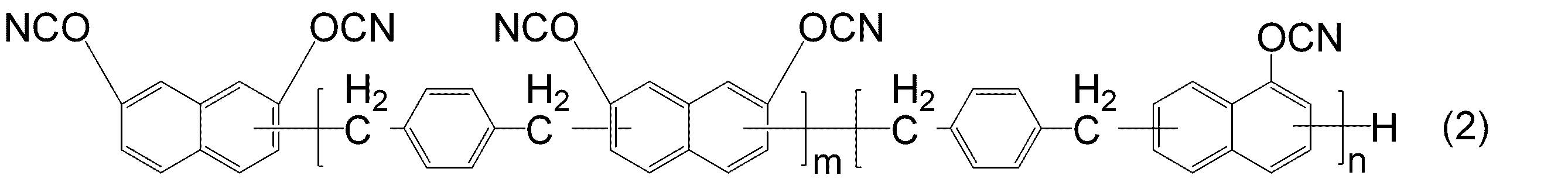
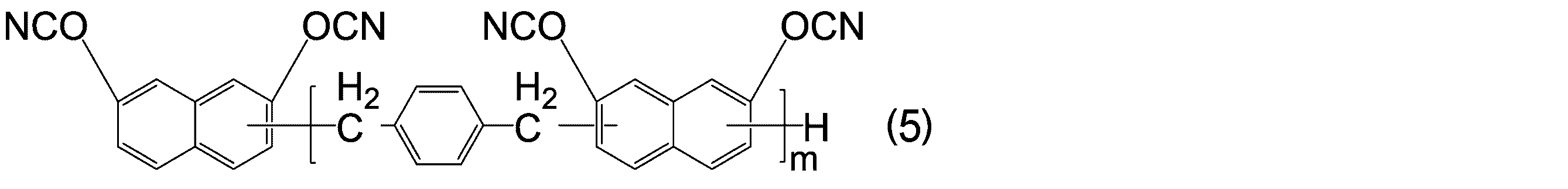
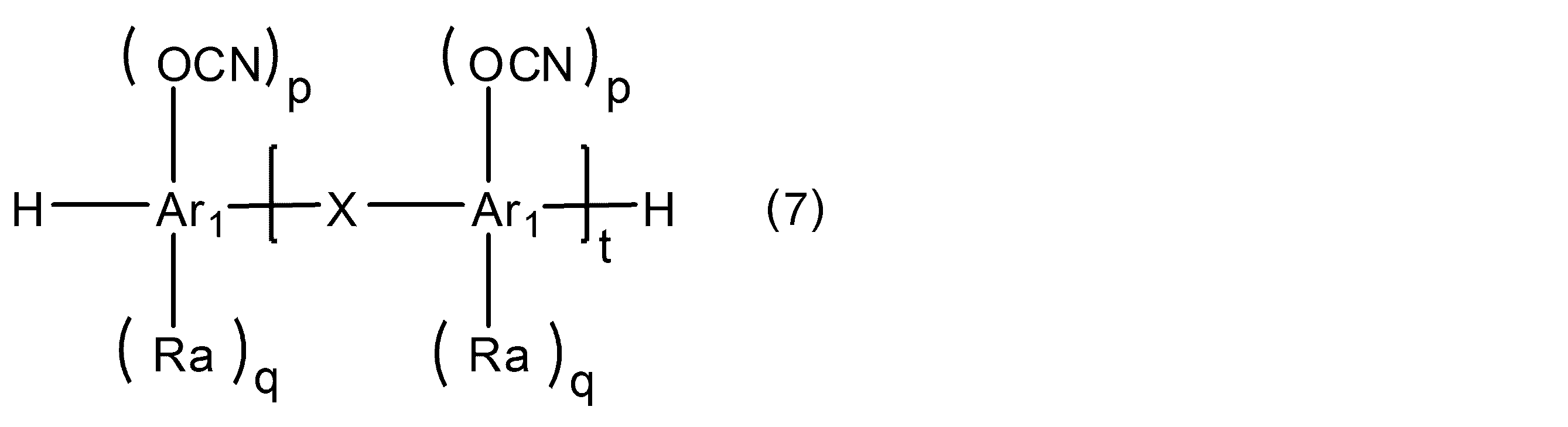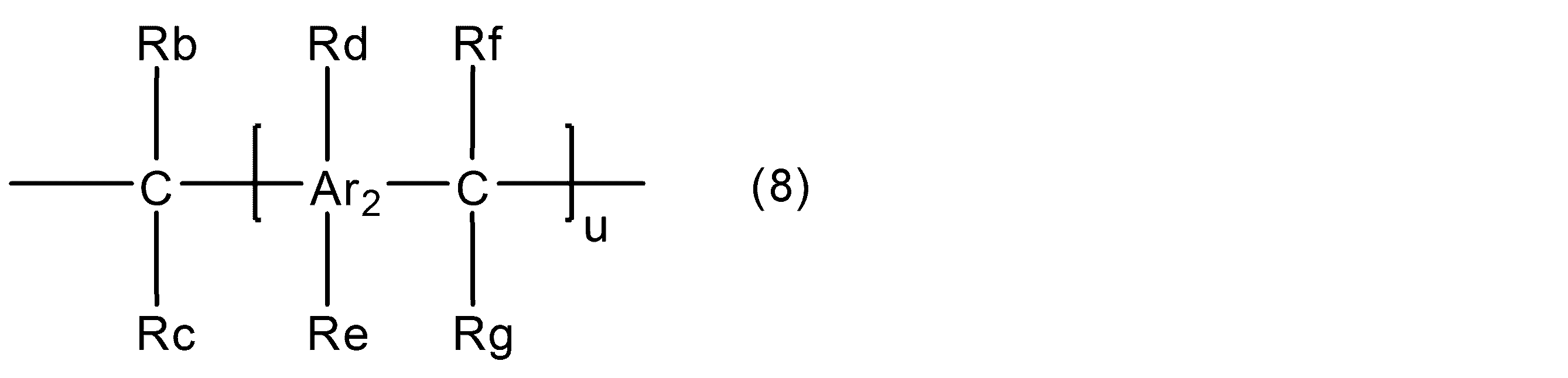WO2016158066A1 - シアン酸エステル化合物、該化合物を含む硬化性樹脂組成物及びその硬化物 - Google Patents
シアン酸エステル化合物、該化合物を含む硬化性樹脂組成物及びその硬化物 Download PDFInfo
- Publication number
- WO2016158066A1 WO2016158066A1 PCT/JP2016/054850 JP2016054850W WO2016158066A1 WO 2016158066 A1 WO2016158066 A1 WO 2016158066A1 JP 2016054850 W JP2016054850 W JP 2016054850W WO 2016158066 A1 WO2016158066 A1 WO 2016158066A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- group
- ester compound
- cyanate ester
- bis
- compound
- Prior art date
Links
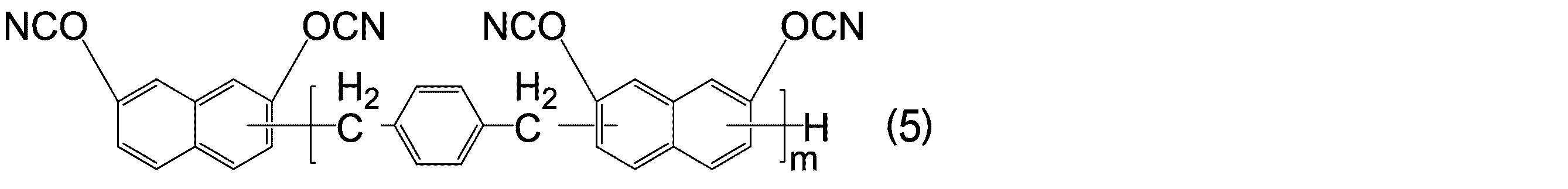
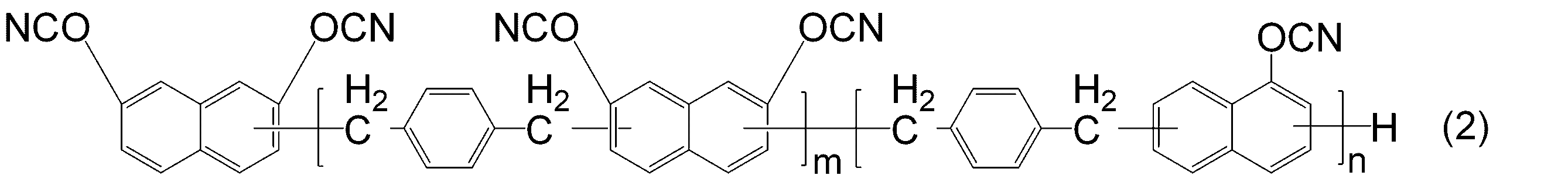
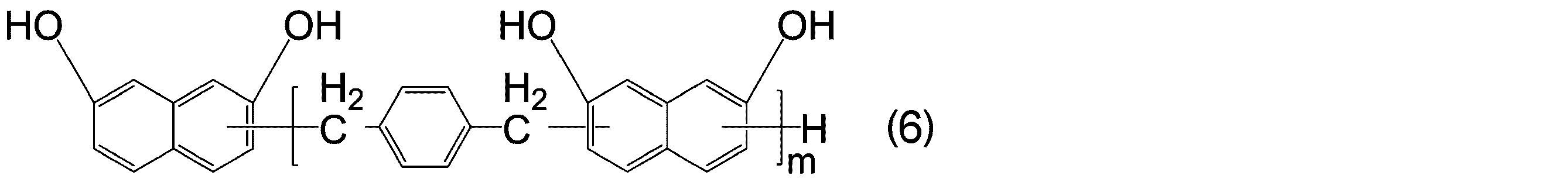
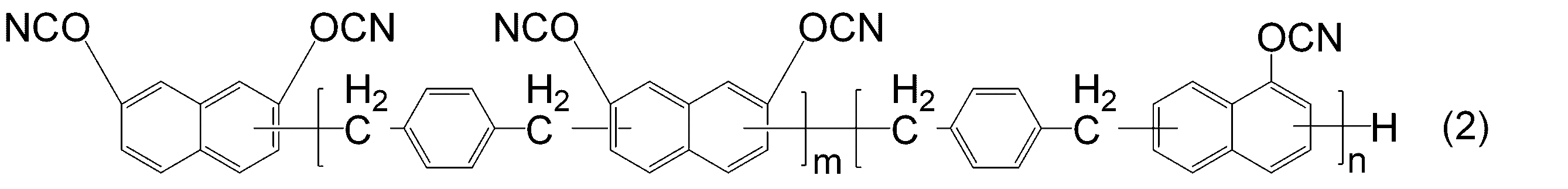
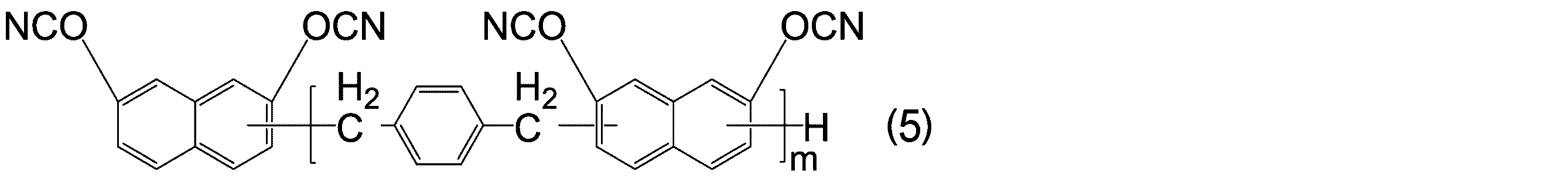
- -1 Cyanic acid ester compound Chemical class 0.000 title claims abstract description 117
- 150000001875 compounds Chemical class 0.000 title claims description 148
- 239000011342 resin composition Substances 0.000 title claims description 56
- XLJMAIOERFSOGZ-UHFFFAOYSA-N anhydrous cyanic acid Natural products OC#N XLJMAIOERFSOGZ-UHFFFAOYSA-N 0.000 title abstract description 5
- 229920005989 resin Polymers 0.000 claims abstract description 110
- 239000011347 resin Substances 0.000 claims abstract description 110
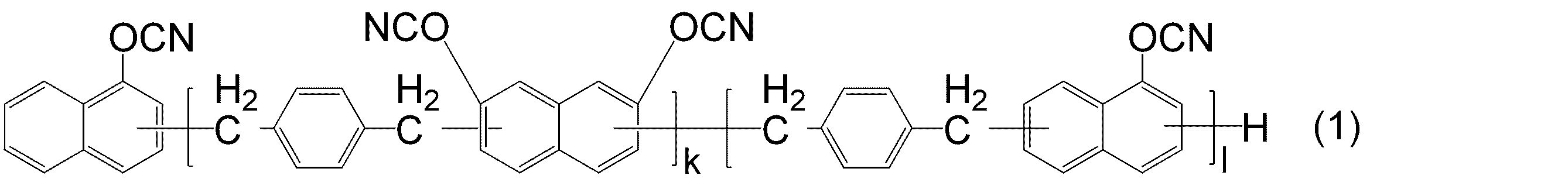
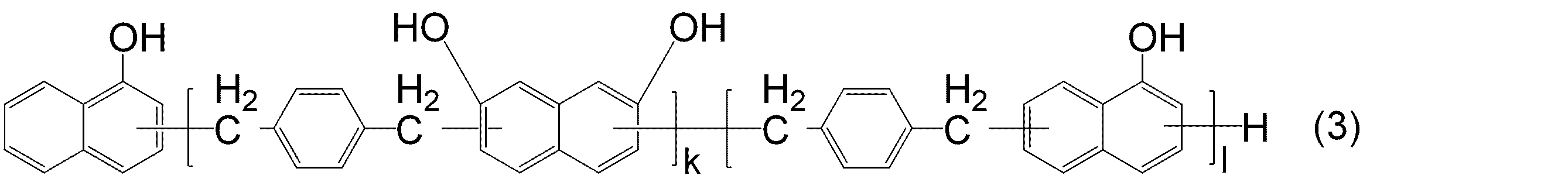
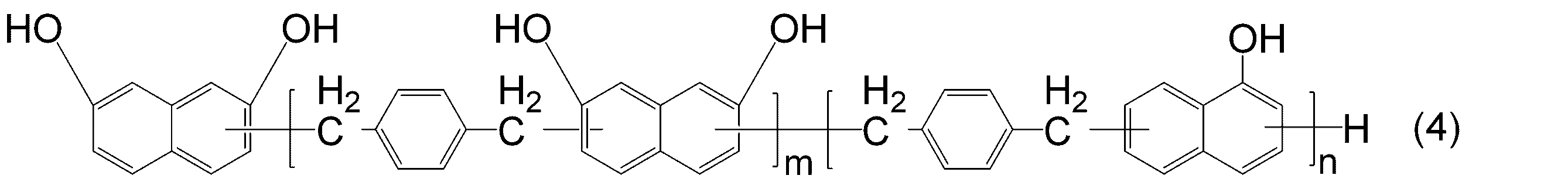
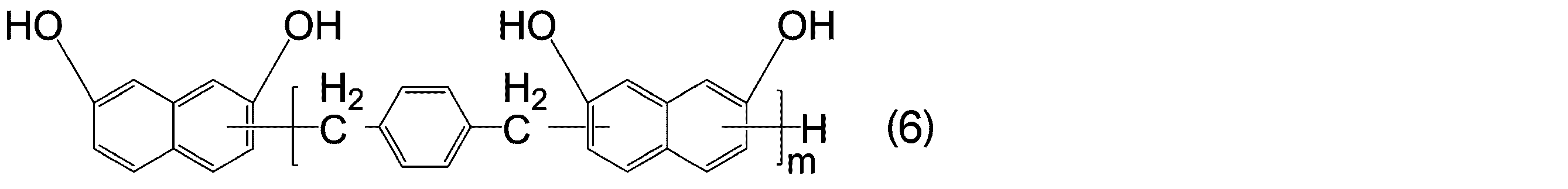
- KJCVRFUGPWSIIH-UHFFFAOYSA-N 1-naphthol Chemical compound C1=CC=C2C(O)=CC=CC2=C1 KJCVRFUGPWSIIH-UHFFFAOYSA-N 0.000 claims abstract description 32
- 239000004643 cyanate ester Substances 0.000 claims description 97
- 239000000463 material Substances 0.000 claims description 38
- 239000000203 mixture Substances 0.000 claims description 32
- 239000003822 epoxy resin Substances 0.000 claims description 30
- 239000005011 phenolic resin Substances 0.000 claims description 30
- 229920000647 polyepoxide Polymers 0.000 claims description 30
- 239000003054 catalyst Substances 0.000 claims description 29
- 230000002378 acidificating effect Effects 0.000 claims description 21
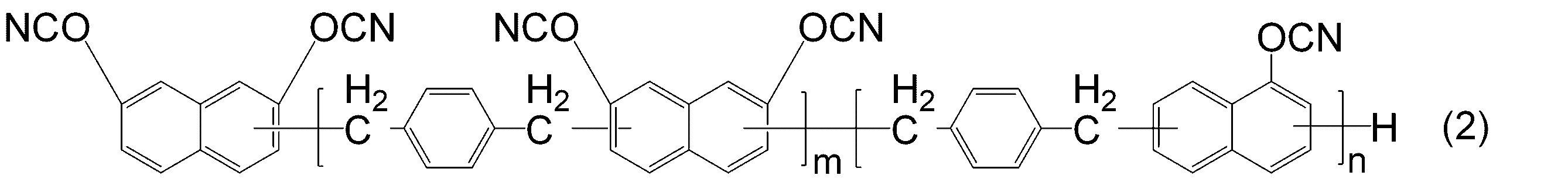
- DFQICHCWIIJABH-UHFFFAOYSA-N naphthalene-2,7-diol Chemical compound C1=CC(O)=CC2=CC(O)=CC=C21 DFQICHCWIIJABH-UHFFFAOYSA-N 0.000 claims description 20
- DAJPMKAQEUGECW-UHFFFAOYSA-N 1,4-bis(methoxymethyl)benzene Chemical compound COCC1=CC=C(COC)C=C1 DAJPMKAQEUGECW-UHFFFAOYSA-N 0.000 claims description 14
- LYCAIKOWRPUZTN-UHFFFAOYSA-N ethylene glycol Natural products OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 claims description 12
- WGCNASOHLSPBMP-UHFFFAOYSA-N hydroxyacetaldehyde Natural products OCC=O WGCNASOHLSPBMP-UHFFFAOYSA-N 0.000 claims description 12
- URLKBWYHVLBVBO-UHFFFAOYSA-N p-dimethylbenzene Natural products CC1=CC=C(C)C=C1 URLKBWYHVLBVBO-UHFFFAOYSA-N 0.000 claims description 12
- 239000000853 adhesive Substances 0.000 claims description 11
- 230000001070 adhesive effect Effects 0.000 claims description 11
- 239000000758 substrate Substances 0.000 claims description 11
- AHHWIHXENZJRFG-UHFFFAOYSA-N oxetane Chemical compound C1COC1 AHHWIHXENZJRFG-UHFFFAOYSA-N 0.000 claims description 10
- 239000003566 sealing material Substances 0.000 claims description 9
- 239000003733 fiber-reinforced composite Substances 0.000 claims description 6
- 239000002904 solvent Substances 0.000 abstract description 68
- 238000000034 method Methods 0.000 description 65
- 238000006243 chemical reaction Methods 0.000 description 55
- 239000000243 solution Substances 0.000 description 52
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 39
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 38
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 36
- 239000000047 product Substances 0.000 description 34
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 33
- ISWSIDIOOBJBQZ-UHFFFAOYSA-N phenol group Chemical group C1(=CC=CC=C1)O ISWSIDIOOBJBQZ-UHFFFAOYSA-N 0.000 description 29
- 239000011521 glass Substances 0.000 description 27
- 125000004093 cyano group Chemical group *C#N 0.000 description 26
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 24
- 150000007514 bases Chemical class 0.000 description 22
- JMANVNJQNLATNU-UHFFFAOYSA-N glycolonitrile Natural products N#CC#N JMANVNJQNLATNU-UHFFFAOYSA-N 0.000 description 22
- 125000004432 carbon atom Chemical group C* 0.000 description 19
- 239000000835 fiber Substances 0.000 description 19
- ZWEHNKRNPOVVGH-UHFFFAOYSA-N 2-Butanone Chemical compound CCC(C)=O ZWEHNKRNPOVVGH-UHFFFAOYSA-N 0.000 description 18
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 17
- 239000002585 base Substances 0.000 description 17
- 229920003986 novolac Polymers 0.000 description 16
- 238000005406 washing Methods 0.000 description 16
- 125000003118 aryl group Chemical group 0.000 description 15
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 14
- IISBACLAFKSPIT-UHFFFAOYSA-N bisphenol A Chemical compound C=1C=C(O)C=CC=1C(C)(C)C1=CC=C(O)C=C1 IISBACLAFKSPIT-UHFFFAOYSA-N 0.000 description 14
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 12
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 12
- NIXOWILDQLNWCW-UHFFFAOYSA-M Acrylate Chemical compound [O-]C(=O)C=C NIXOWILDQLNWCW-UHFFFAOYSA-M 0.000 description 11
- 125000000217 alkyl group Chemical group 0.000 description 11
- 230000015572 biosynthetic process Effects 0.000 description 11
- 229920001002 functional polymer Polymers 0.000 description 11
- 238000004519 manufacturing process Methods 0.000 description 11
- 239000002861 polymer material Substances 0.000 description 11
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 10
- 239000002253 acid Substances 0.000 description 10
- LNEPOXFFQSENCJ-UHFFFAOYSA-N haloperidol Chemical compound C1CC(O)(C=2C=CC(Cl)=CC=2)CCN1CCCC(=O)C1=CC=C(F)C=C1 LNEPOXFFQSENCJ-UHFFFAOYSA-N 0.000 description 10
- 125000004435 hydrogen atom Chemical group [H]* 0.000 description 10
- 229920001568 phenolic resin Polymers 0.000 description 10
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 9
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 9
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 9
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 9
- ZUOUZKKEUPVFJK-UHFFFAOYSA-N diphenyl Chemical compound C1=CC=CC=C1C1=CC=CC=C1 ZUOUZKKEUPVFJK-UHFFFAOYSA-N 0.000 description 9
- 239000012074 organic phase Substances 0.000 description 9
- 238000003786 synthesis reaction Methods 0.000 description 9
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 8
- NTIZESTWPVYFNL-UHFFFAOYSA-N Methyl isobutyl ketone Chemical compound CC(C)CC(C)=O NTIZESTWPVYFNL-UHFFFAOYSA-N 0.000 description 8
- UIHCLUNTQKBZGK-UHFFFAOYSA-N Methyl isobutyl ketone Natural products CCC(C)C(C)=O UIHCLUNTQKBZGK-UHFFFAOYSA-N 0.000 description 8
- JGFZNNIVVJXRND-UHFFFAOYSA-N N,N-Diisopropylethylamine (DIPEA) Chemical compound CCN(C(C)C)C(C)C JGFZNNIVVJXRND-UHFFFAOYSA-N 0.000 description 8
- 238000010521 absorption reaction Methods 0.000 description 8
- 239000000654 additive Substances 0.000 description 8
- XLJMAIOERFSOGZ-UHFFFAOYSA-M cyanate Chemical compound [O-]C#N XLJMAIOERFSOGZ-UHFFFAOYSA-M 0.000 description 8
- 238000007333 cyanation reaction Methods 0.000 description 8
- 239000011256 inorganic filler Substances 0.000 description 8
- 229910003475 inorganic filler Inorganic materials 0.000 description 8
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical compound CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 description 8
- 125000000962 organic group Chemical group 0.000 description 8
- 239000003960 organic solvent Substances 0.000 description 8
- 239000011541 reaction mixture Substances 0.000 description 8
- 239000004065 semiconductor Substances 0.000 description 8
- 238000003756 stirring Methods 0.000 description 8
- GETQZCLCWQTVFV-UHFFFAOYSA-N trimethylamine Chemical compound CN(C)C GETQZCLCWQTVFV-UHFFFAOYSA-N 0.000 description 8
- 239000002966 varnish Substances 0.000 description 8
- KXGFMDJXCMQABM-UHFFFAOYSA-N 2-methoxy-6-methylphenol Chemical compound [CH]OC1=CC=CC([CH])=C1O KXGFMDJXCMQABM-UHFFFAOYSA-N 0.000 description 7
- 239000007788 liquid Substances 0.000 description 7
- 230000007774 longterm Effects 0.000 description 7
- 238000002156 mixing Methods 0.000 description 7
- 150000003512 tertiary amines Chemical class 0.000 description 7
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 7
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 6
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 6
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 6
- 125000003545 alkoxy group Chemical group 0.000 description 6
- 239000004305 biphenyl Substances 0.000 description 6
- 235000010290 biphenyl Nutrition 0.000 description 6
- PXKLMJQFEQBVLD-UHFFFAOYSA-N bisphenol F Chemical compound C1=CC(O)=CC=C1CC1=CC=C(O)C=C1 PXKLMJQFEQBVLD-UHFFFAOYSA-N 0.000 description 6
- QPJDMGCKMHUXFD-UHFFFAOYSA-N cyanogen chloride Chemical compound ClC#N QPJDMGCKMHUXFD-UHFFFAOYSA-N 0.000 description 6
- 125000005843 halogen group Chemical group 0.000 description 6
- IVSZLXZYQVIEFR-UHFFFAOYSA-N m-xylene Chemical group CC1=CC=CC(C)=C1 IVSZLXZYQVIEFR-UHFFFAOYSA-N 0.000 description 6
- 239000012046 mixed solvent Substances 0.000 description 6
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 6
- 150000003923 2,5-pyrrolediones Chemical class 0.000 description 5
- CTQNGGLPUBDAKN-UHFFFAOYSA-N O-Xylene Chemical compound CC1=CC=CC=C1C CTQNGGLPUBDAKN-UHFFFAOYSA-N 0.000 description 5
- 150000001491 aromatic compounds Chemical class 0.000 description 5
- JFDZBHWFFUWGJE-UHFFFAOYSA-N benzenecarbonitrile Natural products N#CC1=CC=CC=C1 JFDZBHWFFUWGJE-UHFFFAOYSA-N 0.000 description 5
- 239000006227 byproduct Substances 0.000 description 5
- 239000002131 composite material Substances 0.000 description 5
- JHIVVAPYMSGYDF-UHFFFAOYSA-N cyclohexanone Chemical compound O=C1CCCCC1 JHIVVAPYMSGYDF-UHFFFAOYSA-N 0.000 description 5
- 239000003365 glass fiber Substances 0.000 description 5
- 230000009477 glass transition Effects 0.000 description 5
- 150000002430 hydrocarbons Chemical class 0.000 description 5
- 230000001965 increasing effect Effects 0.000 description 5
- 150000007530 organic bases Chemical class 0.000 description 5
- 229920001225 polyester resin Polymers 0.000 description 5
- 239000004645 polyester resin Substances 0.000 description 5
- 239000002685 polymerization catalyst Substances 0.000 description 5
- 239000002994 raw material Substances 0.000 description 5
- 239000012783 reinforcing fiber Substances 0.000 description 5
- 239000008096 xylene Substances 0.000 description 5
- HECLRDQVFMWTQS-RGOKHQFPSA-N 1755-01-7 Chemical class C1[C@H]2[C@@H]3CC=C[C@@H]3[C@@H]1C=C2 HECLRDQVFMWTQS-RGOKHQFPSA-N 0.000 description 4
- XKRFYHLGVUSROY-UHFFFAOYSA-N Argon Chemical compound [Ar] XKRFYHLGVUSROY-UHFFFAOYSA-N 0.000 description 4
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 4
- 239000004215 Carbon black (E152) Substances 0.000 description 4
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 description 4
- XTHFKEDIFFGKHM-UHFFFAOYSA-N Dimethoxyethane Chemical compound COCCOC XTHFKEDIFFGKHM-UHFFFAOYSA-N 0.000 description 4
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 4
- WSFSSNUMVMOOMR-UHFFFAOYSA-N Formaldehyde Chemical class O=C WSFSSNUMVMOOMR-UHFFFAOYSA-N 0.000 description 4
- JLTDJTHDQAWBAV-UHFFFAOYSA-N N,N-dimethylaniline Chemical compound CN(C)C1=CC=CC=C1 JLTDJTHDQAWBAV-UHFFFAOYSA-N 0.000 description 4
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 4
- JUJWROOIHBZHMG-UHFFFAOYSA-N Pyridine Chemical compound C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 description 4
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 4
- 150000001252 acrylic acid derivatives Chemical class 0.000 description 4
- 239000005456 alcohol based solvent Substances 0.000 description 4
- MWPLVEDNUUSJAV-UHFFFAOYSA-N anthracene Chemical compound C1=CC=CC2=CC3=CC=CC=C3C=C21 MWPLVEDNUUSJAV-UHFFFAOYSA-N 0.000 description 4
- 239000007864 aqueous solution Substances 0.000 description 4
- 125000003710 aryl alkyl group Chemical group 0.000 description 4
- 230000001588 bifunctional effect Effects 0.000 description 4
- QARVLSVVCXYDNA-UHFFFAOYSA-N bromobenzene Chemical compound BrC1=CC=CC=C1 QARVLSVVCXYDNA-UHFFFAOYSA-N 0.000 description 4
- 239000012295 chemical reaction liquid Substances 0.000 description 4
- MVPPADPHJFYWMZ-UHFFFAOYSA-N chlorobenzene Chemical compound ClC1=CC=CC=C1 MVPPADPHJFYWMZ-UHFFFAOYSA-N 0.000 description 4
- 229930003836 cresol Natural products 0.000 description 4
- BGTOWKSIORTVQH-UHFFFAOYSA-N cyclopentanone Chemical compound O=C1CCCC1 BGTOWKSIORTVQH-UHFFFAOYSA-N 0.000 description 4
- GGSUCNLOZRCGPQ-UHFFFAOYSA-N diethylaniline Chemical compound CCN(CC)C1=CC=CC=C1 GGSUCNLOZRCGPQ-UHFFFAOYSA-N 0.000 description 4
- MTZQAGJQAFMTAQ-UHFFFAOYSA-N ethyl benzoate Chemical compound CCOC(=O)C1=CC=CC=C1 MTZQAGJQAFMTAQ-UHFFFAOYSA-N 0.000 description 4
- NIHNNTQXNPWCJQ-UHFFFAOYSA-N fluorene Chemical compound C1=CC=C2CC3=CC=CC=C3C2=C1 NIHNNTQXNPWCJQ-UHFFFAOYSA-N 0.000 description 4
- 238000010438 heat treatment Methods 0.000 description 4
- 229930195733 hydrocarbon Natural products 0.000 description 4
- LELOWRISYMNNSU-UHFFFAOYSA-N hydrogen cyanide Chemical compound N#C LELOWRISYMNNSU-UHFFFAOYSA-N 0.000 description 4
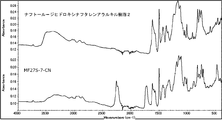
- 238000002329 infrared spectrum Methods 0.000 description 4
- 150000007529 inorganic bases Chemical class 0.000 description 4
- 239000005453 ketone based solvent Substances 0.000 description 4
- 239000002648 laminated material Substances 0.000 description 4
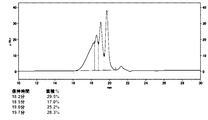
- 238000005259 measurement Methods 0.000 description 4
- 229910052751 metal Inorganic materials 0.000 description 4
- 239000002184 metal Substances 0.000 description 4
- 125000001624 naphthyl group Chemical group 0.000 description 4
- LQNUZADURLCDLV-UHFFFAOYSA-N nitrobenzene Chemical compound [O-][N+](=O)C1=CC=CC=C1 LQNUZADURLCDLV-UHFFFAOYSA-N 0.000 description 4
- 125000004430 oxygen atom Chemical group O* 0.000 description 4
- 239000012071 phase Substances 0.000 description 4
- 230000000704 physical effect Effects 0.000 description 4
- HBMJWWWQQXIZIP-UHFFFAOYSA-N silicon carbide Chemical compound [Si+]#[C-] HBMJWWWQQXIZIP-UHFFFAOYSA-N 0.000 description 4
- 239000000126 substance Substances 0.000 description 4
- VZGDMQKNWNREIO-UHFFFAOYSA-N tetrachloromethane Chemical compound ClC(Cl)(Cl)Cl VZGDMQKNWNREIO-UHFFFAOYSA-N 0.000 description 4
- ZUHZGEOKBKGPSW-UHFFFAOYSA-N tetraglyme Chemical compound COCCOCCOCCOCCOC ZUHZGEOKBKGPSW-UHFFFAOYSA-N 0.000 description 4
- IMFACGCPASFAPR-UHFFFAOYSA-N tributylamine Chemical compound CCCCN(CCCC)CCCC IMFACGCPASFAPR-UHFFFAOYSA-N 0.000 description 4
- 125000002023 trifluoromethyl group Chemical group FC(F)(F)* 0.000 description 4
- 239000002759 woven fabric Substances 0.000 description 4
- QTWJRLJHJPIABL-UHFFFAOYSA-N 2-methylphenol;3-methylphenol;4-methylphenol Chemical compound CC1=CC=C(O)C=C1.CC1=CC=CC(O)=C1.CC1=CC=CC=C1O QTWJRLJHJPIABL-UHFFFAOYSA-N 0.000 description 3
- CMLFRMDBDNHMRA-UHFFFAOYSA-N 2h-1,2-benzoxazine Chemical compound C1=CC=C2C=CNOC2=C1 CMLFRMDBDNHMRA-UHFFFAOYSA-N 0.000 description 3
- VPWNQTHUCYMVMZ-UHFFFAOYSA-N 4,4'-sulfonyldiphenol Chemical compound C1=CC(O)=CC=C1S(=O)(=O)C1=CC=C(O)C=C1 VPWNQTHUCYMVMZ-UHFFFAOYSA-N 0.000 description 3
- XFXPMWWXUTWYJX-UHFFFAOYSA-N Cyanide Chemical compound N#[C-] XFXPMWWXUTWYJX-UHFFFAOYSA-N 0.000 description 3
- 239000004593 Epoxy Substances 0.000 description 3
- HYTRYEXINDDXJK-UHFFFAOYSA-N Ethyl isopropyl ketone Chemical compound CCC(=O)C(C)C HYTRYEXINDDXJK-UHFFFAOYSA-N 0.000 description 3
- 238000005033 Fourier transform infrared spectroscopy Methods 0.000 description 3
- WMFOQBRAJBCJND-UHFFFAOYSA-M Lithium hydroxide Chemical compound [Li+].[OH-] WMFOQBRAJBCJND-UHFFFAOYSA-M 0.000 description 3
- LRHPLDYGYMQRHN-UHFFFAOYSA-N N-Butanol Chemical compound CCCCO LRHPLDYGYMQRHN-UHFFFAOYSA-N 0.000 description 3
- MUBZPKHOEPUJKR-UHFFFAOYSA-N Oxalic acid Chemical compound OC(=O)C(O)=O MUBZPKHOEPUJKR-UHFFFAOYSA-N 0.000 description 3
- KWYUFKZDYYNOTN-UHFFFAOYSA-M Potassium hydroxide Chemical compound [OH-].[K+] KWYUFKZDYYNOTN-UHFFFAOYSA-M 0.000 description 3
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 3
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 3
- 235000011054 acetic acid Nutrition 0.000 description 3
- 125000002723 alicyclic group Chemical group 0.000 description 3
- 239000008346 aqueous phase Substances 0.000 description 3
- 239000004760 aramid Substances 0.000 description 3
- 150000004945 aromatic hydrocarbons Chemical class 0.000 description 3
- 239000004927 clay Substances 0.000 description 3
- 238000002425 crystallisation Methods 0.000 description 3
- 230000008025 crystallization Effects 0.000 description 3
- 238000001035 drying Methods 0.000 description 3
- 230000000694 effects Effects 0.000 description 3
- 239000003759 ester based solvent Substances 0.000 description 3
- 125000004185 ester group Chemical group 0.000 description 3
- 229910052731 fluorine Inorganic materials 0.000 description 3
- OIAUFEASXQPCFE-UHFFFAOYSA-N formaldehyde;1,3-xylene Chemical class O=C.CC1=CC=CC(C)=C1 OIAUFEASXQPCFE-UHFFFAOYSA-N 0.000 description 3
- 150000004820 halides Chemical class 0.000 description 3
- 229910052736 halogen Inorganic materials 0.000 description 3
- 150000002367 halogens Chemical class 0.000 description 3
- 150000008040 ionic compounds Chemical class 0.000 description 3
- PXZQEOJJUGGUIB-UHFFFAOYSA-N isoindolin-1-one Chemical compound C1=CC=C2C(=O)NCC2=C1 PXZQEOJJUGGUIB-UHFFFAOYSA-N 0.000 description 3
- 125000000956 methoxy group Chemical group [H]C([H])([H])O* 0.000 description 3
- 239000004843 novolac epoxy resin Substances 0.000 description 3
- 150000007524 organic acids Chemical class 0.000 description 3
- 239000000123 paper Substances 0.000 description 3
- 150000002989 phenols Chemical class 0.000 description 3
- 125000004437 phosphorous atom Chemical group 0.000 description 3
- 229910052698 phosphorus Inorganic materials 0.000 description 3
- 238000006116 polymerization reaction Methods 0.000 description 3
- 239000000843 powder Substances 0.000 description 3
- 238000007086 side reaction Methods 0.000 description 3
- 229910010271 silicon carbide Inorganic materials 0.000 description 3
- JOXIMZWYDAKGHI-UHFFFAOYSA-N toluene-4-sulfonic acid Chemical compound CC1=CC=C(S(O)(=O)=O)C=C1 JOXIMZWYDAKGHI-UHFFFAOYSA-N 0.000 description 3
- 239000002351 wastewater Substances 0.000 description 3
- UOCLXMDMGBRAIB-UHFFFAOYSA-N 1,1,1-trichloroethane Chemical compound CC(Cl)(Cl)Cl UOCLXMDMGBRAIB-UHFFFAOYSA-N 0.000 description 2
- HCNHNBLSNVSJTJ-UHFFFAOYSA-N 1,1-Bis(4-hydroxyphenyl)ethane Chemical compound C=1C=C(O)C=CC=1C(C)C1=CC=C(O)C=C1 HCNHNBLSNVSJTJ-UHFFFAOYSA-N 0.000 description 2
- SCYULBFZEHDVBN-UHFFFAOYSA-N 1,1-Dichloroethane Chemical compound CC(Cl)Cl SCYULBFZEHDVBN-UHFFFAOYSA-N 0.000 description 2
- JYEUMXHLPRZUAT-UHFFFAOYSA-N 1,2,3-triazine Chemical group C1=CN=NN=C1 JYEUMXHLPRZUAT-UHFFFAOYSA-N 0.000 description 2
- MYRTYDVEIRVNKP-UHFFFAOYSA-N 1,2-Divinylbenzene Chemical compound C=CC1=CC=CC=C1C=C MYRTYDVEIRVNKP-UHFFFAOYSA-N 0.000 description 2
- RYHBNJHYFVUHQT-UHFFFAOYSA-N 1,4-Dioxane Chemical compound C1COCCO1 RYHBNJHYFVUHQT-UHFFFAOYSA-N 0.000 description 2
- XQUPVDVFXZDTLT-UHFFFAOYSA-N 1-[4-[[4-(2,5-dioxopyrrol-1-yl)phenyl]methyl]phenyl]pyrrole-2,5-dione Chemical compound O=C1C=CC(=O)N1C(C=C1)=CC=C1CC1=CC=C(N2C(C=CC2=O)=O)C=C1 XQUPVDVFXZDTLT-UHFFFAOYSA-N 0.000 description 2
- RNFJDJUURJAICM-UHFFFAOYSA-N 2,2,4,4,6,6-hexaphenoxy-1,3,5-triaza-2$l^{5},4$l^{5},6$l^{5}-triphosphacyclohexa-1,3,5-triene Chemical compound N=1P(OC=2C=CC=CC=2)(OC=2C=CC=CC=2)=NP(OC=2C=CC=CC=2)(OC=2C=CC=CC=2)=NP=1(OC=1C=CC=CC=1)OC1=CC=CC=C1 RNFJDJUURJAICM-UHFFFAOYSA-N 0.000 description 2
- GQHTUMJGOHRCHB-UHFFFAOYSA-N 2,3,4,6,7,8,9,10-octahydropyrimido[1,2-a]azepine Chemical compound C1CCCCN2CCCN=C21 GQHTUMJGOHRCHB-UHFFFAOYSA-N 0.000 description 2
- YIWUKEYIRIRTPP-UHFFFAOYSA-N 2-ethylhexan-1-ol Chemical compound CCCCC(CC)CO YIWUKEYIRIRTPP-UHFFFAOYSA-N 0.000 description 2
- JWUJQDFVADABEY-UHFFFAOYSA-N 2-methyltetrahydrofuran Chemical compound CC1CCCO1 JWUJQDFVADABEY-UHFFFAOYSA-N 0.000 description 2
- VTYYLEPIZMXCLO-UHFFFAOYSA-L Calcium carbonate Chemical compound [Ca+2].[O-]C([O-])=O VTYYLEPIZMXCLO-UHFFFAOYSA-L 0.000 description 2
- XDTMQSROBMDMFD-UHFFFAOYSA-N Cyclohexane Chemical compound C1CCCCC1 XDTMQSROBMDMFD-UHFFFAOYSA-N 0.000 description 2
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 2
- CSNNHWWHGAXBCP-UHFFFAOYSA-L Magnesium sulfate Chemical compound [Mg+2].[O-][S+2]([O-])([O-])[O-] CSNNHWWHGAXBCP-UHFFFAOYSA-L 0.000 description 2
- SECXISVLQFMRJM-UHFFFAOYSA-N N-Methylpyrrolidone Chemical compound CN1CCCC1=O SECXISVLQFMRJM-UHFFFAOYSA-N 0.000 description 2
- UFWIBTONFRDIAS-UHFFFAOYSA-N Naphthalene Chemical compound C1=CC=CC2=CC=CC=C21 UFWIBTONFRDIAS-UHFFFAOYSA-N 0.000 description 2
- ATUOYWHBWRKTHZ-UHFFFAOYSA-N Propane Chemical compound CCC ATUOYWHBWRKTHZ-UHFFFAOYSA-N 0.000 description 2
- GSEJCLTVZPLZKY-UHFFFAOYSA-N Triethanolamine Chemical compound OCCN(CCO)CCO GSEJCLTVZPLZKY-UHFFFAOYSA-N 0.000 description 2
- ZJCCRDAZUWHFQH-UHFFFAOYSA-N Trimethylolpropane Chemical compound CCC(CO)(CO)CO ZJCCRDAZUWHFQH-UHFFFAOYSA-N 0.000 description 2
- 239000007983 Tris buffer Substances 0.000 description 2
- XLOMVQKBTHCTTD-UHFFFAOYSA-N Zinc monoxide Chemical compound [Zn]=O XLOMVQKBTHCTTD-UHFFFAOYSA-N 0.000 description 2
- DUEAQFNVHPRQKJ-UHFFFAOYSA-N [4-[1-(4-cyanatophenyl)-2-methylpropyl]phenyl] cyanate Chemical compound C=1C=C(OC#N)C=CC=1C(C(C)C)C1=CC=C(OC#N)C=C1 DUEAQFNVHPRQKJ-UHFFFAOYSA-N 0.000 description 2
- INHGSGHLQLYYND-UHFFFAOYSA-N [4-[2-(4-cyanatophenyl)-1,1,1,3,3,3-hexafluoropropan-2-yl]phenyl] cyanate Chemical compound C=1C=C(OC#N)C=CC=1C(C(F)(F)F)(C(F)(F)F)C1=CC=C(OC#N)C=C1 INHGSGHLQLYYND-UHFFFAOYSA-N 0.000 description 2
- AHZMUXQJTGRNHT-UHFFFAOYSA-N [4-[2-(4-cyanatophenyl)propan-2-yl]phenyl] cyanate Chemical compound C=1C=C(OC#N)C=CC=1C(C)(C)C1=CC=C(OC#N)C=C1 AHZMUXQJTGRNHT-UHFFFAOYSA-N 0.000 description 2
- PNEYBMLMFCGWSK-UHFFFAOYSA-N aluminium oxide Inorganic materials [O-2].[O-2].[O-2].[Al+3].[Al+3] PNEYBMLMFCGWSK-UHFFFAOYSA-N 0.000 description 2
- 229910000147 aluminium phosphate Inorganic materials 0.000 description 2
- 238000004458 analytical method Methods 0.000 description 2
- RDOXTESZEPMUJZ-UHFFFAOYSA-N anisole Chemical compound COC1=CC=CC=C1 RDOXTESZEPMUJZ-UHFFFAOYSA-N 0.000 description 2
- 229910052786 argon Inorganic materials 0.000 description 2
- 229920003235 aromatic polyamide Polymers 0.000 description 2
- 239000012298 atmosphere Substances 0.000 description 2
- TZCXTZWJZNENPQ-UHFFFAOYSA-L barium sulfate Chemical compound [Ba+2].[O-]S([O-])(=O)=O TZCXTZWJZNENPQ-UHFFFAOYSA-L 0.000 description 2
- 150000005130 benzoxazines Chemical class 0.000 description 2
- OSGAYBCDTDRGGQ-UHFFFAOYSA-L calcium sulfate Chemical compound [Ca+2].[O-]S([O-])(=O)=O OSGAYBCDTDRGGQ-UHFFFAOYSA-L 0.000 description 2
- 235000011132 calcium sulphate Nutrition 0.000 description 2
- 235000010261 calcium sulphite Nutrition 0.000 description 2
- 125000002915 carbonyl group Chemical group [*:2]C([*:1])=O 0.000 description 2
- 239000003795 chemical substances by application Substances 0.000 description 2
- 229910052801 chlorine Inorganic materials 0.000 description 2
- 125000001309 chloro group Chemical group Cl* 0.000 description 2
- 238000009833 condensation Methods 0.000 description 2
- 230000005494 condensation Effects 0.000 description 2
- 239000013078 crystal Substances 0.000 description 2
- 125000001651 cyanato group Chemical group [*]OC#N 0.000 description 2
- 125000004122 cyclic group Chemical group 0.000 description 2
- 125000004956 cyclohexylene group Chemical group 0.000 description 2
- 239000008367 deionised water Substances 0.000 description 2
- 229910021641 deionized water Inorganic materials 0.000 description 2
- SBZXBUIDTXKZTM-UHFFFAOYSA-N diglyme Chemical compound COCCOCCOC SBZXBUIDTXKZTM-UHFFFAOYSA-N 0.000 description 2
- XBDQKXXYIPTUBI-UHFFFAOYSA-N dimethylselenoniopropionate Natural products CCC(O)=O XBDQKXXYIPTUBI-UHFFFAOYSA-N 0.000 description 2
- 238000004821 distillation Methods 0.000 description 2
- 239000012153 distilled water Substances 0.000 description 2
- 239000012772 electrical insulation material Substances 0.000 description 2
- 230000005611 electricity Effects 0.000 description 2
- 239000004210 ether based solvent Substances 0.000 description 2
- LZCLXQDLBQLTDK-UHFFFAOYSA-N ethyl 2-hydroxypropanoate Chemical compound CCOC(=O)C(C)O LZCLXQDLBQLTDK-UHFFFAOYSA-N 0.000 description 2
- RRAFCDWBNXTKKO-UHFFFAOYSA-N eugenol Chemical compound COC1=CC(CC=C)=CC=C1O RRAFCDWBNXTKKO-UHFFFAOYSA-N 0.000 description 2
- 239000003063 flame retardant Substances 0.000 description 2
- 125000001153 fluoro group Chemical group F* 0.000 description 2
- HYBBIBNJHNGZAN-UHFFFAOYSA-N furfural Chemical compound O=CC1=CC=CO1 HYBBIBNJHNGZAN-UHFFFAOYSA-N 0.000 description 2
- 239000007789 gas Substances 0.000 description 2
- 238000004817 gas chromatography Methods 0.000 description 2
- 150000008282 halocarbons Chemical class 0.000 description 2
- 239000001307 helium Substances 0.000 description 2
- 229910052734 helium Inorganic materials 0.000 description 2
- SWQJXJOGLNCZEY-UHFFFAOYSA-N helium atom Chemical compound [He] SWQJXJOGLNCZEY-UHFFFAOYSA-N 0.000 description 2
- 238000004128 high performance liquid chromatography Methods 0.000 description 2
- 239000001257 hydrogen Substances 0.000 description 2
- 229910052739 hydrogen Inorganic materials 0.000 description 2
- 238000005470 impregnation Methods 0.000 description 2
- 239000011261 inert gas Substances 0.000 description 2
- 239000011810 insulating material Substances 0.000 description 2
- ZFSLODLOARCGLH-UHFFFAOYSA-N isocyanuric acid Chemical group OC1=NC(O)=NC(O)=N1 ZFSLODLOARCGLH-UHFFFAOYSA-N 0.000 description 2
- 239000002655 kraft paper Substances 0.000 description 2
- JVTAAEKCZFNVCJ-UHFFFAOYSA-N lactic acid Chemical compound CC(O)C(O)=O JVTAAEKCZFNVCJ-UHFFFAOYSA-N 0.000 description 2
- 238000004811 liquid chromatography Methods 0.000 description 2
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 2
- 125000001570 methylene group Chemical group [H]C([H])([*:1])[*:2] 0.000 description 2
- 150000007522 mineralic acids Chemical class 0.000 description 2
- 239000012778 molding material Substances 0.000 description 2
- 239000000178 monomer Substances 0.000 description 2
- 238000000465 moulding Methods 0.000 description 2
- MXHTZQSKTCCMFG-UHFFFAOYSA-N n,n-dibenzyl-1-phenylmethanamine Chemical compound C=1C=CC=CC=1CN(CC=1C=CC=CC=1)CC1=CC=CC=C1 MXHTZQSKTCCMFG-UHFFFAOYSA-N 0.000 description 2
- FRQONEWDWWHIPM-UHFFFAOYSA-N n,n-dicyclohexylcyclohexanamine Chemical compound C1CCCCC1N(C1CCCCC1)C1CCCCC1 FRQONEWDWWHIPM-UHFFFAOYSA-N 0.000 description 2
- ORSUTASIQKBEFU-UHFFFAOYSA-N n,n-diethylbutan-1-amine Chemical compound CCCCN(CC)CC ORSUTASIQKBEFU-UHFFFAOYSA-N 0.000 description 2
- CIXSDMKDSYXUMJ-UHFFFAOYSA-N n,n-diethylcyclohexanamine Chemical compound CCN(CC)C1CCCCC1 CIXSDMKDSYXUMJ-UHFFFAOYSA-N 0.000 description 2
- YWFWDNVOPHGWMX-UHFFFAOYSA-N n,n-dimethyldodecan-1-amine Chemical compound CCCCCCCCCCCCN(C)C YWFWDNVOPHGWMX-UHFFFAOYSA-N 0.000 description 2
- OOHAUGDGCWURIT-UHFFFAOYSA-N n,n-dipentylpentan-1-amine Chemical compound CCCCCN(CCCCC)CCCCC OOHAUGDGCWURIT-UHFFFAOYSA-N 0.000 description 2
- MTHFROHDIWGWFD-UHFFFAOYSA-N n-butyl-n-methylbutan-1-amine Chemical compound CCCCN(C)CCCC MTHFROHDIWGWFD-UHFFFAOYSA-N 0.000 description 2
- WOLFCKKMHUVEPN-UHFFFAOYSA-N n-ethyl-n-methylbutan-1-amine Chemical compound CCCCN(C)CC WOLFCKKMHUVEPN-UHFFFAOYSA-N 0.000 description 2
- DYFFAVRFJWYYQO-UHFFFAOYSA-N n-methyl-n-phenylaniline Chemical compound C=1C=CC=CC=1N(C)C1=CC=CC=C1 DYFFAVRFJWYYQO-UHFFFAOYSA-N 0.000 description 2
- 238000006386 neutralization reaction Methods 0.000 description 2
- 150000002825 nitriles Chemical class 0.000 description 2
- 229910052757 nitrogen Inorganic materials 0.000 description 2
- 125000004433 nitrogen atom Chemical group N* 0.000 description 2
- LYGJENNIWJXYER-UHFFFAOYSA-N nitromethane Chemical compound C[N+]([O-])=O LYGJENNIWJXYER-UHFFFAOYSA-N 0.000 description 2
- 239000004745 nonwoven fabric Substances 0.000 description 2
- 239000010680 novolac-type phenolic resin Substances 0.000 description 2
- 235000005985 organic acids Nutrition 0.000 description 2
- 125000001037 p-tolyl group Chemical group [H]C1=C([H])C(=C([H])C([H])=C1*)C([H])([H])[H] 0.000 description 2
- KJFMBFZCATUALV-UHFFFAOYSA-N phenolphthalein Chemical compound C1=CC(O)=CC=C1C1(C=2C=CC(O)=CC=2)C2=CC=CC=C2C(=O)O1 KJFMBFZCATUALV-UHFFFAOYSA-N 0.000 description 2
- 125000000951 phenoxy group Chemical group [H]C1=C([H])C([H])=C(O*)C([H])=C1[H] 0.000 description 2
- 239000000049 pigment Substances 0.000 description 2
- 239000002798 polar solvent Substances 0.000 description 2
- 229920003192 poly(bis maleimide) Polymers 0.000 description 2
- 229920001721 polyimide Chemical group 0.000 description 2
- 229920000642 polymer Polymers 0.000 description 2
- 125000004805 propylene group Chemical group [H]C([H])([H])C([H])([*:1])C([H])([H])[*:2] 0.000 description 2
- 238000000746 purification Methods 0.000 description 2
- UMJSCPRVCHMLSP-UHFFFAOYSA-N pyridine Natural products COC1=CC=CN=C1 UMJSCPRVCHMLSP-UHFFFAOYSA-N 0.000 description 2
- 230000035484 reaction time Effects 0.000 description 2
- 239000012744 reinforcing agent Substances 0.000 description 2
- 238000000926 separation method Methods 0.000 description 2
- 229920002050 silicone resin Polymers 0.000 description 2
- SQGYOTSLMSWVJD-UHFFFAOYSA-N silver(1+) nitrate Chemical compound [Ag+].[O-]N(=O)=O SQGYOTSLMSWVJD-UHFFFAOYSA-N 0.000 description 2
- 238000005507 spraying Methods 0.000 description 2
- 238000003860 storage Methods 0.000 description 2
- 125000001424 substituent group Chemical group 0.000 description 2
- 238000012360 testing method Methods 0.000 description 2
- 238000004809 thin layer chromatography Methods 0.000 description 2
- 238000001721 transfer moulding Methods 0.000 description 2
- JIAARYAFYJHUJI-UHFFFAOYSA-L zinc dichloride Chemical compound [Cl-].[Cl-].[Zn+2] JIAARYAFYJHUJI-UHFFFAOYSA-L 0.000 description 2
- JRBJLOCMQWBLDF-UHFFFAOYSA-N (2,6-dichlorophenyl) cyanate Chemical compound ClC1=CC=CC(Cl)=C1OC#N JRBJLOCMQWBLDF-UHFFFAOYSA-N 0.000 description 1
- VSHAQOZVUZJKCO-UHFFFAOYSA-N (2,6-ditert-butylphenyl) cyanate Chemical compound CC(C)(C)C1=CC=CC(C(C)(C)C)=C1OC#N VSHAQOZVUZJKCO-UHFFFAOYSA-N 0.000 description 1
- SMTQKQLZBRJCLI-UHFFFAOYSA-N (2-cyanatophenyl) cyanate Chemical compound N#COC1=CC=CC=C1OC#N SMTQKQLZBRJCLI-UHFFFAOYSA-N 0.000 description 1
- ULYBHRQVCDELMO-UHFFFAOYSA-N (2-ethyl-4-nitrophenyl) cyanate Chemical compound CCC1=CC([N+]([O-])=O)=CC=C1OC#N ULYBHRQVCDELMO-UHFFFAOYSA-N 0.000 description 1
- SHBUQGBESNZMCH-UHFFFAOYSA-N (2-nitrophenyl) cyanate Chemical compound [O-][N+](=O)C1=CC=CC=C1OC#N SHBUQGBESNZMCH-UHFFFAOYSA-N 0.000 description 1
- NGGIZAWDYJYQBS-UHFFFAOYSA-N (2-tert-butyl-4-cyanatophenyl) cyanate Chemical compound CC(C)(C)C1=CC(OC#N)=CC=C1OC#N NGGIZAWDYJYQBS-UHFFFAOYSA-N 0.000 description 1
- QSMZSCNZPOTAQR-UHFFFAOYSA-N (3,5-dimethylphenyl) cyanate Chemical compound CC1=CC(C)=CC(OC#N)=C1 QSMZSCNZPOTAQR-UHFFFAOYSA-N 0.000 description 1
- UBWVCMYZYWVZFK-UHFFFAOYSA-N (3-chloro-2-methylphenyl) cyanate Chemical compound CC1=C(Cl)C=CC=C1OC#N UBWVCMYZYWVZFK-UHFFFAOYSA-N 0.000 description 1
- YVOQQUGUJXFKOC-UHFFFAOYSA-N (3-chlorophenyl) cyanate Chemical compound ClC1=CC=CC(OC#N)=C1 YVOQQUGUJXFKOC-UHFFFAOYSA-N 0.000 description 1
- UVKWYPJUDVBJBS-UHFFFAOYSA-N (3-cyanato-2,4,6-trimethylphenyl) cyanate Chemical compound CC1=CC(C)=C(OC#N)C(C)=C1OC#N UVKWYPJUDVBJBS-UHFFFAOYSA-N 0.000 description 1
- OSHDNORJIBTCJW-UHFFFAOYSA-N (3-cyanato-5-methylphenyl) cyanate Chemical compound CC1=CC(OC#N)=CC(OC#N)=C1 OSHDNORJIBTCJW-UHFFFAOYSA-N 0.000 description 1
- QQZZMAPJAKOSNG-UHFFFAOYSA-N (3-cyanatophenyl) cyanate Chemical compound N#COC1=CC=CC(OC#N)=C1 QQZZMAPJAKOSNG-UHFFFAOYSA-N 0.000 description 1
- UNMJLQGKEDTEKJ-UHFFFAOYSA-N (3-ethyloxetan-3-yl)methanol Chemical compound CCC1(CO)COC1 UNMJLQGKEDTEKJ-UHFFFAOYSA-N 0.000 description 1
- MJTRGJDBPFGULK-UHFFFAOYSA-N (4-acetamidophenyl) cyanate Chemical compound CC(=O)NC1=CC=C(OC#N)C=C1 MJTRGJDBPFGULK-UHFFFAOYSA-N 0.000 description 1
- MUAFEFAYKJZTHJ-UHFFFAOYSA-N (4-acetylphenyl) cyanate Chemical compound CC(=O)C1=CC=C(OC#N)C=C1 MUAFEFAYKJZTHJ-UHFFFAOYSA-N 0.000 description 1
- OXYKVVLTXXXVRT-UHFFFAOYSA-N (4-chlorobenzoyl) 4-chlorobenzenecarboperoxoate Chemical compound C1=CC(Cl)=CC=C1C(=O)OOC(=O)C1=CC=C(Cl)C=C1 OXYKVVLTXXXVRT-UHFFFAOYSA-N 0.000 description 1
- XEFGVCMYMDAKON-UHFFFAOYSA-N (4-cyanato-1,5,6-trimethylcyclohexa-2,4-dien-1-yl) cyanate Chemical compound O(C#N)C1=C(C(C(C=C1)(C)OC#N)C)C XEFGVCMYMDAKON-UHFFFAOYSA-N 0.000 description 1
- IJYLPQZZHPXEIH-UHFFFAOYSA-N (4-cyanato-1,5-dimethylcyclohexa-2,4-dien-1-yl) cyanate Chemical compound O(C#N)C1=C(CC(C=C1)(C)OC#N)C IJYLPQZZHPXEIH-UHFFFAOYSA-N 0.000 description 1
- GUGZCSAPOLLKNG-UHFFFAOYSA-N (4-cyanatophenyl) cyanate Chemical compound N#COC1=CC=C(OC#N)C=C1 GUGZCSAPOLLKNG-UHFFFAOYSA-N 0.000 description 1
- FFUGFEZHSFMOTP-UHFFFAOYSA-N (4-cyclohexylphenyl) cyanate Chemical compound C1=CC(OC#N)=CC=C1C1CCCCC1 FFUGFEZHSFMOTP-UHFFFAOYSA-N 0.000 description 1
- LKOYFNVMCUKOJD-UHFFFAOYSA-N (4-ethenylphenyl) cyanate Chemical compound C=CC1=CC=C(OC#N)C=C1 LKOYFNVMCUKOJD-UHFFFAOYSA-N 0.000 description 1
- BYMZMVNQWJIIDN-UHFFFAOYSA-N (4-formylphenyl) cyanate Chemical compound O=CC1=CC=C(OC#N)C=C1 BYMZMVNQWJIIDN-UHFFFAOYSA-N 0.000 description 1
- HZSITCSNYNWEMM-UHFFFAOYSA-N (4-methoxynaphthalen-1-yl) cyanate Chemical compound O(C#N)C1=CC=C(C2=CC=CC=C12)OC HZSITCSNYNWEMM-UHFFFAOYSA-N 0.000 description 1
- RFXSQXFVJCZAMW-UHFFFAOYSA-N (4-methoxyphenyl) cyanate Chemical compound COC1=CC=C(OC#N)C=C1 RFXSQXFVJCZAMW-UHFFFAOYSA-N 0.000 description 1
- UGMKNMPRUHJNQK-UHFFFAOYSA-N (4-methylphenyl) cyanate Chemical compound CC1=CC=C(OC#N)C=C1 UGMKNMPRUHJNQK-UHFFFAOYSA-N 0.000 description 1
- OQOBDFYUAMYTRM-UHFFFAOYSA-N (4-methylsulfanylphenyl) cyanate Chemical compound CSC1=CC=C(OC#N)C=C1 OQOBDFYUAMYTRM-UHFFFAOYSA-N 0.000 description 1
- SGACLTVKXIFYLM-UHFFFAOYSA-N (4-phenylphenyl) cyanate Chemical group C1=CC(OC#N)=CC=C1C1=CC=CC=C1 SGACLTVKXIFYLM-UHFFFAOYSA-N 0.000 description 1
- RUEBPOOTFCZRBC-UHFFFAOYSA-N (5-methyl-2-phenyl-1h-imidazol-4-yl)methanol Chemical compound OCC1=C(C)NC(C=2C=CC=CC=2)=N1 RUEBPOOTFCZRBC-UHFFFAOYSA-N 0.000 description 1
- PCJABUZFXDYTMH-UHFFFAOYSA-N (6-methylnaphthalen-2-yl) cyanate Chemical compound O(C#N)C1=CC2=CC=C(C=C2C=C1)C PCJABUZFXDYTMH-UHFFFAOYSA-N 0.000 description 1
- PWGMXFQYTQPRLC-UHFFFAOYSA-N (7-methoxynaphthalen-2-yl) cyanate Chemical compound O(C#N)C1=CC2=CC(=CC=C2C=C1)OC PWGMXFQYTQPRLC-UHFFFAOYSA-N 0.000 description 1
- VRKVWGGGHMMERE-UHFFFAOYSA-N 1,2-bis(methoxymethyl)benzene Chemical compound COCC1=CC=CC=C1COC VRKVWGGGHMMERE-UHFFFAOYSA-N 0.000 description 1
- QVCUKHQDEZNNOC-UHFFFAOYSA-N 1,2-diazabicyclo[2.2.2]octane Chemical compound C1CC2CCN1NC2 QVCUKHQDEZNNOC-UHFFFAOYSA-N 0.000 description 1
- OUPZKGBUJRBPGC-UHFFFAOYSA-N 1,3,5-tris(oxiran-2-ylmethyl)-1,3,5-triazinane-2,4,6-trione Chemical compound O=C1N(CC2OC2)C(=O)N(CC2OC2)C(=O)N1CC1CO1 OUPZKGBUJRBPGC-UHFFFAOYSA-N 0.000 description 1
- CYSGHNMQYZDMIA-UHFFFAOYSA-N 1,3-Dimethyl-2-imidazolidinon Chemical compound CN1CCN(C)C1=O CYSGHNMQYZDMIA-UHFFFAOYSA-N 0.000 description 1
- SGUVLZREKBPKCE-UHFFFAOYSA-N 1,5-diazabicyclo[4.3.0]-non-5-ene Chemical compound C1CCN=C2CCCN21 SGUVLZREKBPKCE-UHFFFAOYSA-N 0.000 description 1
- MDEDKSYUWOWMHU-UHFFFAOYSA-N 1-[2-ethyl-4-[[3-ethyl-5-methyl-4-(3-methyl-2,5-dioxopyrrol-1-yl)phenyl]methyl]-6-methylphenyl]-3-methylpyrrole-2,5-dione Chemical compound C(C)C=1C=C(C=C(C=1N1C(C(C)=CC1=O)=O)C)CC1=CC(=C(C(=C1)C)N1C(C(C)=CC1=O)=O)CC MDEDKSYUWOWMHU-UHFFFAOYSA-N 0.000 description 1
- IPJGAEWUPXWFPL-UHFFFAOYSA-N 1-[3-(2,5-dioxopyrrol-1-yl)phenyl]pyrrole-2,5-dione Chemical compound O=C1C=CC(=O)N1C1=CC=CC(N2C(C=CC2=O)=O)=C1 IPJGAEWUPXWFPL-UHFFFAOYSA-N 0.000 description 1
- MBNMVPPPCZKPIJ-UHFFFAOYSA-N 1-[3-[3-[3-(2,5-dioxopyrrol-1-yl)phenoxy]phenoxy]phenyl]pyrrole-2,5-dione Chemical compound O=C1C=CC(=O)N1C1=CC=CC(OC=2C=C(OC=3C=C(C=CC=3)N3C(C=CC3=O)=O)C=CC=2)=C1 MBNMVPPPCZKPIJ-UHFFFAOYSA-N 0.000 description 1
- IKFPAKYBSYICFK-UHFFFAOYSA-N 1-[4-(4-propylphenoxy)phenyl]pyrrole-2,5-dione Chemical compound C1=CC(CCC)=CC=C1OC1=CC=C(N2C(C=CC2=O)=O)C=C1 IKFPAKYBSYICFK-UHFFFAOYSA-N 0.000 description 1
- UGJHILWNNSROJV-UHFFFAOYSA-N 1-[4-[3-[4-(2,5-dioxopyrrol-1-yl)phenoxy]phenoxy]phenyl]pyrrole-2,5-dione Chemical compound O=C1C=CC(=O)N1C(C=C1)=CC=C1OC1=CC=CC(OC=2C=CC(=CC=2)N2C(C=CC2=O)=O)=C1 UGJHILWNNSROJV-UHFFFAOYSA-N 0.000 description 1
- BDDKHONBQBEGMA-UHFFFAOYSA-N 1-[4-[[3,5-diethyl-4-(3-methyl-2,5-dioxopyrrol-1-yl)phenyl]methyl]-2,6-diethylphenyl]-3-methylpyrrole-2,5-dione Chemical compound C=1C(CC)=C(N2C(C(C)=CC2=O)=O)C(CC)=CC=1CC(C=C1CC)=CC(CC)=C1N1C(=O)C=C(C)C1=O BDDKHONBQBEGMA-UHFFFAOYSA-N 0.000 description 1
- DRDFUKFBOJQEEO-UHFFFAOYSA-N 1-[4-[[3,5-dimethyl-4-(3-methyl-2,5-dioxopyrrol-1-yl)phenyl]methyl]-2,6-dimethylphenyl]-3-methylpyrrole-2,5-dione Chemical compound O=C1C(C)=CC(=O)N1C(C(=C1)C)=C(C)C=C1CC(C=C1C)=CC(C)=C1N1C(=O)C(C)=CC1=O DRDFUKFBOJQEEO-UHFFFAOYSA-N 0.000 description 1
- NHWYMYDMYCNUKI-UHFFFAOYSA-N 1-[4-[[4-(2,5-dioxopyrrol-1-yl)-3,5-diethylphenyl]methyl]-2,6-diethylphenyl]pyrrole-2,5-dione Chemical compound C=1C(CC)=C(N2C(C=CC2=O)=O)C(CC)=CC=1CC(C=C1CC)=CC(CC)=C1N1C(=O)C=CC1=O NHWYMYDMYCNUKI-UHFFFAOYSA-N 0.000 description 1
- RUORVEVRVBXRIO-UHFFFAOYSA-N 1-[4-[[4-(2,5-dioxopyrrol-1-yl)-3,5-dimethylphenyl]methyl]-2,6-dimethylphenyl]pyrrole-2,5-dione Chemical compound C=1C(C)=C(N2C(C=CC2=O)=O)C(C)=CC=1CC(C=C1C)=CC(C)=C1N1C(=O)C=CC1=O RUORVEVRVBXRIO-UHFFFAOYSA-N 0.000 description 1
- YNSSPVZNXLACMW-UHFFFAOYSA-N 1-[4-[[4-(2,5-dioxopyrrol-1-yl)-3-ethyl-5-methylphenyl]methyl]-2-ethyl-6-methylphenyl]pyrrole-2,5-dione Chemical compound C=1C(C)=C(N2C(C=CC2=O)=O)C(CC)=CC=1CC(C=C1CC)=CC(C)=C1N1C(=O)C=CC1=O YNSSPVZNXLACMW-UHFFFAOYSA-N 0.000 description 1
- IYSVFZBXZVPIFA-UHFFFAOYSA-N 1-ethenyl-4-(4-ethenylphenyl)benzene Chemical group C1=CC(C=C)=CC=C1C1=CC=C(C=C)C=C1 IYSVFZBXZVPIFA-UHFFFAOYSA-N 0.000 description 1
- JOLQKTGDSGKSKJ-UHFFFAOYSA-N 1-ethoxypropan-2-ol Chemical compound CCOCC(C)O JOLQKTGDSGKSKJ-UHFFFAOYSA-N 0.000 description 1
- ARXJGSRGQADJSQ-UHFFFAOYSA-N 1-methoxypropan-2-ol Chemical compound COCC(C)O ARXJGSRGQADJSQ-UHFFFAOYSA-N 0.000 description 1
- SQAINHDHICKHLX-UHFFFAOYSA-N 1-naphthaldehyde Chemical compound C1=CC=C2C(C=O)=CC=CC2=C1 SQAINHDHICKHLX-UHFFFAOYSA-N 0.000 description 1
- PLQNDYUMLMVFCX-UHFFFAOYSA-N 1h-imidazol-2-ylmethanediol Chemical compound OC(O)C1=NC=CN1 PLQNDYUMLMVFCX-UHFFFAOYSA-N 0.000 description 1
- ZWZGXLKXKAPXMZ-UHFFFAOYSA-N 2,2'-dihydroxy-3,3'-dimethoxy-5,5'-dipropyldiphenylmethane Chemical compound COC1=CC(CCC)=CC(CC=2C(=C(OC)C=C(CCC)C=2)O)=C1O ZWZGXLKXKAPXMZ-UHFFFAOYSA-N 0.000 description 1
- XHYFCIYCSYEDCP-UHFFFAOYSA-N 2,2-dimethyloxetane Chemical compound CC1(C)CCO1 XHYFCIYCSYEDCP-UHFFFAOYSA-N 0.000 description 1
- MTJUELTVQKBEPR-UHFFFAOYSA-N 2-(chloromethyl)oxetane Chemical compound ClCC1CCO1 MTJUELTVQKBEPR-UHFFFAOYSA-N 0.000 description 1
- XNWFRZJHXBZDAG-UHFFFAOYSA-N 2-METHOXYETHANOL Chemical compound COCCO XNWFRZJHXBZDAG-UHFFFAOYSA-N 0.000 description 1
- LBLYYCQCTBFVLH-UHFFFAOYSA-N 2-Methylbenzenesulfonic acid Chemical compound CC1=CC=CC=C1S(O)(=O)=O LBLYYCQCTBFVLH-UHFFFAOYSA-N 0.000 description 1
- TXBCBTDQIULDIA-UHFFFAOYSA-N 2-[[3-hydroxy-2,2-bis(hydroxymethyl)propoxy]methyl]-2-(hydroxymethyl)propane-1,3-diol Chemical compound OCC(CO)(CO)COCC(CO)(CO)CO TXBCBTDQIULDIA-UHFFFAOYSA-N 0.000 description 1
- WNZQDUSMALZDQF-UHFFFAOYSA-N 2-benzofuran-1(3H)-one Chemical compound C1=CC=C2C(=O)OCC2=C1 WNZQDUSMALZDQF-UHFFFAOYSA-N 0.000 description 1
- 125000004198 2-fluorophenyl group Chemical group [H]C1=C([H])C(F)=C(*)C([H])=C1[H] 0.000 description 1
- 125000000954 2-hydroxyethyl group Chemical group [H]C([*])([H])C([H])([H])O[H] 0.000 description 1
- LXBGSDVWAMZHDD-UHFFFAOYSA-N 2-methyl-1h-imidazole Chemical compound CC1=NC=CN1 LXBGSDVWAMZHDD-UHFFFAOYSA-N 0.000 description 1
- FZIIBDOXPQOKBP-UHFFFAOYSA-N 2-methyloxetane Chemical compound CC1CCO1 FZIIBDOXPQOKBP-UHFFFAOYSA-N 0.000 description 1
- ZCUJYXPAKHMBAZ-UHFFFAOYSA-N 2-phenyl-1h-imidazole Chemical compound C1=CNC(C=2C=CC=CC=2)=N1 ZCUJYXPAKHMBAZ-UHFFFAOYSA-N 0.000 description 1
- CPFRUJWRXRBCLM-UHFFFAOYSA-N 2-phenylethyl cyanate Chemical compound N#COCCC1=CC=CC=C1 CPFRUJWRXRBCLM-UHFFFAOYSA-N 0.000 description 1
- CPBJMKMKNCRKQB-UHFFFAOYSA-N 3,3-bis(4-hydroxy-3-methylphenyl)-2-benzofuran-1-one Chemical compound C1=C(O)C(C)=CC(C2(C3=CC=CC=C3C(=O)O2)C=2C=C(C)C(O)=CC=2)=C1 CPBJMKMKNCRKQB-UHFFFAOYSA-N 0.000 description 1
- CXURGFRDGROIKG-UHFFFAOYSA-N 3,3-bis(chloromethyl)oxetane Chemical compound ClCC1(CCl)COC1 CXURGFRDGROIKG-UHFFFAOYSA-N 0.000 description 1
- RVGLUKRYMXEQAH-UHFFFAOYSA-N 3,3-dimethyloxetane Chemical compound CC1(C)COC1 RVGLUKRYMXEQAH-UHFFFAOYSA-N 0.000 description 1
- BGDOLELXXPTPFX-UHFFFAOYSA-N 3,4-dihydro-2h-1,2-benzoxazine Chemical group C1=CC=C2ONCCC2=C1 BGDOLELXXPTPFX-UHFFFAOYSA-N 0.000 description 1
- UIDDPPKZYZTEGS-UHFFFAOYSA-N 3-(2-ethyl-4-methylimidazol-1-yl)propanenitrile Chemical compound CCC1=NC(C)=CN1CCC#N UIDDPPKZYZTEGS-UHFFFAOYSA-N 0.000 description 1
- BVYPJEBKDLFIDL-UHFFFAOYSA-N 3-(2-phenylimidazol-1-yl)propanenitrile Chemical compound N#CCCN1C=CN=C1C1=CC=CC=C1 BVYPJEBKDLFIDL-UHFFFAOYSA-N 0.000 description 1
- YRNCACXJEDBLDE-UHFFFAOYSA-N 3-(methoxymethyl)-3-methyloxetane Chemical compound COCC1(C)COC1 YRNCACXJEDBLDE-UHFFFAOYSA-N 0.000 description 1
- VXPSQDAMFATNNG-UHFFFAOYSA-N 3-[2-(2,5-dioxopyrrol-3-yl)phenyl]pyrrole-2,5-dione Chemical compound O=C1NC(=O)C(C=2C(=CC=CC=2)C=2C(NC(=O)C=2)=O)=C1 VXPSQDAMFATNNG-UHFFFAOYSA-N 0.000 description 1
- MOSSLXZUUKTULI-UHFFFAOYSA-N 3-[3-(2,5-dioxopyrrol-3-yl)-4-methylphenyl]pyrrole-2,5-dione Chemical compound CC1=CC=C(C=2C(NC(=O)C=2)=O)C=C1C1=CC(=O)NC1=O MOSSLXZUUKTULI-UHFFFAOYSA-N 0.000 description 1
- VCFJLCCBKJNFKQ-UHFFFAOYSA-N 3-[4-(2,5-dioxopyrrol-3-yl)phenyl]pyrrole-2,5-dione Chemical compound O=C1NC(=O)C(C=2C=CC(=CC=2)C=2C(NC(=O)C=2)=O)=C1 VCFJLCCBKJNFKQ-UHFFFAOYSA-N 0.000 description 1
- GSHIDYWGMUZEOL-UHFFFAOYSA-N 3-[[2-[(2,5-dioxopyrrol-3-yl)methyl]phenyl]methyl]pyrrole-2,5-dione Chemical compound O=C1NC(=O)C(CC=2C(=CC=CC=2)CC=2C(NC(=O)C=2)=O)=C1 GSHIDYWGMUZEOL-UHFFFAOYSA-N 0.000 description 1
- RVFQOYLBPXHFKR-UHFFFAOYSA-N 3-[[3-[(2,5-dioxopyrrol-3-yl)methyl]phenyl]methyl]pyrrole-2,5-dione Chemical compound O=C1NC(=O)C(CC=2C=C(CC=3C(NC(=O)C=3)=O)C=CC=2)=C1 RVFQOYLBPXHFKR-UHFFFAOYSA-N 0.000 description 1
- KAWODUAUIIOGGH-UHFFFAOYSA-N 3-[[4-[(2,5-dioxopyrrol-3-yl)methyl]phenyl]methyl]pyrrole-2,5-dione Chemical compound O=C1NC(=O)C(CC=2C=CC(CC=3C(NC(=O)C=3)=O)=CC=2)=C1 KAWODUAUIIOGGH-UHFFFAOYSA-N 0.000 description 1
- 125000004180 3-fluorophenyl group Chemical group [H]C1=C([H])C(*)=C([H])C(F)=C1[H] 0.000 description 1
- ISLXKUPMVIEQGO-UHFFFAOYSA-N 3-methyl-1-[4-(4-propylphenoxy)phenyl]pyrrole-2,5-dione Chemical compound C1(C(C)=CC(N1C1=CC=C(OC2=CC=C(C=C2)CCC)C=C1)=O)=O ISLXKUPMVIEQGO-UHFFFAOYSA-N 0.000 description 1
- VJQHJNIGWOABDZ-UHFFFAOYSA-N 3-methyloxetane Chemical compound CC1COC1 VJQHJNIGWOABDZ-UHFFFAOYSA-N 0.000 description 1
- UPMLOUAZCHDJJD-UHFFFAOYSA-N 4,4'-Diphenylmethane Diisocyanate Chemical compound C1=CC(N=C=O)=CC=C1CC1=CC=C(N=C=O)C=C1 UPMLOUAZCHDJJD-UHFFFAOYSA-N 0.000 description 1
- 125000001255 4-fluorophenyl group Chemical group [H]C1=C([H])C(*)=C([H])C([H])=C1F 0.000 description 1
- RGHHSNMVTDWUBI-UHFFFAOYSA-N 4-hydroxybenzaldehyde Chemical compound OC1=CC=C(C=O)C=C1 RGHHSNMVTDWUBI-UHFFFAOYSA-N 0.000 description 1
- VPBBZXIPVBVLFZ-UHFFFAOYSA-N 4-phenylbutyl cyanate Chemical compound O(C#N)CCCCC1=CC=CC=C1 VPBBZXIPVBVLFZ-UHFFFAOYSA-N 0.000 description 1
- ULKLGIFJWFIQFF-UHFFFAOYSA-N 5K8XI641G3 Chemical compound CCC1=NC=C(C)N1 ULKLGIFJWFIQFF-UHFFFAOYSA-N 0.000 description 1
- KIFWEUZANBOIJW-UHFFFAOYSA-N 8-phenyloctyl cyanate Chemical compound N#COCCCCCCCCC1=CC=CC=C1 KIFWEUZANBOIJW-UHFFFAOYSA-N 0.000 description 1
- LPEKGGXMPWTOCB-UHFFFAOYSA-N 8beta-(2,3-epoxy-2-methylbutyryloxy)-14-acetoxytithifolin Natural products COC(=O)C(C)O LPEKGGXMPWTOCB-UHFFFAOYSA-N 0.000 description 1
- JWBBBJFKVIQIFQ-UHFFFAOYSA-N 9-phenylnonyl cyanate Chemical compound O(C#N)CCCCCCCCCC1=CC=CC=C1 JWBBBJFKVIQIFQ-UHFFFAOYSA-N 0.000 description 1
- 229910052582 BN Inorganic materials 0.000 description 1
- 239000004342 Benzoyl peroxide Substances 0.000 description 1
- OMPJBNCRMGITSC-UHFFFAOYSA-N Benzoylperoxide Chemical compound C=1C=CC=CC=1C(=O)OOC(=O)C1=CC=CC=C1 OMPJBNCRMGITSC-UHFFFAOYSA-N 0.000 description 1
- ZOXJGFHDIHLPTG-UHFFFAOYSA-N Boron Chemical compound [B] ZOXJGFHDIHLPTG-UHFFFAOYSA-N 0.000 description 1
- PZNSFCLAULLKQX-UHFFFAOYSA-N Boron nitride Chemical compound N#B PZNSFCLAULLKQX-UHFFFAOYSA-N 0.000 description 1
- DKPFZGUDAPQIHT-UHFFFAOYSA-N Butyl acetate Natural products CCCCOC(C)=O DKPFZGUDAPQIHT-UHFFFAOYSA-N 0.000 description 1
- 229920000049 Carbon (fiber) Polymers 0.000 description 1
- NPBVQXIMTZKSBA-UHFFFAOYSA-N Chavibetol Natural products COC1=CC=C(CC=C)C=C1O NPBVQXIMTZKSBA-UHFFFAOYSA-N 0.000 description 1
- 229920000742 Cotton Polymers 0.000 description 1
- LCGLNKUTAGEVQW-UHFFFAOYSA-N Dimethyl ether Chemical compound COC LCGLNKUTAGEVQW-UHFFFAOYSA-N 0.000 description 1
- OTMSDBZUPAUEDD-UHFFFAOYSA-N Ethane Chemical compound CC OTMSDBZUPAUEDD-UHFFFAOYSA-N 0.000 description 1
- 239000005770 Eugenol Substances 0.000 description 1
- PXGOKWXKJXAPGV-UHFFFAOYSA-N Fluorine Chemical compound FF PXGOKWXKJXAPGV-UHFFFAOYSA-N 0.000 description 1
- 238000004566 IR spectroscopy Methods 0.000 description 1
- 229910021578 Iron(III) chloride Inorganic materials 0.000 description 1
- NHTMVDHEPJAVLT-UHFFFAOYSA-N Isooctane Chemical compound CC(C)CC(C)(C)C NHTMVDHEPJAVLT-UHFFFAOYSA-N 0.000 description 1
- CERQOIWHTDAKMF-UHFFFAOYSA-M Methacrylate Chemical compound CC(=C)C([O-])=O CERQOIWHTDAKMF-UHFFFAOYSA-M 0.000 description 1
- XYVQFUJDGOBPQI-UHFFFAOYSA-N Methyl-2-hydoxyisobutyric acid Chemical compound COC(=O)C(C)(C)O XYVQFUJDGOBPQI-UHFFFAOYSA-N 0.000 description 1
- GRYLNZFGIOXLOG-UHFFFAOYSA-N Nitric acid Chemical compound O[N+]([O-])=O GRYLNZFGIOXLOG-UHFFFAOYSA-N 0.000 description 1
- IGFHQQFPSIBGKE-UHFFFAOYSA-N Nonylphenol Natural products CCCCCCCCCC1=CC=C(O)C=C1 IGFHQQFPSIBGKE-UHFFFAOYSA-N 0.000 description 1
- CPNXAOWDQMNJMA-UHFFFAOYSA-N O(C#N)C1=CC=C(C=C1)C1=C(C(=O)O)C=CC(=C1)OC#N.O(C#N)C1=CC=C(C=C1)OC(C1=CC=C(C=C1)OC#N)=O Chemical compound O(C#N)C1=CC=C(C=C1)C1=C(C(=O)O)C=CC(=C1)OC#N.O(C#N)C1=CC=C(C=C1)OC(C1=CC=C(C=C1)OC#N)=O CPNXAOWDQMNJMA-UHFFFAOYSA-N 0.000 description 1
- IPCYSKTUZUBHPN-UHFFFAOYSA-N O(C#N)C1=CC=C(C=C1)C1=C(C=CC(=C1)C(C)C)CC Chemical compound O(C#N)C1=CC=C(C=C1)C1=C(C=CC(=C1)C(C)C)CC IPCYSKTUZUBHPN-UHFFFAOYSA-N 0.000 description 1
- WDQPIBOHJHWKPK-UHFFFAOYSA-N O(C#N)C1=CC=C(C=C1)N1C(C=2C(C1=O)=CC(=CC2)OC=2C=C1C(C(=O)NC1=O)=CC2)=O Chemical compound O(C#N)C1=CC=C(C=C1)N1C(C=2C(C1=O)=CC(=CC2)OC=2C=C1C(C(=O)NC1=O)=CC2)=O WDQPIBOHJHWKPK-UHFFFAOYSA-N 0.000 description 1
- 229930040373 Paraformaldehyde Natural products 0.000 description 1
- OAICVXFJPJFONN-UHFFFAOYSA-N Phosphorus Chemical compound [P] OAICVXFJPJFONN-UHFFFAOYSA-N 0.000 description 1
- 239000004952 Polyamide Substances 0.000 description 1
- 239000004698 Polyethylene Substances 0.000 description 1
- 239000004642 Polyimide Chemical group 0.000 description 1
- 239000004793 Polystyrene Substances 0.000 description 1
- XBDQKXXYIPTUBI-UHFFFAOYSA-M Propionate Chemical compound CCC([O-])=O XBDQKXXYIPTUBI-UHFFFAOYSA-M 0.000 description 1
- UVMRYBDEERADNV-UHFFFAOYSA-N Pseudoeugenol Natural products COC1=CC(C(C)=C)=CC=C1O UVMRYBDEERADNV-UHFFFAOYSA-N 0.000 description 1
- 229910052581 Si3N4 Inorganic materials 0.000 description 1
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical compound [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 description 1
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 description 1
- 235000002597 Solanum melongena Nutrition 0.000 description 1
- 244000061458 Solanum melongena Species 0.000 description 1
- 229910021627 Tin(IV) chloride Inorganic materials 0.000 description 1
- GWEVSGVZZGPLCZ-UHFFFAOYSA-N Titan oxide Chemical compound O=[Ti]=O GWEVSGVZZGPLCZ-UHFFFAOYSA-N 0.000 description 1
- FJVCXYGDZUZPBL-UHFFFAOYSA-N [1-(2-cyanatonaphthalen-1-yl)naphthalen-2-yl] cyanate Chemical group C1=CC=C2C(C3=C4C=CC=CC4=CC=C3OC#N)=C(OC#N)C=CC2=C1 FJVCXYGDZUZPBL-UHFFFAOYSA-N 0.000 description 1
- CMHBPGYKMXHYBG-UHFFFAOYSA-N [3-(trifluoromethyl)phenyl] cyanate Chemical compound FC(F)(F)C1=CC=CC(OC#N)=C1 CMHBPGYKMXHYBG-UHFFFAOYSA-N 0.000 description 1
- SUXGZGQKIFLKOR-UHFFFAOYSA-N [4-(2-phenylpropan-2-yl)phenyl] cyanate Chemical compound C=1C=C(OC#N)C=CC=1C(C)(C)C1=CC=CC=C1 SUXGZGQKIFLKOR-UHFFFAOYSA-N 0.000 description 1
- JSYZVYJUHKETFH-UHFFFAOYSA-N [4-(4-cyanato-2,3,5,6-tetrafluorophenyl)-2,3,5,6-tetrafluorophenyl] cyanate Chemical group O(C#N)C1=C(C(=C(C(=C1F)F)C1=C(C(=C(C(=C1F)F)OC#N)F)F)F)F JSYZVYJUHKETFH-UHFFFAOYSA-N 0.000 description 1
- SNYVZKMCGVGTKN-UHFFFAOYSA-N [4-(4-cyanatophenoxy)phenyl] cyanate Chemical compound C1=CC(OC#N)=CC=C1OC1=CC=C(OC#N)C=C1 SNYVZKMCGVGTKN-UHFFFAOYSA-N 0.000 description 1
- CNUHQZDDTLOZRY-UHFFFAOYSA-N [4-(4-cyanatophenyl)sulfanylphenyl] cyanate Chemical compound C1=CC(OC#N)=CC=C1SC1=CC=C(OC#N)C=C1 CNUHQZDDTLOZRY-UHFFFAOYSA-N 0.000 description 1
- BUPOATPDNYBPMR-UHFFFAOYSA-N [4-(4-cyanatophenyl)sulfonylphenyl] cyanate Chemical compound C=1C=C(OC#N)C=CC=1S(=O)(=O)C1=CC=C(OC#N)C=C1 BUPOATPDNYBPMR-UHFFFAOYSA-N 0.000 description 1
- SYGJXLZLADSQOO-UHFFFAOYSA-N [4-(9H-fluoren-1-yl)phenyl] cyanate Chemical compound O(C#N)C1=CC=C(C=C1)C1=CC=CC=2C3=CC=CC=C3CC1=2 SYGJXLZLADSQOO-UHFFFAOYSA-N 0.000 description 1
- JNCRKOQSRHDNIO-UHFFFAOYSA-N [4-[(4-cyanato-3,5-dimethylphenyl)methyl]-2,6-dimethylphenyl] cyanate Chemical compound CC1=C(OC#N)C(C)=CC(CC=2C=C(C)C(OC#N)=C(C)C=2)=C1 JNCRKOQSRHDNIO-UHFFFAOYSA-N 0.000 description 1
- CGWCRUVZAOUGMY-UHFFFAOYSA-N [4-[(4-cyanatophenyl)-(4-phenylphenyl)methyl]phenyl] cyanate Chemical group O(C#N)C1=CC=C(C=C1)C(C1=CC=C(C=C1)C1=CC=CC=C1)C1=CC=C(C=C1)OC#N CGWCRUVZAOUGMY-UHFFFAOYSA-N 0.000 description 1
- PYICCSHCIIAPJE-UHFFFAOYSA-N [4-[(4-cyanatophenyl)-diphenylmethyl]phenyl] cyanate Chemical compound C1=CC(OC#N)=CC=C1C(C=1C=CC(OC#N)=CC=1)(C=1C=CC=CC=1)C1=CC=CC=C1 PYICCSHCIIAPJE-UHFFFAOYSA-N 0.000 description 1
- NNVFMMULSKOXSJ-UHFFFAOYSA-N [4-[(4-cyanatophenyl)-phenylmethyl]phenyl] cyanate Chemical compound C1=CC(OC#N)=CC=C1C(C=1C=CC(OC#N)=CC=1)C1=CC=CC=C1 NNVFMMULSKOXSJ-UHFFFAOYSA-N 0.000 description 1
- AWWJTNMLTCVUBS-UHFFFAOYSA-N [4-[1,1-bis(4-cyanatophenyl)ethyl]phenyl] cyanate Chemical compound C=1C=C(OC#N)C=CC=1C(C=1C=CC(OC#N)=CC=1)(C)C1=CC=C(OC#N)C=C1 AWWJTNMLTCVUBS-UHFFFAOYSA-N 0.000 description 1
- MWQTYOOOVAXQHW-UHFFFAOYSA-N [4-[1,3-bis(4-cyanatophenyl)propyl]phenyl] cyanate Chemical compound O(C#N)C1=CC=C(C=C1)C(CCC1=CC=C(C=C1)OC#N)C1=CC=C(C=C1)OC#N MWQTYOOOVAXQHW-UHFFFAOYSA-N 0.000 description 1
- XDDXAEDWFZJGSJ-UHFFFAOYSA-N [4-[1-(4-cyanatophenyl)-1-phenylethyl]phenyl] cyanate Chemical compound C=1C=C(OC#N)C=CC=1C(C=1C=CC(OC#N)=CC=1)(C)C1=CC=CC=C1 XDDXAEDWFZJGSJ-UHFFFAOYSA-N 0.000 description 1
- WHXGQAGLFTWDAD-UHFFFAOYSA-N [4-[1-(4-cyanatophenyl)-2,2-dimethylpropyl]phenyl] cyanate Chemical compound C=1C=C(OC#N)C=CC=1C(C(C)(C)C)C1=CC=C(OC#N)C=C1 WHXGQAGLFTWDAD-UHFFFAOYSA-N 0.000 description 1
- VRJVYJZCNABROM-UHFFFAOYSA-N [4-[1-(4-cyanatophenyl)-2-methylbutyl]phenyl] cyanate Chemical compound C=1C=C(OC#N)C=CC=1C(C(C)CC)C1=CC=C(OC#N)C=C1 VRJVYJZCNABROM-UHFFFAOYSA-N 0.000 description 1
- OTFDZGDENDYXCY-UHFFFAOYSA-N [4-[1-(4-cyanatophenyl)-3,3,5-trimethylcyclohexyl]phenyl] cyanate Chemical compound O(C#N)C1=CC=C(C=C1)C1(CC(CC(C1)C)(C)C)C1=CC=C(C=C1)OC#N OTFDZGDENDYXCY-UHFFFAOYSA-N 0.000 description 1
- ALTSFBKKGZFYFL-UHFFFAOYSA-N [4-[1-(4-cyanatophenyl)-3-methylbutyl]phenyl] cyanate Chemical compound C=1C=C(OC#N)C=CC=1C(CC(C)C)C1=CC=C(OC#N)C=C1 ALTSFBKKGZFYFL-UHFFFAOYSA-N 0.000 description 1
- CUVGYGLIVYYKLM-UHFFFAOYSA-N [4-[1-(4-cyanatophenyl)-3-oxo-2-benzofuran-1-yl]phenyl] cyanate Chemical compound C12=CC=CC=C2C(=O)OC1(C=1C=CC(OC#N)=CC=1)C1=CC=C(OC#N)C=C1 CUVGYGLIVYYKLM-UHFFFAOYSA-N 0.000 description 1
- ZFWZYLYLPMWAQM-UHFFFAOYSA-N [4-[1-(4-cyanatophenyl)butyl]phenyl] cyanate Chemical compound C=1C=C(OC#N)C=CC=1C(CCC)C1=CC=C(OC#N)C=C1 ZFWZYLYLPMWAQM-UHFFFAOYSA-N 0.000 description 1
- RZBUVWIMPWGGRK-UHFFFAOYSA-N [4-[1-(4-cyanatophenyl)cyclohexyl]phenyl] cyanate Chemical compound C1=CC(OC#N)=CC=C1C1(C=2C=CC(OC#N)=CC=2)CCCCC1 RZBUVWIMPWGGRK-UHFFFAOYSA-N 0.000 description 1
- BWVPAGYFWDPBIW-UHFFFAOYSA-N [4-[1-(4-cyanatophenyl)cyclopentyl]phenyl] cyanate Chemical compound C1=CC(OC#N)=CC=C1C1(C=2C=CC(OC#N)=CC=2)CCCC1 BWVPAGYFWDPBIW-UHFFFAOYSA-N 0.000 description 1
- SIZDMAYTWUINIG-UHFFFAOYSA-N [4-[1-(4-cyanatophenyl)ethyl]phenyl] cyanate Chemical compound C=1C=C(OC#N)C=CC=1C(C)C1=CC=C(OC#N)C=C1 SIZDMAYTWUINIG-UHFFFAOYSA-N 0.000 description 1
- LNZIRNRHIJPNRY-UHFFFAOYSA-N [4-[1-(4-cyanatophenyl)propyl]phenyl] cyanate Chemical compound C=1C=C(OC#N)C=CC=1C(CC)C1=CC=C(OC#N)C=C1 LNZIRNRHIJPNRY-UHFFFAOYSA-N 0.000 description 1
- OXRLCQGMPCWRFK-UHFFFAOYSA-N [4-[2,2-dichloro-1-(4-cyanatophenyl)ethenyl]phenyl] cyanate Chemical group C=1C=C(OC#N)C=CC=1C(=C(Cl)Cl)C1=CC=C(OC#N)C=C1 OXRLCQGMPCWRFK-UHFFFAOYSA-N 0.000 description 1
- HMIPTJCEKMBTKV-UHFFFAOYSA-N [4-[2-(4-cyanato-3-methylphenyl)propan-2-yl]-2-methylphenyl] cyanate Chemical compound C1=C(OC#N)C(C)=CC(C(C)(C)C=2C=C(C)C(OC#N)=CC=2)=C1 HMIPTJCEKMBTKV-UHFFFAOYSA-N 0.000 description 1
- LCCSXIORAPRMIY-UHFFFAOYSA-N [4-[2-(4-cyanato-3-phenylphenyl)propan-2-yl]-2-phenylphenyl] cyanate Chemical compound C=1C=C(OC#N)C(C=2C=CC=CC=2)=CC=1C(C)(C)C(C=1)=CC=C(OC#N)C=1C1=CC=CC=C1 LCCSXIORAPRMIY-UHFFFAOYSA-N 0.000 description 1
- VJLQKGRQOAVLKN-UHFFFAOYSA-N [4-[2-(4-cyanatophenyl)-3,3-dimethylbutan-2-yl]phenyl] cyanate Chemical compound C=1C=C(OC#N)C=CC=1C(C)(C(C)(C)C)C1=CC=C(OC#N)C=C1 VJLQKGRQOAVLKN-UHFFFAOYSA-N 0.000 description 1
- LYVKPFCEPYMYIN-UHFFFAOYSA-N [4-[2-(4-cyanatophenyl)-3-methylbutan-2-yl]phenyl] cyanate Chemical compound C=1C=C(OC#N)C=CC=1C(C)(C(C)C)C1=CC=C(OC#N)C=C1 LYVKPFCEPYMYIN-UHFFFAOYSA-N 0.000 description 1
- OKOGUHSERRTCOI-UHFFFAOYSA-N [4-[2-(4-cyanatophenyl)-4-methylpentan-2-yl]phenyl] cyanate Chemical compound C=1C=C(OC#N)C=CC=1C(C)(CC(C)C)C1=CC=C(OC#N)C=C1 OKOGUHSERRTCOI-UHFFFAOYSA-N 0.000 description 1
- UPCZANMLPOWPAD-UHFFFAOYSA-N [4-[2-(4-cyanatophenyl)butan-2-yl]phenyl] cyanate Chemical compound C=1C=C(OC#N)C=CC=1C(C)(CC)C1=CC=C(OC#N)C=C1 UPCZANMLPOWPAD-UHFFFAOYSA-N 0.000 description 1
- NHZITVMFMHBTBL-UHFFFAOYSA-N [4-[2-(4-cyanatophenyl)hexan-2-yl]phenyl] cyanate Chemical compound C=1C=C(OC#N)C=CC=1C(C)(CCCC)C1=CC=C(OC#N)C=C1 NHZITVMFMHBTBL-UHFFFAOYSA-N 0.000 description 1
- KGRJSARQPLMHSD-UHFFFAOYSA-N [4-[2-(4-cyanatophenyl)pentan-2-yl]phenyl] cyanate Chemical compound C=1C=C(OC#N)C=CC=1C(C)(CCC)C1=CC=C(OC#N)C=C1 KGRJSARQPLMHSD-UHFFFAOYSA-N 0.000 description 1
- QODYBLNVIHQJIW-UHFFFAOYSA-N [4-[2-[3-[2-(4-cyanatophenyl)propan-2-yl]phenyl]propan-2-yl]phenyl] cyanate Chemical compound C=1C=CC(C(C)(C)C=2C=CC(OC#N)=CC=2)=CC=1C(C)(C)C1=CC=C(OC#N)C=C1 QODYBLNVIHQJIW-UHFFFAOYSA-N 0.000 description 1
- NNRCYOCJRLOBCE-UHFFFAOYSA-N [4-[3-(4-cyanato-3-methylphenyl)-5,7-dimethyl-1-adamantyl]-2-methylphenyl] cyanate Chemical compound CC=1C=C(C=CC1OC#N)C12CC3(CC(CC(C1)(C3)C)(C2)C)C2=CC(=C(C=C2)OC#N)C NNRCYOCJRLOBCE-UHFFFAOYSA-N 0.000 description 1
- JGZANMKVIUZPKB-UHFFFAOYSA-N [4-[3-(4-cyanatophenyl)-1-adamantyl]phenyl] cyanate Chemical compound O(C#N)C1=CC=C(C=C1)C12CC3(CC(CC(C1)C3)C2)C2=CC=C(C=C2)OC#N JGZANMKVIUZPKB-UHFFFAOYSA-N 0.000 description 1
- SEBSTXOGSGMBQA-UHFFFAOYSA-N [4-[3-(4-cyanatophenyl)-1-methyl-2-oxoindol-3-yl]phenyl] cyanate Chemical compound C12=CC=CC=C2N(C)C(=O)C1(C=1C=CC(OC#N)=CC=1)C1=CC=C(OC#N)C=C1 SEBSTXOGSGMBQA-UHFFFAOYSA-N 0.000 description 1
- KHNZTNGNKBPUEM-UHFFFAOYSA-N [4-[3-(4-cyanatophenyl)-2,2,4-trimethylpentan-3-yl]phenyl] cyanate Chemical compound C=1C=C(OC#N)C=CC=1C(C(C)(C)C)(C(C)C)C1=CC=C(OC#N)C=C1 KHNZTNGNKBPUEM-UHFFFAOYSA-N 0.000 description 1
- SABGMTGLIYMFLH-UHFFFAOYSA-N [4-[3-(4-cyanatophenyl)-2,2-dimethylpentan-3-yl]phenyl] cyanate Chemical compound C=1C=C(OC#N)C=CC=1C(C(C)(C)C)(CC)C1=CC=C(OC#N)C=C1 SABGMTGLIYMFLH-UHFFFAOYSA-N 0.000 description 1
- GSDUKIJGTSHOOD-UHFFFAOYSA-N [4-[3-(4-cyanatophenyl)-2-methylheptan-3-yl]phenyl] cyanate Chemical compound C=1C=C(OC#N)C=CC=1C(C(C)C)(CCCC)C1=CC=C(OC#N)C=C1 GSDUKIJGTSHOOD-UHFFFAOYSA-N 0.000 description 1
- BCFLPWOICZLPEV-UHFFFAOYSA-N [4-[3-(4-cyanatophenyl)-2-methylhexan-3-yl]phenyl] cyanate Chemical compound C=1C=C(OC#N)C=CC=1C(C(C)C)(CCC)C1=CC=C(OC#N)C=C1 BCFLPWOICZLPEV-UHFFFAOYSA-N 0.000 description 1
- AFESTADXJPTEQF-UHFFFAOYSA-N [4-[3-(4-cyanatophenyl)-2-phenyl-1,2-dihydroindol-3-yl]phenyl] cyanate Chemical compound C1(=CC=CC=C1)C1NC2=CC=CC=C2C1(C1=CC=C(C=C1)OC#N)C1=CC=C(C=C1)OC#N AFESTADXJPTEQF-UHFFFAOYSA-N 0.000 description 1
- IELUNLXGZJSZCC-UHFFFAOYSA-N [4-[3-(4-cyanatophenyl)-3-oxoprop-1-enyl]phenyl] cyanate Chemical compound C=1C=C(OC#N)C=CC=1C(=O)C=CC1=CC=C(OC#N)C=C1 IELUNLXGZJSZCC-UHFFFAOYSA-N 0.000 description 1
- DIXZEGPVGUCLJG-UHFFFAOYSA-N [4-[3-(4-cyanatophenyl)-5,7-dimethyl-1-adamantyl]phenyl] cyanate Chemical compound O(C#N)C1=CC=C(C=C1)C12CC3(CC(CC(C1)(C3)C)(C2)C)C2=CC=C(C=C2)OC#N DIXZEGPVGUCLJG-UHFFFAOYSA-N 0.000 description 1
- GTJHXCHRCZRACK-UHFFFAOYSA-N [4-[3-(4-cyanatophenyl)heptan-3-yl]phenyl] cyanate Chemical compound C=1C=C(OC#N)C=CC=1C(CC)(CCCC)C1=CC=C(OC#N)C=C1 GTJHXCHRCZRACK-UHFFFAOYSA-N 0.000 description 1
- SIVPMTCZSPKNJC-UHFFFAOYSA-N [4-[3-(4-cyanatophenyl)hexan-3-yl]phenyl] cyanate Chemical compound C=1C=C(OC#N)C=CC=1C(CC)(CCC)C1=CC=C(OC#N)C=C1 SIVPMTCZSPKNJC-UHFFFAOYSA-N 0.000 description 1
- ZSFDETFCXJDSDY-UHFFFAOYSA-N [4-[3-(4-cyanatophenyl)octan-3-yl]phenyl] cyanate Chemical compound C=1C=C(OC#N)C=CC=1C(CC)(CCCCC)C1=CC=C(OC#N)C=C1 ZSFDETFCXJDSDY-UHFFFAOYSA-N 0.000 description 1
- VCTHWSNFKPDIFC-UHFFFAOYSA-N [4-[4-(4-cyanatophenyl)-3-methylheptan-4-yl]phenyl] cyanate Chemical compound C=1C=C(OC#N)C=CC=1C(C(C)CC)(CCC)C1=CC=C(OC#N)C=C1 VCTHWSNFKPDIFC-UHFFFAOYSA-N 0.000 description 1
- NRAWVVBFCLDHDQ-UHFFFAOYSA-N [4-[9-(4-cyanato-3-phenylphenyl)fluoren-9-yl]-2-phenylphenyl] cyanate Chemical compound N#COC1=CC=C(C2(C3=CC=CC=C3C3=CC=CC=C32)C=2C=C(C(OC#N)=CC=2)C=2C=CC=CC=2)C=C1C1=CC=CC=C1 NRAWVVBFCLDHDQ-UHFFFAOYSA-N 0.000 description 1
- RMPWNGZEITYIQC-UHFFFAOYSA-N [4-[[4,6-bis(4-cyanato-N-methylanilino)-1,3,5-triazin-2-yl]-methylamino]phenyl] cyanate Chemical compound CN(C1=CC=C(C=C1)OC#N)C1=NC(=NC(=N1)N(C1=CC=C(C=C1)OC#N)C)N(C1=CC=C(C=C1)OC#N)C RMPWNGZEITYIQC-UHFFFAOYSA-N 0.000 description 1
- HBZHCQUDVXCPJK-UHFFFAOYSA-N [4-[[4-(4-cyanato-N-methylanilino)-6-(N-methylanilino)-1,3,5-triazin-2-yl]-methylamino]phenyl] cyanate Chemical compound CN(C1=CC=C(C=C1)OC#N)C1=NC(=NC(=N1)N(C1=CC=C(C=C1)OC#N)C)N(C1=CC=CC=C1)C HBZHCQUDVXCPJK-UHFFFAOYSA-N 0.000 description 1
- KHHRTIJOAOQNBD-UHFFFAOYSA-N [4-[bis(4-cyanatophenyl)methyl]phenyl] cyanate Chemical compound C1=CC(OC#N)=CC=C1C(C=1C=CC(OC#N)=CC=1)C1=CC=C(OC#N)C=C1 KHHRTIJOAOQNBD-UHFFFAOYSA-N 0.000 description 1
- BAQMVMPLBZLJLP-UHFFFAOYSA-N [4-[tris(4-cyanatophenyl)methyl]phenyl] cyanate Chemical compound O(C#N)C1=CC=C(C=C1)C(C1=CC=C(C=C1)OC#N)(C1=CC=C(C=C1)OC#N)C1=CC=C(C=C1)OC#N BAQMVMPLBZLJLP-UHFFFAOYSA-N 0.000 description 1
- CFUMBHCUWAMIBK-UHFFFAOYSA-N [B+3].[O-]B([O-])[O-] Chemical class [B+3].[O-]B([O-])[O-] CFUMBHCUWAMIBK-UHFFFAOYSA-N 0.000 description 1
- XKTUVNRQONVSGB-UHFFFAOYSA-N [C-]#N.N#CC#N Chemical compound [C-]#N.N#CC#N XKTUVNRQONVSGB-UHFFFAOYSA-N 0.000 description 1
- KXKVLQRXCPHEJC-UHFFFAOYSA-N acetic acid trimethyl ester Natural products COC(C)=O KXKVLQRXCPHEJC-UHFFFAOYSA-N 0.000 description 1
- KSTVMGTVOPUBDE-UHFFFAOYSA-N acetyl chloride;pyridine Chemical compound CC(Cl)=O.C1=CC=NC=C1 KSTVMGTVOPUBDE-UHFFFAOYSA-N 0.000 description 1
- 239000003377 acid catalyst Substances 0.000 description 1
- 239000003929 acidic solution Substances 0.000 description 1
- 150000007513 acids Chemical class 0.000 description 1
- 150000001298 alcohols Chemical class 0.000 description 1
- 125000001931 aliphatic group Chemical group 0.000 description 1
- 150000001338 aliphatic hydrocarbons Chemical class 0.000 description 1
- 229910052783 alkali metal Inorganic materials 0.000 description 1
- 150000008044 alkali metal hydroxides Chemical class 0.000 description 1
- 150000001340 alkali metals Chemical class 0.000 description 1
- 125000004849 alkoxymethyl group Chemical group 0.000 description 1
- 150000003973 alkyl amines Chemical class 0.000 description 1
- 125000002947 alkylene group Chemical group 0.000 description 1
- 229910052782 aluminium Inorganic materials 0.000 description 1
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 1
- WNROFYMDJYEPJX-UHFFFAOYSA-K aluminium hydroxide Chemical compound [OH-].[OH-].[OH-].[Al+3] WNROFYMDJYEPJX-UHFFFAOYSA-K 0.000 description 1
- 150000001412 amines Chemical class 0.000 description 1
- 238000005349 anion exchange Methods 0.000 description 1
- 239000002518 antifoaming agent Substances 0.000 description 1
- 239000003963 antioxidant agent Substances 0.000 description 1
- 230000003078 antioxidant effect Effects 0.000 description 1
- 229920006231 aramid fiber Polymers 0.000 description 1
- 150000004982 aromatic amines Chemical class 0.000 description 1
- QBLDFAIABQKINO-UHFFFAOYSA-N barium borate Chemical compound [Ba+2].[O-]B=O.[O-]B=O QBLDFAIABQKINO-UHFFFAOYSA-N 0.000 description 1
- JRPBQTZRNDNNOP-UHFFFAOYSA-N barium titanate Chemical compound [Ba+2].[Ba+2].[O-][Ti]([O-])([O-])[O-] JRPBQTZRNDNNOP-UHFFFAOYSA-N 0.000 description 1
- 229910002113 barium titanate Inorganic materials 0.000 description 1
- UMIVXZPTRXBADB-UHFFFAOYSA-N benzocyclobutene Chemical compound C1=CC=C2CCC2=C1 UMIVXZPTRXBADB-UHFFFAOYSA-N 0.000 description 1
- 235000019400 benzoyl peroxide Nutrition 0.000 description 1
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 description 1
- 125000006267 biphenyl group Chemical group 0.000 description 1
- OZGHELYGAUYWFC-UHFFFAOYSA-N bis(4-cyanatophenyl) carbonate Chemical compound C=1C=C(OC#N)C=CC=1OC(=O)OC1=CC=C(OC#N)C=C1 OZGHELYGAUYWFC-UHFFFAOYSA-N 0.000 description 1
- 229910001593 boehmite Inorganic materials 0.000 description 1
- 229910021538 borax Inorganic materials 0.000 description 1
- 229910052796 boron Inorganic materials 0.000 description 1
- 229910000019 calcium carbonate Inorganic materials 0.000 description 1
- AXCZMVOFGPJBDE-UHFFFAOYSA-L calcium dihydroxide Chemical compound [OH-].[OH-].[Ca+2] AXCZMVOFGPJBDE-UHFFFAOYSA-L 0.000 description 1
- 239000000920 calcium hydroxide Substances 0.000 description 1
- 229910001861 calcium hydroxide Inorganic materials 0.000 description 1
- GBAOBIBJACZTNA-UHFFFAOYSA-L calcium sulfite Chemical compound [Ca+2].[O-]S([O-])=O GBAOBIBJACZTNA-UHFFFAOYSA-L 0.000 description 1
- 239000004917 carbon fiber Substances 0.000 description 1
- 150000004649 carbonic acid derivatives Chemical class 0.000 description 1
- 238000003763 carbonization Methods 0.000 description 1
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 1
- 230000003197 catalytic effect Effects 0.000 description 1
- 238000005341 cation exchange Methods 0.000 description 1
- 150000001768 cations Chemical class 0.000 description 1
- 239000007795 chemical reaction product Substances 0.000 description 1
- 239000011248 coating agent Substances 0.000 description 1
- 238000000576 coating method Methods 0.000 description 1
- 238000004440 column chromatography Methods 0.000 description 1
- 238000009841 combustion method Methods 0.000 description 1
- 238000002485 combustion reaction Methods 0.000 description 1
- 230000000052 comparative effect Effects 0.000 description 1
- 229940125904 compound 1 Drugs 0.000 description 1
- 229940125782 compound 2 Drugs 0.000 description 1
- 230000006835 compression Effects 0.000 description 1
- 238000007906 compression Methods 0.000 description 1
- 239000012141 concentrate Substances 0.000 description 1
- 238000007796 conventional method Methods 0.000 description 1
- 238000001816 cooling Methods 0.000 description 1
- 229940120693 copper naphthenate Drugs 0.000 description 1
- PMHQVHHXPFUNSP-UHFFFAOYSA-M copper(1+);methylsulfanylmethane;bromide Chemical compound Br[Cu].CSC PMHQVHHXPFUNSP-UHFFFAOYSA-M 0.000 description 1
- SEVNKWFHTNVOLD-UHFFFAOYSA-L copper;3-(4-ethylcyclohexyl)propanoate;3-(3-ethylcyclopentyl)propanoate Chemical compound [Cu+2].CCC1CCC(CCC([O-])=O)C1.CCC1CCC(CCC([O-])=O)CC1 SEVNKWFHTNVOLD-UHFFFAOYSA-L 0.000 description 1
- 239000012043 crude product Substances 0.000 description 1
- ATDGTVJJHBUTRL-UHFFFAOYSA-N cyanogen bromide Chemical compound BrC#N ATDGTVJJHBUTRL-UHFFFAOYSA-N 0.000 description 1
- 125000000753 cycloalkyl group Chemical group 0.000 description 1
- 125000002993 cycloalkylene group Chemical group 0.000 description 1
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 1
- 125000001511 cyclopentyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 1
- 125000004979 cyclopentylene group Chemical group 0.000 description 1
- 238000000354 decomposition reaction Methods 0.000 description 1
- 238000007872 degassing Methods 0.000 description 1
- 230000018044 dehydration Effects 0.000 description 1
- 238000006297 dehydration reaction Methods 0.000 description 1
- 230000006866 deterioration Effects 0.000 description 1
- LSXWFXONGKSEMY-UHFFFAOYSA-N di-tert-butyl peroxide Chemical compound CC(C)(C)OOC(C)(C)C LSXWFXONGKSEMY-UHFFFAOYSA-N 0.000 description 1
- GDVKFRBCXAPAQJ-UHFFFAOYSA-A dialuminum;hexamagnesium;carbonate;hexadecahydroxide Chemical compound [OH-].[OH-].[OH-].[OH-].[OH-].[OH-].[OH-].[OH-].[OH-].[OH-].[OH-].[OH-].[OH-].[OH-].[OH-].[OH-].[Mg+2].[Mg+2].[Mg+2].[Mg+2].[Mg+2].[Mg+2].[Al+3].[Al+3].[O-]C([O-])=O GDVKFRBCXAPAQJ-UHFFFAOYSA-A 0.000 description 1
- 125000004188 dichlorophenyl group Chemical group 0.000 description 1
- QGBSISYHAICWAH-UHFFFAOYSA-N dicyandiamide Chemical compound NC(N)=NC#N QGBSISYHAICWAH-UHFFFAOYSA-N 0.000 description 1
- JVSWJIKNEAIKJW-UHFFFAOYSA-N dimethyl-hexane Natural products CCCCCC(C)C JVSWJIKNEAIKJW-UHFFFAOYSA-N 0.000 description 1
- XXBDWLFCJWSEKW-UHFFFAOYSA-N dimethylbenzylamine Chemical compound CN(C)CC1=CC=CC=C1 XXBDWLFCJWSEKW-UHFFFAOYSA-N 0.000 description 1
- 239000002270 dispersing agent Substances 0.000 description 1
- ODQWQRRAPPTVAG-GZTJUZNOSA-N doxepin Chemical compound C1OC2=CC=CC=C2C(=C/CCN(C)C)/C2=CC=CC=C21 ODQWQRRAPPTVAG-GZTJUZNOSA-N 0.000 description 1
- 239000000975 dye Substances 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 230000002708 enhancing effect Effects 0.000 description 1
- 238000003912 environmental pollution Methods 0.000 description 1
- 125000003700 epoxy group Chemical group 0.000 description 1
- 125000001033 ether group Chemical group 0.000 description 1
- 125000001301 ethoxy group Chemical group [H]C([H])([H])C([H])([H])O* 0.000 description 1
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 1
- 229940116333 ethyl lactate Drugs 0.000 description 1
- 125000000816 ethylene group Chemical group [H]C([H])([*:1])C([H])([H])[*:2] 0.000 description 1
- 229960002217 eugenol Drugs 0.000 description 1
- 238000011156 evaluation Methods 0.000 description 1
- 238000000605 extraction Methods 0.000 description 1
- 239000004744 fabric Substances 0.000 description 1
- 238000009730 filament winding Methods 0.000 description 1
- 238000011049 filling Methods 0.000 description 1
- 238000001914 filtration Methods 0.000 description 1
- 239000012467 final product Substances 0.000 description 1
- 150000002220 fluorenes Chemical class 0.000 description 1
- 239000011737 fluorine Substances 0.000 description 1
- 239000006260 foam Substances 0.000 description 1
- CBYZIWCZNMOEAV-UHFFFAOYSA-N formaldehyde;naphthalene Chemical class O=C.C1=CC=CC2=CC=CC=C21 CBYZIWCZNMOEAV-UHFFFAOYSA-N 0.000 description 1
- 125000000524 functional group Chemical group 0.000 description 1
- 239000005350 fused silica glass Substances 0.000 description 1
- 238000001879 gelation Methods 0.000 description 1
- 238000007429 general method Methods 0.000 description 1
- 229910001679 gibbsite Inorganic materials 0.000 description 1
- 150000002366 halogen compounds Chemical class 0.000 description 1
- 238000009787 hand lay-up Methods 0.000 description 1
- 125000005842 heteroatom Chemical group 0.000 description 1
- FUZZWVXGSFPDMH-UHFFFAOYSA-N hexanoic acid Chemical compound CCCCCC(O)=O FUZZWVXGSFPDMH-UHFFFAOYSA-N 0.000 description 1
- 125000004051 hexyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 229910001701 hydrotalcite Inorganic materials 0.000 description 1
- 229960001545 hydrotalcite Drugs 0.000 description 1
- XLYOFNOQVPJJNP-UHFFFAOYSA-M hydroxide Chemical compound [OH-] XLYOFNOQVPJJNP-UHFFFAOYSA-M 0.000 description 1
- 150000004679 hydroxides Chemical class 0.000 description 1
- FAHBNUUHRFUEAI-UHFFFAOYSA-M hydroxidooxidoaluminium Chemical compound O[Al]=O FAHBNUUHRFUEAI-UHFFFAOYSA-M 0.000 description 1
- 125000005027 hydroxyaryl group Chemical group 0.000 description 1
- 125000004029 hydroxymethyl group Chemical group [H]OC([H])([H])* 0.000 description 1
- RAXXELZNTBOGNW-UHFFFAOYSA-N imidazole Substances C1=CNC=N1 RAXXELZNTBOGNW-UHFFFAOYSA-N 0.000 description 1
- YAMHXTCMCPHKLN-UHFFFAOYSA-N imidazolidin-2-one Chemical compound O=C1NCCN1 YAMHXTCMCPHKLN-UHFFFAOYSA-N 0.000 description 1
- 150000003949 imides Chemical group 0.000 description 1
- 125000001841 imino group Chemical group [H]N=* 0.000 description 1
- 230000001771 impaired effect Effects 0.000 description 1
- 239000012535 impurity Substances 0.000 description 1
- 238000001802 infusion Methods 0.000 description 1
- 239000003112 inhibitor Substances 0.000 description 1
- 239000003999 initiator Substances 0.000 description 1
- 239000012784 inorganic fiber Substances 0.000 description 1
- 229910052500 inorganic mineral Inorganic materials 0.000 description 1
- 238000009413 insulation Methods 0.000 description 1
- 229940079865 intestinal antiinfectives imidazole derivative Drugs 0.000 description 1
- 238000004255 ion exchange chromatography Methods 0.000 description 1
- RBTARNINKXHZNM-UHFFFAOYSA-K iron trichloride Chemical compound Cl[Fe](Cl)Cl RBTARNINKXHZNM-UHFFFAOYSA-K 0.000 description 1
- DLAPQHBZCAAVPQ-UHFFFAOYSA-N iron;pentane-2,4-dione Chemical compound [Fe].CC(=O)CC(C)=O DLAPQHBZCAAVPQ-UHFFFAOYSA-N 0.000 description 1
- 125000002510 isobutoxy group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])O* 0.000 description 1
- 125000000959 isobutyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 description 1
- 125000001972 isopentyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000003253 isopropoxy group Chemical group [H]C([H])([H])C([H])(O*)C([H])([H])[H] 0.000 description 1
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 239000004310 lactic acid Substances 0.000 description 1
- 235000014655 lactic acid Nutrition 0.000 description 1
- 239000000314 lubricant Substances 0.000 description 1
- 125000000040 m-tolyl group Chemical group [H]C1=C([H])C(*)=C([H])C(=C1[H])C([H])([H])[H] 0.000 description 1
- ZLNQQNXFFQJAID-UHFFFAOYSA-L magnesium carbonate Chemical compound [Mg+2].[O-]C([O-])=O ZLNQQNXFFQJAID-UHFFFAOYSA-L 0.000 description 1
- 239000001095 magnesium carbonate Substances 0.000 description 1
- 229910000021 magnesium carbonate Inorganic materials 0.000 description 1
- VTHJTEIRLNZDEV-UHFFFAOYSA-L magnesium dihydroxide Chemical compound [OH-].[OH-].[Mg+2] VTHJTEIRLNZDEV-UHFFFAOYSA-L 0.000 description 1
- 239000000347 magnesium hydroxide Substances 0.000 description 1
- 229910001862 magnesium hydroxide Inorganic materials 0.000 description 1
- 239000000395 magnesium oxide Substances 0.000 description 1
- CPLXHLVBOLITMK-UHFFFAOYSA-N magnesium oxide Inorganic materials [Mg]=O CPLXHLVBOLITMK-UHFFFAOYSA-N 0.000 description 1
- 229910052943 magnesium sulfate Inorganic materials 0.000 description 1
- 235000019341 magnesium sulphate Nutrition 0.000 description 1
- AXZKOIWUVFPNLO-UHFFFAOYSA-N magnesium;oxygen(2-) Chemical compound [O-2].[Mg+2] AXZKOIWUVFPNLO-UHFFFAOYSA-N 0.000 description 1
- 125000005439 maleimidyl group Chemical group C1(C=CC(N1*)=O)=O 0.000 description 1
- 238000000691 measurement method Methods 0.000 description 1
- 238000002844 melting Methods 0.000 description 1
- 230000008018 melting Effects 0.000 description 1
- 150000001247 metal acetylides Chemical class 0.000 description 1
- UZKWTJUDCOPSNM-UHFFFAOYSA-N methoxybenzene Substances CCCCOC=C UZKWTJUDCOPSNM-UHFFFAOYSA-N 0.000 description 1
- BDJSOPWXYLFTNW-UHFFFAOYSA-N methyl 3-methoxypropanoate Chemical compound COCCC(=O)OC BDJSOPWXYLFTNW-UHFFFAOYSA-N 0.000 description 1
- SODPLIPUBHWQPC-UHFFFAOYSA-N methyl 4-cyanatobenzoate Chemical compound COC(=O)C1=CC=C(OC#N)C=C1 SODPLIPUBHWQPC-UHFFFAOYSA-N 0.000 description 1
- 229940057867 methyl lactate Drugs 0.000 description 1
- 239000010445 mica Substances 0.000 description 1
- 229910052618 mica group Inorganic materials 0.000 description 1
- 239000011707 mineral Substances 0.000 description 1
- 235000010755 mineral Nutrition 0.000 description 1
- 229910000476 molybdenum oxide Inorganic materials 0.000 description 1
- JAOPKYRWYXCGOQ-UHFFFAOYSA-N n,n-dimethyl-1-(4-methylphenyl)methanamine Chemical compound CN(C)CC1=CC=C(C)C=C1 JAOPKYRWYXCGOQ-UHFFFAOYSA-N 0.000 description 1
- GEMHFKXPOCTAIP-UHFFFAOYSA-N n,n-dimethyl-n'-phenylcarbamimidoyl chloride Chemical compound CN(C)C(Cl)=NC1=CC=CC=C1 GEMHFKXPOCTAIP-UHFFFAOYSA-N 0.000 description 1
- 125000006606 n-butoxy group Chemical group 0.000 description 1
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000000740 n-pentyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- KJPLRQFMMRYYFL-UHFFFAOYSA-N naphthalen-2-yl cyanate Chemical compound C1=CC=CC2=CC(OC#N)=CC=C21 KJPLRQFMMRYYFL-UHFFFAOYSA-N 0.000 description 1
- 125000001971 neopentyl group Chemical group [H]C([*])([H])C(C([H])([H])[H])(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 229910017604 nitric acid Inorganic materials 0.000 description 1
- 150000004767 nitrides Chemical class 0.000 description 1
- 125000000449 nitro group Chemical group [O-][N+](*)=O 0.000 description 1
- 239000012299 nitrogen atmosphere Substances 0.000 description 1
- SNQQPOLDUKLAAF-UHFFFAOYSA-N nonylphenol Chemical compound CCCCCCCCCC1=CC=CC=C1O SNQQPOLDUKLAAF-UHFFFAOYSA-N 0.000 description 1
- 125000003261 o-tolyl group Chemical group [H]C1=C([H])C(*)=C(C([H])=C1[H])C([H])([H])[H] 0.000 description 1
- 230000003287 optical effect Effects 0.000 description 1
- 235000006408 oxalic acid Nutrition 0.000 description 1
- MMCOUVMKNAHQOY-UHFFFAOYSA-L oxido carbonate Chemical compound [O-]OC([O-])=O MMCOUVMKNAHQOY-UHFFFAOYSA-L 0.000 description 1
- PQQKPALAQIIWST-UHFFFAOYSA-N oxomolybdenum Chemical compound [Mo]=O PQQKPALAQIIWST-UHFFFAOYSA-N 0.000 description 1
- RVTZCBVAJQQJTK-UHFFFAOYSA-N oxygen(2-);zirconium(4+) Chemical compound [O-2].[O-2].[Zr+4] RVTZCBVAJQQJTK-UHFFFAOYSA-N 0.000 description 1
- QBDSZLJBMIMQRS-UHFFFAOYSA-N p-Cumylphenol Chemical compound C=1C=C(O)C=CC=1C(C)(C)C1=CC=CC=C1 QBDSZLJBMIMQRS-UHFFFAOYSA-N 0.000 description 1
- 229920002866 paraformaldehyde Polymers 0.000 description 1
- WXZMFSXDPGVJKK-UHFFFAOYSA-N pentaerythritol Chemical compound OCC(CO)(CO)CO WXZMFSXDPGVJKK-UHFFFAOYSA-N 0.000 description 1
- 125000003538 pentan-3-yl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])C([H])([H])[H] 0.000 description 1
- 150000002978 peroxides Chemical class 0.000 description 1
- NOHFYJWXIIYRIP-UHFFFAOYSA-N phenyl 4-cyanatobenzoate Chemical compound C=1C=C(OC#N)C=CC=1C(=O)OC1=CC=CC=C1 NOHFYJWXIIYRIP-UHFFFAOYSA-N 0.000 description 1
- CWHFDTWZHFRTAB-UHFFFAOYSA-N phenyl cyanate Chemical compound N#COC1=CC=CC=C1 CWHFDTWZHFRTAB-UHFFFAOYSA-N 0.000 description 1
- XYFCBTPGUUZFHI-UHFFFAOYSA-O phosphonium Chemical compound [PH4+] XYFCBTPGUUZFHI-UHFFFAOYSA-O 0.000 description 1
- 239000011574 phosphorus Substances 0.000 description 1
- 150000003018 phosphorus compounds Chemical class 0.000 description 1
- 239000003504 photosensitizing agent Substances 0.000 description 1
- 229920002647 polyamide Polymers 0.000 description 1
- 229920006122 polyamide resin Polymers 0.000 description 1
- 229920000573 polyethylene Polymers 0.000 description 1
- 239000009719 polyimide resin Substances 0.000 description 1
- 229920005862 polyol Polymers 0.000 description 1
- 150000003077 polyols Chemical class 0.000 description 1
- 229920001451 polypropylene glycol Polymers 0.000 description 1
- 229920001296 polysiloxane Polymers 0.000 description 1
- 229920002223 polystyrene Polymers 0.000 description 1
- 238000003918 potentiometric titration Methods 0.000 description 1
- 238000001556 precipitation Methods 0.000 description 1
- 239000002243 precursor Substances 0.000 description 1
- 238000002360 preparation method Methods 0.000 description 1
- BDERNNFJNOPAEC-UHFFFAOYSA-N propan-1-ol Chemical compound CCCO BDERNNFJNOPAEC-UHFFFAOYSA-N 0.000 description 1
- RGBXDEHYFWDBKD-UHFFFAOYSA-N propan-2-yl propan-2-yloxy carbonate Chemical compound CC(C)OOC(=O)OC(C)C RGBXDEHYFWDBKD-UHFFFAOYSA-N 0.000 description 1
- 239000001294 propane Substances 0.000 description 1
- 235000019260 propionic acid Nutrition 0.000 description 1
- 125000002572 propoxy group Chemical group [*]OC([H])([H])C(C([H])([H])[H])([H])[H] 0.000 description 1
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- QQONPFPTGQHPMA-UHFFFAOYSA-N propylene Natural products CC=C QQONPFPTGQHPMA-UHFFFAOYSA-N 0.000 description 1
- LLHKCFNBLRBOGN-UHFFFAOYSA-N propylene glycol methyl ether acetate Chemical compound COCC(C)OC(C)=O LLHKCFNBLRBOGN-UHFFFAOYSA-N 0.000 description 1
- 238000011403 purification operation Methods 0.000 description 1
- 238000000197 pyrolysis Methods 0.000 description 1
- IUVKMZGDUIUOCP-BTNSXGMBSA-N quinbolone Chemical compound O([C@H]1CC[C@H]2[C@H]3[C@@H]([C@]4(C=CC(=O)C=C4CC3)C)CC[C@@]21C)C1=CCCC1 IUVKMZGDUIUOCP-BTNSXGMBSA-N 0.000 description 1
- 230000009257 reactivity Effects 0.000 description 1
- 238000001953 recrystallisation Methods 0.000 description 1
- 239000013557 residual solvent Substances 0.000 description 1
- 229910052702 rhenium Inorganic materials 0.000 description 1
- 150000003839 salts Chemical class 0.000 description 1
- 150000003333 secondary alcohols Chemical class 0.000 description 1
- 150000004756 silanes Chemical class 0.000 description 1
- 150000004760 silicates Chemical class 0.000 description 1
- 239000010703 silicon Substances 0.000 description 1
- 229910052710 silicon Inorganic materials 0.000 description 1
- 239000000377 silicon dioxide Substances 0.000 description 1
- HQVNEWCFYHHQES-UHFFFAOYSA-N silicon nitride Chemical compound N12[Si]34N5[Si]62N3[Si]51N64 HQVNEWCFYHHQES-UHFFFAOYSA-N 0.000 description 1
- 229910001961 silver nitrate Inorganic materials 0.000 description 1
- 229910052938 sodium sulfate Inorganic materials 0.000 description 1
- 235000011152 sodium sulphate Nutrition 0.000 description 1
- 235000010339 sodium tetraborate Nutrition 0.000 description 1
- WSFQLUVWDKCYSW-UHFFFAOYSA-M sodium;2-hydroxy-3-morpholin-4-ylpropane-1-sulfonate Chemical compound [Na+].[O-]S(=O)(=O)CC(O)CN1CCOCC1 WSFQLUVWDKCYSW-UHFFFAOYSA-M 0.000 description 1
- 229910000679 solder Inorganic materials 0.000 description 1
- 238000005476 soldering Methods 0.000 description 1
- 230000003068 static effect Effects 0.000 description 1
- 238000001256 steam distillation Methods 0.000 description 1
- VEALVRVVWBQVSL-UHFFFAOYSA-N strontium titanate Chemical compound [Sr+2].[O-][Ti]([O-])=O VEALVRVVWBQVSL-UHFFFAOYSA-N 0.000 description 1
- 150000005846 sugar alcohols Polymers 0.000 description 1
- LSNNMFCWUKXFEE-UHFFFAOYSA-L sulfite Chemical class [O-]S([O-])=O LSNNMFCWUKXFEE-UHFFFAOYSA-L 0.000 description 1
- 125000000472 sulfonyl group Chemical group *S(*)(=O)=O 0.000 description 1
- 229910052717 sulfur Inorganic materials 0.000 description 1
- 125000004434 sulfur atom Chemical group 0.000 description 1
- 150000003467 sulfuric acid derivatives Chemical class 0.000 description 1
- 239000012756 surface treatment agent Substances 0.000 description 1
- 239000004094 surface-active agent Substances 0.000 description 1
- 230000002194 synthesizing effect Effects 0.000 description 1
- 239000012209 synthetic fiber Substances 0.000 description 1
- 229920002994 synthetic fiber Polymers 0.000 description 1
- 239000000454 talc Substances 0.000 description 1
- 229910052623 talc Inorganic materials 0.000 description 1
- 239000008399 tap water Substances 0.000 description 1
- 235000020679 tap water Nutrition 0.000 description 1
- 125000004213 tert-butoxy group Chemical group [H]C([H])([H])C(O*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 150000003509 tertiary alcohols Chemical class 0.000 description 1
- 230000000930 thermomechanical effect Effects 0.000 description 1
- 229920005992 thermoplastic resin Polymers 0.000 description 1
- 229920001187 thermosetting polymer Polymers 0.000 description 1
- 239000002562 thickening agent Substances 0.000 description 1
- HPGGPRDJHPYFRM-UHFFFAOYSA-J tin(iv) chloride Chemical compound Cl[Sn](Cl)(Cl)Cl HPGGPRDJHPYFRM-UHFFFAOYSA-J 0.000 description 1
- OGIDPMRJRNCKJF-UHFFFAOYSA-N titanium oxide Inorganic materials [Ti]=O OGIDPMRJRNCKJF-UHFFFAOYSA-N 0.000 description 1
- 125000005270 trialkylamine group Chemical group 0.000 description 1
- VLCLHFYFMCKBRP-UHFFFAOYSA-N tricalcium;diborate Chemical compound [Ca+2].[Ca+2].[Ca+2].[O-]B([O-])[O-].[O-]B([O-])[O-] VLCLHFYFMCKBRP-UHFFFAOYSA-N 0.000 description 1
- IMNIMPAHZVJRPE-UHFFFAOYSA-N triethylenediamine Chemical compound C1CN2CCN1CC2 IMNIMPAHZVJRPE-UHFFFAOYSA-N 0.000 description 1
- 125000004360 trifluorophenyl group Chemical group 0.000 description 1
- 125000003258 trimethylene group Chemical group [H]C([H])([*:2])C([H])([H])C([H])([H])[*:1] 0.000 description 1
- BSVBQGMMJUBVOD-UHFFFAOYSA-N trisodium borate Chemical compound [Na+].[Na+].[Na+].[O-]B([O-])[O-] BSVBQGMMJUBVOD-UHFFFAOYSA-N 0.000 description 1
- BIKXLKXABVUSMH-UHFFFAOYSA-N trizinc;diborate Chemical compound [Zn+2].[Zn+2].[Zn+2].[O-]B([O-])[O-].[O-]B([O-])[O-] BIKXLKXABVUSMH-UHFFFAOYSA-N 0.000 description 1
- CMPGARWFYBADJI-UHFFFAOYSA-L tungstic acid Chemical compound O[W](O)(=O)=O CMPGARWFYBADJI-UHFFFAOYSA-L 0.000 description 1
- 239000006097 ultraviolet radiation absorber Substances 0.000 description 1
- 229930195735 unsaturated hydrocarbon Natural products 0.000 description 1
- 238000005292 vacuum distillation Methods 0.000 description 1
- 125000000391 vinyl group Chemical group [H]C([*])=C([H])[H] 0.000 description 1
- 125000005023 xylyl group Chemical group 0.000 description 1
- 239000011592 zinc chloride Substances 0.000 description 1
- 235000005074 zinc chloride Nutrition 0.000 description 1
- 239000011787 zinc oxide Substances 0.000 description 1
- XAEWLETZEZXLHR-UHFFFAOYSA-N zinc;dioxido(dioxo)molybdenum Chemical compound [Zn+2].[O-][Mo]([O-])(=O)=O XAEWLETZEZXLHR-UHFFFAOYSA-N 0.000 description 1
- CHJMFFKHPHCQIJ-UHFFFAOYSA-L zinc;octanoate Chemical compound [Zn+2].CCCCCCCC([O-])=O.CCCCCCCC([O-])=O CHJMFFKHPHCQIJ-UHFFFAOYSA-L 0.000 description 1
- 229910001928 zirconium oxide Inorganic materials 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G73/00—Macromolecular compounds obtained by reactions forming a linkage containing nitrogen with or without oxygen or carbon in the main chain of the macromolecule, not provided for in groups C08G12/00 - C08G71/00
- C08G73/06—Polycondensates having nitrogen-containing heterocyclic rings in the main chain of the macromolecule
- C08G73/0622—Polycondensates containing six-membered rings, not condensed with other rings, with nitrogen atoms as the only ring hetero atoms
- C08G73/0638—Polycondensates containing six-membered rings, not condensed with other rings, with nitrogen atoms as the only ring hetero atoms with at least three nitrogen atoms in the ring
- C08G73/0644—Poly(1,3,5)triazines
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C261/00—Derivatives of cyanic acid
- C07C261/02—Cyanates
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G14/00—Condensation polymers of aldehydes or ketones with two or more other monomers covered by at least two of the groups C08G8/00 - C08G12/00
- C08G14/02—Condensation polymers of aldehydes or ketones with two or more other monomers covered by at least two of the groups C08G8/00 - C08G12/00 of aldehydes
- C08G14/04—Condensation polymers of aldehydes or ketones with two or more other monomers covered by at least two of the groups C08G8/00 - C08G12/00 of aldehydes with phenols
- C08G14/12—Chemically modified polycondensates
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G59/00—Polycondensates containing more than one epoxy group per molecule; Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups
- C08G59/18—Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups ; e.g. general methods of curing
- C08G59/20—Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups ; e.g. general methods of curing characterised by the epoxy compounds used
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G61/00—Macromolecular compounds obtained by reactions forming a carbon-to-carbon link in the main chain of the macromolecule
- C08G61/02—Macromolecular compounds containing only carbon atoms in the main chain of the macromolecule, e.g. polyxylylenes
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G65/00—Macromolecular compounds obtained by reactions forming an ether link in the main chain of the macromolecule
- C08G65/02—Macromolecular compounds obtained by reactions forming an ether link in the main chain of the macromolecule from cyclic ethers by opening of the heterocyclic ring
- C08G65/04—Macromolecular compounds obtained by reactions forming an ether link in the main chain of the macromolecule from cyclic ethers by opening of the heterocyclic ring from cyclic ethers only
- C08G65/06—Cyclic ethers having no atoms other than carbon and hydrogen outside the ring
- C08G65/16—Cyclic ethers having four or more ring atoms
- C08G65/18—Oxetanes
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J5/00—Manufacture of articles or shaped materials containing macromolecular substances
- C08J5/04—Reinforcing macromolecular compounds with loose or coherent fibrous material
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J5/00—Manufacture of articles or shaped materials containing macromolecular substances
- C08J5/24—Impregnating materials with prepolymers which can be polymerised in situ, e.g. manufacture of prepregs
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J5/00—Manufacture of articles or shaped materials containing macromolecular substances
- C08J5/24—Impregnating materials with prepolymers which can be polymerised in situ, e.g. manufacture of prepregs
- C08J5/241—Impregnating materials with prepolymers which can be polymerised in situ, e.g. manufacture of prepregs using inorganic fibres
- C08J5/244—Impregnating materials with prepolymers which can be polymerised in situ, e.g. manufacture of prepregs using inorganic fibres using glass fibres
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K5/00—Use of organic ingredients
- C08K5/16—Nitrogen-containing compounds
- C08K5/29—Compounds containing one or more carbon-to-nitrogen double bonds
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L61/00—Compositions of condensation polymers of aldehydes or ketones; Compositions of derivatives of such polymers
- C08L61/04—Condensation polymers of aldehydes or ketones with phenols only
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L61/00—Compositions of condensation polymers of aldehydes or ketones; Compositions of derivatives of such polymers
- C08L61/34—Condensation polymers of aldehydes or ketones with monomers covered by at least two of the groups C08L61/04, C08L61/18 and C08L61/20
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L63/00—Compositions of epoxy resins; Compositions of derivatives of epoxy resins
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L65/00—Compositions of macromolecular compounds obtained by reactions forming a carbon-to-carbon link in the main chain; Compositions of derivatives of such polymers
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09J—ADHESIVES; NON-MECHANICAL ASPECTS OF ADHESIVE PROCESSES IN GENERAL; ADHESIVE PROCESSES NOT PROVIDED FOR ELSEWHERE; USE OF MATERIALS AS ADHESIVES
- C09J11/00—Features of adhesives not provided for in group C09J9/00, e.g. additives
- C09J11/02—Non-macromolecular additives
- C09J11/06—Non-macromolecular additives organic
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09J—ADHESIVES; NON-MECHANICAL ASPECTS OF ADHESIVE PROCESSES IN GENERAL; ADHESIVE PROCESSES NOT PROVIDED FOR ELSEWHERE; USE OF MATERIALS AS ADHESIVES
- C09J161/00—Adhesives based on condensation polymers of aldehydes or ketones; Adhesives based on derivatives of such polymers
- C09J161/04—Condensation polymers of aldehydes or ketones with phenols only
- C09J161/06—Condensation polymers of aldehydes or ketones with phenols only of aldehydes with phenols
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09J—ADHESIVES; NON-MECHANICAL ASPECTS OF ADHESIVE PROCESSES IN GENERAL; ADHESIVE PROCESSES NOT PROVIDED FOR ELSEWHERE; USE OF MATERIALS AS ADHESIVES
- C09J163/00—Adhesives based on epoxy resins; Adhesives based on derivatives of epoxy resins
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01L—SEMICONDUCTOR DEVICES NOT COVERED BY CLASS H10
- H01L23/00—Details of semiconductor or other solid state devices
- H01L23/12—Mountings, e.g. non-detachable insulating substrates
- H01L23/14—Mountings, e.g. non-detachable insulating substrates characterised by the material or its electrical properties
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01L—SEMICONDUCTOR DEVICES NOT COVERED BY CLASS H10
- H01L23/00—Details of semiconductor or other solid state devices
- H01L23/28—Encapsulations, e.g. encapsulating layers, coatings, e.g. for protection
- H01L23/29—Encapsulations, e.g. encapsulating layers, coatings, e.g. for protection characterised by the material, e.g. carbon
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01L—SEMICONDUCTOR DEVICES NOT COVERED BY CLASS H10
- H01L23/00—Details of semiconductor or other solid state devices
- H01L23/28—Encapsulations, e.g. encapsulating layers, coatings, e.g. for protection
- H01L23/29—Encapsulations, e.g. encapsulating layers, coatings, e.g. for protection characterised by the material, e.g. carbon
- H01L23/293—Organic, e.g. plastic
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01L—SEMICONDUCTOR DEVICES NOT COVERED BY CLASS H10
- H01L23/00—Details of semiconductor or other solid state devices
- H01L23/28—Encapsulations, e.g. encapsulating layers, coatings, e.g. for protection
- H01L23/31—Encapsulations, e.g. encapsulating layers, coatings, e.g. for protection characterised by the arrangement or shape
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G73/00—Macromolecular compounds obtained by reactions forming a linkage containing nitrogen with or without oxygen or carbon in the main chain of the macromolecule, not provided for in groups C08G12/00 - C08G71/00
- C08G73/06—Polycondensates having nitrogen-containing heterocyclic rings in the main chain of the macromolecule
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09J—ADHESIVES; NON-MECHANICAL ASPECTS OF ADHESIVE PROCESSES IN GENERAL; ADHESIVE PROCESSES NOT PROVIDED FOR ELSEWHERE; USE OF MATERIALS AS ADHESIVES
- C09J179/00—Adhesives based on macromolecular compounds obtained by reactions forming in the main chain of the macromolecule a linkage containing nitrogen, with or without oxygen, or carbon only, not provided for in groups C09J161/00 - C09J177/00
- C09J179/04—Polycondensates having nitrogen-containing heterocyclic rings in the main chain; Polyhydrazides; Polyamide acids or similar polyimide precursors
Definitions
- the present invention relates to a cyanate ester compound, a curable resin composition containing the compound, and a cured product thereof.
- the cyanate ester compound is a compound that generates a triazine ring upon curing.
- Cyanate ester compounds are widely used as raw materials for various functional polymer materials such as structural composite materials, adhesives, electrical insulating materials, and electrical and electronic parts due to their high heat resistance and excellent electrical properties. .
- functional polymer materials such as structural composite materials, adhesives, electrical insulating materials, and electrical and electronic parts due to their high heat resistance and excellent electrical properties.
- functional polymer materials such as structural composite materials, adhesives, electrical insulating materials, and electrical and electronic parts due to their high heat resistance and excellent electrical properties.
- the requirements for various physical properties of functional polymer materials have become increasingly severe. Examples of such physical properties include flame retardancy, heat resistance, low thermal expansion coefficient, low water absorption, low dielectric constant, low dielectric loss tangent, weather resistance, chemical resistance, and high fracture toughness.
- flame retardancy heat resistance
- low thermal expansion coefficient low water absorption
- low dielectric constant low dielectric constant
- the functional polymer material may contain a halogen atom or a phosphorus atom.
- the halogen atom may generate a halogen-based gas that may cause environmental pollution during combustion, and lowers the insulation of the final product.
- phosphorus atoms often reduce required physical properties other than flame retardancy (heat resistance, moisture resistance, low water absorption, etc.). Therefore, it is also required to improve the flame retardancy without containing a halogen atom and a phosphorus atom in the functional polymer material.
- a prepreg may be used when manufacturing a laminated board for printed wiring boards.
- a prepreg is prepared by first dissolving a monomer before curing, which is a precursor of a functional polymer material, in a solvent such as methyl ethyl ketone to prepare a varnish, then impregnating the varnish into glass fibers and drying it. Sometimes. Therefore, it is also required to improve solvent solubility for the monomer.
- an isocyanuric acid skeleton-containing cyanate ester compound see Patent Document 2
- a triazine skeleton-containing cyanate ester compound Patent Document 3
- a bifunctional cyanatophenyl type cyanate ester compound in which a hydrogen atom of a methylene group bonding cyanatophenyl groups is substituted with a biphenyl group see Patent Document 4
- See also) or bisphenol A type cyanic acid Combining the ester compound and the imide skeleton-containing cyanate ester compound see Patent Document 2
- a triazine skeleton-containing cyanate ester compound Patent Document 3
- Proposed composition containing novolac-type cyanate, novolac-type phenolic resin and inorganic filler as a composition using cyanate ester compound with heat resistance and long-term high-temperature heat resistance (long-term heat resistance) (Patent Document 8).
- Patent Document 1 in a bifunctional cyanatophenyl type cyanate ester compound, when hydrogen of a methylene group that bonds cyanatophenyl groups is replaced with an alkyl group, flame retardancy (at high temperature) ) Will be reduced.
- Patent Document 1 has no description regarding flame retardancy and long-term heat resistance.
- Patent Documents 2 to 7 has any description regarding the coefficient of thermal expansion, long-term heat resistance and / or solvent solubility.
- the long-term heat resistance test in Patent Document 8 is about 50 hours (250 ° C.), which is not sufficient as the test time. Further, Patent Document 8 has no description regarding the coefficient of thermal expansion and solvent solubility.
- the present invention is a novel cyanate ester compound having excellent solvent solubility, a low thermal expansion coefficient, a cyanate ester compound from which a cured product having excellent flame retardancy and heat resistance can be obtained, and
- An object of the present invention is to provide a curable resin composition containing the compound.
- the present inventors have found that a cyanate ester compound obtained by cyanating a predetermined resin has excellent solvent solubility, and a curable resin composition using such a cyanate ester compound has a coefficient of thermal expansion.
- the present inventors have found that a cured product having low flame resistance and heat resistance can be realized, and the present invention has been achieved. That is, the present invention is as follows.
- k represents an integer of 1 or more
- l represents an integer of 0 or more.
- K and l may be a mixture of different compounds.
- the arrangement of each repeating unit is arbitrary.
- m and n each represent an integer of 0 or more, and at least one of them is 1 or more.
- a mixture of compounds in which m and n are different may be used.
- the arrangement of each repeating unit is arbitrary.
- m represents an integer of 0 or more.
- a mixture of compounds in which m is different may be used.
- the cyanate ester compound according to [1] obtained by cyanating at least one resin selected from the group consisting of naphthol-dihydroxynaphthalene aralkyl resins and dihydroxynaphthalene aralkyl resins.
- the naphthol-dihydroxynaphthalene aralkyl resin is an acid catalyst comprising at least one selected from the group consisting of para-xylene glycol and 1,4-bis (methoxymethyl) benzene and 1-naphthol and 2,7-dihydroxynaphthalene.
- the dihydroxynaphthalene aralkyl resin comprises at least one selected from the group consisting of paraxylene glycol and 1,4-bis (methoxymethyl) benzene and 2,7-dihydroxynaphthalene.
- a curable resin composition comprising the cyanate ester compound according to any one of [1] to [5].
- a prepreg for a structural material comprising: a base material; and the curable resin composition according to [6] or [7], which is impregnated or applied to the base material.
- a sealing material comprising the curable resin composition according to [6] or [7].
- a fiber-reinforced composite material comprising the curable resin composition according to [6] or [7].
- a novel cyanate ester compound having excellent solvent solubility, a low thermal expansion coefficient, a cyanate ester compound capable of obtaining a cured product having excellent flame retardancy and heat resistance, and A curable resin composition containing the compound can be provided.
- 1 is a GPC chart of naphthol-dihydroxynaphthalene aralkyl resin obtained in Examples. It is a GPC chart of the cyanate ester compound obtained in the Example. 2 is an FT-IR chart of naphthol-dihydroxynaphthalene aralkyl resin and cyanate ester compound obtained in Examples. 6 is a GPC chart of naphthol-dihydroxynaphthalene aralkyl resin obtained in another example. It is a GPC chart of the cyanate ester compound obtained in another Example. 4 is an FT-IR chart of naphthol-dihydroxynaphthalene aralkyl resin and cyanate ester compound obtained in another example.
- 6 is a GPC chart of a cyanate ester compound obtained in yet another Example 5.
- 6 is an FT-IR chart of dihydroxynaphthalene aralkyl resin and cyanate ester compound obtained in another example.
- the cyanate ester compound of this embodiment is one kind selected from the group consisting of naphthol-dihydroxynaphthalene aralkyl resins and dihydroxynaphthalene aralkyl resins (hereinafter collectively referred to as “naphthol-dihydroxynaphthalene aralkyl resins etc.”). It is a cyanate ester compound obtained by cyanating the above resin. Moreover, the curable resin composition of this embodiment is a curable resin composition containing the said cyanate ester compound.
- a cured product obtained by curing the curable resin composition, a sealing material including the curable resin composition, a fiber-reinforced composite material, and an adhesive are also provided. .
- the naphthol-dihydroxynaphthalene aralkyl resin or the like used as a raw material for the cyanate ester compound of the present embodiment is not particularly limited. be able to.
- the naphthol-dihydroxynaphthalene aralkyl resin includes at least one selected from the group consisting of paraxylene glycol and 1,4-bis (methoxymethyl) benzene, 1-naphthol, 2,7-dihydroxynaphthalene, Can be obtained by reacting in the presence of an acidic catalyst.
- the dihydroxynaphthalene aralkyl resin is obtained by reacting at least one selected from the group consisting of para-xylene glycol and 1,4-bis (methoxymethyl) benzene with 2,7-dihydroxynaphthalene in the presence of an acidic catalyst. be able to.
- acidic catalysts used in the production of naphthol-dihydroxynaphthalene aralkyl resins and the like include mineral acids such as hydrochloric acid, sulfuric acid and phosphoric acid, organic acids such as oxalic acid, toluenesulfonic acid and acetic acid, heteropolyacids such as tungstic acid, Examples include activated acid clay, inorganic acid, stannic chloride, zinc chloride, and ferric chloride, and other commonly used acidic catalysts such as organic and inorganic acid salts showing acidity. These are used singly or in combination of two or more.
- the amount of the acidic catalyst used is 1 mol of the total hydroxy group of 1-naphthol and 2,7-dihydroxynaphthalene, or 1 mol of the hydroxy group of 2,7-dihydroxynaphthalene when 1-naphthol is not used.
- the amount is preferably 0.005 to 2.0 mol, more preferably 0.01 to 1.1 mol. It is preferable that the amount of catalyst used is 0.005 mol or more because the reaction proceeds more rapidly, the reaction at a lower temperature is possible, and the reaction proceeds more reliably to the end. Moreover, it is preferable for the amount of catalyst used to be 2.0 mol or less, since the labor in post-treatment such as neutralization and purification can be further reduced.
- a solvent inert to the above reaction can also be used.
- the solvent include alcohol solvents such as methanol, ethanol, propanol, and isopropanol, aromatic hydrocarbon solvents such as toluene and xylene, and methyl isobutyl ketone (MIBK), anone, cyclopentanone, and methyl ethyl ketone.
- MIBK methyl isobutyl ketone
- anone, cyclopentanone, and methyl ethyl ketone examples include ketone solvents. These can be used individually by 1 type or in combination of 2 or more types.
- the amount used is preferably 100 parts by mass in total of 1-naphthol and 2,7-dihydroxynaphthalene, or 100 parts by mass of 2,7-dihydroxynaphthalene when 1-naphthol is not used. Is from 5 to 500 parts by weight, more preferably from 10 to 400 parts by weight.
- the reaction temperature in the above reaction is preferably 0 to 300 ° C, more preferably 40 to 230 ° C.
- the reaction time in the above reaction is preferably 1 to 200 hours, more preferably 2 to 150 hours.
- the total amount of 1-naphthol and 2,7-dihydroxynaphthalene used or the total amount of 2,7-dihydroxynaphthalene when 1-naphthol is not used is paraxylene glycol and 1,4
- the amount is preferably 1.2 to 20 mol, more preferably 1.5 to 10 mol, relative to 1 mol of the total amount of bis (methoxymethyl) benzene.
- the softening point of the naphthol-dihydroxynaphthalene aralkyl resin or dihydroxynaphthalene aralkyl resin to be produced can be lowered more appropriately, and the resin can be removed from the reactor after the reaction. It becomes easier.
- the amount used is 20 mol or less, it is possible to further suppress a decrease in one or more structures selected from the group consisting of structures represented by the following formulas (3), (4) and (5), and heat resistance It becomes possible to prevent the fall of property.
- the reaction for obtaining a naphthol-dihydroxynaphthalene aralkyl resin includes at least one selected from the group consisting of paraxylene glycol and 1,4-bis (methoxymethyl) benzene, 1-naphthol, 2 , 7-dihydroxynaphthalene and, if necessary, a mixture of a solvent, an acidic catalyst is added as necessary, and the mixture is heated.
- a mixture of 1-naphthol, 2,7-dihydroxynaphthalene, an acidic catalyst, and, if necessary, a solvent is heated from the group consisting of paraxylene glycol and 1,4-bis (methoxymethyl) benzene.
- the above reaction can also be carried out by gradually adding one or more selected ones.
- the reaction for obtaining the dihydroxynaphthalene aralkyl resin includes at least one selected from the group consisting of paraxylene glycol and 1,4-bis (methoxymethyl) benzene, and 2,7-dihydroxynaphthalene. If necessary, an acidic catalyst is added to a mixture with a solvent as necessary and heated. In addition, at least one selected from the group consisting of paraxylene glycol and 1,4-bis (methoxymethyl) benzene is obtained by heating a mixture of 2,7-dihydroxynaphthalene, an acidic catalyst, and, if necessary, a solvent.
- the above reaction can also be carried out by gradually adding.
- These reactions may be performed with stirring, may be performed in air or in an atmosphere of an inert gas (for example, nitrogen, helium, argon, etc.), or may be performed under normal pressure or pressure.
- the progress of the reaction can be confirmed (or traced) by high performance liquid chromatography (HPLC), thin layer chromatography (TLC), or the like.
- the reaction mixture after completion of the reaction contains one or more selected from the group consisting of unreacted paraxylene glycol and 1,4-bis (methoxymethyl) benzene, unreacted 1-naphthol, unreacted 2,7- Dihydroxynaphthalene, acidic catalyst, side reaction product and the like are included. Therefore, the target compound can be separated and purified by a conventional method such as neutralization, washing with water, filtration, concentration, extraction, crystallization, recrystallization, column chromatography, or a combination of these. Is preferred.
- the naphthol-dihydroxynaphthalene aralkyl resin and the like according to this embodiment, which can be obtained by the above production method, are not particularly limited, but have a structure represented by the following formulas (3), (4), and (6) You may have 1 or more types of structures chosen from the group which consists of.
- k represents an integer of 1 or more
- l represents an integer of 0 or more. It may be a mixture of compounds in which k and l are different.
- sequence of each repeating unit is arbitrary.
- n and n represent an integer of 0 or more, and at least one of them is 1 or more. It may be a mixture of compounds in which m and n are different. Moreover, the arrangement
- sequence of each repeating unit is arbitrary.
- n represents an integer of 0 to 20 in the formula. It may be a mixture of compounds having different m.
- the cyanate ester compound of the present embodiment can be obtained by cyanating a hydroxy group of a naphthol-dihydroxynaphthalene aralkyl resin or the like (hereinafter, this step is also referred to as “cyanation step”).
- the method of cyanation is not particularly limited, and a known method can be adopted. More specifically, a method of reacting naphthol-dihydroxynaphthalene aralkyl resin or the like with cyanogen halide in a solvent in the presence of a basic compound; in a solvent in the presence of a base, the cyanogen halide is always in excess of the base.
- Naphthol-dihydroxynaphtha with continuous plug flow method A method of reacting a trialkylamine and a cyanogen halide such as a nonaralkyl resin (see Japanese Patent No. 3905559); a naphthol-dihydroxynaphthalene aralkyl resin and a cyanogen halide are reacted in a non-aqueous solution in the presence of tert-amine.
- a method of treating a tert-ammonium halide produced as a by-product with a cation and anion exchange pair see Japanese Patent No.
- the reaction substrate naphthol-dihydroxynaphthalene aralkyl resin or the like is converted into a cyanogen halide solution and After dissolving in advance in any of the basic compound solutions, the cyanogen halide solution and the basic compound solution may be contacted.
- a method for bringing the cyanogen halide solution into contact with the basic compound solution for example, (A) a basic compound solution is poured into the stirred cyanogen halide solution. (B) a method in which a cyan halide solution is poured into a stirred basic compound solution, and (C) a cyan halide solution and a basic compound solution are alternately alternated Or a method of supplying them simultaneously.
- the side reaction is further suppressed, and a higher purity cyanate ester compound can be obtained in a high yield. Therefore, the method (A) is preferable. .
- the contact method between the cyanogen halide solution and the basic compound solution may be either a semi-batch format or a continuous flow format.
- the reaction can be completed without leaving the hydroxy group of the naphthol-dihydroxynaphthalene aralkyl resin or the like, and a higher purity cyanate ester compound can be obtained in a high yield. Since it can be obtained, it is preferable to divide and drop the basic compound.
- the number of divisions is not particularly limited, but is preferably 1 to 5 times.
- the type of basic compound may be the same or different for each division.
- Examples of the cyanogen halide used in this embodiment include cyanogen chloride and cyanogen bromide.
- the cyanogen halide may be a cyanide halide obtained by a known production method such as a method of reacting hydrogen cyanide or metal cyanide with halogen, or may be a commercially available product.
- a reaction liquid containing cyanogen halide obtained by reacting hydrogen cyanide or metal cyanide with halogen can be used as it is.
- the amount of cyanogen cyanide used in the cyanation step of the present embodiment relative to naphthol-dihydroxynaphthalene aralkyl resin or the like is preferably 0.5 to 5 mol relative to 1 mol of hydroxy group such as naphthol-dihydroxynaphthalene aralkyl resin.
- the amount is preferably 1.0 to 3.5 mol.
- Examples of the solvent that can be contained in the cyanogen halide solution include ketone solvents such as acetone, methyl ethyl ketone, methyl isobutyl ketone, cyclohexanone, and cyclopentanone, aliphatic hydrocarbon solvents such as n-hexane, cyclohexane, and isooctane, and benzene.
- ketone solvents such as acetone, methyl ethyl ketone, methyl isobutyl ketone, cyclohexanone, and cyclopentanone
- aliphatic hydrocarbon solvents such as n-hexane, cyclohexane, and isooctane
- benzene ketone solvents such as acetone, methyl ethyl ketone, methyl isobutyl ketone, cyclohexanone, and cyclopentanone
- Aromatic hydrocarbon solvents such as toluene and xylene, ether solvents such as diethyl ether, dimethyl cellosolve, diglyme, tetrahydrofuran, methyltetrahydrofuran, dioxane, tetraethylene glycol dimethyl ether, dichloromethane, chloroform, carbon tetrachloride, dichloroethane, trichloroethane , Halogenated hydrocarbon solvents such as chlorobenzene and bromobenzene, methanol, ethanol, isopropanol, methyl sorbol, propylene Alcohol solvents such as glycol monomethyl ether, aprotic polar solvents such as N, N-dimethylformamide, N-methylpyrrolidone, 1,3-dimethyl-2-imidazolidone and dimethyl sulfoxide, and nitrile solvents such as acetonitrile and benzonitrile Nitro solvents such as nitrome
- both organic and inorganic bases can be used, and one of them can be used alone or in combination of two or more.
- the organic base is not particularly limited, but from the viewpoint of obtaining the target compound with higher yield, trimethylamine, triethylamine, tri-n-butylamine, triamylamine, diisopropylethylamine, diethyl-n-butylamine, methyldi-n-butylamine , Methylethyl-n-butylamine, dodecyldimethylamine, tribenzylamine, triethanolamine, N, N-dimethylaniline, N, N-diethylaniline, diphenylmethylamine, pyridine, diethylcyclohexylamine, tricyclohexylamine, 1, Tertiary such as 4-diazabicyclo [2.2.2] octane, 1,8-diazabicyclo [5.4.0] -7-undecene, and 1,5-diazabicyclo [4.3.0] -5-nonene Amines are preferred.
- trimethylamine, triethylamine, tri-n-butylamine, and diisopropylethylamine are more preferable, and triethylamine is particularly preferable from the viewpoint of obtaining the target compound with higher yield.
- the amount of the organic base used is preferably 0.1 to 8 mol, more preferably 1.0 to 3.5 mol, per 1 mol of hydroxy groups such as naphthol-dihydroxynaphthalene aralkyl resin. Thereby, the yield of the cyanate ester compound can be further increased without leaving unreacted naphthol-dihydroxynaphthalene aralkyl resin and the like.
- the inorganic base is not particularly limited, but an alkali metal hydroxide is preferable from the viewpoint of obtaining the target compound in a higher yield.
- an alkali metal hydroxide is preferable from the viewpoint of obtaining the target compound in a higher yield.
- a hydroxide of an alkali metal For example, sodium hydroxide, potassium hydroxide, and lithium hydroxide generally used industrially are mentioned. Among these, sodium hydroxide is particularly preferable because it can be obtained at a low cost.
- the amount of the inorganic base to be used is preferably 1.0 to 5.0 mol, more preferably 1.0 to 3.5 mol, per 1 mol of hydroxy groups such as naphthol-dihydroxynaphthalene aralkyl resin. Thereby, the yield of the cyanate ester compound can be further increased without leaving unreacted naphthol-dihydroxynaphthalene aralkyl resin and the like.
- the basic compound in the reaction of this embodiment, can be used as a solution dissolved in a solvent as described above.
- a solvent for example, an organic solvent or water can be used.
- the amount of the solvent that can be contained in the basic compound solution is preferably 0.1 with respect to 1 part by mass of the naphthol-dihydroxynaphthalene aralkyl resin or the like when the naphthol-dihydroxynaphthalene aralkyl resin or the like is dissolved in the basic compound solution. To 100 parts by mass, more preferably 0.5 to 50 parts by mass. In the case where the naphthol-dihydroxynaphthalene aralkyl resin or the like is not dissolved in the basic compound solution, the amount of the solvent used is preferably 0.1 to 100 parts by mass, more preferably 0. 25 to 50 parts by mass.
- organic solvent for dissolving the basic compound is preferably used when the basic compound is an organic base.
- organic solvents include ketone solvents such as acetone, methyl ethyl ketone, and methyl isobutyl ketone, aromatic hydrocarbon solvents such as benzene, toluene, and xylene, diethyl ether, dimethyl cellosolve, diglyme, tetrahydrofuran, and methyltetrahydrofuran.
- Ether solvents such as dioxane, tetraethylene glycol dimethyl ether, halogenated hydrocarbon solvents such as dichloromethane, chloroform, carbon tetrachloride, dichloroethane, trichloroethane, chlorobenzene, bromobenzene, methanol, ethanol, isopropanol, methylsolvesolve, propylene glycol Alcohol solvents such as monomethyl ether, N, N-dimethylformamide, N-methylpyrrolidone, 1,3-dimethyl-2 Aprotic polar solvents such as imidazolidone and dimethyl sulfoxide, nitrile solvents such as acetonitrile and benzonitrile, nitro solvents such as nitromethane and nitrobenzene, ester solvents such as ethyl acetate and ethyl benzoate, and aliphatics such as cyclohexane A hydrocarbon solvent is mentioned.
- Water in which the basic compound is dissolved is preferably used when the basic compound is an inorganic base.
- the water is not particularly limited, and may be tap water, distilled water, or deionized water. From the viewpoint of obtaining the target cyanate compound more efficiently, distilled water and deionized water with less impurities are preferred.
- the solvent contained in the basic compound solution is water
- a catalytic amount of an organic base is preferred.
- tertiary amines with few side reactions are preferred.
- the tertiary amine may be any of alkylamine, arylamine, and cycloalkylamine.
- trimethylamine, triethylamine, tri-n-butylamine, and diisopropylethylamine are more preferable, and triethylamine is particularly preferable from the viewpoint of solubility in water and the ability to obtain the target compound with higher yield. These are used singly or in combination of two or more.
- the total amount of the solvent used in the cyanation step of the present embodiment is 2.5 to 100 parts by mass with respect to 1 part by mass of naphthol-dihydroxynaphthalene aralkyl resin and the like, and naphthol-dihydroxynaphthalene aralkyl resin and the like are more uniform. From the viewpoint of more efficiently producing a cyanate ester compound.
- the pH of the reaction solution is not particularly limited, but it is preferable to carry out the reaction while maintaining a state where the pH is less than 7.
- a method of adding an acid to the reaction solution is preferable.
- an acid is added to the cyanogen halide solution immediately before the cyanation step, and an acid is added to the reaction system while appropriately measuring the pH of the reaction solution with a pH meter during the reaction.
- the method is such that pH is maintained at a level of less than 7.
- the acid used at that time include inorganic acids such as hydrochloric acid, nitric acid, sulfuric acid, and phosphoric acid, and organic acids such as acetic acid, lactic acid, and propionic acid.
- the reaction temperature in the cyanation step of the present embodiment is a viewpoint that further suppresses the formation of by-products such as imide carbonate, a polymer of a cyanate ester compound, and a dialkylcyanoamide, a viewpoint that suppresses the condensation of the reaction solution, and From the viewpoint of further suppressing the volatilization of cyanogen chloride when cyanogen chloride is used as the cyanogen halide, it is preferably ⁇ 20 to + 50 ° C., more preferably ⁇ 15 to 15 ° C., and further preferably ⁇ 10 to 10 ° C.
- the reaction pressure in the cyanation step of this embodiment may be normal pressure, or may be increased, that is, a pressure exceeding normal pressure. Moreover, you may distribute
- the reaction time is not particularly limited, but the pouring time when the contact method is the method (A) and (B) and the contact time when the method is the method (C) are 1 minute to 20 hours. Preferably, 3 minutes to 10 hours are more preferable. Further, it is preferable to stir while maintaining the reaction temperature for 10 minutes to 10 hours thereafter.
- the target cyanate ester compound can be obtained more economically and more industrially by setting the reaction conditions in the above range.
- the progress of the reaction in the cyanate formation step can be analyzed by liquid chromatography or IR spectrum method. Volatile components such as by-product dicyan and dialkylcyanoamide can be analyzed by gas chromatography.
- the desired cyanate ester compound can be isolated by carrying out the usual post-treatment operations that can be performed after synthesizing the conventional cyanate ester compound and, if desired, separation and purification operations. . Specifically, an organic solvent phase containing a cyanate ester compound is separated from the reaction solution, washed with water, concentrated, precipitated, or crystallized. Alternatively, the organic solvent phase thus collected may be washed with water and then replaced with a solvent. In order to remove excess amines in place of water at the time of washing, a method using an acidic aqueous solution such as dilute hydrochloric acid can also be employed.
- the reaction solution can be dried by a general method using sodium sulfate, magnesium sulfate or the like.
- the organic solvent is distilled off by heating to a temperature of 90 ° C. or lower under reduced pressure.
- a solvent having a low solubility of the cyanate ester compound can be used.
- a method of dropping a hydrocarbon solvent such as an ether solvent, hexane, or an alcohol solvent into the reaction solution, or a method of pouring the reaction solution into the solvent can be employed.
- a method of washing the concentrate of the reaction solution and precipitated crystals with an ether solvent, a hydrocarbon solvent such as hexane, or an alcohol solvent should be adopted.
- the crystals obtained by concentrating the reaction solution may be re-dissolved and then recrystallized. Crystallization may be performed by simply concentrating or cooling the reaction solution.
- the cyanate ester compound obtained by the above production method is not particularly limited, but one or more selected from the group consisting of structures represented by the following formulas (1), (2) and (5) It has a structure.
- k represents an integer of 1 or more
- l represents an integer of 0 or more. It may be a mixture of compounds in which k and l are different.
- sequence of each repeating unit is arbitrary.
- the upper limit of k is not particularly limited, but is preferably 20 or less from the viewpoint of more effectively and reliably achieving the effects of the present invention. From the same viewpoint, the upper limit of l is preferably 20 or less.
- m and n represent an integer of 0 or more, and at least one of them is 1 or more. It may be a mixture of compounds in which m and n are different. Moreover, the arrangement
- the upper limit of m is not particularly limited, but is preferably 20 or less from the viewpoint of more effectively and reliably achieving the effects of the present invention. From the same viewpoint, the upper limit of n is preferably 20 or less.
- m represents an integer of 0 or more. It may be a mixture of compounds having different m.
- the upper limit of m is not particularly limited, but is preferably 20 or less from the viewpoint of more effectively and reliably achieving the effects of the present invention.
- the weight average molecular weight Mw of the cyanate ester compound of the present embodiment is not particularly limited, but is preferably 100 to 5000, more preferably 200 to 3500, and further preferably 200 to 3000.
- the weight average molecular weight Mw is measured by the method described in Examples.
- the obtained cyanate ester compound can be identified by a known method such as NMR.
- the purity of the cyanate ester compound can be analyzed by liquid chromatography or IR spectroscopy.
- Byproducts such as dialkylcyanoamide in the cyanate ester compound and volatile components such as residual solvent can be quantitatively analyzed by gas chromatography.
- Halogen compounds remaining in the cyanate ester compound can be identified by a liquid chromatograph mass spectrometer, and can be quantitatively analyzed by potentiometric titration using a silver nitrate solution or ion chromatography after decomposition by a combustion method. .
- the polymerization reactivity of the cyanate ester compound can be evaluated by gelation time by a hot plate method or a torque measurement method.
- the curable resin composition of the present embodiment contains the cyanate ester compound.
- This curable resin composition includes a cyanate ester compound other than the cyanate ester compound (hereinafter referred to as “another cyanate ester compound”), a maleimide compound, and a phenol resin as long as desired characteristics are not impaired.
- An epoxy resin, an oxetane resin, a benzoxazine compound, and one or more selected from the group consisting of compounds having a polymerizable unsaturated group may be contained.
- cyanate ester compounds include compounds having an aromatic moiety substituted with at least one cyanate group in the molecule, and those other than those obtained by cyanating a naphthol-dihydroxynaphthalene aralkyl resin or the like. If there is no particular limitation. For example, what is represented by following formula (7) is mentioned.
- Ar 1 represents a single bond of a benzene ring, a naphthalene ring or two benzene rings, and when there are a plurality of them, they may be the same or different.
- Each Ra is independently a hydrogen atom, an alkyl group having 1 to 6 carbon atoms, an aryl group having 6 to 12 carbon atoms, an alkoxy group having 1 to 4 carbon atoms, or an alkyl group having 1 to 6 carbon atoms and 6 to 12 carbon atoms.
- the aromatic ring in Ra may have a substituent, and the substituent in Ar 1 and Ra can be selected at any position.
- p represents the number of cyanato groups bonded to Ar 1 , and each independently represents an integer of 1 to 3.
- q represents the number of Ra bonded to Ar 1, and 4-p when Ar 1 is a benzene ring, 6-p when Ar 1 is a naphthalene ring, and 8-p when two benzene rings are single-bonded.
- . t represents an average number of repetitions and is an integer of 0 to 50, and the other cyanate compound may be a mixture of compounds having different t.
- Xs each independently represents a single bond, a divalent organic group having 1 to 50 carbon atoms (a hydrogen atom may be substituted with a hetero atom), a divalent group having 1 to 10 nitrogen atoms.
- An organic group eg, —N—R—N— (where R represents an organic group)
- a carbonyl group —CO—
- a carboxy group —C ( ⁇ O) O—
- a carbonyl dioxide group —OC ( ⁇ O) O—
- a sulfonyl group —SO 2 —
- divalent sulfur atom or a divalent oxygen atom.
- the alkyl group in Ra in the formula (7) may have any of a linear or branched chain structure and a cyclic structure (for example, a cycloalkyl group).
- the hydrogen atom in the alkyl group in formula (7) and the aryl group in Ra may be substituted with a halogen atom such as a fluorine atom or a chlorine atom, an alkoxy group such as a methoxy group or a phenoxy group, or a cyano group.
- a halogen atom such as a fluorine atom or a chlorine atom
- an alkoxy group such as a methoxy group or a phenoxy group
- cyano group for example, a cyano group.
- alkyl group examples include methyl group, ethyl group, propyl group, isopropyl group, n-butyl group, isobutyl group, tert-butyl group, n-pentyl group, 1-ethylpropyl group, 2,2-dimethylpropyl group.
- aryl group examples include phenyl group, xylyl group, mesityl group, naphthyl group, phenoxyphenyl group, ethylphenyl group, o-, m- or p-fluorophenyl group, dichlorophenyl group, dicyanophenyl group, trifluorophenyl.
- alkoxy group examples include a methoxy group, an ethoxy group, a propoxy group, an isopropoxy group, an n-butoxy group, an isobutoxy group, and a tert-butoxy group.
- divalent organic group having 1 to 50 carbon atoms in X in the formula (7) include alkylene groups such as methylene group, ethylene group, trimethylene group and propylene group, cyclopentylene group, cyclohexylene group and trimethyl.
- divalent organic groups having an aromatic ring such as a cycloalkylene group such as cyclohexylene group, biphenylylmethylene group, dimethylmethylene-phenylene-dimethylmethylene group, fluorenediyl group, and phthalidodiyl group.
- the hydrogen atom in the divalent organic group may be substituted with a halogen atom such as a fluorine atom or a chlorine atom, an alkoxy group such as a methoxy group or a phenoxy group, or a cyano group.
- Examples of the divalent organic group having 1 to 10 nitrogen atoms in X in the formula (7) include a group represented by —N—R—N—, an imino group, and a polyimide group.
- Ar 2 represents a benzenetetrayl group, a naphthalenetetrayl group or a biphenyltetrayl group, and when u is 2 or more, they may be the same as or different from each other.
- Rb, Rc, Rf and Rg each independently have at least one hydrogen atom, alkyl group having 1 to 6 carbon atoms, aryl group having 6 to 12 carbon atoms, trifluoromethyl group, or phenolic hydroxy group. An aryl group is shown.
- Rd and Re each independently represent a hydrogen atom, an alkyl group having 1 to 6 carbon atoms, an aryl group having 6 to 12 carbon atoms, an alkoxy group having 1 to 4 carbon atoms, or a hydroxy group.
- u represents an integer of 0 to 5.
- Ar 3 represents a benzenetetrayl group, a naphthalenetetrayl group or a biphenyltetrayl group, and when v is 2 or more, they may be the same as or different from each other.
- Ri and Rj are each independently a hydrogen atom, an alkyl group having 1 to 6 carbon atoms, an aryl group having 6 to 12 carbon atoms, a benzyl group, an alkoxy group having 1 to 4 carbon atoms, a hydroxy group, a trifluoromethyl group, Or an aryl group substituted with at least one cyanato group.
- v represents an integer of 0 to 5, but may be a mixture of compounds having different v.
- X in Formula (7) the bivalent group represented by a following formula is mentioned.
- m represents an integer of 4 to 7.
- Each R independently represents a hydrogen atom or an alkyl group having 1 to 6 carbon atoms.
- Ar 2 in formula (8) and Ar 3 in formula (9) include two carbon atoms represented by formula (8) or two oxygen atoms represented by formula (9).
- a benzenetetrayl group bonded to the 4-position or 1,3-position, the two carbon atoms or the two oxygen atoms are 4,4′-position, 2,4′-position, 2,2′-position, 2,
- the biphenyltetrayl group bonded to the 3′-position, the 3,3′-position, or the 3,4′-position, and the two carbon atoms or the two oxygen atoms are in the 2,6-position, 1,5-position
- Examples thereof include naphthalenetetrayl groups bonded to the 1,6-position, 1,8-position, 1,3-position, 1,4-position, or 2,7-position.
- Rb, Rc, Rd, Re, Rf and Rg in the formula (8) and Ri and Rj in the formula (9) have the same meanings as those in the above formula (7).
- cyanato-substituted aromatic compound represented by the above formula (7) include cyanatobenzene, 1-cyanato-2-, 1-cyanato-3-, or 1-cyanato-4-methylbenzene, 1- Cyanato-2-, 1-cyanato-3-, or 1-cyanato-4-methoxybenzene, 1-cyanato-2,3-, 1-cyanato-2,4-, 1-cyanato-2,5-, 1 -Cyanato-2,6-, 1-cyanato-3,4- or 1-cyanato-3,5-dimethylbenzene, cyanatoethylbenzene, cyanatobutylbenzene, cyanatooctylbenzene, cyanatononylbenzene, 2- ( 4-cyanatophenyl) -2-phenylpropane (cyanate of 4- ⁇ -cumylphenol), 1-cyanato-4-cyclohexylbenzene, 1-cyanato-4-vinylbenzene 1-
- phenol novolak resin and cresol novolak resin phenol, alkyl-substituted phenol or halogen-substituted phenol, formalin, A product obtained by reacting a formaldehyde compound such as paraformaldehyde in an acidic solution), a trisphenol novolak resin (a product obtained by reacting hydroxybenzaldehyde and phenol in the presence of an acidic catalyst), a fluorene novolac resin (9 , 9-bis (hydroxyaryl) fluorenes in the presence of an acidic catalyst), furan ring-containing phenol novolak resin (furfural and phenol reacted in the presence of a basic catalyst), phenol Ara Kill resin, cresol aralkyl resin, naphthol aralkyl resin, a binaphthol aralkyl resin and a biphenyl aralky
- a phenol-modified xylene formaldehyde resin (a product obtained by reacting a xylene formaldehyde resin and a phenol compound in the presence of an acidic catalyst by a known method), a modified naphthalene formaldehyde resin (a known method).
- a naphthalene formaldehyde resin with a hydroxy-substituted aromatic compound in the presence of an acidic catalyst
- a phenol-modified dicyclopentadiene resin a phenol resin having a polynaphthylene ether structure
- examples thereof include those obtained by cyanating a phenol resin such as a polyhydric hydroxynaphthalene compound having two or more groups in one molecule by dehydration condensation in the presence of a basic catalyst. These are not particularly limited. These other cyanate ester compounds can be used alone or in combination of two or more.
- maleimide compound generally known compounds can be used as long as they have one or more maleimide groups in one molecule.
- maleimide compounds include o-phenylene bismaleimide, m-phenylene bismaleimide, p-phenylene bismaleimide, o-phenylene biscitraconimide, m-phenylene biscitraconimide, p-phenylene biscitraconimide, 4,4 ′.
- the phenol resin a phenol resin having two or more hydroxy groups in one molecule is preferable, and generally known resins can be used.
- the phenolic resin include bisphenol A type phenolic resin, bisphenol E type phenolic resin, bisphenol F type phenolic resin, bisphenol S type phenolic resin, phenol novolac resin, bisphenol A novolac type phenolic resin, glycidyl ester type phenolic resin, aralkyl novolac.
- epoxy resin generally known compounds can be used as long as they are compounds having two or more epoxy groups in one molecule.
- epoxy resins include bisphenol A type epoxy resins, bisphenol E type epoxy resins, bisphenol F type epoxy resins, bisphenol S type epoxy resins, bisphenol A novolac type epoxy resins, biphenyl type epoxy resins, phenol novolac type epoxy resins, and cresol novolacs.
- Type epoxy resin xylene novolac type epoxy resin, naphthalene type epoxy resin, anthracene type epoxy resin, trifunctional phenol type epoxy resin, tetrafunctional phenol type epoxy resin, triglycidyl isocyanurate, glycidyl ester type epoxy resin, alicyclic epoxy resin , Dicyclopentadiene novolac epoxy resin, biphenyl novolac epoxy resin, phenol aralkyl novolac epoxy resin, naphtha Aralkyl novolac type epoxy resin, aralkyl novolac type epoxy resin, biphenyl aralkyl type epoxy resin, naphthol aralkyl type epoxy resin, dicyclopentadiene type epoxy resin, polyol type epoxy resin, alicyclic epoxy resin, or halides thereof. Can be mentioned. These epoxy resins can be used alone or in combination of two or more.
- oxetane resin generally known oxetane resins can be used.
- the oxetane resin include alkyl oxetanes such as oxetane, 2-methyloxetane, 2,2-dimethyloxetane, 3-methyloxetane, 3,3-dimethyloxetane, 3-methyl-3-methoxymethyloxetane, 3,3
- oxetane resin include alkyl oxetanes such as oxetane, 2-methyloxetane, 2,2-dimethyloxetane, 3-methyloxetane, 3,3-dimethyloxetane, 3-methyl-3-methoxymethyloxetane, 3,3
- OXT-101 trade name of Toagosei Co., Ltd.
- OXT- 121 trade name of Toagosei Co., Ltd.
- the benzoxazine compound is preferably a compound having two or more dihydrobenzoxazine rings in one molecule, and generally known compounds can be used.
- the benzoxazine compound include BA-BXZ (trade name, manufactured by Konishi Chemical Co., Ltd.), which is a bisphenol A type benzoxazine, and BF-BXZ (trade name, manufactured by Konishi Chemical Co., Ltd.), a bisphenol S type benzoxazine.
- BS-BXZ (trade name, manufactured by Konishi Chemical) and phenolphthalein-type benzoxazine.
- the compound having a polymerizable unsaturated group generally known compounds can be used.
- the compound having a polymerizable unsaturated group include vinyl compounds such as ethylene, propylene, styrene, divinylbenzene, and divinylbiphenyl, methyl (meth) acrylate, 2-hydroxyethyl (meth) acrylate, 2-hydroxypropyl ( Monovalents such as (meth) acrylate, polypropylene glycol di (meth) acrylate, trimethylolpropane di (meth) acrylate, trimethylolpropane tri (meth) acrylate, pentaerythritol tetra (meth) acrylate, dipentaerythritol hexa (meth) acrylate
- epoxy (meth) acrylates such as (meth) acrylates of polyhydric alcohols, bisphenol A type epoxy (meth) acrylates, bisphenol F type epoxy (meth) acrylates,
- the “(meth) acrylate” is a concept including acrylate and methacrylate corresponding to the acrylate.
- the curable resin composition in the present embodiment includes, in addition to the above-described compounds and resins, a cyanate ester compound, a maleimide compound, a phenol resin, an epoxy resin, an oxetane resin, and a compound having a polymerizable unsaturated group.
- a compound that acts as a polymerization catalyst can be blended.
- the polymerization catalyst include metal salts such as zinc octylate, zinc naphthenate, cobalt naphthenate, copper naphthenate, and acetylacetone iron, phenol compounds such as octylphenol and nonylphenol, and alcohols such as 1-butanol and 2-ethylhexanol.
- peroxides such as epoxy-imidazole adduct compounds, benzoyl peroxide, p-chlorobenzoyl peroxide, di-t-butyl peroxide, diisopropyl peroxycarbonate, di-2-ethylhexyl peroxycarbonate, and azo
- An azo compound such as bisisobutyronitrile may be used as a polymerization catalyst.
- Commercially available polymerization catalysts may be used. Examples of commercially available products include Amicure PN-23 (trade name, manufactured by Ajinomoto Fine Techno Co., Ltd.), Novacure HX-3721 (trade name, manufactured by Asahi Kasei Co., Ltd.), and Fujicure FX-1000. (Trade name manufactured by Fuji Kasei Kogyo Co., Ltd.). These polymerization catalysts can be used alone or in combination of two or more.
- the curable resin composition in the present embodiment can include an inorganic filler.
- the inorganic filler include talc, fired clay, unfired clay, mica, E glass, A glass, NE glass, C glass, L glass, D glass, S glass, M glass G20, and short glass fiber (E glass).
- Glass fine powders such as T glass, D glass, S glass, and Q glass.
- Oxides such as magnesium oxide, zirconium oxide and molybdenum oxide, carbonates such as calcium carbonate, magnesium carbonate and hydrotalcite, hydroxides such as aluminum hydroxide, magnesium hydroxide and calcium hydroxide, barium sulfate and calcium sulfate , Sulfates or sulfites such as calcium sulfite, zinc borate, barium metaborate, boron Borates such as aluminum, calcium borate and sodium borate, nitrides such as aluminum nitride, boron nitride, silicon nitride and carbon nitride, carbides such as silicon carbide, titanates such as strontium titanate and barium titanate, Examples include zinc molybdate,
- the surface of the inorganic filler may be further treated with a surface treatment agent in advance.
- a surface treatment agent one or more compounds selected from the group consisting of functional group-containing silanes, cyclic oligosiloxanes, organohalosilanes, and alkylsilazanes can be suitably used.
- treating the surface of an inorganic filler (particularly spherical silica) with organohalosilanes and alkylsilazanes is suitable for hydrophobizing the surface of the inorganic filler, and the curable resin composition. It is preferable in terms of excellent dispersibility of the inorganic filler in the product.
- the curable resin composition of the present embodiment includes a thermoplastic resin, a curing catalyst, a curing accelerator, a color pigment, an antifoaming agent, a surface conditioner, a flame retardant, an ultraviolet absorber, and an antioxidant as necessary.
- Known additives such as photopolymerization initiators, optical brighteners, photosensitizers, dyes, pigments, thickeners, lubricants, flow regulators, dispersants, leveling agents, brighteners, and polymerization inhibitors. You may contain. Moreover, you may contain the solvent as needed. These optional additives can be used alone or in combination of two or more.
- solvent generally known solvents can be used.
- the solvent include ketone solvents such as acetone, methyl ethyl ketone, methyl isobutyl ketone and cyclohexanone, cellosolv solvents such as propylene glycol monomethyl ether and propylene glycol monomethyl ether acetate, methyl lactate, methyl acetate, ethyl acetate, butyl acetate and acetic acid.
- ketone solvents such as acetone, methyl ethyl ketone, methyl isobutyl ketone and cyclohexanone
- cellosolv solvents such as propylene glycol monomethyl ether and propylene glycol monomethyl ether acetate, methyl lactate, methyl acetate, ethyl acetate, butyl acetate and acetic acid.
- Ester solvents such as isoamyl, ethyl lactate, methyl methoxypropionate and methyl hydroxyisobutyrate, alcohol solvents such as methanol, ethanol, isopropanol and 1-ethoxy-2-propanol, and aromatic carbonization such as toluene, xylene and anisole Hydrogen is mentioned. These solvents can be used alone or in combination of two or more.
- the curable resin composition in the present embodiment includes the above-described cyanate ester compound and, if necessary, other cyanate ester compounds, maleimide compounds, phenol resins, epoxy resins, oxetane resins, benzoxazine compounds, and / or , Compounds having polymerizable unsaturated groups and various additives, together with solvents, using known mixers such as high-speed mixers, nauter mixers, ribbon blenders, kneaders, intensive mixers, universal mixers, dissolvers, static mixers, etc. Can be obtained by mixing.
- the mixing method of the cyanate ester compound, various additives and the solvent during mixing is not particularly limited.
- the curable resin composition according to the present embodiment can be cured by being cured by heat or light.
- the cured product can be obtained, for example, by melting the curable resin composition or dissolving it in a solvent, then pouring it into a mold and curing it under normal conditions.
- the curing temperature is preferably in the range of 120 ° C. to 300 ° C. from the viewpoint of further curing and further suppressing deterioration of the cured product.
- the sealing material of this embodiment contains the said curable resin composition, and can be manufactured using the curable resin composition.
- a method for producing the sealing material generally known methods can be appropriately employed, and are not particularly limited.
- the curable resin composition is mixed with various additives and / or solvents that are known to be used when producing a sealing material by using a known mixer.
- a stopping material can be manufactured.
- the well-known thing can be employ
- the structural material prepreg of the present embodiment includes a base material and the curable resin composition impregnated or coated on the base material.
- the prepreg for a structural material can be produced by impregnating or applying the curable resin composition on an inorganic and / or organic fiber substrate and further drying as necessary.
- polyamides such as inorganic fiber base materials, such as glass fiber base materials, such as a glass woven fabric and a glass nonwoven fabric, a polyamide resin fiber, an aromatic polyamide resin fiber, and a fully aromatic polyamide resin fiber
- Synthetic fibers composed of polyester resin fibers, polyimide resin fibers, fluororesin fibers, etc. as main components such as polyester resin fibers, polyester resin fibers, aromatic polyester resin fibers and wholly aromatic polyester resin fibers
- organic fiber base materials such as base materials, kraft paper, cotton linter paper, paper base materials mainly composed of linter and kraft pulp mixed paper.
- the glass which comprises the said glass fiber base material is not specifically limited, For example, E glass, C glass, A glass, S glass, D glass, NE glass, T glass, and H glass are mentioned.
- the method for producing the structural material prepreg is not particularly limited, and generally known methods can be adopted as appropriate.
- a resin varnish is prepared using the curable resin composition described above, a method of immersing the substrate in the resin varnish, a method of applying the resin varnish to the substrate with various coaters, a method of spraying by spraying, etc.
- a prepreg can be produced.
- the method of immersing the base material in the resin varnish is preferable. Thereby, the impregnation property of the resin composition with respect to a base material can be improved.
- a normal impregnation coating equipment can be used.
- a method of manufacturing a prepreg by impregnating an inorganic and / or organic fiber base material with a resin composition varnish, drying, and B-staging can be employed.
- the fiber-reinforced composite material of the present embodiment includes the curable resin composition of the present embodiment, and can be manufactured using the curable resin composition and reinforcing fibers.
- the reinforcing fiber contained in the fiber-reinforced composite material include carbon fibers, glass fibers, aramid fibers, boron fibers, PBO fibers, high-strength polyethylene fibers, alumina fibers, and silicon carbide fibers.
- the form and arrangement of the reinforcing fibers are not particularly limited, and can be appropriately selected from woven fabrics, nonwoven fabrics, mats, knits, braids, unidirectional strands, rovings, choppeds, and the like.
- a preform (a laminate of woven fabrics made of reinforcing fibers, or a structure obtained by stitching them together with stitch yarns, or a fiber structure such as a three-dimensional woven fabric or a braided fabric) is adopted.
- a liquid composite molding method a resin film infusion method, a filament winding method, a hand layup method, and a pultrusion method.
- the resin transfer molding method which is one of the liquid composite molding methods, is to set materials other than preforms such as metal plates, foam cores and honeycomb cores in the mold in advance. Can be applied to various applications. Therefore, the resin transfer molding method is preferably used when mass-producing a composite material having a relatively complicated shape in a short time.
- the adhesive of the present embodiment includes the curable resin composition of the present embodiment, and can be manufactured using the curable resin composition.
- a method for producing an adhesive generally known ones can be appropriately employed, and are not particularly limited.
- the adhesive is obtained by mixing the curable resin composition and various additives and / or solvents that are known to be used in producing an adhesive using a known mixer. Can be manufactured.
- generally the well-known thing can be employ
- the cyanate ester compound of this embodiment has excellent solvent solubility and excellent handleability.
- the curable resin composition of the present embodiment can realize a cured product having a low coefficient of thermal expansion and excellent flame retardancy and heat resistance by including such a cyanate ester compound. It is.
- the curable resin composition according to the present embodiment has excellent low thermal expansion, flame retardancy, and heat resistance, it is extremely useful as a high-functional polymer material.
- This curable resin composition is a material with excellent thermal, electrical and mechanical properties. In addition to electrical insulation materials, sealing materials, adhesives, laminate materials, resists, build-up laminate materials, civil engineering and architecture It is preferably used for fixing materials, structural members, reinforcing agents, molding materials, etc.
- Example 1 Synthesis of cyanate ester compound 1 of naphthol-dihydroxynaphthalene aralkyl resin (hereinafter abbreviated as “MF27S-3-CN”) MF27S-3- represented by the following formula (1) and / or the following formula (2) CN was synthesized as described below.
- k represents an integer of 1 to 20
- l represents an integer of 0 to 20. It may be a mixture of compounds in which k and l are different.
- the arrangement of each repeating unit is arbitrary.
- m and n each represent an integer of 0 to 20, and at least one of them is 1 or more. It may be a mixture of compounds in which m and n are different.
- the arrangement of each repeating unit is arbitrary.
- MF27S-3-OH naphthol-dihydroxynaphthalene aralkyl resin 1
- k represents an integer of 1 to 20
- l represents an integer of 0 to 20. It may be a mixture of compounds in which k and l are different.
- the arrangement of each repeating unit is arbitrary.
- m and n each represent an integer of 0 to 20, and at least one of them is 1 or more. It may be a mixture of compounds in which m and n are different.
- the arrangement of each repeating unit is arbitrary.
- the MF group equivalent of MF27S-3-OH obtained was 174 g / eq. Met.
- solution 2 A solution dissolved in 14.6 g of dichloromethane (hereinafter referred to as “solution 2”) was poured into the reactor over 25 minutes. After completion of the pouring of the solution 2, the inside of the reactor was stirred at the same temperature for 30 minutes to complete the reaction.
- the reaction liquid in the reactor was allowed to stand to separate the organic phase and the aqueous phase.
- the obtained organic phase was washed with 2 L of 0.1N hydrochloric acid and then washed 6 times with 2000 g of water.
- the electrical conductivity of the waste water in the sixth washing with water was 20 ⁇ S / cm, and it was confirmed that the ionic compounds that could be removed were sufficiently removed by washing with water.
- the organic phase after washing with water was concentrated under reduced pressure and finally concentrated to dryness at 90 ° C. for 1 hour to obtain 570 g of the intended cyanate ester compound MF27S-3-CN (black purple viscous product).
- the obtained MF27S-3-CN had a weight average molecular weight Mw of 980.
- the GPC chart is shown in FIG.
- the IR spectrum of MF27S-3-CN showed absorption of a cyanate ester group at 2264 cm ⁇ 1 and no absorption of a hydroxy group.
- the IR chart of the naphthol-dihydroxynaphthalene aralkyl resin 1 (MF27S-3-OH) is shown in FIG.
- MF27S-3-CN could be dissolved in methyl ethyl ethyl ketone at 50% by mass or more at 25 ° C.
- Example 2 Preparation of curable resin composition and production of cured product> 100 parts by mass of MF27S-3-CN, which is the cyanate ester compound obtained in Example 1, was put into an eggplant type flask, heated and melted at 150 ° C., and deaerated with a vacuum pump. Pour MF27S-3-CN after degassing into the mold described in JIS-K7238-2-2009, store in an oven, heat at 180 ° C for 3 hours, and then heat at 250 ° C for 3 hours. To obtain a cured product having a side of 100 mm and a thickness of 2 mm.
- MF27S-7-CN naphthol-dihydroxynaphthalene aralkyl resin cyanate ester compound 2
- CN naphthol-dihydroxynaphthalene aralkyl resin cyanate ester compound 2
- k represents an integer of 1 to 20
- l represents an integer of 0 to 20. It may be a mixture of compounds in which k and l are different.
- the arrangement of each repeating unit is arbitrary.
- m and n each represent an integer of 0 to 20, and at least one of them is 1 or more. It may be a mixture of compounds in which m and n are different.
- the arrangement of each repeating unit is arbitrary.
- MF27S-7-OH naphthol-dihydroxynaphthalene aralkyl resin 2
- k represents an integer of 1 to 20
- l represents an integer of 0 to 20. It may be a mixture of compounds in which k and l are different.
- the arrangement of each repeating unit is arbitrary.
- m and n each represent an integer of 0 to 20, and at least one of them is 1 or more. It may be a mixture of compounds in which m and n are different.
- the arrangement of each repeating unit is arbitrary.
- the mixed solvent was removed by distillation under reduced pressure to obtain 185 g of MF27S-7-OH.
- the obtained MF27S-7-OH had an OH group equivalent of 175 g / eq. Met.
- solution 2 A solution dissolved in 4.0 g of dichloromethane (hereinafter referred to as “solution 2”) was poured into the reactor over 2 minutes. After completion of the pouring of the solution 2, the inside of the reactor was stirred at the same temperature for 30 minutes to complete the reaction.
- the reaction liquid in the reactor was allowed to stand to separate the organic phase and the aqueous phase.
- the obtained organic phase was washed with 100 mL of 0.1N hydrochloric acid, and then 4 times with 100 g of water.
- the electric conductivity of the waste water in the fourth washing with water was 20 ⁇ S / cm, and it was confirmed that the ionic compounds that could be removed were sufficiently removed by washing with water.
- MF27S-7-CN black purple viscous product
- the obtained MF27S-7-CN had a weight average molecular weight Mw of 1350.
- the GPC chart is shown in FIG. Further, IR spectrum of MF27S-7-CN represents the absorption of a cyanate ester group 2239Cm -1 and 2265 cm -1, and the absorption of the hydroxy groups did not.
- the IR chart of the naphthol-dihydroxynaphthalene aralkyl resin 2 (MF27S-7-OH) is shown in FIG. MF27S-7-CN could be dissolved in methyl ethyl ethyl ketone at 50 ° C. or more at 25 ° C. or more.
- Example 4 Instead of using 100 parts by mass of MF27S-3-CN, 50 parts by mass of MF27S-7-CN obtained in Example 3 and 2,2-bis (4-cyanatophenyl) propane (Mitsubishi Gas) A cured product was obtained in the same manner as in Example 2 except that 50 parts by mass of Chemical, trade name “skylex” (hereinafter abbreviated as “TA”) was used.
- TA trade name “skylex”
- MF27S-10-CN cyanate ester compound of dihydroxynaphthalene aralkyl resin
- MF27S-10-CN cyanate ester compound of dihydroxynaphthalene aralkyl resin
- MF27S-10-OH dihydroxynaphthalene aralkyl resin
- the mixed solvent was removed by distillation under reduced pressure to obtain 158 g of MF27S-10-OH.
- the obtained MF27S-10-OH had an OH group equivalent of 131 g / eq. Met.
- solution 2 a solution dissolved in 12.4 g of dichloromethane (hereinafter referred to as “solution 2”) was poured into the reactor over 5 minutes. After completion of the pouring of the solution 2, the inside of the reactor was stirred at the same temperature for 30 minutes to complete the reaction.
- the reaction liquid in the reactor was allowed to stand to separate the organic phase and the aqueous phase.
- the obtained organic phase was washed with 100 mL of 0.1N hydrochloric acid, and then 4 times with 100 g of water.
- the electric conductivity of the waste water in the fourth washing with water was 20 ⁇ S / cm, and it was confirmed that the ionic compounds that could be removed were sufficiently removed by washing with water.
- the organic phase after washing with water was concentrated under reduced pressure and finally concentrated to dryness at 90 ° C. for 1 hour to obtain 28 g of the intended cyanate ester compound MF27S-10-CN (black purple viscous product).
- the obtained MF27S-10-CN had a weight average molecular weight Mw of 1190.
- the GPC chart is shown in FIG.
- the IR spectrum of MF27S-10-CN showed absorption of cyanate ester groups at 2237 cm ⁇ 1 and 2266 cm ⁇ 1 .
- the IR chart is shown in FIG. 9 together with the IR chart of dihydroxynaphthalene aralkyl resin (MF27S-10-OH).
- MF27S-10-CN was able to dissolve 50% by mass or more at 25 ° C. in methyl ethyl ethyl ketone.
- Example 6 Similar to Example 2 except that 50 parts by mass of MF27S-10-CN obtained in Example 5 and 50 parts by mass of TA were used instead of using 100 parts by mass of MF27S-3-CN. A cured product was obtained.
- Example 1 A cured product was obtained in the same manner as in Example 2 except that 100 parts by mass of TA was used instead of 100 parts by mass of MF27S-3-CN. In addition, TA was able to melt
- Glass transition temperature (Tg) Based on JIS-K7244-3 (JIS C6481), using a dynamic viscoelasticity measuring device (manufactured by TA Instruments Japan Co., Ltd., model number “AR2000”), start temperature 30 ° C., end temperature 400 ° C.
- the dynamic viscoelasticity of the cured product was measured under the conditions of a heating rate of 3 ° C./min and a measurement frequency of 1 Hz, and the maximum value of the loss elastic modulus (E ′′) obtained at that time was defined as the glass transition temperature.
- the glass transition temperature is an index of heat resistance.
- thermomechanical analyzer (trade name “TMA / SS7100” manufactured by SII NanoTechnology Co., Ltd.), a cured specimen (dimension: 5 mm) ⁇ 5mm ⁇ 2mm), thermal mechanical analysis in expansion / compression mode was performed under conditions of start temperature 30 ° C, end temperature 330 ° C, heating rate 10 ° C / min, weight 0.05N (49mN). The average thermal expansion amount per 1 ° C. at 60 to 120 ° C. was measured to obtain the thermal expansion coefficient.
- Mass reduction rate (%) (DE) / D ⁇ 100
- D represents the mass at the starting temperature
- E represents the mass at 500 ° C. (the same unit as D).
- flame retardant is defined as a large amount of residue during pyrolysis, that is, a low mass reduction rate.
- the cured product prepared by the above method was stored in a hot air oven in an air atmosphere at 250 ° C. for 360 hours, and the glass transition temperature of the cured product after storage was measured. When the glass transition temperature decrease before and after storage was 20% or less, it was accepted, and when it exceeded 20%, it was rejected.
- the cyanate ester compound such as naphthol-dihydroxynaphthalene aralkyl resin of the present embodiment has excellent solvent solubility and excellent handleability. Further, the cured product of the curable resin composition using the cyanate ester compound of the present embodiment has a low coefficient of thermal expansion, excellent flame retardancy and It was confirmed to have heat resistance.
- the curable resin composition according to the present invention is extremely useful as a high-functional polymer material because it has excellent low thermal expansion, flame retardancy and heat resistance.
- This curable resin composition is a material with excellent thermal, electrical and mechanical properties.
- electrical insulation materials, sealing materials, adhesives, laminate materials, resists, build-up laminate materials, civil engineering and architecture It is preferably used for fixing materials, structural members, reinforcing agents, molding materials, etc. in fields such as electricity / electronics, automobiles, railways, ships, aircraft, sports equipment, arts and crafts, etc., and industrial applicability to these applications There is.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Condensed Matter Physics & Semiconductors (AREA)
- Physics & Mathematics (AREA)
- General Physics & Mathematics (AREA)
- Computer Hardware Design (AREA)
- Microelectronics & Electronic Packaging (AREA)
- Power Engineering (AREA)
- Manufacturing & Machinery (AREA)
- General Chemical & Material Sciences (AREA)
- Inorganic Chemistry (AREA)
- Reinforced Plastic Materials (AREA)
- Phenolic Resins Or Amino Resins (AREA)
- Macromolecular Compounds Obtained By Forming Nitrogen-Containing Linkages In General (AREA)
- Compositions Of Macromolecular Compounds (AREA)
- Polyoxymethylene Polymers And Polymers With Carbon-To-Carbon Bonds (AREA)
- Adhesives Or Adhesive Processes (AREA)
Abstract
Description
本実施形態のシアン酸エステル化合物は、ナフトール-ジヒドロキシナフタレンアラルキル樹脂及びジヒドロキシナフタレンアラルキル樹脂(以下、これらを総称して「ナフトール-ジヒドロキシナフタレンアラルキル樹脂等」ともいう。)からなる群より選ばれる1種以上の樹脂をシアネート化して得られるシアン酸エステル化合物である。また、本実施形態の硬化性樹脂組成物は、上記シアン酸エステル化合物を含む硬化性樹脂組成物である。
本実施形態のシアン酸エステル化合物の原料となるナフトール-ジヒドロキシナフタレンアラルキル樹脂等は、特に限定されないが、例えば特公平7-45557号公報又は特開2014-9336号公報等に記載の方法に従い製造することができる。具体的には、ナフトール-ジヒドロキシナフタレンアラルキル樹脂は、パラキシレングリコール及び1,4-ビス(メトキシメチル)ベンゼンからなる群より選ばれる1種以上と、1-ナフトールと、2,7-ジヒドロキシナフタレンとを酸性触媒存在下に反応させて得ることができる。また、ジヒドロキシナフタレンアラルキル樹脂は、パラキシレングリコール及び1,4-ビス(メトキシメチル)ベンゼンからなる群より選ばれる1種以上と、2,7-ジヒドロキシナフタレンとを酸性触媒存在下に反応させて得ることができる。
本実施形態のシアン酸エステル化合物は、ナフトール-ジヒドロキシナフタレンアラルキル樹脂等が有するヒドロキシ基をシアネート化することで得られる(以下、この工程を「シアネート化工程」ともいう。)。シアネート化の方法は、特に制限されるものではなく、公知の方法を採用することができる。より具体的には、ナフトール-ジヒドロキシナフタレンアラルキル樹脂等とハロゲン化シアンを、溶媒中、塩基性化合物存在下で反応させる方法;溶媒中、塩基の存在下で、ハロゲン化シアンが常に塩基より過剰に存在するようにして、ナフトール-ジヒドロキシナフタレンアラルキル樹脂等とハロゲン化シアンを反応させる方法(米国特許3553244号参照);、塩基として3級アミンを用い、これをハロゲン化シアンよりも過剰に用いながら、溶媒の存在下、ナフトール-ジヒドロキシナフタレンアラルキル樹脂等に3級アミンを添加した後、ハロゲン化シアンを滴下する、又は、ハロゲン化シアンと3級アミンとを併注滴下する方法(特許3319061号公報参照);連続プラグフロー方式で、ナフトール-ジヒドロキシナフタレンアラルキル樹脂等、トリアルキルアミン及びハロゲン化シアンを反応させる方法(特許3905559号公報参照);ナフトール-ジヒドロキシナフタレンアラルキル樹脂等とハロゲン化シアンとを、tert-アミンの存在下、非水溶液中で反応させる際に副生するtert-アンモニウムハライドを、カチオン及びアニオン交換対で処理する方法(特許4055210号公報参照);ナフトール-ジヒドロキシナフタレンアラルキル樹脂等に対して、水と分液可能な溶媒の存在下で、3級アミンとハロゲン化シアンとを同時に添加して反応させた後、水洗分液し、得られた溶液から2級若しくは3級アルコール類又は炭化水素の貧溶媒を用いて沈殿精製する方法(特許2991054号公報参照);あるいは、ナフトール-ジヒドロキシナフタレンアラルキル樹脂等、ハロゲン化シアン、及び3級アミンを、水と有機溶媒との二相系溶媒中、酸性条件下で反応させる方法(特許5026727号公報);等により、本実施形態のシアン酸エステル化合物を得ることができる。
その際に用いる酸としては、例えば、塩酸、硝酸、硫酸、燐酸等の無機酸、並びに、酢酸、乳酸、プロピオン酸等の有機酸が挙げられる。
本実施形態の硬化性樹脂組成物は、上記シアン酸エステル化合物を含むものである。この硬化性樹脂組成物は、所期の特性が損なわれない範囲において、上記シアン酸エステル化合物以外のシアン酸エステル化合物(以下、「他のシアン酸エステル化合物」という。)、マレイミド化合物、フェノール樹脂、エポキシ樹脂、オキセタン樹脂、ベンゾオキサジン化合物、及び重合可能な不飽和基を有する化合物からなる群より選ばれる1種以上を含有していてもよい。
式(8)のRb、Rc、Rd、Re、Rf及びRg、並びに式(9)のRi、及びRjにおけるアルキル基及びアリール基は上記式(7)におけるものと同義である。
本実施形態の封止用材料は、上記硬化性樹脂組成物を含むものであり、その硬化性樹脂組成物を用いて製造することができる。封止用材料の製造方法は、一般に公知のものを適宜採用でき、特に限定されない。例えば、上記硬化性樹脂組成物と、封止用材料を製造する際に用いられることが知られている各種の添加剤及び/又は溶媒等とを、公知のミキサーを用いて混合することで封止用材料を製造することができる。なお、混合の際の、硬化性樹脂組成物、各種添加剤及び溶媒の添加方法は、一般に公知のものを適宜採用でき、特に限定されない。
JIS-K0070に準拠して、ピリジン-塩化アセチル法により、水酸基含有芳香族化合物のOH基当量(g/eq.)を求めた。
シアン酸エステル化合物1gを100gのテトラヒドロフラン(溶媒)に溶解させた溶液10μLを、高速液体クロマトグラフィー(株式会社日立ハイテクノロジーズ社製高速液体クロマトグラフ、製品名「LachromElite」)に注入し分析を実施した。カラムとして東ソー株式会社製の製品名「TSKgel GMHHR-M」(長さ30cm×内径7.8mm)2本を用い、移動相としてテトラヒドロフランを用いた。カラム内での試料の流速は1mL/min.に設定し、検出器としてRI(示差屈折)検出器を用いた。重量平均分子量Mwは、GPC法によりポリスチレンを標準物質として求めた。
ナフトール-ジヒドロキシナフタレンアラルキル樹脂のシアン酸エステル化合物1(以下、「MF27S-3-CN」と略記する。)の合成
下記式(1)及び/又は下記式(2)で表されるMF27S-3-CNを後述のようにして合成した。
まず、下記式(3)及び/又は下記式(4)で表されるMF27S-3-OHを合成した。
次に、上記方法で得られたMF27S-3-OH500g(OH基当量174g/eq.、OH基換算で2.87mol、重量平均分子量Mw760(GPCチャートを図1に示す。))、トリエチルアミン436.2g(4.31mol、MF27S-3-OHのOH基1モルに対して1.5モル)をジクロロメタン3000gに溶解させ、これを溶液1とした。
<硬化性樹脂組成物の調製及び硬化物の作製>
実施例1で得られたシアン酸エステル化合物であるMF27S-3-CN100質量部をナス型フラスコに投入し、150℃で加熱溶融させ、真空ポンプで脱気した。脱気後のMF27S-3-CNを、JIS-K7238-2-2009に記載の型に流し込み、オーブンに収容し、180℃にて3時間加熱し、その後、250℃にて3時間加熱することにより硬化させ、1辺100mm、厚さ2mmの硬化物を得た。
ナフトール-ジヒドロキシナフタレンアラルキル樹脂のシアン酸エステル化合物2(以下、「MF27S-7-CN」と略記する。)の合成
下記式(1)及び/又は下記式(2)で表されるMF27S-7-CNを後述のようにして合成した。
まず、下記式(3)及び/又は下記式(4)で表されるMF27S-7-OHを合成した。
次に、上記方法で得られたMF27S-7-OH15g(OH基当量175g/eq.、OH基換算で0.086mol、重量平均分子量Mw960(GPCチャートを図4に示す。))、及びトリエチルアミン13.0g(0.128mol、MF27S-7-OHのOH基1モルに対して1.5モル)をジクロロメタン90g及びテトラヒドロフラン15gに溶解させ、これを溶液1とした。
MF27S-3-CNを100質量部用いることに代えて、実施例3で得られたMF27S-7-CNを50質量部、及び、2,2-ビス(4-シアナートフェニル)プロパン(三菱ガス化学製、商品名「skylex」、以下、「TA」と略記)を50質量部用いたこと以外は、実施例2と同様にして硬化物を得た。
ジヒドロキシナフタレンアラルキル樹脂のシアン酸エステル化合物(以下、「MF27S-10-CN」と略記する。)の合成
下記式(5)で表されるMF27S-10-CNを後述のようにして合成した。
まず、下記式(6)で表されるMF27S-10-OHを合成した。
次に、上記方法で得られたMF27S-10-OH20g(OH基当量131g/eq.、OH基換算で0.153mol、重量平均分子量Mw650(GPCチャートを図7に示す。))、及びトリエチルアミン23.2g(0.229mol、MF27S-10-OHのOH基1モルに対して1.5モル)をジクロロメタン120g及びテトラヒドロフラン30gに溶解させ、これを溶液1とした。
MF27S-3-CNを100質量部用いることに代えて、実施例5で得られたMF27S-10-CNを50質量部、及び、TAを50質量部用いたこと以外は、実施例2と同様にして硬化物を得た。
MF27S-3-CNを100質量部用いることに代えて、TAを100質量部用いたこと以外は、実施例2と同様にして硬化物を得た。なお、TAは、メチルエチルケトンに対し、25℃で50質量%以上溶解することが可能であった。
[ガラス転移温度(Tg)]
JIS-K7244-3(JIS C6481)に準拠し、動的粘弾性測定装置(ティー・エイ・インスツルメント・ジャパン株式会社製、型番「AR2000」)を用い、開始温度30℃、終了温度400℃、昇温速度3℃/分、測定周波数1Hzの条件にて、硬化物の動的粘弾性を測定し、その際に得られた損失弾性率(E”)の最大値をガラス転移温度とした。ガラス転移温度は耐熱性の指標である。
JIS-K-7197-2012(JIS C6481)に準拠し、熱機械的分析装置(エスアイアイ・ナノテクノロジー株式会社製、商品名「TMA/SS7100」)を用い、硬化物の試験片(寸法:5mm×5mm×2mm)に対し、開始温度30℃、終了温度330℃、昇温速度10℃/分、加重0.05N(49mN)の条件にて、膨張・圧縮モードでの熱機械分析を実施し、60~120℃における1℃当たりの平均熱膨張量を測定し、熱膨張係数を求めた。
JIS-K7120-1987に準拠し、示差熱熱質量同時測定装置(株式会社リガク製、商品名「サーモプラスエボTG8120」)を用い、硬化物の試験片(寸法:3mm×3mm×2mm)について、開始温度40℃、終了温度500℃、昇温速度10℃/分、窒素雰囲気下の条件にて、質量を測定し、500℃における質量減少率を下記式に基づき算出した。
質量減少率(%)=(D-E)/D×100
ここで、Dは開始温度での質量を示し、Eは500℃での質量(Dと同じ単位)をそれぞれ示す。ここで、本明細書では、「難燃性」を、熱分解時の残渣量が多いこと、すなわち、質量減少率が低いことと定義する。
上記方法にて調製した硬化物を、熱風オーブン中にて、空気雰囲気下、250℃で360時間保管し、保管後の硬化物のガラス転移温度を測定した。保管前後でのガラス転移温度の減少幅が20%以下のものを合格とし、20%を超えたものを不合格とした。
Claims (12)
- ナフトール-ジヒドロキシナフタレンアラルキル樹脂及びジヒドロキシナフタレンアラルキル樹脂からなる群より選ばれる1種以上の樹脂をシアネート化して得られる、シアン酸エステル化合物。
- ナフトール-ジヒドロキシナフタレンアラルキル樹脂及びジヒドロキシナフタレンアラルキル樹脂からなる群より選ばれる1種以上の樹脂をシアネート化して得られる、請求項1に記載のシアン酸エステル化合物。
- 前記ナフトール-ジヒドロキシナフタレンアラルキル樹脂が、パラキシレングリコール及び1,4-ビス(メトキシメチル)ベンゼンからなる群より選ばれる1種以上と1-ナフトールと2,7-ジヒドロキシナフタレンとを酸性触媒の存在下に反応させたものであり、前記ジヒドロキシナフタレンアラルキル樹脂が、パラキシレングリコール及び1,4-ビス(メトキシメチル)ベンゼンからなる群より選ばれる1種以上と2,7-ジヒドロキシナフタレンとを酸性触媒の存在下に反応させたものである、請求項2又は3に記載のシアン酸エステル化合物。
- 重量平均分子量Mwが100~5000である、請求項1~4のいずれか一項に記載のシアン酸エステル化合物。
- 請求項1~5のいずれか一項に記載のシアン酸エステル化合物を含む、硬化性樹脂組成物。
- 前記シアン酸エステル化合物以外のシアン酸エステル化合物、マレイミド化合物、フェノール樹脂、エポキシ樹脂、オキセタン樹脂、ベンゾオキサジン化合物及び重合可能な不飽和基を有する化合物からなる群より選択される1種以上を更に含む、請求項6に記載の硬化性樹脂組成物。
- 請求項6又は7に記載の硬化性樹脂組成物を硬化させてなる、硬化物。
- 基材と、
該基材に含浸又は塗布された、請求項6又は7に記載の硬化性樹脂組成物と、
を備える構造材料用プリプレグ。 - 請求項6又は7に記載の硬化性樹脂組成物を含む、封止用材料。
- 請求項6又は7に記載の硬化性樹脂組成物を含む、繊維強化複合材料。
- 請求項6又は7に記載の硬化性樹脂組成物を含む、接着剤。
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020177027330A KR102545337B1 (ko) | 2015-03-31 | 2016-02-19 | 시안산에스테르 화합물, 그 화합물을 함유하는 경화성 수지 조성물 및 그 경화물 |
| CN201680020007.7A CN107406582B (zh) | 2015-03-31 | 2016-02-19 | 氰酸酯化合物、包含该化合物的固化性树脂组合物和其固化物 |
| EP16771950.9A EP3279232B1 (en) | 2015-03-31 | 2016-02-19 | Cyanic acid ester compound, curable resin composition containing said compound, and cured object obtained therefrom |
| US15/560,708 US10370325B2 (en) | 2015-03-31 | 2016-02-19 | Cyanate ester compound, curable resin composition containing the compound, and hardened product thereof |
| JP2017509371A JP6710434B2 (ja) | 2015-03-31 | 2016-02-19 | シアン酸エステル化合物、該化合物を含む硬化性樹脂組成物及びその硬化物 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2015-072689 | 2015-03-31 | ||
| JP2015072689 | 2015-03-31 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2016158066A1 true WO2016158066A1 (ja) | 2016-10-06 |
Family
ID=57006981
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2016/054850 WO2016158066A1 (ja) | 2015-03-31 | 2016-02-19 | シアン酸エステル化合物、該化合物を含む硬化性樹脂組成物及びその硬化物 |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US10370325B2 (ja) |
| EP (1) | EP3279232B1 (ja) |
| JP (1) | JP6710434B2 (ja) |
| KR (1) | KR102545337B1 (ja) |
| CN (1) | CN107406582B (ja) |
| TW (1) | TWI674254B (ja) |
| WO (1) | WO2016158066A1 (ja) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2019044133A1 (ja) * | 2017-08-28 | 2019-03-07 | 京セラ株式会社 | SiC及びGaN素子封止用成形材料組成物、電子部品装置 |
| JP7469747B1 (ja) | 2023-07-27 | 2024-04-17 | 三菱瓦斯化学株式会社 | シアン酸エステル化合物及びその製造方法、樹脂組成物、並びに硬化物 |
| JP7469748B1 (ja) | 2023-07-27 | 2024-04-17 | 三菱瓦斯化学株式会社 | シアン酸エステル化合物及びその製造方法、樹脂組成物、並びに硬化物 |
Families Citing this family (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US11250555B2 (en) * | 2018-11-07 | 2022-02-15 | Brigham Young University | Data acquisition for liquid composite molding |
| CN109825039B (zh) * | 2018-12-28 | 2021-07-30 | 广东生益科技股份有限公司 | 氰酸酯树脂组合物及其用途 |
| CN110330612A (zh) * | 2019-06-27 | 2019-10-15 | 扬州天启新材料股份有限公司 | 一种酚醛型氰酸酯树脂的生产工艺 |
| WO2022005783A1 (en) * | 2020-07-02 | 2022-01-06 | Fujifilm Electronic Materials U.S.A., Inc. | Dielectric film-forming composition |
Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS6031585A (ja) * | 1983-07-28 | 1985-02-18 | Dainippon Ink & Chem Inc | 摩擦材用樹脂組成物 |
| JPH0570549A (ja) * | 1991-09-10 | 1993-03-23 | Dai Ichi Kogyo Seiyaku Co Ltd | エポキシ樹脂組成物 |
| JPH06145309A (ja) * | 1992-11-13 | 1994-05-24 | Mitsui Toatsu Chem Inc | ヒドロキシナフタレン共重合体、そのエポキシ化物、それらの製造方法および用途 |
| JPH06234832A (ja) * | 1993-02-09 | 1994-08-23 | Nippon Steel Chem Co Ltd | 高耐熱性エポキシ樹脂硬化物 |
| JP2014185222A (ja) * | 2013-03-22 | 2014-10-02 | Mitsubishi Gas Chemical Co Inc | 樹脂組成物、プリプレグ、積層板及びプリント配線板 |
| WO2014196501A1 (ja) * | 2013-06-03 | 2014-12-11 | 三菱瓦斯化学株式会社 | プリント配線板材料用樹脂組成物、並びにそれを用いたプリプレグ、樹脂シート、金属箔張積層板、及びプリント配線板 |
| WO2014203866A1 (ja) * | 2013-06-18 | 2014-12-24 | 三菱瓦斯化学株式会社 | 樹脂組成物、プリプレグ、樹脂シート及び金属箔張り積層板 |
| JP2015053341A (ja) * | 2013-09-05 | 2015-03-19 | 信越化学工業株式会社 | 封止材積層複合体、封止後半導体素子搭載基板又は封止後半導体素子形成ウエハ、半導体装置、及び半導体装置の製造方法 |
Family Cites Families (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS514312B1 (ja) | 1969-11-28 | 1976-02-10 | ||
| JPS514312A (en) | 1974-07-02 | 1976-01-14 | Toray Industries | Yojuhorimachuno suibunoyobi teifutsutenfujunbutsujokyohoho |
| US4709008A (en) | 1986-06-30 | 1987-11-24 | Interez, Inc. | Blend of tris (cyanatophenyl) alkane and bis(cyanatophenyl) alkane |
| WO1999005196A1 (en) * | 1997-07-24 | 1999-02-04 | Loctite Corporation | Thermosetting resin compositions useful as underfill sealants |
| JP4843944B2 (ja) * | 2005-01-13 | 2011-12-21 | 三菱瓦斯化学株式会社 | 樹脂組成物並びにこれを用いたプリプレグ及び積層板 |
| JP4654770B2 (ja) | 2005-05-30 | 2011-03-23 | 三菱瓦斯化学株式会社 | シアン酸エステル化合物、難燃性樹脂組成物、並びにこれを用いたプリプレグおよび積層板 |
| CN101283022B (zh) | 2005-10-25 | 2012-06-20 | 三菱瓦斯化学株式会社 | 氰酸酯聚合物 |
| JP2010180147A (ja) | 2009-02-04 | 2010-08-19 | Mitsubishi Gas Chemical Co Inc | シアン酸エステル化合物、およびその硬化物 |
| JP5630133B2 (ja) | 2010-08-05 | 2014-11-26 | 三菱瓦斯化学株式会社 | シアン酸エステル化合物、およびその硬化物 |
| WO2012057144A1 (ja) | 2010-10-29 | 2012-05-03 | 三菱瓦斯化学株式会社 | シアン酸エステル化合物、シアン酸エステル化合物を含む硬化性樹脂組成物、およびその硬化物 |
| WO2013021869A1 (ja) | 2011-08-09 | 2013-02-14 | 三菱瓦斯化学株式会社 | 新規なシアン酸エステル化合物及びその製造方法、並びに該化合物を含む硬化性樹脂組成物及びその硬化物 |
| JP2013053218A (ja) | 2011-09-02 | 2013-03-21 | Kaneka Corp | 熱硬化性樹脂組成物及び半導体封止用樹脂組成物 |
-
2016
- 2016-02-19 CN CN201680020007.7A patent/CN107406582B/zh active Active
- 2016-02-19 US US15/560,708 patent/US10370325B2/en active Active
- 2016-02-19 EP EP16771950.9A patent/EP3279232B1/en active Active
- 2016-02-19 KR KR1020177027330A patent/KR102545337B1/ko active IP Right Grant
- 2016-02-19 JP JP2017509371A patent/JP6710434B2/ja active Active
- 2016-02-19 WO PCT/JP2016/054850 patent/WO2016158066A1/ja active Application Filing
- 2016-02-25 TW TW105105578A patent/TWI674254B/zh active
Patent Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS6031585A (ja) * | 1983-07-28 | 1985-02-18 | Dainippon Ink & Chem Inc | 摩擦材用樹脂組成物 |
| JPH0570549A (ja) * | 1991-09-10 | 1993-03-23 | Dai Ichi Kogyo Seiyaku Co Ltd | エポキシ樹脂組成物 |
| JPH06145309A (ja) * | 1992-11-13 | 1994-05-24 | Mitsui Toatsu Chem Inc | ヒドロキシナフタレン共重合体、そのエポキシ化物、それらの製造方法および用途 |
| JPH06234832A (ja) * | 1993-02-09 | 1994-08-23 | Nippon Steel Chem Co Ltd | 高耐熱性エポキシ樹脂硬化物 |
| JP2014185222A (ja) * | 2013-03-22 | 2014-10-02 | Mitsubishi Gas Chemical Co Inc | 樹脂組成物、プリプレグ、積層板及びプリント配線板 |
| WO2014196501A1 (ja) * | 2013-06-03 | 2014-12-11 | 三菱瓦斯化学株式会社 | プリント配線板材料用樹脂組成物、並びにそれを用いたプリプレグ、樹脂シート、金属箔張積層板、及びプリント配線板 |
| WO2014203866A1 (ja) * | 2013-06-18 | 2014-12-24 | 三菱瓦斯化学株式会社 | 樹脂組成物、プリプレグ、樹脂シート及び金属箔張り積層板 |
| JP2015053341A (ja) * | 2013-09-05 | 2015-03-19 | 信越化学工業株式会社 | 封止材積層複合体、封止後半導体素子搭載基板又は封止後半導体素子形成ウエハ、半導体装置、及び半導体装置の製造方法 |
Non-Patent Citations (3)
| Title |
|---|
| HONG-QIANG YAN ET AL.: "Interpenetrating polymer networks from the novel bismaleimide and cyanate containing naphthalene: Cure and thermal characteristics", EUROPEAN POLYMER JOURNAL, 2009, pages 2383 - 2390, XP026336912 * |
| See also references of EP3279232A4 * |
| TSUNG-HAN HO: "Synthesis of naphthalene containing aralkyl Novolac epoxy resins for electronic application", MACROMOLECULAR MATERIALS AND ENGINEERING, vol. 283, 2000, pages 57 - 61, XP055318296 * |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2019044133A1 (ja) * | 2017-08-28 | 2019-03-07 | 京セラ株式会社 | SiC及びGaN素子封止用成形材料組成物、電子部品装置 |
| CN110603294A (zh) * | 2017-08-28 | 2019-12-20 | 京瓷株式会社 | SiC和GaN元件密封用成型材料组合物、电子部件装置 |
| CN110603294B (zh) * | 2017-08-28 | 2022-07-22 | 京瓷株式会社 | SiC和GaN元件密封用成型材料组合物、电子部件装置 |
| JP7469747B1 (ja) | 2023-07-27 | 2024-04-17 | 三菱瓦斯化学株式会社 | シアン酸エステル化合物及びその製造方法、樹脂組成物、並びに硬化物 |
| JP7469748B1 (ja) | 2023-07-27 | 2024-04-17 | 三菱瓦斯化学株式会社 | シアン酸エステル化合物及びその製造方法、樹脂組成物、並びに硬化物 |
Also Published As
| Publication number | Publication date |
|---|---|
| US10370325B2 (en) | 2019-08-06 |
| CN107406582B (zh) | 2020-01-17 |
| TWI674254B (zh) | 2019-10-11 |
| US20180105488A1 (en) | 2018-04-19 |
| EP3279232A1 (en) | 2018-02-07 |
| JPWO2016158066A1 (ja) | 2018-01-25 |
| TW201638068A (zh) | 2016-11-01 |
| KR20170133356A (ko) | 2017-12-05 |
| KR102545337B1 (ko) | 2023-06-19 |
| EP3279232A4 (en) | 2018-12-19 |
| CN107406582A (zh) | 2017-11-28 |
| EP3279232B1 (en) | 2020-05-06 |
| JP6710434B2 (ja) | 2020-06-17 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6602016B2 (ja) | シアン酸エステル化合物、該化合物を含む硬化性樹脂組成物及びその硬化物 | |
| JP5578299B1 (ja) | ハロゲン化シアンの製造方法、シアン酸エステル化合物及びその製造方法、並びに樹脂組成物 | |
| WO2016158066A1 (ja) | シアン酸エステル化合物、該化合物を含む硬化性樹脂組成物及びその硬化物 | |
| JP6838604B2 (ja) | シアン酸エステル化合物、その製造方法、樹脂組成物、硬化物、プリプレグ、封止用材料、繊維強化複合材料、接着剤、金属箔張積層板、樹脂シート及びプリント配線板 | |
| JP6850432B2 (ja) | シアン酸エステル化合物、樹脂組成物、硬化物、構造材料用プリプレグ、封止用材料、繊維強化複合材料及び接着剤 | |
| JP6468440B2 (ja) | シアン酸エステル化合物、該化合物を含む硬化性樹脂組成物及びその硬化物 | |
| KR20160079006A (ko) | 시안산에스테르 화합물, 그 화합물을 포함하는 경화성 수지 조성물 및 그 경화물 | |
| JP2015209381A (ja) | シアン酸エステル化合物、該化合物を含む硬化性樹脂組成物及びその硬化物 | |
| JP6667126B2 (ja) | シアン酸エステル化合物、該化合物を含む硬化性樹脂組成物及びその硬化物 | |
| JP6071120B2 (ja) | シアン酸エステル化合物及びその製造方法、樹脂組成物、並びに、硬化物 | |
| JP6402448B2 (ja) | シアン酸エステル化合物、該化合物を含む硬化性樹脂組成物及びその硬化物 | |
| JP6288559B2 (ja) | シアン酸エステル化合物、該化合物を含む硬化性樹脂組成物及びその硬化物 | |
| JP6544713B2 (ja) | シアン酸エステル化合物、該化合物を含む硬化性樹脂組成物及びその硬化物 | |
| WO2019198550A1 (ja) | シアン酸エステル化合物、及びその製造方法、樹脂組成物、硬化物、単層樹脂シート、積層樹脂シート、プリプレグ、金属箔張積層板、プリント配線板、封止用材料、繊維強化複合材料、並びに接着剤 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 16771950 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2017509371 Country of ref document: JP Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 15560708 Country of ref document: US |
|
| ENP | Entry into the national phase |
Ref document number: 20177027330 Country of ref document: KR Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| REEP | Request for entry into the european phase |
Ref document number: 2016771950 Country of ref document: EP |