WO2014142146A1 - マイボーム機能不全の治療剤 - Google Patents
マイボーム機能不全の治療剤 Download PDFInfo
- Publication number
- WO2014142146A1 WO2014142146A1 PCT/JP2014/056416 JP2014056416W WO2014142146A1 WO 2014142146 A1 WO2014142146 A1 WO 2014142146A1 JP 2014056416 W JP2014056416 W JP 2014056416W WO 2014142146 A1 WO2014142146 A1 WO 2014142146A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- group
- hydroxymethyl
- hydroxy
- propionyloxy
- therapeutic agent
- Prior art date
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/44—Non condensed pyridines; Hydrogenated derivatives thereof
- A61K31/445—Non condensed piperidines, e.g. piperocaine
- A61K31/4523—Non condensed piperidines, e.g. piperocaine containing further heterocyclic ring systems
- A61K31/453—Non condensed piperidines, e.g. piperocaine containing further heterocyclic ring systems containing a six-membered ring with oxygen as a ring hetero atom
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/4353—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom ortho- or peri-condensed with heterocyclic ring systems
- A61K31/436—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom ortho- or peri-condensed with heterocyclic ring systems the heterocyclic ring system containing a six-membered ring having oxygen as a ring hetero atom, e.g. rapamycin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/66—Phosphorus compounds
- A61K31/675—Phosphorus compounds having nitrogen as a ring hetero atom, e.g. pyridoxal phosphate
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/0048—Eye, e.g. artificial tears
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/10—Dispersions; Emulsions
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/10—Dispersions; Emulsions
- A61K9/107—Emulsions ; Emulsion preconcentrates; Micelles
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P27/00—Drugs for disorders of the senses
- A61P27/02—Ophthalmic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P27/00—Drugs for disorders of the senses
- A61P27/02—Ophthalmic agents
- A61P27/04—Artificial tears; Irrigation solutions
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/06—Antiarrhythmics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/14—Vasoprotectives; Antihaemorrhoidals; Drugs for varicose therapy; Capillary stabilisers
Definitions
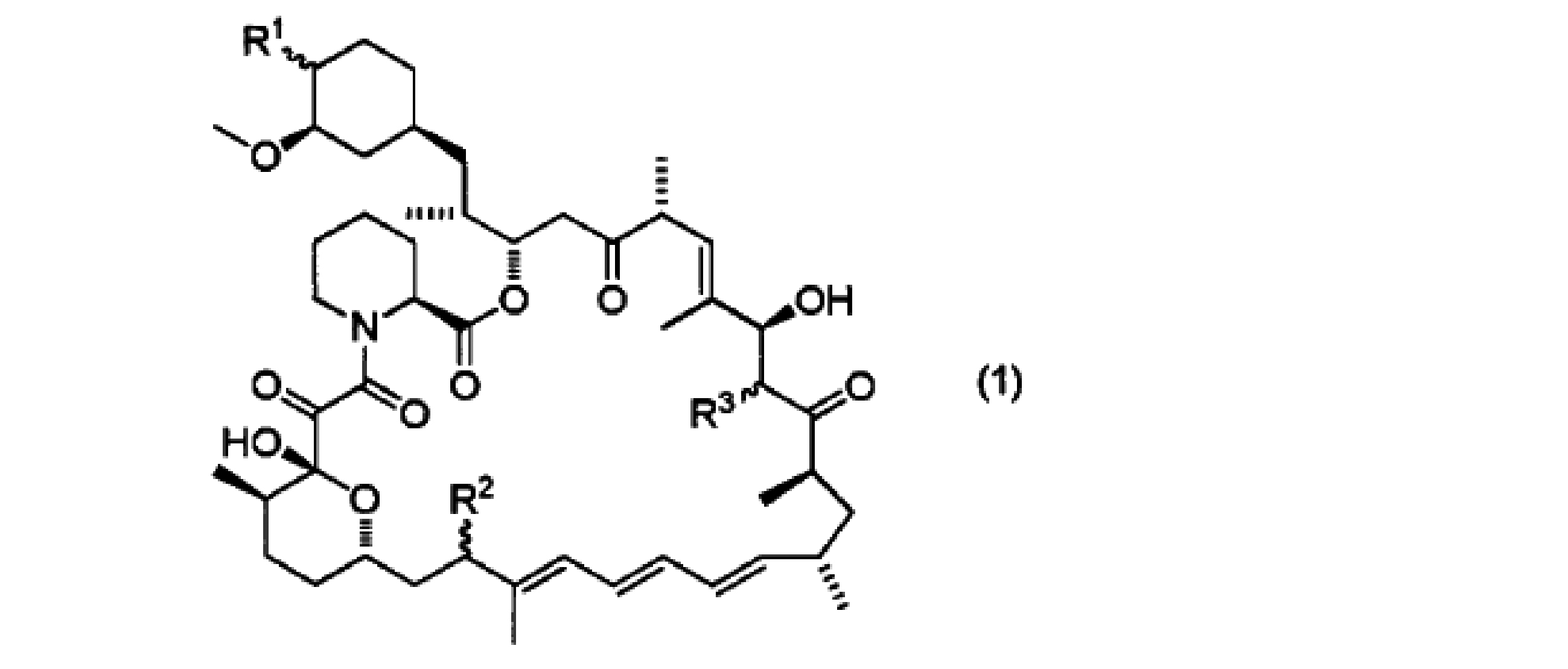
- R 1 is a hydroxy group, methoxy group, hydroxymethoxy group, ethoxy group, 1-hydroxyethoxy group, 2-hydroxyethoxy group, 2-methoxyethoxy group, 2-ethoxyethoxy group, formyloxy group, carboxyoxy group, acetoxy group , Hydroxyacetoxy group, propionyloxy group, 2-hydroxypropionyloxy group, 3-hydroxypropionyloxy group, 2-methylpropionyloxy group, 2- (hydroxymethyl) propionyloxy group, 3-hydroxy-2- (hydroxymethyl) Propionyloxy group, 2,2-dimethylpropionyloxy group, 2- (hydroxymethyl) -2-methylpropionyloxy group, 2,2-bis (hydroxymethyl) propionyloxy group, methylphosphinoyloxy group, dimethylphosphine Represents a finoyloxy group or a 1H-tetrazol-1-yl group; R 2 represents a hydrogen atom,
- the meibomian glands are sebaceous glands in the stencil and have openings at the upper and lower eyelid margins, and lipids secreted from the meibomian glands play various roles in the outer eye.
- Non-Patent Document 1 the symptoms such as eye discomfort in a significant proportion of the patients who visit the ophthalmology complaining, meibomian dysfunction (m eibomian g land d ysfunction: hereinafter also referred to as "MGD”) is the cause It is described that many patients cause a reduction in quality of life.
- MMD meibomian dysfunction
- MGD meibomian glands due to various causes. It is defined as “disease” and a disease accompanied by chronic eye discomfort (see Non-Patent Document 1).
- Non-Patent Document 1 also discloses that MGD is divided into secretion-decreasing MGD and secretion-increasing MGD, and that secretion of meibomian gland fat decreases due to obstruction of the opening of the meibomian gland. ing. Furthermore, Non-Patent Document 1 is positive for three symptoms: ⁇ 1> subjective symptoms such as eye discomfort, ⁇ 2> abnormal findings around the meibomian gland opening, such as vasodilation, and ⁇ 3> meibomian gland opening obstruction findings. Diagnosing the disease as a secreted MGD is also described.
- Non-Patent Document 1 discloses that dry eye may not accompany depending on the amount of tears, stage, or severity. .
- Non-Patent Document 2 discloses a classification method of MGD in the United States, and reduced secretion type MGD (low-delivery state MGD) is further converted to “hyposecretory MGD” and “obstructive MGD (mebibristo Migb)”. It is disclosed that it can be classified into two types. Furthermore, Non-Patent Document 2 suggests that MGD is one of the diseases that cause posterior blepharitis.
- Sirolimus also called rapamycin
- rapamycin is known as an immunosuppressant and is used as an oral preparation in the United States and the like.
- sirolimus derivative a derivative obtained by partially modifying the structure of sirolimus (hereinafter also referred to as “sirolimus derivative”) is known to have the same activity as sirolimus, and this derivative includes deferolimus, everolimus, temsirolimus. Zotarolimus, biolimus, novolimus and the like.
- Patent Document 1 contains an mTOR inhibitor such as sirolimus, everolimus, and temsirolimus, a first surfactant having an HLB index greater than about 10, and a second surfactant having an HLB index greater than about 13. An ophthalmic composition is described. However, Patent Document 1 does not describe whether or not the mTOR inhibitor has a therapeutic effect on MGD.
- Patent Document 2 suggests that pipecolic acid derivatives such as rapamycin can treat visual diseases, and the visual diseases include meibomian adenocarcinoma and internal stye (meibomian stye). Are listed. However, meibomian adenocarcinoma is a disease that is clearly different from MGD, and as described above, internal stye is also a disease different from MGD.
- Patent Documents 1 and 2 do not describe or suggest whether or not the present compound has a therapeutic effect on MGD.
- An object of the present invention is to provide a preventive and / or therapeutic agent for meibomian dysfunction (MGD).
- R 1 is a hydroxy group, methoxy group, hydroxymethoxy group, ethoxy group, 1-hydroxyethoxy group, 2-hydroxyethoxy group, 2-methoxyethoxy group, 2-ethoxyethoxy group, formyloxy group, carboxyoxy group, acetoxy group , Hydroxyacetoxy group, propionyloxy group, 2-hydroxypropionyloxy group, 3-hydroxypropionyloxy group, 2-methylpropionyloxy group, 2- (hydroxymethyl) propionyloxy group, 3-hydroxy-2- (hydroxymethyl) Propionyloxy group, 2,2-dimethylpropionyloxy group, 2- (hydroxymethyl) -2-methylpropionyloxy group, 2,2-bis (hydroxymethyl) propionyloxy group, methylphosphinoyloxy group, dimethylphosphine Represents a finoy
- the present invention relates to a prophylactic and / or therapeutic agent for meibomian dysfunction (MGD) (hereinafter referred to as the following) containing the compound of the above general formula (1) (present compound) or a pharmaceutically acceptable salt thereof as an active ingredient. Also referred to as “this drug”.
- MMD meibomian dysfunction
- the present compound has the above general formula (1), wherein R 1 is a hydroxy group, 2-hydroxyethoxy group, 2-ethoxyethoxy group, 2,2-bis (hydroxymethyl) propionyloxy group, dimethylphosphinoyloxy group.
- R 1 is a hydroxy group, 2-hydroxyethoxy group, 2-ethoxyethoxy group, 2,2-bis (hydroxymethyl) propionyloxy group, dimethylphosphinoyloxy group.
- a group or 1H-tetrazol-1-yl group a compound in which R 2 represents a hydrogen atom, a hydroxy group, a methoxy group or a methylthio group; and R 3 represents a hydrogen atom or a methoxy group, or a pharmaceutically acceptable salt thereof It is preferable that
- R 1 represents a hydroxy group or a dimethylphosphinoyloxy group
- R 2 represents a methoxy group
- R 3 represents a methoxy group
- An acceptable salt is preferred.
- this compound is sirolimus, deforolimus, everolimus, temsirolimus, zotarolimus, biolimus, novolimus, 7-epirapamycin, 7-thiomethyl-rapamycin, 7-epi-thiomethyl-rapamycin, 7-demethoxy-rapamycin, 32-demethoxy-rapamycin
- a pharmaceutically acceptable salt thereof is preferable, and sirolimus, deforolimus or a salt thereof is particularly preferable.
- the administration form of this drug is preferably ophthalmic administration or eyelid skin administration.
- the dosage form of this agent is preferably eye drops, eye ointments or ointments (excluding eye ointments), and the properties of the eye drops are preferably suspensions or emulsions.
- compositions for inhibiting occlusion of meibomian glands containing the present compound or a pharmaceutically acceptable salt thereof as an active ingredient (hereinafter also referred to as “the present composition”). It is.
- the active ingredient of the present composition is preferably sirolimus, deforolimus or a salt thereof.
- MMD meibomian dysfunction
- the present invention relates to a pharmaceutical composition for the prevention and / or treatment.
- Another aspect of the present invention relates to a compound of the above general formula (1) (the present compound) for use in the suppression of meibomian gland obstruction.
- Another aspect of the present invention relates to the compound of the above general formula (1) (the present compound) for use in suppressing the expansion of capillaries around the meibomian gland opening.
- Another aspect of the present invention relates to a compound of the above general formula (1) (present compound) for use in the prevention and / or treatment of meibomian dysfunction (MGD).
- MMD meibomian dysfunction
- Another aspect of the present invention relates to the use of the compound of the above general formula (1) (the present compound) for the manufacture of a composition for suppressing the obstruction of the meibomian glands.
- Another aspect of the present invention relates to the use of the compound of the above general formula (1) (the present compound) for the manufacture of a composition for suppressing the expansion of capillaries around the opening of the meibomian gland opening.
- Another aspect of the present invention relates to the use of the compound of the above general formula (1) (the present compound) for the manufacture of a medicament for preventing and / or treating meibomian dysfunction (MGD).
- MMD meibomian dysfunction
- another aspect of the present invention is a method for suppressing occlusion of meibomian glands, comprising administering the compound of the above general formula (1) (the present compound) to a human or an animal.
- Another aspect of the present invention is a method for suppressing expansion of capillaries around the meibomian gland opening, wherein the compound of the above general formula (1) (the present compound) is administered to humans or animals. Including methods.
- Another aspect of the present invention is a method for preventing and / or treating meibomian dysfunction (MGD), wherein the compound of the above general formula (1) (the present compound) is administered to a human or an animal. Including methods.
- the compound of the above general formula (1) or a pharmaceutically acceptable salt thereof is useful as a preventive and / or therapeutic agent for meibomian dysfunction (MGD).
- This compound has the general formula (1): Is a compound in which each group is a group shown below.
- R 1 is a hydroxy group, methoxy group, hydroxymethoxy group, ethoxy group, 1-hydroxyethoxy group, 2-hydroxyethoxy group, 2-methoxyethoxy group, 2-ethoxyethoxy group, formyloxy group, carboxyoxy group , Acetoxy group, hydroxyacetoxy group, propionyloxy group, 2-hydroxypropionyloxy group, 3-hydroxypropionyloxy group, 2-methylpropionyloxy group, 2- (hydroxymethyl) propionyloxy group, 3-hydroxy-2- ( Hydroxymethyl) propionyloxy group, 2,2-dimethylpropionyloxy group, 2- (hydroxymethyl) -2-methylpropionyloxy group, 2,2-bis (hydroxymethyl) propionyloxy group, methylphosphinoyloxy group, Jime Represents a tyrpphosphinoyloxy group or a 1H-tetrazol-1-yl group; (A2) R 2 represents a hydrogen atom, a hydroxy group,
- R 1 represents a hydroxy group, a 2-hydroxyethoxy group, a 2-ethoxyethoxy group, a 2,2-bis (hydroxymethyl) propionyloxy group, a dimethylphosphinoyloxy group, or a 1H-tetrazol-1-yl group.
- R 2 represents a hydrogen atom, a hydroxy group, a methoxy group, a methylthio group or a 2,4,6-trimethoxyphenyl group
- R 3 represents a hydrogen atom or a methoxy group. That is, a preferred example of the present compound is a combination of the groups listed in the above (B1), (B2) and (B3) in the general formula (1).
- (C) As a more preferable example of this compound, the compound whose each group is the group shown below in the said General formula (1) is mentioned.
- (C1) R 1 represents a hydroxy group, a 2-hydroxyethoxy group, a 2-ethoxyethoxy group, a 2,2-bis (hydroxymethyl) propionyloxy group or a dimethylphosphinoyloxy group;
- (C2) R 2 represents a methoxy group;
- (C3) R 3 represents a methoxy group; That is, a more preferable example of this compound is a compound represented by the above general formula (1), which is a combination of the groups listed in the above (C1), (C2) and (C3).
- the wavy line indicates that the carbon atom bonded to R 1 , R 2 or R 3 can take any configuration of S or R.
- the carbon bonded to R 1 The atom preferably has the R configuration, and when R 2 or R 3 is not a hydrogen atom, the carbon atom bonded to R 2 preferably has the S configuration, and the carbon atom bonded to R 3 preferably has the R configuration.
- a preferred specific example of the present compound is sirolimus or deforolimus, and sirolimus is particularly preferred.
- the present compound or a pharmaceutically acceptable salt thereof can be produced according to a usual method in the field of synthetic organic chemistry.
- sirolimus is commercially available from LKT laboratories (catalog number: R0161). Can also be used.
- Deforolimus can also be produced according to the method described in JP 2005-516065 A.
- Examples of the pharmaceutically acceptable salt of this compound include salts with inorganic acids such as hydrochloric acid, hydrobromic acid, hydroiodic acid, nitric acid, sulfuric acid, phosphoric acid; acetic acid, fumaric acid, maleic acid, succinic acid.
- inorganic acids such as hydrochloric acid, hydrobromic acid, hydroiodic acid, nitric acid, sulfuric acid, phosphoric acid; acetic acid, fumaric acid, maleic acid, succinic acid.
- Acid citric acid, tartaric acid, adipic acid, gluconic acid, glucoheptic acid, glucuronic acid, terephthalic acid, methanesulfonic acid, lactic acid, hippuric acid, 1,2-ethanedisulfonic acid, isethionic acid, lactobionic acid, oleic acid, pamoic acid , Salts with organic acids such as polygalacturonic acid, stearic acid, tannic acid, trifluoromethanesulfonic acid, benzenesulfonic acid, p-toluenesulfonic acid, lauryl sulfate, methyl sulfate, naphthalenesulfonic acid, sulfosalicylic acid; methyl bromide , Quaternary ammonium salts with methyl iodide, etc .; halogens such as bromine ion, chlorine ion, iodine ion And salt
- the present compound or a pharmaceutically acceptable salt may take the form of a hydrate or a solvate.
- crystal polymorphs and crystals Polymorph groups are also within the scope of the present invention.
- the crystal polymorphism group means that the crystal form changes depending on the conditions and state of production, crystallization and storage of these crystals (including the formulated state in this state). Means the individual crystal forms at each stage and the whole process.
- the present compound or a pharmaceutically acceptable salt thereof can be used for prevention and / or treatment of meibomian dysfunction (MGD).
- MMD meibomian dysfunction
- meibomian dysfunction is, for example, “a state in which the function of the meibomian gland has been diffusely abnormal due to various causes and is accompanied by chronic eye discomfort”.
- the state in which the function of the meibomian gland has been diffusely abnormal means, for example, capillar dilation, obstruction of the opening of the meibomian gland, not local meibomian gland abnormality observed in a chalazion, internal stye, etc. It means that abnormalities of meibomian glands such as are recognized diffusely.
- MGD is divided into secretion-decreasing MGD (low-delivery state) and secretion-increasing MGD (high delivery state), and is further expressed as “secretion-reducing MGD (low-delivery state MD)” (hyposecretory MGD (reactive state). And “obstructive MGD (meibionic grand obstruction)”.
- secretion of meibomian gland fat decreases due to obstruction of the opening of the meibomian gland. Further, in the secretory increased MGD, the secretion of meibomian gland fat increases due to various causes.
- the MGD in the present invention includes the meibomian gland infarction. Is also included.
- MGD for which the present compound or a pharmaceutically acceptable salt thereof can be treated is a secretory decreased type MGD.
- MGD may cause dry eye and may cause posterior blepharitis.
- MGD includes “MGD with dry eye and / or posterior blepharitis”, “MGD causing dry eye and / or posterior blepharitis”, “MGD without dry eye and not concomitant” , “MGD that does not cause dry eye”, “MGD that does not involve (not merge with) posterior blepharitis”, and “MGD that does not cause posterior blepharitis”.
- a prophylactic and / or therapeutic agent for MGD means a medicament for preventing and / or treating MGD.
- “treating and / or preventing MGD” means three indicators ( ⁇ 1> subjective symptoms such as eye discomfort, ⁇ 2> vasodilation, which are recognized in MGD patients disclosed in Non-Patent Document 1). At least improvement of "Meibomian gland opening obstruction findings", preferably "Meibomian gland opening such as vasodilation” This means that improvement in “surrounding abnormalities” and "observation of meibomian gland opening obstruction” is observed.
- Improvement of “abnormal findings around meibomian gland opening such as vasodilation” means that, for example, expansion of capillaries around the meibomian gland opening is suppressed.
- “suppressing expansion of the capillary around the meibomian gland opening” means, for example, improving “abnormal findings around the opening of the meibomian gland opening of the blood vessel”.
- Meibomian gland opening occlusion findings means, for example, that the meibomian gland opening occlusion is suppressed.
- “suppressing meibomian gland blockage” means, for example, improving “meibomian gland opening blockage finding”.
- the agent or the composition may contain an active ingredient other than the compound or a pharmaceutically acceptable salt thereof, and the compound or the pharmaceutically acceptable salt thereof is the only effective ingredient. It can also be contained as a component.
- the agent or the composition can be administered, for example, orally or parenterally to a patient, but is preferably administered parenterally.
- parenteral dosage forms include ophthalmic administration (including ophthalmic ointment), subconjunctival administration, intraconjunctival sac administration, subtenon sac administration, and the like, with ophthalmic administration being particularly preferred.
- parenteral administration forms include, for example, skin administration, but eyelid skin administration is particularly preferable as the skin administration in the present invention.
- the present compound or a pharmaceutically acceptable salt thereof is formulated into a dosage form suitable for administration together with a pharmaceutically acceptable additive as necessary.
- the dosage form suitable for oral administration include capsules, fine granules, granules, powders, pills, tablets and the like.
- dosage forms suitable for parenteral administration include eye drops, eye ointments, ointments (excluding eye ointments), injections, intraocular implant preparations (including punctal plugs), inserts, and patches. Agents, gels and the like. In addition, these can be prepared using the normal technique currently used widely in the said field
- the present compound or a pharmaceutically acceptable salt thereof can also be made into a DDS (drug delivery system) preparation such as microspheres.
- the preferred dosage form of the agent or the composition is an eye drop or an eye ointment, and an eye drop is particularly preferable.
- the properties of the eye drops may be dissolved solutions (dissolved eye drops) or suspensions (suspended eye drops). It may be an emulsion (emulsion ophthalmic solution), but is preferably a suspension or an emulsion.
- a preferable dosage form of the agent or the composition is an ointment (excluding an eye ointment).
- This compound or a pharmaceutically acceptable salt thereof includes, for example, excipients such as crystalline cellulose, lactose, glucose, D-mannitol, anhydrous calcium hydrogen phosphate, starch, sucrose; carboxymethylcellulose, carboxymethylcellulose calcium, cloth Disintegrants such as carmellose sodium, crospovidone, starch, partially pregelatinized starch, low substituted hydroxypropylcellulose; binders such as hydroxypropylcellulose, ethylcellulose, gum arabic, starch, partially pregelatinized starch, polyvinylpyrrolidone, polyvinyl alcohol ; Lubricants such as magnesium stearate, calcium stearate, talc, hydrous silicon dioxide, hydrogenated oil; refined sucrose, hydroxypropylmethylcellulose, hydroxypropyl cell Capsules, fine granules, granules, powders, coating agents such as glucose, methylcellulose, ethylcellulose, polyvinyl pyrrolidone, etc
- the eye drops are, for example, oil components such as medium chain fatty acid triglyceride (MCT); isotonic agents such as sodium chloride, potassium chloride, glycerin; buffers such as sodium phosphate, sodium acetate, epsilon-aminocaproic acid Surfactants such as tyloxapol, poloxamer 188, polyoxyethylene sorbitan monooleate, polyoxyl 40 stearate, polyoxyethylene hydrogenated castor oil; stabilizers such as sodium citrate and sodium edetate; benzalkonium chloride , And can be selected and used as needed from preservatives such as parabens.
- the pH of the ophthalmic solution may be within the range acceptable for ophthalmic preparations, but is usually preferably within the range of 4-8.
- the eye ointment or ointment (excluding the eye ointment) can be prepared using, for example, a commonly used base such as white petrolatum or liquid paraffin.
- the injection is necessary, for example, from an isotonic agent such as sodium chloride; a buffering agent such as sodium phosphate; a surfactant such as polyoxyethylene sorbitan monooleate; a thickener such as methylcellulose. It can be selected and used accordingly.
- an isotonic agent such as sodium chloride
- a buffering agent such as sodium phosphate
- a surfactant such as polyoxyethylene sorbitan monooleate
- a thickener such as methylcellulose.
- the preparation for intraocular implant is prepared using, for example, a biodegradable polymer, for example, a biodegradable polymer such as polylactic acid, polyglycolic acid, lactic acid / glycolic acid copolymer, and hydroxypropylcellulose.
- a biodegradable polymer for example, a biodegradable polymer such as polylactic acid, polyglycolic acid, lactic acid / glycolic acid copolymer, and hydroxypropylcellulose.
- the intercalating agent is, for example, a biodegradable polymer, for example, a biodegradable polymer such as hydroxypropylcellulose, hydroxypropylmethylcellulose, carboxyvinyl polymer, polyacrylic acid and the like, pulverized and mixed with the compound, and the powder is compression molded.
- a biodegradable polymer for example, a biodegradable polymer such as hydroxypropylcellulose, hydroxypropylmethylcellulose, carboxyvinyl polymer, polyacrylic acid and the like, pulverized and mixed with the compound, and the powder is compression molded.
- an excipient, a binder, a stabilizer, a pH adjuster and the like can be used as necessary.
- the dose of the present compound or a pharmaceutically acceptable salt thereof can be appropriately changed according to the dosage form, the severity of the patient's symptoms to be administered, age, weight, doctor's judgment, etc. For example, 0.000001 to 1000 mg per day can be administered to an adult once or in several divided doses.
- an ophthalmic solution having an active ingredient concentration of 0.01 to 0.5% (w / v) is divided into once or several times a day, preferably 1 to 2 times a day, more preferably 1 day a day. Can be instilled.
- the present compound or a pharmaceutically acceptable salt thereof is instilled (instilled) as an eye ointment, or administered through the eyelid skin as an ointment (excluding eye ointment), for example, 0.0001 to 1% (for adults) w / w), preferably 0.001-1% (w / w), more preferably 0.01-0.5% (w / w) active ointment once or several times a day Instillation (instillation) or eyelid skin administration can be performed.
- an injection containing 0.000001 to 1000 mg of active ingredient per day is administered to adults in one or several divided doses. can do.
- an intraocular implant preparation containing 0.000001 to 1000 mg of an active ingredient can be implanted into an adult.
- an intercalating agent containing 0.000001 to 1000 mg of an active ingredient can be inserted into an adult.
- sirolimus suspension prepared by suspending in 0.01% (w / v) hydroxypropyl methylcellulose.
- sirolimus what was purchased from LKT Laboratories (catalog number: R0161) was used (hereinafter the same in the examples).
- the eyelid margin of the upper eyelid is equally divided into three fractions of the ear side, the central part, and the nose side, and the meibomian gland opening for each fraction
- the surrounding capillary dilation score was determined, and the sum of the scores of the three fractions was calculated as the score per eye.
- the presence or absence of dilation of the capillaries was determined based on whether or not the capillaries that are normally invisible could be confirmed as a result of the enlargement of the vessel diameter.
- occlusion of the meibomian gland opening part was determined by whether or not the meibomian gland opening part was in a clouded state.
- the group was divided into a physiological saline administration group and a 0.1% sirolimus suspension administration group, and each group was tested with 7 or 8 eyes so as to reduce the variation of the average value of each score.
- Saline solution 50 ⁇ L / eye, twice daily
- 0.1% sirolimus suspension 50 ⁇ L / eye, twice daily
- the periphery of the meibomian gland opening in the upper right eyelid was observed using a slit lamp, the capillary dilation score was determined, and the number of obstructed openings was counted.
- Test results The test results (capillary dilation score and average value of the number of occlusions of the opening) are shown in Table 3.
- Example preparation 0.1% (w / v) sirolimus suspension: prepared by suspending in 0.01% (w / v) hydroxypropyl methylcellulose. 0.1% (w / v) deforolimus suspension: Prepared by suspending in 0.01% (w / v) hyhydroxypropyl methylcellulose. The deforolimus used in this test was produced according to the method described in JP-T-2005-516065.
- the 15 HR-AD feed feeding groups were divided into a physiological saline administration group, a 0.1% sirolimus suspension administration group, and a 0.1% deforolimus suspension administration group (5 mice for each group). From the 29th day after the start, physiological saline (2 ⁇ L / eye, twice daily), 0.1% sirolimus suspension (2 ⁇ L / eye, twice daily), 0.1% (w / v) deforolimus The suspension (2 ⁇ L / eye, twice daily) was instilled for 28 days. On the 42nd and 56th day after the start of feeding, the number of obstructed meibomian gland openings was counted using a slit lamp.
- Test results The test results are shown in Table 4.
- formulation example The pharmaceutical agent of the present invention will be described more specifically with formulation examples, but the present invention is not limited to these formulation examples.
- An eye drop is prepared by adding sirolimus and other components to sterilized purified water and mixing them well.
- the properties of this eye drop are emulsions.
- an ophthalmic solution having a sirolimus concentration of 0.05% (w / v), 0.5% (w / v) or 1% (w / v) can be prepared.
- Formulation Example 2 Eye drops (0.1% (w / v)) 0.1 g of Deforolimus in 100 ml Medium chain triglyceride (MCT) 7.5g Benzalkonium chloride 0.2g Tyloxapol 1.2g Poloxamer 188 1g Glycerin 22.5g Sterilized purified water
- An ophthalmic solution having a sirolimus concentration of 0.05% (w / v), 0.5% (w / v) or 1% (w / v) can be prepared by changing the amount of deforolimus.
- sirolimus to uniformly melted white petrolatum and liquid paraffin, mix them well, and cool slowly to prepare eye ointment or ointment (excluding eye ointment).
- An eye ointment or ointment (eye ointment) having a sirolimus concentration of 0.05% (w / w), 0.1% (w / w) or 0.5% (w / w) by changing the amount of sirolimus can be prepared.
- Defololimus is added to uniformly melted white petrolatum and liquid paraffin, and after thoroughly mixing them, an eye ointment or ointment (excluding eye ointment) is prepared by gradually cooling.
- an ointment or ointment (eye ointment) having a dephorolimus concentration of 0.05% (w / w), 0.1% (w / w) or 0.5% (w / w) can be prepared.
Abstract
Description
R1はヒドロキシ基、メトキシ基、ヒドロキシメトキシ基、エトキシ基、1-ヒドロキエトキシ基、2-ヒドロキシエトキシ基、2-メトキシエトキシ基、2-エトキシエトキシ基、ホルミルオキシ基、カルボキシオキシ基、アセトキシ基、ヒドロキシアセトキシ基、プロピオニルオキシ基、2-ヒドロキシプロピオニルオキシ基、3-ヒドロキシプロピオニルオキシ基、2-メチルプロピオニルオキシ基、2-(ヒドロキシメチル)プロピオニルオキシ基、3-ヒドロキシ-2-(ヒドロキシメチル)プロピオニルオキシ基、2,2-ジメチルプロピオニルオキシ基、2-(ヒドロキシメチル)-2-メチルプロピオニルオキシ基、2,2-ビス(ヒドロキシメチル)プロピオニルオキシ基、メチルホスフィノイルオキシ基、ジメチルホスフィノイルオキシ基または1H-テトラゾール-1-イル基を示し;
R2は水素原子、ヒドロキシ基、メトキシ基、メルカプト基またはメチルチオ基を示し;
R3は水素原子、ヒドロキシ基またはメトキシ基を示し;
波線はR1、R2またはR3と結合した炭素原子がSまたはRのいずれの配置もとりうることを示す]で表される化合物(以下、「本化合物」ともいう)またはその薬学的に許容される塩を有効成分として含有する、マイボーム機能不全の予防および/または治療剤に関する。
R1はヒドロキシ基、メトキシ基、ヒドロキシメトキシ基、エトキシ基、1-ヒドロキエトキシ基、2-ヒドロキシエトキシ基、2-メトキシエトキシ基、2-エトキシエトキシ基、ホルミルオキシ基、カルボキシオキシ基、アセトキシ基、ヒドロキシアセトキシ基、プロピオニルオキシ基、2-ヒドロキシプロピオニルオキシ基、3-ヒドロキシプロピオニルオキシ基、2-メチルプロピオニルオキシ基、2-(ヒドロキシメチル)プロピオニルオキシ基、3-ヒドロキシ-2-(ヒドロキシメチル)プロピオニルオキシ基、2,2-ジメチルプロピオニルオキシ基、2-(ヒドロキシメチル)-2-メチルプロピオニルオキシ基、2,2-ビス(ヒドロキシメチル)プロピオニルオキシ基、メチルホスフィノイルオキシ基、ジメチルホスフィノイルオキシ基または1H-テトラゾール-1-イル基を示し;
R2は水素原子、ヒドロキシ基、メトキシ基、メルカプト基またはメチルチオ基を示し;
R3は水素原子、ヒドロキシ基またはメトキシ基を示し;
波線はR1、R2またはR3と結合した炭素原子がSまたはRのいずれの配置もとりうることを示す]の化合物のまたはその薬学的に許容される塩が、マイボーム腺開口部の閉塞数を減少させることを見出し、そして上記一般式(1)の化合物またはその薬学的に許容される塩が、MGDに対して治療効果を有すること、およびマイボーム腺の閉塞を抑制することを見出し、本発明に至った。
(A2)R2は水素原子、ヒドロキシ基、メトキシ基、メルカプト基、メチルチオ基、フェニル基、2,4,6-トリヒドロキシフェニル基または2,4,6-トリメトキシフェニル基を示し;および
(A3)R3は水素原子、ヒドロキシ基またはメトキシ基を示す。
すなわち、本化合物は、前記一般式(1)で表される化合物において、上記(A1)、(A2)および(A3)に列挙された各基を組み合わせたものである。
(B1)R1がヒドロキシ基、2-ヒドロキシエトキシ基、2-エトキシエトキシ基、2,2-ビス(ヒドロキシメチル)プロピオニルオキシ基、ジメチルホスフィノイルオキシ基または1H-テトラゾール-1-イル基を示し;
(B2)R2が水素原子、ヒドロキシ基、メトキシ基、メチルチオ基または2,4,6-トリメトキシフェニル基を示し;および
(B3)R3が水素原子またはメトキシ基を示す。
すなわち、本化合物の好ましい例は、前記一般式(1)において、上記(B1)、(B2)および(B3)に列挙された各基を組み合わせたものである。
(C1)R1がヒドロキシ基、2-ヒドロキシエトキシ基、2-エトキシエトキシ基、2,2-ビス(ヒドロキシメチル)プロピオニルオキシ基またはジメチルホスフィノイルオキシ基を示し;
(C2)R2がメトキシ基を示し;
(C3)R3がメトキシ基を示し;
すなわち、本化合物のより好ましい例は、前記一般式(1)で表される化合物において、上記(C1)、(C2)および(C3)に列挙された各基を組み合わせたものである。
完全フロイントアジュバンド投与ウサギにおいては、MGDの所見に類似した、マイボーム腺開口部周囲の毛細血管拡張、およびマイボーム腺開口部の閉塞が認められる。本化合物の代表例であるシロリムスが当該毛細血管拡張および閉塞に及ぼす影響を検討した(特開2014-024835を参照)。
0.1%(w/v)シロリムス懸濁液:0.01%(w/v)ヒドロキシプロピルメチルセルロースに懸濁して調製した。なお、シロリムスについては、LKT Laboratories社から購入したもの(カタログ番号:R0161)を使用した(以下、実施例において同じ)。
約2kgの雄性日本白色ウサギの右上眼瞼(3箇所)に、完全フロイントアジュバント10μLをそれぞれ投与した。惹起4日目に、スリットランプを用いて右上眼瞼のマイボーム腺開口部周囲を観察し、毛細血管拡張スコアおよびマイボーム腺開口部閉塞スコアを判定した。
試験結果(毛細血管拡張スコアおよび開口部閉塞数の平均値)を表3に示す。
0.1%シロリムス懸濁液は、マイボーム腺開口部周囲の毛細血管拡張スコアおよび当該開口部の閉塞数を減少させた。すなわち、シロリムスはこれらのマイボーム腺異常を改善させたことから、本化合物またはその薬学的に許容される塩はMGDに対して治療効果を有することが示された。
Hos:HR-1系ヘアレスマウスにHR-AD飼料を給餌すると、MGDの所見に類似した、マイボーム腺開口部の閉塞が認められることが知られている(特願2012-137778)。そこで、本化合物の代表例であるシロリムスおよびデフォロリムスが当該閉塞に及ぼす影響を検討した。
0.1%(w/v)シロリムス懸濁液:0.01%(w/v)ヒドロキシプロピルメチルセルロースに懸濁して調製した。
0.1%(w/v)デフォロリムス懸濁液:0.01%(w/v)ヒヒドロキシプロピルメチルセルロースに懸濁して調製した。なお、本試験に用いたデフォロリムスは、特表2005-516065号公報に記載された方法に従って製造した。
6週齢の雄性Hos:HR-1系ヘアレスマウスを、通常飼料給餌群5匹(CRF-1飼料、オリエンタル酵母工業株式会社製)、HR-AD飼料給餌群15匹(日本農産工業株式会社製)に群分けし、それぞれに通常飼料またはHR-AD飼料を給餌し、自発的に摂取させた。給餌開始後28日目に、スリットランプを用いて、マイボーム腺開口部を観察し、上眼瞼の中央8個のマイボーム腺開口部の中で、閉塞している開口部の数を計測した。なお、マイボーム腺開口部の閉塞の有無は、マイボーム腺開口部が白濁して盛り上がった状態にあるか否かで判定した(特開2014-023526を参照)。
試験結果を表4に示す。
0.1%シロリムス懸濁液、および0.1%デフォロリムス懸濁液は、マイボーム腺開口部の閉塞数を減少させた。すなわち、シロリムスおよびデフォロリムスはマイボーム腺異常を改善させたことから、本化合物またはその薬学的に許容される塩はMGDに対して治療効果を有することが示された。
製剤例を挙げて本発明の薬剤をさらに具体的に説明するが、本発明はこれらの製剤例にのみ限定されるものではない。
100ml中
シロリムス 0.1g
中鎖脂肪酸トリグリセリド(MCT) 7.5g
ベンザルコニウム塩化物 0.2g
チロキサポール 1.2g
ポロキサマー188 1g
グリセリン 22.5g
滅菌精製水 適量
100ml中
デフォロリムス 0.1g
中鎖脂肪酸トリグリセリド(MCT) 7.5g
ベンザルコニウム塩化物 0.2g
チロキサポール 1.2g
ポロキサマー188 1g
グリセリン 22.5g
滅菌精製水 適量
100g中
シロリムス 1g
流動パラフィン 適量
白色ワセリン 適量
100g中
デフォロリムス 1g
流動パラフィン 適量
白色ワセリン 適量
Claims (16)
- 一般式(1):
R1はヒドロキシ基、メトキシ基、ヒドロキシメトキシ基、エトキシ基、1-ヒドロキエトキシ基、2-ヒドロキシエトキシ基、2-メトキシエトキシ基、2-エトキシエトキシ基、ホルミルオキシ基、カルボキシオキシ基、アセトキシ基、ヒドロキシアセトキシ基、プロピオニルオキシ基、2-ヒドロキシプロピオニルオキシ基、3-ヒドロキシプロピオニルオキシ基、2-メチルプロピオニルオキシ基、2-(ヒドロキシメチル)プロピオニルオキシ基、3-ヒドロキシ-2-(ヒドロキシメチル)プロピオニルオキシ基、2,2-ジメチルプロピオニルオキシ基、2-(ヒドロキシメチル)-2-メチルプロピオニルオキシ基、2,2-ビス(ヒドロキシメチル)プロピオニルオキシ基、メチルホスフィノイルオキシ基、ジメチルホスフィノイルオキシ基または1H-テトラゾール-1-イル基を示し;
R2は水素原子、ヒドロキシ基、メトキシ基、メルカプト基またはメチルチオ基を示し;
R3は水素原子、ヒドロキシ基またはメトキシ基を示し;
波線はR1、R2またはR3と結合した炭素原子がSまたはRのいずれの配置もとりうることを示す]
で表される化合物またはその薬学的に許容される塩を有効成分として含有する、マイボーム機能不全の予防および/または治療剤。 - 上記一般式(1)において、R1がヒドロキシ基、2-ヒドロキシエトキシ基、2-エトキシエトキシ基、2,2-ビス(ヒドロキシメチル)プロピオニルオキシ基、ジメチルホスフィノイルオキシ基または1H-テトラゾール-1-イル基を示し;
R2が水素原子、ヒドロキシ基、メトキシ基またはメチルチオ基を示し;
R3が水素原子またはメトキシ基を示す、請求項1記載の予防および/または治療剤。 - 上記一般式(1)において、R1がヒドロキシ基またはジメチルホスフィノイルオキシ基を示し;
R2がメトキシ基を示し;
R3がメトキシ基を示す、請求項1記載の予防および/または治療剤。 - 上記一般式(1)で表される化合物が、シロリムス、デフォロリムス、エベロリムス、テムシロリムス、ゾタロリムス、バイオリムス、ノボリムス、7-エピーラパマイシン、7-チオメチル-ラパマイシン、7-エピ-チオメチル-ラパマイシン、7-デメトキシ-ラパマイシンまたは32-デメトキシ-ラパマイシンである、請求項1記載の予防および/または治療剤。
- 上記一般式(1)で表される化合物がシロリムスである、請求項1記載の予防および/または治療剤。
- 上記一般式(1)で表される化合物がデフォロリムスである、請求項1記載の予防および/または治療剤。
- 前記マイボーム機能不全が、分泌減少型マイボーム機能不全である、請求項1~6のいずれかに記載の予防および/または治療剤。
- 投与形態が点眼投与である、請求項1~7のいずれか一項に記載の予防および/または治療剤。
- 投与剤型が点眼剤または眼軟膏である、請求項1~7のいずれか一項に記載の予防および/または治療剤。
- 投与剤型が点眼剤であり、該点眼剤の性状が、懸濁液またはエマルジョンである、請求項9記載の予防および/または治療剤。
- 投与形態が眼瞼皮膚投与である、請求項1~7のいずれか一項に記載の予防および/または治療剤。
- 投与剤型が軟膏(眼軟膏を除く)である、請求項1~7のいずれか一項に記載の予防および/または治療剤。
- 一般式(1):
R1はヒドロキシ基、メトキシ基、ヒドロキシメトキシ基、エトキシ基、1-ヒドロキエトキシ基、2-ヒドロキシエトキシ基、2-メトキシエトキシ基、2-エトキシエトキシ基、ホルミルオキシ基、カルボキシオキシ基、アセトキシ基、ヒドロキシアセトキシ基、プロピオニルオキシ基、2-ヒドロキシプロピオニルオキシ基、3-ヒドロキシプロピオニルオキシ基、2-メチルプロピオニルオキシ基、2-(ヒドロキシメチル)プロピオニルオキシ基、3-ヒドロキシ-2-(ヒドロキシメチル)プロピオニルオキシ基、2,2-ジメチルプロピオニルオキシ基、2-(ヒドロキシメチル)-2-メチルプロピオニルオキシ基、2,2-ビス(ヒドロキシメチル)プロピオニルオキシ基、メチルホスフィノイルオキシ基、ジメチルホスフィノイルオキシ基または1H-テトラゾール-1-イル基を示し;
R2は水素原子、ヒドロキシ基、メトキシ基、メルカプト基またはメチルチオ基を示し;
R3は水素原子、ヒドロキシ基またはメトキシ基を示し;
波線はR1、R2またはR3と結合した炭素原子がSまたはRのいずれの配置もとりうることを示す]
で表される化合物またはその薬学的に許容される塩を有効成分として含有する、マイボーム腺の閉塞を抑制するための組成物。 - 一般式(1):
R1はヒドロキシ基、メトキシ基、ヒドロキシメトキシ基、エトキシ基、1-ヒドロキエトキシ基、2-ヒドロキシエトキシ基、2-メトキシエトキシ基、2-エトキシエトキシ基、ホルミルオキシ基、カルボキシオキシ基、アセトキシ基、ヒドロキシアセトキシ基、プロピオニルオキシ基、2-ヒドロキシプロピオニルオキシ基、3-ヒドロキシプロピオニルオキシ基、2-メチルプロピオニルオキシ基、2-(ヒドロキシメチル)プロピオニルオキシ基、3-ヒドロキシ-2-(ヒドロキシメチル)プロピオニルオキシ基、2,2-ジメチルプロピオニルオキシ基、2-(ヒドロキシメチル)-2-メチルプロピオニルオキシ基、2,2-ビス(ヒドロキシメチル)プロピオニルオキシ基、メチルホスフィノイルオキシ基、ジメチルホスフィノイルオキシ基または1H-テトラゾール-1-イル基を示し;
R2は水素原子、ヒドロキシ基、メトキシ基、メルカプト基またはメチルチオ基を示し;
R3は水素原子、ヒドロキシ基またはメトキシ基を示し;
波線はR1、R2またはR3と結合した炭素原子がSまたはRのいずれの配置もとりうることを示す]
で表される化合物またはその薬学的に許容される塩を有効成分として含有する、マイボーム腺開口部周辺毛細血管の拡張を抑制するための組成物。 - 上記一般式(1)で表される化合物がシロリムスである、請求項13または14記載の組成物。
- 上記一般式(1)で表される化合物がデフォロリムスである、請求項13または14記載の組成物。
Priority Applications (16)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020207029041A KR102285110B1 (ko) | 2013-03-13 | 2014-03-12 | 마이봄 기능부전의 치료제 |
| EP20164189.1A EP3689348A1 (en) | 2013-03-13 | 2014-03-12 | Therapeutic agent for meibomian gland dysfunction |
| ES14763999T ES2799185T3 (es) | 2013-03-13 | 2014-03-12 | Agente terapéutico para la disfunción meibomiana |
| PL14763999T PL2974728T3 (pl) | 2013-03-13 | 2014-03-12 | Środek terapeutyczny na dysfunkcję gruczołu Meiboma |
| CA2905247A CA2905247C (en) | 2013-03-13 | 2014-03-12 | Therapeutic agent for meibomian gland dysfunction |
| DK14763999.1T DK2974728T3 (da) | 2013-03-13 | 2014-03-12 | Terapeutisk middel til meibomsk dysfunktion |
| US14/775,019 US20160022648A1 (en) | 2013-03-13 | 2014-03-12 | Therapeutic agent for meibomian gland dysfunction |
| CN201480008884.3A CN105073112B (zh) | 2013-03-13 | 2014-03-12 | 睑板腺功能障碍的治疗剂 |
| KR1020157021675A KR102175622B1 (ko) | 2013-03-13 | 2014-03-12 | 마이봄 기능부전의 치료제 |
| EP14763999.1A EP2974728B1 (en) | 2013-03-13 | 2014-03-12 | Therapeutic agent for meibomian dysfunction |
| RU2015143511A RU2709542C2 (ru) | 2013-03-13 | 2014-03-12 | Терапевтическое средство против дисфункции мейбомиевых желез |
| HK16101318.5A HK1213203A1 (zh) | 2013-03-13 | 2016-02-04 | 瞼板腺功能障礙的治療劑 |
| HK16105537.1A HK1217445A1 (zh) | 2013-03-13 | 2016-05-14 | 瞼板腺功能障礙的治療劑 |
| US15/722,257 US20180021314A1 (en) | 2013-03-13 | 2017-10-02 | Therapeutic agent for meibomian dysfunction |
| US16/405,249 US11612590B2 (en) | 2013-03-13 | 2019-05-07 | Therapeutic agent for meibomian dysfunction |
| US18/170,246 US11951098B2 (en) | 2013-03-13 | 2023-02-16 | Therapeutic agent for meibomian dysfunction |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2013050766 | 2013-03-13 | ||
| JP2013-050766 | 2013-03-13 |
Related Child Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US14/775,019 A-371-Of-International US20160022648A1 (en) | 2013-03-13 | 2014-03-12 | Therapeutic agent for meibomian gland dysfunction |
| US15/722,257 Division US20180021314A1 (en) | 2013-03-13 | 2017-10-02 | Therapeutic agent for meibomian dysfunction |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2014142146A1 true WO2014142146A1 (ja) | 2014-09-18 |
Family
ID=51536797
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2014/056416 WO2014142146A1 (ja) | 2013-03-13 | 2014-03-12 | マイボーム機能不全の治療剤 |
Country Status (14)
| Country | Link |
|---|---|
| US (4) | US20160022648A1 (ja) |
| EP (2) | EP3689348A1 (ja) |
| JP (5) | JP6265793B2 (ja) |
| KR (2) | KR102285110B1 (ja) |
| CN (2) | CN105073112B (ja) |
| CA (1) | CA2905247C (ja) |
| DK (1) | DK2974728T3 (ja) |
| ES (1) | ES2799185T3 (ja) |
| HK (3) | HK1213203A1 (ja) |
| PL (1) | PL2974728T3 (ja) |
| PT (1) | PT2974728T (ja) |
| RU (2) | RU2709542C2 (ja) |
| TW (2) | TWI656874B (ja) |
| WO (1) | WO2014142146A1 (ja) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2021132565A1 (ja) | 2019-12-26 | 2021-07-01 | 参天製薬株式会社 | シロリムスまたはその塩を含有する水性懸濁組成物 |
Families Citing this family (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8746883B2 (en) | 2009-04-01 | 2014-06-10 | Tearscience, Inc. | Ocular surface interferometery (OSI) devices and systems for imaging, processing, and/or displaying an ocular tear film |
| WO2013191168A1 (ja) * | 2012-06-19 | 2013-12-27 | 参天製薬株式会社 | 完全フロイントアジュバント投与による眼瞼状態の変化方法 |
| TWI656874B (zh) | 2013-03-13 | 2019-04-21 | 日商參天製藥股份有限公司 | 西羅莫司或其藥學上可容許的鹽之用途 |
| AU2014259619B2 (en) | 2013-05-03 | 2019-08-22 | Tearscience, Inc. | Eyelid illumination systems and methods for imaging meibomian glands for meibomian gland analysis |
| WO2016124601A1 (en) * | 2015-02-02 | 2016-08-11 | Santen Pharmaceutical Co., Ltd | Polyaphrons and palpebral administration thereof |
| CN109999032A (zh) * | 2019-04-30 | 2019-07-12 | 南昌大学 | 雷帕霉素在制备降眼内压的局部滴眼给药药物中的应用 |
| CA3176584A1 (en) * | 2020-03-31 | 2021-10-07 | Santen Pharmaceutical Co., Ltd. | Silver salt-containing ophthalmic aqueous composition filled in resin container |
Citations (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2002522485A (ja) | 1998-08-14 | 2002-07-23 | ジーピーアイ エヌアイエル ホールディングス, インコーポレイテッド | 視覚および記憶の疾患のためのピペコリン酸誘導体 |
| JP2005068101A (ja) * | 2003-08-27 | 2005-03-17 | Nippon Tenganyaku Kenkyusho:Kk | 眼瞼皮膚適用型外用剤 |
| JP2005516065A (ja) | 2002-02-01 | 2005-06-02 | アリアド ジーン セラピューティクス インコーポレイテッド | リン含有化合物およびその用途 |
| JP2007518690A (ja) * | 2003-07-31 | 2007-07-12 | アドバンスト アキュラー システムズ リミテッド | 眼疾患治療 |
| JP2010540682A (ja) | 2007-10-08 | 2010-12-24 | ラックス・バイオサイエンシーズ・インコーポレイテッド | カルシニューリン阻害剤またはmTOR阻害剤を含む眼科用組成物 |
| JP2011516400A (ja) * | 2007-08-16 | 2011-05-26 | ザ スキーペンズ アイ リサーチ インスティチュート インコーポレイテッド | 眼および付属器組織の炎症を処置するための治療組成物 |
| JP2011517659A (ja) * | 2008-01-09 | 2011-06-16 | ザ スキーペンズ アイ リサーチ インスティチュート インコーポレイテッド | 眼の炎症性障害を処置するための治療組成物 |
| JP2012137778A (ja) | 2012-03-19 | 2012-07-19 | Az Electronic Materials Ip Ltd | ケイ素含有微細パターン形成用組成物 |
| WO2012142160A1 (en) * | 2011-04-12 | 2012-10-18 | Rigel Pharmaceuticals, Inc. | Methods for inhibiting allograft rejection |
| JP2012525154A (ja) * | 2009-05-01 | 2012-10-22 | オレゴン ヘルス アンド サイエンス ユニバーシティ | 生体内でヒト肝細胞を増大させる方法 |
| WO2013126599A1 (en) * | 2012-02-21 | 2013-08-29 | Massachusetts Eye & Ear Infirmary | Meibomian gland dysfunction |
| JP2014024835A (ja) | 2012-06-19 | 2014-02-06 | Santen Pharmaceut Co Ltd | 完全フロイントアジュバント投与による眼瞼状態の変化方法 |
| JP2014023526A (ja) | 2012-06-19 | 2014-02-06 | Santen Pharmaceut Co Ltd | ヘアレス動物の眼瞼状態の変化方法 |
Family Cites Families (27)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| ZA935112B (en) * | 1992-07-17 | 1994-02-08 | Smithkline Beecham Corp | Rapamycin derivatives |
| DE19536504C2 (de) * | 1995-09-29 | 1999-09-23 | H Meinert | Verwendung fluorierter Alkane |
| DE19861012A1 (de) * | 1998-03-18 | 1999-09-30 | Pharm Pur Gmbh | Behandlungsmittel für die Ophthalmologie |
| US6864232B1 (en) * | 1998-12-24 | 2005-03-08 | Sucampo Ag | Agent for treating visual cell function disorder |
| US7063857B1 (en) * | 1999-04-30 | 2006-06-20 | Sucampo Ag | Use of macrolide compounds for the treatment of dry eye |
| DE19938668B4 (de) * | 1999-08-14 | 2006-01-26 | Bausch & Lomb Inc. | Tränenersatzmittel |
| US20030018044A1 (en) * | 2000-02-18 | 2003-01-23 | Peyman Gholam A. | Treatment of ocular disease |
| DK1539157T3 (da) * | 2002-09-18 | 2013-10-07 | Univ Pennsylvania | Fremgangsmåde til hæmning af choroidal neovaskularisering |
| PL1646634T3 (pl) * | 2003-07-08 | 2009-04-30 | Novartis Ag | Zastosowanie rapamycyny i pochodnych rapamycyny w leczeniu utraty kości |
| US7585517B2 (en) * | 2003-09-18 | 2009-09-08 | Macusight, Inc. | Transscleral delivery |
| US8541413B2 (en) * | 2004-10-01 | 2013-09-24 | Ramscor, Inc. | Sustained release eye drop formulations |
| US8313763B2 (en) * | 2004-10-04 | 2012-11-20 | Tolmar Therapeutics, Inc. | Sustained delivery formulations of rapamycin compounds |
| EP1655021B1 (en) * | 2004-11-09 | 2008-10-29 | Novagali Pharma SA | Oil-in-water type emulsion with low concentration of cationic agent and positive zeta potential |
| CA2599758A1 (en) | 2005-03-08 | 2006-09-14 | Lifecycle Pharma A/S | Pharmaceutical compositions comprising sirolimus and/or an analogue thereof |
| EP2083834B1 (en) * | 2006-09-13 | 2017-06-21 | Elixir Medical Corporation | Macrocyclic lactone compounds and methods for their use |
| US8088789B2 (en) * | 2006-09-13 | 2012-01-03 | Elixir Medical Corporation | Macrocyclic lactone compounds and methods for their use |
| WO2010044893A1 (en) * | 2008-10-17 | 2010-04-22 | Merck & Co. | Combination therapy |
| ES2645692T3 (es) * | 2008-11-11 | 2017-12-07 | The Board Of Regents,The University Of Texas System | Microcápsulas de rapamicina y su uso para el tratamiento del cáncer |
| MX337849B (es) * | 2009-07-28 | 2016-03-09 | Rigel Pharmaceuticals Inc | Composiciones y metodos para inhibicion de la via jak. |
| EP2335735A1 (en) * | 2009-12-14 | 2011-06-22 | Novaliq GmbH | Pharmaceutical composition for treatment of dry eye syndrome |
| US20120024177A1 (en) * | 2010-07-27 | 2012-02-02 | Alrick Vincent Warner | Method of Printing Fabric-Inspired Designs On Absorbent Articles |
| JP5713179B2 (ja) | 2010-12-28 | 2015-05-07 | スタンレー電気株式会社 | 自動二輪車用プロジェクタ型ヘッドランプ |
| US20130102572A1 (en) | 2011-04-12 | 2013-04-25 | Dow Pharmaceutical Sciences | Methods of treating skin conditions exhibiting telangiectasia |
| WO2013126602A1 (en) | 2012-02-21 | 2013-08-29 | Massachusetts Eye & Ear Infirmary | Inflammatory eye disorders |
| US8765725B2 (en) | 2012-05-08 | 2014-07-01 | Aciex Therapeutics, Inc. | Preparations of hydrophobic therapeutic agents, methods of manufacture and use thereof |
| CA3142049C (en) * | 2012-09-12 | 2023-08-29 | Novaliq Gmbh | Semifluorinated alkane compositions |
| TWI656874B (zh) | 2013-03-13 | 2019-04-21 | 日商參天製藥股份有限公司 | 西羅莫司或其藥學上可容許的鹽之用途 |
-
2014
- 2014-03-12 TW TW107109966A patent/TWI656874B/zh active
- 2014-03-12 PL PL14763999T patent/PL2974728T3/pl unknown
- 2014-03-12 US US14/775,019 patent/US20160022648A1/en not_active Abandoned
- 2014-03-12 DK DK14763999.1T patent/DK2974728T3/da active
- 2014-03-12 PT PT147639991T patent/PT2974728T/pt unknown
- 2014-03-12 EP EP20164189.1A patent/EP3689348A1/en active Pending
- 2014-03-12 RU RU2015143511A patent/RU2709542C2/ru active
- 2014-03-12 EP EP14763999.1A patent/EP2974728B1/en active Active
- 2014-03-12 TW TW103108641A patent/TWI623313B/zh active
- 2014-03-12 RU RU2019139886A patent/RU2019139886A/ru unknown
- 2014-03-12 WO PCT/JP2014/056416 patent/WO2014142146A1/ja active Application Filing
- 2014-03-12 JP JP2014048477A patent/JP6265793B2/ja active Active
- 2014-03-12 CN CN201480008884.3A patent/CN105073112B/zh active Active
- 2014-03-12 CA CA2905247A patent/CA2905247C/en active Active
- 2014-03-12 KR KR1020207029041A patent/KR102285110B1/ko active IP Right Grant
- 2014-03-12 KR KR1020157021675A patent/KR102175622B1/ko active IP Right Grant
- 2014-03-12 CN CN201711069895.4A patent/CN107823202B/zh active Active
- 2014-03-12 ES ES14763999T patent/ES2799185T3/es active Active
-
2016
- 2016-02-04 HK HK16101318.5A patent/HK1213203A1/zh unknown
- 2016-05-14 HK HK16105537.1A patent/HK1217445A1/zh unknown
- 2016-05-14 HK HK18105546.8A patent/HK1245680A1/zh unknown
-
2017
- 2017-10-02 US US15/722,257 patent/US20180021314A1/en not_active Abandoned
- 2017-12-19 JP JP2017242430A patent/JP6495427B2/ja active Active
-
2019
- 2019-03-06 JP JP2019040166A patent/JP2019108383A/ja active Pending
- 2019-05-07 US US16/405,249 patent/US11612590B2/en active Active
-
2020
- 2020-09-08 JP JP2020150230A patent/JP7082162B2/ja active Active
-
2022
- 2022-05-26 JP JP2022085648A patent/JP2022116182A/ja active Pending
-
2023
- 2023-02-16 US US18/170,246 patent/US11951098B2/en active Active
Patent Citations (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2002522485A (ja) | 1998-08-14 | 2002-07-23 | ジーピーアイ エヌアイエル ホールディングス, インコーポレイテッド | 視覚および記憶の疾患のためのピペコリン酸誘導体 |
| JP2005516065A (ja) | 2002-02-01 | 2005-06-02 | アリアド ジーン セラピューティクス インコーポレイテッド | リン含有化合物およびその用途 |
| JP2007518690A (ja) * | 2003-07-31 | 2007-07-12 | アドバンスト アキュラー システムズ リミテッド | 眼疾患治療 |
| JP2005068101A (ja) * | 2003-08-27 | 2005-03-17 | Nippon Tenganyaku Kenkyusho:Kk | 眼瞼皮膚適用型外用剤 |
| JP2011516400A (ja) * | 2007-08-16 | 2011-05-26 | ザ スキーペンズ アイ リサーチ インスティチュート インコーポレイテッド | 眼および付属器組織の炎症を処置するための治療組成物 |
| JP2010540682A (ja) | 2007-10-08 | 2010-12-24 | ラックス・バイオサイエンシーズ・インコーポレイテッド | カルシニューリン阻害剤またはmTOR阻害剤を含む眼科用組成物 |
| JP2011517659A (ja) * | 2008-01-09 | 2011-06-16 | ザ スキーペンズ アイ リサーチ インスティチュート インコーポレイテッド | 眼の炎症性障害を処置するための治療組成物 |
| JP2012525154A (ja) * | 2009-05-01 | 2012-10-22 | オレゴン ヘルス アンド サイエンス ユニバーシティ | 生体内でヒト肝細胞を増大させる方法 |
| WO2012142160A1 (en) * | 2011-04-12 | 2012-10-18 | Rigel Pharmaceuticals, Inc. | Methods for inhibiting allograft rejection |
| WO2013126599A1 (en) * | 2012-02-21 | 2013-08-29 | Massachusetts Eye & Ear Infirmary | Meibomian gland dysfunction |
| JP2012137778A (ja) | 2012-03-19 | 2012-07-19 | Az Electronic Materials Ip Ltd | ケイ素含有微細パターン形成用組成物 |
| JP2014024835A (ja) | 2012-06-19 | 2014-02-06 | Santen Pharmaceut Co Ltd | 完全フロイントアジュバント投与による眼瞼状態の変化方法 |
| JP2014023526A (ja) | 2012-06-19 | 2014-02-06 | Santen Pharmaceut Co Ltd | ヘアレス動物の眼瞼状態の変化方法 |
Non-Patent Citations (5)
| Title |
|---|
| INVESTIGATIVE OPHTHALMOLOGY & VISUAL SCIENCE, vol. 52, no. 4, 2011, pages 1930 - 1937 |
| JOURNAL OF THE EYE, vol. 27, no. 5, 2010, pages 627 - 631 |
| KHANDELWAL P ET AL.: "Androgen regulation of gene expression in human meibomian gland and conjunctival epithelial cells", MOL VIS, vol. 18, 27 April 2012 (2012-04-27), pages 1055 - 1067, XP055281056 * |
| MCKOWN RL ET AL.: "Lacritin and other new proteins of the lacrimal functional unit", EXP EYE RES, vol. 88, no. 5, 1 May 2009 (2009-05-01), pages 848 - 858, XP026024034, DOI: 10.1016/J.EXER.2008.09.002 * |
| WANG M ET AL.: "Effect assessment of subconjunctival injection of rapamycin-loaded microspheres in non-obese diabetic mice with dry eye caused by Sjogren's syndrome", GUOJI YANKE ZAZHI(INT EYE SCI, vol. 13, no. 5, May 2013 (2013-05-01), pages 861 - 864, XP008180980 * |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2021132565A1 (ja) | 2019-12-26 | 2021-07-01 | 参天製薬株式会社 | シロリムスまたはその塩を含有する水性懸濁組成物 |
| KR20220122612A (ko) | 2019-12-26 | 2022-09-02 | 산텐 세이야꾸 가부시키가이샤 | 시롤리무스 또는 그 염을 함유하는 수성 현탁 조성물 |
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6495427B2 (ja) | マイボーム機能不全の治療剤 | |
| RU2406499C2 (ru) | Профилактическое или терапевтическое средство для лечения кератоконъюнктивитных нарушений | |
| JP6509244B2 (ja) | 水晶体硬化抑制剤 | |
| JP7016880B2 (ja) | チオトロピウムを有効成分として含有する近視予防、近視治療および/または近視進行抑制剤 | |
| JP2011144111A (ja) | 軸性近視の予防または治療剤 | |
| US9579309B2 (en) | Prophylactic or therapeutic agent for posterior ocular disease containing tetrahydropyranylaminocyclopentylcarbonyltetrahydropyridopyridine derivative as effective ingredient | |
| JP2010150243A (ja) | ピリジン−3−カルバルデヒドo−(ピペリジン−1−イル−プロピル)−オキシム誘導体を有効成分として含有する網脈絡膜変性疾患の治療剤 | |
| TW202227042A (zh) | 雙重鎮痛/消炎組合物、組合及其使用方法 | |
| JP2009079041A (ja) | リチウム塩を有効成分として含有する後眼部疾患の治療又は予防剤 | |
| KR20160143846A (ko) | 이명 환자의 치료용 약제 | |
| JP2014005276A (ja) | ペプチド誘導体を含有する緑内障の治療剤 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 201480008884.3 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 14763999 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 20157021675 Country of ref document: KR Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 2905247 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 14775019 Country of ref document: US |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2014763999 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 2015143511 Country of ref document: RU Kind code of ref document: A |