryl group, 5-10
- Aryl or "aromatic ring” means an all-carbon monocyclic or fused polycyclic ring (ie, a ring that shares a pair of adjacent carbon atoms), a polycyclic ring having a conjugated II electron system (ie, having an adjacent a ring for a carbon atom), for example, .
- Aryl means an all-carbon aryl group having 5 to 10 carbons
- “5-10 membered aryl” means an all-carbon aryl group having 5 to 10 carbons, including but not limited to phenyl and naphthyl.
- the base ring may be fused to a heteroaryl, heterocyclyl or cycloalkyl ring, wherein the ring to which the parent structure is attached is an aryl ring, including but not limited to:
- the aryl group may be substituted or unsubstituted, and when substituted, the substituent is preferably one or more of the following groups, independently selected from the group consisting of hydrazine, halogen, cyano, nitro, azide, Alkyl, ( : 2-1() alkenyl, ( : 2 - 1) alkynyl, halogen
- heteroaryl refers to a heteroaromatic system containing 5-10 ring atoms
- Heteroaryl refers to a heteroaromatic system containing 5-8 ring atoms, including but not limited to furyl, thienyl, pyridyl, pyrrolyl, alkyl pyrrolyl, pyrimidinyl, pyrazinyl, imidazolyl, tetra Azolyl and the like.
- the heteroaryl ring may be fused to an aryl, heterocyclic or cycloalkyl ring wherein the ring to which the parent structure is attached is a heteroaryl ring including, but not limited to:
- the heteroaryl group may be optionally substituted or unsubstituted, and when substituted, the substituent is preferably one or more of the following groups, independently selected from the group consisting of hydrazine, halogen, cyano, nitro, azide, ( 3 ⁇ 0 alkyl, Alkenyl, Alkenyl group, substituted ⁇ 1-1 fluorenyl group, fluorene substituted ⁇ 1-1 fluorenyl group, ⁇ 3-1 ⁇ cyclodecyl group, 3-10 membered heterocyclic group, ⁇ 5-1 ⁇ aryl group, 5- 10
- Alkenyl refers to an alkyl group as defined above consisting of at least two carbon atoms and at least one carbon-carbon double bond, for example. Alkenyl refers to a straight or branched chain alkenyl group containing from 2 to 10 carbons. These include, but are not limited to, vinyl, 1-propenyl, 2-propenyl, 1-, 2- or 3-butene, and the like.
- the alkenyl group may be substituted or unsubstituted, and when substituted, the substituent is preferably one or more of the following groups, independently selected from the group consisting of hydrazine, halogen, cyano, nitro, azide, alkyl, hydrazine.
- Alkynyl means an alkyl group as defined above, consisting of at least two carbon atoms and at least one carbon-carbon triple bond, for example. Alkynyl refers to a straight or branched alkynyl group containing from 2 to 10 carbons. These include, but are not limited to, ethynyl, 1-propynyl, 2-propenyl, 1 _ , 2 - or 3-butyl, and the like.
- the alkynyl group may be substituted or unsubstituted, and when substituted, the substituent is preferably one or more of the following groups, independently selected from the group consisting of anthraquinone, an aryl group, an aryl group, a nitro group, an azide group, and a 1-10 fluorene group.
- Base ⁇ 2-10 chain pain group, ⁇ 2-10 chain block group, substituted 1-1 fluorenyl group, hydrazine substituted (31-10 fluorenyl group, ⁇ 3-1 ⁇ ring yard group, 3-10 membered heterocyclic ring)
- Base ⁇ 5-1 ⁇ aryl, 5-10 yuan
- Alkyloxy groups of carbon include, but are not limited to, methoxy, ethoxy, propoxy, butoxy, and the like.
- the alkoxy group may be optionally substituted or unsubstituted, and when substituted, the substituent, preferably one or more of the following groups, independently selected from the group consisting of an anthracene, an aryl group, an aryl group, a nitro group, an azide group, 1-10 thiol, ⁇ 2-10 chain pain base, ⁇ 2-10 chain block, substitution ( ⁇ 1-10 fen, ⁇ substituted 1-10 fluorenyl, ⁇ 3-10 fluorenyl, 3-10 Metacyclic heterocyclic group, ⁇ 5-10 aryl group,
- Cycloalkoxy refers to - ⁇ -cycloalkyl, wherein cycloalkyl is as defined above, for example, “Cycloalkoxy” means a cycloalkyloxy group containing from 3 to 10 carbons including, but not limited to, cyclopropoxy, cyclobutoxy, cyclopentyloxy, cyclohexyloxy and the like.
- Heterocyclyloxy means a fluorenyl-heterocyclic group, wherein the definition of the heterocyclic group is as described above, for example, "Heterocyclyloxy” means a heterocyclic oxy group having 3 to 10 carbons including, but not limited to, azetiyloxy, oxetanyloxy, azacyclopentyloxy, nitrogen, oxa Cyclohexyloxy and the like.
- the heterocyclic oxy group may be optionally substituted or unsubstituted, and when substituted, the substituent is preferably one or more of the following groups, independently selected from the group consisting of hydrazine, halogen, cyano, nitro, azide, 0: ⁇ () alkyl, alkenyl, (: 2 - 1 ⁇ chain fast radical, substituted 1-10 fluorenyl, hydrazine substituted 1-10 yard, ⁇ 3-10 ring yard, 3-10 yuan Ring group, ⁇ 5-10 aryl group,
- -0)-8-0-1113 means that the oxygen atom in -0-1113 is bonded to (:0-8 alkyl, wherein 0)alkyl refers to a bond, (:1-8 alkyl is as defined above Said.
- "-0)-8-pass 151116" means that the nitrogen atom in -Nen 151116 is attached to the -8 alkyl group, wherein (: 0 alkyl means a bond, and (: 1-8 alkyl is as defined above).
- "-0)-8 - 3 ⁇ 45)-(:(0)1114" means -111 5 )-(:(0)1114 has a nitrogen atom attached to the 0)-8 alkyl group, wherein 0)alkyl means key, ( ⁇ _ 8 alkyl group defined as described above.
- Hal-substituted alkyl refers to a hydrogen on the alkyl group optionally substituted by a fluorine, chlorine, bromine or iodine atom, including but not limited to difluoromethyl, dichloromethyl. Base, dibromomethyl, trifluoromethyl, trichloromethyl, tribromomethyl and the like.
- Hydrogen is optionally substituted with 1-10 carbon alkyl groups substituted by deuterium atoms. These include, but are not limited to, monomethyl, dimethyl, trimethyl, and the like.
- Halogen means fluoro, chloro, bromo or iodo.
- heterocyclic group optionally substituted by an alkyl group means that an alkyl group may be, but is not necessarily, present, including the case where the heterocyclic group is substituted by an alkyl group and the case where the heterocyclic group is not substituted by an alkyl group.
- Substituted means that one or more hydrogen atoms in the group are each independently substituted with a corresponding number of substituents. It goes without saying that the substituents are only in their possible chemical positions, and those skilled in the art will be able to determine (by experiment or theory) substitutions that may or may not be possible without undue effort. For example, an amino group or a hydroxyl group having a free hydrogen may be unstable when combined with a carbon atom having an unsaturated bond such as an olefin.
- “Pharmaceutical composition” means a mixture comprising one or more of the compounds described herein, or a physiologically/pharmaceutically acceptable salt or prodrug thereof, and other chemical components, as well as other components such as physiological/pharmaceutically acceptable carriers. And excipients.
- the purpose of the pharmaceutical composition is to promote administration to an organism, to facilitate absorption of the active ingredient and to exert biological activity.
- the present invention will be further described in detail with reference to the accompanying drawings, but not by way of limitation.
- the structure of the compound of the present invention is through nuclear magnetic To determine. ⁇ 111 Chemical shift (5) is given in parts per million. The determination of ⁇ 111 is Eight eight ⁇ ⁇ :?
- Liquid chromatography-mass spectrometry For measurement 6120 mass spectrometer. The measurement was performed using an Agilent 12000 ⁇ high pressure liquid chromatograph ⁇ ! ⁇ (:18 150 X 4.6 111111 column) and ⁇ Vaters 2695-2996 high pressure liquid chromatograph ( ⁇ 1 ⁇ 1 ⁇ (:18 150 X 4.6 111111 column) ).
- Column chromatography generally uses Yantai Huanghai silica gel 200 ⁇ 300 mesh silica gel as a carrier.
- the starting materials in the examples of the invention are known and commercially available or can be synthesized or synthesized according to methods known in the art.
- reaction solution is supplemented with 1,4 - dioxane (10 ⁇ 1 ⁇ and water (8 111 ⁇ , then added 3 ⁇ 4 (: ⁇ 3 (3.71, 26.89 ⁇ 1111 ⁇ 1) and ⁇ (1 (( ⁇ (:12(0.655, 0.90 1 ⁇ 1) Pumping, nitrogen protection. Heating to 951: reaction for 2 hours. The reaction was completed, and the reaction mixture was concentrated under reduced pressure. /£8:1) Get 2,2_-dimethyl- Biphenyl] -3,3 ⁇ -diamine (1.8, yield: 95%). 81): 213 [?4+11
- the third step dimethyl 6,6'-(((2,2'-dimethyl-[1,1'-biphenyl 3 ⁇ 4-3,3'-diyl) bis(azacycloalkanyl)) Synthesis of bis(carbonyl)dinitrobutyrate
- Step 4 Synthesis of (2,2'-dimethyl-[1,1'-biphenyl 3 ⁇ 4-3,3'-diyl)bis(5-(hydroxymethyl)-2-pyridineamide)
- Step 5 Synthesis of (2,2'-dimethyl-[1,1'-biphenyl]-3,3'-diyl)bis(5-formyl-2-pyridine amide)
- Second step (2,2'-dimethyl-[1,1'-biphenyl 3 ⁇ 4-3,3'-diyl) bis(4-methyl-5-vinyl-2-pyridine amide) synthesis
- Step 5 (2,2'-Dimethyl-[1,1'-biphenyl]-3,3'-diyl)bis(4-methoxy-5-vinyl-2-pyridineamide )Synthesis 5 ⁇ , 6.8 Placed in 1.4-dioxane (50 1111 ⁇ ) and water ( 10 1111 ⁇ ), followed by potassium carbonate (2.82 20.4 111111 ⁇ 1), acetonitrile trifluoroborate (9.1 ⁇ , 68.0 ⁇ ) 1 ⁇ 1 ⁇ 1) and [1,1'-bis(diphenylphosphino)ferrocene] dichloride button (500 113 ⁇ 4, 0.68 111111 ⁇ 1).
- Step 6 Synthesis of (2,2'-dimethyl-[1,1biphenyl 3 ⁇ 4-3,3'-diyl)bis(5-formyl-4-methoxy-2-pyridine amide)
- Step 2 Synthesis of ethyl 4-cyclopropyl-6-vinyl nicotine
- Second step Synthesis of 2-((dimethyl carbonyl-6-sulfaninyl)amino) acetonitrile
- Tetrahydrothiophene-1 -oxidation 500 13 ⁇ 4, 4.8 11111101 was added to methanol (20 11 ⁇ ), then 111 (0 8 ( ⁇ 4 (4.64 g, 14.4 and carbamic acid according to (1.5, 19.2 ⁇ 1111 ⁇ 1), The reaction was stirred at room temperature for 1 hour. After completion of the reaction, the mixture was concentrated, and the residue was applied tojjjjjjjj , yield: 102.2
- Step 2 Synthesis of 2-((1 -lightyltetrahydro-6-thiophene-1-ylidene)amino)acetonitrile
- the third step synthesis of 1-((2-aminoethyl)imino)tetraammine-111-6-thiophene-1 -oxidation
- the NF-KB-IUC reporter plasmid and human PD-1 plasmid were transfected into Jurkat cells to establish a stable cell line that stably expressed both PD-1 and NF-KB-LUC reporter genes.
- the surface expression level of PD-1 was identified by flow cytometry, and the expression level of the reporter gene was identified by the reaction of the reporter gene after stimulation with OKT-3 and Raiji cells.
- the expression plasmid of human PD-L1 was transfected into Raji cells to obtain a cell line stably expressing PD-L1. Then, Jurkat/NF-KB-luc/PD1 cells were co-cultured with Raji-PD-L1 cells and stimulated with OKT-3. Compounds were added to reflect the compounds to PD-1/PD by reading the reaction of the reporter reaction. Inhibition of the -L1 interaction enhances the T cell activation signaling pathway.
- the specific experimental methods are as follows:
- the series of compounds of the present invention have a strong inhibitory effect on the protein interaction of PD-1/PD-L1, and this inhibition can enhance or restore T cells at the cellular level. activation. 3. Determination of mouse pharmacokinetics 1. Research purposes
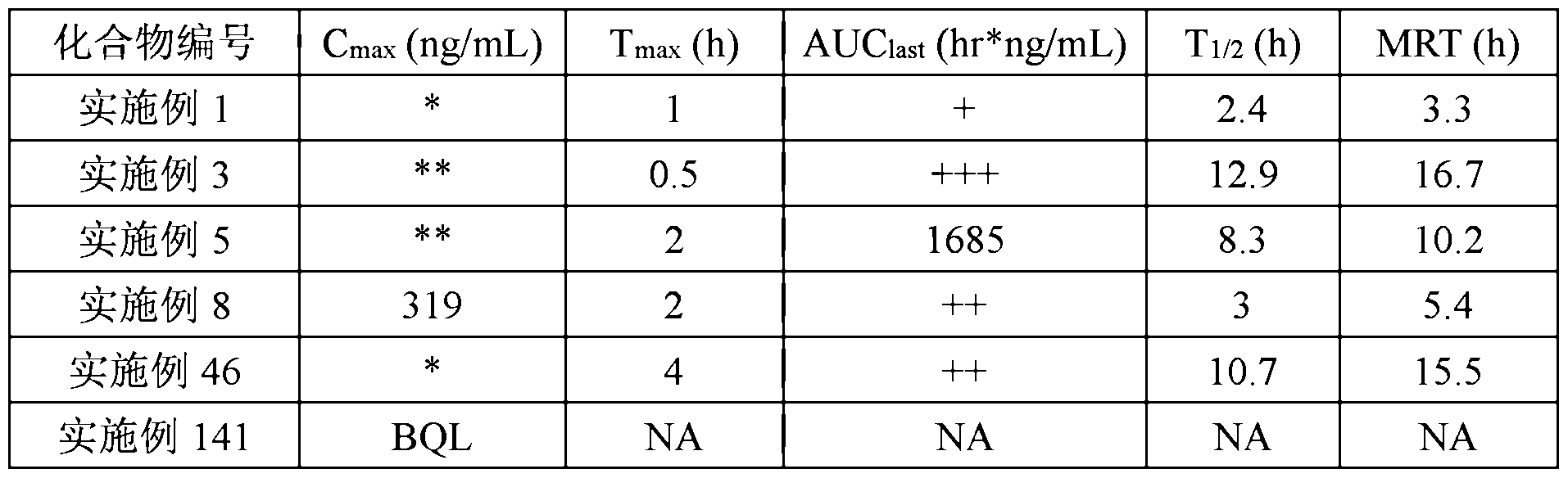
- Example 1 10 mg/kg: Example 1, Example 3, Example 5, Example 8, Example 46, Example 104 and Example 141 compound.
- Example 4 30 mg/kg: Example 4, Example 26 and Example 64 compounds.
- the compounds of this test are derived from the compounds of the specific examples of the invention.
- Blood was taken from the submandibular vein at about 90 ⁇ / time point, anticoagulation of heparin sodium, placed on ice after collection, and centrifuged to separate plasma within 1 hour (centrifugation conditions: 8000 rpm, 6 minutes, 2-8 degrees) ).
- the time of blood collection is 0, 0.25, 0.5, 1, 2, 4, 6, 8, 24 hours.
- the sample is stored in a negative 20 degree refrigerator.
- Plasma sample 40 was added to 160 ice-cold acetonitrile containing an internal standard, vortexed for 3 minutes, and centrifuged at 1 1000 rpm for 5 minutes. Take the supernatant 100 and add it to 100 water, take 5 injections to Analyze. Only the prototype compounds were analyzed for Example 1, Example 3, Example 5, Example 8, Example 46, and Example 141 (see Table 2 for the results); Example 4, Example 26, Example 64, and Example 104 Simultaneous analysis of the protoforms ester and possible ester hydrolysates were Example 2, Example 27, Example 65 and Example 105 (see Table 3 for results).
- Example 4 and Example 104 were orally absorbed into mice and rapidly undergo ester hydrolysis to form the corresponding acid (Example 2 and Example 105) and examples. Compared to Example 1, the metabolism of Example 105 was significantly slower in vivo, and the half-life was increased from 1.6 hours to 20 hours. The methylphenidate compound (Example 26 and Example 64) was orally administered into the body, and almost only the original compound was present. , no ester hydrolyzate was detected.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Pharmacology & Pharmacy (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- General Health & Medical Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Medicinal Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- General Chemical & Material Sciences (AREA)
- Immunology (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Engineering & Computer Science (AREA)
- Epidemiology (AREA)
- Oncology (AREA)
- Communicable Diseases (AREA)
- Diabetes (AREA)
- Hematology (AREA)
- Obesity (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Nitrogen And Oxygen Or Sulfur-Condensed Heterocyclic Ring Systems (AREA)
- Pyridine Compounds (AREA)
- Plural Heterocyclic Compounds (AREA)
Abstract
Description
Claims
Priority Applications (12)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CA3088927A CA3088927C (en) | 2018-02-05 | 2019-01-29 | Biaryl derivative, preparation method therefor and pharmaceutical use thereof |
| EP19748336.5A EP3750887A4 (en) | 2018-02-05 | 2019-01-29 | BIARYLE DERIVATIVE, ITS PREPARATION PROCESS, AND ITS PHARMACEUTICAL APPLICATION |
| CN201980005084.9A CN111448189A (zh) | 2018-02-05 | 2019-01-29 | 一种联芳基衍生物、其制备方法和在药学上的应用 |
| BR112020014459-2A BR112020014459A2 (pt) | 2018-02-05 | 2019-01-29 | Derivado de biarila, método de preparação do mesmo e uso farmacêutico do mesmo |
| US16/967,030 US11459339B2 (en) | 2018-02-05 | 2019-01-29 | Biaryl derivative, preparation method thereof and pharmaceutical application thereof |
| RU2020127950A RU2768830C9 (ru) | 2018-02-05 | 2019-01-29 | Производное биарила, метод его получения и его фармацевтическое применение |
| AU2019214089A AU2019214089B2 (en) | 2018-02-05 | 2019-01-29 | Biaryl derivative, preparation method therefor and pharmaceutical use thereof |
| MX2020008205A MX2020008205A (es) | 2018-02-05 | 2019-01-29 | Derivado de biarilo, metodo de preparacion del mismo y uso farmaceutico del mismo. |
| SG11202006409TA SG11202006409TA (en) | 2018-02-05 | 2019-01-29 | Biaryl derivative, preparation method therefor and pharmaceutical use thereof |
| KR1020207020538A KR102534185B1 (ko) | 2018-02-05 | 2019-01-29 | 비아릴 유도체, 이의 합성법과 그 약학적 용법 |
| JP2020542156A JP7033343B2 (ja) | 2018-02-05 | 2019-01-29 | バイアリール誘導体、その製造方法および薬学上の使用 |
| PH12020551184A PH12020551184A1 (en) | 2018-02-05 | 2020-08-05 | Biaryl derivative, preparation method thereof and pharmaceutical application thereof |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201810111413.5 | 2018-02-05 | ||
| CN201810111413 | 2018-02-05 | ||
| CN201810770644.7 | 2018-07-13 | ||
| CN201810770644 | 2018-07-13 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2019149183A1 true WO2019149183A1 (zh) | 2019-08-08 |
Family
ID=67479570
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/CN2019/073594 WO2019149183A1 (zh) | 2018-02-05 | 2019-01-29 | 一种联芳基衍生物、其制备方法和在药学上的应用 |
Country Status (13)
| Country | Link |
|---|---|
| US (1) | US11459339B2 (zh) |
| EP (1) | EP3750887A4 (zh) |
| JP (1) | JP7033343B2 (zh) |
| KR (1) | KR102534185B1 (zh) |
| CN (1) | CN111448189A (zh) |
| AU (1) | AU2019214089B2 (zh) |
| BR (1) | BR112020014459A2 (zh) |
| CA (1) | CA3088927C (zh) |
| MX (1) | MX2020008205A (zh) |
| PH (1) | PH12020551184A1 (zh) |
| SG (1) | SG11202006409TA (zh) |
| TW (1) | TWI828649B (zh) |
| WO (1) | WO2019149183A1 (zh) |
Cited By (19)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2021008491A1 (zh) * | 2019-07-18 | 2021-01-21 | 上海和誉生物医药科技有限公司 | 一种阻断pd-1/pd-l1相互作用的联苯基衍生物及其制备方法和应用 |
| US11059834B2 (en) | 2017-08-08 | 2021-07-13 | Chemocentryx, Inc. | Macrocyclic immunomodulators |
| WO2021147940A1 (zh) * | 2020-01-21 | 2021-07-29 | 上海华汇拓医药科技有限公司 | 一种pd-1/pd-l1抑制剂及其制备方法和用途 |
| CN113248492A (zh) * | 2020-02-10 | 2021-08-13 | 上海海雁医药科技有限公司 | 杂环取代的含氮六元杂环衍生物及其制法与医药上的用途 |
| JP2021520342A (ja) * | 2018-04-03 | 2021-08-19 | ベータ ファーマシューティカルズ カンパニー リミテッド | 免疫調節物質、組成物及びそれらの方法 |
| US11130740B2 (en) | 2017-04-25 | 2021-09-28 | Arbutus Biopharma Corporation | Substituted 2,3-dihydro-1H-indene analogs and methods using same |
| US11135210B2 (en) | 2018-02-22 | 2021-10-05 | Chemocentryx, Inc. | Indane-amines as PD-L1 antagonists |
| CN113493469A (zh) * | 2020-03-18 | 2021-10-12 | 成都倍特药业股份有限公司 | 可作为免疫调节剂的化合物、其制备方法和应用 |
| WO2022033303A1 (zh) * | 2020-08-11 | 2022-02-17 | 中国人民解放军军事科学院军事医学研究院 | 苄胺类衍生物及其制备方法与用途 |
| US11266643B2 (en) | 2019-05-15 | 2022-03-08 | Chemocentryx, Inc. | Triaryl compounds for treatment of PD-L1 diseases |
| US11426364B2 (en) | 2016-06-27 | 2022-08-30 | Chemocentryx, Inc. | Immunomodulator compounds |
| US11485708B2 (en) | 2019-06-20 | 2022-11-01 | Chemocentryx, Inc. | Compounds for treatment of PD-L1 diseases |
| EP4159720A1 (en) | 2021-09-30 | 2023-04-05 | Recepton Spolka z Ograniczona Odpowiedzialnoscia | Nonsymmetric substituted 1,1'-biphenyl derivatives and uses thereof |
| US11708326B2 (en) | 2017-07-28 | 2023-07-25 | Chemocentryx, Inc. | Immunomodulator compounds |
| US11713307B2 (en) | 2019-10-16 | 2023-08-01 | Chemocentryx, Inc. | Heteroaryl-biphenyl amides for the treatment of PD-L1 diseases |
| WO2023169373A1 (zh) * | 2022-03-07 | 2023-09-14 | 上海和誉生物医药科技有限公司 | 一种全对称联苯基衍生物及其制备方法和应用 |
| CN117343006A (zh) * | 2023-10-08 | 2024-01-05 | 河南科技大学 | 一种arb-272572的制备方法 |
| US11866429B2 (en) | 2019-10-16 | 2024-01-09 | Chemocentryx, Inc. | Heteroaryl-biphenyl amines for the treatment of PD-L1 diseases |
| US11872217B2 (en) | 2019-07-10 | 2024-01-16 | Chemocentryx, Inc. | Indanes as PD-L1 inhibitors |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN105705489A (zh) * | 2013-09-04 | 2016-06-22 | 百时美施贵宝公司 | 用作免疫调节剂的化合物 |
| WO2017106634A1 (en) * | 2015-12-17 | 2017-06-22 | Incyte Corporation | N-phenyl-pyridine-2-carboxamide derivatives and their use as pd-1/pd-l1 protein/protein interaction modulators |
| WO2017222976A1 (en) * | 2016-06-20 | 2017-12-28 | Incyte Corporation | Heterocyclic compounds as immunomodulators |
| WO2018044783A1 (en) * | 2016-08-29 | 2018-03-08 | Incyte Corporation | Heterocyclic compounds as immunomodulators |
| WO2018119221A1 (en) * | 2016-12-22 | 2018-06-28 | Incyte Corporation | Pyridine derivatives as immunomodulators |
| WO2018119224A1 (en) * | 2016-12-22 | 2018-06-28 | Incyte Corporation | Tetrahydro imidazo[4,5-c]pyridine derivatives as pd-l1 internalization inducers |
Family Cites Families (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| AU2017294231B2 (en) * | 2016-07-05 | 2021-09-09 | Guangzhou Maxinovel Pharmaceuticals Co., Ltd | Aromatic acetylene or aromatic ethylene compound, intermediate, preparation method, pharmaceutical composition and use thereof |
| CA3030773A1 (en) * | 2016-08-03 | 2018-02-08 | Arising International, Inc. | Symmetric or semi-symmetric compounds useful as immunomodulators |
| EP3774750A4 (en) | 2018-04-03 | 2021-12-29 | Betta Pharmaceuticals Co., Ltd | Immunomodulators, compositions and methods thereof |
-
2019
- 2019-01-29 WO PCT/CN2019/073594 patent/WO2019149183A1/zh unknown
- 2019-01-29 US US16/967,030 patent/US11459339B2/en active Active
- 2019-01-29 MX MX2020008205A patent/MX2020008205A/es unknown
- 2019-01-29 BR BR112020014459-2A patent/BR112020014459A2/pt active Search and Examination
- 2019-01-29 KR KR1020207020538A patent/KR102534185B1/ko active IP Right Grant
- 2019-01-29 EP EP19748336.5A patent/EP3750887A4/en active Pending
- 2019-01-29 SG SG11202006409TA patent/SG11202006409TA/en unknown
- 2019-01-29 JP JP2020542156A patent/JP7033343B2/ja active Active
- 2019-01-29 AU AU2019214089A patent/AU2019214089B2/en active Active
- 2019-01-29 CA CA3088927A patent/CA3088927C/en active Active
- 2019-01-29 CN CN201980005084.9A patent/CN111448189A/zh active Pending
- 2019-01-30 TW TW108103459A patent/TWI828649B/zh active
-
2020
- 2020-08-05 PH PH12020551184A patent/PH12020551184A1/en unknown
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN105705489A (zh) * | 2013-09-04 | 2016-06-22 | 百时美施贵宝公司 | 用作免疫调节剂的化合物 |
| WO2017106634A1 (en) * | 2015-12-17 | 2017-06-22 | Incyte Corporation | N-phenyl-pyridine-2-carboxamide derivatives and their use as pd-1/pd-l1 protein/protein interaction modulators |
| WO2017222976A1 (en) * | 2016-06-20 | 2017-12-28 | Incyte Corporation | Heterocyclic compounds as immunomodulators |
| WO2018044783A1 (en) * | 2016-08-29 | 2018-03-08 | Incyte Corporation | Heterocyclic compounds as immunomodulators |
| WO2018119221A1 (en) * | 2016-12-22 | 2018-06-28 | Incyte Corporation | Pyridine derivatives as immunomodulators |
| WO2018119224A1 (en) * | 2016-12-22 | 2018-06-28 | Incyte Corporation | Tetrahydro imidazo[4,5-c]pyridine derivatives as pd-l1 internalization inducers |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP3750887A4 * |
Cited By (25)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US11426364B2 (en) | 2016-06-27 | 2022-08-30 | Chemocentryx, Inc. | Immunomodulator compounds |
| US11793771B2 (en) | 2016-06-27 | 2023-10-24 | Chemocentryx, Inc. | Immunomodulator compounds |
| US11130740B2 (en) | 2017-04-25 | 2021-09-28 | Arbutus Biopharma Corporation | Substituted 2,3-dihydro-1H-indene analogs and methods using same |
| US11708326B2 (en) | 2017-07-28 | 2023-07-25 | Chemocentryx, Inc. | Immunomodulator compounds |
| US11691985B2 (en) | 2017-08-08 | 2023-07-04 | Chemocentryx, Inc. | Macrocyclic immunomodulators |
| US11059834B2 (en) | 2017-08-08 | 2021-07-13 | Chemocentryx, Inc. | Macrocyclic immunomodulators |
| US11135210B2 (en) | 2018-02-22 | 2021-10-05 | Chemocentryx, Inc. | Indane-amines as PD-L1 antagonists |
| US11759458B2 (en) | 2018-02-22 | 2023-09-19 | Chemocentryx, Inc. | Indane-amines as PD-L1 antagonists |
| EP3774750A4 (en) * | 2018-04-03 | 2021-12-29 | Betta Pharmaceuticals Co., Ltd | Immunomodulators, compositions and methods thereof |
| JP2021520342A (ja) * | 2018-04-03 | 2021-08-19 | ベータ ファーマシューティカルズ カンパニー リミテッド | 免疫調節物質、組成物及びそれらの方法 |
| US11266643B2 (en) | 2019-05-15 | 2022-03-08 | Chemocentryx, Inc. | Triaryl compounds for treatment of PD-L1 diseases |
| US11485708B2 (en) | 2019-06-20 | 2022-11-01 | Chemocentryx, Inc. | Compounds for treatment of PD-L1 diseases |
| US11872217B2 (en) | 2019-07-10 | 2024-01-16 | Chemocentryx, Inc. | Indanes as PD-L1 inhibitors |
| CN113825751A (zh) * | 2019-07-18 | 2021-12-21 | 上海和誉生物医药科技有限公司 | 一种阻断pd-1/pd-l1相互作用的联苯基衍生物及其制备方法和应用 |
| WO2021008491A1 (zh) * | 2019-07-18 | 2021-01-21 | 上海和誉生物医药科技有限公司 | 一种阻断pd-1/pd-l1相互作用的联苯基衍生物及其制备方法和应用 |
| US11713307B2 (en) | 2019-10-16 | 2023-08-01 | Chemocentryx, Inc. | Heteroaryl-biphenyl amides for the treatment of PD-L1 diseases |
| US11866429B2 (en) | 2019-10-16 | 2024-01-09 | Chemocentryx, Inc. | Heteroaryl-biphenyl amines for the treatment of PD-L1 diseases |
| CN114650993A (zh) * | 2020-01-21 | 2022-06-21 | 上海华汇拓医药科技有限公司 | 一种pd-1/pd-l1抑制剂及其制备方法和用途 |
| WO2021147940A1 (zh) * | 2020-01-21 | 2021-07-29 | 上海华汇拓医药科技有限公司 | 一种pd-1/pd-l1抑制剂及其制备方法和用途 |
| CN113248492A (zh) * | 2020-02-10 | 2021-08-13 | 上海海雁医药科技有限公司 | 杂环取代的含氮六元杂环衍生物及其制法与医药上的用途 |
| CN113493469A (zh) * | 2020-03-18 | 2021-10-12 | 成都倍特药业股份有限公司 | 可作为免疫调节剂的化合物、其制备方法和应用 |
| WO2022033303A1 (zh) * | 2020-08-11 | 2022-02-17 | 中国人民解放军军事科学院军事医学研究院 | 苄胺类衍生物及其制备方法与用途 |
| EP4159720A1 (en) | 2021-09-30 | 2023-04-05 | Recepton Spolka z Ograniczona Odpowiedzialnoscia | Nonsymmetric substituted 1,1'-biphenyl derivatives and uses thereof |
| WO2023169373A1 (zh) * | 2022-03-07 | 2023-09-14 | 上海和誉生物医药科技有限公司 | 一种全对称联苯基衍生物及其制备方法和应用 |
| CN117343006A (zh) * | 2023-10-08 | 2024-01-05 | 河南科技大学 | 一种arb-272572的制备方法 |
Also Published As
| Publication number | Publication date |
|---|---|
| JP7033343B2 (ja) | 2022-03-10 |
| AU2019214089B2 (en) | 2021-02-11 |
| TW201934544A (zh) | 2019-09-01 |
| SG11202006409TA (en) | 2020-08-28 |
| US20210032270A1 (en) | 2021-02-04 |
| RU2020127950A3 (zh) | 2022-03-11 |
| TWI828649B (zh) | 2024-01-11 |
| JP2021512878A (ja) | 2021-05-20 |
| MX2020008205A (es) | 2020-09-18 |
| RU2768830C2 (ru) | 2022-03-24 |
| AU2019214089A1 (en) | 2020-07-09 |
| US11459339B2 (en) | 2022-10-04 |
| CN111448189A (zh) | 2020-07-24 |
| KR102534185B1 (ko) | 2023-05-18 |
| EP3750887A1 (en) | 2020-12-16 |
| BR112020014459A2 (pt) | 2020-12-15 |
| CA3088927C (en) | 2023-03-21 |
| KR20200100717A (ko) | 2020-08-26 |
| CA3088927A1 (en) | 2019-08-08 |
| RU2020127950A (ru) | 2022-03-11 |
| PH12020551184A1 (en) | 2021-05-10 |
| EP3750887A4 (en) | 2021-10-20 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2019149183A1 (zh) | 一种联芳基衍生物、其制备方法和在药学上的应用 | |
| JP6399660B2 (ja) | 癌治療用組成物および方法 | |
| WO2020011209A1 (zh) | 免疫抑制剂及其制备方法和在药学上的应用 | |
| CA3119526A1 (en) | Small molecule degraders of helios and methods of use | |
| JP2023530733A (ja) | 免疫抑制剤、その製造方法及び応用 | |
| US20200347061A1 (en) | Class of amino-substituted nitrogen-containing fused ring compounds, preparation method therefor, and use thereof | |
| WO2018214866A1 (zh) | 一种氮杂芳基衍生物、其制备方法和在药学上的应用 | |
| WO2021008491A1 (zh) | 一种阻断pd-1/pd-l1相互作用的联苯基衍生物及其制备方法和应用 | |
| TWI755418B (zh) | 聯芳組成物和調控激酶級聯之方法 | |
| US20100069443A1 (en) | Compound with benzamide skeleton having cyclooxygenase-1 (cox-1)-selective inhibitory activity | |
| CN114502556B (zh) | 一种联苯基氟代双键衍生物及其制备方法和在药学上的应用 | |
| WO2018090979A1 (zh) | 一种具有抗癌作用的化合物及其制备方法和应用 | |
| RU2768830C9 (ru) | Производное биарила, метод его получения и его фармацевтическое применение | |
| WO2023169373A1 (zh) | 一种全对称联苯基衍生物及其制备方法和应用 | |
| WO2017144010A1 (zh) | (6-(1H-吲唑-6-基)-N-[4-(4-吗啉基)苯基]咪唑并[1,2-a]吡嗪-8-胺)甲磺酸盐的新晶型 | |
| CN112724134A (zh) | 氮杂吲唑联吡啶衍生物髓细胞增殖抑制剂及其制备方法与在制药中的应用 | |
| TW201113274A (en) | Novel ethanediamine hepcidine antagonists |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 19748336 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2019214089 Country of ref document: AU Date of ref document: 20190129 Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 3088927 Country of ref document: CA |
|
| ENP | Entry into the national phase |
Ref document number: 20207020538 Country of ref document: KR Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 2020542156 Country of ref document: JP Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2019748336 Country of ref document: EP Effective date: 20200907 |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01A Ref document number: 112020014459 Country of ref document: BR |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01E Ref document number: 112020014459 Country of ref document: BR Free format text: APRESENTE NOVAS FOLHAS DO RELATORIO DESCRITIVO, QUADRO REIVINDICATORIO E RESUMO TENDO EM VISTA QUE OS CONJUNTOS APRESENTADOS NA PETICAO NO 870200088160 ENCONTRAM-SE EM DESACORDO COM A FORMA PREVISTA DOS ARTS. 15 A 41 DA INSTRUCAO NORMATIVA NO 31/2013 . |
|
| ENP | Entry into the national phase |
Ref document number: 112020014459 Country of ref document: BR Kind code of ref document: A2 Effective date: 20200715 |