WO2016080367A1 - 安定化されたペプチド組成物 - Google Patents
安定化されたペプチド組成物 Download PDFInfo
- Publication number
- WO2016080367A1 WO2016080367A1 PCT/JP2015/082179 JP2015082179W WO2016080367A1 WO 2016080367 A1 WO2016080367 A1 WO 2016080367A1 JP 2015082179 W JP2015082179 W JP 2015082179W WO 2016080367 A1 WO2016080367 A1 WO 2016080367A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- peptide
- amino acid
- seq
- acid sequence
- composition according
- Prior art date
Links
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
- A61K38/04—Peptides having up to 20 amino acids in a fully defined sequence; Derivatives thereof
- A61K38/08—Peptides having 5 to 11 amino acids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/02—Inorganic compounds
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/08—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite containing oxygen, e.g. ethers, acetals, ketones, quinones, aldehydes, peroxides
- A61K47/10—Alcohols; Phenols; Salts thereof, e.g. glycerol; Polyethylene glycols [PEG]; Poloxamers; PEG/POE alkyl ethers
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/26—Carbohydrates, e.g. sugar alcohols, amino sugars, nucleic acids, mono-, di- or oligo-saccharides; Derivatives thereof, e.g. polysorbates, sorbitan fatty acid esters or glycyrrhizin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/30—Macromolecular organic or inorganic compounds, e.g. inorganic polyphosphates
- A61K47/36—Polysaccharides; Derivatives thereof, e.g. gums, starch, alginate, dextrin, hyaluronic acid, chitosan, inulin, agar or pectin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/10—Dispersions; Emulsions
- A61K9/107—Emulsions ; Emulsion preconcentrates; Micelles
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07H—SUGARS; DERIVATIVES THEREOF; NUCLEOSIDES; NUCLEOTIDES; NUCLEIC ACIDS
- C07H3/00—Compounds containing only hydrogen atoms and saccharide radicals having only carbon, hydrogen, and oxygen atoms
- C07H3/04—Disaccharides
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K7/00—Peptides having 5 to 20 amino acids in a fully defined sequence; Derivatives thereof
- C07K7/04—Linear peptides containing only normal peptide links
- C07K7/06—Linear peptides containing only normal peptide links having 5 to 11 amino acids
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02A—TECHNOLOGIES FOR ADAPTATION TO CLIMATE CHANGE
- Y02A50/00—TECHNOLOGIES FOR ADAPTATION TO CLIMATE CHANGE in human health protection, e.g. against extreme weather
- Y02A50/30—Against vector-borne diseases, e.g. mosquito-borne, fly-borne, tick-borne or waterborne diseases whose impact is exacerbated by climate change
Definitions
- the present invention relates to a composition containing a peptide. Specifically, the present invention relates to a composition in which the stability of a plurality of types of peptides is improved by containing a sugar and an inorganic salt, and more specifically, by containing sucrose and sodium chloride.
- Patent Documents 1 to 3 A plurality of peptides having the ability to induce cytotoxic T cells used as vaccines for the treatment or prevention of cancer are known (Patent Documents 1 to 3). Some of these peptides produce an analog when subjected to a long-term stability test, and the residual ratio decreases.
- a preparation containing sodium carbonate and / or arginine (Patent Document 4) is disclosed.
- examples of injections in which proteins or peptides are stabilized by containing saccharides such as sucrose and inorganic salts such as sodium chloride include inactivated dengue virus, poloxamer surfactant, sodium chloride, sucrose and the like.
- Formulation containing (Patent Document 5), Formulation containing CTLA4Ig molecule, sodium chloride, sucrose, etc.
- Patent Document 6 Factor VIII, surfactant, calcium chloride, sucrose, sodium chloride, trisodium citrate, amino acid Formulation containing buffering agent not included
- Patent Document 7 formulation containing highly concentrated protein, sodium chloride, sucrose, etc.
- Patent Document 8 sodium carboxymethylcellulose, sodium chloride, polysorbate 20, Tween 20, 50: 50DL PLG 4A port Preparation containing mer, exendin-4, sucrose, etc.
- Patent Document 9 preparation containing Fc domain-containing peptide, L-arginine, sodium chloride, sucrose, etc.
- Patent Document 10 human serum albumin, sucrose, glutamic acid , A preparation containing sodium chloride, alkali metal phosphate (Patent Document 11), a preparation containing heparan N-sulfatase protein, sodium chloride, buffer, polysorbate surfactant, sucrose, etc.
- Patent Document 12 has been.
- the amino acid sequences of the peptides are different, the stability is also greatly different, and even if the disclosed composition is applied, it does not necessarily contribute to the stability improvement.
- the present invention provides a composition that contains one or more peptides, can dissolve various peptides in water, has high stability, and can be stored for a long period of time.
- the present invention [1] One or more peptides having the amino acid sequence represented by any one of SEQ ID NOs: 1 to 5, or 1 to 2 amino acids of the peptide, each independently substituted, deleted or added A composition comprising a peptide, a saccharide and an inorganic salt, [2] The composition according to [1] above, wherein the second amino acid from the N-terminus has a peptide that is phenylalanine, tyrosine, methionine, tryptophan, or threonine.
- composition according to [1] or [2] above which contains a peptide whose C-terminal amino acid is phenylalanine, leucine, isoleucine, tryptophan or methionine
- the composition according to [1] above comprising 5 types of peptides consisting of the amino acid sequences represented by SEQ ID NOs: 1 to 5,
- composition according to the above [6], wherein the saccharide is sucrose [8] The inorganic salt according to any one of [1] to [7], wherein the inorganic salt is one or more selected from the group consisting of sodium chloride, potassium chloride, magnesium chloride, sodium sulfite, and sodium pyrosulfite.
- composition [9] The composition according to the above [8], wherein the inorganic salt is sodium chloride, [10] The composition according to any one of [1] to [9], which is a freeze-dried preparation, [11] The composition according to the above [10], wherein the weight of the peptide is 1.0 to 20 mg, [12] The composition according to [10] above, wherein the concentration of the peptide when dissolved in water so that the total amount is 2.0 g is 0.05 to 1.0% by weight, [13] The composition according to any one of [10] to [12], wherein the weight of the saccharide is 20 to 70 mg, [14] The composition according to any one of [10] to [12], wherein the weight ratio of the peptide to the saccharide in the composition is 1: 1.7 to 1: 29.2.
- composition [19] The composition according to any one of [10] to [18], which has a pH of 7.0 to 9.5 when dissolved in water, [20] The composition according to any one of [1] to [9], which is a water-in-oil emulsion formulation, [21] The composition according to [20], prepared from the composition according to any one of [10] to [19], [22] The composition according to any one of [1] to [21], which is an injection, [23] The composition according to any one of [10] to [22] above, wherein the oxygen concentration in the vial is 0.01% or more and less than 3.0%, [24] A method of preparing an emulsion by mixing the composition according to any one of [10] to [19] and an adjuvant, [25] A kit comprising the composition according to any one of [1] to [23], [26] At least the following steps: a) a step of mixing a highly stable peptide, saccharide and inorganic salt in a solution having a pH
- composition that is at least 01% and less than 3.0%; [32] Peptide having amino acid sequence represented by SEQ ID NO: 1, peptide having amino acid sequence represented by SEQ ID NO: 2, peptide having amino acid sequence represented by SEQ ID NO: 3, amino acid represented by SEQ ID NO: 4 A composition comprising a peptide having the sequence, a peptide having the amino acid sequence represented by SEQ ID NO: 5, sucrose and sodium chloride, [33] 1.0 to 2.4 mg of the peptide having the amino acid sequence represented by SEQ ID NO: 1, 1.0 to 2.4 mg of the peptide having the amino acid sequence represented by SEQ ID NO: 2, represented by SEQ ID NO: 3 1.0 to 2.4 mg of the peptide having the amino acid sequence shown below, 1.0 to 2.4 mg of the peptide having the amino acid sequence represented by SEQ ID NO: 4 and the peptide having the amino acid sequence represented by SEQ ID NO: 5
- a composition capable of storing one or more peptides in a stable state for a long period of time is provided. Furthermore, stability can be improved by controlling the pH of the solution. In yet another aspect, improved peptide stability can be achieved by controlling the oxygen concentration in the vial.
- the pH dependence of the solubility of each peptide is shown.
- the increase amount immediately after the preparation of the peptide-related analog having the amino acid sequence represented by SEQ ID NO: 1 Show.
- the increase amount immediately after the preparation of the peptide-related analog having the amino acid sequence represented by SEQ ID NO: 2 Show.
- the increase amount immediately after preparation of a peptide-derived related substance having the amino acid sequence represented by SEQ ID NO: 3 Show.
- the residual ratio immediately after the preparation of the peptide having the amino acid sequence represented by SEQ ID NO: 4 when the solution of the composition having a pH of 8 to 11 is allowed to stand for 6 hours at 25 ° C. is shown.
- the residual ratio immediately after the preparation of the peptide having the amino acid sequence represented by SEQ ID NO: 5 when the solution of the composition having a pH of 8 to 11 is allowed to stand for 6 hours at 25 ° C. is shown.
- Derived from a peptide having the amino acid sequence represented by SEQ ID NO: 3 when a freeze-dried preparation of a composition containing any of sucrose, trehalose, glucose, dextran, or glycerin is allowed to stand at 40 ° C. and 75% humidity for 2 weeks.
- the amount of related substances increased immediately after the preparation.
- the amino acid sequence represented by SEQ ID NO: 3 when a freeze-dried preparation of the composition having an amount of sucrose contained in the composition of 30 mg, 60 mg or 100 mg is left to stand for 3 months at 40 ° C. and 75% humidity, respectively.
- the increase amount with respect to immediately after preparation of the related substance derived from the peptide which has is shown.
- the amino acid represented by SEQ ID NO: 3 when a freeze-dried preparation of the composition having an amount of sodium chloride contained in the composition of 0 mg, 2 mg, or 4 mg is allowed to stand for 3 months at 40 ° C. and 75% humidity, respectively.
- the increase amount immediately after preparation of the related substance derived from the peptide which has a sequence is shown.
- the freeze-dried preparation of the composition in which the amount of sodium chloride contained in the composition is 2 mg the dry state of the vial without shrinkage is shown.
- the lyophilized formulation of the composition whose amount of sodium chloride contained in the composition is 4 mg shows the dry state of a completely contracted vial.
- the freeze-dried preparation of the composition having a pH of 8.0, 8.5, 9.0, or 9.5 in solution is allowed to stand for 3 months at 25 ° C. and 60% humidity, respectively.
- the increase amount with respect to immediately after preparation of the related substance derived from the peptide which has an amino acid sequence represented by sequence number 3 is shown.
- “Peptide” is used interchangeably herein and refers to a polymer of 2 to 100 amino acids. This polymer may be linear, branched, or cyclic.
- the amino acid may be natural or non-natural and may be a modified amino acid.
- the term may also include those assembled into a complex of multiple peptide chains.
- the term also encompasses natural or artificially modified amino acid polymers. Such modifications include, for example, disulfide bond formation, glycosylation, lipidation, acetylation, phosphorylation or any other manipulation or modification (eg, conjugation with a labeling component).
- This definition also includes, for example, peptides containing one or more analogs of amino acids (eg, including unnatural amino acids, etc.), peptide-like compounds (eg, peptoids) and other modifications known in the art. Is done.
- Protein cocktail refers to a composition comprising two or more peptides.
- amino acid may be natural or non-natural as long as it satisfies the object of the present invention.
- amino acid “substitution” results from the substitution of one or more amino acids by a different amino acid, respectively, when compared to a reference amino acid sequence (the amino acid sequence of a wild-type protein).
- An amino acid “deletion” is defined as a change in any of the amino acid sequences in which one or more amino acid residues are each not present when compared to the reference amino acid sequence (amino acid sequence of the wild-type protein).
- the An “addition” of an amino acid is either in the amino acid sequence or the N-terminus or C of the amino acid sequence resulting in the addition of one or more amino acid residues, respectively, when compared to a reference amino acid sequence (amino acid sequence of the wild-type protein). It is a change at the end.
- “Peptide having high stability in solution state” means that the residual ratio of the peptide is usually 95% or more, preferably 98% or more when left standing at pH 12, 25 ° C. and 60% relative humidity for 6 hours or more. And, when the peptide is a peptide having the amino acid sequence represented by SEQ ID NO: 3, the increase amount immediately after the preparation of the related substance derived from the peptide having the amino acid sequence represented by SEQ ID NO: 3 is usually pH 11, 25 A peptide that is 0.9% or less, preferably 0.8% or less, more preferably 0.5% or less when left at 6 ° C. and 60% relative humidity for 6 hours or more.
- the “stable peptide lyophilized preparation” is a peptide lyophilized preparation having only the peptide having the amino acid sequence represented by SEQ ID NO: 3, usually when left at 25 ° C. for 3 months or more. This refers to a lyophilized preparation of a peptide having an increase of 2.0% or less, preferably 1.5% or less, more preferably 1.0% or less with respect to immediately after preparation of the related substance derived from the peptide. “The stability of the peptide cocktail is high” means that, in a lyophilized state, the increase amount immediately after the preparation of the peptide-related analog having the amino acid sequence represented by SEQ ID NO: 3 is usually 25 ° C. and 60% relative humidity.
- the “analogous substance derived from the peptide having the amino acid sequence represented by SEQ ID NO: 3” includes an oxidized form and / or a dimer of the peptide having the amino acid sequence represented by SEQ ID NO: 3,
- the related substance derived from the peptide having the amino acid sequence represented by No. 3 particularly refers to an oxidant.
- Oxidized substance refers to a structure in which the cysteine thiol constituting the peptide is oxidized to form sulfinic acid.
- W / O emulsion or “water-in-oil emulsion” refers to the dispersion of an aqueous phase into an oil phase.
- Weight ratio indicates the ratio of the target composition to the peptide in the composition. In particular, the ratio of the weight of the target composition when the weight of the peptide is 1 is shown.
- “Adjuvant” increases the level of immune response in body fluids or cells and prolongs the response period. Therefore, by using an adjuvant, the number of injections and the amount of antigen contained in the vaccine can be reduced, and the vaccine inoculation cost can be kept low.
- Oxygen concentration is the ml volume of oxygen contained in 100 ml of medium such as air, and the value is expressed in%.
- a single dose vial is a vial containing a composition used for a single dose. This includes not only administering the entire amount of the composition contained in the vial, but also administering a partial amount of the composition contained in the vial.
- composition containing the peptide of the present invention can be used as a solution (eg, injection) in which the peptide and the following additives are dissolved.
- the liquid medium of the solution is dried to obtain a solid preparation (eg, lyophilized preparation). ). If it is a solid preparation, the composition can be stored for a long period of time.
- Examples of the peptides included in the present invention include one or more peptides selected from peptides derived from DEPDC1, MPHOSPH1, URLC10, CDCA1 and KOC1 protein.
- Examples of peptides derived from the DEPDC1 protein include peptides having the amino acid sequence EYYELFVNI (SEQ ID NO: 1) (Patent Document 1 (WO2008 / 047473)).
- Patent Document 1 WO2008 / 047473
- the peptide which has amino acid sequence IYNEYIYDL sequence number: 2 is illustrated (patent document 1 (WO2008 / 047473)).
- Examples of peptides derived from the URLC10 protein include peptides having the amino acid sequence RYCNLEGPPI (SEQ ID NO: 3) (Patent Document 2 (WO2006 / 090810)).
- Examples of the peptide derived from the CDCA1 protein include a peptide having the amino acid sequence VYGIRLLEHF (SEQ ID NO: 4) (Patent Document 3 (WO2009 / 153939)).
- a peptide derived from KOC1 protein a peptide having the amino acid sequence KTVNELQNL (SEQ ID NO: 5) is exemplified (Patent Document 2 (WO2006 / 090810)).
- 1 or 2 amino acids of these peptides may be substituted, deleted or added, or 1 or 2 amino acids may be substituted.
- the second amino acid from the N-terminus is phenylalanine, tyrosine, methionine, tryptophan or threonine, and / or the C-terminal amino acid is phenylalanine, leucine, isoleucine, tryptophan or Methionine.
- the peptides included in the present invention are particularly preferably 5 types of peptides consisting of the amino acid sequences represented by SEQ ID NOs: 1 to 5.
- the content of the peptide included in the present invention is preferably 1.0 to 20 mg, more preferably 2.0 to 15 mg in a single-dose vial regardless of the preparation such as a solution preparation and a lyophilized preparation. More preferably, it is 5.0 to 14 mg, particularly preferably 10 to 13 mg, and still more preferably 12 mg. In particular, when 5 types of peptides are contained in a single dose vial, the peptide content is preferably 9 to 13 mg, particularly preferably 10 to 12 mg.
- the concentration of the peptide when the lyophilized preparation in a single dose vial is dissolved in water (eg, distilled water, purified water, physiological saline, etc.) so that the total amount becomes 2.0 g is preferably 0.
- the peptide concentration is preferably 0.45% to 0.65% by weight, particularly preferably 0.5% to 0.6%.
- saccharides included in the present invention saccharides listed in the Japanese Pharmacopoeia, the Japanese Pharmacopoeia Pharmaceutical Standards or the Pharmaceutical Additives Standard, etc. can be used.
- monosaccharide glucose, fructose, ribose, xylose, mannose, maltotriose
- disaccharide lactose, cellobiose, purified sucrose, maltose, trehalose, etc.
- trisaccharide raffinose, etc.
- sugar alcohol D-sorbitol, Inositol, D-mannitol, etc.
- polysaccharides disaccharides
- disaccharides dextran, chondroitin sulfate, hyaluronic acid, dextrin sulfate, etc.
- salts thereof sodium chondroitin sulfate, sodium hyaluronate, etc.
- cyclic saccharides cyclodextrin,
- it is one or more saccharides selected from the group consisting of monosaccharides, disaccharides and polysaccharides. More preferably, it is one or more saccharides selected from the group consisting of glucose, galactose, fructose, sucrose, lactose, maltose, trehalose, dextran and glycerol. Particularly preferred is sucrose.
- the content of the saccharide contained in the present invention is preferably 20 to 70 mg, more preferably 40 to 70 mg, more preferably 40 mg in a single-dose vial regardless of the formulation such as a solution formulation and a lyophilized formulation. 50 to 70 mg, more preferably 55 to 65 mg, particularly preferably 60 mg.
- the weight ratio of peptide to saccharide in the composition of the present invention which is a lyophilized preparation is preferably from 1: 1.7 to 1: 29.2, more preferably from 1: 3.3 to 1: 5. .8, more preferably 1: 4.2 to 1: 5.8, still more preferably 1: 4.6 to 1: 5.4, and particularly preferably 1: 5.
- the concentration of the saccharide when the lyophilized preparation in a single dose vial is dissolved in water eg, distilled water, purified water, physiological saline, etc.
- water eg, distilled water, purified water, physiological saline, etc.
- the total amount becomes 2.0 g is preferably 1 0.0 to 3.5 wt%, more preferably 2.5 to 3.5 wt%, more preferably 2.8 to 3.2 wt%, and even more preferably 2.9 to 3.1 wt% Particularly preferably, it is 3.0% by weight.
- the amount of the saccharide contained in the composition of the present invention is excessive or insufficient, the stability of the peptide is lowered, and impurities derived from the peptide, in particular, an analog derived from a peptide having the amino acid sequence represented by SEQ ID NO: 3 An increase in body can be seen. Therefore, the amount of saccharide in the composition is preferably in the above range.
- the inorganic salt included in the present invention is preferably one or more inorganic salts selected from the group consisting of sodium chloride, potassium chloride, magnesium chloride, sodium sulfite and sodium pyrosulfite. More preferred is sodium chloride.
- the content of the inorganic salt contained in the present invention is preferably 0.1 to 3.0 mg, more preferably 1.3 mg in a single-dose vial, regardless of the formulation such as solution formulation and lyophilized formulation. It is 0 to 3.0 mg, more preferably 1.5 to 2.5 mg, and particularly preferably 2.0 mg.
- the weight ratio of the peptide to the inorganic salt in the composition of the present invention that is a lyophilized preparation is preferably 1: 0.008 to 1: 1.25, more preferably 1: 0.083 to 1: It is 0.25, more preferably 1: 0.125 to 1: 0.208, particularly preferably 1: 0.17.
- the concentration of the inorganic salt when the lyophilized preparation in a single dose vial is dissolved in water eg, distilled water, purified water, physiological saline, etc.
- water eg, distilled water, purified water, physiological saline, etc.
- the concentration of the inorganic salt when the lyophilized preparation in a single dose vial is dissolved in water (eg, distilled water, purified water, physiological saline, etc.) so that the total amount becomes 2.0 g is preferably It is 0.01 to 0.3% by weight, more preferably 0.05 to 0.2% by weight, still more preferably 0.05 to 0.15% by weight, and particularly preferably 0.1% by weight.
- the amount of the inorganic salt contained in the composition of the present invention is below the above range, the stability of the peptide is lowered, and the amount of impurities derived from the peptide can be increased.
- the content of the inorganic salt in the composition is insufficient, an increase in analogs derived from the peptide having the amino acid sequence represented by SEQ ID NO: 3 can be observed.
- the amount of the inorganic salt contained in the composition of the present invention exceeds the above range, when the composition of the present invention is freeze-dried, poor freeze-drying occurs and the shape of the freeze-dried product (cake) is deteriorated. . Therefore, the amount of the inorganic salt in the composition is preferably in the above range.
- a pH adjuster may be contained, and a pH adjuster listed in Japanese Pharmacopoeia, Japanese Pharmacopoeia Standards for Pharmaceuticals or Pharmaceutical Additives Standard, etc. can be used.
- a pH adjuster listed in Japanese Pharmacopoeia, Japanese Pharmacopoeia Standards for Pharmaceuticals or Pharmaceutical Additives Standard, etc. can be used.
- Preferred are hydrochloric acid and sodium hydroxide.
- the amount of these pH adjusting agents in the composition is arbitrary as long as it can be adjusted to a predetermined pH.
- the pH measurement method is carried out according to the pH measurement method described in the general test method of the Japanese Pharmacopoeia. HORIBA Ltd.
- the pH is measured within a range of 20 ° C. to 25 ° C. using a pH meter (HORIBA pH METER F-52) manufactured by the company.
- the pH when the composition of the present invention, which is a lyophilized preparation, is dissolved in water is preferably pH 7.0 to 9.5, more preferably The pH is 8.0 to 9.5, more preferably pH 8.2 to 9.0, and particularly preferably pH 8.4 to 8.6.
- the pH of the composition in the solution state of the present invention exceeds the above range, the stability of the peptide decreases, and the peptide-derived impurities, in particular, the peptide-derived analog having the amino acid sequence represented by SEQ ID NO: 3 The amount can increase.
- the pH of the composition in the solution state of the present invention is lower than the above range, the solubility of the peptide contained in the composition of the present invention is lowered and the peptide may be precipitated.
- peptides having the amino acid sequences represented by SEQ ID NO: 1, SEQ ID NO: 2 and SEQ ID NO: 4 have low solubility at a pH lower than the above range, and can precipitate.
- the optimal composition of the present invention is a composition in which the saccharide is sucrose and the inorganic salt is sodium chloride.
- composition of the present invention is a peptide having the amino acid sequence represented by SEQ ID NO: 1, a peptide having the amino acid sequence represented by SEQ ID NO: 2, a peptide having the amino acid sequence represented by SEQ ID NO: 3, A composition comprising a peptide having an amino acid sequence represented by 4, a peptide having an amino acid sequence represented by SEQ ID NO: 5, sucrose and sodium chloride.
- a more preferred composition of the present invention is 1.0 to 2.4 mg of the peptide having the amino acid sequence represented by SEQ ID NO: 1, and 1.0 to 2.4 mg of the peptide having the amino acid sequence represented by SEQ ID NO: 2.
- the present composition is, for example, a solution preparation, a lyophilized preparation, an emulsion preparation or the like. Preferably, it is a freeze-dried preparation. Freeze-drying is performed according to a known method, for example, using a freeze-dryer (manufactured by KyowaowVacuum.Engineering Co., Ltd).
- the concentration of one kind of peptide is 2.0 mg / ml (10 mg / ml when 5 kinds of peptides are included), for example, distilled water, purified water, preferably Can be prepared by dissolving in water such as purified water for injection and physiological saline, and mixing the solution with an oil phase such as an adjuvant to prepare a water-in-oil (W / O) emulsion.
- W / O water-in-oil
- the composition may be an oral preparation or an injection, but is preferably an injection.
- the oxygen concentration in the vial when sealed in the vial is preferably 0.01% or more and less than 3.0%, more preferably 0.01% or more and less than 2.0%, Preferably, it is 0.01% or more and less than 1.5%, and particularly preferably 0.01% or more and less than 1.0%.
- the stability of the peptide may be reduced and the amount of peptide-derived impurities may be increased.
- an increase in analogs derived from the peptide having the amino acid sequence represented by SEQ ID NO: 3 can be observed. Therefore, the peptide having the amino acid sequence represented by SEQ ID NO: 3 shows a decrease in stability due to oxygen.
- Oxygen concentration is measured non-destructively by irradiating the gas in the vial with a laser and monitoring the amount of absorption using a non-destructive headspace analyzer (such as Lighthouse Instruments). Can be measured.
- a non-destructive headspace analyzer such as Lighthouse Instruments
- the present invention also includes (a) a lyophilized formulation of the composition of the present invention, (b) a reconstituted solution of the lyophilized formulation of the composition of the present invention, and (c) a lyophilized formulation of the composition of the present invention.
- a kit comprising any of the water-in-oil emulsion formulations in which the reconstitution liquid and the adjuvant are mixed is provided.
- the composition of the present invention contained as a lyophilized preparation in the kit of the present invention may be any of the above compositions.
- the present invention also includes at least the following steps: a) a step of mixing a highly stable peptide, saccharide and inorganic salt in a solution having a pH of 12.0 or more and less than 13.0; b) adjusting the liquid prepared in the step a) to a pH of 10.0 or more and less than 12.0; c) a step of mixing a peptide having low stability with the solution prepared in the step b); d) a step of adjusting the liquid prepared in the step c) to a pH of 7.0 or more and less than 9.0; e) lyophilizing the mixture prepared in step d)
- a method for preparing a lyophilized formulation is provided.
- the present invention also provides at least the following steps: a) mixing a peptide having an amino acid sequence represented by SEQ ID NO: 1, a peptide having an amino acid sequence represented by SEQ ID NO: 4, sucrose and sodium chloride in a solution having a pH of 12.0 or more and less than 13.0; b) adjusting the liquid prepared in the step a) to pH 10.0 or more and less than 12.0 with an acidic substance; c) The liquid prepared in the step b) has a peptide having the amino acid sequence represented by SEQ ID NO: 2, a peptide having the amino acid sequence represented by SEQ ID NO: 3, and an amino acid sequence represented by SEQ ID NO: 5.
- the present invention also provides at least the following steps: a) mixing a peptide having an amino acid sequence represented by SEQ ID NO: 1, a peptide having an amino acid sequence represented by SEQ ID NO: 4, sucrose and sodium chloride in a solution having a pH of 12.0 or more and less than 13.0; b) a step of preparing the liquid prepared in the step a) with an acidic substance to have a pH of 11.0 or more and less than 12.0, c) mixing the peptide having the amino acid sequence represented by SEQ ID NO: 2 and the peptide having the amino acid sequence represented by SEQ ID NO: 3 with the solution prepared in the step b); d) a step of preparing the liquid prepared in the step c) to pH 9.0 or more and less than 11.0 with an acidic substance; e) mixing the peptide having the amino acid sequence represented by SEQ ID NO: 5 with the solution prepared in the step d); f) a step of preparing the liquid prepared in the step e) with an acidic substance
- the liquid can be adjusted with a pH adjusting agent.
- the present invention also provides a method for preparing a lyophilized formulation comprising a step of controlling the oxygen concentration in the vial of the lyophilized formulation obtained in the final step to 0.01% or more and less than 3.0%.
- a step of controlling the oxygen concentration in the vial of the lyophilized formulation obtained in the final step to 0.01% or more and less than 3.0%.
- the present invention also provides a freeze-dried preparation prepared by the above-mentioned freeze-dried preparation preparation method.
- the present invention also includes a peptide having the amino acid sequence represented by SEQ ID NO: 3, or a peptide in which 1 to 2 amino acids of the peptide may be substituted, deleted or added, and the oxygen concentration in the vial is Compositions that are 0.01% or more and less than 3.0% are provided.
- the oxygen concentration in the vial of the peptide having the amino acid sequence represented by SEQ ID NO: 3 is 3.0% or more, an increase in analogs derived from the peptide having the amino acid sequence represented by SEQ ID NO: 3 is observed. obtain. Therefore, since the peptide having the amino acid sequence represented by SEQ ID NO: 3 is affected by oxygen, it is preferably stored under low oxygen concentration conditions.
- composition of the present invention thus obtained is excellent in stability even when stored over time. Therefore, the composition of the present invention is useful as a preparation having excellent storage stability and stable drug efficacy.
- the pH of the solution is adjusted to pH 11.5 with an aqueous hydrochloric acid solution, and 15.9 g of the peptide having the amino acid sequence represented by SEQ ID NO: 2 and 16.5 g of the peptide having the amino acid sequence represented by SEQ ID NO: 5 And 16.9 g of a peptide having the amino acid sequence represented by SEQ ID NO: 3 was added.
- the pH of the solution was adjusted to pH 8.4 to 8.6 with an aqueous hydrochloric acid solution, and prepared with purified water for injection so that the total amount was 12500 g.
- the solution of the produced composition was aseptically filtered, and 2.0 g of the solution of the produced composition was dispensed into 3 ml glass vials and freeze-dried.
- the lyophilized composition was sealed in a glass vial and allowed to stand at 25 ° C. and 60% relative humidity, or 40 ° C. and 75% relative humidity for a certain period of time, and the composition was examined for visual appearance, content, and amount of related substances. Went.
- the content and the amount of related substances were measured by liquid chromatography described in the general test method of the Japanese Pharmacopoeia. Purified water for injection was added to the lyophilized composition, and a solution diluted so that the concentration of each peptide was 0.12 mg / mL was used as a sample solution. The content was calculated from the peak area of each drug substance obtained from the sample solution and the peak area of each drug substance obtained from the standard solution described below, and calculated from the dilution ratio and the ratio of the peak areas. The residual ratio of the content during the stability test was the ratio of the content immediately after preparation to the content. The amount of related substances was calculated from the ratio of peak areas by quantifying the peak areas of each drug substance and related substances obtained from the sample solution. The amount of increase in related substances during the stability test was the difference immediately after preparation.
- a column having an octadecyl group was used as the stationary phase, and 1.6 mg / mL phosphoric acid solution and acetonitrile were used as the mobile phase, and measurement was performed by a gradient elution method. The flow rate during the measurement was 1.0 mL / min.
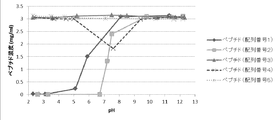
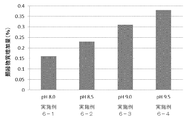
- the pH dependence of the solubility of each peptide is shown in FIG. It was found that the solubility of each peptide having the amino acid sequence represented by SEQ ID NO: 1 or 2 decreases as the pH decreases. Further, it was found that the solubility of the peptide having the amino acid sequence represented by SEQ ID NO: 4 decreases at a neutral pH.
- Purified water for injection and 0.1 mol / L aqueous sodium hydroxide solution are added to 15 mg of the peptide having the amino acid sequence represented by SEQ ID NOs: 1 and 2, respectively, the pH of the solution is adjusted to 10, and the peptide concentration is 1. It was set to 5 mg / g.
- a hydrochloric acid aqueous solution was added to each solution, and the solubility when the pH was gradually lowered was visually confirmed.
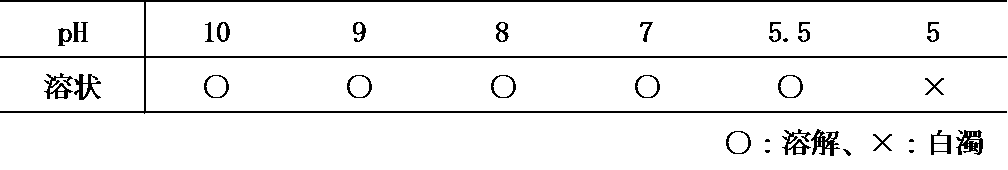
- the peptide having the amino acid sequence represented by SEQ ID NO: 1 was found to become cloudy when the pH of the solution was below 5.6 (Table 1-1). It was also found that the peptide having the amino acid sequence represented by SEQ ID NO: 2 became cloudy when the pH of the solution was below 5.5 (Table 1-2).
- the residual ratio of the peptide having the amino acid sequence represented by SEQ ID NO: 4 and the residual ratio of the peptide having the amino acid sequence represented by SEQ ID NO: 5 were measured.
- the method for measuring the related substance and the peptide content followed the above-described method.
- the results are shown in FIGS. 2-1 to 2-5. It was found that the related substance derived from the peptide having the amino acid sequence represented by SEQ ID NO: 1 did not change significantly depending on the solution pH and was relatively stable (FIG. 2-1). It has been found that the related substance derived from the peptide having the amino acid sequence represented by SEQ ID NO: 2 increases and the stability decreases as the solution pH increases (FIG. 2-2).
- the composition of the composition is as shown in Table 3.
- the above composition was mixed to the concentration shown in the above table, adjusted to pH 8 with an aqueous hydrochloric acid solution, and then 2.5 g of the above composition solution was dispensed into 3 ml glass vials.
- the composition solution was lyophilized.
- the freeze-dried preparation was allowed to stand at 40 ° C. and 75% humidity for 2 weeks, and the amount of increase relative to immediately after preparation of a peptide-related related substance having the amino acid sequence represented by SEQ ID NO: 3 was measured.
- the result is shown in FIG. From this result, it was found that sucrose with the smallest increase in related substances contributes to the stability improvement of the peptide cocktail.
- the composition of the composition is as shown in Table 4.
- the above composition was mixed to the concentration shown in the above table, adjusted to pH 8.5 with an aqueous hydrochloric acid solution, and then 2.0 g of the composition solution was dispensed into 3 ml glass vials and lyophilized.
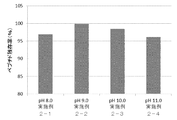
- the freeze-dried preparation was allowed to stand at 40 ° C. and 75% humidity for 3 months, and the amount of increase relative to immediately after preparation of a peptide-derived related substance having the amino acid sequence represented by SEQ ID NO: 3 was measured.
- the result is shown in FIG. From this result, it was found that when the amount of sucrose was 60 mg per vial (volume concentration of 30 mg / g), the increase amount immediately after the preparation of the related substance was low, which contributed to the improvement of the stability of the peptide cocktail.
- the composition of the composition is as shown in Table 5.
- the above composition was mixed to the concentration shown in the above table and adjusted to pH 8.5 with an aqueous hydrochloric acid solution, and 2.0 g of the solution of the composition was dispensed into 3 ml glass vials and lyophilized.
- the freeze-dried preparation was allowed to stand at 40 ° C. and 75% humidity for 3 months, and the increase in the amount of related substances derived from the peptide having the amino acid sequence represented by SEQ ID NO: 3 was measured.
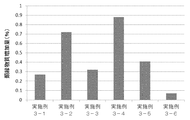
- the result is shown in FIG. From this result, it was found that the amount of sodium chloride was 4.0 mg per vial (2.0 mg / g as the solution concentration), the increase in the related substances was low, and contributed to the stability improvement of the peptide cocktail. .
- Example 5-2 Example 5-3 having a sodium chloride content of 2 mg (1.0 mg / g as a solution concentration) or 4 mg (2.0 mg / g as a solution concentration) per vial 827 vials and 809 vials, respectively, for a total of 1636 vials, were lyophilized under the same conditions.
- FIG. 5-2 is a photograph of a vial with 1 mg / g sodium chloride and no shrinkage.
- FIG. 5-3 is a photograph of a vial fully contracted with 2 mg / g of sodium chloride. From this result, it was found that when the sodium chloride content was 2 mg per vial (1.0 mg / g as the solution concentration), the number of freeze-dried preparations completely contracted was small, and the freeze-dried state was better.
- the sodium chloride content was 2.0 mg per vial (1.0 mg / g as the solution concentration). Judged to be the best.
- the composition of the composition is shown in Table 6.
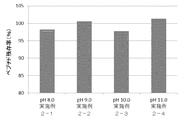
- the above composition was mixed at the concentration shown in the above table, adjusted to pH 8.0, 8.5, 9.0, or 9.5 with an aqueous hydrochloric acid solution, and 2.0 g of the solution of the composition was each 3 ml glass vial. Were aliquoted and lyophilized. The freeze-dried preparation was allowed to stand at 25 ° C. and 60% humidity for 3 months, and the increase in the amount of related substances derived from the peptide having the amino acid sequence represented by SEQ ID NO: 3 was measured. The result is shown in FIG. From this result, it was found that when the solution pH was 8.0, the increase amount of the related substance was low, and contributed to the stability improvement of the peptide cocktail.
- the peptide is dissolved in a stable state in a solution state (the peptide having the amino acid sequence represented by SEQ ID NO: 1 and the peptide having the amino acid sequence represented by SEQ ID NO: 4), Dissolving a peptide having low stability in a solution state (a peptide having an amino acid sequence represented by SEQ ID NO: 2, a peptide having an amino acid sequence represented by SEQ ID NO: 5 and an amino acid sequence represented by SEQ ID NO: 3); did.
- the peptide having high stability in the solution state is preferably dissolved in the order of the peptide having the amino acid sequence represented by SEQ ID NO: 1 and the peptide having the amino acid sequence represented by SEQ ID NO: 4.
- the peptide having low stability in the solution state includes a peptide having the amino acid sequence represented by SEQ ID NO: 2, a peptide having the amino acid sequence represented by SEQ ID NO: 5, and a peptide having the amino acid sequence represented by SEQ ID NO: 3. It was judged that it was preferable to dissolve in this order.
- the oxygen concentration in the vial is preferably less than 3.0% in order to obtain a highly stable peptide lyophilized preparation.
- the present invention is useful as a pharmaceutical composition containing a peptide, particularly as a peptide vaccine preparation for cancer treatment containing a peptide.
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Animal Behavior & Ethology (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Pharmacology & Pharmacy (AREA)
- Epidemiology (AREA)
- Engineering & Computer Science (AREA)
- Organic Chemistry (AREA)
- Biochemistry (AREA)
- Molecular Biology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Immunology (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Inorganic Chemistry (AREA)
- Genetics & Genomics (AREA)
- Gastroenterology & Hepatology (AREA)
- Biotechnology (AREA)
- Dispersion Chemistry (AREA)
- Biophysics (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Medicinal Preparation (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Peptides Or Proteins (AREA)
Abstract
Description
[1]配列番号1~5のいずれかで表されるアミノ酸配列を有する1種または2種以上のペプチド、または該ペプチドの1~2個のアミノ酸がそれぞれ独立して、置換、欠失または付加されていてもよいペプチド、糖類および無機塩を含有する組成物、
[2]N末端から2番目のアミノ酸が、フェニルアラニン、チロシン、メチオニン、トリプトファンまたはトレオニンであるペプチドを有する、前記[1]記載の組成物、
[3]C末端アミノ酸が、フェニルアラニン、ロイシン、イソロイシン、トリプトファンまたはメチオニンであるペプチドを含有する、前記[1]または[2]記載の組成物、
[4]配列番号1~5で表されるアミノ酸配列からなる5種のペプチドを含有する、前記[1]記載の組成物、
[5]糖類が、単糖類、二糖類および多糖類からなる群から選択される1または2以上である、前記[1]~[4]のいずれかに記載の組成物、
[6]糖類が、グルコース、ガラクトース、フルクトース、スクロース、ラクトース、マルトース、トレハロース、デキストランおよびグリセロールからなる群から選択される1または2以上である、前記[5]に記載の組成物、
[7]糖類が、スクロースである、前記[6]記載の組成物、
[8]無機塩が、塩化ナトリウム、塩化カリウム、塩化マグネシウム、亜硫酸ナトリウムおよびピロ亜硫酸ナトリウムからなる群から選択される1または2以上である、前記[1]~[7]のいずれかに記載の組成物、
[9]無機塩が、塩化ナトリウムである、前記[8]記載の組成物、
[10]凍結乾燥製剤である、前記[1]~[9]のいずれかに記載の組成物、
[11]ペプチドの重量が、1.0~20mgである、前記[10]記載の組成物、
[12]全量が2.0gになるように水で溶解した時におけるペプチドの濃度が、0.05~1.0重量%である、前記[10]記載の組成物、
[13]糖類の重量が、20~70mgである、前記[10]~[12]のいずれかに記載の組成物、
[14]組成物中のペプチドと糖類の重量比が、1:1.7~1:29.2である、前記[10]~[12]のいずれかに記載の組成物、
[15]全量が2.0gになるように水で溶解した時における糖類の濃度が、1.0~3.5重量%である、前記[10]~[12]のいずれかに記載の組成物、
[16]無機塩の重量が、0.1~3.0mgである、前記[10]~[15]のいずれかに記載の組成物、
[17]組成物中のペプチドと無機塩の重量比が、1:0.008~1:1.25である、前記[10]~[15]のいずれかに記載の組成物、
[18]全量が2.0gになるように水で溶解した時における無機塩の濃度が、0.01~0.3重量%である、前記[10]~[15]のいずれかに記載の組成物、
[19]水で溶解した時に、pH7.0~9.5である、前記[10]~[18]のいずれかに記載の組成物、
[20]油中水滴エマルション製剤である、前記[1]~[9]のいずれかに記載の組成物、
[21]前記[10]~[19]のいずれかに記載の組成物から調製された前記[20]記載の組成物、
[22]注射剤である、前記[1]~[21]のいずれかに記載の組成物、
[23]バイアル中の酸素濃度が0.01%以上、3.0%未満である、前記[10]~[22]のいずれかに記載の組成物、
[24]前記[10]~[19]のいずれかに記載の組成物とアジュバントを混合することにより、エマルションを調製する方法、
[25]前記[1]~[23]のいずれかに記載の組成物を含むキット、
[26]少なくとも以下の工程:
a)pH12.0以上、13.0未満の液に、安定性の高いペプチド、糖類および無機塩を混合する工程、
b)該a)の工程で調製した液を、pH10.0以上、12.0未満に調整する工程、
c)該b)の工程で調製した液に、安定性の低いペプチドを混合する工程、
d)該c)の工程で調製した液を、pH7.0以上、9.0未満に調整する工程、
e)該d)の工程で調製した混合物を凍結乾燥する工程、
を含有する、凍結乾燥製剤調製法、
[27]少なくとも以下の工程:
a)pH12.0以上、13.0未満の液に、配列番号1で表されるアミノ酸配列を有するペプチド、配列番号4で表されるアミノ酸配列を有するペプチド、スクロースおよび塩化ナトリウムを混合する工程、
b)該a)の工程で調製した液を、酸性物質でpH10.0以上、12.0未満に調整する工程、
c)該b)の工程で調製した液に、配列番号2で表されるアミノ酸配列を有するペプチド、配列番号3で表されるアミノ酸配列を有するペプチドおよび配列番号5で表されるアミノ酸配列を有するペプチドを混合する工程、
d)該c)の工程で調製した液を、酸性物質でpH7.0以上、9.0未満に調整する工程、
e)該d)の工程で調製した混合物を凍結乾燥する工程、
を含有する、凍結乾燥製剤調製法、
[28]少なくとも以下の工程:
a)pH12.0以上、13.0未満の液に、配列番号1で表されるアミノ酸配列を有するペプチド、配列番号4で表されるアミノ酸配列を有するペプチド、スクロースおよび塩化ナトリウムを混合する工程、
b)該a)の工程で調製した液を、酸性物質でpH11.0以上、12.0未満に調製する工程、
c)該b)の工程で調製した液に、配列番号2で表されるアミノ酸配列を有するペプチドおよび配列番号3で表されるアミノ酸配列を有するペプチドを混合する工程、
d)該c)の工程で調製した液を、酸性物質でpH9.0以上、11.0未満に調製する工程、
e)該d)の工程で調製した液に、配列番号5で表されるアミノ酸配列を有するペプチドを混合する工程、
f)該e)の工程で調製した液を、酸性物質でpH7.0以上、9.0未満に調製する工程、
g)該f)の工程で調製した混合物を凍結乾燥する工程、
を含有する、凍結乾燥製剤調製法、
[29]さらに、バイアル中の酸素濃度を0.01%以上、3.0%未満に制御する工程を含有する前記[26]~[28]のいずれかに記載の凍結乾燥製剤調製法、
[30]前記[26]~[28]のいずれかに記載の方法で調製した凍結乾燥製剤、
[31]配列番号3で表されるアミノ酸配列を有するペプチド、または該ペプチドの1~2個のアミノ酸が置換、欠失または付加されていてもよいペプチドを含み、バイアル中の酸素濃度が0.01%以上、3.0%未満である組成物、
[32]配列番号1で表されるアミノ酸配列を有するペプチド、配列番号2で表されるアミノ酸配列を有するペプチド、配列番号3で表されるアミノ酸配列を有するペプチド、配列番号4で表されるアミノ酸配列を有するペプチド、配列番号5で表されるアミノ酸配列を有するペプチド、スクロースおよび塩化ナトリウムを含有する組成物、
[33]配列番号1で表されるアミノ酸配列を有するペプチドが1.0~2.4mg、配列番号2で表されるアミノ酸配列を有するペプチドが1.0~2.4mg、配列番号3で表されるアミノ酸配列を有するペプチドが1.0~2.4mg、配列番号4で表されるアミノ酸配列を有するペプチドが1.0~2.4mg、配列番号5で表されるアミノ酸配列を有するペプチドが1.0~2.4mg、スクロースが20~60mgおよび塩化ナトリウムが1~2mgである前記[32]記載の組成物、
に関する。
アミノ酸の「欠失」は、1つ以上のアミノ酸残基が、それぞれ、参照アミノ酸配列(野生型タンパク質のアミノ酸配列)と比較した場合に存在していない、アミノ酸配列のいずれかにおける変化として規定される。
アミノ酸の「付加」は、参照アミノ酸配列(野生型タンパク質のアミノ酸配列)と比較した場合に、それぞれ、1つ以上のアミノ酸残基の付加を生じた、アミノ酸配列中又はアミノ酸配列のN末端若しくはC末端における変化である。
「安定性の高いペプチド凍結乾燥製剤」とは、配列番号3で表されるアミノ酸配列を有するペプチドのみを有するペプチド凍結乾燥製剤であり、通常、25℃で3か月間以上静置した場合において、該ペプチド由来の類縁物質の調製直後に対する増加量が、2.0%以下、好ましくは1.5%以下、より好ましくは1.0%以下であるペプチドの凍結乾燥製剤をいう。
「ペプチドカクテルの安定性が高い」とは、凍結乾燥状態において、配列番号3で表されるアミノ酸配列を有するペプチド由来の類縁物質の調製直後に対する増加量が、通常、25℃かつ60%相対湿度で3か月間静置した場合において、1.0%以下、好ましくは0.5%以下、より好ましくは0.3%以下、または40℃かつ75%相対湿度で2週間静置した場合において、1.0%以下、好ましくは0.5%以下、より好ましくは0.3%以下、または40℃かつ75%相対湿度で3か月間静置した場合において、1.5%以下、好ましくは1.0%以下、より好ましくは0.5%以下であるペプチドカクテルをいう。
本発明に含まれるペプチドは、好ましくは、N末端から2番目のアミノ酸が、フェニルアラニン、チロシン、メチオニン、トリプトファンまたはトレオニンであり、および/または、C末端のアミノ酸 が、フェニルアラニン、ロイシン、イソロイシン、トリプトファンまたはメチオニンである。
本発明に含まれるペプチドは、特に好ましくは、配列番号1~5で表されるアミノ酸配列からなる5種のペプチドである。
または、投与1回分のバイアルの凍結乾燥製剤を全量が2.0gになるように水(例:蒸留水、精製水、生理食塩水等)で溶解した時におけるペプチドの濃度は、好ましくは、0.05~1.0重量%、より好ましくは、0.1~1.0重量%、さらに好ましくは、0.3~1.0重量%、特に好ましくは、0.4~0.8重量%、さらに好ましくは0.6重量%である。特に投与1回分のバイアル中にペプチドが5種含まれる場合は、ペプチドの濃度は、0.45%~0.65重量%が好ましく、0.5%~0.6%が特に好ましい。
または、凍結乾燥製剤である本発明の組成物中のペプチドと糖類の重量比は、好ましくは、1:1.7~1:29.2、さらに好ましくは、1:3.3~1:5.8、より好ましくは、1:4.2~1:5.8、さらに好ましくは、1:4.6~1:5.4、特に好ましくは、1:5である。
または、投与1回分のバイアルの凍結乾燥製剤を全量が2.0gになるように水(例:蒸留水、精製水、生理食塩水等)で溶解した時における糖類の濃度は、好ましくは、1.0~3.5重量%、さらに好ましくは、2.5~3.5重量%、より好ましくは、2.8~3.2重量%、さらに好ましくは、2.9~3.1重量%、特に好ましくは3.0重量%である。
または、凍結乾燥製剤である本発明の組成物中のペプチドと無機塩の重量比は、好ましくは、1:0.008~1:1.25、より好ましくは、1:0.083~1:0.25、さらに好ましくは、1:0.125~1:0.208、特に好ましくは、1:0.17である。
または、投与1回分のバイアルの凍結乾燥製剤を全量が2.0gになるように水(例:蒸留水、精製水、生理食塩水等)で溶解した時における無機塩の濃度は、好ましくは、0.01~0.3重量%、より好ましくは、0.05~0.2重量%、さらに好ましくは、0.05~0.15重量%、特に好ましくは0.1重量%である。
一方、本発明の組成物中に含まれる無機塩の量が、上記範囲を上回る場合、本発明組成物を凍結乾燥した場合、凍結乾燥不良が起こり、凍結乾燥物(ケーキ)の形状が悪くなる。したがって、組成物中の無機塩の量は、上記範囲が好ましい。
一方、本発明の溶液状態の組成物のpHが、上記範囲を下回る場合、本発明の組成物に含まれるペプチドの溶解度が低下し、ペプチドが析出する恐れがある。特に、配列番号1、配列番号2および配列番号4で表されるアミノ酸配列を有するペプチドは、上記範囲を下回るpHで溶解度が低く、析出し得る。
本発明の凍結乾燥製剤のバイアルをアルミパウチ等の袋で密封して保存することで、バイアル中の酸素濃度を長期間にわたり上記範囲内で維持することができる。
本発明のキットに凍結乾燥製剤として含まれる本発明の組成物は、上記のいずれの組成物であってもよい。
a)pH12.0以上、13.0未満の液に、安定性の高いペプチド、糖類および無機塩を混合する工程、
b)該a)の工程で調製した液を、pH10.0以上、12.0未満に調整する工程、
c)該b)の工程で調製した液に、安定性の低いペプチドを混合する工程、
d)該c)の工程で調製した液を、pH7.0以上、9.0未満に調整する工程、
e)該d)の工程で調製した混合物を凍結乾燥する工程、
を含有する、凍結乾燥製剤調製法を提供する。
a)pH12.0以上、13.0未満の液に、配列番号1で表されるアミノ酸配列を有するペプチド、配列番号4で表されるアミノ酸配列を有するペプチド、スクロースおよび塩化ナトリウムを混合する工程、
b)該a)の工程で調製した液を、酸性物質でpH10.0以上、12.0未満に調整する工程、
c)該b)の工程で調製した液に、配列番号2で表されるアミノ酸配列を有するペプチド、配列番号3で表されるアミノ酸配列を有するペプチドおよび配列番号5で表されるアミノ酸配列を有するペプチドを混合する工程、
d)該c)の工程で調製した液を、酸性物質でpH7.0以上、9.0未満に調整する工程、
e)該d)の工程で調製した混合物を凍結乾燥する工程、
を含有する、凍結乾燥製剤調製法を提供する。
a)pH12.0以上、13.0未満の液に、配列番号1で表されるアミノ酸配列を有するペプチド、配列番号4で表されるアミノ酸配列を有するペプチド、スクロースおよび塩化ナトリウムを混合する工程、
b)該a)の工程で調製した液を、酸性物質でpH11.0以上、12.0未満に調製する工程、
c)該b)の工程で調製した液に、配列番号2で表されるアミノ酸配列を有するペプチドおよび配列番号3で表されるアミノ酸配列を有するペプチドを混合する工程、
d)該c)の工程で調製した液を、酸性物質でpH9.0以上、11.0未満に調製する工程、
e)該d)の工程で調製した液に、配列番号5で表されるアミノ酸配列を有するペプチドを混合する工程、
f)該e)の工程で調製した液を、酸性物質でpH7.0以上、9.0未満に調製する工程、
g)該f)の工程で調製した混合物を凍結乾燥する工程、
を含有する、凍結乾燥製剤調製法を提供する。
配列番号3で表されるアミノ酸配列を有するペプチドは、バイアル中の酸素濃度が3.0%以上であるとき、配列番号3で表されるアミノ酸配列を有するペプチド由来の類縁体の増加がみられ得る。したがって、配列番号3で表されるアミノ酸配列を有するペプチドは、酸素の影響を受けるため、低酸素濃度条件下で保存することが好ましい。
8987.5gの注射用精製水に625gの1mol/L水酸化ナトリウム水溶液を添加し、375gのスクロースおよび12.5gの塩化ナトリウムを添加した。次いで、16.7gの配列番号1で表されるアミノ酸配列を有するペプチドおよび17.7gの配列番号4で表されるアミノ酸配列を有するペプチドを添加した。次いで、該溶液のpHを塩酸水溶液でpH11.5に調整し、15.9gの配列番号2で表されるアミノ酸配列を有するペプチド、16.5gの配列番号5で表されるアミノ酸配列を有するペプチドおよび16.9gの配列番号3で表されるアミノ酸配列を有するペプチドを添加した。次いで、該溶液のpHを塩酸水溶液でpH8.4~8.6に調整し、全量12500gになるよう注射用精製水で調製した。上記製造した組成物の溶液を無菌濾過した後、上記製造した組成物の溶液2.0gをそれぞれ3ml容のガラスバイアルに分注し、凍結乾燥を行った。
上記凍結乾燥組成物をガラスバイアルに密封した状態で25℃かつ60%相対湿度または40℃かつ75%相対湿度で一定期間静置し、該組成物の目視による外観、含量、類縁物質量について試験を行った。
含量および類縁物質量は、日本薬局方の一般試験法に記載の液体クロマトグラフィーにより測定した。凍結乾燥組成物に注射用精製水を添加し、各ペプチドの濃度が0.12mg/mLになるように希釈した溶液を試料溶液とした。含量は試料溶液から得られる各原薬のピーク面積と下記記載の標準溶液から得られる各原薬のピーク面積を定量し、希釈倍率及びピーク面積の比率から算出した。安定性試験時の含量の残存率は対調製直後の含量の比率とした。類縁物質量は試料溶液から得られる各原薬および類縁物質のピーク面積を定量し、ピーク面積の比率から算出した。安定性試験時の類縁物質の増加量は対調製直後の差分とした。
配列番号1で表されるアミノ酸配列を有するペプチドまたは配列番号2で表されるアミノ酸配列を有するペプチドの含量を測定するための標準溶液として、1.6mg/mLのリン酸溶液に上記のペプチドを添加し、各ペプチドの濃度を0.15mg/mLとなるよう希釈した溶液を標準溶液とした。配列番号3で表されるアミノ酸配列を有するペプチド、配列番号4で表されるアミノ酸配列を有するペプチドまたは配列番号5で表されるアミノ酸配列を有するペプチドの含量を測定するための標準溶液として、12.1mg/mLの2‐アミノ‐2‐ヒドロキシメチル‐1,3‐プロパンジオール水溶液に上記のペプチドを添加し、各ペプチドの濃度を0.15mg/mLとなるよう希釈した溶液を標準溶液とした。固定相にはオクタデシル基を有するカラムを用い、移動相には1.6mg/mLのリン酸溶液及びアセトニトリルを使用し、グラジエント溶出法により測定した。測定中の流速は1.0mL/分とした。
pH2~12における、配列番号1~5で表されるアミノ酸配列を有する各ペプチドの溶解度を検討した。注射用水に塩酸または水酸化ナトリウム溶液を入れ、pH調整した溶液に各ペプチドを3mg/mLとなるように混合した。混合後の溶液をフィルターでろ過し、ろ過液をそのまま、または注射用精製水で10~20倍に希釈し、液体クロマトグラフィー(上記の条件と同一)を用いてそれぞれのペプチドのピーク面積を定量した。それぞれのペプチドのピーク面積、ろ過液の希釈倍率、上記の標準溶液のピーク面積および濃度から、ペプチドの濃度を算出した。各ペプチドの溶解性のpH依存を図1に示す。配列番号1または2で表されるアミノ酸配列を有する各ペプチドは、pHが低下するほど溶解度が低下することが判明した。また、配列番号4で表されるアミノ酸配列を有するペプチドは、中性付近のpHで溶解度が低下することが判明した。
上記検討1のように、注射用水に直接ペプチドを添加すると、塊になり溶解しにくくなる。そこで、高pHで完全に溶解させた後、溶液のpHを徐々に下げて白濁するpHを見出すことで、熱力学的な溶解度を検討した。
配列番号1および2で表されるアミノ酸配列を有する各ペプチドをpH10の溶液にそれぞれ溶解させ、その後pHを低下させた場合の溶解度について検討した。配列番号1および2で表されるアミノ酸配列を有するペプチド15mgにそれぞれ注射用精製水および0.1 mol/L水酸化ナトリウム水溶液を加え、溶液のpHを10に調整し、ペプチドの濃度が1.5mg/gとなるようにした。それぞれの溶液に対して塩酸水溶液を加え、pHを徐々に下げたときの溶解性を目視により確認した。配列番号1で表されるアミノ酸配列を有するペプチドは、溶液のpHが5.6を下回ると溶液が白濁することが判明した(表1-1)。また、配列番号2で表されるアミノ酸配列を有するペプチドは、溶液のpHが5.5を下回ると溶液が白濁することが判明した(表1-2)。
pH8~11における、配列番号1~5で表されるアミノ酸配列を有するペプチドの溶液状態での各ペプチドの安定性について検討した。組成物の組成は表2のとおりである。
上記組成の調製した溶液を25℃で6時間静置し、配列番号1で表されるアミノ酸配列を有するペプチド由来の類縁物質の増加量、配列番号2で表されるアミノ酸配列を有するペプチド由来の類縁物質の増加量、配列番号3で表されるアミノ酸配列を有するペプチド由来の類縁物質の増加量をそれぞれ測定した。また、配列番号4で表されるアミノ酸配列を有するペプチドの残存率、配列番号5で表されるアミノ酸配列を有するペプチドの残存率をそれぞれ測定した。類縁物質およびペプチド含量の測定方法は上述の手法に従った。その結果を図2-1~図2-5に示す。配列番号1で表されるアミノ酸配列を有するペプチド由来の類縁物質は、溶液pHによって増加量が著しく変わらず、比較的安定であることが判明した(図2-1)。配列番号2で表されるアミノ酸配列を有するペプチド由来の類縁物質は、溶液pHが高くなると増加し、安定性が低下することが判明した(図2-2)。配列番号3で表されるアミノ酸配列を有するペプチド由来の類縁物質は溶液pHが10.0~11.0の場合に増加し、安定性が低下することが判明した(図2-3)。配列番号4で表されるアミノ酸配列を有するペプチドの残存率は、溶液pHによって著しく変わらず、比較的安定であることが判明した(図2-4)。配列番号5で表されるアミノ酸配列を有するペプチドの残存率は、溶液pHが高くなると著しく低下し、安定性が低下することが判明した(図2-5)。
組成物に含まれる糖類が、スクロース、トレハロース、ブドウ糖、デキストランまたはグリセリンのうち、いずれがペプチドカクテルの安定性向上に寄与するか検討した。組成物の組成は表3のとおりである。
上記組成物を上記表の濃度となるよう混合し、塩酸水溶液でpH8に調整した後、上記組成物の溶液2.5gをそれぞれ3ml容のガラスバイアルに分注した。該組成物の溶液を凍結乾燥した。
該凍結乾燥製剤を40℃、75%湿度下で2週間静置し、配列番号3で表されるアミノ酸配列を有するペプチド由来の類縁物質の調製直後に対する増加量を測定した。その結果を図3に示す。この結果から、類縁物質の増加量が最も低いスクロースがペプチドカクテルの安定性向上に寄与することが判明した。
組成物に含まれるスクロースの量が、いずれの場合にペプチドカクテルの安定性向上に寄与するか検討した。組成物の組成は表4のとおりである。
上記組成物を上記表の濃度となるよう混合し、塩酸水溶液でpH8.5に調整した後、該組成物の溶液2.0gをそれぞれ3ml容のガラスバイアルに分注し、凍結乾燥した。該凍結乾燥製剤を40℃、75%湿度下で3か月間静置し、配列番号3で表されるアミノ酸配列を有するペプチド由来の類縁物質の調製直後に対する増加量を測定した。その結果を図4に示す。この結果から、スクロースの量が1バイアルあたり60mg(容液濃度として30mg/g)の方が、類縁物質の調製直後に対する増加量が低く、ペプチドカクテルの安定性向上に寄与することが判明した。
組成物に含まれる塩化ナトリウムの量が、いずれの場合にペプチドカクテルの安定性向上に寄与するか検討した。組成物の組成は表5のとおりである。
上記組成物を上記の表の濃度となるよう混合し、塩酸水溶液でpH8.5に調整した後、該組成物の溶液2.0gをそれぞれ3ml容のガラスバイアルに分注し、凍結乾燥した。
該凍結乾燥製剤を40℃、75%湿度下で3か月間静置し、配列番号3で表されるアミノ酸配列を有するペプチド由来の類縁物質の増加量を測定した。その結果は、図5に示す。この結果から、塩化ナトリウムの量が1バイアルあたり4.0mg(溶液濃度として2.0mg/g)の方が、類縁物質の増加量が低く、ペプチドカクテルの安定性向上に寄与することが判明した。
凍結乾燥不良により、凍結乾燥物(ケーキ)が完全収縮することで、凍結乾燥物の形状が悪くなったものは不良品となる。そこで、組成物中の塩化ナトリウム含有量による凍結乾燥物の収縮の有無を検討した。塩化ナトリウムの含有量が1バイアルあたり2mg(溶液濃度として1.0mg/g)または4mg(溶液濃度として2.0mg/g)である上記組成物(実施例5-2、実施例5-3)をそれぞれ827バイアル、809バイアル、合計1636バイアル調製し、同条件で凍結乾燥した。そのときの凍結乾燥製剤の乾燥状態の内訳を表5-2に示し、代表的な凍結乾燥物を図5-2、図5-3に示す。図5-2は塩化ナトリウム1mg/gで収縮のないバイアルの写真である。図5-3は塩化ナトリウム2mg/gで完全収縮したバイアルの写真である。この結果から、塩化ナトリウムの含有量が1バイアルあたり2mg(溶液濃度として1.0mg/g)の方が、完全収縮した凍結乾燥製剤の数が少なく、凍結乾燥状態が良いことが判明した。
組成物の溶液のpHが、いずれの場合にペプチドカクテルの安定性向上に寄与するか検討した。組成物の組成は表6のとおりである。
上記組成物を上記の表の濃度で混合し、塩酸水溶液でpH8.0、8.5、9.0または9.5に調整し、該組成物の溶液2.0gをそれぞれ3ml容のガラスバイアルに分注し、凍結乾燥した。
該凍結乾燥製剤を25℃、60%湿度下で3か月間静置し、配列番号3で表されるアミノ酸配列を有するペプチド由来の類縁物質の増加量を測定した。その結果は、図6に示す。この結果から、溶液pHが8.0の方が、類縁物質の増加量が低く、ペプチドカクテルの安定性向上に寄与することが判明した。
配列番号3で表されるアミノ酸配列を有するペプチドの凍結乾燥製剤のバイアル中の酸素濃度を測定することで、ペプチド凍結乾燥製剤の安定性を検討した。凍結乾燥製剤の組成は表7-1のとおりである。
凍結乾燥バイアルを25℃、3か月間静置し、各バイアル中の酸素濃度を非破壊式ヘッドスペースアナライザで測定し、また、配列番号3で表されるアミノ酸配列を有するペプチド由来の類縁物質の量を測定した。その結果は表7-2のとおりである。この結果より、バイアル中の酸素濃度が低いほど、配列番号3で表されるアミノ酸配列を有するペプチド由来の類縁物質の増加量が小さいことが判明した。したがって、安定性の高いペプチド凍結乾燥製剤であるためには、バイアル中の酸素濃度が3.0%未満であることが好ましいことが判明した。
Claims (31)
- 配列番号1~5のいずれかで表されるアミノ酸配列を有する1種または2種以上のペプチド、または該ペプチドの1~2個のアミノ酸がそれぞれ独立して、置換、欠失または付加されていてもよいペプチド、糖類および無機塩を含有する組成物。
- N末端から2番目のアミノ酸が、フェニルアラニン、チロシン、メチオニン、トリプトファンまたはトレオニンであるペプチドを含有する、請求項1記載の組成物。
- C末端アミノ酸が、フェニルアラニン、ロイシン、イソロイシン、トリプトファンまたはメチオニンであるペプチドを含有する、請求項1または2記載の組成物。
- 配列番号1~5で表されるアミノ酸配列からなる5種のペプチドを含有する、請求項1記載の組成物。
- 糖類が、単糖類、二糖類および多糖類からなる群から選択される1または2以上である、請求項1~4のいずれかに記載の組成物。
- 糖類が、グルコース、ガラクトース、フルクトース、スクロース、ラクトース、マルトース、トレハロース、デキストランおよびグリセロールからなる群から選択される1または2以上である、請求項5記載の組成物。
- 糖類が、スクロースである、請求項6記載の組成物。
- 無機塩が、塩化ナトリウム、塩化カリウム、塩化マグネシウム、亜硫酸ナトリウムおよびピロ亜硫酸ナトリウムからなる群から選択される1または2以上である、請求項1~7のいずれかに記載の組成物。
- 無機塩が、塩化ナトリウムである、請求項8記載の組成物。
- 凍結乾燥製剤である、請求項1~9のいずれかに記載の組成物。
- ペプチドの重量が、1.0~20mgである、請求項10記載の組成物。
- 全量が2.0gになるように水で溶解した時におけるペプチドの濃度が、0.05~1.0重量%である、請求項10記載の組成物。
- 糖類の重量が、20~70mgである、請求項10~12のいずれかに記載の組成物。
- 組成物中のペプチドと糖類の重量比が、1:1.7~1:29.2である、請求項10~12のいずれかに記載の組成物。
- 全量が2.0gになるように水で溶解した時における糖類の濃度が、1.0~3.5重量%である、請求項10~12のいずれかに記載の組成物。
- 無機塩の重量が、0.1~3.0mgである、請求項10~15のいずれかに記載の組成物。
- 組成物中のペプチドと無機塩の重量比が、1:0.008~1:1.25である、請求項10~15のいずれかに記載の組成物。
- 全量が2.0gになるように水で溶解した時における無機塩の濃度が、0.01~0.3重量%である、請求項10~15のいずれかに記載の組成物。
- 水で溶解した時に、pH7.0~9.5である、請求項10~18のいずれかに記載の組成物。
- 油中水滴エマルション製剤である、請求項1~9のいずれかに記載の組成物。
- 請求項10~19のいずれかに記載の組成物から調製された請求項20記載の組成物。
- 注射剤である、請求項1~21のいずれかに記載の組成物。
- バイアル中の酸素濃度が0.01%以上、3.0%未満である、請求項10~22のいずれかに記載の組成物。
- 請求項10~19のいずれかに記載の組成物とアジュバントを混合することにより、エマルションを調製する方法。
- 請求項1~23のいずれかに記載の組成物を含むキット。
- 少なくとも以下の工程:
a)pH12.0以上、13.0未満の液に、安定性の高いペプチド、糖類および無機塩を混合する工程、
b)該a)の工程で調製した液を、pH10.0以上、12.0未満に調製する工程、
c)該b)の工程で調製した液に、安定性の低いペプチドを混合する工程、
d)該c)の工程で調製した液を、pH7.0以上、9.0未満に調製する工程、
e)該d)の工程で調製した混合物を凍結乾燥する工程、
を含有する、凍結乾燥製剤調製法。 - 少なくとも以下の工程:
a)pH12.0以上、13.0未満の液に、配列番号1で表されるアミノ酸配列を有するペプチド、配列番号4で表されるアミノ酸配列を有するペプチド、スクロースおよび塩化ナトリウムを混合する工程、
b)該a)の工程で調製した液を、酸性物質でpH10.0以上、12.0未満に調製する工程、
c)該b)の工程で調製した液に、配列番号2で表されるアミノ酸配列を有するペプチド、配列番号3で表されるアミノ酸配列を有するペプチドおよび配列番号5で表されるアミノ酸配列を有するペプチドを混合する工程、
d)該c)の工程で調製した液を、酸性物質でpH7.0以上、9.0未満に調製する工程、
e)該d)の工程で調製した混合物を凍結乾燥する工程、
を含有する、凍結乾燥製剤調製法。 - 少なくとも以下の工程:
a)pH12.0以上、13.0未満の液に、配列番号1で表されるアミノ酸配列を有するペプチド、配列番号4で表されるアミノ酸配列を有するペプチド、スクロースおよび塩化ナトリウムを混合する工程、
b)該a)の工程で調製した液を、酸性物質でpH11.0以上、12.0未満に調製する工程、
c)該b)の工程で調製した液に、配列番号2で表されるアミノ酸配列を有するペプチドおよび配列番号3で表されるアミノ酸配列を有するペプチドを混合する工程、
d)該c)の工程で調製した液を、酸性物質でpH9.0以上、11.0未満に調製する工程、
e)該d)の工程で調製した液に、配列番号5で表されるアミノ酸配列を有するペプチドを混合する工程、
f)該e)の工程で調製した液を、酸性物質でpH7.0以上、9.0未満に調製する工程、
g)該f)の工程で調製した混合物を凍結乾燥する工程、
を含有する、凍結乾燥製剤調製法。 - さらに、バイアル中の酸素濃度を0.01%以上、3.0%未満に制御する工程を含有する請求項26~28のいずれかに記載の凍結乾燥製剤調製法。
- 請求項26~28のいずれかに記載の方法で調製した凍結乾燥製剤。
- 配列番号3で表されるアミノ酸配列を有するペプチド、または該ペプチドの1~2個のアミノ酸が置換、欠失または付加されていてもよいペプチドを含み、バイアル中の酸素濃度が0.01%以上、3.0%未満である組成物。
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US15/527,938 US20180200325A1 (en) | 2014-11-18 | 2015-11-17 | Stabilized peptide composition |
| JP2016560224A JP6781508B2 (ja) | 2014-11-18 | 2015-11-17 | 安定化されたペプチド組成物 |
| EP15861133.5A EP3207936B1 (en) | 2014-11-18 | 2015-11-17 | Stable peptide composition |
| US17/851,339 US20220347259A1 (en) | 2014-11-18 | 2022-06-28 | Stabilized peptide composition |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2014-233604 | 2014-11-18 | ||
| JP2014233604 | 2014-11-18 |
Related Child Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US15/527,938 A-371-Of-International US20180200325A1 (en) | 2014-11-18 | 2015-11-17 | Stabilized peptide composition |
| US17/851,339 Division US20220347259A1 (en) | 2014-11-18 | 2022-06-28 | Stabilized peptide composition |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2016080367A1 true WO2016080367A1 (ja) | 2016-05-26 |
Family
ID=56013903
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2015/082179 WO2016080367A1 (ja) | 2014-11-18 | 2015-11-17 | 安定化されたペプチド組成物 |
Country Status (4)
| Country | Link |
|---|---|
| US (2) | US20180200325A1 (ja) |
| EP (1) | EP3207936B1 (ja) |
| JP (1) | JP6781508B2 (ja) |
| WO (1) | WO2016080367A1 (ja) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2022504937A (ja) * | 2018-10-19 | 2022-01-13 | ジェネンテック, インコーポレイテッド | 畳み込みニューラルネットワークによる凍結乾燥製剤における欠陥検出 |
| WO2022136440A1 (en) | 2020-12-21 | 2022-06-30 | Syngenta Crop Protection Ag | Pesticidal hexatoxin polypeptides and methods of use thereof |
Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2005530714A (ja) * | 2002-03-26 | 2005-10-13 | ソシエテ・ド・コンセイユ・ド・ルシエルシエ・エ・ダアツプリカーション・シヤンテイフイツク・(エス.セー.エール.アー.エス) | 第viii因子を含有する安定な医薬組成物 |
| JP2008530975A (ja) * | 2005-02-25 | 2008-08-14 | オンコセラピー・サイエンス株式会社 | Ttk、urlc10、またはkoc1ポリペプチドを発現する肺癌に対するペプチドワクチン |
| JP2010506826A (ja) * | 2006-10-17 | 2010-03-04 | オンコセラピー・サイエンス株式会社 | Mphosph1またはdepdc1ポリペプチドを発現する癌に対するペプチドワクチン |
| JP2010512356A (ja) * | 2006-12-11 | 2010-04-22 | エフ.ホフマン−ラ ロシュ アーゲー | 非経口aベータ抗体製剤 |
| JP2011524737A (ja) * | 2008-06-19 | 2011-09-08 | オンコセラピー・サイエンス株式会社 | Cdca1エピトープペプチドおよびそれを含むワクチン |
| WO2013133405A1 (ja) * | 2012-03-09 | 2013-09-12 | オンコセラピー・サイエンス株式会社 | ペプチドを含む医薬組成物 |
| JP2014509644A (ja) * | 2011-03-31 | 2014-04-21 | メディ−トックス インク | ボツリヌム毒素の凍結乾燥製剤 |
| JP2014530256A (ja) * | 2011-10-18 | 2014-11-17 | コヒラス・バイオサイエンシズ・インコーポレイテッド | 塩化ナトリウムによって安定化されたエタネルセプト製剤 |
Family Cites Families (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN102614134B (zh) * | 2005-03-25 | 2016-09-07 | 瑞泽恩制药公司 | Vegf拮抗剂制剂 |
| EP2453874A2 (en) * | 2009-07-14 | 2012-05-23 | Biogen Idec MA Inc. | Methods for inhibiting yellow color and peroxide formation in a composition |
| AR092325A1 (es) * | 2012-05-31 | 2015-04-15 | Regeneron Pharma | Formulaciones estabilizadas que contienen anticuerpos anti-dll4 y kit |
| EP2869816A4 (en) * | 2012-07-09 | 2016-04-20 | Coherus Biosciences Inc | FORMANTS OF ETANERCEPT HAVING A REDUCTION MARKED IN INVISIBLE PARTICLES IN THE NU |
-
2015
- 2015-11-17 WO PCT/JP2015/082179 patent/WO2016080367A1/ja active Application Filing
- 2015-11-17 US US15/527,938 patent/US20180200325A1/en not_active Abandoned
- 2015-11-17 JP JP2016560224A patent/JP6781508B2/ja active Active
- 2015-11-17 EP EP15861133.5A patent/EP3207936B1/en active Active
-
2022
- 2022-06-28 US US17/851,339 patent/US20220347259A1/en active Pending
Patent Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2005530714A (ja) * | 2002-03-26 | 2005-10-13 | ソシエテ・ド・コンセイユ・ド・ルシエルシエ・エ・ダアツプリカーション・シヤンテイフイツク・(エス.セー.エール.アー.エス) | 第viii因子を含有する安定な医薬組成物 |
| JP2008530975A (ja) * | 2005-02-25 | 2008-08-14 | オンコセラピー・サイエンス株式会社 | Ttk、urlc10、またはkoc1ポリペプチドを発現する肺癌に対するペプチドワクチン |
| JP2010506826A (ja) * | 2006-10-17 | 2010-03-04 | オンコセラピー・サイエンス株式会社 | Mphosph1またはdepdc1ポリペプチドを発現する癌に対するペプチドワクチン |
| JP2010512356A (ja) * | 2006-12-11 | 2010-04-22 | エフ.ホフマン−ラ ロシュ アーゲー | 非経口aベータ抗体製剤 |
| JP2011524737A (ja) * | 2008-06-19 | 2011-09-08 | オンコセラピー・サイエンス株式会社 | Cdca1エピトープペプチドおよびそれを含むワクチン |
| JP2014509644A (ja) * | 2011-03-31 | 2014-04-21 | メディ−トックス インク | ボツリヌム毒素の凍結乾燥製剤 |
| JP2014530256A (ja) * | 2011-10-18 | 2014-11-17 | コヒラス・バイオサイエンシズ・インコーポレイテッド | 塩化ナトリウムによって安定化されたエタネルセプト製剤 |
| WO2013133405A1 (ja) * | 2012-03-09 | 2013-09-12 | オンコセラピー・サイエンス株式会社 | ペプチドを含む医薬組成物 |
Non-Patent Citations (2)
| Title |
|---|
| See also references of EP3207936A4 * |
| YOSHIHISA SAKAKIBARA: "Iyaku Iryo Kigu Bun'ya ni Okeru Datsusansozai Shiyo no Genjo to Kongo no Tenkai", PHARM TECH JAPAN, vol. 20, no. 10, 2004, pages 2017 - 2024, XP009502950 * |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2022504937A (ja) * | 2018-10-19 | 2022-01-13 | ジェネンテック, インコーポレイテッド | 畳み込みニューラルネットワークによる凍結乾燥製剤における欠陥検出 |
| WO2022136440A1 (en) | 2020-12-21 | 2022-06-30 | Syngenta Crop Protection Ag | Pesticidal hexatoxin polypeptides and methods of use thereof |
Also Published As
| Publication number | Publication date |
|---|---|
| US20220347259A1 (en) | 2022-11-03 |
| EP3207936A1 (en) | 2017-08-23 |
| EP3207936B1 (en) | 2021-04-21 |
| JP6781508B2 (ja) | 2020-11-04 |
| US20180200325A1 (en) | 2018-07-19 |
| EP3207936A4 (en) | 2018-06-06 |
| JPWO2016080367A1 (ja) | 2017-08-24 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5752671B2 (ja) | Vegfアンタゴニスト製剤 | |
| US20220347259A1 (en) | Stabilized peptide composition | |
| TWI766890B (zh) | 於室溫穩定之冷凍乾燥蛋白質 | |
| CA2742659C (en) | Aqueous composition containing follicle-stimulating hormone | |
| WO2017104778A1 (ja) | 抗ヒトtslp受容体抗体含有医薬組成物 | |
| TWI579003B (zh) | 促性腺激素之新穎調製劑 | |
| KR100700963B1 (ko) | 켐토테신의 다당체 유도체를 함유하는 동결건조된 액상제제 | |
| JP2013514340A (ja) | Igf−iタンパク質、緩衝化剤および等張化剤を含む薬学的組成物 | |
| KR20220034053A (ko) | 재조합 단백질의 안정적인 제형 | |
| JP5865374B2 (ja) | B型ランチビオティックの点滴用製剤 | |
| EP3434283A1 (en) | Medicinal composition comprising peg anti-human ngf antibody fab' fragment | |
| TWI728409B (zh) | Hgf冷凍乾燥製劑 | |
| EP4200319A1 (en) | Annexin a1 n-terminal peptide formulations and methods | |
| WO2015056613A1 (ja) | 安定化されたポリペプチド水性製剤 | |
| JPH05112464A (ja) | 安定化された [Leu13−motilin−Hse 医薬組成物 | |
| JP2011516461A (ja) | 液体配合物及び凍結乾燥した配合物 | |
| US20220288167A1 (en) | A stable lyophilized formulation for hybrid fc fused g-csf | |
| JP6671141B2 (ja) | 懸濁液剤 | |
| EA045982B1 (ru) | СТАБИЛЬНЫЙ ЛИОФИЛИЗИРОВАННЫЙ СОСТАВ НА ОСНОВЕ G-CSF, СЛИТОГО С ГИБРИДНЫМ Fc | |
| TW202233225A (zh) | 穩定的酸性纖維母細胞生長因子組合物 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 15861133 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2016560224 Country of ref document: JP Kind code of ref document: A |
|
| REEP | Request for entry into the european phase |
Ref document number: 2015861133 Country of ref document: EP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 15527938 Country of ref document: US |