WO2019022555A1 - 알칼리 퓨전을 통한 폐탈질촉매로부터 선택적 유가금속 회수방법 - Google Patents
알칼리 퓨전을 통한 폐탈질촉매로부터 선택적 유가금속 회수방법 Download PDFInfo
- Publication number
- WO2019022555A1 WO2019022555A1 PCT/KR2018/008523 KR2018008523W WO2019022555A1 WO 2019022555 A1 WO2019022555 A1 WO 2019022555A1 KR 2018008523 W KR2018008523 W KR 2018008523W WO 2019022555 A1 WO2019022555 A1 WO 2019022555A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- alkali
- leaching
- calcium
- tungsten
- denitration catalyst
- Prior art date
Links
- 239000003054 catalyst Substances 0.000 title claims abstract description 106
- 230000004927 fusion Effects 0.000 title claims abstract description 99
- 239000002699 waste material Substances 0.000 title claims abstract description 98
- 238000000034 method Methods 0.000 title claims abstract description 91
- 239000003513 alkali Substances 0.000 title claims abstract description 80
- 229910052751 metal Inorganic materials 0.000 title claims abstract description 73
- 239000002184 metal Substances 0.000 title claims abstract description 73
- 238000011084 recovery Methods 0.000 title claims abstract description 62
- 238000002386 leaching Methods 0.000 claims abstract description 123
- 239000011575 calcium Substances 0.000 claims abstract description 68
- 238000001556 precipitation Methods 0.000 claims abstract description 54
- 238000006243 chemical reaction Methods 0.000 claims abstract description 48
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 claims abstract description 41
- 229910052791 calcium Inorganic materials 0.000 claims abstract description 41
- 229910052783 alkali metal Inorganic materials 0.000 claims abstract description 35
- 150000001340 alkali metals Chemical class 0.000 claims abstract description 35
- 239000002253 acid Substances 0.000 claims abstract description 30
- 238000003756 stirring Methods 0.000 claims abstract description 22
- PBYZMCDFOULPGH-UHFFFAOYSA-N tungstate Chemical compound [O-][W]([O-])(=O)=O PBYZMCDFOULPGH-UHFFFAOYSA-N 0.000 claims abstract description 17
- DNWNZRZGKVWORZ-UHFFFAOYSA-N calcium oxido(dioxo)vanadium Chemical compound [Ca+2].[O-][V](=O)=O.[O-][V](=O)=O DNWNZRZGKVWORZ-UHFFFAOYSA-N 0.000 claims abstract description 13
- 238000001354 calcination Methods 0.000 claims abstract description 11
- CMPGARWFYBADJI-UHFFFAOYSA-L tungstic acid Chemical compound O[W](O)(=O)=O CMPGARWFYBADJI-UHFFFAOYSA-L 0.000 claims abstract description 5
- 238000002156 mixing Methods 0.000 claims abstract description 3
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 claims description 264
- CDBYLPFSWZWCQE-UHFFFAOYSA-L Sodium Carbonate Chemical compound [Na+].[Na+].[O-]C([O-])=O CDBYLPFSWZWCQE-UHFFFAOYSA-L 0.000 claims description 43
- GWEVSGVZZGPLCZ-UHFFFAOYSA-N Titan oxide Chemical compound O=[Ti]=O GWEVSGVZZGPLCZ-UHFFFAOYSA-N 0.000 claims description 39
- 239000011734 sodium Substances 0.000 claims description 34
- 229910000029 sodium carbonate Inorganic materials 0.000 claims description 20
- 239000004408 titanium dioxide Substances 0.000 claims description 19
- 150000002739 metals Chemical class 0.000 claims description 14
- 239000000047 product Substances 0.000 claims description 14
- 239000002244 precipitate Substances 0.000 claims description 12
- 239000003795 chemical substances by application Substances 0.000 claims description 11
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 11
- OGIDPMRJRNCKJF-UHFFFAOYSA-N titanium oxide Inorganic materials [Ti]=O OGIDPMRJRNCKJF-UHFFFAOYSA-N 0.000 claims description 10
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 claims description 8
- UXVMQQNJUSDDNG-UHFFFAOYSA-L Calcium chloride Chemical group [Cl-].[Cl-].[Ca+2] UXVMQQNJUSDDNG-UHFFFAOYSA-L 0.000 claims description 7
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 claims description 6
- 230000001376 precipitating effect Effects 0.000 claims description 5
- AXCZMVOFGPJBDE-UHFFFAOYSA-L calcium dihydroxide Chemical compound [OH-].[OH-].[Ca+2] AXCZMVOFGPJBDE-UHFFFAOYSA-L 0.000 claims description 3
- LLZRNZOLAXHGLL-UHFFFAOYSA-J titanic acid Chemical compound O[Ti](O)(O)O LLZRNZOLAXHGLL-UHFFFAOYSA-J 0.000 claims description 3
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 claims 3
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 claims 2
- 229910001413 alkali metal ion Inorganic materials 0.000 claims 1
- WFKWXMTUELFFGS-UHFFFAOYSA-N tungsten Chemical compound [W] WFKWXMTUELFFGS-UHFFFAOYSA-N 0.000 abstract description 96
- 239000010937 tungsten Substances 0.000 abstract description 96
- LEONUFNNVUYDNQ-UHFFFAOYSA-N vanadium atom Chemical compound [V] LEONUFNNVUYDNQ-UHFFFAOYSA-N 0.000 abstract description 95
- 229910052721 tungsten Inorganic materials 0.000 abstract description 93
- 229910052720 vanadium Inorganic materials 0.000 abstract description 91
- 239000012716 precipitator Substances 0.000 abstract 2
- 229910004829 CaWO4 Inorganic materials 0.000 abstract 1
- 238000000354 decomposition reaction Methods 0.000 abstract 1
- 239000000243 solution Substances 0.000 description 37
- 239000010936 titanium Substances 0.000 description 30
- 230000006399 behavior Effects 0.000 description 18
- 229910052719 titanium Inorganic materials 0.000 description 17
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical compound [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 description 16
- 230000008569 process Effects 0.000 description 15
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 10
- 238000000926 separation method Methods 0.000 description 8
- CWYNVVGOOAEACU-UHFFFAOYSA-N Fe2+ Chemical compound [Fe+2] CWYNVVGOOAEACU-UHFFFAOYSA-N 0.000 description 7
- 230000008859 change Effects 0.000 description 7
- 238000002474 experimental method Methods 0.000 description 7
- 230000000694 effects Effects 0.000 description 6
- 229910010413 TiO 2 Inorganic materials 0.000 description 5
- 150000001875 compounds Chemical class 0.000 description 5
- 230000007423 decrease Effects 0.000 description 5
- 239000011780 sodium chloride Substances 0.000 description 5
- 239000000203 mixture Substances 0.000 description 4
- 229910021542 Vanadium(IV) oxide Inorganic materials 0.000 description 3
- 239000001110 calcium chloride Substances 0.000 description 3
- 229910001628 calcium chloride Inorganic materials 0.000 description 3
- 230000003247 decreasing effect Effects 0.000 description 3
- 238000002354 inductively-coupled plasma atomic emission spectroscopy Methods 0.000 description 3
- MWUXSHHQAYIFBG-UHFFFAOYSA-N nitrogen oxide Inorganic materials O=[N] MWUXSHHQAYIFBG-UHFFFAOYSA-N 0.000 description 3
- 238000004062 sedimentation Methods 0.000 description 3
- DZKDPOPGYFUOGI-UHFFFAOYSA-N tungsten dioxide Inorganic materials O=[W]=O DZKDPOPGYFUOGI-UHFFFAOYSA-N 0.000 description 3
- 241000196324 Embryophyta Species 0.000 description 2
- 238000002441 X-ray diffraction Methods 0.000 description 2
- HUVSVEZKLBFKTA-UHFFFAOYSA-N [Ca].[W] Chemical compound [Ca].[W] HUVSVEZKLBFKTA-UHFFFAOYSA-N 0.000 description 2
- 239000000809 air pollutant Substances 0.000 description 2
- 231100001243 air pollutant Toxicity 0.000 description 2
- 230000008021 deposition Effects 0.000 description 2
- 239000007788 liquid Substances 0.000 description 2
- 230000014759 maintenance of location Effects 0.000 description 2
- 230000004048 modification Effects 0.000 description 2
- 238000012986 modification Methods 0.000 description 2
- 239000010970 precious metal Substances 0.000 description 2
- 230000035484 reaction time Effects 0.000 description 2
- 238000007788 roughening Methods 0.000 description 2
- 239000000126 substance Substances 0.000 description 2
- RZVAJINKPMORJF-UHFFFAOYSA-N Acetaminophen Chemical compound CC(=O)NC1=CC=C(O)C=C1 RZVAJINKPMORJF-UHFFFAOYSA-N 0.000 description 1
- 229910018072 Al 2 O 3 Inorganic materials 0.000 description 1
- QTANTQQOYSUMLC-UHFFFAOYSA-O Ethidium cation Chemical compound C12=CC(N)=CC=C2C2=CC=C(N)C=C2[N+](CC)=C1C1=CC=CC=C1 QTANTQQOYSUMLC-UHFFFAOYSA-O 0.000 description 1
- CBENFWSGALASAD-UHFFFAOYSA-N Ozone Chemical compound [O-][O+]=O CBENFWSGALASAD-UHFFFAOYSA-N 0.000 description 1
- 206010036790 Productive cough Diseases 0.000 description 1
- 229910004298 SiO 2 Inorganic materials 0.000 description 1
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical compound [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 description 1
- 238000003916 acid precipitation Methods 0.000 description 1
- 150000007513 acids Chemical class 0.000 description 1
- 238000013019 agitation Methods 0.000 description 1
- 239000012670 alkaline solution Substances 0.000 description 1
- 229910052782 aluminium Inorganic materials 0.000 description 1
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 1
- 238000004458 analytical method Methods 0.000 description 1
- 239000007864 aqueous solution Substances 0.000 description 1
- 230000033228 biological regulation Effects 0.000 description 1
- 238000010531 catalytic reduction reaction Methods 0.000 description 1
- 238000010276 construction Methods 0.000 description 1
- 239000013078 crystal Substances 0.000 description 1
- 238000010790 dilution Methods 0.000 description 1
- 239000012895 dilution Substances 0.000 description 1
- 230000007613 environmental effect Effects 0.000 description 1
- 230000036571 hydration Effects 0.000 description 1
- 238000006703 hydration reaction Methods 0.000 description 1
- 230000006872 improvement Effects 0.000 description 1
- 238000009434 installation Methods 0.000 description 1
- 239000013067 intermediate product Substances 0.000 description 1
- 238000005342 ion exchange Methods 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- QGLKJKCYBOYXKC-UHFFFAOYSA-N nonaoxidotritungsten Chemical compound O=[W]1(=O)O[W](=O)(=O)O[W](=O)(=O)O1 QGLKJKCYBOYXKC-UHFFFAOYSA-N 0.000 description 1
- IYVLHQRADFNKAU-UHFFFAOYSA-N oxygen(2-);titanium(4+);hydrate Chemical compound O.[O-2].[O-2].[Ti+4] IYVLHQRADFNKAU-UHFFFAOYSA-N 0.000 description 1
- 239000002245 particle Substances 0.000 description 1
- 239000005297 pyrex Substances 0.000 description 1
- 230000036632 reaction speed Effects 0.000 description 1
- 230000008929 regeneration Effects 0.000 description 1
- 238000011069 regeneration method Methods 0.000 description 1
- 229910052710 silicon Inorganic materials 0.000 description 1
- 239000010703 silicon Substances 0.000 description 1
- GROMGGTZECPEKN-UHFFFAOYSA-N sodium metatitanate Chemical compound [Na+].[Na+].[O-][Ti](=O)O[Ti](=O)O[Ti]([O-])=O GROMGGTZECPEKN-UHFFFAOYSA-N 0.000 description 1
- 238000000638 solvent extraction Methods 0.000 description 1
- 210000003802 sputum Anatomy 0.000 description 1
- 208000024794 sputum Diseases 0.000 description 1
- 229910001930 tungsten oxide Inorganic materials 0.000 description 1
- 229910001935 vanadium oxide Inorganic materials 0.000 description 1
- 238000010792 warming Methods 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22B—PRODUCTION AND REFINING OF METALS; PRETREATMENT OF RAW MATERIALS
- C22B34/00—Obtaining refractory metals
- C22B34/30—Obtaining chromium, molybdenum or tungsten
- C22B34/36—Obtaining tungsten
- C22B34/365—Obtaining tungsten from spent catalysts
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22B—PRODUCTION AND REFINING OF METALS; PRETREATMENT OF RAW MATERIALS
- C22B1/00—Preliminary treatment of ores or scrap
- C22B1/02—Roasting processes
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22B—PRODUCTION AND REFINING OF METALS; PRETREATMENT OF RAW MATERIALS
- C22B1/00—Preliminary treatment of ores or scrap
- C22B1/14—Agglomerating; Briquetting; Binding; Granulating
- C22B1/16—Sintering; Agglomerating
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22B—PRODUCTION AND REFINING OF METALS; PRETREATMENT OF RAW MATERIALS
- C22B3/00—Extraction of metal compounds from ores or concentrates by wet processes
- C22B3/04—Extraction of metal compounds from ores or concentrates by wet processes by leaching
- C22B3/12—Extraction of metal compounds from ores or concentrates by wet processes by leaching in inorganic alkaline solutions
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22B—PRODUCTION AND REFINING OF METALS; PRETREATMENT OF RAW MATERIALS
- C22B3/00—Extraction of metal compounds from ores or concentrates by wet processes
- C22B3/20—Treatment or purification of solutions, e.g. obtained by leaching
- C22B3/205—Treatment or purification of solutions, e.g. obtained by leaching using adducts or inclusion complexes
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22B—PRODUCTION AND REFINING OF METALS; PRETREATMENT OF RAW MATERIALS
- C22B34/00—Obtaining refractory metals
- C22B34/10—Obtaining titanium, zirconium or hafnium
- C22B34/12—Obtaining titanium or titanium compounds from ores or scrap by metallurgical processing; preparation of titanium compounds from other titanium compounds see C01G23/00 - C01G23/08
- C22B34/1236—Obtaining titanium or titanium compounds from ores or scrap by metallurgical processing; preparation of titanium compounds from other titanium compounds see C01G23/00 - C01G23/08 obtaining titanium or titanium compounds from ores or scrap by wet processes, e.g. by leaching
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22B—PRODUCTION AND REFINING OF METALS; PRETREATMENT OF RAW MATERIALS
- C22B34/00—Obtaining refractory metals
- C22B34/10—Obtaining titanium, zirconium or hafnium
- C22B34/12—Obtaining titanium or titanium compounds from ores or scrap by metallurgical processing; preparation of titanium compounds from other titanium compounds see C01G23/00 - C01G23/08
- C22B34/1236—Obtaining titanium or titanium compounds from ores or scrap by metallurgical processing; preparation of titanium compounds from other titanium compounds see C01G23/00 - C01G23/08 obtaining titanium or titanium compounds from ores or scrap by wet processes, e.g. by leaching
- C22B34/124—Obtaining titanium or titanium compounds from ores or scrap by metallurgical processing; preparation of titanium compounds from other titanium compounds see C01G23/00 - C01G23/08 obtaining titanium or titanium compounds from ores or scrap by wet processes, e.g. by leaching using acidic solutions or liquors
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22B—PRODUCTION AND REFINING OF METALS; PRETREATMENT OF RAW MATERIALS
- C22B34/00—Obtaining refractory metals
- C22B34/20—Obtaining niobium, tantalum or vanadium
- C22B34/22—Obtaining vanadium
- C22B34/225—Obtaining vanadium from spent catalysts
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22B—PRODUCTION AND REFINING OF METALS; PRETREATMENT OF RAW MATERIALS
- C22B7/00—Working up raw materials other than ores, e.g. scrap, to produce non-ferrous metals and compounds thereof; Methods of a general interest or applied to the winning of more than two metals
- C22B7/006—Wet processes
- C22B7/008—Wet processes by an alkaline or ammoniacal leaching
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22B—PRODUCTION AND REFINING OF METALS; PRETREATMENT OF RAW MATERIALS
- C22B7/00—Working up raw materials other than ores, e.g. scrap, to produce non-ferrous metals and compounds thereof; Methods of a general interest or applied to the winning of more than two metals
- C22B7/009—General processes for recovering metals or metallic compounds from spent catalysts
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02P—CLIMATE CHANGE MITIGATION TECHNOLOGIES IN THE PRODUCTION OR PROCESSING OF GOODS
- Y02P10/00—Technologies related to metal processing
- Y02P10/20—Recycling
Definitions
- the present invention relates to a method for recovering valuable metals capable of selectively recovering vanadium, tungsten and titanium from a waste denitration catalyst.
- SCR Selective catalytic reduction
- the denitration catalyst is generated as waste after 3-4 years of use and 2-3 times of regeneration, and the opportunity cost of the use of the valuable metal in the catalyst increases when the whole waste is discarded.
- the waste denitration catalyst contains vanadium in an amount of 1 to 1.5% (w / w) and tungsten in an amount of 10% (w / w).
- there are various valuable metals such as titanium, aluminum and silicon.
- vanadium and tungsten can be recovered in the form of oxides, but energy efficiency due to an increase in roasting time is low, and there is no means for separating and recovering titanium dioxide separately, There is a problem that can not be recovered.
- the present invention provides a method of separating titanium dioxide, which is difficult to be recovered by conventional acid and alkali leaching methods, in the recovery process of babidium and tungsten.
- the present invention provides a method for producing a waste denitration catalyst, comprising the steps of: (a) adding an alkaline metal to a waste denitration catalyst and mixing and alkali fusion to produce waste products; (b) recovering the alkali leaching solution and the residue by water leaching of the roasting product; (c) adding a precipitant to the alkali leach solution and stirring the precipitate to precipitate with calcium metavanadate, Ca (VO 3 ) 2, or calcium tungstate (CaWO 4 ); And (d) acid decomposing the recovered calcium tungstate to produce tungstic acid.
- the present invention also provides a method for recovering a selected valuable metal from a waste denitration catalyst through alkaline fusion.
- the alkali metal may be added in an amount of 1.1 to 2 equivalents based on 1 equivalent of the waste denitration catalyst.
- the alkali metal may also be sodium carbonate (Na 2 CO 3 ) or sodium hydroxide (NaOH).
- the alkali fusion can be roasted at an output of 1 kw per 100 g of the waste denitration catalyst when microwaves are used.
- the alkali fusion can be roasted at 900 to 1000 DEG C for 20 to 60 minutes.
- the precipitation agent may be a calcium chloride (CaCl 2).
- the pH can be adjusted by adding the precipitation agent and further adding a strong alkali calcium hydroxide [Ca (OH) 2 ].
- stirring after the addition of the precipitant may be carried out at 200 to 600 rpm for 30 minutes to 2 hours.
- the precipitation agent is added so that 1.0 to 1.5 equivalents of calcium is added to the precipitate to adjust the precipitation efficiency of calcium metavanadate to 99 % Or more.
- the precipitation agent is added and reacted so that 1.0 equivalent or less of calcium is added to precipitate the calcium tungstate precipitation efficiency to 1% .
- a precipitant is added to the alkali leach solution and the mixture is reacted with stirring at 333 K to 353 K for 15 minutes or less to precipitate calcium metavanadate and react at 313 K to 353 K for 1 hour to 2 hours to form tungsten Calcium can be precipitated.
- the present invention also relates to a process for recovering the residue, comprising the steps of: (i) acid leaching by adding sulfuric acid or hydrochloric acid to the residue; (ii) adding sodium hydroxide to the acid leaching solution and allowing the reaction to precipitate with meta titanic acid, TiO (OH) 2 ; And (iii) calcining the metatitanic acid to recover titanium dioxide.
- the acid leaching can be carried out at 323 K to 333 K for 2 hours to 3 hours.
- the pH may be adjusted to 2 or less so that the leaching rate may be 99% or more.
- the calcination may be carried out at 850 K to 900 K for 4 to 5 hours.
- vanadium and tungsten can be recovered with high efficiency by adding an excess amount of alkali metal to the waste denitration catalyst, roasting, adding a precipitant of the leached solution which is water leached, and controlling the reaction rate.
- titanium dioxide which has not been recovered effectively by the acid and alkali leaching methods by the conventional soda roasting can be additionally separated and recovered.
- the recovery of vanadium, tungsten, and titanium dioxide can be selectively determined for each valuable metal.
- reaction time in the alkali fusion and precipitation steps can be greatly shortened, which greatly increases the efficiency of the entire valuable metal recovery process.
- FIG. 1 illustrates a process flow of a selective ferrous metal recovery process from a waste denitration catalyst through an alkali fusion according to an embodiment of the present invention.
- FIG. 2 shows a process flow of a selective precious metal recovery method from a waste denitration catalyst through alkali fusion according to another embodiment of the present invention.
- FIG. 4 is a graph showing leaching rates of vanadium and tungsten by roasting temperatures during alkaline fusion for 20 minutes in a selective metal recovery method from a waste denitration catalyst through alkaline fusion according to an embodiment of the present invention.
- FIG. 5 is a graph showing the leaching rates of vanadium and tungsten by the roasting temperature during alkali fusion for 60 minutes in the selective metal recovery method from the waste denitration catalyst through alkaline fusion according to an embodiment of the present invention.
- FIG. 6 is a graph showing leaching rates of vanadium and tungsten according to the equivalents of sodium hydroxide during alkaline fusion in a selective lean metal recovery method from a waste denitration catalyst through alkaline fusion according to an embodiment of the present invention.
- FIG. 7 is a photograph of roughening a selective metal free metal from a waste denitration catalyst through alkaline fusion according to an embodiment of the present invention by roasting using sodium carbonate during alkali fusion.
- FIG. 8 is a graph showing leaching rates of vanadium and tungsten according to roasting time by adding sodium carbonate during alkali fusion in a selective metal recovery method from a waste denitration catalyst through alkaline fusion according to an embodiment of the present invention.
- FIG. 9 is a graph showing leaching rates of vanadium and tungsten according to the equivalents of sodium carbonate during alkaline fusion in a selective lean metal recovery method from a waste denitration catalyst through alkaline fusion according to an embodiment of the present invention.
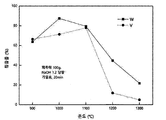
- FIG. 10 is a graph showing leaching rates of vanadium, tungsten, and titanium according to roasting temperature during alkaline fusion in a selective metal recovery method from a waste denitration catalyst through alkaline fusion according to an embodiment of the present invention.
- Fig. 11 is a graph showing the change in temperature at the roasting time through the microwave.
- FIG. 12 is a graph showing leaching rates of vanadium and tungsten according to microwave irradiation time in the case where sodium hydroxide is added in the selective ferrous metal recovery method from a waste denitration catalyst through alkaline fusion according to an embodiment of the present invention .
- FIG. 13 is a graph showing changes in the leaching rate depending on the equivalence of sodium hydroxide during the microwave irradiation in the selective metal recovery method from the waste denitration catalyst through alkaline fusion according to an embodiment of the present invention.
- Figure 14 shows the leach rates of vanadium and tungsten for each microwave output.
- 15 is a graph showing changes in leaching rate depending on equivalents of sodium hydroxide during microwave irradiation in a selective metal recovery method from a waste denitration catalyst through alkaline fusion according to an embodiment of the present invention.
- 16 is a graph showing changes in leaching rates of vanadium and tungsten by equivalents of sodium carbonate in a selective metal recovery method from a waste denitration catalyst through alkaline fusion according to an embodiment of the present invention.
- 17 is a graph showing the sedimentation behavior of vanadium according to the variation of the stirring speed in the selective metal recovery method from the waste denitration catalyst through alkaline fusion according to an embodiment of the present invention.
- 19 is a graph showing deposition rates of vanadium depending on sodium hydroxide concentration and calcium addition amount in a selective metal recovery method from a waste denitration catalyst through alkaline fusion according to an embodiment of the present invention.
- 20 is a graph showing the precipitation rate of tungsten depending on the concentration of sodium hydroxide and the amount of calcium added in the selective metal recovery method from a waste denitration catalyst through alkaline fusion according to an embodiment of the present invention.
- FIG. 21 is a graph showing the precipitation behavior of vanadium and tungsten according to the concentration of sodium hydroxide in the selective ferrous metal recovery method from a waste denitration catalyst through alkaline fusion according to an embodiment of the present invention.
- FIG. 22 is a graph showing the precipitation behavior of vanadium according to temperature in the selective metal recovery method from a waste denitration catalyst through alkaline fusion according to an embodiment of the present invention.
- FIG. 23 is a graph showing precipitation behavior of tungsten depending on temperature in a selective metal recovery method from a waste denitration catalyst through alkaline fusion according to an embodiment of the present invention.
- 24 is a graph showing precipitation behavior of calcium, vanadium and tungsten according to retention time at room temperature in a selective metal recovery method from a waste denitration catalyst through alkaline fusion according to an embodiment of the present invention.
- FIG. 1 illustrates a process flow of a selective ferrous metal recovery process from a waste denitration catalyst through an alkali fusion according to an embodiment of the present invention.
- a method for recovering selective metals from a waste denitration catalyst through alkaline fusion comprises the steps of: (a) adding an alkali metal to a waste denitration catalyst to produce an exhaust product by alkali fusion; (b) recovering the alkali leaching solution and the residue by water leaching of the roasting product; (c) adding a precipitant to the alkali leach solution and stirring the precipitate by precipitating with calcium metavanadate, Ca (VO 3 ) 2, or calcium tungstate (CaWO 4 ); And (d) acid-decomposing the recovered calcium tungstate to produce tungstic acid.
- FIG. 1 illustrates a process flow of a selective ferrous metal recovery process from a waste denitration catalyst through an alkali fusion according to an embodiment of the present invention.
- a method for recovering selective metals from a waste denitration catalyst through alkaline fusion comprises the steps of: (a) adding an alkali metal to a waste denitration catalyst to produce an exhaust product by alkali fusion; (b) recovering the alkali leaching solution and the residue by water leaching of the roasting product; (c) adding a precipitant to the alkali leach solution and stirring the precipitate to precipitate with calcium metavanadate, Ca (VO 3 ) 2, or calcium tungstate (CaWO 4 ); And (d) acid-decomposing the recovered calcium tungstate to produce tungstic acid.
- a waste denitration catalyst is prepared, alkali metal is added and mixed, and an exhaust product is produced through alkali fusion (S100).
- the waste denitration catalyst is crushed and dried in an oven to remove moisture, and it can be prepared to have a size of 100 ⁇ m or less.
- the waste denitration catalyst contains vanadium (V) and tungsten (W) in the form of oxides (V 2 O 5 , WO 3 ) and may contain a large amount of titanium dioxide (TiO 2 ).
- the alkaline fusion is roughened together with alkali metal which is a strong alkali.
- alkali metal which is a strong alkali.
- the amount of alkaline metal added to the amount equivalent to 2 equivalents of the amount equivalent to the amount of waste denitration catalyst is added and reacted to change insoluble compounds such as vanadium and tungsten oxide into water-soluble compounds, which means that the electric power used in roasting is carefully controlled by using an electric furnace or a microwave to greatly increase the energy efficiency of the roasting process.
- the alkali metal may be sodium carbonate (Na 2 CO 3 ) or sodium hydroxide (NaOH).
- the alkali metal is a strong alkali in an aqueous solution, and depending on the selection of the alkali metal, the conditions of alkali fusion, that is, the alkali metal addition equivalent, the roasting temperature and the roasting time may be different.
- the alkali fusion may produce an endosperm product according to the following Reaction Scheme 1 or Reaction Scheme 2.
- the insoluble oxides V 2 O 5 and WO 3 are changed to water-soluble compounds NaVO 3 and Na 2 WO 4 .
- the alkali metal may be added in an amount of 1.1 to 2 equivalents based on 1 equivalent of the waste denitration catalyst.
- the tungsten leaching rate when the alkali metal is sodium hydroxide (NaOH), the tungsten leaching rate is 1.2 equivalents relative to the waste denitration catalyst, 87.5% when roasted at 1000 ° C for 20 minutes and roasted at 900 ° C for 60 minutes , The tungsten leaching rate can be maximized to 91.3%.
- NaOH sodium hydroxide
- the alkali metal is sodium carbonate (Na 2 CO 3 ), it is added in an amount of 1.2 equivalents, and it can be roasted at 1000 ° C. for 20 minutes to achieve a tungsten leaching rate of 99.8%.
- the alkali fusion may be roasted using an electric furnace or a microwave.
- microwave When microwave is used, it is possible to efficiently determine the leaching rate of tungsten and vanadium by precisely controlling the power supply according to the equivalence.
- microwave When microwave is used, it can be roasted with an output of 1 kw per 100 g of waste denitration catalyst.
- tungsten when 1.2 g of sodium hydroxide is added to 300 g of waste denitration catalyst at an output of 3 kw and roasted for 20 minutes, tungsten may exhibit a leaching rate of 100% and 2 equivalents of sodium hydroxide And the leaching rate of tungsten, vanadium and titanium dioxide can be maximized when roasting for 60 minutes.
- 1.2 g of sodium carbonate is added for roasting at an output of 3 kw with respect to 300 g of waste denitration catalyst, and the leaching rate of tungsten and vanadium can be maximized by roasting for 40 minutes. 2.0 eq. The leaching rate of tungsten, vanadium and titanium dioxide can be maximized when roasting.
- the alkali fusion can be roasted at 900 to 1000 DEG C for 20 to 60 minutes.
- the leaching rate (W / V) of leached tungsten and vanadium can be effectively determined
- the leach rate of titanium dioxide can be determined, so that the valuable metal can be selectively recovered.
- the sprouts are water-leached to recover the alkali leaching solution and residue (S200).
- the sprouts are those in which the insoluble compound is changed into a water-soluble compound.
- the alkali leachate may contain water-soluble NaVO 3 and Na 2 WO 4 and the residues may contain sodium titanate, Na 2 TiO 3 or Na 2 Ti 3 O 7 .
- the alkali leaching solution can be precipitated to recover tungsten and vanadium, and when the residue is acid leached, a hydrated titanium dioxide intermediate can be produced.
- the valuable metal can be selectively separated through the precipitation process.
- the precipitation agent is added to the alkali leach solution, and the precipitate is precipitated with calcium metavanadate (Ca (VO 3 ) 2 ] or calcium tungstate (CaWO 4 ) by stirring (S300).
- Ca (VO 3 ) 2 calcium metavanadate
- CaWO 4 calcium tungstate
- the precipitation agent may be a calcium chloride (CaCl 2).
- the calcium chloride can control the leaching rate of tungsten and vanadium through the calcium in the leach solution which is strong alkaline, thereby enabling the separation of the two metals.
- the precipitant may be added and the pH may be adjusted by adding a strong alkali calcium hydroxide [Ca (OH) 2 ].
- calcium metavanadate can be precipitated by adjusting the pH to 13.25, and calcium tungstate can be precipitated at pH 13 or lower.
- Stirring after addition of the precipitant may be carried out at 200 to 600 rpm for 30 minutes to 2 hours.
- the alkali metal is sodium hydroxide and the concentration of sodium hydroxide in the alkaline leaching solution is 0.5 to 0.75 mol / L, 1.0 to 1.5 equivalent of calcium is added to the precipitant so that meta vanadium
- the precipitation efficiency of calcium can be 99% or more.
- the alkali metal is sodium hydroxide and the concentration of sodium hydroxide in the alkali leach solution is 0.5 mol / L or less
- a precipitant is added so that 1.0 equivalent or less of calcium is added and reacted to obtain a calcium tungstate recovery Can be precipitated to 1% or less.
- the precipitation efficiency of tungsten and vanadium can be determined according to the amount of calcium in the precipitant calcium chloride and the concentration of sodium hydroxide in the alkali leaching solution, thereby enabling selective recovery.
- a precipitant is added to the alkali leach solution, and the mixture is reacted with stirring at 333 K to 353 K for 15 minutes or less to precipitate calcium metavanadate and react at 313 K to 353 K for 1 hour to 2 hours to form tungsten Calcium can be precipitated.
- vanadium and tungsten can be separated even at room temperature.
- FIG. 2 shows a process flow of a selective precious metal recovery method from a waste denitration catalyst through alkali fusion according to another embodiment of the present invention.
- an alternative method for recovering valuable metals from a waste denitration catalyst through alkaline fusion comprises recovering the residue, (i) adding sulfuric acid or hydrochloric acid to the residue, ; (ii) adding sodium hydroxide to the acid leaching solution and allowing the reaction to precipitate with meta titanic acid, TiO (OH) 2 ; And (iii) calcining the metatitanic acid to recover titanium dioxide.
- the residue is an intermediate product produced through alkali fusion and contains a large amount of titanium dioxide.
- soda roasting can deposit vanadium and tungsten after roasting, but there is a problem that titanium dioxide can not be selected and separated.
- the residue may be acid-leached again to selectively recover titanium dioxide of high purity.
- the acid leaching can be carried out at 323 K to 333 K for 2 to 3 hours.
- the acid leaching can be carried out according to the following reaction formula (4).
- Sodium hydroxide may be added to the acid leach solution and allowed to react and precipitate with metatitanic acid.
- the precipitation process can be carried out according to the following reaction formula (5).
- TiO 2 ( Aq. ) + 2 NaOH ( Aq. ) TiO (OH) 2 ( s ) + 2 NaCl ( Aq. )
- TiOSO 4 (Aq.) + 2NaOH (Aq.) TiO (OH) 2 (s) + 2Na 2 SO 3 (Aq.)
- metatitanic acid is in the hydrate form of titanium dioxide.
- the pH may be adjusted to 2 or less so that the leaching rate may be 99% or more.
- Precipitation can be easily performed in the range of strong acids, and in particular, when the pH is adjusted to 2 or lower, the leaching rate of the titanium dioxide hydrate may be 99% or more.
- the metatitanic acid may then be calcined to recover the titanium dioxide.
- the calcination may be performed at 900 K or more for 5 hours or more, and in this case, the purity of recovered titanium dioxide is recovered to 90% or more.
- a waste denitration catalyst was prepared, pulverized and analyzed by ICP-AES.
- Vanadium, tungsten and titanium in oxide form were identified and roasting conditions for alkali fusion were derived.
- the microwave power was adjusted to 3 kw, and alkali fusion was performed for 20 minutes for 1.2 equivalents and 60 minutes for 2 equivalents.
- the alkali fusion temperature was performed in the range of 900 to 1000 ⁇ ⁇ .
- the alkali leaching solution and the residue were recovered by adjusting the pulp density to 20% after alkaline fusion and water leaching for 2 hours.
- the average size of the particles was adjusted to 200 ⁇ m or less and was performed at room temperature.
- the calcium addition amount of calcium chloride (CaCl 2 ) as a precipitating agent is added in an amount of 1.0 to 1.5 equivalents, .
- composition was first analyzed using ICP-AES.
- Fig. 3 shows the results of X-ray diffraction analysis of the waste denitration catalyst.
- Table 1 shows the composition of a waste denitration catalyst.
- the amount of alkali added was calculated in accordance with the above-mentioned Reaction Scheme 2.
- FIG. 4 is a graph showing leaching rates of vanadium and tungsten by roasting temperatures during alkaline fusion for 20 minutes in a selective metal recovery method from a waste denitration catalyst through alkaline fusion according to an embodiment of the present invention.
- FIG. 5 is a graph showing the leaching rates of vanadium and tungsten by the roasting temperature during alkali fusion for 60 minutes in the selective metal recovery method from the waste denitration catalyst through alkaline fusion according to an embodiment of the present invention.
- Table 2 shows the leaching rates of vanadium and tungsten by roasting in an electric furnace for each hourly temperature with the alkali being selected as sodium hydroxide, the amount of NaOH being set at 1.2 equivalent to the denitration catalyst.
- FIG. 6 is a graph showing leaching rates of vanadium and tungsten according to the equivalents of sodium hydroxide during alkaline fusion in a selective lean metal recovery method from a waste denitration catalyst through alkaline fusion according to an embodiment of the present invention.
- the leaching rate of tungsten was 85%, which was relatively low at 2 equivalents, but the leaching rate was 91% or more at 1.2 and 1.5 equivalents. It was also confirmed that the amount of NaOH required to leach vanadium and tungsten from the waste denitration catalyst was sufficient to be 1.5 equivalents or more when the calcination temperature was 900 ° C for 1 hour.
- the roasting conditions of alkaline fusion were 1.2 equivalents when alkali metal was sodium hydroxide, and it was confirmed that W / V leaching rate was the highest at 1000 ° C. when the roasting time was 20 minutes and 900 ° C. when the roasting time was 60 minutes.
- Table 3 shows roasting results of sodium carbonate.
- FIG. 7 is a photograph of roughening a selective metal free metal from a waste denitration catalyst through alkaline fusion according to an embodiment of the present invention by roasting using sodium carbonate during alkali fusion.
- FIG. 8 is a graph showing leaching rates of vanadium and tungsten according to roasting time by adding sodium carbonate during alkali fusion in a selective metal recovery method from a waste denitration catalyst through alkaline fusion according to an embodiment of the present invention.
- FIG. 9 is a graph showing leaching rates of vanadium and tungsten according to the equivalents of sodium carbonate during alkaline fusion in a selective lean metal recovery method from a waste denitration catalyst through alkaline fusion according to an embodiment of the present invention.
- the roasting conditions of the alkaline fusion step for the recovery of titanium (Ti) were confirmed by adjusting the amount of alkali required for leaching titanium in the waste denitration catalyst to 2 equivalents.
- the amount of alkali metal to be added was 2 equivalents according to Reaction Scheme 7 above.
- TiO 2 is formed in the form of Na 2 TiO 3 or Na 2 Ti 3 O 7 , and this form is not readily leached into the acid.
- the amount of alkali was changed to 2 equivalents based on Na 2 Ti 3 O 7 in order to convert the titanium into Na 8 Ti 5 O 14 form which is all leached into the acid.
- FIG. 10 is a graph showing leaching rates of vanadium, tungsten, and titanium according to roasting temperature during alkaline fusion in a selective metal recovery method from a waste denitration catalyst through alkaline fusion according to an embodiment of the present invention.
- the leaching rate of Ti was 95% when roasted at 950 ° C. for 1 hour with NaOH, and the leaching rate of Ti was 93% when roasted at 1000 ° C. for 1 hour with Na 2 CO 3 And W / V was also leached at least 98% at this time.
- the amount of alkali is 2 equivalents based on the reaction of Scheme 7, and the case of roasting for 1 hour at a temperature of 950 ° C. or more is the optimum condition Respectively.
- Fig. 11 is a graph showing the change in temperature at the roasting time through the microwave.
- 300 g of the waste denitration catalyst was subjected to 1.2 hour equivalent of NaOH and a microwave power of 3 kw, and the temperature was checked over time. After about 5 minutes, the temperature was maintained at 25 ° C Respectively.
- FIG. 12 is a graph showing leaching rates of vanadium and tungsten according to microwave irradiation time in the case where sodium hydroxide is added in the selective ferrous metal recovery method from a waste denitration catalyst through alkaline fusion according to an embodiment of the present invention .
- the leaching rate of tungsten is close to 100% And more than 85% of vanadium was leached.
- FIG. 13 is a graph showing changes in the leaching rate depending on the equivalence of sodium hydroxide during the microwave irradiation in the selective metal recovery method from the waste denitration catalyst through alkaline fusion according to an embodiment of the present invention.
- the leaching rate was similar to that of the leaching rate per hour. However, at 2 equivalents of NaOH, more than 99% of W / V were leached out.
- Figure 14 shows the leach rates of vanadium and tungsten for each microwave output.
- 3 kw was the result of decreasing the W / V leaching rate with a longer time of microwave irradiation, and the W / V leaching rate of 2 kw was higher. It is considered that the temperature was not high enough to affect the decrease of the leaching rate even if the microwave was irradiated for 1 hour because the output was lower than that of the same amount.
- 15 is a graph showing changes in leaching rate depending on equivalents of sodium hydroxide during microwave irradiation in a selective metal recovery method from a waste denitration catalyst through alkaline fusion according to an embodiment of the present invention.
- 16 is a graph showing changes in leaching rates of vanadium and tungsten by equivalents of sodium carbonate in a selective metal recovery method from a waste denitration catalyst through alkaline fusion according to an embodiment of the present invention.
- the optimum leaching conditions for each metal were determined by confirming that leaching can be selectively performed according to alkali equivalent, roasting temperature, roasting time and microwave power.
- the alkaline leaching solution of denitrification catalyst is a strong alkaline solution of pH 12 or more. Since the separation of two metals by calcium is possible, the precipitation behavior of vanadium and tungsten in the strong alkaline region is clarified and the precipitation of vanadium and tungsten by calcium addition .
- the simulated solution was prepared by dissolving NaVO 3 and NaWO 4 ⁇ 2H 2 O in NaOH solution and using 1 g / L of vanadium and 10 g / L of vanadium and tungsten in the waste denitration catalyst based on vanadium and tungsten contents.
- CaCl 2 was used as a precipitant and 4 mol / L was added to prevent the dilution of vanadium and tungsten due to a large amount of input.
- 1 equivalent of calcium participating in the precipitation reaction of vanadium 1 g / L and tungsten 10 g / L corresponds to 2.57 g / L, and the amount of calcium added is determined by the amount of calcium participating in the precipitation of vanadium and tungsten by the following reaction formula And calculated as 1 equivalent.
- the experimental apparatus used was a constant temperature circulating water tank and Pyrex double jacket reaction tank, and 2.5 ⁇ 2.5 mm cruciform magnetic stir bar was directly stirred.
- the precipitation behaviors of vanadium and tungsten were investigated for 2 hours by adjusting Ca (Ca) addition, NaOH solution concentration, stirring speed and reaction temperature.
- 17 is a graph showing the sedimentation behavior of vanadium according to the variation of the stirring speed in the selective metal recovery method from the waste denitration catalyst through alkaline fusion according to an embodiment of the present invention.
- 19 is a graph showing deposition rates of vanadium depending on sodium hydroxide concentration and calcium addition amount in a selective metal recovery method from a waste denitration catalyst through alkaline fusion according to an embodiment of the present invention.
- the leaching rate of vanadium was checked according to the concentration of NaOH solution and the amount of Ca, the leaching rate was 80% or more in the experimental conditions. Among them, 0.50 - 0.75 mol / L NaOH solution showed a leaching rate of 99% or more at 1.0-1.5 equivalent of Ca addition condition.
- 20 is a graph showing the precipitation rate of tungsten depending on the concentration of sodium hydroxide and the amount of calcium added in the selective metal recovery method from a waste denitration catalyst through alkaline fusion according to an embodiment of the present invention.
- leaching rate tends to decrease as the NaOH concentration increases and the amount of Ca added decreases. Particularly, leaching rate of less than 1% was shown in the case of Ca addition of 1.0 equivalent or less in NaOH solution of 0.5 mol / L or more.
- FIG. 21 is a graph showing the precipitation behavior of vanadium and tungsten according to the concentration of sodium hydroxide in the selective ferrous metal recovery method from a waste denitration catalyst through alkaline fusion according to an embodiment of the present invention.
- the conditions for the separation of vanadium and tungsten can be confirmed by overlapping the precipitation region of not less than 95% of vanadium and not more than 10% of tungsten.
- A is more than 99% of vanadium and less than 1% of tungsten
- a + B is more than 97% of vanadium, less than 1% of tungsten
- a + B + C is less than 1% of vanadium and less than 1% of tungsten. Area.
- vanadium and tungsten can be separated and recovered according to the concentration of sodium hydroxide.
- FIG. 22 is a graph showing the precipitation behavior of vanadium according to temperature in the selective metal recovery method from a waste denitration catalyst through alkaline fusion according to an embodiment of the present invention.
- the precipitation reaction was completed within 45 minutes at 283-313 K, and the reaction was completed within 15 minutes at 333 K or above.
- FIG. 23 is a graph showing precipitation behavior of tungsten depending on temperature in a selective metal recovery method from a waste denitration catalyst through alkaline fusion according to an embodiment of the present invention.
- the precipitation reaction did not occur at a temperature below room temperature (293 K), but at a temperature of 313 K or higher, the precipitation reaction started to take place after the reaction time, and the leaching rate was 10.0% after the completion of the reaction for 2 hours .
- the precipitate started to precipitate with vanadium, and the leaching rate was 98.5% at 333 K and 97.2% at 353 K after the completion of the reaction.
- vanadium and tungsten can be separated at room temperature due to the difference in precipitation reaction speed.
- tungsten is not precipitated as CaWO 4 or less in the room temperature (293 K), it was confirmed that the temperature is settled to the higher CaWO 4.
- 24 is a graph showing precipitation behavior of calcium, vanadium and tungsten according to retention time at room temperature in a selective metal recovery method from a waste denitration catalyst through alkaline fusion according to an embodiment of the present invention.
- Tungsten is not only slower than vanadium but also has a slower precipitation due to the reaction of Ca (OH) 2 with the reaction of Ca (OH) 2 by the solid-liquid reaction, unlike the liquid-liquid reaction of vanadium.
- vanadium was precipitated with Ca (VO 3 ) 2 at room temperature by the precipitation reaction rate of vanadium and tungsten, and tungsten was left in solution,
Abstract
Description
| TiO2 | SiO2 | WO3 | Al2O3 | CaO | V2O5 | Fe2O3 | MgO | MoO3 | 기타 (wt.%) |
| 70.9 | 9.80 | 7.73 | 5.57 | 2.50 | 1.23 | 0.77 | 0.55 | 0.10 | 4.5 |
| 20분 | 60분 | 비고 | |||
| 온도 (℃) | W | V | W | V | |
| 900 | 63.8 | 66.4 | 91.3 | 68.0 | 알칼리 투입량1:1.2 |
| 1000 | 87.5 | 71.3 | 81.4 | 67.4 | |
| 1100 | 79.5 | 77.9 | 43.0 | 13.4 | |
| 1200 | 44.5 | 11.8 | 34.9 | 5.7 | |
| 1300 | 21.6 | 4.9 | - | - | |
| 1 | 2 | 3 | 4 | 비 고 | |
| 당량 | 1.2 | 2 | 1.2 | 2 | |
| 시간(분) | 20 | 20 | 60 | 60 | |
| W | 99.8 | 98.6 | 99.2 | 98.5 | 침출률,% |
| V | 99.6 | 99.4 | 98.6 | 99.1 |
Claims (15)
- (a) 폐탈질촉매에 알칼리금속을 첨가하고 혼합하여 알칼리 퓨전하여 배소산물을 생성하는 단계;(b) 상기 배소산물을 수침출하여 알칼리 침출액 및 잔류물을 회수하는 단계;(c) 상기 알칼리 침출액에 침전제를 첨가하고 교반하여 메타바나듐산 칼슘[calcium metavanadate, Ca(VO3)2] 또는 텅스텐산 칼슘(calcium tungsnate, CaWO4)으로 침전시켜 회수하는 단계; 및(d) 회수된 상기 텅스텐산 칼슘을 산분해하여 텅스텐산을 제조하는 단계를 포함하는 알칼리 퓨전을 통한 폐탈질촉매로부터 선택적 유가금속 회수방법.
- 제1항에 있어서,상기 알칼리금속은상기 폐탈질촉매 1 당량에 대하여 1.1 내지 2 당량으로 첨가하는 것을 특징으로 하는 알칼리 퓨전을 통한 폐탈질촉매로부터 선택적 유가금속 회수방법.
- 제1항에 있어서,상기 알칼리금속은탄산나트륨(Na2CO3) 또는 수산화나트륨(NaOH)인 것을 특징으로 하는 알칼리 퓨전을 통한 폐탈질촉매로부터 선택적 유가금속 회수방법.
- 제1항에 있어서,상기 알칼리 퓨전은마이크로웨이브를 사용하는 경우에 폐탈질촉매 100 g 당 1 kw의 출력으로 배소하는 것을 특징으로 하는 알칼리 퓨전을 통한 폐탈질촉매로부터 선택적 유가금속 회수방법.
- 제1항에 있어서,상기 알칼리 퓨전은 900 내지 1000 ℃에서 20 내지 60분 동안 배소하는 것을 특징으로 하는 알칼리 퓨전을 통한 폐탈질촉매로부터 선택적 유가금속 회수방법.
- 제1항에 있어서,상기 침전제는 염화칼슘(CaCl2)인 것을 특징으로 하는 알칼리 퓨전을 통한 폐탈질촉매로부터 선택적 유가금속 회수방법.
- 제1항에 있어서,상기 침전제를 첨가하고 강알칼리인 수산화칼슘[Ca(OH)2]을 더 첨가하여 pH를 조절하는 것을 특징으로 하는 알칼리 퓨전을 통한 폐탈질촉매로부터 선택적 유가금속 회수방법.
- 제1항에 있어서,상기 침전제를 첨가한 이후의 교반은200 내지 600 rpm으로 30분 내지 2시간 동안 수행되는 것을 특징으로 하는 알칼리 퓨전을 통한 폐탈질촉매로부터 선택적 유가금속 회수방법.
- 제1항 또는 제6항에 있어서,상기 알칼리금속이 수산화나트륨이고, 알칼리 침출액의 수산화나트륨의 농도가 0.5 내지 0.75 mol/L 인 경우에 1.0내지 1.5 당량의 칼슘이 첨가되도록 침전제를 투입하여 반응시켜 메타바나듐산 칼슘의 침전효율을 99% 이상으로 침전시키는 것을 특징으로 하는 알칼리 퓨전을 통한 폐탈질촉매로부터 선택적 유가금속 회수방법.
- 제1항 또는 제6항에 있어서,상기 알칼리금속이 수산화나트륨이고, 알칼리 침출액의 수산화나트륨의 농도가 0.5 mol/L 이하인 경우에 1.0 당량 이하의 칼슘이 첨가되도록 침전제를 투입하고 반응시켜 텅스텐산 칼슘의 회수율을 1% 이하로 침전시키는 것을 특징으로 하는 알칼리 퓨전을 통한 폐탈질촉매로부터 선택적 유가금속 회수방법.
- 제1항에 있어서,상기 알칼리 침출액에 침전제를 첨가하고 교반하고 반응시키되,333 K 내지 353 K에서 15 분 이하로 반응시켜, 메타바나듐산 칼슘을 침전시키고,313 K 내지 353 K 에서 1시간 내지 2시간 동안 반응시켜 텅스텐산 칼슘을 침전시키는 것을 특징으로 하는 알칼리 퓨전을 통한 폐탈질촉매로부터 선택적 유가금속 회수방법.
- 제1항에 있어서,상기 잔류물을 회수하고,(i) 상기 잔류물에 황산 또는 염산을 첨가하여 산침출시키는 단계;(ii) 산침출액에 수산화나트륨을 첨가하고 반응시켜 메타타이타닉 산[meta titanic cid, TiO(OH)2 ]으로 침전시키는 단계; 및(iii) 상기 메타타이타닉 산을 하소하여 이산화티타늄을 회수하는 단계를 더 포함하는 알칼리 퓨전을 통한 폐탈질촉매로부터 선택적 유가금속 회수방법.
- 제12항에 있어서,상기 산침출은 323 K 내지 333 K에서 2 시간 내지 3 시간 동안 수행되는 것을 특징으로 하는 알칼리 퓨전을 통한 폐탈질촉매로부터 선택적 유가금속 회수방법.
- 제12항에 있어서,상기 수산화나트륨을 첨가하고 반응시키는 단계에서 pH를 2 이하로 조절하여 침출률이 99 % 이상인 것을 특징으로 하는 알칼리 퓨전을 통한 폐탈질촉매로부터 선택적 유가금속 회수방법.
- 제12항에 있어서,상기 하소는 850 K 내지 900 K에서 4 시간 내지 5 시간 수행하는 것을 특징으로 하는 알칼리 퓨전을 통한 폐탈질촉매로부터 선택적 유가금속 회수방법.
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2020503954A JP6955621B2 (ja) | 2017-07-27 | 2018-07-27 | アルカリ融解による廃脱窒触媒から選択的な有価金属の回収方法 |
| EP18839016.5A EP3660175A4 (en) | 2017-07-27 | 2018-07-27 | PROCESS FOR SELECTIVE RECOVERY OF VALUABLE METAL FROM A WASTE DENITRIFICATION CATALYST BY ALKALINE MELTING |
| US16/633,257 US20210130928A1 (en) | 2017-07-27 | 2018-07-27 | Method for selective recovery of valuable metal from waste denitrification catalyst through alkali fusion |
| CN201880048569.1A CN110945151A (zh) | 2017-07-27 | 2018-07-27 | 通过碱熔从废脱氮催化剂中选择性地回收有价金属的方法 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR10-2017-0095357 | 2017-07-27 | ||
| KR1020170095357A KR101813233B1 (ko) | 2017-07-27 | 2017-07-27 | 알칼리 퓨전을 통한 폐탈질촉매로부터 선택적 유가금속 회수방법 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2019022555A1 true WO2019022555A1 (ko) | 2019-01-31 |
Family
ID=60939990
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/KR2018/008523 WO2019022555A1 (ko) | 2017-07-27 | 2018-07-27 | 알칼리 퓨전을 통한 폐탈질촉매로부터 선택적 유가금속 회수방법 |
Country Status (6)
| Country | Link |
|---|---|
| US (1) | US20210130928A1 (ko) |
| EP (1) | EP3660175A4 (ko) |
| JP (1) | JP6955621B2 (ko) |
| KR (1) | KR101813233B1 (ko) |
| CN (1) | CN110945151A (ko) |
| WO (1) | WO2019022555A1 (ko) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN110760683A (zh) * | 2019-11-07 | 2020-02-07 | 河钢股份有限公司承德分公司 | 一种废弃scr催化剂提钒并制备碱金属氟钛酸盐的方法及其产品和用途 |
Families Citing this family (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR101994610B1 (ko) * | 2018-03-21 | 2019-09-30 | 이종애 | 마이크로웨이브를 이용한 탈질 폐촉매 이온 분해 장치 |
| KR101957705B1 (ko) * | 2018-10-24 | 2019-03-13 | 한국지질자원연구원 | 폐탈질촉매로부터 고순도 이산화타이타늄 제조방법 |
| CN109628731B (zh) * | 2019-01-31 | 2020-09-04 | 河钢股份有限公司承德分公司 | 一种短流程处理含钒原料提取制备钒及合金粉末的方法 |
| CN111690824A (zh) * | 2019-03-14 | 2020-09-22 | 边悟 | 含钛钨原料酸解提钛提钨及含钛原料酸解提钛的方法 |
| KR102150087B1 (ko) * | 2019-12-24 | 2020-09-01 | 주식회사 한내포티 | 폐탈질 촉매를 이용한 고품위 텅스텐 및 바나듐의 회수 방법 |
| CN112495982A (zh) * | 2020-10-22 | 2021-03-16 | 湖北君集水处理有限公司 | 一种餐厨垃圾催化湿式氧化转化为废水反硝化碳源的方法 |
| CN112143901B (zh) * | 2020-10-27 | 2022-01-28 | 江西理工大学 | 一种从钒钛系废脱硝催化剂分别回收钛钨钒的方法 |
| BR112023018136A2 (pt) * | 2021-03-10 | 2023-10-31 | National Institutes For Quantum Science And Tech | Método para produção de solução inorgânica e aparelho para produção de solução inorgânica |
| CN113215419B (zh) * | 2021-05-11 | 2022-11-11 | 江西理工大学 | 一种从废scr脱硝催化剂提取有价元素的方法 |
| CN114231760A (zh) * | 2021-12-20 | 2022-03-25 | 安徽工业大学 | 一种硫酸工业废钒催化剂回收利用的方法 |
| CN114534706B (zh) * | 2022-01-17 | 2023-04-21 | 北京科技大学 | 一种废脱硝催化剂回收制备钛硅载体的方法 |
| CN114671464B (zh) * | 2022-03-28 | 2023-09-01 | 江苏龙净科杰环保技术有限公司 | 一种新型废催化剂清洗球磨工艺 |
| CN115228464B (zh) * | 2022-04-22 | 2024-01-19 | 南京市生态环境保护科学研究院 | 一种废脱硝催化剂的资源化利用方法 |
| CN115924926A (zh) * | 2022-11-22 | 2023-04-07 | 辽宁智优环保科技有限公司 | 一种高效回收废scr催化剂中有价值组分的方法 |
| CN115717201A (zh) * | 2022-11-25 | 2023-02-28 | 辽宁智优环保科技有限公司 | 一种从废弃scr脱硝催化剂中回收金属氧化物的方法 |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR100573004B1 (ko) * | 2002-04-23 | 2006-04-24 | 주식회사 한길 | 폐탈질 촉매로부터 바나듐, 텅스텐, 티타늄 성분의 분리회수방법 |
| JP2009511755A (ja) * | 2005-10-18 | 2009-03-19 | ミレニアム インオーガニック ケミカルズ、 インコーポレイテッド | チタン鉱石の選鉱 |
| KR101281579B1 (ko) | 2012-09-07 | 2013-07-03 | 한국지질자원연구원 | 배소 및 수침출을 이용한 탈질폐촉매에 함유된 유가금속 침출 방법 |
| KR20140104254A (ko) * | 2013-02-20 | 2014-08-28 | 한국생산기술연구원 | 배기 가스 정화 장치의 폐촉매에 포함된 금속 정량 방법 |
| KR101452179B1 (ko) * | 2014-03-11 | 2014-10-24 | 한국지질자원연구원 | 탈질 폐촉매의 침출용액으로부터 바나듐 및 텅스텐의 회수방법 |
Family Cites Families (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4168296A (en) * | 1976-06-21 | 1979-09-18 | Lundquist Adolph Q | Extracting tungsten from ores and concentrates |
| JPH04114747A (ja) * | 1990-09-05 | 1992-04-15 | Catalysts & Chem Ind Co Ltd | 使用済触媒から金属成分を回収する方法 |
| CN103028394A (zh) * | 2011-10-10 | 2013-04-10 | 河南佰利联化学股份有限公司 | 一种用偏钛酸制备脱硝用二氧化钛的方法 |
| WO2014047728A1 (en) * | 2012-09-26 | 2014-04-03 | Orbite Aluminae Inc. | Processes for preparing alumina and magnesium chloride by hc1 leaching of various materials |
| CN105648241B (zh) * | 2016-03-18 | 2018-03-09 | 昆明冶金研究院 | 废钒钨钛脱硝催化剂中有价金属钨、钒、钛综合回收的方法 |
| CN105969991A (zh) * | 2016-06-18 | 2016-09-28 | 浙江天蓝环保技术股份有限公司 | 一种从失效scr催化剂中提取钛、钒、钨金属氧化物的方法 |
-
2017
- 2017-07-27 KR KR1020170095357A patent/KR101813233B1/ko active IP Right Grant
-
2018
- 2018-07-27 EP EP18839016.5A patent/EP3660175A4/en active Pending
- 2018-07-27 US US16/633,257 patent/US20210130928A1/en active Pending
- 2018-07-27 CN CN201880048569.1A patent/CN110945151A/zh active Pending
- 2018-07-27 JP JP2020503954A patent/JP6955621B2/ja active Active
- 2018-07-27 WO PCT/KR2018/008523 patent/WO2019022555A1/ko active Application Filing
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR100573004B1 (ko) * | 2002-04-23 | 2006-04-24 | 주식회사 한길 | 폐탈질 촉매로부터 바나듐, 텅스텐, 티타늄 성분의 분리회수방법 |
| JP2009511755A (ja) * | 2005-10-18 | 2009-03-19 | ミレニアム インオーガニック ケミカルズ、 インコーポレイテッド | チタン鉱石の選鉱 |
| KR101281579B1 (ko) | 2012-09-07 | 2013-07-03 | 한국지질자원연구원 | 배소 및 수침출을 이용한 탈질폐촉매에 함유된 유가금속 침출 방법 |
| KR20140104254A (ko) * | 2013-02-20 | 2014-08-28 | 한국생산기술연구원 | 배기 가스 정화 장치의 폐촉매에 포함된 금속 정량 방법 |
| KR101452179B1 (ko) * | 2014-03-11 | 2014-10-24 | 한국지질자원연구원 | 탈질 폐촉매의 침출용액으로부터 바나듐 및 텅스텐의 회수방법 |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP3660175A4 |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN110760683A (zh) * | 2019-11-07 | 2020-02-07 | 河钢股份有限公司承德分公司 | 一种废弃scr催化剂提钒并制备碱金属氟钛酸盐的方法及其产品和用途 |
Also Published As
| Publication number | Publication date |
|---|---|
| EP3660175A4 (en) | 2021-05-05 |
| JP2020528964A (ja) | 2020-10-01 |
| KR101813233B1 (ko) | 2017-12-28 |
| CN110945151A (zh) | 2020-03-31 |
| EP3660175A1 (en) | 2020-06-03 |
| JP6955621B2 (ja) | 2021-10-27 |
| US20210130928A1 (en) | 2021-05-06 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2019022555A1 (ko) | 알칼리 퓨전을 통한 폐탈질촉매로부터 선택적 유가금속 회수방법 | |
| WO2023282564A1 (ko) | 리튬폐액과 불화물폐액으로부터 고부가가치 자원 회수방법 | |
| WO2018070726A1 (ko) | 리튬 화합물의 제조 방법 | |
| WO2012081897A2 (ko) | 니켈 함유 원료로부터 페로니켈을 농축 회수하는 방법, 상기 농축된 페로니켈로부터 니켈을 회수하는 방법 및 상기 방법에서 발생하는 철 함유 용액을 재활용하는 방법 | |
| WO2013165071A1 (ko) | 고순도 황산망간일수화물의 제조방법 및 그 제조방법에 의하여 제조된 고순도 황산망간일수화물 | |
| WO2023282565A1 (ko) | 암모니아 용액을 이용한 오산화바나듐의 부분환원방법 및 이로부터 제조된 이산화바나듐 분말 | |
| WO2017188538A1 (ko) | 6불화우라늄 실린더 세척공정에서 발생한 세척폐액의 처리공정에 의한 우라늄 회수방법 | |
| WO2019209012A1 (ko) | 비침지 방식을 통한 유리의 이온교환 방법 | |
| WO2023136464A1 (ko) | 폐 양극재 반응용기로부터 유가금속 및 제올라이트 함유 물질의 회수방법 | |
| WO2024058441A1 (ko) | 바이오시안화물 및 이온성 액체를 이용하여 폐촉매로부터 백금족 금속을 회수하는 공정 | |
| WO2016032284A1 (ko) | 봉형 산화 몰리브덴의 제조방법 및 산화 몰리브덴 복합체의 제조방법 | |
| WO2022035053A1 (ko) | 활물질 회수 장치 및 이를 이용한 활물질 재사용 방법 | |
| WO2012091437A2 (ko) | 일관제철시스템 및 일관제철방법 | |
| WO2018194397A1 (ko) | 적니를 활용한 일메나이트 제련방법 | |
| WO2022060153A1 (ko) | 에스테르 작용기를 포함하는 고분자의 해중합 촉매 및 이를 이용한 해중합 방법 | |
| WO2017146443A1 (ko) | 폴리에틸렌글리콜 디알데히드 유도체의 제조방법 | |
| WO2012081896A2 (ko) | 니켈 함유 원료로부터 니켈을 회수하는 방법 | |
| WO2024076100A1 (ko) | 니켈 함유 물질로부터 수산화니켈 및 황산니켈 회수방법 | |
| WO2023043071A1 (ko) | 양극 활물질의 재생 방법 및 이로부터 재생된 양극 활물질 | |
| WO2023043095A1 (ko) | 탈질폐촉매 함유 유가금속의 추출방법 | |
| WO2023121346A1 (ko) | 황산나트륨을 함유하는 산업부산물로부터 탄산수소나트륨의 제조장치 | |
| WO2014021639A1 (ko) | 오스테나이트계 스테인리스 냉연강판을 제조하기 위한 고속 산세 프로세스 | |
| WO2022025600A1 (ko) | 폐전극으로부터 알루미늄을 선택적으로 제거하는 방법 및 이를 이용하여 폐전극으로부터 금속 성분을 회수하는 방법 | |
| WO2023182561A1 (ko) | 리튬 이차전지 폐기물로부터 용매추출을 이용한 유가금속의 선택적 회수방법 | |
| WO2024010263A1 (ko) | 재생 양극 활물질, 이의 재생 방법 및 이를 포함하는 이차 전지 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 18839016 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2020503954 Country of ref document: JP Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2018839016 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 2018839016 Country of ref document: EP Effective date: 20200227 |