WO2013024960A1 - 멀꿀 추출물을 포함하는 의약 조성물 - Google Patents
멀꿀 추출물을 포함하는 의약 조성물 Download PDFInfo
- Publication number
- WO2013024960A1 WO2013024960A1 PCT/KR2012/003867 KR2012003867W WO2013024960A1 WO 2013024960 A1 WO2013024960 A1 WO 2013024960A1 KR 2012003867 W KR2012003867 W KR 2012003867W WO 2013024960 A1 WO2013024960 A1 WO 2013024960A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- honey
- extract
- composition
- inflammatory
- antipyretic
- Prior art date
Links
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K36/00—Medicinal preparations of undetermined constitution containing material from algae, lichens, fungi or plants, or derivatives thereof, e.g. traditional herbal medicines
- A61K36/18—Magnoliophyta (angiosperms)
- A61K36/185—Magnoliopsida (dicotyledons)
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/96—Cosmetics or similar toiletry preparations characterised by the composition containing materials, or derivatives thereof of undetermined constitution
- A61K8/97—Cosmetics or similar toiletry preparations characterised by the composition containing materials, or derivatives thereof of undetermined constitution from algae, fungi, lichens or plants; from derivatives thereof
- A61K8/9783—Angiosperms [Magnoliophyta]
- A61K8/9789—Magnoliopsida [dicotyledons]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/02—Stomatological preparations, e.g. drugs for caries, aphtae, periodontitis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/04—Drugs for disorders of the alimentary tract or the digestive system for ulcers, gastritis or reflux esophagitis, e.g. antacids, inhibitors of acid secretion, mucosal protectants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/16—Drugs for disorders of the alimentary tract or the digestive system for liver or gallbladder disorders, e.g. hepatoprotective agents, cholagogues, litholytics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/18—Drugs for disorders of the alimentary tract or the digestive system for pancreatic disorders, e.g. pancreatic enzymes
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P11/00—Drugs for disorders of the respiratory system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P11/00—Drugs for disorders of the respiratory system
- A61P11/08—Bronchodilators
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P13/00—Drugs for disorders of the urinary system
- A61P13/12—Drugs for disorders of the urinary system of the kidneys
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
- A61P17/02—Drugs for dermatological disorders for treating wounds, ulcers, burns, scars, keloids, or the like
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P21/00—Drugs for disorders of the muscular or neuromuscular system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P27/00—Drugs for disorders of the senses
- A61P27/02—Ophthalmic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
- A61P29/02—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID] without antiinflammatory effect
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/04—Antibacterial agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/02—Immunomodulators
- A61P37/06—Immunosuppressants, e.g. drugs for graft rejection
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/08—Antiallergic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P39/00—General protective or antinoxious agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P5/00—Drugs for disorders of the endocrine system
- A61P5/14—Drugs for disorders of the endocrine system of the thyroid hormones, e.g. T3, T4
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P7/00—Drugs for disorders of the blood or the extracellular fluid
- A61P7/10—Antioedematous agents; Diuretics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/10—Drugs for disorders of the cardiovascular system for treating ischaemic or atherosclerotic diseases, e.g. antianginal drugs, coronary vasodilators, drugs for myocardial infarction, retinopathy, cerebrovascula insufficiency, renal arteriosclerosis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q19/00—Preparations for care of the skin
Definitions
- antipyretic drug is a drug that acts to lower the body temperature in the fever, and generally called analgesic because it also exhibits analgesic action.
- Another object of the present invention is to provide a composition for antipyretic with an active ingredient of a plant extract, which is less likely to cause problems related to side effects, in order to meet the above demands.
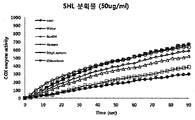
- the honey extract has an excellent anti-inflammatory effect. More specifically, the extract of honey can effectively inhibit NO secretion, inhibit the expression of iNOS associated with NO production, and promote COX (inflammation) in response to the biosynthesis of prostaglandin present in vivo. It was confirmed that it can inhibit cyclooxygenase) enzyme activity. In addition, it was confirmed that the ethyl acetate fraction of the various solvent fractions of the honey leaf extract can effectively inhibit NO production and COX enzyme activity as well as other fractions in the range where toxicity is not a problem.
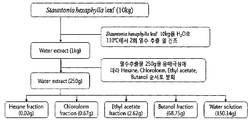
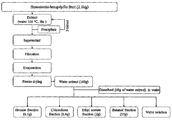
- the extract extracted with the solvent may be further subjected to the fractionation process with one or more solvents selected from the group consisting of hexane, chloroform, ethyl acetate, methylene chloride, ethyl ether, acetone, butanol, water and mixtures thereof.
- solvents selected from the group consisting of hexane, chloroform, ethyl acetate, methylene chloride, ethyl ether, acetone, butanol, water and mixtures thereof.
- Two or more solvents may be used in the fractionation, and each solvent extract may be prepared using sequentially or mixed according to the polarity of the solvent.
- the anti-inflammatory composition can be used for the purpose of treating, preventing or ameliorating the inflammatory disease.
- the honey extract may be preferably a honey leaf extract.
- the antipyretic agent of the present invention may further include a compound or a plant extract having a known antipyretic effect in addition to the active ingredient, and 0.1 parts by weight to 99.9 parts by weight or 0.5 parts by weight to 20 parts by weight based on 100 parts by weight of the active ingredient, respectively. It may be included in parts by weight.
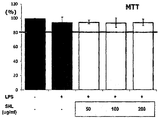
- the cultured cells were aliquoted into 48 well-plates at a density of 50,000 cells / well and further cultured for 24 hours. After 24 hours, the LPS-treated control group without any treatment and LPS and the honey leaf extract and fraction of Example 1 were prepared at various concentrations using DMSO, which was found to have little effect on cell survival. The honey leaf extract and fractions were divided into experimental groups, and further cultured for 24 hours, the culture solution was removed, and the number of living cells was measured by MTT assay. The MTT analysis was performed in the following manner.
- the macrophage (Macrophage primary cell) obtained in Example 4 was adjusted to 1 X 10 5 cells / ml using DMEM medium and then inoculated in a 24 well plate, 18 hours ago in a 5% CO 2 thermostat Incubated. After the incubation, the honey leaf hydrothermal extracts were treated by concentration (0.1 ⁇ g / ml, 1 ⁇ g / ml, 10 ⁇ g / ml, 100 ⁇ g / ml and 200 ⁇ g / ml), and after 1 hour of incubation, LPS (1 ⁇ g / ml). Ml) was treated and cultured under the same conditions as the pre-culture.
- Example 9-1 SD male rat of 5 weeks old (Samtako, Korea) ) was used.
Abstract
Description
| 확인대상 mRNA | primer sequence | Annealing Tm(℃) | |
| iNOS | sense | CAGAGGACCCAGAGACAAG | 50.8 |
| anti-sense | ACCTGATGTTGCCATTGTTG | ||
| 확인대상 mRNA | Primer sequence | Annealing Tm(℃) | |
| TNF-α | sense | GGCAGGTCTACTTTGGAGTCATTGC | 62.2 |
| anti-sense | ACATTCGAGGCTCCAGTGAATTCGG | ||
| IFN-γ | sense | GCGGCTGACTGAACTCAGATTGTAG | 50 |
| anti-sense | GTCACAGTTTTCAGCTGTATAGGG | ||
| IL-1β | sense | TGCAGAGTTCCTACATGGTCAACC | 55 |
| anti-sense | GTGCTGCCTAATGTCCCCTTGAATC | ||
| Normal | LPS | SHL 200 | SHL 200(1h) | Dexamethasone | APAP | |
| 0h | 37.2 | 37.2 | 37.2 | 37.2 | 37.2 | 37.2 |
| 1h | 37.55±0.21 | 38.2±0.68 | 36.65±0.69 | 38.23±0.41 | 37.3±0.52 | 36.58±0.67 |
| 4h | 37.65±0.07 | 37.85±0.33 | 37.20±0.45 | 36.95±0.54 | 37.6±0.12 | 37.53±0.59 |
| 8h | 37.55±0.07 | 37.6±0.61 | 37.7±0.14 | 37.73±0.22 | 37.73±0.13 | 37.78±0.13 |
Claims (16)
- 멀꿀(Stauntonia Hexaphylla) 추출물을 유효성분으로 포함하는 항염 조성물.
- 제1항에 있어서,상기 멀꿀 추출물은 멀꿀 잎 추출물 또는 멀꿀 열매 추출물인 항염 조성물.
- 제1항에 있어서,상기 멀꿀 추출물은 상기 멀꿀 잎 또는 멀꿀 열매를 물, 탄소수 1 내지 5의 알코올 및 이의 혼합물로 이루어진 군 중에서 선택된 1종 이상을 추출용매로 추출한 것인 항염 조성물.
- 제3항에 있어서,상기 멀꿀 추출물은 멀꿀 잎 열수추출물인 항염조성물.
- 제4항에 있어서,상기 멀꿀 잎 열수 추출물은 멀꿀 잎 열수추출물에 에틸아세테이트 또는 클로로포름을 분획용매로 분획한 분획물인 항염조성물.
- 멀꿀(Stauntonia Hexaphylla) 추출물을 유효성분으로 포함하는 염증질환 치료 및 예방용 약학 조성물.
- 제6항에 있어서,상기 멀꿀 추출물은 멀꿀 잎 열수추출물에 에틸아세테이트 또는 클로로포름을 분획용매로 분획한 분획물인 염증질환 치료 및 예방용 약학 조성물.
- 제6항에 있어서,상기 염증 질환은 피부염, 피부근염(dermatomyositis), 다발성 근염(polymyositis), 알레르기, 전신성 홍반성낭창, 천포창, 아프타구내염, 망막염, 위염, 간염, 기관지염, 식도염, 장염, 췌장염, 대장염, 신장염, 욕창, 루푸스, 만성 갑상선염, 다발성 경화증, 패혈증(sepsis), 방사선 손상, 장기이식의 거부반응, 전신부종 및 국소부종으로 이루어진 군 중에서 선택된 어느 하나인 염증질환 치료 및 예방용 약학 조성물.
- 멀꿀(Stauntonia Hexaphylla) 추출물을 유효성분으로 포함하는 염증 완화 또는 완화용 화장료 조성물.
- 제9항에 있어서,상기 멀꿀 추출물은 멀꿀 잎 열수추출물에 에틸아세테이트 또는 클로로포름을 분획용매로 분획한 분획물인 염증 완화 또는 완화용 화장료 조성물.
- 멀꿀(Stauntonia Hexaphylla) 추출물을 유효성분으로 포함하는 해열용 조성물.
- 제11항에 있어서,상기 멀꿀 추출물은 멀꿀 잎을 물, 탄소수 1 내지 5의 알코올 및 이의 혼합물로 이루어진 군 중에서 선택된 1종 이상을 추출용매로 추출한 것인 해열용 조성물.
- 제12항에 있어서,상기 멀꿀 추출물은 멀꿀 잎 열수추출물에 클로로포름, 부탄올 또는 에틸아세테이트를 분획용매로 분획한 분획물인 해열용 조성물.
- 제13항에 있어서,상기 분획용매는 에틸아세테이트 또는 클로로포름인 해열용 조성물.
- 제14항에 있어서,상기 분획용매는 에틸아세테이트인 해열용 조성물.
- 제11항에 있어서,상기 해열용 조성물은 해열제 또는 해열진통제인 해열용 조성물.
Priority Applications (11)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| AU2012295779A AU2012295779A1 (en) | 2011-08-18 | 2012-05-16 | Medical composition containing stauntonia hexaphylla extract |
| PL16176916T PL3097920T3 (pl) | 2011-08-18 | 2012-05-16 | Kompozycja medyczna zawierająca ekstrakt stauntonia hexaphylla |
| CN201280045043.0A CN103826651A (zh) | 2011-08-18 | 2012-05-16 | 含野木瓜提取物的医药组合物 |
| ES12824322.7T ES2620022T3 (es) | 2011-08-18 | 2012-05-16 | Composición química que contiene extracto de Stauntonia Hexaphylla |
| CA2845625A CA2845625C (en) | 2011-08-18 | 2012-05-16 | Medical composition comprising stauntonia hexaphylla extract |
| JP2014525911A JP6049721B2 (ja) | 2011-08-18 | 2012-05-16 | ムベ抽出物を含む医薬組成物 |
| US14/239,521 US9370542B2 (en) | 2011-08-18 | 2012-05-16 | Medical composition containing Stauntonia hexaphylla extract |
| EP16176916.1A EP3097920B1 (en) | 2011-08-18 | 2012-05-16 | Medical composition comprising stauntonia hexaphylla extract |
| EP12824322.7A EP2774617B1 (en) | 2011-08-18 | 2012-05-16 | Medical composition containing stauntonia hexaphylla extract |
| US15/012,553 US20160193265A1 (en) | 2011-08-18 | 2016-02-01 | Medical composition containing stauntonia hexaphylla extract |
| AU2016201841A AU2016201841B2 (en) | 2011-08-18 | 2016-03-23 | Medical Composition Containing Stauntonia Hexaphylla Extract |
Applications Claiming Priority (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020110082023A KR101167589B1 (ko) | 2011-08-18 | 2011-08-18 | 멀꿀 열매 추출물을 포함하는 항염증제 |
| KR10-2011-0082023 | 2011-08-18 | ||
| KR10-2012-0038977 | 2012-04-16 | ||
| KR1020120038977A KR101221617B1 (ko) | 2012-04-16 | 2012-04-16 | 멀꿀 잎 추출물을 포함하는 항염증제 |
| KR10-2012-0050532 | 2012-05-11 | ||
| KR1020120050532A KR101243115B1 (ko) | 2012-04-17 | 2012-05-11 | 멀꿀 잎 추출물을 포함하는 해열제 |
Related Child Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US14/239,521 A-371-Of-International US9370542B2 (en) | 2011-08-18 | 2012-05-16 | Medical composition containing Stauntonia hexaphylla extract |
| US15/012,553 Continuation US20160193265A1 (en) | 2011-08-18 | 2016-02-01 | Medical composition containing stauntonia hexaphylla extract |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2013024960A1 true WO2013024960A1 (ko) | 2013-02-21 |
Family
ID=50185141
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/KR2012/003867 WO2013024960A1 (ko) | 2011-08-18 | 2012-05-16 | 멀꿀 추출물을 포함하는 의약 조성물 |
Country Status (9)
| Country | Link |
|---|---|
| US (2) | US9370542B2 (ko) |
| EP (2) | EP3097920B1 (ko) |
| JP (2) | JP6049721B2 (ko) |
| CN (2) | CN103826651A (ko) |
| AU (2) | AU2012295779A1 (ko) |
| CA (2) | CA2999174C (ko) |
| ES (2) | ES2752249T3 (ko) |
| PL (2) | PL3097920T3 (ko) |
| WO (1) | WO2013024960A1 (ko) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2016520059A (ja) * | 2013-06-30 | 2016-07-11 | ジョンナム バイオインダストリー ファウンデーション | ムベ葉抽出物を有効成分として含む骨組織生成促進用薬学組成物 |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US10137162B1 (en) * | 2018-01-26 | 2018-11-27 | King Saud University | Gastroprotective extracts of Sonchus oleraceus L. |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2001226249A (ja) * | 2000-02-17 | 2001-08-21 | Ichimaru Pharcos Co Ltd | 植物水蒸気蒸留水含有化粧料組成物 |
| KR20090048848A (ko) * | 2007-11-12 | 2009-05-15 | 이스트힐(주) | 오메가오일과 멀꿀나무 추출물을 포함하는 색조 화장료용 복합 분체 |
| KR20110068923A (ko) * | 2009-12-15 | 2011-06-22 | 재단법인 경기바이오센터 | 항히스타민제 조성물 |
Family Cites Families (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH01242596A (ja) * | 1988-03-17 | 1989-09-27 | Zhongguo Yixuekexueyuan Yaowo Yanjiusuo | リグナン化合物 |
| JPH01290694A (ja) * | 1988-05-07 | 1989-11-22 | Zhongguo Yixuekexueyuan Yaowo Yanjiusuo | リグナン化合物 |
| CN1562275A (zh) * | 2004-03-22 | 2005-01-12 | 广州安健实业发展有限公司 | 野木瓜总甙及其注射剂 |
| CN1742816A (zh) * | 2005-10-07 | 2006-03-08 | 张益群 | 一种五角枫根外用药 |
| CN100350932C (zh) * | 2005-12-27 | 2007-11-28 | 王赛 | 非手术治疗鼻炎、咽喉炎的药物 |
| CN101024065A (zh) * | 2007-04-09 | 2007-08-29 | 林水文 | 一种活络油的药物组合物及其制备方法 |
| CN101283784B (zh) * | 2007-09-27 | 2012-01-11 | 贵州大学 | 野木瓜保健固体饮料及其制备方法 |
| CN101879196A (zh) * | 2009-05-08 | 2010-11-10 | 何国增 | 一种中药野木瓜注射液的制备方法 |
| CN102132914A (zh) * | 2010-01-27 | 2011-07-27 | 遵义光勋绿色产业有限公司 | 一种野木瓜饮料的制备方法 |
| CN101926488B (zh) | 2010-04-06 | 2012-05-23 | 贵州大学 | 一种野木瓜果醋饮料的制备方法 |
-
2012
- 2012-05-16 EP EP16176916.1A patent/EP3097920B1/en active Active
- 2012-05-16 CN CN201280045043.0A patent/CN103826651A/zh active Pending
- 2012-05-16 WO PCT/KR2012/003867 patent/WO2013024960A1/ko active Application Filing
- 2012-05-16 EP EP12824322.7A patent/EP2774617B1/en active Active
- 2012-05-16 JP JP2014525911A patent/JP6049721B2/ja active Active
- 2012-05-16 CA CA2999174A patent/CA2999174C/en active Active
- 2012-05-16 AU AU2012295779A patent/AU2012295779A1/en not_active Abandoned
- 2012-05-16 CN CN201810231572.9A patent/CN108392501A/zh active Pending
- 2012-05-16 CA CA2845625A patent/CA2845625C/en active Active
- 2012-05-16 PL PL16176916T patent/PL3097920T3/pl unknown
- 2012-05-16 ES ES16176916T patent/ES2752249T3/es active Active
- 2012-05-16 ES ES12824322.7T patent/ES2620022T3/es active Active
- 2012-05-16 US US14/239,521 patent/US9370542B2/en active Active
- 2012-05-16 PL PL12824322T patent/PL2774617T3/pl unknown
-
2016
- 2016-02-01 US US15/012,553 patent/US20160193265A1/en not_active Abandoned
- 2016-03-23 AU AU2016201841A patent/AU2016201841B2/en active Active
- 2016-06-13 JP JP2016116887A patent/JP6258409B2/ja active Active
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2001226249A (ja) * | 2000-02-17 | 2001-08-21 | Ichimaru Pharcos Co Ltd | 植物水蒸気蒸留水含有化粧料組成物 |
| KR20090048848A (ko) * | 2007-11-12 | 2009-05-15 | 이스트힐(주) | 오메가오일과 멀꿀나무 추출물을 포함하는 색조 화장료용 복합 분체 |
| KR20110068923A (ko) * | 2009-12-15 | 2011-06-22 | 재단법인 경기바이오센터 | 항히스타민제 조성물 |
Non-Patent Citations (4)
| Title |
|---|
| "Remington's Pharmaceutical Science", MACK PUBLISHING COMPANY |
| KIM ET AL.: "Antioxident activity and cell toxicity of pressurised liquid extracts from 20 selected plant species in Jeju, Korea", FOOD CHEMISTRY, vol. 122, no. 3, 2010, pages 546 - 552, XP027039536 * |
| See also references of EP2774617A4 * |
| VILELA FC: "Anti-inflammatory and antipyretic effects of Sonchus oleraceus in rats", J ETHNOPHARMACOL., vol. 127, no. 3, 2010, pages 737 - 41, XP026884192, DOI: doi:10.1016/j.jep.2009.11.030 |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2016520059A (ja) * | 2013-06-30 | 2016-07-11 | ジョンナム バイオインダストリー ファウンデーション | ムベ葉抽出物を有効成分として含む骨組織生成促進用薬学組成物 |
Also Published As
| Publication number | Publication date |
|---|---|
| EP2774617B1 (en) | 2016-12-21 |
| JP6049721B2 (ja) | 2016-12-21 |
| PL3097920T3 (pl) | 2020-03-31 |
| EP2774617A1 (en) | 2014-09-10 |
| CA2999174A1 (en) | 2013-02-21 |
| ES2620022T3 (es) | 2017-06-27 |
| ES2752249T3 (es) | 2020-04-03 |
| EP3097920B1 (en) | 2019-07-31 |
| CA2999174C (en) | 2020-07-14 |
| PL2774617T3 (pl) | 2017-08-31 |
| US20140205691A1 (en) | 2014-07-24 |
| CA2845625C (en) | 2018-05-15 |
| CA2845625A1 (en) | 2013-02-21 |
| AU2016201841B2 (en) | 2017-11-02 |
| CN108392501A (zh) | 2018-08-14 |
| AU2012295779A1 (en) | 2014-03-06 |
| EP3097920A1 (en) | 2016-11-30 |
| EP2774617A4 (en) | 2015-02-18 |
| JP2014524436A (ja) | 2014-09-22 |
| JP2016164202A (ja) | 2016-09-08 |
| JP6258409B2 (ja) | 2018-01-10 |
| AU2016201841A1 (en) | 2016-05-19 |
| US9370542B2 (en) | 2016-06-21 |
| US20160193265A1 (en) | 2016-07-07 |
| CN103826651A (zh) | 2014-05-28 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| KR101221617B1 (ko) | 멀꿀 잎 추출물을 포함하는 항염증제 | |
| WO2016060525A1 (ko) | 쥐꼬리망초 속 식물의 추출물 또는 분획물을 포함하는 조성물 | |
| WO2020218781A1 (ko) | 불멸화 줄기세포 유래 엑소좀 풍부 배양액 및 장미꽃봉오리 추출물을 유효성분으로 포함하는 기능성 조성물 | |
| WO2016032249A1 (ko) | 모새나무 추출물 또는 이의 분획물을 유효성분으로 포함하는 신경염증 또는 퇴행성 뇌신경 질환의 예방 또는 치료용 약학적 조성물 | |
| WO2020076101A1 (ko) | 시지지움 포르모슘 추출물을 유효성분으로 포함하는 피부 미백, 항균 또는 항아토피 조성물 | |
| WO2022260454A1 (ko) | 장수만리화 추출물을 포함하는 아토피 피부염 치료용 또는 피부 장벽 강화용 또는 노화 방지용 조성물 | |
| WO2021080297A1 (ko) | 달맞이꽃 추출물을 유효성분으로 포함하는 비만 또는 비만으로부터 유도된 대사증후군의 예방 또는 치료용 조성물 | |
| WO2020256464A1 (ko) | 항염 활성을 갖는 아피오스 아메리카나 괴경 추출물의 분획물의 알코올성 위염 예방 또는 치료제로서의 용도 및 이의 제조방법 | |
| WO2013024960A1 (ko) | 멀꿀 추출물을 포함하는 의약 조성물 | |
| KR101167589B1 (ko) | 멀꿀 열매 추출물을 포함하는 항염증제 | |
| WO2016093515A1 (ko) | 장수 유전자 활성화용 조성물 | |
| WO2019203515A1 (ko) | 긴병꽃풀 추출물을 함유하는 벌레물림 치료제 | |
| WO2015037855A1 (en) | A composition comprising an extract of combined herbs consisting of acanthopanax koreanum nakai and crinum asiaticum var. japonicum showing preventing activity of baldness and stimulating activity of hair growth | |
| WO2022092523A1 (ko) | 중대가리풀 유래의 추출물을 포함하는 항염, 자가면역 질환 및 비알코올성 지방간 질환 예방 및 치료용 조성물 | |
| WO2018106009A1 (ko) | 단풍잎 추출물 또는 이의 분획물을 포함하는 염증성 안구질환 예방 또는 치료용 조성물 | |
| WO2016122091A1 (ko) | 심비디움(cymbidium) 추출물을 유효성분으로 함유하는 알레르기 질환 또는 접촉성 피부염 예방 및 치료용 약학조성물 | |
| WO2017142368A2 (ko) | 알레르기성 또는 염증성 피부질환의 예방 또는 치료용 조성물 | |
| WO2020204479A1 (ko) | 포도잎 추출물 및 병풀 추출물을 포함하는 정맥순환 장애 개선용 조성물 | |
| WO2014185610A1 (ko) | 밀기울 추출물을 유효성분으로 포함하는 탈모 방지 또는 육모 촉진 조성물 | |
| WO2015152653A1 (ko) | 알파인 웜우드 추출물을 포함하는 조성물 | |
| WO2018155933A1 (ko) | 아쿠아포린-3의 발현을 증가시키는 6,7-디메톡시-2,2-디메틸-2h-크로멘 및 이의 용도 | |
| WO2012096463A2 (ko) | 복합 생약 추출물을 유효성분으로 함유하는 알러지성 또는 비알러지성 피부 질환의 예방 및 개선용 조성물 및 이의 용도 | |
| WO2020032297A1 (ko) | 백반증 증상 개선용 조성물 및 이의 제조방법 | |
| WO2016032250A1 (ko) | 채송화 추출물 또는 이의 분획물을 유효성분으로 포함하는 신경염증 또는 퇴행성 뇌신경 질환의 예방 또는 치료용 약학적 조성물 | |
| WO2023182757A1 (ko) | 뇌졸중의 예방 및 치료용 조성물 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 12824322 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2845625 Country of ref document: CA |
|
| ENP | Entry into the national phase |
Ref document number: 2014525911 Country of ref document: JP Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| REEP | Request for entry into the european phase |
Ref document number: 2012824322 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2012824322 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 2012295779 Country of ref document: AU Date of ref document: 20120516 Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 14239521 Country of ref document: US |