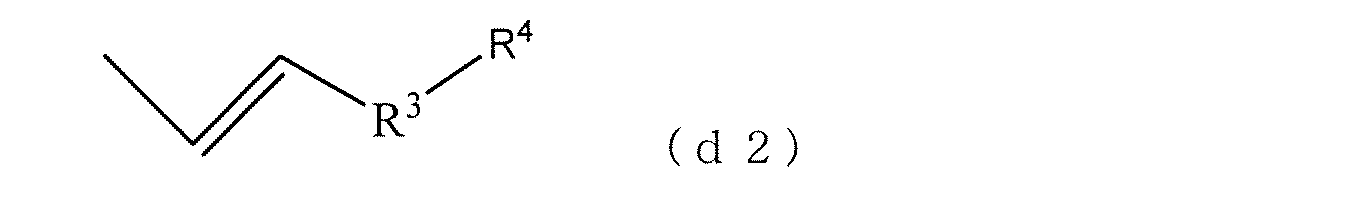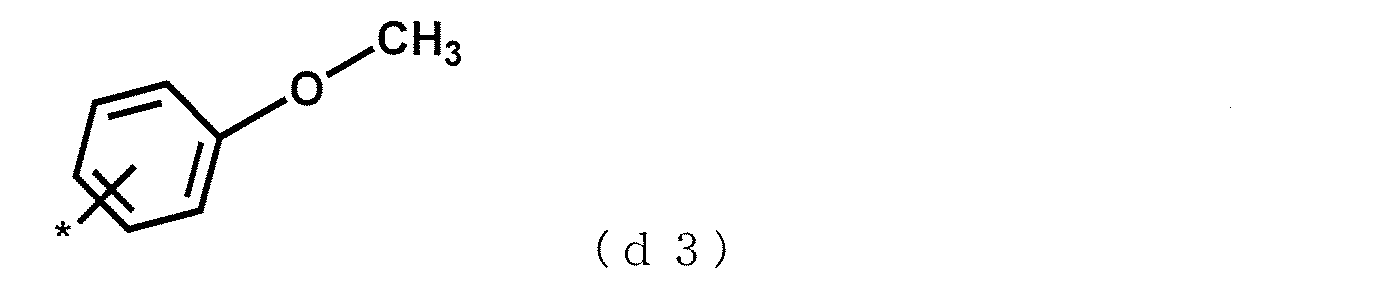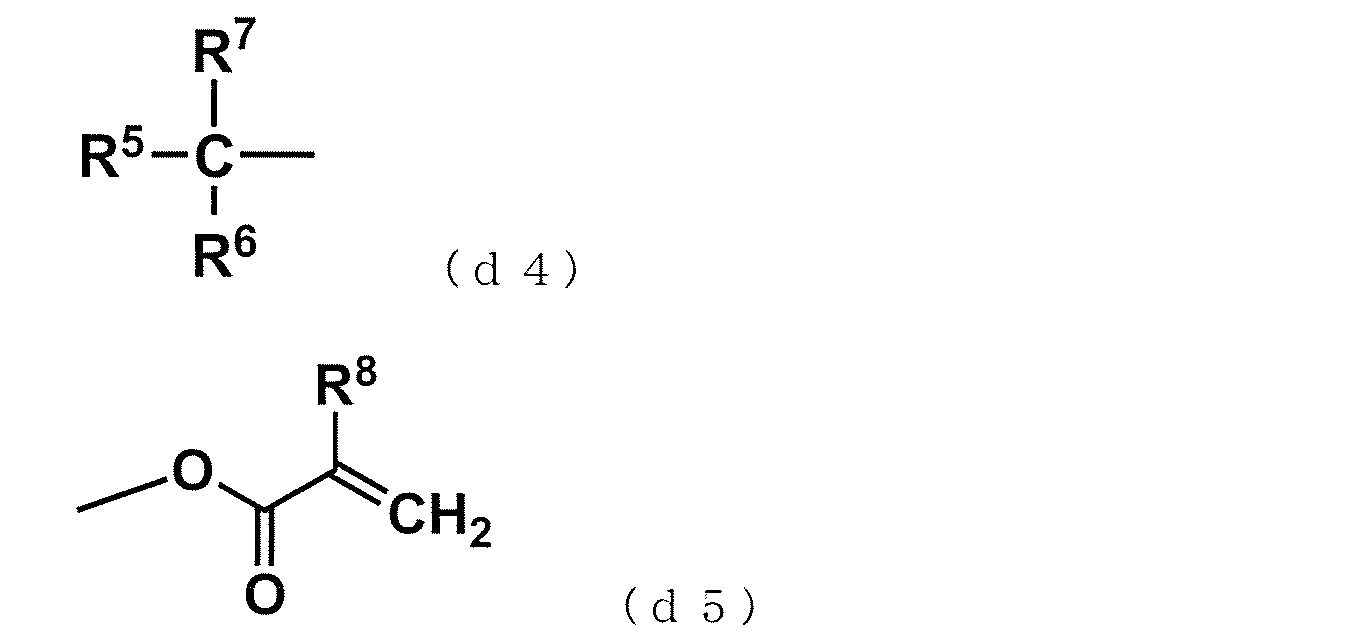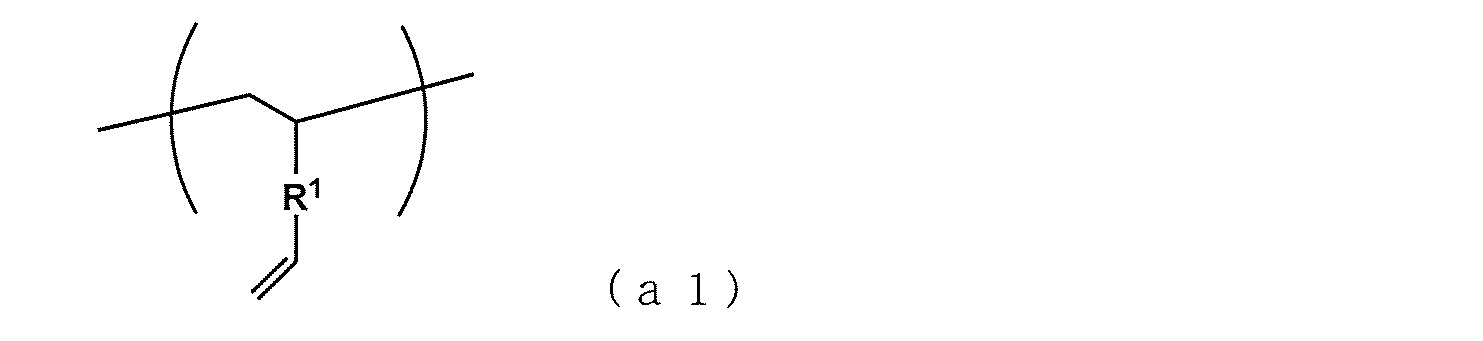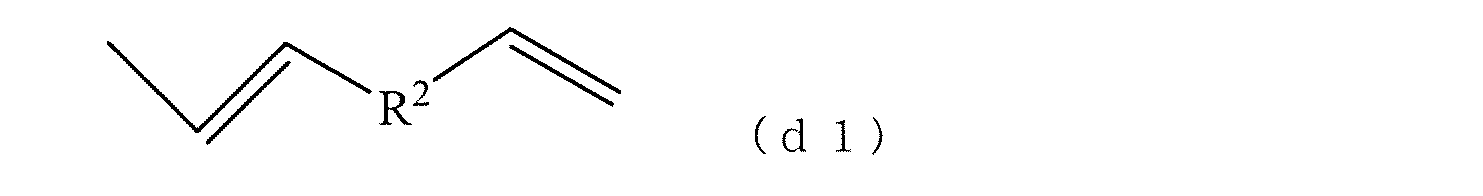WO2018084128A1 - 共重合体ゴム及びその製造方法、並びに架橋ゴム組成物 - Google Patents
共重合体ゴム及びその製造方法、並びに架橋ゴム組成物 Download PDFInfo
- Publication number
- WO2018084128A1 WO2018084128A1 PCT/JP2017/039254 JP2017039254W WO2018084128A1 WO 2018084128 A1 WO2018084128 A1 WO 2018084128A1 JP 2017039254 W JP2017039254 W JP 2017039254W WO 2018084128 A1 WO2018084128 A1 WO 2018084128A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- structural unit
- carbon atoms
- hydrocarbon group
- aromatic
- copolymer
- Prior art date
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F257/00—Macromolecular compounds obtained by polymerising monomers on to polymers of aromatic monomers as defined in group C08F12/00
- C08F257/02—Macromolecular compounds obtained by polymerising monomers on to polymers of aromatic monomers as defined in group C08F12/00 on to polymers of styrene or alkyl-substituted styrenes
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F290/00—Macromolecular compounds obtained by polymerising monomers on to polymers modified by introduction of aliphatic unsaturated end or side groups
- C08F290/02—Macromolecular compounds obtained by polymerising monomers on to polymers modified by introduction of aliphatic unsaturated end or side groups on to polymers modified by introduction of unsaturated end groups
- C08F290/04—Polymers provided for in subclasses C08C or C08F
- C08F290/044—Polymers of aromatic monomers as defined in group C08F12/00
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J3/00—Processes of treating or compounding macromolecular substances
- C08J3/24—Crosslinking, e.g. vulcanising, of macromolecules
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K3/00—Use of inorganic substances as compounding ingredients
- C08K3/01—Use of inorganic substances as compounding ingredients characterized by their specific function
- C08K3/013—Fillers, pigments or reinforcing additives
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L19/00—Compositions of rubbers not provided for in groups C08L7/00 - C08L17/00
- C08L19/003—Precrosslinked rubber; Scrap rubber; Used vulcanised rubber
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L25/00—Compositions of, homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by an aromatic carbocyclic ring; Compositions of derivatives of such polymers
- C08L25/02—Homopolymers or copolymers of hydrocarbons
- C08L25/04—Homopolymers or copolymers of styrene
- C08L25/08—Copolymers of styrene
- C08L25/10—Copolymers of styrene with conjugated dienes
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L9/00—Compositions of homopolymers or copolymers of conjugated diene hydrocarbons
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L9/00—Compositions of homopolymers or copolymers of conjugated diene hydrocarbons
- C08L9/06—Copolymers with styrene
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K3/00—Use of inorganic substances as compounding ingredients
- C08K3/02—Elements
- C08K3/04—Carbon
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02T—CLIMATE CHANGE MITIGATION TECHNOLOGIES RELATED TO TRANSPORTATION
- Y02T10/00—Road transport of goods or passengers
- Y02T10/80—Technologies aiming to reduce greenhouse gasses emissions common to all road transportation technologies
- Y02T10/86—Optimisation of rolling resistance, e.g. weight reduction
Definitions
- the present invention relates to a copolymer rubber having excellent processability and excellent tensile strength and wear resistance, and a crosslinked rubber product obtained by crosslinking the copolymer rubber.
- Conjugated diene rubbers such as SBR (styrene-butadiene rubber), BR (butadiene rubber), and IR (isoprene rubber) styrene-isoprene rubber are excellent in wear resistance, elasticity, water resistance, molding materials, resin modifiers, etc. It is used for various applications.
- This conjugated diene rubber is automobile tires.
- Properties required for a tire include mechanical strength, wear resistance, wet grip properties, and the like (hereinafter, also referred to as strength).
- tires that are excellent in energy saving performance, that is, low fuel consumption have been actively developed. This eco-tyre is required to have low rolling resistance in addition to strength.
- terminal-modified solution polymerization type SBR (terminal-modified S-SBR) has been attracting attention.
- the terminal-modified S-SBR has a functional group at the molecular end of the SBR, and the functional group at the molecular end interacts with the filler.
- the dispersibility of the filler in SBR is improved, and the molecular ends of SBR are constrained to lower the mobility.
- the hysteresis loss (internal friction) of the tire is reduced, and the rolling resistance is reduced. Taking advantage of this characteristic, eco-tires that combine strength and low rolling resistance are being developed.
- Patent Document 1 a block copolymer composed of an ⁇ -methylstyrene block and a butadiene block is synthesized by living anionic polymerization using an organolithium compound as an initiator in a nonpolar solvent, and if necessary, multifunctional By reacting the coupling agent, S-SBR having both high temperature characteristics and rubber properties is obtained.
- Patent Document 2 discloses a star-block interpolymer having a random copolymer block of a conjugated diene and a monovinyl aromatic monomer, a polyconjugated diene block, and a functional group derived from a polyfunctional lithium-based initiator.
- Patent Documents 1 and 2 are considered to have an effect of ensuring the workability of rubber by introducing a branched structure into the rubber component.
- the interaction with the filler for ensuring the strength is not particularly devised, and the contribution to the strength is not sufficient.
- Patent Document 3 a rubber composition in which a predetermined amount of carbon black is blended with a blend rubber containing a plurality of diene rubbers, has a functional group that interacts with the carbon black at the molecular chain end, and A rubber composition comprising a low molecular weight functional group-containing polymer having a polymer structure similar to the rubber component of the diene rubber is disclosed.
- the amount of carbon black distributed in each diene rubber component can be controlled by blending a rubber with a low molecular compound that interacts with carbon black. Therefore, the features of each rubber component can be effectively expressed, and for example, it is possible to achieve both rubber characteristics having a contradiction such as rolling characteristics and wet characteristics.
- Patent Document 4 discloses a crosslinked rubber particle containing a conjugated diene monomer unit, an aromatic vinyl monomer unit and a monomer unit having at least two polymerizable unsaturated groups, and a specific bonding structure.
- the inorganic filler such as silica
- the substances disclosed as the monomer unit having at least two polymerizable unsaturated groups and the monomer unit having a carboxylic acid group, a hydroxyl group and / or an epoxy group are all low molecular weight. is there. Therefore, the reactivity is excessively high, and there is a possibility that gelation proceeds in the crosslinked rubber particles and the rubber composition.
- Patent Document 5 discloses a soluble polyfunctional vinyl aromatic copolymer, but does not teach its use in the production of copolymer rubber.
- the present invention aims to solve such problems and provide a material having workability, strength and homogeneity.
- the present inventors have used the above-mentioned specific polyfunctional vinyl aromatic copolymer compound having both a branched structure and an interaction function with a filler as a constituent unit of the conjugated diene rubber.
- the present inventors have found that the problems can be solved and completed the present invention.
- the polyfunctional vinyl aromatic copolymer (A) comprises a structural unit (a) derived from a divinyl aromatic compound, a structural unit (b) derived from a monovinyl aromatic compound, and a structural unit (d) constituting a terminal.
- At least a part of the structural unit (a) is a vinyl group-containing structural unit (a1) represented by the following formula: (In the formula, R 1 represents an aromatic hydrocarbon group having 6 to 30 carbon atoms.) At least another part of the structural unit (a) is a structural unit constituting the structural unit (d), and the mole fraction of the vinyl group-containing structural unit (a1) relative to the sum of the structural units (a) and (b) is A method for producing a copolymer rubber, characterized in that it has a range of 0.02 to 0.8 and a number average molecular weight of 300 to 100,000.
- a polyfunctional vinyl aromatic copolymer (A) and a conjugated diene compound (B) or a raw material containing a conjugated diene compound (B) and an aromatic vinyl compound (C) are copolymerized and copolymerized.
- the polyfunctional vinyl aromatic copolymer is used as the polyfunctional vinyl aromatic copolymer (A).
- the structural unit (d) constituting the terminal of the polyfunctional vinyl aromatic copolymer (A) is any of the units represented by the following (d1) to (d6). However, in the case of the unit represented by (d4) or (d5), it has the unit represented by (d4) or (d5) at the same time.
- R 2 represents an aromatic hydrocarbon group having 6 to 30 carbon atoms.
- R 3 represents an aromatic hydrocarbon group having 6 to 30 carbon atoms
- R 4 represents a hydrogen atom or a hydrocarbon group having 6 to 30 carbon atoms.
- Y represents an aromatic hydrocarbon group having 6 to 52 carbon atoms which is unsubstituted or substituted with a hydrocarbon group having 1 to 12 carbon atoms
- Z is an aliphatic hydrocarbon group having 1 to 30 carbon atoms
- R 5 represents a hydrogen atom or a hydrocarbon group having 1 to 18 carbon atoms
- R 6 and R 7 represent a hydrocarbon group having 1 to 18 carbon atoms
- R 8 represents a hydrogen atom or a methyl group.
- R 9 is a hydrocarbon group having 4 to 16 carbon atoms having one or more alicyclic hydrocarbon rings.
- Anionic polymerization is suitable as the copolymerization method.
- Another aspect of the present invention is a cross-linking characterized in that a rubber composition is obtained by blending a filler with the copolymer rubber obtained by the above-described method for producing a copolymer rubber and cross-linking by vulcanization. It is a manufacturing method of a rubber composition.
- another embodiment of the present invention is the structural unit (A1) of the polyfunctional vinyl aromatic copolymer, the structural unit (B1) of the conjugated diene compound, or the structural unit (B1) of the conjugated diene compound and the aromatic vinyl.
- the structural unit (A1) contains a structural unit (a) derived from a divinyl aromatic compound, a structural unit (b) derived from a monovinyl aromatic compound, and a structural unit (d) constituting a terminal, a) is contained in an amount of 2 mol% or more and less than 95 mol%, and at least a part of the structural unit (a) is a vinyl group-containing structural unit (a1) represented by the above formula, and at least another structural unit (a)
- the number average molecular weight of the structural unit (A1) in the copolymer rubber is preferably 300 to 100,000.
- the structural unit (d) constituting the terminal in the copolymer rubber may be any of the units represented by the above (d1) to (d6). However, in the case of the unit represented by (d4) or (d5), it has the unit represented by (d4) or (d5) at the same time.
- Another aspect of the present invention is a crosslinked rubber composition containing the above copolymer rubber and a filler, wherein the copolymer rubber has a crosslinked structure. Furthermore, it is a polyfunctional vinyl aromatic copolymer characterized by being used for the copolymer rubber or the crosslinked rubber composition.
- the copolymer rubber of the present invention has a structural unit of a specific polyfunctional vinyl aromatic copolymer having both a branched structure and an interaction function with a filler, it has both workability and strength. Furthermore, it is difficult to form a gel substance, it becomes homogeneous, and can be applied to molding materials, resin modifiers, and the like. Furthermore, since this copolymer rubber contains a filler and is crosslinked, the crosslinked rubber composition is excellent in dispersibility of the filler, and thus is excellent in mechanical strength and wear resistance.
- a raw material containing a polyfunctional vinyl aromatic copolymer (A), a conjugated diene compound (B) and, if necessary, an aromatic vinyl compound (C) as a raw material Copolymerize.
- the polyfunctional vinyl aromatic copolymer (A) is preferably obtained by copolymerizing a divinyl aromatic compound and a monovinyl aromatic compound and having solvent solubility. For example, it may be soluble in toluene, xylene, tetrahydrofuran, dichloroethane or chloroform.
- the number average molecular weight Mn of the polyfunctional vinyl aromatic copolymer (A) is 300 to 100,000, preferably 500 to 5,000.
- the molecular weight distribution Mw / Mn is preferably in the range of 3-20.
- the divinyl aromatic compound gives the structural unit (a), and the monovinyl aromatic compound gives the structural unit (b). Since the divinyl aromatic compound has a plurality of vinyl groups, it is crosslinked and gives a branched structure, but the degree of crosslinking is controlled to the extent that it exhibits solubility.
- the structural unit having an unreacted vinyl group becomes the structural unit (a) or the structural unit (d1) as the structural unit (d) constituting the terminal, and gives a polyfunctional structure having a plurality of vinyl groups.
- the polyfunctional vinyl aromatic copolymer (A) contains 2 mol% or more and less than 95 mol% of the structural unit (a).
- the content is preferably 5 to 90 mol%, more preferably 10 to 90 mol%, and particularly preferably 15 to 85 mol%.
- a part of the structural unit (a) may contain 1 mol% or more of the structural unit (a1), preferably 2 to 80 mol%, more preferably 5 to 70 mol%, still more preferably. Is from 10 to 60 mol%, particularly preferably from 15 to 50 mol%.
- a part of the structural unit (a) is contained as a structural unit (d1) in the structural unit (d) constituting the terminal, and the amount thereof is 0.7 or more per molecule, preferably 1.0.
- the structural unit (a) includes the structural unit (a1) and the structural unit (d1).
- the terminal structural unit (d) may have an average of 2.1 or more, preferably 2.5 or more, more preferably 3 or more per molecule.
- the polyfunctional vinyl aromatic copolymer (A) contains 5 mol% or more and less than 98 mol% of the structural unit (b).
- the content is preferably 10 to 95 mol%, more preferably 10 to 90 mol%, and particularly preferably 15 to 85 mol%.
- a part of the structural unit (b) may be a terminal structural unit (d). This structural unit is referred to as a structural unit (d2).
- the structural unit (a) derived from a divinyl aromatic compound contains a vinyl group as a crosslinking component for developing heat resistance and rigidity, while the structural unit (b) derived from a monovinyl aromatic compound is a curing reaction. Since it does not have a vinyl group involved in the above, it gives flexibility and solubility.
- the terminal structural unit (d) may consist of a part of the structural unit (a) and the structural unit (b), and other monomers other than the divinyl aromatic compound and the monovinyl aromatic compound (G ), And some or all of the structural units generated therefrom may be the terminal structural unit (d).
- This structural unit is referred to as a structural unit (d3).
- the polyfunctional vinyl aromatic copolymer (A) can be obtained by the method described in Patent Document 5 and the like. For example, it can be obtained by polymerizing a monomer containing a divinyl aromatic compound, a monovinyl aromatic compound and, if necessary, another monomer (G) in a homogeneous solvent dissolved in a solvent at a temperature of 20 to 120 ° C.
- divinyl aromatic compounds include divinylbenzene (including each isomer), divinylnaphthalene (including each isomer), and divinylbiphenyl (including each isomer), but are not limited thereto. is not. Moreover, these can be used individually or in combination of 2 or more types. From the viewpoint of moldability, divinylbenzene (m-isomer, p-isomer or a mixture of isomers thereof) is more preferable.
- monovinyl aromatic compounds examples include vinyl aromatic compounds such as styrene, vinyl naphthalene, vinyl biphenyl, o-methyl styrene, m-methyl styrene, p-methyl styrene, o, p-dimethyl styrene, o-ethyl vinyl benzene.
- Nucleoalkyl-substituted vinyl aromatic compounds such as m-ethylvinylbenzene and p-ethylvinylbenzene, etc., including styrene, ethylvinylbenzene (including each isomer), ethylvinylbiphenyl (including each isomer), And ethyl vinyl naphthalene (including each isomer) are preferred.
- R 2 represents an aromatic hydrocarbon group having 6 to 30 carbon atoms.
- Formula (d1) can arise from divinyl aromatic compounds as monomers. Specific examples include divinylbenzene, divinylnaphthalene, divinylanthracene, divinylphenanthrene, divinylbiphenyl, divinyltriphenyl, and the like. Divinylbenzene, divinylnaphthalene, and divinylbiphenyl are preferred because the polyfunctional vinyl aromatic copolymer (A) is easy to handle in rubber solution polymerization and has excellent interaction with the filler.
- R 3 represents an aromatic hydrocarbon group having 6 to 30 carbon atoms
- R 4 represents a hydrogen atom or a hydrocarbon group having 6 to 30 carbon atoms.
- Formula (d2) can arise from monovinyl aromatic compounds as monomers.
- R 3 is a phenyl group, a naphthyl group, or a biphenylyl group because of easy handling in rubber solution polymerization of the polyfunctional vinyl aromatic copolymer (A) and excellent interaction with the filler
- R 4 Is a hydrogen atom or an alkyl group having 1 to 10 carbon atoms.
- Y represents an aromatic hydrocarbon group having 6 to 52 carbon atoms which is unsubstituted or substituted with a hydrocarbon group having 1 to 12 carbon atoms
- Z is an aliphatic hydrocarbon having 1 to 30 carbon atoms.
- Formula (d3) can be generated from anisole, ethoxybenzene, propoxybenzene, butoxybenzene, methoxynaphthalene, methoxybiphenyl, biphenyl ether used as monomers (G). Of these, anisole, butoxybenzene, and methoxynaphthalene are preferred because they are easy to handle in rubber solution polymerization of the polyfunctional vinyl aromatic copolymer (A) and have excellent interaction with the filler.
- R 5 represents a hydrogen atom or a hydrocarbon group having 1 to 18 carbon atoms
- R 6 and R 7 represent a hydrocarbon group having 1 to 18 carbon atoms
- R 6 , R 7 may combine with each other to form a ring.
- R 6 and R 7 form a ring with each other, the total carbon number of R 6 and R 7 is 2 to 36.
- R 8 represents a hydrogen atom or a methyl group.
- Formulas (d4) and (d5) are methyl (meth) acrylate, ethyl (meth) acrylate, n-propyl (meth) acrylate, isopropyl (meth) acrylate used as monomers (G), N-butyl (meth) acrylate, isobutyl (meth) acrylate, sec-butyl (meth) acrylate, t-butyl (meth) acrylate, aliphatic (meth) acrylates of 2-ethylhexyl (meth) acrylate, (Meth) acrylates having an alicyclic ring such as cyclopentyl (meth) acrylate and cyclohexyl methacrylic acid, and (meth) acrylates having an aromatic ring such as phenyl (meth) acrylate and naphthyl (meth) acrylate Etc.
- the polyfunctional vinyl aromatic copolymer (A) is easy to handle in rubber solution polymerization and has excellent interaction with the filler, so that isopropyl (meth) acrylate, isobutyl (meth) acrylate, (meth ) Sec-butyl acrylate, t-butyl (meth) acrylate.
- R 9 is a hydrocarbon group having 4 to 6 carbon atoms having one or more alicyclic hydrocarbon rings. R 9 is bonded to the molecular chain through an adjacent carbon-carbon double bond.
- Formula (d6) can arise from cyclopentene, cyclohexene, norbornene, dicyclopenta-monoene, etc. used as monomers (G).
- norbornene is used because the polyfunctional vinyl aromatic copolymer (A) is easy to handle in rubber solution polymerization and has excellent interaction with the filler.
- An acid catalyst or a Lewis acid catalyst is suitable as a catalyst for copolymerizing a monomer containing a divinyl aromatic compound or a monovinyl aromatic compound and, if necessary, another monomer (G).
- the acid catalyst sulfonic acid catalysts such as alkylsulfonic acid and toluenesulfonic acid can be used. The amount used is 0.1 to 10 moles with respect to a total of 100 moles of all monomer components.
- the Lewis acid catalyst can be used without particular limitation as long as it is a compound composed of a metal ion (acid) and a ligand (base) and can receive an electron pair.
- a metal fluoride or a complex thereof is preferable from the viewpoint of the thermal decomposition resistance of the copolymer obtained, and particularly B, Al, Ga, In, Si, Ge, Sn, Pb, Sb, Bi, Ti, Divalent to hexavalent metal fluorides such as W, Zn, Fe and V or complexes thereof are preferred.
- These catalysts can be used alone or in combination of two or more.
- boron trifluoride ether complexes are most preferably used.
- examples of the ether of the ether complex include diethyl ether and dimethyl ether.
- the Lewis acid catalyst is preferably used in a range of 0.001 to 100 mol, more preferably 0.01 to 50 mol, per 100 mol of all monomer components. Most preferably, it is 0.1 to 10 mol. If desired, one or more Lewis base compounds can be used as a cocatalyst.
- Lewis base compounds include ester compounds such as ethyl acetate, thioester compounds such as methyl mercaptopropionic acid, ketone compounds such as methyl ethyl ketone, amine compounds such as methylamine, ether compounds of diethyl ether, diethyl There are thioether compounds such as sulfide and phosphine compounds such as tripropylphosphine and tributylphosphine.
- the polymerization reaction is carried out, for example, at 20 to 120 ° C., preferably 40 to 100 ° C. in a homogeneous solvent in which the monomer and acid catalyst or Lewis acid catalyst are dissolved in an organic solvent having a dielectric constant of 2.0 to 15.0.
- a cationic copolymerization method is suitable.
- the organic solvent toluene, xylene, n-hexane, cyclohexane, methylcyclohexane or ethylcyclohexane is particularly preferable from the viewpoint of a balance between polymerization activity and solubility.
- the method for recovering the polyfunctional vinyl aromatic copolymer is not particularly limited.
- a commonly used method such as a heat concentration method, a steam stripping method, or precipitation with a poor solvent may be used.
- a copolymer rubber is produced by copolymerizing the polyfunctional vinyl aromatic copolymer (A), the conjugated diene compound (B), and a raw material containing the aromatic vinyl compound (C) if necessary.
- a diene rubber such as butadiene rubber or isoprene rubber can be obtained.
- a co-rubber rubber such as SBR can be obtained. Obtainable.
- a method for producing a copolymer rubber by copolymerizing a conjugated diene compound (B) or a raw material containing the conjugated diene compound (B) and an aromatic vinyl compound (C) is a method in which a polyfunctional vinyl aromatic copolymer (A) is one of the raw materials.
- the methods described in Patent Documents 1 to 4 can be employed except that
- Examples of the conjugated diene compound (B) include 1,3-butadiene, isoprene, 1,3-pentadiene, 2,3-dimethyl-1,3-butadiene, 2-phenyl-1,3-butadiene, and 1,3-hexadiene. Can be used singly or in combination of two or more, among which 1,3-butadiene, isoprene and 2,3-dimethyl-1,3-butadiene are preferred.
- Aromatic vinyl compounds include styrene, ⁇ -methylstyrene, 1-vinylnaphthalene, 3-vinyltoluene, ethylvinylbenzene, divinylbenzene, 4-cyclohexylstyrene, 2,4,6-trimethylstyrene, tert-butoxy.
- Dimethylsilylstyrene, isopropoxydimethylsilylstyrene, and the like can be used alone or in combination of two or more. Among these, styrene is preferable.
- styrene-butadiene rubber SBR
- butadiene rubber BR
- isoprene rubber IR
- a styrene-butadiene rubber (SBR) structure is particularly preferable because of excellent wear resistance, heat resistance, and aging resistance.
- the method for producing a copolymer rubber by copolymerizing raw materials containing these is not limited, but anionic polymerization in a hydrocarbon solvent is preferred.
- a polyfunctional vinyl aromatic copolymer, a conjugated diene compound or a method of living anion polymerization of these with an aromatic vinyl compound using an organic alkali metal compound as an initiator in a hydrocarbon solvent, or a conjugated diene compound there is a multistage reaction method in which a living anion polymerization of an aromatic vinyl compound is performed, and then a reaction is performed by adding a polyfunctional vinyl aromatic copolymer.
- the terminal may be terminally modified with a polymerization terminator having a functional group.
- hydrocarbon solvent used in the above method examples include propane, n-butane, isobutane, n-pentane, isopentane, n-hexane, cyclohexane, propene, 1-butene, isobutene, and trans-2- Butene, cis-2-butene, 1-pentyne, 2-pentyne, 1-hexene, 2-hexene, benzene, toluene, xylene, ethylbenzene, heptane, cyclopentane, methylcyclopentane, methylcyclohexane, 1-pentene, 2- Examples include pentene and cyclohexene.
- organic alkali metal used as the initiator examples include methyl lithium, ethyl lithium, n-propyl lithium, iso-propyl lithium, n-butyl lithium, sec-butyl lithium, tert-octyl lithium, n-decyl lithium, and phenyl.
- examples thereof include lithium, 2-naphthyl lithium, 2-butyl-phenyl lithium, 4-phenyl-butyl lithium, cyclohexyl lithium, and a reaction product of diisopropenylbenzene and butyl lithium.
- the method for using the polyfunctional vinyl aromatic copolymer (A) in the polymerization is not particularly limited, and as an example, a method for initiating polymerization in the presence of the conjugated diene compound (B) and, if necessary, the aromatic vinyl compound (C). (B) and a method of adding an aromatic vinyl compound (C) if necessary after reacting (A) with a polymerization initiator, a conjugated diene compound (B) and an aromatic vinyl compound (C) if necessary.
- Examples thereof include a method of adding a vinyl aromatic copolymer (A) and coupling rubber molecules.
- the weight average molecular weight of the copolymer rubber is preferably in the range of 100,000 to 500,000.
- the reaction can be terminated by adding alkanol or the like, and the solvent can be removed to obtain a copolymer rubber.
- the use ratio of the polyfunctional vinyl aromatic copolymer (A), the conjugated diene compound (B), and the aromatic vinyl compound (C) is preferably in the following range.
- the polyfunctional vinyl aromatic copolymer (A) is 0.001 to 6% by weight, preferably 0.005 to 3% by weight, more preferably 0.01 to 2% by weight.
- the conjugated diene compound (B) Is 94 to 99.999% by weight, and the aromatic vinyl compound (C) is 0 to 70% by weight.
- the aromatic vinyl compound (C) is used, the polyfunctional vinyl aromatic copolymer (A) is in the same range as described above, and the conjugated diene compound (B) is 50 to 97% by weight, preferably 55%.
- the aromatic vinyl compound (C) is 2 to 50% by weight, preferably 5 to 45% by weight. More preferably, the polyfunctional vinyl aromatic copolymer (A) is 0.01 to 23% by weight, more preferably 0.1 to 1% by weight.
- the conjugated diene compound (B) is 50 to 97% by weight, more preferably 55 to 90% by weight.
- the aromatic vinyl compound (C) is preferably 2 to 49% by weight, more preferably 5 to 44% by weight.
- the content ratio of the structural unit (A1) of the polyfunctional vinyl aromatic copolymer, the structural unit (B1) of the conjugated diene compound, and the structural unit (C1) of the aromatic vinyl compound in the copolymer rubber of the present invention is
- the unit (A1) is 0.001 to 6% by weight
- the structural unit (B1) is 29 to 99.999% by weight
- the structural unit (C1) is 0 to 70% by weight.
- It contains a structural unit (a) derived from a divinyl aromatic compound, a structural unit (b) derived from a monovinyl aromatic compound, and a structural unit (d) constituting a terminal, and the structural unit (a) is 2 mol% or more.
- At least part of the structural unit (a) is a vinyl group-containing structural unit (a1) represented by the following formula, (In the formula, R 1 represents an aromatic hydrocarbon group having 6 to 30 carbon atoms.)
- At least another part of the structural unit (a) is a structural unit constituting the structural unit (d), and the mole fraction of the vinyl group-containing structural unit (a1) relative to the sum of the structural units (a) and (b) is The range is from 0.02 to 0.8, and the number average molecular weight is from 300 to 100,000.
- a preferable range is preferably a ratio similar to the ratio of the raw materials used.
- the crosslinked rubber composition of the present invention can be obtained by blending the copolymer rubber with a filler or a crosslinking agent to form a rubber composition, and crosslinking the rubber composition by vulcanization.
- a filler include carbon black and silica. Excellent interaction with carbon black.
- carbon black contained as the filler carbon black of each grade such as SRF, GPF, FEF, HAF, ISAF, SAF and the like can be used. Among these, HAF, ISAF, and SAF are preferable because a rubber elastic body having excellent wear resistance can be obtained.
- the silica may be in the form of particles generally used as a filler, but the primary particle diameter is preferably 50 nm or less. Specific examples of such silica include hydrous silicic acid, anhydrous silicic acid, calcium silicate, and aluminum silicate.
- the filler content is preferably 10 to 120 parts by weight with respect to 100 parts by weight of the total rubber component including the copolymer rubber, and 55 to 100 parts by weight from the viewpoint of reinforcing properties and the effect of improving various physical properties thereby. More preferably, it is part.
- the crosslinking agent contained in the rubber composition is not particularly limited, such as crosslinking with sulfur and crosslinking with peroxide, but sulfur is usually used.
- the content of the cross-linking agent is preferably 0.1 to 5 parts by weight, more preferably 1 to 3 parts by weight with respect to 100 parts by weight of the total rubber component.
- rubber compounding oils such as silane coupling agents, paraffinic oils, naphthenic oils, aroma oils, waxes, anti-aging agents, stearic acid (additives). Sulfur aids and processing aids), zinc oxide, vulcanization accelerators, and the like may be included.
- the rubber composition can be prepared by kneading each component using a kneader such as a plast mill, a banbury mixer, a roll, an internal mixer.
- a kneader such as a plast mill, a banbury mixer, a roll, an internal mixer.
- components other than the crosslinking agent and the vulcanization accelerator may be kneaded, and then the resulting kneaded product may be further kneaded by adding the crosslinking agent and the vulcanization accelerator.
- a kneader such as a plast mill, a banbury mixer, a roll, an internal mixer.
- the rubber composition of the present invention is excellent in mechanical strength and wear resistance, it is suitable as a rubber composition for obtaining tire treads and sidewall members of low fuel consumption tires, large tires, high performance tires and the like. . It can also be suitably used for rubber belts, rubber hoses, footwear materials, and the like.
- the crosslinked rubber composition of the present invention can be obtained by crosslinking the above rubber composition.
- a tire manufactures a tread by extruding and molding the above rubber composition according to the shape of the tire (specifically, the shape of the tread), and heating and pressing it in a vulcanizer. And the target tire can be manufactured by assembling this tread and other parts.
- the present invention will be specifically described by way of examples. However, the present invention is not limited to these examples.
- the part in each example is a weight part when there is no description in particular, and evaluation of each physical property was performed by the method shown below.
- the terminal group is calculated based on the number average molecular weight obtained from the GPC measurement and the total monomer amount obtained from the results of 13 C-NMR and 1 H-NMR measurement and gas chromatograph (GC) analysis. From the amount of the derivative used for introducing the polyfunctional vinyl aromatic copolymer, the number of terminal groups contained in one molecule was calculated.
- the terminal group is generated from a divinyl aromatic compound or a monovinyl aromatic compound.
- the calculation of the end group is based on the data regarding the total amount of each structural unit introduced into the copolymer obtained from GC analysis in addition to the results of 13 C-NMR and 1 H-NMR measurement.
- the amount of structural unit introduced is calculated, and is included in one molecule of the polyfunctional vinyl aromatic copolymer from the amount of the specific structural unit introduced at the terminal and the number average molecular weight obtained from the GPC measurement.
- the number of terminal groups of a specific structural unit was calculated.
- Mooney viscosity (ML (1 + 4) 100 ° C.) According to JIS K6300-1, L-type rotor, preheating 1 minute, rotor operating time 4 minutes, temperature 100 ° C.
- Synthesis example 1 DVB-810 (manufactured by Nippon Steel & Sumikin Chemical Co., Ltd., 81.0 wt% of divinylbenzene component) 320.5 mL (1.82 mol of divinylbenzene component, 0.43 mol of ethylvinylbenzene component), 0.28 mol of n-butyl acetate (36.9 mL), 140 mL of toluene was charged into a 1.0 L reactor, and a solution of 40 mmol of methanesulfonic acid dissolved in 0.12 mol (15.7 mL) of n-butyl acetate at 70 ° C. was added. And reacted for 6 hours.
- copolymer X1 a soluble polyfunctional vinyl aromatic copolymer
- Mn of the obtained copolymer X1 was 1085
- Mw was 12400
- Mw / Mn was 11.4.
- the copolymer X was found to contain the following vinyl group-containing structural unit (a1) and terminal group (a) derived from the divinyl aromatic compound (a). It was confirmed that d1) had the following end group (d2) derived from the monovinyl aromatic compound (b).
- the number of terminal structural units (d1) per molecule was 7.7.
- the copolymer X1 contained the structural unit derived from a divinylbenzene component and ethylvinylbenzene, respectively with respect to the sum total of 84.0 mol% and 16.0 mol%.
- the molar fraction of the vinyl group-containing structural unit (a1) with respect to the entire structure was 0.39.
- the copolymer X1 was soluble in toluene, xylene, THF, dichloroethane, dichloromethane, and chloroform, and no gel was observed.
- Synthesis example 2 DVB-810 276.4 mL, toluene 86.2 mL, and anisole 93.74 mL were charged into a 1.0 L reactor, and a solution of 10 mmol of methanesulfonic acid dissolved in 2 mL of toluene at 50 ° C. was added.
- a copolymer X2 was obtained in the same manner as in Synthesis Example 1 except that the reaction was performed for a period of time. Mn of the obtained copolymer X2 was 1000, Mw was 18800, and Mw / Mn was 18.8.
- the copolymer X2 is the same vinyl group-containing structural unit (a1) as in Synthesis Example 1 derived from the divinyl aromatic compound (a). , And the following end group (d3) derived from anisole.
- the copolymer X2 contains 64.0 mol% of the structural unit (a) derived from the divinylbenzene component with respect to the entire structure, and contains an average of 2.0 terminal structural groups (d3) derived from anisole per molecule. It was. Moreover, the molar fraction of the vinyl group-containing structural unit (a1) with respect to the entire structure was 0.12. Further, it was soluble in toluene, xylene, THF, dichloroethane, dichloromethane and chloroform, and no gel was formed.
- Synthesis example 3 DVB-810 300 mL, toluene 177.6 mL, t-butyl methacrylate 145.6 mL, a solution of 10 mmol of methanesulfonic acid dissolved in 2 mL of toluene at 60 ° C. was added and reacted for 4 hours. 1 to obtain a copolymer X3.
- Mn of the obtained copolymer X3 was 2190, Mw was 13600, and Mw / Mn was 7.2.
- copolymer X3 71 mol% of the structural unit (a) derived from the divinylbenzene component and 1 to 2 of the terminal structural groups (d4) and (d5) derived from t-butyl methacrylate are averaged per molecule. Seven were contained. Further, the molar fraction of the vinyl group-containing structural unit (a1) with respect to the entire structure was 0.53. Copolymer X3 was soluble in toluene, xylene, THF, dichloroethane, dichloromethane, and chloroform, and no gel was observed.
- Example 1 To an autoclave reactor with an internal volume of 0.5 liters purged with nitrogen, 245 g of cyclohexane, 2.5 g of THF, 10 g of styrene, 40 g of 1,3-butadiene, and 0.015 g of the copolymer X1 obtained in Synthesis Example 1 were added. . At 25 ° C., 5 g of a cyclohexane solution containing 50 mg of sec-butyllithium was added to initiate polymerization. The temperature of the reaction solution increased due to the heat of polymerization, and the maximum temperature reached 85 ° C.
- Example 2 Copolymer rubber A, process oil, carbon black, zinc oxide, stearic acid and an antioxidant were kneaded for 4 minutes at 155 ° C. and 60 rpm using a lab plast mill. Sulfur and a vulcanization accelerator were added to the kneaded product obtained by the kneading, kneaded at 70 ° C. and 60 rpm for 1 minute using a lab plast mill, and vulcanized to obtain a crosslinked rubber A.
- Table 2 shows the blending ratio of each additive.
- Table 3 shows the physical properties of the crosslinked rubber A.
- Examples 3 to 4 A copolymer rubber C and a copolymer rubber D were obtained in the same manner as in Example 1, except that the copolymer X2 and the copolymer X3 were used instead of the copolymer X1, respectively.
- the obtained physical properties are shown in Table 1.
- Examples 5-6 A crosslinked rubber C and a crosslinked rubber D were obtained in the same manner as in Example 2 except that the copolymer rubber C and the copolymer rubber D were used in place of the copolymer rubber A, respectively.
- Table 3 shows the obtained physical properties.
- copolymer rubber B was prepared in the same manner as in Example 1 except that 0.005 g of DVB-810 in terms of divinylbenzene was used instead of copolymer X1 synthesized in Synthesis Example 1. Got. Table 1 shows the physical properties of the obtained copolymer rubber B.
- Comparative Example 2 Furthermore, using the copolymer rubber B, a crosslinked rubber B was obtained in the same manner as in Example 1 and the blending ratio of each additive. Table 3 shows the physical properties of the crosslinked rubber B.
- the rubber composition of the present invention gives processability equivalent to that when divinylbenzene, which is a known branching agent, is used, and in vulcanized rubber compounded with carbon black, tensile strength and abrasion resistance It turns out that it is excellent in property.
- the copolymer rubber of the present invention is useful as an elastomer material for tires (particularly tire treads), seismic isolation rubbers, rubber hoses, rubber rollers, footwear materials and the like.
Landscapes
- Chemical & Material Sciences (AREA)
- Health & Medical Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
- Organic Chemistry (AREA)
- Compositions Of Macromolecular Compounds (AREA)
- Addition Polymer Or Copolymer, Post-Treatments, Or Chemical Modifications (AREA)
- Graft Or Block Polymers (AREA)
Abstract
Description
また、特許文献2では、共役ジエン及びモノビニル芳香族モノマーのランダムコポリマーブロックと、ポリ共役ジエンブロックと、多官能性リチウム系開始剤由来の官能基とを有する、星形-ブロックインターポリマーが開示され、転がり抵抗の低減やトラクション特性の改善といった優れた特性を有するタイヤトレッドの作製におけるゴムとして、広く使用することができることが開示されている。
特許文献1及び2の技術は、ゴム成分に分岐構造を導入することで、ゴムの加工性を担保する効果があると考えられる。しかし、強度を担保するためのフィラーとの相互作用については、特段の工夫はなく、強度に対する寄与は十分ではない。
また、特許文献4では、共役ジエン単量体単位、芳香族ビニル単量体単位及び少なくとも2個の重合性不飽和基を有する単量体単位を含む架橋ゴム粒子、並びに、特定の結合構造を有する共役ジエン単量体単位を含む共役ジエン/芳香族ビニル共重合ゴムを含有するゴム組成物が開示され、この架橋ゴム粒子はカルボン酸基、ヒドロキシル基及び/又はエポキシ基を有する単量体単位を含んでも良いと開示されている。この技術は、シリカ等の無機充填剤(フィラー)との適度な相互作用を有することから、無機充填剤の分散性や加工性に優れる。しかし、前記少なくとも2個の重合性不飽和基を有する単量体単位や、カルボン酸基、ヒドロキシル基及び/又はエポキシ基を有する単量体単位として開示されている物質は、いずれも低分子である。そのため、反応性が過剰に高く、架橋ゴム粒子及びゴム組成物においてゲル化が進行する恐れがあった。また、この技術は共役ジエン/芳香族ビニル共重合ゴムとは別途架橋ゴムを合成した上で、その架橋ゴムを共役ジエン/芳香族ビニル共重合ゴムと配合することが必須であり、工程の簡易性の観点で、改善が必要である。
特許文献5は、可溶性多官能ビニル芳香族共重合体を開示するが、これを共重合体ゴムの製造に使用することは教えない。
多官能ビニル芳香族共重合体(A)が、ジビニル芳香族化合物に由来する構造単位(a)、モノビニル芳香族化合物に由来する構造単位(b)、及び末端を構成する構造単位(d)を含有し、構造単位(a)を2モル%以上95モル%未満含有し、構造単位(a)の少なくとも一部は下記式で表されるビニル基含有構造単位(a1)であり、
(式中、R1は炭素数6~30の芳香族炭化水素基を示す。)
構造単位(a)の他の少なくとも一部は上記構造単位(d)を構成する構造単位であり、構造単位(a)及び(b)の総和に対するビニル基含有構造単位(a1)モル分率は0.02~0.8の範囲であり、数平均分子量が300~100000であることを特徴とする共重合体ゴムの製造方法である。
(式中、R2は炭素数6~30の芳香族炭化水素基を示す。)
(式中、R3は炭素数6~30の芳香族炭化水素基を示し、R4は水素原子又は炭素数6~30の炭化水素基を示す。)
(式中、Yは未置換又は炭素数1~12の炭化水素基で置換された炭素数6~52の芳香族炭化水素基を示し、Zは炭素数1~30の脂肪族炭化水素基、又は未置換又は炭素数1~12の炭化水素基で置換された炭素数6~52の芳香族炭化水素基を示す。)
構造単位(A1)を0.001~6重量%、構造単位(B1)を29~99.999重量%及び構造単位(C1)を0~70重量%含み、
構造単位(A1)が、ジビニル芳香族化合物に由来する構造単位(a)、モノビニル芳香族化合物に由来する構造単位(b)、及び末端を構成する構造単位(d)を含有し、構造単位(a)を2モル%以上95モル%未満含有し、構造単位(a)の少なくとも一部は上記式で表されるビニル基含有構造単位(a1)であり、構造単位(a)の他の少なくとも一部は上記構造単位(d)を構成する構造単位であり、構造単位(a)及び(b)の総和に対するビニル基含有構造単位(a1)モル分率は0.02~0.8の範囲であることを特徴とする共重合体ゴムである。
更に、この共重合体ゴムにフィラーを含有し、架橋させた架橋ゴム組成物は、フィラーの分散性に優れることから、機械的強度、耐摩耗性に優れる。
多官能ビニル芳香族共重合体(A)の数平均分子量Mnは、300~100000であり、好ましくは500~5000である。また、分子量分布Mw/Mnは3~20の範囲が好ましい。
そして、構造単位(a)の一部は、上記構造単位(a1)として、これを1モル%以上含むことがよく、好ましくは2~80モル%、より好ましくは5~70モル%、更に好ましくは10~60モル%、特に好ましくは15~50モル%である。
更に、構造単位(a)の一部は、末端を構成する構造単位(d)中に、構造単位(d1)として含まれ、その量は一分子当たり0.7個以上、好ましくは1.0個以上、より好ましくは1.5個以上であることがよい。
なお、上記計算において、構造単位(a)は構造単位(a1)と構造単位(d1)を含む。
また、末端の構造単位(d)は一分子当たり平均2.1個以上、好ましくは2.5個以上、より好ましくは3個以上有することがよい。
式(d2)において、R3は炭素数6~30の芳香族炭化水素基を示し、R4は水素原子又は炭素数6~30の炭化水素基を示す。式(d2)は、モノマーとしてのモノビニル芳香族化合物から生じることができる。具体的には、ビニルトルエン、エチルビニルベンゼン、n-プロピルスチレン、イソプロピルスチレン、ブチルスチレン、メチルビニルナフタレン、エチルビニルナフタレン、メチルビニルフェナンスレン、エチルビニルフェナンスレン、メチルビニルアントラセン、エチルビニルアントラセン、メチルビニルビフェニル、エチルビニルビフェニル、メチルビニルトリフェニル、エチルビニルトリフェニル等が挙げられる。好ましくは、多官能ビニル芳香族共重合体(A)のゴム溶液重合における取扱いの容易性、フィラーとの相互作用に優れることから、R3はフェニル基、ナフチル基、ビフェニリル基であり、R4は水素原子または炭素数1~10のアルキル基である。具体的には、ビニルトルエン、エチルビニルベンゼン、n-プロピルスチレン、イソプロピルスチレン、n-ブチルスチレン、イソブチルスチレン、sec-ブチルスチレン、t-ブチルスチレン、メチルビニルナフタレン、エチルビニルナフタレン、メチルビニルビフェニル、エチルビニルビフェニル、等が挙げられる。より好ましくは、ビニルトルエン、エチルビニルベンゼン、イソプロピルスチレン、t-ブチルスチレンである。
酸触媒としては、アルキルスルホン酸、トルエンスルホン酸等のスルホン酸触媒等が使用できる。その使用量は、全モノマー成分の合計100モルに対し、0.1~10モルである。
ルイス酸触媒は、全モノマー成分の合計100モルに対し、ルイス酸触媒を0.001~100モルの範囲内で使用することがよく、より好ましくは0.01~50モルである。最も好ましくは0.1~10モルである。
また、所望により助触媒として、1種以上のルイス塩基化合物を使用することができる。ルイス塩基化合物の具体例としては、酢酸エチル等のエステル系化合物、メチルメルカプトプロピオン酸等のチオエステル系化合物、メチルエチルケトン等のケトン系化合物、メチルアミン等のアミン系化合物、ジエチルエーテルのエーテル系化合物、ジエチルスルフィド等のチオエーテル系化合物、及びトリプロピルホスフィン、トリブチルホスフィンなどのホスフィン系化合物がある。
有機溶媒としては、重合活性、溶解性のバランスの観点からトルエン、キシレン、n-へキサン、シクロへキサン、メチルシクロへキサン又はエチルシクロへキサンが特に好ましい。
例えば、炭化水素溶媒中において、有機アルカリ金属の化合物を開始剤として用いて、多官能ビニル芳香族共重合体、共役ジエン化合物又はこれらと芳香族ビニル化合物をリビングアニオン重合させる方法や、共役ジエン化合物及び芳香族ビニル化合物をリビングアニオン重合させたのち、多官能ビニル芳香族共重合体を加えて反応を行う多段階反応方法がある。また、必要により末端を、官能基を有する重合停止剤で末端変性してもよい。
多官能ビニル芳香族共重合体(A)は、0.001~6重量%、好ましくは0.005~3重量%、更に好ましくは0.01~2重量%であり、共役ジエン化合物(B)は、94~99.999重量%であり、芳香族ビニル化合物(C)は、0~70重量%である。
芳香族ビニル化合物(C)を使用する場合は、多官能ビニル芳香族共重合体(A)は、上記と同様の範囲であり、共役ジエン化合物(B)は50~97重量%、好ましくは55~90重量%であり、芳香族ビニル化合物(C)は、2~50重量%、好ましくは5~45重量%である。
より好ましくは、多官能ビニル芳香族共重合体(A)は、0.01~23重量%、更に好ましくは0.1~1重量%である。共役ジエン化合物(B)は50~97重量%、更に好ましくは55~90重量%である。芳香族ビニル化合物(C)2~49重量%、更に好ましくは5~44重量%の範囲とすることが好ましい。
ジビニル芳香族化合物に由来する構造単位(a)、モノビニル芳香族化合物に由来する構造単位(b)、及び末端を構成する構造単位(d)を含有し、構造単位(a)を2モル%以上95モル%未満含有し、構造単位(a)の少なくとも一部は下記式で表されるビニル基含有構造単位(a1)であり、
(式中、R1は炭素数6~30の芳香族炭化水素基を示す。)
構造単位(a)の他の少なくとも一部は上記構造単位(d)を構成する構造単位であり、構造単位(a)及び(b)の総和に対するビニル基含有構造単位(a1)モル分率は0.02~0.8の範囲であり、数平均分子量が300~100000である。
好ましい範囲は、上記原料の使用割合と同様な割合となることがよい。
シリカとしては、一般的に充填剤として用いられる粒子状のものであればよいが、一次粒子径が50nm以下のものであることが好ましい。このようなシリカの具体例としては、含水ケイ酸、無水ケイ酸、ケイ酸カルシウム、ケイ酸アルミニウムなどが挙げられる。
分子量及び分子量分布測定は、GPC(東ソー製、HLC-8220GPC)を使用し、溶媒にテトラヒドロフラン(THF)、流量1.0ml/min、カラム温度38℃、単分散ポリスチレンによる検量線を用いて行った。
日本電子製JNM-LA600型核磁気共鳴分光装置を用い、13C-NMR及び1H-NMR分析により決定した。溶媒としてクロロホルム-d1を使用し、テトラメチルシランの共鳴線を内部標準として使用した。
末端基の算出は、上記のGPC測定より得られる数平均分子量と13C-NMR及び1H-NMR測定とガスクロマトグラフ(GC)分析の結果より得られるモノマー総量に対する末端基を導入するために使用した誘導体量とから、多官能ビニル芳香族共重合体1分子中に含まれる末端基数を算出した。なお、合成例1においては、末端基はジビニル芳香族化合物又はモノビニル芳香族化合物から生じる。末端基の算出は、13C-NMR及び1H-NMR測定結果に加えて、GC分析より得られる共重合体中に導入された各構造単位の総量に関するデータより、末端に導入された特定の構造単位の導入量を算出し、この末端に導入された特定の構造単位の導入量と上記のGPC測定より得られる数平均分子量とから、多官能ビニル芳香族共重合体1分子中に含まれる特定の構造単位の末端基数を算出した。
JIS K6300‐1に従って、L形ローター、予熱1分、ローター作動時間4分、温度100℃ で求めた。
共重合体ゴムを5倍重量のシクロヘキサンに溶解して溶液をろ過し、フィルター上の残留物の有無を目視確認した。残留物があればゲル状物ありと判定した。
JIS K6251の引張試験法により300%モジュラスを測定し、比較例1で得られた架橋ゴムの測定値を100として、指数化した。指数値が大きいほど引張り強度に優れることを示す。
JIS K6264に準拠したランボーン型摩耗試験機を使用した方法を用い、スリップ率が30%の摩耗量を測定し、比較例1で得られた架橋ゴムの測定値を100として、指数化した。指数値が大きいほど耐摩耗性は良好である。
DVB-810(新日鉄住金化学製、ジビニルベンゼン成分の含有率81.0wt%)320.5mL(ジビニルベンゼン成分1.82モル、エチルビニルベンゼン成分0.43モル)、酢酸n-ブチル 0.28モル(36.9mL)、トルエン 140mLを1.0Lの反応器内に投入し、70℃で40ミリモルのメタンスルホン酸を酢酸n-ブチル 0.12モル(15.7mL)に溶解させた溶液を添加し、6時間反応させた。重合溶液を水酸化カルシウムで停止させた後、活性アルミナをろ過助剤として、ろ過を行った。それから、60℃で減圧脱揮し、可溶性多官能ビニル芳香族共重合体(共重合体X1)22.6gを得た。
得られた共重合体X1のMnは1085、Mwは12400、Mw/Mnは11.4であった。GC分析、GPC測定、FT-IR、13C‐NMR及び1H‐NMR分析により、共重合体Xは、ジビニル芳香族化合物(a)由来の下記ビニル基含有構造単位(a1)及び末端基(d1)、モノビニル芳香族化合物(b)由来の下記末端基(d2)を有することを確認した。
また、共重合体X1は、トルエン、キシレン、THF、ジクロロエタン、ジクロロメタン、クロロホルムに可溶であり、ゲルの生成は認められなかった。
DVB-810 276.4mL、トルエン 86.2mL、アニソール93.74mLを1.0Lの反応器内に投入し、50℃で10ミリモルのメタンスルホン酸をトルエン2mLに溶解させた溶液を添加し、4時間反応させた他は、合成例1と同様の手法で共重合体X2を得た。
得られた共重合体X2のMnは1000、Mwは18800、Mw/Mnは18.8であった。GC分析、GPC測定、FT-IR、13C‐NMR及び1H‐NMR分析により、共重合体X2は、ジビニル芳香族化合物(a)由来の合成例1と同様な上記ビニル基含有構造単位(a1)、およびアニソール由来の下記末端基(d3)を有することを確認した。
DVB-810 300mL、トルエン 177.6mL、メタクリル酸t-ブチル145.6mL、60℃で10ミリモルのメタンスルホン酸をトルエン2mLに溶解させた溶液を添加し、4時間反応させた他は、合成例1と同様の手法で共重合体X3を得た。
得られた共重合体X3のMnは2190、Mwは13600、Mw/Mnは7.2であった。GC分析、GPC測定、FT-IR、13C‐NMR及び1H‐NMR分析により、共重合体X3は、ジビニル芳香族化合物(a)由来の合成例1と同様な上記ビニル基含有構造単位(a1)、並びにメタクリル酸t-ブチル由来の末端基(d4)(R5、R6、R7=CH3)及び末端基 (d5)(R8=CH3)を有することを確認した。
また、共重合体X3は、トルエン、キシレン、THF、ジクロロエタン、ジクロロメタン、クロロホルムに可溶であり、ゲルの生成は認められなかった。
窒素置換された内容積0.5リットルのオートクレーブ反応器に、シクロヘキサン245g、THF2.5g、スチレン10g、1,3-ブタジエン40g、合成例1で得られた共重合体X1 0.015gを加えた。25℃において、sec-ブチルリチウム50mgを含むシクロヘキサン溶液5gを添加して重合を開始した。重合熱により反応溶液の温度が上昇し、最高温度は85℃に達した。
重合転化率が99%に達したのを確認後、イソプロパノールを50mg添加して重合停止し、反応溶液に、2,6-ジ-tert-ブチル-p-クレゾール(BHT)を添加した。次いで、スチームストリッピングにより脱溶媒を行い、共重合体ゴムAを得た。得られた共重合体ゴムAの物性を表1に示す。
共重合体ゴムA、プロセスオイル、カーボンブラック、酸化亜鉛、ステアリン酸及び老化防止剤を、ラボプラストミルを用い、155℃、60rpmで4分間混練した。
上記混練で得られた混練物に、硫黄と加硫促進剤を加え、ラボプラストミルを用い、70℃、60rpmで1分間混練し、加硫して架橋ゴムAを得た。
各添加物の配合割合を表2に示す。また、架橋ゴムAの物性を表3に示す。
共重合体X1の代わりに、共重合体X2、共重合体X3をそれぞれ使用した他は、実施例1と同様の手法で、共重合体ゴムC、共重合体ゴムDを得た。得られた物性を表1に示す。
共重合体ゴムAの代わりに、共重合体ゴムC、共重合体ゴムDをそれぞれ使用した他は、実施例2と同様の手法で、架橋ゴムC、架橋ゴムDを得た。得られた物性を表3に示す。
実施例1において、合成例1にて合成した共重合体X1の代わりにDVB-810を、ジビニルベンゼン換算にて0.005g使用した以外は、実施例1と同様の手法で共重合体ゴムBを得た。得られた共重合体ゴムBの物性を表1に示す。
さらに、共重合体ゴムBを用いて、実施例1と同様の手法及び各添加物の配合割合で架橋ゴムBを得た。また、架橋ゴムBの物性を表3に示す。
ゴム配合油:出光興産製 ダイナプロセスオイル AC-12
硫黄:鶴見化学工業製 粉末硫黄
酸化亜鉛:三井金属鉱業製 亜鉛華1号
ステアリン酸:日油製
カーボンブラック:新日化カーボン製 ニテロン#300
加硫促進剤:N-tert-ブチルベンゾチアゾール-2-スルフェンアミド
老化防止剤:大内新興化学工業製 ノクセラーNS
配合ゴム
Claims (10)
- 多官能ビニル芳香族共重合体(A)、及び共役ジエン化合物(B)を含む原料を共重合させて共重合体ゴムを製造するに当たり、
多官能ビニル芳香族共重合体(A)が、ジビニル芳香族化合物に由来する構造単位(a)、モノビニル芳香族化合物に由来する構造単位(b)、及び末端を構成する構造単位(d)を含有し、構造単位(a)を2モル%以上95モル%未満含有し、構造単位(a)の少なくとも一部は下記式で表されるビニル基含有構造単位(a1)であり、
(式中、R1は炭素数6~30の芳香族炭化水素基を示す。)
構造単位(a)の他の少なくとも一部は上記構造単位(d)を構成する構造単位であり、構造単位(a)及び(b)の総和に対するビニル基含有構造単位(a1)モル分率は0.02~0.8の範囲であり、数平均分子量が300~100000であることを特徴とする共重合体ゴムの製造方法。 - 多官能ビニル芳香族共重合体(A)、共役ジエン化合物(B)及び芳香族ビニル化合物(C)を含む原料を共重合させて共重合体ゴムを製造することを特徴とする請求項1に記載の共重合体ゴムの製造方法。
- 末端を構成する構造単位(d)が、下記(d1)~(d6)で表される単位のいずれかである(ただし、(d4)又は(d5)で表される単位の場合は、(d4)又は(d5)で表される単位を同時に有する。)請求項1又は2に記載の共重合体ゴムの製造方法。
(式中、R2は炭素数6~30の芳香族炭化水素基を示す。)
(式中、R3は炭素数6~30の芳香族炭化水素基を示し、R4は水素原子又は炭素数6~30の炭化水素基を示す。)
―Y-O-Z (d3)
(式中、Yは未置換又は炭素数1~12の炭化水素基で置換された炭素数6~52の芳香族炭化水素基を示し、Zは炭素数1~30の脂肪族炭化水素基、又は未置換又は炭素数1~12の炭化水素基で置換された炭素数6~52の芳香族炭化水素基を示す。)
(ここで、R5は水素原子又は炭素数1~18の炭化水素基を示し、R6及びR7は炭素数1~18の炭化水素基を示し、R8は水素原子又はメチル基を示す。)
(ここで、R9は1または2以上の脂環式炭化水素環を有する炭素数4~16の炭化水素基である。) - アニオン重合によって共重合することを特徴とする請求項1~3のいずれかに記載の共重合体ゴムの製造方法。
- 請求項1~4のいずれかに記載の共重合体ゴムの製造方法で得られた共重合体ゴムに、フィラーを配合し、加硫により架橋させてゴム組成物を得ることを特徴とする架橋ゴム組成物の製造方法。
- 多官能ビニル芳香族共重合体の構造単位(A1)と、共役ジエン化合物の構造単位(B1)又は共役ジエン化合物の構造単位(B1)と芳香族ビニル化合物の構造単位(C1)を含有する共重合体ゴムであって、
構造単位(A1)を0.001~6重量%、構造単位(B1)を29~99.999重量%及び構造単位(C1)を0~70重量%含み、
構造単位(A1)が、
ジビニル芳香族化合物に由来する構造単位(a)、モノビニル芳香族化合物に由来する構造単位(b)、及び末端を構成する構造単位(d)を含有し、構造単位(a)を2モル%以上95モル%未満含有し、構造単位(a)の少なくとも一部は下記式で表されるビニル基含有構造単位(a1)であり、
(式中、R1は炭素数6~30の芳香族炭化水素基を示す。)
構造単位(a)の他の少なくとも一部は上記構造単位(d)を構成する構造単位であり、構造単位(a)及び(b)の総和に対するビニル基含有構造単位(a1)モル分率は0.02~0.8の範囲であり、数平均分子量が300~100000であることを特徴とする共重合体ゴム。 - 構造単位(A1)の数平均分子量が300~100000である請求項6に記載の共重合体ゴム。
- 末端を構成する構造単位(d)が、下記(d1)~(d6)で表される単位のいずれかである(ただし、(d4)又は(d5)で表される単位の場合は、(d4)又は(d5)で表される単位を同時に有する。)請求項6又は7に記載の共重合体ゴム。
(式中、R2は炭素数6~30の芳香族炭化水素基を示す。)
(式中、R3は炭素数6~30の芳香族炭化水素基を示し、R4は水素原子又は炭素数6~30の炭化水素基を示す。)
―Y-O-Z (d3)
(式中、Yは未置換又は炭素数1~12の炭化水素基で置換された炭素数6~52の芳香族炭化水素基を示し、Zは炭素数1~30の脂肪族炭化水素基、又は未置換又は炭素数1~12の炭化水素基で置換された炭素数6~52の芳香族炭化水素基を示す。)
(ここで、R5は水素原子又は炭素数1~18の炭化水素基を示し、R6及びR7は炭素数1~18の炭化水素基を示し、R8は水素原子又はメチル基を示す。)
(ここで、R9は1または2以上の脂環式炭化水素環を有する炭素数4~16の炭化水素基である。) - 請求項6~8のいずれかに記載の共重合体ゴムと、フィラーを含有し、該共重合体ゴムが架橋構造を有することを特徴とする架橋ゴム組成物。
- 請求項6~8のいずれかに記載の共重合体ゴムに使用することを特徴とする多官能ビニル芳香族共重合体。
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2018549003A JP7079204B2 (ja) | 2016-11-01 | 2017-10-31 | 共重合体ゴム及びその製造方法、並びに架橋ゴム組成物 |
| EP17866778.8A EP3536723B1 (en) | 2016-11-01 | 2017-10-31 | Copolymer rubber and method for producing same, and crosslinked rubber composition |
| KR1020197013922A KR102494998B1 (ko) | 2016-11-01 | 2017-10-31 | 공중합체 고무 및 그 제조 방법, 그리고 가교 고무 조성물 |
| CN201780068061.3A CN109906238B (zh) | 2016-11-01 | 2017-10-31 | 共聚物橡胶及其制造方法、以及交联橡胶组合物及其制法 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2016-214717 | 2016-11-01 | ||
| JP2016214717 | 2016-11-01 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2018084128A1 true WO2018084128A1 (ja) | 2018-05-11 |
Family
ID=62075799
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2017/039254 WO2018084128A1 (ja) | 2016-11-01 | 2017-10-31 | 共重合体ゴム及びその製造方法、並びに架橋ゴム組成物 |
Country Status (5)
| Country | Link |
|---|---|
| EP (1) | EP3536723B1 (ja) |
| JP (1) | JP7079204B2 (ja) |
| KR (1) | KR102494998B1 (ja) |
| CN (1) | CN109906238B (ja) |
| WO (1) | WO2018084128A1 (ja) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2019065662A1 (ja) * | 2017-09-29 | 2019-04-04 | 日鉄ケミカル&マテリアル株式会社 | 共重合体ゴム及びその製造方法、並びに架橋ゴム組成物 |
| WO2020067336A1 (ja) | 2018-09-28 | 2020-04-02 | 日鉄ケミカル&マテリアル株式会社 | 多官能ビニル芳香族共重合体及びその製造方法、それから得られる共重合体ゴム、ゴム組成物、ゴム架橋物及びタイヤ部材 |
| WO2020262371A1 (ja) | 2019-06-25 | 2020-12-30 | 日鉄ケミカル&マテリアル株式会社 | 変性ビニル芳香族系共重合体及びその製造方法、それから得られる変性共役ジエン系共重合体、その組成物、ゴム架橋物及びタイヤ部材 |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR102537249B1 (ko) * | 2017-03-30 | 2023-05-26 | 닛테츠 케미컬 앤드 머티리얼 가부시키가이샤 | 가용성 다관능 비닐 방향족 공중합체, 그 제조 방법, 경화성 수지 조성물 및 그 경화물 |
Citations (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2002000779A1 (fr) | 2000-06-29 | 2002-01-03 | Jsr Corporation | Composition de caoutchouc |
| JP2003073434A (ja) | 2001-06-22 | 2003-03-12 | Kuraray Co Ltd | ブロック共重合体の製造方法 |
| JP2004123873A (ja) | 2002-10-01 | 2004-04-22 | Nippon Steel Chem Co Ltd | 可溶性多官能ビニル芳香族共重合体及びその重合方法 |
| JP2004517202A (ja) | 2000-10-19 | 2004-06-10 | チャイナ・ペトロリアム・アンド・ケミカル・コーポレーション | 星形−ブロックインターポリマー及びそれらの製造 |
| JP2005213381A (ja) | 2004-01-29 | 2005-08-11 | Toyo Tire & Rubber Co Ltd | ゴム組成物 |
| JP2009544824A (ja) * | 2006-07-28 | 2009-12-17 | 株式会社ブリヂストン | 相間領域を有する重合体コア−シェルナノ粒子 |
| JP2010511071A (ja) * | 2006-11-23 | 2010-04-08 | エルジー・ケム・リミテッド | 高分子ブラシを有する微粒子及びその製造方法 |
| JP2013100433A (ja) * | 2011-11-09 | 2013-05-23 | Nippon Steel & Sumikin Chemical Co Ltd | スチレン−メタクリル酸系共重合樹脂組成物の製造方法およびその組成物 |
| WO2016104748A1 (ja) * | 2014-12-26 | 2016-06-30 | 新日鉄住金化学株式会社 | 末端変性可溶性多官能ビニル芳香族共重合体、硬化性樹脂組成物及びこれを用いた光導波路 |
Family Cites Families (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3661873A (en) * | 1971-01-06 | 1972-05-09 | Firestone Tire & Rubber Co | Process and product of treating live polymers with divinyl benzene and a haloalkane |
| US20060177666A1 (en) * | 2005-02-08 | 2006-08-10 | Masanao Kawabe | Curable resin compositions |
| WO2008123199A1 (ja) * | 2007-03-26 | 2008-10-16 | Nippon Steel Chemical Co., Ltd. | 可溶性多官能ビニル芳香族共重合体及びその製造方法 |
| WO2013069077A1 (ja) * | 2011-11-07 | 2013-05-16 | 新日鉄住金化学株式会社 | 高分岐型超高分子量体を含有するスチレン系樹脂組成物の製造方法およびその組成物 |
-
2017
- 2017-10-31 WO PCT/JP2017/039254 patent/WO2018084128A1/ja active Search and Examination
- 2017-10-31 CN CN201780068061.3A patent/CN109906238B/zh active Active
- 2017-10-31 JP JP2018549003A patent/JP7079204B2/ja active Active
- 2017-10-31 EP EP17866778.8A patent/EP3536723B1/en active Active
- 2017-10-31 KR KR1020197013922A patent/KR102494998B1/ko active IP Right Grant
Patent Citations (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2002000779A1 (fr) | 2000-06-29 | 2002-01-03 | Jsr Corporation | Composition de caoutchouc |
| JP2004517202A (ja) | 2000-10-19 | 2004-06-10 | チャイナ・ペトロリアム・アンド・ケミカル・コーポレーション | 星形−ブロックインターポリマー及びそれらの製造 |
| JP2003073434A (ja) | 2001-06-22 | 2003-03-12 | Kuraray Co Ltd | ブロック共重合体の製造方法 |
| JP2004123873A (ja) | 2002-10-01 | 2004-04-22 | Nippon Steel Chem Co Ltd | 可溶性多官能ビニル芳香族共重合体及びその重合方法 |
| JP2005213381A (ja) | 2004-01-29 | 2005-08-11 | Toyo Tire & Rubber Co Ltd | ゴム組成物 |
| JP2009544824A (ja) * | 2006-07-28 | 2009-12-17 | 株式会社ブリヂストン | 相間領域を有する重合体コア−シェルナノ粒子 |
| JP2010511071A (ja) * | 2006-11-23 | 2010-04-08 | エルジー・ケム・リミテッド | 高分子ブラシを有する微粒子及びその製造方法 |
| JP2013100433A (ja) * | 2011-11-09 | 2013-05-23 | Nippon Steel & Sumikin Chemical Co Ltd | スチレン−メタクリル酸系共重合樹脂組成物の製造方法およびその組成物 |
| WO2016104748A1 (ja) * | 2014-12-26 | 2016-06-30 | 新日鉄住金化学株式会社 | 末端変性可溶性多官能ビニル芳香族共重合体、硬化性樹脂組成物及びこれを用いた光導波路 |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP3536723A4 |
Cited By (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2019065662A1 (ja) * | 2017-09-29 | 2019-04-04 | 日鉄ケミカル&マテリアル株式会社 | 共重合体ゴム及びその製造方法、並びに架橋ゴム組成物 |
| JPWO2019065662A1 (ja) * | 2017-09-29 | 2020-10-22 | 日鉄ケミカル&マテリアル株式会社 | 共重合体ゴム及びその製造方法、並びに架橋ゴム組成物 |
| JP7144430B2 (ja) | 2017-09-29 | 2022-09-29 | 日鉄ケミカル&マテリアル株式会社 | 共重合体ゴム及びその製造方法、並びに架橋ゴム組成物 |
| WO2020067336A1 (ja) | 2018-09-28 | 2020-04-02 | 日鉄ケミカル&マテリアル株式会社 | 多官能ビニル芳香族共重合体及びその製造方法、それから得られる共重合体ゴム、ゴム組成物、ゴム架橋物及びタイヤ部材 |
| CN112771089A (zh) * | 2018-09-28 | 2021-05-07 | 日铁化学材料株式会社 | 多官能乙烯基芳香族共聚物及其制造方法、由其获得的共聚物橡胶、橡胶组合物、橡胶交联物和轮胎构件 |
| JPWO2020067336A1 (ja) * | 2018-09-28 | 2021-09-02 | 日鉄ケミカル&マテリアル株式会社 | 多官能ビニル芳香族共重合体及びその製造方法、それから得られる共重合体ゴム、ゴム組成物、ゴム架橋物及びタイヤ部材 |
| CN114933676A (zh) * | 2018-09-28 | 2022-08-23 | 日铁化学材料株式会社 | 多官能乙烯基芳香族共聚物及其制法、共轭二烯系共聚物及其组合物、交联物、轮胎构件 |
| CN112771089B (zh) * | 2018-09-28 | 2022-11-01 | 日铁化学材料株式会社 | 多官能乙烯基芳香族共聚物及其制法、共轭二烯系共聚物及其组合物、交联物、轮胎构件 |
| JP7472027B2 (ja) | 2018-09-28 | 2024-04-22 | 日鉄ケミカル&マテリアル株式会社 | 多官能ビニル芳香族共重合体及びその製造方法、それから得られる共重合体ゴム、ゴム組成物、ゴム架橋物及びタイヤ部材 |
| WO2020262371A1 (ja) | 2019-06-25 | 2020-12-30 | 日鉄ケミカル&マテリアル株式会社 | 変性ビニル芳香族系共重合体及びその製造方法、それから得られる変性共役ジエン系共重合体、その組成物、ゴム架橋物及びタイヤ部材 |
Also Published As
| Publication number | Publication date |
|---|---|
| CN109906238A (zh) | 2019-06-18 |
| KR102494998B1 (ko) | 2023-02-02 |
| CN109906238B (zh) | 2021-09-28 |
| EP3536723A4 (en) | 2020-04-29 |
| EP3536723B1 (en) | 2023-01-25 |
| EP3536723A1 (en) | 2019-09-11 |
| KR20190077408A (ko) | 2019-07-03 |
| JP7079204B2 (ja) | 2022-06-01 |
| JPWO2018084128A1 (ja) | 2019-09-26 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6230736B2 (ja) | 分枝共役ジエン共重合体、ゴム組成物および空気入りタイヤ | |
| JP6639909B2 (ja) | タイヤ用部材 | |
| JP7112406B2 (ja) | タイヤ用ゴム組成物 | |
| JP7458146B2 (ja) | ゴム組成物 | |
| TWI770259B (zh) | 重負載輪胎用橡膠組成物及輪胎 | |
| KR102494998B1 (ko) | 공중합체 고무 및 그 제조 방법, 그리고 가교 고무 조성물 | |
| JP7112408B2 (ja) | 重荷重タイヤ用ゴム組成物およびタイヤ | |
| JP7112405B2 (ja) | タイヤ用ゴム組成物 | |
| WO2012086496A1 (ja) | 共役ジエン系ゴム、ゴム組成物、ゴム架橋物、およびタイヤ | |
| WO2019044891A1 (ja) | 高グリップタイヤ用ゴム組成物 | |
| JP2012251118A (ja) | 共役ジエン系エラストマーおよびその製造方法並びにゴム組成物、ゴム弾性体およびタイヤ | |
| WO2019044890A1 (ja) | 高グリップタイヤ用ゴム組成物 | |
| JP2019094390A (ja) | 変性共役ジエン系重合体の製造方法、重合体組成物、架橋体及びタイヤ | |
| KR20220058896A (ko) | 중합체 조성물, 가교체 및 타이어 | |
| JP4057123B2 (ja) | 重合体の製造方法、重合体及びその重合体を用いたゴム組成物 | |
| JP2018526520A (ja) | 官能化ポリマーの調製方法 | |
| JP2018145239A (ja) | タイヤ用ゴム組成物及び空気入りタイヤ | |
| JP7144430B2 (ja) | 共重合体ゴム及びその製造方法、並びに架橋ゴム組成物 | |
| WO2020158678A1 (ja) | ゴム組成物、架橋体及びタイヤ | |
| JP2020196803A (ja) | 変性共役ジエン系重合体の製造方法、変性共役ジエン系重合体、重合体組成物、架橋体及びタイヤ | |
| JP7140953B2 (ja) | 重合体組成物及びタイヤ | |
| US20240002642A1 (en) | Rubber bale, production method therefor, polymer composition, crosslinked object, and tire | |
| US20230312889A1 (en) | Polymer composition, method for producing same, formulation, crosslinked product, and tire | |
| KR100576592B1 (ko) | 변성 고무, 이의 제조 방법 및 그의 조성물 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| DPE1 | Request for preliminary examination filed after expiration of 19th month from priority date (pct application filed from 20040101) | ||
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 17866778 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2018549003 Country of ref document: JP Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 20197013922 Country of ref document: KR Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 2017866778 Country of ref document: EP Effective date: 20190603 |