WO2019054448A1 - 転動疲労特性に優れた鋼材 - Google Patents
転動疲労特性に優れた鋼材 Download PDFInfo
- Publication number
- WO2019054448A1 WO2019054448A1 PCT/JP2018/034008 JP2018034008W WO2019054448A1 WO 2019054448 A1 WO2019054448 A1 WO 2019054448A1 JP 2018034008 W JP2018034008 W JP 2018034008W WO 2019054448 A1 WO2019054448 A1 WO 2019054448A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- fatigue
- mass
- content
- less
- rem
- Prior art date
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/18—Ferrous alloys, e.g. steel alloys containing chromium
- C22C38/40—Ferrous alloys, e.g. steel alloys containing chromium with nickel
- C22C38/44—Ferrous alloys, e.g. steel alloys containing chromium with nickel with molybdenum or tungsten
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/001—Ferrous alloys, e.g. steel alloys containing N
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/002—Ferrous alloys, e.g. steel alloys containing In, Mg, or other elements not provided for in one single group C22C38/001 - C22C38/60
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/005—Ferrous alloys, e.g. steel alloys containing rare earths, i.e. Sc, Y, Lanthanides
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/02—Ferrous alloys, e.g. steel alloys containing silicon
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/04—Ferrous alloys, e.g. steel alloys containing manganese
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/06—Ferrous alloys, e.g. steel alloys containing aluminium
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/08—Ferrous alloys, e.g. steel alloys containing nickel
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/18—Ferrous alloys, e.g. steel alloys containing chromium
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/18—Ferrous alloys, e.g. steel alloys containing chromium
- C22C38/20—Ferrous alloys, e.g. steel alloys containing chromium with copper
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/18—Ferrous alloys, e.g. steel alloys containing chromium
- C22C38/22—Ferrous alloys, e.g. steel alloys containing chromium with molybdenum or tungsten
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/18—Ferrous alloys, e.g. steel alloys containing chromium
- C22C38/24—Ferrous alloys, e.g. steel alloys containing chromium with vanadium
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/18—Ferrous alloys, e.g. steel alloys containing chromium
- C22C38/26—Ferrous alloys, e.g. steel alloys containing chromium with niobium or tantalum
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/18—Ferrous alloys, e.g. steel alloys containing chromium
- C22C38/28—Ferrous alloys, e.g. steel alloys containing chromium with titanium or zirconium
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/18—Ferrous alloys, e.g. steel alloys containing chromium
- C22C38/32—Ferrous alloys, e.g. steel alloys containing chromium with boron
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/18—Ferrous alloys, e.g. steel alloys containing chromium
- C22C38/40—Ferrous alloys, e.g. steel alloys containing chromium with nickel
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/18—Ferrous alloys, e.g. steel alloys containing chromium
- C22C38/40—Ferrous alloys, e.g. steel alloys containing chromium with nickel
- C22C38/42—Ferrous alloys, e.g. steel alloys containing chromium with nickel with copper
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/18—Ferrous alloys, e.g. steel alloys containing chromium
- C22C38/40—Ferrous alloys, e.g. steel alloys containing chromium with nickel
- C22C38/50—Ferrous alloys, e.g. steel alloys containing chromium with nickel with titanium or zirconium
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/60—Ferrous alloys, e.g. steel alloys containing lead, selenium, tellurium, or antimony, or more than 0.04% by weight of sulfur
-
- C—CHEMISTRY; METALLURGY
- C21—METALLURGY OF IRON
- C21C—PROCESSING OF PIG-IRON, e.g. REFINING, MANUFACTURE OF WROUGHT-IRON OR STEEL; TREATMENT IN MOLTEN STATE OF FERROUS ALLOYS
- C21C7/00—Treating molten ferrous alloys, e.g. steel, not covered by groups C21C1/00 - C21C5/00
- C21C7/04—Removing impurities by adding a treating agent
- C21C7/06—Deoxidising, e.g. killing
-
- C—CHEMISTRY; METALLURGY
- C21—METALLURGY OF IRON
- C21D—MODIFYING THE PHYSICAL STRUCTURE OF FERROUS METALS; GENERAL DEVICES FOR HEAT TREATMENT OF FERROUS OR NON-FERROUS METALS OR ALLOYS; MAKING METAL MALLEABLE, e.g. BY DECARBURISATION OR TEMPERING
- C21D1/00—General methods or devices for heat treatment, e.g. annealing, hardening, quenching or tempering
- C21D1/34—Methods of heating
- C21D1/42—Induction heating
-
- C—CHEMISTRY; METALLURGY
- C21—METALLURGY OF IRON
- C21D—MODIFYING THE PHYSICAL STRUCTURE OF FERROUS METALS; GENERAL DEVICES FOR HEAT TREATMENT OF FERROUS OR NON-FERROUS METALS OR ALLOYS; MAKING METAL MALLEABLE, e.g. BY DECARBURISATION OR TEMPERING
- C21D6/00—Heat treatment of ferrous alloys
- C21D6/002—Heat treatment of ferrous alloys containing Cr
-
- C—CHEMISTRY; METALLURGY
- C21—METALLURGY OF IRON
- C21D—MODIFYING THE PHYSICAL STRUCTURE OF FERROUS METALS; GENERAL DEVICES FOR HEAT TREATMENT OF FERROUS OR NON-FERROUS METALS OR ALLOYS; MAKING METAL MALLEABLE, e.g. BY DECARBURISATION OR TEMPERING
- C21D6/00—Heat treatment of ferrous alloys
- C21D6/005—Heat treatment of ferrous alloys containing Mn
-
- C—CHEMISTRY; METALLURGY
- C21—METALLURGY OF IRON
- C21D—MODIFYING THE PHYSICAL STRUCTURE OF FERROUS METALS; GENERAL DEVICES FOR HEAT TREATMENT OF FERROUS OR NON-FERROUS METALS OR ALLOYS; MAKING METAL MALLEABLE, e.g. BY DECARBURISATION OR TEMPERING
- C21D6/00—Heat treatment of ferrous alloys
- C21D6/008—Heat treatment of ferrous alloys containing Si
-
- C—CHEMISTRY; METALLURGY
- C21—METALLURGY OF IRON
- C21D—MODIFYING THE PHYSICAL STRUCTURE OF FERROUS METALS; GENERAL DEVICES FOR HEAT TREATMENT OF FERROUS OR NON-FERROUS METALS OR ALLOYS; MAKING METAL MALLEABLE, e.g. BY DECARBURISATION OR TEMPERING
- C21D9/00—Heat treatment, e.g. annealing, hardening, quenching or tempering, adapted for particular articles; Furnaces therefor
- C21D9/40—Heat treatment, e.g. annealing, hardening, quenching or tempering, adapted for particular articles; Furnaces therefor for rings; for bearing races
Definitions
- the present invention relates to a steel material excellent in rolling fatigue characteristics, in which the composition of nonmetallic inclusions is controlled.
- the present invention relates to a steel material having good rolling fatigue characteristics in which fatigue damage originating from inclusions is suppressed by using cluster-like oxide-based inclusions as REM-containing inclusions.
- the rolling bearing includes, for example, a “rolling element” in the shape of a ball or a roller, and an “inner ring” and an “outer ring” that transmit a load in contact with the rolling element.
- Steel materials used for rolling members such as rolling elements, inner rings, and outer rings are required to have excellent rolling fatigue characteristics. The inclusions contained in the steel material are desired to be as fine and as small as possible for the purpose of improving the rolling fatigue life.
- oxides such as alumina (Al 2 O 3 ), sulfides such as manganese sulfide (MnS), and nitrides such as titanium nitride (TiN) are known.
- Alumina-based inclusions are formed by combining dissolved oxygen remaining in molten steel refined in a converter or a vacuum processing vessel with Al having a high affinity for oxygen. Also, ladles and the like are often made of alumina-based refractories. Therefore, at the time of deoxidation, by the reaction of the molten steel and the refractory, alumina is eluted as Al in the molten steel and reoxidized to form alumina-based inclusions. Alumina-based inclusions form clusters in the solidified steel and cause a reduction in rolling fatigue life.
- Patent Document 1 discloses a method of forming low-melting inclusions by adding two or more of REM, Mg, and Ca to molten steel. This method is effective in preventing slivers. However, this method can not reduce the size of inclusions to the level required for bearing steels. The reason is that inclusions having a low melting point are easily aggregated and coalesced.
- Patent Document 2 discloses that the content of REM needs to be 0.010% by mass or less in order not to reduce the fatigue life. However, Patent Document 2 does not disclose the mechanism of fatigue life reduction and the existence state of inclusions.
- An object of the present invention is to provide a steel material excellent in rolling fatigue characteristics in view of the problems of the prior art.
- the gist of the present invention is as follows. [1] In mass%, C: 0.10% to 1.50%, Si: 0.01% to 0.80%, Mn: 0.10% to 1.50%, Cr: 0.02% to 2.50%, Al: 0.002% to less than 0.010%, Ce + La + Nd: 0.0001% to 0.0025%, Mg: 0.0005% to 0.0050%, O: 0.0001% to 0.0020%, Ti: 0.000% to less than 0.005%, N: 0.0180% or less, P: 0.030% or less, S: 0.005% or less, Ca: 0.0000% to 0.0010%, V: 0.00 to 0.40%, Mo: 0.00 to 0.60%, Cu: 0.00 to 0.50%, Nb: 0.000 to less than 0.050%, Ni: 0.00 to 2.50%, Pb: 0.00 to 0.10%, Bi: 0.00 to 0.10%, B: 0.0000 to 0.0050%, The balance is Fe and impurities, It is characterized in that the fatigue origin inclusion detected by the ultra
- Ce%, La%, Nd%, Mg% and Al% are atomic weight percentages of Ce, La, Nd, Mg and Al contained in the fatigue origin inclusion, respectively.
- the cluster-like Al-O-based inclusions are reformed into REM-Al-Mg-O-based inclusions to reduce the influence of the oxide-based inclusions on the fatigue characteristics, It is possible to provide a steel material excellent in dynamic fatigue characteristics.
- the present inventors diligently conducted experiments and studies in order to solve the problems of the prior art. As a result, the following findings were obtained by adjusting the content of REM and the content of Al and S.
- (1) The adhesion between the oxide-based inclusions and the base material is improved by modifying the Al-O-based inclusions, which are cluster-like oxides, into REM-Al-Mg-O-based inclusions.
- Al-O-based inclusions which are cluster-like oxides, are coarse and adversely affect fatigue characteristics, so it is better to minimize the amount of Al, but if not added, coarse lower oxides are formed, The effect of reforming oxide inclusions by Mg and REM can not be obtained. Therefore, deoxidation of Al is required at a minimum.
- C 0.10% to 1.50%
- C is an element which secures hardness and improves the fatigue life. In order to secure required strength and hardness, it is necessary to contain C 0.10% or more. However, if the C content exceeds 1.50%, the hardness increases too much, which causes a quench crack. Therefore, the C content is 0.10% to 1.50%. If C: 0.10% to less than 0.45%, it is suitable for steel materials for skin burning. If C: 0.45% to less than 0.90%, it is suitable for steel materials for induction hardening. If C: 0.90% to 1.50%, it is suitable for steel materials for overall hardening.
- the lower limit of the C content is preferably 0.15%.
- the upper limit of the C content is preferably 1.35%.
- Si 0.01% to 0.80% Si is an element that improves the hardenability and improves the fatigue life. In order to acquire this effect, it is necessary to contain Si 0.01% or more. However, if the Si content exceeds 0.80%, the hardenability improving effect is saturated, and further, the deoxidation state is affected, the formation of oxides is affected, and the fatigue characteristics are deteriorated. Therefore, the Si content is set to 0.01% to 0.80%.
- the lower limit of the Si content is preferably 0.07%.
- the upper limit of the Si content is preferably 0.65% or less.
- Mn 0.10% to 1.50%
- Mn is an element that enhances hardenability and strength, and improves fatigue life. In order to acquire this effect, it is necessary to contain Mn 0.10% or more. However, if the Mn content exceeds 1.50%, the effect of improving the hardenability is saturated, which in turn causes a quench crack. Therefore, the Mn content is 0.10% to 1.50%.
- the lower limit of the Mn content is preferably 0.20%.
- the upper limit of the Mn content is preferably 1.20%.
- Cr 0.02% to 2.50% Cr is an element that improves the hardenability and improves the fatigue life. In order to acquire this effect stably, it is preferable to contain Cr 0.02% or more. However, if the Cr content exceeds 2.50%, the effect of improving the hardenability is saturated, which in turn causes a quench crack. Therefore, the upper limit of the Cr content is 2.50%.
- the lower limit of the Cr content is preferably 0.15% or more.
- the upper limit of the Cr content is preferably 2.00% or less.
- the Cr content may be defined as 1.90% or less or 1.80% or less. When used as a steel material for bearings due to skin burning, it is desirable that the Cr content be 0.02 to 1.50%. In the case of using as a steel material for bearings by induction hardening or as a steel material for bearings by whole hardening, it is desirable to set Cr: 0.70 to 2.50%.
- Al 0.002% to less than 0.010%
- Al is T.O. It is necessary to contain 0.002% or more as a deoxidizing element which reduces O (total oxygen amount).
- O total oxygen amount
- the Al content is less than 0.010%.
- the Al content is preferably 0.005% or more with respect to the lower limit.
- the Al content is preferably 0.008% or less with respect to the upper limit.
- Ce + La + Nd 0.0001% to 0.0025% Ce (cerium), La (lanthanum), and Nd (neodymium) are elements classified into the rare earth elements.
- the rare earth element is a generic name of a total of 17 elements in which scandium of atomic number 21 and yttrium of atomic number 39 are added to 15 elements from lanthanum of atomic number 57 to lutetium of 71.
- the rare earth element is a strong deoxidizing element, and plays an extremely important role in the steel for bearing according to the present embodiment.
- the rare earth element alloy for steelmaking is mainly composed of three elements of Ce, La and Nd. Therefore, in the present invention, Ce, La and Nd are limited among the rare earth 17 elements.
- Elements other than the three elements contained in the rare earth alloy are also powerful deoxidizing elements, and exert the same effects as the three elements. It may contain 0.0001% to 0.0025% of any one of Ce, La and Nd, or may contain 0.0001% to 0.0025% of two or more in total.
- Ce, La and Nd are collectively referred to as REM.
- REM reacts with oxygen in molten steel to form REM-based oxides.
- REM-based oxides are also simultaneously taken up with the aggregation and clustering of the alumina oxides in the molten steel. By doing so, the Al-O-based oxide is reformed into REM-Al-Mg-O-based inclusions.
- the function of REM in the bearing steel material according to the present embodiment is as follows.
- the size of the cluster-like inclusions is not significantly different between the case where REM is not added and the case where it is not added.
- the presence of REM-based oxides improves the interface state with the base material, specifically the adhesion, so even if they have the same size, they do not easily become fracture starting points, and fatigue characteristics improves.
- the content effect is insufficient when REM is less than 0.0001%. Accordingly, the lower limit of the REM content is made 0.0001%, preferably 0.0003% or more, more preferably 0.0008% or more. However, if the REM content exceeds 0.0025%, not only the cost increases but also blockage of the casting nozzle tends to occur, which hinders the production of steel. Therefore, the upper limit of the content of REM is 0.0025%, preferably 0.0020%, and more preferably 0.0018%.
- Mg 0.0005% to 0.0050% Mg, like Al, is a strong deoxidizing element and plays an extremely important role in the steel material according to the present embodiment.
- the effect of suppressing the destruction of the cluster oxide starting point is small with Mg alone, the combined use of REM and Mg enhances the effect of improving the fatigue characteristics more than REM alone.
- the Mg content is made 0.0050% or less.
- the Mg content preferably defines the lower limit to 0.0010% or more.
- the Mg content preferably defines the upper limit to be 0.0040% or less.
- O 0.0001% to 0.0020%
- O is an impurity and an element removed from the steel by deoxidation. If it is possible to eliminate O in the steel by deoxidation, alumina clusters are not generated, and the problem to be solved by the present invention does not naturally occur. However, from the viewpoint of technology and cost, 0.0001% or more of O is inevitably contained in the present steel, and the resulting alumina clusters may lower the fatigue properties.
- the present invention improves the fatigue characteristics of a steel containing the same level of oxygen as normally contained, as compared to the prior art. Generally, the O content of steel is often 0.0005% or more. On the other hand, when the O content exceeds 0.0020%, a large amount of oxide such as alumina remains and the fatigue life decreases, so the upper limit of the O content is made 0.0020%.
- the O content is preferably 0.0015% or less.
- Composition ratio of Ce, La, Nd, Mg and Al in fatigue origin inclusions (Ce% + La% + Nd% + Mg%) / Al%: 0.20 or more Al-O based inclusions which are oxides in a cluster form are REM By reforming into —Al—Mg—O inclusions, the adhesion between the oxide inclusions and the base material is improved, and the fatigue characteristics are improved. This effect appears when the composition ratio of Ce, La, Nd, Mg, and Al (Ce% + La% + Nd% + Mg%) / Al% in the fatigue starting inclusions is 0.20% or more. Therefore, (Ce% + La% + Nd% + Mg%) / Al% is set to 0.20 or more. In order to further enhance the above effect, (Ce% + La% + Nd% + Mg%) / Al% is preferably 0.50 or more.
- the Ce%, La%, Nd%, Mg% and Al% in the fatigue origin inclusions are respectively the number of atoms of each element relative to the atomic number sum of Ce, La, Nd, Mg and Al contained in the fatigue origin inclusions. Ratio (atomic weight%). When all three items of Al%, Mg%, and (Ce% + La% + Nd%) are 0.1 or more in the fatigue origin inclusion, the fatigue origin inclusion is “REM-Al-Mg-O It is assumed that it is a system inclusion. Desirably, it is desirable that the fatigue origin inclusions contain both Mg% and (Ce% + La% + Nd%) of 1.0 or more. Although the number of O atoms and the ratio of O are not considered in the calculation of the atomic weight%, the above elements form composite inclusions through O, and O is included in the fatigue origin inclusions. Be
- the balance is iron and impurities.
- impurity in “the balance is iron and impurities” refers to what is unavoidably mixed from the ore as a raw material, scrap, or a manufacturing environment etc., when manufacturing steel industrially.
- the impurities Ti, N, P, S, and Ca need to be limited as follows.
- Ti 0.000% to less than 0.005%
- Ti is an impurity, and if it is present in the steel, it forms TiN and degrades the fatigue properties, so the Ti content is limited to less than 0.005%.
- the Ti content is limited to 0.004% or less.
- N 0.0180% or less
- N is an impurity, and when it is present in steel, it forms nitrides to deteriorate fatigue properties, and also to deteriorate ductility and toughness by strain aging.
- the upper limit of the N content is limited to 0.0180%.
- the N content is limited to 0.0150% or less.
- 0.0000% of N may be sufficient, reduction of N content has industrial restrictions and it is meaningless to make it too low. It may be 0.0020% as a practical lower limit of N possible at ordinary cost.
- P 0.030% or less
- P is an impurity, and when it is present in steel, it segregates at grain boundaries to reduce the fatigue life.
- the upper limit of the P content is limited to 0.030%.
- the P content is limited to 0.020% or less.
- the lower limit of the P content may be 0.000%, but may be 0.001% as an industrial lower limit.
- S when present in steel, forms a sulfide.
- S content exceeds 0.005%, S combines with REM to form a sulfide, reducing REM effective for reforming alumina clusters, and thus reducing the fatigue life. Therefore, the upper limit of the S content is limited to 0.005%.
- the S content is limited to 0.0025% or less.
- the lower limit of the S content may be 0.000%, but may be 0.001% as the industrial lower limit.
- Ca 0.0000% to 0.0010%
- coarse CaO is formed to reduce the fatigue life, so the upper limit is made 0.0010%.
- the Ca content is preferably 0.0002% or less, and more preferably 0.0000%.
- the steel material according to the present embodiment further includes V: 0.00% to 0.40%, Mo: 0.00% to 0.60%, Cu: 0.00% to 0.50%, Nb: 0. 000% to 0.050%, Ni: 0.00% to 2.50%, Pb: 0.00 to 0.10%, Bi: 0.00 to 0.10%, B: 0.0000 to 0..
- V 0.00% to 0.40%
- Mo 0.00% to 0.60%
- Cu 0.00% to 0.50%
- Nb 0. 000% to 0.050%
- Ni 0.00% to 2.50%
- Pb 0.00 to 0.10%
- Bi 0.00 to 0.10%
- B 0.0000 to 0..
- One or more of 0050% may be contained.
- V 0.00% to 0.40%
- V is an element which combines with C and N in steel to form carbides, nitrides or carbonitrides and contributes to the refinement and strengthening of the steel structure.
- V 0.05% or more.
- the V content is more preferably 0.10% or more.
- the V content is set to 0.40%.
- the V content is 0.30% or less.
- Mo 0.00% to 0.60%
- Mo is an element that enhances hardenability and combines with C in the steel to form carbides and contributes to the improvement of the strength of the steel by precipitation strengthening. In order to acquire this effect stably, it is preferable to contain Mo 0.05% or more.
- the Mo content is more preferably 0.10% or more. However, if the Mo content exceeds 0.60%, it will cause a cause of cracking, so the upper limit of the Mo content is made 0.60%.
- the Mo content is preferably 0.50% or less.
- Cu 0.00% to 0.50%
- Cu is an element that contributes to the improvement of fatigue characteristics by strengthening the base material. In order to acquire this effect stably, it is preferable to contain Cu 0.05% or more. However, if the Cu content exceeds 0.50%, cracking occurs during hot working, so the upper limit of the Cu content is made 0.50%.
- the Cu content is preferably 0.35% or less.
- Nb 0.000% to less than 0.050%
- Nb is an element that contributes to the improvement of fatigue characteristics by strengthening the base material. In order to acquire this effect stably, it is preferable to contain Nb 0.005% or more.
- the Nb content is more preferably 0.010% or more. However, if the Nb content is 0.050% or more, the contained effect is saturated and cracks are generated during hot working, so the Nb content is made less than 0.050%.
- the Nb content is preferably 0.030% or less.
- Ni 0.00% to 2.50% or Less Ni is an element that contributes to the improvement of the fatigue life by enhancing the corrosion resistance. In order to acquire this effect stably, it is preferable to contain Ni 0.10% or more.
- the Ni content is more preferably 0.30% or more. However, if the Ni content exceeds 2.50%, the machinability of the steel decreases, so the upper limit of the Ni content is set to 2.50%.
- the Ni content is preferably 2.00% or less.
- Pb 0.00% to 0.10% Pb is added to enhance the machinability of the steel. However, if the Pb content exceeds 0.10%, it is the origin of fatigue cracks and the fatigue strength is reduced. Therefore, the upper limit of the Pb content is 0.10%.
- the Pb content is preferably 0.06% or less.
- Bi 0.00% to 0.10% Bi is added to increase the machinability of the steel. However, if the Bi content exceeds 0.10%, it is a starting point of fatigue cracking and the fatigue strength is reduced. Therefore, the upper limit of the Bi content is 0.10%.
- the Bi content is preferably 0.06% or less.
- B 0.0000% to 0.0050% B is segregated to austenite grain boundaries, and has the effect of enhancing grain boundary strength and improving toughness. However, if the B content exceeds 0.0050%, the austenite grains grow abnormally during heat treatment, and the fatigue strength decreases. Therefore, the upper limit of the B content is set to 0.0050%.
- the B content is preferably 0.0030% or less.
- the above cluster-like oxide is stretched by rolling.
- the interface state with the base material is reformed and the fatigue characteristics are improved by becoming a composite of the alumina alone to the REM oxide regardless of the form or size thereof.
- the order of introducing the deoxidizer is important.
- deoxidation is performed using Al and Mg.
- ladle refining including vacuum degassing is performed.
- REM When REM is added at the initial stage of deoxidation, the REM-O-based oxide is formed and fixed, and the alumina formed later or the Al—Mg—O-based oxide can not be reformed. Therefore, at the beginning of deoxidation, Al and then Mg are added to fix O contained in the molten steel as an oxide. Thereafter, REM is added to reform the cluster-like oxide into REM-Al-Mg-O-based inclusions.
- misch metal an alloy composed of a plurality of rare earth metals
- massive misch metal may be added to molten steel at the end of refining.
- Deoxidation by REM is performed for 60 seconds or more. This is the time required for the added REM to take in oxygen from the Al—Mg—O-based oxide once formed to form the REM-based oxide.
- the Al-O-based cluster oxide can be reformed into REM-Al-Mg-O-based inclusions, and the rolling characteristics of the steel material are improved.
- the generation amount of MnS and the generation amount of TiN existing independently are extremely small, but it is not necessary to completely eliminate it.
- MnS and TiN will not be coarsened from the cluster-like oxide, and they can not be a starting point of fatigue failure.
- the cast slab after heating is heated to a heating temperature and then held for 60 seconds to 60 minutes in a temperature range of 1200 ° C. to 1250 ° C., and then subjected to hot rolling or hot forging.
- Manufacture steel products By using this steel material as a raw material and cutting it into a shape close to the final shape, heat treatment such as carburizing, induction hardening, whole hardening and the like can be performed to make the surface hardness suitable for the bearing.
- the steel materials which concern on this embodiment are C: 0.10%-1.50%, if C: 0.10%-less than 0.45%, it is suitable for steel materials for skin-burning.
- the surface hardness can be set to a Vickers hardness of 700 Hv (measurement load 2.94 N) or more. If C: 0.45% to 1.50%, the hardness of the surface can be set to a Vickers hardness of 650 Hv (measurement load 2.94 N) or more by induction hardening. In addition, C: 0.90% to 1.50% is suitable for steel materials for bearings after overall quenching.
- the rolling member heat-treated by carburizing, induction hardening, whole hardening etc. using the steel materials of the present invention is excellent in fatigue characteristics.
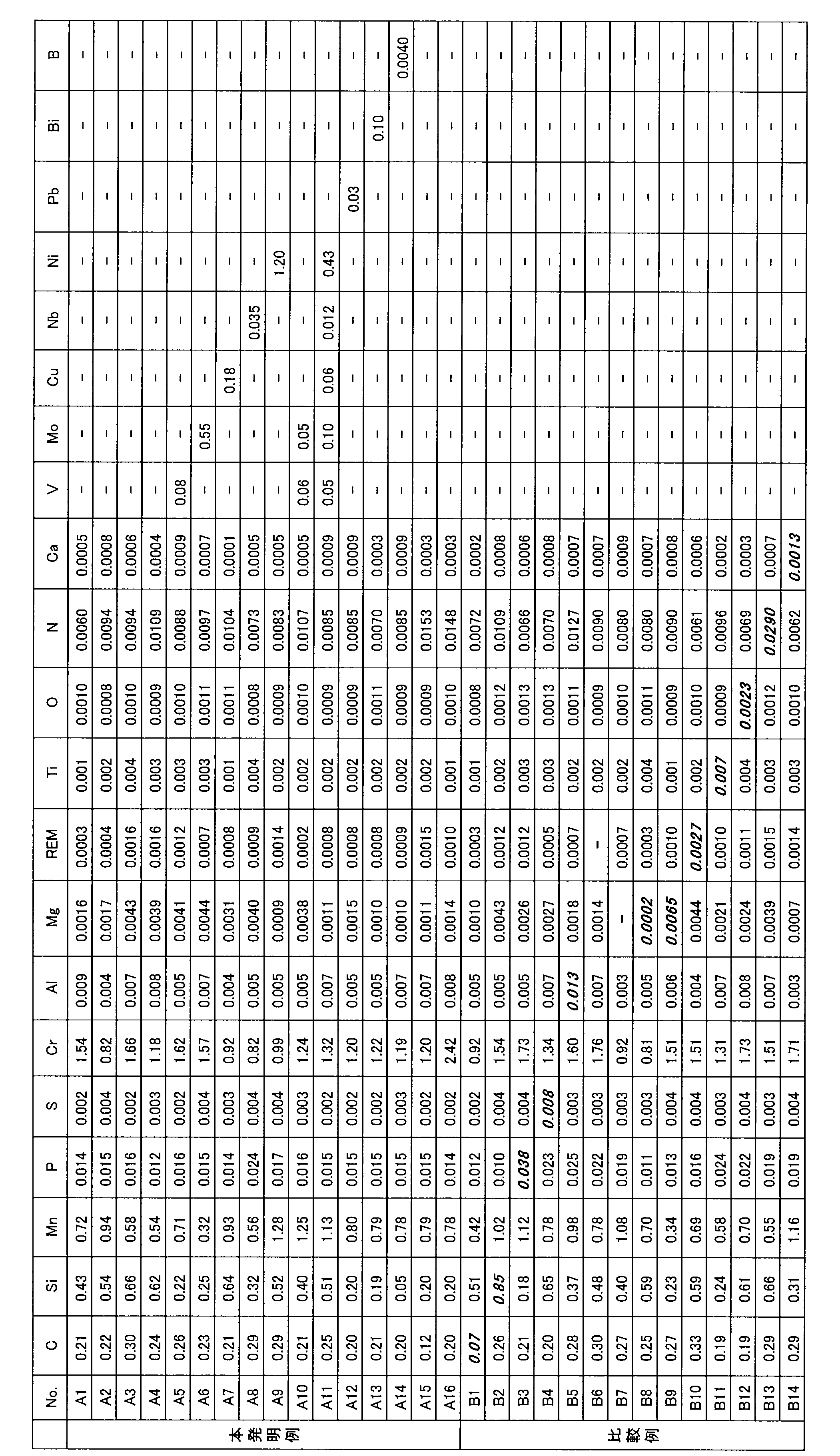
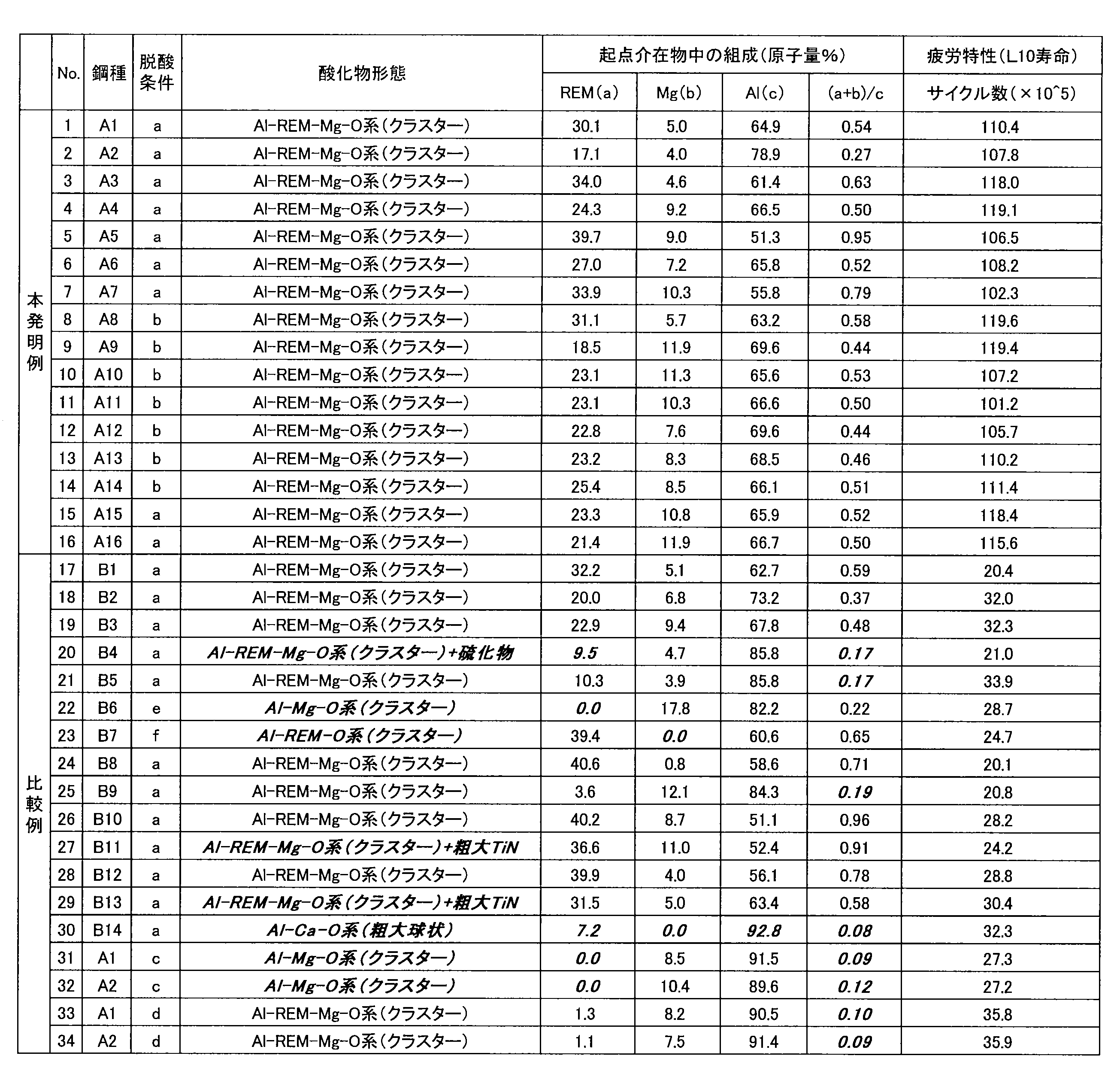
- Example 1 An example in which bearings made of skin burning were assumed No. 1 shown in Table 1.
- A1 to A16 (invention examples), no. A steel grade of each component of B1 to B14 (comparative example) was cast in a 150 kg vacuum melting furnace.
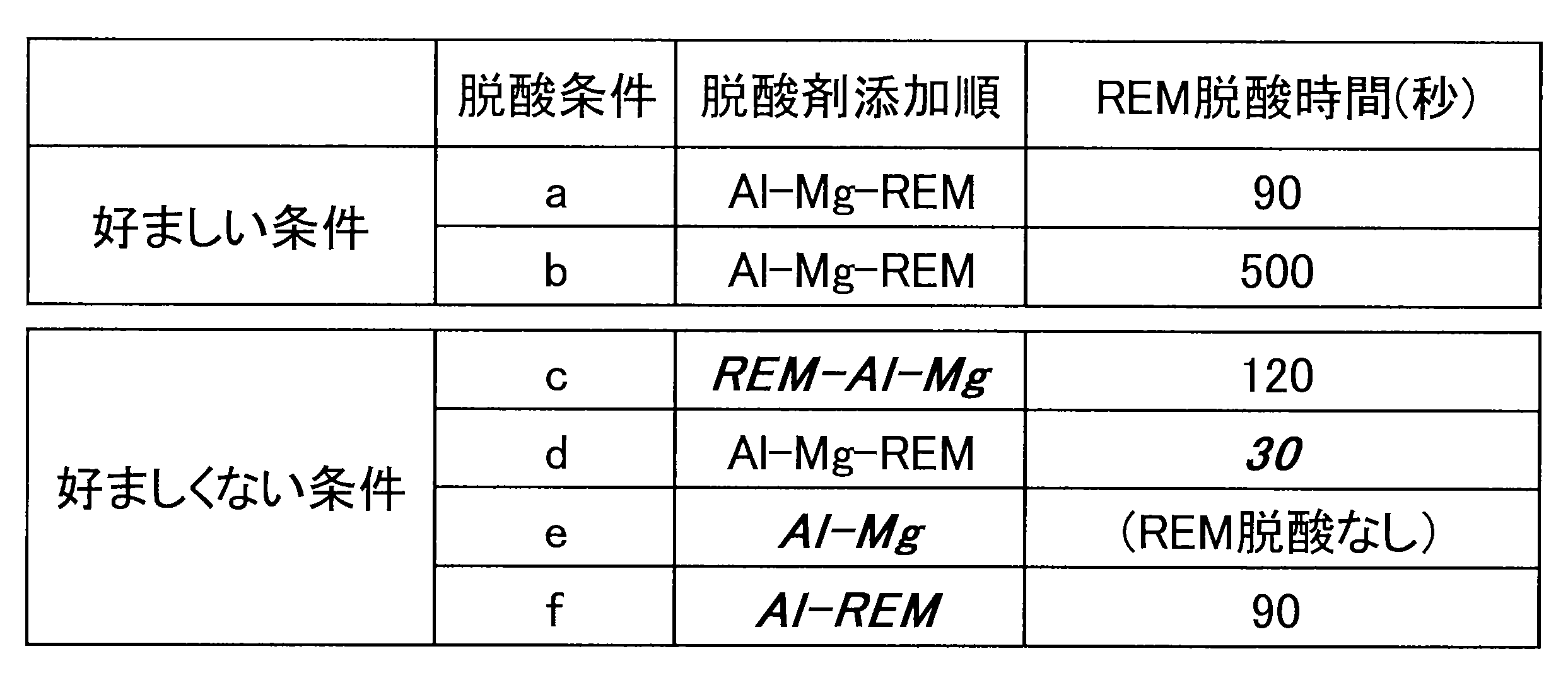
- the deoxidizing conditions were changed as deoxidizing conditions a to f shown in Table 2, and the effects were investigated.
- REM is added, misch metal is added at an estimated yield of 40%.
- a deoxidizer was added in the order of Al, Mg and REM, and steel was extracted after 90 seconds from the addition of REM under deoxidation condition a.
- deoxidizing condition b Under deoxidizing condition b, it was confirmed that 500 seconds had elapsed after addition of REM and steel was released. Under deoxidation condition d, steel was extracted immediately after 30 seconds from the addition of REM.
- deoxidizing condition c a deoxidizing agent was added in the order of REM, Al, and Mg, and the deoxidizing time by REM was 120 seconds.
- deoxidizing conditions e deoxidizing was performed by adding a deoxidizing agent in the order of Al and Mg, and deoxidizing by adding REM was not performed.
- deoxidation condition f a deoxidizer was added in the order of Al and REM to perform deoxidation, and it was confirmed that 90 seconds had elapsed since REM was added and steel was released.
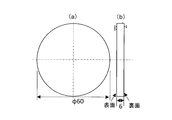
- the rolling fatigue test piece shown in FIG. 1 was extract
- the rolling fatigue test piece is a disc having a thickness of 6.0 mm and a diameter of 60 mm, and the circular surface is perpendicular to the longitudinal direction of the original round bar.
- This rolling fatigue test piece imitates the inner ring and the outer ring in the bearing.
- the circular surface of the rolling fatigue test piece corresponds to the test surface, and the same surface comes into contact with the rolling elements to apply a fatigue load.
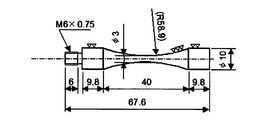
- the evaluation of the fatigue origin inclusions was carried out by an ultrasonic fatigue test using a test piece as shown in FIG.
- the ultrasonic fatigue test pieces for the same test were also collected from the round bar used as the material of the rolling fatigue test pieces.
- the ultrasonic fatigue test pieces were collected such that the longitudinal direction of the test pieces was perpendicular to the longitudinal direction of the raw material round bar.
- the test part is carburized for a sufficiently long time so that the carbon concentration of the surface layer becomes equivalent to the rolling fatigue test piece and carburization can be performed to the center part of the test part, and then hardening treatment and 1hr at 180 ° C. A tempering process was performed. Thereafter, it was finished into a predetermined ultrasonic test piece shape.
- the ultrasonic fatigue test was conducted at a frequency of 20 kHz, a stress ratio of -1, and a constant stress amplitude of 700 to 850 MPa until fracture.
- the composition was analyzed using EDX (energy dispersive X-ray analysis) for the fatigue origin inclusions in this ultrasonic fatigue test, and the total amount of atomic weight% of REM and the atomic weight% of Mg and Al were measured.
- EDX energy dispersive X-ray analysis
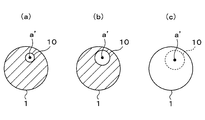
- the fatigue origin inclusions are identified as follows. As shown in FIG. 3, a large number of inclusions a are included between mark points L of the ultrasonic fatigue test piece 1. Among these inclusions a, fatigue failure occurs starting from the inclusion a ′ that generates stress concentration most in the ultrasonic fatigue test.
- the inclusion a ′ that generates the stress concentration most is a fatigue origin inclusion whose stress concentration coefficient is the largest in the ultrasonic fatigue test depending on the size, shape, and the like.
- FIG. 4 is an explanatory view schematically showing how fatigue failure progresses gradually.
- a circular fatigue crack is generated in a cross section perpendicular to the longitudinal direction of the ultrasonic fatigue test piece 1, centering on the inclusion a ′ (the fatigue origin inclusion) .
- the torn surface 10 spreads circularly with the increase in the frequency
- the fractured surface 10 becomes a certain size with the increase of the number of times of stress amplitude, as shown in FIG.
- observation is performed at an acceleration voltage of 20 kV and a magnification of 500 times.
- An example of the reflection electron composition image of the origin inclusion used for composition analysis is shown in FIG.
- region (inclusion area) corresponding to the inclusion in a visual field was specified, the area was extracted, and the chemical composition was measured.
- the major axis of the inclusion is about 100 to 300 ⁇ m, and even when the entire inclusion does not fit in the field of view, the value obtained in one field of view is used as the composition of the inclusion.
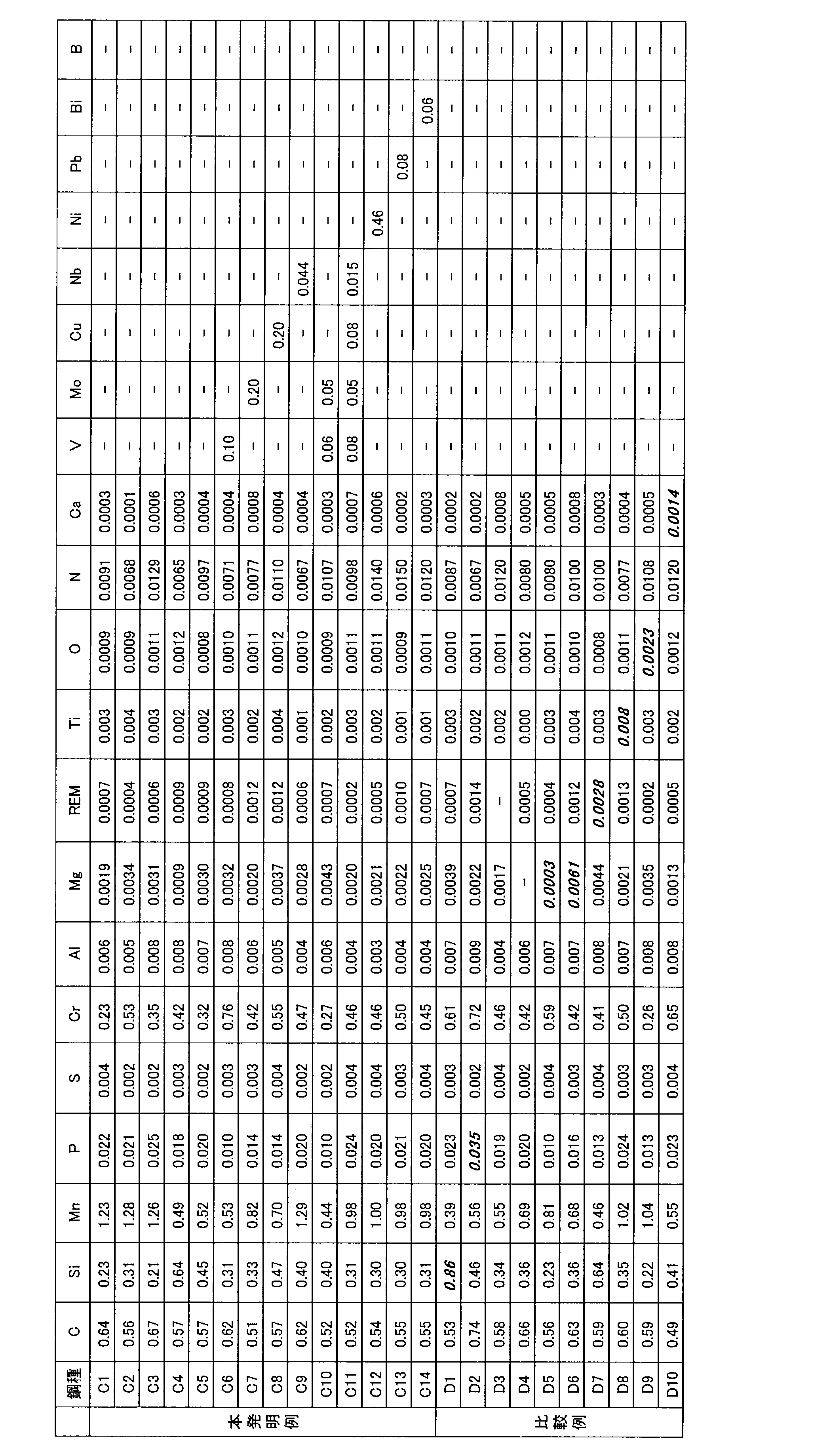
- Table 1 shows the chemical compositions of the respective steel types in Example 1.
- Table 2 shows the deacidification conditions a to f.
- Table 3 shows steel types at various levels (No. 1 to 34), deoxidizing conditions, oxide forms of inclusions of fatigue origin in ultrasonic fatigue test, composition and fatigue characteristics (L10 life) in rolling fatigue test. .
- the fatigue life L10 of the inventive example was 10 7 cycles or more, and was superior to the steel grade serving as the comparative example.
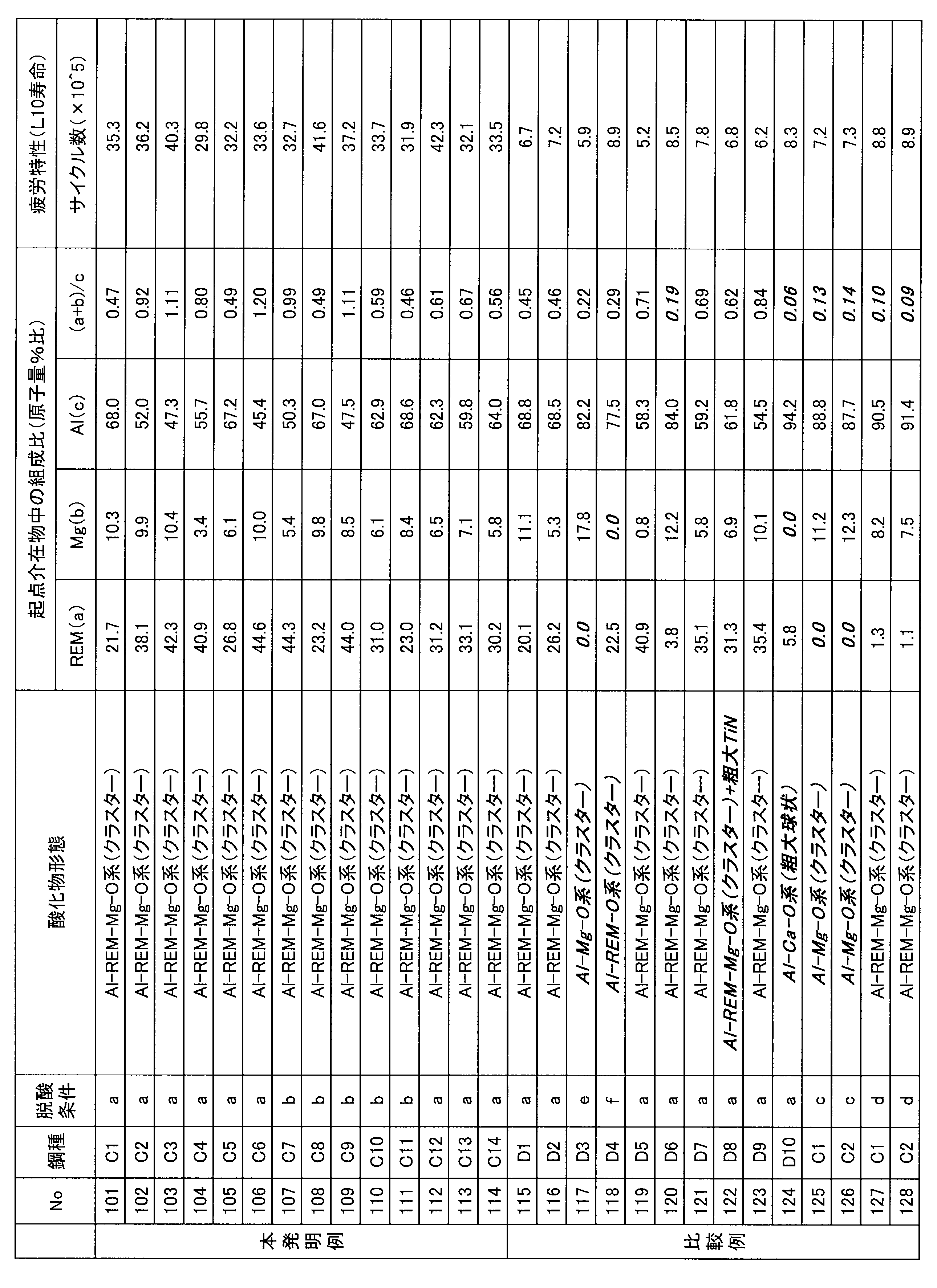
- Example 2 Example assuming bearings by induction hardening No. 1 shown in Table 4.
- C1 to C14 invention examples
- the steel types of the respective components D1 to D10 comparativative examples were cast in a 150 kg vacuum melting furnace, and hot forged into a round bar of ⁇ 80 mm (material for collecting test pieces) as in Example 1.
- the deacidification conditions were the same as in Example 1 under the deacidification conditions a to f shown in Table 2.
- the test part was subjected to induction hardening and tempering at 150 ° C. for 1 hour.
- the induction hardening process was performed under the condition that the surface hardness after tempering became 650 Hv (measurement load 2.94 N) or more.
- test surface was finished to a mirror surface and subjected to a rolling fatigue test.
- the rolling fatigue test was conducted with a Mori type thrust tester (contact surface pressure: 5.33 GPa). For 10 test results for each level (No. 1 to 28 in Table 5), using Weibull statistics, the number of cycles at which 10% of the evaluation samples were broken was evaluated as fatigue characteristics L10.
- the evaluation of the fatigue origin inclusions was carried out by the same ultrasonic fatigue test as in Example 1.
- the heat treatment of the ultrasonic fatigue test pieces was induction hardening in the test section and then tempering at 150 ° C. for 1 hour.
- the induction hardening process was performed after tempering on the conditions which become 650 Hv (measurement load 2.94 N) or more from the test part surface to the center.
- the fatigue test was conducted at a frequency of 20 kHz, a stress ratio of -1, and a constant stress amplitude of 700 to 850 MPa until failure.
- the composition of the starting inclusions in this ultrasonic fatigue test was analyzed using EDX (energy dispersive X-ray analysis), and the total amount of atomic weight% of REM and the atomic weight% of Mg and Al were measured. Also in Table 5, the proportion of O is not considered in the calculation of atomic weight% as in Table 3, but in any of the examples, the starting point inclusion contained O.
- Table 4 shows the chemical compositions of the respective steel types in Example 2.
- Table 5 shows steel types at various levels (No. 101 to 128), deoxidizing conditions, oxide forms of inclusions of fatigue origin in ultrasonic fatigue test, composition and fatigue characteristics (L10 life) in rolling fatigue test. .
- the fatigue characteristics L10 of the inventive example containing an appropriate amount of REM were 10 6 cycles or more, and were superior to the steel types serving as comparative examples.
- Example 3 An example in which the bearing was assumed to be totally quenched No. 1 shown in Table 6.
- E1 to E12 (invention examples), no. A steel grade of each component of F1 to F12 (comparative example) was cast in a 150 kg vacuum melting furnace, and hot forged into a round bar of ⁇ 80 mm (material for collecting test pieces) in the same manner as in Example 1.
- the deacidification conditions were the same as in Example 1 under the deacidification conditions a to f shown in Table 2.
- the test pieces were heated to 850 ° C. and then quenched, and then tempered at 180 ° C. for 1 hour. Furthermore, the test surface was finished to a mirror surface and subjected to a rolling fatigue test.
- the rolling fatigue test was conducted with a Mori type thrust tester (contact surface pressure: 5.33 GPa). For 10 test results for each level (No. 201 to 228 in Table 7), using Weibull statistics, the number of cycles at which 10% of the evaluation samples were broken was evaluated as fatigue characteristics L10.
- the evaluation of the fatigue origin inclusions was carried out by the same ultrasonic fatigue test as in Example 1.
- the heat treatment of the ultrasonic fatigue test piece was performed under the same conditions as the rolling fatigue test piece.
- the fatigue test was conducted at a frequency of 20 kHz, a stress ratio of -1, and a constant stress amplitude of 700 to 850 MPa until failure.
- the composition of the starting inclusions in the ultrasonic fatigue test was analyzed using EDX (energy dispersive X-ray analysis), and the total amount of REM and the atomic weight% of Al and Mg were measured. Also in Table 7, the proportion of O is not considered in the calculation of atomic weight% as in Table 3, but in any of the examples, the starting point inclusion contained O.
- Table 6 shows the chemical compositions of the respective steel types in Example 3.
- Table 7 shows steel types at various levels (No. 201 to 228), deoxidizing conditions, oxide forms of inclusions of fatigue origin in ultrasonic fatigue test, composition and fatigue characteristics (L10 life) in rolling fatigue test. .
- the fatigue characteristics L10 of the inventive example containing an appropriate amount of REM were 5.0 ⁇ 10 6 cycles or more, and were superior to the steel types serving as comparative examples.
- a steel material having excellent fatigue properties can be provided by combining cluster-like Al-O-based inclusions with REM-Al-Mg-O-based inclusions.
Landscapes
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Metallurgy (AREA)
- Organic Chemistry (AREA)
- Mechanical Engineering (AREA)
- Heat Treatment Of Steel (AREA)
- Treatment Of Steel In Its Molten State (AREA)
- Heat Treatment Of Articles (AREA)
Abstract
Description
転がり軸受は、例えば玉形状やコロ形状をした「転動体」と、転動体に接して荷重を伝える「内輪」及び「外輪」」とを備える。転動体や内輪、外輪といった転動部材に使用される鋼材には、優れた転動疲労特性が要求されている。鋼材に含まれる介在物は、転動疲労寿命を向上させる目的から、できるだけ微細でかつ少量であることが望まれている。鋼材に含まれる介在物としては、アルミナ(Al2O3)等の酸化物、硫化マンガン(MnS)等の硫化物、窒化チタン(TiN)等の窒化物が知られている。
[1]
質量%で、
C:0.10%~1.50%、
Si:0.01%~0.80%、
Mn:0.10%~1.50%、
Cr:0.02%~2.50%、
Al:0.002%~0.010%未満、
Ce+La+Nd:0.0001%~0.0025%、
Mg:0.0005%~0.0050%、
O:0.0001%~0.0020%、
Ti:0.000%~0.005%未満、
N:0.0180%以下、
P:0.030%以下、
S:0.005%以下、
Ca:0.0000%~0.0010%、
V:0.00~0.40%、
Mo:0.00~0.60%、
Cu:0.00~0.50%、
Nb:0.000~0.050%未満、
Ni:0.00~2.50%、
Pb:0.00~0.10%、
Bi:0.00~0.10%、
B:0.0000~0.0050%、
残部がFeおよび不純物であり、
超音波疲労試験によって検出される疲労起点介在物がCe、La、Ndの1種以上と、Mg、Al,および、Oを含有し、かつ、その組成比が式(1)を満たすことを特徴とする、転動疲労特性に優れた鋼材。
(Ce%+La%+Nd%+Mg%)/Al%≧0.20 ・・・ 式(1)
但し、式(1)において、Ce%、La%、Nd%、Mg%、Al%は、それぞれ、疲労起点介在物が含有するCe、La、Nd、Mg、Alの原子量%である。
[2]
質量%で、C:0.10%~0.45%未満であり、Cr:0.02~1.50%であることを特徴とする、[1]に記載の転動疲労特性に優れた鋼材。
[3]
質量%で、C:0.45%~0.90%未満であり、Cr:0.70~2.50%であることを特徴とする、[1]に記載の転動疲労特性に優れた鋼材。
[4]
質量%で、C:0.90%~1.50%であり、Cr:0.70~2.50%であることを特徴とする、[1]に記載の転動疲労特性に優れた鋼材。
(1)クラスター状の酸化物であるAl-O系介在物をREM-Al-Mg-O系介在物に改質することで、酸化物系介在物と母材との密着性が向上する。
(2)クラスター状の酸化物であるAl-O系介在物にMgとREM系の介在物を混在させるためには、MgおよびREMとの反応性が高いSは極力含有量を抑えるべきである。
(3)クラスター状の酸化物であるAl-O系介在物は粗大であり、疲労特性に悪影響を及ぼすため、Al量は極力抑えるほうがいいが、非添加では粗大な低級酸化物が形成され、MgおよびREMによる酸化物系介在物の改質効果が得られない。よって、Alの脱酸は最低限必要である。
まず、本実施形態に係る鋼材の成分組成とその限定理由について説明する。なお、下記の元素の含有量に関する%は、質量%を意味する。
Cは、硬さを確保して、疲労寿命を向上させる元素である。所要の強度と硬さとを確保するためには、Cを0.10%以上含有させる必要がある。しかし、C含有量が1.50%を超えると硬さが上昇しすぎて、焼割れの原因となる。したがって、C含有量は、0.10%~1.50%とする。なお、C:0.10%~0.45%未満であれば、肌焼用の鋼材に適している。C:0.45%~0.90%未満であれば、高周波焼入れ用の鋼材に適している。C:0.90%~1.50%であれば、全体焼入れ用の鋼材に適している。また、C含有量の下限は、好ましくは、0.15%である。C含有量の上限は、好ましくは1.35%である。
Siは、焼入れ性を高めて、疲労寿命を向上させる元素である。この効果を得るためには、Siを0.01%以上含有させる必要がある。しかし、Si含有量が、0.80%を超えると、焼入れ性向上効果が飽和し、さらに、脱酸状態に影響し、酸化物の形成に影響を及ぼし疲労特性が低下する。したがって、Si含有量は0.01%~0.80%とする。また、Si含有量の下限は、好ましくは、0.07%とする。Si含有量の上限は、好ましくは0.65%以下とする。
Mnは、焼入れ性を高めて強度を高め、疲労寿命を向上させる元素である。この効果を得るためには、Mnを0.10%以上含有させる必要がある。しかし、Mn含有量が、1.50%を超えると、焼入れ性向上効果が飽和し、かえって、焼割れの原因となる。そのため、Mn含有量は0.10%~1.50%とする。Mn含有量の下限は、好ましくは、0.20%とする。Mn含有量の上限は、好ましくは、1.20%とする。
Crは、焼入れ性を高めて、疲労寿命を向上させる元素である。この効果を安定して得るためには、Crを0.02%以上含有させることが好ましい。しかし、Cr含有量が2.50%を超えると、焼入れ性向上効果が飽和し、かえって焼割れの原因となる。そのため、Cr含有量の上限は2.50%とする。また、Cr含有量の下限は、好ましくは0.15%以上とする。Cr含有量の上限は、好ましくは2.00%以下とする。Cr含有量は、1.90%以下、もしくは1.80%以下と規定してもよい。
なお、肌焼による軸受用の鋼材として用いる場合には、Cr:0.02~1.50%とすることが望ましい。高周波焼き入れによる軸受用の鋼材、または全体焼き入れによる軸受用の鋼材として用いる場合には、Cr:0.70~2.50%とすることが望ましい。
Alは、T.O(全酸素量)を低減する脱酸元素として、0.002%以上を含有させる必要がある。しかし、Al含有量が0.010%以上では、クラスター状のアルミナ量が増加し、MgおよびREM添加によるREM-Al-Mg-O系介在物への改質が十分できないと考えられる。そのため、Al含有量は0.010%未満とする。Al含有量は、好ましくは、下限について0.005%以上とする。Al含有量は、好ましくは、上限について、0.008%以下とする。
Ce(セリウム)、La(ランタン)、およびNd(ネオジム)は希土類元素に分類される元素である。希土類元素とは、原子番号が57のランタンから71のルテシウムまでの15元素に、原子番号が21のスカンジウムと原子番号が39のイットリウムとを加えた合計17元素の総称である。希土類元素は、強力な脱酸元素であり、本実施形態に係る軸受け用鋼材において、極めて重要な役割を担う。製鋼用の希土類元素合金は、Ce、La、および、Ndの3元素が主成分となっているため、本発明では、希土類17元素のうち、Ce、La、および、Ndを限定する。希土類合金に含まれる3元素以外の元素も強力な脱酸元素であることに変わりなく、3元素と同様の効果を発揮する。Ce、La、Ndのいずれか1種を0.0001%~0.0025%含有しても良いし、2種以上を合計で0.0001%~0.0025%含有しても良い。本発明における説明ではCe、La、および、NdをREMと総称する。REMは、まず、溶鋼中の酸素と反応して、REM系の酸化物を生成する。次いで、溶鋼中でのアルミナ酸化物の凝集クラスター化に伴ってREM系酸化物も同時に取り込まれる。そうすることでAl-O系酸化物がREM-Al-Mg-O系介在物へと改質される。
これらの観点から検討した結果、REMが0.0001%未満では含有効果が不十分であることを実験的に知見した。したがって、REM含有量の下限を0.0001%とし、好ましくは、0.0003%以上、より好ましくは、0.0008%以上とする。ただし、REM含有量が0.0025%を超えると、コスト高となるだけでなく、鋳造ノズルの閉塞が発生し易くなり、鋼の製造を阻害する。したがって、REMの含有量の上限は0.0025%であり、好ましくは0.0020%、より好ましくは0.0018%である。
Mgは、Alと同様に強力な脱酸元素であり、本実施形態に係る鋼材において、極めて重要な役割を担う。Mgだけではクラスター酸化物起点の破壊を抑制する効果は小さいが、REMとMgの併用により、REM単独よりもその疲労特性の改善効果が高まる。本効果を得るためには、Mgを0.0005%以上の量で含有させる必要がある。Mg含有量が多いと、酸化物量そのものが増加しREM添加によるREM-Al-Mg-O系介在物を改質ができないと考えられる。そのため、Mg含有量は0.0050%以下とする。Mg含有量は、好ましくは下限を0.0010%以上と規定する。Mg含有量は、好ましくは、上限を0.0040%以下と規定する。
Oは不純物であり、脱酸により鋼から除去される元素である。脱酸により鋼中のOを皆無とすることができればアルミナクラスターは発生せず、本発明が解決しようとする課題はもとより生じない。しかし、技術面およびコスト面から、現在の鋼においては0.0001%以上のOが必然的に含有され、これにより生じるアルミナクラスターにより疲労特性が低下しうる。本発明は、通常含まれるのと同程度の酸素を含有する鋼において、従来に比べて疲労特性を改善したものである。一般的には、鋼のO含有量は0.0005%以上であることが多い。一方、O含有量が0.0020%を超えると、アルミナなどの酸化物が多量に残存し、疲労寿命が低下するので、O含有量の上限を0.0020%とする。O含有量は、好ましくは0.0015%以下である。
クラスター状の酸化物であるAl-O系介在物をREM-Al-Mg-O系介在物に改質することで、酸化物系介在物と母材との密着性が向上し、疲労特性が向上する。この効果は、疲労起点介在物におけるCe、La、Nd、Mg、Alの組成比(Ce%+La%+Nd%+Mg%)/Al%が0.20%以上の場合に発現する。したがって、(Ce%+La%+Nd%+Mg%)/Al%を0.20以上とする。上記の効果をより高めるためには、(Ce%+La%+Nd%+Mg%)/Al%は0.50以上が好ましい。
Tiは不純物であり、鋼中に存在すると、TiNを生成して疲労特性を劣化させるので、Ti含有量を0.005%未満に制限する。好ましくはTi含有量を0.004%以下に制限する。
Nは不純物であり、鋼中に存在すると、窒化物を形成して疲労特性を劣化させ、また、歪時効によって延性及び靭性を劣化させる。N含有量が、0.0180%を超えると、疲労特性、延性、及び、靭性の劣化などの弊害が著しくなる。そのため、N含有量の上限を0.0180%に制限する。好ましくはN含有量を0.0150%以下に制限する。Nは0.0000%でも良いが、N含有量の低減には工業上の制約があり、またあまりに低くすることは意味がない。通常の費用で可能なNの実質的な下限として0.0020%としても良い。
Pは、不純物であり、鋼中に存在すると、結晶粒界に偏析して疲労寿命を低下させる。P含有量が、0.030%を超えると、疲労寿命が低下する。そのため、P含有量の上限を0.030%に制限する。好ましくは、P含有量を0.020%以下に制限する。P含有量の下限は0.000%でもよいが、工業的な下限として0.001%としても良い。
Sは、鋼中に存在すると、硫化物を形成する。S含有量が、0.005%を超えると、SがREMと結合して硫化物を形成し、アルミナクラスターの改質に有効なREMを低減させ、ひいては疲労寿命を低下させる。そのため、S含有量の上限を0.005%に制限する。好ましくは、S含有量を0.0025%以下に制限する。S含有量の下限は0.000%でもよいが、工業的な下限として0.001%としても良い。
Caは、鋼中に存在すると、粗大なCaOが生成して、疲労寿命が低下するので、上限を0.0010%とする。Ca含有量は、好ましくは0.0002%以下であり、0.0000%であればなおよい。
本実施形態に係る鋼材は、さらに、V:0.00%~0.40%、Mo:0.00%~0.60%、Cu:0.00%~0.50%、Nb:0.000%~0.050%、Ni:0.00%~2.50%、Pb:0.00~0.10%、Bi:0.00~0.10%、B:0.0000~0.0050%、の1種以上を含有してもよい。
Vは、鋼中のC及びNと結合して、炭化物、窒化物、又は炭窒化物を形成し、鋼の組織微細化と強化に寄与する元素である。この効果を安定して得るためには、Vを0.05%以上含有させることが好ましい。V含有量は、より好ましくは0.10%以上である。しかし、V含有量が0.40%を超えると、含有効果は飽和するとともに熱間加工時に割れが発生するので、V含有量の上限を0.40%とする。好ましくは、V含有量を0.30%以下とする。
Moは、焼き入れ性を高めるとともに鋼中のCと結合して、炭化物を形成し、析出強化により鋼の強度の向上に寄与する元素である。この効果を安定して得るためには、Moを0.05%以上含有させることが好ましい。Mo含有量は、より好ましくは0.10%以上である。しかし、Mo含有量が0.60%を超えると、かえって焼割れの原因となるので、Mo含有量の上限を0.60%とする。Mo含有量は、好ましくは0.50%以下である。
Cuは、母材の強化による疲労特性の向上に寄与する元素である。この効果を安定して得るためには、Cuを0.05%以上含有させることが好ましい。しかし、Cu含有量が0.50%を超えると、熱間加工時に割れが発生するので、Cu含有量の上限を0.50%とする。Cu含有量は、好ましくは0.35%以下である。
Nbは、母材強化による疲労特性の向上に寄与する元素である。この効果を安定して得るためには、Nbを0.005%以上含有させることが好ましい。Nb含有量は、より好ましくは0.010%以上である。しかし、Nb含有量が0.050%以上になると、含有効果が飽和するとともに熱間加工時に割れが発生するので、Nb含有量を0.050%未満とする。Nb含有量は、好ましくは0.030%以下である。
Niは、耐食性を上げることで疲労寿命の向上に寄与する元素である。この効果を安定して得るためには、Niを0.10%以上含有させることが好ましい。Ni含有量は、より好ましくは0.30%以上である。しかし、Ni含有量が2.50%を超えると、鋼の被削性が低下するので、Ni含有量の上限を2.50%とする。Ni含有量は、好ましくは2.00%以下である。
Pbは、鋼の被削性を高めるために添加される。しかし、Pb含有量が0.10%を超えると疲労き裂の発生起点となり疲労強度が低下する。したがって、Pb含有量の上限は0.10%とする。Pb含有量は好ましくは0.06%以下である。
Biは、鋼の被削性を高めるために添加される。しかし、Bi含有量が0.10%を超えると疲労き裂の発生起点となり疲労強度が低下する。したがって、Bi含有量の上限は0.10%とする。Bi含有量は好ましくは0.06%以下である。
Bは、オーステナイト粒界に偏析し、粒界強度を高めて靭性を改善する効果がある。しかし、B含有量が0.0050%を超えると熱処理時にオーステナイト粒が異常粒成長し疲労強度が低下する。したがって、B含有量の上限は0.0050%とする。B含有量は好ましくは0.0030%以下である。
表1に示すNo.A1~A16(本発明例)、No.B1~B14(比較例)の各成分の鋼種を150kg真空溶解炉で鋳造した。脱酸条件については、表2に示す脱酸条件a~fとして変化させ、影響を調査した。REMを添加する場合には、想定歩留まり40%でミッシュメタルを添加した。
脱酸条件a、b、dは、いずれもAl、Mg、REMの順番で脱酸剤を添加し、脱酸条件aでは、REM添加から90秒経過後、出鋼した。脱酸条件bでは、REM添加から500秒経過したことを確認し出鋼した。脱酸条件dでは、REM添加から30秒経過後すぐに出鋼した。脱酸条件cは、REM、Al、Mgの順番で脱酸剤を添加し、REMによる脱酸時間は120秒とした。脱酸条件eは、Al,Mgの順番で脱酸剤を添加して脱酸を行い、REM添加による脱酸を行わなかった。脱酸条件fは、Al,REMの順番で脱酸剤を添加して脱酸を行い、REM添加から90秒経過したことを確認して出鋼した。
介在物の長径はおよそ100~300μmであり、介在物全体が視野に収まらない場合も、一視野内で得られた値をその介在物の組成として用いる。上記の視野について、ドゥエルタイム0.5μs、プリセット5でEDSによる元素マッピングを実施し、介在物エリアから得られたX線スペクトルよりCe、La、Nd、Mg、Alの原子量%を求め、介在物組成を計測する。EDSのデータ取得及び解析には、EDS分析システムAnalysis
Station(日本電子製)を用いる。なお、表3における原子量%の算出にあたってはOの割合が考慮されていないが、疲労起点介在物はいずれもOを介して形成された複合介在物であり、Oを含んでいた。また、比較例では、疲労起点介在物が他元素(Mn、Ti等)を含むケースもあったが、Ce、La、Nd、Mg、Al以外の元素はいずれも原子量%の算出に考慮されていない。
本発明例の疲労寿命L10は、107サイクル以上であり、比較例となる鋼種より優位であった。
表4に示すNo.C1~C14(本発明例)、No.D1~D10(比較例)の各成分の鋼種を150kg真空溶解炉で鋳造し、実施例1と同様にφ80mmの丸棒(試験片採取のための素材)へと熱間鍛造した。脱酸条件は、実施例1と同様、表2に示す脱酸条件a~fにより行った。転動疲労試験片の採取後、試験部に高周波焼入れ処理と、150℃で1hrの焼戻し処理を行った。高周波焼き入れ処理は、焼き戻し後の表面硬度が650Hv(測定荷重2.94N)以上となる条件にて行った。さらに、試験面を鏡面に仕上げ加工し転動疲労試験に供した。転動疲労試験は森式スラスト型試験機(接触面圧:5.33GPa)で行った。各水準(表5のNo.1~28)に対する10回の試験結果について、ワイブル統計を用いて、評価試料のうちの10%が破壊するサイクル数を疲労特性L10として評価した。
REMを適量含有した、本発明例の疲労特性L10は、106サイクル以上であり、比較例となる鋼種より優位であった。
表6に示すNo.E1~E12(本発明例)、No.F1~F12(比較例)の各成分の鋼種を150kg真空溶解炉で鋳造し、実施例1と同様にφ80mmの丸棒(試験片採取のための素材)へと熱間鍛造した。脱酸条件は、実施例1と同様、表2に示す脱酸条件a~fにより行った。転動疲労試験片の採取後、試験片を850℃に加熱してから焼入れ処理を行い、その後180℃で1hrの焼戻し処理を行った。さらに、試験面を鏡面に仕上げ加工し転動疲労試験に供した。転動疲労試験は森式スラスト型試験機(接触面圧:5.33GPa)で行った。各水準(表7のNo.201~228)に対する10回の試験結果について、ワイブル統計を用いて、評価試料のうちの10%が破壊するサイクル数を疲労特性L10として評価した。
10 破断面
11 同心円状の模様(フィッシュアイ)
L 標点距離
a 介在物
a’ 介在物(疲労起点介在物)
Claims (4)
- 質量%で、
C:0.10%~1.50%、
Si:0.01%~0.80%、
Mn:0.10%~1.50%、
Cr:0.02%~2.50%、
Al:0.002%~0.010%未満、
Ce+La+Nd:0.0001%~0.0025%、
Mg:0.0005%~0.0050%、
O:0.0001%~0.0020%、
Ti:0.000%~0.005%未満、
N:0.0180%以下、
P:0.030%以下、
S:0.005%以下、
Ca:0.0000%~0.0010%、
V:0.00~0.40%、
Mo:0.00~0.60%、
Cu:0.00~0.50%、
Nb:0.000~0.050%未満、
Ni:0.00~2.50%、
Pb:0.00~0.10%、
Bi:0.00~0.10%、
B:0.0000~0.0050%、
残部がFeおよび不純物であり、
超音波疲労試験によって検出される疲労起点介在物がCe、La、Ndの1種以上と、Mg、Al,および、Oを含有し、かつ、その組成比が式(1)を満たすことを特徴とする、転動疲労特性に優れた鋼材。
(Ce%+La%+Nd%+Mg%)/Al%≧0.20 ・・・ 式(1)
但し、式(1)において、Ce%、La%、Nd%、Mg%、Al%は、それぞれ、疲労起点介在物が含有するCe、La、Nd、Mg、Alの原子量%である。 - 質量%で、C:0.10%~0.45%未満であり、Cr:0.02~1.50%であることを特徴とする、請求項1に記載の転動疲労特性に優れた鋼材。
- 質量%で、C:0.45%~0.90%未満であり、Cr:0.70~2.50%であることを特徴とする、請求項1に記載の転動疲労特性に優れた鋼材。
- 質量%で、C:0.90%~1.50%であり、Cr:0.70~2.50%であることを特徴とする、請求項1に記載の転動疲労特性に優れた鋼材。
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020207008332A KR20200044866A (ko) | 2017-09-13 | 2018-09-13 | 전동 피로 특성이 우수한 강재 |
| CN201880057744.3A CN111065755A (zh) | 2017-09-13 | 2018-09-13 | 滚动疲劳特性优异的钢材 |
| JP2019542288A JP6652226B2 (ja) | 2017-09-13 | 2018-09-13 | 転動疲労特性に優れた鋼材 |
| EP18857159.0A EP3647451A4 (en) | 2017-09-13 | 2018-09-13 | STEEL MATERIAL WITH EXCELLENT BEARING FATIGUE CHARACTERISTICS |
| US16/642,700 US20200216937A1 (en) | 2017-09-13 | 2018-09-13 | Steel material excellent in rolling fatigue property |
Applications Claiming Priority (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2017176163 | 2017-09-13 | ||
| JP2017-176161 | 2017-09-13 | ||
| JP2017-176163 | 2017-09-13 | ||
| JP2017176161 | 2017-09-13 | ||
| JP2017-176165 | 2017-09-13 | ||
| JP2017176165 | 2017-09-13 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2019054448A1 true WO2019054448A1 (ja) | 2019-03-21 |
Family
ID=65723945
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2018/034008 WO2019054448A1 (ja) | 2017-09-13 | 2018-09-13 | 転動疲労特性に優れた鋼材 |
Country Status (6)
| Country | Link |
|---|---|
| US (1) | US20200216937A1 (ja) |
| EP (1) | EP3647451A4 (ja) |
| JP (1) | JP6652226B2 (ja) |
| KR (1) | KR20200044866A (ja) |
| CN (1) | CN111065755A (ja) |
| WO (1) | WO2019054448A1 (ja) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US10837488B2 (en) | 2018-07-24 | 2020-11-17 | Roller Bearing Company Of America, Inc. | Roller bearing assembly for use in a fracking pump crank shaft |
| CN112195414A (zh) * | 2020-10-21 | 2021-01-08 | 中泽电气科技有限公司 | 一种配电箱用耐腐蚀不锈钢材料制备方法 |
| JP2021028413A (ja) * | 2019-08-09 | 2021-02-25 | 日本製鉄株式会社 | 浸炭歯車用鋼、浸炭歯車及び浸炭歯車の製造方法 |
Families Citing this family (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR102386638B1 (ko) * | 2017-08-25 | 2022-04-14 | 닛폰세이테츠 가부시키가이샤 | 침탄 베어링 부품용 강재 |
| CN116875901B (zh) * | 2023-07-24 | 2024-06-18 | 鞍钢股份有限公司 | 一种疲劳性能优异的船用720MPa级钢板及制造方法 |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH01279695A (ja) | 1988-04-30 | 1989-11-09 | Nec Home Electron Ltd | 磁気記録再生装置 |
| JPH09263820A (ja) | 1996-03-25 | 1997-10-07 | Kawasaki Steel Corp | クラスターのないAlキルド鋼の製造方法 |
| JPH11279695A (ja) * | 1998-03-27 | 1999-10-12 | Kawasaki Steel Corp | 軸受用鋼線材およびその製造方法 |
| JP2005002420A (ja) * | 2003-06-12 | 2005-01-06 | Nippon Steel Corp | アルミナクラスターの少ない鋼材とその製造方法 |
| JP2009287111A (ja) * | 2008-06-02 | 2009-12-10 | Kobe Steel Ltd | 機械構造用鋼 |
| WO2015076384A1 (ja) * | 2013-11-22 | 2015-05-28 | 新日鐵住金株式会社 | 高炭素鋼板及びその製造方法 |
Family Cites Families (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2002042508A1 (fr) * | 2000-11-21 | 2002-05-30 | Nippon Yakin Kogyo Co., Ltd. | Materiau en alliage nickel-fer destine a un masque perfore parfaitement adapte a la gravure |
| KR100460346B1 (ko) * | 2002-03-25 | 2004-12-08 | 이인성 | 금속간상의 형성이 억제된 내식성, 내취화성, 주조성 및열간가공성이 우수한 슈퍼 듀플렉스 스테인리스강 |
| JP5208354B2 (ja) * | 2005-04-11 | 2013-06-12 | 新日鐵住金株式会社 | オーステナイト系ステンレス鋼 |
| US8475605B2 (en) * | 2010-03-19 | 2013-07-02 | Nippon Steel & Sumitomo Metal Corporation | Surface layer-hardened steel part and method of manufacturing the same |
| MY156205A (en) * | 2010-06-08 | 2016-01-29 | Nippon Steel & Sumitomo Metal Corp | Steel for steel tube with excellent sulfide stress cracking resistance |
| AT509598B1 (de) * | 2010-10-18 | 2011-10-15 | Boehler Edelstahl Gmbh & Co Kg | Verfahren zur herstellung von werkzeugen aus legiertem stahl und werkzeuge insbesondere zur spanabhebenden bearbeitung von metallen |
| WO2012160675A1 (ja) * | 2011-05-25 | 2012-11-29 | 株式会社神戸製鋼所 | 転動疲労特性に優れた鋼材 |
| CN103890209B (zh) * | 2011-10-20 | 2015-11-25 | 新日铁住金株式会社 | 轴承钢及其制造方法 |
| JP5867262B2 (ja) * | 2012-04-23 | 2016-02-24 | 新日鐵住金株式会社 | 耐遅れ破壊特性に優れたレール |
| WO2014061784A1 (ja) * | 2012-10-19 | 2014-04-24 | 新日鐵住金株式会社 | 疲労特性に優れる肌焼鋼 |
| EP2990497B1 (en) * | 2013-04-24 | 2018-06-06 | Nippon Steel & Sumitomo Metal Corporation | Low-oxygen clean steel and low-oxygen clean steel product |
| US10435765B2 (en) * | 2014-04-24 | 2019-10-08 | Nippon Steel Corporation | Wire rod for high strength steel cord |
| JP6462376B2 (ja) * | 2015-01-23 | 2019-01-30 | 株式会社神戸製鋼所 | 転動疲労特性に優れた軸受用鋼材および軸受部品 |
| MX2017012194A (es) * | 2015-03-25 | 2017-12-15 | Jfe Steel Corp | Lamina de acero laminada en frio y metodo de fabricacion para la misma. |
| WO2017090738A1 (ja) * | 2015-11-27 | 2017-06-01 | 新日鐵住金株式会社 | 鋼、浸炭鋼部品、及び浸炭鋼部品の製造方法 |
-
2018
- 2018-09-13 CN CN201880057744.3A patent/CN111065755A/zh active Pending
- 2018-09-13 EP EP18857159.0A patent/EP3647451A4/en not_active Withdrawn
- 2018-09-13 KR KR1020207008332A patent/KR20200044866A/ko not_active Application Discontinuation
- 2018-09-13 JP JP2019542288A patent/JP6652226B2/ja active Active
- 2018-09-13 US US16/642,700 patent/US20200216937A1/en not_active Abandoned
- 2018-09-13 WO PCT/JP2018/034008 patent/WO2019054448A1/ja unknown
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH01279695A (ja) | 1988-04-30 | 1989-11-09 | Nec Home Electron Ltd | 磁気記録再生装置 |
| JPH09263820A (ja) | 1996-03-25 | 1997-10-07 | Kawasaki Steel Corp | クラスターのないAlキルド鋼の製造方法 |
| JPH11279695A (ja) * | 1998-03-27 | 1999-10-12 | Kawasaki Steel Corp | 軸受用鋼線材およびその製造方法 |
| JP2005002420A (ja) * | 2003-06-12 | 2005-01-06 | Nippon Steel Corp | アルミナクラスターの少ない鋼材とその製造方法 |
| JP2009287111A (ja) * | 2008-06-02 | 2009-12-10 | Kobe Steel Ltd | 機械構造用鋼 |
| WO2015076384A1 (ja) * | 2013-11-22 | 2015-05-28 | 新日鐵住金株式会社 | 高炭素鋼板及びその製造方法 |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP3647451A4 * |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US10837488B2 (en) | 2018-07-24 | 2020-11-17 | Roller Bearing Company Of America, Inc. | Roller bearing assembly for use in a fracking pump crank shaft |
| JP2021028413A (ja) * | 2019-08-09 | 2021-02-25 | 日本製鉄株式会社 | 浸炭歯車用鋼、浸炭歯車及び浸炭歯車の製造方法 |
| JP7295417B2 (ja) | 2019-08-09 | 2023-06-21 | 日本製鉄株式会社 | 浸炭歯車用鋼、浸炭歯車及び浸炭歯車の製造方法 |
| CN112195414A (zh) * | 2020-10-21 | 2021-01-08 | 中泽电气科技有限公司 | 一种配电箱用耐腐蚀不锈钢材料制备方法 |
| CN112195414B (zh) * | 2020-10-21 | 2021-10-29 | 中泽电气科技有限公司 | 一种配电箱用耐腐蚀不锈钢材料制备方法 |
Also Published As
| Publication number | Publication date |
|---|---|
| EP3647451A1 (en) | 2020-05-06 |
| JP6652226B2 (ja) | 2020-02-19 |
| US20200216937A1 (en) | 2020-07-09 |
| EP3647451A4 (en) | 2020-11-04 |
| KR20200044866A (ko) | 2020-04-29 |
| CN111065755A (zh) | 2020-04-24 |
| JPWO2019054448A1 (ja) | 2020-03-26 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5652555B2 (ja) | 軸受鋼とその製造方法 | |
| JP6652226B2 (ja) | 転動疲労特性に優れた鋼材 | |
| JP6036997B2 (ja) | 耐疲労特性に優れたばね鋼及びその製造方法 | |
| KR101492782B1 (ko) | 강판 | |
| JP5794397B2 (ja) | 疲労特性に優れる肌焼鋼 | |
| WO2010050238A1 (ja) | 耐摩耗性および靭性に優れたパーライト系レール | |
| JP5794396B2 (ja) | 疲労特性に優れる高周波焼入れ用鋼 | |
| WO2016148037A1 (ja) | 冷間加工性と浸炭熱処理後の靱性に優れる浸炭用鋼板 | |
| JP6628014B1 (ja) | 浸炭処理が行われる部品用の鋼材 | |
| CN112981237A (zh) | 一种球笼式万向节保持架用钢及其生产方法 | |
| CN115261715A (zh) | 一种高温渗碳齿轴用钢及其制造方法 | |
| JP2012132094A (ja) | 軸受材料及び軸受材料の製造方法 | |
| JP5316495B2 (ja) | 軸受鋼鋼材 | |
| WO2018174270A1 (ja) | 線材、及び平鋼線 | |
| JP6301145B2 (ja) | スリーブ・ドッグギヤ | |
| WO2018212196A1 (ja) | 鋼及び部品 | |
| JP2019011510A (ja) | 冷間加工性と浸炭熱処理後の靱性に優れる浸炭用鋼板 | |
| CN111876679B (zh) | 铬钒系热轧钢盘条及其制备方法、以及钢丝和手工具的制备方法 | |
| JP4280923B2 (ja) | 浸炭部品又は浸炭窒化部品用の鋼材 | |
| JP6635100B2 (ja) | 肌焼鋼 | |
| JP2013001940A (ja) | 軸受材料 | |
| WO2019142946A1 (ja) | 軸受鋼部品、および軸受鋼部品用棒鋼 | |
| JP2005307257A5 (ja) | ||
| WO2024169713A1 (zh) | 一种车辆轮毂用轴承钢及其制造方法 | |
| CN115537678B (zh) | 一种高温渗碳齿轮用钢及其制造方法 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 18857159 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2019542288 Country of ref document: JP Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 2018857159 Country of ref document: EP Effective date: 20200128 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 20207008332 Country of ref document: KR Kind code of ref document: A |